User login
MACE Update
Over the last several years, we have come to understand that patients with inflammatory diseases may be at higher risk for major adverse cardiovascular events (MACEs) and cardiovascular death. In an article published online on October 28 in the Annals of the Rheumatic Diseases, Ogdie et al reported on a study of the risk for MACEs among patients with psoriatic arthritis (PsA), rheumatoid arthritis (RA), and psoriasis without known PsA compared with the general population after adjusting for traditional cardiovascular risk factors.
The authors performed a population-based longitudinal cohort study (1994-2010) in The Health Improvement Network (THIN), a primary care medical record database in the United Kingdom. Patients aged 18 to 89 years with the applicable diagnoses were included. Up to 10 unexposed controls were selected for each patient with PsA, matched on practice and index date.
The study outcomes included the following: cardiovascular death, myocardial infarction, cerebrovascular accidents, and the composite outcome (MACE). Cox proportional hazards models were utilized to calculate the hazard ratios (HRs) for each of the outcomes after adjusting for traditional risk factors. The authors’ preliminary hypothesis was that there was an interaction between disease status and disease-modifying antirheumatic drug (DMARD) use.
In the analysis, the authors identified individuals with PsA (N=8706), RA (N=41,752), psoriasis (N=138,424), and unexposed controls (N=81,573). After adjusting for traditional risk factors, the risk for MACE was higher in the following populations: patients with PsA not prescribed a DMARD (HR, 1.24; 95% confidence interval [CI], 1.03-1.49), patients with RA (no DMARD: HR, 1.39; 95% CI, 1.28-1.50; DMARD: HR, 1.58; 95% CI, 1.46-1.70), patients with psoriasis not prescribed a DMARD (HR, 1.08; 95% CI, 1.02-1.15), and patients with severe psoriasis (DMARD: HR, 1.42; 95% CI, 1.17-1.73).
The authors concluded that cardiovascular risk should be addressed with all patients affected by psoriasis, PsA, or RA. These results suggest the need for improved screening and management of traditional cardiovascular risk factors in patients with inflammatory diseases.
What’s the issue?
Although there has been literature on MACE in these diseases, existing studies have not examined the risk for incident MACE including myocardial infarction, cerebrovascular accidents, and cardiovascular death in PsA compared with matched internal controls from a population-based perspective after accounting for the presence of traditional cardiovascular risk factors. This study is an additional piece of evidence that supports the need for comprehensive management of these patients. How do you currently address the concern for MACE?
Over the last several years, we have come to understand that patients with inflammatory diseases may be at higher risk for major adverse cardiovascular events (MACEs) and cardiovascular death. In an article published online on October 28 in the Annals of the Rheumatic Diseases, Ogdie et al reported on a study of the risk for MACEs among patients with psoriatic arthritis (PsA), rheumatoid arthritis (RA), and psoriasis without known PsA compared with the general population after adjusting for traditional cardiovascular risk factors.
The authors performed a population-based longitudinal cohort study (1994-2010) in The Health Improvement Network (THIN), a primary care medical record database in the United Kingdom. Patients aged 18 to 89 years with the applicable diagnoses were included. Up to 10 unexposed controls were selected for each patient with PsA, matched on practice and index date.
The study outcomes included the following: cardiovascular death, myocardial infarction, cerebrovascular accidents, and the composite outcome (MACE). Cox proportional hazards models were utilized to calculate the hazard ratios (HRs) for each of the outcomes after adjusting for traditional risk factors. The authors’ preliminary hypothesis was that there was an interaction between disease status and disease-modifying antirheumatic drug (DMARD) use.
In the analysis, the authors identified individuals with PsA (N=8706), RA (N=41,752), psoriasis (N=138,424), and unexposed controls (N=81,573). After adjusting for traditional risk factors, the risk for MACE was higher in the following populations: patients with PsA not prescribed a DMARD (HR, 1.24; 95% confidence interval [CI], 1.03-1.49), patients with RA (no DMARD: HR, 1.39; 95% CI, 1.28-1.50; DMARD: HR, 1.58; 95% CI, 1.46-1.70), patients with psoriasis not prescribed a DMARD (HR, 1.08; 95% CI, 1.02-1.15), and patients with severe psoriasis (DMARD: HR, 1.42; 95% CI, 1.17-1.73).
The authors concluded that cardiovascular risk should be addressed with all patients affected by psoriasis, PsA, or RA. These results suggest the need for improved screening and management of traditional cardiovascular risk factors in patients with inflammatory diseases.
What’s the issue?
Although there has been literature on MACE in these diseases, existing studies have not examined the risk for incident MACE including myocardial infarction, cerebrovascular accidents, and cardiovascular death in PsA compared with matched internal controls from a population-based perspective after accounting for the presence of traditional cardiovascular risk factors. This study is an additional piece of evidence that supports the need for comprehensive management of these patients. How do you currently address the concern for MACE?
Over the last several years, we have come to understand that patients with inflammatory diseases may be at higher risk for major adverse cardiovascular events (MACEs) and cardiovascular death. In an article published online on October 28 in the Annals of the Rheumatic Diseases, Ogdie et al reported on a study of the risk for MACEs among patients with psoriatic arthritis (PsA), rheumatoid arthritis (RA), and psoriasis without known PsA compared with the general population after adjusting for traditional cardiovascular risk factors.
The authors performed a population-based longitudinal cohort study (1994-2010) in The Health Improvement Network (THIN), a primary care medical record database in the United Kingdom. Patients aged 18 to 89 years with the applicable diagnoses were included. Up to 10 unexposed controls were selected for each patient with PsA, matched on practice and index date.
The study outcomes included the following: cardiovascular death, myocardial infarction, cerebrovascular accidents, and the composite outcome (MACE). Cox proportional hazards models were utilized to calculate the hazard ratios (HRs) for each of the outcomes after adjusting for traditional risk factors. The authors’ preliminary hypothesis was that there was an interaction between disease status and disease-modifying antirheumatic drug (DMARD) use.
In the analysis, the authors identified individuals with PsA (N=8706), RA (N=41,752), psoriasis (N=138,424), and unexposed controls (N=81,573). After adjusting for traditional risk factors, the risk for MACE was higher in the following populations: patients with PsA not prescribed a DMARD (HR, 1.24; 95% confidence interval [CI], 1.03-1.49), patients with RA (no DMARD: HR, 1.39; 95% CI, 1.28-1.50; DMARD: HR, 1.58; 95% CI, 1.46-1.70), patients with psoriasis not prescribed a DMARD (HR, 1.08; 95% CI, 1.02-1.15), and patients with severe psoriasis (DMARD: HR, 1.42; 95% CI, 1.17-1.73).
The authors concluded that cardiovascular risk should be addressed with all patients affected by psoriasis, PsA, or RA. These results suggest the need for improved screening and management of traditional cardiovascular risk factors in patients with inflammatory diseases.
What’s the issue?
Although there has been literature on MACE in these diseases, existing studies have not examined the risk for incident MACE including myocardial infarction, cerebrovascular accidents, and cardiovascular death in PsA compared with matched internal controls from a population-based perspective after accounting for the presence of traditional cardiovascular risk factors. This study is an additional piece of evidence that supports the need for comprehensive management of these patients. How do you currently address the concern for MACE?
Most Common Dermatologic Conditions Encountered by Dermatologists and Nondermatologists
Skin diseases are highly prevalent in the United States, affecting an estimated 1 in 3 Americans at any given time.1,2 In 2009 the direct medical costs associated with skin-related diseases, including health services and prescriptions, was approximately $22 billion; the annual total economic burden was estimated to be closer to $96 billion when factoring in the cost of lost productivity and pay for symptom relief.3,4 Effective and efficient management of skin disease is essential to minimizing cost and morbidity. Nondermatologists traditionally have diagnosed the majority of skin diseases.5,6 In particular, primary care physicians commonly manage dermatologic conditions and often are the first health care providers to encounter patients presenting with skin problems. A predicted shortage of dermatologists will likely contribute to an increase in this trend.7,8 Therefore, it is important to adequately prepare nondermatologists to evaluate and treat the skin conditions that they are most likely to encounter in their scope of practice.
Residents, particularly in primary care specialties, often have opportunities to spend 2 to 4 weeks with a dermatologist to learn about skin diseases; however, the skin conditions most often encountered by dermatologists may differ from those most often encountered by physicians in other specialties. For instance, one study demonstrated a disparity between the most common skin problems seen by dermatologists and internists.9 These dissimilarities should be recognized and addressed in curriculum content. The purpose of this study was to identify and compare the 20 most common dermatologic conditions reported by dermatologists versus those reported by nondermatologists (ie, internists, pediatricians, family physicians, emergency medicine physicians, general surgeons, otolaryngologists) from 2001 to 2010. Data also were analyzed to determine the top 20 conditions referred to dermatologists by nondermatologists as a potential indicator for areas of further improvement within medical education. With this knowledge, we hope educational curricula and self-study can be modified to reflect the current epidemiology of cutaneous diseases, thereby improving patient care.
Methods
Data from 2001 to 2010 were extracted from the National Ambulatory Medical Care Survey (NAMCS), which is an ongoing survey conducted by the National Center for Health Statistics. The NAMCS collects descriptive data regarding ambulatory visits to nonfederal office-based physicians in the United States. Participating physicians are instructed to record information about patient visits for a 1-week period, including patient demographics, insurance status, reason for visit, diagnoses, procedures, therapeutics, and referrals made at that time. Data collected for the NAMCS are entered into a multistage probability sample to produce national estimates. Within dermatology, an average of 118 dermatologists are sampled each year, and over the last 10 years, participation rates have ranged from 47% to 77%.
International Classification of Diseases, Ninth Revision, Clinical Modification codes were identified to determine the diagnoses that could be classified as dermatologic conditions. Select infectious and neoplastic disorders of the skin and mucous membrane conditions were included as well as the codes for skin diseases. Nondermatologic diagnoses and V codes were not included in the study. Data for all providers were studied to identify outpatient visits associated with the primary diagnosis of a dermatologic condition. Minor diagnoses that were considered to be subsets of major diagnoses were combined to allow better analysis of the data. For example, all tinea infections (ie, dermatophytosis of various sites, dermatomycosis unspecified) were combined into 1 diagnosis referred to as tinea because the recognition and treatment of this disease does not vary tremendously by anatomic location. Visits to dermatologists that listed nonspecific diagnoses and codes (eg, other postsurgical status [V45.89], neoplasm of uncertain behavior site unspecified [238.9]) were assumed to be for dermatologic problems.
Sampling weights were applied to obtain estimates for the number of each diagnosis made nationally. All data analyses were performed using SAS software and linear regression models were generated using SAS PROC SURVEYREG.
Data were analyzed to determine the dermatologic conditions most commonly encountered by dermatologists and nondermatologists in emergency medicine, family medicine, general surgery, internal medicine, otolaryngology, and pediatrics; these specialties include physicians who are known to commonly diagnose and treat skin diseases.10 Data also were analyzed to determine the most common conditions referred to dermatologists for treatment by nondermatologists from the selected specialties. Permission to conduct this study was obtained from the Wake Forest University institutional review board (Winston-Salem, North Carolina).
Results
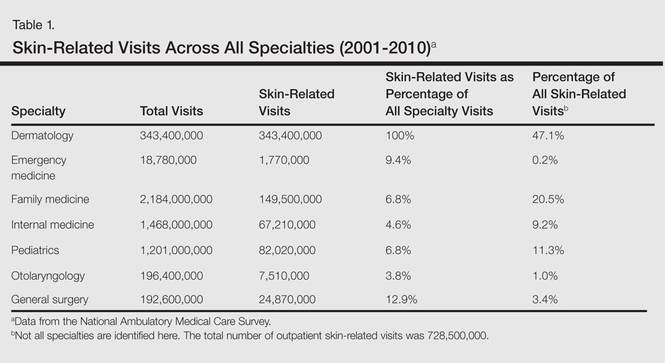
From 2001 to 2010, more than 700 million outpatient visits for skin-related problems were identified, with 676.3 million visits to dermatologists, emergency medicine physicians, family practitioners, general surgeons, internists, otolaryngologists, and pediatricians. More than half (52.9%) of all skin-related visits were addressed by nondermatologists during this time. Among nondermatologists, family practitioners encountered the greatest number of skin diseases (20.5%), followed by pediatricians (11.3%), internists (9.2%), general surgeons (3.4%), otolaryngologists (1.0%), and emergency medicine physicians (0.2%)(Table 1).
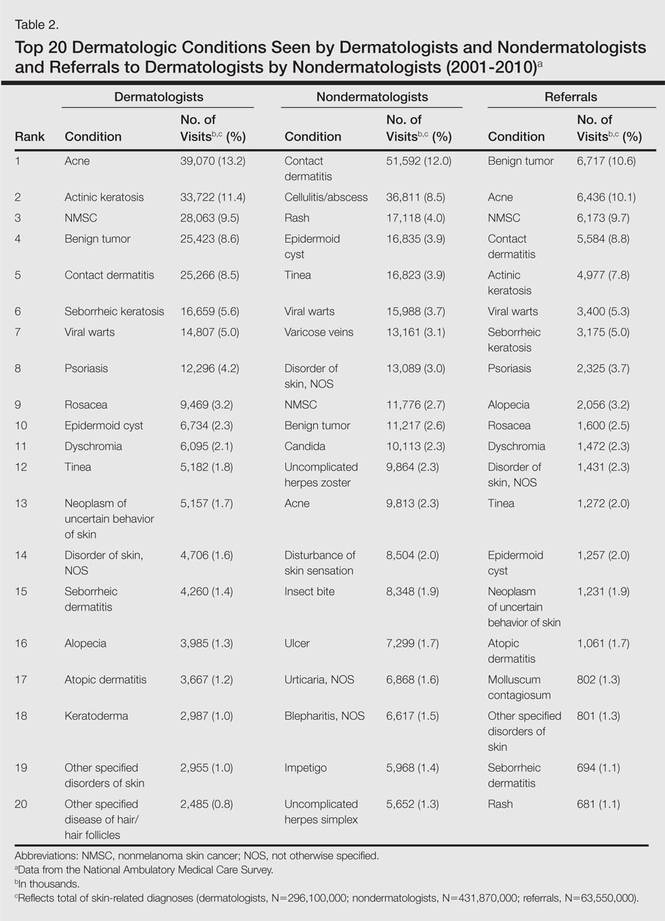
Benign tumors and acne were the most common cutaneous conditions referred to dermatologists by nondermatologists (10.6% and 10.1% of all dermatology referrals, respectively), followed by nonmelanoma skin cancers (9.7%), contact dermatitis (8.8%), and actinic keratosis (7.8%)(Table 2). The top 20 conditions referred to dermatologists accounted for 83.7% of all outpatient referrals to dermatologists.
Among the diseases most frequently reported by nondermatologists, contact dermatitis was the most common (12.0%), with twice the number of visits to nondermatologists for contact dermatitis than to dermatologists (51.6 million vs 25.3 million). In terms of disease categories, infectious skin diseases (ie, bacterial [cellulitis/abscess], viral [warts, herpesvirus], fungal [tinea] and yeast [candida] etiologies) were the most common dermatologic conditions reported by nondermatologists (Table 2).
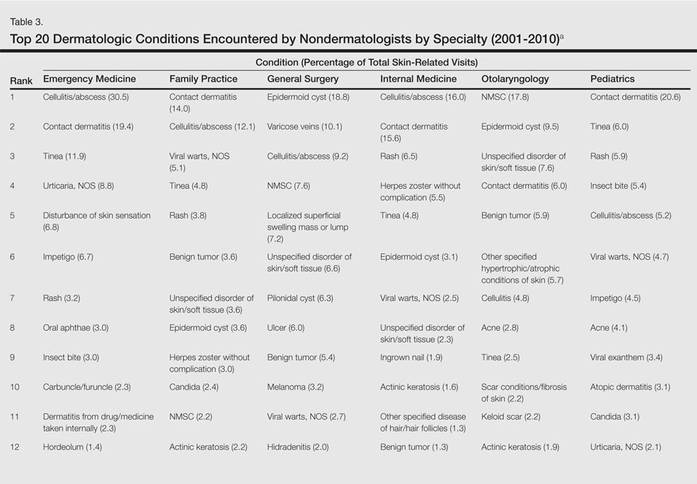
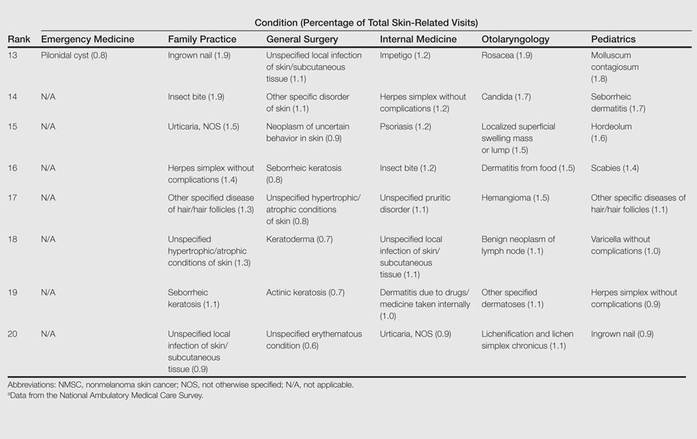
The top 20 dermatologic conditions reported by dermatologists accounted for 85.4% of all diagnoses made by dermatologists. Diseases that were among the top 20 conditions encountered by dermatologists but were not among the top 20 for nondermatologists included actinic keratosis, seborrheic keratosis, atopic dermatitis, psoriasis, alopecia, rosacea, dyschromia, seborrheic dermatitis, follicular disease, and neoplasm of uncertain behavior of skin. Additionally, 5 of the top 20 conditions encountered by dermatologists also were among the top 20 for only 1 individual nondermatologic specialty; these included atopic dermatitis (pediatrics), seborrheic dermatitis (pediatrics), psoriasis (internal medicine), rosacea (otolaryngology), and keratoderma (general surgery). Seborrheic dermatitis, psoriasis, and rosacea also were among the top 20 conditions most commonly referred to dermatologists for treatment by nondermatologists. Table 3 shows the top 20 dermatologic conditions encountered by nondermatologists by comparison.
Comment
According to NAMCS data from 2001 to 2010, visits to nondermatologists accounted for more than half of total outpatient visits for cutaneous diseases in the United States, whereas visits to dermatologists accounted for 47.1%. These findings are consistent with historical data indicating that 30% to 40% of skin-related visits are to dermatologists, and the majority of patients with skin disease are diagnosed by nondermatologists.5,6
Past data indicate that most visits to dermatologists were for evaluation of acne, infections, psoriasis, and neoplasms, whereas most visits to nondermatologists were for evaluation of epidermoid cysts, impetigo, plant dermatitis, cellulitis, and diaper rash.9 Over the last 10 years, acne has been more commonly encountered by nondermatologists, especially pediatricians. Additionally, infectious etiologies have been seen in larger volume by nondermatologists.9 Together, infectious cutaneous conditions make up nearly one-fourth of dermatologic encounters by emergency medicine physicians, internists, and family practitioners but are not within the top 20 diagnoses referred to dermatologists, which suggests that uncomplicated cases of cellulitis, herpes zoster, and other skin-related infections are largely managed by nondermatologists.5,6 Contact dermatitis, often caused by specific allergens such as detergents, solvents, and topical products, was one of the most common reported dermatologic encounters among dermatologists and nondermatologists and also was the fourth most common condition referred to dermatologists by nondermatologists for treatment; however, there may be an element of overuse of the International Classification of Diseases, Ninth Revision code, as any presumed contact dermatitis of unspecified cause can be reported under 692.9 defined as contact dermatitis and other eczema, unspecified cause. The high rate of referrals to dermatologists by nondermatologists may be for patch testing and further management. Additionally, there are no specific codes for allergic or irritant dermatitis, thus these diseases may be lumped together.
Although nearly half of all dermatologic encounters were seen by nondermatologists, dermatologists see a much larger proportion of patients with skin disease than nondermatologists and nondermatologists often have limited exposure to the field of dermatology during residency training. Studies have demonstrated differences in the abilities of dermatologists and nondermatologists to correctly diagnose common cutaneous diseases, which unsurprisingly revealed greater diagnostic accuracy demonstrated by dermatologists.11-16 The increase in acne and skin-related infections reported by nondermatologists is consistent with possible efforts to increase formal training in frequently encountered skin diseases. In one study evaluating the impact of a formal 3-week dermatology curriculum on an internal medicine department, internists demonstrated 100% accuracy in the diagnosis of acne and herpes zoster in contrast to 29% for tinea and 12% for lichen planus.5,6
The current Accreditation Council for Graduate Medical Education guidelines place little emphasis on exposure to dermatology training during residency for internists and pediatricians, as this training is not a required component of these programs.17 Two core problems with current training regarding the evaluation and management of cutaneous disease are minimal exposure to dermatologic conditions in medical school and residency and lack of consensus on the core topics that should be taught to nondermatologists.18 Exposure to dermatologic conditions through rotations in medical school has been shown to increase residents’ self-reported confidence in diagnosing and treating alopecia, cutaneous drug eruptions, warts, acne, rosacea, nonmelanoma skin cancers, sun damage, psoriasis, seborrhea, atopic dermatitis, and contact dermatitis; however, the majority of primary care residents surveyed still felt that this exposure in medical school was inadequate.19
In creating a core curriculum for dermatology training for nondermatologists, it is important to consider the dermatologic conditions that are most frequently encountered by these specialties. Our study revealed that the most commonly encountered dermatologic conditions differ among dermatologists and nondermatologists, with a fair degree of variation even among individual specialties. Failure to recognize these discrepancies has likely contributed to the challenges faced by nondermatologists in the diagnosis and management of dermatologic disease. In this study, contact dermatitis, epidermoid cysts, and skin infections were the most common dermatologic conditions encountered by nondermatologists and also were among the top skin diseases referred to dermatologists by nondermatologists. This finding suggests that nondermatologists are able to identify these conditions but have a tendency to refer approximately 10% of these patients to dermatology for further management. Clinical evaluation and medical management of these cutaneous diseases may be an important area of focus for medical school curricula, as the treatment of these diseases is within the capabilities of the nondermatologist. For example, initial management of dermatitis requires determination of the type of dermatitis (ie, essential, contact, atopic, seborrheic, stasis) and selection of an appropriate topical steroid, with referral to a dermatologist needed for questionable or refractory cases. Although a curriculum cannot be built solely on a list of the top 20 diagnoses provided here, these data may serve as a preliminary platform for medical school dermatology curriculum design. The curriculum also should include serious skin diseases, such as melanoma and severe drug eruptions. Although these conditions are less commonly encountered by nondermatologists, missed diagnosis and/or improper management can be life threatening.
The use of NAMCS data presents a few limitations. For instance, these data only represent outpatient management of skin disease. There is the potential for misdiagnosis and coding errors by the reporting physicians. The volume of data (ie, billions of office visits) prevents verification of diagnostic accuracy. The coding system requires physicians to give a diagnosis but does not provide any means by which to determine the physician’s confidence in that diagnosis. There is no code for “uncertain” or “diagnosis not determined.” Additionally, an “unspecified” diagnosis may reflect uncertainty or may simply imply that no other code accurately described the condition. Despite these limitations, the NAMCS database is a large, nationally representative survey of actual patient visits and represents some of the best data available for a study such as ours.
Conclusion
This study provides an important analysis of the most common outpatient dermatologic conditions encountered by dermatologists and nondermatologists of various specialties and offers a foundation from which to construct curricula for dermatology training tailored to individual specialties based on their needs. In the future, identification of the most common inpatient dermatologic conditions managed by each specialty also may benefit curriculum design.
- Thorpe KE, Florence CS, Joski P. Which medical conditions account for the rise in health care spending? Health Aff (Millwood). 2004;(suppl web exclusives):W4-437-445.
- Johnson ML. Defining the burden of skin disease in the United States—a historical perspective. J Investig Dermatol Symp Proc. 2004;9:108-110.
- Agency for Healthcare Research and Quality. Medical expenditure panel survey. US Department of Health & Human Services Web site. http://meps.ahrq.gov. Accessed November 17, 2014.
- Bickers DR, Lim HW, Margolis D, et al. The burden of skin diseases: 2004 a joint project of the American Academy of Dermatology Association and the Society for Investigative Dermatology. J Am Acad Dermatol. 2006;55:490-500.
- Johnson ML. On teaching dermatology to nondermatologists. Arch Dermatol. 1994;130:850-852.
- Ramsay DL, Weary PE. Primary care in dermatology: whose role should it be? J Am Acad Dermatol. 1996;35:1005-1008.
- Kimball AB, Resneck JS Jr. The US dermatology workforce: a specialty remains in shortage. J Am Acad Dermatol. 2008;59:741-745.
- Resneck JS Jr, Kimball AB. Who else is providing care in dermatology practices? trends in the use of nonphysician clinicians. J Am Acad Dermatol. 2008;58:211-216.
- Feldman SR, Fleischer AB Jr, McConnell RC. Most common dermatologic problems identified by internists, 1990-1994. Arch Intern Med. 1998;158:726-730.
- Ahn CS, Davis SA, Debade TS, et al. Noncosmetic skin-related procedures performed in the United States: an analysis of national ambulatory medical care survey data from 1995 to 2010. Dermatol Surg. 2013;39:1912-1921.
- Antic M, Conen D, Itin PH. Teaching effects of dermatological consultations on nondermatologists in the field of internal medicine. a study of 1290 inpatients. Dermatology. 2004;208:32-37.
- Federman DG, Concato J, Kirsner RS. Comparison of dermatologic diagnoses by primary care practitioners and dermatologists. a review of the literature. Arch Fam Med. 1999;8:170-172.
- Fleischer AB Jr, Herbert CR, Feldman SR, et al. Diagnosis of skin disease by nondermatologists. Am J Manag Care. 2000;6:1149-1156.
- Kirsner RS, Federman DG. Lack of correlation between internists’ ability in dermatology and their patterns of treating patients with skin disease. Arch Dermatol. 1996;132:1043-1046.
- McCarthy GM, Lamb GC, Russell TJ, et al. Primary care-based dermatology practice: internists need more training. J Gen Intern Med. 1991;6:52-56.
- Sellheyer K, Bergfeld WF. A retrospective biopsy study of the clinical diagnostic accuracy of common skin diseases by different specialties compared with dermatology. J Am Acad Dermatol. 2005;52:823-830.
- Medical specialties. Accreditation Council for Graduate Medical Education Web site. http://www.acgme.org/acgmeweb/tabid/368ProgramandInstitutionalGuidelines/MedicalAccreditation.aspx. Accessed November 17, 2014.
- McCleskey PE, Gilson RT, DeVillez RL. Medical student core curriculum in dermatology survey. J Am Acad Dermatol. 2009;61:30-35.
- Hansra NK, O’Sullivan P, Chen CL, et al. Medical school dermatology curriculum: are we adequately preparing primary care physicians? J Am Acad Dermatol. 2009;61:23-29.
Skin diseases are highly prevalent in the United States, affecting an estimated 1 in 3 Americans at any given time.1,2 In 2009 the direct medical costs associated with skin-related diseases, including health services and prescriptions, was approximately $22 billion; the annual total economic burden was estimated to be closer to $96 billion when factoring in the cost of lost productivity and pay for symptom relief.3,4 Effective and efficient management of skin disease is essential to minimizing cost and morbidity. Nondermatologists traditionally have diagnosed the majority of skin diseases.5,6 In particular, primary care physicians commonly manage dermatologic conditions and often are the first health care providers to encounter patients presenting with skin problems. A predicted shortage of dermatologists will likely contribute to an increase in this trend.7,8 Therefore, it is important to adequately prepare nondermatologists to evaluate and treat the skin conditions that they are most likely to encounter in their scope of practice.
Residents, particularly in primary care specialties, often have opportunities to spend 2 to 4 weeks with a dermatologist to learn about skin diseases; however, the skin conditions most often encountered by dermatologists may differ from those most often encountered by physicians in other specialties. For instance, one study demonstrated a disparity between the most common skin problems seen by dermatologists and internists.9 These dissimilarities should be recognized and addressed in curriculum content. The purpose of this study was to identify and compare the 20 most common dermatologic conditions reported by dermatologists versus those reported by nondermatologists (ie, internists, pediatricians, family physicians, emergency medicine physicians, general surgeons, otolaryngologists) from 2001 to 2010. Data also were analyzed to determine the top 20 conditions referred to dermatologists by nondermatologists as a potential indicator for areas of further improvement within medical education. With this knowledge, we hope educational curricula and self-study can be modified to reflect the current epidemiology of cutaneous diseases, thereby improving patient care.
Methods
Data from 2001 to 2010 were extracted from the National Ambulatory Medical Care Survey (NAMCS), which is an ongoing survey conducted by the National Center for Health Statistics. The NAMCS collects descriptive data regarding ambulatory visits to nonfederal office-based physicians in the United States. Participating physicians are instructed to record information about patient visits for a 1-week period, including patient demographics, insurance status, reason for visit, diagnoses, procedures, therapeutics, and referrals made at that time. Data collected for the NAMCS are entered into a multistage probability sample to produce national estimates. Within dermatology, an average of 118 dermatologists are sampled each year, and over the last 10 years, participation rates have ranged from 47% to 77%.
International Classification of Diseases, Ninth Revision, Clinical Modification codes were identified to determine the diagnoses that could be classified as dermatologic conditions. Select infectious and neoplastic disorders of the skin and mucous membrane conditions were included as well as the codes for skin diseases. Nondermatologic diagnoses and V codes were not included in the study. Data for all providers were studied to identify outpatient visits associated with the primary diagnosis of a dermatologic condition. Minor diagnoses that were considered to be subsets of major diagnoses were combined to allow better analysis of the data. For example, all tinea infections (ie, dermatophytosis of various sites, dermatomycosis unspecified) were combined into 1 diagnosis referred to as tinea because the recognition and treatment of this disease does not vary tremendously by anatomic location. Visits to dermatologists that listed nonspecific diagnoses and codes (eg, other postsurgical status [V45.89], neoplasm of uncertain behavior site unspecified [238.9]) were assumed to be for dermatologic problems.
Sampling weights were applied to obtain estimates for the number of each diagnosis made nationally. All data analyses were performed using SAS software and linear regression models were generated using SAS PROC SURVEYREG.
Data were analyzed to determine the dermatologic conditions most commonly encountered by dermatologists and nondermatologists in emergency medicine, family medicine, general surgery, internal medicine, otolaryngology, and pediatrics; these specialties include physicians who are known to commonly diagnose and treat skin diseases.10 Data also were analyzed to determine the most common conditions referred to dermatologists for treatment by nondermatologists from the selected specialties. Permission to conduct this study was obtained from the Wake Forest University institutional review board (Winston-Salem, North Carolina).
Results
From 2001 to 2010, more than 700 million outpatient visits for skin-related problems were identified, with 676.3 million visits to dermatologists, emergency medicine physicians, family practitioners, general surgeons, internists, otolaryngologists, and pediatricians. More than half (52.9%) of all skin-related visits were addressed by nondermatologists during this time. Among nondermatologists, family practitioners encountered the greatest number of skin diseases (20.5%), followed by pediatricians (11.3%), internists (9.2%), general surgeons (3.4%), otolaryngologists (1.0%), and emergency medicine physicians (0.2%)(Table 1).

Benign tumors and acne were the most common cutaneous conditions referred to dermatologists by nondermatologists (10.6% and 10.1% of all dermatology referrals, respectively), followed by nonmelanoma skin cancers (9.7%), contact dermatitis (8.8%), and actinic keratosis (7.8%)(Table 2). The top 20 conditions referred to dermatologists accounted for 83.7% of all outpatient referrals to dermatologists.
Among the diseases most frequently reported by nondermatologists, contact dermatitis was the most common (12.0%), with twice the number of visits to nondermatologists for contact dermatitis than to dermatologists (51.6 million vs 25.3 million). In terms of disease categories, infectious skin diseases (ie, bacterial [cellulitis/abscess], viral [warts, herpesvirus], fungal [tinea] and yeast [candida] etiologies) were the most common dermatologic conditions reported by nondermatologists (Table 2).

The top 20 dermatologic conditions reported by dermatologists accounted for 85.4% of all diagnoses made by dermatologists. Diseases that were among the top 20 conditions encountered by dermatologists but were not among the top 20 for nondermatologists included actinic keratosis, seborrheic keratosis, atopic dermatitis, psoriasis, alopecia, rosacea, dyschromia, seborrheic dermatitis, follicular disease, and neoplasm of uncertain behavior of skin. Additionally, 5 of the top 20 conditions encountered by dermatologists also were among the top 20 for only 1 individual nondermatologic specialty; these included atopic dermatitis (pediatrics), seborrheic dermatitis (pediatrics), psoriasis (internal medicine), rosacea (otolaryngology), and keratoderma (general surgery). Seborrheic dermatitis, psoriasis, and rosacea also were among the top 20 conditions most commonly referred to dermatologists for treatment by nondermatologists. Table 3 shows the top 20 dermatologic conditions encountered by nondermatologists by comparison.


Comment
According to NAMCS data from 2001 to 2010, visits to nondermatologists accounted for more than half of total outpatient visits for cutaneous diseases in the United States, whereas visits to dermatologists accounted for 47.1%. These findings are consistent with historical data indicating that 30% to 40% of skin-related visits are to dermatologists, and the majority of patients with skin disease are diagnosed by nondermatologists.5,6
Past data indicate that most visits to dermatologists were for evaluation of acne, infections, psoriasis, and neoplasms, whereas most visits to nondermatologists were for evaluation of epidermoid cysts, impetigo, plant dermatitis, cellulitis, and diaper rash.9 Over the last 10 years, acne has been more commonly encountered by nondermatologists, especially pediatricians. Additionally, infectious etiologies have been seen in larger volume by nondermatologists.9 Together, infectious cutaneous conditions make up nearly one-fourth of dermatologic encounters by emergency medicine physicians, internists, and family practitioners but are not within the top 20 diagnoses referred to dermatologists, which suggests that uncomplicated cases of cellulitis, herpes zoster, and other skin-related infections are largely managed by nondermatologists.5,6 Contact dermatitis, often caused by specific allergens such as detergents, solvents, and topical products, was one of the most common reported dermatologic encounters among dermatologists and nondermatologists and also was the fourth most common condition referred to dermatologists by nondermatologists for treatment; however, there may be an element of overuse of the International Classification of Diseases, Ninth Revision code, as any presumed contact dermatitis of unspecified cause can be reported under 692.9 defined as contact dermatitis and other eczema, unspecified cause. The high rate of referrals to dermatologists by nondermatologists may be for patch testing and further management. Additionally, there are no specific codes for allergic or irritant dermatitis, thus these diseases may be lumped together.
Although nearly half of all dermatologic encounters were seen by nondermatologists, dermatologists see a much larger proportion of patients with skin disease than nondermatologists and nondermatologists often have limited exposure to the field of dermatology during residency training. Studies have demonstrated differences in the abilities of dermatologists and nondermatologists to correctly diagnose common cutaneous diseases, which unsurprisingly revealed greater diagnostic accuracy demonstrated by dermatologists.11-16 The increase in acne and skin-related infections reported by nondermatologists is consistent with possible efforts to increase formal training in frequently encountered skin diseases. In one study evaluating the impact of a formal 3-week dermatology curriculum on an internal medicine department, internists demonstrated 100% accuracy in the diagnosis of acne and herpes zoster in contrast to 29% for tinea and 12% for lichen planus.5,6
The current Accreditation Council for Graduate Medical Education guidelines place little emphasis on exposure to dermatology training during residency for internists and pediatricians, as this training is not a required component of these programs.17 Two core problems with current training regarding the evaluation and management of cutaneous disease are minimal exposure to dermatologic conditions in medical school and residency and lack of consensus on the core topics that should be taught to nondermatologists.18 Exposure to dermatologic conditions through rotations in medical school has been shown to increase residents’ self-reported confidence in diagnosing and treating alopecia, cutaneous drug eruptions, warts, acne, rosacea, nonmelanoma skin cancers, sun damage, psoriasis, seborrhea, atopic dermatitis, and contact dermatitis; however, the majority of primary care residents surveyed still felt that this exposure in medical school was inadequate.19
In creating a core curriculum for dermatology training for nondermatologists, it is important to consider the dermatologic conditions that are most frequently encountered by these specialties. Our study revealed that the most commonly encountered dermatologic conditions differ among dermatologists and nondermatologists, with a fair degree of variation even among individual specialties. Failure to recognize these discrepancies has likely contributed to the challenges faced by nondermatologists in the diagnosis and management of dermatologic disease. In this study, contact dermatitis, epidermoid cysts, and skin infections were the most common dermatologic conditions encountered by nondermatologists and also were among the top skin diseases referred to dermatologists by nondermatologists. This finding suggests that nondermatologists are able to identify these conditions but have a tendency to refer approximately 10% of these patients to dermatology for further management. Clinical evaluation and medical management of these cutaneous diseases may be an important area of focus for medical school curricula, as the treatment of these diseases is within the capabilities of the nondermatologist. For example, initial management of dermatitis requires determination of the type of dermatitis (ie, essential, contact, atopic, seborrheic, stasis) and selection of an appropriate topical steroid, with referral to a dermatologist needed for questionable or refractory cases. Although a curriculum cannot be built solely on a list of the top 20 diagnoses provided here, these data may serve as a preliminary platform for medical school dermatology curriculum design. The curriculum also should include serious skin diseases, such as melanoma and severe drug eruptions. Although these conditions are less commonly encountered by nondermatologists, missed diagnosis and/or improper management can be life threatening.
The use of NAMCS data presents a few limitations. For instance, these data only represent outpatient management of skin disease. There is the potential for misdiagnosis and coding errors by the reporting physicians. The volume of data (ie, billions of office visits) prevents verification of diagnostic accuracy. The coding system requires physicians to give a diagnosis but does not provide any means by which to determine the physician’s confidence in that diagnosis. There is no code for “uncertain” or “diagnosis not determined.” Additionally, an “unspecified” diagnosis may reflect uncertainty or may simply imply that no other code accurately described the condition. Despite these limitations, the NAMCS database is a large, nationally representative survey of actual patient visits and represents some of the best data available for a study such as ours.
Conclusion
This study provides an important analysis of the most common outpatient dermatologic conditions encountered by dermatologists and nondermatologists of various specialties and offers a foundation from which to construct curricula for dermatology training tailored to individual specialties based on their needs. In the future, identification of the most common inpatient dermatologic conditions managed by each specialty also may benefit curriculum design.
Skin diseases are highly prevalent in the United States, affecting an estimated 1 in 3 Americans at any given time.1,2 In 2009 the direct medical costs associated with skin-related diseases, including health services and prescriptions, was approximately $22 billion; the annual total economic burden was estimated to be closer to $96 billion when factoring in the cost of lost productivity and pay for symptom relief.3,4 Effective and efficient management of skin disease is essential to minimizing cost and morbidity. Nondermatologists traditionally have diagnosed the majority of skin diseases.5,6 In particular, primary care physicians commonly manage dermatologic conditions and often are the first health care providers to encounter patients presenting with skin problems. A predicted shortage of dermatologists will likely contribute to an increase in this trend.7,8 Therefore, it is important to adequately prepare nondermatologists to evaluate and treat the skin conditions that they are most likely to encounter in their scope of practice.
Residents, particularly in primary care specialties, often have opportunities to spend 2 to 4 weeks with a dermatologist to learn about skin diseases; however, the skin conditions most often encountered by dermatologists may differ from those most often encountered by physicians in other specialties. For instance, one study demonstrated a disparity between the most common skin problems seen by dermatologists and internists.9 These dissimilarities should be recognized and addressed in curriculum content. The purpose of this study was to identify and compare the 20 most common dermatologic conditions reported by dermatologists versus those reported by nondermatologists (ie, internists, pediatricians, family physicians, emergency medicine physicians, general surgeons, otolaryngologists) from 2001 to 2010. Data also were analyzed to determine the top 20 conditions referred to dermatologists by nondermatologists as a potential indicator for areas of further improvement within medical education. With this knowledge, we hope educational curricula and self-study can be modified to reflect the current epidemiology of cutaneous diseases, thereby improving patient care.
Methods
Data from 2001 to 2010 were extracted from the National Ambulatory Medical Care Survey (NAMCS), which is an ongoing survey conducted by the National Center for Health Statistics. The NAMCS collects descriptive data regarding ambulatory visits to nonfederal office-based physicians in the United States. Participating physicians are instructed to record information about patient visits for a 1-week period, including patient demographics, insurance status, reason for visit, diagnoses, procedures, therapeutics, and referrals made at that time. Data collected for the NAMCS are entered into a multistage probability sample to produce national estimates. Within dermatology, an average of 118 dermatologists are sampled each year, and over the last 10 years, participation rates have ranged from 47% to 77%.
International Classification of Diseases, Ninth Revision, Clinical Modification codes were identified to determine the diagnoses that could be classified as dermatologic conditions. Select infectious and neoplastic disorders of the skin and mucous membrane conditions were included as well as the codes for skin diseases. Nondermatologic diagnoses and V codes were not included in the study. Data for all providers were studied to identify outpatient visits associated with the primary diagnosis of a dermatologic condition. Minor diagnoses that were considered to be subsets of major diagnoses were combined to allow better analysis of the data. For example, all tinea infections (ie, dermatophytosis of various sites, dermatomycosis unspecified) were combined into 1 diagnosis referred to as tinea because the recognition and treatment of this disease does not vary tremendously by anatomic location. Visits to dermatologists that listed nonspecific diagnoses and codes (eg, other postsurgical status [V45.89], neoplasm of uncertain behavior site unspecified [238.9]) were assumed to be for dermatologic problems.
Sampling weights were applied to obtain estimates for the number of each diagnosis made nationally. All data analyses were performed using SAS software and linear regression models were generated using SAS PROC SURVEYREG.
Data were analyzed to determine the dermatologic conditions most commonly encountered by dermatologists and nondermatologists in emergency medicine, family medicine, general surgery, internal medicine, otolaryngology, and pediatrics; these specialties include physicians who are known to commonly diagnose and treat skin diseases.10 Data also were analyzed to determine the most common conditions referred to dermatologists for treatment by nondermatologists from the selected specialties. Permission to conduct this study was obtained from the Wake Forest University institutional review board (Winston-Salem, North Carolina).
Results
From 2001 to 2010, more than 700 million outpatient visits for skin-related problems were identified, with 676.3 million visits to dermatologists, emergency medicine physicians, family practitioners, general surgeons, internists, otolaryngologists, and pediatricians. More than half (52.9%) of all skin-related visits were addressed by nondermatologists during this time. Among nondermatologists, family practitioners encountered the greatest number of skin diseases (20.5%), followed by pediatricians (11.3%), internists (9.2%), general surgeons (3.4%), otolaryngologists (1.0%), and emergency medicine physicians (0.2%)(Table 1).

Benign tumors and acne were the most common cutaneous conditions referred to dermatologists by nondermatologists (10.6% and 10.1% of all dermatology referrals, respectively), followed by nonmelanoma skin cancers (9.7%), contact dermatitis (8.8%), and actinic keratosis (7.8%)(Table 2). The top 20 conditions referred to dermatologists accounted for 83.7% of all outpatient referrals to dermatologists.
Among the diseases most frequently reported by nondermatologists, contact dermatitis was the most common (12.0%), with twice the number of visits to nondermatologists for contact dermatitis than to dermatologists (51.6 million vs 25.3 million). In terms of disease categories, infectious skin diseases (ie, bacterial [cellulitis/abscess], viral [warts, herpesvirus], fungal [tinea] and yeast [candida] etiologies) were the most common dermatologic conditions reported by nondermatologists (Table 2).

The top 20 dermatologic conditions reported by dermatologists accounted for 85.4% of all diagnoses made by dermatologists. Diseases that were among the top 20 conditions encountered by dermatologists but were not among the top 20 for nondermatologists included actinic keratosis, seborrheic keratosis, atopic dermatitis, psoriasis, alopecia, rosacea, dyschromia, seborrheic dermatitis, follicular disease, and neoplasm of uncertain behavior of skin. Additionally, 5 of the top 20 conditions encountered by dermatologists also were among the top 20 for only 1 individual nondermatologic specialty; these included atopic dermatitis (pediatrics), seborrheic dermatitis (pediatrics), psoriasis (internal medicine), rosacea (otolaryngology), and keratoderma (general surgery). Seborrheic dermatitis, psoriasis, and rosacea also were among the top 20 conditions most commonly referred to dermatologists for treatment by nondermatologists. Table 3 shows the top 20 dermatologic conditions encountered by nondermatologists by comparison.


Comment
According to NAMCS data from 2001 to 2010, visits to nondermatologists accounted for more than half of total outpatient visits for cutaneous diseases in the United States, whereas visits to dermatologists accounted for 47.1%. These findings are consistent with historical data indicating that 30% to 40% of skin-related visits are to dermatologists, and the majority of patients with skin disease are diagnosed by nondermatologists.5,6
Past data indicate that most visits to dermatologists were for evaluation of acne, infections, psoriasis, and neoplasms, whereas most visits to nondermatologists were for evaluation of epidermoid cysts, impetigo, plant dermatitis, cellulitis, and diaper rash.9 Over the last 10 years, acne has been more commonly encountered by nondermatologists, especially pediatricians. Additionally, infectious etiologies have been seen in larger volume by nondermatologists.9 Together, infectious cutaneous conditions make up nearly one-fourth of dermatologic encounters by emergency medicine physicians, internists, and family practitioners but are not within the top 20 diagnoses referred to dermatologists, which suggests that uncomplicated cases of cellulitis, herpes zoster, and other skin-related infections are largely managed by nondermatologists.5,6 Contact dermatitis, often caused by specific allergens such as detergents, solvents, and topical products, was one of the most common reported dermatologic encounters among dermatologists and nondermatologists and also was the fourth most common condition referred to dermatologists by nondermatologists for treatment; however, there may be an element of overuse of the International Classification of Diseases, Ninth Revision code, as any presumed contact dermatitis of unspecified cause can be reported under 692.9 defined as contact dermatitis and other eczema, unspecified cause. The high rate of referrals to dermatologists by nondermatologists may be for patch testing and further management. Additionally, there are no specific codes for allergic or irritant dermatitis, thus these diseases may be lumped together.
Although nearly half of all dermatologic encounters were seen by nondermatologists, dermatologists see a much larger proportion of patients with skin disease than nondermatologists and nondermatologists often have limited exposure to the field of dermatology during residency training. Studies have demonstrated differences in the abilities of dermatologists and nondermatologists to correctly diagnose common cutaneous diseases, which unsurprisingly revealed greater diagnostic accuracy demonstrated by dermatologists.11-16 The increase in acne and skin-related infections reported by nondermatologists is consistent with possible efforts to increase formal training in frequently encountered skin diseases. In one study evaluating the impact of a formal 3-week dermatology curriculum on an internal medicine department, internists demonstrated 100% accuracy in the diagnosis of acne and herpes zoster in contrast to 29% for tinea and 12% for lichen planus.5,6
The current Accreditation Council for Graduate Medical Education guidelines place little emphasis on exposure to dermatology training during residency for internists and pediatricians, as this training is not a required component of these programs.17 Two core problems with current training regarding the evaluation and management of cutaneous disease are minimal exposure to dermatologic conditions in medical school and residency and lack of consensus on the core topics that should be taught to nondermatologists.18 Exposure to dermatologic conditions through rotations in medical school has been shown to increase residents’ self-reported confidence in diagnosing and treating alopecia, cutaneous drug eruptions, warts, acne, rosacea, nonmelanoma skin cancers, sun damage, psoriasis, seborrhea, atopic dermatitis, and contact dermatitis; however, the majority of primary care residents surveyed still felt that this exposure in medical school was inadequate.19
In creating a core curriculum for dermatology training for nondermatologists, it is important to consider the dermatologic conditions that are most frequently encountered by these specialties. Our study revealed that the most commonly encountered dermatologic conditions differ among dermatologists and nondermatologists, with a fair degree of variation even among individual specialties. Failure to recognize these discrepancies has likely contributed to the challenges faced by nondermatologists in the diagnosis and management of dermatologic disease. In this study, contact dermatitis, epidermoid cysts, and skin infections were the most common dermatologic conditions encountered by nondermatologists and also were among the top skin diseases referred to dermatologists by nondermatologists. This finding suggests that nondermatologists are able to identify these conditions but have a tendency to refer approximately 10% of these patients to dermatology for further management. Clinical evaluation and medical management of these cutaneous diseases may be an important area of focus for medical school curricula, as the treatment of these diseases is within the capabilities of the nondermatologist. For example, initial management of dermatitis requires determination of the type of dermatitis (ie, essential, contact, atopic, seborrheic, stasis) and selection of an appropriate topical steroid, with referral to a dermatologist needed for questionable or refractory cases. Although a curriculum cannot be built solely on a list of the top 20 diagnoses provided here, these data may serve as a preliminary platform for medical school dermatology curriculum design. The curriculum also should include serious skin diseases, such as melanoma and severe drug eruptions. Although these conditions are less commonly encountered by nondermatologists, missed diagnosis and/or improper management can be life threatening.
The use of NAMCS data presents a few limitations. For instance, these data only represent outpatient management of skin disease. There is the potential for misdiagnosis and coding errors by the reporting physicians. The volume of data (ie, billions of office visits) prevents verification of diagnostic accuracy. The coding system requires physicians to give a diagnosis but does not provide any means by which to determine the physician’s confidence in that diagnosis. There is no code for “uncertain” or “diagnosis not determined.” Additionally, an “unspecified” diagnosis may reflect uncertainty or may simply imply that no other code accurately described the condition. Despite these limitations, the NAMCS database is a large, nationally representative survey of actual patient visits and represents some of the best data available for a study such as ours.
Conclusion
This study provides an important analysis of the most common outpatient dermatologic conditions encountered by dermatologists and nondermatologists of various specialties and offers a foundation from which to construct curricula for dermatology training tailored to individual specialties based on their needs. In the future, identification of the most common inpatient dermatologic conditions managed by each specialty also may benefit curriculum design.
- Thorpe KE, Florence CS, Joski P. Which medical conditions account for the rise in health care spending? Health Aff (Millwood). 2004;(suppl web exclusives):W4-437-445.
- Johnson ML. Defining the burden of skin disease in the United States—a historical perspective. J Investig Dermatol Symp Proc. 2004;9:108-110.
- Agency for Healthcare Research and Quality. Medical expenditure panel survey. US Department of Health & Human Services Web site. http://meps.ahrq.gov. Accessed November 17, 2014.
- Bickers DR, Lim HW, Margolis D, et al. The burden of skin diseases: 2004 a joint project of the American Academy of Dermatology Association and the Society for Investigative Dermatology. J Am Acad Dermatol. 2006;55:490-500.
- Johnson ML. On teaching dermatology to nondermatologists. Arch Dermatol. 1994;130:850-852.
- Ramsay DL, Weary PE. Primary care in dermatology: whose role should it be? J Am Acad Dermatol. 1996;35:1005-1008.
- Kimball AB, Resneck JS Jr. The US dermatology workforce: a specialty remains in shortage. J Am Acad Dermatol. 2008;59:741-745.
- Resneck JS Jr, Kimball AB. Who else is providing care in dermatology practices? trends in the use of nonphysician clinicians. J Am Acad Dermatol. 2008;58:211-216.
- Feldman SR, Fleischer AB Jr, McConnell RC. Most common dermatologic problems identified by internists, 1990-1994. Arch Intern Med. 1998;158:726-730.
- Ahn CS, Davis SA, Debade TS, et al. Noncosmetic skin-related procedures performed in the United States: an analysis of national ambulatory medical care survey data from 1995 to 2010. Dermatol Surg. 2013;39:1912-1921.
- Antic M, Conen D, Itin PH. Teaching effects of dermatological consultations on nondermatologists in the field of internal medicine. a study of 1290 inpatients. Dermatology. 2004;208:32-37.
- Federman DG, Concato J, Kirsner RS. Comparison of dermatologic diagnoses by primary care practitioners and dermatologists. a review of the literature. Arch Fam Med. 1999;8:170-172.
- Fleischer AB Jr, Herbert CR, Feldman SR, et al. Diagnosis of skin disease by nondermatologists. Am J Manag Care. 2000;6:1149-1156.
- Kirsner RS, Federman DG. Lack of correlation between internists’ ability in dermatology and their patterns of treating patients with skin disease. Arch Dermatol. 1996;132:1043-1046.
- McCarthy GM, Lamb GC, Russell TJ, et al. Primary care-based dermatology practice: internists need more training. J Gen Intern Med. 1991;6:52-56.
- Sellheyer K, Bergfeld WF. A retrospective biopsy study of the clinical diagnostic accuracy of common skin diseases by different specialties compared with dermatology. J Am Acad Dermatol. 2005;52:823-830.
- Medical specialties. Accreditation Council for Graduate Medical Education Web site. http://www.acgme.org/acgmeweb/tabid/368ProgramandInstitutionalGuidelines/MedicalAccreditation.aspx. Accessed November 17, 2014.
- McCleskey PE, Gilson RT, DeVillez RL. Medical student core curriculum in dermatology survey. J Am Acad Dermatol. 2009;61:30-35.
- Hansra NK, O’Sullivan P, Chen CL, et al. Medical school dermatology curriculum: are we adequately preparing primary care physicians? J Am Acad Dermatol. 2009;61:23-29.
- Thorpe KE, Florence CS, Joski P. Which medical conditions account for the rise in health care spending? Health Aff (Millwood). 2004;(suppl web exclusives):W4-437-445.
- Johnson ML. Defining the burden of skin disease in the United States—a historical perspective. J Investig Dermatol Symp Proc. 2004;9:108-110.
- Agency for Healthcare Research and Quality. Medical expenditure panel survey. US Department of Health & Human Services Web site. http://meps.ahrq.gov. Accessed November 17, 2014.
- Bickers DR, Lim HW, Margolis D, et al. The burden of skin diseases: 2004 a joint project of the American Academy of Dermatology Association and the Society for Investigative Dermatology. J Am Acad Dermatol. 2006;55:490-500.
- Johnson ML. On teaching dermatology to nondermatologists. Arch Dermatol. 1994;130:850-852.
- Ramsay DL, Weary PE. Primary care in dermatology: whose role should it be? J Am Acad Dermatol. 1996;35:1005-1008.
- Kimball AB, Resneck JS Jr. The US dermatology workforce: a specialty remains in shortage. J Am Acad Dermatol. 2008;59:741-745.
- Resneck JS Jr, Kimball AB. Who else is providing care in dermatology practices? trends in the use of nonphysician clinicians. J Am Acad Dermatol. 2008;58:211-216.
- Feldman SR, Fleischer AB Jr, McConnell RC. Most common dermatologic problems identified by internists, 1990-1994. Arch Intern Med. 1998;158:726-730.
- Ahn CS, Davis SA, Debade TS, et al. Noncosmetic skin-related procedures performed in the United States: an analysis of national ambulatory medical care survey data from 1995 to 2010. Dermatol Surg. 2013;39:1912-1921.
- Antic M, Conen D, Itin PH. Teaching effects of dermatological consultations on nondermatologists in the field of internal medicine. a study of 1290 inpatients. Dermatology. 2004;208:32-37.
- Federman DG, Concato J, Kirsner RS. Comparison of dermatologic diagnoses by primary care practitioners and dermatologists. a review of the literature. Arch Fam Med. 1999;8:170-172.
- Fleischer AB Jr, Herbert CR, Feldman SR, et al. Diagnosis of skin disease by nondermatologists. Am J Manag Care. 2000;6:1149-1156.
- Kirsner RS, Federman DG. Lack of correlation between internists’ ability in dermatology and their patterns of treating patients with skin disease. Arch Dermatol. 1996;132:1043-1046.
- McCarthy GM, Lamb GC, Russell TJ, et al. Primary care-based dermatology practice: internists need more training. J Gen Intern Med. 1991;6:52-56.
- Sellheyer K, Bergfeld WF. A retrospective biopsy study of the clinical diagnostic accuracy of common skin diseases by different specialties compared with dermatology. J Am Acad Dermatol. 2005;52:823-830.
- Medical specialties. Accreditation Council for Graduate Medical Education Web site. http://www.acgme.org/acgmeweb/tabid/368ProgramandInstitutionalGuidelines/MedicalAccreditation.aspx. Accessed November 17, 2014.
- McCleskey PE, Gilson RT, DeVillez RL. Medical student core curriculum in dermatology survey. J Am Acad Dermatol. 2009;61:30-35.
- Hansra NK, O’Sullivan P, Chen CL, et al. Medical school dermatology curriculum: are we adequately preparing primary care physicians? J Am Acad Dermatol. 2009;61:23-29.
Practice Points
- Approximately half of skin-related visits are to nondermatologists, such as family medicine physicians, pediatricians, and internists.
- Skin conditions that most frequently present to nondermatologists are different from those seen by dermatologists.
- Education efforts in nondermatology specialties should be targeted toward the common skin diseases that present to these specialties to maximize the yield of medical education and improve diagnostic accuracy and patient outcomes.
Oral tofacitinib scores against psoriasis in phase III trial
AMSTERDAM – Oral tofacitinib for psoriasis proved as effective as – and in certain domains better than – subcutaneous etanercept in improving patient-reported quality of life endpoints in a phase III clinical trial.
The double-blind study included 1,101 patients with moderate to severe chronic plaque psoriasis who were randomized to 12 weeks of the oral Janus kinase inhibitor tofacitinib at 5 mg twice daily, 10 mg twice daily, subcutaneous etanercept (Enbrel) at 50 mg twice weekly, or placebo. The findings were presented by Dr. Fernando Valenzuela at the annual congress of the European Academy of Dermatology and Venereology.
Earlier in 2014, at the annual meeting of the American Academy of Dermatology in Denver, Dr. Valenzuela presented the phase III study’s primary results, in which the oral small molecule proved noninferior to the tumor necrosis factor inhibitor in improving PASI scores. At the EADV Congress in Amsterdam, he focused on the secondary outcomes, which arguably matter more to patients than do changes in PASI scores, namely, measures of quality of life and itchiness.
From a baseline Itch Severity Item score of 5, indicative of moderate to severe itching, week 12 scores fell by a mean of 3.2, 4.0, 3.5, and 0.4 points, respectively, in patients on low- or high-dose tofacitinib, etanercept, and placebo. The improvement in itch in patients randomized to tofacitinib at 10 mg twice daily was not only greater than with etanercept, it occurred faster as well, with significant reduction in itch scores documented on day 2 of therapy, reported Dr. Valenzuela, a dermatologist at the University of Chile in Santiago.
The median baseline Dermatology Life Quality Index (DLQI) score was 12. Significant improvement was seen from week 4 in all three active treatment study arms. By week 12, DLQI scores had dropped by an average of 7.3 points in patients on tofacitinib at 5 mg twice daily, 9.7 points in those on 10 mg twice daily, 9.0 points in etanercept-treated patients, and 1.9 points with placebo. Seventy-eight percent of patients on tofacitinib at 10 mg twice daily experienced a 5-point or larger drop in the DLQI by week 12, as did 75% of those on etanercept, 66% of those on low-dose tofacitinib, and 32% of those on placebo.
At baseline, 30% of patients rated their psoriasis as moderate and 70% rated their psoriasis as severe based on Patient Global Assessment. By week 12, more than 50% of patients on tofacitinib at 10 mg twice daily or etanercept rated their skin as clear or almost clear.
More than 70% of patients in all three active treatment arms indicated at week 12 that they were satisfied with their medication. Satisfaction scores were highest, and equally so, in those on high-dose tofacitinib and etanercept.
“Patients using tofacitinib at the low dose had not that good an improvement. It was better improvement than with placebo, but it doesn’t look like etanercept,” Dr. Valenzuela concluded.
Tofacitinib is approved as Xeljanz for the treatment of rheumatoid arthritis, but remains investigational for psoriasis. According to a company statement, Pfizer intends to submit a supplemental New Drug Application (sNDA) to the Food and Drug Administration by early 2015.
The study was funded by Pfizer. Dr. Valenzuela is an adviser to Pfizer, AbbVie, Eli Lilly, Janssen, Merck, and Novartis.
AMSTERDAM – Oral tofacitinib for psoriasis proved as effective as – and in certain domains better than – subcutaneous etanercept in improving patient-reported quality of life endpoints in a phase III clinical trial.
The double-blind study included 1,101 patients with moderate to severe chronic plaque psoriasis who were randomized to 12 weeks of the oral Janus kinase inhibitor tofacitinib at 5 mg twice daily, 10 mg twice daily, subcutaneous etanercept (Enbrel) at 50 mg twice weekly, or placebo. The findings were presented by Dr. Fernando Valenzuela at the annual congress of the European Academy of Dermatology and Venereology.
Earlier in 2014, at the annual meeting of the American Academy of Dermatology in Denver, Dr. Valenzuela presented the phase III study’s primary results, in which the oral small molecule proved noninferior to the tumor necrosis factor inhibitor in improving PASI scores. At the EADV Congress in Amsterdam, he focused on the secondary outcomes, which arguably matter more to patients than do changes in PASI scores, namely, measures of quality of life and itchiness.
From a baseline Itch Severity Item score of 5, indicative of moderate to severe itching, week 12 scores fell by a mean of 3.2, 4.0, 3.5, and 0.4 points, respectively, in patients on low- or high-dose tofacitinib, etanercept, and placebo. The improvement in itch in patients randomized to tofacitinib at 10 mg twice daily was not only greater than with etanercept, it occurred faster as well, with significant reduction in itch scores documented on day 2 of therapy, reported Dr. Valenzuela, a dermatologist at the University of Chile in Santiago.
The median baseline Dermatology Life Quality Index (DLQI) score was 12. Significant improvement was seen from week 4 in all three active treatment study arms. By week 12, DLQI scores had dropped by an average of 7.3 points in patients on tofacitinib at 5 mg twice daily, 9.7 points in those on 10 mg twice daily, 9.0 points in etanercept-treated patients, and 1.9 points with placebo. Seventy-eight percent of patients on tofacitinib at 10 mg twice daily experienced a 5-point or larger drop in the DLQI by week 12, as did 75% of those on etanercept, 66% of those on low-dose tofacitinib, and 32% of those on placebo.
At baseline, 30% of patients rated their psoriasis as moderate and 70% rated their psoriasis as severe based on Patient Global Assessment. By week 12, more than 50% of patients on tofacitinib at 10 mg twice daily or etanercept rated their skin as clear or almost clear.
More than 70% of patients in all three active treatment arms indicated at week 12 that they were satisfied with their medication. Satisfaction scores were highest, and equally so, in those on high-dose tofacitinib and etanercept.
“Patients using tofacitinib at the low dose had not that good an improvement. It was better improvement than with placebo, but it doesn’t look like etanercept,” Dr. Valenzuela concluded.
Tofacitinib is approved as Xeljanz for the treatment of rheumatoid arthritis, but remains investigational for psoriasis. According to a company statement, Pfizer intends to submit a supplemental New Drug Application (sNDA) to the Food and Drug Administration by early 2015.
The study was funded by Pfizer. Dr. Valenzuela is an adviser to Pfizer, AbbVie, Eli Lilly, Janssen, Merck, and Novartis.
AMSTERDAM – Oral tofacitinib for psoriasis proved as effective as – and in certain domains better than – subcutaneous etanercept in improving patient-reported quality of life endpoints in a phase III clinical trial.
The double-blind study included 1,101 patients with moderate to severe chronic plaque psoriasis who were randomized to 12 weeks of the oral Janus kinase inhibitor tofacitinib at 5 mg twice daily, 10 mg twice daily, subcutaneous etanercept (Enbrel) at 50 mg twice weekly, or placebo. The findings were presented by Dr. Fernando Valenzuela at the annual congress of the European Academy of Dermatology and Venereology.
Earlier in 2014, at the annual meeting of the American Academy of Dermatology in Denver, Dr. Valenzuela presented the phase III study’s primary results, in which the oral small molecule proved noninferior to the tumor necrosis factor inhibitor in improving PASI scores. At the EADV Congress in Amsterdam, he focused on the secondary outcomes, which arguably matter more to patients than do changes in PASI scores, namely, measures of quality of life and itchiness.
From a baseline Itch Severity Item score of 5, indicative of moderate to severe itching, week 12 scores fell by a mean of 3.2, 4.0, 3.5, and 0.4 points, respectively, in patients on low- or high-dose tofacitinib, etanercept, and placebo. The improvement in itch in patients randomized to tofacitinib at 10 mg twice daily was not only greater than with etanercept, it occurred faster as well, with significant reduction in itch scores documented on day 2 of therapy, reported Dr. Valenzuela, a dermatologist at the University of Chile in Santiago.
The median baseline Dermatology Life Quality Index (DLQI) score was 12. Significant improvement was seen from week 4 in all three active treatment study arms. By week 12, DLQI scores had dropped by an average of 7.3 points in patients on tofacitinib at 5 mg twice daily, 9.7 points in those on 10 mg twice daily, 9.0 points in etanercept-treated patients, and 1.9 points with placebo. Seventy-eight percent of patients on tofacitinib at 10 mg twice daily experienced a 5-point or larger drop in the DLQI by week 12, as did 75% of those on etanercept, 66% of those on low-dose tofacitinib, and 32% of those on placebo.
At baseline, 30% of patients rated their psoriasis as moderate and 70% rated their psoriasis as severe based on Patient Global Assessment. By week 12, more than 50% of patients on tofacitinib at 10 mg twice daily or etanercept rated their skin as clear or almost clear.
More than 70% of patients in all three active treatment arms indicated at week 12 that they were satisfied with their medication. Satisfaction scores were highest, and equally so, in those on high-dose tofacitinib and etanercept.
“Patients using tofacitinib at the low dose had not that good an improvement. It was better improvement than with placebo, but it doesn’t look like etanercept,” Dr. Valenzuela concluded.
Tofacitinib is approved as Xeljanz for the treatment of rheumatoid arthritis, but remains investigational for psoriasis. According to a company statement, Pfizer intends to submit a supplemental New Drug Application (sNDA) to the Food and Drug Administration by early 2015.
The study was funded by Pfizer. Dr. Valenzuela is an adviser to Pfizer, AbbVie, Eli Lilly, Janssen, Merck, and Novartis.
AT THE EADV CONGRESS
Key clinical point: Multiple measures of quality of life and disease burden improved similarly in psoriasis patients regardless of whether they were on subcutaneous etanercept or the oral Janus kinase inhibitor tofacitinib.
Major finding: From a median baseline Dermatology Life Quality Index score of 12, scores improved by an average of 7.3 points after 12 weeks of tofacitinib at 5 mg twice daily, 9.7 points with tofacitinib at 10 mg twice daily, 9.0 points with etanercept at 50 mg twice weekly, and 1.9 points with placebo.
Data source: A phase III randomized, double-blind prospective study involving 1,101 patients with moderate to severe chronic plaque psoriasis.
Disclosures: The presenter is an adviser to Pfizer, which sponsored the study, and other pharmaceutical companies.
Dermatologists counsel psoriasis patients on obesity, less on alcohol and tobacco
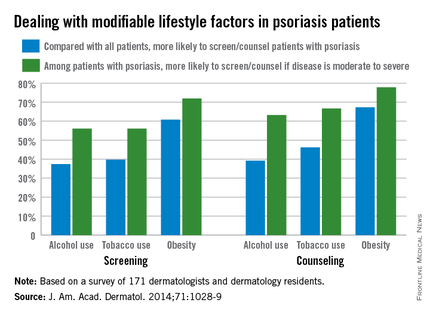
Less than half of dermatologists screen or counsel psoriasis patients about alcohol or tobacco use, compared with all patients, but approximately 60% screen or counsel psoriasis patients about obesity, based on data from an online survey of 171 dermatologists and dermatology residents.
The findings were published in a letter to the editor in the November issue of the Journal of the American Academy of Dermatology (2014;71:1028-9).
However, among patients with psoriasis, more than two-thirds of the respondents said they were more likely to both screen and counsel patients with moderate to severe psoriasis about alcohol, tobacco, and obesity, compared with psoriasis patients overall.
Previous research suggests that if psoriasis patients’ issues with alcohol, tobacco, and obesity are addressed, “patients may experience disease improvement, emphasizing the importance of screening and counseling,” wrote Brandon L. Adler and Aimee E. Krausz, medical students at Albert Einstein College of Medicine, Bronx, N.Y., and their colleagues.
Overall, 87% of respondents believed themselves responsible for screening for the three risk factors, but 56% believed themselves responsible for counseling.
Although the results were limited by the focus on academic dermatologists, the findings suggest a need to improve dermatologists’ confidence in counseling patients, the researchers noted. “Systematic training and effective counseling instruments would empower practitioners to translate this knowledge into clinical practice.”
The study was supported by the Dermatology Foundation Career Development Awards Program.
The researchers had no financial conflicts to disclose.
|
Less than half of dermatologists screen or counsel psoriasis patients about alcohol or tobacco use, compared with all patients, but approximately 60% screen or counsel psoriasis patients about obesity, based on data from an online survey of 171 dermatologists and dermatology residents.
The findings were published in a letter to the editor in the November issue of the Journal of the American Academy of Dermatology (2014;71:1028-9).
However, among patients with psoriasis, more than two-thirds of the respondents said they were more likely to both screen and counsel patients with moderate to severe psoriasis about alcohol, tobacco, and obesity, compared with psoriasis patients overall.
Previous research suggests that if psoriasis patients’ issues with alcohol, tobacco, and obesity are addressed, “patients may experience disease improvement, emphasizing the importance of screening and counseling,” wrote Brandon L. Adler and Aimee E. Krausz, medical students at Albert Einstein College of Medicine, Bronx, N.Y., and their colleagues.
Overall, 87% of respondents believed themselves responsible for screening for the three risk factors, but 56% believed themselves responsible for counseling.
Although the results were limited by the focus on academic dermatologists, the findings suggest a need to improve dermatologists’ confidence in counseling patients, the researchers noted. “Systematic training and effective counseling instruments would empower practitioners to translate this knowledge into clinical practice.”
The study was supported by the Dermatology Foundation Career Development Awards Program.
The researchers had no financial conflicts to disclose.
|

Less than half of dermatologists screen or counsel psoriasis patients about alcohol or tobacco use, compared with all patients, but approximately 60% screen or counsel psoriasis patients about obesity, based on data from an online survey of 171 dermatologists and dermatology residents.
The findings were published in a letter to the editor in the November issue of the Journal of the American Academy of Dermatology (2014;71:1028-9).
However, among patients with psoriasis, more than two-thirds of the respondents said they were more likely to both screen and counsel patients with moderate to severe psoriasis about alcohol, tobacco, and obesity, compared with psoriasis patients overall.
Previous research suggests that if psoriasis patients’ issues with alcohol, tobacco, and obesity are addressed, “patients may experience disease improvement, emphasizing the importance of screening and counseling,” wrote Brandon L. Adler and Aimee E. Krausz, medical students at Albert Einstein College of Medicine, Bronx, N.Y., and their colleagues.
Overall, 87% of respondents believed themselves responsible for screening for the three risk factors, but 56% believed themselves responsible for counseling.
Although the results were limited by the focus on academic dermatologists, the findings suggest a need to improve dermatologists’ confidence in counseling patients, the researchers noted. “Systematic training and effective counseling instruments would empower practitioners to translate this knowledge into clinical practice.”
The study was supported by the Dermatology Foundation Career Development Awards Program.
The researchers had no financial conflicts to disclose.
|

FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Secukinumab tames psoriatic arthritis in FUTURE 2 trial
BOSTON – Secukinumab improved quality of life and symptoms in patients with psoriatic arthritis, regardless of whether they had received prior anti-tumor-necrosis factor therapy or were concurrently receiving methotrexate, Dr. Ian B. McInnes reported at the annual meeting of the American College of Rheumatology.
A 300-mg dose of the investigational drug, which is given subcutaneously, proved to be the most effective dose, Dr. McInnes, of the University of Glasgow, Scotland, said in his presentation of the 24-week results of the FUTURE 2 study. The primary endpoint, a response of at least ACR20, was achieved by 54% of 100 patients given the 300-mg dose and by 51% of 100 patients given the 150-mg dose. The 75-mg dose was far less effective, with a 29% response in 99 patients; the 98 patients given placebo had a 15% response.
At the 150-mg and 300-mg doses, the rates of ACR20 responses were comparable whether or not patients were also taking concomitant methotrexate. Further, the drug’s safety profile was comparable to placebo, Dr. McInnes said.
An ACR50 was achieved by 35% of patients given secukinumab at either 300 mg or 150 mg, and by 18% of those given 75 mg and 7% of those given placebo. About 20% of patients given the higher doses had an ACR70 response, as did 6% of those given 75 mg and 1% of those given placebo.
At the 300-mg dose, secukinumab also resolved dactylitis and enthesitis in approximately half of the affected patients.
Mean improvements in quality of life based on patients’ SF 36 PCS score (Short Form-36 Physical Component Summary) at 24 weeks from baseline were 7.25 in those on the 300-mg dose and 6.39 in those on the 150-mg dose. Those on the 75-mg dose had a 4.38-point mean improvement, whereas patients on placebo had a 1.34-point mean improvement in SF 36 PCS.
No safety signals were noted; adverse events were few and comparable to placebo. Five subjects on the active drug had mild to moderate candidal infections that responded to oral therapy. Neutropenia occurred in one patient in the 300-mg dose group and in one patient in the placebo group, but was transient and patients continued on therapy.
Secukinumab is a fully-human IgG1k monoclonal antibody that selectively targets IL-17A. The drug, manufactured by Novartis, was unanimously recommended for approval by an advisory committee to the Food and Drug Administration. It was administered weekly as a subcutaneous injection for the first 4 weeks of the study, then given again at week 8 and once every 4 weeks thereafter in patients assigned to one of the secukinumab arms of the double-blind, randomized study. Patient assigned to the placebo group were either responders and assigned to receive secukinumab at week 24 and every 4 weeks thereafter or were nonresponders assigned to receive secukinumab at week 16 and every 4 weeks thereafter. Only patients with at least 20% reductions in the number of tender joints or swollen joints continue to receive the drug beyond 1 year.
To be eligible for the study, patients needed to have a diagnosis of active psoriatic arthritis classified by CASPAR criteria and tenderness in at least 3 of 78 joints and swelling of at least 3 of 76 joints. They additionally needed to have an inadequate response to nonsteroidal anti-inflammatory drugs, methotrexate, or anti-TNF therapy
The primary endpoint of the study was ACR20 response at 24 weeks. Secondary endpoints included PASI 75 and PASI 90 responses, change in DAS28-CRP (28-joint Disease Activity Score using C-reactive protein) from baseline, change in SF-36 PCS and HAQ-DI (Health Assessment Questionnaire-Disability Index) from baseline, ACR50 response, the proportion of subjects with dactylitis and enthesitis, and overall safety and tolerability.
As of press time, the FDA was expected to take additional action in January 2015, according to Novartis.
The study was sponsored by Novartis, the maker of secukinumab. Dr. McInnes receives consulting fees from Novartis as well as multiple other pharmaceutical companies.
BOSTON – Secukinumab improved quality of life and symptoms in patients with psoriatic arthritis, regardless of whether they had received prior anti-tumor-necrosis factor therapy or were concurrently receiving methotrexate, Dr. Ian B. McInnes reported at the annual meeting of the American College of Rheumatology.
A 300-mg dose of the investigational drug, which is given subcutaneously, proved to be the most effective dose, Dr. McInnes, of the University of Glasgow, Scotland, said in his presentation of the 24-week results of the FUTURE 2 study. The primary endpoint, a response of at least ACR20, was achieved by 54% of 100 patients given the 300-mg dose and by 51% of 100 patients given the 150-mg dose. The 75-mg dose was far less effective, with a 29% response in 99 patients; the 98 patients given placebo had a 15% response.
At the 150-mg and 300-mg doses, the rates of ACR20 responses were comparable whether or not patients were also taking concomitant methotrexate. Further, the drug’s safety profile was comparable to placebo, Dr. McInnes said.
An ACR50 was achieved by 35% of patients given secukinumab at either 300 mg or 150 mg, and by 18% of those given 75 mg and 7% of those given placebo. About 20% of patients given the higher doses had an ACR70 response, as did 6% of those given 75 mg and 1% of those given placebo.
At the 300-mg dose, secukinumab also resolved dactylitis and enthesitis in approximately half of the affected patients.
Mean improvements in quality of life based on patients’ SF 36 PCS score (Short Form-36 Physical Component Summary) at 24 weeks from baseline were 7.25 in those on the 300-mg dose and 6.39 in those on the 150-mg dose. Those on the 75-mg dose had a 4.38-point mean improvement, whereas patients on placebo had a 1.34-point mean improvement in SF 36 PCS.
No safety signals were noted; adverse events were few and comparable to placebo. Five subjects on the active drug had mild to moderate candidal infections that responded to oral therapy. Neutropenia occurred in one patient in the 300-mg dose group and in one patient in the placebo group, but was transient and patients continued on therapy.
Secukinumab is a fully-human IgG1k monoclonal antibody that selectively targets IL-17A. The drug, manufactured by Novartis, was unanimously recommended for approval by an advisory committee to the Food and Drug Administration. It was administered weekly as a subcutaneous injection for the first 4 weeks of the study, then given again at week 8 and once every 4 weeks thereafter in patients assigned to one of the secukinumab arms of the double-blind, randomized study. Patient assigned to the placebo group were either responders and assigned to receive secukinumab at week 24 and every 4 weeks thereafter or were nonresponders assigned to receive secukinumab at week 16 and every 4 weeks thereafter. Only patients with at least 20% reductions in the number of tender joints or swollen joints continue to receive the drug beyond 1 year.
To be eligible for the study, patients needed to have a diagnosis of active psoriatic arthritis classified by CASPAR criteria and tenderness in at least 3 of 78 joints and swelling of at least 3 of 76 joints. They additionally needed to have an inadequate response to nonsteroidal anti-inflammatory drugs, methotrexate, or anti-TNF therapy
The primary endpoint of the study was ACR20 response at 24 weeks. Secondary endpoints included PASI 75 and PASI 90 responses, change in DAS28-CRP (28-joint Disease Activity Score using C-reactive protein) from baseline, change in SF-36 PCS and HAQ-DI (Health Assessment Questionnaire-Disability Index) from baseline, ACR50 response, the proportion of subjects with dactylitis and enthesitis, and overall safety and tolerability.
As of press time, the FDA was expected to take additional action in January 2015, according to Novartis.
The study was sponsored by Novartis, the maker of secukinumab. Dr. McInnes receives consulting fees from Novartis as well as multiple other pharmaceutical companies.
BOSTON – Secukinumab improved quality of life and symptoms in patients with psoriatic arthritis, regardless of whether they had received prior anti-tumor-necrosis factor therapy or were concurrently receiving methotrexate, Dr. Ian B. McInnes reported at the annual meeting of the American College of Rheumatology.
A 300-mg dose of the investigational drug, which is given subcutaneously, proved to be the most effective dose, Dr. McInnes, of the University of Glasgow, Scotland, said in his presentation of the 24-week results of the FUTURE 2 study. The primary endpoint, a response of at least ACR20, was achieved by 54% of 100 patients given the 300-mg dose and by 51% of 100 patients given the 150-mg dose. The 75-mg dose was far less effective, with a 29% response in 99 patients; the 98 patients given placebo had a 15% response.
At the 150-mg and 300-mg doses, the rates of ACR20 responses were comparable whether or not patients were also taking concomitant methotrexate. Further, the drug’s safety profile was comparable to placebo, Dr. McInnes said.
An ACR50 was achieved by 35% of patients given secukinumab at either 300 mg or 150 mg, and by 18% of those given 75 mg and 7% of those given placebo. About 20% of patients given the higher doses had an ACR70 response, as did 6% of those given 75 mg and 1% of those given placebo.
At the 300-mg dose, secukinumab also resolved dactylitis and enthesitis in approximately half of the affected patients.
Mean improvements in quality of life based on patients’ SF 36 PCS score (Short Form-36 Physical Component Summary) at 24 weeks from baseline were 7.25 in those on the 300-mg dose and 6.39 in those on the 150-mg dose. Those on the 75-mg dose had a 4.38-point mean improvement, whereas patients on placebo had a 1.34-point mean improvement in SF 36 PCS.
No safety signals were noted; adverse events were few and comparable to placebo. Five subjects on the active drug had mild to moderate candidal infections that responded to oral therapy. Neutropenia occurred in one patient in the 300-mg dose group and in one patient in the placebo group, but was transient and patients continued on therapy.
Secukinumab is a fully-human IgG1k monoclonal antibody that selectively targets IL-17A. The drug, manufactured by Novartis, was unanimously recommended for approval by an advisory committee to the Food and Drug Administration. It was administered weekly as a subcutaneous injection for the first 4 weeks of the study, then given again at week 8 and once every 4 weeks thereafter in patients assigned to one of the secukinumab arms of the double-blind, randomized study. Patient assigned to the placebo group were either responders and assigned to receive secukinumab at week 24 and every 4 weeks thereafter or were nonresponders assigned to receive secukinumab at week 16 and every 4 weeks thereafter. Only patients with at least 20% reductions in the number of tender joints or swollen joints continue to receive the drug beyond 1 year.
To be eligible for the study, patients needed to have a diagnosis of active psoriatic arthritis classified by CASPAR criteria and tenderness in at least 3 of 78 joints and swelling of at least 3 of 76 joints. They additionally needed to have an inadequate response to nonsteroidal anti-inflammatory drugs, methotrexate, or anti-TNF therapy
The primary endpoint of the study was ACR20 response at 24 weeks. Secondary endpoints included PASI 75 and PASI 90 responses, change in DAS28-CRP (28-joint Disease Activity Score using C-reactive protein) from baseline, change in SF-36 PCS and HAQ-DI (Health Assessment Questionnaire-Disability Index) from baseline, ACR50 response, the proportion of subjects with dactylitis and enthesitis, and overall safety and tolerability.
As of press time, the FDA was expected to take additional action in January 2015, according to Novartis.
The study was sponsored by Novartis, the maker of secukinumab. Dr. McInnes receives consulting fees from Novartis as well as multiple other pharmaceutical companies.
AT THE ACR ANNUAL MEETING
Key clinical point: Secukinumab could prove to be a major new therapy for psoriatic arthritis.
Major finding: After 24 weeks, a response of at least ACR20 was achieved by 54% of 100 patients given the 300-mg dose of secukinumab.
Data source: The FUTURE 2 study group of nearly 400 patients with psoriatic arthritis who were randomized either to one of three doses of secukinumab or placebo.
Disclosures: The study was sponsored by Novartis, the maker of secukinumab. Dr. McInnes receives consulting fees from Novartis as well as multiple other pharmaceutical companies.
Few psoriatic arthritis patients achieve minimal disease activity on methotrexate
BOSTON – It’s time to test whether methotrexate is really up to snuff as a first-line therapy for psoriatic arthritis, Canadian investigators say.
They base that recommendation on findings from a retrospective study showing that fewer than 18% of patients with psoriatic arthritis treated with methotrexate achieved minimal disease activity (MDA) after 6 months.
They also found evidence to suggest that physicians may overestimate the benefits of methotrexate for psoriatic arthritis.
“Physician-dependent measures reveal good response to methotrexate at 6 months, but patient-reported measures are less responsive, possibly due to side effects, back pain, disease, [and/or] disability,” said Dr. Barry J. Sheane of the division of rheumatology in the department of medicine at the University of Toronto.
The data on the effects of methotrexate in psoriatic arthritis are considerably less impressive than are those seen with tumor necrosis factor–alpha inhibitors (TNFi), Dr. Sheane said. For example, at 24 weeks, 39% of patients treated with adalimumab (Humira) and 52% treated with infliximab (Remicade) had achieved MDA, he noted.
“We suggest that a randomized controlled trial of methotrexate compared to a TNF inhibitor is warranted,” Dr. Sheane said at the annual meeting of the American College of Rheumatology.
He and his colleagues reviewed records on 204 consecutive patients treated for psoriatic arthritis with methotrexate from January 2004 through April 2014. The patients, who had a mean duration of psoriatic arthritis of 6.2 years, were all naive to biological antirheumatic drugs and were initiating methotrexate therapy.
Of the 204 patients, 167 had data sufficient for a 6-month analysis.
The investigators defined MDA after 6 months on methotrexate, the primary endpoint, as the presence of at least five out of the following seven domains: 0-1 tender joints, 0-1 swollen joints, Psoriasis Area Severity Index (PASI) 1 or less or body surface area involved 3% or less, 0-1 tender entheseal points, Health Assessment Questionnaire (HAQ) score of 0.5 or less, patient global disease activity Visual Analog Scale score of 20 or lower, and patient pain Visual Analog Scale score of 15 or lower.
They found that 17.4% of patients (29 of 167) met the physician-rated criteria for MDA at 6 months, even though 82.6% of the patients (138) had a PASI score of 1 or lower, and 58.1% (97) had a swollen joint count of 1 or fewer.
When the patients were asked for their assessment of global disease activity, 13.2% (22) rated disease activity with a prespecified score of 20 or lower. Only 12% of patients (20) reported an HAQ score of 0.5 or less.
The mean methotrexate dose was similar between patients who achieved MDA (17.8 mg/week) and those who did not (17.3 mg/week). The median dose was 17.5 mg/week in each group.
After controlling for sex, baseline sacroiliitis, and duration of psoriasis and psoriatic arthritis at the start of methotrexate in a multivariate model, the authors found that only dactylitis, inflammatory back pain, and mechanical back pain were significantly associated with a lower probability of patients reaching MDA. The variables that did not prove to be associated with achieving MDA after 6 months on methotrexate included erythrocyte sedimentation rate, presence of nail disease, number of clinically damaged joints, and body mass index.
The study was internally funded. Dr. Sheane reported having no relevant disclosures.
BOSTON – It’s time to test whether methotrexate is really up to snuff as a first-line therapy for psoriatic arthritis, Canadian investigators say.
They base that recommendation on findings from a retrospective study showing that fewer than 18% of patients with psoriatic arthritis treated with methotrexate achieved minimal disease activity (MDA) after 6 months.
They also found evidence to suggest that physicians may overestimate the benefits of methotrexate for psoriatic arthritis.
“Physician-dependent measures reveal good response to methotrexate at 6 months, but patient-reported measures are less responsive, possibly due to side effects, back pain, disease, [and/or] disability,” said Dr. Barry J. Sheane of the division of rheumatology in the department of medicine at the University of Toronto.
The data on the effects of methotrexate in psoriatic arthritis are considerably less impressive than are those seen with tumor necrosis factor–alpha inhibitors (TNFi), Dr. Sheane said. For example, at 24 weeks, 39% of patients treated with adalimumab (Humira) and 52% treated with infliximab (Remicade) had achieved MDA, he noted.
“We suggest that a randomized controlled trial of methotrexate compared to a TNF inhibitor is warranted,” Dr. Sheane said at the annual meeting of the American College of Rheumatology.
He and his colleagues reviewed records on 204 consecutive patients treated for psoriatic arthritis with methotrexate from January 2004 through April 2014. The patients, who had a mean duration of psoriatic arthritis of 6.2 years, were all naive to biological antirheumatic drugs and were initiating methotrexate therapy.
Of the 204 patients, 167 had data sufficient for a 6-month analysis.
The investigators defined MDA after 6 months on methotrexate, the primary endpoint, as the presence of at least five out of the following seven domains: 0-1 tender joints, 0-1 swollen joints, Psoriasis Area Severity Index (PASI) 1 or less or body surface area involved 3% or less, 0-1 tender entheseal points, Health Assessment Questionnaire (HAQ) score of 0.5 or less, patient global disease activity Visual Analog Scale score of 20 or lower, and patient pain Visual Analog Scale score of 15 or lower.
They found that 17.4% of patients (29 of 167) met the physician-rated criteria for MDA at 6 months, even though 82.6% of the patients (138) had a PASI score of 1 or lower, and 58.1% (97) had a swollen joint count of 1 or fewer.
When the patients were asked for their assessment of global disease activity, 13.2% (22) rated disease activity with a prespecified score of 20 or lower. Only 12% of patients (20) reported an HAQ score of 0.5 or less.
The mean methotrexate dose was similar between patients who achieved MDA (17.8 mg/week) and those who did not (17.3 mg/week). The median dose was 17.5 mg/week in each group.
After controlling for sex, baseline sacroiliitis, and duration of psoriasis and psoriatic arthritis at the start of methotrexate in a multivariate model, the authors found that only dactylitis, inflammatory back pain, and mechanical back pain were significantly associated with a lower probability of patients reaching MDA. The variables that did not prove to be associated with achieving MDA after 6 months on methotrexate included erythrocyte sedimentation rate, presence of nail disease, number of clinically damaged joints, and body mass index.
The study was internally funded. Dr. Sheane reported having no relevant disclosures.
BOSTON – It’s time to test whether methotrexate is really up to snuff as a first-line therapy for psoriatic arthritis, Canadian investigators say.
They base that recommendation on findings from a retrospective study showing that fewer than 18% of patients with psoriatic arthritis treated with methotrexate achieved minimal disease activity (MDA) after 6 months.
They also found evidence to suggest that physicians may overestimate the benefits of methotrexate for psoriatic arthritis.
“Physician-dependent measures reveal good response to methotrexate at 6 months, but patient-reported measures are less responsive, possibly due to side effects, back pain, disease, [and/or] disability,” said Dr. Barry J. Sheane of the division of rheumatology in the department of medicine at the University of Toronto.
The data on the effects of methotrexate in psoriatic arthritis are considerably less impressive than are those seen with tumor necrosis factor–alpha inhibitors (TNFi), Dr. Sheane said. For example, at 24 weeks, 39% of patients treated with adalimumab (Humira) and 52% treated with infliximab (Remicade) had achieved MDA, he noted.
“We suggest that a randomized controlled trial of methotrexate compared to a TNF inhibitor is warranted,” Dr. Sheane said at the annual meeting of the American College of Rheumatology.
He and his colleagues reviewed records on 204 consecutive patients treated for psoriatic arthritis with methotrexate from January 2004 through April 2014. The patients, who had a mean duration of psoriatic arthritis of 6.2 years, were all naive to biological antirheumatic drugs and were initiating methotrexate therapy.
Of the 204 patients, 167 had data sufficient for a 6-month analysis.
The investigators defined MDA after 6 months on methotrexate, the primary endpoint, as the presence of at least five out of the following seven domains: 0-1 tender joints, 0-1 swollen joints, Psoriasis Area Severity Index (PASI) 1 or less or body surface area involved 3% or less, 0-1 tender entheseal points, Health Assessment Questionnaire (HAQ) score of 0.5 or less, patient global disease activity Visual Analog Scale score of 20 or lower, and patient pain Visual Analog Scale score of 15 or lower.
They found that 17.4% of patients (29 of 167) met the physician-rated criteria for MDA at 6 months, even though 82.6% of the patients (138) had a PASI score of 1 or lower, and 58.1% (97) had a swollen joint count of 1 or fewer.
When the patients were asked for their assessment of global disease activity, 13.2% (22) rated disease activity with a prespecified score of 20 or lower. Only 12% of patients (20) reported an HAQ score of 0.5 or less.
The mean methotrexate dose was similar between patients who achieved MDA (17.8 mg/week) and those who did not (17.3 mg/week). The median dose was 17.5 mg/week in each group.
After controlling for sex, baseline sacroiliitis, and duration of psoriasis and psoriatic arthritis at the start of methotrexate in a multivariate model, the authors found that only dactylitis, inflammatory back pain, and mechanical back pain were significantly associated with a lower probability of patients reaching MDA. The variables that did not prove to be associated with achieving MDA after 6 months on methotrexate included erythrocyte sedimentation rate, presence of nail disease, number of clinically damaged joints, and body mass index.
The study was internally funded. Dr. Sheane reported having no relevant disclosures.
AT THE ACR ANNUAL MEETING
Key clinical point: It may be time to rethink methotrexate’s role as first-line therapy for psoriatic arthritis.
Major finding: Only 17.4% of patients with psoriatic arthritis treated with methotrexate achieved minimal disease activity at 6 months.
Data source: Retrospective study of 204 patients, 167 of whom had data sufficient for an efficacy and dosing analysis at 6 months.
Disclosures: The study was internally funded. Dr. Sheane reported having no relevant disclosures.
TNF-alpha blockers effective in sustaining reduced PsA activity
A state of minimal disease activity in patients with psoriatic arthritis is sustainable over several years by using treatments with tumor necrosis factor–alpha blockers, according to findings from a single-center, retrospective cohort study.
The study is the first to report on the predictors of minimal disease activity (MDA) in patients with psoriatic arthritis (PsA) taking TNF-alpha inhibitors in a clinical setting and “is also the first “to report about PsA patients who achieved and continued to be in MDA state after treatment change,” wrote the study investigators, who were led by Dr. Amir Haddad of the University of Toronto.
The investigators reported on 226 patients with PsA who were not in an MDA state when they presented to the University of Toronto and were treated with TNF-alpha inhibitors during 2000-2012. They had excluded 23 patients who had MDA when treatment began and 57 who were on TNF-alpha blockers prior to enrollment (Arthritis Care Res. 2014 Dec. 2 [doi:10.1002/acr.22529]).
The patients were 65% male with an average diagnosis age of 36.0 years and were followed at 6-12 month intervals. The authors defined MDA according to the criteria provided by Coates et al. (Ann. Rheum. Dis. 2010;69:48-53), which required patients to meet at least five of seven criteria: 0-1 tender joints ; 0-1 swollen joints; Psoriasis Activity and Severity Index of 1 or less or body surface area of 3 or less; patient pain visual analogue score (VAS) of 15 or less; patient global disease activity VAS of 20 or less; health assessment questionnaire of 0.5 or less; tender entheseal points of 1 or less.
A total of 145 patients (64%) achieved MDA status after an average of 1.30 months (standard deviation, 1.51), and 88 (61%) achieved sustained MDA (defined as at least 1 year) for a mean of 3.46 years (standard deviation, 2.25). Another 17 patients remained in an MDA state after reducing their anti–TNF-alpha drug dose, including 9 who withdrew anti–TNF-alpha treatment completely, and these patients remained in an MDA state for a mean of 2.11 years. The investigators noted that “no protocol was used for tapering the dose and it was left to patient’s preference,” and that MDA was sustained for longer periods of time in patients who reduced the TNF-alpha inhibitor dose and for shorter periods in patients who withdrew the treatment.
Male sex and a normal erythrocyte sedimentation rate (ESR) were found to be reliable predictors of achieving MDA. The authors cited previous studies on ankylosing spondylitis or axial spondyloarthritis, and noted that low ESR/C-reactive protein is considered a predictor for nonresponse to TNF-alpha blockers, but caution that the results of their own study may “simply [be] from using different outcome measures for response.”
The authors disclosed that all but one of them are members of the University of Toronto’s Psoriatic Arthritis Program, which is partly funded by the Arthritis Society, the Canadian Institutes of Health Research, and the Krembil Foundation. Additionally, Dr. Haddad disclosed that he was supported by unrestricted educational grants from Janssen and UCB.
A state of minimal disease activity in patients with psoriatic arthritis is sustainable over several years by using treatments with tumor necrosis factor–alpha blockers, according to findings from a single-center, retrospective cohort study.
The study is the first to report on the predictors of minimal disease activity (MDA) in patients with psoriatic arthritis (PsA) taking TNF-alpha inhibitors in a clinical setting and “is also the first “to report about PsA patients who achieved and continued to be in MDA state after treatment change,” wrote the study investigators, who were led by Dr. Amir Haddad of the University of Toronto.
The investigators reported on 226 patients with PsA who were not in an MDA state when they presented to the University of Toronto and were treated with TNF-alpha inhibitors during 2000-2012. They had excluded 23 patients who had MDA when treatment began and 57 who were on TNF-alpha blockers prior to enrollment (Arthritis Care Res. 2014 Dec. 2 [doi:10.1002/acr.22529]).
The patients were 65% male with an average diagnosis age of 36.0 years and were followed at 6-12 month intervals. The authors defined MDA according to the criteria provided by Coates et al. (Ann. Rheum. Dis. 2010;69:48-53), which required patients to meet at least five of seven criteria: 0-1 tender joints ; 0-1 swollen joints; Psoriasis Activity and Severity Index of 1 or less or body surface area of 3 or less; patient pain visual analogue score (VAS) of 15 or less; patient global disease activity VAS of 20 or less; health assessment questionnaire of 0.5 or less; tender entheseal points of 1 or less.
A total of 145 patients (64%) achieved MDA status after an average of 1.30 months (standard deviation, 1.51), and 88 (61%) achieved sustained MDA (defined as at least 1 year) for a mean of 3.46 years (standard deviation, 2.25). Another 17 patients remained in an MDA state after reducing their anti–TNF-alpha drug dose, including 9 who withdrew anti–TNF-alpha treatment completely, and these patients remained in an MDA state for a mean of 2.11 years. The investigators noted that “no protocol was used for tapering the dose and it was left to patient’s preference,” and that MDA was sustained for longer periods of time in patients who reduced the TNF-alpha inhibitor dose and for shorter periods in patients who withdrew the treatment.
Male sex and a normal erythrocyte sedimentation rate (ESR) were found to be reliable predictors of achieving MDA. The authors cited previous studies on ankylosing spondylitis or axial spondyloarthritis, and noted that low ESR/C-reactive protein is considered a predictor for nonresponse to TNF-alpha blockers, but caution that the results of their own study may “simply [be] from using different outcome measures for response.”
The authors disclosed that all but one of them are members of the University of Toronto’s Psoriatic Arthritis Program, which is partly funded by the Arthritis Society, the Canadian Institutes of Health Research, and the Krembil Foundation. Additionally, Dr. Haddad disclosed that he was supported by unrestricted educational grants from Janssen and UCB.
A state of minimal disease activity in patients with psoriatic arthritis is sustainable over several years by using treatments with tumor necrosis factor–alpha blockers, according to findings from a single-center, retrospective cohort study.
The study is the first to report on the predictors of minimal disease activity (MDA) in patients with psoriatic arthritis (PsA) taking TNF-alpha inhibitors in a clinical setting and “is also the first “to report about PsA patients who achieved and continued to be in MDA state after treatment change,” wrote the study investigators, who were led by Dr. Amir Haddad of the University of Toronto.
The investigators reported on 226 patients with PsA who were not in an MDA state when they presented to the University of Toronto and were treated with TNF-alpha inhibitors during 2000-2012. They had excluded 23 patients who had MDA when treatment began and 57 who were on TNF-alpha blockers prior to enrollment (Arthritis Care Res. 2014 Dec. 2 [doi:10.1002/acr.22529]).
The patients were 65% male with an average diagnosis age of 36.0 years and were followed at 6-12 month intervals. The authors defined MDA according to the criteria provided by Coates et al. (Ann. Rheum. Dis. 2010;69:48-53), which required patients to meet at least five of seven criteria: 0-1 tender joints ; 0-1 swollen joints; Psoriasis Activity and Severity Index of 1 or less or body surface area of 3 or less; patient pain visual analogue score (VAS) of 15 or less; patient global disease activity VAS of 20 or less; health assessment questionnaire of 0.5 or less; tender entheseal points of 1 or less.
A total of 145 patients (64%) achieved MDA status after an average of 1.30 months (standard deviation, 1.51), and 88 (61%) achieved sustained MDA (defined as at least 1 year) for a mean of 3.46 years (standard deviation, 2.25). Another 17 patients remained in an MDA state after reducing their anti–TNF-alpha drug dose, including 9 who withdrew anti–TNF-alpha treatment completely, and these patients remained in an MDA state for a mean of 2.11 years. The investigators noted that “no protocol was used for tapering the dose and it was left to patient’s preference,” and that MDA was sustained for longer periods of time in patients who reduced the TNF-alpha inhibitor dose and for shorter periods in patients who withdrew the treatment.
Male sex and a normal erythrocyte sedimentation rate (ESR) were found to be reliable predictors of achieving MDA. The authors cited previous studies on ankylosing spondylitis or axial spondyloarthritis, and noted that low ESR/C-reactive protein is considered a predictor for nonresponse to TNF-alpha blockers, but caution that the results of their own study may “simply [be] from using different outcome measures for response.”
The authors disclosed that all but one of them are members of the University of Toronto’s Psoriatic Arthritis Program, which is partly funded by the Arthritis Society, the Canadian Institutes of Health Research, and the Krembil Foundation. Additionally, Dr. Haddad disclosed that he was supported by unrestricted educational grants from Janssen and UCB.
FROM ARTHRITIS CARE & RESEARCH
Key clinical point: A majority of PsA patients seen in a clinical setting can achieve minimal disease activity on TNF-alpha inhibitors.
Major finding: A total of 64% of PsA patients taking TNF-alpha inhibitors achieved MDA within a mean duration of 1.30 years.
Data source: A retrospective, observational cohort study of 226 patients with PsA.
Disclosures: The authors reported several potential conflicts.
Psoriasis is independently associated with advanced liver fibrosis
AMSTERDAM – Older psoriasis patients have an increased risk of advanced hepatic fibrosis even if they have mild skin disease that has never required systemic therapy.
This finding from the Rotterdam Study raises a red flag: “It could be suggested to screen for liver fibrosis ... before and during potentially hepatotoxic therapies in psoriasis patients, especially in those with metabolic syndrome,” Dr. Ella van der Voort said in presenting the study findings at the annual congress of the European Academy of Dermatology and Venereology.
The Rotterdam Study is an ongoing large, prospective, population-based cohort study which began in 1998. Routine liver screening by FibroScan was incorporated into the study protocol in 2011. The proprietary FibroScan transient elastography device is noninvasive and yields results in 5 minutes, explained Dr. van der Voort, a dermatologist at Erasmus University, Rotterdam, the Netherlands.
In her analysis, Dr. van der Voort reported on 1,535 Rotterdam Study participants with FibroScan results, including 75 with psoriasis. The mean age was 71 years, both in the psoriasis patients and the 1,461 nonpsoriatic controls. The psoriasis patients had mild skin disease, with a mean Psoriasis Area and Severity Index score of 2.0. None of the psoriasis patients had ever received methotrexate or any other systemic therapy for their psoriasis.
The prevalence of advanced liver fibrosis was 3.6% in controls and 8.1% in the psoriasis patients. In a multivariate analysis adjusted for alcohol consumption, alanine aminotransferase levels, steatosis, metabolic syndrome, age, and gender, psoriasis was independently associated with a 2.6-fold increased risk of advanced liver fibrosis.
Among the subgroup of subjects with nonalcoholic fatty liver disease (NAFLD), psoriasis was associated with an even greater likelihood of advanced hepatic fibrosis. This liver finding was present in 15% of 20 patients with mild psoriasis and NAFLD, compared with 4% of 375 nonpsoriatic controls with NAFLD, for an adjusted 4.1-fold increased risk among the group with psoriasis.
Psoriasis patients with advanced liver fibrosis had a relatively high prevalence of the metabolic syndrome, so hepatic screening efforts could be focused on this subgroup, in Dr. van der Voort’s view.
In contrast, serum ALTs were generally within normal range in affected patients. “Using elevated ALTs to trigger screening for liver fibrosis in psoriasis patients is not good advice,” she cautioned.
Citing the advanced age of the Rotterdam Study cohort, Dr. van der Voort noted that her study findings may not be applicable to younger patients with mild psoriasis.
Dr. van der Voort presented another analysis from the Rotterdam Study at the 2013 EADV Congress in Istanbul. In that analysis, she reported that psoriasis was independently associated with a 70% increased risk of NAFLD. Since patients with NAFLD are at increased risk for progression to fibrosis and cirrhosis, the new finding of a 2.6-fold increased risk for advanced liver fibrosis in psoriasis patients doesn’t come as a total surprise. The biggest concern, of course, is that advanced liver fibrosis is, in turn, associated with an increased risk of hepatocellular carcinoma.
The Rotterdam Study is funded by Dutch governmental grants and foundations. Dr. van der Voort reported having no financial conflicts.
AMSTERDAM – Older psoriasis patients have an increased risk of advanced hepatic fibrosis even if they have mild skin disease that has never required systemic therapy.
This finding from the Rotterdam Study raises a red flag: “It could be suggested to screen for liver fibrosis ... before and during potentially hepatotoxic therapies in psoriasis patients, especially in those with metabolic syndrome,” Dr. Ella van der Voort said in presenting the study findings at the annual congress of the European Academy of Dermatology and Venereology.
The Rotterdam Study is an ongoing large, prospective, population-based cohort study which began in 1998. Routine liver screening by FibroScan was incorporated into the study protocol in 2011. The proprietary FibroScan transient elastography device is noninvasive and yields results in 5 minutes, explained Dr. van der Voort, a dermatologist at Erasmus University, Rotterdam, the Netherlands.
In her analysis, Dr. van der Voort reported on 1,535 Rotterdam Study participants with FibroScan results, including 75 with psoriasis. The mean age was 71 years, both in the psoriasis patients and the 1,461 nonpsoriatic controls. The psoriasis patients had mild skin disease, with a mean Psoriasis Area and Severity Index score of 2.0. None of the psoriasis patients had ever received methotrexate or any other systemic therapy for their psoriasis.
The prevalence of advanced liver fibrosis was 3.6% in controls and 8.1% in the psoriasis patients. In a multivariate analysis adjusted for alcohol consumption, alanine aminotransferase levels, steatosis, metabolic syndrome, age, and gender, psoriasis was independently associated with a 2.6-fold increased risk of advanced liver fibrosis.
Among the subgroup of subjects with nonalcoholic fatty liver disease (NAFLD), psoriasis was associated with an even greater likelihood of advanced hepatic fibrosis. This liver finding was present in 15% of 20 patients with mild psoriasis and NAFLD, compared with 4% of 375 nonpsoriatic controls with NAFLD, for an adjusted 4.1-fold increased risk among the group with psoriasis.
Psoriasis patients with advanced liver fibrosis had a relatively high prevalence of the metabolic syndrome, so hepatic screening efforts could be focused on this subgroup, in Dr. van der Voort’s view.
In contrast, serum ALTs were generally within normal range in affected patients. “Using elevated ALTs to trigger screening for liver fibrosis in psoriasis patients is not good advice,” she cautioned.
Citing the advanced age of the Rotterdam Study cohort, Dr. van der Voort noted that her study findings may not be applicable to younger patients with mild psoriasis.
Dr. van der Voort presented another analysis from the Rotterdam Study at the 2013 EADV Congress in Istanbul. In that analysis, she reported that psoriasis was independently associated with a 70% increased risk of NAFLD. Since patients with NAFLD are at increased risk for progression to fibrosis and cirrhosis, the new finding of a 2.6-fold increased risk for advanced liver fibrosis in psoriasis patients doesn’t come as a total surprise. The biggest concern, of course, is that advanced liver fibrosis is, in turn, associated with an increased risk of hepatocellular carcinoma.
The Rotterdam Study is funded by Dutch governmental grants and foundations. Dr. van der Voort reported having no financial conflicts.
AMSTERDAM – Older psoriasis patients have an increased risk of advanced hepatic fibrosis even if they have mild skin disease that has never required systemic therapy.
This finding from the Rotterdam Study raises a red flag: “It could be suggested to screen for liver fibrosis ... before and during potentially hepatotoxic therapies in psoriasis patients, especially in those with metabolic syndrome,” Dr. Ella van der Voort said in presenting the study findings at the annual congress of the European Academy of Dermatology and Venereology.
The Rotterdam Study is an ongoing large, prospective, population-based cohort study which began in 1998. Routine liver screening by FibroScan was incorporated into the study protocol in 2011. The proprietary FibroScan transient elastography device is noninvasive and yields results in 5 minutes, explained Dr. van der Voort, a dermatologist at Erasmus University, Rotterdam, the Netherlands.
In her analysis, Dr. van der Voort reported on 1,535 Rotterdam Study participants with FibroScan results, including 75 with psoriasis. The mean age was 71 years, both in the psoriasis patients and the 1,461 nonpsoriatic controls. The psoriasis patients had mild skin disease, with a mean Psoriasis Area and Severity Index score of 2.0. None of the psoriasis patients had ever received methotrexate or any other systemic therapy for their psoriasis.
The prevalence of advanced liver fibrosis was 3.6% in controls and 8.1% in the psoriasis patients. In a multivariate analysis adjusted for alcohol consumption, alanine aminotransferase levels, steatosis, metabolic syndrome, age, and gender, psoriasis was independently associated with a 2.6-fold increased risk of advanced liver fibrosis.
Among the subgroup of subjects with nonalcoholic fatty liver disease (NAFLD), psoriasis was associated with an even greater likelihood of advanced hepatic fibrosis. This liver finding was present in 15% of 20 patients with mild psoriasis and NAFLD, compared with 4% of 375 nonpsoriatic controls with NAFLD, for an adjusted 4.1-fold increased risk among the group with psoriasis.
Psoriasis patients with advanced liver fibrosis had a relatively high prevalence of the metabolic syndrome, so hepatic screening efforts could be focused on this subgroup, in Dr. van der Voort’s view.
In contrast, serum ALTs were generally within normal range in affected patients. “Using elevated ALTs to trigger screening for liver fibrosis in psoriasis patients is not good advice,” she cautioned.
Citing the advanced age of the Rotterdam Study cohort, Dr. van der Voort noted that her study findings may not be applicable to younger patients with mild psoriasis.
Dr. van der Voort presented another analysis from the Rotterdam Study at the 2013 EADV Congress in Istanbul. In that analysis, she reported that psoriasis was independently associated with a 70% increased risk of NAFLD. Since patients with NAFLD are at increased risk for progression to fibrosis and cirrhosis, the new finding of a 2.6-fold increased risk for advanced liver fibrosis in psoriasis patients doesn’t come as a total surprise. The biggest concern, of course, is that advanced liver fibrosis is, in turn, associated with an increased risk of hepatocellular carcinoma.
The Rotterdam Study is funded by Dutch governmental grants and foundations. Dr. van der Voort reported having no financial conflicts.
AT THE EADV CONGRESS
Key clinical point: Noninvasive screening for advanced hepatic fibrosis may be in order for older patients with mild psoriasis never treated systemically.
Major finding: In a multivariate analysis adjusted for alcohol consumption, alanine aminotransferase levels, steatosis, metabolic syndrome, age, and gender, psoriasis was independently associated with a 2.6-fold increased risk of advanced liver fibrosis.
Data source: This analysis involved noninvasive screening for hepatic fibrosis in 1,535 elderly participants in the Rotterdam Study, an ongoing prospective population-based cohort study.
Disclosures: The Rotterdam Study is funded by Dutch governmental research grants and foundations. The presenter reported having no financial conflicts.
Psoriasis: Brodalumab maintains efficacy through 144 weeks
AMSTERDAM – The majority of psoriasis patients placed on the investigational biologic agent brodalumab maintained a PASI 100 response throughout 144 weeks in a long-term, open-label extension of a phase II study.
“The benefit/risk ratio of brodalumab remains very favorable, and warrants continued development of this anti–interleukin-17 receptor A human monoclonal antibody as a potential treatment for psoriasis,” Dr. Kim Papp declared in presenting the study results at the annual congress of the European Academy of Dermatology and Venereology.
Large phase III clinical trials of brodalumab for the treatment of moderate to severe plaque psoriasis are ongoing.
The parent phase II study was 16 weeks long, double blinded, and placebo controlled. At the study’s end, 181 participants enrolled in the open-label extension, in which they received subcutaneous brodalumab at 210 mg every 2 weeks. The study protocol was amended after about a year, with the dose reduced to 140 mg every 2 weeks in the 119 patients weighing 100 kg or less. Six of those patients subsequently had an inadequate response to the lower dose, and were returned to the higher-dose regimen, explained Dr. Papp of Probity Medical Research in Waterloo, Ont.
Roughly 95% of patients became PASI 75 responders within the first few weeks. There was very little drop-off over time. The PASI 75 response rate was 95% at week 12, 93% at week 48, and 85% at week 144.
At week 144, roughly two-thirds of subjects were deemed clear or almost clear according to Physician Global Assessment.
No new safety signals emerged during this long-term study. The most common adverse events were minor upper respiratory infections. Two percent of patients developed a grade 2 absolute neutrophil count of less than 1,500 per 109/L; however, these were transitory events that resolved without a change in treatment.
In response to an audience question, Dr. Papp said antidrug antibodies did develop in some patients during the course of 144 weeks of treatment, but he had no information as to the clinical effect, if any.
“This is a fairly small study population. I think it makes sense to wait for the phase III results to see if the antibodies affect safety and/or efficacy,” he added.
Dr. Papp reported receiving research support from Amgen, which is codeveloping brodalumab with AstraZeneca/MedImmune.
AMSTERDAM – The majority of psoriasis patients placed on the investigational biologic agent brodalumab maintained a PASI 100 response throughout 144 weeks in a long-term, open-label extension of a phase II study.
“The benefit/risk ratio of brodalumab remains very favorable, and warrants continued development of this anti–interleukin-17 receptor A human monoclonal antibody as a potential treatment for psoriasis,” Dr. Kim Papp declared in presenting the study results at the annual congress of the European Academy of Dermatology and Venereology.
Large phase III clinical trials of brodalumab for the treatment of moderate to severe plaque psoriasis are ongoing.
The parent phase II study was 16 weeks long, double blinded, and placebo controlled. At the study’s end, 181 participants enrolled in the open-label extension, in which they received subcutaneous brodalumab at 210 mg every 2 weeks. The study protocol was amended after about a year, with the dose reduced to 140 mg every 2 weeks in the 119 patients weighing 100 kg or less. Six of those patients subsequently had an inadequate response to the lower dose, and were returned to the higher-dose regimen, explained Dr. Papp of Probity Medical Research in Waterloo, Ont.
Roughly 95% of patients became PASI 75 responders within the first few weeks. There was very little drop-off over time. The PASI 75 response rate was 95% at week 12, 93% at week 48, and 85% at week 144.
At week 144, roughly two-thirds of subjects were deemed clear or almost clear according to Physician Global Assessment.
No new safety signals emerged during this long-term study. The most common adverse events were minor upper respiratory infections. Two percent of patients developed a grade 2 absolute neutrophil count of less than 1,500 per 109/L; however, these were transitory events that resolved without a change in treatment.
In response to an audience question, Dr. Papp said antidrug antibodies did develop in some patients during the course of 144 weeks of treatment, but he had no information as to the clinical effect, if any.
“This is a fairly small study population. I think it makes sense to wait for the phase III results to see if the antibodies affect safety and/or efficacy,” he added.
Dr. Papp reported receiving research support from Amgen, which is codeveloping brodalumab with AstraZeneca/MedImmune.
AMSTERDAM – The majority of psoriasis patients placed on the investigational biologic agent brodalumab maintained a PASI 100 response throughout 144 weeks in a long-term, open-label extension of a phase II study.
“The benefit/risk ratio of brodalumab remains very favorable, and warrants continued development of this anti–interleukin-17 receptor A human monoclonal antibody as a potential treatment for psoriasis,” Dr. Kim Papp declared in presenting the study results at the annual congress of the European Academy of Dermatology and Venereology.
Large phase III clinical trials of brodalumab for the treatment of moderate to severe plaque psoriasis are ongoing.
The parent phase II study was 16 weeks long, double blinded, and placebo controlled. At the study’s end, 181 participants enrolled in the open-label extension, in which they received subcutaneous brodalumab at 210 mg every 2 weeks. The study protocol was amended after about a year, with the dose reduced to 140 mg every 2 weeks in the 119 patients weighing 100 kg or less. Six of those patients subsequently had an inadequate response to the lower dose, and were returned to the higher-dose regimen, explained Dr. Papp of Probity Medical Research in Waterloo, Ont.
Roughly 95% of patients became PASI 75 responders within the first few weeks. There was very little drop-off over time. The PASI 75 response rate was 95% at week 12, 93% at week 48, and 85% at week 144.
At week 144, roughly two-thirds of subjects were deemed clear or almost clear according to Physician Global Assessment.
No new safety signals emerged during this long-term study. The most common adverse events were minor upper respiratory infections. Two percent of patients developed a grade 2 absolute neutrophil count of less than 1,500 per 109/L; however, these were transitory events that resolved without a change in treatment.
In response to an audience question, Dr. Papp said antidrug antibodies did develop in some patients during the course of 144 weeks of treatment, but he had no information as to the clinical effect, if any.
“This is a fairly small study population. I think it makes sense to wait for the phase III results to see if the antibodies affect safety and/or efficacy,” he added.
Dr. Papp reported receiving research support from Amgen, which is codeveloping brodalumab with AstraZeneca/MedImmune.
AT THE EADV CONGRESS
Key clinical point: The investigational interleukin-17 receptor A inhibitor brodalumab maintained strong clinical efficacy throughout 144 weeks of psoriasis treatment.
Major finding: The PASI 90 response rate to brodalumab was 85% at week 12, 83% at week 48, and 74% at week 144.
Data source: A 181-patient, prospective, open-label extension of a phase II study.
Disclosures: Dr. Papp reported receiving financial support from Amgen, which is codeveloping brodalumab with AstraZeneca/MedImmune.
The Top 100
In the October 2014 issue of the Journal of Clinical and Aesthetic Dermatology (2014;7:10-19), Wu et al published the top 100 most-cited psoriasis articles in clinical dermatologic journals from 1970 to 2012. Given the explosion of literature in this area, I was very excited to rush to find the list online.
The authors conducted a citation analysis of major clinical dermatologic journals from 1970 to 2012 limited to the subject of psoriasis. They used the search term psoriasis in the Science Citation Index from 1970 to 2012 and included articles that have received 100 or more citations. The top 100 articles were further stratified by country, institution, and study type.
The authors found that half of the top 100 cited articles were from the United States; 81 of them were original articles. The majority of the top 100 articles were from dermatology programs in the United States, but institutions in the United Kingdom and Germany also made notable contributions.
There were 2 periods of particular note in their analysis. The high numbers of citations from 1985 to 1989 correlated with the elucidation of the immune-mediated pathogenesis of psoriasis at that time. The high number of top citations from 2000 to 2004 correlated with the development of biologic agents in psoriasis therapy.
What’s the issue?
It is interesting to see which studies have been most influential in this highly active area of dermatology. What articles have most influenced your approach to psoriasis?
In the October 2014 issue of the Journal of Clinical and Aesthetic Dermatology (2014;7:10-19), Wu et al published the top 100 most-cited psoriasis articles in clinical dermatologic journals from 1970 to 2012. Given the explosion of literature in this area, I was very excited to rush to find the list online.
The authors conducted a citation analysis of major clinical dermatologic journals from 1970 to 2012 limited to the subject of psoriasis. They used the search term psoriasis in the Science Citation Index from 1970 to 2012 and included articles that have received 100 or more citations. The top 100 articles were further stratified by country, institution, and study type.
The authors found that half of the top 100 cited articles were from the United States; 81 of them were original articles. The majority of the top 100 articles were from dermatology programs in the United States, but institutions in the United Kingdom and Germany also made notable contributions.
There were 2 periods of particular note in their analysis. The high numbers of citations from 1985 to 1989 correlated with the elucidation of the immune-mediated pathogenesis of psoriasis at that time. The high number of top citations from 2000 to 2004 correlated with the development of biologic agents in psoriasis therapy.
What’s the issue?
It is interesting to see which studies have been most influential in this highly active area of dermatology. What articles have most influenced your approach to psoriasis?
In the October 2014 issue of the Journal of Clinical and Aesthetic Dermatology (2014;7:10-19), Wu et al published the top 100 most-cited psoriasis articles in clinical dermatologic journals from 1970 to 2012. Given the explosion of literature in this area, I was very excited to rush to find the list online.
The authors conducted a citation analysis of major clinical dermatologic journals from 1970 to 2012 limited to the subject of psoriasis. They used the search term psoriasis in the Science Citation Index from 1970 to 2012 and included articles that have received 100 or more citations. The top 100 articles were further stratified by country, institution, and study type.
The authors found that half of the top 100 cited articles were from the United States; 81 of them were original articles. The majority of the top 100 articles were from dermatology programs in the United States, but institutions in the United Kingdom and Germany also made notable contributions.
There were 2 periods of particular note in their analysis. The high numbers of citations from 1985 to 1989 correlated with the elucidation of the immune-mediated pathogenesis of psoriasis at that time. The high number of top citations from 2000 to 2004 correlated with the development of biologic agents in psoriasis therapy.
What’s the issue?
It is interesting to see which studies have been most influential in this highly active area of dermatology. What articles have most influenced your approach to psoriasis?