User login
Biosimilars poised to save $54 billion over the next decade
Biosimilars could reduce overall spending on biologic products by $54 billion from 2017 to 2026, according to new research from the Rand Corp.
Given the level of uncertainty surrounding the biosimilars market, however, the range of savings could be as low at $24 billion or as high as $150 billion.
“Because of limited U.S. experience with biosimilars, the key assumptions on market share and biosimilar prices are ‘best guesses’ based on anecdotes or professional opinion,” Andrew Mulcahy, PhD, a health policy researcher at Rand, and his colleagues, wrote in a perspective report.
“Whether actual cost savings end up above or below our baseline estimate hinges in large part on whether manufacturers continue to have a business case to invest in developing and marketing biosimilars,” the authors noted, citing a number of areas, including intellectual property litigation, payment, price competition, nonprice competition from reference biologic manufacturers, naming convention, and interchangeability.
Getting over these hurdles could require legislative or regulatory solutions.
“The pervasive uncertainty in the U.S. biosimilar market – including questions as to whether the market will be sustainable and lead to cost savings, as intended – presents two choices for policymakers,” Dr. Mulcahy and his colleagues wrote. “One strategy is to let the market continue to develop under current policies,” with stability coming from experience.
The alternative could be policy levers to “help steer the U.S. biosimilar market more quickly to a sustainable, competitive state,” they continued. “For example, regulators at the FDA could experiment with new approaches to provide stronger, earlier signals through guidance documents or other mechanisms on expectations surrounding interchangeability and other topics.”
The FDA appears to be moving on the latter. In an Oct. 23 blog post, FDA Commissioner Scott Gottlieb, MD, and Leah Christl, PhD, associate director for therapeutic biologics in the office of new drugs at the FDA’s Center for Drug Evaluation and Research, outlined a number of recent tools to help biosimilar adoption. The resources provide basics such as the basic definition associated with biosimilars (i.e., what is a biosimilar and a reference product, and what it means to be interchangeable), the standards of approval that biosimilars must go through, and easily accessible information on what the FDA is using to review biosimilarity.
“Next, FDA plans to embark on additional research with health care professionals to learn more about the types of information prescribers need to properly communicate with their patients about biosimilars,” Commissioner Gottlieb and Dr. Christl wrote. “An increase in market competition, offered by a growing complement of biosimilars, may lead to meaningfully reduced costs for both patients and our health care system.”
The Centers for Medicare & Medicaid Services also plays a role in developing policy to spur biosimilar adoption, Dr. Mulcahy and his colleagues wrote. They note work being done by the Medicare Payment Advisory Commission on recommendations that could address payment for physician-administered biosimilars under Part B, as well as incentives in the Part D prescription drug program to steer patients and providers toward lower cost biosimilars when appropriate. CMS changed current payment policy for biosimilars for 2018, which may have an effect.
“Beyond FDA regulation, payment, and coverage, both government and industry could play a role in educating patients and providers about the potential cost savings from biosimilars, much like both groups have done for generic drugs,” they stated. “While our study does not address whether policy action is needed now, it is likely that the answer will become clearer over the next 1 to 3 years as the market continues to develop.”
Biosimilars could reduce overall spending on biologic products by $54 billion from 2017 to 2026, according to new research from the Rand Corp.
Given the level of uncertainty surrounding the biosimilars market, however, the range of savings could be as low at $24 billion or as high as $150 billion.
“Because of limited U.S. experience with biosimilars, the key assumptions on market share and biosimilar prices are ‘best guesses’ based on anecdotes or professional opinion,” Andrew Mulcahy, PhD, a health policy researcher at Rand, and his colleagues, wrote in a perspective report.
“Whether actual cost savings end up above or below our baseline estimate hinges in large part on whether manufacturers continue to have a business case to invest in developing and marketing biosimilars,” the authors noted, citing a number of areas, including intellectual property litigation, payment, price competition, nonprice competition from reference biologic manufacturers, naming convention, and interchangeability.
Getting over these hurdles could require legislative or regulatory solutions.
“The pervasive uncertainty in the U.S. biosimilar market – including questions as to whether the market will be sustainable and lead to cost savings, as intended – presents two choices for policymakers,” Dr. Mulcahy and his colleagues wrote. “One strategy is to let the market continue to develop under current policies,” with stability coming from experience.
The alternative could be policy levers to “help steer the U.S. biosimilar market more quickly to a sustainable, competitive state,” they continued. “For example, regulators at the FDA could experiment with new approaches to provide stronger, earlier signals through guidance documents or other mechanisms on expectations surrounding interchangeability and other topics.”
The FDA appears to be moving on the latter. In an Oct. 23 blog post, FDA Commissioner Scott Gottlieb, MD, and Leah Christl, PhD, associate director for therapeutic biologics in the office of new drugs at the FDA’s Center for Drug Evaluation and Research, outlined a number of recent tools to help biosimilar adoption. The resources provide basics such as the basic definition associated with biosimilars (i.e., what is a biosimilar and a reference product, and what it means to be interchangeable), the standards of approval that biosimilars must go through, and easily accessible information on what the FDA is using to review biosimilarity.
“Next, FDA plans to embark on additional research with health care professionals to learn more about the types of information prescribers need to properly communicate with their patients about biosimilars,” Commissioner Gottlieb and Dr. Christl wrote. “An increase in market competition, offered by a growing complement of biosimilars, may lead to meaningfully reduced costs for both patients and our health care system.”
The Centers for Medicare & Medicaid Services also plays a role in developing policy to spur biosimilar adoption, Dr. Mulcahy and his colleagues wrote. They note work being done by the Medicare Payment Advisory Commission on recommendations that could address payment for physician-administered biosimilars under Part B, as well as incentives in the Part D prescription drug program to steer patients and providers toward lower cost biosimilars when appropriate. CMS changed current payment policy for biosimilars for 2018, which may have an effect.
“Beyond FDA regulation, payment, and coverage, both government and industry could play a role in educating patients and providers about the potential cost savings from biosimilars, much like both groups have done for generic drugs,” they stated. “While our study does not address whether policy action is needed now, it is likely that the answer will become clearer over the next 1 to 3 years as the market continues to develop.”
Biosimilars could reduce overall spending on biologic products by $54 billion from 2017 to 2026, according to new research from the Rand Corp.
Given the level of uncertainty surrounding the biosimilars market, however, the range of savings could be as low at $24 billion or as high as $150 billion.
“Because of limited U.S. experience with biosimilars, the key assumptions on market share and biosimilar prices are ‘best guesses’ based on anecdotes or professional opinion,” Andrew Mulcahy, PhD, a health policy researcher at Rand, and his colleagues, wrote in a perspective report.
“Whether actual cost savings end up above or below our baseline estimate hinges in large part on whether manufacturers continue to have a business case to invest in developing and marketing biosimilars,” the authors noted, citing a number of areas, including intellectual property litigation, payment, price competition, nonprice competition from reference biologic manufacturers, naming convention, and interchangeability.
Getting over these hurdles could require legislative or regulatory solutions.
“The pervasive uncertainty in the U.S. biosimilar market – including questions as to whether the market will be sustainable and lead to cost savings, as intended – presents two choices for policymakers,” Dr. Mulcahy and his colleagues wrote. “One strategy is to let the market continue to develop under current policies,” with stability coming from experience.
The alternative could be policy levers to “help steer the U.S. biosimilar market more quickly to a sustainable, competitive state,” they continued. “For example, regulators at the FDA could experiment with new approaches to provide stronger, earlier signals through guidance documents or other mechanisms on expectations surrounding interchangeability and other topics.”
The FDA appears to be moving on the latter. In an Oct. 23 blog post, FDA Commissioner Scott Gottlieb, MD, and Leah Christl, PhD, associate director for therapeutic biologics in the office of new drugs at the FDA’s Center for Drug Evaluation and Research, outlined a number of recent tools to help biosimilar adoption. The resources provide basics such as the basic definition associated with biosimilars (i.e., what is a biosimilar and a reference product, and what it means to be interchangeable), the standards of approval that biosimilars must go through, and easily accessible information on what the FDA is using to review biosimilarity.
“Next, FDA plans to embark on additional research with health care professionals to learn more about the types of information prescribers need to properly communicate with their patients about biosimilars,” Commissioner Gottlieb and Dr. Christl wrote. “An increase in market competition, offered by a growing complement of biosimilars, may lead to meaningfully reduced costs for both patients and our health care system.”
The Centers for Medicare & Medicaid Services also plays a role in developing policy to spur biosimilar adoption, Dr. Mulcahy and his colleagues wrote. They note work being done by the Medicare Payment Advisory Commission on recommendations that could address payment for physician-administered biosimilars under Part B, as well as incentives in the Part D prescription drug program to steer patients and providers toward lower cost biosimilars when appropriate. CMS changed current payment policy for biosimilars for 2018, which may have an effect.
“Beyond FDA regulation, payment, and coverage, both government and industry could play a role in educating patients and providers about the potential cost savings from biosimilars, much like both groups have done for generic drugs,” they stated. “While our study does not address whether policy action is needed now, it is likely that the answer will become clearer over the next 1 to 3 years as the market continues to develop.”
Psoriasis: Biologics bring potential for long-term remission off treatment
GENEVA – Can potent, highly efficacious biologic therapy induce longstanding remission of moderate to severe plaque psoriasis following discontinuation of all treatment?
The answer appears to be yes, in a minority of patients, based on results of a long-term extension study of two pivotal phase 3 clinical trials of secukinumab (Cosentyx), Mark G. Lebwohl, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.

This 2-year extension study provides the first double-blind, long-term evidence regarding the natural history of psoriasis following discontinuation of a contemporary potent targeted therapy.
The results are exciting because spontaneous remission of moderate to severe psoriasis is rare; psoriasis typically reverts to baseline severity following discontinuation of treatment. In the extension study, though, 21% of patients who achieved a 75% reduction in the Psoriasis Area Severity Index (PASI 75) response on secukinumab at the 300-mg dose for 52 weeks and were then crossed over double-blind to placebo remained continuously relapse-free after 12 months on placebo, and 10% remained relapse-free at 2 years, at which point they were unblinded and put back on secukinumab. No psoriasis therapies were permitted during the placebo phase.
Those findings are particularly impressive because participants in the two pivotal trials of secukinumab combined for the treatment withdrawal extension study – ERASURE and FIXTURE – had a mean 14- and 16-year history of psoriasis at baseline.
“Secukinumab appears to have modified the course of chronic moderate to severe psoriasis, despite intervening late in its course. Treating patients early is expected to have an even greater potential for disease modification,” commented Dr. Lebwohl, the Sol and Clara Kest Professor and chair of the department of dermatology at Icahn School of Medicine at Mount Sinai, New York.
That hypothesis is under investigation in the ongoing multinational STEPin trial (Study of the Efficacy of Early Intervention With Secukinumab 300 mg s.c. [subcutaneous] Compared to Narrow-band UVB in Patients With New-onset Moderate to Severe Plaque Psoriasis). STEPin involves about 200 patients with newly diagnosed disease not previously treated with systemic agents or phototherapy.
Dr. Lebwohl focused on the 120 patients in the extension study who had a PASI 75 response to secukinumab at the 300-mg dose and the 100 patients with a PASI 75 response to the 150-mg dose who, after 52 weeks on the fully human anti-interleukin-17A monoclonal antibody, were crossed over double-blind to placebo for 2 years or until relapse occurred. Patients were assessed monthly during the extension study.
Relapse was defined as loss of 50% of the maximum PASI improvement achieved while on secukinumab. The relapse-free rate was better in patients who had been on secukinumab 300 mg than in those on 150 mg. At 1 year on placebo, 21% of patients formerly on secukinumab at the higher dose remained free of relapse, as did 10% at 2 years. In contrast, the 1- and 2-year relapse-free rates in the group formerly on secukinumab 150 mg were lower at 14% and 6%, respectively, even though everyone in the extension study had a PASI 75 response when they went off treatment.
The mean baseline PASI score in the ERASURE and FIXTURE trials was 23. Among patients who remained relapse-free for 1 year, the mean PASI score was dramatically lower, at 0.7; among those in remission for 2 years, it was 0.4.
Importantly, the longer a patient’s history of psoriasis, the less likely a long-term relapse-free experience, the dermatologist observed.
A separate mechanistic study in a different group of psoriasis patients provided a likely explanation for the long relapse-free period seen off therapy in some patients in the extension study. The mechanistic study entailed transcriptional analysis of psoriasis-related genes in lesional and nonlesional skin before and after 1 year of treatment with secukinumab 300 mg. The major finding: 85% of the upregulated genes and 75% of downregulated genes in lesional skin pretreatment reverted to normal levels in healed lesional skin post treatment.
Moreover, global mRNA expression for key genes involved in the interleukin-23/interleukin-17 inflammatory cytokine pathway closely resembled healthy skin following treatment with secukinumab. This is evidence of profound molecular normalization, Dr. Lebwohl said.
These secukinumab studies were sponsored by Novartis. Dr. Lebwohl reported having no personal financial conflicts of interest, although his department receives research funding from Novartis and numerous other pharmaceutical companies.
GENEVA – Can potent, highly efficacious biologic therapy induce longstanding remission of moderate to severe plaque psoriasis following discontinuation of all treatment?
The answer appears to be yes, in a minority of patients, based on results of a long-term extension study of two pivotal phase 3 clinical trials of secukinumab (Cosentyx), Mark G. Lebwohl, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.

This 2-year extension study provides the first double-blind, long-term evidence regarding the natural history of psoriasis following discontinuation of a contemporary potent targeted therapy.
The results are exciting because spontaneous remission of moderate to severe psoriasis is rare; psoriasis typically reverts to baseline severity following discontinuation of treatment. In the extension study, though, 21% of patients who achieved a 75% reduction in the Psoriasis Area Severity Index (PASI 75) response on secukinumab at the 300-mg dose for 52 weeks and were then crossed over double-blind to placebo remained continuously relapse-free after 12 months on placebo, and 10% remained relapse-free at 2 years, at which point they were unblinded and put back on secukinumab. No psoriasis therapies were permitted during the placebo phase.
Those findings are particularly impressive because participants in the two pivotal trials of secukinumab combined for the treatment withdrawal extension study – ERASURE and FIXTURE – had a mean 14- and 16-year history of psoriasis at baseline.
“Secukinumab appears to have modified the course of chronic moderate to severe psoriasis, despite intervening late in its course. Treating patients early is expected to have an even greater potential for disease modification,” commented Dr. Lebwohl, the Sol and Clara Kest Professor and chair of the department of dermatology at Icahn School of Medicine at Mount Sinai, New York.
That hypothesis is under investigation in the ongoing multinational STEPin trial (Study of the Efficacy of Early Intervention With Secukinumab 300 mg s.c. [subcutaneous] Compared to Narrow-band UVB in Patients With New-onset Moderate to Severe Plaque Psoriasis). STEPin involves about 200 patients with newly diagnosed disease not previously treated with systemic agents or phototherapy.
Dr. Lebwohl focused on the 120 patients in the extension study who had a PASI 75 response to secukinumab at the 300-mg dose and the 100 patients with a PASI 75 response to the 150-mg dose who, after 52 weeks on the fully human anti-interleukin-17A monoclonal antibody, were crossed over double-blind to placebo for 2 years or until relapse occurred. Patients were assessed monthly during the extension study.
Relapse was defined as loss of 50% of the maximum PASI improvement achieved while on secukinumab. The relapse-free rate was better in patients who had been on secukinumab 300 mg than in those on 150 mg. At 1 year on placebo, 21% of patients formerly on secukinumab at the higher dose remained free of relapse, as did 10% at 2 years. In contrast, the 1- and 2-year relapse-free rates in the group formerly on secukinumab 150 mg were lower at 14% and 6%, respectively, even though everyone in the extension study had a PASI 75 response when they went off treatment.
The mean baseline PASI score in the ERASURE and FIXTURE trials was 23. Among patients who remained relapse-free for 1 year, the mean PASI score was dramatically lower, at 0.7; among those in remission for 2 years, it was 0.4.
Importantly, the longer a patient’s history of psoriasis, the less likely a long-term relapse-free experience, the dermatologist observed.
A separate mechanistic study in a different group of psoriasis patients provided a likely explanation for the long relapse-free period seen off therapy in some patients in the extension study. The mechanistic study entailed transcriptional analysis of psoriasis-related genes in lesional and nonlesional skin before and after 1 year of treatment with secukinumab 300 mg. The major finding: 85% of the upregulated genes and 75% of downregulated genes in lesional skin pretreatment reverted to normal levels in healed lesional skin post treatment.
Moreover, global mRNA expression for key genes involved in the interleukin-23/interleukin-17 inflammatory cytokine pathway closely resembled healthy skin following treatment with secukinumab. This is evidence of profound molecular normalization, Dr. Lebwohl said.
These secukinumab studies were sponsored by Novartis. Dr. Lebwohl reported having no personal financial conflicts of interest, although his department receives research funding from Novartis and numerous other pharmaceutical companies.
GENEVA – Can potent, highly efficacious biologic therapy induce longstanding remission of moderate to severe plaque psoriasis following discontinuation of all treatment?
The answer appears to be yes, in a minority of patients, based on results of a long-term extension study of two pivotal phase 3 clinical trials of secukinumab (Cosentyx), Mark G. Lebwohl, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.

This 2-year extension study provides the first double-blind, long-term evidence regarding the natural history of psoriasis following discontinuation of a contemporary potent targeted therapy.
The results are exciting because spontaneous remission of moderate to severe psoriasis is rare; psoriasis typically reverts to baseline severity following discontinuation of treatment. In the extension study, though, 21% of patients who achieved a 75% reduction in the Psoriasis Area Severity Index (PASI 75) response on secukinumab at the 300-mg dose for 52 weeks and were then crossed over double-blind to placebo remained continuously relapse-free after 12 months on placebo, and 10% remained relapse-free at 2 years, at which point they were unblinded and put back on secukinumab. No psoriasis therapies were permitted during the placebo phase.
Those findings are particularly impressive because participants in the two pivotal trials of secukinumab combined for the treatment withdrawal extension study – ERASURE and FIXTURE – had a mean 14- and 16-year history of psoriasis at baseline.
“Secukinumab appears to have modified the course of chronic moderate to severe psoriasis, despite intervening late in its course. Treating patients early is expected to have an even greater potential for disease modification,” commented Dr. Lebwohl, the Sol and Clara Kest Professor and chair of the department of dermatology at Icahn School of Medicine at Mount Sinai, New York.
That hypothesis is under investigation in the ongoing multinational STEPin trial (Study of the Efficacy of Early Intervention With Secukinumab 300 mg s.c. [subcutaneous] Compared to Narrow-band UVB in Patients With New-onset Moderate to Severe Plaque Psoriasis). STEPin involves about 200 patients with newly diagnosed disease not previously treated with systemic agents or phototherapy.
Dr. Lebwohl focused on the 120 patients in the extension study who had a PASI 75 response to secukinumab at the 300-mg dose and the 100 patients with a PASI 75 response to the 150-mg dose who, after 52 weeks on the fully human anti-interleukin-17A monoclonal antibody, were crossed over double-blind to placebo for 2 years or until relapse occurred. Patients were assessed monthly during the extension study.
Relapse was defined as loss of 50% of the maximum PASI improvement achieved while on secukinumab. The relapse-free rate was better in patients who had been on secukinumab 300 mg than in those on 150 mg. At 1 year on placebo, 21% of patients formerly on secukinumab at the higher dose remained free of relapse, as did 10% at 2 years. In contrast, the 1- and 2-year relapse-free rates in the group formerly on secukinumab 150 mg were lower at 14% and 6%, respectively, even though everyone in the extension study had a PASI 75 response when they went off treatment.
The mean baseline PASI score in the ERASURE and FIXTURE trials was 23. Among patients who remained relapse-free for 1 year, the mean PASI score was dramatically lower, at 0.7; among those in remission for 2 years, it was 0.4.
Importantly, the longer a patient’s history of psoriasis, the less likely a long-term relapse-free experience, the dermatologist observed.
A separate mechanistic study in a different group of psoriasis patients provided a likely explanation for the long relapse-free period seen off therapy in some patients in the extension study. The mechanistic study entailed transcriptional analysis of psoriasis-related genes in lesional and nonlesional skin before and after 1 year of treatment with secukinumab 300 mg. The major finding: 85% of the upregulated genes and 75% of downregulated genes in lesional skin pretreatment reverted to normal levels in healed lesional skin post treatment.
Moreover, global mRNA expression for key genes involved in the interleukin-23/interleukin-17 inflammatory cytokine pathway closely resembled healthy skin following treatment with secukinumab. This is evidence of profound molecular normalization, Dr. Lebwohl said.
These secukinumab studies were sponsored by Novartis. Dr. Lebwohl reported having no personal financial conflicts of interest, although his department receives research funding from Novartis and numerous other pharmaceutical companies.
AT THE EADV CONGRESS
Key clinical point: , enabling some patients to experience long-term remission off therapy.
Major finding: After 1 and 2 years without any psoriasis therapy following successful treatment with secukinumab for 52 weeks, 21% and 10% of patients, respectively, remained relapse-free.
Data source: This prospective extension of two phase 3 pivotal trials of secukinumab included 220 psoriasis patients who were taken off the biologic after 52 weeks and crossed over double blind to placebo for up to 2 years.
Disclosures: The study was sponsored by Novartis. Dr. Lebwohl reported having no financial conflicts.
Pediatric psoriasis carries sharply increased risk of selected autoimmune comorbidities
GENEVA – Pediatric psoriasis is associated with sharply increased risks of selected autoimmune diseases, according to a cross-sectional study encompassing every child and adolescent in Denmark.
"Even though the absolute risk of many of these conditions remains rare in childhood, clinicians should keep these associations in mind because they can greatly add to the total disease burden. In particular, we advise focusing on extracutaneous symptoms when treating psoriasis in children,” Christoffer Blegvad, MD, said at the annual congress of the European Academy of Dermatology and Venereology.
He presented a cross-sectional study in which Denmark’s vaunted system of comprehensive national registries was harnessed to obtain health information on all individuals under age 18 years living in Denmark as of the end of 2012. The study population comprised 1,925 children and adolescents with dermatologist-diagnosed psoriasis, including those with psoriasis mild enough to be managed with topical therapies, and 1,194,712 youths without psoriasis.
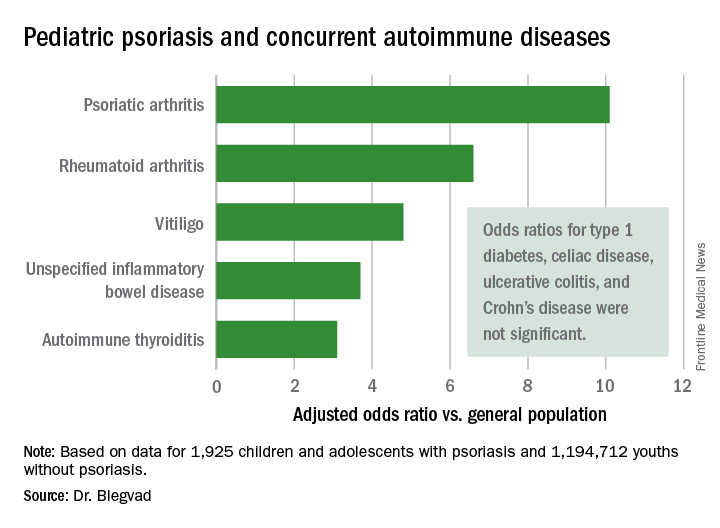
In a first-pass unadjusted analysis, the psoriasis patients were at significantly increased risk for all nine of the autoimmune diseases examined. When the investigators adjusted for age, sex, and an individual’s health care utilization as reflected in his or her number of dermatology visits, the children and adolescents with psoriasis remained at tenfold increased risk for comorbid psoriatic arthritis, 6.6-fold risk for rheumatoid arthritis, 4.8-fold risk for vitiligo, and smaller yet significantly increased risks for several other autoimmune diseases, compared with individuals without psoriasis.
Indeed, while the presence of pediatric psoriasis was associated with an adjusted 4.4-fold increased risk of having at least one autoimmune disease, psoriasis patients with one of the selected autoimmune diseases were at 7.3-fold greater risk of having two or more autoimmune diseases, compared with individuals with one autoimmune disease who didn’t have psoriasis.
This is the first study to show such a clustering effect in either pediatric or adult psoriasis patients. The finding highlights the complex genetic underpinnings of psoriasis, which has previously been shown to share genetic susceptibility loci with inflammatory bowel disease and various other autoimmune diseases, Dr. Blegvad noted.
A small caveat: The psoriatic arthritis category also included individuals with juvenile idiopathic arthritis and juvenile psoriatic arthritis, because the clinical signs and symptoms of the three disorders often are difficult to distinguish in young patients.
Dr. Blegvad reported having no financial conflicts regarding the study, funded by Herlev and Gentofte Hospital and the LEO Foundation.
GENEVA – Pediatric psoriasis is associated with sharply increased risks of selected autoimmune diseases, according to a cross-sectional study encompassing every child and adolescent in Denmark.
"Even though the absolute risk of many of these conditions remains rare in childhood, clinicians should keep these associations in mind because they can greatly add to the total disease burden. In particular, we advise focusing on extracutaneous symptoms when treating psoriasis in children,” Christoffer Blegvad, MD, said at the annual congress of the European Academy of Dermatology and Venereology.
He presented a cross-sectional study in which Denmark’s vaunted system of comprehensive national registries was harnessed to obtain health information on all individuals under age 18 years living in Denmark as of the end of 2012. The study population comprised 1,925 children and adolescents with dermatologist-diagnosed psoriasis, including those with psoriasis mild enough to be managed with topical therapies, and 1,194,712 youths without psoriasis.
In a first-pass unadjusted analysis, the psoriasis patients were at significantly increased risk for all nine of the autoimmune diseases examined. When the investigators adjusted for age, sex, and an individual’s health care utilization as reflected in his or her number of dermatology visits, the children and adolescents with psoriasis remained at tenfold increased risk for comorbid psoriatic arthritis, 6.6-fold risk for rheumatoid arthritis, 4.8-fold risk for vitiligo, and smaller yet significantly increased risks for several other autoimmune diseases, compared with individuals without psoriasis.

Indeed, while the presence of pediatric psoriasis was associated with an adjusted 4.4-fold increased risk of having at least one autoimmune disease, psoriasis patients with one of the selected autoimmune diseases were at 7.3-fold greater risk of having two or more autoimmune diseases, compared with individuals with one autoimmune disease who didn’t have psoriasis.
This is the first study to show such a clustering effect in either pediatric or adult psoriasis patients. The finding highlights the complex genetic underpinnings of psoriasis, which has previously been shown to share genetic susceptibility loci with inflammatory bowel disease and various other autoimmune diseases, Dr. Blegvad noted.
A small caveat: The psoriatic arthritis category also included individuals with juvenile idiopathic arthritis and juvenile psoriatic arthritis, because the clinical signs and symptoms of the three disorders often are difficult to distinguish in young patients.
Dr. Blegvad reported having no financial conflicts regarding the study, funded by Herlev and Gentofte Hospital and the LEO Foundation.
GENEVA – Pediatric psoriasis is associated with sharply increased risks of selected autoimmune diseases, according to a cross-sectional study encompassing every child and adolescent in Denmark.
"Even though the absolute risk of many of these conditions remains rare in childhood, clinicians should keep these associations in mind because they can greatly add to the total disease burden. In particular, we advise focusing on extracutaneous symptoms when treating psoriasis in children,” Christoffer Blegvad, MD, said at the annual congress of the European Academy of Dermatology and Venereology.
He presented a cross-sectional study in which Denmark’s vaunted system of comprehensive national registries was harnessed to obtain health information on all individuals under age 18 years living in Denmark as of the end of 2012. The study population comprised 1,925 children and adolescents with dermatologist-diagnosed psoriasis, including those with psoriasis mild enough to be managed with topical therapies, and 1,194,712 youths without psoriasis.
In a first-pass unadjusted analysis, the psoriasis patients were at significantly increased risk for all nine of the autoimmune diseases examined. When the investigators adjusted for age, sex, and an individual’s health care utilization as reflected in his or her number of dermatology visits, the children and adolescents with psoriasis remained at tenfold increased risk for comorbid psoriatic arthritis, 6.6-fold risk for rheumatoid arthritis, 4.8-fold risk for vitiligo, and smaller yet significantly increased risks for several other autoimmune diseases, compared with individuals without psoriasis.

Indeed, while the presence of pediatric psoriasis was associated with an adjusted 4.4-fold increased risk of having at least one autoimmune disease, psoriasis patients with one of the selected autoimmune diseases were at 7.3-fold greater risk of having two or more autoimmune diseases, compared with individuals with one autoimmune disease who didn’t have psoriasis.
This is the first study to show such a clustering effect in either pediatric or adult psoriasis patients. The finding highlights the complex genetic underpinnings of psoriasis, which has previously been shown to share genetic susceptibility loci with inflammatory bowel disease and various other autoimmune diseases, Dr. Blegvad noted.
A small caveat: The psoriatic arthritis category also included individuals with juvenile idiopathic arthritis and juvenile psoriatic arthritis, because the clinical signs and symptoms of the three disorders often are difficult to distinguish in young patients.
Dr. Blegvad reported having no financial conflicts regarding the study, funded by Herlev and Gentofte Hospital and the LEO Foundation.
AT THE EADV CONGRESS
Key clinical point:
Major finding: Pediatric psoriasis patients were at an adjusted 6.6-fold increased risk of comorbid rheumatoid arthritis, 4.8-fold risk of vitiligo, and significantly increased risks of several other autoimmune diseases, compared with matched youths without psoriasis.
Data source: A cross-sectional study of all children and adolescents living in Denmark at the end of 2012.
Disclosures: The presenter reported having no financial conflicts regarding the study, funded by Herlev and Gentofte Hospital and the LEO Foundation.
Biologic approved for moderate to severe psoriasis in adolescents
The Food and Drug Administration approval of ustekinumab has been expanded to include adolescents aged 12 and older with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy, based on the results of a phase 3 study.
The manufacturer, Janssen Biotech, announced the expanded indication in a press release on Oct. 13.
Ustekinumab, an interleukin-12 and -23 antagonist administered subcutaneously, was first approved by the FDA in 2009 for the same indication in adults; it is also approved for adults with active psoriatic arthritis, and for adults with moderately to severely active Crohn’s disease.
Ustekinumab is marketed as Stelara.
The Food and Drug Administration approval of ustekinumab has been expanded to include adolescents aged 12 and older with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy, based on the results of a phase 3 study.
The manufacturer, Janssen Biotech, announced the expanded indication in a press release on Oct. 13.
Ustekinumab, an interleukin-12 and -23 antagonist administered subcutaneously, was first approved by the FDA in 2009 for the same indication in adults; it is also approved for adults with active psoriatic arthritis, and for adults with moderately to severely active Crohn’s disease.
Ustekinumab is marketed as Stelara.
The Food and Drug Administration approval of ustekinumab has been expanded to include adolescents aged 12 and older with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy, based on the results of a phase 3 study.
The manufacturer, Janssen Biotech, announced the expanded indication in a press release on Oct. 13.
Ustekinumab, an interleukin-12 and -23 antagonist administered subcutaneously, was first approved by the FDA in 2009 for the same indication in adults; it is also approved for adults with active psoriatic arthritis, and for adults with moderately to severely active Crohn’s disease.
Ustekinumab is marketed as Stelara.
Genital psoriasis is the worst: Patients sound off
GENEVA – The great majority of patients with genital psoriasis say their symptoms in the genital area are worse than elsewhere on the body, Kim A. Meeuwis, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
She presented a qualitative study in which 20 patients with longstanding genital psoriasis sounded off, sharing their perspectives on the disease in one-on-one, semistructured, face-to-face interviews.
Genital psoriasis is common. Epidemiologic studies show 30%-60% of psoriasis patients experience genital involvement at some point in the course of their disease. Yet patients seldom discuss their genital psoriasis with their physicians, and the patient perspective on how the experience of genital psoriasis differs from that of having psoriasis at other locations has been addressed only sparsely in the literature. This lack of attention was the impetus for the current study, she explained.
The 20 participants in the study had an average 18-year history of plaque psoriasis, with an average 7.5-year history of genital involvement. The genital psoriasis was rated moderate or severe in 70% of subjects at the time of the study.
The most commonly reported symptoms of genital psoriasis were itch and discomfort, each of which was cited by all study participants. This was followed by erythema, cited by 95%; stinging and burning, also cited by 95%; pain, cited by 85%; scaling, by 75%; and cracking, by 30%.
Of the patients in the study, 85% reported that their pain and/or discomfort were worse in the genital area than at other sites, and 10% said they were highly self-conscious about their genital psoriasis because others had misidentified them as having a sexually transmitted infection.
Since this was a qualitative study, Dr. Meeuwis provided representative quotes from several patients, including one who asserted, “I really only have discomfort on my psoriasis on the rest of my body ... in my genitals is the only place that actually has pain, or the itching is ... really, really bad.”
Dr. Meeuwis said the study results hold an important lesson for physicians who treat psoriasis: “Due to differences in patient experiences between genital and nongenital skin, it’s really important to make time for the specific evaluation of genital involvement in taking care of patients with psoriasis – and to be sure to ask about it.”
Dr. Meeuwis reported serving as a consultant to Eli Lilly, which sponsored the study, as well as being on an advisory board to Beiersdorf.
GENEVA – The great majority of patients with genital psoriasis say their symptoms in the genital area are worse than elsewhere on the body, Kim A. Meeuwis, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
She presented a qualitative study in which 20 patients with longstanding genital psoriasis sounded off, sharing their perspectives on the disease in one-on-one, semistructured, face-to-face interviews.
Genital psoriasis is common. Epidemiologic studies show 30%-60% of psoriasis patients experience genital involvement at some point in the course of their disease. Yet patients seldom discuss their genital psoriasis with their physicians, and the patient perspective on how the experience of genital psoriasis differs from that of having psoriasis at other locations has been addressed only sparsely in the literature. This lack of attention was the impetus for the current study, she explained.
The 20 participants in the study had an average 18-year history of plaque psoriasis, with an average 7.5-year history of genital involvement. The genital psoriasis was rated moderate or severe in 70% of subjects at the time of the study.
The most commonly reported symptoms of genital psoriasis were itch and discomfort, each of which was cited by all study participants. This was followed by erythema, cited by 95%; stinging and burning, also cited by 95%; pain, cited by 85%; scaling, by 75%; and cracking, by 30%.
Of the patients in the study, 85% reported that their pain and/or discomfort were worse in the genital area than at other sites, and 10% said they were highly self-conscious about their genital psoriasis because others had misidentified them as having a sexually transmitted infection.
Since this was a qualitative study, Dr. Meeuwis provided representative quotes from several patients, including one who asserted, “I really only have discomfort on my psoriasis on the rest of my body ... in my genitals is the only place that actually has pain, or the itching is ... really, really bad.”
Dr. Meeuwis said the study results hold an important lesson for physicians who treat psoriasis: “Due to differences in patient experiences between genital and nongenital skin, it’s really important to make time for the specific evaluation of genital involvement in taking care of patients with psoriasis – and to be sure to ask about it.”
Dr. Meeuwis reported serving as a consultant to Eli Lilly, which sponsored the study, as well as being on an advisory board to Beiersdorf.
GENEVA – The great majority of patients with genital psoriasis say their symptoms in the genital area are worse than elsewhere on the body, Kim A. Meeuwis, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
She presented a qualitative study in which 20 patients with longstanding genital psoriasis sounded off, sharing their perspectives on the disease in one-on-one, semistructured, face-to-face interviews.
Genital psoriasis is common. Epidemiologic studies show 30%-60% of psoriasis patients experience genital involvement at some point in the course of their disease. Yet patients seldom discuss their genital psoriasis with their physicians, and the patient perspective on how the experience of genital psoriasis differs from that of having psoriasis at other locations has been addressed only sparsely in the literature. This lack of attention was the impetus for the current study, she explained.
The 20 participants in the study had an average 18-year history of plaque psoriasis, with an average 7.5-year history of genital involvement. The genital psoriasis was rated moderate or severe in 70% of subjects at the time of the study.
The most commonly reported symptoms of genital psoriasis were itch and discomfort, each of which was cited by all study participants. This was followed by erythema, cited by 95%; stinging and burning, also cited by 95%; pain, cited by 85%; scaling, by 75%; and cracking, by 30%.
Of the patients in the study, 85% reported that their pain and/or discomfort were worse in the genital area than at other sites, and 10% said they were highly self-conscious about their genital psoriasis because others had misidentified them as having a sexually transmitted infection.
Since this was a qualitative study, Dr. Meeuwis provided representative quotes from several patients, including one who asserted, “I really only have discomfort on my psoriasis on the rest of my body ... in my genitals is the only place that actually has pain, or the itching is ... really, really bad.”
Dr. Meeuwis said the study results hold an important lesson for physicians who treat psoriasis: “Due to differences in patient experiences between genital and nongenital skin, it’s really important to make time for the specific evaluation of genital involvement in taking care of patients with psoriasis – and to be sure to ask about it.”
Dr. Meeuwis reported serving as a consultant to Eli Lilly, which sponsored the study, as well as being on an advisory board to Beiersdorf.
AT THE EADV CONGRESS
Key clinical point:
Major finding: Of the participants in a study of genital psoriasis, 100% reported that a hallmark of their genital disease was itching and discomfort.
Data source: A qualitative study that involved one-on-one interviews with 20 patients with genital psoriasis, who shared their experiences as to how genital involvement differs from their psoriasis elsewhere.
Disclosures: The study was sponsored by Eli Lilly. The presenter reported serving as a consultant to the company.
Ixekizumab has profound impact on genital psoriasis
GENEVA – The interleukin-17A inhibitor ixekizumab provided rapid clearance of genital psoriasis in a phase 3b clinical trial, with significant improvement seen as early as week 1, Caitriona Ryan, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
The highly targeted monoclonal antibody also improved the intense itching that’s a particularly prominent feature of genital psoriasis.

“Genital psoriasis is a hidden part of psoriasis. Unfortunately, as dermatologists we do a bad job of evaluating our patients for it. They are ashamed and embarrassed to bring up the topic with their dermatologists. Hopefully, this study will create some awareness around the topic,” she said.
This was the first-ever randomized trial to evaluate the effect of a biologic agent specifically on genital psoriasis. It was also the first study of a biologic in psoriasis patients with less than 10% body surface area involved.
“That’s a very important thing,” according to the dermatologist. “There are lots of patients with genital psoriasis who have less than 10% body surface area involved and therefore don’t qualify for biologic therapy, even though their genital psoriasis can be incredibly debilitating.”
The 12-week, multicenter, double-blind trial included 149 patients with a baseline static Physician’s Global Assessment of Genitalia (sPGA-G) score of at least 3 on a 0-5 scale. All participants had failed to respond to at least one topical therapy for their genital psoriasis, such as a corticosteroid, a calcineurin inhibitor, or a vitamin D analog. The subjects averaged a 16-year history of psoriasis and a 9-year history of genital psoriasis. Thirty-eight percent of participants had an involved body surface area of at least 1% but less than 10%.
Patients were randomized to ixekizumab (Taltz) given in the usual way – a subcutaneous loading dose of 160 mg, followed by repeat 80-mg injections every 2 weeks – or placebo.
The primary study endpoint was achievement of an sPGA-G score of 0 or 1, meaning clear or almost clear, as assessed by blinded investigators. At the 12-week mark, the rate was 73% in the ixekizumab group and 8% in controls. The sPGA-G score already differed significantly between the two study arms at the first assessment, after 1 week. The treatment success rate was closely similar in patients with or without at least 10% total body surface area involved.
A key secondary endpoint concerned sexual health. Among patients who at baseline indicated that in the past week, their genital psoriasis “sometimes,” “often,” or “always” limited the frequency of their sexual activity, at week 12, 78% of those in the ixekizumab group answered the same question on the Sexual Frequency Questionnaire “never” or “rarely,” compared with 21% of controls.
“This is huge. It’s such an important part of our patients’ lives, and there was a big difference by week 1,” Dr. Ryan noted.
On another secondary endpoint, 60% of the ixekizumab group reported at least a 3-point improvement in the 0-10 Genital Itch Numeric Rating Scale at week 12, compared with 8% of controls, with a statistically significant difference apparent at week 2.
“Itch is the most frequently reported symptom in our patients with genital psoriasis, and it seems to be much more impactful than itch from psoriasis elsewhere,” Dr. Ryan commented.
The side effect profile of ixekizumab was the same as has been seen in larger, longer-term studies. There were no serious ixekizumab-related adverse events, and no cases of candidiasis.
The study was sponsored by Eli Lilly. Dr. Ryan reported serving as an advisory board member to and/or receiving honoraria from that company and more than half a dozen other pharmaceutical companies.
GENEVA – The interleukin-17A inhibitor ixekizumab provided rapid clearance of genital psoriasis in a phase 3b clinical trial, with significant improvement seen as early as week 1, Caitriona Ryan, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
The highly targeted monoclonal antibody also improved the intense itching that’s a particularly prominent feature of genital psoriasis.

“Genital psoriasis is a hidden part of psoriasis. Unfortunately, as dermatologists we do a bad job of evaluating our patients for it. They are ashamed and embarrassed to bring up the topic with their dermatologists. Hopefully, this study will create some awareness around the topic,” she said.
This was the first-ever randomized trial to evaluate the effect of a biologic agent specifically on genital psoriasis. It was also the first study of a biologic in psoriasis patients with less than 10% body surface area involved.
“That’s a very important thing,” according to the dermatologist. “There are lots of patients with genital psoriasis who have less than 10% body surface area involved and therefore don’t qualify for biologic therapy, even though their genital psoriasis can be incredibly debilitating.”
The 12-week, multicenter, double-blind trial included 149 patients with a baseline static Physician’s Global Assessment of Genitalia (sPGA-G) score of at least 3 on a 0-5 scale. All participants had failed to respond to at least one topical therapy for their genital psoriasis, such as a corticosteroid, a calcineurin inhibitor, or a vitamin D analog. The subjects averaged a 16-year history of psoriasis and a 9-year history of genital psoriasis. Thirty-eight percent of participants had an involved body surface area of at least 1% but less than 10%.
Patients were randomized to ixekizumab (Taltz) given in the usual way – a subcutaneous loading dose of 160 mg, followed by repeat 80-mg injections every 2 weeks – or placebo.
The primary study endpoint was achievement of an sPGA-G score of 0 or 1, meaning clear or almost clear, as assessed by blinded investigators. At the 12-week mark, the rate was 73% in the ixekizumab group and 8% in controls. The sPGA-G score already differed significantly between the two study arms at the first assessment, after 1 week. The treatment success rate was closely similar in patients with or without at least 10% total body surface area involved.
A key secondary endpoint concerned sexual health. Among patients who at baseline indicated that in the past week, their genital psoriasis “sometimes,” “often,” or “always” limited the frequency of their sexual activity, at week 12, 78% of those in the ixekizumab group answered the same question on the Sexual Frequency Questionnaire “never” or “rarely,” compared with 21% of controls.
“This is huge. It’s such an important part of our patients’ lives, and there was a big difference by week 1,” Dr. Ryan noted.
On another secondary endpoint, 60% of the ixekizumab group reported at least a 3-point improvement in the 0-10 Genital Itch Numeric Rating Scale at week 12, compared with 8% of controls, with a statistically significant difference apparent at week 2.
“Itch is the most frequently reported symptom in our patients with genital psoriasis, and it seems to be much more impactful than itch from psoriasis elsewhere,” Dr. Ryan commented.
The side effect profile of ixekizumab was the same as has been seen in larger, longer-term studies. There were no serious ixekizumab-related adverse events, and no cases of candidiasis.
The study was sponsored by Eli Lilly. Dr. Ryan reported serving as an advisory board member to and/or receiving honoraria from that company and more than half a dozen other pharmaceutical companies.
GENEVA – The interleukin-17A inhibitor ixekizumab provided rapid clearance of genital psoriasis in a phase 3b clinical trial, with significant improvement seen as early as week 1, Caitriona Ryan, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
The highly targeted monoclonal antibody also improved the intense itching that’s a particularly prominent feature of genital psoriasis.

“Genital psoriasis is a hidden part of psoriasis. Unfortunately, as dermatologists we do a bad job of evaluating our patients for it. They are ashamed and embarrassed to bring up the topic with their dermatologists. Hopefully, this study will create some awareness around the topic,” she said.
This was the first-ever randomized trial to evaluate the effect of a biologic agent specifically on genital psoriasis. It was also the first study of a biologic in psoriasis patients with less than 10% body surface area involved.
“That’s a very important thing,” according to the dermatologist. “There are lots of patients with genital psoriasis who have less than 10% body surface area involved and therefore don’t qualify for biologic therapy, even though their genital psoriasis can be incredibly debilitating.”
The 12-week, multicenter, double-blind trial included 149 patients with a baseline static Physician’s Global Assessment of Genitalia (sPGA-G) score of at least 3 on a 0-5 scale. All participants had failed to respond to at least one topical therapy for their genital psoriasis, such as a corticosteroid, a calcineurin inhibitor, or a vitamin D analog. The subjects averaged a 16-year history of psoriasis and a 9-year history of genital psoriasis. Thirty-eight percent of participants had an involved body surface area of at least 1% but less than 10%.
Patients were randomized to ixekizumab (Taltz) given in the usual way – a subcutaneous loading dose of 160 mg, followed by repeat 80-mg injections every 2 weeks – or placebo.
The primary study endpoint was achievement of an sPGA-G score of 0 or 1, meaning clear or almost clear, as assessed by blinded investigators. At the 12-week mark, the rate was 73% in the ixekizumab group and 8% in controls. The sPGA-G score already differed significantly between the two study arms at the first assessment, after 1 week. The treatment success rate was closely similar in patients with or without at least 10% total body surface area involved.
A key secondary endpoint concerned sexual health. Among patients who at baseline indicated that in the past week, their genital psoriasis “sometimes,” “often,” or “always” limited the frequency of their sexual activity, at week 12, 78% of those in the ixekizumab group answered the same question on the Sexual Frequency Questionnaire “never” or “rarely,” compared with 21% of controls.
“This is huge. It’s such an important part of our patients’ lives, and there was a big difference by week 1,” Dr. Ryan noted.
On another secondary endpoint, 60% of the ixekizumab group reported at least a 3-point improvement in the 0-10 Genital Itch Numeric Rating Scale at week 12, compared with 8% of controls, with a statistically significant difference apparent at week 2.
“Itch is the most frequently reported symptom in our patients with genital psoriasis, and it seems to be much more impactful than itch from psoriasis elsewhere,” Dr. Ryan commented.
The side effect profile of ixekizumab was the same as has been seen in larger, longer-term studies. There were no serious ixekizumab-related adverse events, and no cases of candidiasis.
The study was sponsored by Eli Lilly. Dr. Ryan reported serving as an advisory board member to and/or receiving honoraria from that company and more than half a dozen other pharmaceutical companies.
AT THE EADV CONGRESS
Key clinical point: First-ever trial of a biologic agent in genital psoriasis shows heartening results.
Major finding: 73% of patients with moderate to severe genital psoriasis were clear or almost clear of their genital disease after 12 weeks of ixekizumab, vs. 8% of controls.
Data source: This was a randomized, double-blind, placebo-controlled, multicenter, 12-week clinical trial in 149 patients with moderate to severe genital psoriasis.
Disclosures: The study was sponsored by Eli Lilly. The presenter reported serving as an advisory board member to and/or receiving honoraria from that company and more than half a dozen other pharmaceutical companies.
Innovations in Dermatology: Brodalumab for Plaque Psoriasis
Baseline lab data is class specific for biologics to treat psoriasis patients
said April Armstrong, MD, of the University of Southern California, Los Angeles.
Keep class-specific considerations in mind when collecting baseline lab data to help support the success of biologics in treating psoriasis, Dr. Armstrong said at the annual Coastal Dermatology Symposium.
When clinicians consider biologics, they must balance efficacy, safety, convenience, and costs of treatment, Dr. Armstrong said.
She addressed general considerations when selecting biologics for psoriasis and stressed the importance of assessing patients for tuberculosis and reviewing underlying cancer risk. Confirm that a patient has no active infections and consider whether a patient has completed all age-appropriate immunizations. Consider a complete blood count and metabolic panel for the following biologics:
- Ustekinumab: Baseline HIV or pregnancy test, and a TB evaluation at baseline as well as annual monitoring.
- Etanercept, adalimumab, infliximab: Baseline TB evaluation and screening hepatitis panel, liver function tests, and blood count, with option to add pregnancy test or HIV test. A liver function test/hepatitis panel is indicated annually, and TB should be monitored annually. Be cautious about using this class of drugs in patients with heart failure, and verify the absence of demyelinating disease in patients prior to prescribing this class of drugs.
- Guselkumab: Baseline TB evaluation, possible pregnancy or HIV tests, followed by annual TB evaluation.
- Secukinumab, ixekizumab, and brodalumab: Baseline TB evaluation, consider HIV or pregnancy tests, followed by annual TB evaluation. Be cautious about using this class of drugs in patients with ulcerative colitis or Crohn’s disease; assess and counsel for increased risk of suicidality when considering brodalumab.
Beyond the general considerations, several other factors can help maximize success with particular biologics, Dr. Armstrong said at the meeting, which is jointly presented by the University of Louisville and Global Academy for Medical Education.
The number of injections given in the first year, which range from 5 (ustekinumab) to 64 (etanercept) is an important consideration for some patients, Dr. Armstrong noted; the number of injections for the remaining biologics are guselkumab, 8; ixekizumab, 17; brodalumab and adalimumab, both 27, and secukinumab, 32. In addition, the IL-17 inhibitors carry some risk of oral candidiasis and inflammatory bowel disease.
This publication and the Global Academy for Medical Education are owned by Frontline Medical News.
Dr. Armstrong disclosed relationships with multiple companies including AbbVie, Janssen, Novartis, Lilly, Regeneron, Sanofi, Modernizing Medicine, and Valeant.
said April Armstrong, MD, of the University of Southern California, Los Angeles.
Keep class-specific considerations in mind when collecting baseline lab data to help support the success of biologics in treating psoriasis, Dr. Armstrong said at the annual Coastal Dermatology Symposium.
When clinicians consider biologics, they must balance efficacy, safety, convenience, and costs of treatment, Dr. Armstrong said.
She addressed general considerations when selecting biologics for psoriasis and stressed the importance of assessing patients for tuberculosis and reviewing underlying cancer risk. Confirm that a patient has no active infections and consider whether a patient has completed all age-appropriate immunizations. Consider a complete blood count and metabolic panel for the following biologics:
- Ustekinumab: Baseline HIV or pregnancy test, and a TB evaluation at baseline as well as annual monitoring.
- Etanercept, adalimumab, infliximab: Baseline TB evaluation and screening hepatitis panel, liver function tests, and blood count, with option to add pregnancy test or HIV test. A liver function test/hepatitis panel is indicated annually, and TB should be monitored annually. Be cautious about using this class of drugs in patients with heart failure, and verify the absence of demyelinating disease in patients prior to prescribing this class of drugs.
- Guselkumab: Baseline TB evaluation, possible pregnancy or HIV tests, followed by annual TB evaluation.
- Secukinumab, ixekizumab, and brodalumab: Baseline TB evaluation, consider HIV or pregnancy tests, followed by annual TB evaluation. Be cautious about using this class of drugs in patients with ulcerative colitis or Crohn’s disease; assess and counsel for increased risk of suicidality when considering brodalumab.
Beyond the general considerations, several other factors can help maximize success with particular biologics, Dr. Armstrong said at the meeting, which is jointly presented by the University of Louisville and Global Academy for Medical Education.
The number of injections given in the first year, which range from 5 (ustekinumab) to 64 (etanercept) is an important consideration for some patients, Dr. Armstrong noted; the number of injections for the remaining biologics are guselkumab, 8; ixekizumab, 17; brodalumab and adalimumab, both 27, and secukinumab, 32. In addition, the IL-17 inhibitors carry some risk of oral candidiasis and inflammatory bowel disease.
This publication and the Global Academy for Medical Education are owned by Frontline Medical News.
Dr. Armstrong disclosed relationships with multiple companies including AbbVie, Janssen, Novartis, Lilly, Regeneron, Sanofi, Modernizing Medicine, and Valeant.
said April Armstrong, MD, of the University of Southern California, Los Angeles.
Keep class-specific considerations in mind when collecting baseline lab data to help support the success of biologics in treating psoriasis, Dr. Armstrong said at the annual Coastal Dermatology Symposium.
When clinicians consider biologics, they must balance efficacy, safety, convenience, and costs of treatment, Dr. Armstrong said.
She addressed general considerations when selecting biologics for psoriasis and stressed the importance of assessing patients for tuberculosis and reviewing underlying cancer risk. Confirm that a patient has no active infections and consider whether a patient has completed all age-appropriate immunizations. Consider a complete blood count and metabolic panel for the following biologics:
- Ustekinumab: Baseline HIV or pregnancy test, and a TB evaluation at baseline as well as annual monitoring.
- Etanercept, adalimumab, infliximab: Baseline TB evaluation and screening hepatitis panel, liver function tests, and blood count, with option to add pregnancy test or HIV test. A liver function test/hepatitis panel is indicated annually, and TB should be monitored annually. Be cautious about using this class of drugs in patients with heart failure, and verify the absence of demyelinating disease in patients prior to prescribing this class of drugs.
- Guselkumab: Baseline TB evaluation, possible pregnancy or HIV tests, followed by annual TB evaluation.
- Secukinumab, ixekizumab, and brodalumab: Baseline TB evaluation, consider HIV or pregnancy tests, followed by annual TB evaluation. Be cautious about using this class of drugs in patients with ulcerative colitis or Crohn’s disease; assess and counsel for increased risk of suicidality when considering brodalumab.
Beyond the general considerations, several other factors can help maximize success with particular biologics, Dr. Armstrong said at the meeting, which is jointly presented by the University of Louisville and Global Academy for Medical Education.
The number of injections given in the first year, which range from 5 (ustekinumab) to 64 (etanercept) is an important consideration for some patients, Dr. Armstrong noted; the number of injections for the remaining biologics are guselkumab, 8; ixekizumab, 17; brodalumab and adalimumab, both 27, and secukinumab, 32. In addition, the IL-17 inhibitors carry some risk of oral candidiasis and inflammatory bowel disease.
This publication and the Global Academy for Medical Education are owned by Frontline Medical News.
Dr. Armstrong disclosed relationships with multiple companies including AbbVie, Janssen, Novartis, Lilly, Regeneron, Sanofi, Modernizing Medicine, and Valeant.
EXPERT ANALYSIS FROM THE COASTAL DERMATOLOGY SYMPOSIUM
New psoriasis therapies coming of age
The pathogenesis theories and treatment approaches to psoriasis have evolved over the past 3 decades, and the latest treatments continue to show safety and effectiveness, according to Alan Menter, MD, chairman of dermatology at Baylor University Medical Center, Dallas.
Before the 1980s, psoriasis was seen as a disease of keratinocyte dysfunction, with treatments that included methotrexate, UVB, and retinoids, Dr. Menter said in a presentation at the annual Coastal Dermatology Symposium. In the 1980s, it was considered an immunologic disease, and then an interleukin (IL)–12/Th1–mediated disease, with anti-CD2, anti-CD11a, and tumor necrosis factor–alpha blocker treatments from 1990 to 2004.
These include risankizumab, which targets the p19 subunit of IL-23 and is being studied for treatment of moderate to severe psoriasis. After one intravenous or subcutaneous dose of risankizumab in a phase 1 study, 16% of patients achieved a Psoriasis Area and Severity Index (PASI) 100, 58% achieved a PASI 90, and 87% achieved a PASI 75, and the publication of phase 2 results are pending, Dr. Menter said. The most common side effects included mild to moderate upper respiratory infections, mild nasopharyngitis, and mild to moderate headaches.
Psoriasis patients treated with guselkumab, which also targets the p19 subunit of IL-23 and was approved in July 2017 for patients with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy, were significantly more likely to be clear or almost clear at 16 weeks, compared with those on placebo in a phase 2 randomized, controlled trial.
“Both IL-23 and IL-17 are promising targets in the treatment of moderate to severe plaque psoriasis,” said Dr. Menter. “It is important to be vigilant in following the safety profile of these drugs both in clinical trials and in postmarketing registries to ensure their long-term safety,” he added.
Additional research on how to curb side effects associated with these new and emerging therapies should target receptors downstream along the IL-23/Th17 pathway, Dr. Menter explained. Findings from a 2015 study suggest that deficiencies in cytokines and receptors further downstream in the IL-23/Th17 pathway “are associated with fewer disorders than deficiencies in upstream components of the pathway,” he said (J Invest Dermatol. 2015 Aug;135[8]:1946-53).
Although concerns about safety remain, avoiding biologics may have a negative impact as well, as moderate to severe psoriasis patients may experience deformed joints, decreased quality of life, heart attacks, strokes, and early death, Dr. Menter said.
Dr. Menter disclosed having received research support and/or serving as a consultant and/or lecturer for AbbVie, Allergan, Amgen, Anacor, Celgene, Dermira, Eli Lilly, Galderma, Janssen Biotech, LEO Pharma, Merck, Neothetics, Novartis, Pfizer, Regeneron, Stiefel, Symbio/Maruho, Vitae, and Xenoport.
The symposium was jointly presented by the University of Louisville and Global Academy for Medical Education. This publication and Global Academy for Medical Education are both owned by Frontline Medical News.
The pathogenesis theories and treatment approaches to psoriasis have evolved over the past 3 decades, and the latest treatments continue to show safety and effectiveness, according to Alan Menter, MD, chairman of dermatology at Baylor University Medical Center, Dallas.
Before the 1980s, psoriasis was seen as a disease of keratinocyte dysfunction, with treatments that included methotrexate, UVB, and retinoids, Dr. Menter said in a presentation at the annual Coastal Dermatology Symposium. In the 1980s, it was considered an immunologic disease, and then an interleukin (IL)–12/Th1–mediated disease, with anti-CD2, anti-CD11a, and tumor necrosis factor–alpha blocker treatments from 1990 to 2004.
These include risankizumab, which targets the p19 subunit of IL-23 and is being studied for treatment of moderate to severe psoriasis. After one intravenous or subcutaneous dose of risankizumab in a phase 1 study, 16% of patients achieved a Psoriasis Area and Severity Index (PASI) 100, 58% achieved a PASI 90, and 87% achieved a PASI 75, and the publication of phase 2 results are pending, Dr. Menter said. The most common side effects included mild to moderate upper respiratory infections, mild nasopharyngitis, and mild to moderate headaches.
Psoriasis patients treated with guselkumab, which also targets the p19 subunit of IL-23 and was approved in July 2017 for patients with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy, were significantly more likely to be clear or almost clear at 16 weeks, compared with those on placebo in a phase 2 randomized, controlled trial.
“Both IL-23 and IL-17 are promising targets in the treatment of moderate to severe plaque psoriasis,” said Dr. Menter. “It is important to be vigilant in following the safety profile of these drugs both in clinical trials and in postmarketing registries to ensure their long-term safety,” he added.
Additional research on how to curb side effects associated with these new and emerging therapies should target receptors downstream along the IL-23/Th17 pathway, Dr. Menter explained. Findings from a 2015 study suggest that deficiencies in cytokines and receptors further downstream in the IL-23/Th17 pathway “are associated with fewer disorders than deficiencies in upstream components of the pathway,” he said (J Invest Dermatol. 2015 Aug;135[8]:1946-53).
Although concerns about safety remain, avoiding biologics may have a negative impact as well, as moderate to severe psoriasis patients may experience deformed joints, decreased quality of life, heart attacks, strokes, and early death, Dr. Menter said.
Dr. Menter disclosed having received research support and/or serving as a consultant and/or lecturer for AbbVie, Allergan, Amgen, Anacor, Celgene, Dermira, Eli Lilly, Galderma, Janssen Biotech, LEO Pharma, Merck, Neothetics, Novartis, Pfizer, Regeneron, Stiefel, Symbio/Maruho, Vitae, and Xenoport.
The symposium was jointly presented by the University of Louisville and Global Academy for Medical Education. This publication and Global Academy for Medical Education are both owned by Frontline Medical News.
The pathogenesis theories and treatment approaches to psoriasis have evolved over the past 3 decades, and the latest treatments continue to show safety and effectiveness, according to Alan Menter, MD, chairman of dermatology at Baylor University Medical Center, Dallas.
Before the 1980s, psoriasis was seen as a disease of keratinocyte dysfunction, with treatments that included methotrexate, UVB, and retinoids, Dr. Menter said in a presentation at the annual Coastal Dermatology Symposium. In the 1980s, it was considered an immunologic disease, and then an interleukin (IL)–12/Th1–mediated disease, with anti-CD2, anti-CD11a, and tumor necrosis factor–alpha blocker treatments from 1990 to 2004.
These include risankizumab, which targets the p19 subunit of IL-23 and is being studied for treatment of moderate to severe psoriasis. After one intravenous or subcutaneous dose of risankizumab in a phase 1 study, 16% of patients achieved a Psoriasis Area and Severity Index (PASI) 100, 58% achieved a PASI 90, and 87% achieved a PASI 75, and the publication of phase 2 results are pending, Dr. Menter said. The most common side effects included mild to moderate upper respiratory infections, mild nasopharyngitis, and mild to moderate headaches.
Psoriasis patients treated with guselkumab, which also targets the p19 subunit of IL-23 and was approved in July 2017 for patients with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy, were significantly more likely to be clear or almost clear at 16 weeks, compared with those on placebo in a phase 2 randomized, controlled trial.
“Both IL-23 and IL-17 are promising targets in the treatment of moderate to severe plaque psoriasis,” said Dr. Menter. “It is important to be vigilant in following the safety profile of these drugs both in clinical trials and in postmarketing registries to ensure their long-term safety,” he added.
Additional research on how to curb side effects associated with these new and emerging therapies should target receptors downstream along the IL-23/Th17 pathway, Dr. Menter explained. Findings from a 2015 study suggest that deficiencies in cytokines and receptors further downstream in the IL-23/Th17 pathway “are associated with fewer disorders than deficiencies in upstream components of the pathway,” he said (J Invest Dermatol. 2015 Aug;135[8]:1946-53).
Although concerns about safety remain, avoiding biologics may have a negative impact as well, as moderate to severe psoriasis patients may experience deformed joints, decreased quality of life, heart attacks, strokes, and early death, Dr. Menter said.
Dr. Menter disclosed having received research support and/or serving as a consultant and/or lecturer for AbbVie, Allergan, Amgen, Anacor, Celgene, Dermira, Eli Lilly, Galderma, Janssen Biotech, LEO Pharma, Merck, Neothetics, Novartis, Pfizer, Regeneron, Stiefel, Symbio/Maruho, Vitae, and Xenoport.
The symposium was jointly presented by the University of Louisville and Global Academy for Medical Education. This publication and Global Academy for Medical Education are both owned by Frontline Medical News.
FROM THE COASTAL DERMATOLOGY SYMPOSIUM
TNF inhibitors associated with fewer side effects than methotrexate in children with psoriasis
Treatment with tumor necrosis factor (TNF) inhibitors was associated with fewer adverse events (AEs) than with methotrexate, in an international, retrospective study of children with psoriasis.
“Patients with pediatric psoriasis treated with methotrexate had a greater risk of having one or more AEs than those treated with TNF-I [tumor necrosis factor inhibitors], although fewer AEs occurred with methotrexate or TNF-I than with other drug classes,” Inge M.G.J. Bronckers, MD, of the department of dermatology at Radboud University, Nijmegen, the Netherlands, and his coauthors reported.
Among those treated with methotrexate, administration of folic acid six to seven times a week was more protective against methotrexate-associated gastrointestinal AEs, than when administered only once a week. The study was published on Sept. 13 in JAMA Dermatology (2017. doi: 10.1001/jamadermatol.2017.3029).
The study evaluated 390 children with moderate to severe psoriasis, treated with at least one systemic medication at 20 centers in Canada, Europe, and the United States, during December 1990-September 2014. They were diagnosed at a mean age of about 8 years, and started systemic therapy a mean of 3 years later. Of the 390 children treated for psoriasis, 270 were treated with methotrexate and 106 were treated with biologics, most often the TNF inhibitor etanercept. The remaining treatments were acitretin, cyclosporine, and fumaric acid esters; almost 19% were treated with more than one medication.
Of those treated with methotrexate, 130 (48.1%) experienced one or more treatment-related AEs, compared with 41 (38.7%) of those treated with a biologic agent (odds ratio, 1.76; P = .03). Almost 25% of those on methotrexate had GI-related AEs, the most common AE; other AEs included elevated transaminase levels and fatigue. Among those on biologics, injection site reactions were the most common (in 18.9%); 12 patients (11.3%) of those on biologics had infections, primarily airway infections.
Compared with those on a TNF inhibitor, patients on methotrexate were more likely to experience GI-related AEs (OR, 11.49; P less than .001) or to discontinue treatment (OR, 5.69; P = .02), the investigators said. But associated infections were more common with TNF inhibitors (OR, 0.36; P = .03), compared with methotrexate. There were no cases of malignancies or tuberculosis.
Folic acid was prescribed to 239 patients receiving methotrexate in one of three regimens: once weekly; six times weekly, avoiding the methotrexate day; and seven times weekly, according to the investigators. Compared with once-weekly treatment, administration six or seven times weekly was associated with a lower probability of developing a GI-related AE (OR, 0.16; P less than .001; OR, 0.21; P = .003, respectively).
“Data are sparse on the relative use of systemic agents and their toxic effects in the pediatric population,” Dr. Bronckers and his coauthors wrote, adding that standardized guidelines and more data concerning children are needed. “Our data suggest that a weekly administration of folic acid could be replaced with a daily or six times weekly administration to reduce GI AEs, although the potential efficacy of six vs. seven times weekly dosing deserves further investigation,” they concluded.
The study was supported by a grant from the International Psoriasis Council. Of the 23 authors, 11 had financial disclosures with various pharmaceutical manufacturers.
Treatment with tumor necrosis factor (TNF) inhibitors was associated with fewer adverse events (AEs) than with methotrexate, in an international, retrospective study of children with psoriasis.
“Patients with pediatric psoriasis treated with methotrexate had a greater risk of having one or more AEs than those treated with TNF-I [tumor necrosis factor inhibitors], although fewer AEs occurred with methotrexate or TNF-I than with other drug classes,” Inge M.G.J. Bronckers, MD, of the department of dermatology at Radboud University, Nijmegen, the Netherlands, and his coauthors reported.
Among those treated with methotrexate, administration of folic acid six to seven times a week was more protective against methotrexate-associated gastrointestinal AEs, than when administered only once a week. The study was published on Sept. 13 in JAMA Dermatology (2017. doi: 10.1001/jamadermatol.2017.3029).
The study evaluated 390 children with moderate to severe psoriasis, treated with at least one systemic medication at 20 centers in Canada, Europe, and the United States, during December 1990-September 2014. They were diagnosed at a mean age of about 8 years, and started systemic therapy a mean of 3 years later. Of the 390 children treated for psoriasis, 270 were treated with methotrexate and 106 were treated with biologics, most often the TNF inhibitor etanercept. The remaining treatments were acitretin, cyclosporine, and fumaric acid esters; almost 19% were treated with more than one medication.
Of those treated with methotrexate, 130 (48.1%) experienced one or more treatment-related AEs, compared with 41 (38.7%) of those treated with a biologic agent (odds ratio, 1.76; P = .03). Almost 25% of those on methotrexate had GI-related AEs, the most common AE; other AEs included elevated transaminase levels and fatigue. Among those on biologics, injection site reactions were the most common (in 18.9%); 12 patients (11.3%) of those on biologics had infections, primarily airway infections.
Compared with those on a TNF inhibitor, patients on methotrexate were more likely to experience GI-related AEs (OR, 11.49; P less than .001) or to discontinue treatment (OR, 5.69; P = .02), the investigators said. But associated infections were more common with TNF inhibitors (OR, 0.36; P = .03), compared with methotrexate. There were no cases of malignancies or tuberculosis.
Folic acid was prescribed to 239 patients receiving methotrexate in one of three regimens: once weekly; six times weekly, avoiding the methotrexate day; and seven times weekly, according to the investigators. Compared with once-weekly treatment, administration six or seven times weekly was associated with a lower probability of developing a GI-related AE (OR, 0.16; P less than .001; OR, 0.21; P = .003, respectively).
“Data are sparse on the relative use of systemic agents and their toxic effects in the pediatric population,” Dr. Bronckers and his coauthors wrote, adding that standardized guidelines and more data concerning children are needed. “Our data suggest that a weekly administration of folic acid could be replaced with a daily or six times weekly administration to reduce GI AEs, although the potential efficacy of six vs. seven times weekly dosing deserves further investigation,” they concluded.
The study was supported by a grant from the International Psoriasis Council. Of the 23 authors, 11 had financial disclosures with various pharmaceutical manufacturers.
Treatment with tumor necrosis factor (TNF) inhibitors was associated with fewer adverse events (AEs) than with methotrexate, in an international, retrospective study of children with psoriasis.
“Patients with pediatric psoriasis treated with methotrexate had a greater risk of having one or more AEs than those treated with TNF-I [tumor necrosis factor inhibitors], although fewer AEs occurred with methotrexate or TNF-I than with other drug classes,” Inge M.G.J. Bronckers, MD, of the department of dermatology at Radboud University, Nijmegen, the Netherlands, and his coauthors reported.
Among those treated with methotrexate, administration of folic acid six to seven times a week was more protective against methotrexate-associated gastrointestinal AEs, than when administered only once a week. The study was published on Sept. 13 in JAMA Dermatology (2017. doi: 10.1001/jamadermatol.2017.3029).
The study evaluated 390 children with moderate to severe psoriasis, treated with at least one systemic medication at 20 centers in Canada, Europe, and the United States, during December 1990-September 2014. They were diagnosed at a mean age of about 8 years, and started systemic therapy a mean of 3 years later. Of the 390 children treated for psoriasis, 270 were treated with methotrexate and 106 were treated with biologics, most often the TNF inhibitor etanercept. The remaining treatments were acitretin, cyclosporine, and fumaric acid esters; almost 19% were treated with more than one medication.
Of those treated with methotrexate, 130 (48.1%) experienced one or more treatment-related AEs, compared with 41 (38.7%) of those treated with a biologic agent (odds ratio, 1.76; P = .03). Almost 25% of those on methotrexate had GI-related AEs, the most common AE; other AEs included elevated transaminase levels and fatigue. Among those on biologics, injection site reactions were the most common (in 18.9%); 12 patients (11.3%) of those on biologics had infections, primarily airway infections.
Compared with those on a TNF inhibitor, patients on methotrexate were more likely to experience GI-related AEs (OR, 11.49; P less than .001) or to discontinue treatment (OR, 5.69; P = .02), the investigators said. But associated infections were more common with TNF inhibitors (OR, 0.36; P = .03), compared with methotrexate. There were no cases of malignancies or tuberculosis.
Folic acid was prescribed to 239 patients receiving methotrexate in one of three regimens: once weekly; six times weekly, avoiding the methotrexate day; and seven times weekly, according to the investigators. Compared with once-weekly treatment, administration six or seven times weekly was associated with a lower probability of developing a GI-related AE (OR, 0.16; P less than .001; OR, 0.21; P = .003, respectively).
“Data are sparse on the relative use of systemic agents and their toxic effects in the pediatric population,” Dr. Bronckers and his coauthors wrote, adding that standardized guidelines and more data concerning children are needed. “Our data suggest that a weekly administration of folic acid could be replaced with a daily or six times weekly administration to reduce GI AEs, although the potential efficacy of six vs. seven times weekly dosing deserves further investigation,” they concluded.
The study was supported by a grant from the International Psoriasis Council. Of the 23 authors, 11 had financial disclosures with various pharmaceutical manufacturers.
FROM JAMA DERMATOLOGY
Key clinical point:
Major finding: Almost half (48.1%) of the children treated with methotrexate experienced one or more treatment-related adverse events, compared with 41 (38.7%) of those treated with a biologic agent (OR, 1.76; P = .03).
Data source: An international, retrospective study of 390 children with moderate to severe psoriasis compared the adverse events associated with different systemic therapies.
Disclosures: The study was supported by a grant from the International Psoriasis Council.