User login
Primary Prevention of Diabetic Kidney Disease: Thumbs Up/Down
LAS VEGAS – Contrary to conventional wisdom, neither ACE inhibitors nor angiotensin receptor blockers have any role to play in primary prevention of diabetic kidney disease, according to Dr. Robert C. Stanton, chief of nephrology at the Harvard University’s Joslin Diabetes Center, Boston.
"I don’t see any unique indication for ACE inhibitors and ARBs for the primary prevention of kidney disease in diabetic patients, especially given that around 70% of diabetes patients will never develop kidney disease. They’re perfectly fine blood pressure pills. But as a magic kidney disease prevention drug, I don’t see any evidence for that. Of course, patients with proteinuria are another issue entirely. Those drugs absolutely are beneficial in that setting," he said at a meeting sponsored by the National Kidney Foundation.
When Dr. Stanton polled his audience electronically during the course of his talk, however, the majority of physicians indicated that they believe ACE inhibitors and ARBs are indeed useful for primary prevention of diabetic kidney disease. The evidence, Dr. Stanton emphasized, shows otherwise.
For example, a well-conducted, randomized, multicenter, placebo-controlled, 5-year clinical trial showed no benefit for enalapril or losartan in preventing kidney disease in patients with type 1 diabetes (N. Engl. J. Med. 2009;361:40-51). And three randomized controlled trials showed no primary preventive benefit for candesartan in more than 5,000 patients with type 1 or type 2 diabetes (Ann. Intern. Med. 2009;151:11-20).
Dr. Stanton noted that lots of other interventions have been proposed for the primary prevention of kidney disease in diabetes patients. Some are supported by solid evidence of benefit, others are not.
Here is his view of the preventive landscape:
• Intensive blood glucose control. "This is the easy one," he said. "A lot of us in the diabetes world feel that a hemoglobin A1c of 7% is the appropriate target for preventing many complications. It’s a reasonable target and should be achieved whether you’re talking about type 1 or type 2 patients."
The nephrologist noted that recent 25-year follow-up data from the landmark Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications study showed that fully 18 years after the intervention ended, patients assigned to intensive blood glucose control still showed highly impressive 50% reductions in the cumulative incidence of both microalbuminuria and end-stage renal disease compared with patients placed on less intensive control (Diabetes Care 2014;37:24-30).
• Smoking cessation. Smoking has been linked to a several-fold increased risk of diabetic kidney disease. "I think of diabetes as an endothelial cell disease, and smoking is the greatest endothelial cell poison we’ve come up with. So stopping smoking is something well worth doing," Dr. Stanton said.
• Blood pressure control. No question exists regarding its renoprotective effect. But recent guidelines are dizzyingly all over the map in terms of target pressure recommendations.
"I’m getting a major headache reading these articles right now. I can show you the data. Good luck! I personally like a target of 130/80 mm Hg or less, particularly when it’s not that hard to get there. But I’d let you decide what particular target you favor," he said.
He prefers 130/80 mm Hg as a target blood pressure for primary prevention of diabetic kidney disease in large part because of a meta-analysis showing that it was associated with a 10% reduction in the risk of developing microalbuminuria and an 11% decrease in end-stage renal disease (PloS Med 2012;9(8):e1001293).
• Weight loss. The growing bariatric surgery literature supports weight loss as a primary preventive strategy.
• Protein intake. There is no role for a low-protein diet – say, less than 0.8 g/kg per day – for primary prevention of kidney disease in diabetes patients. And Dr. Stanton believes a high-protein diet in the range of more than 1.5 or 2 g/kg per day is best avoided in patients with diabetes, although he stressed that the evidence on this score remains sketchy.
Still, "I would not go on a body-building diet or an Atkins-type diet," he cautioned.
• Targeting glomerular hyperfiltration. Studies have shown conflicting results. "For me, there’s no clear role for targeting hyperfiltration," said Dr. Stanton, who cited a comprehensive review that he finds persuasive (Diabetologia 2010;53:2093-104).
The key to developing more effective primary prevention strategies, according to Dr. Stanton, will be first to establish markers that clearly identify the 30% or so of diabetes patients who will go on to develop renal disease, then test novel interventions specifically in that high-risk group.
Promising biomarkers include circulating tumor necrosis factor alpha receptor levels, von Willebrand factor, monocyte chemoattractant factor, asymmetrical dimethylarginine, interleukin-6 and -8, and Fas receptor.
For example, one study showed that patients with type 2 diabetes in the top quartile for circulating TNF receptor 1 had a cumulative 12-year incidence of end-stage renal disease of 54%, compared to just 3% in patients in the other quartiles (J. Am. Soc. Nephrol. 2012;23:507-15).
"Lots of companies are looking at these now. These markers may be coming our way as indicators of people with diabetes who are likely to progress to kidney disease," Dr. Stanton said.
He reported serving as a consultant to Boehringer Ingelheim.
LAS VEGAS – Contrary to conventional wisdom, neither ACE inhibitors nor angiotensin receptor blockers have any role to play in primary prevention of diabetic kidney disease, according to Dr. Robert C. Stanton, chief of nephrology at the Harvard University’s Joslin Diabetes Center, Boston.
"I don’t see any unique indication for ACE inhibitors and ARBs for the primary prevention of kidney disease in diabetic patients, especially given that around 70% of diabetes patients will never develop kidney disease. They’re perfectly fine blood pressure pills. But as a magic kidney disease prevention drug, I don’t see any evidence for that. Of course, patients with proteinuria are another issue entirely. Those drugs absolutely are beneficial in that setting," he said at a meeting sponsored by the National Kidney Foundation.
When Dr. Stanton polled his audience electronically during the course of his talk, however, the majority of physicians indicated that they believe ACE inhibitors and ARBs are indeed useful for primary prevention of diabetic kidney disease. The evidence, Dr. Stanton emphasized, shows otherwise.
For example, a well-conducted, randomized, multicenter, placebo-controlled, 5-year clinical trial showed no benefit for enalapril or losartan in preventing kidney disease in patients with type 1 diabetes (N. Engl. J. Med. 2009;361:40-51). And three randomized controlled trials showed no primary preventive benefit for candesartan in more than 5,000 patients with type 1 or type 2 diabetes (Ann. Intern. Med. 2009;151:11-20).
Dr. Stanton noted that lots of other interventions have been proposed for the primary prevention of kidney disease in diabetes patients. Some are supported by solid evidence of benefit, others are not.
Here is his view of the preventive landscape:
• Intensive blood glucose control. "This is the easy one," he said. "A lot of us in the diabetes world feel that a hemoglobin A1c of 7% is the appropriate target for preventing many complications. It’s a reasonable target and should be achieved whether you’re talking about type 1 or type 2 patients."
The nephrologist noted that recent 25-year follow-up data from the landmark Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications study showed that fully 18 years after the intervention ended, patients assigned to intensive blood glucose control still showed highly impressive 50% reductions in the cumulative incidence of both microalbuminuria and end-stage renal disease compared with patients placed on less intensive control (Diabetes Care 2014;37:24-30).
• Smoking cessation. Smoking has been linked to a several-fold increased risk of diabetic kidney disease. "I think of diabetes as an endothelial cell disease, and smoking is the greatest endothelial cell poison we’ve come up with. So stopping smoking is something well worth doing," Dr. Stanton said.
• Blood pressure control. No question exists regarding its renoprotective effect. But recent guidelines are dizzyingly all over the map in terms of target pressure recommendations.
"I’m getting a major headache reading these articles right now. I can show you the data. Good luck! I personally like a target of 130/80 mm Hg or less, particularly when it’s not that hard to get there. But I’d let you decide what particular target you favor," he said.
He prefers 130/80 mm Hg as a target blood pressure for primary prevention of diabetic kidney disease in large part because of a meta-analysis showing that it was associated with a 10% reduction in the risk of developing microalbuminuria and an 11% decrease in end-stage renal disease (PloS Med 2012;9(8):e1001293).
• Weight loss. The growing bariatric surgery literature supports weight loss as a primary preventive strategy.
• Protein intake. There is no role for a low-protein diet – say, less than 0.8 g/kg per day – for primary prevention of kidney disease in diabetes patients. And Dr. Stanton believes a high-protein diet in the range of more than 1.5 or 2 g/kg per day is best avoided in patients with diabetes, although he stressed that the evidence on this score remains sketchy.
Still, "I would not go on a body-building diet or an Atkins-type diet," he cautioned.
• Targeting glomerular hyperfiltration. Studies have shown conflicting results. "For me, there’s no clear role for targeting hyperfiltration," said Dr. Stanton, who cited a comprehensive review that he finds persuasive (Diabetologia 2010;53:2093-104).
The key to developing more effective primary prevention strategies, according to Dr. Stanton, will be first to establish markers that clearly identify the 30% or so of diabetes patients who will go on to develop renal disease, then test novel interventions specifically in that high-risk group.
Promising biomarkers include circulating tumor necrosis factor alpha receptor levels, von Willebrand factor, monocyte chemoattractant factor, asymmetrical dimethylarginine, interleukin-6 and -8, and Fas receptor.
For example, one study showed that patients with type 2 diabetes in the top quartile for circulating TNF receptor 1 had a cumulative 12-year incidence of end-stage renal disease of 54%, compared to just 3% in patients in the other quartiles (J. Am. Soc. Nephrol. 2012;23:507-15).
"Lots of companies are looking at these now. These markers may be coming our way as indicators of people with diabetes who are likely to progress to kidney disease," Dr. Stanton said.
He reported serving as a consultant to Boehringer Ingelheim.
LAS VEGAS – Contrary to conventional wisdom, neither ACE inhibitors nor angiotensin receptor blockers have any role to play in primary prevention of diabetic kidney disease, according to Dr. Robert C. Stanton, chief of nephrology at the Harvard University’s Joslin Diabetes Center, Boston.
"I don’t see any unique indication for ACE inhibitors and ARBs for the primary prevention of kidney disease in diabetic patients, especially given that around 70% of diabetes patients will never develop kidney disease. They’re perfectly fine blood pressure pills. But as a magic kidney disease prevention drug, I don’t see any evidence for that. Of course, patients with proteinuria are another issue entirely. Those drugs absolutely are beneficial in that setting," he said at a meeting sponsored by the National Kidney Foundation.
When Dr. Stanton polled his audience electronically during the course of his talk, however, the majority of physicians indicated that they believe ACE inhibitors and ARBs are indeed useful for primary prevention of diabetic kidney disease. The evidence, Dr. Stanton emphasized, shows otherwise.
For example, a well-conducted, randomized, multicenter, placebo-controlled, 5-year clinical trial showed no benefit for enalapril or losartan in preventing kidney disease in patients with type 1 diabetes (N. Engl. J. Med. 2009;361:40-51). And three randomized controlled trials showed no primary preventive benefit for candesartan in more than 5,000 patients with type 1 or type 2 diabetes (Ann. Intern. Med. 2009;151:11-20).
Dr. Stanton noted that lots of other interventions have been proposed for the primary prevention of kidney disease in diabetes patients. Some are supported by solid evidence of benefit, others are not.
Here is his view of the preventive landscape:
• Intensive blood glucose control. "This is the easy one," he said. "A lot of us in the diabetes world feel that a hemoglobin A1c of 7% is the appropriate target for preventing many complications. It’s a reasonable target and should be achieved whether you’re talking about type 1 or type 2 patients."
The nephrologist noted that recent 25-year follow-up data from the landmark Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications study showed that fully 18 years after the intervention ended, patients assigned to intensive blood glucose control still showed highly impressive 50% reductions in the cumulative incidence of both microalbuminuria and end-stage renal disease compared with patients placed on less intensive control (Diabetes Care 2014;37:24-30).
• Smoking cessation. Smoking has been linked to a several-fold increased risk of diabetic kidney disease. "I think of diabetes as an endothelial cell disease, and smoking is the greatest endothelial cell poison we’ve come up with. So stopping smoking is something well worth doing," Dr. Stanton said.
• Blood pressure control. No question exists regarding its renoprotective effect. But recent guidelines are dizzyingly all over the map in terms of target pressure recommendations.
"I’m getting a major headache reading these articles right now. I can show you the data. Good luck! I personally like a target of 130/80 mm Hg or less, particularly when it’s not that hard to get there. But I’d let you decide what particular target you favor," he said.
He prefers 130/80 mm Hg as a target blood pressure for primary prevention of diabetic kidney disease in large part because of a meta-analysis showing that it was associated with a 10% reduction in the risk of developing microalbuminuria and an 11% decrease in end-stage renal disease (PloS Med 2012;9(8):e1001293).
• Weight loss. The growing bariatric surgery literature supports weight loss as a primary preventive strategy.
• Protein intake. There is no role for a low-protein diet – say, less than 0.8 g/kg per day – for primary prevention of kidney disease in diabetes patients. And Dr. Stanton believes a high-protein diet in the range of more than 1.5 or 2 g/kg per day is best avoided in patients with diabetes, although he stressed that the evidence on this score remains sketchy.
Still, "I would not go on a body-building diet or an Atkins-type diet," he cautioned.
• Targeting glomerular hyperfiltration. Studies have shown conflicting results. "For me, there’s no clear role for targeting hyperfiltration," said Dr. Stanton, who cited a comprehensive review that he finds persuasive (Diabetologia 2010;53:2093-104).
The key to developing more effective primary prevention strategies, according to Dr. Stanton, will be first to establish markers that clearly identify the 30% or so of diabetes patients who will go on to develop renal disease, then test novel interventions specifically in that high-risk group.
Promising biomarkers include circulating tumor necrosis factor alpha receptor levels, von Willebrand factor, monocyte chemoattractant factor, asymmetrical dimethylarginine, interleukin-6 and -8, and Fas receptor.
For example, one study showed that patients with type 2 diabetes in the top quartile for circulating TNF receptor 1 had a cumulative 12-year incidence of end-stage renal disease of 54%, compared to just 3% in patients in the other quartiles (J. Am. Soc. Nephrol. 2012;23:507-15).
"Lots of companies are looking at these now. These markers may be coming our way as indicators of people with diabetes who are likely to progress to kidney disease," Dr. Stanton said.
He reported serving as a consultant to Boehringer Ingelheim.
EXPERT ANALYSIS FROM SCM 14
New Clinical Practice Guidelines on Pheochromocytomas
CHICAGO – Genetic testing has jumped to the fore in the management of patients diagnosed as having a pheochromocytoma or paraganglioma, according to new clinical practice guidelines released by the Endocrine Society.
Indeed, the new guidelines call for genetic testing to be considered seriously in all patients with a proven pheochromocytoma or paraganglioma (PPGL), Dr. Jacques W. M. Lenders said in presenting highlights of the new guidelines at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
"We recommend that all patients with PPGLs should be engaged in shared decision making for genetic testing. I don’t say that we should do genetic testing in everybody, but we should consider it and engage the patient in the final decision," said Dr. Lenders, who chaired the practice guidelines task force.
The strong emphasis on genetic testing arises from evidence that roughly one-third of all PPGLs are associated with germline mutations. Moreover, susceptibility mutations are present in 12% of patients with absolutely no suggestion of a positive family history. Some of these mutations – for example, those involving succinate dehydrogenase B (SDHB) – are associated with a high risk of metastasis and unfavorable prognosis. Thus, gene-testing results can have a major impact on patients with PPGL as well as their relatives.
Nonetheless, genetic testing in patients with PPGLs remains controversial.
"I must say, we on the guideline task force spent considerable time on what and how to do it," said Dr. Lenders, who is professor and deputy chair of internal medicine at Radboud University in Nijmegen, the Netherlands.
Since simultaneous testing for all the known culprit genes remains for now too expensive to be cost effective, the guidelines include a clinical feature–driven decisional algorithm designed to establish the priorities for genetic testing in a given patient with proven PPGL.
For example, patients with a metastatic PPGL should be tested for SDHB mutations, while those with a paraganglioma should undergo testing for succinate dehydrogenase mutations, according to the guidelines, published in full in concert with ICE/ENDO 2014 (J. Clin. Endocrinol. Metab. 2014;1915-42).
Dr. Lenders noted that PPGLs are uncommon tumors. It is estimated that 0.1%-1% of patients being treated for hypertension have pheochromocytomas, which are adrenal tumors resulting in excess production of epinephrine and norepinephrine. Symptoms can include paroxysmal severe headache, tachycardia, anxiety, and excessive sweating, along with tough-to-control hypertension.
While pheochromocytomas are typically benign, malignant transformation occurs in up to 17% of cases. And although a complete cure is often possible with timely therapy, the fact is that on average a 3-year delay transpires between symptomatic presentation and diagnosis of PPGL. Also, studies show that failure to appropriately follow up on a positive biochemical test is common in clinical practice; as a consequence, PPGLs are often overdiagnosed. For these reasons, Endocrine Society officials deemed PPGLs a priority area in need of practice guidelines.
In addition to routine consideration of genetic testing, other recommendations include:
• Diagnostic biochemical testing: Initial testing should include measurement of plasma free or urinary fractionated metanephrines, preferably using liquid chromatography with electrochemical or mass spectrometric laboratory methods. Immunoassays, although popular in Europe, haven’t yet been adequately validated. In measuring plasma metanephrines, the blood draw should be done with the patient in supine position, using reference standards established in the same position.
"False-positive test results are a major problem in daily clinical practice, and they outweigh by far the number of true-positive test results. That’s very important to realize," the endocrinologist said.
One common cause of false-positive test results are medications that trigger elevated metanephrine levels, according to guideline panelist Dr. William F. Young Jr., professor of medicine and chair of the department of endocrinology, diabetes, metabolism and nutrition at the Mayo Clinic, Rochester, Minn. The top three offending drugs in his experience are tricyclic antidepressants, antipsychotic agents, and levodopa. The guidelines list others, he added.
• Imaging: Once clear biochemical evidence of a PPGL is established, CT is preferred over MRI in order to locate the tumor because of its superior spatial resolution in the thorax, abdomen, and pelvis. 18F-fluorodeoxyglucose positron emission tomography/CT scanning is preferred over 123I-metaiodobenzylguanidine (MIBG) scintigraphy in patients with known metastatic PPGL. 123I-MIBG is best reserved for functional imaging in patients with metastatic PPGL who are being considered for radiotherapy using 131I-MIBG, in patients with an unusually large primary tumor, and in other special circumstances.
• Perioperative medical management: Preoperative blockade with an alpha-adrenergic–receptor blocker beginning 7-14 days before surgery is recommended together with a high-sodium diet and increased fluid intake as the best means of reducing the risk of perioperative cardiovascular problems.
• Surgery: Minimally invasive adrenalectomy is appropriate for most pheochromocytomas; open resection is best reserved for those tumors which are invasive or greater than 6 cm in size. The guidelines recommend open resection for paragangliomas, although laparoscopic surgery is described as reasonable for those which are small, noninvasive, and favorably located. Partial adrenalectomy is advised for patients with a hereditary pheochromocytoma and in other special circumstances.
• Team approach: Because PPGLs are uncommon, they are best managed by multidisciplinary teams at centers of expertise. That’s particularly important in nonstraightforward cases, such as those involving pregnancy, metastasis, diagnostic uncertainty, or surgical complexity, according to the guideline panelists.
All Endocrine Society clinical practice guidelines are funded by the society without any corporate support. Dr. Lenders reported having no financial conflicts.
CHICAGO – Genetic testing has jumped to the fore in the management of patients diagnosed as having a pheochromocytoma or paraganglioma, according to new clinical practice guidelines released by the Endocrine Society.
Indeed, the new guidelines call for genetic testing to be considered seriously in all patients with a proven pheochromocytoma or paraganglioma (PPGL), Dr. Jacques W. M. Lenders said in presenting highlights of the new guidelines at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
"We recommend that all patients with PPGLs should be engaged in shared decision making for genetic testing. I don’t say that we should do genetic testing in everybody, but we should consider it and engage the patient in the final decision," said Dr. Lenders, who chaired the practice guidelines task force.
The strong emphasis on genetic testing arises from evidence that roughly one-third of all PPGLs are associated with germline mutations. Moreover, susceptibility mutations are present in 12% of patients with absolutely no suggestion of a positive family history. Some of these mutations – for example, those involving succinate dehydrogenase B (SDHB) – are associated with a high risk of metastasis and unfavorable prognosis. Thus, gene-testing results can have a major impact on patients with PPGL as well as their relatives.
Nonetheless, genetic testing in patients with PPGLs remains controversial.
"I must say, we on the guideline task force spent considerable time on what and how to do it," said Dr. Lenders, who is professor and deputy chair of internal medicine at Radboud University in Nijmegen, the Netherlands.
Since simultaneous testing for all the known culprit genes remains for now too expensive to be cost effective, the guidelines include a clinical feature–driven decisional algorithm designed to establish the priorities for genetic testing in a given patient with proven PPGL.
For example, patients with a metastatic PPGL should be tested for SDHB mutations, while those with a paraganglioma should undergo testing for succinate dehydrogenase mutations, according to the guidelines, published in full in concert with ICE/ENDO 2014 (J. Clin. Endocrinol. Metab. 2014;1915-42).
Dr. Lenders noted that PPGLs are uncommon tumors. It is estimated that 0.1%-1% of patients being treated for hypertension have pheochromocytomas, which are adrenal tumors resulting in excess production of epinephrine and norepinephrine. Symptoms can include paroxysmal severe headache, tachycardia, anxiety, and excessive sweating, along with tough-to-control hypertension.
While pheochromocytomas are typically benign, malignant transformation occurs in up to 17% of cases. And although a complete cure is often possible with timely therapy, the fact is that on average a 3-year delay transpires between symptomatic presentation and diagnosis of PPGL. Also, studies show that failure to appropriately follow up on a positive biochemical test is common in clinical practice; as a consequence, PPGLs are often overdiagnosed. For these reasons, Endocrine Society officials deemed PPGLs a priority area in need of practice guidelines.
In addition to routine consideration of genetic testing, other recommendations include:
• Diagnostic biochemical testing: Initial testing should include measurement of plasma free or urinary fractionated metanephrines, preferably using liquid chromatography with electrochemical or mass spectrometric laboratory methods. Immunoassays, although popular in Europe, haven’t yet been adequately validated. In measuring plasma metanephrines, the blood draw should be done with the patient in supine position, using reference standards established in the same position.
"False-positive test results are a major problem in daily clinical practice, and they outweigh by far the number of true-positive test results. That’s very important to realize," the endocrinologist said.
One common cause of false-positive test results are medications that trigger elevated metanephrine levels, according to guideline panelist Dr. William F. Young Jr., professor of medicine and chair of the department of endocrinology, diabetes, metabolism and nutrition at the Mayo Clinic, Rochester, Minn. The top three offending drugs in his experience are tricyclic antidepressants, antipsychotic agents, and levodopa. The guidelines list others, he added.
• Imaging: Once clear biochemical evidence of a PPGL is established, CT is preferred over MRI in order to locate the tumor because of its superior spatial resolution in the thorax, abdomen, and pelvis. 18F-fluorodeoxyglucose positron emission tomography/CT scanning is preferred over 123I-metaiodobenzylguanidine (MIBG) scintigraphy in patients with known metastatic PPGL. 123I-MIBG is best reserved for functional imaging in patients with metastatic PPGL who are being considered for radiotherapy using 131I-MIBG, in patients with an unusually large primary tumor, and in other special circumstances.
• Perioperative medical management: Preoperative blockade with an alpha-adrenergic–receptor blocker beginning 7-14 days before surgery is recommended together with a high-sodium diet and increased fluid intake as the best means of reducing the risk of perioperative cardiovascular problems.
• Surgery: Minimally invasive adrenalectomy is appropriate for most pheochromocytomas; open resection is best reserved for those tumors which are invasive or greater than 6 cm in size. The guidelines recommend open resection for paragangliomas, although laparoscopic surgery is described as reasonable for those which are small, noninvasive, and favorably located. Partial adrenalectomy is advised for patients with a hereditary pheochromocytoma and in other special circumstances.
• Team approach: Because PPGLs are uncommon, they are best managed by multidisciplinary teams at centers of expertise. That’s particularly important in nonstraightforward cases, such as those involving pregnancy, metastasis, diagnostic uncertainty, or surgical complexity, according to the guideline panelists.
All Endocrine Society clinical practice guidelines are funded by the society without any corporate support. Dr. Lenders reported having no financial conflicts.
CHICAGO – Genetic testing has jumped to the fore in the management of patients diagnosed as having a pheochromocytoma or paraganglioma, according to new clinical practice guidelines released by the Endocrine Society.
Indeed, the new guidelines call for genetic testing to be considered seriously in all patients with a proven pheochromocytoma or paraganglioma (PPGL), Dr. Jacques W. M. Lenders said in presenting highlights of the new guidelines at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
"We recommend that all patients with PPGLs should be engaged in shared decision making for genetic testing. I don’t say that we should do genetic testing in everybody, but we should consider it and engage the patient in the final decision," said Dr. Lenders, who chaired the practice guidelines task force.
The strong emphasis on genetic testing arises from evidence that roughly one-third of all PPGLs are associated with germline mutations. Moreover, susceptibility mutations are present in 12% of patients with absolutely no suggestion of a positive family history. Some of these mutations – for example, those involving succinate dehydrogenase B (SDHB) – are associated with a high risk of metastasis and unfavorable prognosis. Thus, gene-testing results can have a major impact on patients with PPGL as well as their relatives.
Nonetheless, genetic testing in patients with PPGLs remains controversial.
"I must say, we on the guideline task force spent considerable time on what and how to do it," said Dr. Lenders, who is professor and deputy chair of internal medicine at Radboud University in Nijmegen, the Netherlands.
Since simultaneous testing for all the known culprit genes remains for now too expensive to be cost effective, the guidelines include a clinical feature–driven decisional algorithm designed to establish the priorities for genetic testing in a given patient with proven PPGL.
For example, patients with a metastatic PPGL should be tested for SDHB mutations, while those with a paraganglioma should undergo testing for succinate dehydrogenase mutations, according to the guidelines, published in full in concert with ICE/ENDO 2014 (J. Clin. Endocrinol. Metab. 2014;1915-42).
Dr. Lenders noted that PPGLs are uncommon tumors. It is estimated that 0.1%-1% of patients being treated for hypertension have pheochromocytomas, which are adrenal tumors resulting in excess production of epinephrine and norepinephrine. Symptoms can include paroxysmal severe headache, tachycardia, anxiety, and excessive sweating, along with tough-to-control hypertension.
While pheochromocytomas are typically benign, malignant transformation occurs in up to 17% of cases. And although a complete cure is often possible with timely therapy, the fact is that on average a 3-year delay transpires between symptomatic presentation and diagnosis of PPGL. Also, studies show that failure to appropriately follow up on a positive biochemical test is common in clinical practice; as a consequence, PPGLs are often overdiagnosed. For these reasons, Endocrine Society officials deemed PPGLs a priority area in need of practice guidelines.
In addition to routine consideration of genetic testing, other recommendations include:
• Diagnostic biochemical testing: Initial testing should include measurement of plasma free or urinary fractionated metanephrines, preferably using liquid chromatography with electrochemical or mass spectrometric laboratory methods. Immunoassays, although popular in Europe, haven’t yet been adequately validated. In measuring plasma metanephrines, the blood draw should be done with the patient in supine position, using reference standards established in the same position.
"False-positive test results are a major problem in daily clinical practice, and they outweigh by far the number of true-positive test results. That’s very important to realize," the endocrinologist said.
One common cause of false-positive test results are medications that trigger elevated metanephrine levels, according to guideline panelist Dr. William F. Young Jr., professor of medicine and chair of the department of endocrinology, diabetes, metabolism and nutrition at the Mayo Clinic, Rochester, Minn. The top three offending drugs in his experience are tricyclic antidepressants, antipsychotic agents, and levodopa. The guidelines list others, he added.
• Imaging: Once clear biochemical evidence of a PPGL is established, CT is preferred over MRI in order to locate the tumor because of its superior spatial resolution in the thorax, abdomen, and pelvis. 18F-fluorodeoxyglucose positron emission tomography/CT scanning is preferred over 123I-metaiodobenzylguanidine (MIBG) scintigraphy in patients with known metastatic PPGL. 123I-MIBG is best reserved for functional imaging in patients with metastatic PPGL who are being considered for radiotherapy using 131I-MIBG, in patients with an unusually large primary tumor, and in other special circumstances.
• Perioperative medical management: Preoperative blockade with an alpha-adrenergic–receptor blocker beginning 7-14 days before surgery is recommended together with a high-sodium diet and increased fluid intake as the best means of reducing the risk of perioperative cardiovascular problems.
• Surgery: Minimally invasive adrenalectomy is appropriate for most pheochromocytomas; open resection is best reserved for those tumors which are invasive or greater than 6 cm in size. The guidelines recommend open resection for paragangliomas, although laparoscopic surgery is described as reasonable for those which are small, noninvasive, and favorably located. Partial adrenalectomy is advised for patients with a hereditary pheochromocytoma and in other special circumstances.
• Team approach: Because PPGLs are uncommon, they are best managed by multidisciplinary teams at centers of expertise. That’s particularly important in nonstraightforward cases, such as those involving pregnancy, metastasis, diagnostic uncertainty, or surgical complexity, according to the guideline panelists.
All Endocrine Society clinical practice guidelines are funded by the society without any corporate support. Dr. Lenders reported having no financial conflicts.
AT ICE/ENDO 2014
Think methotrexate for juvenile localized scleroderma
COEUR D’ALENE, IDAHO – Long-term use of methotrexate has a lot going for it as first-line therapy for active juvenile localized scleroderma, according to Dr. Francesco Zulian, chief of pediatric rheumatology at the University of Padua (Italy).
"It’s a drug that’s very old, it’s not expensive, it’s used in many dermatologic and rheumatologic conditions – and it is very useful in patients with scleroderma," he observed in his Sidney Hurwitz Memorial Lecture at the annual meeting of the Society for Pediatric Dermatology.
The initial studies of methotrexate in scleroderma were conducted in adults. Dr. Zulian and colleagues are credited with performing the first randomized, double-blind, prospective clinical trial in pediatric patients, building upon other investigators’ favorable earlier nonrandomized results.
Based upon the randomized trial findings and the subsequent long-term follow-up study, his recommendation for patients with active juvenile localized scleroderma – whether of the linear, pansclerotic, or generalized morphea subtype – is 3 months of initial bridging therapy with a combination of methotrexate plus systemic corticosteroids, followed by at least 24 months of methotrexate without systemic steroids.
In the long-term follow-up study involving 65 patients, treatment was associated with a 74% clinical remission rate. This broke down as approximately a 54% complete remission rate maintained for at least 6 months without treatment, and a 20% clinical remission rate on treatment. Treatment for less than 24 months yielded lesser long-term benefit. Adverse effects were seen in nearly half of patients; however, they were typically mild, and no patients discontinued treatment as a result (J. Am. Acad. Dermatol. 2012;67:1151-6).
"This study shows there is a large group of patients who get better with a relatively mild treatment," Dr. Zulian noted.
The bridging therapy regimen employed in the landmark double-blind, placebo-controlled trial involved oral methotrexate at 15 mg/m2 or a maximum of 20 mg per week, along with prednisone at 1 mg/kg/day or a maximum of 50 mg daily for 3 months (Arthritis Rheum. 2011;63:1998-2006).
In his own clinical practice, Dr. Zulian said, he turns to mycophenolate mofetil (CellCept) in the minority of cases in which bridging therapy with methotrexate and prednisone proves inadequate.
In patients with circumscribed morphea as defined in the international Padua consensus conference guidelines, subsequently formalized by the American College of Rheumatology, the European League Against Rheumatism, and the Pediatric Rheumatism European Society (Arthritis Rheum. 2007;203-12), his recommended treatment is topical corticosteroids, calcipotriol, or phototherapy.
While scleroderma is a rare condition in children, it is nonetheless the third most frequent condition within pediatric rheumatology. For physicians with affected patients who are interested in collaborative research to advance the treatment and understanding of this disease, Dr. Zulian recommended contacting the Juvenile Scleroderma International Network (www.jusinet.org).
Dr. Zulian reported having no financial conflicts with regard to his presentation.
COEUR D’ALENE, IDAHO – Long-term use of methotrexate has a lot going for it as first-line therapy for active juvenile localized scleroderma, according to Dr. Francesco Zulian, chief of pediatric rheumatology at the University of Padua (Italy).
"It’s a drug that’s very old, it’s not expensive, it’s used in many dermatologic and rheumatologic conditions – and it is very useful in patients with scleroderma," he observed in his Sidney Hurwitz Memorial Lecture at the annual meeting of the Society for Pediatric Dermatology.
The initial studies of methotrexate in scleroderma were conducted in adults. Dr. Zulian and colleagues are credited with performing the first randomized, double-blind, prospective clinical trial in pediatric patients, building upon other investigators’ favorable earlier nonrandomized results.
Based upon the randomized trial findings and the subsequent long-term follow-up study, his recommendation for patients with active juvenile localized scleroderma – whether of the linear, pansclerotic, or generalized morphea subtype – is 3 months of initial bridging therapy with a combination of methotrexate plus systemic corticosteroids, followed by at least 24 months of methotrexate without systemic steroids.
In the long-term follow-up study involving 65 patients, treatment was associated with a 74% clinical remission rate. This broke down as approximately a 54% complete remission rate maintained for at least 6 months without treatment, and a 20% clinical remission rate on treatment. Treatment for less than 24 months yielded lesser long-term benefit. Adverse effects were seen in nearly half of patients; however, they were typically mild, and no patients discontinued treatment as a result (J. Am. Acad. Dermatol. 2012;67:1151-6).
"This study shows there is a large group of patients who get better with a relatively mild treatment," Dr. Zulian noted.
The bridging therapy regimen employed in the landmark double-blind, placebo-controlled trial involved oral methotrexate at 15 mg/m2 or a maximum of 20 mg per week, along with prednisone at 1 mg/kg/day or a maximum of 50 mg daily for 3 months (Arthritis Rheum. 2011;63:1998-2006).
In his own clinical practice, Dr. Zulian said, he turns to mycophenolate mofetil (CellCept) in the minority of cases in which bridging therapy with methotrexate and prednisone proves inadequate.
In patients with circumscribed morphea as defined in the international Padua consensus conference guidelines, subsequently formalized by the American College of Rheumatology, the European League Against Rheumatism, and the Pediatric Rheumatism European Society (Arthritis Rheum. 2007;203-12), his recommended treatment is topical corticosteroids, calcipotriol, or phototherapy.
While scleroderma is a rare condition in children, it is nonetheless the third most frequent condition within pediatric rheumatology. For physicians with affected patients who are interested in collaborative research to advance the treatment and understanding of this disease, Dr. Zulian recommended contacting the Juvenile Scleroderma International Network (www.jusinet.org).
Dr. Zulian reported having no financial conflicts with regard to his presentation.
COEUR D’ALENE, IDAHO – Long-term use of methotrexate has a lot going for it as first-line therapy for active juvenile localized scleroderma, according to Dr. Francesco Zulian, chief of pediatric rheumatology at the University of Padua (Italy).
"It’s a drug that’s very old, it’s not expensive, it’s used in many dermatologic and rheumatologic conditions – and it is very useful in patients with scleroderma," he observed in his Sidney Hurwitz Memorial Lecture at the annual meeting of the Society for Pediatric Dermatology.
The initial studies of methotrexate in scleroderma were conducted in adults. Dr. Zulian and colleagues are credited with performing the first randomized, double-blind, prospective clinical trial in pediatric patients, building upon other investigators’ favorable earlier nonrandomized results.
Based upon the randomized trial findings and the subsequent long-term follow-up study, his recommendation for patients with active juvenile localized scleroderma – whether of the linear, pansclerotic, or generalized morphea subtype – is 3 months of initial bridging therapy with a combination of methotrexate plus systemic corticosteroids, followed by at least 24 months of methotrexate without systemic steroids.
In the long-term follow-up study involving 65 patients, treatment was associated with a 74% clinical remission rate. This broke down as approximately a 54% complete remission rate maintained for at least 6 months without treatment, and a 20% clinical remission rate on treatment. Treatment for less than 24 months yielded lesser long-term benefit. Adverse effects were seen in nearly half of patients; however, they were typically mild, and no patients discontinued treatment as a result (J. Am. Acad. Dermatol. 2012;67:1151-6).
"This study shows there is a large group of patients who get better with a relatively mild treatment," Dr. Zulian noted.
The bridging therapy regimen employed in the landmark double-blind, placebo-controlled trial involved oral methotrexate at 15 mg/m2 or a maximum of 20 mg per week, along with prednisone at 1 mg/kg/day or a maximum of 50 mg daily for 3 months (Arthritis Rheum. 2011;63:1998-2006).
In his own clinical practice, Dr. Zulian said, he turns to mycophenolate mofetil (CellCept) in the minority of cases in which bridging therapy with methotrexate and prednisone proves inadequate.
In patients with circumscribed morphea as defined in the international Padua consensus conference guidelines, subsequently formalized by the American College of Rheumatology, the European League Against Rheumatism, and the Pediatric Rheumatism European Society (Arthritis Rheum. 2007;203-12), his recommended treatment is topical corticosteroids, calcipotriol, or phototherapy.
While scleroderma is a rare condition in children, it is nonetheless the third most frequent condition within pediatric rheumatology. For physicians with affected patients who are interested in collaborative research to advance the treatment and understanding of this disease, Dr. Zulian recommended contacting the Juvenile Scleroderma International Network (www.jusinet.org).
Dr. Zulian reported having no financial conflicts with regard to his presentation.
EXPERT ANALYSIS FROM THE SPD ANNUAL MEETING
Rhythm control protocol succeeds in recent onset atrial fibrillation
DALLAS – It’s not the rate; it’s the rhythm control that matters most for a selected subgroup of patients who present to the emergency department with recent onset atrial fibrillation.
The rhythm control approach, essentially the Ottawa Aggressive Protocol, uses intravenous procainamide as first-line therapy and, if the pharmacologic conversion is unsuccessful, subsequent electrical cardioversion by ED physicians. Patients converted to sinus rhythm are discharged home.
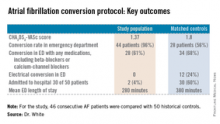
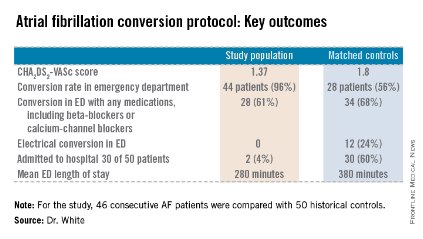
In a single-center prospective study, the rhythm control protocol proved safe, and 96% of recent-onset AF patients were converted in the ED. Among the historical controls, 56% were converted. Moreover, the rhythm control protocol cut ED lengths of stay, reduced hospital admissions, and proved highly popular with patients, who were returned to sinus rhythm and discharged home, noted Dr. Jennifer L. White, who presented the study results at the annual meeting of the Society for Academic Emergency Medicine.
"Is this study practice changing? It was for us. It’s our ED protocol now," said Dr. White, an emergency physician at Doylestown (Pa.) Hospital.
The Doylestown study included 46 consecutive patients who presented to the ED with atrial fibrillation (AF) of less than 48 hours duration. They were compared with 50 historical controls who met the same inclusion criteria and were treated before the ED rhythm protocol was implemented.
Dr. White said it took nearly 2 years to develop an ED protocol for AF conversion that was acceptable to cardiologists, nurses, and hospital administrators. The cardiologists dictated the exclusion criteria: No patients received the protocol if they had coronary artery disease, fever, concurrent ischemia, used medication that prolongs the QT interval, ejection fractions below 35%, hospital admission within the prior 3 months, and use of any antiarrhythmic agent within the past 72 hours. Patients with a history of TIA had to be on an oral anticoagulant. The baseline QTc could not exceed 460 msec.
Follow-up interviews conducted at 30 days showed no strokes or other serious adverse events, and a mean patient satisfaction score of 9.6 out of a possible 10. AF recurred in 4 of 46 patients during the 30-day period; 96% of patients were seen by a cardiologist within 14 days after ED discharge, as recommended by the ED staff.
The protocol put to the test in the ED in Doylestown entailed giving selected patients with recent-onset AF 1 g of procainamide intravenously. If patients converted to sinus rhythm, they were discharged. If not they were offered electrical cardioversion with moderate sedation using propofol, a procedure performed by two ED physicians. Patients who converted were sent home; for those who didn’t, cardiology was called in, and hospital admission usually followed.
"It took a while, honestly, to develop a protocol everyone felt comfortable with. We developed ED order sets for the nurses. There was a lot of education. But now when we say we’re going to cardiovert someone in the ED, no one seems to get uptight and upset about it," according to Dr. White.
The Ottawa Aggressive Protocol has been successfully used in Canada where "they’ve been converting patients in the ED and discharging them home with no bad outcomes. So why, then, are we in the United States admitting these patients for rate control?" Dr. White asked.
The protocol is in the process of being modified in response to the release of the 2014 American Heart Association/American College of Cardiology/Heart Rhythm Society guidelines for the management of patients with AF (J. Am. Coll. Cardiol. 2014 [doi:10.1016/j.acc.2014.03.022]).
Those guidelines recommend that all patients with AF and a CHA2DS2-VASc score of 2 or more be placed on an oral anticoagulant before or immediately following cardioversion.
Dr. White remarked that "personally, I would love to see this happen because I think our exclusion criteria are complicated and not reproducible. My thought is we should (instead) ask these three questions:
• Is the atrial fibrillation of recent onset and the primary diagnosis?
• Is the patient at CHA2DS2-VASc of 0-1 without any significant valvular disease?
• Is the QTc interval less than 460 msec with no other arrhythmia?
If the answer to all three questions is ‘yes,’ then we use ED rhythm control. If not, we consult cardiology," she said.
Dr. White reported having no financial conflicts regarding this study.
DALLAS – It’s not the rate; it’s the rhythm control that matters most for a selected subgroup of patients who present to the emergency department with recent onset atrial fibrillation.
The rhythm control approach, essentially the Ottawa Aggressive Protocol, uses intravenous procainamide as first-line therapy and, if the pharmacologic conversion is unsuccessful, subsequent electrical cardioversion by ED physicians. Patients converted to sinus rhythm are discharged home.
In a single-center prospective study, the rhythm control protocol proved safe, and 96% of recent-onset AF patients were converted in the ED. Among the historical controls, 56% were converted. Moreover, the rhythm control protocol cut ED lengths of stay, reduced hospital admissions, and proved highly popular with patients, who were returned to sinus rhythm and discharged home, noted Dr. Jennifer L. White, who presented the study results at the annual meeting of the Society for Academic Emergency Medicine.
"Is this study practice changing? It was for us. It’s our ED protocol now," said Dr. White, an emergency physician at Doylestown (Pa.) Hospital.
The Doylestown study included 46 consecutive patients who presented to the ED with atrial fibrillation (AF) of less than 48 hours duration. They were compared with 50 historical controls who met the same inclusion criteria and were treated before the ED rhythm protocol was implemented.
Dr. White said it took nearly 2 years to develop an ED protocol for AF conversion that was acceptable to cardiologists, nurses, and hospital administrators. The cardiologists dictated the exclusion criteria: No patients received the protocol if they had coronary artery disease, fever, concurrent ischemia, used medication that prolongs the QT interval, ejection fractions below 35%, hospital admission within the prior 3 months, and use of any antiarrhythmic agent within the past 72 hours. Patients with a history of TIA had to be on an oral anticoagulant. The baseline QTc could not exceed 460 msec.
Follow-up interviews conducted at 30 days showed no strokes or other serious adverse events, and a mean patient satisfaction score of 9.6 out of a possible 10. AF recurred in 4 of 46 patients during the 30-day period; 96% of patients were seen by a cardiologist within 14 days after ED discharge, as recommended by the ED staff.
The protocol put to the test in the ED in Doylestown entailed giving selected patients with recent-onset AF 1 g of procainamide intravenously. If patients converted to sinus rhythm, they were discharged. If not they were offered electrical cardioversion with moderate sedation using propofol, a procedure performed by two ED physicians. Patients who converted were sent home; for those who didn’t, cardiology was called in, and hospital admission usually followed.
"It took a while, honestly, to develop a protocol everyone felt comfortable with. We developed ED order sets for the nurses. There was a lot of education. But now when we say we’re going to cardiovert someone in the ED, no one seems to get uptight and upset about it," according to Dr. White.
The Ottawa Aggressive Protocol has been successfully used in Canada where "they’ve been converting patients in the ED and discharging them home with no bad outcomes. So why, then, are we in the United States admitting these patients for rate control?" Dr. White asked.
The protocol is in the process of being modified in response to the release of the 2014 American Heart Association/American College of Cardiology/Heart Rhythm Society guidelines for the management of patients with AF (J. Am. Coll. Cardiol. 2014 [doi:10.1016/j.acc.2014.03.022]).
Those guidelines recommend that all patients with AF and a CHA2DS2-VASc score of 2 or more be placed on an oral anticoagulant before or immediately following cardioversion.
Dr. White remarked that "personally, I would love to see this happen because I think our exclusion criteria are complicated and not reproducible. My thought is we should (instead) ask these three questions:
• Is the atrial fibrillation of recent onset and the primary diagnosis?
• Is the patient at CHA2DS2-VASc of 0-1 without any significant valvular disease?
• Is the QTc interval less than 460 msec with no other arrhythmia?
If the answer to all three questions is ‘yes,’ then we use ED rhythm control. If not, we consult cardiology," she said.
Dr. White reported having no financial conflicts regarding this study.
DALLAS – It’s not the rate; it’s the rhythm control that matters most for a selected subgroup of patients who present to the emergency department with recent onset atrial fibrillation.
The rhythm control approach, essentially the Ottawa Aggressive Protocol, uses intravenous procainamide as first-line therapy and, if the pharmacologic conversion is unsuccessful, subsequent electrical cardioversion by ED physicians. Patients converted to sinus rhythm are discharged home.
In a single-center prospective study, the rhythm control protocol proved safe, and 96% of recent-onset AF patients were converted in the ED. Among the historical controls, 56% were converted. Moreover, the rhythm control protocol cut ED lengths of stay, reduced hospital admissions, and proved highly popular with patients, who were returned to sinus rhythm and discharged home, noted Dr. Jennifer L. White, who presented the study results at the annual meeting of the Society for Academic Emergency Medicine.
"Is this study practice changing? It was for us. It’s our ED protocol now," said Dr. White, an emergency physician at Doylestown (Pa.) Hospital.
The Doylestown study included 46 consecutive patients who presented to the ED with atrial fibrillation (AF) of less than 48 hours duration. They were compared with 50 historical controls who met the same inclusion criteria and were treated before the ED rhythm protocol was implemented.
Dr. White said it took nearly 2 years to develop an ED protocol for AF conversion that was acceptable to cardiologists, nurses, and hospital administrators. The cardiologists dictated the exclusion criteria: No patients received the protocol if they had coronary artery disease, fever, concurrent ischemia, used medication that prolongs the QT interval, ejection fractions below 35%, hospital admission within the prior 3 months, and use of any antiarrhythmic agent within the past 72 hours. Patients with a history of TIA had to be on an oral anticoagulant. The baseline QTc could not exceed 460 msec.
Follow-up interviews conducted at 30 days showed no strokes or other serious adverse events, and a mean patient satisfaction score of 9.6 out of a possible 10. AF recurred in 4 of 46 patients during the 30-day period; 96% of patients were seen by a cardiologist within 14 days after ED discharge, as recommended by the ED staff.
The protocol put to the test in the ED in Doylestown entailed giving selected patients with recent-onset AF 1 g of procainamide intravenously. If patients converted to sinus rhythm, they were discharged. If not they were offered electrical cardioversion with moderate sedation using propofol, a procedure performed by two ED physicians. Patients who converted were sent home; for those who didn’t, cardiology was called in, and hospital admission usually followed.
"It took a while, honestly, to develop a protocol everyone felt comfortable with. We developed ED order sets for the nurses. There was a lot of education. But now when we say we’re going to cardiovert someone in the ED, no one seems to get uptight and upset about it," according to Dr. White.
The Ottawa Aggressive Protocol has been successfully used in Canada where "they’ve been converting patients in the ED and discharging them home with no bad outcomes. So why, then, are we in the United States admitting these patients for rate control?" Dr. White asked.
The protocol is in the process of being modified in response to the release of the 2014 American Heart Association/American College of Cardiology/Heart Rhythm Society guidelines for the management of patients with AF (J. Am. Coll. Cardiol. 2014 [doi:10.1016/j.acc.2014.03.022]).
Those guidelines recommend that all patients with AF and a CHA2DS2-VASc score of 2 or more be placed on an oral anticoagulant before or immediately following cardioversion.
Dr. White remarked that "personally, I would love to see this happen because I think our exclusion criteria are complicated and not reproducible. My thought is we should (instead) ask these three questions:
• Is the atrial fibrillation of recent onset and the primary diagnosis?
• Is the patient at CHA2DS2-VASc of 0-1 without any significant valvular disease?
• Is the QTc interval less than 460 msec with no other arrhythmia?
If the answer to all three questions is ‘yes,’ then we use ED rhythm control. If not, we consult cardiology," she said.
Dr. White reported having no financial conflicts regarding this study.
AT SAEM 2014
Key clinical point: Select patients with recent-onset AF were safely and effectively converted to sinus rhythm in the ED using the Ottawa Aggressive Protocol.
Major finding: Of patients with AF of less than 48 hours duration, 4% of those managed using intravenous procainamide as first-line therapy with subsequent electrical cardioversion were admitted to the hospital. Of those managed according to the standard protocol, 60% were admitted.
Data source: The single-center, prospective study included 46 consecutive patients with recent-onset AF who presented to the ED and met study inclusion criteria and 50 matched historical controls.
Disclosures: The presenter reported having no financial conflicts regarding this study, conducted with institutional funds.
Ask about sleep sweat before diagnosing primary focal hyperhidrosis
COEUR D’ALENE, IDAHO – An individual who complains of excessive focal sweating that continues during sleep does not – repeat, not – have primary focal hyperhidrosis, according to pediatric dermatologist Dr. Jane S. Bellet.
"If you remember nothing else I tell you today, the sweating must cease during sleep. This is absolutely critical. Otherwise you’re dealing with something completely different. So I really press every single patient on that question," she emphasized at the annual meeting of the Society for Pediatric Dermatology.
Primary focal hyperhidrosis occurs in 1.6% of children and adolescents. Two-thirds of affected patients have a positive family history.
"This is really a life-altering condition, and if untreated it will continue unabated into adulthood. There are effective treatment options. We can make a profound difference for these children and adolescents," she said.
Primary focal hyperhidrosis is a clinical diagnosis. It’s based upon the patient’s history, a review of symptoms, and physical examination. No tests are needed, although Minor’s starch iodine test or the older quinizarin test can be useful as documentation for insurance purposes or to guide botulinum toxin A therapy, according to Dr. Bellet of Duke University, Durham, N.C.
According to the widely accepted, decade-old diagnostic criteria developed by a multispecialty expert working group, the diagnosis requires visible, excessive, focal sweating of the axillae, palms, soles, or face/head for at least 6 months with no apparent cause, plus at least two of six additional criteria. These are onset before age 25 years; bilateral, relatively symmetric sweating; impairment of daily activities; cessation during sleep; a positive family history for the disorder; and at least one episode per week (J. Am. Acad. Dermatol. 2004;51:274-86).
Sweating that continues during sleep is due to one of many possible secondary causes, which are elaborated upon in the multispecialty working group’s report.
"It’s a completely separate algorithm, but if you’re dealing with secondary hyperhidrosis there will always be clues that will send you down that pathway," she said.
The patient history is critical in establishing the diagnosis of primary focal hyperhidrosis and its adverse effect on quality of life and daily activities, which is important to document for insurance purposes.
The physical exam is "pretty basic," according to Dr. Bellet. She uses it as an opportunity to touch the patient and establish a bond.
"Shake hands. It shows the patient you understand their condition, and you know their embarrassment. Resist the temptation to wipe your hands on your pants afterward," she advised.
The review of symptoms is essentially a screen for secondary causes of hyperhidrosis to make sure that the correct diagnosis is indeed primary focal hyperhidrosis. Fever, headache, weight loss, abdominal pain, vomiting, palpitations, anorexia – these point toward a secondary cause. Medications that can cause a generalized sweating problem include antidepressants, antimigraine medications, beta-agonists, pilocarpine, insulin, and GnRH agonists.
Dr. Bellet reported having no financial conflicts of interest regarding her presentation.
COEUR D’ALENE, IDAHO – An individual who complains of excessive focal sweating that continues during sleep does not – repeat, not – have primary focal hyperhidrosis, according to pediatric dermatologist Dr. Jane S. Bellet.
"If you remember nothing else I tell you today, the sweating must cease during sleep. This is absolutely critical. Otherwise you’re dealing with something completely different. So I really press every single patient on that question," she emphasized at the annual meeting of the Society for Pediatric Dermatology.
Primary focal hyperhidrosis occurs in 1.6% of children and adolescents. Two-thirds of affected patients have a positive family history.
"This is really a life-altering condition, and if untreated it will continue unabated into adulthood. There are effective treatment options. We can make a profound difference for these children and adolescents," she said.
Primary focal hyperhidrosis is a clinical diagnosis. It’s based upon the patient’s history, a review of symptoms, and physical examination. No tests are needed, although Minor’s starch iodine test or the older quinizarin test can be useful as documentation for insurance purposes or to guide botulinum toxin A therapy, according to Dr. Bellet of Duke University, Durham, N.C.
According to the widely accepted, decade-old diagnostic criteria developed by a multispecialty expert working group, the diagnosis requires visible, excessive, focal sweating of the axillae, palms, soles, or face/head for at least 6 months with no apparent cause, plus at least two of six additional criteria. These are onset before age 25 years; bilateral, relatively symmetric sweating; impairment of daily activities; cessation during sleep; a positive family history for the disorder; and at least one episode per week (J. Am. Acad. Dermatol. 2004;51:274-86).
Sweating that continues during sleep is due to one of many possible secondary causes, which are elaborated upon in the multispecialty working group’s report.
"It’s a completely separate algorithm, but if you’re dealing with secondary hyperhidrosis there will always be clues that will send you down that pathway," she said.
The patient history is critical in establishing the diagnosis of primary focal hyperhidrosis and its adverse effect on quality of life and daily activities, which is important to document for insurance purposes.
The physical exam is "pretty basic," according to Dr. Bellet. She uses it as an opportunity to touch the patient and establish a bond.
"Shake hands. It shows the patient you understand their condition, and you know their embarrassment. Resist the temptation to wipe your hands on your pants afterward," she advised.
The review of symptoms is essentially a screen for secondary causes of hyperhidrosis to make sure that the correct diagnosis is indeed primary focal hyperhidrosis. Fever, headache, weight loss, abdominal pain, vomiting, palpitations, anorexia – these point toward a secondary cause. Medications that can cause a generalized sweating problem include antidepressants, antimigraine medications, beta-agonists, pilocarpine, insulin, and GnRH agonists.
Dr. Bellet reported having no financial conflicts of interest regarding her presentation.
COEUR D’ALENE, IDAHO – An individual who complains of excessive focal sweating that continues during sleep does not – repeat, not – have primary focal hyperhidrosis, according to pediatric dermatologist Dr. Jane S. Bellet.
"If you remember nothing else I tell you today, the sweating must cease during sleep. This is absolutely critical. Otherwise you’re dealing with something completely different. So I really press every single patient on that question," she emphasized at the annual meeting of the Society for Pediatric Dermatology.
Primary focal hyperhidrosis occurs in 1.6% of children and adolescents. Two-thirds of affected patients have a positive family history.
"This is really a life-altering condition, and if untreated it will continue unabated into adulthood. There are effective treatment options. We can make a profound difference for these children and adolescents," she said.
Primary focal hyperhidrosis is a clinical diagnosis. It’s based upon the patient’s history, a review of symptoms, and physical examination. No tests are needed, although Minor’s starch iodine test or the older quinizarin test can be useful as documentation for insurance purposes or to guide botulinum toxin A therapy, according to Dr. Bellet of Duke University, Durham, N.C.
According to the widely accepted, decade-old diagnostic criteria developed by a multispecialty expert working group, the diagnosis requires visible, excessive, focal sweating of the axillae, palms, soles, or face/head for at least 6 months with no apparent cause, plus at least two of six additional criteria. These are onset before age 25 years; bilateral, relatively symmetric sweating; impairment of daily activities; cessation during sleep; a positive family history for the disorder; and at least one episode per week (J. Am. Acad. Dermatol. 2004;51:274-86).
Sweating that continues during sleep is due to one of many possible secondary causes, which are elaborated upon in the multispecialty working group’s report.
"It’s a completely separate algorithm, but if you’re dealing with secondary hyperhidrosis there will always be clues that will send you down that pathway," she said.
The patient history is critical in establishing the diagnosis of primary focal hyperhidrosis and its adverse effect on quality of life and daily activities, which is important to document for insurance purposes.
The physical exam is "pretty basic," according to Dr. Bellet. She uses it as an opportunity to touch the patient and establish a bond.
"Shake hands. It shows the patient you understand their condition, and you know their embarrassment. Resist the temptation to wipe your hands on your pants afterward," she advised.
The review of symptoms is essentially a screen for secondary causes of hyperhidrosis to make sure that the correct diagnosis is indeed primary focal hyperhidrosis. Fever, headache, weight loss, abdominal pain, vomiting, palpitations, anorexia – these point toward a secondary cause. Medications that can cause a generalized sweating problem include antidepressants, antimigraine medications, beta-agonists, pilocarpine, insulin, and GnRH agonists.
Dr. Bellet reported having no financial conflicts of interest regarding her presentation.
EXPERT ANALYSIS FROM THE SPD ANNUAL MEETING
Five factors predict biphasic reactions in children with anaphylaxis
DALLAS – Five newly recognized clinical predictors are useful in identifying which children with anaphylaxis are at increased risk for a biphasic reaction.
"Children who match none of the five criteria can actually be discharged sooner from the ED [emergency department]. These predictors can potentially improve the efficiency and quality of care in the ED," Dr. Waleed Alqurashi said at the annual meeting of the Society for Academic Emergency Medicine.

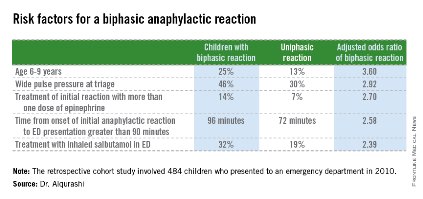
The five risk factors are age of 6-9 years, a wide pulse pressure at triage, treatment of initial reaction requiring more than one dose of epinephrine, time from onset of initial anaphylactic reaction to ED presentation, and treatment with inhaled salbutamol in the ED (see graphic).
The other key – and surprising – finding was that prophylactic administration of systemic corticosteroids was ineffective in preventing biphasic reactions. The result is at odds with classic teaching regarding the benefit of prophylactic steroids in patients with anaphylaxis. "This is the largest study to date of biphasic reactions in children, and we found no association. Also, there’s no biologic plausibility for systemic steroids to prevent anaphylaxis," asserted Dr. Alqurashi of Children’s Hospital of Eastern Ontario, Ottawa.
He presented a multicenter, retrospective cohort study of 484 children who presented with anaphylaxis to an ED during 2010. Biphasic reaction – the recurrence of anaphylactic symptoms at least 1 hour after initial resolution despite no additional exposure to the antigen – occurred in 71 (14.7%).
These biphasic events can be potentially fatal, he said. The 2010 guidelines on anaphylaxis from the National Institute of Allergy and Infectious Diseases underscore the fact that significant knowledge gaps exist regarding the incidence, predictors, and treatment of biphasic reactions (J. Allergy Clin. Immunol. 2010;126:S1-58).
In the Canadian study, 49% of biphasic reactions were sufficiently severe as to require treatment with epinephrine. Three-quarters of these children developed their biphasic reaction prior to ED discharge, at a median of 4.7 hours following onset of the initial reaction. Onset in those whose biphasic reaction occurred after ED discharge was a median of 18.5 hours after onset of the first anaphylactic reaction.
No validated anaphylaxis severity score exists, Dr. Alqurashi remarked, so it wasn’t possible to analyze the relationship between initial reaction severity and likelihood of a subsequent biphasic event. However, several of the clinical predictors identified in this study via multivariate logistic regression analysis are clearly proxies for a more severe reaction.
"If a patient had a severe biphasic reaction, it was more likely to occur within 6 hours. So those with a mild initial anaphylactic reaction, if they don’t match any of these five criteria, can be sent home early. The majority of biphasic reactions occurring after ED discharge did not require epinephrine therapy," he observed.
Anaphylaxis is no longer a rare event, according to Dr. Alqurashi. During the last decade, rates for food-induced anaphylaxis have climbed 350% and for non–food-induced anaphylaxis 230%.
Dr. Marianne Gausche-Hill rose from the audience to comment that she gleaned a slightly different lesson.
"My take-home message from this study is if [patients] didn’t have any of the risk factors, maybe you could discharge them in 6 hours because they’re really unlikely to get into trouble. But if they have any risk factor, it’s probably best just to admit them overnight, which is our standard practice," said Dr. Gausche-Hill, professor of emergency medicine and director of the division of pediatric emergency medicine at Harbor-UCLA Medical Center, Los Angeles.
Dr. Alqurashi reported having no financial conflicts of interest with regard to his study, which was conducted free of commercial support.
DALLAS – Five newly recognized clinical predictors are useful in identifying which children with anaphylaxis are at increased risk for a biphasic reaction.
"Children who match none of the five criteria can actually be discharged sooner from the ED [emergency department]. These predictors can potentially improve the efficiency and quality of care in the ED," Dr. Waleed Alqurashi said at the annual meeting of the Society for Academic Emergency Medicine.

The five risk factors are age of 6-9 years, a wide pulse pressure at triage, treatment of initial reaction requiring more than one dose of epinephrine, time from onset of initial anaphylactic reaction to ED presentation, and treatment with inhaled salbutamol in the ED (see graphic).
The other key – and surprising – finding was that prophylactic administration of systemic corticosteroids was ineffective in preventing biphasic reactions. The result is at odds with classic teaching regarding the benefit of prophylactic steroids in patients with anaphylaxis. "This is the largest study to date of biphasic reactions in children, and we found no association. Also, there’s no biologic plausibility for systemic steroids to prevent anaphylaxis," asserted Dr. Alqurashi of Children’s Hospital of Eastern Ontario, Ottawa.
He presented a multicenter, retrospective cohort study of 484 children who presented with anaphylaxis to an ED during 2010. Biphasic reaction – the recurrence of anaphylactic symptoms at least 1 hour after initial resolution despite no additional exposure to the antigen – occurred in 71 (14.7%).
These biphasic events can be potentially fatal, he said. The 2010 guidelines on anaphylaxis from the National Institute of Allergy and Infectious Diseases underscore the fact that significant knowledge gaps exist regarding the incidence, predictors, and treatment of biphasic reactions (J. Allergy Clin. Immunol. 2010;126:S1-58).
In the Canadian study, 49% of biphasic reactions were sufficiently severe as to require treatment with epinephrine. Three-quarters of these children developed their biphasic reaction prior to ED discharge, at a median of 4.7 hours following onset of the initial reaction. Onset in those whose biphasic reaction occurred after ED discharge was a median of 18.5 hours after onset of the first anaphylactic reaction.
No validated anaphylaxis severity score exists, Dr. Alqurashi remarked, so it wasn’t possible to analyze the relationship between initial reaction severity and likelihood of a subsequent biphasic event. However, several of the clinical predictors identified in this study via multivariate logistic regression analysis are clearly proxies for a more severe reaction.
"If a patient had a severe biphasic reaction, it was more likely to occur within 6 hours. So those with a mild initial anaphylactic reaction, if they don’t match any of these five criteria, can be sent home early. The majority of biphasic reactions occurring after ED discharge did not require epinephrine therapy," he observed.
Anaphylaxis is no longer a rare event, according to Dr. Alqurashi. During the last decade, rates for food-induced anaphylaxis have climbed 350% and for non–food-induced anaphylaxis 230%.
Dr. Marianne Gausche-Hill rose from the audience to comment that she gleaned a slightly different lesson.
"My take-home message from this study is if [patients] didn’t have any of the risk factors, maybe you could discharge them in 6 hours because they’re really unlikely to get into trouble. But if they have any risk factor, it’s probably best just to admit them overnight, which is our standard practice," said Dr. Gausche-Hill, professor of emergency medicine and director of the division of pediatric emergency medicine at Harbor-UCLA Medical Center, Los Angeles.
Dr. Alqurashi reported having no financial conflicts of interest with regard to his study, which was conducted free of commercial support.
DALLAS – Five newly recognized clinical predictors are useful in identifying which children with anaphylaxis are at increased risk for a biphasic reaction.
"Children who match none of the five criteria can actually be discharged sooner from the ED [emergency department]. These predictors can potentially improve the efficiency and quality of care in the ED," Dr. Waleed Alqurashi said at the annual meeting of the Society for Academic Emergency Medicine.

The five risk factors are age of 6-9 years, a wide pulse pressure at triage, treatment of initial reaction requiring more than one dose of epinephrine, time from onset of initial anaphylactic reaction to ED presentation, and treatment with inhaled salbutamol in the ED (see graphic).
The other key – and surprising – finding was that prophylactic administration of systemic corticosteroids was ineffective in preventing biphasic reactions. The result is at odds with classic teaching regarding the benefit of prophylactic steroids in patients with anaphylaxis. "This is the largest study to date of biphasic reactions in children, and we found no association. Also, there’s no biologic plausibility for systemic steroids to prevent anaphylaxis," asserted Dr. Alqurashi of Children’s Hospital of Eastern Ontario, Ottawa.
He presented a multicenter, retrospective cohort study of 484 children who presented with anaphylaxis to an ED during 2010. Biphasic reaction – the recurrence of anaphylactic symptoms at least 1 hour after initial resolution despite no additional exposure to the antigen – occurred in 71 (14.7%).
These biphasic events can be potentially fatal, he said. The 2010 guidelines on anaphylaxis from the National Institute of Allergy and Infectious Diseases underscore the fact that significant knowledge gaps exist regarding the incidence, predictors, and treatment of biphasic reactions (J. Allergy Clin. Immunol. 2010;126:S1-58).
In the Canadian study, 49% of biphasic reactions were sufficiently severe as to require treatment with epinephrine. Three-quarters of these children developed their biphasic reaction prior to ED discharge, at a median of 4.7 hours following onset of the initial reaction. Onset in those whose biphasic reaction occurred after ED discharge was a median of 18.5 hours after onset of the first anaphylactic reaction.
No validated anaphylaxis severity score exists, Dr. Alqurashi remarked, so it wasn’t possible to analyze the relationship between initial reaction severity and likelihood of a subsequent biphasic event. However, several of the clinical predictors identified in this study via multivariate logistic regression analysis are clearly proxies for a more severe reaction.
"If a patient had a severe biphasic reaction, it was more likely to occur within 6 hours. So those with a mild initial anaphylactic reaction, if they don’t match any of these five criteria, can be sent home early. The majority of biphasic reactions occurring after ED discharge did not require epinephrine therapy," he observed.
Anaphylaxis is no longer a rare event, according to Dr. Alqurashi. During the last decade, rates for food-induced anaphylaxis have climbed 350% and for non–food-induced anaphylaxis 230%.
Dr. Marianne Gausche-Hill rose from the audience to comment that she gleaned a slightly different lesson.
"My take-home message from this study is if [patients] didn’t have any of the risk factors, maybe you could discharge them in 6 hours because they’re really unlikely to get into trouble. But if they have any risk factor, it’s probably best just to admit them overnight, which is our standard practice," said Dr. Gausche-Hill, professor of emergency medicine and director of the division of pediatric emergency medicine at Harbor-UCLA Medical Center, Los Angeles.
Dr. Alqurashi reported having no financial conflicts of interest with regard to his study, which was conducted free of commercial support.
AT SAEM 2014
Key clinical point: Children with anaphylaxis may reasonably be discharged from the ED at 6 hours provided they don’t have any of five newly identified clinical predictors of increased risk for a biphasic reaction.
Major finding: In children presenting to the ED with anaphylaxis, 75% of biphasic reactions occurred within 6 hours after the onset of the initial reaction. Biphasic reactions occurring after more than 6 hours were seldom severe.
Data source: This was a multicenter, retrospective cohort study of 484 children who presented to EDs with anaphylaxis during 2010.
Disclosures: Dr. Alqurashi reported having no financial conflicts of interest with regard to his study, which was conducted free of commercial support.
Chronic kidney disease brings often subtle cognitive impairment
LAS VEGAS – Patients on dialysis for end-stage renal disease have a high burden of impaired executive function that’s typically missed by clinicians, Dr. Daniel E. Weiner said at a meeting sponsored by the National Kidney Foundation.
Vascular dementia, which is much more common than Alzheimer’s disease in patients with chronic kidney disease (CKD), impairs executive function and is associated with anatomic white matter brain disease. Executive function is the cognitive domain concerned with attention, processing speed, reasoning, planning, and problem solving. Clinicians tend to miss the presence of cognitive impairment in patients with CKD because they typically rely upon the Mini-Mental State Examination (MMSE) to screen for impaired cognition. And the MMSE focuses on memory difficulties, which are more common in Alzheimer’s disease, rather than the more subtle domain of executive function, he explained.
"We’re trying to teach (CKD patients) about complex topics such as fluid restriction, medication management, and salt intake, and you wonder if it just goes in one ear and out the other because they’re not able to process these complicated issues," observed Dr. Weiner, a nephrologist at Tufts University, Boston.
Dr. Weiner was coauthor of a recent cross-sectional cohort study in which 314 hemodialysis patients at six Boston-area hemodialysis units completed a comprehensive battery of neuropsychological tests assessing memory and executive function. The patients scored markedly worse than general population norms on executive function, but not on memory performance. Moreover, impaired executive function was highly prevalent even in patients with a normal MMSE of 24 or more. The take-home message: be cautious in using an MMSE score of less than 24 to screen for cognitive impairment in dialysis patients (Neurology 2013;80:471-80).
Moderate to severe cognitive impairment is also prevalent in patients on peritoneal dialysis, as shown by investigators at the University of Minnesota. They gave a battery of nine validated neuropsychological tests to 51 peritoneal dialysis patients, 338 hemodialysis patients, and 101 controls without CKD who were matched for age and comorbid conditions. Of the peritoneal dialysis cohort, 31% had severe cognitive impairment, as did 37% of the hemodialysis group and 13% of controls. In an adjusted logistic regression model, peritoneal dialysis was associated with a 2.5-fold increased risk of moderate to severe global cognitive impairment, compared with the no-CKD controls, while hemodialysis patients had a similar 3.16-fold increased risk (Am. J. Kidney Dis. 2011;57:612-20).
"That’s a remarkably high prevalence: one-third of dialysis patients in a moderately well educated Minnesota population with access to medical care performed poorly enough on a neurocognitive battery to be classified as severely cognitively impaired, meaning they flubbed on two different domains of cognitive function," Dr. Weiner commented.
The effect of dialysis itself upon cognition is a matter of continuing controversy. Although some nephrologists posit that a more intensive dialysis regimen would result in improved cognitive performance, that hasn’t been borne out in analyses to date (Kidney International 2011;79:14-22).
"This suggests that the cognitive impairment we see in dialysis patients isn’t related to the dialysis dose. It’s not related to retained solutes, but is more related to the – for lack of a better word – bad humors patients have been exposed to for many years and which have put them into a situation where they require dialysis," according to Dr. Weiner.
"We have patients that drop their systolic blood pressure by 20, 30, 40, even unfortunately 80 mm Hg during dialysis. You can’t imagine that this is good for you. We’re inducing transient microvascular ischemia, which is ultimately manifest structurally: in the kidney we get fibrosis, in the brain we get white matter disease," he continued.
Cognitive dysfunction is associated with increased mortality risk even in CKD patients who are not dialysis dependent. In an analysis of National Health and Nutrition Examination Survey III data, non–dialysis dependent CKD patients in the lowest quartile in terms of cognitive score had a twofold increased risk of mortality, compared with those in the highest quartile (Am. J. Nephrol. 2012;35:49-57).
Chronic kidney disease as defined by an estimated glomerular filtration rate below 60 mL/min/1.73 m2 is also a cardiovascular disease risk equivalent for stroke. Dr. Weiner and coworkers showed that the stroke rate in patients with CKD but no known cardiovascular disease is elevated to roughly the same extent as in patients with known cardiovascular disease but no CKD (Am. J. Kidney Dis. 2006;48:392-401).
More recently, Dr. Weiner and his colleagues showed in a brain MRI study that hemodialysis patients not only have far more white matter disease and cerebral atrophy than controls without kidney disease, they also have a high prevalence of previously unrecognized strokes. The cross-sectional study involved 45 hemodialysis patients and 67 controls, all without a history of stroke. Impressively, 18% of the hemodialysis patients had evidence of a small-vessel infarct on MRI and another 8% had a large-vessel infarct (Am. J. Kidney Dis. 2013;61:271-8).
The high rate of often subtle cognitive impairment among dialysis patients points to the need for alternative patient education strategies. In order to reinforce his educational messages, Dr. Weiner makes an effort to convey the same extensive information to family members and other primary caregivers that he provides to the patients themselves.
He reported having no financial conflicts.
LAS VEGAS – Patients on dialysis for end-stage renal disease have a high burden of impaired executive function that’s typically missed by clinicians, Dr. Daniel E. Weiner said at a meeting sponsored by the National Kidney Foundation.
Vascular dementia, which is much more common than Alzheimer’s disease in patients with chronic kidney disease (CKD), impairs executive function and is associated with anatomic white matter brain disease. Executive function is the cognitive domain concerned with attention, processing speed, reasoning, planning, and problem solving. Clinicians tend to miss the presence of cognitive impairment in patients with CKD because they typically rely upon the Mini-Mental State Examination (MMSE) to screen for impaired cognition. And the MMSE focuses on memory difficulties, which are more common in Alzheimer’s disease, rather than the more subtle domain of executive function, he explained.
"We’re trying to teach (CKD patients) about complex topics such as fluid restriction, medication management, and salt intake, and you wonder if it just goes in one ear and out the other because they’re not able to process these complicated issues," observed Dr. Weiner, a nephrologist at Tufts University, Boston.
Dr. Weiner was coauthor of a recent cross-sectional cohort study in which 314 hemodialysis patients at six Boston-area hemodialysis units completed a comprehensive battery of neuropsychological tests assessing memory and executive function. The patients scored markedly worse than general population norms on executive function, but not on memory performance. Moreover, impaired executive function was highly prevalent even in patients with a normal MMSE of 24 or more. The take-home message: be cautious in using an MMSE score of less than 24 to screen for cognitive impairment in dialysis patients (Neurology 2013;80:471-80).
Moderate to severe cognitive impairment is also prevalent in patients on peritoneal dialysis, as shown by investigators at the University of Minnesota. They gave a battery of nine validated neuropsychological tests to 51 peritoneal dialysis patients, 338 hemodialysis patients, and 101 controls without CKD who were matched for age and comorbid conditions. Of the peritoneal dialysis cohort, 31% had severe cognitive impairment, as did 37% of the hemodialysis group and 13% of controls. In an adjusted logistic regression model, peritoneal dialysis was associated with a 2.5-fold increased risk of moderate to severe global cognitive impairment, compared with the no-CKD controls, while hemodialysis patients had a similar 3.16-fold increased risk (Am. J. Kidney Dis. 2011;57:612-20).
"That’s a remarkably high prevalence: one-third of dialysis patients in a moderately well educated Minnesota population with access to medical care performed poorly enough on a neurocognitive battery to be classified as severely cognitively impaired, meaning they flubbed on two different domains of cognitive function," Dr. Weiner commented.
The effect of dialysis itself upon cognition is a matter of continuing controversy. Although some nephrologists posit that a more intensive dialysis regimen would result in improved cognitive performance, that hasn’t been borne out in analyses to date (Kidney International 2011;79:14-22).
"This suggests that the cognitive impairment we see in dialysis patients isn’t related to the dialysis dose. It’s not related to retained solutes, but is more related to the – for lack of a better word – bad humors patients have been exposed to for many years and which have put them into a situation where they require dialysis," according to Dr. Weiner.
"We have patients that drop their systolic blood pressure by 20, 30, 40, even unfortunately 80 mm Hg during dialysis. You can’t imagine that this is good for you. We’re inducing transient microvascular ischemia, which is ultimately manifest structurally: in the kidney we get fibrosis, in the brain we get white matter disease," he continued.
Cognitive dysfunction is associated with increased mortality risk even in CKD patients who are not dialysis dependent. In an analysis of National Health and Nutrition Examination Survey III data, non–dialysis dependent CKD patients in the lowest quartile in terms of cognitive score had a twofold increased risk of mortality, compared with those in the highest quartile (Am. J. Nephrol. 2012;35:49-57).
Chronic kidney disease as defined by an estimated glomerular filtration rate below 60 mL/min/1.73 m2 is also a cardiovascular disease risk equivalent for stroke. Dr. Weiner and coworkers showed that the stroke rate in patients with CKD but no known cardiovascular disease is elevated to roughly the same extent as in patients with known cardiovascular disease but no CKD (Am. J. Kidney Dis. 2006;48:392-401).
More recently, Dr. Weiner and his colleagues showed in a brain MRI study that hemodialysis patients not only have far more white matter disease and cerebral atrophy than controls without kidney disease, they also have a high prevalence of previously unrecognized strokes. The cross-sectional study involved 45 hemodialysis patients and 67 controls, all without a history of stroke. Impressively, 18% of the hemodialysis patients had evidence of a small-vessel infarct on MRI and another 8% had a large-vessel infarct (Am. J. Kidney Dis. 2013;61:271-8).
The high rate of often subtle cognitive impairment among dialysis patients points to the need for alternative patient education strategies. In order to reinforce his educational messages, Dr. Weiner makes an effort to convey the same extensive information to family members and other primary caregivers that he provides to the patients themselves.
He reported having no financial conflicts.
LAS VEGAS – Patients on dialysis for end-stage renal disease have a high burden of impaired executive function that’s typically missed by clinicians, Dr. Daniel E. Weiner said at a meeting sponsored by the National Kidney Foundation.
Vascular dementia, which is much more common than Alzheimer’s disease in patients with chronic kidney disease (CKD), impairs executive function and is associated with anatomic white matter brain disease. Executive function is the cognitive domain concerned with attention, processing speed, reasoning, planning, and problem solving. Clinicians tend to miss the presence of cognitive impairment in patients with CKD because they typically rely upon the Mini-Mental State Examination (MMSE) to screen for impaired cognition. And the MMSE focuses on memory difficulties, which are more common in Alzheimer’s disease, rather than the more subtle domain of executive function, he explained.
"We’re trying to teach (CKD patients) about complex topics such as fluid restriction, medication management, and salt intake, and you wonder if it just goes in one ear and out the other because they’re not able to process these complicated issues," observed Dr. Weiner, a nephrologist at Tufts University, Boston.
Dr. Weiner was coauthor of a recent cross-sectional cohort study in which 314 hemodialysis patients at six Boston-area hemodialysis units completed a comprehensive battery of neuropsychological tests assessing memory and executive function. The patients scored markedly worse than general population norms on executive function, but not on memory performance. Moreover, impaired executive function was highly prevalent even in patients with a normal MMSE of 24 or more. The take-home message: be cautious in using an MMSE score of less than 24 to screen for cognitive impairment in dialysis patients (Neurology 2013;80:471-80).
Moderate to severe cognitive impairment is also prevalent in patients on peritoneal dialysis, as shown by investigators at the University of Minnesota. They gave a battery of nine validated neuropsychological tests to 51 peritoneal dialysis patients, 338 hemodialysis patients, and 101 controls without CKD who were matched for age and comorbid conditions. Of the peritoneal dialysis cohort, 31% had severe cognitive impairment, as did 37% of the hemodialysis group and 13% of controls. In an adjusted logistic regression model, peritoneal dialysis was associated with a 2.5-fold increased risk of moderate to severe global cognitive impairment, compared with the no-CKD controls, while hemodialysis patients had a similar 3.16-fold increased risk (Am. J. Kidney Dis. 2011;57:612-20).
"That’s a remarkably high prevalence: one-third of dialysis patients in a moderately well educated Minnesota population with access to medical care performed poorly enough on a neurocognitive battery to be classified as severely cognitively impaired, meaning they flubbed on two different domains of cognitive function," Dr. Weiner commented.
The effect of dialysis itself upon cognition is a matter of continuing controversy. Although some nephrologists posit that a more intensive dialysis regimen would result in improved cognitive performance, that hasn’t been borne out in analyses to date (Kidney International 2011;79:14-22).
"This suggests that the cognitive impairment we see in dialysis patients isn’t related to the dialysis dose. It’s not related to retained solutes, but is more related to the – for lack of a better word – bad humors patients have been exposed to for many years and which have put them into a situation where they require dialysis," according to Dr. Weiner.
"We have patients that drop their systolic blood pressure by 20, 30, 40, even unfortunately 80 mm Hg during dialysis. You can’t imagine that this is good for you. We’re inducing transient microvascular ischemia, which is ultimately manifest structurally: in the kidney we get fibrosis, in the brain we get white matter disease," he continued.
Cognitive dysfunction is associated with increased mortality risk even in CKD patients who are not dialysis dependent. In an analysis of National Health and Nutrition Examination Survey III data, non–dialysis dependent CKD patients in the lowest quartile in terms of cognitive score had a twofold increased risk of mortality, compared with those in the highest quartile (Am. J. Nephrol. 2012;35:49-57).
Chronic kidney disease as defined by an estimated glomerular filtration rate below 60 mL/min/1.73 m2 is also a cardiovascular disease risk equivalent for stroke. Dr. Weiner and coworkers showed that the stroke rate in patients with CKD but no known cardiovascular disease is elevated to roughly the same extent as in patients with known cardiovascular disease but no CKD (Am. J. Kidney Dis. 2006;48:392-401).
More recently, Dr. Weiner and his colleagues showed in a brain MRI study that hemodialysis patients not only have far more white matter disease and cerebral atrophy than controls without kidney disease, they also have a high prevalence of previously unrecognized strokes. The cross-sectional study involved 45 hemodialysis patients and 67 controls, all without a history of stroke. Impressively, 18% of the hemodialysis patients had evidence of a small-vessel infarct on MRI and another 8% had a large-vessel infarct (Am. J. Kidney Dis. 2013;61:271-8).
The high rate of often subtle cognitive impairment among dialysis patients points to the need for alternative patient education strategies. In order to reinforce his educational messages, Dr. Weiner makes an effort to convey the same extensive information to family members and other primary caregivers that he provides to the patients themselves.
He reported having no financial conflicts.
EXPERT ANALYSIS FROM SCM 14
Patch testing tricks in atopic dermatitis with concomitant contact dermatitis
COEUR D’ALENE, IDAHO– Contact dermatitis goes together with moderate-to-severe atopic dermatitis like ham and eggs. Reading patch test results in such patients poses unique challenges, because of the impaired skin barrier function intrinsic to atopic dermatitis, coupled with the moist environment created under the occlusive patches, which predisposes to Staphylococcus aureus colonization and superinfection.
All of this makes the evaluation of patch test results in atopic dermatitis more complicated than in patients without contact dermatitis. But there are tricks that greatly reduce the difficulty.
"Patch testing can play a crucial role in the work-up and management of patients with refractory atopic dermatitis. Prior to patch testing, measures should be taken to improve the skin barrier and reduce bacterial overload," Dr. Sharon E. Jacob advised at the annual meeting of the Society for Pediatric Dermatology.
Her recommended preparation program starts 3 weeks prior to the scheduled patch test. The emphasis is on beefing up the disrupted skin barrier through the use of lipid-replenishing emollients and nonalkaline soaps, avoidance of fragrances and other irritant allergens, and preemptive treatment of S. aureus colonization or superinfection, explained Dr. Jacob of Loma Linda (Calif.) University.
At the start of the 3-week countdown, a patient with no clinical signs of skin infection should be checked for nasal carriage of S. aureus. If positive, or if the patient has a remote history of S. aureus colonization or superinfection but no current signs of colonization, such as oozing or crusting, it’s appropriate to begin intranasal, perianal, and umbilical topical mupirocin twice daily for 5 days.
It’s also time to eliminate irritant allergens, start a regimen of dilute bleach baths, embrace the special emollients and soaps, and make sure areas of dermatitis are getting adequately treated with topical steroids, except for the planned patch test area, which must remain steroid free for 7 days prior to the test day.
The lipid-containing emollients, which should contain ceramides or filaggrin degradation products, are to be used all over the body, including the back, until the day before the test.
Examples of the nonalkaline soaps, which are employed to maintain an acidic skin pH, include Aveeno Moisturizing Bar soap, Cetaphil Gentle Cleansing Bar, and Dove Sensitive Skin Unscented Beauty Bar, the pediatric dermatologist continued.
Patients on UV phototherapy for their atopic dermatitis can continue except at the planned patch test site, where it should be avoided for 2 weeks beforehand.
In an adolescent with head and neck atopic dermatitis, a pre–patch test course of oral antifungal therapy is worth considering, according to Dr. Jacob.
For the atopic dermatitis patient with clinical signs of bacterial infection at the pre–patch test office visit, culture the lesions and start a preemptive 10-day course of oral cephalexin at 25-50 mg/kg per day three times daily beginning 3 days prior to patch testing, with the choice of antimicrobial adjusted as warranted by the culture results. These patients also go on the emollients, soaps, dilute bleach baths, and irritant allergen avoidance regimen.
A patient with no signs of skin infection when the test patches are removed can continue with the test readings as scheduled, with no further intervention. However, if signs of infection are present, the patient should immediately go on oral cephalexin if not already on it and take a single bleach bath the same day the patches come off. The bath should be at one-tenth to one-half the customary concentration of 0.005% sodium hypochlorite and should be followed by a fresh water rinse.
The usual patch test reading schedule in atopic dermatitis patients is at 24-48 hours, again at 72-96 hours, and once again at 120 hours. That last reading is vital because patients with atopic dermatitis can have a low irritant threshold; the delayed reading lessens the possibility of reading irritant reactions as positives.
The reading at 72-96 hours is the time for induration testing.
"When evaluating patch test reactions, palpation for induration is absolutely necessary," Dr. Jacob emphasized. "First the evaluator palpates the baseline dermatitis, and then the patch application squares, looking for areas of increased induration and effectively subtracting background."
She reported serving as a consultant to Johnson & Johnson and Medimetriks and as a clinical investigator for SmartPractice, which manufactures patch test kits, the use of which remains nonapproved by the Food and Drug Administration in children.
COEUR D’ALENE, IDAHO– Contact dermatitis goes together with moderate-to-severe atopic dermatitis like ham and eggs. Reading patch test results in such patients poses unique challenges, because of the impaired skin barrier function intrinsic to atopic dermatitis, coupled with the moist environment created under the occlusive patches, which predisposes to Staphylococcus aureus colonization and superinfection.
All of this makes the evaluation of patch test results in atopic dermatitis more complicated than in patients without contact dermatitis. But there are tricks that greatly reduce the difficulty.
"Patch testing can play a crucial role in the work-up and management of patients with refractory atopic dermatitis. Prior to patch testing, measures should be taken to improve the skin barrier and reduce bacterial overload," Dr. Sharon E. Jacob advised at the annual meeting of the Society for Pediatric Dermatology.
Her recommended preparation program starts 3 weeks prior to the scheduled patch test. The emphasis is on beefing up the disrupted skin barrier through the use of lipid-replenishing emollients and nonalkaline soaps, avoidance of fragrances and other irritant allergens, and preemptive treatment of S. aureus colonization or superinfection, explained Dr. Jacob of Loma Linda (Calif.) University.
At the start of the 3-week countdown, a patient with no clinical signs of skin infection should be checked for nasal carriage of S. aureus. If positive, or if the patient has a remote history of S. aureus colonization or superinfection but no current signs of colonization, such as oozing or crusting, it’s appropriate to begin intranasal, perianal, and umbilical topical mupirocin twice daily for 5 days.
It’s also time to eliminate irritant allergens, start a regimen of dilute bleach baths, embrace the special emollients and soaps, and make sure areas of dermatitis are getting adequately treated with topical steroids, except for the planned patch test area, which must remain steroid free for 7 days prior to the test day.
The lipid-containing emollients, which should contain ceramides or filaggrin degradation products, are to be used all over the body, including the back, until the day before the test.
Examples of the nonalkaline soaps, which are employed to maintain an acidic skin pH, include Aveeno Moisturizing Bar soap, Cetaphil Gentle Cleansing Bar, and Dove Sensitive Skin Unscented Beauty Bar, the pediatric dermatologist continued.
Patients on UV phototherapy for their atopic dermatitis can continue except at the planned patch test site, where it should be avoided for 2 weeks beforehand.
In an adolescent with head and neck atopic dermatitis, a pre–patch test course of oral antifungal therapy is worth considering, according to Dr. Jacob.
For the atopic dermatitis patient with clinical signs of bacterial infection at the pre–patch test office visit, culture the lesions and start a preemptive 10-day course of oral cephalexin at 25-50 mg/kg per day three times daily beginning 3 days prior to patch testing, with the choice of antimicrobial adjusted as warranted by the culture results. These patients also go on the emollients, soaps, dilute bleach baths, and irritant allergen avoidance regimen.
A patient with no signs of skin infection when the test patches are removed can continue with the test readings as scheduled, with no further intervention. However, if signs of infection are present, the patient should immediately go on oral cephalexin if not already on it and take a single bleach bath the same day the patches come off. The bath should be at one-tenth to one-half the customary concentration of 0.005% sodium hypochlorite and should be followed by a fresh water rinse.
The usual patch test reading schedule in atopic dermatitis patients is at 24-48 hours, again at 72-96 hours, and once again at 120 hours. That last reading is vital because patients with atopic dermatitis can have a low irritant threshold; the delayed reading lessens the possibility of reading irritant reactions as positives.
The reading at 72-96 hours is the time for induration testing.
"When evaluating patch test reactions, palpation for induration is absolutely necessary," Dr. Jacob emphasized. "First the evaluator palpates the baseline dermatitis, and then the patch application squares, looking for areas of increased induration and effectively subtracting background."
She reported serving as a consultant to Johnson & Johnson and Medimetriks and as a clinical investigator for SmartPractice, which manufactures patch test kits, the use of which remains nonapproved by the Food and Drug Administration in children.
COEUR D’ALENE, IDAHO– Contact dermatitis goes together with moderate-to-severe atopic dermatitis like ham and eggs. Reading patch test results in such patients poses unique challenges, because of the impaired skin barrier function intrinsic to atopic dermatitis, coupled with the moist environment created under the occlusive patches, which predisposes to Staphylococcus aureus colonization and superinfection.
All of this makes the evaluation of patch test results in atopic dermatitis more complicated than in patients without contact dermatitis. But there are tricks that greatly reduce the difficulty.
"Patch testing can play a crucial role in the work-up and management of patients with refractory atopic dermatitis. Prior to patch testing, measures should be taken to improve the skin barrier and reduce bacterial overload," Dr. Sharon E. Jacob advised at the annual meeting of the Society for Pediatric Dermatology.
Her recommended preparation program starts 3 weeks prior to the scheduled patch test. The emphasis is on beefing up the disrupted skin barrier through the use of lipid-replenishing emollients and nonalkaline soaps, avoidance of fragrances and other irritant allergens, and preemptive treatment of S. aureus colonization or superinfection, explained Dr. Jacob of Loma Linda (Calif.) University.
At the start of the 3-week countdown, a patient with no clinical signs of skin infection should be checked for nasal carriage of S. aureus. If positive, or if the patient has a remote history of S. aureus colonization or superinfection but no current signs of colonization, such as oozing or crusting, it’s appropriate to begin intranasal, perianal, and umbilical topical mupirocin twice daily for 5 days.
It’s also time to eliminate irritant allergens, start a regimen of dilute bleach baths, embrace the special emollients and soaps, and make sure areas of dermatitis are getting adequately treated with topical steroids, except for the planned patch test area, which must remain steroid free for 7 days prior to the test day.
The lipid-containing emollients, which should contain ceramides or filaggrin degradation products, are to be used all over the body, including the back, until the day before the test.
Examples of the nonalkaline soaps, which are employed to maintain an acidic skin pH, include Aveeno Moisturizing Bar soap, Cetaphil Gentle Cleansing Bar, and Dove Sensitive Skin Unscented Beauty Bar, the pediatric dermatologist continued.
Patients on UV phototherapy for their atopic dermatitis can continue except at the planned patch test site, where it should be avoided for 2 weeks beforehand.
In an adolescent with head and neck atopic dermatitis, a pre–patch test course of oral antifungal therapy is worth considering, according to Dr. Jacob.
For the atopic dermatitis patient with clinical signs of bacterial infection at the pre–patch test office visit, culture the lesions and start a preemptive 10-day course of oral cephalexin at 25-50 mg/kg per day three times daily beginning 3 days prior to patch testing, with the choice of antimicrobial adjusted as warranted by the culture results. These patients also go on the emollients, soaps, dilute bleach baths, and irritant allergen avoidance regimen.
A patient with no signs of skin infection when the test patches are removed can continue with the test readings as scheduled, with no further intervention. However, if signs of infection are present, the patient should immediately go on oral cephalexin if not already on it and take a single bleach bath the same day the patches come off. The bath should be at one-tenth to one-half the customary concentration of 0.005% sodium hypochlorite and should be followed by a fresh water rinse.
The usual patch test reading schedule in atopic dermatitis patients is at 24-48 hours, again at 72-96 hours, and once again at 120 hours. That last reading is vital because patients with atopic dermatitis can have a low irritant threshold; the delayed reading lessens the possibility of reading irritant reactions as positives.
The reading at 72-96 hours is the time for induration testing.
"When evaluating patch test reactions, palpation for induration is absolutely necessary," Dr. Jacob emphasized. "First the evaluator palpates the baseline dermatitis, and then the patch application squares, looking for areas of increased induration and effectively subtracting background."
She reported serving as a consultant to Johnson & Johnson and Medimetriks and as a clinical investigator for SmartPractice, which manufactures patch test kits, the use of which remains nonapproved by the Food and Drug Administration in children.
EXPERT ANALYSIS FROM THE SPD 2014
Referral-only appointment policy cuts wait times
COEUR D’ALENE, IDAHO – Switching to a referral-only policy in one pediatric dermatology clinic in a children’s hospital reduced the average wait for an initial appointment by 10%.
"A referral-only system may be an effective method for other pediatric dermatology clinics, as well as other pediatric subspecialties, to decrease patient wait time to see a subspecialist," Dr. Tiffany J. Herd concluded at the annual meeting of the Society for Pediatric Dermatology.
Nationally, pediatric dermatology is very close to the top of the list of pediatric subspecialties in terms of patient wait time. Strategies to reduce wait time for pediatric dermatology are urgently needed, noted Dr. Herd of Children’s Mercy Hospitals and Clinics in Kansas City, Mo.
She presented a retrospective study analyzing the impact of implementing a referral-only policy for new outpatient pediatric dermatology appointments at the medical center. The study entailed reviewing the medical records of 6,316 pediatric dermatology clinic patients seen initially either during January-August of 2012–before the switch to a referral-only policy–or in January-August of 2013, after the policy was in place.
Self-referrals accounted for 24% of all initial appointments in 2012, but only 4% a year later. Mean patient wait time fell from 36.4 days in 2012 to 32.9 in 2013, a 9.6% decrease. The median wait time dropped from 34 to 24 days.
The 15 most common diagnoses were the same before and after the policy shift. The top three in both time periods were benign neoplasms of the skin, atopic dermatitis, and acne. Their proportion of total new clinic visits was the same in 2012 and 2013. Indeed, the proportion changed for only one of the top 15 diagnoses: warts, which accounted for 9.2% of initial appointments in 2012, but only 7.4% a year later, according to Dr. Herd.
Of note, however, the complexity of the cases seen in the pediatric dermatology clinic rose substantially following the shift to a referral-only policy. In 2012, 40% of initially referred patients were billed as Level 3 complexity, compared with 68% in 2013.
Patient demographics didn’t change, nor did the type of insurance coverage. These findings suggest that access to pediatric dermatology services remained equitable following implementation of the referral-only policy, she continued.
Primary care physicians are probably capable of handling many of the pediatric dermatologic conditions which they now commonly refer to the subspecialty clinic. An interesting topic for future study will be to see if targeted education of primary care clinicians and pediatric residents regarding the top 15 diagnoses seen in the referral-only pediatric dermatology clinic allows the nondermatologists to reduce their referral rate, Dr. Herd said.
She reported having no financial conflicts regarding this study, which was conducted with institutional funds.
COEUR D’ALENE, IDAHO – Switching to a referral-only policy in one pediatric dermatology clinic in a children’s hospital reduced the average wait for an initial appointment by 10%.
"A referral-only system may be an effective method for other pediatric dermatology clinics, as well as other pediatric subspecialties, to decrease patient wait time to see a subspecialist," Dr. Tiffany J. Herd concluded at the annual meeting of the Society for Pediatric Dermatology.
Nationally, pediatric dermatology is very close to the top of the list of pediatric subspecialties in terms of patient wait time. Strategies to reduce wait time for pediatric dermatology are urgently needed, noted Dr. Herd of Children’s Mercy Hospitals and Clinics in Kansas City, Mo.
She presented a retrospective study analyzing the impact of implementing a referral-only policy for new outpatient pediatric dermatology appointments at the medical center. The study entailed reviewing the medical records of 6,316 pediatric dermatology clinic patients seen initially either during January-August of 2012–before the switch to a referral-only policy–or in January-August of 2013, after the policy was in place.
Self-referrals accounted for 24% of all initial appointments in 2012, but only 4% a year later. Mean patient wait time fell from 36.4 days in 2012 to 32.9 in 2013, a 9.6% decrease. The median wait time dropped from 34 to 24 days.
The 15 most common diagnoses were the same before and after the policy shift. The top three in both time periods were benign neoplasms of the skin, atopic dermatitis, and acne. Their proportion of total new clinic visits was the same in 2012 and 2013. Indeed, the proportion changed for only one of the top 15 diagnoses: warts, which accounted for 9.2% of initial appointments in 2012, but only 7.4% a year later, according to Dr. Herd.
Of note, however, the complexity of the cases seen in the pediatric dermatology clinic rose substantially following the shift to a referral-only policy. In 2012, 40% of initially referred patients were billed as Level 3 complexity, compared with 68% in 2013.
Patient demographics didn’t change, nor did the type of insurance coverage. These findings suggest that access to pediatric dermatology services remained equitable following implementation of the referral-only policy, she continued.
Primary care physicians are probably capable of handling many of the pediatric dermatologic conditions which they now commonly refer to the subspecialty clinic. An interesting topic for future study will be to see if targeted education of primary care clinicians and pediatric residents regarding the top 15 diagnoses seen in the referral-only pediatric dermatology clinic allows the nondermatologists to reduce their referral rate, Dr. Herd said.
She reported having no financial conflicts regarding this study, which was conducted with institutional funds.
COEUR D’ALENE, IDAHO – Switching to a referral-only policy in one pediatric dermatology clinic in a children’s hospital reduced the average wait for an initial appointment by 10%.
"A referral-only system may be an effective method for other pediatric dermatology clinics, as well as other pediatric subspecialties, to decrease patient wait time to see a subspecialist," Dr. Tiffany J. Herd concluded at the annual meeting of the Society for Pediatric Dermatology.
Nationally, pediatric dermatology is very close to the top of the list of pediatric subspecialties in terms of patient wait time. Strategies to reduce wait time for pediatric dermatology are urgently needed, noted Dr. Herd of Children’s Mercy Hospitals and Clinics in Kansas City, Mo.
She presented a retrospective study analyzing the impact of implementing a referral-only policy for new outpatient pediatric dermatology appointments at the medical center. The study entailed reviewing the medical records of 6,316 pediatric dermatology clinic patients seen initially either during January-August of 2012–before the switch to a referral-only policy–or in January-August of 2013, after the policy was in place.
Self-referrals accounted for 24% of all initial appointments in 2012, but only 4% a year later. Mean patient wait time fell from 36.4 days in 2012 to 32.9 in 2013, a 9.6% decrease. The median wait time dropped from 34 to 24 days.
The 15 most common diagnoses were the same before and after the policy shift. The top three in both time periods were benign neoplasms of the skin, atopic dermatitis, and acne. Their proportion of total new clinic visits was the same in 2012 and 2013. Indeed, the proportion changed for only one of the top 15 diagnoses: warts, which accounted for 9.2% of initial appointments in 2012, but only 7.4% a year later, according to Dr. Herd.
Of note, however, the complexity of the cases seen in the pediatric dermatology clinic rose substantially following the shift to a referral-only policy. In 2012, 40% of initially referred patients were billed as Level 3 complexity, compared with 68% in 2013.
Patient demographics didn’t change, nor did the type of insurance coverage. These findings suggest that access to pediatric dermatology services remained equitable following implementation of the referral-only policy, she continued.
Primary care physicians are probably capable of handling many of the pediatric dermatologic conditions which they now commonly refer to the subspecialty clinic. An interesting topic for future study will be to see if targeted education of primary care clinicians and pediatric residents regarding the top 15 diagnoses seen in the referral-only pediatric dermatology clinic allows the nondermatologists to reduce their referral rate, Dr. Herd said.
She reported having no financial conflicts regarding this study, which was conducted with institutional funds.
AT THE SPD ANNUAL MEETING
Key clinical point: The change to a referral-only appointment policy increased the complexity of conditions referred to the pediatric dermatologist.
Major finding: The median wait time for an initial appointment in a pediatric dermatology clinic dropped from 34 to 24 days following a switch to a referral-only appointment policy.
Data source: This retrospective medical chart review involving 6,316 patients compared mean wait times for an initial appointment at a single pediatric dermatology clinic before and after implementation of a referral-only policy.
Disclosures: Dr. Herd reported no financial conflicts with regard to this study, supported by institutional funds.
Early sclerotherapy advised for lymphatic malformations
COEUR D’ALENE, IDAHO – Early therapeutic intervention in children with lymphatic malformations appears to reduce the risk of lesion progression and symptomatic complications, according to a large retrospective study.
"Lymphatic malformations are progressive, but early intervention makes a difference in terms of volume increase and the complication rate. Early intervention should definitely be considered when functional impairment due to local extension is a concern," Dr. Joyce Teng said at the annual meeting of the Society for Pediatric Dermatology.
Lymphatic malformations (LMs) are benign, low-flow congenital vascular anomalies due to abnormal formation of lymphatic vessels in utero. The lesions consist of sequestered lymphatic cysts lined with lymphatic epithelium surrounded by connective tissue stroma. These cysts don’t drain. The result is soft, doughy masses underlying normal-looking epidermis.
Close to two-thirds of LMs occur on the head and neck. They commonly cause pain and swelling, especially during an upper respiratory infection. These head and neck lesions not only cause unsightly facial asymmetry, but as they progress they can press on the airway. Truncal lesions not infrequently send affected patients to the emergency department with acute abdominal pain, explained Dr. Teng, director of pediatric dermatology at Stanford (Calif.) University.
There is no ideal therapy for LMs. Management options include conservative observation, surgical excision, sclerotherapy, and potentially disease-modifying medications, with sildenafil and sirolimus among the agents under study.
In children, sclerotherapy is a particularly attractive alternative to surgical excision. The Stanford experience as described by Dr. Teng indicates that doxycycline and ethanolamine are equally safe and effective sclerosants for this purpose, as is the Japanese agent OK-432, unavailable in North America or Europe.
Dr. Teng presented a retrospective chart review of 336 patients under age 20 years with LMs in Stanford’s large clinical database. Half were female. The LMs were first noted at a mean age of 2.1 years and a median of 2 months. Forty-three percent were macrocystic as defined by septated cystic spaces in excess of 2 cc, while another 35% of patients had a mix of macro- and microcysts.
Disease progression was defined as lesion enlargement at a rate greater than the patient’s growth rate or worsening signs and symptoms. The LM progression rate was 47% in childhood.
The median age at the time of progression was 4.9 years. Consistent with the findings of an earlier study by investigators at Harvard University, Boston, the rate of rapid progression in the Stanford study was greater among 15- to 20-year-olds than in children, probably due to the influence of pubertal hormonal changes. In the retrospective 441-patient Harvard study, the LM progression rate was 40% in childhood, 64% in adolescence, and 95% lifetime (J. Craniofac. Surg. 2012;23:149-52).
Dr. Teng and coworkers found that observation without active treatment was associated with a 2.1-fold increased risk of progression. Also, older age at the time of diagnosis was associated with a higher risk of complications. Indeed, the odds of progression rose by 17% per year of age at diagnosis of LM.
In contrast, factors not associated with progression risk in the Stanford study were gender, lesion location or subtype, or having LMs at multiple sites.
Forty-one children with LMs on the head and neck underwent sclerotherapy at a median age of 1-2 years.
Among 19 patients treated with doxycycline, there were two recurrences. Outcomes were rated as excellent in 53% of cases, meaning two investigators agreed that the two sides of the face looked nearly the same post treatment. Outcomes of doxycycline sclerotherapy were rated good in another 26%. Among 12 ethanolamine-treated patients, outcomes were rated excellent in 42% and good in 25%. One of the 12 patients experienced an LM recurrence, as did 3 of 10 treated with OK-432. No procedural complications occurred in the ethanolamine group. One doxycycline-treated patient experienced a treatment-related hemorrhage.
In a multivariate analysis, doxycycline and ethanolamine didn’t differ in effectiveness. Both performed best in treating macrocystic lesions, where 90% of patients experienced marked LM volume reduction, as did 73% of patients with mixed lesions. The success rate was lowest when treating purely microcystic lesions.
"I’ve talked to lots of interventional radiologists. These cases aren’t all that common, and their choice of sclerosant seems to really depend upon their training and comfort level," Dr. Teng said.
She advised getting a baseline magnetic resonance image (MRI) to confirm the diagnosis of LM and to determine the baseline extent of disease in order to guide future decisions about intervention. In addition, a post-treatment MRI showing LM volume reduction provides objective evidence of therapeutic success.
One audience member, citing the high cost of MRIs, asked if there are less expensive ways to measure the magnitude of treatment response. Dr. Teng replied that imaging is best. She added, however, that the cost of an MRI is reduced by about $800 when sedation isn’t required. And a Stanford study has shown that the use of a child-friendly teaching kit enables most children to undergo an MRI without sedation.
"It’s like a video game they take home and play for a month. When they come back for their MRI more than 80% of kids don’t need sedation anymore. It really cuts down the cost," she said.
Dr. Teng credited University of California, Irvine, medical student Viraat Patel as deserving primary authorship status for this study, conducted under her supervision. The study was supported by the Society for Pediatric Dermatology. Dr. Teng had no financial conflicts.
COEUR D’ALENE, IDAHO – Early therapeutic intervention in children with lymphatic malformations appears to reduce the risk of lesion progression and symptomatic complications, according to a large retrospective study.
"Lymphatic malformations are progressive, but early intervention makes a difference in terms of volume increase and the complication rate. Early intervention should definitely be considered when functional impairment due to local extension is a concern," Dr. Joyce Teng said at the annual meeting of the Society for Pediatric Dermatology.
Lymphatic malformations (LMs) are benign, low-flow congenital vascular anomalies due to abnormal formation of lymphatic vessels in utero. The lesions consist of sequestered lymphatic cysts lined with lymphatic epithelium surrounded by connective tissue stroma. These cysts don’t drain. The result is soft, doughy masses underlying normal-looking epidermis.
Close to two-thirds of LMs occur on the head and neck. They commonly cause pain and swelling, especially during an upper respiratory infection. These head and neck lesions not only cause unsightly facial asymmetry, but as they progress they can press on the airway. Truncal lesions not infrequently send affected patients to the emergency department with acute abdominal pain, explained Dr. Teng, director of pediatric dermatology at Stanford (Calif.) University.
There is no ideal therapy for LMs. Management options include conservative observation, surgical excision, sclerotherapy, and potentially disease-modifying medications, with sildenafil and sirolimus among the agents under study.
In children, sclerotherapy is a particularly attractive alternative to surgical excision. The Stanford experience as described by Dr. Teng indicates that doxycycline and ethanolamine are equally safe and effective sclerosants for this purpose, as is the Japanese agent OK-432, unavailable in North America or Europe.
Dr. Teng presented a retrospective chart review of 336 patients under age 20 years with LMs in Stanford’s large clinical database. Half were female. The LMs were first noted at a mean age of 2.1 years and a median of 2 months. Forty-three percent were macrocystic as defined by septated cystic spaces in excess of 2 cc, while another 35% of patients had a mix of macro- and microcysts.
Disease progression was defined as lesion enlargement at a rate greater than the patient’s growth rate or worsening signs and symptoms. The LM progression rate was 47% in childhood.
The median age at the time of progression was 4.9 years. Consistent with the findings of an earlier study by investigators at Harvard University, Boston, the rate of rapid progression in the Stanford study was greater among 15- to 20-year-olds than in children, probably due to the influence of pubertal hormonal changes. In the retrospective 441-patient Harvard study, the LM progression rate was 40% in childhood, 64% in adolescence, and 95% lifetime (J. Craniofac. Surg. 2012;23:149-52).
Dr. Teng and coworkers found that observation without active treatment was associated with a 2.1-fold increased risk of progression. Also, older age at the time of diagnosis was associated with a higher risk of complications. Indeed, the odds of progression rose by 17% per year of age at diagnosis of LM.
In contrast, factors not associated with progression risk in the Stanford study were gender, lesion location or subtype, or having LMs at multiple sites.
Forty-one children with LMs on the head and neck underwent sclerotherapy at a median age of 1-2 years.
Among 19 patients treated with doxycycline, there were two recurrences. Outcomes were rated as excellent in 53% of cases, meaning two investigators agreed that the two sides of the face looked nearly the same post treatment. Outcomes of doxycycline sclerotherapy were rated good in another 26%. Among 12 ethanolamine-treated patients, outcomes were rated excellent in 42% and good in 25%. One of the 12 patients experienced an LM recurrence, as did 3 of 10 treated with OK-432. No procedural complications occurred in the ethanolamine group. One doxycycline-treated patient experienced a treatment-related hemorrhage.
In a multivariate analysis, doxycycline and ethanolamine didn’t differ in effectiveness. Both performed best in treating macrocystic lesions, where 90% of patients experienced marked LM volume reduction, as did 73% of patients with mixed lesions. The success rate was lowest when treating purely microcystic lesions.
"I’ve talked to lots of interventional radiologists. These cases aren’t all that common, and their choice of sclerosant seems to really depend upon their training and comfort level," Dr. Teng said.
She advised getting a baseline magnetic resonance image (MRI) to confirm the diagnosis of LM and to determine the baseline extent of disease in order to guide future decisions about intervention. In addition, a post-treatment MRI showing LM volume reduction provides objective evidence of therapeutic success.
One audience member, citing the high cost of MRIs, asked if there are less expensive ways to measure the magnitude of treatment response. Dr. Teng replied that imaging is best. She added, however, that the cost of an MRI is reduced by about $800 when sedation isn’t required. And a Stanford study has shown that the use of a child-friendly teaching kit enables most children to undergo an MRI without sedation.
"It’s like a video game they take home and play for a month. When they come back for their MRI more than 80% of kids don’t need sedation anymore. It really cuts down the cost," she said.
Dr. Teng credited University of California, Irvine, medical student Viraat Patel as deserving primary authorship status for this study, conducted under her supervision. The study was supported by the Society for Pediatric Dermatology. Dr. Teng had no financial conflicts.
COEUR D’ALENE, IDAHO – Early therapeutic intervention in children with lymphatic malformations appears to reduce the risk of lesion progression and symptomatic complications, according to a large retrospective study.
"Lymphatic malformations are progressive, but early intervention makes a difference in terms of volume increase and the complication rate. Early intervention should definitely be considered when functional impairment due to local extension is a concern," Dr. Joyce Teng said at the annual meeting of the Society for Pediatric Dermatology.
Lymphatic malformations (LMs) are benign, low-flow congenital vascular anomalies due to abnormal formation of lymphatic vessels in utero. The lesions consist of sequestered lymphatic cysts lined with lymphatic epithelium surrounded by connective tissue stroma. These cysts don’t drain. The result is soft, doughy masses underlying normal-looking epidermis.
Close to two-thirds of LMs occur on the head and neck. They commonly cause pain and swelling, especially during an upper respiratory infection. These head and neck lesions not only cause unsightly facial asymmetry, but as they progress they can press on the airway. Truncal lesions not infrequently send affected patients to the emergency department with acute abdominal pain, explained Dr. Teng, director of pediatric dermatology at Stanford (Calif.) University.
There is no ideal therapy for LMs. Management options include conservative observation, surgical excision, sclerotherapy, and potentially disease-modifying medications, with sildenafil and sirolimus among the agents under study.
In children, sclerotherapy is a particularly attractive alternative to surgical excision. The Stanford experience as described by Dr. Teng indicates that doxycycline and ethanolamine are equally safe and effective sclerosants for this purpose, as is the Japanese agent OK-432, unavailable in North America or Europe.
Dr. Teng presented a retrospective chart review of 336 patients under age 20 years with LMs in Stanford’s large clinical database. Half were female. The LMs were first noted at a mean age of 2.1 years and a median of 2 months. Forty-three percent were macrocystic as defined by septated cystic spaces in excess of 2 cc, while another 35% of patients had a mix of macro- and microcysts.
Disease progression was defined as lesion enlargement at a rate greater than the patient’s growth rate or worsening signs and symptoms. The LM progression rate was 47% in childhood.
The median age at the time of progression was 4.9 years. Consistent with the findings of an earlier study by investigators at Harvard University, Boston, the rate of rapid progression in the Stanford study was greater among 15- to 20-year-olds than in children, probably due to the influence of pubertal hormonal changes. In the retrospective 441-patient Harvard study, the LM progression rate was 40% in childhood, 64% in adolescence, and 95% lifetime (J. Craniofac. Surg. 2012;23:149-52).
Dr. Teng and coworkers found that observation without active treatment was associated with a 2.1-fold increased risk of progression. Also, older age at the time of diagnosis was associated with a higher risk of complications. Indeed, the odds of progression rose by 17% per year of age at diagnosis of LM.
In contrast, factors not associated with progression risk in the Stanford study were gender, lesion location or subtype, or having LMs at multiple sites.
Forty-one children with LMs on the head and neck underwent sclerotherapy at a median age of 1-2 years.
Among 19 patients treated with doxycycline, there were two recurrences. Outcomes were rated as excellent in 53% of cases, meaning two investigators agreed that the two sides of the face looked nearly the same post treatment. Outcomes of doxycycline sclerotherapy were rated good in another 26%. Among 12 ethanolamine-treated patients, outcomes were rated excellent in 42% and good in 25%. One of the 12 patients experienced an LM recurrence, as did 3 of 10 treated with OK-432. No procedural complications occurred in the ethanolamine group. One doxycycline-treated patient experienced a treatment-related hemorrhage.
In a multivariate analysis, doxycycline and ethanolamine didn’t differ in effectiveness. Both performed best in treating macrocystic lesions, where 90% of patients experienced marked LM volume reduction, as did 73% of patients with mixed lesions. The success rate was lowest when treating purely microcystic lesions.
"I’ve talked to lots of interventional radiologists. These cases aren’t all that common, and their choice of sclerosant seems to really depend upon their training and comfort level," Dr. Teng said.
She advised getting a baseline magnetic resonance image (MRI) to confirm the diagnosis of LM and to determine the baseline extent of disease in order to guide future decisions about intervention. In addition, a post-treatment MRI showing LM volume reduction provides objective evidence of therapeutic success.
One audience member, citing the high cost of MRIs, asked if there are less expensive ways to measure the magnitude of treatment response. Dr. Teng replied that imaging is best. She added, however, that the cost of an MRI is reduced by about $800 when sedation isn’t required. And a Stanford study has shown that the use of a child-friendly teaching kit enables most children to undergo an MRI without sedation.
"It’s like a video game they take home and play for a month. When they come back for their MRI more than 80% of kids don’t need sedation anymore. It really cuts down the cost," she said.
Dr. Teng credited University of California, Irvine, medical student Viraat Patel as deserving primary authorship status for this study, conducted under her supervision. The study was supported by the Society for Pediatric Dermatology. Dr. Teng had no financial conflicts.
EXPERT ANALYSIS FROM THE SPD ANNUAL MEETING
Key clinical point: Intervene in the first few years of life in order to minimize the likelihood of later progression of lymphatic malformations.
Major finding: Among 41 children who underwent sclerotherapy before age 2 years for lymphatic malformations on the head and neck, only 5 experienced recurrences.
Data source: This was a retrospective chart review of 336 children and adolescents with lymphatic malformations.
Disclosures: The study was supported by the Society for Pediatric Dermatology. The presenter reported having no financial conflicts.