User login
Disease Burden and Quality of Life in Psoriasis Patients With and Without Comorbid Psoriatic Arthritis: Results From National Psoriasis Foundation Panel Surveys
Approximately 3% of the US adult population, or roughly 5 million Americans, have been diagnosed with psoriasis.1 Psoriasis has a high disease burden, with one review of 817 quality-of-life (QOL) studies concluding that the degree to which psoriasis negatively affects patients’ QOL is comparable with major diseases such as diabetes mellitus or cancer.2
Psoriatic arthritis (PsA) is a chronic, often progressive, inflammatory arthropathy that can lead to permanent joint damage and severe disability.3,4 Psoriatic arthritis is a common comorbidity among patients with psoriasis, affecting 6% to 10% of psoriasis patients overall5 and 20% to 40% of psoriasis patients with more extensive skin involvement.5-7
The comorbidity profile and overall disease impact are not well understood in psoriasis patients with and without comorbid PsA. The primary objective of this study was to compare disease characteristics, comorbidities, and psoriasis-related QOL in patients with moderate to severe psoriasis with and without comorbid PsA.
Methods
Study Design and Participants
Since 2003 the National Psoriasis Foundation (NPF) has conducted semiannual survey panels to collect patient-reported data on topics including disease characteristics, treatment utilization, and psoriasis-related QOL impact. Responses from approximately 5604 individuals have been collected and analyzed from a total of 13 survey panels conducted from 2003-2009 and 2011. No data were available for 2010. In each survey panel, responses from approximately 400 randomly selected respondents were collected, with approximately 300 respondents having moderate to severe psoriasis and 200 respondents being NPF members. Respondents were surveyed via telephone and the Internet.
This study examined 2 main patient groups: (1) adults (>18 years of age) with psoriasis alone and (2) those with comorbid PsA. The 2 study groups were subdivided according to percentage of body surface area (BSA) affected; specifically, patients with psoriasis alone were classified as having mild psoriasis (<3% BSA) or moderate to severe psoriasis (>3% BSA) and patients with PsA were classified as having mild to no psoriasis (<3% BSA) or moderate to severe psoriasis (≥3% BSA).8
Assessments
Comparisons of patient demographics, disease characteristics, comorbidities, psoriasis-related QOL, and symptom impact were made for patients with moderate to severe psoriasis and PsA versus those with moderate to severe psoriasis alone. The overall impact of psoriasis on patients’ physical and emotional QOL was assessed via a 12-item survey with each item rated on an 11-point scale (0=not at all; 5=somewhat; 10=very much). Total QOL scores ranged from 0 to 120. Higher scores indicated greater (ie, worse) impact of psoriasis on the patient’s QOL. To evaluate symptom impact (ie, to what degree a patient felt affected by individual symptoms), ratings from 3 symptoms—itching, irritation, and pain—that were included in the overall QOL assessment also were analyzed individually. Results also were compared for patients with mild psoriasis alone versus those with moderate to severe psoriasis alone as well as for patients with PsA and mild to no psoriasis versus those with PsA and moderate to severe psoriasis.
Statistical Analysis
Adult survey respondents who answered survey questions relevant to the analysis were included. For patient demographics and comorbidities, t tests were used for continuous variables and Χ2 tests were used for categorical variables. For overall psoriasis-related QOL and symptom impact scores, differences among the study groups were compared using analyses of covariance, which adjusted for age, gender, and race. All analyses were conducted using SAS version 9.2.
Results
Patient Demographics
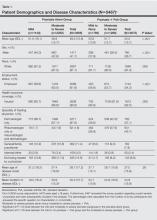
Of 5467 total respondents, 3532 reported moderate to severe psoriasis of which 1280 (36.2%) had comorbid PsA (Table 1). Compared to patients with moderate to severe psoriasis alone, those with moderate to severe psoriasis and comorbid PsA were older, less commonly male, less likely to be employed, had a longer duration of psoriasis, were more likely to be white, and were more likely to have health insurance coverage. Among the psoriasis alone group, those patients reporting moderate to severe psoriasis were younger, less likely to have health insurance coverage, and were less likely to be treated by a dermatologist only than those with mild psoriasis. Among the group with PsA, patients with moderate to severe psoriasis were more likely to be treated by a dermatologist only versus those with mild or no psoriasis (Table 1).
Patient Comorbidities
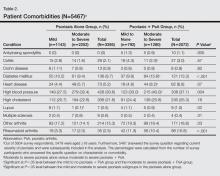
Patients with moderate to severe psoriasis and comorbid PsA reported significantly higher rates of diabetes mellitus, lupus, rheumatoid arthritis, other arthritis, ankylosing spondylitis, and high blood pressure than patients with moderate to severe psoriasis alone (P<.05)(Table 2). In the group with psoriasis alone, patients with moderate to severe psoriasis had significantly lower rates of lupus compared to those with mild psoriasis (P<.05). In the group with PsA, patients with moderate to severe psoriasis were significantly more likely to report diagnoses of colitis, diabetes mellitus, heart disease, and high blood pressure compared to those with mild to no psoriasis (P<.05).
Psoriasis-Related QOL
The psoriasis-related QOL analyses are shown in Table 3. Patients with moderate to severe psoriasis and comorbid PsA reported a greater impact on overall QOL compared to their counterparts with moderate to severe psoriasis alone. Similarly, patients with moderate to severe psoriasis and comorbid PsA reported a greater impact of itching, physical irritation/soreness, and pain symptoms.
Disease severity was associated with psoriasis-related QOL. Compared to patients with mild psoriasis alone, those with moderate to severe psoriasis alone had significantly higher (ie, worse) scores in the categories of itching, irritation, pain, and overall QOL (P<.05). In the PsA group, patients with moderate to severe psoriasis also reported significantly worse itching, irritation, pain, and overall QOL compared to their counterparts with mild to no psoriasis (P<.05).
Comment
The results of this study showed that patient demographics, comorbidities, and psoriasis-related QOL in psoriasis patients varied significantly depending on presence or absence of comorbid PsA as well as disease severity. Patients with more severe cutaneous psoriasis with and without PsA were associated with worse QOL scores and higher rates of self-reported heart disease and hypertension. Among patients with moderate to severe psoriasis, those with comorbid PsA were associated with greater rates of self-reported diabetes mellitus and high blood pressure and worse QOL scores including worse itching, irritation, and pain than those with moderate to severe psoriasis alone.
The present study is one of the first to compare psoriasis-related QOL in patients with and without PsA. A prior study demonstrated patients with PsA had worse physical impairment, fatigue severity, and health status relative to patients with psoriasis alone but no differences in dermatology-specific QOL measures.9 Our analysis revealed that patients with moderate to severe psoriasis and comorbid PsA experienced a greater impact on overall QOL as well as symptoms of itching, irritation, and pain relative to patients with moderate to severe psoriasis alone. Among both treatment groups, reports of overall QOL and itching, irritation, and pain became worse as the disease severity increased. These findings are consistent with prior studies that have shown greater impairment of QOL among patients with severe psoriasis compared to mild psoriasis.10-12
Relative to patients with moderate to severe psoriasis alone, those with moderate to severe psoriasis and comorbid PsA reported a greater prevalence of comorbid diagnoses overall, with notably higher rates of type 2 diabetes mellitus, hypertension, and rheumatoid arthritis. These findings are consistent with an earlier report by Husted et al.13
Limitations
The results of this study should be interpreted in light of several important limitations associated with survey data (ie, patient-reported data, no details on length of therapy or comorbid disease, multiple surveys taken over time, respondents aged >18 years only).
Conclusion
This study provides evidence that patients with moderate to severe psoriasis and comorbid PsA have a higher psoriasis-related disease burden compared to those with moderate to severe psoriasis alone, which suggests that physicians may wish to consider this comorbidity when designing patient treatment plans. Our findings also confirm that disease severity is associated with comorbid conditions and psoriasis-related QOL impact in psoriasis patients. These findings highlight the importance of screening for PsA in patients with psoriasis and effective treatment of skin disease in patients with PsA.
Acknowledgment
The authors thank Maureen Lage, PhD, Healthmetrics, Bonita Springs, Florida, for her work in drafting the manuscript.
1. Kurd SK, Gelfand JM. The prevalence of previously diagnosed and undiagnosed psoriasis in US adults: results from NHANES 2003-2004. J Am Acad Dermatol. 2009;60:218-224.
2. Raho G, Koleva DM, Garattini L, et al. The burden of moderate to severe psoriasis: an overview. Pharmacoeconomics. 2012;30:1005-1013.
3. Gladman DD, Antoni C, Mease P, et al. Psoriatic arthritis: epidemiology, clinical features, course, and outcome. Ann Rheum Dis. 2005;64(suppl 2):14-17.
4. Ogdie A, Langan S, Love T, et al. Prevalence and treatment patterns of psoriatic arthritis in the UK. Rheumatology (Oxford). 2013;52:568-575.
5. Ogdie A, Gelfand JM. Identification of risk factors for psoriatic arthritis: scientific opportunity meets clinical need. Arch Dermatol. 2010;146:785-788.
6. Gelfand JM, Gladman DD, Mease PJ, et al. Epidemiology of psoriatic arthritis in the population of the United States. J Am Acad Dermatol. 2005;53:573.
7. Richardson SK, Gelfand JM. Update on the natural history and systemic treatment of psoriasis. Adv Dermatol. 2008;24:171-196.
8. Armstrong AW, Robertson AD, Wu J, et al. Undertreatment, treatment trends, and treatment dissatisfaction among patients with psoriasis and psoriatic arthritis in the United States: findings from the National Psoriasis Foundation surveys, 2003-2011. JAMA Dermatol. 2013;149:1180-1185.
9. Rosen CF, Mussani F, Chandran V, et al. Patients with psoriatic arthritis have worse quality of life than those with psoriasis alone. Rheumatology (Oxford). 2012;51:571-576.
10. Armstrong AW, Schupp C, Wu J, et al. Quality of life and work productivity impairment among psoriasis patients: findings from the National Psoriasis Foundation survey data 2003-2011. PLoS ONE. 2012;7:e52935.
11. Colombo G, Altomare G, Peris K, et al. Moderate and severe plaque psoriasis: cost-of-illness study in Italy. Ther Clin Risk Manag. 2008;4:559-568.
12. Gelfand JM, Feldman SR, Stern RS, et al. Determinants of quality of life in patients with psoriasis: a study from the US population. J Am Acad Dermatol. 2004;51:704-708.
13. Husted JA, Thavaneswaran A, Chandran V, et al. Cardiovascular and other comorbidities in patients with psoriatic arthritis: a comparison with patients with psoriasis. Arthritis Care Res (Hoboken). 2011;63:1729-1735.
Approximately 3% of the US adult population, or roughly 5 million Americans, have been diagnosed with psoriasis.1 Psoriasis has a high disease burden, with one review of 817 quality-of-life (QOL) studies concluding that the degree to which psoriasis negatively affects patients’ QOL is comparable with major diseases such as diabetes mellitus or cancer.2
Psoriatic arthritis (PsA) is a chronic, often progressive, inflammatory arthropathy that can lead to permanent joint damage and severe disability.3,4 Psoriatic arthritis is a common comorbidity among patients with psoriasis, affecting 6% to 10% of psoriasis patients overall5 and 20% to 40% of psoriasis patients with more extensive skin involvement.5-7
The comorbidity profile and overall disease impact are not well understood in psoriasis patients with and without comorbid PsA. The primary objective of this study was to compare disease characteristics, comorbidities, and psoriasis-related QOL in patients with moderate to severe psoriasis with and without comorbid PsA.
Methods
Study Design and Participants
Since 2003 the National Psoriasis Foundation (NPF) has conducted semiannual survey panels to collect patient-reported data on topics including disease characteristics, treatment utilization, and psoriasis-related QOL impact. Responses from approximately 5604 individuals have been collected and analyzed from a total of 13 survey panels conducted from 2003-2009 and 2011. No data were available for 2010. In each survey panel, responses from approximately 400 randomly selected respondents were collected, with approximately 300 respondents having moderate to severe psoriasis and 200 respondents being NPF members. Respondents were surveyed via telephone and the Internet.
This study examined 2 main patient groups: (1) adults (>18 years of age) with psoriasis alone and (2) those with comorbid PsA. The 2 study groups were subdivided according to percentage of body surface area (BSA) affected; specifically, patients with psoriasis alone were classified as having mild psoriasis (<3% BSA) or moderate to severe psoriasis (>3% BSA) and patients with PsA were classified as having mild to no psoriasis (<3% BSA) or moderate to severe psoriasis (≥3% BSA).8
Assessments
Comparisons of patient demographics, disease characteristics, comorbidities, psoriasis-related QOL, and symptom impact were made for patients with moderate to severe psoriasis and PsA versus those with moderate to severe psoriasis alone. The overall impact of psoriasis on patients’ physical and emotional QOL was assessed via a 12-item survey with each item rated on an 11-point scale (0=not at all; 5=somewhat; 10=very much). Total QOL scores ranged from 0 to 120. Higher scores indicated greater (ie, worse) impact of psoriasis on the patient’s QOL. To evaluate symptom impact (ie, to what degree a patient felt affected by individual symptoms), ratings from 3 symptoms—itching, irritation, and pain—that were included in the overall QOL assessment also were analyzed individually. Results also were compared for patients with mild psoriasis alone versus those with moderate to severe psoriasis alone as well as for patients with PsA and mild to no psoriasis versus those with PsA and moderate to severe psoriasis.
Statistical Analysis
Adult survey respondents who answered survey questions relevant to the analysis were included. For patient demographics and comorbidities, t tests were used for continuous variables and Χ2 tests were used for categorical variables. For overall psoriasis-related QOL and symptom impact scores, differences among the study groups were compared using analyses of covariance, which adjusted for age, gender, and race. All analyses were conducted using SAS version 9.2.
Results
Patient Demographics
Of 5467 total respondents, 3532 reported moderate to severe psoriasis of which 1280 (36.2%) had comorbid PsA (Table 1). Compared to patients with moderate to severe psoriasis alone, those with moderate to severe psoriasis and comorbid PsA were older, less commonly male, less likely to be employed, had a longer duration of psoriasis, were more likely to be white, and were more likely to have health insurance coverage. Among the psoriasis alone group, those patients reporting moderate to severe psoriasis were younger, less likely to have health insurance coverage, and were less likely to be treated by a dermatologist only than those with mild psoriasis. Among the group with PsA, patients with moderate to severe psoriasis were more likely to be treated by a dermatologist only versus those with mild or no psoriasis (Table 1).
Patient Comorbidities
Patients with moderate to severe psoriasis and comorbid PsA reported significantly higher rates of diabetes mellitus, lupus, rheumatoid arthritis, other arthritis, ankylosing spondylitis, and high blood pressure than patients with moderate to severe psoriasis alone (P<.05)(Table 2). In the group with psoriasis alone, patients with moderate to severe psoriasis had significantly lower rates of lupus compared to those with mild psoriasis (P<.05). In the group with PsA, patients with moderate to severe psoriasis were significantly more likely to report diagnoses of colitis, diabetes mellitus, heart disease, and high blood pressure compared to those with mild to no psoriasis (P<.05).
Psoriasis-Related QOL
The psoriasis-related QOL analyses are shown in Table 3. Patients with moderate to severe psoriasis and comorbid PsA reported a greater impact on overall QOL compared to their counterparts with moderate to severe psoriasis alone. Similarly, patients with moderate to severe psoriasis and comorbid PsA reported a greater impact of itching, physical irritation/soreness, and pain symptoms.
Disease severity was associated with psoriasis-related QOL. Compared to patients with mild psoriasis alone, those with moderate to severe psoriasis alone had significantly higher (ie, worse) scores in the categories of itching, irritation, pain, and overall QOL (P<.05). In the PsA group, patients with moderate to severe psoriasis also reported significantly worse itching, irritation, pain, and overall QOL compared to their counterparts with mild to no psoriasis (P<.05).
Comment
The results of this study showed that patient demographics, comorbidities, and psoriasis-related QOL in psoriasis patients varied significantly depending on presence or absence of comorbid PsA as well as disease severity. Patients with more severe cutaneous psoriasis with and without PsA were associated with worse QOL scores and higher rates of self-reported heart disease and hypertension. Among patients with moderate to severe psoriasis, those with comorbid PsA were associated with greater rates of self-reported diabetes mellitus and high blood pressure and worse QOL scores including worse itching, irritation, and pain than those with moderate to severe psoriasis alone.
The present study is one of the first to compare psoriasis-related QOL in patients with and without PsA. A prior study demonstrated patients with PsA had worse physical impairment, fatigue severity, and health status relative to patients with psoriasis alone but no differences in dermatology-specific QOL measures.9 Our analysis revealed that patients with moderate to severe psoriasis and comorbid PsA experienced a greater impact on overall QOL as well as symptoms of itching, irritation, and pain relative to patients with moderate to severe psoriasis alone. Among both treatment groups, reports of overall QOL and itching, irritation, and pain became worse as the disease severity increased. These findings are consistent with prior studies that have shown greater impairment of QOL among patients with severe psoriasis compared to mild psoriasis.10-12
Relative to patients with moderate to severe psoriasis alone, those with moderate to severe psoriasis and comorbid PsA reported a greater prevalence of comorbid diagnoses overall, with notably higher rates of type 2 diabetes mellitus, hypertension, and rheumatoid arthritis. These findings are consistent with an earlier report by Husted et al.13
Limitations
The results of this study should be interpreted in light of several important limitations associated with survey data (ie, patient-reported data, no details on length of therapy or comorbid disease, multiple surveys taken over time, respondents aged >18 years only).
Conclusion
This study provides evidence that patients with moderate to severe psoriasis and comorbid PsA have a higher psoriasis-related disease burden compared to those with moderate to severe psoriasis alone, which suggests that physicians may wish to consider this comorbidity when designing patient treatment plans. Our findings also confirm that disease severity is associated with comorbid conditions and psoriasis-related QOL impact in psoriasis patients. These findings highlight the importance of screening for PsA in patients with psoriasis and effective treatment of skin disease in patients with PsA.
Acknowledgment
The authors thank Maureen Lage, PhD, Healthmetrics, Bonita Springs, Florida, for her work in drafting the manuscript.
Approximately 3% of the US adult population, or roughly 5 million Americans, have been diagnosed with psoriasis.1 Psoriasis has a high disease burden, with one review of 817 quality-of-life (QOL) studies concluding that the degree to which psoriasis negatively affects patients’ QOL is comparable with major diseases such as diabetes mellitus or cancer.2
Psoriatic arthritis (PsA) is a chronic, often progressive, inflammatory arthropathy that can lead to permanent joint damage and severe disability.3,4 Psoriatic arthritis is a common comorbidity among patients with psoriasis, affecting 6% to 10% of psoriasis patients overall5 and 20% to 40% of psoriasis patients with more extensive skin involvement.5-7
The comorbidity profile and overall disease impact are not well understood in psoriasis patients with and without comorbid PsA. The primary objective of this study was to compare disease characteristics, comorbidities, and psoriasis-related QOL in patients with moderate to severe psoriasis with and without comorbid PsA.
Methods
Study Design and Participants
Since 2003 the National Psoriasis Foundation (NPF) has conducted semiannual survey panels to collect patient-reported data on topics including disease characteristics, treatment utilization, and psoriasis-related QOL impact. Responses from approximately 5604 individuals have been collected and analyzed from a total of 13 survey panels conducted from 2003-2009 and 2011. No data were available for 2010. In each survey panel, responses from approximately 400 randomly selected respondents were collected, with approximately 300 respondents having moderate to severe psoriasis and 200 respondents being NPF members. Respondents were surveyed via telephone and the Internet.
This study examined 2 main patient groups: (1) adults (>18 years of age) with psoriasis alone and (2) those with comorbid PsA. The 2 study groups were subdivided according to percentage of body surface area (BSA) affected; specifically, patients with psoriasis alone were classified as having mild psoriasis (<3% BSA) or moderate to severe psoriasis (>3% BSA) and patients with PsA were classified as having mild to no psoriasis (<3% BSA) or moderate to severe psoriasis (≥3% BSA).8
Assessments
Comparisons of patient demographics, disease characteristics, comorbidities, psoriasis-related QOL, and symptom impact were made for patients with moderate to severe psoriasis and PsA versus those with moderate to severe psoriasis alone. The overall impact of psoriasis on patients’ physical and emotional QOL was assessed via a 12-item survey with each item rated on an 11-point scale (0=not at all; 5=somewhat; 10=very much). Total QOL scores ranged from 0 to 120. Higher scores indicated greater (ie, worse) impact of psoriasis on the patient’s QOL. To evaluate symptom impact (ie, to what degree a patient felt affected by individual symptoms), ratings from 3 symptoms—itching, irritation, and pain—that were included in the overall QOL assessment also were analyzed individually. Results also were compared for patients with mild psoriasis alone versus those with moderate to severe psoriasis alone as well as for patients with PsA and mild to no psoriasis versus those with PsA and moderate to severe psoriasis.
Statistical Analysis
Adult survey respondents who answered survey questions relevant to the analysis were included. For patient demographics and comorbidities, t tests were used for continuous variables and Χ2 tests were used for categorical variables. For overall psoriasis-related QOL and symptom impact scores, differences among the study groups were compared using analyses of covariance, which adjusted for age, gender, and race. All analyses were conducted using SAS version 9.2.
Results
Patient Demographics
Of 5467 total respondents, 3532 reported moderate to severe psoriasis of which 1280 (36.2%) had comorbid PsA (Table 1). Compared to patients with moderate to severe psoriasis alone, those with moderate to severe psoriasis and comorbid PsA were older, less commonly male, less likely to be employed, had a longer duration of psoriasis, were more likely to be white, and were more likely to have health insurance coverage. Among the psoriasis alone group, those patients reporting moderate to severe psoriasis were younger, less likely to have health insurance coverage, and were less likely to be treated by a dermatologist only than those with mild psoriasis. Among the group with PsA, patients with moderate to severe psoriasis were more likely to be treated by a dermatologist only versus those with mild or no psoriasis (Table 1).
Patient Comorbidities
Patients with moderate to severe psoriasis and comorbid PsA reported significantly higher rates of diabetes mellitus, lupus, rheumatoid arthritis, other arthritis, ankylosing spondylitis, and high blood pressure than patients with moderate to severe psoriasis alone (P<.05)(Table 2). In the group with psoriasis alone, patients with moderate to severe psoriasis had significantly lower rates of lupus compared to those with mild psoriasis (P<.05). In the group with PsA, patients with moderate to severe psoriasis were significantly more likely to report diagnoses of colitis, diabetes mellitus, heart disease, and high blood pressure compared to those with mild to no psoriasis (P<.05).
Psoriasis-Related QOL
The psoriasis-related QOL analyses are shown in Table 3. Patients with moderate to severe psoriasis and comorbid PsA reported a greater impact on overall QOL compared to their counterparts with moderate to severe psoriasis alone. Similarly, patients with moderate to severe psoriasis and comorbid PsA reported a greater impact of itching, physical irritation/soreness, and pain symptoms.
Disease severity was associated with psoriasis-related QOL. Compared to patients with mild psoriasis alone, those with moderate to severe psoriasis alone had significantly higher (ie, worse) scores in the categories of itching, irritation, pain, and overall QOL (P<.05). In the PsA group, patients with moderate to severe psoriasis also reported significantly worse itching, irritation, pain, and overall QOL compared to their counterparts with mild to no psoriasis (P<.05).
Comment
The results of this study showed that patient demographics, comorbidities, and psoriasis-related QOL in psoriasis patients varied significantly depending on presence or absence of comorbid PsA as well as disease severity. Patients with more severe cutaneous psoriasis with and without PsA were associated with worse QOL scores and higher rates of self-reported heart disease and hypertension. Among patients with moderate to severe psoriasis, those with comorbid PsA were associated with greater rates of self-reported diabetes mellitus and high blood pressure and worse QOL scores including worse itching, irritation, and pain than those with moderate to severe psoriasis alone.
The present study is one of the first to compare psoriasis-related QOL in patients with and without PsA. A prior study demonstrated patients with PsA had worse physical impairment, fatigue severity, and health status relative to patients with psoriasis alone but no differences in dermatology-specific QOL measures.9 Our analysis revealed that patients with moderate to severe psoriasis and comorbid PsA experienced a greater impact on overall QOL as well as symptoms of itching, irritation, and pain relative to patients with moderate to severe psoriasis alone. Among both treatment groups, reports of overall QOL and itching, irritation, and pain became worse as the disease severity increased. These findings are consistent with prior studies that have shown greater impairment of QOL among patients with severe psoriasis compared to mild psoriasis.10-12
Relative to patients with moderate to severe psoriasis alone, those with moderate to severe psoriasis and comorbid PsA reported a greater prevalence of comorbid diagnoses overall, with notably higher rates of type 2 diabetes mellitus, hypertension, and rheumatoid arthritis. These findings are consistent with an earlier report by Husted et al.13
Limitations
The results of this study should be interpreted in light of several important limitations associated with survey data (ie, patient-reported data, no details on length of therapy or comorbid disease, multiple surveys taken over time, respondents aged >18 years only).
Conclusion
This study provides evidence that patients with moderate to severe psoriasis and comorbid PsA have a higher psoriasis-related disease burden compared to those with moderate to severe psoriasis alone, which suggests that physicians may wish to consider this comorbidity when designing patient treatment plans. Our findings also confirm that disease severity is associated with comorbid conditions and psoriasis-related QOL impact in psoriasis patients. These findings highlight the importance of screening for PsA in patients with psoriasis and effective treatment of skin disease in patients with PsA.
Acknowledgment
The authors thank Maureen Lage, PhD, Healthmetrics, Bonita Springs, Florida, for her work in drafting the manuscript.
1. Kurd SK, Gelfand JM. The prevalence of previously diagnosed and undiagnosed psoriasis in US adults: results from NHANES 2003-2004. J Am Acad Dermatol. 2009;60:218-224.
2. Raho G, Koleva DM, Garattini L, et al. The burden of moderate to severe psoriasis: an overview. Pharmacoeconomics. 2012;30:1005-1013.
3. Gladman DD, Antoni C, Mease P, et al. Psoriatic arthritis: epidemiology, clinical features, course, and outcome. Ann Rheum Dis. 2005;64(suppl 2):14-17.
4. Ogdie A, Langan S, Love T, et al. Prevalence and treatment patterns of psoriatic arthritis in the UK. Rheumatology (Oxford). 2013;52:568-575.
5. Ogdie A, Gelfand JM. Identification of risk factors for psoriatic arthritis: scientific opportunity meets clinical need. Arch Dermatol. 2010;146:785-788.
6. Gelfand JM, Gladman DD, Mease PJ, et al. Epidemiology of psoriatic arthritis in the population of the United States. J Am Acad Dermatol. 2005;53:573.
7. Richardson SK, Gelfand JM. Update on the natural history and systemic treatment of psoriasis. Adv Dermatol. 2008;24:171-196.
8. Armstrong AW, Robertson AD, Wu J, et al. Undertreatment, treatment trends, and treatment dissatisfaction among patients with psoriasis and psoriatic arthritis in the United States: findings from the National Psoriasis Foundation surveys, 2003-2011. JAMA Dermatol. 2013;149:1180-1185.
9. Rosen CF, Mussani F, Chandran V, et al. Patients with psoriatic arthritis have worse quality of life than those with psoriasis alone. Rheumatology (Oxford). 2012;51:571-576.
10. Armstrong AW, Schupp C, Wu J, et al. Quality of life and work productivity impairment among psoriasis patients: findings from the National Psoriasis Foundation survey data 2003-2011. PLoS ONE. 2012;7:e52935.
11. Colombo G, Altomare G, Peris K, et al. Moderate and severe plaque psoriasis: cost-of-illness study in Italy. Ther Clin Risk Manag. 2008;4:559-568.
12. Gelfand JM, Feldman SR, Stern RS, et al. Determinants of quality of life in patients with psoriasis: a study from the US population. J Am Acad Dermatol. 2004;51:704-708.
13. Husted JA, Thavaneswaran A, Chandran V, et al. Cardiovascular and other comorbidities in patients with psoriatic arthritis: a comparison with patients with psoriasis. Arthritis Care Res (Hoboken). 2011;63:1729-1735.
1. Kurd SK, Gelfand JM. The prevalence of previously diagnosed and undiagnosed psoriasis in US adults: results from NHANES 2003-2004. J Am Acad Dermatol. 2009;60:218-224.
2. Raho G, Koleva DM, Garattini L, et al. The burden of moderate to severe psoriasis: an overview. Pharmacoeconomics. 2012;30:1005-1013.
3. Gladman DD, Antoni C, Mease P, et al. Psoriatic arthritis: epidemiology, clinical features, course, and outcome. Ann Rheum Dis. 2005;64(suppl 2):14-17.
4. Ogdie A, Langan S, Love T, et al. Prevalence and treatment patterns of psoriatic arthritis in the UK. Rheumatology (Oxford). 2013;52:568-575.
5. Ogdie A, Gelfand JM. Identification of risk factors for psoriatic arthritis: scientific opportunity meets clinical need. Arch Dermatol. 2010;146:785-788.
6. Gelfand JM, Gladman DD, Mease PJ, et al. Epidemiology of psoriatic arthritis in the population of the United States. J Am Acad Dermatol. 2005;53:573.
7. Richardson SK, Gelfand JM. Update on the natural history and systemic treatment of psoriasis. Adv Dermatol. 2008;24:171-196.
8. Armstrong AW, Robertson AD, Wu J, et al. Undertreatment, treatment trends, and treatment dissatisfaction among patients with psoriasis and psoriatic arthritis in the United States: findings from the National Psoriasis Foundation surveys, 2003-2011. JAMA Dermatol. 2013;149:1180-1185.
9. Rosen CF, Mussani F, Chandran V, et al. Patients with psoriatic arthritis have worse quality of life than those with psoriasis alone. Rheumatology (Oxford). 2012;51:571-576.
10. Armstrong AW, Schupp C, Wu J, et al. Quality of life and work productivity impairment among psoriasis patients: findings from the National Psoriasis Foundation survey data 2003-2011. PLoS ONE. 2012;7:e52935.
11. Colombo G, Altomare G, Peris K, et al. Moderate and severe plaque psoriasis: cost-of-illness study in Italy. Ther Clin Risk Manag. 2008;4:559-568.
12. Gelfand JM, Feldman SR, Stern RS, et al. Determinants of quality of life in patients with psoriasis: a study from the US population. J Am Acad Dermatol. 2004;51:704-708.
13. Husted JA, Thavaneswaran A, Chandran V, et al. Cardiovascular and other comorbidities in patients with psoriatic arthritis: a comparison with patients with psoriasis. Arthritis Care Res (Hoboken). 2011;63:1729-1735.
Practice Points
- Patients with psoriatic arthritis (PsA) often have severe cutaneous psoriasis. These patients may be at higher risk for comorbid conditions and impaired quality of life (QOL) compared to patients with psoriasis alone.
- The severity of cutaneous psoriasis, both in patients with psoriasis alone and those with comorbid PsA, is remarkable and is associated with worse QOL scores.
Update on Pediatric Psoriasis
Psoriasis affects 2% to 4% of the US population, with approximately one-third of cases beginning in childhood. The understanding of pediatric psoriasis has developed at a far slower pace than adult disease, with limitations in care including few medications that are approved by the US Food and Drug Administration for pediatric and adolescent use. Recently, a stable fixed-combination dose of calcipo-triene 0.005%–betamethasone dipropionate 0.064% topical suspension was approved for treatment of plaque psoriasis of the scalp in patients aged 12 to 17 years, which hopefully will lead a trend in psoriasis medication approval for children and teenagers.1 Based on a PubMed search of articles indexed for MEDLINE using the search terms pediatric psoriasis, psoriasis, and strep that were published from April 2012 to April 2014, this article reviews newer data to address the issues that surround pediatric psoriasis and to provide an update on prior review articles on pediatric psoriasis.2-5 This article reviews some of the newer literature on clinical presentation and comorbidities in pediatric psoriasis.5 Based on these recent findings, additional screenings including review of obesity parameters are recommended for pediatric patients with psoriasis (Table 1).
Update on Disease Manifestations, Associations, and Comorbidities
Disease Manifestations
A 2013 multicenter study delineated the clinical features of pediatric psoriasis.6 The study was conducted at 8 geographically diverse dermatology clinics in the United States to delineate the clinical manifestations of pediatric psoriasis. In an assessment of 181 participants aged 5 to 17 years, the investigators sought to determine the frequency of disease sites, severity, and guttate disease. Over a period of approximately 2 years, 43.1% of participants were determined to have mild disease and 56.9% had severe disease. Family history of psoriasis was present in 51.4% of participants, with first-degree relatives affected in 59.8% of cases. Scalp involvement at some time was noted in 79.0% of participants, and nail disease was noted in 55% of boys and 29% of girls. Guttate psoriasis was noted in 30% of participants, with more cases in the severe range (35.9%) versus the mild range (21.8%). Additionally, 22.1% of participants had a precipitating streptococcal infection, with the association being more common in pediatric patients with guttate psoriasis than plaque psoriasis.6 This study highlighted that pediatric psoriasis has a genetic basis, is frequently guttate in nature, commonly affects the nails, shows a trend toward being classified as severe, and may be triggered by streptococcal infections.
Streptococcal Infection
Pediatric psoriasis may be triggered or flared by Streptococcus pyogenes (group A β-hemolytic streptococci) infections, specifically β-hemolytic streptococci groups A, C, and G that have streptococcal M protein,2,3,7 and this tendency can be associated with HLA-Cw6 or guttate psoriasis. Newer data have elucidated the role of streptococcal throat infections in psoriasis. Given that streptococcal throat infections are most common in school-aged children, these studies suggest a putative mechanism in pediatric psoriasis for triggering streptococcal infections, which would need to be confirmed in future studies, specifically in pediatric psoriasis patients.
It has been shown that T cells in psoriasis patients recognize common streptococcal M proteins and keratin determinants.7 Ferran et al8 recently demonstrated activation of circulating cutaneous lymphocyte–associated antigen (CLA)+ T cells but not CLA- memory T cells in 27 psoriasis patients (ages not specified) when mixed with streptococcal throat extracts, causing production of IL-17, IP-10, IL-22, and IFN-γ; activation was not found in 6 healthy control patients. Antistreptolysin O levels were correlated with the messenger RNA up- regulation for IL-17, IP-10, IL-22, and IFN-γ, and also correlated with psoriasis area and severity index score in psoriasis patients. In this same study, injection of the activated culture supernatant into mouse skin caused epidermal hyperkeratosis and activation of nonlesional epidermal cells from psoriatic patients. This study thereby delineated some of the potential pathways of the streptococcal induction of psoriasis and psoriatic flares in childhood8; however, confirmation is needed through further study of pediatric psoriatic lymphocyte activity.
Differential Diagnosis
Additions to the extensive differential list have been cited in the recent literature. The differential diagnosis of pediatric psoriasis now includes sodium valproate–induced psoriasiform drug eruption9 and allergic contact dermatitis to methylchloroisothiazolinone and methylisothiazolinone, which are present in many sanitizing hand and diaper wipes and has been reported to cause psoriasiform dermatitis in a periorificial or perineal distribution.10 Clinicians should inquire about the use of these wipes, as caregivers rarely suspect this agent to be causative of the eruption.
Psoriatic Arthritis
Previously, psoriasis and psoriatic arthritis have been linked to autoimmune thyroid disease in adults.11 A study of the Childhood Arthritis & Rheumatology Research Alliance (CARRA) registry showed that family history of psoriasis, autoimmune thyroiditis, Crohn disease, and ankylosing spondylitis in a first-degree relative has been linked to juvenile idiopathic arthritis, highlighting that pediatric psoriasis can be genetically linked or associated with multiple autoimmune conditions and vice versa.12
Obesity, Metabolic Syndrome, and Cardiovascular Risks
Obesity is associated with pediatric psoriasis as highlighted in a growing body of recent literature.13 Excess adiposity as manifested by body mass index in the 85th percentile or greater (37.9% of 155 pediatric psoriasis patients vs 20.5% of 42 controls) and excess central adiposity as manifested by excess waist circumference and increased waist-to-height ratios are more common in pediatric patients with psoriasis than in controls.14
Obesity may be a trigger or associated with increased disease activity in pediatric psoriasis patients. Excess overall adiposity correlates with more severe disease. Obesity parameters may correlate with the onset of psoriasis and with disease severity. In fact, the odds of obesity may be higher in childhood than in adults.14,15 A 2011 report of pediatric psoriasis patients aged 10 to 17 years (n=12) and wart controls (n=6)(mean age, 13.2 and 13.5 years, respectively) demonstrated that 4 of 12 patients with psoriasis and 0 of 6 patients with warts met criteria for metabolic syndrome as defined by 3 of the following: (1) triglycerides greater than or equal to 100 mg/dL; (2) high-density lipoprotein cholesterol less than 50 mg/dL in females and less than 5 mg/dL in males; (3) fasting blood glucose levels greater than or equal to 110 mg/dL, (4) waist circumference greater than the 75th percentile for age and sex; and (5) systolic or diastolic blood pressure greater than the 90th percentile for age, sex, and height.16 These studies highlight that obesity and metabolic syndrome are of concern in pediatric psoriasis patients; however, the best management approach using diet and weight interventions has yet to be identified.
Adiposity may precede the onset of psoriasis. A recent cohort of 27 pediatric psoriasis patients reported that the average age at onset of psoriasis was 8.7 years and the average age at onset of obesity was 4.1 years.15 In this study, 93% (25/27) of patients had adiposity preceding their psoriasis by 2 or more years. It is unclear if this is nature or nurture, as 48% (13/27) of patients had a family history of obesity, 41% (11/27) had a family history of psoriasis, and 48% (13/27) had a family history of hyperlipidemia.15 Therefore, obesity may be cultivated in some psoriatic families. The issue of household influences on diet and obesity needs to be addressed if successful weight management is to be achieved in future studies of pediatric psoriasis.
Cardiovascular risks in the pediatric psoriasis population are the subject of ongoing assessment but will likely mimic studies of adult psoriasis patients when reviewed longitudinally.16 Weight loss and healthy lifestyle interventions likely are beneficial to long-term health, but there is a lack of published data addressing dietary modification as a disease modifier for long-term care of pediatric psoriasis.
Anxiety and Depression
Anxiety and depression have been noted in adults with chronic skin diseases. A recent study assessed 118 patients and caregivers of pediatric patients with atopic dermatitis (n=50), psoriasis (n=25), or vitiligo (n=43) using the Children’s Dermatology Life Quality Index, the Hamilton Anxiety Scale, and the Beck Depression Inventory.17 Anxiety and depression were found in 36% of caregivers of pediatric psoriasis patients and depression was found in 36% of pediatric psoriasis patients, highlighting the need for interventions on a personal and family level to improve quality of life. As a comparator, anxiety was more prevalent in vitiligo caregivers (42%), but depression was only found in 26% of caregivers in the same group. Extent of disease (25%–75% body surface area affected) correlated with both depression and anxiety in the caregivers of pediatric patients with psoriasis as well as with anxiety in caregivers of pediatric patients with increased visible surface area of vitiligo.17 Parental anxiety has been reported at times to be linked to corticosteroid phobia, or corticophobia, which may interfere with disease therapy, as topical corticosteroids are considered the mainstay of therapy in childhood disease.18 Coordinating care with caregivers and addressing their concerns about the safety of medications should be integral to the pediatric psoriasis visit.
Pustular Psoriasis
Pustular psoriasis can be seen in any age group. Researchers recently have attempted to delineate the features and successful management of this severe subset of pediatric psoriasis patients. Twenty-four pediatric pustular psoriasis cases reviewed by Posso-De Los Rios et al19 revealed that 92% (22/24) had generalized and 8% (2/24) had limited acral disease. The mean (standard deviation) age at onset of pediatric pustular psoriasis was 6.3 (4.9) years. Half of the reported cases required more than one intervention. Treatment with acitretin, cyclosporine, and methotrexate was effective, but the investigators identified that there is a true dearth of evidence-based therapeutics in pediatric pustular psoriasis and much rebound with discontinuation.19 Although the subset of pediatric pustular psoriasis is rare, study of evidence-based intervention is needed.
Therapy
Recent reviews of pediatric and adolescent psoriasis highlight the paucity of therapeutic information for these patient populations. Investigators typically focus on topical therapies as the basis of treatment,20 as well as the addition of phototherapy in mild to moderate plaque or guttate psoriasis and biologic or systemic agents in moderate to severe flares of plaque, erythrodermic, or pustular psoriasis.21 Further studies are needed to identify evidence-based therapeutic paradigms for pediatric psoriasis and to pinpoint therapies associated with the best quality of life in patients and their caregivers.
Tumor Necrosis Factor α Inhibitors
Safety and efficacy of etanercept for juvenile idiopathic arthritis including oligoarthritis, enthesitis-related arthritis, and psoriatic arthritis recently was reviewed by Windschall et al22 using data from the German pediatric Biologika in der Kinderrheumatologie registry. Juvenile Arthritis Disease Activity Score 10 improved from baseline for 127 pediatric patients with psoriatic arthritis in 3 to 24 months (mean [standard deviation], 14.7 [6.4], 5.0 [4.6], 5.3 [6.4] at baseline, 3 months, and 24 months, respectively). Overall side effects were relatively higher in the psoriatic arthritis group; the rate of serious (relative risk, 1.39 [0.95-2.03; P=.08]) and nonserious (relative risk, 1.18 [1.02-1.35; P=.03]) adverse events also was elevated. Uveitis risk was greatest in the psoriatic arthritis group and the number of associated cases of inflammatory bowel disease outnumbered those seen in other forms of arthritis. The investigators concluded that monitoring for extra-articular immunopathies should be conducted in pediatric patients with psoriatic arthritis who are undergoing etanercept therapy.22
Tumor necrosis factor α (TNF-α) inhibitors have been associated with triggering psoriasiform dermatitis in pediatric patients treated for inflammatory bowel disease. A Finnish study of infliximab side effects in pediatric patients with inflammatory bowel disease (n=84; Crohn disease: n=64) demonstrated that almost half (47.6% [40/84]) of the participants presented with chronic skin reactions, 23% of which were severe in nature.23 Psoriasiform lesions of the scalp and ears were most common, followed by the periorificial area, genitals, trunk, and extremities. Rare association with HLA-Cw*0602 genotype was noted. Skin manifestations did not correlate with gut inflammation (as determined by fecal calprotectin levels). Discontinuation of therapy rarely was required.23 Other studies also have highlighted this side effect, suggesting an incidence of 2.7% in adults with colitis treated with TNF-α inhibitors24 and 10.5% in pediatric patients with Crohn disease.25 In a study by Sherlock et al,25 pediatric patients with Crohn disease developing psoriasis following infliximab therapy were more likely to be homozygous for specific polymorphisms in the IL-23R gene (rs10489628, rs10789229, and rs1343151).
Methotrexate
For pediatric patients who are being treated with methotrexate, the polyglutamate assay recently has been reported to be helpful in identifying patients needing a dose escalation.26 Higher numbers on the polyglutamate assay are associated with superior response to methotrexate therapy. Doses can be increased after 12 weeks in patients with low assays.26
IL-23
The safety of IL-23 blockade in pediatric psoriasis patients has not yet been established, but data from adult cases have implicated the IL-17 and IL-23 pathways in psoriasis/psoriatic arthritis, including an association with IL-23R polymorphisms27 and increases in soluble IL-20 and IL-22 associated with disease severity and an association of IL-17 levels with activity on the psoriasis area and severity index scores.28 The data are more limited for pediatric cases. Pediatric patients with inflammatory bowel disease who have an IL-23R polymorphism appear to be susceptible to psoriatic flares while on TNF-α inhibitor therapy,25 which suggests that the IL-23 blockade may be of benefit for some pediatric patients with psoriasis or psoriatic arthritis.
Conclusion
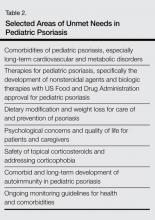
Pediatric psoriasis and psoriatic arthritis have now been identified as being part of the autoimmune spectrum and are associated with metabolic syndrome, including obesity and excess central adiposity, similar to their adult variants. An overview of potential unmet needs in pediatric psoriasis is included in Table 2. These unmet needs include further delineation of diet and weight modification in the care and prevention of psoriasis; expansion of therapeutic trials and US Food and Drug Administration–approved medications for children with psoriasis, especially severe variants such as extensive plaque and pustular disease; and development of guidelines for ongoing monitoring of children with psoriasis. The role of therapeutic interventions and weight management on long-term disease course remains to be shown in extended clinical trials. Despite the great advancements in psoriatic care, knowledge gaps remain in pediatric psoriasis that will need to be addressed in the future.
1. Taclonex Expanded Indication. OptumRx Web site. https://www.optumrx.com/vgnpreview/HCP/Assets/RxNews/Clinical%20Updates_Taclonex_2014-1003.pdf. Published August 29, 2014. Accessed January 28, 2015.
2. Silverberg NB. Update on pediatric psoriasis, part 1: clinical features and demographics. Cutis. 2010;86:118-124.
3. Silverberg NB. Update on pediatric psoriasis, part 2: therapeutic management. Cutis. 2010;86:172-176.
4. Cather JC. Psoriasis in children and women: addressing some special needs. Semin Cutan Med Surg. 2014;33(2 suppl 2):S42-S44.
5. Khorsand K, Sidbury R. Recent advances in pediatric dermatology. Arch Dis Child. 2014;99:944-948.
6. Mercy K, Kwasny M, Cordoro KM, et al. Clinical manifestations of pediatric psoriasis: results of a multicenter study in the United States. Pediatr Dermatol. 2013;30:424-428.
7. Gudjonsson JE, Thorarinsson AM, Sigurgeirsson B, et al. Streptococcal throat infections and exacerbation of chronic plaque psoriasis: a prospective study. Br J Dermatol. 2003;149:530-534.
8. Ferran M, Galván AB, Rincón C, et al. Streptococcus induces circulating CLA(+) memory T-cell-dependent epidermal cell activation in psoriasis. J Invest Dermatol. 2013;133:999-1007.
9. Gul Mert G, Incecik F, Gunasti S, et al. Psoriasiform drug eruption associated with sodium valproate [published online ahead of print November 13, 2013]. Case Rep Pediatr. 2013;2013:823469.
10. Chang MW, Nakrani R. Six children with allergic contact dermatitis to methylisothiazolinone in wet wipes (baby wipes). Pediatrics. 2014;133:e434-e438.
11. Gul U, Gonul M, Kaya I, et al. Autoimmune thyroid disorders in patients with psoriasis. Eur J Dermatol. 2009;19:221-223.
12. Prahalad S, McCracken C, Ponder L, et al. A120: Familial autoimmunity in the CARRA registry. Arthritis Rheumatol. 2014;66(suppl 11):S157.
13. Mercy KM, Paller AS. The relationship between obesity and psoriasis in the pediatric population: implications and future directions. Cutis. 2013;92:107-109.
14. Paller AS, Mercy K, Kwasny MJ, et al. Association of pediatric psoriasis severity with excess and central adiposity: an international cross-sectional study. JAMA Dermatol. 2013;149:166-176.
15. Becker L, Tom WL, Eshagh K, et al. Excess adiposity preceding pediatric psoriasis. JAMA Dermatol. 2014;150:573-574.
16. Volf EM, Levine DE, Michelon MA, et al. Assessor-blinded study of the metabolic syndrome and surrogate markers of increased cardiovascular risk in children with moderate-to-severe psoriasis compared with age-matched population of children with warts. J Drugs Dermatol. 2011;10:900-901.
17. Manzoni AP, Weber MB, Nagatomi AR, et al. Assessing depression and anxiety in the caregivers of pediatric patients with chronic skin disorders. An Bras Dermatol. 2013;88:894-899.
18. Belloni Fortina A, Neri L. Topical steroids and corticophobia. G Ital Dermatol Venereol. 2013;148:651-654.
19. Posso-De Los Rios CJ, Pope E, Lara-Corrales I. A systematic review of systemic medications for pustular psoriasis in pediatrics. Pediatr Dermatol. 2014;31:430-439.
20. Tollefson MM. Diagnosis and management of psoriasis in children. Pediatr Clin North Am. 2014;61:261-277.
21. Fotiadou C, Lazaridou E, Ioannides D. Management of psoriasis in adolescence. Adolesc Health Med Ther. 2014;5:25-34.
22. Windschall D, Müller T, Becker I, et al. Safety and efficacy of etanercept in children with the JIA categories extended oligoarthritis, enthesitis-related arthritis and psoriasis arthritis [published online ahead of print July 18, 2014]. Clin Rheumatol. 2015;34:61-69.
23. Mälkönen T, Wikström A, Heiskanen K, et al. Skin reactions during anti-TNFa therapy for pediatric inflammatory bowel disease: a 2-year prospective study. Inflamm Bowel Dis. 2014;20:1309-1315.
24. Afzali A, Wheat CL, Hu JK, et al. The association of psoriasiform rash with anti-tumor necrosis factor (anti-TNF) therapy in inflammatory bowel disease: a single academic center case series. J Crohns Colitis. 2014;8:480-488.
25. Sherlock ME, Walters T, Tabbers MM, et al. Infliximab-induced psoriasis and psoriasiform skin lesions in pediatric Crohn disease and a potential association with IL-23 receptor polymorphisms. J Pediatr Gastroenterol Nutr. 2013;56:512-518.
26. Rahman SI, Siegfried E, Flanagan KH, et al. The methotrexate polyglutamate assay supports the efficacy of methotrexate for severe inflammatory skin disease in children. J Am Acad Dermatol. 2014;70:252-256.
27. Suzuki E, Mellins ED, Gershwin ME, et al. The IL-23/IL-17 axis in psoriatic arthritis. Autoimmun Rev. 2014;13:496-502.
28. Michalak-Stoma A, Bartosi´nska J, Kowal M, et al. Serum levels of selected Th17 and Th22 cytokines in psoriatic patients. Dis Markers. 2013;35:625-631.
Psoriasis affects 2% to 4% of the US population, with approximately one-third of cases beginning in childhood. The understanding of pediatric psoriasis has developed at a far slower pace than adult disease, with limitations in care including few medications that are approved by the US Food and Drug Administration for pediatric and adolescent use. Recently, a stable fixed-combination dose of calcipo-triene 0.005%–betamethasone dipropionate 0.064% topical suspension was approved for treatment of plaque psoriasis of the scalp in patients aged 12 to 17 years, which hopefully will lead a trend in psoriasis medication approval for children and teenagers.1 Based on a PubMed search of articles indexed for MEDLINE using the search terms pediatric psoriasis, psoriasis, and strep that were published from April 2012 to April 2014, this article reviews newer data to address the issues that surround pediatric psoriasis and to provide an update on prior review articles on pediatric psoriasis.2-5 This article reviews some of the newer literature on clinical presentation and comorbidities in pediatric psoriasis.5 Based on these recent findings, additional screenings including review of obesity parameters are recommended for pediatric patients with psoriasis (Table 1).
Update on Disease Manifestations, Associations, and Comorbidities
Disease Manifestations
A 2013 multicenter study delineated the clinical features of pediatric psoriasis.6 The study was conducted at 8 geographically diverse dermatology clinics in the United States to delineate the clinical manifestations of pediatric psoriasis. In an assessment of 181 participants aged 5 to 17 years, the investigators sought to determine the frequency of disease sites, severity, and guttate disease. Over a period of approximately 2 years, 43.1% of participants were determined to have mild disease and 56.9% had severe disease. Family history of psoriasis was present in 51.4% of participants, with first-degree relatives affected in 59.8% of cases. Scalp involvement at some time was noted in 79.0% of participants, and nail disease was noted in 55% of boys and 29% of girls. Guttate psoriasis was noted in 30% of participants, with more cases in the severe range (35.9%) versus the mild range (21.8%). Additionally, 22.1% of participants had a precipitating streptococcal infection, with the association being more common in pediatric patients with guttate psoriasis than plaque psoriasis.6 This study highlighted that pediatric psoriasis has a genetic basis, is frequently guttate in nature, commonly affects the nails, shows a trend toward being classified as severe, and may be triggered by streptococcal infections.
Streptococcal Infection
Pediatric psoriasis may be triggered or flared by Streptococcus pyogenes (group A β-hemolytic streptococci) infections, specifically β-hemolytic streptococci groups A, C, and G that have streptococcal M protein,2,3,7 and this tendency can be associated with HLA-Cw6 or guttate psoriasis. Newer data have elucidated the role of streptococcal throat infections in psoriasis. Given that streptococcal throat infections are most common in school-aged children, these studies suggest a putative mechanism in pediatric psoriasis for triggering streptococcal infections, which would need to be confirmed in future studies, specifically in pediatric psoriasis patients.
It has been shown that T cells in psoriasis patients recognize common streptococcal M proteins and keratin determinants.7 Ferran et al8 recently demonstrated activation of circulating cutaneous lymphocyte–associated antigen (CLA)+ T cells but not CLA- memory T cells in 27 psoriasis patients (ages not specified) when mixed with streptococcal throat extracts, causing production of IL-17, IP-10, IL-22, and IFN-γ; activation was not found in 6 healthy control patients. Antistreptolysin O levels were correlated with the messenger RNA up- regulation for IL-17, IP-10, IL-22, and IFN-γ, and also correlated with psoriasis area and severity index score in psoriasis patients. In this same study, injection of the activated culture supernatant into mouse skin caused epidermal hyperkeratosis and activation of nonlesional epidermal cells from psoriatic patients. This study thereby delineated some of the potential pathways of the streptococcal induction of psoriasis and psoriatic flares in childhood8; however, confirmation is needed through further study of pediatric psoriatic lymphocyte activity.
Differential Diagnosis
Additions to the extensive differential list have been cited in the recent literature. The differential diagnosis of pediatric psoriasis now includes sodium valproate–induced psoriasiform drug eruption9 and allergic contact dermatitis to methylchloroisothiazolinone and methylisothiazolinone, which are present in many sanitizing hand and diaper wipes and has been reported to cause psoriasiform dermatitis in a periorificial or perineal distribution.10 Clinicians should inquire about the use of these wipes, as caregivers rarely suspect this agent to be causative of the eruption.
Psoriatic Arthritis
Previously, psoriasis and psoriatic arthritis have been linked to autoimmune thyroid disease in adults.11 A study of the Childhood Arthritis & Rheumatology Research Alliance (CARRA) registry showed that family history of psoriasis, autoimmune thyroiditis, Crohn disease, and ankylosing spondylitis in a first-degree relative has been linked to juvenile idiopathic arthritis, highlighting that pediatric psoriasis can be genetically linked or associated with multiple autoimmune conditions and vice versa.12
Obesity, Metabolic Syndrome, and Cardiovascular Risks
Obesity is associated with pediatric psoriasis as highlighted in a growing body of recent literature.13 Excess adiposity as manifested by body mass index in the 85th percentile or greater (37.9% of 155 pediatric psoriasis patients vs 20.5% of 42 controls) and excess central adiposity as manifested by excess waist circumference and increased waist-to-height ratios are more common in pediatric patients with psoriasis than in controls.14
Obesity may be a trigger or associated with increased disease activity in pediatric psoriasis patients. Excess overall adiposity correlates with more severe disease. Obesity parameters may correlate with the onset of psoriasis and with disease severity. In fact, the odds of obesity may be higher in childhood than in adults.14,15 A 2011 report of pediatric psoriasis patients aged 10 to 17 years (n=12) and wart controls (n=6)(mean age, 13.2 and 13.5 years, respectively) demonstrated that 4 of 12 patients with psoriasis and 0 of 6 patients with warts met criteria for metabolic syndrome as defined by 3 of the following: (1) triglycerides greater than or equal to 100 mg/dL; (2) high-density lipoprotein cholesterol less than 50 mg/dL in females and less than 5 mg/dL in males; (3) fasting blood glucose levels greater than or equal to 110 mg/dL, (4) waist circumference greater than the 75th percentile for age and sex; and (5) systolic or diastolic blood pressure greater than the 90th percentile for age, sex, and height.16 These studies highlight that obesity and metabolic syndrome are of concern in pediatric psoriasis patients; however, the best management approach using diet and weight interventions has yet to be identified.
Adiposity may precede the onset of psoriasis. A recent cohort of 27 pediatric psoriasis patients reported that the average age at onset of psoriasis was 8.7 years and the average age at onset of obesity was 4.1 years.15 In this study, 93% (25/27) of patients had adiposity preceding their psoriasis by 2 or more years. It is unclear if this is nature or nurture, as 48% (13/27) of patients had a family history of obesity, 41% (11/27) had a family history of psoriasis, and 48% (13/27) had a family history of hyperlipidemia.15 Therefore, obesity may be cultivated in some psoriatic families. The issue of household influences on diet and obesity needs to be addressed if successful weight management is to be achieved in future studies of pediatric psoriasis.
Cardiovascular risks in the pediatric psoriasis population are the subject of ongoing assessment but will likely mimic studies of adult psoriasis patients when reviewed longitudinally.16 Weight loss and healthy lifestyle interventions likely are beneficial to long-term health, but there is a lack of published data addressing dietary modification as a disease modifier for long-term care of pediatric psoriasis.
Anxiety and Depression
Anxiety and depression have been noted in adults with chronic skin diseases. A recent study assessed 118 patients and caregivers of pediatric patients with atopic dermatitis (n=50), psoriasis (n=25), or vitiligo (n=43) using the Children’s Dermatology Life Quality Index, the Hamilton Anxiety Scale, and the Beck Depression Inventory.17 Anxiety and depression were found in 36% of caregivers of pediatric psoriasis patients and depression was found in 36% of pediatric psoriasis patients, highlighting the need for interventions on a personal and family level to improve quality of life. As a comparator, anxiety was more prevalent in vitiligo caregivers (42%), but depression was only found in 26% of caregivers in the same group. Extent of disease (25%–75% body surface area affected) correlated with both depression and anxiety in the caregivers of pediatric patients with psoriasis as well as with anxiety in caregivers of pediatric patients with increased visible surface area of vitiligo.17 Parental anxiety has been reported at times to be linked to corticosteroid phobia, or corticophobia, which may interfere with disease therapy, as topical corticosteroids are considered the mainstay of therapy in childhood disease.18 Coordinating care with caregivers and addressing their concerns about the safety of medications should be integral to the pediatric psoriasis visit.
Pustular Psoriasis
Pustular psoriasis can be seen in any age group. Researchers recently have attempted to delineate the features and successful management of this severe subset of pediatric psoriasis patients. Twenty-four pediatric pustular psoriasis cases reviewed by Posso-De Los Rios et al19 revealed that 92% (22/24) had generalized and 8% (2/24) had limited acral disease. The mean (standard deviation) age at onset of pediatric pustular psoriasis was 6.3 (4.9) years. Half of the reported cases required more than one intervention. Treatment with acitretin, cyclosporine, and methotrexate was effective, but the investigators identified that there is a true dearth of evidence-based therapeutics in pediatric pustular psoriasis and much rebound with discontinuation.19 Although the subset of pediatric pustular psoriasis is rare, study of evidence-based intervention is needed.
Therapy
Recent reviews of pediatric and adolescent psoriasis highlight the paucity of therapeutic information for these patient populations. Investigators typically focus on topical therapies as the basis of treatment,20 as well as the addition of phototherapy in mild to moderate plaque or guttate psoriasis and biologic or systemic agents in moderate to severe flares of plaque, erythrodermic, or pustular psoriasis.21 Further studies are needed to identify evidence-based therapeutic paradigms for pediatric psoriasis and to pinpoint therapies associated with the best quality of life in patients and their caregivers.
Tumor Necrosis Factor α Inhibitors
Safety and efficacy of etanercept for juvenile idiopathic arthritis including oligoarthritis, enthesitis-related arthritis, and psoriatic arthritis recently was reviewed by Windschall et al22 using data from the German pediatric Biologika in der Kinderrheumatologie registry. Juvenile Arthritis Disease Activity Score 10 improved from baseline for 127 pediatric patients with psoriatic arthritis in 3 to 24 months (mean [standard deviation], 14.7 [6.4], 5.0 [4.6], 5.3 [6.4] at baseline, 3 months, and 24 months, respectively). Overall side effects were relatively higher in the psoriatic arthritis group; the rate of serious (relative risk, 1.39 [0.95-2.03; P=.08]) and nonserious (relative risk, 1.18 [1.02-1.35; P=.03]) adverse events also was elevated. Uveitis risk was greatest in the psoriatic arthritis group and the number of associated cases of inflammatory bowel disease outnumbered those seen in other forms of arthritis. The investigators concluded that monitoring for extra-articular immunopathies should be conducted in pediatric patients with psoriatic arthritis who are undergoing etanercept therapy.22
Tumor necrosis factor α (TNF-α) inhibitors have been associated with triggering psoriasiform dermatitis in pediatric patients treated for inflammatory bowel disease. A Finnish study of infliximab side effects in pediatric patients with inflammatory bowel disease (n=84; Crohn disease: n=64) demonstrated that almost half (47.6% [40/84]) of the participants presented with chronic skin reactions, 23% of which were severe in nature.23 Psoriasiform lesions of the scalp and ears were most common, followed by the periorificial area, genitals, trunk, and extremities. Rare association with HLA-Cw*0602 genotype was noted. Skin manifestations did not correlate with gut inflammation (as determined by fecal calprotectin levels). Discontinuation of therapy rarely was required.23 Other studies also have highlighted this side effect, suggesting an incidence of 2.7% in adults with colitis treated with TNF-α inhibitors24 and 10.5% in pediatric patients with Crohn disease.25 In a study by Sherlock et al,25 pediatric patients with Crohn disease developing psoriasis following infliximab therapy were more likely to be homozygous for specific polymorphisms in the IL-23R gene (rs10489628, rs10789229, and rs1343151).
Methotrexate
For pediatric patients who are being treated with methotrexate, the polyglutamate assay recently has been reported to be helpful in identifying patients needing a dose escalation.26 Higher numbers on the polyglutamate assay are associated with superior response to methotrexate therapy. Doses can be increased after 12 weeks in patients with low assays.26
IL-23
The safety of IL-23 blockade in pediatric psoriasis patients has not yet been established, but data from adult cases have implicated the IL-17 and IL-23 pathways in psoriasis/psoriatic arthritis, including an association with IL-23R polymorphisms27 and increases in soluble IL-20 and IL-22 associated with disease severity and an association of IL-17 levels with activity on the psoriasis area and severity index scores.28 The data are more limited for pediatric cases. Pediatric patients with inflammatory bowel disease who have an IL-23R polymorphism appear to be susceptible to psoriatic flares while on TNF-α inhibitor therapy,25 which suggests that the IL-23 blockade may be of benefit for some pediatric patients with psoriasis or psoriatic arthritis.
Conclusion
Pediatric psoriasis and psoriatic arthritis have now been identified as being part of the autoimmune spectrum and are associated with metabolic syndrome, including obesity and excess central adiposity, similar to their adult variants. An overview of potential unmet needs in pediatric psoriasis is included in Table 2. These unmet needs include further delineation of diet and weight modification in the care and prevention of psoriasis; expansion of therapeutic trials and US Food and Drug Administration–approved medications for children with psoriasis, especially severe variants such as extensive plaque and pustular disease; and development of guidelines for ongoing monitoring of children with psoriasis. The role of therapeutic interventions and weight management on long-term disease course remains to be shown in extended clinical trials. Despite the great advancements in psoriatic care, knowledge gaps remain in pediatric psoriasis that will need to be addressed in the future.
Psoriasis affects 2% to 4% of the US population, with approximately one-third of cases beginning in childhood. The understanding of pediatric psoriasis has developed at a far slower pace than adult disease, with limitations in care including few medications that are approved by the US Food and Drug Administration for pediatric and adolescent use. Recently, a stable fixed-combination dose of calcipo-triene 0.005%–betamethasone dipropionate 0.064% topical suspension was approved for treatment of plaque psoriasis of the scalp in patients aged 12 to 17 years, which hopefully will lead a trend in psoriasis medication approval for children and teenagers.1 Based on a PubMed search of articles indexed for MEDLINE using the search terms pediatric psoriasis, psoriasis, and strep that were published from April 2012 to April 2014, this article reviews newer data to address the issues that surround pediatric psoriasis and to provide an update on prior review articles on pediatric psoriasis.2-5 This article reviews some of the newer literature on clinical presentation and comorbidities in pediatric psoriasis.5 Based on these recent findings, additional screenings including review of obesity parameters are recommended for pediatric patients with psoriasis (Table 1).
Update on Disease Manifestations, Associations, and Comorbidities
Disease Manifestations
A 2013 multicenter study delineated the clinical features of pediatric psoriasis.6 The study was conducted at 8 geographically diverse dermatology clinics in the United States to delineate the clinical manifestations of pediatric psoriasis. In an assessment of 181 participants aged 5 to 17 years, the investigators sought to determine the frequency of disease sites, severity, and guttate disease. Over a period of approximately 2 years, 43.1% of participants were determined to have mild disease and 56.9% had severe disease. Family history of psoriasis was present in 51.4% of participants, with first-degree relatives affected in 59.8% of cases. Scalp involvement at some time was noted in 79.0% of participants, and nail disease was noted in 55% of boys and 29% of girls. Guttate psoriasis was noted in 30% of participants, with more cases in the severe range (35.9%) versus the mild range (21.8%). Additionally, 22.1% of participants had a precipitating streptococcal infection, with the association being more common in pediatric patients with guttate psoriasis than plaque psoriasis.6 This study highlighted that pediatric psoriasis has a genetic basis, is frequently guttate in nature, commonly affects the nails, shows a trend toward being classified as severe, and may be triggered by streptococcal infections.
Streptococcal Infection
Pediatric psoriasis may be triggered or flared by Streptococcus pyogenes (group A β-hemolytic streptococci) infections, specifically β-hemolytic streptococci groups A, C, and G that have streptococcal M protein,2,3,7 and this tendency can be associated with HLA-Cw6 or guttate psoriasis. Newer data have elucidated the role of streptococcal throat infections in psoriasis. Given that streptococcal throat infections are most common in school-aged children, these studies suggest a putative mechanism in pediatric psoriasis for triggering streptococcal infections, which would need to be confirmed in future studies, specifically in pediatric psoriasis patients.
It has been shown that T cells in psoriasis patients recognize common streptococcal M proteins and keratin determinants.7 Ferran et al8 recently demonstrated activation of circulating cutaneous lymphocyte–associated antigen (CLA)+ T cells but not CLA- memory T cells in 27 psoriasis patients (ages not specified) when mixed with streptococcal throat extracts, causing production of IL-17, IP-10, IL-22, and IFN-γ; activation was not found in 6 healthy control patients. Antistreptolysin O levels were correlated with the messenger RNA up- regulation for IL-17, IP-10, IL-22, and IFN-γ, and also correlated with psoriasis area and severity index score in psoriasis patients. In this same study, injection of the activated culture supernatant into mouse skin caused epidermal hyperkeratosis and activation of nonlesional epidermal cells from psoriatic patients. This study thereby delineated some of the potential pathways of the streptococcal induction of psoriasis and psoriatic flares in childhood8; however, confirmation is needed through further study of pediatric psoriatic lymphocyte activity.
Differential Diagnosis
Additions to the extensive differential list have been cited in the recent literature. The differential diagnosis of pediatric psoriasis now includes sodium valproate–induced psoriasiform drug eruption9 and allergic contact dermatitis to methylchloroisothiazolinone and methylisothiazolinone, which are present in many sanitizing hand and diaper wipes and has been reported to cause psoriasiform dermatitis in a periorificial or perineal distribution.10 Clinicians should inquire about the use of these wipes, as caregivers rarely suspect this agent to be causative of the eruption.
Psoriatic Arthritis
Previously, psoriasis and psoriatic arthritis have been linked to autoimmune thyroid disease in adults.11 A study of the Childhood Arthritis & Rheumatology Research Alliance (CARRA) registry showed that family history of psoriasis, autoimmune thyroiditis, Crohn disease, and ankylosing spondylitis in a first-degree relative has been linked to juvenile idiopathic arthritis, highlighting that pediatric psoriasis can be genetically linked or associated with multiple autoimmune conditions and vice versa.12
Obesity, Metabolic Syndrome, and Cardiovascular Risks
Obesity is associated with pediatric psoriasis as highlighted in a growing body of recent literature.13 Excess adiposity as manifested by body mass index in the 85th percentile or greater (37.9% of 155 pediatric psoriasis patients vs 20.5% of 42 controls) and excess central adiposity as manifested by excess waist circumference and increased waist-to-height ratios are more common in pediatric patients with psoriasis than in controls.14
Obesity may be a trigger or associated with increased disease activity in pediatric psoriasis patients. Excess overall adiposity correlates with more severe disease. Obesity parameters may correlate with the onset of psoriasis and with disease severity. In fact, the odds of obesity may be higher in childhood than in adults.14,15 A 2011 report of pediatric psoriasis patients aged 10 to 17 years (n=12) and wart controls (n=6)(mean age, 13.2 and 13.5 years, respectively) demonstrated that 4 of 12 patients with psoriasis and 0 of 6 patients with warts met criteria for metabolic syndrome as defined by 3 of the following: (1) triglycerides greater than or equal to 100 mg/dL; (2) high-density lipoprotein cholesterol less than 50 mg/dL in females and less than 5 mg/dL in males; (3) fasting blood glucose levels greater than or equal to 110 mg/dL, (4) waist circumference greater than the 75th percentile for age and sex; and (5) systolic or diastolic blood pressure greater than the 90th percentile for age, sex, and height.16 These studies highlight that obesity and metabolic syndrome are of concern in pediatric psoriasis patients; however, the best management approach using diet and weight interventions has yet to be identified.
Adiposity may precede the onset of psoriasis. A recent cohort of 27 pediatric psoriasis patients reported that the average age at onset of psoriasis was 8.7 years and the average age at onset of obesity was 4.1 years.15 In this study, 93% (25/27) of patients had adiposity preceding their psoriasis by 2 or more years. It is unclear if this is nature or nurture, as 48% (13/27) of patients had a family history of obesity, 41% (11/27) had a family history of psoriasis, and 48% (13/27) had a family history of hyperlipidemia.15 Therefore, obesity may be cultivated in some psoriatic families. The issue of household influences on diet and obesity needs to be addressed if successful weight management is to be achieved in future studies of pediatric psoriasis.
Cardiovascular risks in the pediatric psoriasis population are the subject of ongoing assessment but will likely mimic studies of adult psoriasis patients when reviewed longitudinally.16 Weight loss and healthy lifestyle interventions likely are beneficial to long-term health, but there is a lack of published data addressing dietary modification as a disease modifier for long-term care of pediatric psoriasis.
Anxiety and Depression
Anxiety and depression have been noted in adults with chronic skin diseases. A recent study assessed 118 patients and caregivers of pediatric patients with atopic dermatitis (n=50), psoriasis (n=25), or vitiligo (n=43) using the Children’s Dermatology Life Quality Index, the Hamilton Anxiety Scale, and the Beck Depression Inventory.17 Anxiety and depression were found in 36% of caregivers of pediatric psoriasis patients and depression was found in 36% of pediatric psoriasis patients, highlighting the need for interventions on a personal and family level to improve quality of life. As a comparator, anxiety was more prevalent in vitiligo caregivers (42%), but depression was only found in 26% of caregivers in the same group. Extent of disease (25%–75% body surface area affected) correlated with both depression and anxiety in the caregivers of pediatric patients with psoriasis as well as with anxiety in caregivers of pediatric patients with increased visible surface area of vitiligo.17 Parental anxiety has been reported at times to be linked to corticosteroid phobia, or corticophobia, which may interfere with disease therapy, as topical corticosteroids are considered the mainstay of therapy in childhood disease.18 Coordinating care with caregivers and addressing their concerns about the safety of medications should be integral to the pediatric psoriasis visit.
Pustular Psoriasis
Pustular psoriasis can be seen in any age group. Researchers recently have attempted to delineate the features and successful management of this severe subset of pediatric psoriasis patients. Twenty-four pediatric pustular psoriasis cases reviewed by Posso-De Los Rios et al19 revealed that 92% (22/24) had generalized and 8% (2/24) had limited acral disease. The mean (standard deviation) age at onset of pediatric pustular psoriasis was 6.3 (4.9) years. Half of the reported cases required more than one intervention. Treatment with acitretin, cyclosporine, and methotrexate was effective, but the investigators identified that there is a true dearth of evidence-based therapeutics in pediatric pustular psoriasis and much rebound with discontinuation.19 Although the subset of pediatric pustular psoriasis is rare, study of evidence-based intervention is needed.
Therapy
Recent reviews of pediatric and adolescent psoriasis highlight the paucity of therapeutic information for these patient populations. Investigators typically focus on topical therapies as the basis of treatment,20 as well as the addition of phototherapy in mild to moderate plaque or guttate psoriasis and biologic or systemic agents in moderate to severe flares of plaque, erythrodermic, or pustular psoriasis.21 Further studies are needed to identify evidence-based therapeutic paradigms for pediatric psoriasis and to pinpoint therapies associated with the best quality of life in patients and their caregivers.
Tumor Necrosis Factor α Inhibitors
Safety and efficacy of etanercept for juvenile idiopathic arthritis including oligoarthritis, enthesitis-related arthritis, and psoriatic arthritis recently was reviewed by Windschall et al22 using data from the German pediatric Biologika in der Kinderrheumatologie registry. Juvenile Arthritis Disease Activity Score 10 improved from baseline for 127 pediatric patients with psoriatic arthritis in 3 to 24 months (mean [standard deviation], 14.7 [6.4], 5.0 [4.6], 5.3 [6.4] at baseline, 3 months, and 24 months, respectively). Overall side effects were relatively higher in the psoriatic arthritis group; the rate of serious (relative risk, 1.39 [0.95-2.03; P=.08]) and nonserious (relative risk, 1.18 [1.02-1.35; P=.03]) adverse events also was elevated. Uveitis risk was greatest in the psoriatic arthritis group and the number of associated cases of inflammatory bowel disease outnumbered those seen in other forms of arthritis. The investigators concluded that monitoring for extra-articular immunopathies should be conducted in pediatric patients with psoriatic arthritis who are undergoing etanercept therapy.22
Tumor necrosis factor α (TNF-α) inhibitors have been associated with triggering psoriasiform dermatitis in pediatric patients treated for inflammatory bowel disease. A Finnish study of infliximab side effects in pediatric patients with inflammatory bowel disease (n=84; Crohn disease: n=64) demonstrated that almost half (47.6% [40/84]) of the participants presented with chronic skin reactions, 23% of which were severe in nature.23 Psoriasiform lesions of the scalp and ears were most common, followed by the periorificial area, genitals, trunk, and extremities. Rare association with HLA-Cw*0602 genotype was noted. Skin manifestations did not correlate with gut inflammation (as determined by fecal calprotectin levels). Discontinuation of therapy rarely was required.23 Other studies also have highlighted this side effect, suggesting an incidence of 2.7% in adults with colitis treated with TNF-α inhibitors24 and 10.5% in pediatric patients with Crohn disease.25 In a study by Sherlock et al,25 pediatric patients with Crohn disease developing psoriasis following infliximab therapy were more likely to be homozygous for specific polymorphisms in the IL-23R gene (rs10489628, rs10789229, and rs1343151).
Methotrexate
For pediatric patients who are being treated with methotrexate, the polyglutamate assay recently has been reported to be helpful in identifying patients needing a dose escalation.26 Higher numbers on the polyglutamate assay are associated with superior response to methotrexate therapy. Doses can be increased after 12 weeks in patients with low assays.26
IL-23
The safety of IL-23 blockade in pediatric psoriasis patients has not yet been established, but data from adult cases have implicated the IL-17 and IL-23 pathways in psoriasis/psoriatic arthritis, including an association with IL-23R polymorphisms27 and increases in soluble IL-20 and IL-22 associated with disease severity and an association of IL-17 levels with activity on the psoriasis area and severity index scores.28 The data are more limited for pediatric cases. Pediatric patients with inflammatory bowel disease who have an IL-23R polymorphism appear to be susceptible to psoriatic flares while on TNF-α inhibitor therapy,25 which suggests that the IL-23 blockade may be of benefit for some pediatric patients with psoriasis or psoriatic arthritis.
Conclusion
Pediatric psoriasis and psoriatic arthritis have now been identified as being part of the autoimmune spectrum and are associated with metabolic syndrome, including obesity and excess central adiposity, similar to their adult variants. An overview of potential unmet needs in pediatric psoriasis is included in Table 2. These unmet needs include further delineation of diet and weight modification in the care and prevention of psoriasis; expansion of therapeutic trials and US Food and Drug Administration–approved medications for children with psoriasis, especially severe variants such as extensive plaque and pustular disease; and development of guidelines for ongoing monitoring of children with psoriasis. The role of therapeutic interventions and weight management on long-term disease course remains to be shown in extended clinical trials. Despite the great advancements in psoriatic care, knowledge gaps remain in pediatric psoriasis that will need to be addressed in the future.
1. Taclonex Expanded Indication. OptumRx Web site. https://www.optumrx.com/vgnpreview/HCP/Assets/RxNews/Clinical%20Updates_Taclonex_2014-1003.pdf. Published August 29, 2014. Accessed January 28, 2015.
2. Silverberg NB. Update on pediatric psoriasis, part 1: clinical features and demographics. Cutis. 2010;86:118-124.
3. Silverberg NB. Update on pediatric psoriasis, part 2: therapeutic management. Cutis. 2010;86:172-176.
4. Cather JC. Psoriasis in children and women: addressing some special needs. Semin Cutan Med Surg. 2014;33(2 suppl 2):S42-S44.
5. Khorsand K, Sidbury R. Recent advances in pediatric dermatology. Arch Dis Child. 2014;99:944-948.
6. Mercy K, Kwasny M, Cordoro KM, et al. Clinical manifestations of pediatric psoriasis: results of a multicenter study in the United States. Pediatr Dermatol. 2013;30:424-428.
7. Gudjonsson JE, Thorarinsson AM, Sigurgeirsson B, et al. Streptococcal throat infections and exacerbation of chronic plaque psoriasis: a prospective study. Br J Dermatol. 2003;149:530-534.
8. Ferran M, Galván AB, Rincón C, et al. Streptococcus induces circulating CLA(+) memory T-cell-dependent epidermal cell activation in psoriasis. J Invest Dermatol. 2013;133:999-1007.
9. Gul Mert G, Incecik F, Gunasti S, et al. Psoriasiform drug eruption associated with sodium valproate [published online ahead of print November 13, 2013]. Case Rep Pediatr. 2013;2013:823469.
10. Chang MW, Nakrani R. Six children with allergic contact dermatitis to methylisothiazolinone in wet wipes (baby wipes). Pediatrics. 2014;133:e434-e438.
11. Gul U, Gonul M, Kaya I, et al. Autoimmune thyroid disorders in patients with psoriasis. Eur J Dermatol. 2009;19:221-223.
12. Prahalad S, McCracken C, Ponder L, et al. A120: Familial autoimmunity in the CARRA registry. Arthritis Rheumatol. 2014;66(suppl 11):S157.
13. Mercy KM, Paller AS. The relationship between obesity and psoriasis in the pediatric population: implications and future directions. Cutis. 2013;92:107-109.
14. Paller AS, Mercy K, Kwasny MJ, et al. Association of pediatric psoriasis severity with excess and central adiposity: an international cross-sectional study. JAMA Dermatol. 2013;149:166-176.
15. Becker L, Tom WL, Eshagh K, et al. Excess adiposity preceding pediatric psoriasis. JAMA Dermatol. 2014;150:573-574.
16. Volf EM, Levine DE, Michelon MA, et al. Assessor-blinded study of the metabolic syndrome and surrogate markers of increased cardiovascular risk in children with moderate-to-severe psoriasis compared with age-matched population of children with warts. J Drugs Dermatol. 2011;10:900-901.
17. Manzoni AP, Weber MB, Nagatomi AR, et al. Assessing depression and anxiety in the caregivers of pediatric patients with chronic skin disorders. An Bras Dermatol. 2013;88:894-899.
18. Belloni Fortina A, Neri L. Topical steroids and corticophobia. G Ital Dermatol Venereol. 2013;148:651-654.
19. Posso-De Los Rios CJ, Pope E, Lara-Corrales I. A systematic review of systemic medications for pustular psoriasis in pediatrics. Pediatr Dermatol. 2014;31:430-439.
20. Tollefson MM. Diagnosis and management of psoriasis in children. Pediatr Clin North Am. 2014;61:261-277.
21. Fotiadou C, Lazaridou E, Ioannides D. Management of psoriasis in adolescence. Adolesc Health Med Ther. 2014;5:25-34.
22. Windschall D, Müller T, Becker I, et al. Safety and efficacy of etanercept in children with the JIA categories extended oligoarthritis, enthesitis-related arthritis and psoriasis arthritis [published online ahead of print July 18, 2014]. Clin Rheumatol. 2015;34:61-69.
23. Mälkönen T, Wikström A, Heiskanen K, et al. Skin reactions during anti-TNFa therapy for pediatric inflammatory bowel disease: a 2-year prospective study. Inflamm Bowel Dis. 2014;20:1309-1315.
24. Afzali A, Wheat CL, Hu JK, et al. The association of psoriasiform rash with anti-tumor necrosis factor (anti-TNF) therapy in inflammatory bowel disease: a single academic center case series. J Crohns Colitis. 2014;8:480-488.
25. Sherlock ME, Walters T, Tabbers MM, et al. Infliximab-induced psoriasis and psoriasiform skin lesions in pediatric Crohn disease and a potential association with IL-23 receptor polymorphisms. J Pediatr Gastroenterol Nutr. 2013;56:512-518.
26. Rahman SI, Siegfried E, Flanagan KH, et al. The methotrexate polyglutamate assay supports the efficacy of methotrexate for severe inflammatory skin disease in children. J Am Acad Dermatol. 2014;70:252-256.
27. Suzuki E, Mellins ED, Gershwin ME, et al. The IL-23/IL-17 axis in psoriatic arthritis. Autoimmun Rev. 2014;13:496-502.
28. Michalak-Stoma A, Bartosi´nska J, Kowal M, et al. Serum levels of selected Th17 and Th22 cytokines in psoriatic patients. Dis Markers. 2013;35:625-631.
1. Taclonex Expanded Indication. OptumRx Web site. https://www.optumrx.com/vgnpreview/HCP/Assets/RxNews/Clinical%20Updates_Taclonex_2014-1003.pdf. Published August 29, 2014. Accessed January 28, 2015.
2. Silverberg NB. Update on pediatric psoriasis, part 1: clinical features and demographics. Cutis. 2010;86:118-124.
3. Silverberg NB. Update on pediatric psoriasis, part 2: therapeutic management. Cutis. 2010;86:172-176.
4. Cather JC. Psoriasis in children and women: addressing some special needs. Semin Cutan Med Surg. 2014;33(2 suppl 2):S42-S44.
5. Khorsand K, Sidbury R. Recent advances in pediatric dermatology. Arch Dis Child. 2014;99:944-948.
6. Mercy K, Kwasny M, Cordoro KM, et al. Clinical manifestations of pediatric psoriasis: results of a multicenter study in the United States. Pediatr Dermatol. 2013;30:424-428.
7. Gudjonsson JE, Thorarinsson AM, Sigurgeirsson B, et al. Streptococcal throat infections and exacerbation of chronic plaque psoriasis: a prospective study. Br J Dermatol. 2003;149:530-534.
8. Ferran M, Galván AB, Rincón C, et al. Streptococcus induces circulating CLA(+) memory T-cell-dependent epidermal cell activation in psoriasis. J Invest Dermatol. 2013;133:999-1007.
9. Gul Mert G, Incecik F, Gunasti S, et al. Psoriasiform drug eruption associated with sodium valproate [published online ahead of print November 13, 2013]. Case Rep Pediatr. 2013;2013:823469.
10. Chang MW, Nakrani R. Six children with allergic contact dermatitis to methylisothiazolinone in wet wipes (baby wipes). Pediatrics. 2014;133:e434-e438.
11. Gul U, Gonul M, Kaya I, et al. Autoimmune thyroid disorders in patients with psoriasis. Eur J Dermatol. 2009;19:221-223.
12. Prahalad S, McCracken C, Ponder L, et al. A120: Familial autoimmunity in the CARRA registry. Arthritis Rheumatol. 2014;66(suppl 11):S157.
13. Mercy KM, Paller AS. The relationship between obesity and psoriasis in the pediatric population: implications and future directions. Cutis. 2013;92:107-109.
14. Paller AS, Mercy K, Kwasny MJ, et al. Association of pediatric psoriasis severity with excess and central adiposity: an international cross-sectional study. JAMA Dermatol. 2013;149:166-176.
15. Becker L, Tom WL, Eshagh K, et al. Excess adiposity preceding pediatric psoriasis. JAMA Dermatol. 2014;150:573-574.
16. Volf EM, Levine DE, Michelon MA, et al. Assessor-blinded study of the metabolic syndrome and surrogate markers of increased cardiovascular risk in children with moderate-to-severe psoriasis compared with age-matched population of children with warts. J Drugs Dermatol. 2011;10:900-901.
17. Manzoni AP, Weber MB, Nagatomi AR, et al. Assessing depression and anxiety in the caregivers of pediatric patients with chronic skin disorders. An Bras Dermatol. 2013;88:894-899.
18. Belloni Fortina A, Neri L. Topical steroids and corticophobia. G Ital Dermatol Venereol. 2013;148:651-654.
19. Posso-De Los Rios CJ, Pope E, Lara-Corrales I. A systematic review of systemic medications for pustular psoriasis in pediatrics. Pediatr Dermatol. 2014;31:430-439.
20. Tollefson MM. Diagnosis and management of psoriasis in children. Pediatr Clin North Am. 2014;61:261-277.
21. Fotiadou C, Lazaridou E, Ioannides D. Management of psoriasis in adolescence. Adolesc Health Med Ther. 2014;5:25-34.
22. Windschall D, Müller T, Becker I, et al. Safety and efficacy of etanercept in children with the JIA categories extended oligoarthritis, enthesitis-related arthritis and psoriasis arthritis [published online ahead of print July 18, 2014]. Clin Rheumatol. 2015;34:61-69.
23. Mälkönen T, Wikström A, Heiskanen K, et al. Skin reactions during anti-TNFa therapy for pediatric inflammatory bowel disease: a 2-year prospective study. Inflamm Bowel Dis. 2014;20:1309-1315.
24. Afzali A, Wheat CL, Hu JK, et al. The association of psoriasiform rash with anti-tumor necrosis factor (anti-TNF) therapy in inflammatory bowel disease: a single academic center case series. J Crohns Colitis. 2014;8:480-488.
25. Sherlock ME, Walters T, Tabbers MM, et al. Infliximab-induced psoriasis and psoriasiform skin lesions in pediatric Crohn disease and a potential association with IL-23 receptor polymorphisms. J Pediatr Gastroenterol Nutr. 2013;56:512-518.
26. Rahman SI, Siegfried E, Flanagan KH, et al. The methotrexate polyglutamate assay supports the efficacy of methotrexate for severe inflammatory skin disease in children. J Am Acad Dermatol. 2014;70:252-256.
27. Suzuki E, Mellins ED, Gershwin ME, et al. The IL-23/IL-17 axis in psoriatic arthritis. Autoimmun Rev. 2014;13:496-502.
28. Michalak-Stoma A, Bartosi´nska J, Kowal M, et al. Serum levels of selected Th17 and Th22 cytokines in psoriatic patients. Dis Markers. 2013;35:625-631.
Practice Points
- The majority of children with psoriasis have severe disease, scalp involvement, and a family history.
- Pediatric psoriasis is associated with metabolic syndrome, especially obesity.
- Anxiety and depression may be noted in children with psoriasis as well as their caregivers.
First Refusal
Treatment success in psoriasis, as in any dermatologic condition, is dependent on many factors. The willingness of patients to follow our suggested therapeutic plans certainly is one of the most important components of this process.
Halioua et al1 analyzed the issue of treatment refusal, which they defined as “a patient actively refusing to take treatment despite physician recommendations,” among psoriasis patients. Treatment refusal is a more complex phenomenon than nonadherence, as it requires an affirmative act that goes beyond more passive acts of not filling prescriptions, taking a medication sporadically, or forgetting to take a medication. Their objective was to investigate refusal of topical treatments by patients living with psoriasis in France as well as the factors that influence such refusal.1
The authors evaluated responses to an Internet study.1 Responses from participants who refused topical therapy (n=50) were compared to individuals who successfully applied topical treatment (n=205). Individuals receiving phototherapy, biologic therapy, and oral treatment were not included in the analysis. Spearman rank correlations completed by Fisher exact tests and Student t tests were performed.1
The researchers found that objective aspects of psoriasis, including comorbidities, localization of lesions, and symptoms associated with psoriasis, were not significant predictors of treatment refusal. The factors that did appear to influence refusal related more to patient perception of disease and its treatment.1
First, treatment refusal was defined by patient attitude toward treatment. In the treatment refusal group, significantly fewer participants reported believing that psoriasis can be managed (20.0% vs 38.5%; P<.01), and significantly more participants in the treatment refusal group reported believing that topical psoriasis treatments never work (58.0% vs 27.5%; odds ratio, 2.09; P<.0001). Additionally, significantly fewer participants in the treatment refusal group were willing to stay on prescription medications long-term (30.0% vs 77.6%; P<.001), and significantly more patients in the treatment refusal group believed that all creams (prescription or over-the-counter) work the same (54.0% vs 31.7%; odds ratio, 1.07; P=.003).1
The physician-patient relationship also influenced refusal. In the treatment refusal group, 60% of participants reported no longer consulting physicians for psoriasis treatment. The main reasons for cessation of medical care were lack of improvement of psoriasis (40%) and feeling that the physician did not take psoriasis seriously (20%). In the treatment acceptance group, only 10% of participants no longer consulted physicians.1 Among participants who continued to consult their physician (40% for the treatment refusal group and 90.2% for the treatment acceptance group), significantly fewer participants in the treatment refusal group reported that they were substantially helped by their physician (50.0% vs 73.0%; P=.03) and that they always followed physician recommendations (65.0% vs 85.4%; P=.02). Additionally, significantly fewer participants in the treatment refusal group considered that their physician took the time to listen to what he/she had to say (65.0% vs 85.9%; P=.02) and that their physician had provided clear instructions on how to utilize the treatment (65.0% vs 83.2%; P=.046).1
Therefore, treatment refusal is an important factor to be considered in the management of psoriasis. The findings of this study indicate possible strategies to reduce patient refusal. For example, enhanced education about the therapeutic options for psoriasis and their benefits could counter negative perceptions about these therapies. It also appears that increased focus on the physician-patient relationship may have a positive impact in this area.
Reference
1. Halioua B, Maury Le Breton A, de Fontaubert A, et al. Treatment refusal among patients with psoriasis [published online ahead of print]. J Dermatolog Treat. 2015;2:1-5.
Treatment success in psoriasis, as in any dermatologic condition, is dependent on many factors. The willingness of patients to follow our suggested therapeutic plans certainly is one of the most important components of this process.
Halioua et al1 analyzed the issue of treatment refusal, which they defined as “a patient actively refusing to take treatment despite physician recommendations,” among psoriasis patients. Treatment refusal is a more complex phenomenon than nonadherence, as it requires an affirmative act that goes beyond more passive acts of not filling prescriptions, taking a medication sporadically, or forgetting to take a medication. Their objective was to investigate refusal of topical treatments by patients living with psoriasis in France as well as the factors that influence such refusal.1
The authors evaluated responses to an Internet study.1 Responses from participants who refused topical therapy (n=50) were compared to individuals who successfully applied topical treatment (n=205). Individuals receiving phototherapy, biologic therapy, and oral treatment were not included in the analysis. Spearman rank correlations completed by Fisher exact tests and Student t tests were performed.1
The researchers found that objective aspects of psoriasis, including comorbidities, localization of lesions, and symptoms associated with psoriasis, were not significant predictors of treatment refusal. The factors that did appear to influence refusal related more to patient perception of disease and its treatment.1
First, treatment refusal was defined by patient attitude toward treatment. In the treatment refusal group, significantly fewer participants reported believing that psoriasis can be managed (20.0% vs 38.5%; P<.01), and significantly more participants in the treatment refusal group reported believing that topical psoriasis treatments never work (58.0% vs 27.5%; odds ratio, 2.09; P<.0001). Additionally, significantly fewer participants in the treatment refusal group were willing to stay on prescription medications long-term (30.0% vs 77.6%; P<.001), and significantly more patients in the treatment refusal group believed that all creams (prescription or over-the-counter) work the same (54.0% vs 31.7%; odds ratio, 1.07; P=.003).1
The physician-patient relationship also influenced refusal. In the treatment refusal group, 60% of participants reported no longer consulting physicians for psoriasis treatment. The main reasons for cessation of medical care were lack of improvement of psoriasis (40%) and feeling that the physician did not take psoriasis seriously (20%). In the treatment acceptance group, only 10% of participants no longer consulted physicians.1 Among participants who continued to consult their physician (40% for the treatment refusal group and 90.2% for the treatment acceptance group), significantly fewer participants in the treatment refusal group reported that they were substantially helped by their physician (50.0% vs 73.0%; P=.03) and that they always followed physician recommendations (65.0% vs 85.4%; P=.02). Additionally, significantly fewer participants in the treatment refusal group considered that their physician took the time to listen to what he/she had to say (65.0% vs 85.9%; P=.02) and that their physician had provided clear instructions on how to utilize the treatment (65.0% vs 83.2%; P=.046).1
Therefore, treatment refusal is an important factor to be considered in the management of psoriasis. The findings of this study indicate possible strategies to reduce patient refusal. For example, enhanced education about the therapeutic options for psoriasis and their benefits could counter negative perceptions about these therapies. It also appears that increased focus on the physician-patient relationship may have a positive impact in this area.
Treatment success in psoriasis, as in any dermatologic condition, is dependent on many factors. The willingness of patients to follow our suggested therapeutic plans certainly is one of the most important components of this process.
Halioua et al1 analyzed the issue of treatment refusal, which they defined as “a patient actively refusing to take treatment despite physician recommendations,” among psoriasis patients. Treatment refusal is a more complex phenomenon than nonadherence, as it requires an affirmative act that goes beyond more passive acts of not filling prescriptions, taking a medication sporadically, or forgetting to take a medication. Their objective was to investigate refusal of topical treatments by patients living with psoriasis in France as well as the factors that influence such refusal.1
The authors evaluated responses to an Internet study.1 Responses from participants who refused topical therapy (n=50) were compared to individuals who successfully applied topical treatment (n=205). Individuals receiving phototherapy, biologic therapy, and oral treatment were not included in the analysis. Spearman rank correlations completed by Fisher exact tests and Student t tests were performed.1
The researchers found that objective aspects of psoriasis, including comorbidities, localization of lesions, and symptoms associated with psoriasis, were not significant predictors of treatment refusal. The factors that did appear to influence refusal related more to patient perception of disease and its treatment.1
First, treatment refusal was defined by patient attitude toward treatment. In the treatment refusal group, significantly fewer participants reported believing that psoriasis can be managed (20.0% vs 38.5%; P<.01), and significantly more participants in the treatment refusal group reported believing that topical psoriasis treatments never work (58.0% vs 27.5%; odds ratio, 2.09; P<.0001). Additionally, significantly fewer participants in the treatment refusal group were willing to stay on prescription medications long-term (30.0% vs 77.6%; P<.001), and significantly more patients in the treatment refusal group believed that all creams (prescription or over-the-counter) work the same (54.0% vs 31.7%; odds ratio, 1.07; P=.003).1
The physician-patient relationship also influenced refusal. In the treatment refusal group, 60% of participants reported no longer consulting physicians for psoriasis treatment. The main reasons for cessation of medical care were lack of improvement of psoriasis (40%) and feeling that the physician did not take psoriasis seriously (20%). In the treatment acceptance group, only 10% of participants no longer consulted physicians.1 Among participants who continued to consult their physician (40% for the treatment refusal group and 90.2% for the treatment acceptance group), significantly fewer participants in the treatment refusal group reported that they were substantially helped by their physician (50.0% vs 73.0%; P=.03) and that they always followed physician recommendations (65.0% vs 85.4%; P=.02). Additionally, significantly fewer participants in the treatment refusal group considered that their physician took the time to listen to what he/she had to say (65.0% vs 85.9%; P=.02) and that their physician had provided clear instructions on how to utilize the treatment (65.0% vs 83.2%; P=.046).1
Therefore, treatment refusal is an important factor to be considered in the management of psoriasis. The findings of this study indicate possible strategies to reduce patient refusal. For example, enhanced education about the therapeutic options for psoriasis and their benefits could counter negative perceptions about these therapies. It also appears that increased focus on the physician-patient relationship may have a positive impact in this area.
Reference
1. Halioua B, Maury Le Breton A, de Fontaubert A, et al. Treatment refusal among patients with psoriasis [published online ahead of print]. J Dermatolog Treat. 2015;2:1-5.
Reference
1. Halioua B, Maury Le Breton A, de Fontaubert A, et al. Treatment refusal among patients with psoriasis [published online ahead of print]. J Dermatolog Treat. 2015;2:1-5.
Interleukin-23 inhibition with tildrakizumab achieves significant psoriasis improvements
A monoclonal antibody targeting an interleukin-23 subunit has shown a significant reduction in psoriasis area and disease severity in 77 psoriasis patients, according to data from a randomized, placebo-controlled phase I trial.
Three doses of intravenous tildrakizumab (0.05–10 mg/kg1) – given 1-2 months apart – were associated with 50%-80% reductions in mean placebo-corrected psoriasis area and severity index (PASI) scores at 112 days after commencing treatment, and was well tolerated with minor adverse events.
Researchers observed 50% reductions in PASI score 308 days after the last administered dose, and all subjects given 3 or 10 mg/kg achieved a 75% reduction in PASI score by day 196, according to a paper published online March 9 in Nature (doi: 10.1038/nature14175).
“Further development of tildrakizumab is warranted based on these results to determine whether selective targeting of IL-23 can provide similar or better efficacy while reducing safety concerns that are associated with other biologic agents currently approved for the treatment of psoriasis,” wrote Dr. Tamara Kopp, from the Juvenis Medical Center, Vienna, and coauthors.
The study was funded by tildrakizumab-developer Merck & Co. Inc, and several authors were employees of Merck & Co. Inc.
A monoclonal antibody targeting an interleukin-23 subunit has shown a significant reduction in psoriasis area and disease severity in 77 psoriasis patients, according to data from a randomized, placebo-controlled phase I trial.
Three doses of intravenous tildrakizumab (0.05–10 mg/kg1) – given 1-2 months apart – were associated with 50%-80% reductions in mean placebo-corrected psoriasis area and severity index (PASI) scores at 112 days after commencing treatment, and was well tolerated with minor adverse events.
Researchers observed 50% reductions in PASI score 308 days after the last administered dose, and all subjects given 3 or 10 mg/kg achieved a 75% reduction in PASI score by day 196, according to a paper published online March 9 in Nature (doi: 10.1038/nature14175).
“Further development of tildrakizumab is warranted based on these results to determine whether selective targeting of IL-23 can provide similar or better efficacy while reducing safety concerns that are associated with other biologic agents currently approved for the treatment of psoriasis,” wrote Dr. Tamara Kopp, from the Juvenis Medical Center, Vienna, and coauthors.
The study was funded by tildrakizumab-developer Merck & Co. Inc, and several authors were employees of Merck & Co. Inc.
A monoclonal antibody targeting an interleukin-23 subunit has shown a significant reduction in psoriasis area and disease severity in 77 psoriasis patients, according to data from a randomized, placebo-controlled phase I trial.
Three doses of intravenous tildrakizumab (0.05–10 mg/kg1) – given 1-2 months apart – were associated with 50%-80% reductions in mean placebo-corrected psoriasis area and severity index (PASI) scores at 112 days after commencing treatment, and was well tolerated with minor adverse events.
Researchers observed 50% reductions in PASI score 308 days after the last administered dose, and all subjects given 3 or 10 mg/kg achieved a 75% reduction in PASI score by day 196, according to a paper published online March 9 in Nature (doi: 10.1038/nature14175).
“Further development of tildrakizumab is warranted based on these results to determine whether selective targeting of IL-23 can provide similar or better efficacy while reducing safety concerns that are associated with other biologic agents currently approved for the treatment of psoriasis,” wrote Dr. Tamara Kopp, from the Juvenis Medical Center, Vienna, and coauthors.
The study was funded by tildrakizumab-developer Merck & Co. Inc, and several authors were employees of Merck & Co. Inc.
VIDEO: Secukinumab ‘exciting’ new agent for psoriasis
KAUAI, HAWAII – The Food and Drug administration recently approved a new class of drug for psoriasis. Secukinumab, indicated for the treatment of mild to severe psoriasis, targets IL-17, a cytokine highly implicated in the disease.
“I’m very excited to try this drug in my patients,” Dr. Kristina Callis Duffin said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
In this report, Dr. Duffin of the University of Utah explains the drug’s mechanism of action, describes how it differs from other biologic agents used to treat psoriasis, and shares promising data showing improved scores on the Psoriasis Area and Severity Index (PASI).
SDEF and this news organization are owned by the same parent company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @whitneymcknight
KAUAI, HAWAII – The Food and Drug administration recently approved a new class of drug for psoriasis. Secukinumab, indicated for the treatment of mild to severe psoriasis, targets IL-17, a cytokine highly implicated in the disease.
“I’m very excited to try this drug in my patients,” Dr. Kristina Callis Duffin said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
In this report, Dr. Duffin of the University of Utah explains the drug’s mechanism of action, describes how it differs from other biologic agents used to treat psoriasis, and shares promising data showing improved scores on the Psoriasis Area and Severity Index (PASI).
SDEF and this news organization are owned by the same parent company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @whitneymcknight
KAUAI, HAWAII – The Food and Drug administration recently approved a new class of drug for psoriasis. Secukinumab, indicated for the treatment of mild to severe psoriasis, targets IL-17, a cytokine highly implicated in the disease.
“I’m very excited to try this drug in my patients,” Dr. Kristina Callis Duffin said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
In this report, Dr. Duffin of the University of Utah explains the drug’s mechanism of action, describes how it differs from other biologic agents used to treat psoriasis, and shares promising data showing improved scores on the Psoriasis Area and Severity Index (PASI).
SDEF and this news organization are owned by the same parent company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @whitneymcknight
AT SDEF HAWAII DERMATOLOGY SEMINAR
Infliximab most common cause of drug-induced liver injury
Infliximab caused liver injury in 8.3% of treated patients in a prospective study, exceeding rates for other tumor necrosis factor-alpha antagonists, investigators reported online in Clinical Gastroenterology and Hepatology.
The findings show that “liver injury associated with the use of TNF-alpha antagonists is more common than previously reported, occurring in 1 in 120 of those exposed to infliximab,” said Dr. Einar S. Björnsson at the University of Iceland in Reykjavik and his associates. Furthermore, neither anti-TNF treatment dose nor baseline antinuclear acid antibody (ANA) status predicted which patients would develop drug-induced liver injury (DILI), the researchers said.
Since emerging in the 1990s, anti-TNF agents have dramatically altered the treatment landscape for autoimmune diseases such as rheumatoid arthritis, psoriasis, and inflammatory bowel disease. Although they are known to cause liver damage in some patients, data on the topic mainly come from single case reports, the researchers said (Clin. Gastroenterol. Hepatol. 2014 [doi: http://dx.doi.org/10.1016/j.cgh.2014.07.062]) To better understand the association, the researchers prospectively studied patients who received anti-TNF agents between 2009 and 2013 at the University Hospital in Iceland. They defined liver injury as aspartate aminotransferase or alanine aminotransferase (ALT) levels that were at least triple the normal upper limit, or alkaline phosphatase levels of at least double the upper limit.
A total of 1,776 patients were treated with anti-TNF agents during the 5-year study period, the researchers reported. In all, 11 developed drug-induced liver injury (DILI), of which nine cases were caused by infliximab, they said. Liver injury developed in 8.3% of patients treated with infliximab, compared with only 3.7% of those who received adalimumab and 2.3% of those given etanercept, they added. In a past analysis, the researchers calculated that one in every 148 patients would develop DILI during 2 years of treatment with infliximab (Gastroenterology 2014;144:1419-25). Patients who developed DILI on one anti-TNF agent were able to switch therapies without DILI recurring, the investigators said. Seven patients were switched from infliximab to adalimumab, etanercept, or both, and one was switched to infliximab after developing DILI on adalimumab, they added.
The researchers also compared the 11 cases to 22 randomized controls matched by age, sex, underlying condition, and treatment. Notably, among the 11 patients diagnosed with DILI, just 1 (9%) was receiving methotrexate at the time of diagnosis, compared with 59% of the controls (P = .009), they reported. “The reason for this is not clear,” they added. “Methotrexate has been shown to lead to a decrease in circulating autoantibodies in cutaneous lupus erythematosus, but the influence of methotrexate could not be confirmed during infliximab treatment.”
Five of the 11 patients with DILI had liver biopsies, of which three showed severe acute hepatitis, two indicated mild unspecified chronic hepatitis, and one showed pure canalicular cholestasis, the researchers reported. About half the patients needed steroids acutely, but “the vast majority” did not need long-term steroid treatment, they said. Exactly how anti-TNF agents cause liver injury remains unclear, they added. Future studies might evaluate whether these drugs trigger CD4 T cells to react against liver cells, as is the case in classic autoimmune hepatitis, they said.
The researchers reported no funding sources and declared having no conflicts of interest.
Infliximab caused liver injury in 8.3% of treated patients in a prospective study, exceeding rates for other tumor necrosis factor-alpha antagonists, investigators reported online in Clinical Gastroenterology and Hepatology.
The findings show that “liver injury associated with the use of TNF-alpha antagonists is more common than previously reported, occurring in 1 in 120 of those exposed to infliximab,” said Dr. Einar S. Björnsson at the University of Iceland in Reykjavik and his associates. Furthermore, neither anti-TNF treatment dose nor baseline antinuclear acid antibody (ANA) status predicted which patients would develop drug-induced liver injury (DILI), the researchers said.
Since emerging in the 1990s, anti-TNF agents have dramatically altered the treatment landscape for autoimmune diseases such as rheumatoid arthritis, psoriasis, and inflammatory bowel disease. Although they are known to cause liver damage in some patients, data on the topic mainly come from single case reports, the researchers said (Clin. Gastroenterol. Hepatol. 2014 [doi: http://dx.doi.org/10.1016/j.cgh.2014.07.062]) To better understand the association, the researchers prospectively studied patients who received anti-TNF agents between 2009 and 2013 at the University Hospital in Iceland. They defined liver injury as aspartate aminotransferase or alanine aminotransferase (ALT) levels that were at least triple the normal upper limit, or alkaline phosphatase levels of at least double the upper limit.
A total of 1,776 patients were treated with anti-TNF agents during the 5-year study period, the researchers reported. In all, 11 developed drug-induced liver injury (DILI), of which nine cases were caused by infliximab, they said. Liver injury developed in 8.3% of patients treated with infliximab, compared with only 3.7% of those who received adalimumab and 2.3% of those given etanercept, they added. In a past analysis, the researchers calculated that one in every 148 patients would develop DILI during 2 years of treatment with infliximab (Gastroenterology 2014;144:1419-25). Patients who developed DILI on one anti-TNF agent were able to switch therapies without DILI recurring, the investigators said. Seven patients were switched from infliximab to adalimumab, etanercept, or both, and one was switched to infliximab after developing DILI on adalimumab, they added.
The researchers also compared the 11 cases to 22 randomized controls matched by age, sex, underlying condition, and treatment. Notably, among the 11 patients diagnosed with DILI, just 1 (9%) was receiving methotrexate at the time of diagnosis, compared with 59% of the controls (P = .009), they reported. “The reason for this is not clear,” they added. “Methotrexate has been shown to lead to a decrease in circulating autoantibodies in cutaneous lupus erythematosus, but the influence of methotrexate could not be confirmed during infliximab treatment.”
Five of the 11 patients with DILI had liver biopsies, of which three showed severe acute hepatitis, two indicated mild unspecified chronic hepatitis, and one showed pure canalicular cholestasis, the researchers reported. About half the patients needed steroids acutely, but “the vast majority” did not need long-term steroid treatment, they said. Exactly how anti-TNF agents cause liver injury remains unclear, they added. Future studies might evaluate whether these drugs trigger CD4 T cells to react against liver cells, as is the case in classic autoimmune hepatitis, they said.
The researchers reported no funding sources and declared having no conflicts of interest.
Infliximab caused liver injury in 8.3% of treated patients in a prospective study, exceeding rates for other tumor necrosis factor-alpha antagonists, investigators reported online in Clinical Gastroenterology and Hepatology.
The findings show that “liver injury associated with the use of TNF-alpha antagonists is more common than previously reported, occurring in 1 in 120 of those exposed to infliximab,” said Dr. Einar S. Björnsson at the University of Iceland in Reykjavik and his associates. Furthermore, neither anti-TNF treatment dose nor baseline antinuclear acid antibody (ANA) status predicted which patients would develop drug-induced liver injury (DILI), the researchers said.
Since emerging in the 1990s, anti-TNF agents have dramatically altered the treatment landscape for autoimmune diseases such as rheumatoid arthritis, psoriasis, and inflammatory bowel disease. Although they are known to cause liver damage in some patients, data on the topic mainly come from single case reports, the researchers said (Clin. Gastroenterol. Hepatol. 2014 [doi: http://dx.doi.org/10.1016/j.cgh.2014.07.062]) To better understand the association, the researchers prospectively studied patients who received anti-TNF agents between 2009 and 2013 at the University Hospital in Iceland. They defined liver injury as aspartate aminotransferase or alanine aminotransferase (ALT) levels that were at least triple the normal upper limit, or alkaline phosphatase levels of at least double the upper limit.
A total of 1,776 patients were treated with anti-TNF agents during the 5-year study period, the researchers reported. In all, 11 developed drug-induced liver injury (DILI), of which nine cases were caused by infliximab, they said. Liver injury developed in 8.3% of patients treated with infliximab, compared with only 3.7% of those who received adalimumab and 2.3% of those given etanercept, they added. In a past analysis, the researchers calculated that one in every 148 patients would develop DILI during 2 years of treatment with infliximab (Gastroenterology 2014;144:1419-25). Patients who developed DILI on one anti-TNF agent were able to switch therapies without DILI recurring, the investigators said. Seven patients were switched from infliximab to adalimumab, etanercept, or both, and one was switched to infliximab after developing DILI on adalimumab, they added.
The researchers also compared the 11 cases to 22 randomized controls matched by age, sex, underlying condition, and treatment. Notably, among the 11 patients diagnosed with DILI, just 1 (9%) was receiving methotrexate at the time of diagnosis, compared with 59% of the controls (P = .009), they reported. “The reason for this is not clear,” they added. “Methotrexate has been shown to lead to a decrease in circulating autoantibodies in cutaneous lupus erythematosus, but the influence of methotrexate could not be confirmed during infliximab treatment.”
Five of the 11 patients with DILI had liver biopsies, of which three showed severe acute hepatitis, two indicated mild unspecified chronic hepatitis, and one showed pure canalicular cholestasis, the researchers reported. About half the patients needed steroids acutely, but “the vast majority” did not need long-term steroid treatment, they said. Exactly how anti-TNF agents cause liver injury remains unclear, they added. Future studies might evaluate whether these drugs trigger CD4 T cells to react against liver cells, as is the case in classic autoimmune hepatitis, they said.
The researchers reported no funding sources and declared having no conflicts of interest.
Key clinical point: Infliximab was the most common anti–tumor necrosis factor-alpha agent linked to liver injury.
Major finding: Rates of drug-induced liver injury were highest among patients treated with infliximab (8.3%), compared with 3.7% for adalimumab and 2.3% for etanercept.
Data source: Prospective study of 11 cases of drug-induced liver injury and 22 controls.
Disclosures: The researchers declared no funding sources or conflicts of interest.
New psoriasis drugs offer treatment advantages
MIAMI BEACH – Two of the newest treatments available for psoriasis – apremilast and secukinumab – are true “game-changers,” according to Dr. David M. Pariser.
Apremilast (Otezla), a recently approved oral phosphodiesterase 4 inhibitor, will be a particularly attractive treatment option for many dermatologists and patients, he said at the South Beach Symposium.
Apremilast has a very limited effect on the immune system, and it’s an oral therapy and thus requires no needles. It is very safe – with “strikingly few” serious adverse events – and no laboratory monitoring is required, he explained.
Efficacy results with apremilast are modest. In the phase III ESTEEM trial, for example, 33% of patients achieved at least 75% improvement (PASI-75), compared with 5% of patients who received placebo, said Dr. Pariser of Eastern Virginia Medical School, Norfolk, and an investigator for the trial.
Further, the drug can be used for almost any patient and type of psoriasis; it is an option for those who want systemic therapy, but who don’t want to go on a biologic or methotrexate, and its use is not precluded by a history of cancer or infections, as is the case with biologics, he added.
Apremilast also will be attractive for dermatologists who do not currently prescribe systemic therapy for psoriasis, or who don’t use aggressive systemic therapy for psoriasis, he said.
“If a prescriber feels safety is more important than efficacy, this might be a good choice,” he said.
Secukinumab (Cosentyx), on the other hand, is a “big gun,” Dr. Pariser said of the biologic, which was approved in January 2015 for the treatment of adults with moderate to severe plaque psoriasis.
“It’s the biggest gun we’ve got now … and it really has a safety profile similar to existing biologics so far,” he said.
The fully human monoclonal antibody inhibits interleukin-17A and is administered by subcutaneous injection. Its safety and efficacy were demonstrated in numerous of studies involving about 4,500 patients. Treatment was associated with significant improvement, compared with placebo, said Dr. Pariser, who also was an investigator on secukinumab trials.
Of note, while the PASI-75 findings for secukinumab are “a nice number but not dramatically higher than things we have had in the past,” the PASI-90 and PASI-100 scores are remarkable, he said.
At the highest dose studied (300 mg given at weeks 1, 2, 3, 4, and 8, and monthly thereafter), PASI-90 was achieved by 59% of patients in one study, and PASI-100 was achieved in 28%.
“That’s significant. We haven’t had that before, and that is really, really, really nice,” he said.
Further, the primary efficacy endpoint of the study was outcome at 12 weeks, but patients continued to improve at least until 16 weeks.
PASI-100 – no psoriasis whatsoever – was 40% at 16 weeks, he said.
In another phase III trial, secukinumab was again shown to be superior to placebo, but it also compared favorably with etanercept, he noted.
Safety was reasonable in the secukinumab trials. No deaths occurred, but there were more serious adverse events and discontinuations in the active treatment group. Nasopharyngitis was the most common serious adverse events, and it occurred in all groups. Upper respiratory tract infections appeared to be more common in the secukinumab patients, he noted.
An important consideration with secukinumab, however, is the need for continuous treatment, he said.
“The bottom line, really, is that patients should stay on it. They will lose effectiveness if they go off of it or take it on an as-needed basis,” he said.
Dr. Pariser is a consultant and/or researcher for Amgen, AbbVie, Celgene, Eli Lilly, Janssen Pharmaceuticals, Merck, and Pfizer.
MIAMI BEACH – Two of the newest treatments available for psoriasis – apremilast and secukinumab – are true “game-changers,” according to Dr. David M. Pariser.
Apremilast (Otezla), a recently approved oral phosphodiesterase 4 inhibitor, will be a particularly attractive treatment option for many dermatologists and patients, he said at the South Beach Symposium.
Apremilast has a very limited effect on the immune system, and it’s an oral therapy and thus requires no needles. It is very safe – with “strikingly few” serious adverse events – and no laboratory monitoring is required, he explained.
Efficacy results with apremilast are modest. In the phase III ESTEEM trial, for example, 33% of patients achieved at least 75% improvement (PASI-75), compared with 5% of patients who received placebo, said Dr. Pariser of Eastern Virginia Medical School, Norfolk, and an investigator for the trial.
Further, the drug can be used for almost any patient and type of psoriasis; it is an option for those who want systemic therapy, but who don’t want to go on a biologic or methotrexate, and its use is not precluded by a history of cancer or infections, as is the case with biologics, he added.
Apremilast also will be attractive for dermatologists who do not currently prescribe systemic therapy for psoriasis, or who don’t use aggressive systemic therapy for psoriasis, he said.
“If a prescriber feels safety is more important than efficacy, this might be a good choice,” he said.
Secukinumab (Cosentyx), on the other hand, is a “big gun,” Dr. Pariser said of the biologic, which was approved in January 2015 for the treatment of adults with moderate to severe plaque psoriasis.
“It’s the biggest gun we’ve got now … and it really has a safety profile similar to existing biologics so far,” he said.
The fully human monoclonal antibody inhibits interleukin-17A and is administered by subcutaneous injection. Its safety and efficacy were demonstrated in numerous of studies involving about 4,500 patients. Treatment was associated with significant improvement, compared with placebo, said Dr. Pariser, who also was an investigator on secukinumab trials.
Of note, while the PASI-75 findings for secukinumab are “a nice number but not dramatically higher than things we have had in the past,” the PASI-90 and PASI-100 scores are remarkable, he said.
At the highest dose studied (300 mg given at weeks 1, 2, 3, 4, and 8, and monthly thereafter), PASI-90 was achieved by 59% of patients in one study, and PASI-100 was achieved in 28%.
“That’s significant. We haven’t had that before, and that is really, really, really nice,” he said.
Further, the primary efficacy endpoint of the study was outcome at 12 weeks, but patients continued to improve at least until 16 weeks.
PASI-100 – no psoriasis whatsoever – was 40% at 16 weeks, he said.
In another phase III trial, secukinumab was again shown to be superior to placebo, but it also compared favorably with etanercept, he noted.
Safety was reasonable in the secukinumab trials. No deaths occurred, but there were more serious adverse events and discontinuations in the active treatment group. Nasopharyngitis was the most common serious adverse events, and it occurred in all groups. Upper respiratory tract infections appeared to be more common in the secukinumab patients, he noted.
An important consideration with secukinumab, however, is the need for continuous treatment, he said.
“The bottom line, really, is that patients should stay on it. They will lose effectiveness if they go off of it or take it on an as-needed basis,” he said.
Dr. Pariser is a consultant and/or researcher for Amgen, AbbVie, Celgene, Eli Lilly, Janssen Pharmaceuticals, Merck, and Pfizer.
MIAMI BEACH – Two of the newest treatments available for psoriasis – apremilast and secukinumab – are true “game-changers,” according to Dr. David M. Pariser.
Apremilast (Otezla), a recently approved oral phosphodiesterase 4 inhibitor, will be a particularly attractive treatment option for many dermatologists and patients, he said at the South Beach Symposium.
Apremilast has a very limited effect on the immune system, and it’s an oral therapy and thus requires no needles. It is very safe – with “strikingly few” serious adverse events – and no laboratory monitoring is required, he explained.
Efficacy results with apremilast are modest. In the phase III ESTEEM trial, for example, 33% of patients achieved at least 75% improvement (PASI-75), compared with 5% of patients who received placebo, said Dr. Pariser of Eastern Virginia Medical School, Norfolk, and an investigator for the trial.
Further, the drug can be used for almost any patient and type of psoriasis; it is an option for those who want systemic therapy, but who don’t want to go on a biologic or methotrexate, and its use is not precluded by a history of cancer or infections, as is the case with biologics, he added.
Apremilast also will be attractive for dermatologists who do not currently prescribe systemic therapy for psoriasis, or who don’t use aggressive systemic therapy for psoriasis, he said.
“If a prescriber feels safety is more important than efficacy, this might be a good choice,” he said.
Secukinumab (Cosentyx), on the other hand, is a “big gun,” Dr. Pariser said of the biologic, which was approved in January 2015 for the treatment of adults with moderate to severe plaque psoriasis.
“It’s the biggest gun we’ve got now … and it really has a safety profile similar to existing biologics so far,” he said.
The fully human monoclonal antibody inhibits interleukin-17A and is administered by subcutaneous injection. Its safety and efficacy were demonstrated in numerous of studies involving about 4,500 patients. Treatment was associated with significant improvement, compared with placebo, said Dr. Pariser, who also was an investigator on secukinumab trials.
Of note, while the PASI-75 findings for secukinumab are “a nice number but not dramatically higher than things we have had in the past,” the PASI-90 and PASI-100 scores are remarkable, he said.
At the highest dose studied (300 mg given at weeks 1, 2, 3, 4, and 8, and monthly thereafter), PASI-90 was achieved by 59% of patients in one study, and PASI-100 was achieved in 28%.
“That’s significant. We haven’t had that before, and that is really, really, really nice,” he said.
Further, the primary efficacy endpoint of the study was outcome at 12 weeks, but patients continued to improve at least until 16 weeks.
PASI-100 – no psoriasis whatsoever – was 40% at 16 weeks, he said.
In another phase III trial, secukinumab was again shown to be superior to placebo, but it also compared favorably with etanercept, he noted.
Safety was reasonable in the secukinumab trials. No deaths occurred, but there were more serious adverse events and discontinuations in the active treatment group. Nasopharyngitis was the most common serious adverse events, and it occurred in all groups. Upper respiratory tract infections appeared to be more common in the secukinumab patients, he noted.
An important consideration with secukinumab, however, is the need for continuous treatment, he said.
“The bottom line, really, is that patients should stay on it. They will lose effectiveness if they go off of it or take it on an as-needed basis,” he said.
Dr. Pariser is a consultant and/or researcher for Amgen, AbbVie, Celgene, Eli Lilly, Janssen Pharmaceuticals, Merck, and Pfizer.
EXPERT ANALYSIS FROM THE ANNUAL SOUTH BEACH SYMPOSIUM
Data suggest link between tonsillectomy, psoriasis improvement
MIAMI BEACH – If all else fails in treating psoriasis, a tonsillectomy may do the trick – at least in patients whose psoriasis is associated with recurrent tonsillitis, according to findings from a systematic review of available data.
“Maybe taking the tonsils out removes resident bacteria like strep whose antigens tend to promote psoriasis,” Dr. Theodore Rosen, professor of dermatology at Baylor College of Medicine, Houston, suggested during a presentation at the South Beach Symposium.
The relationship between streptococcal pharyngitis and guttate psoriasis is well known, he explained during the talk on the latest findings in clinical dermatology.
Indeed, the authors of the systematic review noted that streptococcal infection is associated with psoriasis onset in some patients, but said it was unknown whether tonsillectomy decreases psoriasis symptoms in those patients.
Dr. Tara D. Rachakonda of Salt Lake City and her colleagues included studies dating back to 1960, and identified 20 relevant studies – including 5 controlled studies – involving 545 patients with psoriasis who were evaluated for or underwent tonsillectomy. The overall improvement rate across the studies was about 70% in 410 patients who underwent tonsillectomy, and in one of the studies the response rate was 86%.
In some cases, the response was sustained, but some patients experienced relapse, they reported (JAAD 2015;72:261-75).
The authors concluded that tonsillectomy may be a potential treatment option in patients with recalcitrant psoriasis associated with episodes of tonsillitis, but noted that additional study with long-term follow-up is needed to examine both the extent and persistence of benefit of tonsillectomy in psoriasis patients.
“The evidence is not sufficient to recommend tonsillectomy in all of your psoriasis patients,” Dr. Rosen said, but he noted that it may be something worth considering in patients who still have their tonsils and who are not responding to psoriasis treatments.
Dr. Rosen reported having no relevant financial disclosures.
MIAMI BEACH – If all else fails in treating psoriasis, a tonsillectomy may do the trick – at least in patients whose psoriasis is associated with recurrent tonsillitis, according to findings from a systematic review of available data.
“Maybe taking the tonsils out removes resident bacteria like strep whose antigens tend to promote psoriasis,” Dr. Theodore Rosen, professor of dermatology at Baylor College of Medicine, Houston, suggested during a presentation at the South Beach Symposium.
The relationship between streptococcal pharyngitis and guttate psoriasis is well known, he explained during the talk on the latest findings in clinical dermatology.
Indeed, the authors of the systematic review noted that streptococcal infection is associated with psoriasis onset in some patients, but said it was unknown whether tonsillectomy decreases psoriasis symptoms in those patients.
Dr. Tara D. Rachakonda of Salt Lake City and her colleagues included studies dating back to 1960, and identified 20 relevant studies – including 5 controlled studies – involving 545 patients with psoriasis who were evaluated for or underwent tonsillectomy. The overall improvement rate across the studies was about 70% in 410 patients who underwent tonsillectomy, and in one of the studies the response rate was 86%.
In some cases, the response was sustained, but some patients experienced relapse, they reported (JAAD 2015;72:261-75).
The authors concluded that tonsillectomy may be a potential treatment option in patients with recalcitrant psoriasis associated with episodes of tonsillitis, but noted that additional study with long-term follow-up is needed to examine both the extent and persistence of benefit of tonsillectomy in psoriasis patients.
“The evidence is not sufficient to recommend tonsillectomy in all of your psoriasis patients,” Dr. Rosen said, but he noted that it may be something worth considering in patients who still have their tonsils and who are not responding to psoriasis treatments.
Dr. Rosen reported having no relevant financial disclosures.
MIAMI BEACH – If all else fails in treating psoriasis, a tonsillectomy may do the trick – at least in patients whose psoriasis is associated with recurrent tonsillitis, according to findings from a systematic review of available data.
“Maybe taking the tonsils out removes resident bacteria like strep whose antigens tend to promote psoriasis,” Dr. Theodore Rosen, professor of dermatology at Baylor College of Medicine, Houston, suggested during a presentation at the South Beach Symposium.
The relationship between streptococcal pharyngitis and guttate psoriasis is well known, he explained during the talk on the latest findings in clinical dermatology.
Indeed, the authors of the systematic review noted that streptococcal infection is associated with psoriasis onset in some patients, but said it was unknown whether tonsillectomy decreases psoriasis symptoms in those patients.
Dr. Tara D. Rachakonda of Salt Lake City and her colleagues included studies dating back to 1960, and identified 20 relevant studies – including 5 controlled studies – involving 545 patients with psoriasis who were evaluated for or underwent tonsillectomy. The overall improvement rate across the studies was about 70% in 410 patients who underwent tonsillectomy, and in one of the studies the response rate was 86%.
In some cases, the response was sustained, but some patients experienced relapse, they reported (JAAD 2015;72:261-75).
The authors concluded that tonsillectomy may be a potential treatment option in patients with recalcitrant psoriasis associated with episodes of tonsillitis, but noted that additional study with long-term follow-up is needed to examine both the extent and persistence of benefit of tonsillectomy in psoriasis patients.
“The evidence is not sufficient to recommend tonsillectomy in all of your psoriasis patients,” Dr. Rosen said, but he noted that it may be something worth considering in patients who still have their tonsils and who are not responding to psoriasis treatments.
Dr. Rosen reported having no relevant financial disclosures.
Ixekizumab improves lesions in patients with chronic plaque psoriasis after 20 weeks
Patients with chronic plaque psoriasis who were given ixekizumab improved the severity of scalp and nail lesions in a phase 2 trial, according to Dr. Richard G. Langley of Dalhousie University in Halifax, N.S., and his associates.
In a 20-week randomized, placebo-controlled trial, 142 patients with moderate to severe plaque psoriasis at baseline were injected subcutaneously with ixekizumab at weeks 0, 2, 4, 8, 12, and 16. Ixekizumab, a monoclonal antibody, specifically targets IL-17A, a cytokine involved in the development of psoriasis.
Patients with scalp psoriasis were split into groups and given 10-, 25-, 75- and 150-mg doses of ixekizumab, or placebo; patients with nail psoriasis received 75- and 150-mg doses of ixekizumab. After 20 weeks, patients scalp psoriasis patients in the 25-, 75-, and 150-mg groups and nail psoriasis patients in the 75- and 150-mg groups showed significant improvement from baseline. By week 48, 78% of patients with scalp psoriasis and 51% of patients with nail psoriasis experienced complete resolution of lesions, the investigators reported.
Read the full article in the Journal of the European Academy of Dermatology and Venereology.
Patients with chronic plaque psoriasis who were given ixekizumab improved the severity of scalp and nail lesions in a phase 2 trial, according to Dr. Richard G. Langley of Dalhousie University in Halifax, N.S., and his associates.
In a 20-week randomized, placebo-controlled trial, 142 patients with moderate to severe plaque psoriasis at baseline were injected subcutaneously with ixekizumab at weeks 0, 2, 4, 8, 12, and 16. Ixekizumab, a monoclonal antibody, specifically targets IL-17A, a cytokine involved in the development of psoriasis.
Patients with scalp psoriasis were split into groups and given 10-, 25-, 75- and 150-mg doses of ixekizumab, or placebo; patients with nail psoriasis received 75- and 150-mg doses of ixekizumab. After 20 weeks, patients scalp psoriasis patients in the 25-, 75-, and 150-mg groups and nail psoriasis patients in the 75- and 150-mg groups showed significant improvement from baseline. By week 48, 78% of patients with scalp psoriasis and 51% of patients with nail psoriasis experienced complete resolution of lesions, the investigators reported.
Read the full article in the Journal of the European Academy of Dermatology and Venereology.
Patients with chronic plaque psoriasis who were given ixekizumab improved the severity of scalp and nail lesions in a phase 2 trial, according to Dr. Richard G. Langley of Dalhousie University in Halifax, N.S., and his associates.
In a 20-week randomized, placebo-controlled trial, 142 patients with moderate to severe plaque psoriasis at baseline were injected subcutaneously with ixekizumab at weeks 0, 2, 4, 8, 12, and 16. Ixekizumab, a monoclonal antibody, specifically targets IL-17A, a cytokine involved in the development of psoriasis.
Patients with scalp psoriasis were split into groups and given 10-, 25-, 75- and 150-mg doses of ixekizumab, or placebo; patients with nail psoriasis received 75- and 150-mg doses of ixekizumab. After 20 weeks, patients scalp psoriasis patients in the 25-, 75-, and 150-mg groups and nail psoriasis patients in the 75- and 150-mg groups showed significant improvement from baseline. By week 48, 78% of patients with scalp psoriasis and 51% of patients with nail psoriasis experienced complete resolution of lesions, the investigators reported.
Read the full article in the Journal of the European Academy of Dermatology and Venereology.
Counting Costs
We are all aware of the rising costs of medical care, especially for complex diseases such as psoriasis. The total cost of psoriasis in the United States is unknown. Brezinski et al (JAMA Dermatol. doi:10.1001/jamadermatol.2014.3593) sought to define the economic burden of psoriasis in the United States. They argued that this information is needed to provide the foundation for research, advocacy, and educational efforts within the disease.
The authors searched PubMed and MEDLINE databases for economic investigations on the costs of adult psoriasis in the United States. The primary objective of the analysis was to provide a comprehensive analysis of the literature on the economic burden of psoriasis in the United States. The direct, indirect, intangible, and comorbidity costs of psoriasis were reported based on this systematic literature review and adjusted to 2013 US dollars.
The direct costs included medical costs associated with (1) specialist medical evaluations, (2) hospitalization, (3) prescription medications, (4) phototherapy, (5) medication administration costs, (6) laboratory tests and monitoring studies, and (7) over-the-counter medications and self-care products. The indirect costs were determined by absenteeism and impaired work productivity. Intangible costs were calculated as a measure of the negative effect of psoriasis on quality of life. Finally, comorbidity costs measured the medical evaluations, treatment, and lab monitoring that were directly attributed to comorbid conditions associated with psoriasis.
An initial review of the literature generated 100 articles; 22 studies were included in the systematic review. The direct psoriasis costs ranged from $51.7 billion to $63.2 billion, the indirect costs ranged from $23.9 billion to $35.4 billion, and medical comorbidities were estimated to contribute $36.4 billion annually in 2013 US dollars. The annual cost of psoriasis in the United States amounted to approximately $112 billion in 2013.
The authors concluded that the economic burden of psoriasis was substantial and significant in the United States.
What’s the issue?
In the United States, the economic burden of psoriasis is substantial because this disease is associated with negative physical, psychiatric, and social consequences. In addition, treatment costs continue to rise. How will this analysis of cost influence your future management of psoriasis?
We are all aware of the rising costs of medical care, especially for complex diseases such as psoriasis. The total cost of psoriasis in the United States is unknown. Brezinski et al (JAMA Dermatol. doi:10.1001/jamadermatol.2014.3593) sought to define the economic burden of psoriasis in the United States. They argued that this information is needed to provide the foundation for research, advocacy, and educational efforts within the disease.
The authors searched PubMed and MEDLINE databases for economic investigations on the costs of adult psoriasis in the United States. The primary objective of the analysis was to provide a comprehensive analysis of the literature on the economic burden of psoriasis in the United States. The direct, indirect, intangible, and comorbidity costs of psoriasis were reported based on this systematic literature review and adjusted to 2013 US dollars.
The direct costs included medical costs associated with (1) specialist medical evaluations, (2) hospitalization, (3) prescription medications, (4) phototherapy, (5) medication administration costs, (6) laboratory tests and monitoring studies, and (7) over-the-counter medications and self-care products. The indirect costs were determined by absenteeism and impaired work productivity. Intangible costs were calculated as a measure of the negative effect of psoriasis on quality of life. Finally, comorbidity costs measured the medical evaluations, treatment, and lab monitoring that were directly attributed to comorbid conditions associated with psoriasis.
An initial review of the literature generated 100 articles; 22 studies were included in the systematic review. The direct psoriasis costs ranged from $51.7 billion to $63.2 billion, the indirect costs ranged from $23.9 billion to $35.4 billion, and medical comorbidities were estimated to contribute $36.4 billion annually in 2013 US dollars. The annual cost of psoriasis in the United States amounted to approximately $112 billion in 2013.
The authors concluded that the economic burden of psoriasis was substantial and significant in the United States.
What’s the issue?
In the United States, the economic burden of psoriasis is substantial because this disease is associated with negative physical, psychiatric, and social consequences. In addition, treatment costs continue to rise. How will this analysis of cost influence your future management of psoriasis?
We are all aware of the rising costs of medical care, especially for complex diseases such as psoriasis. The total cost of psoriasis in the United States is unknown. Brezinski et al (JAMA Dermatol. doi:10.1001/jamadermatol.2014.3593) sought to define the economic burden of psoriasis in the United States. They argued that this information is needed to provide the foundation for research, advocacy, and educational efforts within the disease.
The authors searched PubMed and MEDLINE databases for economic investigations on the costs of adult psoriasis in the United States. The primary objective of the analysis was to provide a comprehensive analysis of the literature on the economic burden of psoriasis in the United States. The direct, indirect, intangible, and comorbidity costs of psoriasis were reported based on this systematic literature review and adjusted to 2013 US dollars.
The direct costs included medical costs associated with (1) specialist medical evaluations, (2) hospitalization, (3) prescription medications, (4) phototherapy, (5) medication administration costs, (6) laboratory tests and monitoring studies, and (7) over-the-counter medications and self-care products. The indirect costs were determined by absenteeism and impaired work productivity. Intangible costs were calculated as a measure of the negative effect of psoriasis on quality of life. Finally, comorbidity costs measured the medical evaluations, treatment, and lab monitoring that were directly attributed to comorbid conditions associated with psoriasis.
An initial review of the literature generated 100 articles; 22 studies were included in the systematic review. The direct psoriasis costs ranged from $51.7 billion to $63.2 billion, the indirect costs ranged from $23.9 billion to $35.4 billion, and medical comorbidities were estimated to contribute $36.4 billion annually in 2013 US dollars. The annual cost of psoriasis in the United States amounted to approximately $112 billion in 2013.
The authors concluded that the economic burden of psoriasis was substantial and significant in the United States.
What’s the issue?
In the United States, the economic burden of psoriasis is substantial because this disease is associated with negative physical, psychiatric, and social consequences. In addition, treatment costs continue to rise. How will this analysis of cost influence your future management of psoriasis?