User login
Antirheumatic drugs don’t boost surgical infection risk
SAN DIEGO – Rheumatoid arthritis patients undergoing surgery who stayed on their antirheumatic medication perioperatively didn’t have a higher risk of early postoperative infection compared with those who temporarily stopped treatment before surgery, according to findings from a large national Veterans Affairs study.
Rheumatologists are frequently consulted about this issue. Evidence to guide practice has been scarce, however, and until now many rheumatologists and surgeons have taken a conservative approach, reasoning that the immunosuppressive drugs employed in controlling inflammation in rheumatoid arthritis might also increase the risk of surgical wound infection.
A common practice has been to have RA patients stop their medication a month ahead of elective surgery, or at least two drug half-lives beforehand, then start treatment again roughly a month after the operation, or when the wound has healed. The new Veterans Affairs (VA) study findings suggest this practice may be unnecessary, Dr. Zaki Abou Zahr said at the annual meeting of the American College of Rheumatology.
Dr. Bernard Ng, his senior coinvestigator in the study, added that temporarily stopping antirheumatic agents before surgery may actually be harmful in that it increases the risk of a flare of the RA, which in turn would impede postoperative rehabilitation.
But there is a major caveat regarding the VA study: Participation was restricted to RA patients on only a single conventional disease-modifying antirheumatic drug (DMARD) or biologic agent leading up to surgery. This restriction, imposed to make for a more clear-cut analysis, means that the study results can’t be extrapolated to patients on multidrug therapy. And multidrug therapy is quite common. Indeed, slightly more than half of RA patients in the VA health care system are on combination therapy, most often methotrexate plus a biologic agent, noted Dr. Ng, chief of rheumatology at the VA Puget Sound Health Care System, Seattle.
Dr. Abou Zahr presented the retrospective cohort study involving 6,548 RA patients in VA administrative databases, all of whom were on antirheumatic drug monotherapy prior to surgery. The surgery was of all types, including cardiothoracic, gastrointestinal, vascular, and orthopedic, as well as emergent and elective.
The primary endpoints were the rate of wound infections, both superficial and deep, within 30 days post surgery, and the general infection rate – including pneumonia, sepsis, and urinary tract infections – during the same time frame.
Sixty-two percent of the 1,480 RA patients on a single biologic agent did not stop taking it preoperatively. One key study finding was that neither their postoperative wound infection rate nor their general infection rate differed significantly from rates in patients who temporarily halted their biologic agent. The same held true among the 70% of patients on a single conventional DMARD who did not stop taking their medication preoperatively, according to Dr. Abou Zahr of Baylor College of Medicine, Houston.
Dr. Ng said the investigators plan to extend their work to include RA patients on multiple antirheumatic drugs that they do or don’t temporarily stop when undergoing surgery within the VA system. The researchers also plan to take a close look at patients undergoing specific types of surgery to see if the postoperative infection risk in patients who remain on treatment varies according to their operation.
Dr. Fehmida Zahabi, a rheumatologist from Plano, Tex., who chaired a press conference highlighting the VA study findings, said that while she’d like to see a confirmatory study, "I think we’re getting to the point where we’re saying we should cautiously keep these patients on their medications. That’s what the data suggest."
She noted that before the VA study, the very limited evidence available to guide practice in this area centered on a 12-year-old British randomized trial involving RA patients on methotrexate undergoing elective orthopedic surgery. Those assigned to stop the drug from 2 weeks before surgery to 2 weeks post surgery had significantly more infections, surgical complications, and RA flares within 6 weeks after surgery (Ann. Rheum. Dis. 2001;60:214-7).
As for patients on multidrug therapy who are scheduled for surgery, her inclination until evidence becomes available for guidance is to pare down the regimen preoperatively, while keeping the patient on one or two drugs.
The VA study was funded by the Department of Veterans Affairs. Dr. Abou Zahr and Dr. Ng reported having no conflicts of interest.
SAN DIEGO – Rheumatoid arthritis patients undergoing surgery who stayed on their antirheumatic medication perioperatively didn’t have a higher risk of early postoperative infection compared with those who temporarily stopped treatment before surgery, according to findings from a large national Veterans Affairs study.
Rheumatologists are frequently consulted about this issue. Evidence to guide practice has been scarce, however, and until now many rheumatologists and surgeons have taken a conservative approach, reasoning that the immunosuppressive drugs employed in controlling inflammation in rheumatoid arthritis might also increase the risk of surgical wound infection.
A common practice has been to have RA patients stop their medication a month ahead of elective surgery, or at least two drug half-lives beforehand, then start treatment again roughly a month after the operation, or when the wound has healed. The new Veterans Affairs (VA) study findings suggest this practice may be unnecessary, Dr. Zaki Abou Zahr said at the annual meeting of the American College of Rheumatology.
Dr. Bernard Ng, his senior coinvestigator in the study, added that temporarily stopping antirheumatic agents before surgery may actually be harmful in that it increases the risk of a flare of the RA, which in turn would impede postoperative rehabilitation.
But there is a major caveat regarding the VA study: Participation was restricted to RA patients on only a single conventional disease-modifying antirheumatic drug (DMARD) or biologic agent leading up to surgery. This restriction, imposed to make for a more clear-cut analysis, means that the study results can’t be extrapolated to patients on multidrug therapy. And multidrug therapy is quite common. Indeed, slightly more than half of RA patients in the VA health care system are on combination therapy, most often methotrexate plus a biologic agent, noted Dr. Ng, chief of rheumatology at the VA Puget Sound Health Care System, Seattle.
Dr. Abou Zahr presented the retrospective cohort study involving 6,548 RA patients in VA administrative databases, all of whom were on antirheumatic drug monotherapy prior to surgery. The surgery was of all types, including cardiothoracic, gastrointestinal, vascular, and orthopedic, as well as emergent and elective.
The primary endpoints were the rate of wound infections, both superficial and deep, within 30 days post surgery, and the general infection rate – including pneumonia, sepsis, and urinary tract infections – during the same time frame.
Sixty-two percent of the 1,480 RA patients on a single biologic agent did not stop taking it preoperatively. One key study finding was that neither their postoperative wound infection rate nor their general infection rate differed significantly from rates in patients who temporarily halted their biologic agent. The same held true among the 70% of patients on a single conventional DMARD who did not stop taking their medication preoperatively, according to Dr. Abou Zahr of Baylor College of Medicine, Houston.
Dr. Ng said the investigators plan to extend their work to include RA patients on multiple antirheumatic drugs that they do or don’t temporarily stop when undergoing surgery within the VA system. The researchers also plan to take a close look at patients undergoing specific types of surgery to see if the postoperative infection risk in patients who remain on treatment varies according to their operation.
Dr. Fehmida Zahabi, a rheumatologist from Plano, Tex., who chaired a press conference highlighting the VA study findings, said that while she’d like to see a confirmatory study, "I think we’re getting to the point where we’re saying we should cautiously keep these patients on their medications. That’s what the data suggest."
She noted that before the VA study, the very limited evidence available to guide practice in this area centered on a 12-year-old British randomized trial involving RA patients on methotrexate undergoing elective orthopedic surgery. Those assigned to stop the drug from 2 weeks before surgery to 2 weeks post surgery had significantly more infections, surgical complications, and RA flares within 6 weeks after surgery (Ann. Rheum. Dis. 2001;60:214-7).
As for patients on multidrug therapy who are scheduled for surgery, her inclination until evidence becomes available for guidance is to pare down the regimen preoperatively, while keeping the patient on one or two drugs.
The VA study was funded by the Department of Veterans Affairs. Dr. Abou Zahr and Dr. Ng reported having no conflicts of interest.
SAN DIEGO – Rheumatoid arthritis patients undergoing surgery who stayed on their antirheumatic medication perioperatively didn’t have a higher risk of early postoperative infection compared with those who temporarily stopped treatment before surgery, according to findings from a large national Veterans Affairs study.
Rheumatologists are frequently consulted about this issue. Evidence to guide practice has been scarce, however, and until now many rheumatologists and surgeons have taken a conservative approach, reasoning that the immunosuppressive drugs employed in controlling inflammation in rheumatoid arthritis might also increase the risk of surgical wound infection.
A common practice has been to have RA patients stop their medication a month ahead of elective surgery, or at least two drug half-lives beforehand, then start treatment again roughly a month after the operation, or when the wound has healed. The new Veterans Affairs (VA) study findings suggest this practice may be unnecessary, Dr. Zaki Abou Zahr said at the annual meeting of the American College of Rheumatology.
Dr. Bernard Ng, his senior coinvestigator in the study, added that temporarily stopping antirheumatic agents before surgery may actually be harmful in that it increases the risk of a flare of the RA, which in turn would impede postoperative rehabilitation.
But there is a major caveat regarding the VA study: Participation was restricted to RA patients on only a single conventional disease-modifying antirheumatic drug (DMARD) or biologic agent leading up to surgery. This restriction, imposed to make for a more clear-cut analysis, means that the study results can’t be extrapolated to patients on multidrug therapy. And multidrug therapy is quite common. Indeed, slightly more than half of RA patients in the VA health care system are on combination therapy, most often methotrexate plus a biologic agent, noted Dr. Ng, chief of rheumatology at the VA Puget Sound Health Care System, Seattle.
Dr. Abou Zahr presented the retrospective cohort study involving 6,548 RA patients in VA administrative databases, all of whom were on antirheumatic drug monotherapy prior to surgery. The surgery was of all types, including cardiothoracic, gastrointestinal, vascular, and orthopedic, as well as emergent and elective.
The primary endpoints were the rate of wound infections, both superficial and deep, within 30 days post surgery, and the general infection rate – including pneumonia, sepsis, and urinary tract infections – during the same time frame.
Sixty-two percent of the 1,480 RA patients on a single biologic agent did not stop taking it preoperatively. One key study finding was that neither their postoperative wound infection rate nor their general infection rate differed significantly from rates in patients who temporarily halted their biologic agent. The same held true among the 70% of patients on a single conventional DMARD who did not stop taking their medication preoperatively, according to Dr. Abou Zahr of Baylor College of Medicine, Houston.
Dr. Ng said the investigators plan to extend their work to include RA patients on multiple antirheumatic drugs that they do or don’t temporarily stop when undergoing surgery within the VA system. The researchers also plan to take a close look at patients undergoing specific types of surgery to see if the postoperative infection risk in patients who remain on treatment varies according to their operation.
Dr. Fehmida Zahabi, a rheumatologist from Plano, Tex., who chaired a press conference highlighting the VA study findings, said that while she’d like to see a confirmatory study, "I think we’re getting to the point where we’re saying we should cautiously keep these patients on their medications. That’s what the data suggest."
She noted that before the VA study, the very limited evidence available to guide practice in this area centered on a 12-year-old British randomized trial involving RA patients on methotrexate undergoing elective orthopedic surgery. Those assigned to stop the drug from 2 weeks before surgery to 2 weeks post surgery had significantly more infections, surgical complications, and RA flares within 6 weeks after surgery (Ann. Rheum. Dis. 2001;60:214-7).
As for patients on multidrug therapy who are scheduled for surgery, her inclination until evidence becomes available for guidance is to pare down the regimen preoperatively, while keeping the patient on one or two drugs.
The VA study was funded by the Department of Veterans Affairs. Dr. Abou Zahr and Dr. Ng reported having no conflicts of interest.
AT THE ACR ANNUAL MEETING
Major finding: Rheumatoid arthritis patients who remained on their antirheumatic medication while they underwent various types of surgery did not have a significantly different 30-day wound infection rate than those who stopped treatment temporarily prior to surgery.
Data source: This was a retrospective observational cohort study involving 6,548 rheumatoid arthritis patients undergoing various types of surgery.
Disclosures: The study was funded by the Department of Veterans Affairs. The presenters reported having no financial conflicts.
Systemic sclerosis brings sharply increased MI risk
SAN DIEGO – Patients with systemic sclerosis are at greater than eightfold increased risk of having an acute myocardial infarction during the first year after diagnosis, compared with matched controls, according to the first large population-based cohort study to look at the issue.
After that first year, the MI risk drops off over time. Still, during years 1-5 post diagnosis the MI risk remains more than triple that of the matched general population, Dr. J. Antonio Avina-Zubieta reported at the annual meeting of the American College of Rheumatology.
"Our findings support increased vigilance in cardiovascular disease prevention, surveillance, and risk modification in patients with systemic sclerosis," declared Dr. Avina-Zubieta, a rheumatologist at the University of British Columbia, Vancouver.
He and his coinvestigators harnessed comprehensive provincial medical databases to identify all 1,245 adults diagnosed with rheumatologist-confirmed systemic sclerosis (SSc) in British Columbia during 1996-2010. The SSc patients averaged 53 years of age at the time of their rheumatologic diagnosis. Eighty-three percent were women.
During a mean follow-up period of 3.5 years following diagnosis of SSc, 89 patients experienced an acute MI, as did 289 controls. This translated to an incidence rate of 20.2 cases per 1,000 person-years among patients with SSc, compared with 5.3 per 1,000 among controls.
The risk was 8.2-fold greater in SSc patients than controls during the first year after diagnosis and 3.5 times greater during years 1-5 after diagnosis, but once patients got more than 5 years out from diagnosis there was no longer any significant increase in MI risk.
Patients with SSc who were in the 45- to 59-year-old age group were at greatest risk: They were 7.4 times more likely than controls to have an MI. SSc patients aged 60-74 years had a 4.4-fold greater risk of MI than did controls, and those aged 75 years and older were at 5.6-fold increased risk.
The SSc patients remained at a 4.3-fold increased risk of MI, compared with controls, in a multivariate analysis extensively adjusted for baseline chronic obstructive pulmonary disease, angina, obesity, hormone replacement therapy, Charlson Comorbidity Index, number of hospitalizations during the 12 months prior to diagnosis, and the use of NSAIDs, COX-2 inhibitors, glucocorticoids, lipid-lowering drugs, and antidiabetic medications.
It is well established that patients with rheumatoid arthritis or systemic lupus erythematosus have an increased risk of premature atherosclerotic disease. Up until now, however, information about the risk of major adverse cardiovascular events in patients with SSc has been scarce and has come from nondefinitive studies in selected populations or cross-sectional studies lacking adequate adjustment for potential confounding factors, according to Dr. Avina-Zubieta, who also is a research scientist at the Arthritis Research Centre of Canada in Richmond, B.C.
Why the sharp increase in MI risk during the first year after diagnosis of SSc? He offered two hypotheses. One is that this is a time of particularly great inflammatory load as physicians work to get the disease under control. Also, there is a phenomenon Dr. Avina-Zubieta called "the depletion of susceptibles," whereby, once the patients at higher risk have had their MI early on, those who remain aren’t at sufficient risk to stand out from the general population.
The study was sponsored by the Arthritis Centre of Canada. Dr. Avina-Zubieta reported having no financial conflicts of interest.
SAN DIEGO – Patients with systemic sclerosis are at greater than eightfold increased risk of having an acute myocardial infarction during the first year after diagnosis, compared with matched controls, according to the first large population-based cohort study to look at the issue.
After that first year, the MI risk drops off over time. Still, during years 1-5 post diagnosis the MI risk remains more than triple that of the matched general population, Dr. J. Antonio Avina-Zubieta reported at the annual meeting of the American College of Rheumatology.
"Our findings support increased vigilance in cardiovascular disease prevention, surveillance, and risk modification in patients with systemic sclerosis," declared Dr. Avina-Zubieta, a rheumatologist at the University of British Columbia, Vancouver.
He and his coinvestigators harnessed comprehensive provincial medical databases to identify all 1,245 adults diagnosed with rheumatologist-confirmed systemic sclerosis (SSc) in British Columbia during 1996-2010. The SSc patients averaged 53 years of age at the time of their rheumatologic diagnosis. Eighty-three percent were women.
During a mean follow-up period of 3.5 years following diagnosis of SSc, 89 patients experienced an acute MI, as did 289 controls. This translated to an incidence rate of 20.2 cases per 1,000 person-years among patients with SSc, compared with 5.3 per 1,000 among controls.
The risk was 8.2-fold greater in SSc patients than controls during the first year after diagnosis and 3.5 times greater during years 1-5 after diagnosis, but once patients got more than 5 years out from diagnosis there was no longer any significant increase in MI risk.
Patients with SSc who were in the 45- to 59-year-old age group were at greatest risk: They were 7.4 times more likely than controls to have an MI. SSc patients aged 60-74 years had a 4.4-fold greater risk of MI than did controls, and those aged 75 years and older were at 5.6-fold increased risk.
The SSc patients remained at a 4.3-fold increased risk of MI, compared with controls, in a multivariate analysis extensively adjusted for baseline chronic obstructive pulmonary disease, angina, obesity, hormone replacement therapy, Charlson Comorbidity Index, number of hospitalizations during the 12 months prior to diagnosis, and the use of NSAIDs, COX-2 inhibitors, glucocorticoids, lipid-lowering drugs, and antidiabetic medications.
It is well established that patients with rheumatoid arthritis or systemic lupus erythematosus have an increased risk of premature atherosclerotic disease. Up until now, however, information about the risk of major adverse cardiovascular events in patients with SSc has been scarce and has come from nondefinitive studies in selected populations or cross-sectional studies lacking adequate adjustment for potential confounding factors, according to Dr. Avina-Zubieta, who also is a research scientist at the Arthritis Research Centre of Canada in Richmond, B.C.
Why the sharp increase in MI risk during the first year after diagnosis of SSc? He offered two hypotheses. One is that this is a time of particularly great inflammatory load as physicians work to get the disease under control. Also, there is a phenomenon Dr. Avina-Zubieta called "the depletion of susceptibles," whereby, once the patients at higher risk have had their MI early on, those who remain aren’t at sufficient risk to stand out from the general population.
The study was sponsored by the Arthritis Centre of Canada. Dr. Avina-Zubieta reported having no financial conflicts of interest.
SAN DIEGO – Patients with systemic sclerosis are at greater than eightfold increased risk of having an acute myocardial infarction during the first year after diagnosis, compared with matched controls, according to the first large population-based cohort study to look at the issue.
After that first year, the MI risk drops off over time. Still, during years 1-5 post diagnosis the MI risk remains more than triple that of the matched general population, Dr. J. Antonio Avina-Zubieta reported at the annual meeting of the American College of Rheumatology.
"Our findings support increased vigilance in cardiovascular disease prevention, surveillance, and risk modification in patients with systemic sclerosis," declared Dr. Avina-Zubieta, a rheumatologist at the University of British Columbia, Vancouver.
He and his coinvestigators harnessed comprehensive provincial medical databases to identify all 1,245 adults diagnosed with rheumatologist-confirmed systemic sclerosis (SSc) in British Columbia during 1996-2010. The SSc patients averaged 53 years of age at the time of their rheumatologic diagnosis. Eighty-three percent were women.
During a mean follow-up period of 3.5 years following diagnosis of SSc, 89 patients experienced an acute MI, as did 289 controls. This translated to an incidence rate of 20.2 cases per 1,000 person-years among patients with SSc, compared with 5.3 per 1,000 among controls.
The risk was 8.2-fold greater in SSc patients than controls during the first year after diagnosis and 3.5 times greater during years 1-5 after diagnosis, but once patients got more than 5 years out from diagnosis there was no longer any significant increase in MI risk.
Patients with SSc who were in the 45- to 59-year-old age group were at greatest risk: They were 7.4 times more likely than controls to have an MI. SSc patients aged 60-74 years had a 4.4-fold greater risk of MI than did controls, and those aged 75 years and older were at 5.6-fold increased risk.
The SSc patients remained at a 4.3-fold increased risk of MI, compared with controls, in a multivariate analysis extensively adjusted for baseline chronic obstructive pulmonary disease, angina, obesity, hormone replacement therapy, Charlson Comorbidity Index, number of hospitalizations during the 12 months prior to diagnosis, and the use of NSAIDs, COX-2 inhibitors, glucocorticoids, lipid-lowering drugs, and antidiabetic medications.
It is well established that patients with rheumatoid arthritis or systemic lupus erythematosus have an increased risk of premature atherosclerotic disease. Up until now, however, information about the risk of major adverse cardiovascular events in patients with SSc has been scarce and has come from nondefinitive studies in selected populations or cross-sectional studies lacking adequate adjustment for potential confounding factors, according to Dr. Avina-Zubieta, who also is a research scientist at the Arthritis Research Centre of Canada in Richmond, B.C.
Why the sharp increase in MI risk during the first year after diagnosis of SSc? He offered two hypotheses. One is that this is a time of particularly great inflammatory load as physicians work to get the disease under control. Also, there is a phenomenon Dr. Avina-Zubieta called "the depletion of susceptibles," whereby, once the patients at higher risk have had their MI early on, those who remain aren’t at sufficient risk to stand out from the general population.
The study was sponsored by the Arthritis Centre of Canada. Dr. Avina-Zubieta reported having no financial conflicts of interest.
AT THE ACR ANNUAL MEETING
Major finding: The incidence rate of acute MI following diagnosis of systemic sclerosis was 20.2 events per 1,000 person-years, compared with 5.3 per 1,000 in matched controls.
Data source: Population-based cohort study included all 1,245 adults diagnosed with systemic sclerosis in British Columbia in 1990-2010 and 12,678 matched controls drawn from the general population.
Disclosures: The study was sponsored by the Arthritis Centre of Canada. Dr. Avina-Zubieta reported having no financial conflicts of interest.
Treatment adherence poor among Medicaid beneficiaries with lupus
SAN DIEGO – Fewer than one in three Medicaid beneficiaries with systemic lupus erythematosus are adhering to their recommended treatment regimens at least 80% of the time, results from a large national analysis demonstrated.
"As a physician who spends my professional time taking care of people with lupus, these data are highly concerning," Dr. Jinoos Yazdany said in an interview prior to the annual meeting of the American College of Rheumatology, where the study was presented. "For many individuals with lupus, treatment is very effective and has a proven track record of preventing life-threatening complications and long-term organ damage such as kidney failure. The fact that less than one in three patients are adhering with treatment is alarming and should serve as a call to action for those of providing care to these patients."
Dr. Yazdany, associate director of the lupus clinic at the University of California-San Francisco Medical Center, and her colleagues used MAX (Medicaid Analytic eXtract) data from 2000 to 2006 to identify 23,187 patients with systemic lupus erythematosus (SLE) who were taking at least one immunosuppressive or antimalarial drug. They used pharmacy claims to assess adherence to drugs over a period of 180 days by calculating a medication possession ratio (MPR), defined as the proportion of days covered by the total days’ supply dispensed after the first claim for each drug. The researchers also evaluated the proportion of patients who had an MPR of 80% or greater. The oral drugs studied were hydroxychloroquine, azathioprine, methotrexate, mycophenolate mofetil, cyclosporine, tacrolimus, cyclophosphamide, and leflunomide.
The mean age of the 23,187 patients was 38 years, 94% were female, and the racial and ethnic makeup was diverse (40% black, 34% white, 16% Hispanic, 5% Asian, and 5% other). The highest proportion of SLE patients lived in the southern United States (36%).
Dr. Yazdany reported that the average MPR ranged from 31.1% for tacrolimus to 56% for hydroxychloroquine. In addition, for most drugs, fewer than one in three patients had an MPR of at least 80%. Overall adherence was poorest among those taking tacrolimus (14%) and highest among those taking mycophenolate mofetil (40%).
Across all medications, blacks had lower adherence, compared with whites, and adherence was highest for those residing in the Northeast.
"Our findings underscore the need to understand the reasons behind low adherence in this high-risk and vulnerable group of patients," Dr. Yazdany said. "And we urgently need to develop interventions to improve adherence. Physicians may be unaware of their patient’s adherence with medication, and patients may not be forthcoming about this issue. Concerns about side effects, inadequate understanding of the benefit, and the medication’s cost may be barriers for patients. Treatment-associated side effects may be another important barrier. We need better patient-physician communication around the issue of adherence."
The Medicaid administrative data allowed for an otherwise unobtainable nationwide view of adherence in SLE, but Dr. Yazdany acknowledged certain limitations of the study, including the fact that treatment "may be interrupted for clinically appropriate reasons, so the medication possession ratios in our study may underestimate actual adherence. Also, pharmacy claims are imperfect proxies for whether patients actually take medications that are dispensed, which might lead to an overestimation of adherence."
Dr. Yazdany disclosed that her research is funded by the National Institutes of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases. She had no other relevant financial conflicts to disclose.
SAN DIEGO – Fewer than one in three Medicaid beneficiaries with systemic lupus erythematosus are adhering to their recommended treatment regimens at least 80% of the time, results from a large national analysis demonstrated.
"As a physician who spends my professional time taking care of people with lupus, these data are highly concerning," Dr. Jinoos Yazdany said in an interview prior to the annual meeting of the American College of Rheumatology, where the study was presented. "For many individuals with lupus, treatment is very effective and has a proven track record of preventing life-threatening complications and long-term organ damage such as kidney failure. The fact that less than one in three patients are adhering with treatment is alarming and should serve as a call to action for those of providing care to these patients."
Dr. Yazdany, associate director of the lupus clinic at the University of California-San Francisco Medical Center, and her colleagues used MAX (Medicaid Analytic eXtract) data from 2000 to 2006 to identify 23,187 patients with systemic lupus erythematosus (SLE) who were taking at least one immunosuppressive or antimalarial drug. They used pharmacy claims to assess adherence to drugs over a period of 180 days by calculating a medication possession ratio (MPR), defined as the proportion of days covered by the total days’ supply dispensed after the first claim for each drug. The researchers also evaluated the proportion of patients who had an MPR of 80% or greater. The oral drugs studied were hydroxychloroquine, azathioprine, methotrexate, mycophenolate mofetil, cyclosporine, tacrolimus, cyclophosphamide, and leflunomide.
The mean age of the 23,187 patients was 38 years, 94% were female, and the racial and ethnic makeup was diverse (40% black, 34% white, 16% Hispanic, 5% Asian, and 5% other). The highest proportion of SLE patients lived in the southern United States (36%).
Dr. Yazdany reported that the average MPR ranged from 31.1% for tacrolimus to 56% for hydroxychloroquine. In addition, for most drugs, fewer than one in three patients had an MPR of at least 80%. Overall adherence was poorest among those taking tacrolimus (14%) and highest among those taking mycophenolate mofetil (40%).
Across all medications, blacks had lower adherence, compared with whites, and adherence was highest for those residing in the Northeast.
"Our findings underscore the need to understand the reasons behind low adherence in this high-risk and vulnerable group of patients," Dr. Yazdany said. "And we urgently need to develop interventions to improve adherence. Physicians may be unaware of their patient’s adherence with medication, and patients may not be forthcoming about this issue. Concerns about side effects, inadequate understanding of the benefit, and the medication’s cost may be barriers for patients. Treatment-associated side effects may be another important barrier. We need better patient-physician communication around the issue of adherence."
The Medicaid administrative data allowed for an otherwise unobtainable nationwide view of adherence in SLE, but Dr. Yazdany acknowledged certain limitations of the study, including the fact that treatment "may be interrupted for clinically appropriate reasons, so the medication possession ratios in our study may underestimate actual adherence. Also, pharmacy claims are imperfect proxies for whether patients actually take medications that are dispensed, which might lead to an overestimation of adherence."
Dr. Yazdany disclosed that her research is funded by the National Institutes of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases. She had no other relevant financial conflicts to disclose.
SAN DIEGO – Fewer than one in three Medicaid beneficiaries with systemic lupus erythematosus are adhering to their recommended treatment regimens at least 80% of the time, results from a large national analysis demonstrated.
"As a physician who spends my professional time taking care of people with lupus, these data are highly concerning," Dr. Jinoos Yazdany said in an interview prior to the annual meeting of the American College of Rheumatology, where the study was presented. "For many individuals with lupus, treatment is very effective and has a proven track record of preventing life-threatening complications and long-term organ damage such as kidney failure. The fact that less than one in three patients are adhering with treatment is alarming and should serve as a call to action for those of providing care to these patients."
Dr. Yazdany, associate director of the lupus clinic at the University of California-San Francisco Medical Center, and her colleagues used MAX (Medicaid Analytic eXtract) data from 2000 to 2006 to identify 23,187 patients with systemic lupus erythematosus (SLE) who were taking at least one immunosuppressive or antimalarial drug. They used pharmacy claims to assess adherence to drugs over a period of 180 days by calculating a medication possession ratio (MPR), defined as the proportion of days covered by the total days’ supply dispensed after the first claim for each drug. The researchers also evaluated the proportion of patients who had an MPR of 80% or greater. The oral drugs studied were hydroxychloroquine, azathioprine, methotrexate, mycophenolate mofetil, cyclosporine, tacrolimus, cyclophosphamide, and leflunomide.
The mean age of the 23,187 patients was 38 years, 94% were female, and the racial and ethnic makeup was diverse (40% black, 34% white, 16% Hispanic, 5% Asian, and 5% other). The highest proportion of SLE patients lived in the southern United States (36%).
Dr. Yazdany reported that the average MPR ranged from 31.1% for tacrolimus to 56% for hydroxychloroquine. In addition, for most drugs, fewer than one in three patients had an MPR of at least 80%. Overall adherence was poorest among those taking tacrolimus (14%) and highest among those taking mycophenolate mofetil (40%).
Across all medications, blacks had lower adherence, compared with whites, and adherence was highest for those residing in the Northeast.
"Our findings underscore the need to understand the reasons behind low adherence in this high-risk and vulnerable group of patients," Dr. Yazdany said. "And we urgently need to develop interventions to improve adherence. Physicians may be unaware of their patient’s adherence with medication, and patients may not be forthcoming about this issue. Concerns about side effects, inadequate understanding of the benefit, and the medication’s cost may be barriers for patients. Treatment-associated side effects may be another important barrier. We need better patient-physician communication around the issue of adherence."
The Medicaid administrative data allowed for an otherwise unobtainable nationwide view of adherence in SLE, but Dr. Yazdany acknowledged certain limitations of the study, including the fact that treatment "may be interrupted for clinically appropriate reasons, so the medication possession ratios in our study may underestimate actual adherence. Also, pharmacy claims are imperfect proxies for whether patients actually take medications that are dispensed, which might lead to an overestimation of adherence."
Dr. Yazdany disclosed that her research is funded by the National Institutes of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases. She had no other relevant financial conflicts to disclose.
AT THE ACR ANNUAL MEETING
Major finding: Among Medicaid beneficiaries with SLE, the average medication possession ratio ranged from 31.1% for tacrolimus to 56% for hydroxychloroquine.
Data source: A study of 23,187 Medicaid patients with SLE who were taking at least one immunosuppressive or antimalarial drug between 2000 and 2006.
Disclosures: Dr. Yazdany disclosed that her research is funded by the National Institutes of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases. She had no other relevant financial conflicts to disclose.
Apremilast promising for Behçet’s syndrome
ISTANBUL, TURKEY – The investigational oral phosphodiesterase-4 inhibitor apremilast showed dramatic efficacy in the treatment of oral and genital ulcers of Behçet’s syndrome in a randomized, double-blind clinical trial.
The multicenter phase II study included 111 Behçet’s syndrome patients with active oral ulcers. They were randomized to 12 weeks of double-blind apremilast at 30 mg twice daily or placebo followed by an additional 12 weeks of open-label apremilast for all, then 4 weeks of post-treatment observation.
Participants had a mean of 2.8 oral ulcers at baseline. The primary study endpoint was the number present at week 12: a mean of 0.5 in the apremilast group, compared with 2.1 in controls, Dr. Gülen Hatemi reported at the annual congress of the European Academy of Dermatology and Venereology.
A total of 71% of patients in the apremilast group were oral ulcer–free at week 12, as were 29% of controls, added Dr. Hatemi, a rheumatologist at Istanbul (Turkey) University.
The benefit of apremilast occurred rapidly. The full response was seen at week 2 and was then maintained throughout the remainder of the 12 weeks of double-blind therapy and the following 12 weeks of open-label apremilast. However, oral ulcers returned in full force quickly after treatment cessation.
Pain from oral ulcers on a 0-100 visual analog scale plummeted in the apremilast group by a mean 44.7 points from a baseline of score of 54. In contrast, pain scores in the control group improved by 16 points, which is less than the 20-point threshold accepted as being clinically meaningful.
Behçet’s syndrome is an immune-mediated small-vessel vasculitis. Its mucocutaneous manifestations can be disabling and resistant to conventional therapies, Dr. Hatemi noted.
Only 16 patients had genital ulcers. All 10 randomized to apremilast were free of the ulcers at week 12, as were three of six affected patients in the control group.
Average scores on the Behçet’s Disease Activity Index dropped from 3.4 at baseline to 2.0 at 12 weeks in the apremilast arm while remaining unchanged in controls. Mean scores on the Behçet’s Disease Quality of Life instrument decreased by 4.5 points from a baseline of 12.6 in apremilast-treated patients, compared with a 1.6-point reduction in controls.
The investigators said no serious adverse events were related to apremilast.
Dr. Hatemi received a research grant from Celgene, the study sponsor, which is also developing the oral drug as a possible treatment for psoriasis, psoriatic arthritis, and ankylosing spondylitis.
ISTANBUL, TURKEY – The investigational oral phosphodiesterase-4 inhibitor apremilast showed dramatic efficacy in the treatment of oral and genital ulcers of Behçet’s syndrome in a randomized, double-blind clinical trial.
The multicenter phase II study included 111 Behçet’s syndrome patients with active oral ulcers. They were randomized to 12 weeks of double-blind apremilast at 30 mg twice daily or placebo followed by an additional 12 weeks of open-label apremilast for all, then 4 weeks of post-treatment observation.
Participants had a mean of 2.8 oral ulcers at baseline. The primary study endpoint was the number present at week 12: a mean of 0.5 in the apremilast group, compared with 2.1 in controls, Dr. Gülen Hatemi reported at the annual congress of the European Academy of Dermatology and Venereology.
A total of 71% of patients in the apremilast group were oral ulcer–free at week 12, as were 29% of controls, added Dr. Hatemi, a rheumatologist at Istanbul (Turkey) University.
The benefit of apremilast occurred rapidly. The full response was seen at week 2 and was then maintained throughout the remainder of the 12 weeks of double-blind therapy and the following 12 weeks of open-label apremilast. However, oral ulcers returned in full force quickly after treatment cessation.
Pain from oral ulcers on a 0-100 visual analog scale plummeted in the apremilast group by a mean 44.7 points from a baseline of score of 54. In contrast, pain scores in the control group improved by 16 points, which is less than the 20-point threshold accepted as being clinically meaningful.
Behçet’s syndrome is an immune-mediated small-vessel vasculitis. Its mucocutaneous manifestations can be disabling and resistant to conventional therapies, Dr. Hatemi noted.
Only 16 patients had genital ulcers. All 10 randomized to apremilast were free of the ulcers at week 12, as were three of six affected patients in the control group.
Average scores on the Behçet’s Disease Activity Index dropped from 3.4 at baseline to 2.0 at 12 weeks in the apremilast arm while remaining unchanged in controls. Mean scores on the Behçet’s Disease Quality of Life instrument decreased by 4.5 points from a baseline of 12.6 in apremilast-treated patients, compared with a 1.6-point reduction in controls.
The investigators said no serious adverse events were related to apremilast.
Dr. Hatemi received a research grant from Celgene, the study sponsor, which is also developing the oral drug as a possible treatment for psoriasis, psoriatic arthritis, and ankylosing spondylitis.
ISTANBUL, TURKEY – The investigational oral phosphodiesterase-4 inhibitor apremilast showed dramatic efficacy in the treatment of oral and genital ulcers of Behçet’s syndrome in a randomized, double-blind clinical trial.
The multicenter phase II study included 111 Behçet’s syndrome patients with active oral ulcers. They were randomized to 12 weeks of double-blind apremilast at 30 mg twice daily or placebo followed by an additional 12 weeks of open-label apremilast for all, then 4 weeks of post-treatment observation.
Participants had a mean of 2.8 oral ulcers at baseline. The primary study endpoint was the number present at week 12: a mean of 0.5 in the apremilast group, compared with 2.1 in controls, Dr. Gülen Hatemi reported at the annual congress of the European Academy of Dermatology and Venereology.
A total of 71% of patients in the apremilast group were oral ulcer–free at week 12, as were 29% of controls, added Dr. Hatemi, a rheumatologist at Istanbul (Turkey) University.
The benefit of apremilast occurred rapidly. The full response was seen at week 2 and was then maintained throughout the remainder of the 12 weeks of double-blind therapy and the following 12 weeks of open-label apremilast. However, oral ulcers returned in full force quickly after treatment cessation.
Pain from oral ulcers on a 0-100 visual analog scale plummeted in the apremilast group by a mean 44.7 points from a baseline of score of 54. In contrast, pain scores in the control group improved by 16 points, which is less than the 20-point threshold accepted as being clinically meaningful.
Behçet’s syndrome is an immune-mediated small-vessel vasculitis. Its mucocutaneous manifestations can be disabling and resistant to conventional therapies, Dr. Hatemi noted.
Only 16 patients had genital ulcers. All 10 randomized to apremilast were free of the ulcers at week 12, as were three of six affected patients in the control group.
Average scores on the Behçet’s Disease Activity Index dropped from 3.4 at baseline to 2.0 at 12 weeks in the apremilast arm while remaining unchanged in controls. Mean scores on the Behçet’s Disease Quality of Life instrument decreased by 4.5 points from a baseline of 12.6 in apremilast-treated patients, compared with a 1.6-point reduction in controls.
The investigators said no serious adverse events were related to apremilast.
Dr. Hatemi received a research grant from Celgene, the study sponsor, which is also developing the oral drug as a possible treatment for psoriasis, psoriatic arthritis, and ankylosing spondylitis.
AT THE EADV CONGRESS
Major finding: The mean number of oral ulcers in Behçet’s syndrome patients treated with apremilast quickly dropped from 2.8 at baseline to 0.5, an improvement maintained during 24 weeks of therapy.
Data source: A randomized, double-blind, multicenter, 28-week phase II study involving 111 Behçet’s syndrome patients with active oral ulcers.
Disclosures: The presenter received a research grant from the study sponsor, Celgene.
Systemic sclerosis overlap syndromes called distinct entity
ISTANBUL – Systemic sclerosis overlap syndromes follow a path of disease progression distinctly different from that of limited or diffuse cutaneous systemic sclerosis, according to a new analysis from the German Network for Systemic Scleroderma.
"This study shows for the first time that patients suffering from systemic sclerosis overlap syndromes should be viewed as a distinct systemic sclerosis subset," Dr. Pia Moinzadeh noted at the annual congress of the European Academy of Dermatology and Venereology.
Those with systemic sclerosis overlap syndromes (SSc-OS) are a heterogeneous subgroup of patients who have the clinical features of SSc according to American College of Rheumatology criteria simultaneously with the characteristic findings of at least one other connective tissue disease, such as dermatomyositis, Sjögren’s syndrome, rheumatoid arthritis, or lupus erythematosus.
The registry of the German Network for Systemic Scleroderma is a uniquely large and inclusive ongoing prospective data base directed by a joint committee of dermatologists and rheumatologists. More than 6 years ago, when the investigators realized that a sizable fraction of patients enrolled in the national registry didn’t fit within the classic bimodal SSc categorization scheme composed of two subtypes – limited cutaneous and diffuse cutaneous SSc – they proposed three additional SSc subtypes. These are SSc-OS, undifferentiated scleroderma, and sclerosis sine scleroderma.
The focus of Dr. Moinzadeh’s EADV presentation was on SSc-OS. Of 3,240 SSc patients in the German registry, patients with OS comprise 11%, while those with limited cutaneous SSc make up 43%, those with diffuse cutaneous disease account for 31%, and the remainder of patients have undifferentiated scleroderma or sclerosis sine scleroderma.
The validity of the SSc-OS diagnostic construct has been controversial. Some experts have contended that patients with SSc-OS ought to be lumped under the headings of limited cutaneous SSc or diffuse cutaneous SSc, depending on the extent of skin involvement. However, the unparalleled size, inclusiveness, and long-term prospective follow-up provided by the German registry have resulted in evidence that argues to the contrary, according to Dr. Moinzadeh, a rheumatologist at the University of Cologne (Germany).
She reported on 326 patients with SSc-OS, 1,598 with limited cutaneous SSc, and 996 with diffuse cutaneous SSc prospectively followed through the German registry for a mean of 10.1 years. The follow-up data indicate SSc-OS evolves differently from either limited cutaneous SSc or diffuse cutaneous SSc. It also displays a distinctive pattern of organ involvement and autoantibody positivity, she noted.
The group with SSc-OS had a mean modified Rodnan skin score of 6.7, similar to the 7.2 score in the limited cutaneous SSc group and markedly less than the 15.8 in the diffuse cutaneous SSc patients. While this might suggest that patients with SSc-OS have a milder course of disease comparable to limited cutaneous SSc, in fact they progressed more rapidly, with earlier and more widespread significant organ involvement and a higher disease burden than the limited cutaneous SSc group, according to Dr. Moinzadeh.
Patients with SSc-OS developed musculoskeletal involvement far more frequently than others. Sixty-seven percent were affected, compared with 38% of patients with limited cutaneous SSc and 48% with diffuse cutaneous SSc. The patients in the SSc-OS group also developed musculoskeletal involvement earlier.
Lung fibrosis occurred in 38% of patients with SSc-OS, 27% of patients with limited cutaneous SSc, and 63% with diffuse cutaneous SSc. Cardiac involvement occurred in 15% with SSc-OS, 10% with limited cutaneous SSc, and 19% with diffuse cutaneous SSc. The onset of these manifestations of SSc in the SSc-OS subgroup was significantly earlier than in the limited cutaneous SSc patients but later than with diffuse cutaneous SSc.
Progression to esophageal and renal involvement, as well as pulmonary arterial hypertension, in the SSc-OS group occurred significantly earlier than in the limited cutaneous SSC patients, but again was later than with diffuse cutaneous SSC.
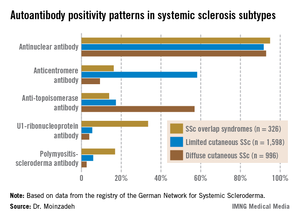
The autoantibody status of the three subgroups showed distinct differences (see graphic).
The German Network for Systemic Scleroderma is funded by the German Federal Ministry of Education and Research. Dr. Moinzadeh reported having no financial conflicts.
ISTANBUL – Systemic sclerosis overlap syndromes follow a path of disease progression distinctly different from that of limited or diffuse cutaneous systemic sclerosis, according to a new analysis from the German Network for Systemic Scleroderma.
"This study shows for the first time that patients suffering from systemic sclerosis overlap syndromes should be viewed as a distinct systemic sclerosis subset," Dr. Pia Moinzadeh noted at the annual congress of the European Academy of Dermatology and Venereology.

Those with systemic sclerosis overlap syndromes (SSc-OS) are a heterogeneous subgroup of patients who have the clinical features of SSc according to American College of Rheumatology criteria simultaneously with the characteristic findings of at least one other connective tissue disease, such as dermatomyositis, Sjögren’s syndrome, rheumatoid arthritis, or lupus erythematosus.
The registry of the German Network for Systemic Scleroderma is a uniquely large and inclusive ongoing prospective data base directed by a joint committee of dermatologists and rheumatologists. More than 6 years ago, when the investigators realized that a sizable fraction of patients enrolled in the national registry didn’t fit within the classic bimodal SSc categorization scheme composed of two subtypes – limited cutaneous and diffuse cutaneous SSc – they proposed three additional SSc subtypes. These are SSc-OS, undifferentiated scleroderma, and sclerosis sine scleroderma.
The focus of Dr. Moinzadeh’s EADV presentation was on SSc-OS. Of 3,240 SSc patients in the German registry, patients with OS comprise 11%, while those with limited cutaneous SSc make up 43%, those with diffuse cutaneous disease account for 31%, and the remainder of patients have undifferentiated scleroderma or sclerosis sine scleroderma.
The validity of the SSc-OS diagnostic construct has been controversial. Some experts have contended that patients with SSc-OS ought to be lumped under the headings of limited cutaneous SSc or diffuse cutaneous SSc, depending on the extent of skin involvement. However, the unparalleled size, inclusiveness, and long-term prospective follow-up provided by the German registry have resulted in evidence that argues to the contrary, according to Dr. Moinzadeh, a rheumatologist at the University of Cologne (Germany).
She reported on 326 patients with SSc-OS, 1,598 with limited cutaneous SSc, and 996 with diffuse cutaneous SSc prospectively followed through the German registry for a mean of 10.1 years. The follow-up data indicate SSc-OS evolves differently from either limited cutaneous SSc or diffuse cutaneous SSc. It also displays a distinctive pattern of organ involvement and autoantibody positivity, she noted.
The group with SSc-OS had a mean modified Rodnan skin score of 6.7, similar to the 7.2 score in the limited cutaneous SSc group and markedly less than the 15.8 in the diffuse cutaneous SSc patients. While this might suggest that patients with SSc-OS have a milder course of disease comparable to limited cutaneous SSc, in fact they progressed more rapidly, with earlier and more widespread significant organ involvement and a higher disease burden than the limited cutaneous SSc group, according to Dr. Moinzadeh.
Patients with SSc-OS developed musculoskeletal involvement far more frequently than others. Sixty-seven percent were affected, compared with 38% of patients with limited cutaneous SSc and 48% with diffuse cutaneous SSc. The patients in the SSc-OS group also developed musculoskeletal involvement earlier.
Lung fibrosis occurred in 38% of patients with SSc-OS, 27% of patients with limited cutaneous SSc, and 63% with diffuse cutaneous SSc. Cardiac involvement occurred in 15% with SSc-OS, 10% with limited cutaneous SSc, and 19% with diffuse cutaneous SSc. The onset of these manifestations of SSc in the SSc-OS subgroup was significantly earlier than in the limited cutaneous SSc patients but later than with diffuse cutaneous SSc.
Progression to esophageal and renal involvement, as well as pulmonary arterial hypertension, in the SSc-OS group occurred significantly earlier than in the limited cutaneous SSC patients, but again was later than with diffuse cutaneous SSC.
The autoantibody status of the three subgroups showed distinct differences (see graphic).
The German Network for Systemic Scleroderma is funded by the German Federal Ministry of Education and Research. Dr. Moinzadeh reported having no financial conflicts.
ISTANBUL – Systemic sclerosis overlap syndromes follow a path of disease progression distinctly different from that of limited or diffuse cutaneous systemic sclerosis, according to a new analysis from the German Network for Systemic Scleroderma.
"This study shows for the first time that patients suffering from systemic sclerosis overlap syndromes should be viewed as a distinct systemic sclerosis subset," Dr. Pia Moinzadeh noted at the annual congress of the European Academy of Dermatology and Venereology.

Those with systemic sclerosis overlap syndromes (SSc-OS) are a heterogeneous subgroup of patients who have the clinical features of SSc according to American College of Rheumatology criteria simultaneously with the characteristic findings of at least one other connective tissue disease, such as dermatomyositis, Sjögren’s syndrome, rheumatoid arthritis, or lupus erythematosus.
The registry of the German Network for Systemic Scleroderma is a uniquely large and inclusive ongoing prospective data base directed by a joint committee of dermatologists and rheumatologists. More than 6 years ago, when the investigators realized that a sizable fraction of patients enrolled in the national registry didn’t fit within the classic bimodal SSc categorization scheme composed of two subtypes – limited cutaneous and diffuse cutaneous SSc – they proposed three additional SSc subtypes. These are SSc-OS, undifferentiated scleroderma, and sclerosis sine scleroderma.
The focus of Dr. Moinzadeh’s EADV presentation was on SSc-OS. Of 3,240 SSc patients in the German registry, patients with OS comprise 11%, while those with limited cutaneous SSc make up 43%, those with diffuse cutaneous disease account for 31%, and the remainder of patients have undifferentiated scleroderma or sclerosis sine scleroderma.
The validity of the SSc-OS diagnostic construct has been controversial. Some experts have contended that patients with SSc-OS ought to be lumped under the headings of limited cutaneous SSc or diffuse cutaneous SSc, depending on the extent of skin involvement. However, the unparalleled size, inclusiveness, and long-term prospective follow-up provided by the German registry have resulted in evidence that argues to the contrary, according to Dr. Moinzadeh, a rheumatologist at the University of Cologne (Germany).
She reported on 326 patients with SSc-OS, 1,598 with limited cutaneous SSc, and 996 with diffuse cutaneous SSc prospectively followed through the German registry for a mean of 10.1 years. The follow-up data indicate SSc-OS evolves differently from either limited cutaneous SSc or diffuse cutaneous SSc. It also displays a distinctive pattern of organ involvement and autoantibody positivity, she noted.
The group with SSc-OS had a mean modified Rodnan skin score of 6.7, similar to the 7.2 score in the limited cutaneous SSc group and markedly less than the 15.8 in the diffuse cutaneous SSc patients. While this might suggest that patients with SSc-OS have a milder course of disease comparable to limited cutaneous SSc, in fact they progressed more rapidly, with earlier and more widespread significant organ involvement and a higher disease burden than the limited cutaneous SSc group, according to Dr. Moinzadeh.
Patients with SSc-OS developed musculoskeletal involvement far more frequently than others. Sixty-seven percent were affected, compared with 38% of patients with limited cutaneous SSc and 48% with diffuse cutaneous SSc. The patients in the SSc-OS group also developed musculoskeletal involvement earlier.
Lung fibrosis occurred in 38% of patients with SSc-OS, 27% of patients with limited cutaneous SSc, and 63% with diffuse cutaneous SSc. Cardiac involvement occurred in 15% with SSc-OS, 10% with limited cutaneous SSc, and 19% with diffuse cutaneous SSc. The onset of these manifestations of SSc in the SSc-OS subgroup was significantly earlier than in the limited cutaneous SSc patients but later than with diffuse cutaneous SSc.
Progression to esophageal and renal involvement, as well as pulmonary arterial hypertension, in the SSc-OS group occurred significantly earlier than in the limited cutaneous SSC patients, but again was later than with diffuse cutaneous SSC.
The autoantibody status of the three subgroups showed distinct differences (see graphic).
The German Network for Systemic Scleroderma is funded by the German Federal Ministry of Education and Research. Dr. Moinzadeh reported having no financial conflicts.
AT THE EADV CONGRESS
Major finding: Musculoskeletal involvement occurred in 67% of patients with systemic sclerosis overlap syndromes, compared with 38% of those with limited cutaneous systemic sclerosis and 48% with diffuse cutaneous systemic sclerosis.
Data source: This analysis from the ongoing prospective national registry maintained by the German Network for Systemic Scleroderma included nearly 3,000 patients with systemic sclerosis followed for a mean of 10.1 years.
Disclosures: The German Network for Systemic Scleroderma is funded by the German Federal Ministry of Education and Research. Dr. Moinzadeh reported having no financial conflicts.
Is PASI 90 becoming the new PASI 75?
ISTANBUL – With the majority of psoriasis patients now achieving PASI 90 responses in randomized trials of the latest-generation biologic agents, a push is on to replace PASI 75 with PASI 90 as the new goal defining treatment success. But some dermatologists have misgivings about raising the bar.
"More and more, editorialists are promoting the idea of silencing psoriasis in all patients. This is a tricky and challenging goal," Dr. Hervé Bachelez said at the annual congress of the European Academy of Dermatology and Venereology.
"You’ve probably noticed that PASI 90 and even PASI 100 are becoming important as secondary endpoints in virtually all clinical trials. It’s good, it’s legitimate, and the PASI 90 probably better reflects the wishes of the patient and the physician than the PASI 75. But we have to wait and see what the caveats of this are. You can say, ‘Let’s push the response rate up to PASI 100 in all patients,’ but the danger is that if you cross a line, you may be unable to precisely regulate the level of immunosuppression in some patients. Basically you can expect some safety issues in real life that you would not see in clinical trials," cautioned Dr. Bachelez, professor of dermatology and head of the inflammatory skin diseases unit at Saint Louis University Hospital, Paris.
Not so many years ago the notion of PASI 90 responses in high double figures seemed a pipedream, he observed. For example, the week-12 PASI 90 rate in published randomized trials of methotrexate was only 9%, while for the first-generation tumor necrosis factor inhibitor etanercept, the rates were 19%-23%. In contrast, among the highlights of this year’s EADV congress were the presentation of results from clinical trials of two investigational interleukin-17 inhibitors: In the pivotal Phase III FIXTURE trial, secukinumab-treated patients had a PASI 90 response rate of 72% at week 16, while the week-16 PASI 90 rate in the Phase II OLE trial was 87% in patients on brodalimab, and even out to week 96, it was 78%.
Underscoring Dr. Bachelez’ concern that the randomized trial experience likely underestimates the true extent of safety hazards posed by potent therapies in daily clinical practice was a report by a consortium of 13 Spanish dermatology departments responsible for the BIOBADADERM registry. The Spanish registry is focused on safety and includes only psoriasis patients on systemic therapy, whether biologics or classic drugs. Among the first 1,042 enrollees receiving systemic therapy, fully 30% would not have been eligible for participation in randomized controlled trials for various reasons, including age greater than 70 years, having chronic kidney or liver disease, a history of hepatitis B or C, HIV infection, or cancer, or having psoriasis of a type other than chronic plaque disease.
The disturbing finding was that during 2,179 person-years of prospective follow-up, the large group of patients ineligible for randomized trials had a 2.7-fold increased risk of serious adverse events compared with patients on systemic therapy who were eligible for study participation.
The number needed to harm was calculated as follows: For every 40 patients treated with systemic therapy for 2.1 years despite not being eligible for randomized trials, one additional serious adverse event can be expected compared with similar treatment in randomized trial-eligible patients, according to the investigators (Arch. Dermatol. 2012;148:463-70). And that’s without pushing the envelope by trying to aim for a PASI 90 response, Dr. Bachelez noted.
A contrary view regarding PASI 90 as an emerging standard of treatment excellence was put forth elsewhere at the meeting by Dr. Peter van de Kerkhof, professor and head of the department of dermatology at Radboud University in Nijmegen, the Netherlands.
He cited multiple studies demonstrating that substantial PASI reductions may not translate into tangible improvements in patients’ quality of life. For example, among psoriasis patients who achieved a PASI 75 response in one European study, 65% still had a Dermatology Life Quality Index (DLQI) score of 2 or more (Eur. J. Dermatol. 2010;20:62-7).
"This implies that there is something more to be wished for by patients, even when PASI 75 is reached," according to Dr. van de Kerkhof.
Moreover, in another trial, even among patients with a PASI score of 0 at week 24, only 70% had an optimal DLQI of 0, not 100% as most dermatologists might expect (Br. J. Dermatol. 2006;154:1161-8).
A recent survey of 2,151 European psoriasis patients and their dermatologists highlighted a substantial degree of dissatisfaction with current therapies. Patients on biologics had higher rates of improvement from severe to moderate or mild disease than did those on any other forms of psoriasis therapy, yet 41% of patients on biologics were dissatisfied with their treatment (J. Dermatolog. Treat. 2013;24:193-8).
"I think it’s extremely important that we continue to innovate treatment possibilities for psoriasis in order to improve outcomes for our patients," Dr. van de Kerkhof said.
What patients really want, surveys suggest, are treatments that render them clear or nearly clear, and do so quickly. In one survey, patients rated as the most important characteristic about a therapy the rapidity with which it could achieve a moderate 50% improvement in symptoms. They rated that as higher in importance to them than the therapy’s long-term risks (Arch. Dermatol. 2007;143:1175-9), Dr. van de Kerkhof noted.
Patients also place a high priority on improvement of a broad array of symptoms that aren’t captured by either the PASI or the DLQI, Dr. Bruce E. Strober noted in a separate presentation. These include itching, plaque-related pain, altered skin appearance, flaking, and bleeding.
For this reason, he and a group of his coworkers have created and are now validating a new tool for the assessment of patient-related outcomes in psoriasis called the Psoriasis Symptom Diary (Value Health 2013;16:1014-22). The 16-item tool takes less than 5 minutes for a patient to fill out and is designed to replace the DLQI both in clinical trials and everyday practice. The goal is to be able to walk into the examination room, take a quick look at the Psoriasis Symptom Diary, and know from that how a patient is currently doing even before asking for the patient to disrobe.
"Let’s face it: Outside of skin cancer, when we’re doing dermatology, we’re in the quality of life business. That means we’re asking patients how they’re doing at every visit for psoriasis, atopic dermatitis, or severe acne. The DLQI does that, but it’s not psoriasis specific," said Dr. Strober, vice chair and director of the clinical trials unit in the department of dermatology at the University of Connecticut, Farmington.
As for the PASI, he doesn’t use it except in structured clinical trials. It’s too time consuming and has a high rate of inter- and intrarater variability.
"In the United States, dermatologists never do PASI scores in their clinics. The PASI score has numerous drawbacks that make it impractical in a regular practice setting," Dr. Strober said. "In my own practice, I routinely do a 5-point Physician’s Global Assessment along with an estimate of involved body surface area. I think that gives you a good picture of the objective level of psoriasis severity."
Dr. Bachelez, Dr. van de Kerkhof, and Dr. Strober each reported receiving research grants from and serving on advisory boards for 9-14 pharmaceutical companies engaged in developing new treatments for psoriasis.
ISTANBUL – With the majority of psoriasis patients now achieving PASI 90 responses in randomized trials of the latest-generation biologic agents, a push is on to replace PASI 75 with PASI 90 as the new goal defining treatment success. But some dermatologists have misgivings about raising the bar.
"More and more, editorialists are promoting the idea of silencing psoriasis in all patients. This is a tricky and challenging goal," Dr. Hervé Bachelez said at the annual congress of the European Academy of Dermatology and Venereology.
"You’ve probably noticed that PASI 90 and even PASI 100 are becoming important as secondary endpoints in virtually all clinical trials. It’s good, it’s legitimate, and the PASI 90 probably better reflects the wishes of the patient and the physician than the PASI 75. But we have to wait and see what the caveats of this are. You can say, ‘Let’s push the response rate up to PASI 100 in all patients,’ but the danger is that if you cross a line, you may be unable to precisely regulate the level of immunosuppression in some patients. Basically you can expect some safety issues in real life that you would not see in clinical trials," cautioned Dr. Bachelez, professor of dermatology and head of the inflammatory skin diseases unit at Saint Louis University Hospital, Paris.
Not so many years ago the notion of PASI 90 responses in high double figures seemed a pipedream, he observed. For example, the week-12 PASI 90 rate in published randomized trials of methotrexate was only 9%, while for the first-generation tumor necrosis factor inhibitor etanercept, the rates were 19%-23%. In contrast, among the highlights of this year’s EADV congress were the presentation of results from clinical trials of two investigational interleukin-17 inhibitors: In the pivotal Phase III FIXTURE trial, secukinumab-treated patients had a PASI 90 response rate of 72% at week 16, while the week-16 PASI 90 rate in the Phase II OLE trial was 87% in patients on brodalimab, and even out to week 96, it was 78%.
Underscoring Dr. Bachelez’ concern that the randomized trial experience likely underestimates the true extent of safety hazards posed by potent therapies in daily clinical practice was a report by a consortium of 13 Spanish dermatology departments responsible for the BIOBADADERM registry. The Spanish registry is focused on safety and includes only psoriasis patients on systemic therapy, whether biologics or classic drugs. Among the first 1,042 enrollees receiving systemic therapy, fully 30% would not have been eligible for participation in randomized controlled trials for various reasons, including age greater than 70 years, having chronic kidney or liver disease, a history of hepatitis B or C, HIV infection, or cancer, or having psoriasis of a type other than chronic plaque disease.
The disturbing finding was that during 2,179 person-years of prospective follow-up, the large group of patients ineligible for randomized trials had a 2.7-fold increased risk of serious adverse events compared with patients on systemic therapy who were eligible for study participation.
The number needed to harm was calculated as follows: For every 40 patients treated with systemic therapy for 2.1 years despite not being eligible for randomized trials, one additional serious adverse event can be expected compared with similar treatment in randomized trial-eligible patients, according to the investigators (Arch. Dermatol. 2012;148:463-70). And that’s without pushing the envelope by trying to aim for a PASI 90 response, Dr. Bachelez noted.
A contrary view regarding PASI 90 as an emerging standard of treatment excellence was put forth elsewhere at the meeting by Dr. Peter van de Kerkhof, professor and head of the department of dermatology at Radboud University in Nijmegen, the Netherlands.
He cited multiple studies demonstrating that substantial PASI reductions may not translate into tangible improvements in patients’ quality of life. For example, among psoriasis patients who achieved a PASI 75 response in one European study, 65% still had a Dermatology Life Quality Index (DLQI) score of 2 or more (Eur. J. Dermatol. 2010;20:62-7).
"This implies that there is something more to be wished for by patients, even when PASI 75 is reached," according to Dr. van de Kerkhof.
Moreover, in another trial, even among patients with a PASI score of 0 at week 24, only 70% had an optimal DLQI of 0, not 100% as most dermatologists might expect (Br. J. Dermatol. 2006;154:1161-8).
A recent survey of 2,151 European psoriasis patients and their dermatologists highlighted a substantial degree of dissatisfaction with current therapies. Patients on biologics had higher rates of improvement from severe to moderate or mild disease than did those on any other forms of psoriasis therapy, yet 41% of patients on biologics were dissatisfied with their treatment (J. Dermatolog. Treat. 2013;24:193-8).
"I think it’s extremely important that we continue to innovate treatment possibilities for psoriasis in order to improve outcomes for our patients," Dr. van de Kerkhof said.
What patients really want, surveys suggest, are treatments that render them clear or nearly clear, and do so quickly. In one survey, patients rated as the most important characteristic about a therapy the rapidity with which it could achieve a moderate 50% improvement in symptoms. They rated that as higher in importance to them than the therapy’s long-term risks (Arch. Dermatol. 2007;143:1175-9), Dr. van de Kerkhof noted.
Patients also place a high priority on improvement of a broad array of symptoms that aren’t captured by either the PASI or the DLQI, Dr. Bruce E. Strober noted in a separate presentation. These include itching, plaque-related pain, altered skin appearance, flaking, and bleeding.
For this reason, he and a group of his coworkers have created and are now validating a new tool for the assessment of patient-related outcomes in psoriasis called the Psoriasis Symptom Diary (Value Health 2013;16:1014-22). The 16-item tool takes less than 5 minutes for a patient to fill out and is designed to replace the DLQI both in clinical trials and everyday practice. The goal is to be able to walk into the examination room, take a quick look at the Psoriasis Symptom Diary, and know from that how a patient is currently doing even before asking for the patient to disrobe.
"Let’s face it: Outside of skin cancer, when we’re doing dermatology, we’re in the quality of life business. That means we’re asking patients how they’re doing at every visit for psoriasis, atopic dermatitis, or severe acne. The DLQI does that, but it’s not psoriasis specific," said Dr. Strober, vice chair and director of the clinical trials unit in the department of dermatology at the University of Connecticut, Farmington.
As for the PASI, he doesn’t use it except in structured clinical trials. It’s too time consuming and has a high rate of inter- and intrarater variability.
"In the United States, dermatologists never do PASI scores in their clinics. The PASI score has numerous drawbacks that make it impractical in a regular practice setting," Dr. Strober said. "In my own practice, I routinely do a 5-point Physician’s Global Assessment along with an estimate of involved body surface area. I think that gives you a good picture of the objective level of psoriasis severity."
Dr. Bachelez, Dr. van de Kerkhof, and Dr. Strober each reported receiving research grants from and serving on advisory boards for 9-14 pharmaceutical companies engaged in developing new treatments for psoriasis.
ISTANBUL – With the majority of psoriasis patients now achieving PASI 90 responses in randomized trials of the latest-generation biologic agents, a push is on to replace PASI 75 with PASI 90 as the new goal defining treatment success. But some dermatologists have misgivings about raising the bar.
"More and more, editorialists are promoting the idea of silencing psoriasis in all patients. This is a tricky and challenging goal," Dr. Hervé Bachelez said at the annual congress of the European Academy of Dermatology and Venereology.
"You’ve probably noticed that PASI 90 and even PASI 100 are becoming important as secondary endpoints in virtually all clinical trials. It’s good, it’s legitimate, and the PASI 90 probably better reflects the wishes of the patient and the physician than the PASI 75. But we have to wait and see what the caveats of this are. You can say, ‘Let’s push the response rate up to PASI 100 in all patients,’ but the danger is that if you cross a line, you may be unable to precisely regulate the level of immunosuppression in some patients. Basically you can expect some safety issues in real life that you would not see in clinical trials," cautioned Dr. Bachelez, professor of dermatology and head of the inflammatory skin diseases unit at Saint Louis University Hospital, Paris.
Not so many years ago the notion of PASI 90 responses in high double figures seemed a pipedream, he observed. For example, the week-12 PASI 90 rate in published randomized trials of methotrexate was only 9%, while for the first-generation tumor necrosis factor inhibitor etanercept, the rates were 19%-23%. In contrast, among the highlights of this year’s EADV congress were the presentation of results from clinical trials of two investigational interleukin-17 inhibitors: In the pivotal Phase III FIXTURE trial, secukinumab-treated patients had a PASI 90 response rate of 72% at week 16, while the week-16 PASI 90 rate in the Phase II OLE trial was 87% in patients on brodalimab, and even out to week 96, it was 78%.
Underscoring Dr. Bachelez’ concern that the randomized trial experience likely underestimates the true extent of safety hazards posed by potent therapies in daily clinical practice was a report by a consortium of 13 Spanish dermatology departments responsible for the BIOBADADERM registry. The Spanish registry is focused on safety and includes only psoriasis patients on systemic therapy, whether biologics or classic drugs. Among the first 1,042 enrollees receiving systemic therapy, fully 30% would not have been eligible for participation in randomized controlled trials for various reasons, including age greater than 70 years, having chronic kidney or liver disease, a history of hepatitis B or C, HIV infection, or cancer, or having psoriasis of a type other than chronic plaque disease.
The disturbing finding was that during 2,179 person-years of prospective follow-up, the large group of patients ineligible for randomized trials had a 2.7-fold increased risk of serious adverse events compared with patients on systemic therapy who were eligible for study participation.
The number needed to harm was calculated as follows: For every 40 patients treated with systemic therapy for 2.1 years despite not being eligible for randomized trials, one additional serious adverse event can be expected compared with similar treatment in randomized trial-eligible patients, according to the investigators (Arch. Dermatol. 2012;148:463-70). And that’s without pushing the envelope by trying to aim for a PASI 90 response, Dr. Bachelez noted.
A contrary view regarding PASI 90 as an emerging standard of treatment excellence was put forth elsewhere at the meeting by Dr. Peter van de Kerkhof, professor and head of the department of dermatology at Radboud University in Nijmegen, the Netherlands.
He cited multiple studies demonstrating that substantial PASI reductions may not translate into tangible improvements in patients’ quality of life. For example, among psoriasis patients who achieved a PASI 75 response in one European study, 65% still had a Dermatology Life Quality Index (DLQI) score of 2 or more (Eur. J. Dermatol. 2010;20:62-7).
"This implies that there is something more to be wished for by patients, even when PASI 75 is reached," according to Dr. van de Kerkhof.
Moreover, in another trial, even among patients with a PASI score of 0 at week 24, only 70% had an optimal DLQI of 0, not 100% as most dermatologists might expect (Br. J. Dermatol. 2006;154:1161-8).
A recent survey of 2,151 European psoriasis patients and their dermatologists highlighted a substantial degree of dissatisfaction with current therapies. Patients on biologics had higher rates of improvement from severe to moderate or mild disease than did those on any other forms of psoriasis therapy, yet 41% of patients on biologics were dissatisfied with their treatment (J. Dermatolog. Treat. 2013;24:193-8).
"I think it’s extremely important that we continue to innovate treatment possibilities for psoriasis in order to improve outcomes for our patients," Dr. van de Kerkhof said.
What patients really want, surveys suggest, are treatments that render them clear or nearly clear, and do so quickly. In one survey, patients rated as the most important characteristic about a therapy the rapidity with which it could achieve a moderate 50% improvement in symptoms. They rated that as higher in importance to them than the therapy’s long-term risks (Arch. Dermatol. 2007;143:1175-9), Dr. van de Kerkhof noted.
Patients also place a high priority on improvement of a broad array of symptoms that aren’t captured by either the PASI or the DLQI, Dr. Bruce E. Strober noted in a separate presentation. These include itching, plaque-related pain, altered skin appearance, flaking, and bleeding.
For this reason, he and a group of his coworkers have created and are now validating a new tool for the assessment of patient-related outcomes in psoriasis called the Psoriasis Symptom Diary (Value Health 2013;16:1014-22). The 16-item tool takes less than 5 minutes for a patient to fill out and is designed to replace the DLQI both in clinical trials and everyday practice. The goal is to be able to walk into the examination room, take a quick look at the Psoriasis Symptom Diary, and know from that how a patient is currently doing even before asking for the patient to disrobe.
"Let’s face it: Outside of skin cancer, when we’re doing dermatology, we’re in the quality of life business. That means we’re asking patients how they’re doing at every visit for psoriasis, atopic dermatitis, or severe acne. The DLQI does that, but it’s not psoriasis specific," said Dr. Strober, vice chair and director of the clinical trials unit in the department of dermatology at the University of Connecticut, Farmington.
As for the PASI, he doesn’t use it except in structured clinical trials. It’s too time consuming and has a high rate of inter- and intrarater variability.
"In the United States, dermatologists never do PASI scores in their clinics. The PASI score has numerous drawbacks that make it impractical in a regular practice setting," Dr. Strober said. "In my own practice, I routinely do a 5-point Physician’s Global Assessment along with an estimate of involved body surface area. I think that gives you a good picture of the objective level of psoriasis severity."
Dr. Bachelez, Dr. van de Kerkhof, and Dr. Strober each reported receiving research grants from and serving on advisory boards for 9-14 pharmaceutical companies engaged in developing new treatments for psoriasis.
EXPERT ANALYSIS FROM THE eadv CONGRESS
New drugs, strategies advance rheumatoid arthritis treatment
New biologics and more aggressive intervention strategies have advanced rheumatoid arthritis treatment over the last decade, Dr. Iain McInnes explains, but remission remains too infrequent. New drugs under development in the coming years offer promise for the future. For more on rheumatoid arthritis advances, visit http://www.rheumatologynews.com.
New biologics and more aggressive intervention strategies have advanced rheumatoid arthritis treatment over the last decade, Dr. Iain McInnes explains, but remission remains too infrequent. New drugs under development in the coming years offer promise for the future. For more on rheumatoid arthritis advances, visit http://www.rheumatologynews.com.
New biologics and more aggressive intervention strategies have advanced rheumatoid arthritis treatment over the last decade, Dr. Iain McInnes explains, but remission remains too infrequent. New drugs under development in the coming years offer promise for the future. For more on rheumatoid arthritis advances, visit http://www.rheumatologynews.com.
Options exist for psoriasis patients with multiple anti-TNF failures
LAS VEGAS – The use of an alternative anti–tumor necrosis factor drug is not necessarily precluded in a psoriasis patient with both skin and joint symptoms who has failed two previous anti-TNF drugs, according to Dr. Kenneth B. Gordon.
Dr. Gordon, professor of dermatology at Northwestern University, Chicago, described a case involving a 52-year-old man with a 20-year history of psoriasis who initially presented with mainly scalp and limited plaque psoriasis, and who was treated with topical corticosteroids and topical calcipotriene. After developing more extensive disease, he was treated successfully with ultraviolet B phototherapy.
Eleven years after first presenting with psoriasis, he presented with peripheral arthritis and enthesitis and was diagnosed with psoriatic arthritis.
The patient was treated initially with sulfasalazine, but failed to respond; methotrexate had only modest benefit, and at higher doses the patient developed liver function test abnormalities due to steatohepatitis, Dr. Gordon said at Perspectives in Rheumatic Diseases 2013.
Etanercept was initiated at 50 mg twice weekly, and the patient had an excellent initial response.
However, after stepping down to 50 mg weekly, his psoriasis flared.
"His joints were doing fine, but his skin was getting bad," Dr. Gordon said, noting that this type of response was also seen in early studies of etanercept, which showed that when doses were reduced to 50 mg weekly after 12 weeks, responses leveled off, with some patients achieving a 75% reduction in the Psoriasis Area and Severity Index (PASI) 75 response, and some losing PASI 75 response.
"It turns out that this occurs in 20%-30% of patients," he said, adding that some controversy exists regarding dosing, because "it’s not entirely clear that if you had just started at the lower dose and just let them go at the 50 mg once a week, that at 6 months they’d be any different than if you gave them the higher dose initially."
The REVEAL study of adalimumab for psoriasis also demonstrated loss of response after 33 weeks among those with an initial PASI 75 response (J. Am. Acad. Dermatol. 2012;66:241-51), he said.
"So clearly, patients lose response. Now, that lessens over time, but in the first year in psoriasis – and my guess is, in psoriatic arthritis as well – you have loss of effect at a relatively high level with all of these medicines," he said.
In fact, in the patient Dr. Gordon presented, a switch to adalimumab at a dose of 40 mg every other week resulted in an excellent initial response in both joints and skin, but skin symptoms returned after 8 months.
"This is something we’re all familiar with – patients doing well on an anti-TNF and then losing effect after a period of time on medication," he said.
So what are the alternatives?
An anti-TNF drug option after the patient cycles through etanercept, adalimumab, and infliximab is certolizumab, which was approved in September for the treatment of psoriatic arthritis. Study data suggest that the drug is "quite effective for psoriasis, was well tolerated, and similar to the other anti-TNFs," he said.
Using certolizumab is a reasonable approach that appears to work for both skin and joint symptoms, he said.
Golimumab is another possible option for treatment, but primary psoriasis data are lacking with this drug.
"It is my feeling that golimumab is probably not as potent for psoriasis as the other anti-TNF agents that we have, but that is an opinion," he said.
As for alternatives to anti-TNF agents, the PHEONIX 2 trial (Lancet 2008;371:1675-84) provided reasonable data in support of the interleukin-12 and -23 monoclonal antibody ustekinumab for skin disease, but patients who failed prior anti-TNF therapy actually had a poorer outcome, so that is something to keep in mind, he said, noting that adding methotrexate may help prevent a flare in patients who are switched from an anti-TNF agent to ustekinumab.
Ustekinumab, which also was approved in September for the treatment of psoriatic arthritis, is likely more useful for the skin, but is not exceptional for joint disease, he said at the meeting, held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Apremilast and tofacitinib, drugs now in development for psoriasis, may also prove useful in the future, as they may "theoretically be of benefit" based on currently available data, he said.
Dr. Gordon has received research support and/or honoraria from AbbVie (which markets adalimumab), Amgen (which markets etanercept), Celgene (which is developing apremilast), Eli Lilly, Janssen (which markets golimumab, infliximab, and ustekinumab), Novartis, and Pfizer (which markets tofacitinib).
LAS VEGAS – The use of an alternative anti–tumor necrosis factor drug is not necessarily precluded in a psoriasis patient with both skin and joint symptoms who has failed two previous anti-TNF drugs, according to Dr. Kenneth B. Gordon.
Dr. Gordon, professor of dermatology at Northwestern University, Chicago, described a case involving a 52-year-old man with a 20-year history of psoriasis who initially presented with mainly scalp and limited plaque psoriasis, and who was treated with topical corticosteroids and topical calcipotriene. After developing more extensive disease, he was treated successfully with ultraviolet B phototherapy.
Eleven years after first presenting with psoriasis, he presented with peripheral arthritis and enthesitis and was diagnosed with psoriatic arthritis.
The patient was treated initially with sulfasalazine, but failed to respond; methotrexate had only modest benefit, and at higher doses the patient developed liver function test abnormalities due to steatohepatitis, Dr. Gordon said at Perspectives in Rheumatic Diseases 2013.
Etanercept was initiated at 50 mg twice weekly, and the patient had an excellent initial response.
However, after stepping down to 50 mg weekly, his psoriasis flared.
"His joints were doing fine, but his skin was getting bad," Dr. Gordon said, noting that this type of response was also seen in early studies of etanercept, which showed that when doses were reduced to 50 mg weekly after 12 weeks, responses leveled off, with some patients achieving a 75% reduction in the Psoriasis Area and Severity Index (PASI) 75 response, and some losing PASI 75 response.
"It turns out that this occurs in 20%-30% of patients," he said, adding that some controversy exists regarding dosing, because "it’s not entirely clear that if you had just started at the lower dose and just let them go at the 50 mg once a week, that at 6 months they’d be any different than if you gave them the higher dose initially."
The REVEAL study of adalimumab for psoriasis also demonstrated loss of response after 33 weeks among those with an initial PASI 75 response (J. Am. Acad. Dermatol. 2012;66:241-51), he said.
"So clearly, patients lose response. Now, that lessens over time, but in the first year in psoriasis – and my guess is, in psoriatic arthritis as well – you have loss of effect at a relatively high level with all of these medicines," he said.
In fact, in the patient Dr. Gordon presented, a switch to adalimumab at a dose of 40 mg every other week resulted in an excellent initial response in both joints and skin, but skin symptoms returned after 8 months.
"This is something we’re all familiar with – patients doing well on an anti-TNF and then losing effect after a period of time on medication," he said.
So what are the alternatives?
An anti-TNF drug option after the patient cycles through etanercept, adalimumab, and infliximab is certolizumab, which was approved in September for the treatment of psoriatic arthritis. Study data suggest that the drug is "quite effective for psoriasis, was well tolerated, and similar to the other anti-TNFs," he said.
Using certolizumab is a reasonable approach that appears to work for both skin and joint symptoms, he said.
Golimumab is another possible option for treatment, but primary psoriasis data are lacking with this drug.
"It is my feeling that golimumab is probably not as potent for psoriasis as the other anti-TNF agents that we have, but that is an opinion," he said.
As for alternatives to anti-TNF agents, the PHEONIX 2 trial (Lancet 2008;371:1675-84) provided reasonable data in support of the interleukin-12 and -23 monoclonal antibody ustekinumab for skin disease, but patients who failed prior anti-TNF therapy actually had a poorer outcome, so that is something to keep in mind, he said, noting that adding methotrexate may help prevent a flare in patients who are switched from an anti-TNF agent to ustekinumab.
Ustekinumab, which also was approved in September for the treatment of psoriatic arthritis, is likely more useful for the skin, but is not exceptional for joint disease, he said at the meeting, held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Apremilast and tofacitinib, drugs now in development for psoriasis, may also prove useful in the future, as they may "theoretically be of benefit" based on currently available data, he said.
Dr. Gordon has received research support and/or honoraria from AbbVie (which markets adalimumab), Amgen (which markets etanercept), Celgene (which is developing apremilast), Eli Lilly, Janssen (which markets golimumab, infliximab, and ustekinumab), Novartis, and Pfizer (which markets tofacitinib).
LAS VEGAS – The use of an alternative anti–tumor necrosis factor drug is not necessarily precluded in a psoriasis patient with both skin and joint symptoms who has failed two previous anti-TNF drugs, according to Dr. Kenneth B. Gordon.
Dr. Gordon, professor of dermatology at Northwestern University, Chicago, described a case involving a 52-year-old man with a 20-year history of psoriasis who initially presented with mainly scalp and limited plaque psoriasis, and who was treated with topical corticosteroids and topical calcipotriene. After developing more extensive disease, he was treated successfully with ultraviolet B phototherapy.
Eleven years after first presenting with psoriasis, he presented with peripheral arthritis and enthesitis and was diagnosed with psoriatic arthritis.
The patient was treated initially with sulfasalazine, but failed to respond; methotrexate had only modest benefit, and at higher doses the patient developed liver function test abnormalities due to steatohepatitis, Dr. Gordon said at Perspectives in Rheumatic Diseases 2013.
Etanercept was initiated at 50 mg twice weekly, and the patient had an excellent initial response.
However, after stepping down to 50 mg weekly, his psoriasis flared.
"His joints were doing fine, but his skin was getting bad," Dr. Gordon said, noting that this type of response was also seen in early studies of etanercept, which showed that when doses were reduced to 50 mg weekly after 12 weeks, responses leveled off, with some patients achieving a 75% reduction in the Psoriasis Area and Severity Index (PASI) 75 response, and some losing PASI 75 response.
"It turns out that this occurs in 20%-30% of patients," he said, adding that some controversy exists regarding dosing, because "it’s not entirely clear that if you had just started at the lower dose and just let them go at the 50 mg once a week, that at 6 months they’d be any different than if you gave them the higher dose initially."
The REVEAL study of adalimumab for psoriasis also demonstrated loss of response after 33 weeks among those with an initial PASI 75 response (J. Am. Acad. Dermatol. 2012;66:241-51), he said.
"So clearly, patients lose response. Now, that lessens over time, but in the first year in psoriasis – and my guess is, in psoriatic arthritis as well – you have loss of effect at a relatively high level with all of these medicines," he said.
In fact, in the patient Dr. Gordon presented, a switch to adalimumab at a dose of 40 mg every other week resulted in an excellent initial response in both joints and skin, but skin symptoms returned after 8 months.
"This is something we’re all familiar with – patients doing well on an anti-TNF and then losing effect after a period of time on medication," he said.
So what are the alternatives?
An anti-TNF drug option after the patient cycles through etanercept, adalimumab, and infliximab is certolizumab, which was approved in September for the treatment of psoriatic arthritis. Study data suggest that the drug is "quite effective for psoriasis, was well tolerated, and similar to the other anti-TNFs," he said.
Using certolizumab is a reasonable approach that appears to work for both skin and joint symptoms, he said.
Golimumab is another possible option for treatment, but primary psoriasis data are lacking with this drug.
"It is my feeling that golimumab is probably not as potent for psoriasis as the other anti-TNF agents that we have, but that is an opinion," he said.
As for alternatives to anti-TNF agents, the PHEONIX 2 trial (Lancet 2008;371:1675-84) provided reasonable data in support of the interleukin-12 and -23 monoclonal antibody ustekinumab for skin disease, but patients who failed prior anti-TNF therapy actually had a poorer outcome, so that is something to keep in mind, he said, noting that adding methotrexate may help prevent a flare in patients who are switched from an anti-TNF agent to ustekinumab.
Ustekinumab, which also was approved in September for the treatment of psoriatic arthritis, is likely more useful for the skin, but is not exceptional for joint disease, he said at the meeting, held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Apremilast and tofacitinib, drugs now in development for psoriasis, may also prove useful in the future, as they may "theoretically be of benefit" based on currently available data, he said.
Dr. Gordon has received research support and/or honoraria from AbbVie (which markets adalimumab), Amgen (which markets etanercept), Celgene (which is developing apremilast), Eli Lilly, Janssen (which markets golimumab, infliximab, and ustekinumab), Novartis, and Pfizer (which markets tofacitinib).
EXPERT ANALYSIS FROM PERSPECTIVES IN RHEUMATIC DISEASES 2013
Treatment options expand for pulmonary arterial hypertension in scleroderma
LAS VEGAS – Scleroderma patients are at particularly high risk for developing pulmonary arterial hypertension, but treatment options are expanding, and with early referral to a PAH specialist, outcomes can be improved, according to Dr. Ronald J. Oudiz.
"In 1995 we had nothing. Now we have nine treatments specifically for PAH, and more that are being looked at," he said at Perspectives in Rheumatic Diseases 2013.
Three major signaling pathways, including the prostacyclin, endothelin, and nitric oxide pathways, form the basis for the available treatment options for this progressive and deadly disease, which has a 36-month survival rate of only about 60%. The treatments include prostacyclin analogues, endothelin receptor antagonists, and phosphodiesterase type 5 (PDE-5) inhibitors, said Dr. Oudiz, professor of medicine at the University of California, Los Angeles, and director of the Liu Center for Pulmonary Hypertension at Harbor-UCLA Medical Center.
The prostacyclin analogues include intravenous epoprostenol, inhaled iloprost, and treprostinil, which can be inhaled or delivered intravenously or subcutaneously. These have been shown to improve performance on the 6-minute walk test in a dose-dependent fashion, Dr. Oudiz noted.
Side effects with prostacyclin analogues can include flushing, headache, rash, thrombocytopenia, infection, and gastrointestinal effects such as diarrhea, nausea, and weight loss.
The endothelin receptor antagonists include bosentan and ambrisentan, which both are delivered orally. Their approval was "revolutionary, because for a while we only had IV drugs," he said.
Side effects with endothelin receptor antagonists include liver function test abnormalities, headache, nasal congestion, and edema. Although these drugs are generally tolerated well, liver function test abnormalities and edema can be troublesome for both patients and physicians as they require close monitoring and dose modification or interruption, he said.
PDE-5 inhibitors include sildenafil and tadalafil, which also are both oral drugs, and which have side effects that are similar to those seen with the endothelin-receptor antagonists, with the addition of myalgia, diarrhea, dyspepsia, and nose bleeds, he noted.
Overall, studies suggest that treatment with PAH drugs not only leads to improved exercise capacity, but probably improves long-term survival. In a meta-analysis of several major PAH drug trials, the majority of studies showed a potential mortality benefit (Eur. Heart. J. 2009;30:394-403), Dr. Oudiz noted.
"I say potential because we know none of the studies assessed in the meta-analysis were powered to examine mortality ... but nevertheless, we believe we’re doing more than just improving exercise capacity," he said.
In fact, in a study published in August in the New England Journal of Medicine, the new endothelin receptor antagonist macitentan was associated with a dose-dependent decrease in the number of outcome events measured, relative to placebo, including death, transplant, and heart failure in patients with PAH, he noted (N. Engl. J. Med. 2013;369:809-18).
"These are some really important endpoints we’re finally starting to meet," he said.
These advances underscore the importance of increased awareness of the risk of PAH in scleroderma and early recognition of the condition, and – since diagnosis and the subtleties of management are complex – they also underscore the importance of early referral to a PAH specialist, he said.
"Early treatment is always preferable to later treatment," he concluded at the meeting held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Dr. Oudiz has received grant or research support from, served as a consultant to, and/or served on a speakers’ bureau for Actelion, Bayer, Gilead, Ikaria, Lung, Pfizer, and United Therapeutics.
LAS VEGAS – Scleroderma patients are at particularly high risk for developing pulmonary arterial hypertension, but treatment options are expanding, and with early referral to a PAH specialist, outcomes can be improved, according to Dr. Ronald J. Oudiz.
"In 1995 we had nothing. Now we have nine treatments specifically for PAH, and more that are being looked at," he said at Perspectives in Rheumatic Diseases 2013.
Three major signaling pathways, including the prostacyclin, endothelin, and nitric oxide pathways, form the basis for the available treatment options for this progressive and deadly disease, which has a 36-month survival rate of only about 60%. The treatments include prostacyclin analogues, endothelin receptor antagonists, and phosphodiesterase type 5 (PDE-5) inhibitors, said Dr. Oudiz, professor of medicine at the University of California, Los Angeles, and director of the Liu Center for Pulmonary Hypertension at Harbor-UCLA Medical Center.
The prostacyclin analogues include intravenous epoprostenol, inhaled iloprost, and treprostinil, which can be inhaled or delivered intravenously or subcutaneously. These have been shown to improve performance on the 6-minute walk test in a dose-dependent fashion, Dr. Oudiz noted.
Side effects with prostacyclin analogues can include flushing, headache, rash, thrombocytopenia, infection, and gastrointestinal effects such as diarrhea, nausea, and weight loss.
The endothelin receptor antagonists include bosentan and ambrisentan, which both are delivered orally. Their approval was "revolutionary, because for a while we only had IV drugs," he said.
Side effects with endothelin receptor antagonists include liver function test abnormalities, headache, nasal congestion, and edema. Although these drugs are generally tolerated well, liver function test abnormalities and edema can be troublesome for both patients and physicians as they require close monitoring and dose modification or interruption, he said.
PDE-5 inhibitors include sildenafil and tadalafil, which also are both oral drugs, and which have side effects that are similar to those seen with the endothelin-receptor antagonists, with the addition of myalgia, diarrhea, dyspepsia, and nose bleeds, he noted.
Overall, studies suggest that treatment with PAH drugs not only leads to improved exercise capacity, but probably improves long-term survival. In a meta-analysis of several major PAH drug trials, the majority of studies showed a potential mortality benefit (Eur. Heart. J. 2009;30:394-403), Dr. Oudiz noted.
"I say potential because we know none of the studies assessed in the meta-analysis were powered to examine mortality ... but nevertheless, we believe we’re doing more than just improving exercise capacity," he said.
In fact, in a study published in August in the New England Journal of Medicine, the new endothelin receptor antagonist macitentan was associated with a dose-dependent decrease in the number of outcome events measured, relative to placebo, including death, transplant, and heart failure in patients with PAH, he noted (N. Engl. J. Med. 2013;369:809-18).
"These are some really important endpoints we’re finally starting to meet," he said.
These advances underscore the importance of increased awareness of the risk of PAH in scleroderma and early recognition of the condition, and – since diagnosis and the subtleties of management are complex – they also underscore the importance of early referral to a PAH specialist, he said.
"Early treatment is always preferable to later treatment," he concluded at the meeting held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Dr. Oudiz has received grant or research support from, served as a consultant to, and/or served on a speakers’ bureau for Actelion, Bayer, Gilead, Ikaria, Lung, Pfizer, and United Therapeutics.
LAS VEGAS – Scleroderma patients are at particularly high risk for developing pulmonary arterial hypertension, but treatment options are expanding, and with early referral to a PAH specialist, outcomes can be improved, according to Dr. Ronald J. Oudiz.
"In 1995 we had nothing. Now we have nine treatments specifically for PAH, and more that are being looked at," he said at Perspectives in Rheumatic Diseases 2013.
Three major signaling pathways, including the prostacyclin, endothelin, and nitric oxide pathways, form the basis for the available treatment options for this progressive and deadly disease, which has a 36-month survival rate of only about 60%. The treatments include prostacyclin analogues, endothelin receptor antagonists, and phosphodiesterase type 5 (PDE-5) inhibitors, said Dr. Oudiz, professor of medicine at the University of California, Los Angeles, and director of the Liu Center for Pulmonary Hypertension at Harbor-UCLA Medical Center.
The prostacyclin analogues include intravenous epoprostenol, inhaled iloprost, and treprostinil, which can be inhaled or delivered intravenously or subcutaneously. These have been shown to improve performance on the 6-minute walk test in a dose-dependent fashion, Dr. Oudiz noted.
Side effects with prostacyclin analogues can include flushing, headache, rash, thrombocytopenia, infection, and gastrointestinal effects such as diarrhea, nausea, and weight loss.
The endothelin receptor antagonists include bosentan and ambrisentan, which both are delivered orally. Their approval was "revolutionary, because for a while we only had IV drugs," he said.
Side effects with endothelin receptor antagonists include liver function test abnormalities, headache, nasal congestion, and edema. Although these drugs are generally tolerated well, liver function test abnormalities and edema can be troublesome for both patients and physicians as they require close monitoring and dose modification or interruption, he said.
PDE-5 inhibitors include sildenafil and tadalafil, which also are both oral drugs, and which have side effects that are similar to those seen with the endothelin-receptor antagonists, with the addition of myalgia, diarrhea, dyspepsia, and nose bleeds, he noted.
Overall, studies suggest that treatment with PAH drugs not only leads to improved exercise capacity, but probably improves long-term survival. In a meta-analysis of several major PAH drug trials, the majority of studies showed a potential mortality benefit (Eur. Heart. J. 2009;30:394-403), Dr. Oudiz noted.
"I say potential because we know none of the studies assessed in the meta-analysis were powered to examine mortality ... but nevertheless, we believe we’re doing more than just improving exercise capacity," he said.
In fact, in a study published in August in the New England Journal of Medicine, the new endothelin receptor antagonist macitentan was associated with a dose-dependent decrease in the number of outcome events measured, relative to placebo, including death, transplant, and heart failure in patients with PAH, he noted (N. Engl. J. Med. 2013;369:809-18).
"These are some really important endpoints we’re finally starting to meet," he said.
These advances underscore the importance of increased awareness of the risk of PAH in scleroderma and early recognition of the condition, and – since diagnosis and the subtleties of management are complex – they also underscore the importance of early referral to a PAH specialist, he said.
"Early treatment is always preferable to later treatment," he concluded at the meeting held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Dr. Oudiz has received grant or research support from, served as a consultant to, and/or served on a speakers’ bureau for Actelion, Bayer, Gilead, Ikaria, Lung, Pfizer, and United Therapeutics.
EXPERT ANALYSIS FROM PERSEPCTIVES IN RHEUMATIC DISEASES 2013
Fixed maintenance secukinumab beats as-needed dosing
ISTANBUL – Psoriasis patients who initially achieve a high-level response to secukinumab are significantly more likely to retain it over the long haul if they follow a fixed maintenance therapy schedule of once-monthly subcutaneous dosing rather than dosing as needed in the event of the start of relapse, according to the phase III SCULPTURE trial.
Secukinumab is an investigational fully human IgG1 monoclonal antibody targeting interleukin-17A, a key player in psoriasis, because it generates downstream proinflammatory cytokines and stimulates keratinocyte growth. Earlier studies indicated the investigational biologic had unprecedented clinical efficacy and rapidity of response. These observations gave rise to the hypothesis in SCULPTURE that maintenance dosing as needed upon relapse would prove noninferior to conventional fixed once-monthly maintenance therapy.
If the study hypothesis proved valid, it would mean less exposure to potential medication side effects, lower drug costs, and greater patient convenience. However, the hypothesis wasn’t borne out in the 966-patient, double-blind, randomized SCULPTURE (Study Comparing Retreatment Upon Start of Relapse), Dr. Ulrich Mrowietz reported at the annual congress of the European Academy of Dermatology and Venereology.
SCULPTURE participants had moderate-to-severe chronic plaque psoriasis despite prior systemic therapies, including biologic agents in many cases. They were randomized double blind to induction therapy involving five once-weekly subcutaneous injections of secukinumab at either 150 or 300 mg. At week 8, 843 participants with a Psoriasis Area and Severity Index (PASI) 75 response were re-randomized to maintenance therapy at the same dose, to be delivered either once monthly or as needed for relapse. The definition of relapse in this study required two elements: loss of PASI 75 response, and at least a 20% fall from the maximum PASI improvement, compared with baseline.
At week 52, 78.2% of patients randomized to 300 mg of secukinumab on a fixed once-monthly schedule still maintained a PASI 75 response. This was a significantly better outcome than the 67.7% PASI 75 rate in patients assigned to secukinumab 300 mg as needed, the 62.1% rate in those on secukinumab 150 mg once monthly, and the 52% PASI 75 rate in patients on secukinumab 150 mg as needed, reported Dr. Mrowietz of the University Medical Center Schleswig-Holstein, Kiel (Germany).
He found a bright spot in the negative results: While patients assigned to as-needed maintenance therapy achieved roughly an absolute 10% lower PASI 75 rate at 1 year, those in the secukinumab 150-mg group did so with only 46% the number of doses received by patients in the fixed monthly therapy group, while those in the retreatment-as-needed with secukinumab 300-mg group got only 39% of the number of doses, compared with patients on fixed monthly therapy.
"Fixed monthly dosing is the best maintenance regimen. But, in selected patients, there may be an opportunity with secukinumab to deviate from the usual fixed dosing regimen in favor of an as-needed approach," he said.
This tradeoff of an absolute 10% reduction in efficacy in return for a dosing regimen that entails less than half as much medication over the course of a year could prove of interest to payers, he noted.
The key, according to Dr. Mrowietz, will be to try to identify criteria helpful in selecting patients with an increased likelihood of a high-level response to the retreat-as-needed management strategy. The phase III trial was completed so recently that those necessary subanalyses have yet to be done.
However, Dr. Kristian Reich, another investigator involved in the secukinumab clinical trials program, drew a different message from SCULPTURE. He observed that these newer biologic agents are so effective that the bar has been raised with regard to patient expectations. Many patients won’t be satisfied with a PASI 75 response once a PASI 90 is achievable. And with fixed monthly maintenance secukinumab, it often is.
Indeed, the week-52 PASI 90 rate in SCULPTURE was a highly robust 59.7% in patients on fixed monthly secukinumab 300 mg and 45.8% for fixed monthly low-dose therapy, compared with the unimpressive 13.8% and 11.2% PASI 90 rates with high- and low-dose as-needed therapy.
"My take from this is that the best way to use this drug for continuous disease control is to give the drug every 4 weeks. What this study tells us is for those patients where you have to stop, where you have to use on-and-off therapy because they go away for 2 months to Africa, or they have a major operation, or for other reasons, these data are reassuring that we can use the drug safely on an intermittent basis," said Dr. Reich of Georg-August University in Göttingen, Germany.
Prior to SCULPTURE, he and other investigators were concerned that intermittent secukinumab therapy might promote the development of harmful antidrug antibodies. But antidrug antibodies arose in only three patients on fixed monthly maintenance therapy and two patients on as-needed therapy, and had no impact upon clinical efficacy or safety, he noted.
Another phase III trial presented in Istanbul was ERASURE (Efficacy and Safety of Subcutaneous Secukinumab for Moderate to Severe Chronic Plaque-Type Psoriasis for Up to 1 Year). Dr. Boni E. Elewski reported on 738 patients with moderate-to-severe chronic plaque psoriasis who were randomized double blind to secukinumab at 150 mg or 300 mg, or to placebo. Participants averaged a PASI score of 22 at baseline, along with 33% body surface area involvement. Psoriatic arthritis was present in 23% of subjects.
The co-primary endpoints in ERASURE were the week 12 PASI 75 response rates and Investigator’s Global Assessment scores of 0/1, indicative of clear or almost clear on a modified 5-point scale. The week-12 PASI 75 rates were 81.6% in the secukinumab 300 mg group, 71.6% with secukinumab 150 mg, and 4.5% with placebo. The week-12 IGA 0/1 rates were 65.3%, 51.2%, and 2.4%, said Dr. Elewski of the University of Alabama, Birmingham.
Among the other notable findings in ERASURE were the 69.8% PASI 90 and 41.6% PASI 100 responses at week 16 in the group assigned to secukinumab 300 mg, the fact that only one patient, on secukinumab 150 mg, developed transient antidrug antibodies, and the complete absence of cardiovascular events during 52 weeks on secukinumab, observed Dr. Elewski, professor of dermatology at the University of Alabama, Birmingham.
Dr. Mrowietz, Dr. Reich, and Dr. Elewski reported having received research grants from and serving as consultants to Novartis, which sponsored the secukinumab clinical trials program. The dermatologists serve in similar capacities with other pharmaceutical companies developing new medications for psoriasis.
ISTANBUL – Psoriasis patients who initially achieve a high-level response to secukinumab are significantly more likely to retain it over the long haul if they follow a fixed maintenance therapy schedule of once-monthly subcutaneous dosing rather than dosing as needed in the event of the start of relapse, according to the phase III SCULPTURE trial.
Secukinumab is an investigational fully human IgG1 monoclonal antibody targeting interleukin-17A, a key player in psoriasis, because it generates downstream proinflammatory cytokines and stimulates keratinocyte growth. Earlier studies indicated the investigational biologic had unprecedented clinical efficacy and rapidity of response. These observations gave rise to the hypothesis in SCULPTURE that maintenance dosing as needed upon relapse would prove noninferior to conventional fixed once-monthly maintenance therapy.
If the study hypothesis proved valid, it would mean less exposure to potential medication side effects, lower drug costs, and greater patient convenience. However, the hypothesis wasn’t borne out in the 966-patient, double-blind, randomized SCULPTURE (Study Comparing Retreatment Upon Start of Relapse), Dr. Ulrich Mrowietz reported at the annual congress of the European Academy of Dermatology and Venereology.
SCULPTURE participants had moderate-to-severe chronic plaque psoriasis despite prior systemic therapies, including biologic agents in many cases. They were randomized double blind to induction therapy involving five once-weekly subcutaneous injections of secukinumab at either 150 or 300 mg. At week 8, 843 participants with a Psoriasis Area and Severity Index (PASI) 75 response were re-randomized to maintenance therapy at the same dose, to be delivered either once monthly or as needed for relapse. The definition of relapse in this study required two elements: loss of PASI 75 response, and at least a 20% fall from the maximum PASI improvement, compared with baseline.
At week 52, 78.2% of patients randomized to 300 mg of secukinumab on a fixed once-monthly schedule still maintained a PASI 75 response. This was a significantly better outcome than the 67.7% PASI 75 rate in patients assigned to secukinumab 300 mg as needed, the 62.1% rate in those on secukinumab 150 mg once monthly, and the 52% PASI 75 rate in patients on secukinumab 150 mg as needed, reported Dr. Mrowietz of the University Medical Center Schleswig-Holstein, Kiel (Germany).
He found a bright spot in the negative results: While patients assigned to as-needed maintenance therapy achieved roughly an absolute 10% lower PASI 75 rate at 1 year, those in the secukinumab 150-mg group did so with only 46% the number of doses received by patients in the fixed monthly therapy group, while those in the retreatment-as-needed with secukinumab 300-mg group got only 39% of the number of doses, compared with patients on fixed monthly therapy.
"Fixed monthly dosing is the best maintenance regimen. But, in selected patients, there may be an opportunity with secukinumab to deviate from the usual fixed dosing regimen in favor of an as-needed approach," he said.
This tradeoff of an absolute 10% reduction in efficacy in return for a dosing regimen that entails less than half as much medication over the course of a year could prove of interest to payers, he noted.
The key, according to Dr. Mrowietz, will be to try to identify criteria helpful in selecting patients with an increased likelihood of a high-level response to the retreat-as-needed management strategy. The phase III trial was completed so recently that those necessary subanalyses have yet to be done.
However, Dr. Kristian Reich, another investigator involved in the secukinumab clinical trials program, drew a different message from SCULPTURE. He observed that these newer biologic agents are so effective that the bar has been raised with regard to patient expectations. Many patients won’t be satisfied with a PASI 75 response once a PASI 90 is achievable. And with fixed monthly maintenance secukinumab, it often is.
Indeed, the week-52 PASI 90 rate in SCULPTURE was a highly robust 59.7% in patients on fixed monthly secukinumab 300 mg and 45.8% for fixed monthly low-dose therapy, compared with the unimpressive 13.8% and 11.2% PASI 90 rates with high- and low-dose as-needed therapy.
"My take from this is that the best way to use this drug for continuous disease control is to give the drug every 4 weeks. What this study tells us is for those patients where you have to stop, where you have to use on-and-off therapy because they go away for 2 months to Africa, or they have a major operation, or for other reasons, these data are reassuring that we can use the drug safely on an intermittent basis," said Dr. Reich of Georg-August University in Göttingen, Germany.
Prior to SCULPTURE, he and other investigators were concerned that intermittent secukinumab therapy might promote the development of harmful antidrug antibodies. But antidrug antibodies arose in only three patients on fixed monthly maintenance therapy and two patients on as-needed therapy, and had no impact upon clinical efficacy or safety, he noted.
Another phase III trial presented in Istanbul was ERASURE (Efficacy and Safety of Subcutaneous Secukinumab for Moderate to Severe Chronic Plaque-Type Psoriasis for Up to 1 Year). Dr. Boni E. Elewski reported on 738 patients with moderate-to-severe chronic plaque psoriasis who were randomized double blind to secukinumab at 150 mg or 300 mg, or to placebo. Participants averaged a PASI score of 22 at baseline, along with 33% body surface area involvement. Psoriatic arthritis was present in 23% of subjects.
The co-primary endpoints in ERASURE were the week 12 PASI 75 response rates and Investigator’s Global Assessment scores of 0/1, indicative of clear or almost clear on a modified 5-point scale. The week-12 PASI 75 rates were 81.6% in the secukinumab 300 mg group, 71.6% with secukinumab 150 mg, and 4.5% with placebo. The week-12 IGA 0/1 rates were 65.3%, 51.2%, and 2.4%, said Dr. Elewski of the University of Alabama, Birmingham.
Among the other notable findings in ERASURE were the 69.8% PASI 90 and 41.6% PASI 100 responses at week 16 in the group assigned to secukinumab 300 mg, the fact that only one patient, on secukinumab 150 mg, developed transient antidrug antibodies, and the complete absence of cardiovascular events during 52 weeks on secukinumab, observed Dr. Elewski, professor of dermatology at the University of Alabama, Birmingham.
Dr. Mrowietz, Dr. Reich, and Dr. Elewski reported having received research grants from and serving as consultants to Novartis, which sponsored the secukinumab clinical trials program. The dermatologists serve in similar capacities with other pharmaceutical companies developing new medications for psoriasis.
ISTANBUL – Psoriasis patients who initially achieve a high-level response to secukinumab are significantly more likely to retain it over the long haul if they follow a fixed maintenance therapy schedule of once-monthly subcutaneous dosing rather than dosing as needed in the event of the start of relapse, according to the phase III SCULPTURE trial.
Secukinumab is an investigational fully human IgG1 monoclonal antibody targeting interleukin-17A, a key player in psoriasis, because it generates downstream proinflammatory cytokines and stimulates keratinocyte growth. Earlier studies indicated the investigational biologic had unprecedented clinical efficacy and rapidity of response. These observations gave rise to the hypothesis in SCULPTURE that maintenance dosing as needed upon relapse would prove noninferior to conventional fixed once-monthly maintenance therapy.
If the study hypothesis proved valid, it would mean less exposure to potential medication side effects, lower drug costs, and greater patient convenience. However, the hypothesis wasn’t borne out in the 966-patient, double-blind, randomized SCULPTURE (Study Comparing Retreatment Upon Start of Relapse), Dr. Ulrich Mrowietz reported at the annual congress of the European Academy of Dermatology and Venereology.
SCULPTURE participants had moderate-to-severe chronic plaque psoriasis despite prior systemic therapies, including biologic agents in many cases. They were randomized double blind to induction therapy involving five once-weekly subcutaneous injections of secukinumab at either 150 or 300 mg. At week 8, 843 participants with a Psoriasis Area and Severity Index (PASI) 75 response were re-randomized to maintenance therapy at the same dose, to be delivered either once monthly or as needed for relapse. The definition of relapse in this study required two elements: loss of PASI 75 response, and at least a 20% fall from the maximum PASI improvement, compared with baseline.
At week 52, 78.2% of patients randomized to 300 mg of secukinumab on a fixed once-monthly schedule still maintained a PASI 75 response. This was a significantly better outcome than the 67.7% PASI 75 rate in patients assigned to secukinumab 300 mg as needed, the 62.1% rate in those on secukinumab 150 mg once monthly, and the 52% PASI 75 rate in patients on secukinumab 150 mg as needed, reported Dr. Mrowietz of the University Medical Center Schleswig-Holstein, Kiel (Germany).
He found a bright spot in the negative results: While patients assigned to as-needed maintenance therapy achieved roughly an absolute 10% lower PASI 75 rate at 1 year, those in the secukinumab 150-mg group did so with only 46% the number of doses received by patients in the fixed monthly therapy group, while those in the retreatment-as-needed with secukinumab 300-mg group got only 39% of the number of doses, compared with patients on fixed monthly therapy.
"Fixed monthly dosing is the best maintenance regimen. But, in selected patients, there may be an opportunity with secukinumab to deviate from the usual fixed dosing regimen in favor of an as-needed approach," he said.
This tradeoff of an absolute 10% reduction in efficacy in return for a dosing regimen that entails less than half as much medication over the course of a year could prove of interest to payers, he noted.
The key, according to Dr. Mrowietz, will be to try to identify criteria helpful in selecting patients with an increased likelihood of a high-level response to the retreat-as-needed management strategy. The phase III trial was completed so recently that those necessary subanalyses have yet to be done.
However, Dr. Kristian Reich, another investigator involved in the secukinumab clinical trials program, drew a different message from SCULPTURE. He observed that these newer biologic agents are so effective that the bar has been raised with regard to patient expectations. Many patients won’t be satisfied with a PASI 75 response once a PASI 90 is achievable. And with fixed monthly maintenance secukinumab, it often is.
Indeed, the week-52 PASI 90 rate in SCULPTURE was a highly robust 59.7% in patients on fixed monthly secukinumab 300 mg and 45.8% for fixed monthly low-dose therapy, compared with the unimpressive 13.8% and 11.2% PASI 90 rates with high- and low-dose as-needed therapy.
"My take from this is that the best way to use this drug for continuous disease control is to give the drug every 4 weeks. What this study tells us is for those patients where you have to stop, where you have to use on-and-off therapy because they go away for 2 months to Africa, or they have a major operation, or for other reasons, these data are reassuring that we can use the drug safely on an intermittent basis," said Dr. Reich of Georg-August University in Göttingen, Germany.
Prior to SCULPTURE, he and other investigators were concerned that intermittent secukinumab therapy might promote the development of harmful antidrug antibodies. But antidrug antibodies arose in only three patients on fixed monthly maintenance therapy and two patients on as-needed therapy, and had no impact upon clinical efficacy or safety, he noted.
Another phase III trial presented in Istanbul was ERASURE (Efficacy and Safety of Subcutaneous Secukinumab for Moderate to Severe Chronic Plaque-Type Psoriasis for Up to 1 Year). Dr. Boni E. Elewski reported on 738 patients with moderate-to-severe chronic plaque psoriasis who were randomized double blind to secukinumab at 150 mg or 300 mg, or to placebo. Participants averaged a PASI score of 22 at baseline, along with 33% body surface area involvement. Psoriatic arthritis was present in 23% of subjects.
The co-primary endpoints in ERASURE were the week 12 PASI 75 response rates and Investigator’s Global Assessment scores of 0/1, indicative of clear or almost clear on a modified 5-point scale. The week-12 PASI 75 rates were 81.6% in the secukinumab 300 mg group, 71.6% with secukinumab 150 mg, and 4.5% with placebo. The week-12 IGA 0/1 rates were 65.3%, 51.2%, and 2.4%, said Dr. Elewski of the University of Alabama, Birmingham.
Among the other notable findings in ERASURE were the 69.8% PASI 90 and 41.6% PASI 100 responses at week 16 in the group assigned to secukinumab 300 mg, the fact that only one patient, on secukinumab 150 mg, developed transient antidrug antibodies, and the complete absence of cardiovascular events during 52 weeks on secukinumab, observed Dr. Elewski, professor of dermatology at the University of Alabama, Birmingham.
Dr. Mrowietz, Dr. Reich, and Dr. Elewski reported having received research grants from and serving as consultants to Novartis, which sponsored the secukinumab clinical trials program. The dermatologists serve in similar capacities with other pharmaceutical companies developing new medications for psoriasis.
AT THE EADV CONGRESS
Major finding: Among psoriasis patients who achieved a PASI 75 response to 5 weeks of induction therapy with subcutaneous secukinumab, 59.7% who were placed on a maintenance regimen of 300 mg every 4 weeks had a PASI 90 response at week 52, compared with 13.8% whose maintenance program involved taking 300 mg on an as-needed basis at the start of relapse.
Data source: The SCULPTURE study was a prospective, double-blind, 52-week, phase III clinical trial including 843 patients.
Disclosures: The study was sponsored by Novartis. Speakers reported having received research grants from and serving as consultants to the company.