User login
Certolizumab achieves sustained skin improvement in psoriatic arthritis
AMSTERDAM – Certolizumab pegol maintained significant improvement in dermatologic outcomes in psoriatic arthritis patients through 96 weeks of treatment in the phase III RAPID-PsA trial.
Moreover, the safety profile of this tumor necrosis factor (TNF) inhibitor was in line with findings from shorter-term studies, including the week 24 report from RAPID-PsA. Treatment-emergent adverse events were similar in type and frequency to those in placebo-treated controls, with the exception of an increased rate of minor upper respiratory tract infections. No cases of tuberculosis occurred.
“There were no new safety issues despite the increased exposure time out to 96 weeks,” Dr. Owen Davies reported at the annual congress of the European Academy of Dermatology and Venereology.
RAPID-PsA is an ongoing 216-week phase III study. It was double-blind and placebo-controlled through the first 24 weeks. The study started out with 409 psoriatic arthritis patients, half of whom had previously failed to response to one nonbiologic disease-modifying antirheumatic drug (DMARD), while the other half had been nonresponders to two or more. Of the 273 patients placed on certolizumab, 80% completed both 48 and 96 weeks of the study, explained Dr. Davies of UCB Pharma in Slough, England.
He focused on the dermatologic outcomes because the arthritis outcomes have previously been reported and served to support certolizumab’s regulatory approval for the treatment of psoriatic arthritis. The biologic is also approved for treatment of rheumatoid arthritis, ankylosing spondylitis, and Crohn’s disease. However, certolizumab’s durability of effect on the psoriatic skin manifestations of psoriatic arthritis hasn’t previously been addressed.
Briefly, at week 12 – the primary endpoint for the joint-related outcomes – 55% of patients achieved an ACR 20 response, compared with 24% on placebo. Moreover, 35% of certolizumab-treated patients had an ACR 50 response at that point, and 20% had an ACR 70 response. The ACR response rate in certolizumab-treated patients was similar regardless of whether or not they had previously been on another anti-TNF biologic.
Dr. Davies addressed in detail the dermatologic outcomes in the 166 psoriatic arthritis patients with at least 3% psoriasis body surface area involvement at baseline. They had an average 10-year disease duration, 24% body surface area involvement, and a baseline Psoriasis Area Severity Index (PASI) score of 12.0.
Dermatologic responses to certolizumab were comparable regardless of whether patients had been randomized to the biologic at 200 mg subcutaneously every 2 weeks or 400 mg once every 4 weeks. As was true for the joint-related responses to certolizumab, the skin responses were similar both in anti-TNF–naive and anti-TNF–experienced patients, he noted.
The PASI 75 response rate in this group of psoriatic arthritis patients with significant skin involvement was 61% at week 24, 65% at week 48, and 53% at week 96. The improvement was even greater in the 71 patients with more severe skin involvement as defined by a baseline PASI score of 10 or more. Certolizumab-treated patients also showed important improvements on the Physician Global Assessment and Dermatology Life Quality Index.
Certolizumab is a pegylated Fab’ fragment of a humanized TNF inhibitor monoclonal antibody.
“Certolizumab is structurally different from other currently available anti-TNF agents, which are either IgG1 monoclonal antibodies or, in the case of etanercept, a receptor fusion protein. Whether or not these structural differences will translate into clinical differences is a question being addressed in ongoing clinical trials,” Dr. Davies said.
The RAPID-PsA study is sponsored by UCB Pharma, where Dr. Davies is employed.
AMSTERDAM – Certolizumab pegol maintained significant improvement in dermatologic outcomes in psoriatic arthritis patients through 96 weeks of treatment in the phase III RAPID-PsA trial.
Moreover, the safety profile of this tumor necrosis factor (TNF) inhibitor was in line with findings from shorter-term studies, including the week 24 report from RAPID-PsA. Treatment-emergent adverse events were similar in type and frequency to those in placebo-treated controls, with the exception of an increased rate of minor upper respiratory tract infections. No cases of tuberculosis occurred.
“There were no new safety issues despite the increased exposure time out to 96 weeks,” Dr. Owen Davies reported at the annual congress of the European Academy of Dermatology and Venereology.
RAPID-PsA is an ongoing 216-week phase III study. It was double-blind and placebo-controlled through the first 24 weeks. The study started out with 409 psoriatic arthritis patients, half of whom had previously failed to response to one nonbiologic disease-modifying antirheumatic drug (DMARD), while the other half had been nonresponders to two or more. Of the 273 patients placed on certolizumab, 80% completed both 48 and 96 weeks of the study, explained Dr. Davies of UCB Pharma in Slough, England.
He focused on the dermatologic outcomes because the arthritis outcomes have previously been reported and served to support certolizumab’s regulatory approval for the treatment of psoriatic arthritis. The biologic is also approved for treatment of rheumatoid arthritis, ankylosing spondylitis, and Crohn’s disease. However, certolizumab’s durability of effect on the psoriatic skin manifestations of psoriatic arthritis hasn’t previously been addressed.
Briefly, at week 12 – the primary endpoint for the joint-related outcomes – 55% of patients achieved an ACR 20 response, compared with 24% on placebo. Moreover, 35% of certolizumab-treated patients had an ACR 50 response at that point, and 20% had an ACR 70 response. The ACR response rate in certolizumab-treated patients was similar regardless of whether or not they had previously been on another anti-TNF biologic.
Dr. Davies addressed in detail the dermatologic outcomes in the 166 psoriatic arthritis patients with at least 3% psoriasis body surface area involvement at baseline. They had an average 10-year disease duration, 24% body surface area involvement, and a baseline Psoriasis Area Severity Index (PASI) score of 12.0.
Dermatologic responses to certolizumab were comparable regardless of whether patients had been randomized to the biologic at 200 mg subcutaneously every 2 weeks or 400 mg once every 4 weeks. As was true for the joint-related responses to certolizumab, the skin responses were similar both in anti-TNF–naive and anti-TNF–experienced patients, he noted.
The PASI 75 response rate in this group of psoriatic arthritis patients with significant skin involvement was 61% at week 24, 65% at week 48, and 53% at week 96. The improvement was even greater in the 71 patients with more severe skin involvement as defined by a baseline PASI score of 10 or more. Certolizumab-treated patients also showed important improvements on the Physician Global Assessment and Dermatology Life Quality Index.
Certolizumab is a pegylated Fab’ fragment of a humanized TNF inhibitor monoclonal antibody.
“Certolizumab is structurally different from other currently available anti-TNF agents, which are either IgG1 monoclonal antibodies or, in the case of etanercept, a receptor fusion protein. Whether or not these structural differences will translate into clinical differences is a question being addressed in ongoing clinical trials,” Dr. Davies said.
The RAPID-PsA study is sponsored by UCB Pharma, where Dr. Davies is employed.
AMSTERDAM – Certolizumab pegol maintained significant improvement in dermatologic outcomes in psoriatic arthritis patients through 96 weeks of treatment in the phase III RAPID-PsA trial.
Moreover, the safety profile of this tumor necrosis factor (TNF) inhibitor was in line with findings from shorter-term studies, including the week 24 report from RAPID-PsA. Treatment-emergent adverse events were similar in type and frequency to those in placebo-treated controls, with the exception of an increased rate of minor upper respiratory tract infections. No cases of tuberculosis occurred.
“There were no new safety issues despite the increased exposure time out to 96 weeks,” Dr. Owen Davies reported at the annual congress of the European Academy of Dermatology and Venereology.
RAPID-PsA is an ongoing 216-week phase III study. It was double-blind and placebo-controlled through the first 24 weeks. The study started out with 409 psoriatic arthritis patients, half of whom had previously failed to response to one nonbiologic disease-modifying antirheumatic drug (DMARD), while the other half had been nonresponders to two or more. Of the 273 patients placed on certolizumab, 80% completed both 48 and 96 weeks of the study, explained Dr. Davies of UCB Pharma in Slough, England.
He focused on the dermatologic outcomes because the arthritis outcomes have previously been reported and served to support certolizumab’s regulatory approval for the treatment of psoriatic arthritis. The biologic is also approved for treatment of rheumatoid arthritis, ankylosing spondylitis, and Crohn’s disease. However, certolizumab’s durability of effect on the psoriatic skin manifestations of psoriatic arthritis hasn’t previously been addressed.
Briefly, at week 12 – the primary endpoint for the joint-related outcomes – 55% of patients achieved an ACR 20 response, compared with 24% on placebo. Moreover, 35% of certolizumab-treated patients had an ACR 50 response at that point, and 20% had an ACR 70 response. The ACR response rate in certolizumab-treated patients was similar regardless of whether or not they had previously been on another anti-TNF biologic.
Dr. Davies addressed in detail the dermatologic outcomes in the 166 psoriatic arthritis patients with at least 3% psoriasis body surface area involvement at baseline. They had an average 10-year disease duration, 24% body surface area involvement, and a baseline Psoriasis Area Severity Index (PASI) score of 12.0.
Dermatologic responses to certolizumab were comparable regardless of whether patients had been randomized to the biologic at 200 mg subcutaneously every 2 weeks or 400 mg once every 4 weeks. As was true for the joint-related responses to certolizumab, the skin responses were similar both in anti-TNF–naive and anti-TNF–experienced patients, he noted.
The PASI 75 response rate in this group of psoriatic arthritis patients with significant skin involvement was 61% at week 24, 65% at week 48, and 53% at week 96. The improvement was even greater in the 71 patients with more severe skin involvement as defined by a baseline PASI score of 10 or more. Certolizumab-treated patients also showed important improvements on the Physician Global Assessment and Dermatology Life Quality Index.
Certolizumab is a pegylated Fab’ fragment of a humanized TNF inhibitor monoclonal antibody.
“Certolizumab is structurally different from other currently available anti-TNF agents, which are either IgG1 monoclonal antibodies or, in the case of etanercept, a receptor fusion protein. Whether or not these structural differences will translate into clinical differences is a question being addressed in ongoing clinical trials,” Dr. Davies said.
The RAPID-PsA study is sponsored by UCB Pharma, where Dr. Davies is employed.
AT THE EADV CONGRESS
Key clinical point: Certolizumab pegol maintains sustained improvement in the dermatologic manifestations of psoriatic arthritis through 96 weeks.
Major finding: Sixty-two percent of psoriatic arthritis patients with at least 3% body surface area involvement at a baseline PASI score of 10 or more still had a PASI 75 dermatologic response after 96 weeks on certolizumab.
Data source: The RAPID-PsA study is an ongoing 216-week, prospective, randomized, multicenter trial involving 409 psoriatic arthritis patients, including 166 with significant skin involvement at baseline.
Disclosures: The study is sponsored by UCB Pharma. The presenter is a full-time company employee.
Oral curcumin shown effective in psoriasis
AMSTERDAM – Oral curcumin proved safe and effective as adjunctive therapy in patients on topical corticosteroids for mild to moderate psoriasis vulgaris in a 12-week, randomized, placebo-controlled clinical trial.
This agent helps fill an unmet need in psoriasis, Dr. Emiliano Antiga observed in presenting the study findings at the annual congress of the European Academy of Dermatology and Venereology.
That’s because by far most of the action in the development of new treatments for psoriasis focuses on biologics and other extremely costly agents targeting patients at the moderate to severe end of the disease spectrum. But psoriasis is a chronic condition, and the many patients with milder disease require long-term therapies that are nontoxic and won’t break the bank. Enter curcumin.
“Oral curcumin is effective, safe, and it is cheap,” declared Dr. Antiga, a dermatologist at the University of Florence (Italy).
Moreover, it has a biologically plausible mechanism of benefit in psoriasis, as was demonstrated in his 60-patient randomized trial. Serum levels of the proinflammatory cytokine interleukin-22 were cut in half in the group assigned to 12 weeks of daily oral curcumin while remaining unchanged in the control group.
Curcumin is derived from turmeric, the dried rhizome of a plant, Curcuma longa. Turmeric is a yellowish Indian spice used in curries. But curcumin has long been used therapeutically in traditional Indian and Chinese medicine. Studies have shown that curcumin has antiproliferative, antiangiogenic, and anti-inflammatory effects. In a small study by other investigators, topical turmeric not only successfully cleared psoriasis lesions, it also suppressed phosphorylase kinase activity, which is important to keratinocyte proliferation (Br. J. Dermatol. 2000;143:937-49).
Dr. Antiga presented a study of 60 patients with mild to moderate psoriasis vulgaris as defined by a baseline median Psoriasis Area and Severity Index (PASI) score of 5.5 who were randomly assigned to 12 weeks of treatment with topical corticosteroids plus 3 g per day of oral curcumin or topical steroids plus placebo. The active-treatment capsules contained curcumin embedded in nanoparticle liposomes to enhance bioavailability.
Forty-nine patients completed the study. The primary endpoint was reduction in PASI values over 12 weeks. Both groups showed improvement – after all, the controls were on active treatment with topical steroids – but the change in PASI scores was significantly greater in the curcumin-treated patients.
Adverse events in the curcumin group were limited to one case of diarrhea. There was one case of nausea and one papular eruption in the control group.
Although IL-22 levels at 12 weeks were halved in the curcumin group and unchanged in controls, levels of the inflammatory cytokines IL-10 and -17 and transforming growth factor–beta remained unchanged in both groups over time.
Dr. Antiga reported having no financial conflicts regarding this study.
AMSTERDAM – Oral curcumin proved safe and effective as adjunctive therapy in patients on topical corticosteroids for mild to moderate psoriasis vulgaris in a 12-week, randomized, placebo-controlled clinical trial.
This agent helps fill an unmet need in psoriasis, Dr. Emiliano Antiga observed in presenting the study findings at the annual congress of the European Academy of Dermatology and Venereology.
That’s because by far most of the action in the development of new treatments for psoriasis focuses on biologics and other extremely costly agents targeting patients at the moderate to severe end of the disease spectrum. But psoriasis is a chronic condition, and the many patients with milder disease require long-term therapies that are nontoxic and won’t break the bank. Enter curcumin.
“Oral curcumin is effective, safe, and it is cheap,” declared Dr. Antiga, a dermatologist at the University of Florence (Italy).
Moreover, it has a biologically plausible mechanism of benefit in psoriasis, as was demonstrated in his 60-patient randomized trial. Serum levels of the proinflammatory cytokine interleukin-22 were cut in half in the group assigned to 12 weeks of daily oral curcumin while remaining unchanged in the control group.
Curcumin is derived from turmeric, the dried rhizome of a plant, Curcuma longa. Turmeric is a yellowish Indian spice used in curries. But curcumin has long been used therapeutically in traditional Indian and Chinese medicine. Studies have shown that curcumin has antiproliferative, antiangiogenic, and anti-inflammatory effects. In a small study by other investigators, topical turmeric not only successfully cleared psoriasis lesions, it also suppressed phosphorylase kinase activity, which is important to keratinocyte proliferation (Br. J. Dermatol. 2000;143:937-49).
Dr. Antiga presented a study of 60 patients with mild to moderate psoriasis vulgaris as defined by a baseline median Psoriasis Area and Severity Index (PASI) score of 5.5 who were randomly assigned to 12 weeks of treatment with topical corticosteroids plus 3 g per day of oral curcumin or topical steroids plus placebo. The active-treatment capsules contained curcumin embedded in nanoparticle liposomes to enhance bioavailability.
Forty-nine patients completed the study. The primary endpoint was reduction in PASI values over 12 weeks. Both groups showed improvement – after all, the controls were on active treatment with topical steroids – but the change in PASI scores was significantly greater in the curcumin-treated patients.
Adverse events in the curcumin group were limited to one case of diarrhea. There was one case of nausea and one papular eruption in the control group.
Although IL-22 levels at 12 weeks were halved in the curcumin group and unchanged in controls, levels of the inflammatory cytokines IL-10 and -17 and transforming growth factor–beta remained unchanged in both groups over time.
Dr. Antiga reported having no financial conflicts regarding this study.
AMSTERDAM – Oral curcumin proved safe and effective as adjunctive therapy in patients on topical corticosteroids for mild to moderate psoriasis vulgaris in a 12-week, randomized, placebo-controlled clinical trial.
This agent helps fill an unmet need in psoriasis, Dr. Emiliano Antiga observed in presenting the study findings at the annual congress of the European Academy of Dermatology and Venereology.
That’s because by far most of the action in the development of new treatments for psoriasis focuses on biologics and other extremely costly agents targeting patients at the moderate to severe end of the disease spectrum. But psoriasis is a chronic condition, and the many patients with milder disease require long-term therapies that are nontoxic and won’t break the bank. Enter curcumin.
“Oral curcumin is effective, safe, and it is cheap,” declared Dr. Antiga, a dermatologist at the University of Florence (Italy).
Moreover, it has a biologically plausible mechanism of benefit in psoriasis, as was demonstrated in his 60-patient randomized trial. Serum levels of the proinflammatory cytokine interleukin-22 were cut in half in the group assigned to 12 weeks of daily oral curcumin while remaining unchanged in the control group.
Curcumin is derived from turmeric, the dried rhizome of a plant, Curcuma longa. Turmeric is a yellowish Indian spice used in curries. But curcumin has long been used therapeutically in traditional Indian and Chinese medicine. Studies have shown that curcumin has antiproliferative, antiangiogenic, and anti-inflammatory effects. In a small study by other investigators, topical turmeric not only successfully cleared psoriasis lesions, it also suppressed phosphorylase kinase activity, which is important to keratinocyte proliferation (Br. J. Dermatol. 2000;143:937-49).
Dr. Antiga presented a study of 60 patients with mild to moderate psoriasis vulgaris as defined by a baseline median Psoriasis Area and Severity Index (PASI) score of 5.5 who were randomly assigned to 12 weeks of treatment with topical corticosteroids plus 3 g per day of oral curcumin or topical steroids plus placebo. The active-treatment capsules contained curcumin embedded in nanoparticle liposomes to enhance bioavailability.
Forty-nine patients completed the study. The primary endpoint was reduction in PASI values over 12 weeks. Both groups showed improvement – after all, the controls were on active treatment with topical steroids – but the change in PASI scores was significantly greater in the curcumin-treated patients.
Adverse events in the curcumin group were limited to one case of diarrhea. There was one case of nausea and one papular eruption in the control group.
Although IL-22 levels at 12 weeks were halved in the curcumin group and unchanged in controls, levels of the inflammatory cytokines IL-10 and -17 and transforming growth factor–beta remained unchanged in both groups over time.
Dr. Antiga reported having no financial conflicts regarding this study.
AT THE EADV CONGRESS
Key clinical point: A common ingredient in Indian curry spice mixes is safe and effective as adjuvant therapy for mild-to-moderate psoriasis.
Major finding: Daily oral curcumin capsules plus topical corticosteroids resulted in a PASI 75 improvement rate of 48% compared with a 12% rate in patients who got topical steroids plus placebo.
Data source: A prospective randomized 12-week clinical trial involving 60 patients with mild to moderate psoriasis vulgaris.
Disclosures: The presenter reported no financial conflicts regarding this study, conducted free of commercial support.
Chronic inflammatory disease patients at greater risk of major CV events
Patients with rheumatoid arthritis, psoriatic arthritis, or psoriasis are at an increased risk of major adverse cardiovascular events when compared with the general population, according to findings from a large cohort study.
All three diseases had statistically similar risks for major adverse cardiovascular events (MACE) after adjustment for age, gender, and traditional CV risk factors, Dr. Alexis Ogdie-Beatty of the University of Pennsylvania, Philadelphia, and her colleagues reported (Ann. Rheum. Dis. 2014 Oct. 30 [doi: 10.1136/annrheumdis-2014-205675]).
The investigators noted that most studies of CV risk in psoriatic arthritis (PsA) patients have been cross-sectional and that three previous population-based cohort studies have evaluated CV risk in psoriasis patients, with PsA patients as a subgroup; however, these three psoriasis studies did not include incident MACE and matched internal control patients with adjustments for traditional CV risk factors.
The investigators used data from the Health Improvement Network, a U.K. primary care medical record database, and compared the number of MACE (myocardial infarction, cerebrovascular accident, and CV death) that occurred during a mean 5 years of follow-up in 41,752 patients with rheumatoid arthritis (RA), 8,706 with PsA, 138,424 with psoriasis, and 81,573 matched controls. There was significant interaction between disease-modifying antirheumatic drug (DMARD) use and disease group (P < .001 for MACE and two components, CV death and cerebrovascular accident; and P = .01 for MI).
The risk of MACE was higher in patients with PsA not prescribed a DMARD (hazard ratio, 1.24; 95% confidence interval, 1.03-1.49). This risk was elevated in RA patients both with DMARD prescriptions (HR, 1.58; 95% CI, 1.46-1.70) and without (HR, 1.39; 95% CI, 1.28-1.50). Patients with severe psoriasis who were prescribed a DMARD had an HR of 1.42 (95% CI, 1.17-1.73), whereas psoriasis patients not prescribed a DMARD had an HR of 1.08 (95% CI, 1.02-1.15).
The results highlight a need for improved screening and management of traditional CV risk factors in patients with inflammatory diseases, the researchers said.
Study limitations included not being able to measure disease severity or the use of over-the-counter NSAIDs, as well as having few records on biologic medications and possibly missing DMARD prescriptions.
The researchers were supported by various grants from the Rheumatology Research Foundation, the National Institutes of Health, the Doris Duke Charitable Foundation, and the Icelandic Research Fund. Several authors reported financial relationships with companies that market drugs for chronic inflammatory diseases.
Patients with rheumatoid arthritis, psoriatic arthritis, or psoriasis are at an increased risk of major adverse cardiovascular events when compared with the general population, according to findings from a large cohort study.
All three diseases had statistically similar risks for major adverse cardiovascular events (MACE) after adjustment for age, gender, and traditional CV risk factors, Dr. Alexis Ogdie-Beatty of the University of Pennsylvania, Philadelphia, and her colleagues reported (Ann. Rheum. Dis. 2014 Oct. 30 [doi: 10.1136/annrheumdis-2014-205675]).
The investigators noted that most studies of CV risk in psoriatic arthritis (PsA) patients have been cross-sectional and that three previous population-based cohort studies have evaluated CV risk in psoriasis patients, with PsA patients as a subgroup; however, these three psoriasis studies did not include incident MACE and matched internal control patients with adjustments for traditional CV risk factors.
The investigators used data from the Health Improvement Network, a U.K. primary care medical record database, and compared the number of MACE (myocardial infarction, cerebrovascular accident, and CV death) that occurred during a mean 5 years of follow-up in 41,752 patients with rheumatoid arthritis (RA), 8,706 with PsA, 138,424 with psoriasis, and 81,573 matched controls. There was significant interaction between disease-modifying antirheumatic drug (DMARD) use and disease group (P < .001 for MACE and two components, CV death and cerebrovascular accident; and P = .01 for MI).
The risk of MACE was higher in patients with PsA not prescribed a DMARD (hazard ratio, 1.24; 95% confidence interval, 1.03-1.49). This risk was elevated in RA patients both with DMARD prescriptions (HR, 1.58; 95% CI, 1.46-1.70) and without (HR, 1.39; 95% CI, 1.28-1.50). Patients with severe psoriasis who were prescribed a DMARD had an HR of 1.42 (95% CI, 1.17-1.73), whereas psoriasis patients not prescribed a DMARD had an HR of 1.08 (95% CI, 1.02-1.15).
The results highlight a need for improved screening and management of traditional CV risk factors in patients with inflammatory diseases, the researchers said.
Study limitations included not being able to measure disease severity or the use of over-the-counter NSAIDs, as well as having few records on biologic medications and possibly missing DMARD prescriptions.
The researchers were supported by various grants from the Rheumatology Research Foundation, the National Institutes of Health, the Doris Duke Charitable Foundation, and the Icelandic Research Fund. Several authors reported financial relationships with companies that market drugs for chronic inflammatory diseases.
Patients with rheumatoid arthritis, psoriatic arthritis, or psoriasis are at an increased risk of major adverse cardiovascular events when compared with the general population, according to findings from a large cohort study.
All three diseases had statistically similar risks for major adverse cardiovascular events (MACE) after adjustment for age, gender, and traditional CV risk factors, Dr. Alexis Ogdie-Beatty of the University of Pennsylvania, Philadelphia, and her colleagues reported (Ann. Rheum. Dis. 2014 Oct. 30 [doi: 10.1136/annrheumdis-2014-205675]).
The investigators noted that most studies of CV risk in psoriatic arthritis (PsA) patients have been cross-sectional and that three previous population-based cohort studies have evaluated CV risk in psoriasis patients, with PsA patients as a subgroup; however, these three psoriasis studies did not include incident MACE and matched internal control patients with adjustments for traditional CV risk factors.
The investigators used data from the Health Improvement Network, a U.K. primary care medical record database, and compared the number of MACE (myocardial infarction, cerebrovascular accident, and CV death) that occurred during a mean 5 years of follow-up in 41,752 patients with rheumatoid arthritis (RA), 8,706 with PsA, 138,424 with psoriasis, and 81,573 matched controls. There was significant interaction between disease-modifying antirheumatic drug (DMARD) use and disease group (P < .001 for MACE and two components, CV death and cerebrovascular accident; and P = .01 for MI).
The risk of MACE was higher in patients with PsA not prescribed a DMARD (hazard ratio, 1.24; 95% confidence interval, 1.03-1.49). This risk was elevated in RA patients both with DMARD prescriptions (HR, 1.58; 95% CI, 1.46-1.70) and without (HR, 1.39; 95% CI, 1.28-1.50). Patients with severe psoriasis who were prescribed a DMARD had an HR of 1.42 (95% CI, 1.17-1.73), whereas psoriasis patients not prescribed a DMARD had an HR of 1.08 (95% CI, 1.02-1.15).
The results highlight a need for improved screening and management of traditional CV risk factors in patients with inflammatory diseases, the researchers said.
Study limitations included not being able to measure disease severity or the use of over-the-counter NSAIDs, as well as having few records on biologic medications and possibly missing DMARD prescriptions.
The researchers were supported by various grants from the Rheumatology Research Foundation, the National Institutes of Health, the Doris Duke Charitable Foundation, and the Icelandic Research Fund. Several authors reported financial relationships with companies that market drugs for chronic inflammatory diseases.
FROM ANNALS OF THE RHEUMATIC DISEASES
Key clinical point: Patients with RA, PsA, and psoriasis have similarly elevated risk for major adverse cardiovascular events when compared with the general population.
Major finding: The risk of MACE was higher in patients with PsA not prescribed a DMARD (hazard ratio, 1.24; 95% confidence interval, 1.03-1.49), compared with matched controls.
Data source: A population-based, longitudinal cohort study of 41,752 RA patients, 8,706 PsA patients, 138,424 psoriasis patients, and 81,573 matched controls.
Disclosures: The researchers were supported by various grants from the Rheumatology Research Foundation, the National Institutes of Health, the Doris Duke Charitable Foundation, and the Icelandic Research Fund. Several authors reported financial relationships with companies that market drugs for chronic inflammatory diseases.
Recent Findings About Plaque Psoriasis
More Evidence Needed on Combined Use of Systemic Agents for Plaque Psoriasis
To evaluate the combined use of systemic agents to achieve disease control in therapy-resistant patients with plaque-type psoriasis, Busard et al (JAMA Dermatol. doi:10.1001/jamadermatol.2014.1111) conducted an electronic search of randomized clinical trials. They found that most studies favored combination therapy, but the significance level and quality of evidence was low. Etanercept plus methotrexate was investigated with an adequate sample size and demonstrated superior efficacy compared to etanercept monotherapy. Although it was associated with an increase in adverse events, the overall safety profile was acceptable.
Practice Point: Patients with plaque psoriasis may benefit from combination therapy with systemic agents, but there is insufficient evidence. Etanercept plus methotrexate may be beneficial, with dose reductions to minimize adverse effects.
>>Read more at JAMA Dermatology
Plaque Psoriasis Familial Risk Highest in First-Degree Relatives
Psoriasis is known to be a highly heritable disease. In a cross-sectional study of 640 consecutive, unrelated adult patients with chronic plaque psoriasis, Di Lernia et al (Clin Exp Dermatol. 2014;39:801-805) found a positive familial history of psoriasis in more than half (59.37%) of patients. Of these patients, 27.18% had at least 1 parent with psoriasis, 16.56% had at least 1 second-degree relative with psoriasis, and 5.31% had 1 third-degree relative with psoriasis.
Practice Point: There is a high familial recurrence risk for plaque psoriasis.
>>Read more at Clinical and Experimental Dermatology
Secukinumab Efficacy for Plaque Psoriasis
Two phase 3, double-blind, 52-week trials (ERASURE and FIXTURE) evaluated subcutaneous secukinumab at a dose of 300 or 150 mg (administed once weekly for 5 weeks, then every 4 weeks), placebo, or (in the FIXTURE study only) etanercept at a dose of 50 mg (administered twice weekly for 12 weeks, then once weekly) in patients with moderate to severe plaque psoriasis. Langley et al, in conjunction with the ERASURE Study Group and FIXTURE Study Group, reported (N Engl J Med. 2014;371:326-338) that the proportion of patients who met the criterion for a reduction of 75% or more from baseline in the psoriasis area and severity index score (PASI 75) at week 12 was higher with each secukinumab dose than with placebo or etanercept. The proportion of patients with a response of 0 (clear) or 1 (almost clear) on the modified investigator global assessment at week 12 was higher with each secukinumab dose than with placebo or etanercept.
This study was funded by Novartis Pharmaceuticals.
Practice Point: Efficacy of secukinumab has been documented in randomized trials of patients with moderate to severe plaque psoriasis, validating IL-17A as a therapeutic target.
>>Read more at the New England Journal of Medicine
More Evidence Needed on Combined Use of Systemic Agents for Plaque Psoriasis
To evaluate the combined use of systemic agents to achieve disease control in therapy-resistant patients with plaque-type psoriasis, Busard et al (JAMA Dermatol. doi:10.1001/jamadermatol.2014.1111) conducted an electronic search of randomized clinical trials. They found that most studies favored combination therapy, but the significance level and quality of evidence was low. Etanercept plus methotrexate was investigated with an adequate sample size and demonstrated superior efficacy compared to etanercept monotherapy. Although it was associated with an increase in adverse events, the overall safety profile was acceptable.
Practice Point: Patients with plaque psoriasis may benefit from combination therapy with systemic agents, but there is insufficient evidence. Etanercept plus methotrexate may be beneficial, with dose reductions to minimize adverse effects.
>>Read more at JAMA Dermatology
Plaque Psoriasis Familial Risk Highest in First-Degree Relatives
Psoriasis is known to be a highly heritable disease. In a cross-sectional study of 640 consecutive, unrelated adult patients with chronic plaque psoriasis, Di Lernia et al (Clin Exp Dermatol. 2014;39:801-805) found a positive familial history of psoriasis in more than half (59.37%) of patients. Of these patients, 27.18% had at least 1 parent with psoriasis, 16.56% had at least 1 second-degree relative with psoriasis, and 5.31% had 1 third-degree relative with psoriasis.
Practice Point: There is a high familial recurrence risk for plaque psoriasis.
>>Read more at Clinical and Experimental Dermatology
Secukinumab Efficacy for Plaque Psoriasis
Two phase 3, double-blind, 52-week trials (ERASURE and FIXTURE) evaluated subcutaneous secukinumab at a dose of 300 or 150 mg (administed once weekly for 5 weeks, then every 4 weeks), placebo, or (in the FIXTURE study only) etanercept at a dose of 50 mg (administered twice weekly for 12 weeks, then once weekly) in patients with moderate to severe plaque psoriasis. Langley et al, in conjunction with the ERASURE Study Group and FIXTURE Study Group, reported (N Engl J Med. 2014;371:326-338) that the proportion of patients who met the criterion for a reduction of 75% or more from baseline in the psoriasis area and severity index score (PASI 75) at week 12 was higher with each secukinumab dose than with placebo or etanercept. The proportion of patients with a response of 0 (clear) or 1 (almost clear) on the modified investigator global assessment at week 12 was higher with each secukinumab dose than with placebo or etanercept.
This study was funded by Novartis Pharmaceuticals.
Practice Point: Efficacy of secukinumab has been documented in randomized trials of patients with moderate to severe plaque psoriasis, validating IL-17A as a therapeutic target.
>>Read more at the New England Journal of Medicine
More Evidence Needed on Combined Use of Systemic Agents for Plaque Psoriasis
To evaluate the combined use of systemic agents to achieve disease control in therapy-resistant patients with plaque-type psoriasis, Busard et al (JAMA Dermatol. doi:10.1001/jamadermatol.2014.1111) conducted an electronic search of randomized clinical trials. They found that most studies favored combination therapy, but the significance level and quality of evidence was low. Etanercept plus methotrexate was investigated with an adequate sample size and demonstrated superior efficacy compared to etanercept monotherapy. Although it was associated with an increase in adverse events, the overall safety profile was acceptable.
Practice Point: Patients with plaque psoriasis may benefit from combination therapy with systemic agents, but there is insufficient evidence. Etanercept plus methotrexate may be beneficial, with dose reductions to minimize adverse effects.
>>Read more at JAMA Dermatology
Plaque Psoriasis Familial Risk Highest in First-Degree Relatives
Psoriasis is known to be a highly heritable disease. In a cross-sectional study of 640 consecutive, unrelated adult patients with chronic plaque psoriasis, Di Lernia et al (Clin Exp Dermatol. 2014;39:801-805) found a positive familial history of psoriasis in more than half (59.37%) of patients. Of these patients, 27.18% had at least 1 parent with psoriasis, 16.56% had at least 1 second-degree relative with psoriasis, and 5.31% had 1 third-degree relative with psoriasis.
Practice Point: There is a high familial recurrence risk for plaque psoriasis.
>>Read more at Clinical and Experimental Dermatology
Secukinumab Efficacy for Plaque Psoriasis
Two phase 3, double-blind, 52-week trials (ERASURE and FIXTURE) evaluated subcutaneous secukinumab at a dose of 300 or 150 mg (administed once weekly for 5 weeks, then every 4 weeks), placebo, or (in the FIXTURE study only) etanercept at a dose of 50 mg (administered twice weekly for 12 weeks, then once weekly) in patients with moderate to severe plaque psoriasis. Langley et al, in conjunction with the ERASURE Study Group and FIXTURE Study Group, reported (N Engl J Med. 2014;371:326-338) that the proportion of patients who met the criterion for a reduction of 75% or more from baseline in the psoriasis area and severity index score (PASI 75) at week 12 was higher with each secukinumab dose than with placebo or etanercept. The proportion of patients with a response of 0 (clear) or 1 (almost clear) on the modified investigator global assessment at week 12 was higher with each secukinumab dose than with placebo or etanercept.
This study was funded by Novartis Pharmaceuticals.
Practice Point: Efficacy of secukinumab has been documented in randomized trials of patients with moderate to severe plaque psoriasis, validating IL-17A as a therapeutic target.
>>Read more at the New England Journal of Medicine
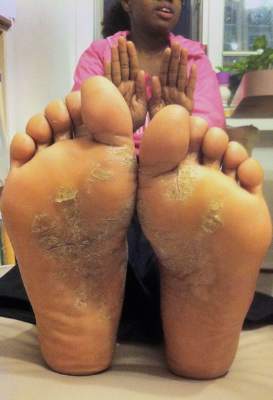
Apremilast succeeds against nail, scalp, palmoplantar psoriasis
LAS VEGAS – A twice-daily 30-mg dose of apremilast significantly improved moderate to severe nail, scalp, and palmoplantar psoriasis, compared with placebo, based on data from 411 adults in the ESTEEM 2 phase III study.
Overall, at 16 weeks, 29% of the patients randomized to apremilast achieved PASI-75, and 56% achieved PASI-50, compared with placebo patients (6% and 20%, respectively). The findings were presented in a poster at SDEF Las Vegas Dermatology Seminar by Dr. Jeffrey Crowley of Bakersfield (Calif.) Dermatology, and his colleagues.
In addition, patients with palmarplantar, nail, and scalp psoriasis who received apremilast showed significantly greater response rates, compared with placebo patients.
At baseline, 266 patients had scores of 1 or greater on the Nail Psoriasis Severity Index (NAPSI). Significantly more of these patients on apremilast achieved NAPSI-50 vs. placebo patients after 16 weeks (45% vs. 19%, respectively). The average percent change in NAPSI score from baseline was 29% for apremilast vs. 7% for placebo.
“Among patients initially randomized to apremilast and maintained on apremilast through week 32, nail psoriasis was further improved at week 32 (55% of patients achieved NAPSI-50)” and improvements were seen at week 32 in patients who switched from placebo to apremilast at 16 weeks, the researchers noted.
Response at 16 weeks was significantly greater on apremilast, compared with placebo, for 269 patients with scalp psoriasis (41% vs. 17%), and for 42 patients with palmoplantar psoriasis (65% vs. 31%).
Most adverse events were mild to moderate, and discontinuation rates because of adverse events were approximately 5% for placebo and treatment patients. The most common adverse events overall were nausea, diarrhea, nasopharyngitis, upper respiratory tract infection, headache, and vomiting.
The findings were limited by the short duration of the study, the researchers noted. However, apremilast was safe and well tolerated, and improvement was noted up to 32 weeks, they added.
Dr. Crowley reported receiving fees from Celgene during the study, and reported financial relationships with other companies, including AbbVie, Amgen, AstraZeneca, Eli Lilly, Janssen, Merck, and Pfizer.
LAS VEGAS – A twice-daily 30-mg dose of apremilast significantly improved moderate to severe nail, scalp, and palmoplantar psoriasis, compared with placebo, based on data from 411 adults in the ESTEEM 2 phase III study.
Overall, at 16 weeks, 29% of the patients randomized to apremilast achieved PASI-75, and 56% achieved PASI-50, compared with placebo patients (6% and 20%, respectively). The findings were presented in a poster at SDEF Las Vegas Dermatology Seminar by Dr. Jeffrey Crowley of Bakersfield (Calif.) Dermatology, and his colleagues.
In addition, patients with palmarplantar, nail, and scalp psoriasis who received apremilast showed significantly greater response rates, compared with placebo patients.
At baseline, 266 patients had scores of 1 or greater on the Nail Psoriasis Severity Index (NAPSI). Significantly more of these patients on apremilast achieved NAPSI-50 vs. placebo patients after 16 weeks (45% vs. 19%, respectively). The average percent change in NAPSI score from baseline was 29% for apremilast vs. 7% for placebo.
“Among patients initially randomized to apremilast and maintained on apremilast through week 32, nail psoriasis was further improved at week 32 (55% of patients achieved NAPSI-50)” and improvements were seen at week 32 in patients who switched from placebo to apremilast at 16 weeks, the researchers noted.
Response at 16 weeks was significantly greater on apremilast, compared with placebo, for 269 patients with scalp psoriasis (41% vs. 17%), and for 42 patients with palmoplantar psoriasis (65% vs. 31%).
Most adverse events were mild to moderate, and discontinuation rates because of adverse events were approximately 5% for placebo and treatment patients. The most common adverse events overall were nausea, diarrhea, nasopharyngitis, upper respiratory tract infection, headache, and vomiting.
The findings were limited by the short duration of the study, the researchers noted. However, apremilast was safe and well tolerated, and improvement was noted up to 32 weeks, they added.
Dr. Crowley reported receiving fees from Celgene during the study, and reported financial relationships with other companies, including AbbVie, Amgen, AstraZeneca, Eli Lilly, Janssen, Merck, and Pfizer.
LAS VEGAS – A twice-daily 30-mg dose of apremilast significantly improved moderate to severe nail, scalp, and palmoplantar psoriasis, compared with placebo, based on data from 411 adults in the ESTEEM 2 phase III study.
Overall, at 16 weeks, 29% of the patients randomized to apremilast achieved PASI-75, and 56% achieved PASI-50, compared with placebo patients (6% and 20%, respectively). The findings were presented in a poster at SDEF Las Vegas Dermatology Seminar by Dr. Jeffrey Crowley of Bakersfield (Calif.) Dermatology, and his colleagues.
In addition, patients with palmarplantar, nail, and scalp psoriasis who received apremilast showed significantly greater response rates, compared with placebo patients.
At baseline, 266 patients had scores of 1 or greater on the Nail Psoriasis Severity Index (NAPSI). Significantly more of these patients on apremilast achieved NAPSI-50 vs. placebo patients after 16 weeks (45% vs. 19%, respectively). The average percent change in NAPSI score from baseline was 29% for apremilast vs. 7% for placebo.
“Among patients initially randomized to apremilast and maintained on apremilast through week 32, nail psoriasis was further improved at week 32 (55% of patients achieved NAPSI-50)” and improvements were seen at week 32 in patients who switched from placebo to apremilast at 16 weeks, the researchers noted.
Response at 16 weeks was significantly greater on apremilast, compared with placebo, for 269 patients with scalp psoriasis (41% vs. 17%), and for 42 patients with palmoplantar psoriasis (65% vs. 31%).
Most adverse events were mild to moderate, and discontinuation rates because of adverse events were approximately 5% for placebo and treatment patients. The most common adverse events overall were nausea, diarrhea, nasopharyngitis, upper respiratory tract infection, headache, and vomiting.
The findings were limited by the short duration of the study, the researchers noted. However, apremilast was safe and well tolerated, and improvement was noted up to 32 weeks, they added.
Dr. Crowley reported receiving fees from Celgene during the study, and reported financial relationships with other companies, including AbbVie, Amgen, AstraZeneca, Eli Lilly, Janssen, Merck, and Pfizer.
AT SDEF LAS VEGAS DERMATOLOGY SEMINAR
Key clinical point: Apremilast significantly improved psoriasis in a subset of patient with nail, scalp, and palmoplantar involvement.
Major finding: After 16 weeks of twice-daily apremilast, significantly more patients with nail psoriasis achieved NAPSI-50, compared with placebo patients (45% vs. 19%, respectively).
Data source: Analysis of data from ESTEEM 2.
Disclosures:Dr. Crowley reported receiving fees from Celgene during the study and reported financial relationships with other companies, including AbbVie, Amgen, AstraZeneca, Eli Lilly, Janssen, Merck, and Pfizer.
VIDEO: Don’t discount topical therapy for psoriasis
LAS VEGAS– Don’t underestimate the value of topical therapy for treating psoriasis patients, Dr. Linda Stein Gold said at Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
Dr. Stein Gold explained how to overcome some of the compliance issues associated with topical therapies for psoriasis, and offered her suggestions for managing side effects in an interview at the meeting.
Dr. Stein Gold disclosed relationships with multiple pharmaceutical companies including Galderma, Stiefel, Allergan, Valeant, Ranbaxy, Promius, and Actavis.
SDEF and this news organization are owned by Frontline Medical Communications.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
LAS VEGAS– Don’t underestimate the value of topical therapy for treating psoriasis patients, Dr. Linda Stein Gold said at Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
Dr. Stein Gold explained how to overcome some of the compliance issues associated with topical therapies for psoriasis, and offered her suggestions for managing side effects in an interview at the meeting.
Dr. Stein Gold disclosed relationships with multiple pharmaceutical companies including Galderma, Stiefel, Allergan, Valeant, Ranbaxy, Promius, and Actavis.
SDEF and this news organization are owned by Frontline Medical Communications.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
LAS VEGAS– Don’t underestimate the value of topical therapy for treating psoriasis patients, Dr. Linda Stein Gold said at Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
Dr. Stein Gold explained how to overcome some of the compliance issues associated with topical therapies for psoriasis, and offered her suggestions for managing side effects in an interview at the meeting.
Dr. Stein Gold disclosed relationships with multiple pharmaceutical companies including Galderma, Stiefel, Allergan, Valeant, Ranbaxy, Promius, and Actavis.
SDEF and this news organization are owned by Frontline Medical Communications.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT SDEF LAS VEGAS DERMATOLOGY SEMINAR
Psoriasis patients’ QOL improved with calcipotriene/betamethasone
LAS VEGAS – Adults with localized psoriasis reported significant improvement in quality of life after 2 and 8 weeks of treatment with a combination topical suspension, based on data from 147 adults.
Calcipotriene 0.005% plus betamethasone diproprionate 0.064% (CBD) has shown safety and effectiveness in adults with psoriasis. Dr. Jerry Bagel, who is in group practice in East Windsor, N.J., and his colleagues documented real-life experiences with CBD and patient-reported outcomes. The findings were presented at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
The average change in quality of life scores from baseline was –4.2 at week 2 and –5.5 at week 8; both were statistically significant. In addition, 38% and 42% of patients at 2 weeks and 8 weeks, respectively, met the secondary endpoint of at least a 5-point improvement on the Dermatology Life Quality Index (DLQI).
The mean percent change in the patients’ perceptions of itching was –19% at week 2 and –29% at week 8, based on an itch visual analog scale, Dr. Bagel noted. Patient satisfaction with treatment also was measured using the Treatment Satisfaction Questionnaire for Medication–9 (TSQM-9), and the average scores for treatment effectiveness, convenience, and satisfaction were greater than 65 on a scale of 0-100 at week 2 and week 8.
Dr. Bagel and his colleagues studied 147 adults aged 18 years and older; patients’ average age was 49 years. More than half (57%) were women; 78% of patients were white. Approximately 56% were characterized as having moderate disease, and the average disease duration was 11 years. Only two patients reported treatment-emergent adverse events, but these were deemed unrelated to the study drug by the researchers.
The findings were limited by the use of self-reports. However, “patients with an extensive history of psoriasis and various levels of disease severity were satisfied with the product” in this real-life treatment profile, he noted.
Dr. Bagel has served as a consultant, speaker, and investigator for multiple companies, including Leo Pharma, which sponsored this study. SDEF and this news organization are owned by Frontline Medical Communications.
LAS VEGAS – Adults with localized psoriasis reported significant improvement in quality of life after 2 and 8 weeks of treatment with a combination topical suspension, based on data from 147 adults.
Calcipotriene 0.005% plus betamethasone diproprionate 0.064% (CBD) has shown safety and effectiveness in adults with psoriasis. Dr. Jerry Bagel, who is in group practice in East Windsor, N.J., and his colleagues documented real-life experiences with CBD and patient-reported outcomes. The findings were presented at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
The average change in quality of life scores from baseline was –4.2 at week 2 and –5.5 at week 8; both were statistically significant. In addition, 38% and 42% of patients at 2 weeks and 8 weeks, respectively, met the secondary endpoint of at least a 5-point improvement on the Dermatology Life Quality Index (DLQI).
The mean percent change in the patients’ perceptions of itching was –19% at week 2 and –29% at week 8, based on an itch visual analog scale, Dr. Bagel noted. Patient satisfaction with treatment also was measured using the Treatment Satisfaction Questionnaire for Medication–9 (TSQM-9), and the average scores for treatment effectiveness, convenience, and satisfaction were greater than 65 on a scale of 0-100 at week 2 and week 8.
Dr. Bagel and his colleagues studied 147 adults aged 18 years and older; patients’ average age was 49 years. More than half (57%) were women; 78% of patients were white. Approximately 56% were characterized as having moderate disease, and the average disease duration was 11 years. Only two patients reported treatment-emergent adverse events, but these were deemed unrelated to the study drug by the researchers.
The findings were limited by the use of self-reports. However, “patients with an extensive history of psoriasis and various levels of disease severity were satisfied with the product” in this real-life treatment profile, he noted.
Dr. Bagel has served as a consultant, speaker, and investigator for multiple companies, including Leo Pharma, which sponsored this study. SDEF and this news organization are owned by Frontline Medical Communications.
LAS VEGAS – Adults with localized psoriasis reported significant improvement in quality of life after 2 and 8 weeks of treatment with a combination topical suspension, based on data from 147 adults.
Calcipotriene 0.005% plus betamethasone diproprionate 0.064% (CBD) has shown safety and effectiveness in adults with psoriasis. Dr. Jerry Bagel, who is in group practice in East Windsor, N.J., and his colleagues documented real-life experiences with CBD and patient-reported outcomes. The findings were presented at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
The average change in quality of life scores from baseline was –4.2 at week 2 and –5.5 at week 8; both were statistically significant. In addition, 38% and 42% of patients at 2 weeks and 8 weeks, respectively, met the secondary endpoint of at least a 5-point improvement on the Dermatology Life Quality Index (DLQI).
The mean percent change in the patients’ perceptions of itching was –19% at week 2 and –29% at week 8, based on an itch visual analog scale, Dr. Bagel noted. Patient satisfaction with treatment also was measured using the Treatment Satisfaction Questionnaire for Medication–9 (TSQM-9), and the average scores for treatment effectiveness, convenience, and satisfaction were greater than 65 on a scale of 0-100 at week 2 and week 8.
Dr. Bagel and his colleagues studied 147 adults aged 18 years and older; patients’ average age was 49 years. More than half (57%) were women; 78% of patients were white. Approximately 56% were characterized as having moderate disease, and the average disease duration was 11 years. Only two patients reported treatment-emergent adverse events, but these were deemed unrelated to the study drug by the researchers.
The findings were limited by the use of self-reports. However, “patients with an extensive history of psoriasis and various levels of disease severity were satisfied with the product” in this real-life treatment profile, he noted.
Dr. Bagel has served as a consultant, speaker, and investigator for multiple companies, including Leo Pharma, which sponsored this study. SDEF and this news organization are owned by Frontline Medical Communications.
AT SDEF LAS VEGAS DERMATOLOGY SEMINAR
Key clinical point: Calcipotriene 0.005% plus betamethasone diproprionate 0.064% was safe and effective and improved patients’ perceived quality of life.
Major finding: The average change in quality of life scores from baseline was –4.2 at week 2 and –5.5 at week 8.
Data source: Data from 147 adults aged 18 years and older with psoraisis vulgaris of an average of 11 years’ duration.
Disclosures: Dr. Bagel has served as a consultant, speaker, and investigator for multiple companies, including Leo Pharma, which sponsored this study.
Novel psoriasis biologic wows with jaw-dropping results
AMSTERDAM– The spectacular long-term efficacy achieved with a novel biologic agent for psoriasis in a first-in-humans, proof-of-concept study has raised the prospect of clinical outcomes continuing to ratchet higher in the treatment of moderate-to-severe chronic plaque psoriasis.
How much higher? Six of nine treated patients followed long-term have maintained a PASI 100 response – that is, completely clear – for up to 66 months after a single subcutaneous injection of the agent known for now as BI 655066, Dr. James G. Krueger reported at the annual congress of the European Academy of Dermatology and Venereology.
“For me, this is one of the most interesting features of this proof-of-concept study,” he added. “If this kind of activity is confirmed in the ongoing phase IIb trial, I think this represents the potential for very long-term disease modification. This could become an important agent in the future to treat psoriasis.”
BI 655066 is a monoclonal antibody that specifically targets the p19 subunit of interleukin (IL)-23. Unlike ustekinumab (Stelara), which blocks both IL-23 and IL-12, BI 655066 selectively blocks only IL-23, which Dr. Krueger believes is the central driving force in activating and sustaining the T-cell subsets responsible for the hyperproliferative and inflammatory reactions that define psoriasis.
“This study is all about testing for the specific pathogenic contribution of IL-23 to psoriasis in a first-in-humans study. Our findings really emphasize the importance of IL-23 in driving the key pathways of psoriasis,” observed Dr. Krueger, professor of investigative dermatology and director of the Milstein Medical Research Program at Rockefeller University, New York.
The study included 39 patients with moderate to severe plaque psoriasis. Their baseline PASI was 18, and they averaged more than a 20-year history of psoriasis. Twenty-four patients were randomized 3:1 to a single intravenous injection of BI 655066 at various doses ranging from 0.01 mg/kg to 5 mg/kg or to placebo in order to get an initial sense of the agent’s safety and tolerability.
In the second part of the study, 15 other participants received a single subcutaneous injection: two got placebo and the rest were randomized to BI 655066 at either 0.25 mg/kg or 1.0 mg/kg. Safety and efficacy were assessed at weeks 0, 2, 4, 12, and 24. In addition, skin biopsies were obtained at weeks 0 and 8 for immunohistochemistry studies and RNA sequencing analysis.
By week 12, the PASI 75 response rate in subcutaneous BI 655066 recipients was 87% and the PASI 90 rate was 58%. At week 24, nine patients elected to continue structured prospective follow-up while remaining off treatment, including six PASI 100 responders. Those six PASI 100 responders remained PASI 100 at ongoing follow-up 48-66 weeks after receiving their single dose of the agent.
Biopsy specimens obtained at week 8 showed normalization of the epidermal psoriasiform hyperplasia which had been present at baseline. A normal-looking granular layer had been reestablished. “This looks essentially like the pattern of normal or nonlesional skin,” according to the dermatologist.
RNA sequencing analysis and gene profiling showed normalized production of the IL-23/IL-17-induced proteins that had been strongly overexpressed at baseline, including lipocalin, beta-defensin, and psoriasin.
“The immune axis is turned down. The number of immune cells is way down, although they’re not completely eliminated. With placebo, you still see a psoriasislike pattern of the disease. With blockade of IL-23, most cases have a gene profile like nonlesional skin. This represents a profound cellular and disease modulation,” Dr. Krueger said.
Among all 39 participants, the only serious adverse event deemed possibly treatment related was a 5-minute transient ischemic attack (TIA) episode in a patient on BI 655066. This caught Dr. Krueger’s attention as a possible red flag; however, he noted that more than 200 patients have since received the biologic agent in the ongoing phase IIb trial, with no reported major adverse cardiovascular events.
“I think that TIA may just be bad luck with small numbers,” he added.
Asked what he thinks might explain the remarkably lengthy disease remission seen following a single dose of the biologic, Dr. Krueger offered two possibilities.
“It may be that IL-23 is necessary to sustain pathogenic clones of memory cells in the skin, and as we get rid of it those clones most likely apoptose. And if you’ve sufficiently removed the clones, then you don’t get the expansion. That’s guess one. Guess two would be that we’ve renormalized tolerance mechanisms in some way. Both of these hypotheses can be tested,” according to Dr. Krueger.
The study was funded by Boehringer Ingelheim. Dr. Krueger reported receiving funding from that pharmaceutical company and nearly two dozen others.
AMSTERDAM– The spectacular long-term efficacy achieved with a novel biologic agent for psoriasis in a first-in-humans, proof-of-concept study has raised the prospect of clinical outcomes continuing to ratchet higher in the treatment of moderate-to-severe chronic plaque psoriasis.
How much higher? Six of nine treated patients followed long-term have maintained a PASI 100 response – that is, completely clear – for up to 66 months after a single subcutaneous injection of the agent known for now as BI 655066, Dr. James G. Krueger reported at the annual congress of the European Academy of Dermatology and Venereology.
“For me, this is one of the most interesting features of this proof-of-concept study,” he added. “If this kind of activity is confirmed in the ongoing phase IIb trial, I think this represents the potential for very long-term disease modification. This could become an important agent in the future to treat psoriasis.”
BI 655066 is a monoclonal antibody that specifically targets the p19 subunit of interleukin (IL)-23. Unlike ustekinumab (Stelara), which blocks both IL-23 and IL-12, BI 655066 selectively blocks only IL-23, which Dr. Krueger believes is the central driving force in activating and sustaining the T-cell subsets responsible for the hyperproliferative and inflammatory reactions that define psoriasis.
“This study is all about testing for the specific pathogenic contribution of IL-23 to psoriasis in a first-in-humans study. Our findings really emphasize the importance of IL-23 in driving the key pathways of psoriasis,” observed Dr. Krueger, professor of investigative dermatology and director of the Milstein Medical Research Program at Rockefeller University, New York.
The study included 39 patients with moderate to severe plaque psoriasis. Their baseline PASI was 18, and they averaged more than a 20-year history of psoriasis. Twenty-four patients were randomized 3:1 to a single intravenous injection of BI 655066 at various doses ranging from 0.01 mg/kg to 5 mg/kg or to placebo in order to get an initial sense of the agent’s safety and tolerability.
In the second part of the study, 15 other participants received a single subcutaneous injection: two got placebo and the rest were randomized to BI 655066 at either 0.25 mg/kg or 1.0 mg/kg. Safety and efficacy were assessed at weeks 0, 2, 4, 12, and 24. In addition, skin biopsies were obtained at weeks 0 and 8 for immunohistochemistry studies and RNA sequencing analysis.
By week 12, the PASI 75 response rate in subcutaneous BI 655066 recipients was 87% and the PASI 90 rate was 58%. At week 24, nine patients elected to continue structured prospective follow-up while remaining off treatment, including six PASI 100 responders. Those six PASI 100 responders remained PASI 100 at ongoing follow-up 48-66 weeks after receiving their single dose of the agent.
Biopsy specimens obtained at week 8 showed normalization of the epidermal psoriasiform hyperplasia which had been present at baseline. A normal-looking granular layer had been reestablished. “This looks essentially like the pattern of normal or nonlesional skin,” according to the dermatologist.
RNA sequencing analysis and gene profiling showed normalized production of the IL-23/IL-17-induced proteins that had been strongly overexpressed at baseline, including lipocalin, beta-defensin, and psoriasin.
“The immune axis is turned down. The number of immune cells is way down, although they’re not completely eliminated. With placebo, you still see a psoriasislike pattern of the disease. With blockade of IL-23, most cases have a gene profile like nonlesional skin. This represents a profound cellular and disease modulation,” Dr. Krueger said.
Among all 39 participants, the only serious adverse event deemed possibly treatment related was a 5-minute transient ischemic attack (TIA) episode in a patient on BI 655066. This caught Dr. Krueger’s attention as a possible red flag; however, he noted that more than 200 patients have since received the biologic agent in the ongoing phase IIb trial, with no reported major adverse cardiovascular events.
“I think that TIA may just be bad luck with small numbers,” he added.
Asked what he thinks might explain the remarkably lengthy disease remission seen following a single dose of the biologic, Dr. Krueger offered two possibilities.
“It may be that IL-23 is necessary to sustain pathogenic clones of memory cells in the skin, and as we get rid of it those clones most likely apoptose. And if you’ve sufficiently removed the clones, then you don’t get the expansion. That’s guess one. Guess two would be that we’ve renormalized tolerance mechanisms in some way. Both of these hypotheses can be tested,” according to Dr. Krueger.
The study was funded by Boehringer Ingelheim. Dr. Krueger reported receiving funding from that pharmaceutical company and nearly two dozen others.
AMSTERDAM– The spectacular long-term efficacy achieved with a novel biologic agent for psoriasis in a first-in-humans, proof-of-concept study has raised the prospect of clinical outcomes continuing to ratchet higher in the treatment of moderate-to-severe chronic plaque psoriasis.
How much higher? Six of nine treated patients followed long-term have maintained a PASI 100 response – that is, completely clear – for up to 66 months after a single subcutaneous injection of the agent known for now as BI 655066, Dr. James G. Krueger reported at the annual congress of the European Academy of Dermatology and Venereology.
“For me, this is one of the most interesting features of this proof-of-concept study,” he added. “If this kind of activity is confirmed in the ongoing phase IIb trial, I think this represents the potential for very long-term disease modification. This could become an important agent in the future to treat psoriasis.”
BI 655066 is a monoclonal antibody that specifically targets the p19 subunit of interleukin (IL)-23. Unlike ustekinumab (Stelara), which blocks both IL-23 and IL-12, BI 655066 selectively blocks only IL-23, which Dr. Krueger believes is the central driving force in activating and sustaining the T-cell subsets responsible for the hyperproliferative and inflammatory reactions that define psoriasis.
“This study is all about testing for the specific pathogenic contribution of IL-23 to psoriasis in a first-in-humans study. Our findings really emphasize the importance of IL-23 in driving the key pathways of psoriasis,” observed Dr. Krueger, professor of investigative dermatology and director of the Milstein Medical Research Program at Rockefeller University, New York.
The study included 39 patients with moderate to severe plaque psoriasis. Their baseline PASI was 18, and they averaged more than a 20-year history of psoriasis. Twenty-four patients were randomized 3:1 to a single intravenous injection of BI 655066 at various doses ranging from 0.01 mg/kg to 5 mg/kg or to placebo in order to get an initial sense of the agent’s safety and tolerability.
In the second part of the study, 15 other participants received a single subcutaneous injection: two got placebo and the rest were randomized to BI 655066 at either 0.25 mg/kg or 1.0 mg/kg. Safety and efficacy were assessed at weeks 0, 2, 4, 12, and 24. In addition, skin biopsies were obtained at weeks 0 and 8 for immunohistochemistry studies and RNA sequencing analysis.
By week 12, the PASI 75 response rate in subcutaneous BI 655066 recipients was 87% and the PASI 90 rate was 58%. At week 24, nine patients elected to continue structured prospective follow-up while remaining off treatment, including six PASI 100 responders. Those six PASI 100 responders remained PASI 100 at ongoing follow-up 48-66 weeks after receiving their single dose of the agent.
Biopsy specimens obtained at week 8 showed normalization of the epidermal psoriasiform hyperplasia which had been present at baseline. A normal-looking granular layer had been reestablished. “This looks essentially like the pattern of normal or nonlesional skin,” according to the dermatologist.
RNA sequencing analysis and gene profiling showed normalized production of the IL-23/IL-17-induced proteins that had been strongly overexpressed at baseline, including lipocalin, beta-defensin, and psoriasin.
“The immune axis is turned down. The number of immune cells is way down, although they’re not completely eliminated. With placebo, you still see a psoriasislike pattern of the disease. With blockade of IL-23, most cases have a gene profile like nonlesional skin. This represents a profound cellular and disease modulation,” Dr. Krueger said.
Among all 39 participants, the only serious adverse event deemed possibly treatment related was a 5-minute transient ischemic attack (TIA) episode in a patient on BI 655066. This caught Dr. Krueger’s attention as a possible red flag; however, he noted that more than 200 patients have since received the biologic agent in the ongoing phase IIb trial, with no reported major adverse cardiovascular events.
“I think that TIA may just be bad luck with small numbers,” he added.
Asked what he thinks might explain the remarkably lengthy disease remission seen following a single dose of the biologic, Dr. Krueger offered two possibilities.
“It may be that IL-23 is necessary to sustain pathogenic clones of memory cells in the skin, and as we get rid of it those clones most likely apoptose. And if you’ve sufficiently removed the clones, then you don’t get the expansion. That’s guess one. Guess two would be that we’ve renormalized tolerance mechanisms in some way. Both of these hypotheses can be tested,” according to Dr. Krueger.
The study was funded by Boehringer Ingelheim. Dr. Krueger reported receiving funding from that pharmaceutical company and nearly two dozen others.
AT THE EADV CONGRESS
Key clinical point: Up to 66 months after receiving a single subcutaneous injection of a biologic agent that selectively blocks interleukin-23, six patients with moderate to severe chronic plaque psoriasis at baseline remained PASI 100 responders with clear skin.
Major finding: The PASI 75 response rate 12 weeks after receiving a single dose of the investigational agent BI 655066 was 87%, and the PASI 90 rate was 58%.
Data source: This was a first-in-humans, proof-of-concept study involving 39 psoriasis patients.
Disclosures: The study was sponsored by Boehringer Ingelheim. The presenter reported receiving research funding from that pharmaceutical company and nearly two dozen others.
Severe diaper rash, cradle cap raise suspicion for pediatric psoriasis
NEWPORT BEACH, CALIF. – A history of severe cradle cap and diaper dermatitis helps to differentiate between pediatric psoriasis and atopic dermatitis, so be sure to ask, according to Dr. Alan Menter, chief of the dermatology division at the Baylor University Medical Center in Dallas.
“Both are markers for later onset of psoriasis, and are much more likely to be a marker for psoriasis than atopic eczema,” he said at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
The tip to ask about cradle cap and diaper dermatitis is based largely on clinical observation, but is more useful than asking about a family history of psoriasis, because people tend to keep psoriasis to themselves, he noted; family members and even spouses might not know. “It’s a very hidden disease, so family history is of little benefit,” he said.
Recent strep infection also may provide a clue, not only for guttate psoriasis but also probably for plaque psoriasis in children, Dr. Menter said. But the sooner pediatric psoriasis is caught and controlled, the better, no matter how it is detected. Aside from the suffering it causes on its own, psoriasis in children has been linked to diabetes, hypertension, fatty liver disease, obesity, and cardiovascular problems, he noted.
The mechanism of action for these comorbidities remains under investigation. Perhaps mothers with psoriasis gain more weight during pregnancy, and their children are heavier at birth, Dr. Menter said.
Crohn’s disease is far more likely in children with psoriasis, too. Dr. Menter noted that he has had referrals where the diagnosis has been missed, even in the setting of long-standing fatigue and diarrhea. “We have to look for it [Crohn’s] in our psoriasis population,” he said.
Children with psoriasis are often teased, taunted, and bullied, sometimes as young as kindergarten age. The emotional stress, loneliness, and depression can have a major impact on school and social growth, Dr. Menter said.
“Treatment of these kids goes beyond prescribing a topical steroid; they need [both] physical and psychological support,” he emphasized. Talk to parents and teachers about how the child is doing in school and other social settings. Parents might know about grades, but not much about their child’s social interactions. To help catch problems, also “take a quality of life index on all your patients with psoriasis,” he said.
It’s important to intervene early and get children’s skin cleared quickly. “[Although] we’d love to treat [everybody] with topicals and wet compresses,” effective treatment sometimes means systemic therapy, he said.
Cyclosporine is a valid rescue option, particularly for more inflammatory disease. “Rarely, if ever, have I seen any hypertension or serum creatinine issues,” Dr. Menter said. “You just have to warn parents to be careful about gums, because you can get gingival hyperplasia, and girls don’t like the mild hypertrichosis you sometimes get around the temples and forearms,” he said.
Etanercept is another option. It not approved for pediatric psoriasis, but if you try hard enough, you can get insurance companies to cover it, Dr. Menter said. “You have to talk about quality of life and how psoriasis has impacted schooling,” among other topics, he explained.
Clinicians looking for child-oriented resources and support materials can recommend the National Psoriasis Foundation to their patients, he noted. SDEF and this news organization are owned by Frontline Medical Communications.
Dr. Menter disclosed financial relationships with Abbott, AbbVie, and numerous other companies.
NEWPORT BEACH, CALIF. – A history of severe cradle cap and diaper dermatitis helps to differentiate between pediatric psoriasis and atopic dermatitis, so be sure to ask, according to Dr. Alan Menter, chief of the dermatology division at the Baylor University Medical Center in Dallas.
“Both are markers for later onset of psoriasis, and are much more likely to be a marker for psoriasis than atopic eczema,” he said at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
The tip to ask about cradle cap and diaper dermatitis is based largely on clinical observation, but is more useful than asking about a family history of psoriasis, because people tend to keep psoriasis to themselves, he noted; family members and even spouses might not know. “It’s a very hidden disease, so family history is of little benefit,” he said.
Recent strep infection also may provide a clue, not only for guttate psoriasis but also probably for plaque psoriasis in children, Dr. Menter said. But the sooner pediatric psoriasis is caught and controlled, the better, no matter how it is detected. Aside from the suffering it causes on its own, psoriasis in children has been linked to diabetes, hypertension, fatty liver disease, obesity, and cardiovascular problems, he noted.
The mechanism of action for these comorbidities remains under investigation. Perhaps mothers with psoriasis gain more weight during pregnancy, and their children are heavier at birth, Dr. Menter said.
Crohn’s disease is far more likely in children with psoriasis, too. Dr. Menter noted that he has had referrals where the diagnosis has been missed, even in the setting of long-standing fatigue and diarrhea. “We have to look for it [Crohn’s] in our psoriasis population,” he said.
Children with psoriasis are often teased, taunted, and bullied, sometimes as young as kindergarten age. The emotional stress, loneliness, and depression can have a major impact on school and social growth, Dr. Menter said.
“Treatment of these kids goes beyond prescribing a topical steroid; they need [both] physical and psychological support,” he emphasized. Talk to parents and teachers about how the child is doing in school and other social settings. Parents might know about grades, but not much about their child’s social interactions. To help catch problems, also “take a quality of life index on all your patients with psoriasis,” he said.
It’s important to intervene early and get children’s skin cleared quickly. “[Although] we’d love to treat [everybody] with topicals and wet compresses,” effective treatment sometimes means systemic therapy, he said.
Cyclosporine is a valid rescue option, particularly for more inflammatory disease. “Rarely, if ever, have I seen any hypertension or serum creatinine issues,” Dr. Menter said. “You just have to warn parents to be careful about gums, because you can get gingival hyperplasia, and girls don’t like the mild hypertrichosis you sometimes get around the temples and forearms,” he said.
Etanercept is another option. It not approved for pediatric psoriasis, but if you try hard enough, you can get insurance companies to cover it, Dr. Menter said. “You have to talk about quality of life and how psoriasis has impacted schooling,” among other topics, he explained.
Clinicians looking for child-oriented resources and support materials can recommend the National Psoriasis Foundation to their patients, he noted. SDEF and this news organization are owned by Frontline Medical Communications.
Dr. Menter disclosed financial relationships with Abbott, AbbVie, and numerous other companies.
NEWPORT BEACH, CALIF. – A history of severe cradle cap and diaper dermatitis helps to differentiate between pediatric psoriasis and atopic dermatitis, so be sure to ask, according to Dr. Alan Menter, chief of the dermatology division at the Baylor University Medical Center in Dallas.
“Both are markers for later onset of psoriasis, and are much more likely to be a marker for psoriasis than atopic eczema,” he said at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
The tip to ask about cradle cap and diaper dermatitis is based largely on clinical observation, but is more useful than asking about a family history of psoriasis, because people tend to keep psoriasis to themselves, he noted; family members and even spouses might not know. “It’s a very hidden disease, so family history is of little benefit,” he said.
Recent strep infection also may provide a clue, not only for guttate psoriasis but also probably for plaque psoriasis in children, Dr. Menter said. But the sooner pediatric psoriasis is caught and controlled, the better, no matter how it is detected. Aside from the suffering it causes on its own, psoriasis in children has been linked to diabetes, hypertension, fatty liver disease, obesity, and cardiovascular problems, he noted.
The mechanism of action for these comorbidities remains under investigation. Perhaps mothers with psoriasis gain more weight during pregnancy, and their children are heavier at birth, Dr. Menter said.
Crohn’s disease is far more likely in children with psoriasis, too. Dr. Menter noted that he has had referrals where the diagnosis has been missed, even in the setting of long-standing fatigue and diarrhea. “We have to look for it [Crohn’s] in our psoriasis population,” he said.
Children with psoriasis are often teased, taunted, and bullied, sometimes as young as kindergarten age. The emotional stress, loneliness, and depression can have a major impact on school and social growth, Dr. Menter said.
“Treatment of these kids goes beyond prescribing a topical steroid; they need [both] physical and psychological support,” he emphasized. Talk to parents and teachers about how the child is doing in school and other social settings. Parents might know about grades, but not much about their child’s social interactions. To help catch problems, also “take a quality of life index on all your patients with psoriasis,” he said.
It’s important to intervene early and get children’s skin cleared quickly. “[Although] we’d love to treat [everybody] with topicals and wet compresses,” effective treatment sometimes means systemic therapy, he said.
Cyclosporine is a valid rescue option, particularly for more inflammatory disease. “Rarely, if ever, have I seen any hypertension or serum creatinine issues,” Dr. Menter said. “You just have to warn parents to be careful about gums, because you can get gingival hyperplasia, and girls don’t like the mild hypertrichosis you sometimes get around the temples and forearms,” he said.
Etanercept is another option. It not approved for pediatric psoriasis, but if you try hard enough, you can get insurance companies to cover it, Dr. Menter said. “You have to talk about quality of life and how psoriasis has impacted schooling,” among other topics, he explained.
Clinicians looking for child-oriented resources and support materials can recommend the National Psoriasis Foundation to their patients, he noted. SDEF and this news organization are owned by Frontline Medical Communications.
Dr. Menter disclosed financial relationships with Abbott, AbbVie, and numerous other companies.
EXPERT ANALYSIS FROM SDEF WOMEN’S AND PEDIATRIC DERMATOLOGY SEMINAR
Psoriasiform lesions linked to anti-TNF treatment
VIENNA – Patients treated with an anti–tumor necrosis factor drug had a 5% annual rate of developing one or more psoriasiform skin lesions in a review of more than 400 patients who received these drugs to treat inflammatory bowel disease at a single center in Rome.
The cohort review confirmed prior reports that smoking is a risk factor for the appearance of psoriasislike skin lesions on patients being treated with an anti–tumor necrosis factor (TNF) drug, such as infliximab (Remicade) or adalimumab (Humira). The Rome experience also showed that in 28 of the 42 patients who developed a psoriasiform lesion, the eruption responded to topical treatment without need to stop or change the anti-TNF regimen. Ten of the 42 patients ultimately had to stop their anti-TNF regimen, Dr. Daniela Pugliese said at the United European Gastroenterology Global Congress.
“There have been several case reports of this, but this is the largest cohort review yet reported by one center,” said Dr. Pugliese, a gastroenterologist in the inflammatory bowel disease (IBD) unit of Complesso Integrato Columbus, Catholic University in Rome.
The review included 402 patients treated with an anti-TNF drug at the unit during 2008-2013, with a median follow-up of 17 months. During follow-up, 42 patients developed a psoriasiform lesion with a biopsy-proven diagnosis, a rate of 5 cases/100 person-years on anti-TNF treatment. The IBD patients averaged 40 years old, and 60% had Crohn’s disease and 40% had ulcerative colitis. About 60% received infliximab treatment, and 40% adalimumab. Most lesions appeared in predilection sites, as well as on palmoplantar surfaces or on the scalp; nearly half the patients had lesions in two or more locations.
A multivariate regression analysis that assessed several demographic and clinical features of all 402 patients identified two parameters that significantly linked with lesion development. Smoking linked with a greater than twofold increased risk for having a psoriasiform lesion (78 of the patients, 19%, smoked), and concurrent treatment with a thiopurine such as azathioprine, linked with a 67% reduced rate of lesion development (85 patients, 21%, were on concurrent thiopurine treatment). All IBD patients seen at the Rome unit who smoked were counseled regarding smoking cessation, Dr. Pugliese said in an interview.
Among the patients who did not respond to topical treatment, four received some benefit from starting treatment with ustekinumab (Stelara), which especially benefited patients who were otherwise difficult to treat, Dr. Pugliese said. Other patients benefited from starting treatment with cyclosporine, methotrexate, or a transient treatment with an oral steroid. Two patients who developed lesions on infliximab switched to adalimumab, and two other patients who had lesions on adalimumab switched to infliximab.
Dr. Pugliese and her associates did not have a good explanation of why patients on anti-TNF drugs develop these lesions, which Dr. Pugliese called “paradoxical.” One possible etiology is that inhibition of TNF-alpha results in uncontrolled production of interferon-alpha by plasmacytoid dendritic cells and this then triggers the psoriasiform eruptions.
A key element in managing these lesions may be early detection and topical treatment while they remain small, commented Dr. C. Janneke van der Woude, head of the IBD unit at Erasmus University Medical Center in Rotterdam, the Netherlands. “Every time we see an IBD patient who is on an anti-TNF drug, we ask whether they have had any itching, allergic reaction, arthritis, eye problem, or headache,” to facilitate early detection of an adverse effect from treatment, she said in an interview.
Dr. Pugliese had no disclosures. Dr. van der Woude said that she has been an adviser to Dr Falk, AbbVie, Janssen, Johnson & Johnson, and Cosmo.
On Twitter@mitchelzoler
VIENNA – Patients treated with an anti–tumor necrosis factor drug had a 5% annual rate of developing one or more psoriasiform skin lesions in a review of more than 400 patients who received these drugs to treat inflammatory bowel disease at a single center in Rome.
The cohort review confirmed prior reports that smoking is a risk factor for the appearance of psoriasislike skin lesions on patients being treated with an anti–tumor necrosis factor (TNF) drug, such as infliximab (Remicade) or adalimumab (Humira). The Rome experience also showed that in 28 of the 42 patients who developed a psoriasiform lesion, the eruption responded to topical treatment without need to stop or change the anti-TNF regimen. Ten of the 42 patients ultimately had to stop their anti-TNF regimen, Dr. Daniela Pugliese said at the United European Gastroenterology Global Congress.
“There have been several case reports of this, but this is the largest cohort review yet reported by one center,” said Dr. Pugliese, a gastroenterologist in the inflammatory bowel disease (IBD) unit of Complesso Integrato Columbus, Catholic University in Rome.
The review included 402 patients treated with an anti-TNF drug at the unit during 2008-2013, with a median follow-up of 17 months. During follow-up, 42 patients developed a psoriasiform lesion with a biopsy-proven diagnosis, a rate of 5 cases/100 person-years on anti-TNF treatment. The IBD patients averaged 40 years old, and 60% had Crohn’s disease and 40% had ulcerative colitis. About 60% received infliximab treatment, and 40% adalimumab. Most lesions appeared in predilection sites, as well as on palmoplantar surfaces or on the scalp; nearly half the patients had lesions in two or more locations.
A multivariate regression analysis that assessed several demographic and clinical features of all 402 patients identified two parameters that significantly linked with lesion development. Smoking linked with a greater than twofold increased risk for having a psoriasiform lesion (78 of the patients, 19%, smoked), and concurrent treatment with a thiopurine such as azathioprine, linked with a 67% reduced rate of lesion development (85 patients, 21%, were on concurrent thiopurine treatment). All IBD patients seen at the Rome unit who smoked were counseled regarding smoking cessation, Dr. Pugliese said in an interview.
Among the patients who did not respond to topical treatment, four received some benefit from starting treatment with ustekinumab (Stelara), which especially benefited patients who were otherwise difficult to treat, Dr. Pugliese said. Other patients benefited from starting treatment with cyclosporine, methotrexate, or a transient treatment with an oral steroid. Two patients who developed lesions on infliximab switched to adalimumab, and two other patients who had lesions on adalimumab switched to infliximab.
Dr. Pugliese and her associates did not have a good explanation of why patients on anti-TNF drugs develop these lesions, which Dr. Pugliese called “paradoxical.” One possible etiology is that inhibition of TNF-alpha results in uncontrolled production of interferon-alpha by plasmacytoid dendritic cells and this then triggers the psoriasiform eruptions.
A key element in managing these lesions may be early detection and topical treatment while they remain small, commented Dr. C. Janneke van der Woude, head of the IBD unit at Erasmus University Medical Center in Rotterdam, the Netherlands. “Every time we see an IBD patient who is on an anti-TNF drug, we ask whether they have had any itching, allergic reaction, arthritis, eye problem, or headache,” to facilitate early detection of an adverse effect from treatment, she said in an interview.
Dr. Pugliese had no disclosures. Dr. van der Woude said that she has been an adviser to Dr Falk, AbbVie, Janssen, Johnson & Johnson, and Cosmo.
On Twitter@mitchelzoler
VIENNA – Patients treated with an anti–tumor necrosis factor drug had a 5% annual rate of developing one or more psoriasiform skin lesions in a review of more than 400 patients who received these drugs to treat inflammatory bowel disease at a single center in Rome.
The cohort review confirmed prior reports that smoking is a risk factor for the appearance of psoriasislike skin lesions on patients being treated with an anti–tumor necrosis factor (TNF) drug, such as infliximab (Remicade) or adalimumab (Humira). The Rome experience also showed that in 28 of the 42 patients who developed a psoriasiform lesion, the eruption responded to topical treatment without need to stop or change the anti-TNF regimen. Ten of the 42 patients ultimately had to stop their anti-TNF regimen, Dr. Daniela Pugliese said at the United European Gastroenterology Global Congress.
“There have been several case reports of this, but this is the largest cohort review yet reported by one center,” said Dr. Pugliese, a gastroenterologist in the inflammatory bowel disease (IBD) unit of Complesso Integrato Columbus, Catholic University in Rome.
The review included 402 patients treated with an anti-TNF drug at the unit during 2008-2013, with a median follow-up of 17 months. During follow-up, 42 patients developed a psoriasiform lesion with a biopsy-proven diagnosis, a rate of 5 cases/100 person-years on anti-TNF treatment. The IBD patients averaged 40 years old, and 60% had Crohn’s disease and 40% had ulcerative colitis. About 60% received infliximab treatment, and 40% adalimumab. Most lesions appeared in predilection sites, as well as on palmoplantar surfaces or on the scalp; nearly half the patients had lesions in two or more locations.
A multivariate regression analysis that assessed several demographic and clinical features of all 402 patients identified two parameters that significantly linked with lesion development. Smoking linked with a greater than twofold increased risk for having a psoriasiform lesion (78 of the patients, 19%, smoked), and concurrent treatment with a thiopurine such as azathioprine, linked with a 67% reduced rate of lesion development (85 patients, 21%, were on concurrent thiopurine treatment). All IBD patients seen at the Rome unit who smoked were counseled regarding smoking cessation, Dr. Pugliese said in an interview.
Among the patients who did not respond to topical treatment, four received some benefit from starting treatment with ustekinumab (Stelara), which especially benefited patients who were otherwise difficult to treat, Dr. Pugliese said. Other patients benefited from starting treatment with cyclosporine, methotrexate, or a transient treatment with an oral steroid. Two patients who developed lesions on infliximab switched to adalimumab, and two other patients who had lesions on adalimumab switched to infliximab.
Dr. Pugliese and her associates did not have a good explanation of why patients on anti-TNF drugs develop these lesions, which Dr. Pugliese called “paradoxical.” One possible etiology is that inhibition of TNF-alpha results in uncontrolled production of interferon-alpha by plasmacytoid dendritic cells and this then triggers the psoriasiform eruptions.
A key element in managing these lesions may be early detection and topical treatment while they remain small, commented Dr. C. Janneke van der Woude, head of the IBD unit at Erasmus University Medical Center in Rotterdam, the Netherlands. “Every time we see an IBD patient who is on an anti-TNF drug, we ask whether they have had any itching, allergic reaction, arthritis, eye problem, or headache,” to facilitate early detection of an adverse effect from treatment, she said in an interview.
Dr. Pugliese had no disclosures. Dr. van der Woude said that she has been an adviser to Dr Falk, AbbVie, Janssen, Johnson & Johnson, and Cosmo.
On Twitter@mitchelzoler
AT UEG WEEK VIENNA 2014
Key clinical point: Inflammatory bowel disease patients on anti–tumor necrosis factor treatment often develop psoriasiform skin lesions.
Major finding: Patients receiving an anti-TNF drug developed psoriasiform lesions at an annual rate of 5%.
Data source: Retrospective review of 402 patients with inflammatory bowel disease treated with an anti-TNF drug at one center in Rome.
Disclosures: Dr. Pugliese had no disclosures. Dr. van der Woude said that she has been an adviser to Dr Falk, AbbVie, Janssen, Johnson & Johnson, and Cosmo.