User login
Psoriasis treatment recommendations address four clinical scenarios
New guidelines on nail psoriasis address four clinical manifestations of the disease. The recommendations by the Medical Board of the National Psoriasis Foundation appeared as a consensus statement in the January issue of JAMA Dermatology.
Limitations in clinical trial data make comparing treatments difficult, noted lead author Dr. Jeffrey J. Crowley of Bakersfield (Calif.) Dermatology and his associates. “There are limited data to evaluate or support the use of combination therapy in nail psoriasis. Thus, treatment options recommended in this review are monotherapy,” the guidelines authors added (JAMA Dermatol. 2015;151:87-94).
To develop the guidelines, the research team searched PubMed for articles on nail psoriasis dating from Jan. 1, 1947 through May 11, 2014. They evaluated these studies for level of evidence based on recommendations for writing guidelines from Dr. Paul G. Shekelle of the VA West Los Angeles Medical Center and his associates (BMJ 1999;318:593-6).
They also polled the Medical Board of the National Psoriasis Foundation regarding their treatment approach for four clinical presentations of nail psoriasis:
• For treatment-naive patients with psoriasis of the nails only (affecting at least 3 of 10 fingernails), the board recommended initial treatment with high-potency topical corticosteroids (with or without calcipotriol), with intralesional corticosteroids as a secondary option. Intralesional corticosteroids have been used for decades, but clinical data supporting their use are “extremely limited,” the guidelines state.
• For extensive nail psoriasis (affecting at least five fingernails and causing moderate to severe pain) that has failed topical treatment, the board recommended adalimumab most enthusiastically, followed by etanercept, intralesional corticosteroids, ustekinumab, methotrexate sodium, and acitretin in decreasing order.
• For concurrent skin and nail disease without joint involvement (defined as skin disease on at least 8% of the body surface and moderately to severely painful dystrophy of at least 5 of 10 nails), the board strongly recommended adalimumab, etanercept, and ustekinumab, and also recommended methotrexate, acitretin, infliximab, and apremilast.
• For concurrent nail, skin, and joint involvement (defined as skin disease on 8% of the body surface, a history of dactylitis and morning stiffness (psoriatic arthritis), and severe, painful involvement of at least 5 of 10 nails), the board most strongly recommended adalimumab, followed by etanercept, ustekinumab, infliximab, methotrexate, apremilast, and golimumab.
Because nails grow slowly, psoriatic joint and skin disease often improve before nail psoriasis does, the authors noted. “Few studies show any significant improvement before 12 weeks, and several studies with etanercept, infliximab, and ustekinumab demonstrate continued improvement beyond 6 months,” they wrote.
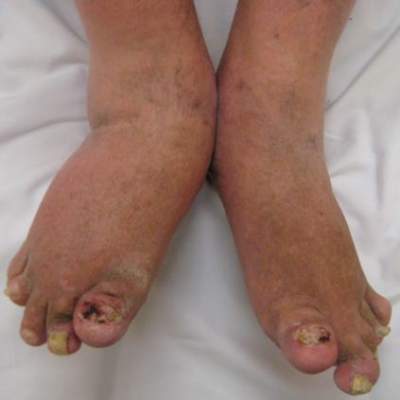
About half of patients with psoriasis have some amount of nail involvement, and about 70% of patients with psoriatic arthritis have nail disease, according to the literature review. Dermatophyte infections can further complicate treatment of nail psoriasis, and immunosuppressive therapies can lead to onychomycosis in patients whose psoriasis includes the toenails, the authors added.
Dr. Crowley reported speaker and consulting honoraria from AbbVie, Abbott, and Amgen, and research funding from Abbott, AbbVie, Amgen, AstraZeneca, Celgene, Eli Lilly, Janssen Pharmaceutica, Merck, Pfizer, and Regeneron Pharmaceuticals. Four coauthors reported advisory, consulting, or financial relationships with Amgen, Abbott, Janssen Biotech Inc., Celgene, Novartis International AG, Abbvie, Merck, Celgene, Leo Pharma, Eli Lilly, Pfizer, and the National Psoriasis Foundation.
New guidelines on nail psoriasis address four clinical manifestations of the disease. The recommendations by the Medical Board of the National Psoriasis Foundation appeared as a consensus statement in the January issue of JAMA Dermatology.
Limitations in clinical trial data make comparing treatments difficult, noted lead author Dr. Jeffrey J. Crowley of Bakersfield (Calif.) Dermatology and his associates. “There are limited data to evaluate or support the use of combination therapy in nail psoriasis. Thus, treatment options recommended in this review are monotherapy,” the guidelines authors added (JAMA Dermatol. 2015;151:87-94).
To develop the guidelines, the research team searched PubMed for articles on nail psoriasis dating from Jan. 1, 1947 through May 11, 2014. They evaluated these studies for level of evidence based on recommendations for writing guidelines from Dr. Paul G. Shekelle of the VA West Los Angeles Medical Center and his associates (BMJ 1999;318:593-6).
They also polled the Medical Board of the National Psoriasis Foundation regarding their treatment approach for four clinical presentations of nail psoriasis:
• For treatment-naive patients with psoriasis of the nails only (affecting at least 3 of 10 fingernails), the board recommended initial treatment with high-potency topical corticosteroids (with or without calcipotriol), with intralesional corticosteroids as a secondary option. Intralesional corticosteroids have been used for decades, but clinical data supporting their use are “extremely limited,” the guidelines state.
• For extensive nail psoriasis (affecting at least five fingernails and causing moderate to severe pain) that has failed topical treatment, the board recommended adalimumab most enthusiastically, followed by etanercept, intralesional corticosteroids, ustekinumab, methotrexate sodium, and acitretin in decreasing order.
• For concurrent skin and nail disease without joint involvement (defined as skin disease on at least 8% of the body surface and moderately to severely painful dystrophy of at least 5 of 10 nails), the board strongly recommended adalimumab, etanercept, and ustekinumab, and also recommended methotrexate, acitretin, infliximab, and apremilast.
• For concurrent nail, skin, and joint involvement (defined as skin disease on 8% of the body surface, a history of dactylitis and morning stiffness (psoriatic arthritis), and severe, painful involvement of at least 5 of 10 nails), the board most strongly recommended adalimumab, followed by etanercept, ustekinumab, infliximab, methotrexate, apremilast, and golimumab.
Because nails grow slowly, psoriatic joint and skin disease often improve before nail psoriasis does, the authors noted. “Few studies show any significant improvement before 12 weeks, and several studies with etanercept, infliximab, and ustekinumab demonstrate continued improvement beyond 6 months,” they wrote.
About half of patients with psoriasis have some amount of nail involvement, and about 70% of patients with psoriatic arthritis have nail disease, according to the literature review. Dermatophyte infections can further complicate treatment of nail psoriasis, and immunosuppressive therapies can lead to onychomycosis in patients whose psoriasis includes the toenails, the authors added.
Dr. Crowley reported speaker and consulting honoraria from AbbVie, Abbott, and Amgen, and research funding from Abbott, AbbVie, Amgen, AstraZeneca, Celgene, Eli Lilly, Janssen Pharmaceutica, Merck, Pfizer, and Regeneron Pharmaceuticals. Four coauthors reported advisory, consulting, or financial relationships with Amgen, Abbott, Janssen Biotech Inc., Celgene, Novartis International AG, Abbvie, Merck, Celgene, Leo Pharma, Eli Lilly, Pfizer, and the National Psoriasis Foundation.
New guidelines on nail psoriasis address four clinical manifestations of the disease. The recommendations by the Medical Board of the National Psoriasis Foundation appeared as a consensus statement in the January issue of JAMA Dermatology.
Limitations in clinical trial data make comparing treatments difficult, noted lead author Dr. Jeffrey J. Crowley of Bakersfield (Calif.) Dermatology and his associates. “There are limited data to evaluate or support the use of combination therapy in nail psoriasis. Thus, treatment options recommended in this review are monotherapy,” the guidelines authors added (JAMA Dermatol. 2015;151:87-94).
To develop the guidelines, the research team searched PubMed for articles on nail psoriasis dating from Jan. 1, 1947 through May 11, 2014. They evaluated these studies for level of evidence based on recommendations for writing guidelines from Dr. Paul G. Shekelle of the VA West Los Angeles Medical Center and his associates (BMJ 1999;318:593-6).
They also polled the Medical Board of the National Psoriasis Foundation regarding their treatment approach for four clinical presentations of nail psoriasis:
• For treatment-naive patients with psoriasis of the nails only (affecting at least 3 of 10 fingernails), the board recommended initial treatment with high-potency topical corticosteroids (with or without calcipotriol), with intralesional corticosteroids as a secondary option. Intralesional corticosteroids have been used for decades, but clinical data supporting their use are “extremely limited,” the guidelines state.
• For extensive nail psoriasis (affecting at least five fingernails and causing moderate to severe pain) that has failed topical treatment, the board recommended adalimumab most enthusiastically, followed by etanercept, intralesional corticosteroids, ustekinumab, methotrexate sodium, and acitretin in decreasing order.
• For concurrent skin and nail disease without joint involvement (defined as skin disease on at least 8% of the body surface and moderately to severely painful dystrophy of at least 5 of 10 nails), the board strongly recommended adalimumab, etanercept, and ustekinumab, and also recommended methotrexate, acitretin, infliximab, and apremilast.
• For concurrent nail, skin, and joint involvement (defined as skin disease on 8% of the body surface, a history of dactylitis and morning stiffness (psoriatic arthritis), and severe, painful involvement of at least 5 of 10 nails), the board most strongly recommended adalimumab, followed by etanercept, ustekinumab, infliximab, methotrexate, apremilast, and golimumab.
Because nails grow slowly, psoriatic joint and skin disease often improve before nail psoriasis does, the authors noted. “Few studies show any significant improvement before 12 weeks, and several studies with etanercept, infliximab, and ustekinumab demonstrate continued improvement beyond 6 months,” they wrote.
About half of patients with psoriasis have some amount of nail involvement, and about 70% of patients with psoriatic arthritis have nail disease, according to the literature review. Dermatophyte infections can further complicate treatment of nail psoriasis, and immunosuppressive therapies can lead to onychomycosis in patients whose psoriasis includes the toenails, the authors added.
Dr. Crowley reported speaker and consulting honoraria from AbbVie, Abbott, and Amgen, and research funding from Abbott, AbbVie, Amgen, AstraZeneca, Celgene, Eli Lilly, Janssen Pharmaceutica, Merck, Pfizer, and Regeneron Pharmaceuticals. Four coauthors reported advisory, consulting, or financial relationships with Amgen, Abbott, Janssen Biotech Inc., Celgene, Novartis International AG, Abbvie, Merck, Celgene, Leo Pharma, Eli Lilly, Pfizer, and the National Psoriasis Foundation.
FROM JAMA DERMATOLOGY
Early psoriatic arthritis treatment with etanercept gives better outcomes
Patients with psoriatic arthritis and psoriasis report having a better response to etanercept the earlier they are treated, according to a post hoc analysis of the PRESTA trial.
Patients with shorter psoriatic arthritis (PsA) duration had greater improvements in arthritis scores and several patient-reported outcomes at 24 weeks of treatment with etanercept 50 mg a week, compared with patients with longer disease duration.
The researchers, led by Dr. Bruce Kirkham from Guy’s and St. Thomas’ NHS Foundation Trust, London, said the results showed “clinicians should consider treating their PsA patients with therapies effective in PsA early rather than late.”
The industry-sponsored PRESTA (Psoriasis Randomized Etanercept Study in Patients with Psoriatic Arthritis) trial was a randomized, blinded, 24-week, multicenter study enrolling adults with active but stable plaque psoriasis involving at least 10% body surface area and active PsA defined as 2 or more swollen joints, 2 or more tender joints, joint pain for 3 months or longer, and a negative serum rheumatoid factor within 6 months prior to baseline.
Overall, 372 patients who received etanercept 50 mg once a week for 24 weeks were included in the current post hoc analysis (Clin. Exp. Rheumatol. 2014 Dec. 22).
Baseline and after treatment changes were compared between patients with PsA disease duration of 2 years or less (n = 103) and those with disease more than 2 years (n = 269).
Baseline efficacy measures were similar between the shorter duration and longer duration groups, with the exception of Physicians Global Assessment (PGA) arthritis score, which was significantly lower in the group with 2 years or less duration (44.9 vs. 51.8; P = .006), the authors reported.
At week 24, joint disease improved, based on the PGA arthritis score, by a significantly greater amount in the shorter duration group (–39.8 vs. –35.7; P = .03).
Clinically meaningful improvements in patient-reported outcomes with etanercept treatment occurred in both groups, the study authors said, but changes in scores from baseline to week 24 were significantly higher in the shorter duration group for visual analog scale reports of joint pain (P = .007) and arthritis activity (P = .01) as well as quality of life on EuroQol 5D utility (P = .046) and EuroQol 5D visual analog scale (P = .04) responses.
The mean number of swollen joints that had improved from baseline to week 24 was not significantly different between the groups, and no significant between-group differences were seen in the percentages of patients achieving the ACR20, ACR50, and ACR70 responses.
While all patients responded to treatment irrespective of disease duration, patients with shorter disease duration had greater improvements on some measures, the authors concluded.
However, the study was limited by the fact that it was a post hoc analysis and the original trial was not designed to explore the effect of early treatment versus later treatment in patients with PsA and moderate-to-severe psoriasis, they noted.
The study was sponsored by Wyeth, which was acquired by Pfizer, the manufacturer of etanercept, in October 2009. Several of the authors declared receiving honoraria from several pharmaceutical companies. Two authors were employees of Pfizer during the PRESTA study and development of the current manuscript, and two other authors are current employees of Pfizer.
Patients with psoriatic arthritis and psoriasis report having a better response to etanercept the earlier they are treated, according to a post hoc analysis of the PRESTA trial.
Patients with shorter psoriatic arthritis (PsA) duration had greater improvements in arthritis scores and several patient-reported outcomes at 24 weeks of treatment with etanercept 50 mg a week, compared with patients with longer disease duration.
The researchers, led by Dr. Bruce Kirkham from Guy’s and St. Thomas’ NHS Foundation Trust, London, said the results showed “clinicians should consider treating their PsA patients with therapies effective in PsA early rather than late.”
The industry-sponsored PRESTA (Psoriasis Randomized Etanercept Study in Patients with Psoriatic Arthritis) trial was a randomized, blinded, 24-week, multicenter study enrolling adults with active but stable plaque psoriasis involving at least 10% body surface area and active PsA defined as 2 or more swollen joints, 2 or more tender joints, joint pain for 3 months or longer, and a negative serum rheumatoid factor within 6 months prior to baseline.
Overall, 372 patients who received etanercept 50 mg once a week for 24 weeks were included in the current post hoc analysis (Clin. Exp. Rheumatol. 2014 Dec. 22).
Baseline and after treatment changes were compared between patients with PsA disease duration of 2 years or less (n = 103) and those with disease more than 2 years (n = 269).
Baseline efficacy measures were similar between the shorter duration and longer duration groups, with the exception of Physicians Global Assessment (PGA) arthritis score, which was significantly lower in the group with 2 years or less duration (44.9 vs. 51.8; P = .006), the authors reported.
At week 24, joint disease improved, based on the PGA arthritis score, by a significantly greater amount in the shorter duration group (–39.8 vs. –35.7; P = .03).
Clinically meaningful improvements in patient-reported outcomes with etanercept treatment occurred in both groups, the study authors said, but changes in scores from baseline to week 24 were significantly higher in the shorter duration group for visual analog scale reports of joint pain (P = .007) and arthritis activity (P = .01) as well as quality of life on EuroQol 5D utility (P = .046) and EuroQol 5D visual analog scale (P = .04) responses.
The mean number of swollen joints that had improved from baseline to week 24 was not significantly different between the groups, and no significant between-group differences were seen in the percentages of patients achieving the ACR20, ACR50, and ACR70 responses.
While all patients responded to treatment irrespective of disease duration, patients with shorter disease duration had greater improvements on some measures, the authors concluded.
However, the study was limited by the fact that it was a post hoc analysis and the original trial was not designed to explore the effect of early treatment versus later treatment in patients with PsA and moderate-to-severe psoriasis, they noted.
The study was sponsored by Wyeth, which was acquired by Pfizer, the manufacturer of etanercept, in October 2009. Several of the authors declared receiving honoraria from several pharmaceutical companies. Two authors were employees of Pfizer during the PRESTA study and development of the current manuscript, and two other authors are current employees of Pfizer.
Patients with psoriatic arthritis and psoriasis report having a better response to etanercept the earlier they are treated, according to a post hoc analysis of the PRESTA trial.
Patients with shorter psoriatic arthritis (PsA) duration had greater improvements in arthritis scores and several patient-reported outcomes at 24 weeks of treatment with etanercept 50 mg a week, compared with patients with longer disease duration.
The researchers, led by Dr. Bruce Kirkham from Guy’s and St. Thomas’ NHS Foundation Trust, London, said the results showed “clinicians should consider treating their PsA patients with therapies effective in PsA early rather than late.”
The industry-sponsored PRESTA (Psoriasis Randomized Etanercept Study in Patients with Psoriatic Arthritis) trial was a randomized, blinded, 24-week, multicenter study enrolling adults with active but stable plaque psoriasis involving at least 10% body surface area and active PsA defined as 2 or more swollen joints, 2 or more tender joints, joint pain for 3 months or longer, and a negative serum rheumatoid factor within 6 months prior to baseline.
Overall, 372 patients who received etanercept 50 mg once a week for 24 weeks were included in the current post hoc analysis (Clin. Exp. Rheumatol. 2014 Dec. 22).
Baseline and after treatment changes were compared between patients with PsA disease duration of 2 years or less (n = 103) and those with disease more than 2 years (n = 269).
Baseline efficacy measures were similar between the shorter duration and longer duration groups, with the exception of Physicians Global Assessment (PGA) arthritis score, which was significantly lower in the group with 2 years or less duration (44.9 vs. 51.8; P = .006), the authors reported.
At week 24, joint disease improved, based on the PGA arthritis score, by a significantly greater amount in the shorter duration group (–39.8 vs. –35.7; P = .03).
Clinically meaningful improvements in patient-reported outcomes with etanercept treatment occurred in both groups, the study authors said, but changes in scores from baseline to week 24 were significantly higher in the shorter duration group for visual analog scale reports of joint pain (P = .007) and arthritis activity (P = .01) as well as quality of life on EuroQol 5D utility (P = .046) and EuroQol 5D visual analog scale (P = .04) responses.
The mean number of swollen joints that had improved from baseline to week 24 was not significantly different between the groups, and no significant between-group differences were seen in the percentages of patients achieving the ACR20, ACR50, and ACR70 responses.
While all patients responded to treatment irrespective of disease duration, patients with shorter disease duration had greater improvements on some measures, the authors concluded.
However, the study was limited by the fact that it was a post hoc analysis and the original trial was not designed to explore the effect of early treatment versus later treatment in patients with PsA and moderate-to-severe psoriasis, they noted.
The study was sponsored by Wyeth, which was acquired by Pfizer, the manufacturer of etanercept, in October 2009. Several of the authors declared receiving honoraria from several pharmaceutical companies. Two authors were employees of Pfizer during the PRESTA study and development of the current manuscript, and two other authors are current employees of Pfizer.
FROM CLINICAL AND EXPERIMENTAL RHEUMATOLOGY
Key clinical point: PsA treatment with etanercept within 2 years of diagnosis may lead to better patient-reported quality of life outcomes than does treatment starting more than 2 years after diagnosis.
Major finding: After 24 weeks of etanercept 50 mg per week, joint disease improved, based on the PGA arthritis score, by a significantly greater amount in patients with PsA for 2 years or less vs. those with the disease for more than 2 years (–39.8 vs. –35.7, respectively; P = .03) .
Data source: A post hoc analysis of 372 patients with PsA and psoriasis who were enrolled in the PRESTA trial.
Disclosures: The study was sponsored by Wyeth, which was acquired by Pfizer, the manufacturer of etanercept, in October 2009. Several of the authors declared receiving honoraria from several pharmaceutical companies. Two authors were employees of Pfizer during the PRESTA study and development of the current manuscript, and two other authors are current employees of Pfizer.
Case series: Ustekinumab for psoriasis helps skin, hurts joints
Ustekinumab treatment was associated with new-onset or worsening psoriatic arthritis in a series of seven patients with psoriasis.
The findings, which support previous observations that patients treated with ustekinumab (Stelara) “sometimes have discordant responses of their skin and joint disease,” underscore the need for regularly asking patients about joint symptoms, and for referral to a rheumatologist for suspected psoriatic arthritis, Ben B. Jones of the University of Utah, Salt Lake City, and his colleagues reported (Br. J. Dermatol. 2014 Dec. 30 [doi:10.1111/bjd.13645]).
All seven patients in the case series had well-controlled psoriasis on ustekinumab. Five had new-onset psoriatic arthritis, and two had worsening psoriatic arthritis on treatment. The patients had phenotypic similarities; most were women over age 49 years, and all five of those with new-onset disease were women. Also, five of the seven patients had exposure to tumor necrosis factor inhibitors prior to switching to ustekinumab.
Three other case series have reported similar findings of marked improvement in cutaneous symptoms with worsening of joint symptoms among patients treated with ustekinumab, the investigators noted, concluding that the findings – which may reflect a lack of efficacy at the administered doses or a need for more frequent dosing – may support arguments that psoriatic arthritis and psoriasis involve distinct inflammatory pathways.
“It is also possible that ustekinumab may trigger or unmask inflammation in the joints of patients with psoriatic arthritis,” they wrote, concluding that “larger epidemiologic studies comparing patients with discordant and concordant cutaneous and articular responses to ustekinumab may better define patients at risk for psoriatic arthritis worsening with ustekinumab.”
Two of the five authors have reported serving as a consultant or advisory board member, receiving payment for lectures, and/or serving as an investigator for Janssen, which markets ustekinumab, as well as other manufacturers of biologics. The other authors reported having no conflicts of interest.
Ustekinumab treatment was associated with new-onset or worsening psoriatic arthritis in a series of seven patients with psoriasis.
The findings, which support previous observations that patients treated with ustekinumab (Stelara) “sometimes have discordant responses of their skin and joint disease,” underscore the need for regularly asking patients about joint symptoms, and for referral to a rheumatologist for suspected psoriatic arthritis, Ben B. Jones of the University of Utah, Salt Lake City, and his colleagues reported (Br. J. Dermatol. 2014 Dec. 30 [doi:10.1111/bjd.13645]).
All seven patients in the case series had well-controlled psoriasis on ustekinumab. Five had new-onset psoriatic arthritis, and two had worsening psoriatic arthritis on treatment. The patients had phenotypic similarities; most were women over age 49 years, and all five of those with new-onset disease were women. Also, five of the seven patients had exposure to tumor necrosis factor inhibitors prior to switching to ustekinumab.
Three other case series have reported similar findings of marked improvement in cutaneous symptoms with worsening of joint symptoms among patients treated with ustekinumab, the investigators noted, concluding that the findings – which may reflect a lack of efficacy at the administered doses or a need for more frequent dosing – may support arguments that psoriatic arthritis and psoriasis involve distinct inflammatory pathways.
“It is also possible that ustekinumab may trigger or unmask inflammation in the joints of patients with psoriatic arthritis,” they wrote, concluding that “larger epidemiologic studies comparing patients with discordant and concordant cutaneous and articular responses to ustekinumab may better define patients at risk for psoriatic arthritis worsening with ustekinumab.”
Two of the five authors have reported serving as a consultant or advisory board member, receiving payment for lectures, and/or serving as an investigator for Janssen, which markets ustekinumab, as well as other manufacturers of biologics. The other authors reported having no conflicts of interest.
Ustekinumab treatment was associated with new-onset or worsening psoriatic arthritis in a series of seven patients with psoriasis.
The findings, which support previous observations that patients treated with ustekinumab (Stelara) “sometimes have discordant responses of their skin and joint disease,” underscore the need for regularly asking patients about joint symptoms, and for referral to a rheumatologist for suspected psoriatic arthritis, Ben B. Jones of the University of Utah, Salt Lake City, and his colleagues reported (Br. J. Dermatol. 2014 Dec. 30 [doi:10.1111/bjd.13645]).
All seven patients in the case series had well-controlled psoriasis on ustekinumab. Five had new-onset psoriatic arthritis, and two had worsening psoriatic arthritis on treatment. The patients had phenotypic similarities; most were women over age 49 years, and all five of those with new-onset disease were women. Also, five of the seven patients had exposure to tumor necrosis factor inhibitors prior to switching to ustekinumab.
Three other case series have reported similar findings of marked improvement in cutaneous symptoms with worsening of joint symptoms among patients treated with ustekinumab, the investigators noted, concluding that the findings – which may reflect a lack of efficacy at the administered doses or a need for more frequent dosing – may support arguments that psoriatic arthritis and psoriasis involve distinct inflammatory pathways.
“It is also possible that ustekinumab may trigger or unmask inflammation in the joints of patients with psoriatic arthritis,” they wrote, concluding that “larger epidemiologic studies comparing patients with discordant and concordant cutaneous and articular responses to ustekinumab may better define patients at risk for psoriatic arthritis worsening with ustekinumab.”
Two of the five authors have reported serving as a consultant or advisory board member, receiving payment for lectures, and/or serving as an investigator for Janssen, which markets ustekinumab, as well as other manufacturers of biologics. The other authors reported having no conflicts of interest.
Key clinical point: Increasing evidence suggests that ustekinumab is associated with discordant joint and skin responses.
Major finding: Five of seven psoriasis patients treated with ustekinumab experienced new-onset psoriatic arthritis, and two had worsening psoriatic arthritis.
Data source: A series of seven cases.
Disclosures: Two of the five authors have reported serving as a consultant or advisory board member, receiving payment for lectures, and/or serving as an investigator for Janssen, which markets ustekinumab, as well as other manufacturers of biologics. The other authors reported having no conflicts of interest.
Framingham score underestimates CVD risk in psoriatic arthritis patients
Most newly diagnosed psoriatic arthritis patients have an increased risk for cardiovascular disease that is markedly underestimated by the Framingham Risk Score, according to findings from a retrospective, population-based, cohort study.
The mean Framingham Risk Score in 126 patients with psoriatic arthritis who were aged 30 years or older and who had no prior cardiovascular disease (CVD) history, was 9.7% during the first 10 years of follow-up. However, the 10-year cumulative incidence of CVD events was nearly double that at 17% (standardized incidence ratio, 1.80), Dr. Floranne C. Ernste of the Mayo Clinic, Rochester, Minn., and her colleagues reported (Arthritis Care Res. 2015 Jan. 7 [doi:10.1002/acr.22536]).
Age-based analysis showed that the CVD risk in these patients was consistently twice as high as predicted by the FRS beginning after age 40 years, the authors noted.
The findings underscore the importance of CVD risk assessment in patients with psoriatic arthritis but suggest that the Framingham Risk Score may not be applicable in such patients, the investigators said, adding that “This study serves to illustrate the important need for further research to focus on the development of CVD risk assessment tools specific to psoriatic arthritis patients.”
The findings also suggest that aggressive therapy may be warranted early in the course of psoriatic arthritis to “attenuate the long-term burden of CVD,” they said.
This study was supported by the Rochester Epidemiology Project and by Amgen. Dr. Ernste reported having no disclosures.
Most newly diagnosed psoriatic arthritis patients have an increased risk for cardiovascular disease that is markedly underestimated by the Framingham Risk Score, according to findings from a retrospective, population-based, cohort study.
The mean Framingham Risk Score in 126 patients with psoriatic arthritis who were aged 30 years or older and who had no prior cardiovascular disease (CVD) history, was 9.7% during the first 10 years of follow-up. However, the 10-year cumulative incidence of CVD events was nearly double that at 17% (standardized incidence ratio, 1.80), Dr. Floranne C. Ernste of the Mayo Clinic, Rochester, Minn., and her colleagues reported (Arthritis Care Res. 2015 Jan. 7 [doi:10.1002/acr.22536]).
Age-based analysis showed that the CVD risk in these patients was consistently twice as high as predicted by the FRS beginning after age 40 years, the authors noted.
The findings underscore the importance of CVD risk assessment in patients with psoriatic arthritis but suggest that the Framingham Risk Score may not be applicable in such patients, the investigators said, adding that “This study serves to illustrate the important need for further research to focus on the development of CVD risk assessment tools specific to psoriatic arthritis patients.”
The findings also suggest that aggressive therapy may be warranted early in the course of psoriatic arthritis to “attenuate the long-term burden of CVD,” they said.
This study was supported by the Rochester Epidemiology Project and by Amgen. Dr. Ernste reported having no disclosures.
Most newly diagnosed psoriatic arthritis patients have an increased risk for cardiovascular disease that is markedly underestimated by the Framingham Risk Score, according to findings from a retrospective, population-based, cohort study.
The mean Framingham Risk Score in 126 patients with psoriatic arthritis who were aged 30 years or older and who had no prior cardiovascular disease (CVD) history, was 9.7% during the first 10 years of follow-up. However, the 10-year cumulative incidence of CVD events was nearly double that at 17% (standardized incidence ratio, 1.80), Dr. Floranne C. Ernste of the Mayo Clinic, Rochester, Minn., and her colleagues reported (Arthritis Care Res. 2015 Jan. 7 [doi:10.1002/acr.22536]).
Age-based analysis showed that the CVD risk in these patients was consistently twice as high as predicted by the FRS beginning after age 40 years, the authors noted.
The findings underscore the importance of CVD risk assessment in patients with psoriatic arthritis but suggest that the Framingham Risk Score may not be applicable in such patients, the investigators said, adding that “This study serves to illustrate the important need for further research to focus on the development of CVD risk assessment tools specific to psoriatic arthritis patients.”
The findings also suggest that aggressive therapy may be warranted early in the course of psoriatic arthritis to “attenuate the long-term burden of CVD,” they said.
This study was supported by the Rochester Epidemiology Project and by Amgen. Dr. Ernste reported having no disclosures.
FROM ARTHRITIS CARE & RESEARCH
Key clinical point: The Framingham Risk Score may not be applicable for estimating CVD risk in psoriatic arthritis patients.
Major finding: The 10-year cumulative CVD incidence rate was 17%, compared with 9.7% predicted by Framingham Risk Score.
Data source: A retrospective, population-based, cohort study of 126 patients.
Disclosures: This study was supported by the Rochester Epidemiology Project and by Amgen. Dr. Ernste reported having no disclosures.
Annual costs of psoriasis costs top $112 billion
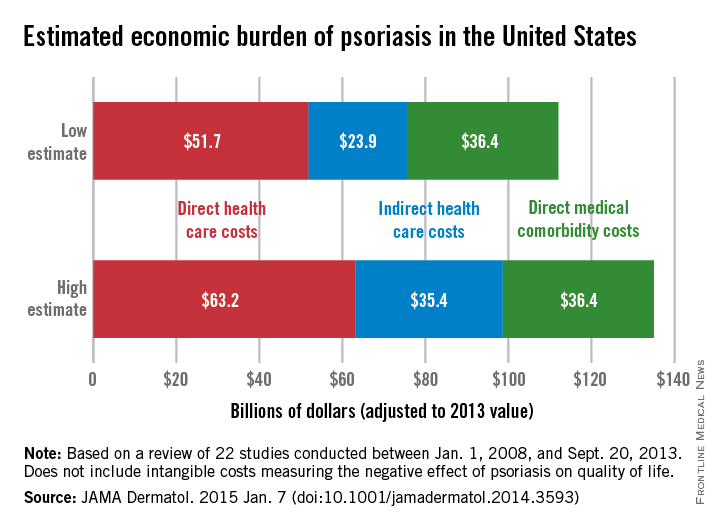
The total economic burden of psoriasis in the United States is at least $112 billion per year, and possibly as high as $135 billion, investigators estimated in a study published Jan. 7 in JAMA Dermatology.
Dr. Elizabeth A. Brezinski of the University of California, Davis, in Sacramento, and her associates, reviewed 22 studies conducted between Jan. 1, 2008, and Sept. 20, 2013, adjusting the results to 2013 dollars.
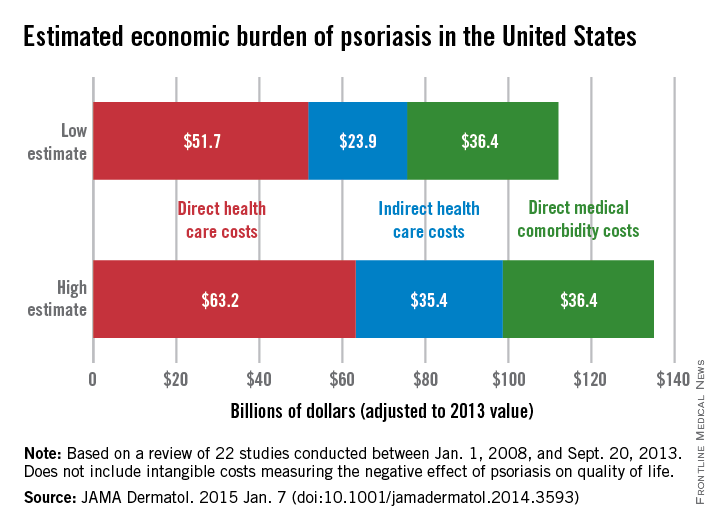
Estimates for the direct medical cost of psoriasis care ranged from $51.7 billion to $63.2 billion per year. Indirect costs from absenteeism or working while sick contributed another $23.9-$35.4 billion, with comorbidity costs estimated at $36.4 billion annually (JAMA Dermatol. 2014 Jan. 7 [doi:10.1001/jamadermatol.2014.3593]).
Intangible costs on quality of life, which were not included in the annual figures, were estimated to be $85.1 billion over the lifetimes of the psoriasis patient population (7.4 million as of 2013), they said.
“Defining the economic burden of psoriasis from a societal perspective is the foundation for innovating and providing access to cost-effective therapies that will result in improved patient outcomes,” Dr. Brezinski and her coauthors wrote.
One of the researchers reported serving as an investigator for, or consultant to, AbbVie, Amgen, Celgene, Janssen, Lilly, Merck, Pfizer, and UCB. No other disclosures were reported.
The total economic burden of psoriasis in the United States is at least $112 billion per year, and possibly as high as $135 billion, investigators estimated in a study published Jan. 7 in JAMA Dermatology.
Dr. Elizabeth A. Brezinski of the University of California, Davis, in Sacramento, and her associates, reviewed 22 studies conducted between Jan. 1, 2008, and Sept. 20, 2013, adjusting the results to 2013 dollars.

Estimates for the direct medical cost of psoriasis care ranged from $51.7 billion to $63.2 billion per year. Indirect costs from absenteeism or working while sick contributed another $23.9-$35.4 billion, with comorbidity costs estimated at $36.4 billion annually (JAMA Dermatol. 2014 Jan. 7 [doi:10.1001/jamadermatol.2014.3593]).
Intangible costs on quality of life, which were not included in the annual figures, were estimated to be $85.1 billion over the lifetimes of the psoriasis patient population (7.4 million as of 2013), they said.
“Defining the economic burden of psoriasis from a societal perspective is the foundation for innovating and providing access to cost-effective therapies that will result in improved patient outcomes,” Dr. Brezinski and her coauthors wrote.
One of the researchers reported serving as an investigator for, or consultant to, AbbVie, Amgen, Celgene, Janssen, Lilly, Merck, Pfizer, and UCB. No other disclosures were reported.
The total economic burden of psoriasis in the United States is at least $112 billion per year, and possibly as high as $135 billion, investigators estimated in a study published Jan. 7 in JAMA Dermatology.
Dr. Elizabeth A. Brezinski of the University of California, Davis, in Sacramento, and her associates, reviewed 22 studies conducted between Jan. 1, 2008, and Sept. 20, 2013, adjusting the results to 2013 dollars.

Estimates for the direct medical cost of psoriasis care ranged from $51.7 billion to $63.2 billion per year. Indirect costs from absenteeism or working while sick contributed another $23.9-$35.4 billion, with comorbidity costs estimated at $36.4 billion annually (JAMA Dermatol. 2014 Jan. 7 [doi:10.1001/jamadermatol.2014.3593]).
Intangible costs on quality of life, which were not included in the annual figures, were estimated to be $85.1 billion over the lifetimes of the psoriasis patient population (7.4 million as of 2013), they said.
“Defining the economic burden of psoriasis from a societal perspective is the foundation for innovating and providing access to cost-effective therapies that will result in improved patient outcomes,” Dr. Brezinski and her coauthors wrote.
One of the researchers reported serving as an investigator for, or consultant to, AbbVie, Amgen, Celgene, Janssen, Lilly, Merck, Pfizer, and UCB. No other disclosures were reported.
FROM JAMA DERMATOLOGY
Immunogenicity to TNF-alpha blockers varies in psoriatic arthritis
Immunogenicity to three TNF-alpha blocking agents seems to vary substantially among patients with psoriatic arthritis, and the use of methotrexate appears to attenuate the presence of antidrug antibodies, according to findings from a cross-sectional study.
Although researchers have looked at the immunogenicity of TNF-alpha blockers in patients with diseases such as rheumatoid arthritis (RA), ankylosing spondylitis, psoriasis, and inflammatory bowel disease, their immunogenicity in psoriatic arthritis (PsA) patients has not been fully investigated, noted the current study’s investigators, led by Dr. Michael Zisapel of the department of rheumatology at Tel Aviv University (J. Rheumatol. 2014 Nov. 15 [doi:10.3899/jrheum.140685]).
The prevalence of antidrug antibodies (ADAb) to TNF-alpha blockers has been reported to be 20%-40% in RA, 25%-64% in ankylosing spondylitis, and about 33% in psoriasis, and evidence has shown that this is significantly reduced by the use of methotrexate, they said.
The study involved 93 patients with PsA who were taking adalimumab (n = 48), infliximab (n = 24), and etanercept (n = 21). A quarter of the patients were taking methotrexate at an average dose of 13.3 mg/week.
Overall, 77% of the patients had therapeutic drug levels. The prevalence of immunogenicity in the entire group was 33.3%, and one-fifth of patients had high concentrations of antibodies.
High levels of ADAb were found in 29% of patients taking adalimumab and 21% of the patients taking infliximab. No ADAb levels were found in patients taking etanercept.
Interestingly, the patients taking adalimumab demonstrated an immunogenicity of 54%, but only half of them (29%) showed high concentrations of antibodies.
“This finding suggests that a variety of levels might be found that might affect the significance of immunogenicity differently among patients producing ADAb,” the investigators wrote.
Fewer methotrexate-treated patients had high ADAb concentrations, compared with patients not taking methotrexate (16.7% vs. 21.7%).
A clear correlation was found between the presence of immunogenicity, lower drug levels, and decreased clinical response in PsA patients, just as other studies have found in RA and ankylosing spondylitis patients, the authors noted.
“Our results suggest that the use of methotrexate should be strongly considered in addition to monoclonal antibodies,” they said.
Limitations of their study included the small number of patients and the use of a bridging ELISA test which is reliable but considered to be less accurate than radioimmunoassay.
No disclosure information was available.
Immunogenicity to three TNF-alpha blocking agents seems to vary substantially among patients with psoriatic arthritis, and the use of methotrexate appears to attenuate the presence of antidrug antibodies, according to findings from a cross-sectional study.
Although researchers have looked at the immunogenicity of TNF-alpha blockers in patients with diseases such as rheumatoid arthritis (RA), ankylosing spondylitis, psoriasis, and inflammatory bowel disease, their immunogenicity in psoriatic arthritis (PsA) patients has not been fully investigated, noted the current study’s investigators, led by Dr. Michael Zisapel of the department of rheumatology at Tel Aviv University (J. Rheumatol. 2014 Nov. 15 [doi:10.3899/jrheum.140685]).
The prevalence of antidrug antibodies (ADAb) to TNF-alpha blockers has been reported to be 20%-40% in RA, 25%-64% in ankylosing spondylitis, and about 33% in psoriasis, and evidence has shown that this is significantly reduced by the use of methotrexate, they said.
The study involved 93 patients with PsA who were taking adalimumab (n = 48), infliximab (n = 24), and etanercept (n = 21). A quarter of the patients were taking methotrexate at an average dose of 13.3 mg/week.
Overall, 77% of the patients had therapeutic drug levels. The prevalence of immunogenicity in the entire group was 33.3%, and one-fifth of patients had high concentrations of antibodies.
High levels of ADAb were found in 29% of patients taking adalimumab and 21% of the patients taking infliximab. No ADAb levels were found in patients taking etanercept.
Interestingly, the patients taking adalimumab demonstrated an immunogenicity of 54%, but only half of them (29%) showed high concentrations of antibodies.
“This finding suggests that a variety of levels might be found that might affect the significance of immunogenicity differently among patients producing ADAb,” the investigators wrote.
Fewer methotrexate-treated patients had high ADAb concentrations, compared with patients not taking methotrexate (16.7% vs. 21.7%).
A clear correlation was found between the presence of immunogenicity, lower drug levels, and decreased clinical response in PsA patients, just as other studies have found in RA and ankylosing spondylitis patients, the authors noted.
“Our results suggest that the use of methotrexate should be strongly considered in addition to monoclonal antibodies,” they said.
Limitations of their study included the small number of patients and the use of a bridging ELISA test which is reliable but considered to be less accurate than radioimmunoassay.
No disclosure information was available.
Immunogenicity to three TNF-alpha blocking agents seems to vary substantially among patients with psoriatic arthritis, and the use of methotrexate appears to attenuate the presence of antidrug antibodies, according to findings from a cross-sectional study.
Although researchers have looked at the immunogenicity of TNF-alpha blockers in patients with diseases such as rheumatoid arthritis (RA), ankylosing spondylitis, psoriasis, and inflammatory bowel disease, their immunogenicity in psoriatic arthritis (PsA) patients has not been fully investigated, noted the current study’s investigators, led by Dr. Michael Zisapel of the department of rheumatology at Tel Aviv University (J. Rheumatol. 2014 Nov. 15 [doi:10.3899/jrheum.140685]).
The prevalence of antidrug antibodies (ADAb) to TNF-alpha blockers has been reported to be 20%-40% in RA, 25%-64% in ankylosing spondylitis, and about 33% in psoriasis, and evidence has shown that this is significantly reduced by the use of methotrexate, they said.
The study involved 93 patients with PsA who were taking adalimumab (n = 48), infliximab (n = 24), and etanercept (n = 21). A quarter of the patients were taking methotrexate at an average dose of 13.3 mg/week.
Overall, 77% of the patients had therapeutic drug levels. The prevalence of immunogenicity in the entire group was 33.3%, and one-fifth of patients had high concentrations of antibodies.
High levels of ADAb were found in 29% of patients taking adalimumab and 21% of the patients taking infliximab. No ADAb levels were found in patients taking etanercept.
Interestingly, the patients taking adalimumab demonstrated an immunogenicity of 54%, but only half of them (29%) showed high concentrations of antibodies.
“This finding suggests that a variety of levels might be found that might affect the significance of immunogenicity differently among patients producing ADAb,” the investigators wrote.
Fewer methotrexate-treated patients had high ADAb concentrations, compared with patients not taking methotrexate (16.7% vs. 21.7%).
A clear correlation was found between the presence of immunogenicity, lower drug levels, and decreased clinical response in PsA patients, just as other studies have found in RA and ankylosing spondylitis patients, the authors noted.
“Our results suggest that the use of methotrexate should be strongly considered in addition to monoclonal antibodies,” they said.
Limitations of their study included the small number of patients and the use of a bridging ELISA test which is reliable but considered to be less accurate than radioimmunoassay.
No disclosure information was available.
FROM JOURNAL OF RHEUMATOLOGY
Key clinical point: The use of methotrexate should be strongly considered in addition to TNF-alpha blockers in patients with psoriatic arthritis to reduce the presence and influence of antidrug antibodies.
Major finding: High levels of antidrug antibodies were found in 29% of patients taking adalimumab, 21% of the patients taking infliximab, and no patients taking etanercept.
Data source: A cross-sectional study of 93 consecutive psoriatic arthritis patients.
Disclosures: No disclosure information was available.
Psoriasis patients have higher rate of low back pain
The prevalence of low back pain appears to be higher in people with psoriasis than in the general population, according to an analysis of national survey data.
The findings may change the way psoriasis patients are managed when they present to primary care or specialty clinics with sudden-onset back and buttock pain, said first author of the study, Dr. Nicole Thom of the division of rheumatology at Cedars-Sinai Medical Center, Los Angeles, and her colleagues (Arthritis Care Res. 2014 Dec. 2 [doi:10.1002/acr.22528]).
Using data from the 2009-2010 U.S. National Health and Nutrition Examination Survey of 6,684 adults, the researchers identified 5,103 people who had answered questions on back pain. A total of 148 had psoriasis and 5 had psoriatic arthritis (PsA).
People with psoriasis/PsA had a significantly higher prevalence of axial pain as measured using the 3-month duration criteria, compared with people without the disease (31.1% vs. 18.9%; P = .04). They were also more likely to have alternating buttock pain (7.2% vs. 2.4%; P = .03) and meet Berlin 7b and 8a criteria for inflammatory back pain (P = .04 and P = .02, respectively). The prevalence of spondyloarthritis was significantly higher in the psoriasis/PsA group when using Amor or European Spondyloarthritis Study Group criteria (14.3% vs. 1.5%; P = .001). Sudden onset of axial pain was also higher in the psoriasis/PsA group (23.3% vs. 13.0%; P = .01), the researchers reported.
“The internist or family medicine physician should include inflammatory back pain in their differential diagnosis,” the study authors suggest.
With more and more research continuing to support multiple comorbidities in psoriasis, it also raises the question as to whether rheumatologists, dermatologists, and other health care professionals should be screening for them, they said.
The work was supported in part by grants from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and a National Center for Research Resources grant to the Clinical Translational Science Institute at the University of California, Los Angeles.
The prevalence of low back pain appears to be higher in people with psoriasis than in the general population, according to an analysis of national survey data.
The findings may change the way psoriasis patients are managed when they present to primary care or specialty clinics with sudden-onset back and buttock pain, said first author of the study, Dr. Nicole Thom of the division of rheumatology at Cedars-Sinai Medical Center, Los Angeles, and her colleagues (Arthritis Care Res. 2014 Dec. 2 [doi:10.1002/acr.22528]).
Using data from the 2009-2010 U.S. National Health and Nutrition Examination Survey of 6,684 adults, the researchers identified 5,103 people who had answered questions on back pain. A total of 148 had psoriasis and 5 had psoriatic arthritis (PsA).
People with psoriasis/PsA had a significantly higher prevalence of axial pain as measured using the 3-month duration criteria, compared with people without the disease (31.1% vs. 18.9%; P = .04). They were also more likely to have alternating buttock pain (7.2% vs. 2.4%; P = .03) and meet Berlin 7b and 8a criteria for inflammatory back pain (P = .04 and P = .02, respectively). The prevalence of spondyloarthritis was significantly higher in the psoriasis/PsA group when using Amor or European Spondyloarthritis Study Group criteria (14.3% vs. 1.5%; P = .001). Sudden onset of axial pain was also higher in the psoriasis/PsA group (23.3% vs. 13.0%; P = .01), the researchers reported.
“The internist or family medicine physician should include inflammatory back pain in their differential diagnosis,” the study authors suggest.
With more and more research continuing to support multiple comorbidities in psoriasis, it also raises the question as to whether rheumatologists, dermatologists, and other health care professionals should be screening for them, they said.
The work was supported in part by grants from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and a National Center for Research Resources grant to the Clinical Translational Science Institute at the University of California, Los Angeles.
The prevalence of low back pain appears to be higher in people with psoriasis than in the general population, according to an analysis of national survey data.
The findings may change the way psoriasis patients are managed when they present to primary care or specialty clinics with sudden-onset back and buttock pain, said first author of the study, Dr. Nicole Thom of the division of rheumatology at Cedars-Sinai Medical Center, Los Angeles, and her colleagues (Arthritis Care Res. 2014 Dec. 2 [doi:10.1002/acr.22528]).
Using data from the 2009-2010 U.S. National Health and Nutrition Examination Survey of 6,684 adults, the researchers identified 5,103 people who had answered questions on back pain. A total of 148 had psoriasis and 5 had psoriatic arthritis (PsA).
People with psoriasis/PsA had a significantly higher prevalence of axial pain as measured using the 3-month duration criteria, compared with people without the disease (31.1% vs. 18.9%; P = .04). They were also more likely to have alternating buttock pain (7.2% vs. 2.4%; P = .03) and meet Berlin 7b and 8a criteria for inflammatory back pain (P = .04 and P = .02, respectively). The prevalence of spondyloarthritis was significantly higher in the psoriasis/PsA group when using Amor or European Spondyloarthritis Study Group criteria (14.3% vs. 1.5%; P = .001). Sudden onset of axial pain was also higher in the psoriasis/PsA group (23.3% vs. 13.0%; P = .01), the researchers reported.
“The internist or family medicine physician should include inflammatory back pain in their differential diagnosis,” the study authors suggest.
With more and more research continuing to support multiple comorbidities in psoriasis, it also raises the question as to whether rheumatologists, dermatologists, and other health care professionals should be screening for them, they said.
The work was supported in part by grants from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and a National Center for Research Resources grant to the Clinical Translational Science Institute at the University of California, Los Angeles.
FROM ARTHRITIS CARE & RESEARCH
Calcipotriene 0.005%–Betamethasone Dipropionate 0.064% Ointment Versus Topical Suspension in the Treatment of Plaque Psoriasis: A Randomized Pilot Study of Patient Preference
Psoriasis is a chronic relapsing inflammatory skin and joint disease that affects 1% to 3% of the US population.1 In cases of mild to moderate disease, topical agents including corticosteroids, vitamin D analogues, and retinoids are the mainstay of therapy. The need for long-term treatment can be frustrating for patients and treatment adherence often is problematic, resulting in poor outcomes. Reported adherence rates to topical psoriasis treatments range from 27% to 73%.2-6
Topical agents for treatment of psoriasis are available in various formulations, including creams, lotions, gels, ointments, solutions, and shampoos. For topical psoriasis therapies, vehicle formulation plays a major role in both delivery of the active drug and treatment adherence. Patients often cite poor cosmetic characteristics (eg, product feels too sticky or greasy, product feels unpleasant/has a bad texture, product is too messy, product application is too time consuming/takes too long to rub in) as reasons for poor treatment adherence.2,6-9 Psoriasis patients tend to prefer formulations that are not as messy such as solutions and foams versus creams, gels, and ointments.10
Ointments have been favored by physicians for the treatment of psoriasis because of the belief that their occlusive properties result in greater potency; however, a systematic review of clinical trials of different formulations of clobetasol propionate did not find that ointments were more effective than other vehicles.11 Furthermore, if a patient finds an ointment to be cosmetically unacceptable, he/she will be less inclined to use the medication as prescribed, regardless of its potency.
The objective of this study was to conduct a preliminary assessment of patient preference for ointment versus topical suspension formulations of calcipotriene 0.005%–betamethasone dipropionate 0.064% for treatment of plaque psoriasis. The specific attributes that were found to be appealing or unappealing by participants for each formulation also were evaluated.
Methods
Study Design and Participants
This open-label, investigator-blinded, crossover, prospective, single-center study evaluated patient preference for ointment versus topical suspension formulations of calcipo-triene 0.005%–betamethasone dipropionate 0.064% in the treatment of plaque psoriasis. The study protocol was approved by the Wake Forest School of Medicine (Winston-Salem, North Carolina) institutional review board.
Participants were recruited from the Wake Forest School of Medicine dermatology clinic. Inclusion criteria included men and women with mild to moderate plaque-type psoriasis who were 18 years of age or older; participants also were required to have an investigator global assessment (IGA) score of 2 (mild) or 3 (moderate) on a 5-point scale and 1% to 10% body surface involvement on the trunk or extremities.
Exclusion criteria included use of a topical or systemic psoriasis treatment within 2 or 4 weeks of baseline, respectively. Women who were pregnant, breastfeeding, planning to become pregnant, or could potentially become pregnant and were not using a medically accepted form of contraception also were excluded from the study. Patients with other serious skin conditions or any other chronic medical conditions that were not well controlled also were considered ineligible.
As a pilot study, a sample size of 20 participants was needed based on available funding.
Assessments
At baseline, the diagnosis of plaque-type psoriasis in each participant was confirmed by the investigator. Each participant’s medical history was obtained and all prior and current medications were reviewed to ensure eligibility criteria were met. Female participants of childbearing potential also underwent a urine pregnancy test. Consent was obtained from all enrolled participants. Investigators assessed the severity of psoriasis at baseline using the IGA.
All participants then were randomized (1:1 randomization) for treatment with either calcipotriene 0.005%–betamethasone dipropionate 0.064% ointment or calcipotriene 0.005%–betamethasone dipropionate 0.064% topical suspension. A simple random sampling chart was prepared and used by the study staff to determine treatment group allotment. Participants were instructed to apply the assigned study drug to affected areas of the body, excluding the scalp, face, and intertriginous areas, once daily for 3 days. Participants and study staff were aware of the study product being used, while investigators remained blinded. Participants also were asked to maintain a daily medication diary noting when the medication was applied.
Participants returned on day 3 for crossover treatment. Participants were asked to complete a subjective participant preference survey and study staff reviewed concomitant medications and adverse events. The packaging for the initial study drug was collected and weighed, and the crossover drug was dispensed to each participant to be applied once daily for 3 days.
Participants returned on day 6 or 7 for follow-up and were again asked to complete the subjective participant preference survey; study staff reviewed concomitant medications and adverse events. The packaging for the crossover study drug was collected and weighed, and the participant’s medication diary also was collected.
Subjective Participant Preference Survey
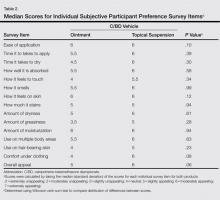
The subjective participant preference survey consisted of 15 questions relating to the participant’s experience with the study drug (eg, how the product felt to touch, amount of greasiness, time it took to apply). The final survey question asked participants to rate the overall appeal of the vehicle. Participants responded to the questions using a 7-point grading scale (1=extremely unappealing; 4=neutral; 7=extremely appealing). Total preference scores could range from 15 to 105.
End Points
The primary end point was the mean total preference score for each study drug obtained from the subjective participant preference surveys. Secondary end points included median values for individual survey questions and treatment adherence, which was measured from self-reported medication diary entries.
Statistical Analysis
Participant characteristics were reported with percentages for dichotomous data and median and ranges for other data. Subjective participant preference survey scores were calculated by taking the mean (standard deviation [SD]) sum of the scores for each individual survey item for both products. A generalized linear model that accounted for possible carryover and period effects was used to compare the difference of individual participant scores for the 2 products using SAS software. The mean (SD) amount of product used was reported and correlated to the preference score using a Spearman rank correlation. Total and individual survey scores were compared between sexes using Wilcoxon rank sum tests.
Results
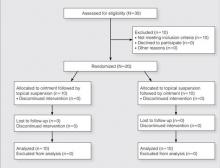
Participants were enrolled in and completed the study from January 2013 to March 2013. The Figure presents a diagram of the Consolidated Standards of Reporting Trials. Thirty patients were screened; 10 patients did not meet eligibility criteria. Twenty patients were enrolled in the study with 10 patients randomized to each study arm. All 20 participants completed the study.
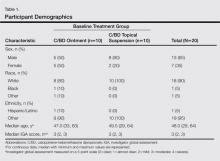
Participant demographics are described in Table 1. The median age was 48 years (range 29–64 years). The majority of participants were male (13/20) and white (18/20). The median IGA score was 3 (range, 2–3).
The mean (SD) total preference score for the calcipotriene 0.005%–betamethasone dipropionate 0.064% ointment formulation was 73.5 (19.4) and 80 (20.2) for the topical suspension formulation. The difference between means was -6.5 (95% confidence interval, -19.7-6.8; P=.32 after adjusting for possible carryover and period effects). Participants used a mean (SD) of 11 (11.4) g of product per study arm for the ointment formulation and 8.8 (6.6) g for the topical suspension formulation. There was no correlation between the amount of product used and preference for product (Spearman r=-0.01; P=.94). No statistically significant difference in product preference among men versus women was noted when considering total preference score or median scores of individual survey questions. Median overall appeal rating for the ointment formulation was 5 (slightly appealing) versus 6 (moderately appealing) for the topical suspension formulation, approaching statistical significance with P=.06 (Table 2). No significant carryover effects from one product to the other were noted (P=.64). The mean (SD) total preference scores were 81.1 (18.4) and 77.6 (21.2) in participants who used the topical suspension first followed by the ointment. In participants who used the ointment first followed by the topical suspension, the mean (SD) total preference scores were 69.4 (17.6) and 78.9 (22.8). Self-reported treatment adherence according to the participant’s daily medication diary was 100%.
Adverse effects during the study included 1 report of neck and back muscle pain and 1 report of sinusitis; neither was considered to be related to the study drugs.
Comment
Psoriasis is a chronic disease that can be difficult to treat, and treatment compliance often is poor. Multiple topical agents often are needed for adequate disease control, and adherence can be an even greater hurdle than with monotherapy. Combination products such as calcipotriene–betamethasone dipropionate offer the potential advantage of once-daily application. Adherence to once-daily application regimens for treatment of psoriasis has been shown to be greater than twice-daily application (82% vs 44%).4 However, the vehicle may be an adherence barrier for some patients.
Calcipotriene 0.005%–betamethasone dipropionate 0.064% topical suspension originally was indicated for the treatment of psoriasis of the scalp; however, it is now also indicated for treatment of psoriasis of the body.12 This topical suspension formulation is less messy, which could potentially be more cosmetically appealing and useful for improving treatment adherence.
Overall, the participants in our small pilot study showed a preference for the topical suspension versus the ointment formulation. The difference was substantial but was not statistically significant. This result is consistent with a previous study in which patients were found to prefer solutions that were less greasy compared to messy sticky ointments.10 Although the topical suspension received a higher average rating from participants for how it felt to touch and how it felt under clothing than the ointment and the ointment was rated on average as more greasy (none of these individual items achieved statistical significance), the calcipotriene–betamethasone dipropionate ointment was still rated as slightly appealing overall (Table 2). This result supports the need for physicians to discuss individual patient preferences when choosing the most appropriate vehicle for topical psoriasis treatment.
In our study, participants were found to use less product during treatment with the topical suspension versus the ointment, likely because the topical suspension formulation is thinner and spreads easier; however, participants rated the ease of application of the 2 products equally (Table 2). Ease of application was rated moderately appealing and time to apply was rated slightly to moderately appealing, which is important because patients often cite these factors as barriers to treatment adherence.2,6,13
The occlusive properties of ointment formulations provide moisturization by preventing water loss, a property that can be desirable when treating psoriatic plaques. Unlike the ointment, which was formulated with petrolatum and mineral oil, the calcipotriene–betamethasone dipropionate topical suspension was formulated with hydrogenated castor oil and mineral oil to provide moisturization. Nevertheless, participants found both the ointment and topical suspension to be moderately appealing (median score, 6; P=.94) for moisturization.
Limitations of this pilot study include the small sample size, which restricted the extent of subgroup analyses and the generalizability of our results. The small sample size also may or may not have contributed to the lack of statistical significance in the majority of the outcomes. This study provides pilot data that can be used to define a larger study; however, we do not think that a larger study is needed, as patients can be offered both vehicles in a practical clinical setting and can choose the product that is right for them. The short treatment duration of 3 days for each formulation also is a limitation, as a patient’s preference may change over time with longer use of the product. Treatment efficacy, which was not measured in this study, also could have an effect on patient preference, which could be assessed over a longer treatment period. Additionally, the study drugs may not be representative of all ointments or topical suspensions in their cosmetic appeal.
Conclusion
In this small cohort of plaque psoriasis patients, a calcipotriene–betamethasone dipropionate topical suspension was preferred over an ointment formulation, but in clinical practice it may be best to allow patients to choose the vehicle formulation that is most desirable on an individual basis. The topical suspension provides clinicians with an alternative that not only has the benefits of a combination product but also has been found to be appealing to patients.
1. Parisi R, Symmons DP, Griffiths CE, et al. Global epidemiology of psoriasis: a systematic review of incidence and prevalence. J Invest Dermatol. 2013;133:377-385.
2. Brown KK, Rehmus WE, Kimball AB. Determining the relative importance of patient motivations for nonadherence to topical corticosteroid therapy in psoriasis. J Am Acad Dermatol. 2006;55:607-613.
3. Carroll CL, Feldman SR, Camacho FT, et al. Adherence to topical therapy decreases during the course of an 8-week psoriasis clinical trial: commonly used methods of measuring adherence to topical therapy overestimate actual use. J Am Acad Dermatol. 2004;51:212-216.
4. Zaghloul SS, Goodfield MJ. Objective assessment of compliance with psoriasis treatment. Arch Dermatol. 2004;140:408-414.
5. Richards HL, Fortune DG, O’Sullivan TM, et al. Patients with psoriasis and their compliance with medication. J Am Acad Dermatol. 1999;41:581-583.
6. Fouere S, Adjadj L, Pawin H. How patients experience psoriasis: results from a European survey. J Eur Acad Dermatol Venereol. 2005;19:S2-S6.
7. van de Kerkhof PC, Steegers-Theunissen RP, Kuipers MV. Evaluation of topical drug treatment in psoriasis. Dermatology. 1998;197:31-36.
8. Devaux S, Castela A, Archier E, et al. Adherence to topical treatment in psoriasis: a systematic literature review. J Eur Acad Dermatol Venereol. 2012;26(suppl 3):S61-S67.
9. Chan SA, Hussain F, Lawson LG, et al. Factors affecting adherence to treatment of psoriasis: comparing biologic therapy to other modalities. J Dermatolog Treat. 2013;24:64-69.
10. Housman TS, Mellen BG, Rapp SR, et al. Patients with psoriasis prefer solution and foam vehicles: a quantitative assessment of vehicle preference. Cutis. 2002;70:327-332.
11. Warino L, Balkrishnan R, Feldman SR. Clobetasol propionate for psoriasis: are ointments really more potent? J Drugs Dermatol. 2006;5:527-532.
12. Menter A, Gold LS, Bukhalo M, et al. Calcipotriene plus betamethasone dipropionate topical suspension for the treatment of mild to moderate psoriasis vulgaris on the body: a randomized, double-blind, vehicle-controlled trial. J Drugs Dermatol. 2013;12:92-98.
13. Krueger G, Koo J, Lebwohl M, et al. The impact of psoriasis on quality of life: results of a 1998 National Psoriasis Foundation patient-membership survey. Arch Dermatol. 2001;137:280-284.
Psoriasis is a chronic relapsing inflammatory skin and joint disease that affects 1% to 3% of the US population.1 In cases of mild to moderate disease, topical agents including corticosteroids, vitamin D analogues, and retinoids are the mainstay of therapy. The need for long-term treatment can be frustrating for patients and treatment adherence often is problematic, resulting in poor outcomes. Reported adherence rates to topical psoriasis treatments range from 27% to 73%.2-6
Topical agents for treatment of psoriasis are available in various formulations, including creams, lotions, gels, ointments, solutions, and shampoos. For topical psoriasis therapies, vehicle formulation plays a major role in both delivery of the active drug and treatment adherence. Patients often cite poor cosmetic characteristics (eg, product feels too sticky or greasy, product feels unpleasant/has a bad texture, product is too messy, product application is too time consuming/takes too long to rub in) as reasons for poor treatment adherence.2,6-9 Psoriasis patients tend to prefer formulations that are not as messy such as solutions and foams versus creams, gels, and ointments.10
Ointments have been favored by physicians for the treatment of psoriasis because of the belief that their occlusive properties result in greater potency; however, a systematic review of clinical trials of different formulations of clobetasol propionate did not find that ointments were more effective than other vehicles.11 Furthermore, if a patient finds an ointment to be cosmetically unacceptable, he/she will be less inclined to use the medication as prescribed, regardless of its potency.
The objective of this study was to conduct a preliminary assessment of patient preference for ointment versus topical suspension formulations of calcipotriene 0.005%–betamethasone dipropionate 0.064% for treatment of plaque psoriasis. The specific attributes that were found to be appealing or unappealing by participants for each formulation also were evaluated.
Methods
Study Design and Participants
This open-label, investigator-blinded, crossover, prospective, single-center study evaluated patient preference for ointment versus topical suspension formulations of calcipo-triene 0.005%–betamethasone dipropionate 0.064% in the treatment of plaque psoriasis. The study protocol was approved by the Wake Forest School of Medicine (Winston-Salem, North Carolina) institutional review board.
Participants were recruited from the Wake Forest School of Medicine dermatology clinic. Inclusion criteria included men and women with mild to moderate plaque-type psoriasis who were 18 years of age or older; participants also were required to have an investigator global assessment (IGA) score of 2 (mild) or 3 (moderate) on a 5-point scale and 1% to 10% body surface involvement on the trunk or extremities.
Exclusion criteria included use of a topical or systemic psoriasis treatment within 2 or 4 weeks of baseline, respectively. Women who were pregnant, breastfeeding, planning to become pregnant, or could potentially become pregnant and were not using a medically accepted form of contraception also were excluded from the study. Patients with other serious skin conditions or any other chronic medical conditions that were not well controlled also were considered ineligible.
As a pilot study, a sample size of 20 participants was needed based on available funding.
Assessments
At baseline, the diagnosis of plaque-type psoriasis in each participant was confirmed by the investigator. Each participant’s medical history was obtained and all prior and current medications were reviewed to ensure eligibility criteria were met. Female participants of childbearing potential also underwent a urine pregnancy test. Consent was obtained from all enrolled participants. Investigators assessed the severity of psoriasis at baseline using the IGA.
All participants then were randomized (1:1 randomization) for treatment with either calcipotriene 0.005%–betamethasone dipropionate 0.064% ointment or calcipotriene 0.005%–betamethasone dipropionate 0.064% topical suspension. A simple random sampling chart was prepared and used by the study staff to determine treatment group allotment. Participants were instructed to apply the assigned study drug to affected areas of the body, excluding the scalp, face, and intertriginous areas, once daily for 3 days. Participants and study staff were aware of the study product being used, while investigators remained blinded. Participants also were asked to maintain a daily medication diary noting when the medication was applied.
Participants returned on day 3 for crossover treatment. Participants were asked to complete a subjective participant preference survey and study staff reviewed concomitant medications and adverse events. The packaging for the initial study drug was collected and weighed, and the crossover drug was dispensed to each participant to be applied once daily for 3 days.
Participants returned on day 6 or 7 for follow-up and were again asked to complete the subjective participant preference survey; study staff reviewed concomitant medications and adverse events. The packaging for the crossover study drug was collected and weighed, and the participant’s medication diary also was collected.
Subjective Participant Preference Survey
The subjective participant preference survey consisted of 15 questions relating to the participant’s experience with the study drug (eg, how the product felt to touch, amount of greasiness, time it took to apply). The final survey question asked participants to rate the overall appeal of the vehicle. Participants responded to the questions using a 7-point grading scale (1=extremely unappealing; 4=neutral; 7=extremely appealing). Total preference scores could range from 15 to 105.
End Points
The primary end point was the mean total preference score for each study drug obtained from the subjective participant preference surveys. Secondary end points included median values for individual survey questions and treatment adherence, which was measured from self-reported medication diary entries.
Statistical Analysis
Participant characteristics were reported with percentages for dichotomous data and median and ranges for other data. Subjective participant preference survey scores were calculated by taking the mean (standard deviation [SD]) sum of the scores for each individual survey item for both products. A generalized linear model that accounted for possible carryover and period effects was used to compare the difference of individual participant scores for the 2 products using SAS software. The mean (SD) amount of product used was reported and correlated to the preference score using a Spearman rank correlation. Total and individual survey scores were compared between sexes using Wilcoxon rank sum tests.
Results
Participants were enrolled in and completed the study from January 2013 to March 2013. The Figure presents a diagram of the Consolidated Standards of Reporting Trials. Thirty patients were screened; 10 patients did not meet eligibility criteria. Twenty patients were enrolled in the study with 10 patients randomized to each study arm. All 20 participants completed the study.
Participant demographics are described in Table 1. The median age was 48 years (range 29–64 years). The majority of participants were male (13/20) and white (18/20). The median IGA score was 3 (range, 2–3).
The mean (SD) total preference score for the calcipotriene 0.005%–betamethasone dipropionate 0.064% ointment formulation was 73.5 (19.4) and 80 (20.2) for the topical suspension formulation. The difference between means was -6.5 (95% confidence interval, -19.7-6.8; P=.32 after adjusting for possible carryover and period effects). Participants used a mean (SD) of 11 (11.4) g of product per study arm for the ointment formulation and 8.8 (6.6) g for the topical suspension formulation. There was no correlation between the amount of product used and preference for product (Spearman r=-0.01; P=.94). No statistically significant difference in product preference among men versus women was noted when considering total preference score or median scores of individual survey questions. Median overall appeal rating for the ointment formulation was 5 (slightly appealing) versus 6 (moderately appealing) for the topical suspension formulation, approaching statistical significance with P=.06 (Table 2). No significant carryover effects from one product to the other were noted (P=.64). The mean (SD) total preference scores were 81.1 (18.4) and 77.6 (21.2) in participants who used the topical suspension first followed by the ointment. In participants who used the ointment first followed by the topical suspension, the mean (SD) total preference scores were 69.4 (17.6) and 78.9 (22.8). Self-reported treatment adherence according to the participant’s daily medication diary was 100%.
Adverse effects during the study included 1 report of neck and back muscle pain and 1 report of sinusitis; neither was considered to be related to the study drugs.
Comment
Psoriasis is a chronic disease that can be difficult to treat, and treatment compliance often is poor. Multiple topical agents often are needed for adequate disease control, and adherence can be an even greater hurdle than with monotherapy. Combination products such as calcipotriene–betamethasone dipropionate offer the potential advantage of once-daily application. Adherence to once-daily application regimens for treatment of psoriasis has been shown to be greater than twice-daily application (82% vs 44%).4 However, the vehicle may be an adherence barrier for some patients.
Calcipotriene 0.005%–betamethasone dipropionate 0.064% topical suspension originally was indicated for the treatment of psoriasis of the scalp; however, it is now also indicated for treatment of psoriasis of the body.12 This topical suspension formulation is less messy, which could potentially be more cosmetically appealing and useful for improving treatment adherence.
Overall, the participants in our small pilot study showed a preference for the topical suspension versus the ointment formulation. The difference was substantial but was not statistically significant. This result is consistent with a previous study in which patients were found to prefer solutions that were less greasy compared to messy sticky ointments.10 Although the topical suspension received a higher average rating from participants for how it felt to touch and how it felt under clothing than the ointment and the ointment was rated on average as more greasy (none of these individual items achieved statistical significance), the calcipotriene–betamethasone dipropionate ointment was still rated as slightly appealing overall (Table 2). This result supports the need for physicians to discuss individual patient preferences when choosing the most appropriate vehicle for topical psoriasis treatment.
In our study, participants were found to use less product during treatment with the topical suspension versus the ointment, likely because the topical suspension formulation is thinner and spreads easier; however, participants rated the ease of application of the 2 products equally (Table 2). Ease of application was rated moderately appealing and time to apply was rated slightly to moderately appealing, which is important because patients often cite these factors as barriers to treatment adherence.2,6,13
The occlusive properties of ointment formulations provide moisturization by preventing water loss, a property that can be desirable when treating psoriatic plaques. Unlike the ointment, which was formulated with petrolatum and mineral oil, the calcipotriene–betamethasone dipropionate topical suspension was formulated with hydrogenated castor oil and mineral oil to provide moisturization. Nevertheless, participants found both the ointment and topical suspension to be moderately appealing (median score, 6; P=.94) for moisturization.
Limitations of this pilot study include the small sample size, which restricted the extent of subgroup analyses and the generalizability of our results. The small sample size also may or may not have contributed to the lack of statistical significance in the majority of the outcomes. This study provides pilot data that can be used to define a larger study; however, we do not think that a larger study is needed, as patients can be offered both vehicles in a practical clinical setting and can choose the product that is right for them. The short treatment duration of 3 days for each formulation also is a limitation, as a patient’s preference may change over time with longer use of the product. Treatment efficacy, which was not measured in this study, also could have an effect on patient preference, which could be assessed over a longer treatment period. Additionally, the study drugs may not be representative of all ointments or topical suspensions in their cosmetic appeal.
Conclusion
In this small cohort of plaque psoriasis patients, a calcipotriene–betamethasone dipropionate topical suspension was preferred over an ointment formulation, but in clinical practice it may be best to allow patients to choose the vehicle formulation that is most desirable on an individual basis. The topical suspension provides clinicians with an alternative that not only has the benefits of a combination product but also has been found to be appealing to patients.
Psoriasis is a chronic relapsing inflammatory skin and joint disease that affects 1% to 3% of the US population.1 In cases of mild to moderate disease, topical agents including corticosteroids, vitamin D analogues, and retinoids are the mainstay of therapy. The need for long-term treatment can be frustrating for patients and treatment adherence often is problematic, resulting in poor outcomes. Reported adherence rates to topical psoriasis treatments range from 27% to 73%.2-6
Topical agents for treatment of psoriasis are available in various formulations, including creams, lotions, gels, ointments, solutions, and shampoos. For topical psoriasis therapies, vehicle formulation plays a major role in both delivery of the active drug and treatment adherence. Patients often cite poor cosmetic characteristics (eg, product feels too sticky or greasy, product feels unpleasant/has a bad texture, product is too messy, product application is too time consuming/takes too long to rub in) as reasons for poor treatment adherence.2,6-9 Psoriasis patients tend to prefer formulations that are not as messy such as solutions and foams versus creams, gels, and ointments.10
Ointments have been favored by physicians for the treatment of psoriasis because of the belief that their occlusive properties result in greater potency; however, a systematic review of clinical trials of different formulations of clobetasol propionate did not find that ointments were more effective than other vehicles.11 Furthermore, if a patient finds an ointment to be cosmetically unacceptable, he/she will be less inclined to use the medication as prescribed, regardless of its potency.
The objective of this study was to conduct a preliminary assessment of patient preference for ointment versus topical suspension formulations of calcipotriene 0.005%–betamethasone dipropionate 0.064% for treatment of plaque psoriasis. The specific attributes that were found to be appealing or unappealing by participants for each formulation also were evaluated.
Methods
Study Design and Participants
This open-label, investigator-blinded, crossover, prospective, single-center study evaluated patient preference for ointment versus topical suspension formulations of calcipo-triene 0.005%–betamethasone dipropionate 0.064% in the treatment of plaque psoriasis. The study protocol was approved by the Wake Forest School of Medicine (Winston-Salem, North Carolina) institutional review board.
Participants were recruited from the Wake Forest School of Medicine dermatology clinic. Inclusion criteria included men and women with mild to moderate plaque-type psoriasis who were 18 years of age or older; participants also were required to have an investigator global assessment (IGA) score of 2 (mild) or 3 (moderate) on a 5-point scale and 1% to 10% body surface involvement on the trunk or extremities.
Exclusion criteria included use of a topical or systemic psoriasis treatment within 2 or 4 weeks of baseline, respectively. Women who were pregnant, breastfeeding, planning to become pregnant, or could potentially become pregnant and were not using a medically accepted form of contraception also were excluded from the study. Patients with other serious skin conditions or any other chronic medical conditions that were not well controlled also were considered ineligible.
As a pilot study, a sample size of 20 participants was needed based on available funding.
Assessments
At baseline, the diagnosis of plaque-type psoriasis in each participant was confirmed by the investigator. Each participant’s medical history was obtained and all prior and current medications were reviewed to ensure eligibility criteria were met. Female participants of childbearing potential also underwent a urine pregnancy test. Consent was obtained from all enrolled participants. Investigators assessed the severity of psoriasis at baseline using the IGA.
All participants then were randomized (1:1 randomization) for treatment with either calcipotriene 0.005%–betamethasone dipropionate 0.064% ointment or calcipotriene 0.005%–betamethasone dipropionate 0.064% topical suspension. A simple random sampling chart was prepared and used by the study staff to determine treatment group allotment. Participants were instructed to apply the assigned study drug to affected areas of the body, excluding the scalp, face, and intertriginous areas, once daily for 3 days. Participants and study staff were aware of the study product being used, while investigators remained blinded. Participants also were asked to maintain a daily medication diary noting when the medication was applied.
Participants returned on day 3 for crossover treatment. Participants were asked to complete a subjective participant preference survey and study staff reviewed concomitant medications and adverse events. The packaging for the initial study drug was collected and weighed, and the crossover drug was dispensed to each participant to be applied once daily for 3 days.
Participants returned on day 6 or 7 for follow-up and were again asked to complete the subjective participant preference survey; study staff reviewed concomitant medications and adverse events. The packaging for the crossover study drug was collected and weighed, and the participant’s medication diary also was collected.
Subjective Participant Preference Survey
The subjective participant preference survey consisted of 15 questions relating to the participant’s experience with the study drug (eg, how the product felt to touch, amount of greasiness, time it took to apply). The final survey question asked participants to rate the overall appeal of the vehicle. Participants responded to the questions using a 7-point grading scale (1=extremely unappealing; 4=neutral; 7=extremely appealing). Total preference scores could range from 15 to 105.
End Points
The primary end point was the mean total preference score for each study drug obtained from the subjective participant preference surveys. Secondary end points included median values for individual survey questions and treatment adherence, which was measured from self-reported medication diary entries.
Statistical Analysis
Participant characteristics were reported with percentages for dichotomous data and median and ranges for other data. Subjective participant preference survey scores were calculated by taking the mean (standard deviation [SD]) sum of the scores for each individual survey item for both products. A generalized linear model that accounted for possible carryover and period effects was used to compare the difference of individual participant scores for the 2 products using SAS software. The mean (SD) amount of product used was reported and correlated to the preference score using a Spearman rank correlation. Total and individual survey scores were compared between sexes using Wilcoxon rank sum tests.
Results
Participants were enrolled in and completed the study from January 2013 to March 2013. The Figure presents a diagram of the Consolidated Standards of Reporting Trials. Thirty patients were screened; 10 patients did not meet eligibility criteria. Twenty patients were enrolled in the study with 10 patients randomized to each study arm. All 20 participants completed the study.
Participant demographics are described in Table 1. The median age was 48 years (range 29–64 years). The majority of participants were male (13/20) and white (18/20). The median IGA score was 3 (range, 2–3).
The mean (SD) total preference score for the calcipotriene 0.005%–betamethasone dipropionate 0.064% ointment formulation was 73.5 (19.4) and 80 (20.2) for the topical suspension formulation. The difference between means was -6.5 (95% confidence interval, -19.7-6.8; P=.32 after adjusting for possible carryover and period effects). Participants used a mean (SD) of 11 (11.4) g of product per study arm for the ointment formulation and 8.8 (6.6) g for the topical suspension formulation. There was no correlation between the amount of product used and preference for product (Spearman r=-0.01; P=.94). No statistically significant difference in product preference among men versus women was noted when considering total preference score or median scores of individual survey questions. Median overall appeal rating for the ointment formulation was 5 (slightly appealing) versus 6 (moderately appealing) for the topical suspension formulation, approaching statistical significance with P=.06 (Table 2). No significant carryover effects from one product to the other were noted (P=.64). The mean (SD) total preference scores were 81.1 (18.4) and 77.6 (21.2) in participants who used the topical suspension first followed by the ointment. In participants who used the ointment first followed by the topical suspension, the mean (SD) total preference scores were 69.4 (17.6) and 78.9 (22.8). Self-reported treatment adherence according to the participant’s daily medication diary was 100%.
Adverse effects during the study included 1 report of neck and back muscle pain and 1 report of sinusitis; neither was considered to be related to the study drugs.
Comment
Psoriasis is a chronic disease that can be difficult to treat, and treatment compliance often is poor. Multiple topical agents often are needed for adequate disease control, and adherence can be an even greater hurdle than with monotherapy. Combination products such as calcipotriene–betamethasone dipropionate offer the potential advantage of once-daily application. Adherence to once-daily application regimens for treatment of psoriasis has been shown to be greater than twice-daily application (82% vs 44%).4 However, the vehicle may be an adherence barrier for some patients.
Calcipotriene 0.005%–betamethasone dipropionate 0.064% topical suspension originally was indicated for the treatment of psoriasis of the scalp; however, it is now also indicated for treatment of psoriasis of the body.12 This topical suspension formulation is less messy, which could potentially be more cosmetically appealing and useful for improving treatment adherence.
Overall, the participants in our small pilot study showed a preference for the topical suspension versus the ointment formulation. The difference was substantial but was not statistically significant. This result is consistent with a previous study in which patients were found to prefer solutions that were less greasy compared to messy sticky ointments.10 Although the topical suspension received a higher average rating from participants for how it felt to touch and how it felt under clothing than the ointment and the ointment was rated on average as more greasy (none of these individual items achieved statistical significance), the calcipotriene–betamethasone dipropionate ointment was still rated as slightly appealing overall (Table 2). This result supports the need for physicians to discuss individual patient preferences when choosing the most appropriate vehicle for topical psoriasis treatment.
In our study, participants were found to use less product during treatment with the topical suspension versus the ointment, likely because the topical suspension formulation is thinner and spreads easier; however, participants rated the ease of application of the 2 products equally (Table 2). Ease of application was rated moderately appealing and time to apply was rated slightly to moderately appealing, which is important because patients often cite these factors as barriers to treatment adherence.2,6,13
The occlusive properties of ointment formulations provide moisturization by preventing water loss, a property that can be desirable when treating psoriatic plaques. Unlike the ointment, which was formulated with petrolatum and mineral oil, the calcipotriene–betamethasone dipropionate topical suspension was formulated with hydrogenated castor oil and mineral oil to provide moisturization. Nevertheless, participants found both the ointment and topical suspension to be moderately appealing (median score, 6; P=.94) for moisturization.
Limitations of this pilot study include the small sample size, which restricted the extent of subgroup analyses and the generalizability of our results. The small sample size also may or may not have contributed to the lack of statistical significance in the majority of the outcomes. This study provides pilot data that can be used to define a larger study; however, we do not think that a larger study is needed, as patients can be offered both vehicles in a practical clinical setting and can choose the product that is right for them. The short treatment duration of 3 days for each formulation also is a limitation, as a patient’s preference may change over time with longer use of the product. Treatment efficacy, which was not measured in this study, also could have an effect on patient preference, which could be assessed over a longer treatment period. Additionally, the study drugs may not be representative of all ointments or topical suspensions in their cosmetic appeal.
Conclusion
In this small cohort of plaque psoriasis patients, a calcipotriene–betamethasone dipropionate topical suspension was preferred over an ointment formulation, but in clinical practice it may be best to allow patients to choose the vehicle formulation that is most desirable on an individual basis. The topical suspension provides clinicians with an alternative that not only has the benefits of a combination product but also has been found to be appealing to patients.
1. Parisi R, Symmons DP, Griffiths CE, et al. Global epidemiology of psoriasis: a systematic review of incidence and prevalence. J Invest Dermatol. 2013;133:377-385.
2. Brown KK, Rehmus WE, Kimball AB. Determining the relative importance of patient motivations for nonadherence to topical corticosteroid therapy in psoriasis. J Am Acad Dermatol. 2006;55:607-613.
3. Carroll CL, Feldman SR, Camacho FT, et al. Adherence to topical therapy decreases during the course of an 8-week psoriasis clinical trial: commonly used methods of measuring adherence to topical therapy overestimate actual use. J Am Acad Dermatol. 2004;51:212-216.
4. Zaghloul SS, Goodfield MJ. Objective assessment of compliance with psoriasis treatment. Arch Dermatol. 2004;140:408-414.
5. Richards HL, Fortune DG, O’Sullivan TM, et al. Patients with psoriasis and their compliance with medication. J Am Acad Dermatol. 1999;41:581-583.
6. Fouere S, Adjadj L, Pawin H. How patients experience psoriasis: results from a European survey. J Eur Acad Dermatol Venereol. 2005;19:S2-S6.
7. van de Kerkhof PC, Steegers-Theunissen RP, Kuipers MV. Evaluation of topical drug treatment in psoriasis. Dermatology. 1998;197:31-36.
8. Devaux S, Castela A, Archier E, et al. Adherence to topical treatment in psoriasis: a systematic literature review. J Eur Acad Dermatol Venereol. 2012;26(suppl 3):S61-S67.
9. Chan SA, Hussain F, Lawson LG, et al. Factors affecting adherence to treatment of psoriasis: comparing biologic therapy to other modalities. J Dermatolog Treat. 2013;24:64-69.
10. Housman TS, Mellen BG, Rapp SR, et al. Patients with psoriasis prefer solution and foam vehicles: a quantitative assessment of vehicle preference. Cutis. 2002;70:327-332.
11. Warino L, Balkrishnan R, Feldman SR. Clobetasol propionate for psoriasis: are ointments really more potent? J Drugs Dermatol. 2006;5:527-532.
12. Menter A, Gold LS, Bukhalo M, et al. Calcipotriene plus betamethasone dipropionate topical suspension for the treatment of mild to moderate psoriasis vulgaris on the body: a randomized, double-blind, vehicle-controlled trial. J Drugs Dermatol. 2013;12:92-98.
13. Krueger G, Koo J, Lebwohl M, et al. The impact of psoriasis on quality of life: results of a 1998 National Psoriasis Foundation patient-membership survey. Arch Dermatol. 2001;137:280-284.
1. Parisi R, Symmons DP, Griffiths CE, et al. Global epidemiology of psoriasis: a systematic review of incidence and prevalence. J Invest Dermatol. 2013;133:377-385.
2. Brown KK, Rehmus WE, Kimball AB. Determining the relative importance of patient motivations for nonadherence to topical corticosteroid therapy in psoriasis. J Am Acad Dermatol. 2006;55:607-613.
3. Carroll CL, Feldman SR, Camacho FT, et al. Adherence to topical therapy decreases during the course of an 8-week psoriasis clinical trial: commonly used methods of measuring adherence to topical therapy overestimate actual use. J Am Acad Dermatol. 2004;51:212-216.
4. Zaghloul SS, Goodfield MJ. Objective assessment of compliance with psoriasis treatment. Arch Dermatol. 2004;140:408-414.
5. Richards HL, Fortune DG, O’Sullivan TM, et al. Patients with psoriasis and their compliance with medication. J Am Acad Dermatol. 1999;41:581-583.
6. Fouere S, Adjadj L, Pawin H. How patients experience psoriasis: results from a European survey. J Eur Acad Dermatol Venereol. 2005;19:S2-S6.
7. van de Kerkhof PC, Steegers-Theunissen RP, Kuipers MV. Evaluation of topical drug treatment in psoriasis. Dermatology. 1998;197:31-36.
8. Devaux S, Castela A, Archier E, et al. Adherence to topical treatment in psoriasis: a systematic literature review. J Eur Acad Dermatol Venereol. 2012;26(suppl 3):S61-S67.
9. Chan SA, Hussain F, Lawson LG, et al. Factors affecting adherence to treatment of psoriasis: comparing biologic therapy to other modalities. J Dermatolog Treat. 2013;24:64-69.
10. Housman TS, Mellen BG, Rapp SR, et al. Patients with psoriasis prefer solution and foam vehicles: a quantitative assessment of vehicle preference. Cutis. 2002;70:327-332.
11. Warino L, Balkrishnan R, Feldman SR. Clobetasol propionate for psoriasis: are ointments really more potent? J Drugs Dermatol. 2006;5:527-532.
12. Menter A, Gold LS, Bukhalo M, et al. Calcipotriene plus betamethasone dipropionate topical suspension for the treatment of mild to moderate psoriasis vulgaris on the body: a randomized, double-blind, vehicle-controlled trial. J Drugs Dermatol. 2013;12:92-98.
13. Krueger G, Koo J, Lebwohl M, et al. The impact of psoriasis on quality of life: results of a 1998 National Psoriasis Foundation patient-membership survey. Arch Dermatol. 2001;137:280-284.
Practice Points
- Patient preference plays an important role in adherence to treatment regimens in chronic skin diseases such as psoriasis.
- The topical suspension formulation of calcipotriene 0.005%–betamethasone dipropionate 0.064% was preferred by psoriasis patients over the ointment; however, the availability of the 2 formulations provides patients with options and allows them to choose which product they prefer.
Ixekizumab linked to decrease in psoriasis-related sexual difficulties
AMSTERDAM – Patient complaints of psoriasis-related sexual difficulties decreased significantly in response to treatment with the investigational biologic agent ixekizumab in a phase 2 dose-ranging study.
Sexual problems attributed by psoriasis patients to their skin condition are a common, underappreciated, and understudied problem. Most physicians simply don’t ask. But when they do, as was done formally in this study, it turned out that at baseline roughly one-third of the 142 participating subjects with moderate to severe chronic plaque psoriasis reported some degree of sexual problems they believed due specifically to their skin disease, Dr. Lyn Guenther reported at the annual congress of the European Academy of Dermatology and Venereology.
That rate dropped precipitously and in dose-dependent fashion in response to subcutaneous therapy with ixekizumab, a super-potent psoriasis medication directed against interleukin 17A, according to Dr. Guenther, professor of dermatology at the University of Western Ontario, London.
The study entailed double-blind randomization of the 142 participants to subcutaneous injections of ixekizumab at 10, 25, 75, or 150 mg or placebo at 0, 2, 4, 8, 12, and 16 weeks. The standard primary outcome -- the proportion of patients with a PASI 75 improvement at 12 weeks -- has previously been reported as highly positive (N. Engl. J. Med. 2012;366:1190-9).
For her secondary analysis of psoriasis-related sexual difficulties and their response to treatment, Dr. Guenther used as her metric the patients’ response to item 9 on the Dermatology Life Quality Index, which all subjects completed at weeks 0, 8, and 16. Item 9 asks the extent to which the responder’s skin “caused any sexual difficulties” during the past week. The options range from 0 (none at all) to 3 (very much). She categorized a response of 1 or more as evidence of sexual difficulties. And because of the relatively small sample size in this study, she lumped together as the low-dose therapy group those patients assigned to ixekizumab at 10 or 25 mg, with the high-dose group being comprised of patients on 75 or
After 8 weeks on low-dose ixekizumab, the proportion of patients reporting any skin disease-related sexual difficulties within the past week fell from a baseline of 30% to 16%. After 8 weeks of high-dose ixekizumab, the figure was just 7%. Those rates remained unchanged at 16 weeks. In contrast, the placebo group remained unchanged over time, with 32% of patients still reporting sexual difficulties caused by their skin disease at week 8.
The degree to which a patient’s psoriasis improved in response to therapy with the humanized monoclonal antibody was closely related to the reduction in skin-related sexual problems. Among the 76 ixekizumab-treated patients who achieved a PASI 75 response at week 16, the rate of self-reported sexual difficulties within the previous week was 7%. For those with less than a PASI 75 response, the rate was 24%.
An impressively high 38% percent of patients on high-dose ixekizumab achieved a PASI 100 response. Only 5% of them reported any skin-related sexual difficulties at week 8, as did 9% at week 16.
Dr. Guenther also looked at the data restricting the analysis to patients with more severe baseline sexual impairment as defined by a response of 2 or 3 on item 9 of the DLQI. Among patients on high-dose izekizumab, the rate dropped from 10.5% at baseline to 1.8% at week 8 to zero at week 16. For patients on low-dose izekizumab, the progression was 13.8% to 8.8% to 3.5% at week 16. Rates remained unchanged over time in the control group.
Although this phase 2 study was limited in size, Dr. Guenther found much the same thing earlier in the much larger phase 3 PHOENIX 1 and 2 trials of ustekinumab (Stelara), which together featured 1,334 psoriasis patients randomized to the human anti-interkelukin-12/23 monoclonal antibody. In that analysis, the proportion of ustekinumab-treated patients with impaired sexual function as assessed by DLQI item 9 plunged from 22.4% at baseline to 2.7% at week 12, compared with no change in placebo-treated controls. The bigger the PASI improvement, the greater the reduction in psoriasis-related sexual dysfunction (J. Eur. Acad. Dermatol. Venereol. 2011;25:851-7).
The phase 2 ixekizumab study was funded by Eli Lilly. Dr. Guenther is a consultant to the company. Positive primary outcomes in three pivotal phase 3 clinical trials of ixekizumab totalling nearly 3,900 randomized psoriasis patients have since been reported. The company plans to apply for marketing approval of the biologic in the first half of 2015.
AMSTERDAM – Patient complaints of psoriasis-related sexual difficulties decreased significantly in response to treatment with the investigational biologic agent ixekizumab in a phase 2 dose-ranging study.
Sexual problems attributed by psoriasis patients to their skin condition are a common, underappreciated, and understudied problem. Most physicians simply don’t ask. But when they do, as was done formally in this study, it turned out that at baseline roughly one-third of the 142 participating subjects with moderate to severe chronic plaque psoriasis reported some degree of sexual problems they believed due specifically to their skin disease, Dr. Lyn Guenther reported at the annual congress of the European Academy of Dermatology and Venereology.
That rate dropped precipitously and in dose-dependent fashion in response to subcutaneous therapy with ixekizumab, a super-potent psoriasis medication directed against interleukin 17A, according to Dr. Guenther, professor of dermatology at the University of Western Ontario, London.
The study entailed double-blind randomization of the 142 participants to subcutaneous injections of ixekizumab at 10, 25, 75, or 150 mg or placebo at 0, 2, 4, 8, 12, and 16 weeks. The standard primary outcome -- the proportion of patients with a PASI 75 improvement at 12 weeks -- has previously been reported as highly positive (N. Engl. J. Med. 2012;366:1190-9).
For her secondary analysis of psoriasis-related sexual difficulties and their response to treatment, Dr. Guenther used as her metric the patients’ response to item 9 on the Dermatology Life Quality Index, which all subjects completed at weeks 0, 8, and 16. Item 9 asks the extent to which the responder’s skin “caused any sexual difficulties” during the past week. The options range from 0 (none at all) to 3 (very much). She categorized a response of 1 or more as evidence of sexual difficulties. And because of the relatively small sample size in this study, she lumped together as the low-dose therapy group those patients assigned to ixekizumab at 10 or 25 mg, with the high-dose group being comprised of patients on 75 or
After 8 weeks on low-dose ixekizumab, the proportion of patients reporting any skin disease-related sexual difficulties within the past week fell from a baseline of 30% to 16%. After 8 weeks of high-dose ixekizumab, the figure was just 7%. Those rates remained unchanged at 16 weeks. In contrast, the placebo group remained unchanged over time, with 32% of patients still reporting sexual difficulties caused by their skin disease at week 8.
The degree to which a patient’s psoriasis improved in response to therapy with the humanized monoclonal antibody was closely related to the reduction in skin-related sexual problems. Among the 76 ixekizumab-treated patients who achieved a PASI 75 response at week 16, the rate of self-reported sexual difficulties within the previous week was 7%. For those with less than a PASI 75 response, the rate was 24%.
An impressively high 38% percent of patients on high-dose ixekizumab achieved a PASI 100 response. Only 5% of them reported any skin-related sexual difficulties at week 8, as did 9% at week 16.
Dr. Guenther also looked at the data restricting the analysis to patients with more severe baseline sexual impairment as defined by a response of 2 or 3 on item 9 of the DLQI. Among patients on high-dose izekizumab, the rate dropped from 10.5% at baseline to 1.8% at week 8 to zero at week 16. For patients on low-dose izekizumab, the progression was 13.8% to 8.8% to 3.5% at week 16. Rates remained unchanged over time in the control group.
Although this phase 2 study was limited in size, Dr. Guenther found much the same thing earlier in the much larger phase 3 PHOENIX 1 and 2 trials of ustekinumab (Stelara), which together featured 1,334 psoriasis patients randomized to the human anti-interkelukin-12/23 monoclonal antibody. In that analysis, the proportion of ustekinumab-treated patients with impaired sexual function as assessed by DLQI item 9 plunged from 22.4% at baseline to 2.7% at week 12, compared with no change in placebo-treated controls. The bigger the PASI improvement, the greater the reduction in psoriasis-related sexual dysfunction (J. Eur. Acad. Dermatol. Venereol. 2011;25:851-7).
The phase 2 ixekizumab study was funded by Eli Lilly. Dr. Guenther is a consultant to the company. Positive primary outcomes in three pivotal phase 3 clinical trials of ixekizumab totalling nearly 3,900 randomized psoriasis patients have since been reported. The company plans to apply for marketing approval of the biologic in the first half of 2015.
AMSTERDAM – Patient complaints of psoriasis-related sexual difficulties decreased significantly in response to treatment with the investigational biologic agent ixekizumab in a phase 2 dose-ranging study.
Sexual problems attributed by psoriasis patients to their skin condition are a common, underappreciated, and understudied problem. Most physicians simply don’t ask. But when they do, as was done formally in this study, it turned out that at baseline roughly one-third of the 142 participating subjects with moderate to severe chronic plaque psoriasis reported some degree of sexual problems they believed due specifically to their skin disease, Dr. Lyn Guenther reported at the annual congress of the European Academy of Dermatology and Venereology.
That rate dropped precipitously and in dose-dependent fashion in response to subcutaneous therapy with ixekizumab, a super-potent psoriasis medication directed against interleukin 17A, according to Dr. Guenther, professor of dermatology at the University of Western Ontario, London.
The study entailed double-blind randomization of the 142 participants to subcutaneous injections of ixekizumab at 10, 25, 75, or 150 mg or placebo at 0, 2, 4, 8, 12, and 16 weeks. The standard primary outcome -- the proportion of patients with a PASI 75 improvement at 12 weeks -- has previously been reported as highly positive (N. Engl. J. Med. 2012;366:1190-9).
For her secondary analysis of psoriasis-related sexual difficulties and their response to treatment, Dr. Guenther used as her metric the patients’ response to item 9 on the Dermatology Life Quality Index, which all subjects completed at weeks 0, 8, and 16. Item 9 asks the extent to which the responder’s skin “caused any sexual difficulties” during the past week. The options range from 0 (none at all) to 3 (very much). She categorized a response of 1 or more as evidence of sexual difficulties. And because of the relatively small sample size in this study, she lumped together as the low-dose therapy group those patients assigned to ixekizumab at 10 or 25 mg, with the high-dose group being comprised of patients on 75 or
After 8 weeks on low-dose ixekizumab, the proportion of patients reporting any skin disease-related sexual difficulties within the past week fell from a baseline of 30% to 16%. After 8 weeks of high-dose ixekizumab, the figure was just 7%. Those rates remained unchanged at 16 weeks. In contrast, the placebo group remained unchanged over time, with 32% of patients still reporting sexual difficulties caused by their skin disease at week 8.
The degree to which a patient’s psoriasis improved in response to therapy with the humanized monoclonal antibody was closely related to the reduction in skin-related sexual problems. Among the 76 ixekizumab-treated patients who achieved a PASI 75 response at week 16, the rate of self-reported sexual difficulties within the previous week was 7%. For those with less than a PASI 75 response, the rate was 24%.
An impressively high 38% percent of patients on high-dose ixekizumab achieved a PASI 100 response. Only 5% of them reported any skin-related sexual difficulties at week 8, as did 9% at week 16.
Dr. Guenther also looked at the data restricting the analysis to patients with more severe baseline sexual impairment as defined by a response of 2 or 3 on item 9 of the DLQI. Among patients on high-dose izekizumab, the rate dropped from 10.5% at baseline to 1.8% at week 8 to zero at week 16. For patients on low-dose izekizumab, the progression was 13.8% to 8.8% to 3.5% at week 16. Rates remained unchanged over time in the control group.
Although this phase 2 study was limited in size, Dr. Guenther found much the same thing earlier in the much larger phase 3 PHOENIX 1 and 2 trials of ustekinumab (Stelara), which together featured 1,334 psoriasis patients randomized to the human anti-interkelukin-12/23 monoclonal antibody. In that analysis, the proportion of ustekinumab-treated patients with impaired sexual function as assessed by DLQI item 9 plunged from 22.4% at baseline to 2.7% at week 12, compared with no change in placebo-treated controls. The bigger the PASI improvement, the greater the reduction in psoriasis-related sexual dysfunction (J. Eur. Acad. Dermatol. Venereol. 2011;25:851-7).
The phase 2 ixekizumab study was funded by Eli Lilly. Dr. Guenther is a consultant to the company. Positive primary outcomes in three pivotal phase 3 clinical trials of ixekizumab totalling nearly 3,900 randomized psoriasis patients have since been reported. The company plans to apply for marketing approval of the biologic in the first half of 2015.
AT THE EADV CONGRESS
Key clinical point: Psoriasis patients have a high rate of sexual problems they attribute to their skin disease, and which decrease with effective psoriasis therapy.
Major finding: The prevalence of self-reported psoriasis-related sexual difficulties within the previous week fell from 32% at baseline to 7% among patients with a PASI 75 response to ixekizumab at week 16.
Data source: This was a double-blind, phase 2, dose-ranging study involving 142 patients with moderate to severe chronic plaque psoriasis randomized to 16 weeks of ixekizumab or placebo.
Disclosures: The study was funded by Eli Lilly. The presenter is a consultant to the company.
Consider phototherapy for certain psoriasis patients
LAS VEGAS – Psoriasis patients who are unresponsive to topical therapy, do not have psoriatic arthritis, and can make regular office visits may be candidates for successful treatment with phototherapy.
A patient with type II skin and extensive plaque psoriasis could be treated with a targeted NB-UVB laser, Dr. Kenneth B. Gordon said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
However, it’s important to evaluate each patient individually on phototherapy decisions, rather than relying on published protocols, noted Dr. Gordon of Northwestern University Feinberg School of Medicine in Chicago.
The advantages of the NB-UVB laser for psoriasis include a high rate of response (data show more than 75% of patients achieve PASI 75), the ability to avoid treating uninvolved skin, and potential long-term clearing of lesions, he said.
For someone with extensive plaque psoriasis, Dr. Gordon said he would start with 3 treatments per week, at 200 mJ, and increase the fluence by 25 mJ if necessary. “A good response can be expected in 4-6 weeks,” he said.
As for long-term treatment, “I leave it up to the patient but offer long-term maintenance,” said Dr. Gordon. Long-term efficacy of NB-UVB for plaque psoriasis has not been well studied, and safety data are unknown. “It is unclear whether there is a skin cancer risk,” he said. Reducing the number of exposures for a patient on maintenance therapy may be an option, he added.
Short-term treatment with NB-UVB can be effective in some psoriasis patients, such as in cases of an acute flare of guttate psoriasis, Dr. Gordon noted. Psoralen and UV light therapy (PUVA) is another option, he said.
Phototherapy also has a role in managing psoriasis in conjunction with retinoids, Dr. Gordon said. He cited an example of a 59-year-old woman with palmar psoriasis. For this patient, he said he would recommend starting with acitretin for approximately 1 month, if the patient tolerates it. “If the acitretin works by itself, no need to add phototherapy,” he said. However, if UVB is added, account for the photosensitizing agent, he emphasized. “Decrease the starting dose by 25-50 mJ or decrease the skin type by one,” he said.
Some psoriasis patients will not benefit from phototherapy, particularly those with erythroderma, said Dr. Gordon. “If skin is highly inflamed, phototherapy could induce easy burning, Koebnerization,” or other complications, he noted.
Dr. Gordon disclosed that his department at Northwestern derives income from phototherapy, but he personally does not. He also disclosed receiving research grants from and/or serving as a consultant to multiple companies including AbbVie, Amgen, Celgene, Eli Lilly, and Janssen.
LAS VEGAS – Psoriasis patients who are unresponsive to topical therapy, do not have psoriatic arthritis, and can make regular office visits may be candidates for successful treatment with phototherapy.
A patient with type II skin and extensive plaque psoriasis could be treated with a targeted NB-UVB laser, Dr. Kenneth B. Gordon said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
However, it’s important to evaluate each patient individually on phototherapy decisions, rather than relying on published protocols, noted Dr. Gordon of Northwestern University Feinberg School of Medicine in Chicago.
The advantages of the NB-UVB laser for psoriasis include a high rate of response (data show more than 75% of patients achieve PASI 75), the ability to avoid treating uninvolved skin, and potential long-term clearing of lesions, he said.
For someone with extensive plaque psoriasis, Dr. Gordon said he would start with 3 treatments per week, at 200 mJ, and increase the fluence by 25 mJ if necessary. “A good response can be expected in 4-6 weeks,” he said.
As for long-term treatment, “I leave it up to the patient but offer long-term maintenance,” said Dr. Gordon. Long-term efficacy of NB-UVB for plaque psoriasis has not been well studied, and safety data are unknown. “It is unclear whether there is a skin cancer risk,” he said. Reducing the number of exposures for a patient on maintenance therapy may be an option, he added.
Short-term treatment with NB-UVB can be effective in some psoriasis patients, such as in cases of an acute flare of guttate psoriasis, Dr. Gordon noted. Psoralen and UV light therapy (PUVA) is another option, he said.
Phototherapy also has a role in managing psoriasis in conjunction with retinoids, Dr. Gordon said. He cited an example of a 59-year-old woman with palmar psoriasis. For this patient, he said he would recommend starting with acitretin for approximately 1 month, if the patient tolerates it. “If the acitretin works by itself, no need to add phototherapy,” he said. However, if UVB is added, account for the photosensitizing agent, he emphasized. “Decrease the starting dose by 25-50 mJ or decrease the skin type by one,” he said.
Some psoriasis patients will not benefit from phototherapy, particularly those with erythroderma, said Dr. Gordon. “If skin is highly inflamed, phototherapy could induce easy burning, Koebnerization,” or other complications, he noted.
Dr. Gordon disclosed that his department at Northwestern derives income from phototherapy, but he personally does not. He also disclosed receiving research grants from and/or serving as a consultant to multiple companies including AbbVie, Amgen, Celgene, Eli Lilly, and Janssen.
LAS VEGAS – Psoriasis patients who are unresponsive to topical therapy, do not have psoriatic arthritis, and can make regular office visits may be candidates for successful treatment with phototherapy.
A patient with type II skin and extensive plaque psoriasis could be treated with a targeted NB-UVB laser, Dr. Kenneth B. Gordon said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
However, it’s important to evaluate each patient individually on phototherapy decisions, rather than relying on published protocols, noted Dr. Gordon of Northwestern University Feinberg School of Medicine in Chicago.
The advantages of the NB-UVB laser for psoriasis include a high rate of response (data show more than 75% of patients achieve PASI 75), the ability to avoid treating uninvolved skin, and potential long-term clearing of lesions, he said.
For someone with extensive plaque psoriasis, Dr. Gordon said he would start with 3 treatments per week, at 200 mJ, and increase the fluence by 25 mJ if necessary. “A good response can be expected in 4-6 weeks,” he said.
As for long-term treatment, “I leave it up to the patient but offer long-term maintenance,” said Dr. Gordon. Long-term efficacy of NB-UVB for plaque psoriasis has not been well studied, and safety data are unknown. “It is unclear whether there is a skin cancer risk,” he said. Reducing the number of exposures for a patient on maintenance therapy may be an option, he added.
Short-term treatment with NB-UVB can be effective in some psoriasis patients, such as in cases of an acute flare of guttate psoriasis, Dr. Gordon noted. Psoralen and UV light therapy (PUVA) is another option, he said.
Phototherapy also has a role in managing psoriasis in conjunction with retinoids, Dr. Gordon said. He cited an example of a 59-year-old woman with palmar psoriasis. For this patient, he said he would recommend starting with acitretin for approximately 1 month, if the patient tolerates it. “If the acitretin works by itself, no need to add phototherapy,” he said. However, if UVB is added, account for the photosensitizing agent, he emphasized. “Decrease the starting dose by 25-50 mJ or decrease the skin type by one,” he said.
Some psoriasis patients will not benefit from phototherapy, particularly those with erythroderma, said Dr. Gordon. “If skin is highly inflamed, phototherapy could induce easy burning, Koebnerization,” or other complications, he noted.
Dr. Gordon disclosed that his department at Northwestern derives income from phototherapy, but he personally does not. He also disclosed receiving research grants from and/or serving as a consultant to multiple companies including AbbVie, Amgen, Celgene, Eli Lilly, and Janssen.
EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR