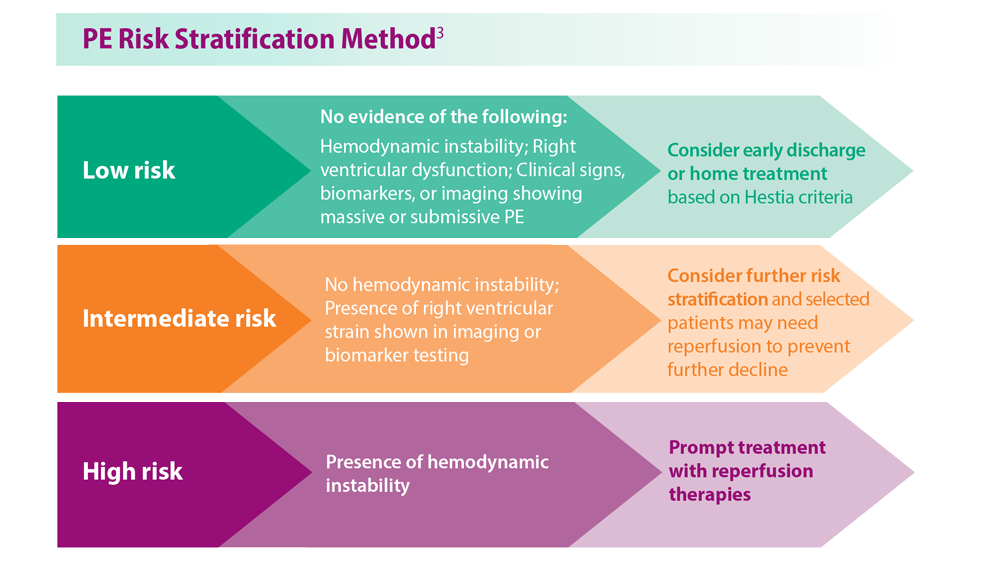
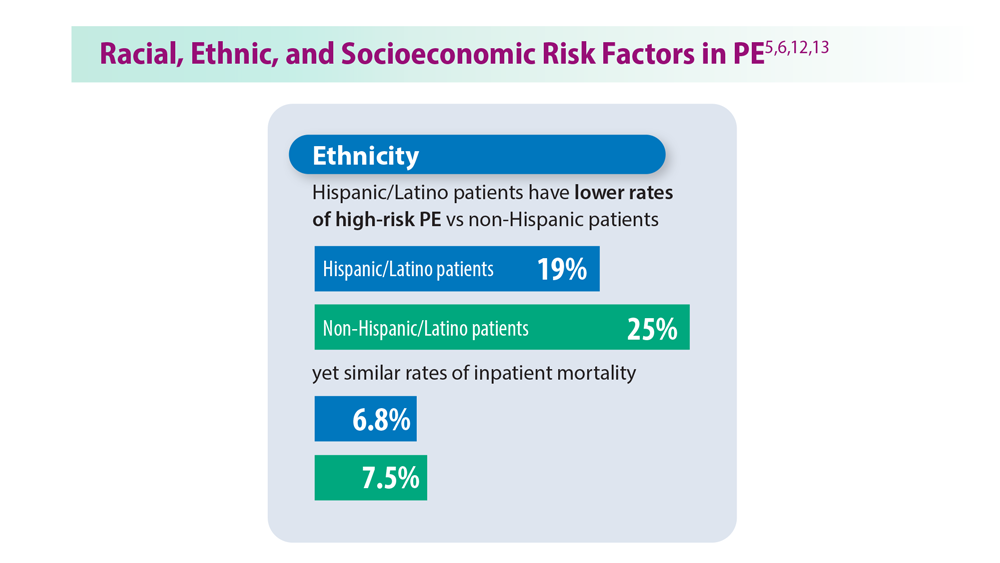
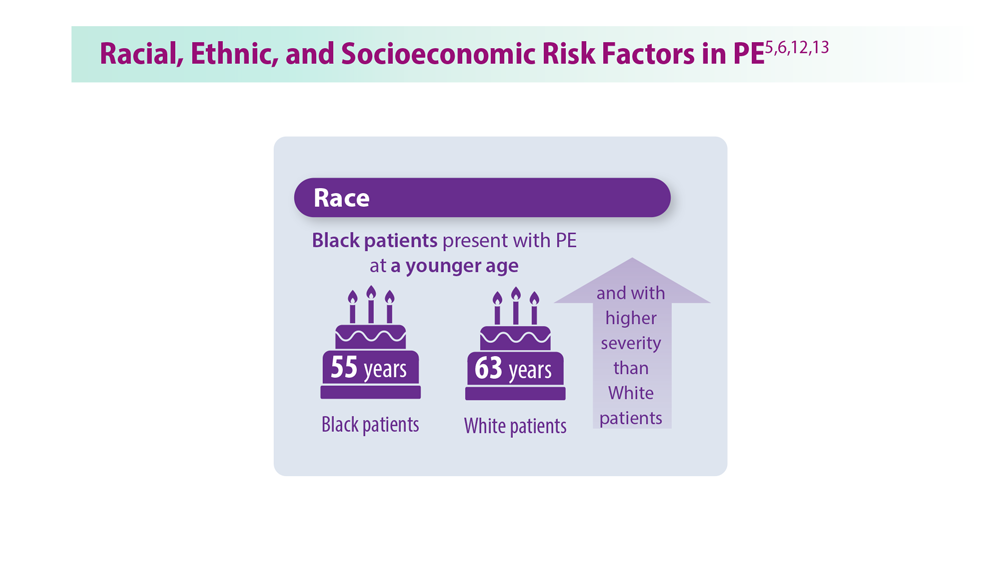
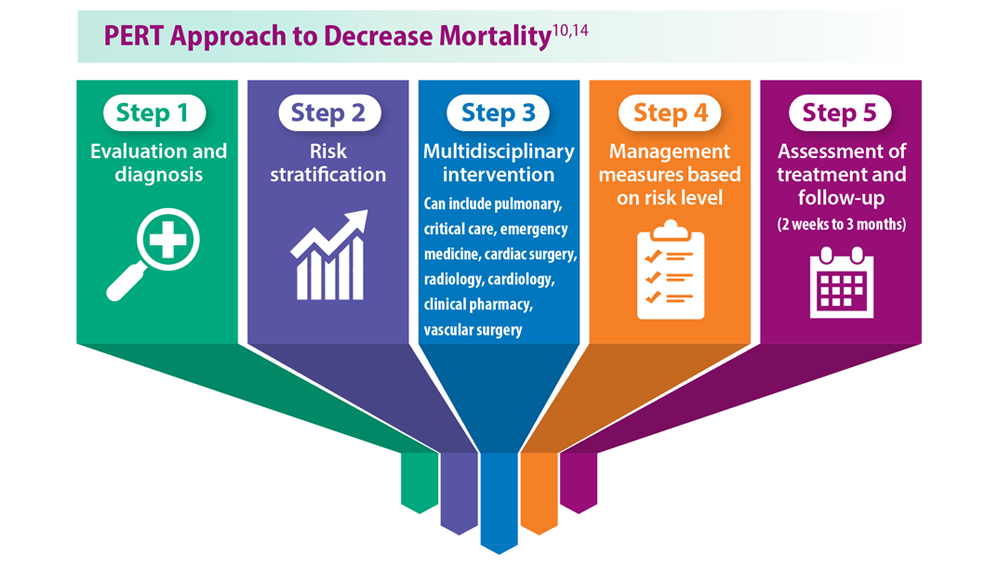
User login
MD-IQ only
Use of SGLT2 Inhibitors Associated With Better Survival in PAH
BOSTON — The use of sodium-glucose cotransporter-2 (SGLT2) inhibitors is associated with reduced short- and long-term mortality among patients with pulmonary arterial hypertension (PAH), according to results from a new propensity score–matched analysis.
“There are a lot of new studies that show benefits [of SGLT2 inhibitors] in heart failure, in [chronic kidney disease], and of course, in diabetes. There are studies that show that SGLT2 inhibitors can have an impact on inflammatory cascades, fibrosis, and vascular remodeling in general. Together, all this data triggered this idea for me, and that’s when I decided to conduct further studies,” said Irakli Lemonjava, MD, who presented the study at the American College of Chest Physicians (CHEST) 2024 Annual Meeting.
The researchers drew data on 125,634 adult patients from the TriNetX database who were diagnosed with PAH after January 1, 2013. They used propensity score matching to account for demographic characteristics and 10 organ system disorders to compare patients with exposure to SGLT2 inhibitors (canagliflozin, dapagliflozin, empagliflozin, or ertugliflozin; n = 6238) with those without such exposure (n = 6243).
At 1 year, 8.1% of patients taking SGLT2 inhibitors had died, compared with 15.5% of patients not taking SGLT2 inhibitors (risk reduction [RR], 0.52; P < .0001). The values were 13% and 22.5% (RR, 0.579; P < .0001) at 3 years and 14.6% and 25% at 5 years (RR, 0.583; P < .0001).
The study generated discussion during the Q&A period following the talk. One audience member asked if the group was able to access patients both inside and outside the United States. “Because I wonder if access to GLP2 inhibitors is actually a surrogate marker for access to other medications,” the questioner said.
Although the finding is intriguing, it shouldn’t change clinical practice, according to Lemonjava. “I don’t think we can make any changes based on what I shared today. Our purpose was to trigger the question. I think the numbers are so impressive that it will trigger more studies. I think if in the future it’s demonstrated by clinical trials that [SGLT2 inhibitors are beneficial], it will not be a problem to prescribe for someone with pulmonary arterial hypertension because they do not have many side effects,” he said. Lemonjava is a resident physician at Jefferson Einstein Philadelphia Hospital, Philadelphia.
Session co-moderator said Syed Rehan Quadery, MD, praised the study but emphasized the remaining uncertainty. “It’s an excellent proof of concept study. More trials need to [be done] on it, and we don’t understand the mechanism of action in which it improves survival in patients with pulmonary artery hypertension. The majority of the patients with pulmonary hypertension are much older and they have comorbidities, including cardiovascular risk factors, and maybe that is one of the ways in which this drug helps. Plus, there are multiple mechanisms in which it may be working, including anti-inflammatory as well as antiproliferative mechanisms through inhibiting the Notch-3 signaling pathway,” said Quadery, who is a consultant respiratory physician at National Pulmonary Hypertension Unit, Dublin, Ireland.
Quadery and his co-moderator Zeenat Safdar, MD, both noted that SGLT2 inhibitors have already been demonstrated to improve outcomes in heart failure. “[SGLT2 inhibition] improves survival, it decreases hospitalization, it improves morbidity and mortality. There are a lot of things that can be shown in different [animal or in vitro] models. In humans, we actually don’t know exactly how it works, but we know that it does. If it works in left heart failure, it also [could] work in right heart failure,” said Safdar, who is the director of the Houston Methodist Lung Center, Houston Methodist Hospital, Houston.
The study was independently supported. Lemonjava, Quadery, and Safdar reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
BOSTON — The use of sodium-glucose cotransporter-2 (SGLT2) inhibitors is associated with reduced short- and long-term mortality among patients with pulmonary arterial hypertension (PAH), according to results from a new propensity score–matched analysis.
“There are a lot of new studies that show benefits [of SGLT2 inhibitors] in heart failure, in [chronic kidney disease], and of course, in diabetes. There are studies that show that SGLT2 inhibitors can have an impact on inflammatory cascades, fibrosis, and vascular remodeling in general. Together, all this data triggered this idea for me, and that’s when I decided to conduct further studies,” said Irakli Lemonjava, MD, who presented the study at the American College of Chest Physicians (CHEST) 2024 Annual Meeting.
The researchers drew data on 125,634 adult patients from the TriNetX database who were diagnosed with PAH after January 1, 2013. They used propensity score matching to account for demographic characteristics and 10 organ system disorders to compare patients with exposure to SGLT2 inhibitors (canagliflozin, dapagliflozin, empagliflozin, or ertugliflozin; n = 6238) with those without such exposure (n = 6243).
At 1 year, 8.1% of patients taking SGLT2 inhibitors had died, compared with 15.5% of patients not taking SGLT2 inhibitors (risk reduction [RR], 0.52; P < .0001). The values were 13% and 22.5% (RR, 0.579; P < .0001) at 3 years and 14.6% and 25% at 5 years (RR, 0.583; P < .0001).
The study generated discussion during the Q&A period following the talk. One audience member asked if the group was able to access patients both inside and outside the United States. “Because I wonder if access to GLP2 inhibitors is actually a surrogate marker for access to other medications,” the questioner said.
Although the finding is intriguing, it shouldn’t change clinical practice, according to Lemonjava. “I don’t think we can make any changes based on what I shared today. Our purpose was to trigger the question. I think the numbers are so impressive that it will trigger more studies. I think if in the future it’s demonstrated by clinical trials that [SGLT2 inhibitors are beneficial], it will not be a problem to prescribe for someone with pulmonary arterial hypertension because they do not have many side effects,” he said. Lemonjava is a resident physician at Jefferson Einstein Philadelphia Hospital, Philadelphia.
Session co-moderator said Syed Rehan Quadery, MD, praised the study but emphasized the remaining uncertainty. “It’s an excellent proof of concept study. More trials need to [be done] on it, and we don’t understand the mechanism of action in which it improves survival in patients with pulmonary artery hypertension. The majority of the patients with pulmonary hypertension are much older and they have comorbidities, including cardiovascular risk factors, and maybe that is one of the ways in which this drug helps. Plus, there are multiple mechanisms in which it may be working, including anti-inflammatory as well as antiproliferative mechanisms through inhibiting the Notch-3 signaling pathway,” said Quadery, who is a consultant respiratory physician at National Pulmonary Hypertension Unit, Dublin, Ireland.
Quadery and his co-moderator Zeenat Safdar, MD, both noted that SGLT2 inhibitors have already been demonstrated to improve outcomes in heart failure. “[SGLT2 inhibition] improves survival, it decreases hospitalization, it improves morbidity and mortality. There are a lot of things that can be shown in different [animal or in vitro] models. In humans, we actually don’t know exactly how it works, but we know that it does. If it works in left heart failure, it also [could] work in right heart failure,” said Safdar, who is the director of the Houston Methodist Lung Center, Houston Methodist Hospital, Houston.
The study was independently supported. Lemonjava, Quadery, and Safdar reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
BOSTON — The use of sodium-glucose cotransporter-2 (SGLT2) inhibitors is associated with reduced short- and long-term mortality among patients with pulmonary arterial hypertension (PAH), according to results from a new propensity score–matched analysis.
“There are a lot of new studies that show benefits [of SGLT2 inhibitors] in heart failure, in [chronic kidney disease], and of course, in diabetes. There are studies that show that SGLT2 inhibitors can have an impact on inflammatory cascades, fibrosis, and vascular remodeling in general. Together, all this data triggered this idea for me, and that’s when I decided to conduct further studies,” said Irakli Lemonjava, MD, who presented the study at the American College of Chest Physicians (CHEST) 2024 Annual Meeting.
The researchers drew data on 125,634 adult patients from the TriNetX database who were diagnosed with PAH after January 1, 2013. They used propensity score matching to account for demographic characteristics and 10 organ system disorders to compare patients with exposure to SGLT2 inhibitors (canagliflozin, dapagliflozin, empagliflozin, or ertugliflozin; n = 6238) with those without such exposure (n = 6243).
At 1 year, 8.1% of patients taking SGLT2 inhibitors had died, compared with 15.5% of patients not taking SGLT2 inhibitors (risk reduction [RR], 0.52; P < .0001). The values were 13% and 22.5% (RR, 0.579; P < .0001) at 3 years and 14.6% and 25% at 5 years (RR, 0.583; P < .0001).
The study generated discussion during the Q&A period following the talk. One audience member asked if the group was able to access patients both inside and outside the United States. “Because I wonder if access to GLP2 inhibitors is actually a surrogate marker for access to other medications,” the questioner said.
Although the finding is intriguing, it shouldn’t change clinical practice, according to Lemonjava. “I don’t think we can make any changes based on what I shared today. Our purpose was to trigger the question. I think the numbers are so impressive that it will trigger more studies. I think if in the future it’s demonstrated by clinical trials that [SGLT2 inhibitors are beneficial], it will not be a problem to prescribe for someone with pulmonary arterial hypertension because they do not have many side effects,” he said. Lemonjava is a resident physician at Jefferson Einstein Philadelphia Hospital, Philadelphia.
Session co-moderator said Syed Rehan Quadery, MD, praised the study but emphasized the remaining uncertainty. “It’s an excellent proof of concept study. More trials need to [be done] on it, and we don’t understand the mechanism of action in which it improves survival in patients with pulmonary artery hypertension. The majority of the patients with pulmonary hypertension are much older and they have comorbidities, including cardiovascular risk factors, and maybe that is one of the ways in which this drug helps. Plus, there are multiple mechanisms in which it may be working, including anti-inflammatory as well as antiproliferative mechanisms through inhibiting the Notch-3 signaling pathway,” said Quadery, who is a consultant respiratory physician at National Pulmonary Hypertension Unit, Dublin, Ireland.
Quadery and his co-moderator Zeenat Safdar, MD, both noted that SGLT2 inhibitors have already been demonstrated to improve outcomes in heart failure. “[SGLT2 inhibition] improves survival, it decreases hospitalization, it improves morbidity and mortality. There are a lot of things that can be shown in different [animal or in vitro] models. In humans, we actually don’t know exactly how it works, but we know that it does. If it works in left heart failure, it also [could] work in right heart failure,” said Safdar, who is the director of the Houston Methodist Lung Center, Houston Methodist Hospital, Houston.
The study was independently supported. Lemonjava, Quadery, and Safdar reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
FROM CHEST 2024
Current Hydroxychloroquine Use in Lupus May Provide Protection Against Cardiovascular Events
TOPLINE:
Current use of hydroxychloroquine is associated with a lower risk for myocardial infarction (MI), stroke, and other thromboembolic events in patients with systemic lupus erythematosus (SLE). This protective effect diminishes after discontinuation of hydroxychloroquine treatment.
METHODOLOGY:
- Researchers used a nested case-control design to evaluate the association between exposure to hydroxychloroquine and the risk for cardiovascular events in patients with SLE.
- They included 52,883 adults with SLE (mean age, 44.23 years; 86.6% women) identified from the National System of Health Databases, which includes 99% of the French population.
- Among these, 1981 individuals with composite cardiovascular conditions were matched with 16,892 control individuals without cardiovascular conditions.
- Patients were categorized on the basis of hydroxychloroquine exposure into current users (last exposure within 90 days before a cardiovascular event), remote users (91-365 days before), and nonusers (no exposure within 365 days).
- The study outcomes included a composite of cardiovascular events, including MI, stroke (including transient ischemic attack), and other thromboembolic events such as phlebitis, thrombophlebitis, venous thrombosis, venous thromboembolism, and pulmonary embolism.
TAKEAWAY:
- Current hydroxychloroquine users had lower odds of experiencing a composite cardiovascular outcome than nonusers (adjusted odds ratio [aOR], 0.63; 95% CI, 0.57-0.70).
- The odds of MI (aOR, 0.72; 95% CI, 0.60-0.87), stroke (aOR, 0.71; 95% CI, 0.61-0.83), and other thromboembolic events (aOR, 0.58; 95% CI, 0.48-0.69) were also lower among current users than among nonusers.
- No significant association was found for remote hydroxychloroquine exposure and the risk for composite cardiovascular events, MI, stroke, and other thromboembolic events.
IN PRACTICE:
“These findings support the protective association of hydroxychloroquine against CV [cardiovascular] events and underscore the importance of continuous hydroxychloroquine therapy for patients diagnosed with SLE,” the authors wrote.
SOURCE:
The study was led by Lamiae Grimaldi-Bensouda, PharmD, PhD, Department of Pharmacology, Hospital Group Paris-Saclay, Assistance Publique-Hôpitaux de Paris, France. It was published online on August 30, 2024, in JAMA Network Open.
LIMITATIONS:
The observational nature of the study may have introduced confounding. Current hydroxychloroquine users were younger than nonusers, with an average age difference of almost 5 years. Current hydroxychloroquine users had a twofold longer duration of onset of SLE and had a higher prevalence of chronic kidney disease compared with nonusers.
DISCLOSURES:
This study was funded by the Banque pour l’Investissement, Deeptech. Some authors declared having financial ties with various institutions and companies outside of the current study.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Current use of hydroxychloroquine is associated with a lower risk for myocardial infarction (MI), stroke, and other thromboembolic events in patients with systemic lupus erythematosus (SLE). This protective effect diminishes after discontinuation of hydroxychloroquine treatment.
METHODOLOGY:
- Researchers used a nested case-control design to evaluate the association between exposure to hydroxychloroquine and the risk for cardiovascular events in patients with SLE.
- They included 52,883 adults with SLE (mean age, 44.23 years; 86.6% women) identified from the National System of Health Databases, which includes 99% of the French population.
- Among these, 1981 individuals with composite cardiovascular conditions were matched with 16,892 control individuals without cardiovascular conditions.
- Patients were categorized on the basis of hydroxychloroquine exposure into current users (last exposure within 90 days before a cardiovascular event), remote users (91-365 days before), and nonusers (no exposure within 365 days).
- The study outcomes included a composite of cardiovascular events, including MI, stroke (including transient ischemic attack), and other thromboembolic events such as phlebitis, thrombophlebitis, venous thrombosis, venous thromboembolism, and pulmonary embolism.
TAKEAWAY:
- Current hydroxychloroquine users had lower odds of experiencing a composite cardiovascular outcome than nonusers (adjusted odds ratio [aOR], 0.63; 95% CI, 0.57-0.70).
- The odds of MI (aOR, 0.72; 95% CI, 0.60-0.87), stroke (aOR, 0.71; 95% CI, 0.61-0.83), and other thromboembolic events (aOR, 0.58; 95% CI, 0.48-0.69) were also lower among current users than among nonusers.
- No significant association was found for remote hydroxychloroquine exposure and the risk for composite cardiovascular events, MI, stroke, and other thromboembolic events.
IN PRACTICE:
“These findings support the protective association of hydroxychloroquine against CV [cardiovascular] events and underscore the importance of continuous hydroxychloroquine therapy for patients diagnosed with SLE,” the authors wrote.
SOURCE:
The study was led by Lamiae Grimaldi-Bensouda, PharmD, PhD, Department of Pharmacology, Hospital Group Paris-Saclay, Assistance Publique-Hôpitaux de Paris, France. It was published online on August 30, 2024, in JAMA Network Open.
LIMITATIONS:
The observational nature of the study may have introduced confounding. Current hydroxychloroquine users were younger than nonusers, with an average age difference of almost 5 years. Current hydroxychloroquine users had a twofold longer duration of onset of SLE and had a higher prevalence of chronic kidney disease compared with nonusers.
DISCLOSURES:
This study was funded by the Banque pour l’Investissement, Deeptech. Some authors declared having financial ties with various institutions and companies outside of the current study.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Current use of hydroxychloroquine is associated with a lower risk for myocardial infarction (MI), stroke, and other thromboembolic events in patients with systemic lupus erythematosus (SLE). This protective effect diminishes after discontinuation of hydroxychloroquine treatment.
METHODOLOGY:
- Researchers used a nested case-control design to evaluate the association between exposure to hydroxychloroquine and the risk for cardiovascular events in patients with SLE.
- They included 52,883 adults with SLE (mean age, 44.23 years; 86.6% women) identified from the National System of Health Databases, which includes 99% of the French population.
- Among these, 1981 individuals with composite cardiovascular conditions were matched with 16,892 control individuals without cardiovascular conditions.
- Patients were categorized on the basis of hydroxychloroquine exposure into current users (last exposure within 90 days before a cardiovascular event), remote users (91-365 days before), and nonusers (no exposure within 365 days).
- The study outcomes included a composite of cardiovascular events, including MI, stroke (including transient ischemic attack), and other thromboembolic events such as phlebitis, thrombophlebitis, venous thrombosis, venous thromboembolism, and pulmonary embolism.
TAKEAWAY:
- Current hydroxychloroquine users had lower odds of experiencing a composite cardiovascular outcome than nonusers (adjusted odds ratio [aOR], 0.63; 95% CI, 0.57-0.70).
- The odds of MI (aOR, 0.72; 95% CI, 0.60-0.87), stroke (aOR, 0.71; 95% CI, 0.61-0.83), and other thromboembolic events (aOR, 0.58; 95% CI, 0.48-0.69) were also lower among current users than among nonusers.
- No significant association was found for remote hydroxychloroquine exposure and the risk for composite cardiovascular events, MI, stroke, and other thromboembolic events.
IN PRACTICE:
“These findings support the protective association of hydroxychloroquine against CV [cardiovascular] events and underscore the importance of continuous hydroxychloroquine therapy for patients diagnosed with SLE,” the authors wrote.
SOURCE:
The study was led by Lamiae Grimaldi-Bensouda, PharmD, PhD, Department of Pharmacology, Hospital Group Paris-Saclay, Assistance Publique-Hôpitaux de Paris, France. It was published online on August 30, 2024, in JAMA Network Open.
LIMITATIONS:
The observational nature of the study may have introduced confounding. Current hydroxychloroquine users were younger than nonusers, with an average age difference of almost 5 years. Current hydroxychloroquine users had a twofold longer duration of onset of SLE and had a higher prevalence of chronic kidney disease compared with nonusers.
DISCLOSURES:
This study was funded by the Banque pour l’Investissement, Deeptech. Some authors declared having financial ties with various institutions and companies outside of the current study.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Excess Thrombotic Risk in RA Has No Clear Driving Factor
LIVERPOOL, ENGLAND — People with rheumatoid arthritis (RA) have a consistently higher risk for venous thromboembolism (VTE) than the general population, but the reasons for this remain unclear, research presented at the annual meeting of the British Society for Rheumatology (BSR) reaffirmed.
Regardless of age, sex, body mass index (BMI), duration of disease, use of estrogen-based oral contraceptives, or hormone replacement therapy (HRT), people with RA are more likely to experience a pulmonary embolism or deep vein thrombosis than those without RA.
However, “these are rare events,” James Galloway, MBChB, PhD, professor of rheumatology and deputy head of the Centre for Rheumatic Diseases at King’s College London in England, said at the meeting.
In one analysis of data from 117,050 individuals living in England and Wales that are held within a large primary care practice database, Dr. Galloway and colleagues found that the unadjusted incidence of VTE in people diagnosed with RA (n = 23,410) was 0.44% vs 0.26% for matched controls within the general population (n = 93,640).
RA and VTE Risk
The overall risk for VTE was 46% higher among people with RA than among those without, although the absolute difference was small, Dr. Galloway reported.
“RA is associated with an increased risk of VTE; that’s been well described over the years,” Dr. Galloway told this news organization. Past research into why there is an elevated risk for VTE in patients with RA has often focused on the role of disease activity and inflammation.
“In the last few years, a new class of drugs, the JAK [Janus kinase] inhibitors, have emerged in which we have seen a signal of increased VTE risk from a number of studies. And I think that puts a spotlight on our understanding of VTE risk,” Dr. Galloway said.
He added “JAK inhibitors are very powerful at controlling inflammation, but if you take away inflammation, there is still an excess risk. What else could be driving that?”
To examine the excess risk for VTE seen in people with RA, Dr. Galloway and colleagues performed three separate analyses using data collected between January 1999 and December 2018 by the Royal College of General Practitioners Research and Surveillance Center.
One analysis looked at VTE risk according to age, sex, and BMI; another looked at the effect of the duration of RA; and a third analysis focused on the use of estrogen-based oral contraceptives or HRT.
For all three analyses, those with RA were matched in a 4:1 ratio to people from the general population without RA on the basis of current age, sex, calendar time, and years since registration at the primary care practice.
Observational Data Challenged
“These are observational data, so it’s important to weigh up the strengths and limitations,” Dr. Galloway acknowledged. Strengths are the large sample size and long follow-up provided by the database, which assesses and monitors more than 2000 primary care practices in England and Wales.
Confounding is still possible, despite adjusting for multiple factors that included sociodemographic factors; clinical features; and VTE risk factors such as smoking status, alcohol use, thrombophilia, reduced mobility, lower limb fracture, and a family history of VTE if data had been available. There wasn’t information on disease activity, for example, and disease duration was used as a surrogate marker for this.
Sitting in the audience, Marwan Bukhari, MBBS, PhD, challenged the population-matching process.
“Do you think maybe it was the matching that was the problem?” asked Dr. Bukhari, who is consultant rheumatologist at University Hospitals of Morecambe Bay NHS Foundation Trust and an honorary senior lecturer at the University of Manchester, both in England.
“They’re not entirely matched completely, correctly. Even if it is 4:1, there’s a difference between the populations,” he said.
Age, Sex, and Bodyweight
Over an average of 8.2 years’ follow-up, the adjusted hazard ratios (aHRs) comparing VTE risk in women and men with and without RA were a respective 1.62 and 1.52. The corresponding aHRs for VTE according to different age groups were 2.13 for age 18-49 years, 1.57 for age 50-69 years, and 1.34 for age 70 years and older.
“The highest excess risk was in the youngest age group,” Dr. Galloway pointed out, “but all age groups showing a significant increased risk of venous thromboembolism.”
Similar findings were seen across different BMI categories, with the highest risk occurring in those in the lowest BMI group. The aHRs were 1.66, 1.60, and 1.41 for the BMI categories of less than 25 kg/m2, 25-30 kg/m2, and more than 30 kg/m2, respectively.
Duration of RA
As for disease duration, nearly two thirds (63.9%) of the 23,410 adults with RA included in this analysis were included at or within 2 years of a diagnosis of RA, 7.8% within 2-5 years of diagnosis, 9.8% within 5-10 years of diagnosis, and 18.5% at 10 or more years after diagnosis.
The aHR for an increased relative risk for VTE in people with RA vs the control group ranged from 1.49 for 0-2 years of diagnosis up to 1.63 for more than 10 years since diagnosis.
“We could see no evidence that the VTE excess risk in rheumatoid arthritis was with a specific time since diagnosis,” Dr. Galloway said in the interview. “It appears that the risk is increased in people with established RA, whether you’ve had the disease for 2 years or 10 years.”
Similar findings were also seen when they looked at aHRs for pulmonary embolism (1.46-2.02) and deep vein thrombosis (1.43-1.89) separately.
Oral Contraceptives and HRT
Data on the use of estrogen-based oral contraceptives or HRT were detailed in a virtual poster presentation. In this analysis, there were 16,664 women with and 65,448 without RA, and the average follow-up was 8.3 years.
“The number of people available for this analysis was small, and bigger studies are needed,” Dr. Galloway said in the interview. Indeed, in the RA group, just 3.3% had used an estrogen-based oral contraceptive and 4.5% had used HRT compared with 3.9% and 3.8% in the control group, respectively.
The overall VTE risk was 52% higher in women with RA than in those without RA.
Risk for VTE was higher among women with RA regardless of the use of estrogen-based oral contraceptives or not (aHRs, 1.43 and 1.52, respectively) and regardless of the use of HRT or not (aHRs, 2.32 and 1.51).
Assess and Monitor
Together these data increase understanding of how age, gender, obesity, duration of disease, and estrogen-based contraception and HRT may make a difference to someone’s VTE risk.
“In all people with RA, we observe an increased risk of venous thromboembolism, and that is both relevant in a contemporary era when we think about prescribing and the different risks of drugs we use for therapeutic strategies,” Dr. Galloway said.
The overall take-home message, he said, is that VTE risk should be considered in everyone with RA and assessed and monitored accordingly. This includes those who may have traditionally been thought of as having a lower risk than others, such as men vs women, younger vs older individuals, and those who may have had RA for a few years.
The research was funded by Pfizer. Dr. Galloway reported receiving honoraria from Pfizer, AbbVie, Biovitrum, Bristol Myers Squibb, Celgene, Chugai, Galapagos, Janssen, Lilly, Novartis, Roche, Sanofi, Sobi, and UCB. Two coauthors of the work were employees of Pfizer. Dr. Bukhari had no conflicts of interest and was not involved in the research.
A version of this article appeared on Medscape.com.
LIVERPOOL, ENGLAND — People with rheumatoid arthritis (RA) have a consistently higher risk for venous thromboembolism (VTE) than the general population, but the reasons for this remain unclear, research presented at the annual meeting of the British Society for Rheumatology (BSR) reaffirmed.
Regardless of age, sex, body mass index (BMI), duration of disease, use of estrogen-based oral contraceptives, or hormone replacement therapy (HRT), people with RA are more likely to experience a pulmonary embolism or deep vein thrombosis than those without RA.
However, “these are rare events,” James Galloway, MBChB, PhD, professor of rheumatology and deputy head of the Centre for Rheumatic Diseases at King’s College London in England, said at the meeting.
In one analysis of data from 117,050 individuals living in England and Wales that are held within a large primary care practice database, Dr. Galloway and colleagues found that the unadjusted incidence of VTE in people diagnosed with RA (n = 23,410) was 0.44% vs 0.26% for matched controls within the general population (n = 93,640).
RA and VTE Risk
The overall risk for VTE was 46% higher among people with RA than among those without, although the absolute difference was small, Dr. Galloway reported.
“RA is associated with an increased risk of VTE; that’s been well described over the years,” Dr. Galloway told this news organization. Past research into why there is an elevated risk for VTE in patients with RA has often focused on the role of disease activity and inflammation.
“In the last few years, a new class of drugs, the JAK [Janus kinase] inhibitors, have emerged in which we have seen a signal of increased VTE risk from a number of studies. And I think that puts a spotlight on our understanding of VTE risk,” Dr. Galloway said.
He added “JAK inhibitors are very powerful at controlling inflammation, but if you take away inflammation, there is still an excess risk. What else could be driving that?”
To examine the excess risk for VTE seen in people with RA, Dr. Galloway and colleagues performed three separate analyses using data collected between January 1999 and December 2018 by the Royal College of General Practitioners Research and Surveillance Center.
One analysis looked at VTE risk according to age, sex, and BMI; another looked at the effect of the duration of RA; and a third analysis focused on the use of estrogen-based oral contraceptives or HRT.
For all three analyses, those with RA were matched in a 4:1 ratio to people from the general population without RA on the basis of current age, sex, calendar time, and years since registration at the primary care practice.
Observational Data Challenged
“These are observational data, so it’s important to weigh up the strengths and limitations,” Dr. Galloway acknowledged. Strengths are the large sample size and long follow-up provided by the database, which assesses and monitors more than 2000 primary care practices in England and Wales.
Confounding is still possible, despite adjusting for multiple factors that included sociodemographic factors; clinical features; and VTE risk factors such as smoking status, alcohol use, thrombophilia, reduced mobility, lower limb fracture, and a family history of VTE if data had been available. There wasn’t information on disease activity, for example, and disease duration was used as a surrogate marker for this.
Sitting in the audience, Marwan Bukhari, MBBS, PhD, challenged the population-matching process.
“Do you think maybe it was the matching that was the problem?” asked Dr. Bukhari, who is consultant rheumatologist at University Hospitals of Morecambe Bay NHS Foundation Trust and an honorary senior lecturer at the University of Manchester, both in England.
“They’re not entirely matched completely, correctly. Even if it is 4:1, there’s a difference between the populations,” he said.
Age, Sex, and Bodyweight
Over an average of 8.2 years’ follow-up, the adjusted hazard ratios (aHRs) comparing VTE risk in women and men with and without RA were a respective 1.62 and 1.52. The corresponding aHRs for VTE according to different age groups were 2.13 for age 18-49 years, 1.57 for age 50-69 years, and 1.34 for age 70 years and older.
“The highest excess risk was in the youngest age group,” Dr. Galloway pointed out, “but all age groups showing a significant increased risk of venous thromboembolism.”
Similar findings were seen across different BMI categories, with the highest risk occurring in those in the lowest BMI group. The aHRs were 1.66, 1.60, and 1.41 for the BMI categories of less than 25 kg/m2, 25-30 kg/m2, and more than 30 kg/m2, respectively.
Duration of RA
As for disease duration, nearly two thirds (63.9%) of the 23,410 adults with RA included in this analysis were included at or within 2 years of a diagnosis of RA, 7.8% within 2-5 years of diagnosis, 9.8% within 5-10 years of diagnosis, and 18.5% at 10 or more years after diagnosis.
The aHR for an increased relative risk for VTE in people with RA vs the control group ranged from 1.49 for 0-2 years of diagnosis up to 1.63 for more than 10 years since diagnosis.
“We could see no evidence that the VTE excess risk in rheumatoid arthritis was with a specific time since diagnosis,” Dr. Galloway said in the interview. “It appears that the risk is increased in people with established RA, whether you’ve had the disease for 2 years or 10 years.”
Similar findings were also seen when they looked at aHRs for pulmonary embolism (1.46-2.02) and deep vein thrombosis (1.43-1.89) separately.
Oral Contraceptives and HRT
Data on the use of estrogen-based oral contraceptives or HRT were detailed in a virtual poster presentation. In this analysis, there were 16,664 women with and 65,448 without RA, and the average follow-up was 8.3 years.
“The number of people available for this analysis was small, and bigger studies are needed,” Dr. Galloway said in the interview. Indeed, in the RA group, just 3.3% had used an estrogen-based oral contraceptive and 4.5% had used HRT compared with 3.9% and 3.8% in the control group, respectively.
The overall VTE risk was 52% higher in women with RA than in those without RA.
Risk for VTE was higher among women with RA regardless of the use of estrogen-based oral contraceptives or not (aHRs, 1.43 and 1.52, respectively) and regardless of the use of HRT or not (aHRs, 2.32 and 1.51).
Assess and Monitor
Together these data increase understanding of how age, gender, obesity, duration of disease, and estrogen-based contraception and HRT may make a difference to someone’s VTE risk.
“In all people with RA, we observe an increased risk of venous thromboembolism, and that is both relevant in a contemporary era when we think about prescribing and the different risks of drugs we use for therapeutic strategies,” Dr. Galloway said.
The overall take-home message, he said, is that VTE risk should be considered in everyone with RA and assessed and monitored accordingly. This includes those who may have traditionally been thought of as having a lower risk than others, such as men vs women, younger vs older individuals, and those who may have had RA for a few years.
The research was funded by Pfizer. Dr. Galloway reported receiving honoraria from Pfizer, AbbVie, Biovitrum, Bristol Myers Squibb, Celgene, Chugai, Galapagos, Janssen, Lilly, Novartis, Roche, Sanofi, Sobi, and UCB. Two coauthors of the work were employees of Pfizer. Dr. Bukhari had no conflicts of interest and was not involved in the research.
A version of this article appeared on Medscape.com.
LIVERPOOL, ENGLAND — People with rheumatoid arthritis (RA) have a consistently higher risk for venous thromboembolism (VTE) than the general population, but the reasons for this remain unclear, research presented at the annual meeting of the British Society for Rheumatology (BSR) reaffirmed.
Regardless of age, sex, body mass index (BMI), duration of disease, use of estrogen-based oral contraceptives, or hormone replacement therapy (HRT), people with RA are more likely to experience a pulmonary embolism or deep vein thrombosis than those without RA.
However, “these are rare events,” James Galloway, MBChB, PhD, professor of rheumatology and deputy head of the Centre for Rheumatic Diseases at King’s College London in England, said at the meeting.
In one analysis of data from 117,050 individuals living in England and Wales that are held within a large primary care practice database, Dr. Galloway and colleagues found that the unadjusted incidence of VTE in people diagnosed with RA (n = 23,410) was 0.44% vs 0.26% for matched controls within the general population (n = 93,640).
RA and VTE Risk
The overall risk for VTE was 46% higher among people with RA than among those without, although the absolute difference was small, Dr. Galloway reported.
“RA is associated with an increased risk of VTE; that’s been well described over the years,” Dr. Galloway told this news organization. Past research into why there is an elevated risk for VTE in patients with RA has often focused on the role of disease activity and inflammation.
“In the last few years, a new class of drugs, the JAK [Janus kinase] inhibitors, have emerged in which we have seen a signal of increased VTE risk from a number of studies. And I think that puts a spotlight on our understanding of VTE risk,” Dr. Galloway said.
He added “JAK inhibitors are very powerful at controlling inflammation, but if you take away inflammation, there is still an excess risk. What else could be driving that?”
To examine the excess risk for VTE seen in people with RA, Dr. Galloway and colleagues performed three separate analyses using data collected between January 1999 and December 2018 by the Royal College of General Practitioners Research and Surveillance Center.
One analysis looked at VTE risk according to age, sex, and BMI; another looked at the effect of the duration of RA; and a third analysis focused on the use of estrogen-based oral contraceptives or HRT.
For all three analyses, those with RA were matched in a 4:1 ratio to people from the general population without RA on the basis of current age, sex, calendar time, and years since registration at the primary care practice.
Observational Data Challenged
“These are observational data, so it’s important to weigh up the strengths and limitations,” Dr. Galloway acknowledged. Strengths are the large sample size and long follow-up provided by the database, which assesses and monitors more than 2000 primary care practices in England and Wales.
Confounding is still possible, despite adjusting for multiple factors that included sociodemographic factors; clinical features; and VTE risk factors such as smoking status, alcohol use, thrombophilia, reduced mobility, lower limb fracture, and a family history of VTE if data had been available. There wasn’t information on disease activity, for example, and disease duration was used as a surrogate marker for this.
Sitting in the audience, Marwan Bukhari, MBBS, PhD, challenged the population-matching process.
“Do you think maybe it was the matching that was the problem?” asked Dr. Bukhari, who is consultant rheumatologist at University Hospitals of Morecambe Bay NHS Foundation Trust and an honorary senior lecturer at the University of Manchester, both in England.
“They’re not entirely matched completely, correctly. Even if it is 4:1, there’s a difference between the populations,” he said.
Age, Sex, and Bodyweight
Over an average of 8.2 years’ follow-up, the adjusted hazard ratios (aHRs) comparing VTE risk in women and men with and without RA were a respective 1.62 and 1.52. The corresponding aHRs for VTE according to different age groups were 2.13 for age 18-49 years, 1.57 for age 50-69 years, and 1.34 for age 70 years and older.
“The highest excess risk was in the youngest age group,” Dr. Galloway pointed out, “but all age groups showing a significant increased risk of venous thromboembolism.”
Similar findings were seen across different BMI categories, with the highest risk occurring in those in the lowest BMI group. The aHRs were 1.66, 1.60, and 1.41 for the BMI categories of less than 25 kg/m2, 25-30 kg/m2, and more than 30 kg/m2, respectively.
Duration of RA
As for disease duration, nearly two thirds (63.9%) of the 23,410 adults with RA included in this analysis were included at or within 2 years of a diagnosis of RA, 7.8% within 2-5 years of diagnosis, 9.8% within 5-10 years of diagnosis, and 18.5% at 10 or more years after diagnosis.
The aHR for an increased relative risk for VTE in people with RA vs the control group ranged from 1.49 for 0-2 years of diagnosis up to 1.63 for more than 10 years since diagnosis.
“We could see no evidence that the VTE excess risk in rheumatoid arthritis was with a specific time since diagnosis,” Dr. Galloway said in the interview. “It appears that the risk is increased in people with established RA, whether you’ve had the disease for 2 years or 10 years.”
Similar findings were also seen when they looked at aHRs for pulmonary embolism (1.46-2.02) and deep vein thrombosis (1.43-1.89) separately.
Oral Contraceptives and HRT
Data on the use of estrogen-based oral contraceptives or HRT were detailed in a virtual poster presentation. In this analysis, there were 16,664 women with and 65,448 without RA, and the average follow-up was 8.3 years.
“The number of people available for this analysis was small, and bigger studies are needed,” Dr. Galloway said in the interview. Indeed, in the RA group, just 3.3% had used an estrogen-based oral contraceptive and 4.5% had used HRT compared with 3.9% and 3.8% in the control group, respectively.
The overall VTE risk was 52% higher in women with RA than in those without RA.
Risk for VTE was higher among women with RA regardless of the use of estrogen-based oral contraceptives or not (aHRs, 1.43 and 1.52, respectively) and regardless of the use of HRT or not (aHRs, 2.32 and 1.51).
Assess and Monitor
Together these data increase understanding of how age, gender, obesity, duration of disease, and estrogen-based contraception and HRT may make a difference to someone’s VTE risk.
“In all people with RA, we observe an increased risk of venous thromboembolism, and that is both relevant in a contemporary era when we think about prescribing and the different risks of drugs we use for therapeutic strategies,” Dr. Galloway said.
The overall take-home message, he said, is that VTE risk should be considered in everyone with RA and assessed and monitored accordingly. This includes those who may have traditionally been thought of as having a lower risk than others, such as men vs women, younger vs older individuals, and those who may have had RA for a few years.
The research was funded by Pfizer. Dr. Galloway reported receiving honoraria from Pfizer, AbbVie, Biovitrum, Bristol Myers Squibb, Celgene, Chugai, Galapagos, Janssen, Lilly, Novartis, Roche, Sanofi, Sobi, and UCB. Two coauthors of the work were employees of Pfizer. Dr. Bukhari had no conflicts of interest and was not involved in the research.
A version of this article appeared on Medscape.com.
FROM BSR 2024
Review Finds No Short-term MACE, VTE risk with JAK Inhibitors For Dermatoses
, at least in the short term, say the authors of a new meta-analysis published in JAMA Dermatology.
Considering data on over 17,000 patients with different dermatoses from 45 placebo-controlled randomized clinical trials with an average follow up of 16 weeks, they found there was no significant increase in the occurrence of major adverse cardiovascular events (MACE) or venous thromboembolism (VTE) in people with dermatoses treated with JAK-STAT inhibitors, compared with placebo.
The I² statistic was 0.00% for both MACE and VTE comparing the two arms, indicating that the results were unlikely to be due to chance. There was no increased risk in MACE between those on placebo and those on JAK-STAT inhibitors, with a risk ratio (RR) of 0.47; or for VTE risk, with an RR of 0.46.
Similar findings were obtained when data were analyzed according to the dermatological condition being treated, mechanism of action of the medication, or whether the medication carried a boxed warning.
These data “suggest inconsistency with established sentiments,” that JAK-STAT inhibitors increase the risk for cardiovascular events, Patrick Ireland, MD, of the University of New South Wales, Randwick, Australia, and coauthors wrote in the article. “This may be owing to the limited time frames in which these rare events could be adequately captured, or the ages of enrolled patients being too young to realize the well established heightened risks of developing MACE and VTE,” they suggested.
However, the findings challenge the notion that the cardiovascular complications of these drugs are the same in all patients; dermatological use may not be associated with the same risks as with use for rheumatologic indications.
Class-Wide Boxed Warning
“JAK-STAT [inhibitors] have had some pretty indemnifying data against their use, with the ORAL [Surveillance] study demonstrating increased all-cause mortality, cardiovascular events, venous thromboembolism, and malignancy,” Dr. Ireland said in an interview.
ORAL Surveillance was an open-label, postmarketing trial conducted in patients with rheumatoid arthritis treated with tofacitinib or a tumor necrosis factor (TNF) inhibitor. The results led the US Food and Drug Administration to require information about the risks of serious heart-related events, cancer, blood clots, and death in a boxed warning for JAK-STAT inhibitors in 2022.
“I think it’s important to recognize that these [ORAL Surveillance participants] are very different patients to the typical dermatological patient being treated with a JAK-STAT [inhibitors], with newer studies demonstrating a much safer profile than initially thought,” Dr. Ireland said.
Examining Risk in Dermatological Conditions
The meta-analysis performed by Dr. Ireland and associates focused specifically on the risk for MACE and VTE in patients being treated for dermatological conditions, and included trials published up until June 2023. Only trials that had included a placebo arm were considered; pooled analyses, long-term extension trial data, post hoc analyses, and pediatric-specific trials were excluded.
Most (25) of the trials were phase 2b or phase 3 trials, 18 were phase 2 to 2b, and two were phase 1 trials. The studies included 12,996 participants, mostly with atopic dermatitis or psoriasis, who were treated with JAK-STAT inhibitors, which included baricitinib (2846 patients), tofacitinib (2470), upadacitinib (2218), abrocitinib (1904), and deucravacitinib (1492), among others. There were 4925 patients on placebo.
Overall, MACE — defined as a combined endpoint of acute myocardial infarction, stroke, cardiovascular mortality, heart failure, and unstable angina, as well as arterial embolism — occurred in 13 of the JAK-STAT inhibitor-treated patients and in four of those on placebo. VTE — defined as deep vein thrombosis, pulmonary embolism, and any unusual site thrombosis — was reported in eight JAK-STAT inhibitor-treated patients and in one patient on placebo.
The pooled incidence ratios for MACE and VTE were calculated as 0.20 per 100 person exposure years (PEY) for JAK-STAT inhibitor treatment and 0.13 PEY for placebo. The pooled RRs comparing the two treatment groups were a respective 1.13 for MACE and 2.79 for VTE, but neither RR reached statistical significance.
No difference was seen between the treatment arms in terms of treatment emergent adverse events (RR, 1.05), serious adverse events (RR, 0.92), or study discontinuation because of adverse events (RR, 0.94).
Reassuring Results?
Dr. Ireland and coauthors said the finding should help to reassure clinicians that the short-term use of JAK-STAT inhibitors in patients with dermatological conditions with low cardiovascular risk profiles “appears to be both safe and well tolerated.” They cautioned, however, that “clinicians must remain judicious” when using these medications for longer periods and in high-risk patient populations.
This was a pragmatic meta-analysis that provides useful information for dermatologists, Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, DC, said in an interview.
“When there are safety concerns, I think that’s where data like this are so important to not just allay the fears of practitioners, but also to arm the practitioner with information for when they discuss a possible treatment with a patient,” said Dr. Friedman, who was not involved in the study.
“What’s unique here is that they’re looking at any possible use of JAK inhibitors for dermatological disease,” so this represents patients that dermatologists would be seeing, he added.
“The limitation here is time, we only can say so much about the safety of the medication with the data that we have,” Dr. Friedman said. Almost 4 months is “a good amount of time” to know about the cardiovascular risks, he said, but added, what happens then? Will the risk increase and will patients need to be switched to another medication?
“There’s no line in the sand,” with regard to using a JAK-STAT inhibitor. “If you look at the label, they’re not meant to be used incrementally,” but as ongoing treatment, while considering the needs of the patient and the relative risks and benefits, he said.
With that in mind, “the open label extension studies for all these [JAK-STAT inhibitors] are really, really important to get a sense of ‘do new signals emerge down the road.’ ”
The meta-analysis received no commercial funding. One author of the work reported personal fees from several pharmaceutical companies which were done outside of analysis. Dr. Friedman has received research funding from or acted as a consultant for several pharmaceutical companies including, Incyte, Pfizer, Eli Lily, and AbbVie.
, at least in the short term, say the authors of a new meta-analysis published in JAMA Dermatology.
Considering data on over 17,000 patients with different dermatoses from 45 placebo-controlled randomized clinical trials with an average follow up of 16 weeks, they found there was no significant increase in the occurrence of major adverse cardiovascular events (MACE) or venous thromboembolism (VTE) in people with dermatoses treated with JAK-STAT inhibitors, compared with placebo.
The I² statistic was 0.00% for both MACE and VTE comparing the two arms, indicating that the results were unlikely to be due to chance. There was no increased risk in MACE between those on placebo and those on JAK-STAT inhibitors, with a risk ratio (RR) of 0.47; or for VTE risk, with an RR of 0.46.
Similar findings were obtained when data were analyzed according to the dermatological condition being treated, mechanism of action of the medication, or whether the medication carried a boxed warning.
These data “suggest inconsistency with established sentiments,” that JAK-STAT inhibitors increase the risk for cardiovascular events, Patrick Ireland, MD, of the University of New South Wales, Randwick, Australia, and coauthors wrote in the article. “This may be owing to the limited time frames in which these rare events could be adequately captured, or the ages of enrolled patients being too young to realize the well established heightened risks of developing MACE and VTE,” they suggested.
However, the findings challenge the notion that the cardiovascular complications of these drugs are the same in all patients; dermatological use may not be associated with the same risks as with use for rheumatologic indications.
Class-Wide Boxed Warning
“JAK-STAT [inhibitors] have had some pretty indemnifying data against their use, with the ORAL [Surveillance] study demonstrating increased all-cause mortality, cardiovascular events, venous thromboembolism, and malignancy,” Dr. Ireland said in an interview.
ORAL Surveillance was an open-label, postmarketing trial conducted in patients with rheumatoid arthritis treated with tofacitinib or a tumor necrosis factor (TNF) inhibitor. The results led the US Food and Drug Administration to require information about the risks of serious heart-related events, cancer, blood clots, and death in a boxed warning for JAK-STAT inhibitors in 2022.
“I think it’s important to recognize that these [ORAL Surveillance participants] are very different patients to the typical dermatological patient being treated with a JAK-STAT [inhibitors], with newer studies demonstrating a much safer profile than initially thought,” Dr. Ireland said.
Examining Risk in Dermatological Conditions
The meta-analysis performed by Dr. Ireland and associates focused specifically on the risk for MACE and VTE in patients being treated for dermatological conditions, and included trials published up until June 2023. Only trials that had included a placebo arm were considered; pooled analyses, long-term extension trial data, post hoc analyses, and pediatric-specific trials were excluded.
Most (25) of the trials were phase 2b or phase 3 trials, 18 were phase 2 to 2b, and two were phase 1 trials. The studies included 12,996 participants, mostly with atopic dermatitis or psoriasis, who were treated with JAK-STAT inhibitors, which included baricitinib (2846 patients), tofacitinib (2470), upadacitinib (2218), abrocitinib (1904), and deucravacitinib (1492), among others. There were 4925 patients on placebo.
Overall, MACE — defined as a combined endpoint of acute myocardial infarction, stroke, cardiovascular mortality, heart failure, and unstable angina, as well as arterial embolism — occurred in 13 of the JAK-STAT inhibitor-treated patients and in four of those on placebo. VTE — defined as deep vein thrombosis, pulmonary embolism, and any unusual site thrombosis — was reported in eight JAK-STAT inhibitor-treated patients and in one patient on placebo.
The pooled incidence ratios for MACE and VTE were calculated as 0.20 per 100 person exposure years (PEY) for JAK-STAT inhibitor treatment and 0.13 PEY for placebo. The pooled RRs comparing the two treatment groups were a respective 1.13 for MACE and 2.79 for VTE, but neither RR reached statistical significance.
No difference was seen between the treatment arms in terms of treatment emergent adverse events (RR, 1.05), serious adverse events (RR, 0.92), or study discontinuation because of adverse events (RR, 0.94).
Reassuring Results?
Dr. Ireland and coauthors said the finding should help to reassure clinicians that the short-term use of JAK-STAT inhibitors in patients with dermatological conditions with low cardiovascular risk profiles “appears to be both safe and well tolerated.” They cautioned, however, that “clinicians must remain judicious” when using these medications for longer periods and in high-risk patient populations.
This was a pragmatic meta-analysis that provides useful information for dermatologists, Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, DC, said in an interview.
“When there are safety concerns, I think that’s where data like this are so important to not just allay the fears of practitioners, but also to arm the practitioner with information for when they discuss a possible treatment with a patient,” said Dr. Friedman, who was not involved in the study.
“What’s unique here is that they’re looking at any possible use of JAK inhibitors for dermatological disease,” so this represents patients that dermatologists would be seeing, he added.
“The limitation here is time, we only can say so much about the safety of the medication with the data that we have,” Dr. Friedman said. Almost 4 months is “a good amount of time” to know about the cardiovascular risks, he said, but added, what happens then? Will the risk increase and will patients need to be switched to another medication?
“There’s no line in the sand,” with regard to using a JAK-STAT inhibitor. “If you look at the label, they’re not meant to be used incrementally,” but as ongoing treatment, while considering the needs of the patient and the relative risks and benefits, he said.
With that in mind, “the open label extension studies for all these [JAK-STAT inhibitors] are really, really important to get a sense of ‘do new signals emerge down the road.’ ”
The meta-analysis received no commercial funding. One author of the work reported personal fees from several pharmaceutical companies which were done outside of analysis. Dr. Friedman has received research funding from or acted as a consultant for several pharmaceutical companies including, Incyte, Pfizer, Eli Lily, and AbbVie.
, at least in the short term, say the authors of a new meta-analysis published in JAMA Dermatology.
Considering data on over 17,000 patients with different dermatoses from 45 placebo-controlled randomized clinical trials with an average follow up of 16 weeks, they found there was no significant increase in the occurrence of major adverse cardiovascular events (MACE) or venous thromboembolism (VTE) in people with dermatoses treated with JAK-STAT inhibitors, compared with placebo.
The I² statistic was 0.00% for both MACE and VTE comparing the two arms, indicating that the results were unlikely to be due to chance. There was no increased risk in MACE between those on placebo and those on JAK-STAT inhibitors, with a risk ratio (RR) of 0.47; or for VTE risk, with an RR of 0.46.
Similar findings were obtained when data were analyzed according to the dermatological condition being treated, mechanism of action of the medication, or whether the medication carried a boxed warning.
These data “suggest inconsistency with established sentiments,” that JAK-STAT inhibitors increase the risk for cardiovascular events, Patrick Ireland, MD, of the University of New South Wales, Randwick, Australia, and coauthors wrote in the article. “This may be owing to the limited time frames in which these rare events could be adequately captured, or the ages of enrolled patients being too young to realize the well established heightened risks of developing MACE and VTE,” they suggested.
However, the findings challenge the notion that the cardiovascular complications of these drugs are the same in all patients; dermatological use may not be associated with the same risks as with use for rheumatologic indications.
Class-Wide Boxed Warning
“JAK-STAT [inhibitors] have had some pretty indemnifying data against their use, with the ORAL [Surveillance] study demonstrating increased all-cause mortality, cardiovascular events, venous thromboembolism, and malignancy,” Dr. Ireland said in an interview.
ORAL Surveillance was an open-label, postmarketing trial conducted in patients with rheumatoid arthritis treated with tofacitinib or a tumor necrosis factor (TNF) inhibitor. The results led the US Food and Drug Administration to require information about the risks of serious heart-related events, cancer, blood clots, and death in a boxed warning for JAK-STAT inhibitors in 2022.
“I think it’s important to recognize that these [ORAL Surveillance participants] are very different patients to the typical dermatological patient being treated with a JAK-STAT [inhibitors], with newer studies demonstrating a much safer profile than initially thought,” Dr. Ireland said.
Examining Risk in Dermatological Conditions
The meta-analysis performed by Dr. Ireland and associates focused specifically on the risk for MACE and VTE in patients being treated for dermatological conditions, and included trials published up until June 2023. Only trials that had included a placebo arm were considered; pooled analyses, long-term extension trial data, post hoc analyses, and pediatric-specific trials were excluded.
Most (25) of the trials were phase 2b or phase 3 trials, 18 were phase 2 to 2b, and two were phase 1 trials. The studies included 12,996 participants, mostly with atopic dermatitis or psoriasis, who were treated with JAK-STAT inhibitors, which included baricitinib (2846 patients), tofacitinib (2470), upadacitinib (2218), abrocitinib (1904), and deucravacitinib (1492), among others. There were 4925 patients on placebo.
Overall, MACE — defined as a combined endpoint of acute myocardial infarction, stroke, cardiovascular mortality, heart failure, and unstable angina, as well as arterial embolism — occurred in 13 of the JAK-STAT inhibitor-treated patients and in four of those on placebo. VTE — defined as deep vein thrombosis, pulmonary embolism, and any unusual site thrombosis — was reported in eight JAK-STAT inhibitor-treated patients and in one patient on placebo.
The pooled incidence ratios for MACE and VTE were calculated as 0.20 per 100 person exposure years (PEY) for JAK-STAT inhibitor treatment and 0.13 PEY for placebo. The pooled RRs comparing the two treatment groups were a respective 1.13 for MACE and 2.79 for VTE, but neither RR reached statistical significance.
No difference was seen between the treatment arms in terms of treatment emergent adverse events (RR, 1.05), serious adverse events (RR, 0.92), or study discontinuation because of adverse events (RR, 0.94).
Reassuring Results?
Dr. Ireland and coauthors said the finding should help to reassure clinicians that the short-term use of JAK-STAT inhibitors in patients with dermatological conditions with low cardiovascular risk profiles “appears to be both safe and well tolerated.” They cautioned, however, that “clinicians must remain judicious” when using these medications for longer periods and in high-risk patient populations.
This was a pragmatic meta-analysis that provides useful information for dermatologists, Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, DC, said in an interview.
“When there are safety concerns, I think that’s where data like this are so important to not just allay the fears of practitioners, but also to arm the practitioner with information for when they discuss a possible treatment with a patient,” said Dr. Friedman, who was not involved in the study.
“What’s unique here is that they’re looking at any possible use of JAK inhibitors for dermatological disease,” so this represents patients that dermatologists would be seeing, he added.
“The limitation here is time, we only can say so much about the safety of the medication with the data that we have,” Dr. Friedman said. Almost 4 months is “a good amount of time” to know about the cardiovascular risks, he said, but added, what happens then? Will the risk increase and will patients need to be switched to another medication?
“There’s no line in the sand,” with regard to using a JAK-STAT inhibitor. “If you look at the label, they’re not meant to be used incrementally,” but as ongoing treatment, while considering the needs of the patient and the relative risks and benefits, he said.
With that in mind, “the open label extension studies for all these [JAK-STAT inhibitors] are really, really important to get a sense of ‘do new signals emerge down the road.’ ”
The meta-analysis received no commercial funding. One author of the work reported personal fees from several pharmaceutical companies which were done outside of analysis. Dr. Friedman has received research funding from or acted as a consultant for several pharmaceutical companies including, Incyte, Pfizer, Eli Lily, and AbbVie.
FROM JAMA DERMATOLOGY
Do Statins Offset Venous Thrombosis Risk With Hormone Therapy?
This transcript has been edited for clarity.
This is Dr JoAnn Manson, professor of medicine at Harvard Medical School and Brigham and Women’s Hospital. I’d like to talk with you about a recent report in JAMA Network Open on the subject of whether statin therapy may be able to offset some of the excess risk for venous thromboembolism (VTE) among women taking menopausal hormone therapy.
It’s an important issue because we know that menopausal hormone therapy, especially oral therapy, is linked to an excess risk for VTE, approximately doubling of risk in the randomized clinical trials. There is also emerging evidence from some randomized trials, such as the Jupiter trial, that step therapy may be linked to a reduction in risk. This may be related to anti-inflammatory or antithrombotic effects of statin therapy.
The authors made use of a very large administrative claims database, Optum Health, to look at more than 15 million annual members. They were able to identify 2000 women with a diagnostic code for VTE treatment. The women were between ages 50 and 64 years, and they were compared with 200,000 controls without VTE, matched in 10-to-1 fashion.
About 50% of the women were taking oral hormone therapy, and about 50% took non-oral transdermal or other non-oral formulations of hormone therapy. The odds ratio for VTE was 1.53 among the women who did not also have prescription records for statin therapy. They were able to look at prescribed prescriptions for both the hormone therapy and the statins. Among the women prescribed hormone therapy and also low- to intermediate-dose statins, the odds ratio was 1.29. So that was quite a mitigation of the elevated risk. Among the women taking high-intensity statins, the odds ratio was 1.06, and there was no significant elevation.
We do need more data and more research on this question. One approach would be a meta-analysis of all of the existing randomized trials of hormone therapy in recent years wherein there was increased uptake of statin therapy to look at this question not only for VTE but also for coronary heart disease, stroke, and other CVD outcomes to see whether statin therapy is associated with some attenuation of the excess risk. We also need a targeted randomized trial of statins vs placebo among women who have clear indications for hormone therapy but may be at some increased risk for VTE. That type of trial would be extremely helpful.
These include choosing a transdermal rather than an oral formulation of hormone therapy and using lower doses of hormone therapy. Also, women who are clear candidates for hormone therapy and also for statins, it’s obvious that statins could be co-prescribed. Even among women who are clear candidates for hormone therapy but only intermediate borderline candidates for statin therapy, the prescription of statins might be considered in that clinical scenario to try to mitigate that excess risk for VTE.
JoAnn E. Manson, MD, DrPH, has disclosed the following relevant financial relationships: Received study pill donation and infrastructure support from: Mars Symbioscience (for the COSMOS trial).
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
This is Dr JoAnn Manson, professor of medicine at Harvard Medical School and Brigham and Women’s Hospital. I’d like to talk with you about a recent report in JAMA Network Open on the subject of whether statin therapy may be able to offset some of the excess risk for venous thromboembolism (VTE) among women taking menopausal hormone therapy.
It’s an important issue because we know that menopausal hormone therapy, especially oral therapy, is linked to an excess risk for VTE, approximately doubling of risk in the randomized clinical trials. There is also emerging evidence from some randomized trials, such as the Jupiter trial, that step therapy may be linked to a reduction in risk. This may be related to anti-inflammatory or antithrombotic effects of statin therapy.
The authors made use of a very large administrative claims database, Optum Health, to look at more than 15 million annual members. They were able to identify 2000 women with a diagnostic code for VTE treatment. The women were between ages 50 and 64 years, and they were compared with 200,000 controls without VTE, matched in 10-to-1 fashion.
About 50% of the women were taking oral hormone therapy, and about 50% took non-oral transdermal or other non-oral formulations of hormone therapy. The odds ratio for VTE was 1.53 among the women who did not also have prescription records for statin therapy. They were able to look at prescribed prescriptions for both the hormone therapy and the statins. Among the women prescribed hormone therapy and also low- to intermediate-dose statins, the odds ratio was 1.29. So that was quite a mitigation of the elevated risk. Among the women taking high-intensity statins, the odds ratio was 1.06, and there was no significant elevation.
We do need more data and more research on this question. One approach would be a meta-analysis of all of the existing randomized trials of hormone therapy in recent years wherein there was increased uptake of statin therapy to look at this question not only for VTE but also for coronary heart disease, stroke, and other CVD outcomes to see whether statin therapy is associated with some attenuation of the excess risk. We also need a targeted randomized trial of statins vs placebo among women who have clear indications for hormone therapy but may be at some increased risk for VTE. That type of trial would be extremely helpful.
These include choosing a transdermal rather than an oral formulation of hormone therapy and using lower doses of hormone therapy. Also, women who are clear candidates for hormone therapy and also for statins, it’s obvious that statins could be co-prescribed. Even among women who are clear candidates for hormone therapy but only intermediate borderline candidates for statin therapy, the prescription of statins might be considered in that clinical scenario to try to mitigate that excess risk for VTE.
JoAnn E. Manson, MD, DrPH, has disclosed the following relevant financial relationships: Received study pill donation and infrastructure support from: Mars Symbioscience (for the COSMOS trial).
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
This is Dr JoAnn Manson, professor of medicine at Harvard Medical School and Brigham and Women’s Hospital. I’d like to talk with you about a recent report in JAMA Network Open on the subject of whether statin therapy may be able to offset some of the excess risk for venous thromboembolism (VTE) among women taking menopausal hormone therapy.
It’s an important issue because we know that menopausal hormone therapy, especially oral therapy, is linked to an excess risk for VTE, approximately doubling of risk in the randomized clinical trials. There is also emerging evidence from some randomized trials, such as the Jupiter trial, that step therapy may be linked to a reduction in risk. This may be related to anti-inflammatory or antithrombotic effects of statin therapy.
The authors made use of a very large administrative claims database, Optum Health, to look at more than 15 million annual members. They were able to identify 2000 women with a diagnostic code for VTE treatment. The women were between ages 50 and 64 years, and they were compared with 200,000 controls without VTE, matched in 10-to-1 fashion.
About 50% of the women were taking oral hormone therapy, and about 50% took non-oral transdermal or other non-oral formulations of hormone therapy. The odds ratio for VTE was 1.53 among the women who did not also have prescription records for statin therapy. They were able to look at prescribed prescriptions for both the hormone therapy and the statins. Among the women prescribed hormone therapy and also low- to intermediate-dose statins, the odds ratio was 1.29. So that was quite a mitigation of the elevated risk. Among the women taking high-intensity statins, the odds ratio was 1.06, and there was no significant elevation.
We do need more data and more research on this question. One approach would be a meta-analysis of all of the existing randomized trials of hormone therapy in recent years wherein there was increased uptake of statin therapy to look at this question not only for VTE but also for coronary heart disease, stroke, and other CVD outcomes to see whether statin therapy is associated with some attenuation of the excess risk. We also need a targeted randomized trial of statins vs placebo among women who have clear indications for hormone therapy but may be at some increased risk for VTE. That type of trial would be extremely helpful.
These include choosing a transdermal rather than an oral formulation of hormone therapy and using lower doses of hormone therapy. Also, women who are clear candidates for hormone therapy and also for statins, it’s obvious that statins could be co-prescribed. Even among women who are clear candidates for hormone therapy but only intermediate borderline candidates for statin therapy, the prescription of statins might be considered in that clinical scenario to try to mitigate that excess risk for VTE.
JoAnn E. Manson, MD, DrPH, has disclosed the following relevant financial relationships: Received study pill donation and infrastructure support from: Mars Symbioscience (for the COSMOS trial).
A version of this article appeared on Medscape.com.
Pregnancy in rheumatic disease quadruples risk of cardiovascular events
SAN DIEGO – Pregnant individuals with autoimmune rheumatic diseases (ARDs) are at least four times more likely to experience an acute cardiovascular event (CVE) than are pregnant individuals without these conditions, according to new research presented at the annual meeting of the American College of Rheumatology. Pregnant individuals with primary antiphospholipid syndrome (APS) had a 15-fold increase in CVE risk.
Patients who experienced CVEs were also more likely to experience preterm birth and other adverse pregnancy outcomes (APOs).
Rashmi Dhital, MD, a rheumatology fellow at the University of California, San Diego, and colleagues examined the medical records of pregnant individuals in California who had delivered singleton live-born infants from 2005 to 2020. Using data from the Study of Outcomes in Mothers and Infants (SOMI) database, an administrative population-based birth cohort in California, they identified more than 7 million individuals, 19,340 with ARDs and 7,758 with APS.
They then analyzed how many patients experienced an acute CVE during pregnancy and up to 6 weeks after giving birth.
CVEs occurred in 2.0% of patients with ARDs, 6.9% of individuals with APS, and 0.4% of women without these conditions. CVE risk was four times higher in the ARDs group (adjusted relative risk, 4.1; 95% confidence interval, 3.7-4.5) and nearly 15 times higher in the APS group (aRR, 14.7; 95% CI, 13.5-16.0) than in the comparison group. Patients with systemic lupus erythematosus (SLE) had a sixfold higher risk of CVE, which was further exacerbated by concomitant APS (18-fold higher risk) or lupus nephritis (15-fold higher risk).
Dr. Dhital also classified CVEs as either venous thromboembolism and non-VTE events. Pregnant patients with APS had a high risk for VTE-only CVE (40-fold greater) and a 3.7-fold higher risk of non-VTE events, compared with pregnant patients without these conditions. Patients with SLE along with lupus nephritis had a 20-fold increased risk of VTE-only CVE and an 11-fold higher risk of non-VTE CVE.
Although the study grouped rheumatic diseases together, “lupus is generally driving these results,” Sharon Kolasinski, MD, of the University of Pennsylvania, Philadelphia, noted in an interview. She moderated the plenary session where the research was presented. “If you take out lupus, then what is the risk? That would be an interesting question.”
Between 25% and 30% of all CVEs occurred in the postpartum period, highlighting the importance of close monitoring of cardiovascular risks and events in women with ARDs or APS both during pregnancy and postpartum, Dr. Dhital noted.
Recognizing these risks “can sometimes be challenging due to a lower suspicion of CVE in younger patients, and also symptoms overlap with normal pregnancy,” Dr. Dhital said during her plenary presentation. Working with other clinical teams could help physicians detect these risks in patients.
“It’s important for us to remember that there’s increased risk of cardiovascular events in pregnancy in our patients. It’s uncommon, but it’s not zero,” added Dr. Kolasinski, and this study highlighted when physicians should be more focused about that risk.
Dr. Dhital noted there were some limitations to the study that are inherent in using administrative databases for research that relies on ICD codes, including “the availability of information on disease activity, medications, and labs, which may restrict clinical interpretation.”
SOMI data reinforced by National Inpatient Sample study
The findings were complemented by a study using the National Inpatient Sample database to explore CVE risk in pregnant individuals with various rheumatic diseases. Lead author Karun Shrestha, MD, a resident physician at St. Barnabas Hospital in New York, and colleagues identified delivery hospitalizations from 2016 to 2019 for individuals with SLE, RA, and systemic vasculitis and looked for CVEs including preeclampsia, peripartum cardiomyopathy (PPCM), heart failure, stroke, cardiac arrhythmias, and VTE.
Out of over 3.4 million delivery hospitalizations, researchers identified 5,900 individuals with SLE, 4,895 with RA, and 325 with vasculitis. After adjusting for confounding factors such as race, age, insurance, and other comorbidities, SLE was identified as an independent risk factor for preeclampsia (odds ratio, 1.5; 95% CI, 1.1-2.1), arrhythmia (OR, 3.17; 95% CI, 1.73-5.79), and venous thrombosis (OR, 8.4; 95% CI, 2.9-22.1). Vasculitis was tied to increased risk for preeclampsia (OR, 4.7; 95% CI, 2-11.3), stroke (OR, 513.3; 95% CI, 114-2,284), heart failure (OR, 24.17; 95% CI, 4.68-124.6), and PPCM (OR, 66.7; 95% CI, 8.7-509.4). RA was tied to an increased risk for preeclampsia (OR, 1.5; 95% CI, 1.05-2.1).
Patients with SLE or vasculitis had longer, more costly hospital stays, compared with those without these conditions, and they experienced higher rates of in-hospital mortality. While previous research has demonstrated that patients with SLE have higher risk of cardiac events, there is less literature on CVE risk in pregnancies for vasculitis, Dr. Shrestha said in an interview.
“It’s something to work on,” he said.
Adverse pregnancy outcomes higher with ARDs, APS
In a second abstract also led by Dr. Dhital using SOMI data, researchers found that pregnant individuals with ARDs or APS had a higher risk of experiencing an APO – preterm birth or small-for-gestational age – than individuals without these conditions. CVEs exacerbated that risk, regardless of underlying chronic health conditions.
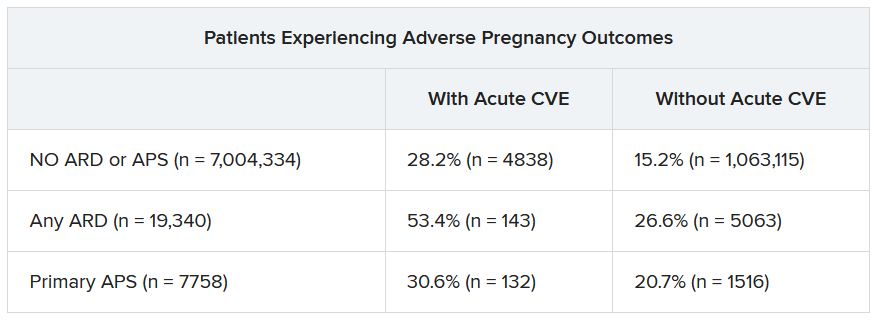
Over half of patients with an ARD and a CVE during pregnancy experienced an APO – most commonly preterm birth. More than one in four pregnant individuals without ARD or APS who experienced a CVE also had an APO.
After differentiating CVEs as either VTE and non-VTE events, patients with ARD and a non-VTE CVE had a fivefold greater risk of early preterm birth (< 32 weeks) and a threefold higher risk of moderate preterm birth (32 to < 34 weeks).
“These findings highlight the need for close monitoring and management of pregnant women, not only for adverse outcomes, but also for cardiovascular risks and events, in order to identify those at the highest risk for adverse outcomes,” the authors wrote. “This need is particularly significant for individuals with ARDs, as 53.4% of our population with an ARD and CVE in pregnancy experienced an APO.”
Dr. Dhital, Dr. Kolasinski, and Dr. Shrestha disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
SAN DIEGO – Pregnant individuals with autoimmune rheumatic diseases (ARDs) are at least four times more likely to experience an acute cardiovascular event (CVE) than are pregnant individuals without these conditions, according to new research presented at the annual meeting of the American College of Rheumatology. Pregnant individuals with primary antiphospholipid syndrome (APS) had a 15-fold increase in CVE risk.
Patients who experienced CVEs were also more likely to experience preterm birth and other adverse pregnancy outcomes (APOs).
Rashmi Dhital, MD, a rheumatology fellow at the University of California, San Diego, and colleagues examined the medical records of pregnant individuals in California who had delivered singleton live-born infants from 2005 to 2020. Using data from the Study of Outcomes in Mothers and Infants (SOMI) database, an administrative population-based birth cohort in California, they identified more than 7 million individuals, 19,340 with ARDs and 7,758 with APS.
They then analyzed how many patients experienced an acute CVE during pregnancy and up to 6 weeks after giving birth.
CVEs occurred in 2.0% of patients with ARDs, 6.9% of individuals with APS, and 0.4% of women without these conditions. CVE risk was four times higher in the ARDs group (adjusted relative risk, 4.1; 95% confidence interval, 3.7-4.5) and nearly 15 times higher in the APS group (aRR, 14.7; 95% CI, 13.5-16.0) than in the comparison group. Patients with systemic lupus erythematosus (SLE) had a sixfold higher risk of CVE, which was further exacerbated by concomitant APS (18-fold higher risk) or lupus nephritis (15-fold higher risk).
Dr. Dhital also classified CVEs as either venous thromboembolism and non-VTE events. Pregnant patients with APS had a high risk for VTE-only CVE (40-fold greater) and a 3.7-fold higher risk of non-VTE events, compared with pregnant patients without these conditions. Patients with SLE along with lupus nephritis had a 20-fold increased risk of VTE-only CVE and an 11-fold higher risk of non-VTE CVE.
Although the study grouped rheumatic diseases together, “lupus is generally driving these results,” Sharon Kolasinski, MD, of the University of Pennsylvania, Philadelphia, noted in an interview. She moderated the plenary session where the research was presented. “If you take out lupus, then what is the risk? That would be an interesting question.”
Between 25% and 30% of all CVEs occurred in the postpartum period, highlighting the importance of close monitoring of cardiovascular risks and events in women with ARDs or APS both during pregnancy and postpartum, Dr. Dhital noted.
Recognizing these risks “can sometimes be challenging due to a lower suspicion of CVE in younger patients, and also symptoms overlap with normal pregnancy,” Dr. Dhital said during her plenary presentation. Working with other clinical teams could help physicians detect these risks in patients.
“It’s important for us to remember that there’s increased risk of cardiovascular events in pregnancy in our patients. It’s uncommon, but it’s not zero,” added Dr. Kolasinski, and this study highlighted when physicians should be more focused about that risk.
Dr. Dhital noted there were some limitations to the study that are inherent in using administrative databases for research that relies on ICD codes, including “the availability of information on disease activity, medications, and labs, which may restrict clinical interpretation.”
SOMI data reinforced by National Inpatient Sample study
The findings were complemented by a study using the National Inpatient Sample database to explore CVE risk in pregnant individuals with various rheumatic diseases. Lead author Karun Shrestha, MD, a resident physician at St. Barnabas Hospital in New York, and colleagues identified delivery hospitalizations from 2016 to 2019 for individuals with SLE, RA, and systemic vasculitis and looked for CVEs including preeclampsia, peripartum cardiomyopathy (PPCM), heart failure, stroke, cardiac arrhythmias, and VTE.
Out of over 3.4 million delivery hospitalizations, researchers identified 5,900 individuals with SLE, 4,895 with RA, and 325 with vasculitis. After adjusting for confounding factors such as race, age, insurance, and other comorbidities, SLE was identified as an independent risk factor for preeclampsia (odds ratio, 1.5; 95% CI, 1.1-2.1), arrhythmia (OR, 3.17; 95% CI, 1.73-5.79), and venous thrombosis (OR, 8.4; 95% CI, 2.9-22.1). Vasculitis was tied to increased risk for preeclampsia (OR, 4.7; 95% CI, 2-11.3), stroke (OR, 513.3; 95% CI, 114-2,284), heart failure (OR, 24.17; 95% CI, 4.68-124.6), and PPCM (OR, 66.7; 95% CI, 8.7-509.4). RA was tied to an increased risk for preeclampsia (OR, 1.5; 95% CI, 1.05-2.1).
Patients with SLE or vasculitis had longer, more costly hospital stays, compared with those without these conditions, and they experienced higher rates of in-hospital mortality. While previous research has demonstrated that patients with SLE have higher risk of cardiac events, there is less literature on CVE risk in pregnancies for vasculitis, Dr. Shrestha said in an interview.
“It’s something to work on,” he said.
Adverse pregnancy outcomes higher with ARDs, APS
In a second abstract also led by Dr. Dhital using SOMI data, researchers found that pregnant individuals with ARDs or APS had a higher risk of experiencing an APO – preterm birth or small-for-gestational age – than individuals without these conditions. CVEs exacerbated that risk, regardless of underlying chronic health conditions.

Over half of patients with an ARD and a CVE during pregnancy experienced an APO – most commonly preterm birth. More than one in four pregnant individuals without ARD or APS who experienced a CVE also had an APO.
After differentiating CVEs as either VTE and non-VTE events, patients with ARD and a non-VTE CVE had a fivefold greater risk of early preterm birth (< 32 weeks) and a threefold higher risk of moderate preterm birth (32 to < 34 weeks).
“These findings highlight the need for close monitoring and management of pregnant women, not only for adverse outcomes, but also for cardiovascular risks and events, in order to identify those at the highest risk for adverse outcomes,” the authors wrote. “This need is particularly significant for individuals with ARDs, as 53.4% of our population with an ARD and CVE in pregnancy experienced an APO.”
Dr. Dhital, Dr. Kolasinski, and Dr. Shrestha disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
SAN DIEGO – Pregnant individuals with autoimmune rheumatic diseases (ARDs) are at least four times more likely to experience an acute cardiovascular event (CVE) than are pregnant individuals without these conditions, according to new research presented at the annual meeting of the American College of Rheumatology. Pregnant individuals with primary antiphospholipid syndrome (APS) had a 15-fold increase in CVE risk.
Patients who experienced CVEs were also more likely to experience preterm birth and other adverse pregnancy outcomes (APOs).
Rashmi Dhital, MD, a rheumatology fellow at the University of California, San Diego, and colleagues examined the medical records of pregnant individuals in California who had delivered singleton live-born infants from 2005 to 2020. Using data from the Study of Outcomes in Mothers and Infants (SOMI) database, an administrative population-based birth cohort in California, they identified more than 7 million individuals, 19,340 with ARDs and 7,758 with APS.
They then analyzed how many patients experienced an acute CVE during pregnancy and up to 6 weeks after giving birth.
CVEs occurred in 2.0% of patients with ARDs, 6.9% of individuals with APS, and 0.4% of women without these conditions. CVE risk was four times higher in the ARDs group (adjusted relative risk, 4.1; 95% confidence interval, 3.7-4.5) and nearly 15 times higher in the APS group (aRR, 14.7; 95% CI, 13.5-16.0) than in the comparison group. Patients with systemic lupus erythematosus (SLE) had a sixfold higher risk of CVE, which was further exacerbated by concomitant APS (18-fold higher risk) or lupus nephritis (15-fold higher risk).
Dr. Dhital also classified CVEs as either venous thromboembolism and non-VTE events. Pregnant patients with APS had a high risk for VTE-only CVE (40-fold greater) and a 3.7-fold higher risk of non-VTE events, compared with pregnant patients without these conditions. Patients with SLE along with lupus nephritis had a 20-fold increased risk of VTE-only CVE and an 11-fold higher risk of non-VTE CVE.
Although the study grouped rheumatic diseases together, “lupus is generally driving these results,” Sharon Kolasinski, MD, of the University of Pennsylvania, Philadelphia, noted in an interview. She moderated the plenary session where the research was presented. “If you take out lupus, then what is the risk? That would be an interesting question.”
Between 25% and 30% of all CVEs occurred in the postpartum period, highlighting the importance of close monitoring of cardiovascular risks and events in women with ARDs or APS both during pregnancy and postpartum, Dr. Dhital noted.
Recognizing these risks “can sometimes be challenging due to a lower suspicion of CVE in younger patients, and also symptoms overlap with normal pregnancy,” Dr. Dhital said during her plenary presentation. Working with other clinical teams could help physicians detect these risks in patients.
“It’s important for us to remember that there’s increased risk of cardiovascular events in pregnancy in our patients. It’s uncommon, but it’s not zero,” added Dr. Kolasinski, and this study highlighted when physicians should be more focused about that risk.
Dr. Dhital noted there were some limitations to the study that are inherent in using administrative databases for research that relies on ICD codes, including “the availability of information on disease activity, medications, and labs, which may restrict clinical interpretation.”
SOMI data reinforced by National Inpatient Sample study
The findings were complemented by a study using the National Inpatient Sample database to explore CVE risk in pregnant individuals with various rheumatic diseases. Lead author Karun Shrestha, MD, a resident physician at St. Barnabas Hospital in New York, and colleagues identified delivery hospitalizations from 2016 to 2019 for individuals with SLE, RA, and systemic vasculitis and looked for CVEs including preeclampsia, peripartum cardiomyopathy (PPCM), heart failure, stroke, cardiac arrhythmias, and VTE.
Out of over 3.4 million delivery hospitalizations, researchers identified 5,900 individuals with SLE, 4,895 with RA, and 325 with vasculitis. After adjusting for confounding factors such as race, age, insurance, and other comorbidities, SLE was identified as an independent risk factor for preeclampsia (odds ratio, 1.5; 95% CI, 1.1-2.1), arrhythmia (OR, 3.17; 95% CI, 1.73-5.79), and venous thrombosis (OR, 8.4; 95% CI, 2.9-22.1). Vasculitis was tied to increased risk for preeclampsia (OR, 4.7; 95% CI, 2-11.3), stroke (OR, 513.3; 95% CI, 114-2,284), heart failure (OR, 24.17; 95% CI, 4.68-124.6), and PPCM (OR, 66.7; 95% CI, 8.7-509.4). RA was tied to an increased risk for preeclampsia (OR, 1.5; 95% CI, 1.05-2.1).
Patients with SLE or vasculitis had longer, more costly hospital stays, compared with those without these conditions, and they experienced higher rates of in-hospital mortality. While previous research has demonstrated that patients with SLE have higher risk of cardiac events, there is less literature on CVE risk in pregnancies for vasculitis, Dr. Shrestha said in an interview.
“It’s something to work on,” he said.
Adverse pregnancy outcomes higher with ARDs, APS
In a second abstract also led by Dr. Dhital using SOMI data, researchers found that pregnant individuals with ARDs or APS had a higher risk of experiencing an APO – preterm birth or small-for-gestational age – than individuals without these conditions. CVEs exacerbated that risk, regardless of underlying chronic health conditions.

Over half of patients with an ARD and a CVE during pregnancy experienced an APO – most commonly preterm birth. More than one in four pregnant individuals without ARD or APS who experienced a CVE also had an APO.
After differentiating CVEs as either VTE and non-VTE events, patients with ARD and a non-VTE CVE had a fivefold greater risk of early preterm birth (< 32 weeks) and a threefold higher risk of moderate preterm birth (32 to < 34 weeks).
“These findings highlight the need for close monitoring and management of pregnant women, not only for adverse outcomes, but also for cardiovascular risks and events, in order to identify those at the highest risk for adverse outcomes,” the authors wrote. “This need is particularly significant for individuals with ARDs, as 53.4% of our population with an ARD and CVE in pregnancy experienced an APO.”
Dr. Dhital, Dr. Kolasinski, and Dr. Shrestha disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT ACR 2023
Review finds no CV or VTE risk signal with use of JAK inhibitors for skin indications
, results from a systematic literature review, and meta-analysis showed.
“There remains a knowledge gap regarding the risk of JAK inhibitor use and VTE and/or MACE in the dermatologic population,” researchers led by Michael S. Garshick, MD, a cardiologist at New York University Langone Health, wrote in their study, which was published online in JAMA Dermatology . “Pooled safety studies suggest that the risk of MACE and VTE may be lower in patients treated with JAK inhibitors for a dermatologic indication than the risk observed in the ORAL Surveillance study, which may be related to the younger age and better health status of those enrolled in trials for dermatologic indications.” The results of that study, which included patients with rheumatoid arthritis only, resulted in the addition of a boxed warning in the labels for topical and oral JAK inhibitors regarding the increased risk of MACE, VTE, serious infections, malignancies, and death .
For the review – thought to be the first to specifically evaluate these risks for dermatologic indications – the researchers searched PubMed and ClinicalTrials.gov from inception through April 1, 2023, for phase 3 dermatology randomized clinical trials (RCTs) to evaluate the risk of MACE, VTE, and all-cause mortality with JAK inhibitors, compared with placebo or an active comparator in the treatment of immune-mediated inflammatory skin diseases. They followed Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines and used a random-effects model and the DerSimonian-Laird method to calculate adverse events with odds ratios.
The database search yielded 35 RCTs with a total of 20,651 patients. Their mean age was 38.5 years, 54% were male, and the mean follow-up time was 4.9 months. Of the 35 trials, most (21) involved patients with atopic dermatitis, followed by psoriasis/psoriatic arthritis (9 trials), alopecia areata (3 trials) and vitiligo (2 trials).
The researchers found no significant difference between JAK inhibitors and placebo/active comparator in composite MACE and all-cause mortality (odds ratio, 0.83; 95% confidence interval, 0.44-1.57) or in VTE (OR, 0.52; 95% CI, 0.26-1.04).
In a secondary analysis, which included additional psoriatic arthritis RCTs, no significant differences between the treatment and placebo/active comparator groups were observed. Similarly, subgroup analyses of oral versus topical JAK inhibitors and a sensitivity analysis that excluded pediatric trials showed no significant differences between patients exposed to JAK inhibitors and those not exposed.
The researchers acknowledged certain limitations of the review, including the lack of access to patient-level data, the fact that most trials only included short-term follow-up, and that the findings have limited generalizability to an older patient population. “It remains unclear if the cardiovascular risks of JAK inhibitors are primarily due to patient level cardiovascular risk factors or are drug mediated,” they concluded. “Dermatologists should carefully select patients and assess baseline cardiovascular risk factors when considering JAK therapy. Cardiovascular risk assessment should continue for the duration of treatment.”
Raj Chovatiya, MD, PhD, assistant professor of dermatology and director of the center for eczema and itch at Northwestern University, Chicago, who was asked to comment on the study results, characterized the findings as reassuring to dermatologists who may be reluctant to initiate therapy with JAK inhibitors based on concerns about safety signals for MACE, VTE, and all-cause mortality.
“These data systematically show that across medications and across conditions, there doesn’t appear to be an increased signal for these events during the short-term, placebo-controlled period which generally spans a few months in most studies,” he told this news organization. The findings, he added, “align well with our clinical experience to date for JAK inhibitor use in inflammatory skin disease. Short-term safety, particularly in relation to boxed warning events such MACE, VTE, and all-cause mortality, have generally been favorable with real-world use. It’s good to have a rigorous statistical analysis to refer to when setting patient expectations.”
However, he noted that these data only examined short-term safety during the placebo or active comparator-controlled periods. “Considering that events like MACE or VTE may take many months or years to manifest, continued long-term data generation is needed to fully answer the question of risk,” he said.
Dr. Garshick disclosed that he received grants from Pfizer and personal fees from Bristol Myers Squibb during the conduct of the study and personal fees from Kiniksa Pharmaceuticals outside the submitted work. Several other coauthors reported having advisory board roles and/or having received funding or support from several pharmaceutical companies. Dr. Chovatiya disclosed that he is a consultant to, a speaker for, investigator, and/or a member of the advisory board for several pharmaceutical companies, including those that develop JAK inhibitors.
, results from a systematic literature review, and meta-analysis showed.
“There remains a knowledge gap regarding the risk of JAK inhibitor use and VTE and/or MACE in the dermatologic population,” researchers led by Michael S. Garshick, MD, a cardiologist at New York University Langone Health, wrote in their study, which was published online in JAMA Dermatology . “Pooled safety studies suggest that the risk of MACE and VTE may be lower in patients treated with JAK inhibitors for a dermatologic indication than the risk observed in the ORAL Surveillance study, which may be related to the younger age and better health status of those enrolled in trials for dermatologic indications.” The results of that study, which included patients with rheumatoid arthritis only, resulted in the addition of a boxed warning in the labels for topical and oral JAK inhibitors regarding the increased risk of MACE, VTE, serious infections, malignancies, and death .
For the review – thought to be the first to specifically evaluate these risks for dermatologic indications – the researchers searched PubMed and ClinicalTrials.gov from inception through April 1, 2023, for phase 3 dermatology randomized clinical trials (RCTs) to evaluate the risk of MACE, VTE, and all-cause mortality with JAK inhibitors, compared with placebo or an active comparator in the treatment of immune-mediated inflammatory skin diseases. They followed Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines and used a random-effects model and the DerSimonian-Laird method to calculate adverse events with odds ratios.
The database search yielded 35 RCTs with a total of 20,651 patients. Their mean age was 38.5 years, 54% were male, and the mean follow-up time was 4.9 months. Of the 35 trials, most (21) involved patients with atopic dermatitis, followed by psoriasis/psoriatic arthritis (9 trials), alopecia areata (3 trials) and vitiligo (2 trials).
The researchers found no significant difference between JAK inhibitors and placebo/active comparator in composite MACE and all-cause mortality (odds ratio, 0.83; 95% confidence interval, 0.44-1.57) or in VTE (OR, 0.52; 95% CI, 0.26-1.04).
In a secondary analysis, which included additional psoriatic arthritis RCTs, no significant differences between the treatment and placebo/active comparator groups were observed. Similarly, subgroup analyses of oral versus topical JAK inhibitors and a sensitivity analysis that excluded pediatric trials showed no significant differences between patients exposed to JAK inhibitors and those not exposed.
The researchers acknowledged certain limitations of the review, including the lack of access to patient-level data, the fact that most trials only included short-term follow-up, and that the findings have limited generalizability to an older patient population. “It remains unclear if the cardiovascular risks of JAK inhibitors are primarily due to patient level cardiovascular risk factors or are drug mediated,” they concluded. “Dermatologists should carefully select patients and assess baseline cardiovascular risk factors when considering JAK therapy. Cardiovascular risk assessment should continue for the duration of treatment.”
Raj Chovatiya, MD, PhD, assistant professor of dermatology and director of the center for eczema and itch at Northwestern University, Chicago, who was asked to comment on the study results, characterized the findings as reassuring to dermatologists who may be reluctant to initiate therapy with JAK inhibitors based on concerns about safety signals for MACE, VTE, and all-cause mortality.
“These data systematically show that across medications and across conditions, there doesn’t appear to be an increased signal for these events during the short-term, placebo-controlled period which generally spans a few months in most studies,” he told this news organization. The findings, he added, “align well with our clinical experience to date for JAK inhibitor use in inflammatory skin disease. Short-term safety, particularly in relation to boxed warning events such MACE, VTE, and all-cause mortality, have generally been favorable with real-world use. It’s good to have a rigorous statistical analysis to refer to when setting patient expectations.”
However, he noted that these data only examined short-term safety during the placebo or active comparator-controlled periods. “Considering that events like MACE or VTE may take many months or years to manifest, continued long-term data generation is needed to fully answer the question of risk,” he said.
Dr. Garshick disclosed that he received grants from Pfizer and personal fees from Bristol Myers Squibb during the conduct of the study and personal fees from Kiniksa Pharmaceuticals outside the submitted work. Several other coauthors reported having advisory board roles and/or having received funding or support from several pharmaceutical companies. Dr. Chovatiya disclosed that he is a consultant to, a speaker for, investigator, and/or a member of the advisory board for several pharmaceutical companies, including those that develop JAK inhibitors.
, results from a systematic literature review, and meta-analysis showed.
“There remains a knowledge gap regarding the risk of JAK inhibitor use and VTE and/or MACE in the dermatologic population,” researchers led by Michael S. Garshick, MD, a cardiologist at New York University Langone Health, wrote in their study, which was published online in JAMA Dermatology . “Pooled safety studies suggest that the risk of MACE and VTE may be lower in patients treated with JAK inhibitors for a dermatologic indication than the risk observed in the ORAL Surveillance study, which may be related to the younger age and better health status of those enrolled in trials for dermatologic indications.” The results of that study, which included patients with rheumatoid arthritis only, resulted in the addition of a boxed warning in the labels for topical and oral JAK inhibitors regarding the increased risk of MACE, VTE, serious infections, malignancies, and death .
For the review – thought to be the first to specifically evaluate these risks for dermatologic indications – the researchers searched PubMed and ClinicalTrials.gov from inception through April 1, 2023, for phase 3 dermatology randomized clinical trials (RCTs) to evaluate the risk of MACE, VTE, and all-cause mortality with JAK inhibitors, compared with placebo or an active comparator in the treatment of immune-mediated inflammatory skin diseases. They followed Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines and used a random-effects model and the DerSimonian-Laird method to calculate adverse events with odds ratios.
The database search yielded 35 RCTs with a total of 20,651 patients. Their mean age was 38.5 years, 54% were male, and the mean follow-up time was 4.9 months. Of the 35 trials, most (21) involved patients with atopic dermatitis, followed by psoriasis/psoriatic arthritis (9 trials), alopecia areata (3 trials) and vitiligo (2 trials).
The researchers found no significant difference between JAK inhibitors and placebo/active comparator in composite MACE and all-cause mortality (odds ratio, 0.83; 95% confidence interval, 0.44-1.57) or in VTE (OR, 0.52; 95% CI, 0.26-1.04).
In a secondary analysis, which included additional psoriatic arthritis RCTs, no significant differences between the treatment and placebo/active comparator groups were observed. Similarly, subgroup analyses of oral versus topical JAK inhibitors and a sensitivity analysis that excluded pediatric trials showed no significant differences between patients exposed to JAK inhibitors and those not exposed.
The researchers acknowledged certain limitations of the review, including the lack of access to patient-level data, the fact that most trials only included short-term follow-up, and that the findings have limited generalizability to an older patient population. “It remains unclear if the cardiovascular risks of JAK inhibitors are primarily due to patient level cardiovascular risk factors or are drug mediated,” they concluded. “Dermatologists should carefully select patients and assess baseline cardiovascular risk factors when considering JAK therapy. Cardiovascular risk assessment should continue for the duration of treatment.”
Raj Chovatiya, MD, PhD, assistant professor of dermatology and director of the center for eczema and itch at Northwestern University, Chicago, who was asked to comment on the study results, characterized the findings as reassuring to dermatologists who may be reluctant to initiate therapy with JAK inhibitors based on concerns about safety signals for MACE, VTE, and all-cause mortality.
“These data systematically show that across medications and across conditions, there doesn’t appear to be an increased signal for these events during the short-term, placebo-controlled period which generally spans a few months in most studies,” he told this news organization. The findings, he added, “align well with our clinical experience to date for JAK inhibitor use in inflammatory skin disease. Short-term safety, particularly in relation to boxed warning events such MACE, VTE, and all-cause mortality, have generally been favorable with real-world use. It’s good to have a rigorous statistical analysis to refer to when setting patient expectations.”
However, he noted that these data only examined short-term safety during the placebo or active comparator-controlled periods. “Considering that events like MACE or VTE may take many months or years to manifest, continued long-term data generation is needed to fully answer the question of risk,” he said.
Dr. Garshick disclosed that he received grants from Pfizer and personal fees from Bristol Myers Squibb during the conduct of the study and personal fees from Kiniksa Pharmaceuticals outside the submitted work. Several other coauthors reported having advisory board roles and/or having received funding or support from several pharmaceutical companies. Dr. Chovatiya disclosed that he is a consultant to, a speaker for, investigator, and/or a member of the advisory board for several pharmaceutical companies, including those that develop JAK inhibitors.
FROM JAMA DERMATOLOGY
Supplemental oxygen fails to improve echocardiographic measures in PE patients
compared with ambient oxygen in a pilot study of 70 individuals.
Anticoagulation monotherapy is the standard of care for patients with intermediate-risk pulmonary embolism (PE), but persistent short-term complication rates may approach 10%, wrote Deisy Barrios, MD, of Hospital Ramón y Cajal (IRYCIS), Madrid, and colleagues. Additional strategies are needed, and the use of supplemental oxygen in non-hypoxemic patients with intermediate-risk PE has not been explored, they said.
In a study published in the journal Chest, the researchers recruited 36 women and 34 men who were non-hypoxemic with stable PE and intermediate risk, defined as echocardiographic RV enlargement. The study recruitment ended prematurely because of the COVID-19 pandemic. The mean age of the participants was 67.3 years. Patients were randomized within 24 hours of hospital admission to anticoagulation plus supplemental oxygen or anticoagulation alone. The groups were similar in echocardiographic mean RV end-diameter and RV/LV ratios at baseline.
The intervention patients received supplemental oxygen at a 35% concentration (7 L/min) continuously for 48 hours via a face mask, and through a nasal cannula during meal times.
The primary outcome was normalization of right ventricle size (defined as an RV/LV diameter ratio less than 1.0 from the subcostal or apical view) at 48 hours after randomization. Secondary outcomes included change in the right ventricle/left ventricle diameter as measured at 48 hours and 7 days after randomization compared to baseline.
The proportion of patients with an RV/LV ratio of 1.0 or less at 48 hours was not significantly different between the intervention and control groups (42.4% vs. 21.6%, P = .08). Similarly, the proportion of patients with an RV/LV ratio of 1.0 or less at 7 days was not significantly different between the groups (76% vs. 70%).
The between-group reduction in RV/LV ratio was significantly greater in the supplemental oxygen group vs. the control group from baseline to 48 hours (0.28 vs. 0.12 P = .02).
However, the within-group mean RV/LV ratio was significantly reduced in both the supplemental oxygen group and the control group compared to baseline at 48 hours and at 7 days after randomization.
None of the patients experienced hemodynamic collapse or recurrent venous thromboembolism during the follow-up period.
The findings were limited by several factors including the small sample size and open-label design, and lack of power to detect clinical outcomes, the researchers noted.
However, the results suggest that although supplemental oxygen had no significant impact of RV/LV normalization, “supplemental oxygen might increase the likelihood of reducing echocardiographic RV dilatation,” and the findings warrant a definitive clinical outcomes trial of supplemental oxygen vs. ambient air to improve outcomes in non-hypoxemic patients with intermediate-risk PE, they concluded.
The study was supported by the Instituto de Salud Carlos III. Dr. Barrios had no financial conflicts to disclose.
compared with ambient oxygen in a pilot study of 70 individuals.
Anticoagulation monotherapy is the standard of care for patients with intermediate-risk pulmonary embolism (PE), but persistent short-term complication rates may approach 10%, wrote Deisy Barrios, MD, of Hospital Ramón y Cajal (IRYCIS), Madrid, and colleagues. Additional strategies are needed, and the use of supplemental oxygen in non-hypoxemic patients with intermediate-risk PE has not been explored, they said.
In a study published in the journal Chest, the researchers recruited 36 women and 34 men who were non-hypoxemic with stable PE and intermediate risk, defined as echocardiographic RV enlargement. The study recruitment ended prematurely because of the COVID-19 pandemic. The mean age of the participants was 67.3 years. Patients were randomized within 24 hours of hospital admission to anticoagulation plus supplemental oxygen or anticoagulation alone. The groups were similar in echocardiographic mean RV end-diameter and RV/LV ratios at baseline.
The intervention patients received supplemental oxygen at a 35% concentration (7 L/min) continuously for 48 hours via a face mask, and through a nasal cannula during meal times.
The primary outcome was normalization of right ventricle size (defined as an RV/LV diameter ratio less than 1.0 from the subcostal or apical view) at 48 hours after randomization. Secondary outcomes included change in the right ventricle/left ventricle diameter as measured at 48 hours and 7 days after randomization compared to baseline.
The proportion of patients with an RV/LV ratio of 1.0 or less at 48 hours was not significantly different between the intervention and control groups (42.4% vs. 21.6%, P = .08). Similarly, the proportion of patients with an RV/LV ratio of 1.0 or less at 7 days was not significantly different between the groups (76% vs. 70%).
The between-group reduction in RV/LV ratio was significantly greater in the supplemental oxygen group vs. the control group from baseline to 48 hours (0.28 vs. 0.12 P = .02).
However, the within-group mean RV/LV ratio was significantly reduced in both the supplemental oxygen group and the control group compared to baseline at 48 hours and at 7 days after randomization.
None of the patients experienced hemodynamic collapse or recurrent venous thromboembolism during the follow-up period.
The findings were limited by several factors including the small sample size and open-label design, and lack of power to detect clinical outcomes, the researchers noted.
However, the results suggest that although supplemental oxygen had no significant impact of RV/LV normalization, “supplemental oxygen might increase the likelihood of reducing echocardiographic RV dilatation,” and the findings warrant a definitive clinical outcomes trial of supplemental oxygen vs. ambient air to improve outcomes in non-hypoxemic patients with intermediate-risk PE, they concluded.
The study was supported by the Instituto de Salud Carlos III. Dr. Barrios had no financial conflicts to disclose.
compared with ambient oxygen in a pilot study of 70 individuals.
Anticoagulation monotherapy is the standard of care for patients with intermediate-risk pulmonary embolism (PE), but persistent short-term complication rates may approach 10%, wrote Deisy Barrios, MD, of Hospital Ramón y Cajal (IRYCIS), Madrid, and colleagues. Additional strategies are needed, and the use of supplemental oxygen in non-hypoxemic patients with intermediate-risk PE has not been explored, they said.
In a study published in the journal Chest, the researchers recruited 36 women and 34 men who were non-hypoxemic with stable PE and intermediate risk, defined as echocardiographic RV enlargement. The study recruitment ended prematurely because of the COVID-19 pandemic. The mean age of the participants was 67.3 years. Patients were randomized within 24 hours of hospital admission to anticoagulation plus supplemental oxygen or anticoagulation alone. The groups were similar in echocardiographic mean RV end-diameter and RV/LV ratios at baseline.
The intervention patients received supplemental oxygen at a 35% concentration (7 L/min) continuously for 48 hours via a face mask, and through a nasal cannula during meal times.
The primary outcome was normalization of right ventricle size (defined as an RV/LV diameter ratio less than 1.0 from the subcostal or apical view) at 48 hours after randomization. Secondary outcomes included change in the right ventricle/left ventricle diameter as measured at 48 hours and 7 days after randomization compared to baseline.
The proportion of patients with an RV/LV ratio of 1.0 or less at 48 hours was not significantly different between the intervention and control groups (42.4% vs. 21.6%, P = .08). Similarly, the proportion of patients with an RV/LV ratio of 1.0 or less at 7 days was not significantly different between the groups (76% vs. 70%).
The between-group reduction in RV/LV ratio was significantly greater in the supplemental oxygen group vs. the control group from baseline to 48 hours (0.28 vs. 0.12 P = .02).
However, the within-group mean RV/LV ratio was significantly reduced in both the supplemental oxygen group and the control group compared to baseline at 48 hours and at 7 days after randomization.
None of the patients experienced hemodynamic collapse or recurrent venous thromboembolism during the follow-up period.
The findings were limited by several factors including the small sample size and open-label design, and lack of power to detect clinical outcomes, the researchers noted.
However, the results suggest that although supplemental oxygen had no significant impact of RV/LV normalization, “supplemental oxygen might increase the likelihood of reducing echocardiographic RV dilatation,” and the findings warrant a definitive clinical outcomes trial of supplemental oxygen vs. ambient air to improve outcomes in non-hypoxemic patients with intermediate-risk PE, they concluded.
The study was supported by the Instituto de Salud Carlos III. Dr. Barrios had no financial conflicts to disclose.
FROM THE JOURNAL CHEST
Decreasing Pulmonary Embolism-Related Mortality
- Centers for Disease Control and Prevention. Data and statistics on venous thromboembolism. Last reviewed June 28, 2023. Accessed July 18, 2023. https://www.cdc.gov/ncbddd/dvt/data.html
- Becattini C et al. Chest. 2016;149(1):192-200. doi:10.1378/chest.15-0808
- Triantafyllou GA et al. Semin Respir Crit Care Med. 2021;42(2):183-198.doi:10.1055/s-0041-1722898
- Ng ACC et al. Respiration. 2013;85(5):408-416. doi:10.1159/000342024
- Phillips AR et al. J Am Heart Assoc. 2021;10(17):e021818. doi:10.1161/JAHA.121.021818
- Wadhera RK et al. J Am Heart Assoc. 2021;10(13):e021117. doi:10.1161/JAHA.121.021117
- Bashir R et al. JACC Cardiovasc Interv. 2022;15(23):2427-2436. doi:10.1016/j.jcin.2022.09.011
- Patel NJ et al. Int J Cardiol. 2019;287:116-117. doi:10.1016/j.ijcard.2019.04.029
- Li X et al. Ann Transl Med. 2021;9(10):838. doi:10.21037/atm-21-975
- Rivera-Lebron BN et al. Chest. 2021;159(1):347-355. doi:10.1016/j.chest.2020.07.065
- Noto JG, Rali P. Pulm Circ. 2022;12(1):e12021. doi:10.1002/pul2.12021
- Snyder DJ et al. Vasc Med. 2023;28(3):222-232. doi:10.1177/1358863X231157441
- Bikdeli B et al. Semin Thromb Hemost. 2023. doi:10.1055/s-0043-1764231
- Fleitas Sosa D et al. Eur Respir Rev. 2022;31(165):220023. doi:10.1183/16000617.0023-2022
- Pulmonary embolism - thrombus removal with catheter-directed therapy (PE-TRACT). ClinicalTrials.gov. Updated July 17, 2023. Accessed July 18, 2023. https://clinicaltrials.gov/ct2/show/NCT05591118
- The PEERLESS study (PEERLESS). ClinicalTrials.gov. Updated Jun 23, 2023. Accessed July 18, 2023. https://clinicaltrials.gov/ct2/show/NCT05111613
- Inari Medical, Inc. Inari Medical announces Peerless II, a randomized controlled trial evaluating clinical outcomes of the FlowTriever® system vs. anticoagulation in pulmonary embolism patients [press release]. Published May 22,2023. Accessed July 18, 2023. https://ir.inarimedical.com/news-releases/news-release-details/inari-medical-announces-peerless-ii-randomized-controlled-trial
- Ultrasound-facilitated, catheter-directed, thrombolysis in intermediate-high risk pulmonary embolism (HI-PEITHO). ClinicalTrials.gov. Updated July 17, 2023. Accessed July 18, 2023. https://clinicaltrials.gov/ct2/show/NCT04790370
- Comparison of two pulmonary embolism treatments. ClinicalTrials.gov. Updated May 31, 2023. Accessed July 18, 2023. https://clinicaltrials.gov/ct2/show/NCT05684796
- Pulmonary Embolism International THrOmbolysis Study-3 (PEITHO-3).ClinicalTrials.gov. Updated June 8, 2023. Accessed July 18, 2023. https://clinicaltrials.gov/ct2/show/NCT04430569
- Study of the long-term safety and outcomes of treating pulmonary embolism with the Indigo Aspiration System. ClinicalTrials.gov. Updated May 11, 2023. Accessed July 18, 2023. https://clinicaltrials.gov/ct2/show/NCT04798261
- Bashir R et al. J Am Coll Cardiol Intv. 2022;15(23):2427-2436. doi:10.1016/j.jcin.2022.09.011
- Centers for Disease Control and Prevention. Data and statistics on venous thromboembolism. Last reviewed June 28, 2023. Accessed July 18, 2023. https://www.cdc.gov/ncbddd/dvt/data.html
- Becattini C et al. Chest. 2016;149(1):192-200. doi:10.1378/chest.15-0808
- Triantafyllou GA et al. Semin Respir Crit Care Med. 2021;42(2):183-198.doi:10.1055/s-0041-1722898
- Ng ACC et al. Respiration. 2013;85(5):408-416. doi:10.1159/000342024
- Phillips AR et al. J Am Heart Assoc. 2021;10(17):e021818. doi:10.1161/JAHA.121.021818
- Wadhera RK et al. J Am Heart Assoc. 2021;10(13):e021117. doi:10.1161/JAHA.121.021117
- Bashir R et al. JACC Cardiovasc Interv. 2022;15(23):2427-2436. doi:10.1016/j.jcin.2022.09.011
- Patel NJ et al. Int J Cardiol. 2019;287:116-117. doi:10.1016/j.ijcard.2019.04.029
- Li X et al. Ann Transl Med. 2021;9(10):838. doi:10.21037/atm-21-975
- Rivera-Lebron BN et al. Chest. 2021;159(1):347-355. doi:10.1016/j.chest.2020.07.065
- Noto JG, Rali P. Pulm Circ. 2022;12(1):e12021. doi:10.1002/pul2.12021
- Snyder DJ et al. Vasc Med. 2023;28(3):222-232. doi:10.1177/1358863X231157441
- Bikdeli B et al. Semin Thromb Hemost. 2023. doi:10.1055/s-0043-1764231
- Fleitas Sosa D et al. Eur Respir Rev. 2022;31(165):220023. doi:10.1183/16000617.0023-2022
- Pulmonary embolism - thrombus removal with catheter-directed therapy (PE-TRACT). ClinicalTrials.gov. Updated July 17, 2023. Accessed July 18, 2023. https://clinicaltrials.gov/ct2/show/NCT05591118
- The PEERLESS study (PEERLESS). ClinicalTrials.gov. Updated Jun 23, 2023. Accessed July 18, 2023. https://clinicaltrials.gov/ct2/show/NCT05111613
- Inari Medical, Inc. Inari Medical announces Peerless II, a randomized controlled trial evaluating clinical outcomes of the FlowTriever® system vs. anticoagulation in pulmonary embolism patients [press release]. Published May 22,2023. Accessed July 18, 2023. https://ir.inarimedical.com/news-releases/news-release-details/inari-medical-announces-peerless-ii-randomized-controlled-trial
- Ultrasound-facilitated, catheter-directed, thrombolysis in intermediate-high risk pulmonary embolism (HI-PEITHO). ClinicalTrials.gov. Updated July 17, 2023. Accessed July 18, 2023. https://clinicaltrials.gov/ct2/show/NCT04790370
- Comparison of two pulmonary embolism treatments. ClinicalTrials.gov. Updated May 31, 2023. Accessed July 18, 2023. https://clinicaltrials.gov/ct2/show/NCT05684796
- Pulmonary Embolism International THrOmbolysis Study-3 (PEITHO-3).ClinicalTrials.gov. Updated June 8, 2023. Accessed July 18, 2023. https://clinicaltrials.gov/ct2/show/NCT04430569
- Study of the long-term safety and outcomes of treating pulmonary embolism with the Indigo Aspiration System. ClinicalTrials.gov. Updated May 11, 2023. Accessed July 18, 2023. https://clinicaltrials.gov/ct2/show/NCT04798261
- Bashir R et al. J Am Coll Cardiol Intv. 2022;15(23):2427-2436. doi:10.1016/j.jcin.2022.09.011
- Centers for Disease Control and Prevention. Data and statistics on venous thromboembolism. Last reviewed June 28, 2023. Accessed July 18, 2023. https://www.cdc.gov/ncbddd/dvt/data.html
- Becattini C et al. Chest. 2016;149(1):192-200. doi:10.1378/chest.15-0808
- Triantafyllou GA et al. Semin Respir Crit Care Med. 2021;42(2):183-198.doi:10.1055/s-0041-1722898
- Ng ACC et al. Respiration. 2013;85(5):408-416. doi:10.1159/000342024
- Phillips AR et al. J Am Heart Assoc. 2021;10(17):e021818. doi:10.1161/JAHA.121.021818
- Wadhera RK et al. J Am Heart Assoc. 2021;10(13):e021117. doi:10.1161/JAHA.121.021117
- Bashir R et al. JACC Cardiovasc Interv. 2022;15(23):2427-2436. doi:10.1016/j.jcin.2022.09.011
- Patel NJ et al. Int J Cardiol. 2019;287:116-117. doi:10.1016/j.ijcard.2019.04.029
- Li X et al. Ann Transl Med. 2021;9(10):838. doi:10.21037/atm-21-975
- Rivera-Lebron BN et al. Chest. 2021;159(1):347-355. doi:10.1016/j.chest.2020.07.065
- Noto JG, Rali P. Pulm Circ. 2022;12(1):e12021. doi:10.1002/pul2.12021
- Snyder DJ et al. Vasc Med. 2023;28(3):222-232. doi:10.1177/1358863X231157441
- Bikdeli B et al. Semin Thromb Hemost. 2023. doi:10.1055/s-0043-1764231
- Fleitas Sosa D et al. Eur Respir Rev. 2022;31(165):220023. doi:10.1183/16000617.0023-2022
- Pulmonary embolism - thrombus removal with catheter-directed therapy (PE-TRACT). ClinicalTrials.gov. Updated July 17, 2023. Accessed July 18, 2023. https://clinicaltrials.gov/ct2/show/NCT05591118
- The PEERLESS study (PEERLESS). ClinicalTrials.gov. Updated Jun 23, 2023. Accessed July 18, 2023. https://clinicaltrials.gov/ct2/show/NCT05111613
- Inari Medical, Inc. Inari Medical announces Peerless II, a randomized controlled trial evaluating clinical outcomes of the FlowTriever® system vs. anticoagulation in pulmonary embolism patients [press release]. Published May 22,2023. Accessed July 18, 2023. https://ir.inarimedical.com/news-releases/news-release-details/inari-medical-announces-peerless-ii-randomized-controlled-trial
- Ultrasound-facilitated, catheter-directed, thrombolysis in intermediate-high risk pulmonary embolism (HI-PEITHO). ClinicalTrials.gov. Updated July 17, 2023. Accessed July 18, 2023. https://clinicaltrials.gov/ct2/show/NCT04790370
- Comparison of two pulmonary embolism treatments. ClinicalTrials.gov. Updated May 31, 2023. Accessed July 18, 2023. https://clinicaltrials.gov/ct2/show/NCT05684796
- Pulmonary Embolism International THrOmbolysis Study-3 (PEITHO-3).ClinicalTrials.gov. Updated June 8, 2023. Accessed July 18, 2023. https://clinicaltrials.gov/ct2/show/NCT04430569
- Study of the long-term safety and outcomes of treating pulmonary embolism with the Indigo Aspiration System. ClinicalTrials.gov. Updated May 11, 2023. Accessed July 18, 2023. https://clinicaltrials.gov/ct2/show/NCT04798261
- Bashir R et al. J Am Coll Cardiol Intv. 2022;15(23):2427-2436. doi:10.1016/j.jcin.2022.09.011
Longer edoxaban may benefit cancer patients with distal DVT
Patients with active cancer and newly diagnosed isolated distal deep vein thrombosis (DVT) who received 12 months of edoxaban (Savaysa) had fewer thrombotic events at 1 year than those who received 3 months of treatment, without significantly increased bleeding, in the ONCO-DVT trial.
However, lead author Yugo Yamashita, MD, of Kyoto University noted that caution is needed when determining anticoagulation strategies in individual patients with distal DVT, especially those with high risk for bleeding.
Dr. Yamashita presented the results at the annual congress of the European Society of Cardiology, and the trial was simultaneously published in the journal Circulation.
“This is the first and only randomized trial to show the superiority of longer duration over shorter duration of anticoagulation therapy for reducing thrombotic events in cancer patients with isolated distal DVT,” he said in a press briefing.
The results provide support for 12 months of edoxaban in patients with active cancer and isolated distal DVD, he said in an email.
However, “considering the risk of bleeding associated with anticoagulation therapy, physicians should make the decision of anticoagulation strategies for these patients based on risk-benefit balance of anticoagulation therapy in individual patients,” he stressed.
The take-home message for clinicians is that, “if you find minor DVT in cancer patients, please be careful, because their thrombotic risk was not low” in this trial, Dr. Yamashita said.
The study was conducted in Japan, so whether or not the results are generalizable to other populations is not clear. “Subgroup analysis based on body weight did not show any signal of different effect,” he noted, which suggests that the main results could be applied to other populations, including the U.S. population. However, “generalizability of the current results should be carried out carefully.”
Caution needed when translating findings into clinical practice
The assigned discussant, Teresa Lopez-Fernandez, MD, from La Paz University Hospital, Madrid, who was co-chairperson of 2022 ESC guidelines on cardio-oncology, noted that the optimal anticoagulation therapy strategy is unclear in patients with cancer and isolated distal DVT.
“2022 ESC guidelines on cardio-oncology and [European Society for Medical Oncology] guidelines from this year,” she said, “are both in agreement that we need to prolong anticoagulation [therapy to prevent venous thromboembolism (VTE)] when active cancer exists, and particularly in patients with metastatic cancer. The problem is that none of this text refers specifically to distal DVT.”
The ONCO-DVT trial sheds light on this, but there are a few points to consider when interpreting the findings.
Major bleeding was slightly increased in the 12-month vs 3-month edoxaban groups, although this was not statistically significant, she noted. Moreover, 75% of the patients were treated with low-dose edoxaban, mainly due to their low weight. Also, bleeding risk probably differs in different cancer types.
“These are important things that we need to keep in mind when we try to transfer this data to [inform] our clinical practice,” Dr. Lopez-Fernandez said.
She drew attention to a recent study based on RIETE registry data that suggests that “isolated distal DVT is a big problem for patients with cancer in comparison with noncancer patients, where it seems it’s a low-risk problem.”
The main takeaways from ONCO-DVT, Dr. Lopez-Fernandez said, are that it confirms that cancer-associated isolated distal DVT is a marker of poor prognosis, and it supports the need for extended anticoagulation in patients with active, ongoing cancer and isolated distal DVT.
However, “we need to be cautious to try to really understand what the bleeding risks of these patients are,” she said, “particularly because it is not always easy to transfer the results from an Asian population to other populations.”
There is also a need for further studies with other doses, with other novel oral anticoagulants, and in patients at high risk for bleeding, in clinical practice.
Dr. Yamashita said that the study suggests that there is a potential benefit of prolonged duration of anticoagulant therapy for some patients with isolated distal DVT, but not all patients should receive this dosing strategy, because some patients may be at high risk for bleeding or VTE recurrence. A subanalysis of data from ONCO-DVT study should shed further light on this.
“We need to individualize our risk stratification,” Dr. Lopez-Fernandez said, adding that notably, “a lot of patients in the 12-month group did not continue with the 12-month treatment,” which may have affected bleeding results. Dr. Yamashita agreed.
Study design and findings
From April 2019 to June 2022, the researchers enrolled and randomly assigned 604 patients with active cancer who had newly diagnosed isolated distal DVT, confirmed by ultrasonography, and were scheduled for DVT treatment with anticoagulation therapy, at 60 centers.
Active cancer was defined as a cancer diagnosis or cancer treatment (surgery, chemotherapy, radiotherapy, etc.) within 6 months of randomization, or current recurrence, local invasion, distant metastases, or hematopoietic malignancy without complete remission.
The most common reasons for ultrasonography were elevated D-dimer levels (62%) and suspected DVT because of symptoms (20%).
The patients had a mean age of 70.8 years and 28% were men. The most common cancer sites were ovaries (14%), uterus (13%), lung (11%), colon (9%), and pancreas (8%), followed by stomach, blood, and breast (each 5%).
The patients were randomly assigned 1:1 to receive 12 months or 3 months of oral edoxaban at a dose of 60 mg once daily or 30 mg once daily in patients with body weight of 60 kg or less, creatinine clearance of 30-50 mL/minute, or concomitant treatment with a potent P-glycoprotein inhibitor.
After excluding 3 patients who withdrew consent, 601 patients were included in the intention-to-treat population: 296 patients in the 12-month edoxaban group and 305 patients in the 3-month edoxaban group.
About 70% of patients had a body weight of 60 kg or less and about 22% had a creatinine clearance less than 50 mL/min. About three quarters received the lower dose of edoxaban.
In the 12-month edoxaban group, 223 patients completed the 1-year follow-up (66 patients had died and 7 were lost to follow-up). In the 3-month edoxaban group, 224 patients completed the 1-year follow-up (77 had died and 4 were lost to follow-up).
In the 12-month edoxaban group, 41% of the patients had discontinued treatment by 12 months. In the 3-month edoxaban group, 41% of patients had discontinued treatment by 3 months.
The primary endpoint – a symptomatic recurrent VTE event or VTE-related death – occurred in 3 of the 222 patients (1.2%) in the 12-month edoxaban group and in 22 of the 210 (8.5%) in the 3-month edoxaban group (odds ratio,0.13; 95% confidence interval, 0.03-0.44, P < .001). There were no VTE-related deaths.
The major secondary endpoint – major bleeding, according to International Society on Thrombosis and Hemostasis criteria – occurred in 28 of the 210 patients (10.2%) in the 12-month edoxaban group and in 22 of the 217 (7.6%) in the 3-month edoxaban group (OR, 1.34; 95% CI, 0.75-2.41, P = NS).
The researchers acknowledged that study limitations include an open-label design, a lower-than-expected primary endpoint rate, and less than high adherence to edoxaban, as well as the need for caution when generalizing the results to other populations.
The study was funded by Daiichi Sankyo. Dr. Yamashita disclosed receiving lecture fees from Bayer Healthcare, Bristol-Myers Squibb, Pfizer, and Daiichi Sankyo, and grant support from Bayer Healthcare and Daiichi Sankyo. Dr. Lopez-Fernandez disclosed receiving speaker fees from Phillips, Janssen, Daiichi Sankyo, Myocardial Solutions, AstraZeneca, Pfizer, Beigene, and Bayer not related to this study.
A version of this article appeared on Medscape.com.
Patients with active cancer and newly diagnosed isolated distal deep vein thrombosis (DVT) who received 12 months of edoxaban (Savaysa) had fewer thrombotic events at 1 year than those who received 3 months of treatment, without significantly increased bleeding, in the ONCO-DVT trial.
However, lead author Yugo Yamashita, MD, of Kyoto University noted that caution is needed when determining anticoagulation strategies in individual patients with distal DVT, especially those with high risk for bleeding.
Dr. Yamashita presented the results at the annual congress of the European Society of Cardiology, and the trial was simultaneously published in the journal Circulation.
“This is the first and only randomized trial to show the superiority of longer duration over shorter duration of anticoagulation therapy for reducing thrombotic events in cancer patients with isolated distal DVT,” he said in a press briefing.
The results provide support for 12 months of edoxaban in patients with active cancer and isolated distal DVD, he said in an email.
However, “considering the risk of bleeding associated with anticoagulation therapy, physicians should make the decision of anticoagulation strategies for these patients based on risk-benefit balance of anticoagulation therapy in individual patients,” he stressed.
The take-home message for clinicians is that, “if you find minor DVT in cancer patients, please be careful, because their thrombotic risk was not low” in this trial, Dr. Yamashita said.
The study was conducted in Japan, so whether or not the results are generalizable to other populations is not clear. “Subgroup analysis based on body weight did not show any signal of different effect,” he noted, which suggests that the main results could be applied to other populations, including the U.S. population. However, “generalizability of the current results should be carried out carefully.”
Caution needed when translating findings into clinical practice
The assigned discussant, Teresa Lopez-Fernandez, MD, from La Paz University Hospital, Madrid, who was co-chairperson of 2022 ESC guidelines on cardio-oncology, noted that the optimal anticoagulation therapy strategy is unclear in patients with cancer and isolated distal DVT.
“2022 ESC guidelines on cardio-oncology and [European Society for Medical Oncology] guidelines from this year,” she said, “are both in agreement that we need to prolong anticoagulation [therapy to prevent venous thromboembolism (VTE)] when active cancer exists, and particularly in patients with metastatic cancer. The problem is that none of this text refers specifically to distal DVT.”
The ONCO-DVT trial sheds light on this, but there are a few points to consider when interpreting the findings.
Major bleeding was slightly increased in the 12-month vs 3-month edoxaban groups, although this was not statistically significant, she noted. Moreover, 75% of the patients were treated with low-dose edoxaban, mainly due to their low weight. Also, bleeding risk probably differs in different cancer types.
“These are important things that we need to keep in mind when we try to transfer this data to [inform] our clinical practice,” Dr. Lopez-Fernandez said.
She drew attention to a recent study based on RIETE registry data that suggests that “isolated distal DVT is a big problem for patients with cancer in comparison with noncancer patients, where it seems it’s a low-risk problem.”
The main takeaways from ONCO-DVT, Dr. Lopez-Fernandez said, are that it confirms that cancer-associated isolated distal DVT is a marker of poor prognosis, and it supports the need for extended anticoagulation in patients with active, ongoing cancer and isolated distal DVT.
However, “we need to be cautious to try to really understand what the bleeding risks of these patients are,” she said, “particularly because it is not always easy to transfer the results from an Asian population to other populations.”
There is also a need for further studies with other doses, with other novel oral anticoagulants, and in patients at high risk for bleeding, in clinical practice.
Dr. Yamashita said that the study suggests that there is a potential benefit of prolonged duration of anticoagulant therapy for some patients with isolated distal DVT, but not all patients should receive this dosing strategy, because some patients may be at high risk for bleeding or VTE recurrence. A subanalysis of data from ONCO-DVT study should shed further light on this.
“We need to individualize our risk stratification,” Dr. Lopez-Fernandez said, adding that notably, “a lot of patients in the 12-month group did not continue with the 12-month treatment,” which may have affected bleeding results. Dr. Yamashita agreed.
Study design and findings
From April 2019 to June 2022, the researchers enrolled and randomly assigned 604 patients with active cancer who had newly diagnosed isolated distal DVT, confirmed by ultrasonography, and were scheduled for DVT treatment with anticoagulation therapy, at 60 centers.
Active cancer was defined as a cancer diagnosis or cancer treatment (surgery, chemotherapy, radiotherapy, etc.) within 6 months of randomization, or current recurrence, local invasion, distant metastases, or hematopoietic malignancy without complete remission.
The most common reasons for ultrasonography were elevated D-dimer levels (62%) and suspected DVT because of symptoms (20%).
The patients had a mean age of 70.8 years and 28% were men. The most common cancer sites were ovaries (14%), uterus (13%), lung (11%), colon (9%), and pancreas (8%), followed by stomach, blood, and breast (each 5%).
The patients were randomly assigned 1:1 to receive 12 months or 3 months of oral edoxaban at a dose of 60 mg once daily or 30 mg once daily in patients with body weight of 60 kg or less, creatinine clearance of 30-50 mL/minute, or concomitant treatment with a potent P-glycoprotein inhibitor.
After excluding 3 patients who withdrew consent, 601 patients were included in the intention-to-treat population: 296 patients in the 12-month edoxaban group and 305 patients in the 3-month edoxaban group.
About 70% of patients had a body weight of 60 kg or less and about 22% had a creatinine clearance less than 50 mL/min. About three quarters received the lower dose of edoxaban.
In the 12-month edoxaban group, 223 patients completed the 1-year follow-up (66 patients had died and 7 were lost to follow-up). In the 3-month edoxaban group, 224 patients completed the 1-year follow-up (77 had died and 4 were lost to follow-up).
In the 12-month edoxaban group, 41% of the patients had discontinued treatment by 12 months. In the 3-month edoxaban group, 41% of patients had discontinued treatment by 3 months.
The primary endpoint – a symptomatic recurrent VTE event or VTE-related death – occurred in 3 of the 222 patients (1.2%) in the 12-month edoxaban group and in 22 of the 210 (8.5%) in the 3-month edoxaban group (odds ratio,0.13; 95% confidence interval, 0.03-0.44, P < .001). There were no VTE-related deaths.
The major secondary endpoint – major bleeding, according to International Society on Thrombosis and Hemostasis criteria – occurred in 28 of the 210 patients (10.2%) in the 12-month edoxaban group and in 22 of the 217 (7.6%) in the 3-month edoxaban group (OR, 1.34; 95% CI, 0.75-2.41, P = NS).
The researchers acknowledged that study limitations include an open-label design, a lower-than-expected primary endpoint rate, and less than high adherence to edoxaban, as well as the need for caution when generalizing the results to other populations.
The study was funded by Daiichi Sankyo. Dr. Yamashita disclosed receiving lecture fees from Bayer Healthcare, Bristol-Myers Squibb, Pfizer, and Daiichi Sankyo, and grant support from Bayer Healthcare and Daiichi Sankyo. Dr. Lopez-Fernandez disclosed receiving speaker fees from Phillips, Janssen, Daiichi Sankyo, Myocardial Solutions, AstraZeneca, Pfizer, Beigene, and Bayer not related to this study.
A version of this article appeared on Medscape.com.
Patients with active cancer and newly diagnosed isolated distal deep vein thrombosis (DVT) who received 12 months of edoxaban (Savaysa) had fewer thrombotic events at 1 year than those who received 3 months of treatment, without significantly increased bleeding, in the ONCO-DVT trial.
However, lead author Yugo Yamashita, MD, of Kyoto University noted that caution is needed when determining anticoagulation strategies in individual patients with distal DVT, especially those with high risk for bleeding.
Dr. Yamashita presented the results at the annual congress of the European Society of Cardiology, and the trial was simultaneously published in the journal Circulation.
“This is the first and only randomized trial to show the superiority of longer duration over shorter duration of anticoagulation therapy for reducing thrombotic events in cancer patients with isolated distal DVT,” he said in a press briefing.
The results provide support for 12 months of edoxaban in patients with active cancer and isolated distal DVD, he said in an email.
However, “considering the risk of bleeding associated with anticoagulation therapy, physicians should make the decision of anticoagulation strategies for these patients based on risk-benefit balance of anticoagulation therapy in individual patients,” he stressed.
The take-home message for clinicians is that, “if you find minor DVT in cancer patients, please be careful, because their thrombotic risk was not low” in this trial, Dr. Yamashita said.
The study was conducted in Japan, so whether or not the results are generalizable to other populations is not clear. “Subgroup analysis based on body weight did not show any signal of different effect,” he noted, which suggests that the main results could be applied to other populations, including the U.S. population. However, “generalizability of the current results should be carried out carefully.”
Caution needed when translating findings into clinical practice
The assigned discussant, Teresa Lopez-Fernandez, MD, from La Paz University Hospital, Madrid, who was co-chairperson of 2022 ESC guidelines on cardio-oncology, noted that the optimal anticoagulation therapy strategy is unclear in patients with cancer and isolated distal DVT.
“2022 ESC guidelines on cardio-oncology and [European Society for Medical Oncology] guidelines from this year,” she said, “are both in agreement that we need to prolong anticoagulation [therapy to prevent venous thromboembolism (VTE)] when active cancer exists, and particularly in patients with metastatic cancer. The problem is that none of this text refers specifically to distal DVT.”
The ONCO-DVT trial sheds light on this, but there are a few points to consider when interpreting the findings.
Major bleeding was slightly increased in the 12-month vs 3-month edoxaban groups, although this was not statistically significant, she noted. Moreover, 75% of the patients were treated with low-dose edoxaban, mainly due to their low weight. Also, bleeding risk probably differs in different cancer types.
“These are important things that we need to keep in mind when we try to transfer this data to [inform] our clinical practice,” Dr. Lopez-Fernandez said.
She drew attention to a recent study based on RIETE registry data that suggests that “isolated distal DVT is a big problem for patients with cancer in comparison with noncancer patients, where it seems it’s a low-risk problem.”
The main takeaways from ONCO-DVT, Dr. Lopez-Fernandez said, are that it confirms that cancer-associated isolated distal DVT is a marker of poor prognosis, and it supports the need for extended anticoagulation in patients with active, ongoing cancer and isolated distal DVT.
However, “we need to be cautious to try to really understand what the bleeding risks of these patients are,” she said, “particularly because it is not always easy to transfer the results from an Asian population to other populations.”
There is also a need for further studies with other doses, with other novel oral anticoagulants, and in patients at high risk for bleeding, in clinical practice.
Dr. Yamashita said that the study suggests that there is a potential benefit of prolonged duration of anticoagulant therapy for some patients with isolated distal DVT, but not all patients should receive this dosing strategy, because some patients may be at high risk for bleeding or VTE recurrence. A subanalysis of data from ONCO-DVT study should shed further light on this.
“We need to individualize our risk stratification,” Dr. Lopez-Fernandez said, adding that notably, “a lot of patients in the 12-month group did not continue with the 12-month treatment,” which may have affected bleeding results. Dr. Yamashita agreed.
Study design and findings
From April 2019 to June 2022, the researchers enrolled and randomly assigned 604 patients with active cancer who had newly diagnosed isolated distal DVT, confirmed by ultrasonography, and were scheduled for DVT treatment with anticoagulation therapy, at 60 centers.
Active cancer was defined as a cancer diagnosis or cancer treatment (surgery, chemotherapy, radiotherapy, etc.) within 6 months of randomization, or current recurrence, local invasion, distant metastases, or hematopoietic malignancy without complete remission.
The most common reasons for ultrasonography were elevated D-dimer levels (62%) and suspected DVT because of symptoms (20%).
The patients had a mean age of 70.8 years and 28% were men. The most common cancer sites were ovaries (14%), uterus (13%), lung (11%), colon (9%), and pancreas (8%), followed by stomach, blood, and breast (each 5%).
The patients were randomly assigned 1:1 to receive 12 months or 3 months of oral edoxaban at a dose of 60 mg once daily or 30 mg once daily in patients with body weight of 60 kg or less, creatinine clearance of 30-50 mL/minute, or concomitant treatment with a potent P-glycoprotein inhibitor.
After excluding 3 patients who withdrew consent, 601 patients were included in the intention-to-treat population: 296 patients in the 12-month edoxaban group and 305 patients in the 3-month edoxaban group.
About 70% of patients had a body weight of 60 kg or less and about 22% had a creatinine clearance less than 50 mL/min. About three quarters received the lower dose of edoxaban.
In the 12-month edoxaban group, 223 patients completed the 1-year follow-up (66 patients had died and 7 were lost to follow-up). In the 3-month edoxaban group, 224 patients completed the 1-year follow-up (77 had died and 4 were lost to follow-up).
In the 12-month edoxaban group, 41% of the patients had discontinued treatment by 12 months. In the 3-month edoxaban group, 41% of patients had discontinued treatment by 3 months.
The primary endpoint – a symptomatic recurrent VTE event or VTE-related death – occurred in 3 of the 222 patients (1.2%) in the 12-month edoxaban group and in 22 of the 210 (8.5%) in the 3-month edoxaban group (odds ratio,0.13; 95% confidence interval, 0.03-0.44, P < .001). There were no VTE-related deaths.
The major secondary endpoint – major bleeding, according to International Society on Thrombosis and Hemostasis criteria – occurred in 28 of the 210 patients (10.2%) in the 12-month edoxaban group and in 22 of the 217 (7.6%) in the 3-month edoxaban group (OR, 1.34; 95% CI, 0.75-2.41, P = NS).
The researchers acknowledged that study limitations include an open-label design, a lower-than-expected primary endpoint rate, and less than high adherence to edoxaban, as well as the need for caution when generalizing the results to other populations.
The study was funded by Daiichi Sankyo. Dr. Yamashita disclosed receiving lecture fees from Bayer Healthcare, Bristol-Myers Squibb, Pfizer, and Daiichi Sankyo, and grant support from Bayer Healthcare and Daiichi Sankyo. Dr. Lopez-Fernandez disclosed receiving speaker fees from Phillips, Janssen, Daiichi Sankyo, Myocardial Solutions, AstraZeneca, Pfizer, Beigene, and Bayer not related to this study.
A version of this article appeared on Medscape.com.
FROM THE ESC CONGRESS 2023