User login
Role of aspirin explored in primary prevention of CVD in systemic rheumatic diseases
Low-dose aspirin may be considered for the primary prevention of cardiovascular disease (CVD) in patients with autoimmune systemic rheumatic diseases who are at particularly high risk because of their individual cardiovascular risk profile, according to authors of a new review article in the journal Rheumatology who acknowledge the controversial nature of the issue, because while significant cardiovascular benefit from aspirin for secondary prevention is well established, it has not been for primary prevention.
Secondary prevention with daily, low-dose aspirin is part of aggressive, comprehensive risk modification in patients who have experienced an MI or stroke or are considered at high risk for CVD. But when it comes to primary prevention of the onset of disease, the authors, led by Serena Fasano, MD, PhD, of the rheumatology unit at the University of Campania, Naples, Italy, acknowledged the contradictory positions of international guidelines and uncertainty over balancing benefit versus harm – including risk of mortality in the context of excess bleeding. They called for “robust data” from high-quality randomized, controlled trials for subgroups of patients with specific rheumatologic diseases in order to better answer the question of aspirin for primary prevention.
“This review is devoted to reporting the present knowledge on the effectiveness of low-dose [aspirin] in primary CV prevention in a number of autoimmune systemic rheumatic diseases, not a systematic review or meta-analysis,” the authors stated. “We are not claiming to have covered more than a selection of the literature for each disease. Available data are not high-quality data and do not provide firm conclusions.”
The authors focused primarily on accelerated, rather than spontaneous, atherosclerosis or buildup of plaque in artery walls, implicated in ischemic heart diseases such as MI and ischemic cerebrovascular diseases such as stroke. They looked at its association with autoimmune rheumatic diseases, primarily systemic lupus erythematosus (SLE) and RA, but also including antiphospholipid syndrome, systemic sclerosis, mixed connective tissue disease, dermatomyositis/polymyositis, primary Sjögren’s syndrome, and systemic vasculitis.
They shared results from a review of 167 patients with SLE consecutively admitted to their tertiary medical center who had not previously experienced a cardiovascular event and who were prescribed low-dose (100 mg) aspirin on their first visit and followed for 8 years. The cardiovascular event-free rate was higher in the aspirin group and no excess bleeding was noted, although this may be attributable to a younger patient population and routine use of proton pump inhibitors. Subsequently, hydroxychloroquine was added to the aspirin treatment and was associated with further reduction in cardiovascular events.
The research group also conducted a retrospective analysis of 746 patients with RA consecutively admitted to four tertiary medical centers who hadn’t experienced a cardiovascular event previously. Incidence of cardiovascular events was significantly lower in aspirin-treated patients.
Individualized aspirin prescribing with cardiologist comanagement
There may be a modest benefit of using low-dose aspirin on a long-term basis, but that benefit needs to be offset by the risk of bleeds, said M. Elaine Husni, MD, MPH, vice chair of rheumatology and director of the Arthritis and Musculoskeletal Center at the Cleveland Clinic. It’s important to remind clinicians of cardiovascular risk, she said. “But the message for rheumatologists is it needs to be prescribed on an individual basis, rather than based on diagnosis of a rheumatic condition – at least until we have better evidence.”
Dr. Husni recommended keeping an open mind regarding individual approaches – for example, low-dose aspirin plus statins. A composite approach to prevention likely is called for, including attention to lifestyle issues such as smoking cessation, exercise, and weight loss. “That kind of complexity in decision-making highlights the need for comanagement with a cardiologist,” she said. “I’m a big believer in comanagement. At my multidisciplinary medical center, I am able to pick up the phone and talk to a cardiologist with whom our group has a relationship.” If physicians don’t have that kind of relationship with a cardiology group, she suggested reaching out to establish one.
The review paper could give some guidance to rheumatologists for use on an individual case, Michael Nurmohamed, MD, PhD, of the Amsterdam Rheumatology and Immunology Center in the Netherlands commented in an interview. “However, firm recommendations cannot be given as proper investigations are still lacking, as acknowledged by the authors. In addition, the review paper itself has some methodological constraints. Although this is a narrative review, the search strategy should have been specified, and a quality assessment of the individual studies is lacking.”
There is no doubt that the CVD burden in RA and other rheumatologic conditions is substantially increased in comparison to the general population, Dr. Nurmohamed said. That has been assessed by several well-designed, prospective, controlled studies. Other relatively frequent inflammatory arthropathies, including ankylosing spondylitis and psoriatic arthritis, also pose cardiovascular risk.
“Aspirin cannot be recommended for primary CVD prevention in inflammatory arthropathies due to the absence of adequate studies. That’s why the EULAR [European League Against Rheumatism] guidelines did not recommend its use,” he said. Currently, a EULAR task force is developing evidence-based guidelines for primary CVD prevention in the diseases discussed by Fasano et al., where the use of aspirin will be reassessed. “As these guidelines will consider the methodological quality of the underlying studies, they will enable a more refined use of aspirin in daily clinical practice.”
Primary prevention of CVD using aspirin is not currently the standard of care in taking care of patients with rheumatologic disease in the Netherlands, Ronald F. van Vollenhoven, MD, PhD, Dr. Nurmohamed’s colleague and director of the Amsterdam Rheumatology and Immunology Center and the chair of the department of rheumatology and clinical immunology at the Amsterdam University Medical Center, said in an interview.
“One reason may be the limited data, as highlighted in the review by Dr. Fasano and colleagues. However, another consideration is the problem of polypharmacy. Rheumatic diseases usually require chronic treatment, sometimes with multiple medications. This makes it even more of a concern to add an additional medication, even a relatively innocuous one such as low-dose aspirin,” he said.
Dr. Husni, Dr. Nurmohamed, and Dr. van Vollenhoven reported having no relevant disclosures. The authors of the review article had no relevant disclosures.
SOURCE: Fasano S et al. Rheumatology. 2020 Aug 25. doi: 10.1093/rheumatology/keaa335.
Low-dose aspirin may be considered for the primary prevention of cardiovascular disease (CVD) in patients with autoimmune systemic rheumatic diseases who are at particularly high risk because of their individual cardiovascular risk profile, according to authors of a new review article in the journal Rheumatology who acknowledge the controversial nature of the issue, because while significant cardiovascular benefit from aspirin for secondary prevention is well established, it has not been for primary prevention.
Secondary prevention with daily, low-dose aspirin is part of aggressive, comprehensive risk modification in patients who have experienced an MI or stroke or are considered at high risk for CVD. But when it comes to primary prevention of the onset of disease, the authors, led by Serena Fasano, MD, PhD, of the rheumatology unit at the University of Campania, Naples, Italy, acknowledged the contradictory positions of international guidelines and uncertainty over balancing benefit versus harm – including risk of mortality in the context of excess bleeding. They called for “robust data” from high-quality randomized, controlled trials for subgroups of patients with specific rheumatologic diseases in order to better answer the question of aspirin for primary prevention.
“This review is devoted to reporting the present knowledge on the effectiveness of low-dose [aspirin] in primary CV prevention in a number of autoimmune systemic rheumatic diseases, not a systematic review or meta-analysis,” the authors stated. “We are not claiming to have covered more than a selection of the literature for each disease. Available data are not high-quality data and do not provide firm conclusions.”
The authors focused primarily on accelerated, rather than spontaneous, atherosclerosis or buildup of plaque in artery walls, implicated in ischemic heart diseases such as MI and ischemic cerebrovascular diseases such as stroke. They looked at its association with autoimmune rheumatic diseases, primarily systemic lupus erythematosus (SLE) and RA, but also including antiphospholipid syndrome, systemic sclerosis, mixed connective tissue disease, dermatomyositis/polymyositis, primary Sjögren’s syndrome, and systemic vasculitis.
They shared results from a review of 167 patients with SLE consecutively admitted to their tertiary medical center who had not previously experienced a cardiovascular event and who were prescribed low-dose (100 mg) aspirin on their first visit and followed for 8 years. The cardiovascular event-free rate was higher in the aspirin group and no excess bleeding was noted, although this may be attributable to a younger patient population and routine use of proton pump inhibitors. Subsequently, hydroxychloroquine was added to the aspirin treatment and was associated with further reduction in cardiovascular events.
The research group also conducted a retrospective analysis of 746 patients with RA consecutively admitted to four tertiary medical centers who hadn’t experienced a cardiovascular event previously. Incidence of cardiovascular events was significantly lower in aspirin-treated patients.
Individualized aspirin prescribing with cardiologist comanagement
There may be a modest benefit of using low-dose aspirin on a long-term basis, but that benefit needs to be offset by the risk of bleeds, said M. Elaine Husni, MD, MPH, vice chair of rheumatology and director of the Arthritis and Musculoskeletal Center at the Cleveland Clinic. It’s important to remind clinicians of cardiovascular risk, she said. “But the message for rheumatologists is it needs to be prescribed on an individual basis, rather than based on diagnosis of a rheumatic condition – at least until we have better evidence.”
Dr. Husni recommended keeping an open mind regarding individual approaches – for example, low-dose aspirin plus statins. A composite approach to prevention likely is called for, including attention to lifestyle issues such as smoking cessation, exercise, and weight loss. “That kind of complexity in decision-making highlights the need for comanagement with a cardiologist,” she said. “I’m a big believer in comanagement. At my multidisciplinary medical center, I am able to pick up the phone and talk to a cardiologist with whom our group has a relationship.” If physicians don’t have that kind of relationship with a cardiology group, she suggested reaching out to establish one.
The review paper could give some guidance to rheumatologists for use on an individual case, Michael Nurmohamed, MD, PhD, of the Amsterdam Rheumatology and Immunology Center in the Netherlands commented in an interview. “However, firm recommendations cannot be given as proper investigations are still lacking, as acknowledged by the authors. In addition, the review paper itself has some methodological constraints. Although this is a narrative review, the search strategy should have been specified, and a quality assessment of the individual studies is lacking.”
There is no doubt that the CVD burden in RA and other rheumatologic conditions is substantially increased in comparison to the general population, Dr. Nurmohamed said. That has been assessed by several well-designed, prospective, controlled studies. Other relatively frequent inflammatory arthropathies, including ankylosing spondylitis and psoriatic arthritis, also pose cardiovascular risk.
“Aspirin cannot be recommended for primary CVD prevention in inflammatory arthropathies due to the absence of adequate studies. That’s why the EULAR [European League Against Rheumatism] guidelines did not recommend its use,” he said. Currently, a EULAR task force is developing evidence-based guidelines for primary CVD prevention in the diseases discussed by Fasano et al., where the use of aspirin will be reassessed. “As these guidelines will consider the methodological quality of the underlying studies, they will enable a more refined use of aspirin in daily clinical practice.”
Primary prevention of CVD using aspirin is not currently the standard of care in taking care of patients with rheumatologic disease in the Netherlands, Ronald F. van Vollenhoven, MD, PhD, Dr. Nurmohamed’s colleague and director of the Amsterdam Rheumatology and Immunology Center and the chair of the department of rheumatology and clinical immunology at the Amsterdam University Medical Center, said in an interview.
“One reason may be the limited data, as highlighted in the review by Dr. Fasano and colleagues. However, another consideration is the problem of polypharmacy. Rheumatic diseases usually require chronic treatment, sometimes with multiple medications. This makes it even more of a concern to add an additional medication, even a relatively innocuous one such as low-dose aspirin,” he said.
Dr. Husni, Dr. Nurmohamed, and Dr. van Vollenhoven reported having no relevant disclosures. The authors of the review article had no relevant disclosures.
SOURCE: Fasano S et al. Rheumatology. 2020 Aug 25. doi: 10.1093/rheumatology/keaa335.
Low-dose aspirin may be considered for the primary prevention of cardiovascular disease (CVD) in patients with autoimmune systemic rheumatic diseases who are at particularly high risk because of their individual cardiovascular risk profile, according to authors of a new review article in the journal Rheumatology who acknowledge the controversial nature of the issue, because while significant cardiovascular benefit from aspirin for secondary prevention is well established, it has not been for primary prevention.
Secondary prevention with daily, low-dose aspirin is part of aggressive, comprehensive risk modification in patients who have experienced an MI or stroke or are considered at high risk for CVD. But when it comes to primary prevention of the onset of disease, the authors, led by Serena Fasano, MD, PhD, of the rheumatology unit at the University of Campania, Naples, Italy, acknowledged the contradictory positions of international guidelines and uncertainty over balancing benefit versus harm – including risk of mortality in the context of excess bleeding. They called for “robust data” from high-quality randomized, controlled trials for subgroups of patients with specific rheumatologic diseases in order to better answer the question of aspirin for primary prevention.
“This review is devoted to reporting the present knowledge on the effectiveness of low-dose [aspirin] in primary CV prevention in a number of autoimmune systemic rheumatic diseases, not a systematic review or meta-analysis,” the authors stated. “We are not claiming to have covered more than a selection of the literature for each disease. Available data are not high-quality data and do not provide firm conclusions.”
The authors focused primarily on accelerated, rather than spontaneous, atherosclerosis or buildup of plaque in artery walls, implicated in ischemic heart diseases such as MI and ischemic cerebrovascular diseases such as stroke. They looked at its association with autoimmune rheumatic diseases, primarily systemic lupus erythematosus (SLE) and RA, but also including antiphospholipid syndrome, systemic sclerosis, mixed connective tissue disease, dermatomyositis/polymyositis, primary Sjögren’s syndrome, and systemic vasculitis.
They shared results from a review of 167 patients with SLE consecutively admitted to their tertiary medical center who had not previously experienced a cardiovascular event and who were prescribed low-dose (100 mg) aspirin on their first visit and followed for 8 years. The cardiovascular event-free rate was higher in the aspirin group and no excess bleeding was noted, although this may be attributable to a younger patient population and routine use of proton pump inhibitors. Subsequently, hydroxychloroquine was added to the aspirin treatment and was associated with further reduction in cardiovascular events.
The research group also conducted a retrospective analysis of 746 patients with RA consecutively admitted to four tertiary medical centers who hadn’t experienced a cardiovascular event previously. Incidence of cardiovascular events was significantly lower in aspirin-treated patients.
Individualized aspirin prescribing with cardiologist comanagement
There may be a modest benefit of using low-dose aspirin on a long-term basis, but that benefit needs to be offset by the risk of bleeds, said M. Elaine Husni, MD, MPH, vice chair of rheumatology and director of the Arthritis and Musculoskeletal Center at the Cleveland Clinic. It’s important to remind clinicians of cardiovascular risk, she said. “But the message for rheumatologists is it needs to be prescribed on an individual basis, rather than based on diagnosis of a rheumatic condition – at least until we have better evidence.”
Dr. Husni recommended keeping an open mind regarding individual approaches – for example, low-dose aspirin plus statins. A composite approach to prevention likely is called for, including attention to lifestyle issues such as smoking cessation, exercise, and weight loss. “That kind of complexity in decision-making highlights the need for comanagement with a cardiologist,” she said. “I’m a big believer in comanagement. At my multidisciplinary medical center, I am able to pick up the phone and talk to a cardiologist with whom our group has a relationship.” If physicians don’t have that kind of relationship with a cardiology group, she suggested reaching out to establish one.
The review paper could give some guidance to rheumatologists for use on an individual case, Michael Nurmohamed, MD, PhD, of the Amsterdam Rheumatology and Immunology Center in the Netherlands commented in an interview. “However, firm recommendations cannot be given as proper investigations are still lacking, as acknowledged by the authors. In addition, the review paper itself has some methodological constraints. Although this is a narrative review, the search strategy should have been specified, and a quality assessment of the individual studies is lacking.”
There is no doubt that the CVD burden in RA and other rheumatologic conditions is substantially increased in comparison to the general population, Dr. Nurmohamed said. That has been assessed by several well-designed, prospective, controlled studies. Other relatively frequent inflammatory arthropathies, including ankylosing spondylitis and psoriatic arthritis, also pose cardiovascular risk.
“Aspirin cannot be recommended for primary CVD prevention in inflammatory arthropathies due to the absence of adequate studies. That’s why the EULAR [European League Against Rheumatism] guidelines did not recommend its use,” he said. Currently, a EULAR task force is developing evidence-based guidelines for primary CVD prevention in the diseases discussed by Fasano et al., where the use of aspirin will be reassessed. “As these guidelines will consider the methodological quality of the underlying studies, they will enable a more refined use of aspirin in daily clinical practice.”
Primary prevention of CVD using aspirin is not currently the standard of care in taking care of patients with rheumatologic disease in the Netherlands, Ronald F. van Vollenhoven, MD, PhD, Dr. Nurmohamed’s colleague and director of the Amsterdam Rheumatology and Immunology Center and the chair of the department of rheumatology and clinical immunology at the Amsterdam University Medical Center, said in an interview.
“One reason may be the limited data, as highlighted in the review by Dr. Fasano and colleagues. However, another consideration is the problem of polypharmacy. Rheumatic diseases usually require chronic treatment, sometimes with multiple medications. This makes it even more of a concern to add an additional medication, even a relatively innocuous one such as low-dose aspirin,” he said.
Dr. Husni, Dr. Nurmohamed, and Dr. van Vollenhoven reported having no relevant disclosures. The authors of the review article had no relevant disclosures.
SOURCE: Fasano S et al. Rheumatology. 2020 Aug 25. doi: 10.1093/rheumatology/keaa335.
FROM RHEUMATOLOGY
Late-onset neutropenia more common than expected in patients on rituximab
A new study has found that late-onset neutropenia is a notably common and occasionally serious occurrence in rituximab-treated patients with autoimmune diseases.
“The literature on late-onset neutropenia – or LON – has, to date, been limited in size and scope,” first author Reza Zonozi, MD, of Massachusetts General Hospital in Boston, said in an interview. “At the Vasculitis and Glomerulonephritis Center at Mass General, we’ve seen a number of cases of LON. Even though most are incidental and can be self-limiting, some can be severe and associated with sepsis. As such, we’ve come to appreciate it as one of the more concerning side effects of rituximab.
“Our hope was to offer a practical analysis of LON, how often it happens, and what it looks like,” he added, “as well as to share our approach to its management.” Their findings were published in Arthritis & Rheumatology.
To investigate the incidence, clinical features and outcomes of LON, the researchers launched a study of 738 adult patients with autoimmune diseases who were being treated with rituximab-induced continuous B-cell depletion. For the purposes of this study, LON was defined as an unexplained absolute neutrophil count of less than 1,000 cells/mcL during the period of B-cell depletion. Regarding disease type, 529 of the patients had antineutrophil cytoplasmic antibody–associated vasculitis (AAV), 73 had membranous nephropathy (MN), 59 had minimal change disease or focal segmental glomerulosclerosis (MCD/FSGS), 24 had lupus nephritis, and 53 had another autoimmune disease. Their average age was 58, and 53% were female.
All patients received a median of eight doses of rituximab – most commonly administered as one 1,000-mg IV dose every 4-6 months – and were in a state of B-cell depletion for a median of 2.5 years. Two months of low-dose daily oral cyclophosphamide was also used concurrently in 70% (n = 515) of patients. Glucocorticoids were used in 95% (n = 698) of patients.
During follow-up, 107 episodes of LON occurred in 71 patients. At 1, 2, and 5 years of continuous B-cell depletion, the incidence of LON was 6.6% (95% confidence interval, 5.0%-8.7%), 7.9% (95% CI, 6.1%-10.2%), and 13.5% (95% CI, 10.4%-17.4%), respectively. The first year following treatment initiation saw a much higher incidence rate of 7.2 per 100 person-years (95% CI, 5.4-9.6), compared with the rate thereafter of 1.5 per 100 person-years (95% CI, 1.0-2.3). LON occurred at a median of 4.1 months (interquartile range, 1.6-23.1) after the first rituximab infusion. The most common treatment for a LON episode was filgrastim.
Of the 107 episodes, 63 (59%) were asymptomatic. No infections were identified in asymptomatic episodes, while infections were identified in all symptomatic episodes. The most common symptom was a fever, and all 30 patients with LON and fever were hospitalized for management of febrile neutropenia. Four of the episodes included gingival soreness, and eight were complicated by sepsis. All the sepsis cases were resolved with standard therapy. One patient died with multiple relapsing LON.
Of the 71 patients with LON, 9 were not rechallenged with rituximab. A total of four of those patients had second LON episodes. Of the 62 patients who were rechallenged, 13 had second LON episodes over a median follow-up period of 2.4 years. The cumulative incidence of recurrent LON at 1, 2, and 5 years after rechallenge was 11.5% (95% CI, 5.6%-22.6%), 23.4% (95% CI, 13.8%-38.2%), and 30.4% (95% CI, 16.9%-50.9%), respectively.
Percentagewise, LON occurred significantly more often in patients with lupus nephritis (25%) than in patients with AAV (10.4%), MN (8.2%), or other diseases (7.6%) (P = .03). LON did not occur in any of the patients with MCD/FSGS. After multivariable analysis, lupus nephritis was associated with higher odds of developing LON (adjusted hazard ratio, 2.96; 95% CI, 1.10-8.01). A multivariable model also found that patients treated with cyclophosphamide and rituximab had higher odds of developing LON, compared with patients who did not receive cyclophosphamide (aHR, 1.98; 95% CI, 1.06-3.71).
Still more to learn about what leads to LON
“In large part, these findings quantify what our experience has been with LON in clinical practice,” Dr. Zonozi said. “It is indeed common, it’s often incidental, and most cases are reversible and respond well to treatment. But it can be associated with severe infections, including sepsis, and warrants close monitoring.”
In an interview, Md Yuzaiful Md Yusof, MBChB, PhD, observed that this incidence rate was notably higher than what he’d seen previously. Dr. Md Yusof presented at EULAR Congress 2015 on rituximab and LON, finding that 23 patients (2.5%) from a cohort of 912 developed rituximab-associated neutropenia.
“Most of our cases were in patients with rheumatoid arthritis,” he added, “so it may just be a difference in cohorts.”
Regardless, he applauded additional research in this area, noting that “the etiology of rituximab-associated LON is still unclear. The reasons behind this occurrence need investigating, particularly in regard to severe neutropenia cases. If we can find the predictors of those, it will be extremely helpful for the future of treatment.”
Dr. Zonozi agreed that “more investigation is needed to accurately define the mechanism of LON, which remains unknown. This will likely lead to more targeted strategies to both prevent and treat it.”
The authors acknowledged their study’s limitations, including being a single-center study that relied on retrospective data collection. They also acknowledged that, because the center is a nephrology-based practice, there was a low number of certain diseases like RA, opening up the possibility that “rates of LON are different” in those patients.
Two authors’ work on the study was funded by grants from the National Institutes of Health. The authors disclosed no potential conflicts of interest.
SOURCE: Zonozi R et al. Arthritis Rheumatol. 2020 Sep 6. doi: 10.1002/art.41501.
A new study has found that late-onset neutropenia is a notably common and occasionally serious occurrence in rituximab-treated patients with autoimmune diseases.
“The literature on late-onset neutropenia – or LON – has, to date, been limited in size and scope,” first author Reza Zonozi, MD, of Massachusetts General Hospital in Boston, said in an interview. “At the Vasculitis and Glomerulonephritis Center at Mass General, we’ve seen a number of cases of LON. Even though most are incidental and can be self-limiting, some can be severe and associated with sepsis. As such, we’ve come to appreciate it as one of the more concerning side effects of rituximab.
“Our hope was to offer a practical analysis of LON, how often it happens, and what it looks like,” he added, “as well as to share our approach to its management.” Their findings were published in Arthritis & Rheumatology.
To investigate the incidence, clinical features and outcomes of LON, the researchers launched a study of 738 adult patients with autoimmune diseases who were being treated with rituximab-induced continuous B-cell depletion. For the purposes of this study, LON was defined as an unexplained absolute neutrophil count of less than 1,000 cells/mcL during the period of B-cell depletion. Regarding disease type, 529 of the patients had antineutrophil cytoplasmic antibody–associated vasculitis (AAV), 73 had membranous nephropathy (MN), 59 had minimal change disease or focal segmental glomerulosclerosis (MCD/FSGS), 24 had lupus nephritis, and 53 had another autoimmune disease. Their average age was 58, and 53% were female.
All patients received a median of eight doses of rituximab – most commonly administered as one 1,000-mg IV dose every 4-6 months – and were in a state of B-cell depletion for a median of 2.5 years. Two months of low-dose daily oral cyclophosphamide was also used concurrently in 70% (n = 515) of patients. Glucocorticoids were used in 95% (n = 698) of patients.
During follow-up, 107 episodes of LON occurred in 71 patients. At 1, 2, and 5 years of continuous B-cell depletion, the incidence of LON was 6.6% (95% confidence interval, 5.0%-8.7%), 7.9% (95% CI, 6.1%-10.2%), and 13.5% (95% CI, 10.4%-17.4%), respectively. The first year following treatment initiation saw a much higher incidence rate of 7.2 per 100 person-years (95% CI, 5.4-9.6), compared with the rate thereafter of 1.5 per 100 person-years (95% CI, 1.0-2.3). LON occurred at a median of 4.1 months (interquartile range, 1.6-23.1) after the first rituximab infusion. The most common treatment for a LON episode was filgrastim.
Of the 107 episodes, 63 (59%) were asymptomatic. No infections were identified in asymptomatic episodes, while infections were identified in all symptomatic episodes. The most common symptom was a fever, and all 30 patients with LON and fever were hospitalized for management of febrile neutropenia. Four of the episodes included gingival soreness, and eight were complicated by sepsis. All the sepsis cases were resolved with standard therapy. One patient died with multiple relapsing LON.
Of the 71 patients with LON, 9 were not rechallenged with rituximab. A total of four of those patients had second LON episodes. Of the 62 patients who were rechallenged, 13 had second LON episodes over a median follow-up period of 2.4 years. The cumulative incidence of recurrent LON at 1, 2, and 5 years after rechallenge was 11.5% (95% CI, 5.6%-22.6%), 23.4% (95% CI, 13.8%-38.2%), and 30.4% (95% CI, 16.9%-50.9%), respectively.
Percentagewise, LON occurred significantly more often in patients with lupus nephritis (25%) than in patients with AAV (10.4%), MN (8.2%), or other diseases (7.6%) (P = .03). LON did not occur in any of the patients with MCD/FSGS. After multivariable analysis, lupus nephritis was associated with higher odds of developing LON (adjusted hazard ratio, 2.96; 95% CI, 1.10-8.01). A multivariable model also found that patients treated with cyclophosphamide and rituximab had higher odds of developing LON, compared with patients who did not receive cyclophosphamide (aHR, 1.98; 95% CI, 1.06-3.71).
Still more to learn about what leads to LON
“In large part, these findings quantify what our experience has been with LON in clinical practice,” Dr. Zonozi said. “It is indeed common, it’s often incidental, and most cases are reversible and respond well to treatment. But it can be associated with severe infections, including sepsis, and warrants close monitoring.”
In an interview, Md Yuzaiful Md Yusof, MBChB, PhD, observed that this incidence rate was notably higher than what he’d seen previously. Dr. Md Yusof presented at EULAR Congress 2015 on rituximab and LON, finding that 23 patients (2.5%) from a cohort of 912 developed rituximab-associated neutropenia.
“Most of our cases were in patients with rheumatoid arthritis,” he added, “so it may just be a difference in cohorts.”
Regardless, he applauded additional research in this area, noting that “the etiology of rituximab-associated LON is still unclear. The reasons behind this occurrence need investigating, particularly in regard to severe neutropenia cases. If we can find the predictors of those, it will be extremely helpful for the future of treatment.”
Dr. Zonozi agreed that “more investigation is needed to accurately define the mechanism of LON, which remains unknown. This will likely lead to more targeted strategies to both prevent and treat it.”
The authors acknowledged their study’s limitations, including being a single-center study that relied on retrospective data collection. They also acknowledged that, because the center is a nephrology-based practice, there was a low number of certain diseases like RA, opening up the possibility that “rates of LON are different” in those patients.
Two authors’ work on the study was funded by grants from the National Institutes of Health. The authors disclosed no potential conflicts of interest.
SOURCE: Zonozi R et al. Arthritis Rheumatol. 2020 Sep 6. doi: 10.1002/art.41501.
A new study has found that late-onset neutropenia is a notably common and occasionally serious occurrence in rituximab-treated patients with autoimmune diseases.
“The literature on late-onset neutropenia – or LON – has, to date, been limited in size and scope,” first author Reza Zonozi, MD, of Massachusetts General Hospital in Boston, said in an interview. “At the Vasculitis and Glomerulonephritis Center at Mass General, we’ve seen a number of cases of LON. Even though most are incidental and can be self-limiting, some can be severe and associated with sepsis. As such, we’ve come to appreciate it as one of the more concerning side effects of rituximab.
“Our hope was to offer a practical analysis of LON, how often it happens, and what it looks like,” he added, “as well as to share our approach to its management.” Their findings were published in Arthritis & Rheumatology.
To investigate the incidence, clinical features and outcomes of LON, the researchers launched a study of 738 adult patients with autoimmune diseases who were being treated with rituximab-induced continuous B-cell depletion. For the purposes of this study, LON was defined as an unexplained absolute neutrophil count of less than 1,000 cells/mcL during the period of B-cell depletion. Regarding disease type, 529 of the patients had antineutrophil cytoplasmic antibody–associated vasculitis (AAV), 73 had membranous nephropathy (MN), 59 had minimal change disease or focal segmental glomerulosclerosis (MCD/FSGS), 24 had lupus nephritis, and 53 had another autoimmune disease. Their average age was 58, and 53% were female.
All patients received a median of eight doses of rituximab – most commonly administered as one 1,000-mg IV dose every 4-6 months – and were in a state of B-cell depletion for a median of 2.5 years. Two months of low-dose daily oral cyclophosphamide was also used concurrently in 70% (n = 515) of patients. Glucocorticoids were used in 95% (n = 698) of patients.
During follow-up, 107 episodes of LON occurred in 71 patients. At 1, 2, and 5 years of continuous B-cell depletion, the incidence of LON was 6.6% (95% confidence interval, 5.0%-8.7%), 7.9% (95% CI, 6.1%-10.2%), and 13.5% (95% CI, 10.4%-17.4%), respectively. The first year following treatment initiation saw a much higher incidence rate of 7.2 per 100 person-years (95% CI, 5.4-9.6), compared with the rate thereafter of 1.5 per 100 person-years (95% CI, 1.0-2.3). LON occurred at a median of 4.1 months (interquartile range, 1.6-23.1) after the first rituximab infusion. The most common treatment for a LON episode was filgrastim.
Of the 107 episodes, 63 (59%) were asymptomatic. No infections were identified in asymptomatic episodes, while infections were identified in all symptomatic episodes. The most common symptom was a fever, and all 30 patients with LON and fever were hospitalized for management of febrile neutropenia. Four of the episodes included gingival soreness, and eight were complicated by sepsis. All the sepsis cases were resolved with standard therapy. One patient died with multiple relapsing LON.
Of the 71 patients with LON, 9 were not rechallenged with rituximab. A total of four of those patients had second LON episodes. Of the 62 patients who were rechallenged, 13 had second LON episodes over a median follow-up period of 2.4 years. The cumulative incidence of recurrent LON at 1, 2, and 5 years after rechallenge was 11.5% (95% CI, 5.6%-22.6%), 23.4% (95% CI, 13.8%-38.2%), and 30.4% (95% CI, 16.9%-50.9%), respectively.
Percentagewise, LON occurred significantly more often in patients with lupus nephritis (25%) than in patients with AAV (10.4%), MN (8.2%), or other diseases (7.6%) (P = .03). LON did not occur in any of the patients with MCD/FSGS. After multivariable analysis, lupus nephritis was associated with higher odds of developing LON (adjusted hazard ratio, 2.96; 95% CI, 1.10-8.01). A multivariable model also found that patients treated with cyclophosphamide and rituximab had higher odds of developing LON, compared with patients who did not receive cyclophosphamide (aHR, 1.98; 95% CI, 1.06-3.71).
Still more to learn about what leads to LON
“In large part, these findings quantify what our experience has been with LON in clinical practice,” Dr. Zonozi said. “It is indeed common, it’s often incidental, and most cases are reversible and respond well to treatment. But it can be associated with severe infections, including sepsis, and warrants close monitoring.”
In an interview, Md Yuzaiful Md Yusof, MBChB, PhD, observed that this incidence rate was notably higher than what he’d seen previously. Dr. Md Yusof presented at EULAR Congress 2015 on rituximab and LON, finding that 23 patients (2.5%) from a cohort of 912 developed rituximab-associated neutropenia.
“Most of our cases were in patients with rheumatoid arthritis,” he added, “so it may just be a difference in cohorts.”
Regardless, he applauded additional research in this area, noting that “the etiology of rituximab-associated LON is still unclear. The reasons behind this occurrence need investigating, particularly in regard to severe neutropenia cases. If we can find the predictors of those, it will be extremely helpful for the future of treatment.”
Dr. Zonozi agreed that “more investigation is needed to accurately define the mechanism of LON, which remains unknown. This will likely lead to more targeted strategies to both prevent and treat it.”
The authors acknowledged their study’s limitations, including being a single-center study that relied on retrospective data collection. They also acknowledged that, because the center is a nephrology-based practice, there was a low number of certain diseases like RA, opening up the possibility that “rates of LON are different” in those patients.
Two authors’ work on the study was funded by grants from the National Institutes of Health. The authors disclosed no potential conflicts of interest.
SOURCE: Zonozi R et al. Arthritis Rheumatol. 2020 Sep 6. doi: 10.1002/art.41501.
FROM ARTHRITIS & RHEUMATOLOGY
High disability after a year of RA treatment signals increased mortality risk
over the course of up to 10 years of follow-up, according to an analysis of patients enrolled in the Canadian Early Arthritis Cohort (CATCH).
Higher Disease Activity Score in 28 joints (DAS28) at follow-up was also associated with higher all-cause mortality among the patients, who all took at least one conventional synthetic or biologic disease-modifying antirheumatic drug during the first year. Higher DAS28 scores in previous studies has been associated with increased disability as measured by the HAQ, Safoora Fatima, MD, of the University of Western Ontario, London, and colleagues wrote in Arthritis & Rheumatology.
“Combining our study findings with this association suggests that poorer disease control (high DAS28) within the first treatment year for RA may lead to increased disability (high HAQ scores) which in turn may contribute to higher mortality. This may indicate that RA patients who do not have a deep response in the first year to treatment have higher subsequent mortality,” the researchers wrote.
In addition to higher HAQ scores, all-cause mortality was independently associated with age, male sex, lower education, smoking, more comorbidities, higher baseline disease activity, and glucocorticoid use. “This is helpful in a clinical setting as it can guide physician-patient discussions in terms of risk factors associated with prognosis, prescribing glucocorticoids, counseling on smoking cessation, monitoring treatment responses, and focusing on patient education,” the authors wrote.
While the impact of increased disease activity and damage likely plays a role in the association between high HAQ score and increased mortality, the authors noted that “comorbidities could be causing deaths and those with comorbidities in [early RA] have less chance of remission and more functional impairment at 1 year versus those without any comorbidities, as has been shown [before] in the CATCH [early RA] cohort.”
Dr. Fatima and associates studied 1,724 patients with RA who had a symptom duration of less than 1 year at the time of enrollment in CATCH during 2007-2017. These patients had a mean age of 55 years, and 72% were women. Over the 10-year follow up period, 62 patients (2.4%) died. HAQ scores proved to be significantly higher at both baseline and 1 year for those who died, going from 1.2 to 0.9, compared with scores moving from 1.0 to 0.5 among patients who did not die. (The HAQ has eight categories that are each scored 0-3, with 0 meaning no self-reported functional impairment and 3 meaning severe functional impairment.) Similarly, DAS28 scores were significantly higher at both time points for patients who died versus those who lived, declining from 5.4 to 3.6 for deceased and from 4.9 to 2.8 for nondeceased patients in a year.
Whereas HAQ at baseline was not significantly associated with all-cause mortality in a multivariate, discrete-time survival model that adjusted for age, gender, comorbidities, disease activity, smoking, education, seropositivity, symptom duration, and glucocorticoid use, the association between HAQ at 1 year and death remained statistically significant with a hazard ratio of 1.87.
The authors noted that potential confounders may not have been adjusted for in the comparisons, such as “variable access to advanced therapies, other comorbidities not in the standardized comorbidity questionnaire, [and] severity of comorbidities.”
CATCH has been funded over many years by multiple companies including Amgen and Pfizer Canada, AbbVie, Medexus, Eli Lilly Canada, Merck Canada, Sandoz, Hoffman–La Roche, Janssen, UCB Canada, Bristol-Myers Squibb Canada, and Sanofi Genzyme. The authors had no disclosures.
SOURCE: Fatima S et al. Arthritis Rheumatol. 2020 Sep 6. doi: 10.1002/art.41513.
over the course of up to 10 years of follow-up, according to an analysis of patients enrolled in the Canadian Early Arthritis Cohort (CATCH).
Higher Disease Activity Score in 28 joints (DAS28) at follow-up was also associated with higher all-cause mortality among the patients, who all took at least one conventional synthetic or biologic disease-modifying antirheumatic drug during the first year. Higher DAS28 scores in previous studies has been associated with increased disability as measured by the HAQ, Safoora Fatima, MD, of the University of Western Ontario, London, and colleagues wrote in Arthritis & Rheumatology.
“Combining our study findings with this association suggests that poorer disease control (high DAS28) within the first treatment year for RA may lead to increased disability (high HAQ scores) which in turn may contribute to higher mortality. This may indicate that RA patients who do not have a deep response in the first year to treatment have higher subsequent mortality,” the researchers wrote.
In addition to higher HAQ scores, all-cause mortality was independently associated with age, male sex, lower education, smoking, more comorbidities, higher baseline disease activity, and glucocorticoid use. “This is helpful in a clinical setting as it can guide physician-patient discussions in terms of risk factors associated with prognosis, prescribing glucocorticoids, counseling on smoking cessation, monitoring treatment responses, and focusing on patient education,” the authors wrote.
While the impact of increased disease activity and damage likely plays a role in the association between high HAQ score and increased mortality, the authors noted that “comorbidities could be causing deaths and those with comorbidities in [early RA] have less chance of remission and more functional impairment at 1 year versus those without any comorbidities, as has been shown [before] in the CATCH [early RA] cohort.”
Dr. Fatima and associates studied 1,724 patients with RA who had a symptom duration of less than 1 year at the time of enrollment in CATCH during 2007-2017. These patients had a mean age of 55 years, and 72% were women. Over the 10-year follow up period, 62 patients (2.4%) died. HAQ scores proved to be significantly higher at both baseline and 1 year for those who died, going from 1.2 to 0.9, compared with scores moving from 1.0 to 0.5 among patients who did not die. (The HAQ has eight categories that are each scored 0-3, with 0 meaning no self-reported functional impairment and 3 meaning severe functional impairment.) Similarly, DAS28 scores were significantly higher at both time points for patients who died versus those who lived, declining from 5.4 to 3.6 for deceased and from 4.9 to 2.8 for nondeceased patients in a year.
Whereas HAQ at baseline was not significantly associated with all-cause mortality in a multivariate, discrete-time survival model that adjusted for age, gender, comorbidities, disease activity, smoking, education, seropositivity, symptom duration, and glucocorticoid use, the association between HAQ at 1 year and death remained statistically significant with a hazard ratio of 1.87.
The authors noted that potential confounders may not have been adjusted for in the comparisons, such as “variable access to advanced therapies, other comorbidities not in the standardized comorbidity questionnaire, [and] severity of comorbidities.”
CATCH has been funded over many years by multiple companies including Amgen and Pfizer Canada, AbbVie, Medexus, Eli Lilly Canada, Merck Canada, Sandoz, Hoffman–La Roche, Janssen, UCB Canada, Bristol-Myers Squibb Canada, and Sanofi Genzyme. The authors had no disclosures.
SOURCE: Fatima S et al. Arthritis Rheumatol. 2020 Sep 6. doi: 10.1002/art.41513.
over the course of up to 10 years of follow-up, according to an analysis of patients enrolled in the Canadian Early Arthritis Cohort (CATCH).
Higher Disease Activity Score in 28 joints (DAS28) at follow-up was also associated with higher all-cause mortality among the patients, who all took at least one conventional synthetic or biologic disease-modifying antirheumatic drug during the first year. Higher DAS28 scores in previous studies has been associated with increased disability as measured by the HAQ, Safoora Fatima, MD, of the University of Western Ontario, London, and colleagues wrote in Arthritis & Rheumatology.
“Combining our study findings with this association suggests that poorer disease control (high DAS28) within the first treatment year for RA may lead to increased disability (high HAQ scores) which in turn may contribute to higher mortality. This may indicate that RA patients who do not have a deep response in the first year to treatment have higher subsequent mortality,” the researchers wrote.
In addition to higher HAQ scores, all-cause mortality was independently associated with age, male sex, lower education, smoking, more comorbidities, higher baseline disease activity, and glucocorticoid use. “This is helpful in a clinical setting as it can guide physician-patient discussions in terms of risk factors associated with prognosis, prescribing glucocorticoids, counseling on smoking cessation, monitoring treatment responses, and focusing on patient education,” the authors wrote.
While the impact of increased disease activity and damage likely plays a role in the association between high HAQ score and increased mortality, the authors noted that “comorbidities could be causing deaths and those with comorbidities in [early RA] have less chance of remission and more functional impairment at 1 year versus those without any comorbidities, as has been shown [before] in the CATCH [early RA] cohort.”
Dr. Fatima and associates studied 1,724 patients with RA who had a symptom duration of less than 1 year at the time of enrollment in CATCH during 2007-2017. These patients had a mean age of 55 years, and 72% were women. Over the 10-year follow up period, 62 patients (2.4%) died. HAQ scores proved to be significantly higher at both baseline and 1 year for those who died, going from 1.2 to 0.9, compared with scores moving from 1.0 to 0.5 among patients who did not die. (The HAQ has eight categories that are each scored 0-3, with 0 meaning no self-reported functional impairment and 3 meaning severe functional impairment.) Similarly, DAS28 scores were significantly higher at both time points for patients who died versus those who lived, declining from 5.4 to 3.6 for deceased and from 4.9 to 2.8 for nondeceased patients in a year.
Whereas HAQ at baseline was not significantly associated with all-cause mortality in a multivariate, discrete-time survival model that adjusted for age, gender, comorbidities, disease activity, smoking, education, seropositivity, symptom duration, and glucocorticoid use, the association between HAQ at 1 year and death remained statistically significant with a hazard ratio of 1.87.
The authors noted that potential confounders may not have been adjusted for in the comparisons, such as “variable access to advanced therapies, other comorbidities not in the standardized comorbidity questionnaire, [and] severity of comorbidities.”
CATCH has been funded over many years by multiple companies including Amgen and Pfizer Canada, AbbVie, Medexus, Eli Lilly Canada, Merck Canada, Sandoz, Hoffman–La Roche, Janssen, UCB Canada, Bristol-Myers Squibb Canada, and Sanofi Genzyme. The authors had no disclosures.
SOURCE: Fatima S et al. Arthritis Rheumatol. 2020 Sep 6. doi: 10.1002/art.41513.
FROM ARTHRITIS & RHEUMATOLOGY
TNF inhibitors linked to inflammatory CNS events
, new research suggests
The nested case-control study included more than 200 participants with diseases such as rheumatoid arthritis, psoriasis, and Crohn’s disease. Results showed that exposure to TNF inhibitors was significantly associated with increased risk for demyelinating CNS events, such as multiple sclerosis, and nondemyelinating events, such as meningitis and encephalitis.
Interestingly, disease-specific secondary analyses showed that the strongest association for inflammatory events was in patients with rheumatoid arthritis.
Lead author Amy Kunchok, MD, of Mayo Clinic, Rochester, Minn., noted that “these are highly effective therapies for patients” and that these CNS events are likely uncommon.
“Our study has observed an association, but this does not imply causality. Therefore, we are not cautioning against using these therapies in appropriate patients,” Dr. Kunchok said in an interview.
“Rather, we recommend that clinicians assessing patients with both inflammatory demyelinating and nondemyelinating CNS events consider a detailed evaluation of the medication history, particularly in patients with coexistent autoimmune diseases who may have a current or past history of biological therapies,” she said.
The findings were published in JAMA Neurology.
Poorly understood
TNF inhibitors “are common therapies for certain autoimmune diseases,” the investigators noted.
Previously, a link between exposure to these inhibitors and inflammatory CNS events “has been postulated but is poorly understood,” they wrote.
In the current study, they examined records for 106 patients who were treated at Mayo clinics in Minnesota, Arizona, or Florida from January 2003 through February 2019. All participants had been diagnosed with an autoimmune disease that the Food and Drug Administration has listed as an indication for TNF inhibitor use. This included rheumatoid arthritis (n = 48), ankylosing spondylitis (n = 4), psoriasis and psoriatic arthritis (n = 21), Crohn’s disease (n = 27), and ulcerative colitis (n = 6). Their records also showed diagnostic codes for the inflammatory demyelinating CNS events of relapsing-remitting or primary progressive MS, clinically isolated syndrome, radiologically isolated syndrome, neuromyelitis optica spectrum disorder, and transverse myelitis or for the inflammatory nondemyelinating CNS events of meningitis, meningoencephalitis, encephalitis, neurosarcoidosis, and CNS vasculitis. The investigators also included 106 age-, sex-, and autoimmune disease–matched participants 1:1 to act as the control group.
In the total study population, 64% were women and the median age at disease onset was 52 years. In addition, 60% of the patient group and 40% of the control group were exposed to TNF inhibitors.
Novel finding?
Results showed that TNF inhibitor exposure was significantly linked to increased risk for developing any inflammatory CNS event (adjusted odds ratio, 3.01; 95% CI, 1.55-5.82; P = .001). When the outcomes were stratified by class of inflammatory event, these results were similar. The aOR was 3.09 (95% CI, 1.19-8.04; P = .02) for inflammatory demyelinating CNS events and was 2.97 (95% CI, 1.15-7.65; P = .02) for inflammatory nondemyelinating events.
Dr. Kunchok noted that the association between the inhibitors and nondemyelinating events was “a novel finding from this study.”
In secondary analyses, patients with rheumatoid arthritis and exposure to TNF inhibitors had the strongest association with any inflammatory CNS event (aOR, 4.82; 95% CI, 1.62-14.36; P = .005).
A pooled cohort comprising only the participants with the other autoimmune diseases did not show a significant association between exposure to TNF inhibitors and development of CNS events (P = .09).
“Because of the lack of power, further stratification by individual autoimmune diseases was not analyzed,” the investigators reported.
Although the overall findings showed that exposure to TNF inhibitors was linked to increased risk for inflammatory events, whether this association “represents de novo or exacerbated inflammatory pathways requires further research,” the authors wrote.
Dr. Kunchok added that more research, especially population-based studies, is also needed to examine the incidence of these inflammatory CNS events in patients exposed to TNF-alpha inhibitors.
Adds to the literature
In an accompanying editorial, Jeffrey M. Gelfand, MD, department of neurology at the University of California, San Francisco, and Jinoos Yazdany, MD, Zuckerberg San Francisco General Hospital at UCSF, noted that although the study adds to the literature, the magnitude of the risk found “remains unclear.”
“Randomized clinical trials are not suited to the study of rare adverse events,” Dr. Gelfand and Dr. Yazdany wrote. They agree with Dr. Kunchok that “next steps should include population-based observational studies that control for disease severity.”
Still, the current study provides additional evidence of rare adverse events in patients receiving TNF inhibitors, they noted. So how should prescribers proceed?
“As with all treatments, the risk-benefit ratio for the individual patient’s situation must be weighed and appropriate counseling must be given to facilitate shared decision-making discussions,” wrote the editorialists.
“Given what is known about the risk of harm, avoiding TNF inhibitors is advisable in patients with known MS,” they wrote.
In addition, neurologic consultation can be helpful for clarifying diagnoses and providing advice on monitoring strategies for TNF inhibitor treatment in those with possible MS or other demyelinating conditions, noted the editorialists.
“In patients who develop new concerning neurological symptoms while receiving TNF inhibitor treatment, timely evaluation is indicated, including consideration of neuroinflammatory, infectious, and neurological diagnoses that may be unrelated to treatment,” they added.
“Broader awareness of risks that studies such as this one by Kunchok et al provide can ... encourage timelier recognition of potential TNF inhibitor–associated neuroinflammatory events and may improve outcomes for patients,” Dr. Gelfand and Dr. Yazdany concluded.
The study was funded by a grant from the National Center for Advancing Translational Sciences. Dr. Kunchok reports having received research funding from Biogen outside this study. A full list of disclosures for the other study authors is in the original article. Dr. Gelfand reports having received g rants for a clinical trial from Genentech and consulting fees from Biogen, Alexion, Theranica, Impel Neuropharma, Advanced Clinical, Biohaven, and Satsuma. Dr. Yazdany reports having received grants from Pfizer and consulting fees from AstraZeneca and Eli Lilly outside the submitted work.
A version of this article originally appeared on Medscape.com.
, new research suggests
The nested case-control study included more than 200 participants with diseases such as rheumatoid arthritis, psoriasis, and Crohn’s disease. Results showed that exposure to TNF inhibitors was significantly associated with increased risk for demyelinating CNS events, such as multiple sclerosis, and nondemyelinating events, such as meningitis and encephalitis.
Interestingly, disease-specific secondary analyses showed that the strongest association for inflammatory events was in patients with rheumatoid arthritis.
Lead author Amy Kunchok, MD, of Mayo Clinic, Rochester, Minn., noted that “these are highly effective therapies for patients” and that these CNS events are likely uncommon.
“Our study has observed an association, but this does not imply causality. Therefore, we are not cautioning against using these therapies in appropriate patients,” Dr. Kunchok said in an interview.
“Rather, we recommend that clinicians assessing patients with both inflammatory demyelinating and nondemyelinating CNS events consider a detailed evaluation of the medication history, particularly in patients with coexistent autoimmune diseases who may have a current or past history of biological therapies,” she said.
The findings were published in JAMA Neurology.
Poorly understood
TNF inhibitors “are common therapies for certain autoimmune diseases,” the investigators noted.
Previously, a link between exposure to these inhibitors and inflammatory CNS events “has been postulated but is poorly understood,” they wrote.
In the current study, they examined records for 106 patients who were treated at Mayo clinics in Minnesota, Arizona, or Florida from January 2003 through February 2019. All participants had been diagnosed with an autoimmune disease that the Food and Drug Administration has listed as an indication for TNF inhibitor use. This included rheumatoid arthritis (n = 48), ankylosing spondylitis (n = 4), psoriasis and psoriatic arthritis (n = 21), Crohn’s disease (n = 27), and ulcerative colitis (n = 6). Their records also showed diagnostic codes for the inflammatory demyelinating CNS events of relapsing-remitting or primary progressive MS, clinically isolated syndrome, radiologically isolated syndrome, neuromyelitis optica spectrum disorder, and transverse myelitis or for the inflammatory nondemyelinating CNS events of meningitis, meningoencephalitis, encephalitis, neurosarcoidosis, and CNS vasculitis. The investigators also included 106 age-, sex-, and autoimmune disease–matched participants 1:1 to act as the control group.
In the total study population, 64% were women and the median age at disease onset was 52 years. In addition, 60% of the patient group and 40% of the control group were exposed to TNF inhibitors.
Novel finding?
Results showed that TNF inhibitor exposure was significantly linked to increased risk for developing any inflammatory CNS event (adjusted odds ratio, 3.01; 95% CI, 1.55-5.82; P = .001). When the outcomes were stratified by class of inflammatory event, these results were similar. The aOR was 3.09 (95% CI, 1.19-8.04; P = .02) for inflammatory demyelinating CNS events and was 2.97 (95% CI, 1.15-7.65; P = .02) for inflammatory nondemyelinating events.
Dr. Kunchok noted that the association between the inhibitors and nondemyelinating events was “a novel finding from this study.”
In secondary analyses, patients with rheumatoid arthritis and exposure to TNF inhibitors had the strongest association with any inflammatory CNS event (aOR, 4.82; 95% CI, 1.62-14.36; P = .005).
A pooled cohort comprising only the participants with the other autoimmune diseases did not show a significant association between exposure to TNF inhibitors and development of CNS events (P = .09).
“Because of the lack of power, further stratification by individual autoimmune diseases was not analyzed,” the investigators reported.
Although the overall findings showed that exposure to TNF inhibitors was linked to increased risk for inflammatory events, whether this association “represents de novo or exacerbated inflammatory pathways requires further research,” the authors wrote.
Dr. Kunchok added that more research, especially population-based studies, is also needed to examine the incidence of these inflammatory CNS events in patients exposed to TNF-alpha inhibitors.
Adds to the literature
In an accompanying editorial, Jeffrey M. Gelfand, MD, department of neurology at the University of California, San Francisco, and Jinoos Yazdany, MD, Zuckerberg San Francisco General Hospital at UCSF, noted that although the study adds to the literature, the magnitude of the risk found “remains unclear.”
“Randomized clinical trials are not suited to the study of rare adverse events,” Dr. Gelfand and Dr. Yazdany wrote. They agree with Dr. Kunchok that “next steps should include population-based observational studies that control for disease severity.”
Still, the current study provides additional evidence of rare adverse events in patients receiving TNF inhibitors, they noted. So how should prescribers proceed?
“As with all treatments, the risk-benefit ratio for the individual patient’s situation must be weighed and appropriate counseling must be given to facilitate shared decision-making discussions,” wrote the editorialists.
“Given what is known about the risk of harm, avoiding TNF inhibitors is advisable in patients with known MS,” they wrote.
In addition, neurologic consultation can be helpful for clarifying diagnoses and providing advice on monitoring strategies for TNF inhibitor treatment in those with possible MS or other demyelinating conditions, noted the editorialists.
“In patients who develop new concerning neurological symptoms while receiving TNF inhibitor treatment, timely evaluation is indicated, including consideration of neuroinflammatory, infectious, and neurological diagnoses that may be unrelated to treatment,” they added.
“Broader awareness of risks that studies such as this one by Kunchok et al provide can ... encourage timelier recognition of potential TNF inhibitor–associated neuroinflammatory events and may improve outcomes for patients,” Dr. Gelfand and Dr. Yazdany concluded.
The study was funded by a grant from the National Center for Advancing Translational Sciences. Dr. Kunchok reports having received research funding from Biogen outside this study. A full list of disclosures for the other study authors is in the original article. Dr. Gelfand reports having received g rants for a clinical trial from Genentech and consulting fees from Biogen, Alexion, Theranica, Impel Neuropharma, Advanced Clinical, Biohaven, and Satsuma. Dr. Yazdany reports having received grants from Pfizer and consulting fees from AstraZeneca and Eli Lilly outside the submitted work.
A version of this article originally appeared on Medscape.com.
, new research suggests
The nested case-control study included more than 200 participants with diseases such as rheumatoid arthritis, psoriasis, and Crohn’s disease. Results showed that exposure to TNF inhibitors was significantly associated with increased risk for demyelinating CNS events, such as multiple sclerosis, and nondemyelinating events, such as meningitis and encephalitis.
Interestingly, disease-specific secondary analyses showed that the strongest association for inflammatory events was in patients with rheumatoid arthritis.
Lead author Amy Kunchok, MD, of Mayo Clinic, Rochester, Minn., noted that “these are highly effective therapies for patients” and that these CNS events are likely uncommon.
“Our study has observed an association, but this does not imply causality. Therefore, we are not cautioning against using these therapies in appropriate patients,” Dr. Kunchok said in an interview.
“Rather, we recommend that clinicians assessing patients with both inflammatory demyelinating and nondemyelinating CNS events consider a detailed evaluation of the medication history, particularly in patients with coexistent autoimmune diseases who may have a current or past history of biological therapies,” she said.
The findings were published in JAMA Neurology.
Poorly understood
TNF inhibitors “are common therapies for certain autoimmune diseases,” the investigators noted.
Previously, a link between exposure to these inhibitors and inflammatory CNS events “has been postulated but is poorly understood,” they wrote.
In the current study, they examined records for 106 patients who were treated at Mayo clinics in Minnesota, Arizona, or Florida from January 2003 through February 2019. All participants had been diagnosed with an autoimmune disease that the Food and Drug Administration has listed as an indication for TNF inhibitor use. This included rheumatoid arthritis (n = 48), ankylosing spondylitis (n = 4), psoriasis and psoriatic arthritis (n = 21), Crohn’s disease (n = 27), and ulcerative colitis (n = 6). Their records also showed diagnostic codes for the inflammatory demyelinating CNS events of relapsing-remitting or primary progressive MS, clinically isolated syndrome, radiologically isolated syndrome, neuromyelitis optica spectrum disorder, and transverse myelitis or for the inflammatory nondemyelinating CNS events of meningitis, meningoencephalitis, encephalitis, neurosarcoidosis, and CNS vasculitis. The investigators also included 106 age-, sex-, and autoimmune disease–matched participants 1:1 to act as the control group.
In the total study population, 64% were women and the median age at disease onset was 52 years. In addition, 60% of the patient group and 40% of the control group were exposed to TNF inhibitors.
Novel finding?
Results showed that TNF inhibitor exposure was significantly linked to increased risk for developing any inflammatory CNS event (adjusted odds ratio, 3.01; 95% CI, 1.55-5.82; P = .001). When the outcomes were stratified by class of inflammatory event, these results were similar. The aOR was 3.09 (95% CI, 1.19-8.04; P = .02) for inflammatory demyelinating CNS events and was 2.97 (95% CI, 1.15-7.65; P = .02) for inflammatory nondemyelinating events.
Dr. Kunchok noted that the association between the inhibitors and nondemyelinating events was “a novel finding from this study.”
In secondary analyses, patients with rheumatoid arthritis and exposure to TNF inhibitors had the strongest association with any inflammatory CNS event (aOR, 4.82; 95% CI, 1.62-14.36; P = .005).
A pooled cohort comprising only the participants with the other autoimmune diseases did not show a significant association between exposure to TNF inhibitors and development of CNS events (P = .09).
“Because of the lack of power, further stratification by individual autoimmune diseases was not analyzed,” the investigators reported.
Although the overall findings showed that exposure to TNF inhibitors was linked to increased risk for inflammatory events, whether this association “represents de novo or exacerbated inflammatory pathways requires further research,” the authors wrote.
Dr. Kunchok added that more research, especially population-based studies, is also needed to examine the incidence of these inflammatory CNS events in patients exposed to TNF-alpha inhibitors.
Adds to the literature
In an accompanying editorial, Jeffrey M. Gelfand, MD, department of neurology at the University of California, San Francisco, and Jinoos Yazdany, MD, Zuckerberg San Francisco General Hospital at UCSF, noted that although the study adds to the literature, the magnitude of the risk found “remains unclear.”
“Randomized clinical trials are not suited to the study of rare adverse events,” Dr. Gelfand and Dr. Yazdany wrote. They agree with Dr. Kunchok that “next steps should include population-based observational studies that control for disease severity.”
Still, the current study provides additional evidence of rare adverse events in patients receiving TNF inhibitors, they noted. So how should prescribers proceed?
“As with all treatments, the risk-benefit ratio for the individual patient’s situation must be weighed and appropriate counseling must be given to facilitate shared decision-making discussions,” wrote the editorialists.
“Given what is known about the risk of harm, avoiding TNF inhibitors is advisable in patients with known MS,” they wrote.
In addition, neurologic consultation can be helpful for clarifying diagnoses and providing advice on monitoring strategies for TNF inhibitor treatment in those with possible MS or other demyelinating conditions, noted the editorialists.
“In patients who develop new concerning neurological symptoms while receiving TNF inhibitor treatment, timely evaluation is indicated, including consideration of neuroinflammatory, infectious, and neurological diagnoses that may be unrelated to treatment,” they added.
“Broader awareness of risks that studies such as this one by Kunchok et al provide can ... encourage timelier recognition of potential TNF inhibitor–associated neuroinflammatory events and may improve outcomes for patients,” Dr. Gelfand and Dr. Yazdany concluded.
The study was funded by a grant from the National Center for Advancing Translational Sciences. Dr. Kunchok reports having received research funding from Biogen outside this study. A full list of disclosures for the other study authors is in the original article. Dr. Gelfand reports having received g rants for a clinical trial from Genentech and consulting fees from Biogen, Alexion, Theranica, Impel Neuropharma, Advanced Clinical, Biohaven, and Satsuma. Dr. Yazdany reports having received grants from Pfizer and consulting fees from AstraZeneca and Eli Lilly outside the submitted work.
A version of this article originally appeared on Medscape.com.
Humira topped drug-revenue list for 2019
Humira outsold all other drugs in 2019 in terms of revenue as cytokine inhibitor medications earned their way to three of the first four spots on the pharmaceutical best-seller list, according to a new analysis from the IQVIA Institute for Human Data Science.
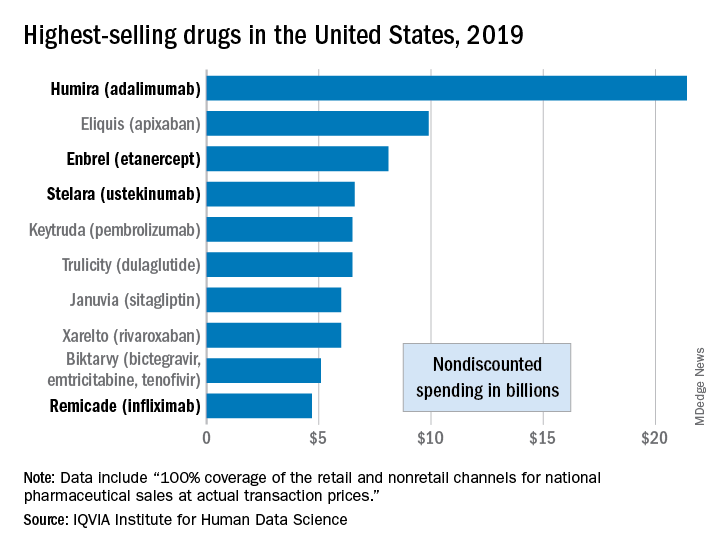
Sales of Humira (adalimumab) amounted to $21.4 billion before discounting, Murray Aitken, the institute’s executive director, and associates wrote in their analysis. That’s more than double the total of the anticoagulant Eliquis (apixaban), which brought in $9.9 billion in its last year before generic forms became available.
The next two spots were filled by the tumor necrosis factor inhibitor Enbrel (etanercept) with $8.1 billion in sales and the interleukin 12/23 inhibitor Stelara (ustekinumab) with sales totaling $6.6 billion, followed by the chemotherapy drug Keytruda (pembrolizumab) close behind after racking up $6.5 billion in sales, the researchers reported.
Total nondiscounted spending on all drugs in the U.S. market came to $511 billion in 2019, an increase of 5.7% over the $484 billion spent in 2018, based on data from the July 2020 IQVIA National Sales Perspectives.
These figures are “not adjusted for estimates of off-invoice discounts and rebates,” the authors noted, but they include “prescription and insulin products sold into chain and independent pharmacies, food store pharmacies, mail service pharmacies, long-term care facilities, hospitals, clinics, and other institutional settings.”
Those “discounts and rebates” do exist, however, and they can add up. Drug sales for 2019, “after deducting negotiated rebates, discounts, and other forms of price concessions, such as patient coupons or vouchers that offset out-of-pocket costs,” were $235 billion less than overall nondiscounted spending, the report noted.
Now that we’ve shown you the money, let’s take a quick look at volume. The leading drugs by number of dispensed prescriptions in 2019 were, not surprisingly, quite different. First, with 118 million prescriptions, was atorvastatin, followed by levothyroxine (113 million), lisinopril (96), amlodipine (89), and metoprolol (85), Mr. Aitken and associates reported.
Altogether, over 4.2 billion prescriptions were dispensed last year, with a couple of caveats: 90-day and 30-day fills were both counted as one prescription, and OTC drugs were not included, they pointed out.
Humira outsold all other drugs in 2019 in terms of revenue as cytokine inhibitor medications earned their way to three of the first four spots on the pharmaceutical best-seller list, according to a new analysis from the IQVIA Institute for Human Data Science.

Sales of Humira (adalimumab) amounted to $21.4 billion before discounting, Murray Aitken, the institute’s executive director, and associates wrote in their analysis. That’s more than double the total of the anticoagulant Eliquis (apixaban), which brought in $9.9 billion in its last year before generic forms became available.
The next two spots were filled by the tumor necrosis factor inhibitor Enbrel (etanercept) with $8.1 billion in sales and the interleukin 12/23 inhibitor Stelara (ustekinumab) with sales totaling $6.6 billion, followed by the chemotherapy drug Keytruda (pembrolizumab) close behind after racking up $6.5 billion in sales, the researchers reported.
Total nondiscounted spending on all drugs in the U.S. market came to $511 billion in 2019, an increase of 5.7% over the $484 billion spent in 2018, based on data from the July 2020 IQVIA National Sales Perspectives.
These figures are “not adjusted for estimates of off-invoice discounts and rebates,” the authors noted, but they include “prescription and insulin products sold into chain and independent pharmacies, food store pharmacies, mail service pharmacies, long-term care facilities, hospitals, clinics, and other institutional settings.”
Those “discounts and rebates” do exist, however, and they can add up. Drug sales for 2019, “after deducting negotiated rebates, discounts, and other forms of price concessions, such as patient coupons or vouchers that offset out-of-pocket costs,” were $235 billion less than overall nondiscounted spending, the report noted.
Now that we’ve shown you the money, let’s take a quick look at volume. The leading drugs by number of dispensed prescriptions in 2019 were, not surprisingly, quite different. First, with 118 million prescriptions, was atorvastatin, followed by levothyroxine (113 million), lisinopril (96), amlodipine (89), and metoprolol (85), Mr. Aitken and associates reported.
Altogether, over 4.2 billion prescriptions were dispensed last year, with a couple of caveats: 90-day and 30-day fills were both counted as one prescription, and OTC drugs were not included, they pointed out.
Humira outsold all other drugs in 2019 in terms of revenue as cytokine inhibitor medications earned their way to three of the first four spots on the pharmaceutical best-seller list, according to a new analysis from the IQVIA Institute for Human Data Science.

Sales of Humira (adalimumab) amounted to $21.4 billion before discounting, Murray Aitken, the institute’s executive director, and associates wrote in their analysis. That’s more than double the total of the anticoagulant Eliquis (apixaban), which brought in $9.9 billion in its last year before generic forms became available.
The next two spots were filled by the tumor necrosis factor inhibitor Enbrel (etanercept) with $8.1 billion in sales and the interleukin 12/23 inhibitor Stelara (ustekinumab) with sales totaling $6.6 billion, followed by the chemotherapy drug Keytruda (pembrolizumab) close behind after racking up $6.5 billion in sales, the researchers reported.
Total nondiscounted spending on all drugs in the U.S. market came to $511 billion in 2019, an increase of 5.7% over the $484 billion spent in 2018, based on data from the July 2020 IQVIA National Sales Perspectives.
These figures are “not adjusted for estimates of off-invoice discounts and rebates,” the authors noted, but they include “prescription and insulin products sold into chain and independent pharmacies, food store pharmacies, mail service pharmacies, long-term care facilities, hospitals, clinics, and other institutional settings.”
Those “discounts and rebates” do exist, however, and they can add up. Drug sales for 2019, “after deducting negotiated rebates, discounts, and other forms of price concessions, such as patient coupons or vouchers that offset out-of-pocket costs,” were $235 billion less than overall nondiscounted spending, the report noted.
Now that we’ve shown you the money, let’s take a quick look at volume. The leading drugs by number of dispensed prescriptions in 2019 were, not surprisingly, quite different. First, with 118 million prescriptions, was atorvastatin, followed by levothyroxine (113 million), lisinopril (96), amlodipine (89), and metoprolol (85), Mr. Aitken and associates reported.
Altogether, over 4.2 billion prescriptions were dispensed last year, with a couple of caveats: 90-day and 30-day fills were both counted as one prescription, and OTC drugs were not included, they pointed out.
RA patients show decreased risk for new-onset type 2 diabetes
Patients with RA were at lower risk for developing incident type 2 diabetes mellitus (T2DM) in comparison with patients with hypertension, psoriatic arthritis (PsA), or osteoarthritis, as well as the general population without RA in a retrospective cohort study of a large, nationwide, commercial health insurance claims database.
This result goes against what the study researchers from the division of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital and Harvard Medical School, both in Boston, initially hypothesized: The “risk of incident T2DM in RA patients would be similar to or less than PsA and [hypertension] patients, but higher, compared to general non-RA and OA patients.”
Prior epidemiologic studies of the relationship between RA and incident diabetes have yielded inconclusive results suggesting a small increase or no increase in risk of T2DM in patients with RA, possibly because of differences in the risk of T2DM in comparison groups used by previous studies to calculate relative risk, first author Yinzhu Jin and colleagues noted in their report published in Arthritis Care & Research.
After mining a nationwide U.S. commercial health insurance claims database, the Optum Clinformatics Data Mart, for claims data from Jan. 1, 2005, to Dec. 31, 2017, the researchers matched a total of 108,568 patients in RA, general population non-RA, hypertension, and OA cohorts based on age, sex, and index date (the date of disease-specific medication dispensing). Overall, 77% of those patients were female and had a mean age of nearly 56 years, whereas 48% of patients with PsA were female and their mean age was nearly 49 years. (PsA patients were not matched because of smaller numbers.)
During a median follow-up period of 1.4-1.8 years across the comparison groups, the crude incidence rate for diabetes per 1,000 person-years in the cohorts was 7.0 for RA, 7.4 for general non-RA, 12.3 for hypertension, 7.8 for OA, and 9.9 for PsA. The hazard ratios and 95% confidence interval for risk of diabetes in patients with RA – after adjustment for more than 40 baseline covariates that included demographics, comorbidities, medication use, and health care utilization – was 0.72 (0.66-0.78) in comparison withh the general non-RA cohort, 0.65 (0.60-0.71) in comparison with the hypertension cohort, 0.75 (0.69-0.81) in comparison with the OA cohort, and 0.76 (0.67-0.86) in comparison with the PsA cohort. These values correspond to RA patients having a 24%-35% lower risk of incident diabetes versus the comparison groups, the researchers noted. They observed results consistent to these when they conducted a sensitivity analysis using a 1-year lag time from the index date before starting follow-up.
The lower risk of T2DM in patients with RA in comparison with patients in the non-RA cohort “may be, in part, due to the effect of biologic DMARD [disease-modifying antirheumatic drug] treatment in RA which likely modifies the risk of DM,” the researchers wrote. “Both the increasing use of biologic DMARDs for RA in the U.S. over the last decade and our cohort entry criteria for the RA cohort (i.e., at least one dispensing of a DMARD) may explain the finding of the lower risk of DM in RA.”
The results found with the other three cohorts did not surprise the researchers. The reduced risk of diabetes among RA patients versus those with OA jibes with “higher rates of obesity and other comorbidities in patients with OA” as well as findings from a recent study that found a higher incidence rate of diabetes in OA, compared with RA. Ms. Jin and colleagues also acknowledged it is well known that “hypertension and PsA are associated with metabolic dysregulation and increase the risk of diabetes.”
The researchers defined patients with RA as having at least twoinpatient or outpatient ICD-9 or ICD-10 diagnosis codes of RA, separated by 7-365 days and having at least one dispensing for DMARDs within 1 year from the first RA diagnosis date, and defined the primary outcome of incident T2DM as at least one inpatient or outpatient diagnosis of T2DM plus at least one dispensing of an antidiabetic drug. They set the general non-RA cohort by selecting patients with any inpatient or outpatient diagnosis codes and a dispensing of any medications, and the hypertension, PsA, and OA comparator groups as having at least two inpatient or outpatient disease-specific ICD-9/ICD-10 codes separated by 7-365 days and at least one dispensing of disease-specific medication within 1 year from the first diagnosis date. They excluded patients with RA, PsA, or psoriasis diagnosis or disease-specific medication dispensing any time prior to or on the index date (the date of disease-specific medication dispensing).
The researchers recognized that the conclusions that can be drawn from the study are limited by the “potential misclassification of cohorts and covariates” because they “mainly used diagnosis codes and pharmacy dispensing records in claims data,” and some “important covariates such as baseline obesity are likely underreported and not adequately captured in claims data.” The level of covariate misclassification also may have been different across the study cohorts on “unmeasured covariates such as body mass index, diet, and physical activity, as well as disease specific measures,” thus introducing residual confounding. They also could not “examine potential difference in the risk of T2DM in untreated or undertreated RA patients” because “RA and all the non-RA comparator cohorts were required to use a disease-specific drug,” they wrote.
“While systemic inflammation in RA is thought to increase the risk of [cardiovascular disease] and cardiovascular risk factors such as DM, our findings suggest having RA itself does not confer an increased risk of DM. Future study should determine whether untreated RA or undertreated RA is associated with a greater risk of developing DM,” the researchers concluded.
The study was supported by a research grant from Bristol-Myers Squibb, which “played no role in the study design, data analysis or interpretation of data or presentation of results,” the researchers said. The company was “given the opportunity to make nonbinding comments on a draft of the manuscript, but the authors retained the right of publication and to determine the final wording.” One author reported receiving research grants from Brigham and Women’s Hospital from Pfizer, AbbVie, Bristol-Myers Squibb, and Roche for unrelated topics.
SOURCE: Jin Y et al. Arthritis Care Res. 2020 Aug 4. doi: 10.1002/acr.24343.
Patients with RA were at lower risk for developing incident type 2 diabetes mellitus (T2DM) in comparison with patients with hypertension, psoriatic arthritis (PsA), or osteoarthritis, as well as the general population without RA in a retrospective cohort study of a large, nationwide, commercial health insurance claims database.
This result goes against what the study researchers from the division of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital and Harvard Medical School, both in Boston, initially hypothesized: The “risk of incident T2DM in RA patients would be similar to or less than PsA and [hypertension] patients, but higher, compared to general non-RA and OA patients.”
Prior epidemiologic studies of the relationship between RA and incident diabetes have yielded inconclusive results suggesting a small increase or no increase in risk of T2DM in patients with RA, possibly because of differences in the risk of T2DM in comparison groups used by previous studies to calculate relative risk, first author Yinzhu Jin and colleagues noted in their report published in Arthritis Care & Research.
After mining a nationwide U.S. commercial health insurance claims database, the Optum Clinformatics Data Mart, for claims data from Jan. 1, 2005, to Dec. 31, 2017, the researchers matched a total of 108,568 patients in RA, general population non-RA, hypertension, and OA cohorts based on age, sex, and index date (the date of disease-specific medication dispensing). Overall, 77% of those patients were female and had a mean age of nearly 56 years, whereas 48% of patients with PsA were female and their mean age was nearly 49 years. (PsA patients were not matched because of smaller numbers.)
During a median follow-up period of 1.4-1.8 years across the comparison groups, the crude incidence rate for diabetes per 1,000 person-years in the cohorts was 7.0 for RA, 7.4 for general non-RA, 12.3 for hypertension, 7.8 for OA, and 9.9 for PsA. The hazard ratios and 95% confidence interval for risk of diabetes in patients with RA – after adjustment for more than 40 baseline covariates that included demographics, comorbidities, medication use, and health care utilization – was 0.72 (0.66-0.78) in comparison withh the general non-RA cohort, 0.65 (0.60-0.71) in comparison with the hypertension cohort, 0.75 (0.69-0.81) in comparison with the OA cohort, and 0.76 (0.67-0.86) in comparison with the PsA cohort. These values correspond to RA patients having a 24%-35% lower risk of incident diabetes versus the comparison groups, the researchers noted. They observed results consistent to these when they conducted a sensitivity analysis using a 1-year lag time from the index date before starting follow-up.
The lower risk of T2DM in patients with RA in comparison with patients in the non-RA cohort “may be, in part, due to the effect of biologic DMARD [disease-modifying antirheumatic drug] treatment in RA which likely modifies the risk of DM,” the researchers wrote. “Both the increasing use of biologic DMARDs for RA in the U.S. over the last decade and our cohort entry criteria for the RA cohort (i.e., at least one dispensing of a DMARD) may explain the finding of the lower risk of DM in RA.”
The results found with the other three cohorts did not surprise the researchers. The reduced risk of diabetes among RA patients versus those with OA jibes with “higher rates of obesity and other comorbidities in patients with OA” as well as findings from a recent study that found a higher incidence rate of diabetes in OA, compared with RA. Ms. Jin and colleagues also acknowledged it is well known that “hypertension and PsA are associated with metabolic dysregulation and increase the risk of diabetes.”
The researchers defined patients with RA as having at least twoinpatient or outpatient ICD-9 or ICD-10 diagnosis codes of RA, separated by 7-365 days and having at least one dispensing for DMARDs within 1 year from the first RA diagnosis date, and defined the primary outcome of incident T2DM as at least one inpatient or outpatient diagnosis of T2DM plus at least one dispensing of an antidiabetic drug. They set the general non-RA cohort by selecting patients with any inpatient or outpatient diagnosis codes and a dispensing of any medications, and the hypertension, PsA, and OA comparator groups as having at least two inpatient or outpatient disease-specific ICD-9/ICD-10 codes separated by 7-365 days and at least one dispensing of disease-specific medication within 1 year from the first diagnosis date. They excluded patients with RA, PsA, or psoriasis diagnosis or disease-specific medication dispensing any time prior to or on the index date (the date of disease-specific medication dispensing).
The researchers recognized that the conclusions that can be drawn from the study are limited by the “potential misclassification of cohorts and covariates” because they “mainly used diagnosis codes and pharmacy dispensing records in claims data,” and some “important covariates such as baseline obesity are likely underreported and not adequately captured in claims data.” The level of covariate misclassification also may have been different across the study cohorts on “unmeasured covariates such as body mass index, diet, and physical activity, as well as disease specific measures,” thus introducing residual confounding. They also could not “examine potential difference in the risk of T2DM in untreated or undertreated RA patients” because “RA and all the non-RA comparator cohorts were required to use a disease-specific drug,” they wrote.
“While systemic inflammation in RA is thought to increase the risk of [cardiovascular disease] and cardiovascular risk factors such as DM, our findings suggest having RA itself does not confer an increased risk of DM. Future study should determine whether untreated RA or undertreated RA is associated with a greater risk of developing DM,” the researchers concluded.
The study was supported by a research grant from Bristol-Myers Squibb, which “played no role in the study design, data analysis or interpretation of data or presentation of results,” the researchers said. The company was “given the opportunity to make nonbinding comments on a draft of the manuscript, but the authors retained the right of publication and to determine the final wording.” One author reported receiving research grants from Brigham and Women’s Hospital from Pfizer, AbbVie, Bristol-Myers Squibb, and Roche for unrelated topics.
SOURCE: Jin Y et al. Arthritis Care Res. 2020 Aug 4. doi: 10.1002/acr.24343.
Patients with RA were at lower risk for developing incident type 2 diabetes mellitus (T2DM) in comparison with patients with hypertension, psoriatic arthritis (PsA), or osteoarthritis, as well as the general population without RA in a retrospective cohort study of a large, nationwide, commercial health insurance claims database.
This result goes against what the study researchers from the division of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital and Harvard Medical School, both in Boston, initially hypothesized: The “risk of incident T2DM in RA patients would be similar to or less than PsA and [hypertension] patients, but higher, compared to general non-RA and OA patients.”
Prior epidemiologic studies of the relationship between RA and incident diabetes have yielded inconclusive results suggesting a small increase or no increase in risk of T2DM in patients with RA, possibly because of differences in the risk of T2DM in comparison groups used by previous studies to calculate relative risk, first author Yinzhu Jin and colleagues noted in their report published in Arthritis Care & Research.
After mining a nationwide U.S. commercial health insurance claims database, the Optum Clinformatics Data Mart, for claims data from Jan. 1, 2005, to Dec. 31, 2017, the researchers matched a total of 108,568 patients in RA, general population non-RA, hypertension, and OA cohorts based on age, sex, and index date (the date of disease-specific medication dispensing). Overall, 77% of those patients were female and had a mean age of nearly 56 years, whereas 48% of patients with PsA were female and their mean age was nearly 49 years. (PsA patients were not matched because of smaller numbers.)
During a median follow-up period of 1.4-1.8 years across the comparison groups, the crude incidence rate for diabetes per 1,000 person-years in the cohorts was 7.0 for RA, 7.4 for general non-RA, 12.3 for hypertension, 7.8 for OA, and 9.9 for PsA. The hazard ratios and 95% confidence interval for risk of diabetes in patients with RA – after adjustment for more than 40 baseline covariates that included demographics, comorbidities, medication use, and health care utilization – was 0.72 (0.66-0.78) in comparison withh the general non-RA cohort, 0.65 (0.60-0.71) in comparison with the hypertension cohort, 0.75 (0.69-0.81) in comparison with the OA cohort, and 0.76 (0.67-0.86) in comparison with the PsA cohort. These values correspond to RA patients having a 24%-35% lower risk of incident diabetes versus the comparison groups, the researchers noted. They observed results consistent to these when they conducted a sensitivity analysis using a 1-year lag time from the index date before starting follow-up.
The lower risk of T2DM in patients with RA in comparison with patients in the non-RA cohort “may be, in part, due to the effect of biologic DMARD [disease-modifying antirheumatic drug] treatment in RA which likely modifies the risk of DM,” the researchers wrote. “Both the increasing use of biologic DMARDs for RA in the U.S. over the last decade and our cohort entry criteria for the RA cohort (i.e., at least one dispensing of a DMARD) may explain the finding of the lower risk of DM in RA.”
The results found with the other three cohorts did not surprise the researchers. The reduced risk of diabetes among RA patients versus those with OA jibes with “higher rates of obesity and other comorbidities in patients with OA” as well as findings from a recent study that found a higher incidence rate of diabetes in OA, compared with RA. Ms. Jin and colleagues also acknowledged it is well known that “hypertension and PsA are associated with metabolic dysregulation and increase the risk of diabetes.”
The researchers defined patients with RA as having at least twoinpatient or outpatient ICD-9 or ICD-10 diagnosis codes of RA, separated by 7-365 days and having at least one dispensing for DMARDs within 1 year from the first RA diagnosis date, and defined the primary outcome of incident T2DM as at least one inpatient or outpatient diagnosis of T2DM plus at least one dispensing of an antidiabetic drug. They set the general non-RA cohort by selecting patients with any inpatient or outpatient diagnosis codes and a dispensing of any medications, and the hypertension, PsA, and OA comparator groups as having at least two inpatient or outpatient disease-specific ICD-9/ICD-10 codes separated by 7-365 days and at least one dispensing of disease-specific medication within 1 year from the first diagnosis date. They excluded patients with RA, PsA, or psoriasis diagnosis or disease-specific medication dispensing any time prior to or on the index date (the date of disease-specific medication dispensing).
The researchers recognized that the conclusions that can be drawn from the study are limited by the “potential misclassification of cohorts and covariates” because they “mainly used diagnosis codes and pharmacy dispensing records in claims data,” and some “important covariates such as baseline obesity are likely underreported and not adequately captured in claims data.” The level of covariate misclassification also may have been different across the study cohorts on “unmeasured covariates such as body mass index, diet, and physical activity, as well as disease specific measures,” thus introducing residual confounding. They also could not “examine potential difference in the risk of T2DM in untreated or undertreated RA patients” because “RA and all the non-RA comparator cohorts were required to use a disease-specific drug,” they wrote.
“While systemic inflammation in RA is thought to increase the risk of [cardiovascular disease] and cardiovascular risk factors such as DM, our findings suggest having RA itself does not confer an increased risk of DM. Future study should determine whether untreated RA or undertreated RA is associated with a greater risk of developing DM,” the researchers concluded.
The study was supported by a research grant from Bristol-Myers Squibb, which “played no role in the study design, data analysis or interpretation of data or presentation of results,” the researchers said. The company was “given the opportunity to make nonbinding comments on a draft of the manuscript, but the authors retained the right of publication and to determine the final wording.” One author reported receiving research grants from Brigham and Women’s Hospital from Pfizer, AbbVie, Bristol-Myers Squibb, and Roche for unrelated topics.
SOURCE: Jin Y et al. Arthritis Care Res. 2020 Aug 4. doi: 10.1002/acr.24343.
FROM ARTHRITIS CARE & RESEARCH
Chloroquine linked to serious psychiatric side effects
Chloroquine may be associated with serious psychiatric side effects, even in patients with no family or personal history of psychiatric disorders, a new review suggests.
In a letter to the editor published online July 28 in The Journal of Clinical Psychiatry, the authors summarize data from several studies published as far back as 1993 and as recently as May 2020.
“In addition to previously reported side effects, chloroquine could also induce psychiatric side effects which are polymorphic and can persist even after stopping the drug,” lead author Florence Gressier, MD, PhD, CESP, Inserm, department of psychiatry, Le Kremlin Bicêtre, France, said in an interview.
“In COVID-19 patients who may still be [undergoing treatment] with chloroquine, close psychiatric assessment and monitoring should be performed,” she said.
Heated controversy
Following findings of a small French study that suggested efficacy in lowering the viral load in patients with COVID-19, President Donald Trump expressed optimism regarding the role of hydroxychloroquine in treating COVID-19, calling it a “game changer”.
Other studies, however, have called into question both the efficacy and the safety of hydroxychloroquine in treating COVID-19. On June 15, the Food and Drug Administration revoked the emergency use authorization it had given in March to chloroquine and hydroxychloroquine for the treatment of COVID-19.
Nevertheless, hydroxychloroquine continues to be prescribed for COVID-19. For example, an article that appeared in Click2Houston on June 15 quoted the chief medical officer of Houston’s United Memorial Center as saying he plans to continue prescribing hydroxychloroquine for patients with COVID-19 until he finds a better alternative.
As discussed in a Medscape expert commentary, a group of physicians who held a “white coat summit” in front of the U.S. Supreme Court building promoted the use of hydroxychloroquine for the treatment of COVID-19. The video of their summit was retweeted by President Trump and garnered millions of views before it was taken down by Twitter, Facebook, and YouTube.
Sudden onset
For the new review, “we wanted to alert the public and practitioners on the potentially psychiatric risks induced by chloroquine, as it could be taken as self-medication or potentially still prescribed,” Dr. Gressier said.
“We think the format of the letter to the editor allows information to be provided in a concise and clear manner,” she added.
According to the FDA’s Adverse Event Reporting System database, 12% of reported adverse events (520 of 4,336) following the use of chloroquine that occurred between the fourth quarter of 2012 and the fourth quarter of 2019 were neuropsychiatric. These events included amnesia, delirium, hallucinations, depression, and loss of consciousness, the authors write.
The researchers acknowledged that the incidence of psychiatric adverse effects associated with the use of chloroquine is “unclear in the absence of high-quality, randomized placebo-controlled trials of its safety.” Nevertheless, they pointed out that there have been reports of insomnia and depression when the drug was used as prophylaxis against malaria .
Moreover, some case series or case reports describe symptoms such as depression, anxiety, agitation, violent outburst, suicidal ideation, and psychosis in patients who have been treated with chloroquine for malaria, lupus erythematosus, and rheumatoid arthritis .
“In contrast to many other psychoses, chloroquine psychosis may be more affective and include prominent visual hallucinations, symptoms of derealization, and disorders of thought, with preserved insight,” the authors wrote.
They noted that the frequency of symptoms does not appear to be connected to the cumulative dose or the duration of treatment, and the onset of psychosis or other adverse effects is usually “sudden.”
In addition, they warn that the drug’s psychiatric effects may go unnoticed, especially because COVID-19 itself has been associated with neuropsychiatric symptoms, making it hard to distinguish between symptoms caused by the illness and those caused by the drug.
Although the psychiatric symptoms typically occur early after treatment initiation, some “subtle” symptoms might persist after stopping the drug, possibly owing to its “extremely long” half-life, the authors stated.
Dr. Gressier noted that practicing clinicians should look up reports about self-medication with chloroquine “and warn their patients about the risk induced by chloroquine.”
Safe but ‘not benign’
Nilanjana Bose, MD, MBA, a rheumatologist at the Rheumatology Center of Houston, said she uses hydroxychloroquine “all the time” in clinical practice to treat patients with rheumatic conditions.
“I cannot comment on whether it [hydroxychloroquine or chloroquine] is a potential prophylactic or treatment for COVID-19, but I can say that, from a safety point of view, as a rheumatologist who uses hydroxychloroquine at a dose of 400 mg/day, I do not think we need to worry about serious [psychiatric] side effects,” Dr. Bose said in an interview.
Because clinicians are trying all types of possible treatments for COVID-19, “if this medication has possible efficacy, it is a great medicine from a rheumatologic perspective and is safe,” she added.
Nevertheless, the drug is “not benign, and regular side effects will be there, and of course, higher doses will cause more side effects,” said Dr. Bose, who was not involved in authoring the letter.
She counsels patients about potential psychiatric side effects of hydroxychloroquine because some of her patients have complained about irritability, worsening anxiety and depression, and difficulty sleeping.
Be wary
James “Jimmy” Potash, MD, MPH, Henry Phipps Professor of Psychiatry and Behavioral Sciences, Johns Hopkins Medicine, Baltimore, said in an interview that the “take-home message of this letter is that serious psychiatric effects, psychotic illness in particular,” can occur in individuals who take chloroquine and hydroxychloroquine.
In addition, “these are potentially very concerning side effects that psychiatrists should be aware of,” noted Dr. Potash, department director and psychiatrist-in-chief at Johns Hopkins.
He said that one of his patients who had been “completely psychiatrically healthy” took chloroquine prophylactically prior to traveling overseas. After she began taking the drug, she had an episode of mania that resolved once she discontinued the medication and received treatment for the mania.
“If you add potential psychiatric side effects to the other side effects that can result from these medications, that adds up to a pretty important reason to be wary of taking them, particularly for the indication of COVID-19, where the level of evidence that it helps in any way is still quite weak,” Dr. Potash said.
In an interview, Remington Nevin, MD, MPH, DrPH, executive director at the Quinism Foundation, White River Junction, Vt., a nonprofit organization that supports and promotes education and research on disorders caused by poisoning by quinoline drugs; and faculty associate in the department of mental health at Johns Hopkins Bloomberg School of Public Health, said that the authors of the letter “are to be commended for their efforts in raising awareness of the potentially lasting and disabling psychiatric effects of chloroquine and hydroxychloroquine, which, as with similar effects from other synthetic quinoline antimalarials, have occasionally been overlooked or misattributed to other conditions.”
He added: “I have proposed that the chronic neuropsychiatric effects of this class of drug are best considered not as side effects but as signs and symptoms of a disorder known as chronic quinoline encephalopathy caused by poisoning of the central nervous system.”
Dr. Gressier and the other letter authors, Dr. Bose, and Dr. Potash have reported no relevant financial relationships. Dr. Nevin has been retained as a consultant and expert witness in legal cases involving claims of adverse effects from quinoline antimalarial drugs.
A version of this article originally appeared on Medscape.com.
Chloroquine may be associated with serious psychiatric side effects, even in patients with no family or personal history of psychiatric disorders, a new review suggests.
In a letter to the editor published online July 28 in The Journal of Clinical Psychiatry, the authors summarize data from several studies published as far back as 1993 and as recently as May 2020.
“In addition to previously reported side effects, chloroquine could also induce psychiatric side effects which are polymorphic and can persist even after stopping the drug,” lead author Florence Gressier, MD, PhD, CESP, Inserm, department of psychiatry, Le Kremlin Bicêtre, France, said in an interview.
“In COVID-19 patients who may still be [undergoing treatment] with chloroquine, close psychiatric assessment and monitoring should be performed,” she said.
Heated controversy
Following findings of a small French study that suggested efficacy in lowering the viral load in patients with COVID-19, President Donald Trump expressed optimism regarding the role of hydroxychloroquine in treating COVID-19, calling it a “game changer”.
Other studies, however, have called into question both the efficacy and the safety of hydroxychloroquine in treating COVID-19. On June 15, the Food and Drug Administration revoked the emergency use authorization it had given in March to chloroquine and hydroxychloroquine for the treatment of COVID-19.
Nevertheless, hydroxychloroquine continues to be prescribed for COVID-19. For example, an article that appeared in Click2Houston on June 15 quoted the chief medical officer of Houston’s United Memorial Center as saying he plans to continue prescribing hydroxychloroquine for patients with COVID-19 until he finds a better alternative.
As discussed in a Medscape expert commentary, a group of physicians who held a “white coat summit” in front of the U.S. Supreme Court building promoted the use of hydroxychloroquine for the treatment of COVID-19. The video of their summit was retweeted by President Trump and garnered millions of views before it was taken down by Twitter, Facebook, and YouTube.
Sudden onset
For the new review, “we wanted to alert the public and practitioners on the potentially psychiatric risks induced by chloroquine, as it could be taken as self-medication or potentially still prescribed,” Dr. Gressier said.
“We think the format of the letter to the editor allows information to be provided in a concise and clear manner,” she added.
According to the FDA’s Adverse Event Reporting System database, 12% of reported adverse events (520 of 4,336) following the use of chloroquine that occurred between the fourth quarter of 2012 and the fourth quarter of 2019 were neuropsychiatric. These events included amnesia, delirium, hallucinations, depression, and loss of consciousness, the authors write.
The researchers acknowledged that the incidence of psychiatric adverse effects associated with the use of chloroquine is “unclear in the absence of high-quality, randomized placebo-controlled trials of its safety.” Nevertheless, they pointed out that there have been reports of insomnia and depression when the drug was used as prophylaxis against malaria .
Moreover, some case series or case reports describe symptoms such as depression, anxiety, agitation, violent outburst, suicidal ideation, and psychosis in patients who have been treated with chloroquine for malaria, lupus erythematosus, and rheumatoid arthritis .
“In contrast to many other psychoses, chloroquine psychosis may be more affective and include prominent visual hallucinations, symptoms of derealization, and disorders of thought, with preserved insight,” the authors wrote.
They noted that the frequency of symptoms does not appear to be connected to the cumulative dose or the duration of treatment, and the onset of psychosis or other adverse effects is usually “sudden.”
In addition, they warn that the drug’s psychiatric effects may go unnoticed, especially because COVID-19 itself has been associated with neuropsychiatric symptoms, making it hard to distinguish between symptoms caused by the illness and those caused by the drug.
Although the psychiatric symptoms typically occur early after treatment initiation, some “subtle” symptoms might persist after stopping the drug, possibly owing to its “extremely long” half-life, the authors stated.
Dr. Gressier noted that practicing clinicians should look up reports about self-medication with chloroquine “and warn their patients about the risk induced by chloroquine.”
Safe but ‘not benign’
Nilanjana Bose, MD, MBA, a rheumatologist at the Rheumatology Center of Houston, said she uses hydroxychloroquine “all the time” in clinical practice to treat patients with rheumatic conditions.
“I cannot comment on whether it [hydroxychloroquine or chloroquine] is a potential prophylactic or treatment for COVID-19, but I can say that, from a safety point of view, as a rheumatologist who uses hydroxychloroquine at a dose of 400 mg/day, I do not think we need to worry about serious [psychiatric] side effects,” Dr. Bose said in an interview.
Because clinicians are trying all types of possible treatments for COVID-19, “if this medication has possible efficacy, it is a great medicine from a rheumatologic perspective and is safe,” she added.
Nevertheless, the drug is “not benign, and regular side effects will be there, and of course, higher doses will cause more side effects,” said Dr. Bose, who was not involved in authoring the letter.
She counsels patients about potential psychiatric side effects of hydroxychloroquine because some of her patients have complained about irritability, worsening anxiety and depression, and difficulty sleeping.
Be wary
James “Jimmy” Potash, MD, MPH, Henry Phipps Professor of Psychiatry and Behavioral Sciences, Johns Hopkins Medicine, Baltimore, said in an interview that the “take-home message of this letter is that serious psychiatric effects, psychotic illness in particular,” can occur in individuals who take chloroquine and hydroxychloroquine.
In addition, “these are potentially very concerning side effects that psychiatrists should be aware of,” noted Dr. Potash, department director and psychiatrist-in-chief at Johns Hopkins.
He said that one of his patients who had been “completely psychiatrically healthy” took chloroquine prophylactically prior to traveling overseas. After she began taking the drug, she had an episode of mania that resolved once she discontinued the medication and received treatment for the mania.
“If you add potential psychiatric side effects to the other side effects that can result from these medications, that adds up to a pretty important reason to be wary of taking them, particularly for the indication of COVID-19, where the level of evidence that it helps in any way is still quite weak,” Dr. Potash said.
In an interview, Remington Nevin, MD, MPH, DrPH, executive director at the Quinism Foundation, White River Junction, Vt., a nonprofit organization that supports and promotes education and research on disorders caused by poisoning by quinoline drugs; and faculty associate in the department of mental health at Johns Hopkins Bloomberg School of Public Health, said that the authors of the letter “are to be commended for their efforts in raising awareness of the potentially lasting and disabling psychiatric effects of chloroquine and hydroxychloroquine, which, as with similar effects from other synthetic quinoline antimalarials, have occasionally been overlooked or misattributed to other conditions.”
He added: “I have proposed that the chronic neuropsychiatric effects of this class of drug are best considered not as side effects but as signs and symptoms of a disorder known as chronic quinoline encephalopathy caused by poisoning of the central nervous system.”
Dr. Gressier and the other letter authors, Dr. Bose, and Dr. Potash have reported no relevant financial relationships. Dr. Nevin has been retained as a consultant and expert witness in legal cases involving claims of adverse effects from quinoline antimalarial drugs.
A version of this article originally appeared on Medscape.com.
Chloroquine may be associated with serious psychiatric side effects, even in patients with no family or personal history of psychiatric disorders, a new review suggests.
In a letter to the editor published online July 28 in The Journal of Clinical Psychiatry, the authors summarize data from several studies published as far back as 1993 and as recently as May 2020.
“In addition to previously reported side effects, chloroquine could also induce psychiatric side effects which are polymorphic and can persist even after stopping the drug,” lead author Florence Gressier, MD, PhD, CESP, Inserm, department of psychiatry, Le Kremlin Bicêtre, France, said in an interview.
“In COVID-19 patients who may still be [undergoing treatment] with chloroquine, close psychiatric assessment and monitoring should be performed,” she said.
Heated controversy
Following findings of a small French study that suggested efficacy in lowering the viral load in patients with COVID-19, President Donald Trump expressed optimism regarding the role of hydroxychloroquine in treating COVID-19, calling it a “game changer”.
Other studies, however, have called into question both the efficacy and the safety of hydroxychloroquine in treating COVID-19. On June 15, the Food and Drug Administration revoked the emergency use authorization it had given in March to chloroquine and hydroxychloroquine for the treatment of COVID-19.
Nevertheless, hydroxychloroquine continues to be prescribed for COVID-19. For example, an article that appeared in Click2Houston on June 15 quoted the chief medical officer of Houston’s United Memorial Center as saying he plans to continue prescribing hydroxychloroquine for patients with COVID-19 until he finds a better alternative.
As discussed in a Medscape expert commentary, a group of physicians who held a “white coat summit” in front of the U.S. Supreme Court building promoted the use of hydroxychloroquine for the treatment of COVID-19. The video of their summit was retweeted by President Trump and garnered millions of views before it was taken down by Twitter, Facebook, and YouTube.
Sudden onset
For the new review, “we wanted to alert the public and practitioners on the potentially psychiatric risks induced by chloroquine, as it could be taken as self-medication or potentially still prescribed,” Dr. Gressier said.
“We think the format of the letter to the editor allows information to be provided in a concise and clear manner,” she added.
According to the FDA’s Adverse Event Reporting System database, 12% of reported adverse events (520 of 4,336) following the use of chloroquine that occurred between the fourth quarter of 2012 and the fourth quarter of 2019 were neuropsychiatric. These events included amnesia, delirium, hallucinations, depression, and loss of consciousness, the authors write.
The researchers acknowledged that the incidence of psychiatric adverse effects associated with the use of chloroquine is “unclear in the absence of high-quality, randomized placebo-controlled trials of its safety.” Nevertheless, they pointed out that there have been reports of insomnia and depression when the drug was used as prophylaxis against malaria .
Moreover, some case series or case reports describe symptoms such as depression, anxiety, agitation, violent outburst, suicidal ideation, and psychosis in patients who have been treated with chloroquine for malaria, lupus erythematosus, and rheumatoid arthritis .
“In contrast to many other psychoses, chloroquine psychosis may be more affective and include prominent visual hallucinations, symptoms of derealization, and disorders of thought, with preserved insight,” the authors wrote.
They noted that the frequency of symptoms does not appear to be connected to the cumulative dose or the duration of treatment, and the onset of psychosis or other adverse effects is usually “sudden.”
In addition, they warn that the drug’s psychiatric effects may go unnoticed, especially because COVID-19 itself has been associated with neuropsychiatric symptoms, making it hard to distinguish between symptoms caused by the illness and those caused by the drug.
Although the psychiatric symptoms typically occur early after treatment initiation, some “subtle” symptoms might persist after stopping the drug, possibly owing to its “extremely long” half-life, the authors stated.
Dr. Gressier noted that practicing clinicians should look up reports about self-medication with chloroquine “and warn their patients about the risk induced by chloroquine.”
Safe but ‘not benign’
Nilanjana Bose, MD, MBA, a rheumatologist at the Rheumatology Center of Houston, said she uses hydroxychloroquine “all the time” in clinical practice to treat patients with rheumatic conditions.
“I cannot comment on whether it [hydroxychloroquine or chloroquine] is a potential prophylactic or treatment for COVID-19, but I can say that, from a safety point of view, as a rheumatologist who uses hydroxychloroquine at a dose of 400 mg/day, I do not think we need to worry about serious [psychiatric] side effects,” Dr. Bose said in an interview.
Because clinicians are trying all types of possible treatments for COVID-19, “if this medication has possible efficacy, it is a great medicine from a rheumatologic perspective and is safe,” she added.
Nevertheless, the drug is “not benign, and regular side effects will be there, and of course, higher doses will cause more side effects,” said Dr. Bose, who was not involved in authoring the letter.
She counsels patients about potential psychiatric side effects of hydroxychloroquine because some of her patients have complained about irritability, worsening anxiety and depression, and difficulty sleeping.
Be wary
James “Jimmy” Potash, MD, MPH, Henry Phipps Professor of Psychiatry and Behavioral Sciences, Johns Hopkins Medicine, Baltimore, said in an interview that the “take-home message of this letter is that serious psychiatric effects, psychotic illness in particular,” can occur in individuals who take chloroquine and hydroxychloroquine.
In addition, “these are potentially very concerning side effects that psychiatrists should be aware of,” noted Dr. Potash, department director and psychiatrist-in-chief at Johns Hopkins.
He said that one of his patients who had been “completely psychiatrically healthy” took chloroquine prophylactically prior to traveling overseas. After she began taking the drug, she had an episode of mania that resolved once she discontinued the medication and received treatment for the mania.
“If you add potential psychiatric side effects to the other side effects that can result from these medications, that adds up to a pretty important reason to be wary of taking them, particularly for the indication of COVID-19, where the level of evidence that it helps in any way is still quite weak,” Dr. Potash said.
In an interview, Remington Nevin, MD, MPH, DrPH, executive director at the Quinism Foundation, White River Junction, Vt., a nonprofit organization that supports and promotes education and research on disorders caused by poisoning by quinoline drugs; and faculty associate in the department of mental health at Johns Hopkins Bloomberg School of Public Health, said that the authors of the letter “are to be commended for their efforts in raising awareness of the potentially lasting and disabling psychiatric effects of chloroquine and hydroxychloroquine, which, as with similar effects from other synthetic quinoline antimalarials, have occasionally been overlooked or misattributed to other conditions.”
He added: “I have proposed that the chronic neuropsychiatric effects of this class of drug are best considered not as side effects but as signs and symptoms of a disorder known as chronic quinoline encephalopathy caused by poisoning of the central nervous system.”
Dr. Gressier and the other letter authors, Dr. Bose, and Dr. Potash have reported no relevant financial relationships. Dr. Nevin has been retained as a consultant and expert witness in legal cases involving claims of adverse effects from quinoline antimalarial drugs.
A version of this article originally appeared on Medscape.com.
Doctors hesitated to embrace biosimilar infliximab in first 2 years
Physicians have been slow to embrace biosimilar versions of infliximab, but are more likely to prescribe it to new patients, based on data from a review of nearly 50,000 infliximab claims through Medicare in the first 2 years that biosimilars were available in the United States.
“Although biosimilar versions are as safe and effective as the biologic, patients and physicians may be more reluctant to switch from a working biologic regimen in a chronic setting than an acute one,” wrote Alice J. Chen, PhD, of the University of Southern California, Los Angeles, and colleagues.
In a research letter published in JAMA Internal Medicine, the investigators examined prescribing patterns of physicians switching between the originator infliximab (Remicade) and two of its biosimilars (Inflectra and Renflexis).
They reviewed infliximab use and reimbursement in the 100% Medicare Part B quarterly claims database from Jan. 1, 2017, to Dec. 31, 2018. The study population included Medicare patients classified as new if they had no infliximab claims in the prior 6 months; those with claims were considered returning patients.
In a comparison of claims reflecting 49,771 patients and 4,289 physicians in 2018, a total of 1,418 new patients (17.4%) and 4,495 (10.8%) returning patients used a biosimilar. “Of returning patients, half used the biosimilar version exclusively, whereas the other half switched between biologic and biosimilar versions,” the researchers noted.
Of the 4,289 physicians who prescribed infliximab, 3,124 prescribed no biosimilars, 1,015 prescribed both biologics and biosimilars, and 150 prescribed biosimilars only. Of the physicians who prescribed both, approximately 61% switched some patients from the biologic to the biosimilar; “the remainder kept individual patients on only 1 version of the drug but treated patients with both versions,” the researchers wrote.
The adoption of biosimilars may be slower for chronic vs. acute conditions, the researchers noted. “Prescribers may hesitate to switch clinically stable chronic patients from biologic regimens if they are unfamiliar with the biosimilar or face financial disincentives from prescribing it.”
The study findings were limited by several factors including the use of only 2 years of data and a focus only on Medicare Part B. Switching medications may have been influenced by factors such as lower copays for patients and rebates or discounts for physicians; however, “further research is needed to better understand biosimilar pricing dynamics and the barriers to adopting biosimilars for chronic conditions,” they concluded.
The study was supported by the Leonard D. Schaeffer Center for Health Policy & Economics at the University of Southern California, Los Angeles, and the National Institute on Aging. Lead author Dr. Chen also disclosed receiving personal fees from Amgen outside of the current study.
SOURCE: Chen AJ et al. JAMA Intern Med. 2020 July 20. doi: 10.1001/jamainternmed.2020.3188.
Physicians have been slow to embrace biosimilar versions of infliximab, but are more likely to prescribe it to new patients, based on data from a review of nearly 50,000 infliximab claims through Medicare in the first 2 years that biosimilars were available in the United States.
“Although biosimilar versions are as safe and effective as the biologic, patients and physicians may be more reluctant to switch from a working biologic regimen in a chronic setting than an acute one,” wrote Alice J. Chen, PhD, of the University of Southern California, Los Angeles, and colleagues.
In a research letter published in JAMA Internal Medicine, the investigators examined prescribing patterns of physicians switching between the originator infliximab (Remicade) and two of its biosimilars (Inflectra and Renflexis).
They reviewed infliximab use and reimbursement in the 100% Medicare Part B quarterly claims database from Jan. 1, 2017, to Dec. 31, 2018. The study population included Medicare patients classified as new if they had no infliximab claims in the prior 6 months; those with claims were considered returning patients.
In a comparison of claims reflecting 49,771 patients and 4,289 physicians in 2018, a total of 1,418 new patients (17.4%) and 4,495 (10.8%) returning patients used a biosimilar. “Of returning patients, half used the biosimilar version exclusively, whereas the other half switched between biologic and biosimilar versions,” the researchers noted.
Of the 4,289 physicians who prescribed infliximab, 3,124 prescribed no biosimilars, 1,015 prescribed both biologics and biosimilars, and 150 prescribed biosimilars only. Of the physicians who prescribed both, approximately 61% switched some patients from the biologic to the biosimilar; “the remainder kept individual patients on only 1 version of the drug but treated patients with both versions,” the researchers wrote.
The adoption of biosimilars may be slower for chronic vs. acute conditions, the researchers noted. “Prescribers may hesitate to switch clinically stable chronic patients from biologic regimens if they are unfamiliar with the biosimilar or face financial disincentives from prescribing it.”
The study findings were limited by several factors including the use of only 2 years of data and a focus only on Medicare Part B. Switching medications may have been influenced by factors such as lower copays for patients and rebates or discounts for physicians; however, “further research is needed to better understand biosimilar pricing dynamics and the barriers to adopting biosimilars for chronic conditions,” they concluded.
The study was supported by the Leonard D. Schaeffer Center for Health Policy & Economics at the University of Southern California, Los Angeles, and the National Institute on Aging. Lead author Dr. Chen also disclosed receiving personal fees from Amgen outside of the current study.
SOURCE: Chen AJ et al. JAMA Intern Med. 2020 July 20. doi: 10.1001/jamainternmed.2020.3188.
Physicians have been slow to embrace biosimilar versions of infliximab, but are more likely to prescribe it to new patients, based on data from a review of nearly 50,000 infliximab claims through Medicare in the first 2 years that biosimilars were available in the United States.
“Although biosimilar versions are as safe and effective as the biologic, patients and physicians may be more reluctant to switch from a working biologic regimen in a chronic setting than an acute one,” wrote Alice J. Chen, PhD, of the University of Southern California, Los Angeles, and colleagues.
In a research letter published in JAMA Internal Medicine, the investigators examined prescribing patterns of physicians switching between the originator infliximab (Remicade) and two of its biosimilars (Inflectra and Renflexis).
They reviewed infliximab use and reimbursement in the 100% Medicare Part B quarterly claims database from Jan. 1, 2017, to Dec. 31, 2018. The study population included Medicare patients classified as new if they had no infliximab claims in the prior 6 months; those with claims were considered returning patients.
In a comparison of claims reflecting 49,771 patients and 4,289 physicians in 2018, a total of 1,418 new patients (17.4%) and 4,495 (10.8%) returning patients used a biosimilar. “Of returning patients, half used the biosimilar version exclusively, whereas the other half switched between biologic and biosimilar versions,” the researchers noted.
Of the 4,289 physicians who prescribed infliximab, 3,124 prescribed no biosimilars, 1,015 prescribed both biologics and biosimilars, and 150 prescribed biosimilars only. Of the physicians who prescribed both, approximately 61% switched some patients from the biologic to the biosimilar; “the remainder kept individual patients on only 1 version of the drug but treated patients with both versions,” the researchers wrote.
The adoption of biosimilars may be slower for chronic vs. acute conditions, the researchers noted. “Prescribers may hesitate to switch clinically stable chronic patients from biologic regimens if they are unfamiliar with the biosimilar or face financial disincentives from prescribing it.”
The study findings were limited by several factors including the use of only 2 years of data and a focus only on Medicare Part B. Switching medications may have been influenced by factors such as lower copays for patients and rebates or discounts for physicians; however, “further research is needed to better understand biosimilar pricing dynamics and the barriers to adopting biosimilars for chronic conditions,” they concluded.
The study was supported by the Leonard D. Schaeffer Center for Health Policy & Economics at the University of Southern California, Los Angeles, and the National Institute on Aging. Lead author Dr. Chen also disclosed receiving personal fees from Amgen outside of the current study.
SOURCE: Chen AJ et al. JAMA Intern Med. 2020 July 20. doi: 10.1001/jamainternmed.2020.3188.
FROM JAMA INTERNAL MEDICINE
Key clinical point: A total of 17% of patients new to infliximab received a biosimilar in 2018, compared with 11% of returning patients.
Major finding: Biosimilar infliximab accounted for 10% of the market share 2 years after the product was introduced.
Study details: The data come from a review of infliximab claims across 49,771 patients and 4,289 physicians who prescribed infliximab in 2018.
Disclosures: The study was supported by the Leonard D. Schaeffer Center for Health Policy & Economics at the University of Southern California, Los Angeles, and the National Institute on Aging. Lead author Dr. Chen also disclosed receiving personal fees from Amgen outside of the current study.
Source: Chen AJ et al. JAMA Intern Med. 2020 July 20. doi: 10.1001/jamainternmed.2020.3188.
Ready for PRIME time? Newly ID’d cells predict RA flares
A newly identified circulating cell type may be a reliable marker for impending RA flares. The discovery and description of the cells, which bear a “striking” similarity to synovial fibroblasts, provide important clues to the origins of RA and progressive joint inflammation, investigators say.
By studying longitudinally collected blood samples from four patients with RA over 4 years, Dana E. Orange, MD and colleagues at Rockefeller University, New York, identified a pattern of B-cell activation and expansion of circulating cells that are negative for CD45 and CD31 expression, and positive for PDPN, dubbed preinflammatory mesenchymal or “PRIME” cells.
Expansion of PRIME cells in circulation increased dramatically in the weeks leading up to a flare and decreased during a flare, suggesting the possibility of a serum assay for predicting flares and allowing for early intervention to ameliorate or prevent disabling consequences, the investigators wrote in a study published in the New England Journal of Medicine.
“Our hope is that this will be a diagnostic in the future, but we need to study it in more patients to see how it will perform,” Dr. Orange said in an interview, adding that the cells, if shown to be pathogenic, could also be targets for new therapeutic strategies.
RNA sequencing
Dr. Orange and colleagues discovered the PRIME cells through a novel clinical and technical protocol involving home collection of blood by patients and longitudinal RNA sequencing to study gene expression profiles during times of both disease quiescence and flares, and noticed a distinct pattern of PRIME cell expansion, depletion, and gene expression.
“Looking at their gene expression profiles, they overlapped with fibroblasts that reside in inflamed rheumatoid arthritis synovium, and in an animal model those types of fibroblasts were important for allowing entry of inflammatory infiltrates around the joint,” she said.
PRIME cells may be a precursor of synovial fibroblasts, which have been implicated by some researchers in the spread of RA between joints, Dr. Orange added.
Patients do homework
The investigators began by enrolling four patients, followed for 1-4 years, who met 2010 American College of Rheumatology–European League Against Rheumatism criteria and who were seropositive for anti–cyclic citrullinated peptide antibodies.
They assessed disease activity from patient homes weekly or during escalation of flares up to four times daily, with the Routine Assessment of Patient Index Data 3 (RAPID3) questionnaire, as well as monthly clinic visits. At clinic visits during flares, disease activity was assessed using both the RAPID3 and 28-joint Disease Activity Score.
The patients performed fingerstick blood collection and mailed the samples overnight each week to Rockefeller University, where RNA was extracted and sequenced. The investigators identified gene transcripts that were differentially expressed in blood prior to flares, and compared them with data profiles derived from synovial single-cell RNA sequencing.
To validate the findings, the researchers used flow cytometry and sorted blood-cell RNA sequencing of samples from an additional 19 patients with RA.
They found that a total of 2,613 genes were differentially expressed during a flare, compared with baseline, and that expression of 1,437 of these genes was increased during a flare, with the remaining 1,176 decreased during flares.
Before the storm
Focusing on two flare-antecedent clusters of genes, they identified one cluster of transcripts that increased 2 weeks before a flare, enriched with genes coding for developmental pathways for naive B cells (that is, not yet exposed to antigens) and leukocytes.
The second cluster included gene transcripts that increased during the week before a flare, then decreased over the duration of the flare. Genes in this cluster were enriched for pathways that were unexpected in typical blood specimens, including genes involved in cartilage morphogenesis, endochondral bone growth, and extracellular matrix organization. The gene activity suggested the presence of a mesenchymal cell, they wrote.
The RNA expression profiles of these newly identified PRIME cells were very similar to those of synovial fibroblasts, and the investigators speculated that PRIME cells may be synovial fibroblast precursors.
They proposed a model of RA exacerbation in which PRIME cells become activated by B cells in the weeks immediately preceding a flare, and then migrate out of blood into the synovium.
The investigators are currently investigating “how reproducible this signature is in different flares in patients on different types of background therapy, and then we’re very interested in looking at the upstream triggers of the B cell and the PRIME cell,” Dr. Orange said.
“One of the reasons this is very exciting is that there are these signatures that can be found when patients are clearly asymptomatic but about to flare, and if we can intervene at that time, then the patients won’t have to live through a flare, they won’t have to have that experience,” she said.
Pros and cons
In an editorial accompanying the study, Ellen M. Gravallese, MD, from Brigham and Women’s Hospital in Boston and William H. Robinson, MD, PhD, from Stanford (Calif.) University and the Veterans Affairs Palo Alto (Calif.) Health Care System wrote that the study demonstrates an important method for identifying genetic contributions to many different types of disease.
“Orange and colleagues show that intensively collected longitudinal data from a small sample of patients can be used to identify dysregulated transcriptional signatures that are not recognized by classical cross-sectional studies. This study illustrates the exciting potential of longitudinal genomics to identify key antecedents of disease flares in an approach that may be applicable to the investigation of pathogenic and protective immune responses in a wide range of human diseases,” they wrote.
Rheumatology researcher Christopher D. Buckley, MBBS, DPhil, from the University of Birmingham (England), who was not involved in the study, said that the use of blood samples is both a strength and a weakness of the study.
“Blood is much easier to get than synovial tissue, but synovial tissue is important. If I’m trying to look at the blood and trying to make an inference about what’s going on in the synovium, if I don’t look at the synovium I don’t know what the link between the blood and synovium is,” he said in an interview.
On the plus side, “the big advantage about looking at blood is that you do multiple time points, which is really cool,” he said.
Dr. Buckley is a coauthor of a recent paper in Nature Medicine – published after the study by Dr. Orange and colleagues was accepted by the New England Journal of Medicine – showing that a population of macrophages in synovium was highly predictive of remission in patients with RA, and that therapeutic modulation of these macrophages has the potential as a treatment strategy for RA.
“We are very keen to understand the cellular basis of disease. We’re very good at understanding genes, but genes have to work in cells, and cells make organs, so the cells are critical,” he said.
The paper adds fuel to a controversy that has been raging among rheumatology researchers for more than a decade: the “flying fibroblast” hypothesis, which suggests that fibroblasts can migrate from one joint to another, hence spreading the disease in a manner akin to cancer metastases.
“It’s been quite controversial whether these cells like fibroblasts can exist in the blood, or whether they’re found in sufficient number in the blood,” Dr. Buckley said. “The fact that they have identified these PRIME cells is fascinating, because that’s going to cause us to go back and reinvestigate the whole flying fibroblast story.”
His colleague John Isaacs, MBBS, PhD, from Newcastle (England) University, is principal investigator for the BIO-FLARE study, in which participants with RA stop taking their disease-modifying antirheumatic drugs under close supervision of researchers. The investigators then study the patients looking for flare signals as well as the biology of flares themselves.
“As it happens, our protocols would not pick up this particular cell, because we have not been focusing on the stroma, at least not in peripheral blood. We’ve all been looking at synovium as part of BIO-FLARE,” he said in an interview. “We will be looking for this cell now that we have seen this research, and certainly we would want to replicate.”
He agreed with Dr. Buckley’s observation that the PRIME cell data may revive the flying fibroblast hypothesis. “This is a great paper in a top clinical journal. What isn’t there is mechanism. That’s the thing we’re all going to want to understand now: Where do the cells come from, how do they actually trigger flares, and how do they go down as flare starts?”
Both Dr. Buckley and Dr. Isaacs agreed that the study findings point to important new avenues of research, but also noted that the study was small, involving a total of only 23 patients, and that replication of the findings and elucidation of the mechanism of PRIME cell generation and disposition will be required.
The study was supported by grants from the National Institutes of Health, Simons Foundation, Robertson Foundation, Rheumatology Research Foundation, Bernard and Irene Schwartz Foundation, the Iris and Junming Le Foundation, and Rockefeller University. Dr. Orange disclosed a provisional patent for the discovery of the PRIME cells. Dr. Buckley and Dr. Isaacs reported no relevant conflicts of interest.
SOURCE: Orange DE et al. N Engl J Med. 2020 Jul 15. doi: 10.1056/NEJMoa2004114.
A newly identified circulating cell type may be a reliable marker for impending RA flares. The discovery and description of the cells, which bear a “striking” similarity to synovial fibroblasts, provide important clues to the origins of RA and progressive joint inflammation, investigators say.
By studying longitudinally collected blood samples from four patients with RA over 4 years, Dana E. Orange, MD and colleagues at Rockefeller University, New York, identified a pattern of B-cell activation and expansion of circulating cells that are negative for CD45 and CD31 expression, and positive for PDPN, dubbed preinflammatory mesenchymal or “PRIME” cells.
Expansion of PRIME cells in circulation increased dramatically in the weeks leading up to a flare and decreased during a flare, suggesting the possibility of a serum assay for predicting flares and allowing for early intervention to ameliorate or prevent disabling consequences, the investigators wrote in a study published in the New England Journal of Medicine.
“Our hope is that this will be a diagnostic in the future, but we need to study it in more patients to see how it will perform,” Dr. Orange said in an interview, adding that the cells, if shown to be pathogenic, could also be targets for new therapeutic strategies.
RNA sequencing
Dr. Orange and colleagues discovered the PRIME cells through a novel clinical and technical protocol involving home collection of blood by patients and longitudinal RNA sequencing to study gene expression profiles during times of both disease quiescence and flares, and noticed a distinct pattern of PRIME cell expansion, depletion, and gene expression.
“Looking at their gene expression profiles, they overlapped with fibroblasts that reside in inflamed rheumatoid arthritis synovium, and in an animal model those types of fibroblasts were important for allowing entry of inflammatory infiltrates around the joint,” she said.
PRIME cells may be a precursor of synovial fibroblasts, which have been implicated by some researchers in the spread of RA between joints, Dr. Orange added.
Patients do homework
The investigators began by enrolling four patients, followed for 1-4 years, who met 2010 American College of Rheumatology–European League Against Rheumatism criteria and who were seropositive for anti–cyclic citrullinated peptide antibodies.
They assessed disease activity from patient homes weekly or during escalation of flares up to four times daily, with the Routine Assessment of Patient Index Data 3 (RAPID3) questionnaire, as well as monthly clinic visits. At clinic visits during flares, disease activity was assessed using both the RAPID3 and 28-joint Disease Activity Score.
The patients performed fingerstick blood collection and mailed the samples overnight each week to Rockefeller University, where RNA was extracted and sequenced. The investigators identified gene transcripts that were differentially expressed in blood prior to flares, and compared them with data profiles derived from synovial single-cell RNA sequencing.
To validate the findings, the researchers used flow cytometry and sorted blood-cell RNA sequencing of samples from an additional 19 patients with RA.
They found that a total of 2,613 genes were differentially expressed during a flare, compared with baseline, and that expression of 1,437 of these genes was increased during a flare, with the remaining 1,176 decreased during flares.
Before the storm
Focusing on two flare-antecedent clusters of genes, they identified one cluster of transcripts that increased 2 weeks before a flare, enriched with genes coding for developmental pathways for naive B cells (that is, not yet exposed to antigens) and leukocytes.
The second cluster included gene transcripts that increased during the week before a flare, then decreased over the duration of the flare. Genes in this cluster were enriched for pathways that were unexpected in typical blood specimens, including genes involved in cartilage morphogenesis, endochondral bone growth, and extracellular matrix organization. The gene activity suggested the presence of a mesenchymal cell, they wrote.
The RNA expression profiles of these newly identified PRIME cells were very similar to those of synovial fibroblasts, and the investigators speculated that PRIME cells may be synovial fibroblast precursors.
They proposed a model of RA exacerbation in which PRIME cells become activated by B cells in the weeks immediately preceding a flare, and then migrate out of blood into the synovium.
The investigators are currently investigating “how reproducible this signature is in different flares in patients on different types of background therapy, and then we’re very interested in looking at the upstream triggers of the B cell and the PRIME cell,” Dr. Orange said.
“One of the reasons this is very exciting is that there are these signatures that can be found when patients are clearly asymptomatic but about to flare, and if we can intervene at that time, then the patients won’t have to live through a flare, they won’t have to have that experience,” she said.
Pros and cons
In an editorial accompanying the study, Ellen M. Gravallese, MD, from Brigham and Women’s Hospital in Boston and William H. Robinson, MD, PhD, from Stanford (Calif.) University and the Veterans Affairs Palo Alto (Calif.) Health Care System wrote that the study demonstrates an important method for identifying genetic contributions to many different types of disease.
“Orange and colleagues show that intensively collected longitudinal data from a small sample of patients can be used to identify dysregulated transcriptional signatures that are not recognized by classical cross-sectional studies. This study illustrates the exciting potential of longitudinal genomics to identify key antecedents of disease flares in an approach that may be applicable to the investigation of pathogenic and protective immune responses in a wide range of human diseases,” they wrote.
Rheumatology researcher Christopher D. Buckley, MBBS, DPhil, from the University of Birmingham (England), who was not involved in the study, said that the use of blood samples is both a strength and a weakness of the study.
“Blood is much easier to get than synovial tissue, but synovial tissue is important. If I’m trying to look at the blood and trying to make an inference about what’s going on in the synovium, if I don’t look at the synovium I don’t know what the link between the blood and synovium is,” he said in an interview.
On the plus side, “the big advantage about looking at blood is that you do multiple time points, which is really cool,” he said.
Dr. Buckley is a coauthor of a recent paper in Nature Medicine – published after the study by Dr. Orange and colleagues was accepted by the New England Journal of Medicine – showing that a population of macrophages in synovium was highly predictive of remission in patients with RA, and that therapeutic modulation of these macrophages has the potential as a treatment strategy for RA.
“We are very keen to understand the cellular basis of disease. We’re very good at understanding genes, but genes have to work in cells, and cells make organs, so the cells are critical,” he said.
The paper adds fuel to a controversy that has been raging among rheumatology researchers for more than a decade: the “flying fibroblast” hypothesis, which suggests that fibroblasts can migrate from one joint to another, hence spreading the disease in a manner akin to cancer metastases.
“It’s been quite controversial whether these cells like fibroblasts can exist in the blood, or whether they’re found in sufficient number in the blood,” Dr. Buckley said. “The fact that they have identified these PRIME cells is fascinating, because that’s going to cause us to go back and reinvestigate the whole flying fibroblast story.”
His colleague John Isaacs, MBBS, PhD, from Newcastle (England) University, is principal investigator for the BIO-FLARE study, in which participants with RA stop taking their disease-modifying antirheumatic drugs under close supervision of researchers. The investigators then study the patients looking for flare signals as well as the biology of flares themselves.
“As it happens, our protocols would not pick up this particular cell, because we have not been focusing on the stroma, at least not in peripheral blood. We’ve all been looking at synovium as part of BIO-FLARE,” he said in an interview. “We will be looking for this cell now that we have seen this research, and certainly we would want to replicate.”
He agreed with Dr. Buckley’s observation that the PRIME cell data may revive the flying fibroblast hypothesis. “This is a great paper in a top clinical journal. What isn’t there is mechanism. That’s the thing we’re all going to want to understand now: Where do the cells come from, how do they actually trigger flares, and how do they go down as flare starts?”
Both Dr. Buckley and Dr. Isaacs agreed that the study findings point to important new avenues of research, but also noted that the study was small, involving a total of only 23 patients, and that replication of the findings and elucidation of the mechanism of PRIME cell generation and disposition will be required.
The study was supported by grants from the National Institutes of Health, Simons Foundation, Robertson Foundation, Rheumatology Research Foundation, Bernard and Irene Schwartz Foundation, the Iris and Junming Le Foundation, and Rockefeller University. Dr. Orange disclosed a provisional patent for the discovery of the PRIME cells. Dr. Buckley and Dr. Isaacs reported no relevant conflicts of interest.
SOURCE: Orange DE et al. N Engl J Med. 2020 Jul 15. doi: 10.1056/NEJMoa2004114.
A newly identified circulating cell type may be a reliable marker for impending RA flares. The discovery and description of the cells, which bear a “striking” similarity to synovial fibroblasts, provide important clues to the origins of RA and progressive joint inflammation, investigators say.
By studying longitudinally collected blood samples from four patients with RA over 4 years, Dana E. Orange, MD and colleagues at Rockefeller University, New York, identified a pattern of B-cell activation and expansion of circulating cells that are negative for CD45 and CD31 expression, and positive for PDPN, dubbed preinflammatory mesenchymal or “PRIME” cells.
Expansion of PRIME cells in circulation increased dramatically in the weeks leading up to a flare and decreased during a flare, suggesting the possibility of a serum assay for predicting flares and allowing for early intervention to ameliorate or prevent disabling consequences, the investigators wrote in a study published in the New England Journal of Medicine.
“Our hope is that this will be a diagnostic in the future, but we need to study it in more patients to see how it will perform,” Dr. Orange said in an interview, adding that the cells, if shown to be pathogenic, could also be targets for new therapeutic strategies.
RNA sequencing
Dr. Orange and colleagues discovered the PRIME cells through a novel clinical and technical protocol involving home collection of blood by patients and longitudinal RNA sequencing to study gene expression profiles during times of both disease quiescence and flares, and noticed a distinct pattern of PRIME cell expansion, depletion, and gene expression.
“Looking at their gene expression profiles, they overlapped with fibroblasts that reside in inflamed rheumatoid arthritis synovium, and in an animal model those types of fibroblasts were important for allowing entry of inflammatory infiltrates around the joint,” she said.
PRIME cells may be a precursor of synovial fibroblasts, which have been implicated by some researchers in the spread of RA between joints, Dr. Orange added.
Patients do homework
The investigators began by enrolling four patients, followed for 1-4 years, who met 2010 American College of Rheumatology–European League Against Rheumatism criteria and who were seropositive for anti–cyclic citrullinated peptide antibodies.
They assessed disease activity from patient homes weekly or during escalation of flares up to four times daily, with the Routine Assessment of Patient Index Data 3 (RAPID3) questionnaire, as well as monthly clinic visits. At clinic visits during flares, disease activity was assessed using both the RAPID3 and 28-joint Disease Activity Score.
The patients performed fingerstick blood collection and mailed the samples overnight each week to Rockefeller University, where RNA was extracted and sequenced. The investigators identified gene transcripts that were differentially expressed in blood prior to flares, and compared them with data profiles derived from synovial single-cell RNA sequencing.
To validate the findings, the researchers used flow cytometry and sorted blood-cell RNA sequencing of samples from an additional 19 patients with RA.
They found that a total of 2,613 genes were differentially expressed during a flare, compared with baseline, and that expression of 1,437 of these genes was increased during a flare, with the remaining 1,176 decreased during flares.
Before the storm
Focusing on two flare-antecedent clusters of genes, they identified one cluster of transcripts that increased 2 weeks before a flare, enriched with genes coding for developmental pathways for naive B cells (that is, not yet exposed to antigens) and leukocytes.
The second cluster included gene transcripts that increased during the week before a flare, then decreased over the duration of the flare. Genes in this cluster were enriched for pathways that were unexpected in typical blood specimens, including genes involved in cartilage morphogenesis, endochondral bone growth, and extracellular matrix organization. The gene activity suggested the presence of a mesenchymal cell, they wrote.
The RNA expression profiles of these newly identified PRIME cells were very similar to those of synovial fibroblasts, and the investigators speculated that PRIME cells may be synovial fibroblast precursors.
They proposed a model of RA exacerbation in which PRIME cells become activated by B cells in the weeks immediately preceding a flare, and then migrate out of blood into the synovium.
The investigators are currently investigating “how reproducible this signature is in different flares in patients on different types of background therapy, and then we’re very interested in looking at the upstream triggers of the B cell and the PRIME cell,” Dr. Orange said.
“One of the reasons this is very exciting is that there are these signatures that can be found when patients are clearly asymptomatic but about to flare, and if we can intervene at that time, then the patients won’t have to live through a flare, they won’t have to have that experience,” she said.
Pros and cons
In an editorial accompanying the study, Ellen M. Gravallese, MD, from Brigham and Women’s Hospital in Boston and William H. Robinson, MD, PhD, from Stanford (Calif.) University and the Veterans Affairs Palo Alto (Calif.) Health Care System wrote that the study demonstrates an important method for identifying genetic contributions to many different types of disease.
“Orange and colleagues show that intensively collected longitudinal data from a small sample of patients can be used to identify dysregulated transcriptional signatures that are not recognized by classical cross-sectional studies. This study illustrates the exciting potential of longitudinal genomics to identify key antecedents of disease flares in an approach that may be applicable to the investigation of pathogenic and protective immune responses in a wide range of human diseases,” they wrote.
Rheumatology researcher Christopher D. Buckley, MBBS, DPhil, from the University of Birmingham (England), who was not involved in the study, said that the use of blood samples is both a strength and a weakness of the study.
“Blood is much easier to get than synovial tissue, but synovial tissue is important. If I’m trying to look at the blood and trying to make an inference about what’s going on in the synovium, if I don’t look at the synovium I don’t know what the link between the blood and synovium is,” he said in an interview.
On the plus side, “the big advantage about looking at blood is that you do multiple time points, which is really cool,” he said.
Dr. Buckley is a coauthor of a recent paper in Nature Medicine – published after the study by Dr. Orange and colleagues was accepted by the New England Journal of Medicine – showing that a population of macrophages in synovium was highly predictive of remission in patients with RA, and that therapeutic modulation of these macrophages has the potential as a treatment strategy for RA.
“We are very keen to understand the cellular basis of disease. We’re very good at understanding genes, but genes have to work in cells, and cells make organs, so the cells are critical,” he said.
The paper adds fuel to a controversy that has been raging among rheumatology researchers for more than a decade: the “flying fibroblast” hypothesis, which suggests that fibroblasts can migrate from one joint to another, hence spreading the disease in a manner akin to cancer metastases.
“It’s been quite controversial whether these cells like fibroblasts can exist in the blood, or whether they’re found in sufficient number in the blood,” Dr. Buckley said. “The fact that they have identified these PRIME cells is fascinating, because that’s going to cause us to go back and reinvestigate the whole flying fibroblast story.”
His colleague John Isaacs, MBBS, PhD, from Newcastle (England) University, is principal investigator for the BIO-FLARE study, in which participants with RA stop taking their disease-modifying antirheumatic drugs under close supervision of researchers. The investigators then study the patients looking for flare signals as well as the biology of flares themselves.
“As it happens, our protocols would not pick up this particular cell, because we have not been focusing on the stroma, at least not in peripheral blood. We’ve all been looking at synovium as part of BIO-FLARE,” he said in an interview. “We will be looking for this cell now that we have seen this research, and certainly we would want to replicate.”
He agreed with Dr. Buckley’s observation that the PRIME cell data may revive the flying fibroblast hypothesis. “This is a great paper in a top clinical journal. What isn’t there is mechanism. That’s the thing we’re all going to want to understand now: Where do the cells come from, how do they actually trigger flares, and how do they go down as flare starts?”
Both Dr. Buckley and Dr. Isaacs agreed that the study findings point to important new avenues of research, but also noted that the study was small, involving a total of only 23 patients, and that replication of the findings and elucidation of the mechanism of PRIME cell generation and disposition will be required.
The study was supported by grants from the National Institutes of Health, Simons Foundation, Robertson Foundation, Rheumatology Research Foundation, Bernard and Irene Schwartz Foundation, the Iris and Junming Le Foundation, and Rockefeller University. Dr. Orange disclosed a provisional patent for the discovery of the PRIME cells. Dr. Buckley and Dr. Isaacs reported no relevant conflicts of interest.
SOURCE: Orange DE et al. N Engl J Med. 2020 Jul 15. doi: 10.1056/NEJMoa2004114.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: A newly identified cell type may be predictive of RA flares.
Major finding: Preinflammatory mesenchymal cells were expanded in the week preceding a flare and decreased during the flare.
Study details: A longitudinal observational study of 23 patients with RA.
Disclosures: The study was supported by grants from the National Institutes of Health, Simons Foundation, Robertson Foundation, Rheumatology Research Foundation, Bernard and Irene Schwartz Foundation, the Iris and Junming Le Foundation, and Rockefeller University. Dr. Orange disclosed a provisional patent for the discovery of the preinflammatory mesenchymal cells.
Source: Orange DE et al. N Engl J Med. 2020 Jul 15. doi: 10.1056/NEJMoa2004114.
Steroids linked to increased hypertension in RA
Although the adverse effects of systemic glucocorticoids (GCs) are well known, their association with hypertension in rheumatoid arthritis (RA) has been unclear. Now, a large population-based study shows that the drugs are linked to a 17% overall increased risk for incident hypertension among patients with RA.
Further, when the researchers stratified participants by dose category, they found that doses higher than 7.5 mg were significantly associated with hypertension. Cumulative dosage was not tied to any clear pattern of risk.
The authors, led by Ruth E. Costello, a researcher at the Centre for Epidemiology Versus Arthritis in the Centre for Musculoskeletal Research at the University of Manchester (England) concluded that patients who are taking these drugs for the treatment of RA should be monitored for high blood pressure, which is an important but modifiable cardiovascular risk factor, and treated appropriately.
The results of Ms. Costello and colleagues’ study were published June 27 in Rheumatology.
“While fractures associated with these steroid drugs are well studied, hypertension is a side effect that seems to have been less well studied, and yet it is an important cardiovascular risk factor that can be managed,” Ms. Costello said in an interview.
To better understand the possible association, Ms. Costello and colleagues identified 17,760 patients who were newly diagnosed with RA between 1992 and 2019 and were included in the Clinical Practice Research Datalink, which represents about 7% of the U.K. population. None of the patients had hypertension at initial RA diagnosis. Slightly more than two-thirds were women (68.1%), and the mean age was 56.3 years.
Of those patients, 7,421 (41.8%) were prescribed GCs during postdiagnosis follow-up. Most patients (73%) were followed for at least 2 years.
Patients who used GCs were slightly older than never-users (mean age, 57.7 vs. 55.3 years), were predominantly women, had a history of smoking, and had more comorbidities.
The overall incidence rate (IR) of hypertension was 64.1 per 1,000 person-years (95% confidence interval, 62.5-65.7). There were 6,243 cases of incident hypertension over 97,547 person-years of follow-up.
Among those exposed to GCs, 1,321 patients developed hypertension, for an IR of 87.6 per 1,000 person-years. Among unexposed participants, the IR for hypertension was 59.7 per 1,000 person-years. In Cox proportional hazards modeling, GC use was associated with a 17% increased risk for hypertension (hazard ratio, 1.17; 95% CI, 1.10-1.24).
The researchers noted that 40% of GC users with hypertension were not prescribed an antihypertensive agent at any point during the study. “Whilst some may have been offered lifestyle advice, left untreated this has important implications in terms of addressing modifiable risk factors in an RA population already at increased risk of CV disease,” they wrote.
They noted that cardiovascular disease is a major driver of the elevated mortality risk seen among adults with RA compared with the general population and that recent treatment recommendations address management of cardiovascular risks in these patients.
“There are several routes by which GCs may promote cardiovascular disease, including hypertension, metabolic changes, diabetes, and weight gain. We don’t currently know the extent to which each of these individual mechanisms may be increasing cardiovascular disease,” said Ms. Costello.
“Glucocorticoids increase fluid retention and promote obesity and hypertension,” said Rajat S. Bhatt, MD, a rheumatologist at Prime Rheumatology and Memorial Hermann Katy Hospital in Richmond, Texas, who sees hypertension in GC users in his clinical practice. “So patients need to be monitored for these risk factors,” he said in an interview.
Although hypertension may be a significant factor in the increase in cardiovascular disease in the RA population, Dr. Bhatt said the major driver is likely the intrinsic inflammatory state caused by the disease itself. As to why the GC-hypertension connection has flown under the radar in RA, he added, “That specific link has been difficult to tease out since RA patients are often on multiple medications.”
In regard to the role of dosage, Dr. Bhatt said that hypertension risk increases with higher GC doses, as the U.K. study indicates, and usually subsides when patients stop using GCs.
“Whether the observed dose association is causal or influenced by the underlying disease severity, our results suggest we should be vigilant in patients on all doses of GC, especially higher doses,” Ms. Costello added.
In regard to using drugs that are less cardiotoxic than GCs, Dr. Bhatt said that there are clinical scenarios in which GC therapy is the best choice, so just switching to nonsteroidal drugs is no panacea. “All RA drugs have adverse side effects, and anyway, the goal of rheumatology treatment is always to get patients off corticosteroids as soon as possible,” he said.
Ms. Costello and colleagues noted that their results are consonant with earlier research, including a single-center, cross-sectional study in which less than 6 months’ use of prednisolone at a median dose of 7.5 mg was associated with hypertension. In a German registry study, among patients who received doses of less than 7.5 mg for less than 6 months, there were higher rates of self-reported elevations in blood pressure.
The findings are at odds, however, with a recent matched-cohort study, which also used data from the Clinical Practice Research Datalink. That study found no association between GC use and hypertension.
GCs have come under increasing scrutiny in regard to several diseases. A study published July 7 found that even short-term courses of a few days’ duration entail risks for serious adverse events.
Ms. Costello’s group says that an estimate of GC-related incident hypertension in RA should allow more informed treatment decisions and that their findings highlight the ongoing need to monitor for and address this risk.
The study was supported by the Centre for Epidemiology Versus Arthritis and by the National Institute for Health Research Manchester Biomedical Research Centre. Coauthor William G. Dixon, PhD, has received consultancy fees from Google and Bayer unrelated to this study. Dr. Bhatt has disclosed no relevant financial relationships.
SOURCE: Costello RE et al. Rheumatology. 2020 June 27. doi: 10.1093/rheumatology/keaa209.
A version of this article originally appeared on Medscape.com.
Although the adverse effects of systemic glucocorticoids (GCs) are well known, their association with hypertension in rheumatoid arthritis (RA) has been unclear. Now, a large population-based study shows that the drugs are linked to a 17% overall increased risk for incident hypertension among patients with RA.
Further, when the researchers stratified participants by dose category, they found that doses higher than 7.5 mg were significantly associated with hypertension. Cumulative dosage was not tied to any clear pattern of risk.
The authors, led by Ruth E. Costello, a researcher at the Centre for Epidemiology Versus Arthritis in the Centre for Musculoskeletal Research at the University of Manchester (England) concluded that patients who are taking these drugs for the treatment of RA should be monitored for high blood pressure, which is an important but modifiable cardiovascular risk factor, and treated appropriately.
The results of Ms. Costello and colleagues’ study were published June 27 in Rheumatology.
“While fractures associated with these steroid drugs are well studied, hypertension is a side effect that seems to have been less well studied, and yet it is an important cardiovascular risk factor that can be managed,” Ms. Costello said in an interview.
To better understand the possible association, Ms. Costello and colleagues identified 17,760 patients who were newly diagnosed with RA between 1992 and 2019 and were included in the Clinical Practice Research Datalink, which represents about 7% of the U.K. population. None of the patients had hypertension at initial RA diagnosis. Slightly more than two-thirds were women (68.1%), and the mean age was 56.3 years.
Of those patients, 7,421 (41.8%) were prescribed GCs during postdiagnosis follow-up. Most patients (73%) were followed for at least 2 years.
Patients who used GCs were slightly older than never-users (mean age, 57.7 vs. 55.3 years), were predominantly women, had a history of smoking, and had more comorbidities.
The overall incidence rate (IR) of hypertension was 64.1 per 1,000 person-years (95% confidence interval, 62.5-65.7). There were 6,243 cases of incident hypertension over 97,547 person-years of follow-up.
Among those exposed to GCs, 1,321 patients developed hypertension, for an IR of 87.6 per 1,000 person-years. Among unexposed participants, the IR for hypertension was 59.7 per 1,000 person-years. In Cox proportional hazards modeling, GC use was associated with a 17% increased risk for hypertension (hazard ratio, 1.17; 95% CI, 1.10-1.24).
The researchers noted that 40% of GC users with hypertension were not prescribed an antihypertensive agent at any point during the study. “Whilst some may have been offered lifestyle advice, left untreated this has important implications in terms of addressing modifiable risk factors in an RA population already at increased risk of CV disease,” they wrote.
They noted that cardiovascular disease is a major driver of the elevated mortality risk seen among adults with RA compared with the general population and that recent treatment recommendations address management of cardiovascular risks in these patients.
“There are several routes by which GCs may promote cardiovascular disease, including hypertension, metabolic changes, diabetes, and weight gain. We don’t currently know the extent to which each of these individual mechanisms may be increasing cardiovascular disease,” said Ms. Costello.
“Glucocorticoids increase fluid retention and promote obesity and hypertension,” said Rajat S. Bhatt, MD, a rheumatologist at Prime Rheumatology and Memorial Hermann Katy Hospital in Richmond, Texas, who sees hypertension in GC users in his clinical practice. “So patients need to be monitored for these risk factors,” he said in an interview.
Although hypertension may be a significant factor in the increase in cardiovascular disease in the RA population, Dr. Bhatt said the major driver is likely the intrinsic inflammatory state caused by the disease itself. As to why the GC-hypertension connection has flown under the radar in RA, he added, “That specific link has been difficult to tease out since RA patients are often on multiple medications.”
In regard to the role of dosage, Dr. Bhatt said that hypertension risk increases with higher GC doses, as the U.K. study indicates, and usually subsides when patients stop using GCs.
“Whether the observed dose association is causal or influenced by the underlying disease severity, our results suggest we should be vigilant in patients on all doses of GC, especially higher doses,” Ms. Costello added.
In regard to using drugs that are less cardiotoxic than GCs, Dr. Bhatt said that there are clinical scenarios in which GC therapy is the best choice, so just switching to nonsteroidal drugs is no panacea. “All RA drugs have adverse side effects, and anyway, the goal of rheumatology treatment is always to get patients off corticosteroids as soon as possible,” he said.
Ms. Costello and colleagues noted that their results are consonant with earlier research, including a single-center, cross-sectional study in which less than 6 months’ use of prednisolone at a median dose of 7.5 mg was associated with hypertension. In a German registry study, among patients who received doses of less than 7.5 mg for less than 6 months, there were higher rates of self-reported elevations in blood pressure.
The findings are at odds, however, with a recent matched-cohort study, which also used data from the Clinical Practice Research Datalink. That study found no association between GC use and hypertension.
GCs have come under increasing scrutiny in regard to several diseases. A study published July 7 found that even short-term courses of a few days’ duration entail risks for serious adverse events.
Ms. Costello’s group says that an estimate of GC-related incident hypertension in RA should allow more informed treatment decisions and that their findings highlight the ongoing need to monitor for and address this risk.
The study was supported by the Centre for Epidemiology Versus Arthritis and by the National Institute for Health Research Manchester Biomedical Research Centre. Coauthor William G. Dixon, PhD, has received consultancy fees from Google and Bayer unrelated to this study. Dr. Bhatt has disclosed no relevant financial relationships.
SOURCE: Costello RE et al. Rheumatology. 2020 June 27. doi: 10.1093/rheumatology/keaa209.
A version of this article originally appeared on Medscape.com.
Although the adverse effects of systemic glucocorticoids (GCs) are well known, their association with hypertension in rheumatoid arthritis (RA) has been unclear. Now, a large population-based study shows that the drugs are linked to a 17% overall increased risk for incident hypertension among patients with RA.
Further, when the researchers stratified participants by dose category, they found that doses higher than 7.5 mg were significantly associated with hypertension. Cumulative dosage was not tied to any clear pattern of risk.
The authors, led by Ruth E. Costello, a researcher at the Centre for Epidemiology Versus Arthritis in the Centre for Musculoskeletal Research at the University of Manchester (England) concluded that patients who are taking these drugs for the treatment of RA should be monitored for high blood pressure, which is an important but modifiable cardiovascular risk factor, and treated appropriately.
The results of Ms. Costello and colleagues’ study were published June 27 in Rheumatology.
“While fractures associated with these steroid drugs are well studied, hypertension is a side effect that seems to have been less well studied, and yet it is an important cardiovascular risk factor that can be managed,” Ms. Costello said in an interview.
To better understand the possible association, Ms. Costello and colleagues identified 17,760 patients who were newly diagnosed with RA between 1992 and 2019 and were included in the Clinical Practice Research Datalink, which represents about 7% of the U.K. population. None of the patients had hypertension at initial RA diagnosis. Slightly more than two-thirds were women (68.1%), and the mean age was 56.3 years.
Of those patients, 7,421 (41.8%) were prescribed GCs during postdiagnosis follow-up. Most patients (73%) were followed for at least 2 years.
Patients who used GCs were slightly older than never-users (mean age, 57.7 vs. 55.3 years), were predominantly women, had a history of smoking, and had more comorbidities.
The overall incidence rate (IR) of hypertension was 64.1 per 1,000 person-years (95% confidence interval, 62.5-65.7). There were 6,243 cases of incident hypertension over 97,547 person-years of follow-up.
Among those exposed to GCs, 1,321 patients developed hypertension, for an IR of 87.6 per 1,000 person-years. Among unexposed participants, the IR for hypertension was 59.7 per 1,000 person-years. In Cox proportional hazards modeling, GC use was associated with a 17% increased risk for hypertension (hazard ratio, 1.17; 95% CI, 1.10-1.24).
The researchers noted that 40% of GC users with hypertension were not prescribed an antihypertensive agent at any point during the study. “Whilst some may have been offered lifestyle advice, left untreated this has important implications in terms of addressing modifiable risk factors in an RA population already at increased risk of CV disease,” they wrote.
They noted that cardiovascular disease is a major driver of the elevated mortality risk seen among adults with RA compared with the general population and that recent treatment recommendations address management of cardiovascular risks in these patients.
“There are several routes by which GCs may promote cardiovascular disease, including hypertension, metabolic changes, diabetes, and weight gain. We don’t currently know the extent to which each of these individual mechanisms may be increasing cardiovascular disease,” said Ms. Costello.
“Glucocorticoids increase fluid retention and promote obesity and hypertension,” said Rajat S. Bhatt, MD, a rheumatologist at Prime Rheumatology and Memorial Hermann Katy Hospital in Richmond, Texas, who sees hypertension in GC users in his clinical practice. “So patients need to be monitored for these risk factors,” he said in an interview.
Although hypertension may be a significant factor in the increase in cardiovascular disease in the RA population, Dr. Bhatt said the major driver is likely the intrinsic inflammatory state caused by the disease itself. As to why the GC-hypertension connection has flown under the radar in RA, he added, “That specific link has been difficult to tease out since RA patients are often on multiple medications.”
In regard to the role of dosage, Dr. Bhatt said that hypertension risk increases with higher GC doses, as the U.K. study indicates, and usually subsides when patients stop using GCs.
“Whether the observed dose association is causal or influenced by the underlying disease severity, our results suggest we should be vigilant in patients on all doses of GC, especially higher doses,” Ms. Costello added.
In regard to using drugs that are less cardiotoxic than GCs, Dr. Bhatt said that there are clinical scenarios in which GC therapy is the best choice, so just switching to nonsteroidal drugs is no panacea. “All RA drugs have adverse side effects, and anyway, the goal of rheumatology treatment is always to get patients off corticosteroids as soon as possible,” he said.
Ms. Costello and colleagues noted that their results are consonant with earlier research, including a single-center, cross-sectional study in which less than 6 months’ use of prednisolone at a median dose of 7.5 mg was associated with hypertension. In a German registry study, among patients who received doses of less than 7.5 mg for less than 6 months, there were higher rates of self-reported elevations in blood pressure.
The findings are at odds, however, with a recent matched-cohort study, which also used data from the Clinical Practice Research Datalink. That study found no association between GC use and hypertension.
GCs have come under increasing scrutiny in regard to several diseases. A study published July 7 found that even short-term courses of a few days’ duration entail risks for serious adverse events.
Ms. Costello’s group says that an estimate of GC-related incident hypertension in RA should allow more informed treatment decisions and that their findings highlight the ongoing need to monitor for and address this risk.
The study was supported by the Centre for Epidemiology Versus Arthritis and by the National Institute for Health Research Manchester Biomedical Research Centre. Coauthor William G. Dixon, PhD, has received consultancy fees from Google and Bayer unrelated to this study. Dr. Bhatt has disclosed no relevant financial relationships.
SOURCE: Costello RE et al. Rheumatology. 2020 June 27. doi: 10.1093/rheumatology/keaa209.
A version of this article originally appeared on Medscape.com.
FROM RHEUMATOLOGY










