User login
Even a few days of steroids may be risky, new study suggests
Extended use of corticosteroids for chronic inflammatory conditions puts patients at risk for serious adverse events (AEs), including cardiovascular disease, osteoporosis, cataracts, and diabetes. Now, a growing body of evidence suggests that even short bursts of these drugs are associated with serious risks.
Most recently, a population-based study of more than 2.6 million people found that taking corticosteroids for 14 days or less was associated with a substantially greater risk for gastrointestinal (GI) bleeding, sepsis, and heart failure, particularly within the first 30 days after therapy.
In the study, Tsung-Chieh Yao, MD, PhD, a professor in the division of allergy, asthma, and rheumatology in the department of pediatrics at Chang Gung Memorial Hospital in Taoyuan, Taiwan, and colleagues used a self-controlled case series to analyze data from Taiwan’s National Health Insurance Research Database of medical claims. They compared patients’ conditions in the period from 5 to 90 days before treatment to conditions from the periods from 5 to 30 days and from 31 to 90 days after therapy.
With a median duration of 3 days of treatment, the incidence rate ratios (IRRs) were 1.80 (95% confidence interval, 1.75-1.84) for GI bleeding, 1.99 (95% CI, 1.70-2.32) for sepsis, and 2.37 (95% CI, 2.13-2.63) for heart failure.
Given the findings, physicians should weigh the benefits against the risks of rare but potentially serious consequences of these anti-inflammatory drugs, according to the authors.
“After initiating patients on oral steroid bursts, physicians should be on the lookout for these severe adverse events, particularly within the first month after initiation of steroid therapy,” Dr. Yao said in an interview.
The findings were published online July 6 in Annals of Internal Medicine.
Of the 15,859,129 adult Asians in the Taiwanese database, the study included 2,623,327 adults aged 20-64 years who received single steroid bursts (14 days or less) between Jan. 1, 2013, and Dec. 31, 2015.
Almost 60% of the indications were for skin disorders, such as eczema and urticaria, and for respiratory tract infections, such as sinusitis and acute pharyngitis. Among specialties, dermatology, otolaryngology, family practice, internal medicine, and pediatrics accounted for 88% of prescriptions.
“Our findings are important for physicians and guideline developers because short-term use of oral corticosteroids is common and the real-world safety of this approach remains unclear,” the authors wrote. They acknowledged that the database did not provide information on such potential confounders as disease severity and lifestyle factors, nor did it include children and vulnerable individuals, which may limit the generalizability of the results.
The findings echo those of a 2017 cohort study conducted by researchers at the University of Michigan in Ann Arbor. That study, by Akbar K. Waljee, MD, assistant professor of gastroenterology, University of Michigan, Ann Arbor, and colleagues, included data on more than 1.5 million privately insured U.S. adults. The researchers included somewhat longer steroid bursts of up to 30 days’ duration and found that use of the drugs was associated with a greater than fivefold increased risk for sepsis, a more than threefold increased risk for venous thromboembolism, and a nearly twofold increased risk for fracture within 30 days of starting treatment.
Furthermore, the elevated risk persisted at prednisone-equivalent doses of less than 20 mg/d (IRR, 4.02 for sepsis, 3.61 for venous thromboembolism, and 1.83 for fracture; all P < .001).
The U.S. study also found that during the 3-year period from 2012 to 2014, more than 20% of patients were prescribed short-term oral corticosteroids.
“Both studies indicate that these short-term regimens are more common in the real world than was previously thought and are not risk free,” Dr. Yao said.
Recognition that corticosteroids are associated with adverse events has been building for decades, according to the authors of an editorial that accompanies the new study.
“However, we commonly use short corticosteroid ‘bursts’ for minor ailments despite a lack of evidence for meaningful benefit. We are now learning that bursts as short as 3 days may increase risk for serious AEs, even in young and healthy people,” wrote editorialists Beth I. Wallace, MD, of the Center for Clinical Management Research at the VA Ann Arbor Healthcare System and the Institute for Healthcare Policy and Innovation at Michigan Medicine, Ann Arbor, and Dr. Waljee, who led the 2017 study.
Dr. Wallace and Dr. Waljee drew parallels between corticosteroid bursts and other short-term regimens, such as of antibiotics and opiates, in which prescriber preference and sometimes patient pressure play a role. “All of these treatments have well-defined indications but can cause net harm when used. We can thus conceive of a corticosteroid stewardship model of targeted interventions that aims to reduce inappropriate prescribing,” they wrote.
In an interview, Dr. Wallace, a rheumatologist who prescribes oral steroids fairly frequently, noted that the Taiwan study is the first to investigate steroid bursts. “Up till now, these very short courses have flown under the radar. Clinicians very commonly prescribe short courses to help relieve symptoms of self-limited conditions like bronchitis, and we assume that because the exposure duration is short, the risks are low, especially for patients who are otherwise healthy.”
She warned that the data in the current study indicate that these short bursts – even at the lower end of the 1- to 2-week courses American physicians prescribe most often – carry small but real increases in risk for serious AEs. “And these increases were seen in young, healthy people, not just in people with preexisting conditions,” she said. “So, we might need to start thinking harder about how we are prescribing even these very short courses of steroids and try to use steroids only when their meaningful benefits really outweigh the risk.”
She noted that a patient with a chronic inflammatory condition such as rheumatoid arthritis may benefit substantially from short-term steroids to treat a disease flare. In that specific case, the benefits of short-term steroids may outweigh the risks, Dr. Wallace said.
But not everyone thinks a new strategy is needed. For Whitney A. High, MD, associate professor of dermatology and pathology at the University of Colorado at Denver, Aurora, the overprescribing of short-term corticosteroids is not a problem, and dermatologists are already exercising caution.
“I only prescribe these drugs short term to, at a guess, about 1 in 40 patients and only when a patient is miserable and quality of life is being seriously affected,” he said in an interview. “And that’s something that can’t be measured in a database study like the one from Taiwan but only in a risk-benefit analysis,” he said.
Furthermore, dermatologists have other drugs and technologies in their armamentarium, including topical steroids with occlusion or with wet wraps, phototherapy, phosphodiesterase inhibitors, calcipotriene, methotrexate and other immunosuppressive agents, and biologics. “In fact, many of these agents are specifically referred to as steroid-sparing,” Dr. High said.
Nor does he experience much pressure from patients to prescribe these drugs. “While occasionally I may encounter a patient who places pressure on me for oral steroids, it’s probably not nearly as frequently as providers in other fields are pressured to prescribe antibiotics or narcotics,” he said.
According to the Taiwanese researchers, the next step is to conduct more studies, including clinical trials, to determine optimal use of corticosteroids by monitoring adverse events. In the meantime, for practitioners such as Dr. Wallace and Dr. High, there is ample evidence from several recent studies of the harms of short-term corticosteroids, whereas the benefits for patients with self-limiting conditions remain uncertain. “This and other studies like it quite appropriately remind providers to avoid oral steroids when they’re not necessary and to seek alternatives where possible,” Dr. High said.
The study was supported by the National Health Research Institutes of Taiwan, the Ministry of Science and Technology of Taiwan, the Chang Gung Medical Foundation, and the Eunice Kennedy Shriver National Institute of Child Health and Human Development of the National Institutes of Health (NIH). Dr. Yao has disclosed no relevant financial relationships. Dr. Wu has received grants from GlaxoSmithKline outside the submitted work. The editorialists and Dr. High have disclosed no relevant financial relationships. Dr. Wallace received an NIH grant during the writing of the editorial.
A version of this article originally appeared on Medscape.com.
Extended use of corticosteroids for chronic inflammatory conditions puts patients at risk for serious adverse events (AEs), including cardiovascular disease, osteoporosis, cataracts, and diabetes. Now, a growing body of evidence suggests that even short bursts of these drugs are associated with serious risks.
Most recently, a population-based study of more than 2.6 million people found that taking corticosteroids for 14 days or less was associated with a substantially greater risk for gastrointestinal (GI) bleeding, sepsis, and heart failure, particularly within the first 30 days after therapy.
In the study, Tsung-Chieh Yao, MD, PhD, a professor in the division of allergy, asthma, and rheumatology in the department of pediatrics at Chang Gung Memorial Hospital in Taoyuan, Taiwan, and colleagues used a self-controlled case series to analyze data from Taiwan’s National Health Insurance Research Database of medical claims. They compared patients’ conditions in the period from 5 to 90 days before treatment to conditions from the periods from 5 to 30 days and from 31 to 90 days after therapy.
With a median duration of 3 days of treatment, the incidence rate ratios (IRRs) were 1.80 (95% confidence interval, 1.75-1.84) for GI bleeding, 1.99 (95% CI, 1.70-2.32) for sepsis, and 2.37 (95% CI, 2.13-2.63) for heart failure.
Given the findings, physicians should weigh the benefits against the risks of rare but potentially serious consequences of these anti-inflammatory drugs, according to the authors.
“After initiating patients on oral steroid bursts, physicians should be on the lookout for these severe adverse events, particularly within the first month after initiation of steroid therapy,” Dr. Yao said in an interview.
The findings were published online July 6 in Annals of Internal Medicine.
Of the 15,859,129 adult Asians in the Taiwanese database, the study included 2,623,327 adults aged 20-64 years who received single steroid bursts (14 days or less) between Jan. 1, 2013, and Dec. 31, 2015.
Almost 60% of the indications were for skin disorders, such as eczema and urticaria, and for respiratory tract infections, such as sinusitis and acute pharyngitis. Among specialties, dermatology, otolaryngology, family practice, internal medicine, and pediatrics accounted for 88% of prescriptions.
“Our findings are important for physicians and guideline developers because short-term use of oral corticosteroids is common and the real-world safety of this approach remains unclear,” the authors wrote. They acknowledged that the database did not provide information on such potential confounders as disease severity and lifestyle factors, nor did it include children and vulnerable individuals, which may limit the generalizability of the results.
The findings echo those of a 2017 cohort study conducted by researchers at the University of Michigan in Ann Arbor. That study, by Akbar K. Waljee, MD, assistant professor of gastroenterology, University of Michigan, Ann Arbor, and colleagues, included data on more than 1.5 million privately insured U.S. adults. The researchers included somewhat longer steroid bursts of up to 30 days’ duration and found that use of the drugs was associated with a greater than fivefold increased risk for sepsis, a more than threefold increased risk for venous thromboembolism, and a nearly twofold increased risk for fracture within 30 days of starting treatment.
Furthermore, the elevated risk persisted at prednisone-equivalent doses of less than 20 mg/d (IRR, 4.02 for sepsis, 3.61 for venous thromboembolism, and 1.83 for fracture; all P < .001).
The U.S. study also found that during the 3-year period from 2012 to 2014, more than 20% of patients were prescribed short-term oral corticosteroids.
“Both studies indicate that these short-term regimens are more common in the real world than was previously thought and are not risk free,” Dr. Yao said.
Recognition that corticosteroids are associated with adverse events has been building for decades, according to the authors of an editorial that accompanies the new study.
“However, we commonly use short corticosteroid ‘bursts’ for minor ailments despite a lack of evidence for meaningful benefit. We are now learning that bursts as short as 3 days may increase risk for serious AEs, even in young and healthy people,” wrote editorialists Beth I. Wallace, MD, of the Center for Clinical Management Research at the VA Ann Arbor Healthcare System and the Institute for Healthcare Policy and Innovation at Michigan Medicine, Ann Arbor, and Dr. Waljee, who led the 2017 study.
Dr. Wallace and Dr. Waljee drew parallels between corticosteroid bursts and other short-term regimens, such as of antibiotics and opiates, in which prescriber preference and sometimes patient pressure play a role. “All of these treatments have well-defined indications but can cause net harm when used. We can thus conceive of a corticosteroid stewardship model of targeted interventions that aims to reduce inappropriate prescribing,” they wrote.
In an interview, Dr. Wallace, a rheumatologist who prescribes oral steroids fairly frequently, noted that the Taiwan study is the first to investigate steroid bursts. “Up till now, these very short courses have flown under the radar. Clinicians very commonly prescribe short courses to help relieve symptoms of self-limited conditions like bronchitis, and we assume that because the exposure duration is short, the risks are low, especially for patients who are otherwise healthy.”
She warned that the data in the current study indicate that these short bursts – even at the lower end of the 1- to 2-week courses American physicians prescribe most often – carry small but real increases in risk for serious AEs. “And these increases were seen in young, healthy people, not just in people with preexisting conditions,” she said. “So, we might need to start thinking harder about how we are prescribing even these very short courses of steroids and try to use steroids only when their meaningful benefits really outweigh the risk.”
She noted that a patient with a chronic inflammatory condition such as rheumatoid arthritis may benefit substantially from short-term steroids to treat a disease flare. In that specific case, the benefits of short-term steroids may outweigh the risks, Dr. Wallace said.
But not everyone thinks a new strategy is needed. For Whitney A. High, MD, associate professor of dermatology and pathology at the University of Colorado at Denver, Aurora, the overprescribing of short-term corticosteroids is not a problem, and dermatologists are already exercising caution.
“I only prescribe these drugs short term to, at a guess, about 1 in 40 patients and only when a patient is miserable and quality of life is being seriously affected,” he said in an interview. “And that’s something that can’t be measured in a database study like the one from Taiwan but only in a risk-benefit analysis,” he said.
Furthermore, dermatologists have other drugs and technologies in their armamentarium, including topical steroids with occlusion or with wet wraps, phototherapy, phosphodiesterase inhibitors, calcipotriene, methotrexate and other immunosuppressive agents, and biologics. “In fact, many of these agents are specifically referred to as steroid-sparing,” Dr. High said.
Nor does he experience much pressure from patients to prescribe these drugs. “While occasionally I may encounter a patient who places pressure on me for oral steroids, it’s probably not nearly as frequently as providers in other fields are pressured to prescribe antibiotics or narcotics,” he said.
According to the Taiwanese researchers, the next step is to conduct more studies, including clinical trials, to determine optimal use of corticosteroids by monitoring adverse events. In the meantime, for practitioners such as Dr. Wallace and Dr. High, there is ample evidence from several recent studies of the harms of short-term corticosteroids, whereas the benefits for patients with self-limiting conditions remain uncertain. “This and other studies like it quite appropriately remind providers to avoid oral steroids when they’re not necessary and to seek alternatives where possible,” Dr. High said.
The study was supported by the National Health Research Institutes of Taiwan, the Ministry of Science and Technology of Taiwan, the Chang Gung Medical Foundation, and the Eunice Kennedy Shriver National Institute of Child Health and Human Development of the National Institutes of Health (NIH). Dr. Yao has disclosed no relevant financial relationships. Dr. Wu has received grants from GlaxoSmithKline outside the submitted work. The editorialists and Dr. High have disclosed no relevant financial relationships. Dr. Wallace received an NIH grant during the writing of the editorial.
A version of this article originally appeared on Medscape.com.
Extended use of corticosteroids for chronic inflammatory conditions puts patients at risk for serious adverse events (AEs), including cardiovascular disease, osteoporosis, cataracts, and diabetes. Now, a growing body of evidence suggests that even short bursts of these drugs are associated with serious risks.
Most recently, a population-based study of more than 2.6 million people found that taking corticosteroids for 14 days or less was associated with a substantially greater risk for gastrointestinal (GI) bleeding, sepsis, and heart failure, particularly within the first 30 days after therapy.
In the study, Tsung-Chieh Yao, MD, PhD, a professor in the division of allergy, asthma, and rheumatology in the department of pediatrics at Chang Gung Memorial Hospital in Taoyuan, Taiwan, and colleagues used a self-controlled case series to analyze data from Taiwan’s National Health Insurance Research Database of medical claims. They compared patients’ conditions in the period from 5 to 90 days before treatment to conditions from the periods from 5 to 30 days and from 31 to 90 days after therapy.
With a median duration of 3 days of treatment, the incidence rate ratios (IRRs) were 1.80 (95% confidence interval, 1.75-1.84) for GI bleeding, 1.99 (95% CI, 1.70-2.32) for sepsis, and 2.37 (95% CI, 2.13-2.63) for heart failure.
Given the findings, physicians should weigh the benefits against the risks of rare but potentially serious consequences of these anti-inflammatory drugs, according to the authors.
“After initiating patients on oral steroid bursts, physicians should be on the lookout for these severe adverse events, particularly within the first month after initiation of steroid therapy,” Dr. Yao said in an interview.
The findings were published online July 6 in Annals of Internal Medicine.
Of the 15,859,129 adult Asians in the Taiwanese database, the study included 2,623,327 adults aged 20-64 years who received single steroid bursts (14 days or less) between Jan. 1, 2013, and Dec. 31, 2015.
Almost 60% of the indications were for skin disorders, such as eczema and urticaria, and for respiratory tract infections, such as sinusitis and acute pharyngitis. Among specialties, dermatology, otolaryngology, family practice, internal medicine, and pediatrics accounted for 88% of prescriptions.
“Our findings are important for physicians and guideline developers because short-term use of oral corticosteroids is common and the real-world safety of this approach remains unclear,” the authors wrote. They acknowledged that the database did not provide information on such potential confounders as disease severity and lifestyle factors, nor did it include children and vulnerable individuals, which may limit the generalizability of the results.
The findings echo those of a 2017 cohort study conducted by researchers at the University of Michigan in Ann Arbor. That study, by Akbar K. Waljee, MD, assistant professor of gastroenterology, University of Michigan, Ann Arbor, and colleagues, included data on more than 1.5 million privately insured U.S. adults. The researchers included somewhat longer steroid bursts of up to 30 days’ duration and found that use of the drugs was associated with a greater than fivefold increased risk for sepsis, a more than threefold increased risk for venous thromboembolism, and a nearly twofold increased risk for fracture within 30 days of starting treatment.
Furthermore, the elevated risk persisted at prednisone-equivalent doses of less than 20 mg/d (IRR, 4.02 for sepsis, 3.61 for venous thromboembolism, and 1.83 for fracture; all P < .001).
The U.S. study also found that during the 3-year period from 2012 to 2014, more than 20% of patients were prescribed short-term oral corticosteroids.
“Both studies indicate that these short-term regimens are more common in the real world than was previously thought and are not risk free,” Dr. Yao said.
Recognition that corticosteroids are associated with adverse events has been building for decades, according to the authors of an editorial that accompanies the new study.
“However, we commonly use short corticosteroid ‘bursts’ for minor ailments despite a lack of evidence for meaningful benefit. We are now learning that bursts as short as 3 days may increase risk for serious AEs, even in young and healthy people,” wrote editorialists Beth I. Wallace, MD, of the Center for Clinical Management Research at the VA Ann Arbor Healthcare System and the Institute for Healthcare Policy and Innovation at Michigan Medicine, Ann Arbor, and Dr. Waljee, who led the 2017 study.
Dr. Wallace and Dr. Waljee drew parallels between corticosteroid bursts and other short-term regimens, such as of antibiotics and opiates, in which prescriber preference and sometimes patient pressure play a role. “All of these treatments have well-defined indications but can cause net harm when used. We can thus conceive of a corticosteroid stewardship model of targeted interventions that aims to reduce inappropriate prescribing,” they wrote.
In an interview, Dr. Wallace, a rheumatologist who prescribes oral steroids fairly frequently, noted that the Taiwan study is the first to investigate steroid bursts. “Up till now, these very short courses have flown under the radar. Clinicians very commonly prescribe short courses to help relieve symptoms of self-limited conditions like bronchitis, and we assume that because the exposure duration is short, the risks are low, especially for patients who are otherwise healthy.”
She warned that the data in the current study indicate that these short bursts – even at the lower end of the 1- to 2-week courses American physicians prescribe most often – carry small but real increases in risk for serious AEs. “And these increases were seen in young, healthy people, not just in people with preexisting conditions,” she said. “So, we might need to start thinking harder about how we are prescribing even these very short courses of steroids and try to use steroids only when their meaningful benefits really outweigh the risk.”
She noted that a patient with a chronic inflammatory condition such as rheumatoid arthritis may benefit substantially from short-term steroids to treat a disease flare. In that specific case, the benefits of short-term steroids may outweigh the risks, Dr. Wallace said.
But not everyone thinks a new strategy is needed. For Whitney A. High, MD, associate professor of dermatology and pathology at the University of Colorado at Denver, Aurora, the overprescribing of short-term corticosteroids is not a problem, and dermatologists are already exercising caution.
“I only prescribe these drugs short term to, at a guess, about 1 in 40 patients and only when a patient is miserable and quality of life is being seriously affected,” he said in an interview. “And that’s something that can’t be measured in a database study like the one from Taiwan but only in a risk-benefit analysis,” he said.
Furthermore, dermatologists have other drugs and technologies in their armamentarium, including topical steroids with occlusion or with wet wraps, phototherapy, phosphodiesterase inhibitors, calcipotriene, methotrexate and other immunosuppressive agents, and biologics. “In fact, many of these agents are specifically referred to as steroid-sparing,” Dr. High said.
Nor does he experience much pressure from patients to prescribe these drugs. “While occasionally I may encounter a patient who places pressure on me for oral steroids, it’s probably not nearly as frequently as providers in other fields are pressured to prescribe antibiotics or narcotics,” he said.
According to the Taiwanese researchers, the next step is to conduct more studies, including clinical trials, to determine optimal use of corticosteroids by monitoring adverse events. In the meantime, for practitioners such as Dr. Wallace and Dr. High, there is ample evidence from several recent studies of the harms of short-term corticosteroids, whereas the benefits for patients with self-limiting conditions remain uncertain. “This and other studies like it quite appropriately remind providers to avoid oral steroids when they’re not necessary and to seek alternatives where possible,” Dr. High said.
The study was supported by the National Health Research Institutes of Taiwan, the Ministry of Science and Technology of Taiwan, the Chang Gung Medical Foundation, and the Eunice Kennedy Shriver National Institute of Child Health and Human Development of the National Institutes of Health (NIH). Dr. Yao has disclosed no relevant financial relationships. Dr. Wu has received grants from GlaxoSmithKline outside the submitted work. The editorialists and Dr. High have disclosed no relevant financial relationships. Dr. Wallace received an NIH grant during the writing of the editorial.
A version of this article originally appeared on Medscape.com.
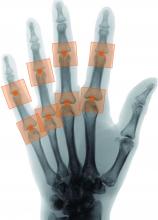
Automated RA image scoring could be coming
A novel program that aims to automate the Sharp-van der Heijde scoring of radiographs of patients with rheumatoid arthritis has shown good reliability in identifying regions of interest and matching human reader scoring for joint-space narrowing, according to a report given at the annual European Congress of Rheumatology, held online this year because of COVID-19.
First author and presenter Thomas Deimel, MD, and colleagues at the Medical University of Vienna said their program, called autoscoRA, may be a solution to the problem of readers having to make subjective calls on the severity of damage seen on radiographs.
Although the work continues to be validated, Dr. Deimel, a resident at the university, is confident in the system as is. “I think for joint space narrowing, we’re there at the point where this could be used and could be as good as a human reader in terms of reliability,” he said in an interview. To find out, the group plans to compare the variability between autoscoRA and a gold-standard human reader against the variability seen between human readers. If the two measures of variability are similar, it would provide a strong endorsement.
The effort is far from the first to develop an automatic scoring system for RA images, but no fully automated system has emerged as reliable, according to Dr. Deimel. He thinks one main issue for others has been lack of access to a sufficient data set to train systems. It can be difficult to find enough training images because many types of joint damage are comparatively uncommon. The problem is made even worse because images can be hard to interpret: The shapes that the system must decipher can be misleading, especially in positions of tendon insertion or ligament attachment that can resemble damage. Differing angles of view between various training images can also complicate matters.
The autoscoRA program is based on modifications of a form of convolutional neural network called the VGG16 architecture. The team used 2,207 images from 270 patients to train autoscoRA, 1,150 images from 133 patients for validation, and 1,834 images from 237 patients to test it.
The group had access to a high-quality data set of almost 6,000 hand radiographs from their institution, the result of foresight of principal investigator Daniel Aletaha, MD, and his predecessor Josef Smolen, MD. They “thought ahead and started collecting data and had all of it scored,” Dr. Deimel said. The work wasn’t all completed ahead of time, though. Dr. Deimel had to pull images from the hospital’s system sort through them manually.
The group also benefited from close proximity to computer scientists, including coauthor Georg Langs from the Medical University of Vienna’s computational imaging research lab. “We were lucky that we have a computer science department that is very much involved in medical imaging,” Dr. Deimel said.
The trained system successfully identified regions of interest in 96% of joints. It calculated the same score as the human reader in 80.5% of metacarpophalangeal joints and 72.3% of proximal interphalangeal joints. It deviated by more than 1 point from the gold-standard score in just 1.8% of metacarpophalangeal joints and 1.7% of proximal interphalangeal joints.
The researchers aim next to extend the program to bone erosions and also to images of the wrists and feet. They also hope to use scores from the program in clinical trials to measure a treatment’s effect, in registries of routine patient visits where thousands of such images along with clinical data could form the basis of informative observational studies, and in clinical practice, though likely with human oversight.
The study received no outside financial support. Dr. Deimel had no relevant financial disclosures. Mr. Langs reported being cofounder and shareholder of contextflow and receiving grants from Novartis, Siemens Healthineers, and NVIDIA. Dr. Aletaha reported financial relationships with many companies marketing drugs for rheumatoid arthritis.
SOURCE: Deimel T et al. Ann Rheum Dis. 2020 Jun;79(suppl 1):39-40.
A novel program that aims to automate the Sharp-van der Heijde scoring of radiographs of patients with rheumatoid arthritis has shown good reliability in identifying regions of interest and matching human reader scoring for joint-space narrowing, according to a report given at the annual European Congress of Rheumatology, held online this year because of COVID-19.
First author and presenter Thomas Deimel, MD, and colleagues at the Medical University of Vienna said their program, called autoscoRA, may be a solution to the problem of readers having to make subjective calls on the severity of damage seen on radiographs.
Although the work continues to be validated, Dr. Deimel, a resident at the university, is confident in the system as is. “I think for joint space narrowing, we’re there at the point where this could be used and could be as good as a human reader in terms of reliability,” he said in an interview. To find out, the group plans to compare the variability between autoscoRA and a gold-standard human reader against the variability seen between human readers. If the two measures of variability are similar, it would provide a strong endorsement.
The effort is far from the first to develop an automatic scoring system for RA images, but no fully automated system has emerged as reliable, according to Dr. Deimel. He thinks one main issue for others has been lack of access to a sufficient data set to train systems. It can be difficult to find enough training images because many types of joint damage are comparatively uncommon. The problem is made even worse because images can be hard to interpret: The shapes that the system must decipher can be misleading, especially in positions of tendon insertion or ligament attachment that can resemble damage. Differing angles of view between various training images can also complicate matters.
The autoscoRA program is based on modifications of a form of convolutional neural network called the VGG16 architecture. The team used 2,207 images from 270 patients to train autoscoRA, 1,150 images from 133 patients for validation, and 1,834 images from 237 patients to test it.
The group had access to a high-quality data set of almost 6,000 hand radiographs from their institution, the result of foresight of principal investigator Daniel Aletaha, MD, and his predecessor Josef Smolen, MD. They “thought ahead and started collecting data and had all of it scored,” Dr. Deimel said. The work wasn’t all completed ahead of time, though. Dr. Deimel had to pull images from the hospital’s system sort through them manually.
The group also benefited from close proximity to computer scientists, including coauthor Georg Langs from the Medical University of Vienna’s computational imaging research lab. “We were lucky that we have a computer science department that is very much involved in medical imaging,” Dr. Deimel said.
The trained system successfully identified regions of interest in 96% of joints. It calculated the same score as the human reader in 80.5% of metacarpophalangeal joints and 72.3% of proximal interphalangeal joints. It deviated by more than 1 point from the gold-standard score in just 1.8% of metacarpophalangeal joints and 1.7% of proximal interphalangeal joints.
The researchers aim next to extend the program to bone erosions and also to images of the wrists and feet. They also hope to use scores from the program in clinical trials to measure a treatment’s effect, in registries of routine patient visits where thousands of such images along with clinical data could form the basis of informative observational studies, and in clinical practice, though likely with human oversight.
The study received no outside financial support. Dr. Deimel had no relevant financial disclosures. Mr. Langs reported being cofounder and shareholder of contextflow and receiving grants from Novartis, Siemens Healthineers, and NVIDIA. Dr. Aletaha reported financial relationships with many companies marketing drugs for rheumatoid arthritis.
SOURCE: Deimel T et al. Ann Rheum Dis. 2020 Jun;79(suppl 1):39-40.
A novel program that aims to automate the Sharp-van der Heijde scoring of radiographs of patients with rheumatoid arthritis has shown good reliability in identifying regions of interest and matching human reader scoring for joint-space narrowing, according to a report given at the annual European Congress of Rheumatology, held online this year because of COVID-19.
First author and presenter Thomas Deimel, MD, and colleagues at the Medical University of Vienna said their program, called autoscoRA, may be a solution to the problem of readers having to make subjective calls on the severity of damage seen on radiographs.
Although the work continues to be validated, Dr. Deimel, a resident at the university, is confident in the system as is. “I think for joint space narrowing, we’re there at the point where this could be used and could be as good as a human reader in terms of reliability,” he said in an interview. To find out, the group plans to compare the variability between autoscoRA and a gold-standard human reader against the variability seen between human readers. If the two measures of variability are similar, it would provide a strong endorsement.
The effort is far from the first to develop an automatic scoring system for RA images, but no fully automated system has emerged as reliable, according to Dr. Deimel. He thinks one main issue for others has been lack of access to a sufficient data set to train systems. It can be difficult to find enough training images because many types of joint damage are comparatively uncommon. The problem is made even worse because images can be hard to interpret: The shapes that the system must decipher can be misleading, especially in positions of tendon insertion or ligament attachment that can resemble damage. Differing angles of view between various training images can also complicate matters.
The autoscoRA program is based on modifications of a form of convolutional neural network called the VGG16 architecture. The team used 2,207 images from 270 patients to train autoscoRA, 1,150 images from 133 patients for validation, and 1,834 images from 237 patients to test it.
The group had access to a high-quality data set of almost 6,000 hand radiographs from their institution, the result of foresight of principal investigator Daniel Aletaha, MD, and his predecessor Josef Smolen, MD. They “thought ahead and started collecting data and had all of it scored,” Dr. Deimel said. The work wasn’t all completed ahead of time, though. Dr. Deimel had to pull images from the hospital’s system sort through them manually.
The group also benefited from close proximity to computer scientists, including coauthor Georg Langs from the Medical University of Vienna’s computational imaging research lab. “We were lucky that we have a computer science department that is very much involved in medical imaging,” Dr. Deimel said.
The trained system successfully identified regions of interest in 96% of joints. It calculated the same score as the human reader in 80.5% of metacarpophalangeal joints and 72.3% of proximal interphalangeal joints. It deviated by more than 1 point from the gold-standard score in just 1.8% of metacarpophalangeal joints and 1.7% of proximal interphalangeal joints.
The researchers aim next to extend the program to bone erosions and also to images of the wrists and feet. They also hope to use scores from the program in clinical trials to measure a treatment’s effect, in registries of routine patient visits where thousands of such images along with clinical data could form the basis of informative observational studies, and in clinical practice, though likely with human oversight.
The study received no outside financial support. Dr. Deimel had no relevant financial disclosures. Mr. Langs reported being cofounder and shareholder of contextflow and receiving grants from Novartis, Siemens Healthineers, and NVIDIA. Dr. Aletaha reported financial relationships with many companies marketing drugs for rheumatoid arthritis.
SOURCE: Deimel T et al. Ann Rheum Dis. 2020 Jun;79(suppl 1):39-40.
FROM THE EULAR 2020 E-CONGRESS
ACIP approves flu vaccine recommendations for 2020-2021
– Fluzone high-dose quadrivalent, which replaces the trivalent Fluzone high-dose and Fluad quadrivalent (Seqirus), according to the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
At a virtual meeting on June 24, the committee voted unanimously to approve the vaccine recommendations for annual influenza immunization of all individuals aged 6 months and older. They also voted to accept some guidance and language changes to the recommendations.
The past flu season was unique in its overlap with the emergence of the COVID-19 coronavirus, which likely contributed to a third peak in reported cases of influenza-like illness at approximately week 14 of last season, said Lisa Grohskopf, MD, of the CDC’s influenza division, who presented data on last year’s activity and the updates for next season.
The CDC estimates that 39,000,000-56,000,000 flu illnesses occurred in the United States from Oct. 1, 2019, to April 4, 2020, said Dr. Grohskopf. Estimates also suggest as many as 740,000 hospitalizations and 62,000 deaths related to the seasonal flu.
Preliminary results of vaccine effectiveness showed 39% overall for the 2019-2020 season, with more substantial protection against influenza B and lower protection against A/H1N1pmd09.
Vaccine safety data from the Vaccine Adverse Event Reporting System and Vaccine Safety Datalink showed no new safety concerns for any flu vaccine types used last year, Dr. Grohskopf noted.
Based on this information, three components (A/H1N1pdm09, A/H3N2, and B/Victoria) have been updated for the 2020-2021 vaccines, said Dr. Grohskopf. The egg-based influenza vaccines will include hemagglutinin derived from an A/Guangdong-Maonan/SWL1536/2019(H1N1)pdm09–like virus, an A/Hong Kong/2671/2019(H3N2)–like virus and a B/Washington/02/2019 (Victoria lineage)–like virus, and (for quadrivalent vaccines) a B/Phuket/3073/2013 (Yamagata lineage)–like virus.
Nonegg vaccines will contain hemagglutinin derived from an A/Hawaii/70/2019 (H1N1)pdm09–like virus, an A/Hong Kong/45/2019 (H3N2)–like virus, a B/Washington/02/2019 (Victoria lineage)–like virus, and a B/Phuket/3073/2013 (Yamagata lineage)–like virus.
New guidance for next year’s flu season includes a change to the language in the contraindications and precautions table to simply read “Contraindications,” with more details in the text explaining package insert contraindications and ACIP recommendations, Dr. Grohskopf said. In addition, updated guidance clarifies that live-attenuated influenza vaccine quadravalents (LAIV4) should not be used in patients with cochlear implants, active cerebrospinal fluid leaks, and anatomical or functional asplenia, based on ACIP’s review of the latest evidence and the availability of alternative vaccines.
ACIP also updated guidance on the use of antivirals and LAIV4. Based on half-lives, language was added indicating that clinicians should assume interference if antivirals are given within certain intervals of LAIV4, Dr. Grohskopf explained. “Newer antivirals peramivir and baloxavir have longer half-lives than oseltamivir and zanamivir, and insufficient data are available on the use of LAIV4 in the setting of antiviral use.”
The ACIP members had no financial conflicts to disclose.
– Fluzone high-dose quadrivalent, which replaces the trivalent Fluzone high-dose and Fluad quadrivalent (Seqirus), according to the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
At a virtual meeting on June 24, the committee voted unanimously to approve the vaccine recommendations for annual influenza immunization of all individuals aged 6 months and older. They also voted to accept some guidance and language changes to the recommendations.
The past flu season was unique in its overlap with the emergence of the COVID-19 coronavirus, which likely contributed to a third peak in reported cases of influenza-like illness at approximately week 14 of last season, said Lisa Grohskopf, MD, of the CDC’s influenza division, who presented data on last year’s activity and the updates for next season.
The CDC estimates that 39,000,000-56,000,000 flu illnesses occurred in the United States from Oct. 1, 2019, to April 4, 2020, said Dr. Grohskopf. Estimates also suggest as many as 740,000 hospitalizations and 62,000 deaths related to the seasonal flu.
Preliminary results of vaccine effectiveness showed 39% overall for the 2019-2020 season, with more substantial protection against influenza B and lower protection against A/H1N1pmd09.
Vaccine safety data from the Vaccine Adverse Event Reporting System and Vaccine Safety Datalink showed no new safety concerns for any flu vaccine types used last year, Dr. Grohskopf noted.
Based on this information, three components (A/H1N1pdm09, A/H3N2, and B/Victoria) have been updated for the 2020-2021 vaccines, said Dr. Grohskopf. The egg-based influenza vaccines will include hemagglutinin derived from an A/Guangdong-Maonan/SWL1536/2019(H1N1)pdm09–like virus, an A/Hong Kong/2671/2019(H3N2)–like virus and a B/Washington/02/2019 (Victoria lineage)–like virus, and (for quadrivalent vaccines) a B/Phuket/3073/2013 (Yamagata lineage)–like virus.
Nonegg vaccines will contain hemagglutinin derived from an A/Hawaii/70/2019 (H1N1)pdm09–like virus, an A/Hong Kong/45/2019 (H3N2)–like virus, a B/Washington/02/2019 (Victoria lineage)–like virus, and a B/Phuket/3073/2013 (Yamagata lineage)–like virus.
New guidance for next year’s flu season includes a change to the language in the contraindications and precautions table to simply read “Contraindications,” with more details in the text explaining package insert contraindications and ACIP recommendations, Dr. Grohskopf said. In addition, updated guidance clarifies that live-attenuated influenza vaccine quadravalents (LAIV4) should not be used in patients with cochlear implants, active cerebrospinal fluid leaks, and anatomical or functional asplenia, based on ACIP’s review of the latest evidence and the availability of alternative vaccines.
ACIP also updated guidance on the use of antivirals and LAIV4. Based on half-lives, language was added indicating that clinicians should assume interference if antivirals are given within certain intervals of LAIV4, Dr. Grohskopf explained. “Newer antivirals peramivir and baloxavir have longer half-lives than oseltamivir and zanamivir, and insufficient data are available on the use of LAIV4 in the setting of antiviral use.”
The ACIP members had no financial conflicts to disclose.
– Fluzone high-dose quadrivalent, which replaces the trivalent Fluzone high-dose and Fluad quadrivalent (Seqirus), according to the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
At a virtual meeting on June 24, the committee voted unanimously to approve the vaccine recommendations for annual influenza immunization of all individuals aged 6 months and older. They also voted to accept some guidance and language changes to the recommendations.
The past flu season was unique in its overlap with the emergence of the COVID-19 coronavirus, which likely contributed to a third peak in reported cases of influenza-like illness at approximately week 14 of last season, said Lisa Grohskopf, MD, of the CDC’s influenza division, who presented data on last year’s activity and the updates for next season.
The CDC estimates that 39,000,000-56,000,000 flu illnesses occurred in the United States from Oct. 1, 2019, to April 4, 2020, said Dr. Grohskopf. Estimates also suggest as many as 740,000 hospitalizations and 62,000 deaths related to the seasonal flu.
Preliminary results of vaccine effectiveness showed 39% overall for the 2019-2020 season, with more substantial protection against influenza B and lower protection against A/H1N1pmd09.
Vaccine safety data from the Vaccine Adverse Event Reporting System and Vaccine Safety Datalink showed no new safety concerns for any flu vaccine types used last year, Dr. Grohskopf noted.
Based on this information, three components (A/H1N1pdm09, A/H3N2, and B/Victoria) have been updated for the 2020-2021 vaccines, said Dr. Grohskopf. The egg-based influenza vaccines will include hemagglutinin derived from an A/Guangdong-Maonan/SWL1536/2019(H1N1)pdm09–like virus, an A/Hong Kong/2671/2019(H3N2)–like virus and a B/Washington/02/2019 (Victoria lineage)–like virus, and (for quadrivalent vaccines) a B/Phuket/3073/2013 (Yamagata lineage)–like virus.
Nonegg vaccines will contain hemagglutinin derived from an A/Hawaii/70/2019 (H1N1)pdm09–like virus, an A/Hong Kong/45/2019 (H3N2)–like virus, a B/Washington/02/2019 (Victoria lineage)–like virus, and a B/Phuket/3073/2013 (Yamagata lineage)–like virus.
New guidance for next year’s flu season includes a change to the language in the contraindications and precautions table to simply read “Contraindications,” with more details in the text explaining package insert contraindications and ACIP recommendations, Dr. Grohskopf said. In addition, updated guidance clarifies that live-attenuated influenza vaccine quadravalents (LAIV4) should not be used in patients with cochlear implants, active cerebrospinal fluid leaks, and anatomical or functional asplenia, based on ACIP’s review of the latest evidence and the availability of alternative vaccines.
ACIP also updated guidance on the use of antivirals and LAIV4. Based on half-lives, language was added indicating that clinicians should assume interference if antivirals are given within certain intervals of LAIV4, Dr. Grohskopf explained. “Newer antivirals peramivir and baloxavir have longer half-lives than oseltamivir and zanamivir, and insufficient data are available on the use of LAIV4 in the setting of antiviral use.”
The ACIP members had no financial conflicts to disclose.
EULAR gives pointers on intra-articular injection best practices
New EULAR recommendations for the intra-articular (IA) treatment of arthropathies aim to facilitate uniformity and quality of care for this mainstay of rheumatologic practice, according to a report on the new guidance that was presented at the annual European Congress of Rheumatology, held online this year due to COVID-19.
Until now there were no official recommendations on how best to use it in everyday practice. “This is the first time that there’s been a joint effort to develop evidence-based recommendations,” Jacqueline Usón, MD, PhD, associate professor medicine at Rey Juan Carlos University in Madrid, said in an interview. “Everything that we are saying is pretty logical, but it’s nice to see it put in recommendations based on evidence.”
IA therapy has been around for decades and is key for treating adults with a number of different conditions where synovitis, effusion, pain, or all three, are present, such as inflammatory arthritis and osteoarthritis, Dr. Usón observed during her presentation.
“Today, commonly used injectables are not only corticosteroids but also local anesthetics, hyaluronic acid, blood products, and maybe pharmaceuticals,” she said, adding that “there is a wide variation in the way intra-articular therapies are used and delivered to patients.” Health professionals also have very different views and habits depending on geographic locations and health care systems, she observed. Ironing out the variation was one of the main objectives of the recommendations.
As one of the two conveners of the EULAR task force behind the recommendations, Dr. Usón, herself a rheumatologist at University Hospital of Móstoles, pointed out that the task force brought together a range of specialties – rheumatologists, orthopedic surgeons, radiologists, nuclear medicine specialists, among others, as well as patients – to ensure that the best advice could be given.
The task force followed EULAR standard operating procedures for developing recommendations, with discussion groups, systematic literature reviews, and Delphi technique-based consensus all being employed. The literature search considered publications from 1946 up until 2019.
“We agreed on the need for more background information from health professionals and patients, so we developed two surveys: One for health professionals with 160 items, [for which] we obtained 186 responses from 26 countries; and the patient survey was made up of 44 items, translated into 10 different languages, and we obtained 200 responses,” she said.
The results of the systematic literature review and surveys were used to help form expert consensus, leading to 5 overarching principles and 11 recommendations that look at before, during, and after intra-articular therapy.
Five overarching principles
The first overarching principle recognizes the widespread use of IA therapies and that their use is specific to the disease that is being treated and “may not be interchangeable across indications,” Dr. Usón said. The second principle concerns improving patient-centered outcomes, which are “those that are relevant to the patient,” and include the benefits, harms, preferences, or implications for self-management.
“Contextual factors are important and contribute to the effect of IAT [intra-articular treatment],” she said, discussing the third principle. “These include effective communication, patient expectations, or settings [where the procedure takes place]. In addition, one should take into account that the route of delivery has in itself a placebo effect. We found that in different RCTs [randomized controlled trials], the pooled placebo effect of IA saline is moderate to large.”
The fourth principle looks at ensuring that patients and clinicians make an informed and shared decision, which is again highlighted by the first recommendation. The fifth, and last, overarching principle acknowledges that IA injections may be given by a range of health care professionals.
Advice for before, during, and after injection
Patients need to be “fully informed of the nature of the procedure, the injectable used, and potential effects – benefits and risks – [and] informed consent should be obtained and documented,” said Dr. Usón, outlining the first recommendation. “That seems common,” she said in the interview, “but when we did the survey, we realize that many patients didn’t [give consent], and the doctors didn’t even ask for it. This is why it’s a very general statement, and it’s our first recommendation. The agreement was 99%!”
The recommendations also look at the optimal settings for performing injections, such as providing a professional and private, well-lighted room, and having a resuscitation kit nearby in case patients faint. Accuracy is important, Dr. Usón said, and imaging, such as ultrasound, should be used where available to ensure accurate injection into the joint. This is an area where further research could be performed, she said, urging young rheumatologists and health professionals to consider this. “Intra-articular therapy is something that you learn and do, but you never really investigate in it,” she said.
One recommendation states that when intra-articular injections are being given to pregnant patients, the safety of injected compound must be considered, both for the mother and for the fetus. There is another recommendation on the need to perform IA injections under aseptic conditions, and another stating that patients should be offered local anesthetics, after explaining the pros and cons.
Special populations of patients are also considered, Dr. Usón said. For example, the guidance advises warning patients with diabetes of the risk of transient glycemia after IA glucocorticoids and the need to monitor their blood glucose levels carefully for a couple of days afterward.
As a rule, “IAT is not a contraindication to people with clotting or bleeding disorders, or taking antithrombotic medications,” she said, unless they are at a high risk of bleeding.
Importantly, the recommendations cover when IAT can be performed after joint replacement surgery (after at least 3 months), and the need to “avoid overuse of injected joints” while also avoiding complete immobilization for at least 24 hours afterward. The recommendations very generally cover re-injections, but not how long intervals between injections should be. When asked about interval duration after her presentation, Dr. Usón said that the usual advice is to give IA injections no more than 2-3 times a year, but it depends on the injectable.
“It wasn’t our intention to review the efficacy and the safety of the different injectables, nor to review the use of IAT in different types of joint diseases,” she said. “We do lack a lot of information, a lot of evidence in this, and I really would hope that new rheumatologists start looking into and start investigating in this topic,” she added.
Recommendations will increase awareness of good clinical practice
“IA injections are commonly administered in the rheumatology setting. This is because [IA injection] is often a useful treatment for acute flare of arthritis, particularly when it is limited to a few joints,” observed Ai Lyn Tan, MD, associate professor and honorary consultant rheumatologist at the Leeds (England) Institute of Rheumatic and Musculoskeletal Medicine.
IA injection “also relieves symptoms relatively quickly for patients; however, the response can be variable, and there are side effects associated with IA injections,” Dr. Tan added in an interview.
There is a lack of universally accepted recommendations, Dr. Tan observed, noting that while there might be some local guidelines on how to safely perform IA injections these were often not standardized and were subject to being continually updated to try to improve the experience for patients.
“It is therefore timely to learn about the new EULAR recommendations for IA injections. The advantage of this will be to increase awareness of good clinical practice for performing IA injections.”
Dr. Tan had no relevant conflicts of interest.
SOURCE: EULAR COVID-19 Recommendations. E-congress content available until Sept. 1, 2020.
New EULAR recommendations for the intra-articular (IA) treatment of arthropathies aim to facilitate uniformity and quality of care for this mainstay of rheumatologic practice, according to a report on the new guidance that was presented at the annual European Congress of Rheumatology, held online this year due to COVID-19.
Until now there were no official recommendations on how best to use it in everyday practice. “This is the first time that there’s been a joint effort to develop evidence-based recommendations,” Jacqueline Usón, MD, PhD, associate professor medicine at Rey Juan Carlos University in Madrid, said in an interview. “Everything that we are saying is pretty logical, but it’s nice to see it put in recommendations based on evidence.”
IA therapy has been around for decades and is key for treating adults with a number of different conditions where synovitis, effusion, pain, or all three, are present, such as inflammatory arthritis and osteoarthritis, Dr. Usón observed during her presentation.
“Today, commonly used injectables are not only corticosteroids but also local anesthetics, hyaluronic acid, blood products, and maybe pharmaceuticals,” she said, adding that “there is a wide variation in the way intra-articular therapies are used and delivered to patients.” Health professionals also have very different views and habits depending on geographic locations and health care systems, she observed. Ironing out the variation was one of the main objectives of the recommendations.
As one of the two conveners of the EULAR task force behind the recommendations, Dr. Usón, herself a rheumatologist at University Hospital of Móstoles, pointed out that the task force brought together a range of specialties – rheumatologists, orthopedic surgeons, radiologists, nuclear medicine specialists, among others, as well as patients – to ensure that the best advice could be given.
The task force followed EULAR standard operating procedures for developing recommendations, with discussion groups, systematic literature reviews, and Delphi technique-based consensus all being employed. The literature search considered publications from 1946 up until 2019.
“We agreed on the need for more background information from health professionals and patients, so we developed two surveys: One for health professionals with 160 items, [for which] we obtained 186 responses from 26 countries; and the patient survey was made up of 44 items, translated into 10 different languages, and we obtained 200 responses,” she said.
The results of the systematic literature review and surveys were used to help form expert consensus, leading to 5 overarching principles and 11 recommendations that look at before, during, and after intra-articular therapy.
Five overarching principles
The first overarching principle recognizes the widespread use of IA therapies and that their use is specific to the disease that is being treated and “may not be interchangeable across indications,” Dr. Usón said. The second principle concerns improving patient-centered outcomes, which are “those that are relevant to the patient,” and include the benefits, harms, preferences, or implications for self-management.
“Contextual factors are important and contribute to the effect of IAT [intra-articular treatment],” she said, discussing the third principle. “These include effective communication, patient expectations, or settings [where the procedure takes place]. In addition, one should take into account that the route of delivery has in itself a placebo effect. We found that in different RCTs [randomized controlled trials], the pooled placebo effect of IA saline is moderate to large.”
The fourth principle looks at ensuring that patients and clinicians make an informed and shared decision, which is again highlighted by the first recommendation. The fifth, and last, overarching principle acknowledges that IA injections may be given by a range of health care professionals.
Advice for before, during, and after injection
Patients need to be “fully informed of the nature of the procedure, the injectable used, and potential effects – benefits and risks – [and] informed consent should be obtained and documented,” said Dr. Usón, outlining the first recommendation. “That seems common,” she said in the interview, “but when we did the survey, we realize that many patients didn’t [give consent], and the doctors didn’t even ask for it. This is why it’s a very general statement, and it’s our first recommendation. The agreement was 99%!”
The recommendations also look at the optimal settings for performing injections, such as providing a professional and private, well-lighted room, and having a resuscitation kit nearby in case patients faint. Accuracy is important, Dr. Usón said, and imaging, such as ultrasound, should be used where available to ensure accurate injection into the joint. This is an area where further research could be performed, she said, urging young rheumatologists and health professionals to consider this. “Intra-articular therapy is something that you learn and do, but you never really investigate in it,” she said.
One recommendation states that when intra-articular injections are being given to pregnant patients, the safety of injected compound must be considered, both for the mother and for the fetus. There is another recommendation on the need to perform IA injections under aseptic conditions, and another stating that patients should be offered local anesthetics, after explaining the pros and cons.
Special populations of patients are also considered, Dr. Usón said. For example, the guidance advises warning patients with diabetes of the risk of transient glycemia after IA glucocorticoids and the need to monitor their blood glucose levels carefully for a couple of days afterward.
As a rule, “IAT is not a contraindication to people with clotting or bleeding disorders, or taking antithrombotic medications,” she said, unless they are at a high risk of bleeding.
Importantly, the recommendations cover when IAT can be performed after joint replacement surgery (after at least 3 months), and the need to “avoid overuse of injected joints” while also avoiding complete immobilization for at least 24 hours afterward. The recommendations very generally cover re-injections, but not how long intervals between injections should be. When asked about interval duration after her presentation, Dr. Usón said that the usual advice is to give IA injections no more than 2-3 times a year, but it depends on the injectable.
“It wasn’t our intention to review the efficacy and the safety of the different injectables, nor to review the use of IAT in different types of joint diseases,” she said. “We do lack a lot of information, a lot of evidence in this, and I really would hope that new rheumatologists start looking into and start investigating in this topic,” she added.
Recommendations will increase awareness of good clinical practice
“IA injections are commonly administered in the rheumatology setting. This is because [IA injection] is often a useful treatment for acute flare of arthritis, particularly when it is limited to a few joints,” observed Ai Lyn Tan, MD, associate professor and honorary consultant rheumatologist at the Leeds (England) Institute of Rheumatic and Musculoskeletal Medicine.
IA injection “also relieves symptoms relatively quickly for patients; however, the response can be variable, and there are side effects associated with IA injections,” Dr. Tan added in an interview.
There is a lack of universally accepted recommendations, Dr. Tan observed, noting that while there might be some local guidelines on how to safely perform IA injections these were often not standardized and were subject to being continually updated to try to improve the experience for patients.
“It is therefore timely to learn about the new EULAR recommendations for IA injections. The advantage of this will be to increase awareness of good clinical practice for performing IA injections.”
Dr. Tan had no relevant conflicts of interest.
SOURCE: EULAR COVID-19 Recommendations. E-congress content available until Sept. 1, 2020.
New EULAR recommendations for the intra-articular (IA) treatment of arthropathies aim to facilitate uniformity and quality of care for this mainstay of rheumatologic practice, according to a report on the new guidance that was presented at the annual European Congress of Rheumatology, held online this year due to COVID-19.
Until now there were no official recommendations on how best to use it in everyday practice. “This is the first time that there’s been a joint effort to develop evidence-based recommendations,” Jacqueline Usón, MD, PhD, associate professor medicine at Rey Juan Carlos University in Madrid, said in an interview. “Everything that we are saying is pretty logical, but it’s nice to see it put in recommendations based on evidence.”
IA therapy has been around for decades and is key for treating adults with a number of different conditions where synovitis, effusion, pain, or all three, are present, such as inflammatory arthritis and osteoarthritis, Dr. Usón observed during her presentation.
“Today, commonly used injectables are not only corticosteroids but also local anesthetics, hyaluronic acid, blood products, and maybe pharmaceuticals,” she said, adding that “there is a wide variation in the way intra-articular therapies are used and delivered to patients.” Health professionals also have very different views and habits depending on geographic locations and health care systems, she observed. Ironing out the variation was one of the main objectives of the recommendations.
As one of the two conveners of the EULAR task force behind the recommendations, Dr. Usón, herself a rheumatologist at University Hospital of Móstoles, pointed out that the task force brought together a range of specialties – rheumatologists, orthopedic surgeons, radiologists, nuclear medicine specialists, among others, as well as patients – to ensure that the best advice could be given.
The task force followed EULAR standard operating procedures for developing recommendations, with discussion groups, systematic literature reviews, and Delphi technique-based consensus all being employed. The literature search considered publications from 1946 up until 2019.
“We agreed on the need for more background information from health professionals and patients, so we developed two surveys: One for health professionals with 160 items, [for which] we obtained 186 responses from 26 countries; and the patient survey was made up of 44 items, translated into 10 different languages, and we obtained 200 responses,” she said.
The results of the systematic literature review and surveys were used to help form expert consensus, leading to 5 overarching principles and 11 recommendations that look at before, during, and after intra-articular therapy.
Five overarching principles
The first overarching principle recognizes the widespread use of IA therapies and that their use is specific to the disease that is being treated and “may not be interchangeable across indications,” Dr. Usón said. The second principle concerns improving patient-centered outcomes, which are “those that are relevant to the patient,” and include the benefits, harms, preferences, or implications for self-management.
“Contextual factors are important and contribute to the effect of IAT [intra-articular treatment],” she said, discussing the third principle. “These include effective communication, patient expectations, or settings [where the procedure takes place]. In addition, one should take into account that the route of delivery has in itself a placebo effect. We found that in different RCTs [randomized controlled trials], the pooled placebo effect of IA saline is moderate to large.”
The fourth principle looks at ensuring that patients and clinicians make an informed and shared decision, which is again highlighted by the first recommendation. The fifth, and last, overarching principle acknowledges that IA injections may be given by a range of health care professionals.
Advice for before, during, and after injection
Patients need to be “fully informed of the nature of the procedure, the injectable used, and potential effects – benefits and risks – [and] informed consent should be obtained and documented,” said Dr. Usón, outlining the first recommendation. “That seems common,” she said in the interview, “but when we did the survey, we realize that many patients didn’t [give consent], and the doctors didn’t even ask for it. This is why it’s a very general statement, and it’s our first recommendation. The agreement was 99%!”
The recommendations also look at the optimal settings for performing injections, such as providing a professional and private, well-lighted room, and having a resuscitation kit nearby in case patients faint. Accuracy is important, Dr. Usón said, and imaging, such as ultrasound, should be used where available to ensure accurate injection into the joint. This is an area where further research could be performed, she said, urging young rheumatologists and health professionals to consider this. “Intra-articular therapy is something that you learn and do, but you never really investigate in it,” she said.
One recommendation states that when intra-articular injections are being given to pregnant patients, the safety of injected compound must be considered, both for the mother and for the fetus. There is another recommendation on the need to perform IA injections under aseptic conditions, and another stating that patients should be offered local anesthetics, after explaining the pros and cons.
Special populations of patients are also considered, Dr. Usón said. For example, the guidance advises warning patients with diabetes of the risk of transient glycemia after IA glucocorticoids and the need to monitor their blood glucose levels carefully for a couple of days afterward.
As a rule, “IAT is not a contraindication to people with clotting or bleeding disorders, or taking antithrombotic medications,” she said, unless they are at a high risk of bleeding.
Importantly, the recommendations cover when IAT can be performed after joint replacement surgery (after at least 3 months), and the need to “avoid overuse of injected joints” while also avoiding complete immobilization for at least 24 hours afterward. The recommendations very generally cover re-injections, but not how long intervals between injections should be. When asked about interval duration after her presentation, Dr. Usón said that the usual advice is to give IA injections no more than 2-3 times a year, but it depends on the injectable.
“It wasn’t our intention to review the efficacy and the safety of the different injectables, nor to review the use of IAT in different types of joint diseases,” she said. “We do lack a lot of information, a lot of evidence in this, and I really would hope that new rheumatologists start looking into and start investigating in this topic,” she added.
Recommendations will increase awareness of good clinical practice
“IA injections are commonly administered in the rheumatology setting. This is because [IA injection] is often a useful treatment for acute flare of arthritis, particularly when it is limited to a few joints,” observed Ai Lyn Tan, MD, associate professor and honorary consultant rheumatologist at the Leeds (England) Institute of Rheumatic and Musculoskeletal Medicine.
IA injection “also relieves symptoms relatively quickly for patients; however, the response can be variable, and there are side effects associated with IA injections,” Dr. Tan added in an interview.
There is a lack of universally accepted recommendations, Dr. Tan observed, noting that while there might be some local guidelines on how to safely perform IA injections these were often not standardized and were subject to being continually updated to try to improve the experience for patients.
“It is therefore timely to learn about the new EULAR recommendations for IA injections. The advantage of this will be to increase awareness of good clinical practice for performing IA injections.”
Dr. Tan had no relevant conflicts of interest.
SOURCE: EULAR COVID-19 Recommendations. E-congress content available until Sept. 1, 2020.
FROM THE EULAR 2020 E-CONGRESS
New registry focuses on rheumatic immune-related AEs of cancer therapy
Its first findings were reported at the annual European Congress of Rheumatology, held online this year due to COVID-19.
“We have limited knowledge on the interrelationships between malignant and rheumatic diseases on both the clinical and molecular level, and we have a large unmet need for management guidelines in the case of the coincidence of both disease entities,” noted lead author Karolina Benesova, MD, of the department of hematology, oncology, and rheumatology at University Hospital Heidelberg (Germany).
The TRheuMa registry – Therapy-Induced Rheumatic Symptoms in Patients with Malignancy – is one of three registries in a multicenter observational project exploring various contexts between malignant and rheumatic diseases. Over its first 22 months, the registry recruited 69 patients having rheumatic symptoms as a result of immune checkpoint inhibitor therapy or other cancer therapies.
Registry findings
The largest shares of patients had non–small cell lung cancer (38%) or melanoma (33%), Dr. Benesova reported. The immune checkpoint inhibitors most commonly received were pembrolizumab (Keytruda), nivolumab (Opdivo), and ipilimumab (Yervoy).
The immune-related adverse events usually presented with symptoms of de novo spondyloarthritis or psoriatic arthritis (42%), late-onset RA (17%), or polymyalgia rheumatica (14%). But 16% of the patients were experiencing a flare of a preexisting rheumatic and musculoskeletal disease.
Laboratory findings differed somewhat from those of classical rheumatic and musculoskeletal diseases, according to Dr. Benesova. Specific findings were rare; in particular, most patients did not have detectable autoantibodies. However, 76% had an elevated C-reactive protein level and 39% had an elevated soluble CD25 level. In addition, nearly all patients (96%) undergoing joint ultrasound had pathologic findings.
“Based on our experiences from interdisciplinary care together with our local oncologists, we have developed a therapeutic algorithm for rheumatic immune-related adverse events,” she reported, noting that the algorithm is consistent with recently published recommendations in this area.
The large majority of patients were adequately treated with prednisone at a dose greater than 10 mg (40%) or at a dose of 10 mg or less with or without an NSAID (40%), while some received NSAID monotherapy (14%).
“We have a growing proportion of patients on conventional or biological [disease-modifying antirheumatic drugs],” Dr. Benesova noted. “These are mostly patients with preexisting rheumatic and musculoskeletal disease or highly suspected de novo classical rheumatic and musculoskeletal disease under checkpoint inhibitor therapy.”
Patients with melanoma having a rheumatic immune-related adverse event had a better response to their therapy than historical counterparts who did not have such events: 39% of the former had a complete response, relative to merely 4% of the latter.
Only a small proportion of patients overall (9%) had to discontinue immune checkpoint inhibitor therapy because of their adverse event, and some of them may be eligible for rechallenge if their cancer progresses, Dr. Benesova noted.
“There is still a lot to be done,” she stated, such as better elucidating the nature of these adverse events [whether transient side effects or a triggering of chronic rheumatic and musculoskeletal diseases], the need for a defensive treatment strategy, and the advisability of closer monitoring of high-risk patients given immune checkpoint inhibitors. “We are aiming at solving these questions in the next few years,” she concluded.
Findings in context
“Registries are important to gain prospective data on patient outcomes,” Sabina Sandigursky, MD, an instructor in the division of rheumatology at the Laura and Isaac Perlmutter Cancer Center at New York University, commented in an interview. “One must be careful, while interpreting these data, especially since they are not randomized, controlled trials.”
Patterns may differ at other centers, too, she pointed out. “The German registry reported a predominance of spondyloarthritis-like disease; however, our patients have a predominance of small-joint involvement. It is unclear what accounts for this difference.”
Individual institutions in North America are similarly collecting data on this patient population, with efforts underway to compile those data to provide a larger picture, according to Dr. Sandigursky.
“Many of the syndromes that we consider to be rheumatic immune-related adverse events have been well described by groups from the U.S., Canada, Australia, and European Union,” she concluded. “From this registry, we can observe how patients are being treated in real time since this information is largely consensus based.”
The study did not receive any specific funding. Dr. Benesova disclosed grant/research support from AbbVie, Novartis, Rheumaliga Baden-Wurttemberg, and the University of Heidelberg, and consultancies, speaker fees, and/or travel reimbursements from AbbVie, Bristol-Myers Squibb, Gilead, Janssen, Medac, Merck Sharp & Dohme, Mundipharma, Novartis, Pfizer, Roche, and UCB. Some of her coauthors also disclosed financial relationships with industry. Dr. Sandigursky disclosed having no relevant conflicts of interest.
SOURCE: Benesova K et al. Ann Rheum Dis 2020;79[suppl 1]:168-9, Abstract OP0270.
Its first findings were reported at the annual European Congress of Rheumatology, held online this year due to COVID-19.
“We have limited knowledge on the interrelationships between malignant and rheumatic diseases on both the clinical and molecular level, and we have a large unmet need for management guidelines in the case of the coincidence of both disease entities,” noted lead author Karolina Benesova, MD, of the department of hematology, oncology, and rheumatology at University Hospital Heidelberg (Germany).
The TRheuMa registry – Therapy-Induced Rheumatic Symptoms in Patients with Malignancy – is one of three registries in a multicenter observational project exploring various contexts between malignant and rheumatic diseases. Over its first 22 months, the registry recruited 69 patients having rheumatic symptoms as a result of immune checkpoint inhibitor therapy or other cancer therapies.
Registry findings
The largest shares of patients had non–small cell lung cancer (38%) or melanoma (33%), Dr. Benesova reported. The immune checkpoint inhibitors most commonly received were pembrolizumab (Keytruda), nivolumab (Opdivo), and ipilimumab (Yervoy).
The immune-related adverse events usually presented with symptoms of de novo spondyloarthritis or psoriatic arthritis (42%), late-onset RA (17%), or polymyalgia rheumatica (14%). But 16% of the patients were experiencing a flare of a preexisting rheumatic and musculoskeletal disease.
Laboratory findings differed somewhat from those of classical rheumatic and musculoskeletal diseases, according to Dr. Benesova. Specific findings were rare; in particular, most patients did not have detectable autoantibodies. However, 76% had an elevated C-reactive protein level and 39% had an elevated soluble CD25 level. In addition, nearly all patients (96%) undergoing joint ultrasound had pathologic findings.
“Based on our experiences from interdisciplinary care together with our local oncologists, we have developed a therapeutic algorithm for rheumatic immune-related adverse events,” she reported, noting that the algorithm is consistent with recently published recommendations in this area.
The large majority of patients were adequately treated with prednisone at a dose greater than 10 mg (40%) or at a dose of 10 mg or less with or without an NSAID (40%), while some received NSAID monotherapy (14%).
“We have a growing proportion of patients on conventional or biological [disease-modifying antirheumatic drugs],” Dr. Benesova noted. “These are mostly patients with preexisting rheumatic and musculoskeletal disease or highly suspected de novo classical rheumatic and musculoskeletal disease under checkpoint inhibitor therapy.”
Patients with melanoma having a rheumatic immune-related adverse event had a better response to their therapy than historical counterparts who did not have such events: 39% of the former had a complete response, relative to merely 4% of the latter.
Only a small proportion of patients overall (9%) had to discontinue immune checkpoint inhibitor therapy because of their adverse event, and some of them may be eligible for rechallenge if their cancer progresses, Dr. Benesova noted.
“There is still a lot to be done,” she stated, such as better elucidating the nature of these adverse events [whether transient side effects or a triggering of chronic rheumatic and musculoskeletal diseases], the need for a defensive treatment strategy, and the advisability of closer monitoring of high-risk patients given immune checkpoint inhibitors. “We are aiming at solving these questions in the next few years,” she concluded.
Findings in context
“Registries are important to gain prospective data on patient outcomes,” Sabina Sandigursky, MD, an instructor in the division of rheumatology at the Laura and Isaac Perlmutter Cancer Center at New York University, commented in an interview. “One must be careful, while interpreting these data, especially since they are not randomized, controlled trials.”
Patterns may differ at other centers, too, she pointed out. “The German registry reported a predominance of spondyloarthritis-like disease; however, our patients have a predominance of small-joint involvement. It is unclear what accounts for this difference.”
Individual institutions in North America are similarly collecting data on this patient population, with efforts underway to compile those data to provide a larger picture, according to Dr. Sandigursky.
“Many of the syndromes that we consider to be rheumatic immune-related adverse events have been well described by groups from the U.S., Canada, Australia, and European Union,” she concluded. “From this registry, we can observe how patients are being treated in real time since this information is largely consensus based.”
The study did not receive any specific funding. Dr. Benesova disclosed grant/research support from AbbVie, Novartis, Rheumaliga Baden-Wurttemberg, and the University of Heidelberg, and consultancies, speaker fees, and/or travel reimbursements from AbbVie, Bristol-Myers Squibb, Gilead, Janssen, Medac, Merck Sharp & Dohme, Mundipharma, Novartis, Pfizer, Roche, and UCB. Some of her coauthors also disclosed financial relationships with industry. Dr. Sandigursky disclosed having no relevant conflicts of interest.
SOURCE: Benesova K et al. Ann Rheum Dis 2020;79[suppl 1]:168-9, Abstract OP0270.
Its first findings were reported at the annual European Congress of Rheumatology, held online this year due to COVID-19.
“We have limited knowledge on the interrelationships between malignant and rheumatic diseases on both the clinical and molecular level, and we have a large unmet need for management guidelines in the case of the coincidence of both disease entities,” noted lead author Karolina Benesova, MD, of the department of hematology, oncology, and rheumatology at University Hospital Heidelberg (Germany).
The TRheuMa registry – Therapy-Induced Rheumatic Symptoms in Patients with Malignancy – is one of three registries in a multicenter observational project exploring various contexts between malignant and rheumatic diseases. Over its first 22 months, the registry recruited 69 patients having rheumatic symptoms as a result of immune checkpoint inhibitor therapy or other cancer therapies.
Registry findings
The largest shares of patients had non–small cell lung cancer (38%) or melanoma (33%), Dr. Benesova reported. The immune checkpoint inhibitors most commonly received were pembrolizumab (Keytruda), nivolumab (Opdivo), and ipilimumab (Yervoy).
The immune-related adverse events usually presented with symptoms of de novo spondyloarthritis or psoriatic arthritis (42%), late-onset RA (17%), or polymyalgia rheumatica (14%). But 16% of the patients were experiencing a flare of a preexisting rheumatic and musculoskeletal disease.
Laboratory findings differed somewhat from those of classical rheumatic and musculoskeletal diseases, according to Dr. Benesova. Specific findings were rare; in particular, most patients did not have detectable autoantibodies. However, 76% had an elevated C-reactive protein level and 39% had an elevated soluble CD25 level. In addition, nearly all patients (96%) undergoing joint ultrasound had pathologic findings.
“Based on our experiences from interdisciplinary care together with our local oncologists, we have developed a therapeutic algorithm for rheumatic immune-related adverse events,” she reported, noting that the algorithm is consistent with recently published recommendations in this area.
The large majority of patients were adequately treated with prednisone at a dose greater than 10 mg (40%) or at a dose of 10 mg or less with or without an NSAID (40%), while some received NSAID monotherapy (14%).
“We have a growing proportion of patients on conventional or biological [disease-modifying antirheumatic drugs],” Dr. Benesova noted. “These are mostly patients with preexisting rheumatic and musculoskeletal disease or highly suspected de novo classical rheumatic and musculoskeletal disease under checkpoint inhibitor therapy.”
Patients with melanoma having a rheumatic immune-related adverse event had a better response to their therapy than historical counterparts who did not have such events: 39% of the former had a complete response, relative to merely 4% of the latter.
Only a small proportion of patients overall (9%) had to discontinue immune checkpoint inhibitor therapy because of their adverse event, and some of them may be eligible for rechallenge if their cancer progresses, Dr. Benesova noted.
“There is still a lot to be done,” she stated, such as better elucidating the nature of these adverse events [whether transient side effects or a triggering of chronic rheumatic and musculoskeletal diseases], the need for a defensive treatment strategy, and the advisability of closer monitoring of high-risk patients given immune checkpoint inhibitors. “We are aiming at solving these questions in the next few years,” she concluded.
Findings in context
“Registries are important to gain prospective data on patient outcomes,” Sabina Sandigursky, MD, an instructor in the division of rheumatology at the Laura and Isaac Perlmutter Cancer Center at New York University, commented in an interview. “One must be careful, while interpreting these data, especially since they are not randomized, controlled trials.”
Patterns may differ at other centers, too, she pointed out. “The German registry reported a predominance of spondyloarthritis-like disease; however, our patients have a predominance of small-joint involvement. It is unclear what accounts for this difference.”
Individual institutions in North America are similarly collecting data on this patient population, with efforts underway to compile those data to provide a larger picture, according to Dr. Sandigursky.
“Many of the syndromes that we consider to be rheumatic immune-related adverse events have been well described by groups from the U.S., Canada, Australia, and European Union,” she concluded. “From this registry, we can observe how patients are being treated in real time since this information is largely consensus based.”
The study did not receive any specific funding. Dr. Benesova disclosed grant/research support from AbbVie, Novartis, Rheumaliga Baden-Wurttemberg, and the University of Heidelberg, and consultancies, speaker fees, and/or travel reimbursements from AbbVie, Bristol-Myers Squibb, Gilead, Janssen, Medac, Merck Sharp & Dohme, Mundipharma, Novartis, Pfizer, Roche, and UCB. Some of her coauthors also disclosed financial relationships with industry. Dr. Sandigursky disclosed having no relevant conflicts of interest.
SOURCE: Benesova K et al. Ann Rheum Dis 2020;79[suppl 1]:168-9, Abstract OP0270.
FROM THE EULAR 2020 E-CONGRESS
Seropositivity in RA linked with doubled pneumonia incidence
from a single U.S. medical system.
“Patients with seropositive RA, particularly RF [rheumatoid factor]-positive RA, had increased risk for pneumonia throughout the RA disease course that was not explained by measured confounders, including smoking status, multimorbidity, medications, and [erythrocyte sedimentation rate] level,” Jeffrey A. Sparks, MD, said at the annual European Congress of Rheumatology, held online this year due to COVID-19.
“There has been much interest about the relationship between lung inflammation and the generation of RF and CCP [cyclic citrullinated protein] prior to the onset of RA. We hypothesized that patients with seropositive RA might have subclinical lung injury that could predispose them to pneumonia after clinical RA onset,” Dr. Sparks said in an interview. “Pneumonia is one of the most common serious infections in both patients with RA and the general population, and it causes serious morbidity and mortality.”
The doubled relative risk for pneumonia seen in the findings “translates into a clinically meaningful finding when considering the high rate and the many patients at risk since RA is relatively common,” said Dr. Sparks, a rheumatologist at Brigham and Women’s Hospital in Boston.
“Patients with RF-positive RA who present with symptoms concerning for pneumonia should be evaluated carefully for this and for other possible pulmonary manifestations of RA. Vaccination for pneumonia should be strongly considered for patients with RA who are on disease-modifying antirheumatic drugs, and we hope that our report encourages clinicians and patients” to undertake vaccination, he said.
His study used a database of more than 60,000 patients diagnosed with RA as of November 2013 in the records of a large Boston-area medical system that includes physicians affiliated with Brigham and Women’s Hospital and Massachusetts General Hospital. The researchers applied a validated algorithm for calculating a patient’s probability of having RA, and at the level of 97% probability they narrowed the cohort down to just under 10,000 patients. Additional winnowing because of missing data or a history of pneumonia yielded a study group of 4,110, which included 3,279 (80%) who were seropositive for either or both CCP and RF, and 831 (20%) who were seronegative. During a median follow-up of 7.8 years and total follow-up of more than 32,000 patient-years, the overall pneumonia incidence was 5.8%, with a 2.8% rate among the seronegatives and a 6.6% rate among seropositives. After adjustment for age, sex, glucocorticoid use, disease-modifying antirheumatic drug use, and several other possible confounders, the researchers calculated a 99% relative increased rate of pneumonia among all seropositive patients, compared with the seronegatives.
Further analysis looked at pneumonia incidence rates among patients positive only for CCP antibody, positive only for RF antibody, or both, compared with seronegative patients. This showed that CCP seropositivity had no statistically significant link with incident pneumonia, while RF seropositivity linked with a statistically significant, roughly twofold higher rate. Only 6% of all seropositive patients were positive only for CCP antibody, 59% were positive specifically for RF antibody, and 35% for both.
The data Dr. Sparks presented did not include information on pneumonia type, the timing of the pneumonia, compared with the onset of RA, disease activity, or smoking intensity.
“We anticipated that both RF positive and CCP positive would each be associated with pneumonia, so it was somewhat surprising that we only detected this for RF,” Dr. Sparks said. But he added that, because the number of patients with only CCP positivity was relatively so small, “it is still possible that CCP [antibody] could also increase pneumonia risk.”
The study had no commercial funding. Dr. Sparks had no disclosures.
SOURCE: Sparks JA et al. Ann Rheum Dis. 2020 Jun;79[suppl 1]:73, Abstract OP0111.
from a single U.S. medical system.
“Patients with seropositive RA, particularly RF [rheumatoid factor]-positive RA, had increased risk for pneumonia throughout the RA disease course that was not explained by measured confounders, including smoking status, multimorbidity, medications, and [erythrocyte sedimentation rate] level,” Jeffrey A. Sparks, MD, said at the annual European Congress of Rheumatology, held online this year due to COVID-19.
“There has been much interest about the relationship between lung inflammation and the generation of RF and CCP [cyclic citrullinated protein] prior to the onset of RA. We hypothesized that patients with seropositive RA might have subclinical lung injury that could predispose them to pneumonia after clinical RA onset,” Dr. Sparks said in an interview. “Pneumonia is one of the most common serious infections in both patients with RA and the general population, and it causes serious morbidity and mortality.”
The doubled relative risk for pneumonia seen in the findings “translates into a clinically meaningful finding when considering the high rate and the many patients at risk since RA is relatively common,” said Dr. Sparks, a rheumatologist at Brigham and Women’s Hospital in Boston.
“Patients with RF-positive RA who present with symptoms concerning for pneumonia should be evaluated carefully for this and for other possible pulmonary manifestations of RA. Vaccination for pneumonia should be strongly considered for patients with RA who are on disease-modifying antirheumatic drugs, and we hope that our report encourages clinicians and patients” to undertake vaccination, he said.
His study used a database of more than 60,000 patients diagnosed with RA as of November 2013 in the records of a large Boston-area medical system that includes physicians affiliated with Brigham and Women’s Hospital and Massachusetts General Hospital. The researchers applied a validated algorithm for calculating a patient’s probability of having RA, and at the level of 97% probability they narrowed the cohort down to just under 10,000 patients. Additional winnowing because of missing data or a history of pneumonia yielded a study group of 4,110, which included 3,279 (80%) who were seropositive for either or both CCP and RF, and 831 (20%) who were seronegative. During a median follow-up of 7.8 years and total follow-up of more than 32,000 patient-years, the overall pneumonia incidence was 5.8%, with a 2.8% rate among the seronegatives and a 6.6% rate among seropositives. After adjustment for age, sex, glucocorticoid use, disease-modifying antirheumatic drug use, and several other possible confounders, the researchers calculated a 99% relative increased rate of pneumonia among all seropositive patients, compared with the seronegatives.
Further analysis looked at pneumonia incidence rates among patients positive only for CCP antibody, positive only for RF antibody, or both, compared with seronegative patients. This showed that CCP seropositivity had no statistically significant link with incident pneumonia, while RF seropositivity linked with a statistically significant, roughly twofold higher rate. Only 6% of all seropositive patients were positive only for CCP antibody, 59% were positive specifically for RF antibody, and 35% for both.
The data Dr. Sparks presented did not include information on pneumonia type, the timing of the pneumonia, compared with the onset of RA, disease activity, or smoking intensity.
“We anticipated that both RF positive and CCP positive would each be associated with pneumonia, so it was somewhat surprising that we only detected this for RF,” Dr. Sparks said. But he added that, because the number of patients with only CCP positivity was relatively so small, “it is still possible that CCP [antibody] could also increase pneumonia risk.”
The study had no commercial funding. Dr. Sparks had no disclosures.
SOURCE: Sparks JA et al. Ann Rheum Dis. 2020 Jun;79[suppl 1]:73, Abstract OP0111.
from a single U.S. medical system.
“Patients with seropositive RA, particularly RF [rheumatoid factor]-positive RA, had increased risk for pneumonia throughout the RA disease course that was not explained by measured confounders, including smoking status, multimorbidity, medications, and [erythrocyte sedimentation rate] level,” Jeffrey A. Sparks, MD, said at the annual European Congress of Rheumatology, held online this year due to COVID-19.
“There has been much interest about the relationship between lung inflammation and the generation of RF and CCP [cyclic citrullinated protein] prior to the onset of RA. We hypothesized that patients with seropositive RA might have subclinical lung injury that could predispose them to pneumonia after clinical RA onset,” Dr. Sparks said in an interview. “Pneumonia is one of the most common serious infections in both patients with RA and the general population, and it causes serious morbidity and mortality.”
The doubled relative risk for pneumonia seen in the findings “translates into a clinically meaningful finding when considering the high rate and the many patients at risk since RA is relatively common,” said Dr. Sparks, a rheumatologist at Brigham and Women’s Hospital in Boston.
“Patients with RF-positive RA who present with symptoms concerning for pneumonia should be evaluated carefully for this and for other possible pulmonary manifestations of RA. Vaccination for pneumonia should be strongly considered for patients with RA who are on disease-modifying antirheumatic drugs, and we hope that our report encourages clinicians and patients” to undertake vaccination, he said.
His study used a database of more than 60,000 patients diagnosed with RA as of November 2013 in the records of a large Boston-area medical system that includes physicians affiliated with Brigham and Women’s Hospital and Massachusetts General Hospital. The researchers applied a validated algorithm for calculating a patient’s probability of having RA, and at the level of 97% probability they narrowed the cohort down to just under 10,000 patients. Additional winnowing because of missing data or a history of pneumonia yielded a study group of 4,110, which included 3,279 (80%) who were seropositive for either or both CCP and RF, and 831 (20%) who were seronegative. During a median follow-up of 7.8 years and total follow-up of more than 32,000 patient-years, the overall pneumonia incidence was 5.8%, with a 2.8% rate among the seronegatives and a 6.6% rate among seropositives. After adjustment for age, sex, glucocorticoid use, disease-modifying antirheumatic drug use, and several other possible confounders, the researchers calculated a 99% relative increased rate of pneumonia among all seropositive patients, compared with the seronegatives.
Further analysis looked at pneumonia incidence rates among patients positive only for CCP antibody, positive only for RF antibody, or both, compared with seronegative patients. This showed that CCP seropositivity had no statistically significant link with incident pneumonia, while RF seropositivity linked with a statistically significant, roughly twofold higher rate. Only 6% of all seropositive patients were positive only for CCP antibody, 59% were positive specifically for RF antibody, and 35% for both.
The data Dr. Sparks presented did not include information on pneumonia type, the timing of the pneumonia, compared with the onset of RA, disease activity, or smoking intensity.
“We anticipated that both RF positive and CCP positive would each be associated with pneumonia, so it was somewhat surprising that we only detected this for RF,” Dr. Sparks said. But he added that, because the number of patients with only CCP positivity was relatively so small, “it is still possible that CCP [antibody] could also increase pneumonia risk.”
The study had no commercial funding. Dr. Sparks had no disclosures.
SOURCE: Sparks JA et al. Ann Rheum Dis. 2020 Jun;79[suppl 1]:73, Abstract OP0111.
FROM THE EULAR 2020 E-CONGRESS
EULAR’s COVID-19 recommendations offer no surprises
As might be expected, the “EULAR [European League Against Rheumatism] provisional recommendations for the management of rheumatic and musculoskeletal diseases [RMDs] in the context of SARS-CoV-2” concur with much of the guidance already released on how best to manage patients during the current pandemic.
Highlights of the five overarching principles are that, contrary to earlier expectations, “there is no indication that patients with RMDs have an additional, or have a higher, risk of contracting the virus, or that they fare a worse course” than the general population, said the task force convener Robert Landewé, MD, PhD, professor of rheumatology at the University of Amsterdam.
“The second pertinent highlight is that, when it comes to managerial discussions, whether or not to stop or to start treatment for RMDs, rheumatologists should definitely be involved,” Dr. Landewé said during a live session at the annual European Congress of Rheumatology, held online this year due to COVID-19. “In practice, something that happens very often is that immunosuppressive drugs are stopped by medical specialists involved in the care of COVID but without any expertise in treating patients with rheumatic diseases. We should try to avoid that situation.”
The third highlight, something many rheumatologists may already be well aware of, is that rheumatology drugs are being used to treat COVID-19 patients without RMDs and a shortage of disease-modifying antirheumatic drugs (DMARDs) agents is a real possibility. As such, the fifth overarching highlight states that the availability of both synthetic and biologic DMARDs is “a delicate societal responsibility” and that “the off-label use of DMARDs in COVID-19 outside the context of clinical trials should be discouraged.”
The EULAR recommendation are now published online in Annals of the Rheumatic Diseases and they are “what you could call an unprecedented set of recommendations,” Dr. Landewé said. “We have never done this before,” he added, referring to the speed and way in which they had to be put together, remotely, and with little scientific evidence currently available. “Three months ago we hadn’t even heard about the virus.”
From the first patient being identified in the Hubei province of China in November 2019, to the first U.S. patient in the state of Washington on Jan. 20, 2020, and to the first European patient identified a little over 10 days later, the COVID-19 pandemic has taken the world by storm. It was only declared a pandemic on March 11, 2020, however, and Dr. Landewé noted that the response to the pandemic had been very variable – some countries locking down their borders early, while others took their time to make an appropriate response, if at all.
The rheumatology community was particularly concerned, Dr. Landewé said, because people with autoimmune diseases who were taking immunosuppressant drugs might be at higher risk for becoming infected with SARS-CoV-2, and may be at higher risk than others for a worse disease course. Thankfully, that seems not to be the case according to data that are emerging from new registries that have been set up, including EULAR’s own COVID-19 registry.
There are 13 recommendations that cover 4 themes: general measures and prevention of SARS-CoV-2 infection; the management of RMD patients during the pandemic; the management of RMD patients who have COVID-19; and the prevention of other pulmonary infections in RMD patients.
Highlighting the first three general recommendations, Dr. Landewé said: “Follow the regular guidelines in your country; if a patient with RMD does not have symptoms of COVID-19, simply continue RMD treatments,” albeit with a couple of exceptions.
The next four recommendation highlights are to avoid visits to the hospital or to the office; use remote monitoring via the telephone, for example; and if visits cannot be avoided, then take appropriate precautions. Finally, if you suspect a patient has COVID-19, do a test.
If patients test positive, then the next four recommendations cover what to do, such as continuing use of RMD treatments, but in the case of glucocorticoids this should be the lowest possible dose necessary. There is no consensus on what to do in cases of mild symptoms; the recommendation is to “decide on a case-by-case basis,” said Dr. Landewé. If a patient’s symptoms worsen, then “seek expert advice immediately and follow local treatment recommendations. The rheumatologist is not the expert to treat COVID-19,” he added. That responsibility lies with the pulmonologist, infectious disease specialist, or maybe the intensive care specialist, depending on local situations.
On the whole, the EULAR recommendations are pretty similar to those already released by the American College of Rheumatology, said Ted Mikuls, MD, of the University of Nebraska Medical Center, Omaha. The ACR recommendations are “slightly more prescriptive”, he suggested, with 25 final guidance statements. For example, general statements focused not only on the use of glucocorticoids, but also other medicines, such as antihypertensives.
“There’s really not a [lot of], I would say, major differences in the two efforts and that’s ... somewhat reassuring that we’re approaching the unknown from very different parts of the world, and driving in a very similar place,” commented Dr. Mikuls, who is a member of the ACR COVID-19 recommendations task force.
“I think one of the very important similarities that I would highlight is that, in the absence of known exposure, in the absence of COVID-19 infection, our panel felt very strongly about the importance of continuing rheumatic disease treatments,” Dr. Mikuls observed. The ACR guidelines also touch upon societal perspectives, including “some statements that were made very specific to lupus, and the use of antimalarials, given supply chain issues that we have encountered.”
Dr. Mikuls also said that the American recommendations emphasized that “you really have to manage active inflammatory rheumatic disease. Even in the context of the COVID-19 pandemic, given what we saw as the potential risk of unchecked inflammation and unchecked rheumatic disease.”
One notable difference, however, is that the European recommendations advise on immunizations and pneumonia prophylaxis, saying that all patients without COVID-19 symptoms should make sure they are up to date with any recommended vaccinations, “with a particular focus on pneumococcal and influenza vaccinations,” Dr. Landewé said.
Another difference is that the ACR recommendations are a living document and could potentially be updated monthly if the evidence arrives to allow that. In that sense, the American guidance is more agile, with EULAR expecting to update its recommendations every 3 months.
“The current evidence is extremely sparse and fragmented,” Dr. Landewé said. “We, as a task force are essentially flying blindly. We also have to cover many jurisdictions within Europe, with many conflicting opinions. So the last word to say is that updates are truly necessary, but we have to wait a while.”
SOURCE: Landewé RB et al. Ann Rheum Dis. 2020 Jun 5. doi: 10.1136/annrheumdis-2020-217877.
As might be expected, the “EULAR [European League Against Rheumatism] provisional recommendations for the management of rheumatic and musculoskeletal diseases [RMDs] in the context of SARS-CoV-2” concur with much of the guidance already released on how best to manage patients during the current pandemic.
Highlights of the five overarching principles are that, contrary to earlier expectations, “there is no indication that patients with RMDs have an additional, or have a higher, risk of contracting the virus, or that they fare a worse course” than the general population, said the task force convener Robert Landewé, MD, PhD, professor of rheumatology at the University of Amsterdam.
“The second pertinent highlight is that, when it comes to managerial discussions, whether or not to stop or to start treatment for RMDs, rheumatologists should definitely be involved,” Dr. Landewé said during a live session at the annual European Congress of Rheumatology, held online this year due to COVID-19. “In practice, something that happens very often is that immunosuppressive drugs are stopped by medical specialists involved in the care of COVID but without any expertise in treating patients with rheumatic diseases. We should try to avoid that situation.”
The third highlight, something many rheumatologists may already be well aware of, is that rheumatology drugs are being used to treat COVID-19 patients without RMDs and a shortage of disease-modifying antirheumatic drugs (DMARDs) agents is a real possibility. As such, the fifth overarching highlight states that the availability of both synthetic and biologic DMARDs is “a delicate societal responsibility” and that “the off-label use of DMARDs in COVID-19 outside the context of clinical trials should be discouraged.”
The EULAR recommendation are now published online in Annals of the Rheumatic Diseases and they are “what you could call an unprecedented set of recommendations,” Dr. Landewé said. “We have never done this before,” he added, referring to the speed and way in which they had to be put together, remotely, and with little scientific evidence currently available. “Three months ago we hadn’t even heard about the virus.”
From the first patient being identified in the Hubei province of China in November 2019, to the first U.S. patient in the state of Washington on Jan. 20, 2020, and to the first European patient identified a little over 10 days later, the COVID-19 pandemic has taken the world by storm. It was only declared a pandemic on March 11, 2020, however, and Dr. Landewé noted that the response to the pandemic had been very variable – some countries locking down their borders early, while others took their time to make an appropriate response, if at all.
The rheumatology community was particularly concerned, Dr. Landewé said, because people with autoimmune diseases who were taking immunosuppressant drugs might be at higher risk for becoming infected with SARS-CoV-2, and may be at higher risk than others for a worse disease course. Thankfully, that seems not to be the case according to data that are emerging from new registries that have been set up, including EULAR’s own COVID-19 registry.
There are 13 recommendations that cover 4 themes: general measures and prevention of SARS-CoV-2 infection; the management of RMD patients during the pandemic; the management of RMD patients who have COVID-19; and the prevention of other pulmonary infections in RMD patients.
Highlighting the first three general recommendations, Dr. Landewé said: “Follow the regular guidelines in your country; if a patient with RMD does not have symptoms of COVID-19, simply continue RMD treatments,” albeit with a couple of exceptions.
The next four recommendation highlights are to avoid visits to the hospital or to the office; use remote monitoring via the telephone, for example; and if visits cannot be avoided, then take appropriate precautions. Finally, if you suspect a patient has COVID-19, do a test.
If patients test positive, then the next four recommendations cover what to do, such as continuing use of RMD treatments, but in the case of glucocorticoids this should be the lowest possible dose necessary. There is no consensus on what to do in cases of mild symptoms; the recommendation is to “decide on a case-by-case basis,” said Dr. Landewé. If a patient’s symptoms worsen, then “seek expert advice immediately and follow local treatment recommendations. The rheumatologist is not the expert to treat COVID-19,” he added. That responsibility lies with the pulmonologist, infectious disease specialist, or maybe the intensive care specialist, depending on local situations.
On the whole, the EULAR recommendations are pretty similar to those already released by the American College of Rheumatology, said Ted Mikuls, MD, of the University of Nebraska Medical Center, Omaha. The ACR recommendations are “slightly more prescriptive”, he suggested, with 25 final guidance statements. For example, general statements focused not only on the use of glucocorticoids, but also other medicines, such as antihypertensives.
“There’s really not a [lot of], I would say, major differences in the two efforts and that’s ... somewhat reassuring that we’re approaching the unknown from very different parts of the world, and driving in a very similar place,” commented Dr. Mikuls, who is a member of the ACR COVID-19 recommendations task force.
“I think one of the very important similarities that I would highlight is that, in the absence of known exposure, in the absence of COVID-19 infection, our panel felt very strongly about the importance of continuing rheumatic disease treatments,” Dr. Mikuls observed. The ACR guidelines also touch upon societal perspectives, including “some statements that were made very specific to lupus, and the use of antimalarials, given supply chain issues that we have encountered.”
Dr. Mikuls also said that the American recommendations emphasized that “you really have to manage active inflammatory rheumatic disease. Even in the context of the COVID-19 pandemic, given what we saw as the potential risk of unchecked inflammation and unchecked rheumatic disease.”
One notable difference, however, is that the European recommendations advise on immunizations and pneumonia prophylaxis, saying that all patients without COVID-19 symptoms should make sure they are up to date with any recommended vaccinations, “with a particular focus on pneumococcal and influenza vaccinations,” Dr. Landewé said.
Another difference is that the ACR recommendations are a living document and could potentially be updated monthly if the evidence arrives to allow that. In that sense, the American guidance is more agile, with EULAR expecting to update its recommendations every 3 months.
“The current evidence is extremely sparse and fragmented,” Dr. Landewé said. “We, as a task force are essentially flying blindly. We also have to cover many jurisdictions within Europe, with many conflicting opinions. So the last word to say is that updates are truly necessary, but we have to wait a while.”
SOURCE: Landewé RB et al. Ann Rheum Dis. 2020 Jun 5. doi: 10.1136/annrheumdis-2020-217877.
As might be expected, the “EULAR [European League Against Rheumatism] provisional recommendations for the management of rheumatic and musculoskeletal diseases [RMDs] in the context of SARS-CoV-2” concur with much of the guidance already released on how best to manage patients during the current pandemic.
Highlights of the five overarching principles are that, contrary to earlier expectations, “there is no indication that patients with RMDs have an additional, or have a higher, risk of contracting the virus, or that they fare a worse course” than the general population, said the task force convener Robert Landewé, MD, PhD, professor of rheumatology at the University of Amsterdam.
“The second pertinent highlight is that, when it comes to managerial discussions, whether or not to stop or to start treatment for RMDs, rheumatologists should definitely be involved,” Dr. Landewé said during a live session at the annual European Congress of Rheumatology, held online this year due to COVID-19. “In practice, something that happens very often is that immunosuppressive drugs are stopped by medical specialists involved in the care of COVID but without any expertise in treating patients with rheumatic diseases. We should try to avoid that situation.”
The third highlight, something many rheumatologists may already be well aware of, is that rheumatology drugs are being used to treat COVID-19 patients without RMDs and a shortage of disease-modifying antirheumatic drugs (DMARDs) agents is a real possibility. As such, the fifth overarching highlight states that the availability of both synthetic and biologic DMARDs is “a delicate societal responsibility” and that “the off-label use of DMARDs in COVID-19 outside the context of clinical trials should be discouraged.”
The EULAR recommendation are now published online in Annals of the Rheumatic Diseases and they are “what you could call an unprecedented set of recommendations,” Dr. Landewé said. “We have never done this before,” he added, referring to the speed and way in which they had to be put together, remotely, and with little scientific evidence currently available. “Three months ago we hadn’t even heard about the virus.”
From the first patient being identified in the Hubei province of China in November 2019, to the first U.S. patient in the state of Washington on Jan. 20, 2020, and to the first European patient identified a little over 10 days later, the COVID-19 pandemic has taken the world by storm. It was only declared a pandemic on March 11, 2020, however, and Dr. Landewé noted that the response to the pandemic had been very variable – some countries locking down their borders early, while others took their time to make an appropriate response, if at all.
The rheumatology community was particularly concerned, Dr. Landewé said, because people with autoimmune diseases who were taking immunosuppressant drugs might be at higher risk for becoming infected with SARS-CoV-2, and may be at higher risk than others for a worse disease course. Thankfully, that seems not to be the case according to data that are emerging from new registries that have been set up, including EULAR’s own COVID-19 registry.
There are 13 recommendations that cover 4 themes: general measures and prevention of SARS-CoV-2 infection; the management of RMD patients during the pandemic; the management of RMD patients who have COVID-19; and the prevention of other pulmonary infections in RMD patients.
Highlighting the first three general recommendations, Dr. Landewé said: “Follow the regular guidelines in your country; if a patient with RMD does not have symptoms of COVID-19, simply continue RMD treatments,” albeit with a couple of exceptions.
The next four recommendation highlights are to avoid visits to the hospital or to the office; use remote monitoring via the telephone, for example; and if visits cannot be avoided, then take appropriate precautions. Finally, if you suspect a patient has COVID-19, do a test.
If patients test positive, then the next four recommendations cover what to do, such as continuing use of RMD treatments, but in the case of glucocorticoids this should be the lowest possible dose necessary. There is no consensus on what to do in cases of mild symptoms; the recommendation is to “decide on a case-by-case basis,” said Dr. Landewé. If a patient’s symptoms worsen, then “seek expert advice immediately and follow local treatment recommendations. The rheumatologist is not the expert to treat COVID-19,” he added. That responsibility lies with the pulmonologist, infectious disease specialist, or maybe the intensive care specialist, depending on local situations.
On the whole, the EULAR recommendations are pretty similar to those already released by the American College of Rheumatology, said Ted Mikuls, MD, of the University of Nebraska Medical Center, Omaha. The ACR recommendations are “slightly more prescriptive”, he suggested, with 25 final guidance statements. For example, general statements focused not only on the use of glucocorticoids, but also other medicines, such as antihypertensives.
“There’s really not a [lot of], I would say, major differences in the two efforts and that’s ... somewhat reassuring that we’re approaching the unknown from very different parts of the world, and driving in a very similar place,” commented Dr. Mikuls, who is a member of the ACR COVID-19 recommendations task force.
“I think one of the very important similarities that I would highlight is that, in the absence of known exposure, in the absence of COVID-19 infection, our panel felt very strongly about the importance of continuing rheumatic disease treatments,” Dr. Mikuls observed. The ACR guidelines also touch upon societal perspectives, including “some statements that were made very specific to lupus, and the use of antimalarials, given supply chain issues that we have encountered.”
Dr. Mikuls also said that the American recommendations emphasized that “you really have to manage active inflammatory rheumatic disease. Even in the context of the COVID-19 pandemic, given what we saw as the potential risk of unchecked inflammation and unchecked rheumatic disease.”
One notable difference, however, is that the European recommendations advise on immunizations and pneumonia prophylaxis, saying that all patients without COVID-19 symptoms should make sure they are up to date with any recommended vaccinations, “with a particular focus on pneumococcal and influenza vaccinations,” Dr. Landewé said.
Another difference is that the ACR recommendations are a living document and could potentially be updated monthly if the evidence arrives to allow that. In that sense, the American guidance is more agile, with EULAR expecting to update its recommendations every 3 months.
“The current evidence is extremely sparse and fragmented,” Dr. Landewé said. “We, as a task force are essentially flying blindly. We also have to cover many jurisdictions within Europe, with many conflicting opinions. So the last word to say is that updates are truly necessary, but we have to wait a while.”
SOURCE: Landewé RB et al. Ann Rheum Dis. 2020 Jun 5. doi: 10.1136/annrheumdis-2020-217877.
FROM THE EULAR 2020 E-CONGRESS
JAK inhibitors go the distance in RA patients
Patients with rheumatoid arthritis remained on therapy longer with the relatively new JAK inhibitors than with TNF inhibitors, according to the large international JAK-pot study, offering encouraging signals about the efficacy and safety of JAK inhibitors in these patients.
“We saw that efficacy with JAK inhibitors was at least as good as other current drugs on the market,” said investigator Kim Lauper, MD, from the University of Geneva in Switzerland and the University of Manchester in the United Kingdom.
“We don’t have datasets on JAK inhibitors over a long period of time, but we do have a lot of registers,” Dr. Lauper told Medscape Medical News.
“In general, we were really happy to see no big difference in effectiveness” for these disease-modifying antirheumatic drugs (DMARDs) for patients with RA, she said.
In many countries, JAK inhibitors have only recently been approved as a treatment for RA, Lauper explained. In the past several years, baricitinib, tofacitinib, and upadacitinib have been approved by the U.S. Food and Drug Administration.
For their study, Dr. Lauper and her colleagues analyzed data from registers in 19 countries.
When JAK inhibitors became available in each country, the team assessed effectiveness by comparing how long patients remained on JAK inhibitors or on long-available biologics. Dr. Lauper presented the findings at the virtual European League Against Rheumatism (EULAR) 2020 Congress.
“In general, we know that drug retention is a measure of both effectiveness and safety,” she explained.
Of the 25,521 patients in the 19 registers, 6,063 started on a JAK inhibitor during the 3-year study period, 13,879 started on a TNF inhibitor, 2,348 started on abatacept, and 3,231 started on an interleukin-6 inhibitor.
Three-quarters of patients were women (average age, 55 years), and average time since the diagnosis of RA was 10 years.
At baseline, patients taking JAK inhibitors had higher levels of C-reactive protein and disease activity than patients taking a biologic. They had also been treated previously with more traditional and biologic DMARDs.
Ineffectiveness was the most common reason for discontinuing a drug, cited by 49% of patients, followed by adverse events, cited by 21%.
The rate of discontinuation was lower for JAK inhibitors than for TNF inhibitors, after adjustment. However, the discontinuation rate for JAK inhibitors, abatacept, and IL-6 inhibitors was comparable.
The observational nature of the study was a limitation, Dr. Lauper acknowledged, explaining that “we couldn’t adjust for confounding factors that were not measured.”
Notably, there were large variations in JAK inhibitor retention rates in the different countries, which surprised both Dr. Lauper and Loreto Carmona, MD, PhD, from the Musculoskeletal Health Institute in Madrid.
“It’s very interesting because there’s not much heterogeneity with abatacept and IL inhibitors,” said Dr. Carmona, who is chair of the EULAR abstract selection committee.
“It’s all over the spectrum with JAK inhibitors,” she told Medscape Medical News. But “what the research shows is that JAK inhibitors are maintained for longer, which means maybe the mix of efficacy, low toxicity, and adherence, on the whole, is better in JAK inhibitors.”
The study was funded by Pfizer. Dr. Lauper and Dr. Carmona have disclosed no relevant financial relationships.
This story first appeared on Medscape.com.
Patients with rheumatoid arthritis remained on therapy longer with the relatively new JAK inhibitors than with TNF inhibitors, according to the large international JAK-pot study, offering encouraging signals about the efficacy and safety of JAK inhibitors in these patients.
“We saw that efficacy with JAK inhibitors was at least as good as other current drugs on the market,” said investigator Kim Lauper, MD, from the University of Geneva in Switzerland and the University of Manchester in the United Kingdom.
“We don’t have datasets on JAK inhibitors over a long period of time, but we do have a lot of registers,” Dr. Lauper told Medscape Medical News.
“In general, we were really happy to see no big difference in effectiveness” for these disease-modifying antirheumatic drugs (DMARDs) for patients with RA, she said.
In many countries, JAK inhibitors have only recently been approved as a treatment for RA, Lauper explained. In the past several years, baricitinib, tofacitinib, and upadacitinib have been approved by the U.S. Food and Drug Administration.
For their study, Dr. Lauper and her colleagues analyzed data from registers in 19 countries.
When JAK inhibitors became available in each country, the team assessed effectiveness by comparing how long patients remained on JAK inhibitors or on long-available biologics. Dr. Lauper presented the findings at the virtual European League Against Rheumatism (EULAR) 2020 Congress.
“In general, we know that drug retention is a measure of both effectiveness and safety,” she explained.
Of the 25,521 patients in the 19 registers, 6,063 started on a JAK inhibitor during the 3-year study period, 13,879 started on a TNF inhibitor, 2,348 started on abatacept, and 3,231 started on an interleukin-6 inhibitor.
Three-quarters of patients were women (average age, 55 years), and average time since the diagnosis of RA was 10 years.
At baseline, patients taking JAK inhibitors had higher levels of C-reactive protein and disease activity than patients taking a biologic. They had also been treated previously with more traditional and biologic DMARDs.
Ineffectiveness was the most common reason for discontinuing a drug, cited by 49% of patients, followed by adverse events, cited by 21%.
The rate of discontinuation was lower for JAK inhibitors than for TNF inhibitors, after adjustment. However, the discontinuation rate for JAK inhibitors, abatacept, and IL-6 inhibitors was comparable.
The observational nature of the study was a limitation, Dr. Lauper acknowledged, explaining that “we couldn’t adjust for confounding factors that were not measured.”
Notably, there were large variations in JAK inhibitor retention rates in the different countries, which surprised both Dr. Lauper and Loreto Carmona, MD, PhD, from the Musculoskeletal Health Institute in Madrid.
“It’s very interesting because there’s not much heterogeneity with abatacept and IL inhibitors,” said Dr. Carmona, who is chair of the EULAR abstract selection committee.
“It’s all over the spectrum with JAK inhibitors,” she told Medscape Medical News. But “what the research shows is that JAK inhibitors are maintained for longer, which means maybe the mix of efficacy, low toxicity, and adherence, on the whole, is better in JAK inhibitors.”
The study was funded by Pfizer. Dr. Lauper and Dr. Carmona have disclosed no relevant financial relationships.
This story first appeared on Medscape.com.
Patients with rheumatoid arthritis remained on therapy longer with the relatively new JAK inhibitors than with TNF inhibitors, according to the large international JAK-pot study, offering encouraging signals about the efficacy and safety of JAK inhibitors in these patients.
“We saw that efficacy with JAK inhibitors was at least as good as other current drugs on the market,” said investigator Kim Lauper, MD, from the University of Geneva in Switzerland and the University of Manchester in the United Kingdom.
“We don’t have datasets on JAK inhibitors over a long period of time, but we do have a lot of registers,” Dr. Lauper told Medscape Medical News.
“In general, we were really happy to see no big difference in effectiveness” for these disease-modifying antirheumatic drugs (DMARDs) for patients with RA, she said.
In many countries, JAK inhibitors have only recently been approved as a treatment for RA, Lauper explained. In the past several years, baricitinib, tofacitinib, and upadacitinib have been approved by the U.S. Food and Drug Administration.
For their study, Dr. Lauper and her colleagues analyzed data from registers in 19 countries.
When JAK inhibitors became available in each country, the team assessed effectiveness by comparing how long patients remained on JAK inhibitors or on long-available biologics. Dr. Lauper presented the findings at the virtual European League Against Rheumatism (EULAR) 2020 Congress.
“In general, we know that drug retention is a measure of both effectiveness and safety,” she explained.
Of the 25,521 patients in the 19 registers, 6,063 started on a JAK inhibitor during the 3-year study period, 13,879 started on a TNF inhibitor, 2,348 started on abatacept, and 3,231 started on an interleukin-6 inhibitor.
Three-quarters of patients were women (average age, 55 years), and average time since the diagnosis of RA was 10 years.
At baseline, patients taking JAK inhibitors had higher levels of C-reactive protein and disease activity than patients taking a biologic. They had also been treated previously with more traditional and biologic DMARDs.
Ineffectiveness was the most common reason for discontinuing a drug, cited by 49% of patients, followed by adverse events, cited by 21%.
The rate of discontinuation was lower for JAK inhibitors than for TNF inhibitors, after adjustment. However, the discontinuation rate for JAK inhibitors, abatacept, and IL-6 inhibitors was comparable.
The observational nature of the study was a limitation, Dr. Lauper acknowledged, explaining that “we couldn’t adjust for confounding factors that were not measured.”
Notably, there were large variations in JAK inhibitor retention rates in the different countries, which surprised both Dr. Lauper and Loreto Carmona, MD, PhD, from the Musculoskeletal Health Institute in Madrid.
“It’s very interesting because there’s not much heterogeneity with abatacept and IL inhibitors,” said Dr. Carmona, who is chair of the EULAR abstract selection committee.
“It’s all over the spectrum with JAK inhibitors,” she told Medscape Medical News. But “what the research shows is that JAK inhibitors are maintained for longer, which means maybe the mix of efficacy, low toxicity, and adherence, on the whole, is better in JAK inhibitors.”
The study was funded by Pfizer. Dr. Lauper and Dr. Carmona have disclosed no relevant financial relationships.
This story first appeared on Medscape.com.
Opioid use up after TNF inhibitor for inflammatory arthritis
Opioid use does not decline after patients with inflammatory arthritis start TNF inhibitor therapy; in fact, average use appears to increase, results from a new study show.
“Starting a TNF inhibitor, you would think the pain would go down, and we were hoping the dose of opioids would go down with it,” said investigator Olafur Palsson, MD, from the University of Iceland in Reykjavik and Lund University in Sweden.
“But this research shows that the insertion of a TNF inhibitor has only a minor effect on that,” he told Medscape Medical News.
The findings are an “important reminder” to rheumatologists that they should broaden their consideration of other pain treatments and techniques for patients with inflammatory arthritis, Dr. Palsson said. “They should focus on trying other tactics to get patients’ pain and stiffness under control; there may be some underlying factors.”
The investigators compared opioid prescription rates in 940 patients with rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, and undifferentiated arthritis with a control group of 4,700 matched subjects. Dr. Palsson presented the findings at the virtual European League Against Rheumatism (EULAR) 2020 Congress.
The team assessed nationwide databases that capture all patients taking biologics for rheumatic diseases and more than 90% of all drug prescriptions. They found that patients with inflammatory arthritis in Iceland were more likely to have received at least one opioid prescription than control subjects (75% vs. 43%).
During the study period, average yearly opioid dose rose much more in the patient group than in the control group. And 2 years after the initiation of TNF inhibitors, the number of patients taking opioids was unchanged from baseline, at about 40%.
Overall, the patient group was prescribed nearly six times more opioids than the control group. The investigators used a bootstrapping analysis to obtain a reliable confidence interval.
“In a way, the data are extremely skewed,” Dr. Palsson explained. “Most patients were taking very low doses of opioids and a few were taking extremely high doses. It’s hard to do a statistical analysis.”
“With bootstrapping, you don’t detect small fluctuations in data,” he said, acknowledging this study limitation. Also, “prescription data don’t necessarily reflect consumption” of a drug. People prescribed high doses may not necessarily be consuming high doses.”
Additionally, the risk for addiction is low when opioids are used as intended, said John Isaacs, MBBS, PhD, from Newcastle University in Newcastle Upon Tyne, United Kingdom, who is chair of the EULAR scientific program committee.
To alleviate chronic pain, opioids “should, in any case, only be part of a comprehensive therapy program in which doctors, psychologists, and physiotherapists work together,” Dr. Isaacs said in a EULAR news release.
Dr. Palsson has disclosed no relevant financial relationships. Dr. Isaacs is a consultant or has received honoraria or grants from Pfizer, AbbVie, Amgen, Merck, Roche, and UCB.
This article first appeared on Medscape.com.
Opioid use does not decline after patients with inflammatory arthritis start TNF inhibitor therapy; in fact, average use appears to increase, results from a new study show.
“Starting a TNF inhibitor, you would think the pain would go down, and we were hoping the dose of opioids would go down with it,” said investigator Olafur Palsson, MD, from the University of Iceland in Reykjavik and Lund University in Sweden.
“But this research shows that the insertion of a TNF inhibitor has only a minor effect on that,” he told Medscape Medical News.
The findings are an “important reminder” to rheumatologists that they should broaden their consideration of other pain treatments and techniques for patients with inflammatory arthritis, Dr. Palsson said. “They should focus on trying other tactics to get patients’ pain and stiffness under control; there may be some underlying factors.”
The investigators compared opioid prescription rates in 940 patients with rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, and undifferentiated arthritis with a control group of 4,700 matched subjects. Dr. Palsson presented the findings at the virtual European League Against Rheumatism (EULAR) 2020 Congress.
The team assessed nationwide databases that capture all patients taking biologics for rheumatic diseases and more than 90% of all drug prescriptions. They found that patients with inflammatory arthritis in Iceland were more likely to have received at least one opioid prescription than control subjects (75% vs. 43%).
During the study period, average yearly opioid dose rose much more in the patient group than in the control group. And 2 years after the initiation of TNF inhibitors, the number of patients taking opioids was unchanged from baseline, at about 40%.
Overall, the patient group was prescribed nearly six times more opioids than the control group. The investigators used a bootstrapping analysis to obtain a reliable confidence interval.
“In a way, the data are extremely skewed,” Dr. Palsson explained. “Most patients were taking very low doses of opioids and a few were taking extremely high doses. It’s hard to do a statistical analysis.”
“With bootstrapping, you don’t detect small fluctuations in data,” he said, acknowledging this study limitation. Also, “prescription data don’t necessarily reflect consumption” of a drug. People prescribed high doses may not necessarily be consuming high doses.”
Additionally, the risk for addiction is low when opioids are used as intended, said John Isaacs, MBBS, PhD, from Newcastle University in Newcastle Upon Tyne, United Kingdom, who is chair of the EULAR scientific program committee.
To alleviate chronic pain, opioids “should, in any case, only be part of a comprehensive therapy program in which doctors, psychologists, and physiotherapists work together,” Dr. Isaacs said in a EULAR news release.
Dr. Palsson has disclosed no relevant financial relationships. Dr. Isaacs is a consultant or has received honoraria or grants from Pfizer, AbbVie, Amgen, Merck, Roche, and UCB.
This article first appeared on Medscape.com.
Opioid use does not decline after patients with inflammatory arthritis start TNF inhibitor therapy; in fact, average use appears to increase, results from a new study show.
“Starting a TNF inhibitor, you would think the pain would go down, and we were hoping the dose of opioids would go down with it,” said investigator Olafur Palsson, MD, from the University of Iceland in Reykjavik and Lund University in Sweden.
“But this research shows that the insertion of a TNF inhibitor has only a minor effect on that,” he told Medscape Medical News.
The findings are an “important reminder” to rheumatologists that they should broaden their consideration of other pain treatments and techniques for patients with inflammatory arthritis, Dr. Palsson said. “They should focus on trying other tactics to get patients’ pain and stiffness under control; there may be some underlying factors.”
The investigators compared opioid prescription rates in 940 patients with rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, and undifferentiated arthritis with a control group of 4,700 matched subjects. Dr. Palsson presented the findings at the virtual European League Against Rheumatism (EULAR) 2020 Congress.
The team assessed nationwide databases that capture all patients taking biologics for rheumatic diseases and more than 90% of all drug prescriptions. They found that patients with inflammatory arthritis in Iceland were more likely to have received at least one opioid prescription than control subjects (75% vs. 43%).
During the study period, average yearly opioid dose rose much more in the patient group than in the control group. And 2 years after the initiation of TNF inhibitors, the number of patients taking opioids was unchanged from baseline, at about 40%.
Overall, the patient group was prescribed nearly six times more opioids than the control group. The investigators used a bootstrapping analysis to obtain a reliable confidence interval.
“In a way, the data are extremely skewed,” Dr. Palsson explained. “Most patients were taking very low doses of opioids and a few were taking extremely high doses. It’s hard to do a statistical analysis.”
“With bootstrapping, you don’t detect small fluctuations in data,” he said, acknowledging this study limitation. Also, “prescription data don’t necessarily reflect consumption” of a drug. People prescribed high doses may not necessarily be consuming high doses.”
Additionally, the risk for addiction is low when opioids are used as intended, said John Isaacs, MBBS, PhD, from Newcastle University in Newcastle Upon Tyne, United Kingdom, who is chair of the EULAR scientific program committee.
To alleviate chronic pain, opioids “should, in any case, only be part of a comprehensive therapy program in which doctors, psychologists, and physiotherapists work together,” Dr. Isaacs said in a EULAR news release.
Dr. Palsson has disclosed no relevant financial relationships. Dr. Isaacs is a consultant or has received honoraria or grants from Pfizer, AbbVie, Amgen, Merck, Roche, and UCB.
This article first appeared on Medscape.com.
TNF inhibitors cut odds of VTE in RA patients
The risk for venous thromboembolism is almost 50% lower in patients with RA taking TNF inhibitors than in those taking conventional synthetic disease-modifying antirheumatic drugs (DMARDs), according to data from the German RABBIT registry.
“Some rheumatologists have thought TNF inhibitors could increase the risk for venous thromboembolism events, but we don’t think this is true, based on our findings,” said investigator Anja Strangfeld, MD, PhD, from the German Rheumatism Research Center in Berlin.
The risk is more than one-third lower in RA patients treated with other newer biologics, such as abatacept, rituximab, sarilumab, and tocilizumab.
However, risk for a serious venous thromboembolism is twice as high in patients with C-reactive protein (CRP) levels above 5 mg/L and is nearly three times as high in patients 65 years and older.
For the study, Dr. Strangfeld and her colleagues followed about 11,000 patients for more than 10 years. The findings were presented at the European League Against Rheumatism (EULAR) 2020 Congress.
“Patients with RA have a greater risk for venous thromboembolism compared with the general population, but we didn’t know the risk conveyed by different DMARD treatments,” Dr. Strangfeld told Medscape Medical News. “It is also evident that higher age and lower capacity for physical function increase the risk, which was not so surprising.”
Chronic inflammation in RA patients elevates the risk for deep vein and pulmonary thrombosis by two to three times, said John Isaacs, MBBS, PhD, from Newcastle University in Newcastle Upon Tyne, United Kingdom, who is chair of the EULAR scientific program committee.
Among the supporting studies Dr. Isaacs discussed during an online press conference was a Swedish trial of more than 46,000 RA patients, which had been presented earlier by Viktor Molander, a PhD candidate from the Karolinska Institute in Stockholm (abstract OP0034).
Mr. Molander’s team showed that one in 100 patients with high disease activity will develop venous thromboembolism within a year, which is twice the number of events seen among patients in remission.
Combined with the RABBIT data, both studies “show if you can control their disease in the right way, you’re not only helping rheumatoid arthritis patients feel better, but you could be prolonging their lives,” Dr. Isaacs said.
The prospective RABBIT study followed RA patients who began receiving a new DMARD after treatment failed with at least one conventional synthetic DMARD, such as methotrexate or leflunomide. At baseline, those taking TNF inhibitors or other biologics had higher CRP levels on average, as well as a higher rate of existing cardiovascular disease. They also received glucocorticoids, such as prednisone, more often.
The observational nature of the RABBIT study is a weakness, Dr. Strangfeld said, and it could not prove cause and effect. But the methodology had several strengths, including input on patient factors from participating rheumatologists at least every 6 months.
“We enrolled patients at the start of treatment and observed them, regardless of any treatment changes, for up to 10 years,” she added. “That’s a really long observation period.”
The RABBIT data can help shape treatment decisions, said Loreto Carmona, MD, PhD, from the Musculoskeletal Health Institute in Madrid, who is chair of the EULAR abstract selection committee.
For a woman with RA who smokes and takes oral contraceptives, for example, “if she has high levels of inflammation, I think it’s okay to use TNF inhibitors, where maybe in the past we wouldn’t have thought that,” she said.
“The TNF inhibitors are actually reducing the inflammation and, therefore, reducing the risk,” Dr. Carmona told Medscape Medical News. “It could be an effect of using the drugs on people with higher levels of inflammation. It’s an indirect protective effect.”
The study was funded by a joint unconditional grant from AbbVie, Amgen, BMS, Fresenius-Kabi, Hexal, Lilly, MSD, Mylan, Pfizer, Roche, Samsung Bioepis, Sanofi-Aventis, and UCB. Dr. Strangfeld is on the speakers bureau of AbbVie, BMS, Pfizer, Roche and Sanofi-Aventis. Dr. Isaacs is a consultant or has received honoraria or grants from Pfizer, AbbVie, Amgen, Merck, Roche, and UCB. Dr. Carmona has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
The risk for venous thromboembolism is almost 50% lower in patients with RA taking TNF inhibitors than in those taking conventional synthetic disease-modifying antirheumatic drugs (DMARDs), according to data from the German RABBIT registry.
“Some rheumatologists have thought TNF inhibitors could increase the risk for venous thromboembolism events, but we don’t think this is true, based on our findings,” said investigator Anja Strangfeld, MD, PhD, from the German Rheumatism Research Center in Berlin.
The risk is more than one-third lower in RA patients treated with other newer biologics, such as abatacept, rituximab, sarilumab, and tocilizumab.
However, risk for a serious venous thromboembolism is twice as high in patients with C-reactive protein (CRP) levels above 5 mg/L and is nearly three times as high in patients 65 years and older.
For the study, Dr. Strangfeld and her colleagues followed about 11,000 patients for more than 10 years. The findings were presented at the European League Against Rheumatism (EULAR) 2020 Congress.
“Patients with RA have a greater risk for venous thromboembolism compared with the general population, but we didn’t know the risk conveyed by different DMARD treatments,” Dr. Strangfeld told Medscape Medical News. “It is also evident that higher age and lower capacity for physical function increase the risk, which was not so surprising.”
Chronic inflammation in RA patients elevates the risk for deep vein and pulmonary thrombosis by two to three times, said John Isaacs, MBBS, PhD, from Newcastle University in Newcastle Upon Tyne, United Kingdom, who is chair of the EULAR scientific program committee.
Among the supporting studies Dr. Isaacs discussed during an online press conference was a Swedish trial of more than 46,000 RA patients, which had been presented earlier by Viktor Molander, a PhD candidate from the Karolinska Institute in Stockholm (abstract OP0034).
Mr. Molander’s team showed that one in 100 patients with high disease activity will develop venous thromboembolism within a year, which is twice the number of events seen among patients in remission.
Combined with the RABBIT data, both studies “show if you can control their disease in the right way, you’re not only helping rheumatoid arthritis patients feel better, but you could be prolonging their lives,” Dr. Isaacs said.
The prospective RABBIT study followed RA patients who began receiving a new DMARD after treatment failed with at least one conventional synthetic DMARD, such as methotrexate or leflunomide. At baseline, those taking TNF inhibitors or other biologics had higher CRP levels on average, as well as a higher rate of existing cardiovascular disease. They also received glucocorticoids, such as prednisone, more often.
The observational nature of the RABBIT study is a weakness, Dr. Strangfeld said, and it could not prove cause and effect. But the methodology had several strengths, including input on patient factors from participating rheumatologists at least every 6 months.
“We enrolled patients at the start of treatment and observed them, regardless of any treatment changes, for up to 10 years,” she added. “That’s a really long observation period.”
The RABBIT data can help shape treatment decisions, said Loreto Carmona, MD, PhD, from the Musculoskeletal Health Institute in Madrid, who is chair of the EULAR abstract selection committee.
For a woman with RA who smokes and takes oral contraceptives, for example, “if she has high levels of inflammation, I think it’s okay to use TNF inhibitors, where maybe in the past we wouldn’t have thought that,” she said.
“The TNF inhibitors are actually reducing the inflammation and, therefore, reducing the risk,” Dr. Carmona told Medscape Medical News. “It could be an effect of using the drugs on people with higher levels of inflammation. It’s an indirect protective effect.”
The study was funded by a joint unconditional grant from AbbVie, Amgen, BMS, Fresenius-Kabi, Hexal, Lilly, MSD, Mylan, Pfizer, Roche, Samsung Bioepis, Sanofi-Aventis, and UCB. Dr. Strangfeld is on the speakers bureau of AbbVie, BMS, Pfizer, Roche and Sanofi-Aventis. Dr. Isaacs is a consultant or has received honoraria or grants from Pfizer, AbbVie, Amgen, Merck, Roche, and UCB. Dr. Carmona has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
The risk for venous thromboembolism is almost 50% lower in patients with RA taking TNF inhibitors than in those taking conventional synthetic disease-modifying antirheumatic drugs (DMARDs), according to data from the German RABBIT registry.
“Some rheumatologists have thought TNF inhibitors could increase the risk for venous thromboembolism events, but we don’t think this is true, based on our findings,” said investigator Anja Strangfeld, MD, PhD, from the German Rheumatism Research Center in Berlin.
The risk is more than one-third lower in RA patients treated with other newer biologics, such as abatacept, rituximab, sarilumab, and tocilizumab.
However, risk for a serious venous thromboembolism is twice as high in patients with C-reactive protein (CRP) levels above 5 mg/L and is nearly three times as high in patients 65 years and older.
For the study, Dr. Strangfeld and her colleagues followed about 11,000 patients for more than 10 years. The findings were presented at the European League Against Rheumatism (EULAR) 2020 Congress.
“Patients with RA have a greater risk for venous thromboembolism compared with the general population, but we didn’t know the risk conveyed by different DMARD treatments,” Dr. Strangfeld told Medscape Medical News. “It is also evident that higher age and lower capacity for physical function increase the risk, which was not so surprising.”
Chronic inflammation in RA patients elevates the risk for deep vein and pulmonary thrombosis by two to three times, said John Isaacs, MBBS, PhD, from Newcastle University in Newcastle Upon Tyne, United Kingdom, who is chair of the EULAR scientific program committee.
Among the supporting studies Dr. Isaacs discussed during an online press conference was a Swedish trial of more than 46,000 RA patients, which had been presented earlier by Viktor Molander, a PhD candidate from the Karolinska Institute in Stockholm (abstract OP0034).
Mr. Molander’s team showed that one in 100 patients with high disease activity will develop venous thromboembolism within a year, which is twice the number of events seen among patients in remission.
Combined with the RABBIT data, both studies “show if you can control their disease in the right way, you’re not only helping rheumatoid arthritis patients feel better, but you could be prolonging their lives,” Dr. Isaacs said.
The prospective RABBIT study followed RA patients who began receiving a new DMARD after treatment failed with at least one conventional synthetic DMARD, such as methotrexate or leflunomide. At baseline, those taking TNF inhibitors or other biologics had higher CRP levels on average, as well as a higher rate of existing cardiovascular disease. They also received glucocorticoids, such as prednisone, more often.
The observational nature of the RABBIT study is a weakness, Dr. Strangfeld said, and it could not prove cause and effect. But the methodology had several strengths, including input on patient factors from participating rheumatologists at least every 6 months.
“We enrolled patients at the start of treatment and observed them, regardless of any treatment changes, for up to 10 years,” she added. “That’s a really long observation period.”
The RABBIT data can help shape treatment decisions, said Loreto Carmona, MD, PhD, from the Musculoskeletal Health Institute in Madrid, who is chair of the EULAR abstract selection committee.
For a woman with RA who smokes and takes oral contraceptives, for example, “if she has high levels of inflammation, I think it’s okay to use TNF inhibitors, where maybe in the past we wouldn’t have thought that,” she said.
“The TNF inhibitors are actually reducing the inflammation and, therefore, reducing the risk,” Dr. Carmona told Medscape Medical News. “It could be an effect of using the drugs on people with higher levels of inflammation. It’s an indirect protective effect.”
The study was funded by a joint unconditional grant from AbbVie, Amgen, BMS, Fresenius-Kabi, Hexal, Lilly, MSD, Mylan, Pfizer, Roche, Samsung Bioepis, Sanofi-Aventis, and UCB. Dr. Strangfeld is on the speakers bureau of AbbVie, BMS, Pfizer, Roche and Sanofi-Aventis. Dr. Isaacs is a consultant or has received honoraria or grants from Pfizer, AbbVie, Amgen, Merck, Roche, and UCB. Dr. Carmona has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.