User login
Novel predictor of poor outcomes in lupus pregnancies
SAN DIEGO – Alteration in the balance of placentally secreted angiogenic factors early in pregnancy provides a potent new predictor of subsequent preeclampsia and other poor outcomes in pregnant women with systemic lupus erythematosus and/or antiphospholipid antibody syndrome.
Patients with systemic lupus erythematosus (SLE) and/or antiphospholipid antibody syndrome (APS) who had an elevated ratio of a splice variant of vascular endothelial growth factor R1 called sFLT1 to placental growth factor (PlGF) when measured at 16-19 weeks’ gestation were at 13.8-fold increased relative risk of preeclampsia before 34 weeks, compared with patients with an sFLT1/PlGF ratio below that cut-point in the large, multicenter, observational PROMISSE (Predictors of Pregnancy Outcome: Biomarkers in Antiphospholipid Antibody Syndrome and SLE) study, Dr. Jane E. Salmon reported at the annual meeting of the American College of Rheumatology.
"Nearly half of the patients with an SFLT1/PlGF ratio greater than 3.45 when measured at 16-19 weeks’ gestation will develop early preeclampsia. On the other hand, a low ratio, as well as low levels of sFLT1 or high levels of PlGF, can reassure physicians and patients that preterm preeclampsia is unlikely: a 3% chance," said Dr. Salmon, professor of medicine and of ob.gyn. at Cornell University and a rheumatologist at the Hospital for Special Surgery, both in New York.
Pregnancy in patients with lupus is associated with obstetric complications placing both mother and fetus at great risk. Yet, until now it hasn’t been possible to predict which patients will have poor outcomes.
The key to identifying those at high risk lies in a recognition that preeclampsia and other poor outcomes are dramatic manifestations of placental insufficiency, which actually begins, initially silently, early in pregnancy. The maternal hypertension, proteinuria, thrombocytopenia, and other end-organ manifestations of preeclampsia are caused by maternal endothelial dysfunction mediated by placental secretion of antiangiogenic factors. Angiogenic growth factors, such as PIGF and vascular endothelial growth factor (VEGF), are essential to a healthy endothelium. But placentally secreted sFLT1 binds to these two angiogenic growth factors, rendering them unavailable to the endothelium, she explained.
Overexpression of sFLT1 in multiple animal models results in hypertension and proteinuria, the hallmarks of preeclampsia. Moreover, cancer patients treated with VEGF inhibitors often develop these two conditions. Based in part on these observations, Dr. Salmon and her coinvestigators turned to the PROMISSE study to test their hypothesis that elevated levels of antiangiogenic factors early in pregnancy predict poor outcomes in patients with SLE and/or APS. The prospective study involved 503 pregnant women with SLE and/or APS and 204 healthy controls, all with monthly blood draws starting before 12 weeks’ gestation.
The composite outcome of preeclampsia, small for gestational age, indicated preterm delivery, and other adverse events occurred in 37% of SLE patients who also had APS. The rate was 16% in patients with SLE alone, 26% in those with APS alone, and 3% in controls.
Subjects with SLE and/or APS who developed preeclampsia and other pregnancy complications displayed significantly higher levels of sFLT1 beginning at 12 weeks and sustained through 31 weeks’ gestation, compared with those with normal pregnancies. Moreover, PlGF levels were significantly lower during weeks 16-31 in the patients with pregnancy complications. The investigators determined that the best predictor of pregnancy complications was the ratio of antiangiogenic sFLT1 to angiogenic PlGF. And the optimal cut-point was 3.45.
Audience members said that while a predictive test for preeclampsia is most welcome, the fact remains that physicians don’t have a lot to offer in terms of prevention or treatment of this feared pregnancy complication. Dr. Salmon responded that the SFLT1/PlGF ratio can be used to risk-stratify pregnant lupus patients for future interventional trials with new drugs looking at new pathways. Already, for example, other investigators have reported some success using a strategy targeting sFLT1 itself. In a small study, they found that women with severe preeclampsia who had their blood run through a heparin column that binds and removes sFLT1 were able to maintain their pregnancies for up to 2 weeks.
"It’s a tiny, open-label trial involving a device, but I think that will move forward," she predicted.
The PROMISSE study was funded by the National Institutes of Health, the Alliance for Lupus Research, and the Mary Kirkland Center for Lupus Research at the Hospital for Special Surgery. Dr. Salmon reported having received research grants from and/or serving as a consultant to Alexion, Novartis, and Roche.
SLE, APS, vascular endothelial growth factor R1, sFLT1, placental growth factor, PlGF, preeclampsia, PROMISSE study, Predictors of Pregnancy Outcome: Biomarkers in Antiphospholipid Antibody Syndrome and SLE study, Dr. Jane E. Salmon, annual meeting of the American College of Rheumatology, early preeclampsia,
SAN DIEGO – Alteration in the balance of placentally secreted angiogenic factors early in pregnancy provides a potent new predictor of subsequent preeclampsia and other poor outcomes in pregnant women with systemic lupus erythematosus and/or antiphospholipid antibody syndrome.
Patients with systemic lupus erythematosus (SLE) and/or antiphospholipid antibody syndrome (APS) who had an elevated ratio of a splice variant of vascular endothelial growth factor R1 called sFLT1 to placental growth factor (PlGF) when measured at 16-19 weeks’ gestation were at 13.8-fold increased relative risk of preeclampsia before 34 weeks, compared with patients with an sFLT1/PlGF ratio below that cut-point in the large, multicenter, observational PROMISSE (Predictors of Pregnancy Outcome: Biomarkers in Antiphospholipid Antibody Syndrome and SLE) study, Dr. Jane E. Salmon reported at the annual meeting of the American College of Rheumatology.
"Nearly half of the patients with an SFLT1/PlGF ratio greater than 3.45 when measured at 16-19 weeks’ gestation will develop early preeclampsia. On the other hand, a low ratio, as well as low levels of sFLT1 or high levels of PlGF, can reassure physicians and patients that preterm preeclampsia is unlikely: a 3% chance," said Dr. Salmon, professor of medicine and of ob.gyn. at Cornell University and a rheumatologist at the Hospital for Special Surgery, both in New York.
Pregnancy in patients with lupus is associated with obstetric complications placing both mother and fetus at great risk. Yet, until now it hasn’t been possible to predict which patients will have poor outcomes.
The key to identifying those at high risk lies in a recognition that preeclampsia and other poor outcomes are dramatic manifestations of placental insufficiency, which actually begins, initially silently, early in pregnancy. The maternal hypertension, proteinuria, thrombocytopenia, and other end-organ manifestations of preeclampsia are caused by maternal endothelial dysfunction mediated by placental secretion of antiangiogenic factors. Angiogenic growth factors, such as PIGF and vascular endothelial growth factor (VEGF), are essential to a healthy endothelium. But placentally secreted sFLT1 binds to these two angiogenic growth factors, rendering them unavailable to the endothelium, she explained.
Overexpression of sFLT1 in multiple animal models results in hypertension and proteinuria, the hallmarks of preeclampsia. Moreover, cancer patients treated with VEGF inhibitors often develop these two conditions. Based in part on these observations, Dr. Salmon and her coinvestigators turned to the PROMISSE study to test their hypothesis that elevated levels of antiangiogenic factors early in pregnancy predict poor outcomes in patients with SLE and/or APS. The prospective study involved 503 pregnant women with SLE and/or APS and 204 healthy controls, all with monthly blood draws starting before 12 weeks’ gestation.
The composite outcome of preeclampsia, small for gestational age, indicated preterm delivery, and other adverse events occurred in 37% of SLE patients who also had APS. The rate was 16% in patients with SLE alone, 26% in those with APS alone, and 3% in controls.
Subjects with SLE and/or APS who developed preeclampsia and other pregnancy complications displayed significantly higher levels of sFLT1 beginning at 12 weeks and sustained through 31 weeks’ gestation, compared with those with normal pregnancies. Moreover, PlGF levels were significantly lower during weeks 16-31 in the patients with pregnancy complications. The investigators determined that the best predictor of pregnancy complications was the ratio of antiangiogenic sFLT1 to angiogenic PlGF. And the optimal cut-point was 3.45.
Audience members said that while a predictive test for preeclampsia is most welcome, the fact remains that physicians don’t have a lot to offer in terms of prevention or treatment of this feared pregnancy complication. Dr. Salmon responded that the SFLT1/PlGF ratio can be used to risk-stratify pregnant lupus patients for future interventional trials with new drugs looking at new pathways. Already, for example, other investigators have reported some success using a strategy targeting sFLT1 itself. In a small study, they found that women with severe preeclampsia who had their blood run through a heparin column that binds and removes sFLT1 were able to maintain their pregnancies for up to 2 weeks.
"It’s a tiny, open-label trial involving a device, but I think that will move forward," she predicted.
The PROMISSE study was funded by the National Institutes of Health, the Alliance for Lupus Research, and the Mary Kirkland Center for Lupus Research at the Hospital for Special Surgery. Dr. Salmon reported having received research grants from and/or serving as a consultant to Alexion, Novartis, and Roche.
SAN DIEGO – Alteration in the balance of placentally secreted angiogenic factors early in pregnancy provides a potent new predictor of subsequent preeclampsia and other poor outcomes in pregnant women with systemic lupus erythematosus and/or antiphospholipid antibody syndrome.
Patients with systemic lupus erythematosus (SLE) and/or antiphospholipid antibody syndrome (APS) who had an elevated ratio of a splice variant of vascular endothelial growth factor R1 called sFLT1 to placental growth factor (PlGF) when measured at 16-19 weeks’ gestation were at 13.8-fold increased relative risk of preeclampsia before 34 weeks, compared with patients with an sFLT1/PlGF ratio below that cut-point in the large, multicenter, observational PROMISSE (Predictors of Pregnancy Outcome: Biomarkers in Antiphospholipid Antibody Syndrome and SLE) study, Dr. Jane E. Salmon reported at the annual meeting of the American College of Rheumatology.
"Nearly half of the patients with an SFLT1/PlGF ratio greater than 3.45 when measured at 16-19 weeks’ gestation will develop early preeclampsia. On the other hand, a low ratio, as well as low levels of sFLT1 or high levels of PlGF, can reassure physicians and patients that preterm preeclampsia is unlikely: a 3% chance," said Dr. Salmon, professor of medicine and of ob.gyn. at Cornell University and a rheumatologist at the Hospital for Special Surgery, both in New York.
Pregnancy in patients with lupus is associated with obstetric complications placing both mother and fetus at great risk. Yet, until now it hasn’t been possible to predict which patients will have poor outcomes.
The key to identifying those at high risk lies in a recognition that preeclampsia and other poor outcomes are dramatic manifestations of placental insufficiency, which actually begins, initially silently, early in pregnancy. The maternal hypertension, proteinuria, thrombocytopenia, and other end-organ manifestations of preeclampsia are caused by maternal endothelial dysfunction mediated by placental secretion of antiangiogenic factors. Angiogenic growth factors, such as PIGF and vascular endothelial growth factor (VEGF), are essential to a healthy endothelium. But placentally secreted sFLT1 binds to these two angiogenic growth factors, rendering them unavailable to the endothelium, she explained.
Overexpression of sFLT1 in multiple animal models results in hypertension and proteinuria, the hallmarks of preeclampsia. Moreover, cancer patients treated with VEGF inhibitors often develop these two conditions. Based in part on these observations, Dr. Salmon and her coinvestigators turned to the PROMISSE study to test their hypothesis that elevated levels of antiangiogenic factors early in pregnancy predict poor outcomes in patients with SLE and/or APS. The prospective study involved 503 pregnant women with SLE and/or APS and 204 healthy controls, all with monthly blood draws starting before 12 weeks’ gestation.
The composite outcome of preeclampsia, small for gestational age, indicated preterm delivery, and other adverse events occurred in 37% of SLE patients who also had APS. The rate was 16% in patients with SLE alone, 26% in those with APS alone, and 3% in controls.
Subjects with SLE and/or APS who developed preeclampsia and other pregnancy complications displayed significantly higher levels of sFLT1 beginning at 12 weeks and sustained through 31 weeks’ gestation, compared with those with normal pregnancies. Moreover, PlGF levels were significantly lower during weeks 16-31 in the patients with pregnancy complications. The investigators determined that the best predictor of pregnancy complications was the ratio of antiangiogenic sFLT1 to angiogenic PlGF. And the optimal cut-point was 3.45.
Audience members said that while a predictive test for preeclampsia is most welcome, the fact remains that physicians don’t have a lot to offer in terms of prevention or treatment of this feared pregnancy complication. Dr. Salmon responded that the SFLT1/PlGF ratio can be used to risk-stratify pregnant lupus patients for future interventional trials with new drugs looking at new pathways. Already, for example, other investigators have reported some success using a strategy targeting sFLT1 itself. In a small study, they found that women with severe preeclampsia who had their blood run through a heparin column that binds and removes sFLT1 were able to maintain their pregnancies for up to 2 weeks.
"It’s a tiny, open-label trial involving a device, but I think that will move forward," she predicted.
The PROMISSE study was funded by the National Institutes of Health, the Alliance for Lupus Research, and the Mary Kirkland Center for Lupus Research at the Hospital for Special Surgery. Dr. Salmon reported having received research grants from and/or serving as a consultant to Alexion, Novartis, and Roche.
SLE, APS, vascular endothelial growth factor R1, sFLT1, placental growth factor, PlGF, preeclampsia, PROMISSE study, Predictors of Pregnancy Outcome: Biomarkers in Antiphospholipid Antibody Syndrome and SLE study, Dr. Jane E. Salmon, annual meeting of the American College of Rheumatology, early preeclampsia,
SLE, APS, vascular endothelial growth factor R1, sFLT1, placental growth factor, PlGF, preeclampsia, PROMISSE study, Predictors of Pregnancy Outcome: Biomarkers in Antiphospholipid Antibody Syndrome and SLE study, Dr. Jane E. Salmon, annual meeting of the American College of Rheumatology, early preeclampsia,
AT THE ACR ANNUAL MEETING
Major finding: Pregnant patients with SLE and/or APS had nearly a 14-fold increased risk of developing preeclampsia before 34 weeks’ gestation when they displayed an abnormal ratio of two key placentally derived angiogenic factors at 16-19 weeks’ gestation.
Data source: This analysis included 503 pregnant women with SLE and/or APS and 204 pregnant controls participating in the prospective, observational, multicenter PROMISSE study.
Disclosures: The study was funded by the National Institutes of Health, the Alliance for Lupus Research, and the Mary Kirkland Center for Lupus Research at the Hospital for Special Surgery. The presenter reported no relevant financial interests.
Impact of psoriasis on sexual activity
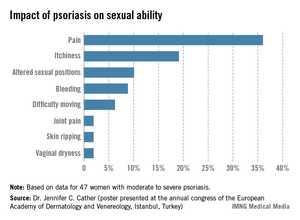
One third of a group of women with psoriasis reported that the pain associated with their condition interfered with their sexual activity, according to findings from a survey presented by Dr. Jennifer C. Cather.
Based on responses from a survey of 60 women with moderate to severe psoriasis, the specific complaints that were the most common ways in which psoriasis interfered with sexual activity were itchiness (19%), the need to adjust sexual position (10%), and bleeding (9%), Dr. Cather reported at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar. The survey was part of an effort to determine the impact of psoriasis on women’s sexual activity, desires, and relationships.
The data were previously presented in a poster at the annual congress of the European Academy of Dermatology and Venereology (Istanbul.
SDEF and this news organization are owned by Frontline Medical Communications. Dr. Cather disclosed that she is a consultant, speaker, or researcher for AbbVie, Novartis, Leo, Janssen, Amgen, Celgene, Merck, and Pfizer.
One third of a group of women with psoriasis reported that the pain associated with their condition interfered with their sexual activity, according to findings from a survey presented by Dr. Jennifer C. Cather.
Based on responses from a survey of 60 women with moderate to severe psoriasis, the specific complaints that were the most common ways in which psoriasis interfered with sexual activity were itchiness (19%), the need to adjust sexual position (10%), and bleeding (9%), Dr. Cather reported at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar. The survey was part of an effort to determine the impact of psoriasis on women’s sexual activity, desires, and relationships.
The data were previously presented in a poster at the annual congress of the European Academy of Dermatology and Venereology (Istanbul.
SDEF and this news organization are owned by Frontline Medical Communications. Dr. Cather disclosed that she is a consultant, speaker, or researcher for AbbVie, Novartis, Leo, Janssen, Amgen, Celgene, Merck, and Pfizer.

One third of a group of women with psoriasis reported that the pain associated with their condition interfered with their sexual activity, according to findings from a survey presented by Dr. Jennifer C. Cather.
Based on responses from a survey of 60 women with moderate to severe psoriasis, the specific complaints that were the most common ways in which psoriasis interfered with sexual activity were itchiness (19%), the need to adjust sexual position (10%), and bleeding (9%), Dr. Cather reported at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar. The survey was part of an effort to determine the impact of psoriasis on women’s sexual activity, desires, and relationships.
The data were previously presented in a poster at the annual congress of the European Academy of Dermatology and Venereology (Istanbul.
SDEF and this news organization are owned by Frontline Medical Communications. Dr. Cather disclosed that she is a consultant, speaker, or researcher for AbbVie, Novartis, Leo, Janssen, Amgen, Celgene, Merck, and Pfizer.

EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
Treat-to-target approach for psoriatic arthritis found beneficial
SAN DIEGO – Compared with standard care, intensive management of psoriatic arthritis using a treat-to-target approach significantly improved joint and skin outcomes for patients newly diagnosed with the disease, results from a multicenter, randomized controlled trial showed.
"Treating to target works in this disease, and it’s going to result in better long-term outcomes," lead investigator Dr. Philip Helliwell said during a press briefing at the annual meeting of the American College of Rheumatology.
Dr. Helliwell and his associates at eight centers in the United Kingdom randomized 206 patients with early psoriatic arthritis to standard care or intensive management, and followed them for 48 weeks. Patients in the standard care group were treated by a rheumatologist with no set protocol and no limitations, while those in the intensive management group followed a strict treatment protocol with escalation of therapy if minimal disease activity criteria were not met.
Patients in the intensive management group were started on methotrexate with rapid escalation to a dose of *25 mg/week after 6 weeks if they tolerated the drug. If they did not meet minimal disease criteria after 12 weeks, they received a more powerful combination of disease-modifying antirheumatic drugs (DMARDs).
After another 12 weeks, patients in the intensive management group were given anti–tumor necrosis factor therapy if they had three or more tender joints. If they had fewer than three tender or swollen joints but did not meet the minimal disease activity criteria, they were given methotrexate and an alternative DMARD. Patients in the standard care group were treated with DMARDs, but with no set time limits for drug therapy escalation or measurements to reach.
The primary outcome measures were the proportion of patients in both groups who achieved ACR20, ACR50, and ACR70 criteria for disease activity, which represent disease improvement of 20%, 50%, and 70%, respectively. Dr. Helliwell reported that compared with the standard care group, a higher proportion of patients in the intensive management group achieved ACR20 (62% vs. 45%, respectively), ACR50 (51% vs. 25%), and ACR70 (38% vs. 17%). A higher proportion of patients in the intensive management group also achieved a Psoriasis Area and Severity Index 75 compared with their counterparts in the standard care group (59% vs. 33%).
Research in psoriatic arthritis has "lagged behind that of rheumatoid arthritis for years in terms of pathogenesis and treatment paradigms," noted Dr. Helliwell, a senior lecturer in rheumatology at the Institute of Rheumatic and Musculoskeletal Medicine at the University of Leeds (England). "I think the study we’ve reported brings psoriatic arthritis right up to date alongside RA."
He said that up to one-third of people with psoriasis will develop psoriatic arthritis, "so it’s not an insignificant arthritis. It’s often not recognized. One survey we did of people with psoriasis in the community in the U.K. found that up to half of the people with psoriatic arthritis didn’t know they had it. They’d seen their doctor for various reasons and had been told they’d had another form of arthritis, or they were fobbed off with other diagnoses."
The researchers have yet to perform a cost analysis comparing the two treatment groups.
The study was funded by Arthritis Research UK and Pfizer. Dr. Helliwell disclosed that he has received consulting fees from Pfizer.
*Correction 11/11/13: A previous version of this story misstated the methotrexate dosage used in the study. This version has been updated to reflect the correct dosage.
SAN DIEGO – Compared with standard care, intensive management of psoriatic arthritis using a treat-to-target approach significantly improved joint and skin outcomes for patients newly diagnosed with the disease, results from a multicenter, randomized controlled trial showed.
"Treating to target works in this disease, and it’s going to result in better long-term outcomes," lead investigator Dr. Philip Helliwell said during a press briefing at the annual meeting of the American College of Rheumatology.
Dr. Helliwell and his associates at eight centers in the United Kingdom randomized 206 patients with early psoriatic arthritis to standard care or intensive management, and followed them for 48 weeks. Patients in the standard care group were treated by a rheumatologist with no set protocol and no limitations, while those in the intensive management group followed a strict treatment protocol with escalation of therapy if minimal disease activity criteria were not met.
Patients in the intensive management group were started on methotrexate with rapid escalation to a dose of *25 mg/week after 6 weeks if they tolerated the drug. If they did not meet minimal disease criteria after 12 weeks, they received a more powerful combination of disease-modifying antirheumatic drugs (DMARDs).
After another 12 weeks, patients in the intensive management group were given anti–tumor necrosis factor therapy if they had three or more tender joints. If they had fewer than three tender or swollen joints but did not meet the minimal disease activity criteria, they were given methotrexate and an alternative DMARD. Patients in the standard care group were treated with DMARDs, but with no set time limits for drug therapy escalation or measurements to reach.
The primary outcome measures were the proportion of patients in both groups who achieved ACR20, ACR50, and ACR70 criteria for disease activity, which represent disease improvement of 20%, 50%, and 70%, respectively. Dr. Helliwell reported that compared with the standard care group, a higher proportion of patients in the intensive management group achieved ACR20 (62% vs. 45%, respectively), ACR50 (51% vs. 25%), and ACR70 (38% vs. 17%). A higher proportion of patients in the intensive management group also achieved a Psoriasis Area and Severity Index 75 compared with their counterparts in the standard care group (59% vs. 33%).
Research in psoriatic arthritis has "lagged behind that of rheumatoid arthritis for years in terms of pathogenesis and treatment paradigms," noted Dr. Helliwell, a senior lecturer in rheumatology at the Institute of Rheumatic and Musculoskeletal Medicine at the University of Leeds (England). "I think the study we’ve reported brings psoriatic arthritis right up to date alongside RA."
He said that up to one-third of people with psoriasis will develop psoriatic arthritis, "so it’s not an insignificant arthritis. It’s often not recognized. One survey we did of people with psoriasis in the community in the U.K. found that up to half of the people with psoriatic arthritis didn’t know they had it. They’d seen their doctor for various reasons and had been told they’d had another form of arthritis, or they were fobbed off with other diagnoses."
The researchers have yet to perform a cost analysis comparing the two treatment groups.
The study was funded by Arthritis Research UK and Pfizer. Dr. Helliwell disclosed that he has received consulting fees from Pfizer.
*Correction 11/11/13: A previous version of this story misstated the methotrexate dosage used in the study. This version has been updated to reflect the correct dosage.
SAN DIEGO – Compared with standard care, intensive management of psoriatic arthritis using a treat-to-target approach significantly improved joint and skin outcomes for patients newly diagnosed with the disease, results from a multicenter, randomized controlled trial showed.
"Treating to target works in this disease, and it’s going to result in better long-term outcomes," lead investigator Dr. Philip Helliwell said during a press briefing at the annual meeting of the American College of Rheumatology.
Dr. Helliwell and his associates at eight centers in the United Kingdom randomized 206 patients with early psoriatic arthritis to standard care or intensive management, and followed them for 48 weeks. Patients in the standard care group were treated by a rheumatologist with no set protocol and no limitations, while those in the intensive management group followed a strict treatment protocol with escalation of therapy if minimal disease activity criteria were not met.
Patients in the intensive management group were started on methotrexate with rapid escalation to a dose of *25 mg/week after 6 weeks if they tolerated the drug. If they did not meet minimal disease criteria after 12 weeks, they received a more powerful combination of disease-modifying antirheumatic drugs (DMARDs).
After another 12 weeks, patients in the intensive management group were given anti–tumor necrosis factor therapy if they had three or more tender joints. If they had fewer than three tender or swollen joints but did not meet the minimal disease activity criteria, they were given methotrexate and an alternative DMARD. Patients in the standard care group were treated with DMARDs, but with no set time limits for drug therapy escalation or measurements to reach.
The primary outcome measures were the proportion of patients in both groups who achieved ACR20, ACR50, and ACR70 criteria for disease activity, which represent disease improvement of 20%, 50%, and 70%, respectively. Dr. Helliwell reported that compared with the standard care group, a higher proportion of patients in the intensive management group achieved ACR20 (62% vs. 45%, respectively), ACR50 (51% vs. 25%), and ACR70 (38% vs. 17%). A higher proportion of patients in the intensive management group also achieved a Psoriasis Area and Severity Index 75 compared with their counterparts in the standard care group (59% vs. 33%).
Research in psoriatic arthritis has "lagged behind that of rheumatoid arthritis for years in terms of pathogenesis and treatment paradigms," noted Dr. Helliwell, a senior lecturer in rheumatology at the Institute of Rheumatic and Musculoskeletal Medicine at the University of Leeds (England). "I think the study we’ve reported brings psoriatic arthritis right up to date alongside RA."
He said that up to one-third of people with psoriasis will develop psoriatic arthritis, "so it’s not an insignificant arthritis. It’s often not recognized. One survey we did of people with psoriasis in the community in the U.K. found that up to half of the people with psoriatic arthritis didn’t know they had it. They’d seen their doctor for various reasons and had been told they’d had another form of arthritis, or they were fobbed off with other diagnoses."
The researchers have yet to perform a cost analysis comparing the two treatment groups.
The study was funded by Arthritis Research UK and Pfizer. Dr. Helliwell disclosed that he has received consulting fees from Pfizer.
*Correction 11/11/13: A previous version of this story misstated the methotrexate dosage used in the study. This version has been updated to reflect the correct dosage.
AT THE ACR ANNUAL MEETING
Major finding: At 48 weeks, a higher proportion of psoriatic arthritis patients in the intensive management group achieved ACR70, compared with those in the standard care group (38% vs. 17%, respectively).
Data source: A multicenter trial of 206 patients with early psoriatic arthritis who were randomized to standard care or intensive management and followed for 48 weeks.
Disclosures: The study was funded by Arthritis Research UK and Pfizer. Dr. Helliwell disclosed that he has received consulting fees from Pfizer.
Women’s sexuality diminished by psoriasis
LAS VEGAS – Psoriasis has a negative impact on women’s sexual desire, sexual ability, and sexual relationships, according to Dr. Jennifer Cather of the Modern Dermatology-Aesthetics Center in Dallas.
Dr. Cather and her colleagues are developing a clinical tool to assess the disease’s sexual impact; as part of those efforts, they conducted 60 interviews with moderately to severely psoriatic women with a mean age of 41 years.
The survey results showed that sexuality was clearly another reason why it’s important to keep psoriasis in check. Pain and itchiness during sex, self-consciousness and embarrassment, and avoidance of dating and intimate relationships were among the most common problems the women reported.
"We really didn’t appreciate the impact psoriasis had on relationships" before the survey, Dr. Cather said at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar. "The sexual impact has been underappreciated," she said.
Those problems might help explain why women with psoriasis, especially those aged younger than 35 years, tend to have fewer babies, she added. Psoriatic women also have higher rates of induced and spontaneous abortions and are more likely to have preterm and underweight births.
Some of the women surveyed said that they worried about passing psoriasis to their children. "Maybe there’s voluntary childlessness," Dr. Cather said. But when women in her practice mention they don’t want kids for fear of passing on the disease, "I answer right back, ‘You don’t have to have what you have. I can help you with it,’ " she said.
Methotrexate, acitretin, and psoralen photochemotherapy (PUVA) are contraindicated during pregnancy, but ultraviolet B (UVB) treatments are safe, said Dr. Cather. She also uses tumor necrosis factor inhibitors, which are FDA pregnancy category B agents; she prefers etanercept for its short half-life (just over 4 days) and because ob.gyns. are usually familiar with it. The Organization of Teratology Information Specialists (OTIS) keeps the etanercept and adalimumab pregnancy registries, and "there’s [been] no signal to date" for those drugs, she said.
Psoriatic women should know that the odds are with them for having a normal pregnancy, and that pregnancy is likely to help clear their skin, Dr. Cather said.
But because half of the pregnancies in the United States are unplanned, it’s important to discuss pregnancy – and psoriasis treatment during pregnancy – as part of routine care. "It’s difficult if a psoriasis patient calls you in a panic because they’re not sure their drug is safe, and not sure their doctor will let them continue it,’ she said. Some ob.gyns. are comfortable with letting women stay on their psoriasis therapy, while others want them to quit everything, even topical steroids.
"In my clinic, when a psoriasis patient gets pregnant, usually we’ve planned for it and are excited about it, and we’ve had some dialogue with the ob.gyn.," she explained.
SDEF and this news organization are owned by Frontline Medical Communications. Dr. Cather is a consultant, speaker, or researcher for AbbVie, Novartis, Leo, Janssen, Amgen, Celgene, Merck, and Pfizer.
LAS VEGAS – Psoriasis has a negative impact on women’s sexual desire, sexual ability, and sexual relationships, according to Dr. Jennifer Cather of the Modern Dermatology-Aesthetics Center in Dallas.
Dr. Cather and her colleagues are developing a clinical tool to assess the disease’s sexual impact; as part of those efforts, they conducted 60 interviews with moderately to severely psoriatic women with a mean age of 41 years.
The survey results showed that sexuality was clearly another reason why it’s important to keep psoriasis in check. Pain and itchiness during sex, self-consciousness and embarrassment, and avoidance of dating and intimate relationships were among the most common problems the women reported.
"We really didn’t appreciate the impact psoriasis had on relationships" before the survey, Dr. Cather said at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar. "The sexual impact has been underappreciated," she said.
Those problems might help explain why women with psoriasis, especially those aged younger than 35 years, tend to have fewer babies, she added. Psoriatic women also have higher rates of induced and spontaneous abortions and are more likely to have preterm and underweight births.
Some of the women surveyed said that they worried about passing psoriasis to their children. "Maybe there’s voluntary childlessness," Dr. Cather said. But when women in her practice mention they don’t want kids for fear of passing on the disease, "I answer right back, ‘You don’t have to have what you have. I can help you with it,’ " she said.
Methotrexate, acitretin, and psoralen photochemotherapy (PUVA) are contraindicated during pregnancy, but ultraviolet B (UVB) treatments are safe, said Dr. Cather. She also uses tumor necrosis factor inhibitors, which are FDA pregnancy category B agents; she prefers etanercept for its short half-life (just over 4 days) and because ob.gyns. are usually familiar with it. The Organization of Teratology Information Specialists (OTIS) keeps the etanercept and adalimumab pregnancy registries, and "there’s [been] no signal to date" for those drugs, she said.
Psoriatic women should know that the odds are with them for having a normal pregnancy, and that pregnancy is likely to help clear their skin, Dr. Cather said.
But because half of the pregnancies in the United States are unplanned, it’s important to discuss pregnancy – and psoriasis treatment during pregnancy – as part of routine care. "It’s difficult if a psoriasis patient calls you in a panic because they’re not sure their drug is safe, and not sure their doctor will let them continue it,’ she said. Some ob.gyns. are comfortable with letting women stay on their psoriasis therapy, while others want them to quit everything, even topical steroids.
"In my clinic, when a psoriasis patient gets pregnant, usually we’ve planned for it and are excited about it, and we’ve had some dialogue with the ob.gyn.," she explained.
SDEF and this news organization are owned by Frontline Medical Communications. Dr. Cather is a consultant, speaker, or researcher for AbbVie, Novartis, Leo, Janssen, Amgen, Celgene, Merck, and Pfizer.
LAS VEGAS – Psoriasis has a negative impact on women’s sexual desire, sexual ability, and sexual relationships, according to Dr. Jennifer Cather of the Modern Dermatology-Aesthetics Center in Dallas.
Dr. Cather and her colleagues are developing a clinical tool to assess the disease’s sexual impact; as part of those efforts, they conducted 60 interviews with moderately to severely psoriatic women with a mean age of 41 years.
The survey results showed that sexuality was clearly another reason why it’s important to keep psoriasis in check. Pain and itchiness during sex, self-consciousness and embarrassment, and avoidance of dating and intimate relationships were among the most common problems the women reported.
"We really didn’t appreciate the impact psoriasis had on relationships" before the survey, Dr. Cather said at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar. "The sexual impact has been underappreciated," she said.
Those problems might help explain why women with psoriasis, especially those aged younger than 35 years, tend to have fewer babies, she added. Psoriatic women also have higher rates of induced and spontaneous abortions and are more likely to have preterm and underweight births.
Some of the women surveyed said that they worried about passing psoriasis to their children. "Maybe there’s voluntary childlessness," Dr. Cather said. But when women in her practice mention they don’t want kids for fear of passing on the disease, "I answer right back, ‘You don’t have to have what you have. I can help you with it,’ " she said.
Methotrexate, acitretin, and psoralen photochemotherapy (PUVA) are contraindicated during pregnancy, but ultraviolet B (UVB) treatments are safe, said Dr. Cather. She also uses tumor necrosis factor inhibitors, which are FDA pregnancy category B agents; she prefers etanercept for its short half-life (just over 4 days) and because ob.gyns. are usually familiar with it. The Organization of Teratology Information Specialists (OTIS) keeps the etanercept and adalimumab pregnancy registries, and "there’s [been] no signal to date" for those drugs, she said.
Psoriatic women should know that the odds are with them for having a normal pregnancy, and that pregnancy is likely to help clear their skin, Dr. Cather said.
But because half of the pregnancies in the United States are unplanned, it’s important to discuss pregnancy – and psoriasis treatment during pregnancy – as part of routine care. "It’s difficult if a psoriasis patient calls you in a panic because they’re not sure their drug is safe, and not sure their doctor will let them continue it,’ she said. Some ob.gyns. are comfortable with letting women stay on their psoriasis therapy, while others want them to quit everything, even topical steroids.
"In my clinic, when a psoriasis patient gets pregnant, usually we’ve planned for it and are excited about it, and we’ve had some dialogue with the ob.gyn.," she explained.
SDEF and this news organization are owned by Frontline Medical Communications. Dr. Cather is a consultant, speaker, or researcher for AbbVie, Novartis, Leo, Janssen, Amgen, Celgene, Merck, and Pfizer.
EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
Wet dressings work for pruritus when other options fail
LAS VEGAS – Wet dressing, a technique forgotten in most places but in continual use at the Mayo Clinic for more than 80 years, knocks out intractable pruritus – whatever its cause – in children and adults, according to Dr. Mark Davis, chair of the division of clinical dermatology at the clinic in Rochester, Minn.
"It’s a simple technique that works extraordinarily well for any itchy condition from head to toe, and has virtually no side effects. It stops itching reliably, when nothing else has worked," including prednisone, methotrexate, phototherapy, and elimination diets, among other strategies, he said at Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
Even so, "there’s remarkably little on this in the literature, and what’s published is mostly just in kids, but in adults it works brilliantly, too, particularly for atopic dermatitis. We use it when people come in itching from anything, like psoriasis," he said (J. Am. Acad. Dermatol. 2009;60:792-800).
"The commonest question [we hear from people] is ‘I’ve been going to doctors for years. Why didn’t anybody tell me about this?’ " said Dr. Davis.
If their pruritus is severe enough, patients will be admitted to the Mayo Clinic and have wet cloths applied to wherever the itch happens to be – above the waist, below the waist, the feet, or even the entire body, including the face – with a dry blanket on top if needed to ward of the chill. Patients can get up from bed for a bathroom break when the dressings are changed every 3 hours.
Topical steroids are used with the dressings up to three times a day; 1% percent hydrocortisone for the face or genitals, 0.1% or 0.05% triamcinolone elsewhere. The Mayo Clinic has never had a case of hypothalamic-pituitary-adrenal axis suppression with the technique, and insurance usually covers the cost for 3 days, Dr. Davis said.
When outpatient treatment is sufficient, pruritic patients are instructed to put on wet pajamas or long johns, or hop into the shower in dry ones, and then leave them on for 30 minutes to an hour, 3-4 times a day. Nurses, with the help of handouts and videos, teach patients how to do this, and call them every 24 hours to see how they are getting along.
Hospital patients get the same instructions at discharge. "Initially, they have to do [it] at least once a day for a number of weeks, and then they can use [the technique] on an as-needed basis, maybe once or twice a week," Dr. Davis said.
"Wet dressings went out of favor" in most places because "they are just so much trouble," he said. At present, the technique is "largely unknown," as is the reason why it works, he added.
Dr. Davis has no relevant disclosures. SDEF and this news organization are owned by Frontline Medical Communications.
LAS VEGAS – Wet dressing, a technique forgotten in most places but in continual use at the Mayo Clinic for more than 80 years, knocks out intractable pruritus – whatever its cause – in children and adults, according to Dr. Mark Davis, chair of the division of clinical dermatology at the clinic in Rochester, Minn.
"It’s a simple technique that works extraordinarily well for any itchy condition from head to toe, and has virtually no side effects. It stops itching reliably, when nothing else has worked," including prednisone, methotrexate, phototherapy, and elimination diets, among other strategies, he said at Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
Even so, "there’s remarkably little on this in the literature, and what’s published is mostly just in kids, but in adults it works brilliantly, too, particularly for atopic dermatitis. We use it when people come in itching from anything, like psoriasis," he said (J. Am. Acad. Dermatol. 2009;60:792-800).
"The commonest question [we hear from people] is ‘I’ve been going to doctors for years. Why didn’t anybody tell me about this?’ " said Dr. Davis.
If their pruritus is severe enough, patients will be admitted to the Mayo Clinic and have wet cloths applied to wherever the itch happens to be – above the waist, below the waist, the feet, or even the entire body, including the face – with a dry blanket on top if needed to ward of the chill. Patients can get up from bed for a bathroom break when the dressings are changed every 3 hours.
Topical steroids are used with the dressings up to three times a day; 1% percent hydrocortisone for the face or genitals, 0.1% or 0.05% triamcinolone elsewhere. The Mayo Clinic has never had a case of hypothalamic-pituitary-adrenal axis suppression with the technique, and insurance usually covers the cost for 3 days, Dr. Davis said.
When outpatient treatment is sufficient, pruritic patients are instructed to put on wet pajamas or long johns, or hop into the shower in dry ones, and then leave them on for 30 minutes to an hour, 3-4 times a day. Nurses, with the help of handouts and videos, teach patients how to do this, and call them every 24 hours to see how they are getting along.
Hospital patients get the same instructions at discharge. "Initially, they have to do [it] at least once a day for a number of weeks, and then they can use [the technique] on an as-needed basis, maybe once or twice a week," Dr. Davis said.
"Wet dressings went out of favor" in most places because "they are just so much trouble," he said. At present, the technique is "largely unknown," as is the reason why it works, he added.
Dr. Davis has no relevant disclosures. SDEF and this news organization are owned by Frontline Medical Communications.
LAS VEGAS – Wet dressing, a technique forgotten in most places but in continual use at the Mayo Clinic for more than 80 years, knocks out intractable pruritus – whatever its cause – in children and adults, according to Dr. Mark Davis, chair of the division of clinical dermatology at the clinic in Rochester, Minn.
"It’s a simple technique that works extraordinarily well for any itchy condition from head to toe, and has virtually no side effects. It stops itching reliably, when nothing else has worked," including prednisone, methotrexate, phototherapy, and elimination diets, among other strategies, he said at Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
Even so, "there’s remarkably little on this in the literature, and what’s published is mostly just in kids, but in adults it works brilliantly, too, particularly for atopic dermatitis. We use it when people come in itching from anything, like psoriasis," he said (J. Am. Acad. Dermatol. 2009;60:792-800).
"The commonest question [we hear from people] is ‘I’ve been going to doctors for years. Why didn’t anybody tell me about this?’ " said Dr. Davis.
If their pruritus is severe enough, patients will be admitted to the Mayo Clinic and have wet cloths applied to wherever the itch happens to be – above the waist, below the waist, the feet, or even the entire body, including the face – with a dry blanket on top if needed to ward of the chill. Patients can get up from bed for a bathroom break when the dressings are changed every 3 hours.
Topical steroids are used with the dressings up to three times a day; 1% percent hydrocortisone for the face or genitals, 0.1% or 0.05% triamcinolone elsewhere. The Mayo Clinic has never had a case of hypothalamic-pituitary-adrenal axis suppression with the technique, and insurance usually covers the cost for 3 days, Dr. Davis said.
When outpatient treatment is sufficient, pruritic patients are instructed to put on wet pajamas or long johns, or hop into the shower in dry ones, and then leave them on for 30 minutes to an hour, 3-4 times a day. Nurses, with the help of handouts and videos, teach patients how to do this, and call them every 24 hours to see how they are getting along.
Hospital patients get the same instructions at discharge. "Initially, they have to do [it] at least once a day for a number of weeks, and then they can use [the technique] on an as-needed basis, maybe once or twice a week," Dr. Davis said.
"Wet dressings went out of favor" in most places because "they are just so much trouble," he said. At present, the technique is "largely unknown," as is the reason why it works, he added.
Dr. Davis has no relevant disclosures. SDEF and this news organization are owned by Frontline Medical Communications.
AT SDEF LAS VEGAS DERMATOLOGY SEMINAR
Patient education on heart failure risk is crucial in psoriasis
As evidence supporting an association between psoriasis and cardiovascular disease continues to mount, dermatologists may be the first line of defense in lowering heart failure risk.
"The increased risk of cardiovascular disease for patients with psoriasis may be of a similar magnitude as other well-described CV risk factors, such as uncontrolled hypertension," said Dr. Bruce E. Strober of the University of Connecticut, Farmington. "Further, epidemiological studies show that psoriasis patients have a shortened life expectancy, likely as a result of their experience with CV comorbitidies."
Patient education is essential, he noted at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar. "At the very least, dermatologists should alert patients of the link between psoriasis and CV disease, and remind these patients of the necessity of having a primary care physician who monitors conventional risk factors of CV disease. Patients should be reminded that psoriasis is ‘systemic disease of inflammation’ that creates risks beyond the skin and may shorten life expectancy."
"Dermatologists who care for moderate to severe psoriasis patients should measure blood pressure and draw baseline blood tests assessing for abnormalities of cholesterol, triglycerides, kidney function, liver function, and blood glucose. Abnormalities should prompt an appropriate referral to a primary care physician," he noted.
In a population-based Dutch study presented at the annual congress of the European Society of Cardiology in September, adults with mild psoriasis developed 4.02 cases of new-onset heart failure per 1,000 person-years of follow-up, compared with 4.50/1,000 person-years in patients with severe psoriasis; both of which were significantly higher than the rate of 2.27/1,000 person-years in the general population.
Data from a 2006 study found that diabetes, hypertension, hyperlipidemia, and obesity were more prevalent in psoriasis patients, when compared with controls (J. Am. Acad. Dermatol. 2006;55:829-35). Diabetes and obesity were significantly more prevalent in patients with severe psoriasis, compared with patients with mild psoriasis.
Although the reasons for the increased cardiovascular risk in psoriasis patients remain unknown, possible causes include the use of dyslipidemic therapies, including corticosteroids, acitretin, and cyclosporine, as well as uncontrolled inflammation that could lead to endothelial dysfunction and dyslipidemia, said Dr. Strober. The prevalence of other associated and/or independent risk factors including obesity, hypertension, smoking, and alcohol misuse in psoriasis patients, also could play a role.
Dr. Strober disclosed relationships with multiple pharmaceutical companies including Abbott, Amgen, Janssen, Pfizer, Novartis, and Celgene. SDEF and this news organization are owned by Frontline Medical Communications.
As evidence supporting an association between psoriasis and cardiovascular disease continues to mount, dermatologists may be the first line of defense in lowering heart failure risk.
"The increased risk of cardiovascular disease for patients with psoriasis may be of a similar magnitude as other well-described CV risk factors, such as uncontrolled hypertension," said Dr. Bruce E. Strober of the University of Connecticut, Farmington. "Further, epidemiological studies show that psoriasis patients have a shortened life expectancy, likely as a result of their experience with CV comorbitidies."
Patient education is essential, he noted at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar. "At the very least, dermatologists should alert patients of the link between psoriasis and CV disease, and remind these patients of the necessity of having a primary care physician who monitors conventional risk factors of CV disease. Patients should be reminded that psoriasis is ‘systemic disease of inflammation’ that creates risks beyond the skin and may shorten life expectancy."
"Dermatologists who care for moderate to severe psoriasis patients should measure blood pressure and draw baseline blood tests assessing for abnormalities of cholesterol, triglycerides, kidney function, liver function, and blood glucose. Abnormalities should prompt an appropriate referral to a primary care physician," he noted.
In a population-based Dutch study presented at the annual congress of the European Society of Cardiology in September, adults with mild psoriasis developed 4.02 cases of new-onset heart failure per 1,000 person-years of follow-up, compared with 4.50/1,000 person-years in patients with severe psoriasis; both of which were significantly higher than the rate of 2.27/1,000 person-years in the general population.
Data from a 2006 study found that diabetes, hypertension, hyperlipidemia, and obesity were more prevalent in psoriasis patients, when compared with controls (J. Am. Acad. Dermatol. 2006;55:829-35). Diabetes and obesity were significantly more prevalent in patients with severe psoriasis, compared with patients with mild psoriasis.
Although the reasons for the increased cardiovascular risk in psoriasis patients remain unknown, possible causes include the use of dyslipidemic therapies, including corticosteroids, acitretin, and cyclosporine, as well as uncontrolled inflammation that could lead to endothelial dysfunction and dyslipidemia, said Dr. Strober. The prevalence of other associated and/or independent risk factors including obesity, hypertension, smoking, and alcohol misuse in psoriasis patients, also could play a role.
Dr. Strober disclosed relationships with multiple pharmaceutical companies including Abbott, Amgen, Janssen, Pfizer, Novartis, and Celgene. SDEF and this news organization are owned by Frontline Medical Communications.
As evidence supporting an association between psoriasis and cardiovascular disease continues to mount, dermatologists may be the first line of defense in lowering heart failure risk.
"The increased risk of cardiovascular disease for patients with psoriasis may be of a similar magnitude as other well-described CV risk factors, such as uncontrolled hypertension," said Dr. Bruce E. Strober of the University of Connecticut, Farmington. "Further, epidemiological studies show that psoriasis patients have a shortened life expectancy, likely as a result of their experience with CV comorbitidies."
Patient education is essential, he noted at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar. "At the very least, dermatologists should alert patients of the link between psoriasis and CV disease, and remind these patients of the necessity of having a primary care physician who monitors conventional risk factors of CV disease. Patients should be reminded that psoriasis is ‘systemic disease of inflammation’ that creates risks beyond the skin and may shorten life expectancy."
"Dermatologists who care for moderate to severe psoriasis patients should measure blood pressure and draw baseline blood tests assessing for abnormalities of cholesterol, triglycerides, kidney function, liver function, and blood glucose. Abnormalities should prompt an appropriate referral to a primary care physician," he noted.
In a population-based Dutch study presented at the annual congress of the European Society of Cardiology in September, adults with mild psoriasis developed 4.02 cases of new-onset heart failure per 1,000 person-years of follow-up, compared with 4.50/1,000 person-years in patients with severe psoriasis; both of which were significantly higher than the rate of 2.27/1,000 person-years in the general population.
Data from a 2006 study found that diabetes, hypertension, hyperlipidemia, and obesity were more prevalent in psoriasis patients, when compared with controls (J. Am. Acad. Dermatol. 2006;55:829-35). Diabetes and obesity were significantly more prevalent in patients with severe psoriasis, compared with patients with mild psoriasis.
Although the reasons for the increased cardiovascular risk in psoriasis patients remain unknown, possible causes include the use of dyslipidemic therapies, including corticosteroids, acitretin, and cyclosporine, as well as uncontrolled inflammation that could lead to endothelial dysfunction and dyslipidemia, said Dr. Strober. The prevalence of other associated and/or independent risk factors including obesity, hypertension, smoking, and alcohol misuse in psoriasis patients, also could play a role.
Dr. Strober disclosed relationships with multiple pharmaceutical companies including Abbott, Amgen, Janssen, Pfizer, Novartis, and Celgene. SDEF and this news organization are owned by Frontline Medical Communications.
EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
Product News: 11 2013
Cimzia
UCB, Inc, obtains US Food and Drug Administration approval of Cimzia (certolizumab pegol), a tumor necrosis factor blocker for the treatment of active psoriatic arthritis in adults. Cimzia also is indicated in adults for the treatment of moderate to severe rheumatoid arthritis and to reduce signs and symptoms of moderately to severely active Crohn disease. For more information, visit www.cimzia.com.
La Roche-Posay Antiaging Products
La Roche-Posay Laboratoire Dermatologique releases 3 antiaging products: Mela-D Deep Cleansing Brightening Foaming Cream, Redermic [R] Eyes, and Substiane [+] Serum. The Mela-D product is a deep-cleansing and brightening formula with lipohy-droxy acid that combats dark spots and gives the skin a clean and smooth appearance. Redermic [R] Eyes combines 0.01% pure retinol with a retinol booster complex and caffeine to reduce crow’s-feet and dark circles to rejuvenate the eye area. Substiane [+] Serum increases the volume and elasticity of the skin. All products are physician dispensed or available in select drugstores and online. For more information, visit www.laroche-posay.us.
Otrexup
Antares Pharma Inc obtains US Food and Drug Administration approval of Otrexup (methotrexate), a folate analog metabolic inhibitor for the treatment of severe, recalcitrant, disabling psoriasis in adults who have not responded to other therapies. Otrexup is self-administered subcutaneously once weekly via an easy-to-use, single-dose, disposable autoinjector, which utilizes Vibex Medi-Jet technology. Otrexup also is indicated for adults with severe active rheumatoid arthritis and for children with active polyarticular juvenile idiopathic arthritis. For more information, visit www.otrexup.com.
Valeant Partners With National Coalition Against Domestic
Violence Valeant Pharmaceuticals International, Inc, announces a partnership with the National Coalition Against Domestic Violence (NCADV) to help improve the lives of women. Valeant pledges to donate a portion of sales from Medicis and Obagi products to support the initiatives of the NCADV. For more information, visit enddomesticviolence.valeant.com.
If you would like your product included in Product News, please e-mail a press release to Melissa Steiger at msteiger@frontlinemedcom.com.
Cimzia
UCB, Inc, obtains US Food and Drug Administration approval of Cimzia (certolizumab pegol), a tumor necrosis factor blocker for the treatment of active psoriatic arthritis in adults. Cimzia also is indicated in adults for the treatment of moderate to severe rheumatoid arthritis and to reduce signs and symptoms of moderately to severely active Crohn disease. For more information, visit www.cimzia.com.
La Roche-Posay Antiaging Products
La Roche-Posay Laboratoire Dermatologique releases 3 antiaging products: Mela-D Deep Cleansing Brightening Foaming Cream, Redermic [R] Eyes, and Substiane [+] Serum. The Mela-D product is a deep-cleansing and brightening formula with lipohy-droxy acid that combats dark spots and gives the skin a clean and smooth appearance. Redermic [R] Eyes combines 0.01% pure retinol with a retinol booster complex and caffeine to reduce crow’s-feet and dark circles to rejuvenate the eye area. Substiane [+] Serum increases the volume and elasticity of the skin. All products are physician dispensed or available in select drugstores and online. For more information, visit www.laroche-posay.us.
Otrexup
Antares Pharma Inc obtains US Food and Drug Administration approval of Otrexup (methotrexate), a folate analog metabolic inhibitor for the treatment of severe, recalcitrant, disabling psoriasis in adults who have not responded to other therapies. Otrexup is self-administered subcutaneously once weekly via an easy-to-use, single-dose, disposable autoinjector, which utilizes Vibex Medi-Jet technology. Otrexup also is indicated for adults with severe active rheumatoid arthritis and for children with active polyarticular juvenile idiopathic arthritis. For more information, visit www.otrexup.com.
Valeant Partners With National Coalition Against Domestic
Violence Valeant Pharmaceuticals International, Inc, announces a partnership with the National Coalition Against Domestic Violence (NCADV) to help improve the lives of women. Valeant pledges to donate a portion of sales from Medicis and Obagi products to support the initiatives of the NCADV. For more information, visit enddomesticviolence.valeant.com.
If you would like your product included in Product News, please e-mail a press release to Melissa Steiger at msteiger@frontlinemedcom.com.
Cimzia
UCB, Inc, obtains US Food and Drug Administration approval of Cimzia (certolizumab pegol), a tumor necrosis factor blocker for the treatment of active psoriatic arthritis in adults. Cimzia also is indicated in adults for the treatment of moderate to severe rheumatoid arthritis and to reduce signs and symptoms of moderately to severely active Crohn disease. For more information, visit www.cimzia.com.
La Roche-Posay Antiaging Products
La Roche-Posay Laboratoire Dermatologique releases 3 antiaging products: Mela-D Deep Cleansing Brightening Foaming Cream, Redermic [R] Eyes, and Substiane [+] Serum. The Mela-D product is a deep-cleansing and brightening formula with lipohy-droxy acid that combats dark spots and gives the skin a clean and smooth appearance. Redermic [R] Eyes combines 0.01% pure retinol with a retinol booster complex and caffeine to reduce crow’s-feet and dark circles to rejuvenate the eye area. Substiane [+] Serum increases the volume and elasticity of the skin. All products are physician dispensed or available in select drugstores and online. For more information, visit www.laroche-posay.us.
Otrexup
Antares Pharma Inc obtains US Food and Drug Administration approval of Otrexup (methotrexate), a folate analog metabolic inhibitor for the treatment of severe, recalcitrant, disabling psoriasis in adults who have not responded to other therapies. Otrexup is self-administered subcutaneously once weekly via an easy-to-use, single-dose, disposable autoinjector, which utilizes Vibex Medi-Jet technology. Otrexup also is indicated for adults with severe active rheumatoid arthritis and for children with active polyarticular juvenile idiopathic arthritis. For more information, visit www.otrexup.com.
Valeant Partners With National Coalition Against Domestic
Violence Valeant Pharmaceuticals International, Inc, announces a partnership with the National Coalition Against Domestic Violence (NCADV) to help improve the lives of women. Valeant pledges to donate a portion of sales from Medicis and Obagi products to support the initiatives of the NCADV. For more information, visit enddomesticviolence.valeant.com.
If you would like your product included in Product News, please e-mail a press release to Melissa Steiger at msteiger@frontlinemedcom.com.
Disease Burden and Treatment Adherence in Psoriasis Patients
Test your knowledge on QOL issues for patients with psoriasis with MD-IQ: the medical intelligence quiz. Click here to answer 5 questions.
Biologic therapy doesn’t preclude vaccinating psoriasis patients
LAS VEGAS – Dermatologists, like all physicians, play an important role not just in the treatment of disease, but also in the prevention of disease, and that means vaccinating patients when indicated, according to Dr. Stephen K. Tyring.
The varicella vaccines, including the chicken pox and shingles vaccines, and the human papillomavirus (HPV), hepatitis, and influenza virus vaccines are among those to consider offering to patients when appropriate, Dr. Tyring said at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
Concerns arise, however, when it comes to vaccinating psoriasis patients who are – or will be – treated with biologics.
Ideally, vaccination should be offered before treatment is initiated, he said, but for some patients – such as those who are under the age for which their insurance company will cover a vaccine (age 50 or 60 for the shingles vaccine, depending on the insurance company, for example) – this may not be possible. In those already taking a biologic drug, the question is how to vaccinate safely and without interfering with treatment efficacy, he said.
"What we usually do – and there’s no absolute golden rule – is ask the patient to stop the drug for one to two half-lives of the drug," said Dr. Tyring of the University of Texas, Houston. We suggest stopping one half-life for killed, subunit, or recombinant virus vaccines and stopping for two half-lives for live attenuated virus vaccines.
With the tumor necrosis factor (TNF) inhibitor etanercept (Enbrel), for example, that means skipping one to two weekly injections.
"That’s a little bit more than a half-life, but if a patient is using Enbrel once or twice a week, the next week, instead of injecting the Enbrel, they can just come in and get the shingles vaccine [Zostavax]," he said.
Adalimumab (Humira), another anti-TNF drug, has a longer half-life, so that has to be taken into account. Things get a little more complicated when it comes to patients treated with ustekinumab (Stelera), which has a particularly long half-life, he noted.
"It’s not quite as easy, because it’s not clear how long to wait after Stelera to give a vaccine. Most efficacy is seen in the first month, and when patients come back at 3 months, they often are starting to get a little psoriasis back. Therefore, 2 months following an injection of Stelera is about the right time to get the vaccine; that way you get the minimum immunosuppression and the maximum chance to respond," he said.
Similarly, with other vaccines like the Gardasil HPV vaccine and the hepatitis A and B vaccines that require a series of shots, the biologic should be stopped for a half-life. This will mean skipping biologic dosing multiple times, but in most cases this won’t be problematic, because it typically takes about 3 months for a patient who is clear to experience significant psoriasis recurrence.
"There’s really no danger in skipping," he said.
While some may advocate waiting two half-lives after vaccination, most experts agree that is unnecessary for killed, recombinant, or subunit (for example, injectable influenza) virus vaccines but is advisable for live attenuated virus vaccines like the intranasal influenza virus vaccine or the herpes zoster vaccine Zostavax, he said.
Dr. Tyring also noted during his presentation that in his experience, concerns about increased infection risk in patients taking TNF inhibitors have been unfounded. In fact, treatment appears to provide an unexpected benefit for those who do become infected with herpes zoster: a reduced risk of postherpetic neuralgia, even among older individuals who are generally at particularly high risk.
After noticing this benefit in his own patients, Dr. Tyring asked his colleagues and found that they, too, had noticed a similar pattern. In 2011, he and his colleagues published a retrospective study of 206 patients on TNF inhibitors who developed herpes zoster, and with only two exceptions involving patients on both a TNF inhibitor and methotrexate, patients universally experienced milder symptoms if they developed shingles while on a TNF inhibitor, he said, noting that this is much lower than rates reported in the literature in the general population (J. Med. Virol. 2011;83:2051-5).
SDEF and this news organization are owned by Frontline Medical Communications.
LAS VEGAS – Dermatologists, like all physicians, play an important role not just in the treatment of disease, but also in the prevention of disease, and that means vaccinating patients when indicated, according to Dr. Stephen K. Tyring.
The varicella vaccines, including the chicken pox and shingles vaccines, and the human papillomavirus (HPV), hepatitis, and influenza virus vaccines are among those to consider offering to patients when appropriate, Dr. Tyring said at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
Concerns arise, however, when it comes to vaccinating psoriasis patients who are – or will be – treated with biologics.
Ideally, vaccination should be offered before treatment is initiated, he said, but for some patients – such as those who are under the age for which their insurance company will cover a vaccine (age 50 or 60 for the shingles vaccine, depending on the insurance company, for example) – this may not be possible. In those already taking a biologic drug, the question is how to vaccinate safely and without interfering with treatment efficacy, he said.
"What we usually do – and there’s no absolute golden rule – is ask the patient to stop the drug for one to two half-lives of the drug," said Dr. Tyring of the University of Texas, Houston. We suggest stopping one half-life for killed, subunit, or recombinant virus vaccines and stopping for two half-lives for live attenuated virus vaccines.
With the tumor necrosis factor (TNF) inhibitor etanercept (Enbrel), for example, that means skipping one to two weekly injections.
"That’s a little bit more than a half-life, but if a patient is using Enbrel once or twice a week, the next week, instead of injecting the Enbrel, they can just come in and get the shingles vaccine [Zostavax]," he said.
Adalimumab (Humira), another anti-TNF drug, has a longer half-life, so that has to be taken into account. Things get a little more complicated when it comes to patients treated with ustekinumab (Stelera), which has a particularly long half-life, he noted.
"It’s not quite as easy, because it’s not clear how long to wait after Stelera to give a vaccine. Most efficacy is seen in the first month, and when patients come back at 3 months, they often are starting to get a little psoriasis back. Therefore, 2 months following an injection of Stelera is about the right time to get the vaccine; that way you get the minimum immunosuppression and the maximum chance to respond," he said.
Similarly, with other vaccines like the Gardasil HPV vaccine and the hepatitis A and B vaccines that require a series of shots, the biologic should be stopped for a half-life. This will mean skipping biologic dosing multiple times, but in most cases this won’t be problematic, because it typically takes about 3 months for a patient who is clear to experience significant psoriasis recurrence.
"There’s really no danger in skipping," he said.
While some may advocate waiting two half-lives after vaccination, most experts agree that is unnecessary for killed, recombinant, or subunit (for example, injectable influenza) virus vaccines but is advisable for live attenuated virus vaccines like the intranasal influenza virus vaccine or the herpes zoster vaccine Zostavax, he said.
Dr. Tyring also noted during his presentation that in his experience, concerns about increased infection risk in patients taking TNF inhibitors have been unfounded. In fact, treatment appears to provide an unexpected benefit for those who do become infected with herpes zoster: a reduced risk of postherpetic neuralgia, even among older individuals who are generally at particularly high risk.
After noticing this benefit in his own patients, Dr. Tyring asked his colleagues and found that they, too, had noticed a similar pattern. In 2011, he and his colleagues published a retrospective study of 206 patients on TNF inhibitors who developed herpes zoster, and with only two exceptions involving patients on both a TNF inhibitor and methotrexate, patients universally experienced milder symptoms if they developed shingles while on a TNF inhibitor, he said, noting that this is much lower than rates reported in the literature in the general population (J. Med. Virol. 2011;83:2051-5).
SDEF and this news organization are owned by Frontline Medical Communications.
LAS VEGAS – Dermatologists, like all physicians, play an important role not just in the treatment of disease, but also in the prevention of disease, and that means vaccinating patients when indicated, according to Dr. Stephen K. Tyring.
The varicella vaccines, including the chicken pox and shingles vaccines, and the human papillomavirus (HPV), hepatitis, and influenza virus vaccines are among those to consider offering to patients when appropriate, Dr. Tyring said at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
Concerns arise, however, when it comes to vaccinating psoriasis patients who are – or will be – treated with biologics.
Ideally, vaccination should be offered before treatment is initiated, he said, but for some patients – such as those who are under the age for which their insurance company will cover a vaccine (age 50 or 60 for the shingles vaccine, depending on the insurance company, for example) – this may not be possible. In those already taking a biologic drug, the question is how to vaccinate safely and without interfering with treatment efficacy, he said.
"What we usually do – and there’s no absolute golden rule – is ask the patient to stop the drug for one to two half-lives of the drug," said Dr. Tyring of the University of Texas, Houston. We suggest stopping one half-life for killed, subunit, or recombinant virus vaccines and stopping for two half-lives for live attenuated virus vaccines.
With the tumor necrosis factor (TNF) inhibitor etanercept (Enbrel), for example, that means skipping one to two weekly injections.
"That’s a little bit more than a half-life, but if a patient is using Enbrel once or twice a week, the next week, instead of injecting the Enbrel, they can just come in and get the shingles vaccine [Zostavax]," he said.
Adalimumab (Humira), another anti-TNF drug, has a longer half-life, so that has to be taken into account. Things get a little more complicated when it comes to patients treated with ustekinumab (Stelera), which has a particularly long half-life, he noted.
"It’s not quite as easy, because it’s not clear how long to wait after Stelera to give a vaccine. Most efficacy is seen in the first month, and when patients come back at 3 months, they often are starting to get a little psoriasis back. Therefore, 2 months following an injection of Stelera is about the right time to get the vaccine; that way you get the minimum immunosuppression and the maximum chance to respond," he said.
Similarly, with other vaccines like the Gardasil HPV vaccine and the hepatitis A and B vaccines that require a series of shots, the biologic should be stopped for a half-life. This will mean skipping biologic dosing multiple times, but in most cases this won’t be problematic, because it typically takes about 3 months for a patient who is clear to experience significant psoriasis recurrence.
"There’s really no danger in skipping," he said.
While some may advocate waiting two half-lives after vaccination, most experts agree that is unnecessary for killed, recombinant, or subunit (for example, injectable influenza) virus vaccines but is advisable for live attenuated virus vaccines like the intranasal influenza virus vaccine or the herpes zoster vaccine Zostavax, he said.
Dr. Tyring also noted during his presentation that in his experience, concerns about increased infection risk in patients taking TNF inhibitors have been unfounded. In fact, treatment appears to provide an unexpected benefit for those who do become infected with herpes zoster: a reduced risk of postherpetic neuralgia, even among older individuals who are generally at particularly high risk.
After noticing this benefit in his own patients, Dr. Tyring asked his colleagues and found that they, too, had noticed a similar pattern. In 2011, he and his colleagues published a retrospective study of 206 patients on TNF inhibitors who developed herpes zoster, and with only two exceptions involving patients on both a TNF inhibitor and methotrexate, patients universally experienced milder symptoms if they developed shingles while on a TNF inhibitor, he said, noting that this is much lower than rates reported in the literature in the general population (J. Med. Virol. 2011;83:2051-5).
SDEF and this news organization are owned by Frontline Medical Communications.
EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
Don’t brush off topical therapies for psoriasis
Biologics and systemic therapies command much of the spotlight for treating psoriasis, but topical therapy remains an effective option for many psoriasis patients, according to Dr. Linda Stein Gold.
In fact, up to 80% of psoriasis patients can be adequately treated with topical therapy (N. Engl. J. Med. 2005;352:1899-912), said Dr. Stein Gold at the Skin Disease Education Foundation’s (SDEF’s) annual Las Vegas dermatology seminar.
She offered several principles to help clinicians make the most of topical therapies by troubleshooting potential problems.
• Check the amount. One gram of most topical psoriasis products covers 4% of body surface area per application, so a 60-gram tube should treat 4% of body surface area for a month, said Dr. Stein Gold, citing guidelines developed by Dr. Alan Menter and his colleagues and approved by the American Academy of Dermatology in 2009.
• Don’t miss corticosteroid allergies. "We are missing this," said Dr. Stein Gold, director of dermatology research at Henry Ford Hospital in Detroit. Suspect a possible allergy if a patient returns with a worsening rash after using hydrocortisone, for example. Data have shown that between 0.2% and 5% of all dermatitis patients have a steroid allergy. "Allergy to the active molecule or to the vehicle should be suspected in all patients who don’t respond as expected to topical steroids," she noted.
• Visit (or revisit) vitamin D. Another plus for topical therapy is the usefulness of topical vitamin D for tricky areas, such as the forehead, armpit, groin, and the area behind the ears. Data from a randomized trial of 75 patients found calcitriol ointment to be significantly more effective against target lesions and better tolerated by patients than calcipotriene ointment (Br. J. Dermatol. 2003;148:326-33).
• Don’t forget coal tar. Simple, but effective, coal tar is a proven safe topical psoriasis treatment and is available in several vehicles, including solution and foam. Data from a study of more than 13,000 patients with psoriasis and eczema found that coal tar was safe, and that it did not increase the risk for skin cancer (J. Invest. Dermatol. 2010;130:953-61).
The future of topical psoriasis therapy is not static, said Dr. Stein Gold. New molecules – notably topical Janus kinase inhibitors and phosphodiesterase-4 inhibitors – are currently being explored in clinical trials.
Dr. Stein Gold disclosed relationships with Leo, Medicis, and other companies. SDEF and this news organization are owned by Frontline Medical Communications.
Biologics and systemic therapies command much of the spotlight for treating psoriasis, but topical therapy remains an effective option for many psoriasis patients, according to Dr. Linda Stein Gold.
In fact, up to 80% of psoriasis patients can be adequately treated with topical therapy (N. Engl. J. Med. 2005;352:1899-912), said Dr. Stein Gold at the Skin Disease Education Foundation’s (SDEF’s) annual Las Vegas dermatology seminar.
She offered several principles to help clinicians make the most of topical therapies by troubleshooting potential problems.
• Check the amount. One gram of most topical psoriasis products covers 4% of body surface area per application, so a 60-gram tube should treat 4% of body surface area for a month, said Dr. Stein Gold, citing guidelines developed by Dr. Alan Menter and his colleagues and approved by the American Academy of Dermatology in 2009.
• Don’t miss corticosteroid allergies. "We are missing this," said Dr. Stein Gold, director of dermatology research at Henry Ford Hospital in Detroit. Suspect a possible allergy if a patient returns with a worsening rash after using hydrocortisone, for example. Data have shown that between 0.2% and 5% of all dermatitis patients have a steroid allergy. "Allergy to the active molecule or to the vehicle should be suspected in all patients who don’t respond as expected to topical steroids," she noted.
• Visit (or revisit) vitamin D. Another plus for topical therapy is the usefulness of topical vitamin D for tricky areas, such as the forehead, armpit, groin, and the area behind the ears. Data from a randomized trial of 75 patients found calcitriol ointment to be significantly more effective against target lesions and better tolerated by patients than calcipotriene ointment (Br. J. Dermatol. 2003;148:326-33).
• Don’t forget coal tar. Simple, but effective, coal tar is a proven safe topical psoriasis treatment and is available in several vehicles, including solution and foam. Data from a study of more than 13,000 patients with psoriasis and eczema found that coal tar was safe, and that it did not increase the risk for skin cancer (J. Invest. Dermatol. 2010;130:953-61).
The future of topical psoriasis therapy is not static, said Dr. Stein Gold. New molecules – notably topical Janus kinase inhibitors and phosphodiesterase-4 inhibitors – are currently being explored in clinical trials.
Dr. Stein Gold disclosed relationships with Leo, Medicis, and other companies. SDEF and this news organization are owned by Frontline Medical Communications.
Biologics and systemic therapies command much of the spotlight for treating psoriasis, but topical therapy remains an effective option for many psoriasis patients, according to Dr. Linda Stein Gold.
In fact, up to 80% of psoriasis patients can be adequately treated with topical therapy (N. Engl. J. Med. 2005;352:1899-912), said Dr. Stein Gold at the Skin Disease Education Foundation’s (SDEF’s) annual Las Vegas dermatology seminar.
She offered several principles to help clinicians make the most of topical therapies by troubleshooting potential problems.
• Check the amount. One gram of most topical psoriasis products covers 4% of body surface area per application, so a 60-gram tube should treat 4% of body surface area for a month, said Dr. Stein Gold, citing guidelines developed by Dr. Alan Menter and his colleagues and approved by the American Academy of Dermatology in 2009.
• Don’t miss corticosteroid allergies. "We are missing this," said Dr. Stein Gold, director of dermatology research at Henry Ford Hospital in Detroit. Suspect a possible allergy if a patient returns with a worsening rash after using hydrocortisone, for example. Data have shown that between 0.2% and 5% of all dermatitis patients have a steroid allergy. "Allergy to the active molecule or to the vehicle should be suspected in all patients who don’t respond as expected to topical steroids," she noted.
• Visit (or revisit) vitamin D. Another plus for topical therapy is the usefulness of topical vitamin D for tricky areas, such as the forehead, armpit, groin, and the area behind the ears. Data from a randomized trial of 75 patients found calcitriol ointment to be significantly more effective against target lesions and better tolerated by patients than calcipotriene ointment (Br. J. Dermatol. 2003;148:326-33).
• Don’t forget coal tar. Simple, but effective, coal tar is a proven safe topical psoriasis treatment and is available in several vehicles, including solution and foam. Data from a study of more than 13,000 patients with psoriasis and eczema found that coal tar was safe, and that it did not increase the risk for skin cancer (J. Invest. Dermatol. 2010;130:953-61).
The future of topical psoriasis therapy is not static, said Dr. Stein Gold. New molecules – notably topical Janus kinase inhibitors and phosphodiesterase-4 inhibitors – are currently being explored in clinical trials.
Dr. Stein Gold disclosed relationships with Leo, Medicis, and other companies. SDEF and this news organization are owned by Frontline Medical Communications.
EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR