User login
Launch of adalimumab biosimilar Amjevita postponed
Amgen, maker of the adalimumab biosimilar Amjevita (adalimumab-atto) has reached an agreement with AbbVie, manufacturer of the originator adalimumab Humira, that halts marketing of Amjevita in the United States until 2023 and in Europe until 2018, according to a company statement.
The deal between the two manufacturers settles a patent infringement lawsuit that AbbVie brought against Amgen after it received Food and Drug Administration approval in September 2016 for seven of Humira’s nine indications: rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, plaque psoriasis, and polyarticular juvenile idiopathic arthritis. Amjevita is not approved for two of Humira’s indications, hidradenitis suppurativa and uveitis.
Amgen said in its statement that AbbVie will grant patent licenses for the use and sale of Amjevita worldwide, on a country-by-country basis, with current expectations that marketing will begin in Europe on Oct. 16, 2018, and in the United States on Jan. 31, 2023. Amjevita is named Amgevita in Europe.
Amgen, maker of the adalimumab biosimilar Amjevita (adalimumab-atto) has reached an agreement with AbbVie, manufacturer of the originator adalimumab Humira, that halts marketing of Amjevita in the United States until 2023 and in Europe until 2018, according to a company statement.
The deal between the two manufacturers settles a patent infringement lawsuit that AbbVie brought against Amgen after it received Food and Drug Administration approval in September 2016 for seven of Humira’s nine indications: rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, plaque psoriasis, and polyarticular juvenile idiopathic arthritis. Amjevita is not approved for two of Humira’s indications, hidradenitis suppurativa and uveitis.
Amgen said in its statement that AbbVie will grant patent licenses for the use and sale of Amjevita worldwide, on a country-by-country basis, with current expectations that marketing will begin in Europe on Oct. 16, 2018, and in the United States on Jan. 31, 2023. Amjevita is named Amgevita in Europe.
Amgen, maker of the adalimumab biosimilar Amjevita (adalimumab-atto) has reached an agreement with AbbVie, manufacturer of the originator adalimumab Humira, that halts marketing of Amjevita in the United States until 2023 and in Europe until 2018, according to a company statement.
The deal between the two manufacturers settles a patent infringement lawsuit that AbbVie brought against Amgen after it received Food and Drug Administration approval in September 2016 for seven of Humira’s nine indications: rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, plaque psoriasis, and polyarticular juvenile idiopathic arthritis. Amjevita is not approved for two of Humira’s indications, hidradenitis suppurativa and uveitis.
Amgen said in its statement that AbbVie will grant patent licenses for the use and sale of Amjevita worldwide, on a country-by-country basis, with current expectations that marketing will begin in Europe on Oct. 16, 2018, and in the United States on Jan. 31, 2023. Amjevita is named Amgevita in Europe.
Swedish study finds low risk of developing psoriasis in bariatric surgery patients
Obese patients who undergo bariatric surgery have a lower risk of later developing psoriasis, according to results of nonrandomized, longitudinal intervention trial.
Cristina Maglio, MD, of the University of Gothenburg, Sweden, and her associates found that over a 26-year follow-up period, the adjusted hazard ratio (HR) of developing psoriasis was 0.65 (95% confidence interval [CI], 0.47-0.89; P = .008) for patients who underwent bariatric surgery, compared with those who received conventional, nonsurgical obesity treatments. Psoriasis developed in 3.6% of 1,991 patients in the surgery group during follow-up and in 5.1% of 2,018 control patients during follow-up.
Conversely, the difference in the risk of developing psoriatic arthritis (PsA), experienced by up to one-third of patients with psoriasis, was not statistically significant (HR, 0.77; 95% CI, 0.43-1.37; P = .287). PsA developed in 1% of subjects from the surgery group and 1.3% from the control group.
To understand how surgery affected the development of psoriasis or psoriatic arthritis, the researchers conducted a trial with a control group and surgery group. In the control group, 2,018 patients received standard obesity treatments that included recommendations on eating behavior, food selection, and physical activity. The 1,991 patients in the surgery group underwent gastric banding (375), vertical banded gastroplasty (1,354), or gastric bypass (262). At the start of the study, patients were evaluated for baseline measurements, then again at 6 months. After the 6-month mark, patients were reevaluated at 1, 2, 3, 4, 6, 8, 10, 15, and 20 years, respectively. All study participants, regardless of trial group, were examined and presented patient health questionnaires at each follow-up. The endpoint for this study was the first diagnosis of either psoriasis or PsA. Body mass index decreased significantly in the surgery group, compared with virtually no change in the control group.
Vertical banded gastroplasty was found to significantly lower the incidence of psoriasis, compared with usual treatment. But using gastric banding as a reference, vertical banded gastroplasty (HR, 0.80; 95% CI, 0.46-1.39; P = .418) and gastric bypass (HR, 0.71; 95% CI, 0.29-1.71; P = 0.439) were found to have similar effects on the prevention of psoriasis.
The researchers also identified several risk factors that significantly increased the risk of developing psoriasis. Smoking (HR, 1.75; 95% CI, 1.26-2.42; P = .001), a known risk factor in the development of psoriasis, and the length of time a patient had been obese (HR, 1.28; 95% CI, 1.05-1.55; P = .014) were found to be independently associated with an increased risk of psoriasis.
As part of their risk analysis, Dr. Maglio and her colleagues analyzed the interactions of baseline risk factors such as BMI and obesity duration with the bariatric surgery. This analysis found no significant interactions between baseline risk factors and bariatric surgery. It did reveal that patients who were older at baseline evaluation had slightly better responses to bariatric surgery with lower incidences of psoriasis, compared with younger patients, but the differences were not statistically significant.
“The preventive role of bariatric surgery on the risk of psoriasis has been recently highlighted by a retrospective Danish study (JAMA Surg. 2017 Apr 1;152[4]:344-9),” noted Dr. Maglio and her colleagues. “However, we lent strength to the previous results by confirming this association in a large prospective intervention trial designed to examine the effect of bariatric surgery on obesity-related comorbidities in comparison with usual obesity care.
This study was funded in part by the National Institutes of Diabetes and Digestive and Kidney Diseases, the Swedish Rheumatism association, the Swedish Research Council, the University of Gothenburg, and the Swedish federal government. Dr. Anna Rudin reported that part of her salary at Sahlgrenska University is supported by a grant from AstraZeneca. Dr. Lena M.S. Carlsson has received lecture fees from AstraZeneca, Johnson & Johnson, and Merck Sharp and Dohme. Dr. Maglio and Dr. Markku Peltonen had no relevant financial disclosures.
SOURCE: Maglio et al. Obesity. 2017. Dec; 25[12]:2068-73.
Obese patients who undergo bariatric surgery have a lower risk of later developing psoriasis, according to results of nonrandomized, longitudinal intervention trial.
Cristina Maglio, MD, of the University of Gothenburg, Sweden, and her associates found that over a 26-year follow-up period, the adjusted hazard ratio (HR) of developing psoriasis was 0.65 (95% confidence interval [CI], 0.47-0.89; P = .008) for patients who underwent bariatric surgery, compared with those who received conventional, nonsurgical obesity treatments. Psoriasis developed in 3.6% of 1,991 patients in the surgery group during follow-up and in 5.1% of 2,018 control patients during follow-up.
Conversely, the difference in the risk of developing psoriatic arthritis (PsA), experienced by up to one-third of patients with psoriasis, was not statistically significant (HR, 0.77; 95% CI, 0.43-1.37; P = .287). PsA developed in 1% of subjects from the surgery group and 1.3% from the control group.
To understand how surgery affected the development of psoriasis or psoriatic arthritis, the researchers conducted a trial with a control group and surgery group. In the control group, 2,018 patients received standard obesity treatments that included recommendations on eating behavior, food selection, and physical activity. The 1,991 patients in the surgery group underwent gastric banding (375), vertical banded gastroplasty (1,354), or gastric bypass (262). At the start of the study, patients were evaluated for baseline measurements, then again at 6 months. After the 6-month mark, patients were reevaluated at 1, 2, 3, 4, 6, 8, 10, 15, and 20 years, respectively. All study participants, regardless of trial group, were examined and presented patient health questionnaires at each follow-up. The endpoint for this study was the first diagnosis of either psoriasis or PsA. Body mass index decreased significantly in the surgery group, compared with virtually no change in the control group.
Vertical banded gastroplasty was found to significantly lower the incidence of psoriasis, compared with usual treatment. But using gastric banding as a reference, vertical banded gastroplasty (HR, 0.80; 95% CI, 0.46-1.39; P = .418) and gastric bypass (HR, 0.71; 95% CI, 0.29-1.71; P = 0.439) were found to have similar effects on the prevention of psoriasis.
The researchers also identified several risk factors that significantly increased the risk of developing psoriasis. Smoking (HR, 1.75; 95% CI, 1.26-2.42; P = .001), a known risk factor in the development of psoriasis, and the length of time a patient had been obese (HR, 1.28; 95% CI, 1.05-1.55; P = .014) were found to be independently associated with an increased risk of psoriasis.
As part of their risk analysis, Dr. Maglio and her colleagues analyzed the interactions of baseline risk factors such as BMI and obesity duration with the bariatric surgery. This analysis found no significant interactions between baseline risk factors and bariatric surgery. It did reveal that patients who were older at baseline evaluation had slightly better responses to bariatric surgery with lower incidences of psoriasis, compared with younger patients, but the differences were not statistically significant.
“The preventive role of bariatric surgery on the risk of psoriasis has been recently highlighted by a retrospective Danish study (JAMA Surg. 2017 Apr 1;152[4]:344-9),” noted Dr. Maglio and her colleagues. “However, we lent strength to the previous results by confirming this association in a large prospective intervention trial designed to examine the effect of bariatric surgery on obesity-related comorbidities in comparison with usual obesity care.
This study was funded in part by the National Institutes of Diabetes and Digestive and Kidney Diseases, the Swedish Rheumatism association, the Swedish Research Council, the University of Gothenburg, and the Swedish federal government. Dr. Anna Rudin reported that part of her salary at Sahlgrenska University is supported by a grant from AstraZeneca. Dr. Lena M.S. Carlsson has received lecture fees from AstraZeneca, Johnson & Johnson, and Merck Sharp and Dohme. Dr. Maglio and Dr. Markku Peltonen had no relevant financial disclosures.
SOURCE: Maglio et al. Obesity. 2017. Dec; 25[12]:2068-73.
Obese patients who undergo bariatric surgery have a lower risk of later developing psoriasis, according to results of nonrandomized, longitudinal intervention trial.
Cristina Maglio, MD, of the University of Gothenburg, Sweden, and her associates found that over a 26-year follow-up period, the adjusted hazard ratio (HR) of developing psoriasis was 0.65 (95% confidence interval [CI], 0.47-0.89; P = .008) for patients who underwent bariatric surgery, compared with those who received conventional, nonsurgical obesity treatments. Psoriasis developed in 3.6% of 1,991 patients in the surgery group during follow-up and in 5.1% of 2,018 control patients during follow-up.
Conversely, the difference in the risk of developing psoriatic arthritis (PsA), experienced by up to one-third of patients with psoriasis, was not statistically significant (HR, 0.77; 95% CI, 0.43-1.37; P = .287). PsA developed in 1% of subjects from the surgery group and 1.3% from the control group.
To understand how surgery affected the development of psoriasis or psoriatic arthritis, the researchers conducted a trial with a control group and surgery group. In the control group, 2,018 patients received standard obesity treatments that included recommendations on eating behavior, food selection, and physical activity. The 1,991 patients in the surgery group underwent gastric banding (375), vertical banded gastroplasty (1,354), or gastric bypass (262). At the start of the study, patients were evaluated for baseline measurements, then again at 6 months. After the 6-month mark, patients were reevaluated at 1, 2, 3, 4, 6, 8, 10, 15, and 20 years, respectively. All study participants, regardless of trial group, were examined and presented patient health questionnaires at each follow-up. The endpoint for this study was the first diagnosis of either psoriasis or PsA. Body mass index decreased significantly in the surgery group, compared with virtually no change in the control group.
Vertical banded gastroplasty was found to significantly lower the incidence of psoriasis, compared with usual treatment. But using gastric banding as a reference, vertical banded gastroplasty (HR, 0.80; 95% CI, 0.46-1.39; P = .418) and gastric bypass (HR, 0.71; 95% CI, 0.29-1.71; P = 0.439) were found to have similar effects on the prevention of psoriasis.
The researchers also identified several risk factors that significantly increased the risk of developing psoriasis. Smoking (HR, 1.75; 95% CI, 1.26-2.42; P = .001), a known risk factor in the development of psoriasis, and the length of time a patient had been obese (HR, 1.28; 95% CI, 1.05-1.55; P = .014) were found to be independently associated with an increased risk of psoriasis.
As part of their risk analysis, Dr. Maglio and her colleagues analyzed the interactions of baseline risk factors such as BMI and obesity duration with the bariatric surgery. This analysis found no significant interactions between baseline risk factors and bariatric surgery. It did reveal that patients who were older at baseline evaluation had slightly better responses to bariatric surgery with lower incidences of psoriasis, compared with younger patients, but the differences were not statistically significant.
“The preventive role of bariatric surgery on the risk of psoriasis has been recently highlighted by a retrospective Danish study (JAMA Surg. 2017 Apr 1;152[4]:344-9),” noted Dr. Maglio and her colleagues. “However, we lent strength to the previous results by confirming this association in a large prospective intervention trial designed to examine the effect of bariatric surgery on obesity-related comorbidities in comparison with usual obesity care.
This study was funded in part by the National Institutes of Diabetes and Digestive and Kidney Diseases, the Swedish Rheumatism association, the Swedish Research Council, the University of Gothenburg, and the Swedish federal government. Dr. Anna Rudin reported that part of her salary at Sahlgrenska University is supported by a grant from AstraZeneca. Dr. Lena M.S. Carlsson has received lecture fees from AstraZeneca, Johnson & Johnson, and Merck Sharp and Dohme. Dr. Maglio and Dr. Markku Peltonen had no relevant financial disclosures.
SOURCE: Maglio et al. Obesity. 2017. Dec; 25[12]:2068-73.
FROM OBESITY
Key clinical point:
Major finding: Obese patients who underwent bariatric surgery had a lower incidence of psoriasis over a 26-year period (HR, 0.65; 95% CI: 0.47-0.89; P = .008), compared with usual care.
Study details: Swedish Obese Subjects study, a longitudinal, nonrandomized intervention trial comprising 1,991 surgery group patients and 2,018 control patients.
Disclosures: This study was funded in part by the National Institutes of Diabetes and Digestive and Kidney Diseases, the Swedish Rheumatism association, the Swedish Research Council, the University of Gothenburg, and the Swedish federal government. Dr. Anna Rudin reported that part of her salary at Sahlgrenska University is supported by a grant from AstraZeneca. Dr. Lena M.S. Carlsson has received lecture fees from AstraZeneca, Johnson & Johnson, and Merck Sharp and Dohme. Dr. Maglio and Dr. Markku Peltonen had no relevant financial disclosures.
Source: Maglio et al. Obesity. 2017. Dec; 25[12]:2068-2073.
Smoking linked to increased psoriasis risk
Current and former smokers were significantly more likely to have psoriasis than were nonsmokers in an analysis of the Korean National Health Insurance database.
Multivariate analyses produced adjusted incidence rates of 1.14 for current smokers (n = 132,566) and 1.11 for former smokers (n = 47,477), compared with nonsmokers (n = 320,435), indicating “that smoking status is an independent potential risk factor for psoriasis,” reported Eun Joo Lee, PhD, of the National Health Insurance Service in Wonjusi, South Korea, and associates (J Am Acad Dermatol. 2017;77[3]:573-5).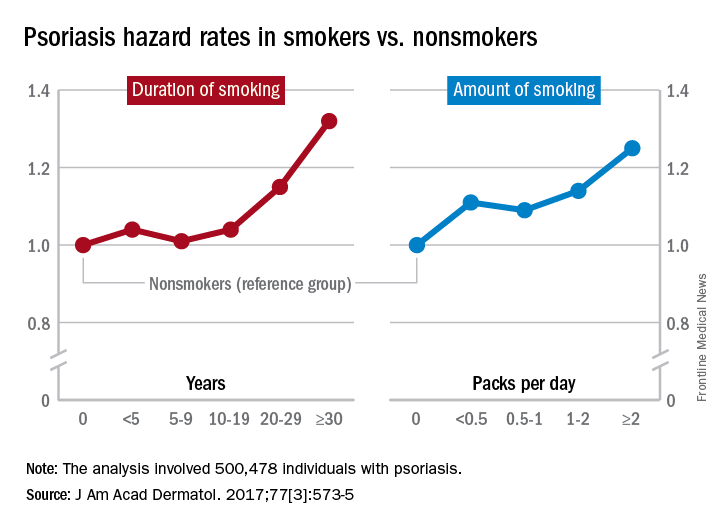
The study was supported by a grant from the National Research Foundation of Korea that was funded by the Korean government. The investigators did not declare any conflicts of interest.
Current and former smokers were significantly more likely to have psoriasis than were nonsmokers in an analysis of the Korean National Health Insurance database.
Multivariate analyses produced adjusted incidence rates of 1.14 for current smokers (n = 132,566) and 1.11 for former smokers (n = 47,477), compared with nonsmokers (n = 320,435), indicating “that smoking status is an independent potential risk factor for psoriasis,” reported Eun Joo Lee, PhD, of the National Health Insurance Service in Wonjusi, South Korea, and associates (J Am Acad Dermatol. 2017;77[3]:573-5).
The study was supported by a grant from the National Research Foundation of Korea that was funded by the Korean government. The investigators did not declare any conflicts of interest.
Current and former smokers were significantly more likely to have psoriasis than were nonsmokers in an analysis of the Korean National Health Insurance database.
Multivariate analyses produced adjusted incidence rates of 1.14 for current smokers (n = 132,566) and 1.11 for former smokers (n = 47,477), compared with nonsmokers (n = 320,435), indicating “that smoking status is an independent potential risk factor for psoriasis,” reported Eun Joo Lee, PhD, of the National Health Insurance Service in Wonjusi, South Korea, and associates (J Am Acad Dermatol. 2017;77[3]:573-5).
The study was supported by a grant from the National Research Foundation of Korea that was funded by the Korean government. The investigators did not declare any conflicts of interest.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
50 years of pediatric dermatology
The world in pediatric dermatology has changed in incredible ways since 1967. In fact, pediatric dermatology was not an organized specialty until years later! This article will look back at some of the history of pediatric dermatology, exploring how different the field was 50 years ago, and how it has evolved into the vibrant field that it is. By looking at some disease states, and differences in practice in relation to the care of dermatologic conditions in children both by pediatricians and dermatologists, we can see the tremendous evolution in our understanding and management of pediatric skin conditions, and perhaps gain insight into the future.
Pediatric dermatology was fairly “neonatal” 50 years ago, with only a few practitioners in the field. Recognizing that up to 30% of pediatric primary care visits include a skin-related problem, and that there was limited training about skin diseases among primary care practitioners and inconsistent training amongst dermatologists, there was a clinical need for establishing the subspecialty of pediatric dermatology. The first international symposium was held in Mexico City in October 1972, and with this meeting the International Society of Pediatric Dermatology was founded. The Society for Pediatric Dermatology (SPD) began in 1973, with Alvin Jacobs, MD, Samuel Weinberg, MD, Nancy Esterly, MD, Sidney Hurwitz, MD, William Weston, MD, and Coleman Jacobson, MD, as some of the initial “founding mothers and fathers.” The journal Pediatric Dermatology released its first issue in 1982 (35 years ago), and the American Academy of Pediatrics did not have a section of dermatology until 1986.
Pediatrics and dermatology: The interface
Many of the first generation of pediatric dermatologists trained as pediatricians prior to pursuing their dermatology work, with some being “assigned” dermatology as pediatric experts, while others did formal residencies in dermatology. This history is important, as pediatric dermatology was, and remains, integrated with pediatrics, even while training in dermatology residencies became standard practice. An important part of the development of the field has been the education of pediatricians and dermatologists by pediatric dermatologists, with a strong sensibility that improved training for both generalists and specialists about pediatric skin disease would yield better care for patients and families.
Initially, there were very few pediatric or dermatology programs in the United States that had pediatric dermatologists. Over the succeeding decades, this is now less common, although even now there are still dermatology and pediatric residency programs that do not have a pediatric dermatologist for either training or to serve their patients. The founding leaders of the SPD set a tone of collaboration nationally and internationally, reaching out to pediatric colleagues and dermatology associates from around the world, and establishing superb educational programs for the exchange of ideas, presentation of challenging cases, and promoting state of the art knowledge of the field. Through annual meetings of the SPD, conferences immediately preceding the American Academy of Dermatology annual meetings, the World Congress of Pediatric Dermatology, and other regional and international meetings, the field developed as the number of practitioners grew, and as the specialized published literature reflected new knowledge in diagnosis and therapy.
Building upon the history of collaboration and reflecting the maturation of the field with a desire to influence the breadth and quantity of research in pediatric dermatology, the Pediatric Dermatology Research Alliance (PeDRA) was formed in 2012. This organization was formed to promote and facilitate high quality collaborative clinical, translational, educational, and basic science research in pediatric dermatology with a vision to create sustainable, collaborative networks to better understand, prevent, treat, and cure dermatologic diseases in children. This network is now composed of over 230 members representing over 68 institutions from the United States and Canada, but including involvement globally from Mexico, Europe, and the Middle East.
Examples of changing perspectives: hemangiomas
A good way to look at evolution of the field is take a look at some of the similarities and differences in clinical practice in relation to common and uncommon disease states.
A great example is hemangiomas. Some of the first natural history studies on hemangiomas were done in the early 1960s, establishing that many lesions had a typical clinical course of fairly rapid growth, plateau, and involution over time. Of course, the identification of hemangiomas of infancy (or “HOI” in the trade), was confused with vascular malformations, and no one had recognized variant tumors that were distinct, such as rapidly involuting and noninvoluting congenital hemangiomas (RICHs or NICHs), tufted angiomas, and hemangioendotheliomas. PHACE syndrome (posterior fossa brain malformations) had yet to be described (that was done in 1996 by Ilona Frieden and her colleagues). For a time period, hemangiomas were treated with X-rays, before the negative impact of such radiation was acknowledged. For many years after that, even deforming and functionally significant lesions were “followed clinically” for natural involution, presumably a backlash from the radiation therapy interventions.
This story also reflects how organized research efforts helped with the evolution of knowledge and clinical care. The Hemangioma of Infancy Group was formed to take a collaborative approach to characterize and study hemangiomas and related tumors. Beginning with energetic, insightful pediatric dermatologists, and little funding, they changed our knowledge base of how hemangiomas present, the risk factors for their development and the characteristics and multiple organ findings associated with PHACE and other syndromic hemangiomas.
Procedural pediatric dermatology: Tremendous revolution in surgery and laser
The first generation of pediatric dermatologists were considered medical dermatologist specialists. And how important this specialty work was! Acne, atopic dermatitis, psoriasis, diaper and seborrheic dermatitis, and rare genetic syndromes, these conditions were a major part of the work of early pediatric dermatologists (and remain so now). What was not common was for pediatric dermatologists to have procedural or surgical practices, while this now is routinely part of the work of specialists in the field. How did this shift occur?
The fundamental shift began to occur with the introduction of the pulsed dye laser in 1989 and the publication of a seminal article in the New England Journal of Medicine (1989 Feb 16;320[7]:416-21) on its utility in treating port-wine stains in children with minimal scarring. Vascular lesions including port-wine stains were common, and pediatric dermatologists managed these patients for both diagnosis and medical management. Also, dermatology residencies at this time offered training in cutaneous surgery, excisions (including Mohs surgery) and repairs, and trainees in pediatric dermatology were “trained up” to high levels of expertise. As lasers were incorporated into dermatology residency work and practices, pediatric dermatologists had the exposure and skill to do this work. An added advantage was having the pediatric knowledge of how to handle children and adolescents in an age appropriate manner, and consideration of methods to minimize the pain and anxiety of procedures. Within a few years, pediatric dermatologists were at the forefront of the use of topical anesthetics (EMLA and liposomal lidocaine) and had general anesthesia privileges for laser and excisional surgery.
So while pediatric dermatologists still do “small procedures” every hour in most practices (cryotherapy for warts, cantharidin for molluscum, shave and punch biopsies), a subset now have extensive procedural practices, which in recent years has extended to pigment lesion lasers (to treat nevus of Ota), hair lasers (to treat perineal areas to prevent pilonidal cyst recurrence or to treat hirsutism), and combinations of lasers to treat hypertrophic, constrictive, and/or deforming scars).
Inflammatory skin disorders: Bread and butter ... and peanut butter?
The care of pediatric inflammatory skin disorders has evolved, but more slowly for some diseases than others. Acne vulgaris now is recognized as much more common under age 12 years than previously, presumably reflecting earlier pubertal changes in our preteens. Over the past 30 years, therapy has evolved with the use of topical retinoids (still underused by pediatricians, considered a “practice gap”), hormonal therapy with combined oral contraceptives, and oral isotretinoin, a powerful but highly effective systemic agent for severe and refractory acne. Specific pediatric guidelines came much later. Pediatric acne expert recommendations were formulated by the American Acne and Rosacea Society and endorsed by the American Academy of Pediatrics in 2013 (Pediatrics. 2013;131:S163-86). Over the past few years, there is a push by experts for more judicious use of antibiotics for acne (oral and topical) to minimize the emergence of bacterial resistance.
Psoriasis has been a condition that has been “behind the revolution,” in that no biologic agent was approved for pediatric psoriasis in the United States until several months ago, lagging behind Europe and elsewhere in the world by almost a decade. Adult psoriasis has been recognized to be associated with a broad set of comorbidities, including obesity and early heart disease, and there is now research on how children are at risk as well, and new recommendations on how to screen children with psoriasis. Moderate to severe psoriasis in adults is now tremendously controllable with biologic agents targeting TNF-alpha, IL 12/23, and IL-17. Etanercept has been approved for children with psoriasis aged 4 years and older, and other biologic agents are under study.
Atopic dermatitis now is ready for its revolution! AD has increased in prevalence from around 5% of the pediatric population 30-plus years ago to 10%-15%. Treatment of most individuals has remained the same over the decades: Good skin care, frequent moisturizers, topical corticosteroids for flares, management of infection if noted. The topical calcineurin inhibitors (TCIs) broadened the therapeutic approach when introduced in 2000 and 2001, but the boxed warning resulted in some practitioners minimizing their utilization of these useful agents.

It has been recognized for years that children with AD have higher risk of developing food allergies than children without AD. A changing understanding of how early food exposure may induce tolerance is changing the world of allergy and influencing the care of children with AD. This is where the peanut butter (or other processed peanut, such as “Bamba”) may be life saving. New guidelines have come from the National Institute of Allergy and Infectious Diseases recommending that infants with severe eczema (or egg allergy, or both) have introduction of age-appropriate peanut-containing food as early as 4-6 months of age to reduce the risk of development of peanut allergy. It is recommended that these infants undergo early evaluation for possible sensitization to peanut protein, with referral to allergists for skin prick tests or serum IgE screens (though if positive, referral to allergists is appropriate), and assess the safety of going ahead with early feeding. It is hoped that following these new guidelines can minimize the development of peanut allergy.
The future
Where will pediatric skin disease, or more importantly, skin health over a lifetime be in 50 years? Can we cure or prevent the consequences of our lethal and life altering genetic diseases such as epidermolysis bullosa or our neurocutaneous disorders? Will our new insights into birthmarks (they are mostly somatic mutations) allow us to form specific, personalized therapies to minimize their impact? Will we be using computers equipped with imaging devices and algorithms to assess our patients’ moles, papules, and nodules? Will our vaccines have wiped out warts, molluscum, and perhaps, acne? Will we have cured our inflammatory skin disorders, or perhaps prevented them by interventions in the neonatal period? No predictions will be offered here, other than that we can look forward to incredible changes for our future generations of health care practitioners, patients, and families.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego and professor of dermatology and pediatrics at the University of California, San Diego. Dr. Eichenfield has served as a consultant for Anacor/Pfizer and Regeneron/Sanofi. Email him at pdnews@frontlinemedcom.com.
The world in pediatric dermatology has changed in incredible ways since 1967. In fact, pediatric dermatology was not an organized specialty until years later! This article will look back at some of the history of pediatric dermatology, exploring how different the field was 50 years ago, and how it has evolved into the vibrant field that it is. By looking at some disease states, and differences in practice in relation to the care of dermatologic conditions in children both by pediatricians and dermatologists, we can see the tremendous evolution in our understanding and management of pediatric skin conditions, and perhaps gain insight into the future.
Pediatric dermatology was fairly “neonatal” 50 years ago, with only a few practitioners in the field. Recognizing that up to 30% of pediatric primary care visits include a skin-related problem, and that there was limited training about skin diseases among primary care practitioners and inconsistent training amongst dermatologists, there was a clinical need for establishing the subspecialty of pediatric dermatology. The first international symposium was held in Mexico City in October 1972, and with this meeting the International Society of Pediatric Dermatology was founded. The Society for Pediatric Dermatology (SPD) began in 1973, with Alvin Jacobs, MD, Samuel Weinberg, MD, Nancy Esterly, MD, Sidney Hurwitz, MD, William Weston, MD, and Coleman Jacobson, MD, as some of the initial “founding mothers and fathers.” The journal Pediatric Dermatology released its first issue in 1982 (35 years ago), and the American Academy of Pediatrics did not have a section of dermatology until 1986.
Pediatrics and dermatology: The interface
Many of the first generation of pediatric dermatologists trained as pediatricians prior to pursuing their dermatology work, with some being “assigned” dermatology as pediatric experts, while others did formal residencies in dermatology. This history is important, as pediatric dermatology was, and remains, integrated with pediatrics, even while training in dermatology residencies became standard practice. An important part of the development of the field has been the education of pediatricians and dermatologists by pediatric dermatologists, with a strong sensibility that improved training for both generalists and specialists about pediatric skin disease would yield better care for patients and families.
Initially, there were very few pediatric or dermatology programs in the United States that had pediatric dermatologists. Over the succeeding decades, this is now less common, although even now there are still dermatology and pediatric residency programs that do not have a pediatric dermatologist for either training or to serve their patients. The founding leaders of the SPD set a tone of collaboration nationally and internationally, reaching out to pediatric colleagues and dermatology associates from around the world, and establishing superb educational programs for the exchange of ideas, presentation of challenging cases, and promoting state of the art knowledge of the field. Through annual meetings of the SPD, conferences immediately preceding the American Academy of Dermatology annual meetings, the World Congress of Pediatric Dermatology, and other regional and international meetings, the field developed as the number of practitioners grew, and as the specialized published literature reflected new knowledge in diagnosis and therapy.
Building upon the history of collaboration and reflecting the maturation of the field with a desire to influence the breadth and quantity of research in pediatric dermatology, the Pediatric Dermatology Research Alliance (PeDRA) was formed in 2012. This organization was formed to promote and facilitate high quality collaborative clinical, translational, educational, and basic science research in pediatric dermatology with a vision to create sustainable, collaborative networks to better understand, prevent, treat, and cure dermatologic diseases in children. This network is now composed of over 230 members representing over 68 institutions from the United States and Canada, but including involvement globally from Mexico, Europe, and the Middle East.
Examples of changing perspectives: hemangiomas
A good way to look at evolution of the field is take a look at some of the similarities and differences in clinical practice in relation to common and uncommon disease states.
A great example is hemangiomas. Some of the first natural history studies on hemangiomas were done in the early 1960s, establishing that many lesions had a typical clinical course of fairly rapid growth, plateau, and involution over time. Of course, the identification of hemangiomas of infancy (or “HOI” in the trade), was confused with vascular malformations, and no one had recognized variant tumors that were distinct, such as rapidly involuting and noninvoluting congenital hemangiomas (RICHs or NICHs), tufted angiomas, and hemangioendotheliomas. PHACE syndrome (posterior fossa brain malformations) had yet to be described (that was done in 1996 by Ilona Frieden and her colleagues). For a time period, hemangiomas were treated with X-rays, before the negative impact of such radiation was acknowledged. For many years after that, even deforming and functionally significant lesions were “followed clinically” for natural involution, presumably a backlash from the radiation therapy interventions.
This story also reflects how organized research efforts helped with the evolution of knowledge and clinical care. The Hemangioma of Infancy Group was formed to take a collaborative approach to characterize and study hemangiomas and related tumors. Beginning with energetic, insightful pediatric dermatologists, and little funding, they changed our knowledge base of how hemangiomas present, the risk factors for their development and the characteristics and multiple organ findings associated with PHACE and other syndromic hemangiomas.
Procedural pediatric dermatology: Tremendous revolution in surgery and laser
The first generation of pediatric dermatologists were considered medical dermatologist specialists. And how important this specialty work was! Acne, atopic dermatitis, psoriasis, diaper and seborrheic dermatitis, and rare genetic syndromes, these conditions were a major part of the work of early pediatric dermatologists (and remain so now). What was not common was for pediatric dermatologists to have procedural or surgical practices, while this now is routinely part of the work of specialists in the field. How did this shift occur?
The fundamental shift began to occur with the introduction of the pulsed dye laser in 1989 and the publication of a seminal article in the New England Journal of Medicine (1989 Feb 16;320[7]:416-21) on its utility in treating port-wine stains in children with minimal scarring. Vascular lesions including port-wine stains were common, and pediatric dermatologists managed these patients for both diagnosis and medical management. Also, dermatology residencies at this time offered training in cutaneous surgery, excisions (including Mohs surgery) and repairs, and trainees in pediatric dermatology were “trained up” to high levels of expertise. As lasers were incorporated into dermatology residency work and practices, pediatric dermatologists had the exposure and skill to do this work. An added advantage was having the pediatric knowledge of how to handle children and adolescents in an age appropriate manner, and consideration of methods to minimize the pain and anxiety of procedures. Within a few years, pediatric dermatologists were at the forefront of the use of topical anesthetics (EMLA and liposomal lidocaine) and had general anesthesia privileges for laser and excisional surgery.
So while pediatric dermatologists still do “small procedures” every hour in most practices (cryotherapy for warts, cantharidin for molluscum, shave and punch biopsies), a subset now have extensive procedural practices, which in recent years has extended to pigment lesion lasers (to treat nevus of Ota), hair lasers (to treat perineal areas to prevent pilonidal cyst recurrence or to treat hirsutism), and combinations of lasers to treat hypertrophic, constrictive, and/or deforming scars).
Inflammatory skin disorders: Bread and butter ... and peanut butter?
The care of pediatric inflammatory skin disorders has evolved, but more slowly for some diseases than others. Acne vulgaris now is recognized as much more common under age 12 years than previously, presumably reflecting earlier pubertal changes in our preteens. Over the past 30 years, therapy has evolved with the use of topical retinoids (still underused by pediatricians, considered a “practice gap”), hormonal therapy with combined oral contraceptives, and oral isotretinoin, a powerful but highly effective systemic agent for severe and refractory acne. Specific pediatric guidelines came much later. Pediatric acne expert recommendations were formulated by the American Acne and Rosacea Society and endorsed by the American Academy of Pediatrics in 2013 (Pediatrics. 2013;131:S163-86). Over the past few years, there is a push by experts for more judicious use of antibiotics for acne (oral and topical) to minimize the emergence of bacterial resistance.
Psoriasis has been a condition that has been “behind the revolution,” in that no biologic agent was approved for pediatric psoriasis in the United States until several months ago, lagging behind Europe and elsewhere in the world by almost a decade. Adult psoriasis has been recognized to be associated with a broad set of comorbidities, including obesity and early heart disease, and there is now research on how children are at risk as well, and new recommendations on how to screen children with psoriasis. Moderate to severe psoriasis in adults is now tremendously controllable with biologic agents targeting TNF-alpha, IL 12/23, and IL-17. Etanercept has been approved for children with psoriasis aged 4 years and older, and other biologic agents are under study.
Atopic dermatitis now is ready for its revolution! AD has increased in prevalence from around 5% of the pediatric population 30-plus years ago to 10%-15%. Treatment of most individuals has remained the same over the decades: Good skin care, frequent moisturizers, topical corticosteroids for flares, management of infection if noted. The topical calcineurin inhibitors (TCIs) broadened the therapeutic approach when introduced in 2000 and 2001, but the boxed warning resulted in some practitioners minimizing their utilization of these useful agents.

It has been recognized for years that children with AD have higher risk of developing food allergies than children without AD. A changing understanding of how early food exposure may induce tolerance is changing the world of allergy and influencing the care of children with AD. This is where the peanut butter (or other processed peanut, such as “Bamba”) may be life saving. New guidelines have come from the National Institute of Allergy and Infectious Diseases recommending that infants with severe eczema (or egg allergy, or both) have introduction of age-appropriate peanut-containing food as early as 4-6 months of age to reduce the risk of development of peanut allergy. It is recommended that these infants undergo early evaluation for possible sensitization to peanut protein, with referral to allergists for skin prick tests or serum IgE screens (though if positive, referral to allergists is appropriate), and assess the safety of going ahead with early feeding. It is hoped that following these new guidelines can minimize the development of peanut allergy.
The future
Where will pediatric skin disease, or more importantly, skin health over a lifetime be in 50 years? Can we cure or prevent the consequences of our lethal and life altering genetic diseases such as epidermolysis bullosa or our neurocutaneous disorders? Will our new insights into birthmarks (they are mostly somatic mutations) allow us to form specific, personalized therapies to minimize their impact? Will we be using computers equipped with imaging devices and algorithms to assess our patients’ moles, papules, and nodules? Will our vaccines have wiped out warts, molluscum, and perhaps, acne? Will we have cured our inflammatory skin disorders, or perhaps prevented them by interventions in the neonatal period? No predictions will be offered here, other than that we can look forward to incredible changes for our future generations of health care practitioners, patients, and families.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego and professor of dermatology and pediatrics at the University of California, San Diego. Dr. Eichenfield has served as a consultant for Anacor/Pfizer and Regeneron/Sanofi. Email him at pdnews@frontlinemedcom.com.
The world in pediatric dermatology has changed in incredible ways since 1967. In fact, pediatric dermatology was not an organized specialty until years later! This article will look back at some of the history of pediatric dermatology, exploring how different the field was 50 years ago, and how it has evolved into the vibrant field that it is. By looking at some disease states, and differences in practice in relation to the care of dermatologic conditions in children both by pediatricians and dermatologists, we can see the tremendous evolution in our understanding and management of pediatric skin conditions, and perhaps gain insight into the future.
Pediatric dermatology was fairly “neonatal” 50 years ago, with only a few practitioners in the field. Recognizing that up to 30% of pediatric primary care visits include a skin-related problem, and that there was limited training about skin diseases among primary care practitioners and inconsistent training amongst dermatologists, there was a clinical need for establishing the subspecialty of pediatric dermatology. The first international symposium was held in Mexico City in October 1972, and with this meeting the International Society of Pediatric Dermatology was founded. The Society for Pediatric Dermatology (SPD) began in 1973, with Alvin Jacobs, MD, Samuel Weinberg, MD, Nancy Esterly, MD, Sidney Hurwitz, MD, William Weston, MD, and Coleman Jacobson, MD, as some of the initial “founding mothers and fathers.” The journal Pediatric Dermatology released its first issue in 1982 (35 years ago), and the American Academy of Pediatrics did not have a section of dermatology until 1986.
Pediatrics and dermatology: The interface
Many of the first generation of pediatric dermatologists trained as pediatricians prior to pursuing their dermatology work, with some being “assigned” dermatology as pediatric experts, while others did formal residencies in dermatology. This history is important, as pediatric dermatology was, and remains, integrated with pediatrics, even while training in dermatology residencies became standard practice. An important part of the development of the field has been the education of pediatricians and dermatologists by pediatric dermatologists, with a strong sensibility that improved training for both generalists and specialists about pediatric skin disease would yield better care for patients and families.
Initially, there were very few pediatric or dermatology programs in the United States that had pediatric dermatologists. Over the succeeding decades, this is now less common, although even now there are still dermatology and pediatric residency programs that do not have a pediatric dermatologist for either training or to serve their patients. The founding leaders of the SPD set a tone of collaboration nationally and internationally, reaching out to pediatric colleagues and dermatology associates from around the world, and establishing superb educational programs for the exchange of ideas, presentation of challenging cases, and promoting state of the art knowledge of the field. Through annual meetings of the SPD, conferences immediately preceding the American Academy of Dermatology annual meetings, the World Congress of Pediatric Dermatology, and other regional and international meetings, the field developed as the number of practitioners grew, and as the specialized published literature reflected new knowledge in diagnosis and therapy.
Building upon the history of collaboration and reflecting the maturation of the field with a desire to influence the breadth and quantity of research in pediatric dermatology, the Pediatric Dermatology Research Alliance (PeDRA) was formed in 2012. This organization was formed to promote and facilitate high quality collaborative clinical, translational, educational, and basic science research in pediatric dermatology with a vision to create sustainable, collaborative networks to better understand, prevent, treat, and cure dermatologic diseases in children. This network is now composed of over 230 members representing over 68 institutions from the United States and Canada, but including involvement globally from Mexico, Europe, and the Middle East.
Examples of changing perspectives: hemangiomas
A good way to look at evolution of the field is take a look at some of the similarities and differences in clinical practice in relation to common and uncommon disease states.
A great example is hemangiomas. Some of the first natural history studies on hemangiomas were done in the early 1960s, establishing that many lesions had a typical clinical course of fairly rapid growth, plateau, and involution over time. Of course, the identification of hemangiomas of infancy (or “HOI” in the trade), was confused with vascular malformations, and no one had recognized variant tumors that were distinct, such as rapidly involuting and noninvoluting congenital hemangiomas (RICHs or NICHs), tufted angiomas, and hemangioendotheliomas. PHACE syndrome (posterior fossa brain malformations) had yet to be described (that was done in 1996 by Ilona Frieden and her colleagues). For a time period, hemangiomas were treated with X-rays, before the negative impact of such radiation was acknowledged. For many years after that, even deforming and functionally significant lesions were “followed clinically” for natural involution, presumably a backlash from the radiation therapy interventions.
This story also reflects how organized research efforts helped with the evolution of knowledge and clinical care. The Hemangioma of Infancy Group was formed to take a collaborative approach to characterize and study hemangiomas and related tumors. Beginning with energetic, insightful pediatric dermatologists, and little funding, they changed our knowledge base of how hemangiomas present, the risk factors for their development and the characteristics and multiple organ findings associated with PHACE and other syndromic hemangiomas.
Procedural pediatric dermatology: Tremendous revolution in surgery and laser
The first generation of pediatric dermatologists were considered medical dermatologist specialists. And how important this specialty work was! Acne, atopic dermatitis, psoriasis, diaper and seborrheic dermatitis, and rare genetic syndromes, these conditions were a major part of the work of early pediatric dermatologists (and remain so now). What was not common was for pediatric dermatologists to have procedural or surgical practices, while this now is routinely part of the work of specialists in the field. How did this shift occur?
The fundamental shift began to occur with the introduction of the pulsed dye laser in 1989 and the publication of a seminal article in the New England Journal of Medicine (1989 Feb 16;320[7]:416-21) on its utility in treating port-wine stains in children with minimal scarring. Vascular lesions including port-wine stains were common, and pediatric dermatologists managed these patients for both diagnosis and medical management. Also, dermatology residencies at this time offered training in cutaneous surgery, excisions (including Mohs surgery) and repairs, and trainees in pediatric dermatology were “trained up” to high levels of expertise. As lasers were incorporated into dermatology residency work and practices, pediatric dermatologists had the exposure and skill to do this work. An added advantage was having the pediatric knowledge of how to handle children and adolescents in an age appropriate manner, and consideration of methods to minimize the pain and anxiety of procedures. Within a few years, pediatric dermatologists were at the forefront of the use of topical anesthetics (EMLA and liposomal lidocaine) and had general anesthesia privileges for laser and excisional surgery.
So while pediatric dermatologists still do “small procedures” every hour in most practices (cryotherapy for warts, cantharidin for molluscum, shave and punch biopsies), a subset now have extensive procedural practices, which in recent years has extended to pigment lesion lasers (to treat nevus of Ota), hair lasers (to treat perineal areas to prevent pilonidal cyst recurrence or to treat hirsutism), and combinations of lasers to treat hypertrophic, constrictive, and/or deforming scars).
Inflammatory skin disorders: Bread and butter ... and peanut butter?
The care of pediatric inflammatory skin disorders has evolved, but more slowly for some diseases than others. Acne vulgaris now is recognized as much more common under age 12 years than previously, presumably reflecting earlier pubertal changes in our preteens. Over the past 30 years, therapy has evolved with the use of topical retinoids (still underused by pediatricians, considered a “practice gap”), hormonal therapy with combined oral contraceptives, and oral isotretinoin, a powerful but highly effective systemic agent for severe and refractory acne. Specific pediatric guidelines came much later. Pediatric acne expert recommendations were formulated by the American Acne and Rosacea Society and endorsed by the American Academy of Pediatrics in 2013 (Pediatrics. 2013;131:S163-86). Over the past few years, there is a push by experts for more judicious use of antibiotics for acne (oral and topical) to minimize the emergence of bacterial resistance.
Psoriasis has been a condition that has been “behind the revolution,” in that no biologic agent was approved for pediatric psoriasis in the United States until several months ago, lagging behind Europe and elsewhere in the world by almost a decade. Adult psoriasis has been recognized to be associated with a broad set of comorbidities, including obesity and early heart disease, and there is now research on how children are at risk as well, and new recommendations on how to screen children with psoriasis. Moderate to severe psoriasis in adults is now tremendously controllable with biologic agents targeting TNF-alpha, IL 12/23, and IL-17. Etanercept has been approved for children with psoriasis aged 4 years and older, and other biologic agents are under study.
Atopic dermatitis now is ready for its revolution! AD has increased in prevalence from around 5% of the pediatric population 30-plus years ago to 10%-15%. Treatment of most individuals has remained the same over the decades: Good skin care, frequent moisturizers, topical corticosteroids for flares, management of infection if noted. The topical calcineurin inhibitors (TCIs) broadened the therapeutic approach when introduced in 2000 and 2001, but the boxed warning resulted in some practitioners minimizing their utilization of these useful agents.

It has been recognized for years that children with AD have higher risk of developing food allergies than children without AD. A changing understanding of how early food exposure may induce tolerance is changing the world of allergy and influencing the care of children with AD. This is where the peanut butter (or other processed peanut, such as “Bamba”) may be life saving. New guidelines have come from the National Institute of Allergy and Infectious Diseases recommending that infants with severe eczema (or egg allergy, or both) have introduction of age-appropriate peanut-containing food as early as 4-6 months of age to reduce the risk of development of peanut allergy. It is recommended that these infants undergo early evaluation for possible sensitization to peanut protein, with referral to allergists for skin prick tests or serum IgE screens (though if positive, referral to allergists is appropriate), and assess the safety of going ahead with early feeding. It is hoped that following these new guidelines can minimize the development of peanut allergy.
The future
Where will pediatric skin disease, or more importantly, skin health over a lifetime be in 50 years? Can we cure or prevent the consequences of our lethal and life altering genetic diseases such as epidermolysis bullosa or our neurocutaneous disorders? Will our new insights into birthmarks (they are mostly somatic mutations) allow us to form specific, personalized therapies to minimize their impact? Will we be using computers equipped with imaging devices and algorithms to assess our patients’ moles, papules, and nodules? Will our vaccines have wiped out warts, molluscum, and perhaps, acne? Will we have cured our inflammatory skin disorders, or perhaps prevented them by interventions in the neonatal period? No predictions will be offered here, other than that we can look forward to incredible changes for our future generations of health care practitioners, patients, and families.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego and professor of dermatology and pediatrics at the University of California, San Diego. Dr. Eichenfield has served as a consultant for Anacor/Pfizer and Regeneron/Sanofi. Email him at pdnews@frontlinemedcom.com.
FDA approves second adalimumab biosimilar for multiple conditions
The Food and Drug Administration has approved Cyltezo (adalimumab-adbm) for multiple conditions.
Cyltezo is an injectable tumor necrosis factor blocker, and is a biosimilar to adalimumab (Humira). The drug is indicated to treat moderate to severe active rheumatoid arthritis, active psoriatic arthritis, active ankylosing spondylitis, moderate to severe active Crohn’s disease, moderate to severe active ulcerative colitis, moderately to severely active polyarticular juvenile idiopathic arthritis in patients 4 years of age and older, and moderate to severe plaque psoriasis.
Find the Cyltezo labeling information here.
The Food and Drug Administration has approved Cyltezo (adalimumab-adbm) for multiple conditions.
Cyltezo is an injectable tumor necrosis factor blocker, and is a biosimilar to adalimumab (Humira). The drug is indicated to treat moderate to severe active rheumatoid arthritis, active psoriatic arthritis, active ankylosing spondylitis, moderate to severe active Crohn’s disease, moderate to severe active ulcerative colitis, moderately to severely active polyarticular juvenile idiopathic arthritis in patients 4 years of age and older, and moderate to severe plaque psoriasis.
Find the Cyltezo labeling information here.
The Food and Drug Administration has approved Cyltezo (adalimumab-adbm) for multiple conditions.
Cyltezo is an injectable tumor necrosis factor blocker, and is a biosimilar to adalimumab (Humira). The drug is indicated to treat moderate to severe active rheumatoid arthritis, active psoriatic arthritis, active ankylosing spondylitis, moderate to severe active Crohn’s disease, moderate to severe active ulcerative colitis, moderately to severely active polyarticular juvenile idiopathic arthritis in patients 4 years of age and older, and moderate to severe plaque psoriasis.
Find the Cyltezo labeling information here.
Noninvasive NASH test could help monitor hepatotoxicity in patients on methotrexate
A noninvasive test for nonalcoholic steatohepatitis (NASH) and hepatic fibrosis could be used to help detect worsening hepatic fibrosis in psoriasis patients who are on long-term methotrexate therapy, according to the authors of a retrospective study published online Aug. 23.
To evaluate the use of the noninvasive test to monitor for hepatic fibrosis in this group of patients and guide the management of methotrexate (MTX) without a liver biopsy, investigators conducted an analysis of 107 patients with psoriasis who were on long-term MTX treatment, for whom the NASH FibroSure test was used between January 2007 and December 2013. All the patients were white, fifty were men, the mean age was 83 years, and almost all of the patients had a body mass index of 28 or more (16% had a BMI between 28 and 30, and 81% had a BMI over 30).
The NASH FibroSure test, which was developed for use in patients suspected of having nonalcoholic fatty liver disease (NAFLD), combines analyses of 10 biochemical markers combined with age, sex, height, and weight to calculate the degree of hepatic fibrosis. The test has a reported sensitivity of 83% and a specificity of 78% in detecting risk of significant fibrosis, according to Bruce Bauer, MD, of Pariser Dermatology Specialists, Norfolk, Va., and his coinvestigators (JAMA Dermatol. 2017 Aug 23. doi: 10.1001/jamadermatol.2017.2083).
Among the 107 patients, the investigators found a statistically significant correlation “between worsening fibrosis scores and cumulative methotrexate” dose among women (P = .02), but not among men (P = .11). In addition, women with a BMI of 28 or more were more likely to have worsening fibrosis scores (P = .03). “There were no differences between men and women with regard to prevalence of a BMI of 28 or more, diabetes, age older than 65 years, or chronic kidney disease,” which they said, suggests that among women, “obesity influences the progression of fibrosis scores.”
The investigators advised providers not to disregard any potential warning signs when using FibroSure on men. “No differences between the cohorts were observed that would explain the association of worsening fibrosis scores and cumulative MTX dose among women but not men,” they wrote. “However, given the implications of the progression of hepatic fibrosis, we still recommend discontinuing MTX for male patients who demonstrate worsening fibrosis scores.”
While the test may not be able to replace liver biopsy, based on these results, the investigators noted that noninvasive tests such as this one could help significantly reduce the number of liver biopsies needed.
They pointed out that because the study was conducted at one center, the next step is to conduct “a prospective, randomized, multi-institutional analysis of NASH FibroSure and liver biopsies for patients with psoriasis receiving MTX vs. other treatments, including a larger cohort of men and women with different racial and ethnic backgrounds.”
One of the four authors reported receiving research funding from T2 Biosystems. Dr. Bauer and the two other authors reported no relevant financial disclosures. The study was funded by the Marshfield Clinic Resident Research Program.
ezimmerman@frontlinemedcom.com
On Twitter @eaztweets
A noninvasive test for nonalcoholic steatohepatitis (NASH) and hepatic fibrosis could be used to help detect worsening hepatic fibrosis in psoriasis patients who are on long-term methotrexate therapy, according to the authors of a retrospective study published online Aug. 23.
To evaluate the use of the noninvasive test to monitor for hepatic fibrosis in this group of patients and guide the management of methotrexate (MTX) without a liver biopsy, investigators conducted an analysis of 107 patients with psoriasis who were on long-term MTX treatment, for whom the NASH FibroSure test was used between January 2007 and December 2013. All the patients were white, fifty were men, the mean age was 83 years, and almost all of the patients had a body mass index of 28 or more (16% had a BMI between 28 and 30, and 81% had a BMI over 30).
The NASH FibroSure test, which was developed for use in patients suspected of having nonalcoholic fatty liver disease (NAFLD), combines analyses of 10 biochemical markers combined with age, sex, height, and weight to calculate the degree of hepatic fibrosis. The test has a reported sensitivity of 83% and a specificity of 78% in detecting risk of significant fibrosis, according to Bruce Bauer, MD, of Pariser Dermatology Specialists, Norfolk, Va., and his coinvestigators (JAMA Dermatol. 2017 Aug 23. doi: 10.1001/jamadermatol.2017.2083).
Among the 107 patients, the investigators found a statistically significant correlation “between worsening fibrosis scores and cumulative methotrexate” dose among women (P = .02), but not among men (P = .11). In addition, women with a BMI of 28 or more were more likely to have worsening fibrosis scores (P = .03). “There were no differences between men and women with regard to prevalence of a BMI of 28 or more, diabetes, age older than 65 years, or chronic kidney disease,” which they said, suggests that among women, “obesity influences the progression of fibrosis scores.”
The investigators advised providers not to disregard any potential warning signs when using FibroSure on men. “No differences between the cohorts were observed that would explain the association of worsening fibrosis scores and cumulative MTX dose among women but not men,” they wrote. “However, given the implications of the progression of hepatic fibrosis, we still recommend discontinuing MTX for male patients who demonstrate worsening fibrosis scores.”
While the test may not be able to replace liver biopsy, based on these results, the investigators noted that noninvasive tests such as this one could help significantly reduce the number of liver biopsies needed.
They pointed out that because the study was conducted at one center, the next step is to conduct “a prospective, randomized, multi-institutional analysis of NASH FibroSure and liver biopsies for patients with psoriasis receiving MTX vs. other treatments, including a larger cohort of men and women with different racial and ethnic backgrounds.”
One of the four authors reported receiving research funding from T2 Biosystems. Dr. Bauer and the two other authors reported no relevant financial disclosures. The study was funded by the Marshfield Clinic Resident Research Program.
ezimmerman@frontlinemedcom.com
On Twitter @eaztweets
A noninvasive test for nonalcoholic steatohepatitis (NASH) and hepatic fibrosis could be used to help detect worsening hepatic fibrosis in psoriasis patients who are on long-term methotrexate therapy, according to the authors of a retrospective study published online Aug. 23.
To evaluate the use of the noninvasive test to monitor for hepatic fibrosis in this group of patients and guide the management of methotrexate (MTX) without a liver biopsy, investigators conducted an analysis of 107 patients with psoriasis who were on long-term MTX treatment, for whom the NASH FibroSure test was used between January 2007 and December 2013. All the patients were white, fifty were men, the mean age was 83 years, and almost all of the patients had a body mass index of 28 or more (16% had a BMI between 28 and 30, and 81% had a BMI over 30).
The NASH FibroSure test, which was developed for use in patients suspected of having nonalcoholic fatty liver disease (NAFLD), combines analyses of 10 biochemical markers combined with age, sex, height, and weight to calculate the degree of hepatic fibrosis. The test has a reported sensitivity of 83% and a specificity of 78% in detecting risk of significant fibrosis, according to Bruce Bauer, MD, of Pariser Dermatology Specialists, Norfolk, Va., and his coinvestigators (JAMA Dermatol. 2017 Aug 23. doi: 10.1001/jamadermatol.2017.2083).
Among the 107 patients, the investigators found a statistically significant correlation “between worsening fibrosis scores and cumulative methotrexate” dose among women (P = .02), but not among men (P = .11). In addition, women with a BMI of 28 or more were more likely to have worsening fibrosis scores (P = .03). “There were no differences between men and women with regard to prevalence of a BMI of 28 or more, diabetes, age older than 65 years, or chronic kidney disease,” which they said, suggests that among women, “obesity influences the progression of fibrosis scores.”
The investigators advised providers not to disregard any potential warning signs when using FibroSure on men. “No differences between the cohorts were observed that would explain the association of worsening fibrosis scores and cumulative MTX dose among women but not men,” they wrote. “However, given the implications of the progression of hepatic fibrosis, we still recommend discontinuing MTX for male patients who demonstrate worsening fibrosis scores.”
While the test may not be able to replace liver biopsy, based on these results, the investigators noted that noninvasive tests such as this one could help significantly reduce the number of liver biopsies needed.
They pointed out that because the study was conducted at one center, the next step is to conduct “a prospective, randomized, multi-institutional analysis of NASH FibroSure and liver biopsies for patients with psoriasis receiving MTX vs. other treatments, including a larger cohort of men and women with different racial and ethnic backgrounds.”
One of the four authors reported receiving research funding from T2 Biosystems. Dr. Bauer and the two other authors reported no relevant financial disclosures. The study was funded by the Marshfield Clinic Resident Research Program.
ezimmerman@frontlinemedcom.com
On Twitter @eaztweets
FROM JAMA DERMATOLOGY
Key clinical point:
Major finding: There was a significant correlation between worsening fibrosis scores on the test and cumulative methotrexate dose among women (P = .02), but not among men (P = .11).
Data source: A retrospective single-center study analyzing the test in 107 psoriasis patients on methotrexate, collected between January 2007 and December 2013.
Disclosures: One of the four authors received research funding from T2 Biosystems. There were no other financial disclosures.
Psoriasis, psoriatic arthritis research makes headway at GRAPPA meeting
The agenda for this year’s Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) annual meeting included sessions on juvenile psoriatic arthritis (PsA), the microbiome in psoriasis and PsA, and setting up longitudinal cohort studies. There was also a workshop to define the clinical fit and feasibility of PsA outcome measures for clinical trials and updates on several GRAPPA-associated projects.
The meeting, held July 13-15 in Amsterdam, opened with the annual trainee session. This year’s trainee session attracted more than 40 abstracts, of which 6 were selected for oral presentations on a variety of topics including outcome measure assessment, imaging, the microbiome in PsA, and T-cell subsets in synovial fluid.
The jPsA session also emphasized the need for efforts to better define this disease entity and to study outcomes related to it. The current International League of Associations for Rheumatology criteria for juvenile idiopathic arthritis split jPsA into other subgroups (for example, enthesitis-associated arthritis, undifferentiated arthritis, etc).
Three investigators – Matt Stoll, MD, PhD, Devy Zisman, MD, and Elizabeth Mellins, MD – also presented studies of the epidemiology of jPsA.
Among the most intriguing sessions at the GRAPPA meeting was a series of three talks by Jose Scher, MD, Hok Bing Thio, MD, PhD, and Dirk Elewaut, PhD, that introduced the audience to the complexity of the micro-organisms that call the human host “home” and the potential role in the development of inflammatory conditions, in particular psoriasis and PsA. These talks touched on the potential uses of the microbiome of the gut, skin, and oral mucosa in predicting therapy response and modulating the immune system.
Dafna Gladman, MD, led a session on “how to set up a cohort.” In this session, speakers provided a road map for setting up a longitudinal cohort and discussed opportunities and challenges along the way. This session provided a foundation for a meeting that followed the annual meeting, the GRAPPA Research Collaborative Network meeting (held July 15-16). The RCN meeting aimed to develop a plan for a GRAPPA research network supporting collaborative research to identify biomarkers and outcomes in psoriasis and PsA. Speakers/panelists from academics and industry kicked off the meeting with a discussion of prior experiences in establishing international cohort studies. Subsequent sessions presented individual aspects of beginning a longitudinal cohort study for biomarkers, including methods for sample collection, regulatory processes, and data collected at potential sites; capturing and harmonizing clinical data for such studies; and policies for publication of RCN studies.
This year’s workshop was focused on the GRAPPA-Outcome Measures in Rheumatology (OMERACT) working group’s plan to develop a Core Outcome Measure Set, a collection of outcome measurement instruments to be used in randomized, controlled trials (RCTs) for PsA. During the plenary session, the team presented a process for the group to evaluate instruments, including assessment of match to the domain or concept of interest, feasibility, construct validity, and discrimination (including reliability and the ability to distinguish between two groups such as responders and nonresponders). Breakout groups then discussed one of the six tools being considered and voted on match to the domain of interest and feasibility for RCTs.
In a skin session, ongoing efforts to standardize and simplify the measurement of skin psoriasis in both the clinic and RCTs were presented. While the Psoriasis Area and Severity Index (PASI) is the current standard for measuring psoriasis severity and response to therapy in RCTs, the PASI is challenging for use in clinical practice. Joseph Merola, MD, presented data to support the use of the psoriasis Physician Global Assessment (PGA) x body surface area (BSA), a simpler measure than the PASI, as a potential substitute for the PASI. Updates from the International Dermatology Outcome Measures board included reporting of the psoriasis core domain set and a summary of the PsA symptoms working group’s efforts to identify patient-reported outcomes to identify and measure PsA among patients enrolled in psoriasis RCTs. April Armstrong, MD, discussed the National Psoriasis Foundation’s efforts to develop treat-to-target goals for psoriasis. Finally, Laura Coates, MBChB, PhD, presented a report on her efforts to examine a variety of psoriasis cut points for minimal disease activity and very low disease activity outcomes.
Dr. Ogdie is director of the Penn Psoriatic Arthritis Clinic at the University of Pennsylvania, Philadelphia, and is a member of the GRAPPA Steering Committee.
The agenda for this year’s Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) annual meeting included sessions on juvenile psoriatic arthritis (PsA), the microbiome in psoriasis and PsA, and setting up longitudinal cohort studies. There was also a workshop to define the clinical fit and feasibility of PsA outcome measures for clinical trials and updates on several GRAPPA-associated projects.
The meeting, held July 13-15 in Amsterdam, opened with the annual trainee session. This year’s trainee session attracted more than 40 abstracts, of which 6 were selected for oral presentations on a variety of topics including outcome measure assessment, imaging, the microbiome in PsA, and T-cell subsets in synovial fluid.
The jPsA session also emphasized the need for efforts to better define this disease entity and to study outcomes related to it. The current International League of Associations for Rheumatology criteria for juvenile idiopathic arthritis split jPsA into other subgroups (for example, enthesitis-associated arthritis, undifferentiated arthritis, etc).
Three investigators – Matt Stoll, MD, PhD, Devy Zisman, MD, and Elizabeth Mellins, MD – also presented studies of the epidemiology of jPsA.
Among the most intriguing sessions at the GRAPPA meeting was a series of three talks by Jose Scher, MD, Hok Bing Thio, MD, PhD, and Dirk Elewaut, PhD, that introduced the audience to the complexity of the micro-organisms that call the human host “home” and the potential role in the development of inflammatory conditions, in particular psoriasis and PsA. These talks touched on the potential uses of the microbiome of the gut, skin, and oral mucosa in predicting therapy response and modulating the immune system.
Dafna Gladman, MD, led a session on “how to set up a cohort.” In this session, speakers provided a road map for setting up a longitudinal cohort and discussed opportunities and challenges along the way. This session provided a foundation for a meeting that followed the annual meeting, the GRAPPA Research Collaborative Network meeting (held July 15-16). The RCN meeting aimed to develop a plan for a GRAPPA research network supporting collaborative research to identify biomarkers and outcomes in psoriasis and PsA. Speakers/panelists from academics and industry kicked off the meeting with a discussion of prior experiences in establishing international cohort studies. Subsequent sessions presented individual aspects of beginning a longitudinal cohort study for biomarkers, including methods for sample collection, regulatory processes, and data collected at potential sites; capturing and harmonizing clinical data for such studies; and policies for publication of RCN studies.
This year’s workshop was focused on the GRAPPA-Outcome Measures in Rheumatology (OMERACT) working group’s plan to develop a Core Outcome Measure Set, a collection of outcome measurement instruments to be used in randomized, controlled trials (RCTs) for PsA. During the plenary session, the team presented a process for the group to evaluate instruments, including assessment of match to the domain or concept of interest, feasibility, construct validity, and discrimination (including reliability and the ability to distinguish between two groups such as responders and nonresponders). Breakout groups then discussed one of the six tools being considered and voted on match to the domain of interest and feasibility for RCTs.
In a skin session, ongoing efforts to standardize and simplify the measurement of skin psoriasis in both the clinic and RCTs were presented. While the Psoriasis Area and Severity Index (PASI) is the current standard for measuring psoriasis severity and response to therapy in RCTs, the PASI is challenging for use in clinical practice. Joseph Merola, MD, presented data to support the use of the psoriasis Physician Global Assessment (PGA) x body surface area (BSA), a simpler measure than the PASI, as a potential substitute for the PASI. Updates from the International Dermatology Outcome Measures board included reporting of the psoriasis core domain set and a summary of the PsA symptoms working group’s efforts to identify patient-reported outcomes to identify and measure PsA among patients enrolled in psoriasis RCTs. April Armstrong, MD, discussed the National Psoriasis Foundation’s efforts to develop treat-to-target goals for psoriasis. Finally, Laura Coates, MBChB, PhD, presented a report on her efforts to examine a variety of psoriasis cut points for minimal disease activity and very low disease activity outcomes.
Dr. Ogdie is director of the Penn Psoriatic Arthritis Clinic at the University of Pennsylvania, Philadelphia, and is a member of the GRAPPA Steering Committee.
The agenda for this year’s Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) annual meeting included sessions on juvenile psoriatic arthritis (PsA), the microbiome in psoriasis and PsA, and setting up longitudinal cohort studies. There was also a workshop to define the clinical fit and feasibility of PsA outcome measures for clinical trials and updates on several GRAPPA-associated projects.
The meeting, held July 13-15 in Amsterdam, opened with the annual trainee session. This year’s trainee session attracted more than 40 abstracts, of which 6 were selected for oral presentations on a variety of topics including outcome measure assessment, imaging, the microbiome in PsA, and T-cell subsets in synovial fluid.
The jPsA session also emphasized the need for efforts to better define this disease entity and to study outcomes related to it. The current International League of Associations for Rheumatology criteria for juvenile idiopathic arthritis split jPsA into other subgroups (for example, enthesitis-associated arthritis, undifferentiated arthritis, etc).
Three investigators – Matt Stoll, MD, PhD, Devy Zisman, MD, and Elizabeth Mellins, MD – also presented studies of the epidemiology of jPsA.
Among the most intriguing sessions at the GRAPPA meeting was a series of three talks by Jose Scher, MD, Hok Bing Thio, MD, PhD, and Dirk Elewaut, PhD, that introduced the audience to the complexity of the micro-organisms that call the human host “home” and the potential role in the development of inflammatory conditions, in particular psoriasis and PsA. These talks touched on the potential uses of the microbiome of the gut, skin, and oral mucosa in predicting therapy response and modulating the immune system.
Dafna Gladman, MD, led a session on “how to set up a cohort.” In this session, speakers provided a road map for setting up a longitudinal cohort and discussed opportunities and challenges along the way. This session provided a foundation for a meeting that followed the annual meeting, the GRAPPA Research Collaborative Network meeting (held July 15-16). The RCN meeting aimed to develop a plan for a GRAPPA research network supporting collaborative research to identify biomarkers and outcomes in psoriasis and PsA. Speakers/panelists from academics and industry kicked off the meeting with a discussion of prior experiences in establishing international cohort studies. Subsequent sessions presented individual aspects of beginning a longitudinal cohort study for biomarkers, including methods for sample collection, regulatory processes, and data collected at potential sites; capturing and harmonizing clinical data for such studies; and policies for publication of RCN studies.
This year’s workshop was focused on the GRAPPA-Outcome Measures in Rheumatology (OMERACT) working group’s plan to develop a Core Outcome Measure Set, a collection of outcome measurement instruments to be used in randomized, controlled trials (RCTs) for PsA. During the plenary session, the team presented a process for the group to evaluate instruments, including assessment of match to the domain or concept of interest, feasibility, construct validity, and discrimination (including reliability and the ability to distinguish between two groups such as responders and nonresponders). Breakout groups then discussed one of the six tools being considered and voted on match to the domain of interest and feasibility for RCTs.
In a skin session, ongoing efforts to standardize and simplify the measurement of skin psoriasis in both the clinic and RCTs were presented. While the Psoriasis Area and Severity Index (PASI) is the current standard for measuring psoriasis severity and response to therapy in RCTs, the PASI is challenging for use in clinical practice. Joseph Merola, MD, presented data to support the use of the psoriasis Physician Global Assessment (PGA) x body surface area (BSA), a simpler measure than the PASI, as a potential substitute for the PASI. Updates from the International Dermatology Outcome Measures board included reporting of the psoriasis core domain set and a summary of the PsA symptoms working group’s efforts to identify patient-reported outcomes to identify and measure PsA among patients enrolled in psoriasis RCTs. April Armstrong, MD, discussed the National Psoriasis Foundation’s efforts to develop treat-to-target goals for psoriasis. Finally, Laura Coates, MBChB, PhD, presented a report on her efforts to examine a variety of psoriasis cut points for minimal disease activity and very low disease activity outcomes.
Dr. Ogdie is director of the Penn Psoriatic Arthritis Clinic at the University of Pennsylvania, Philadelphia, and is a member of the GRAPPA Steering Committee.
Study finds secukinumab effective for moderate to severe scalp psoriasis
Secukinumab is a safe and effective treatment option for patients with moderate to severe scalp psoriasis, according to Dr. Jerry Bagel of the Psoriasis Treatment Center of Central New Jersey, and his associates.
After 12 weeks of treatment with 300 mg of secukinumab, administered subcutaneously, 52.9% of the 50 patients who received secukinumab achieved a 90% reduction in their Psoriasis Scalp Severity Index score, and 35.3% achieved complete clearance of scalp psoriasis. Only 2% of the 47 patients in the placebo group achieved PSSI 90, and no one achieved complete clearance, in the phase 3b study.
“These promising results demonstrate the possibility of establishing PSSI 90 as a new benchmark for scalp psoriasis treatment outcome,” the investigators noted. Secukinumab (Cosentyx) is approved for moderate to severe plaque psoriasis, active psoriatic arthritis, and in adult patients who are candidates for systemic therapy or phototherapy, and ankylosing spondylitis.
Find the study in the Journal of the American Academy of Dermatology (doi: 10.1016/j.jaad.2017.05.033).
Secukinumab is a safe and effective treatment option for patients with moderate to severe scalp psoriasis, according to Dr. Jerry Bagel of the Psoriasis Treatment Center of Central New Jersey, and his associates.
After 12 weeks of treatment with 300 mg of secukinumab, administered subcutaneously, 52.9% of the 50 patients who received secukinumab achieved a 90% reduction in their Psoriasis Scalp Severity Index score, and 35.3% achieved complete clearance of scalp psoriasis. Only 2% of the 47 patients in the placebo group achieved PSSI 90, and no one achieved complete clearance, in the phase 3b study.
“These promising results demonstrate the possibility of establishing PSSI 90 as a new benchmark for scalp psoriasis treatment outcome,” the investigators noted. Secukinumab (Cosentyx) is approved for moderate to severe plaque psoriasis, active psoriatic arthritis, and in adult patients who are candidates for systemic therapy or phototherapy, and ankylosing spondylitis.
Find the study in the Journal of the American Academy of Dermatology (doi: 10.1016/j.jaad.2017.05.033).
Secukinumab is a safe and effective treatment option for patients with moderate to severe scalp psoriasis, according to Dr. Jerry Bagel of the Psoriasis Treatment Center of Central New Jersey, and his associates.
After 12 weeks of treatment with 300 mg of secukinumab, administered subcutaneously, 52.9% of the 50 patients who received secukinumab achieved a 90% reduction in their Psoriasis Scalp Severity Index score, and 35.3% achieved complete clearance of scalp psoriasis. Only 2% of the 47 patients in the placebo group achieved PSSI 90, and no one achieved complete clearance, in the phase 3b study.
“These promising results demonstrate the possibility of establishing PSSI 90 as a new benchmark for scalp psoriasis treatment outcome,” the investigators noted. Secukinumab (Cosentyx) is approved for moderate to severe plaque psoriasis, active psoriatic arthritis, and in adult patients who are candidates for systemic therapy or phototherapy, and ankylosing spondylitis.
Find the study in the Journal of the American Academy of Dermatology (doi: 10.1016/j.jaad.2017.05.033).
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Alopecia may be permanent in one in four pediatric HSCT patients
CHICAGO – Late dermatologic manifestations in children who have received hematopoietic stem cell transplants may be more common than previously thought, according to results of a new study.
Johanna Song, MD, and her collaborators reported that, in their prospective pediatric study, 25% of patients had permanent alopecia and 16% had psoriasis, noting that late nonmalignant skin effects of hematopoietic stem cell transplantation (HSCT) have been studied primarily retrospectively, and in adults. Vitiligo and nail changes were also seen.
In a poster presentation at the World Congress of Dermatology, Dr. Song and her colleagues noted that these figures are higher than the previously reported pediatric rates of 1.7% for vitiligo and 15.6% for permanent alopecia. “Early recognition of these late effects can facilitate prompt and appropriate treatment, if desired,” they said.
The single-center, cross-sectional cohort study tracked pediatric patients over an 18-month period and included patients who were at least 1 year post allogeneic HSCT and had not relapsed. Patients who were not English speaking were excluded.
The median age of the 85 patients enrolled in the study was 13.8 years, and participants were a median of 3.6 years post transplant at the time of enrollment. The study’s analysis attempted to determine which patient, transplant, and disease factors might be associated with the late nonmalignant skin changes, according to Dr. Song, a resident dermatologist at Harvard University, Boston, and her colleagues.
Most – 52– of the patients (61.2%) had hematologic malignancies; 12 patients (14.1%) received their transplant for bone marrow failure, and 11 patients (12.5%) had immunodeficiency. Three patients (3.5%) received HSCT for other malignancies, and seven (8.2%) for nonmalignant diseases.
Diffuse hair thinning was seen in 13 (62%) of the 21 patients who had alopecia, while 11 (52%) of the patients with alopecia had an androgenetic hair loss pattern. Chronic graft versus host disease (GVHD), skin chronic GVHD, a HSCT regimen that included busulfan conditioning, and a family history of early male-pattern alopecia were all significantly associated with post-HSCT permanent alopecia (P less than .05 for all).
The patients with androgenetic alopecia may be experiencing an accelerated time course of a condition to which they are already genetically disposed, noted Dr. Song and her colleagues.
Psoriasis was commonly seen on the scalp, affecting 11 of the 14 patients with psoriasis (79%), and involved the face in five of the patients (36%). Just one patient had psoriasis elsewhere on the body. There was a nonsignificant trend towards human leukocyte antigen mismatch among patients who had psoriasis. Although “psoriasis may be a marker of persistent immune dysregulation,” the investigators said, they did not identify any associated risk factors that would point toward this mechanism in their analysis.
Twelve patients (14%) had vitiligo, with halo nevi seen in four of these patients. Children who were younger than age 10 years and those who received their transplant for primary immunodeficiency were significantly more likely to have vitiligo (P less than .05 for both). Specific possible mechanisms triggering vitiligo could include thymic dysfunction resulting in loss of self-tolerance, and donor alloreactivity against the patient’s host antigens, according to the authors.
Nail changes such as pterygium and nail pitting, ridging, or thickening were seen in just five patients, all of whom had chronic GVHD of the skin. “Nail changes are likely a result of persistent inflammation and immune dysregulation from chronic GVHD,” said Dr. Song.
These late effects “can significantly impact patients’ quality of life,” according to the authors, who called for larger studies that follow pediatric HSCT patients longitudinally, beginning before the transplant – and for more investigation into the pathogenesis of specific late effects.
Dr. Song reported no relevant conflicts of interest.
koakes@frontlinemedcom.com
On Twitter @karioakes
CHICAGO – Late dermatologic manifestations in children who have received hematopoietic stem cell transplants may be more common than previously thought, according to results of a new study.
Johanna Song, MD, and her collaborators reported that, in their prospective pediatric study, 25% of patients had permanent alopecia and 16% had psoriasis, noting that late nonmalignant skin effects of hematopoietic stem cell transplantation (HSCT) have been studied primarily retrospectively, and in adults. Vitiligo and nail changes were also seen.
In a poster presentation at the World Congress of Dermatology, Dr. Song and her colleagues noted that these figures are higher than the previously reported pediatric rates of 1.7% for vitiligo and 15.6% for permanent alopecia. “Early recognition of these late effects can facilitate prompt and appropriate treatment, if desired,” they said.
The single-center, cross-sectional cohort study tracked pediatric patients over an 18-month period and included patients who were at least 1 year post allogeneic HSCT and had not relapsed. Patients who were not English speaking were excluded.
The median age of the 85 patients enrolled in the study was 13.8 years, and participants were a median of 3.6 years post transplant at the time of enrollment. The study’s analysis attempted to determine which patient, transplant, and disease factors might be associated with the late nonmalignant skin changes, according to Dr. Song, a resident dermatologist at Harvard University, Boston, and her colleagues.
Most – 52– of the patients (61.2%) had hematologic malignancies; 12 patients (14.1%) received their transplant for bone marrow failure, and 11 patients (12.5%) had immunodeficiency. Three patients (3.5%) received HSCT for other malignancies, and seven (8.2%) for nonmalignant diseases.
Diffuse hair thinning was seen in 13 (62%) of the 21 patients who had alopecia, while 11 (52%) of the patients with alopecia had an androgenetic hair loss pattern. Chronic graft versus host disease (GVHD), skin chronic GVHD, a HSCT regimen that included busulfan conditioning, and a family history of early male-pattern alopecia were all significantly associated with post-HSCT permanent alopecia (P less than .05 for all).
The patients with androgenetic alopecia may be experiencing an accelerated time course of a condition to which they are already genetically disposed, noted Dr. Song and her colleagues.
Psoriasis was commonly seen on the scalp, affecting 11 of the 14 patients with psoriasis (79%), and involved the face in five of the patients (36%). Just one patient had psoriasis elsewhere on the body. There was a nonsignificant trend towards human leukocyte antigen mismatch among patients who had psoriasis. Although “psoriasis may be a marker of persistent immune dysregulation,” the investigators said, they did not identify any associated risk factors that would point toward this mechanism in their analysis.
Twelve patients (14%) had vitiligo, with halo nevi seen in four of these patients. Children who were younger than age 10 years and those who received their transplant for primary immunodeficiency were significantly more likely to have vitiligo (P less than .05 for both). Specific possible mechanisms triggering vitiligo could include thymic dysfunction resulting in loss of self-tolerance, and donor alloreactivity against the patient’s host antigens, according to the authors.
Nail changes such as pterygium and nail pitting, ridging, or thickening were seen in just five patients, all of whom had chronic GVHD of the skin. “Nail changes are likely a result of persistent inflammation and immune dysregulation from chronic GVHD,” said Dr. Song.
These late effects “can significantly impact patients’ quality of life,” according to the authors, who called for larger studies that follow pediatric HSCT patients longitudinally, beginning before the transplant – and for more investigation into the pathogenesis of specific late effects.
Dr. Song reported no relevant conflicts of interest.
koakes@frontlinemedcom.com
On Twitter @karioakes
CHICAGO – Late dermatologic manifestations in children who have received hematopoietic stem cell transplants may be more common than previously thought, according to results of a new study.
Johanna Song, MD, and her collaborators reported that, in their prospective pediatric study, 25% of patients had permanent alopecia and 16% had psoriasis, noting that late nonmalignant skin effects of hematopoietic stem cell transplantation (HSCT) have been studied primarily retrospectively, and in adults. Vitiligo and nail changes were also seen.
In a poster presentation at the World Congress of Dermatology, Dr. Song and her colleagues noted that these figures are higher than the previously reported pediatric rates of 1.7% for vitiligo and 15.6% for permanent alopecia. “Early recognition of these late effects can facilitate prompt and appropriate treatment, if desired,” they said.
The single-center, cross-sectional cohort study tracked pediatric patients over an 18-month period and included patients who were at least 1 year post allogeneic HSCT and had not relapsed. Patients who were not English speaking were excluded.
The median age of the 85 patients enrolled in the study was 13.8 years, and participants were a median of 3.6 years post transplant at the time of enrollment. The study’s analysis attempted to determine which patient, transplant, and disease factors might be associated with the late nonmalignant skin changes, according to Dr. Song, a resident dermatologist at Harvard University, Boston, and her colleagues.
Most – 52– of the patients (61.2%) had hematologic malignancies; 12 patients (14.1%) received their transplant for bone marrow failure, and 11 patients (12.5%) had immunodeficiency. Three patients (3.5%) received HSCT for other malignancies, and seven (8.2%) for nonmalignant diseases.
Diffuse hair thinning was seen in 13 (62%) of the 21 patients who had alopecia, while 11 (52%) of the patients with alopecia had an androgenetic hair loss pattern. Chronic graft versus host disease (GVHD), skin chronic GVHD, a HSCT regimen that included busulfan conditioning, and a family history of early male-pattern alopecia were all significantly associated with post-HSCT permanent alopecia (P less than .05 for all).
The patients with androgenetic alopecia may be experiencing an accelerated time course of a condition to which they are already genetically disposed, noted Dr. Song and her colleagues.
Psoriasis was commonly seen on the scalp, affecting 11 of the 14 patients with psoriasis (79%), and involved the face in five of the patients (36%). Just one patient had psoriasis elsewhere on the body. There was a nonsignificant trend towards human leukocyte antigen mismatch among patients who had psoriasis. Although “psoriasis may be a marker of persistent immune dysregulation,” the investigators said, they did not identify any associated risk factors that would point toward this mechanism in their analysis.
Twelve patients (14%) had vitiligo, with halo nevi seen in four of these patients. Children who were younger than age 10 years and those who received their transplant for primary immunodeficiency were significantly more likely to have vitiligo (P less than .05 for both). Specific possible mechanisms triggering vitiligo could include thymic dysfunction resulting in loss of self-tolerance, and donor alloreactivity against the patient’s host antigens, according to the authors.
Nail changes such as pterygium and nail pitting, ridging, or thickening were seen in just five patients, all of whom had chronic GVHD of the skin. “Nail changes are likely a result of persistent inflammation and immune dysregulation from chronic GVHD,” said Dr. Song.
These late effects “can significantly impact patients’ quality of life,” according to the authors, who called for larger studies that follow pediatric HSCT patients longitudinally, beginning before the transplant – and for more investigation into the pathogenesis of specific late effects.
Dr. Song reported no relevant conflicts of interest.
koakes@frontlinemedcom.com
On Twitter @karioakes
AT WCPD 2017
Key clinical point:
Major finding: Permanent alopecia was seen in 25% of patients, and 16% had psoriasis a median of 3.6 years after transplant.
Data source: A single-center prospective study of 85 children in routine post-HSCT follow-up.
Disclosures: Dr. Song reported no conflicts of interest.
Adalimumab for Hidradenitis Suppurativa
We applaud Kimball et al1 on their report that adalimumab demonstrated clinical improvement in patients with hidradenitis suppurativa (HS) versus placebo in 2 phase 3 trials. Hidradenitis suppurativa is a chronic relapsing condition with painful subcutaneous abscesses, malodorous drainage, sinus tract formation, and scarring that typically occurs in the axillae and anogenital region. It impairs the quality of life for these patients, as evidenced by higher Dermatology Life Quality Index scores compared to psoriasis, pimples, hand rash, atopic eczema, or control.2
The exact pathogenesis of HS is unknown but likely involves a complex interaction of genetic, hormonal, immunologic, and environmental factors.3 The levels of inflammatory cytokines are elevated in HS lesions, specifically IL-1β, tumor necrosis factor α, IL-10, and CXCL9, as well as monokines from IFN-γ, IL-11, and IL-17A. Additionally, the dermis of affected regions contains IL-12– and IL-23–containing macrophages along with IL-17–producing T cells.3 These findings reveal many potential therapeutic targets for the treatment of HS.
PIONEER I and PIONEER II are similarly designed 36-week phase 3 trials of 633 patients with HS who were unresponsive to oral antibiotic treatment.1 By week 12, a significantly greater proportion of patients receiving adalimumab demonstrated clinical improvement (≥50% reduction in total abscess and nodule count) compared to placebo in both trials (PIONEER I: 41.8% vs 26.0%, P=.003; PIONEER II: 58.9% vs 27.6%, P<.001). Secondary end points (inflammatory-nodule count, pain score, and disease severity) were only achieved in PIONEER II. The difference in clinical improvement between the trials is likely due to higher baseline disease severity in the HS patients in PIONEER I versus PIONEER II. No new safety risks were reported and were in accordance with prior adalimumab trials for other diseases. Notably, 10 paradoxical psoriasislike eruptions were reported.
Adalimumab is the first and only US Food and Drug Administration–approved therapy for HS. Further understanding of the pathogenesis of HS may result in additional biologic treatments for HS. We encourage the manufacturers of other biologic therapies, such as infliximab,4 ustekinumab,5 anakinra,6 secukinumab, ixekizumab, and brodalumab, to consider conducting further clinical trials in HS to enhance the therapeutic options available for this debilitating disease.
- Kimball AB, Okun MM, Williams DA, et al. Two Phase 3 trials of adalimumab for hidradenitis suppurativa. N Engl J Med. 2016;375:422-434.
- Vinding GR, Knudsen KM, Ellervik C, et al. Self-reported skin morbidities and health-related quality of life: a population-based nested case-control study. Dermatology. 2014;228:261-268.
- Deckers IE, van der Zee HH, Prens EP. Epidemiology of hidradenitis suppurativa: prevalence, pathogenesis, and factors associated with the development of HS. Curr Dermatol Rep. 2014;3:54-60.
- Ingram JR, Woo PN, Chua SL, et al. Interventions for hidradenitis suppurativa: a Cochrane systematic review incorporating GRADE assessment of evidence quality. Br J Dermatol. 2016;174:970-978.
- Blok JL, Li K, Brodmerkel C, et al. Ustekinumab in hidradenitis suppurativa: clinical results and a search for potential biomarkers in serum. Br J Dermatol. 2016;174:839-846.
- Tzanetakou V, Kanni T, Giatrakou S, et al. Safety and efficacy of anakinra in severe hidradenitis suppurativa: a randomized clinical trial. JAMA Dermatol. 2016;152:52-59.
We applaud Kimball et al1 on their report that adalimumab demonstrated clinical improvement in patients with hidradenitis suppurativa (HS) versus placebo in 2 phase 3 trials. Hidradenitis suppurativa is a chronic relapsing condition with painful subcutaneous abscesses, malodorous drainage, sinus tract formation, and scarring that typically occurs in the axillae and anogenital region. It impairs the quality of life for these patients, as evidenced by higher Dermatology Life Quality Index scores compared to psoriasis, pimples, hand rash, atopic eczema, or control.2
The exact pathogenesis of HS is unknown but likely involves a complex interaction of genetic, hormonal, immunologic, and environmental factors.3 The levels of inflammatory cytokines are elevated in HS lesions, specifically IL-1β, tumor necrosis factor α, IL-10, and CXCL9, as well as monokines from IFN-γ, IL-11, and IL-17A. Additionally, the dermis of affected regions contains IL-12– and IL-23–containing macrophages along with IL-17–producing T cells.3 These findings reveal many potential therapeutic targets for the treatment of HS.
PIONEER I and PIONEER II are similarly designed 36-week phase 3 trials of 633 patients with HS who were unresponsive to oral antibiotic treatment.1 By week 12, a significantly greater proportion of patients receiving adalimumab demonstrated clinical improvement (≥50% reduction in total abscess and nodule count) compared to placebo in both trials (PIONEER I: 41.8% vs 26.0%, P=.003; PIONEER II: 58.9% vs 27.6%, P<.001). Secondary end points (inflammatory-nodule count, pain score, and disease severity) were only achieved in PIONEER II. The difference in clinical improvement between the trials is likely due to higher baseline disease severity in the HS patients in PIONEER I versus PIONEER II. No new safety risks were reported and were in accordance with prior adalimumab trials for other diseases. Notably, 10 paradoxical psoriasislike eruptions were reported.
Adalimumab is the first and only US Food and Drug Administration–approved therapy for HS. Further understanding of the pathogenesis of HS may result in additional biologic treatments for HS. We encourage the manufacturers of other biologic therapies, such as infliximab,4 ustekinumab,5 anakinra,6 secukinumab, ixekizumab, and brodalumab, to consider conducting further clinical trials in HS to enhance the therapeutic options available for this debilitating disease.
We applaud Kimball et al1 on their report that adalimumab demonstrated clinical improvement in patients with hidradenitis suppurativa (HS) versus placebo in 2 phase 3 trials. Hidradenitis suppurativa is a chronic relapsing condition with painful subcutaneous abscesses, malodorous drainage, sinus tract formation, and scarring that typically occurs in the axillae and anogenital region. It impairs the quality of life for these patients, as evidenced by higher Dermatology Life Quality Index scores compared to psoriasis, pimples, hand rash, atopic eczema, or control.2
The exact pathogenesis of HS is unknown but likely involves a complex interaction of genetic, hormonal, immunologic, and environmental factors.3 The levels of inflammatory cytokines are elevated in HS lesions, specifically IL-1β, tumor necrosis factor α, IL-10, and CXCL9, as well as monokines from IFN-γ, IL-11, and IL-17A. Additionally, the dermis of affected regions contains IL-12– and IL-23–containing macrophages along with IL-17–producing T cells.3 These findings reveal many potential therapeutic targets for the treatment of HS.
PIONEER I and PIONEER II are similarly designed 36-week phase 3 trials of 633 patients with HS who were unresponsive to oral antibiotic treatment.1 By week 12, a significantly greater proportion of patients receiving adalimumab demonstrated clinical improvement (≥50% reduction in total abscess and nodule count) compared to placebo in both trials (PIONEER I: 41.8% vs 26.0%, P=.003; PIONEER II: 58.9% vs 27.6%, P<.001). Secondary end points (inflammatory-nodule count, pain score, and disease severity) were only achieved in PIONEER II. The difference in clinical improvement between the trials is likely due to higher baseline disease severity in the HS patients in PIONEER I versus PIONEER II. No new safety risks were reported and were in accordance with prior adalimumab trials for other diseases. Notably, 10 paradoxical psoriasislike eruptions were reported.
Adalimumab is the first and only US Food and Drug Administration–approved therapy for HS. Further understanding of the pathogenesis of HS may result in additional biologic treatments for HS. We encourage the manufacturers of other biologic therapies, such as infliximab,4 ustekinumab,5 anakinra,6 secukinumab, ixekizumab, and brodalumab, to consider conducting further clinical trials in HS to enhance the therapeutic options available for this debilitating disease.
- Kimball AB, Okun MM, Williams DA, et al. Two Phase 3 trials of adalimumab for hidradenitis suppurativa. N Engl J Med. 2016;375:422-434.
- Vinding GR, Knudsen KM, Ellervik C, et al. Self-reported skin morbidities and health-related quality of life: a population-based nested case-control study. Dermatology. 2014;228:261-268.
- Deckers IE, van der Zee HH, Prens EP. Epidemiology of hidradenitis suppurativa: prevalence, pathogenesis, and factors associated with the development of HS. Curr Dermatol Rep. 2014;3:54-60.
- Ingram JR, Woo PN, Chua SL, et al. Interventions for hidradenitis suppurativa: a Cochrane systematic review incorporating GRADE assessment of evidence quality. Br J Dermatol. 2016;174:970-978.
- Blok JL, Li K, Brodmerkel C, et al. Ustekinumab in hidradenitis suppurativa: clinical results and a search for potential biomarkers in serum. Br J Dermatol. 2016;174:839-846.
- Tzanetakou V, Kanni T, Giatrakou S, et al. Safety and efficacy of anakinra in severe hidradenitis suppurativa: a randomized clinical trial. JAMA Dermatol. 2016;152:52-59.
- Kimball AB, Okun MM, Williams DA, et al. Two Phase 3 trials of adalimumab for hidradenitis suppurativa. N Engl J Med. 2016;375:422-434.
- Vinding GR, Knudsen KM, Ellervik C, et al. Self-reported skin morbidities and health-related quality of life: a population-based nested case-control study. Dermatology. 2014;228:261-268.
- Deckers IE, van der Zee HH, Prens EP. Epidemiology of hidradenitis suppurativa: prevalence, pathogenesis, and factors associated with the development of HS. Curr Dermatol Rep. 2014;3:54-60.
- Ingram JR, Woo PN, Chua SL, et al. Interventions for hidradenitis suppurativa: a Cochrane systematic review incorporating GRADE assessment of evidence quality. Br J Dermatol. 2016;174:970-978.
- Blok JL, Li K, Brodmerkel C, et al. Ustekinumab in hidradenitis suppurativa: clinical results and a search for potential biomarkers in serum. Br J Dermatol. 2016;174:839-846.
- Tzanetakou V, Kanni T, Giatrakou S, et al. Safety and efficacy of anakinra in severe hidradenitis suppurativa: a randomized clinical trial. JAMA Dermatol. 2016;152:52-59.