User login
Apremilast meets psoriasis endpoints at week 32
SAN FRANCISCO – Apremilast met its primary endpoint at week 32 with no new or serious safety signals among patients with moderate to severe plaque psoriasis, according to phase III data from the ongoing LIBERATE study.
At week 16, 40% of patients who received oral apremilast achieved PASI-75, compared with 12% of the placebo group, Dr. Kristian Reich reported at the annual meeting of the American Academy of Dermatology.
By week 32, PASI-75 response for apremilast had further risen to 53%, he added. “This drug may not be a quick fix, but the longer you give it, the higher the response,” said Dr. Reich of SCIderm Research Institute and Dermatologikum Hamburg in Germany.
The randomized, double-blind LIBERATE study compared the safety and efficacy of apremilast (30 mg twice daily) and injectable etanercept (50 mg weekly) with placebo among 250 patients with plaque psoriasis who had not previously received biologic therapy. Patients received one of the three treatments through week 16, and then all were switched to or continued apremilast.
At week 16, PASI-75 response rates were 40% for apremilast, 48% for etanercept, and 12% for placebo (P < .0001 for apremilast versus placebo), reported Dr. Reich. The high rate of response to placebo might have occurred because patients in the study were treatment naive, he said. At week 32, PASI-75 response rates were 53% for patients who received apremilast from baseline, and 61% for patients who switched from etanercept to apremilast at week 16.
Based on the results, “I would probably use apremilast more in the moderate [psoriasis] space than in the severe space, but its efficacy does not correlate with baseline disease severity, as far as I know,” said Dr. Reich. Apremilast also beat placebo in analyses of secondary endpoints, including static physician global assessment of clear or almost clear and the Dermatology Quality of Life Index, he added.
Safety analyses showed that the drug was generally well tolerated. Fewer than 4% of patients discontinued treatment because of adverse events, the most common of which were diarrhea, nausea, vomiting, and headache (including tension headache). No new side effects emerged after patients switched from etanercept to apremilast at week 16, he said.
A post hoc analysis found that apremilast was noninferior to etanercept (P > .05) in terms of PASI-75, although the study was not powered to directly compare the two biologics, Dr. Reich noted.
Celgene Corporation makes apremilast and sponsored the study. Dr. Reich reported serving as a speaker for and receiving honoraria from Celgene.
SAN FRANCISCO – Apremilast met its primary endpoint at week 32 with no new or serious safety signals among patients with moderate to severe plaque psoriasis, according to phase III data from the ongoing LIBERATE study.
At week 16, 40% of patients who received oral apremilast achieved PASI-75, compared with 12% of the placebo group, Dr. Kristian Reich reported at the annual meeting of the American Academy of Dermatology.
By week 32, PASI-75 response for apremilast had further risen to 53%, he added. “This drug may not be a quick fix, but the longer you give it, the higher the response,” said Dr. Reich of SCIderm Research Institute and Dermatologikum Hamburg in Germany.
The randomized, double-blind LIBERATE study compared the safety and efficacy of apremilast (30 mg twice daily) and injectable etanercept (50 mg weekly) with placebo among 250 patients with plaque psoriasis who had not previously received biologic therapy. Patients received one of the three treatments through week 16, and then all were switched to or continued apremilast.
At week 16, PASI-75 response rates were 40% for apremilast, 48% for etanercept, and 12% for placebo (P < .0001 for apremilast versus placebo), reported Dr. Reich. The high rate of response to placebo might have occurred because patients in the study were treatment naive, he said. At week 32, PASI-75 response rates were 53% for patients who received apremilast from baseline, and 61% for patients who switched from etanercept to apremilast at week 16.
Based on the results, “I would probably use apremilast more in the moderate [psoriasis] space than in the severe space, but its efficacy does not correlate with baseline disease severity, as far as I know,” said Dr. Reich. Apremilast also beat placebo in analyses of secondary endpoints, including static physician global assessment of clear or almost clear and the Dermatology Quality of Life Index, he added.
Safety analyses showed that the drug was generally well tolerated. Fewer than 4% of patients discontinued treatment because of adverse events, the most common of which were diarrhea, nausea, vomiting, and headache (including tension headache). No new side effects emerged after patients switched from etanercept to apremilast at week 16, he said.
A post hoc analysis found that apremilast was noninferior to etanercept (P > .05) in terms of PASI-75, although the study was not powered to directly compare the two biologics, Dr. Reich noted.
Celgene Corporation makes apremilast and sponsored the study. Dr. Reich reported serving as a speaker for and receiving honoraria from Celgene.
SAN FRANCISCO – Apremilast met its primary endpoint at week 32 with no new or serious safety signals among patients with moderate to severe plaque psoriasis, according to phase III data from the ongoing LIBERATE study.
At week 16, 40% of patients who received oral apremilast achieved PASI-75, compared with 12% of the placebo group, Dr. Kristian Reich reported at the annual meeting of the American Academy of Dermatology.
By week 32, PASI-75 response for apremilast had further risen to 53%, he added. “This drug may not be a quick fix, but the longer you give it, the higher the response,” said Dr. Reich of SCIderm Research Institute and Dermatologikum Hamburg in Germany.
The randomized, double-blind LIBERATE study compared the safety and efficacy of apremilast (30 mg twice daily) and injectable etanercept (50 mg weekly) with placebo among 250 patients with plaque psoriasis who had not previously received biologic therapy. Patients received one of the three treatments through week 16, and then all were switched to or continued apremilast.
At week 16, PASI-75 response rates were 40% for apremilast, 48% for etanercept, and 12% for placebo (P < .0001 for apremilast versus placebo), reported Dr. Reich. The high rate of response to placebo might have occurred because patients in the study were treatment naive, he said. At week 32, PASI-75 response rates were 53% for patients who received apremilast from baseline, and 61% for patients who switched from etanercept to apremilast at week 16.
Based on the results, “I would probably use apremilast more in the moderate [psoriasis] space than in the severe space, but its efficacy does not correlate with baseline disease severity, as far as I know,” said Dr. Reich. Apremilast also beat placebo in analyses of secondary endpoints, including static physician global assessment of clear or almost clear and the Dermatology Quality of Life Index, he added.
Safety analyses showed that the drug was generally well tolerated. Fewer than 4% of patients discontinued treatment because of adverse events, the most common of which were diarrhea, nausea, vomiting, and headache (including tension headache). No new side effects emerged after patients switched from etanercept to apremilast at week 16, he said.
A post hoc analysis found that apremilast was noninferior to etanercept (P > .05) in terms of PASI-75, although the study was not powered to directly compare the two biologics, Dr. Reich noted.
Celgene Corporation makes apremilast and sponsored the study. Dr. Reich reported serving as a speaker for and receiving honoraria from Celgene.
AT THE AAD ANNUAL MEETING
Key clinical point: Oral apremilast beat placebo and showed durable efficacy in patients with moderate to severe plaque psoriasis.
Major finding: PASI-75 response rates were 40% at week 16 (compared with 12% for placebo) and 53% at week 32.
Data source: Ongoing phase IIIb study of apremilast, etanercept, and placebo among 250 patients with moderate to severe plaque psoriasis.
Disclosures: Celgene Corporation makes apremilast and sponsored the study. Dr. Reich reported serving as a speaker for and receiving honoraria from Celgene.
Ixekizumab met psoriasis endpoints by week 12, with durable response at 60 weeks
SAN FRANCISCO – More than 80% of psoriasis patients who received ixekizumab achieved a 75% reduction in the Psoriasis Area and Severity Index score and static Physician Global Assessment scores of clear or almost clear skin at 12 weeks, based on the results of a phase III trial.
In addition, 35% of patients who received 80 mg ixekizumab twice monthly for 12 weeks achieved complete resolution of their psoriasis plaques (PASI 100), reported Dr. Kenneth Gordon of Northwestern University in Chicago. “The overall safety profile for ixekizumab was acceptable during both the induction and maintenance phases,” Dr. Gordon said at the annual meeting of the American Academy of Dermatology.
Ixekizumab is a monoclonal antibody that targets interleukin (IL)-17A, a major cytokine in the pathogenesis of psoriasis. Dr. Gordon and his associates conducted a randomized induction and withdrawal trial that compared twice-monthly or monthly treatment of the biologic with placebo among 1,296 patients. During the 12-week induction phase, 431 patients received placebo, 433 received 80 mg ixekizumab every 2 weeks, and 432 received 80 mg ixekizumab every 4 weeks, said Dr. Gordon. He and his associates then rerandomized the responders (based on PASI 75 and static Patient Global Assessment [sPGA] scores) to one of the three protocols, and followed these patients until week 60.
About 80% of patients who received ixekizumab every 2 or 4 weeks achieved sPGA scores of 0 or 1 (clear or almost clear) at 12 weeks, compared with 3% of the placebo group, said Dr. Gordon. Rates of PASI 75 response at 12 weeks were 89% for patients treated twice monthly, 83% for patients treated monthly, and 4% for the placebo group. Rates of PASI 100 were 35%, 33%, and 0%, respectively.
Among initial responders who were rerandomized to monthly ixekizumab, 73% maintained sPGA scores of clear or almost clear at week 60, while 78% maintained or achieved PASI 75, and 52% maintained or achieved PASI 100, said Dr. Gordon. The investigators also found a significant positive linear correlation between PASI response and scores on the Dermatology Quality of Life Index (DLQI), with P values ranging from less than .001 to less than .002, Dr. Gordon reported. “We are seeing that clearance is very important in quality of life,” he added. “More patients reported no itching or other negative impact on quality of life with higher levels of response.”
Serious adverse events at week 12 affected 1.4% of the twice-monthly ixekizumab group, 2.8% of the monthly ixekizumab group, and 1.2% of the placebo group, Dr. Gordon reported. Rates of candidiasis were similar among all three arms. Between weeks 12 and 60, the monthly treatment group had three major adverse cardiac events, but exposure-adjusted rates of adverse events for this group were similar to those during the induction period, he added.
Eli Lilly sponsored the trial. Dr. Gordon reported receiving research funding from Eli Lilly and several other pharmaceutical companies.
SAN FRANCISCO – More than 80% of psoriasis patients who received ixekizumab achieved a 75% reduction in the Psoriasis Area and Severity Index score and static Physician Global Assessment scores of clear or almost clear skin at 12 weeks, based on the results of a phase III trial.
In addition, 35% of patients who received 80 mg ixekizumab twice monthly for 12 weeks achieved complete resolution of their psoriasis plaques (PASI 100), reported Dr. Kenneth Gordon of Northwestern University in Chicago. “The overall safety profile for ixekizumab was acceptable during both the induction and maintenance phases,” Dr. Gordon said at the annual meeting of the American Academy of Dermatology.
Ixekizumab is a monoclonal antibody that targets interleukin (IL)-17A, a major cytokine in the pathogenesis of psoriasis. Dr. Gordon and his associates conducted a randomized induction and withdrawal trial that compared twice-monthly or monthly treatment of the biologic with placebo among 1,296 patients. During the 12-week induction phase, 431 patients received placebo, 433 received 80 mg ixekizumab every 2 weeks, and 432 received 80 mg ixekizumab every 4 weeks, said Dr. Gordon. He and his associates then rerandomized the responders (based on PASI 75 and static Patient Global Assessment [sPGA] scores) to one of the three protocols, and followed these patients until week 60.
About 80% of patients who received ixekizumab every 2 or 4 weeks achieved sPGA scores of 0 or 1 (clear or almost clear) at 12 weeks, compared with 3% of the placebo group, said Dr. Gordon. Rates of PASI 75 response at 12 weeks were 89% for patients treated twice monthly, 83% for patients treated monthly, and 4% for the placebo group. Rates of PASI 100 were 35%, 33%, and 0%, respectively.
Among initial responders who were rerandomized to monthly ixekizumab, 73% maintained sPGA scores of clear or almost clear at week 60, while 78% maintained or achieved PASI 75, and 52% maintained or achieved PASI 100, said Dr. Gordon. The investigators also found a significant positive linear correlation between PASI response and scores on the Dermatology Quality of Life Index (DLQI), with P values ranging from less than .001 to less than .002, Dr. Gordon reported. “We are seeing that clearance is very important in quality of life,” he added. “More patients reported no itching or other negative impact on quality of life with higher levels of response.”
Serious adverse events at week 12 affected 1.4% of the twice-monthly ixekizumab group, 2.8% of the monthly ixekizumab group, and 1.2% of the placebo group, Dr. Gordon reported. Rates of candidiasis were similar among all three arms. Between weeks 12 and 60, the monthly treatment group had three major adverse cardiac events, but exposure-adjusted rates of adverse events for this group were similar to those during the induction period, he added.
Eli Lilly sponsored the trial. Dr. Gordon reported receiving research funding from Eli Lilly and several other pharmaceutical companies.
SAN FRANCISCO – More than 80% of psoriasis patients who received ixekizumab achieved a 75% reduction in the Psoriasis Area and Severity Index score and static Physician Global Assessment scores of clear or almost clear skin at 12 weeks, based on the results of a phase III trial.
In addition, 35% of patients who received 80 mg ixekizumab twice monthly for 12 weeks achieved complete resolution of their psoriasis plaques (PASI 100), reported Dr. Kenneth Gordon of Northwestern University in Chicago. “The overall safety profile for ixekizumab was acceptable during both the induction and maintenance phases,” Dr. Gordon said at the annual meeting of the American Academy of Dermatology.
Ixekizumab is a monoclonal antibody that targets interleukin (IL)-17A, a major cytokine in the pathogenesis of psoriasis. Dr. Gordon and his associates conducted a randomized induction and withdrawal trial that compared twice-monthly or monthly treatment of the biologic with placebo among 1,296 patients. During the 12-week induction phase, 431 patients received placebo, 433 received 80 mg ixekizumab every 2 weeks, and 432 received 80 mg ixekizumab every 4 weeks, said Dr. Gordon. He and his associates then rerandomized the responders (based on PASI 75 and static Patient Global Assessment [sPGA] scores) to one of the three protocols, and followed these patients until week 60.
About 80% of patients who received ixekizumab every 2 or 4 weeks achieved sPGA scores of 0 or 1 (clear or almost clear) at 12 weeks, compared with 3% of the placebo group, said Dr. Gordon. Rates of PASI 75 response at 12 weeks were 89% for patients treated twice monthly, 83% for patients treated monthly, and 4% for the placebo group. Rates of PASI 100 were 35%, 33%, and 0%, respectively.
Among initial responders who were rerandomized to monthly ixekizumab, 73% maintained sPGA scores of clear or almost clear at week 60, while 78% maintained or achieved PASI 75, and 52% maintained or achieved PASI 100, said Dr. Gordon. The investigators also found a significant positive linear correlation between PASI response and scores on the Dermatology Quality of Life Index (DLQI), with P values ranging from less than .001 to less than .002, Dr. Gordon reported. “We are seeing that clearance is very important in quality of life,” he added. “More patients reported no itching or other negative impact on quality of life with higher levels of response.”
Serious adverse events at week 12 affected 1.4% of the twice-monthly ixekizumab group, 2.8% of the monthly ixekizumab group, and 1.2% of the placebo group, Dr. Gordon reported. Rates of candidiasis were similar among all three arms. Between weeks 12 and 60, the monthly treatment group had three major adverse cardiac events, but exposure-adjusted rates of adverse events for this group were similar to those during the induction period, he added.
Eli Lilly sponsored the trial. Dr. Gordon reported receiving research funding from Eli Lilly and several other pharmaceutical companies.
AT THE AAD ANNUAL MEETING
Key clinical point: The investigational anti-IL-17A antibody ixekizumab met its endpoints at week 12 in patients with plaque psoriasis, and showed durability of response at 60 weeks.
Major finding: More than 80% of treated patients achieved PASI 75 and static Physician Global Assessment scores of clear or almost clear skin at 12 weeks.
Data source: A phase III trial of 1,296 patients with plaque psoriasis.
Disclosures: Eli Lilly sponsored the trial. Dr. Gordon reported receiving research funding from Eli Lilly and several other pharmaceutical companies.
Traditional risk factors raise cardiovascular risk in PsA
Psoriatic arthritis is associated with higher rates of self-reported angina pectoris, percutaneous coronary intervention, hypertension, obesity, and smoking than are observed in the general population, a large population-based cohort study found.
However, the overall rate of cardiovascular (CV) disease (defined as one or more of angina pectoris, myocardial infarction, cerebrovascular stroke, coronary artery bypass graft, or percutaneous coronary intervention) was 8% in both the 338 patients with psoriatic arthritis (PsA) and the 50,468 controls in the Nord-Trøndelag Health Study. A similar percentage of patients with PsA (13.4%) and controls (11.3%) were deemed to be at very high 10-year risk for a fatal cardiovascular event based on the Systematic Coronary Risk Evaluation (SCORE) algorithm, and the median risk calculated by the algorithm was similar for both groups, Dr. Agnete Malm Gulati of St. Olavs Hospital, Trondheim, Norway, and her coauthors reported. But patients with PsA had significantly higher C-reactive protein levels, body mass index, and diastolic blood pressure, which are not included in the SCORE algorithm.
Patients with PsA had lived with the disease for a mean of 9.3 years, and both patients and controls had a mean age of about 54 years.
The study’s authors said their findings supported previous suggestions that the increased risk of CV disease in psoriatic arthritis was largely a reflection of an increased prevalence of conventional risk factors such as obesity, smoking, and hypertension (Ann. Rheum. Dis. 2015 March 26 [doi:10.1136/annrheumdis-2014-206824]).
“In RA [rheumatoid arthritis], it is shown that atherosclerosis cannot solely be explained by the presence of traditional CV risk factors, and chronic inflammation has also been proposed as being responsible for the enhanced development of atherosclerosis in these patients, [and] this may also be the case for patients with PsA,” the investigators wrote.
The study was supported by Revmafondet i Trondheim, and one author reported financial support from Pfizer. There were no other conflicts of interest declared.
Psoriatic arthritis is associated with higher rates of self-reported angina pectoris, percutaneous coronary intervention, hypertension, obesity, and smoking than are observed in the general population, a large population-based cohort study found.
However, the overall rate of cardiovascular (CV) disease (defined as one or more of angina pectoris, myocardial infarction, cerebrovascular stroke, coronary artery bypass graft, or percutaneous coronary intervention) was 8% in both the 338 patients with psoriatic arthritis (PsA) and the 50,468 controls in the Nord-Trøndelag Health Study. A similar percentage of patients with PsA (13.4%) and controls (11.3%) were deemed to be at very high 10-year risk for a fatal cardiovascular event based on the Systematic Coronary Risk Evaluation (SCORE) algorithm, and the median risk calculated by the algorithm was similar for both groups, Dr. Agnete Malm Gulati of St. Olavs Hospital, Trondheim, Norway, and her coauthors reported. But patients with PsA had significantly higher C-reactive protein levels, body mass index, and diastolic blood pressure, which are not included in the SCORE algorithm.
Patients with PsA had lived with the disease for a mean of 9.3 years, and both patients and controls had a mean age of about 54 years.
The study’s authors said their findings supported previous suggestions that the increased risk of CV disease in psoriatic arthritis was largely a reflection of an increased prevalence of conventional risk factors such as obesity, smoking, and hypertension (Ann. Rheum. Dis. 2015 March 26 [doi:10.1136/annrheumdis-2014-206824]).
“In RA [rheumatoid arthritis], it is shown that atherosclerosis cannot solely be explained by the presence of traditional CV risk factors, and chronic inflammation has also been proposed as being responsible for the enhanced development of atherosclerosis in these patients, [and] this may also be the case for patients with PsA,” the investigators wrote.
The study was supported by Revmafondet i Trondheim, and one author reported financial support from Pfizer. There were no other conflicts of interest declared.
Psoriatic arthritis is associated with higher rates of self-reported angina pectoris, percutaneous coronary intervention, hypertension, obesity, and smoking than are observed in the general population, a large population-based cohort study found.
However, the overall rate of cardiovascular (CV) disease (defined as one or more of angina pectoris, myocardial infarction, cerebrovascular stroke, coronary artery bypass graft, or percutaneous coronary intervention) was 8% in both the 338 patients with psoriatic arthritis (PsA) and the 50,468 controls in the Nord-Trøndelag Health Study. A similar percentage of patients with PsA (13.4%) and controls (11.3%) were deemed to be at very high 10-year risk for a fatal cardiovascular event based on the Systematic Coronary Risk Evaluation (SCORE) algorithm, and the median risk calculated by the algorithm was similar for both groups, Dr. Agnete Malm Gulati of St. Olavs Hospital, Trondheim, Norway, and her coauthors reported. But patients with PsA had significantly higher C-reactive protein levels, body mass index, and diastolic blood pressure, which are not included in the SCORE algorithm.
Patients with PsA had lived with the disease for a mean of 9.3 years, and both patients and controls had a mean age of about 54 years.
The study’s authors said their findings supported previous suggestions that the increased risk of CV disease in psoriatic arthritis was largely a reflection of an increased prevalence of conventional risk factors such as obesity, smoking, and hypertension (Ann. Rheum. Dis. 2015 March 26 [doi:10.1136/annrheumdis-2014-206824]).
“In RA [rheumatoid arthritis], it is shown that atherosclerosis cannot solely be explained by the presence of traditional CV risk factors, and chronic inflammation has also been proposed as being responsible for the enhanced development of atherosclerosis in these patients, [and] this may also be the case for patients with PsA,” the investigators wrote.
The study was supported by Revmafondet i Trondheim, and one author reported financial support from Pfizer. There were no other conflicts of interest declared.
FROM ANNALS OF THE RHEUMATIC DISEASES
Key clinical point: The increased risk of cardiovascular disease in psoriatic arthritis is largely because of an increased prevalence of conventional risk factors such as obesity, smoking, and hypertension.
Major finding: Psoriatic arthritis is associated with higher rates of self-reported angina pectoris, percutaneous coronary intervention, hypertension, obesity, and smoking.
Data source: A prospective, population-based cohort study of 338 patients with psoriatic arthritis and 50,468 controls.
Disclosures: The study was supported by Revmafondet i Trondheim, and one author reported financial support from Pfizer. There were no other conflicts of interest declared.
No increased risk of lung disease with methotrexate
Methotrexate is not associated with an increased risk of pulmonary disease in patients taking it for the treatment of psoriatic arthritis, psoriasis, or inflammatory bowel disease, a meta-analysis has found.
The analysis of results from seven double-blind, randomized, controlled studies, involving a total of 1,640 participants, showed no increased risk of total adverse respiratory events – infectious or noninfectious – or pulmonary deaths in patients taking methotrexate, compared with controls, according to Dr. Richard Conway of the department of rheumatology at Galway (Ireland) University Hospitals and his coauthors.
Methotrexate has previously been implicated as a cause of lung toxicity, and the prevalence of methotrexate-related interstitial lung disease has been reported as high as 11.6% in rheumatoid arthritis, but studies of methotrexate-induced lung disease are confounded by the higher risk of pulmonary infections among patients with rheumatoid arthritis, the authors said (BMJ 2015 [doi:10.1136/bmj.h1269]).
“These findings, coupled with those of a previous study in rheumatoid arthritis, suggest that methotrexate-related lung disease is rare, if it exists at all,” the investigators wrote.
The investigators had no specific source of funding for the study and had no conflicts of interest to declare.
Methotrexate is not associated with an increased risk of pulmonary disease in patients taking it for the treatment of psoriatic arthritis, psoriasis, or inflammatory bowel disease, a meta-analysis has found.
The analysis of results from seven double-blind, randomized, controlled studies, involving a total of 1,640 participants, showed no increased risk of total adverse respiratory events – infectious or noninfectious – or pulmonary deaths in patients taking methotrexate, compared with controls, according to Dr. Richard Conway of the department of rheumatology at Galway (Ireland) University Hospitals and his coauthors.
Methotrexate has previously been implicated as a cause of lung toxicity, and the prevalence of methotrexate-related interstitial lung disease has been reported as high as 11.6% in rheumatoid arthritis, but studies of methotrexate-induced lung disease are confounded by the higher risk of pulmonary infections among patients with rheumatoid arthritis, the authors said (BMJ 2015 [doi:10.1136/bmj.h1269]).
“These findings, coupled with those of a previous study in rheumatoid arthritis, suggest that methotrexate-related lung disease is rare, if it exists at all,” the investigators wrote.
The investigators had no specific source of funding for the study and had no conflicts of interest to declare.
Methotrexate is not associated with an increased risk of pulmonary disease in patients taking it for the treatment of psoriatic arthritis, psoriasis, or inflammatory bowel disease, a meta-analysis has found.
The analysis of results from seven double-blind, randomized, controlled studies, involving a total of 1,640 participants, showed no increased risk of total adverse respiratory events – infectious or noninfectious – or pulmonary deaths in patients taking methotrexate, compared with controls, according to Dr. Richard Conway of the department of rheumatology at Galway (Ireland) University Hospitals and his coauthors.
Methotrexate has previously been implicated as a cause of lung toxicity, and the prevalence of methotrexate-related interstitial lung disease has been reported as high as 11.6% in rheumatoid arthritis, but studies of methotrexate-induced lung disease are confounded by the higher risk of pulmonary infections among patients with rheumatoid arthritis, the authors said (BMJ 2015 [doi:10.1136/bmj.h1269]).
“These findings, coupled with those of a previous study in rheumatoid arthritis, suggest that methotrexate-related lung disease is rare, if it exists at all,” the investigators wrote.
The investigators had no specific source of funding for the study and had no conflicts of interest to declare.
FROM BMJ
Key clinical point: Methotrexate is not associated with an increased risk of pulmonary disease.
Major finding: There was no increased risk of total adverse respiratory events – infectious or noninfectious – or pulmonary deaths in patients taking methotrexate, compared with controls.
Data source: Meta-analysis of seven double-blind, randomized, controlled studies, involving a total of 1,640 participants.
Disclosures: The investigators had no specific source of funding for the study and had no conflicts of interest to declare.
Methotrexate and biologics linked to higher zoster risk in psoriasis
The combination of methotrexate and biologics for the treatment of psoriasis may increase the risk of herpes zoster infection, according to a large, database cohort study published in JAMA Dermatology.
Analysis of medical records for 95,941 patients with psoriasis showed treatment with both biologic medications and methotrexate was associated with a significant 66% increase in the incidence of herpes zoster, compared with a control group, during more than 11 years of follow-up. Episodes of herpes zoster (HZ) occurring in a patient following at least 6 consecutive treatment-free months or in a patient who did not receive any of the listed medications at any time during follow-up were counted as HZ episodes in the control group. The multivariate regression analysis was adjusted for age, sex, psoriasis severity, Charlson comorbidity index, steroid administration per year, and socioeconomic status.
The study found phototherapy, methotrexate alone, cyclosporine, or biologic medications as single agents did not significantly increase the risk, while acitretin therapy was associated with a significant 31% decrease in the incidence of herpes zoster (JAMA Dermatol. 2015 March 22 [doi:10.1001/jamadermatol.2014.4956]).
“Our study results might suggest the need for prophylactic vaccination against VZV [varicella zoster virus] in patients with psoriasis and multiple risk factors for HZ (eg, age, female sex) as part of the preparation for biologic medication treatments in the appropriate clinical setting,” wrote Dr. Guy Shalom of Soroka Medical Center, Beer-Sheva, Israel, and his coauthors.
One author reported consultancies and research grants from a range of pharmaceutical companies. There were no other disclosures.
The combination of methotrexate and biologics for the treatment of psoriasis may increase the risk of herpes zoster infection, according to a large, database cohort study published in JAMA Dermatology.
Analysis of medical records for 95,941 patients with psoriasis showed treatment with both biologic medications and methotrexate was associated with a significant 66% increase in the incidence of herpes zoster, compared with a control group, during more than 11 years of follow-up. Episodes of herpes zoster (HZ) occurring in a patient following at least 6 consecutive treatment-free months or in a patient who did not receive any of the listed medications at any time during follow-up were counted as HZ episodes in the control group. The multivariate regression analysis was adjusted for age, sex, psoriasis severity, Charlson comorbidity index, steroid administration per year, and socioeconomic status.
The study found phototherapy, methotrexate alone, cyclosporine, or biologic medications as single agents did not significantly increase the risk, while acitretin therapy was associated with a significant 31% decrease in the incidence of herpes zoster (JAMA Dermatol. 2015 March 22 [doi:10.1001/jamadermatol.2014.4956]).
“Our study results might suggest the need for prophylactic vaccination against VZV [varicella zoster virus] in patients with psoriasis and multiple risk factors for HZ (eg, age, female sex) as part of the preparation for biologic medication treatments in the appropriate clinical setting,” wrote Dr. Guy Shalom of Soroka Medical Center, Beer-Sheva, Israel, and his coauthors.
One author reported consultancies and research grants from a range of pharmaceutical companies. There were no other disclosures.
The combination of methotrexate and biologics for the treatment of psoriasis may increase the risk of herpes zoster infection, according to a large, database cohort study published in JAMA Dermatology.
Analysis of medical records for 95,941 patients with psoriasis showed treatment with both biologic medications and methotrexate was associated with a significant 66% increase in the incidence of herpes zoster, compared with a control group, during more than 11 years of follow-up. Episodes of herpes zoster (HZ) occurring in a patient following at least 6 consecutive treatment-free months or in a patient who did not receive any of the listed medications at any time during follow-up were counted as HZ episodes in the control group. The multivariate regression analysis was adjusted for age, sex, psoriasis severity, Charlson comorbidity index, steroid administration per year, and socioeconomic status.
The study found phototherapy, methotrexate alone, cyclosporine, or biologic medications as single agents did not significantly increase the risk, while acitretin therapy was associated with a significant 31% decrease in the incidence of herpes zoster (JAMA Dermatol. 2015 March 22 [doi:10.1001/jamadermatol.2014.4956]).
“Our study results might suggest the need for prophylactic vaccination against VZV [varicella zoster virus] in patients with psoriasis and multiple risk factors for HZ (eg, age, female sex) as part of the preparation for biologic medication treatments in the appropriate clinical setting,” wrote Dr. Guy Shalom of Soroka Medical Center, Beer-Sheva, Israel, and his coauthors.
One author reported consultancies and research grants from a range of pharmaceutical companies. There were no other disclosures.
FROM JAMA DERMATOLOGY
Key clinical point: The combination of methotrexate and biologics for the treatment of psoriasis may increase the risk of herpes zoster infection.
Major finding: Combination therapy with both biologic medications and methotrexate was associated with a significant 66% increase in the incidence of herpes zoster over more than 11 years of follow-up
Data source: Analysis of medical records for 95,941 patients with psoriasis.
Disclosures: One author reported consultancies and research grants from a range of pharmaceutical companies. There were no other disclosures.
Secukinumab beat ustekinumab for psoriasis, with difference by week four
SAN FRANCISCO– Secukinumab met its primary endpoint in a head-to-head trial, achieving 21% more PASI 90 responses than ustekinumab in patients with moderate to severe plaque psoriasis, according to interim, week 16 results from the phase IIIb CLEAR trial.
Patients’ responses to the two biologic agents had diverged by four weeks of treatment, when half the secukinumab group had achieved the secondary endpoint of PASI 75, compared with 20.6% of the ustekinumab group (P < .0001), Dr. Diamant Thaci said at the annual meeting of the American Academy of Dermatology.
“Secukinumab was superior to ustekinumab, even at early time points,” said Dr. Thaci of the Comprehensive Center for Inflammation Medicine at University Medical School Schleswig-Holstein, Lübeck, Germany. “This was very remarkable. Especially remarkable was the early difference for PASI 75.”
The biologic agents also showed similar safety profiles, and secukinumab yielded no new safety signals besides those observed in its pivotal phase III studies. “The most common adverse events were what we have seen in daily practice, including headache, nasopharyngitis, diarrhea, fatigue, and arthralgia,” Dr. Thaci said.
Secukinumab works by binding specifically to the interleukin-17A cytokine, thereby blocking its interaction with its receptor. The 52-week, phase IIIb CLEAR trial, which is underway at 134 sites in 24 countries, is comparing the safety and efficacy of secukinumab and ustekinumab in 679 patients with moderate to severe plaque psoriasis. Patients had baseline PASI scores of at least 12 and at least 10% body surface area involvement, no previous exposure to either biologic agent, and inadequate responses to topical treatments or phototherapy. Researchers randomized the patients to either 300 mg secukinumab given subcutaneously at baseline, at weeks one through four, and every four weeks thereafter, or to ustekinumab, dosed according to label.
The study cohorts resembled one another demographically at baseline. At week 16, 79% of patients given secukinumab had achieved a PASI 90 response, compared with 57.6% of the ustekinumab group (P < .0001). Furthermore, 44.3% of the secukinumab arm had achieved the secondary endpoint of completely clear skin (a PASI 100 response), compared with 28.4% of the ustekinumab group (P < .0001), Dr. Thaci reported.
After an average of 110 to 111 days of treatment, infections and infestations developed in 29.3% of the secukinumab group and 25.3% of ustekinumab group. Three percent of each group developed serious but non-fatal adverse events, with no deaths reported to date.
“Secukinumab treatment – even at early time points – has demonstrated superiority to ustekinumab in clearing the skin of subjects with moderate to severe psoriasis, with a comparable safety profile,” Dr. Thaci concluded. “It is very clear that we will be able to show and to observe that his agent is leading to improved quality of life.”
Dr. Thaci reported receiving research support from Dignity Sciences.
SAN FRANCISCO– Secukinumab met its primary endpoint in a head-to-head trial, achieving 21% more PASI 90 responses than ustekinumab in patients with moderate to severe plaque psoriasis, according to interim, week 16 results from the phase IIIb CLEAR trial.
Patients’ responses to the two biologic agents had diverged by four weeks of treatment, when half the secukinumab group had achieved the secondary endpoint of PASI 75, compared with 20.6% of the ustekinumab group (P < .0001), Dr. Diamant Thaci said at the annual meeting of the American Academy of Dermatology.
“Secukinumab was superior to ustekinumab, even at early time points,” said Dr. Thaci of the Comprehensive Center for Inflammation Medicine at University Medical School Schleswig-Holstein, Lübeck, Germany. “This was very remarkable. Especially remarkable was the early difference for PASI 75.”
The biologic agents also showed similar safety profiles, and secukinumab yielded no new safety signals besides those observed in its pivotal phase III studies. “The most common adverse events were what we have seen in daily practice, including headache, nasopharyngitis, diarrhea, fatigue, and arthralgia,” Dr. Thaci said.
Secukinumab works by binding specifically to the interleukin-17A cytokine, thereby blocking its interaction with its receptor. The 52-week, phase IIIb CLEAR trial, which is underway at 134 sites in 24 countries, is comparing the safety and efficacy of secukinumab and ustekinumab in 679 patients with moderate to severe plaque psoriasis. Patients had baseline PASI scores of at least 12 and at least 10% body surface area involvement, no previous exposure to either biologic agent, and inadequate responses to topical treatments or phototherapy. Researchers randomized the patients to either 300 mg secukinumab given subcutaneously at baseline, at weeks one through four, and every four weeks thereafter, or to ustekinumab, dosed according to label.
The study cohorts resembled one another demographically at baseline. At week 16, 79% of patients given secukinumab had achieved a PASI 90 response, compared with 57.6% of the ustekinumab group (P < .0001). Furthermore, 44.3% of the secukinumab arm had achieved the secondary endpoint of completely clear skin (a PASI 100 response), compared with 28.4% of the ustekinumab group (P < .0001), Dr. Thaci reported.
After an average of 110 to 111 days of treatment, infections and infestations developed in 29.3% of the secukinumab group and 25.3% of ustekinumab group. Three percent of each group developed serious but non-fatal adverse events, with no deaths reported to date.
“Secukinumab treatment – even at early time points – has demonstrated superiority to ustekinumab in clearing the skin of subjects with moderate to severe psoriasis, with a comparable safety profile,” Dr. Thaci concluded. “It is very clear that we will be able to show and to observe that his agent is leading to improved quality of life.”
Dr. Thaci reported receiving research support from Dignity Sciences.
SAN FRANCISCO– Secukinumab met its primary endpoint in a head-to-head trial, achieving 21% more PASI 90 responses than ustekinumab in patients with moderate to severe plaque psoriasis, according to interim, week 16 results from the phase IIIb CLEAR trial.
Patients’ responses to the two biologic agents had diverged by four weeks of treatment, when half the secukinumab group had achieved the secondary endpoint of PASI 75, compared with 20.6% of the ustekinumab group (P < .0001), Dr. Diamant Thaci said at the annual meeting of the American Academy of Dermatology.
“Secukinumab was superior to ustekinumab, even at early time points,” said Dr. Thaci of the Comprehensive Center for Inflammation Medicine at University Medical School Schleswig-Holstein, Lübeck, Germany. “This was very remarkable. Especially remarkable was the early difference for PASI 75.”
The biologic agents also showed similar safety profiles, and secukinumab yielded no new safety signals besides those observed in its pivotal phase III studies. “The most common adverse events were what we have seen in daily practice, including headache, nasopharyngitis, diarrhea, fatigue, and arthralgia,” Dr. Thaci said.
Secukinumab works by binding specifically to the interleukin-17A cytokine, thereby blocking its interaction with its receptor. The 52-week, phase IIIb CLEAR trial, which is underway at 134 sites in 24 countries, is comparing the safety and efficacy of secukinumab and ustekinumab in 679 patients with moderate to severe plaque psoriasis. Patients had baseline PASI scores of at least 12 and at least 10% body surface area involvement, no previous exposure to either biologic agent, and inadequate responses to topical treatments or phototherapy. Researchers randomized the patients to either 300 mg secukinumab given subcutaneously at baseline, at weeks one through four, and every four weeks thereafter, or to ustekinumab, dosed according to label.
The study cohorts resembled one another demographically at baseline. At week 16, 79% of patients given secukinumab had achieved a PASI 90 response, compared with 57.6% of the ustekinumab group (P < .0001). Furthermore, 44.3% of the secukinumab arm had achieved the secondary endpoint of completely clear skin (a PASI 100 response), compared with 28.4% of the ustekinumab group (P < .0001), Dr. Thaci reported.
After an average of 110 to 111 days of treatment, infections and infestations developed in 29.3% of the secukinumab group and 25.3% of ustekinumab group. Three percent of each group developed serious but non-fatal adverse events, with no deaths reported to date.
“Secukinumab treatment – even at early time points – has demonstrated superiority to ustekinumab in clearing the skin of subjects with moderate to severe psoriasis, with a comparable safety profile,” Dr. Thaci concluded. “It is very clear that we will be able to show and to observe that his agent is leading to improved quality of life.”
Dr. Thaci reported receiving research support from Dignity Sciences.
Key clinical point:Secukinumab achieved significantly better responses than ustekinumab in the treatment of moderate to severe psoriasis.
Major finding:At week 16, 79% of patients given secukinumab had achieved a PASI 90 response, compared with 57.6% of the ustekinumab group (P < .0001).
Data source: Interim results from a phase IIIb trial comparing secukinumab and ustekinumab in 679 patients with moderate to severe plaque psoriasis.
Disclosures: Dr. Thaci reported receiving research support from Dignity Sciences.
Brodalumab achieved primary endpoints for moderate to severe psoriasis at 52 weeks
SAN FRANCISCO– Significantly more psoriasis patients who received the investigational biologic agent brodalumab achieved a PASI 100 response compared with those who received ustekinumab, and clinical responses persisted through 52 weeks, according to data from the pivotal phase III AMAGINE-2 trial.
Brodalumab also met its co-primary endpoints (PASI 75 and sPGA 0 or 1) compared with placebo at week 12 when given at doses of either 210 or 140 mg every two weeks, Dr. Mark Lebwohl said at the annual meeting of the American Academy of Dermatology.
But patients maintained the best responses at the higher brodalumab dose, Dr. Lebwohl, professor and chair of dermatology at the Icahn School of Medicine at Mount Sinai, New York, reported. “Nearly 44% of patients in this group had not a dot of psoriasis left,” he said.
The interleukin-17 (IL-17) receptor and cytokine family play a key role in the pathogenesis of plaque psoriasis. Brodalumab works by binding the IL-17 receptor, thereby blocking binding by the A, F, and A/F IL-17 cytokines. The AMAGINE-2 trial is the last of a trio of phase III studies to assess brodalumab’s safety and efficacy in patients with moderate to severe plaque psoriasis, Dr. Lebwohl and his associates noted. The findings are consistent with those from earlier trials, they said.
For the study, the researchers enrolled 1,831 patients with moderate to severe plaque psoriasis, of whom 1,776 completed the 12-week induction phase. During induction, patients received either 210 or 140 mg brodalumab, 45 mg of ustekinumab (or 90 mg if they weighed more than 100 kg), or placebo. At week 12, patients were re-randomized to one of the brodalumab or ustekinumab arms.
Fully 44% of patients who received 210 mg brodalumab achieved total clearance of skin disease, or Psoriasis Area Severity Index (PASI) 100 – twice the proportion of the ustekinumab group (22%; P < .001), Dr. Lebwohl said. The 210-mg brodalumab dose also achieved the highest PASI 75 response rate (86%, compared with 70% for ustekinumab, 67% for 140 mg brodalumab, and 8% for placebo), although the adjusted p-value comparing 210 mg brodalumab and ustekinumab did not reach statistical significance (P = .078), he noted. Finally, 79% of patients who received 210 mg brodalumab and 58% of those who received 140 mg brodalumab achieved clear or almost clear skin at week 12 according to the static Physician Global Assessment (sPGA), compared with only 4% of the placebo group (P < .001), he reported.
Brodalumab’s safety profile during the 12-week induction phase resembled that for previous trials, said Dr. Lebwohl. The most common adverse events were nasopharyngitis, upper respiratory tract infection, headache, and arthralgia. “But the punch line was Candida,” he said. Candidiasis affected 0.6% of patients in the placebo arm, compared with 1.4% for brodalumab-treated patients at week 12. By week 52, about 4% to 6.5% of treated patients had developed Candida infections.
Similarly small proportions of patients across all arms experienced serious side effects (1% to 2.6%) during the placebo-controlled period, noted Dr. Lebwohl. After adjusting for exposure time, rates of adverse events were similar for all groups, he said. “However, due to disparity in patient-years of exposure between treatment groups, we cannot draw conclusions about potential dose effects,” he added.
Dr. Lebwohl reported receiving research support from Amgen, which is developing brodalumab together with AstraZeneca/MedImmune.
SAN FRANCISCO– Significantly more psoriasis patients who received the investigational biologic agent brodalumab achieved a PASI 100 response compared with those who received ustekinumab, and clinical responses persisted through 52 weeks, according to data from the pivotal phase III AMAGINE-2 trial.
Brodalumab also met its co-primary endpoints (PASI 75 and sPGA 0 or 1) compared with placebo at week 12 when given at doses of either 210 or 140 mg every two weeks, Dr. Mark Lebwohl said at the annual meeting of the American Academy of Dermatology.
But patients maintained the best responses at the higher brodalumab dose, Dr. Lebwohl, professor and chair of dermatology at the Icahn School of Medicine at Mount Sinai, New York, reported. “Nearly 44% of patients in this group had not a dot of psoriasis left,” he said.
The interleukin-17 (IL-17) receptor and cytokine family play a key role in the pathogenesis of plaque psoriasis. Brodalumab works by binding the IL-17 receptor, thereby blocking binding by the A, F, and A/F IL-17 cytokines. The AMAGINE-2 trial is the last of a trio of phase III studies to assess brodalumab’s safety and efficacy in patients with moderate to severe plaque psoriasis, Dr. Lebwohl and his associates noted. The findings are consistent with those from earlier trials, they said.
For the study, the researchers enrolled 1,831 patients with moderate to severe plaque psoriasis, of whom 1,776 completed the 12-week induction phase. During induction, patients received either 210 or 140 mg brodalumab, 45 mg of ustekinumab (or 90 mg if they weighed more than 100 kg), or placebo. At week 12, patients were re-randomized to one of the brodalumab or ustekinumab arms.
Fully 44% of patients who received 210 mg brodalumab achieved total clearance of skin disease, or Psoriasis Area Severity Index (PASI) 100 – twice the proportion of the ustekinumab group (22%; P < .001), Dr. Lebwohl said. The 210-mg brodalumab dose also achieved the highest PASI 75 response rate (86%, compared with 70% for ustekinumab, 67% for 140 mg brodalumab, and 8% for placebo), although the adjusted p-value comparing 210 mg brodalumab and ustekinumab did not reach statistical significance (P = .078), he noted. Finally, 79% of patients who received 210 mg brodalumab and 58% of those who received 140 mg brodalumab achieved clear or almost clear skin at week 12 according to the static Physician Global Assessment (sPGA), compared with only 4% of the placebo group (P < .001), he reported.
Brodalumab’s safety profile during the 12-week induction phase resembled that for previous trials, said Dr. Lebwohl. The most common adverse events were nasopharyngitis, upper respiratory tract infection, headache, and arthralgia. “But the punch line was Candida,” he said. Candidiasis affected 0.6% of patients in the placebo arm, compared with 1.4% for brodalumab-treated patients at week 12. By week 52, about 4% to 6.5% of treated patients had developed Candida infections.
Similarly small proportions of patients across all arms experienced serious side effects (1% to 2.6%) during the placebo-controlled period, noted Dr. Lebwohl. After adjusting for exposure time, rates of adverse events were similar for all groups, he said. “However, due to disparity in patient-years of exposure between treatment groups, we cannot draw conclusions about potential dose effects,” he added.
Dr. Lebwohl reported receiving research support from Amgen, which is developing brodalumab together with AstraZeneca/MedImmune.
SAN FRANCISCO– Significantly more psoriasis patients who received the investigational biologic agent brodalumab achieved a PASI 100 response compared with those who received ustekinumab, and clinical responses persisted through 52 weeks, according to data from the pivotal phase III AMAGINE-2 trial.
Brodalumab also met its co-primary endpoints (PASI 75 and sPGA 0 or 1) compared with placebo at week 12 when given at doses of either 210 or 140 mg every two weeks, Dr. Mark Lebwohl said at the annual meeting of the American Academy of Dermatology.
But patients maintained the best responses at the higher brodalumab dose, Dr. Lebwohl, professor and chair of dermatology at the Icahn School of Medicine at Mount Sinai, New York, reported. “Nearly 44% of patients in this group had not a dot of psoriasis left,” he said.
The interleukin-17 (IL-17) receptor and cytokine family play a key role in the pathogenesis of plaque psoriasis. Brodalumab works by binding the IL-17 receptor, thereby blocking binding by the A, F, and A/F IL-17 cytokines. The AMAGINE-2 trial is the last of a trio of phase III studies to assess brodalumab’s safety and efficacy in patients with moderate to severe plaque psoriasis, Dr. Lebwohl and his associates noted. The findings are consistent with those from earlier trials, they said.
For the study, the researchers enrolled 1,831 patients with moderate to severe plaque psoriasis, of whom 1,776 completed the 12-week induction phase. During induction, patients received either 210 or 140 mg brodalumab, 45 mg of ustekinumab (or 90 mg if they weighed more than 100 kg), or placebo. At week 12, patients were re-randomized to one of the brodalumab or ustekinumab arms.
Fully 44% of patients who received 210 mg brodalumab achieved total clearance of skin disease, or Psoriasis Area Severity Index (PASI) 100 – twice the proportion of the ustekinumab group (22%; P < .001), Dr. Lebwohl said. The 210-mg brodalumab dose also achieved the highest PASI 75 response rate (86%, compared with 70% for ustekinumab, 67% for 140 mg brodalumab, and 8% for placebo), although the adjusted p-value comparing 210 mg brodalumab and ustekinumab did not reach statistical significance (P = .078), he noted. Finally, 79% of patients who received 210 mg brodalumab and 58% of those who received 140 mg brodalumab achieved clear or almost clear skin at week 12 according to the static Physician Global Assessment (sPGA), compared with only 4% of the placebo group (P < .001), he reported.
Brodalumab’s safety profile during the 12-week induction phase resembled that for previous trials, said Dr. Lebwohl. The most common adverse events were nasopharyngitis, upper respiratory tract infection, headache, and arthralgia. “But the punch line was Candida,” he said. Candidiasis affected 0.6% of patients in the placebo arm, compared with 1.4% for brodalumab-treated patients at week 12. By week 52, about 4% to 6.5% of treated patients had developed Candida infections.
Similarly small proportions of patients across all arms experienced serious side effects (1% to 2.6%) during the placebo-controlled period, noted Dr. Lebwohl. After adjusting for exposure time, rates of adverse events were similar for all groups, he said. “However, due to disparity in patient-years of exposure between treatment groups, we cannot draw conclusions about potential dose effects,” he added.
Dr. Lebwohl reported receiving research support from Amgen, which is developing brodalumab together with AstraZeneca/MedImmune.
Key clinical point: At 52 weeks, brodalumab met its PASI 100 endpoint compared with ustekinumab in the pivotal phase III AMAGINE-2 trial.
Major finding: Forty-four percent of patients who received 210 mg brodalumab achieved PASI 100 compared with 22% of the ustekinumab group (P < .001).
Data source: Randomized, placebo-controlled phase III trial of brodalumab, ustekinumab, and placebo in 1,831 patients with moderate to severe plaque psoriasis.
Disclosures: Dr. Lebwohl reported receiving research support from Amgen, which is developing brodalumab together with AstraZeneca/MedImmune.
Halting biologics before surgery tied to flares in psoriasis, psoriatic arthritis
Interrupting biologic therapy before surgery led to flares in psoriasis and psoriatic arthritis and did not appear to prevent postoperative complications in a small, retrospective cohort study.
“Our findings are in keeping with most of the existing literature on this topic,” said Dr. Waseem Bakkour and his associates at the University of Manchester (England). “However, it is important to acknowledge the deficiencies of our data, in particular the small data set and retrospective study design with numerous complexities associated with interpreting it” (J. Eur. Acad. Dermatol. Venereol. 2015 Mar. 2 [doi:10.1111/jdv.12997]).
The British Association of Dermatologists and the British Society for Rheumatology recommend stopping biologics for at least four half-lives before surgery, but the guideline is based mostly on retrospective studies of rheumatoid arthritis and inflammatory bowel disease, the researchers said. For their study, they reviewed electronic health records from 42 patients with psoriasis and psoriatic arthritis who underwent 77 major and minor surgical procedures during a 6-year period. Discontinuing biologic therapy before surgery was linked to a significant risk of flare of psoriasis or psoriatic arthritis (40% with stoppage vs. 8.7% with continuation; P = .003). For three-quarters of procedures, patients continued biologic therapy (usually etanercept, but also adalimumab and infliximab), with no apparent effect on rates of postoperative infections or delayed wound healing. About 48% of procedures required general anesthesia, and most of the rest were skin surgeries.
The findings contradict those from a larger retrospective study (Arthritis Care Res. 2006;55:333-7) that linked biologic therapy before orthopedic surgery to a fourfold rise in the odds of postoperative infections, the investigators noted. “Whilst the current evidence, not surprisingly, suggests a link between stopping treatment and disease flare, it remains equivocal regarding the question of whether continuing biologic therapy perioperatively increases the risk of postsurgical complications,” they wrote.
The authors reported no funding sources. They disclosed financial and advisory relationships with many companies that manufacture biologic therapies.
Interrupting biologic therapy before surgery led to flares in psoriasis and psoriatic arthritis and did not appear to prevent postoperative complications in a small, retrospective cohort study.
“Our findings are in keeping with most of the existing literature on this topic,” said Dr. Waseem Bakkour and his associates at the University of Manchester (England). “However, it is important to acknowledge the deficiencies of our data, in particular the small data set and retrospective study design with numerous complexities associated with interpreting it” (J. Eur. Acad. Dermatol. Venereol. 2015 Mar. 2 [doi:10.1111/jdv.12997]).
The British Association of Dermatologists and the British Society for Rheumatology recommend stopping biologics for at least four half-lives before surgery, but the guideline is based mostly on retrospective studies of rheumatoid arthritis and inflammatory bowel disease, the researchers said. For their study, they reviewed electronic health records from 42 patients with psoriasis and psoriatic arthritis who underwent 77 major and minor surgical procedures during a 6-year period. Discontinuing biologic therapy before surgery was linked to a significant risk of flare of psoriasis or psoriatic arthritis (40% with stoppage vs. 8.7% with continuation; P = .003). For three-quarters of procedures, patients continued biologic therapy (usually etanercept, but also adalimumab and infliximab), with no apparent effect on rates of postoperative infections or delayed wound healing. About 48% of procedures required general anesthesia, and most of the rest were skin surgeries.
The findings contradict those from a larger retrospective study (Arthritis Care Res. 2006;55:333-7) that linked biologic therapy before orthopedic surgery to a fourfold rise in the odds of postoperative infections, the investigators noted. “Whilst the current evidence, not surprisingly, suggests a link between stopping treatment and disease flare, it remains equivocal regarding the question of whether continuing biologic therapy perioperatively increases the risk of postsurgical complications,” they wrote.
The authors reported no funding sources. They disclosed financial and advisory relationships with many companies that manufacture biologic therapies.
Interrupting biologic therapy before surgery led to flares in psoriasis and psoriatic arthritis and did not appear to prevent postoperative complications in a small, retrospective cohort study.
“Our findings are in keeping with most of the existing literature on this topic,” said Dr. Waseem Bakkour and his associates at the University of Manchester (England). “However, it is important to acknowledge the deficiencies of our data, in particular the small data set and retrospective study design with numerous complexities associated with interpreting it” (J. Eur. Acad. Dermatol. Venereol. 2015 Mar. 2 [doi:10.1111/jdv.12997]).
The British Association of Dermatologists and the British Society for Rheumatology recommend stopping biologics for at least four half-lives before surgery, but the guideline is based mostly on retrospective studies of rheumatoid arthritis and inflammatory bowel disease, the researchers said. For their study, they reviewed electronic health records from 42 patients with psoriasis and psoriatic arthritis who underwent 77 major and minor surgical procedures during a 6-year period. Discontinuing biologic therapy before surgery was linked to a significant risk of flare of psoriasis or psoriatic arthritis (40% with stoppage vs. 8.7% with continuation; P = .003). For three-quarters of procedures, patients continued biologic therapy (usually etanercept, but also adalimumab and infliximab), with no apparent effect on rates of postoperative infections or delayed wound healing. About 48% of procedures required general anesthesia, and most of the rest were skin surgeries.
The findings contradict those from a larger retrospective study (Arthritis Care Res. 2006;55:333-7) that linked biologic therapy before orthopedic surgery to a fourfold rise in the odds of postoperative infections, the investigators noted. “Whilst the current evidence, not surprisingly, suggests a link between stopping treatment and disease flare, it remains equivocal regarding the question of whether continuing biologic therapy perioperatively increases the risk of postsurgical complications,” they wrote.
The authors reported no funding sources. They disclosed financial and advisory relationships with many companies that manufacture biologic therapies.
FROM THE JOURNAL OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Key clinical point: Interrupting biologic therapy before surgery led to flares in psoriasis and psoriatic arthritis.
Major finding: Discontinuing biologic therapy before surgery was associated with a significant risk of flare (40% with stoppage vs. 8.7% with continuation; P = .003).
Data source: A retrospective cohort study of 42 patients with psoriasis and/or psoriatic arthritis who underwent 77 surgical procedures.
Disclosures: The authors reported no funding sources. They disclosed financial and advisory relationships with many companies that manufacture biologic therapies.
Novel Psoriasis Therapies and Patient Outcomes, Part 1: Topical Medications
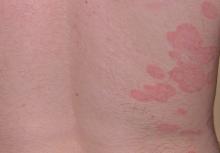
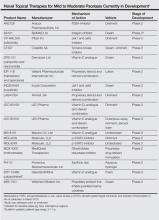
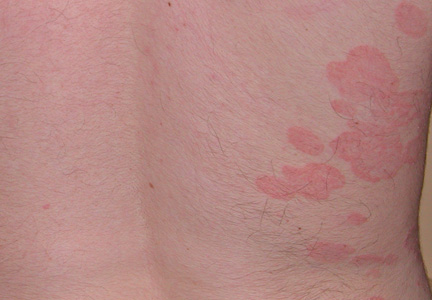
Topical therapies are a mainstay in the management of patients with mild to moderate psoriasis (Figure). Presently, US Food and Drug Administration–approved topical medications that are commercially available for use in patients with psoriasis include corticosteroids, vitamin D3 analogues, calcineurin inhibitors, retinoids, anthralin, and tar-based formulations.1 In recent years, research has furthered our understanding of the molecular mechanisms underlying the pathogenesis of psoriasis and has afforded the development of more targeted therapies. Novel topical medications currently in phase 2 and phase 3 clinical trials are discussed in this article, and a summary is provided in the Table.
AN2728 (Phosphodiesterase 4 Inhibitor)
AN2728 (Anacor Pharmaceuticals, Inc) is a phosphodiesterase 4 inhibitor that blocks the inactivation of cyclic adenosine monophosphate, resulting in decreased production of inflammatory cytokines (eg, IL-6, IL-12, IL-23, tumor necrosis factor α [TNF-α]).2,3 In a randomized, double-blind, phase 2 clinical trial (N=35), 40% of patients treated with AN2728 ointment 5% reported improvement of more than 2 points in overall target plaque severity score versus 6% of patients treated with vehicle. In another randomized, double-blind, dose-response trial of 145 patients, those treated with AN2728 ointment 2% twice daily reported a 60% improvement versus 40% improvement in those treated with AN2728 ointment 0.5% once daily.3 In total, 3 phase 1 trials (registered at www.clinicaltrials.gov with the identifiers NCT01258088, NCT00762658, NCT00763204) and 4 phase 2 trials (NCT01029405, NCT00755196, NCT00759161, NCT01300052) have been completed; results were not available at the time of publication.
AS101 (Integrin Inhibitor)
AS101 (BioMAS Ltd), or ammonium trichloro (dioxoethylene-o,o') tellurate, acts as stimulator of regulatory T cells and a redox modulator inhibiting the leukocyte integrins α4β1 and α4β7 that enable CD4+ T-cell and macrophage extravasation; it also limits expression of the inflammatory cytokines IL-6 and IL-17.4 A randomized, placebo-controlled, double-blind, phase 2 study evaluating the efficacy of AS101 cream 4% twice daily for 12 weeks was withdrawn prior to enrollment (NCT00788424).
Tofacitinib (Janus Kinase 1 and 3 Inhibitor)
Tofacitinib (formerly known as CP-690,550)(Pfizer Inc) is a selective Janus kinase (Jak) 1 and Jak3 inhibitor that limits expression of cytokines that promote inflammation (eg, IFN-γ) and inhibits helper T cells (TH17) by downregulating expression of the IL-23 receptor. Epidermal keratinocyte proliferation in psoriasis is activated by TH17 cells that release IL-17 as well as TH1 cells that release IFN-γ and tumor necrosis factor. A phase 2a trial showed statistically significant improvement from baseline in the target plaque severity score for tofacitinib ointment 2% (least squares mean, −54.4%) versus vehicle (least squares mean, −41.5%).5 Two other phase 2 trials (NCT01246583, NCT00678561) assessing the efficacy, safety, tolerability, and pharmacokinetics of tofacitinib ointment in patients with mild to moderate psoriasis have been completed; results were not available at the time of publication. A phase 2b study that compared 2 dose strengths of tofacitinib ointment—10 mg/g and 20 mg/g—versus placebo over a 12-week period also was completed (NCT01831466); results were not available at the time of publication.
CT327 (Tyrosine Kinase Inhibitor)
CT327 (Creabilis SA) is a tyrosine kinase A (TrkA) inhibitor that affords a novel perspective in the treatment of pruritus by shifting the focus to sensory neurons. In a phase 2b study of 160 patients, a 60% change in the visual analog scale was noted at 8 weeks in the treatment group versus 21% in the placebo group.6 Two other phase 2 studies have been completed, one with a cream formulation of pegylated K252a (NCT00995969) and another with an ointment formulation (NCT01465282); results were not available at the time of publication.
DPS-101 (Vitamin D Analogue)
DPS-101 (Dermipsor Ltd) is a combination of calcipotriol and niacinamide. Calcipotriol is a vitamin D3 analogue that increases IL-10 expression while decreasing IL-8 expression.7 It curbs epidermal keratinocyte proliferation by limiting the expression of polo-like kinase 2 and early growth response-1.8 It also may induce keratinocyte apoptosis.9 Niacinamide is the amide of vitamin B3 and inhibits proinflammatory cytokines such as TNF-α, IL-1β, IL-6, and IL-8.10 In a dose-response phase 2b trial of 168 patients, DPS-101 demonstrated better results than either calcipotriol or niacinamide alone.11
IDP-118 (Proprietary Steroid and Retinoid Combination)
IDP-118 (Valeant Pharmaceuticals International, Inc) is a combination of halobetasol propionate (HP) 0.01% (a topical corticosteroid) and tazar-otene 0.045% (a selective topical retinoid) in a lotion formulation. In isolation, tazarotene is as effective as a mid to highly potent corticosteroid, but irritation may limit its tolerability. The use of combination treatments of mid to highly potent corticosteroids and tazarotene has shown enhanced tolerability and therapeutic efficacy.12 Ongoing studies include a phase 1 trial and a phase 2 trial to evaluate low- and high-strength preparations of IDP-118, respectively (NCT01670513). Another phase 2 trial evaluating the efficacy and safety of IDP-118 lotion (HP 0.01% and tazarotene 0.045%) versus IDP-118 monad HP 0.01% lotion, IDP-118 monad tazar-otene 0.045% lotion, and placebo has been completed (NCT02045277); results were not available at the time of publication.
Ruxolitinib (Jak1 and Jak2 Inhibitor)
Ruxolitinib (formerly known as INCB18424)(Incyte Corporation) is a selective Jak1 and Jak2 inhibitor. A phase 2 trial of ruxolitinib showed a 53% decline in the score for mean total lesions in patients treated with ruxolitinib phosphate cream 1% (P=.033) versus 54% in those treated with ruxolitinib phosphate cream 1.5% (P=.056) and 32% in those treated with placebo.13 Three other phase 2 studies (NCT00617994, NCT00820950, NCT00778700) have been completed; results were not available at the time of publication.
LAS41004 (Proprietary Steroid and Retinoid Combination)
LAS41004 (Almirall, SA) is an ointment containing the corticosteroid betamethasone dipropionate and the retinoid bexarotene that is being evaluated for treatment of mild to moderate psoriasis. Five phase 2 studies (NCT01119339, NCT01283698, NCT01360944, NCT02111499, NCT01462643) have been completed; results were not available at the time of publication. A randomized, double-blind, phase 2a study (NCT02180464) with a left-right design assessing clinical response to LAS41004 versus control in patients with mild to moderate psoriasis was actively recruiting at the time of publication.
LEO 80190 (Vitamin D3 Analogue and Steroid Combination)
LEO 80190 (LEO Pharma) is a combination of the vitamin D3 analogue calcipotriol and the corticosteroid hydrocortisone. It was developed as a treatment for sensitive areas such as the face and intertriginous regions. A randomized, investigator-blind, phase 3 trial (NCT00640822) of LEO 80190 ointment versus tacalcitol ointment and placebo once daily for 8 weeks demonstrated controlled disease of the face in 56.8% (183/322) of patients in the LEO 80190 group, 46.4% (147/317) in the tacalcitol group, and 36.3% (37/102) in the placebo group.14 Another phase 2 study (NCT00704262) and 2 phase 3 studies (NCT00691002, NCT01007591) have been completed; results were not available at the time of publication.
LEO 90100 (Vitamin D Analogue and Steroid Combination)
LEO 90100 (LEO Pharma) contains the vitamin D3 analogue calcipotriol and the corticosteroid betamethasone. Three phase 2 studies (NCT01347255, NCT01536886, NCT01536938) and a phase 3 study (NCT01866163) examining the efficacy and safety of various vehicles and formulations of LEO 90100 have been completed; results were not available at the time of publication. Another phase 3 study (NCT02132936) is ongoing but not recruiting participants. Other completed studies whose results were not yet available include a phase 1 pharmacodynamic study (NCT01946386), a phase 1 study that used patch testing to assess the degree of skin irritation and sensitization associated with LEO 90100 (NCT01935869), and a phase 2 study examining the impact of LEO 90100 on calcium metabolism and the hypothalamic-pituitary-adrenal axis (NCT01600222).
M518101 (Vitamin D Analogue)
M518101 (Maruho Co, Ltd) is a novel topical vitamin D3 analogue. Phase 1 (NCT01844973) and phase 2 (NCT01301157, NCT00884169) trials evaluating the safety, pharmacokinetics, and efficacy of M518101 have been completed; results were not available at the time of publication. A phase 3 study (NCT01989429) assessing the safety and therapeutic efficacy of M518101 according to changes in the modified psoriasis area and severity index over an 8-week treatment period also has been completed; results were not yet available. Three phase 3 studies assessing the safety and therapeutic efficacy of M518101 are ongoing: one is currently closed to recruitment (NCT01908595) and 2 are actively recruiting participants at the time of publication (NCT01878461, NCT01873677).
MOL4239 and MOL4249 (Phosphorylated Signal Transducer and Activator of Transcription 3 Inhibitors)
MOL4239 (Moleculin, LLC) is a novel topical agent for use in mild to moderate psoriasis that acts via phosphorylated signal transducer and activator of transcription 3 (p-STAT3) inhibition.15 The p-STAT3 protein has increased expression in psoriasis.16 A phase 2 trial of MOL4239 ointment (NCT01826201) has been completed, showing a greater mean (standard deviation) change in the psoriasis severity score in lesions treated at 28 days with MOL4239 ointment 10% (−1.9 [1.45]) versus lesions treated with placebo ointment (−1.5 [1.87]).17
MOL4249 (Moleculin, LLC) is more potent than MOL4239 with better lipid solubility. In the MOL4249 subset of a placebo-controlled, double-blind, phase 2a study of 16 patients with mild to moderate psoriasis, 10% (1/10) of patients experienced complete clearance of psoriatic plaques, 30% (3/10) of patients experienced 75% or greater improvement, and 50% (5/10) of patients experienced 50% or greater improvement compared to 17% (1/6) in the placebo group. Currently, a phase 2a contralateral study, a phase 2b psoriasis area and severity index trial, and a phase 3 pivotal trial are planned, according to the manufacturer.18
MQX-5902 (Dihydrofolate Reductase Inhibitor)
MQX-5902 (MediQuest Therapeutics) is a topical preparation of methotrexate for the treatment of fingernail psoriasis. Methotrexate is a dihydrofolate reductase inhibitor and antimetabolite that inhibits folic acid metabolism, thereby disrupting DNA synthesis.19 A phase 2b dose-ranging trial (NCT00666354) was designed to assess the therapeutic efficacy and safety of MQX-5902 delivered via a proprietary drug delivery formulation in fingernail psoriasis; the outcome of this trial was not available at the time of publication.
PH-10 (Xanthine Dye)
PH-10 (Provectus Biopharmaceuticals, Inc) is a topical aqueous hydrogel derived from rose bengal disodium that may be beneficial in treating skin conditions such as atopic dermatitis and mild to moderate psoriasis. Rose bengal disodium is a hydrophilic xanthine dye with diagnostic utility in ophthalmology and gastroenterology as well as projected use as a melanoma treatment as demonstrated in phase 1 and phase 2 clinical trials of PV-10 (Provectus Biopharmaceuticals, Inc).20 Two phase 2 studies assessing the safety and therapeutic efficacy of PH-10 in psoriasis (NCT01247818, NCT00941278) have been completed; results were not available at the time of publication.
STF115469 (Vitamin D Analogue)
STF115469 (GlaxoSmithKline) is a calcipotriene foam. At the time of publication, a randomized, placebo-controlled, double-blind, phase 3 trial (NCT01582932) of this vitamin D3 analogue with a projected enrollment of 180 participants was actively recruiting patients aged 2 to 11 years with mild to moderate plaque psoriasis to study the efficacy, safety, and tolerability of STF115469, as well as its pharmacokinetics and pharmacodynamics.
WBI-1001 (Proprietary Product)
WBI-1001 (Welichem Biotech Inc), or 2-isopropyl-5-[(E)-2-phenylethenyl] benzene-1, 3-diol, is a novel proprietary agent that inhibits proinflammatory cytokines (eg, IFN-γ, TNF-α). A randomized, placebo-controlled, double-blind, phase 1 trial (NCT00830817) assessing the efficacy, safety, tolerability, and pharmacokinetics of WBI-1001 has been completed; results were not available at the time of publication. Another randomized, placebo-controlled, double-blind, phase 2 trial (NCT01098721) evaluating its efficacy and safety according to the physician’s global assessment demonstrated a therapeutic benefit of 62.8% in patients treated with WBI-1001 cream 1% versus 13.0% in those treated with a placebo after a 12-week treatment period (P<.0001).21 WBI-1001 may offer a novel approach in the treatment of mild to moderate psoriasis.
Conclusion
Enhanced knowledge of the underlying pathogeneses of psoriasis and psoriatic arthritis has identified new therapeutic targets and enabled the development of exciting novel treatments for these conditions. The topical agents currently in phase 2 and phase 3 clinical trials show promise in enhancing the way physicians treat psoriasis. There is hope for more individualized treatment regimens with improved tolerability and better safety profiles with increased therapeutic efficacy. As our understanding of the molecular underpinnings of psoriasis continues to deepen, it will afford the development of even more innovative therapeutics for use in the management of psoriasis.
1. Mason A, Mason J, Cork M, et al. Topical treatments for chronic plaque psoriasis: an abridged Cochrane systematic review. J Am Acad Dermatol. 2013;69:799-807.
2. Nazarian R, Weinberg JM. AN-2728, a PDE4 inhibitor for the potential topical treatment of psoriasis and atopic dermatitis. Curr Opin Investig Drugs. 2009;10:1236-1242.
3. Moustafa F, Feldman SR. A review of phosphodiesterase-inhibition and the potential role for phosphodiesterase 4-inhibitors in clinical dermatology. Dermatol Online J. 2014;20:22608.
4. Halpert G, Sredni B. The effect of the novel tellurium compound AS101 on autoimmune diseases. Autoimmun Rev. 2014;13:1230-1235.
5. Ports WC, Khan S, Lan S, et al. A randomized phase 2a efficacy and safety trial of the topical Janus kinase inhibitor tofacitinib in the treatment of chronic plaque psoriasis. Br J Dermatol. 2013;169:137-145.
6. Yosipovitch G, Roblin D, Traversa S, et al. A novel topical targeted anti-pruritic treatment in phase 2b development for chronic pruritus. Paper presented at: 72nd Annual Meeting of the American Academy of Dermatology; March 21-25, 2014; Denver, CO.
7. Kang S, Yi S, Griffiths CE, et al. Calcipotriene-induced improvement in psoriasis is associated with reduced interleukin-8 and increased interleukin-10 levels within lesions. Br J Dermatol. 1998;138:77-83.
8. Kristl J, Slanc P, Krasna M, et al. Calcipotriol affects keratinocyte proliferation by decreasing expression of early growth response-1 and polo-like kinase-2. Pharm Res. 2008;25:521-529.
9. Tiberio R, Bozzo C, Pertusi G, et al. Calcipotriol induces apoptosis in psoriatic keratinocytes. Clin Exp Dermatol. 2009;34:e972-e974.
10. Luger T, Seite S, Humbert P, et al. Recommendations for adjunctive basic skin care in patients with psoriasis. Eur J Dermatol. 2014;24:194-200.
11. Dermipsor reports good results in DPS-101 Phase IIb study for plaque psoriasis [press release]. Evaluate Web site. http://www.evaluategroup.com/Universal/View.aspx?type=Story&id=250042. Published October 15, 2007. Accessed February 13, 2015.
12. Rivera AM, Hsu S. Topical halobetasol propionate in the treatment of plaque psoriasis: a review. Am J Clin Dermatol. 2005;6:311-316.
13. Punwani N, Scherle P, Flores R, et al. Preliminary clinical activity of a topical JAK1/2 inhibitor in the treatment of psoriasis. J Am Acad Dermatol. 2012;67:658-664.
14. Efficacy and safety of calcipotriol plus hydrocortisone ointment compared with tacalcitol ointment in patients with psoriasis on the face and skin folds (NCT00640822). https://clinicaltrials.gov/ct2/show/results/NCT00640822?term=NCT00640822&rank=1. Updated October 21, 2013. Accessed May 30, 2014.
15. Product candidates: targeting p-STAT3 for improved psoriasis treatment. Moleculin Web site. http://moleculin.com/product-candidates/mol4239. Accessed February 13, 2015.
16. Chowdhari S, Saini N. hsa-miR-4516 mediated downregulation of STAT3/CDK6/UBE2N plays a role in PUVA induced apoptosis in keratinocytes. J Cell Physiol. 2014;229:1630-1638.
17. Paired psoriasis lesion, comparative, study to evaluate MOL4239 in psoriasis (NCT01826201). https://clinicaltrials.gov/ct2/show/results/NCT01826201?term=NCT01826201&rank=1§=X01256#all. Updated December 22, 2014. Accessed February 25, 2015.
18. Clinical development pipeline. Moleculin Web site. http://moleculin.com/clinical-trials/psoriasis-trials. Accessed February 13, 2015.
19. de la Brassinne M, Nikkels A. Psoriasis: state of the art 2013. Acta Clin Belg. 2013;68:433-441.
20. Ross MI. Intralesional therapy with PV-10 (Rose Bengal) for in-transit melanoma. J Surg Oncol. 2014;109:314-319.
21. Bissonnette R, Bolduc C, Maari C, et al. Efficacy and safety of topical WBI-1001 in patients with mild to moderate psoriasis: results from a randomized, double-blind placebo-controlled, phase II trial. J Eur Acad Dermatol Venereol. 2012;26:1516-1521.
Topical therapies are a mainstay in the management of patients with mild to moderate psoriasis (Figure). Presently, US Food and Drug Administration–approved topical medications that are commercially available for use in patients with psoriasis include corticosteroids, vitamin D3 analogues, calcineurin inhibitors, retinoids, anthralin, and tar-based formulations.1 In recent years, research has furthered our understanding of the molecular mechanisms underlying the pathogenesis of psoriasis and has afforded the development of more targeted therapies. Novel topical medications currently in phase 2 and phase 3 clinical trials are discussed in this article, and a summary is provided in the Table.
AN2728 (Phosphodiesterase 4 Inhibitor)
AN2728 (Anacor Pharmaceuticals, Inc) is a phosphodiesterase 4 inhibitor that blocks the inactivation of cyclic adenosine monophosphate, resulting in decreased production of inflammatory cytokines (eg, IL-6, IL-12, IL-23, tumor necrosis factor α [TNF-α]).2,3 In a randomized, double-blind, phase 2 clinical trial (N=35), 40% of patients treated with AN2728 ointment 5% reported improvement of more than 2 points in overall target plaque severity score versus 6% of patients treated with vehicle. In another randomized, double-blind, dose-response trial of 145 patients, those treated with AN2728 ointment 2% twice daily reported a 60% improvement versus 40% improvement in those treated with AN2728 ointment 0.5% once daily.3 In total, 3 phase 1 trials (registered at www.clinicaltrials.gov with the identifiers NCT01258088, NCT00762658, NCT00763204) and 4 phase 2 trials (NCT01029405, NCT00755196, NCT00759161, NCT01300052) have been completed; results were not available at the time of publication.
AS101 (Integrin Inhibitor)
AS101 (BioMAS Ltd), or ammonium trichloro (dioxoethylene-o,o') tellurate, acts as stimulator of regulatory T cells and a redox modulator inhibiting the leukocyte integrins α4β1 and α4β7 that enable CD4+ T-cell and macrophage extravasation; it also limits expression of the inflammatory cytokines IL-6 and IL-17.4 A randomized, placebo-controlled, double-blind, phase 2 study evaluating the efficacy of AS101 cream 4% twice daily for 12 weeks was withdrawn prior to enrollment (NCT00788424).
Tofacitinib (Janus Kinase 1 and 3 Inhibitor)
Tofacitinib (formerly known as CP-690,550)(Pfizer Inc) is a selective Janus kinase (Jak) 1 and Jak3 inhibitor that limits expression of cytokines that promote inflammation (eg, IFN-γ) and inhibits helper T cells (TH17) by downregulating expression of the IL-23 receptor. Epidermal keratinocyte proliferation in psoriasis is activated by TH17 cells that release IL-17 as well as TH1 cells that release IFN-γ and tumor necrosis factor. A phase 2a trial showed statistically significant improvement from baseline in the target plaque severity score for tofacitinib ointment 2% (least squares mean, −54.4%) versus vehicle (least squares mean, −41.5%).5 Two other phase 2 trials (NCT01246583, NCT00678561) assessing the efficacy, safety, tolerability, and pharmacokinetics of tofacitinib ointment in patients with mild to moderate psoriasis have been completed; results were not available at the time of publication. A phase 2b study that compared 2 dose strengths of tofacitinib ointment—10 mg/g and 20 mg/g—versus placebo over a 12-week period also was completed (NCT01831466); results were not available at the time of publication.
CT327 (Tyrosine Kinase Inhibitor)
CT327 (Creabilis SA) is a tyrosine kinase A (TrkA) inhibitor that affords a novel perspective in the treatment of pruritus by shifting the focus to sensory neurons. In a phase 2b study of 160 patients, a 60% change in the visual analog scale was noted at 8 weeks in the treatment group versus 21% in the placebo group.6 Two other phase 2 studies have been completed, one with a cream formulation of pegylated K252a (NCT00995969) and another with an ointment formulation (NCT01465282); results were not available at the time of publication.
DPS-101 (Vitamin D Analogue)
DPS-101 (Dermipsor Ltd) is a combination of calcipotriol and niacinamide. Calcipotriol is a vitamin D3 analogue that increases IL-10 expression while decreasing IL-8 expression.7 It curbs epidermal keratinocyte proliferation by limiting the expression of polo-like kinase 2 and early growth response-1.8 It also may induce keratinocyte apoptosis.9 Niacinamide is the amide of vitamin B3 and inhibits proinflammatory cytokines such as TNF-α, IL-1β, IL-6, and IL-8.10 In a dose-response phase 2b trial of 168 patients, DPS-101 demonstrated better results than either calcipotriol or niacinamide alone.11
IDP-118 (Proprietary Steroid and Retinoid Combination)
IDP-118 (Valeant Pharmaceuticals International, Inc) is a combination of halobetasol propionate (HP) 0.01% (a topical corticosteroid) and tazar-otene 0.045% (a selective topical retinoid) in a lotion formulation. In isolation, tazarotene is as effective as a mid to highly potent corticosteroid, but irritation may limit its tolerability. The use of combination treatments of mid to highly potent corticosteroids and tazarotene has shown enhanced tolerability and therapeutic efficacy.12 Ongoing studies include a phase 1 trial and a phase 2 trial to evaluate low- and high-strength preparations of IDP-118, respectively (NCT01670513). Another phase 2 trial evaluating the efficacy and safety of IDP-118 lotion (HP 0.01% and tazarotene 0.045%) versus IDP-118 monad HP 0.01% lotion, IDP-118 monad tazar-otene 0.045% lotion, and placebo has been completed (NCT02045277); results were not available at the time of publication.
Ruxolitinib (Jak1 and Jak2 Inhibitor)
Ruxolitinib (formerly known as INCB18424)(Incyte Corporation) is a selective Jak1 and Jak2 inhibitor. A phase 2 trial of ruxolitinib showed a 53% decline in the score for mean total lesions in patients treated with ruxolitinib phosphate cream 1% (P=.033) versus 54% in those treated with ruxolitinib phosphate cream 1.5% (P=.056) and 32% in those treated with placebo.13 Three other phase 2 studies (NCT00617994, NCT00820950, NCT00778700) have been completed; results were not available at the time of publication.
LAS41004 (Proprietary Steroid and Retinoid Combination)
LAS41004 (Almirall, SA) is an ointment containing the corticosteroid betamethasone dipropionate and the retinoid bexarotene that is being evaluated for treatment of mild to moderate psoriasis. Five phase 2 studies (NCT01119339, NCT01283698, NCT01360944, NCT02111499, NCT01462643) have been completed; results were not available at the time of publication. A randomized, double-blind, phase 2a study (NCT02180464) with a left-right design assessing clinical response to LAS41004 versus control in patients with mild to moderate psoriasis was actively recruiting at the time of publication.
LEO 80190 (Vitamin D3 Analogue and Steroid Combination)
LEO 80190 (LEO Pharma) is a combination of the vitamin D3 analogue calcipotriol and the corticosteroid hydrocortisone. It was developed as a treatment for sensitive areas such as the face and intertriginous regions. A randomized, investigator-blind, phase 3 trial (NCT00640822) of LEO 80190 ointment versus tacalcitol ointment and placebo once daily for 8 weeks demonstrated controlled disease of the face in 56.8% (183/322) of patients in the LEO 80190 group, 46.4% (147/317) in the tacalcitol group, and 36.3% (37/102) in the placebo group.14 Another phase 2 study (NCT00704262) and 2 phase 3 studies (NCT00691002, NCT01007591) have been completed; results were not available at the time of publication.
LEO 90100 (Vitamin D Analogue and Steroid Combination)
LEO 90100 (LEO Pharma) contains the vitamin D3 analogue calcipotriol and the corticosteroid betamethasone. Three phase 2 studies (NCT01347255, NCT01536886, NCT01536938) and a phase 3 study (NCT01866163) examining the efficacy and safety of various vehicles and formulations of LEO 90100 have been completed; results were not available at the time of publication. Another phase 3 study (NCT02132936) is ongoing but not recruiting participants. Other completed studies whose results were not yet available include a phase 1 pharmacodynamic study (NCT01946386), a phase 1 study that used patch testing to assess the degree of skin irritation and sensitization associated with LEO 90100 (NCT01935869), and a phase 2 study examining the impact of LEO 90100 on calcium metabolism and the hypothalamic-pituitary-adrenal axis (NCT01600222).
M518101 (Vitamin D Analogue)
M518101 (Maruho Co, Ltd) is a novel topical vitamin D3 analogue. Phase 1 (NCT01844973) and phase 2 (NCT01301157, NCT00884169) trials evaluating the safety, pharmacokinetics, and efficacy of M518101 have been completed; results were not available at the time of publication. A phase 3 study (NCT01989429) assessing the safety and therapeutic efficacy of M518101 according to changes in the modified psoriasis area and severity index over an 8-week treatment period also has been completed; results were not yet available. Three phase 3 studies assessing the safety and therapeutic efficacy of M518101 are ongoing: one is currently closed to recruitment (NCT01908595) and 2 are actively recruiting participants at the time of publication (NCT01878461, NCT01873677).
MOL4239 and MOL4249 (Phosphorylated Signal Transducer and Activator of Transcription 3 Inhibitors)
MOL4239 (Moleculin, LLC) is a novel topical agent for use in mild to moderate psoriasis that acts via phosphorylated signal transducer and activator of transcription 3 (p-STAT3) inhibition.15 The p-STAT3 protein has increased expression in psoriasis.16 A phase 2 trial of MOL4239 ointment (NCT01826201) has been completed, showing a greater mean (standard deviation) change in the psoriasis severity score in lesions treated at 28 days with MOL4239 ointment 10% (−1.9 [1.45]) versus lesions treated with placebo ointment (−1.5 [1.87]).17
MOL4249 (Moleculin, LLC) is more potent than MOL4239 with better lipid solubility. In the MOL4249 subset of a placebo-controlled, double-blind, phase 2a study of 16 patients with mild to moderate psoriasis, 10% (1/10) of patients experienced complete clearance of psoriatic plaques, 30% (3/10) of patients experienced 75% or greater improvement, and 50% (5/10) of patients experienced 50% or greater improvement compared to 17% (1/6) in the placebo group. Currently, a phase 2a contralateral study, a phase 2b psoriasis area and severity index trial, and a phase 3 pivotal trial are planned, according to the manufacturer.18
MQX-5902 (Dihydrofolate Reductase Inhibitor)
MQX-5902 (MediQuest Therapeutics) is a topical preparation of methotrexate for the treatment of fingernail psoriasis. Methotrexate is a dihydrofolate reductase inhibitor and antimetabolite that inhibits folic acid metabolism, thereby disrupting DNA synthesis.19 A phase 2b dose-ranging trial (NCT00666354) was designed to assess the therapeutic efficacy and safety of MQX-5902 delivered via a proprietary drug delivery formulation in fingernail psoriasis; the outcome of this trial was not available at the time of publication.
PH-10 (Xanthine Dye)
PH-10 (Provectus Biopharmaceuticals, Inc) is a topical aqueous hydrogel derived from rose bengal disodium that may be beneficial in treating skin conditions such as atopic dermatitis and mild to moderate psoriasis. Rose bengal disodium is a hydrophilic xanthine dye with diagnostic utility in ophthalmology and gastroenterology as well as projected use as a melanoma treatment as demonstrated in phase 1 and phase 2 clinical trials of PV-10 (Provectus Biopharmaceuticals, Inc).20 Two phase 2 studies assessing the safety and therapeutic efficacy of PH-10 in psoriasis (NCT01247818, NCT00941278) have been completed; results were not available at the time of publication.
STF115469 (Vitamin D Analogue)
STF115469 (GlaxoSmithKline) is a calcipotriene foam. At the time of publication, a randomized, placebo-controlled, double-blind, phase 3 trial (NCT01582932) of this vitamin D3 analogue with a projected enrollment of 180 participants was actively recruiting patients aged 2 to 11 years with mild to moderate plaque psoriasis to study the efficacy, safety, and tolerability of STF115469, as well as its pharmacokinetics and pharmacodynamics.
WBI-1001 (Proprietary Product)
WBI-1001 (Welichem Biotech Inc), or 2-isopropyl-5-[(E)-2-phenylethenyl] benzene-1, 3-diol, is a novel proprietary agent that inhibits proinflammatory cytokines (eg, IFN-γ, TNF-α). A randomized, placebo-controlled, double-blind, phase 1 trial (NCT00830817) assessing the efficacy, safety, tolerability, and pharmacokinetics of WBI-1001 has been completed; results were not available at the time of publication. Another randomized, placebo-controlled, double-blind, phase 2 trial (NCT01098721) evaluating its efficacy and safety according to the physician’s global assessment demonstrated a therapeutic benefit of 62.8% in patients treated with WBI-1001 cream 1% versus 13.0% in those treated with a placebo after a 12-week treatment period (P<.0001).21 WBI-1001 may offer a novel approach in the treatment of mild to moderate psoriasis.
Conclusion
Enhanced knowledge of the underlying pathogeneses of psoriasis and psoriatic arthritis has identified new therapeutic targets and enabled the development of exciting novel treatments for these conditions. The topical agents currently in phase 2 and phase 3 clinical trials show promise in enhancing the way physicians treat psoriasis. There is hope for more individualized treatment regimens with improved tolerability and better safety profiles with increased therapeutic efficacy. As our understanding of the molecular underpinnings of psoriasis continues to deepen, it will afford the development of even more innovative therapeutics for use in the management of psoriasis.
Topical therapies are a mainstay in the management of patients with mild to moderate psoriasis (Figure). Presently, US Food and Drug Administration–approved topical medications that are commercially available for use in patients with psoriasis include corticosteroids, vitamin D3 analogues, calcineurin inhibitors, retinoids, anthralin, and tar-based formulations.1 In recent years, research has furthered our understanding of the molecular mechanisms underlying the pathogenesis of psoriasis and has afforded the development of more targeted therapies. Novel topical medications currently in phase 2 and phase 3 clinical trials are discussed in this article, and a summary is provided in the Table.
AN2728 (Phosphodiesterase 4 Inhibitor)
AN2728 (Anacor Pharmaceuticals, Inc) is a phosphodiesterase 4 inhibitor that blocks the inactivation of cyclic adenosine monophosphate, resulting in decreased production of inflammatory cytokines (eg, IL-6, IL-12, IL-23, tumor necrosis factor α [TNF-α]).2,3 In a randomized, double-blind, phase 2 clinical trial (N=35), 40% of patients treated with AN2728 ointment 5% reported improvement of more than 2 points in overall target plaque severity score versus 6% of patients treated with vehicle. In another randomized, double-blind, dose-response trial of 145 patients, those treated with AN2728 ointment 2% twice daily reported a 60% improvement versus 40% improvement in those treated with AN2728 ointment 0.5% once daily.3 In total, 3 phase 1 trials (registered at www.clinicaltrials.gov with the identifiers NCT01258088, NCT00762658, NCT00763204) and 4 phase 2 trials (NCT01029405, NCT00755196, NCT00759161, NCT01300052) have been completed; results were not available at the time of publication.
AS101 (Integrin Inhibitor)
AS101 (BioMAS Ltd), or ammonium trichloro (dioxoethylene-o,o') tellurate, acts as stimulator of regulatory T cells and a redox modulator inhibiting the leukocyte integrins α4β1 and α4β7 that enable CD4+ T-cell and macrophage extravasation; it also limits expression of the inflammatory cytokines IL-6 and IL-17.4 A randomized, placebo-controlled, double-blind, phase 2 study evaluating the efficacy of AS101 cream 4% twice daily for 12 weeks was withdrawn prior to enrollment (NCT00788424).
Tofacitinib (Janus Kinase 1 and 3 Inhibitor)
Tofacitinib (formerly known as CP-690,550)(Pfizer Inc) is a selective Janus kinase (Jak) 1 and Jak3 inhibitor that limits expression of cytokines that promote inflammation (eg, IFN-γ) and inhibits helper T cells (TH17) by downregulating expression of the IL-23 receptor. Epidermal keratinocyte proliferation in psoriasis is activated by TH17 cells that release IL-17 as well as TH1 cells that release IFN-γ and tumor necrosis factor. A phase 2a trial showed statistically significant improvement from baseline in the target plaque severity score for tofacitinib ointment 2% (least squares mean, −54.4%) versus vehicle (least squares mean, −41.5%).5 Two other phase 2 trials (NCT01246583, NCT00678561) assessing the efficacy, safety, tolerability, and pharmacokinetics of tofacitinib ointment in patients with mild to moderate psoriasis have been completed; results were not available at the time of publication. A phase 2b study that compared 2 dose strengths of tofacitinib ointment—10 mg/g and 20 mg/g—versus placebo over a 12-week period also was completed (NCT01831466); results were not available at the time of publication.
CT327 (Tyrosine Kinase Inhibitor)
CT327 (Creabilis SA) is a tyrosine kinase A (TrkA) inhibitor that affords a novel perspective in the treatment of pruritus by shifting the focus to sensory neurons. In a phase 2b study of 160 patients, a 60% change in the visual analog scale was noted at 8 weeks in the treatment group versus 21% in the placebo group.6 Two other phase 2 studies have been completed, one with a cream formulation of pegylated K252a (NCT00995969) and another with an ointment formulation (NCT01465282); results were not available at the time of publication.
DPS-101 (Vitamin D Analogue)
DPS-101 (Dermipsor Ltd) is a combination of calcipotriol and niacinamide. Calcipotriol is a vitamin D3 analogue that increases IL-10 expression while decreasing IL-8 expression.7 It curbs epidermal keratinocyte proliferation by limiting the expression of polo-like kinase 2 and early growth response-1.8 It also may induce keratinocyte apoptosis.9 Niacinamide is the amide of vitamin B3 and inhibits proinflammatory cytokines such as TNF-α, IL-1β, IL-6, and IL-8.10 In a dose-response phase 2b trial of 168 patients, DPS-101 demonstrated better results than either calcipotriol or niacinamide alone.11
IDP-118 (Proprietary Steroid and Retinoid Combination)
IDP-118 (Valeant Pharmaceuticals International, Inc) is a combination of halobetasol propionate (HP) 0.01% (a topical corticosteroid) and tazar-otene 0.045% (a selective topical retinoid) in a lotion formulation. In isolation, tazarotene is as effective as a mid to highly potent corticosteroid, but irritation may limit its tolerability. The use of combination treatments of mid to highly potent corticosteroids and tazarotene has shown enhanced tolerability and therapeutic efficacy.12 Ongoing studies include a phase 1 trial and a phase 2 trial to evaluate low- and high-strength preparations of IDP-118, respectively (NCT01670513). Another phase 2 trial evaluating the efficacy and safety of IDP-118 lotion (HP 0.01% and tazarotene 0.045%) versus IDP-118 monad HP 0.01% lotion, IDP-118 monad tazar-otene 0.045% lotion, and placebo has been completed (NCT02045277); results were not available at the time of publication.
Ruxolitinib (Jak1 and Jak2 Inhibitor)
Ruxolitinib (formerly known as INCB18424)(Incyte Corporation) is a selective Jak1 and Jak2 inhibitor. A phase 2 trial of ruxolitinib showed a 53% decline in the score for mean total lesions in patients treated with ruxolitinib phosphate cream 1% (P=.033) versus 54% in those treated with ruxolitinib phosphate cream 1.5% (P=.056) and 32% in those treated with placebo.13 Three other phase 2 studies (NCT00617994, NCT00820950, NCT00778700) have been completed; results were not available at the time of publication.
LAS41004 (Proprietary Steroid and Retinoid Combination)
LAS41004 (Almirall, SA) is an ointment containing the corticosteroid betamethasone dipropionate and the retinoid bexarotene that is being evaluated for treatment of mild to moderate psoriasis. Five phase 2 studies (NCT01119339, NCT01283698, NCT01360944, NCT02111499, NCT01462643) have been completed; results were not available at the time of publication. A randomized, double-blind, phase 2a study (NCT02180464) with a left-right design assessing clinical response to LAS41004 versus control in patients with mild to moderate psoriasis was actively recruiting at the time of publication.
LEO 80190 (Vitamin D3 Analogue and Steroid Combination)
LEO 80190 (LEO Pharma) is a combination of the vitamin D3 analogue calcipotriol and the corticosteroid hydrocortisone. It was developed as a treatment for sensitive areas such as the face and intertriginous regions. A randomized, investigator-blind, phase 3 trial (NCT00640822) of LEO 80190 ointment versus tacalcitol ointment and placebo once daily for 8 weeks demonstrated controlled disease of the face in 56.8% (183/322) of patients in the LEO 80190 group, 46.4% (147/317) in the tacalcitol group, and 36.3% (37/102) in the placebo group.14 Another phase 2 study (NCT00704262) and 2 phase 3 studies (NCT00691002, NCT01007591) have been completed; results were not available at the time of publication.
LEO 90100 (Vitamin D Analogue and Steroid Combination)
LEO 90100 (LEO Pharma) contains the vitamin D3 analogue calcipotriol and the corticosteroid betamethasone. Three phase 2 studies (NCT01347255, NCT01536886, NCT01536938) and a phase 3 study (NCT01866163) examining the efficacy and safety of various vehicles and formulations of LEO 90100 have been completed; results were not available at the time of publication. Another phase 3 study (NCT02132936) is ongoing but not recruiting participants. Other completed studies whose results were not yet available include a phase 1 pharmacodynamic study (NCT01946386), a phase 1 study that used patch testing to assess the degree of skin irritation and sensitization associated with LEO 90100 (NCT01935869), and a phase 2 study examining the impact of LEO 90100 on calcium metabolism and the hypothalamic-pituitary-adrenal axis (NCT01600222).
M518101 (Vitamin D Analogue)
M518101 (Maruho Co, Ltd) is a novel topical vitamin D3 analogue. Phase 1 (NCT01844973) and phase 2 (NCT01301157, NCT00884169) trials evaluating the safety, pharmacokinetics, and efficacy of M518101 have been completed; results were not available at the time of publication. A phase 3 study (NCT01989429) assessing the safety and therapeutic efficacy of M518101 according to changes in the modified psoriasis area and severity index over an 8-week treatment period also has been completed; results were not yet available. Three phase 3 studies assessing the safety and therapeutic efficacy of M518101 are ongoing: one is currently closed to recruitment (NCT01908595) and 2 are actively recruiting participants at the time of publication (NCT01878461, NCT01873677).
MOL4239 and MOL4249 (Phosphorylated Signal Transducer and Activator of Transcription 3 Inhibitors)
MOL4239 (Moleculin, LLC) is a novel topical agent for use in mild to moderate psoriasis that acts via phosphorylated signal transducer and activator of transcription 3 (p-STAT3) inhibition.15 The p-STAT3 protein has increased expression in psoriasis.16 A phase 2 trial of MOL4239 ointment (NCT01826201) has been completed, showing a greater mean (standard deviation) change in the psoriasis severity score in lesions treated at 28 days with MOL4239 ointment 10% (−1.9 [1.45]) versus lesions treated with placebo ointment (−1.5 [1.87]).17
MOL4249 (Moleculin, LLC) is more potent than MOL4239 with better lipid solubility. In the MOL4249 subset of a placebo-controlled, double-blind, phase 2a study of 16 patients with mild to moderate psoriasis, 10% (1/10) of patients experienced complete clearance of psoriatic plaques, 30% (3/10) of patients experienced 75% or greater improvement, and 50% (5/10) of patients experienced 50% or greater improvement compared to 17% (1/6) in the placebo group. Currently, a phase 2a contralateral study, a phase 2b psoriasis area and severity index trial, and a phase 3 pivotal trial are planned, according to the manufacturer.18
MQX-5902 (Dihydrofolate Reductase Inhibitor)
MQX-5902 (MediQuest Therapeutics) is a topical preparation of methotrexate for the treatment of fingernail psoriasis. Methotrexate is a dihydrofolate reductase inhibitor and antimetabolite that inhibits folic acid metabolism, thereby disrupting DNA synthesis.19 A phase 2b dose-ranging trial (NCT00666354) was designed to assess the therapeutic efficacy and safety of MQX-5902 delivered via a proprietary drug delivery formulation in fingernail psoriasis; the outcome of this trial was not available at the time of publication.
PH-10 (Xanthine Dye)
PH-10 (Provectus Biopharmaceuticals, Inc) is a topical aqueous hydrogel derived from rose bengal disodium that may be beneficial in treating skin conditions such as atopic dermatitis and mild to moderate psoriasis. Rose bengal disodium is a hydrophilic xanthine dye with diagnostic utility in ophthalmology and gastroenterology as well as projected use as a melanoma treatment as demonstrated in phase 1 and phase 2 clinical trials of PV-10 (Provectus Biopharmaceuticals, Inc).20 Two phase 2 studies assessing the safety and therapeutic efficacy of PH-10 in psoriasis (NCT01247818, NCT00941278) have been completed; results were not available at the time of publication.
STF115469 (Vitamin D Analogue)
STF115469 (GlaxoSmithKline) is a calcipotriene foam. At the time of publication, a randomized, placebo-controlled, double-blind, phase 3 trial (NCT01582932) of this vitamin D3 analogue with a projected enrollment of 180 participants was actively recruiting patients aged 2 to 11 years with mild to moderate plaque psoriasis to study the efficacy, safety, and tolerability of STF115469, as well as its pharmacokinetics and pharmacodynamics.
WBI-1001 (Proprietary Product)
WBI-1001 (Welichem Biotech Inc), or 2-isopropyl-5-[(E)-2-phenylethenyl] benzene-1, 3-diol, is a novel proprietary agent that inhibits proinflammatory cytokines (eg, IFN-γ, TNF-α). A randomized, placebo-controlled, double-blind, phase 1 trial (NCT00830817) assessing the efficacy, safety, tolerability, and pharmacokinetics of WBI-1001 has been completed; results were not available at the time of publication. Another randomized, placebo-controlled, double-blind, phase 2 trial (NCT01098721) evaluating its efficacy and safety according to the physician’s global assessment demonstrated a therapeutic benefit of 62.8% in patients treated with WBI-1001 cream 1% versus 13.0% in those treated with a placebo after a 12-week treatment period (P<.0001).21 WBI-1001 may offer a novel approach in the treatment of mild to moderate psoriasis.
Conclusion
Enhanced knowledge of the underlying pathogeneses of psoriasis and psoriatic arthritis has identified new therapeutic targets and enabled the development of exciting novel treatments for these conditions. The topical agents currently in phase 2 and phase 3 clinical trials show promise in enhancing the way physicians treat psoriasis. There is hope for more individualized treatment regimens with improved tolerability and better safety profiles with increased therapeutic efficacy. As our understanding of the molecular underpinnings of psoriasis continues to deepen, it will afford the development of even more innovative therapeutics for use in the management of psoriasis.
1. Mason A, Mason J, Cork M, et al. Topical treatments for chronic plaque psoriasis: an abridged Cochrane systematic review. J Am Acad Dermatol. 2013;69:799-807.
2. Nazarian R, Weinberg JM. AN-2728, a PDE4 inhibitor for the potential topical treatment of psoriasis and atopic dermatitis. Curr Opin Investig Drugs. 2009;10:1236-1242.
3. Moustafa F, Feldman SR. A review of phosphodiesterase-inhibition and the potential role for phosphodiesterase 4-inhibitors in clinical dermatology. Dermatol Online J. 2014;20:22608.
4. Halpert G, Sredni B. The effect of the novel tellurium compound AS101 on autoimmune diseases. Autoimmun Rev. 2014;13:1230-1235.
5. Ports WC, Khan S, Lan S, et al. A randomized phase 2a efficacy and safety trial of the topical Janus kinase inhibitor tofacitinib in the treatment of chronic plaque psoriasis. Br J Dermatol. 2013;169:137-145.
6. Yosipovitch G, Roblin D, Traversa S, et al. A novel topical targeted anti-pruritic treatment in phase 2b development for chronic pruritus. Paper presented at: 72nd Annual Meeting of the American Academy of Dermatology; March 21-25, 2014; Denver, CO.
7. Kang S, Yi S, Griffiths CE, et al. Calcipotriene-induced improvement in psoriasis is associated with reduced interleukin-8 and increased interleukin-10 levels within lesions. Br J Dermatol. 1998;138:77-83.
8. Kristl J, Slanc P, Krasna M, et al. Calcipotriol affects keratinocyte proliferation by decreasing expression of early growth response-1 and polo-like kinase-2. Pharm Res. 2008;25:521-529.
9. Tiberio R, Bozzo C, Pertusi G, et al. Calcipotriol induces apoptosis in psoriatic keratinocytes. Clin Exp Dermatol. 2009;34:e972-e974.
10. Luger T, Seite S, Humbert P, et al. Recommendations for adjunctive basic skin care in patients with psoriasis. Eur J Dermatol. 2014;24:194-200.
11. Dermipsor reports good results in DPS-101 Phase IIb study for plaque psoriasis [press release]. Evaluate Web site. http://www.evaluategroup.com/Universal/View.aspx?type=Story&id=250042. Published October 15, 2007. Accessed February 13, 2015.
12. Rivera AM, Hsu S. Topical halobetasol propionate in the treatment of plaque psoriasis: a review. Am J Clin Dermatol. 2005;6:311-316.
13. Punwani N, Scherle P, Flores R, et al. Preliminary clinical activity of a topical JAK1/2 inhibitor in the treatment of psoriasis. J Am Acad Dermatol. 2012;67:658-664.
14. Efficacy and safety of calcipotriol plus hydrocortisone ointment compared with tacalcitol ointment in patients with psoriasis on the face and skin folds (NCT00640822). https://clinicaltrials.gov/ct2/show/results/NCT00640822?term=NCT00640822&rank=1. Updated October 21, 2013. Accessed May 30, 2014.
15. Product candidates: targeting p-STAT3 for improved psoriasis treatment. Moleculin Web site. http://moleculin.com/product-candidates/mol4239. Accessed February 13, 2015.
16. Chowdhari S, Saini N. hsa-miR-4516 mediated downregulation of STAT3/CDK6/UBE2N plays a role in PUVA induced apoptosis in keratinocytes. J Cell Physiol. 2014;229:1630-1638.
17. Paired psoriasis lesion, comparative, study to evaluate MOL4239 in psoriasis (NCT01826201). https://clinicaltrials.gov/ct2/show/results/NCT01826201?term=NCT01826201&rank=1§=X01256#all. Updated December 22, 2014. Accessed February 25, 2015.
18. Clinical development pipeline. Moleculin Web site. http://moleculin.com/clinical-trials/psoriasis-trials. Accessed February 13, 2015.
19. de la Brassinne M, Nikkels A. Psoriasis: state of the art 2013. Acta Clin Belg. 2013;68:433-441.
20. Ross MI. Intralesional therapy with PV-10 (Rose Bengal) for in-transit melanoma. J Surg Oncol. 2014;109:314-319.
21. Bissonnette R, Bolduc C, Maari C, et al. Efficacy and safety of topical WBI-1001 in patients with mild to moderate psoriasis: results from a randomized, double-blind placebo-controlled, phase II trial. J Eur Acad Dermatol Venereol. 2012;26:1516-1521.
1. Mason A, Mason J, Cork M, et al. Topical treatments for chronic plaque psoriasis: an abridged Cochrane systematic review. J Am Acad Dermatol. 2013;69:799-807.
2. Nazarian R, Weinberg JM. AN-2728, a PDE4 inhibitor for the potential topical treatment of psoriasis and atopic dermatitis. Curr Opin Investig Drugs. 2009;10:1236-1242.
3. Moustafa F, Feldman SR. A review of phosphodiesterase-inhibition and the potential role for phosphodiesterase 4-inhibitors in clinical dermatology. Dermatol Online J. 2014;20:22608.
4. Halpert G, Sredni B. The effect of the novel tellurium compound AS101 on autoimmune diseases. Autoimmun Rev. 2014;13:1230-1235.
5. Ports WC, Khan S, Lan S, et al. A randomized phase 2a efficacy and safety trial of the topical Janus kinase inhibitor tofacitinib in the treatment of chronic plaque psoriasis. Br J Dermatol. 2013;169:137-145.
6. Yosipovitch G, Roblin D, Traversa S, et al. A novel topical targeted anti-pruritic treatment in phase 2b development for chronic pruritus. Paper presented at: 72nd Annual Meeting of the American Academy of Dermatology; March 21-25, 2014; Denver, CO.
7. Kang S, Yi S, Griffiths CE, et al. Calcipotriene-induced improvement in psoriasis is associated with reduced interleukin-8 and increased interleukin-10 levels within lesions. Br J Dermatol. 1998;138:77-83.
8. Kristl J, Slanc P, Krasna M, et al. Calcipotriol affects keratinocyte proliferation by decreasing expression of early growth response-1 and polo-like kinase-2. Pharm Res. 2008;25:521-529.
9. Tiberio R, Bozzo C, Pertusi G, et al. Calcipotriol induces apoptosis in psoriatic keratinocytes. Clin Exp Dermatol. 2009;34:e972-e974.
10. Luger T, Seite S, Humbert P, et al. Recommendations for adjunctive basic skin care in patients with psoriasis. Eur J Dermatol. 2014;24:194-200.
11. Dermipsor reports good results in DPS-101 Phase IIb study for plaque psoriasis [press release]. Evaluate Web site. http://www.evaluategroup.com/Universal/View.aspx?type=Story&id=250042. Published October 15, 2007. Accessed February 13, 2015.
12. Rivera AM, Hsu S. Topical halobetasol propionate in the treatment of plaque psoriasis: a review. Am J Clin Dermatol. 2005;6:311-316.
13. Punwani N, Scherle P, Flores R, et al. Preliminary clinical activity of a topical JAK1/2 inhibitor in the treatment of psoriasis. J Am Acad Dermatol. 2012;67:658-664.
14. Efficacy and safety of calcipotriol plus hydrocortisone ointment compared with tacalcitol ointment in patients with psoriasis on the face and skin folds (NCT00640822). https://clinicaltrials.gov/ct2/show/results/NCT00640822?term=NCT00640822&rank=1. Updated October 21, 2013. Accessed May 30, 2014.
15. Product candidates: targeting p-STAT3 for improved psoriasis treatment. Moleculin Web site. http://moleculin.com/product-candidates/mol4239. Accessed February 13, 2015.
16. Chowdhari S, Saini N. hsa-miR-4516 mediated downregulation of STAT3/CDK6/UBE2N plays a role in PUVA induced apoptosis in keratinocytes. J Cell Physiol. 2014;229:1630-1638.
17. Paired psoriasis lesion, comparative, study to evaluate MOL4239 in psoriasis (NCT01826201). https://clinicaltrials.gov/ct2/show/results/NCT01826201?term=NCT01826201&rank=1§=X01256#all. Updated December 22, 2014. Accessed February 25, 2015.
18. Clinical development pipeline. Moleculin Web site. http://moleculin.com/clinical-trials/psoriasis-trials. Accessed February 13, 2015.
19. de la Brassinne M, Nikkels A. Psoriasis: state of the art 2013. Acta Clin Belg. 2013;68:433-441.
20. Ross MI. Intralesional therapy with PV-10 (Rose Bengal) for in-transit melanoma. J Surg Oncol. 2014;109:314-319.
21. Bissonnette R, Bolduc C, Maari C, et al. Efficacy and safety of topical WBI-1001 in patients with mild to moderate psoriasis: results from a randomized, double-blind placebo-controlled, phase II trial. J Eur Acad Dermatol Venereol. 2012;26:1516-1521.
Practice Points
- Topical therapies are the cornerstone of treating patients with mild to moderate psoriasis. Commercially available medications approved by the US Food and Drug Administration for use in patients with psoriasis include corticosteroids, vitamin D3 analogues, calcineurin inhibitors, retinoids, anthralin, and tar-based formulations.
- Recent developments in our understanding of inflammatory mediators and the underlying pathogenesis of psoriasis have revealed new potential therapeutic targets, leading to the discovery of many promising treatments.
- Novel topical therapies currently in phase 2 and phase 3 clinical trials for patients with mild to moderate psoriasis may offer hope to patients who have reported a suboptimal therapeutic response to conventional treatments.
New Systemic Therapies for Psoriasis
Psoriasis is a common chronic inflammatory skin disease affecting 1% to 8% of the world population, depending on the country.1 Psoriasis can greatly impact quality of life in affected individuals, even in those with limited body surface involvement.2 Studies have demonstrated a high degree of psychological distress associated with psoriasis, leading to depression and poor self-esteem.3
Over the last decade, our improved understanding of the autoimmune inflammatory pathways and the associated changing concepts in psoriasis pathogenesis have led to the development of biological drugs targeting specific components of effector immune mechanisms, and these biological drugs have revolutionized the treatment of psoriasis.4 Although response rates of these biological agents are greater compared to those of conventional systemic drugs,5 current biological drugs fail to demonstrate efficacy in some patients or lose their efficacy over time. In addition to the high costs associated with these drugs, these limitations have driven a continued search for alternative therapies.
Helper T cells (TH17) and the proinflammatory cytokine IL-17 have been shown to play a key role in the pathophysiology of psoriasis, bridging innate and adaptive immune responses. IL-17 is involved in the modulation of proinflammatory cytokines, hematopoietic growth factors, antimicrobial peptides, and chemokines. Increased TH17 activity and high levels of IL-17 have been found in psoriatic plaques, and increased levels of TH17 are found in the plasma of psoriasis patients.6 Increased IL-17 induces neutrophilia, inflammation, and angiogenesis.7 Other cytokines that are highly upregulated in involved skin are tumor necrosis factor a (TNF-α), IL-23, IL-22, and IL-21.8 IL-23 is involved in regulating TH17 cells and is a potent activator of keratinocyte proliferation.9 Blockade of IL-12/23 causes downregulation of TH17 and TH22 cell responses.10 As IL-17 has a key role in protecting skin and mucous membranes from bacterial and fungal infections, IL-17 inhibition can potentially interfere with the inflammatory cascade. However, available data suggest that sufficient residual IL-17 activity remains to maintain immunity against infections.11
Currently approved biological agents for psoriasis target proinflammatory cytokines such as TNF-α, or the p40 subunit of IL-12 and IL-23. A number of novel targeted therapies including biologics as well as small molecule inhibitors targeting various cytokines and molecules involved in the pathogenesis of psoriasis are currently in different stages of development (Table). These drugs include 3 IL-17 inhibitors (secukinumab, ixekizumab, and brodalumab); 2 IL-23 blockers (tildrakizumab and guselkumab); and small molecule inhibitors that target the kinase pathway including apremilast (a phosphodiesterase 4 [PDE4] inhibitor), as well as tofacitinib, baricitinib, and ruxolitinib (Janus kinase [Jak] inhibitors). Small molecule inhibitors can be administered orally and are less expensive to produce than biological agents. This article reviews available data on these new systemic agents in the pipeline.
Novel Biologics
Secukinumab
Secukinumab is a fully human monoclonal IgG1k antibody that selectively binds and neutralizes IL-17A.12 It is the first of the IL-17 antibodies to receive approval for the treatment of moderate to severe psoriasis. In 2 phase 3, double-blind, 52-week trials—ERASURE (Efficacy of Response and Safety of Two Fixed Secukinumab Regimens in Psoriasis) and FIXTURE (Full Year Investigative Examination of Secukinumab vs Etanercept Using Two Dosing Regimens to Determine Efficacy in Psoriasis)—participants were randomly assigned to receive subcutaneous secukinumab at doses of 300 mg (n=245 and n=327, respectively) or 150 mg (n=245 and n=327, respectively) once weekly for 5 weeks then every 4 weeks, or placebo (n=248 and n=326, respectively); in the FIXTURE study only, an etanercept group (n=326) was given a 50-mg dose twice weekly for 12 weeks then once weekly.13
In the ERASURE study, the proportion of participants showing a reduction of 75% or more in psoriasis area and severity index (PASI) score from baseline to week 12 was 81.6% with secukinumab 300 mg, 71.6% with secukinumab 150 mg, and 4.5% with placebo.13 Secondary end point results demonstrated the proportion of participants showing a 90% reduction in PASI score was 59.2% with secukinumab 300 mg and 39.1% with secukinumab 150 mg, which were both superior to placebo (1.2%). The proportion of participants who met the criteria for 100% reduction in PASI score at week 12 also was greater with each secukinumab dose than with placebo.13
In the FIXTURE study, the proportion of participants showing a reduction of 75% or more from baseline in PASI score at week 12 was 77.1% with secukinumab 300 mg, 67.0% with secukinumab 150 mg, 44.0% with etanercept, and 4.9% with placebo.13 Secondary end point results demonstrated the proportion of participants showing a 90% reduction in PASI score was 54.2% with secukinumab 300 mg, 41.9% with secukinumab 150 mg, 20.7% with etanercept, and 1.5% with placebo. The speed of response, which was assessed as the median time to a 50% reduction in mean PASI score from baseline, was significantly shorter with both doses of secukinumab (3.0 weeks and 3.9 weeks, respectively) than with etanercept (7.0 weeks)(P<.001 for both).13
In the FIXTURE study, incidences of adverse events (AEs) were similar in the secukinumab and etanercept groups during both the induction period and the entire treatment period.13 The most common AEs in the secukinumab groups were nasopharyngitis, headache, and diarrhea. The rates of infections or infestations during the induction period were 26.7% with secukinumab 300 mg, 30.9% with secukinumab 150 mg, 24.5% with etanercept, and 19.3% with placebo. Candidal infections were more common with secukinumab than with etanercept during the entire treatment period (4.7% and 2.3% of participants in the secukinumab 300 mg and 150 mg groups, respectively, reported mild or moderate candidal infections). None of these infections resulted in chronic mucocutaneous candidiasis or discontinuation of the study drug and all resolved on their own or with standard therapy. Candidal infection was reported in 1.2% of participants in the etanercept group. Responses at week 12 were sustained in the majority of participants through week 52 with continued secukinumab therapy every 4 weeks. Grade 3 neutropenia occurred in 1.0% of secukinumab-treated participants and in none of the participants in the etanercept group. There were no apparent dose-related differences between the secukinumab groups with respect to AEs, with the exception of mild and moderate candidal infections.13
These efficacy data are impressive and no specific serious safety concerns have been identified to date. However, IL-17A plays a key role in host defense, particularly in mucocutaneous immunity against Candida albicans,14 as well as in hematopoiesis through stimulation of granulopoiesis and neutrophil trafficking,15 and thus we need to remain watchful with regards to Candida albicans infections and neutropenia.
Ixekizumab
Ixekizumab is a humanized IgG4 anti–IL-17A monoclonal antibody. In a phase 2, double-blind, placebo-controlled trial, 142 participants with chronic moderate to severe plaque psoriasis were randomly assigned to receive 150-mg (n=28), 75-mg (n=29), 25-mg (n=30), or 10-mg (n=28) subcutaneous injections of ixekizumab or placebo (n=27) at weeks 0, 2, 4, 8, 12, and 16.16 At 12 weeks, the percentage of participants who achieved a 75% reduction in PASI score from baseline was significantly greater with ixekizumab (82.1% with 150-mg dose, 82.8% with 75-mg dose, 76.7% with 25-mg dose) than with placebo (7.7%)(P<.001 for each comparison), except with the 10-mg dose. Similarly, a greater percentage of participants in the same ixekizumab groups achieved a 90% reduction (71.4% with 150-mg dose, 58.6% with 75-mg dose, 50.0% with 25-mg dose) and a 100% reduction (39.3% with 150-mg dose, 37.9% with 75-mg dose) in PASI score compared to placebo (0%)(P<.001 for each comparison). Significant reductions in PASI scores were evident as early as week 1 in the 150-mg and 75-mg groups, and these reductions were sustained for 20 weeks (P<.05).16 Phase 3 studies of ixekizumab currently are underway.
Brodalumab
The third IL-17 blocker in the pipeline is brodalumab, a human monoclonal antibody against IL-17RA, which blocks signaling of IL-17A and IL-17F as well as the IL-17A/F heterodimer, all of which are involved in the inflammatory process of psoriasis. Brodalumab was evaluated in a phase 2, double-blind, placebo-controlled, dose-ranging study of 198 participants who were randomized to receive 70 mg (n=39), 140 mg (n=39), 210 mg (n=40), or 280 mg (n=42) of brodalumab or placebo (n=38).17 At week 12, improvements of at least 75% and at least 90% in PASI score were achieved by 77% and 72%, respectively, in the 140-mg group, and 82% and 75%, respectively, in the 210-mg group compared to 0% of the placebo group (P<.001 for all comparisons). One hundred percent improvement in PASI was achieved by 38% of participants in the 140-mg group and 62% in the 210-mg group. No participants in the placebo group demonstrated improvement of 75% or higher. The most common AEs were nasopharyngitis, upper respiratory tract infection, arthralgia, and injection-site erythema. Serious AEs reported during the study included renal colic (1 participant), ec-topic pregnancy (1 participant), and grade 3 asymptomatic neutropenia (2 participants). Both cases of neutropenia were noted at the first assessment after brodalumab initiation (week 2) and resolved when the study drug was withheld.17
Results for this new IL-17 blocker are encouraging, but phase 3 data of brodalumab will need to be awaited.
Tildrakizumab
Tildrakizumab is a humanized IgG1 monoclonal antibody that blocks the p19 subunit of IL-23. In a randomized, double-blind, phase 2b trial, 355 adults with moderate to severe psoriasis were randomized to receive subcutaneous injections of tildrakizumab (5 mg, 25 mg, 100 mg, or 200 mg) or placebo.18 In part 1 of the study, injections were administered at weeks 0 and 4. Part 2 of the study started at week 16. In part 2, responders with a 75% improvement in PASI score in the 5- and 25-mg groups continued their dose, while responders in the 100- or 200-mg groups were randomized again to continue the same dose or a reduced dose (100 mg to 25 mg; 200 mg to 100 mg) every 12 weeks from weeks 16 to 52. Those in the placebo group received tildrakizumab 25 mg every 12 weeks in part 2. The primary end point was the mean change in PASI score from baseline to week 16, which was significantly greater in all tildrakizumab groups than in the placebo group (P<.001 for all comparisons). Improvements of 75% in PASI score were achieved by 74% in the 200-mg group, 66% in the 100-mg group, 64% in the 25-mg group, and 33% in the 5-mg group. In contrast, 4.9% in the placebo group achieved an improvement of 75%. At week 52, no loss of efficacy was seen in those participants who had achieved 75% improvement in PASI score at week 16 and had continued their prior doses. The rates of AEs seen in the tildrakizumab groups were 60% to 71% compared to 69% in the placebo group. The most common AE was nasopharyngitis, occurring in 12% to 20% of participants in each group. Serious AEs were uncommon.18 Phase 3 studies are currently underway.19
Guselkumab
Guselkumab is a human IgG1 monoclonal antibody in clinical development that specifically blocks the p19 subunit of IL-23. In a double-blind, placebo-controlled, phase 1 study, 24 participants with moderate to severe plaque psoriasis were randomized to receive a single 10-mg (n=5), 30-mg (n=5), 100-mg (n=5), or 300-mg (n=5) dose of guselkumab or placebo (n=4).20 At week 12, 50% in the 10-mg group, 60% in both the 30- and 100-mg groups, and 100% in the 300-mg group showed 75% improvement in PASI score versus 0% in the placebo group. Improvements in PASI scores were generally maintained through week 24. The rates of AEs were 65% (13/20) in the combined guselkumab group and 50% (2/4) in the placebo group.20
Small Molecule Inhibitors
In contrast to biologics, which mainly target soluble cytokine or cellular receptors, small molecule inhibitors target enzymes within signaling pathways. Small molecule inhibitors have some advantages over biologics in that they are relatively inexpensive to produce and can be administered orally; thus, they may be preferred by some patients over injectable drugs. There are several agents that are undergoing clinical trials in psoriasis, including PDE4 inhibitors and Jak inhibitors.
Apremilast
Apremilast is an oral small molecule PDE4 inhibitor that was approved by the US Food and Drug Administration in March 2014 for the treatment of adult patients with active psoriatic arthritis; an indication for moderate to severe plaque psoriasis was approved in September 2014.21 Phosphodiesterase 4 is a cyclic adenosine monophosphate–specific phosphodiesterase inhibitor, which is dominant in inflammatory cells. Inhibition of PDE4 increases intracellular cyclic adenosine monophosphate levels, thus downregulating proinflammatory cytokines such as TNF-α, IFN-γ, IL-2, IL-12, and IL-23, and increasing the production of anti-inflammatory cytokines such as IL-10.22
Phase 2 and phase 3 studies have demonstrated the clinical efficacy of apremilast in the treatment of patients with moderate to severe plaque psoriasis. In a 16-week randomized, placebo-controlled, phase 3 trial (ESTEEM 2), 408 participants were randomized to receive oral apremilast 30 mg twice daily (n=275) or placebo (n=138).23 Improvement of 75% in PASI score was achieved by 29% of participants in the apremilast group at week 16. The most common AEs were diarrhea (16%) and nausea (18%), which were predominantly mild, occurring most commonly in the first week and resolving within 1 month. No cases of severe diarrhea or severe nausea were reported. Apremilast had no apparent effect on the results of hematological or serum chemistry tests.23 Although the US Food and Drug Administration warns of a possible link between apremilast and depression,24 data are mostly related to roflumilast, another PDE4 inhibitor. Studies in patients with chronic obstructive pulmonary disease have noted increased cases of depression (1.21% vs 0.82%) and suicidal ideation/attempt (0.03% vs 0.02%) in patients treated with roflumilast versus placebo.25
Jak Inhibitors
Janus kinases are a family of intracellular tyrosine kinases that connect several cytokine receptors to the signal transducer and activator of transcription pathways.26 There are 4 Jak family members: Jak1, Jak2, Jak3, and tyrosine kinase 2. Janus kinases 1 and 2 have roles in interferon signaling, while Jak3 transduces signals from IL-2, IL-7, IL-15, and IL-21, which are T-cell growth and survival factors.
Tofacitinib is a novel oral signal transduction molecule that blocks the Jak3 pathway. A phase 2b, 12-week, dose-ranging study was conducted to assess the efficacy and safety of 3 twice-daily regimens of tofacitinib versus placebo in patients with moderate to severe chronic plaque psoriasis.27 One hundred and ninety-seven participants were randomized to receive oral tofacitinib (2 mg, 5 mg, or 15 mg; n=49 each) or placebo (n=50) twice daily for 12 weeks with a 4-week follow-up period. The primary end point was the proportion of participants achieving at least a 75% reduction in PASI score at week 12 (25.0% with 2 mg, 40.8% with 5 mg, 66.7% with 15 mg, 2.0% with placebo). Similarly, a higher proportion of participants achieving 90% reduction in PASI score was seen at weeks 8 and 12 in all tofacitinib-treated participants versus placebo. The most common AEs were upper respiratory tract infection, sinusitis, nasopharyngitis, and headache. A number of changes in laboratory parameters occurred in the tofacitinib groups. Mild dose-related decreases in hemoglobin were noted at week 12 for all tofacitinib groups, and a small increase (mean, 0.04 mg/dL) in serum creatinine levels was observed in the 15-mg group. Decreases in neutrophil counts were observed with higher doses of tofacitinib, with the maximum mean decrease of 0.9×103/mm3 from baseline observed in the 15-mg group at week 4. After weeks 4 through 8, mean neutrophil counts began to return to baseline levels. Dose-related increases in total cholesterol, high-density lipoprotein cholesterol, and low-density lipoprotein cholesterol were observed by week 2 and remained at this level through week 12; mean lipid levels decreased to baseline levels after cessation of active treatment. One participant in the 15-mg group developed an elevated alanine aminotransferase level that was more than 2.5 times the highest normal limit. Three participants experienced 5 serious AEs.27 These early results show that tofacitinib can be a safe and effective treatment in patients with psoriasis, but further data from phase 3 studies will need to be awaited.
Another Jak inhibitor under investigationfor the treatment of psoriasis is ruxolitinib, an inhibitor of Jak1 and Jak2, which has been primarily studied as a topical agent for milder cases of the disease.28
Conclusion
Many new drugs are currently on the horizon and will increase our armamentarium for treating psoriasis. Some of these agents promise greater levels of efficacy than currently used therapies. Although this review focuses on systemic agents, there also are a number of topical formulations in the pipeline. These new agents will certainly increase our options when choosing the most suitable treatment for a patient with psoriasis, but safety will remain a primary concern, and time and experience will tell whether efficacy outweighs any potential side effects.
1. Parisi R, Symmons DP, Griffiths CE, et al. Global epidemiology of psoriasis: a systematic review of incidence and prevalence. J Invest Dermatol. 2013;133:377-385.
2. Rapp SR, Feldman SR, Exum ML, et al. Psoriasis causes as much disability as other major medical diseases. J Am Acad Dermatol. 1999;41(3 pt 1):401-407.
3. Shah R, Bewley A. Psoriasis: ‘the badge of shame.’ a case report of a psychological intervention to reduce and potentially clear chronic skin disease. Clin Exp Dermatol. 2014;39:600-603.
4. Vincent FB, Morand EF, Murphy K, et al. Antidrug antibodies (ADAb) to tumour necrosis factor (TNF)-specific neutralising agents in chronic inflammatory diseases: a real issue, a clinical perspective. Ann Rheum Dis. 2013;72:165-178.
5. Chi CC, Wang SH. Efficacy and cost-efficacy of biologic therapies for moderate to severe psoriasis: a meta-analysis and cost-efficacy analysis using the intention-to-treat principle. Biomed Res Int. 2014;2014:862851.
6. Kagami S, Rizzo HL, Lee JJ, et al. Circulating Th17, Th22, and Th1 cells are increased in psoriasis. J Invest Dermatol. 2010;130:1373-1383.
7. Leonardi CL, Gordon KB. New and emerging therapies in psoriasis. Semin Cutan Med Surg. 2014;33(2, suppl 2):S37-S41.
8. Lowes MA, Kikuchi T, Fuentes-Duculan J, et al. Psoriasis vulgaris lesions contain discrete populations of Th1 and Th17 T cells. J Invest Dermatol. 2008;128:1207-1211.
9. Nickoloff BJ, Qin JZ, Nestle FO. Immunopathogenesis of psoriasis. Clin Rev Allergy Immunol. 2007;33:45-56.
10. Fitch E, Harper E, Skorcheva I, et al. Pathophysiology of psoriasis: recent advances on IL-23 and Th17 cytokines. Curr Rheumatol Rep. 2007;9:461-467.
11. Adami S, Cavani A, Rossi F, et al. The role of interleukin-17A in psoriatic disease. BioDrugs. 2014;28:487-497.
12. Hueber W, Patel DD, Dryja T, et al. Effects of AIN457, a fully human antibody to interleukin-17A, on psoriasis, rheumatoid arthritis, and uveitis. Sci Transl Med. 2010;2:52ra72.
13. Langley RG, Elewski BE, Lebwohl M, et al. Secukinumab in plaque psoriasis—results of two phase 3 trials. N Engl J Med. 2014;371:326-338.
14. Puel A, Cypowyj S, Bustamante J, et al. Chronic mucocutaneous candidiasis in humans with inborn errors of interleukin-17 immunity. Science. 2011;332:65-68.
15. Krstic A, Mojsilovic S, Jovcic G, et al. The potential of interleukin-17 to mediate hematopoietic response. Immunol Res. 2012;52:34-41.
16. Leonardi C, Matheson R, Zachariae C, et al. Anti-interleukin-17 monoclonal antibody ixekizumab in chronic plaque psoriasis. N Engl J Med. 2012;366:1190-1199.
17. Papp KA, Leonardi C, Menter A, et al. Brodalumab, an anti-interleukin-17-receptor antibody for psoriasis. N Engl J Med. 2012;366:1181-1189.
18. Langley RGB, Thaci D, Papp KA, et al. MK-3222, an anti–IL-23p19 humanized monoclonal antibody, provides significant improvement in psoriasis over 52 weeks of treatment that is maintained after discontinuation of dosing. Poster presented at: 73rd Annual Meeting of the American Academy of Dermatology; March 21-25, 2014; Denver, CO. Poster 8056.
19. Tausend W, Downing C, Tyring S. Systematic review of interleukin-12, interleukin-17, and interleukin-23 pathway inhibitors for the treatment of moderate-to-severe chronic plaque psoriasis: ustekinumab, briakinumab, tildrakizumab, guselkumab, secukinumab, ixekizumab, and brodalumab. J Cutan Med Surg. 2014;18:156-169.
20. Sofen H, Smith S, Matheson RT, et al. Guselkumab (an IL-23-specific mAb) demonstrates clinical and molecular response in patients with moderate-to-severe psoriasis. J Allergy Clin Immunol. 2014;133:1032-1040.
21. Schafer PH, Parton A, Capone L, et al. Apremilast is a selective PDE4 inhibitor with regulatory effects on innate immunity. Cell Signal. 2014;26:2016-2029.
22. van de Kerkhof PC. Apremilast: a step forward in the treatment of psoriasis? Lancet. 2012;380:708-709.
23. Paul C, Crowley J, Cather J, et al. Apremilast, an oral phosphodiesterase 4 inhibitor, in patients with moderate to severe psoriasis: 16-week results of a phase 3, randomized, controlled trial (ESTEEM 2). Poster presented at: 73rd Annual Meeting of the American Academy of Dermatology; March 21-25, 2014; Denver, CO. Poster 8412.
24. Otezla [product insert]. Summit, NJ: Celgene Corporation; 2014.
25. Rabe KF. Update on roflumilast, a phosphodiesterase 4 inhibitor for the treatment of chronic obstructive pulmonary disease. Br J Pharmacol. 2011;163:53-67.
26. Palanivel JA, Macbeth AE, Chetty NC, et al. An insight into JAK-STAT signalling in dermatology. Clin Exp Dermatol. 2014;39:513-518.
27. Papp KA, Menter A, Strober B, et al. Efficacy and safety of tofacitinib, an oral Janus kinase inhibitor, in the treatment of psoriasis: a phase 2b randomized placebo-controlled dose-ranging study. Br J Dermatol. 2012;167:668-677.
28. Hsu L, Armstrong AW. JAK inhibitors: treatment efficacy and safety profile in patients with psoriasis. J Immunol Res. 2014;2014:283617.
Psoriasis is a common chronic inflammatory skin disease affecting 1% to 8% of the world population, depending on the country.1 Psoriasis can greatly impact quality of life in affected individuals, even in those with limited body surface involvement.2 Studies have demonstrated a high degree of psychological distress associated with psoriasis, leading to depression and poor self-esteem.3
Over the last decade, our improved understanding of the autoimmune inflammatory pathways and the associated changing concepts in psoriasis pathogenesis have led to the development of biological drugs targeting specific components of effector immune mechanisms, and these biological drugs have revolutionized the treatment of psoriasis.4 Although response rates of these biological agents are greater compared to those of conventional systemic drugs,5 current biological drugs fail to demonstrate efficacy in some patients or lose their efficacy over time. In addition to the high costs associated with these drugs, these limitations have driven a continued search for alternative therapies.
Helper T cells (TH17) and the proinflammatory cytokine IL-17 have been shown to play a key role in the pathophysiology of psoriasis, bridging innate and adaptive immune responses. IL-17 is involved in the modulation of proinflammatory cytokines, hematopoietic growth factors, antimicrobial peptides, and chemokines. Increased TH17 activity and high levels of IL-17 have been found in psoriatic plaques, and increased levels of TH17 are found in the plasma of psoriasis patients.6 Increased IL-17 induces neutrophilia, inflammation, and angiogenesis.7 Other cytokines that are highly upregulated in involved skin are tumor necrosis factor a (TNF-α), IL-23, IL-22, and IL-21.8 IL-23 is involved in regulating TH17 cells and is a potent activator of keratinocyte proliferation.9 Blockade of IL-12/23 causes downregulation of TH17 and TH22 cell responses.10 As IL-17 has a key role in protecting skin and mucous membranes from bacterial and fungal infections, IL-17 inhibition can potentially interfere with the inflammatory cascade. However, available data suggest that sufficient residual IL-17 activity remains to maintain immunity against infections.11
Currently approved biological agents for psoriasis target proinflammatory cytokines such as TNF-α, or the p40 subunit of IL-12 and IL-23. A number of novel targeted therapies including biologics as well as small molecule inhibitors targeting various cytokines and molecules involved in the pathogenesis of psoriasis are currently in different stages of development (Table). These drugs include 3 IL-17 inhibitors (secukinumab, ixekizumab, and brodalumab); 2 IL-23 blockers (tildrakizumab and guselkumab); and small molecule inhibitors that target the kinase pathway including apremilast (a phosphodiesterase 4 [PDE4] inhibitor), as well as tofacitinib, baricitinib, and ruxolitinib (Janus kinase [Jak] inhibitors). Small molecule inhibitors can be administered orally and are less expensive to produce than biological agents. This article reviews available data on these new systemic agents in the pipeline.
Novel Biologics
Secukinumab
Secukinumab is a fully human monoclonal IgG1k antibody that selectively binds and neutralizes IL-17A.12 It is the first of the IL-17 antibodies to receive approval for the treatment of moderate to severe psoriasis. In 2 phase 3, double-blind, 52-week trials—ERASURE (Efficacy of Response and Safety of Two Fixed Secukinumab Regimens in Psoriasis) and FIXTURE (Full Year Investigative Examination of Secukinumab vs Etanercept Using Two Dosing Regimens to Determine Efficacy in Psoriasis)—participants were randomly assigned to receive subcutaneous secukinumab at doses of 300 mg (n=245 and n=327, respectively) or 150 mg (n=245 and n=327, respectively) once weekly for 5 weeks then every 4 weeks, or placebo (n=248 and n=326, respectively); in the FIXTURE study only, an etanercept group (n=326) was given a 50-mg dose twice weekly for 12 weeks then once weekly.13
In the ERASURE study, the proportion of participants showing a reduction of 75% or more in psoriasis area and severity index (PASI) score from baseline to week 12 was 81.6% with secukinumab 300 mg, 71.6% with secukinumab 150 mg, and 4.5% with placebo.13 Secondary end point results demonstrated the proportion of participants showing a 90% reduction in PASI score was 59.2% with secukinumab 300 mg and 39.1% with secukinumab 150 mg, which were both superior to placebo (1.2%). The proportion of participants who met the criteria for 100% reduction in PASI score at week 12 also was greater with each secukinumab dose than with placebo.13
In the FIXTURE study, the proportion of participants showing a reduction of 75% or more from baseline in PASI score at week 12 was 77.1% with secukinumab 300 mg, 67.0% with secukinumab 150 mg, 44.0% with etanercept, and 4.9% with placebo.13 Secondary end point results demonstrated the proportion of participants showing a 90% reduction in PASI score was 54.2% with secukinumab 300 mg, 41.9% with secukinumab 150 mg, 20.7% with etanercept, and 1.5% with placebo. The speed of response, which was assessed as the median time to a 50% reduction in mean PASI score from baseline, was significantly shorter with both doses of secukinumab (3.0 weeks and 3.9 weeks, respectively) than with etanercept (7.0 weeks)(P<.001 for both).13
In the FIXTURE study, incidences of adverse events (AEs) were similar in the secukinumab and etanercept groups during both the induction period and the entire treatment period.13 The most common AEs in the secukinumab groups were nasopharyngitis, headache, and diarrhea. The rates of infections or infestations during the induction period were 26.7% with secukinumab 300 mg, 30.9% with secukinumab 150 mg, 24.5% with etanercept, and 19.3% with placebo. Candidal infections were more common with secukinumab than with etanercept during the entire treatment period (4.7% and 2.3% of participants in the secukinumab 300 mg and 150 mg groups, respectively, reported mild or moderate candidal infections). None of these infections resulted in chronic mucocutaneous candidiasis or discontinuation of the study drug and all resolved on their own or with standard therapy. Candidal infection was reported in 1.2% of participants in the etanercept group. Responses at week 12 were sustained in the majority of participants through week 52 with continued secukinumab therapy every 4 weeks. Grade 3 neutropenia occurred in 1.0% of secukinumab-treated participants and in none of the participants in the etanercept group. There were no apparent dose-related differences between the secukinumab groups with respect to AEs, with the exception of mild and moderate candidal infections.13
These efficacy data are impressive and no specific serious safety concerns have been identified to date. However, IL-17A plays a key role in host defense, particularly in mucocutaneous immunity against Candida albicans,14 as well as in hematopoiesis through stimulation of granulopoiesis and neutrophil trafficking,15 and thus we need to remain watchful with regards to Candida albicans infections and neutropenia.
Ixekizumab
Ixekizumab is a humanized IgG4 anti–IL-17A monoclonal antibody. In a phase 2, double-blind, placebo-controlled trial, 142 participants with chronic moderate to severe plaque psoriasis were randomly assigned to receive 150-mg (n=28), 75-mg (n=29), 25-mg (n=30), or 10-mg (n=28) subcutaneous injections of ixekizumab or placebo (n=27) at weeks 0, 2, 4, 8, 12, and 16.16 At 12 weeks, the percentage of participants who achieved a 75% reduction in PASI score from baseline was significantly greater with ixekizumab (82.1% with 150-mg dose, 82.8% with 75-mg dose, 76.7% with 25-mg dose) than with placebo (7.7%)(P<.001 for each comparison), except with the 10-mg dose. Similarly, a greater percentage of participants in the same ixekizumab groups achieved a 90% reduction (71.4% with 150-mg dose, 58.6% with 75-mg dose, 50.0% with 25-mg dose) and a 100% reduction (39.3% with 150-mg dose, 37.9% with 75-mg dose) in PASI score compared to placebo (0%)(P<.001 for each comparison). Significant reductions in PASI scores were evident as early as week 1 in the 150-mg and 75-mg groups, and these reductions were sustained for 20 weeks (P<.05).16 Phase 3 studies of ixekizumab currently are underway.
Brodalumab
The third IL-17 blocker in the pipeline is brodalumab, a human monoclonal antibody against IL-17RA, which blocks signaling of IL-17A and IL-17F as well as the IL-17A/F heterodimer, all of which are involved in the inflammatory process of psoriasis. Brodalumab was evaluated in a phase 2, double-blind, placebo-controlled, dose-ranging study of 198 participants who were randomized to receive 70 mg (n=39), 140 mg (n=39), 210 mg (n=40), or 280 mg (n=42) of brodalumab or placebo (n=38).17 At week 12, improvements of at least 75% and at least 90% in PASI score were achieved by 77% and 72%, respectively, in the 140-mg group, and 82% and 75%, respectively, in the 210-mg group compared to 0% of the placebo group (P<.001 for all comparisons). One hundred percent improvement in PASI was achieved by 38% of participants in the 140-mg group and 62% in the 210-mg group. No participants in the placebo group demonstrated improvement of 75% or higher. The most common AEs were nasopharyngitis, upper respiratory tract infection, arthralgia, and injection-site erythema. Serious AEs reported during the study included renal colic (1 participant), ec-topic pregnancy (1 participant), and grade 3 asymptomatic neutropenia (2 participants). Both cases of neutropenia were noted at the first assessment after brodalumab initiation (week 2) and resolved when the study drug was withheld.17
Results for this new IL-17 blocker are encouraging, but phase 3 data of brodalumab will need to be awaited.
Tildrakizumab
Tildrakizumab is a humanized IgG1 monoclonal antibody that blocks the p19 subunit of IL-23. In a randomized, double-blind, phase 2b trial, 355 adults with moderate to severe psoriasis were randomized to receive subcutaneous injections of tildrakizumab (5 mg, 25 mg, 100 mg, or 200 mg) or placebo.18 In part 1 of the study, injections were administered at weeks 0 and 4. Part 2 of the study started at week 16. In part 2, responders with a 75% improvement in PASI score in the 5- and 25-mg groups continued their dose, while responders in the 100- or 200-mg groups were randomized again to continue the same dose or a reduced dose (100 mg to 25 mg; 200 mg to 100 mg) every 12 weeks from weeks 16 to 52. Those in the placebo group received tildrakizumab 25 mg every 12 weeks in part 2. The primary end point was the mean change in PASI score from baseline to week 16, which was significantly greater in all tildrakizumab groups than in the placebo group (P<.001 for all comparisons). Improvements of 75% in PASI score were achieved by 74% in the 200-mg group, 66% in the 100-mg group, 64% in the 25-mg group, and 33% in the 5-mg group. In contrast, 4.9% in the placebo group achieved an improvement of 75%. At week 52, no loss of efficacy was seen in those participants who had achieved 75% improvement in PASI score at week 16 and had continued their prior doses. The rates of AEs seen in the tildrakizumab groups were 60% to 71% compared to 69% in the placebo group. The most common AE was nasopharyngitis, occurring in 12% to 20% of participants in each group. Serious AEs were uncommon.18 Phase 3 studies are currently underway.19
Guselkumab
Guselkumab is a human IgG1 monoclonal antibody in clinical development that specifically blocks the p19 subunit of IL-23. In a double-blind, placebo-controlled, phase 1 study, 24 participants with moderate to severe plaque psoriasis were randomized to receive a single 10-mg (n=5), 30-mg (n=5), 100-mg (n=5), or 300-mg (n=5) dose of guselkumab or placebo (n=4).20 At week 12, 50% in the 10-mg group, 60% in both the 30- and 100-mg groups, and 100% in the 300-mg group showed 75% improvement in PASI score versus 0% in the placebo group. Improvements in PASI scores were generally maintained through week 24. The rates of AEs were 65% (13/20) in the combined guselkumab group and 50% (2/4) in the placebo group.20
Small Molecule Inhibitors
In contrast to biologics, which mainly target soluble cytokine or cellular receptors, small molecule inhibitors target enzymes within signaling pathways. Small molecule inhibitors have some advantages over biologics in that they are relatively inexpensive to produce and can be administered orally; thus, they may be preferred by some patients over injectable drugs. There are several agents that are undergoing clinical trials in psoriasis, including PDE4 inhibitors and Jak inhibitors.
Apremilast
Apremilast is an oral small molecule PDE4 inhibitor that was approved by the US Food and Drug Administration in March 2014 for the treatment of adult patients with active psoriatic arthritis; an indication for moderate to severe plaque psoriasis was approved in September 2014.21 Phosphodiesterase 4 is a cyclic adenosine monophosphate–specific phosphodiesterase inhibitor, which is dominant in inflammatory cells. Inhibition of PDE4 increases intracellular cyclic adenosine monophosphate levels, thus downregulating proinflammatory cytokines such as TNF-α, IFN-γ, IL-2, IL-12, and IL-23, and increasing the production of anti-inflammatory cytokines such as IL-10.22
Phase 2 and phase 3 studies have demonstrated the clinical efficacy of apremilast in the treatment of patients with moderate to severe plaque psoriasis. In a 16-week randomized, placebo-controlled, phase 3 trial (ESTEEM 2), 408 participants were randomized to receive oral apremilast 30 mg twice daily (n=275) or placebo (n=138).23 Improvement of 75% in PASI score was achieved by 29% of participants in the apremilast group at week 16. The most common AEs were diarrhea (16%) and nausea (18%), which were predominantly mild, occurring most commonly in the first week and resolving within 1 month. No cases of severe diarrhea or severe nausea were reported. Apremilast had no apparent effect on the results of hematological or serum chemistry tests.23 Although the US Food and Drug Administration warns of a possible link between apremilast and depression,24 data are mostly related to roflumilast, another PDE4 inhibitor. Studies in patients with chronic obstructive pulmonary disease have noted increased cases of depression (1.21% vs 0.82%) and suicidal ideation/attempt (0.03% vs 0.02%) in patients treated with roflumilast versus placebo.25
Jak Inhibitors
Janus kinases are a family of intracellular tyrosine kinases that connect several cytokine receptors to the signal transducer and activator of transcription pathways.26 There are 4 Jak family members: Jak1, Jak2, Jak3, and tyrosine kinase 2. Janus kinases 1 and 2 have roles in interferon signaling, while Jak3 transduces signals from IL-2, IL-7, IL-15, and IL-21, which are T-cell growth and survival factors.
Tofacitinib is a novel oral signal transduction molecule that blocks the Jak3 pathway. A phase 2b, 12-week, dose-ranging study was conducted to assess the efficacy and safety of 3 twice-daily regimens of tofacitinib versus placebo in patients with moderate to severe chronic plaque psoriasis.27 One hundred and ninety-seven participants were randomized to receive oral tofacitinib (2 mg, 5 mg, or 15 mg; n=49 each) or placebo (n=50) twice daily for 12 weeks with a 4-week follow-up period. The primary end point was the proportion of participants achieving at least a 75% reduction in PASI score at week 12 (25.0% with 2 mg, 40.8% with 5 mg, 66.7% with 15 mg, 2.0% with placebo). Similarly, a higher proportion of participants achieving 90% reduction in PASI score was seen at weeks 8 and 12 in all tofacitinib-treated participants versus placebo. The most common AEs were upper respiratory tract infection, sinusitis, nasopharyngitis, and headache. A number of changes in laboratory parameters occurred in the tofacitinib groups. Mild dose-related decreases in hemoglobin were noted at week 12 for all tofacitinib groups, and a small increase (mean, 0.04 mg/dL) in serum creatinine levels was observed in the 15-mg group. Decreases in neutrophil counts were observed with higher doses of tofacitinib, with the maximum mean decrease of 0.9×103/mm3 from baseline observed in the 15-mg group at week 4. After weeks 4 through 8, mean neutrophil counts began to return to baseline levels. Dose-related increases in total cholesterol, high-density lipoprotein cholesterol, and low-density lipoprotein cholesterol were observed by week 2 and remained at this level through week 12; mean lipid levels decreased to baseline levels after cessation of active treatment. One participant in the 15-mg group developed an elevated alanine aminotransferase level that was more than 2.5 times the highest normal limit. Three participants experienced 5 serious AEs.27 These early results show that tofacitinib can be a safe and effective treatment in patients with psoriasis, but further data from phase 3 studies will need to be awaited.
Another Jak inhibitor under investigationfor the treatment of psoriasis is ruxolitinib, an inhibitor of Jak1 and Jak2, which has been primarily studied as a topical agent for milder cases of the disease.28
Conclusion
Many new drugs are currently on the horizon and will increase our armamentarium for treating psoriasis. Some of these agents promise greater levels of efficacy than currently used therapies. Although this review focuses on systemic agents, there also are a number of topical formulations in the pipeline. These new agents will certainly increase our options when choosing the most suitable treatment for a patient with psoriasis, but safety will remain a primary concern, and time and experience will tell whether efficacy outweighs any potential side effects.
Psoriasis is a common chronic inflammatory skin disease affecting 1% to 8% of the world population, depending on the country.1 Psoriasis can greatly impact quality of life in affected individuals, even in those with limited body surface involvement.2 Studies have demonstrated a high degree of psychological distress associated with psoriasis, leading to depression and poor self-esteem.3
Over the last decade, our improved understanding of the autoimmune inflammatory pathways and the associated changing concepts in psoriasis pathogenesis have led to the development of biological drugs targeting specific components of effector immune mechanisms, and these biological drugs have revolutionized the treatment of psoriasis.4 Although response rates of these biological agents are greater compared to those of conventional systemic drugs,5 current biological drugs fail to demonstrate efficacy in some patients or lose their efficacy over time. In addition to the high costs associated with these drugs, these limitations have driven a continued search for alternative therapies.
Helper T cells (TH17) and the proinflammatory cytokine IL-17 have been shown to play a key role in the pathophysiology of psoriasis, bridging innate and adaptive immune responses. IL-17 is involved in the modulation of proinflammatory cytokines, hematopoietic growth factors, antimicrobial peptides, and chemokines. Increased TH17 activity and high levels of IL-17 have been found in psoriatic plaques, and increased levels of TH17 are found in the plasma of psoriasis patients.6 Increased IL-17 induces neutrophilia, inflammation, and angiogenesis.7 Other cytokines that are highly upregulated in involved skin are tumor necrosis factor a (TNF-α), IL-23, IL-22, and IL-21.8 IL-23 is involved in regulating TH17 cells and is a potent activator of keratinocyte proliferation.9 Blockade of IL-12/23 causes downregulation of TH17 and TH22 cell responses.10 As IL-17 has a key role in protecting skin and mucous membranes from bacterial and fungal infections, IL-17 inhibition can potentially interfere with the inflammatory cascade. However, available data suggest that sufficient residual IL-17 activity remains to maintain immunity against infections.11
Currently approved biological agents for psoriasis target proinflammatory cytokines such as TNF-α, or the p40 subunit of IL-12 and IL-23. A number of novel targeted therapies including biologics as well as small molecule inhibitors targeting various cytokines and molecules involved in the pathogenesis of psoriasis are currently in different stages of development (Table). These drugs include 3 IL-17 inhibitors (secukinumab, ixekizumab, and brodalumab); 2 IL-23 blockers (tildrakizumab and guselkumab); and small molecule inhibitors that target the kinase pathway including apremilast (a phosphodiesterase 4 [PDE4] inhibitor), as well as tofacitinib, baricitinib, and ruxolitinib (Janus kinase [Jak] inhibitors). Small molecule inhibitors can be administered orally and are less expensive to produce than biological agents. This article reviews available data on these new systemic agents in the pipeline.
Novel Biologics
Secukinumab
Secukinumab is a fully human monoclonal IgG1k antibody that selectively binds and neutralizes IL-17A.12 It is the first of the IL-17 antibodies to receive approval for the treatment of moderate to severe psoriasis. In 2 phase 3, double-blind, 52-week trials—ERASURE (Efficacy of Response and Safety of Two Fixed Secukinumab Regimens in Psoriasis) and FIXTURE (Full Year Investigative Examination of Secukinumab vs Etanercept Using Two Dosing Regimens to Determine Efficacy in Psoriasis)—participants were randomly assigned to receive subcutaneous secukinumab at doses of 300 mg (n=245 and n=327, respectively) or 150 mg (n=245 and n=327, respectively) once weekly for 5 weeks then every 4 weeks, or placebo (n=248 and n=326, respectively); in the FIXTURE study only, an etanercept group (n=326) was given a 50-mg dose twice weekly for 12 weeks then once weekly.13
In the ERASURE study, the proportion of participants showing a reduction of 75% or more in psoriasis area and severity index (PASI) score from baseline to week 12 was 81.6% with secukinumab 300 mg, 71.6% with secukinumab 150 mg, and 4.5% with placebo.13 Secondary end point results demonstrated the proportion of participants showing a 90% reduction in PASI score was 59.2% with secukinumab 300 mg and 39.1% with secukinumab 150 mg, which were both superior to placebo (1.2%). The proportion of participants who met the criteria for 100% reduction in PASI score at week 12 also was greater with each secukinumab dose than with placebo.13
In the FIXTURE study, the proportion of participants showing a reduction of 75% or more from baseline in PASI score at week 12 was 77.1% with secukinumab 300 mg, 67.0% with secukinumab 150 mg, 44.0% with etanercept, and 4.9% with placebo.13 Secondary end point results demonstrated the proportion of participants showing a 90% reduction in PASI score was 54.2% with secukinumab 300 mg, 41.9% with secukinumab 150 mg, 20.7% with etanercept, and 1.5% with placebo. The speed of response, which was assessed as the median time to a 50% reduction in mean PASI score from baseline, was significantly shorter with both doses of secukinumab (3.0 weeks and 3.9 weeks, respectively) than with etanercept (7.0 weeks)(P<.001 for both).13
In the FIXTURE study, incidences of adverse events (AEs) were similar in the secukinumab and etanercept groups during both the induction period and the entire treatment period.13 The most common AEs in the secukinumab groups were nasopharyngitis, headache, and diarrhea. The rates of infections or infestations during the induction period were 26.7% with secukinumab 300 mg, 30.9% with secukinumab 150 mg, 24.5% with etanercept, and 19.3% with placebo. Candidal infections were more common with secukinumab than with etanercept during the entire treatment period (4.7% and 2.3% of participants in the secukinumab 300 mg and 150 mg groups, respectively, reported mild or moderate candidal infections). None of these infections resulted in chronic mucocutaneous candidiasis or discontinuation of the study drug and all resolved on their own or with standard therapy. Candidal infection was reported in 1.2% of participants in the etanercept group. Responses at week 12 were sustained in the majority of participants through week 52 with continued secukinumab therapy every 4 weeks. Grade 3 neutropenia occurred in 1.0% of secukinumab-treated participants and in none of the participants in the etanercept group. There were no apparent dose-related differences between the secukinumab groups with respect to AEs, with the exception of mild and moderate candidal infections.13
These efficacy data are impressive and no specific serious safety concerns have been identified to date. However, IL-17A plays a key role in host defense, particularly in mucocutaneous immunity against Candida albicans,14 as well as in hematopoiesis through stimulation of granulopoiesis and neutrophil trafficking,15 and thus we need to remain watchful with regards to Candida albicans infections and neutropenia.
Ixekizumab
Ixekizumab is a humanized IgG4 anti–IL-17A monoclonal antibody. In a phase 2, double-blind, placebo-controlled trial, 142 participants with chronic moderate to severe plaque psoriasis were randomly assigned to receive 150-mg (n=28), 75-mg (n=29), 25-mg (n=30), or 10-mg (n=28) subcutaneous injections of ixekizumab or placebo (n=27) at weeks 0, 2, 4, 8, 12, and 16.16 At 12 weeks, the percentage of participants who achieved a 75% reduction in PASI score from baseline was significantly greater with ixekizumab (82.1% with 150-mg dose, 82.8% with 75-mg dose, 76.7% with 25-mg dose) than with placebo (7.7%)(P<.001 for each comparison), except with the 10-mg dose. Similarly, a greater percentage of participants in the same ixekizumab groups achieved a 90% reduction (71.4% with 150-mg dose, 58.6% with 75-mg dose, 50.0% with 25-mg dose) and a 100% reduction (39.3% with 150-mg dose, 37.9% with 75-mg dose) in PASI score compared to placebo (0%)(P<.001 for each comparison). Significant reductions in PASI scores were evident as early as week 1 in the 150-mg and 75-mg groups, and these reductions were sustained for 20 weeks (P<.05).16 Phase 3 studies of ixekizumab currently are underway.
Brodalumab
The third IL-17 blocker in the pipeline is brodalumab, a human monoclonal antibody against IL-17RA, which blocks signaling of IL-17A and IL-17F as well as the IL-17A/F heterodimer, all of which are involved in the inflammatory process of psoriasis. Brodalumab was evaluated in a phase 2, double-blind, placebo-controlled, dose-ranging study of 198 participants who were randomized to receive 70 mg (n=39), 140 mg (n=39), 210 mg (n=40), or 280 mg (n=42) of brodalumab or placebo (n=38).17 At week 12, improvements of at least 75% and at least 90% in PASI score were achieved by 77% and 72%, respectively, in the 140-mg group, and 82% and 75%, respectively, in the 210-mg group compared to 0% of the placebo group (P<.001 for all comparisons). One hundred percent improvement in PASI was achieved by 38% of participants in the 140-mg group and 62% in the 210-mg group. No participants in the placebo group demonstrated improvement of 75% or higher. The most common AEs were nasopharyngitis, upper respiratory tract infection, arthralgia, and injection-site erythema. Serious AEs reported during the study included renal colic (1 participant), ec-topic pregnancy (1 participant), and grade 3 asymptomatic neutropenia (2 participants). Both cases of neutropenia were noted at the first assessment after brodalumab initiation (week 2) and resolved when the study drug was withheld.17
Results for this new IL-17 blocker are encouraging, but phase 3 data of brodalumab will need to be awaited.
Tildrakizumab
Tildrakizumab is a humanized IgG1 monoclonal antibody that blocks the p19 subunit of IL-23. In a randomized, double-blind, phase 2b trial, 355 adults with moderate to severe psoriasis were randomized to receive subcutaneous injections of tildrakizumab (5 mg, 25 mg, 100 mg, or 200 mg) or placebo.18 In part 1 of the study, injections were administered at weeks 0 and 4. Part 2 of the study started at week 16. In part 2, responders with a 75% improvement in PASI score in the 5- and 25-mg groups continued their dose, while responders in the 100- or 200-mg groups were randomized again to continue the same dose or a reduced dose (100 mg to 25 mg; 200 mg to 100 mg) every 12 weeks from weeks 16 to 52. Those in the placebo group received tildrakizumab 25 mg every 12 weeks in part 2. The primary end point was the mean change in PASI score from baseline to week 16, which was significantly greater in all tildrakizumab groups than in the placebo group (P<.001 for all comparisons). Improvements of 75% in PASI score were achieved by 74% in the 200-mg group, 66% in the 100-mg group, 64% in the 25-mg group, and 33% in the 5-mg group. In contrast, 4.9% in the placebo group achieved an improvement of 75%. At week 52, no loss of efficacy was seen in those participants who had achieved 75% improvement in PASI score at week 16 and had continued their prior doses. The rates of AEs seen in the tildrakizumab groups were 60% to 71% compared to 69% in the placebo group. The most common AE was nasopharyngitis, occurring in 12% to 20% of participants in each group. Serious AEs were uncommon.18 Phase 3 studies are currently underway.19
Guselkumab
Guselkumab is a human IgG1 monoclonal antibody in clinical development that specifically blocks the p19 subunit of IL-23. In a double-blind, placebo-controlled, phase 1 study, 24 participants with moderate to severe plaque psoriasis were randomized to receive a single 10-mg (n=5), 30-mg (n=5), 100-mg (n=5), or 300-mg (n=5) dose of guselkumab or placebo (n=4).20 At week 12, 50% in the 10-mg group, 60% in both the 30- and 100-mg groups, and 100% in the 300-mg group showed 75% improvement in PASI score versus 0% in the placebo group. Improvements in PASI scores were generally maintained through week 24. The rates of AEs were 65% (13/20) in the combined guselkumab group and 50% (2/4) in the placebo group.20
Small Molecule Inhibitors
In contrast to biologics, which mainly target soluble cytokine or cellular receptors, small molecule inhibitors target enzymes within signaling pathways. Small molecule inhibitors have some advantages over biologics in that they are relatively inexpensive to produce and can be administered orally; thus, they may be preferred by some patients over injectable drugs. There are several agents that are undergoing clinical trials in psoriasis, including PDE4 inhibitors and Jak inhibitors.
Apremilast
Apremilast is an oral small molecule PDE4 inhibitor that was approved by the US Food and Drug Administration in March 2014 for the treatment of adult patients with active psoriatic arthritis; an indication for moderate to severe plaque psoriasis was approved in September 2014.21 Phosphodiesterase 4 is a cyclic adenosine monophosphate–specific phosphodiesterase inhibitor, which is dominant in inflammatory cells. Inhibition of PDE4 increases intracellular cyclic adenosine monophosphate levels, thus downregulating proinflammatory cytokines such as TNF-α, IFN-γ, IL-2, IL-12, and IL-23, and increasing the production of anti-inflammatory cytokines such as IL-10.22
Phase 2 and phase 3 studies have demonstrated the clinical efficacy of apremilast in the treatment of patients with moderate to severe plaque psoriasis. In a 16-week randomized, placebo-controlled, phase 3 trial (ESTEEM 2), 408 participants were randomized to receive oral apremilast 30 mg twice daily (n=275) or placebo (n=138).23 Improvement of 75% in PASI score was achieved by 29% of participants in the apremilast group at week 16. The most common AEs were diarrhea (16%) and nausea (18%), which were predominantly mild, occurring most commonly in the first week and resolving within 1 month. No cases of severe diarrhea or severe nausea were reported. Apremilast had no apparent effect on the results of hematological or serum chemistry tests.23 Although the US Food and Drug Administration warns of a possible link between apremilast and depression,24 data are mostly related to roflumilast, another PDE4 inhibitor. Studies in patients with chronic obstructive pulmonary disease have noted increased cases of depression (1.21% vs 0.82%) and suicidal ideation/attempt (0.03% vs 0.02%) in patients treated with roflumilast versus placebo.25
Jak Inhibitors
Janus kinases are a family of intracellular tyrosine kinases that connect several cytokine receptors to the signal transducer and activator of transcription pathways.26 There are 4 Jak family members: Jak1, Jak2, Jak3, and tyrosine kinase 2. Janus kinases 1 and 2 have roles in interferon signaling, while Jak3 transduces signals from IL-2, IL-7, IL-15, and IL-21, which are T-cell growth and survival factors.
Tofacitinib is a novel oral signal transduction molecule that blocks the Jak3 pathway. A phase 2b, 12-week, dose-ranging study was conducted to assess the efficacy and safety of 3 twice-daily regimens of tofacitinib versus placebo in patients with moderate to severe chronic plaque psoriasis.27 One hundred and ninety-seven participants were randomized to receive oral tofacitinib (2 mg, 5 mg, or 15 mg; n=49 each) or placebo (n=50) twice daily for 12 weeks with a 4-week follow-up period. The primary end point was the proportion of participants achieving at least a 75% reduction in PASI score at week 12 (25.0% with 2 mg, 40.8% with 5 mg, 66.7% with 15 mg, 2.0% with placebo). Similarly, a higher proportion of participants achieving 90% reduction in PASI score was seen at weeks 8 and 12 in all tofacitinib-treated participants versus placebo. The most common AEs were upper respiratory tract infection, sinusitis, nasopharyngitis, and headache. A number of changes in laboratory parameters occurred in the tofacitinib groups. Mild dose-related decreases in hemoglobin were noted at week 12 for all tofacitinib groups, and a small increase (mean, 0.04 mg/dL) in serum creatinine levels was observed in the 15-mg group. Decreases in neutrophil counts were observed with higher doses of tofacitinib, with the maximum mean decrease of 0.9×103/mm3 from baseline observed in the 15-mg group at week 4. After weeks 4 through 8, mean neutrophil counts began to return to baseline levels. Dose-related increases in total cholesterol, high-density lipoprotein cholesterol, and low-density lipoprotein cholesterol were observed by week 2 and remained at this level through week 12; mean lipid levels decreased to baseline levels after cessation of active treatment. One participant in the 15-mg group developed an elevated alanine aminotransferase level that was more than 2.5 times the highest normal limit. Three participants experienced 5 serious AEs.27 These early results show that tofacitinib can be a safe and effective treatment in patients with psoriasis, but further data from phase 3 studies will need to be awaited.
Another Jak inhibitor under investigationfor the treatment of psoriasis is ruxolitinib, an inhibitor of Jak1 and Jak2, which has been primarily studied as a topical agent for milder cases of the disease.28
Conclusion
Many new drugs are currently on the horizon and will increase our armamentarium for treating psoriasis. Some of these agents promise greater levels of efficacy than currently used therapies. Although this review focuses on systemic agents, there also are a number of topical formulations in the pipeline. These new agents will certainly increase our options when choosing the most suitable treatment for a patient with psoriasis, but safety will remain a primary concern, and time and experience will tell whether efficacy outweighs any potential side effects.
1. Parisi R, Symmons DP, Griffiths CE, et al. Global epidemiology of psoriasis: a systematic review of incidence and prevalence. J Invest Dermatol. 2013;133:377-385.
2. Rapp SR, Feldman SR, Exum ML, et al. Psoriasis causes as much disability as other major medical diseases. J Am Acad Dermatol. 1999;41(3 pt 1):401-407.
3. Shah R, Bewley A. Psoriasis: ‘the badge of shame.’ a case report of a psychological intervention to reduce and potentially clear chronic skin disease. Clin Exp Dermatol. 2014;39:600-603.
4. Vincent FB, Morand EF, Murphy K, et al. Antidrug antibodies (ADAb) to tumour necrosis factor (TNF)-specific neutralising agents in chronic inflammatory diseases: a real issue, a clinical perspective. Ann Rheum Dis. 2013;72:165-178.
5. Chi CC, Wang SH. Efficacy and cost-efficacy of biologic therapies for moderate to severe psoriasis: a meta-analysis and cost-efficacy analysis using the intention-to-treat principle. Biomed Res Int. 2014;2014:862851.
6. Kagami S, Rizzo HL, Lee JJ, et al. Circulating Th17, Th22, and Th1 cells are increased in psoriasis. J Invest Dermatol. 2010;130:1373-1383.
7. Leonardi CL, Gordon KB. New and emerging therapies in psoriasis. Semin Cutan Med Surg. 2014;33(2, suppl 2):S37-S41.
8. Lowes MA, Kikuchi T, Fuentes-Duculan J, et al. Psoriasis vulgaris lesions contain discrete populations of Th1 and Th17 T cells. J Invest Dermatol. 2008;128:1207-1211.
9. Nickoloff BJ, Qin JZ, Nestle FO. Immunopathogenesis of psoriasis. Clin Rev Allergy Immunol. 2007;33:45-56.
10. Fitch E, Harper E, Skorcheva I, et al. Pathophysiology of psoriasis: recent advances on IL-23 and Th17 cytokines. Curr Rheumatol Rep. 2007;9:461-467.
11. Adami S, Cavani A, Rossi F, et al. The role of interleukin-17A in psoriatic disease. BioDrugs. 2014;28:487-497.
12. Hueber W, Patel DD, Dryja T, et al. Effects of AIN457, a fully human antibody to interleukin-17A, on psoriasis, rheumatoid arthritis, and uveitis. Sci Transl Med. 2010;2:52ra72.
13. Langley RG, Elewski BE, Lebwohl M, et al. Secukinumab in plaque psoriasis—results of two phase 3 trials. N Engl J Med. 2014;371:326-338.
14. Puel A, Cypowyj S, Bustamante J, et al. Chronic mucocutaneous candidiasis in humans with inborn errors of interleukin-17 immunity. Science. 2011;332:65-68.
15. Krstic A, Mojsilovic S, Jovcic G, et al. The potential of interleukin-17 to mediate hematopoietic response. Immunol Res. 2012;52:34-41.
16. Leonardi C, Matheson R, Zachariae C, et al. Anti-interleukin-17 monoclonal antibody ixekizumab in chronic plaque psoriasis. N Engl J Med. 2012;366:1190-1199.
17. Papp KA, Leonardi C, Menter A, et al. Brodalumab, an anti-interleukin-17-receptor antibody for psoriasis. N Engl J Med. 2012;366:1181-1189.
18. Langley RGB, Thaci D, Papp KA, et al. MK-3222, an anti–IL-23p19 humanized monoclonal antibody, provides significant improvement in psoriasis over 52 weeks of treatment that is maintained after discontinuation of dosing. Poster presented at: 73rd Annual Meeting of the American Academy of Dermatology; March 21-25, 2014; Denver, CO. Poster 8056.
19. Tausend W, Downing C, Tyring S. Systematic review of interleukin-12, interleukin-17, and interleukin-23 pathway inhibitors for the treatment of moderate-to-severe chronic plaque psoriasis: ustekinumab, briakinumab, tildrakizumab, guselkumab, secukinumab, ixekizumab, and brodalumab. J Cutan Med Surg. 2014;18:156-169.
20. Sofen H, Smith S, Matheson RT, et al. Guselkumab (an IL-23-specific mAb) demonstrates clinical and molecular response in patients with moderate-to-severe psoriasis. J Allergy Clin Immunol. 2014;133:1032-1040.
21. Schafer PH, Parton A, Capone L, et al. Apremilast is a selective PDE4 inhibitor with regulatory effects on innate immunity. Cell Signal. 2014;26:2016-2029.
22. van de Kerkhof PC. Apremilast: a step forward in the treatment of psoriasis? Lancet. 2012;380:708-709.
23. Paul C, Crowley J, Cather J, et al. Apremilast, an oral phosphodiesterase 4 inhibitor, in patients with moderate to severe psoriasis: 16-week results of a phase 3, randomized, controlled trial (ESTEEM 2). Poster presented at: 73rd Annual Meeting of the American Academy of Dermatology; March 21-25, 2014; Denver, CO. Poster 8412.
24. Otezla [product insert]. Summit, NJ: Celgene Corporation; 2014.
25. Rabe KF. Update on roflumilast, a phosphodiesterase 4 inhibitor for the treatment of chronic obstructive pulmonary disease. Br J Pharmacol. 2011;163:53-67.
26. Palanivel JA, Macbeth AE, Chetty NC, et al. An insight into JAK-STAT signalling in dermatology. Clin Exp Dermatol. 2014;39:513-518.
27. Papp KA, Menter A, Strober B, et al. Efficacy and safety of tofacitinib, an oral Janus kinase inhibitor, in the treatment of psoriasis: a phase 2b randomized placebo-controlled dose-ranging study. Br J Dermatol. 2012;167:668-677.
28. Hsu L, Armstrong AW. JAK inhibitors: treatment efficacy and safety profile in patients with psoriasis. J Immunol Res. 2014;2014:283617.
1. Parisi R, Symmons DP, Griffiths CE, et al. Global epidemiology of psoriasis: a systematic review of incidence and prevalence. J Invest Dermatol. 2013;133:377-385.
2. Rapp SR, Feldman SR, Exum ML, et al. Psoriasis causes as much disability as other major medical diseases. J Am Acad Dermatol. 1999;41(3 pt 1):401-407.
3. Shah R, Bewley A. Psoriasis: ‘the badge of shame.’ a case report of a psychological intervention to reduce and potentially clear chronic skin disease. Clin Exp Dermatol. 2014;39:600-603.
4. Vincent FB, Morand EF, Murphy K, et al. Antidrug antibodies (ADAb) to tumour necrosis factor (TNF)-specific neutralising agents in chronic inflammatory diseases: a real issue, a clinical perspective. Ann Rheum Dis. 2013;72:165-178.
5. Chi CC, Wang SH. Efficacy and cost-efficacy of biologic therapies for moderate to severe psoriasis: a meta-analysis and cost-efficacy analysis using the intention-to-treat principle. Biomed Res Int. 2014;2014:862851.
6. Kagami S, Rizzo HL, Lee JJ, et al. Circulating Th17, Th22, and Th1 cells are increased in psoriasis. J Invest Dermatol. 2010;130:1373-1383.
7. Leonardi CL, Gordon KB. New and emerging therapies in psoriasis. Semin Cutan Med Surg. 2014;33(2, suppl 2):S37-S41.
8. Lowes MA, Kikuchi T, Fuentes-Duculan J, et al. Psoriasis vulgaris lesions contain discrete populations of Th1 and Th17 T cells. J Invest Dermatol. 2008;128:1207-1211.
9. Nickoloff BJ, Qin JZ, Nestle FO. Immunopathogenesis of psoriasis. Clin Rev Allergy Immunol. 2007;33:45-56.
10. Fitch E, Harper E, Skorcheva I, et al. Pathophysiology of psoriasis: recent advances on IL-23 and Th17 cytokines. Curr Rheumatol Rep. 2007;9:461-467.
11. Adami S, Cavani A, Rossi F, et al. The role of interleukin-17A in psoriatic disease. BioDrugs. 2014;28:487-497.
12. Hueber W, Patel DD, Dryja T, et al. Effects of AIN457, a fully human antibody to interleukin-17A, on psoriasis, rheumatoid arthritis, and uveitis. Sci Transl Med. 2010;2:52ra72.
13. Langley RG, Elewski BE, Lebwohl M, et al. Secukinumab in plaque psoriasis—results of two phase 3 trials. N Engl J Med. 2014;371:326-338.
14. Puel A, Cypowyj S, Bustamante J, et al. Chronic mucocutaneous candidiasis in humans with inborn errors of interleukin-17 immunity. Science. 2011;332:65-68.
15. Krstic A, Mojsilovic S, Jovcic G, et al. The potential of interleukin-17 to mediate hematopoietic response. Immunol Res. 2012;52:34-41.
16. Leonardi C, Matheson R, Zachariae C, et al. Anti-interleukin-17 monoclonal antibody ixekizumab in chronic plaque psoriasis. N Engl J Med. 2012;366:1190-1199.
17. Papp KA, Leonardi C, Menter A, et al. Brodalumab, an anti-interleukin-17-receptor antibody for psoriasis. N Engl J Med. 2012;366:1181-1189.
18. Langley RGB, Thaci D, Papp KA, et al. MK-3222, an anti–IL-23p19 humanized monoclonal antibody, provides significant improvement in psoriasis over 52 weeks of treatment that is maintained after discontinuation of dosing. Poster presented at: 73rd Annual Meeting of the American Academy of Dermatology; March 21-25, 2014; Denver, CO. Poster 8056.
19. Tausend W, Downing C, Tyring S. Systematic review of interleukin-12, interleukin-17, and interleukin-23 pathway inhibitors for the treatment of moderate-to-severe chronic plaque psoriasis: ustekinumab, briakinumab, tildrakizumab, guselkumab, secukinumab, ixekizumab, and brodalumab. J Cutan Med Surg. 2014;18:156-169.
20. Sofen H, Smith S, Matheson RT, et al. Guselkumab (an IL-23-specific mAb) demonstrates clinical and molecular response in patients with moderate-to-severe psoriasis. J Allergy Clin Immunol. 2014;133:1032-1040.
21. Schafer PH, Parton A, Capone L, et al. Apremilast is a selective PDE4 inhibitor with regulatory effects on innate immunity. Cell Signal. 2014;26:2016-2029.
22. van de Kerkhof PC. Apremilast: a step forward in the treatment of psoriasis? Lancet. 2012;380:708-709.
23. Paul C, Crowley J, Cather J, et al. Apremilast, an oral phosphodiesterase 4 inhibitor, in patients with moderate to severe psoriasis: 16-week results of a phase 3, randomized, controlled trial (ESTEEM 2). Poster presented at: 73rd Annual Meeting of the American Academy of Dermatology; March 21-25, 2014; Denver, CO. Poster 8412.
24. Otezla [product insert]. Summit, NJ: Celgene Corporation; 2014.
25. Rabe KF. Update on roflumilast, a phosphodiesterase 4 inhibitor for the treatment of chronic obstructive pulmonary disease. Br J Pharmacol. 2011;163:53-67.
26. Palanivel JA, Macbeth AE, Chetty NC, et al. An insight into JAK-STAT signalling in dermatology. Clin Exp Dermatol. 2014;39:513-518.
27. Papp KA, Menter A, Strober B, et al. Efficacy and safety of tofacitinib, an oral Janus kinase inhibitor, in the treatment of psoriasis: a phase 2b randomized placebo-controlled dose-ranging study. Br J Dermatol. 2012;167:668-677.
28. Hsu L, Armstrong AW. JAK inhibitors: treatment efficacy and safety profile in patients with psoriasis. J Immunol Res. 2014;2014:283617.
Practice Points
- Secukinumab is an anti–IL-17 antibody approved for the treatment of psoriasis. It is indicated for the treatment of moderate to severe plaque psoriasis in adult patients who are candidates for systemic therapy or phototherapy.
- The new biological agents have shown promising results with some patients achieving psoriasis area and severity index scores of 90 and 100.
- A number of small molecule inhibitors also are in the pipeline, with apremilast the first one to have reached approval for psoriasis.