User login
Should clinicians recommend vitamin D for psychiatric patients during COVID-19?
Amid a flurry of conflicting reports concerning the efficacy of vitamin D for COVID-19 patients, a sense of consternation has emerged in the health care sector regarding its overall utility.
Vitamin D plays a critical role in the restorative function of mental health. Low vitamin D levels correlate with mood disorders as well as the development of schizophrenia. In light of the rise in mental health dysfunction and the body of evidence examined to develop this article, we recommend that patients continue to incorporate regular vitamin D supplementation during the course of the pandemic with the goal of preventing deterioration of well-being. Recent studies have generally overlooked the role of vitamin D in mental health by primarily focusing on the immediacy of therapeutic management for medical disorders within the context of COVID-19.
What is the role of vitamin D in human physiology?
Vitamins play an integral role in homeostatic metabolism. Vitamin D, in particular, is intimately responsible for regulating the body’s underlying phosphorus and calcium balance, thereby facilitating bone mineralization.1 As an immunomodulatory hormone, vitamin D coordinates activities across innate and adaptive immune systems, providing defense against autoimmune diseases and miscellaneous infections.2
It is uncommon for people to be affected with vitamin D deficiency in equatorial zones, yet an Indonesian study uncovered low vitamin D effects (hypovitaminosis D) in virtually all of the patients in its COVID-19 case series.3
Likewise, a study conducted in Spain indicated that a whopping 82.2% of the COVID-19 patients endorsed clinically deficient levels of vitamin D, often within the context of severe presentation. Those patients also expressed elevated inflammatory markers, namely, D-dimer and ferritin.4
Comparable studies across the globe continue to support a correlative, if not causative, role for hypovitaminosis D and susceptibility to COVID-19. Mental health awareness entails healthy emotional interactions, preservation of well-being, and the ability to govern one’s thoughts and actions in accordance with societal expectations against the backdrop of ongoing psychosocial stressors. Such awareness helps ensure that people can make resourceful choices and meaningful associations, and can handle stress. We know that mental health is pivotal in dictating one’s overall health. This article provides a detailed exploration of the dynamics of mental health, COVID-19, and vitamin D.
The rationale for vitamin D supplementation therapy in COVID-19
When it comes to respiratory tract infections (RTI) such as COVID-19, influenza, and pneumonia, considerable interest has been generated with respect to the therapeutic efficacy of vitamin D in the acute setting. Vitamin D, as an inflammatory modulator, exerts a protective effect in patients with RTI, especially in those with deviations from baseline vitamin D levels.5
What is the rationale for administering vitamin D supplementation therapy for COVID-19? It has been noted that emergent cases of COVID-19 arise during the autumn months for European countries6 and there is also a firmly established connection between the amount of solar radiation/UV exposure (or the lack thereof) and influenza outbreaks,7 further underscoring the relevance of vitamin D levels. Despite those observations, wholesale implementation of vitamin D therapy should not be used in the acute setting for conditions such as COVID-19 or pneumonia as it is not supported by evidence-based practices. Despite the compound’s inherent antimicrobial actions,8 four randomized clinical trials involving pediatric subjects failed to demonstrate a significantly beneficial response (for example, radiographic resolution) to adjunctive supplementation during the course of acute pneumonia symptomatology.9 Likewise, data collected from a randomized controlled trial confirmed the suspicion that high-dose vitamin D therapy has no tangible effect, tied to mortality or otherwise, on moderate or severe presentations of COVID-19.10
Revisiting vitamin D supplementation therapy for mental health patients with COVID-19
It is clear that recent studies have undermined the overall applicability of vitamin D therapy with respect to acute presentations of COVID-19. However, our team would like to underscore the importance of vitamin D supplementation with respect to maintenance of the integrity of underlying mental health processes.
Numerous studies (for example, cross-sectional, cohort, case-control) have uncovered a statistically significant relationship between vitamin D deficiency and depression, including variants such as postpartum and antepartum depression. It should be noted that the pathophysiology for those variables is not entirely known and that the overall clinical utility of supplementation therapy has not previously been recommended because of existing gaps in the literature.11
In another prospective study involving a relatively small sample size, subjects with seasonal affective disorder (SAD) were either exposed to 10,000 IUs of vitamin D or phototherapy, and depression endpoints were evaluated via the Hamilton Rating Scale for Depression, the SIGH-SAD, and the SAD-8 depression scale. Improvements in 25-hydroxyvitamin D (25-OH D) levels correlated with improvements in depression metrics. However, subjects exposed to phototherapy sessions did not exhibit any meaningful improvements in clinical outcome.12
It is also possible that vitamin D deficiency is reflective of an overall poor nutritional status. People with schizophrenia have frequently been observed to have vitamin D deficiency with more than half of all patients also manifesting symptoms of osteoporosis, a condition that often necessitates vitamin D supplementation. The literature shows that the jury is still out regarding the applicability of vitamin D supplementation for schizophrenia patients, with numerous conflicting studies, including one randomized trial indicating an improvement in positive and negative symptoms as well as in the metabolic profile.13
However, in light of the rather large and growing body of evidence suggesting an increased risk of deterioration, psychological distress, and worsened prognosis during the pandemic coupled with the presence of medical and/or mental health morbidities, it would be sensible for psychiatric patients, especially those with preexisting deviations from baseline vitamin D levels, to consider vitamin D supplementation.
Vitamin D supplementation therapy, as a preventive, but not curative measure – one that is also low cost/high benefit – allows for the patient to be in a much better position from the perspective of her/his general health and nutritional status to tackle the ongoing psychosocial challenges of the pandemic and/or COVID-19 exposure.
Dr. Aman is a faculty member in the biology department at City Colleges of Chicago. She is a postdoctoral researcher at the International Maternal and Child Health Foundation (IMCHF) in Montreal; fellow, medical staff development, American Academy of Medical Management; and master online teacher (MOT) at the University of Illinois at Chicago. Dr. Aman disclosed no relevant relationships. Dr. Islam is a medical writer for the IMCHF and is based in New York. He is a postdoctoral fellow, psychopharmacologist, and a board-certified medical specialist. He disclosed no relevant financial relationships. Dr. Dhillon is a staff neurologist at Brigham and Women’s Hospital in Boston and is affiliated with Sturdy Memorial Hospital in Attleboro, Mass. He is on the speakers bureaus/advisory boards of Biogen, Bristol Myers Squibb, Genzyme, and Teva Neuroscience. Mr. Zaid Ulhaq Choudhry is a research assistant at the IMCHF. He has no disclosures. Dr. Zia Choudhry (Mr. Choudhry’s father) is chief scientific officer and head of the department of mental health and clinical research at the IMCHF. Dr. Choudhry has no disclosures.
References
1. van Driel M and van Leeuwen JPTM. Mol Cellular Endocrinol. 2017;453:46-51.
2. Charoenngam N and Holick MF. Nutrients. 2020 Jul 15;12(7):2097. doi: 103390/nu12072097.
3. Pinzon RT et al. Trop Med Health. 2020 Dec 20;48:102. doi: 10.1186/S41182-020-00277-w.
4. Hernández JL et al. J Clin Endocrinol Metab. 2021 Mar;106(3)e1343-53.
5. Martineau AR et al. BMJ. 2017;356:i6583. doi: 1136/bmj.i6583.
6. Walrand S. Sci Rep. 2021 Jan 21;11(1981). doi: 10.1038/s41598-021-81419-w.
7. Moan J. et al. Dermatoendocrinol. 2009 Nov-Dec;1(6):307-9.
8. Fabri M et al. Sci Transl Med. 2011 Oct 12;3(104):104ra102. doi: 10.1126/scitranslmed.3003045.
9. Slow S et al. Sci Rep. 2018 Sep 14;8(1):13829. doi: 10.1038/s41598-018-32162-2.
10. Berman R. “Study confirms high doses of vitamin D have no effect on COVID-19.” Medical News Today. 2021 May 4.
11. Menon V et al. Indian J Psychol Med. 2020 Jan-Feb;42(1):11-21.
12. Gloth 3rd FM et al. Nutr Health Aging. 1999;3(1):5-7.
13. Cui X et al. Mol Psychiatry. 2021 Jan 26. doi:10.1038/s41380-021-01025-0.
Amid a flurry of conflicting reports concerning the efficacy of vitamin D for COVID-19 patients, a sense of consternation has emerged in the health care sector regarding its overall utility.
Vitamin D plays a critical role in the restorative function of mental health. Low vitamin D levels correlate with mood disorders as well as the development of schizophrenia. In light of the rise in mental health dysfunction and the body of evidence examined to develop this article, we recommend that patients continue to incorporate regular vitamin D supplementation during the course of the pandemic with the goal of preventing deterioration of well-being. Recent studies have generally overlooked the role of vitamin D in mental health by primarily focusing on the immediacy of therapeutic management for medical disorders within the context of COVID-19.
What is the role of vitamin D in human physiology?
Vitamins play an integral role in homeostatic metabolism. Vitamin D, in particular, is intimately responsible for regulating the body’s underlying phosphorus and calcium balance, thereby facilitating bone mineralization.1 As an immunomodulatory hormone, vitamin D coordinates activities across innate and adaptive immune systems, providing defense against autoimmune diseases and miscellaneous infections.2
It is uncommon for people to be affected with vitamin D deficiency in equatorial zones, yet an Indonesian study uncovered low vitamin D effects (hypovitaminosis D) in virtually all of the patients in its COVID-19 case series.3
Likewise, a study conducted in Spain indicated that a whopping 82.2% of the COVID-19 patients endorsed clinically deficient levels of vitamin D, often within the context of severe presentation. Those patients also expressed elevated inflammatory markers, namely, D-dimer and ferritin.4
Comparable studies across the globe continue to support a correlative, if not causative, role for hypovitaminosis D and susceptibility to COVID-19. Mental health awareness entails healthy emotional interactions, preservation of well-being, and the ability to govern one’s thoughts and actions in accordance with societal expectations against the backdrop of ongoing psychosocial stressors. Such awareness helps ensure that people can make resourceful choices and meaningful associations, and can handle stress. We know that mental health is pivotal in dictating one’s overall health. This article provides a detailed exploration of the dynamics of mental health, COVID-19, and vitamin D.
The rationale for vitamin D supplementation therapy in COVID-19
When it comes to respiratory tract infections (RTI) such as COVID-19, influenza, and pneumonia, considerable interest has been generated with respect to the therapeutic efficacy of vitamin D in the acute setting. Vitamin D, as an inflammatory modulator, exerts a protective effect in patients with RTI, especially in those with deviations from baseline vitamin D levels.5
What is the rationale for administering vitamin D supplementation therapy for COVID-19? It has been noted that emergent cases of COVID-19 arise during the autumn months for European countries6 and there is also a firmly established connection between the amount of solar radiation/UV exposure (or the lack thereof) and influenza outbreaks,7 further underscoring the relevance of vitamin D levels. Despite those observations, wholesale implementation of vitamin D therapy should not be used in the acute setting for conditions such as COVID-19 or pneumonia as it is not supported by evidence-based practices. Despite the compound’s inherent antimicrobial actions,8 four randomized clinical trials involving pediatric subjects failed to demonstrate a significantly beneficial response (for example, radiographic resolution) to adjunctive supplementation during the course of acute pneumonia symptomatology.9 Likewise, data collected from a randomized controlled trial confirmed the suspicion that high-dose vitamin D therapy has no tangible effect, tied to mortality or otherwise, on moderate or severe presentations of COVID-19.10
Revisiting vitamin D supplementation therapy for mental health patients with COVID-19
It is clear that recent studies have undermined the overall applicability of vitamin D therapy with respect to acute presentations of COVID-19. However, our team would like to underscore the importance of vitamin D supplementation with respect to maintenance of the integrity of underlying mental health processes.
Numerous studies (for example, cross-sectional, cohort, case-control) have uncovered a statistically significant relationship between vitamin D deficiency and depression, including variants such as postpartum and antepartum depression. It should be noted that the pathophysiology for those variables is not entirely known and that the overall clinical utility of supplementation therapy has not previously been recommended because of existing gaps in the literature.11
In another prospective study involving a relatively small sample size, subjects with seasonal affective disorder (SAD) were either exposed to 10,000 IUs of vitamin D or phototherapy, and depression endpoints were evaluated via the Hamilton Rating Scale for Depression, the SIGH-SAD, and the SAD-8 depression scale. Improvements in 25-hydroxyvitamin D (25-OH D) levels correlated with improvements in depression metrics. However, subjects exposed to phototherapy sessions did not exhibit any meaningful improvements in clinical outcome.12
It is also possible that vitamin D deficiency is reflective of an overall poor nutritional status. People with schizophrenia have frequently been observed to have vitamin D deficiency with more than half of all patients also manifesting symptoms of osteoporosis, a condition that often necessitates vitamin D supplementation. The literature shows that the jury is still out regarding the applicability of vitamin D supplementation for schizophrenia patients, with numerous conflicting studies, including one randomized trial indicating an improvement in positive and negative symptoms as well as in the metabolic profile.13
However, in light of the rather large and growing body of evidence suggesting an increased risk of deterioration, psychological distress, and worsened prognosis during the pandemic coupled with the presence of medical and/or mental health morbidities, it would be sensible for psychiatric patients, especially those with preexisting deviations from baseline vitamin D levels, to consider vitamin D supplementation.
Vitamin D supplementation therapy, as a preventive, but not curative measure – one that is also low cost/high benefit – allows for the patient to be in a much better position from the perspective of her/his general health and nutritional status to tackle the ongoing psychosocial challenges of the pandemic and/or COVID-19 exposure.
Dr. Aman is a faculty member in the biology department at City Colleges of Chicago. She is a postdoctoral researcher at the International Maternal and Child Health Foundation (IMCHF) in Montreal; fellow, medical staff development, American Academy of Medical Management; and master online teacher (MOT) at the University of Illinois at Chicago. Dr. Aman disclosed no relevant relationships. Dr. Islam is a medical writer for the IMCHF and is based in New York. He is a postdoctoral fellow, psychopharmacologist, and a board-certified medical specialist. He disclosed no relevant financial relationships. Dr. Dhillon is a staff neurologist at Brigham and Women’s Hospital in Boston and is affiliated with Sturdy Memorial Hospital in Attleboro, Mass. He is on the speakers bureaus/advisory boards of Biogen, Bristol Myers Squibb, Genzyme, and Teva Neuroscience. Mr. Zaid Ulhaq Choudhry is a research assistant at the IMCHF. He has no disclosures. Dr. Zia Choudhry (Mr. Choudhry’s father) is chief scientific officer and head of the department of mental health and clinical research at the IMCHF. Dr. Choudhry has no disclosures.
References
1. van Driel M and van Leeuwen JPTM. Mol Cellular Endocrinol. 2017;453:46-51.
2. Charoenngam N and Holick MF. Nutrients. 2020 Jul 15;12(7):2097. doi: 103390/nu12072097.
3. Pinzon RT et al. Trop Med Health. 2020 Dec 20;48:102. doi: 10.1186/S41182-020-00277-w.
4. Hernández JL et al. J Clin Endocrinol Metab. 2021 Mar;106(3)e1343-53.
5. Martineau AR et al. BMJ. 2017;356:i6583. doi: 1136/bmj.i6583.
6. Walrand S. Sci Rep. 2021 Jan 21;11(1981). doi: 10.1038/s41598-021-81419-w.
7. Moan J. et al. Dermatoendocrinol. 2009 Nov-Dec;1(6):307-9.
8. Fabri M et al. Sci Transl Med. 2011 Oct 12;3(104):104ra102. doi: 10.1126/scitranslmed.3003045.
9. Slow S et al. Sci Rep. 2018 Sep 14;8(1):13829. doi: 10.1038/s41598-018-32162-2.
10. Berman R. “Study confirms high doses of vitamin D have no effect on COVID-19.” Medical News Today. 2021 May 4.
11. Menon V et al. Indian J Psychol Med. 2020 Jan-Feb;42(1):11-21.
12. Gloth 3rd FM et al. Nutr Health Aging. 1999;3(1):5-7.
13. Cui X et al. Mol Psychiatry. 2021 Jan 26. doi:10.1038/s41380-021-01025-0.
Amid a flurry of conflicting reports concerning the efficacy of vitamin D for COVID-19 patients, a sense of consternation has emerged in the health care sector regarding its overall utility.
Vitamin D plays a critical role in the restorative function of mental health. Low vitamin D levels correlate with mood disorders as well as the development of schizophrenia. In light of the rise in mental health dysfunction and the body of evidence examined to develop this article, we recommend that patients continue to incorporate regular vitamin D supplementation during the course of the pandemic with the goal of preventing deterioration of well-being. Recent studies have generally overlooked the role of vitamin D in mental health by primarily focusing on the immediacy of therapeutic management for medical disorders within the context of COVID-19.
What is the role of vitamin D in human physiology?
Vitamins play an integral role in homeostatic metabolism. Vitamin D, in particular, is intimately responsible for regulating the body’s underlying phosphorus and calcium balance, thereby facilitating bone mineralization.1 As an immunomodulatory hormone, vitamin D coordinates activities across innate and adaptive immune systems, providing defense against autoimmune diseases and miscellaneous infections.2
It is uncommon for people to be affected with vitamin D deficiency in equatorial zones, yet an Indonesian study uncovered low vitamin D effects (hypovitaminosis D) in virtually all of the patients in its COVID-19 case series.3
Likewise, a study conducted in Spain indicated that a whopping 82.2% of the COVID-19 patients endorsed clinically deficient levels of vitamin D, often within the context of severe presentation. Those patients also expressed elevated inflammatory markers, namely, D-dimer and ferritin.4
Comparable studies across the globe continue to support a correlative, if not causative, role for hypovitaminosis D and susceptibility to COVID-19. Mental health awareness entails healthy emotional interactions, preservation of well-being, and the ability to govern one’s thoughts and actions in accordance with societal expectations against the backdrop of ongoing psychosocial stressors. Such awareness helps ensure that people can make resourceful choices and meaningful associations, and can handle stress. We know that mental health is pivotal in dictating one’s overall health. This article provides a detailed exploration of the dynamics of mental health, COVID-19, and vitamin D.
The rationale for vitamin D supplementation therapy in COVID-19
When it comes to respiratory tract infections (RTI) such as COVID-19, influenza, and pneumonia, considerable interest has been generated with respect to the therapeutic efficacy of vitamin D in the acute setting. Vitamin D, as an inflammatory modulator, exerts a protective effect in patients with RTI, especially in those with deviations from baseline vitamin D levels.5
What is the rationale for administering vitamin D supplementation therapy for COVID-19? It has been noted that emergent cases of COVID-19 arise during the autumn months for European countries6 and there is also a firmly established connection between the amount of solar radiation/UV exposure (or the lack thereof) and influenza outbreaks,7 further underscoring the relevance of vitamin D levels. Despite those observations, wholesale implementation of vitamin D therapy should not be used in the acute setting for conditions such as COVID-19 or pneumonia as it is not supported by evidence-based practices. Despite the compound’s inherent antimicrobial actions,8 four randomized clinical trials involving pediatric subjects failed to demonstrate a significantly beneficial response (for example, radiographic resolution) to adjunctive supplementation during the course of acute pneumonia symptomatology.9 Likewise, data collected from a randomized controlled trial confirmed the suspicion that high-dose vitamin D therapy has no tangible effect, tied to mortality or otherwise, on moderate or severe presentations of COVID-19.10
Revisiting vitamin D supplementation therapy for mental health patients with COVID-19
It is clear that recent studies have undermined the overall applicability of vitamin D therapy with respect to acute presentations of COVID-19. However, our team would like to underscore the importance of vitamin D supplementation with respect to maintenance of the integrity of underlying mental health processes.
Numerous studies (for example, cross-sectional, cohort, case-control) have uncovered a statistically significant relationship between vitamin D deficiency and depression, including variants such as postpartum and antepartum depression. It should be noted that the pathophysiology for those variables is not entirely known and that the overall clinical utility of supplementation therapy has not previously been recommended because of existing gaps in the literature.11
In another prospective study involving a relatively small sample size, subjects with seasonal affective disorder (SAD) were either exposed to 10,000 IUs of vitamin D or phototherapy, and depression endpoints were evaluated via the Hamilton Rating Scale for Depression, the SIGH-SAD, and the SAD-8 depression scale. Improvements in 25-hydroxyvitamin D (25-OH D) levels correlated with improvements in depression metrics. However, subjects exposed to phototherapy sessions did not exhibit any meaningful improvements in clinical outcome.12
It is also possible that vitamin D deficiency is reflective of an overall poor nutritional status. People with schizophrenia have frequently been observed to have vitamin D deficiency with more than half of all patients also manifesting symptoms of osteoporosis, a condition that often necessitates vitamin D supplementation. The literature shows that the jury is still out regarding the applicability of vitamin D supplementation for schizophrenia patients, with numerous conflicting studies, including one randomized trial indicating an improvement in positive and negative symptoms as well as in the metabolic profile.13
However, in light of the rather large and growing body of evidence suggesting an increased risk of deterioration, psychological distress, and worsened prognosis during the pandemic coupled with the presence of medical and/or mental health morbidities, it would be sensible for psychiatric patients, especially those with preexisting deviations from baseline vitamin D levels, to consider vitamin D supplementation.
Vitamin D supplementation therapy, as a preventive, but not curative measure – one that is also low cost/high benefit – allows for the patient to be in a much better position from the perspective of her/his general health and nutritional status to tackle the ongoing psychosocial challenges of the pandemic and/or COVID-19 exposure.
Dr. Aman is a faculty member in the biology department at City Colleges of Chicago. She is a postdoctoral researcher at the International Maternal and Child Health Foundation (IMCHF) in Montreal; fellow, medical staff development, American Academy of Medical Management; and master online teacher (MOT) at the University of Illinois at Chicago. Dr. Aman disclosed no relevant relationships. Dr. Islam is a medical writer for the IMCHF and is based in New York. He is a postdoctoral fellow, psychopharmacologist, and a board-certified medical specialist. He disclosed no relevant financial relationships. Dr. Dhillon is a staff neurologist at Brigham and Women’s Hospital in Boston and is affiliated with Sturdy Memorial Hospital in Attleboro, Mass. He is on the speakers bureaus/advisory boards of Biogen, Bristol Myers Squibb, Genzyme, and Teva Neuroscience. Mr. Zaid Ulhaq Choudhry is a research assistant at the IMCHF. He has no disclosures. Dr. Zia Choudhry (Mr. Choudhry’s father) is chief scientific officer and head of the department of mental health and clinical research at the IMCHF. Dr. Choudhry has no disclosures.
References
1. van Driel M and van Leeuwen JPTM. Mol Cellular Endocrinol. 2017;453:46-51.
2. Charoenngam N and Holick MF. Nutrients. 2020 Jul 15;12(7):2097. doi: 103390/nu12072097.
3. Pinzon RT et al. Trop Med Health. 2020 Dec 20;48:102. doi: 10.1186/S41182-020-00277-w.
4. Hernández JL et al. J Clin Endocrinol Metab. 2021 Mar;106(3)e1343-53.
5. Martineau AR et al. BMJ. 2017;356:i6583. doi: 1136/bmj.i6583.
6. Walrand S. Sci Rep. 2021 Jan 21;11(1981). doi: 10.1038/s41598-021-81419-w.
7. Moan J. et al. Dermatoendocrinol. 2009 Nov-Dec;1(6):307-9.
8. Fabri M et al. Sci Transl Med. 2011 Oct 12;3(104):104ra102. doi: 10.1126/scitranslmed.3003045.
9. Slow S et al. Sci Rep. 2018 Sep 14;8(1):13829. doi: 10.1038/s41598-018-32162-2.
10. Berman R. “Study confirms high doses of vitamin D have no effect on COVID-19.” Medical News Today. 2021 May 4.
11. Menon V et al. Indian J Psychol Med. 2020 Jan-Feb;42(1):11-21.
12. Gloth 3rd FM et al. Nutr Health Aging. 1999;3(1):5-7.
13. Cui X et al. Mol Psychiatry. 2021 Jan 26. doi:10.1038/s41380-021-01025-0.
Early interventions for psychosis
Neuroscience research over the past half century has failed to significantly advance the treatment of severe mental illness.1,2 Hence, evidence that a longer duration of untreated psychosis (DUP) aggravates—and early intervention with medication and social supports ameliorates—the long-term adverse consequences of psychotic disorders generated a great deal of interest.3,4 This knowledge led to the development of diverse early intervention services worldwide aimed at this putative “critical window.” It raised the possibility that appropriate interventions could prevent the long-term disability that makes chronic psychosis one of the most debilitating disorders.5,6 However, even beyond the varied cultural and economic confounds, it is difficult to assess, compare, and optimize program effectiveness.7 Obstacles include paucity of sufficiently powered, well-designed randomized controlled trials (RCTs), the absence of diagnostic biomarkers or other prognostic indicators to better account for the inherent heterogeneity in the population and associated outcomes, and the absence of modifiable risk factors that can guide interventions and provide intermediate outcomes.4,8-10
To better appreciate these issues, it is important to distinguish whether a program is designed to prevent psychosis, or to mitigate the effects of psychosis. Two models include the:
- Prevention model, which focuses on young individuals who are not yet overtly psychotic but at high risk
- First-episode recovery model, which focuses on those who have experienced a first episode of psychosis (FEP) but have not yet developed a chronic disorder.
Both models share long-term goals and are hampered by many of the same issues summarized above. They both deviate markedly from the standard medical model by including psychosocial services designed to promote restoration of a self-defined trajectory to greater independence.11-14 The 2 differ, however, in the challenges they must overcome to produce their sample populations and establish effective interventions.10,15,16
In this article, we provide a succinct overview of these issues and a set of recommendations based on a “strength-based” approach. This approach focuses on finding common ground between patients, their support system, and the treatment team in the service of empowering patients to resume responsibility for transition to adulthood.
The prevention model
While most prevention initiatives in medicine rely on the growing ability to target specific pathophysiologic pathways,3 preventing psychosis relies on clinical evidence showing that DUP and early interventions predict a better course of severe mental illness.17 In contrast, initiatives such as normalizing neonatal neuronal pathways are more consistent with the strategy utilized in other fields but have yet to yield a pathophysiologic target for psychosis.3,18
Initial efforts to identify ‘at-risk’ individuals
The prevention model of psychosis is based on the ability to identify young individuals at high risk for developing a psychotic disorder (Figure). The first screening measures were focused on prodromal psychosis (eg, significant loss of function, family history, and “intermittent” and “attenuated” psychotic symptoms). When applied to referred (ie, pre-screened) samples, 30% to 40% of this group who met criteria transitioned to psychosis over the next 1 to 3 years despite antidepressant and psychosocial interventions.19 Comprising 8 academic medical centers, the North American Prodrome Longitudinal Study (NAPLS) produced similar results using the Structured Interview for Prodromal Syndromes (SIPS).17 Thus, 30% to 50% of pre-screened individuals referred by school counselors and mental health professionals met SIPS criteria, and 35% of these individuals transitioned to psychosis over 30 months. The validity of this measure was further supported by the fact that higher baseline levels of unusual thought content, suspicion/paranoia, social impairment, and substance abuse successfully distinguished approximately 80% of those who transitioned to psychosis. The results of this first generation of screening studies were exciting because they seemed to demonstrate that highly concentrated samples of young persons at high risk of developing psychosis could be identified, and that fine-tuning the screening criteria could produce even more enriched samples (ie, positive predictive power).

Initial interventions produced promising results
The development of effective screening measures led to reports of effective treatment interventions. These were largely applied in a clinical staging model that restricted antipsychotic medications to those who failed to improve after receiving potentially “less toxic” interventions (eg, omega-3 polyunsaturated fatty acids and other antioxidants; psychotherapy; cognitive-behavioral therapy [CBT]; family therapy).5 While study designs were typically quasi-experimental, the interventions appeared to dramatically diminish the transition to psychosis (ie, approximately 50%).
Continue to: The first generation...
The first generation of RCTs appeared to confirm these results, although sample sizes were small, and most study designs assessed only a single intervention. Initial meta-analyses of these data reported that both CBT and antipsychotics appeared to prevent approximately one-half of individuals from becoming psychotic at 12 months, and more than one-third at 2 to 4 years, compared with treatment as usual.20
While some researchers challenged the validity of these findings,21-23 the results generated tremendous international enthusiasm and calls for widespread implementation.6 The number of early intervention services (EIS) centers increased dramatically worldwide, and in 2014 the National Institute for Health and Care Excellence released standards for interventions to prevent transition to psychosis.24 These included close monitoring, CBT and family interventions, and avoiding antipsychotics when possible.24
Focusing on sensitivity over specificity
The first generation of studies generated by the prevention model relied on outreach programs or referrals, which produced small samples of carefully selected, pre-screened individuals (Figure, Pre-screened) who were then screened again to establish the high-risk sample.25 While approximately 33% of these individuals became psychotic, the screening process required a very efficient means of eliminating those not at high-risk (given the ultimate target population represented only approximately .5% of young people) (Figure). The pre-screening and screening processes in these first-generation studies were labor-intensive but could only identify approximately 5% of those individuals destined to become psychotic over the next 2 or 3 years. Thus, alternative methods to enhance sensitivity were needed to extend programming to the general population.
Second-generation pre-screening (Figure; Step 1). New pre-screening methods were identified that captured more individuals destined to become psychotic. For example, approximately 90% of this population were registered in health care organizations (eg, health maintenance organizations) and received a psychiatric diagnosis in the year prior to the onset of psychosis (true positives).8 These samples, however, contained a much higher percentage of persons not destined to become psychotic, and somehow the issue of specificity (decreasing false positives) was minimized.8,9 For example, pre-screened samples contained 20 to 50 individuals not destined to become psychotic for each one who did.26 Since screening measures could only eliminate approximately 20% of this group (Figure, Step 2, page 25), second-generation transition rates fell from 30% to 40% to 2% to 10%.27,28
Other pre-screening approaches were introduced, but they also focused on capturing more of those destined to become psychotic (sensitivity) than eliminating those who would not (specificity). For instance, Australia opened more than 100 “Headspace” community centers nationwide designed to promote engagement and self-esteem in youth experiencing anxiety; depression; stress; relationship, work, or school problems; or bullying.13 Most services were free and included mental health staff who screened for psychosis and provided a wide range of services in a destigmatized setting. These methods identified at least an additional 5% to 7% of individuals destined to become psychotic, but to our knowledge, no data have been published on whether they helped eliminate those who did not.
Continue to: Second-generation screening
Second-generation screening (Figure, Step 2). A second screening aims to retain those pre-screened individuals who will become psychotic (ie, minimizing false negatives) while further minimizing those who do not (ie, minimizing false positives). The addition of cognitive, neural (eg, structural MRI; neurophysiologic), and biochemical (eg, inflammatory immune and stress) markers to the risk calculators have produced a sensitivity close to 100%.8,9 Unfortunately, these studies downplayed specificity, which remained approximately 20%.8,9 Specificity is critical not just because of concerns about stigma (ie, labeling people as pre-psychotic when they are not) but also because of the adverse effects of antipsychotic medications and the effects on future program development (interventions are costly and labor-intensive). Also, diluting the pool with individuals not at risk makes it nearly impossible to identify effective interventions (ie, power).27,28
While some studies focused on increasing specificity (to approximately 75%), this leads to an unacceptable loss of sensitivity (from 90% to 60%),29 with 40% of pre-screened individuals who would become psychotic being eliminated from the study population. The addition of other biological markers (eg, salivary cortisol)30 and use of learning health systems may be able to enhance these numbers (initial reports of specificity = 87% and sensitivity = 85%).8,9 This is accomplished by integrating artificial and human intelligence measures of clinical (symptom and neurocognitive measures) and biological (eg, polygenetic risk scores; gray matter volume) variables.31 However, even if these results are replicated, more effective pre-screening measures will be required.
Identifying a suitable sample population for prevention program studies is clearly more complicated than for FEP studies, where one can usually identify many of those in the at-risk population by their first hospitalization for psychotic symptoms. The issues of false positives (eg, substance-induced psychosis) and negatives (eg, slow deterioration, prominent negative symptoms) are important concerns, but proportionately far less significant.
Prevention and FEP interventions
Once a study sample is constituted, 1 to 3 years of treatment interventions are initiated. Interventions for prevention programs typically include CBT directed at attenuated psychosis (eg, reframing or de-catastrophizing unusual thoughts and minimizing distress associated with unusual perceptions); case management to facilitate personal, educational, and vocational goals; and family therapy in single or multi-group formats to educate one’s support system about the risk state and to minimize adverse familial responses.14 Many programs also include supported education or employment services to promote reintegration in age-appropriate activities; group therapy focused on substance abuse and social skills training; cognitive remediation to ameliorate the cognitive dysfunction; and an array of pharmacologic interventions designed to delay or prevent transition to psychosis or to alleviate symptoms. While most interventions are similar, FEP programs have recently included peer support staff. This appears to instill hope in newly diagnosed patients, provide role models, and provide peer supporters an opportunity to use their experiences to help others and earn income.32
The breadth and depth of these services are critical because retention in the program is highly dependent on participant engagement, which in turn is highly dependent on whether the program can help individuals get what they want (eg, friends, employment, education, more autonomy, physical health). The setting and atmosphere of the treatment program and the willingness/ability of staff to meet participants in the community are also important elements.11,12 In this context, the Headspace community centers are having an impact far beyond Australia and may prove to be a particularly good model.13
Continue to: Assessing prevention and FEP interventions
Assessing prevention and FEP interventions
The second generation of studies of prevention programs has not confirmed, let alone extended, the earlier findings and meta-analyses. A 2020 report concluded CBT was still the most promising intervention; it was more effective than control treatments at 12 and 18 months, although not at 6, 24, or 48 months.33 This review included controlled, open-label, and naturalistic studies that assessed family therapy; omega-3 polyunsaturated fatty acids; integrated psychological therapy (a package of interventions that included family education, CBT, social skills training, and cognitive remediation); N-methyl-
While these disappointing findings are at least partly attributable to the methodological challenges described above and in the Figure, other factors may hinder establishing effective interventions. In contrast to FEP studies, those focused on prevention had a very ambitious agenda (eliminating psychosis) and tended to downplay more modest intermediate outcomes. These studies also tended to assess new ideas with small samples rather than pursue promising findings with larger multi-site studies focused on a group of interventions. The authors of a Cochrane review observed “There is the impression that in this whole area there is a triumph of hope over adversity. There is the repeated hope invested in another—often unique—study question and then a study of fewer than 100 participants are completed. This results in the set of comparisons reported here, all 9 of which are too underpowered to really highlight clear differences.”34 To use a baseball analogy, it seems that investigators are “swinging for the fence” when a few singles are what’s really needed.
From the outset, the goals of FEP studies were more modest, largely ignoring the task of developing consensus definitions of recovery that require following patients for up to 5 to 10 years. Instead, they use intermediate endpoints based on adapting treatments that already appeared effective in patients with chronic mental disorders.35 As a consequence, researchers examining FEP demonstrated clear, albeit limited, salutary effects using large multi-site trials and previously established outcome measures.3,10,36 For instance, the Recovery After an Initial Schizophrenia Episode-Early Treatment Program (RAISE-ETP) study was a 2-year, multi-site RCT (N = 404) funded by the National Institute of Mental Health (NIMH). The investigators reported improved indices of social function (eg, quality of life; education and work participation) and total ratings of psychopathology and depression compared with treatment as usual. Furthermore, they established that DUP predicted treatment response.35 The latter finding was underscored by improvement being limited to the 50% with <74 weeks DUP. Annual costs of the program per 1 standard deviation improvement in quality of life were approximately $1,000 for patients with <74 weeks DUP and $40,000 for those with >74 weeks DUP. Concurrent meta-analyses confirmed and extended these findings,16 showing higher remission rates; diminished relapses and hospital admissions; greater engagement in programming; greater involvement in work and school; improved quality of life; and other steps toward recovery. These studies were also able to establish a clear benefit of antipsychotic medications, particularly a high acceptance of long-acting injectable antipsychotic formulations, which promoted adherence and decreased some adverse events37; and early use of clozapine therapy, which improved remission rates and longer-term outcomes.38 Other findings underscored the need to anticipate and address new problems associated with effective antipsychotic therapy (eg, antipsychotic response correlates with weight gain, a particularly intolerable adverse event for this age group).39 Providing pre-emptive strategies such as exercise groups and nutritional education may be necessary to maintain adherence.
Limitations of FEP studies
The effect sizes in these FEP studies were small to medium on outcome measures tracking recovery and associated indicators (eg, global functioning, school/work participation, treatment engagement); the number needed to treat for each of these was >10. There is no clear evidence that recovery programs such as RAISE-ETP actually reduce longer-term disability. Most studies showed disability payments increased while clinical benefits tended to fade over time. In addition, by grouping interventions together, the studies made it difficult to identify effective vs ineffective treatments, let alone determine how best to personalize therapy for participants in future studies.
The next generation of FEP studies
While limited in scope, the results of the recent FEP studies justify a next generation of recovery interventions designed to address these shortcomings and optimize program outcomes.39 Most previous FEP studies were conducted in community mental health center settings, thus eliminating the need to transition services developed in academia into the “real world.” The next generation of NIMH studies will be primarily conducted in analogous settings under the Early Psychosis Intervention Network (EPINET).40 EPINET’s study design echoes that responsible for the stepwise successes in the late 20th century that produced cures for the deadliest childhood cancer, acute lymphoblastic leukemia (ALL). This disease was successfully treated by modifying diverse evidence-based practices without relying on pharmacologic or other major treatment breakthroughs. Despite this, the effort yielded successful personalized interventions that were not obtainable for other severe childhood conditions.40 EPINET hopes to automate much of these stepwise advances with a learning health system. This program relies on data routinely collected in clinical practice to drive the process of scientific discovery. Specifically, it determines the relationships between clinical features, biologic measures, treatment characteristics, and symptomatic and functional outcomes. EPINET aims to accelerate our understanding of biomarkers of psychosis risk and onset, as well as factors associated with recovery and cure. Dashboard displays of outcomes will allow for real-time comparisons within and across early intervention clinics. This in turn identifies performance gaps and drives continuous quality improvement.
Continue to: Barriers to optimizing program efficacy for both models
Barriers to optimizing program efficacy for both models
Unfortunately, there are stark differences between ALL and severe mental disorders that potentially jeopardize the achievement of these aims, despite the advances in data analytic abilities that drive the learning health system. Specifically, the heterogeneity of psychotic illnesses and the absence of reliable prognostic and modifiable risk markers (responsible for failed efforts to enhance treatment of serious mental illness over the last half century1,2,41) are unlikely to be resolved by a learning health system. These measures are vital to determine whether specific interventions are effective, particularly given the absence of a randomized control group in the EPINET/learning health system design. Fortunately, however, the National Institutes for Health has recently initiated the Accelerating Medicines Partnership–Schizophrenia (AMP-SCZ). This approach seeks “promising biological markers that can help identify those at risk of developing schizophrenia as early as possible, track the progression of symptoms and other outcomes and ultimately define targets for treatment development.”42 The Box1,4,9,10,36,41,43-45 describes some of the challenges involved in identifying biomarkers of severe mental illness.
Box
Biomarkers and modifiable risk factors4,9,10,41,43 are at the core of personalized medicine and its ultimate objective (ie, theragnostics). This is the ability to identify the correct intervention for a disorder based on a biomarker of the illness.10,36 The inability to identify biomarkers of severe mental illness is multifactorial but in part may be attributable to “looking in all the wrong places.”41 By focusing on neural processes that generate psychiatric symptomatology, investigators are assuming they can bridge the “mind gap”1 and specifically distinguish between pathological, compensatory, or collateral measures of poorly characterized limbic neural functions.41
It may be more productive to identify a pathological process within the limbic system that produces a medical condition as well as the mental disorder. If one can isolate the pathologic limbic circuit activity responsible for a medical condition, one may be able to reproduce this in animal models and determine whether analogous processes contribute to the core features of the mental illness. Characterization of the aberrant neural circuit in animal models also could yield targets for future therapies. For example, episodic water intoxication in a discrete subset of patients with schizophrenia44 appears to arise from a stress diathesis produced by anterior hippocampal pathology that disrupts regulation of antidiuretic hormone, oxytocin, and hypothalamic-pituitary-adrenal axis secretion. These patients also exhibit psychogenic polydipsia that may be a consequence of the same hippocampal pathology that disrupts ventral striatal and lateral hypothalamic circuits. These circuits, in turn, also modulate motivated behaviors and cognitive processes likely relevant to psychosis.45
A strength-based approach
The absence of sufficiently powered RCTs for prevention studies and the reliance on intermediate outcomes for FEP studies leaves unanswered whether such programs can effectively prevent chronic psychosis at a cost society is willing to pay. Still, substantial evidence indicates that outreach, long-acting injectable antipsychotics, early consideration of clozapine, family therapy, CBT for psychosis/attenuated psychosis, and services focused on competitive employment can preserve social and occupational functioning.16,34 Until these broader questions are more definitively addressed, it seems reasonable to apply what we have learned (Table11,12,35,37-39,46).
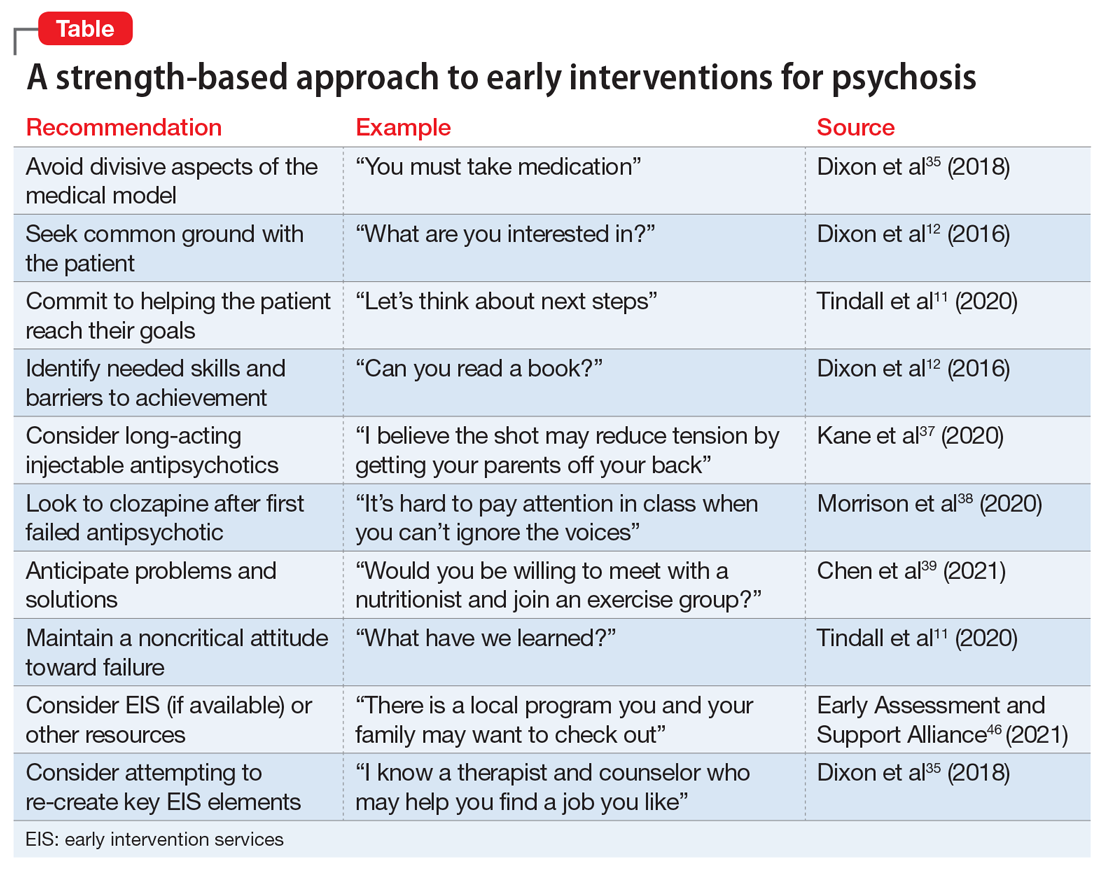
Simply avoiding the most divisive aspects of the medical model that inadvertently promote stigma and undercut self-confidence may help maintain patients’ willingness to learn how best to apply their strengths and manage their limitations.11 The progression to enduring psychotic features (eg, fixed delusions) may reflect ongoing social isolation and alienation. A strength-based approach seeks first to establish common goals (eg, school, work, friends, family support, housing, leaving home) and then works to empower the patient to successfully reach those goals.35 This typically involves giving them the opportunity to fail, avoiding criticism when they do, and focusing on these experiences as learning opportunities from which success can ultimately result.
It is difficult to offer all these services in a typical private practice setting. Instead, it may make more sense to use one of the hundreds of early intervention services programs in the United States.46 If a psychiatric clinician is dedicated to working with this population, it may also be possible to establish ongoing relationships with primary care physicians, family and CBT therapists, family support services (eg, National Alliance on Mental Illness), caseworkers and employment counselors. In essence, a psychiatrist may be able re-create a multidisciplinary effort by taking advantage of the expertise of these various professionals. The challenge is to create a consistent message for patients and families in the absence of regular meetings with the clinical team, although the recent reliance on and improved sophistication of virtual meetings may help. Psychiatrists often play a critical role even when the patient is not prescribed medication, partly because they are most comfortable handling the risks and may have the most comprehensive understanding of the issues at play. When medications are appropriate and patients with FEP are willing to take them, early consideration of long-acting injectable antipsychotics and clozapine may provide better stabilization and diminish the risk of earlier and more frequent relapses.
Bottom Line
Early interventions for psychosis include the prevention model and the first-episode recovery model. It is difficult to assess, compare, and optimize the effectiveness of such programs. Current evidence supports a ‘strength-based’ approach focused on finding common ground between patients, their support system, and the treatment team.
Related Resources
- Early Assessment and Support Alliance. National Early Psychosis Directory. https://easacommunity.org/nationaldirectory.php
- Kane JM, Robinson DG, Schooler NR, et al. Comprehensive versus usual community care for first-episode psychosis: 2-year outcomes from the NIMH RAISE Early Treatment Program. Am J Psychiatry. 2016 ;173(4):362-372
Drug Brand Name
Clozapine • Clozaril
1. Hyman SE. Revolution stalled. Sci Transl Med. 2012;4(155):155cm11. doi: 10.1126/scitranslmed.3003142
2. Harrington A. Mind fixers: psychiatry’s troubled search for the biology of mental illness. W.W. Norton & Company; 2019.
3. Millan MJ, Andrieux A, Bartzokis G, et al. Altering the course of schizophrenia: progress and perspectives. Nat Rev Drug Discov. 2016;15(7):485-515.
4. Lieberman JA, Small SA, Girgis RR. Early detection and preventive intervention in schizophrenia: from fantasy to reality. Am J Psychiatry. 2019;176(10):794-810.
5. McGorry PD, Nelson B, Nordentoft M, et al. Intervention in individuals at ultra-high risk for psychosis: a review and future directions. J Clin Psychiatry. 2009;70(9):1206-1212.
6. Csillag C, Nordentoft M, Mizuno M, et al. Early intervention in psychosis: From clinical intervention to health system implementation. Early Interv Psychiatry. 2018;12(4):757-764.
7. McGorry PD, Ratheesh A, O’Donoghue B. Early intervention—an implementation challenge for 21st century mental health care. JAMA Psychiatry. 2018;75(6):545-546.
8. Rosenheck R. Toward dissemination of secondary prevention for psychosis. Am J Psychiatry. 2018;175(5):393-394.
9. Fusar-Poli P, Salazar de Pablo G, Correll CU, et al. Prevention of psychosis: advances in detection, prognosis, and intervention. JAMA Psychiatry. 2020;77(7):755-765.
10. Oliver D, Reilly TJ, Baccaredda Boy O, et al. What causes the onset of psychosis in individuals at clinical high risk? A meta-analysis of risk and protective factors. Schizophr Bull. 2020;46(1):110-120.
11. Tindall R, Simmons M, Allott K, et al. Disengagement processes within an early intervention service for first-episode psychosis: a longitudinal, qualitative, multi-perspective study. Front Psychiatry. 2020;11:565-565.
12. Dixon LB, Holoshitz Y, Nossel I. Treatment engagement of individuals experiencing mental illness: review and update. World Psychiatry. 2016;15(1):13-20.
13. Rickwood D, Paraskakis M, Quin D, et al. Australia’s innovation in youth mental health care: The headspace centre model. Early Interv Psychiatry. 2019;13(1):159-166.
14. Woodberry KA, Shapiro DI, Bryant C, et al. Progress and future directions in research on the psychosis prodrome: a review for clinicians. Harv Rev Psychiatry. 2016;24(2):87-103.
15. Gupta T, Mittal VA. Advances in clinical staging, early intervention, and the prevention of psychosis. F1000Res. 2019;8:F1000 Faculty Rev-2027. doi: 10.12688/f1000research.20346.1
16. Correll CU, Galling B, Pawar A, et al. Comparison of early intervention services vs treatment as usual for early-phase psychosis: a systematic review, meta-analysis, and meta-regression. JAMA Psychiatry. 2018;75(6):555-565.
17. Cannon TD, Cadenhead K, Cornblatt B, et al. Prediction of psychosis in youth at high clinical risk: a multisite longitudinal study in North America. Arch Gen Psychiatry. 2008;65(1):28-37.
18. Sommer IE, Bearden CE, van Dellen E, et al. Early interventions in risk groups for schizophrenia: what are we waiting for? NPJ Schizophr. 2016;2(1):16003-16003.
19. McGorry PD, Nelson B. Clinical high risk for psychosis—not seeing the trees for the wood. JAMA Psychiatry. 2020;77(7):559-560.
20. van der Gaag M, Smit F, Bechdolf A, et al. Preventing a first episode of psychosis: meta-analysis of randomized controlled prevention trials of 12 month and longer-term follow-ups. Schizophr Res. 2013;149(1):56-62.
21. Marshall M, Rathbone J. Early intervention for psychosis. Cochrane Database Syst Rev. 2011;(6):CD004718. doi: 10.1002/14651858.CD004718.pub3
22. Heinssen RK, Insel TR. Preventing the onset of psychosis: not quite there yet. Schizophr Bull. 2015;41(1):28-29.
23. Amos AJ. Evidence that treatment prevents transition to psychosis in ultra-high-risk patients remains questionable. Schizophr Res. 2014;153(1):240.
24. National Institute for Health and Care Excellence. Psychosis and schizophrenia in adults: prevention and management. Clinical guideline [CG178]. 1.3.7 How to deliver psychological interventions. Published February 12, 2014. Updated March 1, 2014. Accessed August 30, 2021. https://www.nice.org.uk/guidance/cg178/chapter/recommendations#how-to-deliver-psychological-interventions
25. Fusar-Poli P, Werbeloff N, Rutigliano G, et al. Transdiagnostic risk calculator for the automatic detection of individuals at risk and the prediction of psychosis: second replication in an independent National Health Service Trust. Schizophr Bull. 2019;45(3):562-570.
26. Fusar-Poli P, Oliver D, Spada G, et al. The case for improved transdiagnostic detection of first-episode psychosis: electronic health record cohort study. Schizophr Res. 2021;228:547-554.
27. Fusar-Poli P. Negative psychosis prevention trials. JAMA Psychiatry. 2017;74(6):651.
28. Cuijpers P, Smit F, Furukawa TA. Most at‐risk individuals will not develop a mental disorder: the limited predictive strength of risk factors. World Psychiatry. 2021;20(2):224-225.
29. Carrión RE, Cornblatt BA, Burton CZ, et al. Personalized prediction of psychosis: external validation of the NAPLS-2 psychosis risk calculator with the EDIPPP Project. Am J Psychiatry. 2016;173(10):989-996.
30. Worthington MA, Walker EF, Addington J, et al. Incorporating cortisol into the NAPLS2 individualized risk calculator for prediction of psychosis. Schizophr Res. 2021;227:95-100.
31. Koutsouleris N, Dwyer DB, Degenhardt F, et al. Multimodal machine learning workflows for prediction of psychosis in patients with clinical high-risk syndromes and recent-onset depression. JAMA Psychiatry. 2021;78(2):195-209.
32. Simmons MB, Grace D, Fava NJ, et al. The experiences of youth mental health peer workers over time: a qualitative study with longitudinal analysis. Community Ment Health J. 2020;56(5):906-914.
33. Devoe DJ, Farris MS, Townes P, et al. Interventions and transition in youth at risk of psychosis: a systematic review and meta-analyses. J Clin Psychiatry. 2020;81(3):17r12053. doi: 10.4088/JCP.17r12053
34. Bosnjak Kuharic D, Kekin I, Hew J, et al. Interventions for prodromal stage of psychosis. Cochrane Database Syst Rev. 2019;2019(11):CD012236
35. Dixon LB, Goldman HH, Srihari VH, et al. Transforming the treatment of schizophrenia in the United States: The RAISE Initiative. Annu Rev Clin Psychol. 2018;14:237-258.
36. Friedman-Yakoobian MS, Parrish EM, Eack SM, et al. Neurocognitive and social cognitive training for youth at clinical high risk (CHR) for psychosis: a randomized controlled feasibility trial. Schizophr Res. 2020;S0920-9964(20)30461-8. doi: 10.1016/j.schres.2020.09.005
37. Kane JM, Schooler NR, Marcy P, et al. Effect of long-acting injectable antipsychotics vs usual care on time to first hospitalization in early-phase schizophrenia: a randomized clinical trial. JAMA Psychiatry. 2020;77(12):1217-1224.
38. Morrison AP, Pyle M, Maughan D, et al. Antipsychotic medication versus psychological intervention versus a combination of both in adolescents with first-episode psychosis (MAPS): a multicentre, three-arm, randomised controlled pilot and feasibility study. Lancet Psychiatry. 2020;7(9):788-800.
39. Chen YQ, Li XR, Zhang L, et al. Therapeutic response is associated with antipsychotic-induced weight gain in drug-naive first-episode patients with schizophrenia: an 8-week prospective study. J Clin Psychiatry. 2021;82(3):20m13469. doi: 10.4088/JCP.20m13469
40. Insel TR. RAISE-ing our expectations for first-episode psychosis. Am J Psychiatry. 2016;173(4):311-312.
41. Tandon R, Goldman M. Overview of neurobiology. In: Janicak PG, Marder SR, Tandon R, et al, eds. Schizophrenia: recent advances in diagnosis and treatment. Springer; 2014:27-33.
42. National Institutes of Health. Accelerating Medicines Partnership. Schizophrenia. Accessed August 30, 2021. https://www.nih.gov/research-training/accelerating-medicines-partnership-amp/schizophrenia
43. Guloksuz S, van Os J. The slow death of the concept of schizophrenia and the painful birth of the psychosis spectrum. Psychol Med. 2018;48(2):229-244.
44. Christ-Crain M, Bichet DG, Fenske WK, et al. Diabetes insipidus. Nat Rev Dis Primers. 2019;5(1):54.
45. Ahmadi L, Goldman MB. Primary polydipsia: update. Best Pract Res Clin Endocrinol Metab. 2020;34(5):101469. doi: 10.1016/j.beem.2020.101469
46. Early Assessment and Support Alliance. National Early Psychosis Directory. Accessed August 30, 2021. https://easacommunity.org/national-directory.php
Neuroscience research over the past half century has failed to significantly advance the treatment of severe mental illness.1,2 Hence, evidence that a longer duration of untreated psychosis (DUP) aggravates—and early intervention with medication and social supports ameliorates—the long-term adverse consequences of psychotic disorders generated a great deal of interest.3,4 This knowledge led to the development of diverse early intervention services worldwide aimed at this putative “critical window.” It raised the possibility that appropriate interventions could prevent the long-term disability that makes chronic psychosis one of the most debilitating disorders.5,6 However, even beyond the varied cultural and economic confounds, it is difficult to assess, compare, and optimize program effectiveness.7 Obstacles include paucity of sufficiently powered, well-designed randomized controlled trials (RCTs), the absence of diagnostic biomarkers or other prognostic indicators to better account for the inherent heterogeneity in the population and associated outcomes, and the absence of modifiable risk factors that can guide interventions and provide intermediate outcomes.4,8-10
To better appreciate these issues, it is important to distinguish whether a program is designed to prevent psychosis, or to mitigate the effects of psychosis. Two models include the:
- Prevention model, which focuses on young individuals who are not yet overtly psychotic but at high risk
- First-episode recovery model, which focuses on those who have experienced a first episode of psychosis (FEP) but have not yet developed a chronic disorder.
Both models share long-term goals and are hampered by many of the same issues summarized above. They both deviate markedly from the standard medical model by including psychosocial services designed to promote restoration of a self-defined trajectory to greater independence.11-14 The 2 differ, however, in the challenges they must overcome to produce their sample populations and establish effective interventions.10,15,16
In this article, we provide a succinct overview of these issues and a set of recommendations based on a “strength-based” approach. This approach focuses on finding common ground between patients, their support system, and the treatment team in the service of empowering patients to resume responsibility for transition to adulthood.
The prevention model
While most prevention initiatives in medicine rely on the growing ability to target specific pathophysiologic pathways,3 preventing psychosis relies on clinical evidence showing that DUP and early interventions predict a better course of severe mental illness.17 In contrast, initiatives such as normalizing neonatal neuronal pathways are more consistent with the strategy utilized in other fields but have yet to yield a pathophysiologic target for psychosis.3,18
Initial efforts to identify ‘at-risk’ individuals
The prevention model of psychosis is based on the ability to identify young individuals at high risk for developing a psychotic disorder (Figure). The first screening measures were focused on prodromal psychosis (eg, significant loss of function, family history, and “intermittent” and “attenuated” psychotic symptoms). When applied to referred (ie, pre-screened) samples, 30% to 40% of this group who met criteria transitioned to psychosis over the next 1 to 3 years despite antidepressant and psychosocial interventions.19 Comprising 8 academic medical centers, the North American Prodrome Longitudinal Study (NAPLS) produced similar results using the Structured Interview for Prodromal Syndromes (SIPS).17 Thus, 30% to 50% of pre-screened individuals referred by school counselors and mental health professionals met SIPS criteria, and 35% of these individuals transitioned to psychosis over 30 months. The validity of this measure was further supported by the fact that higher baseline levels of unusual thought content, suspicion/paranoia, social impairment, and substance abuse successfully distinguished approximately 80% of those who transitioned to psychosis. The results of this first generation of screening studies were exciting because they seemed to demonstrate that highly concentrated samples of young persons at high risk of developing psychosis could be identified, and that fine-tuning the screening criteria could produce even more enriched samples (ie, positive predictive power).

Initial interventions produced promising results
The development of effective screening measures led to reports of effective treatment interventions. These were largely applied in a clinical staging model that restricted antipsychotic medications to those who failed to improve after receiving potentially “less toxic” interventions (eg, omega-3 polyunsaturated fatty acids and other antioxidants; psychotherapy; cognitive-behavioral therapy [CBT]; family therapy).5 While study designs were typically quasi-experimental, the interventions appeared to dramatically diminish the transition to psychosis (ie, approximately 50%).
Continue to: The first generation...
The first generation of RCTs appeared to confirm these results, although sample sizes were small, and most study designs assessed only a single intervention. Initial meta-analyses of these data reported that both CBT and antipsychotics appeared to prevent approximately one-half of individuals from becoming psychotic at 12 months, and more than one-third at 2 to 4 years, compared with treatment as usual.20
While some researchers challenged the validity of these findings,21-23 the results generated tremendous international enthusiasm and calls for widespread implementation.6 The number of early intervention services (EIS) centers increased dramatically worldwide, and in 2014 the National Institute for Health and Care Excellence released standards for interventions to prevent transition to psychosis.24 These included close monitoring, CBT and family interventions, and avoiding antipsychotics when possible.24
Focusing on sensitivity over specificity
The first generation of studies generated by the prevention model relied on outreach programs or referrals, which produced small samples of carefully selected, pre-screened individuals (Figure, Pre-screened) who were then screened again to establish the high-risk sample.25 While approximately 33% of these individuals became psychotic, the screening process required a very efficient means of eliminating those not at high-risk (given the ultimate target population represented only approximately .5% of young people) (Figure). The pre-screening and screening processes in these first-generation studies were labor-intensive but could only identify approximately 5% of those individuals destined to become psychotic over the next 2 or 3 years. Thus, alternative methods to enhance sensitivity were needed to extend programming to the general population.
Second-generation pre-screening (Figure; Step 1). New pre-screening methods were identified that captured more individuals destined to become psychotic. For example, approximately 90% of this population were registered in health care organizations (eg, health maintenance organizations) and received a psychiatric diagnosis in the year prior to the onset of psychosis (true positives).8 These samples, however, contained a much higher percentage of persons not destined to become psychotic, and somehow the issue of specificity (decreasing false positives) was minimized.8,9 For example, pre-screened samples contained 20 to 50 individuals not destined to become psychotic for each one who did.26 Since screening measures could only eliminate approximately 20% of this group (Figure, Step 2, page 25), second-generation transition rates fell from 30% to 40% to 2% to 10%.27,28
Other pre-screening approaches were introduced, but they also focused on capturing more of those destined to become psychotic (sensitivity) than eliminating those who would not (specificity). For instance, Australia opened more than 100 “Headspace” community centers nationwide designed to promote engagement and self-esteem in youth experiencing anxiety; depression; stress; relationship, work, or school problems; or bullying.13 Most services were free and included mental health staff who screened for psychosis and provided a wide range of services in a destigmatized setting. These methods identified at least an additional 5% to 7% of individuals destined to become psychotic, but to our knowledge, no data have been published on whether they helped eliminate those who did not.
Continue to: Second-generation screening
Second-generation screening (Figure, Step 2). A second screening aims to retain those pre-screened individuals who will become psychotic (ie, minimizing false negatives) while further minimizing those who do not (ie, minimizing false positives). The addition of cognitive, neural (eg, structural MRI; neurophysiologic), and biochemical (eg, inflammatory immune and stress) markers to the risk calculators have produced a sensitivity close to 100%.8,9 Unfortunately, these studies downplayed specificity, which remained approximately 20%.8,9 Specificity is critical not just because of concerns about stigma (ie, labeling people as pre-psychotic when they are not) but also because of the adverse effects of antipsychotic medications and the effects on future program development (interventions are costly and labor-intensive). Also, diluting the pool with individuals not at risk makes it nearly impossible to identify effective interventions (ie, power).27,28
While some studies focused on increasing specificity (to approximately 75%), this leads to an unacceptable loss of sensitivity (from 90% to 60%),29 with 40% of pre-screened individuals who would become psychotic being eliminated from the study population. The addition of other biological markers (eg, salivary cortisol)30 and use of learning health systems may be able to enhance these numbers (initial reports of specificity = 87% and sensitivity = 85%).8,9 This is accomplished by integrating artificial and human intelligence measures of clinical (symptom and neurocognitive measures) and biological (eg, polygenetic risk scores; gray matter volume) variables.31 However, even if these results are replicated, more effective pre-screening measures will be required.
Identifying a suitable sample population for prevention program studies is clearly more complicated than for FEP studies, where one can usually identify many of those in the at-risk population by their first hospitalization for psychotic symptoms. The issues of false positives (eg, substance-induced psychosis) and negatives (eg, slow deterioration, prominent negative symptoms) are important concerns, but proportionately far less significant.
Prevention and FEP interventions
Once a study sample is constituted, 1 to 3 years of treatment interventions are initiated. Interventions for prevention programs typically include CBT directed at attenuated psychosis (eg, reframing or de-catastrophizing unusual thoughts and minimizing distress associated with unusual perceptions); case management to facilitate personal, educational, and vocational goals; and family therapy in single or multi-group formats to educate one’s support system about the risk state and to minimize adverse familial responses.14 Many programs also include supported education or employment services to promote reintegration in age-appropriate activities; group therapy focused on substance abuse and social skills training; cognitive remediation to ameliorate the cognitive dysfunction; and an array of pharmacologic interventions designed to delay or prevent transition to psychosis or to alleviate symptoms. While most interventions are similar, FEP programs have recently included peer support staff. This appears to instill hope in newly diagnosed patients, provide role models, and provide peer supporters an opportunity to use their experiences to help others and earn income.32
The breadth and depth of these services are critical because retention in the program is highly dependent on participant engagement, which in turn is highly dependent on whether the program can help individuals get what they want (eg, friends, employment, education, more autonomy, physical health). The setting and atmosphere of the treatment program and the willingness/ability of staff to meet participants in the community are also important elements.11,12 In this context, the Headspace community centers are having an impact far beyond Australia and may prove to be a particularly good model.13
Continue to: Assessing prevention and FEP interventions
Assessing prevention and FEP interventions
The second generation of studies of prevention programs has not confirmed, let alone extended, the earlier findings and meta-analyses. A 2020 report concluded CBT was still the most promising intervention; it was more effective than control treatments at 12 and 18 months, although not at 6, 24, or 48 months.33 This review included controlled, open-label, and naturalistic studies that assessed family therapy; omega-3 polyunsaturated fatty acids; integrated psychological therapy (a package of interventions that included family education, CBT, social skills training, and cognitive remediation); N-methyl-
While these disappointing findings are at least partly attributable to the methodological challenges described above and in the Figure, other factors may hinder establishing effective interventions. In contrast to FEP studies, those focused on prevention had a very ambitious agenda (eliminating psychosis) and tended to downplay more modest intermediate outcomes. These studies also tended to assess new ideas with small samples rather than pursue promising findings with larger multi-site studies focused on a group of interventions. The authors of a Cochrane review observed “There is the impression that in this whole area there is a triumph of hope over adversity. There is the repeated hope invested in another—often unique—study question and then a study of fewer than 100 participants are completed. This results in the set of comparisons reported here, all 9 of which are too underpowered to really highlight clear differences.”34 To use a baseball analogy, it seems that investigators are “swinging for the fence” when a few singles are what’s really needed.
From the outset, the goals of FEP studies were more modest, largely ignoring the task of developing consensus definitions of recovery that require following patients for up to 5 to 10 years. Instead, they use intermediate endpoints based on adapting treatments that already appeared effective in patients with chronic mental disorders.35 As a consequence, researchers examining FEP demonstrated clear, albeit limited, salutary effects using large multi-site trials and previously established outcome measures.3,10,36 For instance, the Recovery After an Initial Schizophrenia Episode-Early Treatment Program (RAISE-ETP) study was a 2-year, multi-site RCT (N = 404) funded by the National Institute of Mental Health (NIMH). The investigators reported improved indices of social function (eg, quality of life; education and work participation) and total ratings of psychopathology and depression compared with treatment as usual. Furthermore, they established that DUP predicted treatment response.35 The latter finding was underscored by improvement being limited to the 50% with <74 weeks DUP. Annual costs of the program per 1 standard deviation improvement in quality of life were approximately $1,000 for patients with <74 weeks DUP and $40,000 for those with >74 weeks DUP. Concurrent meta-analyses confirmed and extended these findings,16 showing higher remission rates; diminished relapses and hospital admissions; greater engagement in programming; greater involvement in work and school; improved quality of life; and other steps toward recovery. These studies were also able to establish a clear benefit of antipsychotic medications, particularly a high acceptance of long-acting injectable antipsychotic formulations, which promoted adherence and decreased some adverse events37; and early use of clozapine therapy, which improved remission rates and longer-term outcomes.38 Other findings underscored the need to anticipate and address new problems associated with effective antipsychotic therapy (eg, antipsychotic response correlates with weight gain, a particularly intolerable adverse event for this age group).39 Providing pre-emptive strategies such as exercise groups and nutritional education may be necessary to maintain adherence.
Limitations of FEP studies
The effect sizes in these FEP studies were small to medium on outcome measures tracking recovery and associated indicators (eg, global functioning, school/work participation, treatment engagement); the number needed to treat for each of these was >10. There is no clear evidence that recovery programs such as RAISE-ETP actually reduce longer-term disability. Most studies showed disability payments increased while clinical benefits tended to fade over time. In addition, by grouping interventions together, the studies made it difficult to identify effective vs ineffective treatments, let alone determine how best to personalize therapy for participants in future studies.
The next generation of FEP studies
While limited in scope, the results of the recent FEP studies justify a next generation of recovery interventions designed to address these shortcomings and optimize program outcomes.39 Most previous FEP studies were conducted in community mental health center settings, thus eliminating the need to transition services developed in academia into the “real world.” The next generation of NIMH studies will be primarily conducted in analogous settings under the Early Psychosis Intervention Network (EPINET).40 EPINET’s study design echoes that responsible for the stepwise successes in the late 20th century that produced cures for the deadliest childhood cancer, acute lymphoblastic leukemia (ALL). This disease was successfully treated by modifying diverse evidence-based practices without relying on pharmacologic or other major treatment breakthroughs. Despite this, the effort yielded successful personalized interventions that were not obtainable for other severe childhood conditions.40 EPINET hopes to automate much of these stepwise advances with a learning health system. This program relies on data routinely collected in clinical practice to drive the process of scientific discovery. Specifically, it determines the relationships between clinical features, biologic measures, treatment characteristics, and symptomatic and functional outcomes. EPINET aims to accelerate our understanding of biomarkers of psychosis risk and onset, as well as factors associated with recovery and cure. Dashboard displays of outcomes will allow for real-time comparisons within and across early intervention clinics. This in turn identifies performance gaps and drives continuous quality improvement.
Continue to: Barriers to optimizing program efficacy for both models
Barriers to optimizing program efficacy for both models
Unfortunately, there are stark differences between ALL and severe mental disorders that potentially jeopardize the achievement of these aims, despite the advances in data analytic abilities that drive the learning health system. Specifically, the heterogeneity of psychotic illnesses and the absence of reliable prognostic and modifiable risk markers (responsible for failed efforts to enhance treatment of serious mental illness over the last half century1,2,41) are unlikely to be resolved by a learning health system. These measures are vital to determine whether specific interventions are effective, particularly given the absence of a randomized control group in the EPINET/learning health system design. Fortunately, however, the National Institutes for Health has recently initiated the Accelerating Medicines Partnership–Schizophrenia (AMP-SCZ). This approach seeks “promising biological markers that can help identify those at risk of developing schizophrenia as early as possible, track the progression of symptoms and other outcomes and ultimately define targets for treatment development.”42 The Box1,4,9,10,36,41,43-45 describes some of the challenges involved in identifying biomarkers of severe mental illness.
Box
Biomarkers and modifiable risk factors4,9,10,41,43 are at the core of personalized medicine and its ultimate objective (ie, theragnostics). This is the ability to identify the correct intervention for a disorder based on a biomarker of the illness.10,36 The inability to identify biomarkers of severe mental illness is multifactorial but in part may be attributable to “looking in all the wrong places.”41 By focusing on neural processes that generate psychiatric symptomatology, investigators are assuming they can bridge the “mind gap”1 and specifically distinguish between pathological, compensatory, or collateral measures of poorly characterized limbic neural functions.41
It may be more productive to identify a pathological process within the limbic system that produces a medical condition as well as the mental disorder. If one can isolate the pathologic limbic circuit activity responsible for a medical condition, one may be able to reproduce this in animal models and determine whether analogous processes contribute to the core features of the mental illness. Characterization of the aberrant neural circuit in animal models also could yield targets for future therapies. For example, episodic water intoxication in a discrete subset of patients with schizophrenia44 appears to arise from a stress diathesis produced by anterior hippocampal pathology that disrupts regulation of antidiuretic hormone, oxytocin, and hypothalamic-pituitary-adrenal axis secretion. These patients also exhibit psychogenic polydipsia that may be a consequence of the same hippocampal pathology that disrupts ventral striatal and lateral hypothalamic circuits. These circuits, in turn, also modulate motivated behaviors and cognitive processes likely relevant to psychosis.45
A strength-based approach
The absence of sufficiently powered RCTs for prevention studies and the reliance on intermediate outcomes for FEP studies leaves unanswered whether such programs can effectively prevent chronic psychosis at a cost society is willing to pay. Still, substantial evidence indicates that outreach, long-acting injectable antipsychotics, early consideration of clozapine, family therapy, CBT for psychosis/attenuated psychosis, and services focused on competitive employment can preserve social and occupational functioning.16,34 Until these broader questions are more definitively addressed, it seems reasonable to apply what we have learned (Table11,12,35,37-39,46).

Simply avoiding the most divisive aspects of the medical model that inadvertently promote stigma and undercut self-confidence may help maintain patients’ willingness to learn how best to apply their strengths and manage their limitations.11 The progression to enduring psychotic features (eg, fixed delusions) may reflect ongoing social isolation and alienation. A strength-based approach seeks first to establish common goals (eg, school, work, friends, family support, housing, leaving home) and then works to empower the patient to successfully reach those goals.35 This typically involves giving them the opportunity to fail, avoiding criticism when they do, and focusing on these experiences as learning opportunities from which success can ultimately result.
It is difficult to offer all these services in a typical private practice setting. Instead, it may make more sense to use one of the hundreds of early intervention services programs in the United States.46 If a psychiatric clinician is dedicated to working with this population, it may also be possible to establish ongoing relationships with primary care physicians, family and CBT therapists, family support services (eg, National Alliance on Mental Illness), caseworkers and employment counselors. In essence, a psychiatrist may be able re-create a multidisciplinary effort by taking advantage of the expertise of these various professionals. The challenge is to create a consistent message for patients and families in the absence of regular meetings with the clinical team, although the recent reliance on and improved sophistication of virtual meetings may help. Psychiatrists often play a critical role even when the patient is not prescribed medication, partly because they are most comfortable handling the risks and may have the most comprehensive understanding of the issues at play. When medications are appropriate and patients with FEP are willing to take them, early consideration of long-acting injectable antipsychotics and clozapine may provide better stabilization and diminish the risk of earlier and more frequent relapses.
Bottom Line
Early interventions for psychosis include the prevention model and the first-episode recovery model. It is difficult to assess, compare, and optimize the effectiveness of such programs. Current evidence supports a ‘strength-based’ approach focused on finding common ground between patients, their support system, and the treatment team.
Related Resources
- Early Assessment and Support Alliance. National Early Psychosis Directory. https://easacommunity.org/nationaldirectory.php
- Kane JM, Robinson DG, Schooler NR, et al. Comprehensive versus usual community care for first-episode psychosis: 2-year outcomes from the NIMH RAISE Early Treatment Program. Am J Psychiatry. 2016 ;173(4):362-372
Drug Brand Name
Clozapine • Clozaril
Neuroscience research over the past half century has failed to significantly advance the treatment of severe mental illness.1,2 Hence, evidence that a longer duration of untreated psychosis (DUP) aggravates—and early intervention with medication and social supports ameliorates—the long-term adverse consequences of psychotic disorders generated a great deal of interest.3,4 This knowledge led to the development of diverse early intervention services worldwide aimed at this putative “critical window.” It raised the possibility that appropriate interventions could prevent the long-term disability that makes chronic psychosis one of the most debilitating disorders.5,6 However, even beyond the varied cultural and economic confounds, it is difficult to assess, compare, and optimize program effectiveness.7 Obstacles include paucity of sufficiently powered, well-designed randomized controlled trials (RCTs), the absence of diagnostic biomarkers or other prognostic indicators to better account for the inherent heterogeneity in the population and associated outcomes, and the absence of modifiable risk factors that can guide interventions and provide intermediate outcomes.4,8-10
To better appreciate these issues, it is important to distinguish whether a program is designed to prevent psychosis, or to mitigate the effects of psychosis. Two models include the:
- Prevention model, which focuses on young individuals who are not yet overtly psychotic but at high risk
- First-episode recovery model, which focuses on those who have experienced a first episode of psychosis (FEP) but have not yet developed a chronic disorder.
Both models share long-term goals and are hampered by many of the same issues summarized above. They both deviate markedly from the standard medical model by including psychosocial services designed to promote restoration of a self-defined trajectory to greater independence.11-14 The 2 differ, however, in the challenges they must overcome to produce their sample populations and establish effective interventions.10,15,16
In this article, we provide a succinct overview of these issues and a set of recommendations based on a “strength-based” approach. This approach focuses on finding common ground between patients, their support system, and the treatment team in the service of empowering patients to resume responsibility for transition to adulthood.
The prevention model
While most prevention initiatives in medicine rely on the growing ability to target specific pathophysiologic pathways,3 preventing psychosis relies on clinical evidence showing that DUP and early interventions predict a better course of severe mental illness.17 In contrast, initiatives such as normalizing neonatal neuronal pathways are more consistent with the strategy utilized in other fields but have yet to yield a pathophysiologic target for psychosis.3,18
Initial efforts to identify ‘at-risk’ individuals
The prevention model of psychosis is based on the ability to identify young individuals at high risk for developing a psychotic disorder (Figure). The first screening measures were focused on prodromal psychosis (eg, significant loss of function, family history, and “intermittent” and “attenuated” psychotic symptoms). When applied to referred (ie, pre-screened) samples, 30% to 40% of this group who met criteria transitioned to psychosis over the next 1 to 3 years despite antidepressant and psychosocial interventions.19 Comprising 8 academic medical centers, the North American Prodrome Longitudinal Study (NAPLS) produced similar results using the Structured Interview for Prodromal Syndromes (SIPS).17 Thus, 30% to 50% of pre-screened individuals referred by school counselors and mental health professionals met SIPS criteria, and 35% of these individuals transitioned to psychosis over 30 months. The validity of this measure was further supported by the fact that higher baseline levels of unusual thought content, suspicion/paranoia, social impairment, and substance abuse successfully distinguished approximately 80% of those who transitioned to psychosis. The results of this first generation of screening studies were exciting because they seemed to demonstrate that highly concentrated samples of young persons at high risk of developing psychosis could be identified, and that fine-tuning the screening criteria could produce even more enriched samples (ie, positive predictive power).

Initial interventions produced promising results
The development of effective screening measures led to reports of effective treatment interventions. These were largely applied in a clinical staging model that restricted antipsychotic medications to those who failed to improve after receiving potentially “less toxic” interventions (eg, omega-3 polyunsaturated fatty acids and other antioxidants; psychotherapy; cognitive-behavioral therapy [CBT]; family therapy).5 While study designs were typically quasi-experimental, the interventions appeared to dramatically diminish the transition to psychosis (ie, approximately 50%).
Continue to: The first generation...
The first generation of RCTs appeared to confirm these results, although sample sizes were small, and most study designs assessed only a single intervention. Initial meta-analyses of these data reported that both CBT and antipsychotics appeared to prevent approximately one-half of individuals from becoming psychotic at 12 months, and more than one-third at 2 to 4 years, compared with treatment as usual.20
While some researchers challenged the validity of these findings,21-23 the results generated tremendous international enthusiasm and calls for widespread implementation.6 The number of early intervention services (EIS) centers increased dramatically worldwide, and in 2014 the National Institute for Health and Care Excellence released standards for interventions to prevent transition to psychosis.24 These included close monitoring, CBT and family interventions, and avoiding antipsychotics when possible.24
Focusing on sensitivity over specificity
The first generation of studies generated by the prevention model relied on outreach programs or referrals, which produced small samples of carefully selected, pre-screened individuals (Figure, Pre-screened) who were then screened again to establish the high-risk sample.25 While approximately 33% of these individuals became psychotic, the screening process required a very efficient means of eliminating those not at high-risk (given the ultimate target population represented only approximately .5% of young people) (Figure). The pre-screening and screening processes in these first-generation studies were labor-intensive but could only identify approximately 5% of those individuals destined to become psychotic over the next 2 or 3 years. Thus, alternative methods to enhance sensitivity were needed to extend programming to the general population.
Second-generation pre-screening (Figure; Step 1). New pre-screening methods were identified that captured more individuals destined to become psychotic. For example, approximately 90% of this population were registered in health care organizations (eg, health maintenance organizations) and received a psychiatric diagnosis in the year prior to the onset of psychosis (true positives).8 These samples, however, contained a much higher percentage of persons not destined to become psychotic, and somehow the issue of specificity (decreasing false positives) was minimized.8,9 For example, pre-screened samples contained 20 to 50 individuals not destined to become psychotic for each one who did.26 Since screening measures could only eliminate approximately 20% of this group (Figure, Step 2, page 25), second-generation transition rates fell from 30% to 40% to 2% to 10%.27,28
Other pre-screening approaches were introduced, but they also focused on capturing more of those destined to become psychotic (sensitivity) than eliminating those who would not (specificity). For instance, Australia opened more than 100 “Headspace” community centers nationwide designed to promote engagement and self-esteem in youth experiencing anxiety; depression; stress; relationship, work, or school problems; or bullying.13 Most services were free and included mental health staff who screened for psychosis and provided a wide range of services in a destigmatized setting. These methods identified at least an additional 5% to 7% of individuals destined to become psychotic, but to our knowledge, no data have been published on whether they helped eliminate those who did not.
Continue to: Second-generation screening
Second-generation screening (Figure, Step 2). A second screening aims to retain those pre-screened individuals who will become psychotic (ie, minimizing false negatives) while further minimizing those who do not (ie, minimizing false positives). The addition of cognitive, neural (eg, structural MRI; neurophysiologic), and biochemical (eg, inflammatory immune and stress) markers to the risk calculators have produced a sensitivity close to 100%.8,9 Unfortunately, these studies downplayed specificity, which remained approximately 20%.8,9 Specificity is critical not just because of concerns about stigma (ie, labeling people as pre-psychotic when they are not) but also because of the adverse effects of antipsychotic medications and the effects on future program development (interventions are costly and labor-intensive). Also, diluting the pool with individuals not at risk makes it nearly impossible to identify effective interventions (ie, power).27,28
While some studies focused on increasing specificity (to approximately 75%), this leads to an unacceptable loss of sensitivity (from 90% to 60%),29 with 40% of pre-screened individuals who would become psychotic being eliminated from the study population. The addition of other biological markers (eg, salivary cortisol)30 and use of learning health systems may be able to enhance these numbers (initial reports of specificity = 87% and sensitivity = 85%).8,9 This is accomplished by integrating artificial and human intelligence measures of clinical (symptom and neurocognitive measures) and biological (eg, polygenetic risk scores; gray matter volume) variables.31 However, even if these results are replicated, more effective pre-screening measures will be required.
Identifying a suitable sample population for prevention program studies is clearly more complicated than for FEP studies, where one can usually identify many of those in the at-risk population by their first hospitalization for psychotic symptoms. The issues of false positives (eg, substance-induced psychosis) and negatives (eg, slow deterioration, prominent negative symptoms) are important concerns, but proportionately far less significant.
Prevention and FEP interventions
Once a study sample is constituted, 1 to 3 years of treatment interventions are initiated. Interventions for prevention programs typically include CBT directed at attenuated psychosis (eg, reframing or de-catastrophizing unusual thoughts and minimizing distress associated with unusual perceptions); case management to facilitate personal, educational, and vocational goals; and family therapy in single or multi-group formats to educate one’s support system about the risk state and to minimize adverse familial responses.14 Many programs also include supported education or employment services to promote reintegration in age-appropriate activities; group therapy focused on substance abuse and social skills training; cognitive remediation to ameliorate the cognitive dysfunction; and an array of pharmacologic interventions designed to delay or prevent transition to psychosis or to alleviate symptoms. While most interventions are similar, FEP programs have recently included peer support staff. This appears to instill hope in newly diagnosed patients, provide role models, and provide peer supporters an opportunity to use their experiences to help others and earn income.32
The breadth and depth of these services are critical because retention in the program is highly dependent on participant engagement, which in turn is highly dependent on whether the program can help individuals get what they want (eg, friends, employment, education, more autonomy, physical health). The setting and atmosphere of the treatment program and the willingness/ability of staff to meet participants in the community are also important elements.11,12 In this context, the Headspace community centers are having an impact far beyond Australia and may prove to be a particularly good model.13
Continue to: Assessing prevention and FEP interventions
Assessing prevention and FEP interventions
The second generation of studies of prevention programs has not confirmed, let alone extended, the earlier findings and meta-analyses. A 2020 report concluded CBT was still the most promising intervention; it was more effective than control treatments at 12 and 18 months, although not at 6, 24, or 48 months.33 This review included controlled, open-label, and naturalistic studies that assessed family therapy; omega-3 polyunsaturated fatty acids; integrated psychological therapy (a package of interventions that included family education, CBT, social skills training, and cognitive remediation); N-methyl-
While these disappointing findings are at least partly attributable to the methodological challenges described above and in the Figure, other factors may hinder establishing effective interventions. In contrast to FEP studies, those focused on prevention had a very ambitious agenda (eliminating psychosis) and tended to downplay more modest intermediate outcomes. These studies also tended to assess new ideas with small samples rather than pursue promising findings with larger multi-site studies focused on a group of interventions. The authors of a Cochrane review observed “There is the impression that in this whole area there is a triumph of hope over adversity. There is the repeated hope invested in another—often unique—study question and then a study of fewer than 100 participants are completed. This results in the set of comparisons reported here, all 9 of which are too underpowered to really highlight clear differences.”34 To use a baseball analogy, it seems that investigators are “swinging for the fence” when a few singles are what’s really needed.
From the outset, the goals of FEP studies were more modest, largely ignoring the task of developing consensus definitions of recovery that require following patients for up to 5 to 10 years. Instead, they use intermediate endpoints based on adapting treatments that already appeared effective in patients with chronic mental disorders.35 As a consequence, researchers examining FEP demonstrated clear, albeit limited, salutary effects using large multi-site trials and previously established outcome measures.3,10,36 For instance, the Recovery After an Initial Schizophrenia Episode-Early Treatment Program (RAISE-ETP) study was a 2-year, multi-site RCT (N = 404) funded by the National Institute of Mental Health (NIMH). The investigators reported improved indices of social function (eg, quality of life; education and work participation) and total ratings of psychopathology and depression compared with treatment as usual. Furthermore, they established that DUP predicted treatment response.35 The latter finding was underscored by improvement being limited to the 50% with <74 weeks DUP. Annual costs of the program per 1 standard deviation improvement in quality of life were approximately $1,000 for patients with <74 weeks DUP and $40,000 for those with >74 weeks DUP. Concurrent meta-analyses confirmed and extended these findings,16 showing higher remission rates; diminished relapses and hospital admissions; greater engagement in programming; greater involvement in work and school; improved quality of life; and other steps toward recovery. These studies were also able to establish a clear benefit of antipsychotic medications, particularly a high acceptance of long-acting injectable antipsychotic formulations, which promoted adherence and decreased some adverse events37; and early use of clozapine therapy, which improved remission rates and longer-term outcomes.38 Other findings underscored the need to anticipate and address new problems associated with effective antipsychotic therapy (eg, antipsychotic response correlates with weight gain, a particularly intolerable adverse event for this age group).39 Providing pre-emptive strategies such as exercise groups and nutritional education may be necessary to maintain adherence.
Limitations of FEP studies
The effect sizes in these FEP studies were small to medium on outcome measures tracking recovery and associated indicators (eg, global functioning, school/work participation, treatment engagement); the number needed to treat for each of these was >10. There is no clear evidence that recovery programs such as RAISE-ETP actually reduce longer-term disability. Most studies showed disability payments increased while clinical benefits tended to fade over time. In addition, by grouping interventions together, the studies made it difficult to identify effective vs ineffective treatments, let alone determine how best to personalize therapy for participants in future studies.
The next generation of FEP studies
While limited in scope, the results of the recent FEP studies justify a next generation of recovery interventions designed to address these shortcomings and optimize program outcomes.39 Most previous FEP studies were conducted in community mental health center settings, thus eliminating the need to transition services developed in academia into the “real world.” The next generation of NIMH studies will be primarily conducted in analogous settings under the Early Psychosis Intervention Network (EPINET).40 EPINET’s study design echoes that responsible for the stepwise successes in the late 20th century that produced cures for the deadliest childhood cancer, acute lymphoblastic leukemia (ALL). This disease was successfully treated by modifying diverse evidence-based practices without relying on pharmacologic or other major treatment breakthroughs. Despite this, the effort yielded successful personalized interventions that were not obtainable for other severe childhood conditions.40 EPINET hopes to automate much of these stepwise advances with a learning health system. This program relies on data routinely collected in clinical practice to drive the process of scientific discovery. Specifically, it determines the relationships between clinical features, biologic measures, treatment characteristics, and symptomatic and functional outcomes. EPINET aims to accelerate our understanding of biomarkers of psychosis risk and onset, as well as factors associated with recovery and cure. Dashboard displays of outcomes will allow for real-time comparisons within and across early intervention clinics. This in turn identifies performance gaps and drives continuous quality improvement.
Continue to: Barriers to optimizing program efficacy for both models
Barriers to optimizing program efficacy for both models
Unfortunately, there are stark differences between ALL and severe mental disorders that potentially jeopardize the achievement of these aims, despite the advances in data analytic abilities that drive the learning health system. Specifically, the heterogeneity of psychotic illnesses and the absence of reliable prognostic and modifiable risk markers (responsible for failed efforts to enhance treatment of serious mental illness over the last half century1,2,41) are unlikely to be resolved by a learning health system. These measures are vital to determine whether specific interventions are effective, particularly given the absence of a randomized control group in the EPINET/learning health system design. Fortunately, however, the National Institutes for Health has recently initiated the Accelerating Medicines Partnership–Schizophrenia (AMP-SCZ). This approach seeks “promising biological markers that can help identify those at risk of developing schizophrenia as early as possible, track the progression of symptoms and other outcomes and ultimately define targets for treatment development.”42 The Box1,4,9,10,36,41,43-45 describes some of the challenges involved in identifying biomarkers of severe mental illness.
Box
Biomarkers and modifiable risk factors4,9,10,41,43 are at the core of personalized medicine and its ultimate objective (ie, theragnostics). This is the ability to identify the correct intervention for a disorder based on a biomarker of the illness.10,36 The inability to identify biomarkers of severe mental illness is multifactorial but in part may be attributable to “looking in all the wrong places.”41 By focusing on neural processes that generate psychiatric symptomatology, investigators are assuming they can bridge the “mind gap”1 and specifically distinguish between pathological, compensatory, or collateral measures of poorly characterized limbic neural functions.41
It may be more productive to identify a pathological process within the limbic system that produces a medical condition as well as the mental disorder. If one can isolate the pathologic limbic circuit activity responsible for a medical condition, one may be able to reproduce this in animal models and determine whether analogous processes contribute to the core features of the mental illness. Characterization of the aberrant neural circuit in animal models also could yield targets for future therapies. For example, episodic water intoxication in a discrete subset of patients with schizophrenia44 appears to arise from a stress diathesis produced by anterior hippocampal pathology that disrupts regulation of antidiuretic hormone, oxytocin, and hypothalamic-pituitary-adrenal axis secretion. These patients also exhibit psychogenic polydipsia that may be a consequence of the same hippocampal pathology that disrupts ventral striatal and lateral hypothalamic circuits. These circuits, in turn, also modulate motivated behaviors and cognitive processes likely relevant to psychosis.45
A strength-based approach
The absence of sufficiently powered RCTs for prevention studies and the reliance on intermediate outcomes for FEP studies leaves unanswered whether such programs can effectively prevent chronic psychosis at a cost society is willing to pay. Still, substantial evidence indicates that outreach, long-acting injectable antipsychotics, early consideration of clozapine, family therapy, CBT for psychosis/attenuated psychosis, and services focused on competitive employment can preserve social and occupational functioning.16,34 Until these broader questions are more definitively addressed, it seems reasonable to apply what we have learned (Table11,12,35,37-39,46).

Simply avoiding the most divisive aspects of the medical model that inadvertently promote stigma and undercut self-confidence may help maintain patients’ willingness to learn how best to apply their strengths and manage their limitations.11 The progression to enduring psychotic features (eg, fixed delusions) may reflect ongoing social isolation and alienation. A strength-based approach seeks first to establish common goals (eg, school, work, friends, family support, housing, leaving home) and then works to empower the patient to successfully reach those goals.35 This typically involves giving them the opportunity to fail, avoiding criticism when they do, and focusing on these experiences as learning opportunities from which success can ultimately result.
It is difficult to offer all these services in a typical private practice setting. Instead, it may make more sense to use one of the hundreds of early intervention services programs in the United States.46 If a psychiatric clinician is dedicated to working with this population, it may also be possible to establish ongoing relationships with primary care physicians, family and CBT therapists, family support services (eg, National Alliance on Mental Illness), caseworkers and employment counselors. In essence, a psychiatrist may be able re-create a multidisciplinary effort by taking advantage of the expertise of these various professionals. The challenge is to create a consistent message for patients and families in the absence of regular meetings with the clinical team, although the recent reliance on and improved sophistication of virtual meetings may help. Psychiatrists often play a critical role even when the patient is not prescribed medication, partly because they are most comfortable handling the risks and may have the most comprehensive understanding of the issues at play. When medications are appropriate and patients with FEP are willing to take them, early consideration of long-acting injectable antipsychotics and clozapine may provide better stabilization and diminish the risk of earlier and more frequent relapses.
Bottom Line
Early interventions for psychosis include the prevention model and the first-episode recovery model. It is difficult to assess, compare, and optimize the effectiveness of such programs. Current evidence supports a ‘strength-based’ approach focused on finding common ground between patients, their support system, and the treatment team.
Related Resources
- Early Assessment and Support Alliance. National Early Psychosis Directory. https://easacommunity.org/nationaldirectory.php
- Kane JM, Robinson DG, Schooler NR, et al. Comprehensive versus usual community care for first-episode psychosis: 2-year outcomes from the NIMH RAISE Early Treatment Program. Am J Psychiatry. 2016 ;173(4):362-372
Drug Brand Name
Clozapine • Clozaril
1. Hyman SE. Revolution stalled. Sci Transl Med. 2012;4(155):155cm11. doi: 10.1126/scitranslmed.3003142
2. Harrington A. Mind fixers: psychiatry’s troubled search for the biology of mental illness. W.W. Norton & Company; 2019.
3. Millan MJ, Andrieux A, Bartzokis G, et al. Altering the course of schizophrenia: progress and perspectives. Nat Rev Drug Discov. 2016;15(7):485-515.
4. Lieberman JA, Small SA, Girgis RR. Early detection and preventive intervention in schizophrenia: from fantasy to reality. Am J Psychiatry. 2019;176(10):794-810.
5. McGorry PD, Nelson B, Nordentoft M, et al. Intervention in individuals at ultra-high risk for psychosis: a review and future directions. J Clin Psychiatry. 2009;70(9):1206-1212.
6. Csillag C, Nordentoft M, Mizuno M, et al. Early intervention in psychosis: From clinical intervention to health system implementation. Early Interv Psychiatry. 2018;12(4):757-764.
7. McGorry PD, Ratheesh A, O’Donoghue B. Early intervention—an implementation challenge for 21st century mental health care. JAMA Psychiatry. 2018;75(6):545-546.
8. Rosenheck R. Toward dissemination of secondary prevention for psychosis. Am J Psychiatry. 2018;175(5):393-394.
9. Fusar-Poli P, Salazar de Pablo G, Correll CU, et al. Prevention of psychosis: advances in detection, prognosis, and intervention. JAMA Psychiatry. 2020;77(7):755-765.
10. Oliver D, Reilly TJ, Baccaredda Boy O, et al. What causes the onset of psychosis in individuals at clinical high risk? A meta-analysis of risk and protective factors. Schizophr Bull. 2020;46(1):110-120.
11. Tindall R, Simmons M, Allott K, et al. Disengagement processes within an early intervention service for first-episode psychosis: a longitudinal, qualitative, multi-perspective study. Front Psychiatry. 2020;11:565-565.
12. Dixon LB, Holoshitz Y, Nossel I. Treatment engagement of individuals experiencing mental illness: review and update. World Psychiatry. 2016;15(1):13-20.
13. Rickwood D, Paraskakis M, Quin D, et al. Australia’s innovation in youth mental health care: The headspace centre model. Early Interv Psychiatry. 2019;13(1):159-166.
14. Woodberry KA, Shapiro DI, Bryant C, et al. Progress and future directions in research on the psychosis prodrome: a review for clinicians. Harv Rev Psychiatry. 2016;24(2):87-103.
15. Gupta T, Mittal VA. Advances in clinical staging, early intervention, and the prevention of psychosis. F1000Res. 2019;8:F1000 Faculty Rev-2027. doi: 10.12688/f1000research.20346.1
16. Correll CU, Galling B, Pawar A, et al. Comparison of early intervention services vs treatment as usual for early-phase psychosis: a systematic review, meta-analysis, and meta-regression. JAMA Psychiatry. 2018;75(6):555-565.
17. Cannon TD, Cadenhead K, Cornblatt B, et al. Prediction of psychosis in youth at high clinical risk: a multisite longitudinal study in North America. Arch Gen Psychiatry. 2008;65(1):28-37.
18. Sommer IE, Bearden CE, van Dellen E, et al. Early interventions in risk groups for schizophrenia: what are we waiting for? NPJ Schizophr. 2016;2(1):16003-16003.
19. McGorry PD, Nelson B. Clinical high risk for psychosis—not seeing the trees for the wood. JAMA Psychiatry. 2020;77(7):559-560.
20. van der Gaag M, Smit F, Bechdolf A, et al. Preventing a first episode of psychosis: meta-analysis of randomized controlled prevention trials of 12 month and longer-term follow-ups. Schizophr Res. 2013;149(1):56-62.
21. Marshall M, Rathbone J. Early intervention for psychosis. Cochrane Database Syst Rev. 2011;(6):CD004718. doi: 10.1002/14651858.CD004718.pub3
22. Heinssen RK, Insel TR. Preventing the onset of psychosis: not quite there yet. Schizophr Bull. 2015;41(1):28-29.
23. Amos AJ. Evidence that treatment prevents transition to psychosis in ultra-high-risk patients remains questionable. Schizophr Res. 2014;153(1):240.
24. National Institute for Health and Care Excellence. Psychosis and schizophrenia in adults: prevention and management. Clinical guideline [CG178]. 1.3.7 How to deliver psychological interventions. Published February 12, 2014. Updated March 1, 2014. Accessed August 30, 2021. https://www.nice.org.uk/guidance/cg178/chapter/recommendations#how-to-deliver-psychological-interventions
25. Fusar-Poli P, Werbeloff N, Rutigliano G, et al. Transdiagnostic risk calculator for the automatic detection of individuals at risk and the prediction of psychosis: second replication in an independent National Health Service Trust. Schizophr Bull. 2019;45(3):562-570.
26. Fusar-Poli P, Oliver D, Spada G, et al. The case for improved transdiagnostic detection of first-episode psychosis: electronic health record cohort study. Schizophr Res. 2021;228:547-554.
27. Fusar-Poli P. Negative psychosis prevention trials. JAMA Psychiatry. 2017;74(6):651.
28. Cuijpers P, Smit F, Furukawa TA. Most at‐risk individuals will not develop a mental disorder: the limited predictive strength of risk factors. World Psychiatry. 2021;20(2):224-225.
29. Carrión RE, Cornblatt BA, Burton CZ, et al. Personalized prediction of psychosis: external validation of the NAPLS-2 psychosis risk calculator with the EDIPPP Project. Am J Psychiatry. 2016;173(10):989-996.
30. Worthington MA, Walker EF, Addington J, et al. Incorporating cortisol into the NAPLS2 individualized risk calculator for prediction of psychosis. Schizophr Res. 2021;227:95-100.
31. Koutsouleris N, Dwyer DB, Degenhardt F, et al. Multimodal machine learning workflows for prediction of psychosis in patients with clinical high-risk syndromes and recent-onset depression. JAMA Psychiatry. 2021;78(2):195-209.
32. Simmons MB, Grace D, Fava NJ, et al. The experiences of youth mental health peer workers over time: a qualitative study with longitudinal analysis. Community Ment Health J. 2020;56(5):906-914.
33. Devoe DJ, Farris MS, Townes P, et al. Interventions and transition in youth at risk of psychosis: a systematic review and meta-analyses. J Clin Psychiatry. 2020;81(3):17r12053. doi: 10.4088/JCP.17r12053
34. Bosnjak Kuharic D, Kekin I, Hew J, et al. Interventions for prodromal stage of psychosis. Cochrane Database Syst Rev. 2019;2019(11):CD012236
35. Dixon LB, Goldman HH, Srihari VH, et al. Transforming the treatment of schizophrenia in the United States: The RAISE Initiative. Annu Rev Clin Psychol. 2018;14:237-258.
36. Friedman-Yakoobian MS, Parrish EM, Eack SM, et al. Neurocognitive and social cognitive training for youth at clinical high risk (CHR) for psychosis: a randomized controlled feasibility trial. Schizophr Res. 2020;S0920-9964(20)30461-8. doi: 10.1016/j.schres.2020.09.005
37. Kane JM, Schooler NR, Marcy P, et al. Effect of long-acting injectable antipsychotics vs usual care on time to first hospitalization in early-phase schizophrenia: a randomized clinical trial. JAMA Psychiatry. 2020;77(12):1217-1224.
38. Morrison AP, Pyle M, Maughan D, et al. Antipsychotic medication versus psychological intervention versus a combination of both in adolescents with first-episode psychosis (MAPS): a multicentre, three-arm, randomised controlled pilot and feasibility study. Lancet Psychiatry. 2020;7(9):788-800.
39. Chen YQ, Li XR, Zhang L, et al. Therapeutic response is associated with antipsychotic-induced weight gain in drug-naive first-episode patients with schizophrenia: an 8-week prospective study. J Clin Psychiatry. 2021;82(3):20m13469. doi: 10.4088/JCP.20m13469
40. Insel TR. RAISE-ing our expectations for first-episode psychosis. Am J Psychiatry. 2016;173(4):311-312.
41. Tandon R, Goldman M. Overview of neurobiology. In: Janicak PG, Marder SR, Tandon R, et al, eds. Schizophrenia: recent advances in diagnosis and treatment. Springer; 2014:27-33.
42. National Institutes of Health. Accelerating Medicines Partnership. Schizophrenia. Accessed August 30, 2021. https://www.nih.gov/research-training/accelerating-medicines-partnership-amp/schizophrenia
43. Guloksuz S, van Os J. The slow death of the concept of schizophrenia and the painful birth of the psychosis spectrum. Psychol Med. 2018;48(2):229-244.
44. Christ-Crain M, Bichet DG, Fenske WK, et al. Diabetes insipidus. Nat Rev Dis Primers. 2019;5(1):54.
45. Ahmadi L, Goldman MB. Primary polydipsia: update. Best Pract Res Clin Endocrinol Metab. 2020;34(5):101469. doi: 10.1016/j.beem.2020.101469
46. Early Assessment and Support Alliance. National Early Psychosis Directory. Accessed August 30, 2021. https://easacommunity.org/national-directory.php
1. Hyman SE. Revolution stalled. Sci Transl Med. 2012;4(155):155cm11. doi: 10.1126/scitranslmed.3003142
2. Harrington A. Mind fixers: psychiatry’s troubled search for the biology of mental illness. W.W. Norton & Company; 2019.
3. Millan MJ, Andrieux A, Bartzokis G, et al. Altering the course of schizophrenia: progress and perspectives. Nat Rev Drug Discov. 2016;15(7):485-515.
4. Lieberman JA, Small SA, Girgis RR. Early detection and preventive intervention in schizophrenia: from fantasy to reality. Am J Psychiatry. 2019;176(10):794-810.
5. McGorry PD, Nelson B, Nordentoft M, et al. Intervention in individuals at ultra-high risk for psychosis: a review and future directions. J Clin Psychiatry. 2009;70(9):1206-1212.
6. Csillag C, Nordentoft M, Mizuno M, et al. Early intervention in psychosis: From clinical intervention to health system implementation. Early Interv Psychiatry. 2018;12(4):757-764.
7. McGorry PD, Ratheesh A, O’Donoghue B. Early intervention—an implementation challenge for 21st century mental health care. JAMA Psychiatry. 2018;75(6):545-546.
8. Rosenheck R. Toward dissemination of secondary prevention for psychosis. Am J Psychiatry. 2018;175(5):393-394.
9. Fusar-Poli P, Salazar de Pablo G, Correll CU, et al. Prevention of psychosis: advances in detection, prognosis, and intervention. JAMA Psychiatry. 2020;77(7):755-765.
10. Oliver D, Reilly TJ, Baccaredda Boy O, et al. What causes the onset of psychosis in individuals at clinical high risk? A meta-analysis of risk and protective factors. Schizophr Bull. 2020;46(1):110-120.
11. Tindall R, Simmons M, Allott K, et al. Disengagement processes within an early intervention service for first-episode psychosis: a longitudinal, qualitative, multi-perspective study. Front Psychiatry. 2020;11:565-565.
12. Dixon LB, Holoshitz Y, Nossel I. Treatment engagement of individuals experiencing mental illness: review and update. World Psychiatry. 2016;15(1):13-20.
13. Rickwood D, Paraskakis M, Quin D, et al. Australia’s innovation in youth mental health care: The headspace centre model. Early Interv Psychiatry. 2019;13(1):159-166.
14. Woodberry KA, Shapiro DI, Bryant C, et al. Progress and future directions in research on the psychosis prodrome: a review for clinicians. Harv Rev Psychiatry. 2016;24(2):87-103.
15. Gupta T, Mittal VA. Advances in clinical staging, early intervention, and the prevention of psychosis. F1000Res. 2019;8:F1000 Faculty Rev-2027. doi: 10.12688/f1000research.20346.1
16. Correll CU, Galling B, Pawar A, et al. Comparison of early intervention services vs treatment as usual for early-phase psychosis: a systematic review, meta-analysis, and meta-regression. JAMA Psychiatry. 2018;75(6):555-565.
17. Cannon TD, Cadenhead K, Cornblatt B, et al. Prediction of psychosis in youth at high clinical risk: a multisite longitudinal study in North America. Arch Gen Psychiatry. 2008;65(1):28-37.
18. Sommer IE, Bearden CE, van Dellen E, et al. Early interventions in risk groups for schizophrenia: what are we waiting for? NPJ Schizophr. 2016;2(1):16003-16003.
19. McGorry PD, Nelson B. Clinical high risk for psychosis—not seeing the trees for the wood. JAMA Psychiatry. 2020;77(7):559-560.
20. van der Gaag M, Smit F, Bechdolf A, et al. Preventing a first episode of psychosis: meta-analysis of randomized controlled prevention trials of 12 month and longer-term follow-ups. Schizophr Res. 2013;149(1):56-62.
21. Marshall M, Rathbone J. Early intervention for psychosis. Cochrane Database Syst Rev. 2011;(6):CD004718. doi: 10.1002/14651858.CD004718.pub3
22. Heinssen RK, Insel TR. Preventing the onset of psychosis: not quite there yet. Schizophr Bull. 2015;41(1):28-29.
23. Amos AJ. Evidence that treatment prevents transition to psychosis in ultra-high-risk patients remains questionable. Schizophr Res. 2014;153(1):240.
24. National Institute for Health and Care Excellence. Psychosis and schizophrenia in adults: prevention and management. Clinical guideline [CG178]. 1.3.7 How to deliver psychological interventions. Published February 12, 2014. Updated March 1, 2014. Accessed August 30, 2021. https://www.nice.org.uk/guidance/cg178/chapter/recommendations#how-to-deliver-psychological-interventions
25. Fusar-Poli P, Werbeloff N, Rutigliano G, et al. Transdiagnostic risk calculator for the automatic detection of individuals at risk and the prediction of psychosis: second replication in an independent National Health Service Trust. Schizophr Bull. 2019;45(3):562-570.
26. Fusar-Poli P, Oliver D, Spada G, et al. The case for improved transdiagnostic detection of first-episode psychosis: electronic health record cohort study. Schizophr Res. 2021;228:547-554.
27. Fusar-Poli P. Negative psychosis prevention trials. JAMA Psychiatry. 2017;74(6):651.
28. Cuijpers P, Smit F, Furukawa TA. Most at‐risk individuals will not develop a mental disorder: the limited predictive strength of risk factors. World Psychiatry. 2021;20(2):224-225.
29. Carrión RE, Cornblatt BA, Burton CZ, et al. Personalized prediction of psychosis: external validation of the NAPLS-2 psychosis risk calculator with the EDIPPP Project. Am J Psychiatry. 2016;173(10):989-996.
30. Worthington MA, Walker EF, Addington J, et al. Incorporating cortisol into the NAPLS2 individualized risk calculator for prediction of psychosis. Schizophr Res. 2021;227:95-100.
31. Koutsouleris N, Dwyer DB, Degenhardt F, et al. Multimodal machine learning workflows for prediction of psychosis in patients with clinical high-risk syndromes and recent-onset depression. JAMA Psychiatry. 2021;78(2):195-209.
32. Simmons MB, Grace D, Fava NJ, et al. The experiences of youth mental health peer workers over time: a qualitative study with longitudinal analysis. Community Ment Health J. 2020;56(5):906-914.
33. Devoe DJ, Farris MS, Townes P, et al. Interventions and transition in youth at risk of psychosis: a systematic review and meta-analyses. J Clin Psychiatry. 2020;81(3):17r12053. doi: 10.4088/JCP.17r12053
34. Bosnjak Kuharic D, Kekin I, Hew J, et al. Interventions for prodromal stage of psychosis. Cochrane Database Syst Rev. 2019;2019(11):CD012236
35. Dixon LB, Goldman HH, Srihari VH, et al. Transforming the treatment of schizophrenia in the United States: The RAISE Initiative. Annu Rev Clin Psychol. 2018;14:237-258.
36. Friedman-Yakoobian MS, Parrish EM, Eack SM, et al. Neurocognitive and social cognitive training for youth at clinical high risk (CHR) for psychosis: a randomized controlled feasibility trial. Schizophr Res. 2020;S0920-9964(20)30461-8. doi: 10.1016/j.schres.2020.09.005
37. Kane JM, Schooler NR, Marcy P, et al. Effect of long-acting injectable antipsychotics vs usual care on time to first hospitalization in early-phase schizophrenia: a randomized clinical trial. JAMA Psychiatry. 2020;77(12):1217-1224.
38. Morrison AP, Pyle M, Maughan D, et al. Antipsychotic medication versus psychological intervention versus a combination of both in adolescents with first-episode psychosis (MAPS): a multicentre, three-arm, randomised controlled pilot and feasibility study. Lancet Psychiatry. 2020;7(9):788-800.
39. Chen YQ, Li XR, Zhang L, et al. Therapeutic response is associated with antipsychotic-induced weight gain in drug-naive first-episode patients with schizophrenia: an 8-week prospective study. J Clin Psychiatry. 2021;82(3):20m13469. doi: 10.4088/JCP.20m13469
40. Insel TR. RAISE-ing our expectations for first-episode psychosis. Am J Psychiatry. 2016;173(4):311-312.
41. Tandon R, Goldman M. Overview of neurobiology. In: Janicak PG, Marder SR, Tandon R, et al, eds. Schizophrenia: recent advances in diagnosis and treatment. Springer; 2014:27-33.
42. National Institutes of Health. Accelerating Medicines Partnership. Schizophrenia. Accessed August 30, 2021. https://www.nih.gov/research-training/accelerating-medicines-partnership-amp/schizophrenia
43. Guloksuz S, van Os J. The slow death of the concept of schizophrenia and the painful birth of the psychosis spectrum. Psychol Med. 2018;48(2):229-244.
44. Christ-Crain M, Bichet DG, Fenske WK, et al. Diabetes insipidus. Nat Rev Dis Primers. 2019;5(1):54.
45. Ahmadi L, Goldman MB. Primary polydipsia: update. Best Pract Res Clin Endocrinol Metab. 2020;34(5):101469. doi: 10.1016/j.beem.2020.101469
46. Early Assessment and Support Alliance. National Early Psychosis Directory. Accessed August 30, 2021. https://easacommunity.org/national-directory.php
Persistent altered mental status
CASE Sluggish, weak, and incoherent
Mr. O, age 24, who has a history of schizophrenia and obesity, presents to the emergency department (ED) for altered mental status (AMS). His mother reports that he has been sluggish, weak, incoherent, had no appetite, and that on the day before admission, he was drinking excessive amounts of water and urinating every 10 minutes.
HISTORY Multiple ineffective antipsychotics
Mr. O was diagnosed with schizophrenia at age 21 and struggled with medication adherence, which resulted in multiple hospitalizations for stabilization. Trials of haloperidol, risperidone, paliperidone palmitate, and valproic acid had been ineffective. At the time of admission, his psychotropic medication regimen is fluphenazine decanoate, 25 mg injection every 2 weeks; clozapine, 50 mg/d; lithium carbonate, 300 mg twice a day; benztropine, 2 mg every night; and trazodone, 50 mg every night.
EVALUATION Fever, tachycardia, and diabetic ketoacidosis
Upon arrival to the ED, Mr. O is obtunded, unable to follow commands, and does not respond to painful stimuli. On physical exam, he has a fever of 38.4°C (reference range 35.1°C to 37.9°C); tachycardia with a heart rate of 142 beats per minute (bpm) (reference range 60 to 100); tachypnea with a respiratory rate of 35 breaths per minute (reference range 12 to 20); a blood pressure of 116/76 mmHg (reference range 90/60 to 130/80); and hypoxemia with an oxygen saturation of 90% on room air (reference range 94% to 100%).
Mr. O is admitted to the hospital and his laboratory workup indicates diabetic ketoacidosis (DKA), with a glucose of 1,700 mg/dL; anion gap of 30 (reference range 4 to 12 mmol/L); pH 7.04 (reference range 7.32 to 7.42); serum bicarbonate 6 (reference range 20 to 24 mEq/L); beta-hydroxybutyrate 11.04 (reference range 0 to 0.27 mmol/L); urine ketones, serum osmolality 407 (reference range 280 to 300 mOsm/kg); and an elevated white blood cell count of 18.4 (reference range 4.5 to 11.0 × 109/L). A CT scan of the head is negative for acute pathology.
Initially, all psychotropic medications are held. On Day 3 of hospitalization, psychiatry is consulted and clozapine, 50 mg/d; lithium, 300 mg/d; and benztropine, 1 mg at night, are restarted; however, fluphenazine decanoate and trazodone are held. The team recommends IV haloperidol, 2 mg as needed for agitation; however, it is never administered.
Imaging rules out deep vein thrombosis, cardiac dysfunction, and stroke, but a CT chest scan is notable for bilateral lung infiltrates, which suggests aspiration pneumonia.
Mr. O is diagnosed with diabetes, complicated by DKA, and is treated in the intensive care unit (ICU). Despite resolution of the DKA, he remains altered with fever and tachycardia.
Continue to: On Day 6 of hospitalization...
On Day 6 of hospitalization, Mr. O continues to be tachycardic and obtunded with nuchal rigidity. The team decides to transfer Mr. O to another hospital for a higher level of care and continued workup of his persistent AMS.
Immediately upon arrival at the second hospital, infectious disease and neurology teams are consulted for further evaluation. Mr. O’s AMS continues despite no clear signs of infection or other neurologic insults.
[polldaddy:10930631]
The authors’ observations
Based on Mr. O’s psychiatric history and laboratory results, the first medical team concluded his initial AMS was likely secondary to DKA; however, the AMS continued after the DKA resolved. At the second hospital, Mr. O’s treatment team continued to dig for answers.
EVALUATION Exploring the differential diagnosis
At the second hospital, Mr. O is admitted to the ICU with fever (37.8°C), tachycardia (120 bpm), tachypnea, withdrawal from painful stimuli, decreased reflexes, and muscle rigidity, including clenched jaw. The differential diagnoses include meningitis, sepsis from aspiration pneumonia, severe metabolic encephalopathy with prolonged recovery, central pontine myelinolysis, anoxic brain injury, and subclinical seizures.
Empiric vancomycin, 1.75 g every 12 hours; ceftriaxone, 2 g/d; and acyclovir, 900 mg every 8 hours are started for meningoencephalitis, and all psychotropic medications are discontinued. Case reports have documented a relationship between hyperglycemic hyperosmolar syndrome (HHS) and malignant hyperthermia in rare cases1; however, HHS is ruled out based on Mr. O’s laboratory results.A lumbar puncture and imaging rules out CNS infection. Antibiotic treatment is narrowed to ampicillin-sulbactam due to Mr. O’s prior CT chest showing concern for aspiration pneumonia. An MRI of the brain rules out central pontine myelinolysis, acute stroke, and anoxic brain injury, and an EEG shows nonspecific encephalopathy. On Day 10 of hospitalization, a neurologic exam shows flaccid paralysis and bilateral clonus, and Mr. O is mute. On Day 14 of hospitalization, his fever resolves, and his blood cultures are negative. On Day 15 of hospitalization, Mr. O’s creatine kinase (CK) level is elevated at 1,308 U/L (reference range 26 to 192 U/L), suggesting rhabdomyolysis.
Continue to: Given the neurologic exam findings...
Given the neurologic exam findings, and the limited evidence of infection, the differential diagnosis for Mr. O’s AMS is broadened to include catatonia, neuroleptic malignant syndrome (NMS), serotonin syndrome, and autoimmune encephalitis. The psychiatry team evaluates Mr. O for catatonia. He scores 14 on the Bush-Francis Catatonia Rating Scale, with findings of immobility/stupor, mutism, staring, autonomic instability, and withdrawal indicating the presence of catatonia.2
The authors’ observations
When Mr. O was transferred to the second hospital, the primary concern was to rule out meningitis due to his unstable vitals, obtunded mental state, and nuchal rigidity. A comprehensive infectious workup, including lumbar puncture, was imperative because infection can not only lead to AMS, but also precipitate episodes of DKA. Mr. O’s persistently abnormal vital signs indicated an underlying process may have been missed by focusing on treating DKA.
TREATMENT Finally, the diagnosis is established
A lorazepam challenge is performed, and Mr. O receives 4 mg of lorazepam over 24 hours with little change in his catatonia symptoms. Given his persistent fever, tachycardia, and an elevated CK levels in the context of recent exposure to antipsychotic medications, Mr. O is diagnosed with NMS (Table 13,4 ) and is started on bromocriptine, 5 mg 3 times daily.
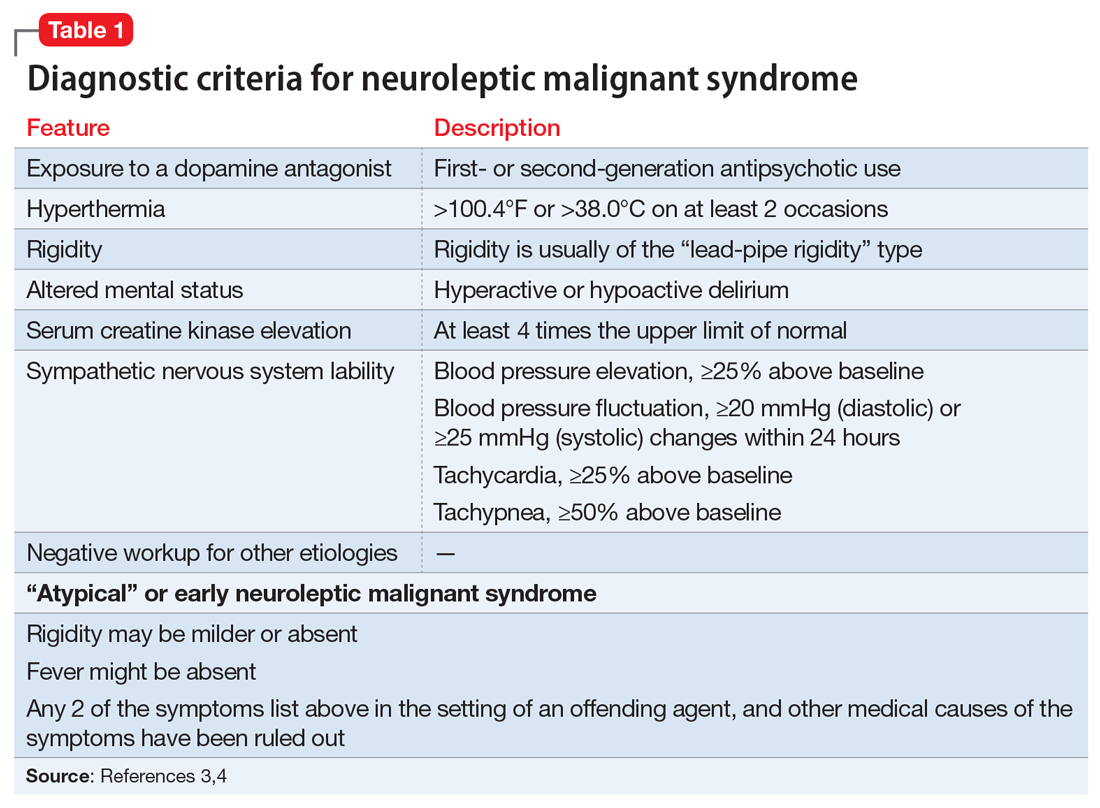
[polldaddy:10930632]
The authors’ observations
Mr. O’s complicated medical state—starting with DKA, halting the use of antipsychotic medications, and the suspicion of catatonia due to his history of schizophrenia—all distracted from the ultimate diagnosis of NMS as the cause of his enduring AMS and autonomic instability. Catatonia and NMS have overlapping symptomatology, including rigidity, autonomic instability, and stupor, which make the diagnosis of either condition complicated. A positive lorazepam test to diagnose catatonia is defined as a marked reduction in catatonia symptoms (typically a 50% reduction) as measured on a standardized rating scale.5 However, a negative lorazepam challenge does not definitely rule out catatonia because some cases are resistant to benzodiazepines.6
NMS risk factors relevant in this case include male sex, young age, acute medical illness, dehydration, and exposure to multiple psychotropic medications, including 2 antipsychotics, clozapine and fluphenazine.7 DKA is especially pertinent due to its acute onset and cause of significant dehydration. NMS can occur at any point of antipsychotic exposure, although the risk is highest during the initial weeks of treatment and during dosage changes. Unfortunately, Mr. O’s treatment team was unable to determine whether his medication had been recently changed, so it is not known what role this may have played in the development of NMS. Although first-generation antipsychotics are considered more likely to cause NMS, second-generation antipsychotics (SGAs) dominate the treatment of schizophrenia and bipolar disorder, and these medications also can cause NMS.8 As occurred in this case, long-acting injectable antipsychotics can be easily forgotten when not administered in the hospital, and their presence in the body persists for weeks. For example, the half-life of fluphenazine decanoate is approximately 10 days, and the half-life of haloperidol decanoate is 21 days.9
Continue to: OUTCOME Improvement with bromocriptine
OUTCOME Improvement with bromocriptine
After 4 days of bromocriptine, 5 mg 3 times daily, Mr. O is more alert, able to say “hello,” and can follow 1-step commands. By Day 26 of hospitalization, his CK levels decrease to 296 U/L, his CSF autoimmune panel is negative, and he is able to participate in physical therapy. After failing multiple swallow tests, Mr. O requires a percutaneous endoscopic gastrostomy (PEG) tube. He is discharged from the hospital to a long-term acute care facility with the plan to taper bromocriptine and restart a psychotropic regimen with his outpatient psychiatrist. At the time of discharge, he is able to sit at the edge of the bed independently, state his name, and respond to questions with multiple-word answers.
[polldaddy:10930633]
The authors’ observations
The most common pharmacologic treatments for NMS are dantrolene, bromocriptine, benzodiazepines (lorazepam or diazepam), and amantadine.3 Mild cases of NMS should be treated with discontinuation of all antipsychotics, supportive care, and benzodiazepines.3 Bromocriptine or amantadine are more appropriate for moderate cases and dantrolene for severe cases of NMS.3 All antipsychotics should be discontinued while a patient is experiencing an episode of NMS; however, once the NMS has resolved, clinicians must thoroughly evaluate the risks and benefits of restarting antipsychotic medication. After a patient has experienced an episode of NMS, clinicians generally should avoid prescribing the agent(s) that caused NMS and long-acting injections, and slowly titrate a low-potency SGA such as quetiapine.10Table 23,11,12 outlines the pharmacologic treatment of NMS.
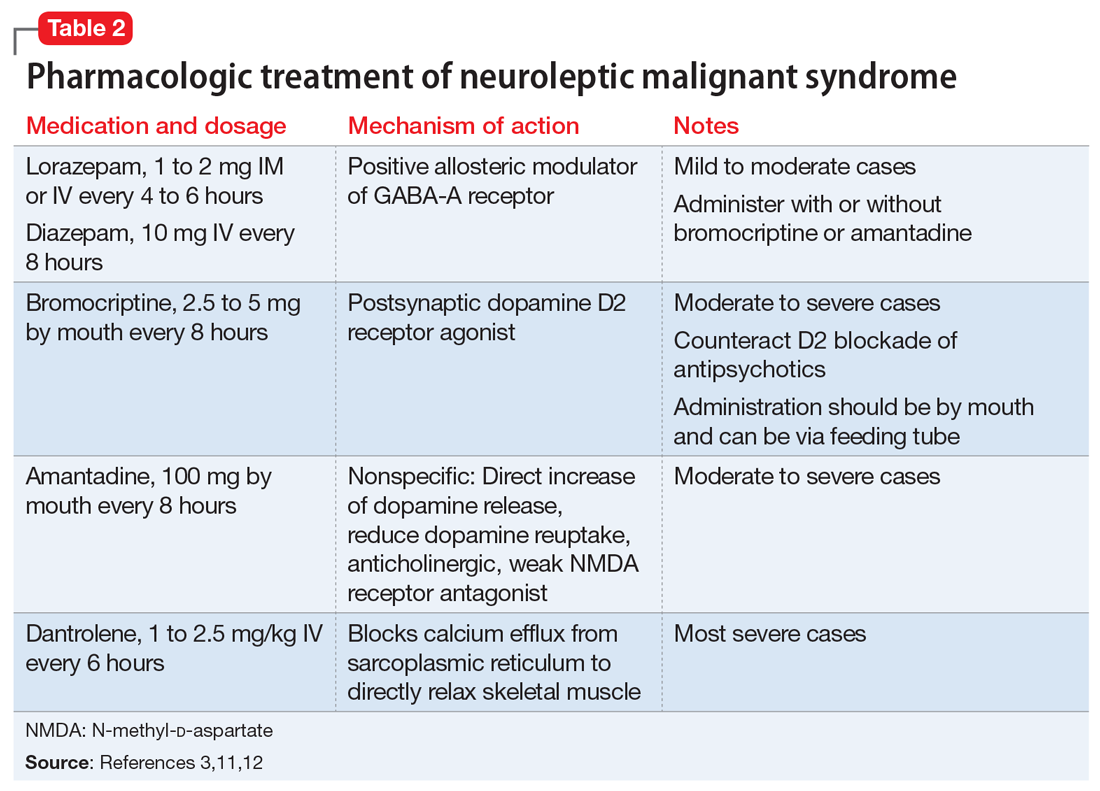
Bottom Line
Neuroleptic malignant syndrome (NMS) should always be part of the differential diagnosis in patients with mental illness and altered mental status. The risk of NMS is especially high in patients with acute medical illness and exposure to antipsychotic medications.
Related Resource
- Turner AH, Kim JJ, McCarron RM. Differentiating serotonin syndrome and neuroleptic malignant syndrome. Current Psychiatry. 2019;18(2):30-36.
Drug Brand Names
Acyclovir • Zovirax
Amantadine • Gocovri
Ampicillin-sulbactam • Unasyn
Aripiprazole • Abilify Maintena
Benztropine • Cogentin
Bromocriptine • Cycloset, Parlodel
Ceftriaxone • Rocephin
Clozapine • Clozaril
Dantrolene • Dantrium
Diazepam • Valium
Haloperidol • Haldol
Lithium • Eskalith, Lithobid
Lorazepam • Ativan
Paliperidone palmitate • Invega Sustenna
Quetiapine • Seroquel
Risperidone • Risperdal
Valproate sodium • Depakote
Trazodone • Oleptro
Vancomycin • Vancocin
1. Zeitler P, Haqq A, Rosenbloom A, et al. Hyperglycemic hyperosmolar syndrome in children: pathophysiological considerations and suggested guidelines for treatment. J Pediatr. 2011;158(1):9-14.e1-2. doi: 10.1016/j.jpeds.2010.09.048
2. Francis A. Catatonia: diagnosis, classification, and treatment. Curr Psychiatry Rep. 2010;12(3):180-185. doi: 10.1007/s11920-010-0113-y
3. Pileggi DJ, Cook AM. Neuroleptic malignant syndrome. Ann Pharmacother. 2016;50(11):973-981. doi:10.1177/1060028016657553
4. Gurrera RJ, Caroff SN, Cohen A, et al. An international consensus study of neuroleptic malignant syndrome diagnostic criteria using the Delphi method. J Clin Psychiatry. 2011;72(9):1222-1228. doi:10.4088/JCP.10m06438
5. Sienaert P, Dhossche DM, Vancampfort D, et al. A clinical review of the treatment of catatonia. Front Psychiatry. 2014;5:181. doi:10.3389/fpsyt.2014.00181
6. Daniels J. Catatonia: clinical aspects and neurobiological correlates. J Neuropsychiatry Clin Neurosci. 2009;21(4):371-380. doi:10.1176/jnp.2009.21.4.371
7. Bhanushali MJ, Tuite PJ. The evaluation and management of patients with neuroleptic malignant syndrome. Neurol Clin. 2004;22(2):389-411. doi:10.1016/j.ncl.2003.12.006
8. Tse L, Barr AM, Scarapicchia V, et al. Neuroleptic malignant syndrome: a review from a clinically oriented perspective. Curr Neuropharmacol. 2015;13(3):395-406. doi:10.2174/1570159x13999150424113345
9. Correll CU, Kim E, Sliwa JK, et al. Pharmacokinetic characteristics of long-acting injectable antipsychotics for schizophrenia: an overview. CNS Drugs. 2021;35(1):39-59. doi:10.1007/s40263-020-00779-5
10. Strawn JR, Keck PE Jr, Caroff SN. Neuroleptic malignant syndrome. Am J Psychiatry. 2007;164(6):870-876. doi:10.1176/ajp.2007.164.6.870
11. Griffin CE 3rd, Kaye AM, Bueno FR, et al. Benzodiazepine pharmacology and central nervous system-mediated effects. Ochsner J. 2013;13(2):214-223.
12. Reulbach U, Dütsch C, Biermann T, et al. Managing an effective treatment for neuroleptic malignant syndrome. Crit Care. 2007;11(1):R4. doi:10.1186/cc5148
CASE Sluggish, weak, and incoherent
Mr. O, age 24, who has a history of schizophrenia and obesity, presents to the emergency department (ED) for altered mental status (AMS). His mother reports that he has been sluggish, weak, incoherent, had no appetite, and that on the day before admission, he was drinking excessive amounts of water and urinating every 10 minutes.
HISTORY Multiple ineffective antipsychotics
Mr. O was diagnosed with schizophrenia at age 21 and struggled with medication adherence, which resulted in multiple hospitalizations for stabilization. Trials of haloperidol, risperidone, paliperidone palmitate, and valproic acid had been ineffective. At the time of admission, his psychotropic medication regimen is fluphenazine decanoate, 25 mg injection every 2 weeks; clozapine, 50 mg/d; lithium carbonate, 300 mg twice a day; benztropine, 2 mg every night; and trazodone, 50 mg every night.
EVALUATION Fever, tachycardia, and diabetic ketoacidosis
Upon arrival to the ED, Mr. O is obtunded, unable to follow commands, and does not respond to painful stimuli. On physical exam, he has a fever of 38.4°C (reference range 35.1°C to 37.9°C); tachycardia with a heart rate of 142 beats per minute (bpm) (reference range 60 to 100); tachypnea with a respiratory rate of 35 breaths per minute (reference range 12 to 20); a blood pressure of 116/76 mmHg (reference range 90/60 to 130/80); and hypoxemia with an oxygen saturation of 90% on room air (reference range 94% to 100%).
Mr. O is admitted to the hospital and his laboratory workup indicates diabetic ketoacidosis (DKA), with a glucose of 1,700 mg/dL; anion gap of 30 (reference range 4 to 12 mmol/L); pH 7.04 (reference range 7.32 to 7.42); serum bicarbonate 6 (reference range 20 to 24 mEq/L); beta-hydroxybutyrate 11.04 (reference range 0 to 0.27 mmol/L); urine ketones, serum osmolality 407 (reference range 280 to 300 mOsm/kg); and an elevated white blood cell count of 18.4 (reference range 4.5 to 11.0 × 109/L). A CT scan of the head is negative for acute pathology.
Initially, all psychotropic medications are held. On Day 3 of hospitalization, psychiatry is consulted and clozapine, 50 mg/d; lithium, 300 mg/d; and benztropine, 1 mg at night, are restarted; however, fluphenazine decanoate and trazodone are held. The team recommends IV haloperidol, 2 mg as needed for agitation; however, it is never administered.
Imaging rules out deep vein thrombosis, cardiac dysfunction, and stroke, but a CT chest scan is notable for bilateral lung infiltrates, which suggests aspiration pneumonia.
Mr. O is diagnosed with diabetes, complicated by DKA, and is treated in the intensive care unit (ICU). Despite resolution of the DKA, he remains altered with fever and tachycardia.
Continue to: On Day 6 of hospitalization...
On Day 6 of hospitalization, Mr. O continues to be tachycardic and obtunded with nuchal rigidity. The team decides to transfer Mr. O to another hospital for a higher level of care and continued workup of his persistent AMS.
Immediately upon arrival at the second hospital, infectious disease and neurology teams are consulted for further evaluation. Mr. O’s AMS continues despite no clear signs of infection or other neurologic insults.
[polldaddy:10930631]
The authors’ observations
Based on Mr. O’s psychiatric history and laboratory results, the first medical team concluded his initial AMS was likely secondary to DKA; however, the AMS continued after the DKA resolved. At the second hospital, Mr. O’s treatment team continued to dig for answers.
EVALUATION Exploring the differential diagnosis
At the second hospital, Mr. O is admitted to the ICU with fever (37.8°C), tachycardia (120 bpm), tachypnea, withdrawal from painful stimuli, decreased reflexes, and muscle rigidity, including clenched jaw. The differential diagnoses include meningitis, sepsis from aspiration pneumonia, severe metabolic encephalopathy with prolonged recovery, central pontine myelinolysis, anoxic brain injury, and subclinical seizures.
Empiric vancomycin, 1.75 g every 12 hours; ceftriaxone, 2 g/d; and acyclovir, 900 mg every 8 hours are started for meningoencephalitis, and all psychotropic medications are discontinued. Case reports have documented a relationship between hyperglycemic hyperosmolar syndrome (HHS) and malignant hyperthermia in rare cases1; however, HHS is ruled out based on Mr. O’s laboratory results.A lumbar puncture and imaging rules out CNS infection. Antibiotic treatment is narrowed to ampicillin-sulbactam due to Mr. O’s prior CT chest showing concern for aspiration pneumonia. An MRI of the brain rules out central pontine myelinolysis, acute stroke, and anoxic brain injury, and an EEG shows nonspecific encephalopathy. On Day 10 of hospitalization, a neurologic exam shows flaccid paralysis and bilateral clonus, and Mr. O is mute. On Day 14 of hospitalization, his fever resolves, and his blood cultures are negative. On Day 15 of hospitalization, Mr. O’s creatine kinase (CK) level is elevated at 1,308 U/L (reference range 26 to 192 U/L), suggesting rhabdomyolysis.
Continue to: Given the neurologic exam findings...
Given the neurologic exam findings, and the limited evidence of infection, the differential diagnosis for Mr. O’s AMS is broadened to include catatonia, neuroleptic malignant syndrome (NMS), serotonin syndrome, and autoimmune encephalitis. The psychiatry team evaluates Mr. O for catatonia. He scores 14 on the Bush-Francis Catatonia Rating Scale, with findings of immobility/stupor, mutism, staring, autonomic instability, and withdrawal indicating the presence of catatonia.2
The authors’ observations
When Mr. O was transferred to the second hospital, the primary concern was to rule out meningitis due to his unstable vitals, obtunded mental state, and nuchal rigidity. A comprehensive infectious workup, including lumbar puncture, was imperative because infection can not only lead to AMS, but also precipitate episodes of DKA. Mr. O’s persistently abnormal vital signs indicated an underlying process may have been missed by focusing on treating DKA.
TREATMENT Finally, the diagnosis is established
A lorazepam challenge is performed, and Mr. O receives 4 mg of lorazepam over 24 hours with little change in his catatonia symptoms. Given his persistent fever, tachycardia, and an elevated CK levels in the context of recent exposure to antipsychotic medications, Mr. O is diagnosed with NMS (Table 13,4 ) and is started on bromocriptine, 5 mg 3 times daily.

[polldaddy:10930632]
The authors’ observations
Mr. O’s complicated medical state—starting with DKA, halting the use of antipsychotic medications, and the suspicion of catatonia due to his history of schizophrenia—all distracted from the ultimate diagnosis of NMS as the cause of his enduring AMS and autonomic instability. Catatonia and NMS have overlapping symptomatology, including rigidity, autonomic instability, and stupor, which make the diagnosis of either condition complicated. A positive lorazepam test to diagnose catatonia is defined as a marked reduction in catatonia symptoms (typically a 50% reduction) as measured on a standardized rating scale.5 However, a negative lorazepam challenge does not definitely rule out catatonia because some cases are resistant to benzodiazepines.6
NMS risk factors relevant in this case include male sex, young age, acute medical illness, dehydration, and exposure to multiple psychotropic medications, including 2 antipsychotics, clozapine and fluphenazine.7 DKA is especially pertinent due to its acute onset and cause of significant dehydration. NMS can occur at any point of antipsychotic exposure, although the risk is highest during the initial weeks of treatment and during dosage changes. Unfortunately, Mr. O’s treatment team was unable to determine whether his medication had been recently changed, so it is not known what role this may have played in the development of NMS. Although first-generation antipsychotics are considered more likely to cause NMS, second-generation antipsychotics (SGAs) dominate the treatment of schizophrenia and bipolar disorder, and these medications also can cause NMS.8 As occurred in this case, long-acting injectable antipsychotics can be easily forgotten when not administered in the hospital, and their presence in the body persists for weeks. For example, the half-life of fluphenazine decanoate is approximately 10 days, and the half-life of haloperidol decanoate is 21 days.9
Continue to: OUTCOME Improvement with bromocriptine
OUTCOME Improvement with bromocriptine
After 4 days of bromocriptine, 5 mg 3 times daily, Mr. O is more alert, able to say “hello,” and can follow 1-step commands. By Day 26 of hospitalization, his CK levels decrease to 296 U/L, his CSF autoimmune panel is negative, and he is able to participate in physical therapy. After failing multiple swallow tests, Mr. O requires a percutaneous endoscopic gastrostomy (PEG) tube. He is discharged from the hospital to a long-term acute care facility with the plan to taper bromocriptine and restart a psychotropic regimen with his outpatient psychiatrist. At the time of discharge, he is able to sit at the edge of the bed independently, state his name, and respond to questions with multiple-word answers.
[polldaddy:10930633]
The authors’ observations
The most common pharmacologic treatments for NMS are dantrolene, bromocriptine, benzodiazepines (lorazepam or diazepam), and amantadine.3 Mild cases of NMS should be treated with discontinuation of all antipsychotics, supportive care, and benzodiazepines.3 Bromocriptine or amantadine are more appropriate for moderate cases and dantrolene for severe cases of NMS.3 All antipsychotics should be discontinued while a patient is experiencing an episode of NMS; however, once the NMS has resolved, clinicians must thoroughly evaluate the risks and benefits of restarting antipsychotic medication. After a patient has experienced an episode of NMS, clinicians generally should avoid prescribing the agent(s) that caused NMS and long-acting injections, and slowly titrate a low-potency SGA such as quetiapine.10Table 23,11,12 outlines the pharmacologic treatment of NMS.

Bottom Line
Neuroleptic malignant syndrome (NMS) should always be part of the differential diagnosis in patients with mental illness and altered mental status. The risk of NMS is especially high in patients with acute medical illness and exposure to antipsychotic medications.
Related Resource
- Turner AH, Kim JJ, McCarron RM. Differentiating serotonin syndrome and neuroleptic malignant syndrome. Current Psychiatry. 2019;18(2):30-36.
Drug Brand Names
Acyclovir • Zovirax
Amantadine • Gocovri
Ampicillin-sulbactam • Unasyn
Aripiprazole • Abilify Maintena
Benztropine • Cogentin
Bromocriptine • Cycloset, Parlodel
Ceftriaxone • Rocephin
Clozapine • Clozaril
Dantrolene • Dantrium
Diazepam • Valium
Haloperidol • Haldol
Lithium • Eskalith, Lithobid
Lorazepam • Ativan
Paliperidone palmitate • Invega Sustenna
Quetiapine • Seroquel
Risperidone • Risperdal
Valproate sodium • Depakote
Trazodone • Oleptro
Vancomycin • Vancocin
CASE Sluggish, weak, and incoherent
Mr. O, age 24, who has a history of schizophrenia and obesity, presents to the emergency department (ED) for altered mental status (AMS). His mother reports that he has been sluggish, weak, incoherent, had no appetite, and that on the day before admission, he was drinking excessive amounts of water and urinating every 10 minutes.
HISTORY Multiple ineffective antipsychotics
Mr. O was diagnosed with schizophrenia at age 21 and struggled with medication adherence, which resulted in multiple hospitalizations for stabilization. Trials of haloperidol, risperidone, paliperidone palmitate, and valproic acid had been ineffective. At the time of admission, his psychotropic medication regimen is fluphenazine decanoate, 25 mg injection every 2 weeks; clozapine, 50 mg/d; lithium carbonate, 300 mg twice a day; benztropine, 2 mg every night; and trazodone, 50 mg every night.
EVALUATION Fever, tachycardia, and diabetic ketoacidosis
Upon arrival to the ED, Mr. O is obtunded, unable to follow commands, and does not respond to painful stimuli. On physical exam, he has a fever of 38.4°C (reference range 35.1°C to 37.9°C); tachycardia with a heart rate of 142 beats per minute (bpm) (reference range 60 to 100); tachypnea with a respiratory rate of 35 breaths per minute (reference range 12 to 20); a blood pressure of 116/76 mmHg (reference range 90/60 to 130/80); and hypoxemia with an oxygen saturation of 90% on room air (reference range 94% to 100%).
Mr. O is admitted to the hospital and his laboratory workup indicates diabetic ketoacidosis (DKA), with a glucose of 1,700 mg/dL; anion gap of 30 (reference range 4 to 12 mmol/L); pH 7.04 (reference range 7.32 to 7.42); serum bicarbonate 6 (reference range 20 to 24 mEq/L); beta-hydroxybutyrate 11.04 (reference range 0 to 0.27 mmol/L); urine ketones, serum osmolality 407 (reference range 280 to 300 mOsm/kg); and an elevated white blood cell count of 18.4 (reference range 4.5 to 11.0 × 109/L). A CT scan of the head is negative for acute pathology.
Initially, all psychotropic medications are held. On Day 3 of hospitalization, psychiatry is consulted and clozapine, 50 mg/d; lithium, 300 mg/d; and benztropine, 1 mg at night, are restarted; however, fluphenazine decanoate and trazodone are held. The team recommends IV haloperidol, 2 mg as needed for agitation; however, it is never administered.
Imaging rules out deep vein thrombosis, cardiac dysfunction, and stroke, but a CT chest scan is notable for bilateral lung infiltrates, which suggests aspiration pneumonia.
Mr. O is diagnosed with diabetes, complicated by DKA, and is treated in the intensive care unit (ICU). Despite resolution of the DKA, he remains altered with fever and tachycardia.
Continue to: On Day 6 of hospitalization...
On Day 6 of hospitalization, Mr. O continues to be tachycardic and obtunded with nuchal rigidity. The team decides to transfer Mr. O to another hospital for a higher level of care and continued workup of his persistent AMS.
Immediately upon arrival at the second hospital, infectious disease and neurology teams are consulted for further evaluation. Mr. O’s AMS continues despite no clear signs of infection or other neurologic insults.
[polldaddy:10930631]
The authors’ observations
Based on Mr. O’s psychiatric history and laboratory results, the first medical team concluded his initial AMS was likely secondary to DKA; however, the AMS continued after the DKA resolved. At the second hospital, Mr. O’s treatment team continued to dig for answers.
EVALUATION Exploring the differential diagnosis
At the second hospital, Mr. O is admitted to the ICU with fever (37.8°C), tachycardia (120 bpm), tachypnea, withdrawal from painful stimuli, decreased reflexes, and muscle rigidity, including clenched jaw. The differential diagnoses include meningitis, sepsis from aspiration pneumonia, severe metabolic encephalopathy with prolonged recovery, central pontine myelinolysis, anoxic brain injury, and subclinical seizures.
Empiric vancomycin, 1.75 g every 12 hours; ceftriaxone, 2 g/d; and acyclovir, 900 mg every 8 hours are started for meningoencephalitis, and all psychotropic medications are discontinued. Case reports have documented a relationship between hyperglycemic hyperosmolar syndrome (HHS) and malignant hyperthermia in rare cases1; however, HHS is ruled out based on Mr. O’s laboratory results.A lumbar puncture and imaging rules out CNS infection. Antibiotic treatment is narrowed to ampicillin-sulbactam due to Mr. O’s prior CT chest showing concern for aspiration pneumonia. An MRI of the brain rules out central pontine myelinolysis, acute stroke, and anoxic brain injury, and an EEG shows nonspecific encephalopathy. On Day 10 of hospitalization, a neurologic exam shows flaccid paralysis and bilateral clonus, and Mr. O is mute. On Day 14 of hospitalization, his fever resolves, and his blood cultures are negative. On Day 15 of hospitalization, Mr. O’s creatine kinase (CK) level is elevated at 1,308 U/L (reference range 26 to 192 U/L), suggesting rhabdomyolysis.
Continue to: Given the neurologic exam findings...
Given the neurologic exam findings, and the limited evidence of infection, the differential diagnosis for Mr. O’s AMS is broadened to include catatonia, neuroleptic malignant syndrome (NMS), serotonin syndrome, and autoimmune encephalitis. The psychiatry team evaluates Mr. O for catatonia. He scores 14 on the Bush-Francis Catatonia Rating Scale, with findings of immobility/stupor, mutism, staring, autonomic instability, and withdrawal indicating the presence of catatonia.2
The authors’ observations
When Mr. O was transferred to the second hospital, the primary concern was to rule out meningitis due to his unstable vitals, obtunded mental state, and nuchal rigidity. A comprehensive infectious workup, including lumbar puncture, was imperative because infection can not only lead to AMS, but also precipitate episodes of DKA. Mr. O’s persistently abnormal vital signs indicated an underlying process may have been missed by focusing on treating DKA.
TREATMENT Finally, the diagnosis is established
A lorazepam challenge is performed, and Mr. O receives 4 mg of lorazepam over 24 hours with little change in his catatonia symptoms. Given his persistent fever, tachycardia, and an elevated CK levels in the context of recent exposure to antipsychotic medications, Mr. O is diagnosed with NMS (Table 13,4 ) and is started on bromocriptine, 5 mg 3 times daily.

[polldaddy:10930632]
The authors’ observations
Mr. O’s complicated medical state—starting with DKA, halting the use of antipsychotic medications, and the suspicion of catatonia due to his history of schizophrenia—all distracted from the ultimate diagnosis of NMS as the cause of his enduring AMS and autonomic instability. Catatonia and NMS have overlapping symptomatology, including rigidity, autonomic instability, and stupor, which make the diagnosis of either condition complicated. A positive lorazepam test to diagnose catatonia is defined as a marked reduction in catatonia symptoms (typically a 50% reduction) as measured on a standardized rating scale.5 However, a negative lorazepam challenge does not definitely rule out catatonia because some cases are resistant to benzodiazepines.6
NMS risk factors relevant in this case include male sex, young age, acute medical illness, dehydration, and exposure to multiple psychotropic medications, including 2 antipsychotics, clozapine and fluphenazine.7 DKA is especially pertinent due to its acute onset and cause of significant dehydration. NMS can occur at any point of antipsychotic exposure, although the risk is highest during the initial weeks of treatment and during dosage changes. Unfortunately, Mr. O’s treatment team was unable to determine whether his medication had been recently changed, so it is not known what role this may have played in the development of NMS. Although first-generation antipsychotics are considered more likely to cause NMS, second-generation antipsychotics (SGAs) dominate the treatment of schizophrenia and bipolar disorder, and these medications also can cause NMS.8 As occurred in this case, long-acting injectable antipsychotics can be easily forgotten when not administered in the hospital, and their presence in the body persists for weeks. For example, the half-life of fluphenazine decanoate is approximately 10 days, and the half-life of haloperidol decanoate is 21 days.9
Continue to: OUTCOME Improvement with bromocriptine
OUTCOME Improvement with bromocriptine
After 4 days of bromocriptine, 5 mg 3 times daily, Mr. O is more alert, able to say “hello,” and can follow 1-step commands. By Day 26 of hospitalization, his CK levels decrease to 296 U/L, his CSF autoimmune panel is negative, and he is able to participate in physical therapy. After failing multiple swallow tests, Mr. O requires a percutaneous endoscopic gastrostomy (PEG) tube. He is discharged from the hospital to a long-term acute care facility with the plan to taper bromocriptine and restart a psychotropic regimen with his outpatient psychiatrist. At the time of discharge, he is able to sit at the edge of the bed independently, state his name, and respond to questions with multiple-word answers.
[polldaddy:10930633]
The authors’ observations
The most common pharmacologic treatments for NMS are dantrolene, bromocriptine, benzodiazepines (lorazepam or diazepam), and amantadine.3 Mild cases of NMS should be treated with discontinuation of all antipsychotics, supportive care, and benzodiazepines.3 Bromocriptine or amantadine are more appropriate for moderate cases and dantrolene for severe cases of NMS.3 All antipsychotics should be discontinued while a patient is experiencing an episode of NMS; however, once the NMS has resolved, clinicians must thoroughly evaluate the risks and benefits of restarting antipsychotic medication. After a patient has experienced an episode of NMS, clinicians generally should avoid prescribing the agent(s) that caused NMS and long-acting injections, and slowly titrate a low-potency SGA such as quetiapine.10Table 23,11,12 outlines the pharmacologic treatment of NMS.

Bottom Line
Neuroleptic malignant syndrome (NMS) should always be part of the differential diagnosis in patients with mental illness and altered mental status. The risk of NMS is especially high in patients with acute medical illness and exposure to antipsychotic medications.
Related Resource
- Turner AH, Kim JJ, McCarron RM. Differentiating serotonin syndrome and neuroleptic malignant syndrome. Current Psychiatry. 2019;18(2):30-36.
Drug Brand Names
Acyclovir • Zovirax
Amantadine • Gocovri
Ampicillin-sulbactam • Unasyn
Aripiprazole • Abilify Maintena
Benztropine • Cogentin
Bromocriptine • Cycloset, Parlodel
Ceftriaxone • Rocephin
Clozapine • Clozaril
Dantrolene • Dantrium
Diazepam • Valium
Haloperidol • Haldol
Lithium • Eskalith, Lithobid
Lorazepam • Ativan
Paliperidone palmitate • Invega Sustenna
Quetiapine • Seroquel
Risperidone • Risperdal
Valproate sodium • Depakote
Trazodone • Oleptro
Vancomycin • Vancocin
1. Zeitler P, Haqq A, Rosenbloom A, et al. Hyperglycemic hyperosmolar syndrome in children: pathophysiological considerations and suggested guidelines for treatment. J Pediatr. 2011;158(1):9-14.e1-2. doi: 10.1016/j.jpeds.2010.09.048
2. Francis A. Catatonia: diagnosis, classification, and treatment. Curr Psychiatry Rep. 2010;12(3):180-185. doi: 10.1007/s11920-010-0113-y
3. Pileggi DJ, Cook AM. Neuroleptic malignant syndrome. Ann Pharmacother. 2016;50(11):973-981. doi:10.1177/1060028016657553
4. Gurrera RJ, Caroff SN, Cohen A, et al. An international consensus study of neuroleptic malignant syndrome diagnostic criteria using the Delphi method. J Clin Psychiatry. 2011;72(9):1222-1228. doi:10.4088/JCP.10m06438
5. Sienaert P, Dhossche DM, Vancampfort D, et al. A clinical review of the treatment of catatonia. Front Psychiatry. 2014;5:181. doi:10.3389/fpsyt.2014.00181
6. Daniels J. Catatonia: clinical aspects and neurobiological correlates. J Neuropsychiatry Clin Neurosci. 2009;21(4):371-380. doi:10.1176/jnp.2009.21.4.371
7. Bhanushali MJ, Tuite PJ. The evaluation and management of patients with neuroleptic malignant syndrome. Neurol Clin. 2004;22(2):389-411. doi:10.1016/j.ncl.2003.12.006
8. Tse L, Barr AM, Scarapicchia V, et al. Neuroleptic malignant syndrome: a review from a clinically oriented perspective. Curr Neuropharmacol. 2015;13(3):395-406. doi:10.2174/1570159x13999150424113345
9. Correll CU, Kim E, Sliwa JK, et al. Pharmacokinetic characteristics of long-acting injectable antipsychotics for schizophrenia: an overview. CNS Drugs. 2021;35(1):39-59. doi:10.1007/s40263-020-00779-5
10. Strawn JR, Keck PE Jr, Caroff SN. Neuroleptic malignant syndrome. Am J Psychiatry. 2007;164(6):870-876. doi:10.1176/ajp.2007.164.6.870
11. Griffin CE 3rd, Kaye AM, Bueno FR, et al. Benzodiazepine pharmacology and central nervous system-mediated effects. Ochsner J. 2013;13(2):214-223.
12. Reulbach U, Dütsch C, Biermann T, et al. Managing an effective treatment for neuroleptic malignant syndrome. Crit Care. 2007;11(1):R4. doi:10.1186/cc5148
1. Zeitler P, Haqq A, Rosenbloom A, et al. Hyperglycemic hyperosmolar syndrome in children: pathophysiological considerations and suggested guidelines for treatment. J Pediatr. 2011;158(1):9-14.e1-2. doi: 10.1016/j.jpeds.2010.09.048
2. Francis A. Catatonia: diagnosis, classification, and treatment. Curr Psychiatry Rep. 2010;12(3):180-185. doi: 10.1007/s11920-010-0113-y
3. Pileggi DJ, Cook AM. Neuroleptic malignant syndrome. Ann Pharmacother. 2016;50(11):973-981. doi:10.1177/1060028016657553
4. Gurrera RJ, Caroff SN, Cohen A, et al. An international consensus study of neuroleptic malignant syndrome diagnostic criteria using the Delphi method. J Clin Psychiatry. 2011;72(9):1222-1228. doi:10.4088/JCP.10m06438
5. Sienaert P, Dhossche DM, Vancampfort D, et al. A clinical review of the treatment of catatonia. Front Psychiatry. 2014;5:181. doi:10.3389/fpsyt.2014.00181
6. Daniels J. Catatonia: clinical aspects and neurobiological correlates. J Neuropsychiatry Clin Neurosci. 2009;21(4):371-380. doi:10.1176/jnp.2009.21.4.371
7. Bhanushali MJ, Tuite PJ. The evaluation and management of patients with neuroleptic malignant syndrome. Neurol Clin. 2004;22(2):389-411. doi:10.1016/j.ncl.2003.12.006
8. Tse L, Barr AM, Scarapicchia V, et al. Neuroleptic malignant syndrome: a review from a clinically oriented perspective. Curr Neuropharmacol. 2015;13(3):395-406. doi:10.2174/1570159x13999150424113345
9. Correll CU, Kim E, Sliwa JK, et al. Pharmacokinetic characteristics of long-acting injectable antipsychotics for schizophrenia: an overview. CNS Drugs. 2021;35(1):39-59. doi:10.1007/s40263-020-00779-5
10. Strawn JR, Keck PE Jr, Caroff SN. Neuroleptic malignant syndrome. Am J Psychiatry. 2007;164(6):870-876. doi:10.1176/ajp.2007.164.6.870
11. Griffin CE 3rd, Kaye AM, Bueno FR, et al. Benzodiazepine pharmacology and central nervous system-mediated effects. Ochsner J. 2013;13(2):214-223.
12. Reulbach U, Dütsch C, Biermann T, et al. Managing an effective treatment for neuroleptic malignant syndrome. Crit Care. 2007;11(1):R4. doi:10.1186/cc5148
COVID-19: Greater mortality among psych patients remains a mystery
Antipsychotics are not responsible for the increased COVID-related death rate among patients with serious mental illness (SMI), new research shows.
The significant increase in COVID-19 mortality that continues to be reported among those with schizophrenia and schizoaffective disorder “underscores the importance of protective interventions for this group, including priority vaccination,” study investigator Katlyn Nemani, MD, research assistant professor, department of psychiatry, New York University, told this news organization.
The study was published online September 22 in JAMA Psychiatry.
Threefold increase in death
Previous research has linked a diagnosis of a schizophrenia spectrum disorder, which includes schizophrenia and schizoaffective disorder, to an almost threefold increase in mortality among patients with COVID-19.
Some population-based research has also reported a link between antipsychotic medication use and increased risk for COVID-related mortality, but these studies did not take psychiatric diagnoses into account.
“This raised the question of whether the increased risk observed in this population is related to underlying psychiatric illness or its treatment,” said Dr. Nemani.
The retrospective cohort study included 464 adults (mean age, 53 years) who were diagnosed with COVID-19 between March 3, 2020, and Feb. 17, 2021, and who had previously been diagnosed with schizophrenia spectrum disorder or bipolar disorder. Of these, 42.2% were treated with an antipsychotic medication.
The primary endpoint was death within 60 days of COVID-19 diagnosis. Covariates included sociodemographic characteristics, such as patient-reported race and ethnicity, age, and insurance type, a psychiatric diagnosis, medical comorbidities, and smoking status.
Of the total, 41 patients (8.8%) died. The 60-day fatality rate was 13.7% among patients with a schizophrenia spectrum disorder (n = 182) and 5.7% among patients with bipolar disorder (n = 282).
Antipsychotic treatment was not significantly associated with mortality (odds ratio, 1.00; 95% confidence interval, 0.48-2.08; P = .99).
“This suggests that antipsychotic medication is unlikely to be responsible for the increased risk we’ve observed in this population, although this finding needs to be replicated,” said Dr. Nemani.
Surprise finding
A diagnosis of a schizophrenia spectrum disorder was associated with an almost threefold increased risk for mortality compared with bipolar disorder (OR, 2.88; 95% CI, 1.36-6.11; P = .006).
“This was a surprising finding,” said Dr. Nemani.
She noted that there is evidence suggesting the immune system may play a role in the pathogenesis of schizophrenia, and research has shown that pneumonia and infection are among the leading causes of premature mortality in this population.
As well, several potential risk factors disproportionately affect people with serious mental illness, including an increase in the prevalence of medical comorbidities such as cardiovascular disease and diabetes, socioeconomic disadvantages, and barriers to accessing timely care. Prior studies have also found that people with SMI are less likely to receive preventive care interventions, including vaccination, said Dr. Nemani.
However, these factors are unlikely to fully account for the increased risk found in the study, she said.
“Our study population was limited to people who had received treatment within the NYU Langone Health System. We took a comprehensive list of sociodemographic and medical risk factors into account, and our research was conducted prior to the availability of COVID-19 vaccines,” she said.
Further research is necessary to understand what underlies the increase in susceptibility to severe infection among patients with schizophrenia and to identify interventions that may mitigate risk, said Dr. Nemani.
“This includes evaluating systems-level factors, such as access to preventive interventions and treatment, as well as investigating underlying immune mechanisms that may contribute to severe and fatal infection,” she said.
The researchers could not validate psychiatric diagnoses or capture deaths not documented in the electronic health record. In addition, the limited sample size precluded analysis of the use of individual antipsychotic medications, which may differ in their associated effects.
“It’s possible individual antipsychotic medications may be associated with harmful or protective effects,” said Dr. Nemani.
The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Antipsychotics are not responsible for the increased COVID-related death rate among patients with serious mental illness (SMI), new research shows.
The significant increase in COVID-19 mortality that continues to be reported among those with schizophrenia and schizoaffective disorder “underscores the importance of protective interventions for this group, including priority vaccination,” study investigator Katlyn Nemani, MD, research assistant professor, department of psychiatry, New York University, told this news organization.
The study was published online September 22 in JAMA Psychiatry.
Threefold increase in death
Previous research has linked a diagnosis of a schizophrenia spectrum disorder, which includes schizophrenia and schizoaffective disorder, to an almost threefold increase in mortality among patients with COVID-19.
Some population-based research has also reported a link between antipsychotic medication use and increased risk for COVID-related mortality, but these studies did not take psychiatric diagnoses into account.
“This raised the question of whether the increased risk observed in this population is related to underlying psychiatric illness or its treatment,” said Dr. Nemani.
The retrospective cohort study included 464 adults (mean age, 53 years) who were diagnosed with COVID-19 between March 3, 2020, and Feb. 17, 2021, and who had previously been diagnosed with schizophrenia spectrum disorder or bipolar disorder. Of these, 42.2% were treated with an antipsychotic medication.
The primary endpoint was death within 60 days of COVID-19 diagnosis. Covariates included sociodemographic characteristics, such as patient-reported race and ethnicity, age, and insurance type, a psychiatric diagnosis, medical comorbidities, and smoking status.
Of the total, 41 patients (8.8%) died. The 60-day fatality rate was 13.7% among patients with a schizophrenia spectrum disorder (n = 182) and 5.7% among patients with bipolar disorder (n = 282).
Antipsychotic treatment was not significantly associated with mortality (odds ratio, 1.00; 95% confidence interval, 0.48-2.08; P = .99).
“This suggests that antipsychotic medication is unlikely to be responsible for the increased risk we’ve observed in this population, although this finding needs to be replicated,” said Dr. Nemani.
Surprise finding
A diagnosis of a schizophrenia spectrum disorder was associated with an almost threefold increased risk for mortality compared with bipolar disorder (OR, 2.88; 95% CI, 1.36-6.11; P = .006).
“This was a surprising finding,” said Dr. Nemani.
She noted that there is evidence suggesting the immune system may play a role in the pathogenesis of schizophrenia, and research has shown that pneumonia and infection are among the leading causes of premature mortality in this population.
As well, several potential risk factors disproportionately affect people with serious mental illness, including an increase in the prevalence of medical comorbidities such as cardiovascular disease and diabetes, socioeconomic disadvantages, and barriers to accessing timely care. Prior studies have also found that people with SMI are less likely to receive preventive care interventions, including vaccination, said Dr. Nemani.
However, these factors are unlikely to fully account for the increased risk found in the study, she said.
“Our study population was limited to people who had received treatment within the NYU Langone Health System. We took a comprehensive list of sociodemographic and medical risk factors into account, and our research was conducted prior to the availability of COVID-19 vaccines,” she said.
Further research is necessary to understand what underlies the increase in susceptibility to severe infection among patients with schizophrenia and to identify interventions that may mitigate risk, said Dr. Nemani.
“This includes evaluating systems-level factors, such as access to preventive interventions and treatment, as well as investigating underlying immune mechanisms that may contribute to severe and fatal infection,” she said.
The researchers could not validate psychiatric diagnoses or capture deaths not documented in the electronic health record. In addition, the limited sample size precluded analysis of the use of individual antipsychotic medications, which may differ in their associated effects.
“It’s possible individual antipsychotic medications may be associated with harmful or protective effects,” said Dr. Nemani.
The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Antipsychotics are not responsible for the increased COVID-related death rate among patients with serious mental illness (SMI), new research shows.
The significant increase in COVID-19 mortality that continues to be reported among those with schizophrenia and schizoaffective disorder “underscores the importance of protective interventions for this group, including priority vaccination,” study investigator Katlyn Nemani, MD, research assistant professor, department of psychiatry, New York University, told this news organization.
The study was published online September 22 in JAMA Psychiatry.
Threefold increase in death
Previous research has linked a diagnosis of a schizophrenia spectrum disorder, which includes schizophrenia and schizoaffective disorder, to an almost threefold increase in mortality among patients with COVID-19.
Some population-based research has also reported a link between antipsychotic medication use and increased risk for COVID-related mortality, but these studies did not take psychiatric diagnoses into account.
“This raised the question of whether the increased risk observed in this population is related to underlying psychiatric illness or its treatment,” said Dr. Nemani.
The retrospective cohort study included 464 adults (mean age, 53 years) who were diagnosed with COVID-19 between March 3, 2020, and Feb. 17, 2021, and who had previously been diagnosed with schizophrenia spectrum disorder or bipolar disorder. Of these, 42.2% were treated with an antipsychotic medication.
The primary endpoint was death within 60 days of COVID-19 diagnosis. Covariates included sociodemographic characteristics, such as patient-reported race and ethnicity, age, and insurance type, a psychiatric diagnosis, medical comorbidities, and smoking status.
Of the total, 41 patients (8.8%) died. The 60-day fatality rate was 13.7% among patients with a schizophrenia spectrum disorder (n = 182) and 5.7% among patients with bipolar disorder (n = 282).
Antipsychotic treatment was not significantly associated with mortality (odds ratio, 1.00; 95% confidence interval, 0.48-2.08; P = .99).
“This suggests that antipsychotic medication is unlikely to be responsible for the increased risk we’ve observed in this population, although this finding needs to be replicated,” said Dr. Nemani.
Surprise finding
A diagnosis of a schizophrenia spectrum disorder was associated with an almost threefold increased risk for mortality compared with bipolar disorder (OR, 2.88; 95% CI, 1.36-6.11; P = .006).
“This was a surprising finding,” said Dr. Nemani.
She noted that there is evidence suggesting the immune system may play a role in the pathogenesis of schizophrenia, and research has shown that pneumonia and infection are among the leading causes of premature mortality in this population.
As well, several potential risk factors disproportionately affect people with serious mental illness, including an increase in the prevalence of medical comorbidities such as cardiovascular disease and diabetes, socioeconomic disadvantages, and barriers to accessing timely care. Prior studies have also found that people with SMI are less likely to receive preventive care interventions, including vaccination, said Dr. Nemani.
However, these factors are unlikely to fully account for the increased risk found in the study, she said.
“Our study population was limited to people who had received treatment within the NYU Langone Health System. We took a comprehensive list of sociodemographic and medical risk factors into account, and our research was conducted prior to the availability of COVID-19 vaccines,” she said.
Further research is necessary to understand what underlies the increase in susceptibility to severe infection among patients with schizophrenia and to identify interventions that may mitigate risk, said Dr. Nemani.
“This includes evaluating systems-level factors, such as access to preventive interventions and treatment, as well as investigating underlying immune mechanisms that may contribute to severe and fatal infection,” she said.
The researchers could not validate psychiatric diagnoses or capture deaths not documented in the electronic health record. In addition, the limited sample size precluded analysis of the use of individual antipsychotic medications, which may differ in their associated effects.
“It’s possible individual antipsychotic medications may be associated with harmful or protective effects,” said Dr. Nemani.
The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Antipsychotic effective for bipolar depression in phase 3 trial
Results of a phase 3 study show that treatment with lumateperone (Caplyta) significantly improved depressive symptoms for patients with major depressive episodes associated with both bipolar I and bipolar II disorders.
“Bipolar depression represents the most prevalent and debilitating presentation of bipolar disorder. There is a critical need for more treatments that are effective and have favorable safety profiles,” study investigator Gary S. Sachs, MD, associate clinical professor in psychiatry, Harvard Medical School, Boston, said in a company news release.
“The strong efficacy and impressive safety results reported in this trial for a broad patient population position lumateperone as a potentially important advancement in the treatment of this disorder,” said Dr. Sachs, who is also founding director of the Bipolar Clinic and Research Program at Massachusetts General Hospital, Boston.
The findings were published online September 23 in the American Journal of Psychiatry.
First-in-class antipsychotic
Lumateperone is a first-in-class antipsychotic that acts synergistically through the serotonergic, dopaminergic, and glutamatergic systems.
It was approved by the U.S. Food and Drug Administration in late 2019 for the treatment of adults with schizophrenia, as reported at the time by this news organization.
All were randomly allocated in a 1:1 ratio to receive 6 weeks of lumateperone monotherapy at 42 mg/d or matching placebo.
At day 43, lumateperone treatment was associated with significantly greater improvement from baseline in Montgomery-Åsberg Depression Rating Scale (MADRS) score in comparison with placebo (drug-placebo difference, -4.6 points; P < .0001; effect size = -0.56), which met the study’s primary endpoint.
The study drug led to significant improvement in MADRS total score as early as the first week, which was the first time point measured. Improvement continued throughout the study.
Treatment with lumateperone also led to significantly greater improvement in the key secondary endpoints of total score on the severity scale of the Clinical Global Impressions Scale–Bipolar Version (CGI-BP-S) (P < .0001; effect size = -0.46) and the CGI-BP-S depression score (P < .001; effect size = -50).
In addition, it was superior to placebo both for patients with bipolar I disorder and those with bipolar II disorder.
Somnolence and nausea were the most commonly reported adverse events associated with lumateperone. Minimal changes were observed in weight and vital signs and in results of metabolic or endocrine assessments. These findings are in line with previous studies involving patients with schizophrenia.
The incidence of extrapyramidal symptom–related events was low and was similar to those with placebo.
The company has submitted a supplemental new drug application for lumateperone for the treatment of bipolar depression, which is currently under review with the FDA. The target action date is December 17.
A version of this article first appeared on Medscape.com.
Results of a phase 3 study show that treatment with lumateperone (Caplyta) significantly improved depressive symptoms for patients with major depressive episodes associated with both bipolar I and bipolar II disorders.
“Bipolar depression represents the most prevalent and debilitating presentation of bipolar disorder. There is a critical need for more treatments that are effective and have favorable safety profiles,” study investigator Gary S. Sachs, MD, associate clinical professor in psychiatry, Harvard Medical School, Boston, said in a company news release.
“The strong efficacy and impressive safety results reported in this trial for a broad patient population position lumateperone as a potentially important advancement in the treatment of this disorder,” said Dr. Sachs, who is also founding director of the Bipolar Clinic and Research Program at Massachusetts General Hospital, Boston.
The findings were published online September 23 in the American Journal of Psychiatry.
First-in-class antipsychotic
Lumateperone is a first-in-class antipsychotic that acts synergistically through the serotonergic, dopaminergic, and glutamatergic systems.
It was approved by the U.S. Food and Drug Administration in late 2019 for the treatment of adults with schizophrenia, as reported at the time by this news organization.
All were randomly allocated in a 1:1 ratio to receive 6 weeks of lumateperone monotherapy at 42 mg/d or matching placebo.
At day 43, lumateperone treatment was associated with significantly greater improvement from baseline in Montgomery-Åsberg Depression Rating Scale (MADRS) score in comparison with placebo (drug-placebo difference, -4.6 points; P < .0001; effect size = -0.56), which met the study’s primary endpoint.
The study drug led to significant improvement in MADRS total score as early as the first week, which was the first time point measured. Improvement continued throughout the study.
Treatment with lumateperone also led to significantly greater improvement in the key secondary endpoints of total score on the severity scale of the Clinical Global Impressions Scale–Bipolar Version (CGI-BP-S) (P < .0001; effect size = -0.46) and the CGI-BP-S depression score (P < .001; effect size = -50).
In addition, it was superior to placebo both for patients with bipolar I disorder and those with bipolar II disorder.
Somnolence and nausea were the most commonly reported adverse events associated with lumateperone. Minimal changes were observed in weight and vital signs and in results of metabolic or endocrine assessments. These findings are in line with previous studies involving patients with schizophrenia.
The incidence of extrapyramidal symptom–related events was low and was similar to those with placebo.
The company has submitted a supplemental new drug application for lumateperone for the treatment of bipolar depression, which is currently under review with the FDA. The target action date is December 17.
A version of this article first appeared on Medscape.com.
Results of a phase 3 study show that treatment with lumateperone (Caplyta) significantly improved depressive symptoms for patients with major depressive episodes associated with both bipolar I and bipolar II disorders.
“Bipolar depression represents the most prevalent and debilitating presentation of bipolar disorder. There is a critical need for more treatments that are effective and have favorable safety profiles,” study investigator Gary S. Sachs, MD, associate clinical professor in psychiatry, Harvard Medical School, Boston, said in a company news release.
“The strong efficacy and impressive safety results reported in this trial for a broad patient population position lumateperone as a potentially important advancement in the treatment of this disorder,” said Dr. Sachs, who is also founding director of the Bipolar Clinic and Research Program at Massachusetts General Hospital, Boston.
The findings were published online September 23 in the American Journal of Psychiatry.
First-in-class antipsychotic
Lumateperone is a first-in-class antipsychotic that acts synergistically through the serotonergic, dopaminergic, and glutamatergic systems.
It was approved by the U.S. Food and Drug Administration in late 2019 for the treatment of adults with schizophrenia, as reported at the time by this news organization.
All were randomly allocated in a 1:1 ratio to receive 6 weeks of lumateperone monotherapy at 42 mg/d or matching placebo.
At day 43, lumateperone treatment was associated with significantly greater improvement from baseline in Montgomery-Åsberg Depression Rating Scale (MADRS) score in comparison with placebo (drug-placebo difference, -4.6 points; P < .0001; effect size = -0.56), which met the study’s primary endpoint.
The study drug led to significant improvement in MADRS total score as early as the first week, which was the first time point measured. Improvement continued throughout the study.
Treatment with lumateperone also led to significantly greater improvement in the key secondary endpoints of total score on the severity scale of the Clinical Global Impressions Scale–Bipolar Version (CGI-BP-S) (P < .0001; effect size = -0.46) and the CGI-BP-S depression score (P < .001; effect size = -50).
In addition, it was superior to placebo both for patients with bipolar I disorder and those with bipolar II disorder.
Somnolence and nausea were the most commonly reported adverse events associated with lumateperone. Minimal changes were observed in weight and vital signs and in results of metabolic or endocrine assessments. These findings are in line with previous studies involving patients with schizophrenia.
The incidence of extrapyramidal symptom–related events was low and was similar to those with placebo.
The company has submitted a supplemental new drug application for lumateperone for the treatment of bipolar depression, which is currently under review with the FDA. The target action date is December 17.
A version of this article first appeared on Medscape.com.
Gut health ‘vitally important’ for mental health
Disturbances in gut microbiota are associated with depletion of anti-inflammatory bacteria and proliferation of proinflammatory bacteria, a pattern tied to several major psychiatric disorders including depression, bipolar disorder (BD), schizophrenia, and anxiety, new research shows.
A meta-analysis of 59 studies, encompassing roughly 2,600 patients with psychiatric conditions, showed a decrease in microbial richness in patients with psychiatric conditions versus controls.
In addition, those with depression, anxiety, BD, and psychosis had a similar set of abnormalities in the microbiota, particularly lower levels of Faecalibacterium and Coprococcus – two types of bacteria that have an anti-inflammatory effect in gut – and higher levels of Eggerthella, a bacterium with proinflammatory effects.
“The wealth of evidence we have summarized clearly demonstrates that the gut microbiota is vitally important to the wider mental health of individuals,” lead author Viktoriya Nikolova, MRes, Centre for Affective Disorders, King’s College London, said in an interview.
“While it is still too early to recommend specific interventions, it’s clear that clinicians need to place a greater awareness of gut health when considering the treatment of certain psychiatric disorders,” she said.
The study was published online Sept. 15, 2021, in JAMA Psychiatry.
Reliable biomarkers
“Evidence of gut microbiota perturbations has accumulated for multiple psychiatric disorders, with microbiota signatures proposed as potential biomarkers,” the authors wrote.
However, “while there is a wealth of evidence to suggest that abnormalities within the composition of the gut microbiota are connected to a number of psychiatric disorders, there haven’t been any attempts to evaluate the specificity of this evidence – that is, if these changes are unique to specific disorders or shared across many,” Ms. Nikolova said.
Previous research in individual disorders has identified “patterns that may be promising biomarker targets,” with the potential to “improve diagnostic accuracy, guide treatment, and assist the monitoring of treatment response,” the authors noted.
“We wanted to see if we could reliably establish biomarkers for individual conditions in an effort to further our understanding of the relationship between mental illness and gut microbiota,” said Ms. Nikolova.
The researchers wanted to “evaluate the specificity and reproducibility of gut microbiota alterations and delineate those with potential to become biomarkers.”
They identified 59 studies (64 case-control comparisons; n = 2,643 patients, 2,336 controls). Most (54.2%) were conducted in East Asia, followed by Westernized populations (40.7%) and Africa (1.7%).
These studies evaluated diversity or abundance of gut microbes in adult populations encompassing an array of psychiatric disorders: major depressive disorder (MDD), BD, psychosis and schizophrenia, eating disorders (anorexia nervosa and bulimia nervosa), anxiety, obsessive-compulsive disorder (OCD), PTSD, and ADHD.
Although studies were similar in exclusion criteria, few attempted to minimize dietary changes or control dietary intake. In addition, use of psychiatric medication also “varied substantially.”
The researchers conducted several analyses, with primary outcomes consisting of “community-level measures of gut microbiota composition (alpha and beta diversity) as well as taxonomic findings at the phylum, family, and genus levels (relative abundance).”
Alpha diversity provides a “summary of the microbial community in individual samples,” which “can be compared across groups to evaluate the role of a particular factor (in this case psychiatric diagnosis) on the richness (number of species) and evenness (how well each species is represented) in the sample.”
Beta diversity, on the other hand, “measures interindividual (between samples) diversity that assesses similarity of communities, compared with the other samples analyzed.”
Control samples consisted of participants without the relevant condition.
Biological overlap?
The alpha-diversity meta-analysis encompassed 34 studies (n = 1,519 patients, 1,429 controls). The researchers found significant decreases in microbial richness in patients, compared with controls (observed species standardized mean difference, −0.26; 95% CI, −0.47 to −0.06; Chao1 SMD, −0.5; 95% CI, −0.79 to −0.21). On the other hand, when they examined each diagnosis separately, they found consistent decreases only in bipolar disorder. There was a small, nonsignificant decrease in phylogenetic diversity between groups.
MDD, psychosis, and schizophrenia were the only conditions in which differences in beta diversity were consistently observed.
“These findings suggest there is reliable evidence for differences in the shared phylogenetic structure in MDD and psychosis and schizophrenia compared with controls,” the authors write.
However, “method of measurement and method of patient classification (symptom vs. diagnosis based) may affect findings,” they added.
When they focused on relative abundance, they found “little evidence” of disorder specificity, but rather a “transdiagnostic pattern of microbiota signatures.”
In particular, depleted levels of Faecalibacterium and Coprococcus and enriched levels of Eggerthella were “consistently shared” between MDD, BD, psychosis and schizophrenia, and anxiety, “suggesting these disorders are characterized by a reduction of anti-inflammatory butyrate-producing bacteria, while proinflammatory genera are enriched.”
“The finding that these perturbations do not appear to be disorder-specific suggests that the microbiota is affected in a similar manner by conditions such as depression, anxiety, bipolar disorder, and psychosis,” said Ms. Nikolova.
“We have seen similar findings from previous meta-analyses of inflammatory marker studies and genetic studies, for example, suggesting that there is a biological overlap between these conditions, which we have now also seen in the microbiota.”
The authors highlighted potential confounders, including study region and medication use.
Conditions such as MDD, psychosis, and schizophrenia were “largely investigated in the East,” while anorexia nervosa and OCD were primarily investigated in the West.
Moreover, comparing results from medication-free studies with those in which 80% or more of patients were taking psychiatric medication showed increases in bacterial families Lactobacillaceae, Klebsiella, Streptococcus, and Megasphaera only in medicated groups, and decreases in Dialister.
In light of these confounders, the findings should be considered “preliminary,” the investigators noted.
Greater standardization needed
Commenting on the study, Emeran Mayer, MD, director of the Oppenheimer Center for Neurobiology of Stress and Resilience at the University of California, Los Angeles, said it is “intriguing to speculate that low-grade immune activation due to reduced production of butyrate may be such a generalized factor affecting microbial composition shared similarly in several brain disorders. However, such a mechanism has not been confirmed in mechanistic studies to date.”
In addition, the study “lumps together a large number of studies and heterogeneous patient populations, with and without centrally acting medication, without adequate dietary history, studied in different ethnic populations, studied with highly variable collection and analysis methods, including highly variable sample and study sizes for different diseases, and using only measures of microbial composition but not function,” cautioned Dr. Mayer, who was not involved in the research.
Future studies “with much greater standardization of subject populations and clinical and biological analyses techniques should be performed to reevaluate the results of the current study and confirm or reject the main hypotheses,” asserted Dr. Mayer, who is also the founding director of the UCLA Brain Gut Microbiome Center.
Ms. Nikolova is funded by a Medical Research Council PhD Studentship. Other sources of funding include the National Institute for Health Research Biomedical Research Centre at South London and Maudsley National Health Service Foundation Trust and King’s College London. Ms. Nikolova has disclosed no relevant financial relationships. Dr. Mayer is a scientific advisory board member of Danone, Axial Therapeutics, Viome, Amare, Mahana Therapeutics, Pendulum, Bloom Biosciences, and APC Microbiome Ireland.
A version of this article first appeared on Medscape.com .
Disturbances in gut microbiota are associated with depletion of anti-inflammatory bacteria and proliferation of proinflammatory bacteria, a pattern tied to several major psychiatric disorders including depression, bipolar disorder (BD), schizophrenia, and anxiety, new research shows.
A meta-analysis of 59 studies, encompassing roughly 2,600 patients with psychiatric conditions, showed a decrease in microbial richness in patients with psychiatric conditions versus controls.
In addition, those with depression, anxiety, BD, and psychosis had a similar set of abnormalities in the microbiota, particularly lower levels of Faecalibacterium and Coprococcus – two types of bacteria that have an anti-inflammatory effect in gut – and higher levels of Eggerthella, a bacterium with proinflammatory effects.
“The wealth of evidence we have summarized clearly demonstrates that the gut microbiota is vitally important to the wider mental health of individuals,” lead author Viktoriya Nikolova, MRes, Centre for Affective Disorders, King’s College London, said in an interview.
“While it is still too early to recommend specific interventions, it’s clear that clinicians need to place a greater awareness of gut health when considering the treatment of certain psychiatric disorders,” she said.
The study was published online Sept. 15, 2021, in JAMA Psychiatry.
Reliable biomarkers
“Evidence of gut microbiota perturbations has accumulated for multiple psychiatric disorders, with microbiota signatures proposed as potential biomarkers,” the authors wrote.
However, “while there is a wealth of evidence to suggest that abnormalities within the composition of the gut microbiota are connected to a number of psychiatric disorders, there haven’t been any attempts to evaluate the specificity of this evidence – that is, if these changes are unique to specific disorders or shared across many,” Ms. Nikolova said.
Previous research in individual disorders has identified “patterns that may be promising biomarker targets,” with the potential to “improve diagnostic accuracy, guide treatment, and assist the monitoring of treatment response,” the authors noted.
“We wanted to see if we could reliably establish biomarkers for individual conditions in an effort to further our understanding of the relationship between mental illness and gut microbiota,” said Ms. Nikolova.
The researchers wanted to “evaluate the specificity and reproducibility of gut microbiota alterations and delineate those with potential to become biomarkers.”
They identified 59 studies (64 case-control comparisons; n = 2,643 patients, 2,336 controls). Most (54.2%) were conducted in East Asia, followed by Westernized populations (40.7%) and Africa (1.7%).
These studies evaluated diversity or abundance of gut microbes in adult populations encompassing an array of psychiatric disorders: major depressive disorder (MDD), BD, psychosis and schizophrenia, eating disorders (anorexia nervosa and bulimia nervosa), anxiety, obsessive-compulsive disorder (OCD), PTSD, and ADHD.
Although studies were similar in exclusion criteria, few attempted to minimize dietary changes or control dietary intake. In addition, use of psychiatric medication also “varied substantially.”
The researchers conducted several analyses, with primary outcomes consisting of “community-level measures of gut microbiota composition (alpha and beta diversity) as well as taxonomic findings at the phylum, family, and genus levels (relative abundance).”
Alpha diversity provides a “summary of the microbial community in individual samples,” which “can be compared across groups to evaluate the role of a particular factor (in this case psychiatric diagnosis) on the richness (number of species) and evenness (how well each species is represented) in the sample.”
Beta diversity, on the other hand, “measures interindividual (between samples) diversity that assesses similarity of communities, compared with the other samples analyzed.”
Control samples consisted of participants without the relevant condition.
Biological overlap?
The alpha-diversity meta-analysis encompassed 34 studies (n = 1,519 patients, 1,429 controls). The researchers found significant decreases in microbial richness in patients, compared with controls (observed species standardized mean difference, −0.26; 95% CI, −0.47 to −0.06; Chao1 SMD, −0.5; 95% CI, −0.79 to −0.21). On the other hand, when they examined each diagnosis separately, they found consistent decreases only in bipolar disorder. There was a small, nonsignificant decrease in phylogenetic diversity between groups.
MDD, psychosis, and schizophrenia were the only conditions in which differences in beta diversity were consistently observed.
“These findings suggest there is reliable evidence for differences in the shared phylogenetic structure in MDD and psychosis and schizophrenia compared with controls,” the authors write.
However, “method of measurement and method of patient classification (symptom vs. diagnosis based) may affect findings,” they added.
When they focused on relative abundance, they found “little evidence” of disorder specificity, but rather a “transdiagnostic pattern of microbiota signatures.”
In particular, depleted levels of Faecalibacterium and Coprococcus and enriched levels of Eggerthella were “consistently shared” between MDD, BD, psychosis and schizophrenia, and anxiety, “suggesting these disorders are characterized by a reduction of anti-inflammatory butyrate-producing bacteria, while proinflammatory genera are enriched.”
“The finding that these perturbations do not appear to be disorder-specific suggests that the microbiota is affected in a similar manner by conditions such as depression, anxiety, bipolar disorder, and psychosis,” said Ms. Nikolova.
“We have seen similar findings from previous meta-analyses of inflammatory marker studies and genetic studies, for example, suggesting that there is a biological overlap between these conditions, which we have now also seen in the microbiota.”
The authors highlighted potential confounders, including study region and medication use.
Conditions such as MDD, psychosis, and schizophrenia were “largely investigated in the East,” while anorexia nervosa and OCD were primarily investigated in the West.
Moreover, comparing results from medication-free studies with those in which 80% or more of patients were taking psychiatric medication showed increases in bacterial families Lactobacillaceae, Klebsiella, Streptococcus, and Megasphaera only in medicated groups, and decreases in Dialister.
In light of these confounders, the findings should be considered “preliminary,” the investigators noted.
Greater standardization needed
Commenting on the study, Emeran Mayer, MD, director of the Oppenheimer Center for Neurobiology of Stress and Resilience at the University of California, Los Angeles, said it is “intriguing to speculate that low-grade immune activation due to reduced production of butyrate may be such a generalized factor affecting microbial composition shared similarly in several brain disorders. However, such a mechanism has not been confirmed in mechanistic studies to date.”
In addition, the study “lumps together a large number of studies and heterogeneous patient populations, with and without centrally acting medication, without adequate dietary history, studied in different ethnic populations, studied with highly variable collection and analysis methods, including highly variable sample and study sizes for different diseases, and using only measures of microbial composition but not function,” cautioned Dr. Mayer, who was not involved in the research.
Future studies “with much greater standardization of subject populations and clinical and biological analyses techniques should be performed to reevaluate the results of the current study and confirm or reject the main hypotheses,” asserted Dr. Mayer, who is also the founding director of the UCLA Brain Gut Microbiome Center.
Ms. Nikolova is funded by a Medical Research Council PhD Studentship. Other sources of funding include the National Institute for Health Research Biomedical Research Centre at South London and Maudsley National Health Service Foundation Trust and King’s College London. Ms. Nikolova has disclosed no relevant financial relationships. Dr. Mayer is a scientific advisory board member of Danone, Axial Therapeutics, Viome, Amare, Mahana Therapeutics, Pendulum, Bloom Biosciences, and APC Microbiome Ireland.
A version of this article first appeared on Medscape.com .
Disturbances in gut microbiota are associated with depletion of anti-inflammatory bacteria and proliferation of proinflammatory bacteria, a pattern tied to several major psychiatric disorders including depression, bipolar disorder (BD), schizophrenia, and anxiety, new research shows.
A meta-analysis of 59 studies, encompassing roughly 2,600 patients with psychiatric conditions, showed a decrease in microbial richness in patients with psychiatric conditions versus controls.
In addition, those with depression, anxiety, BD, and psychosis had a similar set of abnormalities in the microbiota, particularly lower levels of Faecalibacterium and Coprococcus – two types of bacteria that have an anti-inflammatory effect in gut – and higher levels of Eggerthella, a bacterium with proinflammatory effects.
“The wealth of evidence we have summarized clearly demonstrates that the gut microbiota is vitally important to the wider mental health of individuals,” lead author Viktoriya Nikolova, MRes, Centre for Affective Disorders, King’s College London, said in an interview.
“While it is still too early to recommend specific interventions, it’s clear that clinicians need to place a greater awareness of gut health when considering the treatment of certain psychiatric disorders,” she said.
The study was published online Sept. 15, 2021, in JAMA Psychiatry.
Reliable biomarkers
“Evidence of gut microbiota perturbations has accumulated for multiple psychiatric disorders, with microbiota signatures proposed as potential biomarkers,” the authors wrote.
However, “while there is a wealth of evidence to suggest that abnormalities within the composition of the gut microbiota are connected to a number of psychiatric disorders, there haven’t been any attempts to evaluate the specificity of this evidence – that is, if these changes are unique to specific disorders or shared across many,” Ms. Nikolova said.
Previous research in individual disorders has identified “patterns that may be promising biomarker targets,” with the potential to “improve diagnostic accuracy, guide treatment, and assist the monitoring of treatment response,” the authors noted.
“We wanted to see if we could reliably establish biomarkers for individual conditions in an effort to further our understanding of the relationship between mental illness and gut microbiota,” said Ms. Nikolova.
The researchers wanted to “evaluate the specificity and reproducibility of gut microbiota alterations and delineate those with potential to become biomarkers.”
They identified 59 studies (64 case-control comparisons; n = 2,643 patients, 2,336 controls). Most (54.2%) were conducted in East Asia, followed by Westernized populations (40.7%) and Africa (1.7%).
These studies evaluated diversity or abundance of gut microbes in adult populations encompassing an array of psychiatric disorders: major depressive disorder (MDD), BD, psychosis and schizophrenia, eating disorders (anorexia nervosa and bulimia nervosa), anxiety, obsessive-compulsive disorder (OCD), PTSD, and ADHD.
Although studies were similar in exclusion criteria, few attempted to minimize dietary changes or control dietary intake. In addition, use of psychiatric medication also “varied substantially.”
The researchers conducted several analyses, with primary outcomes consisting of “community-level measures of gut microbiota composition (alpha and beta diversity) as well as taxonomic findings at the phylum, family, and genus levels (relative abundance).”
Alpha diversity provides a “summary of the microbial community in individual samples,” which “can be compared across groups to evaluate the role of a particular factor (in this case psychiatric diagnosis) on the richness (number of species) and evenness (how well each species is represented) in the sample.”
Beta diversity, on the other hand, “measures interindividual (between samples) diversity that assesses similarity of communities, compared with the other samples analyzed.”
Control samples consisted of participants without the relevant condition.
Biological overlap?
The alpha-diversity meta-analysis encompassed 34 studies (n = 1,519 patients, 1,429 controls). The researchers found significant decreases in microbial richness in patients, compared with controls (observed species standardized mean difference, −0.26; 95% CI, −0.47 to −0.06; Chao1 SMD, −0.5; 95% CI, −0.79 to −0.21). On the other hand, when they examined each diagnosis separately, they found consistent decreases only in bipolar disorder. There was a small, nonsignificant decrease in phylogenetic diversity between groups.
MDD, psychosis, and schizophrenia were the only conditions in which differences in beta diversity were consistently observed.
“These findings suggest there is reliable evidence for differences in the shared phylogenetic structure in MDD and psychosis and schizophrenia compared with controls,” the authors write.
However, “method of measurement and method of patient classification (symptom vs. diagnosis based) may affect findings,” they added.
When they focused on relative abundance, they found “little evidence” of disorder specificity, but rather a “transdiagnostic pattern of microbiota signatures.”
In particular, depleted levels of Faecalibacterium and Coprococcus and enriched levels of Eggerthella were “consistently shared” between MDD, BD, psychosis and schizophrenia, and anxiety, “suggesting these disorders are characterized by a reduction of anti-inflammatory butyrate-producing bacteria, while proinflammatory genera are enriched.”
“The finding that these perturbations do not appear to be disorder-specific suggests that the microbiota is affected in a similar manner by conditions such as depression, anxiety, bipolar disorder, and psychosis,” said Ms. Nikolova.
“We have seen similar findings from previous meta-analyses of inflammatory marker studies and genetic studies, for example, suggesting that there is a biological overlap between these conditions, which we have now also seen in the microbiota.”
The authors highlighted potential confounders, including study region and medication use.
Conditions such as MDD, psychosis, and schizophrenia were “largely investigated in the East,” while anorexia nervosa and OCD were primarily investigated in the West.
Moreover, comparing results from medication-free studies with those in which 80% or more of patients were taking psychiatric medication showed increases in bacterial families Lactobacillaceae, Klebsiella, Streptococcus, and Megasphaera only in medicated groups, and decreases in Dialister.
In light of these confounders, the findings should be considered “preliminary,” the investigators noted.
Greater standardization needed
Commenting on the study, Emeran Mayer, MD, director of the Oppenheimer Center for Neurobiology of Stress and Resilience at the University of California, Los Angeles, said it is “intriguing to speculate that low-grade immune activation due to reduced production of butyrate may be such a generalized factor affecting microbial composition shared similarly in several brain disorders. However, such a mechanism has not been confirmed in mechanistic studies to date.”
In addition, the study “lumps together a large number of studies and heterogeneous patient populations, with and without centrally acting medication, without adequate dietary history, studied in different ethnic populations, studied with highly variable collection and analysis methods, including highly variable sample and study sizes for different diseases, and using only measures of microbial composition but not function,” cautioned Dr. Mayer, who was not involved in the research.
Future studies “with much greater standardization of subject populations and clinical and biological analyses techniques should be performed to reevaluate the results of the current study and confirm or reject the main hypotheses,” asserted Dr. Mayer, who is also the founding director of the UCLA Brain Gut Microbiome Center.
Ms. Nikolova is funded by a Medical Research Council PhD Studentship. Other sources of funding include the National Institute for Health Research Biomedical Research Centre at South London and Maudsley National Health Service Foundation Trust and King’s College London. Ms. Nikolova has disclosed no relevant financial relationships. Dr. Mayer is a scientific advisory board member of Danone, Axial Therapeutics, Viome, Amare, Mahana Therapeutics, Pendulum, Bloom Biosciences, and APC Microbiome Ireland.
A version of this article first appeared on Medscape.com .
The role of probiotics in mental health
In 1950, at Staten Island’s Sea View Hospital, a group of patients with terminal tuberculosis were given a new antibiotic called isoniazid, which caused some unexpected side effects. The patients reported euphoria, mental stimulation, and improved sleep, and even began socializing with more vigor. The press was all over the case, writing about the sick “dancing in the halls tho’ they had holes in their lungs.” Soon doctors started prescribing isoniazid as the first-ever antidepressant.
The Sea View Hospital experiment was an early hint that changing the composition of the gut microbiome – in this case, via antibiotics – might affect our mental health. Yet only in the last 2 decades has research into connections between what we ingest and psychiatric disorders really taken off. In 2004, a landmark study showed that germ-free mice (born in such sterile conditions that they lacked a microbiome) had an exaggerated stress response. The effects were reversed, however, if the mice were fed a bacterial strain, Bifidobacterium infantis, a probiotic. This sparked academic interest, and thousands of research papers followed.
According to Stephen Ilardi, PhD, a clinical psychologist at the University of Kansas, Lawrence, focusing on the etiology and treatment of depression, now is the “time of exciting discovery” in the field of probiotics and psychiatric disorders, although, admittedly, a lot still remains unknown.
Gut microbiome profiles in mental health disorders
We humans have about 100 trillion microbes residing in our guts. Some of these are archaea, some fungi, some protozoans and even viruses, but most are bacteria. Things like diet, sleep, and stress can all impact the composition of our gut microbiome. When the microbiome differs considerably from the typical, doctors and researchers describe it as dysbiosis, or imbalance. Studies have uncovered dysbiosis in patients with depression, anxiety, schizophrenia, and bipolar disorder.
“I think there is now pretty good evidence that the gut microbiome is actually an important factor in a number of psychiatric disorders,” says Allan Young, MBChB, clinical psychiatrist at King’s College London. The gut microbiome composition does seem to differ between psychiatric patients and the healthy. In depression, for example, a recent review of nine studies found an increase on the genus level in Streptococcus and Oscillibacter and low abundance of Lactobacillus and Coprococcus, among others. In generalized anxiety disorder, meanwhile, there appears to be an increase in Fusobacteria and Escherichia/Shigella .
For Dr. Ilardi, the next important question is whether there are plausible mechanisms that could explain how gut microbiota may influence brain function. And, it appears there are.
“The microbes in the gut can release neurotransmitters into blood that cross into the brain and influence brain function. They can release hormones into the blood that again cross into the brain. They’ve got a lot of tricks up their sleeve,” he says.
One particularly important pathway runs through the vagus nerve – the longest nerve that emerges directly from the brain, connecting it to the gut. Another is the immune pathway. Gut bacteria can interact with immune cells and reduce cytokine production, which in turn can reduce systemic inflammation. Inflammatory processes have been implicated in both depression and bipolar disorder. What’s more, gut microbes can upregulate the expression of a protein called BDNF – brain-derived neurotrophic factor – which helps the development and survival of nerve cells in the brain.
Probiotics’ promise varies for different conditions
As the pathways by which gut dysbiosis may influence psychiatric disorders become clearer, the next logical step is to try to influence the composition of the microbiome to prevent and treat depression, anxiety, or schizophrenia. That’s where probiotics come in.
The evidence for the effects of probiotics – live microorganisms which, when ingested in adequate amounts, confer a health benefit – so far is the strongest for depression, says Viktoriya Nikolova, MRes, MSc, a PhD student and researcher at King’s College London. In their 2021 meta-analysis of seven trials, Mr. Nikolova and colleagues revealed that probiotics can significantly reduce depressive symptoms after just 8 weeks. There was a caveat, however – the probiotics only worked when used in addition to an approved antidepressant. Another meta-analysis, published in 2018, also showed that probiotics, when compared with placebo, improve mood in people with depressive symptoms (here, no antidepressant treatment was necessary).
Roumen Milev, MD, PhD, a neuroscientist at Queen’s University, Kingston, Ont., and coauthor of a review on probiotics and depression published in the Annals of General Psychiatry, warns, however, that the research is still in its infancy. “,” he says.
When it comes to using probiotics to relieve anxiety, “the evidence in the animal literature is really compelling,” says Dr. Ilardi. Human studies are less convincing, however, which Dr. Dr. Ilardi showed in his 2018 review and meta-analysis involving 743 animals and 1,527 humans. “Studies are small for the most part, and some of them aren’t terribly well conducted, and they often use very low doses of probiotics,” he says. One of the larger double-blind and placebo-controlled trials showed that supplementation with Lactobacillus plantarum helps reduce stress and anxiety, while the levels of proinflammatory cytokines go down. Another meta-analysis, published in June, revealed that, when it comes to reducing stress and anxiety in youth, the results are mixed.
Evidence of probiotics’ efficiency in schizophrenia is emerging, yet also limited. A 2019 review concluded that currently available results only “hint” at a possibility that probiotics could make a difference in schizophrenia. Similarly, a 2020 review summed up that the role of probiotics in bipolar disorder “remains unclear and underexplored.”
Better studies, remaining questions
Apart from small samples, one issue with research on probiotics is that they generally tend to use varied doses of different strains of bacteria, or even multistrain mixtures, making it tough to compare results. Although there are hundreds of species of bacteria in the human gut, only a few have been evaluated for their antidepressant or antianxiety effects.
“To make it even worse, it’s almost certainly the case that depending on a person’s actual genetics or maybe their epigenetics, a strain that is helpful for one person may not be helpful for another. There is almost certainly no one-size-fits-all probiotic formulation,” says Dr. Ilardi.
Another critical question that remains to be answered is that of potential side effects.
“Probiotics are often seen as food supplements, so they don’t follow under the same regulations as drugs would,” says Mr. Nikolova. “They don’t necessarily have to follow the pattern of drug trials in many countries, which means that the monitoring of side effects is not the requirement.”
That’s something that worries King’s College psychiatrist Young too. “If you are giving it to modulate how the brain works, you could potentially induce psychiatric symptoms or a psychiatric disorder. There could be allergic reactions. There could be lots of different things,” he says.
When you search the web for “probiotics,” chances are you will come across sites boasting amazing effects that such products can have on cardiovascular heath, the immune system, and yes, mental well-being. Many also sell various probiotic supplements “formulated” for your gut health or improved moods. However, many such commercially available strains have never been actually tested in clinical trials. What’s more, according to Kathrin Cohen Kadosh, PhD, a neuroscientist at University of Surrey (England), “it is not always clear whether the different strains actually reach the gut intact.”
For now, considering the limited research evidence, a safer bet is to try to improve gut health through consumption of fermented foods that naturally contain probiotics, such as miso, kefir, or sauerkraut. Alternatively, you could reach for prebiotics, such as foods containing fiber (prebiotics enhance the growth of beneficial gut microbes). This, Dr. Kadosh says, could be “a gentler way of improving gut health” than popping a pill. Whether an improved mental well-being might follow still remains to be seen.
A version of this article first appeared on Medscape.com.
In 1950, at Staten Island’s Sea View Hospital, a group of patients with terminal tuberculosis were given a new antibiotic called isoniazid, which caused some unexpected side effects. The patients reported euphoria, mental stimulation, and improved sleep, and even began socializing with more vigor. The press was all over the case, writing about the sick “dancing in the halls tho’ they had holes in their lungs.” Soon doctors started prescribing isoniazid as the first-ever antidepressant.
The Sea View Hospital experiment was an early hint that changing the composition of the gut microbiome – in this case, via antibiotics – might affect our mental health. Yet only in the last 2 decades has research into connections between what we ingest and psychiatric disorders really taken off. In 2004, a landmark study showed that germ-free mice (born in such sterile conditions that they lacked a microbiome) had an exaggerated stress response. The effects were reversed, however, if the mice were fed a bacterial strain, Bifidobacterium infantis, a probiotic. This sparked academic interest, and thousands of research papers followed.
According to Stephen Ilardi, PhD, a clinical psychologist at the University of Kansas, Lawrence, focusing on the etiology and treatment of depression, now is the “time of exciting discovery” in the field of probiotics and psychiatric disorders, although, admittedly, a lot still remains unknown.
Gut microbiome profiles in mental health disorders
We humans have about 100 trillion microbes residing in our guts. Some of these are archaea, some fungi, some protozoans and even viruses, but most are bacteria. Things like diet, sleep, and stress can all impact the composition of our gut microbiome. When the microbiome differs considerably from the typical, doctors and researchers describe it as dysbiosis, or imbalance. Studies have uncovered dysbiosis in patients with depression, anxiety, schizophrenia, and bipolar disorder.
“I think there is now pretty good evidence that the gut microbiome is actually an important factor in a number of psychiatric disorders,” says Allan Young, MBChB, clinical psychiatrist at King’s College London. The gut microbiome composition does seem to differ between psychiatric patients and the healthy. In depression, for example, a recent review of nine studies found an increase on the genus level in Streptococcus and Oscillibacter and low abundance of Lactobacillus and Coprococcus, among others. In generalized anxiety disorder, meanwhile, there appears to be an increase in Fusobacteria and Escherichia/Shigella .
For Dr. Ilardi, the next important question is whether there are plausible mechanisms that could explain how gut microbiota may influence brain function. And, it appears there are.
“The microbes in the gut can release neurotransmitters into blood that cross into the brain and influence brain function. They can release hormones into the blood that again cross into the brain. They’ve got a lot of tricks up their sleeve,” he says.
One particularly important pathway runs through the vagus nerve – the longest nerve that emerges directly from the brain, connecting it to the gut. Another is the immune pathway. Gut bacteria can interact with immune cells and reduce cytokine production, which in turn can reduce systemic inflammation. Inflammatory processes have been implicated in both depression and bipolar disorder. What’s more, gut microbes can upregulate the expression of a protein called BDNF – brain-derived neurotrophic factor – which helps the development and survival of nerve cells in the brain.
Probiotics’ promise varies for different conditions
As the pathways by which gut dysbiosis may influence psychiatric disorders become clearer, the next logical step is to try to influence the composition of the microbiome to prevent and treat depression, anxiety, or schizophrenia. That’s where probiotics come in.
The evidence for the effects of probiotics – live microorganisms which, when ingested in adequate amounts, confer a health benefit – so far is the strongest for depression, says Viktoriya Nikolova, MRes, MSc, a PhD student and researcher at King’s College London. In their 2021 meta-analysis of seven trials, Mr. Nikolova and colleagues revealed that probiotics can significantly reduce depressive symptoms after just 8 weeks. There was a caveat, however – the probiotics only worked when used in addition to an approved antidepressant. Another meta-analysis, published in 2018, also showed that probiotics, when compared with placebo, improve mood in people with depressive symptoms (here, no antidepressant treatment was necessary).
Roumen Milev, MD, PhD, a neuroscientist at Queen’s University, Kingston, Ont., and coauthor of a review on probiotics and depression published in the Annals of General Psychiatry, warns, however, that the research is still in its infancy. “,” he says.
When it comes to using probiotics to relieve anxiety, “the evidence in the animal literature is really compelling,” says Dr. Ilardi. Human studies are less convincing, however, which Dr. Dr. Ilardi showed in his 2018 review and meta-analysis involving 743 animals and 1,527 humans. “Studies are small for the most part, and some of them aren’t terribly well conducted, and they often use very low doses of probiotics,” he says. One of the larger double-blind and placebo-controlled trials showed that supplementation with Lactobacillus plantarum helps reduce stress and anxiety, while the levels of proinflammatory cytokines go down. Another meta-analysis, published in June, revealed that, when it comes to reducing stress and anxiety in youth, the results are mixed.
Evidence of probiotics’ efficiency in schizophrenia is emerging, yet also limited. A 2019 review concluded that currently available results only “hint” at a possibility that probiotics could make a difference in schizophrenia. Similarly, a 2020 review summed up that the role of probiotics in bipolar disorder “remains unclear and underexplored.”
Better studies, remaining questions
Apart from small samples, one issue with research on probiotics is that they generally tend to use varied doses of different strains of bacteria, or even multistrain mixtures, making it tough to compare results. Although there are hundreds of species of bacteria in the human gut, only a few have been evaluated for their antidepressant or antianxiety effects.
“To make it even worse, it’s almost certainly the case that depending on a person’s actual genetics or maybe their epigenetics, a strain that is helpful for one person may not be helpful for another. There is almost certainly no one-size-fits-all probiotic formulation,” says Dr. Ilardi.
Another critical question that remains to be answered is that of potential side effects.
“Probiotics are often seen as food supplements, so they don’t follow under the same regulations as drugs would,” says Mr. Nikolova. “They don’t necessarily have to follow the pattern of drug trials in many countries, which means that the monitoring of side effects is not the requirement.”
That’s something that worries King’s College psychiatrist Young too. “If you are giving it to modulate how the brain works, you could potentially induce psychiatric symptoms or a psychiatric disorder. There could be allergic reactions. There could be lots of different things,” he says.
When you search the web for “probiotics,” chances are you will come across sites boasting amazing effects that such products can have on cardiovascular heath, the immune system, and yes, mental well-being. Many also sell various probiotic supplements “formulated” for your gut health or improved moods. However, many such commercially available strains have never been actually tested in clinical trials. What’s more, according to Kathrin Cohen Kadosh, PhD, a neuroscientist at University of Surrey (England), “it is not always clear whether the different strains actually reach the gut intact.”
For now, considering the limited research evidence, a safer bet is to try to improve gut health through consumption of fermented foods that naturally contain probiotics, such as miso, kefir, or sauerkraut. Alternatively, you could reach for prebiotics, such as foods containing fiber (prebiotics enhance the growth of beneficial gut microbes). This, Dr. Kadosh says, could be “a gentler way of improving gut health” than popping a pill. Whether an improved mental well-being might follow still remains to be seen.
A version of this article first appeared on Medscape.com.
In 1950, at Staten Island’s Sea View Hospital, a group of patients with terminal tuberculosis were given a new antibiotic called isoniazid, which caused some unexpected side effects. The patients reported euphoria, mental stimulation, and improved sleep, and even began socializing with more vigor. The press was all over the case, writing about the sick “dancing in the halls tho’ they had holes in their lungs.” Soon doctors started prescribing isoniazid as the first-ever antidepressant.
The Sea View Hospital experiment was an early hint that changing the composition of the gut microbiome – in this case, via antibiotics – might affect our mental health. Yet only in the last 2 decades has research into connections between what we ingest and psychiatric disorders really taken off. In 2004, a landmark study showed that germ-free mice (born in such sterile conditions that they lacked a microbiome) had an exaggerated stress response. The effects were reversed, however, if the mice were fed a bacterial strain, Bifidobacterium infantis, a probiotic. This sparked academic interest, and thousands of research papers followed.
According to Stephen Ilardi, PhD, a clinical psychologist at the University of Kansas, Lawrence, focusing on the etiology and treatment of depression, now is the “time of exciting discovery” in the field of probiotics and psychiatric disorders, although, admittedly, a lot still remains unknown.
Gut microbiome profiles in mental health disorders
We humans have about 100 trillion microbes residing in our guts. Some of these are archaea, some fungi, some protozoans and even viruses, but most are bacteria. Things like diet, sleep, and stress can all impact the composition of our gut microbiome. When the microbiome differs considerably from the typical, doctors and researchers describe it as dysbiosis, or imbalance. Studies have uncovered dysbiosis in patients with depression, anxiety, schizophrenia, and bipolar disorder.
“I think there is now pretty good evidence that the gut microbiome is actually an important factor in a number of psychiatric disorders,” says Allan Young, MBChB, clinical psychiatrist at King’s College London. The gut microbiome composition does seem to differ between psychiatric patients and the healthy. In depression, for example, a recent review of nine studies found an increase on the genus level in Streptococcus and Oscillibacter and low abundance of Lactobacillus and Coprococcus, among others. In generalized anxiety disorder, meanwhile, there appears to be an increase in Fusobacteria and Escherichia/Shigella .
For Dr. Ilardi, the next important question is whether there are plausible mechanisms that could explain how gut microbiota may influence brain function. And, it appears there are.
“The microbes in the gut can release neurotransmitters into blood that cross into the brain and influence brain function. They can release hormones into the blood that again cross into the brain. They’ve got a lot of tricks up their sleeve,” he says.
One particularly important pathway runs through the vagus nerve – the longest nerve that emerges directly from the brain, connecting it to the gut. Another is the immune pathway. Gut bacteria can interact with immune cells and reduce cytokine production, which in turn can reduce systemic inflammation. Inflammatory processes have been implicated in both depression and bipolar disorder. What’s more, gut microbes can upregulate the expression of a protein called BDNF – brain-derived neurotrophic factor – which helps the development and survival of nerve cells in the brain.
Probiotics’ promise varies for different conditions
As the pathways by which gut dysbiosis may influence psychiatric disorders become clearer, the next logical step is to try to influence the composition of the microbiome to prevent and treat depression, anxiety, or schizophrenia. That’s where probiotics come in.
The evidence for the effects of probiotics – live microorganisms which, when ingested in adequate amounts, confer a health benefit – so far is the strongest for depression, says Viktoriya Nikolova, MRes, MSc, a PhD student and researcher at King’s College London. In their 2021 meta-analysis of seven trials, Mr. Nikolova and colleagues revealed that probiotics can significantly reduce depressive symptoms after just 8 weeks. There was a caveat, however – the probiotics only worked when used in addition to an approved antidepressant. Another meta-analysis, published in 2018, also showed that probiotics, when compared with placebo, improve mood in people with depressive symptoms (here, no antidepressant treatment was necessary).
Roumen Milev, MD, PhD, a neuroscientist at Queen’s University, Kingston, Ont., and coauthor of a review on probiotics and depression published in the Annals of General Psychiatry, warns, however, that the research is still in its infancy. “,” he says.
When it comes to using probiotics to relieve anxiety, “the evidence in the animal literature is really compelling,” says Dr. Ilardi. Human studies are less convincing, however, which Dr. Dr. Ilardi showed in his 2018 review and meta-analysis involving 743 animals and 1,527 humans. “Studies are small for the most part, and some of them aren’t terribly well conducted, and they often use very low doses of probiotics,” he says. One of the larger double-blind and placebo-controlled trials showed that supplementation with Lactobacillus plantarum helps reduce stress and anxiety, while the levels of proinflammatory cytokines go down. Another meta-analysis, published in June, revealed that, when it comes to reducing stress and anxiety in youth, the results are mixed.
Evidence of probiotics’ efficiency in schizophrenia is emerging, yet also limited. A 2019 review concluded that currently available results only “hint” at a possibility that probiotics could make a difference in schizophrenia. Similarly, a 2020 review summed up that the role of probiotics in bipolar disorder “remains unclear and underexplored.”
Better studies, remaining questions
Apart from small samples, one issue with research on probiotics is that they generally tend to use varied doses of different strains of bacteria, or even multistrain mixtures, making it tough to compare results. Although there are hundreds of species of bacteria in the human gut, only a few have been evaluated for their antidepressant or antianxiety effects.
“To make it even worse, it’s almost certainly the case that depending on a person’s actual genetics or maybe their epigenetics, a strain that is helpful for one person may not be helpful for another. There is almost certainly no one-size-fits-all probiotic formulation,” says Dr. Ilardi.
Another critical question that remains to be answered is that of potential side effects.
“Probiotics are often seen as food supplements, so they don’t follow under the same regulations as drugs would,” says Mr. Nikolova. “They don’t necessarily have to follow the pattern of drug trials in many countries, which means that the monitoring of side effects is not the requirement.”
That’s something that worries King’s College psychiatrist Young too. “If you are giving it to modulate how the brain works, you could potentially induce psychiatric symptoms or a psychiatric disorder. There could be allergic reactions. There could be lots of different things,” he says.
When you search the web for “probiotics,” chances are you will come across sites boasting amazing effects that such products can have on cardiovascular heath, the immune system, and yes, mental well-being. Many also sell various probiotic supplements “formulated” for your gut health or improved moods. However, many such commercially available strains have never been actually tested in clinical trials. What’s more, according to Kathrin Cohen Kadosh, PhD, a neuroscientist at University of Surrey (England), “it is not always clear whether the different strains actually reach the gut intact.”
For now, considering the limited research evidence, a safer bet is to try to improve gut health through consumption of fermented foods that naturally contain probiotics, such as miso, kefir, or sauerkraut. Alternatively, you could reach for prebiotics, such as foods containing fiber (prebiotics enhance the growth of beneficial gut microbes). This, Dr. Kadosh says, could be “a gentler way of improving gut health” than popping a pill. Whether an improved mental well-being might follow still remains to be seen.
A version of this article first appeared on Medscape.com.
Antipsychotics tied to increased breast cancer risk
Use of antipsychotics that increase prolactin levels is significantly associated with an increased risk for breast cancer in women with schizophrenia, new research suggests. However, at least one expert says that, at this point, clinical implications are premature.
Investigators compared data from Finnish nationwide registers on more than 30,000 women diagnosed with schizophrenia. Of those patients, 1,069 were diagnosed with breast cancer. Results showed that long-term exposure to prolactin-increasing antipsychotics was associated with a 56% increased risk of developing breast cancer in comparison with exposure of short duration. No significant association was found with cumulative exposure to prolactin-sparing antipsychotics.
“In case of planning for long-term antipsychotic [therapy], prefer non–prolactin-raising antipsychotics in females and inform patients about a potential risk to allow for informed shared decision-making,” study coauthor Christoph U. Correll, MD, professor of psychiatry and molecular medicine at Hofstra University, Hempstead, N.Y., told this news organization.
“ he said.
The study was published online Aug. 30, 2021, in The Lancet.
A ‘relevant contribution’
Breast cancer is 25% more prevalent among women with schizophrenia than among women in the general population. Antipsychotics have long been suspected as a potential culprit, but research results have been inconsistent, said Dr. Correll.
In addition, high concentrations of prolactin are associated with a higher risk of developing breast cancer, but most previous research did not distinguish between antipsychotics that increased prolactin levels those that did not.
Dr. Correll and colleagues “wanted to add to this literature by utilizing a generalizable nationwide sample with a sufficient large number of patients and sufficiently long follow-up to address the clinically very relevant question whether antipsychotic use could increase the risk of breast cancer.”
They also believed that grouping antipsychotics into prolactin-raising and non–prolactin-raising agents would be “a relevant contribution.”
The researchers drew on data from several large Finnish databases to conduct a nested case-control study of 30,785 women aged at least16 years who were diagnosed with schizophrenia between 1972 and 2014.
Of these patients, 1,069 received an initial diagnosis of invasive breast cancer (after being diagnosed with schizophrenia) between 2000 and 2017. These case patients were compared to 5,339 matched control patients. The mean age of the case patients and the control patients was 62 years. The mean time since initial diagnosis of schizophrenia was 24 years.
Antipsychotic use was divided into three periods: less than 1 year, 1-4 years, and ≥5 years. Antipsychotics were further divided into prolactin-increasing or prolactin-sparing drugs (for example, clozapine, quetiapine, or aripiprazole). Breast cancer was divided into either lobular or ductal adenocarcinoma.
In their statistical analyses, the researchers adjusted for an array of covariates, including previous diagnoses of other medical conditions, drugs that may modify the risk for breast cancer (for example, beta-blockers, calcium channel blockers, spironolactone, loop diuretics, and statins), substance misuse, suicide attempt, parity, and use of hormone replacement therapy (HRT).
‘Clinically meaningful’ risk
Ductal adenocarcinoma was more common than lobular adenocarcinoma (73% vs. 20% among case patients). A higher proportion of case patients used cardiovascular medications and HRT, compared with control patients.
A higher proportion of case patients had used prolactin-increasing antipsychotics for at least 5 years, compared with control patients (71.4% vs. 64.3%; adjusted odds ratio, 1.56; 95% CI, 1.27-1.92; P < .0001) in comparison with minimal exposure (<1 year) to prolactin-increasing antipsychotics.
On the other hand, a similar proportion of case patients and control patients used prolactin-sparing antipsychotics for at least 5 years (8.3 vs. 8.2%; aOR, 1.19; 95% CI, 0.90-1.58); the OR of 1.19 was not deemed significant.
Although exposure of ≥5 years to prolactin-increasing antipsychotics was associated with an increased risk for both types of adenocarcinoma, the risk was higher for lobular than for ductal disease (aOR, 2.36; 95% CI, 1.46-3.82 vs. aOR, 1.42; 95% CI, 1.12-1.80).
“Conservatively, if we subtract the 19% nonsignificantly increased odds with prolactin-sparing antipsychotics from the 56% significantly increased odds with prolactin-increasing antipsychotics, we obtain a 37% relative increase in odds,” the authors noted.
“Using a lifetime incidence of breast cancer in women in the general population of about 12%, with a somewhat higher lifetime incidence in patients with schizophrenia than the general population, this difference between prolactin-increasing versus prolactin-sparing antipsychotics in breast cancer risk upon exposure of 5 or more years would correspond to about a 4% (37% x 12%) increase in absolute breast cancer odds with prolactin-increasing antipsychotic treatment” – a difference the authors call “clinically meaningful.”
Correll noted that although the study was conducted in a Finnish population, the findings are generalizable to other populations.
Clinical implications premature?
Commenting on the study, Anton Pottegård, MScPharm, PhD, DMSc, professor of pharmacoepidemiology, department of public health, University of Southern Denmark, Odense, expressed concern that “this new study is fairly aggressive in its recommendation [that] we need to pay attention to hyperprolactinemia, as this seems to cause breast cancer.”
Dr. Pottegård, who is also the head of research, Hospital Pharmacy Funen, Odense University Hospital, who was not involved with the study, said he does not “think that the full body of the literature supports such a direct conclusion and/or direct inference to clinical practice.”
Although “this is an important study to further this work, I do not think we are at a place (yet) where it should lead to different action from clinicians,” Dr. Pottegård cautioned.
Also commenting on the study, Mary Seeman, MDCM, DSc, professor emeritus of neurosciences and clinical translation, department of psychiatry, University of Toronto, called the question of whether prolactin-increasing antipsychotics increase breast cancer risk “very complicated because the incidence of breast cancer ... is higher in women with schizophrenia than in other women.”
Dr. Seeman, who was not involved with the study, pointed to other reasons for the increased risk, including higher rates of obesity, substance abuse, cigarette smoking, stress, and sedentary behavior, all of which raise prolactin levels. Additionally, “protective factors such as pregnancies and breastfeeding are less frequent in women with schizophrenia than in their peers.” Women with schizophrenia also “tend not to do breast screening, see their doctors less often, follow doctors’ orders less rigorously, and obtain treatment less often.”
The take-home message “is to prescribe prolactin-sparing medication to women if at all possible – but until we know more, that is good advice, although not always possible because the illness for which the antipsychotics are prescribed may not respond to those particular medications,” Dr. Seeman said.
The study was funded by the Finnish Ministry of Social Affairs and Health through the developmental fund for Niuvanniemi Hospital. Funding was also provided to individual researchers by the Academy of Finland, the Finnish Medical Foundation, and the Emil Aaltonen foundation. Dr. Correll has been a consultant or advisor to or has received honoraria from numerous companies. He has provided expert testimony for Janssen and Otsuka; received royalties from UpToDate and is a stock option holder of LB Pharma; served on a data safety monitoring board for Lundbeck, Rovi, Supernus, and Teva; and received grant support from Janssen and Takeda. Dr. Pottegård and Dr. Seeman disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Use of antipsychotics that increase prolactin levels is significantly associated with an increased risk for breast cancer in women with schizophrenia, new research suggests. However, at least one expert says that, at this point, clinical implications are premature.
Investigators compared data from Finnish nationwide registers on more than 30,000 women diagnosed with schizophrenia. Of those patients, 1,069 were diagnosed with breast cancer. Results showed that long-term exposure to prolactin-increasing antipsychotics was associated with a 56% increased risk of developing breast cancer in comparison with exposure of short duration. No significant association was found with cumulative exposure to prolactin-sparing antipsychotics.
“In case of planning for long-term antipsychotic [therapy], prefer non–prolactin-raising antipsychotics in females and inform patients about a potential risk to allow for informed shared decision-making,” study coauthor Christoph U. Correll, MD, professor of psychiatry and molecular medicine at Hofstra University, Hempstead, N.Y., told this news organization.
“ he said.
The study was published online Aug. 30, 2021, in The Lancet.
A ‘relevant contribution’
Breast cancer is 25% more prevalent among women with schizophrenia than among women in the general population. Antipsychotics have long been suspected as a potential culprit, but research results have been inconsistent, said Dr. Correll.
In addition, high concentrations of prolactin are associated with a higher risk of developing breast cancer, but most previous research did not distinguish between antipsychotics that increased prolactin levels those that did not.
Dr. Correll and colleagues “wanted to add to this literature by utilizing a generalizable nationwide sample with a sufficient large number of patients and sufficiently long follow-up to address the clinically very relevant question whether antipsychotic use could increase the risk of breast cancer.”
They also believed that grouping antipsychotics into prolactin-raising and non–prolactin-raising agents would be “a relevant contribution.”
The researchers drew on data from several large Finnish databases to conduct a nested case-control study of 30,785 women aged at least16 years who were diagnosed with schizophrenia between 1972 and 2014.
Of these patients, 1,069 received an initial diagnosis of invasive breast cancer (after being diagnosed with schizophrenia) between 2000 and 2017. These case patients were compared to 5,339 matched control patients. The mean age of the case patients and the control patients was 62 years. The mean time since initial diagnosis of schizophrenia was 24 years.
Antipsychotic use was divided into three periods: less than 1 year, 1-4 years, and ≥5 years. Antipsychotics were further divided into prolactin-increasing or prolactin-sparing drugs (for example, clozapine, quetiapine, or aripiprazole). Breast cancer was divided into either lobular or ductal adenocarcinoma.
In their statistical analyses, the researchers adjusted for an array of covariates, including previous diagnoses of other medical conditions, drugs that may modify the risk for breast cancer (for example, beta-blockers, calcium channel blockers, spironolactone, loop diuretics, and statins), substance misuse, suicide attempt, parity, and use of hormone replacement therapy (HRT).
‘Clinically meaningful’ risk
Ductal adenocarcinoma was more common than lobular adenocarcinoma (73% vs. 20% among case patients). A higher proportion of case patients used cardiovascular medications and HRT, compared with control patients.
A higher proportion of case patients had used prolactin-increasing antipsychotics for at least 5 years, compared with control patients (71.4% vs. 64.3%; adjusted odds ratio, 1.56; 95% CI, 1.27-1.92; P < .0001) in comparison with minimal exposure (<1 year) to prolactin-increasing antipsychotics.
On the other hand, a similar proportion of case patients and control patients used prolactin-sparing antipsychotics for at least 5 years (8.3 vs. 8.2%; aOR, 1.19; 95% CI, 0.90-1.58); the OR of 1.19 was not deemed significant.
Although exposure of ≥5 years to prolactin-increasing antipsychotics was associated with an increased risk for both types of adenocarcinoma, the risk was higher for lobular than for ductal disease (aOR, 2.36; 95% CI, 1.46-3.82 vs. aOR, 1.42; 95% CI, 1.12-1.80).
“Conservatively, if we subtract the 19% nonsignificantly increased odds with prolactin-sparing antipsychotics from the 56% significantly increased odds with prolactin-increasing antipsychotics, we obtain a 37% relative increase in odds,” the authors noted.
“Using a lifetime incidence of breast cancer in women in the general population of about 12%, with a somewhat higher lifetime incidence in patients with schizophrenia than the general population, this difference between prolactin-increasing versus prolactin-sparing antipsychotics in breast cancer risk upon exposure of 5 or more years would correspond to about a 4% (37% x 12%) increase in absolute breast cancer odds with prolactin-increasing antipsychotic treatment” – a difference the authors call “clinically meaningful.”
Correll noted that although the study was conducted in a Finnish population, the findings are generalizable to other populations.
Clinical implications premature?
Commenting on the study, Anton Pottegård, MScPharm, PhD, DMSc, professor of pharmacoepidemiology, department of public health, University of Southern Denmark, Odense, expressed concern that “this new study is fairly aggressive in its recommendation [that] we need to pay attention to hyperprolactinemia, as this seems to cause breast cancer.”
Dr. Pottegård, who is also the head of research, Hospital Pharmacy Funen, Odense University Hospital, who was not involved with the study, said he does not “think that the full body of the literature supports such a direct conclusion and/or direct inference to clinical practice.”
Although “this is an important study to further this work, I do not think we are at a place (yet) where it should lead to different action from clinicians,” Dr. Pottegård cautioned.
Also commenting on the study, Mary Seeman, MDCM, DSc, professor emeritus of neurosciences and clinical translation, department of psychiatry, University of Toronto, called the question of whether prolactin-increasing antipsychotics increase breast cancer risk “very complicated because the incidence of breast cancer ... is higher in women with schizophrenia than in other women.”
Dr. Seeman, who was not involved with the study, pointed to other reasons for the increased risk, including higher rates of obesity, substance abuse, cigarette smoking, stress, and sedentary behavior, all of which raise prolactin levels. Additionally, “protective factors such as pregnancies and breastfeeding are less frequent in women with schizophrenia than in their peers.” Women with schizophrenia also “tend not to do breast screening, see their doctors less often, follow doctors’ orders less rigorously, and obtain treatment less often.”
The take-home message “is to prescribe prolactin-sparing medication to women if at all possible – but until we know more, that is good advice, although not always possible because the illness for which the antipsychotics are prescribed may not respond to those particular medications,” Dr. Seeman said.
The study was funded by the Finnish Ministry of Social Affairs and Health through the developmental fund for Niuvanniemi Hospital. Funding was also provided to individual researchers by the Academy of Finland, the Finnish Medical Foundation, and the Emil Aaltonen foundation. Dr. Correll has been a consultant or advisor to or has received honoraria from numerous companies. He has provided expert testimony for Janssen and Otsuka; received royalties from UpToDate and is a stock option holder of LB Pharma; served on a data safety monitoring board for Lundbeck, Rovi, Supernus, and Teva; and received grant support from Janssen and Takeda. Dr. Pottegård and Dr. Seeman disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Use of antipsychotics that increase prolactin levels is significantly associated with an increased risk for breast cancer in women with schizophrenia, new research suggests. However, at least one expert says that, at this point, clinical implications are premature.
Investigators compared data from Finnish nationwide registers on more than 30,000 women diagnosed with schizophrenia. Of those patients, 1,069 were diagnosed with breast cancer. Results showed that long-term exposure to prolactin-increasing antipsychotics was associated with a 56% increased risk of developing breast cancer in comparison with exposure of short duration. No significant association was found with cumulative exposure to prolactin-sparing antipsychotics.
“In case of planning for long-term antipsychotic [therapy], prefer non–prolactin-raising antipsychotics in females and inform patients about a potential risk to allow for informed shared decision-making,” study coauthor Christoph U. Correll, MD, professor of psychiatry and molecular medicine at Hofstra University, Hempstead, N.Y., told this news organization.
“ he said.
The study was published online Aug. 30, 2021, in The Lancet.
A ‘relevant contribution’
Breast cancer is 25% more prevalent among women with schizophrenia than among women in the general population. Antipsychotics have long been suspected as a potential culprit, but research results have been inconsistent, said Dr. Correll.
In addition, high concentrations of prolactin are associated with a higher risk of developing breast cancer, but most previous research did not distinguish between antipsychotics that increased prolactin levels those that did not.
Dr. Correll and colleagues “wanted to add to this literature by utilizing a generalizable nationwide sample with a sufficient large number of patients and sufficiently long follow-up to address the clinically very relevant question whether antipsychotic use could increase the risk of breast cancer.”
They also believed that grouping antipsychotics into prolactin-raising and non–prolactin-raising agents would be “a relevant contribution.”
The researchers drew on data from several large Finnish databases to conduct a nested case-control study of 30,785 women aged at least16 years who were diagnosed with schizophrenia between 1972 and 2014.
Of these patients, 1,069 received an initial diagnosis of invasive breast cancer (after being diagnosed with schizophrenia) between 2000 and 2017. These case patients were compared to 5,339 matched control patients. The mean age of the case patients and the control patients was 62 years. The mean time since initial diagnosis of schizophrenia was 24 years.
Antipsychotic use was divided into three periods: less than 1 year, 1-4 years, and ≥5 years. Antipsychotics were further divided into prolactin-increasing or prolactin-sparing drugs (for example, clozapine, quetiapine, or aripiprazole). Breast cancer was divided into either lobular or ductal adenocarcinoma.
In their statistical analyses, the researchers adjusted for an array of covariates, including previous diagnoses of other medical conditions, drugs that may modify the risk for breast cancer (for example, beta-blockers, calcium channel blockers, spironolactone, loop diuretics, and statins), substance misuse, suicide attempt, parity, and use of hormone replacement therapy (HRT).
‘Clinically meaningful’ risk
Ductal adenocarcinoma was more common than lobular adenocarcinoma (73% vs. 20% among case patients). A higher proportion of case patients used cardiovascular medications and HRT, compared with control patients.
A higher proportion of case patients had used prolactin-increasing antipsychotics for at least 5 years, compared with control patients (71.4% vs. 64.3%; adjusted odds ratio, 1.56; 95% CI, 1.27-1.92; P < .0001) in comparison with minimal exposure (<1 year) to prolactin-increasing antipsychotics.
On the other hand, a similar proportion of case patients and control patients used prolactin-sparing antipsychotics for at least 5 years (8.3 vs. 8.2%; aOR, 1.19; 95% CI, 0.90-1.58); the OR of 1.19 was not deemed significant.
Although exposure of ≥5 years to prolactin-increasing antipsychotics was associated with an increased risk for both types of adenocarcinoma, the risk was higher for lobular than for ductal disease (aOR, 2.36; 95% CI, 1.46-3.82 vs. aOR, 1.42; 95% CI, 1.12-1.80).
“Conservatively, if we subtract the 19% nonsignificantly increased odds with prolactin-sparing antipsychotics from the 56% significantly increased odds with prolactin-increasing antipsychotics, we obtain a 37% relative increase in odds,” the authors noted.
“Using a lifetime incidence of breast cancer in women in the general population of about 12%, with a somewhat higher lifetime incidence in patients with schizophrenia than the general population, this difference between prolactin-increasing versus prolactin-sparing antipsychotics in breast cancer risk upon exposure of 5 or more years would correspond to about a 4% (37% x 12%) increase in absolute breast cancer odds with prolactin-increasing antipsychotic treatment” – a difference the authors call “clinically meaningful.”
Correll noted that although the study was conducted in a Finnish population, the findings are generalizable to other populations.
Clinical implications premature?
Commenting on the study, Anton Pottegård, MScPharm, PhD, DMSc, professor of pharmacoepidemiology, department of public health, University of Southern Denmark, Odense, expressed concern that “this new study is fairly aggressive in its recommendation [that] we need to pay attention to hyperprolactinemia, as this seems to cause breast cancer.”
Dr. Pottegård, who is also the head of research, Hospital Pharmacy Funen, Odense University Hospital, who was not involved with the study, said he does not “think that the full body of the literature supports such a direct conclusion and/or direct inference to clinical practice.”
Although “this is an important study to further this work, I do not think we are at a place (yet) where it should lead to different action from clinicians,” Dr. Pottegård cautioned.
Also commenting on the study, Mary Seeman, MDCM, DSc, professor emeritus of neurosciences and clinical translation, department of psychiatry, University of Toronto, called the question of whether prolactin-increasing antipsychotics increase breast cancer risk “very complicated because the incidence of breast cancer ... is higher in women with schizophrenia than in other women.”
Dr. Seeman, who was not involved with the study, pointed to other reasons for the increased risk, including higher rates of obesity, substance abuse, cigarette smoking, stress, and sedentary behavior, all of which raise prolactin levels. Additionally, “protective factors such as pregnancies and breastfeeding are less frequent in women with schizophrenia than in their peers.” Women with schizophrenia also “tend not to do breast screening, see their doctors less often, follow doctors’ orders less rigorously, and obtain treatment less often.”
The take-home message “is to prescribe prolactin-sparing medication to women if at all possible – but until we know more, that is good advice, although not always possible because the illness for which the antipsychotics are prescribed may not respond to those particular medications,” Dr. Seeman said.
The study was funded by the Finnish Ministry of Social Affairs and Health through the developmental fund for Niuvanniemi Hospital. Funding was also provided to individual researchers by the Academy of Finland, the Finnish Medical Foundation, and the Emil Aaltonen foundation. Dr. Correll has been a consultant or advisor to or has received honoraria from numerous companies. He has provided expert testimony for Janssen and Otsuka; received royalties from UpToDate and is a stock option holder of LB Pharma; served on a data safety monitoring board for Lundbeck, Rovi, Supernus, and Teva; and received grant support from Janssen and Takeda. Dr. Pottegård and Dr. Seeman disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Optimal antipsychotic dose for schizophrenia relapse identified
A middle-of-the-road dose of an antipsychotic appears to be optimal for relapse prevention in stable schizophrenia, new research suggests.
Results of a meta-analysis show a 5-mg/day equivalent risperidone dose worked best. Higher doses were associated with more adverse events without showing substantial gains in relapse prevention, and lower doses were associated with greater relapse risk.
“The safest approach is to just to carry on with 5 mg,” which in many cases represents a full dose, lead author Stefan Leucht, MD, professor, department of psychiatry and psychotherapy, Technical University of Munich School of Medicine, Germany, told this news organization.
However, he added, patient preferences and other factors should be considered in dosage decision-making.
The findings were published online August 18 in JAMA Psychiatry.
Unique meta-analysis
Antipsychotic drugs are effective for short-term treatment of schizophrenia and prevention of relapse but are associated with movement disorders, weight gain, and other metabolic changes. They are also associated with even more severe adverse events, including tardive dyskinesia and increased cardiovascular risk.
For years, researchers have tried to find the optimal dose of antipsychotic drugs to prevent relapse in patients with stable schizophrenia while mitigating adverse event risk.
For the meta-analysis, researchers searched for fixed-dose, randomized, blinded, or open trials that lasted longer than 3 months and compared two first-generation antipsychotics – haloperidol or fluphenazine – or a second-generation antipsychotic with placebo or a different dose of the same drug.
The analysis included 26 studies with 72 individual dose arms and 4,776 participants with stable schizophrenia.
Researchers used a dose-response meta-analysis. Unlike a simple meta-analysis that provides an “arbitrary” cut-off of superiority of one drug over placebo or another drug, a dose-response meta-analysis gives a plot or curve “that shows how this evolves with different doses,” Dr. Leucht noted.
The investigators estimated dose-response curves for each antipsychotic drug compared with placebo separately and as a group.
They did not have enough data for most of the single antipsychotics, so they converted doses to risperidone equivalents for a pooled analysis across drugs. They chose risperidone because its equivalents “are pretty well-defined,” said Dr. Leucht.
Go slow to go low
For the primary outcome of relapse, the dose-response curve showed a hyperbolic shape with a clear plateau. Initially, the plot decreased sharply but then flattened at about 5-mg/day risperidone equivalent (odds ratio, 0.20; 95% confidence interval, 0.13-0.31; relative risk, 0.43; 95% CI, 0.31-0.57).
“We were a little disappointed because we hoped that a dose lower than 5 mg would be most efficacious in terms of relapse rate because this would have reduced the side-effect burden,” Dr. Leucht said.
Nevertheless, he emphasized that doses lower than 5 mg/day risperidone equivalent are not completely ineffective. For example, the 2.5-mg dose reduced risk to relapse in relative terms by about 40% (RR, 0.63).
Dr. Leucht also pointed out there is “huge interindividual variability.” Therefore, “It just means for the average patient it’s safest, let’s say, to keep her or him on 5 mg,” he said.
When lowering the dose, Dr. Leucht noted clinicians should “be very careful and to do it very slowly. It should be very small reductions every 3 to 6 months.”
For the secondary endpoint of rehospitalizations, the shape of the curve was similar to the one for relapse but with lower rates.
“If patients need to be rehospitalized, it usually means that the relapse was major and not only a minor increase in symptoms,” said Dr. Leucht.
The curves for all-cause discontinuation and reduction in overall symptoms were also similar to that of relapse.
However, the curve for dropouts because of adverse events showed that higher doses led to more adverse events. For example, with 5-mg/day dose, the OR was 1.4 (95% CI, 0.87-2.25) and the RR was 1.38 (95% CI, 0.87-2.15), but for the 15-mg/day dose, the OR was 2.88 (95% CI, 1.52-5.45) and the RR was 2.68 (95% CI, 1.49-4.62).
Patient preference key
The data were insufficient to assess differences between men and women or between older and younger patients, Dr. Leucht noted.
However, post-hoc subgroup analyses turned up some interesting findings, he added. For example, patients who take high-potency first-generation antipsychotics such as haloperidol might do well on a lower dose, said Dr. Leucht.
“They may need a dose even lower than 5 mg, perhaps something like 2.5 mg, because these drugs bind so strongly to dopamine receptors,” he said.
He reiterated that patient preferences should always be considered when deciding on antipsychotic dosage.
“Many patients will say they don’t want to relapse anymore, but others will say these drugs have horrible side effects, and they want to go on a lower dose,” said Dr. Leucht.
Clinicians should also factor in patient characteristics, such as comorbidities or substance abuse, as well as severity of past relapses and properties of individual drugs, he added.
Reflects real-world experience
Commenting on the findings, Thomas Sedlak, MD, PhD, director, Schizophrenia and Psychosis Consult Clinic and assistant professor of psychiatry and behavioral sciences, Johns Hopkins School of Medicine, Baltimore, said the research “is a fine addition” to a previous analysis that explored dose-response relationships of antipsychotic drugs in the acute phase.
Crunching all the data from studies that have different types of patients and extracting a single dosage that provides maximum benefit is “a great challenge,” said Dr. Sedlak, who was not involved with the research.
The fact that most patients won’t get additional benefit above 5 mg, at which point they start getting more adverse events, and that 2.5 mg is sufficient for certain subgroups “agrees well with the experience of many who use these medications regularly,” Dr. Sedlak said.
However, he cautioned that psychiatrists “don’t always intuitively know which patients fall into which dose category or who might require clozapine.”
“Clinicians need to be mindful that it’s easy to overshoot an optimal dose and elicit side effects,” said Dr. Sedlak.
He also noted that severely ill patients are often underrepresented in clinical trials because they are too impaired to participate, “so they may have a different optimal dosage,” he concluded.
Dr. Leucht has reported receiving personal fees for consulting, advising, and/or speaking outside the submitted work from Angelini, Boehringer Ingelheim, Geodon & Richter, Janssen, Johnson & Johnson, Lundbeck, LTS Lohmann, MSD, Otsuka, Recordati, Sanofi Aventis, Sandoz, Sunovion, Teva, Eisai, Rovi, and Amiabel. Dr. Sedlak has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A middle-of-the-road dose of an antipsychotic appears to be optimal for relapse prevention in stable schizophrenia, new research suggests.
Results of a meta-analysis show a 5-mg/day equivalent risperidone dose worked best. Higher doses were associated with more adverse events without showing substantial gains in relapse prevention, and lower doses were associated with greater relapse risk.
“The safest approach is to just to carry on with 5 mg,” which in many cases represents a full dose, lead author Stefan Leucht, MD, professor, department of psychiatry and psychotherapy, Technical University of Munich School of Medicine, Germany, told this news organization.
However, he added, patient preferences and other factors should be considered in dosage decision-making.
The findings were published online August 18 in JAMA Psychiatry.
Unique meta-analysis
Antipsychotic drugs are effective for short-term treatment of schizophrenia and prevention of relapse but are associated with movement disorders, weight gain, and other metabolic changes. They are also associated with even more severe adverse events, including tardive dyskinesia and increased cardiovascular risk.
For years, researchers have tried to find the optimal dose of antipsychotic drugs to prevent relapse in patients with stable schizophrenia while mitigating adverse event risk.
For the meta-analysis, researchers searched for fixed-dose, randomized, blinded, or open trials that lasted longer than 3 months and compared two first-generation antipsychotics – haloperidol or fluphenazine – or a second-generation antipsychotic with placebo or a different dose of the same drug.
The analysis included 26 studies with 72 individual dose arms and 4,776 participants with stable schizophrenia.
Researchers used a dose-response meta-analysis. Unlike a simple meta-analysis that provides an “arbitrary” cut-off of superiority of one drug over placebo or another drug, a dose-response meta-analysis gives a plot or curve “that shows how this evolves with different doses,” Dr. Leucht noted.
The investigators estimated dose-response curves for each antipsychotic drug compared with placebo separately and as a group.
They did not have enough data for most of the single antipsychotics, so they converted doses to risperidone equivalents for a pooled analysis across drugs. They chose risperidone because its equivalents “are pretty well-defined,” said Dr. Leucht.
Go slow to go low
For the primary outcome of relapse, the dose-response curve showed a hyperbolic shape with a clear plateau. Initially, the plot decreased sharply but then flattened at about 5-mg/day risperidone equivalent (odds ratio, 0.20; 95% confidence interval, 0.13-0.31; relative risk, 0.43; 95% CI, 0.31-0.57).
“We were a little disappointed because we hoped that a dose lower than 5 mg would be most efficacious in terms of relapse rate because this would have reduced the side-effect burden,” Dr. Leucht said.
Nevertheless, he emphasized that doses lower than 5 mg/day risperidone equivalent are not completely ineffective. For example, the 2.5-mg dose reduced risk to relapse in relative terms by about 40% (RR, 0.63).
Dr. Leucht also pointed out there is “huge interindividual variability.” Therefore, “It just means for the average patient it’s safest, let’s say, to keep her or him on 5 mg,” he said.
When lowering the dose, Dr. Leucht noted clinicians should “be very careful and to do it very slowly. It should be very small reductions every 3 to 6 months.”
For the secondary endpoint of rehospitalizations, the shape of the curve was similar to the one for relapse but with lower rates.
“If patients need to be rehospitalized, it usually means that the relapse was major and not only a minor increase in symptoms,” said Dr. Leucht.
The curves for all-cause discontinuation and reduction in overall symptoms were also similar to that of relapse.
However, the curve for dropouts because of adverse events showed that higher doses led to more adverse events. For example, with 5-mg/day dose, the OR was 1.4 (95% CI, 0.87-2.25) and the RR was 1.38 (95% CI, 0.87-2.15), but for the 15-mg/day dose, the OR was 2.88 (95% CI, 1.52-5.45) and the RR was 2.68 (95% CI, 1.49-4.62).
Patient preference key
The data were insufficient to assess differences between men and women or between older and younger patients, Dr. Leucht noted.
However, post-hoc subgroup analyses turned up some interesting findings, he added. For example, patients who take high-potency first-generation antipsychotics such as haloperidol might do well on a lower dose, said Dr. Leucht.
“They may need a dose even lower than 5 mg, perhaps something like 2.5 mg, because these drugs bind so strongly to dopamine receptors,” he said.
He reiterated that patient preferences should always be considered when deciding on antipsychotic dosage.
“Many patients will say they don’t want to relapse anymore, but others will say these drugs have horrible side effects, and they want to go on a lower dose,” said Dr. Leucht.
Clinicians should also factor in patient characteristics, such as comorbidities or substance abuse, as well as severity of past relapses and properties of individual drugs, he added.
Reflects real-world experience
Commenting on the findings, Thomas Sedlak, MD, PhD, director, Schizophrenia and Psychosis Consult Clinic and assistant professor of psychiatry and behavioral sciences, Johns Hopkins School of Medicine, Baltimore, said the research “is a fine addition” to a previous analysis that explored dose-response relationships of antipsychotic drugs in the acute phase.
Crunching all the data from studies that have different types of patients and extracting a single dosage that provides maximum benefit is “a great challenge,” said Dr. Sedlak, who was not involved with the research.
The fact that most patients won’t get additional benefit above 5 mg, at which point they start getting more adverse events, and that 2.5 mg is sufficient for certain subgroups “agrees well with the experience of many who use these medications regularly,” Dr. Sedlak said.
However, he cautioned that psychiatrists “don’t always intuitively know which patients fall into which dose category or who might require clozapine.”
“Clinicians need to be mindful that it’s easy to overshoot an optimal dose and elicit side effects,” said Dr. Sedlak.
He also noted that severely ill patients are often underrepresented in clinical trials because they are too impaired to participate, “so they may have a different optimal dosage,” he concluded.
Dr. Leucht has reported receiving personal fees for consulting, advising, and/or speaking outside the submitted work from Angelini, Boehringer Ingelheim, Geodon & Richter, Janssen, Johnson & Johnson, Lundbeck, LTS Lohmann, MSD, Otsuka, Recordati, Sanofi Aventis, Sandoz, Sunovion, Teva, Eisai, Rovi, and Amiabel. Dr. Sedlak has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A middle-of-the-road dose of an antipsychotic appears to be optimal for relapse prevention in stable schizophrenia, new research suggests.
Results of a meta-analysis show a 5-mg/day equivalent risperidone dose worked best. Higher doses were associated with more adverse events without showing substantial gains in relapse prevention, and lower doses were associated with greater relapse risk.
“The safest approach is to just to carry on with 5 mg,” which in many cases represents a full dose, lead author Stefan Leucht, MD, professor, department of psychiatry and psychotherapy, Technical University of Munich School of Medicine, Germany, told this news organization.
However, he added, patient preferences and other factors should be considered in dosage decision-making.
The findings were published online August 18 in JAMA Psychiatry.
Unique meta-analysis
Antipsychotic drugs are effective for short-term treatment of schizophrenia and prevention of relapse but are associated with movement disorders, weight gain, and other metabolic changes. They are also associated with even more severe adverse events, including tardive dyskinesia and increased cardiovascular risk.
For years, researchers have tried to find the optimal dose of antipsychotic drugs to prevent relapse in patients with stable schizophrenia while mitigating adverse event risk.
For the meta-analysis, researchers searched for fixed-dose, randomized, blinded, or open trials that lasted longer than 3 months and compared two first-generation antipsychotics – haloperidol or fluphenazine – or a second-generation antipsychotic with placebo or a different dose of the same drug.
The analysis included 26 studies with 72 individual dose arms and 4,776 participants with stable schizophrenia.
Researchers used a dose-response meta-analysis. Unlike a simple meta-analysis that provides an “arbitrary” cut-off of superiority of one drug over placebo or another drug, a dose-response meta-analysis gives a plot or curve “that shows how this evolves with different doses,” Dr. Leucht noted.
The investigators estimated dose-response curves for each antipsychotic drug compared with placebo separately and as a group.
They did not have enough data for most of the single antipsychotics, so they converted doses to risperidone equivalents for a pooled analysis across drugs. They chose risperidone because its equivalents “are pretty well-defined,” said Dr. Leucht.
Go slow to go low
For the primary outcome of relapse, the dose-response curve showed a hyperbolic shape with a clear plateau. Initially, the plot decreased sharply but then flattened at about 5-mg/day risperidone equivalent (odds ratio, 0.20; 95% confidence interval, 0.13-0.31; relative risk, 0.43; 95% CI, 0.31-0.57).
“We were a little disappointed because we hoped that a dose lower than 5 mg would be most efficacious in terms of relapse rate because this would have reduced the side-effect burden,” Dr. Leucht said.
Nevertheless, he emphasized that doses lower than 5 mg/day risperidone equivalent are not completely ineffective. For example, the 2.5-mg dose reduced risk to relapse in relative terms by about 40% (RR, 0.63).
Dr. Leucht also pointed out there is “huge interindividual variability.” Therefore, “It just means for the average patient it’s safest, let’s say, to keep her or him on 5 mg,” he said.
When lowering the dose, Dr. Leucht noted clinicians should “be very careful and to do it very slowly. It should be very small reductions every 3 to 6 months.”
For the secondary endpoint of rehospitalizations, the shape of the curve was similar to the one for relapse but with lower rates.
“If patients need to be rehospitalized, it usually means that the relapse was major and not only a minor increase in symptoms,” said Dr. Leucht.
The curves for all-cause discontinuation and reduction in overall symptoms were also similar to that of relapse.
However, the curve for dropouts because of adverse events showed that higher doses led to more adverse events. For example, with 5-mg/day dose, the OR was 1.4 (95% CI, 0.87-2.25) and the RR was 1.38 (95% CI, 0.87-2.15), but for the 15-mg/day dose, the OR was 2.88 (95% CI, 1.52-5.45) and the RR was 2.68 (95% CI, 1.49-4.62).
Patient preference key
The data were insufficient to assess differences between men and women or between older and younger patients, Dr. Leucht noted.
However, post-hoc subgroup analyses turned up some interesting findings, he added. For example, patients who take high-potency first-generation antipsychotics such as haloperidol might do well on a lower dose, said Dr. Leucht.
“They may need a dose even lower than 5 mg, perhaps something like 2.5 mg, because these drugs bind so strongly to dopamine receptors,” he said.
He reiterated that patient preferences should always be considered when deciding on antipsychotic dosage.
“Many patients will say they don’t want to relapse anymore, but others will say these drugs have horrible side effects, and they want to go on a lower dose,” said Dr. Leucht.
Clinicians should also factor in patient characteristics, such as comorbidities or substance abuse, as well as severity of past relapses and properties of individual drugs, he added.
Reflects real-world experience
Commenting on the findings, Thomas Sedlak, MD, PhD, director, Schizophrenia and Psychosis Consult Clinic and assistant professor of psychiatry and behavioral sciences, Johns Hopkins School of Medicine, Baltimore, said the research “is a fine addition” to a previous analysis that explored dose-response relationships of antipsychotic drugs in the acute phase.
Crunching all the data from studies that have different types of patients and extracting a single dosage that provides maximum benefit is “a great challenge,” said Dr. Sedlak, who was not involved with the research.
The fact that most patients won’t get additional benefit above 5 mg, at which point they start getting more adverse events, and that 2.5 mg is sufficient for certain subgroups “agrees well with the experience of many who use these medications regularly,” Dr. Sedlak said.
However, he cautioned that psychiatrists “don’t always intuitively know which patients fall into which dose category or who might require clozapine.”
“Clinicians need to be mindful that it’s easy to overshoot an optimal dose and elicit side effects,” said Dr. Sedlak.
He also noted that severely ill patients are often underrepresented in clinical trials because they are too impaired to participate, “so they may have a different optimal dosage,” he concluded.
Dr. Leucht has reported receiving personal fees for consulting, advising, and/or speaking outside the submitted work from Angelini, Boehringer Ingelheim, Geodon & Richter, Janssen, Johnson & Johnson, Lundbeck, LTS Lohmann, MSD, Otsuka, Recordati, Sanofi Aventis, Sandoz, Sunovion, Teva, Eisai, Rovi, and Amiabel. Dr. Sedlak has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FDA approves first twice-yearly antipsychotic for schizophrenia
The U.S. Food and Drug Administration has approved a 6-month injection form of the long-acting atypical antipsychotic paliperidone palmitate (Invega Hafyera, Janssen Pharmaceuticals) for the treatment of schizophrenia in adults, the company has announced.
This marks the “first-and-only twice-yearly injectable” approved for treating schizophrenia, the company added in a press release.
Before transitioning to the 6-month form, patients must be adequately treated for a minimum of 4 months with the company’s 1-month formulation of paliperidone (Invega Sustenna), or with the 3-month version (Invega Trinza) for at least one 3-month injection cycle.
The FDA approved the twice-yearly formulation on the basis of results from a 12-month, randomized, double-blind, phase 3 study that enrolled 702 adults with schizophrenia from 20 countries.
“The phase 3 trial results provide compelling evidence that 6-month paliperidone palmitate offers longer-term symptom control with the fewest doses per year, which may support greater patient adherence,” Gustavo Alva, MD, medical director at ATP Clinical Research, Costa Mesa, Calif., and 6-month paliperidone palmitate clinical trial investigator, said in the release.
Noninferiority results
In the phase 3 trial, the twice-yearly version of the drug proved noninferior to the 3-month version on the primary endpoint of time to first relapse at the end of 12 months, with 92.5% and 95% of patients, respectively, relapse-free at 12 months.
Relapse was defined as psychiatric hospitalization, increase in Positive and Negative Syndrome Scale (PANSS) total score, increase in individual PANSS item scores, self-injury, violent behavior, or suicidal/homicidal ideation.
The safety profile observed in the trial was in line with prior studies of the 1-month and 3-month versions, with no new safety signals, the researchers note.
The most common adverse reactions affecting at least 5% of participants in the clinical trial receiving twice-year paliperidone were upper respiratory tract infection (12%), injection site reaction (11%), weight gain (9%), headache (7%), and parkinsonism (5%).
Relapse is common in adults with schizophrenia, often because of missed doses of medication, the company said in the news release.
, while research continues to demonstrate that stronger medication adherence means better patient outcomes,” Dr. Alva said.
Recently updated evidence-based guidelines from the American Psychiatric Association recommend consideration of long-acting injectables for appropriate adults living with schizophrenia.
“Long-acting injectable treatments offer a number of advantages, compared to oral medication for schizophrenia, including relief from needing to remember to take medication daily, lower discontinuation rates, and sustained treatment over longer periods,” Bill Martin, PhD, with Janssen Research & Development, said in the release.
“Today’s approval enables us to rethink how we manage this chronic disease by offering patients and caregivers the potential for a life less defined by schizophrenia medication,” Dr. Martin added.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has approved a 6-month injection form of the long-acting atypical antipsychotic paliperidone palmitate (Invega Hafyera, Janssen Pharmaceuticals) for the treatment of schizophrenia in adults, the company has announced.
This marks the “first-and-only twice-yearly injectable” approved for treating schizophrenia, the company added in a press release.
Before transitioning to the 6-month form, patients must be adequately treated for a minimum of 4 months with the company’s 1-month formulation of paliperidone (Invega Sustenna), or with the 3-month version (Invega Trinza) for at least one 3-month injection cycle.
The FDA approved the twice-yearly formulation on the basis of results from a 12-month, randomized, double-blind, phase 3 study that enrolled 702 adults with schizophrenia from 20 countries.
“The phase 3 trial results provide compelling evidence that 6-month paliperidone palmitate offers longer-term symptom control with the fewest doses per year, which may support greater patient adherence,” Gustavo Alva, MD, medical director at ATP Clinical Research, Costa Mesa, Calif., and 6-month paliperidone palmitate clinical trial investigator, said in the release.
Noninferiority results
In the phase 3 trial, the twice-yearly version of the drug proved noninferior to the 3-month version on the primary endpoint of time to first relapse at the end of 12 months, with 92.5% and 95% of patients, respectively, relapse-free at 12 months.
Relapse was defined as psychiatric hospitalization, increase in Positive and Negative Syndrome Scale (PANSS) total score, increase in individual PANSS item scores, self-injury, violent behavior, or suicidal/homicidal ideation.
The safety profile observed in the trial was in line with prior studies of the 1-month and 3-month versions, with no new safety signals, the researchers note.
The most common adverse reactions affecting at least 5% of participants in the clinical trial receiving twice-year paliperidone were upper respiratory tract infection (12%), injection site reaction (11%), weight gain (9%), headache (7%), and parkinsonism (5%).
Relapse is common in adults with schizophrenia, often because of missed doses of medication, the company said in the news release.
, while research continues to demonstrate that stronger medication adherence means better patient outcomes,” Dr. Alva said.
Recently updated evidence-based guidelines from the American Psychiatric Association recommend consideration of long-acting injectables for appropriate adults living with schizophrenia.
“Long-acting injectable treatments offer a number of advantages, compared to oral medication for schizophrenia, including relief from needing to remember to take medication daily, lower discontinuation rates, and sustained treatment over longer periods,” Bill Martin, PhD, with Janssen Research & Development, said in the release.
“Today’s approval enables us to rethink how we manage this chronic disease by offering patients and caregivers the potential for a life less defined by schizophrenia medication,” Dr. Martin added.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has approved a 6-month injection form of the long-acting atypical antipsychotic paliperidone palmitate (Invega Hafyera, Janssen Pharmaceuticals) for the treatment of schizophrenia in adults, the company has announced.
This marks the “first-and-only twice-yearly injectable” approved for treating schizophrenia, the company added in a press release.
Before transitioning to the 6-month form, patients must be adequately treated for a minimum of 4 months with the company’s 1-month formulation of paliperidone (Invega Sustenna), or with the 3-month version (Invega Trinza) for at least one 3-month injection cycle.
The FDA approved the twice-yearly formulation on the basis of results from a 12-month, randomized, double-blind, phase 3 study that enrolled 702 adults with schizophrenia from 20 countries.
“The phase 3 trial results provide compelling evidence that 6-month paliperidone palmitate offers longer-term symptom control with the fewest doses per year, which may support greater patient adherence,” Gustavo Alva, MD, medical director at ATP Clinical Research, Costa Mesa, Calif., and 6-month paliperidone palmitate clinical trial investigator, said in the release.
Noninferiority results
In the phase 3 trial, the twice-yearly version of the drug proved noninferior to the 3-month version on the primary endpoint of time to first relapse at the end of 12 months, with 92.5% and 95% of patients, respectively, relapse-free at 12 months.
Relapse was defined as psychiatric hospitalization, increase in Positive and Negative Syndrome Scale (PANSS) total score, increase in individual PANSS item scores, self-injury, violent behavior, or suicidal/homicidal ideation.
The safety profile observed in the trial was in line with prior studies of the 1-month and 3-month versions, with no new safety signals, the researchers note.
The most common adverse reactions affecting at least 5% of participants in the clinical trial receiving twice-year paliperidone were upper respiratory tract infection (12%), injection site reaction (11%), weight gain (9%), headache (7%), and parkinsonism (5%).
Relapse is common in adults with schizophrenia, often because of missed doses of medication, the company said in the news release.
, while research continues to demonstrate that stronger medication adherence means better patient outcomes,” Dr. Alva said.
Recently updated evidence-based guidelines from the American Psychiatric Association recommend consideration of long-acting injectables for appropriate adults living with schizophrenia.
“Long-acting injectable treatments offer a number of advantages, compared to oral medication for schizophrenia, including relief from needing to remember to take medication daily, lower discontinuation rates, and sustained treatment over longer periods,” Bill Martin, PhD, with Janssen Research & Development, said in the release.
“Today’s approval enables us to rethink how we manage this chronic disease by offering patients and caregivers the potential for a life less defined by schizophrenia medication,” Dr. Martin added.
A version of this article first appeared on Medscape.com.