User login
Medication-assisted recovery for opioid use disorder: A guide
Medication-assisted recovery (MAR)—the preferred terminology for the service formerly known as medication-assisted treatment—entails a comprehensive set of interventions for managing opioid use disorder (OUD), including medications for opioid use disorder (MOUD). Despite the benefits of MAR—reducing opioid use, opioid-related mortality, and health care costs1-3—only 11% of patients with a diagnosis of OUD received MOUD in 2020.3
Primary care physicians, including family physicians, are well positioned to provide MAR across the patient’s lifespan. However, many family medicine clinicians do not possess the logistical knowledge or resources to implement this service.4 In this article, we describe options for, and barriers to, MAR and societal issues that have an impact on the care of these patients.
Pathophysiology of OUD
Opioids relieve pain by stimulating μ-opioid receptors and activating the brain’s reward system. These pleasurable effects motivate repeated use.5 Frequent opioid exposure causes neuroadaptation, tolerance, and dependence. For patients with OUD who are misusing illicit or prescription opioids, periods of abstinence following neuroadaptation lead to withdrawal symptoms that vary in intensity, depending on the drug, dose, and duration of use. Upregulated noradrenergic tone and dopamine deficiency manifest as numerous signs and symptoms of withdrawal, including5:
- Physiologic: secretory (diaphoresis, rhinorrhea, lacrimation, vomiting, diarrhea) and stimulatory (mydriasis, piloerection, hypertension, tachycardia, insomnia)
- Psychological: pain, cravings, dysphoria, anxiety.
A single episode of opioid withdrawal is not directly life-threatening, but untreated episodes can progressively amplify negative feedback and reinforce continued opioid use.6 Left untreated, withdrawal can be terminal.
Medication-assisted recovery: Effective intervention
MAR services that integrate medical, behavioral, and psychosocial programs can reduce mortality from OUD 2-fold.7,8 A meta-analysis found that, when MAR services are rendered in primary care, treatment retention improves by 25% (number needed to treat [NNT] = 6) and ongoing illicit opioid use is reduced by 50% (NNT = 6), relative to care at a specialty clinic9—highlighting a role for family medicine clinicians in treating OUD.
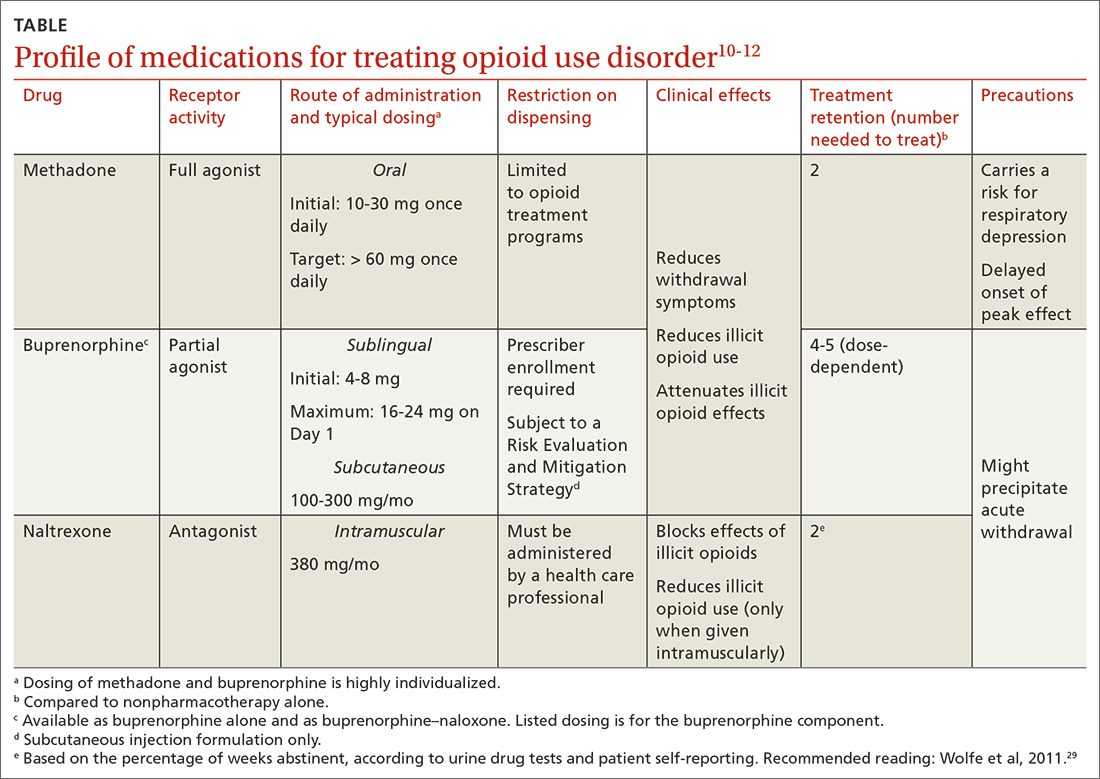
All 3 US Food and Drug Administration (FDA)–approved MOUD (methadone, buprenorphine, and naltrexone) reduce cravings; 2 (methadone and buprenorphine) mitigate withdrawal symptoms by activating the μ-opioid receptor; and naltrexone diminishes the reinforcing effects of use (TABLE10-12). It is crucial to recognize the pharmacologic distinctions among MOUD because untreated withdrawal syndromes increase dropout from treatment programs and subsequent relapse.13
The Hx of medication-assisted recovery
To understand the landscape of MAR, it is important to understand the history of opioid treatment in the United States. In 1966, Congress passed the Narcotic Addiction Rehabilitation Act (NARA), which secured federal assistance by which state and local governments could develop drug treatment programs.14 NARA permitted legal offenders with OUD to be civilly committed to treatment programs, rather than prosecuted. However, limited resources and a burgeoning population led, instead, to low-cost outpatient programs saddled by strict requirements that lacked a basis for improving clinical outcomes.
Continue to: At the time NARA...
At the time NARA was passed by Congress, OUD was viewed—inaccurately—as a criminal problem, not a medical one. Subsequent legislation was crafted through that lens, which has placed a heavy burden on patients until today.14 Although medical understanding of OUD has advanced tremendously over the past 50 years, treatment remains siloed from mainstream medicine, even in primary care.
There is no one-size-fits-all approach to MAR, and relapse is common. Patient-specific factors and the availability of resources should be considered when designing the most individualized, advantageous plan for MAR.
Methadone
Background. Methadone has the most extensive history for treating OUD and consistently has demonstrated efficacy.13 A meta-analysis of randomized controlled trials comparing methadone to nonpharmacotherapy alone found that methadone improved treatment retention by an absolute 57% (NNT = 2).10
Methadone was approved by the FDA for detoxification and maintenance treatment in the early 1970s, although the Narcotic Addict Treatment Act (NATA) of 1974 restricted dispensing of maintenance treatment to highly regulated clinics known as opioid treatment programs (OTPs).14 NATA required the treating physician to register with the US Drug Enforcement Agency (DEA) and to comply with conservative dosing regimens and observed dosing.
Over time, regulations evolved to give the physician greater flexibility in developing a care plan, allowing “take-home” doses, and improving patients’ access to care. Although access to methadone for the treatment of OUD remains limited to federally certified OTPs, regulations facilitate incorporation of a whole-person approach to care, including counseling, individual and group therapy, and toxicology testing.7
Continue to: Clinical considerations
Clinical considerations. Methadone requires slow titration. For patients starting methadone as an outpatient, federal law15 limits the initial dose to 30 mg and requires physician documentation when the first-day total dosage exceeds 40 mg. This dosing constraint makes it challenging to provide care because a daily dosage ≥ 60 mg has been found to produce, first, higher program retention (relative risk = 1.36; 95% CI, 1.13-1.63) and, second, greater reduction in illicit opioid use (relative risk = 1.59; 95% CI, 1.16-2.18) than is seen in patients who receive a lower daily dosage.16
Due to a prolonged elimination half-life, methadone reaches steady-state in 3 to 5 days. Patients and their families should be educated that withdrawal symptoms might not feel fully managed in the first few days of therapy and that time is required to experience safely the regimen’s full effects.
Aggressive dose-titration during methadone induction can result in drug accumulation and respiratory depression. The risk for methadone-related mortality is highest in the first 2 weeks of therapy, mostly related to overdose potential if the drug is combined with other opioids.17
Buprenorphine
Background. The prescribing rate for buprenorphine, particularly in primary care, is accelerating.18 A meta-analysis of randomized controlled trials found that11:
- compared to placebo, buprenorphine, at any dosage, improves treatment retention by an absolute 21% to 28% (NNT = 4-5)
- patients receiving high-dose buprenorphine (≥ 16 mg/d) had fewer evident cases of illicit opioid use.
Unlike methadone, buprenorphine exerts partial agonism at the μ-opioid receptor, resulting in a so-called ceiling effect that significantly reduces the adverse effect profile, including respiratory depression and euphoria, relative to a full-agonist opioid, such as methadone.19
Continue to: Whereas accessing methadone...
Whereas accessing methadone is limited to OTPs, buprenorphine is available for office-based treatment. By hosting OUD treatment and primary care in the same place, primary care physicians can provide comprehensive medical care including and beyond OUD, thereby improving retention and managing comorbidity.20
Integrated models involving support staff—eg, nurses, behavioral health providers, and pharmacists—have produced the greatest success with office-based treatment models.21 Office-based treatment normalizes OUD as a chronic disease managed by the primary care physician, enabling concurrent harm-reduction strategies; medication reconciliation; and convenient, regular prescribing intervals (eg, every 30 days).22 Nevertheless, access to buprenorphine is limited. Because buprenorphine is a controlled substance, the Ryan Haight Online Pharmacy Consumer Protection Act of 2008 prevents initial prescribing of buprenorphine without in-person evaluation. Telehealth consultations increased access to buprenorphine through temporary exceptions during the COVID-19 pandemic. However, revised rules and regulations for telehealth visits for these controlled substances are forthcoming from the DEA as temporary exceptions for telehealth consultations come to an end. Additionally, prescribing buprenorphine for OUD requires that the treating physician undergo specific training and obtain qualifications, which have evolved over time through federal legislation.
The Drug Addiction Treatment Act of 2000 (DATA 2000) authorized what is known as an X-waiver, which allows physicians to prescribe controlled substances for office-based treatment of OUD, provided that:
- they are registered to do so with the Substance Abuse and Mental Health Services Administration and the DEA
- they have had subspecialty training in addiction or completed an 8-hour training course
- they are able to refer patients to appropriate counseling and ancillary services.
DATA 2000 restricted patient panel sizes to 30 patients in the first year, expanding thereafter upon appropriate certification.
The Comprehensive Addiction and Recovery Act of 2016 (CARA) and the Substance Use Disorder Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities Act of 2018 (the SUPPORT Act) collectively extended prescribing authority for MOUD to other qualifying practitioners (eg, advanced practice clinicians). Despite these attempts to expand access to services, the overdose death rate has continued to increase.
Continue to: To further expand access to MAR...
To further expand access to MAR, the US Department of Health and Human Services updated its practice guidelines in April 2021, allowing clinicians to bypass X-waiver training requirements by applying for a notification-of-intent (NOI) buprenorphine waiver.a However, clinicians are still limited to prescribing buprenorphine for 30 patients at a time. Clinicians who undergo complete X-waiver training may prescribe for 100 patients in the first year and, if eligible, 275 patients thereafter.
In addition, as a component of the Consolidation Appropriations Act of 2023, Congress passed the Mainstreaming Addiction Treatment Act of 2021, or MAT 2021, and Medication Access and Training Expansion Act of 2021, or MATE 2021. MAT eliminated the X-waiver, NOI, and restrictions on the number of patients for whom a provider could prescribe buprenorphine, under federal authority; however, restrictions within one’s state might limit the ability to prescribe buprenorphine. MATE 2021 is an educational requirement for licensing by the DEA (at application and renewal) that will require prescribers to complete 8 hours of training in substance use disorders starting in June 2023.
Use of the monthly injectable extended-release buprenorphine productb is limited by an FDA Risk Evaluation and Mitigation Strategy (REMS) program, which requires specialized training and certification by the prescriber, distributor, and administering clinician. REMS reduces buprenorphine accessibility due to time, cost, and regulatory barriers; although such restrictions have been instituted with the patient’s safety in mind, any limitation to buprenorphine prescribing, apart from controlled substance licensure, serves only to limit access to a primary component of MAR.
Clinical considerations. Due to the competitive nature of buprenorphine and its high affinity for the μ-opioid receptor, the drug can displace other opioid agonists and precipitate acute withdrawal. The withdrawal experience can thereby condition fear and disfavor toward buprenorphine among patients.
It is vital, therefore, that (1) patients’ expectations for treatment be managed appropriately and (2) the treating physician be prepared to provide additional buprenorphine for adequate maintenance doses and utilize adjunct comfort agents (clonidine, nonsteroidal anti-inflammatory drugs, ondansetron) to manage acute withdrawal symptoms. Newer buprenorphine dosing strategies, such as micro-induction and macro-induction, have emerged to curtail these risks.23,24 This is an evolving area of MAR; newer low-threshold initiation strategies25 (see “Low-threshold MOUD prescribing models,” in the text that follows) and evidence that supports micro-induction26 might eliminate the practice of requiring active withdrawal for treatment.
Continue to: Regardless of the strategy...
Regardless of the strategy for dosing buprenorphine, it’s critical that patients be educated on how to initiate treatment outside a clinical setting, such as at home, where they occupy a familiar haven during a potentially uncomfortable time and can be as effective at initiation as they would be in a clinical setting, with no difference in precipitation of adverse effects.
At-home induction might be more appropriate for patients who are not yet in significant enough withdrawal while in the physician's office.27 Guidance should be provided on dosing instructions, self-assessment of withdrawal symptoms, and, if applicable, patience with the slow-dissolving sublingual tablet or film formulation.
Naltrexone
Background. Naltrexone is available as an oral tablet and an extended-release, once-monthly intramuscular injection; the latter has demonstrated superiority in MAR.28 Oral naltrexone has limited supporting evidence, is inferior to other MOUD options, and should not be used to treat OUD.7 Altogether, approval of naltrexone for OUD is controversial, due to potentially unethical trials and approval processes,29 although a multicenter randomized controlled trial demonstrated the drug’s noninferiority with respect to treatment retention relative to buprenorphine.30 Used over time, naltrexone does not relieve withdrawal symptoms but can reduce cravings.
Clinical considerations. There are numerous clinical barriers that limit the use of naltrexone.
First, patients should be abstinent from opioids for 7 to 14 days prior to starting therapy; usually, this means undergoing medically supervised withdrawal in a controlled environment. This is an obvious limitation for patients who are constrained financially—those who lack, or have inadequate, health insurance or are unable to be away from their job for an extended time.
Continue to: Second, because naltrexone...
Second, because naltrexone does not address withdrawal symptoms, supportive therapies should be incorporated into the treatment plan, including:
- clonidine for hyperadrenergic symptoms (anxiety, diaphoresis, hypertension)
- nonopioid analgesics for pain
- antiemetics, such as ondansetron and metoclopramide, for nausea or vomiting
- loperamide for diarrhea
- diphenhydramine for insomnia.
Third, patients taking naltrexone have a diminished response to opioids. This complicates pain management in the event of an emergent surgical procedure.
Last, when naltrexone wears off, patients are effectively opioid-naïve, which increases the risk for overdose in those who stop therapy abruptly.29 The increased risk for overdose should be communicated to all patients with OUD who are being treated with naltrexone.
This nonopioid option is appealing to policymakers and is often prioritized in the criminal justice system; however, the decreased efficacy of naltrexone (compared to methadone and buprenorphine), potential for overdose, and challenges in initiating treatment are concerning and limit the drug’s use in many real-world settings.
Because naltrexone is not a controlled substance, regulations regarding maintaining inventory and distribution are more flexible.
Continue to: Overall, the cost-effectiveness...
Overall, the cost-effectiveness of intramuscular naltrexone is unclear. State-administered insurance programs vary in their requirements for coverage of naltrexone treatment.31
Comprehensive medication reconciliation is vital
Overall fragmentation of care within OTPs places patients at risk for adverse events, such as drug interactions.32 Under Title 42 of the US Code,33 patients must provide written consent for an OTP provider to disclose their history of a substance use disorder. Allowing the patient to decide which medical providers can access their treatment records for an OUD benefits patient confidentiality but poses numerous issues worth exploring.
All prescribed controlled substances are recorded in the prescription drug monitoring program, or PDMP, a state-level electronic database accessible to health care professionals to inform prescribing decisions and identify drug interactions. The PDMP has substantially reduced opioid overprescribing and improved identification of patients at risk for overdose or misuse of opioids.
Unlike all other controlled substances, however, prescriptions ordered by an OTP are not recorded in the PDMP (although there are recent exceptions to this scenario). Without such information, a physician might not have important information about the patient when making medical decisions—placing the patient at risk for harmful outcomes, such as drug–drug and drug–disease interactions.
For example: Methadone is associated with a prolonged QT interval,34 increasing the risk for a fatal arrhythmia. Concurrent QT-prolonging medications, such as azithromycin and citalopram, further increase this risk.35 Because methadone dispensing is isolated from the patient’s medical record, the clinician who prescribes MOUD has an incomplete patient history and could make a potentially fatal treatment decision.
Continue to: Diversion is unlikely
Diversion is unlikely
Health care providers often express concern about diversion in MOUD. However, misuse and diversion rates of methadone and buprenorphine have declined steadily since 2011, and, in fact, are actually lower than the diversion rate of prescription antibiotics.36
Regardless, diversion of buprenorphine should not be a concern for physicians prescribing MOUD. Although a prescriber might worry about manipulation of the formulation of buprenorphine for intravenous administration, addition of naloxone to buprenorphine in tablet form diminishes the potential for overdose. Additionally, the ceiling effect of buprenorphine limits the likelihood of significant respiratory depression and euphoria.
Should buprenorphine reach a patient for whom it was not prescribed, it is highly unlikely that an overdose would result. Rather, the medication would protect against the effects of illicit opioids and relieve withdrawal symptoms. Most people with OUD who have misused buprenorphine have done so to relieve withdrawal symptoms,37 not to experience intoxication.
Health care deserts
So-called health care deserts in parts of the United States are an ongoing problem that disproportionately affects lower-income and segregated Black and Hispanic communities38—communities that shoulder the highest burden of OUD and OUD-related mortality39 and whose populace is in greatest need of MAR. Even when health care is accessible in such a desert, some clinicians and pharmacies refuse to prescribe or dispense MOUD because of the accompanying stigma of OUD.
A MAR desert, like a pharmacy desert, is a geographic region—one without access to a MAR or an OTP provider, thereby preventing patients from reaching appropriate care; for some patients, having to travel to the nearest provider can render treatment inaccessible.40
Continue to: Efforts are in place to identify...
Efforts are in place to identify areas at greatest need of OUD-related medical services, such as heat maps that identify areas of increased utilization of emergency medical services for opioid overdose. State-run programs have been implemented to increase access, such as the Illinois Helpline (https://helplineil.org) that provides support and resources for patients, friends, family, and providers.
Novel solutions
Key strategies to increase access to care and slow the opioid epidemic include low-threshold prescribing of MOUD and mobile OTPs.41
Low-threshold MOUD prescribing models. Adoption of one of these models in a medical practice that provides MAR might increase absolute enrollment. A low-threshold prescribing model involves42:
- same-day treatment
- leniency with respect to abstinence periods and a concomitant substance use disorder
- enhanced accessibility to MOUD through nontraditional medical settings.
Low-threshold prescribing is flexible in regard to patients’ needs and bypasses many of the barriers discussed in this article. Impressive multicenter success has been achieved by the CA Bridge program in California (https://cabridge.org), including an increase in recognition of OUD, treatment initiations, and outpatient engagement.25
The cost-effectiveness of low-threshold MOUD prescribing programs remains to be determined.
Mobile OTPs. In July 2021, the DEA authorized a mobile component to existing OTP registrants that is permitted to dispense methadone and buprenorphine. Mobile units are physically separate from the OTP but have similar functions, depending on available space. Services that cannot be provided on the mobile unit of an OTP must be available at its brick-and-mortar location.7 Logistically, OTP registrants no longer need a separate registration to implement a mobile unit, thus expanding care to patients in underserved or remote areas who often encounter barriers to access.43
Conclusion
Understanding the distinct clinical and accessibility benefits and limitations among available MOUD is essential for prescribing clinicians. Accessing treatment is limited by federal regulation, stigma, and the existence of health care deserts that limit access to necessary care for patients with OUD. Newer harm-reduction models, such as low-threshold prescribing and mobile OTPs, represent progress, but many patients remain untreated.
a At buprenorphine.samhsa.gov/forms/select-practitioner-type.php
b Sold under the brand name Sublocade.
CORRESPONDENCE
Jennie B. Jarrett, PharmD, MMedEd, Department of Pharmacy Practice, University of Illinois Chicago College of Pharmacy, 833 South Wood Street (MC 886), Chicago, IL 60612; jarrett8@uic.edu
1. Baser O, Chalk M, Fiellin DA, et al. Cost and utilization outcomes of opioid-dependence treatments. Am J Manag Care. 2011;17(suppl 8):S235-S248.
2. Gibson A, Degenhardt L, Mattick RP, et al. Exposure to opioid maintenance treatment reduces long-term mortality. Addiction. 2008;103:462-468. doi: 10.1111/j.1360-0443.2007.02090.x
3. Substance Abuse and Mental Health Services Administration. Key Substance Use and Mental Health Indicators in the United States: Results From the 2020 National Survey on Drug Use and Health. HHS Publication PEP21-07-01-003, NSDUH Series H-56. 2021. Accessed March 19, 2023. www.samhsa.gov/data/sites/default/files/reports/rpt35325/NSDUHFFRPDFWHTMLFiles2020/2020NSDUHFFR1PDFW102121.pdf
4. Haffajee RL, Andraka-Christou B, Attermann J, et al. A mixed-method comparison of physician-reported beliefs about and barriers to treatment with medications for opioid use disorder. Subst Abuse Treat Prev Policy. 2020;15:69. doi: 10.1186/s13011-020-00312-3
5. Kosten TR, George TP. The neurobiology of opioid dependence: implications for treatment. Sci Pract Perspect. 2002;1:13-20. doi: 10.1151/spp021113
6. Koob GF. Neurobiology of opioid addiction: opponent process, hyperkatifeia, and negative reinforcement. Biol Psychiatry. 2020;87:44-53. doi: 10.1016/j.biopsych.2019.05.023
7. Substance Abuse and Mental Health Services Administration. Medications for Opioid Use Disorder. For Health care and Addiction Professionals, Policymakers, Patients, and Families. Treatment Improvement Protocol TIP 63. Publication No. PEP21-02-01-002. 2021. Accessed March 19, 2023. https://store.samhsa.gov/sites/default/files/pep21-02-01-002.pdf
8. Sordo L, Barrio G, Bravo MJ, et al. Mortality risk during and after opioid substitution treatment: systematic review and meta-analysis of cohort studies. BMJ. 2017;357:j1550. doi: 10.1136/bmj.j1550
9. Korownyk C, Perry D, Ton J, et al. Opioid use disorder in primary care: PEER umbrella systematic review of systematic reviews. Can Fam Physician. 2019;65:e194-e206.
10. Mattick RP, Breen C, Kimber J, et al. Methadone maintenance therapy versus no opioid replacement therapy for opioid dependence. Cochrane Database Syst Rev. 2009;(3):CD002209. doi: 10.1002/14651858.CD002209.pub2
11. Mattick RP, Breen C, Kimber J, et al. Buprenorphine maintenance versus placebo or methadone maintenance for opioid dependence. Cochrane Database Syst Rev. 2014;(2):CD002207. doi: 10.1002/14651858.CD002207.pub4
12. Krupitsky E, Nunes EV, Ling W, et al. Injectable extended-release naltrexone for opioid dependence: a double-blind, placebo-controlled, multicentre randomised trial. Lancet. 2011;377:1506-1513. doi: 10.1016/S0140-6736(11)60358-9
13. Soyka M, Zingg C, Koller G, et al. Retention rate and substance use in methadone and buprenorphine maintenance therapy and predictors of outcome: results from a randomized study. Int J Neuropsychopharmacol. 2008;11:641-653. doi: 10.1017/S146114570700836X
14. Institute of Medicine Committee on Federal Regulation of Methadone Treatment; Rettig R, Yarmolinsky A, eds. Federal Regulation of Methadone Treatment. National Academies Press; 1995.
15. 42 eCFR §8. Medication assisted treatment for opioid use disorders. Revised March 15, 2023. Accessed March 23, 2023. www.ecfr.gov/current/title-42/chapter-I/subchapter-A/part-8?toc=1
16. Faggiano F, Vigna-Taglianti F, Versino E, et al. Methadone maintenance at different dosages for opioid dependence. Cochrane Database Syst Rev. 2003;(3):CD002208. doi: 10.1002/14651858.CD002208
17. Baxter LE Sr, Campbell A, Deshields M, et al. Safe methadone induction and stabilization: report of an expert panel. J Addict Med. 2013;7:377-386. doi: 10.1097/01.ADM.0000435321.39251.d7
18. Olfson M, Zhang VS, Schoenbaum M, et al. Trends in buprenorphine treatment in the United States, 2009-2018. JAMA. 2020;323:276-277. doi: 10.1001/jama.2019.18913
19. Walsh SL, Preston KL, Stitzer ML, et al. Clinical pharmacology of buprenorphine: ceiling effects at high doses. Clin Pharmacol Ther. 1994;55:569-580. doi: 10.1038/clpt.1994.71
20. Walley AY, Palmisano J, Sorensen-Alawad A, et al. Engagement and substance dependence in a primary care-based addiction treatment program for people infected with HIV and people at high-risk for HIV infection. J Subst Abuse Treat. 2015;59:59-66. doi: 10.1016/j.jsat.2015.07.007
21. Lagisetty P, Klasa K, Bush C, et al. Primary care models for treating opioid use disorders: what actually works? A systematic review. PloS One. 2017;12:e0186315. doi: 10.1371/journal.pone.0186315
22. Du CX, Shi J, Tetrault JM, et al. Primary care and medication management characteristics among patients receiving office-based opioid treatment with buprenorphine. Fam Pract. 2022;39:234-240. doi: 10.1093/fampra/cmab166
23. Herring AA, Vosooghi AA, Luftig J, et al. High-dose buprenorphine induction in the emergency department for treatment of opioid use disorder. JAMA Netw Open. 2021;4:e2117128. doi: 10.1001/jamanetworkopen.2021.17128
24. Hämmig R, Kemter A, Strasser J, et al. Use of microdoses for induction of buprenorphine treatment with overlapping full opioid agonist use: the Bernese method. Subst Abuse Rehabil. 2016;7:99-105. doi: 10.2147/SAR.S109919
25. Snyder H, Kalmin MM, Moulin A, et al. Rapid adoption of low-threshold buprenorphine treatment at California emergency departments participating in the CA Bridge Program. Ann Emerg Med. 2021;78:759-772. doi: 10.1016/j.annemergmed.2021.05.024
26. Wong JSH, Nikoo M, Westenberg JN, et al. Comparing rapid micro-induction and standard induction of buprenorphine/naloxone for treatment of opioid use disorder: protocol for an open-label, parallel-group, superiority, randomized controlled trial. Addict Sci Clin Pract. 2021;16:11. doi: 10.1186/s13722-021-00220-2
27. Lee JD, Vocci F, Fiellin DA. Unobserved “home” induction onto buprenorphine. J Addict Med. 2014;8:299-308. doi: 10.1097/ADM.0000000000000059
28. Krupitsky E, Zvartau E, Blokhina E, et al. Randomized trial of long-acting sustained-release naltrexone implant vs oral naltrexone or placebo for preventing relapse to opioid dependence. Arch Gen Psychiatry. 2012;69:973-981. doi: 10.1001/archgenpsychiatry.2012.1a
29. Wolfe D, Carrieri MP, Dasgupta N, et al. Concerns about injectable naltrexone for opioid dependence. Lancet. 2011;377:1468-1470. doi: 10.1016/S0140-6736(10)62056-9
30. Tanum L, Solli KK, Latif ZEH, et al. Effectiveness of injectable extended-release naltrexone vs daily buprenorphine–naloxone for opioid dependence: a randomized clinical noninferiority trial. JAMA Psychiatry. 2017;74:1197-1205. doi: 10.1001/jamapsychiatry.2017.3206
31. Murphy SM, Polsky D, Lee JD, et al. Cost-effectiveness of extended release naltrexone to prevent relapse among criminal justice-involved individuals with a history of opioid use disorder. Addiction. 2017;112:1440-1450. doi: 10.1111/add.13807
32. Ferrari A, Coccia CPR, Bertolini A, et al. Methadone—metabolism, pharmacokinetics and interactions. Pharmacol Res. 2004;50:551-559. doi: 10.1016/j.phrs.2004.05.002
33. 42 eCFR Part 2. Confidentiality of substance use disorder patient records. January 18, 2017. Accessed March 23, 2023. www.ecfr.gov/current/title-42/chapter-I/subchapter-A/part-2
34. Kao DP, Haigney MCP, Mehler PS, et al. Arrhythmia associated with buprenorphine and methadone reported to the Food and Drug Administration. Addiction. 2015;110:1468-1475. doi: 10.1111/add.13013
35. Tisdale JE, Chung MK, Campbell KB, et al; . Drug-induced arrhythmias: a scientific statement from the American Heart Association. Circulation. 2020;142:e214-e233. doi: 10.1161/CIR.0000000000000905
36. Leshner AI, Mancher M, eds. Barriers to broader use of medications to treat opioid use disorder. In: Medications for Opioid Use Disorder Save Lives. National Academies Press; 2019:109-136.
37. Chilcoat HD, Amick HR, Sherwood MR, et al. Buprenorphine in the United States: Motives for abuse, misuse, and diversion. J Subst Abuse Treat. 2019;104:148-157. doi: 10.1016/j.jsat. 2019.07.005
38. Qato DM, Daviglus ML, Wilder J, et al. “Pharmacy deserts” are prevalent in Chicago’s predominantly minority communities, raising medication access concerns. Health Aff (Millwood). 2014;33:1958-1965. doi: 10.1377/hlthaff.2013.1397
39. Mason M, Soliman R, Kim HS, et al. Disparities by sex and race and ethnicity in death rates due to opioid overdose among adults 55 years or older, 1999 to 2019. JAMA Netw Open. 2022;5:e2142982. doi: 10.1001/jamanetworkopen.2021.42982
40. Rosenblum A, Cleland CM, Fong C, et al. Distance traveled and cross-state commuting to opioid treatment programs in the United States. J Environ Public Health. 2011;2011:948789. doi: 10.1155/2011/948789
41. Chan B, Hoffman KA, Bougatsos C, et al. Mobile methadone medication units: a brief history, scoping review and research opportunity. J Subst Abuse Treat. 2021;129:108483. doi: 10.1016/j.jsat.2021.108483
42. Jakubowski A, Fox A. Defining low-threshold buprenorphine treatment. J Addict Med. 2020;14:95-98. doi: 10.1097/ADM.0000000000000555
43. Messmer SE, Elmes AT, Jimenez AD, et al. Outcomes of a mobile medical unit for low-threshold buprenorphine access targeting opioid overdose hot spots in Chicago. J Subst Use Addict Treat. 2023;209054. doi: 10.1016/j.josat.2023.209054
Medication-assisted recovery (MAR)—the preferred terminology for the service formerly known as medication-assisted treatment—entails a comprehensive set of interventions for managing opioid use disorder (OUD), including medications for opioid use disorder (MOUD). Despite the benefits of MAR—reducing opioid use, opioid-related mortality, and health care costs1-3—only 11% of patients with a diagnosis of OUD received MOUD in 2020.3
Primary care physicians, including family physicians, are well positioned to provide MAR across the patient’s lifespan. However, many family medicine clinicians do not possess the logistical knowledge or resources to implement this service.4 In this article, we describe options for, and barriers to, MAR and societal issues that have an impact on the care of these patients.
Pathophysiology of OUD
Opioids relieve pain by stimulating μ-opioid receptors and activating the brain’s reward system. These pleasurable effects motivate repeated use.5 Frequent opioid exposure causes neuroadaptation, tolerance, and dependence. For patients with OUD who are misusing illicit or prescription opioids, periods of abstinence following neuroadaptation lead to withdrawal symptoms that vary in intensity, depending on the drug, dose, and duration of use. Upregulated noradrenergic tone and dopamine deficiency manifest as numerous signs and symptoms of withdrawal, including5:
- Physiologic: secretory (diaphoresis, rhinorrhea, lacrimation, vomiting, diarrhea) and stimulatory (mydriasis, piloerection, hypertension, tachycardia, insomnia)
- Psychological: pain, cravings, dysphoria, anxiety.
A single episode of opioid withdrawal is not directly life-threatening, but untreated episodes can progressively amplify negative feedback and reinforce continued opioid use.6 Left untreated, withdrawal can be terminal.

Medication-assisted recovery: Effective intervention
MAR services that integrate medical, behavioral, and psychosocial programs can reduce mortality from OUD 2-fold.7,8 A meta-analysis found that, when MAR services are rendered in primary care, treatment retention improves by 25% (number needed to treat [NNT] = 6) and ongoing illicit opioid use is reduced by 50% (NNT = 6), relative to care at a specialty clinic9—highlighting a role for family medicine clinicians in treating OUD.
All 3 US Food and Drug Administration (FDA)–approved MOUD (methadone, buprenorphine, and naltrexone) reduce cravings; 2 (methadone and buprenorphine) mitigate withdrawal symptoms by activating the μ-opioid receptor; and naltrexone diminishes the reinforcing effects of use (TABLE10-12). It is crucial to recognize the pharmacologic distinctions among MOUD because untreated withdrawal syndromes increase dropout from treatment programs and subsequent relapse.13

The Hx of medication-assisted recovery
To understand the landscape of MAR, it is important to understand the history of opioid treatment in the United States. In 1966, Congress passed the Narcotic Addiction Rehabilitation Act (NARA), which secured federal assistance by which state and local governments could develop drug treatment programs.14 NARA permitted legal offenders with OUD to be civilly committed to treatment programs, rather than prosecuted. However, limited resources and a burgeoning population led, instead, to low-cost outpatient programs saddled by strict requirements that lacked a basis for improving clinical outcomes.
Continue to: At the time NARA...
At the time NARA was passed by Congress, OUD was viewed—inaccurately—as a criminal problem, not a medical one. Subsequent legislation was crafted through that lens, which has placed a heavy burden on patients until today.14 Although medical understanding of OUD has advanced tremendously over the past 50 years, treatment remains siloed from mainstream medicine, even in primary care.
There is no one-size-fits-all approach to MAR, and relapse is common. Patient-specific factors and the availability of resources should be considered when designing the most individualized, advantageous plan for MAR.
Methadone
Background. Methadone has the most extensive history for treating OUD and consistently has demonstrated efficacy.13 A meta-analysis of randomized controlled trials comparing methadone to nonpharmacotherapy alone found that methadone improved treatment retention by an absolute 57% (NNT = 2).10
Methadone was approved by the FDA for detoxification and maintenance treatment in the early 1970s, although the Narcotic Addict Treatment Act (NATA) of 1974 restricted dispensing of maintenance treatment to highly regulated clinics known as opioid treatment programs (OTPs).14 NATA required the treating physician to register with the US Drug Enforcement Agency (DEA) and to comply with conservative dosing regimens and observed dosing.
Over time, regulations evolved to give the physician greater flexibility in developing a care plan, allowing “take-home” doses, and improving patients’ access to care. Although access to methadone for the treatment of OUD remains limited to federally certified OTPs, regulations facilitate incorporation of a whole-person approach to care, including counseling, individual and group therapy, and toxicology testing.7
Continue to: Clinical considerations
Clinical considerations. Methadone requires slow titration. For patients starting methadone as an outpatient, federal law15 limits the initial dose to 30 mg and requires physician documentation when the first-day total dosage exceeds 40 mg. This dosing constraint makes it challenging to provide care because a daily dosage ≥ 60 mg has been found to produce, first, higher program retention (relative risk = 1.36; 95% CI, 1.13-1.63) and, second, greater reduction in illicit opioid use (relative risk = 1.59; 95% CI, 1.16-2.18) than is seen in patients who receive a lower daily dosage.16
Due to a prolonged elimination half-life, methadone reaches steady-state in 3 to 5 days. Patients and their families should be educated that withdrawal symptoms might not feel fully managed in the first few days of therapy and that time is required to experience safely the regimen’s full effects.
Aggressive dose-titration during methadone induction can result in drug accumulation and respiratory depression. The risk for methadone-related mortality is highest in the first 2 weeks of therapy, mostly related to overdose potential if the drug is combined with other opioids.17
Buprenorphine
Background. The prescribing rate for buprenorphine, particularly in primary care, is accelerating.18 A meta-analysis of randomized controlled trials found that11:
- compared to placebo, buprenorphine, at any dosage, improves treatment retention by an absolute 21% to 28% (NNT = 4-5)
- patients receiving high-dose buprenorphine (≥ 16 mg/d) had fewer evident cases of illicit opioid use.
Unlike methadone, buprenorphine exerts partial agonism at the μ-opioid receptor, resulting in a so-called ceiling effect that significantly reduces the adverse effect profile, including respiratory depression and euphoria, relative to a full-agonist opioid, such as methadone.19
Continue to: Whereas accessing methadone...
Whereas accessing methadone is limited to OTPs, buprenorphine is available for office-based treatment. By hosting OUD treatment and primary care in the same place, primary care physicians can provide comprehensive medical care including and beyond OUD, thereby improving retention and managing comorbidity.20
Integrated models involving support staff—eg, nurses, behavioral health providers, and pharmacists—have produced the greatest success with office-based treatment models.21 Office-based treatment normalizes OUD as a chronic disease managed by the primary care physician, enabling concurrent harm-reduction strategies; medication reconciliation; and convenient, regular prescribing intervals (eg, every 30 days).22 Nevertheless, access to buprenorphine is limited. Because buprenorphine is a controlled substance, the Ryan Haight Online Pharmacy Consumer Protection Act of 2008 prevents initial prescribing of buprenorphine without in-person evaluation. Telehealth consultations increased access to buprenorphine through temporary exceptions during the COVID-19 pandemic. However, revised rules and regulations for telehealth visits for these controlled substances are forthcoming from the DEA as temporary exceptions for telehealth consultations come to an end. Additionally, prescribing buprenorphine for OUD requires that the treating physician undergo specific training and obtain qualifications, which have evolved over time through federal legislation.
The Drug Addiction Treatment Act of 2000 (DATA 2000) authorized what is known as an X-waiver, which allows physicians to prescribe controlled substances for office-based treatment of OUD, provided that:
- they are registered to do so with the Substance Abuse and Mental Health Services Administration and the DEA
- they have had subspecialty training in addiction or completed an 8-hour training course
- they are able to refer patients to appropriate counseling and ancillary services.
DATA 2000 restricted patient panel sizes to 30 patients in the first year, expanding thereafter upon appropriate certification.
The Comprehensive Addiction and Recovery Act of 2016 (CARA) and the Substance Use Disorder Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities Act of 2018 (the SUPPORT Act) collectively extended prescribing authority for MOUD to other qualifying practitioners (eg, advanced practice clinicians). Despite these attempts to expand access to services, the overdose death rate has continued to increase.
Continue to: To further expand access to MAR...
To further expand access to MAR, the US Department of Health and Human Services updated its practice guidelines in April 2021, allowing clinicians to bypass X-waiver training requirements by applying for a notification-of-intent (NOI) buprenorphine waiver.a However, clinicians are still limited to prescribing buprenorphine for 30 patients at a time. Clinicians who undergo complete X-waiver training may prescribe for 100 patients in the first year and, if eligible, 275 patients thereafter.
In addition, as a component of the Consolidation Appropriations Act of 2023, Congress passed the Mainstreaming Addiction Treatment Act of 2021, or MAT 2021, and Medication Access and Training Expansion Act of 2021, or MATE 2021. MAT eliminated the X-waiver, NOI, and restrictions on the number of patients for whom a provider could prescribe buprenorphine, under federal authority; however, restrictions within one’s state might limit the ability to prescribe buprenorphine. MATE 2021 is an educational requirement for licensing by the DEA (at application and renewal) that will require prescribers to complete 8 hours of training in substance use disorders starting in June 2023.
Use of the monthly injectable extended-release buprenorphine productb is limited by an FDA Risk Evaluation and Mitigation Strategy (REMS) program, which requires specialized training and certification by the prescriber, distributor, and administering clinician. REMS reduces buprenorphine accessibility due to time, cost, and regulatory barriers; although such restrictions have been instituted with the patient’s safety in mind, any limitation to buprenorphine prescribing, apart from controlled substance licensure, serves only to limit access to a primary component of MAR.
Clinical considerations. Due to the competitive nature of buprenorphine and its high affinity for the μ-opioid receptor, the drug can displace other opioid agonists and precipitate acute withdrawal. The withdrawal experience can thereby condition fear and disfavor toward buprenorphine among patients.
It is vital, therefore, that (1) patients’ expectations for treatment be managed appropriately and (2) the treating physician be prepared to provide additional buprenorphine for adequate maintenance doses and utilize adjunct comfort agents (clonidine, nonsteroidal anti-inflammatory drugs, ondansetron) to manage acute withdrawal symptoms. Newer buprenorphine dosing strategies, such as micro-induction and macro-induction, have emerged to curtail these risks.23,24 This is an evolving area of MAR; newer low-threshold initiation strategies25 (see “Low-threshold MOUD prescribing models,” in the text that follows) and evidence that supports micro-induction26 might eliminate the practice of requiring active withdrawal for treatment.
Continue to: Regardless of the strategy...
Regardless of the strategy for dosing buprenorphine, it’s critical that patients be educated on how to initiate treatment outside a clinical setting, such as at home, where they occupy a familiar haven during a potentially uncomfortable time and can be as effective at initiation as they would be in a clinical setting, with no difference in precipitation of adverse effects.
At-home induction might be more appropriate for patients who are not yet in significant enough withdrawal while in the physician's office.27 Guidance should be provided on dosing instructions, self-assessment of withdrawal symptoms, and, if applicable, patience with the slow-dissolving sublingual tablet or film formulation.
Naltrexone
Background. Naltrexone is available as an oral tablet and an extended-release, once-monthly intramuscular injection; the latter has demonstrated superiority in MAR.28 Oral naltrexone has limited supporting evidence, is inferior to other MOUD options, and should not be used to treat OUD.7 Altogether, approval of naltrexone for OUD is controversial, due to potentially unethical trials and approval processes,29 although a multicenter randomized controlled trial demonstrated the drug’s noninferiority with respect to treatment retention relative to buprenorphine.30 Used over time, naltrexone does not relieve withdrawal symptoms but can reduce cravings.
Clinical considerations. There are numerous clinical barriers that limit the use of naltrexone.
First, patients should be abstinent from opioids for 7 to 14 days prior to starting therapy; usually, this means undergoing medically supervised withdrawal in a controlled environment. This is an obvious limitation for patients who are constrained financially—those who lack, or have inadequate, health insurance or are unable to be away from their job for an extended time.
Continue to: Second, because naltrexone...
Second, because naltrexone does not address withdrawal symptoms, supportive therapies should be incorporated into the treatment plan, including:
- clonidine for hyperadrenergic symptoms (anxiety, diaphoresis, hypertension)
- nonopioid analgesics for pain
- antiemetics, such as ondansetron and metoclopramide, for nausea or vomiting
- loperamide for diarrhea
- diphenhydramine for insomnia.
Third, patients taking naltrexone have a diminished response to opioids. This complicates pain management in the event of an emergent surgical procedure.
Last, when naltrexone wears off, patients are effectively opioid-naïve, which increases the risk for overdose in those who stop therapy abruptly.29 The increased risk for overdose should be communicated to all patients with OUD who are being treated with naltrexone.
This nonopioid option is appealing to policymakers and is often prioritized in the criminal justice system; however, the decreased efficacy of naltrexone (compared to methadone and buprenorphine), potential for overdose, and challenges in initiating treatment are concerning and limit the drug’s use in many real-world settings.
Because naltrexone is not a controlled substance, regulations regarding maintaining inventory and distribution are more flexible.
Continue to: Overall, the cost-effectiveness...
Overall, the cost-effectiveness of intramuscular naltrexone is unclear. State-administered insurance programs vary in their requirements for coverage of naltrexone treatment.31
Comprehensive medication reconciliation is vital
Overall fragmentation of care within OTPs places patients at risk for adverse events, such as drug interactions.32 Under Title 42 of the US Code,33 patients must provide written consent for an OTP provider to disclose their history of a substance use disorder. Allowing the patient to decide which medical providers can access their treatment records for an OUD benefits patient confidentiality but poses numerous issues worth exploring.
All prescribed controlled substances are recorded in the prescription drug monitoring program, or PDMP, a state-level electronic database accessible to health care professionals to inform prescribing decisions and identify drug interactions. The PDMP has substantially reduced opioid overprescribing and improved identification of patients at risk for overdose or misuse of opioids.
Unlike all other controlled substances, however, prescriptions ordered by an OTP are not recorded in the PDMP (although there are recent exceptions to this scenario). Without such information, a physician might not have important information about the patient when making medical decisions—placing the patient at risk for harmful outcomes, such as drug–drug and drug–disease interactions.
For example: Methadone is associated with a prolonged QT interval,34 increasing the risk for a fatal arrhythmia. Concurrent QT-prolonging medications, such as azithromycin and citalopram, further increase this risk.35 Because methadone dispensing is isolated from the patient’s medical record, the clinician who prescribes MOUD has an incomplete patient history and could make a potentially fatal treatment decision.
Continue to: Diversion is unlikely
Diversion is unlikely
Health care providers often express concern about diversion in MOUD. However, misuse and diversion rates of methadone and buprenorphine have declined steadily since 2011, and, in fact, are actually lower than the diversion rate of prescription antibiotics.36
Regardless, diversion of buprenorphine should not be a concern for physicians prescribing MOUD. Although a prescriber might worry about manipulation of the formulation of buprenorphine for intravenous administration, addition of naloxone to buprenorphine in tablet form diminishes the potential for overdose. Additionally, the ceiling effect of buprenorphine limits the likelihood of significant respiratory depression and euphoria.
Should buprenorphine reach a patient for whom it was not prescribed, it is highly unlikely that an overdose would result. Rather, the medication would protect against the effects of illicit opioids and relieve withdrawal symptoms. Most people with OUD who have misused buprenorphine have done so to relieve withdrawal symptoms,37 not to experience intoxication.
Health care deserts
So-called health care deserts in parts of the United States are an ongoing problem that disproportionately affects lower-income and segregated Black and Hispanic communities38—communities that shoulder the highest burden of OUD and OUD-related mortality39 and whose populace is in greatest need of MAR. Even when health care is accessible in such a desert, some clinicians and pharmacies refuse to prescribe or dispense MOUD because of the accompanying stigma of OUD.
A MAR desert, like a pharmacy desert, is a geographic region—one without access to a MAR or an OTP provider, thereby preventing patients from reaching appropriate care; for some patients, having to travel to the nearest provider can render treatment inaccessible.40
Continue to: Efforts are in place to identify...
Efforts are in place to identify areas at greatest need of OUD-related medical services, such as heat maps that identify areas of increased utilization of emergency medical services for opioid overdose. State-run programs have been implemented to increase access, such as the Illinois Helpline (https://helplineil.org) that provides support and resources for patients, friends, family, and providers.
Novel solutions
Key strategies to increase access to care and slow the opioid epidemic include low-threshold prescribing of MOUD and mobile OTPs.41
Low-threshold MOUD prescribing models. Adoption of one of these models in a medical practice that provides MAR might increase absolute enrollment. A low-threshold prescribing model involves42:
- same-day treatment
- leniency with respect to abstinence periods and a concomitant substance use disorder
- enhanced accessibility to MOUD through nontraditional medical settings.
Low-threshold prescribing is flexible in regard to patients’ needs and bypasses many of the barriers discussed in this article. Impressive multicenter success has been achieved by the CA Bridge program in California (https://cabridge.org), including an increase in recognition of OUD, treatment initiations, and outpatient engagement.25
The cost-effectiveness of low-threshold MOUD prescribing programs remains to be determined.
Mobile OTPs. In July 2021, the DEA authorized a mobile component to existing OTP registrants that is permitted to dispense methadone and buprenorphine. Mobile units are physically separate from the OTP but have similar functions, depending on available space. Services that cannot be provided on the mobile unit of an OTP must be available at its brick-and-mortar location.7 Logistically, OTP registrants no longer need a separate registration to implement a mobile unit, thus expanding care to patients in underserved or remote areas who often encounter barriers to access.43
Conclusion
Understanding the distinct clinical and accessibility benefits and limitations among available MOUD is essential for prescribing clinicians. Accessing treatment is limited by federal regulation, stigma, and the existence of health care deserts that limit access to necessary care for patients with OUD. Newer harm-reduction models, such as low-threshold prescribing and mobile OTPs, represent progress, but many patients remain untreated.
a At buprenorphine.samhsa.gov/forms/select-practitioner-type.php
b Sold under the brand name Sublocade.
CORRESPONDENCE
Jennie B. Jarrett, PharmD, MMedEd, Department of Pharmacy Practice, University of Illinois Chicago College of Pharmacy, 833 South Wood Street (MC 886), Chicago, IL 60612; jarrett8@uic.edu
Medication-assisted recovery (MAR)—the preferred terminology for the service formerly known as medication-assisted treatment—entails a comprehensive set of interventions for managing opioid use disorder (OUD), including medications for opioid use disorder (MOUD). Despite the benefits of MAR—reducing opioid use, opioid-related mortality, and health care costs1-3—only 11% of patients with a diagnosis of OUD received MOUD in 2020.3
Primary care physicians, including family physicians, are well positioned to provide MAR across the patient’s lifespan. However, many family medicine clinicians do not possess the logistical knowledge or resources to implement this service.4 In this article, we describe options for, and barriers to, MAR and societal issues that have an impact on the care of these patients.
Pathophysiology of OUD
Opioids relieve pain by stimulating μ-opioid receptors and activating the brain’s reward system. These pleasurable effects motivate repeated use.5 Frequent opioid exposure causes neuroadaptation, tolerance, and dependence. For patients with OUD who are misusing illicit or prescription opioids, periods of abstinence following neuroadaptation lead to withdrawal symptoms that vary in intensity, depending on the drug, dose, and duration of use. Upregulated noradrenergic tone and dopamine deficiency manifest as numerous signs and symptoms of withdrawal, including5:
- Physiologic: secretory (diaphoresis, rhinorrhea, lacrimation, vomiting, diarrhea) and stimulatory (mydriasis, piloerection, hypertension, tachycardia, insomnia)
- Psychological: pain, cravings, dysphoria, anxiety.
A single episode of opioid withdrawal is not directly life-threatening, but untreated episodes can progressively amplify negative feedback and reinforce continued opioid use.6 Left untreated, withdrawal can be terminal.

Medication-assisted recovery: Effective intervention
MAR services that integrate medical, behavioral, and psychosocial programs can reduce mortality from OUD 2-fold.7,8 A meta-analysis found that, when MAR services are rendered in primary care, treatment retention improves by 25% (number needed to treat [NNT] = 6) and ongoing illicit opioid use is reduced by 50% (NNT = 6), relative to care at a specialty clinic9—highlighting a role for family medicine clinicians in treating OUD.
All 3 US Food and Drug Administration (FDA)–approved MOUD (methadone, buprenorphine, and naltrexone) reduce cravings; 2 (methadone and buprenorphine) mitigate withdrawal symptoms by activating the μ-opioid receptor; and naltrexone diminishes the reinforcing effects of use (TABLE10-12). It is crucial to recognize the pharmacologic distinctions among MOUD because untreated withdrawal syndromes increase dropout from treatment programs and subsequent relapse.13

The Hx of medication-assisted recovery
To understand the landscape of MAR, it is important to understand the history of opioid treatment in the United States. In 1966, Congress passed the Narcotic Addiction Rehabilitation Act (NARA), which secured federal assistance by which state and local governments could develop drug treatment programs.14 NARA permitted legal offenders with OUD to be civilly committed to treatment programs, rather than prosecuted. However, limited resources and a burgeoning population led, instead, to low-cost outpatient programs saddled by strict requirements that lacked a basis for improving clinical outcomes.
Continue to: At the time NARA...
At the time NARA was passed by Congress, OUD was viewed—inaccurately—as a criminal problem, not a medical one. Subsequent legislation was crafted through that lens, which has placed a heavy burden on patients until today.14 Although medical understanding of OUD has advanced tremendously over the past 50 years, treatment remains siloed from mainstream medicine, even in primary care.
There is no one-size-fits-all approach to MAR, and relapse is common. Patient-specific factors and the availability of resources should be considered when designing the most individualized, advantageous plan for MAR.
Methadone
Background. Methadone has the most extensive history for treating OUD and consistently has demonstrated efficacy.13 A meta-analysis of randomized controlled trials comparing methadone to nonpharmacotherapy alone found that methadone improved treatment retention by an absolute 57% (NNT = 2).10
Methadone was approved by the FDA for detoxification and maintenance treatment in the early 1970s, although the Narcotic Addict Treatment Act (NATA) of 1974 restricted dispensing of maintenance treatment to highly regulated clinics known as opioid treatment programs (OTPs).14 NATA required the treating physician to register with the US Drug Enforcement Agency (DEA) and to comply with conservative dosing regimens and observed dosing.
Over time, regulations evolved to give the physician greater flexibility in developing a care plan, allowing “take-home” doses, and improving patients’ access to care. Although access to methadone for the treatment of OUD remains limited to federally certified OTPs, regulations facilitate incorporation of a whole-person approach to care, including counseling, individual and group therapy, and toxicology testing.7
Continue to: Clinical considerations
Clinical considerations. Methadone requires slow titration. For patients starting methadone as an outpatient, federal law15 limits the initial dose to 30 mg and requires physician documentation when the first-day total dosage exceeds 40 mg. This dosing constraint makes it challenging to provide care because a daily dosage ≥ 60 mg has been found to produce, first, higher program retention (relative risk = 1.36; 95% CI, 1.13-1.63) and, second, greater reduction in illicit opioid use (relative risk = 1.59; 95% CI, 1.16-2.18) than is seen in patients who receive a lower daily dosage.16
Due to a prolonged elimination half-life, methadone reaches steady-state in 3 to 5 days. Patients and their families should be educated that withdrawal symptoms might not feel fully managed in the first few days of therapy and that time is required to experience safely the regimen’s full effects.
Aggressive dose-titration during methadone induction can result in drug accumulation and respiratory depression. The risk for methadone-related mortality is highest in the first 2 weeks of therapy, mostly related to overdose potential if the drug is combined with other opioids.17
Buprenorphine
Background. The prescribing rate for buprenorphine, particularly in primary care, is accelerating.18 A meta-analysis of randomized controlled trials found that11:
- compared to placebo, buprenorphine, at any dosage, improves treatment retention by an absolute 21% to 28% (NNT = 4-5)
- patients receiving high-dose buprenorphine (≥ 16 mg/d) had fewer evident cases of illicit opioid use.
Unlike methadone, buprenorphine exerts partial agonism at the μ-opioid receptor, resulting in a so-called ceiling effect that significantly reduces the adverse effect profile, including respiratory depression and euphoria, relative to a full-agonist opioid, such as methadone.19
Continue to: Whereas accessing methadone...
Whereas accessing methadone is limited to OTPs, buprenorphine is available for office-based treatment. By hosting OUD treatment and primary care in the same place, primary care physicians can provide comprehensive medical care including and beyond OUD, thereby improving retention and managing comorbidity.20
Integrated models involving support staff—eg, nurses, behavioral health providers, and pharmacists—have produced the greatest success with office-based treatment models.21 Office-based treatment normalizes OUD as a chronic disease managed by the primary care physician, enabling concurrent harm-reduction strategies; medication reconciliation; and convenient, regular prescribing intervals (eg, every 30 days).22 Nevertheless, access to buprenorphine is limited. Because buprenorphine is a controlled substance, the Ryan Haight Online Pharmacy Consumer Protection Act of 2008 prevents initial prescribing of buprenorphine without in-person evaluation. Telehealth consultations increased access to buprenorphine through temporary exceptions during the COVID-19 pandemic. However, revised rules and regulations for telehealth visits for these controlled substances are forthcoming from the DEA as temporary exceptions for telehealth consultations come to an end. Additionally, prescribing buprenorphine for OUD requires that the treating physician undergo specific training and obtain qualifications, which have evolved over time through federal legislation.
The Drug Addiction Treatment Act of 2000 (DATA 2000) authorized what is known as an X-waiver, which allows physicians to prescribe controlled substances for office-based treatment of OUD, provided that:
- they are registered to do so with the Substance Abuse and Mental Health Services Administration and the DEA
- they have had subspecialty training in addiction or completed an 8-hour training course
- they are able to refer patients to appropriate counseling and ancillary services.
DATA 2000 restricted patient panel sizes to 30 patients in the first year, expanding thereafter upon appropriate certification.
The Comprehensive Addiction and Recovery Act of 2016 (CARA) and the Substance Use Disorder Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities Act of 2018 (the SUPPORT Act) collectively extended prescribing authority for MOUD to other qualifying practitioners (eg, advanced practice clinicians). Despite these attempts to expand access to services, the overdose death rate has continued to increase.
Continue to: To further expand access to MAR...
To further expand access to MAR, the US Department of Health and Human Services updated its practice guidelines in April 2021, allowing clinicians to bypass X-waiver training requirements by applying for a notification-of-intent (NOI) buprenorphine waiver.a However, clinicians are still limited to prescribing buprenorphine for 30 patients at a time. Clinicians who undergo complete X-waiver training may prescribe for 100 patients in the first year and, if eligible, 275 patients thereafter.
In addition, as a component of the Consolidation Appropriations Act of 2023, Congress passed the Mainstreaming Addiction Treatment Act of 2021, or MAT 2021, and Medication Access and Training Expansion Act of 2021, or MATE 2021. MAT eliminated the X-waiver, NOI, and restrictions on the number of patients for whom a provider could prescribe buprenorphine, under federal authority; however, restrictions within one’s state might limit the ability to prescribe buprenorphine. MATE 2021 is an educational requirement for licensing by the DEA (at application and renewal) that will require prescribers to complete 8 hours of training in substance use disorders starting in June 2023.
Use of the monthly injectable extended-release buprenorphine productb is limited by an FDA Risk Evaluation and Mitigation Strategy (REMS) program, which requires specialized training and certification by the prescriber, distributor, and administering clinician. REMS reduces buprenorphine accessibility due to time, cost, and regulatory barriers; although such restrictions have been instituted with the patient’s safety in mind, any limitation to buprenorphine prescribing, apart from controlled substance licensure, serves only to limit access to a primary component of MAR.
Clinical considerations. Due to the competitive nature of buprenorphine and its high affinity for the μ-opioid receptor, the drug can displace other opioid agonists and precipitate acute withdrawal. The withdrawal experience can thereby condition fear and disfavor toward buprenorphine among patients.
It is vital, therefore, that (1) patients’ expectations for treatment be managed appropriately and (2) the treating physician be prepared to provide additional buprenorphine for adequate maintenance doses and utilize adjunct comfort agents (clonidine, nonsteroidal anti-inflammatory drugs, ondansetron) to manage acute withdrawal symptoms. Newer buprenorphine dosing strategies, such as micro-induction and macro-induction, have emerged to curtail these risks.23,24 This is an evolving area of MAR; newer low-threshold initiation strategies25 (see “Low-threshold MOUD prescribing models,” in the text that follows) and evidence that supports micro-induction26 might eliminate the practice of requiring active withdrawal for treatment.
Continue to: Regardless of the strategy...
Regardless of the strategy for dosing buprenorphine, it’s critical that patients be educated on how to initiate treatment outside a clinical setting, such as at home, where they occupy a familiar haven during a potentially uncomfortable time and can be as effective at initiation as they would be in a clinical setting, with no difference in precipitation of adverse effects.
At-home induction might be more appropriate for patients who are not yet in significant enough withdrawal while in the physician's office.27 Guidance should be provided on dosing instructions, self-assessment of withdrawal symptoms, and, if applicable, patience with the slow-dissolving sublingual tablet or film formulation.
Naltrexone
Background. Naltrexone is available as an oral tablet and an extended-release, once-monthly intramuscular injection; the latter has demonstrated superiority in MAR.28 Oral naltrexone has limited supporting evidence, is inferior to other MOUD options, and should not be used to treat OUD.7 Altogether, approval of naltrexone for OUD is controversial, due to potentially unethical trials and approval processes,29 although a multicenter randomized controlled trial demonstrated the drug’s noninferiority with respect to treatment retention relative to buprenorphine.30 Used over time, naltrexone does not relieve withdrawal symptoms but can reduce cravings.
Clinical considerations. There are numerous clinical barriers that limit the use of naltrexone.
First, patients should be abstinent from opioids for 7 to 14 days prior to starting therapy; usually, this means undergoing medically supervised withdrawal in a controlled environment. This is an obvious limitation for patients who are constrained financially—those who lack, or have inadequate, health insurance or are unable to be away from their job for an extended time.
Continue to: Second, because naltrexone...
Second, because naltrexone does not address withdrawal symptoms, supportive therapies should be incorporated into the treatment plan, including:
- clonidine for hyperadrenergic symptoms (anxiety, diaphoresis, hypertension)
- nonopioid analgesics for pain
- antiemetics, such as ondansetron and metoclopramide, for nausea or vomiting
- loperamide for diarrhea
- diphenhydramine for insomnia.
Third, patients taking naltrexone have a diminished response to opioids. This complicates pain management in the event of an emergent surgical procedure.
Last, when naltrexone wears off, patients are effectively opioid-naïve, which increases the risk for overdose in those who stop therapy abruptly.29 The increased risk for overdose should be communicated to all patients with OUD who are being treated with naltrexone.
This nonopioid option is appealing to policymakers and is often prioritized in the criminal justice system; however, the decreased efficacy of naltrexone (compared to methadone and buprenorphine), potential for overdose, and challenges in initiating treatment are concerning and limit the drug’s use in many real-world settings.
Because naltrexone is not a controlled substance, regulations regarding maintaining inventory and distribution are more flexible.
Continue to: Overall, the cost-effectiveness...
Overall, the cost-effectiveness of intramuscular naltrexone is unclear. State-administered insurance programs vary in their requirements for coverage of naltrexone treatment.31
Comprehensive medication reconciliation is vital
Overall fragmentation of care within OTPs places patients at risk for adverse events, such as drug interactions.32 Under Title 42 of the US Code,33 patients must provide written consent for an OTP provider to disclose their history of a substance use disorder. Allowing the patient to decide which medical providers can access their treatment records for an OUD benefits patient confidentiality but poses numerous issues worth exploring.
All prescribed controlled substances are recorded in the prescription drug monitoring program, or PDMP, a state-level electronic database accessible to health care professionals to inform prescribing decisions and identify drug interactions. The PDMP has substantially reduced opioid overprescribing and improved identification of patients at risk for overdose or misuse of opioids.
Unlike all other controlled substances, however, prescriptions ordered by an OTP are not recorded in the PDMP (although there are recent exceptions to this scenario). Without such information, a physician might not have important information about the patient when making medical decisions—placing the patient at risk for harmful outcomes, such as drug–drug and drug–disease interactions.
For example: Methadone is associated with a prolonged QT interval,34 increasing the risk for a fatal arrhythmia. Concurrent QT-prolonging medications, such as azithromycin and citalopram, further increase this risk.35 Because methadone dispensing is isolated from the patient’s medical record, the clinician who prescribes MOUD has an incomplete patient history and could make a potentially fatal treatment decision.
Continue to: Diversion is unlikely
Diversion is unlikely
Health care providers often express concern about diversion in MOUD. However, misuse and diversion rates of methadone and buprenorphine have declined steadily since 2011, and, in fact, are actually lower than the diversion rate of prescription antibiotics.36
Regardless, diversion of buprenorphine should not be a concern for physicians prescribing MOUD. Although a prescriber might worry about manipulation of the formulation of buprenorphine for intravenous administration, addition of naloxone to buprenorphine in tablet form diminishes the potential for overdose. Additionally, the ceiling effect of buprenorphine limits the likelihood of significant respiratory depression and euphoria.
Should buprenorphine reach a patient for whom it was not prescribed, it is highly unlikely that an overdose would result. Rather, the medication would protect against the effects of illicit opioids and relieve withdrawal symptoms. Most people with OUD who have misused buprenorphine have done so to relieve withdrawal symptoms,37 not to experience intoxication.
Health care deserts
So-called health care deserts in parts of the United States are an ongoing problem that disproportionately affects lower-income and segregated Black and Hispanic communities38—communities that shoulder the highest burden of OUD and OUD-related mortality39 and whose populace is in greatest need of MAR. Even when health care is accessible in such a desert, some clinicians and pharmacies refuse to prescribe or dispense MOUD because of the accompanying stigma of OUD.
A MAR desert, like a pharmacy desert, is a geographic region—one without access to a MAR or an OTP provider, thereby preventing patients from reaching appropriate care; for some patients, having to travel to the nearest provider can render treatment inaccessible.40
Continue to: Efforts are in place to identify...
Efforts are in place to identify areas at greatest need of OUD-related medical services, such as heat maps that identify areas of increased utilization of emergency medical services for opioid overdose. State-run programs have been implemented to increase access, such as the Illinois Helpline (https://helplineil.org) that provides support and resources for patients, friends, family, and providers.
Novel solutions
Key strategies to increase access to care and slow the opioid epidemic include low-threshold prescribing of MOUD and mobile OTPs.41
Low-threshold MOUD prescribing models. Adoption of one of these models in a medical practice that provides MAR might increase absolute enrollment. A low-threshold prescribing model involves42:
- same-day treatment
- leniency with respect to abstinence periods and a concomitant substance use disorder
- enhanced accessibility to MOUD through nontraditional medical settings.
Low-threshold prescribing is flexible in regard to patients’ needs and bypasses many of the barriers discussed in this article. Impressive multicenter success has been achieved by the CA Bridge program in California (https://cabridge.org), including an increase in recognition of OUD, treatment initiations, and outpatient engagement.25
The cost-effectiveness of low-threshold MOUD prescribing programs remains to be determined.
Mobile OTPs. In July 2021, the DEA authorized a mobile component to existing OTP registrants that is permitted to dispense methadone and buprenorphine. Mobile units are physically separate from the OTP but have similar functions, depending on available space. Services that cannot be provided on the mobile unit of an OTP must be available at its brick-and-mortar location.7 Logistically, OTP registrants no longer need a separate registration to implement a mobile unit, thus expanding care to patients in underserved or remote areas who often encounter barriers to access.43
Conclusion
Understanding the distinct clinical and accessibility benefits and limitations among available MOUD is essential for prescribing clinicians. Accessing treatment is limited by federal regulation, stigma, and the existence of health care deserts that limit access to necessary care for patients with OUD. Newer harm-reduction models, such as low-threshold prescribing and mobile OTPs, represent progress, but many patients remain untreated.
a At buprenorphine.samhsa.gov/forms/select-practitioner-type.php
b Sold under the brand name Sublocade.
CORRESPONDENCE
Jennie B. Jarrett, PharmD, MMedEd, Department of Pharmacy Practice, University of Illinois Chicago College of Pharmacy, 833 South Wood Street (MC 886), Chicago, IL 60612; jarrett8@uic.edu
1. Baser O, Chalk M, Fiellin DA, et al. Cost and utilization outcomes of opioid-dependence treatments. Am J Manag Care. 2011;17(suppl 8):S235-S248.
2. Gibson A, Degenhardt L, Mattick RP, et al. Exposure to opioid maintenance treatment reduces long-term mortality. Addiction. 2008;103:462-468. doi: 10.1111/j.1360-0443.2007.02090.x
3. Substance Abuse and Mental Health Services Administration. Key Substance Use and Mental Health Indicators in the United States: Results From the 2020 National Survey on Drug Use and Health. HHS Publication PEP21-07-01-003, NSDUH Series H-56. 2021. Accessed March 19, 2023. www.samhsa.gov/data/sites/default/files/reports/rpt35325/NSDUHFFRPDFWHTMLFiles2020/2020NSDUHFFR1PDFW102121.pdf
4. Haffajee RL, Andraka-Christou B, Attermann J, et al. A mixed-method comparison of physician-reported beliefs about and barriers to treatment with medications for opioid use disorder. Subst Abuse Treat Prev Policy. 2020;15:69. doi: 10.1186/s13011-020-00312-3
5. Kosten TR, George TP. The neurobiology of opioid dependence: implications for treatment. Sci Pract Perspect. 2002;1:13-20. doi: 10.1151/spp021113
6. Koob GF. Neurobiology of opioid addiction: opponent process, hyperkatifeia, and negative reinforcement. Biol Psychiatry. 2020;87:44-53. doi: 10.1016/j.biopsych.2019.05.023
7. Substance Abuse and Mental Health Services Administration. Medications for Opioid Use Disorder. For Health care and Addiction Professionals, Policymakers, Patients, and Families. Treatment Improvement Protocol TIP 63. Publication No. PEP21-02-01-002. 2021. Accessed March 19, 2023. https://store.samhsa.gov/sites/default/files/pep21-02-01-002.pdf
8. Sordo L, Barrio G, Bravo MJ, et al. Mortality risk during and after opioid substitution treatment: systematic review and meta-analysis of cohort studies. BMJ. 2017;357:j1550. doi: 10.1136/bmj.j1550
9. Korownyk C, Perry D, Ton J, et al. Opioid use disorder in primary care: PEER umbrella systematic review of systematic reviews. Can Fam Physician. 2019;65:e194-e206.
10. Mattick RP, Breen C, Kimber J, et al. Methadone maintenance therapy versus no opioid replacement therapy for opioid dependence. Cochrane Database Syst Rev. 2009;(3):CD002209. doi: 10.1002/14651858.CD002209.pub2
11. Mattick RP, Breen C, Kimber J, et al. Buprenorphine maintenance versus placebo or methadone maintenance for opioid dependence. Cochrane Database Syst Rev. 2014;(2):CD002207. doi: 10.1002/14651858.CD002207.pub4
12. Krupitsky E, Nunes EV, Ling W, et al. Injectable extended-release naltrexone for opioid dependence: a double-blind, placebo-controlled, multicentre randomised trial. Lancet. 2011;377:1506-1513. doi: 10.1016/S0140-6736(11)60358-9
13. Soyka M, Zingg C, Koller G, et al. Retention rate and substance use in methadone and buprenorphine maintenance therapy and predictors of outcome: results from a randomized study. Int J Neuropsychopharmacol. 2008;11:641-653. doi: 10.1017/S146114570700836X
14. Institute of Medicine Committee on Federal Regulation of Methadone Treatment; Rettig R, Yarmolinsky A, eds. Federal Regulation of Methadone Treatment. National Academies Press; 1995.
15. 42 eCFR §8. Medication assisted treatment for opioid use disorders. Revised March 15, 2023. Accessed March 23, 2023. www.ecfr.gov/current/title-42/chapter-I/subchapter-A/part-8?toc=1
16. Faggiano F, Vigna-Taglianti F, Versino E, et al. Methadone maintenance at different dosages for opioid dependence. Cochrane Database Syst Rev. 2003;(3):CD002208. doi: 10.1002/14651858.CD002208
17. Baxter LE Sr, Campbell A, Deshields M, et al. Safe methadone induction and stabilization: report of an expert panel. J Addict Med. 2013;7:377-386. doi: 10.1097/01.ADM.0000435321.39251.d7
18. Olfson M, Zhang VS, Schoenbaum M, et al. Trends in buprenorphine treatment in the United States, 2009-2018. JAMA. 2020;323:276-277. doi: 10.1001/jama.2019.18913
19. Walsh SL, Preston KL, Stitzer ML, et al. Clinical pharmacology of buprenorphine: ceiling effects at high doses. Clin Pharmacol Ther. 1994;55:569-580. doi: 10.1038/clpt.1994.71
20. Walley AY, Palmisano J, Sorensen-Alawad A, et al. Engagement and substance dependence in a primary care-based addiction treatment program for people infected with HIV and people at high-risk for HIV infection. J Subst Abuse Treat. 2015;59:59-66. doi: 10.1016/j.jsat.2015.07.007
21. Lagisetty P, Klasa K, Bush C, et al. Primary care models for treating opioid use disorders: what actually works? A systematic review. PloS One. 2017;12:e0186315. doi: 10.1371/journal.pone.0186315
22. Du CX, Shi J, Tetrault JM, et al. Primary care and medication management characteristics among patients receiving office-based opioid treatment with buprenorphine. Fam Pract. 2022;39:234-240. doi: 10.1093/fampra/cmab166
23. Herring AA, Vosooghi AA, Luftig J, et al. High-dose buprenorphine induction in the emergency department for treatment of opioid use disorder. JAMA Netw Open. 2021;4:e2117128. doi: 10.1001/jamanetworkopen.2021.17128
24. Hämmig R, Kemter A, Strasser J, et al. Use of microdoses for induction of buprenorphine treatment with overlapping full opioid agonist use: the Bernese method. Subst Abuse Rehabil. 2016;7:99-105. doi: 10.2147/SAR.S109919
25. Snyder H, Kalmin MM, Moulin A, et al. Rapid adoption of low-threshold buprenorphine treatment at California emergency departments participating in the CA Bridge Program. Ann Emerg Med. 2021;78:759-772. doi: 10.1016/j.annemergmed.2021.05.024
26. Wong JSH, Nikoo M, Westenberg JN, et al. Comparing rapid micro-induction and standard induction of buprenorphine/naloxone for treatment of opioid use disorder: protocol for an open-label, parallel-group, superiority, randomized controlled trial. Addict Sci Clin Pract. 2021;16:11. doi: 10.1186/s13722-021-00220-2
27. Lee JD, Vocci F, Fiellin DA. Unobserved “home” induction onto buprenorphine. J Addict Med. 2014;8:299-308. doi: 10.1097/ADM.0000000000000059
28. Krupitsky E, Zvartau E, Blokhina E, et al. Randomized trial of long-acting sustained-release naltrexone implant vs oral naltrexone or placebo for preventing relapse to opioid dependence. Arch Gen Psychiatry. 2012;69:973-981. doi: 10.1001/archgenpsychiatry.2012.1a
29. Wolfe D, Carrieri MP, Dasgupta N, et al. Concerns about injectable naltrexone for opioid dependence. Lancet. 2011;377:1468-1470. doi: 10.1016/S0140-6736(10)62056-9
30. Tanum L, Solli KK, Latif ZEH, et al. Effectiveness of injectable extended-release naltrexone vs daily buprenorphine–naloxone for opioid dependence: a randomized clinical noninferiority trial. JAMA Psychiatry. 2017;74:1197-1205. doi: 10.1001/jamapsychiatry.2017.3206
31. Murphy SM, Polsky D, Lee JD, et al. Cost-effectiveness of extended release naltrexone to prevent relapse among criminal justice-involved individuals with a history of opioid use disorder. Addiction. 2017;112:1440-1450. doi: 10.1111/add.13807
32. Ferrari A, Coccia CPR, Bertolini A, et al. Methadone—metabolism, pharmacokinetics and interactions. Pharmacol Res. 2004;50:551-559. doi: 10.1016/j.phrs.2004.05.002
33. 42 eCFR Part 2. Confidentiality of substance use disorder patient records. January 18, 2017. Accessed March 23, 2023. www.ecfr.gov/current/title-42/chapter-I/subchapter-A/part-2
34. Kao DP, Haigney MCP, Mehler PS, et al. Arrhythmia associated with buprenorphine and methadone reported to the Food and Drug Administration. Addiction. 2015;110:1468-1475. doi: 10.1111/add.13013
35. Tisdale JE, Chung MK, Campbell KB, et al; . Drug-induced arrhythmias: a scientific statement from the American Heart Association. Circulation. 2020;142:e214-e233. doi: 10.1161/CIR.0000000000000905
36. Leshner AI, Mancher M, eds. Barriers to broader use of medications to treat opioid use disorder. In: Medications for Opioid Use Disorder Save Lives. National Academies Press; 2019:109-136.
37. Chilcoat HD, Amick HR, Sherwood MR, et al. Buprenorphine in the United States: Motives for abuse, misuse, and diversion. J Subst Abuse Treat. 2019;104:148-157. doi: 10.1016/j.jsat. 2019.07.005
38. Qato DM, Daviglus ML, Wilder J, et al. “Pharmacy deserts” are prevalent in Chicago’s predominantly minority communities, raising medication access concerns. Health Aff (Millwood). 2014;33:1958-1965. doi: 10.1377/hlthaff.2013.1397
39. Mason M, Soliman R, Kim HS, et al. Disparities by sex and race and ethnicity in death rates due to opioid overdose among adults 55 years or older, 1999 to 2019. JAMA Netw Open. 2022;5:e2142982. doi: 10.1001/jamanetworkopen.2021.42982
40. Rosenblum A, Cleland CM, Fong C, et al. Distance traveled and cross-state commuting to opioid treatment programs in the United States. J Environ Public Health. 2011;2011:948789. doi: 10.1155/2011/948789
41. Chan B, Hoffman KA, Bougatsos C, et al. Mobile methadone medication units: a brief history, scoping review and research opportunity. J Subst Abuse Treat. 2021;129:108483. doi: 10.1016/j.jsat.2021.108483
42. Jakubowski A, Fox A. Defining low-threshold buprenorphine treatment. J Addict Med. 2020;14:95-98. doi: 10.1097/ADM.0000000000000555
43. Messmer SE, Elmes AT, Jimenez AD, et al. Outcomes of a mobile medical unit for low-threshold buprenorphine access targeting opioid overdose hot spots in Chicago. J Subst Use Addict Treat. 2023;209054. doi: 10.1016/j.josat.2023.209054
1. Baser O, Chalk M, Fiellin DA, et al. Cost and utilization outcomes of opioid-dependence treatments. Am J Manag Care. 2011;17(suppl 8):S235-S248.
2. Gibson A, Degenhardt L, Mattick RP, et al. Exposure to opioid maintenance treatment reduces long-term mortality. Addiction. 2008;103:462-468. doi: 10.1111/j.1360-0443.2007.02090.x
3. Substance Abuse and Mental Health Services Administration. Key Substance Use and Mental Health Indicators in the United States: Results From the 2020 National Survey on Drug Use and Health. HHS Publication PEP21-07-01-003, NSDUH Series H-56. 2021. Accessed March 19, 2023. www.samhsa.gov/data/sites/default/files/reports/rpt35325/NSDUHFFRPDFWHTMLFiles2020/2020NSDUHFFR1PDFW102121.pdf
4. Haffajee RL, Andraka-Christou B, Attermann J, et al. A mixed-method comparison of physician-reported beliefs about and barriers to treatment with medications for opioid use disorder. Subst Abuse Treat Prev Policy. 2020;15:69. doi: 10.1186/s13011-020-00312-3
5. Kosten TR, George TP. The neurobiology of opioid dependence: implications for treatment. Sci Pract Perspect. 2002;1:13-20. doi: 10.1151/spp021113
6. Koob GF. Neurobiology of opioid addiction: opponent process, hyperkatifeia, and negative reinforcement. Biol Psychiatry. 2020;87:44-53. doi: 10.1016/j.biopsych.2019.05.023
7. Substance Abuse and Mental Health Services Administration. Medications for Opioid Use Disorder. For Health care and Addiction Professionals, Policymakers, Patients, and Families. Treatment Improvement Protocol TIP 63. Publication No. PEP21-02-01-002. 2021. Accessed March 19, 2023. https://store.samhsa.gov/sites/default/files/pep21-02-01-002.pdf
8. Sordo L, Barrio G, Bravo MJ, et al. Mortality risk during and after opioid substitution treatment: systematic review and meta-analysis of cohort studies. BMJ. 2017;357:j1550. doi: 10.1136/bmj.j1550
9. Korownyk C, Perry D, Ton J, et al. Opioid use disorder in primary care: PEER umbrella systematic review of systematic reviews. Can Fam Physician. 2019;65:e194-e206.
10. Mattick RP, Breen C, Kimber J, et al. Methadone maintenance therapy versus no opioid replacement therapy for opioid dependence. Cochrane Database Syst Rev. 2009;(3):CD002209. doi: 10.1002/14651858.CD002209.pub2
11. Mattick RP, Breen C, Kimber J, et al. Buprenorphine maintenance versus placebo or methadone maintenance for opioid dependence. Cochrane Database Syst Rev. 2014;(2):CD002207. doi: 10.1002/14651858.CD002207.pub4
12. Krupitsky E, Nunes EV, Ling W, et al. Injectable extended-release naltrexone for opioid dependence: a double-blind, placebo-controlled, multicentre randomised trial. Lancet. 2011;377:1506-1513. doi: 10.1016/S0140-6736(11)60358-9
13. Soyka M, Zingg C, Koller G, et al. Retention rate and substance use in methadone and buprenorphine maintenance therapy and predictors of outcome: results from a randomized study. Int J Neuropsychopharmacol. 2008;11:641-653. doi: 10.1017/S146114570700836X
14. Institute of Medicine Committee on Federal Regulation of Methadone Treatment; Rettig R, Yarmolinsky A, eds. Federal Regulation of Methadone Treatment. National Academies Press; 1995.
15. 42 eCFR §8. Medication assisted treatment for opioid use disorders. Revised March 15, 2023. Accessed March 23, 2023. www.ecfr.gov/current/title-42/chapter-I/subchapter-A/part-8?toc=1
16. Faggiano F, Vigna-Taglianti F, Versino E, et al. Methadone maintenance at different dosages for opioid dependence. Cochrane Database Syst Rev. 2003;(3):CD002208. doi: 10.1002/14651858.CD002208
17. Baxter LE Sr, Campbell A, Deshields M, et al. Safe methadone induction and stabilization: report of an expert panel. J Addict Med. 2013;7:377-386. doi: 10.1097/01.ADM.0000435321.39251.d7
18. Olfson M, Zhang VS, Schoenbaum M, et al. Trends in buprenorphine treatment in the United States, 2009-2018. JAMA. 2020;323:276-277. doi: 10.1001/jama.2019.18913
19. Walsh SL, Preston KL, Stitzer ML, et al. Clinical pharmacology of buprenorphine: ceiling effects at high doses. Clin Pharmacol Ther. 1994;55:569-580. doi: 10.1038/clpt.1994.71
20. Walley AY, Palmisano J, Sorensen-Alawad A, et al. Engagement and substance dependence in a primary care-based addiction treatment program for people infected with HIV and people at high-risk for HIV infection. J Subst Abuse Treat. 2015;59:59-66. doi: 10.1016/j.jsat.2015.07.007
21. Lagisetty P, Klasa K, Bush C, et al. Primary care models for treating opioid use disorders: what actually works? A systematic review. PloS One. 2017;12:e0186315. doi: 10.1371/journal.pone.0186315
22. Du CX, Shi J, Tetrault JM, et al. Primary care and medication management characteristics among patients receiving office-based opioid treatment with buprenorphine. Fam Pract. 2022;39:234-240. doi: 10.1093/fampra/cmab166
23. Herring AA, Vosooghi AA, Luftig J, et al. High-dose buprenorphine induction in the emergency department for treatment of opioid use disorder. JAMA Netw Open. 2021;4:e2117128. doi: 10.1001/jamanetworkopen.2021.17128
24. Hämmig R, Kemter A, Strasser J, et al. Use of microdoses for induction of buprenorphine treatment with overlapping full opioid agonist use: the Bernese method. Subst Abuse Rehabil. 2016;7:99-105. doi: 10.2147/SAR.S109919
25. Snyder H, Kalmin MM, Moulin A, et al. Rapid adoption of low-threshold buprenorphine treatment at California emergency departments participating in the CA Bridge Program. Ann Emerg Med. 2021;78:759-772. doi: 10.1016/j.annemergmed.2021.05.024
26. Wong JSH, Nikoo M, Westenberg JN, et al. Comparing rapid micro-induction and standard induction of buprenorphine/naloxone for treatment of opioid use disorder: protocol for an open-label, parallel-group, superiority, randomized controlled trial. Addict Sci Clin Pract. 2021;16:11. doi: 10.1186/s13722-021-00220-2
27. Lee JD, Vocci F, Fiellin DA. Unobserved “home” induction onto buprenorphine. J Addict Med. 2014;8:299-308. doi: 10.1097/ADM.0000000000000059
28. Krupitsky E, Zvartau E, Blokhina E, et al. Randomized trial of long-acting sustained-release naltrexone implant vs oral naltrexone or placebo for preventing relapse to opioid dependence. Arch Gen Psychiatry. 2012;69:973-981. doi: 10.1001/archgenpsychiatry.2012.1a
29. Wolfe D, Carrieri MP, Dasgupta N, et al. Concerns about injectable naltrexone for opioid dependence. Lancet. 2011;377:1468-1470. doi: 10.1016/S0140-6736(10)62056-9
30. Tanum L, Solli KK, Latif ZEH, et al. Effectiveness of injectable extended-release naltrexone vs daily buprenorphine–naloxone for opioid dependence: a randomized clinical noninferiority trial. JAMA Psychiatry. 2017;74:1197-1205. doi: 10.1001/jamapsychiatry.2017.3206
31. Murphy SM, Polsky D, Lee JD, et al. Cost-effectiveness of extended release naltrexone to prevent relapse among criminal justice-involved individuals with a history of opioid use disorder. Addiction. 2017;112:1440-1450. doi: 10.1111/add.13807
32. Ferrari A, Coccia CPR, Bertolini A, et al. Methadone—metabolism, pharmacokinetics and interactions. Pharmacol Res. 2004;50:551-559. doi: 10.1016/j.phrs.2004.05.002
33. 42 eCFR Part 2. Confidentiality of substance use disorder patient records. January 18, 2017. Accessed March 23, 2023. www.ecfr.gov/current/title-42/chapter-I/subchapter-A/part-2
34. Kao DP, Haigney MCP, Mehler PS, et al. Arrhythmia associated with buprenorphine and methadone reported to the Food and Drug Administration. Addiction. 2015;110:1468-1475. doi: 10.1111/add.13013
35. Tisdale JE, Chung MK, Campbell KB, et al; . Drug-induced arrhythmias: a scientific statement from the American Heart Association. Circulation. 2020;142:e214-e233. doi: 10.1161/CIR.0000000000000905
36. Leshner AI, Mancher M, eds. Barriers to broader use of medications to treat opioid use disorder. In: Medications for Opioid Use Disorder Save Lives. National Academies Press; 2019:109-136.
37. Chilcoat HD, Amick HR, Sherwood MR, et al. Buprenorphine in the United States: Motives for abuse, misuse, and diversion. J Subst Abuse Treat. 2019;104:148-157. doi: 10.1016/j.jsat. 2019.07.005
38. Qato DM, Daviglus ML, Wilder J, et al. “Pharmacy deserts” are prevalent in Chicago’s predominantly minority communities, raising medication access concerns. Health Aff (Millwood). 2014;33:1958-1965. doi: 10.1377/hlthaff.2013.1397
39. Mason M, Soliman R, Kim HS, et al. Disparities by sex and race and ethnicity in death rates due to opioid overdose among adults 55 years or older, 1999 to 2019. JAMA Netw Open. 2022;5:e2142982. doi: 10.1001/jamanetworkopen.2021.42982
40. Rosenblum A, Cleland CM, Fong C, et al. Distance traveled and cross-state commuting to opioid treatment programs in the United States. J Environ Public Health. 2011;2011:948789. doi: 10.1155/2011/948789
41. Chan B, Hoffman KA, Bougatsos C, et al. Mobile methadone medication units: a brief history, scoping review and research opportunity. J Subst Abuse Treat. 2021;129:108483. doi: 10.1016/j.jsat.2021.108483
42. Jakubowski A, Fox A. Defining low-threshold buprenorphine treatment. J Addict Med. 2020;14:95-98. doi: 10.1097/ADM.0000000000000555
43. Messmer SE, Elmes AT, Jimenez AD, et al. Outcomes of a mobile medical unit for low-threshold buprenorphine access targeting opioid overdose hot spots in Chicago. J Subst Use Addict Treat. 2023;209054. doi: 10.1016/j.josat.2023.209054
PRACTICE RECOMMENDATIONS
› Consider resource availability (eg, treatment programs and regulatory barriers), in addition to patient- and medicationspecific factors, when designing the most individualized, advantageous medication-assisted recovery plan, to reduce the risk for mortality. B
› Schedule early (< 2 weeks) and frequent follow-up with patients who are starting medications for opioid use disorder (particularly methadone), to manage risk when mortality is highest and to support recovery. C
› Set and manage patient expectations for control of withdrawal symptoms when initiating medications for opioid use disorder (particularly buprenorphine). B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Thoughts on the CDC update on opioid prescribing guidelines
The media is filled with stories about the opioid crisis. We have all heard the horror stories of addiction and overdose, as well as “pill mill” doctors. In fact, more than 932,000 people have died of drug overdose since 1999 and, in recent years, approximately 75% of drug overdoses involved opioids.
Yet, they still have their place in the treatment of pain.
The CDC updated the 2016 guidelines for prescribing opioids for pain in 2022. They cover when to initiate prescribing of opioids, selecting appropriate opioids and doses, and deciding the duration of therapy. The guidelines do a great job providing evidence-based recommendations while at the same time keeping the problems with opioids in the picture.
For primary care doctors, pain is one of the most common complaints we see – from broken bones to low back pain to cancer pain. It is important to note that the current guidelines exclude pain from sickle cell disease, cancer-related pain, palliative care, and end-of-life care. The guidelines apply to acute, subacute, and chronic pain. Pain is a complex symptom and often needs a multipronged approach. We make a mistake if we just prescribe a pain medication without understanding the root cause of the pain.
The guidelines suggest starting with nonopioid medications and incorporating nonmedicinal modes of treatments, such as physical therapy, as well. Opioids should be started at the lowest dose and for the shortest duration. Immediate-release medications are preferred over long-acting or extended-release ones. The patient should always be informed of the risks and benefits.
While the guidelines do a great job recommending how to prescribe opioids, they do not go into any depth discussing other treatment options. Perhaps knowledge of other treatment modalities would help primary care physicians avoid opioid prescribing. When treating our patients, it is important to educate them on how to manage their own symptoms.
The guidelines also advise tapering patients who may have been on high-dose opioids for long periods of time. Doctors know this is a very difficult task. However, resources to help with this are often lacking. For example, rehab may not be covered under a patient’s insurance, or it may be cheaper to take an opioid than to go to physical therapy. Although the recommendation is to taper, community assets may not support this. Guidelines are one thing, but the rest of the health care system needs to catch up to them and make them practical.
Primary care doctors often utilize our physical medicine, rehabilitation, and pain management specialists to assist in managing our patients’ pain. Here too, access to this resource is often difficult to come by. Depending on a patient’s insurance, it can take months to get an appointment.
In general, the current guidelines offer 12 key recommendations when prescribing opioids. They are a great reference; however, we need more real-life tools. For many of us in primary care, these guidelines support what we’ve been doing all along.
Primary care doctors will surely play a huge role in addressing the opioid crisis. We can prescribe opioids appropriately, but it doesn’t erase the problems of those patients who were overprescribed in the past. Many still seek out these medications whether for monetary reasons or just for the high. It is often easy to blame the patient but the one in control is the one with the prescription pad. Yet, it is important to remember that many of these patients are in real pain and need help.
Often, it is simpler to just prescribe a pain medication than it is to explain why one is not appropriate. As primary care doctors, we need to be effective ambassadors of appropriate opioid prescribing and often that means doing the hard thing and saying no to a patient.
Dr. Girgis practices family medicine in South River, N.J., and is a clinical assistant professor of family medicine at Robert Wood Johnson Medical School, New Brunswick, N.J.
fpnews@mdedge.com
The media is filled with stories about the opioid crisis. We have all heard the horror stories of addiction and overdose, as well as “pill mill” doctors. In fact, more than 932,000 people have died of drug overdose since 1999 and, in recent years, approximately 75% of drug overdoses involved opioids.
Yet, they still have their place in the treatment of pain.
The CDC updated the 2016 guidelines for prescribing opioids for pain in 2022. They cover when to initiate prescribing of opioids, selecting appropriate opioids and doses, and deciding the duration of therapy. The guidelines do a great job providing evidence-based recommendations while at the same time keeping the problems with opioids in the picture.
For primary care doctors, pain is one of the most common complaints we see – from broken bones to low back pain to cancer pain. It is important to note that the current guidelines exclude pain from sickle cell disease, cancer-related pain, palliative care, and end-of-life care. The guidelines apply to acute, subacute, and chronic pain. Pain is a complex symptom and often needs a multipronged approach. We make a mistake if we just prescribe a pain medication without understanding the root cause of the pain.
The guidelines suggest starting with nonopioid medications and incorporating nonmedicinal modes of treatments, such as physical therapy, as well. Opioids should be started at the lowest dose and for the shortest duration. Immediate-release medications are preferred over long-acting or extended-release ones. The patient should always be informed of the risks and benefits.
While the guidelines do a great job recommending how to prescribe opioids, they do not go into any depth discussing other treatment options. Perhaps knowledge of other treatment modalities would help primary care physicians avoid opioid prescribing. When treating our patients, it is important to educate them on how to manage their own symptoms.
The guidelines also advise tapering patients who may have been on high-dose opioids for long periods of time. Doctors know this is a very difficult task. However, resources to help with this are often lacking. For example, rehab may not be covered under a patient’s insurance, or it may be cheaper to take an opioid than to go to physical therapy. Although the recommendation is to taper, community assets may not support this. Guidelines are one thing, but the rest of the health care system needs to catch up to them and make them practical.
Primary care doctors often utilize our physical medicine, rehabilitation, and pain management specialists to assist in managing our patients’ pain. Here too, access to this resource is often difficult to come by. Depending on a patient’s insurance, it can take months to get an appointment.
In general, the current guidelines offer 12 key recommendations when prescribing opioids. They are a great reference; however, we need more real-life tools. For many of us in primary care, these guidelines support what we’ve been doing all along.
Primary care doctors will surely play a huge role in addressing the opioid crisis. We can prescribe opioids appropriately, but it doesn’t erase the problems of those patients who were overprescribed in the past. Many still seek out these medications whether for monetary reasons or just for the high. It is often easy to blame the patient but the one in control is the one with the prescription pad. Yet, it is important to remember that many of these patients are in real pain and need help.
Often, it is simpler to just prescribe a pain medication than it is to explain why one is not appropriate. As primary care doctors, we need to be effective ambassadors of appropriate opioid prescribing and often that means doing the hard thing and saying no to a patient.
Dr. Girgis practices family medicine in South River, N.J., and is a clinical assistant professor of family medicine at Robert Wood Johnson Medical School, New Brunswick, N.J.
fpnews@mdedge.com
The media is filled with stories about the opioid crisis. We have all heard the horror stories of addiction and overdose, as well as “pill mill” doctors. In fact, more than 932,000 people have died of drug overdose since 1999 and, in recent years, approximately 75% of drug overdoses involved opioids.
Yet, they still have their place in the treatment of pain.
The CDC updated the 2016 guidelines for prescribing opioids for pain in 2022. They cover when to initiate prescribing of opioids, selecting appropriate opioids and doses, and deciding the duration of therapy. The guidelines do a great job providing evidence-based recommendations while at the same time keeping the problems with opioids in the picture.
For primary care doctors, pain is one of the most common complaints we see – from broken bones to low back pain to cancer pain. It is important to note that the current guidelines exclude pain from sickle cell disease, cancer-related pain, palliative care, and end-of-life care. The guidelines apply to acute, subacute, and chronic pain. Pain is a complex symptom and often needs a multipronged approach. We make a mistake if we just prescribe a pain medication without understanding the root cause of the pain.
The guidelines suggest starting with nonopioid medications and incorporating nonmedicinal modes of treatments, such as physical therapy, as well. Opioids should be started at the lowest dose and for the shortest duration. Immediate-release medications are preferred over long-acting or extended-release ones. The patient should always be informed of the risks and benefits.
While the guidelines do a great job recommending how to prescribe opioids, they do not go into any depth discussing other treatment options. Perhaps knowledge of other treatment modalities would help primary care physicians avoid opioid prescribing. When treating our patients, it is important to educate them on how to manage their own symptoms.
The guidelines also advise tapering patients who may have been on high-dose opioids for long periods of time. Doctors know this is a very difficult task. However, resources to help with this are often lacking. For example, rehab may not be covered under a patient’s insurance, or it may be cheaper to take an opioid than to go to physical therapy. Although the recommendation is to taper, community assets may not support this. Guidelines are one thing, but the rest of the health care system needs to catch up to them and make them practical.
Primary care doctors often utilize our physical medicine, rehabilitation, and pain management specialists to assist in managing our patients’ pain. Here too, access to this resource is often difficult to come by. Depending on a patient’s insurance, it can take months to get an appointment.
In general, the current guidelines offer 12 key recommendations when prescribing opioids. They are a great reference; however, we need more real-life tools. For many of us in primary care, these guidelines support what we’ve been doing all along.
Primary care doctors will surely play a huge role in addressing the opioid crisis. We can prescribe opioids appropriately, but it doesn’t erase the problems of those patients who were overprescribed in the past. Many still seek out these medications whether for monetary reasons or just for the high. It is often easy to blame the patient but the one in control is the one with the prescription pad. Yet, it is important to remember that many of these patients are in real pain and need help.
Often, it is simpler to just prescribe a pain medication than it is to explain why one is not appropriate. As primary care doctors, we need to be effective ambassadors of appropriate opioid prescribing and often that means doing the hard thing and saying no to a patient.
Dr. Girgis practices family medicine in South River, N.J., and is a clinical assistant professor of family medicine at Robert Wood Johnson Medical School, New Brunswick, N.J.
fpnews@mdedge.com
Watching feasible for asymptomatic kidney stones
Many patients with asymptomatic renal stones can qualify for an active surveillance program, Swiss researchers report at the American Urological Association 2023 Annual Meeting.
Kevin Stritt, MD, chief resident in the urology department at Lausanne University Hospital, said kidney stones often pass without symptoms. But until now, data on the frequency of asymptomatic, spontaneous passage of stones have been lacking.
The new data come from the NOSTONE trial, a prospective, multicenter, double-blind, placebo-controlled randomized trial to assess the efficacy of hydrochlorothiazide in the prevention of recurrence in patients with recurrent calcium-containing kidney stones.
Dr. Stritt and colleagues evaluated the natural history of asymptomatic renal stones during a median follow-up of 35 months. “We found for the first time that a relevant number of kidney stone passages [39%] were asymptomatic, spontaneous stone passages,” Dr. Stritt told this news organization.
All asymptomatic spontaneous stone passages were analyzed in a comparison of the total number of kidney stones on low-dose, nonintravenous contrast CT imaging at the beginning and end of the 3-year follow-up.
Of the 403 stones passed spontaneously, 61% (245) were symptomatic stone passages and 39% (158) were asymptomatic stone passages, Dr. Stritt told this news organization.
Asymptomatic stones were a median size of 2.4 mm, and symptomatic stones were 2.15 mm, which was not significantly different (P = .366), according to the researchers. Dr. Stritt said the spontaneous passage of asymptomatic stones was largely influenced by a higher number of stones on CT imaging at randomization (P = .001) and a lower total stone volume (P = .001).
Ephrem Olweny, MD, an assistant professor of urology and section chief of endourology at Rush University Medical Center in Chicago, said previous studies have found that the rate of spontaneous passage of kidney stones ranges from 3% to 29%.
“But this secondary analysis of data from a prior multicenter prospective randomized trial offers higher-quality data that will be of value in guiding patient counseling,” Dr. Olweny said.
“Observation should be initially offered to these patients. However, patients should be informed that 52% are likely to develop symptoms, and some may indeed opt for preemptive surgical removal,” he added.
David Schulsinger, MD, an associate professor in the department of urology at Stony Brook (N.Y.) University Hospital, said the incidence of kidney stones has been increasing worldwide, affecting approximately 12% of men and 6% of women. Dehydration and diets high in sodium and calcium are major factors, he said.
Patients with a history of stones have a 50% risk of recurrence in the next 5 years, and an 80% risk in their lifetime, he added.
Dr. Schulsinger said the message from the Swiss study is that urologists can be “comfortable” watching small stones, those averaging 2.4 mm or less in size. “But if a patient has a 7- or 8-mm stone, you might be more inclined to manage that patient a little bit more aggressively.”
Roughly half of patients with stones less than 2 mm will pass it in about 8 days, he said.
Dr. Olweny noted that the study was a secondary analysis of data from a randomized controlled trial that evaluated the efficacy of thiazides in preventing the recurrence of calcium stones. “The original study was not specifically designed to look at asymptomatic stone passage rates for small renal stones, and therefore, the observed rates may not reflect the most precise estimates,” he said.
Dr. Stritt said his group has not studied the size limit of stones that pass spontaneously without symptoms. “This study could serve to construct recurrence prediction models based on medical history and stone burden on CT imaging. More well-designed research on this topic is urgently needed,” he said. “These results should encourage urologists to counsel patients about the possibility of an active surveillance strategy when smaller kidney stones are present.”
The author and independent commentators have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Many patients with asymptomatic renal stones can qualify for an active surveillance program, Swiss researchers report at the American Urological Association 2023 Annual Meeting.
Kevin Stritt, MD, chief resident in the urology department at Lausanne University Hospital, said kidney stones often pass without symptoms. But until now, data on the frequency of asymptomatic, spontaneous passage of stones have been lacking.
The new data come from the NOSTONE trial, a prospective, multicenter, double-blind, placebo-controlled randomized trial to assess the efficacy of hydrochlorothiazide in the prevention of recurrence in patients with recurrent calcium-containing kidney stones.
Dr. Stritt and colleagues evaluated the natural history of asymptomatic renal stones during a median follow-up of 35 months. “We found for the first time that a relevant number of kidney stone passages [39%] were asymptomatic, spontaneous stone passages,” Dr. Stritt told this news organization.
All asymptomatic spontaneous stone passages were analyzed in a comparison of the total number of kidney stones on low-dose, nonintravenous contrast CT imaging at the beginning and end of the 3-year follow-up.
Of the 403 stones passed spontaneously, 61% (245) were symptomatic stone passages and 39% (158) were asymptomatic stone passages, Dr. Stritt told this news organization.
Asymptomatic stones were a median size of 2.4 mm, and symptomatic stones were 2.15 mm, which was not significantly different (P = .366), according to the researchers. Dr. Stritt said the spontaneous passage of asymptomatic stones was largely influenced by a higher number of stones on CT imaging at randomization (P = .001) and a lower total stone volume (P = .001).
Ephrem Olweny, MD, an assistant professor of urology and section chief of endourology at Rush University Medical Center in Chicago, said previous studies have found that the rate of spontaneous passage of kidney stones ranges from 3% to 29%.
“But this secondary analysis of data from a prior multicenter prospective randomized trial offers higher-quality data that will be of value in guiding patient counseling,” Dr. Olweny said.
“Observation should be initially offered to these patients. However, patients should be informed that 52% are likely to develop symptoms, and some may indeed opt for preemptive surgical removal,” he added.
David Schulsinger, MD, an associate professor in the department of urology at Stony Brook (N.Y.) University Hospital, said the incidence of kidney stones has been increasing worldwide, affecting approximately 12% of men and 6% of women. Dehydration and diets high in sodium and calcium are major factors, he said.
Patients with a history of stones have a 50% risk of recurrence in the next 5 years, and an 80% risk in their lifetime, he added.
Dr. Schulsinger said the message from the Swiss study is that urologists can be “comfortable” watching small stones, those averaging 2.4 mm or less in size. “But if a patient has a 7- or 8-mm stone, you might be more inclined to manage that patient a little bit more aggressively.”
Roughly half of patients with stones less than 2 mm will pass it in about 8 days, he said.
Dr. Olweny noted that the study was a secondary analysis of data from a randomized controlled trial that evaluated the efficacy of thiazides in preventing the recurrence of calcium stones. “The original study was not specifically designed to look at asymptomatic stone passage rates for small renal stones, and therefore, the observed rates may not reflect the most precise estimates,” he said.
Dr. Stritt said his group has not studied the size limit of stones that pass spontaneously without symptoms. “This study could serve to construct recurrence prediction models based on medical history and stone burden on CT imaging. More well-designed research on this topic is urgently needed,” he said. “These results should encourage urologists to counsel patients about the possibility of an active surveillance strategy when smaller kidney stones are present.”
The author and independent commentators have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Many patients with asymptomatic renal stones can qualify for an active surveillance program, Swiss researchers report at the American Urological Association 2023 Annual Meeting.
Kevin Stritt, MD, chief resident in the urology department at Lausanne University Hospital, said kidney stones often pass without symptoms. But until now, data on the frequency of asymptomatic, spontaneous passage of stones have been lacking.
The new data come from the NOSTONE trial, a prospective, multicenter, double-blind, placebo-controlled randomized trial to assess the efficacy of hydrochlorothiazide in the prevention of recurrence in patients with recurrent calcium-containing kidney stones.
Dr. Stritt and colleagues evaluated the natural history of asymptomatic renal stones during a median follow-up of 35 months. “We found for the first time that a relevant number of kidney stone passages [39%] were asymptomatic, spontaneous stone passages,” Dr. Stritt told this news organization.
All asymptomatic spontaneous stone passages were analyzed in a comparison of the total number of kidney stones on low-dose, nonintravenous contrast CT imaging at the beginning and end of the 3-year follow-up.
Of the 403 stones passed spontaneously, 61% (245) were symptomatic stone passages and 39% (158) were asymptomatic stone passages, Dr. Stritt told this news organization.
Asymptomatic stones were a median size of 2.4 mm, and symptomatic stones were 2.15 mm, which was not significantly different (P = .366), according to the researchers. Dr. Stritt said the spontaneous passage of asymptomatic stones was largely influenced by a higher number of stones on CT imaging at randomization (P = .001) and a lower total stone volume (P = .001).
Ephrem Olweny, MD, an assistant professor of urology and section chief of endourology at Rush University Medical Center in Chicago, said previous studies have found that the rate of spontaneous passage of kidney stones ranges from 3% to 29%.
“But this secondary analysis of data from a prior multicenter prospective randomized trial offers higher-quality data that will be of value in guiding patient counseling,” Dr. Olweny said.
“Observation should be initially offered to these patients. However, patients should be informed that 52% are likely to develop symptoms, and some may indeed opt for preemptive surgical removal,” he added.
David Schulsinger, MD, an associate professor in the department of urology at Stony Brook (N.Y.) University Hospital, said the incidence of kidney stones has been increasing worldwide, affecting approximately 12% of men and 6% of women. Dehydration and diets high in sodium and calcium are major factors, he said.
Patients with a history of stones have a 50% risk of recurrence in the next 5 years, and an 80% risk in their lifetime, he added.
Dr. Schulsinger said the message from the Swiss study is that urologists can be “comfortable” watching small stones, those averaging 2.4 mm or less in size. “But if a patient has a 7- or 8-mm stone, you might be more inclined to manage that patient a little bit more aggressively.”
Roughly half of patients with stones less than 2 mm will pass it in about 8 days, he said.
Dr. Olweny noted that the study was a secondary analysis of data from a randomized controlled trial that evaluated the efficacy of thiazides in preventing the recurrence of calcium stones. “The original study was not specifically designed to look at asymptomatic stone passage rates for small renal stones, and therefore, the observed rates may not reflect the most precise estimates,” he said.
Dr. Stritt said his group has not studied the size limit of stones that pass spontaneously without symptoms. “This study could serve to construct recurrence prediction models based on medical history and stone burden on CT imaging. More well-designed research on this topic is urgently needed,” he said. “These results should encourage urologists to counsel patients about the possibility of an active surveillance strategy when smaller kidney stones are present.”
The author and independent commentators have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Interdisciplinary program reduced pediatric pain without pharmacology
WASHINGTON – A nonpharmacologic, interdisciplinary program significantly improved chronic pain in children and the quality of life for their families, based on data from 115 individuals.
Up to 40% of children experience chronic pain that affects their physical, psychosocial, and educational functioning, said Jessica Campanile, BA, a medical student at the University of Pennsylvania, Philadelphia, in a presentation at the Pediatric Academic Societies annual meeting.
Although interdisciplinary pediatric pain rehabilitation programs have shown positive outcomes, very few use only nonpharmacologic treatments, said Ms. Campanile. In addition, few studies have explored the effects of a hospital-based program on the patients and their families.
Ms. Campanile and colleagues conducted a retrospective cohort study of participants in an outpatient pain rehabilitation program at the Children’s Hospital of Philadelphia between April 2016 and December 2019. Patients were evaluated by a pediatric rheumatologist, psychologist, and physical and occupational therapists.
Patients engaged in 2-3 hours of physical therapy (PT) and 2-3 hours of occupational therapy (OT) in a 1:1 ratio at least 5 days a week. Physical activities included stepping into and out of a tub, carrying laundry, and desensitizing to allodynia as needed. Participants also received individual and group cognitive-behavior therapy interventions from psychologists, and psychological support during PT and OT sessions if needed. Parents/caregivers were invited to separate individual and group therapy sessions as part of the program. The median age at admission to the program was 15 years, and 79% of the participants were female. Patients participated the program for a median of 17 days, and 87% were outpatients who came to the hospital for the program.
Pain was assessed based on the 0-10 verbal pain intensity scale, energy was assessed on a scale of 0-100, and functional disability was assessed on a scale of 0-60, with higher scores indicating more pain, more energy, and more self-perceived disability, respectively.
Overall, scores on measures of pain, disability, allodynia, and energy improved significantly from baseline to discharge from the program. Verbal pain intensity scores decreased on average from 7 to 5, disability scores decreased from 26 to 9, the proportion of patients reporting allodynia decreased from 86% to 61%, and the energy level score increased from 70 to 77. The trend continued at the first follow-up visit, conducted 2-3 months after discharge from the program. Notably, pain intensity further decreased from a median of 5 at program completion to a median of 2 at the first follow-up, Ms. Campanile said. Improvements in allodynia also were sustained at the first follow-up.
Quality of life measures related to physical, emotional, social, and cognitive function also improved significantly from baseline to completion of the program.
In addition, scores on a quality of life family impact survey improved significantly; in particular, parent health-related quality of life scores (Parent HRQoL) improved from 60 at baseline to 71 at the end of the program on a scale of 0 to 100. The study findings were limited by several factors including the relatively short duration and use of a convenience sample from a retrospective cohort, with data limited to electronic health records, Ms. Campanile said. The study also was not powered to examine differential treatments based on psychiatric conditions, and any psychiatric conditions were based on self-reports.
However, the results support the value of a nonpharmacologic interdisciplinary program as “a robust treatment for youth with chronic idiopathic pain, for both patients and the family unit,” she said.
“This study also supports the need for and benefit of additional counseling for patients and their caregivers prior to and during enrollment in a pain rehabilitation program,” she concluded.
Study supports effectiveness of drug-free pain management
“The management of pain in any age group can be challenging, especially with current concerns for opioid dependence and abuse,” Cathy Haut, DNP, CPNP-AC, CPNP-PC, a pediatric nurse practitioner in Rehoboth Beach, Del., said in an interview.
“Chronic pain affects daily life for all populations, but for children, adolescents, and their families, it can have a long-lasting impact on growth and development, psychosocial and physical well-being,” Dr. Haut said. “Determining and testing nonpharmacologic alternative methods of pain control are extremely important.”
Given the debilitating effects of chronic pain, and the potential side effects and dependence that have been associated with use of pharmacologic modes of pain control, unique and creative solutions have begun to emerge and need further attention and study, she said.
However, “despite published research supporting the use of alternative and complementary approaches to pain control in children and adolescents, nonpharmacologic, collaborative, interprofessional approaches to pain control have not been widely shared in the literature,” she said.
“Barriers to this type of program include first and foremost a potential lack of financial and workforce-related resources,” Dr. Haut said. “Patient and family attendance at frequent health visits, daily or even every other day, may also hinder success, but opportunities for telehealth and family training to learn physical and occupational skills within this type of program may be beginning solutions.”
Additional research should be conducted at multiple children’s hospitals, with a larger number of children and adolescents at varying ages, with pain related to different diagnoses, and with the inclusion of collaborative methodology, said Dr. Haut. “The current study had some limitations, including the small sample size, predominantly female sex, and a short participation time frame utilizing retrospective review. Completing prospective research over a longer time frame can also yield generalizable results applicable to varied populations.”
The study received no outside funding. Ms. Campanile had no financial conflicts to disclose. Dr. Haut had no financial conflicts to disclose, and serves on the editorial advisory board of Pediatric News.
WASHINGTON – A nonpharmacologic, interdisciplinary program significantly improved chronic pain in children and the quality of life for their families, based on data from 115 individuals.
Up to 40% of children experience chronic pain that affects their physical, psychosocial, and educational functioning, said Jessica Campanile, BA, a medical student at the University of Pennsylvania, Philadelphia, in a presentation at the Pediatric Academic Societies annual meeting.
Although interdisciplinary pediatric pain rehabilitation programs have shown positive outcomes, very few use only nonpharmacologic treatments, said Ms. Campanile. In addition, few studies have explored the effects of a hospital-based program on the patients and their families.
Ms. Campanile and colleagues conducted a retrospective cohort study of participants in an outpatient pain rehabilitation program at the Children’s Hospital of Philadelphia between April 2016 and December 2019. Patients were evaluated by a pediatric rheumatologist, psychologist, and physical and occupational therapists.
Patients engaged in 2-3 hours of physical therapy (PT) and 2-3 hours of occupational therapy (OT) in a 1:1 ratio at least 5 days a week. Physical activities included stepping into and out of a tub, carrying laundry, and desensitizing to allodynia as needed. Participants also received individual and group cognitive-behavior therapy interventions from psychologists, and psychological support during PT and OT sessions if needed. Parents/caregivers were invited to separate individual and group therapy sessions as part of the program. The median age at admission to the program was 15 years, and 79% of the participants were female. Patients participated the program for a median of 17 days, and 87% were outpatients who came to the hospital for the program.
Pain was assessed based on the 0-10 verbal pain intensity scale, energy was assessed on a scale of 0-100, and functional disability was assessed on a scale of 0-60, with higher scores indicating more pain, more energy, and more self-perceived disability, respectively.
Overall, scores on measures of pain, disability, allodynia, and energy improved significantly from baseline to discharge from the program. Verbal pain intensity scores decreased on average from 7 to 5, disability scores decreased from 26 to 9, the proportion of patients reporting allodynia decreased from 86% to 61%, and the energy level score increased from 70 to 77. The trend continued at the first follow-up visit, conducted 2-3 months after discharge from the program. Notably, pain intensity further decreased from a median of 5 at program completion to a median of 2 at the first follow-up, Ms. Campanile said. Improvements in allodynia also were sustained at the first follow-up.
Quality of life measures related to physical, emotional, social, and cognitive function also improved significantly from baseline to completion of the program.
In addition, scores on a quality of life family impact survey improved significantly; in particular, parent health-related quality of life scores (Parent HRQoL) improved from 60 at baseline to 71 at the end of the program on a scale of 0 to 100. The study findings were limited by several factors including the relatively short duration and use of a convenience sample from a retrospective cohort, with data limited to electronic health records, Ms. Campanile said. The study also was not powered to examine differential treatments based on psychiatric conditions, and any psychiatric conditions were based on self-reports.
However, the results support the value of a nonpharmacologic interdisciplinary program as “a robust treatment for youth with chronic idiopathic pain, for both patients and the family unit,” she said.
“This study also supports the need for and benefit of additional counseling for patients and their caregivers prior to and during enrollment in a pain rehabilitation program,” she concluded.
Study supports effectiveness of drug-free pain management
“The management of pain in any age group can be challenging, especially with current concerns for opioid dependence and abuse,” Cathy Haut, DNP, CPNP-AC, CPNP-PC, a pediatric nurse practitioner in Rehoboth Beach, Del., said in an interview.
“Chronic pain affects daily life for all populations, but for children, adolescents, and their families, it can have a long-lasting impact on growth and development, psychosocial and physical well-being,” Dr. Haut said. “Determining and testing nonpharmacologic alternative methods of pain control are extremely important.”
Given the debilitating effects of chronic pain, and the potential side effects and dependence that have been associated with use of pharmacologic modes of pain control, unique and creative solutions have begun to emerge and need further attention and study, she said.
However, “despite published research supporting the use of alternative and complementary approaches to pain control in children and adolescents, nonpharmacologic, collaborative, interprofessional approaches to pain control have not been widely shared in the literature,” she said.
“Barriers to this type of program include first and foremost a potential lack of financial and workforce-related resources,” Dr. Haut said. “Patient and family attendance at frequent health visits, daily or even every other day, may also hinder success, but opportunities for telehealth and family training to learn physical and occupational skills within this type of program may be beginning solutions.”
Additional research should be conducted at multiple children’s hospitals, with a larger number of children and adolescents at varying ages, with pain related to different diagnoses, and with the inclusion of collaborative methodology, said Dr. Haut. “The current study had some limitations, including the small sample size, predominantly female sex, and a short participation time frame utilizing retrospective review. Completing prospective research over a longer time frame can also yield generalizable results applicable to varied populations.”
The study received no outside funding. Ms. Campanile had no financial conflicts to disclose. Dr. Haut had no financial conflicts to disclose, and serves on the editorial advisory board of Pediatric News.
WASHINGTON – A nonpharmacologic, interdisciplinary program significantly improved chronic pain in children and the quality of life for their families, based on data from 115 individuals.
Up to 40% of children experience chronic pain that affects their physical, psychosocial, and educational functioning, said Jessica Campanile, BA, a medical student at the University of Pennsylvania, Philadelphia, in a presentation at the Pediatric Academic Societies annual meeting.
Although interdisciplinary pediatric pain rehabilitation programs have shown positive outcomes, very few use only nonpharmacologic treatments, said Ms. Campanile. In addition, few studies have explored the effects of a hospital-based program on the patients and their families.
Ms. Campanile and colleagues conducted a retrospective cohort study of participants in an outpatient pain rehabilitation program at the Children’s Hospital of Philadelphia between April 2016 and December 2019. Patients were evaluated by a pediatric rheumatologist, psychologist, and physical and occupational therapists.
Patients engaged in 2-3 hours of physical therapy (PT) and 2-3 hours of occupational therapy (OT) in a 1:1 ratio at least 5 days a week. Physical activities included stepping into and out of a tub, carrying laundry, and desensitizing to allodynia as needed. Participants also received individual and group cognitive-behavior therapy interventions from psychologists, and psychological support during PT and OT sessions if needed. Parents/caregivers were invited to separate individual and group therapy sessions as part of the program. The median age at admission to the program was 15 years, and 79% of the participants were female. Patients participated the program for a median of 17 days, and 87% were outpatients who came to the hospital for the program.
Pain was assessed based on the 0-10 verbal pain intensity scale, energy was assessed on a scale of 0-100, and functional disability was assessed on a scale of 0-60, with higher scores indicating more pain, more energy, and more self-perceived disability, respectively.
Overall, scores on measures of pain, disability, allodynia, and energy improved significantly from baseline to discharge from the program. Verbal pain intensity scores decreased on average from 7 to 5, disability scores decreased from 26 to 9, the proportion of patients reporting allodynia decreased from 86% to 61%, and the energy level score increased from 70 to 77. The trend continued at the first follow-up visit, conducted 2-3 months after discharge from the program. Notably, pain intensity further decreased from a median of 5 at program completion to a median of 2 at the first follow-up, Ms. Campanile said. Improvements in allodynia also were sustained at the first follow-up.
Quality of life measures related to physical, emotional, social, and cognitive function also improved significantly from baseline to completion of the program.
In addition, scores on a quality of life family impact survey improved significantly; in particular, parent health-related quality of life scores (Parent HRQoL) improved from 60 at baseline to 71 at the end of the program on a scale of 0 to 100. The study findings were limited by several factors including the relatively short duration and use of a convenience sample from a retrospective cohort, with data limited to electronic health records, Ms. Campanile said. The study also was not powered to examine differential treatments based on psychiatric conditions, and any psychiatric conditions were based on self-reports.
However, the results support the value of a nonpharmacologic interdisciplinary program as “a robust treatment for youth with chronic idiopathic pain, for both patients and the family unit,” she said.
“This study also supports the need for and benefit of additional counseling for patients and their caregivers prior to and during enrollment in a pain rehabilitation program,” she concluded.
Study supports effectiveness of drug-free pain management
“The management of pain in any age group can be challenging, especially with current concerns for opioid dependence and abuse,” Cathy Haut, DNP, CPNP-AC, CPNP-PC, a pediatric nurse practitioner in Rehoboth Beach, Del., said in an interview.
“Chronic pain affects daily life for all populations, but for children, adolescents, and their families, it can have a long-lasting impact on growth and development, psychosocial and physical well-being,” Dr. Haut said. “Determining and testing nonpharmacologic alternative methods of pain control are extremely important.”
Given the debilitating effects of chronic pain, and the potential side effects and dependence that have been associated with use of pharmacologic modes of pain control, unique and creative solutions have begun to emerge and need further attention and study, she said.
However, “despite published research supporting the use of alternative and complementary approaches to pain control in children and adolescents, nonpharmacologic, collaborative, interprofessional approaches to pain control have not been widely shared in the literature,” she said.
“Barriers to this type of program include first and foremost a potential lack of financial and workforce-related resources,” Dr. Haut said. “Patient and family attendance at frequent health visits, daily or even every other day, may also hinder success, but opportunities for telehealth and family training to learn physical and occupational skills within this type of program may be beginning solutions.”
Additional research should be conducted at multiple children’s hospitals, with a larger number of children and adolescents at varying ages, with pain related to different diagnoses, and with the inclusion of collaborative methodology, said Dr. Haut. “The current study had some limitations, including the small sample size, predominantly female sex, and a short participation time frame utilizing retrospective review. Completing prospective research over a longer time frame can also yield generalizable results applicable to varied populations.”
The study received no outside funding. Ms. Campanile had no financial conflicts to disclose. Dr. Haut had no financial conflicts to disclose, and serves on the editorial advisory board of Pediatric News.
FROM PAS 2023
Cancer pain declines with cannabis use
in a study.
Physician-prescribed cannabis, particularly cannabinoids, has been shown to ease cancer-related pain in adult cancer patients, who often find inadequate pain relief from medications including opioids, Saro Aprikian, MSc, a medical student at the Royal College of Surgeons, Dublin, and colleagues, wrote in their paper.
However, real-world data on the safety and effectiveness of cannabis in the cancer population and the impact on use of other medications are lacking, the researchers said.
In the study, published in BMJ Supportive & Palliative Care, the researchers reviewed data from 358 adults with cancer who were part of a multicenter cannabis registry in Canada between May 2015 and October 2018.
The average age of the patients was 57.6 years, and 48% were men. The top three cancer diagnoses in the study population were genitorurinary, breast, and colorectal.
Pain was the most common reason for obtaining a medical cannabis prescription, cited by 72.4% of patients.
Data were collected at follow-up visits conducted every 3 months over 1 year. Pain was assessed via the Brief Pain Inventory (BPI) and revised Edmonton Symptom Assessment System (ESAS-r) questionnaires and compared to baseline values. Patients rated their pain intensity on a sliding scale of 0 (none) to 10 (worst possible). Pain relief was rated on a scale of 0% (none) to 100% (complete).
Compared to baseline scores, patients showed significant decreases at 3, 6 and 9 months for BPI worst pain (5.5 at baseline, 3.6 for 3, 6, and 9 months) average pain (4.1 at baseline, 2.4, 2.3, and 2.7 for 3, 6, and 9 months, respectively), overall pain severity (2.7 at baseline, 2.3, 2.3, and 2.4 at 3, 6, and 9 months, respectively), and pain interference with daily life (4.3 at baseline, 2.4, 2.2, and 2.4 at 3, 6, and 9 months, respectively; P less than .01 for all four pain measures).
“Pain severity as reported in the ESAS-r decreased significantly at 3-month, 6-month and 9-month follow-ups,” the researchers noted.
In addition, total medication burden based on the medication quantification scale (MQS) and morphine equivalent daily dose (MEDD) were recorded at 3, 6, 9, and 12 months. MQS scores decreased compared to baseline at 3, 6, 9, and 12 months in 10%, 23.5%, 26.2%, and 31.6% of patients, respectively. Also compared with baseline, 11.1%, 31.3%, and 14.3% of patients reported decreases in MEDD scores at 3, 6, and 9 months, respectively.
Overall, products with equal amounts of active ingredients tetrahydrocannabinol (THC) and cannabidiol (CBD) were more effective than were those with a predominance of either THC or CBD, the researchers wrote.
Medical cannabis was well-tolerated; a total of 15 moderate to severe side effects were reported by 11 patients, 13 of which were minor. The most common side effects were sleepiness and fatigue, and five patients discontinued their medical cannabis because of side effects. The two serious side effects reported during the study period – pneumonia and a cardiovascular event – were deemed unlikely related to the patients’ medicinal cannabis use.
The findings were limited by several factors, including the observational design, which prevented conclusions about causality, the researchers noted. Other limitations included the loss of many patients to follow-up and incomplete data on other prescription medications in many cases.
The results support the use of medical cannabis by cancer patients as an adjunct pain relief strategy and a way to potentially reduce the use of other medications such as opioids, the authors concluded.
The study was supported by the Canadian Consortium for the Investigation of Cannabinoids, Collège des Médecins du Québec, and the Canopy Growth Corporation. The researchers had no financial conflicts to disclose.
in a study.
Physician-prescribed cannabis, particularly cannabinoids, has been shown to ease cancer-related pain in adult cancer patients, who often find inadequate pain relief from medications including opioids, Saro Aprikian, MSc, a medical student at the Royal College of Surgeons, Dublin, and colleagues, wrote in their paper.
However, real-world data on the safety and effectiveness of cannabis in the cancer population and the impact on use of other medications are lacking, the researchers said.
In the study, published in BMJ Supportive & Palliative Care, the researchers reviewed data from 358 adults with cancer who were part of a multicenter cannabis registry in Canada between May 2015 and October 2018.
The average age of the patients was 57.6 years, and 48% were men. The top three cancer diagnoses in the study population were genitorurinary, breast, and colorectal.
Pain was the most common reason for obtaining a medical cannabis prescription, cited by 72.4% of patients.
Data were collected at follow-up visits conducted every 3 months over 1 year. Pain was assessed via the Brief Pain Inventory (BPI) and revised Edmonton Symptom Assessment System (ESAS-r) questionnaires and compared to baseline values. Patients rated their pain intensity on a sliding scale of 0 (none) to 10 (worst possible). Pain relief was rated on a scale of 0% (none) to 100% (complete).
Compared to baseline scores, patients showed significant decreases at 3, 6 and 9 months for BPI worst pain (5.5 at baseline, 3.6 for 3, 6, and 9 months) average pain (4.1 at baseline, 2.4, 2.3, and 2.7 for 3, 6, and 9 months, respectively), overall pain severity (2.7 at baseline, 2.3, 2.3, and 2.4 at 3, 6, and 9 months, respectively), and pain interference with daily life (4.3 at baseline, 2.4, 2.2, and 2.4 at 3, 6, and 9 months, respectively; P less than .01 for all four pain measures).
“Pain severity as reported in the ESAS-r decreased significantly at 3-month, 6-month and 9-month follow-ups,” the researchers noted.
In addition, total medication burden based on the medication quantification scale (MQS) and morphine equivalent daily dose (MEDD) were recorded at 3, 6, 9, and 12 months. MQS scores decreased compared to baseline at 3, 6, 9, and 12 months in 10%, 23.5%, 26.2%, and 31.6% of patients, respectively. Also compared with baseline, 11.1%, 31.3%, and 14.3% of patients reported decreases in MEDD scores at 3, 6, and 9 months, respectively.
Overall, products with equal amounts of active ingredients tetrahydrocannabinol (THC) and cannabidiol (CBD) were more effective than were those with a predominance of either THC or CBD, the researchers wrote.
Medical cannabis was well-tolerated; a total of 15 moderate to severe side effects were reported by 11 patients, 13 of which were minor. The most common side effects were sleepiness and fatigue, and five patients discontinued their medical cannabis because of side effects. The two serious side effects reported during the study period – pneumonia and a cardiovascular event – were deemed unlikely related to the patients’ medicinal cannabis use.
The findings were limited by several factors, including the observational design, which prevented conclusions about causality, the researchers noted. Other limitations included the loss of many patients to follow-up and incomplete data on other prescription medications in many cases.
The results support the use of medical cannabis by cancer patients as an adjunct pain relief strategy and a way to potentially reduce the use of other medications such as opioids, the authors concluded.
The study was supported by the Canadian Consortium for the Investigation of Cannabinoids, Collège des Médecins du Québec, and the Canopy Growth Corporation. The researchers had no financial conflicts to disclose.
in a study.
Physician-prescribed cannabis, particularly cannabinoids, has been shown to ease cancer-related pain in adult cancer patients, who often find inadequate pain relief from medications including opioids, Saro Aprikian, MSc, a medical student at the Royal College of Surgeons, Dublin, and colleagues, wrote in their paper.
However, real-world data on the safety and effectiveness of cannabis in the cancer population and the impact on use of other medications are lacking, the researchers said.
In the study, published in BMJ Supportive & Palliative Care, the researchers reviewed data from 358 adults with cancer who were part of a multicenter cannabis registry in Canada between May 2015 and October 2018.
The average age of the patients was 57.6 years, and 48% were men. The top three cancer diagnoses in the study population were genitorurinary, breast, and colorectal.
Pain was the most common reason for obtaining a medical cannabis prescription, cited by 72.4% of patients.
Data were collected at follow-up visits conducted every 3 months over 1 year. Pain was assessed via the Brief Pain Inventory (BPI) and revised Edmonton Symptom Assessment System (ESAS-r) questionnaires and compared to baseline values. Patients rated their pain intensity on a sliding scale of 0 (none) to 10 (worst possible). Pain relief was rated on a scale of 0% (none) to 100% (complete).
Compared to baseline scores, patients showed significant decreases at 3, 6 and 9 months for BPI worst pain (5.5 at baseline, 3.6 for 3, 6, and 9 months) average pain (4.1 at baseline, 2.4, 2.3, and 2.7 for 3, 6, and 9 months, respectively), overall pain severity (2.7 at baseline, 2.3, 2.3, and 2.4 at 3, 6, and 9 months, respectively), and pain interference with daily life (4.3 at baseline, 2.4, 2.2, and 2.4 at 3, 6, and 9 months, respectively; P less than .01 for all four pain measures).
“Pain severity as reported in the ESAS-r decreased significantly at 3-month, 6-month and 9-month follow-ups,” the researchers noted.
In addition, total medication burden based on the medication quantification scale (MQS) and morphine equivalent daily dose (MEDD) were recorded at 3, 6, 9, and 12 months. MQS scores decreased compared to baseline at 3, 6, 9, and 12 months in 10%, 23.5%, 26.2%, and 31.6% of patients, respectively. Also compared with baseline, 11.1%, 31.3%, and 14.3% of patients reported decreases in MEDD scores at 3, 6, and 9 months, respectively.
Overall, products with equal amounts of active ingredients tetrahydrocannabinol (THC) and cannabidiol (CBD) were more effective than were those with a predominance of either THC or CBD, the researchers wrote.
Medical cannabis was well-tolerated; a total of 15 moderate to severe side effects were reported by 11 patients, 13 of which were minor. The most common side effects were sleepiness and fatigue, and five patients discontinued their medical cannabis because of side effects. The two serious side effects reported during the study period – pneumonia and a cardiovascular event – were deemed unlikely related to the patients’ medicinal cannabis use.
The findings were limited by several factors, including the observational design, which prevented conclusions about causality, the researchers noted. Other limitations included the loss of many patients to follow-up and incomplete data on other prescription medications in many cases.
The results support the use of medical cannabis by cancer patients as an adjunct pain relief strategy and a way to potentially reduce the use of other medications such as opioids, the authors concluded.
The study was supported by the Canadian Consortium for the Investigation of Cannabinoids, Collège des Médecins du Québec, and the Canopy Growth Corporation. The researchers had no financial conflicts to disclose.
FROM BMJ SUPPORTIVE & PALLIATIVE CARE
Head-to-head comparison of migraine meds reveals top options
BOSTON – , a new real-world analysis of data on more than 3 million migraine attacks shows.
The findings “align with results of clinical trials and recommendations from clinical treatment guidelines” and provide insights to complement clinical practice, said study investigator Chia-Chun Chiang, MD, a neurologist with Mayo Clinic, Rochester, Minn.
The findings were presented at the 2023 annual meeting of the American Academy of Neurology.
The power of big data
Despite a wide variety of acute migraine medications that are available, large-scale, head-to-head comparisons of treatment effectiveness from real-world patient experience reports are lacking, Dr. Chiang explained.
“To the best of our knowledge, this is the first study that simultaneously compared multiple acute migraine medications using a Big Data analysis approach based on real-world patient-provided data,” she said.
The researchers extracted more than 10 million self-reported migraine attack records from a migraine smartphone app called Migraine Buddy, where users can document whether a treatment was helpful, somewhat helpful, unsure, or unhelpful.
They analyzed 25 acute medications among seven classes: acetaminophen, nonsteroidal anti-inflammatory drugs (NSAIDs), triptans, combination analgesics (acetaminophen/aspirin/caffeine), ergots, antiemetics, and opioids. The newer gepants and ditan medication classes of medications were not included because of the relatively lower numbers of usage when data was extracted (2014-2020).
The researchers employed a two-level nested logistic regression model to analyze the odds of treatment effectiveness of each medication by adjusting concurrent medications and the covariance within the same user.
The final analysis included more than 3.1 million migraine attacks among 278,000 users globally.
Using ibuprofen as the reference, triptans, ergots, and antiemetics had the highest efficacy with mean odds ratios of 4.8, 3.02, and 2.67, respectively, followed by opioids (OR, 2.49), NSAIDs (OR, 1.94), combination analgesics (OR, 1.69), others (OR, 1.49), and acetaminophen (OR, 0.83).
Individual medications with the highest patient-reported effectiveness were eletriptan (Relpax; OR, 6.1), zolmitriptan (Zomig; OR, 5.7) and sumatriptan (Zecuity; OR, 5.2).
This migraine medication comparative effectiveness analysis, based on patient-reported outcomes, “supports and complements the treatment recommendations from national headache societies based on randomized controlled trials and meta-analyses and strongly supports the use of triptans,” Dr. Chiang said.
End of trial-and-error?
Commenting on this research, Shaheen Lakhan, MD, PhD, a neurologist and researcher in Boston, said “This is a great study of Big Data in that it shows the power of the smartphone to collect real-world data and smart researchers like at Mayo Clinic to analyze them.”
“The study sheds light on how different therapeutics compare with each other. The next iteration of this line of research, I would hope, would be to determine if particular medications are effective for a particular migraine population, and even down to individuals with migraine,” said Dr. Lakhan, who wasn’t involved in the study.
“Once those models are appropriately built, long gone will be the era of trial-and-error medicine,” Dr. Lakhan added.
The study had no specific funding. Dr. Chiang has served as a consultant for Satsuma. Dr. Lakhan reports no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
BOSTON – , a new real-world analysis of data on more than 3 million migraine attacks shows.
The findings “align with results of clinical trials and recommendations from clinical treatment guidelines” and provide insights to complement clinical practice, said study investigator Chia-Chun Chiang, MD, a neurologist with Mayo Clinic, Rochester, Minn.
The findings were presented at the 2023 annual meeting of the American Academy of Neurology.
The power of big data
Despite a wide variety of acute migraine medications that are available, large-scale, head-to-head comparisons of treatment effectiveness from real-world patient experience reports are lacking, Dr. Chiang explained.
“To the best of our knowledge, this is the first study that simultaneously compared multiple acute migraine medications using a Big Data analysis approach based on real-world patient-provided data,” she said.
The researchers extracted more than 10 million self-reported migraine attack records from a migraine smartphone app called Migraine Buddy, where users can document whether a treatment was helpful, somewhat helpful, unsure, or unhelpful.
They analyzed 25 acute medications among seven classes: acetaminophen, nonsteroidal anti-inflammatory drugs (NSAIDs), triptans, combination analgesics (acetaminophen/aspirin/caffeine), ergots, antiemetics, and opioids. The newer gepants and ditan medication classes of medications were not included because of the relatively lower numbers of usage when data was extracted (2014-2020).
The researchers employed a two-level nested logistic regression model to analyze the odds of treatment effectiveness of each medication by adjusting concurrent medications and the covariance within the same user.
The final analysis included more than 3.1 million migraine attacks among 278,000 users globally.
Using ibuprofen as the reference, triptans, ergots, and antiemetics had the highest efficacy with mean odds ratios of 4.8, 3.02, and 2.67, respectively, followed by opioids (OR, 2.49), NSAIDs (OR, 1.94), combination analgesics (OR, 1.69), others (OR, 1.49), and acetaminophen (OR, 0.83).
Individual medications with the highest patient-reported effectiveness were eletriptan (Relpax; OR, 6.1), zolmitriptan (Zomig; OR, 5.7) and sumatriptan (Zecuity; OR, 5.2).
This migraine medication comparative effectiveness analysis, based on patient-reported outcomes, “supports and complements the treatment recommendations from national headache societies based on randomized controlled trials and meta-analyses and strongly supports the use of triptans,” Dr. Chiang said.
End of trial-and-error?
Commenting on this research, Shaheen Lakhan, MD, PhD, a neurologist and researcher in Boston, said “This is a great study of Big Data in that it shows the power of the smartphone to collect real-world data and smart researchers like at Mayo Clinic to analyze them.”
“The study sheds light on how different therapeutics compare with each other. The next iteration of this line of research, I would hope, would be to determine if particular medications are effective for a particular migraine population, and even down to individuals with migraine,” said Dr. Lakhan, who wasn’t involved in the study.
“Once those models are appropriately built, long gone will be the era of trial-and-error medicine,” Dr. Lakhan added.
The study had no specific funding. Dr. Chiang has served as a consultant for Satsuma. Dr. Lakhan reports no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
BOSTON – , a new real-world analysis of data on more than 3 million migraine attacks shows.
The findings “align with results of clinical trials and recommendations from clinical treatment guidelines” and provide insights to complement clinical practice, said study investigator Chia-Chun Chiang, MD, a neurologist with Mayo Clinic, Rochester, Minn.
The findings were presented at the 2023 annual meeting of the American Academy of Neurology.
The power of big data
Despite a wide variety of acute migraine medications that are available, large-scale, head-to-head comparisons of treatment effectiveness from real-world patient experience reports are lacking, Dr. Chiang explained.
“To the best of our knowledge, this is the first study that simultaneously compared multiple acute migraine medications using a Big Data analysis approach based on real-world patient-provided data,” she said.
The researchers extracted more than 10 million self-reported migraine attack records from a migraine smartphone app called Migraine Buddy, where users can document whether a treatment was helpful, somewhat helpful, unsure, or unhelpful.
They analyzed 25 acute medications among seven classes: acetaminophen, nonsteroidal anti-inflammatory drugs (NSAIDs), triptans, combination analgesics (acetaminophen/aspirin/caffeine), ergots, antiemetics, and opioids. The newer gepants and ditan medication classes of medications were not included because of the relatively lower numbers of usage when data was extracted (2014-2020).
The researchers employed a two-level nested logistic regression model to analyze the odds of treatment effectiveness of each medication by adjusting concurrent medications and the covariance within the same user.
The final analysis included more than 3.1 million migraine attacks among 278,000 users globally.
Using ibuprofen as the reference, triptans, ergots, and antiemetics had the highest efficacy with mean odds ratios of 4.8, 3.02, and 2.67, respectively, followed by opioids (OR, 2.49), NSAIDs (OR, 1.94), combination analgesics (OR, 1.69), others (OR, 1.49), and acetaminophen (OR, 0.83).
Individual medications with the highest patient-reported effectiveness were eletriptan (Relpax; OR, 6.1), zolmitriptan (Zomig; OR, 5.7) and sumatriptan (Zecuity; OR, 5.2).
This migraine medication comparative effectiveness analysis, based on patient-reported outcomes, “supports and complements the treatment recommendations from national headache societies based on randomized controlled trials and meta-analyses and strongly supports the use of triptans,” Dr. Chiang said.
End of trial-and-error?
Commenting on this research, Shaheen Lakhan, MD, PhD, a neurologist and researcher in Boston, said “This is a great study of Big Data in that it shows the power of the smartphone to collect real-world data and smart researchers like at Mayo Clinic to analyze them.”
“The study sheds light on how different therapeutics compare with each other. The next iteration of this line of research, I would hope, would be to determine if particular medications are effective for a particular migraine population, and even down to individuals with migraine,” said Dr. Lakhan, who wasn’t involved in the study.
“Once those models are appropriately built, long gone will be the era of trial-and-error medicine,” Dr. Lakhan added.
The study had no specific funding. Dr. Chiang has served as a consultant for Satsuma. Dr. Lakhan reports no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
AT AAN 2023
FDA expands atogepant approval to include chronic migraine
The approval makes atogepant the first, and only, oral calcitonin gene-related peptide receptor antagonist approved to prevent migraine across frequencies, including episodic and chronic, the company said in a news release.
The FDA initially approved atogepant in 2021 for the prevention of episodic migraine in adults.
Once-daily atogepant is available in three doses – 10 mg, 30 mg, and 60 mg – for prevention of episodic migraine. However, only the 60-mg dose of medication is indicated for the preventive treatment of chronic migraine.
The expanded indication in chronic migraine is based on positive results of the phase 3 PROGRESS trial, which evaluated atogepant in more than 700 adults with chronic migraine.
The trial met the primary endpoint of statistically significant reduction from baseline in mean monthly migraine days with atogepant compared with placebo across the 12-week treatment period.
Treatment with atogepant also led to statistically significant improvements in all six secondary endpoints, including the proportion of patients that achieved at least a 50% reduction in mean monthly migraine days across 12 weeks and improvements in function and reduction in activity impairment caused by migraine.
The efficacy results are consistent with those in the ADVANCE episodic migraine trial.
The overall safety profile of atogepant is consistent with the episodic migraine patient population, with the most common adverse events including constipation, nausea, and fatigue/sleepiness.
“The FDA approval is an important milestone, providing those most impacted by migraine with a new, safe, and effective treatment option in a convenient, once-daily pill,” Peter McAllister, MD, director of the New England Center for Neurology and Headache, Stamford, Conn., said in the news release.
The data demonstrate that atogepant “helps reduce the burden of migraine by delivering improvements in function, with high response rates and sustained efficacy over 12 weeks. These are critical factors neurologists and headache specialists consider when prescribing a treatment option, particularly for those with chronic migraine,” Dr. McAllister added.
A version of this article originally appeared on Medscape.com.
The approval makes atogepant the first, and only, oral calcitonin gene-related peptide receptor antagonist approved to prevent migraine across frequencies, including episodic and chronic, the company said in a news release.
The FDA initially approved atogepant in 2021 for the prevention of episodic migraine in adults.
Once-daily atogepant is available in three doses – 10 mg, 30 mg, and 60 mg – for prevention of episodic migraine. However, only the 60-mg dose of medication is indicated for the preventive treatment of chronic migraine.
The expanded indication in chronic migraine is based on positive results of the phase 3 PROGRESS trial, which evaluated atogepant in more than 700 adults with chronic migraine.
The trial met the primary endpoint of statistically significant reduction from baseline in mean monthly migraine days with atogepant compared with placebo across the 12-week treatment period.
Treatment with atogepant also led to statistically significant improvements in all six secondary endpoints, including the proportion of patients that achieved at least a 50% reduction in mean monthly migraine days across 12 weeks and improvements in function and reduction in activity impairment caused by migraine.
The efficacy results are consistent with those in the ADVANCE episodic migraine trial.
The overall safety profile of atogepant is consistent with the episodic migraine patient population, with the most common adverse events including constipation, nausea, and fatigue/sleepiness.
“The FDA approval is an important milestone, providing those most impacted by migraine with a new, safe, and effective treatment option in a convenient, once-daily pill,” Peter McAllister, MD, director of the New England Center for Neurology and Headache, Stamford, Conn., said in the news release.
The data demonstrate that atogepant “helps reduce the burden of migraine by delivering improvements in function, with high response rates and sustained efficacy over 12 weeks. These are critical factors neurologists and headache specialists consider when prescribing a treatment option, particularly for those with chronic migraine,” Dr. McAllister added.
A version of this article originally appeared on Medscape.com.
The approval makes atogepant the first, and only, oral calcitonin gene-related peptide receptor antagonist approved to prevent migraine across frequencies, including episodic and chronic, the company said in a news release.
The FDA initially approved atogepant in 2021 for the prevention of episodic migraine in adults.
Once-daily atogepant is available in three doses – 10 mg, 30 mg, and 60 mg – for prevention of episodic migraine. However, only the 60-mg dose of medication is indicated for the preventive treatment of chronic migraine.
The expanded indication in chronic migraine is based on positive results of the phase 3 PROGRESS trial, which evaluated atogepant in more than 700 adults with chronic migraine.
The trial met the primary endpoint of statistically significant reduction from baseline in mean monthly migraine days with atogepant compared with placebo across the 12-week treatment period.
Treatment with atogepant also led to statistically significant improvements in all six secondary endpoints, including the proportion of patients that achieved at least a 50% reduction in mean monthly migraine days across 12 weeks and improvements in function and reduction in activity impairment caused by migraine.
The efficacy results are consistent with those in the ADVANCE episodic migraine trial.
The overall safety profile of atogepant is consistent with the episodic migraine patient population, with the most common adverse events including constipation, nausea, and fatigue/sleepiness.
“The FDA approval is an important milestone, providing those most impacted by migraine with a new, safe, and effective treatment option in a convenient, once-daily pill,” Peter McAllister, MD, director of the New England Center for Neurology and Headache, Stamford, Conn., said in the news release.
The data demonstrate that atogepant “helps reduce the burden of migraine by delivering improvements in function, with high response rates and sustained efficacy over 12 weeks. These are critical factors neurologists and headache specialists consider when prescribing a treatment option, particularly for those with chronic migraine,” Dr. McAllister added.
A version of this article originally appeared on Medscape.com.
Musculoskeletal disorders prevalent in orchestra musicians
PARIS – For orchestra musicians, performance is everything. So, it’s no wonder that musculoskeletal disorders – a reality for so many of these professionals – are not openly discussed. Physical pain is often pushed aside, unexpressed, until one day the suffering gets to be too much, the ability to play is impacted, and all the effort to keep things under wraps and under control culminates in burnout.
Anne Maugue was one of the speakers at the French College of General Medicine’s 16th Congress of General Medicine. Ms. Maugue is a postdoctoral researcher at Côte d’Azur University, Nice, France. She also plays flute in the Monte-Carlo Philharmonic Orchestra. Through her presentation to the physicians, she sought to raise awareness about MSDs in professional musicians, as well as the associated psychosocial risk factors. “If caught early enough, this pain can often be successfully treated.”
High prevalence
“You’re a violinist in a major symphony orchestra. It’s Sunday night, 8 o’clock, and you’ve just come off the stage. A few minutes ago, you felt a sharp pain in your right arm – a pain that is now, already, overwhelming. The conductor accused you of not being focused, of not concentrating. You know that you have another rehearsal in just a few hours, Monday morning. So, what do you do – other than hope that the pain goes away by then? Where can you turn to get help?”
With this opening scenario, Ms. Maugue was able to immediately orient the attendees to the realities that professional musicians face.
Pain is far from anecdotal. In professional orchestras, its prevalence over 12 months is between 41% and 93%. “An elite athlete has a full training staff they can turn to. An elite musician, on the other hand, usually only has their general practitioner – and that’s assuming the musician even reaches out to get treatment to begin with.
“The fact is that most of the time musicians only care about the pain when it becomes chronic, when it causes discomfort that affects their playing,” said Ms. Maugue.
How, then, does one evaluate this problem? In a Danish study, musicians rated the musculoskeletal problems they had experienced in the preceding 7 days. When the researchers compared those reports with findings from a clinical examination, they found that the examiners were not able to identify which musicians had reported problems. Why? Because a diagnosis does not reflect the severity or the impact, both of which are subjective.
“When faced with pain, the musician’s initial reaction is denial,” said Ms. Maugue. “The pain is often attributed to something other than the physicality of playing their instrument. They then turn to self-care, to colleagues. It’s only much later that they consult a medical professional.”
As a result, the physician is seldom aware of the musician’s psychological distress and has no sense of how long it’s been since the pain first started.
Work environment
Carrying around an instrument all the time and maintaining nonergonomic postures for extended periods are just two of the factors that put professional musicians at risk of physical pain. Not to be forgotten, Ms. Maugue added, are the work-related pressures. Musicians are not immune to issues with their work environment. They can feel like they aren’t getting the resources they need, proper recognition from their leaders, or support from their colleagues. In the end, such feelings can engender a sense of unfairness – and that acts as a stressor that can give rise to MSDs.
Evidence of this phenomenon can be found in the results of a study that Ms. Maugue conducted. Out of 440 French orchestra musicians (44% women), 64% said they had experienced MSD-related pain in the preceding 12 months and 61% in the preceding 7 days.
Using industrial and organizational psychology scales of measurement, Ms. Maugue was able to show, through hierarchical regression, that “emotional exhaustion and MSD-related pain occur when the environment in which people work causes them to feel a sense of unfairness.”
Early detection
Finally, Ms. Maugue encouraged general practitioners to ask every patient whether he or she plays a musical instrument. If the answer is yes, get an idea about any pain that he or she may have been feeling in the back, neck, and upper extremities so that prompt treatment can be given.
“There are other studies underway that are looking to better characterize instrumental activity and to enable more effective management by sports medicine departments,” said Ms. Maugue. “But back to patients with MSDs. It’s important to understand everything about their playing. Where do they practice? How often do they practice? What’s their posture like when they play? What’s the tempo of the music they’re working on? Because what we see in professional musicians is likely to be seen in amateur musicians as well – particularly in young people who study at a conservatory,” where not much is being done to prevent MSDs.
“If professional musicians are given treatment early on, half of them can be permanently cured,” she concluded. “And then, just like elite athletes, they’ll be able to get right back to playing.”
This article was translated from Medscape’s French edition and a version appeared on Medscape.com.
PARIS – For orchestra musicians, performance is everything. So, it’s no wonder that musculoskeletal disorders – a reality for so many of these professionals – are not openly discussed. Physical pain is often pushed aside, unexpressed, until one day the suffering gets to be too much, the ability to play is impacted, and all the effort to keep things under wraps and under control culminates in burnout.
Anne Maugue was one of the speakers at the French College of General Medicine’s 16th Congress of General Medicine. Ms. Maugue is a postdoctoral researcher at Côte d’Azur University, Nice, France. She also plays flute in the Monte-Carlo Philharmonic Orchestra. Through her presentation to the physicians, she sought to raise awareness about MSDs in professional musicians, as well as the associated psychosocial risk factors. “If caught early enough, this pain can often be successfully treated.”
High prevalence
“You’re a violinist in a major symphony orchestra. It’s Sunday night, 8 o’clock, and you’ve just come off the stage. A few minutes ago, you felt a sharp pain in your right arm – a pain that is now, already, overwhelming. The conductor accused you of not being focused, of not concentrating. You know that you have another rehearsal in just a few hours, Monday morning. So, what do you do – other than hope that the pain goes away by then? Where can you turn to get help?”
With this opening scenario, Ms. Maugue was able to immediately orient the attendees to the realities that professional musicians face.
Pain is far from anecdotal. In professional orchestras, its prevalence over 12 months is between 41% and 93%. “An elite athlete has a full training staff they can turn to. An elite musician, on the other hand, usually only has their general practitioner – and that’s assuming the musician even reaches out to get treatment to begin with.
“The fact is that most of the time musicians only care about the pain when it becomes chronic, when it causes discomfort that affects their playing,” said Ms. Maugue.
How, then, does one evaluate this problem? In a Danish study, musicians rated the musculoskeletal problems they had experienced in the preceding 7 days. When the researchers compared those reports with findings from a clinical examination, they found that the examiners were not able to identify which musicians had reported problems. Why? Because a diagnosis does not reflect the severity or the impact, both of which are subjective.
“When faced with pain, the musician’s initial reaction is denial,” said Ms. Maugue. “The pain is often attributed to something other than the physicality of playing their instrument. They then turn to self-care, to colleagues. It’s only much later that they consult a medical professional.”
As a result, the physician is seldom aware of the musician’s psychological distress and has no sense of how long it’s been since the pain first started.
Work environment
Carrying around an instrument all the time and maintaining nonergonomic postures for extended periods are just two of the factors that put professional musicians at risk of physical pain. Not to be forgotten, Ms. Maugue added, are the work-related pressures. Musicians are not immune to issues with their work environment. They can feel like they aren’t getting the resources they need, proper recognition from their leaders, or support from their colleagues. In the end, such feelings can engender a sense of unfairness – and that acts as a stressor that can give rise to MSDs.
Evidence of this phenomenon can be found in the results of a study that Ms. Maugue conducted. Out of 440 French orchestra musicians (44% women), 64% said they had experienced MSD-related pain in the preceding 12 months and 61% in the preceding 7 days.
Using industrial and organizational psychology scales of measurement, Ms. Maugue was able to show, through hierarchical regression, that “emotional exhaustion and MSD-related pain occur when the environment in which people work causes them to feel a sense of unfairness.”
Early detection
Finally, Ms. Maugue encouraged general practitioners to ask every patient whether he or she plays a musical instrument. If the answer is yes, get an idea about any pain that he or she may have been feeling in the back, neck, and upper extremities so that prompt treatment can be given.
“There are other studies underway that are looking to better characterize instrumental activity and to enable more effective management by sports medicine departments,” said Ms. Maugue. “But back to patients with MSDs. It’s important to understand everything about their playing. Where do they practice? How often do they practice? What’s their posture like when they play? What’s the tempo of the music they’re working on? Because what we see in professional musicians is likely to be seen in amateur musicians as well – particularly in young people who study at a conservatory,” where not much is being done to prevent MSDs.
“If professional musicians are given treatment early on, half of them can be permanently cured,” she concluded. “And then, just like elite athletes, they’ll be able to get right back to playing.”
This article was translated from Medscape’s French edition and a version appeared on Medscape.com.
PARIS – For orchestra musicians, performance is everything. So, it’s no wonder that musculoskeletal disorders – a reality for so many of these professionals – are not openly discussed. Physical pain is often pushed aside, unexpressed, until one day the suffering gets to be too much, the ability to play is impacted, and all the effort to keep things under wraps and under control culminates in burnout.
Anne Maugue was one of the speakers at the French College of General Medicine’s 16th Congress of General Medicine. Ms. Maugue is a postdoctoral researcher at Côte d’Azur University, Nice, France. She also plays flute in the Monte-Carlo Philharmonic Orchestra. Through her presentation to the physicians, she sought to raise awareness about MSDs in professional musicians, as well as the associated psychosocial risk factors. “If caught early enough, this pain can often be successfully treated.”
High prevalence
“You’re a violinist in a major symphony orchestra. It’s Sunday night, 8 o’clock, and you’ve just come off the stage. A few minutes ago, you felt a sharp pain in your right arm – a pain that is now, already, overwhelming. The conductor accused you of not being focused, of not concentrating. You know that you have another rehearsal in just a few hours, Monday morning. So, what do you do – other than hope that the pain goes away by then? Where can you turn to get help?”
With this opening scenario, Ms. Maugue was able to immediately orient the attendees to the realities that professional musicians face.
Pain is far from anecdotal. In professional orchestras, its prevalence over 12 months is between 41% and 93%. “An elite athlete has a full training staff they can turn to. An elite musician, on the other hand, usually only has their general practitioner – and that’s assuming the musician even reaches out to get treatment to begin with.
“The fact is that most of the time musicians only care about the pain when it becomes chronic, when it causes discomfort that affects their playing,” said Ms. Maugue.
How, then, does one evaluate this problem? In a Danish study, musicians rated the musculoskeletal problems they had experienced in the preceding 7 days. When the researchers compared those reports with findings from a clinical examination, they found that the examiners were not able to identify which musicians had reported problems. Why? Because a diagnosis does not reflect the severity or the impact, both of which are subjective.
“When faced with pain, the musician’s initial reaction is denial,” said Ms. Maugue. “The pain is often attributed to something other than the physicality of playing their instrument. They then turn to self-care, to colleagues. It’s only much later that they consult a medical professional.”
As a result, the physician is seldom aware of the musician’s psychological distress and has no sense of how long it’s been since the pain first started.
Work environment
Carrying around an instrument all the time and maintaining nonergonomic postures for extended periods are just two of the factors that put professional musicians at risk of physical pain. Not to be forgotten, Ms. Maugue added, are the work-related pressures. Musicians are not immune to issues with their work environment. They can feel like they aren’t getting the resources they need, proper recognition from their leaders, or support from their colleagues. In the end, such feelings can engender a sense of unfairness – and that acts as a stressor that can give rise to MSDs.
Evidence of this phenomenon can be found in the results of a study that Ms. Maugue conducted. Out of 440 French orchestra musicians (44% women), 64% said they had experienced MSD-related pain in the preceding 12 months and 61% in the preceding 7 days.
Using industrial and organizational psychology scales of measurement, Ms. Maugue was able to show, through hierarchical regression, that “emotional exhaustion and MSD-related pain occur when the environment in which people work causes them to feel a sense of unfairness.”
Early detection
Finally, Ms. Maugue encouraged general practitioners to ask every patient whether he or she plays a musical instrument. If the answer is yes, get an idea about any pain that he or she may have been feeling in the back, neck, and upper extremities so that prompt treatment can be given.
“There are other studies underway that are looking to better characterize instrumental activity and to enable more effective management by sports medicine departments,” said Ms. Maugue. “But back to patients with MSDs. It’s important to understand everything about their playing. Where do they practice? How often do they practice? What’s their posture like when they play? What’s the tempo of the music they’re working on? Because what we see in professional musicians is likely to be seen in amateur musicians as well – particularly in young people who study at a conservatory,” where not much is being done to prevent MSDs.
“If professional musicians are given treatment early on, half of them can be permanently cured,” she concluded. “And then, just like elite athletes, they’ll be able to get right back to playing.”
This article was translated from Medscape’s French edition and a version appeared on Medscape.com.
Spinal cord stimulation may help diabetic neuropathy
according to research that released early, prior to its presentation at the annual meeting of the American Academy of Neurology.
The data represents the longest follow-up available for spinal cord stimulation at a frequency higher than the 60 Hz initially approved for diabetic neuropathy by the Food and Drug Administration, according to lead author Erika A. Petersen, MD, a professor of neurosurgery and the residency program director at the University of Arkansas for Medical Sciences, Little Rock.
“You would expect that somebody who continues to have diabetes for 24 months and has neuropathy would have worse neuropathy after 2 years, and what we’re seeing is that people were stable or better in terms of their nerve function at 2 years,” Dr. Petersen said in an interview. “So that’s really revolutionary.”
Encouraging preliminary findings
The findings are “promising and preliminary,” John D. Markman, MD, a professor in neurology and neurosurgery, vice chair for clinical research, and director of the Translational Pain Research Program at the University of Rochester (N.Y.) Medical Center, said in an interview. Dr. Markman, who was not involved in this study, said that, though the results are encouraging, it’s “less clear how much of [the pain improvement] is due to what we would consider to be on-target, pain-relieving benefit from stimulation versus other factors like expectation.” The crossover rate and amount of reduction in pain intensity are promising, but “I think that excitement is weighed against the fact that this is an open-label study.”
An underused treatment
Although spinal cord stimulation has been around since the late 1960s, its use only picked up steam in the 2000s, when it became more frequently used to treat chronic nerve damage related to neuropathic pain syndromes, Dr. Petersen explained. The FDA approved the treatment’s new indication for diabetic neuropathy in 2015, and data from Abbott and Medtronic have shown benefits from spinal cord stimulation at 60 Hz, but some patients are uncomfortable with the vibration or tingling feelings the devices can cause at that frequency.
“They describe creepy crawlies or ants crawling over the feet, or pins and needles, and painful sensitivity,” Dr. Petersen said. “You create a vibration feeling in the same zone where they already have those feelings of buzzing and pain and vibration, and it’s sometimes actually even more uncomfortable and less satisfying to them in terms of relief” with the spinal cord stimulation at 60 Hz, she said, “so there’s a lot of attrition in terms of who will actually use it.”
At 10 kHz, however, “people don’t feel any vibration or tingling associated with it; it just jams the signal of the pain,” she said. The difference between the frequencies is like that between “a lifeguard whistle and a dog whistle.”
Testing high-frequency stimulation
The new findings included the 24-month follow-up data from a randomized controlled trial that assessed the effectiveness of high-frequency spinal cord stimulation for painful diabetic neuropathy. The original 216 participants enrolled in the trial had diabetic neuropathy symptoms for at least 12 months and either could no not tolerate or did not respond to medications. Enrollment criteria also included lower-limb pain intensity of at least 5 on a 0-10 visual analogy scale and hemoglobin A1c of no more than 10%.
For the first 6 months of the trial – before crossover was offered – participants were randomly assigned to receive either 10 kHz of spinal cord stimulation along with conventional medical management or to receive conventional medical management alone. The 6-month data from 187 patients, as reported in April 2021 in JAMA Neurology, revealed that 79% of those receiving spinal cord stimulation experienced at least 50% improved pain relief without worsening of their baseline neurologic deficits, compared with only 5% of those receiving only conventional treatments.
Average pain levels increased 2% in the control participants compared with a decrease of 76% in those with the spinal cord stimulation devices. In addition, 62% of the patients receiving spinal cord stimulation demonstration neurologic improvement in reflexes, strength, movement and sensation, compared with 3% of those in the control group. The study’s findings led the FDA to approve the device using 10 kHz.
At 6 months, 93% of control patients crossed over to receiving spinal cord stimulation while none with the devices opted to stop their spinal cord stimulation. The 12-month data revealed that 85% of those receiving spinal cord stimulation experienced at least 50% pain relief, with the average pain relief at 74%. Patients also reported statistically significant improved quality of life as well as less interference with sleep, mood, and daily activities from pain.
Two years after baseline, patients’ pain relief was maintained with average 80% improvement, and 66% of patients showed neurologic improvement since baseline. Though no patients had devices removed because of ineffectiveness, five patients’ devices were removed because of infection while infections in three other patients resolved.
“Being able to offer something that is not a pharmaceutical, without the side effects, that shows an even longer durability to that response is a really important finding at this point,” Dr. Petersen said.
Surgical considerations
Among the estimated 37 million Americans with type 1 or 2 diabetes, approximately one quarter of them experience some level of painful diabetic neuropathy, but medication and other medical management strategies are not always adequate in treating their pain. After a 1-week trial of spinal cord stimulation, the devices are implanted under the skin and rechargeable through the skin for up to 10 years, after which they can be replaced.
An appropriate candidate for spinal cord stimulation would be someone for whom existing non-invasive pain relief options, including medications, are ineffective or intolerable, Dr. Petersen and Dr. Markman both said. An adequate trial of medication is not “one size fits all” and will vary by each patient, added Dr. Markman, who is also interested in whether this study’s participants were able to have a reduction in use of pain relief medications.
“I think there’s a significant number of patients out there who can benefit from this, so I think that’s why it’s promising and exciting,” Dr. Markman said. “I do think it’s important to see if this actually allows them to be on less medication or whether stimulation turns out to be another treatment in addition to their baseline treatments.” The challenge is identifying “which patients are most likely to be benefiting from this and which are most likely to be harmed.”
Aside from infection from implantation, other possible risks include pain at the battery site and, in rare cases, a need for reoperation because of migration of the leads, he said.
Improvement in symptom severity and quality of life
After the wound from the implant has completely healed, Dr. Petersen said patients using the devices do not have any activity restrictions outside of magnetic interference, such as MRIs. “I’ve had people go back-country kayaking, scuba diving, fishing with their grandkids, all sorts of all sorts of things. If patients need to go through a scanner of any kind, they should ask whether it’s safe for pacemakers since these devices are like a “pacemaker for pain.
“I had a patient bring solar chargers with him so that he could recharge his battery in the backwoods while kayaking because that’s the level of improvement in pain that he got – from barely being able to walk down the hall to feeling comfortable being off the grid and active again,” Dr. Petersen said. “Those kinds of improvements in quality of life are massive.”
The study findings may also suggest that spinal cord stimulation can benefit a broader population of patients experiencing neuropathic pain, Dr. Markman said.
“There’s an extraordinary unmet need for treatments for neuropathy, and one important question here is the extent to which diabetic peripheral neuropathy and the response that we’re seeing here is a proxy for a broader effect across many neuropathies that are caused by other conditions other than diabetes,” Dr. Markman said. “There’s a lot of reason to think that this will be helpful not just for diabetes-related neuropathic pain, but for other types of neuropathic pain that have similar clinical presentations or clinical symptom patterns to diabetic peripheral neuropathy.”
The study was funded by Nevro, who manufactures the devices. Dr. Petersen and Dr. Markman both reported consulting with, receiving support from, holding stock options with, and serving on the data safety monitoring boards and advisory boards of numerous pharmaceutical companies.
according to research that released early, prior to its presentation at the annual meeting of the American Academy of Neurology.
The data represents the longest follow-up available for spinal cord stimulation at a frequency higher than the 60 Hz initially approved for diabetic neuropathy by the Food and Drug Administration, according to lead author Erika A. Petersen, MD, a professor of neurosurgery and the residency program director at the University of Arkansas for Medical Sciences, Little Rock.
“You would expect that somebody who continues to have diabetes for 24 months and has neuropathy would have worse neuropathy after 2 years, and what we’re seeing is that people were stable or better in terms of their nerve function at 2 years,” Dr. Petersen said in an interview. “So that’s really revolutionary.”
Encouraging preliminary findings
The findings are “promising and preliminary,” John D. Markman, MD, a professor in neurology and neurosurgery, vice chair for clinical research, and director of the Translational Pain Research Program at the University of Rochester (N.Y.) Medical Center, said in an interview. Dr. Markman, who was not involved in this study, said that, though the results are encouraging, it’s “less clear how much of [the pain improvement] is due to what we would consider to be on-target, pain-relieving benefit from stimulation versus other factors like expectation.” The crossover rate and amount of reduction in pain intensity are promising, but “I think that excitement is weighed against the fact that this is an open-label study.”
An underused treatment
Although spinal cord stimulation has been around since the late 1960s, its use only picked up steam in the 2000s, when it became more frequently used to treat chronic nerve damage related to neuropathic pain syndromes, Dr. Petersen explained. The FDA approved the treatment’s new indication for diabetic neuropathy in 2015, and data from Abbott and Medtronic have shown benefits from spinal cord stimulation at 60 Hz, but some patients are uncomfortable with the vibration or tingling feelings the devices can cause at that frequency.
“They describe creepy crawlies or ants crawling over the feet, or pins and needles, and painful sensitivity,” Dr. Petersen said. “You create a vibration feeling in the same zone where they already have those feelings of buzzing and pain and vibration, and it’s sometimes actually even more uncomfortable and less satisfying to them in terms of relief” with the spinal cord stimulation at 60 Hz, she said, “so there’s a lot of attrition in terms of who will actually use it.”
At 10 kHz, however, “people don’t feel any vibration or tingling associated with it; it just jams the signal of the pain,” she said. The difference between the frequencies is like that between “a lifeguard whistle and a dog whistle.”
Testing high-frequency stimulation
The new findings included the 24-month follow-up data from a randomized controlled trial that assessed the effectiveness of high-frequency spinal cord stimulation for painful diabetic neuropathy. The original 216 participants enrolled in the trial had diabetic neuropathy symptoms for at least 12 months and either could no not tolerate or did not respond to medications. Enrollment criteria also included lower-limb pain intensity of at least 5 on a 0-10 visual analogy scale and hemoglobin A1c of no more than 10%.
For the first 6 months of the trial – before crossover was offered – participants were randomly assigned to receive either 10 kHz of spinal cord stimulation along with conventional medical management or to receive conventional medical management alone. The 6-month data from 187 patients, as reported in April 2021 in JAMA Neurology, revealed that 79% of those receiving spinal cord stimulation experienced at least 50% improved pain relief without worsening of their baseline neurologic deficits, compared with only 5% of those receiving only conventional treatments.
Average pain levels increased 2% in the control participants compared with a decrease of 76% in those with the spinal cord stimulation devices. In addition, 62% of the patients receiving spinal cord stimulation demonstration neurologic improvement in reflexes, strength, movement and sensation, compared with 3% of those in the control group. The study’s findings led the FDA to approve the device using 10 kHz.
At 6 months, 93% of control patients crossed over to receiving spinal cord stimulation while none with the devices opted to stop their spinal cord stimulation. The 12-month data revealed that 85% of those receiving spinal cord stimulation experienced at least 50% pain relief, with the average pain relief at 74%. Patients also reported statistically significant improved quality of life as well as less interference with sleep, mood, and daily activities from pain.
Two years after baseline, patients’ pain relief was maintained with average 80% improvement, and 66% of patients showed neurologic improvement since baseline. Though no patients had devices removed because of ineffectiveness, five patients’ devices were removed because of infection while infections in three other patients resolved.
“Being able to offer something that is not a pharmaceutical, without the side effects, that shows an even longer durability to that response is a really important finding at this point,” Dr. Petersen said.
Surgical considerations
Among the estimated 37 million Americans with type 1 or 2 diabetes, approximately one quarter of them experience some level of painful diabetic neuropathy, but medication and other medical management strategies are not always adequate in treating their pain. After a 1-week trial of spinal cord stimulation, the devices are implanted under the skin and rechargeable through the skin for up to 10 years, after which they can be replaced.
An appropriate candidate for spinal cord stimulation would be someone for whom existing non-invasive pain relief options, including medications, are ineffective or intolerable, Dr. Petersen and Dr. Markman both said. An adequate trial of medication is not “one size fits all” and will vary by each patient, added Dr. Markman, who is also interested in whether this study’s participants were able to have a reduction in use of pain relief medications.
“I think there’s a significant number of patients out there who can benefit from this, so I think that’s why it’s promising and exciting,” Dr. Markman said. “I do think it’s important to see if this actually allows them to be on less medication or whether stimulation turns out to be another treatment in addition to their baseline treatments.” The challenge is identifying “which patients are most likely to be benefiting from this and which are most likely to be harmed.”
Aside from infection from implantation, other possible risks include pain at the battery site and, in rare cases, a need for reoperation because of migration of the leads, he said.
Improvement in symptom severity and quality of life
After the wound from the implant has completely healed, Dr. Petersen said patients using the devices do not have any activity restrictions outside of magnetic interference, such as MRIs. “I’ve had people go back-country kayaking, scuba diving, fishing with their grandkids, all sorts of all sorts of things. If patients need to go through a scanner of any kind, they should ask whether it’s safe for pacemakers since these devices are like a “pacemaker for pain.
“I had a patient bring solar chargers with him so that he could recharge his battery in the backwoods while kayaking because that’s the level of improvement in pain that he got – from barely being able to walk down the hall to feeling comfortable being off the grid and active again,” Dr. Petersen said. “Those kinds of improvements in quality of life are massive.”
The study findings may also suggest that spinal cord stimulation can benefit a broader population of patients experiencing neuropathic pain, Dr. Markman said.
“There’s an extraordinary unmet need for treatments for neuropathy, and one important question here is the extent to which diabetic peripheral neuropathy and the response that we’re seeing here is a proxy for a broader effect across many neuropathies that are caused by other conditions other than diabetes,” Dr. Markman said. “There’s a lot of reason to think that this will be helpful not just for diabetes-related neuropathic pain, but for other types of neuropathic pain that have similar clinical presentations or clinical symptom patterns to diabetic peripheral neuropathy.”
The study was funded by Nevro, who manufactures the devices. Dr. Petersen and Dr. Markman both reported consulting with, receiving support from, holding stock options with, and serving on the data safety monitoring boards and advisory boards of numerous pharmaceutical companies.
according to research that released early, prior to its presentation at the annual meeting of the American Academy of Neurology.
The data represents the longest follow-up available for spinal cord stimulation at a frequency higher than the 60 Hz initially approved for diabetic neuropathy by the Food and Drug Administration, according to lead author Erika A. Petersen, MD, a professor of neurosurgery and the residency program director at the University of Arkansas for Medical Sciences, Little Rock.
“You would expect that somebody who continues to have diabetes for 24 months and has neuropathy would have worse neuropathy after 2 years, and what we’re seeing is that people were stable or better in terms of their nerve function at 2 years,” Dr. Petersen said in an interview. “So that’s really revolutionary.”
Encouraging preliminary findings
The findings are “promising and preliminary,” John D. Markman, MD, a professor in neurology and neurosurgery, vice chair for clinical research, and director of the Translational Pain Research Program at the University of Rochester (N.Y.) Medical Center, said in an interview. Dr. Markman, who was not involved in this study, said that, though the results are encouraging, it’s “less clear how much of [the pain improvement] is due to what we would consider to be on-target, pain-relieving benefit from stimulation versus other factors like expectation.” The crossover rate and amount of reduction in pain intensity are promising, but “I think that excitement is weighed against the fact that this is an open-label study.”
An underused treatment
Although spinal cord stimulation has been around since the late 1960s, its use only picked up steam in the 2000s, when it became more frequently used to treat chronic nerve damage related to neuropathic pain syndromes, Dr. Petersen explained. The FDA approved the treatment’s new indication for diabetic neuropathy in 2015, and data from Abbott and Medtronic have shown benefits from spinal cord stimulation at 60 Hz, but some patients are uncomfortable with the vibration or tingling feelings the devices can cause at that frequency.
“They describe creepy crawlies or ants crawling over the feet, or pins and needles, and painful sensitivity,” Dr. Petersen said. “You create a vibration feeling in the same zone where they already have those feelings of buzzing and pain and vibration, and it’s sometimes actually even more uncomfortable and less satisfying to them in terms of relief” with the spinal cord stimulation at 60 Hz, she said, “so there’s a lot of attrition in terms of who will actually use it.”
At 10 kHz, however, “people don’t feel any vibration or tingling associated with it; it just jams the signal of the pain,” she said. The difference between the frequencies is like that between “a lifeguard whistle and a dog whistle.”
Testing high-frequency stimulation
The new findings included the 24-month follow-up data from a randomized controlled trial that assessed the effectiveness of high-frequency spinal cord stimulation for painful diabetic neuropathy. The original 216 participants enrolled in the trial had diabetic neuropathy symptoms for at least 12 months and either could no not tolerate or did not respond to medications. Enrollment criteria also included lower-limb pain intensity of at least 5 on a 0-10 visual analogy scale and hemoglobin A1c of no more than 10%.
For the first 6 months of the trial – before crossover was offered – participants were randomly assigned to receive either 10 kHz of spinal cord stimulation along with conventional medical management or to receive conventional medical management alone. The 6-month data from 187 patients, as reported in April 2021 in JAMA Neurology, revealed that 79% of those receiving spinal cord stimulation experienced at least 50% improved pain relief without worsening of their baseline neurologic deficits, compared with only 5% of those receiving only conventional treatments.
Average pain levels increased 2% in the control participants compared with a decrease of 76% in those with the spinal cord stimulation devices. In addition, 62% of the patients receiving spinal cord stimulation demonstration neurologic improvement in reflexes, strength, movement and sensation, compared with 3% of those in the control group. The study’s findings led the FDA to approve the device using 10 kHz.
At 6 months, 93% of control patients crossed over to receiving spinal cord stimulation while none with the devices opted to stop their spinal cord stimulation. The 12-month data revealed that 85% of those receiving spinal cord stimulation experienced at least 50% pain relief, with the average pain relief at 74%. Patients also reported statistically significant improved quality of life as well as less interference with sleep, mood, and daily activities from pain.
Two years after baseline, patients’ pain relief was maintained with average 80% improvement, and 66% of patients showed neurologic improvement since baseline. Though no patients had devices removed because of ineffectiveness, five patients’ devices were removed because of infection while infections in three other patients resolved.
“Being able to offer something that is not a pharmaceutical, without the side effects, that shows an even longer durability to that response is a really important finding at this point,” Dr. Petersen said.
Surgical considerations
Among the estimated 37 million Americans with type 1 or 2 diabetes, approximately one quarter of them experience some level of painful diabetic neuropathy, but medication and other medical management strategies are not always adequate in treating their pain. After a 1-week trial of spinal cord stimulation, the devices are implanted under the skin and rechargeable through the skin for up to 10 years, after which they can be replaced.
An appropriate candidate for spinal cord stimulation would be someone for whom existing non-invasive pain relief options, including medications, are ineffective or intolerable, Dr. Petersen and Dr. Markman both said. An adequate trial of medication is not “one size fits all” and will vary by each patient, added Dr. Markman, who is also interested in whether this study’s participants were able to have a reduction in use of pain relief medications.
“I think there’s a significant number of patients out there who can benefit from this, so I think that’s why it’s promising and exciting,” Dr. Markman said. “I do think it’s important to see if this actually allows them to be on less medication or whether stimulation turns out to be another treatment in addition to their baseline treatments.” The challenge is identifying “which patients are most likely to be benefiting from this and which are most likely to be harmed.”
Aside from infection from implantation, other possible risks include pain at the battery site and, in rare cases, a need for reoperation because of migration of the leads, he said.
Improvement in symptom severity and quality of life
After the wound from the implant has completely healed, Dr. Petersen said patients using the devices do not have any activity restrictions outside of magnetic interference, such as MRIs. “I’ve had people go back-country kayaking, scuba diving, fishing with their grandkids, all sorts of all sorts of things. If patients need to go through a scanner of any kind, they should ask whether it’s safe for pacemakers since these devices are like a “pacemaker for pain.
“I had a patient bring solar chargers with him so that he could recharge his battery in the backwoods while kayaking because that’s the level of improvement in pain that he got – from barely being able to walk down the hall to feeling comfortable being off the grid and active again,” Dr. Petersen said. “Those kinds of improvements in quality of life are massive.”
The study findings may also suggest that spinal cord stimulation can benefit a broader population of patients experiencing neuropathic pain, Dr. Markman said.
“There’s an extraordinary unmet need for treatments for neuropathy, and one important question here is the extent to which diabetic peripheral neuropathy and the response that we’re seeing here is a proxy for a broader effect across many neuropathies that are caused by other conditions other than diabetes,” Dr. Markman said. “There’s a lot of reason to think that this will be helpful not just for diabetes-related neuropathic pain, but for other types of neuropathic pain that have similar clinical presentations or clinical symptom patterns to diabetic peripheral neuropathy.”
The study was funded by Nevro, who manufactures the devices. Dr. Petersen and Dr. Markman both reported consulting with, receiving support from, holding stock options with, and serving on the data safety monitoring boards and advisory boards of numerous pharmaceutical companies.
FROM AAN 2023
Battlefield Acupuncture vs Ketorolac for Treating Pain in the Emergency Department
Acute pain is a primary symptom for many patients who present to the emergency department (ED). The ED team is challenged with relieving pain while limiting harm from medications.1 A 2017 National Health Interview Survey showed that compared with nonveterans, more veterans reported pain in the previous 3 months, and the rate of severe pain was 40% higher in the veteran group especially among those who served during the era of wars in Afghanistan and Iraq.2
The American College of Emergency Physicians guidelines pain management guidelines recommend patient-centered shared decision making that includes patient education about treatment goals and expectations, and short- and long-term risks, as well as a preference toward pharmacologic treatment with nonopioid analgesics except for patients with severe pain or pain refractory to other drug and treatment modalities.3 There is a lack of evidence regarding superior efficacy of either opioid or nonopioid analgesics; therefore, the use of nonopioid analgesics, such as oral or topical nonsteroidal anti-inflammatory drugs (NSAIDs) or central analgesics, such as acetaminophen, is preferred for treating acute pain to mitigate adverse effects (AEs) and risks associated with opioid use.1,3,4 The US Department of Veterans Affairs (VA) and Department of Defense (DoD) guideline on managing opioid therapy for chronic pain, updated in 2017 and 2022, similarly recommends alternatives to opioids for mild-to-moderate acute pain and encourages multimodal pain care.5 However, use of other pharmacologic treatments, such as NSAIDs, is limited by AE profiles, patient contraindications, and severity of acute pain etiologies. There is a need for the expanded use of nonpharmacologic treatments for addressing pain in the veteran population.
The American College of Emergency Physicians guidelines recommend nonpharmacologic modalities, such as applying heat or cold, physical therapy, cognitive behavioral therapy, and acupuncture.3 A 2014 study reported that 37% to 46% of active duty and reserve military personnel use complementary and alternative medicine (CAM) for a variety of ailments, and there is increasing interest in the use of CAM as adjuncts to traditional therapies.6 According to one study, some CAM therapies are used significantly more by military personnel than used by civilians.7 However, the percentage of the veteran population using acupuncture in this study was small, and more information is needed to assess its use.
Auricular acupuncture originated in traditional Chinese medicine.8 Contemporary auricular acupuncture experts view this modality as a self-contained microsystem mapping portions of the ear to specific parts of the body and internal organs. The analgesic effects may be mediated through the central nervous system by local release of endorphins through nerve fiber activation and neurotransmitters—including serotonin, dopamine, and norepinephrine—leading to pre- and postsynaptic suppression of pain transmission.
Battlefield acupuncture (BFA) uses 5 set points anatomically located on each ear.9 Practitioners use small semipermanent, dartlike acupuncture needles. Patients could experience pain relief in a few minutes, which can last minutes, hours, days, weeks, or months depending on the pathology of the pain. This procedure developed in 2001 has been studied for different pain types and has shown benefit when used for postsurgical pain, chronic spinal cord injury−related neuropathic pain, and general chronic pain, as well as for other indications, such as insomnia, depression, and weight loss.8,10-13 In 2018, a randomized controlled trial compared postintervention numeric rating scale (NRS) pain scores in patients presenting to the ED with acute or acute-on-chronic lower back pain who received BFA as an adjunct to standard care vs standard care alone.14 Patients receiving BFA as an adjunct to standard care were found to have mean postintervention pain scores 1.7 points lower than those receiving standard care alone. This study demonstrated that BFA was feasible and well tolerated for lower back pain in the ED as an adjunct to standard care. The study was limited by the adjunct use of BFA rather than as monotherapy and by the practitioners’ discretion regarding standard care, which was not defined by the study’s authors.
The Jesse Brown Veterans Affairs Medical Center (JBVAMC) in Chicago, Illinois, offers several CAM modalities, such as exercise/movement therapy, chiropractic, art/music therapy, and relaxation workshops, which are widely used by veterans. Recent evidence suggests BFA could reduce pain scores as an adjunct or an alternative to pharmacologic therapy. We are interested in how CAM therapies, such as BFA, can help avoid AEs associated with opioid or NSAID therapy.
At the JBVAMC ED, ketorolac 15 mg is the preferred first-line treatment of acute, noncancer pain, based on the results of previous studies. In 2018 BFA was offered first to veterans presenting with acute or acute-on-chronic pain to the ED; however, its effectiveness for pain reduction vs ketorolac has not been evaluated in this patient population. Limited literature is available on BFA and its use in the ED. To our knowledge, this was the first observational study assessing the difference between a single session of BFA vs a single dose of ketorolac in treating noncancer acute or acute-on-chronic pain in the ED.
Methods
This study was a retrospective chart review of patients who presented to the JBVAMC ED with acute pain or acute-on-chronic pain, who received ketorolac or BFA. The study population was generated from a list of all IV and intramuscular (IM) ketorolac unit dose orders verified from June 1, 2018, through August 30, 2019, and a list of all BFA procedure notes signed from June 1, 2018, through August 30, 2019. Patients were included in the study if they had documented administration of IV or IM ketorolac or BFA between June 1, 2018, and August 30, 2019. Patients who received ketorolac doses other than 15 mg, the intervention was administered outside of the ED, received adjunct treatment in addition to the treatment intervention in the ED, had no baseline NRS pain score documented before the intervention, had an NRS pain score of < 4, had no postintervention NRS pain score documented within 6 hours, had a treatment indication other than pain, or had active cancer were excluded. As in previous JBVAMC studies, we used NRS pain score cutoffs (mild, moderate, severe, and very severe) based on Woo and colleagues’ meta-analysis and excluded scores < 4.15
Endpoints
The primary endpoint was the mean difference in NRS pain score before and after the intervention, determined by comparing the NRS pain score documented at triage to the ED with the first documented NRS pain score at least 30 minutes to 6 hours after treatment administration. The secondary endpoints included the number of patients prescribed pain medication at discharge, the number of patients who were discharged with no medications, and the number of patients admitted to the hospital. The safety endpoint included any AEs of the intervention. Subgroup analyses were performed comparing the mean difference in NRS pain score among subgroups classified by severity of baseline NRS pain score and pain location.
Statistical Analysis
Baseline characteristics and endpoints were analyzed using descriptive statistics. Categorical data were analyzed using Fisher exact test and z test for proportions, and continuous data were compared using t test and paired t test. An 80% power calculation determined that 84 patients per group were needed to detect a statistically significant difference in pain score reduction of 1.3 at a type-1 error rate of 0.05. The sample size was based on a calculation performed in a previously published study that compared IV ketorolac at 3 single-dose regimens for treating acute pain in the ED.16 The 1.3 pain score reduction is considered the minimum clinically significant difference in pain that could be detected with the NRS.17
Results
Sixty-one patients received BFA during the study period: 31 were excluded (26 received adjunct treatment in the ED, 2 had active cancer documented, 2 had an indication other than pain, and 1 received BFA outside of the ED), leaving 30 patients in the BFA cohort. During the study period, 1299 patients received ketorolac.
Baseline characteristics were similar between the 2 groups except for the average baseline NRS pain score, which was statistically significantly higher in the BFA vs ketorolac group (8.7 vs 7.7, respectively; P = .02). The mean age was 51 years in the BFA group and 48 years in the ketorolac group. Most patients in each cohort were male: 80% in the BFA group and 71% in the ketorolac group. The most common types of pain documented as the chief ED presentation included back, lower extremity, and head.
Endpoints
The mean difference in NRS pain score was 3.9 for the BFA group and 5.1 for the ketorolac group. Both were clinically and statistically significant reductions (P = .03 and P < .01), but the difference between the intervention groups in NRS score reduction was not statistically significant (P = .07).
For the secondary endpoint of outpatient prescriptions written at discharge, there was no significant difference between the groups except for oral NSAIDs, which were more likely to be prescribed to patients who received ketorolac (P = .01).
Subgroup Analysis
An analysis was performed for subgroups classified by baseline NRS pain score (mild: 4; moderate, 5 - 6; severe, 7 - 9; and very severe, 10). Data for mild pain was limited because a small number of patients received interventions. For moderate pain, the mean difference in NRS pain score for BFA and ketorolac was 3.5 and 3.8, respectively; for severe pain, 3.4 and 5.3; and for very severe pain, 4.6 and 6.4. There was a larger difference in the preintervention and postintervention NRS pain scores within severe pain and very severe pain groups.
Discussion
Both interventions resulted in a significant reduction in the mean NRS pain score of about 4 to 5 points within their group, and BFA resulted in a similar NRS pain score reduction compared with ketorolac 15 mg. Because the baseline NRS pain scores were significantly different between the BFA and ketorolac groups,
In this study, more patients in the BFA group presented to the ED with lower extremity pain, such as gout or neuropathy, compared with the ketorolac group; however, BFA did not result in a significantly different pain score reduction in this subgroup compared with ketorolac. Patients receiving BFA were more likely to receive topical analgesics or muscle relaxants at discharge; whereas those receiving ketorolac were significantly more likely to receive oral NSAIDs. Patients in this study also were more likely to be admitted to the hospital if they received ketorolac; however, for these patients, pain was secondary to their chief presentation, and the admitting physician’s familiarity with ketorolac might have been the reason for choosing this intervention. Reasons for the admissions were surgical observation, psychiatric stabilization, kidney/gallstones, rule out of acute coronary syndrome, pneumonia, and proctitis in the ketorolac group, and suicidal ideations in the BFA group.
Limitations
As a limited number of patients received BFA at JBVAMC, the study was not sufficiently powered to detect a difference in the primary outcome. Because BFA required a consultation to be entered in the electronic health record, in addition to time needed to perform the procedure, practitioners might have preferred IV/IM ketorolac during busy times in the ED, potentially leading to underrepresentation in the BFA group. Prescribing preferences might have differed among the rotating physicians, timing of the documentation of the NRS pain score could have differed based on the treatment intervention, and the investigators were unable to control or accurately assess whether patients had taken an analgesic medication before presenting to the ED.
Conclusions
NRS pain score reduction with BFA did not differ compared with ketorolac 15 mg for treating acute and acute-on-chronic pain in the ED. Although this study was underpowered, these results add to the limited existing literature, suggesting that both interventions could result in clinically significant pain score reductions for patients presenting to the ED with severe and very severe pain, making BFA a viable nonpharmacologic option. Future studies could include investigating the benefit of BFA in the veteran population by studying larger samples in the ED, surveying patients after their interventions to identify rates AEs, and exploring the use of BFA for chronic pain in the outpatient setting.
1. Cantrill SV, Brown MD, Carlisle RJ, et al. Clinical policy: critical issues in the prescribing of opioids for adult patients in the emergency department. Ann Emerg Med. 2012;60(4):499-525. doi:10.1016/j.annemergmed.2012.06.013
2. Nahin RL. Severe pain in veterans: the effect of age and sex, and comparisons with the general population. J Pain. 2017;18(3):247-254. doi:10.1016/j.jpain.2016.10.021
3. Motov S, Strayer R, Hayes BD, et al. The treatment of acute pain in the emergency department: a white paper position statement prepared for the American Academy of Emergency Medicine. J Emerg Med. 2018;54(5):731-736. doi:10.1016/j.jemermed.2018.01.020
4. Samcam I, Papa L. Acute pain management in the emergency department. In: Prostran M, ed. Pain Management. IntechOpen; 2016. doi:10.5772/62861
5. Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline for the use of opioids in the management of chronic pain. Accessed February 15, 2023. https://www.healthquality.va.gov/guidelines/Pain/cot/VADoDOpioidsCPG.pdf
6. Davis MT, Mulvaney-Day N, Larson MJ, Hoover R, Mauch D. Complementary and alternative medicine among veterans and military personnel: a synthesis of population surveys. Med Care. 2014;52(12 suppl 5):S83-590. doi:10.1097/MLR.0000000000000227
7. Goertz C, Marriott BP, Finch FD, et al. Military report more complementary and alternative medicine use than civilians. J Altern Complement Med. 2013;19(6):509-517. doi:10.1089/acm.2012.0108
8. King HC, Hickey AH, Connelly C. Auricular acupuncture: a brief introduction for military providers. Mil Med. 2013;178(8):867-874. doi:10.7205/MILMED-D-13-00075
9. Niemtzow RC. Battlefield acupuncture. Medical Acupunct. 2007;19(4):225-228. doi:10.1089/acu.2007.0603
10. Collinsworth KM, Goss DL. Battlefield acupuncture and physical therapy versus physical therapy alone after shoulder surgery. Med Acupunct. 2019;31(4):228-238. doi:10.1089/acu.2019.1372
11. Estores I, Chen K, Jackson B, Lao L, Gorman PH. Auricular acupuncture for spinal cord injury related neuropathic pain: a pilot controlled clinical trial. J Spinal Cord Med. 2017;40(4):432-438. doi:10.1080/10790268.2016.1141489
12. Federman DG, Radhakrishnan K, Gabriel L, Poulin LM, Kravetz JD. Group battlefield acupuncture in primary care for veterans with pain. South Med J. 2018;111(10):619-624. doi:10.14423/SMJ.0000000000000877
13. Garner BK, Hopkinson SG, Ketz AK, Landis CA, Trego LL. Auricular acupuncture for chronic pain and insomnia: a randomized clinical trial. Med Acupunct. 2018;30(5):262-272. doi:10.1089/acu.2018.1294
14. Fox LM, Murakami M, Danesh H, Manini AF. Battlefield acupuncture to treat low back pain in the emergency department. Am J Emerg Med. 2018; 36:1045-1048. doi:10.1016/j.ajem.2018.02.038
15. Woo A, Lechner B, Fu T, et al. Cut points for mild, moderate, and severe pain among cancer and non-cancer patients: a literature review. Ann Palliat Med. 2015;4(4):176-183. doi:10.3978/j.issn.2224-5820.2015.09.04
16. Motov S, Yasavolian M, Likourezos A, et al. Comparison of intravenous ketorolac at three single-dose regimens for treating acute pain in the emergency department: a randomized controlled trial. Ann Emerg Med. 2017;70(2):177-184. doi:10.1016/j.annemergmed.2016.10.014
17. Bijur PE, Latimer CT, Gallagher EJ. Validation of a verbally administered numerical rating scale of acute pain for use in the emergency department. Acad Emerg Med. 2003;10:390-392. doi:10.1111/j.1553-2712.2003.tb01355.
Acute pain is a primary symptom for many patients who present to the emergency department (ED). The ED team is challenged with relieving pain while limiting harm from medications.1 A 2017 National Health Interview Survey showed that compared with nonveterans, more veterans reported pain in the previous 3 months, and the rate of severe pain was 40% higher in the veteran group especially among those who served during the era of wars in Afghanistan and Iraq.2
The American College of Emergency Physicians guidelines pain management guidelines recommend patient-centered shared decision making that includes patient education about treatment goals and expectations, and short- and long-term risks, as well as a preference toward pharmacologic treatment with nonopioid analgesics except for patients with severe pain or pain refractory to other drug and treatment modalities.3 There is a lack of evidence regarding superior efficacy of either opioid or nonopioid analgesics; therefore, the use of nonopioid analgesics, such as oral or topical nonsteroidal anti-inflammatory drugs (NSAIDs) or central analgesics, such as acetaminophen, is preferred for treating acute pain to mitigate adverse effects (AEs) and risks associated with opioid use.1,3,4 The US Department of Veterans Affairs (VA) and Department of Defense (DoD) guideline on managing opioid therapy for chronic pain, updated in 2017 and 2022, similarly recommends alternatives to opioids for mild-to-moderate acute pain and encourages multimodal pain care.5 However, use of other pharmacologic treatments, such as NSAIDs, is limited by AE profiles, patient contraindications, and severity of acute pain etiologies. There is a need for the expanded use of nonpharmacologic treatments for addressing pain in the veteran population.
The American College of Emergency Physicians guidelines recommend nonpharmacologic modalities, such as applying heat or cold, physical therapy, cognitive behavioral therapy, and acupuncture.3 A 2014 study reported that 37% to 46% of active duty and reserve military personnel use complementary and alternative medicine (CAM) for a variety of ailments, and there is increasing interest in the use of CAM as adjuncts to traditional therapies.6 According to one study, some CAM therapies are used significantly more by military personnel than used by civilians.7 However, the percentage of the veteran population using acupuncture in this study was small, and more information is needed to assess its use.
Auricular acupuncture originated in traditional Chinese medicine.8 Contemporary auricular acupuncture experts view this modality as a self-contained microsystem mapping portions of the ear to specific parts of the body and internal organs. The analgesic effects may be mediated through the central nervous system by local release of endorphins through nerve fiber activation and neurotransmitters—including serotonin, dopamine, and norepinephrine—leading to pre- and postsynaptic suppression of pain transmission.
Battlefield acupuncture (BFA) uses 5 set points anatomically located on each ear.9 Practitioners use small semipermanent, dartlike acupuncture needles. Patients could experience pain relief in a few minutes, which can last minutes, hours, days, weeks, or months depending on the pathology of the pain. This procedure developed in 2001 has been studied for different pain types and has shown benefit when used for postsurgical pain, chronic spinal cord injury−related neuropathic pain, and general chronic pain, as well as for other indications, such as insomnia, depression, and weight loss.8,10-13 In 2018, a randomized controlled trial compared postintervention numeric rating scale (NRS) pain scores in patients presenting to the ED with acute or acute-on-chronic lower back pain who received BFA as an adjunct to standard care vs standard care alone.14 Patients receiving BFA as an adjunct to standard care were found to have mean postintervention pain scores 1.7 points lower than those receiving standard care alone. This study demonstrated that BFA was feasible and well tolerated for lower back pain in the ED as an adjunct to standard care. The study was limited by the adjunct use of BFA rather than as monotherapy and by the practitioners’ discretion regarding standard care, which was not defined by the study’s authors.
The Jesse Brown Veterans Affairs Medical Center (JBVAMC) in Chicago, Illinois, offers several CAM modalities, such as exercise/movement therapy, chiropractic, art/music therapy, and relaxation workshops, which are widely used by veterans. Recent evidence suggests BFA could reduce pain scores as an adjunct or an alternative to pharmacologic therapy. We are interested in how CAM therapies, such as BFA, can help avoid AEs associated with opioid or NSAID therapy.
At the JBVAMC ED, ketorolac 15 mg is the preferred first-line treatment of acute, noncancer pain, based on the results of previous studies. In 2018 BFA was offered first to veterans presenting with acute or acute-on-chronic pain to the ED; however, its effectiveness for pain reduction vs ketorolac has not been evaluated in this patient population. Limited literature is available on BFA and its use in the ED. To our knowledge, this was the first observational study assessing the difference between a single session of BFA vs a single dose of ketorolac in treating noncancer acute or acute-on-chronic pain in the ED.
Methods
This study was a retrospective chart review of patients who presented to the JBVAMC ED with acute pain or acute-on-chronic pain, who received ketorolac or BFA. The study population was generated from a list of all IV and intramuscular (IM) ketorolac unit dose orders verified from June 1, 2018, through August 30, 2019, and a list of all BFA procedure notes signed from June 1, 2018, through August 30, 2019. Patients were included in the study if they had documented administration of IV or IM ketorolac or BFA between June 1, 2018, and August 30, 2019. Patients who received ketorolac doses other than 15 mg, the intervention was administered outside of the ED, received adjunct treatment in addition to the treatment intervention in the ED, had no baseline NRS pain score documented before the intervention, had an NRS pain score of < 4, had no postintervention NRS pain score documented within 6 hours, had a treatment indication other than pain, or had active cancer were excluded. As in previous JBVAMC studies, we used NRS pain score cutoffs (mild, moderate, severe, and very severe) based on Woo and colleagues’ meta-analysis and excluded scores < 4.15
Endpoints
The primary endpoint was the mean difference in NRS pain score before and after the intervention, determined by comparing the NRS pain score documented at triage to the ED with the first documented NRS pain score at least 30 minutes to 6 hours after treatment administration. The secondary endpoints included the number of patients prescribed pain medication at discharge, the number of patients who were discharged with no medications, and the number of patients admitted to the hospital. The safety endpoint included any AEs of the intervention. Subgroup analyses were performed comparing the mean difference in NRS pain score among subgroups classified by severity of baseline NRS pain score and pain location.
Statistical Analysis
Baseline characteristics and endpoints were analyzed using descriptive statistics. Categorical data were analyzed using Fisher exact test and z test for proportions, and continuous data were compared using t test and paired t test. An 80% power calculation determined that 84 patients per group were needed to detect a statistically significant difference in pain score reduction of 1.3 at a type-1 error rate of 0.05. The sample size was based on a calculation performed in a previously published study that compared IV ketorolac at 3 single-dose regimens for treating acute pain in the ED.16 The 1.3 pain score reduction is considered the minimum clinically significant difference in pain that could be detected with the NRS.17
Results
Sixty-one patients received BFA during the study period: 31 were excluded (26 received adjunct treatment in the ED, 2 had active cancer documented, 2 had an indication other than pain, and 1 received BFA outside of the ED), leaving 30 patients in the BFA cohort. During the study period, 1299 patients received ketorolac.
Baseline characteristics were similar between the 2 groups except for the average baseline NRS pain score, which was statistically significantly higher in the BFA vs ketorolac group (8.7 vs 7.7, respectively; P = .02). The mean age was 51 years in the BFA group and 48 years in the ketorolac group. Most patients in each cohort were male: 80% in the BFA group and 71% in the ketorolac group. The most common types of pain documented as the chief ED presentation included back, lower extremity, and head.
Endpoints
The mean difference in NRS pain score was 3.9 for the BFA group and 5.1 for the ketorolac group. Both were clinically and statistically significant reductions (P = .03 and P < .01), but the difference between the intervention groups in NRS score reduction was not statistically significant (P = .07).
For the secondary endpoint of outpatient prescriptions written at discharge, there was no significant difference between the groups except for oral NSAIDs, which were more likely to be prescribed to patients who received ketorolac (P = .01).
Subgroup Analysis
An analysis was performed for subgroups classified by baseline NRS pain score (mild: 4; moderate, 5 - 6; severe, 7 - 9; and very severe, 10). Data for mild pain was limited because a small number of patients received interventions. For moderate pain, the mean difference in NRS pain score for BFA and ketorolac was 3.5 and 3.8, respectively; for severe pain, 3.4 and 5.3; and for very severe pain, 4.6 and 6.4. There was a larger difference in the preintervention and postintervention NRS pain scores within severe pain and very severe pain groups.
Discussion
Both interventions resulted in a significant reduction in the mean NRS pain score of about 4 to 5 points within their group, and BFA resulted in a similar NRS pain score reduction compared with ketorolac 15 mg. Because the baseline NRS pain scores were significantly different between the BFA and ketorolac groups,
In this study, more patients in the BFA group presented to the ED with lower extremity pain, such as gout or neuropathy, compared with the ketorolac group; however, BFA did not result in a significantly different pain score reduction in this subgroup compared with ketorolac. Patients receiving BFA were more likely to receive topical analgesics or muscle relaxants at discharge; whereas those receiving ketorolac were significantly more likely to receive oral NSAIDs. Patients in this study also were more likely to be admitted to the hospital if they received ketorolac; however, for these patients, pain was secondary to their chief presentation, and the admitting physician’s familiarity with ketorolac might have been the reason for choosing this intervention. Reasons for the admissions were surgical observation, psychiatric stabilization, kidney/gallstones, rule out of acute coronary syndrome, pneumonia, and proctitis in the ketorolac group, and suicidal ideations in the BFA group.
Limitations
As a limited number of patients received BFA at JBVAMC, the study was not sufficiently powered to detect a difference in the primary outcome. Because BFA required a consultation to be entered in the electronic health record, in addition to time needed to perform the procedure, practitioners might have preferred IV/IM ketorolac during busy times in the ED, potentially leading to underrepresentation in the BFA group. Prescribing preferences might have differed among the rotating physicians, timing of the documentation of the NRS pain score could have differed based on the treatment intervention, and the investigators were unable to control or accurately assess whether patients had taken an analgesic medication before presenting to the ED.
Conclusions
NRS pain score reduction with BFA did not differ compared with ketorolac 15 mg for treating acute and acute-on-chronic pain in the ED. Although this study was underpowered, these results add to the limited existing literature, suggesting that both interventions could result in clinically significant pain score reductions for patients presenting to the ED with severe and very severe pain, making BFA a viable nonpharmacologic option. Future studies could include investigating the benefit of BFA in the veteran population by studying larger samples in the ED, surveying patients after their interventions to identify rates AEs, and exploring the use of BFA for chronic pain in the outpatient setting.
Acute pain is a primary symptom for many patients who present to the emergency department (ED). The ED team is challenged with relieving pain while limiting harm from medications.1 A 2017 National Health Interview Survey showed that compared with nonveterans, more veterans reported pain in the previous 3 months, and the rate of severe pain was 40% higher in the veteran group especially among those who served during the era of wars in Afghanistan and Iraq.2
The American College of Emergency Physicians guidelines pain management guidelines recommend patient-centered shared decision making that includes patient education about treatment goals and expectations, and short- and long-term risks, as well as a preference toward pharmacologic treatment with nonopioid analgesics except for patients with severe pain or pain refractory to other drug and treatment modalities.3 There is a lack of evidence regarding superior efficacy of either opioid or nonopioid analgesics; therefore, the use of nonopioid analgesics, such as oral or topical nonsteroidal anti-inflammatory drugs (NSAIDs) or central analgesics, such as acetaminophen, is preferred for treating acute pain to mitigate adverse effects (AEs) and risks associated with opioid use.1,3,4 The US Department of Veterans Affairs (VA) and Department of Defense (DoD) guideline on managing opioid therapy for chronic pain, updated in 2017 and 2022, similarly recommends alternatives to opioids for mild-to-moderate acute pain and encourages multimodal pain care.5 However, use of other pharmacologic treatments, such as NSAIDs, is limited by AE profiles, patient contraindications, and severity of acute pain etiologies. There is a need for the expanded use of nonpharmacologic treatments for addressing pain in the veteran population.
The American College of Emergency Physicians guidelines recommend nonpharmacologic modalities, such as applying heat or cold, physical therapy, cognitive behavioral therapy, and acupuncture.3 A 2014 study reported that 37% to 46% of active duty and reserve military personnel use complementary and alternative medicine (CAM) for a variety of ailments, and there is increasing interest in the use of CAM as adjuncts to traditional therapies.6 According to one study, some CAM therapies are used significantly more by military personnel than used by civilians.7 However, the percentage of the veteran population using acupuncture in this study was small, and more information is needed to assess its use.
Auricular acupuncture originated in traditional Chinese medicine.8 Contemporary auricular acupuncture experts view this modality as a self-contained microsystem mapping portions of the ear to specific parts of the body and internal organs. The analgesic effects may be mediated through the central nervous system by local release of endorphins through nerve fiber activation and neurotransmitters—including serotonin, dopamine, and norepinephrine—leading to pre- and postsynaptic suppression of pain transmission.
Battlefield acupuncture (BFA) uses 5 set points anatomically located on each ear.9 Practitioners use small semipermanent, dartlike acupuncture needles. Patients could experience pain relief in a few minutes, which can last minutes, hours, days, weeks, or months depending on the pathology of the pain. This procedure developed in 2001 has been studied for different pain types and has shown benefit when used for postsurgical pain, chronic spinal cord injury−related neuropathic pain, and general chronic pain, as well as for other indications, such as insomnia, depression, and weight loss.8,10-13 In 2018, a randomized controlled trial compared postintervention numeric rating scale (NRS) pain scores in patients presenting to the ED with acute or acute-on-chronic lower back pain who received BFA as an adjunct to standard care vs standard care alone.14 Patients receiving BFA as an adjunct to standard care were found to have mean postintervention pain scores 1.7 points lower than those receiving standard care alone. This study demonstrated that BFA was feasible and well tolerated for lower back pain in the ED as an adjunct to standard care. The study was limited by the adjunct use of BFA rather than as monotherapy and by the practitioners’ discretion regarding standard care, which was not defined by the study’s authors.
The Jesse Brown Veterans Affairs Medical Center (JBVAMC) in Chicago, Illinois, offers several CAM modalities, such as exercise/movement therapy, chiropractic, art/music therapy, and relaxation workshops, which are widely used by veterans. Recent evidence suggests BFA could reduce pain scores as an adjunct or an alternative to pharmacologic therapy. We are interested in how CAM therapies, such as BFA, can help avoid AEs associated with opioid or NSAID therapy.
At the JBVAMC ED, ketorolac 15 mg is the preferred first-line treatment of acute, noncancer pain, based on the results of previous studies. In 2018 BFA was offered first to veterans presenting with acute or acute-on-chronic pain to the ED; however, its effectiveness for pain reduction vs ketorolac has not been evaluated in this patient population. Limited literature is available on BFA and its use in the ED. To our knowledge, this was the first observational study assessing the difference between a single session of BFA vs a single dose of ketorolac in treating noncancer acute or acute-on-chronic pain in the ED.
Methods
This study was a retrospective chart review of patients who presented to the JBVAMC ED with acute pain or acute-on-chronic pain, who received ketorolac or BFA. The study population was generated from a list of all IV and intramuscular (IM) ketorolac unit dose orders verified from June 1, 2018, through August 30, 2019, and a list of all BFA procedure notes signed from June 1, 2018, through August 30, 2019. Patients were included in the study if they had documented administration of IV or IM ketorolac or BFA between June 1, 2018, and August 30, 2019. Patients who received ketorolac doses other than 15 mg, the intervention was administered outside of the ED, received adjunct treatment in addition to the treatment intervention in the ED, had no baseline NRS pain score documented before the intervention, had an NRS pain score of < 4, had no postintervention NRS pain score documented within 6 hours, had a treatment indication other than pain, or had active cancer were excluded. As in previous JBVAMC studies, we used NRS pain score cutoffs (mild, moderate, severe, and very severe) based on Woo and colleagues’ meta-analysis and excluded scores < 4.15
Endpoints
The primary endpoint was the mean difference in NRS pain score before and after the intervention, determined by comparing the NRS pain score documented at triage to the ED with the first documented NRS pain score at least 30 minutes to 6 hours after treatment administration. The secondary endpoints included the number of patients prescribed pain medication at discharge, the number of patients who were discharged with no medications, and the number of patients admitted to the hospital. The safety endpoint included any AEs of the intervention. Subgroup analyses were performed comparing the mean difference in NRS pain score among subgroups classified by severity of baseline NRS pain score and pain location.
Statistical Analysis
Baseline characteristics and endpoints were analyzed using descriptive statistics. Categorical data were analyzed using Fisher exact test and z test for proportions, and continuous data were compared using t test and paired t test. An 80% power calculation determined that 84 patients per group were needed to detect a statistically significant difference in pain score reduction of 1.3 at a type-1 error rate of 0.05. The sample size was based on a calculation performed in a previously published study that compared IV ketorolac at 3 single-dose regimens for treating acute pain in the ED.16 The 1.3 pain score reduction is considered the minimum clinically significant difference in pain that could be detected with the NRS.17
Results
Sixty-one patients received BFA during the study period: 31 were excluded (26 received adjunct treatment in the ED, 2 had active cancer documented, 2 had an indication other than pain, and 1 received BFA outside of the ED), leaving 30 patients in the BFA cohort. During the study period, 1299 patients received ketorolac.
Baseline characteristics were similar between the 2 groups except for the average baseline NRS pain score, which was statistically significantly higher in the BFA vs ketorolac group (8.7 vs 7.7, respectively; P = .02). The mean age was 51 years in the BFA group and 48 years in the ketorolac group. Most patients in each cohort were male: 80% in the BFA group and 71% in the ketorolac group. The most common types of pain documented as the chief ED presentation included back, lower extremity, and head.
Endpoints
The mean difference in NRS pain score was 3.9 for the BFA group and 5.1 for the ketorolac group. Both were clinically and statistically significant reductions (P = .03 and P < .01), but the difference between the intervention groups in NRS score reduction was not statistically significant (P = .07).
For the secondary endpoint of outpatient prescriptions written at discharge, there was no significant difference between the groups except for oral NSAIDs, which were more likely to be prescribed to patients who received ketorolac (P = .01).
Subgroup Analysis
An analysis was performed for subgroups classified by baseline NRS pain score (mild: 4; moderate, 5 - 6; severe, 7 - 9; and very severe, 10). Data for mild pain was limited because a small number of patients received interventions. For moderate pain, the mean difference in NRS pain score for BFA and ketorolac was 3.5 and 3.8, respectively; for severe pain, 3.4 and 5.3; and for very severe pain, 4.6 and 6.4. There was a larger difference in the preintervention and postintervention NRS pain scores within severe pain and very severe pain groups.
Discussion
Both interventions resulted in a significant reduction in the mean NRS pain score of about 4 to 5 points within their group, and BFA resulted in a similar NRS pain score reduction compared with ketorolac 15 mg. Because the baseline NRS pain scores were significantly different between the BFA and ketorolac groups,
In this study, more patients in the BFA group presented to the ED with lower extremity pain, such as gout or neuropathy, compared with the ketorolac group; however, BFA did not result in a significantly different pain score reduction in this subgroup compared with ketorolac. Patients receiving BFA were more likely to receive topical analgesics or muscle relaxants at discharge; whereas those receiving ketorolac were significantly more likely to receive oral NSAIDs. Patients in this study also were more likely to be admitted to the hospital if they received ketorolac; however, for these patients, pain was secondary to their chief presentation, and the admitting physician’s familiarity with ketorolac might have been the reason for choosing this intervention. Reasons for the admissions were surgical observation, psychiatric stabilization, kidney/gallstones, rule out of acute coronary syndrome, pneumonia, and proctitis in the ketorolac group, and suicidal ideations in the BFA group.
Limitations
As a limited number of patients received BFA at JBVAMC, the study was not sufficiently powered to detect a difference in the primary outcome. Because BFA required a consultation to be entered in the electronic health record, in addition to time needed to perform the procedure, practitioners might have preferred IV/IM ketorolac during busy times in the ED, potentially leading to underrepresentation in the BFA group. Prescribing preferences might have differed among the rotating physicians, timing of the documentation of the NRS pain score could have differed based on the treatment intervention, and the investigators were unable to control or accurately assess whether patients had taken an analgesic medication before presenting to the ED.
Conclusions
NRS pain score reduction with BFA did not differ compared with ketorolac 15 mg for treating acute and acute-on-chronic pain in the ED. Although this study was underpowered, these results add to the limited existing literature, suggesting that both interventions could result in clinically significant pain score reductions for patients presenting to the ED with severe and very severe pain, making BFA a viable nonpharmacologic option. Future studies could include investigating the benefit of BFA in the veteran population by studying larger samples in the ED, surveying patients after their interventions to identify rates AEs, and exploring the use of BFA for chronic pain in the outpatient setting.
1. Cantrill SV, Brown MD, Carlisle RJ, et al. Clinical policy: critical issues in the prescribing of opioids for adult patients in the emergency department. Ann Emerg Med. 2012;60(4):499-525. doi:10.1016/j.annemergmed.2012.06.013
2. Nahin RL. Severe pain in veterans: the effect of age and sex, and comparisons with the general population. J Pain. 2017;18(3):247-254. doi:10.1016/j.jpain.2016.10.021
3. Motov S, Strayer R, Hayes BD, et al. The treatment of acute pain in the emergency department: a white paper position statement prepared for the American Academy of Emergency Medicine. J Emerg Med. 2018;54(5):731-736. doi:10.1016/j.jemermed.2018.01.020
4. Samcam I, Papa L. Acute pain management in the emergency department. In: Prostran M, ed. Pain Management. IntechOpen; 2016. doi:10.5772/62861
5. Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline for the use of opioids in the management of chronic pain. Accessed February 15, 2023. https://www.healthquality.va.gov/guidelines/Pain/cot/VADoDOpioidsCPG.pdf
6. Davis MT, Mulvaney-Day N, Larson MJ, Hoover R, Mauch D. Complementary and alternative medicine among veterans and military personnel: a synthesis of population surveys. Med Care. 2014;52(12 suppl 5):S83-590. doi:10.1097/MLR.0000000000000227
7. Goertz C, Marriott BP, Finch FD, et al. Military report more complementary and alternative medicine use than civilians. J Altern Complement Med. 2013;19(6):509-517. doi:10.1089/acm.2012.0108
8. King HC, Hickey AH, Connelly C. Auricular acupuncture: a brief introduction for military providers. Mil Med. 2013;178(8):867-874. doi:10.7205/MILMED-D-13-00075
9. Niemtzow RC. Battlefield acupuncture. Medical Acupunct. 2007;19(4):225-228. doi:10.1089/acu.2007.0603
10. Collinsworth KM, Goss DL. Battlefield acupuncture and physical therapy versus physical therapy alone after shoulder surgery. Med Acupunct. 2019;31(4):228-238. doi:10.1089/acu.2019.1372
11. Estores I, Chen K, Jackson B, Lao L, Gorman PH. Auricular acupuncture for spinal cord injury related neuropathic pain: a pilot controlled clinical trial. J Spinal Cord Med. 2017;40(4):432-438. doi:10.1080/10790268.2016.1141489
12. Federman DG, Radhakrishnan K, Gabriel L, Poulin LM, Kravetz JD. Group battlefield acupuncture in primary care for veterans with pain. South Med J. 2018;111(10):619-624. doi:10.14423/SMJ.0000000000000877
13. Garner BK, Hopkinson SG, Ketz AK, Landis CA, Trego LL. Auricular acupuncture for chronic pain and insomnia: a randomized clinical trial. Med Acupunct. 2018;30(5):262-272. doi:10.1089/acu.2018.1294
14. Fox LM, Murakami M, Danesh H, Manini AF. Battlefield acupuncture to treat low back pain in the emergency department. Am J Emerg Med. 2018; 36:1045-1048. doi:10.1016/j.ajem.2018.02.038
15. Woo A, Lechner B, Fu T, et al. Cut points for mild, moderate, and severe pain among cancer and non-cancer patients: a literature review. Ann Palliat Med. 2015;4(4):176-183. doi:10.3978/j.issn.2224-5820.2015.09.04
16. Motov S, Yasavolian M, Likourezos A, et al. Comparison of intravenous ketorolac at three single-dose regimens for treating acute pain in the emergency department: a randomized controlled trial. Ann Emerg Med. 2017;70(2):177-184. doi:10.1016/j.annemergmed.2016.10.014
17. Bijur PE, Latimer CT, Gallagher EJ. Validation of a verbally administered numerical rating scale of acute pain for use in the emergency department. Acad Emerg Med. 2003;10:390-392. doi:10.1111/j.1553-2712.2003.tb01355.
1. Cantrill SV, Brown MD, Carlisle RJ, et al. Clinical policy: critical issues in the prescribing of opioids for adult patients in the emergency department. Ann Emerg Med. 2012;60(4):499-525. doi:10.1016/j.annemergmed.2012.06.013
2. Nahin RL. Severe pain in veterans: the effect of age and sex, and comparisons with the general population. J Pain. 2017;18(3):247-254. doi:10.1016/j.jpain.2016.10.021
3. Motov S, Strayer R, Hayes BD, et al. The treatment of acute pain in the emergency department: a white paper position statement prepared for the American Academy of Emergency Medicine. J Emerg Med. 2018;54(5):731-736. doi:10.1016/j.jemermed.2018.01.020
4. Samcam I, Papa L. Acute pain management in the emergency department. In: Prostran M, ed. Pain Management. IntechOpen; 2016. doi:10.5772/62861
5. Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline for the use of opioids in the management of chronic pain. Accessed February 15, 2023. https://www.healthquality.va.gov/guidelines/Pain/cot/VADoDOpioidsCPG.pdf
6. Davis MT, Mulvaney-Day N, Larson MJ, Hoover R, Mauch D. Complementary and alternative medicine among veterans and military personnel: a synthesis of population surveys. Med Care. 2014;52(12 suppl 5):S83-590. doi:10.1097/MLR.0000000000000227
7. Goertz C, Marriott BP, Finch FD, et al. Military report more complementary and alternative medicine use than civilians. J Altern Complement Med. 2013;19(6):509-517. doi:10.1089/acm.2012.0108
8. King HC, Hickey AH, Connelly C. Auricular acupuncture: a brief introduction for military providers. Mil Med. 2013;178(8):867-874. doi:10.7205/MILMED-D-13-00075
9. Niemtzow RC. Battlefield acupuncture. Medical Acupunct. 2007;19(4):225-228. doi:10.1089/acu.2007.0603
10. Collinsworth KM, Goss DL. Battlefield acupuncture and physical therapy versus physical therapy alone after shoulder surgery. Med Acupunct. 2019;31(4):228-238. doi:10.1089/acu.2019.1372
11. Estores I, Chen K, Jackson B, Lao L, Gorman PH. Auricular acupuncture for spinal cord injury related neuropathic pain: a pilot controlled clinical trial. J Spinal Cord Med. 2017;40(4):432-438. doi:10.1080/10790268.2016.1141489
12. Federman DG, Radhakrishnan K, Gabriel L, Poulin LM, Kravetz JD. Group battlefield acupuncture in primary care for veterans with pain. South Med J. 2018;111(10):619-624. doi:10.14423/SMJ.0000000000000877
13. Garner BK, Hopkinson SG, Ketz AK, Landis CA, Trego LL. Auricular acupuncture for chronic pain and insomnia: a randomized clinical trial. Med Acupunct. 2018;30(5):262-272. doi:10.1089/acu.2018.1294
14. Fox LM, Murakami M, Danesh H, Manini AF. Battlefield acupuncture to treat low back pain in the emergency department. Am J Emerg Med. 2018; 36:1045-1048. doi:10.1016/j.ajem.2018.02.038
15. Woo A, Lechner B, Fu T, et al. Cut points for mild, moderate, and severe pain among cancer and non-cancer patients: a literature review. Ann Palliat Med. 2015;4(4):176-183. doi:10.3978/j.issn.2224-5820.2015.09.04
16. Motov S, Yasavolian M, Likourezos A, et al. Comparison of intravenous ketorolac at three single-dose regimens for treating acute pain in the emergency department: a randomized controlled trial. Ann Emerg Med. 2017;70(2):177-184. doi:10.1016/j.annemergmed.2016.10.014
17. Bijur PE, Latimer CT, Gallagher EJ. Validation of a verbally administered numerical rating scale of acute pain for use in the emergency department. Acad Emerg Med. 2003;10:390-392. doi:10.1111/j.1553-2712.2003.tb01355.