User login
Head bath promising for primary headaches
DALLAS – A 30-minute cold-water head bath shows promise as a nonpharmacologic treatment for acute migraine or tension headache.
The key to this approach is that the water starts out lukewarm, cooling slowly to cold over the course of the first 15 minutes. It makes for a very different experience than abrupt immersion in an ice bath or placement of an ice pack against the head, which can actually worsen pain; indeed, many pain studies use a cold-water bath as the standardized pain stimulus, Dr. James R. Miner noted at the annual meeting of the Society for Academic Emergency Medicine.
He presented a prospective, observational, proof-of-concept study involving 18 adults who presented to an emergency department with a primary headache – that is, either migraine, migrainous headache not meeting full diagnostic criteria for migraine, or tension headache. Their mean age was 29 years, and all were free from known vascular disease. These were patients whose headaches were sufficiently severe that ED physicians had slated them for treatment with opioids, triptans, antiemetics, or other medications widely used to treat acute primary headache in the ED.
Instead, participants were placed in what Dr. Miner termed his migraine head box for 60 minutes. The jury-rigged box looks much like the sort of porcelain sink used for hair washing in salons. An icepack is placed at the bottom of the sink of lukewarm water, and the patient then lies back and submerges his or her head in the gradually chilling water to a level just below the ears.
The subjects’ median baseline self-rated pain score on a 0- to 100-mm visual analog scale was 78 mm. Ten patients described their head pain as severe and eight as moderate.
After 30 minutes in the migraine head box, patients reported a median 19.5-mm drop in their pain score, and nine patients now rated their headache as mild. And 60 minutes in the head box brought a modest additional median 2-mm reduction in pain scores, and one additional patient who rated the pain as mild, according to Dr. Miner of Hennepin County Medical Center, Minneapolis.
Seven of the 18 patients received rescue medications. In a follow-up phone call at 72 hours, one-third of subjects reported experiencing a rebound headache after leaving the ED, a rate Dr. Miner found surprisingly high.
He stressed that these are early days for the migraine head box. Planned future studies will include controls given a sham intervention, as well as measurement of water temperatures to learn if outcomes are optimized at a certain temperature. Efforts will also be made to identify the mechanism of benefit.
"I don’t think the cold water therapy has anything to do with an anti-inflammatory effect," Dr. Miner speculated. "Most likely, the low-level innocuous stimulation is causing an afferent decrease in pain at the thalamic level."
He noted that the etiology of primary headaches remains elusive.
"You can go to a lot of different meetings and hear a lot of different theories. I can say that over the course of my career the leading contender for what causes these headaches has changed almost every 3 or 4 years. But I think there’s pretty good agreement that a lot of the pain is thalamically mediated hyperesthesia, although whether this is a result of vasospasm or spreading cortical depression immediately prior to the headache is a subject of conjecture," Dr. Miner commented.
"I think one of the reasons that we struggle with so many different drug classes that are all similarly effective for these headaches is that we don’t know what’s truly causing the headache," he added.
Audience members applauded his effort to develop a fast-acting nonpharmacologic treatment for what is a very common diagnosis in the ED. As one physician noted, "I suspect ED physicians, because of the greatly increased concern about prescribing opiates, are going to be told they need to be using other forms of treatment."
Dr. Miner reported receiving grants for acute pain research from the National Institute of Justice and Taser International.
DALLAS – A 30-minute cold-water head bath shows promise as a nonpharmacologic treatment for acute migraine or tension headache.
The key to this approach is that the water starts out lukewarm, cooling slowly to cold over the course of the first 15 minutes. It makes for a very different experience than abrupt immersion in an ice bath or placement of an ice pack against the head, which can actually worsen pain; indeed, many pain studies use a cold-water bath as the standardized pain stimulus, Dr. James R. Miner noted at the annual meeting of the Society for Academic Emergency Medicine.
He presented a prospective, observational, proof-of-concept study involving 18 adults who presented to an emergency department with a primary headache – that is, either migraine, migrainous headache not meeting full diagnostic criteria for migraine, or tension headache. Their mean age was 29 years, and all were free from known vascular disease. These were patients whose headaches were sufficiently severe that ED physicians had slated them for treatment with opioids, triptans, antiemetics, or other medications widely used to treat acute primary headache in the ED.
Instead, participants were placed in what Dr. Miner termed his migraine head box for 60 minutes. The jury-rigged box looks much like the sort of porcelain sink used for hair washing in salons. An icepack is placed at the bottom of the sink of lukewarm water, and the patient then lies back and submerges his or her head in the gradually chilling water to a level just below the ears.
The subjects’ median baseline self-rated pain score on a 0- to 100-mm visual analog scale was 78 mm. Ten patients described their head pain as severe and eight as moderate.
After 30 minutes in the migraine head box, patients reported a median 19.5-mm drop in their pain score, and nine patients now rated their headache as mild. And 60 minutes in the head box brought a modest additional median 2-mm reduction in pain scores, and one additional patient who rated the pain as mild, according to Dr. Miner of Hennepin County Medical Center, Minneapolis.
Seven of the 18 patients received rescue medications. In a follow-up phone call at 72 hours, one-third of subjects reported experiencing a rebound headache after leaving the ED, a rate Dr. Miner found surprisingly high.
He stressed that these are early days for the migraine head box. Planned future studies will include controls given a sham intervention, as well as measurement of water temperatures to learn if outcomes are optimized at a certain temperature. Efforts will also be made to identify the mechanism of benefit.
"I don’t think the cold water therapy has anything to do with an anti-inflammatory effect," Dr. Miner speculated. "Most likely, the low-level innocuous stimulation is causing an afferent decrease in pain at the thalamic level."
He noted that the etiology of primary headaches remains elusive.
"You can go to a lot of different meetings and hear a lot of different theories. I can say that over the course of my career the leading contender for what causes these headaches has changed almost every 3 or 4 years. But I think there’s pretty good agreement that a lot of the pain is thalamically mediated hyperesthesia, although whether this is a result of vasospasm or spreading cortical depression immediately prior to the headache is a subject of conjecture," Dr. Miner commented.
"I think one of the reasons that we struggle with so many different drug classes that are all similarly effective for these headaches is that we don’t know what’s truly causing the headache," he added.
Audience members applauded his effort to develop a fast-acting nonpharmacologic treatment for what is a very common diagnosis in the ED. As one physician noted, "I suspect ED physicians, because of the greatly increased concern about prescribing opiates, are going to be told they need to be using other forms of treatment."
Dr. Miner reported receiving grants for acute pain research from the National Institute of Justice and Taser International.
DALLAS – A 30-minute cold-water head bath shows promise as a nonpharmacologic treatment for acute migraine or tension headache.
The key to this approach is that the water starts out lukewarm, cooling slowly to cold over the course of the first 15 minutes. It makes for a very different experience than abrupt immersion in an ice bath or placement of an ice pack against the head, which can actually worsen pain; indeed, many pain studies use a cold-water bath as the standardized pain stimulus, Dr. James R. Miner noted at the annual meeting of the Society for Academic Emergency Medicine.
He presented a prospective, observational, proof-of-concept study involving 18 adults who presented to an emergency department with a primary headache – that is, either migraine, migrainous headache not meeting full diagnostic criteria for migraine, or tension headache. Their mean age was 29 years, and all were free from known vascular disease. These were patients whose headaches were sufficiently severe that ED physicians had slated them for treatment with opioids, triptans, antiemetics, or other medications widely used to treat acute primary headache in the ED.
Instead, participants were placed in what Dr. Miner termed his migraine head box for 60 minutes. The jury-rigged box looks much like the sort of porcelain sink used for hair washing in salons. An icepack is placed at the bottom of the sink of lukewarm water, and the patient then lies back and submerges his or her head in the gradually chilling water to a level just below the ears.
The subjects’ median baseline self-rated pain score on a 0- to 100-mm visual analog scale was 78 mm. Ten patients described their head pain as severe and eight as moderate.
After 30 minutes in the migraine head box, patients reported a median 19.5-mm drop in their pain score, and nine patients now rated their headache as mild. And 60 minutes in the head box brought a modest additional median 2-mm reduction in pain scores, and one additional patient who rated the pain as mild, according to Dr. Miner of Hennepin County Medical Center, Minneapolis.
Seven of the 18 patients received rescue medications. In a follow-up phone call at 72 hours, one-third of subjects reported experiencing a rebound headache after leaving the ED, a rate Dr. Miner found surprisingly high.
He stressed that these are early days for the migraine head box. Planned future studies will include controls given a sham intervention, as well as measurement of water temperatures to learn if outcomes are optimized at a certain temperature. Efforts will also be made to identify the mechanism of benefit.
"I don’t think the cold water therapy has anything to do with an anti-inflammatory effect," Dr. Miner speculated. "Most likely, the low-level innocuous stimulation is causing an afferent decrease in pain at the thalamic level."
He noted that the etiology of primary headaches remains elusive.
"You can go to a lot of different meetings and hear a lot of different theories. I can say that over the course of my career the leading contender for what causes these headaches has changed almost every 3 or 4 years. But I think there’s pretty good agreement that a lot of the pain is thalamically mediated hyperesthesia, although whether this is a result of vasospasm or spreading cortical depression immediately prior to the headache is a subject of conjecture," Dr. Miner commented.
"I think one of the reasons that we struggle with so many different drug classes that are all similarly effective for these headaches is that we don’t know what’s truly causing the headache," he added.
Audience members applauded his effort to develop a fast-acting nonpharmacologic treatment for what is a very common diagnosis in the ED. As one physician noted, "I suspect ED physicians, because of the greatly increased concern about prescribing opiates, are going to be told they need to be using other forms of treatment."
Dr. Miner reported receiving grants for acute pain research from the National Institute of Justice and Taser International.
AT THE SAEM ANNUAL MEETING
Key clinical point: Reasonably effective treatment for primary headaches via placement of the back of a patient’s head in a slowly cooling water bath warrants further studies.
Major finding: Median self-rated head pain severity dropped from 78 on a 0-100 scale to 58.5 after 30 minutes in a migraine head box.
Data source: This was a prospective, observational, pilot study involving 18 adults who presented to an emergency department with moderate or severe migraine or tension headache.
Disclosures: The presenter reported having no financial conflicts with regard to this study.
Program Prevented Antipsychotic-induced Weight Gain in Youth
CHICAGO – An innovative multidisciplinary lifestyle intervention in youth with first-episode psychosis can prevent the marked weight gain and other adverse cardiometabolic effects that typically arise during the first months of treatment with antipsychotic agents.
"Antipsychotic-induced weight gain can be halted through individualized lifestyle and life-skills interventions. Weight stability in the face of antipsychotic therapy is a realistic and attainable goal," Dr. Katherine Samaras said at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
The multidisciplinary Australian effort, known as the Keeping the Body in Mind Program, is carried out by Dr. Samaras, an endocrinologist at St. Vincent’s Hospital in Sydney, Australia, together with a psychiatrist, a dietician, and an exercise physiologist. Their motivation in developing the program stems from studies documenting a 20-year life expectancy shortfall in patients with major mental illness, compared with the general population, which Dr. Sue Bailey, past president of the Royal College of Psychiatrists, has called "one of the biggest health scandals of our time."
In addition, as an endocrinologist Dr. Samaras was disturbed to see children and youth on antipsychotic agents in the diabetes clinic on virtually a daily basis. Her own clinical experience was underscored in a recent Tennessee Medicaid program study which found that 6- to 17-year-olds using antipsychotics were at more than threefold increased risk of type 2 diabetes. The risk was evident within the first year and grew with increasing cumulative dose (JAMA Psychiatry 2013;70:1067-75).
"As an endocrinologist, I expect youth with type 1 diabetes to have parity with respect to life expectancy, to maintain their current health, and to develop in education and life skills and have fulfilling life experiences. Imagine if we applied the diabetes care and prevention models we use every day in children with type 1 diabetes to youth with severe mental illness on antipsychotic medications," she mused.
The program is restricted to youth with first-episode psychosis who have been on antipsychotic medication for less than 4 weeks at enrollment. The program entailed weekly individualized counseling and monitoring by a dietician and an exercise physiologist, daily access to a gym converted from a staff conference room in the first-episode psychosis unit, and weekly group life-skills training classes in cooking, shopping, and budgeting.
"There may be very little family support for these people. They’re often living in shelters," Dr. Samaras explained.
She presented a 12-week pilot study involving 16 patients in the Keeping the Body in Mind Program and 12 sociodemographically similar controls in a more conventional Sydney first-episode psychosis program without lifestyle interventions. The subjects were 15-25 years old (mean age, 20 years). The most common psychiatric diagnosis was schizophreniform disorder, followed by bipolar disorder and major depression with psychotic features.
Over the course of 12 weeks, the lifestyle intervention group gained an average of 1.2 kg, compared with 7.3 kg in controls. Moreover, just 12% of the Keeping the Body in Mind Program participants experienced clinically significant weight gain, predefined by the investigators as a greater than 7% increase, compared with 75% of controls. Waist circumference, body mass index, lipids, blood pressure, and fasting blood glucose all remained essentially unchanged over time in the program participants. The group’s aerobic fitness as reflected in peak oxygen intake (VO2max) improved significantly. In contrast, all of these cardiometabolic parameters deteriorated significantly in the control group.
Dr. Samaras noted that most antipsychotic-induced weight gain occurs relatively early in the course of chronic treatment: In one representative study, the average gain was 12 kg during the first 24 months, another 4 kg in the following year, and an additional 3 kg at the 4-year mark.
However, when asked how long young patients with a first episode of major mental illness should remain involved in a lifestyle intervention program such as Keeping the Body in Mind, she was adamant: "I believe that as long as they’re on an antipsychotic agent they should receive dietetic and exercise physiologist support. The key is for us to walk along the path every step of the way for as long as these people need antipsychotics, and not to abandon them to the neglect that I think has characterized the physical health care of mental patients."
The study was supported by the Mental Health and Drug and Alcohol Office of the Ministry of Health for New South Wales. The presenter reported having no financial conflicts.
CHICAGO – An innovative multidisciplinary lifestyle intervention in youth with first-episode psychosis can prevent the marked weight gain and other adverse cardiometabolic effects that typically arise during the first months of treatment with antipsychotic agents.
"Antipsychotic-induced weight gain can be halted through individualized lifestyle and life-skills interventions. Weight stability in the face of antipsychotic therapy is a realistic and attainable goal," Dr. Katherine Samaras said at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
The multidisciplinary Australian effort, known as the Keeping the Body in Mind Program, is carried out by Dr. Samaras, an endocrinologist at St. Vincent’s Hospital in Sydney, Australia, together with a psychiatrist, a dietician, and an exercise physiologist. Their motivation in developing the program stems from studies documenting a 20-year life expectancy shortfall in patients with major mental illness, compared with the general population, which Dr. Sue Bailey, past president of the Royal College of Psychiatrists, has called "one of the biggest health scandals of our time."
In addition, as an endocrinologist Dr. Samaras was disturbed to see children and youth on antipsychotic agents in the diabetes clinic on virtually a daily basis. Her own clinical experience was underscored in a recent Tennessee Medicaid program study which found that 6- to 17-year-olds using antipsychotics were at more than threefold increased risk of type 2 diabetes. The risk was evident within the first year and grew with increasing cumulative dose (JAMA Psychiatry 2013;70:1067-75).
"As an endocrinologist, I expect youth with type 1 diabetes to have parity with respect to life expectancy, to maintain their current health, and to develop in education and life skills and have fulfilling life experiences. Imagine if we applied the diabetes care and prevention models we use every day in children with type 1 diabetes to youth with severe mental illness on antipsychotic medications," she mused.
The program is restricted to youth with first-episode psychosis who have been on antipsychotic medication for less than 4 weeks at enrollment. The program entailed weekly individualized counseling and monitoring by a dietician and an exercise physiologist, daily access to a gym converted from a staff conference room in the first-episode psychosis unit, and weekly group life-skills training classes in cooking, shopping, and budgeting.
"There may be very little family support for these people. They’re often living in shelters," Dr. Samaras explained.
She presented a 12-week pilot study involving 16 patients in the Keeping the Body in Mind Program and 12 sociodemographically similar controls in a more conventional Sydney first-episode psychosis program without lifestyle interventions. The subjects were 15-25 years old (mean age, 20 years). The most common psychiatric diagnosis was schizophreniform disorder, followed by bipolar disorder and major depression with psychotic features.
Over the course of 12 weeks, the lifestyle intervention group gained an average of 1.2 kg, compared with 7.3 kg in controls. Moreover, just 12% of the Keeping the Body in Mind Program participants experienced clinically significant weight gain, predefined by the investigators as a greater than 7% increase, compared with 75% of controls. Waist circumference, body mass index, lipids, blood pressure, and fasting blood glucose all remained essentially unchanged over time in the program participants. The group’s aerobic fitness as reflected in peak oxygen intake (VO2max) improved significantly. In contrast, all of these cardiometabolic parameters deteriorated significantly in the control group.
Dr. Samaras noted that most antipsychotic-induced weight gain occurs relatively early in the course of chronic treatment: In one representative study, the average gain was 12 kg during the first 24 months, another 4 kg in the following year, and an additional 3 kg at the 4-year mark.
However, when asked how long young patients with a first episode of major mental illness should remain involved in a lifestyle intervention program such as Keeping the Body in Mind, she was adamant: "I believe that as long as they’re on an antipsychotic agent they should receive dietetic and exercise physiologist support. The key is for us to walk along the path every step of the way for as long as these people need antipsychotics, and not to abandon them to the neglect that I think has characterized the physical health care of mental patients."
The study was supported by the Mental Health and Drug and Alcohol Office of the Ministry of Health for New South Wales. The presenter reported having no financial conflicts.
CHICAGO – An innovative multidisciplinary lifestyle intervention in youth with first-episode psychosis can prevent the marked weight gain and other adverse cardiometabolic effects that typically arise during the first months of treatment with antipsychotic agents.
"Antipsychotic-induced weight gain can be halted through individualized lifestyle and life-skills interventions. Weight stability in the face of antipsychotic therapy is a realistic and attainable goal," Dr. Katherine Samaras said at the joint meeting of the International Congress of Endocrinology and the Endocrine Society.
The multidisciplinary Australian effort, known as the Keeping the Body in Mind Program, is carried out by Dr. Samaras, an endocrinologist at St. Vincent’s Hospital in Sydney, Australia, together with a psychiatrist, a dietician, and an exercise physiologist. Their motivation in developing the program stems from studies documenting a 20-year life expectancy shortfall in patients with major mental illness, compared with the general population, which Dr. Sue Bailey, past president of the Royal College of Psychiatrists, has called "one of the biggest health scandals of our time."
In addition, as an endocrinologist Dr. Samaras was disturbed to see children and youth on antipsychotic agents in the diabetes clinic on virtually a daily basis. Her own clinical experience was underscored in a recent Tennessee Medicaid program study which found that 6- to 17-year-olds using antipsychotics were at more than threefold increased risk of type 2 diabetes. The risk was evident within the first year and grew with increasing cumulative dose (JAMA Psychiatry 2013;70:1067-75).
"As an endocrinologist, I expect youth with type 1 diabetes to have parity with respect to life expectancy, to maintain their current health, and to develop in education and life skills and have fulfilling life experiences. Imagine if we applied the diabetes care and prevention models we use every day in children with type 1 diabetes to youth with severe mental illness on antipsychotic medications," she mused.
The program is restricted to youth with first-episode psychosis who have been on antipsychotic medication for less than 4 weeks at enrollment. The program entailed weekly individualized counseling and monitoring by a dietician and an exercise physiologist, daily access to a gym converted from a staff conference room in the first-episode psychosis unit, and weekly group life-skills training classes in cooking, shopping, and budgeting.
"There may be very little family support for these people. They’re often living in shelters," Dr. Samaras explained.
She presented a 12-week pilot study involving 16 patients in the Keeping the Body in Mind Program and 12 sociodemographically similar controls in a more conventional Sydney first-episode psychosis program without lifestyle interventions. The subjects were 15-25 years old (mean age, 20 years). The most common psychiatric diagnosis was schizophreniform disorder, followed by bipolar disorder and major depression with psychotic features.
Over the course of 12 weeks, the lifestyle intervention group gained an average of 1.2 kg, compared with 7.3 kg in controls. Moreover, just 12% of the Keeping the Body in Mind Program participants experienced clinically significant weight gain, predefined by the investigators as a greater than 7% increase, compared with 75% of controls. Waist circumference, body mass index, lipids, blood pressure, and fasting blood glucose all remained essentially unchanged over time in the program participants. The group’s aerobic fitness as reflected in peak oxygen intake (VO2max) improved significantly. In contrast, all of these cardiometabolic parameters deteriorated significantly in the control group.
Dr. Samaras noted that most antipsychotic-induced weight gain occurs relatively early in the course of chronic treatment: In one representative study, the average gain was 12 kg during the first 24 months, another 4 kg in the following year, and an additional 3 kg at the 4-year mark.
However, when asked how long young patients with a first episode of major mental illness should remain involved in a lifestyle intervention program such as Keeping the Body in Mind, she was adamant: "I believe that as long as they’re on an antipsychotic agent they should receive dietetic and exercise physiologist support. The key is for us to walk along the path every step of the way for as long as these people need antipsychotics, and not to abandon them to the neglect that I think has characterized the physical health care of mental patients."
The study was supported by the Mental Health and Drug and Alcohol Office of the Ministry of Health for New South Wales. The presenter reported having no financial conflicts.
AT ICE/ENDO 2014
VIDEO: Rivaroxaban provides advantages for cardioversion in AF
BARCELONA – The first-ever prospective, randomized trial of a novel oral anticoagulant in patients with atrial fibrillation undergoing elective cardioversion showed oral rivaroxaban at 20 mg once daily to be an effective and safe alternative to standard-of-care warfarin. But the study, known as X-VeRT, also showed rivaroxaban offers something in addition: more expeditious cardioversion amenable to reliable scheduling.
Dr. Riccardo Cappato, who presented the X-VeRT results at the annual congress of the European Society of Cardiology, explains in this video interview.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
BARCELONA – The first-ever prospective, randomized trial of a novel oral anticoagulant in patients with atrial fibrillation undergoing elective cardioversion showed oral rivaroxaban at 20 mg once daily to be an effective and safe alternative to standard-of-care warfarin. But the study, known as X-VeRT, also showed rivaroxaban offers something in addition: more expeditious cardioversion amenable to reliable scheduling.
Dr. Riccardo Cappato, who presented the X-VeRT results at the annual congress of the European Society of Cardiology, explains in this video interview.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
BARCELONA – The first-ever prospective, randomized trial of a novel oral anticoagulant in patients with atrial fibrillation undergoing elective cardioversion showed oral rivaroxaban at 20 mg once daily to be an effective and safe alternative to standard-of-care warfarin. But the study, known as X-VeRT, also showed rivaroxaban offers something in addition: more expeditious cardioversion amenable to reliable scheduling.
Dr. Riccardo Cappato, who presented the X-VeRT results at the annual congress of the European Society of Cardiology, explains in this video interview.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE ESC CONGRESS 2014
Rivaroxaban shows advantages in AF cardioversion
BARCELONA – Rivaroxaban appears to be a safe and effective alternative to warfarin in patients with atrial fibrillation undergoing elective cardioversion, according to the findings of the first-ever prospective, randomized trial of a novel oral anticoagulant for this application.
Additionally, the X-VeRT trial showed that rivaroxaban (Xarelto) may offer an attractive, highly practical advantage over warfarin, the current guideline-recommended standard of care for cardioversion: Namely, rivaroxaban enabled patients to undergo the procedure more expeditiously, Dr. Riccardo Cappato noted in presenting the X-VeRT findings at the annual congress of the European Society of Cardiology.
Indeed, patients in the early-cardioversion arm of the trial safely underwent the procedure as early as 4 hours after taking their first 20-mg dose of rivaroxaban.
Moreover, patients in the delayed-conversion study arm, which required at least 3 consecutive weeks of effective oral anticoagulation preprocedurally, underwent cardioversion an average of 8 days earlier if randomized to rivaroxaban rather than to warfarin or another vitamin K antagonist. That’s because many warfarin-treated patients couldn’t maintain their international normalized ratio (INR) within the target range of 2.0-3.0 for 3 straight weeks, explained Dr. Cappato, professor of electrophysiology and chief of the arrhythmia and electrophysiology center at the University of Milan’s San Donato Polyclinic Hospital.
X-VeRT included 1,504 patients with nonvalvular atrial fibrillation (AF) at 141 centers in 16 countries. All were scheduled for elective cardioversion. They were randomized 2:1 to rivaroxaban 20 mg once daily or to warfarin at a dose adjusted to maintain an INR of 2.0-3.0.
Of those patients, 872 were assigned to an early-cardioversion strategy. Their cardioversion took place after 1-5 days on study medication, provided transesophageal echocardiography had ruled out a left atrial thrombus or they were already on chronic warfarin with their last three INRs in the target range. The other 632 patients were cardioverted using a delayed strategy in which they had to be on study medication for 3-8 weeks before the procedure.
The primary efficacy outcome in X-VeRT was the composite rate of stroke, TIA, peripheral embolism, MI, and cardiovascular death. The incidence was 0.51% in the rivaroxaban group and not statistically different at 1.02% in the warfarin group. The primary safety outcome – major bleeding – occurred in 0.6% of rivaroxaban-treated patients and 0.8% on warfarin.
The median time to cardioversion in the early-cardioversion strategy arm was similar regardless of which drug was used. Of note, however, in the delayed-strategy group, the median time to cardioversion was 22 days in patients on rivaroxaban, compared with 30 days with warfarin.
Of patients in the delayed-strategy group, 77% of those on rivaroxaban were cardioverted as scheduled, compared with 36% on warfarin. Only one patient in the rivaroxaban group was unable to undergo cardioversion before the 8-week cutoff because of inadequate anticoagulation as defined by less than 80% compliance in pill taking; in contrast, 95 warfarin-treated patients in the delayed-strategy group weren’t cardioverted because they missed the 8-week cutoff because of problematic INRs.
In an interview, ESC spokesman Dr. Jurrien Ten Berg predicted X-VeRT will be practice changing. He believes that on the basis of these study results, many physicians will view elective cardioversion as an excellent time to switch patients from warfarin, with all its inherent problems, to rivaroxaban, especially if they qualify for early cardioversion.
"I think this will absolutely change our policy. Here we’re talking about a once-daily pill that makes it possible to do a cardioversion early, and I think that’s a major advantage. If you delay the cardioversion, anything can happen. We know the vitamin K antagonists are unreliable, especially in the first weeks, when even if the INR is fine you don’t really know if the patient is well anticoagulated. For me, the totally of evidence, including the retrospective analyses of the large atrial fibrillation stroke prevention trials, is enough now to use a NOAC for several days and then do an early cardioversion," said Dr. Ten Berg, a cardiologist at St. Antonius Hospital, Nieuwegein, the Netherlands.
Discussant Dr. Christoph Bode called X-VeRT "a landmark trial" and "brilliant work."
"This should be included in the next update of the guidelines," said Dr. Bode, professor and chair of internal medicine and cardiology at the University of Freiburg, Germany.
However, Dr. Steven Nissen took a more skeptical, albeit clearly a minority, view.
"I think this study shows neither safety nor efficacy for this regimen. There were just a handful of events in both groups, too few to draw any conclusions. I don’t think the guidelines should be changed. I think the proper interpretation of the study is that it’s inconclusive," said Dr. Nissen, chair of the department of cardiovascular medicine at the Cleveland Clinic.
Dr. Cappato noted in response that he’d stated at the outset that X-VeRT, even at more than 1,500 patients, was underpowered statistically. Event rates in well-anticoagulated patients undergoing cardioversion are so low that a study to establish noninferiority for rivaroxaban would require 25,000-30,000 participants, which is not going to happen.
"Many physicians are already switching to NOACs [novel oral anticoagulants] for cardioversion despite the absence of good evidence. We thought bringing forward this solid, methodologically sound information from X-VeRT would provide more consistent support for those who are doing this or considering it," the cardiologist said.
Simultaneous with his presentation in Barcelona, the X-VeRT (Explore the Efficacy and Safety of Once-Daily Oral Rivaroxaban for the Prevention of Cardiovascular Events in Patients With Nonvalvular Atrial Fibrillation Scheduled for Cardioversion) study was published online (Eur. Heart J. 2014 [doi:10.1093/eurheart/ehu367]).
Dr. Cappato reported receiving investigator fees from and serving as a consultant to and on speakers bureaus for numerous pharmaceutical companies, including Bayer HealthCare, which sponsored X-VeRT. Dr. Bode has received honoraria from Bayer HealthCare. Dr. Ten Berg and Dr. Nissen reported having no financial conflicts.
BARCELONA – Rivaroxaban appears to be a safe and effective alternative to warfarin in patients with atrial fibrillation undergoing elective cardioversion, according to the findings of the first-ever prospective, randomized trial of a novel oral anticoagulant for this application.
Additionally, the X-VeRT trial showed that rivaroxaban (Xarelto) may offer an attractive, highly practical advantage over warfarin, the current guideline-recommended standard of care for cardioversion: Namely, rivaroxaban enabled patients to undergo the procedure more expeditiously, Dr. Riccardo Cappato noted in presenting the X-VeRT findings at the annual congress of the European Society of Cardiology.
Indeed, patients in the early-cardioversion arm of the trial safely underwent the procedure as early as 4 hours after taking their first 20-mg dose of rivaroxaban.
Moreover, patients in the delayed-conversion study arm, which required at least 3 consecutive weeks of effective oral anticoagulation preprocedurally, underwent cardioversion an average of 8 days earlier if randomized to rivaroxaban rather than to warfarin or another vitamin K antagonist. That’s because many warfarin-treated patients couldn’t maintain their international normalized ratio (INR) within the target range of 2.0-3.0 for 3 straight weeks, explained Dr. Cappato, professor of electrophysiology and chief of the arrhythmia and electrophysiology center at the University of Milan’s San Donato Polyclinic Hospital.
X-VeRT included 1,504 patients with nonvalvular atrial fibrillation (AF) at 141 centers in 16 countries. All were scheduled for elective cardioversion. They were randomized 2:1 to rivaroxaban 20 mg once daily or to warfarin at a dose adjusted to maintain an INR of 2.0-3.0.
Of those patients, 872 were assigned to an early-cardioversion strategy. Their cardioversion took place after 1-5 days on study medication, provided transesophageal echocardiography had ruled out a left atrial thrombus or they were already on chronic warfarin with their last three INRs in the target range. The other 632 patients were cardioverted using a delayed strategy in which they had to be on study medication for 3-8 weeks before the procedure.
The primary efficacy outcome in X-VeRT was the composite rate of stroke, TIA, peripheral embolism, MI, and cardiovascular death. The incidence was 0.51% in the rivaroxaban group and not statistically different at 1.02% in the warfarin group. The primary safety outcome – major bleeding – occurred in 0.6% of rivaroxaban-treated patients and 0.8% on warfarin.
The median time to cardioversion in the early-cardioversion strategy arm was similar regardless of which drug was used. Of note, however, in the delayed-strategy group, the median time to cardioversion was 22 days in patients on rivaroxaban, compared with 30 days with warfarin.
Of patients in the delayed-strategy group, 77% of those on rivaroxaban were cardioverted as scheduled, compared with 36% on warfarin. Only one patient in the rivaroxaban group was unable to undergo cardioversion before the 8-week cutoff because of inadequate anticoagulation as defined by less than 80% compliance in pill taking; in contrast, 95 warfarin-treated patients in the delayed-strategy group weren’t cardioverted because they missed the 8-week cutoff because of problematic INRs.
In an interview, ESC spokesman Dr. Jurrien Ten Berg predicted X-VeRT will be practice changing. He believes that on the basis of these study results, many physicians will view elective cardioversion as an excellent time to switch patients from warfarin, with all its inherent problems, to rivaroxaban, especially if they qualify for early cardioversion.
"I think this will absolutely change our policy. Here we’re talking about a once-daily pill that makes it possible to do a cardioversion early, and I think that’s a major advantage. If you delay the cardioversion, anything can happen. We know the vitamin K antagonists are unreliable, especially in the first weeks, when even if the INR is fine you don’t really know if the patient is well anticoagulated. For me, the totally of evidence, including the retrospective analyses of the large atrial fibrillation stroke prevention trials, is enough now to use a NOAC for several days and then do an early cardioversion," said Dr. Ten Berg, a cardiologist at St. Antonius Hospital, Nieuwegein, the Netherlands.
Discussant Dr. Christoph Bode called X-VeRT "a landmark trial" and "brilliant work."
"This should be included in the next update of the guidelines," said Dr. Bode, professor and chair of internal medicine and cardiology at the University of Freiburg, Germany.
However, Dr. Steven Nissen took a more skeptical, albeit clearly a minority, view.
"I think this study shows neither safety nor efficacy for this regimen. There were just a handful of events in both groups, too few to draw any conclusions. I don’t think the guidelines should be changed. I think the proper interpretation of the study is that it’s inconclusive," said Dr. Nissen, chair of the department of cardiovascular medicine at the Cleveland Clinic.
Dr. Cappato noted in response that he’d stated at the outset that X-VeRT, even at more than 1,500 patients, was underpowered statistically. Event rates in well-anticoagulated patients undergoing cardioversion are so low that a study to establish noninferiority for rivaroxaban would require 25,000-30,000 participants, which is not going to happen.
"Many physicians are already switching to NOACs [novel oral anticoagulants] for cardioversion despite the absence of good evidence. We thought bringing forward this solid, methodologically sound information from X-VeRT would provide more consistent support for those who are doing this or considering it," the cardiologist said.
Simultaneous with his presentation in Barcelona, the X-VeRT (Explore the Efficacy and Safety of Once-Daily Oral Rivaroxaban for the Prevention of Cardiovascular Events in Patients With Nonvalvular Atrial Fibrillation Scheduled for Cardioversion) study was published online (Eur. Heart J. 2014 [doi:10.1093/eurheart/ehu367]).
Dr. Cappato reported receiving investigator fees from and serving as a consultant to and on speakers bureaus for numerous pharmaceutical companies, including Bayer HealthCare, which sponsored X-VeRT. Dr. Bode has received honoraria from Bayer HealthCare. Dr. Ten Berg and Dr. Nissen reported having no financial conflicts.
BARCELONA – Rivaroxaban appears to be a safe and effective alternative to warfarin in patients with atrial fibrillation undergoing elective cardioversion, according to the findings of the first-ever prospective, randomized trial of a novel oral anticoagulant for this application.
Additionally, the X-VeRT trial showed that rivaroxaban (Xarelto) may offer an attractive, highly practical advantage over warfarin, the current guideline-recommended standard of care for cardioversion: Namely, rivaroxaban enabled patients to undergo the procedure more expeditiously, Dr. Riccardo Cappato noted in presenting the X-VeRT findings at the annual congress of the European Society of Cardiology.
Indeed, patients in the early-cardioversion arm of the trial safely underwent the procedure as early as 4 hours after taking their first 20-mg dose of rivaroxaban.
Moreover, patients in the delayed-conversion study arm, which required at least 3 consecutive weeks of effective oral anticoagulation preprocedurally, underwent cardioversion an average of 8 days earlier if randomized to rivaroxaban rather than to warfarin or another vitamin K antagonist. That’s because many warfarin-treated patients couldn’t maintain their international normalized ratio (INR) within the target range of 2.0-3.0 for 3 straight weeks, explained Dr. Cappato, professor of electrophysiology and chief of the arrhythmia and electrophysiology center at the University of Milan’s San Donato Polyclinic Hospital.
X-VeRT included 1,504 patients with nonvalvular atrial fibrillation (AF) at 141 centers in 16 countries. All were scheduled for elective cardioversion. They were randomized 2:1 to rivaroxaban 20 mg once daily or to warfarin at a dose adjusted to maintain an INR of 2.0-3.0.
Of those patients, 872 were assigned to an early-cardioversion strategy. Their cardioversion took place after 1-5 days on study medication, provided transesophageal echocardiography had ruled out a left atrial thrombus or they were already on chronic warfarin with their last three INRs in the target range. The other 632 patients were cardioverted using a delayed strategy in which they had to be on study medication for 3-8 weeks before the procedure.
The primary efficacy outcome in X-VeRT was the composite rate of stroke, TIA, peripheral embolism, MI, and cardiovascular death. The incidence was 0.51% in the rivaroxaban group and not statistically different at 1.02% in the warfarin group. The primary safety outcome – major bleeding – occurred in 0.6% of rivaroxaban-treated patients and 0.8% on warfarin.
The median time to cardioversion in the early-cardioversion strategy arm was similar regardless of which drug was used. Of note, however, in the delayed-strategy group, the median time to cardioversion was 22 days in patients on rivaroxaban, compared with 30 days with warfarin.
Of patients in the delayed-strategy group, 77% of those on rivaroxaban were cardioverted as scheduled, compared with 36% on warfarin. Only one patient in the rivaroxaban group was unable to undergo cardioversion before the 8-week cutoff because of inadequate anticoagulation as defined by less than 80% compliance in pill taking; in contrast, 95 warfarin-treated patients in the delayed-strategy group weren’t cardioverted because they missed the 8-week cutoff because of problematic INRs.
In an interview, ESC spokesman Dr. Jurrien Ten Berg predicted X-VeRT will be practice changing. He believes that on the basis of these study results, many physicians will view elective cardioversion as an excellent time to switch patients from warfarin, with all its inherent problems, to rivaroxaban, especially if they qualify for early cardioversion.
"I think this will absolutely change our policy. Here we’re talking about a once-daily pill that makes it possible to do a cardioversion early, and I think that’s a major advantage. If you delay the cardioversion, anything can happen. We know the vitamin K antagonists are unreliable, especially in the first weeks, when even if the INR is fine you don’t really know if the patient is well anticoagulated. For me, the totally of evidence, including the retrospective analyses of the large atrial fibrillation stroke prevention trials, is enough now to use a NOAC for several days and then do an early cardioversion," said Dr. Ten Berg, a cardiologist at St. Antonius Hospital, Nieuwegein, the Netherlands.
Discussant Dr. Christoph Bode called X-VeRT "a landmark trial" and "brilliant work."
"This should be included in the next update of the guidelines," said Dr. Bode, professor and chair of internal medicine and cardiology at the University of Freiburg, Germany.
However, Dr. Steven Nissen took a more skeptical, albeit clearly a minority, view.
"I think this study shows neither safety nor efficacy for this regimen. There were just a handful of events in both groups, too few to draw any conclusions. I don’t think the guidelines should be changed. I think the proper interpretation of the study is that it’s inconclusive," said Dr. Nissen, chair of the department of cardiovascular medicine at the Cleveland Clinic.
Dr. Cappato noted in response that he’d stated at the outset that X-VeRT, even at more than 1,500 patients, was underpowered statistically. Event rates in well-anticoagulated patients undergoing cardioversion are so low that a study to establish noninferiority for rivaroxaban would require 25,000-30,000 participants, which is not going to happen.
"Many physicians are already switching to NOACs [novel oral anticoagulants] for cardioversion despite the absence of good evidence. We thought bringing forward this solid, methodologically sound information from X-VeRT would provide more consistent support for those who are doing this or considering it," the cardiologist said.
Simultaneous with his presentation in Barcelona, the X-VeRT (Explore the Efficacy and Safety of Once-Daily Oral Rivaroxaban for the Prevention of Cardiovascular Events in Patients With Nonvalvular Atrial Fibrillation Scheduled for Cardioversion) study was published online (Eur. Heart J. 2014 [doi:10.1093/eurheart/ehu367]).
Dr. Cappato reported receiving investigator fees from and serving as a consultant to and on speakers bureaus for numerous pharmaceutical companies, including Bayer HealthCare, which sponsored X-VeRT. Dr. Bode has received honoraria from Bayer HealthCare. Dr. Ten Berg and Dr. Nissen reported having no financial conflicts.
AT THE ESC CONGRESS 2014
Key clinical point: Patients in atrial fibrillation were able to safely undergo cardioversion as early as 4 hours after taking their first 20-mg dose of oral rivaroxaban.
Major finding: Patients with AF assigned to rivaroxaban in conjunction with cardioversion had a 0.51% rate of major cardiovascular events, similar to the 1.02% rate in patients on warfarin or other vitamin K antagonists. These rates weren’t statistically different; nor were the major bleeding rates in the two groups.
Data source: X-VeRT was a randomized, prospective, open-label, phase IIIb clinical trial involving 1,504 patients at 141 centers in 16 countries.
Disclosures: The X-VeRT trial was sponsored by Bayer HealthCare. The presenter serves as a consultant to and on speakers bureaus for Bayer and other pharmaceutical companies.
VIDEO: Long-term impact of high cholesterol in younger adults underappreciated
BARCELONA – Dr. Ann Marie Navar-Boggan has a beef with current American and European cholesterol guidelines, which she believes seriously underestimate the long-term adverse impact of hypercholesterolemia in younger adults.
Here are the data Dr. Navar-Boggan of the Duke Clinical Research Institute, Durham, N.C., presented at the annual congress of the European Society of Cardiology to bolster her argument for more aggressive treatment of hypercholesterolemia in this population.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
BARCELONA – Dr. Ann Marie Navar-Boggan has a beef with current American and European cholesterol guidelines, which she believes seriously underestimate the long-term adverse impact of hypercholesterolemia in younger adults.
Here are the data Dr. Navar-Boggan of the Duke Clinical Research Institute, Durham, N.C., presented at the annual congress of the European Society of Cardiology to bolster her argument for more aggressive treatment of hypercholesterolemia in this population.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
BARCELONA – Dr. Ann Marie Navar-Boggan has a beef with current American and European cholesterol guidelines, which she believes seriously underestimate the long-term adverse impact of hypercholesterolemia in younger adults.
Here are the data Dr. Navar-Boggan of the Duke Clinical Research Institute, Durham, N.C., presented at the annual congress of the European Society of Cardiology to bolster her argument for more aggressive treatment of hypercholesterolemia in this population.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE ESC CONGRESS 2014
VIDEO: Less may be best when ablating persistent AF
BARCELONA – Current guidelines recommend more extensive ablation procedures beyond pulmonary vein isolation for persistent atrial fibrillation. At the annual congress of the European Society of Cardiology, Dr. Atul Verma explains in this video interview that the fresh results of STAR-AF 2, the largest-ever randomized trial of popular ablation strategies in persistent AF, turn the conventional wisdom on its head and suggest that less ablation may be more.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
BARCELONA – Current guidelines recommend more extensive ablation procedures beyond pulmonary vein isolation for persistent atrial fibrillation. At the annual congress of the European Society of Cardiology, Dr. Atul Verma explains in this video interview that the fresh results of STAR-AF 2, the largest-ever randomized trial of popular ablation strategies in persistent AF, turn the conventional wisdom on its head and suggest that less ablation may be more.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
BARCELONA – Current guidelines recommend more extensive ablation procedures beyond pulmonary vein isolation for persistent atrial fibrillation. At the annual congress of the European Society of Cardiology, Dr. Atul Verma explains in this video interview that the fresh results of STAR-AF 2, the largest-ever randomized trial of popular ablation strategies in persistent AF, turn the conventional wisdom on its head and suggest that less ablation may be more.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE ESC CONGRESS 2014
Onychomycosis: Not Just For Adults
COEUR D’ALENE, IDAHO – Conventional wisdom holds that onychomycosis is rare in children. Not so.
Fully one-third of children and adolescents who presented with a nail complaint to a prominent dermatologic nail disorders center were diagnosed with mycologically confirmed onychomycosis, Dr. Julie Jefferson reported at the annual meeting of the Society for Pediatric Dermatology.
That’s a substantially higher prevalence than the 15.5% figure reported by Spanish investigators in a 20-year retrospective study (Mycoses 2011;54:450-3). It’s also well below the 47% prevalence recently reported in Denver in a 5-year retrospective study, where Trychophyton rubrum was the most common pathogen, and the highest prevalence of onychomycosis in the pediatric population was seen in 6- to 10-year-olds (Pediatr. Dermatol. 2014;31:106-8), noted Dr. Jefferson of Johns Hopkins University, Baltimore.
The investigators in both Spain and Denver observed that the prevalence of pediatric onychomycosis appears to be increasing in recent years.
Dr. Jefferson presented a retrospective study that included 917 patients up to age 18 years who presented to the Oregon Dermatology and Research Center, Portland, in a recent 6-year period. One or more nail disorders were diagnosed in 11%. The mean age at presentation was 9.4 years, with a mean 2.4-year duration of the condition prior to presentation. Toenails were affected in 47 patients, fingernails in 37, and both in 18. Fourteen patients had two nail disorders, 9 had three, and 2 had four distinct nail disorders.
The etiologies ranged widely, from infections to congenital and hereditary malformations, tumors, inflammatory processes, and systemic diseases.
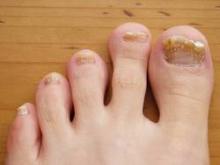
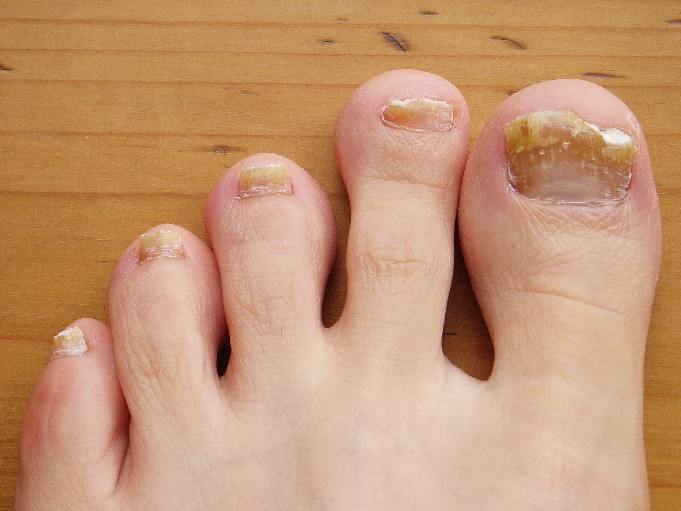
The most common nail condition was onychomycosis, diagnosed in 34 patients. Thus, 3.7% of all pediatric patients presenting to the dermatology center for any reason were diagnosed with onychomycosis, a higher rate than previously reported by others.
Other conditions included 13 cases of longitudinal melanonychia, 11 of disappearing nail bed, 9 of retronychia, 8 cases of congenital malalignment, 8 cases of trachyonychia, 7 of paronychia, 5 cases of psoriasis, and 2 of lichen planus.
Three of four patients with an ingrown toenail also had congenital malalignment of the affected great toenail, supporting the notion put forth by other investigators that congenital malalignment of the great toenail predisposes to ingrown toenails, according to Dr. Jefferson.
She reported having no financial conflicts related to this study.
COEUR D’ALENE, IDAHO – Conventional wisdom holds that onychomycosis is rare in children. Not so.
Fully one-third of children and adolescents who presented with a nail complaint to a prominent dermatologic nail disorders center were diagnosed with mycologically confirmed onychomycosis, Dr. Julie Jefferson reported at the annual meeting of the Society for Pediatric Dermatology.
That’s a substantially higher prevalence than the 15.5% figure reported by Spanish investigators in a 20-year retrospective study (Mycoses 2011;54:450-3). It’s also well below the 47% prevalence recently reported in Denver in a 5-year retrospective study, where Trychophyton rubrum was the most common pathogen, and the highest prevalence of onychomycosis in the pediatric population was seen in 6- to 10-year-olds (Pediatr. Dermatol. 2014;31:106-8), noted Dr. Jefferson of Johns Hopkins University, Baltimore.
The investigators in both Spain and Denver observed that the prevalence of pediatric onychomycosis appears to be increasing in recent years.
Dr. Jefferson presented a retrospective study that included 917 patients up to age 18 years who presented to the Oregon Dermatology and Research Center, Portland, in a recent 6-year period. One or more nail disorders were diagnosed in 11%. The mean age at presentation was 9.4 years, with a mean 2.4-year duration of the condition prior to presentation. Toenails were affected in 47 patients, fingernails in 37, and both in 18. Fourteen patients had two nail disorders, 9 had three, and 2 had four distinct nail disorders.
The etiologies ranged widely, from infections to congenital and hereditary malformations, tumors, inflammatory processes, and systemic diseases.
The most common nail condition was onychomycosis, diagnosed in 34 patients. Thus, 3.7% of all pediatric patients presenting to the dermatology center for any reason were diagnosed with onychomycosis, a higher rate than previously reported by others.
Other conditions included 13 cases of longitudinal melanonychia, 11 of disappearing nail bed, 9 of retronychia, 8 cases of congenital malalignment, 8 cases of trachyonychia, 7 of paronychia, 5 cases of psoriasis, and 2 of lichen planus.
Three of four patients with an ingrown toenail also had congenital malalignment of the affected great toenail, supporting the notion put forth by other investigators that congenital malalignment of the great toenail predisposes to ingrown toenails, according to Dr. Jefferson.
She reported having no financial conflicts related to this study.
COEUR D’ALENE, IDAHO – Conventional wisdom holds that onychomycosis is rare in children. Not so.
Fully one-third of children and adolescents who presented with a nail complaint to a prominent dermatologic nail disorders center were diagnosed with mycologically confirmed onychomycosis, Dr. Julie Jefferson reported at the annual meeting of the Society for Pediatric Dermatology.
That’s a substantially higher prevalence than the 15.5% figure reported by Spanish investigators in a 20-year retrospective study (Mycoses 2011;54:450-3). It’s also well below the 47% prevalence recently reported in Denver in a 5-year retrospective study, where Trychophyton rubrum was the most common pathogen, and the highest prevalence of onychomycosis in the pediatric population was seen in 6- to 10-year-olds (Pediatr. Dermatol. 2014;31:106-8), noted Dr. Jefferson of Johns Hopkins University, Baltimore.
The investigators in both Spain and Denver observed that the prevalence of pediatric onychomycosis appears to be increasing in recent years.
Dr. Jefferson presented a retrospective study that included 917 patients up to age 18 years who presented to the Oregon Dermatology and Research Center, Portland, in a recent 6-year period. One or more nail disorders were diagnosed in 11%. The mean age at presentation was 9.4 years, with a mean 2.4-year duration of the condition prior to presentation. Toenails were affected in 47 patients, fingernails in 37, and both in 18. Fourteen patients had two nail disorders, 9 had three, and 2 had four distinct nail disorders.
The etiologies ranged widely, from infections to congenital and hereditary malformations, tumors, inflammatory processes, and systemic diseases.
The most common nail condition was onychomycosis, diagnosed in 34 patients. Thus, 3.7% of all pediatric patients presenting to the dermatology center for any reason were diagnosed with onychomycosis, a higher rate than previously reported by others.
Other conditions included 13 cases of longitudinal melanonychia, 11 of disappearing nail bed, 9 of retronychia, 8 cases of congenital malalignment, 8 cases of trachyonychia, 7 of paronychia, 5 cases of psoriasis, and 2 of lichen planus.
Three of four patients with an ingrown toenail also had congenital malalignment of the affected great toenail, supporting the notion put forth by other investigators that congenital malalignment of the great toenail predisposes to ingrown toenails, according to Dr. Jefferson.
She reported having no financial conflicts related to this study.
AT THE SPD ANNUAL MEETING
Emergency Physicians May Be Overprescribing PPIs
DALLAS – The frequency at which U.S. emergency department physicians prescribed proton pump inhibitors more than doubled during 2001-2010, despite mounting safety concerns surrounding this class of medications.
"More education may be needed to ensure ED providers are familiar with the appropriate indications for PPI use. The big thing that I’m hoping will be taken away from this study is that because of the increase in prescribing PPIs [proton pump inhibitors] and the concerns about safety, that we’re going to be more vigilant in educating ourselves and each other about appropriate use of these medications," Dr. Maryann Mazer-Amirshahi said at the annual meeting of the Society for Academic Emergency Medicine.
Overprescribing of PPIs has been well documented in primary care offices, gastroenterology clinics, and inpatient settings. Up until now, however, prescribing patterns in the ED haven’t been well documented. The Centers for Disease Control and Prevention’s annual National Hospital Ambulatory Care Survey provided an opportunity to do so via a weighted nationally representative sample of ED visits, explained Dr. Mazer-Amirshahi of Children’s National Medical Center, Washington.
She presented a retrospective analysis of survey data for the years 2001-2010, during which the annual number of adult ED visits climbed from 20.1 million to 28.3 million. Meanwhile, PPI prescribing increased from 3% of adult patients in 2001 to 7.2% in 2010.
"I think that’s pretty significant when you’re talking about more than 7% of 28 million ED visits every year," she commented.
While PPI prescribing more than doubled during the study years, the use of alternative medications declined. Histamine2 blocker use dropped from 6.8% in 2001 to 5.7% in 2010, while the use of antacids decreased from 7.2% to 5.5%.
PPI prescribing rose in EDs in hospitals of all types: nonprofit, for-profit, and government. It increased in all regions of the country and across all payer types, including self-payment. Of note, the number of ED prescriptions increased to a greater extent in teaching hospitals, with a 276% increase, as compared with a 118% increase in nonteaching hospitals. Prescribing of PPIs by attending ED physicians climbed by 122%, by 185% by emergency medicine residents, and by 345% by mid-level providers.
In 2001, 3.3% of ED patients aged 65 years or older received a PPI. By 2010, this figure had climbed to 6.8%, a 104% increase. This trend is of particular concern because the elderly are the group at highest risk of PPI-related adverse events, including osteoporotic fractures, hypomagnesemia, drug-drug interactions, stent thrombosis, Clostridium difficile colitis, and community acquired pneumonia, Dr. Mazer-Amirshahi noted.
Roughly half of patients who got a PPI in the ED during the study years did not have a clear gastrointestinal complaint as the primary reason for their visit, suggesting that much of the ED prescribing of PPIs was not for an approved indication, she continued.
Dr. Mazer-Amirshahi observed that PPI prescribing has received special attention in the Choosing Wisely Program sponsored by the American Board of Internal Medicine, which recommends conducting a drug regimen review before prescribing a PPI in order to avoid drug-drug interactions. It’s also important to know whether a patient has osteoporosis before prescribing a PPI for longer than a few weeks. And there are additional reasons to think twice before prescribing a PPI in the ED.
"In the ED, we generally want to give our patients rapid symptom relief. PPIs have a delayed onset of action. They take 12-24 hours to take effect, so in many situations we might be better off giving an H2 blocker, which acts faster and is less costly," she said.
Dr. Mazer-Amirshahi reported having no financial conflicts regarding her study.
DALLAS – The frequency at which U.S. emergency department physicians prescribed proton pump inhibitors more than doubled during 2001-2010, despite mounting safety concerns surrounding this class of medications.
"More education may be needed to ensure ED providers are familiar with the appropriate indications for PPI use. The big thing that I’m hoping will be taken away from this study is that because of the increase in prescribing PPIs [proton pump inhibitors] and the concerns about safety, that we’re going to be more vigilant in educating ourselves and each other about appropriate use of these medications," Dr. Maryann Mazer-Amirshahi said at the annual meeting of the Society for Academic Emergency Medicine.
Overprescribing of PPIs has been well documented in primary care offices, gastroenterology clinics, and inpatient settings. Up until now, however, prescribing patterns in the ED haven’t been well documented. The Centers for Disease Control and Prevention’s annual National Hospital Ambulatory Care Survey provided an opportunity to do so via a weighted nationally representative sample of ED visits, explained Dr. Mazer-Amirshahi of Children’s National Medical Center, Washington.
She presented a retrospective analysis of survey data for the years 2001-2010, during which the annual number of adult ED visits climbed from 20.1 million to 28.3 million. Meanwhile, PPI prescribing increased from 3% of adult patients in 2001 to 7.2% in 2010.
"I think that’s pretty significant when you’re talking about more than 7% of 28 million ED visits every year," she commented.
While PPI prescribing more than doubled during the study years, the use of alternative medications declined. Histamine2 blocker use dropped from 6.8% in 2001 to 5.7% in 2010, while the use of antacids decreased from 7.2% to 5.5%.
PPI prescribing rose in EDs in hospitals of all types: nonprofit, for-profit, and government. It increased in all regions of the country and across all payer types, including self-payment. Of note, the number of ED prescriptions increased to a greater extent in teaching hospitals, with a 276% increase, as compared with a 118% increase in nonteaching hospitals. Prescribing of PPIs by attending ED physicians climbed by 122%, by 185% by emergency medicine residents, and by 345% by mid-level providers.
In 2001, 3.3% of ED patients aged 65 years or older received a PPI. By 2010, this figure had climbed to 6.8%, a 104% increase. This trend is of particular concern because the elderly are the group at highest risk of PPI-related adverse events, including osteoporotic fractures, hypomagnesemia, drug-drug interactions, stent thrombosis, Clostridium difficile colitis, and community acquired pneumonia, Dr. Mazer-Amirshahi noted.
Roughly half of patients who got a PPI in the ED during the study years did not have a clear gastrointestinal complaint as the primary reason for their visit, suggesting that much of the ED prescribing of PPIs was not for an approved indication, she continued.
Dr. Mazer-Amirshahi observed that PPI prescribing has received special attention in the Choosing Wisely Program sponsored by the American Board of Internal Medicine, which recommends conducting a drug regimen review before prescribing a PPI in order to avoid drug-drug interactions. It’s also important to know whether a patient has osteoporosis before prescribing a PPI for longer than a few weeks. And there are additional reasons to think twice before prescribing a PPI in the ED.
"In the ED, we generally want to give our patients rapid symptom relief. PPIs have a delayed onset of action. They take 12-24 hours to take effect, so in many situations we might be better off giving an H2 blocker, which acts faster and is less costly," she said.
Dr. Mazer-Amirshahi reported having no financial conflicts regarding her study.
DALLAS – The frequency at which U.S. emergency department physicians prescribed proton pump inhibitors more than doubled during 2001-2010, despite mounting safety concerns surrounding this class of medications.
"More education may be needed to ensure ED providers are familiar with the appropriate indications for PPI use. The big thing that I’m hoping will be taken away from this study is that because of the increase in prescribing PPIs [proton pump inhibitors] and the concerns about safety, that we’re going to be more vigilant in educating ourselves and each other about appropriate use of these medications," Dr. Maryann Mazer-Amirshahi said at the annual meeting of the Society for Academic Emergency Medicine.
Overprescribing of PPIs has been well documented in primary care offices, gastroenterology clinics, and inpatient settings. Up until now, however, prescribing patterns in the ED haven’t been well documented. The Centers for Disease Control and Prevention’s annual National Hospital Ambulatory Care Survey provided an opportunity to do so via a weighted nationally representative sample of ED visits, explained Dr. Mazer-Amirshahi of Children’s National Medical Center, Washington.
She presented a retrospective analysis of survey data for the years 2001-2010, during which the annual number of adult ED visits climbed from 20.1 million to 28.3 million. Meanwhile, PPI prescribing increased from 3% of adult patients in 2001 to 7.2% in 2010.
"I think that’s pretty significant when you’re talking about more than 7% of 28 million ED visits every year," she commented.
While PPI prescribing more than doubled during the study years, the use of alternative medications declined. Histamine2 blocker use dropped from 6.8% in 2001 to 5.7% in 2010, while the use of antacids decreased from 7.2% to 5.5%.
PPI prescribing rose in EDs in hospitals of all types: nonprofit, for-profit, and government. It increased in all regions of the country and across all payer types, including self-payment. Of note, the number of ED prescriptions increased to a greater extent in teaching hospitals, with a 276% increase, as compared with a 118% increase in nonteaching hospitals. Prescribing of PPIs by attending ED physicians climbed by 122%, by 185% by emergency medicine residents, and by 345% by mid-level providers.
In 2001, 3.3% of ED patients aged 65 years or older received a PPI. By 2010, this figure had climbed to 6.8%, a 104% increase. This trend is of particular concern because the elderly are the group at highest risk of PPI-related adverse events, including osteoporotic fractures, hypomagnesemia, drug-drug interactions, stent thrombosis, Clostridium difficile colitis, and community acquired pneumonia, Dr. Mazer-Amirshahi noted.
Roughly half of patients who got a PPI in the ED during the study years did not have a clear gastrointestinal complaint as the primary reason for their visit, suggesting that much of the ED prescribing of PPIs was not for an approved indication, she continued.
Dr. Mazer-Amirshahi observed that PPI prescribing has received special attention in the Choosing Wisely Program sponsored by the American Board of Internal Medicine, which recommends conducting a drug regimen review before prescribing a PPI in order to avoid drug-drug interactions. It’s also important to know whether a patient has osteoporosis before prescribing a PPI for longer than a few weeks. And there are additional reasons to think twice before prescribing a PPI in the ED.
"In the ED, we generally want to give our patients rapid symptom relief. PPIs have a delayed onset of action. They take 12-24 hours to take effect, so in many situations we might be better off giving an H2 blocker, which acts faster and is less costly," she said.
Dr. Mazer-Amirshahi reported having no financial conflicts regarding her study.
AT SAEM 2014
VIDEO: Alirocumab study showing sharp reduction in MACE promising but not definitive
CHICAGO – Discussant Dr. Robert M. Califf called the results of the ODYSSEY LONG TERM trial presented by Dr. Jennifer G. Robinson at the annual congress of the European Society of Cardiology "alluring" and "fantastic," with a reported 54% reduction in major adverse cardiovascular events in high-risk, statin-treated patients on add-on alirocumab. But neither he nor Dr. Robinson consider these results to be the final word on the subject.
Here’s what Dr. Robinson had to say about the investigational PCSK9 inhibitor’s performance in the study.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – Discussant Dr. Robert M. Califf called the results of the ODYSSEY LONG TERM trial presented by Dr. Jennifer G. Robinson at the annual congress of the European Society of Cardiology "alluring" and "fantastic," with a reported 54% reduction in major adverse cardiovascular events in high-risk, statin-treated patients on add-on alirocumab. But neither he nor Dr. Robinson consider these results to be the final word on the subject.
Here’s what Dr. Robinson had to say about the investigational PCSK9 inhibitor’s performance in the study.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – Discussant Dr. Robert M. Califf called the results of the ODYSSEY LONG TERM trial presented by Dr. Jennifer G. Robinson at the annual congress of the European Society of Cardiology "alluring" and "fantastic," with a reported 54% reduction in major adverse cardiovascular events in high-risk, statin-treated patients on add-on alirocumab. But neither he nor Dr. Robinson consider these results to be the final word on the subject.
Here’s what Dr. Robinson had to say about the investigational PCSK9 inhibitor’s performance in the study.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE ESC CONGRESS 2014
Alirocumab sharply reduced major cardiovascular events
BARCELONA – When added to maximally tolerated statin therapy, the investigational PCSK9 inhibitor alirocumab resulted in a further 54% reduction in major cardiovascular events among high-cardiovascular-risk patients, based on a post-hoc analysis of a large randomized controlled Phase-3 trial.
The ODYSSEY LONG TERM trial is the largest and longest study of a PCSK9 (proprotein convertase subtilisin/kexin type 9) inhibitor to report results to date, with roughly 1,900 patient-years of double-blind exposure to alirocumab. And although the ongoing trial is primarily a safety study, it is also now the first PCSK9 trial to provide what everyone watching the development of this novel drug class has been eagerly awaiting: clinical outcomes data, albeit in this case from a post-hoc secondary analysis.
“This is the first trial with any of the PCSK9 inhibitors to suggest that there will be a further significant reduction in cardiovascular events when added on to maximized statin therapy,” Dr. Jennifer G. Robinson said in presenting interim results of ODYSSEY LONG TERM at the annual congress of the European Society of Cardiology.
“We’re on the right track in terms of trying to achieve further reduction in cardiovascular events through additional lipid lowering. But this is not the definitive evidence. We need the prospective outcomes trials to validate this data and also to establish the long-term safety of these drugs when added to the statins,” cautioned Dr. Robinson, professor of epidemiology and of medicine and director of the prevention intervention center at the University of Iowa, Iowa City.
Nonetheless, on the basis of the dramatic LDL-lowering and reassuring evidence of safety shown in ODYSSEY LONG TERM and the other double-blind phase III trials presented at the congress, Sanofi and Regeneron announced plans to file for U.S. and European Union marketing approval of alirocumab before the end of the year. The proposed indication will be for LDL lowering, which regulatory agencies have accepted as a surrogate endpoint for prevention of clinical events.
Meanwhile, the definitive ODYSSEY OUTCOMES trial is underway in 18,000 patients with acute coronary syndromes, with prospective evaluation of CV outcomes as its primary endpoint. The composite endpoint employed in the big OUTCOMES trial is identical to that used in the ODYSSEY LONG TERM post hoc analysis.
ODYSSEY LONG TERM includes 2,341 patients at high CV risk and an LDL level greater than 70 mg/dL despite maximally tolerated statin therapy. The patients fall into two categories: those with heterozygous familial hypercholesterolemia and others at very high risk because of known coronary heart disease. Participants were randomized 2:1 to 150 mg of alirocumab by self-administered subcutaneous injection at home every 2 weeks or placebo in addition to their statin.
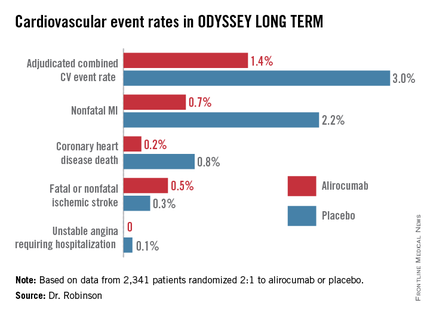
In the interim post-hoc efficacy analysis at 65 weeks, the combined rate of cardiac death, nonfatal MI, stroke, and unstable angina requiring hospitalization was 1.4% in the alirocumab arm compared to 3.0% in placebo-treated controls, for a highly significant 54% relative risk reduction (see graphic).
At 24 weeks, the alirocumab group showed a mean 62% reduction in LDL compared to placebo, a difference that remained constant at 52 weeks. The average LDL level at 52 weeks in the alirocumab group was 53 mg/dL, down from 123 mg/dL on active treatment at baseline; 79% of alirocumab-treated patients achieved an LDL below 70 mg/dL.
The incidence and types of adverse events in the alirocumab arm were essentially the same as with placebo, with no signal of problems in any domains, including neurocognitive function or allergic reactions.
ODYSSEY FH I and FH II
In a separate presentation, Dr. Michel Farnier reported on the alirocumab experience in 735 patients with heterozygous familial hypercholesterolemia in two phase III trials known as ODYSSEY FH I and FH II. At baseline, all were above their LDL goal despite maximally tolerated statin therapy, in two-thirds of cases with add-on ezetimibe. Participants were randomized 2:1 to add-on alirocumab at 75 mg every 2 weeks or to placebo.
The alirocumab-treated patients had 58% and 51% reductions in LDL, compared to actively treated controls at 24 weeks in the FH I and FH II trials. Of the alirocumab-treated patients, 72% and 81% achieved their prespecified LDL goal at 24 weeks, compared with 2% and 11% of controls.
“We have never before seen these kinds of percentages of patients with familial hypercholesterolemia reaching these LDL levels,” commented Dr. Farnier of Point Medical in Dijon, France.
ODYSSEY COMBO II
At the same hot-line clinical trials session, Dr. Christopher P. Cannon reported that alirocumab markedly outperformed ezetimibe as add-on therapy in the 720-patient, phase III, double-blind ODYSSEY COMBO II trial.
In this study, patients at very high CV risk who were unable to reach their desired goal of an LDL below 70 mg/dL despite maximum tolerated statin doses were randomized 2:1 to alirocumab at 75 mg once every 2 weeks or oral ezetimibe (Zetia) at its approved dose of 10 mg/day as an active comparator. Each participant also received placebo therapy.
By week 24, patients on alirocumab plus high-dose statin averaged a 51% reduction in LDL compared to baseline, versus a 21% reduction with ezetimibe plus statin. These effects were maintained at 1 year, with no evidence of tolerance.
Of patients on alirocumab, 77% achieved an LDL goal of less than 70 mg/dL at week 24, compared with 45% on ezetimibe. In addition, 60% of the alirocumab group had an LDL below 50 mg/dL, as did 15% on ezetimibe.
The study design called for patients in the alirocumab group who still had an LDL above 70 mg/dL at week 12 to be uptitrated from 75 mg to 150 mg every 2 weeks. But only 20% of patients needed to do so, according to Dr. Cannon, professor of medicine at Harvard University, Boston.
Will high-risk patient adhere long-term to treatment by self-injection? Dr. Cannon thinks so. He noted that 85% of patients in ODYSSEY COMBO II remained adherent to the biweekly self-injection protocol through 1 year.
“That has been a very pleasant surprise,” the cardiologist said. “The notion of injections for cholesterol management is foreign. It was a big surprise to us that patients really did it.”
‘Great news’ from alirocumab
The alirocumab results in the heterozygous familial hypercholesterolemia trials are “great news for patients with this disease,” discussant Dr. Robert M. Califf said. He noted that recent estimates put the prevalence of heterozygous familial hypercholesterolemia at roughly 1 in 250 persons in the general population, making the genetic disorder considerably more common than most physicians realize.
“These are people who have a terrible disease where a massive reduction in LDL, it seems to me, is clearly worthwhile,” commented Dr. Califf, professor of medicine and vice chancellor for clinical and translational research at Duke University in Durham, N.C.
As for Dr. Robinson’s ODYSSEY LONG TERM data showing a 54% reduction in major CV events, he said “It’s alluring. It looks great. It looks fantastic. And it sets up the large events trial. The only caution out of all of this from my perspective is that any study with less than 100 events is something that should be regarded with great interest but is not definitive.”
If approved, alirocumab could see widespread use, especially if the ODYSSEY OUTCOMES results prove positive. After all, heterozygous familial hypercholesterolemia is now recognized to be one of the most common of all inherited diseases. And Dr. Cannon said that in clinical trials in post-acute coronary syndrome patients placed on high-dose statins, it’s common for only about 40% to achieve an LDL level below 70 mg/dL. In clinical practice, the rate is probably even lower.*
“Down the line I think this level of LDL lowering is needed very badly. To get high-risk patients who have high cholesterol despite maximally dosed statins or statin-intolerance down to an LDL level of 50 mg/dL would be a pretty good thing. This will be applicable to millions of patients,” he predicted.*
Dr. Robinson said that at the American Heart Association meeting this November she and her ODYSSEY LONG TERM coinvestigators plan to present subgroup analyses looking at alirocumab efficacy and safety in patients with achieved LDL levels below 25 and even 15 mg/dL.
Dr. Robinson, Dr. Cannon, and Dr. Farnier reported receiving research grants and consultants’ fees from Sanofi and Regeneron as well as other pharmaceutical companies. Dr. Califf reported having no financial conflicts.
bjancin@frontlinemedcom.com
*CORRECTION: An earlier version of this story misattributed these statements.
BARCELONA – When added to maximally tolerated statin therapy, the investigational PCSK9 inhibitor alirocumab resulted in a further 54% reduction in major cardiovascular events among high-cardiovascular-risk patients, based on a post-hoc analysis of a large randomized controlled Phase-3 trial.
The ODYSSEY LONG TERM trial is the largest and longest study of a PCSK9 (proprotein convertase subtilisin/kexin type 9) inhibitor to report results to date, with roughly 1,900 patient-years of double-blind exposure to alirocumab. And although the ongoing trial is primarily a safety study, it is also now the first PCSK9 trial to provide what everyone watching the development of this novel drug class has been eagerly awaiting: clinical outcomes data, albeit in this case from a post-hoc secondary analysis.
“This is the first trial with any of the PCSK9 inhibitors to suggest that there will be a further significant reduction in cardiovascular events when added on to maximized statin therapy,” Dr. Jennifer G. Robinson said in presenting interim results of ODYSSEY LONG TERM at the annual congress of the European Society of Cardiology.

“We’re on the right track in terms of trying to achieve further reduction in cardiovascular events through additional lipid lowering. But this is not the definitive evidence. We need the prospective outcomes trials to validate this data and also to establish the long-term safety of these drugs when added to the statins,” cautioned Dr. Robinson, professor of epidemiology and of medicine and director of the prevention intervention center at the University of Iowa, Iowa City.
Nonetheless, on the basis of the dramatic LDL-lowering and reassuring evidence of safety shown in ODYSSEY LONG TERM and the other double-blind phase III trials presented at the congress, Sanofi and Regeneron announced plans to file for U.S. and European Union marketing approval of alirocumab before the end of the year. The proposed indication will be for LDL lowering, which regulatory agencies have accepted as a surrogate endpoint for prevention of clinical events.
Meanwhile, the definitive ODYSSEY OUTCOMES trial is underway in 18,000 patients with acute coronary syndromes, with prospective evaluation of CV outcomes as its primary endpoint. The composite endpoint employed in the big OUTCOMES trial is identical to that used in the ODYSSEY LONG TERM post hoc analysis.
ODYSSEY LONG TERM includes 2,341 patients at high CV risk and an LDL level greater than 70 mg/dL despite maximally tolerated statin therapy. The patients fall into two categories: those with heterozygous familial hypercholesterolemia and others at very high risk because of known coronary heart disease. Participants were randomized 2:1 to 150 mg of alirocumab by self-administered subcutaneous injection at home every 2 weeks or placebo in addition to their statin.
In the interim post-hoc efficacy analysis at 65 weeks, the combined rate of cardiac death, nonfatal MI, stroke, and unstable angina requiring hospitalization was 1.4% in the alirocumab arm compared to 3.0% in placebo-treated controls, for a highly significant 54% relative risk reduction (see graphic).
At 24 weeks, the alirocumab group showed a mean 62% reduction in LDL compared to placebo, a difference that remained constant at 52 weeks. The average LDL level at 52 weeks in the alirocumab group was 53 mg/dL, down from 123 mg/dL on active treatment at baseline; 79% of alirocumab-treated patients achieved an LDL below 70 mg/dL.
The incidence and types of adverse events in the alirocumab arm were essentially the same as with placebo, with no signal of problems in any domains, including neurocognitive function or allergic reactions.
ODYSSEY FH I and FH II
In a separate presentation, Dr. Michel Farnier reported on the alirocumab experience in 735 patients with heterozygous familial hypercholesterolemia in two phase III trials known as ODYSSEY FH I and FH II. At baseline, all were above their LDL goal despite maximally tolerated statin therapy, in two-thirds of cases with add-on ezetimibe. Participants were randomized 2:1 to add-on alirocumab at 75 mg every 2 weeks or to placebo.
The alirocumab-treated patients had 58% and 51% reductions in LDL, compared to actively treated controls at 24 weeks in the FH I and FH II trials. Of the alirocumab-treated patients, 72% and 81% achieved their prespecified LDL goal at 24 weeks, compared with 2% and 11% of controls.
“We have never before seen these kinds of percentages of patients with familial hypercholesterolemia reaching these LDL levels,” commented Dr. Farnier of Point Medical in Dijon, France.
ODYSSEY COMBO II
At the same hot-line clinical trials session, Dr. Christopher P. Cannon reported that alirocumab markedly outperformed ezetimibe as add-on therapy in the 720-patient, phase III, double-blind ODYSSEY COMBO II trial.
In this study, patients at very high CV risk who were unable to reach their desired goal of an LDL below 70 mg/dL despite maximum tolerated statin doses were randomized 2:1 to alirocumab at 75 mg once every 2 weeks or oral ezetimibe (Zetia) at its approved dose of 10 mg/day as an active comparator. Each participant also received placebo therapy.
By week 24, patients on alirocumab plus high-dose statin averaged a 51% reduction in LDL compared to baseline, versus a 21% reduction with ezetimibe plus statin. These effects were maintained at 1 year, with no evidence of tolerance.
Of patients on alirocumab, 77% achieved an LDL goal of less than 70 mg/dL at week 24, compared with 45% on ezetimibe. In addition, 60% of the alirocumab group had an LDL below 50 mg/dL, as did 15% on ezetimibe.
The study design called for patients in the alirocumab group who still had an LDL above 70 mg/dL at week 12 to be uptitrated from 75 mg to 150 mg every 2 weeks. But only 20% of patients needed to do so, according to Dr. Cannon, professor of medicine at Harvard University, Boston.
Will high-risk patient adhere long-term to treatment by self-injection? Dr. Cannon thinks so. He noted that 85% of patients in ODYSSEY COMBO II remained adherent to the biweekly self-injection protocol through 1 year.
“That has been a very pleasant surprise,” the cardiologist said. “The notion of injections for cholesterol management is foreign. It was a big surprise to us that patients really did it.”
‘Great news’ from alirocumab
The alirocumab results in the heterozygous familial hypercholesterolemia trials are “great news for patients with this disease,” discussant Dr. Robert M. Califf said. He noted that recent estimates put the prevalence of heterozygous familial hypercholesterolemia at roughly 1 in 250 persons in the general population, making the genetic disorder considerably more common than most physicians realize.
“These are people who have a terrible disease where a massive reduction in LDL, it seems to me, is clearly worthwhile,” commented Dr. Califf, professor of medicine and vice chancellor for clinical and translational research at Duke University in Durham, N.C.
As for Dr. Robinson’s ODYSSEY LONG TERM data showing a 54% reduction in major CV events, he said “It’s alluring. It looks great. It looks fantastic. And it sets up the large events trial. The only caution out of all of this from my perspective is that any study with less than 100 events is something that should be regarded with great interest but is not definitive.”
If approved, alirocumab could see widespread use, especially if the ODYSSEY OUTCOMES results prove positive. After all, heterozygous familial hypercholesterolemia is now recognized to be one of the most common of all inherited diseases. And Dr. Cannon said that in clinical trials in post-acute coronary syndrome patients placed on high-dose statins, it’s common for only about 40% to achieve an LDL level below 70 mg/dL. In clinical practice, the rate is probably even lower.*
“Down the line I think this level of LDL lowering is needed very badly. To get high-risk patients who have high cholesterol despite maximally dosed statins or statin-intolerance down to an LDL level of 50 mg/dL would be a pretty good thing. This will be applicable to millions of patients,” he predicted.*
Dr. Robinson said that at the American Heart Association meeting this November she and her ODYSSEY LONG TERM coinvestigators plan to present subgroup analyses looking at alirocumab efficacy and safety in patients with achieved LDL levels below 25 and even 15 mg/dL.
Dr. Robinson, Dr. Cannon, and Dr. Farnier reported receiving research grants and consultants’ fees from Sanofi and Regeneron as well as other pharmaceutical companies. Dr. Califf reported having no financial conflicts.
bjancin@frontlinemedcom.com
*CORRECTION: An earlier version of this story misattributed these statements.
BARCELONA – When added to maximally tolerated statin therapy, the investigational PCSK9 inhibitor alirocumab resulted in a further 54% reduction in major cardiovascular events among high-cardiovascular-risk patients, based on a post-hoc analysis of a large randomized controlled Phase-3 trial.
The ODYSSEY LONG TERM trial is the largest and longest study of a PCSK9 (proprotein convertase subtilisin/kexin type 9) inhibitor to report results to date, with roughly 1,900 patient-years of double-blind exposure to alirocumab. And although the ongoing trial is primarily a safety study, it is also now the first PCSK9 trial to provide what everyone watching the development of this novel drug class has been eagerly awaiting: clinical outcomes data, albeit in this case from a post-hoc secondary analysis.
“This is the first trial with any of the PCSK9 inhibitors to suggest that there will be a further significant reduction in cardiovascular events when added on to maximized statin therapy,” Dr. Jennifer G. Robinson said in presenting interim results of ODYSSEY LONG TERM at the annual congress of the European Society of Cardiology.

“We’re on the right track in terms of trying to achieve further reduction in cardiovascular events through additional lipid lowering. But this is not the definitive evidence. We need the prospective outcomes trials to validate this data and also to establish the long-term safety of these drugs when added to the statins,” cautioned Dr. Robinson, professor of epidemiology and of medicine and director of the prevention intervention center at the University of Iowa, Iowa City.
Nonetheless, on the basis of the dramatic LDL-lowering and reassuring evidence of safety shown in ODYSSEY LONG TERM and the other double-blind phase III trials presented at the congress, Sanofi and Regeneron announced plans to file for U.S. and European Union marketing approval of alirocumab before the end of the year. The proposed indication will be for LDL lowering, which regulatory agencies have accepted as a surrogate endpoint for prevention of clinical events.
Meanwhile, the definitive ODYSSEY OUTCOMES trial is underway in 18,000 patients with acute coronary syndromes, with prospective evaluation of CV outcomes as its primary endpoint. The composite endpoint employed in the big OUTCOMES trial is identical to that used in the ODYSSEY LONG TERM post hoc analysis.
ODYSSEY LONG TERM includes 2,341 patients at high CV risk and an LDL level greater than 70 mg/dL despite maximally tolerated statin therapy. The patients fall into two categories: those with heterozygous familial hypercholesterolemia and others at very high risk because of known coronary heart disease. Participants were randomized 2:1 to 150 mg of alirocumab by self-administered subcutaneous injection at home every 2 weeks or placebo in addition to their statin.
In the interim post-hoc efficacy analysis at 65 weeks, the combined rate of cardiac death, nonfatal MI, stroke, and unstable angina requiring hospitalization was 1.4% in the alirocumab arm compared to 3.0% in placebo-treated controls, for a highly significant 54% relative risk reduction (see graphic).
At 24 weeks, the alirocumab group showed a mean 62% reduction in LDL compared to placebo, a difference that remained constant at 52 weeks. The average LDL level at 52 weeks in the alirocumab group was 53 mg/dL, down from 123 mg/dL on active treatment at baseline; 79% of alirocumab-treated patients achieved an LDL below 70 mg/dL.
The incidence and types of adverse events in the alirocumab arm were essentially the same as with placebo, with no signal of problems in any domains, including neurocognitive function or allergic reactions.
ODYSSEY FH I and FH II
In a separate presentation, Dr. Michel Farnier reported on the alirocumab experience in 735 patients with heterozygous familial hypercholesterolemia in two phase III trials known as ODYSSEY FH I and FH II. At baseline, all were above their LDL goal despite maximally tolerated statin therapy, in two-thirds of cases with add-on ezetimibe. Participants were randomized 2:1 to add-on alirocumab at 75 mg every 2 weeks or to placebo.
The alirocumab-treated patients had 58% and 51% reductions in LDL, compared to actively treated controls at 24 weeks in the FH I and FH II trials. Of the alirocumab-treated patients, 72% and 81% achieved their prespecified LDL goal at 24 weeks, compared with 2% and 11% of controls.
“We have never before seen these kinds of percentages of patients with familial hypercholesterolemia reaching these LDL levels,” commented Dr. Farnier of Point Medical in Dijon, France.
ODYSSEY COMBO II
At the same hot-line clinical trials session, Dr. Christopher P. Cannon reported that alirocumab markedly outperformed ezetimibe as add-on therapy in the 720-patient, phase III, double-blind ODYSSEY COMBO II trial.
In this study, patients at very high CV risk who were unable to reach their desired goal of an LDL below 70 mg/dL despite maximum tolerated statin doses were randomized 2:1 to alirocumab at 75 mg once every 2 weeks or oral ezetimibe (Zetia) at its approved dose of 10 mg/day as an active comparator. Each participant also received placebo therapy.
By week 24, patients on alirocumab plus high-dose statin averaged a 51% reduction in LDL compared to baseline, versus a 21% reduction with ezetimibe plus statin. These effects were maintained at 1 year, with no evidence of tolerance.
Of patients on alirocumab, 77% achieved an LDL goal of less than 70 mg/dL at week 24, compared with 45% on ezetimibe. In addition, 60% of the alirocumab group had an LDL below 50 mg/dL, as did 15% on ezetimibe.
The study design called for patients in the alirocumab group who still had an LDL above 70 mg/dL at week 12 to be uptitrated from 75 mg to 150 mg every 2 weeks. But only 20% of patients needed to do so, according to Dr. Cannon, professor of medicine at Harvard University, Boston.
Will high-risk patient adhere long-term to treatment by self-injection? Dr. Cannon thinks so. He noted that 85% of patients in ODYSSEY COMBO II remained adherent to the biweekly self-injection protocol through 1 year.
“That has been a very pleasant surprise,” the cardiologist said. “The notion of injections for cholesterol management is foreign. It was a big surprise to us that patients really did it.”
‘Great news’ from alirocumab
The alirocumab results in the heterozygous familial hypercholesterolemia trials are “great news for patients with this disease,” discussant Dr. Robert M. Califf said. He noted that recent estimates put the prevalence of heterozygous familial hypercholesterolemia at roughly 1 in 250 persons in the general population, making the genetic disorder considerably more common than most physicians realize.
“These are people who have a terrible disease where a massive reduction in LDL, it seems to me, is clearly worthwhile,” commented Dr. Califf, professor of medicine and vice chancellor for clinical and translational research at Duke University in Durham, N.C.
As for Dr. Robinson’s ODYSSEY LONG TERM data showing a 54% reduction in major CV events, he said “It’s alluring. It looks great. It looks fantastic. And it sets up the large events trial. The only caution out of all of this from my perspective is that any study with less than 100 events is something that should be regarded with great interest but is not definitive.”
If approved, alirocumab could see widespread use, especially if the ODYSSEY OUTCOMES results prove positive. After all, heterozygous familial hypercholesterolemia is now recognized to be one of the most common of all inherited diseases. And Dr. Cannon said that in clinical trials in post-acute coronary syndrome patients placed on high-dose statins, it’s common for only about 40% to achieve an LDL level below 70 mg/dL. In clinical practice, the rate is probably even lower.*
“Down the line I think this level of LDL lowering is needed very badly. To get high-risk patients who have high cholesterol despite maximally dosed statins or statin-intolerance down to an LDL level of 50 mg/dL would be a pretty good thing. This will be applicable to millions of patients,” he predicted.*
Dr. Robinson said that at the American Heart Association meeting this November she and her ODYSSEY LONG TERM coinvestigators plan to present subgroup analyses looking at alirocumab efficacy and safety in patients with achieved LDL levels below 25 and even 15 mg/dL.
Dr. Robinson, Dr. Cannon, and Dr. Farnier reported receiving research grants and consultants’ fees from Sanofi and Regeneron as well as other pharmaceutical companies. Dr. Califf reported having no financial conflicts.
bjancin@frontlinemedcom.com
*CORRECTION: An earlier version of this story misattributed these statements.
AT THE ESC CONGRESS 2014
Key clinical point: The lower-is-better approach to LDL reduction gets a big boost from a study that showed adding alirocumab to maximally tolerated statin therapy was associated with 54% fewer major adverse cardiovascular events compared to statin plus placebo in high-cardiovascular-risk patients.
Major finding: The incidence of the composite cardiovascular event endpoint at 65 weeks was 1.4% in those on alirocumab plus maximally tolerated statin therapy and 3.0% in those on placebo plus statin.
Data source: This was an interim post hoc analysis from the Phase-3, double-blind, randomized, prospective ODYSSEY LONG TERM trial of more than 2,300 randomized patients.
Disclosures: The ODYSSEY clinical trials program is sponsored by Sanofi and Regeneron. The study presenters reported receiving research grants and consultants’ fees from those and other companies.