User login
VIDEO: As biosimilars arrive in U.S., treatment questions arise
MAUI, HAWAII – There’s been fairly brisk uptake of biosimilar infliximab in Europe, both for new patients and as a possible switch from Remicade.
That biosimilar infliximab, Remsima, was submitted for U.S. approval in 2014, and a Food and Drug Administration advisory panel recently recommended approval of a biosimilar for filgrastim (Neupogen).
The agents are in the vanguard of what is sure to be an expanding market in the United States as biologics come off patent. Additional infliximab replacements are in the works, as well as biosimilars for etanercept, adalimumab, rituximab, and others.
For rheumatologists, that could mean less expensive treatments for patients, but it’s also likely to make treatment more complicated. Given the complexity of the molecules, the differences between biosimilars and familiar brands could be a bit more marked than those between small-molecule generics and their branded counterparts.
In a video interview at the 2015 Rheumatology Winter Clinical Symposium, Dr. Arthur F. Kavanaugh, a rheumatology professor at the University of California, San Diego, outlined the latest developments and shared his thoughts on the rapidly evolving field.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
MAUI, HAWAII – There’s been fairly brisk uptake of biosimilar infliximab in Europe, both for new patients and as a possible switch from Remicade.
That biosimilar infliximab, Remsima, was submitted for U.S. approval in 2014, and a Food and Drug Administration advisory panel recently recommended approval of a biosimilar for filgrastim (Neupogen).
The agents are in the vanguard of what is sure to be an expanding market in the United States as biologics come off patent. Additional infliximab replacements are in the works, as well as biosimilars for etanercept, adalimumab, rituximab, and others.
For rheumatologists, that could mean less expensive treatments for patients, but it’s also likely to make treatment more complicated. Given the complexity of the molecules, the differences between biosimilars and familiar brands could be a bit more marked than those between small-molecule generics and their branded counterparts.
In a video interview at the 2015 Rheumatology Winter Clinical Symposium, Dr. Arthur F. Kavanaugh, a rheumatology professor at the University of California, San Diego, outlined the latest developments and shared his thoughts on the rapidly evolving field.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
MAUI, HAWAII – There’s been fairly brisk uptake of biosimilar infliximab in Europe, both for new patients and as a possible switch from Remicade.
That biosimilar infliximab, Remsima, was submitted for U.S. approval in 2014, and a Food and Drug Administration advisory panel recently recommended approval of a biosimilar for filgrastim (Neupogen).
The agents are in the vanguard of what is sure to be an expanding market in the United States as biologics come off patent. Additional infliximab replacements are in the works, as well as biosimilars for etanercept, adalimumab, rituximab, and others.
For rheumatologists, that could mean less expensive treatments for patients, but it’s also likely to make treatment more complicated. Given the complexity of the molecules, the differences between biosimilars and familiar brands could be a bit more marked than those between small-molecule generics and their branded counterparts.
In a video interview at the 2015 Rheumatology Winter Clinical Symposium, Dr. Arthur F. Kavanaugh, a rheumatology professor at the University of California, San Diego, outlined the latest developments and shared his thoughts on the rapidly evolving field.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT RWCS 2015
Unique psoriatic arthritis genetic risk loci discovered
Several genetic risk loci unique to psoriatic arthritis have been identified in a study of samples from 1,962 patients with the disease and 8,923 healthy population controls of white ancestry, according to Dr. John Bowes and his associates.
The investigators used Immunochip genotyping array to fine map previously reported immune-related susceptibility loci, including known psoriasis susceptibility loci, to identify novel psoriatic arthritis (PsA) susceptibility loci.
They found a risk locus specific to PsA at chromosome 5q31, and they detected PsA risk variants at chromosome 1q31, a known psoriasis susceptibility locus. They found independent associations between PsA and three human leukocyte antigen (HLA) genes as well as three non-HLA loci. Finally, they determined that the PsA genetic risk variants preferentially associated with epigenetic markers of open chromatin in CD8+ memory primary T cells, supporting “their importance in the underlying disease mechanism.”
Until this point, the majority of risk loci associated with PsA were shared with psoriasis, but the discovery of unique risk loci could begin to explain fundamental differences between psoriasis and PsA, the investigators said.
Read the full study published Feb. 5 in Nature Communications (doi:10.1038/ncomms7046).
Several genetic risk loci unique to psoriatic arthritis have been identified in a study of samples from 1,962 patients with the disease and 8,923 healthy population controls of white ancestry, according to Dr. John Bowes and his associates.
The investigators used Immunochip genotyping array to fine map previously reported immune-related susceptibility loci, including known psoriasis susceptibility loci, to identify novel psoriatic arthritis (PsA) susceptibility loci.
They found a risk locus specific to PsA at chromosome 5q31, and they detected PsA risk variants at chromosome 1q31, a known psoriasis susceptibility locus. They found independent associations between PsA and three human leukocyte antigen (HLA) genes as well as three non-HLA loci. Finally, they determined that the PsA genetic risk variants preferentially associated with epigenetic markers of open chromatin in CD8+ memory primary T cells, supporting “their importance in the underlying disease mechanism.”
Until this point, the majority of risk loci associated with PsA were shared with psoriasis, but the discovery of unique risk loci could begin to explain fundamental differences between psoriasis and PsA, the investigators said.
Read the full study published Feb. 5 in Nature Communications (doi:10.1038/ncomms7046).
Several genetic risk loci unique to psoriatic arthritis have been identified in a study of samples from 1,962 patients with the disease and 8,923 healthy population controls of white ancestry, according to Dr. John Bowes and his associates.
The investigators used Immunochip genotyping array to fine map previously reported immune-related susceptibility loci, including known psoriasis susceptibility loci, to identify novel psoriatic arthritis (PsA) susceptibility loci.
They found a risk locus specific to PsA at chromosome 5q31, and they detected PsA risk variants at chromosome 1q31, a known psoriasis susceptibility locus. They found independent associations between PsA and three human leukocyte antigen (HLA) genes as well as three non-HLA loci. Finally, they determined that the PsA genetic risk variants preferentially associated with epigenetic markers of open chromatin in CD8+ memory primary T cells, supporting “their importance in the underlying disease mechanism.”
Until this point, the majority of risk loci associated with PsA were shared with psoriasis, but the discovery of unique risk loci could begin to explain fundamental differences between psoriasis and PsA, the investigators said.
Read the full study published Feb. 5 in Nature Communications (doi:10.1038/ncomms7046).
Manage Your Dermatology Practice: Managing Difficult Patient Encounters
Difficult patient encounters in the dermatology office can be navigated through honest physician-patient communication regarding problems within the office and insurance coverage. Dr. Gary Goldenberg provides tips on communicating with patients about cosmetic procedures that may be noncovered services as well as diagnoses such as melanoma and psoriasis. He also advises how to work through a long list of questions patients may bring to their visit.
Difficult patient encounters in the dermatology office can be navigated through honest physician-patient communication regarding problems within the office and insurance coverage. Dr. Gary Goldenberg provides tips on communicating with patients about cosmetic procedures that may be noncovered services as well as diagnoses such as melanoma and psoriasis. He also advises how to work through a long list of questions patients may bring to their visit.
Difficult patient encounters in the dermatology office can be navigated through honest physician-patient communication regarding problems within the office and insurance coverage. Dr. Gary Goldenberg provides tips on communicating with patients about cosmetic procedures that may be noncovered services as well as diagnoses such as melanoma and psoriasis. He also advises how to work through a long list of questions patients may bring to their visit.
Most cost-effective psoriasis treatment: methotrexate
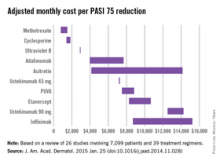
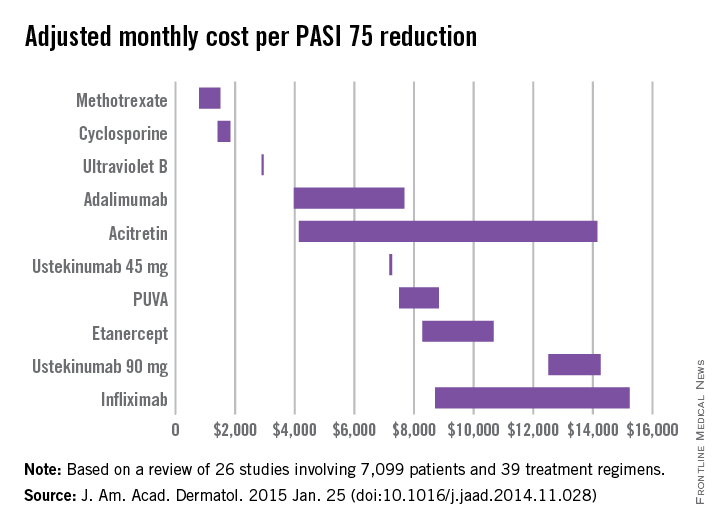
Methotrexate is the most cost-effective systemic treatment for psoriasis in terms of the number of patients needed to treat to achieve a PASI 75, according to a literature review in the Journal of the American Academy of Dermatology.
Methotrexate (2.5-mg tablet) cost an adjusted $794-$1,503 a month per number needed to treat (NNT) for a 75% reduction in Psoriasis Area and Severity Index (PASI 75) score, Dr. Logan S. D’Souza and Dr. Michael J. Payette of the department of dermatology at the University of Connecticut in Farmington reported (J. Am. Acad. Dermatol. 2015 Jan. 25 [doi:10.1016/j.jaad.2014.11.028]).
Coming in second was cyclosporine (25- and 100-mg tablets) at $1,410-$1,844 in monthly cost per PASI 75, followed by narrow-band ultraviolet B light phototherapy at $2,925 and adalimumab (40-mg subcutaneous injection) at $3,975-$7,679, the investigators said.
The most expensive of the 10 Food and Drug Administration–approved treatments included in the study was infliximab (100-mg vial) at $8,705-$15,236 per month. The next most expensive treatment was ustekinumab (90-mg subcutaneous injection), which cost $12,505-$14,257 per month.
“While our data indicate that methotrexate and cyclosporine are more cost efficacious in reaching PASI 75 than other medications approved by the FDA, their potential toxicities may limit their long-term use,” Dr. D’Souza and Dr. Payette noted. “Our study is not meant to dictate treatments based solely on costs; rather, our analysis should help both clinicians and patients in choosing between potential treatments,” they added.
For this analysis, 26 studies involving 7,099 patients and 39 treatment regimens met the investigators’ search criteria.
Dr. D’Souza declared no conflicts of interest. Dr. Payette has been an adviser to Amgen.
Methotrexate is the most cost-effective systemic treatment for psoriasis in terms of the number of patients needed to treat to achieve a PASI 75, according to a literature review in the Journal of the American Academy of Dermatology.
Methotrexate (2.5-mg tablet) cost an adjusted $794-$1,503 a month per number needed to treat (NNT) for a 75% reduction in Psoriasis Area and Severity Index (PASI 75) score, Dr. Logan S. D’Souza and Dr. Michael J. Payette of the department of dermatology at the University of Connecticut in Farmington reported (J. Am. Acad. Dermatol. 2015 Jan. 25 [doi:10.1016/j.jaad.2014.11.028]).
Coming in second was cyclosporine (25- and 100-mg tablets) at $1,410-$1,844 in monthly cost per PASI 75, followed by narrow-band ultraviolet B light phototherapy at $2,925 and adalimumab (40-mg subcutaneous injection) at $3,975-$7,679, the investigators said.
The most expensive of the 10 Food and Drug Administration–approved treatments included in the study was infliximab (100-mg vial) at $8,705-$15,236 per month. The next most expensive treatment was ustekinumab (90-mg subcutaneous injection), which cost $12,505-$14,257 per month.
“While our data indicate that methotrexate and cyclosporine are more cost efficacious in reaching PASI 75 than other medications approved by the FDA, their potential toxicities may limit their long-term use,” Dr. D’Souza and Dr. Payette noted. “Our study is not meant to dictate treatments based solely on costs; rather, our analysis should help both clinicians and patients in choosing between potential treatments,” they added.
For this analysis, 26 studies involving 7,099 patients and 39 treatment regimens met the investigators’ search criteria.
Dr. D’Souza declared no conflicts of interest. Dr. Payette has been an adviser to Amgen.
Methotrexate is the most cost-effective systemic treatment for psoriasis in terms of the number of patients needed to treat to achieve a PASI 75, according to a literature review in the Journal of the American Academy of Dermatology.
Methotrexate (2.5-mg tablet) cost an adjusted $794-$1,503 a month per number needed to treat (NNT) for a 75% reduction in Psoriasis Area and Severity Index (PASI 75) score, Dr. Logan S. D’Souza and Dr. Michael J. Payette of the department of dermatology at the University of Connecticut in Farmington reported (J. Am. Acad. Dermatol. 2015 Jan. 25 [doi:10.1016/j.jaad.2014.11.028]).
Coming in second was cyclosporine (25- and 100-mg tablets) at $1,410-$1,844 in monthly cost per PASI 75, followed by narrow-band ultraviolet B light phototherapy at $2,925 and adalimumab (40-mg subcutaneous injection) at $3,975-$7,679, the investigators said.
The most expensive of the 10 Food and Drug Administration–approved treatments included in the study was infliximab (100-mg vial) at $8,705-$15,236 per month. The next most expensive treatment was ustekinumab (90-mg subcutaneous injection), which cost $12,505-$14,257 per month.
“While our data indicate that methotrexate and cyclosporine are more cost efficacious in reaching PASI 75 than other medications approved by the FDA, their potential toxicities may limit their long-term use,” Dr. D’Souza and Dr. Payette noted. “Our study is not meant to dictate treatments based solely on costs; rather, our analysis should help both clinicians and patients in choosing between potential treatments,” they added.
For this analysis, 26 studies involving 7,099 patients and 39 treatment regimens met the investigators’ search criteria.
Dr. D’Souza declared no conflicts of interest. Dr. Payette has been an adviser to Amgen.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Survey: Psoriasis/psoriatic arthritis undertreated
ORLANDO – Patients with psoriasis and psoriatic arthritis have a high level of dissatisfaction with current treatment options and have numerous unmet health care needs, according to the U.S. findings from the Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP) survey.
Of 1,005 U.S. adults who participated in the population-based telephone survey, 735 had psoriasis diagnosed by a health care provider, and 270 had psoriatic arthritis diagnosed – with or without a diagnosis of psoriasis. Half of those surveyed reported dissatisfaction with available oral and biologic therapies, Dr. Mark Lebwohl of Mount Sinai Hospital, New York, and his colleagues reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
Most respondents with psoriasis reported moderate symptoms (40%), with about 30% each reporting mild or severe symptoms. Itching was the most bothersome symptom, occurring in about 42% of patients, followed by flaking (26%) and scales (22%). Itching was the greatest contributor to estimates of disease severity.
Psoriatic arthritis patients were more likely to report severe symptoms (56%), while 12% reported mild symptoms, and about a third reported moderate symptoms. Itching was the most bothersome symptom among these patients, followed by pain (21%) and flaking (19%). Pain and joint swelling were the greatest contributors to estimates of disease severity in this group, the investigators said.
The majority of respondents (57%) had seen a doctor in the past year. For psoriasis patients, that doctor was most often a dermatologist (57%), while psoriatic arthritis patients most often saw a rheumatologist (38%).
A third of patients most often saw a primary care physician.
The main reasons cited for not having seen a doctor in the past year were having mild or no symptoms, and a feeling that a health care provider would be unable to help.
Of concern, patterns reported by the patients indicated widespread undertreatment of psoriasis and psoriatic arthritis, they said.
More than one in five psoriasis patients (22%) reported no treatment at the time of the survey, and although most described their disease as moderate or severe, only 23% reported ever discussing the use of conventional oral or biologic therapies with their health care providers, and only 9% were currently receiving systemic therapy; most were using prescription topical therapy.
Psoriatic arthritis patients were much more likely than were psoriasis patients to have ever used a conventional oral or biologic therapy, but only 50% were receiving systemic therapy, while 26% reported use of biologic therapy and 24% reported use of an oral therapy.
Methotrexate was the conventional oral therapy used most often (68% of the time), followed by cyclosporine and acitretin (in 14% and 12% of patients, respectively), and etanercept was the biologic therapy used most often (56%) followed by adalimumab (used by 48%).
The most common reasons for discontinuing treatment were concerns about safety, issues with tolerability, and lack or loss of effectiveness. The greatest contributors to treatment burden among those using conventional oral therapies were side effects and the need for laboratory monitoring, and the greatest contributors among those using biologics were concerns about long-term safety, anxiety or fear, pain, and inconvenience associated with self injection.
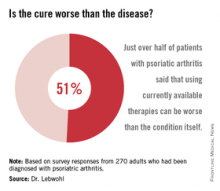
More than half of all respondents with psoriatic arthritis (51%) agreed that using currently available therapies can be worse than the condition itself, and 88% said better therapies are needed.
These results underscore the need for improved screening, as many patients with joint symptoms had not been diagnosed with psoriatic arthritis. They also demonstrate a need for improved assessment of disease severity; pruritus – which was reported as the most bothersome symptom – “is not included in most assessment tools,” the investigators noted.
Further, the results highlight the need for a higher level of treatment in many patients, they said, explaining that survey responses indicated substantial undertreatment in numerous cases. Many patients were receiving no treatment or only topical treatment, and many who were experiencing symptoms were not currently seeking care from a health care provider.
The MAPP survey is the first large-scale, multinational, population-based survey of psoriasis and psoriatic arthritis patients. The survey included 3,426 patients and 781 physicians in North America and Europe. The U.S. findings, which indicate little systematic implementation of treatment algorithms as well as widespread undertreatment, are particularly important given that psoriasis affects 3.2% of adults in the United States, and that 30% of those patients have psoriatic arthritis, the researchers noted.
Most of these patients report that their condition affects their emotional well-being, their quality of life, and their daily activities, they noted.
“These findings deserve attention and action to improve the care and lives of patients with psoriasis and psoriatic arthritis,” they concluded.
The survey was sponsored by Celgene, which markets apremilast (Otezla). Dr. Lebwohl disclosed ties to numerous pharmaceutical companies, including several that market drugs for psoriasis and psoriatic arthritis.
ORLANDO – Patients with psoriasis and psoriatic arthritis have a high level of dissatisfaction with current treatment options and have numerous unmet health care needs, according to the U.S. findings from the Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP) survey.
Of 1,005 U.S. adults who participated in the population-based telephone survey, 735 had psoriasis diagnosed by a health care provider, and 270 had psoriatic arthritis diagnosed – with or without a diagnosis of psoriasis. Half of those surveyed reported dissatisfaction with available oral and biologic therapies, Dr. Mark Lebwohl of Mount Sinai Hospital, New York, and his colleagues reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
Most respondents with psoriasis reported moderate symptoms (40%), with about 30% each reporting mild or severe symptoms. Itching was the most bothersome symptom, occurring in about 42% of patients, followed by flaking (26%) and scales (22%). Itching was the greatest contributor to estimates of disease severity.
Psoriatic arthritis patients were more likely to report severe symptoms (56%), while 12% reported mild symptoms, and about a third reported moderate symptoms. Itching was the most bothersome symptom among these patients, followed by pain (21%) and flaking (19%). Pain and joint swelling were the greatest contributors to estimates of disease severity in this group, the investigators said.
The majority of respondents (57%) had seen a doctor in the past year. For psoriasis patients, that doctor was most often a dermatologist (57%), while psoriatic arthritis patients most often saw a rheumatologist (38%).
A third of patients most often saw a primary care physician.
The main reasons cited for not having seen a doctor in the past year were having mild or no symptoms, and a feeling that a health care provider would be unable to help.
Of concern, patterns reported by the patients indicated widespread undertreatment of psoriasis and psoriatic arthritis, they said.
More than one in five psoriasis patients (22%) reported no treatment at the time of the survey, and although most described their disease as moderate or severe, only 23% reported ever discussing the use of conventional oral or biologic therapies with their health care providers, and only 9% were currently receiving systemic therapy; most were using prescription topical therapy.
Psoriatic arthritis patients were much more likely than were psoriasis patients to have ever used a conventional oral or biologic therapy, but only 50% were receiving systemic therapy, while 26% reported use of biologic therapy and 24% reported use of an oral therapy.
Methotrexate was the conventional oral therapy used most often (68% of the time), followed by cyclosporine and acitretin (in 14% and 12% of patients, respectively), and etanercept was the biologic therapy used most often (56%) followed by adalimumab (used by 48%).
The most common reasons for discontinuing treatment were concerns about safety, issues with tolerability, and lack or loss of effectiveness. The greatest contributors to treatment burden among those using conventional oral therapies were side effects and the need for laboratory monitoring, and the greatest contributors among those using biologics were concerns about long-term safety, anxiety or fear, pain, and inconvenience associated with self injection.
More than half of all respondents with psoriatic arthritis (51%) agreed that using currently available therapies can be worse than the condition itself, and 88% said better therapies are needed.
These results underscore the need for improved screening, as many patients with joint symptoms had not been diagnosed with psoriatic arthritis. They also demonstrate a need for improved assessment of disease severity; pruritus – which was reported as the most bothersome symptom – “is not included in most assessment tools,” the investigators noted.
Further, the results highlight the need for a higher level of treatment in many patients, they said, explaining that survey responses indicated substantial undertreatment in numerous cases. Many patients were receiving no treatment or only topical treatment, and many who were experiencing symptoms were not currently seeking care from a health care provider.
The MAPP survey is the first large-scale, multinational, population-based survey of psoriasis and psoriatic arthritis patients. The survey included 3,426 patients and 781 physicians in North America and Europe. The U.S. findings, which indicate little systematic implementation of treatment algorithms as well as widespread undertreatment, are particularly important given that psoriasis affects 3.2% of adults in the United States, and that 30% of those patients have psoriatic arthritis, the researchers noted.
Most of these patients report that their condition affects their emotional well-being, their quality of life, and their daily activities, they noted.
“These findings deserve attention and action to improve the care and lives of patients with psoriasis and psoriatic arthritis,” they concluded.
The survey was sponsored by Celgene, which markets apremilast (Otezla). Dr. Lebwohl disclosed ties to numerous pharmaceutical companies, including several that market drugs for psoriasis and psoriatic arthritis.
ORLANDO – Patients with psoriasis and psoriatic arthritis have a high level of dissatisfaction with current treatment options and have numerous unmet health care needs, according to the U.S. findings from the Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP) survey.
Of 1,005 U.S. adults who participated in the population-based telephone survey, 735 had psoriasis diagnosed by a health care provider, and 270 had psoriatic arthritis diagnosed – with or without a diagnosis of psoriasis. Half of those surveyed reported dissatisfaction with available oral and biologic therapies, Dr. Mark Lebwohl of Mount Sinai Hospital, New York, and his colleagues reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
Most respondents with psoriasis reported moderate symptoms (40%), with about 30% each reporting mild or severe symptoms. Itching was the most bothersome symptom, occurring in about 42% of patients, followed by flaking (26%) and scales (22%). Itching was the greatest contributor to estimates of disease severity.
Psoriatic arthritis patients were more likely to report severe symptoms (56%), while 12% reported mild symptoms, and about a third reported moderate symptoms. Itching was the most bothersome symptom among these patients, followed by pain (21%) and flaking (19%). Pain and joint swelling were the greatest contributors to estimates of disease severity in this group, the investigators said.
The majority of respondents (57%) had seen a doctor in the past year. For psoriasis patients, that doctor was most often a dermatologist (57%), while psoriatic arthritis patients most often saw a rheumatologist (38%).
A third of patients most often saw a primary care physician.
The main reasons cited for not having seen a doctor in the past year were having mild or no symptoms, and a feeling that a health care provider would be unable to help.
Of concern, patterns reported by the patients indicated widespread undertreatment of psoriasis and psoriatic arthritis, they said.
More than one in five psoriasis patients (22%) reported no treatment at the time of the survey, and although most described their disease as moderate or severe, only 23% reported ever discussing the use of conventional oral or biologic therapies with their health care providers, and only 9% were currently receiving systemic therapy; most were using prescription topical therapy.
Psoriatic arthritis patients were much more likely than were psoriasis patients to have ever used a conventional oral or biologic therapy, but only 50% were receiving systemic therapy, while 26% reported use of biologic therapy and 24% reported use of an oral therapy.
Methotrexate was the conventional oral therapy used most often (68% of the time), followed by cyclosporine and acitretin (in 14% and 12% of patients, respectively), and etanercept was the biologic therapy used most often (56%) followed by adalimumab (used by 48%).
The most common reasons for discontinuing treatment were concerns about safety, issues with tolerability, and lack or loss of effectiveness. The greatest contributors to treatment burden among those using conventional oral therapies were side effects and the need for laboratory monitoring, and the greatest contributors among those using biologics were concerns about long-term safety, anxiety or fear, pain, and inconvenience associated with self injection.
More than half of all respondents with psoriatic arthritis (51%) agreed that using currently available therapies can be worse than the condition itself, and 88% said better therapies are needed.
These results underscore the need for improved screening, as many patients with joint symptoms had not been diagnosed with psoriatic arthritis. They also demonstrate a need for improved assessment of disease severity; pruritus – which was reported as the most bothersome symptom – “is not included in most assessment tools,” the investigators noted.
Further, the results highlight the need for a higher level of treatment in many patients, they said, explaining that survey responses indicated substantial undertreatment in numerous cases. Many patients were receiving no treatment or only topical treatment, and many who were experiencing symptoms were not currently seeking care from a health care provider.
The MAPP survey is the first large-scale, multinational, population-based survey of psoriasis and psoriatic arthritis patients. The survey included 3,426 patients and 781 physicians in North America and Europe. The U.S. findings, which indicate little systematic implementation of treatment algorithms as well as widespread undertreatment, are particularly important given that psoriasis affects 3.2% of adults in the United States, and that 30% of those patients have psoriatic arthritis, the researchers noted.
Most of these patients report that their condition affects their emotional well-being, their quality of life, and their daily activities, they noted.
“These findings deserve attention and action to improve the care and lives of patients with psoriasis and psoriatic arthritis,” they concluded.
The survey was sponsored by Celgene, which markets apremilast (Otezla). Dr. Lebwohl disclosed ties to numerous pharmaceutical companies, including several that market drugs for psoriasis and psoriatic arthritis.
AT THE ODAC CONFERENCE
Key clinical point: Psoriasis and psoriatic arthritis treatment algorithms are underused, and patients are often undertreated.
Major finding: Among survey respondents, 22% were on no treatment, and 23% reported ever discussing conventional oral or biologic therapies with a health care provider.
Data source: A telephone survey (MAPP) of 735 psoriasis and 270 psoriatic arthritis patients.
Disclosures: The survey was sponsored by Celgene, which markets apremilast (Otezla). Dr. Lebwohl disclosed ties to numerous pharmaceutical companies, including several that market drugs for psoriasis and psoriatic arthritis.
Joint mechanical stress: A psoriatic arthritis trigger in genetically susceptible individuals?
Parallel cases of twins developing “symmetrical” dactylitis in two digits following similarly sustained foot injuries support the hypothesis that joint mechanical stress is a major trigger of psoriatic arthritis and dactylitis in genetically susceptible individuals, according to a scientific letter published Jan. 28 in Annals of the Rheumatic Diseases.
A team of researchers led by Dr. Jennifer Ng of Griffith University and Paradise Arthritis and Rheumatology in Southport, Australia, described the cases of identical twins, 54-year old women with a history of psoriasis, both of whom developed psoriatic arthritis of the respective right and left second toes. Both sisters had no arthritis prior to the joint injuries, and one of the patients had to be treated with methotrexate 15 mg weekly with almost complete resolution of the swelling and pain.
Although researchers are still exploring the precise mechanisms that link joint injury and psoriatic arthritis onset, the authors assert they have proof of concept of how site specific injury may result in biomechanical triggering events in genetically susceptible hosts.
Read the entire article here: doi:10.1136/annrheumdis-2014-206784.
Parallel cases of twins developing “symmetrical” dactylitis in two digits following similarly sustained foot injuries support the hypothesis that joint mechanical stress is a major trigger of psoriatic arthritis and dactylitis in genetically susceptible individuals, according to a scientific letter published Jan. 28 in Annals of the Rheumatic Diseases.
A team of researchers led by Dr. Jennifer Ng of Griffith University and Paradise Arthritis and Rheumatology in Southport, Australia, described the cases of identical twins, 54-year old women with a history of psoriasis, both of whom developed psoriatic arthritis of the respective right and left second toes. Both sisters had no arthritis prior to the joint injuries, and one of the patients had to be treated with methotrexate 15 mg weekly with almost complete resolution of the swelling and pain.
Although researchers are still exploring the precise mechanisms that link joint injury and psoriatic arthritis onset, the authors assert they have proof of concept of how site specific injury may result in biomechanical triggering events in genetically susceptible hosts.
Read the entire article here: doi:10.1136/annrheumdis-2014-206784.
Parallel cases of twins developing “symmetrical” dactylitis in two digits following similarly sustained foot injuries support the hypothesis that joint mechanical stress is a major trigger of psoriatic arthritis and dactylitis in genetically susceptible individuals, according to a scientific letter published Jan. 28 in Annals of the Rheumatic Diseases.
A team of researchers led by Dr. Jennifer Ng of Griffith University and Paradise Arthritis and Rheumatology in Southport, Australia, described the cases of identical twins, 54-year old women with a history of psoriasis, both of whom developed psoriatic arthritis of the respective right and left second toes. Both sisters had no arthritis prior to the joint injuries, and one of the patients had to be treated with methotrexate 15 mg weekly with almost complete resolution of the swelling and pain.
Although researchers are still exploring the precise mechanisms that link joint injury and psoriatic arthritis onset, the authors assert they have proof of concept of how site specific injury may result in biomechanical triggering events in genetically susceptible hosts.
Read the entire article here: doi:10.1136/annrheumdis-2014-206784.
Tonsillectomy and Psoriasis
We are all aware that infections, particularly streptococcal infection, can be associated with psoriasis, especially the guttate variety. A logical question emanating from this fact is: Would tonsillectomy and adenoidectomy have any impact on psoriasis and its symptoms?
In a November 2014 article published online in the Journal of the American Academy of Dermatology, Rachakonda et al (doi:10.1016/j.jaad.2014.10.013) performed an extensive literature review to evaluate if tonsillectomy reduces psoriasis severity. The authors searched the following sources: MEDLINE, CINAHL, Cochrane, Embase, Web of Science, and Ovid databases (August 1, 1960, to September 12, 2013). In addition, they executed a manual search of selected references. Through this process, they identified observational studies and clinical trials examining psoriasis after tonsillectomy.
In the analysis, the authors included data from 20 articles from the 53 years they examined. From this literature, they included 545 patients with psoriasis who were either evaluated for or underwent tonsillectomy. Of 410 patients with psoriasis who actually underwent tonsillectomy, 290 experienced improvement in their psoriasis. Although some individuals who underwent tonsillectomy experienced sustained improvement in their disease, others experienced relapse following the procedure. The authors noted that their study was limited. Fifteen of 20 analyzed publications were case reports or series that lacked control groups. In addition, they noted that a publication bias that favored the reporting of improved cases needs to be considered.
Based on this comprehensive systematic review on the effect of tonsillectomy on psoriasis, the authors concluded that although tonsillectomy is effective in ameliorating psoriasis in a subpopulation of patients, there are insufficient data to describe the differences in clinical characteristics between responders versus nonresponders. Tonsillectomy may be a potential option for patients with recalcitrant psoriasis that is associated with occurrences of tonsillitis. Studies with long-term follow-up are needed to elucidate more clearly the extent and persistence of benefit of tonsillectomy in psoriasis.
What’s the issue?
Tonsillectomy represents an intriguing option not commonly considered for those with resistant disease. Based on the current data, will you discuss tonsillectomy with your patients?
We want to know your views! Tell us what you think.
Reader Comment
This concept so intrigued me when I heard Dr. Susan Katz discuss it at the NYU Advances in Medicine conference last June that I had the discussion with one of my patients, and she opted to have tonsillectomy this past fall. So far, she seems to improving, but I have not yet discontinued her long-term biologic therapy. At the same conference, Dr. Katz presented the idea that delaying antibiotic treatment of streptococcal pharyngitis by a few days might actually improve strep clearance rates by allowing the immune system to mount a greater response to the infection. Waiting for culture results before initiating antibiotic therapy might be prudent not only because it might reduce the unnecessary use of antibiotics in non-streptococcal pharyngitis, but because it might actually improve the long-term prognosis of patients (with or without psoriasis) who do have strep by reducing the odds of a chronic carrier state. Thanks for highlighting this area of study. Fascinating to consider that there might be a surgical cure for some psoriatic patients.
—Jennifer Goldwasser, MD (Scarsdale, New York)
We are all aware that infections, particularly streptococcal infection, can be associated with psoriasis, especially the guttate variety. A logical question emanating from this fact is: Would tonsillectomy and adenoidectomy have any impact on psoriasis and its symptoms?
In a November 2014 article published online in the Journal of the American Academy of Dermatology, Rachakonda et al (doi:10.1016/j.jaad.2014.10.013) performed an extensive literature review to evaluate if tonsillectomy reduces psoriasis severity. The authors searched the following sources: MEDLINE, CINAHL, Cochrane, Embase, Web of Science, and Ovid databases (August 1, 1960, to September 12, 2013). In addition, they executed a manual search of selected references. Through this process, they identified observational studies and clinical trials examining psoriasis after tonsillectomy.
In the analysis, the authors included data from 20 articles from the 53 years they examined. From this literature, they included 545 patients with psoriasis who were either evaluated for or underwent tonsillectomy. Of 410 patients with psoriasis who actually underwent tonsillectomy, 290 experienced improvement in their psoriasis. Although some individuals who underwent tonsillectomy experienced sustained improvement in their disease, others experienced relapse following the procedure. The authors noted that their study was limited. Fifteen of 20 analyzed publications were case reports or series that lacked control groups. In addition, they noted that a publication bias that favored the reporting of improved cases needs to be considered.
Based on this comprehensive systematic review on the effect of tonsillectomy on psoriasis, the authors concluded that although tonsillectomy is effective in ameliorating psoriasis in a subpopulation of patients, there are insufficient data to describe the differences in clinical characteristics between responders versus nonresponders. Tonsillectomy may be a potential option for patients with recalcitrant psoriasis that is associated with occurrences of tonsillitis. Studies with long-term follow-up are needed to elucidate more clearly the extent and persistence of benefit of tonsillectomy in psoriasis.
What’s the issue?
Tonsillectomy represents an intriguing option not commonly considered for those with resistant disease. Based on the current data, will you discuss tonsillectomy with your patients?
We want to know your views! Tell us what you think.
Reader Comment
This concept so intrigued me when I heard Dr. Susan Katz discuss it at the NYU Advances in Medicine conference last June that I had the discussion with one of my patients, and she opted to have tonsillectomy this past fall. So far, she seems to improving, but I have not yet discontinued her long-term biologic therapy. At the same conference, Dr. Katz presented the idea that delaying antibiotic treatment of streptococcal pharyngitis by a few days might actually improve strep clearance rates by allowing the immune system to mount a greater response to the infection. Waiting for culture results before initiating antibiotic therapy might be prudent not only because it might reduce the unnecessary use of antibiotics in non-streptococcal pharyngitis, but because it might actually improve the long-term prognosis of patients (with or without psoriasis) who do have strep by reducing the odds of a chronic carrier state. Thanks for highlighting this area of study. Fascinating to consider that there might be a surgical cure for some psoriatic patients.
—Jennifer Goldwasser, MD (Scarsdale, New York)
We are all aware that infections, particularly streptococcal infection, can be associated with psoriasis, especially the guttate variety. A logical question emanating from this fact is: Would tonsillectomy and adenoidectomy have any impact on psoriasis and its symptoms?
In a November 2014 article published online in the Journal of the American Academy of Dermatology, Rachakonda et al (doi:10.1016/j.jaad.2014.10.013) performed an extensive literature review to evaluate if tonsillectomy reduces psoriasis severity. The authors searched the following sources: MEDLINE, CINAHL, Cochrane, Embase, Web of Science, and Ovid databases (August 1, 1960, to September 12, 2013). In addition, they executed a manual search of selected references. Through this process, they identified observational studies and clinical trials examining psoriasis after tonsillectomy.
In the analysis, the authors included data from 20 articles from the 53 years they examined. From this literature, they included 545 patients with psoriasis who were either evaluated for or underwent tonsillectomy. Of 410 patients with psoriasis who actually underwent tonsillectomy, 290 experienced improvement in their psoriasis. Although some individuals who underwent tonsillectomy experienced sustained improvement in their disease, others experienced relapse following the procedure. The authors noted that their study was limited. Fifteen of 20 analyzed publications were case reports or series that lacked control groups. In addition, they noted that a publication bias that favored the reporting of improved cases needs to be considered.
Based on this comprehensive systematic review on the effect of tonsillectomy on psoriasis, the authors concluded that although tonsillectomy is effective in ameliorating psoriasis in a subpopulation of patients, there are insufficient data to describe the differences in clinical characteristics between responders versus nonresponders. Tonsillectomy may be a potential option for patients with recalcitrant psoriasis that is associated with occurrences of tonsillitis. Studies with long-term follow-up are needed to elucidate more clearly the extent and persistence of benefit of tonsillectomy in psoriasis.
What’s the issue?
Tonsillectomy represents an intriguing option not commonly considered for those with resistant disease. Based on the current data, will you discuss tonsillectomy with your patients?
We want to know your views! Tell us what you think.
Reader Comment
This concept so intrigued me when I heard Dr. Susan Katz discuss it at the NYU Advances in Medicine conference last June that I had the discussion with one of my patients, and she opted to have tonsillectomy this past fall. So far, she seems to improving, but I have not yet discontinued her long-term biologic therapy. At the same conference, Dr. Katz presented the idea that delaying antibiotic treatment of streptococcal pharyngitis by a few days might actually improve strep clearance rates by allowing the immune system to mount a greater response to the infection. Waiting for culture results before initiating antibiotic therapy might be prudent not only because it might reduce the unnecessary use of antibiotics in non-streptococcal pharyngitis, but because it might actually improve the long-term prognosis of patients (with or without psoriasis) who do have strep by reducing the odds of a chronic carrier state. Thanks for highlighting this area of study. Fascinating to consider that there might be a surgical cure for some psoriatic patients.
—Jennifer Goldwasser, MD (Scarsdale, New York)
Corticosteroid spray quickly, safely relieves psoriasis scaling
ORLANDO – A topical corticosteroid spray quickly and effectively reduced scaling in patients with plaque psoriasis, according to pooled phase III study results.
After 1 week of twice daily application of 0.25% Topicort spray (TaroPharma, Hawthorne, N.Y.), scaling was clear or almost clear in 29% of 120 treated patients, and was mild in an additional 40% of patients. At 4 weeks, scaling was clear or almost clear in 61% of patients, and mild in an additional 23%, Dr. Brian Keegan of East Windsor, N.J., reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
Additionally, 70% of patients experienced global improvement in psoriasis after 4 weeks of treatment; psoriasis was clear or almost clear in 42% of patients, and mild in 28%, Dr. Keegan said.
Study participants were adults with 10%-86% of body area affected by plaque psoriasis. Most of the patients (58%) were aged 41-to-64 years with a mean affected body surface area of 17%. Most had moderate scaling, but about a third of patients had severe or very severe scaling.
They applied Topicort 0.25% spray twice daily for 4 weeks.
The only adverse events that occurred in more than 1% of patients were dryness, irritation, and pruritus at the application site, which each occurred in 2%-3% of patients.
Treatment with Topicort – a class 1 super-potent corticosteroid indicated for up to 4 weeks of treatment of plaque psoriasis – quickly and safely relieved scaling in the majority of patients treated, Dr. Keegan concluded, noting that the findings are important given that scaling is very common among patients with plaque psoriasis, many of whom find it to be bothersome.
In fact, 94% of more than 17,000 participants in a National Psoriasis Foundation survey reported being bothered by scaling, he noted.
This study was sponsored by TaroPharma.
ORLANDO – A topical corticosteroid spray quickly and effectively reduced scaling in patients with plaque psoriasis, according to pooled phase III study results.
After 1 week of twice daily application of 0.25% Topicort spray (TaroPharma, Hawthorne, N.Y.), scaling was clear or almost clear in 29% of 120 treated patients, and was mild in an additional 40% of patients. At 4 weeks, scaling was clear or almost clear in 61% of patients, and mild in an additional 23%, Dr. Brian Keegan of East Windsor, N.J., reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
Additionally, 70% of patients experienced global improvement in psoriasis after 4 weeks of treatment; psoriasis was clear or almost clear in 42% of patients, and mild in 28%, Dr. Keegan said.
Study participants were adults with 10%-86% of body area affected by plaque psoriasis. Most of the patients (58%) were aged 41-to-64 years with a mean affected body surface area of 17%. Most had moderate scaling, but about a third of patients had severe or very severe scaling.
They applied Topicort 0.25% spray twice daily for 4 weeks.
The only adverse events that occurred in more than 1% of patients were dryness, irritation, and pruritus at the application site, which each occurred in 2%-3% of patients.
Treatment with Topicort – a class 1 super-potent corticosteroid indicated for up to 4 weeks of treatment of plaque psoriasis – quickly and safely relieved scaling in the majority of patients treated, Dr. Keegan concluded, noting that the findings are important given that scaling is very common among patients with plaque psoriasis, many of whom find it to be bothersome.
In fact, 94% of more than 17,000 participants in a National Psoriasis Foundation survey reported being bothered by scaling, he noted.
This study was sponsored by TaroPharma.
ORLANDO – A topical corticosteroid spray quickly and effectively reduced scaling in patients with plaque psoriasis, according to pooled phase III study results.
After 1 week of twice daily application of 0.25% Topicort spray (TaroPharma, Hawthorne, N.Y.), scaling was clear or almost clear in 29% of 120 treated patients, and was mild in an additional 40% of patients. At 4 weeks, scaling was clear or almost clear in 61% of patients, and mild in an additional 23%, Dr. Brian Keegan of East Windsor, N.J., reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
Additionally, 70% of patients experienced global improvement in psoriasis after 4 weeks of treatment; psoriasis was clear or almost clear in 42% of patients, and mild in 28%, Dr. Keegan said.
Study participants were adults with 10%-86% of body area affected by plaque psoriasis. Most of the patients (58%) were aged 41-to-64 years with a mean affected body surface area of 17%. Most had moderate scaling, but about a third of patients had severe or very severe scaling.
They applied Topicort 0.25% spray twice daily for 4 weeks.
The only adverse events that occurred in more than 1% of patients were dryness, irritation, and pruritus at the application site, which each occurred in 2%-3% of patients.
Treatment with Topicort – a class 1 super-potent corticosteroid indicated for up to 4 weeks of treatment of plaque psoriasis – quickly and safely relieved scaling in the majority of patients treated, Dr. Keegan concluded, noting that the findings are important given that scaling is very common among patients with plaque psoriasis, many of whom find it to be bothersome.
In fact, 94% of more than 17,000 participants in a National Psoriasis Foundation survey reported being bothered by scaling, he noted.
This study was sponsored by TaroPharma.
AT THE ODAC CONFERENCE
Key clinical point: Psoriasis patients with scaling are likely to respond to corticosteroid spray treatment.
Major finding: At 4 weeks, scaling was clear, almost clear, or mild in 84% of patients.
Data source: An analysis of pooled phase III trial data for 120 patients.
Disclosures: This study was sponsored by TaroPharma.
Apremilast looks good for psoriasis, psoriatic arthritis
New studies have led to a number of newly approved medications and therapies for patients suffering from psoriasis and psoriatic arthritis, and several more are on the horizon as clinicians and researchers continue finding new ways to effectively treat these conditions.
“This presentation will cover the new agents recently approved and nearing approval over the next 12 months,” Dr. J. Mark Jackson said in an interview. “It will highlight the general efficacy, safety, and dosing of each agent and will also highlight what is on the way.” Dr. Jackson, of the departments of medicine and dermatology at the University of Louisville (Ky.) discussed his findings at the meeting sponsored by Global Academy for Medical Education.
Among the new agents that Dr. Jackson discussed is apremilast (Otezla by Celgene), which has been approved for treatment of both psoriatic arthritis (PsA) and psoriasis (PsO), as of Sept. 23, 2014. The central mechanism of the medication is an intracellular phosphodieterase-4 (PDE-4) inhibitor, and it is administered orally with a titrated dose over the first 2 weeks of treatment from 10 mg once a day to the maintenance dosage of 30 mg twice a day.
“Apremilast is the newest agent approved for PsO and PsA and demonstrates efficacy for both,” explained Dr. Jackson, who also stated that apremilast should warrant further study to test its “utility in the treatment of atopic and endogenous dermatitis, with studies [currently] ongoing.”
Apremilast was tested against a placebo, with doses of apremilast titrated during the first week of administration, in the ESTEEM 1 trial (Study to Evaluate Safety and Effectiveness of Oral Apremilast in Patients With Moderate to Severe Plaque Psoriasis). After 16 weeks, psoriasis area and severity index (PASI) 75, PASI 50, and static Physicians Global Assessment (sPGA) levels were all significantly higher in subjects using apremilast than those randomized into the placebo cohort. “The new interleukin 17 agents have very robust PASI 75 responses and will be a great addition to the market,” said Dr. Jackson.
Similarly robust results were also seen in apremilast used to treat PsA in the PALACE 1 trial (Study of Apremilast to Treat Active Psoriatic Arthritis). In this randomized, double-blind, placebo-controlled study, ACR 20 (arthritis self management), ACR 50, and ACR 70 scores were significantly higher after 24 weeks in subjects given apremilast 30 mg twice daily and apremilast 20 mg twice daily, than in subjects who were on placebo.
Furthermore, Dr. Jackson explained that the aforementioned interleukin-17 agents are “also being investigated for PsA, and it will be interesting to see how they compare in efficacy to the currently approved agents.”
Treatment options that Dr. Jackson spoke about include certolizumab (Cimzia by UCB/Dermira) and golimumab (Simponi by Janssen), both of which are already approved for PsA, and the former of which is in a phase III trial for PsO. Dr. Jackson also discussed several treatments currently in phase III trials, such as secukinumab by Novartis, brodalumab by Amgen and AstraZeneca, and tofacitinib by Pfizer – all of which are for use against both PsO and PsA, the latter being topically applied for PsO – and tildrakizumab by Merck and Sun Pharma for PsO only.
Global Academy and this news organization are owned by the same parent company.
Dr. Jackson said he has received research, honoraria, consulting and/or other support from the following companies: Abbvie, Amgen, Celgene, Galderma, Genentech, Janssen, Lilly, Medicis, Medimetriks, Novartis, Pfizer, Promius, Topica, and TopMD.
New studies have led to a number of newly approved medications and therapies for patients suffering from psoriasis and psoriatic arthritis, and several more are on the horizon as clinicians and researchers continue finding new ways to effectively treat these conditions.
“This presentation will cover the new agents recently approved and nearing approval over the next 12 months,” Dr. J. Mark Jackson said in an interview. “It will highlight the general efficacy, safety, and dosing of each agent and will also highlight what is on the way.” Dr. Jackson, of the departments of medicine and dermatology at the University of Louisville (Ky.) discussed his findings at the meeting sponsored by Global Academy for Medical Education.
Among the new agents that Dr. Jackson discussed is apremilast (Otezla by Celgene), which has been approved for treatment of both psoriatic arthritis (PsA) and psoriasis (PsO), as of Sept. 23, 2014. The central mechanism of the medication is an intracellular phosphodieterase-4 (PDE-4) inhibitor, and it is administered orally with a titrated dose over the first 2 weeks of treatment from 10 mg once a day to the maintenance dosage of 30 mg twice a day.
“Apremilast is the newest agent approved for PsO and PsA and demonstrates efficacy for both,” explained Dr. Jackson, who also stated that apremilast should warrant further study to test its “utility in the treatment of atopic and endogenous dermatitis, with studies [currently] ongoing.”
Apremilast was tested against a placebo, with doses of apremilast titrated during the first week of administration, in the ESTEEM 1 trial (Study to Evaluate Safety and Effectiveness of Oral Apremilast in Patients With Moderate to Severe Plaque Psoriasis). After 16 weeks, psoriasis area and severity index (PASI) 75, PASI 50, and static Physicians Global Assessment (sPGA) levels were all significantly higher in subjects using apremilast than those randomized into the placebo cohort. “The new interleukin 17 agents have very robust PASI 75 responses and will be a great addition to the market,” said Dr. Jackson.
Similarly robust results were also seen in apremilast used to treat PsA in the PALACE 1 trial (Study of Apremilast to Treat Active Psoriatic Arthritis). In this randomized, double-blind, placebo-controlled study, ACR 20 (arthritis self management), ACR 50, and ACR 70 scores were significantly higher after 24 weeks in subjects given apremilast 30 mg twice daily and apremilast 20 mg twice daily, than in subjects who were on placebo.
Furthermore, Dr. Jackson explained that the aforementioned interleukin-17 agents are “also being investigated for PsA, and it will be interesting to see how they compare in efficacy to the currently approved agents.”
Treatment options that Dr. Jackson spoke about include certolizumab (Cimzia by UCB/Dermira) and golimumab (Simponi by Janssen), both of which are already approved for PsA, and the former of which is in a phase III trial for PsO. Dr. Jackson also discussed several treatments currently in phase III trials, such as secukinumab by Novartis, brodalumab by Amgen and AstraZeneca, and tofacitinib by Pfizer – all of which are for use against both PsO and PsA, the latter being topically applied for PsO – and tildrakizumab by Merck and Sun Pharma for PsO only.
Global Academy and this news organization are owned by the same parent company.
Dr. Jackson said he has received research, honoraria, consulting and/or other support from the following companies: Abbvie, Amgen, Celgene, Galderma, Genentech, Janssen, Lilly, Medicis, Medimetriks, Novartis, Pfizer, Promius, Topica, and TopMD.
New studies have led to a number of newly approved medications and therapies for patients suffering from psoriasis and psoriatic arthritis, and several more are on the horizon as clinicians and researchers continue finding new ways to effectively treat these conditions.
“This presentation will cover the new agents recently approved and nearing approval over the next 12 months,” Dr. J. Mark Jackson said in an interview. “It will highlight the general efficacy, safety, and dosing of each agent and will also highlight what is on the way.” Dr. Jackson, of the departments of medicine and dermatology at the University of Louisville (Ky.) discussed his findings at the meeting sponsored by Global Academy for Medical Education.
Among the new agents that Dr. Jackson discussed is apremilast (Otezla by Celgene), which has been approved for treatment of both psoriatic arthritis (PsA) and psoriasis (PsO), as of Sept. 23, 2014. The central mechanism of the medication is an intracellular phosphodieterase-4 (PDE-4) inhibitor, and it is administered orally with a titrated dose over the first 2 weeks of treatment from 10 mg once a day to the maintenance dosage of 30 mg twice a day.
“Apremilast is the newest agent approved for PsO and PsA and demonstrates efficacy for both,” explained Dr. Jackson, who also stated that apremilast should warrant further study to test its “utility in the treatment of atopic and endogenous dermatitis, with studies [currently] ongoing.”
Apremilast was tested against a placebo, with doses of apremilast titrated during the first week of administration, in the ESTEEM 1 trial (Study to Evaluate Safety and Effectiveness of Oral Apremilast in Patients With Moderate to Severe Plaque Psoriasis). After 16 weeks, psoriasis area and severity index (PASI) 75, PASI 50, and static Physicians Global Assessment (sPGA) levels were all significantly higher in subjects using apremilast than those randomized into the placebo cohort. “The new interleukin 17 agents have very robust PASI 75 responses and will be a great addition to the market,” said Dr. Jackson.
Similarly robust results were also seen in apremilast used to treat PsA in the PALACE 1 trial (Study of Apremilast to Treat Active Psoriatic Arthritis). In this randomized, double-blind, placebo-controlled study, ACR 20 (arthritis self management), ACR 50, and ACR 70 scores were significantly higher after 24 weeks in subjects given apremilast 30 mg twice daily and apremilast 20 mg twice daily, than in subjects who were on placebo.
Furthermore, Dr. Jackson explained that the aforementioned interleukin-17 agents are “also being investigated for PsA, and it will be interesting to see how they compare in efficacy to the currently approved agents.”
Treatment options that Dr. Jackson spoke about include certolizumab (Cimzia by UCB/Dermira) and golimumab (Simponi by Janssen), both of which are already approved for PsA, and the former of which is in a phase III trial for PsO. Dr. Jackson also discussed several treatments currently in phase III trials, such as secukinumab by Novartis, brodalumab by Amgen and AstraZeneca, and tofacitinib by Pfizer – all of which are for use against both PsO and PsA, the latter being topically applied for PsO – and tildrakizumab by Merck and Sun Pharma for PsO only.
Global Academy and this news organization are owned by the same parent company.
Dr. Jackson said he has received research, honoraria, consulting and/or other support from the following companies: Abbvie, Amgen, Celgene, Galderma, Genentech, Janssen, Lilly, Medicis, Medimetriks, Novartis, Pfizer, Promius, Topica, and TopMD.
EXPERT ANALYSIS FROM THE CARIBBEAN DERMATOLOGY SYMPOSIUM
Secukinumab earns FDA approval for plaque psoriasis
The U.S. Food and Drug Administration has approved secukinumab for the treatment of moderate to severe plaque psoriasis in adults.
The injectable interleukin-17A inhibitor is indicated for psoriasis patients who are candidates for systemic therapy and/or phototherapy, according to a press release from the FDA. Novartis Pharmaceuticals will market secukinumab as Cosentyx.
The Jan. 21 approval was based on data from randomized, placebo-controlled clinical trials involving 2,403 patients. The most common side effects reported in the trials included upper respiratory infections and diarrhea.
The biologic is being approved with a medication guide “to inform patients that, because Cosentyx is a medicine that affects the immune system, patients may have a greater risk of getting an infection,” according to the FDA release. Consequently, the FDA advised clinicians to be cautious when prescribing secukinumab to patients with chronic or recurrent infections, and to those with active Crohn’s disease.
The FDA approval followed an advisory committee meeting in October 2014.
The U.S. Food and Drug Administration has approved secukinumab for the treatment of moderate to severe plaque psoriasis in adults.
The injectable interleukin-17A inhibitor is indicated for psoriasis patients who are candidates for systemic therapy and/or phototherapy, according to a press release from the FDA. Novartis Pharmaceuticals will market secukinumab as Cosentyx.
The Jan. 21 approval was based on data from randomized, placebo-controlled clinical trials involving 2,403 patients. The most common side effects reported in the trials included upper respiratory infections and diarrhea.
The biologic is being approved with a medication guide “to inform patients that, because Cosentyx is a medicine that affects the immune system, patients may have a greater risk of getting an infection,” according to the FDA release. Consequently, the FDA advised clinicians to be cautious when prescribing secukinumab to patients with chronic or recurrent infections, and to those with active Crohn’s disease.
The FDA approval followed an advisory committee meeting in October 2014.
The U.S. Food and Drug Administration has approved secukinumab for the treatment of moderate to severe plaque psoriasis in adults.
The injectable interleukin-17A inhibitor is indicated for psoriasis patients who are candidates for systemic therapy and/or phototherapy, according to a press release from the FDA. Novartis Pharmaceuticals will market secukinumab as Cosentyx.
The Jan. 21 approval was based on data from randomized, placebo-controlled clinical trials involving 2,403 patients. The most common side effects reported in the trials included upper respiratory infections and diarrhea.
The biologic is being approved with a medication guide “to inform patients that, because Cosentyx is a medicine that affects the immune system, patients may have a greater risk of getting an infection,” according to the FDA release. Consequently, the FDA advised clinicians to be cautious when prescribing secukinumab to patients with chronic or recurrent infections, and to those with active Crohn’s disease.
The FDA approval followed an advisory committee meeting in October 2014.