User login
Desoximetasone spray succeeds in psoriasis studies
Significantly more psoriasis patients randomized to a 0.25% desoximetasone spray showed clinical success and treatment success, compared with those who used a placebo vehicle, based on data from a pair of phase III studies. The findings were published in the December issue of Journal of Drugs in Dermatology by Dr. Leon Kircik of Indiana University School of Medicine, Indianapolis, and his colleagues.
The study population included adults with moderate to severe plaque psoriasis. The patients used the spray twice daily for 28 days, and they were assessed at baseline and during the study using the Physician Global Assessment score and the Total Lesion Severity Score. No significant differences in adverse events were reported between the treatment and placebo groups, and no patients reported stinging or burning from the spray formulation (J. Drugs Dermatol. 2013;12:1404-10).
Significantly more psoriasis patients randomized to a 0.25% desoximetasone spray showed clinical success and treatment success, compared with those who used a placebo vehicle, based on data from a pair of phase III studies. The findings were published in the December issue of Journal of Drugs in Dermatology by Dr. Leon Kircik of Indiana University School of Medicine, Indianapolis, and his colleagues.
The study population included adults with moderate to severe plaque psoriasis. The patients used the spray twice daily for 28 days, and they were assessed at baseline and during the study using the Physician Global Assessment score and the Total Lesion Severity Score. No significant differences in adverse events were reported between the treatment and placebo groups, and no patients reported stinging or burning from the spray formulation (J. Drugs Dermatol. 2013;12:1404-10).
Significantly more psoriasis patients randomized to a 0.25% desoximetasone spray showed clinical success and treatment success, compared with those who used a placebo vehicle, based on data from a pair of phase III studies. The findings were published in the December issue of Journal of Drugs in Dermatology by Dr. Leon Kircik of Indiana University School of Medicine, Indianapolis, and his colleagues.
The study population included adults with moderate to severe plaque psoriasis. The patients used the spray twice daily for 28 days, and they were assessed at baseline and during the study using the Physician Global Assessment score and the Total Lesion Severity Score. No significant differences in adverse events were reported between the treatment and placebo groups, and no patients reported stinging or burning from the spray formulation (J. Drugs Dermatol. 2013;12:1404-10).
FROM JOURNAL OF DRUGS IN DERMATOLOGY
Consider small-fiber neuropathies in systemic lupus erythematosus
Small-fiber neuropathy is one of the most common types of peripheral neuropathy affecting patients with systemic lupus erythematosus, but it isn’t even mentioned in the American College of Rheumatology neuropsychiatric case definitions of manifestations of the disorder, according to a retrospective analysis of cohort of 2,097 patients with SLE.
Other types of peripheral neuropathy, such as acute inflammatory demyelinating neuropathies (for example, Guillain-Barré syndrome), plexopathies, and mononeuritis multiplex, are well described in the ACR-NPSLE case definitions but occur much less frequently. This, combined with the fact that small-fiber neuropathies often present as "unorthodox" pain patterns, indicates that they are underdiagnosed, said Dr. Amin Oomatia of the University of Cambridge, England, and his coinvestigators at John Hopkins University, Baltimore.
Small-fiber neuropathies arise through mechanisms that are distinct from those of other neuropathies and require different diagnostic strategies to be properly identified. In particular, small-fiber neuropathies do not always conform to the "stocking-and-glove" pattern of pain that is typical of other neuropathies in SLE, so it is likely that many affected patients "may be regarded in routine clinical care as having a ‘nonorganic’ pain disorder.
"Our findings suggest that rheumatologists and other clinicians who confront SLE patients with seemingly improbable pain patterns should consider the diagnosis of a small-fiber neuropathy," the investigators wrote, especially since it may occur in the face of normal electrodiagnostic studies.
Dr. Oomatia and his colleagues based these conclusions on their retrospective study of one medical center’s 25-year experience treating 2,097 SLE patients – the Johns Hopkins Lupus Cohort. Using details in a database of patients’ electronic medical records, they identified 82 patients who had peripheral neuropathies related to SLE.
Only one patient had peripheral neuropathy attributable to Guillain-Barré syndrome, only one patient had a plexopathy, and only six patients had mononeuritis multiplex, demonstrating that these are very infrequent complications of SLE even though they are included in ACR case definitions.
In contrast, 14 patients (17% of those with peripheral neuropathy) had biopsy-proven small-fiber neuropathies, and most of them presented with "an entirely different and unorthodox pain distribution" characterized as patchy, asymmetric, or proximal.
In particular, nine patients had pain affecting the face, torso, and/or proximal extremities. Three had burning pain over their entire bodies, the investigators said (Arthritis Rheum. 2013 Dec. 10 [doi:10.1002/art.38302]).
In these cases, punch skin biopsy showed abnormalities that disproportionately affected the proximal thigh, "which is considered a surrogate indicator of proximal-most dorsal root ganglia neuronal cell loss," they wrote. In contrast, other patients who had the typical distal pattern of neuropathic pain showed decreased intraepidermal nerve-fiber densities in the distal leg, a surrogate indicator of distal-most axonal degeneration.
Another distinguishing feature of small-fiber neuropathy was its association with a history of herpes zoster virus, opportunistic infections, and osteoporotic fractures, all unrelated to corticosteroid dose, Dr. Oomatia and his associates said.
This study was supported in part by the National Institutes of Health and the National Center for Research Resources. No potential financial conflicts of interest were reported.
Small-fiber neuropathy is one of the most common types of peripheral neuropathy affecting patients with systemic lupus erythematosus, but it isn’t even mentioned in the American College of Rheumatology neuropsychiatric case definitions of manifestations of the disorder, according to a retrospective analysis of cohort of 2,097 patients with SLE.
Other types of peripheral neuropathy, such as acute inflammatory demyelinating neuropathies (for example, Guillain-Barré syndrome), plexopathies, and mononeuritis multiplex, are well described in the ACR-NPSLE case definitions but occur much less frequently. This, combined with the fact that small-fiber neuropathies often present as "unorthodox" pain patterns, indicates that they are underdiagnosed, said Dr. Amin Oomatia of the University of Cambridge, England, and his coinvestigators at John Hopkins University, Baltimore.
Small-fiber neuropathies arise through mechanisms that are distinct from those of other neuropathies and require different diagnostic strategies to be properly identified. In particular, small-fiber neuropathies do not always conform to the "stocking-and-glove" pattern of pain that is typical of other neuropathies in SLE, so it is likely that many affected patients "may be regarded in routine clinical care as having a ‘nonorganic’ pain disorder.
"Our findings suggest that rheumatologists and other clinicians who confront SLE patients with seemingly improbable pain patterns should consider the diagnosis of a small-fiber neuropathy," the investigators wrote, especially since it may occur in the face of normal electrodiagnostic studies.
Dr. Oomatia and his colleagues based these conclusions on their retrospective study of one medical center’s 25-year experience treating 2,097 SLE patients – the Johns Hopkins Lupus Cohort. Using details in a database of patients’ electronic medical records, they identified 82 patients who had peripheral neuropathies related to SLE.
Only one patient had peripheral neuropathy attributable to Guillain-Barré syndrome, only one patient had a plexopathy, and only six patients had mononeuritis multiplex, demonstrating that these are very infrequent complications of SLE even though they are included in ACR case definitions.
In contrast, 14 patients (17% of those with peripheral neuropathy) had biopsy-proven small-fiber neuropathies, and most of them presented with "an entirely different and unorthodox pain distribution" characterized as patchy, asymmetric, or proximal.
In particular, nine patients had pain affecting the face, torso, and/or proximal extremities. Three had burning pain over their entire bodies, the investigators said (Arthritis Rheum. 2013 Dec. 10 [doi:10.1002/art.38302]).
In these cases, punch skin biopsy showed abnormalities that disproportionately affected the proximal thigh, "which is considered a surrogate indicator of proximal-most dorsal root ganglia neuronal cell loss," they wrote. In contrast, other patients who had the typical distal pattern of neuropathic pain showed decreased intraepidermal nerve-fiber densities in the distal leg, a surrogate indicator of distal-most axonal degeneration.
Another distinguishing feature of small-fiber neuropathy was its association with a history of herpes zoster virus, opportunistic infections, and osteoporotic fractures, all unrelated to corticosteroid dose, Dr. Oomatia and his associates said.
This study was supported in part by the National Institutes of Health and the National Center for Research Resources. No potential financial conflicts of interest were reported.
Small-fiber neuropathy is one of the most common types of peripheral neuropathy affecting patients with systemic lupus erythematosus, but it isn’t even mentioned in the American College of Rheumatology neuropsychiatric case definitions of manifestations of the disorder, according to a retrospective analysis of cohort of 2,097 patients with SLE.
Other types of peripheral neuropathy, such as acute inflammatory demyelinating neuropathies (for example, Guillain-Barré syndrome), plexopathies, and mononeuritis multiplex, are well described in the ACR-NPSLE case definitions but occur much less frequently. This, combined with the fact that small-fiber neuropathies often present as "unorthodox" pain patterns, indicates that they are underdiagnosed, said Dr. Amin Oomatia of the University of Cambridge, England, and his coinvestigators at John Hopkins University, Baltimore.
Small-fiber neuropathies arise through mechanisms that are distinct from those of other neuropathies and require different diagnostic strategies to be properly identified. In particular, small-fiber neuropathies do not always conform to the "stocking-and-glove" pattern of pain that is typical of other neuropathies in SLE, so it is likely that many affected patients "may be regarded in routine clinical care as having a ‘nonorganic’ pain disorder.
"Our findings suggest that rheumatologists and other clinicians who confront SLE patients with seemingly improbable pain patterns should consider the diagnosis of a small-fiber neuropathy," the investigators wrote, especially since it may occur in the face of normal electrodiagnostic studies.
Dr. Oomatia and his colleagues based these conclusions on their retrospective study of one medical center’s 25-year experience treating 2,097 SLE patients – the Johns Hopkins Lupus Cohort. Using details in a database of patients’ electronic medical records, they identified 82 patients who had peripheral neuropathies related to SLE.
Only one patient had peripheral neuropathy attributable to Guillain-Barré syndrome, only one patient had a plexopathy, and only six patients had mononeuritis multiplex, demonstrating that these are very infrequent complications of SLE even though they are included in ACR case definitions.
In contrast, 14 patients (17% of those with peripheral neuropathy) had biopsy-proven small-fiber neuropathies, and most of them presented with "an entirely different and unorthodox pain distribution" characterized as patchy, asymmetric, or proximal.
In particular, nine patients had pain affecting the face, torso, and/or proximal extremities. Three had burning pain over their entire bodies, the investigators said (Arthritis Rheum. 2013 Dec. 10 [doi:10.1002/art.38302]).
In these cases, punch skin biopsy showed abnormalities that disproportionately affected the proximal thigh, "which is considered a surrogate indicator of proximal-most dorsal root ganglia neuronal cell loss," they wrote. In contrast, other patients who had the typical distal pattern of neuropathic pain showed decreased intraepidermal nerve-fiber densities in the distal leg, a surrogate indicator of distal-most axonal degeneration.
Another distinguishing feature of small-fiber neuropathy was its association with a history of herpes zoster virus, opportunistic infections, and osteoporotic fractures, all unrelated to corticosteroid dose, Dr. Oomatia and his associates said.
This study was supported in part by the National Institutes of Health and the National Center for Research Resources. No potential financial conflicts of interest were reported.
FROM ARTHRITIS AND RHEUMATISM
Major finding: A total of 14 patients, or 17% of 82 with peripheral neuropathies, had biopsy-proven small-fiber neuropathies and often presented with unorthodox patterns of pain.
Data source: A retrospective analysis of data regarding 2,097 consecutive patients with SLE registered in the Johns Hopkins Lupus Cohort during a 25-year period, including 82 who developed peripheral neuropathies related to the disease.
Disclosures: This study was supported in part by the National Institutes of Health and the National Center for Research Resources. No potential financial conflicts of interest were reported.
Maternal lupus doubled autism risk
SAN DIEGO – The risk of autism spectrum disorders is more than doubled among children born to mothers with systemic lupus erythematosus, according to the first-ever controlled study to address the question.
That being said, women with SLE can be reassured that despite this elevated relative risk, the absolute risk that their child will be diagnosed with an autism spectrum disorder (ASD) is low – less than 1 in 50 – Dr. Evelyne Vinet said at the annual meeting of the American College of Rheumatology.
The increased risk of ASD in children born to women with SLE documented in this large study was not mediated by in utero exposure to medications for SLE, including antimalarials, immunosuppressive agents, corticosteroids, and antidepressants. Use of those drugs in pregnancy wasn’t associated with any increased risk. In light of this, further research is warranted into a highly promising alternative hypothesis: that in utero exposure to SLE-related autoantibodies, such as anti-DNA and antiphospholipid antibodies, may play a causative role, said Dr. Vinet, a rheumatologist at McGill University, Montreal.
Children born to mothers with SLE also had an increased likelihood of being diagnosed with attention-deficit/hyperactivity disorder in this study. However, in contrast to the situation with ASD, the increased risk of ADHD appeared to result from exposure to medications in utero – specifically, antidepressants and possibly immunosuppressives – rather than to maternal SLE per se, she added.
Dr. Vinet presented an analysis of the OSLER (Offspring of Systemic Lupus Erythematosus Mothers Registry) database, the world’s largest cohort of children born to mothers with SLE. OSLER includes all women with a diagnosis of SLE hospitalized for childbirth in the province of Quebec since 1989. This study included 509 women who had 719 children after they had been diagnosed with SLE, as well as 5,824 controls matched for age and year of delivery along with their 8,493 children. The mean maternal age was 30.3 years. The children were followed out to a mean age of 9.1 years.
ASD was diagnosed in 1.4% of children born to mothers with SLE, compared with 0.6% of control children. The mean age at diagnosis of ASD was noticeably lower in the offspring of mothers with SLE: 3.8 years, compared with 5.7 years in the control children.
Women with SLE had higher rates of hypertension, asthma, and diabetes than did controls at the time of delivery. They also had higher rates of obstetric complications, including preterm birth, small for gestational age, and gestational diabetes. In a multivariate analysis adjusted for these variables, maternal SLE remained independently associated with a 2.3-fold increased risk of diagnosis of an ASD in offspring.
Complete and reliable records of prescription drug use during pregnancy were available only for the roughly 20% of mothers belonging to the provincial medication public assistance program. Of the 18 cases of ASD diagnosed in the children of 1,925 mothers covered by the program, only one occurred among the 155 offspring of SLE mothers; that child had been exposed to corticosteroids in utero. Of the 17 cases of ASD diagnosed in the control group, 16 had no exposures to the medications under scrutiny, and one involved in utero exposure to an anticonvulsant.
The OSLER database doesn’t include information about maternal autoantibody levels, but Dr. Vinet and her coinvestigators have made the collection of such data a top priority in light of recent studies in animal models of SLE by other investigators. Those studies showed that SLE-related autoantibodies, including n-methyl-d-aspartate receptor antibodies and antiphospholipid antibodies, as well as interleukin-6 and other cytokines, alter fetal brain development and induce behavioral anomalies suggestive of autism, such as withdrawal, in the mouse offspring.
In addition, French investigators recently reported that 3 of 36 children born to mothers with antiphospholipid syndrome developed ASD, although Dr. Vinet noted that this small but intriguing study lacked a control group (Semin. Arthritis Rheum. 2013 Aug. 1 [doi: 10.1016/j.semarthrit.2013.07.001]).
ADHD was diagnosed in 9.9% of the children of mothers with SLE in the OSLER database, compared with 6.1% of controls. It was diagnosed when the children were older: at a mean age of 12.5 years, compared with 7.8 years in the controls. In a multivariate analysis adjusted for in utero drug exposures, the association between ADHD and maternal SLE was no longer significant. However, in utero exposure to antidepressant medication, regardless of whether or not a mother had SLE, was associated with a hefty 3.7-fold increase in ADHD in the offspring. Because of the relatively small number of mothers on antidepressant medication, Dr. Vinet is now collaborating with investigators in other Canadian provinces having more comprehensive maternal medication exposure data to take a closer look at this tentative link between in utero drug exposure and ADHD.
This study of the OSLER database was funded by the Canadian Institutes of Health Research. Dr. Vinet reported having no financial conflicts.
SAN DIEGO – The risk of autism spectrum disorders is more than doubled among children born to mothers with systemic lupus erythematosus, according to the first-ever controlled study to address the question.
That being said, women with SLE can be reassured that despite this elevated relative risk, the absolute risk that their child will be diagnosed with an autism spectrum disorder (ASD) is low – less than 1 in 50 – Dr. Evelyne Vinet said at the annual meeting of the American College of Rheumatology.
The increased risk of ASD in children born to women with SLE documented in this large study was not mediated by in utero exposure to medications for SLE, including antimalarials, immunosuppressive agents, corticosteroids, and antidepressants. Use of those drugs in pregnancy wasn’t associated with any increased risk. In light of this, further research is warranted into a highly promising alternative hypothesis: that in utero exposure to SLE-related autoantibodies, such as anti-DNA and antiphospholipid antibodies, may play a causative role, said Dr. Vinet, a rheumatologist at McGill University, Montreal.
Children born to mothers with SLE also had an increased likelihood of being diagnosed with attention-deficit/hyperactivity disorder in this study. However, in contrast to the situation with ASD, the increased risk of ADHD appeared to result from exposure to medications in utero – specifically, antidepressants and possibly immunosuppressives – rather than to maternal SLE per se, she added.
Dr. Vinet presented an analysis of the OSLER (Offspring of Systemic Lupus Erythematosus Mothers Registry) database, the world’s largest cohort of children born to mothers with SLE. OSLER includes all women with a diagnosis of SLE hospitalized for childbirth in the province of Quebec since 1989. This study included 509 women who had 719 children after they had been diagnosed with SLE, as well as 5,824 controls matched for age and year of delivery along with their 8,493 children. The mean maternal age was 30.3 years. The children were followed out to a mean age of 9.1 years.
ASD was diagnosed in 1.4% of children born to mothers with SLE, compared with 0.6% of control children. The mean age at diagnosis of ASD was noticeably lower in the offspring of mothers with SLE: 3.8 years, compared with 5.7 years in the control children.
Women with SLE had higher rates of hypertension, asthma, and diabetes than did controls at the time of delivery. They also had higher rates of obstetric complications, including preterm birth, small for gestational age, and gestational diabetes. In a multivariate analysis adjusted for these variables, maternal SLE remained independently associated with a 2.3-fold increased risk of diagnosis of an ASD in offspring.
Complete and reliable records of prescription drug use during pregnancy were available only for the roughly 20% of mothers belonging to the provincial medication public assistance program. Of the 18 cases of ASD diagnosed in the children of 1,925 mothers covered by the program, only one occurred among the 155 offspring of SLE mothers; that child had been exposed to corticosteroids in utero. Of the 17 cases of ASD diagnosed in the control group, 16 had no exposures to the medications under scrutiny, and one involved in utero exposure to an anticonvulsant.
The OSLER database doesn’t include information about maternal autoantibody levels, but Dr. Vinet and her coinvestigators have made the collection of such data a top priority in light of recent studies in animal models of SLE by other investigators. Those studies showed that SLE-related autoantibodies, including n-methyl-d-aspartate receptor antibodies and antiphospholipid antibodies, as well as interleukin-6 and other cytokines, alter fetal brain development and induce behavioral anomalies suggestive of autism, such as withdrawal, in the mouse offspring.
In addition, French investigators recently reported that 3 of 36 children born to mothers with antiphospholipid syndrome developed ASD, although Dr. Vinet noted that this small but intriguing study lacked a control group (Semin. Arthritis Rheum. 2013 Aug. 1 [doi: 10.1016/j.semarthrit.2013.07.001]).
ADHD was diagnosed in 9.9% of the children of mothers with SLE in the OSLER database, compared with 6.1% of controls. It was diagnosed when the children were older: at a mean age of 12.5 years, compared with 7.8 years in the controls. In a multivariate analysis adjusted for in utero drug exposures, the association between ADHD and maternal SLE was no longer significant. However, in utero exposure to antidepressant medication, regardless of whether or not a mother had SLE, was associated with a hefty 3.7-fold increase in ADHD in the offspring. Because of the relatively small number of mothers on antidepressant medication, Dr. Vinet is now collaborating with investigators in other Canadian provinces having more comprehensive maternal medication exposure data to take a closer look at this tentative link between in utero drug exposure and ADHD.
This study of the OSLER database was funded by the Canadian Institutes of Health Research. Dr. Vinet reported having no financial conflicts.
SAN DIEGO – The risk of autism spectrum disorders is more than doubled among children born to mothers with systemic lupus erythematosus, according to the first-ever controlled study to address the question.
That being said, women with SLE can be reassured that despite this elevated relative risk, the absolute risk that their child will be diagnosed with an autism spectrum disorder (ASD) is low – less than 1 in 50 – Dr. Evelyne Vinet said at the annual meeting of the American College of Rheumatology.
The increased risk of ASD in children born to women with SLE documented in this large study was not mediated by in utero exposure to medications for SLE, including antimalarials, immunosuppressive agents, corticosteroids, and antidepressants. Use of those drugs in pregnancy wasn’t associated with any increased risk. In light of this, further research is warranted into a highly promising alternative hypothesis: that in utero exposure to SLE-related autoantibodies, such as anti-DNA and antiphospholipid antibodies, may play a causative role, said Dr. Vinet, a rheumatologist at McGill University, Montreal.
Children born to mothers with SLE also had an increased likelihood of being diagnosed with attention-deficit/hyperactivity disorder in this study. However, in contrast to the situation with ASD, the increased risk of ADHD appeared to result from exposure to medications in utero – specifically, antidepressants and possibly immunosuppressives – rather than to maternal SLE per se, she added.
Dr. Vinet presented an analysis of the OSLER (Offspring of Systemic Lupus Erythematosus Mothers Registry) database, the world’s largest cohort of children born to mothers with SLE. OSLER includes all women with a diagnosis of SLE hospitalized for childbirth in the province of Quebec since 1989. This study included 509 women who had 719 children after they had been diagnosed with SLE, as well as 5,824 controls matched for age and year of delivery along with their 8,493 children. The mean maternal age was 30.3 years. The children were followed out to a mean age of 9.1 years.
ASD was diagnosed in 1.4% of children born to mothers with SLE, compared with 0.6% of control children. The mean age at diagnosis of ASD was noticeably lower in the offspring of mothers with SLE: 3.8 years, compared with 5.7 years in the control children.
Women with SLE had higher rates of hypertension, asthma, and diabetes than did controls at the time of delivery. They also had higher rates of obstetric complications, including preterm birth, small for gestational age, and gestational diabetes. In a multivariate analysis adjusted for these variables, maternal SLE remained independently associated with a 2.3-fold increased risk of diagnosis of an ASD in offspring.
Complete and reliable records of prescription drug use during pregnancy were available only for the roughly 20% of mothers belonging to the provincial medication public assistance program. Of the 18 cases of ASD diagnosed in the children of 1,925 mothers covered by the program, only one occurred among the 155 offspring of SLE mothers; that child had been exposed to corticosteroids in utero. Of the 17 cases of ASD diagnosed in the control group, 16 had no exposures to the medications under scrutiny, and one involved in utero exposure to an anticonvulsant.
The OSLER database doesn’t include information about maternal autoantibody levels, but Dr. Vinet and her coinvestigators have made the collection of such data a top priority in light of recent studies in animal models of SLE by other investigators. Those studies showed that SLE-related autoantibodies, including n-methyl-d-aspartate receptor antibodies and antiphospholipid antibodies, as well as interleukin-6 and other cytokines, alter fetal brain development and induce behavioral anomalies suggestive of autism, such as withdrawal, in the mouse offspring.
In addition, French investigators recently reported that 3 of 36 children born to mothers with antiphospholipid syndrome developed ASD, although Dr. Vinet noted that this small but intriguing study lacked a control group (Semin. Arthritis Rheum. 2013 Aug. 1 [doi: 10.1016/j.semarthrit.2013.07.001]).
ADHD was diagnosed in 9.9% of the children of mothers with SLE in the OSLER database, compared with 6.1% of controls. It was diagnosed when the children were older: at a mean age of 12.5 years, compared with 7.8 years in the controls. In a multivariate analysis adjusted for in utero drug exposures, the association between ADHD and maternal SLE was no longer significant. However, in utero exposure to antidepressant medication, regardless of whether or not a mother had SLE, was associated with a hefty 3.7-fold increase in ADHD in the offspring. Because of the relatively small number of mothers on antidepressant medication, Dr. Vinet is now collaborating with investigators in other Canadian provinces having more comprehensive maternal medication exposure data to take a closer look at this tentative link between in utero drug exposure and ADHD.
This study of the OSLER database was funded by the Canadian Institutes of Health Research. Dr. Vinet reported having no financial conflicts.
AT THE ACR ANNUAL MEETING
Major finding: Children born to mothers with systemic lupus erythematosus had an adjusted 2.3-fold increased risk of being diagnosed with an autism spectrum disorder, compared with controls.
Data source: This study from the world’s largest cohort of children born to mothers with SLE included 509 affected mothers and their 719 children, as well as 5,824 matched control mothers and their 8,493 children.
Disclosures: This study of the OSLER database was funded by the Canadian Institutes of Health Research. Dr. Vinet reported having no financial conflicts.
Remission reinduction with rituximab a possibility for ANCA-associated vasculitis
SAN DIEGO – Retreatment of granulomatosis with polyangiitis or microscopic polyangiitis with rituximab may be safe and effective in reinducing remission, a prospective trial has shown.
"The vast majority of patients with ANCA [antineutrophil cytoplasmic antibody]–associated vasculitis are able to achieve disease remission initially," Dr. Eli Miloslavsky said at the annual meeting of the American College of Rheumatology. "However, there’s a high rate of flare, as frequent as 55% over the first 3 years. Therefore, it’s critical to determine the best remission agent in relapsing disease."
Dr. Miloslavsky presented data from 17 patients in the RAVE (Rituximab in ANCA–Associated Vasculitis) trial who received two courses of rituximab (RTX) and were followed for an average of 301 days. Patients with a severe flare were eligible to receive an open-label course of RTX between 6 and 18 months (375 mg/m2 once a week for 4 weeks). Severe flare was defined as having a Birmingham Vasculitis Activity Score for Wegener’s Granulomatosis (BVAS/WG) of greater than 3 or one major BVAS/WG item. Outcomes were complete remission (no disease activity and being off of steroids), complete response (no disease activity and taking 10 g of prednisone or less), remission (no disease activity regardless of the prednisone dose), limited flare (BVAS/WG of 3 or less), and severe flare (BVAS/WG of greater than 3 or one major disease activity item). At baseline, 82% of patients who received two courses of rituximab were proteinase 3 positive and 88% had granulomatosis with polyangiitis as the clinical diagnosis.
Of the 17 patients, 11 (65%) had relapsing disease at study entry. After receiving a second course of RTX, 15 patients (88%) achieved remission in an average of 2 months, 12 (71%) had at least a complete response in an average of 5 months, and 8 (47%) reached complete remission in an average of 6 months, reported Dr. Miloslavsky of the department of rheumatology at Massachusetts General Hospital, Boston.
At the 12-month time point, 13 patients (76%) had achieved complete responses and 8 (47%) had reached complete remission.
Four flares occurred during the study. "They were all limited and the time to flare was approximately 8 months after receiving RTX," he said.
Three severe adverse events occurred, including one death (a patient with diffuse alveolar hemorrhage who did not improve and died 7 weeks after the initial flare), one case of metastatic colon cancer, and one case of severe sinusitis.
Dr. Miloslavsky acknowledged certain limitations of the study, including the small sample size, the lack of a comparison group, the lack of long-term follow-up, and the limited generalizability to myeloperoxidase-ANCA–positive patients.
The trial was funded by the Immune Tolerance Network, which is supported by the National Institute of Allergy and Infectious Diseases. Partial funding was also derived from Genentech and Biogen Idec.
SAN DIEGO – Retreatment of granulomatosis with polyangiitis or microscopic polyangiitis with rituximab may be safe and effective in reinducing remission, a prospective trial has shown.
"The vast majority of patients with ANCA [antineutrophil cytoplasmic antibody]–associated vasculitis are able to achieve disease remission initially," Dr. Eli Miloslavsky said at the annual meeting of the American College of Rheumatology. "However, there’s a high rate of flare, as frequent as 55% over the first 3 years. Therefore, it’s critical to determine the best remission agent in relapsing disease."
Dr. Miloslavsky presented data from 17 patients in the RAVE (Rituximab in ANCA–Associated Vasculitis) trial who received two courses of rituximab (RTX) and were followed for an average of 301 days. Patients with a severe flare were eligible to receive an open-label course of RTX between 6 and 18 months (375 mg/m2 once a week for 4 weeks). Severe flare was defined as having a Birmingham Vasculitis Activity Score for Wegener’s Granulomatosis (BVAS/WG) of greater than 3 or one major BVAS/WG item. Outcomes were complete remission (no disease activity and being off of steroids), complete response (no disease activity and taking 10 g of prednisone or less), remission (no disease activity regardless of the prednisone dose), limited flare (BVAS/WG of 3 or less), and severe flare (BVAS/WG of greater than 3 or one major disease activity item). At baseline, 82% of patients who received two courses of rituximab were proteinase 3 positive and 88% had granulomatosis with polyangiitis as the clinical diagnosis.
Of the 17 patients, 11 (65%) had relapsing disease at study entry. After receiving a second course of RTX, 15 patients (88%) achieved remission in an average of 2 months, 12 (71%) had at least a complete response in an average of 5 months, and 8 (47%) reached complete remission in an average of 6 months, reported Dr. Miloslavsky of the department of rheumatology at Massachusetts General Hospital, Boston.
At the 12-month time point, 13 patients (76%) had achieved complete responses and 8 (47%) had reached complete remission.
Four flares occurred during the study. "They were all limited and the time to flare was approximately 8 months after receiving RTX," he said.
Three severe adverse events occurred, including one death (a patient with diffuse alveolar hemorrhage who did not improve and died 7 weeks after the initial flare), one case of metastatic colon cancer, and one case of severe sinusitis.
Dr. Miloslavsky acknowledged certain limitations of the study, including the small sample size, the lack of a comparison group, the lack of long-term follow-up, and the limited generalizability to myeloperoxidase-ANCA–positive patients.
The trial was funded by the Immune Tolerance Network, which is supported by the National Institute of Allergy and Infectious Diseases. Partial funding was also derived from Genentech and Biogen Idec.
SAN DIEGO – Retreatment of granulomatosis with polyangiitis or microscopic polyangiitis with rituximab may be safe and effective in reinducing remission, a prospective trial has shown.
"The vast majority of patients with ANCA [antineutrophil cytoplasmic antibody]–associated vasculitis are able to achieve disease remission initially," Dr. Eli Miloslavsky said at the annual meeting of the American College of Rheumatology. "However, there’s a high rate of flare, as frequent as 55% over the first 3 years. Therefore, it’s critical to determine the best remission agent in relapsing disease."
Dr. Miloslavsky presented data from 17 patients in the RAVE (Rituximab in ANCA–Associated Vasculitis) trial who received two courses of rituximab (RTX) and were followed for an average of 301 days. Patients with a severe flare were eligible to receive an open-label course of RTX between 6 and 18 months (375 mg/m2 once a week for 4 weeks). Severe flare was defined as having a Birmingham Vasculitis Activity Score for Wegener’s Granulomatosis (BVAS/WG) of greater than 3 or one major BVAS/WG item. Outcomes were complete remission (no disease activity and being off of steroids), complete response (no disease activity and taking 10 g of prednisone or less), remission (no disease activity regardless of the prednisone dose), limited flare (BVAS/WG of 3 or less), and severe flare (BVAS/WG of greater than 3 or one major disease activity item). At baseline, 82% of patients who received two courses of rituximab were proteinase 3 positive and 88% had granulomatosis with polyangiitis as the clinical diagnosis.
Of the 17 patients, 11 (65%) had relapsing disease at study entry. After receiving a second course of RTX, 15 patients (88%) achieved remission in an average of 2 months, 12 (71%) had at least a complete response in an average of 5 months, and 8 (47%) reached complete remission in an average of 6 months, reported Dr. Miloslavsky of the department of rheumatology at Massachusetts General Hospital, Boston.
At the 12-month time point, 13 patients (76%) had achieved complete responses and 8 (47%) had reached complete remission.
Four flares occurred during the study. "They were all limited and the time to flare was approximately 8 months after receiving RTX," he said.
Three severe adverse events occurred, including one death (a patient with diffuse alveolar hemorrhage who did not improve and died 7 weeks after the initial flare), one case of metastatic colon cancer, and one case of severe sinusitis.
Dr. Miloslavsky acknowledged certain limitations of the study, including the small sample size, the lack of a comparison group, the lack of long-term follow-up, and the limited generalizability to myeloperoxidase-ANCA–positive patients.
The trial was funded by the Immune Tolerance Network, which is supported by the National Institute of Allergy and Infectious Diseases. Partial funding was also derived from Genentech and Biogen Idec.
AT THE ACR ANNUAL MEETING
Major finding: Six months after receiving a second course of rituximab, 47% of patients achieved complete remission.
Data source: A study of17 patients in the rituximab in ANCA-associated vasculitis trial who received two courses of rituximab and were followed for an average of 301 days.
Disclosures: The trial was funded by the Immune Tolerance Network, which is supported by the National Institute of Allergy and Infectious Diseases. Partial funding was also derived from Genentech and Biogen Idec.
Antimalarials prove protective against long-term lupus damage
SAN DIEGO – Potentially modifiable risk factors for future irreversible organ damage in lupus patients include hypertension, the use of corticosteroids, and higher levels of inflammation early on, according to findings from the SLICC (Systemic Lupus International Collaborating Clinics) Inception Cohort Study.
In addition, the study identified the use of antimalarial drugs as the one significant protective factor against steady accrual of irreversible organ damage in lupus patients.
"These findings help us pave the way to consider whether, firstly, one could use damage as a primary endpoint in future clinical trials in lupus – somewhat akin to how the erosion score is used in rheumatoid arthritis – and secondly, the results suggest particular interventions that might be important in reducing the risk of damage over time," Dr. Ian N. Bruce said at the annual meeting of the American College of Rheumatology.
The study also identified several fixed and unmodifiable risk factors for irreversible damage in lupus patients: older age at diagnosis, male gender, and being black or white Americans, added Dr. Bruce, professor of rheumatology at the University of Manchester (U.K.) and chair of the SLICC research group.
The SLICC Inception Cohort Study involves 1,722 patients at 31 centers in 11 countries in North America, Europe, and Asia who enrolled within 15 months after being formally diagnosed with systemic lupus erythematosus based upon the 1997 ACR criteria. They averaged 35 years of age and had an average of 4.25 comprehensive annual follow-up visits during the study period.
Irreversible organ damage was assessed using the SLICC/ACR Damage Index, or SDI. At baseline, 35% of patients had at least one item of damage as indicated by an SDI score of 1 or more. Over time, damage rates slowly and steadily increased such that by 6 years of follow-up 51% of participants had an SDI of at least 1.
In a multivariate analysis, patients with an SDI score of 1 at baseline had a highly significant 37% reduction in the risk of increasing their score during follow-up if they were taking antimalarials, compared with those not taking antimalarials.
On the other hand, patients with a baseline SDI of 1 were 61% more likely to experience an increase in their damage score during follow-up if they had hypertension and 43% more likely to do so if they were on corticosteroids than if they weren’t. Moreover, their risk of going from an SDI of 1 to a higher SDI indicative of mounting damage increased by 10% for every 3-point increase on the SLE Disease Activity Index (SLEDAI).
Patients with a baseline SDI of 0 were 64% more likely to progress to a score of 1 or more during follow-up if they were taking corticosteroids and 71% more likely to do so if they were hypertensive. Their risk also increased by 17% for each 3-point increase in SLEDAI. Men had a 48% greater risk of going from an SDI of 0 to 1 or more than women. Asians were 40% less likely to develop irreversible damage.
Each 1-point increase in SDI score was associated with a 46% increased risk of mortality, as well as with poorer health-related quality of life, especially as reflected in SF-36 physical component scores.
Session chair Dr. Roberto Caricchio of Temple University, Philadelphia, called the SLICC study "very important work."
"It teaches us to be aggressive up-front with our lupus patients, which we often aren’t. We tend to spare ourselves because it’s a chronic disease, and we know we’ll see these patients for the next 20 years, so we try to spare them from certain therapies," said Dr. Caricchio.
Dr. Bruce concurred. "I think a concerted effort to switch the disease off in almost a treat-to-target way, getting people into remission, may well be very important with regard to avoiding long-term damage. If we could do that without using steroids, that would be ideal," he commented.
"SLICC is interested in the fact that most clinical trials in lupus to date have taken a very small subsection of the population, those with high disease activity, and used a particular biologic agent or new molecule to show that it improved disease activity. But actually the majority of people with lupus – around 60% have low-grade, grumbling disease and are on low-dose steroids. And those are the ones who accumulate damage. I think we need to have a paradigm shift in how we do clinical trials in lupus and think about doing lupus trials against a damage endpoint," the rheumatologist continued.
Power calculations based upon the SLICC Inception Cohort Study suggest such trials could be relatively modest in size, he added.
SLICC receives financial support from GlaxoSmithKline, Bristol-Myers Squibb, and Human Genome Sciences. Dr. Bruce reported receiving research funding from GlaxoSmithKline, Bristol-Myers Squibb, Roche, and UCB.
SAN DIEGO – Potentially modifiable risk factors for future irreversible organ damage in lupus patients include hypertension, the use of corticosteroids, and higher levels of inflammation early on, according to findings from the SLICC (Systemic Lupus International Collaborating Clinics) Inception Cohort Study.
In addition, the study identified the use of antimalarial drugs as the one significant protective factor against steady accrual of irreversible organ damage in lupus patients.
"These findings help us pave the way to consider whether, firstly, one could use damage as a primary endpoint in future clinical trials in lupus – somewhat akin to how the erosion score is used in rheumatoid arthritis – and secondly, the results suggest particular interventions that might be important in reducing the risk of damage over time," Dr. Ian N. Bruce said at the annual meeting of the American College of Rheumatology.
The study also identified several fixed and unmodifiable risk factors for irreversible damage in lupus patients: older age at diagnosis, male gender, and being black or white Americans, added Dr. Bruce, professor of rheumatology at the University of Manchester (U.K.) and chair of the SLICC research group.
The SLICC Inception Cohort Study involves 1,722 patients at 31 centers in 11 countries in North America, Europe, and Asia who enrolled within 15 months after being formally diagnosed with systemic lupus erythematosus based upon the 1997 ACR criteria. They averaged 35 years of age and had an average of 4.25 comprehensive annual follow-up visits during the study period.
Irreversible organ damage was assessed using the SLICC/ACR Damage Index, or SDI. At baseline, 35% of patients had at least one item of damage as indicated by an SDI score of 1 or more. Over time, damage rates slowly and steadily increased such that by 6 years of follow-up 51% of participants had an SDI of at least 1.
In a multivariate analysis, patients with an SDI score of 1 at baseline had a highly significant 37% reduction in the risk of increasing their score during follow-up if they were taking antimalarials, compared with those not taking antimalarials.
On the other hand, patients with a baseline SDI of 1 were 61% more likely to experience an increase in their damage score during follow-up if they had hypertension and 43% more likely to do so if they were on corticosteroids than if they weren’t. Moreover, their risk of going from an SDI of 1 to a higher SDI indicative of mounting damage increased by 10% for every 3-point increase on the SLE Disease Activity Index (SLEDAI).
Patients with a baseline SDI of 0 were 64% more likely to progress to a score of 1 or more during follow-up if they were taking corticosteroids and 71% more likely to do so if they were hypertensive. Their risk also increased by 17% for each 3-point increase in SLEDAI. Men had a 48% greater risk of going from an SDI of 0 to 1 or more than women. Asians were 40% less likely to develop irreversible damage.
Each 1-point increase in SDI score was associated with a 46% increased risk of mortality, as well as with poorer health-related quality of life, especially as reflected in SF-36 physical component scores.
Session chair Dr. Roberto Caricchio of Temple University, Philadelphia, called the SLICC study "very important work."
"It teaches us to be aggressive up-front with our lupus patients, which we often aren’t. We tend to spare ourselves because it’s a chronic disease, and we know we’ll see these patients for the next 20 years, so we try to spare them from certain therapies," said Dr. Caricchio.
Dr. Bruce concurred. "I think a concerted effort to switch the disease off in almost a treat-to-target way, getting people into remission, may well be very important with regard to avoiding long-term damage. If we could do that without using steroids, that would be ideal," he commented.
"SLICC is interested in the fact that most clinical trials in lupus to date have taken a very small subsection of the population, those with high disease activity, and used a particular biologic agent or new molecule to show that it improved disease activity. But actually the majority of people with lupus – around 60% have low-grade, grumbling disease and are on low-dose steroids. And those are the ones who accumulate damage. I think we need to have a paradigm shift in how we do clinical trials in lupus and think about doing lupus trials against a damage endpoint," the rheumatologist continued.
Power calculations based upon the SLICC Inception Cohort Study suggest such trials could be relatively modest in size, he added.
SLICC receives financial support from GlaxoSmithKline, Bristol-Myers Squibb, and Human Genome Sciences. Dr. Bruce reported receiving research funding from GlaxoSmithKline, Bristol-Myers Squibb, Roche, and UCB.
SAN DIEGO – Potentially modifiable risk factors for future irreversible organ damage in lupus patients include hypertension, the use of corticosteroids, and higher levels of inflammation early on, according to findings from the SLICC (Systemic Lupus International Collaborating Clinics) Inception Cohort Study.
In addition, the study identified the use of antimalarial drugs as the one significant protective factor against steady accrual of irreversible organ damage in lupus patients.
"These findings help us pave the way to consider whether, firstly, one could use damage as a primary endpoint in future clinical trials in lupus – somewhat akin to how the erosion score is used in rheumatoid arthritis – and secondly, the results suggest particular interventions that might be important in reducing the risk of damage over time," Dr. Ian N. Bruce said at the annual meeting of the American College of Rheumatology.
The study also identified several fixed and unmodifiable risk factors for irreversible damage in lupus patients: older age at diagnosis, male gender, and being black or white Americans, added Dr. Bruce, professor of rheumatology at the University of Manchester (U.K.) and chair of the SLICC research group.
The SLICC Inception Cohort Study involves 1,722 patients at 31 centers in 11 countries in North America, Europe, and Asia who enrolled within 15 months after being formally diagnosed with systemic lupus erythematosus based upon the 1997 ACR criteria. They averaged 35 years of age and had an average of 4.25 comprehensive annual follow-up visits during the study period.
Irreversible organ damage was assessed using the SLICC/ACR Damage Index, or SDI. At baseline, 35% of patients had at least one item of damage as indicated by an SDI score of 1 or more. Over time, damage rates slowly and steadily increased such that by 6 years of follow-up 51% of participants had an SDI of at least 1.
In a multivariate analysis, patients with an SDI score of 1 at baseline had a highly significant 37% reduction in the risk of increasing their score during follow-up if they were taking antimalarials, compared with those not taking antimalarials.
On the other hand, patients with a baseline SDI of 1 were 61% more likely to experience an increase in their damage score during follow-up if they had hypertension and 43% more likely to do so if they were on corticosteroids than if they weren’t. Moreover, their risk of going from an SDI of 1 to a higher SDI indicative of mounting damage increased by 10% for every 3-point increase on the SLE Disease Activity Index (SLEDAI).
Patients with a baseline SDI of 0 were 64% more likely to progress to a score of 1 or more during follow-up if they were taking corticosteroids and 71% more likely to do so if they were hypertensive. Their risk also increased by 17% for each 3-point increase in SLEDAI. Men had a 48% greater risk of going from an SDI of 0 to 1 or more than women. Asians were 40% less likely to develop irreversible damage.
Each 1-point increase in SDI score was associated with a 46% increased risk of mortality, as well as with poorer health-related quality of life, especially as reflected in SF-36 physical component scores.
Session chair Dr. Roberto Caricchio of Temple University, Philadelphia, called the SLICC study "very important work."
"It teaches us to be aggressive up-front with our lupus patients, which we often aren’t. We tend to spare ourselves because it’s a chronic disease, and we know we’ll see these patients for the next 20 years, so we try to spare them from certain therapies," said Dr. Caricchio.
Dr. Bruce concurred. "I think a concerted effort to switch the disease off in almost a treat-to-target way, getting people into remission, may well be very important with regard to avoiding long-term damage. If we could do that without using steroids, that would be ideal," he commented.
"SLICC is interested in the fact that most clinical trials in lupus to date have taken a very small subsection of the population, those with high disease activity, and used a particular biologic agent or new molecule to show that it improved disease activity. But actually the majority of people with lupus – around 60% have low-grade, grumbling disease and are on low-dose steroids. And those are the ones who accumulate damage. I think we need to have a paradigm shift in how we do clinical trials in lupus and think about doing lupus trials against a damage endpoint," the rheumatologist continued.
Power calculations based upon the SLICC Inception Cohort Study suggest such trials could be relatively modest in size, he added.
SLICC receives financial support from GlaxoSmithKline, Bristol-Myers Squibb, and Human Genome Sciences. Dr. Bruce reported receiving research funding from GlaxoSmithKline, Bristol-Myers Squibb, Roche, and UCB.
AT THE ACR ANNUAL MEETING
Major finding: Patients with recently diagnosed SLE and no significant organ damage at baseline were 71% more likely to develop irreversible organ damage during follow-up if they were hypertensive and 64% more likely to do so if they were taking corticosteroids. Antimalarial drugs showed a protective effect against accrual of damage.
Data source: The Systemic Lupus International Collaborating Clinics Inception Cohort Study involves 1,722 patients at 31 centers in 11 countries, all recruited within 15 months after diagnosis.
Disclosures: The study group receives funding from GlaxoSmithKline, Bristol-Myers Squibb, and Human Genome Sciences. The presenter receives research grants from GlaxoSmithKline, Bristol-Myers Squibb, and several other pharmaceutical companies.
Depression accounts for psoriatics’ increased MI risk
SAN DIEGO – Depression is an independent risk factor for acute myocardial infarction in patients with psoriasis or psoriatic arthritis, a large population-based cohort study indicates.
In this study of more than 10,000 British Columbians with psoriasis and/or psoriatic arthritis, the increased risk of MI was confined to the patient subset having comorbid depression, Lindsay C. Burns, Ph.D., reported at the annual meeting of the American College of Rheumatology.
"These data underscore the need to actively screen for depression among psoriasis and psoriatic arthritis patients and closely monitor cardiovascular health in this high-risk group to improve long-term survival," declared Dr. Burns of the University of British Columbia, Vancouver.
She and her coinvestigators mined the comprehensive health records available for 4.1 million adults through British Columbia’s universal medical insurance coverage system in order to identify all 10,041 patients who were diagnosed with psoriasis by a dermatologist or psoriatic arthritis by a rheumatologist during 1996-2006, and who at that time had no history of MI. The patients were matched by age, gender, and years of follow-up with 47,415 controls.
Acute MI occurred in 268 patients with psoriasis or psoriatic arthritis, for an incidence rate of 5.8 cases per 1,000 person-years. The incidence rate of physician-diagnosed depression was 3.4 per 1,000 person-years in the psoriatic group, with a 10-year prevalence of 21.6%.
In a multivariate regression analysis adjusted for comorbid conditions, socioeconomic status, health resource utilization, age, and gender, individuals with psoriasis or psoriatic arthritis were 26% more likely to be depressed than controls. Diagnosis of depression in psoriatic patients during the follow-up period increased their risk of having an MI by an adjusted 80% compared with psoriatic patients without the psychiatric diagnosis.
Psoriatic subjects without depression had a statistically nonsignificant 10% increased risk of MI compared with nonpsoriatic controls. In contrast, psoriatic patients with diagnosed depression had a 60% greater MI risk than controls, according to Dr. Burns.
The study was funded by the Arthritis Research Center of Canada. Dr. Burns reported having no financial conflicts of interest.
SAN DIEGO – Depression is an independent risk factor for acute myocardial infarction in patients with psoriasis or psoriatic arthritis, a large population-based cohort study indicates.
In this study of more than 10,000 British Columbians with psoriasis and/or psoriatic arthritis, the increased risk of MI was confined to the patient subset having comorbid depression, Lindsay C. Burns, Ph.D., reported at the annual meeting of the American College of Rheumatology.
"These data underscore the need to actively screen for depression among psoriasis and psoriatic arthritis patients and closely monitor cardiovascular health in this high-risk group to improve long-term survival," declared Dr. Burns of the University of British Columbia, Vancouver.
She and her coinvestigators mined the comprehensive health records available for 4.1 million adults through British Columbia’s universal medical insurance coverage system in order to identify all 10,041 patients who were diagnosed with psoriasis by a dermatologist or psoriatic arthritis by a rheumatologist during 1996-2006, and who at that time had no history of MI. The patients were matched by age, gender, and years of follow-up with 47,415 controls.
Acute MI occurred in 268 patients with psoriasis or psoriatic arthritis, for an incidence rate of 5.8 cases per 1,000 person-years. The incidence rate of physician-diagnosed depression was 3.4 per 1,000 person-years in the psoriatic group, with a 10-year prevalence of 21.6%.
In a multivariate regression analysis adjusted for comorbid conditions, socioeconomic status, health resource utilization, age, and gender, individuals with psoriasis or psoriatic arthritis were 26% more likely to be depressed than controls. Diagnosis of depression in psoriatic patients during the follow-up period increased their risk of having an MI by an adjusted 80% compared with psoriatic patients without the psychiatric diagnosis.
Psoriatic subjects without depression had a statistically nonsignificant 10% increased risk of MI compared with nonpsoriatic controls. In contrast, psoriatic patients with diagnosed depression had a 60% greater MI risk than controls, according to Dr. Burns.
The study was funded by the Arthritis Research Center of Canada. Dr. Burns reported having no financial conflicts of interest.
SAN DIEGO – Depression is an independent risk factor for acute myocardial infarction in patients with psoriasis or psoriatic arthritis, a large population-based cohort study indicates.
In this study of more than 10,000 British Columbians with psoriasis and/or psoriatic arthritis, the increased risk of MI was confined to the patient subset having comorbid depression, Lindsay C. Burns, Ph.D., reported at the annual meeting of the American College of Rheumatology.
"These data underscore the need to actively screen for depression among psoriasis and psoriatic arthritis patients and closely monitor cardiovascular health in this high-risk group to improve long-term survival," declared Dr. Burns of the University of British Columbia, Vancouver.
She and her coinvestigators mined the comprehensive health records available for 4.1 million adults through British Columbia’s universal medical insurance coverage system in order to identify all 10,041 patients who were diagnosed with psoriasis by a dermatologist or psoriatic arthritis by a rheumatologist during 1996-2006, and who at that time had no history of MI. The patients were matched by age, gender, and years of follow-up with 47,415 controls.
Acute MI occurred in 268 patients with psoriasis or psoriatic arthritis, for an incidence rate of 5.8 cases per 1,000 person-years. The incidence rate of physician-diagnosed depression was 3.4 per 1,000 person-years in the psoriatic group, with a 10-year prevalence of 21.6%.
In a multivariate regression analysis adjusted for comorbid conditions, socioeconomic status, health resource utilization, age, and gender, individuals with psoriasis or psoriatic arthritis were 26% more likely to be depressed than controls. Diagnosis of depression in psoriatic patients during the follow-up period increased their risk of having an MI by an adjusted 80% compared with psoriatic patients without the psychiatric diagnosis.
Psoriatic subjects without depression had a statistically nonsignificant 10% increased risk of MI compared with nonpsoriatic controls. In contrast, psoriatic patients with diagnosed depression had a 60% greater MI risk than controls, according to Dr. Burns.
The study was funded by the Arthritis Research Center of Canada. Dr. Burns reported having no financial conflicts of interest.
AT THE ACR ANNUAL MEETING
Major finding: Patients with psoriasis or psoriatic arthritis who developed comorbid depression had a highly significant 80% increased risk of acute MI during follow-up, compared with those without depression.
Data source: This was a population-based cohort study involving 10,041 adults diagnosed with psoriasis or psoriatic arthritis in British Columbia during 1996-2006 and more than 47,000 controls.
Disclosures: The study was funded by the Arthritis Research Center of Canada. Dr. Burns reported having no financial conflicts of interest.
Apremilast’s positive study results made it the talk of ACR 2013
SAN DIEGO – The novel oral phosphodiesterase-4 inhibitor apremilast cut an impressively wide swath through the annual meeting of the American College of Rheumatology on the strength of positive results in three separate pivotal phase III trials for psoriatic arthritis and a favorable phase II study in Behçet’s syndrome.
Dr. Alvin F. Wells reported that apremilast resulted in clinically meaningful improvement in psoriatic arthritis symptoms, physical function, and associated skin psoriasis at 16 weeks in the PALACE 4 trial. Moreover, the improvement remained durable through 52 weeks in the phase III clinical trial.
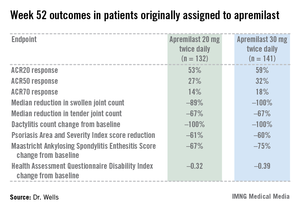
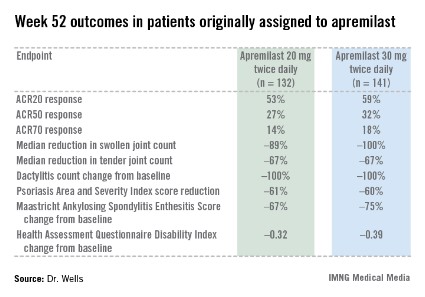
PALACE 4 compared apremilast against placebo as a first-line treatment in 527 patients with psoriatic arthritis not previously treated with a disease-modifying antirheumatic drug. This was a population with active disease: a mean baseline 11 swollen and 20 tender joints; a Health Assessment Questionnaire Disability Index score averaging 1.07; and a 50% prevalence of dactylitis, with a mean severity score of 2.0. Sixty-five percent of patients had enthesitis, with a median Maastricht Ankylosing Spondylitis Enthesitis Score (MASES) of 3.0. Participants had a mean 16-year history of psoriasis and a 3.4-year duration of psoriatic arthritis.
The primary study endpoint was attainment of an American College of Rheumatology 20% improvement (ACR20) response at 16 weeks. This was achieved in 29.2% of patients randomized to apremilast at 20 mg twice daily, 32.3% on 30 mg twice daily, and 16.9% on placebo. At week 16, patients on placebo were re-randomized to apremilast at 20 mg or 30 mg twice weekly. By week 52, an ACR20 response was achieved by 53% of patients in the apremilast 20 mg twice-daily group and 59% of those on the higher dose, according to Dr. Wells, director of the Rheumatology and Immunotherapy Center in Franklin, Wisc.
Apremilast also scored well on numerous secondary endpoints (see chart). For example, the swollen joint count decreased by a median of 89% over the course of 52 weeks in patients on apremilast at 20 mg twice daily.
"The way I like to think of that result is at least 50% of patients had an 89% improvement in their swollen joint count," he explained.
During the first 16 weeks of PALACE 4, there were fewer serious adverse events in apremilast-treated patients than with placebo-treated patients. The most frequent side effects of apremilast were nausea, diarrhea, and headache, affecting 16%, 12%, and 8%, respectively, of patients in the higher-dose arm. These adverse events were almost exclusively mild or moderate, they began within the first 2 weeks of treatment, and they resolved in 4 weeks despite continued treatment. Less than 2% of apremilast-treated patients dropped out of the study due to nausea or diarrhea through week 52.
PALACE 4 differed from the PALACE 2 and 3 trials, also presented at the ACR meeting, in that PALACE 2 and 3 involved only patients with psoriatic arthritis previously treated with biologic agents or other disease-modifying antirheumatic drugs. All three studies had the same design, and the results in terms of both efficacy and safety were consistent across the full clinical trial program. No clinically meaningful changes in laboratory values were seen in any of the PALACE trials, suggesting ongoing lab monitoring may not be necessary.
Also at the ACR annual meeting, Dr. Gülen Hatemi of Istanbul University, Turkey, in a reprise of her report several weeks earlier at the annual congress of the European Academy of Dermatology and Venereology, presented a phase II study showing apremilast to be highly effective in treating the oral ulcers that are the cardinal feature of Behçet’s syndrome.
Celgene filed for approval of apremilast for psoriatic arthritis with the Food and Drug Administration earlier this year and anticipates a decision in March 2014. The company also plans to apply to the European regulatory agency for the same indication before year’s end, and to petition the FDA for approval in psoriasis within the same time frame. A 500-patient phase III study of apremilast in ankylosing spondylitis, known as POSTURE, is well underway.
PALACE 4 was sponsored by Celgene. Dr. Wells reported receiving a research grant from the company.
SAN DIEGO – The novel oral phosphodiesterase-4 inhibitor apremilast cut an impressively wide swath through the annual meeting of the American College of Rheumatology on the strength of positive results in three separate pivotal phase III trials for psoriatic arthritis and a favorable phase II study in Behçet’s syndrome.
Dr. Alvin F. Wells reported that apremilast resulted in clinically meaningful improvement in psoriatic arthritis symptoms, physical function, and associated skin psoriasis at 16 weeks in the PALACE 4 trial. Moreover, the improvement remained durable through 52 weeks in the phase III clinical trial.

PALACE 4 compared apremilast against placebo as a first-line treatment in 527 patients with psoriatic arthritis not previously treated with a disease-modifying antirheumatic drug. This was a population with active disease: a mean baseline 11 swollen and 20 tender joints; a Health Assessment Questionnaire Disability Index score averaging 1.07; and a 50% prevalence of dactylitis, with a mean severity score of 2.0. Sixty-five percent of patients had enthesitis, with a median Maastricht Ankylosing Spondylitis Enthesitis Score (MASES) of 3.0. Participants had a mean 16-year history of psoriasis and a 3.4-year duration of psoriatic arthritis.
The primary study endpoint was attainment of an American College of Rheumatology 20% improvement (ACR20) response at 16 weeks. This was achieved in 29.2% of patients randomized to apremilast at 20 mg twice daily, 32.3% on 30 mg twice daily, and 16.9% on placebo. At week 16, patients on placebo were re-randomized to apremilast at 20 mg or 30 mg twice weekly. By week 52, an ACR20 response was achieved by 53% of patients in the apremilast 20 mg twice-daily group and 59% of those on the higher dose, according to Dr. Wells, director of the Rheumatology and Immunotherapy Center in Franklin, Wisc.
Apremilast also scored well on numerous secondary endpoints (see chart). For example, the swollen joint count decreased by a median of 89% over the course of 52 weeks in patients on apremilast at 20 mg twice daily.
"The way I like to think of that result is at least 50% of patients had an 89% improvement in their swollen joint count," he explained.
During the first 16 weeks of PALACE 4, there were fewer serious adverse events in apremilast-treated patients than with placebo-treated patients. The most frequent side effects of apremilast were nausea, diarrhea, and headache, affecting 16%, 12%, and 8%, respectively, of patients in the higher-dose arm. These adverse events were almost exclusively mild or moderate, they began within the first 2 weeks of treatment, and they resolved in 4 weeks despite continued treatment. Less than 2% of apremilast-treated patients dropped out of the study due to nausea or diarrhea through week 52.
PALACE 4 differed from the PALACE 2 and 3 trials, also presented at the ACR meeting, in that PALACE 2 and 3 involved only patients with psoriatic arthritis previously treated with biologic agents or other disease-modifying antirheumatic drugs. All three studies had the same design, and the results in terms of both efficacy and safety were consistent across the full clinical trial program. No clinically meaningful changes in laboratory values were seen in any of the PALACE trials, suggesting ongoing lab monitoring may not be necessary.
Also at the ACR annual meeting, Dr. Gülen Hatemi of Istanbul University, Turkey, in a reprise of her report several weeks earlier at the annual congress of the European Academy of Dermatology and Venereology, presented a phase II study showing apremilast to be highly effective in treating the oral ulcers that are the cardinal feature of Behçet’s syndrome.
Celgene filed for approval of apremilast for psoriatic arthritis with the Food and Drug Administration earlier this year and anticipates a decision in March 2014. The company also plans to apply to the European regulatory agency for the same indication before year’s end, and to petition the FDA for approval in psoriasis within the same time frame. A 500-patient phase III study of apremilast in ankylosing spondylitis, known as POSTURE, is well underway.
PALACE 4 was sponsored by Celgene. Dr. Wells reported receiving a research grant from the company.
SAN DIEGO – The novel oral phosphodiesterase-4 inhibitor apremilast cut an impressively wide swath through the annual meeting of the American College of Rheumatology on the strength of positive results in three separate pivotal phase III trials for psoriatic arthritis and a favorable phase II study in Behçet’s syndrome.
Dr. Alvin F. Wells reported that apremilast resulted in clinically meaningful improvement in psoriatic arthritis symptoms, physical function, and associated skin psoriasis at 16 weeks in the PALACE 4 trial. Moreover, the improvement remained durable through 52 weeks in the phase III clinical trial.

PALACE 4 compared apremilast against placebo as a first-line treatment in 527 patients with psoriatic arthritis not previously treated with a disease-modifying antirheumatic drug. This was a population with active disease: a mean baseline 11 swollen and 20 tender joints; a Health Assessment Questionnaire Disability Index score averaging 1.07; and a 50% prevalence of dactylitis, with a mean severity score of 2.0. Sixty-five percent of patients had enthesitis, with a median Maastricht Ankylosing Spondylitis Enthesitis Score (MASES) of 3.0. Participants had a mean 16-year history of psoriasis and a 3.4-year duration of psoriatic arthritis.
The primary study endpoint was attainment of an American College of Rheumatology 20% improvement (ACR20) response at 16 weeks. This was achieved in 29.2% of patients randomized to apremilast at 20 mg twice daily, 32.3% on 30 mg twice daily, and 16.9% on placebo. At week 16, patients on placebo were re-randomized to apremilast at 20 mg or 30 mg twice weekly. By week 52, an ACR20 response was achieved by 53% of patients in the apremilast 20 mg twice-daily group and 59% of those on the higher dose, according to Dr. Wells, director of the Rheumatology and Immunotherapy Center in Franklin, Wisc.
Apremilast also scored well on numerous secondary endpoints (see chart). For example, the swollen joint count decreased by a median of 89% over the course of 52 weeks in patients on apremilast at 20 mg twice daily.
"The way I like to think of that result is at least 50% of patients had an 89% improvement in their swollen joint count," he explained.
During the first 16 weeks of PALACE 4, there were fewer serious adverse events in apremilast-treated patients than with placebo-treated patients. The most frequent side effects of apremilast were nausea, diarrhea, and headache, affecting 16%, 12%, and 8%, respectively, of patients in the higher-dose arm. These adverse events were almost exclusively mild or moderate, they began within the first 2 weeks of treatment, and they resolved in 4 weeks despite continued treatment. Less than 2% of apremilast-treated patients dropped out of the study due to nausea or diarrhea through week 52.
PALACE 4 differed from the PALACE 2 and 3 trials, also presented at the ACR meeting, in that PALACE 2 and 3 involved only patients with psoriatic arthritis previously treated with biologic agents or other disease-modifying antirheumatic drugs. All three studies had the same design, and the results in terms of both efficacy and safety were consistent across the full clinical trial program. No clinically meaningful changes in laboratory values were seen in any of the PALACE trials, suggesting ongoing lab monitoring may not be necessary.
Also at the ACR annual meeting, Dr. Gülen Hatemi of Istanbul University, Turkey, in a reprise of her report several weeks earlier at the annual congress of the European Academy of Dermatology and Venereology, presented a phase II study showing apremilast to be highly effective in treating the oral ulcers that are the cardinal feature of Behçet’s syndrome.
Celgene filed for approval of apremilast for psoriatic arthritis with the Food and Drug Administration earlier this year and anticipates a decision in March 2014. The company also plans to apply to the European regulatory agency for the same indication before year’s end, and to petition the FDA for approval in psoriasis within the same time frame. A 500-patient phase III study of apremilast in ankylosing spondylitis, known as POSTURE, is well underway.
PALACE 4 was sponsored by Celgene. Dr. Wells reported receiving a research grant from the company.
AT THE ACR ANNUAL MEETING
Major finding: After 16 weeks of treatment, the investigational drug apremilast achieved an ACR20 response of 29.2% at 20 mg twice daily as first-line therapy in patients with psoriatic arthritis and 32.3% at 30 mg twice daily, whereas placebo-treated controls achieved a rate of 16.9%.
Data source: The PALACE 4 study was a pivotal phase III, randomized, placebo-controlled trial including 527 patients with active psoriatic arthritis.
Disclosures: PALACE 4 was sponsored by Celgene. Dr. Wells reported receiving a research grant from the company.
Cyclophosphamide and rituximab combo reduced severe lupus flares
SAN DIEGO – The combination of intravenous cyclophosphamide and rituximab shows promise for the reduction of lupus flares, both renal and nonrenal, in patients with severe systemic lupus erythematosus, according to Dr. Ali Shahzad.
Moreover, this benefit did not come at the cost of a significant increase in infections, as compared with intravenous cyclophosphamide monotherapy, Dr. Shahzad reported at the annual meeting of the American College of Rheumatology.
He reported on 43 patients with severe, recurrent SLE. Thirty-one were placed on intravenous cyclophosphamide monotherapy administered according to the National Institutes of Health standard protocol for the treatment of lupus nephritis. The other 12 got cyclophosphamide plus two 1,000-mg doses of rituximab (Rituxan) given 15 days apart. The combination regimen was given at the physician’s discretion for recalcitrant or recurrent flares of lupus nephritis in 10 of 12 cases, and for treatment-resistant CNS lupus or other extrarenal lupus in the other 2. Eight of the 12 recipients of combination therapy had previously been treated with intravenous cyclophosphamide, in most cases for lupus nephritis.
In the combination therapy group, the median duration of follow-up prior to dual therapy was 36 months, with an additional 21 months of follow-up after receiving the combination. Prior to combination therapy, these 12 patients collectively had 38 lupus flares, 26 of which featured renal involvement. In contrast, post treatment they developed just 13 flares, only 3 of which were renal, according to Dr. Shahzad of the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Bethesda, Md.
The 43 SLE patients had a total of 15 bacterial, 6 fungal, and 18 viral infections. And although there was a consistent trend toward higher rates in the combination therapy group, the differences fell far short of statistical significance.
Dr. Shahzad acknowledged the small sample size and retrospective design as important study limitations. However, based upon these encouraging, albeit preliminary, study findings, he and his NIH colleagues said they are planning a prospective clinical trial examining the efficacy and tolerability of the intravenous cyclophosphamide/rituximab combination in patients with severe, recurrent SLE.
Dr. Shahzad reported having no financial conflicts regarding this NIH-sponsored study.
SAN DIEGO – The combination of intravenous cyclophosphamide and rituximab shows promise for the reduction of lupus flares, both renal and nonrenal, in patients with severe systemic lupus erythematosus, according to Dr. Ali Shahzad.
Moreover, this benefit did not come at the cost of a significant increase in infections, as compared with intravenous cyclophosphamide monotherapy, Dr. Shahzad reported at the annual meeting of the American College of Rheumatology.
He reported on 43 patients with severe, recurrent SLE. Thirty-one were placed on intravenous cyclophosphamide monotherapy administered according to the National Institutes of Health standard protocol for the treatment of lupus nephritis. The other 12 got cyclophosphamide plus two 1,000-mg doses of rituximab (Rituxan) given 15 days apart. The combination regimen was given at the physician’s discretion for recalcitrant or recurrent flares of lupus nephritis in 10 of 12 cases, and for treatment-resistant CNS lupus or other extrarenal lupus in the other 2. Eight of the 12 recipients of combination therapy had previously been treated with intravenous cyclophosphamide, in most cases for lupus nephritis.
In the combination therapy group, the median duration of follow-up prior to dual therapy was 36 months, with an additional 21 months of follow-up after receiving the combination. Prior to combination therapy, these 12 patients collectively had 38 lupus flares, 26 of which featured renal involvement. In contrast, post treatment they developed just 13 flares, only 3 of which were renal, according to Dr. Shahzad of the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Bethesda, Md.
The 43 SLE patients had a total of 15 bacterial, 6 fungal, and 18 viral infections. And although there was a consistent trend toward higher rates in the combination therapy group, the differences fell far short of statistical significance.
Dr. Shahzad acknowledged the small sample size and retrospective design as important study limitations. However, based upon these encouraging, albeit preliminary, study findings, he and his NIH colleagues said they are planning a prospective clinical trial examining the efficacy and tolerability of the intravenous cyclophosphamide/rituximab combination in patients with severe, recurrent SLE.
Dr. Shahzad reported having no financial conflicts regarding this NIH-sponsored study.
SAN DIEGO – The combination of intravenous cyclophosphamide and rituximab shows promise for the reduction of lupus flares, both renal and nonrenal, in patients with severe systemic lupus erythematosus, according to Dr. Ali Shahzad.
Moreover, this benefit did not come at the cost of a significant increase in infections, as compared with intravenous cyclophosphamide monotherapy, Dr. Shahzad reported at the annual meeting of the American College of Rheumatology.
He reported on 43 patients with severe, recurrent SLE. Thirty-one were placed on intravenous cyclophosphamide monotherapy administered according to the National Institutes of Health standard protocol for the treatment of lupus nephritis. The other 12 got cyclophosphamide plus two 1,000-mg doses of rituximab (Rituxan) given 15 days apart. The combination regimen was given at the physician’s discretion for recalcitrant or recurrent flares of lupus nephritis in 10 of 12 cases, and for treatment-resistant CNS lupus or other extrarenal lupus in the other 2. Eight of the 12 recipients of combination therapy had previously been treated with intravenous cyclophosphamide, in most cases for lupus nephritis.
In the combination therapy group, the median duration of follow-up prior to dual therapy was 36 months, with an additional 21 months of follow-up after receiving the combination. Prior to combination therapy, these 12 patients collectively had 38 lupus flares, 26 of which featured renal involvement. In contrast, post treatment they developed just 13 flares, only 3 of which were renal, according to Dr. Shahzad of the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Bethesda, Md.
The 43 SLE patients had a total of 15 bacterial, 6 fungal, and 18 viral infections. And although there was a consistent trend toward higher rates in the combination therapy group, the differences fell far short of statistical significance.
Dr. Shahzad acknowledged the small sample size and retrospective design as important study limitations. However, based upon these encouraging, albeit preliminary, study findings, he and his NIH colleagues said they are planning a prospective clinical trial examining the efficacy and tolerability of the intravenous cyclophosphamide/rituximab combination in patients with severe, recurrent SLE.
Dr. Shahzad reported having no financial conflicts regarding this NIH-sponsored study.
AT THE ACR ANNUAL MEETING
Major finding: Patients with severe recurrent systemic lupus erythematosus collectively had 38 lupus flares, including 26 with renal involvement, during a median 36-month period before undergoing combination therapy with intravenous cyclophosphamide and rituximab; they had 13 flares, only 3 of which featured renal involvement, during 21 months afterward.
Data source: A retrospective study including 43 patients with severe recurrent SLE, 31 of whom were placed on intravenous cyclophosphamide monotherapy, while 12 received combination therapy with cyclophosphamide plus rituximab.
Disclosures: This study was sponsored by the National Institutes of Health. The presenter, an employee of the National Institute of Arthritis and Musculoskeletal and Skin Diseases, reported having no relevant financial conflicts.
Psoriasis drug research aimed at severe disease
ISTANBUL, TURKEY – The current intensive period of new drug development in psoriasis is cause for enthusiastic celebration by patients and physicians alike. That is, except for one glaring research gap.
Almost all of the promising drugs now in phase-II or -III clinical trials are systemic agents aimed at the minority of psoriasis patients with more severe disease. There’s not nearly as much in the pipeline for the many patients with mild psoriasis who are most appropriately treated with topical therapy, Dr. Hervé Bachelez observed at the annual congress of the European Academy of Dermatology and Venereology.
Indeed, he identified only two promising topical agents advancing through the developmental pipeline: Pfizer’s tofacitinib, a small-molecule Janus kinase inhibitor under study in both topical and oral formulations, and Anacor’s AN2728, a boron-containing molecule that inhibits phosphodiesterase-4 (PDE-4).
Industry’s lack of enthusiasm for developing new topical agents is difficult to understand, given that 84% of all U.S. prescriptions written for psoriasis in 2009 were for topical agents, according to IMS. Moreover, patients clearly believe there is a major unmet need for new and better topicals: a recent survey of 2,151 European psoriasis patients and their dermatologists found that only 45% of patients on topical therapy were satisfied with their treatment. And a mere 35% of dermatologists rated current topical treatments as satisfactory (J. Dermatolog. Treat. 2013;24:193-8).
The great bulk of the action in psoriasis drug development now is focused on injectable biologics and oral small molecules. Among them are three interleukin-17 inhibitors: Novartis’ secukinumab, Amgen’s brodalumab, and Eli Lilly’s ixekizumab. Celgene is developing apremilast as an oral PDE-4 inhibitor. Merck and Janssen Biotech are developing MK-3222 and CNTO 1959, respectively, as interleukin-23 inhibitors.
Dr. Bachelez, professor of dermatology and head of the inflammatory skin diseases unit at Saint Louis University Hospital, Paris, reported serving on the advisory boards of 10 pharmaceutical companies.
ISTANBUL, TURKEY – The current intensive period of new drug development in psoriasis is cause for enthusiastic celebration by patients and physicians alike. That is, except for one glaring research gap.
Almost all of the promising drugs now in phase-II or -III clinical trials are systemic agents aimed at the minority of psoriasis patients with more severe disease. There’s not nearly as much in the pipeline for the many patients with mild psoriasis who are most appropriately treated with topical therapy, Dr. Hervé Bachelez observed at the annual congress of the European Academy of Dermatology and Venereology.
Indeed, he identified only two promising topical agents advancing through the developmental pipeline: Pfizer’s tofacitinib, a small-molecule Janus kinase inhibitor under study in both topical and oral formulations, and Anacor’s AN2728, a boron-containing molecule that inhibits phosphodiesterase-4 (PDE-4).
Industry’s lack of enthusiasm for developing new topical agents is difficult to understand, given that 84% of all U.S. prescriptions written for psoriasis in 2009 were for topical agents, according to IMS. Moreover, patients clearly believe there is a major unmet need for new and better topicals: a recent survey of 2,151 European psoriasis patients and their dermatologists found that only 45% of patients on topical therapy were satisfied with their treatment. And a mere 35% of dermatologists rated current topical treatments as satisfactory (J. Dermatolog. Treat. 2013;24:193-8).
The great bulk of the action in psoriasis drug development now is focused on injectable biologics and oral small molecules. Among them are three interleukin-17 inhibitors: Novartis’ secukinumab, Amgen’s brodalumab, and Eli Lilly’s ixekizumab. Celgene is developing apremilast as an oral PDE-4 inhibitor. Merck and Janssen Biotech are developing MK-3222 and CNTO 1959, respectively, as interleukin-23 inhibitors.
Dr. Bachelez, professor of dermatology and head of the inflammatory skin diseases unit at Saint Louis University Hospital, Paris, reported serving on the advisory boards of 10 pharmaceutical companies.
ISTANBUL, TURKEY – The current intensive period of new drug development in psoriasis is cause for enthusiastic celebration by patients and physicians alike. That is, except for one glaring research gap.
Almost all of the promising drugs now in phase-II or -III clinical trials are systemic agents aimed at the minority of psoriasis patients with more severe disease. There’s not nearly as much in the pipeline for the many patients with mild psoriasis who are most appropriately treated with topical therapy, Dr. Hervé Bachelez observed at the annual congress of the European Academy of Dermatology and Venereology.
Indeed, he identified only two promising topical agents advancing through the developmental pipeline: Pfizer’s tofacitinib, a small-molecule Janus kinase inhibitor under study in both topical and oral formulations, and Anacor’s AN2728, a boron-containing molecule that inhibits phosphodiesterase-4 (PDE-4).
Industry’s lack of enthusiasm for developing new topical agents is difficult to understand, given that 84% of all U.S. prescriptions written for psoriasis in 2009 were for topical agents, according to IMS. Moreover, patients clearly believe there is a major unmet need for new and better topicals: a recent survey of 2,151 European psoriasis patients and their dermatologists found that only 45% of patients on topical therapy were satisfied with their treatment. And a mere 35% of dermatologists rated current topical treatments as satisfactory (J. Dermatolog. Treat. 2013;24:193-8).
The great bulk of the action in psoriasis drug development now is focused on injectable biologics and oral small molecules. Among them are three interleukin-17 inhibitors: Novartis’ secukinumab, Amgen’s brodalumab, and Eli Lilly’s ixekizumab. Celgene is developing apremilast as an oral PDE-4 inhibitor. Merck and Janssen Biotech are developing MK-3222 and CNTO 1959, respectively, as interleukin-23 inhibitors.
Dr. Bachelez, professor of dermatology and head of the inflammatory skin diseases unit at Saint Louis University Hospital, Paris, reported serving on the advisory boards of 10 pharmaceutical companies.
EXPERT ANALYSIS FROM THE EADV CONGRESS
Abatacept may reduce maintenance immunosuppression need in lupus nephritis
ATLANTA – Adding abatacept to low-dose intravenous cyclophosphamide did not improve the rate of complete renal response at 1 year, compared with IV cyclophosphamide alone, in patients with class III or IV lupus nephritis in the randomized, controlled ACCESS Trial.
The phase II findings suggested, however, that abatacept reduced the need for maintenance immunosuppression, and they also demonstrated for the first time that low-dose IV cyclophosphamide may have applicability in racially and ethnically diverse patients with lupus nephritis, Dr. Brad H. Rovin reported at Kidney Week 2013.
The overall complete renal response rates were similar at 33% and 31%, respectively, in 134 patients with class III or IV lupus nephritis who were enrolled in the trial, treated with a steroid taper and 500 mg of IV cyclophosphamide every 2 weeks for 6 weeks followed by 2 mg/kg of azathioprine daily. Patients then were randomized to receive abatacept or placebo at week 0, 2, and 4 and then monthly.
Complete plus partial responses occurred in 59% of patients in each arm, Dr. Rovin said at the conference, which was sponsored by the American Society of Nephrology.
Patients in the study had a urine protein-to-creatinine ratio (UPCR) greater than 1. Complete renal response was defined as a UPCR of less than 0.5, serum creatinine at normal or within 25% of baseline, and prednisone dose tapered to 10 mg daily or less. African Americans comprised 39% of the cohort, and Hispanic/Mestizo patients comprised 40%.
Among the African American patients, 33% in the treatment group, compared with 16% in the placebo group, achieved complete renal response. This difference was not statistically significant.
This finding is important, because while low-dose cyclophosphamide – the "Euro-Lupus regimen" – has been shown to be as effective as standard and more toxic higher dosing, studies had primarily included a European Caucasian population. It was unclear whether the low-dose regimen would be as effective in a multiracial and multiethnic population, said Dr. Rovin of Ohio State University Medical Center.
No differences were seen between the treatment and control groups with respect to anti-dsDNA or complement levels and/or the frequency of serious or infectious adverse events or study withdrawals.
Of note, azathioprine was stopped in treatment group patients who achieved complete renal response – but not control group patients who achieved complete renal response. These patients were followed to 52 weeks. At that time, 50% and 62% of the treatment group and control group patients, respectively, who were complete responders at 24 weeks still met the criteria for complete response, Dr. Rovin said.
While abatacept did not increase complete response rates, the treatment was associated with maintenance of complete renal response to 1 year despite discontinuation of maintenance immunosuppression in the treated patients and continued azathioprine treatment in the control group. This suggests that abatacept may reduce the need for maintenance immunosuppression, he said.
While it was disappointing that the addition of abatacept to background induction therapy with low-dose IV cyclophosphamide and corticosteroids did not improve the complete renal response rate in this study – despite murine models suggesting that the use of abatacept and IV cyclophosphamide act synergistically to arrest lupus nephritis progression – the findings offer two very important lessons for clinical practice, he said.
"We can take the low-dose cyclophosphamide regimen and potentially use that with good results in our [multiethnic, multiracial] patient population that we see here in this country, and I think that’s a huge step forward. It gives us another option to use besides mycophenolate mofetil and high-dose cyclophosphamide," he said, adding that the low-dose IV cyclophosphamide regimen is well tolerated and delivers a total dose of only 3 g of the drug – a dose that generally does not predispose to premature ovarian failure.
This is particularly important, because women of childbearing years comprise a majority of the patient population with lupus nephritis, he explained.
"We know from the Euro-Lupus trial that the low-dose regimen, over 10 years, provided the same maintenance of renal function as the high-dose regimen, so we have very good long-term follow-up. We don’t quite have that yet with mycophenolate mofetil, so I think that for those of us who are very frequent users of cyclophosphamide, this is a very good alternative to the high-dose, highly toxic regimen that has for so long been the standard of care," he said.
This study was funded by the National Institute of Allergy and Infectious Diseases via the Immune Tolerance Network. Bristol Myers Squibb provided the study drug. Dr. Rovin reported ties to Genentech, Questcor, TEVA, Biogen-IDEC, HGS, Johnson & Johnson, Roche, BMS, and TG Therapeutics.
ATLANTA – Adding abatacept to low-dose intravenous cyclophosphamide did not improve the rate of complete renal response at 1 year, compared with IV cyclophosphamide alone, in patients with class III or IV lupus nephritis in the randomized, controlled ACCESS Trial.
The phase II findings suggested, however, that abatacept reduced the need for maintenance immunosuppression, and they also demonstrated for the first time that low-dose IV cyclophosphamide may have applicability in racially and ethnically diverse patients with lupus nephritis, Dr. Brad H. Rovin reported at Kidney Week 2013.
The overall complete renal response rates were similar at 33% and 31%, respectively, in 134 patients with class III or IV lupus nephritis who were enrolled in the trial, treated with a steroid taper and 500 mg of IV cyclophosphamide every 2 weeks for 6 weeks followed by 2 mg/kg of azathioprine daily. Patients then were randomized to receive abatacept or placebo at week 0, 2, and 4 and then monthly.
Complete plus partial responses occurred in 59% of patients in each arm, Dr. Rovin said at the conference, which was sponsored by the American Society of Nephrology.
Patients in the study had a urine protein-to-creatinine ratio (UPCR) greater than 1. Complete renal response was defined as a UPCR of less than 0.5, serum creatinine at normal or within 25% of baseline, and prednisone dose tapered to 10 mg daily or less. African Americans comprised 39% of the cohort, and Hispanic/Mestizo patients comprised 40%.
Among the African American patients, 33% in the treatment group, compared with 16% in the placebo group, achieved complete renal response. This difference was not statistically significant.
This finding is important, because while low-dose cyclophosphamide – the "Euro-Lupus regimen" – has been shown to be as effective as standard and more toxic higher dosing, studies had primarily included a European Caucasian population. It was unclear whether the low-dose regimen would be as effective in a multiracial and multiethnic population, said Dr. Rovin of Ohio State University Medical Center.
No differences were seen between the treatment and control groups with respect to anti-dsDNA or complement levels and/or the frequency of serious or infectious adverse events or study withdrawals.
Of note, azathioprine was stopped in treatment group patients who achieved complete renal response – but not control group patients who achieved complete renal response. These patients were followed to 52 weeks. At that time, 50% and 62% of the treatment group and control group patients, respectively, who were complete responders at 24 weeks still met the criteria for complete response, Dr. Rovin said.
While abatacept did not increase complete response rates, the treatment was associated with maintenance of complete renal response to 1 year despite discontinuation of maintenance immunosuppression in the treated patients and continued azathioprine treatment in the control group. This suggests that abatacept may reduce the need for maintenance immunosuppression, he said.
While it was disappointing that the addition of abatacept to background induction therapy with low-dose IV cyclophosphamide and corticosteroids did not improve the complete renal response rate in this study – despite murine models suggesting that the use of abatacept and IV cyclophosphamide act synergistically to arrest lupus nephritis progression – the findings offer two very important lessons for clinical practice, he said.
"We can take the low-dose cyclophosphamide regimen and potentially use that with good results in our [multiethnic, multiracial] patient population that we see here in this country, and I think that’s a huge step forward. It gives us another option to use besides mycophenolate mofetil and high-dose cyclophosphamide," he said, adding that the low-dose IV cyclophosphamide regimen is well tolerated and delivers a total dose of only 3 g of the drug – a dose that generally does not predispose to premature ovarian failure.
This is particularly important, because women of childbearing years comprise a majority of the patient population with lupus nephritis, he explained.
"We know from the Euro-Lupus trial that the low-dose regimen, over 10 years, provided the same maintenance of renal function as the high-dose regimen, so we have very good long-term follow-up. We don’t quite have that yet with mycophenolate mofetil, so I think that for those of us who are very frequent users of cyclophosphamide, this is a very good alternative to the high-dose, highly toxic regimen that has for so long been the standard of care," he said.
This study was funded by the National Institute of Allergy and Infectious Diseases via the Immune Tolerance Network. Bristol Myers Squibb provided the study drug. Dr. Rovin reported ties to Genentech, Questcor, TEVA, Biogen-IDEC, HGS, Johnson & Johnson, Roche, BMS, and TG Therapeutics.
ATLANTA – Adding abatacept to low-dose intravenous cyclophosphamide did not improve the rate of complete renal response at 1 year, compared with IV cyclophosphamide alone, in patients with class III or IV lupus nephritis in the randomized, controlled ACCESS Trial.
The phase II findings suggested, however, that abatacept reduced the need for maintenance immunosuppression, and they also demonstrated for the first time that low-dose IV cyclophosphamide may have applicability in racially and ethnically diverse patients with lupus nephritis, Dr. Brad H. Rovin reported at Kidney Week 2013.
The overall complete renal response rates were similar at 33% and 31%, respectively, in 134 patients with class III or IV lupus nephritis who were enrolled in the trial, treated with a steroid taper and 500 mg of IV cyclophosphamide every 2 weeks for 6 weeks followed by 2 mg/kg of azathioprine daily. Patients then were randomized to receive abatacept or placebo at week 0, 2, and 4 and then monthly.
Complete plus partial responses occurred in 59% of patients in each arm, Dr. Rovin said at the conference, which was sponsored by the American Society of Nephrology.
Patients in the study had a urine protein-to-creatinine ratio (UPCR) greater than 1. Complete renal response was defined as a UPCR of less than 0.5, serum creatinine at normal or within 25% of baseline, and prednisone dose tapered to 10 mg daily or less. African Americans comprised 39% of the cohort, and Hispanic/Mestizo patients comprised 40%.
Among the African American patients, 33% in the treatment group, compared with 16% in the placebo group, achieved complete renal response. This difference was not statistically significant.
This finding is important, because while low-dose cyclophosphamide – the "Euro-Lupus regimen" – has been shown to be as effective as standard and more toxic higher dosing, studies had primarily included a European Caucasian population. It was unclear whether the low-dose regimen would be as effective in a multiracial and multiethnic population, said Dr. Rovin of Ohio State University Medical Center.
No differences were seen between the treatment and control groups with respect to anti-dsDNA or complement levels and/or the frequency of serious or infectious adverse events or study withdrawals.
Of note, azathioprine was stopped in treatment group patients who achieved complete renal response – but not control group patients who achieved complete renal response. These patients were followed to 52 weeks. At that time, 50% and 62% of the treatment group and control group patients, respectively, who were complete responders at 24 weeks still met the criteria for complete response, Dr. Rovin said.
While abatacept did not increase complete response rates, the treatment was associated with maintenance of complete renal response to 1 year despite discontinuation of maintenance immunosuppression in the treated patients and continued azathioprine treatment in the control group. This suggests that abatacept may reduce the need for maintenance immunosuppression, he said.
While it was disappointing that the addition of abatacept to background induction therapy with low-dose IV cyclophosphamide and corticosteroids did not improve the complete renal response rate in this study – despite murine models suggesting that the use of abatacept and IV cyclophosphamide act synergistically to arrest lupus nephritis progression – the findings offer two very important lessons for clinical practice, he said.
"We can take the low-dose cyclophosphamide regimen and potentially use that with good results in our [multiethnic, multiracial] patient population that we see here in this country, and I think that’s a huge step forward. It gives us another option to use besides mycophenolate mofetil and high-dose cyclophosphamide," he said, adding that the low-dose IV cyclophosphamide regimen is well tolerated and delivers a total dose of only 3 g of the drug – a dose that generally does not predispose to premature ovarian failure.
This is particularly important, because women of childbearing years comprise a majority of the patient population with lupus nephritis, he explained.
"We know from the Euro-Lupus trial that the low-dose regimen, over 10 years, provided the same maintenance of renal function as the high-dose regimen, so we have very good long-term follow-up. We don’t quite have that yet with mycophenolate mofetil, so I think that for those of us who are very frequent users of cyclophosphamide, this is a very good alternative to the high-dose, highly toxic regimen that has for so long been the standard of care," he said.
This study was funded by the National Institute of Allergy and Infectious Diseases via the Immune Tolerance Network. Bristol Myers Squibb provided the study drug. Dr. Rovin reported ties to Genentech, Questcor, TEVA, Biogen-IDEC, HGS, Johnson & Johnson, Roche, BMS, and TG Therapeutics.
AT KIDNEY WEEK 2013
Major finding: Complete renal response rates were similar at 33% and 31% in treatment vs. placebo group patients, respectively.
Data source: Phase II of the randomized, placebo-controlled ACCESS trial involving 134 patients.
Disclosures: This study was funded by the National Institute of Allergy and Infectious Diseases via the Immune Tolerance Network. Bristol Myers Squibb provided the study drug. Dr. Rovin reported ties to Genentech, Questcor, TEVA, Biogen-IDEC, HGS, Johnson & Johnson, Roche, BMS, and TG Therapeutics.