User login
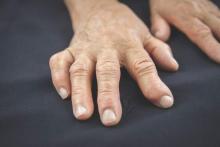
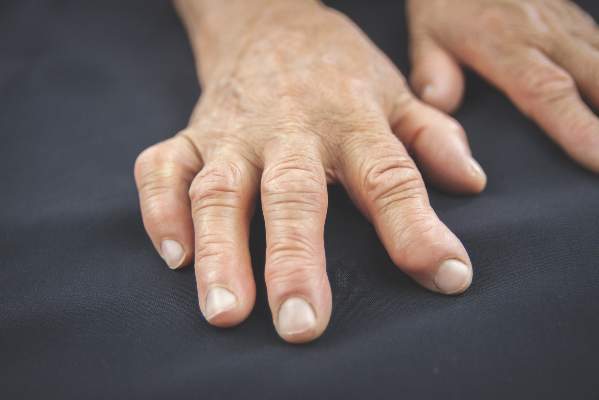
Anti-CarP antibodies linked to worse outcome in early arthritis
MANCHESTER, ENGLAND – Anti–carbamylated protein antibodies were associated with both increased disability and greater disease progression over time in patients with early inflammatory arthritis, data from a 20-year retrospective, observational analysis showed.
According to the research, presented at the British Society for Rheumatology annual conference, patients who were also positive for anti–citrullinated protein antibodies (ACPAs) fared worse long term than those who were ACPA negative.
The results showed that anti–carbamylated protein (anti-CarP) antibodies may provide additional prognostic information to current antibody tests in inflammatory arthritis (IA), but their measurement is not yet ready for general use, said Dr. Jennifer Humphreys of the Arthritis Research UK Centre for Epidemiology at the University of Manchester.
“We know that inflammatory arthritis patients who are rheumatoid factor [RF] or ACPA positive have an increased risk of persistent disease and a poor prognosis,” she said.
“In particular, ACPA[s] are associated with severe radiological progression [and] increased disability and mortality. However, not all patients who lack these antibodies do well,” Dr. Humphreys said.
Research at the University of Leiden (the Netherlands) has found that ACPA-negative patients have anti-CarP antibodies in their sera and that these autoantibodies precede the onset of symptoms and predict the development of rheumatoid arthritis (RA) in patients with arthralgia. These antibodies have also been associated with radiological damage, she said.
Because there were few data on the relationship to disease activity and long-term outcomes in the presence of these antibodies, Dr. Humphreys and her associates decided to look at this using data from the Norfolk Arthritis Register (NOAR), she said. The U.K. team worked in collaboration with the Dutch researchers who performed the anti-CarP tests on serum samples taken from 1,995 patients with early IA enrolled in NOAR from 1990 onward.
At baseline, 1,222 patients met American College of Rheumatology/European League Against Rheumatism 2010 criteria for RA. The mean age of onset was 55 years for both the total population with IA and for those with RA. About two-thirds of the participants were women, and their median symptom duration at enrollment was 33 weeks.
Long-term disability was assessed using the health assessment questionnaire (HAQ), and disease activity was evaluated using the disease activity in 28 joints (DAS28) score. The median baseline DAS28 score was 3.81 for the total cohort and 4.45 for patients with RA, and the median baseline HAQ scores were a respective 0.875 and 1.125.
When the team looked at patient baseline characteristics according to baseline anti-CarP status, they found that the 460 (23%) patients who were positive for these autoantibodies were more likely to have higher baseline DAS28 scores (4.23 vs. 3.73), HAQ scores (1.125 vs. 0.875), and C-reactive protein levels (14 mg/L vs. 7 mg/L) than the 1,543 patients who tested anti-CarP negative. Patients positive for anti-Carp antibodies were also less likely to be female (58% vs 68%) and were slightly older at onset (55 vs. 53 years).
Dr. Humphreys noted that there was a lot of overlap among the presence of anti-CarP, ACPA, and RF. Of the 460 who were anti-CarP positive, 124 were positive only for anti-CarP, 148 were also RF positive, 30 were also ACPA positive, and 158 had all three antibodies.
“Patients who were anti-CarP antibody positive had higher HAQ scores at baseline, and this higher level of disability persisted throughout follow-up, and that association remained significant in the multivariate model,” Dr. Humphreys said.
“When we stratified by ACPA status, there was a statistically significant association in the ACPA-negative subgroup but not in the ACPA-positive subgroup,” she added. However, she noted that the beta coefficients for the association between anti-CarP antibodies and HAQ scores over time were similar (0.12 for the total cohort, 0.14 for the ACPA-negative patients, and 0.09 for the ACPA-positive patients) and that confidence intervals were wide.
Similar findings were seen for the association between anti-CarP antibodies and the DAS28 over time, with respective beta coefficients of 0.23, 01.18, and 0.25 for the total cohort, the ACPA-negative subgroup, and the ACPA-positive subgroup. Again, confidence intervals were wide.
The researchers looked at the additive effect of having anti-CarP and ACPA, and found that while there is no association between RF positivity and long-term disability, there were independent associations with both ACPA positivity and anti-CarP antibody positivity, with respective beta coefficients of –0.05, 0.12, and 0.14.
“And we see the exact same pattern with disease activity over time,” Dr. Humphreys reported.
Thus, measuring anti-CarP antibodies “may be a useful test to do in addition to both RF and ACPA,” she suggested, adding that measuring these antibodies “may be particularly valuable in patients who lack other antibodies.”
NOAR is funded by Arthritis Research UK. Dr. Humphreys had no relevant financial disclosures.
MANCHESTER, ENGLAND – Anti–carbamylated protein antibodies were associated with both increased disability and greater disease progression over time in patients with early inflammatory arthritis, data from a 20-year retrospective, observational analysis showed.
According to the research, presented at the British Society for Rheumatology annual conference, patients who were also positive for anti–citrullinated protein antibodies (ACPAs) fared worse long term than those who were ACPA negative.
The results showed that anti–carbamylated protein (anti-CarP) antibodies may provide additional prognostic information to current antibody tests in inflammatory arthritis (IA), but their measurement is not yet ready for general use, said Dr. Jennifer Humphreys of the Arthritis Research UK Centre for Epidemiology at the University of Manchester.
“We know that inflammatory arthritis patients who are rheumatoid factor [RF] or ACPA positive have an increased risk of persistent disease and a poor prognosis,” she said.
“In particular, ACPA[s] are associated with severe radiological progression [and] increased disability and mortality. However, not all patients who lack these antibodies do well,” Dr. Humphreys said.
Research at the University of Leiden (the Netherlands) has found that ACPA-negative patients have anti-CarP antibodies in their sera and that these autoantibodies precede the onset of symptoms and predict the development of rheumatoid arthritis (RA) in patients with arthralgia. These antibodies have also been associated with radiological damage, she said.
Because there were few data on the relationship to disease activity and long-term outcomes in the presence of these antibodies, Dr. Humphreys and her associates decided to look at this using data from the Norfolk Arthritis Register (NOAR), she said. The U.K. team worked in collaboration with the Dutch researchers who performed the anti-CarP tests on serum samples taken from 1,995 patients with early IA enrolled in NOAR from 1990 onward.
At baseline, 1,222 patients met American College of Rheumatology/European League Against Rheumatism 2010 criteria for RA. The mean age of onset was 55 years for both the total population with IA and for those with RA. About two-thirds of the participants were women, and their median symptom duration at enrollment was 33 weeks.
Long-term disability was assessed using the health assessment questionnaire (HAQ), and disease activity was evaluated using the disease activity in 28 joints (DAS28) score. The median baseline DAS28 score was 3.81 for the total cohort and 4.45 for patients with RA, and the median baseline HAQ scores were a respective 0.875 and 1.125.
When the team looked at patient baseline characteristics according to baseline anti-CarP status, they found that the 460 (23%) patients who were positive for these autoantibodies were more likely to have higher baseline DAS28 scores (4.23 vs. 3.73), HAQ scores (1.125 vs. 0.875), and C-reactive protein levels (14 mg/L vs. 7 mg/L) than the 1,543 patients who tested anti-CarP negative. Patients positive for anti-Carp antibodies were also less likely to be female (58% vs 68%) and were slightly older at onset (55 vs. 53 years).
Dr. Humphreys noted that there was a lot of overlap among the presence of anti-CarP, ACPA, and RF. Of the 460 who were anti-CarP positive, 124 were positive only for anti-CarP, 148 were also RF positive, 30 were also ACPA positive, and 158 had all three antibodies.
“Patients who were anti-CarP antibody positive had higher HAQ scores at baseline, and this higher level of disability persisted throughout follow-up, and that association remained significant in the multivariate model,” Dr. Humphreys said.
“When we stratified by ACPA status, there was a statistically significant association in the ACPA-negative subgroup but not in the ACPA-positive subgroup,” she added. However, she noted that the beta coefficients for the association between anti-CarP antibodies and HAQ scores over time were similar (0.12 for the total cohort, 0.14 for the ACPA-negative patients, and 0.09 for the ACPA-positive patients) and that confidence intervals were wide.
Similar findings were seen for the association between anti-CarP antibodies and the DAS28 over time, with respective beta coefficients of 0.23, 01.18, and 0.25 for the total cohort, the ACPA-negative subgroup, and the ACPA-positive subgroup. Again, confidence intervals were wide.
The researchers looked at the additive effect of having anti-CarP and ACPA, and found that while there is no association between RF positivity and long-term disability, there were independent associations with both ACPA positivity and anti-CarP antibody positivity, with respective beta coefficients of –0.05, 0.12, and 0.14.
“And we see the exact same pattern with disease activity over time,” Dr. Humphreys reported.
Thus, measuring anti-CarP antibodies “may be a useful test to do in addition to both RF and ACPA,” she suggested, adding that measuring these antibodies “may be particularly valuable in patients who lack other antibodies.”
NOAR is funded by Arthritis Research UK. Dr. Humphreys had no relevant financial disclosures.
MANCHESTER, ENGLAND – Anti–carbamylated protein antibodies were associated with both increased disability and greater disease progression over time in patients with early inflammatory arthritis, data from a 20-year retrospective, observational analysis showed.
According to the research, presented at the British Society for Rheumatology annual conference, patients who were also positive for anti–citrullinated protein antibodies (ACPAs) fared worse long term than those who were ACPA negative.
The results showed that anti–carbamylated protein (anti-CarP) antibodies may provide additional prognostic information to current antibody tests in inflammatory arthritis (IA), but their measurement is not yet ready for general use, said Dr. Jennifer Humphreys of the Arthritis Research UK Centre for Epidemiology at the University of Manchester.
“We know that inflammatory arthritis patients who are rheumatoid factor [RF] or ACPA positive have an increased risk of persistent disease and a poor prognosis,” she said.
“In particular, ACPA[s] are associated with severe radiological progression [and] increased disability and mortality. However, not all patients who lack these antibodies do well,” Dr. Humphreys said.
Research at the University of Leiden (the Netherlands) has found that ACPA-negative patients have anti-CarP antibodies in their sera and that these autoantibodies precede the onset of symptoms and predict the development of rheumatoid arthritis (RA) in patients with arthralgia. These antibodies have also been associated with radiological damage, she said.
Because there were few data on the relationship to disease activity and long-term outcomes in the presence of these antibodies, Dr. Humphreys and her associates decided to look at this using data from the Norfolk Arthritis Register (NOAR), she said. The U.K. team worked in collaboration with the Dutch researchers who performed the anti-CarP tests on serum samples taken from 1,995 patients with early IA enrolled in NOAR from 1990 onward.
At baseline, 1,222 patients met American College of Rheumatology/European League Against Rheumatism 2010 criteria for RA. The mean age of onset was 55 years for both the total population with IA and for those with RA. About two-thirds of the participants were women, and their median symptom duration at enrollment was 33 weeks.
Long-term disability was assessed using the health assessment questionnaire (HAQ), and disease activity was evaluated using the disease activity in 28 joints (DAS28) score. The median baseline DAS28 score was 3.81 for the total cohort and 4.45 for patients with RA, and the median baseline HAQ scores were a respective 0.875 and 1.125.
When the team looked at patient baseline characteristics according to baseline anti-CarP status, they found that the 460 (23%) patients who were positive for these autoantibodies were more likely to have higher baseline DAS28 scores (4.23 vs. 3.73), HAQ scores (1.125 vs. 0.875), and C-reactive protein levels (14 mg/L vs. 7 mg/L) than the 1,543 patients who tested anti-CarP negative. Patients positive for anti-Carp antibodies were also less likely to be female (58% vs 68%) and were slightly older at onset (55 vs. 53 years).
Dr. Humphreys noted that there was a lot of overlap among the presence of anti-CarP, ACPA, and RF. Of the 460 who were anti-CarP positive, 124 were positive only for anti-CarP, 148 were also RF positive, 30 were also ACPA positive, and 158 had all three antibodies.
“Patients who were anti-CarP antibody positive had higher HAQ scores at baseline, and this higher level of disability persisted throughout follow-up, and that association remained significant in the multivariate model,” Dr. Humphreys said.
“When we stratified by ACPA status, there was a statistically significant association in the ACPA-negative subgroup but not in the ACPA-positive subgroup,” she added. However, she noted that the beta coefficients for the association between anti-CarP antibodies and HAQ scores over time were similar (0.12 for the total cohort, 0.14 for the ACPA-negative patients, and 0.09 for the ACPA-positive patients) and that confidence intervals were wide.
Similar findings were seen for the association between anti-CarP antibodies and the DAS28 over time, with respective beta coefficients of 0.23, 01.18, and 0.25 for the total cohort, the ACPA-negative subgroup, and the ACPA-positive subgroup. Again, confidence intervals were wide.
The researchers looked at the additive effect of having anti-CarP and ACPA, and found that while there is no association between RF positivity and long-term disability, there were independent associations with both ACPA positivity and anti-CarP antibody positivity, with respective beta coefficients of –0.05, 0.12, and 0.14.
“And we see the exact same pattern with disease activity over time,” Dr. Humphreys reported.
Thus, measuring anti-CarP antibodies “may be a useful test to do in addition to both RF and ACPA,” she suggested, adding that measuring these antibodies “may be particularly valuable in patients who lack other antibodies.”
NOAR is funded by Arthritis Research UK. Dr. Humphreys had no relevant financial disclosures.
AT RHEUMATOLOGY 2015
Key clinical point: Anti-CarP antibodies may provide additional prognostic information to current antibody tests in early inflammatory arthritis.
Major finding: Anti-CarP antibodies were associated with both increased disability and higher disease progression over time.
Data source: A 20-year retrospective, observational study of 1,995 patients who were a median of 55 years old at diagnosis of early IA and who were enrolled in the Norfolk Arthritis Register.
Disclosures: NOAR is funded by Arthritis Research UK. Dr. Humphreys had no relevant financial disclosures.
BSR: Subclinical synovitis common in RA remission
MANCHESTER, U.K. – Subclinical synovitis on ultrasound was apparent in almost half of patients with early rheumatoid arthritis deemed to be in clinical remission and in two-thirds of those with low-disease activity in an observational study.
Power Doppler ultrasound changes were seen in 43% of patients in remission and in 65% of those with LDA. Grey-scale ultrasound changes also were found in 78% of the cohort studied, suggesting synovial hypertrophy. The changes correlated with disease activity. Alterations in synovial T-cell populations also were observed.
Attaining and maintaining clinical remission is a long-established primary treatment goal in rheumatoid arthritis (RA), but current criteria used to determine if this is being achieved are largely subjective, noted Dr. Hanna Gul, who presented the research at the for Rheumatology annual conference.
“Over the years there have been many attempts to define clinical remission criteria using disease activity indices,” said Dr. Gul, a clinical research fellow and specialist registrar at the Leeds Institute of Rheumatic and Musculoskeletal Medicine at the University of Leeds (England).
However, current criteria used in clinical practice and in clinical trials – most commonly the DAS28 – tend to be based on composite scores of disease, can vary in their interpretation, and do not include objective imaging or immunological assessments, she said.
While patients do seem to be increasingly achieving remission, this is not always associated with good outcomes as structural and functional progression still occurs, Dr. Gul said.
“This raises the question regarding the validity of clinical criteria,” she suggested. “Subsequently, the concept of ‘true remission’ has arisen, which is described as the absence of intra- and extra-articular inflammation, immunologic activity, and structural and functional progression.”
To look at the true remission state in more depth using disease activity criteria, imaging, and immunologic markers, Dr. Gul and her colleagues conducted a retrospective, cross-sectional study of 632 patients with early inflammatory arthritis who were considered to be in clinical remission.
Clinical remission was defined as a DAS28-CRP of less than 3.2. A total of 512 patients had a score of less than 2.6, considered as being in ‘strict’ remission, and the remaining 120 had a score of 2.6 to 3.2, having low disease activity (LDA).
The mean age of patients in the strict remission group was 58 years, and in the LDA group it was 57 years. Most of study subjects were female (70% vs. 67%), had anti-citrullinated antibodies (70% and 68%), and were rheumatoid factor positive (59% and 67%).
The median duration of disease was 6.7 months longer in the patients achieving clinical remission, at 12.9 months versus 6.2 months in the LDA group, and patients in the strict remission group had been in remission for an average of 7.6 months (in the LDA group no patient was in strict remission by definition).
The majority (75% in both groups) had been or was being treated with conventional disease-modifying drugs (csDMARDs) either alone (40%-45%) or in combination (30%-34%). About 12.5% of patients in the remission group, and 7.5% of those in the LDA group were being treated with biologics, mostly in combination with a csDMARD.
“In terms of clinical characteristics, the levels of CRP and swollen and tender joints were generally low,” Dr. Gul reported. “There was also overlap between remission and low disease activity states.”
Various patient-reported outcomes were assessed and showed that the levels of disability and functional impairment were generally low, but that there were differences between the remission and LDA states, with the best outcomes reported in patients with a CRP, tender and swollen joint count of zero.
Flow cytometry was used to analyze T-cell populations in 51 patients compared to 120 healthy controls. Results showed that there was a 9% decrease from the normal range in the percentage of naive CD4+ T-cells, a 26% increase in inflammation-related cells, and a 50% decrease in regulatory T-cells.
“This is the most extensive examination of T-cell abnormalities in RA and confirms T-regulatory abnormalities in patients with clinical remission,” Dr. Gul observed.
“Further research is required for understanding the implications, specifically with regard to T-cell abnormalities as predictors of flare,” she said.
The research was supported by the U.K. National Institute for Health Research. Dr. Gul had no disclosures.
MANCHESTER, U.K. – Subclinical synovitis on ultrasound was apparent in almost half of patients with early rheumatoid arthritis deemed to be in clinical remission and in two-thirds of those with low-disease activity in an observational study.
Power Doppler ultrasound changes were seen in 43% of patients in remission and in 65% of those with LDA. Grey-scale ultrasound changes also were found in 78% of the cohort studied, suggesting synovial hypertrophy. The changes correlated with disease activity. Alterations in synovial T-cell populations also were observed.
Attaining and maintaining clinical remission is a long-established primary treatment goal in rheumatoid arthritis (RA), but current criteria used to determine if this is being achieved are largely subjective, noted Dr. Hanna Gul, who presented the research at the for Rheumatology annual conference.
“Over the years there have been many attempts to define clinical remission criteria using disease activity indices,” said Dr. Gul, a clinical research fellow and specialist registrar at the Leeds Institute of Rheumatic and Musculoskeletal Medicine at the University of Leeds (England).
However, current criteria used in clinical practice and in clinical trials – most commonly the DAS28 – tend to be based on composite scores of disease, can vary in their interpretation, and do not include objective imaging or immunological assessments, she said.
While patients do seem to be increasingly achieving remission, this is not always associated with good outcomes as structural and functional progression still occurs, Dr. Gul said.
“This raises the question regarding the validity of clinical criteria,” she suggested. “Subsequently, the concept of ‘true remission’ has arisen, which is described as the absence of intra- and extra-articular inflammation, immunologic activity, and structural and functional progression.”
To look at the true remission state in more depth using disease activity criteria, imaging, and immunologic markers, Dr. Gul and her colleagues conducted a retrospective, cross-sectional study of 632 patients with early inflammatory arthritis who were considered to be in clinical remission.
Clinical remission was defined as a DAS28-CRP of less than 3.2. A total of 512 patients had a score of less than 2.6, considered as being in ‘strict’ remission, and the remaining 120 had a score of 2.6 to 3.2, having low disease activity (LDA).
The mean age of patients in the strict remission group was 58 years, and in the LDA group it was 57 years. Most of study subjects were female (70% vs. 67%), had anti-citrullinated antibodies (70% and 68%), and were rheumatoid factor positive (59% and 67%).
The median duration of disease was 6.7 months longer in the patients achieving clinical remission, at 12.9 months versus 6.2 months in the LDA group, and patients in the strict remission group had been in remission for an average of 7.6 months (in the LDA group no patient was in strict remission by definition).
The majority (75% in both groups) had been or was being treated with conventional disease-modifying drugs (csDMARDs) either alone (40%-45%) or in combination (30%-34%). About 12.5% of patients in the remission group, and 7.5% of those in the LDA group were being treated with biologics, mostly in combination with a csDMARD.
“In terms of clinical characteristics, the levels of CRP and swollen and tender joints were generally low,” Dr. Gul reported. “There was also overlap between remission and low disease activity states.”
Various patient-reported outcomes were assessed and showed that the levels of disability and functional impairment were generally low, but that there were differences between the remission and LDA states, with the best outcomes reported in patients with a CRP, tender and swollen joint count of zero.
Flow cytometry was used to analyze T-cell populations in 51 patients compared to 120 healthy controls. Results showed that there was a 9% decrease from the normal range in the percentage of naive CD4+ T-cells, a 26% increase in inflammation-related cells, and a 50% decrease in regulatory T-cells.
“This is the most extensive examination of T-cell abnormalities in RA and confirms T-regulatory abnormalities in patients with clinical remission,” Dr. Gul observed.
“Further research is required for understanding the implications, specifically with regard to T-cell abnormalities as predictors of flare,” she said.
The research was supported by the U.K. National Institute for Health Research. Dr. Gul had no disclosures.
MANCHESTER, U.K. – Subclinical synovitis on ultrasound was apparent in almost half of patients with early rheumatoid arthritis deemed to be in clinical remission and in two-thirds of those with low-disease activity in an observational study.
Power Doppler ultrasound changes were seen in 43% of patients in remission and in 65% of those with LDA. Grey-scale ultrasound changes also were found in 78% of the cohort studied, suggesting synovial hypertrophy. The changes correlated with disease activity. Alterations in synovial T-cell populations also were observed.
Attaining and maintaining clinical remission is a long-established primary treatment goal in rheumatoid arthritis (RA), but current criteria used to determine if this is being achieved are largely subjective, noted Dr. Hanna Gul, who presented the research at the for Rheumatology annual conference.
“Over the years there have been many attempts to define clinical remission criteria using disease activity indices,” said Dr. Gul, a clinical research fellow and specialist registrar at the Leeds Institute of Rheumatic and Musculoskeletal Medicine at the University of Leeds (England).
However, current criteria used in clinical practice and in clinical trials – most commonly the DAS28 – tend to be based on composite scores of disease, can vary in their interpretation, and do not include objective imaging or immunological assessments, she said.
While patients do seem to be increasingly achieving remission, this is not always associated with good outcomes as structural and functional progression still occurs, Dr. Gul said.
“This raises the question regarding the validity of clinical criteria,” she suggested. “Subsequently, the concept of ‘true remission’ has arisen, which is described as the absence of intra- and extra-articular inflammation, immunologic activity, and structural and functional progression.”
To look at the true remission state in more depth using disease activity criteria, imaging, and immunologic markers, Dr. Gul and her colleagues conducted a retrospective, cross-sectional study of 632 patients with early inflammatory arthritis who were considered to be in clinical remission.
Clinical remission was defined as a DAS28-CRP of less than 3.2. A total of 512 patients had a score of less than 2.6, considered as being in ‘strict’ remission, and the remaining 120 had a score of 2.6 to 3.2, having low disease activity (LDA).
The mean age of patients in the strict remission group was 58 years, and in the LDA group it was 57 years. Most of study subjects were female (70% vs. 67%), had anti-citrullinated antibodies (70% and 68%), and were rheumatoid factor positive (59% and 67%).
The median duration of disease was 6.7 months longer in the patients achieving clinical remission, at 12.9 months versus 6.2 months in the LDA group, and patients in the strict remission group had been in remission for an average of 7.6 months (in the LDA group no patient was in strict remission by definition).
The majority (75% in both groups) had been or was being treated with conventional disease-modifying drugs (csDMARDs) either alone (40%-45%) or in combination (30%-34%). About 12.5% of patients in the remission group, and 7.5% of those in the LDA group were being treated with biologics, mostly in combination with a csDMARD.
“In terms of clinical characteristics, the levels of CRP and swollen and tender joints were generally low,” Dr. Gul reported. “There was also overlap between remission and low disease activity states.”
Various patient-reported outcomes were assessed and showed that the levels of disability and functional impairment were generally low, but that there were differences between the remission and LDA states, with the best outcomes reported in patients with a CRP, tender and swollen joint count of zero.
Flow cytometry was used to analyze T-cell populations in 51 patients compared to 120 healthy controls. Results showed that there was a 9% decrease from the normal range in the percentage of naive CD4+ T-cells, a 26% increase in inflammation-related cells, and a 50% decrease in regulatory T-cells.
“This is the most extensive examination of T-cell abnormalities in RA and confirms T-regulatory abnormalities in patients with clinical remission,” Dr. Gul observed.
“Further research is required for understanding the implications, specifically with regard to T-cell abnormalities as predictors of flare,” she said.
The research was supported by the U.K. National Institute for Health Research. Dr. Gul had no disclosures.
AT RHEUMATOLOGY 2015
Key clinical point: Patients with RA in clinical remission defined via clinical criteria alone may still have subclinical inflammation.
Major finding: Power Doppler ultrasound changes were seen in 43% of patients in remission and in 65% of those with LDA.
Data source: Retrospective, cross-sectional, observational study of 632 patients with early inflammatory arthritis in clinical remission (DAS28-CRP < 3.2).
Disclosures: The research was supported by the U.K. National Institute for Health Research. Dr. Gul had no disclosures.
Synovial pathotype could help guide anti-TNF therapy
MANCHESTER, U.K. – Analysis of synovial tissue taken by ultrasound-guided biopsy could help stratify patients with rheumatoid arthritis and decide if treatment with a tumor necrosis factor inhibitor is appropriate, according to research presented at the British Society for Rheumatology annual conference.
In a small yet intriguing study, Dr. Maria Di Cicco of Queen Mary University of London and her colleagues found that patients who had a lymphoid synovial pathotype were more likely to respond to treatment with the anti-TNF inhibitor certolizumab pegol than if they had other synovial pathotypes.
“Rheumatoid arthritis is a clinically heterogeneous disease to the extent that we should think of it as a syndrome rather than a disease,” Dr. Di Cicco said. “This heterogeneity is expressed as a variable outcome and response to treatment,” she added.
Although biologic drugs have dramatically changed how patients are treated, 30%-40% of patients do not respond to treatment and the way biologic treatment is selected for RA is more down to trial and error than personalized medicine, Dr. Di Cicco observed. This is concerning, as the number of biologic medicines available is exponentially increasing and ways of matching the right drug to the right patient need to be found.
The aim of the current study was to see if different synovial subtypes could influence patients’ first response to treatment with anti-TNF therapy. Recent research looking at synovial tissue suggests that there are three clear synovial subtypes: myeloid, Pauci-immune, and lymphoid.
The myeloid and Pauci-immune pathotypes are characterized by diffuse cellular infiltration distributed evenly through the synovium. The lymphoid synovial pathotype, on the other hand, is characterized by the presence of large lymphocytic aggregates that are known as ectopic lymphoid structures (ELS). Dr. Di Cicco noted that ELS had been linked to B-cell proliferation within the synovium and to promoting in situ autoantibody production and class switching.
For the study, 28 patients with RA on stable doses of methotrexate who qualified for anti-TNF treatment according to U.K. National Institute for Health and Care Excellence guidance were recruited at a single center. All patients underwent ultrasound-guided biopsy of an active joint, which tended to be a small joint such as the wrist or metacarpophalangeal, before they were treated with certolizumab pegol. Twenty patients had a repeat synovial biopsy when the clinical efficacy of treatment was assessed at 12 weeks.
A minimum of six samples were taken from each biopsied joint, and this material underwent immunohistochemical analysis for the presence of T cells expressing CD3, B cells expressing CD20, macrophages expressing CD68, and plasma cells expressing CD138.
Samples were then classified as being the lymphoid pathotype if they contained mainly B and T cells and formed large lymphocytic aggregates or ELS. Myeloid subtype was defined as the presence of sublining macrophages and Pauci-immune if none of these features was present.
Thirteen (46.4%) patients were found to the have the Pauci-immune pathotype, with the lymphoid pathotype being the next most common (39.3% of patients), and four (14.3%) had the myeloid pathotype.
Apart from no prior oral steroid use in patients with the lymphoid pathotype versus 75% of patients with the myeloid pathotype and 38.5% of patients with the Pauci-immune pathotype, there were no significant differences among the groups in terms of demographics, clinical features, inflammatory markers, antibody or erosive status, or other treatments.
“After 12 weeks of therapy, the myeloid and the lymphoid groups showed a significantly higher fall in [Disease Activity Score] 28 compared to the Pauci-immune group,” Dr. Di Cicco reported.
“We also assessed achievement of EULAR response, and 71% of patients did respond. Half of them were good responders and half of them were moderate responders,” she added.
EULAR response by synovial pathotype was 100% for patients with the lymphoid pathotype, 75% for those with the myeloid pathotype, and 46% for those with the Pauci-immune pathotype.
Greater decreases in synovial thickening and power Doppler ultrasound also were seen in the myeloid and lymphoid pathotypes, compared with the Pauci-immune pathotype, which appeared more resistant to change in response to the anti-TNF therapy.
In the 20 patients who had a repeat biopsy, 9 had ELS at baseline. After treatment, three patients were ELS negative, four remained ELS positive, and two were ungraded.
“Our work shows that the presence of synovial ectopic lymphoid structures defining a lymphoid pathotype is a predictor of clinical response to anti-TNF-alpha,” Dr. Di Cicco said. Although further research and confirmation is needed, “we think that synovial tissue analysis could be considered as a promising tool to stratify RA patients and guide therapeutic decision,” she said.
Dr. Di Cicco had no disclosures.
MANCHESTER, U.K. – Analysis of synovial tissue taken by ultrasound-guided biopsy could help stratify patients with rheumatoid arthritis and decide if treatment with a tumor necrosis factor inhibitor is appropriate, according to research presented at the British Society for Rheumatology annual conference.
In a small yet intriguing study, Dr. Maria Di Cicco of Queen Mary University of London and her colleagues found that patients who had a lymphoid synovial pathotype were more likely to respond to treatment with the anti-TNF inhibitor certolizumab pegol than if they had other synovial pathotypes.
“Rheumatoid arthritis is a clinically heterogeneous disease to the extent that we should think of it as a syndrome rather than a disease,” Dr. Di Cicco said. “This heterogeneity is expressed as a variable outcome and response to treatment,” she added.
Although biologic drugs have dramatically changed how patients are treated, 30%-40% of patients do not respond to treatment and the way biologic treatment is selected for RA is more down to trial and error than personalized medicine, Dr. Di Cicco observed. This is concerning, as the number of biologic medicines available is exponentially increasing and ways of matching the right drug to the right patient need to be found.
The aim of the current study was to see if different synovial subtypes could influence patients’ first response to treatment with anti-TNF therapy. Recent research looking at synovial tissue suggests that there are three clear synovial subtypes: myeloid, Pauci-immune, and lymphoid.
The myeloid and Pauci-immune pathotypes are characterized by diffuse cellular infiltration distributed evenly through the synovium. The lymphoid synovial pathotype, on the other hand, is characterized by the presence of large lymphocytic aggregates that are known as ectopic lymphoid structures (ELS). Dr. Di Cicco noted that ELS had been linked to B-cell proliferation within the synovium and to promoting in situ autoantibody production and class switching.
For the study, 28 patients with RA on stable doses of methotrexate who qualified for anti-TNF treatment according to U.K. National Institute for Health and Care Excellence guidance were recruited at a single center. All patients underwent ultrasound-guided biopsy of an active joint, which tended to be a small joint such as the wrist or metacarpophalangeal, before they were treated with certolizumab pegol. Twenty patients had a repeat synovial biopsy when the clinical efficacy of treatment was assessed at 12 weeks.
A minimum of six samples were taken from each biopsied joint, and this material underwent immunohistochemical analysis for the presence of T cells expressing CD3, B cells expressing CD20, macrophages expressing CD68, and plasma cells expressing CD138.
Samples were then classified as being the lymphoid pathotype if they contained mainly B and T cells and formed large lymphocytic aggregates or ELS. Myeloid subtype was defined as the presence of sublining macrophages and Pauci-immune if none of these features was present.
Thirteen (46.4%) patients were found to the have the Pauci-immune pathotype, with the lymphoid pathotype being the next most common (39.3% of patients), and four (14.3%) had the myeloid pathotype.
Apart from no prior oral steroid use in patients with the lymphoid pathotype versus 75% of patients with the myeloid pathotype and 38.5% of patients with the Pauci-immune pathotype, there were no significant differences among the groups in terms of demographics, clinical features, inflammatory markers, antibody or erosive status, or other treatments.
“After 12 weeks of therapy, the myeloid and the lymphoid groups showed a significantly higher fall in [Disease Activity Score] 28 compared to the Pauci-immune group,” Dr. Di Cicco reported.
“We also assessed achievement of EULAR response, and 71% of patients did respond. Half of them were good responders and half of them were moderate responders,” she added.
EULAR response by synovial pathotype was 100% for patients with the lymphoid pathotype, 75% for those with the myeloid pathotype, and 46% for those with the Pauci-immune pathotype.
Greater decreases in synovial thickening and power Doppler ultrasound also were seen in the myeloid and lymphoid pathotypes, compared with the Pauci-immune pathotype, which appeared more resistant to change in response to the anti-TNF therapy.
In the 20 patients who had a repeat biopsy, 9 had ELS at baseline. After treatment, three patients were ELS negative, four remained ELS positive, and two were ungraded.
“Our work shows that the presence of synovial ectopic lymphoid structures defining a lymphoid pathotype is a predictor of clinical response to anti-TNF-alpha,” Dr. Di Cicco said. Although further research and confirmation is needed, “we think that synovial tissue analysis could be considered as a promising tool to stratify RA patients and guide therapeutic decision,” she said.
Dr. Di Cicco had no disclosures.
MANCHESTER, U.K. – Analysis of synovial tissue taken by ultrasound-guided biopsy could help stratify patients with rheumatoid arthritis and decide if treatment with a tumor necrosis factor inhibitor is appropriate, according to research presented at the British Society for Rheumatology annual conference.
In a small yet intriguing study, Dr. Maria Di Cicco of Queen Mary University of London and her colleagues found that patients who had a lymphoid synovial pathotype were more likely to respond to treatment with the anti-TNF inhibitor certolizumab pegol than if they had other synovial pathotypes.
“Rheumatoid arthritis is a clinically heterogeneous disease to the extent that we should think of it as a syndrome rather than a disease,” Dr. Di Cicco said. “This heterogeneity is expressed as a variable outcome and response to treatment,” she added.
Although biologic drugs have dramatically changed how patients are treated, 30%-40% of patients do not respond to treatment and the way biologic treatment is selected for RA is more down to trial and error than personalized medicine, Dr. Di Cicco observed. This is concerning, as the number of biologic medicines available is exponentially increasing and ways of matching the right drug to the right patient need to be found.
The aim of the current study was to see if different synovial subtypes could influence patients’ first response to treatment with anti-TNF therapy. Recent research looking at synovial tissue suggests that there are three clear synovial subtypes: myeloid, Pauci-immune, and lymphoid.
The myeloid and Pauci-immune pathotypes are characterized by diffuse cellular infiltration distributed evenly through the synovium. The lymphoid synovial pathotype, on the other hand, is characterized by the presence of large lymphocytic aggregates that are known as ectopic lymphoid structures (ELS). Dr. Di Cicco noted that ELS had been linked to B-cell proliferation within the synovium and to promoting in situ autoantibody production and class switching.
For the study, 28 patients with RA on stable doses of methotrexate who qualified for anti-TNF treatment according to U.K. National Institute for Health and Care Excellence guidance were recruited at a single center. All patients underwent ultrasound-guided biopsy of an active joint, which tended to be a small joint such as the wrist or metacarpophalangeal, before they were treated with certolizumab pegol. Twenty patients had a repeat synovial biopsy when the clinical efficacy of treatment was assessed at 12 weeks.
A minimum of six samples were taken from each biopsied joint, and this material underwent immunohistochemical analysis for the presence of T cells expressing CD3, B cells expressing CD20, macrophages expressing CD68, and plasma cells expressing CD138.
Samples were then classified as being the lymphoid pathotype if they contained mainly B and T cells and formed large lymphocytic aggregates or ELS. Myeloid subtype was defined as the presence of sublining macrophages and Pauci-immune if none of these features was present.
Thirteen (46.4%) patients were found to the have the Pauci-immune pathotype, with the lymphoid pathotype being the next most common (39.3% of patients), and four (14.3%) had the myeloid pathotype.
Apart from no prior oral steroid use in patients with the lymphoid pathotype versus 75% of patients with the myeloid pathotype and 38.5% of patients with the Pauci-immune pathotype, there were no significant differences among the groups in terms of demographics, clinical features, inflammatory markers, antibody or erosive status, or other treatments.
“After 12 weeks of therapy, the myeloid and the lymphoid groups showed a significantly higher fall in [Disease Activity Score] 28 compared to the Pauci-immune group,” Dr. Di Cicco reported.
“We also assessed achievement of EULAR response, and 71% of patients did respond. Half of them were good responders and half of them were moderate responders,” she added.
EULAR response by synovial pathotype was 100% for patients with the lymphoid pathotype, 75% for those with the myeloid pathotype, and 46% for those with the Pauci-immune pathotype.
Greater decreases in synovial thickening and power Doppler ultrasound also were seen in the myeloid and lymphoid pathotypes, compared with the Pauci-immune pathotype, which appeared more resistant to change in response to the anti-TNF therapy.
In the 20 patients who had a repeat biopsy, 9 had ELS at baseline. After treatment, three patients were ELS negative, four remained ELS positive, and two were ungraded.
“Our work shows that the presence of synovial ectopic lymphoid structures defining a lymphoid pathotype is a predictor of clinical response to anti-TNF-alpha,” Dr. Di Cicco said. Although further research and confirmation is needed, “we think that synovial tissue analysis could be considered as a promising tool to stratify RA patients and guide therapeutic decision,” she said.
Dr. Di Cicco had no disclosures.
AT RHEUMATOLOGY 2015
Key clinical point: Early data suggest that synovial tissue analysis may be a potential tool to guide anti-TNF therapy, although more data are needed.
Major finding: EULAR response by synovial pathotype was 100% for patients with the lymphoid pathotype, 75% for those with the myeloid pathotype, and 46% for those with the Pauci-immune pathotype.
Data source: 28 patients with rheumatoid arthritis needing first anti-TNF treatment.
Disclosures: Dr. Di Cicco had no disclosures.
VA study whittles obesity paradox in RA down to size
Weight loss is a strong predictor of death in rheumatoid arthritis, with the greatest risk of death in low body mass index patients who were previously obese.
“Our study is the first to demonstrate a strong and independent association between recent weight loss, as opposed to BMI per se, and risk of subsequent death in patients with RA. These observations represent an important step in explaining the ‘obesity paradox’ observed in this disease,” Dr. Joshua F. Baker and his colleagues wrote in a study published online May 4 in Arthritis & Rheumatology (doi:10.1002/art.39136).
Although obesity is associated with a higher incidence of a number of diseases, including cardiovascular disease, diabetes, and certain cancers, several epidemiologic studies in the elderly and chronic disease states have reported that overweight patients may have lower mortality than normal-weight patients.
More recent evidence, however, suggests that weight loss due to the development of chronic illness is a significant confounder and may explain the seemingly protective effect of obesity on mortality. Studies that fail to consider weight loss that occurs in chronic illnesses such as RA are likely to underestimate causal associations between greater weight and death, noted Dr. Baker, a rheumatologist at the University of Pennsylvania, Philadelphia, and the Philadelphia VA Medical Center.
To unravel the “obesity paradox” in RA, the investigators examined multiple BMI measures over time and date of death for 1,674 patients with rheumatologist-diagnosed RA in the Veterans Affairs Rheumatoid Arthritis registry. The annualized rate of change in BMI was determined from the slope of BMI over time over all available visits in the preceding 13 months. Cox multivariable proportional hazard models assessed associations between BMI measures and mortality.
At enrollment, the average duration of RA was 7.4 years, average C-reactive protein was 0.8 mg/dL, average BMI was 28.5 kg/m², and maximum BMI was 33.3 kg/m². The cohort’s average age was 63.5 years and 91% were male.
Among the 1,674 patients, there were 312 deaths in 9,183 person-years of follow-up. The median follow-up was 5.5 years, and the median interval between visits was 105 days.
A modest decline in BMI of ≥1 kg/m² over the preceding observation period was associated with an increased risk of death before and after adjustment for multiple potential confounders, including demographics, comorbidities, BMI, smoking, and RA therapies (hazard ratio, 1.99; P < .001), Dr. Baker reported.
This remained significant in a subsample of observation periods after further adjustment for C-reactive protein and physical function (HR, 1.81; P < .001).
The analysis revealed a dose-dependent increase in the risk of death based on the annualized rate of decrease in BMI, he noted.
A loss of less than 2 kg/m² of BMI per year did not have an increased risk of death in the subsequent observation period after multivariable adjustment (HR, 1.12; P = .4). In contrast, a loss of 2-3 kg/m² of BMI per year increased the risk of death by about 1.5 times (HR, 1.65; P = .02) and a loss of more than 3 kg/m² per year was associated with the greatest risk of death (HR, 2.49; P < .001).
“This observation supports the common dogma that more rapid weight loss is a poor prognostic sign, while slower changes should be less alarming,” the authors wrote.
In multivariable models, maximum BMI was not associated with risk of death (HR, 1.00; P = .80).
In models including both current and maximum BMI, however, a lower current BMI and greater maximum BMI category were each independently associated with a greater risk of death, Dr. Baker and his associates reported.
Among patients with a similar maximum BMI, a low current BMI of less than 20 kg/m² is associated with a greater risk of death (HR, 3.82; P < .001). Similarly, among patients with a similar current BMI, those with an obese maximum BMI over 30 kg/m² were at an increased risk of death (HR, 2.22; P = .001).
This suggests that a patient with a low BMI at a given visit and a history of BMI in the obese range is at a dramatically increased risk of death, compared with a normal-weight patient with a consistently normal weight (HR, 8.52; P < .001), the authors reported.
Overall, the observations do not support a biologically protective role of obesity in RA and support close monitoring of patients with RA who demonstrate unintentional weight loss, they concluded.
“The next steps in this area are to understand more specifically how body composition may change over time in the disease and how this may tie in with the observations here with weight loss and the obesity paradox,” Dr. Baker said in an interview. “In general, I think it is important to understand how weight and body composition changes themselves impact long-term outcomes such as comorbid disease, fractures, cardiovascular disease, and death.”
The VARA registry is supported by the Nebraska Arthritis Outcomes Research Center at the University of Nebraska Medical Center and by the Veterans Affairs Health Services Research and Development Program of the Veterans Health Administration. The investigators were supported by awards from the VA, a Rheumatology Research Foundation Investigator Award, and the National Institute of Arthritis and Musculoskeletal and Skin Disorders.
On Twitter @pwendl
Weight loss is a strong predictor of death in rheumatoid arthritis, with the greatest risk of death in low body mass index patients who were previously obese.
“Our study is the first to demonstrate a strong and independent association between recent weight loss, as opposed to BMI per se, and risk of subsequent death in patients with RA. These observations represent an important step in explaining the ‘obesity paradox’ observed in this disease,” Dr. Joshua F. Baker and his colleagues wrote in a study published online May 4 in Arthritis & Rheumatology (doi:10.1002/art.39136).
Although obesity is associated with a higher incidence of a number of diseases, including cardiovascular disease, diabetes, and certain cancers, several epidemiologic studies in the elderly and chronic disease states have reported that overweight patients may have lower mortality than normal-weight patients.
More recent evidence, however, suggests that weight loss due to the development of chronic illness is a significant confounder and may explain the seemingly protective effect of obesity on mortality. Studies that fail to consider weight loss that occurs in chronic illnesses such as RA are likely to underestimate causal associations between greater weight and death, noted Dr. Baker, a rheumatologist at the University of Pennsylvania, Philadelphia, and the Philadelphia VA Medical Center.
To unravel the “obesity paradox” in RA, the investigators examined multiple BMI measures over time and date of death for 1,674 patients with rheumatologist-diagnosed RA in the Veterans Affairs Rheumatoid Arthritis registry. The annualized rate of change in BMI was determined from the slope of BMI over time over all available visits in the preceding 13 months. Cox multivariable proportional hazard models assessed associations between BMI measures and mortality.
At enrollment, the average duration of RA was 7.4 years, average C-reactive protein was 0.8 mg/dL, average BMI was 28.5 kg/m², and maximum BMI was 33.3 kg/m². The cohort’s average age was 63.5 years and 91% were male.
Among the 1,674 patients, there were 312 deaths in 9,183 person-years of follow-up. The median follow-up was 5.5 years, and the median interval between visits was 105 days.
A modest decline in BMI of ≥1 kg/m² over the preceding observation period was associated with an increased risk of death before and after adjustment for multiple potential confounders, including demographics, comorbidities, BMI, smoking, and RA therapies (hazard ratio, 1.99; P < .001), Dr. Baker reported.
This remained significant in a subsample of observation periods after further adjustment for C-reactive protein and physical function (HR, 1.81; P < .001).
The analysis revealed a dose-dependent increase in the risk of death based on the annualized rate of decrease in BMI, he noted.
A loss of less than 2 kg/m² of BMI per year did not have an increased risk of death in the subsequent observation period after multivariable adjustment (HR, 1.12; P = .4). In contrast, a loss of 2-3 kg/m² of BMI per year increased the risk of death by about 1.5 times (HR, 1.65; P = .02) and a loss of more than 3 kg/m² per year was associated with the greatest risk of death (HR, 2.49; P < .001).
“This observation supports the common dogma that more rapid weight loss is a poor prognostic sign, while slower changes should be less alarming,” the authors wrote.
In multivariable models, maximum BMI was not associated with risk of death (HR, 1.00; P = .80).
In models including both current and maximum BMI, however, a lower current BMI and greater maximum BMI category were each independently associated with a greater risk of death, Dr. Baker and his associates reported.
Among patients with a similar maximum BMI, a low current BMI of less than 20 kg/m² is associated with a greater risk of death (HR, 3.82; P < .001). Similarly, among patients with a similar current BMI, those with an obese maximum BMI over 30 kg/m² were at an increased risk of death (HR, 2.22; P = .001).
This suggests that a patient with a low BMI at a given visit and a history of BMI in the obese range is at a dramatically increased risk of death, compared with a normal-weight patient with a consistently normal weight (HR, 8.52; P < .001), the authors reported.
Overall, the observations do not support a biologically protective role of obesity in RA and support close monitoring of patients with RA who demonstrate unintentional weight loss, they concluded.
“The next steps in this area are to understand more specifically how body composition may change over time in the disease and how this may tie in with the observations here with weight loss and the obesity paradox,” Dr. Baker said in an interview. “In general, I think it is important to understand how weight and body composition changes themselves impact long-term outcomes such as comorbid disease, fractures, cardiovascular disease, and death.”
The VARA registry is supported by the Nebraska Arthritis Outcomes Research Center at the University of Nebraska Medical Center and by the Veterans Affairs Health Services Research and Development Program of the Veterans Health Administration. The investigators were supported by awards from the VA, a Rheumatology Research Foundation Investigator Award, and the National Institute of Arthritis and Musculoskeletal and Skin Disorders.
On Twitter @pwendl
Weight loss is a strong predictor of death in rheumatoid arthritis, with the greatest risk of death in low body mass index patients who were previously obese.
“Our study is the first to demonstrate a strong and independent association between recent weight loss, as opposed to BMI per se, and risk of subsequent death in patients with RA. These observations represent an important step in explaining the ‘obesity paradox’ observed in this disease,” Dr. Joshua F. Baker and his colleagues wrote in a study published online May 4 in Arthritis & Rheumatology (doi:10.1002/art.39136).
Although obesity is associated with a higher incidence of a number of diseases, including cardiovascular disease, diabetes, and certain cancers, several epidemiologic studies in the elderly and chronic disease states have reported that overweight patients may have lower mortality than normal-weight patients.
More recent evidence, however, suggests that weight loss due to the development of chronic illness is a significant confounder and may explain the seemingly protective effect of obesity on mortality. Studies that fail to consider weight loss that occurs in chronic illnesses such as RA are likely to underestimate causal associations between greater weight and death, noted Dr. Baker, a rheumatologist at the University of Pennsylvania, Philadelphia, and the Philadelphia VA Medical Center.
To unravel the “obesity paradox” in RA, the investigators examined multiple BMI measures over time and date of death for 1,674 patients with rheumatologist-diagnosed RA in the Veterans Affairs Rheumatoid Arthritis registry. The annualized rate of change in BMI was determined from the slope of BMI over time over all available visits in the preceding 13 months. Cox multivariable proportional hazard models assessed associations between BMI measures and mortality.
At enrollment, the average duration of RA was 7.4 years, average C-reactive protein was 0.8 mg/dL, average BMI was 28.5 kg/m², and maximum BMI was 33.3 kg/m². The cohort’s average age was 63.5 years and 91% were male.
Among the 1,674 patients, there were 312 deaths in 9,183 person-years of follow-up. The median follow-up was 5.5 years, and the median interval between visits was 105 days.
A modest decline in BMI of ≥1 kg/m² over the preceding observation period was associated with an increased risk of death before and after adjustment for multiple potential confounders, including demographics, comorbidities, BMI, smoking, and RA therapies (hazard ratio, 1.99; P < .001), Dr. Baker reported.
This remained significant in a subsample of observation periods after further adjustment for C-reactive protein and physical function (HR, 1.81; P < .001).
The analysis revealed a dose-dependent increase in the risk of death based on the annualized rate of decrease in BMI, he noted.
A loss of less than 2 kg/m² of BMI per year did not have an increased risk of death in the subsequent observation period after multivariable adjustment (HR, 1.12; P = .4). In contrast, a loss of 2-3 kg/m² of BMI per year increased the risk of death by about 1.5 times (HR, 1.65; P = .02) and a loss of more than 3 kg/m² per year was associated with the greatest risk of death (HR, 2.49; P < .001).
“This observation supports the common dogma that more rapid weight loss is a poor prognostic sign, while slower changes should be less alarming,” the authors wrote.
In multivariable models, maximum BMI was not associated with risk of death (HR, 1.00; P = .80).
In models including both current and maximum BMI, however, a lower current BMI and greater maximum BMI category were each independently associated with a greater risk of death, Dr. Baker and his associates reported.
Among patients with a similar maximum BMI, a low current BMI of less than 20 kg/m² is associated with a greater risk of death (HR, 3.82; P < .001). Similarly, among patients with a similar current BMI, those with an obese maximum BMI over 30 kg/m² were at an increased risk of death (HR, 2.22; P = .001).
This suggests that a patient with a low BMI at a given visit and a history of BMI in the obese range is at a dramatically increased risk of death, compared with a normal-weight patient with a consistently normal weight (HR, 8.52; P < .001), the authors reported.
Overall, the observations do not support a biologically protective role of obesity in RA and support close monitoring of patients with RA who demonstrate unintentional weight loss, they concluded.
“The next steps in this area are to understand more specifically how body composition may change over time in the disease and how this may tie in with the observations here with weight loss and the obesity paradox,” Dr. Baker said in an interview. “In general, I think it is important to understand how weight and body composition changes themselves impact long-term outcomes such as comorbid disease, fractures, cardiovascular disease, and death.”
The VARA registry is supported by the Nebraska Arthritis Outcomes Research Center at the University of Nebraska Medical Center and by the Veterans Affairs Health Services Research and Development Program of the Veterans Health Administration. The investigators were supported by awards from the VA, a Rheumatology Research Foundation Investigator Award, and the National Institute of Arthritis and Musculoskeletal and Skin Disorders.
On Twitter @pwendl
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point: Weight loss is a strong independent predictor of death in rheumatoid arthritis.
Major finding: Weight loss of more than 3 kg/m² per year was associated with the greatest risk of death (HR, 2.49; P < .001).
Data source: A national sample of 1,674 patients in the Veterans Affairs Rheumatoid Arthritis registry.
Disclosures: The VARA registry is supported by the Nebraska Arthritis Outcomes Research Center at the University of Nebraska Medical Center and by the Veterans Affairs Health Services Research and Development Program of the Veterans Health Administration. The investigators were supported by awards from the VA, a Rheumatology Research Foundation Investigator Award, and by the National Institute of Arthritis and Musculoskeletal and Skin Disorders.
Consider LDL-C and HDL-C when estimating CV risk in RA
Despite a similar relationship between lipid levels and cardiovascular disease risk in patients with or without rheumatoid arthritis, people with RA have an almost two-fold increased risk of having a major cardiovascular event, research shows.
The findings confirm that existing CV risk calculators are suboptimal for patients with RA and that improved methods of measuring risk in this population are needed, say the study authors from the Brigham and Women’s Hospital in Boston. They also show that there may be potential benefit in considering both LDL-C and HDL-C levels when estimating CV risk in RA, according to Dr. Katherine P. Liao and her associates from Brigham and Women’s Hospital, Boston (Arthritis & Rheumatology 2015; [doi: 10.1002/art.39165]).
While there have been many studies examining the relationship between low density lipoprotein cholesterol levels (LDL-C) and CV risk, only one study described the relationship between high density lipoprotein cholesterol levels (HDL-C) with CV risk among RA patients. It found higher levels of HDL-C were associated with lower CV risk in people with RA, consistent with the general population. “[This] suggests that HDL-C levels may provide important information for estimating CV risk independent of LDL-C,” according to Dr. Liao and her associates.
To assess the relationship between LDL-C and HDL-C with CV risk in RA patients compared with the general population, the researchers compared 16,085 RA patients with 48,499 matched controls without RA from the United Healthcare database. In line with other studies the researchers found evidence of the “lipid paradox” – where low and high LDL-C levels confer a similar CV risk – in both groups.
Looking at HDL-C levels, the risk of a major cardiovascular event (MACE) was highest among patients with lower levels compared with higher levels in both RA and non-RA subjects. For example subjects in the highest quintile had a 55% lower risk for MACE (hazard ratio, 0.45; 95% confidence interval, 0.48-0.72) compared with the lowest quintile after adjusting for CV risk factors. However,in a model that included both LDL-C and HDL-C levels, patients with RA had an elevated risk of MACE (HR 1.7; 95% CI, 1.5-1.9) independent of LDL-C levels.
“This magnitude of increased CV risk is similar to studies published a decade ago, suggesting that current practice and new RA therapies have not closed the gap between CV risk in RA compared to non-RA,” the researchers wrote. The finding also highlighted the potential benefit of considering both LDL-C and HDL-C levels when estimating CV risk in RA,” they said. One potential explanation for the independent significant association between HDL-C levels and CV-risk was that HDL-C levels appeared to remain relatively stable throughout changes in inflammation and may correlate better than LDL-C for CV risk, they suggested.
Overall the findings supported the notion that LDL-C levels may be a suboptimal measure for CV risk assessment in RA and demonstrated the need for improved methods for CV risk assessment, they said. The TransAtlantic cardiovascular risk calculator is under development to address this “major need in optimizing care for RA patients,” they added.
Despite a similar relationship between lipid levels and cardiovascular disease risk in patients with or without rheumatoid arthritis, people with RA have an almost two-fold increased risk of having a major cardiovascular event, research shows.
The findings confirm that existing CV risk calculators are suboptimal for patients with RA and that improved methods of measuring risk in this population are needed, say the study authors from the Brigham and Women’s Hospital in Boston. They also show that there may be potential benefit in considering both LDL-C and HDL-C levels when estimating CV risk in RA, according to Dr. Katherine P. Liao and her associates from Brigham and Women’s Hospital, Boston (Arthritis & Rheumatology 2015; [doi: 10.1002/art.39165]).
While there have been many studies examining the relationship between low density lipoprotein cholesterol levels (LDL-C) and CV risk, only one study described the relationship between high density lipoprotein cholesterol levels (HDL-C) with CV risk among RA patients. It found higher levels of HDL-C were associated with lower CV risk in people with RA, consistent with the general population. “[This] suggests that HDL-C levels may provide important information for estimating CV risk independent of LDL-C,” according to Dr. Liao and her associates.
To assess the relationship between LDL-C and HDL-C with CV risk in RA patients compared with the general population, the researchers compared 16,085 RA patients with 48,499 matched controls without RA from the United Healthcare database. In line with other studies the researchers found evidence of the “lipid paradox” – where low and high LDL-C levels confer a similar CV risk – in both groups.
Looking at HDL-C levels, the risk of a major cardiovascular event (MACE) was highest among patients with lower levels compared with higher levels in both RA and non-RA subjects. For example subjects in the highest quintile had a 55% lower risk for MACE (hazard ratio, 0.45; 95% confidence interval, 0.48-0.72) compared with the lowest quintile after adjusting for CV risk factors. However,in a model that included both LDL-C and HDL-C levels, patients with RA had an elevated risk of MACE (HR 1.7; 95% CI, 1.5-1.9) independent of LDL-C levels.
“This magnitude of increased CV risk is similar to studies published a decade ago, suggesting that current practice and new RA therapies have not closed the gap between CV risk in RA compared to non-RA,” the researchers wrote. The finding also highlighted the potential benefit of considering both LDL-C and HDL-C levels when estimating CV risk in RA,” they said. One potential explanation for the independent significant association between HDL-C levels and CV-risk was that HDL-C levels appeared to remain relatively stable throughout changes in inflammation and may correlate better than LDL-C for CV risk, they suggested.
Overall the findings supported the notion that LDL-C levels may be a suboptimal measure for CV risk assessment in RA and demonstrated the need for improved methods for CV risk assessment, they said. The TransAtlantic cardiovascular risk calculator is under development to address this “major need in optimizing care for RA patients,” they added.
Despite a similar relationship between lipid levels and cardiovascular disease risk in patients with or without rheumatoid arthritis, people with RA have an almost two-fold increased risk of having a major cardiovascular event, research shows.
The findings confirm that existing CV risk calculators are suboptimal for patients with RA and that improved methods of measuring risk in this population are needed, say the study authors from the Brigham and Women’s Hospital in Boston. They also show that there may be potential benefit in considering both LDL-C and HDL-C levels when estimating CV risk in RA, according to Dr. Katherine P. Liao and her associates from Brigham and Women’s Hospital, Boston (Arthritis & Rheumatology 2015; [doi: 10.1002/art.39165]).
While there have been many studies examining the relationship between low density lipoprotein cholesterol levels (LDL-C) and CV risk, only one study described the relationship between high density lipoprotein cholesterol levels (HDL-C) with CV risk among RA patients. It found higher levels of HDL-C were associated with lower CV risk in people with RA, consistent with the general population. “[This] suggests that HDL-C levels may provide important information for estimating CV risk independent of LDL-C,” according to Dr. Liao and her associates.
To assess the relationship between LDL-C and HDL-C with CV risk in RA patients compared with the general population, the researchers compared 16,085 RA patients with 48,499 matched controls without RA from the United Healthcare database. In line with other studies the researchers found evidence of the “lipid paradox” – where low and high LDL-C levels confer a similar CV risk – in both groups.
Looking at HDL-C levels, the risk of a major cardiovascular event (MACE) was highest among patients with lower levels compared with higher levels in both RA and non-RA subjects. For example subjects in the highest quintile had a 55% lower risk for MACE (hazard ratio, 0.45; 95% confidence interval, 0.48-0.72) compared with the lowest quintile after adjusting for CV risk factors. However,in a model that included both LDL-C and HDL-C levels, patients with RA had an elevated risk of MACE (HR 1.7; 95% CI, 1.5-1.9) independent of LDL-C levels.
“This magnitude of increased CV risk is similar to studies published a decade ago, suggesting that current practice and new RA therapies have not closed the gap between CV risk in RA compared to non-RA,” the researchers wrote. The finding also highlighted the potential benefit of considering both LDL-C and HDL-C levels when estimating CV risk in RA,” they said. One potential explanation for the independent significant association between HDL-C levels and CV-risk was that HDL-C levels appeared to remain relatively stable throughout changes in inflammation and may correlate better than LDL-C for CV risk, they suggested.
Overall the findings supported the notion that LDL-C levels may be a suboptimal measure for CV risk assessment in RA and demonstrated the need for improved methods for CV risk assessment, they said. The TransAtlantic cardiovascular risk calculator is under development to address this “major need in optimizing care for RA patients,” they added.
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point: Current CV risk assessments in RA are suboptimal; there may be benefit in considering both LDL-C and HDL-C levels when estimating CV risk in RA.
Major finding: The relationship between lipid levels and CV risk was similar in RA patients and the general population despite a 1.7 fold increased risk of a CV event for RA patients.
Data source: 16,085 RA patients and 48,499 matched controls (3:1 ratio) without RA from the United Healthcare database
Disclosures: The study was funded by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health. The authors declared research grant support from Amgen, Lilly, and Pfizer.
TRACE-RA: Statin therapy may prevent CVD in RA
MANCHESTER, ENGLAND – Results of the Trial of Atorvastatin for the Primary Prevention of Cardiovascular Events in Patients with Rheumatoid Arthritis (TRACE-RA) suggest that daily treatment with atorvastatin may have a primary protective effect on the development of cardiovascular disease in patients with rheumatoid arthritis.
There was a 34% reduction in the primary composite cardiovascular endpoint, which, although not significant, showed a trend for benefit that was similar to that seen with other statin studies in the general population, according to Dr. George Kitas, a consultant rheumatologist and director of research and development at the Dudley (England) Group NHS Foundation Trust.
“The event rate we observed was actually lower than we had calculated on,” Dr. Kitas said in an interview at the British Society for Rheumatology annual conference. The anticipated event rate was 1.8%, but the observed event rate was 0.76%, he explained. As a result, the trial was terminated early as there would not be sufficient events over the planned time course of the trial.
“However, if you correct for a couple of baseline differences, particularly smoking and ethnicity, and also for compliance, the effect on the CVD event rate is actually statistically significant,” Dr. Kitas observed.
He noted that low-density lipoprotein cholesterol (LDL-C) was lowered by a significant 1.07 mmol/L (19.26 mg/dL) in the 1,492 patients randomized to atorvastatin versus a 0.14 mmol/L (2.52 mg/dL) decrease in the 1,492 patients randomized to placebo (P < .001).
Statins are established as a means of both primary and secondary prevention of CVD in the general population, but few patients with RA were included in the seminal statin trials. The aim of TRACE-RA was therefore to look at statins as a possible means for primary prevention in patients with high-grade inflammation such as RA.
TRACE-RA was a prospective, double-blind trial of 2,986 patients with RA aged 50 years or older or who had a disease duration of more than 10 years and who had been recruited at 102 participating centers between 2007 and 2012.
Patients had been randomized to treatment with atorvastatin 40 mg/day or placebo for the primary prevention of cardiovascular events and the median follow-up was 2.53 years, with an overall 7,908 years of patient follow-up. All patients were given lifestyle advice on how to reduce their cardiovascular risk.
The primary endpoint was a composite of CVD death, nonfatal myocardial infarction, cerebrovascular accident (excluding hemorrhagic stroke), transient ischemic attack, hospitalized angina, and coronary and noncoronary revascularization. A total of 33 of these CVD events occurred in 24 atorvastatin-treated patients and 52 events occurred in 36 patients on placebo. Of these, the majority were coronary events.
Adverse event rates in the atorvastatin and placebo arms were a respective 19.7% and 19.5%. “The side effects that were observed in the statin arm were virtually identical to the side effects that were observed in the placebo arm,” Dr. Kitas noted.
“So, what we think we can say from TRACE-RA is firstly that statins are ‘safe’ in people with rheumatoid arthritis. Secondly, that they have virtually identical effects, both in terms of LDL-C reduction and in terms of hard cardiovascular events, as what one would expect with equivalent doses of statins in the general population,” Dr. Kitas said.
“In practical terms, I would suggest that we all assess our patients regularly for their cardiovascular risk and make sure that we do not hesitate to start a statin in those who fulfill criteria according to the relevant national guidelines.”
Commenting on the study, Dr. Ernest Choy of Cardiff University, Wales, said that this is the largest randomized, controlled trial to look at preventing cardiovascular mortality in patients with RA.
“The message is that cardiovascular risk can potentially be reduced by a statin, especially in high-risk patients,” Dr. Choy said, noting that it also “highlights the importance of monitoring for cardiovascular disease.”
TRACE-RA was funded by Arthritis Research UK and the British Heart Foundation. Pfizer UK provided the study medication and an unrestricted grant for the TRACE-RA biobank. Dr. Kitas had no financial conflicts of interest to disclose.
MANCHESTER, ENGLAND – Results of the Trial of Atorvastatin for the Primary Prevention of Cardiovascular Events in Patients with Rheumatoid Arthritis (TRACE-RA) suggest that daily treatment with atorvastatin may have a primary protective effect on the development of cardiovascular disease in patients with rheumatoid arthritis.
There was a 34% reduction in the primary composite cardiovascular endpoint, which, although not significant, showed a trend for benefit that was similar to that seen with other statin studies in the general population, according to Dr. George Kitas, a consultant rheumatologist and director of research and development at the Dudley (England) Group NHS Foundation Trust.
“The event rate we observed was actually lower than we had calculated on,” Dr. Kitas said in an interview at the British Society for Rheumatology annual conference. The anticipated event rate was 1.8%, but the observed event rate was 0.76%, he explained. As a result, the trial was terminated early as there would not be sufficient events over the planned time course of the trial.
“However, if you correct for a couple of baseline differences, particularly smoking and ethnicity, and also for compliance, the effect on the CVD event rate is actually statistically significant,” Dr. Kitas observed.
He noted that low-density lipoprotein cholesterol (LDL-C) was lowered by a significant 1.07 mmol/L (19.26 mg/dL) in the 1,492 patients randomized to atorvastatin versus a 0.14 mmol/L (2.52 mg/dL) decrease in the 1,492 patients randomized to placebo (P < .001).
Statins are established as a means of both primary and secondary prevention of CVD in the general population, but few patients with RA were included in the seminal statin trials. The aim of TRACE-RA was therefore to look at statins as a possible means for primary prevention in patients with high-grade inflammation such as RA.
TRACE-RA was a prospective, double-blind trial of 2,986 patients with RA aged 50 years or older or who had a disease duration of more than 10 years and who had been recruited at 102 participating centers between 2007 and 2012.
Patients had been randomized to treatment with atorvastatin 40 mg/day or placebo for the primary prevention of cardiovascular events and the median follow-up was 2.53 years, with an overall 7,908 years of patient follow-up. All patients were given lifestyle advice on how to reduce their cardiovascular risk.
The primary endpoint was a composite of CVD death, nonfatal myocardial infarction, cerebrovascular accident (excluding hemorrhagic stroke), transient ischemic attack, hospitalized angina, and coronary and noncoronary revascularization. A total of 33 of these CVD events occurred in 24 atorvastatin-treated patients and 52 events occurred in 36 patients on placebo. Of these, the majority were coronary events.
Adverse event rates in the atorvastatin and placebo arms were a respective 19.7% and 19.5%. “The side effects that were observed in the statin arm were virtually identical to the side effects that were observed in the placebo arm,” Dr. Kitas noted.
“So, what we think we can say from TRACE-RA is firstly that statins are ‘safe’ in people with rheumatoid arthritis. Secondly, that they have virtually identical effects, both in terms of LDL-C reduction and in terms of hard cardiovascular events, as what one would expect with equivalent doses of statins in the general population,” Dr. Kitas said.
“In practical terms, I would suggest that we all assess our patients regularly for their cardiovascular risk and make sure that we do not hesitate to start a statin in those who fulfill criteria according to the relevant national guidelines.”
Commenting on the study, Dr. Ernest Choy of Cardiff University, Wales, said that this is the largest randomized, controlled trial to look at preventing cardiovascular mortality in patients with RA.
“The message is that cardiovascular risk can potentially be reduced by a statin, especially in high-risk patients,” Dr. Choy said, noting that it also “highlights the importance of monitoring for cardiovascular disease.”
TRACE-RA was funded by Arthritis Research UK and the British Heart Foundation. Pfizer UK provided the study medication and an unrestricted grant for the TRACE-RA biobank. Dr. Kitas had no financial conflicts of interest to disclose.
MANCHESTER, ENGLAND – Results of the Trial of Atorvastatin for the Primary Prevention of Cardiovascular Events in Patients with Rheumatoid Arthritis (TRACE-RA) suggest that daily treatment with atorvastatin may have a primary protective effect on the development of cardiovascular disease in patients with rheumatoid arthritis.
There was a 34% reduction in the primary composite cardiovascular endpoint, which, although not significant, showed a trend for benefit that was similar to that seen with other statin studies in the general population, according to Dr. George Kitas, a consultant rheumatologist and director of research and development at the Dudley (England) Group NHS Foundation Trust.
“The event rate we observed was actually lower than we had calculated on,” Dr. Kitas said in an interview at the British Society for Rheumatology annual conference. The anticipated event rate was 1.8%, but the observed event rate was 0.76%, he explained. As a result, the trial was terminated early as there would not be sufficient events over the planned time course of the trial.
“However, if you correct for a couple of baseline differences, particularly smoking and ethnicity, and also for compliance, the effect on the CVD event rate is actually statistically significant,” Dr. Kitas observed.
He noted that low-density lipoprotein cholesterol (LDL-C) was lowered by a significant 1.07 mmol/L (19.26 mg/dL) in the 1,492 patients randomized to atorvastatin versus a 0.14 mmol/L (2.52 mg/dL) decrease in the 1,492 patients randomized to placebo (P < .001).
Statins are established as a means of both primary and secondary prevention of CVD in the general population, but few patients with RA were included in the seminal statin trials. The aim of TRACE-RA was therefore to look at statins as a possible means for primary prevention in patients with high-grade inflammation such as RA.
TRACE-RA was a prospective, double-blind trial of 2,986 patients with RA aged 50 years or older or who had a disease duration of more than 10 years and who had been recruited at 102 participating centers between 2007 and 2012.
Patients had been randomized to treatment with atorvastatin 40 mg/day or placebo for the primary prevention of cardiovascular events and the median follow-up was 2.53 years, with an overall 7,908 years of patient follow-up. All patients were given lifestyle advice on how to reduce their cardiovascular risk.
The primary endpoint was a composite of CVD death, nonfatal myocardial infarction, cerebrovascular accident (excluding hemorrhagic stroke), transient ischemic attack, hospitalized angina, and coronary and noncoronary revascularization. A total of 33 of these CVD events occurred in 24 atorvastatin-treated patients and 52 events occurred in 36 patients on placebo. Of these, the majority were coronary events.
Adverse event rates in the atorvastatin and placebo arms were a respective 19.7% and 19.5%. “The side effects that were observed in the statin arm were virtually identical to the side effects that were observed in the placebo arm,” Dr. Kitas noted.
“So, what we think we can say from TRACE-RA is firstly that statins are ‘safe’ in people with rheumatoid arthritis. Secondly, that they have virtually identical effects, both in terms of LDL-C reduction and in terms of hard cardiovascular events, as what one would expect with equivalent doses of statins in the general population,” Dr. Kitas said.
“In practical terms, I would suggest that we all assess our patients regularly for their cardiovascular risk and make sure that we do not hesitate to start a statin in those who fulfill criteria according to the relevant national guidelines.”
Commenting on the study, Dr. Ernest Choy of Cardiff University, Wales, said that this is the largest randomized, controlled trial to look at preventing cardiovascular mortality in patients with RA.
“The message is that cardiovascular risk can potentially be reduced by a statin, especially in high-risk patients,” Dr. Choy said, noting that it also “highlights the importance of monitoring for cardiovascular disease.”
TRACE-RA was funded by Arthritis Research UK and the British Heart Foundation. Pfizer UK provided the study medication and an unrestricted grant for the TRACE-RA biobank. Dr. Kitas had no financial conflicts of interest to disclose.
AT RHEUMATOLOGY 2015
Key clinical point: Statin therapy may be as beneficial in patients with RA as it is in the general population for the primary prevention of cardiovascular events.
Major finding: The hazard ratio for the reduction in the primary composite cardiovascular endpoint was 0.66 (P = .0119).
Data source: Multicenter, double-blind trial of 2,986 patients with RA aged older than 50 years or with more than 10 years’ disease duration randomized to treatment with atorvastatin 40 mg/day or placebo for the primary prevention of cardiovascular events.
Disclosures: TRACE-RA was funded by Arthritis Research UK and the British Heart Foundation. Pfizer UK provided the study medication and an unrestricted grant for the TRACE-RA biobank. Dr. Kitas had no financial conflicts of interest to disclose.
RA susceptibility gene linked with disease severity, treatment response
Specific haplotypes of the of the HLA-DRB1 gene – well known to confer increased risk for rheumatoid arthritis – proved to be strong, independent predictors of radiological disease severity, risk of death, and response to tumor necrosis factor inhibitor therapy in a three-cohort study involving more than 6,000 patients.
The report represents the first analysis to find significant predictors for those outcomes based on 16 haplotypes that are derived from three amino acid positions of the HLA-DRB1 gene. Two of those amino acid positions, 71 and 74, are within the classic shared epitope, which has been “associated with the development of anticitrullinated protein antibodies and has been consistently associated with markers of severe disease, such as radiological joint damage and mortality in patients with RA,” wrote Dr. Sebastien Viatte of the University of Manchester (England) and his coauthors (JAMA 2015;313:1645-56).
The third amino acid position examined in the study, position 11, which lies outside the shared epitope, was associated with increased risk for radiological damage, mortality, and response to tumor necrosis factor (TNF) inhibitor treatment, the latter of which neither position 71 or 74 was associated with.
To find these significant predictors of prognosis, the investigators examined the three amino acid positions in a large prospective cohort of patients with inflammatory polyarthritis or RA who were recruited at disease onset in the Norfolk Arthritis Register. They then independently validated the results for mortality and radiological damage in a replication cohort of patients from the Early Rheumatoid Arthritis Study study and for treatment response in patients from the Biologics in Rheumatoid Arthritis Genetics and Genomics Study Syndicate cohort. All the patients were from the United Kingdom and had self-reported white ancestry.
Overall, radiographic outcome was associated with amino acid positions 11, 71, and 74 independently of each other and “completely superseded the shared epitope model.” Analyses of the effects of the three amino acid positions on all-cause mortality showed similar results. Change in the 28-joint Disease Activity Score or European League Against Rheumatism response was not associated with anticitrullinated protein antibodies or the shared epitope, but was significantly associated with valine at position 11.
Valine at position 11 consistently had the strongest effects on increasing risk for radiographic damage, mortality, and better TNF inhibitor treatment response. Patients with RA and valine at position 11 of HLA-DRB1 had the strongest association with radiological damage (odds ratio, 1.75; 95% confidence interval, 1.51-2.05) as measured by Larsen score. “Valine at [position 11] represents what we believe is the strongest single genetic association with radiographic damage identified to date,” they wrote.
Valine at position 11 also was associated with higher all-cause mortality in patients with inflammatory polyarthritis (hazard ratio, 1.16; 95% CI, 1.03-1.31; P = .01) and with better EULAR response to TNF inhibitor therapy (OR, 1.14; 95% CI, 1.01-1.30; P = .04).
The investigators found that the 16 haplotypes that existed in the cohorts based on the amino acid positions at 11, 71, and 74 of HLA-DRB1 could be used to construct a hierarchy of risk for the three outcome measures. For instance, they noted that the valine-containing VKA haplotype was associated with RA susceptibility (OR, 4.44; 95% CI, 4.02-4.91), joint erosions in patients with RA (OR, 1.82; 95% CI, 1.35-2.46), and a better response to TNF inhibitor drugs (OR, 1.23; 95% CI, 1.06-1.43). Although a 23% increase in the odds of good response to TNF inhibitors is “modest,” the researchers wrote, “it refers to the likelihood of moving from the EULAR [European League Against Rheumatism] category of none to a moderate response, or from a moderate to a good response for every copy of the VKA haplotype carried by the patient, compared with noncarriers.
“Replication of these findings in other cohorts is needed as a next step in evaluating the role of HLA-DRBl haplotype analysis for management of RA,” the authors concluded.
The study was funded by a grant from Arthritis Research UK. Several authors reported receiving fees or grant support from companies that market drugs for RA.
Although the findings of Dr. Viatte and his colleagues do not have immediate clinical implications, identification of the precise HLA variants that influence disease course is of “great interest.”
The findings add to the ability to predict outcomes of RA, which could help optimize therapeutic strategies for different patients. They also shed light on the molecular mechanisms that determine disease course and mortality.

|
Dr. David T. Felson |
However, the increased risk of disease severity conferred by these HLA-DRB1 haplotypes defined by amino acids at positions 11, 71, and 74 has only a minor influence on disease outcomes in comparison to the larger explanatory power of environmental and lifestyle factors and also biomarkers. The best use of this data in predicting RA outcomes and optimizing treatment plans will be in updated prediction algorithms.
Dr. David T. Felson is with the clinical epidemiology research and training unit at Boston University and the National Institute for Health Research biomedical research unit at the University of Manchester (England). Dr. Lars Klareskog is with the rheumatology unit at Karolinska Institute in Stockholm. Their comments are taken from an editorial that accompanied Dr. Viatte and colleagues’ paper (JAMA 2015;313:1623-4). They reported having no conflicts of interest.
Although the findings of Dr. Viatte and his colleagues do not have immediate clinical implications, identification of the precise HLA variants that influence disease course is of “great interest.”
The findings add to the ability to predict outcomes of RA, which could help optimize therapeutic strategies for different patients. They also shed light on the molecular mechanisms that determine disease course and mortality.

|
Dr. David T. Felson |
However, the increased risk of disease severity conferred by these HLA-DRB1 haplotypes defined by amino acids at positions 11, 71, and 74 has only a minor influence on disease outcomes in comparison to the larger explanatory power of environmental and lifestyle factors and also biomarkers. The best use of this data in predicting RA outcomes and optimizing treatment plans will be in updated prediction algorithms.
Dr. David T. Felson is with the clinical epidemiology research and training unit at Boston University and the National Institute for Health Research biomedical research unit at the University of Manchester (England). Dr. Lars Klareskog is with the rheumatology unit at Karolinska Institute in Stockholm. Their comments are taken from an editorial that accompanied Dr. Viatte and colleagues’ paper (JAMA 2015;313:1623-4). They reported having no conflicts of interest.
Although the findings of Dr. Viatte and his colleagues do not have immediate clinical implications, identification of the precise HLA variants that influence disease course is of “great interest.”
The findings add to the ability to predict outcomes of RA, which could help optimize therapeutic strategies for different patients. They also shed light on the molecular mechanisms that determine disease course and mortality.

|
Dr. David T. Felson |
However, the increased risk of disease severity conferred by these HLA-DRB1 haplotypes defined by amino acids at positions 11, 71, and 74 has only a minor influence on disease outcomes in comparison to the larger explanatory power of environmental and lifestyle factors and also biomarkers. The best use of this data in predicting RA outcomes and optimizing treatment plans will be in updated prediction algorithms.
Dr. David T. Felson is with the clinical epidemiology research and training unit at Boston University and the National Institute for Health Research biomedical research unit at the University of Manchester (England). Dr. Lars Klareskog is with the rheumatology unit at Karolinska Institute in Stockholm. Their comments are taken from an editorial that accompanied Dr. Viatte and colleagues’ paper (JAMA 2015;313:1623-4). They reported having no conflicts of interest.
Specific haplotypes of the of the HLA-DRB1 gene – well known to confer increased risk for rheumatoid arthritis – proved to be strong, independent predictors of radiological disease severity, risk of death, and response to tumor necrosis factor inhibitor therapy in a three-cohort study involving more than 6,000 patients.
The report represents the first analysis to find significant predictors for those outcomes based on 16 haplotypes that are derived from three amino acid positions of the HLA-DRB1 gene. Two of those amino acid positions, 71 and 74, are within the classic shared epitope, which has been “associated with the development of anticitrullinated protein antibodies and has been consistently associated with markers of severe disease, such as radiological joint damage and mortality in patients with RA,” wrote Dr. Sebastien Viatte of the University of Manchester (England) and his coauthors (JAMA 2015;313:1645-56).
The third amino acid position examined in the study, position 11, which lies outside the shared epitope, was associated with increased risk for radiological damage, mortality, and response to tumor necrosis factor (TNF) inhibitor treatment, the latter of which neither position 71 or 74 was associated with.
To find these significant predictors of prognosis, the investigators examined the three amino acid positions in a large prospective cohort of patients with inflammatory polyarthritis or RA who were recruited at disease onset in the Norfolk Arthritis Register. They then independently validated the results for mortality and radiological damage in a replication cohort of patients from the Early Rheumatoid Arthritis Study study and for treatment response in patients from the Biologics in Rheumatoid Arthritis Genetics and Genomics Study Syndicate cohort. All the patients were from the United Kingdom and had self-reported white ancestry.
Overall, radiographic outcome was associated with amino acid positions 11, 71, and 74 independently of each other and “completely superseded the shared epitope model.” Analyses of the effects of the three amino acid positions on all-cause mortality showed similar results. Change in the 28-joint Disease Activity Score or European League Against Rheumatism response was not associated with anticitrullinated protein antibodies or the shared epitope, but was significantly associated with valine at position 11.
Valine at position 11 consistently had the strongest effects on increasing risk for radiographic damage, mortality, and better TNF inhibitor treatment response. Patients with RA and valine at position 11 of HLA-DRB1 had the strongest association with radiological damage (odds ratio, 1.75; 95% confidence interval, 1.51-2.05) as measured by Larsen score. “Valine at [position 11] represents what we believe is the strongest single genetic association with radiographic damage identified to date,” they wrote.
Valine at position 11 also was associated with higher all-cause mortality in patients with inflammatory polyarthritis (hazard ratio, 1.16; 95% CI, 1.03-1.31; P = .01) and with better EULAR response to TNF inhibitor therapy (OR, 1.14; 95% CI, 1.01-1.30; P = .04).
The investigators found that the 16 haplotypes that existed in the cohorts based on the amino acid positions at 11, 71, and 74 of HLA-DRB1 could be used to construct a hierarchy of risk for the three outcome measures. For instance, they noted that the valine-containing VKA haplotype was associated with RA susceptibility (OR, 4.44; 95% CI, 4.02-4.91), joint erosions in patients with RA (OR, 1.82; 95% CI, 1.35-2.46), and a better response to TNF inhibitor drugs (OR, 1.23; 95% CI, 1.06-1.43). Although a 23% increase in the odds of good response to TNF inhibitors is “modest,” the researchers wrote, “it refers to the likelihood of moving from the EULAR [European League Against Rheumatism] category of none to a moderate response, or from a moderate to a good response for every copy of the VKA haplotype carried by the patient, compared with noncarriers.
“Replication of these findings in other cohorts is needed as a next step in evaluating the role of HLA-DRBl haplotype analysis for management of RA,” the authors concluded.
The study was funded by a grant from Arthritis Research UK. Several authors reported receiving fees or grant support from companies that market drugs for RA.
Specific haplotypes of the of the HLA-DRB1 gene – well known to confer increased risk for rheumatoid arthritis – proved to be strong, independent predictors of radiological disease severity, risk of death, and response to tumor necrosis factor inhibitor therapy in a three-cohort study involving more than 6,000 patients.
The report represents the first analysis to find significant predictors for those outcomes based on 16 haplotypes that are derived from three amino acid positions of the HLA-DRB1 gene. Two of those amino acid positions, 71 and 74, are within the classic shared epitope, which has been “associated with the development of anticitrullinated protein antibodies and has been consistently associated with markers of severe disease, such as radiological joint damage and mortality in patients with RA,” wrote Dr. Sebastien Viatte of the University of Manchester (England) and his coauthors (JAMA 2015;313:1645-56).
The third amino acid position examined in the study, position 11, which lies outside the shared epitope, was associated with increased risk for radiological damage, mortality, and response to tumor necrosis factor (TNF) inhibitor treatment, the latter of which neither position 71 or 74 was associated with.
To find these significant predictors of prognosis, the investigators examined the three amino acid positions in a large prospective cohort of patients with inflammatory polyarthritis or RA who were recruited at disease onset in the Norfolk Arthritis Register. They then independently validated the results for mortality and radiological damage in a replication cohort of patients from the Early Rheumatoid Arthritis Study study and for treatment response in patients from the Biologics in Rheumatoid Arthritis Genetics and Genomics Study Syndicate cohort. All the patients were from the United Kingdom and had self-reported white ancestry.
Overall, radiographic outcome was associated with amino acid positions 11, 71, and 74 independently of each other and “completely superseded the shared epitope model.” Analyses of the effects of the three amino acid positions on all-cause mortality showed similar results. Change in the 28-joint Disease Activity Score or European League Against Rheumatism response was not associated with anticitrullinated protein antibodies or the shared epitope, but was significantly associated with valine at position 11.
Valine at position 11 consistently had the strongest effects on increasing risk for radiographic damage, mortality, and better TNF inhibitor treatment response. Patients with RA and valine at position 11 of HLA-DRB1 had the strongest association with radiological damage (odds ratio, 1.75; 95% confidence interval, 1.51-2.05) as measured by Larsen score. “Valine at [position 11] represents what we believe is the strongest single genetic association with radiographic damage identified to date,” they wrote.
Valine at position 11 also was associated with higher all-cause mortality in patients with inflammatory polyarthritis (hazard ratio, 1.16; 95% CI, 1.03-1.31; P = .01) and with better EULAR response to TNF inhibitor therapy (OR, 1.14; 95% CI, 1.01-1.30; P = .04).
The investigators found that the 16 haplotypes that existed in the cohorts based on the amino acid positions at 11, 71, and 74 of HLA-DRB1 could be used to construct a hierarchy of risk for the three outcome measures. For instance, they noted that the valine-containing VKA haplotype was associated with RA susceptibility (OR, 4.44; 95% CI, 4.02-4.91), joint erosions in patients with RA (OR, 1.82; 95% CI, 1.35-2.46), and a better response to TNF inhibitor drugs (OR, 1.23; 95% CI, 1.06-1.43). Although a 23% increase in the odds of good response to TNF inhibitors is “modest,” the researchers wrote, “it refers to the likelihood of moving from the EULAR [European League Against Rheumatism] category of none to a moderate response, or from a moderate to a good response for every copy of the VKA haplotype carried by the patient, compared with noncarriers.
“Replication of these findings in other cohorts is needed as a next step in evaluating the role of HLA-DRBl haplotype analysis for management of RA,” the authors concluded.
The study was funded by a grant from Arthritis Research UK. Several authors reported receiving fees or grant support from companies that market drugs for RA.
FROM JAMA
Key clinical finding: The HLA-DRB1 haplotype status of patients with RA may affect radiological damage, mortality, and response to anti-TNF agents.
Major finding: Patients with RA and valine at position 11 of HLA-DRB1 had the strongest association with radiological damage (odds ratio, 1.75; 95% confidence interval, 1.51-2.05) as measured by Larsen score.
Data source: More than 6,000 RA patients from the Norfolk Arthritis Registry, Early Rheumatoid Arthritis Study, and the Biologics in Rheumatoid Arthritis Genetics and Genomics Study Syndicate cohort.
Disclosures: The study was funded by a grant from Arthritis Research UK. Several authors reported receiving fees or grant support from companies that market drugs for RA.
Etanercept maintenance therapy feasible in some RA patients
Rheumatoid arthritis patients with low disease activity are less likely to flare if they continue their treatment with etanercept and methotrexate, even if they take half the dose of the anti-TNF agent, a double-blind, randomized trial showed.
The researchers conducting the trial, called DOSERA (Dose Reduction or Discontinuation of Etanercept in Methotrexate-Treated Rheumatoid Arthritis Patients Who Have Achieved a Stable Low Disease Activity-State), led by Dr. Ronald F. van Vollenhoven of the Karolinska Institute in Stockholm, reported that the proportion of patients who successfully maintained low disease activity was significantly higher with etanercept 50 mg (52%; P = .007) and 25 mg (44%; P = .044), compared with placebo (13%). They defined failure as an increase in 28-joint Disease Activity Score (DAS28) of 0.6 or more and an absolute value of greater than 3.2 or disease progression as determined by investigator/patient assessment.
According to the authors, the results are important because, theoretically, they represent the first controlled demonstration that an induction maintenance strategy can be applied in some patients with established RA for whom the use of an anti-TNF agent is clinically necessary.
From a clinical practice point of view, the results also suggest that it might be possible to reduce dosages in some patients while maintaining the same favorable disease state.
“However, a full assessment of this possibility would require considerably more robust data in a larger population on the full clinical as well as radiographic effects, both short term and long term,” Dr. van Vollenhoven and his associates wrote, adding that “it is not possible to know whether these data would apply equally to other anti-TNF agents.”
Median time to flare was significantly shorter for patients taking methotrexate plus placebo at 6 weeks, compared with etanercept 50 mg at 48 weeks (P = .001) and 25 mg at 36 weeks (P < .001), the researchers reported (Ann. Rheum. Dis. 2015 April 14 [doi:10.1136/annrheumdis-2014-205726]).
Most patients (91%) who flared and then restarted open-label etanercept 50 mg/week responded promptly, with median times until low disease activity or remission being 6.0 weeks for the 50-mg group, 5.9 weeks for the 25-mg group, and 3.9 weeks for the placebo group.
Adverse events were consistent with the patient population and known side effects of etanercept and methotrexate.
The study had several weaknesses, including its limited size and associated wide confidence intervals, the researchers noted, resulting in “uncertainty regarding the comparison of the clinical efficacies of full-dose and reduced-dose [etanercept] continuation.”
Etanercept also is not approved at doses other than 50 mg for the treatment of adults with RA. A dose reduction was therefore not supported by the product label and also entails some practical difficulties, Dr. van Vollenhoven and his associates added.
The study included 91 patients who had been receiving etanercept, 50 mg/week, plus methotrexate, 7.5-25 mg/week for at least 14 months and had a DAS28 score of 3.2 or lower.
After an 8-week run-in phase, 73 patients were randomized in a double-blind design to etanercept 50 mg/week plus methotrexate, etanercept 25 mg/week plus methotrexate, or placebo plus methotrexate for 48 weeks.
The study was sponsored by Wyeth, which was acquired by Pfizer. The investigators designed the study protocol prior to the agreement by Wyeth to support the study. Some of the authors reported receiving research support and/or honoraria from numerous pharmaceutical companies or serving as consultants to them. Several reported being employees of Wyeth or Pfizer at the time of the study.
Rheumatoid arthritis patients with low disease activity are less likely to flare if they continue their treatment with etanercept and methotrexate, even if they take half the dose of the anti-TNF agent, a double-blind, randomized trial showed.
The researchers conducting the trial, called DOSERA (Dose Reduction or Discontinuation of Etanercept in Methotrexate-Treated Rheumatoid Arthritis Patients Who Have Achieved a Stable Low Disease Activity-State), led by Dr. Ronald F. van Vollenhoven of the Karolinska Institute in Stockholm, reported that the proportion of patients who successfully maintained low disease activity was significantly higher with etanercept 50 mg (52%; P = .007) and 25 mg (44%; P = .044), compared with placebo (13%). They defined failure as an increase in 28-joint Disease Activity Score (DAS28) of 0.6 or more and an absolute value of greater than 3.2 or disease progression as determined by investigator/patient assessment.
According to the authors, the results are important because, theoretically, they represent the first controlled demonstration that an induction maintenance strategy can be applied in some patients with established RA for whom the use of an anti-TNF agent is clinically necessary.
From a clinical practice point of view, the results also suggest that it might be possible to reduce dosages in some patients while maintaining the same favorable disease state.
“However, a full assessment of this possibility would require considerably more robust data in a larger population on the full clinical as well as radiographic effects, both short term and long term,” Dr. van Vollenhoven and his associates wrote, adding that “it is not possible to know whether these data would apply equally to other anti-TNF agents.”
Median time to flare was significantly shorter for patients taking methotrexate plus placebo at 6 weeks, compared with etanercept 50 mg at 48 weeks (P = .001) and 25 mg at 36 weeks (P < .001), the researchers reported (Ann. Rheum. Dis. 2015 April 14 [doi:10.1136/annrheumdis-2014-205726]).
Most patients (91%) who flared and then restarted open-label etanercept 50 mg/week responded promptly, with median times until low disease activity or remission being 6.0 weeks for the 50-mg group, 5.9 weeks for the 25-mg group, and 3.9 weeks for the placebo group.
Adverse events were consistent with the patient population and known side effects of etanercept and methotrexate.
The study had several weaknesses, including its limited size and associated wide confidence intervals, the researchers noted, resulting in “uncertainty regarding the comparison of the clinical efficacies of full-dose and reduced-dose [etanercept] continuation.”
Etanercept also is not approved at doses other than 50 mg for the treatment of adults with RA. A dose reduction was therefore not supported by the product label and also entails some practical difficulties, Dr. van Vollenhoven and his associates added.
The study included 91 patients who had been receiving etanercept, 50 mg/week, plus methotrexate, 7.5-25 mg/week for at least 14 months and had a DAS28 score of 3.2 or lower.
After an 8-week run-in phase, 73 patients were randomized in a double-blind design to etanercept 50 mg/week plus methotrexate, etanercept 25 mg/week plus methotrexate, or placebo plus methotrexate for 48 weeks.
The study was sponsored by Wyeth, which was acquired by Pfizer. The investigators designed the study protocol prior to the agreement by Wyeth to support the study. Some of the authors reported receiving research support and/or honoraria from numerous pharmaceutical companies or serving as consultants to them. Several reported being employees of Wyeth or Pfizer at the time of the study.
Rheumatoid arthritis patients with low disease activity are less likely to flare if they continue their treatment with etanercept and methotrexate, even if they take half the dose of the anti-TNF agent, a double-blind, randomized trial showed.
The researchers conducting the trial, called DOSERA (Dose Reduction or Discontinuation of Etanercept in Methotrexate-Treated Rheumatoid Arthritis Patients Who Have Achieved a Stable Low Disease Activity-State), led by Dr. Ronald F. van Vollenhoven of the Karolinska Institute in Stockholm, reported that the proportion of patients who successfully maintained low disease activity was significantly higher with etanercept 50 mg (52%; P = .007) and 25 mg (44%; P = .044), compared with placebo (13%). They defined failure as an increase in 28-joint Disease Activity Score (DAS28) of 0.6 or more and an absolute value of greater than 3.2 or disease progression as determined by investigator/patient assessment.
According to the authors, the results are important because, theoretically, they represent the first controlled demonstration that an induction maintenance strategy can be applied in some patients with established RA for whom the use of an anti-TNF agent is clinically necessary.
From a clinical practice point of view, the results also suggest that it might be possible to reduce dosages in some patients while maintaining the same favorable disease state.
“However, a full assessment of this possibility would require considerably more robust data in a larger population on the full clinical as well as radiographic effects, both short term and long term,” Dr. van Vollenhoven and his associates wrote, adding that “it is not possible to know whether these data would apply equally to other anti-TNF agents.”
Median time to flare was significantly shorter for patients taking methotrexate plus placebo at 6 weeks, compared with etanercept 50 mg at 48 weeks (P = .001) and 25 mg at 36 weeks (P < .001), the researchers reported (Ann. Rheum. Dis. 2015 April 14 [doi:10.1136/annrheumdis-2014-205726]).
Most patients (91%) who flared and then restarted open-label etanercept 50 mg/week responded promptly, with median times until low disease activity or remission being 6.0 weeks for the 50-mg group, 5.9 weeks for the 25-mg group, and 3.9 weeks for the placebo group.
Adverse events were consistent with the patient population and known side effects of etanercept and methotrexate.
The study had several weaknesses, including its limited size and associated wide confidence intervals, the researchers noted, resulting in “uncertainty regarding the comparison of the clinical efficacies of full-dose and reduced-dose [etanercept] continuation.”
Etanercept also is not approved at doses other than 50 mg for the treatment of adults with RA. A dose reduction was therefore not supported by the product label and also entails some practical difficulties, Dr. van Vollenhoven and his associates added.
The study included 91 patients who had been receiving etanercept, 50 mg/week, plus methotrexate, 7.5-25 mg/week for at least 14 months and had a DAS28 score of 3.2 or lower.
After an 8-week run-in phase, 73 patients were randomized in a double-blind design to etanercept 50 mg/week plus methotrexate, etanercept 25 mg/week plus methotrexate, or placebo plus methotrexate for 48 weeks.
The study was sponsored by Wyeth, which was acquired by Pfizer. The investigators designed the study protocol prior to the agreement by Wyeth to support the study. Some of the authors reported receiving research support and/or honoraria from numerous pharmaceutical companies or serving as consultants to them. Several reported being employees of Wyeth or Pfizer at the time of the study.
FROM ANNALS OF THE RHEUMATIC DISEASES
Key clinical point: An induction maintenance strategy with etanercept can be applied in some patients with established RA for whom the use of an anti-TNF agent is clinically necessary.
Major Finding: The proportion of patients who successfully maintained low disease activity was significantly higher with etanercept 50 mg (52%; P = .007) and 25 mg (44%; P = .044), compared with placebo (13%).
Data Source: A double-blind, randomized study of 73 RA patients with low disease activity.
Disclosures: The study was sponsored by Wyeth, which was acquired by Pfizer. The investigators designed the study protocol prior to the agreement by Wyeth to support the study. Some of the authors reported receiving research support and/or honoraria from numerous pharmaceutical companies or serving as consultants to them. Several reported being employees of Wyeth or Pfizer at the time of the study.
Contamination prompts voluntary injectables recall
Mylan N.V. has announced a voluntary recall of specific lots of injectable products for conditions including rheumatoid arthritis, severe psoriasis, lung cancer, breast cancer, ovarian cancer, and acute nonlymphocytic leukemia because foreign particles were observed during testing of retention samples, according to a statement issued by the company on April 23.
The specific lot numbers can be found on the company’s website.
Affected products include methotrexate injection, USP 25 mg/mL, which is indicated for adult rheumatoid arthritis, severe psoriasis, and some neoplastic diseases. According to Mylan, the contaminated lot was distributed in the United States between Jan. 16, 2014, and March 25, 2014.
Other recalled injectable products include gemcitabine, USP 200 mg, an intravenous product for ovarian cancer, breast cancer, non–small cell lung cancer, and pancreatic cancer; carboplatin 10 mg/mL, indicated for advanced ovarian cancer; and cytarabine, indicated for remission induction in acute nonlymphocytic leukemia in adults and children. The lots for these products were distributed in the United States in various periods of several months during 2014.
The recall is being conducted with the knowledge of the Food and Drug Administration, and any adverse events or quality problems should be reported to the FDA’s MedWatch adverse event reporting program.
Mylan N.V. has announced a voluntary recall of specific lots of injectable products for conditions including rheumatoid arthritis, severe psoriasis, lung cancer, breast cancer, ovarian cancer, and acute nonlymphocytic leukemia because foreign particles were observed during testing of retention samples, according to a statement issued by the company on April 23.
The specific lot numbers can be found on the company’s website.
Affected products include methotrexate injection, USP 25 mg/mL, which is indicated for adult rheumatoid arthritis, severe psoriasis, and some neoplastic diseases. According to Mylan, the contaminated lot was distributed in the United States between Jan. 16, 2014, and March 25, 2014.
Other recalled injectable products include gemcitabine, USP 200 mg, an intravenous product for ovarian cancer, breast cancer, non–small cell lung cancer, and pancreatic cancer; carboplatin 10 mg/mL, indicated for advanced ovarian cancer; and cytarabine, indicated for remission induction in acute nonlymphocytic leukemia in adults and children. The lots for these products were distributed in the United States in various periods of several months during 2014.
The recall is being conducted with the knowledge of the Food and Drug Administration, and any adverse events or quality problems should be reported to the FDA’s MedWatch adverse event reporting program.
Mylan N.V. has announced a voluntary recall of specific lots of injectable products for conditions including rheumatoid arthritis, severe psoriasis, lung cancer, breast cancer, ovarian cancer, and acute nonlymphocytic leukemia because foreign particles were observed during testing of retention samples, according to a statement issued by the company on April 23.
The specific lot numbers can be found on the company’s website.
Affected products include methotrexate injection, USP 25 mg/mL, which is indicated for adult rheumatoid arthritis, severe psoriasis, and some neoplastic diseases. According to Mylan, the contaminated lot was distributed in the United States between Jan. 16, 2014, and March 25, 2014.
Other recalled injectable products include gemcitabine, USP 200 mg, an intravenous product for ovarian cancer, breast cancer, non–small cell lung cancer, and pancreatic cancer; carboplatin 10 mg/mL, indicated for advanced ovarian cancer; and cytarabine, indicated for remission induction in acute nonlymphocytic leukemia in adults and children. The lots for these products were distributed in the United States in various periods of several months during 2014.
The recall is being conducted with the knowledge of the Food and Drug Administration, and any adverse events or quality problems should be reported to the FDA’s MedWatch adverse event reporting program.
Epitope from ACPA derived from RA patients common in nature
The monoclonal anti–citrullinated protein antibody CCP-Ab1 present in patients with rheumatoid arthritis cross-reacts with both various autoantigens and many plant and microbial proteins, according to Dr. Reina Tsuda and associates.
The investigators determined that the essential epitope specific for CCP-Ab1 included six essential amino acid residues. They found that this epitope was shared by 38 human proteins, 56 viral proteins, 1,383 fungal proteins, 547 bacterial proteins, and 1,072 plant proteins. CCP-Ab1 reacted successfully with all of the epitope-sharing proteins, including proteins found in the environment, such as proteins consumed from plants as part of a regular diet.
While the wide variety of environmental factors suggest that RA should be more common, the generation of ACPA requires at least two prerequisite risk factors, and “citrullinated peptides more strongly interact with HLA-DR4 molecules and trigger strong CD4+ T cells activation. Thus, a minor population of subjects who have multiple risk factors may develop RA,” the investigators noted.
The full study is available in Arthritis & Rheumatology (doi:10.1002/art.39161).
The monoclonal anti–citrullinated protein antibody CCP-Ab1 present in patients with rheumatoid arthritis cross-reacts with both various autoantigens and many plant and microbial proteins, according to Dr. Reina Tsuda and associates.
The investigators determined that the essential epitope specific for CCP-Ab1 included six essential amino acid residues. They found that this epitope was shared by 38 human proteins, 56 viral proteins, 1,383 fungal proteins, 547 bacterial proteins, and 1,072 plant proteins. CCP-Ab1 reacted successfully with all of the epitope-sharing proteins, including proteins found in the environment, such as proteins consumed from plants as part of a regular diet.
While the wide variety of environmental factors suggest that RA should be more common, the generation of ACPA requires at least two prerequisite risk factors, and “citrullinated peptides more strongly interact with HLA-DR4 molecules and trigger strong CD4+ T cells activation. Thus, a minor population of subjects who have multiple risk factors may develop RA,” the investigators noted.
The full study is available in Arthritis & Rheumatology (doi:10.1002/art.39161).
The monoclonal anti–citrullinated protein antibody CCP-Ab1 present in patients with rheumatoid arthritis cross-reacts with both various autoantigens and many plant and microbial proteins, according to Dr. Reina Tsuda and associates.
The investigators determined that the essential epitope specific for CCP-Ab1 included six essential amino acid residues. They found that this epitope was shared by 38 human proteins, 56 viral proteins, 1,383 fungal proteins, 547 bacterial proteins, and 1,072 plant proteins. CCP-Ab1 reacted successfully with all of the epitope-sharing proteins, including proteins found in the environment, such as proteins consumed from plants as part of a regular diet.
While the wide variety of environmental factors suggest that RA should be more common, the generation of ACPA requires at least two prerequisite risk factors, and “citrullinated peptides more strongly interact with HLA-DR4 molecules and trigger strong CD4+ T cells activation. Thus, a minor population of subjects who have multiple risk factors may develop RA,” the investigators noted.
The full study is available in Arthritis & Rheumatology (doi:10.1002/art.39161).