User login
Strength of morning RA symptoms are correlated with disease’s activity
Rheumatoid arthritis (RA) patients’ severity of morning stiffness and morning pain have stronger correlations with their disease’s activity than does the duration of their morning stiffness, according to an analysis of 350 patients who participated in a randomized, controlled trial.
During the 12-week study of patients with symptomatic RA despite treatment with disease-modifying antirheumatic drugs, 231 of the patients added delayed-release prednisone to their treatment and the remaining 119 patients took a placebo. The patients’ disease activity was measured with the ACR20, 28-joint Disease Activity Score (DAS28), and the Health Assessment Questionnaire-Disability Index, while Pearson correlations were used to determine relationships between patients’ responses to treatment and their disease activity.
The correlations between severity of morning stiffness or intensity of pain on waking and DAS28 or ACR20 ranged from 0.44 to 0.48 for the full study cohort. The weaker correlation between duration of morning stiffness and disease activity ranged from 0.24 to 0.28. The researchers also found a statistically significant 0.91 correlation between the severity of morning stiffness and intensity of morning pain.
“These findings suggest that severity and duration of morning stiffness may provide subtly different measures of RA and its impact on patients. The superior measurement characteristics of the severity of morning stiffness favor this as the preferred outcome measure,” wrote first author Dr. Maarten Boers and his colleagues.
Find the full study in Arthritis Care & Research (doi:10.1002/acr.22592).
Rheumatoid arthritis (RA) patients’ severity of morning stiffness and morning pain have stronger correlations with their disease’s activity than does the duration of their morning stiffness, according to an analysis of 350 patients who participated in a randomized, controlled trial.
During the 12-week study of patients with symptomatic RA despite treatment with disease-modifying antirheumatic drugs, 231 of the patients added delayed-release prednisone to their treatment and the remaining 119 patients took a placebo. The patients’ disease activity was measured with the ACR20, 28-joint Disease Activity Score (DAS28), and the Health Assessment Questionnaire-Disability Index, while Pearson correlations were used to determine relationships between patients’ responses to treatment and their disease activity.
The correlations between severity of morning stiffness or intensity of pain on waking and DAS28 or ACR20 ranged from 0.44 to 0.48 for the full study cohort. The weaker correlation between duration of morning stiffness and disease activity ranged from 0.24 to 0.28. The researchers also found a statistically significant 0.91 correlation between the severity of morning stiffness and intensity of morning pain.
“These findings suggest that severity and duration of morning stiffness may provide subtly different measures of RA and its impact on patients. The superior measurement characteristics of the severity of morning stiffness favor this as the preferred outcome measure,” wrote first author Dr. Maarten Boers and his colleagues.
Find the full study in Arthritis Care & Research (doi:10.1002/acr.22592).
Rheumatoid arthritis (RA) patients’ severity of morning stiffness and morning pain have stronger correlations with their disease’s activity than does the duration of their morning stiffness, according to an analysis of 350 patients who participated in a randomized, controlled trial.
During the 12-week study of patients with symptomatic RA despite treatment with disease-modifying antirheumatic drugs, 231 of the patients added delayed-release prednisone to their treatment and the remaining 119 patients took a placebo. The patients’ disease activity was measured with the ACR20, 28-joint Disease Activity Score (DAS28), and the Health Assessment Questionnaire-Disability Index, while Pearson correlations were used to determine relationships between patients’ responses to treatment and their disease activity.
The correlations between severity of morning stiffness or intensity of pain on waking and DAS28 or ACR20 ranged from 0.44 to 0.48 for the full study cohort. The weaker correlation between duration of morning stiffness and disease activity ranged from 0.24 to 0.28. The researchers also found a statistically significant 0.91 correlation between the severity of morning stiffness and intensity of morning pain.
“These findings suggest that severity and duration of morning stiffness may provide subtly different measures of RA and its impact on patients. The superior measurement characteristics of the severity of morning stiffness favor this as the preferred outcome measure,” wrote first author Dr. Maarten Boers and his colleagues.
Find the full study in Arthritis Care & Research (doi:10.1002/acr.22592).
RA patients face significant out-of-pocket costs under Medicare
Medicare Part D recipients with rheumatoid arthritis face an average of over $2,700 per year in out-of-pocket expenses for biologic disease-modifying antirheumatic drugs, a figure that constitutes a “substantial disincentive” to adherence, according to Dr. Jinoos Yazdany of the division of rheumatology at the University of California, San Francisco, and her colleagues.
For patients with rheumatoid arthritis (RA) who do not respond to less costly nonbiologic disease-modifying antirheumatic drugs (DMARDs), early and aggressive treatment with biologic agents is key to slowing disease progression. However, biologic DMARDs can cost $20,000 or more per year, and may need to be taken over the course of a lifetime.
Dr. Yazdany and her colleagues’ current study examined 2013 data for all 2,737 Medicare Part D formularies, drawn from the Centers for Medicare & Medicaid Services Prescription Drug Plan Formulary and Pharmacy Network Files. Outpatient formularies from all 50 states and the District of Columbia were included. Although all plans covered at least one biologic DMARD, 95% required prior authorization and patients’ coinsurance averaged nearly 30% (Arthritis Rheumatol. 2015 April 21 [doi:10.1002/art.39079]).
A study from 2006 previously showed that estimated out-of-pocket costs for biological DMARDs would exceed $4,000 annually for low-income Medicare beneficiaries in the first year of Part D (Arthritis Rheum. 2009;61:745-54). The current study indicates that even 8 years after Part D plans were implemented, they continued “to use high cost sharing as a primary cost-control mechanism for biologic DMARDs, placing a substantial financial burden on patients who require such drugs for adequate control of their RA symptoms,” the investigators wrote.
Dr. Yazdany and her associates reported that during the period before spending limits were reached and catastrophic coverage was triggered, mean patient cost-sharing for biologic DMARDs was $835 per month. Infliximab was the least expensive option at $269 per month, while anakinra cost patients a mean $2,993 per month. Medicare Advantage plans were more likely to cover biologic agents, but at a higher average patient cost of $862 per month.
Of the nine nonbiologic DMARDs, six were covered by almost all plans with copayments of $5-$10 per month; the least expensive option in each plan averaged $48 per year in out-of-pocket costs. Prior authorizations were not needed for nonbiologic agents.
Dr. Yazdany and colleagues raised the question of whether the policies of prior authorizations, tiered coverage, and high levels of patient cost-sharing contribute to achieving high value care for those with RA who fail first-line therapy. Evidence-based guidelines would move these patients to biologic therapy, but the high level of patient cost-sharing in the current Medicare payment structure has previously been shown to reduce adherence, potentially speeding disease progression and worsening outcomes.
“We have avoided the issue of cost-effectiveness analysis,” Dr. Yazdany said, noting that to date, Medicare has not used reference pricing or negotiated drug costs, in contrast to government payers in many other countries. “We should all work to decrease out-of-pocket costs for people grappling with this serious disease,” she said. “The current situation is really unacceptable. That’s why we need to talk about it.”
Policies for better patient stratification are needed to reduce overall out-of-pocket expenses for those with RA using biologic DMARDs. This study is a very important step, and should help to generate more – and much-needed – conversation around this issue.

|
| Dr. Michael Ward |
Adding more information about younger RA patients will help refine knowledge of who should be using these agents and when they are most effective. There is a growing effort to establish a multilayer research database that would cover a younger population to augment the large and publicly available Medicare database of those 65 and older. CMS data are widely used and publicly available, and effectively track a national population, but are limited by capturing only information about older adults who are further along in their disease course.
An important part of the cost-effectiveness conversation going forward will revolve around understanding whether there is utility in using biologic DMARDs very early in the disease process of RA to gain control of the disease. This early intervention may allow patients to taper off medication at a later point. However, clinicians currently don’t have clear indications for when to taper a patient off a biologic DMARD. Better delineation of answers to these clinical questions will help to clarify the cost-effectiveness analysis.
Dr. Michael M. Ward is a senior investigator at the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Bethesda, Md. These remarks were drawn from a telephone interview. He reported no disclosures.
Policies for better patient stratification are needed to reduce overall out-of-pocket expenses for those with RA using biologic DMARDs. This study is a very important step, and should help to generate more – and much-needed – conversation around this issue.

|
| Dr. Michael Ward |
Adding more information about younger RA patients will help refine knowledge of who should be using these agents and when they are most effective. There is a growing effort to establish a multilayer research database that would cover a younger population to augment the large and publicly available Medicare database of those 65 and older. CMS data are widely used and publicly available, and effectively track a national population, but are limited by capturing only information about older adults who are further along in their disease course.
An important part of the cost-effectiveness conversation going forward will revolve around understanding whether there is utility in using biologic DMARDs very early in the disease process of RA to gain control of the disease. This early intervention may allow patients to taper off medication at a later point. However, clinicians currently don’t have clear indications for when to taper a patient off a biologic DMARD. Better delineation of answers to these clinical questions will help to clarify the cost-effectiveness analysis.
Dr. Michael M. Ward is a senior investigator at the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Bethesda, Md. These remarks were drawn from a telephone interview. He reported no disclosures.
Policies for better patient stratification are needed to reduce overall out-of-pocket expenses for those with RA using biologic DMARDs. This study is a very important step, and should help to generate more – and much-needed – conversation around this issue.

|
| Dr. Michael Ward |
Adding more information about younger RA patients will help refine knowledge of who should be using these agents and when they are most effective. There is a growing effort to establish a multilayer research database that would cover a younger population to augment the large and publicly available Medicare database of those 65 and older. CMS data are widely used and publicly available, and effectively track a national population, but are limited by capturing only information about older adults who are further along in their disease course.
An important part of the cost-effectiveness conversation going forward will revolve around understanding whether there is utility in using biologic DMARDs very early in the disease process of RA to gain control of the disease. This early intervention may allow patients to taper off medication at a later point. However, clinicians currently don’t have clear indications for when to taper a patient off a biologic DMARD. Better delineation of answers to these clinical questions will help to clarify the cost-effectiveness analysis.
Dr. Michael M. Ward is a senior investigator at the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Bethesda, Md. These remarks were drawn from a telephone interview. He reported no disclosures.
Medicare Part D recipients with rheumatoid arthritis face an average of over $2,700 per year in out-of-pocket expenses for biologic disease-modifying antirheumatic drugs, a figure that constitutes a “substantial disincentive” to adherence, according to Dr. Jinoos Yazdany of the division of rheumatology at the University of California, San Francisco, and her colleagues.
For patients with rheumatoid arthritis (RA) who do not respond to less costly nonbiologic disease-modifying antirheumatic drugs (DMARDs), early and aggressive treatment with biologic agents is key to slowing disease progression. However, biologic DMARDs can cost $20,000 or more per year, and may need to be taken over the course of a lifetime.
Dr. Yazdany and her colleagues’ current study examined 2013 data for all 2,737 Medicare Part D formularies, drawn from the Centers for Medicare & Medicaid Services Prescription Drug Plan Formulary and Pharmacy Network Files. Outpatient formularies from all 50 states and the District of Columbia were included. Although all plans covered at least one biologic DMARD, 95% required prior authorization and patients’ coinsurance averaged nearly 30% (Arthritis Rheumatol. 2015 April 21 [doi:10.1002/art.39079]).
A study from 2006 previously showed that estimated out-of-pocket costs for biological DMARDs would exceed $4,000 annually for low-income Medicare beneficiaries in the first year of Part D (Arthritis Rheum. 2009;61:745-54). The current study indicates that even 8 years after Part D plans were implemented, they continued “to use high cost sharing as a primary cost-control mechanism for biologic DMARDs, placing a substantial financial burden on patients who require such drugs for adequate control of their RA symptoms,” the investigators wrote.
Dr. Yazdany and her associates reported that during the period before spending limits were reached and catastrophic coverage was triggered, mean patient cost-sharing for biologic DMARDs was $835 per month. Infliximab was the least expensive option at $269 per month, while anakinra cost patients a mean $2,993 per month. Medicare Advantage plans were more likely to cover biologic agents, but at a higher average patient cost of $862 per month.
Of the nine nonbiologic DMARDs, six were covered by almost all plans with copayments of $5-$10 per month; the least expensive option in each plan averaged $48 per year in out-of-pocket costs. Prior authorizations were not needed for nonbiologic agents.
Dr. Yazdany and colleagues raised the question of whether the policies of prior authorizations, tiered coverage, and high levels of patient cost-sharing contribute to achieving high value care for those with RA who fail first-line therapy. Evidence-based guidelines would move these patients to biologic therapy, but the high level of patient cost-sharing in the current Medicare payment structure has previously been shown to reduce adherence, potentially speeding disease progression and worsening outcomes.
“We have avoided the issue of cost-effectiveness analysis,” Dr. Yazdany said, noting that to date, Medicare has not used reference pricing or negotiated drug costs, in contrast to government payers in many other countries. “We should all work to decrease out-of-pocket costs for people grappling with this serious disease,” she said. “The current situation is really unacceptable. That’s why we need to talk about it.”
Medicare Part D recipients with rheumatoid arthritis face an average of over $2,700 per year in out-of-pocket expenses for biologic disease-modifying antirheumatic drugs, a figure that constitutes a “substantial disincentive” to adherence, according to Dr. Jinoos Yazdany of the division of rheumatology at the University of California, San Francisco, and her colleagues.
For patients with rheumatoid arthritis (RA) who do not respond to less costly nonbiologic disease-modifying antirheumatic drugs (DMARDs), early and aggressive treatment with biologic agents is key to slowing disease progression. However, biologic DMARDs can cost $20,000 or more per year, and may need to be taken over the course of a lifetime.
Dr. Yazdany and her colleagues’ current study examined 2013 data for all 2,737 Medicare Part D formularies, drawn from the Centers for Medicare & Medicaid Services Prescription Drug Plan Formulary and Pharmacy Network Files. Outpatient formularies from all 50 states and the District of Columbia were included. Although all plans covered at least one biologic DMARD, 95% required prior authorization and patients’ coinsurance averaged nearly 30% (Arthritis Rheumatol. 2015 April 21 [doi:10.1002/art.39079]).
A study from 2006 previously showed that estimated out-of-pocket costs for biological DMARDs would exceed $4,000 annually for low-income Medicare beneficiaries in the first year of Part D (Arthritis Rheum. 2009;61:745-54). The current study indicates that even 8 years after Part D plans were implemented, they continued “to use high cost sharing as a primary cost-control mechanism for biologic DMARDs, placing a substantial financial burden on patients who require such drugs for adequate control of their RA symptoms,” the investigators wrote.
Dr. Yazdany and her associates reported that during the period before spending limits were reached and catastrophic coverage was triggered, mean patient cost-sharing for biologic DMARDs was $835 per month. Infliximab was the least expensive option at $269 per month, while anakinra cost patients a mean $2,993 per month. Medicare Advantage plans were more likely to cover biologic agents, but at a higher average patient cost of $862 per month.
Of the nine nonbiologic DMARDs, six were covered by almost all plans with copayments of $5-$10 per month; the least expensive option in each plan averaged $48 per year in out-of-pocket costs. Prior authorizations were not needed for nonbiologic agents.
Dr. Yazdany and colleagues raised the question of whether the policies of prior authorizations, tiered coverage, and high levels of patient cost-sharing contribute to achieving high value care for those with RA who fail first-line therapy. Evidence-based guidelines would move these patients to biologic therapy, but the high level of patient cost-sharing in the current Medicare payment structure has previously been shown to reduce adherence, potentially speeding disease progression and worsening outcomes.
“We have avoided the issue of cost-effectiveness analysis,” Dr. Yazdany said, noting that to date, Medicare has not used reference pricing or negotiated drug costs, in contrast to government payers in many other countries. “We should all work to decrease out-of-pocket costs for people grappling with this serious disease,” she said. “The current situation is really unacceptable. That’s why we need to talk about it.”
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point: Patients with rheumatoid arthritis face significant cost sharing for biologic disease modifying agents under Medicare Part D.
Major findings: Mean out-of-pocket costs for Medicare Part D recipients receiving these treatments were over $2,700 per year before catastrophic coverage began paying 95% of charges.
Data source: Cross-sectional analysis of 2,737 Medicare Part D formularies drawn from the Centers for Medicare & Medicaid Services’ database, including a comparison of Medicare Advantage to stand-alone Medicare Part D plans and a comparison of biologic and nonbiologic disease modifying drugs.
Disclosures: The Agency for Healthcare Research and Quality and the Hawaii Medical Service Association Endowed Chair in Health Services and Quality Research funded the study. The authors reported no conflicts of interest.
Mast cells predict hypersensitive reactions in rituximab desensitization
HOUSTON – For rituximab patients undergoing desensitization, mast cell degranulation is a more effective predictor of hypersensitive reactions (HSR) than are the traditionally employed skin tests, according to a study presented at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
“In this particular study, we were interested in investigating the roles of immunoglobulin E (IgE) in the mast cells in rituximab hypersensitivity,” said Dr. Johnson T. Wong of Massachusetts General Hospital, Boston, who presented the findings during a session dedicated to “Novel Insights in Drug Allergy.”
The study comprised 25 patients with persistent or severe rituximab sensitivity (RITS) who were treated at Massachusetts General between 2008 and 2013. Dr. Wong and his coinvestigators performed skin testing on 18 subjects, of which 7 (39%) reacted positively at least once during the course of testing.
HSR reactions occurred at similar rates in subjects who reacted positively and negatively to skin testing. Tryptase levels were taken in 18 subjects (72%), during 67% of the HSR desensitizations and 21% of the asymptomatic desensitizations.
Overall, elevated levels were noted in 21% of the HSR desensitizations.
The elevated tryptase levels in patients experiencing HSR indicates that mast cell degranulation is likely a major contributing factor in causing HSR, with cytokine release and tumor lysis also playing an important role, Dr. Wong said. Mast cell degranulations also points to an association with IgE mechanisms as a reliable indicator of HSR likelihood.
Dr. Wong and his coauthors concluded via tryptase level testing that asymptomatic mast cell degranulation happened very rarely, as only 1 out of 27 assessments (3%) indicated that result. One patient was excluded from the HSR desensitization cohort’s results because of probable mast cell activation syndrome, which caused abnormally elevated tryptase levels throughout the study period.
“In both positive and negative skin test patients, our desensitization protocol was able to reduce the reaction rate to less than 30%, so around 70% of patients had no reaction at all during desensitization,” said Dr. Wong, adding that nearly all desensitizations were completed successfully.
Dr. Wong had no disclosures.
HOUSTON – For rituximab patients undergoing desensitization, mast cell degranulation is a more effective predictor of hypersensitive reactions (HSR) than are the traditionally employed skin tests, according to a study presented at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
“In this particular study, we were interested in investigating the roles of immunoglobulin E (IgE) in the mast cells in rituximab hypersensitivity,” said Dr. Johnson T. Wong of Massachusetts General Hospital, Boston, who presented the findings during a session dedicated to “Novel Insights in Drug Allergy.”
The study comprised 25 patients with persistent or severe rituximab sensitivity (RITS) who were treated at Massachusetts General between 2008 and 2013. Dr. Wong and his coinvestigators performed skin testing on 18 subjects, of which 7 (39%) reacted positively at least once during the course of testing.
HSR reactions occurred at similar rates in subjects who reacted positively and negatively to skin testing. Tryptase levels were taken in 18 subjects (72%), during 67% of the HSR desensitizations and 21% of the asymptomatic desensitizations.
Overall, elevated levels were noted in 21% of the HSR desensitizations.
The elevated tryptase levels in patients experiencing HSR indicates that mast cell degranulation is likely a major contributing factor in causing HSR, with cytokine release and tumor lysis also playing an important role, Dr. Wong said. Mast cell degranulations also points to an association with IgE mechanisms as a reliable indicator of HSR likelihood.
Dr. Wong and his coauthors concluded via tryptase level testing that asymptomatic mast cell degranulation happened very rarely, as only 1 out of 27 assessments (3%) indicated that result. One patient was excluded from the HSR desensitization cohort’s results because of probable mast cell activation syndrome, which caused abnormally elevated tryptase levels throughout the study period.
“In both positive and negative skin test patients, our desensitization protocol was able to reduce the reaction rate to less than 30%, so around 70% of patients had no reaction at all during desensitization,” said Dr. Wong, adding that nearly all desensitizations were completed successfully.
Dr. Wong had no disclosures.
HOUSTON – For rituximab patients undergoing desensitization, mast cell degranulation is a more effective predictor of hypersensitive reactions (HSR) than are the traditionally employed skin tests, according to a study presented at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
“In this particular study, we were interested in investigating the roles of immunoglobulin E (IgE) in the mast cells in rituximab hypersensitivity,” said Dr. Johnson T. Wong of Massachusetts General Hospital, Boston, who presented the findings during a session dedicated to “Novel Insights in Drug Allergy.”
The study comprised 25 patients with persistent or severe rituximab sensitivity (RITS) who were treated at Massachusetts General between 2008 and 2013. Dr. Wong and his coinvestigators performed skin testing on 18 subjects, of which 7 (39%) reacted positively at least once during the course of testing.
HSR reactions occurred at similar rates in subjects who reacted positively and negatively to skin testing. Tryptase levels were taken in 18 subjects (72%), during 67% of the HSR desensitizations and 21% of the asymptomatic desensitizations.
Overall, elevated levels were noted in 21% of the HSR desensitizations.
The elevated tryptase levels in patients experiencing HSR indicates that mast cell degranulation is likely a major contributing factor in causing HSR, with cytokine release and tumor lysis also playing an important role, Dr. Wong said. Mast cell degranulations also points to an association with IgE mechanisms as a reliable indicator of HSR likelihood.
Dr. Wong and his coauthors concluded via tryptase level testing that asymptomatic mast cell degranulation happened very rarely, as only 1 out of 27 assessments (3%) indicated that result. One patient was excluded from the HSR desensitization cohort’s results because of probable mast cell activation syndrome, which caused abnormally elevated tryptase levels throughout the study period.
“In both positive and negative skin test patients, our desensitization protocol was able to reduce the reaction rate to less than 30%, so around 70% of patients had no reaction at all during desensitization,” said Dr. Wong, adding that nearly all desensitizations were completed successfully.
Dr. Wong had no disclosures.
AT THE 2015 AAAAI ANNUAL MEETING
Key clinical point: Look to mast cell degranulation, not skin tests, for accurate information regarding the likelihood of hypersensitive reactions in rituximab patients undergoing desensitization.
Major finding: Tryptase levels were elevated in 21% of patients who experienced hypersensitive reactions during desensitization.
Data source: Study of 25 patients with rituximab sensitivity at Massachusetts General Hospital.
Disclosures: Dr. Wong reported no relevant financial disclosures.
Calcium supplements not linked to coronary artery calcification in RA patients
For patients with rheumatoid arthritis taking oral calcium supplements, higher levels of calcium in the bloodstream were not associated with an increased risk of coronary atherosclerosis, according to a newly published study in Arthritis and Rheumatology.
Dr. Laura Geraldino-Pardilla of Columbia University, New York, and her associates reviewed data from 145 RA patients without known CVD from a prospective cohort study, and used a multidetector CT scan to assess the patients’ levels of coronary artery calcium (CAC), a measure of coronary atherosclerosis, at baseline and through regular follow-ups.
They found baseline CAC scores greater than 100 units were significantly less frequent in the higher-dose supplemental calcium group (at least 1,000 mg/day) than in the lower-dose group (less than 1,000mg/day) (OR 0.28, 95% CI 0.11-0.74), even after adjustment for relevant confounders (OR 0.30, 95% CI 0.09-0.93).
Read the entire article online at Arthritis & Rheumatology 2015 (doi: 10.1002/art.39100).
For patients with rheumatoid arthritis taking oral calcium supplements, higher levels of calcium in the bloodstream were not associated with an increased risk of coronary atherosclerosis, according to a newly published study in Arthritis and Rheumatology.
Dr. Laura Geraldino-Pardilla of Columbia University, New York, and her associates reviewed data from 145 RA patients without known CVD from a prospective cohort study, and used a multidetector CT scan to assess the patients’ levels of coronary artery calcium (CAC), a measure of coronary atherosclerosis, at baseline and through regular follow-ups.
They found baseline CAC scores greater than 100 units were significantly less frequent in the higher-dose supplemental calcium group (at least 1,000 mg/day) than in the lower-dose group (less than 1,000mg/day) (OR 0.28, 95% CI 0.11-0.74), even after adjustment for relevant confounders (OR 0.30, 95% CI 0.09-0.93).
Read the entire article online at Arthritis & Rheumatology 2015 (doi: 10.1002/art.39100).
For patients with rheumatoid arthritis taking oral calcium supplements, higher levels of calcium in the bloodstream were not associated with an increased risk of coronary atherosclerosis, according to a newly published study in Arthritis and Rheumatology.
Dr. Laura Geraldino-Pardilla of Columbia University, New York, and her associates reviewed data from 145 RA patients without known CVD from a prospective cohort study, and used a multidetector CT scan to assess the patients’ levels of coronary artery calcium (CAC), a measure of coronary atherosclerosis, at baseline and through regular follow-ups.
They found baseline CAC scores greater than 100 units were significantly less frequent in the higher-dose supplemental calcium group (at least 1,000 mg/day) than in the lower-dose group (less than 1,000mg/day) (OR 0.28, 95% CI 0.11-0.74), even after adjustment for relevant confounders (OR 0.30, 95% CI 0.09-0.93).
Read the entire article online at Arthritis & Rheumatology 2015 (doi: 10.1002/art.39100).
CXCL4 and CXCL7 expression increases in early stages of RA
Expression of the CXCL4 and CXCL7 chemokines seems to be increased in the early stages of rheumatoid arthritis, according to Dr. Lorraine Yeo of the University of Birmingham, England, and her associates.
Compared to patients in the non-inflamed control group, those with early RA had significantly higher mRNA levels of the CXCL4 and CXCL7 chemokines. Protein expression of the two chemokines was also much higher in patients with early RA compared to those whose arthritis was resolving or had long established RA. Significantly elevated extravascular CXCL7 levels were also present in early RA patients.
“It will be important to investigate whether the production of CXCL4 and CXCL7 observed in the synovium in early RA is reflected by elevated levels in plasma samples. Future use of these chemokines as biomarkers for prediction of progression to RA will depend on replication in other independent cohorts,” the investigators concluded.Find the full study in Annals of the Rheumatic Diseases (doi:10.1136/annrheumdis-2014-206921).
Expression of the CXCL4 and CXCL7 chemokines seems to be increased in the early stages of rheumatoid arthritis, according to Dr. Lorraine Yeo of the University of Birmingham, England, and her associates.
Compared to patients in the non-inflamed control group, those with early RA had significantly higher mRNA levels of the CXCL4 and CXCL7 chemokines. Protein expression of the two chemokines was also much higher in patients with early RA compared to those whose arthritis was resolving or had long established RA. Significantly elevated extravascular CXCL7 levels were also present in early RA patients.
“It will be important to investigate whether the production of CXCL4 and CXCL7 observed in the synovium in early RA is reflected by elevated levels in plasma samples. Future use of these chemokines as biomarkers for prediction of progression to RA will depend on replication in other independent cohorts,” the investigators concluded.Find the full study in Annals of the Rheumatic Diseases (doi:10.1136/annrheumdis-2014-206921).
Expression of the CXCL4 and CXCL7 chemokines seems to be increased in the early stages of rheumatoid arthritis, according to Dr. Lorraine Yeo of the University of Birmingham, England, and her associates.
Compared to patients in the non-inflamed control group, those with early RA had significantly higher mRNA levels of the CXCL4 and CXCL7 chemokines. Protein expression of the two chemokines was also much higher in patients with early RA compared to those whose arthritis was resolving or had long established RA. Significantly elevated extravascular CXCL7 levels were also present in early RA patients.
“It will be important to investigate whether the production of CXCL4 and CXCL7 observed in the synovium in early RA is reflected by elevated levels in plasma samples. Future use of these chemokines as biomarkers for prediction of progression to RA will depend on replication in other independent cohorts,” the investigators concluded.Find the full study in Annals of the Rheumatic Diseases (doi:10.1136/annrheumdis-2014-206921).
Rheumatologists support biosimilars, with caveats
Biosimilars are poised to flood the U.S. pharmaceutical market, bringing biologic therapies within reach of many patients who previously could not pay for them. In a position statement issued last month, the American College of Rheumatology voiced its support for the new agents, but only if they are carefully regulated and tracked – and if prescribing decisions are left up to physicians and patients, not pharmacists or payers.
“As more biologic drugs are used to treat rheumatic diseases, rheumatologists are increasingly concerned about their high cost and patients being unable to afford them,” ACR president William St.Clair said in a written statement. “We agree that less-expensive biologic therapies are needed, and recognize that biosimilars provide an opportunity to reduce treatment costs.”
The ACR position statement follows the U.S. Food and Drug Administration’s approval on March 6 of the first biosimilar, Sandoz’s Zarxio (filgrastim-sndz), which is used in cancer treatment and is based on Amgen’s Neupogen (filgrastim). Patents on many other biologics will expire by about 2020, clearing the way for biosimilars used in rheumatology, dermatology, and other fields. Among these, Celltrion’s infliximab biosimilar Remsima (CT-P13) was submitted for FDA approval last year for the treatment of rheumatoid arthritis, Crohn’s disease, and several other conditions. The FDA postponed a March 17 meeting to review Celltrion’s application, stating that it had requested more information from the company.
The ACR has echoed the FDA’s apparent caution regarding biosimilars. Although only the clinically inactive components of biosimilars are allowed to differ from their reference agents, today’s biologics are so molecularly complex that biosimilars need rigorous studies in humans to assess their safety and efficacy, the organization stated. “It is uncertain whether patients will respond to these drugs the same way they would to an original biologic, because biologics are very sensitive to manufacturing changes,” Dr. St.Clair said. “Even minor differences in a biosimilar’s molecular structure, purity, or other chemical properties could change the way a patient responds to the drug.”
For this reason, postmarketing studies in children and adults also will be essential, the ACR stated. Dr. Scott Zashin, a rheumatologist at Presbyterian Hospital, Dallas, and who is also with the University of Texas in Dallas, agreed. “Studies will need to be conducted to determine the benefit and risks,” he said in an interview. “In addition, registries would be needed to help assess long-term safety in larger numbers of patients than is feasible in a clinical trial.”
The ACR also raised concerns about switching patients back and forth between originator biologics and biosimilars, even if the biosimilar met stringent criteria demonstrating its interchangeability with the reference agent. “Experience outside the United States with biosimilar erythropoietin has indicated that a serious adverse reaction due to immunogenicity is a valid and very real concern for biosimilar products,” the ACR stated. “At this time, there are too many unknown variables to presume that repeated switching of biologic drugs would be safe practice.”
Because the FDA has not specified what would be needed to prove that a biologic and its biosimilar are truly interchangeable in the treatment of rheumatic conditions, pharmacists should not be allowed to substitute a biosimilar for a prescribed biologic without written approval from the physician, the ACR stated. But if pharmacists are legally allowed to substitute a biosimilar, they should be required to notify both physicians and patients so that patients can be monitored for adverse effects, the organization added.
Dr. Zashin agreed. “I would pretty much treat the biosimilars as generics, and leave it up to the FDA to decide if they are inferior,” he said. “But there are some generics on the market now (small molecules, not biologics) that I feel are inferior to the brand drug. This would be no different if biosimilars are approved.” For that reason, he would not accept a pharmacist substituting a biosimilar if he prescribed the branded drug, unless the patient preferred and specifically requested the less-expensive product, he said.
Along the same lines, the ACR believes that physicians should be able to designate a prescription as “brand only” if they are concerned about the safety of a biosimilar and that payers should not be able to force patients to switch to a biosimilar if they are already responding well to the biologic. But under current circumstances, “the insurance company would dictate what the patient could receive,” Dr. Zashin cautioned. If the patient refused the biosimilar, “I expect they would penalize the patient and make the brand quite costly.”
Finally, the ACR position statement recommended that biosimilars have distinctive names to easily distinguish them from their reference agents. Otherwise, name confusion could lead to prescribing errors and mistakes during surveillance for adverse events, the organization said.
“We will continue to follow the various issues surrounding the distribution, monitoring, and reimbursement of biosimilars very closely as state and federal policies are proposed that affect our patients and the rheumatologists who serve them,” Dr. St.Clair concluded. “Ensuring patients have easy access to affordable treatment options and rheumatology care continues to be a high priority for us.”
Biosimilars are poised to flood the U.S. pharmaceutical market, bringing biologic therapies within reach of many patients who previously could not pay for them. In a position statement issued last month, the American College of Rheumatology voiced its support for the new agents, but only if they are carefully regulated and tracked – and if prescribing decisions are left up to physicians and patients, not pharmacists or payers.
“As more biologic drugs are used to treat rheumatic diseases, rheumatologists are increasingly concerned about their high cost and patients being unable to afford them,” ACR president William St.Clair said in a written statement. “We agree that less-expensive biologic therapies are needed, and recognize that biosimilars provide an opportunity to reduce treatment costs.”
The ACR position statement follows the U.S. Food and Drug Administration’s approval on March 6 of the first biosimilar, Sandoz’s Zarxio (filgrastim-sndz), which is used in cancer treatment and is based on Amgen’s Neupogen (filgrastim). Patents on many other biologics will expire by about 2020, clearing the way for biosimilars used in rheumatology, dermatology, and other fields. Among these, Celltrion’s infliximab biosimilar Remsima (CT-P13) was submitted for FDA approval last year for the treatment of rheumatoid arthritis, Crohn’s disease, and several other conditions. The FDA postponed a March 17 meeting to review Celltrion’s application, stating that it had requested more information from the company.
The ACR has echoed the FDA’s apparent caution regarding biosimilars. Although only the clinically inactive components of biosimilars are allowed to differ from their reference agents, today’s biologics are so molecularly complex that biosimilars need rigorous studies in humans to assess their safety and efficacy, the organization stated. “It is uncertain whether patients will respond to these drugs the same way they would to an original biologic, because biologics are very sensitive to manufacturing changes,” Dr. St.Clair said. “Even minor differences in a biosimilar’s molecular structure, purity, or other chemical properties could change the way a patient responds to the drug.”
For this reason, postmarketing studies in children and adults also will be essential, the ACR stated. Dr. Scott Zashin, a rheumatologist at Presbyterian Hospital, Dallas, and who is also with the University of Texas in Dallas, agreed. “Studies will need to be conducted to determine the benefit and risks,” he said in an interview. “In addition, registries would be needed to help assess long-term safety in larger numbers of patients than is feasible in a clinical trial.”
The ACR also raised concerns about switching patients back and forth between originator biologics and biosimilars, even if the biosimilar met stringent criteria demonstrating its interchangeability with the reference agent. “Experience outside the United States with biosimilar erythropoietin has indicated that a serious adverse reaction due to immunogenicity is a valid and very real concern for biosimilar products,” the ACR stated. “At this time, there are too many unknown variables to presume that repeated switching of biologic drugs would be safe practice.”
Because the FDA has not specified what would be needed to prove that a biologic and its biosimilar are truly interchangeable in the treatment of rheumatic conditions, pharmacists should not be allowed to substitute a biosimilar for a prescribed biologic without written approval from the physician, the ACR stated. But if pharmacists are legally allowed to substitute a biosimilar, they should be required to notify both physicians and patients so that patients can be monitored for adverse effects, the organization added.
Dr. Zashin agreed. “I would pretty much treat the biosimilars as generics, and leave it up to the FDA to decide if they are inferior,” he said. “But there are some generics on the market now (small molecules, not biologics) that I feel are inferior to the brand drug. This would be no different if biosimilars are approved.” For that reason, he would not accept a pharmacist substituting a biosimilar if he prescribed the branded drug, unless the patient preferred and specifically requested the less-expensive product, he said.
Along the same lines, the ACR believes that physicians should be able to designate a prescription as “brand only” if they are concerned about the safety of a biosimilar and that payers should not be able to force patients to switch to a biosimilar if they are already responding well to the biologic. But under current circumstances, “the insurance company would dictate what the patient could receive,” Dr. Zashin cautioned. If the patient refused the biosimilar, “I expect they would penalize the patient and make the brand quite costly.”
Finally, the ACR position statement recommended that biosimilars have distinctive names to easily distinguish them from their reference agents. Otherwise, name confusion could lead to prescribing errors and mistakes during surveillance for adverse events, the organization said.
“We will continue to follow the various issues surrounding the distribution, monitoring, and reimbursement of biosimilars very closely as state and federal policies are proposed that affect our patients and the rheumatologists who serve them,” Dr. St.Clair concluded. “Ensuring patients have easy access to affordable treatment options and rheumatology care continues to be a high priority for us.”
Biosimilars are poised to flood the U.S. pharmaceutical market, bringing biologic therapies within reach of many patients who previously could not pay for them. In a position statement issued last month, the American College of Rheumatology voiced its support for the new agents, but only if they are carefully regulated and tracked – and if prescribing decisions are left up to physicians and patients, not pharmacists or payers.
“As more biologic drugs are used to treat rheumatic diseases, rheumatologists are increasingly concerned about their high cost and patients being unable to afford them,” ACR president William St.Clair said in a written statement. “We agree that less-expensive biologic therapies are needed, and recognize that biosimilars provide an opportunity to reduce treatment costs.”
The ACR position statement follows the U.S. Food and Drug Administration’s approval on March 6 of the first biosimilar, Sandoz’s Zarxio (filgrastim-sndz), which is used in cancer treatment and is based on Amgen’s Neupogen (filgrastim). Patents on many other biologics will expire by about 2020, clearing the way for biosimilars used in rheumatology, dermatology, and other fields. Among these, Celltrion’s infliximab biosimilar Remsima (CT-P13) was submitted for FDA approval last year for the treatment of rheumatoid arthritis, Crohn’s disease, and several other conditions. The FDA postponed a March 17 meeting to review Celltrion’s application, stating that it had requested more information from the company.
The ACR has echoed the FDA’s apparent caution regarding biosimilars. Although only the clinically inactive components of biosimilars are allowed to differ from their reference agents, today’s biologics are so molecularly complex that biosimilars need rigorous studies in humans to assess their safety and efficacy, the organization stated. “It is uncertain whether patients will respond to these drugs the same way they would to an original biologic, because biologics are very sensitive to manufacturing changes,” Dr. St.Clair said. “Even minor differences in a biosimilar’s molecular structure, purity, or other chemical properties could change the way a patient responds to the drug.”
For this reason, postmarketing studies in children and adults also will be essential, the ACR stated. Dr. Scott Zashin, a rheumatologist at Presbyterian Hospital, Dallas, and who is also with the University of Texas in Dallas, agreed. “Studies will need to be conducted to determine the benefit and risks,” he said in an interview. “In addition, registries would be needed to help assess long-term safety in larger numbers of patients than is feasible in a clinical trial.”
The ACR also raised concerns about switching patients back and forth between originator biologics and biosimilars, even if the biosimilar met stringent criteria demonstrating its interchangeability with the reference agent. “Experience outside the United States with biosimilar erythropoietin has indicated that a serious adverse reaction due to immunogenicity is a valid and very real concern for biosimilar products,” the ACR stated. “At this time, there are too many unknown variables to presume that repeated switching of biologic drugs would be safe practice.”
Because the FDA has not specified what would be needed to prove that a biologic and its biosimilar are truly interchangeable in the treatment of rheumatic conditions, pharmacists should not be allowed to substitute a biosimilar for a prescribed biologic without written approval from the physician, the ACR stated. But if pharmacists are legally allowed to substitute a biosimilar, they should be required to notify both physicians and patients so that patients can be monitored for adverse effects, the organization added.
Dr. Zashin agreed. “I would pretty much treat the biosimilars as generics, and leave it up to the FDA to decide if they are inferior,” he said. “But there are some generics on the market now (small molecules, not biologics) that I feel are inferior to the brand drug. This would be no different if biosimilars are approved.” For that reason, he would not accept a pharmacist substituting a biosimilar if he prescribed the branded drug, unless the patient preferred and specifically requested the less-expensive product, he said.
Along the same lines, the ACR believes that physicians should be able to designate a prescription as “brand only” if they are concerned about the safety of a biosimilar and that payers should not be able to force patients to switch to a biosimilar if they are already responding well to the biologic. But under current circumstances, “the insurance company would dictate what the patient could receive,” Dr. Zashin cautioned. If the patient refused the biosimilar, “I expect they would penalize the patient and make the brand quite costly.”
Finally, the ACR position statement recommended that biosimilars have distinctive names to easily distinguish them from their reference agents. Otherwise, name confusion could lead to prescribing errors and mistakes during surveillance for adverse events, the organization said.
“We will continue to follow the various issues surrounding the distribution, monitoring, and reimbursement of biosimilars very closely as state and federal policies are proposed that affect our patients and the rheumatologists who serve them,” Dr. St.Clair concluded. “Ensuring patients have easy access to affordable treatment options and rheumatology care continues to be a high priority for us.”
Home-administered biologics for RA more common with Medicare Part D subsidies
For Medicare beneficiaries with rheumatoid arthritis, out-of-pocket costs are closely linked to whether a patient is more likely to receive facility- or home-administered biologic treatment, according to a retrospective analysis of how patient cost sharing affects utilization and spending patterns.
Medicare Part D beneficiaries with rheumatoid arthritis (RA) who receive a low-income subsidy (LIS) are more likely to receive biologic medication that can be self-injected at home, the study found, while those without subsidies face greater out-of-pocket costs for outpatient medications and are more likely to receive facility-administered drugs. Lead study author Dr. Jinoos Yazdany noted, “Our findings shed light on the complexity of current Medicare drug coverage policies for biologic DMARDs.”
Dr. Yazdany and her associates at the University of California, San Francisco, used 2009 Medicare claims to obtain a random nationwide sample of 5% of all Medicare beneficiaries with RA who were dispensed at least one RA medication. Of the 6,932 beneficiaries identified, 1,812 (26.1%) were dispensed a biologic disease-modifying antirheumatic drug (DMARD). Utilization patterns of the remainder, who received a nonbiologic DMARD, were used for comparison. In 2009, the biologic DMARDs infliximab, abatacept, and rituximab were all facility-administered medications reimbursable under Medicare Part B, while etanercept, adalimumab, and anakinra were available for home self-injection and were therefore Part D drugs (Arthritis Care Res. 2015 March 16 [doi:10.1002/acr.22580]).
Examining biologic DMARD use and costs, the study calculated costs to Medicare for both home- and facility-administered medications. Part D dispensing data were used to calculate costs to patients by determining the actual out-of-pocket dollar costs for beneficiaries. This information was not available for Part B events, so investigators used a range from 0%-20% cost-sharing to estimate patient costs.
Patient costs for Medicare Part D were further divided by LIS status, since LIS confers minimal cost sharing for outpatient drugs. By contrast, nonsubsidized Part D recipients incur variable costs; the current Medicare Part D coverage gap – also known as the doughnut hole – means that up to 100% of the cost of medications may be borne by the patient at certain points in the coverage cycle.
Multinomial regression analysis showed that those receiving a facility-administered biologic DMARD under Part B were significantly less likely than those receiving nonbiologic DMARDs to have a LIS (relative risk, 0.58; 95% confidence interval, 0.48-0.69). Recipients of self-injected DMARDs under Part D, in contrast, were much more likely to have a LIS than were those in the nonbiologic DMARD group (RR, 2.98; 95% CI, 2.50-3.56). Annual Part D out-of-pocket costs for those receiving biologics with no LIS were a mean $3,751, compared with a mean $72 for the LIS Part D group. However, beneficiaries with no LIS paid a maximum of $2,584 out of pocket for biologic DMARDs through Part B (assuming 20% supplemental insurance coverage).
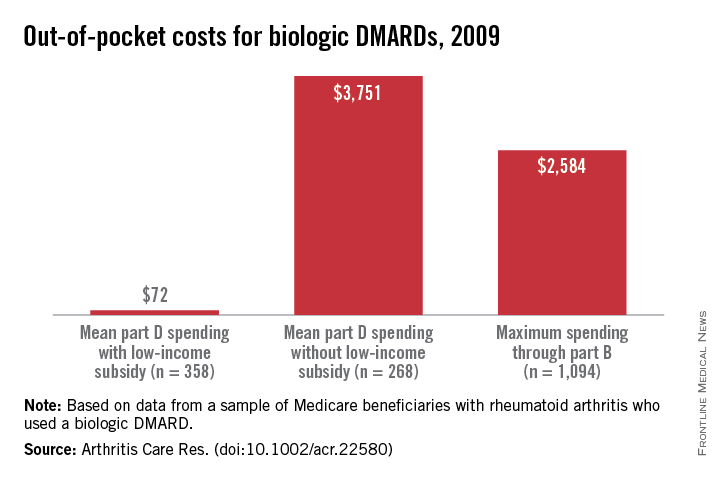
Since the Affordable Care Act will reform Part D to help close the coverage gap, the study also projected how ACA reform, once adopted, would affect cost sharing and use patterns. Dr. Yazdany and her colleagues projected out-of-pocket biologic DMARD costs into full reform implementation in the year 2020, calculating that nonsubsidized out-of-pocket expenses would still be $2,588. The authors noted that “patient cost sharing is an important determinant of drug choice in Medicare’s part B versus part D programs ... the current system risks significant financial burden to many patients.”
When asked to comment on the study, Kaleb Michaud, Ph.D., assistant professor of rheumatology at the University of Nebraska Medical Center, Omaha, and codirector of the National Data Bank for Rheumatic Diseases, noted that this well-constructed study made very good use of Medicare data to point to a fundamental problem with reimbursement for rheumatic disease medications. “Rather than asking, ‘What’s the best drug for this patient?’ we find ourselves asking, ‘What drug can this patient afford?’ ” he said.
The study was funded by the National Institutes of Health, a Research Allocation and Evaluation Award from the University of California, San Francisco, and the Rosaline Russell Medical Research Center for Arthritis. The authors reported that they had no conflicts of interest.
For Medicare beneficiaries with rheumatoid arthritis, out-of-pocket costs are closely linked to whether a patient is more likely to receive facility- or home-administered biologic treatment, according to a retrospective analysis of how patient cost sharing affects utilization and spending patterns.
Medicare Part D beneficiaries with rheumatoid arthritis (RA) who receive a low-income subsidy (LIS) are more likely to receive biologic medication that can be self-injected at home, the study found, while those without subsidies face greater out-of-pocket costs for outpatient medications and are more likely to receive facility-administered drugs. Lead study author Dr. Jinoos Yazdany noted, “Our findings shed light on the complexity of current Medicare drug coverage policies for biologic DMARDs.”
Dr. Yazdany and her associates at the University of California, San Francisco, used 2009 Medicare claims to obtain a random nationwide sample of 5% of all Medicare beneficiaries with RA who were dispensed at least one RA medication. Of the 6,932 beneficiaries identified, 1,812 (26.1%) were dispensed a biologic disease-modifying antirheumatic drug (DMARD). Utilization patterns of the remainder, who received a nonbiologic DMARD, were used for comparison. In 2009, the biologic DMARDs infliximab, abatacept, and rituximab were all facility-administered medications reimbursable under Medicare Part B, while etanercept, adalimumab, and anakinra were available for home self-injection and were therefore Part D drugs (Arthritis Care Res. 2015 March 16 [doi:10.1002/acr.22580]).
Examining biologic DMARD use and costs, the study calculated costs to Medicare for both home- and facility-administered medications. Part D dispensing data were used to calculate costs to patients by determining the actual out-of-pocket dollar costs for beneficiaries. This information was not available for Part B events, so investigators used a range from 0%-20% cost-sharing to estimate patient costs.
Patient costs for Medicare Part D were further divided by LIS status, since LIS confers minimal cost sharing for outpatient drugs. By contrast, nonsubsidized Part D recipients incur variable costs; the current Medicare Part D coverage gap – also known as the doughnut hole – means that up to 100% of the cost of medications may be borne by the patient at certain points in the coverage cycle.
Multinomial regression analysis showed that those receiving a facility-administered biologic DMARD under Part B were significantly less likely than those receiving nonbiologic DMARDs to have a LIS (relative risk, 0.58; 95% confidence interval, 0.48-0.69). Recipients of self-injected DMARDs under Part D, in contrast, were much more likely to have a LIS than were those in the nonbiologic DMARD group (RR, 2.98; 95% CI, 2.50-3.56). Annual Part D out-of-pocket costs for those receiving biologics with no LIS were a mean $3,751, compared with a mean $72 for the LIS Part D group. However, beneficiaries with no LIS paid a maximum of $2,584 out of pocket for biologic DMARDs through Part B (assuming 20% supplemental insurance coverage).

Since the Affordable Care Act will reform Part D to help close the coverage gap, the study also projected how ACA reform, once adopted, would affect cost sharing and use patterns. Dr. Yazdany and her colleagues projected out-of-pocket biologic DMARD costs into full reform implementation in the year 2020, calculating that nonsubsidized out-of-pocket expenses would still be $2,588. The authors noted that “patient cost sharing is an important determinant of drug choice in Medicare’s part B versus part D programs ... the current system risks significant financial burden to many patients.”
When asked to comment on the study, Kaleb Michaud, Ph.D., assistant professor of rheumatology at the University of Nebraska Medical Center, Omaha, and codirector of the National Data Bank for Rheumatic Diseases, noted that this well-constructed study made very good use of Medicare data to point to a fundamental problem with reimbursement for rheumatic disease medications. “Rather than asking, ‘What’s the best drug for this patient?’ we find ourselves asking, ‘What drug can this patient afford?’ ” he said.
The study was funded by the National Institutes of Health, a Research Allocation and Evaluation Award from the University of California, San Francisco, and the Rosaline Russell Medical Research Center for Arthritis. The authors reported that they had no conflicts of interest.
For Medicare beneficiaries with rheumatoid arthritis, out-of-pocket costs are closely linked to whether a patient is more likely to receive facility- or home-administered biologic treatment, according to a retrospective analysis of how patient cost sharing affects utilization and spending patterns.
Medicare Part D beneficiaries with rheumatoid arthritis (RA) who receive a low-income subsidy (LIS) are more likely to receive biologic medication that can be self-injected at home, the study found, while those without subsidies face greater out-of-pocket costs for outpatient medications and are more likely to receive facility-administered drugs. Lead study author Dr. Jinoos Yazdany noted, “Our findings shed light on the complexity of current Medicare drug coverage policies for biologic DMARDs.”
Dr. Yazdany and her associates at the University of California, San Francisco, used 2009 Medicare claims to obtain a random nationwide sample of 5% of all Medicare beneficiaries with RA who were dispensed at least one RA medication. Of the 6,932 beneficiaries identified, 1,812 (26.1%) were dispensed a biologic disease-modifying antirheumatic drug (DMARD). Utilization patterns of the remainder, who received a nonbiologic DMARD, were used for comparison. In 2009, the biologic DMARDs infliximab, abatacept, and rituximab were all facility-administered medications reimbursable under Medicare Part B, while etanercept, adalimumab, and anakinra were available for home self-injection and were therefore Part D drugs (Arthritis Care Res. 2015 March 16 [doi:10.1002/acr.22580]).
Examining biologic DMARD use and costs, the study calculated costs to Medicare for both home- and facility-administered medications. Part D dispensing data were used to calculate costs to patients by determining the actual out-of-pocket dollar costs for beneficiaries. This information was not available for Part B events, so investigators used a range from 0%-20% cost-sharing to estimate patient costs.
Patient costs for Medicare Part D were further divided by LIS status, since LIS confers minimal cost sharing for outpatient drugs. By contrast, nonsubsidized Part D recipients incur variable costs; the current Medicare Part D coverage gap – also known as the doughnut hole – means that up to 100% of the cost of medications may be borne by the patient at certain points in the coverage cycle.
Multinomial regression analysis showed that those receiving a facility-administered biologic DMARD under Part B were significantly less likely than those receiving nonbiologic DMARDs to have a LIS (relative risk, 0.58; 95% confidence interval, 0.48-0.69). Recipients of self-injected DMARDs under Part D, in contrast, were much more likely to have a LIS than were those in the nonbiologic DMARD group (RR, 2.98; 95% CI, 2.50-3.56). Annual Part D out-of-pocket costs for those receiving biologics with no LIS were a mean $3,751, compared with a mean $72 for the LIS Part D group. However, beneficiaries with no LIS paid a maximum of $2,584 out of pocket for biologic DMARDs through Part B (assuming 20% supplemental insurance coverage).

Since the Affordable Care Act will reform Part D to help close the coverage gap, the study also projected how ACA reform, once adopted, would affect cost sharing and use patterns. Dr. Yazdany and her colleagues projected out-of-pocket biologic DMARD costs into full reform implementation in the year 2020, calculating that nonsubsidized out-of-pocket expenses would still be $2,588. The authors noted that “patient cost sharing is an important determinant of drug choice in Medicare’s part B versus part D programs ... the current system risks significant financial burden to many patients.”
When asked to comment on the study, Kaleb Michaud, Ph.D., assistant professor of rheumatology at the University of Nebraska Medical Center, Omaha, and codirector of the National Data Bank for Rheumatic Diseases, noted that this well-constructed study made very good use of Medicare data to point to a fundamental problem with reimbursement for rheumatic disease medications. “Rather than asking, ‘What’s the best drug for this patient?’ we find ourselves asking, ‘What drug can this patient afford?’ ” he said.
The study was funded by the National Institutes of Health, a Research Allocation and Evaluation Award from the University of California, San Francisco, and the Rosaline Russell Medical Research Center for Arthritis. The authors reported that they had no conflicts of interest.
FROM ARTHRITIS CARE & RESEARCH
Key clinical point: Medicare beneficiaries who receive Part D low-income subsidies (LIS) are more likely to receive home-administered biologics for rheumatoid arthritis.
Major finding: LIS Medicare beneficiaries were more likely to receive Part D–reimbursable biologics (RR 2.98, 95% CI, 2.50-3.56); out of pocket Part D costs were lower for LIS beneficiaries ($72) than for those not on subsidies ($3,751).
Data source: Retrospective, nationwide analysis of a random 5% sample of 2009 Medicare beneficiaries with RA and receiving biologic medication.
Disclosures: The study was funded by the National Institutes of Health, a Research Allocation and Evaluation Award from the University of California, San Francisco, and the Rosaline Russell Medical Research Center for Arthritis. The authors reported that they had no conflicts of interest.
Treat-to-target approach for RA has cardiovascular benefits
Rheumatoid arthritis patients have a significantly reduced risk of cardiovascular events if their disease is under control, based on an analysis of data from the CORRONA registry.
Investigators led by Dr. Daniel H. Solomon from Brigham and Women’s Hospital in Boston used longitudinal data from almost 25,000 RA patients who were tracked for a median of 2.7 years to find a 21% reduction (95% confidence interval, 13%-29%) in the risk of a composite outcome of the first confirmed myocardial infarction, stroke, or CV death event for each 10-point decline in the Clinical Disease Activity Index and a 53% reduction (95% confidence interval, 30%-68%) from high disease activity to remission.
“These results add significant new information regarding the importance of sustained control of RA disease activity, not only for improvement in pain and function, but also for reduced CV risk,” the authors wrote (Arthritis Rheumatol. 2015 [doi:10.1002/art.39098]).
The findings held true even after researchers adjusted for the use of immunomodulatory treatment, suggesting that controlling disease activity may be a more important management strategy than use of an immunomodulator, at least in the context of preventing CV events, they said.
“This is a hypothesis worth testing especially as enthusiasm grows for use of combination synthetic DMARDs [disease-modifying antirheumatic drugs],” they wrote.
Another clinical implication of the findings was that the adoption of a treat-to-target strategy in RA might be beneficial not only because of the observed improvement in pain and function but also because of a reduction in CV risk, they said.
“While these findings should not be interpreted to mean that traditional risk factors are not important, they do support the current RA recommendations for treating to low disease activity or remission,” they added.
In an accompanying editorial, Dr. Michael T. Nurmohamed of the department of rheumatology at VU University, Amsterdam, agreed with the authors that controlling disease activity (from a CV point of view) counted more than the drug used to achieve the low disease activity state or remission (Arthritis Rheumatol. 2015 [doi:10.1002/art.39096]).
“As inflammation plays a pivotal role in atherosclerotic disease it is not surprising that antirheumatic drugs such as methotrexate, the IL-1 inhibitor canakinumab and the IL-6 inhibitor tocilizumab are presently investigated in several trials as secondary prevention for recurrent CV events in ‘general population’ patients,” he said.
Although the current state-of-art treatment goals for patients with RA appear also to improve the CV risk of patients, it is important to realize there is still no evidence from randomized trials that RA treatment itself reduces CV risk. And despite the fact that rheumatologists know that traditional CV risk factors are important in RA, the management of CV risk factors in daily clinical practice are still poor, he said.
“Another challenge for the next years is to fulfill this unmet need for effective CV risk management implementation strategies,” he concluded.
The researchers declared receiving financial support from various pharmaceutical companies, but none were specifically linked to the current study. The study was supported by CORRONA, which has received subscription fees from a variety of pharmaceutical companies in the past 2 years, but none relating specifically to this study.
Rheumatoid arthritis patients have a significantly reduced risk of cardiovascular events if their disease is under control, based on an analysis of data from the CORRONA registry.
Investigators led by Dr. Daniel H. Solomon from Brigham and Women’s Hospital in Boston used longitudinal data from almost 25,000 RA patients who were tracked for a median of 2.7 years to find a 21% reduction (95% confidence interval, 13%-29%) in the risk of a composite outcome of the first confirmed myocardial infarction, stroke, or CV death event for each 10-point decline in the Clinical Disease Activity Index and a 53% reduction (95% confidence interval, 30%-68%) from high disease activity to remission.
“These results add significant new information regarding the importance of sustained control of RA disease activity, not only for improvement in pain and function, but also for reduced CV risk,” the authors wrote (Arthritis Rheumatol. 2015 [doi:10.1002/art.39098]).
The findings held true even after researchers adjusted for the use of immunomodulatory treatment, suggesting that controlling disease activity may be a more important management strategy than use of an immunomodulator, at least in the context of preventing CV events, they said.
“This is a hypothesis worth testing especially as enthusiasm grows for use of combination synthetic DMARDs [disease-modifying antirheumatic drugs],” they wrote.
Another clinical implication of the findings was that the adoption of a treat-to-target strategy in RA might be beneficial not only because of the observed improvement in pain and function but also because of a reduction in CV risk, they said.
“While these findings should not be interpreted to mean that traditional risk factors are not important, they do support the current RA recommendations for treating to low disease activity or remission,” they added.
In an accompanying editorial, Dr. Michael T. Nurmohamed of the department of rheumatology at VU University, Amsterdam, agreed with the authors that controlling disease activity (from a CV point of view) counted more than the drug used to achieve the low disease activity state or remission (Arthritis Rheumatol. 2015 [doi:10.1002/art.39096]).
“As inflammation plays a pivotal role in atherosclerotic disease it is not surprising that antirheumatic drugs such as methotrexate, the IL-1 inhibitor canakinumab and the IL-6 inhibitor tocilizumab are presently investigated in several trials as secondary prevention for recurrent CV events in ‘general population’ patients,” he said.
Although the current state-of-art treatment goals for patients with RA appear also to improve the CV risk of patients, it is important to realize there is still no evidence from randomized trials that RA treatment itself reduces CV risk. And despite the fact that rheumatologists know that traditional CV risk factors are important in RA, the management of CV risk factors in daily clinical practice are still poor, he said.
“Another challenge for the next years is to fulfill this unmet need for effective CV risk management implementation strategies,” he concluded.
The researchers declared receiving financial support from various pharmaceutical companies, but none were specifically linked to the current study. The study was supported by CORRONA, which has received subscription fees from a variety of pharmaceutical companies in the past 2 years, but none relating specifically to this study.
Rheumatoid arthritis patients have a significantly reduced risk of cardiovascular events if their disease is under control, based on an analysis of data from the CORRONA registry.
Investigators led by Dr. Daniel H. Solomon from Brigham and Women’s Hospital in Boston used longitudinal data from almost 25,000 RA patients who were tracked for a median of 2.7 years to find a 21% reduction (95% confidence interval, 13%-29%) in the risk of a composite outcome of the first confirmed myocardial infarction, stroke, or CV death event for each 10-point decline in the Clinical Disease Activity Index and a 53% reduction (95% confidence interval, 30%-68%) from high disease activity to remission.
“These results add significant new information regarding the importance of sustained control of RA disease activity, not only for improvement in pain and function, but also for reduced CV risk,” the authors wrote (Arthritis Rheumatol. 2015 [doi:10.1002/art.39098]).
The findings held true even after researchers adjusted for the use of immunomodulatory treatment, suggesting that controlling disease activity may be a more important management strategy than use of an immunomodulator, at least in the context of preventing CV events, they said.
“This is a hypothesis worth testing especially as enthusiasm grows for use of combination synthetic DMARDs [disease-modifying antirheumatic drugs],” they wrote.
Another clinical implication of the findings was that the adoption of a treat-to-target strategy in RA might be beneficial not only because of the observed improvement in pain and function but also because of a reduction in CV risk, they said.
“While these findings should not be interpreted to mean that traditional risk factors are not important, they do support the current RA recommendations for treating to low disease activity or remission,” they added.
In an accompanying editorial, Dr. Michael T. Nurmohamed of the department of rheumatology at VU University, Amsterdam, agreed with the authors that controlling disease activity (from a CV point of view) counted more than the drug used to achieve the low disease activity state or remission (Arthritis Rheumatol. 2015 [doi:10.1002/art.39096]).
“As inflammation plays a pivotal role in atherosclerotic disease it is not surprising that antirheumatic drugs such as methotrexate, the IL-1 inhibitor canakinumab and the IL-6 inhibitor tocilizumab are presently investigated in several trials as secondary prevention for recurrent CV events in ‘general population’ patients,” he said.
Although the current state-of-art treatment goals for patients with RA appear also to improve the CV risk of patients, it is important to realize there is still no evidence from randomized trials that RA treatment itself reduces CV risk. And despite the fact that rheumatologists know that traditional CV risk factors are important in RA, the management of CV risk factors in daily clinical practice are still poor, he said.
“Another challenge for the next years is to fulfill this unmet need for effective CV risk management implementation strategies,” he concluded.
The researchers declared receiving financial support from various pharmaceutical companies, but none were specifically linked to the current study. The study was supported by CORRONA, which has received subscription fees from a variety of pharmaceutical companies in the past 2 years, but none relating specifically to this study.
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point: In a large cohort of RA patients, a clear dose-response effect was observed with reductions in disease activity associated with reduced CV risk, independent of immunomodulatory treatments.
Major finding: RA patients had a 21% reduction in the risk of a composite outcome of the first confirmed myocardial infarction, stroke, or CV death event for each 10-point decline in the Clinical Disease Activity Index and a 53% reduction from high disease activity to remission.
Data source: 24,989 patients with RA who were part of the ongoing CORRONA patient registry and were followed for a median of 2.7 years.
Disclosures: The researchers declared receiving financial support from various pharmaceutical companies, but none were specifically linked to the current study. The study was supported by CORRONA, which has received subscription fees from a variety of pharmaceutical companies in the past 2 years, but none relating specifically to this study.
MicroRNAs may prove helpful as a rheumatoid arthritis therapy biomarker
Levels of certain microRNAs were associated with response to treatment with an anti–tumor necrosis factor–alpha agent and conventional disease-modifying antirheumatic drugs in patients with rheumatoid arthritis, according to a study conducted by Dr. Carmen Castro-Villegas and her associates.
In patients who responded to the combination therapy, 91% had overexpressed miRNA, while 9% had downregulated miRNA. Of the 10 miRNA selected for analysis, 6 had been significantly upregulated by the therapy (miR-16-5p, miR-23-3p, miR125b-5p, miR-126-3p, miR-146a-5p, miR-223-3p), and only patients who responded to the therapy showed an increase in these miRNA. The miRNA increase also paralleled a reduction in TNF-alpha, interleukins, rheumatoid factor, and C-reactive protein.
Further analyses showed that miR-23-3p and miR-223-3p can act to predict patients who would not benefit from combination therapy with anti-TNF-alpha agents and conventional disease-modifying antirheumatic drugs or indicate treatment efficacy or the degree of response, the investigators said.
Find the full study in Arthritis Research & Therapy (doi:10.1186/s13075-015-0555-z).
Levels of certain microRNAs were associated with response to treatment with an anti–tumor necrosis factor–alpha agent and conventional disease-modifying antirheumatic drugs in patients with rheumatoid arthritis, according to a study conducted by Dr. Carmen Castro-Villegas and her associates.
In patients who responded to the combination therapy, 91% had overexpressed miRNA, while 9% had downregulated miRNA. Of the 10 miRNA selected for analysis, 6 had been significantly upregulated by the therapy (miR-16-5p, miR-23-3p, miR125b-5p, miR-126-3p, miR-146a-5p, miR-223-3p), and only patients who responded to the therapy showed an increase in these miRNA. The miRNA increase also paralleled a reduction in TNF-alpha, interleukins, rheumatoid factor, and C-reactive protein.
Further analyses showed that miR-23-3p and miR-223-3p can act to predict patients who would not benefit from combination therapy with anti-TNF-alpha agents and conventional disease-modifying antirheumatic drugs or indicate treatment efficacy or the degree of response, the investigators said.
Find the full study in Arthritis Research & Therapy (doi:10.1186/s13075-015-0555-z).
Levels of certain microRNAs were associated with response to treatment with an anti–tumor necrosis factor–alpha agent and conventional disease-modifying antirheumatic drugs in patients with rheumatoid arthritis, according to a study conducted by Dr. Carmen Castro-Villegas and her associates.
In patients who responded to the combination therapy, 91% had overexpressed miRNA, while 9% had downregulated miRNA. Of the 10 miRNA selected for analysis, 6 had been significantly upregulated by the therapy (miR-16-5p, miR-23-3p, miR125b-5p, miR-126-3p, miR-146a-5p, miR-223-3p), and only patients who responded to the therapy showed an increase in these miRNA. The miRNA increase also paralleled a reduction in TNF-alpha, interleukins, rheumatoid factor, and C-reactive protein.
Further analyses showed that miR-23-3p and miR-223-3p can act to predict patients who would not benefit from combination therapy with anti-TNF-alpha agents and conventional disease-modifying antirheumatic drugs or indicate treatment efficacy or the degree of response, the investigators said.
Find the full study in Arthritis Research & Therapy (doi:10.1186/s13075-015-0555-z).
Anti-interleukin 20 antibody effective for active RA
An anti-interleukin 20 recombinant human monoclonal antibody, dubbed NNC0109-0012, was effective in patients with seropositive rheumatoid arthritis who had not responded to methotrexate therapy in a phase IIa, double-blind, placebo-controlled trial. The study comprised 67 patients.
Dr. Ladislav Šenolt and his associates reported notable improvement with NNC0109-0012 treatment within the first week of treatment. During the 12-week trial period, there was an overall improvement of 0.88 points on the 28-joint Disease Activity Score using C-reactive protein (DAS28-CRP) over placebo. The result was driven by the improvement of seropositive RA patients, who had a decline of 1.66 points on DAS28-CRP when compared with placebo. No significant improvement was found in seronegative patients.
Just under 60% of seropositive patients achieved American College of Rheumatology 20 level of response, while 21% of those in the placebo group achieved ACR 20. Just under half of seropositive patients achieved ACR 50, compared with 14% of the placebo group, and 35% of seropositive patients achieved ACR 70, a marker no one in the placebo group reached.
The most common side effects were injection site reactions and infections; no serious infections or side effects associated with NNC0109-0012 were reported, the investigators said.
Find the full study in Arthritis & Rheumatology (doi:10.1002/art.39083).
An anti-interleukin 20 recombinant human monoclonal antibody, dubbed NNC0109-0012, was effective in patients with seropositive rheumatoid arthritis who had not responded to methotrexate therapy in a phase IIa, double-blind, placebo-controlled trial. The study comprised 67 patients.
Dr. Ladislav Šenolt and his associates reported notable improvement with NNC0109-0012 treatment within the first week of treatment. During the 12-week trial period, there was an overall improvement of 0.88 points on the 28-joint Disease Activity Score using C-reactive protein (DAS28-CRP) over placebo. The result was driven by the improvement of seropositive RA patients, who had a decline of 1.66 points on DAS28-CRP when compared with placebo. No significant improvement was found in seronegative patients.
Just under 60% of seropositive patients achieved American College of Rheumatology 20 level of response, while 21% of those in the placebo group achieved ACR 20. Just under half of seropositive patients achieved ACR 50, compared with 14% of the placebo group, and 35% of seropositive patients achieved ACR 70, a marker no one in the placebo group reached.
The most common side effects were injection site reactions and infections; no serious infections or side effects associated with NNC0109-0012 were reported, the investigators said.
Find the full study in Arthritis & Rheumatology (doi:10.1002/art.39083).
An anti-interleukin 20 recombinant human monoclonal antibody, dubbed NNC0109-0012, was effective in patients with seropositive rheumatoid arthritis who had not responded to methotrexate therapy in a phase IIa, double-blind, placebo-controlled trial. The study comprised 67 patients.
Dr. Ladislav Šenolt and his associates reported notable improvement with NNC0109-0012 treatment within the first week of treatment. During the 12-week trial period, there was an overall improvement of 0.88 points on the 28-joint Disease Activity Score using C-reactive protein (DAS28-CRP) over placebo. The result was driven by the improvement of seropositive RA patients, who had a decline of 1.66 points on DAS28-CRP when compared with placebo. No significant improvement was found in seronegative patients.
Just under 60% of seropositive patients achieved American College of Rheumatology 20 level of response, while 21% of those in the placebo group achieved ACR 20. Just under half of seropositive patients achieved ACR 50, compared with 14% of the placebo group, and 35% of seropositive patients achieved ACR 70, a marker no one in the placebo group reached.
The most common side effects were injection site reactions and infections; no serious infections or side effects associated with NNC0109-0012 were reported, the investigators said.
Find the full study in Arthritis & Rheumatology (doi:10.1002/art.39083).









