User login
ESC issues cascade of new cardiology practice guidelines
BARCELONA – Routine use of a new risk calculator tool to estimate the 5-year risk of sudden cardiac death is recommended in all patients with hypertrophic cardiomyopathy in updated guidelines launched at the annual congress of the European Society of Cardiology.
“Based on that estimate, you can now have an intelligent conversation with the patient about what the threshold is for implantable cardioverter-defibrillator placement given an individual’s risk/benefit ratio,” explained Dr. Perry M. Elliott, chair of the guidelines task force and professor of inherited cardiovascular disease at The Heart Hospital, London.
The hypertrophic cardiomyopathy guidelines were among five new practice guidelines introduced at the annual congress. Others addressed acute pulmonary embolism, noncardiac surgery, myocardial revascularization, and aortic diseases.
Here are selected highlights of the new guidelines:
• Hypertrophic cardiomyopathy. The novel sudden cardiac death risk prognostication tool, known as HCM Risk-SCD, is a major innovation in the new guidelines. The risk calculator grew out of a recent multicenter study of nearly 3,700 patients conducted by Dr. Elliott and his coinvestigators (Eur. Heart J. 2014;35:2010-20).
Now physicians can plug a series of validated risk factors into an online calculator and receive an estimated 5-year risk figure. The input factors include maximal wall thickness, left atrial diameter, family history of sudden cardiac death, unexplained syncope, and maximal left ventricular outflow gradient. If the estimated 5-year risk is 6% or more and the patient has a life expectancy of more than a year, implantation of a cardioverter-defibrillator should be seriously considered, according to the guidelines.
“What we’re trying to do in these guidelines is to change a mind-set, because hypertrophic cardiomyopathy is not really a diagnosis; it actually represents a family of diseases. Running throughout this entire document is an emphasis on individualization, from diagnosis all the way through treatment,” Dr. Elliott said. “There’s a strong emphasis on making a specific diagnosis if you possibly can, because these subtypes of cardiomyopathy have totally different natural histories and in the future will have very different treatments.”
Other highlights of the new guidelines include a stepwise approach to management of left ventricular outflow tract obstruction and heart failure, advice on reproduction, suggestions regarding simple laboratory tests with diagnostic utility, and guidance on the effective utilization of ECG, echocardiography, and cardiac magnetic resonance imaging.
“We show a number of echocardiographic red flags, some of which are perhaps not readily appreciated in everyday practice,” Dr. Elliott said. “For example, the presence of hypertrophy with impaired systolic function immediately narrows down your diagnosis to one of five or six different conditions.”
• Pulmonary embolism. The new guidelines place the novel oral anticoagulants rivaroxaban (Xarelto), apixaban (Eliquis), dabigatran (Pradaxa), and edoxaban (Savaysa) on equal footing with once-standard warfarin.
Task force chair Dr. Stavros Konstantinides highlighted as new in the 2014 guidelines a strong recommendation that patients who do not have a high-risk pulmonary embolism – that is, those who are not in shock or hypotensive – should be scored using the Pulmonary Embolism Severity Index or another validated clinical risk prediction score. The purpose is to distinguish between those at low versus intermediate risk.
Those at intermediate risk should undergo right ventricular imaging via CT or echocardiography, along with a biomarker test for myocardial injury. If both the imaging study and biomarker are positive, the patient is classified as being at intermediate-high risk; if not, intermediate-low. This has treatment implications, since it’s recommended that systemic thrombolysis be considered only in those with intermediate-high-risk pulmonary embolism and clinical signs of hemodynamic decompensation, explained Dr. Konstantinides of University Medical Center in Mainz (Germany).
• Noncardiac surgery. Perioperative initiation of beta-blocker therapy is no longer recommended routinely, but may be considered in patients scheduled for high-risk noncardiac surgery who also have two or more cardiovascular risk factors or known ischemic heart disease. Atenolol and bisoprolol are recommended as the perioperative beta-blockers of choice. If a patient is already on a beta-blocker prior to surgery, the drug can be continued perioperatively, according to task force cochair Dr. Juhani Knuuti of the University of Turku (Finland).
The guidelines emphasize that prophylactic coronary revascularization is seldom indicated in patients undergoing noncardiac surgery that has a low or intermediate cardiovascular risk.
• Myocardial revascularization. These guidelines, with more than 300 recommendations, are the lengthiest of the five new sets of guidelines. The myocardial revascularization guidelines rely heavily upon the findings of a recent meta-analysis conducted by task force cochairs Dr. Stephan Windecker of Bern (Switzerland) University and Dr. Philippe Kohl of the University of Liege (Belgium) and their coinvestigators. The meta-analysis encompassed 100 randomized controlled trials with nearly 94,000 randomized patients and more than 262,000 patient-years of follow-u p (BMJ 2014 June 23 [doi: 10.1136.bmj.g3859]).
The guidelines introduce substantial changes in the recommended method of revascularization in various situations. There is a strong emphasis on risk stratification using the SYNTAX score; in fact, the guidelines include a primer on how to calculate it.
Based largely on the results of the meta-analysis as well as 5-year follow-up in the SYNTAX trial (Lancet 2013;381:629-38), the guidelines now regard percutaneous coronary intervention as equivalent to coronary artery bypass graft surgery in several patient subsets where CABG was previously preferred. These include patients with left main or triple-vessel disease and a SYNTAX score below 22. In patients with one- or two-vessel disease and proximal left anterior descending coronary artery stenosis, both PCI and CABG get a Class I recommendation.
The new guidelines emphasize the value of intracoronary fractional flow reserve measurement to identify hemodynamically relevant lesions warranting revascularization in patients with stable coronary artery disease lacking noninvasive evidence of ischemia.
• Aortic diseases. Routine screening for abdominal aortic aneurysm via ultrasound is recommended in all men over age 65 and “may be considered” in women over 65 with a history of smoking, according to the new guidelines. That’s a much stronger proscreening stance than has been taken by the U.S. Preventive Services Task Force.
Former ESC guidelines were confined to aortic dissection and focused on the thoracic aorta. The new guidelines expand in scope to include intramural hematomas, thoracic and abdominal aneurysms, aortic valve lesions, and penetrating ulcers.
“We are not only dealing with the thoracic aorta, but are taking the holistic view of the aorta as one organ,” according to guidelines task force cochair Dr. Raimund Erbel, professor of medicine at the University of Essen (Germany).
“When an aortic aneurysm is identified at any location, assessment of the entire aorta and aortic valve is recommended at baseline and during follow-up,” he added.
All of the new guidelines can be downloaded in their entirely at the ESC website (escardio.org/guidelines).
BARCELONA – Routine use of a new risk calculator tool to estimate the 5-year risk of sudden cardiac death is recommended in all patients with hypertrophic cardiomyopathy in updated guidelines launched at the annual congress of the European Society of Cardiology.
“Based on that estimate, you can now have an intelligent conversation with the patient about what the threshold is for implantable cardioverter-defibrillator placement given an individual’s risk/benefit ratio,” explained Dr. Perry M. Elliott, chair of the guidelines task force and professor of inherited cardiovascular disease at The Heart Hospital, London.
The hypertrophic cardiomyopathy guidelines were among five new practice guidelines introduced at the annual congress. Others addressed acute pulmonary embolism, noncardiac surgery, myocardial revascularization, and aortic diseases.
Here are selected highlights of the new guidelines:
• Hypertrophic cardiomyopathy. The novel sudden cardiac death risk prognostication tool, known as HCM Risk-SCD, is a major innovation in the new guidelines. The risk calculator grew out of a recent multicenter study of nearly 3,700 patients conducted by Dr. Elliott and his coinvestigators (Eur. Heart J. 2014;35:2010-20).
Now physicians can plug a series of validated risk factors into an online calculator and receive an estimated 5-year risk figure. The input factors include maximal wall thickness, left atrial diameter, family history of sudden cardiac death, unexplained syncope, and maximal left ventricular outflow gradient. If the estimated 5-year risk is 6% or more and the patient has a life expectancy of more than a year, implantation of a cardioverter-defibrillator should be seriously considered, according to the guidelines.
“What we’re trying to do in these guidelines is to change a mind-set, because hypertrophic cardiomyopathy is not really a diagnosis; it actually represents a family of diseases. Running throughout this entire document is an emphasis on individualization, from diagnosis all the way through treatment,” Dr. Elliott said. “There’s a strong emphasis on making a specific diagnosis if you possibly can, because these subtypes of cardiomyopathy have totally different natural histories and in the future will have very different treatments.”
Other highlights of the new guidelines include a stepwise approach to management of left ventricular outflow tract obstruction and heart failure, advice on reproduction, suggestions regarding simple laboratory tests with diagnostic utility, and guidance on the effective utilization of ECG, echocardiography, and cardiac magnetic resonance imaging.
“We show a number of echocardiographic red flags, some of which are perhaps not readily appreciated in everyday practice,” Dr. Elliott said. “For example, the presence of hypertrophy with impaired systolic function immediately narrows down your diagnosis to one of five or six different conditions.”
• Pulmonary embolism. The new guidelines place the novel oral anticoagulants rivaroxaban (Xarelto), apixaban (Eliquis), dabigatran (Pradaxa), and edoxaban (Savaysa) on equal footing with once-standard warfarin.
Task force chair Dr. Stavros Konstantinides highlighted as new in the 2014 guidelines a strong recommendation that patients who do not have a high-risk pulmonary embolism – that is, those who are not in shock or hypotensive – should be scored using the Pulmonary Embolism Severity Index or another validated clinical risk prediction score. The purpose is to distinguish between those at low versus intermediate risk.
Those at intermediate risk should undergo right ventricular imaging via CT or echocardiography, along with a biomarker test for myocardial injury. If both the imaging study and biomarker are positive, the patient is classified as being at intermediate-high risk; if not, intermediate-low. This has treatment implications, since it’s recommended that systemic thrombolysis be considered only in those with intermediate-high-risk pulmonary embolism and clinical signs of hemodynamic decompensation, explained Dr. Konstantinides of University Medical Center in Mainz (Germany).
• Noncardiac surgery. Perioperative initiation of beta-blocker therapy is no longer recommended routinely, but may be considered in patients scheduled for high-risk noncardiac surgery who also have two or more cardiovascular risk factors or known ischemic heart disease. Atenolol and bisoprolol are recommended as the perioperative beta-blockers of choice. If a patient is already on a beta-blocker prior to surgery, the drug can be continued perioperatively, according to task force cochair Dr. Juhani Knuuti of the University of Turku (Finland).
The guidelines emphasize that prophylactic coronary revascularization is seldom indicated in patients undergoing noncardiac surgery that has a low or intermediate cardiovascular risk.
• Myocardial revascularization. These guidelines, with more than 300 recommendations, are the lengthiest of the five new sets of guidelines. The myocardial revascularization guidelines rely heavily upon the findings of a recent meta-analysis conducted by task force cochairs Dr. Stephan Windecker of Bern (Switzerland) University and Dr. Philippe Kohl of the University of Liege (Belgium) and their coinvestigators. The meta-analysis encompassed 100 randomized controlled trials with nearly 94,000 randomized patients and more than 262,000 patient-years of follow-u p (BMJ 2014 June 23 [doi: 10.1136.bmj.g3859]).
The guidelines introduce substantial changes in the recommended method of revascularization in various situations. There is a strong emphasis on risk stratification using the SYNTAX score; in fact, the guidelines include a primer on how to calculate it.
Based largely on the results of the meta-analysis as well as 5-year follow-up in the SYNTAX trial (Lancet 2013;381:629-38), the guidelines now regard percutaneous coronary intervention as equivalent to coronary artery bypass graft surgery in several patient subsets where CABG was previously preferred. These include patients with left main or triple-vessel disease and a SYNTAX score below 22. In patients with one- or two-vessel disease and proximal left anterior descending coronary artery stenosis, both PCI and CABG get a Class I recommendation.
The new guidelines emphasize the value of intracoronary fractional flow reserve measurement to identify hemodynamically relevant lesions warranting revascularization in patients with stable coronary artery disease lacking noninvasive evidence of ischemia.
• Aortic diseases. Routine screening for abdominal aortic aneurysm via ultrasound is recommended in all men over age 65 and “may be considered” in women over 65 with a history of smoking, according to the new guidelines. That’s a much stronger proscreening stance than has been taken by the U.S. Preventive Services Task Force.
Former ESC guidelines were confined to aortic dissection and focused on the thoracic aorta. The new guidelines expand in scope to include intramural hematomas, thoracic and abdominal aneurysms, aortic valve lesions, and penetrating ulcers.
“We are not only dealing with the thoracic aorta, but are taking the holistic view of the aorta as one organ,” according to guidelines task force cochair Dr. Raimund Erbel, professor of medicine at the University of Essen (Germany).
“When an aortic aneurysm is identified at any location, assessment of the entire aorta and aortic valve is recommended at baseline and during follow-up,” he added.
All of the new guidelines can be downloaded in their entirely at the ESC website (escardio.org/guidelines).
BARCELONA – Routine use of a new risk calculator tool to estimate the 5-year risk of sudden cardiac death is recommended in all patients with hypertrophic cardiomyopathy in updated guidelines launched at the annual congress of the European Society of Cardiology.
“Based on that estimate, you can now have an intelligent conversation with the patient about what the threshold is for implantable cardioverter-defibrillator placement given an individual’s risk/benefit ratio,” explained Dr. Perry M. Elliott, chair of the guidelines task force and professor of inherited cardiovascular disease at The Heart Hospital, London.
The hypertrophic cardiomyopathy guidelines were among five new practice guidelines introduced at the annual congress. Others addressed acute pulmonary embolism, noncardiac surgery, myocardial revascularization, and aortic diseases.
Here are selected highlights of the new guidelines:
• Hypertrophic cardiomyopathy. The novel sudden cardiac death risk prognostication tool, known as HCM Risk-SCD, is a major innovation in the new guidelines. The risk calculator grew out of a recent multicenter study of nearly 3,700 patients conducted by Dr. Elliott and his coinvestigators (Eur. Heart J. 2014;35:2010-20).
Now physicians can plug a series of validated risk factors into an online calculator and receive an estimated 5-year risk figure. The input factors include maximal wall thickness, left atrial diameter, family history of sudden cardiac death, unexplained syncope, and maximal left ventricular outflow gradient. If the estimated 5-year risk is 6% or more and the patient has a life expectancy of more than a year, implantation of a cardioverter-defibrillator should be seriously considered, according to the guidelines.
“What we’re trying to do in these guidelines is to change a mind-set, because hypertrophic cardiomyopathy is not really a diagnosis; it actually represents a family of diseases. Running throughout this entire document is an emphasis on individualization, from diagnosis all the way through treatment,” Dr. Elliott said. “There’s a strong emphasis on making a specific diagnosis if you possibly can, because these subtypes of cardiomyopathy have totally different natural histories and in the future will have very different treatments.”
Other highlights of the new guidelines include a stepwise approach to management of left ventricular outflow tract obstruction and heart failure, advice on reproduction, suggestions regarding simple laboratory tests with diagnostic utility, and guidance on the effective utilization of ECG, echocardiography, and cardiac magnetic resonance imaging.
“We show a number of echocardiographic red flags, some of which are perhaps not readily appreciated in everyday practice,” Dr. Elliott said. “For example, the presence of hypertrophy with impaired systolic function immediately narrows down your diagnosis to one of five or six different conditions.”
• Pulmonary embolism. The new guidelines place the novel oral anticoagulants rivaroxaban (Xarelto), apixaban (Eliquis), dabigatran (Pradaxa), and edoxaban (Savaysa) on equal footing with once-standard warfarin.
Task force chair Dr. Stavros Konstantinides highlighted as new in the 2014 guidelines a strong recommendation that patients who do not have a high-risk pulmonary embolism – that is, those who are not in shock or hypotensive – should be scored using the Pulmonary Embolism Severity Index or another validated clinical risk prediction score. The purpose is to distinguish between those at low versus intermediate risk.
Those at intermediate risk should undergo right ventricular imaging via CT or echocardiography, along with a biomarker test for myocardial injury. If both the imaging study and biomarker are positive, the patient is classified as being at intermediate-high risk; if not, intermediate-low. This has treatment implications, since it’s recommended that systemic thrombolysis be considered only in those with intermediate-high-risk pulmonary embolism and clinical signs of hemodynamic decompensation, explained Dr. Konstantinides of University Medical Center in Mainz (Germany).
• Noncardiac surgery. Perioperative initiation of beta-blocker therapy is no longer recommended routinely, but may be considered in patients scheduled for high-risk noncardiac surgery who also have two or more cardiovascular risk factors or known ischemic heart disease. Atenolol and bisoprolol are recommended as the perioperative beta-blockers of choice. If a patient is already on a beta-blocker prior to surgery, the drug can be continued perioperatively, according to task force cochair Dr. Juhani Knuuti of the University of Turku (Finland).
The guidelines emphasize that prophylactic coronary revascularization is seldom indicated in patients undergoing noncardiac surgery that has a low or intermediate cardiovascular risk.
• Myocardial revascularization. These guidelines, with more than 300 recommendations, are the lengthiest of the five new sets of guidelines. The myocardial revascularization guidelines rely heavily upon the findings of a recent meta-analysis conducted by task force cochairs Dr. Stephan Windecker of Bern (Switzerland) University and Dr. Philippe Kohl of the University of Liege (Belgium) and their coinvestigators. The meta-analysis encompassed 100 randomized controlled trials with nearly 94,000 randomized patients and more than 262,000 patient-years of follow-u p (BMJ 2014 June 23 [doi: 10.1136.bmj.g3859]).
The guidelines introduce substantial changes in the recommended method of revascularization in various situations. There is a strong emphasis on risk stratification using the SYNTAX score; in fact, the guidelines include a primer on how to calculate it.
Based largely on the results of the meta-analysis as well as 5-year follow-up in the SYNTAX trial (Lancet 2013;381:629-38), the guidelines now regard percutaneous coronary intervention as equivalent to coronary artery bypass graft surgery in several patient subsets where CABG was previously preferred. These include patients with left main or triple-vessel disease and a SYNTAX score below 22. In patients with one- or two-vessel disease and proximal left anterior descending coronary artery stenosis, both PCI and CABG get a Class I recommendation.
The new guidelines emphasize the value of intracoronary fractional flow reserve measurement to identify hemodynamically relevant lesions warranting revascularization in patients with stable coronary artery disease lacking noninvasive evidence of ischemia.
• Aortic diseases. Routine screening for abdominal aortic aneurysm via ultrasound is recommended in all men over age 65 and “may be considered” in women over 65 with a history of smoking, according to the new guidelines. That’s a much stronger proscreening stance than has been taken by the U.S. Preventive Services Task Force.
Former ESC guidelines were confined to aortic dissection and focused on the thoracic aorta. The new guidelines expand in scope to include intramural hematomas, thoracic and abdominal aneurysms, aortic valve lesions, and penetrating ulcers.
“We are not only dealing with the thoracic aorta, but are taking the holistic view of the aorta as one organ,” according to guidelines task force cochair Dr. Raimund Erbel, professor of medicine at the University of Essen (Germany).
“When an aortic aneurysm is identified at any location, assessment of the entire aorta and aortic valve is recommended at baseline and during follow-up,” he added.
All of the new guidelines can be downloaded in their entirely at the ESC website (escardio.org/guidelines).
AT THE ESC CONGRESS 2014
Diabetes Increases Risk for Atrial Fibrillation
BARCELONA – Adults with diabetes mellitus are at increased risk of subsequent new-onset atrial fibrillation – and the younger the age at diabetes onset, the greater the likelihood of developing the arrhythmia.
That’s the key finding from a Danish national registry study in which all 5,168,416 Danish adults without atrial fibrillation in 1996 were followed through 2012 for development of atrial fibrillation (AF). The study population included 75,197 Danes with diabetes at baseline and another 235,327 who developed the disease during follow-up, Dr. Jannik L. Pallisgaard explained at the annual congress of the European Society of Cardiology.
During follow-up, 5.6% of those with diabetes and 3.3% of those without diabetes developed AF. The mean time from diabetes onset to AF onset was 5 years, reported Dr. Pallisgaard of the University of Copenhagen.
"What was particularly interesting, I think, is that we found the youngest patients were the group at highest risk" of developing AF, he said. "We suggest that starting at the onset of diabetes, routine pulse palpation, ECGs, and focused patient interviews asking about any signs of atrial fibrillation could prove beneficial in detecting the arrhythmia."
The incidence rate ratio for developing AF per 1,000 person-years of follow-up was roughly 2.5-fold greater in 18- to 39-year-olds with diabetes than in their nondiabetic peers. From this peak rate in young adults, the magnitude of relative risk dropped in stepwise fashion with age: The variability in risk was lower in 40- to 60-year-old diabetics than in the 18- to 39-year olds and lower still in 65- to 74-year olds. Variability in the incidence rate ratio finally bottomed out at a still statistically significant 1.3-fold increased risk of developing AF in diabetic individuals ages 75 and older compared to their nondiabetic peers.
Dr. Pallisgaard noted that while the relative risk of developing AF was greatest in the 18- to 39-year-olds, the absolute number of new cases of AF was far greater in older patients because there were so many more of them with diabetes. He cautioned that as the obesity epidemic leads to more and more patients developing type 2 diabetes at younger ages, more cases of AF can be expected in young adults.
Dr. Pallisgaard cited two likely mechanisms underlying the observed increased risk of AF in diabetic patients: left ventricular hypertrophy and vascular inflammation, which are both often present in the diabetic population.
He reported having no financial conflicts regarding this study, conducted with Danish institutional research funds.
BARCELONA – Adults with diabetes mellitus are at increased risk of subsequent new-onset atrial fibrillation – and the younger the age at diabetes onset, the greater the likelihood of developing the arrhythmia.
That’s the key finding from a Danish national registry study in which all 5,168,416 Danish adults without atrial fibrillation in 1996 were followed through 2012 for development of atrial fibrillation (AF). The study population included 75,197 Danes with diabetes at baseline and another 235,327 who developed the disease during follow-up, Dr. Jannik L. Pallisgaard explained at the annual congress of the European Society of Cardiology.
During follow-up, 5.6% of those with diabetes and 3.3% of those without diabetes developed AF. The mean time from diabetes onset to AF onset was 5 years, reported Dr. Pallisgaard of the University of Copenhagen.
"What was particularly interesting, I think, is that we found the youngest patients were the group at highest risk" of developing AF, he said. "We suggest that starting at the onset of diabetes, routine pulse palpation, ECGs, and focused patient interviews asking about any signs of atrial fibrillation could prove beneficial in detecting the arrhythmia."
The incidence rate ratio for developing AF per 1,000 person-years of follow-up was roughly 2.5-fold greater in 18- to 39-year-olds with diabetes than in their nondiabetic peers. From this peak rate in young adults, the magnitude of relative risk dropped in stepwise fashion with age: The variability in risk was lower in 40- to 60-year-old diabetics than in the 18- to 39-year olds and lower still in 65- to 74-year olds. Variability in the incidence rate ratio finally bottomed out at a still statistically significant 1.3-fold increased risk of developing AF in diabetic individuals ages 75 and older compared to their nondiabetic peers.
Dr. Pallisgaard noted that while the relative risk of developing AF was greatest in the 18- to 39-year-olds, the absolute number of new cases of AF was far greater in older patients because there were so many more of them with diabetes. He cautioned that as the obesity epidemic leads to more and more patients developing type 2 diabetes at younger ages, more cases of AF can be expected in young adults.
Dr. Pallisgaard cited two likely mechanisms underlying the observed increased risk of AF in diabetic patients: left ventricular hypertrophy and vascular inflammation, which are both often present in the diabetic population.
He reported having no financial conflicts regarding this study, conducted with Danish institutional research funds.
BARCELONA – Adults with diabetes mellitus are at increased risk of subsequent new-onset atrial fibrillation – and the younger the age at diabetes onset, the greater the likelihood of developing the arrhythmia.
That’s the key finding from a Danish national registry study in which all 5,168,416 Danish adults without atrial fibrillation in 1996 were followed through 2012 for development of atrial fibrillation (AF). The study population included 75,197 Danes with diabetes at baseline and another 235,327 who developed the disease during follow-up, Dr. Jannik L. Pallisgaard explained at the annual congress of the European Society of Cardiology.
During follow-up, 5.6% of those with diabetes and 3.3% of those without diabetes developed AF. The mean time from diabetes onset to AF onset was 5 years, reported Dr. Pallisgaard of the University of Copenhagen.
"What was particularly interesting, I think, is that we found the youngest patients were the group at highest risk" of developing AF, he said. "We suggest that starting at the onset of diabetes, routine pulse palpation, ECGs, and focused patient interviews asking about any signs of atrial fibrillation could prove beneficial in detecting the arrhythmia."
The incidence rate ratio for developing AF per 1,000 person-years of follow-up was roughly 2.5-fold greater in 18- to 39-year-olds with diabetes than in their nondiabetic peers. From this peak rate in young adults, the magnitude of relative risk dropped in stepwise fashion with age: The variability in risk was lower in 40- to 60-year-old diabetics than in the 18- to 39-year olds and lower still in 65- to 74-year olds. Variability in the incidence rate ratio finally bottomed out at a still statistically significant 1.3-fold increased risk of developing AF in diabetic individuals ages 75 and older compared to their nondiabetic peers.
Dr. Pallisgaard noted that while the relative risk of developing AF was greatest in the 18- to 39-year-olds, the absolute number of new cases of AF was far greater in older patients because there were so many more of them with diabetes. He cautioned that as the obesity epidemic leads to more and more patients developing type 2 diabetes at younger ages, more cases of AF can be expected in young adults.
Dr. Pallisgaard cited two likely mechanisms underlying the observed increased risk of AF in diabetic patients: left ventricular hypertrophy and vascular inflammation, which are both often present in the diabetic population.
He reported having no financial conflicts regarding this study, conducted with Danish institutional research funds.
AT THE ESC CONGRESS 2014
Short-course amiodarone post AF ablation cuts hospitalizations
BARCELONA – Eight weeks of oral amiodarone beginning right after catheter ablation for atrial fibrillation more than halved the rates of cardioversion and arrhythmia-related hospitalization for 3 months in the AMIO-CAT trial.
This was a consequence of the sharp reduction in the incidence of AF lasting more than 30 seconds during that interval: 34% in the amiodarone group, compared with 53% in placebo-treated controls, Dr. Stine Darkner reported at the annual congress of the European Society of Cardiology.
“Patients undergoing catheter ablation for AF quite often experience arrhythmias within the first few months after ablation. The ablation itself is thought to cause some of these arrhythmias. It is quite a distressing experience for the patient,” observed Dr. Darkner of Copenhagen University Hospital.
While the benefits at 3 months were significant for the study population as a whole, upon closer analysis the benefits reached statistical significance only in patients treated for persistent AF. Although trends were favorable in subjects with paroxysmal AF, the benefits were less pronounced.
“The implication is that short-term amiodarone should perhaps not be standard treatment after ablation, but should be considered in patients with persistent AF,” she concluded.
At 6 months, there was no significant difference in the cumulative AF recurrence rate, which was the prespecified primary outcome. The rates were 52% in the amiodarone group and 61% with placebo, according to Dr. Darkner.
AMIO-CAT (Amiodarone After Catheter Ablation for Atrial Fibrillation) was the first double-blind, placebo-controlled clinical trial to evaluate the impact of a short course of amiodarone on recovery after AF ablation. The study comprised 212 patients undergoing radiofrequency ablation for paroxysmal or persistent AF.
During the first 3 months post ablation, a time that electrophysiologists call “the blanking period,” the amiodarone group experienced a 57% lower risk of atrial tachyarrythmia-related hospitalization and a 64% reduction in cardioversions, compared with controls.
The side effects reported by patients on amiodarone were transient and are familiar in nature to physicians who prescribe the antiarrhythmic agent: sleep disruption, GI complaints, photosensitivity, and asymptomatic changes in thyroid hormone levels. None of the side effects was serious. Quality of life as measured by the SF-36 was not reduced in the amiodarone group, despite the increase in side effects.
The dosing regimen in the study was amiodarone at 400 mg on the night of the ablation, then 400 mg twice daily for 13 days, followed by amiodarone at 200 mg twice daily for 2 weeks, and finally 200 mg once daily for 4 weeks.
Discussant Karl-Heinz Kuck said he wasn’t surprised by the lack of benefit at 6 months in AMIO-CAT.
“We know that in patients with paroxysmal or persistent AF, the predominant cause of atrial arrhythmia recurrence post ablation at any point in time is pulmonary vein reconduction, which is unlikely to be altered by 8 weeks of amiodarone, especially after discontinuing the drug. We know that AF begets AF, but we don’t know that sinus rhythm begets sinus rhythm,” said Dr. Kuck, head of cardiology at St. Georg Hospital in Hamburg, Germany.
A more promising strategy to reduce the long-term AF recurrence risk is to create more durable lesions for pulmonary vein isolation, he added.
Another discussant, Dr. Paulus Kirchhof, took an upbeat view of AMIO-CAT.
“My take is that your secondary outcome – reduced hospitalizations – may actually have greater impact for the patient, for patient care, and for payers, than the primary outcome of arrhythmia recurrence,” said Dr. Kirchhof, professor of cardiovascular sciences at the University of Birmingham (England).
Dr. Darkner noted that while she and her coworkers didn’t carry out an economic analysis, it’s quite likely that the reduction in hospitalization and cardioversions would translate into decreased cost of postablation care.
Simultaneously with her presentation in Barcelona, the AMIO-CAT results were published online (Eur. Heart J. 2014 PMID: 25182250).
AMIO-CAT was funded by the Danish Heart Foundation and the Heart Center Research Committee at Copenhagen University Hospital. Dr. Darkner reported having no financial conflicts. Dr. Kirchhof and Dr. Kuck disclosed receiving research grants from and/or serving as consultant to numerous medical device and pharmaceutical companies.
BARCELONA – Eight weeks of oral amiodarone beginning right after catheter ablation for atrial fibrillation more than halved the rates of cardioversion and arrhythmia-related hospitalization for 3 months in the AMIO-CAT trial.
This was a consequence of the sharp reduction in the incidence of AF lasting more than 30 seconds during that interval: 34% in the amiodarone group, compared with 53% in placebo-treated controls, Dr. Stine Darkner reported at the annual congress of the European Society of Cardiology.
“Patients undergoing catheter ablation for AF quite often experience arrhythmias within the first few months after ablation. The ablation itself is thought to cause some of these arrhythmias. It is quite a distressing experience for the patient,” observed Dr. Darkner of Copenhagen University Hospital.
While the benefits at 3 months were significant for the study population as a whole, upon closer analysis the benefits reached statistical significance only in patients treated for persistent AF. Although trends were favorable in subjects with paroxysmal AF, the benefits were less pronounced.
“The implication is that short-term amiodarone should perhaps not be standard treatment after ablation, but should be considered in patients with persistent AF,” she concluded.
At 6 months, there was no significant difference in the cumulative AF recurrence rate, which was the prespecified primary outcome. The rates were 52% in the amiodarone group and 61% with placebo, according to Dr. Darkner.
AMIO-CAT (Amiodarone After Catheter Ablation for Atrial Fibrillation) was the first double-blind, placebo-controlled clinical trial to evaluate the impact of a short course of amiodarone on recovery after AF ablation. The study comprised 212 patients undergoing radiofrequency ablation for paroxysmal or persistent AF.
During the first 3 months post ablation, a time that electrophysiologists call “the blanking period,” the amiodarone group experienced a 57% lower risk of atrial tachyarrythmia-related hospitalization and a 64% reduction in cardioversions, compared with controls.
The side effects reported by patients on amiodarone were transient and are familiar in nature to physicians who prescribe the antiarrhythmic agent: sleep disruption, GI complaints, photosensitivity, and asymptomatic changes in thyroid hormone levels. None of the side effects was serious. Quality of life as measured by the SF-36 was not reduced in the amiodarone group, despite the increase in side effects.
The dosing regimen in the study was amiodarone at 400 mg on the night of the ablation, then 400 mg twice daily for 13 days, followed by amiodarone at 200 mg twice daily for 2 weeks, and finally 200 mg once daily for 4 weeks.
Discussant Karl-Heinz Kuck said he wasn’t surprised by the lack of benefit at 6 months in AMIO-CAT.
“We know that in patients with paroxysmal or persistent AF, the predominant cause of atrial arrhythmia recurrence post ablation at any point in time is pulmonary vein reconduction, which is unlikely to be altered by 8 weeks of amiodarone, especially after discontinuing the drug. We know that AF begets AF, but we don’t know that sinus rhythm begets sinus rhythm,” said Dr. Kuck, head of cardiology at St. Georg Hospital in Hamburg, Germany.
A more promising strategy to reduce the long-term AF recurrence risk is to create more durable lesions for pulmonary vein isolation, he added.
Another discussant, Dr. Paulus Kirchhof, took an upbeat view of AMIO-CAT.
“My take is that your secondary outcome – reduced hospitalizations – may actually have greater impact for the patient, for patient care, and for payers, than the primary outcome of arrhythmia recurrence,” said Dr. Kirchhof, professor of cardiovascular sciences at the University of Birmingham (England).
Dr. Darkner noted that while she and her coworkers didn’t carry out an economic analysis, it’s quite likely that the reduction in hospitalization and cardioversions would translate into decreased cost of postablation care.
Simultaneously with her presentation in Barcelona, the AMIO-CAT results were published online (Eur. Heart J. 2014 PMID: 25182250).
AMIO-CAT was funded by the Danish Heart Foundation and the Heart Center Research Committee at Copenhagen University Hospital. Dr. Darkner reported having no financial conflicts. Dr. Kirchhof and Dr. Kuck disclosed receiving research grants from and/or serving as consultant to numerous medical device and pharmaceutical companies.
BARCELONA – Eight weeks of oral amiodarone beginning right after catheter ablation for atrial fibrillation more than halved the rates of cardioversion and arrhythmia-related hospitalization for 3 months in the AMIO-CAT trial.
This was a consequence of the sharp reduction in the incidence of AF lasting more than 30 seconds during that interval: 34% in the amiodarone group, compared with 53% in placebo-treated controls, Dr. Stine Darkner reported at the annual congress of the European Society of Cardiology.
“Patients undergoing catheter ablation for AF quite often experience arrhythmias within the first few months after ablation. The ablation itself is thought to cause some of these arrhythmias. It is quite a distressing experience for the patient,” observed Dr. Darkner of Copenhagen University Hospital.
While the benefits at 3 months were significant for the study population as a whole, upon closer analysis the benefits reached statistical significance only in patients treated for persistent AF. Although trends were favorable in subjects with paroxysmal AF, the benefits were less pronounced.
“The implication is that short-term amiodarone should perhaps not be standard treatment after ablation, but should be considered in patients with persistent AF,” she concluded.
At 6 months, there was no significant difference in the cumulative AF recurrence rate, which was the prespecified primary outcome. The rates were 52% in the amiodarone group and 61% with placebo, according to Dr. Darkner.
AMIO-CAT (Amiodarone After Catheter Ablation for Atrial Fibrillation) was the first double-blind, placebo-controlled clinical trial to evaluate the impact of a short course of amiodarone on recovery after AF ablation. The study comprised 212 patients undergoing radiofrequency ablation for paroxysmal or persistent AF.
During the first 3 months post ablation, a time that electrophysiologists call “the blanking period,” the amiodarone group experienced a 57% lower risk of atrial tachyarrythmia-related hospitalization and a 64% reduction in cardioversions, compared with controls.
The side effects reported by patients on amiodarone were transient and are familiar in nature to physicians who prescribe the antiarrhythmic agent: sleep disruption, GI complaints, photosensitivity, and asymptomatic changes in thyroid hormone levels. None of the side effects was serious. Quality of life as measured by the SF-36 was not reduced in the amiodarone group, despite the increase in side effects.
The dosing regimen in the study was amiodarone at 400 mg on the night of the ablation, then 400 mg twice daily for 13 days, followed by amiodarone at 200 mg twice daily for 2 weeks, and finally 200 mg once daily for 4 weeks.
Discussant Karl-Heinz Kuck said he wasn’t surprised by the lack of benefit at 6 months in AMIO-CAT.
“We know that in patients with paroxysmal or persistent AF, the predominant cause of atrial arrhythmia recurrence post ablation at any point in time is pulmonary vein reconduction, which is unlikely to be altered by 8 weeks of amiodarone, especially after discontinuing the drug. We know that AF begets AF, but we don’t know that sinus rhythm begets sinus rhythm,” said Dr. Kuck, head of cardiology at St. Georg Hospital in Hamburg, Germany.
A more promising strategy to reduce the long-term AF recurrence risk is to create more durable lesions for pulmonary vein isolation, he added.
Another discussant, Dr. Paulus Kirchhof, took an upbeat view of AMIO-CAT.
“My take is that your secondary outcome – reduced hospitalizations – may actually have greater impact for the patient, for patient care, and for payers, than the primary outcome of arrhythmia recurrence,” said Dr. Kirchhof, professor of cardiovascular sciences at the University of Birmingham (England).
Dr. Darkner noted that while she and her coworkers didn’t carry out an economic analysis, it’s quite likely that the reduction in hospitalization and cardioversions would translate into decreased cost of postablation care.
Simultaneously with her presentation in Barcelona, the AMIO-CAT results were published online (Eur. Heart J. 2014 PMID: 25182250).
AMIO-CAT was funded by the Danish Heart Foundation and the Heart Center Research Committee at Copenhagen University Hospital. Dr. Darkner reported having no financial conflicts. Dr. Kirchhof and Dr. Kuck disclosed receiving research grants from and/or serving as consultant to numerous medical device and pharmaceutical companies.
AT THE ESC CONGRESS 2014
Key clinical point: Eight weeks of amiodarone after catheter ablation of persistent AF is worth considering.
Major finding: During the first 3 months following catheter ablation of patients with paroxysmal or persistent AF, patients on a short course of oral amiodarone had a 57% reduction in atrial tachyarrhythmia-related hospitalizations and a 64% decrease in the risk of cardioversions compared with placebo-treated controls.
Data source: The AMIO-CAT trial, a randomized, double-blind Danish study of 212 patients randomized to 8 weeks of amiodarone or placebo following radiofrequency ablation of paroxysmal or persistent AF.
Disclosures: The Danish Heart Foundation and Copenhagen University Hospital funded the study. The presenter reported having no financial conflicts of interest.
LCZ696 shows promise as novel antihypertensive agent
BARCELONA – The novel investigational agent LCZ696 – now grabbing attention for its dramatic mortality benefit in patients with heart failure with reduced ejection fraction – also appears to have a bright future as an antihypertensive medication.
LCZ696, a first-in-class ARNI, or angiotensin receptor neprilysin inhibitor, showed impressive blood pressure–lowering efficacy in obese and overweight hypertensive patients in a pair of 8-week, randomized, double-blind, controlled clinical trials presented at the annual congress of the European Society of Cardiology.
In another hypertension study presented at the meeting, LCZ696 added to amlodipine resulted in blood pressure control as defined by values below 140/90 mm Hg in 68.5% of Asian patients with uncontrolled hypertension on amlodipine monotherapy.
LCZ696 comprises sacubitril, which boosts levels of endogenous vasoactive peptides by inhibition of neprilysin, in combination with the angiotensin receptor blocker valsartan.
Dr. Luis M. Ruilope presented the pooled results of two multicenter, randomized, 8-week, double-blind trials in a total of 848 patients with mild to moderate hypertension, of whom 347 were obese, 368 were overweight, and 133 were normal weight. Participants were randomized to once-daily oral LCZ696 at 400 mg or valsartan at 320 mg.
LCZ696 proved more effective than valsartan in lowering 24-hour ambulatory blood pressure, as well as office blood pressure and pulse pressure, in all three patient groups. In obese patients, for example, LCZ696 reduced mean office systolic blood pressure by 7.5 mm Hg more than did valsartan, mean office diastolic blood pressure by 2.9 mm Hg more, and mean office pulse pressure by 4.6 mm Hg more. Both drugs were generally well tolerated and were free of serious side effects.
It was important to demonstrate LCZ696’s efficacy in these studies of overweight and obese patients because heavy patients comprise a key segment of the hypertensive population. Indeed, hypertension is roughly twice as prevalent in obese as in nonobese individuals, noted Dr. Ruilope, who os the head of the hypertension unit at the Hospital 12 de Octubre, Madrid.
Separately, Dr. Jiguang Wang of the Shanghai (China) Institute of Hypertension presented an 8-week, double-blind, multicenter clinical trial in which 266 Asian patients with systolic hypertension not controlled with amlodipine monotherapy were randomized to LCZ696 at 200 mg/day plus amlodipine at 5 mg/day, or to amlodipine alone at 5 mg/day.
Importantly, all subjects had hypertension previously uncontrolled with amlodipine. This is a common situation in Asian populations, in whom calcium channel blockers such as amlodipine often activate both the renin-angiotensin-aldosterone system (RAAS) and the sympathetic nervous system, thereby limiting treatment efficacy. The hypothesis underlying this study was that the neprilysin inhibition achieved by adding the ARNI to amlodipine would counteract the sympathetic and RAAS activation, with resultant greater blood pressure–lowering efficacy, Dr. Wang explained.
Over the course of 8 weeks, mean 24-hour ambulatory systolic blood pressure dropped significantly, by 13.9 mm Hg from a baseline of 139 mm Hg in patients treated with LCZ696 added on to amlodipine, compared with a 0.8–mm Hg decrease in patients on amlodipine only. Control of sitting systolic blood pressure was achieved in 72% of the combination therapy group compared to 35% on amlodipine monotherapy.
LCZ696 was the talk of ESC 2014 as a consequence of its impressive performance in the landmark PARADIGM-HF trial, in which it reduced the relative risks of cardiovascular death and hospitalization for heart failure by 20% and 21%, respectively, in patients with heart failure with reduced ejection fraction on optimal background therapy.
Dr. Ruilope and Dr. Wang reported receiving consulting fees from Novartis, which funded the studies and is developing LCZ696 for the marketplace.
BARCELONA – The novel investigational agent LCZ696 – now grabbing attention for its dramatic mortality benefit in patients with heart failure with reduced ejection fraction – also appears to have a bright future as an antihypertensive medication.
LCZ696, a first-in-class ARNI, or angiotensin receptor neprilysin inhibitor, showed impressive blood pressure–lowering efficacy in obese and overweight hypertensive patients in a pair of 8-week, randomized, double-blind, controlled clinical trials presented at the annual congress of the European Society of Cardiology.
In another hypertension study presented at the meeting, LCZ696 added to amlodipine resulted in blood pressure control as defined by values below 140/90 mm Hg in 68.5% of Asian patients with uncontrolled hypertension on amlodipine monotherapy.
LCZ696 comprises sacubitril, which boosts levels of endogenous vasoactive peptides by inhibition of neprilysin, in combination with the angiotensin receptor blocker valsartan.
Dr. Luis M. Ruilope presented the pooled results of two multicenter, randomized, 8-week, double-blind trials in a total of 848 patients with mild to moderate hypertension, of whom 347 were obese, 368 were overweight, and 133 were normal weight. Participants were randomized to once-daily oral LCZ696 at 400 mg or valsartan at 320 mg.
LCZ696 proved more effective than valsartan in lowering 24-hour ambulatory blood pressure, as well as office blood pressure and pulse pressure, in all three patient groups. In obese patients, for example, LCZ696 reduced mean office systolic blood pressure by 7.5 mm Hg more than did valsartan, mean office diastolic blood pressure by 2.9 mm Hg more, and mean office pulse pressure by 4.6 mm Hg more. Both drugs were generally well tolerated and were free of serious side effects.
It was important to demonstrate LCZ696’s efficacy in these studies of overweight and obese patients because heavy patients comprise a key segment of the hypertensive population. Indeed, hypertension is roughly twice as prevalent in obese as in nonobese individuals, noted Dr. Ruilope, who os the head of the hypertension unit at the Hospital 12 de Octubre, Madrid.
Separately, Dr. Jiguang Wang of the Shanghai (China) Institute of Hypertension presented an 8-week, double-blind, multicenter clinical trial in which 266 Asian patients with systolic hypertension not controlled with amlodipine monotherapy were randomized to LCZ696 at 200 mg/day plus amlodipine at 5 mg/day, or to amlodipine alone at 5 mg/day.
Importantly, all subjects had hypertension previously uncontrolled with amlodipine. This is a common situation in Asian populations, in whom calcium channel blockers such as amlodipine often activate both the renin-angiotensin-aldosterone system (RAAS) and the sympathetic nervous system, thereby limiting treatment efficacy. The hypothesis underlying this study was that the neprilysin inhibition achieved by adding the ARNI to amlodipine would counteract the sympathetic and RAAS activation, with resultant greater blood pressure–lowering efficacy, Dr. Wang explained.
Over the course of 8 weeks, mean 24-hour ambulatory systolic blood pressure dropped significantly, by 13.9 mm Hg from a baseline of 139 mm Hg in patients treated with LCZ696 added on to amlodipine, compared with a 0.8–mm Hg decrease in patients on amlodipine only. Control of sitting systolic blood pressure was achieved in 72% of the combination therapy group compared to 35% on amlodipine monotherapy.
LCZ696 was the talk of ESC 2014 as a consequence of its impressive performance in the landmark PARADIGM-HF trial, in which it reduced the relative risks of cardiovascular death and hospitalization for heart failure by 20% and 21%, respectively, in patients with heart failure with reduced ejection fraction on optimal background therapy.
Dr. Ruilope and Dr. Wang reported receiving consulting fees from Novartis, which funded the studies and is developing LCZ696 for the marketplace.
BARCELONA – The novel investigational agent LCZ696 – now grabbing attention for its dramatic mortality benefit in patients with heart failure with reduced ejection fraction – also appears to have a bright future as an antihypertensive medication.
LCZ696, a first-in-class ARNI, or angiotensin receptor neprilysin inhibitor, showed impressive blood pressure–lowering efficacy in obese and overweight hypertensive patients in a pair of 8-week, randomized, double-blind, controlled clinical trials presented at the annual congress of the European Society of Cardiology.
In another hypertension study presented at the meeting, LCZ696 added to amlodipine resulted in blood pressure control as defined by values below 140/90 mm Hg in 68.5% of Asian patients with uncontrolled hypertension on amlodipine monotherapy.
LCZ696 comprises sacubitril, which boosts levels of endogenous vasoactive peptides by inhibition of neprilysin, in combination with the angiotensin receptor blocker valsartan.
Dr. Luis M. Ruilope presented the pooled results of two multicenter, randomized, 8-week, double-blind trials in a total of 848 patients with mild to moderate hypertension, of whom 347 were obese, 368 were overweight, and 133 were normal weight. Participants were randomized to once-daily oral LCZ696 at 400 mg or valsartan at 320 mg.
LCZ696 proved more effective than valsartan in lowering 24-hour ambulatory blood pressure, as well as office blood pressure and pulse pressure, in all three patient groups. In obese patients, for example, LCZ696 reduced mean office systolic blood pressure by 7.5 mm Hg more than did valsartan, mean office diastolic blood pressure by 2.9 mm Hg more, and mean office pulse pressure by 4.6 mm Hg more. Both drugs were generally well tolerated and were free of serious side effects.
It was important to demonstrate LCZ696’s efficacy in these studies of overweight and obese patients because heavy patients comprise a key segment of the hypertensive population. Indeed, hypertension is roughly twice as prevalent in obese as in nonobese individuals, noted Dr. Ruilope, who os the head of the hypertension unit at the Hospital 12 de Octubre, Madrid.
Separately, Dr. Jiguang Wang of the Shanghai (China) Institute of Hypertension presented an 8-week, double-blind, multicenter clinical trial in which 266 Asian patients with systolic hypertension not controlled with amlodipine monotherapy were randomized to LCZ696 at 200 mg/day plus amlodipine at 5 mg/day, or to amlodipine alone at 5 mg/day.
Importantly, all subjects had hypertension previously uncontrolled with amlodipine. This is a common situation in Asian populations, in whom calcium channel blockers such as amlodipine often activate both the renin-angiotensin-aldosterone system (RAAS) and the sympathetic nervous system, thereby limiting treatment efficacy. The hypothesis underlying this study was that the neprilysin inhibition achieved by adding the ARNI to amlodipine would counteract the sympathetic and RAAS activation, with resultant greater blood pressure–lowering efficacy, Dr. Wang explained.
Over the course of 8 weeks, mean 24-hour ambulatory systolic blood pressure dropped significantly, by 13.9 mm Hg from a baseline of 139 mm Hg in patients treated with LCZ696 added on to amlodipine, compared with a 0.8–mm Hg decrease in patients on amlodipine only. Control of sitting systolic blood pressure was achieved in 72% of the combination therapy group compared to 35% on amlodipine monotherapy.
LCZ696 was the talk of ESC 2014 as a consequence of its impressive performance in the landmark PARADIGM-HF trial, in which it reduced the relative risks of cardiovascular death and hospitalization for heart failure by 20% and 21%, respectively, in patients with heart failure with reduced ejection fraction on optimal background therapy.
Dr. Ruilope and Dr. Wang reported receiving consulting fees from Novartis, which funded the studies and is developing LCZ696 for the marketplace.
AT THE ESC CONGRESS 2014
Wearable defibrillator impresses as bridge to ICD decision
BARCELONA – Use of the LifeVest wearable cardioverter defibrillator for 3 months is a safe and effective means of buying time to decide whether to place a permanent implantable cardioverter defibrillator for primary prevention of sudden cardiac death in potential candidates, according to the first report from a large prospective U.S. registry.
The wearable cardioverter defibrillator (WCD) simultaneously provides patient monitoring and reliable protection against fatal arrhythmias while physicians wait to see if, for example, a patient’s left ventricular ejection fraction, depressed to less than 35% immediately post-MI, will recover over the first several months or if life-threatening arrhythmias will arise warranting implantable cardioverter defibrillator (ICD) implantation, Dr. Valentina Kutyifa explained while presenting the results of the WEARIT-II registry at the annual congress of the European Society of Cardiology.
"The WCD seems to be a powerful risk-assessment tool to identify patients at a high risk for sudden cardiac death who need subsequent ICD implantation. The WCD can be used as a bridge to a decision for appropriate ICD implantation in patients with a low ejection fraction who are immediately post-MI, or following coronary revascularization, or with new-onset dilated cardiomyopathy who are at high risk for sudden cardiac death until stabilization, or who have an inherited arrhythmia or congenital disorder," said Dr. Kutyifa of the University of Rochester (N.Y.).
She reported on 2,000 U.S. patients in those categories who participated in the prospective WEARIT-II (Prospective Registry and Follow-Up of Patients Using the Wearable Defibrillator) registry. All received from their physicians a 3-month prescription for the LifeVest WCD, which is covered by Medicare and by most health plans. Of the 2,000 patients, 927 had a left ventricular ejection fraction (LVEF) of 35% or below owing to nonischemic cardiomyopathy, 805 had ischemic cardiomyopathy, and 268 had an inherited or congenital arrhythmogenic disorder, such as long QT syndrome or arrhythmogenic right ventricular dysplasia.
Participants’ median ejection fraction at enrollment was 25%, and 52% of subjects had heart failure symptoms at that time. Patients wore the WCD for a median of 22.5 hours per day, with no difference in wear time by disease etiology.
In terms of WCD safety, the inappropriate shock rate was just 0.5% in 2,000 patients during 3 months. No study deaths occurred related to unsuccessful attempts at termination of ventricular tachycardia (VT) or ventricular fibrillation (VF) by the WCD. Three patients died while wearing the WCD, and in all three cases the WCD detected asystole at the time of death.
During 90 days of WCD wear, 2.1% of patients experienced VT or VF, for an event rate of 22 per 100 person-years. The WCD administered a shock for VT/VF in 1.1% of patients. Sustained VT or VF occurred in 1.4% of patients, while 3.6% of participants experienced atrial arrhythmias or supraventricular tachycardia, with an event rate of 121 per 100 person-years.
At the end of the 3-month period of WCD use, 42% of patients with ischemic cardiomyopathy got an ICD, as did 36% of those with nonischemic cardiomyopathy and 46% who had a congenital or inherited condition. The main reason for not implanting an ICD was a boost in ejection fraction.
The WCD frequently detected arrhythmias which facilitated the decision whether to implant an ICD or not. Eighty-five percent of patients with VT/VF treated with a WCD shock got an ICD, as did 65% of those with sustained VT that terminated spontaneously during the wearable device’s detection time. Of patients with WCD-detected atrial arrhythmias, 48% received an ICD, as did 39% of those with no arrhythmias during the 3-month period.
Dr. Kutyifa characterized the WCD as helping to fill an unmet need for improved selection of patients for primary ICD therapy. In the landmark MADIT-II trial, for example, only 4% of patients received an appropriate ICD shock during 4 years of follow-up (N. Engl. J. Med. 2002;346:877-82).
Asked about the cost-effectiveness of WCD as a bridge to the ICD decision, she replied that such data weren’t included in the WEARIT-II registry.
"The up-front cost of the device may seem rather high, but if you think about the fact that if we use the WCD and are then able to make the decision to not implant an ICD in patients who don’t need it, we really need to look at the long-term savings: the cost of the ICD, battery changes, and issues of possible lead extraction. So I think long-term this management strategy would be cost-effective," the electrophysiologist said, adding that the WCD is also rentable.
At the congress-closing conference highlights overview session, Dr. Sylvia G. Priori singled out the WEARIT-II registry as one of the top developments in the field of arrhythmias presented at this year’s meeting. She noted that the WCD has been available for several years – Medicare covers it, as do most U.S. health plans – yet until now many physicians have wanted to see stronger evidence of safety and effectiveness before incorporating the WCD into their own practices. WEARIT-II, she said, provides that supporting evidence.
"This was quite an important study. It showed the device is able to recognize life-threatening arrhythmias, so it is safe, and it does not deliver an excessive number of inappropriate shocks. And there were no deaths related to the device," commented Dr. Priori of the University of Pavia (Italy).
"The take-home message is that, yes, we know there are patients where we are reluctant to immediately say that an ICD should be implanted forever, and now we should consider testing with this WCD, which has a low risk of inappropriate shocks and a good record in shocking patients out of the arrhythmia," she added.
The WEARIT-II registry is now expanding via enrollment of patients in Europe and Israel. The registry is funded by Zoll, which markets the WCD. Dr. Kutyifa reported having received research grants from the company as well as from other device manufacturers.
BARCELONA – Use of the LifeVest wearable cardioverter defibrillator for 3 months is a safe and effective means of buying time to decide whether to place a permanent implantable cardioverter defibrillator for primary prevention of sudden cardiac death in potential candidates, according to the first report from a large prospective U.S. registry.
The wearable cardioverter defibrillator (WCD) simultaneously provides patient monitoring and reliable protection against fatal arrhythmias while physicians wait to see if, for example, a patient’s left ventricular ejection fraction, depressed to less than 35% immediately post-MI, will recover over the first several months or if life-threatening arrhythmias will arise warranting implantable cardioverter defibrillator (ICD) implantation, Dr. Valentina Kutyifa explained while presenting the results of the WEARIT-II registry at the annual congress of the European Society of Cardiology.
"The WCD seems to be a powerful risk-assessment tool to identify patients at a high risk for sudden cardiac death who need subsequent ICD implantation. The WCD can be used as a bridge to a decision for appropriate ICD implantation in patients with a low ejection fraction who are immediately post-MI, or following coronary revascularization, or with new-onset dilated cardiomyopathy who are at high risk for sudden cardiac death until stabilization, or who have an inherited arrhythmia or congenital disorder," said Dr. Kutyifa of the University of Rochester (N.Y.).
She reported on 2,000 U.S. patients in those categories who participated in the prospective WEARIT-II (Prospective Registry and Follow-Up of Patients Using the Wearable Defibrillator) registry. All received from their physicians a 3-month prescription for the LifeVest WCD, which is covered by Medicare and by most health plans. Of the 2,000 patients, 927 had a left ventricular ejection fraction (LVEF) of 35% or below owing to nonischemic cardiomyopathy, 805 had ischemic cardiomyopathy, and 268 had an inherited or congenital arrhythmogenic disorder, such as long QT syndrome or arrhythmogenic right ventricular dysplasia.
Participants’ median ejection fraction at enrollment was 25%, and 52% of subjects had heart failure symptoms at that time. Patients wore the WCD for a median of 22.5 hours per day, with no difference in wear time by disease etiology.
In terms of WCD safety, the inappropriate shock rate was just 0.5% in 2,000 patients during 3 months. No study deaths occurred related to unsuccessful attempts at termination of ventricular tachycardia (VT) or ventricular fibrillation (VF) by the WCD. Three patients died while wearing the WCD, and in all three cases the WCD detected asystole at the time of death.
During 90 days of WCD wear, 2.1% of patients experienced VT or VF, for an event rate of 22 per 100 person-years. The WCD administered a shock for VT/VF in 1.1% of patients. Sustained VT or VF occurred in 1.4% of patients, while 3.6% of participants experienced atrial arrhythmias or supraventricular tachycardia, with an event rate of 121 per 100 person-years.
At the end of the 3-month period of WCD use, 42% of patients with ischemic cardiomyopathy got an ICD, as did 36% of those with nonischemic cardiomyopathy and 46% who had a congenital or inherited condition. The main reason for not implanting an ICD was a boost in ejection fraction.
The WCD frequently detected arrhythmias which facilitated the decision whether to implant an ICD or not. Eighty-five percent of patients with VT/VF treated with a WCD shock got an ICD, as did 65% of those with sustained VT that terminated spontaneously during the wearable device’s detection time. Of patients with WCD-detected atrial arrhythmias, 48% received an ICD, as did 39% of those with no arrhythmias during the 3-month period.
Dr. Kutyifa characterized the WCD as helping to fill an unmet need for improved selection of patients for primary ICD therapy. In the landmark MADIT-II trial, for example, only 4% of patients received an appropriate ICD shock during 4 years of follow-up (N. Engl. J. Med. 2002;346:877-82).
Asked about the cost-effectiveness of WCD as a bridge to the ICD decision, she replied that such data weren’t included in the WEARIT-II registry.
"The up-front cost of the device may seem rather high, but if you think about the fact that if we use the WCD and are then able to make the decision to not implant an ICD in patients who don’t need it, we really need to look at the long-term savings: the cost of the ICD, battery changes, and issues of possible lead extraction. So I think long-term this management strategy would be cost-effective," the electrophysiologist said, adding that the WCD is also rentable.
At the congress-closing conference highlights overview session, Dr. Sylvia G. Priori singled out the WEARIT-II registry as one of the top developments in the field of arrhythmias presented at this year’s meeting. She noted that the WCD has been available for several years – Medicare covers it, as do most U.S. health plans – yet until now many physicians have wanted to see stronger evidence of safety and effectiveness before incorporating the WCD into their own practices. WEARIT-II, she said, provides that supporting evidence.
"This was quite an important study. It showed the device is able to recognize life-threatening arrhythmias, so it is safe, and it does not deliver an excessive number of inappropriate shocks. And there were no deaths related to the device," commented Dr. Priori of the University of Pavia (Italy).
"The take-home message is that, yes, we know there are patients where we are reluctant to immediately say that an ICD should be implanted forever, and now we should consider testing with this WCD, which has a low risk of inappropriate shocks and a good record in shocking patients out of the arrhythmia," she added.
The WEARIT-II registry is now expanding via enrollment of patients in Europe and Israel. The registry is funded by Zoll, which markets the WCD. Dr. Kutyifa reported having received research grants from the company as well as from other device manufacturers.
BARCELONA – Use of the LifeVest wearable cardioverter defibrillator for 3 months is a safe and effective means of buying time to decide whether to place a permanent implantable cardioverter defibrillator for primary prevention of sudden cardiac death in potential candidates, according to the first report from a large prospective U.S. registry.
The wearable cardioverter defibrillator (WCD) simultaneously provides patient monitoring and reliable protection against fatal arrhythmias while physicians wait to see if, for example, a patient’s left ventricular ejection fraction, depressed to less than 35% immediately post-MI, will recover over the first several months or if life-threatening arrhythmias will arise warranting implantable cardioverter defibrillator (ICD) implantation, Dr. Valentina Kutyifa explained while presenting the results of the WEARIT-II registry at the annual congress of the European Society of Cardiology.
"The WCD seems to be a powerful risk-assessment tool to identify patients at a high risk for sudden cardiac death who need subsequent ICD implantation. The WCD can be used as a bridge to a decision for appropriate ICD implantation in patients with a low ejection fraction who are immediately post-MI, or following coronary revascularization, or with new-onset dilated cardiomyopathy who are at high risk for sudden cardiac death until stabilization, or who have an inherited arrhythmia or congenital disorder," said Dr. Kutyifa of the University of Rochester (N.Y.).
She reported on 2,000 U.S. patients in those categories who participated in the prospective WEARIT-II (Prospective Registry and Follow-Up of Patients Using the Wearable Defibrillator) registry. All received from their physicians a 3-month prescription for the LifeVest WCD, which is covered by Medicare and by most health plans. Of the 2,000 patients, 927 had a left ventricular ejection fraction (LVEF) of 35% or below owing to nonischemic cardiomyopathy, 805 had ischemic cardiomyopathy, and 268 had an inherited or congenital arrhythmogenic disorder, such as long QT syndrome or arrhythmogenic right ventricular dysplasia.
Participants’ median ejection fraction at enrollment was 25%, and 52% of subjects had heart failure symptoms at that time. Patients wore the WCD for a median of 22.5 hours per day, with no difference in wear time by disease etiology.
In terms of WCD safety, the inappropriate shock rate was just 0.5% in 2,000 patients during 3 months. No study deaths occurred related to unsuccessful attempts at termination of ventricular tachycardia (VT) or ventricular fibrillation (VF) by the WCD. Three patients died while wearing the WCD, and in all three cases the WCD detected asystole at the time of death.
During 90 days of WCD wear, 2.1% of patients experienced VT or VF, for an event rate of 22 per 100 person-years. The WCD administered a shock for VT/VF in 1.1% of patients. Sustained VT or VF occurred in 1.4% of patients, while 3.6% of participants experienced atrial arrhythmias or supraventricular tachycardia, with an event rate of 121 per 100 person-years.
At the end of the 3-month period of WCD use, 42% of patients with ischemic cardiomyopathy got an ICD, as did 36% of those with nonischemic cardiomyopathy and 46% who had a congenital or inherited condition. The main reason for not implanting an ICD was a boost in ejection fraction.
The WCD frequently detected arrhythmias which facilitated the decision whether to implant an ICD or not. Eighty-five percent of patients with VT/VF treated with a WCD shock got an ICD, as did 65% of those with sustained VT that terminated spontaneously during the wearable device’s detection time. Of patients with WCD-detected atrial arrhythmias, 48% received an ICD, as did 39% of those with no arrhythmias during the 3-month period.
Dr. Kutyifa characterized the WCD as helping to fill an unmet need for improved selection of patients for primary ICD therapy. In the landmark MADIT-II trial, for example, only 4% of patients received an appropriate ICD shock during 4 years of follow-up (N. Engl. J. Med. 2002;346:877-82).
Asked about the cost-effectiveness of WCD as a bridge to the ICD decision, she replied that such data weren’t included in the WEARIT-II registry.
"The up-front cost of the device may seem rather high, but if you think about the fact that if we use the WCD and are then able to make the decision to not implant an ICD in patients who don’t need it, we really need to look at the long-term savings: the cost of the ICD, battery changes, and issues of possible lead extraction. So I think long-term this management strategy would be cost-effective," the electrophysiologist said, adding that the WCD is also rentable.
At the congress-closing conference highlights overview session, Dr. Sylvia G. Priori singled out the WEARIT-II registry as one of the top developments in the field of arrhythmias presented at this year’s meeting. She noted that the WCD has been available for several years – Medicare covers it, as do most U.S. health plans – yet until now many physicians have wanted to see stronger evidence of safety and effectiveness before incorporating the WCD into their own practices. WEARIT-II, she said, provides that supporting evidence.
"This was quite an important study. It showed the device is able to recognize life-threatening arrhythmias, so it is safe, and it does not deliver an excessive number of inappropriate shocks. And there were no deaths related to the device," commented Dr. Priori of the University of Pavia (Italy).
"The take-home message is that, yes, we know there are patients where we are reluctant to immediately say that an ICD should be implanted forever, and now we should consider testing with this WCD, which has a low risk of inappropriate shocks and a good record in shocking patients out of the arrhythmia," she added.
The WEARIT-II registry is now expanding via enrollment of patients in Europe and Israel. The registry is funded by Zoll, which markets the WCD. Dr. Kutyifa reported having received research grants from the company as well as from other device manufacturers.
AT THE ESC CONGRESS 2014
Key clinical point: The temporary use of a commercially available wearable cardioverter defibrillator buys physicians time to decide whether or not to take the big step of permanent implantable cardioverter defibrillator placement in potential candidates for whom questions exist.
Major finding: During 3 months use of the LifeVest wearable cardioverter defibrillator, the vest detected VT/VF and administered an arrhythmia-ending shock in 1.1% of device wearers with an LVEF of 35% or less.
Data source: The prospective WEARIT-II registry includes to date 2,000 patients who are potential candidates for prophylactic implantation of a permanent cardioverter defibrillator for primary prevention of sudden cardiac arrest.
Disclosures: The presenter reported receiving research grant support from Zoll, which funded the WEARIT-II registry.
Pulmonary vein isolation alone may be best ablative procedure for persistent atrial fibrillation
BARCELONA – More extensive catheter ablation procedures offered no benefit over pulmonary vein isolation alone for persistent atrial fibrillation in the largest-ever randomized trial examining outcomes of the three most popular ablation strategies.
"This study, the STAR AF 2 trial, will force a change in thinking both in the guidelines as well as in clinical practice," Dr. Atul Verma predicted, in presenting the study findings at the annual congress of the European Society of Cardiology.
Because of a widespread belief that catheter ablation success rates are probably lower in persistent AF than in paroxysmal AF, guidelines suggest "operators should consider more ablation based on linear lesions or complex fractionated electrograms," in addition to pulmonary vein isolation, in treating patients with persistent AF (Heart Rhythm 2012;9:632-96). The guidelines noted, however, that there is little evidence to support this recommendation.
The STAR AF 2 trial was conducted to learn if more complex ablation procedures really do provide greater efficacy than pulmonary vein isolation (PVI) alone. The study included 589 patients at 48 centers in 12 countries. All patients had persistent AF refractory to at least one antiarrhythmic drug and were about to undergo their first-ever catheter ablation.
Participants were randomized 1:4:4 to PVI alone with the procedural endpoint of entrance and exit block by circular mapping catheter, or PVI plus mapping and ablation of complex fractionated electrograms during AF identified using a validated 3-D mapping system, or PVI plus a left atrial roof line and another line along the mitral valve isthmus with the endpoint of bidirectional block confirmed by prespecified pacing maneuvers.
Patients remained blinded as to which of the three treatments they received. They were prospectively followed with 24-hour Holter monitoring at 3, 6, 9, 12, and 18 months along with weekly transtelephonic monitoring transmissions or at any time they felt symptoms.
Successful PVI was achieved in 97% of patients, complex fractionated electrograms were eliminated in 80% of patients assigned to that strategy, and both target lines were blocked in 74% of patients who underwent linear ablation.
The primary outcome was freedom from a documented episode of AF lasting more than 30 seconds after one procedure with or without antiarrhythmic medication through 18 months. The rates were 59% with PVI only, 48% with PVI plus complex fractionated electrograms, and 44% with PVI and linear ablation. These rates weren’t significantly different.
There were downsides to the two more elaborate ablation strategies. Procedural times were roughly 1 hour longer. Moreover, mean fluoroscopy time was 29 minutes in the PVI-only group, compared with 41 and 42 minutes with the more complex procedures. That translates to 44% more radiation exposure for both operators and patients, with absolutely no resultant added benefit over PVI alone, noted Dr. Verma, an electrophysiologist at Southlake Regional Health Center in Newmarket, Ont.
Complication rates across the board in STAR AF 2 were among the lowest ever reported in a multicenter clinical trial of catheter ablation. Of note, however, the sole fatal complication was the result of an atrial esophageal fistula in a patient assigned to PVI plus electrogram ablation.
Discussant Dr. Jagmeet P. Singh, director of the cardiac resynchronization therapy program at Massachusetts General Hospital, Boston, called STAR AF 2 "a fantastic trial."
"This study surely advocates that less ablation is more – and less works quite well," he said, noting that the roughly 50% success rate at 18 months with PVI alone is comparable to prior published success rates in paroxysmal AF.
Discussant Dr. Paulus Kirchhof said his own recent informal survey of high-volume catheter ablation centers in the United States and Europe indicated roughly one-third do PVI alone for patients with persistent AF, one-third do PVI plus ablation of complex fractionated electrograms, and one-third do PVI plus linear ablation.
"So I would say this was a question at equipoise," added Dr. Kirchhof, professor of cardiovascular sciences at the University of Birmingham (England).
Zeroing in on the added fluoroscopy time associated with the more complex ablation procedures, he noted that observational data suggest lengthier fluoroscopy may be associated with silent, subclinical brain lesions. Based upon the STAR 2 AF results, therefore, a reasonable strategy now for persistent AF is to do PVI alone, then wait and see what happens before considering additional ablation procedures later, he said.
"More importantly, I think this study shows we have to go back to the drawing board. The time pattern of AF – its duration, whether it’s paroxysmal or persistent, the left atrial size – all these things we believe identify patients who need more therapy, they may not actually help us. We just have to accept that not all patients with AF are the same, and that the pattern of AF does not discriminate so well. I think what we can really learn from this trial moving forward is that we need a clinical classification of AF patients. We have to define the patient who would benefit before we continue to develop ever-more intensive interventional strategies," he commented.
Factors worthy of further study as potential tools for separating AF patients into subgroups for treatment purposes include markers of atrial fibrosis, whether by imaging, blood, or ECG patterns; markers of parasympathetic/sympathetic imbalance; clinical markers of abnormal calcium metabolism; or blood markers, Dr. Kirchhof added.
The STAR AF 2 trial was funded by St. Jude Medical. Dr. Verma, Dr. Singh, and Dr. Kirchhof reported receiving grant support from St. Jude Medical as well as other pharmaceutical and medical device companies. In addition, Dr. Verma and Dr. Singh have served on advisory boards for St. Jude.
BARCELONA – More extensive catheter ablation procedures offered no benefit over pulmonary vein isolation alone for persistent atrial fibrillation in the largest-ever randomized trial examining outcomes of the three most popular ablation strategies.
"This study, the STAR AF 2 trial, will force a change in thinking both in the guidelines as well as in clinical practice," Dr. Atul Verma predicted, in presenting the study findings at the annual congress of the European Society of Cardiology.
Because of a widespread belief that catheter ablation success rates are probably lower in persistent AF than in paroxysmal AF, guidelines suggest "operators should consider more ablation based on linear lesions or complex fractionated electrograms," in addition to pulmonary vein isolation, in treating patients with persistent AF (Heart Rhythm 2012;9:632-96). The guidelines noted, however, that there is little evidence to support this recommendation.
The STAR AF 2 trial was conducted to learn if more complex ablation procedures really do provide greater efficacy than pulmonary vein isolation (PVI) alone. The study included 589 patients at 48 centers in 12 countries. All patients had persistent AF refractory to at least one antiarrhythmic drug and were about to undergo their first-ever catheter ablation.
Participants were randomized 1:4:4 to PVI alone with the procedural endpoint of entrance and exit block by circular mapping catheter, or PVI plus mapping and ablation of complex fractionated electrograms during AF identified using a validated 3-D mapping system, or PVI plus a left atrial roof line and another line along the mitral valve isthmus with the endpoint of bidirectional block confirmed by prespecified pacing maneuvers.
Patients remained blinded as to which of the three treatments they received. They were prospectively followed with 24-hour Holter monitoring at 3, 6, 9, 12, and 18 months along with weekly transtelephonic monitoring transmissions or at any time they felt symptoms.
Successful PVI was achieved in 97% of patients, complex fractionated electrograms were eliminated in 80% of patients assigned to that strategy, and both target lines were blocked in 74% of patients who underwent linear ablation.
The primary outcome was freedom from a documented episode of AF lasting more than 30 seconds after one procedure with or without antiarrhythmic medication through 18 months. The rates were 59% with PVI only, 48% with PVI plus complex fractionated electrograms, and 44% with PVI and linear ablation. These rates weren’t significantly different.
There were downsides to the two more elaborate ablation strategies. Procedural times were roughly 1 hour longer. Moreover, mean fluoroscopy time was 29 minutes in the PVI-only group, compared with 41 and 42 minutes with the more complex procedures. That translates to 44% more radiation exposure for both operators and patients, with absolutely no resultant added benefit over PVI alone, noted Dr. Verma, an electrophysiologist at Southlake Regional Health Center in Newmarket, Ont.
Complication rates across the board in STAR AF 2 were among the lowest ever reported in a multicenter clinical trial of catheter ablation. Of note, however, the sole fatal complication was the result of an atrial esophageal fistula in a patient assigned to PVI plus electrogram ablation.
Discussant Dr. Jagmeet P. Singh, director of the cardiac resynchronization therapy program at Massachusetts General Hospital, Boston, called STAR AF 2 "a fantastic trial."
"This study surely advocates that less ablation is more – and less works quite well," he said, noting that the roughly 50% success rate at 18 months with PVI alone is comparable to prior published success rates in paroxysmal AF.
Discussant Dr. Paulus Kirchhof said his own recent informal survey of high-volume catheter ablation centers in the United States and Europe indicated roughly one-third do PVI alone for patients with persistent AF, one-third do PVI plus ablation of complex fractionated electrograms, and one-third do PVI plus linear ablation.
"So I would say this was a question at equipoise," added Dr. Kirchhof, professor of cardiovascular sciences at the University of Birmingham (England).
Zeroing in on the added fluoroscopy time associated with the more complex ablation procedures, he noted that observational data suggest lengthier fluoroscopy may be associated with silent, subclinical brain lesions. Based upon the STAR 2 AF results, therefore, a reasonable strategy now for persistent AF is to do PVI alone, then wait and see what happens before considering additional ablation procedures later, he said.
"More importantly, I think this study shows we have to go back to the drawing board. The time pattern of AF – its duration, whether it’s paroxysmal or persistent, the left atrial size – all these things we believe identify patients who need more therapy, they may not actually help us. We just have to accept that not all patients with AF are the same, and that the pattern of AF does not discriminate so well. I think what we can really learn from this trial moving forward is that we need a clinical classification of AF patients. We have to define the patient who would benefit before we continue to develop ever-more intensive interventional strategies," he commented.
Factors worthy of further study as potential tools for separating AF patients into subgroups for treatment purposes include markers of atrial fibrosis, whether by imaging, blood, or ECG patterns; markers of parasympathetic/sympathetic imbalance; clinical markers of abnormal calcium metabolism; or blood markers, Dr. Kirchhof added.
The STAR AF 2 trial was funded by St. Jude Medical. Dr. Verma, Dr. Singh, and Dr. Kirchhof reported receiving grant support from St. Jude Medical as well as other pharmaceutical and medical device companies. In addition, Dr. Verma and Dr. Singh have served on advisory boards for St. Jude.
BARCELONA – More extensive catheter ablation procedures offered no benefit over pulmonary vein isolation alone for persistent atrial fibrillation in the largest-ever randomized trial examining outcomes of the three most popular ablation strategies.
"This study, the STAR AF 2 trial, will force a change in thinking both in the guidelines as well as in clinical practice," Dr. Atul Verma predicted, in presenting the study findings at the annual congress of the European Society of Cardiology.
Because of a widespread belief that catheter ablation success rates are probably lower in persistent AF than in paroxysmal AF, guidelines suggest "operators should consider more ablation based on linear lesions or complex fractionated electrograms," in addition to pulmonary vein isolation, in treating patients with persistent AF (Heart Rhythm 2012;9:632-96). The guidelines noted, however, that there is little evidence to support this recommendation.
The STAR AF 2 trial was conducted to learn if more complex ablation procedures really do provide greater efficacy than pulmonary vein isolation (PVI) alone. The study included 589 patients at 48 centers in 12 countries. All patients had persistent AF refractory to at least one antiarrhythmic drug and were about to undergo their first-ever catheter ablation.
Participants were randomized 1:4:4 to PVI alone with the procedural endpoint of entrance and exit block by circular mapping catheter, or PVI plus mapping and ablation of complex fractionated electrograms during AF identified using a validated 3-D mapping system, or PVI plus a left atrial roof line and another line along the mitral valve isthmus with the endpoint of bidirectional block confirmed by prespecified pacing maneuvers.
Patients remained blinded as to which of the three treatments they received. They were prospectively followed with 24-hour Holter monitoring at 3, 6, 9, 12, and 18 months along with weekly transtelephonic monitoring transmissions or at any time they felt symptoms.
Successful PVI was achieved in 97% of patients, complex fractionated electrograms were eliminated in 80% of patients assigned to that strategy, and both target lines were blocked in 74% of patients who underwent linear ablation.
The primary outcome was freedom from a documented episode of AF lasting more than 30 seconds after one procedure with or without antiarrhythmic medication through 18 months. The rates were 59% with PVI only, 48% with PVI plus complex fractionated electrograms, and 44% with PVI and linear ablation. These rates weren’t significantly different.
There were downsides to the two more elaborate ablation strategies. Procedural times were roughly 1 hour longer. Moreover, mean fluoroscopy time was 29 minutes in the PVI-only group, compared with 41 and 42 minutes with the more complex procedures. That translates to 44% more radiation exposure for both operators and patients, with absolutely no resultant added benefit over PVI alone, noted Dr. Verma, an electrophysiologist at Southlake Regional Health Center in Newmarket, Ont.
Complication rates across the board in STAR AF 2 were among the lowest ever reported in a multicenter clinical trial of catheter ablation. Of note, however, the sole fatal complication was the result of an atrial esophageal fistula in a patient assigned to PVI plus electrogram ablation.
Discussant Dr. Jagmeet P. Singh, director of the cardiac resynchronization therapy program at Massachusetts General Hospital, Boston, called STAR AF 2 "a fantastic trial."
"This study surely advocates that less ablation is more – and less works quite well," he said, noting that the roughly 50% success rate at 18 months with PVI alone is comparable to prior published success rates in paroxysmal AF.
Discussant Dr. Paulus Kirchhof said his own recent informal survey of high-volume catheter ablation centers in the United States and Europe indicated roughly one-third do PVI alone for patients with persistent AF, one-third do PVI plus ablation of complex fractionated electrograms, and one-third do PVI plus linear ablation.
"So I would say this was a question at equipoise," added Dr. Kirchhof, professor of cardiovascular sciences at the University of Birmingham (England).
Zeroing in on the added fluoroscopy time associated with the more complex ablation procedures, he noted that observational data suggest lengthier fluoroscopy may be associated with silent, subclinical brain lesions. Based upon the STAR 2 AF results, therefore, a reasonable strategy now for persistent AF is to do PVI alone, then wait and see what happens before considering additional ablation procedures later, he said.
"More importantly, I think this study shows we have to go back to the drawing board. The time pattern of AF – its duration, whether it’s paroxysmal or persistent, the left atrial size – all these things we believe identify patients who need more therapy, they may not actually help us. We just have to accept that not all patients with AF are the same, and that the pattern of AF does not discriminate so well. I think what we can really learn from this trial moving forward is that we need a clinical classification of AF patients. We have to define the patient who would benefit before we continue to develop ever-more intensive interventional strategies," he commented.
Factors worthy of further study as potential tools for separating AF patients into subgroups for treatment purposes include markers of atrial fibrosis, whether by imaging, blood, or ECG patterns; markers of parasympathetic/sympathetic imbalance; clinical markers of abnormal calcium metabolism; or blood markers, Dr. Kirchhof added.
The STAR AF 2 trial was funded by St. Jude Medical. Dr. Verma, Dr. Singh, and Dr. Kirchhof reported receiving grant support from St. Jude Medical as well as other pharmaceutical and medical device companies. In addition, Dr. Verma and Dr. Singh have served on advisory boards for St. Jude.
AT THE ESC CONGRESS 2014
Key clinical point: Pulmonary vein isolation alone may offer advantages over more elaborate procedures for persistent atrial fibrillation.
Major finding: The rates of freedom from a documented episode of AF lasting more than 30 seconds were 59% with pulmonary vein isolation only, 48% with PVI plus complex fractionated electrograms, and 44% with PVI and linear ablation.
Data source: The STAR AF 2 trial was a randomized, multicenter prospective study in which 589 patients with persistent AF were randomized to one of three popular catheter ablation strategies.
Disclosures: The study was funded by St. Jude Medical. The presenter has received research grants from and served on advisory boards for St. Jude and other medical device and pharmaceutical companies.
High-dose statins don’t prevent postop AF
BARCELONA – Intensive perioperative statin therapy in patients undergoing CABG surgery doesn’t protect against postop atrial fibrillation or myocardial injury, according to a large randomized clinical trial hailed as the "definitive" study addressing this issue.
"There are many reasons why these patients should be put on statin treatment, but the prevention of postop complications is not one of them," Dr. Barbara Casadei said in presenting the findings of the Statin Therapy in Cardiac Surgery (STICS) trial at the annual congress of the European Society of Cardiology.
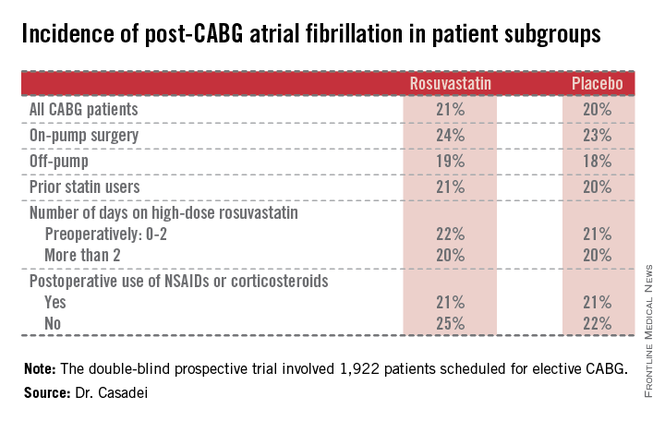
The STICS results are at odds with conventional wisdom. ESC guidelines give a favorable class IIa, level of evidence B recommendation that "statins should be considered for prevention of new-onset atrial fibrillation after coronary artery bypass grafting, either isolated or in combination with valvular interventions."
"STICS was a very carefully conducted, large scale, robust study that I think has definitely closed the door on this issue," commented Dr. Keith A.A. Fox, professor of cardiology at the University of Edinburgh and chair of the scientific and clinical program committee at ESC Congress 2014.
STICS was a double-blind prospective trial in which 1,922 patients scheduled for elective CABG were randomized to 20 mg per day of rosuvastatin (Crestor) or placebo starting up to 8 days prior to surgery and continued for 5 days postop. All participants were in sinus rhythm preoperatively, with no history of AF, said Dr. Casadei, professor of cardiovascular medicine at the University of Oxford, England.
The two coprimary endpoints in STICS were the incidence of new-onset AF during 5 days of postop Holter monitoring, and evidence of postop myocardial injury as demonstrated in serial troponin I assays.
Postop AF occurred in 21% of those given high-intensity therapy with rosuvastatin and 20% of placebo-treated controls. There was no subgroup where rosuvastatin was protective (see graphic).
Troponin I measurements obtained 6, 24, 48, and 120 hours postop showed areas under the curve that were superimposable in the two study groups, meaning perioperative high-dose statin therapy provided absolutely no protection against postop cardiac muscle injury.
Mean hospital length of stay and ICU time didn’t differ between the two groups, either.
The impetus for conducting STICS was recognition that the guidelines’ endorsement of perioperative high-dose statin therapy in conjunction with cardiac surgery was based upon a series of small randomized trials with serious limitations. Although the results of a meta-analysis of the 14 prior trials looked impressive at first glance – a 17% incidence of postop AF in statin-treated patients, compared with 30% in controls, for a near-halving of the risk of this important complication – these 14 studies totaled 1,300 patients, and there were many methodologic shortcomings.
The STICS researchers hypothesized that a large, well-designed trial – bigger than all previous studies combined – would shore up the previously shaky supporting evidence and perhaps provide grounds for statins to win a new indication from regulatory agencies. Post-CABG AF is associated with a doubled risk of stroke and mortality, and excess hospital costs of $8,000-$18,000 dollars per patient.
Discussant Dr. Paulus Kirchhof, a member of the task force that developed the current ESC guidelines (Europace 2010;12:1360-420), said those guidelines now clearly need to be revisited. Beyond that, he added, STICS provides important new contributions in understanding the pathophysiology of AF.
"We know that AF is caused by several vicious circles, and we believe that inflammation could influence those and cause AF. And we also thought that postop AF was the condition where inflammation plays the biggest role. Based upon the negative results with this anti-inflammatory intervention, I think we have to question this concept a bit," said Dr. Kirchhof, professor of cardiovascular sciences at the University of Birmingham, England.
Dr. Casadei countered that she’s not ready to write off postop inflammation entirely as a major trigger of new-onset AF following CABG.
"The inflammation is there. We know from experimental work in animals that there is a strong association between inflammation and postop atrial fibrillation, but whether the association is causal, I think, is still debated. However, it may be that the anti-inflammatory effect of statins is not sufficiently strong to actually prevent this complication," she said.
Discussant Dr. Steven Nissen praised STICS as "an outstanding trial."
"I also think there’s a terribly important lesson here, which is the power of self-delusion in medicine. When we base our guidelines on small, poorly controlled trials, we are often making mistakes. This is one of countless examples where when someone finally does a careful, thoughtful trial, we find out that something that people believe just isn’t true. We can’t cut corners with evidence. We need good randomized trials," declared Dr. Nissen, chair of the department of cardiovascular medicine at the Cleveland Clinic.
The STICS trial was funded primarily by the British Heart Foundation, the Oxford Biomedical Research Center, and the UK Medical Research Council. In addition, Dr. Casadei reported receiving an unrestricted grant from AstraZeneca in conjunction with the trial.

|
| Dr. Hiren Shah |
There are two key lessons from the results of the STICS trial. First, extrapolation of results from biochemical pathways and measured cellular markers does not always translate into meaningful clinical outcomes. Thus, it has long been known from several large trials that statin therapy effectively and rapidly lowers CRP levels both in hyper- and normocholesterolemic patients and that statins are effective in decreasing systemic inflammation. It has also been known that inflammation contributes to the development and maintenance of AF, so it was postulated that by improving endothelial nitric oxide availability, reducing inflammation, and decreasing oxidative stress, and through neurohormonal activation, statins would reduce the incidence of post-op AF. This link was so strong that clinical guidelines adopted limited data from small trials to make treatment recommendations.
This leads us to consider the second key lesson from this study. Trials with small sample size, even when combined across many other trials (1,300 patients were involved across 14 trials in this case), do not always yield reliable results, especially when they have significant limitations, notably not always being blind and having been performed in statin-naive patients only. The large, randomized, and well-designed STICS trial puts to rest an important issue, given the high prevalence of AF after cardiac surgery, which is associated with a longer length of stay, an increased risk of stroke, higher mortality, and greater costs, and should prompt us to consider further evaluation of different strategies to reduce this significant complication.
Dr. Hiren Shah is medical director of the medicine and cardiac telemetry hospitalist unit at Northwestern Memorial Hospital in Chicago and an adviser to Hospitalist News. He is the national chair of the Clinician Committee for ACP’s Initiative on Stroke Prevention and Atrial Fibrillation and is the lead physician for the Society of Hospital Medicine’s National Atrial Fibrillation Initiative.

|
| Dr. Hiren Shah |
There are two key lessons from the results of the STICS trial. First, extrapolation of results from biochemical pathways and measured cellular markers does not always translate into meaningful clinical outcomes. Thus, it has long been known from several large trials that statin therapy effectively and rapidly lowers CRP levels both in hyper- and normocholesterolemic patients and that statins are effective in decreasing systemic inflammation. It has also been known that inflammation contributes to the development and maintenance of AF, so it was postulated that by improving endothelial nitric oxide availability, reducing inflammation, and decreasing oxidative stress, and through neurohormonal activation, statins would reduce the incidence of post-op AF. This link was so strong that clinical guidelines adopted limited data from small trials to make treatment recommendations.
This leads us to consider the second key lesson from this study. Trials with small sample size, even when combined across many other trials (1,300 patients were involved across 14 trials in this case), do not always yield reliable results, especially when they have significant limitations, notably not always being blind and having been performed in statin-naive patients only. The large, randomized, and well-designed STICS trial puts to rest an important issue, given the high prevalence of AF after cardiac surgery, which is associated with a longer length of stay, an increased risk of stroke, higher mortality, and greater costs, and should prompt us to consider further evaluation of different strategies to reduce this significant complication.
Dr. Hiren Shah is medical director of the medicine and cardiac telemetry hospitalist unit at Northwestern Memorial Hospital in Chicago and an adviser to Hospitalist News. He is the national chair of the Clinician Committee for ACP’s Initiative on Stroke Prevention and Atrial Fibrillation and is the lead physician for the Society of Hospital Medicine’s National Atrial Fibrillation Initiative.

|
| Dr. Hiren Shah |
There are two key lessons from the results of the STICS trial. First, extrapolation of results from biochemical pathways and measured cellular markers does not always translate into meaningful clinical outcomes. Thus, it has long been known from several large trials that statin therapy effectively and rapidly lowers CRP levels both in hyper- and normocholesterolemic patients and that statins are effective in decreasing systemic inflammation. It has also been known that inflammation contributes to the development and maintenance of AF, so it was postulated that by improving endothelial nitric oxide availability, reducing inflammation, and decreasing oxidative stress, and through neurohormonal activation, statins would reduce the incidence of post-op AF. This link was so strong that clinical guidelines adopted limited data from small trials to make treatment recommendations.
This leads us to consider the second key lesson from this study. Trials with small sample size, even when combined across many other trials (1,300 patients were involved across 14 trials in this case), do not always yield reliable results, especially when they have significant limitations, notably not always being blind and having been performed in statin-naive patients only. The large, randomized, and well-designed STICS trial puts to rest an important issue, given the high prevalence of AF after cardiac surgery, which is associated with a longer length of stay, an increased risk of stroke, higher mortality, and greater costs, and should prompt us to consider further evaluation of different strategies to reduce this significant complication.
Dr. Hiren Shah is medical director of the medicine and cardiac telemetry hospitalist unit at Northwestern Memorial Hospital in Chicago and an adviser to Hospitalist News. He is the national chair of the Clinician Committee for ACP’s Initiative on Stroke Prevention and Atrial Fibrillation and is the lead physician for the Society of Hospital Medicine’s National Atrial Fibrillation Initiative.
BARCELONA – Intensive perioperative statin therapy in patients undergoing CABG surgery doesn’t protect against postop atrial fibrillation or myocardial injury, according to a large randomized clinical trial hailed as the "definitive" study addressing this issue.
"There are many reasons why these patients should be put on statin treatment, but the prevention of postop complications is not one of them," Dr. Barbara Casadei said in presenting the findings of the Statin Therapy in Cardiac Surgery (STICS) trial at the annual congress of the European Society of Cardiology.

The STICS results are at odds with conventional wisdom. ESC guidelines give a favorable class IIa, level of evidence B recommendation that "statins should be considered for prevention of new-onset atrial fibrillation after coronary artery bypass grafting, either isolated or in combination with valvular interventions."
"STICS was a very carefully conducted, large scale, robust study that I think has definitely closed the door on this issue," commented Dr. Keith A.A. Fox, professor of cardiology at the University of Edinburgh and chair of the scientific and clinical program committee at ESC Congress 2014.
STICS was a double-blind prospective trial in which 1,922 patients scheduled for elective CABG were randomized to 20 mg per day of rosuvastatin (Crestor) or placebo starting up to 8 days prior to surgery and continued for 5 days postop. All participants were in sinus rhythm preoperatively, with no history of AF, said Dr. Casadei, professor of cardiovascular medicine at the University of Oxford, England.
The two coprimary endpoints in STICS were the incidence of new-onset AF during 5 days of postop Holter monitoring, and evidence of postop myocardial injury as demonstrated in serial troponin I assays.
Postop AF occurred in 21% of those given high-intensity therapy with rosuvastatin and 20% of placebo-treated controls. There was no subgroup where rosuvastatin was protective (see graphic).
Troponin I measurements obtained 6, 24, 48, and 120 hours postop showed areas under the curve that were superimposable in the two study groups, meaning perioperative high-dose statin therapy provided absolutely no protection against postop cardiac muscle injury.
Mean hospital length of stay and ICU time didn’t differ between the two groups, either.
The impetus for conducting STICS was recognition that the guidelines’ endorsement of perioperative high-dose statin therapy in conjunction with cardiac surgery was based upon a series of small randomized trials with serious limitations. Although the results of a meta-analysis of the 14 prior trials looked impressive at first glance – a 17% incidence of postop AF in statin-treated patients, compared with 30% in controls, for a near-halving of the risk of this important complication – these 14 studies totaled 1,300 patients, and there were many methodologic shortcomings.
The STICS researchers hypothesized that a large, well-designed trial – bigger than all previous studies combined – would shore up the previously shaky supporting evidence and perhaps provide grounds for statins to win a new indication from regulatory agencies. Post-CABG AF is associated with a doubled risk of stroke and mortality, and excess hospital costs of $8,000-$18,000 dollars per patient.
Discussant Dr. Paulus Kirchhof, a member of the task force that developed the current ESC guidelines (Europace 2010;12:1360-420), said those guidelines now clearly need to be revisited. Beyond that, he added, STICS provides important new contributions in understanding the pathophysiology of AF.
"We know that AF is caused by several vicious circles, and we believe that inflammation could influence those and cause AF. And we also thought that postop AF was the condition where inflammation plays the biggest role. Based upon the negative results with this anti-inflammatory intervention, I think we have to question this concept a bit," said Dr. Kirchhof, professor of cardiovascular sciences at the University of Birmingham, England.
Dr. Casadei countered that she’s not ready to write off postop inflammation entirely as a major trigger of new-onset AF following CABG.
"The inflammation is there. We know from experimental work in animals that there is a strong association between inflammation and postop atrial fibrillation, but whether the association is causal, I think, is still debated. However, it may be that the anti-inflammatory effect of statins is not sufficiently strong to actually prevent this complication," she said.
Discussant Dr. Steven Nissen praised STICS as "an outstanding trial."
"I also think there’s a terribly important lesson here, which is the power of self-delusion in medicine. When we base our guidelines on small, poorly controlled trials, we are often making mistakes. This is one of countless examples where when someone finally does a careful, thoughtful trial, we find out that something that people believe just isn’t true. We can’t cut corners with evidence. We need good randomized trials," declared Dr. Nissen, chair of the department of cardiovascular medicine at the Cleveland Clinic.
The STICS trial was funded primarily by the British Heart Foundation, the Oxford Biomedical Research Center, and the UK Medical Research Council. In addition, Dr. Casadei reported receiving an unrestricted grant from AstraZeneca in conjunction with the trial.
BARCELONA – Intensive perioperative statin therapy in patients undergoing CABG surgery doesn’t protect against postop atrial fibrillation or myocardial injury, according to a large randomized clinical trial hailed as the "definitive" study addressing this issue.
"There are many reasons why these patients should be put on statin treatment, but the prevention of postop complications is not one of them," Dr. Barbara Casadei said in presenting the findings of the Statin Therapy in Cardiac Surgery (STICS) trial at the annual congress of the European Society of Cardiology.

The STICS results are at odds with conventional wisdom. ESC guidelines give a favorable class IIa, level of evidence B recommendation that "statins should be considered for prevention of new-onset atrial fibrillation after coronary artery bypass grafting, either isolated or in combination with valvular interventions."
"STICS was a very carefully conducted, large scale, robust study that I think has definitely closed the door on this issue," commented Dr. Keith A.A. Fox, professor of cardiology at the University of Edinburgh and chair of the scientific and clinical program committee at ESC Congress 2014.
STICS was a double-blind prospective trial in which 1,922 patients scheduled for elective CABG were randomized to 20 mg per day of rosuvastatin (Crestor) or placebo starting up to 8 days prior to surgery and continued for 5 days postop. All participants were in sinus rhythm preoperatively, with no history of AF, said Dr. Casadei, professor of cardiovascular medicine at the University of Oxford, England.
The two coprimary endpoints in STICS were the incidence of new-onset AF during 5 days of postop Holter monitoring, and evidence of postop myocardial injury as demonstrated in serial troponin I assays.
Postop AF occurred in 21% of those given high-intensity therapy with rosuvastatin and 20% of placebo-treated controls. There was no subgroup where rosuvastatin was protective (see graphic).
Troponin I measurements obtained 6, 24, 48, and 120 hours postop showed areas under the curve that were superimposable in the two study groups, meaning perioperative high-dose statin therapy provided absolutely no protection against postop cardiac muscle injury.
Mean hospital length of stay and ICU time didn’t differ between the two groups, either.
The impetus for conducting STICS was recognition that the guidelines’ endorsement of perioperative high-dose statin therapy in conjunction with cardiac surgery was based upon a series of small randomized trials with serious limitations. Although the results of a meta-analysis of the 14 prior trials looked impressive at first glance – a 17% incidence of postop AF in statin-treated patients, compared with 30% in controls, for a near-halving of the risk of this important complication – these 14 studies totaled 1,300 patients, and there were many methodologic shortcomings.
The STICS researchers hypothesized that a large, well-designed trial – bigger than all previous studies combined – would shore up the previously shaky supporting evidence and perhaps provide grounds for statins to win a new indication from regulatory agencies. Post-CABG AF is associated with a doubled risk of stroke and mortality, and excess hospital costs of $8,000-$18,000 dollars per patient.
Discussant Dr. Paulus Kirchhof, a member of the task force that developed the current ESC guidelines (Europace 2010;12:1360-420), said those guidelines now clearly need to be revisited. Beyond that, he added, STICS provides important new contributions in understanding the pathophysiology of AF.
"We know that AF is caused by several vicious circles, and we believe that inflammation could influence those and cause AF. And we also thought that postop AF was the condition where inflammation plays the biggest role. Based upon the negative results with this anti-inflammatory intervention, I think we have to question this concept a bit," said Dr. Kirchhof, professor of cardiovascular sciences at the University of Birmingham, England.
Dr. Casadei countered that she’s not ready to write off postop inflammation entirely as a major trigger of new-onset AF following CABG.
"The inflammation is there. We know from experimental work in animals that there is a strong association between inflammation and postop atrial fibrillation, but whether the association is causal, I think, is still debated. However, it may be that the anti-inflammatory effect of statins is not sufficiently strong to actually prevent this complication," she said.
Discussant Dr. Steven Nissen praised STICS as "an outstanding trial."
"I also think there’s a terribly important lesson here, which is the power of self-delusion in medicine. When we base our guidelines on small, poorly controlled trials, we are often making mistakes. This is one of countless examples where when someone finally does a careful, thoughtful trial, we find out that something that people believe just isn’t true. We can’t cut corners with evidence. We need good randomized trials," declared Dr. Nissen, chair of the department of cardiovascular medicine at the Cleveland Clinic.
The STICS trial was funded primarily by the British Heart Foundation, the Oxford Biomedical Research Center, and the UK Medical Research Council. In addition, Dr. Casadei reported receiving an unrestricted grant from AstraZeneca in conjunction with the trial.
AT THE ESC CONGRESS 2014
Key clinical point: Perioperative statin therapy in patients undergoing CABG failed to protect against new-onset postop atrial fibrillation.
Major finding: The incidence of postop atrial fibrillation within 5 days post-CABG was 21% in patients randomized to 20 mg/day of rosuvastatin and 20% in placebo-treated controls.
Data source: The multicenter STICS trial included 1,922 randomized patients scheduled for elective CABG.
Disclosures: STICS was funded by the British Heart Foundation, the Oxford Biomedical Research Center, and the UK Medical Research Council. The presenter reported having received a research grant from AstraZeneca.
Stroke risk skyrocketed after intracranial hemorrhage in warfarin users
BARCELONA – Patients who have an intracranial hemorrhage while on warfarin for atrial fibrillation are at sharply increased risk for an ischemic stroke during the following year, a period when many of them are off warfarin, according to a Danish national study.
The absolute risk of a first ischemic stroke among survivors of an intracranial hemorrhage (ICH) while on warfarin was roughly 10% within the first month, 20% within 3 months, and 32% at 1 year. These rates exclude strokes that occurred within the first 7 days after an ICH, Dr. Peter B. Nielsen reported at the annual congress of the European Society of Cardiology.
The design of this real-world national registry study doesn’t permit definitive conclusions to be drawn regarding causality. It is telling, however, that the rate of warfarin use among Danish atrial fibrillation (AF) patients in the year after an ICH plunged by 72%; thus, only about one in four of patients who regularly filled warfarin prescriptions up until the time of their ICH did so afterward, according to Dr. Nielsen of Aalborg (Denmark) University.
He reported on 58,815 warfarin-treated Danes with AF with no prior ICH, ischemic stroke, transient ischemic attack, or systemic embolism at baseline who were followed for up to 13 years, during which 1,639 of them were diagnosed with ICH.
During 261,681 person-years of follow-up of the cohort that remained free of ICH, 6,843 patients were diagnosed with a first ischemic stroke, transient ischemic attack, or systemic embolism. Among the group who had an ICH, that rate was 3.67-fold greater.
Moreover, all-cause mortality occurred in 946 patients with an ICH during 2,404 person-years of follow-up, a rate 5.6-fold greater than in those who remained on warfarin and free of ICH.
When the analysis was restricted to only 2 years of follow-up, the event rate ratios became even more dramatic: AF patients with an ICH were at subsequent 5.4- and 11.8-fold greater risks of ischemic stroke and all-cause mortality, respectively, within the next 2 years, compared with those who were free of ICH.
The optimal timing of resumption of warfarin following an ICH is unclear. Various small studies have come up with recommendations ranging from 1 to 30 weeks, Dr. Nielsen noted.
"Caution may lead to undertreatment and increased risk of ischemic stroke and thrombotic events," he noted. "The question now in my mind is, Who, having survived an ICH, can benefit from a return to warfarin or a switch to a novel oral anticoagulant?"
Discussant Dr. Christopher B. Granger, commenting on the Danish study results, said, "I think this is very important information that there’s this extraordinary risk of ischemic stroke in the year after ICH. I don’t think this has been described before. It of course puts us in a tough bind because we don’t know the safety of returning to warfarin use. This raises the issue in my mind of whether there might be a role for a left atrial appendage occlusion device as an option in this population that’s presumably going to be at high risk of recurrent ICH upon resumption of oral anticoagulation," said Dr. Granger, professor of medicine and director of the cardiac care unit at Duke University Medical Center in Durham, N.C.
When it was pointed out that 3 months of oral anticoagulant therapy is required after placement of the Watchman, a left atrial appendage occlusion device, he responded, "Yes, at least the way it has been developed so far – but this might be an opportunity to test an alternative approach."
Dr. Nielsen and Dr. Granger reported having no relevant financial conflicts.
BARCELONA – Patients who have an intracranial hemorrhage while on warfarin for atrial fibrillation are at sharply increased risk for an ischemic stroke during the following year, a period when many of them are off warfarin, according to a Danish national study.
The absolute risk of a first ischemic stroke among survivors of an intracranial hemorrhage (ICH) while on warfarin was roughly 10% within the first month, 20% within 3 months, and 32% at 1 year. These rates exclude strokes that occurred within the first 7 days after an ICH, Dr. Peter B. Nielsen reported at the annual congress of the European Society of Cardiology.
The design of this real-world national registry study doesn’t permit definitive conclusions to be drawn regarding causality. It is telling, however, that the rate of warfarin use among Danish atrial fibrillation (AF) patients in the year after an ICH plunged by 72%; thus, only about one in four of patients who regularly filled warfarin prescriptions up until the time of their ICH did so afterward, according to Dr. Nielsen of Aalborg (Denmark) University.
He reported on 58,815 warfarin-treated Danes with AF with no prior ICH, ischemic stroke, transient ischemic attack, or systemic embolism at baseline who were followed for up to 13 years, during which 1,639 of them were diagnosed with ICH.
During 261,681 person-years of follow-up of the cohort that remained free of ICH, 6,843 patients were diagnosed with a first ischemic stroke, transient ischemic attack, or systemic embolism. Among the group who had an ICH, that rate was 3.67-fold greater.
Moreover, all-cause mortality occurred in 946 patients with an ICH during 2,404 person-years of follow-up, a rate 5.6-fold greater than in those who remained on warfarin and free of ICH.
When the analysis was restricted to only 2 years of follow-up, the event rate ratios became even more dramatic: AF patients with an ICH were at subsequent 5.4- and 11.8-fold greater risks of ischemic stroke and all-cause mortality, respectively, within the next 2 years, compared with those who were free of ICH.
The optimal timing of resumption of warfarin following an ICH is unclear. Various small studies have come up with recommendations ranging from 1 to 30 weeks, Dr. Nielsen noted.
"Caution may lead to undertreatment and increased risk of ischemic stroke and thrombotic events," he noted. "The question now in my mind is, Who, having survived an ICH, can benefit from a return to warfarin or a switch to a novel oral anticoagulant?"
Discussant Dr. Christopher B. Granger, commenting on the Danish study results, said, "I think this is very important information that there’s this extraordinary risk of ischemic stroke in the year after ICH. I don’t think this has been described before. It of course puts us in a tough bind because we don’t know the safety of returning to warfarin use. This raises the issue in my mind of whether there might be a role for a left atrial appendage occlusion device as an option in this population that’s presumably going to be at high risk of recurrent ICH upon resumption of oral anticoagulation," said Dr. Granger, professor of medicine and director of the cardiac care unit at Duke University Medical Center in Durham, N.C.
When it was pointed out that 3 months of oral anticoagulant therapy is required after placement of the Watchman, a left atrial appendage occlusion device, he responded, "Yes, at least the way it has been developed so far – but this might be an opportunity to test an alternative approach."
Dr. Nielsen and Dr. Granger reported having no relevant financial conflicts.
BARCELONA – Patients who have an intracranial hemorrhage while on warfarin for atrial fibrillation are at sharply increased risk for an ischemic stroke during the following year, a period when many of them are off warfarin, according to a Danish national study.
The absolute risk of a first ischemic stroke among survivors of an intracranial hemorrhage (ICH) while on warfarin was roughly 10% within the first month, 20% within 3 months, and 32% at 1 year. These rates exclude strokes that occurred within the first 7 days after an ICH, Dr. Peter B. Nielsen reported at the annual congress of the European Society of Cardiology.
The design of this real-world national registry study doesn’t permit definitive conclusions to be drawn regarding causality. It is telling, however, that the rate of warfarin use among Danish atrial fibrillation (AF) patients in the year after an ICH plunged by 72%; thus, only about one in four of patients who regularly filled warfarin prescriptions up until the time of their ICH did so afterward, according to Dr. Nielsen of Aalborg (Denmark) University.
He reported on 58,815 warfarin-treated Danes with AF with no prior ICH, ischemic stroke, transient ischemic attack, or systemic embolism at baseline who were followed for up to 13 years, during which 1,639 of them were diagnosed with ICH.
During 261,681 person-years of follow-up of the cohort that remained free of ICH, 6,843 patients were diagnosed with a first ischemic stroke, transient ischemic attack, or systemic embolism. Among the group who had an ICH, that rate was 3.67-fold greater.
Moreover, all-cause mortality occurred in 946 patients with an ICH during 2,404 person-years of follow-up, a rate 5.6-fold greater than in those who remained on warfarin and free of ICH.
When the analysis was restricted to only 2 years of follow-up, the event rate ratios became even more dramatic: AF patients with an ICH were at subsequent 5.4- and 11.8-fold greater risks of ischemic stroke and all-cause mortality, respectively, within the next 2 years, compared with those who were free of ICH.
The optimal timing of resumption of warfarin following an ICH is unclear. Various small studies have come up with recommendations ranging from 1 to 30 weeks, Dr. Nielsen noted.
"Caution may lead to undertreatment and increased risk of ischemic stroke and thrombotic events," he noted. "The question now in my mind is, Who, having survived an ICH, can benefit from a return to warfarin or a switch to a novel oral anticoagulant?"
Discussant Dr. Christopher B. Granger, commenting on the Danish study results, said, "I think this is very important information that there’s this extraordinary risk of ischemic stroke in the year after ICH. I don’t think this has been described before. It of course puts us in a tough bind because we don’t know the safety of returning to warfarin use. This raises the issue in my mind of whether there might be a role for a left atrial appendage occlusion device as an option in this population that’s presumably going to be at high risk of recurrent ICH upon resumption of oral anticoagulation," said Dr. Granger, professor of medicine and director of the cardiac care unit at Duke University Medical Center in Durham, N.C.
When it was pointed out that 3 months of oral anticoagulant therapy is required after placement of the Watchman, a left atrial appendage occlusion device, he responded, "Yes, at least the way it has been developed so far – but this might be an opportunity to test an alternative approach."
Dr. Nielsen and Dr. Granger reported having no relevant financial conflicts.
AT THE ESC CONGRESS 2014
Key clinical point: Taking patients with atrial fibrillation off warfarin, even temporarily, after an intracranial hemorrhage comes at a high cost in terms of the subsequent rate of ischemic stroke.
Major finding: The incidence of a first ischemic stroke was 32% during the first year after patients on warfarin for atrial fibrillation had an intracranial hemorrhage.
Data source: A Danish national registry study that included nearly 59,000 patients on warfarin for atrial fibrillation who were followed for up to 13 years.
Disclosures: The study was supported by institutional funds. The presenter reported having no relevant financial conflicts.
Pneumococcal Vaccine Protects Against Cardiac and Cerebrovascular Events
BARCELONA – Influenza vaccine has been shown to provide protection against cardiovascular events, but can the same be said for pneumococcal vaccine?
Yes, particularly in the elderly and in patients at high baseline cardiovascular risk, according to a meta-analysis presented by Dr. Dimitrios Terentes-Printzios at the annual congress of the European Society of Cardiology.
He analyzed 11 published studies comprising 332,267 subjects followed for a mean of 20 months. Because the studies focused on different populations and in some cases reached conflicting conclusions, he performed a series of subgroup analyses to gain a clearer picture.
One of these analyses found that the cardioprotective effects of pneumococcal vaccination wane over time. In studies with follow-up of less than 1 year, the relative risk of total cardiovascular events was 0.72, meaning that patients who received pneumococcal vaccine had a significant 28% relative risk reduction compared with those who did not. In studies with follow-up in excess of 1 year, however, there was no cardioprotective effect, according to Dr. Terentes-Printzios of Athens Medical School.
Significant protection against total cardiovascular events was seen in elderly vaccinated patients, with a 20% relative risk reduction, and in subjects at high baseline cardiovascular risk, who had an 8% risk reduction if they received pneumococcal vaccine.
Breaking down the specific endpoints, subjects who got pneumococcal vaccine had a statistically significant 8% reduction in the risk of cardiovascular mortality. However, vaccination provided no significant protective effect against acute MI or cerebrovascular events except in the elderly, where the relative risk reductions were 10% and 14%, respectively.
These cardio- and cerebrovascular protective benefits of the pneumococcal vaccine can be viewed as added value, given that the primary reason physicians prescribe the vaccine is its demonstrated ability to reduce the risk of invasive pneumococcal infection by up to 60%.
Dr. Terentes-Printzios reported having no financial conflicts.
BARCELONA – Influenza vaccine has been shown to provide protection against cardiovascular events, but can the same be said for pneumococcal vaccine?
Yes, particularly in the elderly and in patients at high baseline cardiovascular risk, according to a meta-analysis presented by Dr. Dimitrios Terentes-Printzios at the annual congress of the European Society of Cardiology.
He analyzed 11 published studies comprising 332,267 subjects followed for a mean of 20 months. Because the studies focused on different populations and in some cases reached conflicting conclusions, he performed a series of subgroup analyses to gain a clearer picture.
One of these analyses found that the cardioprotective effects of pneumococcal vaccination wane over time. In studies with follow-up of less than 1 year, the relative risk of total cardiovascular events was 0.72, meaning that patients who received pneumococcal vaccine had a significant 28% relative risk reduction compared with those who did not. In studies with follow-up in excess of 1 year, however, there was no cardioprotective effect, according to Dr. Terentes-Printzios of Athens Medical School.
Significant protection against total cardiovascular events was seen in elderly vaccinated patients, with a 20% relative risk reduction, and in subjects at high baseline cardiovascular risk, who had an 8% risk reduction if they received pneumococcal vaccine.
Breaking down the specific endpoints, subjects who got pneumococcal vaccine had a statistically significant 8% reduction in the risk of cardiovascular mortality. However, vaccination provided no significant protective effect against acute MI or cerebrovascular events except in the elderly, where the relative risk reductions were 10% and 14%, respectively.
These cardio- and cerebrovascular protective benefits of the pneumococcal vaccine can be viewed as added value, given that the primary reason physicians prescribe the vaccine is its demonstrated ability to reduce the risk of invasive pneumococcal infection by up to 60%.
Dr. Terentes-Printzios reported having no financial conflicts.
BARCELONA – Influenza vaccine has been shown to provide protection against cardiovascular events, but can the same be said for pneumococcal vaccine?
Yes, particularly in the elderly and in patients at high baseline cardiovascular risk, according to a meta-analysis presented by Dr. Dimitrios Terentes-Printzios at the annual congress of the European Society of Cardiology.
He analyzed 11 published studies comprising 332,267 subjects followed for a mean of 20 months. Because the studies focused on different populations and in some cases reached conflicting conclusions, he performed a series of subgroup analyses to gain a clearer picture.
One of these analyses found that the cardioprotective effects of pneumococcal vaccination wane over time. In studies with follow-up of less than 1 year, the relative risk of total cardiovascular events was 0.72, meaning that patients who received pneumococcal vaccine had a significant 28% relative risk reduction compared with those who did not. In studies with follow-up in excess of 1 year, however, there was no cardioprotective effect, according to Dr. Terentes-Printzios of Athens Medical School.
Significant protection against total cardiovascular events was seen in elderly vaccinated patients, with a 20% relative risk reduction, and in subjects at high baseline cardiovascular risk, who had an 8% risk reduction if they received pneumococcal vaccine.
Breaking down the specific endpoints, subjects who got pneumococcal vaccine had a statistically significant 8% reduction in the risk of cardiovascular mortality. However, vaccination provided no significant protective effect against acute MI or cerebrovascular events except in the elderly, where the relative risk reductions were 10% and 14%, respectively.
These cardio- and cerebrovascular protective benefits of the pneumococcal vaccine can be viewed as added value, given that the primary reason physicians prescribe the vaccine is its demonstrated ability to reduce the risk of invasive pneumococcal infection by up to 60%.
Dr. Terentes-Printzios reported having no financial conflicts.
AT THE ESC CONGRESS 2014
Pneumococcal vaccine protects against cardiac and cerebrovascular events
BARCELONA – Influenza vaccine has been shown to provide protection against cardiovascular events, but can the same be said for pneumococcal vaccine?
Yes, particularly in the elderly and in patients at high baseline cardiovascular risk, according to a meta-analysis presented by Dr. Dimitrios Terentes-Printzios at the annual congress of the European Society of Cardiology.
He analyzed 11 published studies comprising 332,267 subjects followed for a mean of 20 months. Because the studies focused on different populations and in some cases reached conflicting conclusions, he performed a series of subgroup analyses to gain a clearer picture.
One of these analyses found that the cardioprotective effects of pneumococcal vaccination wane over time. In studies with follow-up of less than 1 year, the relative risk of total cardiovascular events was 0.72, meaning that patients who received pneumococcal vaccine had a significant 28% relative risk reduction compared with those who did not. In studies with follow-up in excess of 1 year, however, there was no cardioprotective effect, according to Dr. Terentes-Printzios of Athens Medical School.
Significant protection against total cardiovascular events was seen in elderly vaccinated patients, with a 20% relative risk reduction, and in subjects at high baseline cardiovascular risk, who had an 8% risk reduction if they received pneumococcal vaccine.
Breaking down the specific endpoints, subjects who got pneumococcal vaccine had a statistically significant 8% reduction in the risk of cardiovascular mortality. However, vaccination provided no significant protective effect against acute MI or cerebrovascular events except in the elderly, where the relative risk reductions were 10% and 14%, respectively.
These cardio- and cerebrovascular protective benefits of the pneumococcal vaccine can be viewed as added value, given that the primary reason physicians prescribe the vaccine is its demonstrated ability to reduce the risk of invasive pneumococcal infection by up to 60%.
Dr. Terentes-Printzios reported having no financial conflicts.
Many patients with pneumonia are known to be at increased risk for cardiovascular events. The American Heart Association’s Get With The Guidelines – Resuscitation database found that in many stroke and cardiopulmonary arrests, pneumonia was a preexisting condition or was acquired during hospitalization.
Hence, it’s expected that preventing pneumonia with vaccination will also prevent subsequent death from cardiovascular reasons. Pneumonia vaccination will prevent not only cardiovascular events but death in general.
This study found more compelling reasons for us in the front line to vaccinate eligible candidates for flu and pneumonia, especially with fall already here.
Many patients with pneumonia are known to be at increased risk for cardiovascular events. The American Heart Association’s Get With The Guidelines – Resuscitation database found that in many stroke and cardiopulmonary arrests, pneumonia was a preexisting condition or was acquired during hospitalization.
Hence, it’s expected that preventing pneumonia with vaccination will also prevent subsequent death from cardiovascular reasons. Pneumonia vaccination will prevent not only cardiovascular events but death in general.
This study found more compelling reasons for us in the front line to vaccinate eligible candidates for flu and pneumonia, especially with fall already here.
Many patients with pneumonia are known to be at increased risk for cardiovascular events. The American Heart Association’s Get With The Guidelines – Resuscitation database found that in many stroke and cardiopulmonary arrests, pneumonia was a preexisting condition or was acquired during hospitalization.
Hence, it’s expected that preventing pneumonia with vaccination will also prevent subsequent death from cardiovascular reasons. Pneumonia vaccination will prevent not only cardiovascular events but death in general.
This study found more compelling reasons for us in the front line to vaccinate eligible candidates for flu and pneumonia, especially with fall already here.
BARCELONA – Influenza vaccine has been shown to provide protection against cardiovascular events, but can the same be said for pneumococcal vaccine?
Yes, particularly in the elderly and in patients at high baseline cardiovascular risk, according to a meta-analysis presented by Dr. Dimitrios Terentes-Printzios at the annual congress of the European Society of Cardiology.
He analyzed 11 published studies comprising 332,267 subjects followed for a mean of 20 months. Because the studies focused on different populations and in some cases reached conflicting conclusions, he performed a series of subgroup analyses to gain a clearer picture.
One of these analyses found that the cardioprotective effects of pneumococcal vaccination wane over time. In studies with follow-up of less than 1 year, the relative risk of total cardiovascular events was 0.72, meaning that patients who received pneumococcal vaccine had a significant 28% relative risk reduction compared with those who did not. In studies with follow-up in excess of 1 year, however, there was no cardioprotective effect, according to Dr. Terentes-Printzios of Athens Medical School.
Significant protection against total cardiovascular events was seen in elderly vaccinated patients, with a 20% relative risk reduction, and in subjects at high baseline cardiovascular risk, who had an 8% risk reduction if they received pneumococcal vaccine.
Breaking down the specific endpoints, subjects who got pneumococcal vaccine had a statistically significant 8% reduction in the risk of cardiovascular mortality. However, vaccination provided no significant protective effect against acute MI or cerebrovascular events except in the elderly, where the relative risk reductions were 10% and 14%, respectively.
These cardio- and cerebrovascular protective benefits of the pneumococcal vaccine can be viewed as added value, given that the primary reason physicians prescribe the vaccine is its demonstrated ability to reduce the risk of invasive pneumococcal infection by up to 60%.
Dr. Terentes-Printzios reported having no financial conflicts.
BARCELONA – Influenza vaccine has been shown to provide protection against cardiovascular events, but can the same be said for pneumococcal vaccine?
Yes, particularly in the elderly and in patients at high baseline cardiovascular risk, according to a meta-analysis presented by Dr. Dimitrios Terentes-Printzios at the annual congress of the European Society of Cardiology.
He analyzed 11 published studies comprising 332,267 subjects followed for a mean of 20 months. Because the studies focused on different populations and in some cases reached conflicting conclusions, he performed a series of subgroup analyses to gain a clearer picture.
One of these analyses found that the cardioprotective effects of pneumococcal vaccination wane over time. In studies with follow-up of less than 1 year, the relative risk of total cardiovascular events was 0.72, meaning that patients who received pneumococcal vaccine had a significant 28% relative risk reduction compared with those who did not. In studies with follow-up in excess of 1 year, however, there was no cardioprotective effect, according to Dr. Terentes-Printzios of Athens Medical School.
Significant protection against total cardiovascular events was seen in elderly vaccinated patients, with a 20% relative risk reduction, and in subjects at high baseline cardiovascular risk, who had an 8% risk reduction if they received pneumococcal vaccine.
Breaking down the specific endpoints, subjects who got pneumococcal vaccine had a statistically significant 8% reduction in the risk of cardiovascular mortality. However, vaccination provided no significant protective effect against acute MI or cerebrovascular events except in the elderly, where the relative risk reductions were 10% and 14%, respectively.
These cardio- and cerebrovascular protective benefits of the pneumococcal vaccine can be viewed as added value, given that the primary reason physicians prescribe the vaccine is its demonstrated ability to reduce the risk of invasive pneumococcal infection by up to 60%.
Dr. Terentes-Printzios reported having no financial conflicts.
AT THE ESC CONGRESS 2014
Key clinical point: Pneumococcal vaccine provides ancillary cardio- and cerebrovascular protective benefits, particularly in the elderly.
Major finding: Patients who got the pneumococcal vaccine had a statistically significant 8% reduction in the risk of cardiovascular mortality.
Data source: A meta-analysis of 11 studies comprising more than 332,000 patients.
Disclosures: The presenter reported having no conflicts of interest regarding this study.