User login
Humira, Enbrel costs highest in the United States
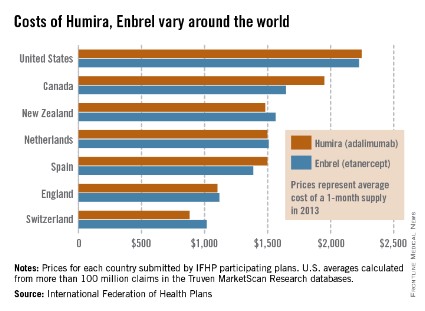
The costs of Humira and Enbrel are higher in the United States, compared with other industrialized countries included in the International Federation of Health Plans’ 2013 Comparative Price Report.
The average cost of a 1-month supply was $2,246 for Humira (adalimumab) in 2013 and $2,225 for Enbrel (etanercept). In Canada, which had second-highest cost for both drugs, Humira cost $1,950 per month and Enbrel cost $1,646 per month, the IFHP reported.
Switzerland had the lowest cost for both drugs among the countries included in the comparison: $881 for Humira and $1,017 for Enbrel.
In the United States, Humira is approved for the treatment of rheumatoid arthritis, juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, and plaque psoriasis. Enbrel is approved for RA, polyarticular juvenile idiopathic arthritis in patients aged 2 years or older, psoriatic arthritis, ankylosing spondylitis, and plaque psoriasis.
The IFHP comprises more than 100 member companies in 25 countries. For the survey, the price for each country was submitted by participating member plans. Some prices are drawn from the public sector, some from the private, and some from both. U.S. averages were calculated from more than 100 million claims in the Truven MarketScan Research databases.
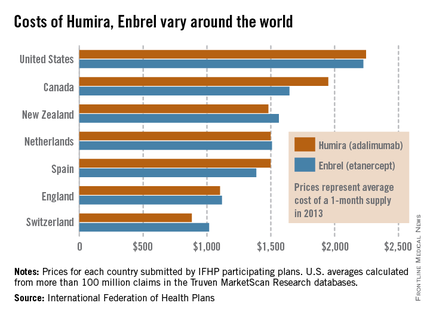
The costs of Humira and Enbrel are higher in the United States, compared with other industrialized countries included in the International Federation of Health Plans’ 2013 Comparative Price Report.
The average cost of a 1-month supply was $2,246 for Humira (adalimumab) in 2013 and $2,225 for Enbrel (etanercept). In Canada, which had second-highest cost for both drugs, Humira cost $1,950 per month and Enbrel cost $1,646 per month, the IFHP reported.
Switzerland had the lowest cost for both drugs among the countries included in the comparison: $881 for Humira and $1,017 for Enbrel.
In the United States, Humira is approved for the treatment of rheumatoid arthritis, juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, and plaque psoriasis. Enbrel is approved for RA, polyarticular juvenile idiopathic arthritis in patients aged 2 years or older, psoriatic arthritis, ankylosing spondylitis, and plaque psoriasis.
The IFHP comprises more than 100 member companies in 25 countries. For the survey, the price for each country was submitted by participating member plans. Some prices are drawn from the public sector, some from the private, and some from both. U.S. averages were calculated from more than 100 million claims in the Truven MarketScan Research databases.

The costs of Humira and Enbrel are higher in the United States, compared with other industrialized countries included in the International Federation of Health Plans’ 2013 Comparative Price Report.
The average cost of a 1-month supply was $2,246 for Humira (adalimumab) in 2013 and $2,225 for Enbrel (etanercept). In Canada, which had second-highest cost for both drugs, Humira cost $1,950 per month and Enbrel cost $1,646 per month, the IFHP reported.
Switzerland had the lowest cost for both drugs among the countries included in the comparison: $881 for Humira and $1,017 for Enbrel.
In the United States, Humira is approved for the treatment of rheumatoid arthritis, juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, and plaque psoriasis. Enbrel is approved for RA, polyarticular juvenile idiopathic arthritis in patients aged 2 years or older, psoriatic arthritis, ankylosing spondylitis, and plaque psoriasis.
The IFHP comprises more than 100 member companies in 25 countries. For the survey, the price for each country was submitted by participating member plans. Some prices are drawn from the public sector, some from the private, and some from both. U.S. averages were calculated from more than 100 million claims in the Truven MarketScan Research databases.

Alopecia Areata Universalis Complicating Daclizumab Therapy for Uveitis
Practice Question Answers: Medications in Dermatology, Part 2
1. A 40-year-old woman is diagnosed with systemic lupus erythematosus. You discuss treatment options and decide to start hydroxychloroquine. What laboratory tests and monitoring are required prior to starting this medication?
a. complete blood cell count with differential and glucose-6-phosphate dehydrogenase
b. complete blood cell count with differential and complete metabolic profile
c. ophthalmology evaluation and glucose-6-phosphate dehydrogenase
d. b and c
2. Two months ago you saw a 30-year-old woman with a history of severe atopic dermatitis. She had been using topical steroids with not much improvement. You decided to start a systemic medication. Within 1 month of drug initiation, she called your office to tell you that she is much better but has noticed unwanted hair on her face lately. Which medication is most likely implicated?
a. cyclosporine
b. dapsone
c. hydroxychloroquine
d. methotrexate
3. A 70-year-old man with type 2 diabetes mellitus who drinks 10 cans of beer per week presents to the emergency department with a 3-day history of diffuse tense bullae and pruritus on the legs and trunk. Direct immunofluorescence displayed linear deposition of IgG and C3 at the dermoepidermal junction, confirming your clinical diagnosis. What is the best long-term treatment option for this patient?
a. combination of oral steroids plus methotrexate
b. oral steroids and mycophenolate mofetil
c. oral steroids only
d. topical steroids only
4. A 45-year-old Venezuelan man presents with painful nodules on his bilateral lower legs. A biopsy demonstrates acid-fast bacilli, and a multidrug regimen is initiated for erythema nodosum leprosum. Which of the following is the mechanism of action of the treatment that is US Food and Drug Administration approved for this condition?
a. inhibits chemotaxis
b. inhibits dihydrofolate reductase
c. inhibits tumor necrosis factor α
d. suppresses T-cell function and B-cell antibody production
5. A patient consults her physician because of several side effects from a medication she started 2 weeks ago due to erythematous to violaceous papules on the legs from palpable purpura. She reports diarrhea, abdominal pain, and fatigue. Which medication is she taking?
a. azathioprine
b. colchicine
c. dapsone
d. methotrexate
1. A 40-year-old woman is diagnosed with systemic lupus erythematosus. You discuss treatment options and decide to start hydroxychloroquine. What laboratory tests and monitoring are required prior to starting this medication?
a. complete blood cell count with differential and glucose-6-phosphate dehydrogenase
b. complete blood cell count with differential and complete metabolic profile
c. ophthalmology evaluation and glucose-6-phosphate dehydrogenase
d. b and c
2. Two months ago you saw a 30-year-old woman with a history of severe atopic dermatitis. She had been using topical steroids with not much improvement. You decided to start a systemic medication. Within 1 month of drug initiation, she called your office to tell you that she is much better but has noticed unwanted hair on her face lately. Which medication is most likely implicated?
a. cyclosporine
b. dapsone
c. hydroxychloroquine
d. methotrexate
3. A 70-year-old man with type 2 diabetes mellitus who drinks 10 cans of beer per week presents to the emergency department with a 3-day history of diffuse tense bullae and pruritus on the legs and trunk. Direct immunofluorescence displayed linear deposition of IgG and C3 at the dermoepidermal junction, confirming your clinical diagnosis. What is the best long-term treatment option for this patient?
a. combination of oral steroids plus methotrexate
b. oral steroids and mycophenolate mofetil
c. oral steroids only
d. topical steroids only
4. A 45-year-old Venezuelan man presents with painful nodules on his bilateral lower legs. A biopsy demonstrates acid-fast bacilli, and a multidrug regimen is initiated for erythema nodosum leprosum. Which of the following is the mechanism of action of the treatment that is US Food and Drug Administration approved for this condition?
a. inhibits chemotaxis
b. inhibits dihydrofolate reductase
c. inhibits tumor necrosis factor α
d. suppresses T-cell function and B-cell antibody production
5. A patient consults her physician because of several side effects from a medication she started 2 weeks ago due to erythematous to violaceous papules on the legs from palpable purpura. She reports diarrhea, abdominal pain, and fatigue. Which medication is she taking?
a. azathioprine
b. colchicine
c. dapsone
d. methotrexate
1. A 40-year-old woman is diagnosed with systemic lupus erythematosus. You discuss treatment options and decide to start hydroxychloroquine. What laboratory tests and monitoring are required prior to starting this medication?
a. complete blood cell count with differential and glucose-6-phosphate dehydrogenase
b. complete blood cell count with differential and complete metabolic profile
c. ophthalmology evaluation and glucose-6-phosphate dehydrogenase
d. b and c
2. Two months ago you saw a 30-year-old woman with a history of severe atopic dermatitis. She had been using topical steroids with not much improvement. You decided to start a systemic medication. Within 1 month of drug initiation, she called your office to tell you that she is much better but has noticed unwanted hair on her face lately. Which medication is most likely implicated?
a. cyclosporine
b. dapsone
c. hydroxychloroquine
d. methotrexate
3. A 70-year-old man with type 2 diabetes mellitus who drinks 10 cans of beer per week presents to the emergency department with a 3-day history of diffuse tense bullae and pruritus on the legs and trunk. Direct immunofluorescence displayed linear deposition of IgG and C3 at the dermoepidermal junction, confirming your clinical diagnosis. What is the best long-term treatment option for this patient?
a. combination of oral steroids plus methotrexate
b. oral steroids and mycophenolate mofetil
c. oral steroids only
d. topical steroids only
4. A 45-year-old Venezuelan man presents with painful nodules on his bilateral lower legs. A biopsy demonstrates acid-fast bacilli, and a multidrug regimen is initiated for erythema nodosum leprosum. Which of the following is the mechanism of action of the treatment that is US Food and Drug Administration approved for this condition?
a. inhibits chemotaxis
b. inhibits dihydrofolate reductase
c. inhibits tumor necrosis factor α
d. suppresses T-cell function and B-cell antibody production
5. A patient consults her physician because of several side effects from a medication she started 2 weeks ago due to erythematous to violaceous papules on the legs from palpable purpura. She reports diarrhea, abdominal pain, and fatigue. Which medication is she taking?
a. azathioprine
b. colchicine
c. dapsone
d. methotrexate
Medications in Dermatology, Part 2: Immunosuppressives
The Impact of Diet and Exercise in Psoriasis
It is well established that increased body mass index and weight gain are risk factors for psoriasis, and the prevalence of obesity in patients with psoriasis is higher than in the general population. However, there are limited data concerning the role of diet and exercise in psoriasis.
Naldi et al (Br J Dermatol. 2014;170:634-642) assessed the impact of dietary intervention in combination with physical exercise for weight loss on improving psoriasis in overweight or obese individuals. The investigators evaluated 303 overweight or obese patients with moderate to severe chronic plaque psoriasis who did not achieve clearance after 4 weeks of continuous systemic treatment. Patients were randomized to 2 regimens: a 20-week quantitative and qualitative dietary plan associated with physical exercise for weight loss, or simple informative counseling at baseline about the utility of weight loss for clinical control of psoriatic disease. The main outcome was any reduction of the psoriasis area and severity index (PASI) from baseline to week 20.
Analysis of the intention-to-treat population showed a median reduction in the PASI score of 48% (95% confidence interval, 33.3%-58.3%) in the diet arm and 25.5% (95% confidence interval, 18.2%-33.3%) in the counseling arm (P=.02). The weight-loss target (a ≥5% reduction from baseline) was reached by 29.8% of patients in the diet arm compared to 14.5% in the counseling arm (P=.001).
The authors concluded that a 20-week dietetic intervention associated with increased physical exercise reduced psoriasis severity in systemically treated overweight or obese patients with active psoriasis.
What’s the issue?
As we would expect, a direct dietary intervention had a great impact on the study population. Will you try to adopt a structured dietary intervention in your patient population?
It is well established that increased body mass index and weight gain are risk factors for psoriasis, and the prevalence of obesity in patients with psoriasis is higher than in the general population. However, there are limited data concerning the role of diet and exercise in psoriasis.
Naldi et al (Br J Dermatol. 2014;170:634-642) assessed the impact of dietary intervention in combination with physical exercise for weight loss on improving psoriasis in overweight or obese individuals. The investigators evaluated 303 overweight or obese patients with moderate to severe chronic plaque psoriasis who did not achieve clearance after 4 weeks of continuous systemic treatment. Patients were randomized to 2 regimens: a 20-week quantitative and qualitative dietary plan associated with physical exercise for weight loss, or simple informative counseling at baseline about the utility of weight loss for clinical control of psoriatic disease. The main outcome was any reduction of the psoriasis area and severity index (PASI) from baseline to week 20.
Analysis of the intention-to-treat population showed a median reduction in the PASI score of 48% (95% confidence interval, 33.3%-58.3%) in the diet arm and 25.5% (95% confidence interval, 18.2%-33.3%) in the counseling arm (P=.02). The weight-loss target (a ≥5% reduction from baseline) was reached by 29.8% of patients in the diet arm compared to 14.5% in the counseling arm (P=.001).
The authors concluded that a 20-week dietetic intervention associated with increased physical exercise reduced psoriasis severity in systemically treated overweight or obese patients with active psoriasis.
What’s the issue?
As we would expect, a direct dietary intervention had a great impact on the study population. Will you try to adopt a structured dietary intervention in your patient population?
It is well established that increased body mass index and weight gain are risk factors for psoriasis, and the prevalence of obesity in patients with psoriasis is higher than in the general population. However, there are limited data concerning the role of diet and exercise in psoriasis.
Naldi et al (Br J Dermatol. 2014;170:634-642) assessed the impact of dietary intervention in combination with physical exercise for weight loss on improving psoriasis in overweight or obese individuals. The investigators evaluated 303 overweight or obese patients with moderate to severe chronic plaque psoriasis who did not achieve clearance after 4 weeks of continuous systemic treatment. Patients were randomized to 2 regimens: a 20-week quantitative and qualitative dietary plan associated with physical exercise for weight loss, or simple informative counseling at baseline about the utility of weight loss for clinical control of psoriatic disease. The main outcome was any reduction of the psoriasis area and severity index (PASI) from baseline to week 20.
Analysis of the intention-to-treat population showed a median reduction in the PASI score of 48% (95% confidence interval, 33.3%-58.3%) in the diet arm and 25.5% (95% confidence interval, 18.2%-33.3%) in the counseling arm (P=.02). The weight-loss target (a ≥5% reduction from baseline) was reached by 29.8% of patients in the diet arm compared to 14.5% in the counseling arm (P=.001).
The authors concluded that a 20-week dietetic intervention associated with increased physical exercise reduced psoriasis severity in systemically treated overweight or obese patients with active psoriasis.
What’s the issue?
As we would expect, a direct dietary intervention had a great impact on the study population. Will you try to adopt a structured dietary intervention in your patient population?
Denver AAD 2014

With the annual meeting of the American Academy of Dermatology (AAD) set in the “mile-high city” now behind us, we must begin to convert didactic sessions into improved outcomes in our daily patient encounters. The enormous variety of lectures and frequently overlapping schedules can make this event a whirlwind for unseasoned attendees. I aim to enlighten those attending future meetings about individual sessions of particularly high value to the dermatologist in training. As a disclaimer, my primary interests are in medical dermatology, so the content of the courses I recommend is by no means comprehensive; however, residents need to have a solid fund of medical knowledge to function in any practice setting and, perhaps more importantly, to pass the boards examination!
I think the session that takes the cake for utility and value for residents is “High Yield ‘Power Hour’ for Residents,” which was led by a group of education-oriented Harvard University dermatologists. The power “hour” in fact lasted 2 hours, during which there was a variety of material presented covering pediatric dermatology, allergic dermatitis, infectious disease dermatology, blistering disorders, and pharmacology. The presenters showed incredible enthusiasm for their respective topics, and their passion also was evident in the high-yield handouts that they provided that were jam-packed with tables, bullet points, and frequently tested material. I would recommend that attendees save or print the handouts and avoid taking extensive notes on them during the session. Sit back, relax, and just soak in the lectures; later on, review the handouts. Also, be sure to arrive early—this session fills up fast—and fill out your evaluation! These lecturers deserve credit for their presentations and deserve a much larger room to accommodate residents that are otherwise willing to sit on the floor, crowd against the walls, and peek in through the doorway to listen.
I also feel residents benefit from lectures that provide us with practical information regarding complicated medical problems. I greatly enjoyed the symposium “Biologics: Perils and Promise” led by Canadian Dermatology Association President Richard Langley, MD. This conglomerate of experts addressed the risk for infection and malignancy with the use of biologics as well as pediatric use. The symposium also touched on the cardiovascular risk inherent in psoriasis and new developments suggesting that the treatment of psoriasis decreases overall systemic inflammation and possibly even cardiovascular risk. Another symposium in the same vein was “Systemic Therapies for Dermatologists: A Comprehensive Review and Update,” which was divided into short lectures discussing agents from acitretin to Zelboraf (vemurafenib). Expert insight on the use of these drugs was invaluable for those of us who train in programs where we are not frequently exposed to these agents.
For those residents interested in an overview of dermatopathology, “Dermatopathology Made Simple” led by Christine Ko, MD, was a lightning-fast tour through the subject. Dr. Ko categorized diagnoses based on pattern and also compared similar pathology side-by-side, focusing on key differences to help cinch the correct diagnosis. Although this talk was only 2 hours, it covered surprising breadth, as more than 170 different cases were presented. The handout accompanying this talk was excellent and served as a condensed review of all the material she covered.
Any resident attending an AAD meeting should first review the schedule carefully and then find topics that would be most beneficial during training and later on during practice. The recommendations I have made summarize material that will benefit all trainees in dermatology. Even those of us who will be continuing on to a fellowship need to have a handle on these topics to remain current and better communicate with colleagues. I look forward to seeing you all at future meetings!

With the annual meeting of the American Academy of Dermatology (AAD) set in the “mile-high city” now behind us, we must begin to convert didactic sessions into improved outcomes in our daily patient encounters. The enormous variety of lectures and frequently overlapping schedules can make this event a whirlwind for unseasoned attendees. I aim to enlighten those attending future meetings about individual sessions of particularly high value to the dermatologist in training. As a disclaimer, my primary interests are in medical dermatology, so the content of the courses I recommend is by no means comprehensive; however, residents need to have a solid fund of medical knowledge to function in any practice setting and, perhaps more importantly, to pass the boards examination!
I think the session that takes the cake for utility and value for residents is “High Yield ‘Power Hour’ for Residents,” which was led by a group of education-oriented Harvard University dermatologists. The power “hour” in fact lasted 2 hours, during which there was a variety of material presented covering pediatric dermatology, allergic dermatitis, infectious disease dermatology, blistering disorders, and pharmacology. The presenters showed incredible enthusiasm for their respective topics, and their passion also was evident in the high-yield handouts that they provided that were jam-packed with tables, bullet points, and frequently tested material. I would recommend that attendees save or print the handouts and avoid taking extensive notes on them during the session. Sit back, relax, and just soak in the lectures; later on, review the handouts. Also, be sure to arrive early—this session fills up fast—and fill out your evaluation! These lecturers deserve credit for their presentations and deserve a much larger room to accommodate residents that are otherwise willing to sit on the floor, crowd against the walls, and peek in through the doorway to listen.
I also feel residents benefit from lectures that provide us with practical information regarding complicated medical problems. I greatly enjoyed the symposium “Biologics: Perils and Promise” led by Canadian Dermatology Association President Richard Langley, MD. This conglomerate of experts addressed the risk for infection and malignancy with the use of biologics as well as pediatric use. The symposium also touched on the cardiovascular risk inherent in psoriasis and new developments suggesting that the treatment of psoriasis decreases overall systemic inflammation and possibly even cardiovascular risk. Another symposium in the same vein was “Systemic Therapies for Dermatologists: A Comprehensive Review and Update,” which was divided into short lectures discussing agents from acitretin to Zelboraf (vemurafenib). Expert insight on the use of these drugs was invaluable for those of us who train in programs where we are not frequently exposed to these agents.
For those residents interested in an overview of dermatopathology, “Dermatopathology Made Simple” led by Christine Ko, MD, was a lightning-fast tour through the subject. Dr. Ko categorized diagnoses based on pattern and also compared similar pathology side-by-side, focusing on key differences to help cinch the correct diagnosis. Although this talk was only 2 hours, it covered surprising breadth, as more than 170 different cases were presented. The handout accompanying this talk was excellent and served as a condensed review of all the material she covered.
Any resident attending an AAD meeting should first review the schedule carefully and then find topics that would be most beneficial during training and later on during practice. The recommendations I have made summarize material that will benefit all trainees in dermatology. Even those of us who will be continuing on to a fellowship need to have a handle on these topics to remain current and better communicate with colleagues. I look forward to seeing you all at future meetings!

With the annual meeting of the American Academy of Dermatology (AAD) set in the “mile-high city” now behind us, we must begin to convert didactic sessions into improved outcomes in our daily patient encounters. The enormous variety of lectures and frequently overlapping schedules can make this event a whirlwind for unseasoned attendees. I aim to enlighten those attending future meetings about individual sessions of particularly high value to the dermatologist in training. As a disclaimer, my primary interests are in medical dermatology, so the content of the courses I recommend is by no means comprehensive; however, residents need to have a solid fund of medical knowledge to function in any practice setting and, perhaps more importantly, to pass the boards examination!
I think the session that takes the cake for utility and value for residents is “High Yield ‘Power Hour’ for Residents,” which was led by a group of education-oriented Harvard University dermatologists. The power “hour” in fact lasted 2 hours, during which there was a variety of material presented covering pediatric dermatology, allergic dermatitis, infectious disease dermatology, blistering disorders, and pharmacology. The presenters showed incredible enthusiasm for their respective topics, and their passion also was evident in the high-yield handouts that they provided that were jam-packed with tables, bullet points, and frequently tested material. I would recommend that attendees save or print the handouts and avoid taking extensive notes on them during the session. Sit back, relax, and just soak in the lectures; later on, review the handouts. Also, be sure to arrive early—this session fills up fast—and fill out your evaluation! These lecturers deserve credit for their presentations and deserve a much larger room to accommodate residents that are otherwise willing to sit on the floor, crowd against the walls, and peek in through the doorway to listen.
I also feel residents benefit from lectures that provide us with practical information regarding complicated medical problems. I greatly enjoyed the symposium “Biologics: Perils and Promise” led by Canadian Dermatology Association President Richard Langley, MD. This conglomerate of experts addressed the risk for infection and malignancy with the use of biologics as well as pediatric use. The symposium also touched on the cardiovascular risk inherent in psoriasis and new developments suggesting that the treatment of psoriasis decreases overall systemic inflammation and possibly even cardiovascular risk. Another symposium in the same vein was “Systemic Therapies for Dermatologists: A Comprehensive Review and Update,” which was divided into short lectures discussing agents from acitretin to Zelboraf (vemurafenib). Expert insight on the use of these drugs was invaluable for those of us who train in programs where we are not frequently exposed to these agents.
For those residents interested in an overview of dermatopathology, “Dermatopathology Made Simple” led by Christine Ko, MD, was a lightning-fast tour through the subject. Dr. Ko categorized diagnoses based on pattern and also compared similar pathology side-by-side, focusing on key differences to help cinch the correct diagnosis. Although this talk was only 2 hours, it covered surprising breadth, as more than 170 different cases were presented. The handout accompanying this talk was excellent and served as a condensed review of all the material she covered.
Any resident attending an AAD meeting should first review the schedule carefully and then find topics that would be most beneficial during training and later on during practice. The recommendations I have made summarize material that will benefit all trainees in dermatology. Even those of us who will be continuing on to a fellowship need to have a handle on these topics to remain current and better communicate with colleagues. I look forward to seeing you all at future meetings!
Key studies show distinctive features of pediatric psoriasis
WAIKOLOA, HAWAII – Guttate psoriasis in children warrants more aggressive monitoring and treatment in an effort to head off more severe disease later, according to Dr. Wynnis L. Tom.
Pediatric psoriasis presenting initially as guttate disease is more likely to progress to severe psoriasis prior to adulthood than if the initial presentation took the form of plaque psoriasis, according to data from a multicenter U.S. study of the clinical manifestations of pediatric psoriasis.
The cross-sectional study included 181 children aged 5-17 years with plaque psoriasis, and the results highlighted important differences between childhood-onset and adult-onset disease, Dr. Tom said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
About 40% of cases of pediatric psoriasis that presented initially as guttate psoriasis progressed to chronic disease. That was not a higher proportion than in children whose initial disease was plaque psoriasis, said Dr. Tom of the University of California, San Diego, and Rady Children’s Hospital. However, 36% of youths with severe psoriasis had a history of disease onset with guttate morphology compared with 22% of those with mild disease, she said.
The peak severity of psoriasis was defined historically as either mild or severe based upon body surface area involvement and Physician Global Assessment.
Overall, 79% of study participants had a history of scalp psoriasis and 39% had a history of nail involvement. However, these disease expressions were unrelated to psoriasis severity.
Boys were three times more likely than girls to have had nail involvement, but 60% less likely to have a history of scalp involvement. These sex-related differences probably reflect koebnerization, Dr. Tom said.
Approximately 5% of the patients had psoriasis restricted to their face alone. Among those with body involvement, the face was included in nearly half of cases.
Session chair Dr. Lawrence F. Eichenfield praised this study (Pediatr. Dermatol. 2013;30:424-8), on which Dr. Tom was a coauthor, as one of the top articles in the field of pediatric dermatology published within the past year. Although various studies indicate that 27%-45% of all cases of psoriasis begin in childhood, the clinical aspects of pediatric psoriasis haven’t been well characterized until now, said Dr. Eichenfield, professor of clinical pediatrics and medicine and chief of pediatric and adolescent dermatology at the University of California, San Diego.
Dr. Eichenfield also singled out Dr. Tom as coauthor of yet another of his top picks of recently published studies on the topic of pediatric psoriasis. This cross-sectional study included 409 psoriasis patients aged 5-17 years in nine countries. The prevalence of excess adiposity as defined by a body mass index at the 85th percentile or greater was 38% among the psoriatic children compared with 21% in matched controls. The likelihood of obesity as defined by a BMI in the 95th percentile or higher was 4.9-fold greater in children with severe psoriasis than in controls, and 3.6-fold greater in those with mild psoriasis compared than in controls. Among U.S. patients, the psoriasis/obesity association was magnified such that American children with severe and mild psoriasis were respectively 7.6-fold and 4.7-fold more likely to be obese than controls (JAMA Dermatol. 2013;149:166-76).
Central adiposity – an element of metabolic syndrome associated with increased cardiovascular risk in adults – was present as defined by waist circumference greater than the 90th percentile in 21% of youths with severe psoriasis, 14% with mild disease, and 9% of controls. In U.S. participants, these figures ballooned to 31%, 21%, and 12%, respectively.
Dr. Tom cited evidence from yet another study in which she was a coinvestigator suggesting that obesity may be a risk factor for pediatric psoriasis, rather than the other way around. In this three-center study, nearly all of a group of pediatric psoriasis patients were overweight or obese for at least 2 years prior to their psoriasis onset.
Current National Psoriasis Foundation guidelines recommend initiating cardiovascular risk factor screening "as early as age 20." Dr. Tom is among those seeking to revise the guidelines to extend screening to pediatric psoriasis patients. She said she now recommends consideration of cardiovascular screening in overweight and obese pediatric psoriasis patients – including monitoring of blood pressure, lipids, fasting blood glucose, and liver enzymes – along with hard-hitting guidance on dietary modifications, weight loss, and the importance of exercise.
"Ongoing studies are assessing the need for screening labs for nonoverweight children with psoriasis. Hopefully this will be sorted out soon as a group in our field. We’re hoping for updated guidelines," she said.
Dr. Tom disclosed serving as a financially uncompensated investigator for Amgen and Anacor.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Guttate psoriasis in children warrants more aggressive monitoring and treatment in an effort to head off more severe disease later, according to Dr. Wynnis L. Tom.
Pediatric psoriasis presenting initially as guttate disease is more likely to progress to severe psoriasis prior to adulthood than if the initial presentation took the form of plaque psoriasis, according to data from a multicenter U.S. study of the clinical manifestations of pediatric psoriasis.
The cross-sectional study included 181 children aged 5-17 years with plaque psoriasis, and the results highlighted important differences between childhood-onset and adult-onset disease, Dr. Tom said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
About 40% of cases of pediatric psoriasis that presented initially as guttate psoriasis progressed to chronic disease. That was not a higher proportion than in children whose initial disease was plaque psoriasis, said Dr. Tom of the University of California, San Diego, and Rady Children’s Hospital. However, 36% of youths with severe psoriasis had a history of disease onset with guttate morphology compared with 22% of those with mild disease, she said.
The peak severity of psoriasis was defined historically as either mild or severe based upon body surface area involvement and Physician Global Assessment.
Overall, 79% of study participants had a history of scalp psoriasis and 39% had a history of nail involvement. However, these disease expressions were unrelated to psoriasis severity.
Boys were three times more likely than girls to have had nail involvement, but 60% less likely to have a history of scalp involvement. These sex-related differences probably reflect koebnerization, Dr. Tom said.
Approximately 5% of the patients had psoriasis restricted to their face alone. Among those with body involvement, the face was included in nearly half of cases.
Session chair Dr. Lawrence F. Eichenfield praised this study (Pediatr. Dermatol. 2013;30:424-8), on which Dr. Tom was a coauthor, as one of the top articles in the field of pediatric dermatology published within the past year. Although various studies indicate that 27%-45% of all cases of psoriasis begin in childhood, the clinical aspects of pediatric psoriasis haven’t been well characterized until now, said Dr. Eichenfield, professor of clinical pediatrics and medicine and chief of pediatric and adolescent dermatology at the University of California, San Diego.
Dr. Eichenfield also singled out Dr. Tom as coauthor of yet another of his top picks of recently published studies on the topic of pediatric psoriasis. This cross-sectional study included 409 psoriasis patients aged 5-17 years in nine countries. The prevalence of excess adiposity as defined by a body mass index at the 85th percentile or greater was 38% among the psoriatic children compared with 21% in matched controls. The likelihood of obesity as defined by a BMI in the 95th percentile or higher was 4.9-fold greater in children with severe psoriasis than in controls, and 3.6-fold greater in those with mild psoriasis compared than in controls. Among U.S. patients, the psoriasis/obesity association was magnified such that American children with severe and mild psoriasis were respectively 7.6-fold and 4.7-fold more likely to be obese than controls (JAMA Dermatol. 2013;149:166-76).
Central adiposity – an element of metabolic syndrome associated with increased cardiovascular risk in adults – was present as defined by waist circumference greater than the 90th percentile in 21% of youths with severe psoriasis, 14% with mild disease, and 9% of controls. In U.S. participants, these figures ballooned to 31%, 21%, and 12%, respectively.
Dr. Tom cited evidence from yet another study in which she was a coinvestigator suggesting that obesity may be a risk factor for pediatric psoriasis, rather than the other way around. In this three-center study, nearly all of a group of pediatric psoriasis patients were overweight or obese for at least 2 years prior to their psoriasis onset.
Current National Psoriasis Foundation guidelines recommend initiating cardiovascular risk factor screening "as early as age 20." Dr. Tom is among those seeking to revise the guidelines to extend screening to pediatric psoriasis patients. She said she now recommends consideration of cardiovascular screening in overweight and obese pediatric psoriasis patients – including monitoring of blood pressure, lipids, fasting blood glucose, and liver enzymes – along with hard-hitting guidance on dietary modifications, weight loss, and the importance of exercise.
"Ongoing studies are assessing the need for screening labs for nonoverweight children with psoriasis. Hopefully this will be sorted out soon as a group in our field. We’re hoping for updated guidelines," she said.
Dr. Tom disclosed serving as a financially uncompensated investigator for Amgen and Anacor.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Guttate psoriasis in children warrants more aggressive monitoring and treatment in an effort to head off more severe disease later, according to Dr. Wynnis L. Tom.
Pediatric psoriasis presenting initially as guttate disease is more likely to progress to severe psoriasis prior to adulthood than if the initial presentation took the form of plaque psoriasis, according to data from a multicenter U.S. study of the clinical manifestations of pediatric psoriasis.
The cross-sectional study included 181 children aged 5-17 years with plaque psoriasis, and the results highlighted important differences between childhood-onset and adult-onset disease, Dr. Tom said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
About 40% of cases of pediatric psoriasis that presented initially as guttate psoriasis progressed to chronic disease. That was not a higher proportion than in children whose initial disease was plaque psoriasis, said Dr. Tom of the University of California, San Diego, and Rady Children’s Hospital. However, 36% of youths with severe psoriasis had a history of disease onset with guttate morphology compared with 22% of those with mild disease, she said.
The peak severity of psoriasis was defined historically as either mild or severe based upon body surface area involvement and Physician Global Assessment.
Overall, 79% of study participants had a history of scalp psoriasis and 39% had a history of nail involvement. However, these disease expressions were unrelated to psoriasis severity.
Boys were three times more likely than girls to have had nail involvement, but 60% less likely to have a history of scalp involvement. These sex-related differences probably reflect koebnerization, Dr. Tom said.
Approximately 5% of the patients had psoriasis restricted to their face alone. Among those with body involvement, the face was included in nearly half of cases.
Session chair Dr. Lawrence F. Eichenfield praised this study (Pediatr. Dermatol. 2013;30:424-8), on which Dr. Tom was a coauthor, as one of the top articles in the field of pediatric dermatology published within the past year. Although various studies indicate that 27%-45% of all cases of psoriasis begin in childhood, the clinical aspects of pediatric psoriasis haven’t been well characterized until now, said Dr. Eichenfield, professor of clinical pediatrics and medicine and chief of pediatric and adolescent dermatology at the University of California, San Diego.
Dr. Eichenfield also singled out Dr. Tom as coauthor of yet another of his top picks of recently published studies on the topic of pediatric psoriasis. This cross-sectional study included 409 psoriasis patients aged 5-17 years in nine countries. The prevalence of excess adiposity as defined by a body mass index at the 85th percentile or greater was 38% among the psoriatic children compared with 21% in matched controls. The likelihood of obesity as defined by a BMI in the 95th percentile or higher was 4.9-fold greater in children with severe psoriasis than in controls, and 3.6-fold greater in those with mild psoriasis compared than in controls. Among U.S. patients, the psoriasis/obesity association was magnified such that American children with severe and mild psoriasis were respectively 7.6-fold and 4.7-fold more likely to be obese than controls (JAMA Dermatol. 2013;149:166-76).
Central adiposity – an element of metabolic syndrome associated with increased cardiovascular risk in adults – was present as defined by waist circumference greater than the 90th percentile in 21% of youths with severe psoriasis, 14% with mild disease, and 9% of controls. In U.S. participants, these figures ballooned to 31%, 21%, and 12%, respectively.
Dr. Tom cited evidence from yet another study in which she was a coinvestigator suggesting that obesity may be a risk factor for pediatric psoriasis, rather than the other way around. In this three-center study, nearly all of a group of pediatric psoriasis patients were overweight or obese for at least 2 years prior to their psoriasis onset.
Current National Psoriasis Foundation guidelines recommend initiating cardiovascular risk factor screening "as early as age 20." Dr. Tom is among those seeking to revise the guidelines to extend screening to pediatric psoriasis patients. She said she now recommends consideration of cardiovascular screening in overweight and obese pediatric psoriasis patients – including monitoring of blood pressure, lipids, fasting blood glucose, and liver enzymes – along with hard-hitting guidance on dietary modifications, weight loss, and the importance of exercise.
"Ongoing studies are assessing the need for screening labs for nonoverweight children with psoriasis. Hopefully this will be sorted out soon as a group in our field. We’re hoping for updated guidelines," she said.
Dr. Tom disclosed serving as a financially uncompensated investigator for Amgen and Anacor.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Ustekinumab again linked to cardiovascular events
WAIKOLOA, HAWAII – The anti-interleukin-12/23 biologic agents ustekinumab and briakinumab were associated with a statistically significant 4.23-fold increased risk of major adverse cardiac events in the latest meta-analysis of placebo-controlled clinical trials conducted in patients with chronic plaque psoriasis.
"What are the implications? It’s probably a class effect. That’s the way I practice. I think there is a slightly increased risk of myocardial infarction in our anti-IL-12/23-treated patients. So I think you should consider all of your options when selecting a biologic therapy. We know that our psoriasis patients typically have multiple cardiac risk factors, and that TNF antagonists are cardioprotective," Dr. Craig L. Leonardi said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
The latest industry-independent meta-analysis (J. Eur. Acad. Dermatol. Venereol. 2013;27:622-7), led by dermatologists at Dessau (Germany) Medical Center, examined the same nine phase-II and phase-III placebo-controlled randomized trials scrutinized in an earlier meta-analysis, also industry independent, carried out by Dr. Leonardi and coinvestigators (JAMA 2011;306:864-71).
The earlier meta-analysis identified 10 major adverse cardiovascular events (MACE) in 3,179 IL-12/23-treated subjects and none among 1,474 placebo-treated controls. This signal was deemed not statistically significant, although Dr. Leonardi and coworkers noted that their meta-analysis may have been underpowered to detect a small increase in risk.
Given that the two meta-analyses relied upon essentially the same data, how did they reach such different conclusions? It’s all in the statistical methods. Without getting geeky about the statistical fine points, Dr. Leonardi noted that his group used the Mantel-Haenszel fixed-effects model, while the Dessau group employed the Peto method, which their statisticians deemed more appropriate on the basis of its previously established superior performance in detecting rare events.
The Peto method is named for Sir Richard Peto, a renowned University of Oxford epidemiologist knighted for his statistical contributions.
Dr. Leonardi, a dermatologist in private practice in St. Louis and a clinical professor of dermatology at Saint Louis University, wasn’t about to quibble about statistics. Taken together, he said, the story conveyed by the two meta-analyses is one of a small but real increase in the absolute risk of MACE in psoriasis patients exposed to anti-IL-12/23 biologics. Based upon this evidence, when he starts a psoriasis patient on ustekinumab (Stelara), he now generally does so at the lower 45-mg dose regardless of the patient’s weight, even though the 90-mg dose is approved for use in patients weighing more than 100 kg.
"And I’m placing my patients on aspirin at 81 mg/day while we await further data," he added.
He anticipates that clinically meaningful data will eventually come from the Psoriasis Longitudinal Assessment and Registry (PSOLAR) study, an ongoing observational registry that has enrolled nearly 12,000 psoriasis patients for a planned follow-up period of at least 8 years at 266 investigative sites in 15 countries. The Janssen-funded registry includes roughly 3,800 patients on ustekinumab, lesser numbers on the other biologics, as well as a large group on phototherapy and no biologics.
Dr. Leonardi presented an early interim analysis in which the rate of MACE in the ustekinumab group was 0.28 per 100 patient-years. This was numerically slightly lower than but still comparable to the rates observed with infliximab and other biologics, and half the rate in patients on no biologics. He stressed, however, that these are unadjusted rates. Planned formal comparisons will require longer follow-up periods, more MACE, and multivariate analysis to control for baseline differences in comorbid conditions and cardiovascular risk factors. The no-biologics group, for example, was significantly older than patients on biologic agents, because once patients become Medicare eligible it is quite difficult to get a prescription for a biologic. And of course cardiovascular risk climbs with advancing age.
Dr. Leonardi is a PSOLAR coinvestigator, and a big fan of the project’s potential.
"Don’t wait for an NIH-sponsored disease-specific registry. The fact is the government is not going to fund it, so we have to do the best we can with the tools we have. PSOLAR is the largest psoriasis registry in the world. It will allow us to answer questions about the emergence of comorbid diseases like psoriatic arthritis and cardiovascular disease, and treatment-specific questions like rates of infection, cancer, and MACE, as well as unanticipated issues nobody knew about," he said.
Dr. Leonardi disclosed ties with Janssen and nearly two dozen other pharmaceutical companies.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The anti-interleukin-12/23 biologic agents ustekinumab and briakinumab were associated with a statistically significant 4.23-fold increased risk of major adverse cardiac events in the latest meta-analysis of placebo-controlled clinical trials conducted in patients with chronic plaque psoriasis.
"What are the implications? It’s probably a class effect. That’s the way I practice. I think there is a slightly increased risk of myocardial infarction in our anti-IL-12/23-treated patients. So I think you should consider all of your options when selecting a biologic therapy. We know that our psoriasis patients typically have multiple cardiac risk factors, and that TNF antagonists are cardioprotective," Dr. Craig L. Leonardi said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
The latest industry-independent meta-analysis (J. Eur. Acad. Dermatol. Venereol. 2013;27:622-7), led by dermatologists at Dessau (Germany) Medical Center, examined the same nine phase-II and phase-III placebo-controlled randomized trials scrutinized in an earlier meta-analysis, also industry independent, carried out by Dr. Leonardi and coinvestigators (JAMA 2011;306:864-71).
The earlier meta-analysis identified 10 major adverse cardiovascular events (MACE) in 3,179 IL-12/23-treated subjects and none among 1,474 placebo-treated controls. This signal was deemed not statistically significant, although Dr. Leonardi and coworkers noted that their meta-analysis may have been underpowered to detect a small increase in risk.
Given that the two meta-analyses relied upon essentially the same data, how did they reach such different conclusions? It’s all in the statistical methods. Without getting geeky about the statistical fine points, Dr. Leonardi noted that his group used the Mantel-Haenszel fixed-effects model, while the Dessau group employed the Peto method, which their statisticians deemed more appropriate on the basis of its previously established superior performance in detecting rare events.
The Peto method is named for Sir Richard Peto, a renowned University of Oxford epidemiologist knighted for his statistical contributions.
Dr. Leonardi, a dermatologist in private practice in St. Louis and a clinical professor of dermatology at Saint Louis University, wasn’t about to quibble about statistics. Taken together, he said, the story conveyed by the two meta-analyses is one of a small but real increase in the absolute risk of MACE in psoriasis patients exposed to anti-IL-12/23 biologics. Based upon this evidence, when he starts a psoriasis patient on ustekinumab (Stelara), he now generally does so at the lower 45-mg dose regardless of the patient’s weight, even though the 90-mg dose is approved for use in patients weighing more than 100 kg.
"And I’m placing my patients on aspirin at 81 mg/day while we await further data," he added.
He anticipates that clinically meaningful data will eventually come from the Psoriasis Longitudinal Assessment and Registry (PSOLAR) study, an ongoing observational registry that has enrolled nearly 12,000 psoriasis patients for a planned follow-up period of at least 8 years at 266 investigative sites in 15 countries. The Janssen-funded registry includes roughly 3,800 patients on ustekinumab, lesser numbers on the other biologics, as well as a large group on phototherapy and no biologics.
Dr. Leonardi presented an early interim analysis in which the rate of MACE in the ustekinumab group was 0.28 per 100 patient-years. This was numerically slightly lower than but still comparable to the rates observed with infliximab and other biologics, and half the rate in patients on no biologics. He stressed, however, that these are unadjusted rates. Planned formal comparisons will require longer follow-up periods, more MACE, and multivariate analysis to control for baseline differences in comorbid conditions and cardiovascular risk factors. The no-biologics group, for example, was significantly older than patients on biologic agents, because once patients become Medicare eligible it is quite difficult to get a prescription for a biologic. And of course cardiovascular risk climbs with advancing age.
Dr. Leonardi is a PSOLAR coinvestigator, and a big fan of the project’s potential.
"Don’t wait for an NIH-sponsored disease-specific registry. The fact is the government is not going to fund it, so we have to do the best we can with the tools we have. PSOLAR is the largest psoriasis registry in the world. It will allow us to answer questions about the emergence of comorbid diseases like psoriatic arthritis and cardiovascular disease, and treatment-specific questions like rates of infection, cancer, and MACE, as well as unanticipated issues nobody knew about," he said.
Dr. Leonardi disclosed ties with Janssen and nearly two dozen other pharmaceutical companies.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The anti-interleukin-12/23 biologic agents ustekinumab and briakinumab were associated with a statistically significant 4.23-fold increased risk of major adverse cardiac events in the latest meta-analysis of placebo-controlled clinical trials conducted in patients with chronic plaque psoriasis.
"What are the implications? It’s probably a class effect. That’s the way I practice. I think there is a slightly increased risk of myocardial infarction in our anti-IL-12/23-treated patients. So I think you should consider all of your options when selecting a biologic therapy. We know that our psoriasis patients typically have multiple cardiac risk factors, and that TNF antagonists are cardioprotective," Dr. Craig L. Leonardi said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
The latest industry-independent meta-analysis (J. Eur. Acad. Dermatol. Venereol. 2013;27:622-7), led by dermatologists at Dessau (Germany) Medical Center, examined the same nine phase-II and phase-III placebo-controlled randomized trials scrutinized in an earlier meta-analysis, also industry independent, carried out by Dr. Leonardi and coinvestigators (JAMA 2011;306:864-71).
The earlier meta-analysis identified 10 major adverse cardiovascular events (MACE) in 3,179 IL-12/23-treated subjects and none among 1,474 placebo-treated controls. This signal was deemed not statistically significant, although Dr. Leonardi and coworkers noted that their meta-analysis may have been underpowered to detect a small increase in risk.
Given that the two meta-analyses relied upon essentially the same data, how did they reach such different conclusions? It’s all in the statistical methods. Without getting geeky about the statistical fine points, Dr. Leonardi noted that his group used the Mantel-Haenszel fixed-effects model, while the Dessau group employed the Peto method, which their statisticians deemed more appropriate on the basis of its previously established superior performance in detecting rare events.
The Peto method is named for Sir Richard Peto, a renowned University of Oxford epidemiologist knighted for his statistical contributions.
Dr. Leonardi, a dermatologist in private practice in St. Louis and a clinical professor of dermatology at Saint Louis University, wasn’t about to quibble about statistics. Taken together, he said, the story conveyed by the two meta-analyses is one of a small but real increase in the absolute risk of MACE in psoriasis patients exposed to anti-IL-12/23 biologics. Based upon this evidence, when he starts a psoriasis patient on ustekinumab (Stelara), he now generally does so at the lower 45-mg dose regardless of the patient’s weight, even though the 90-mg dose is approved for use in patients weighing more than 100 kg.
"And I’m placing my patients on aspirin at 81 mg/day while we await further data," he added.
He anticipates that clinically meaningful data will eventually come from the Psoriasis Longitudinal Assessment and Registry (PSOLAR) study, an ongoing observational registry that has enrolled nearly 12,000 psoriasis patients for a planned follow-up period of at least 8 years at 266 investigative sites in 15 countries. The Janssen-funded registry includes roughly 3,800 patients on ustekinumab, lesser numbers on the other biologics, as well as a large group on phototherapy and no biologics.
Dr. Leonardi presented an early interim analysis in which the rate of MACE in the ustekinumab group was 0.28 per 100 patient-years. This was numerically slightly lower than but still comparable to the rates observed with infliximab and other biologics, and half the rate in patients on no biologics. He stressed, however, that these are unadjusted rates. Planned formal comparisons will require longer follow-up periods, more MACE, and multivariate analysis to control for baseline differences in comorbid conditions and cardiovascular risk factors. The no-biologics group, for example, was significantly older than patients on biologic agents, because once patients become Medicare eligible it is quite difficult to get a prescription for a biologic. And of course cardiovascular risk climbs with advancing age.
Dr. Leonardi is a PSOLAR coinvestigator, and a big fan of the project’s potential.
"Don’t wait for an NIH-sponsored disease-specific registry. The fact is the government is not going to fund it, so we have to do the best we can with the tools we have. PSOLAR is the largest psoriasis registry in the world. It will allow us to answer questions about the emergence of comorbid diseases like psoriatic arthritis and cardiovascular disease, and treatment-specific questions like rates of infection, cancer, and MACE, as well as unanticipated issues nobody knew about," he said.
Dr. Leonardi disclosed ties with Janssen and nearly two dozen other pharmaceutical companies.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Psoriatic pruritus improves with TrkA-blocking drug
An investigational topical drug that targets a nerve sensitization pathway showed good efficacy against chronic itch in patients with pruritic psoriasis.
CT327 works by inhibiting the kinase pathway of TrkA, a receptor that controls uptake of nerve growth factor, Dr. Gil Yosipovitch said at the annual meeting of the American Academy of Dermatology.
In a placebo-controlled trial of 160 patients with pruritic psoriasis, CT327 reduced itch by a mean of 60%, said Dr. Yosipovitch, chair of dermatology at the Temple University, Philadelphia.
"Sixty percent is not 90%," he said. "But when we consider that most of the antipruritic drugs have about a 15% efficacy in comparison to a vehicle, this is an effect we should consider. It’s well tolerated, and there were no application site reactions. I see potential in this drug."
CT327, developed by Creabilis, is a selective kinase inhibitor that targets TrkA, the receptor for nerve growth factor (NGF). NGF, which is overexpressed in atopic dermatitis and psoriatic skin, sensitizes neural networks that transmit the itch sensation to the brain, Dr. Yosipovitch said.
The phase IIb study comprised 160 adults with mild to moderate psoriasis, with at least a 10% body surface area involvement. Their mean score on the modified Psoriasis Area and Severity Index (mPASI) was about 9. Most (97%) had pruritus; 69% reported this as at least moderate, with a score of 40 on a 100-point visual analog scale (VAS). A third of the group reported severe pruritus, with a VAS of more than 70. They were randomized to four groups: an emollient placebo or CT327 in concentrations of 0.05%, 0.1%, or 0.5%.
The primary endpoint was reduction on a validated 100-point pruritus visual analog scale. The secondary endpoint was change on the mPASI.
By the end of 8 weeks, most patients (108) had experienced a significant reduction on the pruritus VAS. Reductions were about 35 points for the 0.1% group and 38 points for the 0.05% and 0.5% groups. These changes represented about a 60% decrease overall, Dr. Yosipovitch said.
Changes began early and continued along a steep trajectory, reaching a significant difference from baseline by week 2. However, he pointed out, by that point, the placebo group was similarly improved. This was probably because of the benefit of the daily emollient vehicle, he noted.
The curves began to separate shortly thereafter. By week 4, all of the treatment groups had experienced about a 30-point reduction on the VAS, while the placebo group had actually increased its score slightly. The active groups continued to improve over the next 4 weeks, while the placebo group plateaued at about a 20-point decrease from baseline.
The study concluded with 4 weeks of non–treatment follow-up. During that time, the placebo group stayed at about a 20-point decline and the active groups rebounded to about that level, "suggesting that there really is something going on here biologically," Dr. Yosipovitch said.
Patients using CT327 also experienced significant – although not overwhelming – benefit on the mPASI score. About 45% responded at the 50% level, compared with 23% of those using the emollient placebo. The active groups had similar improvements, with reductions on the mPASI of between 3 and 4 points. Those in the placebo group experienced about a 1-point improvement.
Dr. Yosipovitch said he was "not too impressed" with the mPASI changes. However, he noted, "Breaking the itch-scratch cycle at its source may have broader effects on other symptoms of psoriasis."
Based on the study results, Creabilis has decided to move forward into phase III testing with the 0.05% concentration. "Preparations are already underway," Dr. Yosipovitch said in an interview. "We also have a second phase IIb study ongoing that is investigating CT327 in patients with atopic dermatitis, which will be used to help finalize our phase III programs. This study should be completed in the middle of this year."
The NGF/TrkA pathway is implicated in other disorders as well, Dr. Yosipovitch said. CT327 might be investigated in some of these.
"These include disorders [in which] pruritus is important, such as pruritus with cutaneous T cell lymphoma, where NGF/TrkA has been shown to play a role. The pathway has also been shown to play an important role in chronic neuropathic and inflammatory pain. Molecules targeting NGF have already shown clinical efficacy."
Creabilis is investigating a related molecule, CT340, an inhibitor of both TrkA and MAP2K kinase, for treating those chronic pain conditions.
Creabilis funded the study. Dr. Yosipovitch is on the company’s scientific advisory board.
On Twitter @alz_gal
An investigational topical drug that targets a nerve sensitization pathway showed good efficacy against chronic itch in patients with pruritic psoriasis.
CT327 works by inhibiting the kinase pathway of TrkA, a receptor that controls uptake of nerve growth factor, Dr. Gil Yosipovitch said at the annual meeting of the American Academy of Dermatology.
In a placebo-controlled trial of 160 patients with pruritic psoriasis, CT327 reduced itch by a mean of 60%, said Dr. Yosipovitch, chair of dermatology at the Temple University, Philadelphia.
"Sixty percent is not 90%," he said. "But when we consider that most of the antipruritic drugs have about a 15% efficacy in comparison to a vehicle, this is an effect we should consider. It’s well tolerated, and there were no application site reactions. I see potential in this drug."
CT327, developed by Creabilis, is a selective kinase inhibitor that targets TrkA, the receptor for nerve growth factor (NGF). NGF, which is overexpressed in atopic dermatitis and psoriatic skin, sensitizes neural networks that transmit the itch sensation to the brain, Dr. Yosipovitch said.
The phase IIb study comprised 160 adults with mild to moderate psoriasis, with at least a 10% body surface area involvement. Their mean score on the modified Psoriasis Area and Severity Index (mPASI) was about 9. Most (97%) had pruritus; 69% reported this as at least moderate, with a score of 40 on a 100-point visual analog scale (VAS). A third of the group reported severe pruritus, with a VAS of more than 70. They were randomized to four groups: an emollient placebo or CT327 in concentrations of 0.05%, 0.1%, or 0.5%.
The primary endpoint was reduction on a validated 100-point pruritus visual analog scale. The secondary endpoint was change on the mPASI.
By the end of 8 weeks, most patients (108) had experienced a significant reduction on the pruritus VAS. Reductions were about 35 points for the 0.1% group and 38 points for the 0.05% and 0.5% groups. These changes represented about a 60% decrease overall, Dr. Yosipovitch said.
Changes began early and continued along a steep trajectory, reaching a significant difference from baseline by week 2. However, he pointed out, by that point, the placebo group was similarly improved. This was probably because of the benefit of the daily emollient vehicle, he noted.
The curves began to separate shortly thereafter. By week 4, all of the treatment groups had experienced about a 30-point reduction on the VAS, while the placebo group had actually increased its score slightly. The active groups continued to improve over the next 4 weeks, while the placebo group plateaued at about a 20-point decrease from baseline.
The study concluded with 4 weeks of non–treatment follow-up. During that time, the placebo group stayed at about a 20-point decline and the active groups rebounded to about that level, "suggesting that there really is something going on here biologically," Dr. Yosipovitch said.
Patients using CT327 also experienced significant – although not overwhelming – benefit on the mPASI score. About 45% responded at the 50% level, compared with 23% of those using the emollient placebo. The active groups had similar improvements, with reductions on the mPASI of between 3 and 4 points. Those in the placebo group experienced about a 1-point improvement.
Dr. Yosipovitch said he was "not too impressed" with the mPASI changes. However, he noted, "Breaking the itch-scratch cycle at its source may have broader effects on other symptoms of psoriasis."
Based on the study results, Creabilis has decided to move forward into phase III testing with the 0.05% concentration. "Preparations are already underway," Dr. Yosipovitch said in an interview. "We also have a second phase IIb study ongoing that is investigating CT327 in patients with atopic dermatitis, which will be used to help finalize our phase III programs. This study should be completed in the middle of this year."
The NGF/TrkA pathway is implicated in other disorders as well, Dr. Yosipovitch said. CT327 might be investigated in some of these.
"These include disorders [in which] pruritus is important, such as pruritus with cutaneous T cell lymphoma, where NGF/TrkA has been shown to play a role. The pathway has also been shown to play an important role in chronic neuropathic and inflammatory pain. Molecules targeting NGF have already shown clinical efficacy."
Creabilis is investigating a related molecule, CT340, an inhibitor of both TrkA and MAP2K kinase, for treating those chronic pain conditions.
Creabilis funded the study. Dr. Yosipovitch is on the company’s scientific advisory board.
On Twitter @alz_gal
An investigational topical drug that targets a nerve sensitization pathway showed good efficacy against chronic itch in patients with pruritic psoriasis.
CT327 works by inhibiting the kinase pathway of TrkA, a receptor that controls uptake of nerve growth factor, Dr. Gil Yosipovitch said at the annual meeting of the American Academy of Dermatology.
In a placebo-controlled trial of 160 patients with pruritic psoriasis, CT327 reduced itch by a mean of 60%, said Dr. Yosipovitch, chair of dermatology at the Temple University, Philadelphia.
"Sixty percent is not 90%," he said. "But when we consider that most of the antipruritic drugs have about a 15% efficacy in comparison to a vehicle, this is an effect we should consider. It’s well tolerated, and there were no application site reactions. I see potential in this drug."
CT327, developed by Creabilis, is a selective kinase inhibitor that targets TrkA, the receptor for nerve growth factor (NGF). NGF, which is overexpressed in atopic dermatitis and psoriatic skin, sensitizes neural networks that transmit the itch sensation to the brain, Dr. Yosipovitch said.
The phase IIb study comprised 160 adults with mild to moderate psoriasis, with at least a 10% body surface area involvement. Their mean score on the modified Psoriasis Area and Severity Index (mPASI) was about 9. Most (97%) had pruritus; 69% reported this as at least moderate, with a score of 40 on a 100-point visual analog scale (VAS). A third of the group reported severe pruritus, with a VAS of more than 70. They were randomized to four groups: an emollient placebo or CT327 in concentrations of 0.05%, 0.1%, or 0.5%.
The primary endpoint was reduction on a validated 100-point pruritus visual analog scale. The secondary endpoint was change on the mPASI.
By the end of 8 weeks, most patients (108) had experienced a significant reduction on the pruritus VAS. Reductions were about 35 points for the 0.1% group and 38 points for the 0.05% and 0.5% groups. These changes represented about a 60% decrease overall, Dr. Yosipovitch said.
Changes began early and continued along a steep trajectory, reaching a significant difference from baseline by week 2. However, he pointed out, by that point, the placebo group was similarly improved. This was probably because of the benefit of the daily emollient vehicle, he noted.
The curves began to separate shortly thereafter. By week 4, all of the treatment groups had experienced about a 30-point reduction on the VAS, while the placebo group had actually increased its score slightly. The active groups continued to improve over the next 4 weeks, while the placebo group plateaued at about a 20-point decrease from baseline.
The study concluded with 4 weeks of non–treatment follow-up. During that time, the placebo group stayed at about a 20-point decline and the active groups rebounded to about that level, "suggesting that there really is something going on here biologically," Dr. Yosipovitch said.
Patients using CT327 also experienced significant – although not overwhelming – benefit on the mPASI score. About 45% responded at the 50% level, compared with 23% of those using the emollient placebo. The active groups had similar improvements, with reductions on the mPASI of between 3 and 4 points. Those in the placebo group experienced about a 1-point improvement.
Dr. Yosipovitch said he was "not too impressed" with the mPASI changes. However, he noted, "Breaking the itch-scratch cycle at its source may have broader effects on other symptoms of psoriasis."
Based on the study results, Creabilis has decided to move forward into phase III testing with the 0.05% concentration. "Preparations are already underway," Dr. Yosipovitch said in an interview. "We also have a second phase IIb study ongoing that is investigating CT327 in patients with atopic dermatitis, which will be used to help finalize our phase III programs. This study should be completed in the middle of this year."
The NGF/TrkA pathway is implicated in other disorders as well, Dr. Yosipovitch said. CT327 might be investigated in some of these.
"These include disorders [in which] pruritus is important, such as pruritus with cutaneous T cell lymphoma, where NGF/TrkA has been shown to play a role. The pathway has also been shown to play an important role in chronic neuropathic and inflammatory pain. Molecules targeting NGF have already shown clinical efficacy."
Creabilis is investigating a related molecule, CT340, an inhibitor of both TrkA and MAP2K kinase, for treating those chronic pain conditions.
Creabilis funded the study. Dr. Yosipovitch is on the company’s scientific advisory board.
On Twitter @alz_gal
AT THE AAD ANNUAL MEETING
Major finding: The investigational drug CT327 reduced pruritus scores by 60% in a group of pruritic psoriatic patients.
Data source: The randomized, placebo-controlled trial of 160 patients.
Disclosures: Creabilis funded the study. Dr. Gil Yosipovitch is on the company’s scientific advisory board.
Tofacitinib found noninferior to etanercept for treating psoriasis
DENVER – Key results from a phase III trial found that an investigational Janus kinase inhibitor was noninferior to a biologic agent for treating psoriasis.
During a late-breaking session at the annual meeting of the American Academy of Dermatology, Dr. Fernando Valenzuela presented findings from a multicenter, double-dummy 12-week comparison of tofacitinib vs. etanercept or placebo for the treatment of patients with moderate to severe psoriasis. Tofacitinib is an investigational Janus kinase inhibitor being developed by Pfizer.
Twice-daily tofacitinib at 5 mg and 10 mg was effective in patients with moderate to severe psoriasis, and the efficacy of twice-daily tofacitinib at 10 mg was similar to twice-weekly etanercept at 50 mg and was superior to placebo.
"Plaque psoriasis is a chronic, immune-mediated systemic disease [that] can lead to major quality of life impairment in its moderate to severe forms," said Dr. Valenzuela of the department of dermatology at the University of Chile Clinical Hospital, Santiago. "The JAK/STAT [Janus kinase/signal transducers and activators of transcription] signaling pathway is implicated in the pathogenesis of chronic immune-mediated diseases including psoriasis. Tofacitinib has previously demonstrated significant efficacy in a placebo-controlled, phase II study in patients with moderate to severe plaque psoriasis."
In the 12-week multicenter trial known as OPT Compare, which was conducted in 23 countries outside of North America, he and his associates assessed the noninferiority/superiority of two doses of oral tofacitinib vs. high-dose etanercept or placebo. To be eligible for the trial, patients had to be at least 18 years of age; have moderate to severe chronic plaque psoriasis; be candidates for systemic therapy or phototherapy; have a Psoriasis Area and Severity Index (PASI) of 12 or greater; have a Physician’s Global Assessment (PGA) of moderate or severe; have psoriasis involvement of 10% or greater body surface area; and had failed to respond, tolerate, or have a contraindication to at least one systemic therapy. Patients previously treated with etanercept or who previously experienced failure to treatment with a tumor necrosis factor therapy were excluded from the trial.
Patients were randomized 3:3:3:1 in the following fashion over a 12-week period: 329 received tofacitinib b.i.d.; 330 received tofacitinib 10 mg b.i.d.; 335 received etanercept 50 mg subcutaneously twice weekly; and 107 received placebo. The coprimary efficacy endpoints were the proportion of patients achieving at least a 75% reduction in PASI score at week 12 from baseline and a PGA score of "clear" or "almost clear" at week 12. The researchers used a fixed sequence procedure to assess noninferiority/superiority sequentially in order to control overall type 1 error. The mean age of patients was 44 years and about 82% of patients had moderate disease.
Dr. Valenzuela reported that at 12 weeks, 39.5%, 63.6%, 58.8%, and 5.6% of patients achieved PASI 75 responses and 47.1%, 68.2%, 66.3%, and 15.0% achieved PGA responses with tofacitinib 5 mg twice daily, tofacitinib 10 mg twice daily, etanercept 50 mg twice weekly, and placebo, respectively. Tofacitinib 10 mg twice a day was noninferior to etanercept and superior to placebo (P less than .0001), but tofacitinib 5 mg twice a day did not meet the noninferiority criterion vs. etanercept.
Over 12 weeks, the safety and tolerability of either dose of tofacitinib was similar to that of etanercept. "The most frequent adverse events were infections, most commonly nasopharyngitis and upper respiratory tract infection," Dr. Valenzuela said. "Hypercholesterolemia and dyslipidemia were more common in tofacitinib recipients, as was an increase in creatine phosphokinase*." Injection site reactions were most frequent among etanercept recipients.
The study was sponsored by Pfizer. Dr. Valenzuela disclosed that he has been a principal investigator, member of a scientific advisory board, or speaker for Pfizer, Janssen, Eli Lilly, Merck, and Novartis.
*Correction, 3/27/2014: An earlier version of this article misstated the term creatine phosphokinase.
DENVER – Key results from a phase III trial found that an investigational Janus kinase inhibitor was noninferior to a biologic agent for treating psoriasis.
During a late-breaking session at the annual meeting of the American Academy of Dermatology, Dr. Fernando Valenzuela presented findings from a multicenter, double-dummy 12-week comparison of tofacitinib vs. etanercept or placebo for the treatment of patients with moderate to severe psoriasis. Tofacitinib is an investigational Janus kinase inhibitor being developed by Pfizer.
Twice-daily tofacitinib at 5 mg and 10 mg was effective in patients with moderate to severe psoriasis, and the efficacy of twice-daily tofacitinib at 10 mg was similar to twice-weekly etanercept at 50 mg and was superior to placebo.
"Plaque psoriasis is a chronic, immune-mediated systemic disease [that] can lead to major quality of life impairment in its moderate to severe forms," said Dr. Valenzuela of the department of dermatology at the University of Chile Clinical Hospital, Santiago. "The JAK/STAT [Janus kinase/signal transducers and activators of transcription] signaling pathway is implicated in the pathogenesis of chronic immune-mediated diseases including psoriasis. Tofacitinib has previously demonstrated significant efficacy in a placebo-controlled, phase II study in patients with moderate to severe plaque psoriasis."
In the 12-week multicenter trial known as OPT Compare, which was conducted in 23 countries outside of North America, he and his associates assessed the noninferiority/superiority of two doses of oral tofacitinib vs. high-dose etanercept or placebo. To be eligible for the trial, patients had to be at least 18 years of age; have moderate to severe chronic plaque psoriasis; be candidates for systemic therapy or phototherapy; have a Psoriasis Area and Severity Index (PASI) of 12 or greater; have a Physician’s Global Assessment (PGA) of moderate or severe; have psoriasis involvement of 10% or greater body surface area; and had failed to respond, tolerate, or have a contraindication to at least one systemic therapy. Patients previously treated with etanercept or who previously experienced failure to treatment with a tumor necrosis factor therapy were excluded from the trial.
Patients were randomized 3:3:3:1 in the following fashion over a 12-week period: 329 received tofacitinib b.i.d.; 330 received tofacitinib 10 mg b.i.d.; 335 received etanercept 50 mg subcutaneously twice weekly; and 107 received placebo. The coprimary efficacy endpoints were the proportion of patients achieving at least a 75% reduction in PASI score at week 12 from baseline and a PGA score of "clear" or "almost clear" at week 12. The researchers used a fixed sequence procedure to assess noninferiority/superiority sequentially in order to control overall type 1 error. The mean age of patients was 44 years and about 82% of patients had moderate disease.
Dr. Valenzuela reported that at 12 weeks, 39.5%, 63.6%, 58.8%, and 5.6% of patients achieved PASI 75 responses and 47.1%, 68.2%, 66.3%, and 15.0% achieved PGA responses with tofacitinib 5 mg twice daily, tofacitinib 10 mg twice daily, etanercept 50 mg twice weekly, and placebo, respectively. Tofacitinib 10 mg twice a day was noninferior to etanercept and superior to placebo (P less than .0001), but tofacitinib 5 mg twice a day did not meet the noninferiority criterion vs. etanercept.
Over 12 weeks, the safety and tolerability of either dose of tofacitinib was similar to that of etanercept. "The most frequent adverse events were infections, most commonly nasopharyngitis and upper respiratory tract infection," Dr. Valenzuela said. "Hypercholesterolemia and dyslipidemia were more common in tofacitinib recipients, as was an increase in creatine phosphokinase*." Injection site reactions were most frequent among etanercept recipients.
The study was sponsored by Pfizer. Dr. Valenzuela disclosed that he has been a principal investigator, member of a scientific advisory board, or speaker for Pfizer, Janssen, Eli Lilly, Merck, and Novartis.
*Correction, 3/27/2014: An earlier version of this article misstated the term creatine phosphokinase.
DENVER – Key results from a phase III trial found that an investigational Janus kinase inhibitor was noninferior to a biologic agent for treating psoriasis.
During a late-breaking session at the annual meeting of the American Academy of Dermatology, Dr. Fernando Valenzuela presented findings from a multicenter, double-dummy 12-week comparison of tofacitinib vs. etanercept or placebo for the treatment of patients with moderate to severe psoriasis. Tofacitinib is an investigational Janus kinase inhibitor being developed by Pfizer.
Twice-daily tofacitinib at 5 mg and 10 mg was effective in patients with moderate to severe psoriasis, and the efficacy of twice-daily tofacitinib at 10 mg was similar to twice-weekly etanercept at 50 mg and was superior to placebo.
"Plaque psoriasis is a chronic, immune-mediated systemic disease [that] can lead to major quality of life impairment in its moderate to severe forms," said Dr. Valenzuela of the department of dermatology at the University of Chile Clinical Hospital, Santiago. "The JAK/STAT [Janus kinase/signal transducers and activators of transcription] signaling pathway is implicated in the pathogenesis of chronic immune-mediated diseases including psoriasis. Tofacitinib has previously demonstrated significant efficacy in a placebo-controlled, phase II study in patients with moderate to severe plaque psoriasis."
In the 12-week multicenter trial known as OPT Compare, which was conducted in 23 countries outside of North America, he and his associates assessed the noninferiority/superiority of two doses of oral tofacitinib vs. high-dose etanercept or placebo. To be eligible for the trial, patients had to be at least 18 years of age; have moderate to severe chronic plaque psoriasis; be candidates for systemic therapy or phototherapy; have a Psoriasis Area and Severity Index (PASI) of 12 or greater; have a Physician’s Global Assessment (PGA) of moderate or severe; have psoriasis involvement of 10% or greater body surface area; and had failed to respond, tolerate, or have a contraindication to at least one systemic therapy. Patients previously treated with etanercept or who previously experienced failure to treatment with a tumor necrosis factor therapy were excluded from the trial.
Patients were randomized 3:3:3:1 in the following fashion over a 12-week period: 329 received tofacitinib b.i.d.; 330 received tofacitinib 10 mg b.i.d.; 335 received etanercept 50 mg subcutaneously twice weekly; and 107 received placebo. The coprimary efficacy endpoints were the proportion of patients achieving at least a 75% reduction in PASI score at week 12 from baseline and a PGA score of "clear" or "almost clear" at week 12. The researchers used a fixed sequence procedure to assess noninferiority/superiority sequentially in order to control overall type 1 error. The mean age of patients was 44 years and about 82% of patients had moderate disease.
Dr. Valenzuela reported that at 12 weeks, 39.5%, 63.6%, 58.8%, and 5.6% of patients achieved PASI 75 responses and 47.1%, 68.2%, 66.3%, and 15.0% achieved PGA responses with tofacitinib 5 mg twice daily, tofacitinib 10 mg twice daily, etanercept 50 mg twice weekly, and placebo, respectively. Tofacitinib 10 mg twice a day was noninferior to etanercept and superior to placebo (P less than .0001), but tofacitinib 5 mg twice a day did not meet the noninferiority criterion vs. etanercept.
Over 12 weeks, the safety and tolerability of either dose of tofacitinib was similar to that of etanercept. "The most frequent adverse events were infections, most commonly nasopharyngitis and upper respiratory tract infection," Dr. Valenzuela said. "Hypercholesterolemia and dyslipidemia were more common in tofacitinib recipients, as was an increase in creatine phosphokinase*." Injection site reactions were most frequent among etanercept recipients.
The study was sponsored by Pfizer. Dr. Valenzuela disclosed that he has been a principal investigator, member of a scientific advisory board, or speaker for Pfizer, Janssen, Eli Lilly, Merck, and Novartis.
*Correction, 3/27/2014: An earlier version of this article misstated the term creatine phosphokinase.
AT THE AAD ANNUAL MEETING
Major finding: Twice-daily tofacitinib at 5 mg and 10 mg was effective in patients with moderate to severe psoriasis, and the efficacy of twice-daily tofacitinib at 10 mg was similar to twice-weekly etanercept at 50 mg and was superior to placebo.
Data source: A phase III, multicenter, double dummy trial in which patients with moderate to severe psoriasis were randomized 3:3:3:1 in the following fashion over a 12-week period: 329 received tofacitinib twice a day; 330 received tofacitinib 10 mg twice a day; 335 received etanercept 50 mg subcutaneously twice weekly; and 107 received placebo.
Disclosures: The study was sponsored by Pfizer. Dr. Valenzuela disclosed that he has been a principal investigator, member of a scientific advisory board, or speaker for Pfizer, Janssen, Eli Lilly, Merck, and Novartis.