User login
Diabetes increases risk of atrial fibrillation
BARCELONA – Adults with diabetes mellitus are at increased risk of subsequent new-onset atrial fibrillation – and the younger the age at diabetes onset, the greater the likelihood of developing the arrhythmia.
That’s the key finding from a Danish national registry study in which all 5,168,416 Danish adults without atrial fibrillation in 1996 were followed through 2012 for development of atrial fibrillation (AF). The study population included 75,197 Danes with diabetes at baseline and another 235,327 who developed the disease during follow-up, Dr. Jannik L. Pallisgaard explained at the annual congress of the European Society of Cardiology.
During follow-up, 5.6% of those with diabetes and 3.3% of those without diabetes developed AF. The mean time from diabetes onset to AF onset was 5 years, reported Dr. Pallisgaard of the University of Copenhagen.
"What was particularly interesting, I think, is that we found the youngest patients were the group at highest risk" of developing AF, he said. "We suggest that starting at the onset of diabetes, routine pulse palpation, ECGs, and focused patient interviews asking about any signs of atrial fibrillation could prove beneficial in detecting the arrhythmia."
The incidence rate ratio for developing AF per 1,000 person-years of follow-up was roughly 2.5-fold greater in 18- to 39-year-olds with diabetes than in their nondiabetic peers. From this peak rate in young adults, the magnitude of relative risk dropped in stepwise fashion with age: The variability in risk was lower in 40- to 60-year-old diabetics than in the 18- to 39-year olds and lower still in 65- to 74-year olds. Variability in the incidence rate ratio finally bottomed out at a still statistically significant 1.3-fold increased risk of developing AF in diabetic individuals ages 75 and older compared to their nondiabetic peers.
Dr. Pallisgaard noted that while the relative risk of developing AF was greatest in the 18- to 39-year-olds, the absolute number of new cases of AF was far greater in older patients because there were so many more of them with diabetes. He cautioned that as the obesity epidemic leads to more and more patients developing type 2 diabetes at younger ages, more cases of AF can be expected in young adults.
Dr. Pallisgaard cited two likely mechanisms underlying the observed increased risk of AF in diabetic patients: left ventricular hypertrophy and vascular inflammation, which are both often present in the diabetic population.
He reported having no financial conflicts regarding this study, conducted with Danish institutional research funds.
BARCELONA – Adults with diabetes mellitus are at increased risk of subsequent new-onset atrial fibrillation – and the younger the age at diabetes onset, the greater the likelihood of developing the arrhythmia.
That’s the key finding from a Danish national registry study in which all 5,168,416 Danish adults without atrial fibrillation in 1996 were followed through 2012 for development of atrial fibrillation (AF). The study population included 75,197 Danes with diabetes at baseline and another 235,327 who developed the disease during follow-up, Dr. Jannik L. Pallisgaard explained at the annual congress of the European Society of Cardiology.
During follow-up, 5.6% of those with diabetes and 3.3% of those without diabetes developed AF. The mean time from diabetes onset to AF onset was 5 years, reported Dr. Pallisgaard of the University of Copenhagen.
"What was particularly interesting, I think, is that we found the youngest patients were the group at highest risk" of developing AF, he said. "We suggest that starting at the onset of diabetes, routine pulse palpation, ECGs, and focused patient interviews asking about any signs of atrial fibrillation could prove beneficial in detecting the arrhythmia."
The incidence rate ratio for developing AF per 1,000 person-years of follow-up was roughly 2.5-fold greater in 18- to 39-year-olds with diabetes than in their nondiabetic peers. From this peak rate in young adults, the magnitude of relative risk dropped in stepwise fashion with age: The variability in risk was lower in 40- to 60-year-old diabetics than in the 18- to 39-year olds and lower still in 65- to 74-year olds. Variability in the incidence rate ratio finally bottomed out at a still statistically significant 1.3-fold increased risk of developing AF in diabetic individuals ages 75 and older compared to their nondiabetic peers.
Dr. Pallisgaard noted that while the relative risk of developing AF was greatest in the 18- to 39-year-olds, the absolute number of new cases of AF was far greater in older patients because there were so many more of them with diabetes. He cautioned that as the obesity epidemic leads to more and more patients developing type 2 diabetes at younger ages, more cases of AF can be expected in young adults.
Dr. Pallisgaard cited two likely mechanisms underlying the observed increased risk of AF in diabetic patients: left ventricular hypertrophy and vascular inflammation, which are both often present in the diabetic population.
He reported having no financial conflicts regarding this study, conducted with Danish institutional research funds.
BARCELONA – Adults with diabetes mellitus are at increased risk of subsequent new-onset atrial fibrillation – and the younger the age at diabetes onset, the greater the likelihood of developing the arrhythmia.
That’s the key finding from a Danish national registry study in which all 5,168,416 Danish adults without atrial fibrillation in 1996 were followed through 2012 for development of atrial fibrillation (AF). The study population included 75,197 Danes with diabetes at baseline and another 235,327 who developed the disease during follow-up, Dr. Jannik L. Pallisgaard explained at the annual congress of the European Society of Cardiology.
During follow-up, 5.6% of those with diabetes and 3.3% of those without diabetes developed AF. The mean time from diabetes onset to AF onset was 5 years, reported Dr. Pallisgaard of the University of Copenhagen.
"What was particularly interesting, I think, is that we found the youngest patients were the group at highest risk" of developing AF, he said. "We suggest that starting at the onset of diabetes, routine pulse palpation, ECGs, and focused patient interviews asking about any signs of atrial fibrillation could prove beneficial in detecting the arrhythmia."
The incidence rate ratio for developing AF per 1,000 person-years of follow-up was roughly 2.5-fold greater in 18- to 39-year-olds with diabetes than in their nondiabetic peers. From this peak rate in young adults, the magnitude of relative risk dropped in stepwise fashion with age: The variability in risk was lower in 40- to 60-year-old diabetics than in the 18- to 39-year olds and lower still in 65- to 74-year olds. Variability in the incidence rate ratio finally bottomed out at a still statistically significant 1.3-fold increased risk of developing AF in diabetic individuals ages 75 and older compared to their nondiabetic peers.
Dr. Pallisgaard noted that while the relative risk of developing AF was greatest in the 18- to 39-year-olds, the absolute number of new cases of AF was far greater in older patients because there were so many more of them with diabetes. He cautioned that as the obesity epidemic leads to more and more patients developing type 2 diabetes at younger ages, more cases of AF can be expected in young adults.
Dr. Pallisgaard cited two likely mechanisms underlying the observed increased risk of AF in diabetic patients: left ventricular hypertrophy and vascular inflammation, which are both often present in the diabetic population.
He reported having no financial conflicts regarding this study, conducted with Danish institutional research funds.
AT THE ESC CONGRESS 2014
Key clinical point: Starting at the onset of diabetes, routine pulse palpation, ECGs, and patient interviews focused on signs of atrial fibrillation might improve detection of the arrhythmia.
Major finding: During follow-up, 5.6% of those with diabetes and 3.3% of those without diabetes developed AF.
Data source: This was a national registry study including all of the nearly 5.2 million Danish adults without atrial fibrillation in 1996. Follow-up ran through 2012.
Disclosures: The presenter reported having no financial conflicts regarding this study, funded by Danish institutional research grants.
Testosterone replacement enhances aerobic capacity in mobility-limited men
CHICAGO – Testosterone replacement therapy in mobility-impaired, sedentary older men with low testosterone levels improved two different measures of aerobic capacity in a placebo-controlled, randomized 6-month clinical trial.
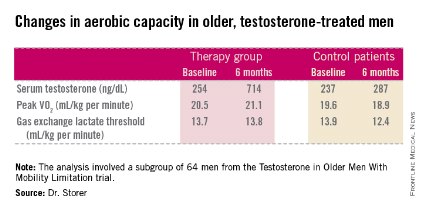
The age-related decline in peak oxygen uptake during exercise, or peak VO2, was 3.4-fold less in the testosterone-treated men than would be expected based on published population norms. Moreover, the rate of decline in peak VO2 in placebo-treated controls was nearly twice the expected rate for the age-matched general population; this accelerated decline was probably due to their limited mobility and low testosterone levels, Thomas W. Storer, Ph.D., said at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.

"This study is the first to show enhanced endurance performance as a result of testosterone therapy in men who have difficulty performing some physical tasks but are otherwise healthy. This is something we think is going to be clinically meaningful," said Dr. Storer, director of the exercise physiology and physical performance laboratory at Brigham and Women’s Hospital, Boston.
He presented an analysis of a subset of participants in the prospective, randomized Testosterone in Older Men With Mobility Limitation (TOM) trial, in which subjects with low total or free testosterone levels were placed on 10 mg/day of testosterone gel or placebo gel for 6 months. The subgroup consisted of 64 men, mean age 73, who underwent formal testing of aerobic capacity via measurement of changes over time in peak VO2 and gas exchange lactate threshold during symptom-limited exercise cycling.
The gas exchange lactate threshold is a good functional measure of the ability to do work over a prolonged period. The rate remained steady during the 6-month study in the testosterone-treated men but declined significantly – and to a greater-than-expected extent based on normative values – in the placebo-treated controls (see chart).
"We think the mechanisms involved in this benefit are many," Dr. Storer said in an interview. Among them are testosterone’s demonstrated ability to increase muscle mass and thereby generate more force during exercise; increased RBC formation; stimulation of tissue capillarity in order to allow more blood flow to the exercising muscle; and stimulation of mitochondrial biogenesis, which increases oxygen uptake by muscle tissue.
However, he added that although these results are quite promising, he doesn’t think this work is ready for prime time application in daily clinical practice. He plans to further evaluate the safety of this treatment and the durability of the effects in a study with larger patient numbers and longer treatment.
The study was funded by the National Institute on Aging, the Claude D. Pepper Older Americans Independence Center, and Boston University. Dr. Storer reported having no relevant financial conflicts.
CHICAGO – Testosterone replacement therapy in mobility-impaired, sedentary older men with low testosterone levels improved two different measures of aerobic capacity in a placebo-controlled, randomized 6-month clinical trial.
The age-related decline in peak oxygen uptake during exercise, or peak VO2, was 3.4-fold less in the testosterone-treated men than would be expected based on published population norms. Moreover, the rate of decline in peak VO2 in placebo-treated controls was nearly twice the expected rate for the age-matched general population; this accelerated decline was probably due to their limited mobility and low testosterone levels, Thomas W. Storer, Ph.D., said at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.

"This study is the first to show enhanced endurance performance as a result of testosterone therapy in men who have difficulty performing some physical tasks but are otherwise healthy. This is something we think is going to be clinically meaningful," said Dr. Storer, director of the exercise physiology and physical performance laboratory at Brigham and Women’s Hospital, Boston.
He presented an analysis of a subset of participants in the prospective, randomized Testosterone in Older Men With Mobility Limitation (TOM) trial, in which subjects with low total or free testosterone levels were placed on 10 mg/day of testosterone gel or placebo gel for 6 months. The subgroup consisted of 64 men, mean age 73, who underwent formal testing of aerobic capacity via measurement of changes over time in peak VO2 and gas exchange lactate threshold during symptom-limited exercise cycling.
The gas exchange lactate threshold is a good functional measure of the ability to do work over a prolonged period. The rate remained steady during the 6-month study in the testosterone-treated men but declined significantly – and to a greater-than-expected extent based on normative values – in the placebo-treated controls (see chart).
"We think the mechanisms involved in this benefit are many," Dr. Storer said in an interview. Among them are testosterone’s demonstrated ability to increase muscle mass and thereby generate more force during exercise; increased RBC formation; stimulation of tissue capillarity in order to allow more blood flow to the exercising muscle; and stimulation of mitochondrial biogenesis, which increases oxygen uptake by muscle tissue.
However, he added that although these results are quite promising, he doesn’t think this work is ready for prime time application in daily clinical practice. He plans to further evaluate the safety of this treatment and the durability of the effects in a study with larger patient numbers and longer treatment.
The study was funded by the National Institute on Aging, the Claude D. Pepper Older Americans Independence Center, and Boston University. Dr. Storer reported having no relevant financial conflicts.
CHICAGO – Testosterone replacement therapy in mobility-impaired, sedentary older men with low testosterone levels improved two different measures of aerobic capacity in a placebo-controlled, randomized 6-month clinical trial.
The age-related decline in peak oxygen uptake during exercise, or peak VO2, was 3.4-fold less in the testosterone-treated men than would be expected based on published population norms. Moreover, the rate of decline in peak VO2 in placebo-treated controls was nearly twice the expected rate for the age-matched general population; this accelerated decline was probably due to their limited mobility and low testosterone levels, Thomas W. Storer, Ph.D., said at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.

"This study is the first to show enhanced endurance performance as a result of testosterone therapy in men who have difficulty performing some physical tasks but are otherwise healthy. This is something we think is going to be clinically meaningful," said Dr. Storer, director of the exercise physiology and physical performance laboratory at Brigham and Women’s Hospital, Boston.
He presented an analysis of a subset of participants in the prospective, randomized Testosterone in Older Men With Mobility Limitation (TOM) trial, in which subjects with low total or free testosterone levels were placed on 10 mg/day of testosterone gel or placebo gel for 6 months. The subgroup consisted of 64 men, mean age 73, who underwent formal testing of aerobic capacity via measurement of changes over time in peak VO2 and gas exchange lactate threshold during symptom-limited exercise cycling.
The gas exchange lactate threshold is a good functional measure of the ability to do work over a prolonged period. The rate remained steady during the 6-month study in the testosterone-treated men but declined significantly – and to a greater-than-expected extent based on normative values – in the placebo-treated controls (see chart).
"We think the mechanisms involved in this benefit are many," Dr. Storer said in an interview. Among them are testosterone’s demonstrated ability to increase muscle mass and thereby generate more force during exercise; increased RBC formation; stimulation of tissue capillarity in order to allow more blood flow to the exercising muscle; and stimulation of mitochondrial biogenesis, which increases oxygen uptake by muscle tissue.
However, he added that although these results are quite promising, he doesn’t think this work is ready for prime time application in daily clinical practice. He plans to further evaluate the safety of this treatment and the durability of the effects in a study with larger patient numbers and longer treatment.
The study was funded by the National Institute on Aging, the Claude D. Pepper Older Americans Independence Center, and Boston University. Dr. Storer reported having no relevant financial conflicts.
AT ICE/ENDO 2014
Key clinical point: Testosterone replacement therapy neutralizes the age-related decline in aerobic capacity in older men with low testosterone and mobility limitation.
Major finding: The age-related decline in peak VO2 in testosterone takers was 3.4-fold less than expected based on age-related norms.
Data source: A subanalysis of data from the randomized, prospective TOM trial, in which participants with low testosterone were randomized to 10 mg/day of testosterone gel or placebo for 6 months.
Disclosures: TOM was funded by the National Institute on Aging, the Claude D. Pepper Older Americans Independence Center, and Boston University.
Mifepristone for Cushing’s brings sustained weight loss
CHICAGO – Mifepristone therapy for Cushing’s syndrome provides an important side benefit: clinically meaningful weight loss that persists over time.
That’s a key finding from the long-term extension phase of the SEISMIC study, a 24-week, multicenter study that led to Food and Drug Administration approval of mifepristone (Korlym) in 2012 as the first and only medication indicated for the treatment of endogenous Cushing’s syndrome.
In the long-term extension phase of the SEISMIC study, mifepristone therapy for up to 3.5 years was associated with a mean 9.3% weight loss from baseline, Dr. Henry G. Fein reported at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
Of patients who lost at least 5% of their body weight during the initial 24-week treatment period in the SEISMIC study, 83% maintained that amount of weight loss through the long-term extension phase. A sustained weight loss of that scope is likely to translate into a reduced risk of cardiovascular disease and metabolic dysfunction, said Dr. Fein of Johns Hopkins University, Baltimore.
Endogenous Cushing’s syndrome is a rare disease, and the phase III SEISMIC study (J. Clin. Endocrinol. Metab. 2012;97:2039-49), while pivotal, was small, involving 50 patients treated with the glucocorticoid receptor antagonist at 300-1,200 mg once daily for 24 weeks. Afterward, 29 patients underwent a 6-week period off drug for safety assessment and then went back on mifepristone for a median of 29.2 months and a maximum of 3.5 years in the extension study.
At baseline, the mean body weight in these 29 patients was 105.4 kg. It dropped to 97.2 kg by week 24. Six weeks later, when patients went back on mifepristone, their mean weight was 98.6 kg. At last follow-up – with patients still on the drug – their mean weight was 95.1 kg, a 9.3% decrease from baseline.
Eighteen of 29 patients achieved a 5% or better weight loss by week 24; at the most recent follow-up, 15 of the 18 (83%) maintained or improved upon that degree of weight loss. Moreover, of the 10 patients who lost at least 10% of their body weight during the first 24 weeks of treatment, 8 maintained that amount of weight loss by study’s end.
Surgery is accepted as the treatment of choice for most patients with Cushing’s syndrome. However, various studies have shown that 10%-45% of surgically treated patients have persistent or recurrent hypercortisolism postoperatively, with accompanying weight gain. That’s where mifepristone plays a key role. In addition, the drug is valuable in patients who aren’t surgical candidates, the endocrinologist noted.
Mifepristone was formerly known as RU-486, "the abortion pill."
The SEISMIC study and its long-term extension were funded by Corcept Therapeutics. Dr. Fein is on the company’s speakers bureau.
CHICAGO – Mifepristone therapy for Cushing’s syndrome provides an important side benefit: clinically meaningful weight loss that persists over time.
That’s a key finding from the long-term extension phase of the SEISMIC study, a 24-week, multicenter study that led to Food and Drug Administration approval of mifepristone (Korlym) in 2012 as the first and only medication indicated for the treatment of endogenous Cushing’s syndrome.
In the long-term extension phase of the SEISMIC study, mifepristone therapy for up to 3.5 years was associated with a mean 9.3% weight loss from baseline, Dr. Henry G. Fein reported at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
Of patients who lost at least 5% of their body weight during the initial 24-week treatment period in the SEISMIC study, 83% maintained that amount of weight loss through the long-term extension phase. A sustained weight loss of that scope is likely to translate into a reduced risk of cardiovascular disease and metabolic dysfunction, said Dr. Fein of Johns Hopkins University, Baltimore.
Endogenous Cushing’s syndrome is a rare disease, and the phase III SEISMIC study (J. Clin. Endocrinol. Metab. 2012;97:2039-49), while pivotal, was small, involving 50 patients treated with the glucocorticoid receptor antagonist at 300-1,200 mg once daily for 24 weeks. Afterward, 29 patients underwent a 6-week period off drug for safety assessment and then went back on mifepristone for a median of 29.2 months and a maximum of 3.5 years in the extension study.
At baseline, the mean body weight in these 29 patients was 105.4 kg. It dropped to 97.2 kg by week 24. Six weeks later, when patients went back on mifepristone, their mean weight was 98.6 kg. At last follow-up – with patients still on the drug – their mean weight was 95.1 kg, a 9.3% decrease from baseline.
Eighteen of 29 patients achieved a 5% or better weight loss by week 24; at the most recent follow-up, 15 of the 18 (83%) maintained or improved upon that degree of weight loss. Moreover, of the 10 patients who lost at least 10% of their body weight during the first 24 weeks of treatment, 8 maintained that amount of weight loss by study’s end.
Surgery is accepted as the treatment of choice for most patients with Cushing’s syndrome. However, various studies have shown that 10%-45% of surgically treated patients have persistent or recurrent hypercortisolism postoperatively, with accompanying weight gain. That’s where mifepristone plays a key role. In addition, the drug is valuable in patients who aren’t surgical candidates, the endocrinologist noted.
Mifepristone was formerly known as RU-486, "the abortion pill."
The SEISMIC study and its long-term extension were funded by Corcept Therapeutics. Dr. Fein is on the company’s speakers bureau.
CHICAGO – Mifepristone therapy for Cushing’s syndrome provides an important side benefit: clinically meaningful weight loss that persists over time.
That’s a key finding from the long-term extension phase of the SEISMIC study, a 24-week, multicenter study that led to Food and Drug Administration approval of mifepristone (Korlym) in 2012 as the first and only medication indicated for the treatment of endogenous Cushing’s syndrome.
In the long-term extension phase of the SEISMIC study, mifepristone therapy for up to 3.5 years was associated with a mean 9.3% weight loss from baseline, Dr. Henry G. Fein reported at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
Of patients who lost at least 5% of their body weight during the initial 24-week treatment period in the SEISMIC study, 83% maintained that amount of weight loss through the long-term extension phase. A sustained weight loss of that scope is likely to translate into a reduced risk of cardiovascular disease and metabolic dysfunction, said Dr. Fein of Johns Hopkins University, Baltimore.
Endogenous Cushing’s syndrome is a rare disease, and the phase III SEISMIC study (J. Clin. Endocrinol. Metab. 2012;97:2039-49), while pivotal, was small, involving 50 patients treated with the glucocorticoid receptor antagonist at 300-1,200 mg once daily for 24 weeks. Afterward, 29 patients underwent a 6-week period off drug for safety assessment and then went back on mifepristone for a median of 29.2 months and a maximum of 3.5 years in the extension study.
At baseline, the mean body weight in these 29 patients was 105.4 kg. It dropped to 97.2 kg by week 24. Six weeks later, when patients went back on mifepristone, their mean weight was 98.6 kg. At last follow-up – with patients still on the drug – their mean weight was 95.1 kg, a 9.3% decrease from baseline.
Eighteen of 29 patients achieved a 5% or better weight loss by week 24; at the most recent follow-up, 15 of the 18 (83%) maintained or improved upon that degree of weight loss. Moreover, of the 10 patients who lost at least 10% of their body weight during the first 24 weeks of treatment, 8 maintained that amount of weight loss by study’s end.
Surgery is accepted as the treatment of choice for most patients with Cushing’s syndrome. However, various studies have shown that 10%-45% of surgically treated patients have persistent or recurrent hypercortisolism postoperatively, with accompanying weight gain. That’s where mifepristone plays a key role. In addition, the drug is valuable in patients who aren’t surgical candidates, the endocrinologist noted.
Mifepristone was formerly known as RU-486, "the abortion pill."
The SEISMIC study and its long-term extension were funded by Corcept Therapeutics. Dr. Fein is on the company’s speakers bureau.
AT ICE/ENDO 2014
Key clinical point: Treatment of Cushing’s syndrome with the glucocorticoid receptor antagonist mifepristone results in sustained significant weight loss, with its attendant cardiometabolic benefits.
Major finding: More than 80% of patients with Cushing’s syndrome who lost at least 5% of their initial body weight during 24 weeks of mifepristone therapy maintained that degree of weight loss for up to 3.5 years.
Data source: The long-term extension study of SEISMIC, an open-label, multicenter study involving 50 mifepristone-treated patients with Cushing’s syndrome.
Disclosures: The study was funded by Corcept Therapeutics, and its presenter is a member of the company’s speakers bureau.
Juvenile facial linear scleroderma is a neurocutaneous disease
COEUR D’ALENE, IDAHO – Juvenile linear scleroderma of the face – traditionally considered a disease limited to the skin – now is more properly viewed as a neurocutaneous disorder, Dr. Francesco Zulian said in his Sidney Hurwitz Memorial Lecture at the annual meeting of the Society for Pediatric Dermatology.
"We need acute neurologic screening at onset of juvenile linear scleroderma of the face and careful monitoring during follow-up," according to Dr. Zulian, chief of pediatric rheumatology at the University of Padua (Italy).
In his series of 77 patients with juvenile linear scleroderma of the face, 46 (60%) proved to have CNS involvement based upon positive brain MRI or EEG findings. The MRI was positive in 73% of patients with neurologic symptoms, which included seizures, chronic headache, cranial nerve palsy, hemiparesis, and behavioral abnormalities.
Moreover, one-quarter of the 24 neurologically asymptomatic patients had organic brain lesions, as evidenced by multiple white and gray matter involvement on MRI. Similarly, 27% of neurologically asymptomatic patients had abnormal EEG findings.
"I am thinking – and other people agree with me – that probably we are dealing with a neurocutaneous condition," he said.
The onset of localized scleroderma of the face occurred at an average age of 5.3 years, which is 2 years earlier than disease onset in patients with juvenile localized scleroderma at other sites. Patients with linear scleroderma of the face who developed neurologic symptoms did so within 5 years of skin disease onset.
How best to monitor patients with juvenile linear scleroderma of the face for occult brain involvement remains an unresolved issue. MRI is expensive and requires sedation in children. Dr. Zulian has turned instead to cone beam CT, a relatively new imaging technique used in maxillofacial surgery and dentistry. It differs from standard CT in that it uses less radiation, takes less time, doesn’t involve sedation, and costs significantly less than MRI or conventional CT. These advantages permit more frequent patient monitoring.
Cone beam CT allows for the creation of three-dimensional images so that physicians can calculate the size of soft tissue and bone abnormalities and monitor their changes over time. Currently, Dr. Zulian is evaluating the utility of this imaging method in a prospective study involving 15 patients. He said he plans to complete the study and present the findings next year.
Dr. Zulian reported having no relevant financial conflicts.
COEUR D’ALENE, IDAHO – Juvenile linear scleroderma of the face – traditionally considered a disease limited to the skin – now is more properly viewed as a neurocutaneous disorder, Dr. Francesco Zulian said in his Sidney Hurwitz Memorial Lecture at the annual meeting of the Society for Pediatric Dermatology.
"We need acute neurologic screening at onset of juvenile linear scleroderma of the face and careful monitoring during follow-up," according to Dr. Zulian, chief of pediatric rheumatology at the University of Padua (Italy).
In his series of 77 patients with juvenile linear scleroderma of the face, 46 (60%) proved to have CNS involvement based upon positive brain MRI or EEG findings. The MRI was positive in 73% of patients with neurologic symptoms, which included seizures, chronic headache, cranial nerve palsy, hemiparesis, and behavioral abnormalities.
Moreover, one-quarter of the 24 neurologically asymptomatic patients had organic brain lesions, as evidenced by multiple white and gray matter involvement on MRI. Similarly, 27% of neurologically asymptomatic patients had abnormal EEG findings.
"I am thinking – and other people agree with me – that probably we are dealing with a neurocutaneous condition," he said.
The onset of localized scleroderma of the face occurred at an average age of 5.3 years, which is 2 years earlier than disease onset in patients with juvenile localized scleroderma at other sites. Patients with linear scleroderma of the face who developed neurologic symptoms did so within 5 years of skin disease onset.
How best to monitor patients with juvenile linear scleroderma of the face for occult brain involvement remains an unresolved issue. MRI is expensive and requires sedation in children. Dr. Zulian has turned instead to cone beam CT, a relatively new imaging technique used in maxillofacial surgery and dentistry. It differs from standard CT in that it uses less radiation, takes less time, doesn’t involve sedation, and costs significantly less than MRI or conventional CT. These advantages permit more frequent patient monitoring.
Cone beam CT allows for the creation of three-dimensional images so that physicians can calculate the size of soft tissue and bone abnormalities and monitor their changes over time. Currently, Dr. Zulian is evaluating the utility of this imaging method in a prospective study involving 15 patients. He said he plans to complete the study and present the findings next year.
Dr. Zulian reported having no relevant financial conflicts.
COEUR D’ALENE, IDAHO – Juvenile linear scleroderma of the face – traditionally considered a disease limited to the skin – now is more properly viewed as a neurocutaneous disorder, Dr. Francesco Zulian said in his Sidney Hurwitz Memorial Lecture at the annual meeting of the Society for Pediatric Dermatology.
"We need acute neurologic screening at onset of juvenile linear scleroderma of the face and careful monitoring during follow-up," according to Dr. Zulian, chief of pediatric rheumatology at the University of Padua (Italy).
In his series of 77 patients with juvenile linear scleroderma of the face, 46 (60%) proved to have CNS involvement based upon positive brain MRI or EEG findings. The MRI was positive in 73% of patients with neurologic symptoms, which included seizures, chronic headache, cranial nerve palsy, hemiparesis, and behavioral abnormalities.
Moreover, one-quarter of the 24 neurologically asymptomatic patients had organic brain lesions, as evidenced by multiple white and gray matter involvement on MRI. Similarly, 27% of neurologically asymptomatic patients had abnormal EEG findings.
"I am thinking – and other people agree with me – that probably we are dealing with a neurocutaneous condition," he said.
The onset of localized scleroderma of the face occurred at an average age of 5.3 years, which is 2 years earlier than disease onset in patients with juvenile localized scleroderma at other sites. Patients with linear scleroderma of the face who developed neurologic symptoms did so within 5 years of skin disease onset.
How best to monitor patients with juvenile linear scleroderma of the face for occult brain involvement remains an unresolved issue. MRI is expensive and requires sedation in children. Dr. Zulian has turned instead to cone beam CT, a relatively new imaging technique used in maxillofacial surgery and dentistry. It differs from standard CT in that it uses less radiation, takes less time, doesn’t involve sedation, and costs significantly less than MRI or conventional CT. These advantages permit more frequent patient monitoring.
Cone beam CT allows for the creation of three-dimensional images so that physicians can calculate the size of soft tissue and bone abnormalities and monitor their changes over time. Currently, Dr. Zulian is evaluating the utility of this imaging method in a prospective study involving 15 patients. He said he plans to complete the study and present the findings next year.
Dr. Zulian reported having no relevant financial conflicts.
EXPERT ANALYSIS FROM THE SPD ANNUAL MEETING
Autologous punch grafts unequalled for segmental vitiligo
COEUR D’ALENE, IDAHO – Autologous grafting is a treatment of unmatched effectiveness for pediatric patients with segmental vitiligo unresponsive to medical modalities, according to Dr. Pearl E. Grimes, director of the Vitiligo and Pigmentation Institute of Southern California and a dermatologist at the University of California, Los Angeles.
"Segmental vitiligo is really the only type of vitiligo I think I can cure, and that’s because we get our absolute best responses with autologous grafting in combination with phototherapy," she said at the annual meeting of the Society for Pediatric Dermatology.
She favors the use of 1-mm punch grafts over more elaborate techniques such as split-thickness grafts, blister roof grafts, and cultured cell transplantation. Autologous grafting using 1-mm punch grafts is simple, and long-term follow-up studies have documented the persistence of repigmentation for more than 5 years in patients with segmental vitiligo.
"It’s so, so easy," Dr. Grimes said. "And the great thing about punch grafts is this technique doesn’t require a lot of special equipment, unlike sheet grafts and the other methods. All you need are iris scissors and jeweler’s forceps."
"I’ve harvested anywhere from 10 to 250 grafts at one setting. We anesthetize a donor area on the hip, harvest the 1-mm punch grafts, place them in saline in sterile petri dishes, remove 1-mm punch grafts from the anesthetized recipient site, and then with jeweler’s forceps you place those harvested 1-mm grafts in those 1-mm recipient holes. You cover the areas with Steri-Strips, which stay in place for 7 days. Then you remove them and start phototherapy, either narrow-band UVB, PUVA [psoralen and UVA], or with an excimer laser," she explained.
In her experience, aggressive topical therapy using high-potency corticosteroids and tacrolimus (Protopic) often provides good results in treating segmental vitiligo on the face.
"If you have segmental vitiligo on the trunk and extremity areas, it’s much more difficult to get good results with topical therapies. Those are the patients who ultimately will need to be grafted," according to the dermatologist.
She stressed that when using autologous grafting to treat patients with generalized vitiligo, it’s imperative to stabilize them beforehand. This is accomplished using systemic steroids: for example, 5-10 mg/day of prednisone for 2 weeks, or mini-pulse dosing of betamethasone at 5 mg twice weekly.
"When I see kids with segmental vitiligo in my initial consultation, I always tell them and their parents that if I were going to get vitiligo, I’d choose segmental versus the generalized variety. That’s because of all the types of vitiligo that we see, the segmental variety is the most predictable. It burns itself out in about 95% of patients in that first year, so we know that in the overwhelming majority of patients it’s not going to spread beyond that dermatome," Dr. Grimes said.
She made a plea for physicians to be more aggressive in treating childhood vitiligo. Often they are reluctant to treat. Yet it has been shown across the board, for all treatment modalities, that efficacy is significantly greater in children than adults.
"Children are the ones who are more likely to give you that greater than 75% repigmentation. It’s a great reason to treat children early," Dr. Grimes said.
She reported having performed clinical research and/or serving as a consultant to a dozen pharmaceutical and cosmetics companies.
COEUR D’ALENE, IDAHO – Autologous grafting is a treatment of unmatched effectiveness for pediatric patients with segmental vitiligo unresponsive to medical modalities, according to Dr. Pearl E. Grimes, director of the Vitiligo and Pigmentation Institute of Southern California and a dermatologist at the University of California, Los Angeles.
"Segmental vitiligo is really the only type of vitiligo I think I can cure, and that’s because we get our absolute best responses with autologous grafting in combination with phototherapy," she said at the annual meeting of the Society for Pediatric Dermatology.
She favors the use of 1-mm punch grafts over more elaborate techniques such as split-thickness grafts, blister roof grafts, and cultured cell transplantation. Autologous grafting using 1-mm punch grafts is simple, and long-term follow-up studies have documented the persistence of repigmentation for more than 5 years in patients with segmental vitiligo.
"It’s so, so easy," Dr. Grimes said. "And the great thing about punch grafts is this technique doesn’t require a lot of special equipment, unlike sheet grafts and the other methods. All you need are iris scissors and jeweler’s forceps."
"I’ve harvested anywhere from 10 to 250 grafts at one setting. We anesthetize a donor area on the hip, harvest the 1-mm punch grafts, place them in saline in sterile petri dishes, remove 1-mm punch grafts from the anesthetized recipient site, and then with jeweler’s forceps you place those harvested 1-mm grafts in those 1-mm recipient holes. You cover the areas with Steri-Strips, which stay in place for 7 days. Then you remove them and start phototherapy, either narrow-band UVB, PUVA [psoralen and UVA], or with an excimer laser," she explained.
In her experience, aggressive topical therapy using high-potency corticosteroids and tacrolimus (Protopic) often provides good results in treating segmental vitiligo on the face.
"If you have segmental vitiligo on the trunk and extremity areas, it’s much more difficult to get good results with topical therapies. Those are the patients who ultimately will need to be grafted," according to the dermatologist.
She stressed that when using autologous grafting to treat patients with generalized vitiligo, it’s imperative to stabilize them beforehand. This is accomplished using systemic steroids: for example, 5-10 mg/day of prednisone for 2 weeks, or mini-pulse dosing of betamethasone at 5 mg twice weekly.
"When I see kids with segmental vitiligo in my initial consultation, I always tell them and their parents that if I were going to get vitiligo, I’d choose segmental versus the generalized variety. That’s because of all the types of vitiligo that we see, the segmental variety is the most predictable. It burns itself out in about 95% of patients in that first year, so we know that in the overwhelming majority of patients it’s not going to spread beyond that dermatome," Dr. Grimes said.
She made a plea for physicians to be more aggressive in treating childhood vitiligo. Often they are reluctant to treat. Yet it has been shown across the board, for all treatment modalities, that efficacy is significantly greater in children than adults.
"Children are the ones who are more likely to give you that greater than 75% repigmentation. It’s a great reason to treat children early," Dr. Grimes said.
She reported having performed clinical research and/or serving as a consultant to a dozen pharmaceutical and cosmetics companies.
COEUR D’ALENE, IDAHO – Autologous grafting is a treatment of unmatched effectiveness for pediatric patients with segmental vitiligo unresponsive to medical modalities, according to Dr. Pearl E. Grimes, director of the Vitiligo and Pigmentation Institute of Southern California and a dermatologist at the University of California, Los Angeles.
"Segmental vitiligo is really the only type of vitiligo I think I can cure, and that’s because we get our absolute best responses with autologous grafting in combination with phototherapy," she said at the annual meeting of the Society for Pediatric Dermatology.
She favors the use of 1-mm punch grafts over more elaborate techniques such as split-thickness grafts, blister roof grafts, and cultured cell transplantation. Autologous grafting using 1-mm punch grafts is simple, and long-term follow-up studies have documented the persistence of repigmentation for more than 5 years in patients with segmental vitiligo.
"It’s so, so easy," Dr. Grimes said. "And the great thing about punch grafts is this technique doesn’t require a lot of special equipment, unlike sheet grafts and the other methods. All you need are iris scissors and jeweler’s forceps."
"I’ve harvested anywhere from 10 to 250 grafts at one setting. We anesthetize a donor area on the hip, harvest the 1-mm punch grafts, place them in saline in sterile petri dishes, remove 1-mm punch grafts from the anesthetized recipient site, and then with jeweler’s forceps you place those harvested 1-mm grafts in those 1-mm recipient holes. You cover the areas with Steri-Strips, which stay in place for 7 days. Then you remove them and start phototherapy, either narrow-band UVB, PUVA [psoralen and UVA], or with an excimer laser," she explained.
In her experience, aggressive topical therapy using high-potency corticosteroids and tacrolimus (Protopic) often provides good results in treating segmental vitiligo on the face.
"If you have segmental vitiligo on the trunk and extremity areas, it’s much more difficult to get good results with topical therapies. Those are the patients who ultimately will need to be grafted," according to the dermatologist.
She stressed that when using autologous grafting to treat patients with generalized vitiligo, it’s imperative to stabilize them beforehand. This is accomplished using systemic steroids: for example, 5-10 mg/day of prednisone for 2 weeks, or mini-pulse dosing of betamethasone at 5 mg twice weekly.
"When I see kids with segmental vitiligo in my initial consultation, I always tell them and their parents that if I were going to get vitiligo, I’d choose segmental versus the generalized variety. That’s because of all the types of vitiligo that we see, the segmental variety is the most predictable. It burns itself out in about 95% of patients in that first year, so we know that in the overwhelming majority of patients it’s not going to spread beyond that dermatome," Dr. Grimes said.
She made a plea for physicians to be more aggressive in treating childhood vitiligo. Often they are reluctant to treat. Yet it has been shown across the board, for all treatment modalities, that efficacy is significantly greater in children than adults.
"Children are the ones who are more likely to give you that greater than 75% repigmentation. It’s a great reason to treat children early," Dr. Grimes said.
She reported having performed clinical research and/or serving as a consultant to a dozen pharmaceutical and cosmetics companies.
EXPERT ANALYSIS FROM THE SPD ANNUAL MEETING
Effective treatments abound for children with heavy sweating in the hands and feet
COEUR D’ALENE, IDAHO – By the time patients seek a physician’s help for palmar/plantar focal hyperhidrosis, they may be well beyond the point at which even potent topical antiperspirants will help.
"Most people think there is nothing that can be done at all [for these patients]. But there are effective options. Even if you’re not going to offer them, get your patients to someone who will. We can make a profound difference for children and adolescents," Dr. Jane S. Bellet said at the annual meeting of the Society for Pediatric Dermatology.
In fact, more potent therapies are available, ranging from iontophoresis to oral medications, botulinum toxin A injections, and surgical thoracic sympathetectomy.
Oral medications: "I’ve really changed my practice in the last 5 years. I didn’t use systemic medications very much. I use them much more frequently now. I think you can get a very nice response," said Dr. Bellet, a pediatric dermatologist at Duke University in Durham, N.C.
She typically turns to the anticholinergic agents glycopyrrolate and oxybutynin. Glycopyrrolate, marketed as Cuvposa in a cherry-flavored solution at 1 mg/5 mL, does not have an indication from the Food and Drug Administration for treatment of pediatric hyperhidrosis, she noted. However, it is FDA-approved in 3- to 16-year-olds for severe chronic drooling caused by neurologic disorders.
"Although we don’t have an indication for hyperhidrosis, we do have a pediatric indication, and I think many times that puts our parents at ease," she observed.
Glycopyrrolate is cost effective and painless, although approximately 30% of children treated for hyperhidrosis will develop dry mouth and/or dry eyes.
"I would definitely add glycopyrrolate to your armamentarium. I think the biggest concern most of us have is the side effects. Speak about them with the family ahead of time, guide them as to what to expect, and stop if it becomes intolerable," Dr. Bellet said.
A recent randomized, prospective, controlled clinical trial in 45 children aged 7-14 years with palmar hyperhidrosis showed excellent outcomes with oxybutynin (Ditropan), with more than 85% experiencing at least moderate improvement in sweating and 80% gaining improved quality of life (Pediatr. Dermatol. 2014;31:48-53).
Iontophoresis: "This is a wonderful treatment for palms and soles," said Dr. Bellet.
The treatment entails placing the hands or feet in a water bath tray filled with tap water through which direct electric current is running. The proposed mechanism of benefit is that hydrolysis of the bath water results in accumulation of hydrogen ions, which then induce sweat gland destruction.
The chief disadvantage of iontophoresis is that it is labor intensive. Unless the family rents or buys a home unit, the patient typically must visit the physician’s office on a daily basis initially, placing the hands or feet in the bath for 10 minutes, followed by a second 10-minute round after the polarity is switched. Eventually, many patients can step down to two or three 20- to 30-minute sessions per week, then perhaps once-weekly maintenance therapy, she said.
Adding glycopyrrolate to the water bath has been shown to result in longer improvement in a study in both children and adults (Australas. J. Dermatol. 2004;45:208-12); however, this poses a greater risk of systemic absorption in children.
"Interestingly enough, some insurance companies will cover iontophoresis if you add glycopyrrolate to the water, but they won’t cover regular iontophoresis," Dr. Bellet said.
In response to audience inquiries, she indicated she uses the Fischer MD-1a galvanic unit. It’s simple to operate, costs about $700 including accessories, and can be rented with the payments applied to a later purchase.
Botulinum toxin A: First using this product for hyperhidrosis is off-label therapy, Dr. Bellet emphasized. Second, the doses required to treat palmar/plantar hyperhidrosis – 75-100 units per palm or sole – are much higher than in treating the axillae. Also, injecting botulinum toxin A into the palms and soles is extraordinarily painful. Children typically require general anesthesia, because the alternative methods employed with mixed results in adults, including EMLA cream, ice packs, and ethyl chloride spray, don’t cut it in younger patients, Dr. Bellet noted.
That being said, botulinum toxin A therapy works quite well in children and adolescents. However, it’s important to explain to patients and parents up front that the injections can cause transient weakness of the hand muscles because of the diffusion of the toxin from dermis to muscles; for a budding pianist or baseball pitcher, that can be a deal-breaker, she said. Also, Dr. Bellet added, repeated injections can result in atrophy of the thenar and hypothenar eminences, with resultant irreversible weakness.
Thoracic sympathectomy: Not many American thoracic surgeons do sympathectomy to treat hyperhidrosis on a frequent basis, but those with extensive experience obtain outstanding results, Dr. Bellet said. To treat palmar hyperhidrosis, the nerve is clipped at the top of the third or fourth rib; for the soles, it’s at the fourth and fifth rib. The youngest reported treated patients have been 8 years old.
Dr. Bellet recommended the International Hyperhidrosis Society (www.sweathelp.org) as an excellent source of further information.
Dr. Bellet reported having no financial conflicts of interest regarding her presentation.
How do you treat palmar/plantar hyperhidrosis in children? Take our Quick Poll on the Skin & Allergy News homepage.
COEUR D’ALENE, IDAHO – By the time patients seek a physician’s help for palmar/plantar focal hyperhidrosis, they may be well beyond the point at which even potent topical antiperspirants will help.
"Most people think there is nothing that can be done at all [for these patients]. But there are effective options. Even if you’re not going to offer them, get your patients to someone who will. We can make a profound difference for children and adolescents," Dr. Jane S. Bellet said at the annual meeting of the Society for Pediatric Dermatology.
In fact, more potent therapies are available, ranging from iontophoresis to oral medications, botulinum toxin A injections, and surgical thoracic sympathetectomy.
Oral medications: "I’ve really changed my practice in the last 5 years. I didn’t use systemic medications very much. I use them much more frequently now. I think you can get a very nice response," said Dr. Bellet, a pediatric dermatologist at Duke University in Durham, N.C.
She typically turns to the anticholinergic agents glycopyrrolate and oxybutynin. Glycopyrrolate, marketed as Cuvposa in a cherry-flavored solution at 1 mg/5 mL, does not have an indication from the Food and Drug Administration for treatment of pediatric hyperhidrosis, she noted. However, it is FDA-approved in 3- to 16-year-olds for severe chronic drooling caused by neurologic disorders.
"Although we don’t have an indication for hyperhidrosis, we do have a pediatric indication, and I think many times that puts our parents at ease," she observed.
Glycopyrrolate is cost effective and painless, although approximately 30% of children treated for hyperhidrosis will develop dry mouth and/or dry eyes.
"I would definitely add glycopyrrolate to your armamentarium. I think the biggest concern most of us have is the side effects. Speak about them with the family ahead of time, guide them as to what to expect, and stop if it becomes intolerable," Dr. Bellet said.
A recent randomized, prospective, controlled clinical trial in 45 children aged 7-14 years with palmar hyperhidrosis showed excellent outcomes with oxybutynin (Ditropan), with more than 85% experiencing at least moderate improvement in sweating and 80% gaining improved quality of life (Pediatr. Dermatol. 2014;31:48-53).
Iontophoresis: "This is a wonderful treatment for palms and soles," said Dr. Bellet.
The treatment entails placing the hands or feet in a water bath tray filled with tap water through which direct electric current is running. The proposed mechanism of benefit is that hydrolysis of the bath water results in accumulation of hydrogen ions, which then induce sweat gland destruction.
The chief disadvantage of iontophoresis is that it is labor intensive. Unless the family rents or buys a home unit, the patient typically must visit the physician’s office on a daily basis initially, placing the hands or feet in the bath for 10 minutes, followed by a second 10-minute round after the polarity is switched. Eventually, many patients can step down to two or three 20- to 30-minute sessions per week, then perhaps once-weekly maintenance therapy, she said.
Adding glycopyrrolate to the water bath has been shown to result in longer improvement in a study in both children and adults (Australas. J. Dermatol. 2004;45:208-12); however, this poses a greater risk of systemic absorption in children.
"Interestingly enough, some insurance companies will cover iontophoresis if you add glycopyrrolate to the water, but they won’t cover regular iontophoresis," Dr. Bellet said.
In response to audience inquiries, she indicated she uses the Fischer MD-1a galvanic unit. It’s simple to operate, costs about $700 including accessories, and can be rented with the payments applied to a later purchase.
Botulinum toxin A: First using this product for hyperhidrosis is off-label therapy, Dr. Bellet emphasized. Second, the doses required to treat palmar/plantar hyperhidrosis – 75-100 units per palm or sole – are much higher than in treating the axillae. Also, injecting botulinum toxin A into the palms and soles is extraordinarily painful. Children typically require general anesthesia, because the alternative methods employed with mixed results in adults, including EMLA cream, ice packs, and ethyl chloride spray, don’t cut it in younger patients, Dr. Bellet noted.
That being said, botulinum toxin A therapy works quite well in children and adolescents. However, it’s important to explain to patients and parents up front that the injections can cause transient weakness of the hand muscles because of the diffusion of the toxin from dermis to muscles; for a budding pianist or baseball pitcher, that can be a deal-breaker, she said. Also, Dr. Bellet added, repeated injections can result in atrophy of the thenar and hypothenar eminences, with resultant irreversible weakness.
Thoracic sympathectomy: Not many American thoracic surgeons do sympathectomy to treat hyperhidrosis on a frequent basis, but those with extensive experience obtain outstanding results, Dr. Bellet said. To treat palmar hyperhidrosis, the nerve is clipped at the top of the third or fourth rib; for the soles, it’s at the fourth and fifth rib. The youngest reported treated patients have been 8 years old.
Dr. Bellet recommended the International Hyperhidrosis Society (www.sweathelp.org) as an excellent source of further information.
Dr. Bellet reported having no financial conflicts of interest regarding her presentation.
How do you treat palmar/plantar hyperhidrosis in children? Take our Quick Poll on the Skin & Allergy News homepage.
COEUR D’ALENE, IDAHO – By the time patients seek a physician’s help for palmar/plantar focal hyperhidrosis, they may be well beyond the point at which even potent topical antiperspirants will help.
"Most people think there is nothing that can be done at all [for these patients]. But there are effective options. Even if you’re not going to offer them, get your patients to someone who will. We can make a profound difference for children and adolescents," Dr. Jane S. Bellet said at the annual meeting of the Society for Pediatric Dermatology.
In fact, more potent therapies are available, ranging from iontophoresis to oral medications, botulinum toxin A injections, and surgical thoracic sympathetectomy.
Oral medications: "I’ve really changed my practice in the last 5 years. I didn’t use systemic medications very much. I use them much more frequently now. I think you can get a very nice response," said Dr. Bellet, a pediatric dermatologist at Duke University in Durham, N.C.
She typically turns to the anticholinergic agents glycopyrrolate and oxybutynin. Glycopyrrolate, marketed as Cuvposa in a cherry-flavored solution at 1 mg/5 mL, does not have an indication from the Food and Drug Administration for treatment of pediatric hyperhidrosis, she noted. However, it is FDA-approved in 3- to 16-year-olds for severe chronic drooling caused by neurologic disorders.
"Although we don’t have an indication for hyperhidrosis, we do have a pediatric indication, and I think many times that puts our parents at ease," she observed.
Glycopyrrolate is cost effective and painless, although approximately 30% of children treated for hyperhidrosis will develop dry mouth and/or dry eyes.
"I would definitely add glycopyrrolate to your armamentarium. I think the biggest concern most of us have is the side effects. Speak about them with the family ahead of time, guide them as to what to expect, and stop if it becomes intolerable," Dr. Bellet said.
A recent randomized, prospective, controlled clinical trial in 45 children aged 7-14 years with palmar hyperhidrosis showed excellent outcomes with oxybutynin (Ditropan), with more than 85% experiencing at least moderate improvement in sweating and 80% gaining improved quality of life (Pediatr. Dermatol. 2014;31:48-53).
Iontophoresis: "This is a wonderful treatment for palms and soles," said Dr. Bellet.
The treatment entails placing the hands or feet in a water bath tray filled with tap water through which direct electric current is running. The proposed mechanism of benefit is that hydrolysis of the bath water results in accumulation of hydrogen ions, which then induce sweat gland destruction.
The chief disadvantage of iontophoresis is that it is labor intensive. Unless the family rents or buys a home unit, the patient typically must visit the physician’s office on a daily basis initially, placing the hands or feet in the bath for 10 minutes, followed by a second 10-minute round after the polarity is switched. Eventually, many patients can step down to two or three 20- to 30-minute sessions per week, then perhaps once-weekly maintenance therapy, she said.
Adding glycopyrrolate to the water bath has been shown to result in longer improvement in a study in both children and adults (Australas. J. Dermatol. 2004;45:208-12); however, this poses a greater risk of systemic absorption in children.
"Interestingly enough, some insurance companies will cover iontophoresis if you add glycopyrrolate to the water, but they won’t cover regular iontophoresis," Dr. Bellet said.
In response to audience inquiries, she indicated she uses the Fischer MD-1a galvanic unit. It’s simple to operate, costs about $700 including accessories, and can be rented with the payments applied to a later purchase.
Botulinum toxin A: First using this product for hyperhidrosis is off-label therapy, Dr. Bellet emphasized. Second, the doses required to treat palmar/plantar hyperhidrosis – 75-100 units per palm or sole – are much higher than in treating the axillae. Also, injecting botulinum toxin A into the palms and soles is extraordinarily painful. Children typically require general anesthesia, because the alternative methods employed with mixed results in adults, including EMLA cream, ice packs, and ethyl chloride spray, don’t cut it in younger patients, Dr. Bellet noted.
That being said, botulinum toxin A therapy works quite well in children and adolescents. However, it’s important to explain to patients and parents up front that the injections can cause transient weakness of the hand muscles because of the diffusion of the toxin from dermis to muscles; for a budding pianist or baseball pitcher, that can be a deal-breaker, she said. Also, Dr. Bellet added, repeated injections can result in atrophy of the thenar and hypothenar eminences, with resultant irreversible weakness.
Thoracic sympathectomy: Not many American thoracic surgeons do sympathectomy to treat hyperhidrosis on a frequent basis, but those with extensive experience obtain outstanding results, Dr. Bellet said. To treat palmar hyperhidrosis, the nerve is clipped at the top of the third or fourth rib; for the soles, it’s at the fourth and fifth rib. The youngest reported treated patients have been 8 years old.
Dr. Bellet recommended the International Hyperhidrosis Society (www.sweathelp.org) as an excellent source of further information.
Dr. Bellet reported having no financial conflicts of interest regarding her presentation.
How do you treat palmar/plantar hyperhidrosis in children? Take our Quick Poll on the Skin & Allergy News homepage.
EXPERT ANALYSIS FROM THE SPD ANNUAL MEETING
Onychomycosis: Not just for adults
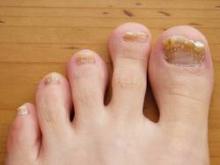
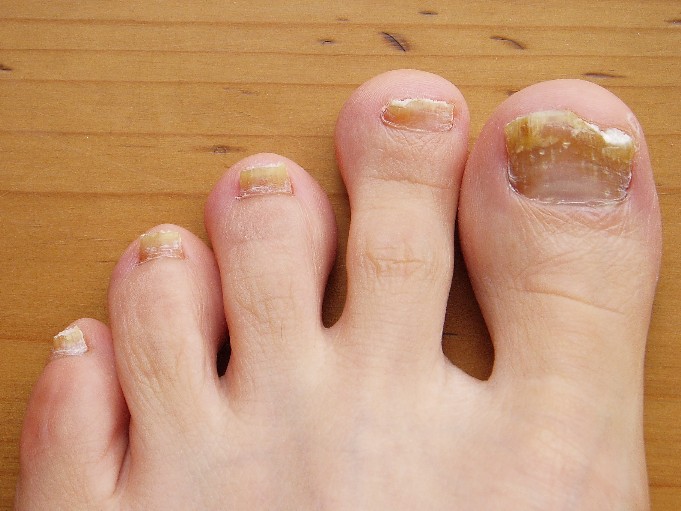
COEUR D’ALENE, IDAHO – Conventional wisdom holds that onychomycosis is rare in children. Not so.
Fully one-third of children and adolescents who presented with a nail complaint to a prominent dermatologic nail disorders center were diagnosed with mycologically confirmed onychomycosis, Dr. Julie Jefferson reported at the annual meeting of the Society for Pediatric Dermatology.
That’s a substantially higher prevalence than the 15.5% figure reported by Spanish investigators in a 20-year retrospective study (Mycoses 2011;54:450-3). It’s also well below the 47% prevalence recently reported in Denver in a 5-year retrospective study, where Trychophyton rubrum was the most common pathogen, and the highest prevalence of onychomycosis in the pediatric population was seen in 6- to 10-year-olds (Pediatr. Dermatol. 2014;31:106-8), noted Dr. Jefferson of Johns Hopkins University, Baltimore.
The investigators in both Span and Denver observed that the prevalence of pediatric onychomycosis appears to be increasing in recent years.
Dr. Jefferson presented a retrospective study that included 917 patients up to age 18 years who presented to the Oregon Dermatology and Research Center, Portland, in a recent 6-year period. One or more nail disorders were diagnosed in 11%. The mean age at presentation was 9.4 years, with a mean 2.4-year duration of the condition prior to presentation. Toenails were affected in 47 patients, fingernails in 37, and both in 18. Fourteen patients had two nail disorders, 9 had three, and 2 had four distinct nail disorders.
The etiologies ranged widely, from infections to congenital and hereditary malformations, tumors, inflammatory processes, and systemic diseases.
The most common nail condition was onychomycosis, diagnosed in 34 patients. Thus, 3.7% of all pediatric patients presenting to the dermatology center for any reason were diagnosed with onychomycosis, a higher rate than previously reported by others.
Other conditions included 13 cases of longitudinal melanonychia, 11 of disappearing nail bed, 9 of retronychia, 8 cases of congenital malalignment, 8 cases of trachyonychia, 7 of paronychia, 5 cases of psoriasis, and 2 of lichen planus.
Three of four patients with an ingrown toenail also had congenital malalignment of the affected great toenail, supporting the notion put forth by other investigators that congenital malalignment of the great toenail predisposes to ingrown toenails, according to Dr. Jefferson.
She reported having no financial conflicts related to this study.
COEUR D’ALENE, IDAHO – Conventional wisdom holds that onychomycosis is rare in children. Not so.
Fully one-third of children and adolescents who presented with a nail complaint to a prominent dermatologic nail disorders center were diagnosed with mycologically confirmed onychomycosis, Dr. Julie Jefferson reported at the annual meeting of the Society for Pediatric Dermatology.
That’s a substantially higher prevalence than the 15.5% figure reported by Spanish investigators in a 20-year retrospective study (Mycoses 2011;54:450-3). It’s also well below the 47% prevalence recently reported in Denver in a 5-year retrospective study, where Trychophyton rubrum was the most common pathogen, and the highest prevalence of onychomycosis in the pediatric population was seen in 6- to 10-year-olds (Pediatr. Dermatol. 2014;31:106-8), noted Dr. Jefferson of Johns Hopkins University, Baltimore.
The investigators in both Span and Denver observed that the prevalence of pediatric onychomycosis appears to be increasing in recent years.
Dr. Jefferson presented a retrospective study that included 917 patients up to age 18 years who presented to the Oregon Dermatology and Research Center, Portland, in a recent 6-year period. One or more nail disorders were diagnosed in 11%. The mean age at presentation was 9.4 years, with a mean 2.4-year duration of the condition prior to presentation. Toenails were affected in 47 patients, fingernails in 37, and both in 18. Fourteen patients had two nail disorders, 9 had three, and 2 had four distinct nail disorders.
The etiologies ranged widely, from infections to congenital and hereditary malformations, tumors, inflammatory processes, and systemic diseases.
The most common nail condition was onychomycosis, diagnosed in 34 patients. Thus, 3.7% of all pediatric patients presenting to the dermatology center for any reason were diagnosed with onychomycosis, a higher rate than previously reported by others.
Other conditions included 13 cases of longitudinal melanonychia, 11 of disappearing nail bed, 9 of retronychia, 8 cases of congenital malalignment, 8 cases of trachyonychia, 7 of paronychia, 5 cases of psoriasis, and 2 of lichen planus.
Three of four patients with an ingrown toenail also had congenital malalignment of the affected great toenail, supporting the notion put forth by other investigators that congenital malalignment of the great toenail predisposes to ingrown toenails, according to Dr. Jefferson.
She reported having no financial conflicts related to this study.
COEUR D’ALENE, IDAHO – Conventional wisdom holds that onychomycosis is rare in children. Not so.
Fully one-third of children and adolescents who presented with a nail complaint to a prominent dermatologic nail disorders center were diagnosed with mycologically confirmed onychomycosis, Dr. Julie Jefferson reported at the annual meeting of the Society for Pediatric Dermatology.
That’s a substantially higher prevalence than the 15.5% figure reported by Spanish investigators in a 20-year retrospective study (Mycoses 2011;54:450-3). It’s also well below the 47% prevalence recently reported in Denver in a 5-year retrospective study, where Trychophyton rubrum was the most common pathogen, and the highest prevalence of onychomycosis in the pediatric population was seen in 6- to 10-year-olds (Pediatr. Dermatol. 2014;31:106-8), noted Dr. Jefferson of Johns Hopkins University, Baltimore.
The investigators in both Span and Denver observed that the prevalence of pediatric onychomycosis appears to be increasing in recent years.
Dr. Jefferson presented a retrospective study that included 917 patients up to age 18 years who presented to the Oregon Dermatology and Research Center, Portland, in a recent 6-year period. One or more nail disorders were diagnosed in 11%. The mean age at presentation was 9.4 years, with a mean 2.4-year duration of the condition prior to presentation. Toenails were affected in 47 patients, fingernails in 37, and both in 18. Fourteen patients had two nail disorders, 9 had three, and 2 had four distinct nail disorders.
The etiologies ranged widely, from infections to congenital and hereditary malformations, tumors, inflammatory processes, and systemic diseases.
The most common nail condition was onychomycosis, diagnosed in 34 patients. Thus, 3.7% of all pediatric patients presenting to the dermatology center for any reason were diagnosed with onychomycosis, a higher rate than previously reported by others.
Other conditions included 13 cases of longitudinal melanonychia, 11 of disappearing nail bed, 9 of retronychia, 8 cases of congenital malalignment, 8 cases of trachyonychia, 7 of paronychia, 5 cases of psoriasis, and 2 of lichen planus.
Three of four patients with an ingrown toenail also had congenital malalignment of the affected great toenail, supporting the notion put forth by other investigators that congenital malalignment of the great toenail predisposes to ingrown toenails, according to Dr. Jefferson.
She reported having no financial conflicts related to this study.
AT THE SPD ANNUAL MEETING
Key clinical point: Onychomycosis appears to be rising in children and adolescents, and needs to be considered in the differential diagnosis of pediatric nail abnormalities.
Major finding: Onychomycosis was diagnosed in 3.7% of all children and adolescents who presented for any reason to a tertiary dermatology center, and in 33% of the 102 who presented for evaluation of a nail disorder.
Data source: This was a 6-year, single-center, retrospective study of 917 patients up to 18 years of age.
Disclosures: The presenter reported having no financial conflicts with regard to this study.
Check thyroid function in children with vitiligo
COEUR D’ALENE, IDAHO – Every child and adolescent with vitiligo should undergo a laboratory evaluation that includes thyroid function tests and antibody levels, Dr. Pearl E. Grimes advised at the annual meeting of the Society for Pediatric Dermatology.
"At some point, we used to debate whether thyroid disease really is more common in the pediatric population with vitiligo. But now we have multiple studies in the literature to suggest that it is. And that certainly has also been my own clinical experience in looking at thyroid function tests as well as thyroid antibodies for the past 25 years in the pediatric population," said Dr. Grimes, director of the Vitiligo and Pigmentation Institute of Southern California and a dermatologist at the University of California, Los Angeles.
Vitiligo is an autoimmune disease. As such, it is well known to be associated with other autoimmune diseases in adults, with Hashimoto’s thyroiditis and other forms of thyroid disease being the most common comorbid autoimmune conditions.
Dr. Grimes cited two recent studies showing a sharply increased prevalence of thyroid abnormalities as well in the pediatric population with vitiligo.
Investigators at the Netherlands Institute for Pigment Disorders in Amsterdam reported on 260 children and adolescents with vitiligo who underwent measurement of thyroid-stimulating hormone, free thyroxine, and antithyroid peroxidase antibody levels. The results indicated 6.2% had autoimmune thyroiditis with thyroid hormone disturbances, a prevalence far greater than that seen in the general pediatric population. Moreover, 10.5% of patients had elevated levels of antithyroid peroxidase antibodies without disturbance of thyroid hormone, a condition known to be associated with an increased risk of developing overt thyroid disease down the road (Horm. Res. Paediatr. 2013;79:137-44).
In another study, investigators at Children’s Hospital in Izmir, Turkey, retrospectively reviewed laboratory findings in 79 vitiligo patients, aged 2-15 years. Fully one-quarter had abnormal results on thyroid function tests and/or elevated thyroid autoantibodies (Indian J. Endocrinol. Metab. 2013;1096-9).
Dr. Grimes recommended that a routine laboratory screening panel for children and adolescents with vitiligo should "at a bare minimum" consist of a CBC with differential, a thyroid panel, thyroid antibodies, and an anti-nuclear antibody level.
"I’ve been looking at ANAs [antinuclear antibody test results] in children and adults with vitiligo for a very long time. I find that about 30% of patients, including kids, will have a positive ANA. What’s the significance? A small cohort of vitiligo patients will go on to develop lupus. Although lupus is not one of our more common autoimmune diseases, it happens. If the ANA titer is less than 1:160, I typically don’t get a lupus panel; but if it is greater than 1:160, I will follow up with a lupus panel – and that determines whether I will send that patient for a rheumatologic consultation. The ANA is also very important in determining whether a vitiligo patient is a candidate for phototherapy. If there’s a high-titer ANA, I will not place a patient on narrow band UVB (therapy)," according to Dr. Grimes.
She reported having performed clinical research for and serving as a consultant to several pharmaceutical and cosmetics companies.
COEUR D’ALENE, IDAHO – Every child and adolescent with vitiligo should undergo a laboratory evaluation that includes thyroid function tests and antibody levels, Dr. Pearl E. Grimes advised at the annual meeting of the Society for Pediatric Dermatology.
"At some point, we used to debate whether thyroid disease really is more common in the pediatric population with vitiligo. But now we have multiple studies in the literature to suggest that it is. And that certainly has also been my own clinical experience in looking at thyroid function tests as well as thyroid antibodies for the past 25 years in the pediatric population," said Dr. Grimes, director of the Vitiligo and Pigmentation Institute of Southern California and a dermatologist at the University of California, Los Angeles.
Vitiligo is an autoimmune disease. As such, it is well known to be associated with other autoimmune diseases in adults, with Hashimoto’s thyroiditis and other forms of thyroid disease being the most common comorbid autoimmune conditions.
Dr. Grimes cited two recent studies showing a sharply increased prevalence of thyroid abnormalities as well in the pediatric population with vitiligo.
Investigators at the Netherlands Institute for Pigment Disorders in Amsterdam reported on 260 children and adolescents with vitiligo who underwent measurement of thyroid-stimulating hormone, free thyroxine, and antithyroid peroxidase antibody levels. The results indicated 6.2% had autoimmune thyroiditis with thyroid hormone disturbances, a prevalence far greater than that seen in the general pediatric population. Moreover, 10.5% of patients had elevated levels of antithyroid peroxidase antibodies without disturbance of thyroid hormone, a condition known to be associated with an increased risk of developing overt thyroid disease down the road (Horm. Res. Paediatr. 2013;79:137-44).
In another study, investigators at Children’s Hospital in Izmir, Turkey, retrospectively reviewed laboratory findings in 79 vitiligo patients, aged 2-15 years. Fully one-quarter had abnormal results on thyroid function tests and/or elevated thyroid autoantibodies (Indian J. Endocrinol. Metab. 2013;1096-9).
Dr. Grimes recommended that a routine laboratory screening panel for children and adolescents with vitiligo should "at a bare minimum" consist of a CBC with differential, a thyroid panel, thyroid antibodies, and an anti-nuclear antibody level.
"I’ve been looking at ANAs [antinuclear antibody test results] in children and adults with vitiligo for a very long time. I find that about 30% of patients, including kids, will have a positive ANA. What’s the significance? A small cohort of vitiligo patients will go on to develop lupus. Although lupus is not one of our more common autoimmune diseases, it happens. If the ANA titer is less than 1:160, I typically don’t get a lupus panel; but if it is greater than 1:160, I will follow up with a lupus panel – and that determines whether I will send that patient for a rheumatologic consultation. The ANA is also very important in determining whether a vitiligo patient is a candidate for phototherapy. If there’s a high-titer ANA, I will not place a patient on narrow band UVB (therapy)," according to Dr. Grimes.
She reported having performed clinical research for and serving as a consultant to several pharmaceutical and cosmetics companies.
COEUR D’ALENE, IDAHO – Every child and adolescent with vitiligo should undergo a laboratory evaluation that includes thyroid function tests and antibody levels, Dr. Pearl E. Grimes advised at the annual meeting of the Society for Pediatric Dermatology.
"At some point, we used to debate whether thyroid disease really is more common in the pediatric population with vitiligo. But now we have multiple studies in the literature to suggest that it is. And that certainly has also been my own clinical experience in looking at thyroid function tests as well as thyroid antibodies for the past 25 years in the pediatric population," said Dr. Grimes, director of the Vitiligo and Pigmentation Institute of Southern California and a dermatologist at the University of California, Los Angeles.
Vitiligo is an autoimmune disease. As such, it is well known to be associated with other autoimmune diseases in adults, with Hashimoto’s thyroiditis and other forms of thyroid disease being the most common comorbid autoimmune conditions.
Dr. Grimes cited two recent studies showing a sharply increased prevalence of thyroid abnormalities as well in the pediatric population with vitiligo.
Investigators at the Netherlands Institute for Pigment Disorders in Amsterdam reported on 260 children and adolescents with vitiligo who underwent measurement of thyroid-stimulating hormone, free thyroxine, and antithyroid peroxidase antibody levels. The results indicated 6.2% had autoimmune thyroiditis with thyroid hormone disturbances, a prevalence far greater than that seen in the general pediatric population. Moreover, 10.5% of patients had elevated levels of antithyroid peroxidase antibodies without disturbance of thyroid hormone, a condition known to be associated with an increased risk of developing overt thyroid disease down the road (Horm. Res. Paediatr. 2013;79:137-44).
In another study, investigators at Children’s Hospital in Izmir, Turkey, retrospectively reviewed laboratory findings in 79 vitiligo patients, aged 2-15 years. Fully one-quarter had abnormal results on thyroid function tests and/or elevated thyroid autoantibodies (Indian J. Endocrinol. Metab. 2013;1096-9).
Dr. Grimes recommended that a routine laboratory screening panel for children and adolescents with vitiligo should "at a bare minimum" consist of a CBC with differential, a thyroid panel, thyroid antibodies, and an anti-nuclear antibody level.
"I’ve been looking at ANAs [antinuclear antibody test results] in children and adults with vitiligo for a very long time. I find that about 30% of patients, including kids, will have a positive ANA. What’s the significance? A small cohort of vitiligo patients will go on to develop lupus. Although lupus is not one of our more common autoimmune diseases, it happens. If the ANA titer is less than 1:160, I typically don’t get a lupus panel; but if it is greater than 1:160, I will follow up with a lupus panel – and that determines whether I will send that patient for a rheumatologic consultation. The ANA is also very important in determining whether a vitiligo patient is a candidate for phototherapy. If there’s a high-titer ANA, I will not place a patient on narrow band UVB (therapy)," according to Dr. Grimes.
She reported having performed clinical research for and serving as a consultant to several pharmaceutical and cosmetics companies.
EXPERT ANALYSIS FROM THE SPD ANNUAL MEETING
Maintenance therapy options for childhood alopecia areata
COEUR D’ALENE, IDAHO – Systemic corticosteroids usually bring an impressive initial response for children with alopecia areata. The big question is, ‘What do you try next?’
Children can’t safely remain on long-term systemic steroids. And the great majority of responders will relapse once steroids are discontinued. The challenge is to find a safe, well-tolerated drug with long-term effectiveness as a maintenance therapy.
"For me, when I look at the list of drugs for maintenance therapy in alopecia areata, which includes cyclosporine, azathioprine, and sulfasalazine, the drug I’m most comfortable giving to children for a prolonged time is methotrexate. That’s why this drug has really become my go-to maintenance therapy for patients with alopecia areata," Dr. Ruth Ann Vleugels said at the annual meeting of the Society for Pediatric Dermatology.
"I will give them 2-3 months of systemic steroids as a boost while their methotrexate kicks in; but after that, it’s maintenance therapy with methotrexate at 1 mg/kg/week," added Dr. Vleugels, director of the Autoimmune Skin Disease Program at Brigham and Women’s Hospital, Boston.
Of course, methotrexate doesn’t work as maintenance therapy for all children with alopecia areata. Patients who don’t respond to the initial several months of combination therapy with systemic steroids won’t respond to maintenance therapy with methotrexate alone.
For those few patients who need second-line maintenance therapy, Dr. Vleugels’ drug of choice is the transplant anti-rejection medication mycophenolate mofetil (CellCept), which is used off-label for alopecia areata. Mycophenolate mofetil also is her rescue agent as maintenance therapy for children with systemic lupus erythematosus, cutaneous lupus erythematosus, juvenile dermatomyositis, and linear morphea with progressive hemifacial atrophy.
In patients with progressive juvenile systemic sclerosis, Dr. Vleugels and her Boston colleagues now use mycophenolate mofetil, rather than methotrexate, as their first-line therapy based upon favorable results in their practice and prospective studies in adults (J. Rheumatol. 2012;39:1241-7).
The mycophenolate mofetil dosing in children with alopecia areata is 600 mg/m2 given twice daily, or 1 g twice daily in patients bigger than 1.5 m2. She uses half the dose for the first 2-3 weeks, then titrates up to full-dose therapy in order to minimize GI side effects, which she said are far and away the most common adverse effects. Smaller children do best with the liquid formulation.
A common clinical dilemma involves the child who experiences intolerable GI upset on mycophenolate mofetil. Dr. Vleugels offered two suggestions: One is to tell the parents to ignore the package insert and have their child take the drug with meals.
"No one can handle this drug on an empty stomach," she said.
A caveat: mycophenolate mofetil cannot be taken with antacids.
If mealtime dosing doesn’t solve the GI upset problem, the alternative is to switch to mycophenolic acid (Myfortic), which is enteric-coated, has better bioavailability, and is better tolerated. It’s also more expensive than mycophenolate mofetil and will require prior authorization from payers. The dose conversion is as follows: 500 mg mycophenolate mofetil equals 360 mg of mycophenolic acid.
Dr. Vleugels reported having no financial conflicts with regard to her presentation.
COEUR D’ALENE, IDAHO – Systemic corticosteroids usually bring an impressive initial response for children with alopecia areata. The big question is, ‘What do you try next?’
Children can’t safely remain on long-term systemic steroids. And the great majority of responders will relapse once steroids are discontinued. The challenge is to find a safe, well-tolerated drug with long-term effectiveness as a maintenance therapy.
"For me, when I look at the list of drugs for maintenance therapy in alopecia areata, which includes cyclosporine, azathioprine, and sulfasalazine, the drug I’m most comfortable giving to children for a prolonged time is methotrexate. That’s why this drug has really become my go-to maintenance therapy for patients with alopecia areata," Dr. Ruth Ann Vleugels said at the annual meeting of the Society for Pediatric Dermatology.
"I will give them 2-3 months of systemic steroids as a boost while their methotrexate kicks in; but after that, it’s maintenance therapy with methotrexate at 1 mg/kg/week," added Dr. Vleugels, director of the Autoimmune Skin Disease Program at Brigham and Women’s Hospital, Boston.
Of course, methotrexate doesn’t work as maintenance therapy for all children with alopecia areata. Patients who don’t respond to the initial several months of combination therapy with systemic steroids won’t respond to maintenance therapy with methotrexate alone.
For those few patients who need second-line maintenance therapy, Dr. Vleugels’ drug of choice is the transplant anti-rejection medication mycophenolate mofetil (CellCept), which is used off-label for alopecia areata. Mycophenolate mofetil also is her rescue agent as maintenance therapy for children with systemic lupus erythematosus, cutaneous lupus erythematosus, juvenile dermatomyositis, and linear morphea with progressive hemifacial atrophy.
In patients with progressive juvenile systemic sclerosis, Dr. Vleugels and her Boston colleagues now use mycophenolate mofetil, rather than methotrexate, as their first-line therapy based upon favorable results in their practice and prospective studies in adults (J. Rheumatol. 2012;39:1241-7).
The mycophenolate mofetil dosing in children with alopecia areata is 600 mg/m2 given twice daily, or 1 g twice daily in patients bigger than 1.5 m2. She uses half the dose for the first 2-3 weeks, then titrates up to full-dose therapy in order to minimize GI side effects, which she said are far and away the most common adverse effects. Smaller children do best with the liquid formulation.
A common clinical dilemma involves the child who experiences intolerable GI upset on mycophenolate mofetil. Dr. Vleugels offered two suggestions: One is to tell the parents to ignore the package insert and have their child take the drug with meals.
"No one can handle this drug on an empty stomach," she said.
A caveat: mycophenolate mofetil cannot be taken with antacids.
If mealtime dosing doesn’t solve the GI upset problem, the alternative is to switch to mycophenolic acid (Myfortic), which is enteric-coated, has better bioavailability, and is better tolerated. It’s also more expensive than mycophenolate mofetil and will require prior authorization from payers. The dose conversion is as follows: 500 mg mycophenolate mofetil equals 360 mg of mycophenolic acid.
Dr. Vleugels reported having no financial conflicts with regard to her presentation.
COEUR D’ALENE, IDAHO – Systemic corticosteroids usually bring an impressive initial response for children with alopecia areata. The big question is, ‘What do you try next?’
Children can’t safely remain on long-term systemic steroids. And the great majority of responders will relapse once steroids are discontinued. The challenge is to find a safe, well-tolerated drug with long-term effectiveness as a maintenance therapy.
"For me, when I look at the list of drugs for maintenance therapy in alopecia areata, which includes cyclosporine, azathioprine, and sulfasalazine, the drug I’m most comfortable giving to children for a prolonged time is methotrexate. That’s why this drug has really become my go-to maintenance therapy for patients with alopecia areata," Dr. Ruth Ann Vleugels said at the annual meeting of the Society for Pediatric Dermatology.
"I will give them 2-3 months of systemic steroids as a boost while their methotrexate kicks in; but after that, it’s maintenance therapy with methotrexate at 1 mg/kg/week," added Dr. Vleugels, director of the Autoimmune Skin Disease Program at Brigham and Women’s Hospital, Boston.
Of course, methotrexate doesn’t work as maintenance therapy for all children with alopecia areata. Patients who don’t respond to the initial several months of combination therapy with systemic steroids won’t respond to maintenance therapy with methotrexate alone.
For those few patients who need second-line maintenance therapy, Dr. Vleugels’ drug of choice is the transplant anti-rejection medication mycophenolate mofetil (CellCept), which is used off-label for alopecia areata. Mycophenolate mofetil also is her rescue agent as maintenance therapy for children with systemic lupus erythematosus, cutaneous lupus erythematosus, juvenile dermatomyositis, and linear morphea with progressive hemifacial atrophy.
In patients with progressive juvenile systemic sclerosis, Dr. Vleugels and her Boston colleagues now use mycophenolate mofetil, rather than methotrexate, as their first-line therapy based upon favorable results in their practice and prospective studies in adults (J. Rheumatol. 2012;39:1241-7).
The mycophenolate mofetil dosing in children with alopecia areata is 600 mg/m2 given twice daily, or 1 g twice daily in patients bigger than 1.5 m2. She uses half the dose for the first 2-3 weeks, then titrates up to full-dose therapy in order to minimize GI side effects, which she said are far and away the most common adverse effects. Smaller children do best with the liquid formulation.
A common clinical dilemma involves the child who experiences intolerable GI upset on mycophenolate mofetil. Dr. Vleugels offered two suggestions: One is to tell the parents to ignore the package insert and have their child take the drug with meals.
"No one can handle this drug on an empty stomach," she said.
A caveat: mycophenolate mofetil cannot be taken with antacids.
If mealtime dosing doesn’t solve the GI upset problem, the alternative is to switch to mycophenolic acid (Myfortic), which is enteric-coated, has better bioavailability, and is better tolerated. It’s also more expensive than mycophenolate mofetil and will require prior authorization from payers. The dose conversion is as follows: 500 mg mycophenolate mofetil equals 360 mg of mycophenolic acid.
Dr. Vleugels reported having no financial conflicts with regard to her presentation.
EXPERT ANALYSIS FROM THE SPD ANNUAL MEETING
Big gains shown in ED pediatric readiness
DALLAS – Emergency departments have substantially improved their readiness to receive pediatric patients since a 2009 multispecialty call to action was triggered by documented major deficiencies in preparedness, the results of a comprehensive new survey indicate.
"We’ve seen significant improvement in all ED patient volume classes in terms of median pediatric readiness scores. The data are pretty exciting. It says we’re moving in the right direction," Dr. Marianne Gausche-Hill said in presenting the survey findings at the annual meeting of the Society for Academic Emergency Medicine.

The key outcome measure in the national survey was a weighted ED pediatric readiness score based upon the extent to which an ED was compliant with the major recommendations of the 2009 joint American Academy of Pediatrics/American College of Emergency Physicians/Emergency Nurses Association guidelines for the care of children in EDs (Pediatrics 2009;124:1233-44.
The guidelines lay out in concrete terms what ED pediatric readiness entails in terms of specialized equipment, medications, policies and protocols, safety issues, staff, and professional education.
Lack of recommended equipment needed to handle pediatric emergencies, such as laryngeal mask airways for children, was a major problem identified in the first survey. Indeed, at that time only 6% of EDs had all the recommended equipment and supplies. In contrast, the new survey showed that today EDs have a median of 91% of all recommended equipment; in higher-volume EDs that figure rises to virtually 100%, according to Dr. Gausche-Hill.
Overall, the median ED pediatric readiness score in the new survey was 69 on a 0-100 scale, up sharply from a median of 55 in an earlier nationwide survey, conducted in 2003 and published in 2007, for which Dr. Gausche-Hill also was the lead author (Pediatrics 2007;120:1229-37).
A test score of 55 gets a big red F in any classroom not grading on the curve. So this disappointing performance was an impetus for the 2009 joint guidelines, of which Dr. Gausche-Hill was a lead coauthor. The guidelines were endorsed by nearly two dozen organizations, including the American Academy of Family Physicians and the American College of Surgeons.
A theme emphasized in the joint guidelines is that all EDs need to be prepared to meet the unique needs of pediatric patients; 90% of all ED visits by patients under age 15 are to nonchildren’s hospitals, and roughly one-third of these visits are to EDs in rural and remote areas, which generally scored poorly in the initial national survey.
Another impetus for issuing the guidelines was recognition that pediatric ED visits are increasing even as the number of EDs nationwide is declining, worsening overcrowding, explained Dr. Gausche-Hill, professor of emergency medicine at the University of California, Los Angeles, and director of emergency medical services at Harbor-UCLA Medical Center.
She and her coinvestigators in the large coalition known as the National Pediatric Readiness Project sent the survey to more than 5,000 U.S. EDs. The response rate was an impressive 83%, despite the fact that the 55-question survey addressed 189 items and took a full hour to complete. Dr. Gausche-Hill attributed this remarkable response rate to on-site advocates’ passion for improved ED pediatric preparedness. Another factor: survey respondents received immediate feedback about how their hospital’s ED score compared with the average score of other hospitals with similar patient volume, as well as information about the top three things their hospital needs to do to reach a preparedness score of at least 80, which is the coalition’s short-term goal.
In analyzing the ED scores, the single most effective way most hospitals can improve their ED pediatric readiness score is for the hospital ED medical director to appoint a physician and/or a nurse pediatric emergency care coordinator. Hospitals with a coordinator scored higher, and the 42% of hospitals with both a physician and a nurse pediatric emergency care coordinator scored highest of all. Hospitals with at least one pediatric emergency care coordinator were more than fivefold more likely to have a quality improvement plan in place for ED pediatric patients, as recommended in a 2006 Institute of Medicine report that described the nation’s ED pediatric readiness as uneven.
In the new survey, 82% of EDs reported one or more barriers to compliance with the pediatric readiness guidelines. These barriers will be the focus of future quality improvement initiatives.
What’s next? Dr. Gausche-Hill said the coalition is reaching out to health care corporate groups in an effort to convince them to make changes in their hospitals.
"We’ve seen the Hospital Corporation of America make a huge initiative and change their average readiness score from 66 to 91 in the intervention hospitals. Also, Kaiser Permanente has initiated a readiness initiative," she said.
The plan is to keep the survey’s internet portal open so that hospitals can enter updated data and show continuous quality improvement. In addition, the coalition’s website – www.pediatricreadiness.org – includes tools to improve readiness.
Dr. Gausche-Hill reported having no financial conflicts regarding this project.
DALLAS – Emergency departments have substantially improved their readiness to receive pediatric patients since a 2009 multispecialty call to action was triggered by documented major deficiencies in preparedness, the results of a comprehensive new survey indicate.
"We’ve seen significant improvement in all ED patient volume classes in terms of median pediatric readiness scores. The data are pretty exciting. It says we’re moving in the right direction," Dr. Marianne Gausche-Hill said in presenting the survey findings at the annual meeting of the Society for Academic Emergency Medicine.

The key outcome measure in the national survey was a weighted ED pediatric readiness score based upon the extent to which an ED was compliant with the major recommendations of the 2009 joint American Academy of Pediatrics/American College of Emergency Physicians/Emergency Nurses Association guidelines for the care of children in EDs (Pediatrics 2009;124:1233-44.
The guidelines lay out in concrete terms what ED pediatric readiness entails in terms of specialized equipment, medications, policies and protocols, safety issues, staff, and professional education.
Lack of recommended equipment needed to handle pediatric emergencies, such as laryngeal mask airways for children, was a major problem identified in the first survey. Indeed, at that time only 6% of EDs had all the recommended equipment and supplies. In contrast, the new survey showed that today EDs have a median of 91% of all recommended equipment; in higher-volume EDs that figure rises to virtually 100%, according to Dr. Gausche-Hill.
Overall, the median ED pediatric readiness score in the new survey was 69 on a 0-100 scale, up sharply from a median of 55 in an earlier nationwide survey, conducted in 2003 and published in 2007, for which Dr. Gausche-Hill also was the lead author (Pediatrics 2007;120:1229-37).
A test score of 55 gets a big red F in any classroom not grading on the curve. So this disappointing performance was an impetus for the 2009 joint guidelines, of which Dr. Gausche-Hill was a lead coauthor. The guidelines were endorsed by nearly two dozen organizations, including the American Academy of Family Physicians and the American College of Surgeons.
A theme emphasized in the joint guidelines is that all EDs need to be prepared to meet the unique needs of pediatric patients; 90% of all ED visits by patients under age 15 are to nonchildren’s hospitals, and roughly one-third of these visits are to EDs in rural and remote areas, which generally scored poorly in the initial national survey.
Another impetus for issuing the guidelines was recognition that pediatric ED visits are increasing even as the number of EDs nationwide is declining, worsening overcrowding, explained Dr. Gausche-Hill, professor of emergency medicine at the University of California, Los Angeles, and director of emergency medical services at Harbor-UCLA Medical Center.
She and her coinvestigators in the large coalition known as the National Pediatric Readiness Project sent the survey to more than 5,000 U.S. EDs. The response rate was an impressive 83%, despite the fact that the 55-question survey addressed 189 items and took a full hour to complete. Dr. Gausche-Hill attributed this remarkable response rate to on-site advocates’ passion for improved ED pediatric preparedness. Another factor: survey respondents received immediate feedback about how their hospital’s ED score compared with the average score of other hospitals with similar patient volume, as well as information about the top three things their hospital needs to do to reach a preparedness score of at least 80, which is the coalition’s short-term goal.
In analyzing the ED scores, the single most effective way most hospitals can improve their ED pediatric readiness score is for the hospital ED medical director to appoint a physician and/or a nurse pediatric emergency care coordinator. Hospitals with a coordinator scored higher, and the 42% of hospitals with both a physician and a nurse pediatric emergency care coordinator scored highest of all. Hospitals with at least one pediatric emergency care coordinator were more than fivefold more likely to have a quality improvement plan in place for ED pediatric patients, as recommended in a 2006 Institute of Medicine report that described the nation’s ED pediatric readiness as uneven.
In the new survey, 82% of EDs reported one or more barriers to compliance with the pediatric readiness guidelines. These barriers will be the focus of future quality improvement initiatives.
What’s next? Dr. Gausche-Hill said the coalition is reaching out to health care corporate groups in an effort to convince them to make changes in their hospitals.
"We’ve seen the Hospital Corporation of America make a huge initiative and change their average readiness score from 66 to 91 in the intervention hospitals. Also, Kaiser Permanente has initiated a readiness initiative," she said.
The plan is to keep the survey’s internet portal open so that hospitals can enter updated data and show continuous quality improvement. In addition, the coalition’s website – www.pediatricreadiness.org – includes tools to improve readiness.
Dr. Gausche-Hill reported having no financial conflicts regarding this project.
DALLAS – Emergency departments have substantially improved their readiness to receive pediatric patients since a 2009 multispecialty call to action was triggered by documented major deficiencies in preparedness, the results of a comprehensive new survey indicate.
"We’ve seen significant improvement in all ED patient volume classes in terms of median pediatric readiness scores. The data are pretty exciting. It says we’re moving in the right direction," Dr. Marianne Gausche-Hill said in presenting the survey findings at the annual meeting of the Society for Academic Emergency Medicine.

The key outcome measure in the national survey was a weighted ED pediatric readiness score based upon the extent to which an ED was compliant with the major recommendations of the 2009 joint American Academy of Pediatrics/American College of Emergency Physicians/Emergency Nurses Association guidelines for the care of children in EDs (Pediatrics 2009;124:1233-44.
The guidelines lay out in concrete terms what ED pediatric readiness entails in terms of specialized equipment, medications, policies and protocols, safety issues, staff, and professional education.
Lack of recommended equipment needed to handle pediatric emergencies, such as laryngeal mask airways for children, was a major problem identified in the first survey. Indeed, at that time only 6% of EDs had all the recommended equipment and supplies. In contrast, the new survey showed that today EDs have a median of 91% of all recommended equipment; in higher-volume EDs that figure rises to virtually 100%, according to Dr. Gausche-Hill.
Overall, the median ED pediatric readiness score in the new survey was 69 on a 0-100 scale, up sharply from a median of 55 in an earlier nationwide survey, conducted in 2003 and published in 2007, for which Dr. Gausche-Hill also was the lead author (Pediatrics 2007;120:1229-37).
A test score of 55 gets a big red F in any classroom not grading on the curve. So this disappointing performance was an impetus for the 2009 joint guidelines, of which Dr. Gausche-Hill was a lead coauthor. The guidelines were endorsed by nearly two dozen organizations, including the American Academy of Family Physicians and the American College of Surgeons.
A theme emphasized in the joint guidelines is that all EDs need to be prepared to meet the unique needs of pediatric patients; 90% of all ED visits by patients under age 15 are to nonchildren’s hospitals, and roughly one-third of these visits are to EDs in rural and remote areas, which generally scored poorly in the initial national survey.
Another impetus for issuing the guidelines was recognition that pediatric ED visits are increasing even as the number of EDs nationwide is declining, worsening overcrowding, explained Dr. Gausche-Hill, professor of emergency medicine at the University of California, Los Angeles, and director of emergency medical services at Harbor-UCLA Medical Center.
She and her coinvestigators in the large coalition known as the National Pediatric Readiness Project sent the survey to more than 5,000 U.S. EDs. The response rate was an impressive 83%, despite the fact that the 55-question survey addressed 189 items and took a full hour to complete. Dr. Gausche-Hill attributed this remarkable response rate to on-site advocates’ passion for improved ED pediatric preparedness. Another factor: survey respondents received immediate feedback about how their hospital’s ED score compared with the average score of other hospitals with similar patient volume, as well as information about the top three things their hospital needs to do to reach a preparedness score of at least 80, which is the coalition’s short-term goal.
In analyzing the ED scores, the single most effective way most hospitals can improve their ED pediatric readiness score is for the hospital ED medical director to appoint a physician and/or a nurse pediatric emergency care coordinator. Hospitals with a coordinator scored higher, and the 42% of hospitals with both a physician and a nurse pediatric emergency care coordinator scored highest of all. Hospitals with at least one pediatric emergency care coordinator were more than fivefold more likely to have a quality improvement plan in place for ED pediatric patients, as recommended in a 2006 Institute of Medicine report that described the nation’s ED pediatric readiness as uneven.
In the new survey, 82% of EDs reported one or more barriers to compliance with the pediatric readiness guidelines. These barriers will be the focus of future quality improvement initiatives.
What’s next? Dr. Gausche-Hill said the coalition is reaching out to health care corporate groups in an effort to convince them to make changes in their hospitals.
"We’ve seen the Hospital Corporation of America make a huge initiative and change their average readiness score from 66 to 91 in the intervention hospitals. Also, Kaiser Permanente has initiated a readiness initiative," she said.
The plan is to keep the survey’s internet portal open so that hospitals can enter updated data and show continuous quality improvement. In addition, the coalition’s website – www.pediatricreadiness.org – includes tools to improve readiness.
Dr. Gausche-Hill reported having no financial conflicts regarding this project.
AT SAEM 2014
Key clinical point: The nation’s hospital emergency departments are markedly better prepared to treat pediatric patients than they were 7 years ago.
Major finding: The median ED pediatric readiness score in a new national survey was 69 on a 0-100 scale, as compared to 55 in a survey published 7 years ago.
Data source: This detailed survey was sent to all of the more than 5,000 EDs in the U.S. and its territories. The response rate was 83%.
Disclosures: The National Pediatric Readiness Project is supported primarily by funding from federal agencies. The study presenter reported having no financial conflicts.