User login
Real-Time Monitoring of Melanoma Markers Predicts Relapse
SEOUL, SOUTH KOREA – Serial monitoring of melanoma tumor marker levels in peripheral blood using a novel quantitative real-time reverse-transcriptase polymerase chain reaction method after surgical resection of melanoma has shown promise for the early detection of patients at high risk for disease progression.
The real-time polymerase chain reaction (PCR) assay measures circulating levels of five markers unique to melanoma cells: glycoprotein 100 (gp100), melanoma antigen gene-3 (MAGE-3), tyrosinase, melanoma marker A (Melan-A), and melanoma inhibitory activity (MIA) protein, Dr. Spyridon Gkalpakiotis explained at the World Congress of Dermatology.
He reported on 65 patients who underwent peripheral blood testing and analysis of the five markers every 3 months for the first 2 years after resection of their stage II or III melanoma, for a total of 2,925 PCR assays.
Twenty-six patients experienced elevated test results. All 26 relapsed during 5 years of follow-up; the 5-year survival rate in this group was 65%.
In contrast, only 1 of 39 patients with consistently negative real-time PCR assays experienced disease progression; 5-year survival in PCR-negative patients was 97%, reported Dr. Gkalpakiotis of Charles University in Prague.
MAGE-3 was expressed in 21 patients with disease progression. The next most sensitive markers of melanoma progression were MIA and gp100.
Dr. Gkalpakiotis declared having no financial conflicts.
SEOUL, SOUTH KOREA – Serial monitoring of melanoma tumor marker levels in peripheral blood using a novel quantitative real-time reverse-transcriptase polymerase chain reaction method after surgical resection of melanoma has shown promise for the early detection of patients at high risk for disease progression.
The real-time polymerase chain reaction (PCR) assay measures circulating levels of five markers unique to melanoma cells: glycoprotein 100 (gp100), melanoma antigen gene-3 (MAGE-3), tyrosinase, melanoma marker A (Melan-A), and melanoma inhibitory activity (MIA) protein, Dr. Spyridon Gkalpakiotis explained at the World Congress of Dermatology.
He reported on 65 patients who underwent peripheral blood testing and analysis of the five markers every 3 months for the first 2 years after resection of their stage II or III melanoma, for a total of 2,925 PCR assays.
Twenty-six patients experienced elevated test results. All 26 relapsed during 5 years of follow-up; the 5-year survival rate in this group was 65%.
In contrast, only 1 of 39 patients with consistently negative real-time PCR assays experienced disease progression; 5-year survival in PCR-negative patients was 97%, reported Dr. Gkalpakiotis of Charles University in Prague.
MAGE-3 was expressed in 21 patients with disease progression. The next most sensitive markers of melanoma progression were MIA and gp100.
Dr. Gkalpakiotis declared having no financial conflicts.
SEOUL, SOUTH KOREA – Serial monitoring of melanoma tumor marker levels in peripheral blood using a novel quantitative real-time reverse-transcriptase polymerase chain reaction method after surgical resection of melanoma has shown promise for the early detection of patients at high risk for disease progression.
The real-time polymerase chain reaction (PCR) assay measures circulating levels of five markers unique to melanoma cells: glycoprotein 100 (gp100), melanoma antigen gene-3 (MAGE-3), tyrosinase, melanoma marker A (Melan-A), and melanoma inhibitory activity (MIA) protein, Dr. Spyridon Gkalpakiotis explained at the World Congress of Dermatology.
He reported on 65 patients who underwent peripheral blood testing and analysis of the five markers every 3 months for the first 2 years after resection of their stage II or III melanoma, for a total of 2,925 PCR assays.
Twenty-six patients experienced elevated test results. All 26 relapsed during 5 years of follow-up; the 5-year survival rate in this group was 65%.
In contrast, only 1 of 39 patients with consistently negative real-time PCR assays experienced disease progression; 5-year survival in PCR-negative patients was 97%, reported Dr. Gkalpakiotis of Charles University in Prague.
MAGE-3 was expressed in 21 patients with disease progression. The next most sensitive markers of melanoma progression were MIA and gp100.
Dr. Gkalpakiotis declared having no financial conflicts.
FROM THE WORLD CONGRESS OF DERMATOLOGY
Melanoma Treatment Ipilimumab Approved Europe-Wide
The European Commission has approved ipilimumab, a novel immunotherapy, for the second-line treatment of adult patients with previously treated, advanced melanoma, the medicine’s manufacturer said July 14.
Ipilimumab (Bristol-Myers Squibb’s Yervoy) was recommended in May by the European Medicines Agency for the indication, and was approved in March by the U.S. Food and Drug Administration. Ipilimumab, a monoclonal antibody, causes tumor cell death by blocking the inhibitory signal of CTLA-4, resulting in T-cell activation, proliferation, and lymphocyte infiltration into tumors.
The medication was shown in one manufacturer-funded, randomized, controlled trial to prolong overall survival for an unprecedented amount of time for people with metastasized unresectable melanoma (N. Engl. J. Med. 2010; 363:711-23).
Patients in the trial (n = 676) received ipilimumab plus glycoprotein 100 (gp100), gp100 alone, or ipilimumab alone, in four intravenous infusions at 3 mg/kg of body weight over 12 weeks. Patients who received the combination therapy or ipilimumab alone saw a median overall survival of about 10 months, compared with 6.4 months for those receiving only gp100. Gp100 was not found to improve the efficacy of ipilimumab.
Responding patients could receive additional courses of ipilimumab, and more than a fifth of patients in the ipilimumab arms survived 2 years or longer.
Adverse reactions to ipilimumab were mostly immune related, and included enterocolitis, hepatitis, dermatitis (including toxic epidermal necrolysis), neuropathy, and endocrinopathy.
European Commission approval means that ipilimumab will be available to patients throughout the European Union. However, pricing may differ, depending on procurement and patient-access schemes in each country. In the United States, ipilimumab is estimated to cost $120,000 per course of treatment.
The European Commission has approved ipilimumab, a novel immunotherapy, for the second-line treatment of adult patients with previously treated, advanced melanoma, the medicine’s manufacturer said July 14.
Ipilimumab (Bristol-Myers Squibb’s Yervoy) was recommended in May by the European Medicines Agency for the indication, and was approved in March by the U.S. Food and Drug Administration. Ipilimumab, a monoclonal antibody, causes tumor cell death by blocking the inhibitory signal of CTLA-4, resulting in T-cell activation, proliferation, and lymphocyte infiltration into tumors.
The medication was shown in one manufacturer-funded, randomized, controlled trial to prolong overall survival for an unprecedented amount of time for people with metastasized unresectable melanoma (N. Engl. J. Med. 2010; 363:711-23).
Patients in the trial (n = 676) received ipilimumab plus glycoprotein 100 (gp100), gp100 alone, or ipilimumab alone, in four intravenous infusions at 3 mg/kg of body weight over 12 weeks. Patients who received the combination therapy or ipilimumab alone saw a median overall survival of about 10 months, compared with 6.4 months for those receiving only gp100. Gp100 was not found to improve the efficacy of ipilimumab.
Responding patients could receive additional courses of ipilimumab, and more than a fifth of patients in the ipilimumab arms survived 2 years or longer.
Adverse reactions to ipilimumab were mostly immune related, and included enterocolitis, hepatitis, dermatitis (including toxic epidermal necrolysis), neuropathy, and endocrinopathy.
European Commission approval means that ipilimumab will be available to patients throughout the European Union. However, pricing may differ, depending on procurement and patient-access schemes in each country. In the United States, ipilimumab is estimated to cost $120,000 per course of treatment.
The European Commission has approved ipilimumab, a novel immunotherapy, for the second-line treatment of adult patients with previously treated, advanced melanoma, the medicine’s manufacturer said July 14.
Ipilimumab (Bristol-Myers Squibb’s Yervoy) was recommended in May by the European Medicines Agency for the indication, and was approved in March by the U.S. Food and Drug Administration. Ipilimumab, a monoclonal antibody, causes tumor cell death by blocking the inhibitory signal of CTLA-4, resulting in T-cell activation, proliferation, and lymphocyte infiltration into tumors.
The medication was shown in one manufacturer-funded, randomized, controlled trial to prolong overall survival for an unprecedented amount of time for people with metastasized unresectable melanoma (N. Engl. J. Med. 2010; 363:711-23).
Patients in the trial (n = 676) received ipilimumab plus glycoprotein 100 (gp100), gp100 alone, or ipilimumab alone, in four intravenous infusions at 3 mg/kg of body weight over 12 weeks. Patients who received the combination therapy or ipilimumab alone saw a median overall survival of about 10 months, compared with 6.4 months for those receiving only gp100. Gp100 was not found to improve the efficacy of ipilimumab.
Responding patients could receive additional courses of ipilimumab, and more than a fifth of patients in the ipilimumab arms survived 2 years or longer.
Adverse reactions to ipilimumab were mostly immune related, and included enterocolitis, hepatitis, dermatitis (including toxic epidermal necrolysis), neuropathy, and endocrinopathy.
European Commission approval means that ipilimumab will be available to patients throughout the European Union. However, pricing may differ, depending on procurement and patient-access schemes in each country. In the United States, ipilimumab is estimated to cost $120,000 per course of treatment.
Cancer-Protective Effects Seen for Type IV Allergies
Patients with a history of contact allergies may be less likely to develop nonmelanoma skin cancer, brain cancer, and breast cancer, a study has shown.
In research published online July 11 in BMJ Open (doi:10.1136/bmjopen-2011-000084), investigators conversely found diagnoses of bladder cancer more likely in people with a history of contact allergies.
Any protective effects could possibly be explained by the stimulation of natural killer T (NKT) cells through contact allergic reactions, the observational study’s lead author, Kaare Engkilde, Ph.D., of the national allergy research center at Copenhagen University Hospital, said in an interview. "You would probably think that [contact allergy] could lead to skin cancer, but that is not what we found," Dr. Engkilde said.
Contact allergic reactions are delayed reactions caused by chemicals small enough to penetrate the skin – triggers can include perfumes, hair dyes, cobalt, nickel, and formaldehyde. Animal studies have shown that contact allergens have the ability to increase the number of NKT cells, at least temporarily, Dr. Engkilde said.
For their research, Dr. Engkilde and his colleagues used a database of 16,922 Danish patients patch tested for type IV allergies between 1984 and 2008. Of these, 6,065 (35.8%) had at least one positive reaction (26.1% of men, 41.4% of women). Linkage with the Danish Cancer Registry showed 3,200, or 18.9%, of dermatitis patients had a benign tumor, a malignant cancer diagnosis, or both, and 1,207 (37.7%) of these patients had had a positive patch test reaction.
Dr. Engkilde and his colleagues looked specifically at 15 types of cancers, all of which affected at least 40 people in the study population.
The investigators found an inverse association between diagnosed contact allergy and nonmelanoma skin cancer (OR, 0.82) along with breast cancer (OR, 0.80) among both men and women. Dr. Engkilde and his colleagues also noted an inverse trend for brain cancer among women with contact allergies, though the P value was above.05 (OR, 0.36).
The findings related to nonmelanoma skin cancers were on some level expected, Dr. Engkilde said; however, the researchers said they were surprised not to see any protective effect against melanomas.
Dr. Engkilde said his group had no hypothesis for the inverse breast cancer finding, but noted that an earlier study had found that self-reported perfume allergy, a type IV allergy, was related inversely to brain cancer incidence (Am. J. Epidemiol. 2007;166:941-50).
The positive association between contact allergy and bladder cancer (OR, 1.44) was more likely related to accumulations of chemical metabolites of type IV allergens in the bladder than to an effect on NKT cells or another immune response, the investigators said.
They cautioned that the relationships between type IV reactions and cancers were "uncertain and not necessarily the result of causality," and that future analyses would have to adjust for social class and smoking, as the latter can increase the risk of developing nickel contact allergies and several types of cancer.
Also, they wrote, "studies focusing on specific chemical exposures are required to further our understanding of the role of contact allergies in the development of cancer." However, if the relationships are in fact etiologic, the findings have "implications for understanding how contact allergy can affect cancer development and vice versa," they said.
Dr. Engkilde and his coauthors received research support from Aage Bang’s Foundation and the Capital Region’s Research Foundation, and disclosed no competing interests.
Patients with a history of contact allergies may be less likely to develop nonmelanoma skin cancer, brain cancer, and breast cancer, a study has shown.
In research published online July 11 in BMJ Open (doi:10.1136/bmjopen-2011-000084), investigators conversely found diagnoses of bladder cancer more likely in people with a history of contact allergies.
Any protective effects could possibly be explained by the stimulation of natural killer T (NKT) cells through contact allergic reactions, the observational study’s lead author, Kaare Engkilde, Ph.D., of the national allergy research center at Copenhagen University Hospital, said in an interview. "You would probably think that [contact allergy] could lead to skin cancer, but that is not what we found," Dr. Engkilde said.
Contact allergic reactions are delayed reactions caused by chemicals small enough to penetrate the skin – triggers can include perfumes, hair dyes, cobalt, nickel, and formaldehyde. Animal studies have shown that contact allergens have the ability to increase the number of NKT cells, at least temporarily, Dr. Engkilde said.
For their research, Dr. Engkilde and his colleagues used a database of 16,922 Danish patients patch tested for type IV allergies between 1984 and 2008. Of these, 6,065 (35.8%) had at least one positive reaction (26.1% of men, 41.4% of women). Linkage with the Danish Cancer Registry showed 3,200, or 18.9%, of dermatitis patients had a benign tumor, a malignant cancer diagnosis, or both, and 1,207 (37.7%) of these patients had had a positive patch test reaction.
Dr. Engkilde and his colleagues looked specifically at 15 types of cancers, all of which affected at least 40 people in the study population.
The investigators found an inverse association between diagnosed contact allergy and nonmelanoma skin cancer (OR, 0.82) along with breast cancer (OR, 0.80) among both men and women. Dr. Engkilde and his colleagues also noted an inverse trend for brain cancer among women with contact allergies, though the P value was above.05 (OR, 0.36).
The findings related to nonmelanoma skin cancers were on some level expected, Dr. Engkilde said; however, the researchers said they were surprised not to see any protective effect against melanomas.
Dr. Engkilde said his group had no hypothesis for the inverse breast cancer finding, but noted that an earlier study had found that self-reported perfume allergy, a type IV allergy, was related inversely to brain cancer incidence (Am. J. Epidemiol. 2007;166:941-50).
The positive association between contact allergy and bladder cancer (OR, 1.44) was more likely related to accumulations of chemical metabolites of type IV allergens in the bladder than to an effect on NKT cells or another immune response, the investigators said.
They cautioned that the relationships between type IV reactions and cancers were "uncertain and not necessarily the result of causality," and that future analyses would have to adjust for social class and smoking, as the latter can increase the risk of developing nickel contact allergies and several types of cancer.
Also, they wrote, "studies focusing on specific chemical exposures are required to further our understanding of the role of contact allergies in the development of cancer." However, if the relationships are in fact etiologic, the findings have "implications for understanding how contact allergy can affect cancer development and vice versa," they said.
Dr. Engkilde and his coauthors received research support from Aage Bang’s Foundation and the Capital Region’s Research Foundation, and disclosed no competing interests.
Patients with a history of contact allergies may be less likely to develop nonmelanoma skin cancer, brain cancer, and breast cancer, a study has shown.
In research published online July 11 in BMJ Open (doi:10.1136/bmjopen-2011-000084), investigators conversely found diagnoses of bladder cancer more likely in people with a history of contact allergies.
Any protective effects could possibly be explained by the stimulation of natural killer T (NKT) cells through contact allergic reactions, the observational study’s lead author, Kaare Engkilde, Ph.D., of the national allergy research center at Copenhagen University Hospital, said in an interview. "You would probably think that [contact allergy] could lead to skin cancer, but that is not what we found," Dr. Engkilde said.
Contact allergic reactions are delayed reactions caused by chemicals small enough to penetrate the skin – triggers can include perfumes, hair dyes, cobalt, nickel, and formaldehyde. Animal studies have shown that contact allergens have the ability to increase the number of NKT cells, at least temporarily, Dr. Engkilde said.
For their research, Dr. Engkilde and his colleagues used a database of 16,922 Danish patients patch tested for type IV allergies between 1984 and 2008. Of these, 6,065 (35.8%) had at least one positive reaction (26.1% of men, 41.4% of women). Linkage with the Danish Cancer Registry showed 3,200, or 18.9%, of dermatitis patients had a benign tumor, a malignant cancer diagnosis, or both, and 1,207 (37.7%) of these patients had had a positive patch test reaction.
Dr. Engkilde and his colleagues looked specifically at 15 types of cancers, all of which affected at least 40 people in the study population.
The investigators found an inverse association between diagnosed contact allergy and nonmelanoma skin cancer (OR, 0.82) along with breast cancer (OR, 0.80) among both men and women. Dr. Engkilde and his colleagues also noted an inverse trend for brain cancer among women with contact allergies, though the P value was above.05 (OR, 0.36).
The findings related to nonmelanoma skin cancers were on some level expected, Dr. Engkilde said; however, the researchers said they were surprised not to see any protective effect against melanomas.
Dr. Engkilde said his group had no hypothesis for the inverse breast cancer finding, but noted that an earlier study had found that self-reported perfume allergy, a type IV allergy, was related inversely to brain cancer incidence (Am. J. Epidemiol. 2007;166:941-50).
The positive association between contact allergy and bladder cancer (OR, 1.44) was more likely related to accumulations of chemical metabolites of type IV allergens in the bladder than to an effect on NKT cells or another immune response, the investigators said.
They cautioned that the relationships between type IV reactions and cancers were "uncertain and not necessarily the result of causality," and that future analyses would have to adjust for social class and smoking, as the latter can increase the risk of developing nickel contact allergies and several types of cancer.
Also, they wrote, "studies focusing on specific chemical exposures are required to further our understanding of the role of contact allergies in the development of cancer." However, if the relationships are in fact etiologic, the findings have "implications for understanding how contact allergy can affect cancer development and vice versa," they said.
Dr. Engkilde and his coauthors received research support from Aage Bang’s Foundation and the Capital Region’s Research Foundation, and disclosed no competing interests.
FROM BMJ OPEN
Major Finding: The investigators found an inverse association between diagnosed contact allergy and nonmelanoma skin cancer (OR, 0.82) along with breast cancer (OR 0.80) among both men and women.
Data Source: A database of 16,922 Danish patients patch tested for type IV allergies between 1984 and 2008.
Disclosures: Dr. Engkilde and his coauthors received research support from Aage Bang’s Foundation and the Capital Region’s Research Foundation, and disclosed no competing interests.
Steaming Veins: The Skinny Podcast
In this month's podcast, Skin & Allergy News' reporters discuss cancer, wrinkles, and veins.
Positive clinical trial results for new melanoma therapies were presented at the American Society of Clinical Oncology. Jeff Evans interviews Dr. Lynn Schuchter about the findings, and what they mean for patients.
Also in this episode, Heidi Splete offers a report on the link between wrinkles and bone health, which may have you sending your wrinkled patients to the bone doctor.
Then, Sherry Boschert talks with Dr. Rene Milleret about a new endovenous ablation technique for varicose veins that uses steam; the procedure is catching on in Europe.
In this month's Cosmetic Counter segment, Dr. Lily Talakoub tackles nanotechnology.
And finally, Dr. Alan Rockoff assures everyone that, contrary to any rumors, he has not kicked the bucket.
In this month's podcast, Skin & Allergy News' reporters discuss cancer, wrinkles, and veins.
Positive clinical trial results for new melanoma therapies were presented at the American Society of Clinical Oncology. Jeff Evans interviews Dr. Lynn Schuchter about the findings, and what they mean for patients.
Also in this episode, Heidi Splete offers a report on the link between wrinkles and bone health, which may have you sending your wrinkled patients to the bone doctor.
Then, Sherry Boschert talks with Dr. Rene Milleret about a new endovenous ablation technique for varicose veins that uses steam; the procedure is catching on in Europe.
In this month's Cosmetic Counter segment, Dr. Lily Talakoub tackles nanotechnology.
And finally, Dr. Alan Rockoff assures everyone that, contrary to any rumors, he has not kicked the bucket.
In this month's podcast, Skin & Allergy News' reporters discuss cancer, wrinkles, and veins.
Positive clinical trial results for new melanoma therapies were presented at the American Society of Clinical Oncology. Jeff Evans interviews Dr. Lynn Schuchter about the findings, and what they mean for patients.
Also in this episode, Heidi Splete offers a report on the link between wrinkles and bone health, which may have you sending your wrinkled patients to the bone doctor.
Then, Sherry Boschert talks with Dr. Rene Milleret about a new endovenous ablation technique for varicose veins that uses steam; the procedure is catching on in Europe.
In this month's Cosmetic Counter segment, Dr. Lily Talakoub tackles nanotechnology.
And finally, Dr. Alan Rockoff assures everyone that, contrary to any rumors, he has not kicked the bucket.
Purse String Stitch Handy for Lip Defects
LAS VEGAS – A simple purse string stitch can provide an elegant closure of challenging lip defects after Mohs surgery.
"It's quite easy to perform. It's useful for numerous areas of the lip. It's low-risk, with low morbidity, and requires little down time," yet provides an excellent aesthetic outcome, Dr. Kenny J. Omlin said at the annual meeting of the American College of Mohs Surgery.
The keys to reconstruction using the purse string stitch start with undermining the entire surgical wound in the subdermal plane to decrease sheering forces.
Next, uniformly place an absorbable suture in the deep dermis using a small needle, with circumferential tissue advancement to distribute the tension uniformly, explained Dr. Omlin, chief of Mohs surgery for Kaiser Permanente Napa-Solano County, Vacaville, Calif.
"There is a uniform stitch all the way around the perimeter" of the wound with particular attention to precisely aligning the vermilion/cutaneous lip junction, he said.
The purse string stitch creates a trestle-like framework that supports normal wound healing.
As with any reconstructions on the lower cosmetic subunits of the face, he tells patients to practice a "ventriloquist act" while healing and not talk much or move their mouths much.
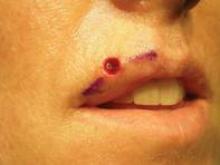
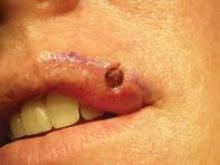
One of his patients provided an excellent case-control comparison of wound closures. She initially presented with a basal cell carcinoma that intersected both the cutaneous and vermilion margins of her left upper lip.
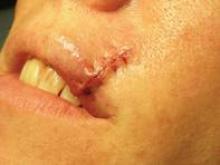
After Mohs surgery, Dr. Omlin did a standard, complex linear closure, followed later by two pulsed-dye laser treatments. At 1-year follow-up, the patient was satisfied with an acceptable cosmetic outcome.
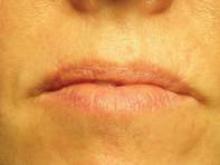
Six months later, she presented with a nearly identical basal cell carcinoma on the right upper lip. This time, Dr. Omlin used a purse string stitch after Mohs surgery. "It takes all of 5-10 minutes," he noted. The aesthetic result was "nearly perfect" a month later, said Dr. Omlin, also of the University of California, Davis.
On the central upper lip, "a lot of our older patients tend to have absent philtral columns or an absent Cupid's bow. Again, this is an excellent place for a purse string stitch," he said.
For patients on warfarin, the purse string stitch is great for hemostasis when repairing Mohs defects of the lip, Dr. Omlin added.
He also likes to use it for defects at the oral commissure. "Sure, you can use an elaborate cross-lip commissuroplasty or other elaborate techniques," but a simple purse string stitch reproduces the accordion-like structure of the oral commissure.
After wound granulation and healing, at 3 months it's hard to tell that a defect was ever there, he said.
In an interview after his presentation, he said he was pleased by the enthusiasm expressed by other attendees at the meeting for his simple surgical pearl.
Dr. Omlin said he has no relevant financial disclosures.
LAS VEGAS – A simple purse string stitch can provide an elegant closure of challenging lip defects after Mohs surgery.
"It's quite easy to perform. It's useful for numerous areas of the lip. It's low-risk, with low morbidity, and requires little down time," yet provides an excellent aesthetic outcome, Dr. Kenny J. Omlin said at the annual meeting of the American College of Mohs Surgery.
The keys to reconstruction using the purse string stitch start with undermining the entire surgical wound in the subdermal plane to decrease sheering forces.
Next, uniformly place an absorbable suture in the deep dermis using a small needle, with circumferential tissue advancement to distribute the tension uniformly, explained Dr. Omlin, chief of Mohs surgery for Kaiser Permanente Napa-Solano County, Vacaville, Calif.
"There is a uniform stitch all the way around the perimeter" of the wound with particular attention to precisely aligning the vermilion/cutaneous lip junction, he said.
The purse string stitch creates a trestle-like framework that supports normal wound healing.
As with any reconstructions on the lower cosmetic subunits of the face, he tells patients to practice a "ventriloquist act" while healing and not talk much or move their mouths much.
One of his patients provided an excellent case-control comparison of wound closures. She initially presented with a basal cell carcinoma that intersected both the cutaneous and vermilion margins of her left upper lip.
After Mohs surgery, Dr. Omlin did a standard, complex linear closure, followed later by two pulsed-dye laser treatments. At 1-year follow-up, the patient was satisfied with an acceptable cosmetic outcome.
Six months later, she presented with a nearly identical basal cell carcinoma on the right upper lip. This time, Dr. Omlin used a purse string stitch after Mohs surgery. "It takes all of 5-10 minutes," he noted. The aesthetic result was "nearly perfect" a month later, said Dr. Omlin, also of the University of California, Davis.
On the central upper lip, "a lot of our older patients tend to have absent philtral columns or an absent Cupid's bow. Again, this is an excellent place for a purse string stitch," he said.
For patients on warfarin, the purse string stitch is great for hemostasis when repairing Mohs defects of the lip, Dr. Omlin added.
He also likes to use it for defects at the oral commissure. "Sure, you can use an elaborate cross-lip commissuroplasty or other elaborate techniques," but a simple purse string stitch reproduces the accordion-like structure of the oral commissure.
After wound granulation and healing, at 3 months it's hard to tell that a defect was ever there, he said.
In an interview after his presentation, he said he was pleased by the enthusiasm expressed by other attendees at the meeting for his simple surgical pearl.
Dr. Omlin said he has no relevant financial disclosures.
LAS VEGAS – A simple purse string stitch can provide an elegant closure of challenging lip defects after Mohs surgery.
"It's quite easy to perform. It's useful for numerous areas of the lip. It's low-risk, with low morbidity, and requires little down time," yet provides an excellent aesthetic outcome, Dr. Kenny J. Omlin said at the annual meeting of the American College of Mohs Surgery.
The keys to reconstruction using the purse string stitch start with undermining the entire surgical wound in the subdermal plane to decrease sheering forces.
Next, uniformly place an absorbable suture in the deep dermis using a small needle, with circumferential tissue advancement to distribute the tension uniformly, explained Dr. Omlin, chief of Mohs surgery for Kaiser Permanente Napa-Solano County, Vacaville, Calif.
"There is a uniform stitch all the way around the perimeter" of the wound with particular attention to precisely aligning the vermilion/cutaneous lip junction, he said.
The purse string stitch creates a trestle-like framework that supports normal wound healing.
As with any reconstructions on the lower cosmetic subunits of the face, he tells patients to practice a "ventriloquist act" while healing and not talk much or move their mouths much.
One of his patients provided an excellent case-control comparison of wound closures. She initially presented with a basal cell carcinoma that intersected both the cutaneous and vermilion margins of her left upper lip.
After Mohs surgery, Dr. Omlin did a standard, complex linear closure, followed later by two pulsed-dye laser treatments. At 1-year follow-up, the patient was satisfied with an acceptable cosmetic outcome.
Six months later, she presented with a nearly identical basal cell carcinoma on the right upper lip. This time, Dr. Omlin used a purse string stitch after Mohs surgery. "It takes all of 5-10 minutes," he noted. The aesthetic result was "nearly perfect" a month later, said Dr. Omlin, also of the University of California, Davis.
On the central upper lip, "a lot of our older patients tend to have absent philtral columns or an absent Cupid's bow. Again, this is an excellent place for a purse string stitch," he said.
For patients on warfarin, the purse string stitch is great for hemostasis when repairing Mohs defects of the lip, Dr. Omlin added.
He also likes to use it for defects at the oral commissure. "Sure, you can use an elaborate cross-lip commissuroplasty or other elaborate techniques," but a simple purse string stitch reproduces the accordion-like structure of the oral commissure.
After wound granulation and healing, at 3 months it's hard to tell that a defect was ever there, he said.
In an interview after his presentation, he said he was pleased by the enthusiasm expressed by other attendees at the meeting for his simple surgical pearl.
Dr. Omlin said he has no relevant financial disclosures.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE AMERICAN COLLEGE OF MOHS SURGERY
Anchoring Cartilage Grafts to Alar Rim Is Simple, Effective
LAS VEGAS – Traditional techniques for anchoring cartilage grafts after Mohs surgery on the nose might be more complicated than necessary.
Well-known textbook descriptions of cartilage grafting for alar rim reconstruction involve harvesting a large piece of cartilage and securing the grafted cartilage with multiple sutures medially to the lower lateral cartilage and laterally to the periosteum of the piriform aperture of the maxilla.
This technique is appropriate for large defects that require reconstruction of the entire alar subunit, but is unnecessary for many of the smaller alar defects that commonly are encountered in Mohs surgery, Dr. Ravi S. Krishnan said.
"While this does produce nice results, I don't like it for two reasons," he said at the annual meeting of the American College of Mohs Surgery. "It requires a very large cartilage graft, and it often requires more effort than you sometimes need."
In his approach to performing a cartilage graft with a skin graft on top of it, he starts the conventional way by making some stab incisions on either side of the wound to create a pocket for the cartilage graft. What most surgeons would do next is to place either a figure-of-eight suture or some interrupted sutures to hold the graft in place.
"While these techniques are fine, I sometimes still have problems with them getting the cartilage graft flush against the alar remnant," noted Dr. Krishnan of Virginia Mason Medical Center, Seattle.
Instead, he starts suturing from inside the nose, pushing the suture through the nose behind the cartilage graft, then pulling it back through the cartilage graft and the nose, finally pulling inferiorly as the knot is tied. He repeats this process, so that there are two sutures anchoring the cartilage graft along the alar rim. These sutures are left in place for 2 weeks to allow some fibrosis to occur.
"The reason I like this technique is because it pulls the graft inferiorly so that it's perfectly flush against the alar rim remnant," he said. It also leaves more of the wound base exposed so that any overlying skin graft will be well perfused.
This is not necessarily a new technique, Dr. Krishnan said, but he could find no published description of it. It doesn't necessarily give better results, but it's easier to execute than are traditional methods, he added.
His techniques for anchoring cartilage grafts along the alar rim also work well with flaps including nasolabial transposition flaps, bilobed flaps, and interpolated paranasal flaps, resulting in good contour and symmetry and very acceptable results, he believes.
He typically follows these patients for 6 months after surgery, and while it's possible that the cartilage graft could shift after 6 months, "I doubt this would be the case."
One small drawback is that epithelium can start growing over the anchoring sutures during the 2 weeks that they are left in place, making them difficult to remove.
"Some people worry about infection, but I've never seen an infection with this technique," he added. All patients in his practice that receive cartilage grafts get perioperative antibiotics.
The advantages outweigh any potential drawbacks of the technique, in his opinion. It's easy to perform, and results are at least as good as those with more difficult techniques. His method precisely places the cartilage "exactly where you want it" along the alar rim, and apposes the cartilage graft to the mucosal lining, he said. When used in conjunction with a full-thickness skin graft, it allows the skin graft to come into contact with as much of the base of the wound as possible.
"It is important to remember that this technique is applicable only to smaller alar defects," he said. "For larger alar defects, using a large cartilage graft secured in the traditional manner is the preferred approach."
Dr. Krishnan said he has no relevant financial disclosures.
LAS VEGAS – Traditional techniques for anchoring cartilage grafts after Mohs surgery on the nose might be more complicated than necessary.
Well-known textbook descriptions of cartilage grafting for alar rim reconstruction involve harvesting a large piece of cartilage and securing the grafted cartilage with multiple sutures medially to the lower lateral cartilage and laterally to the periosteum of the piriform aperture of the maxilla.
This technique is appropriate for large defects that require reconstruction of the entire alar subunit, but is unnecessary for many of the smaller alar defects that commonly are encountered in Mohs surgery, Dr. Ravi S. Krishnan said.
"While this does produce nice results, I don't like it for two reasons," he said at the annual meeting of the American College of Mohs Surgery. "It requires a very large cartilage graft, and it often requires more effort than you sometimes need."
In his approach to performing a cartilage graft with a skin graft on top of it, he starts the conventional way by making some stab incisions on either side of the wound to create a pocket for the cartilage graft. What most surgeons would do next is to place either a figure-of-eight suture or some interrupted sutures to hold the graft in place.
"While these techniques are fine, I sometimes still have problems with them getting the cartilage graft flush against the alar remnant," noted Dr. Krishnan of Virginia Mason Medical Center, Seattle.
Instead, he starts suturing from inside the nose, pushing the suture through the nose behind the cartilage graft, then pulling it back through the cartilage graft and the nose, finally pulling inferiorly as the knot is tied. He repeats this process, so that there are two sutures anchoring the cartilage graft along the alar rim. These sutures are left in place for 2 weeks to allow some fibrosis to occur.
"The reason I like this technique is because it pulls the graft inferiorly so that it's perfectly flush against the alar rim remnant," he said. It also leaves more of the wound base exposed so that any overlying skin graft will be well perfused.
This is not necessarily a new technique, Dr. Krishnan said, but he could find no published description of it. It doesn't necessarily give better results, but it's easier to execute than are traditional methods, he added.
His techniques for anchoring cartilage grafts along the alar rim also work well with flaps including nasolabial transposition flaps, bilobed flaps, and interpolated paranasal flaps, resulting in good contour and symmetry and very acceptable results, he believes.
He typically follows these patients for 6 months after surgery, and while it's possible that the cartilage graft could shift after 6 months, "I doubt this would be the case."
One small drawback is that epithelium can start growing over the anchoring sutures during the 2 weeks that they are left in place, making them difficult to remove.
"Some people worry about infection, but I've never seen an infection with this technique," he added. All patients in his practice that receive cartilage grafts get perioperative antibiotics.
The advantages outweigh any potential drawbacks of the technique, in his opinion. It's easy to perform, and results are at least as good as those with more difficult techniques. His method precisely places the cartilage "exactly where you want it" along the alar rim, and apposes the cartilage graft to the mucosal lining, he said. When used in conjunction with a full-thickness skin graft, it allows the skin graft to come into contact with as much of the base of the wound as possible.
"It is important to remember that this technique is applicable only to smaller alar defects," he said. "For larger alar defects, using a large cartilage graft secured in the traditional manner is the preferred approach."
Dr. Krishnan said he has no relevant financial disclosures.
LAS VEGAS – Traditional techniques for anchoring cartilage grafts after Mohs surgery on the nose might be more complicated than necessary.
Well-known textbook descriptions of cartilage grafting for alar rim reconstruction involve harvesting a large piece of cartilage and securing the grafted cartilage with multiple sutures medially to the lower lateral cartilage and laterally to the periosteum of the piriform aperture of the maxilla.
This technique is appropriate for large defects that require reconstruction of the entire alar subunit, but is unnecessary for many of the smaller alar defects that commonly are encountered in Mohs surgery, Dr. Ravi S. Krishnan said.
"While this does produce nice results, I don't like it for two reasons," he said at the annual meeting of the American College of Mohs Surgery. "It requires a very large cartilage graft, and it often requires more effort than you sometimes need."
In his approach to performing a cartilage graft with a skin graft on top of it, he starts the conventional way by making some stab incisions on either side of the wound to create a pocket for the cartilage graft. What most surgeons would do next is to place either a figure-of-eight suture or some interrupted sutures to hold the graft in place.
"While these techniques are fine, I sometimes still have problems with them getting the cartilage graft flush against the alar remnant," noted Dr. Krishnan of Virginia Mason Medical Center, Seattle.
Instead, he starts suturing from inside the nose, pushing the suture through the nose behind the cartilage graft, then pulling it back through the cartilage graft and the nose, finally pulling inferiorly as the knot is tied. He repeats this process, so that there are two sutures anchoring the cartilage graft along the alar rim. These sutures are left in place for 2 weeks to allow some fibrosis to occur.
"The reason I like this technique is because it pulls the graft inferiorly so that it's perfectly flush against the alar rim remnant," he said. It also leaves more of the wound base exposed so that any overlying skin graft will be well perfused.
This is not necessarily a new technique, Dr. Krishnan said, but he could find no published description of it. It doesn't necessarily give better results, but it's easier to execute than are traditional methods, he added.
His techniques for anchoring cartilage grafts along the alar rim also work well with flaps including nasolabial transposition flaps, bilobed flaps, and interpolated paranasal flaps, resulting in good contour and symmetry and very acceptable results, he believes.
He typically follows these patients for 6 months after surgery, and while it's possible that the cartilage graft could shift after 6 months, "I doubt this would be the case."
One small drawback is that epithelium can start growing over the anchoring sutures during the 2 weeks that they are left in place, making them difficult to remove.
"Some people worry about infection, but I've never seen an infection with this technique," he added. All patients in his practice that receive cartilage grafts get perioperative antibiotics.
The advantages outweigh any potential drawbacks of the technique, in his opinion. It's easy to perform, and results are at least as good as those with more difficult techniques. His method precisely places the cartilage "exactly where you want it" along the alar rim, and apposes the cartilage graft to the mucosal lining, he said. When used in conjunction with a full-thickness skin graft, it allows the skin graft to come into contact with as much of the base of the wound as possible.
"It is important to remember that this technique is applicable only to smaller alar defects," he said. "For larger alar defects, using a large cartilage graft secured in the traditional manner is the preferred approach."
Dr. Krishnan said he has no relevant financial disclosures.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE AMERICAN COLLEGE OF MOHS SURGERY
Childhood Cancer Survivors at Higher Risk for Melanoma
CHICAGO – Survivors of childhood cancer have a nearly threefold increased risk of melanoma, compared with the general population, according to an analysis of the Childhood Cancer Survivor Study.
Among 14,358 5-year survivors, the cumulative incidence of a subsequent melanoma was 0.52% at 35 years from their initial cancer. The standard incidence ratio for subsequent melanoma was 2.83 and excess absolute risk 0.11/1,000 person-years.
"Although the cumulative incidence is low, this is potentially a significant problem," lead author Dr. Alberto Pappo said at the annual meeting of the American Society of Clinical Oncology. "Our findings justify increased awareness and the need for improved adherence to recommended surveillance guidelines in childhood cancer survivors."
Dr. Pappo said the analysis was sparked by a report last year from the Childhood Cancer Survivor Study showing that 5-year survivors had an increased cumulative incidence of subsequent neoplasms (20.5%) as well as second malignant neoplasms (8%) (J. Natl. Cancer Inst. 2010 Jul 21;102:1083-95).
The 14,358 patients in the current analysis were diagnosed with childhood cancer between 1970 and 1986, and compared with participants in the Surveillance, Epidemiology, and End Results (SEER) database. Median follow-up was 24 years.
In all, 53 survivors developed 59 melanomas, said Dr. Pappo, director of the solid tumor division at St. Jude Children’s Research Hospital, Memphis. Among them, two patients had ocular melanoma, nine had in situ disease, and two had other secondary malignancies before the diagnosis of melanoma. Five patients developed two or more melanomas, he pointed out.
The median age at the time of melanoma diagnosis was 32 years, and median time for tumor development was 20.7 years. At the time of the analysis, 43 of the 53 patients (81%) were still alive. Four patients died of melanoma.
Among the 53 melanoma patients, primary diagnoses included acute lymphoblastic leukemia (15), lymphoma (14), bone/soft tissue (15), brain (5), Wilms' tumor (3), and neuroblastoma (1).
The cumulative incidence of melanoma ranged from 0.29% for a primary diagnosis of brain cancer to 0.87% for survivors of soft tissue and bone sarcoma. The incidence was 0.43% for childhood leukemia and 0.55% for lymphoma.
Survivors who developed melanoma were significantly more likely to be more than 10 years old at the time of their initial cancer diagnosis (P less than .001) and to have a family history of cancer (P = .01). However, univariate analysis by age at diagnosis, sex, family history of cancer, treatment era, race, and alkylating score identified no significant risk factors, he said.
Attendees asked whether there is a cut point at which clinicians should pay particular attention to melanoma in this population. Dr. Pappo replied, "I am not sure if annual skin examinations are recommended for all childhood cancer survivors or just for those who receive radiotherapy, but perhaps for specific subsets, such as those with ALL or soft tissue sarcomas, that may be something worth implementing."
Dr. Pappo pointed out that the researchers are seeking more detailed tumor location and radiotherapy exposure data and that the study lacked data on factors known to contribute to melanoma such as sun exposure, number of nevi, and modifying genes.
Invited discussant Dr. Louise C. Strong, a professor of genetics at the University of Texas M.D. Anderson Cancer Center, Houston, called for further studies to elucidate the risk factors and genetics responsible for the increased incidence of melanoma observed in the study. She went on to say that studies in other populations at higher-risk for melanoma, such as transplant and immunosuppressed patients, have shown that prognosis for early stage I and II melanoma is the same as the general population but may actually be worse for late-stage disease.
"To me this is mostly a good news message because as long as we can educate our patients and can develop prevention and intervention programs, then it should work," she said.
The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
CHICAGO – Survivors of childhood cancer have a nearly threefold increased risk of melanoma, compared with the general population, according to an analysis of the Childhood Cancer Survivor Study.
Among 14,358 5-year survivors, the cumulative incidence of a subsequent melanoma was 0.52% at 35 years from their initial cancer. The standard incidence ratio for subsequent melanoma was 2.83 and excess absolute risk 0.11/1,000 person-years.
"Although the cumulative incidence is low, this is potentially a significant problem," lead author Dr. Alberto Pappo said at the annual meeting of the American Society of Clinical Oncology. "Our findings justify increased awareness and the need for improved adherence to recommended surveillance guidelines in childhood cancer survivors."
Dr. Pappo said the analysis was sparked by a report last year from the Childhood Cancer Survivor Study showing that 5-year survivors had an increased cumulative incidence of subsequent neoplasms (20.5%) as well as second malignant neoplasms (8%) (J. Natl. Cancer Inst. 2010 Jul 21;102:1083-95).
The 14,358 patients in the current analysis were diagnosed with childhood cancer between 1970 and 1986, and compared with participants in the Surveillance, Epidemiology, and End Results (SEER) database. Median follow-up was 24 years.
In all, 53 survivors developed 59 melanomas, said Dr. Pappo, director of the solid tumor division at St. Jude Children’s Research Hospital, Memphis. Among them, two patients had ocular melanoma, nine had in situ disease, and two had other secondary malignancies before the diagnosis of melanoma. Five patients developed two or more melanomas, he pointed out.
The median age at the time of melanoma diagnosis was 32 years, and median time for tumor development was 20.7 years. At the time of the analysis, 43 of the 53 patients (81%) were still alive. Four patients died of melanoma.
Among the 53 melanoma patients, primary diagnoses included acute lymphoblastic leukemia (15), lymphoma (14), bone/soft tissue (15), brain (5), Wilms' tumor (3), and neuroblastoma (1).
The cumulative incidence of melanoma ranged from 0.29% for a primary diagnosis of brain cancer to 0.87% for survivors of soft tissue and bone sarcoma. The incidence was 0.43% for childhood leukemia and 0.55% for lymphoma.
Survivors who developed melanoma were significantly more likely to be more than 10 years old at the time of their initial cancer diagnosis (P less than .001) and to have a family history of cancer (P = .01). However, univariate analysis by age at diagnosis, sex, family history of cancer, treatment era, race, and alkylating score identified no significant risk factors, he said.
Attendees asked whether there is a cut point at which clinicians should pay particular attention to melanoma in this population. Dr. Pappo replied, "I am not sure if annual skin examinations are recommended for all childhood cancer survivors or just for those who receive radiotherapy, but perhaps for specific subsets, such as those with ALL or soft tissue sarcomas, that may be something worth implementing."
Dr. Pappo pointed out that the researchers are seeking more detailed tumor location and radiotherapy exposure data and that the study lacked data on factors known to contribute to melanoma such as sun exposure, number of nevi, and modifying genes.
Invited discussant Dr. Louise C. Strong, a professor of genetics at the University of Texas M.D. Anderson Cancer Center, Houston, called for further studies to elucidate the risk factors and genetics responsible for the increased incidence of melanoma observed in the study. She went on to say that studies in other populations at higher-risk for melanoma, such as transplant and immunosuppressed patients, have shown that prognosis for early stage I and II melanoma is the same as the general population but may actually be worse for late-stage disease.
"To me this is mostly a good news message because as long as we can educate our patients and can develop prevention and intervention programs, then it should work," she said.
The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
CHICAGO – Survivors of childhood cancer have a nearly threefold increased risk of melanoma, compared with the general population, according to an analysis of the Childhood Cancer Survivor Study.
Among 14,358 5-year survivors, the cumulative incidence of a subsequent melanoma was 0.52% at 35 years from their initial cancer. The standard incidence ratio for subsequent melanoma was 2.83 and excess absolute risk 0.11/1,000 person-years.
"Although the cumulative incidence is low, this is potentially a significant problem," lead author Dr. Alberto Pappo said at the annual meeting of the American Society of Clinical Oncology. "Our findings justify increased awareness and the need for improved adherence to recommended surveillance guidelines in childhood cancer survivors."
Dr. Pappo said the analysis was sparked by a report last year from the Childhood Cancer Survivor Study showing that 5-year survivors had an increased cumulative incidence of subsequent neoplasms (20.5%) as well as second malignant neoplasms (8%) (J. Natl. Cancer Inst. 2010 Jul 21;102:1083-95).
The 14,358 patients in the current analysis were diagnosed with childhood cancer between 1970 and 1986, and compared with participants in the Surveillance, Epidemiology, and End Results (SEER) database. Median follow-up was 24 years.
In all, 53 survivors developed 59 melanomas, said Dr. Pappo, director of the solid tumor division at St. Jude Children’s Research Hospital, Memphis. Among them, two patients had ocular melanoma, nine had in situ disease, and two had other secondary malignancies before the diagnosis of melanoma. Five patients developed two or more melanomas, he pointed out.
The median age at the time of melanoma diagnosis was 32 years, and median time for tumor development was 20.7 years. At the time of the analysis, 43 of the 53 patients (81%) were still alive. Four patients died of melanoma.
Among the 53 melanoma patients, primary diagnoses included acute lymphoblastic leukemia (15), lymphoma (14), bone/soft tissue (15), brain (5), Wilms' tumor (3), and neuroblastoma (1).
The cumulative incidence of melanoma ranged from 0.29% for a primary diagnosis of brain cancer to 0.87% for survivors of soft tissue and bone sarcoma. The incidence was 0.43% for childhood leukemia and 0.55% for lymphoma.
Survivors who developed melanoma were significantly more likely to be more than 10 years old at the time of their initial cancer diagnosis (P less than .001) and to have a family history of cancer (P = .01). However, univariate analysis by age at diagnosis, sex, family history of cancer, treatment era, race, and alkylating score identified no significant risk factors, he said.
Attendees asked whether there is a cut point at which clinicians should pay particular attention to melanoma in this population. Dr. Pappo replied, "I am not sure if annual skin examinations are recommended for all childhood cancer survivors or just for those who receive radiotherapy, but perhaps for specific subsets, such as those with ALL or soft tissue sarcomas, that may be something worth implementing."
Dr. Pappo pointed out that the researchers are seeking more detailed tumor location and radiotherapy exposure data and that the study lacked data on factors known to contribute to melanoma such as sun exposure, number of nevi, and modifying genes.
Invited discussant Dr. Louise C. Strong, a professor of genetics at the University of Texas M.D. Anderson Cancer Center, Houston, called for further studies to elucidate the risk factors and genetics responsible for the increased incidence of melanoma observed in the study. She went on to say that studies in other populations at higher-risk for melanoma, such as transplant and immunosuppressed patients, have shown that prognosis for early stage I and II melanoma is the same as the general population but may actually be worse for late-stage disease.
"To me this is mostly a good news message because as long as we can educate our patients and can develop prevention and intervention programs, then it should work," she said.
The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF CLINICAL ONCOLOGY
Major Finding: The cumulative incidence of a first subsequent melanoma was 0.52% at 35 years from initial cancer.
Data Source: Retrospective analysis of 14,358 5-year childhood cancer survivors.
Disclosures: The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
Commentary: Prepare for Reimbursement Turbulence
Increases in the volume of dermatologic procedures over the past 15 years are the result of dermatology's reinvention of itself as a surgical specialty, as well as a result of what is a very real skin cancer epidemic.
But that, coupled with the disproportionate percentage of the total reimbursed relative value unit (RVU) pool going to dermatologists, has raised red flags for those charged with cutting costs, and has left the specialty particularly vulnerable to cuts in reimbursement.
Between 1995 and 2008, skin biopsies increased by 76%, destructions by 64%, excisions by 17%, actinic keratoses (AKs) of 15 or greater by 17%, and use of pathology code 88305 by 81%. Even more striking - and potentially at the expense of cuts of up to 25%-30% in reimbursement for these procedures - Mohs surgery is up about 400%, according to the most recent data.
The powers that be aren't looking too favorably upon these increases, especially considering that from 1992 to 2002, the total reimbursed RVU pool percentage for dermatologists increased from 2.5% to 2.9%, while dermatologists accounted for only 1% of all physicians.
This increase is appropriate, given the overhead required for the surgical components of dermatology practice and the increased demand, but it has led to a view of dermatologists as greedy and overpaid.
I don't think we're the greedy dermatologists that many make us out to be; we're office-based, frontline specialists responding to an unrecognized epidemic. But those charged with cutting costs don't see it that way, because they either don't understand or refuse to acknowledge that there is a very real skin cancer epidemic in this country.
A review of multiple Medicare and Ambulatory Medical Care Service databases showed that the number of procedures for skin cancer increased 77% from nearly 1.2 million in 1992 to nearly 2.1 million in 2006. The most recent estimate is that the total number of nonmelanoma skin cancers in the U.S. population is about 4 million.
There are a number of misconceptions about dermatology that are contributing to the specialty's being under siege, and some of them are from dermatologists themselves.
Among the fundamental misconceptions by MedPAC, Congress, and the Centers for Medicare and Medicaid Services are the following:
- What dermatologists do is not important and is mostly cosmetic.
- The increase in RVUs comes from waste and abuse, and from unimportant minor-procedure codes that pay too much.
- There is no skin cancer epidemic.
- New money is not needed in the payment pool for health care.
Dermatologists themselves are also guilty of misconceptions about the specialty. For example, there is a misconception among dermatologists that training more dermatologists and physician extenders will benefit dermatology, when in actuality they will increase utilization and trigger more scrutiny and punishment in the form of cuts to reimbursement.
Similarly, promoting dermatology services to the public won't help until the utilization constraints on the payer side have been solved.
In the next 5 years, it is likely that physicians - and particularly specialists - will take the brunt of any cost-savings attempts by Congress. Dermatology, in particular, will be targeted for cuts because of increased utilization.
I also predict that in the next 5 years the skin cancer epidemic will continue as baby boomers age; that Mohs surgery will not regain its multiple surgery reduction exemption; that appropriateness criteria for Mohs, AK destruction, and perhaps even shaves, skin biopsies, and pathology will emerge; that existing ambulatory surgery centers will become more valuable because new centers will become difficult to license; that Mohs surgeons will band together in groups; and that cosmetic procedures will continue to increase as baby boomers age.
In the face of so many misconceptions and some dire predictions about the future of the specialty, it is imperative that dermatologists work together to educate Congress, MedPAC, and the CMS about the skin cancer epidemic, and prove that dermatologists are the solution, not the problem.
We have to show them that as their constituents live longer, they get more diseases per unit of time, and that we aren't making this up to make more money.
We also have to convince them that epidemics require new money to allow them to be addressed adequately.
This will require a coordinated agenda at the highest levels. Quality must be defined and measured on our terms, and utilization must be controlled or we will risk losing our specialty. We must define our peer groups within dermatology, and a list must be developed of acceptable and unacceptable nominees to the Independent Payment Advisory Board - the panel appointed by Congress to determine payment structure, which is perhaps the biggest threat to the specialty.
What else can you do? Join the American Medical Association (AMA), not because of their politics but because our representation is dependent on your membership. Also, donate to the SkinPAC. This is critical to get politicians elected who understand us. And elect strong, decisive people - not necessarily the nicest people - to your leadership and board of directors because there are some challenges and tough decisions ahead.
Dr. Coldiron is president of the American College of Mohs Surgery and a clinical assistant professor of dermatology at the University of Cincinnati. He has a private practice in Cincinnati.
Increases in the volume of dermatologic procedures over the past 15 years are the result of dermatology's reinvention of itself as a surgical specialty, as well as a result of what is a very real skin cancer epidemic.
But that, coupled with the disproportionate percentage of the total reimbursed relative value unit (RVU) pool going to dermatologists, has raised red flags for those charged with cutting costs, and has left the specialty particularly vulnerable to cuts in reimbursement.
Between 1995 and 2008, skin biopsies increased by 76%, destructions by 64%, excisions by 17%, actinic keratoses (AKs) of 15 or greater by 17%, and use of pathology code 88305 by 81%. Even more striking - and potentially at the expense of cuts of up to 25%-30% in reimbursement for these procedures - Mohs surgery is up about 400%, according to the most recent data.
The powers that be aren't looking too favorably upon these increases, especially considering that from 1992 to 2002, the total reimbursed RVU pool percentage for dermatologists increased from 2.5% to 2.9%, while dermatologists accounted for only 1% of all physicians.
This increase is appropriate, given the overhead required for the surgical components of dermatology practice and the increased demand, but it has led to a view of dermatologists as greedy and overpaid.
I don't think we're the greedy dermatologists that many make us out to be; we're office-based, frontline specialists responding to an unrecognized epidemic. But those charged with cutting costs don't see it that way, because they either don't understand or refuse to acknowledge that there is a very real skin cancer epidemic in this country.
A review of multiple Medicare and Ambulatory Medical Care Service databases showed that the number of procedures for skin cancer increased 77% from nearly 1.2 million in 1992 to nearly 2.1 million in 2006. The most recent estimate is that the total number of nonmelanoma skin cancers in the U.S. population is about 4 million.
There are a number of misconceptions about dermatology that are contributing to the specialty's being under siege, and some of them are from dermatologists themselves.
Among the fundamental misconceptions by MedPAC, Congress, and the Centers for Medicare and Medicaid Services are the following:
- What dermatologists do is not important and is mostly cosmetic.
- The increase in RVUs comes from waste and abuse, and from unimportant minor-procedure codes that pay too much.
- There is no skin cancer epidemic.
- New money is not needed in the payment pool for health care.
Dermatologists themselves are also guilty of misconceptions about the specialty. For example, there is a misconception among dermatologists that training more dermatologists and physician extenders will benefit dermatology, when in actuality they will increase utilization and trigger more scrutiny and punishment in the form of cuts to reimbursement.
Similarly, promoting dermatology services to the public won't help until the utilization constraints on the payer side have been solved.
In the next 5 years, it is likely that physicians - and particularly specialists - will take the brunt of any cost-savings attempts by Congress. Dermatology, in particular, will be targeted for cuts because of increased utilization.
I also predict that in the next 5 years the skin cancer epidemic will continue as baby boomers age; that Mohs surgery will not regain its multiple surgery reduction exemption; that appropriateness criteria for Mohs, AK destruction, and perhaps even shaves, skin biopsies, and pathology will emerge; that existing ambulatory surgery centers will become more valuable because new centers will become difficult to license; that Mohs surgeons will band together in groups; and that cosmetic procedures will continue to increase as baby boomers age.
In the face of so many misconceptions and some dire predictions about the future of the specialty, it is imperative that dermatologists work together to educate Congress, MedPAC, and the CMS about the skin cancer epidemic, and prove that dermatologists are the solution, not the problem.
We have to show them that as their constituents live longer, they get more diseases per unit of time, and that we aren't making this up to make more money.
We also have to convince them that epidemics require new money to allow them to be addressed adequately.
This will require a coordinated agenda at the highest levels. Quality must be defined and measured on our terms, and utilization must be controlled or we will risk losing our specialty. We must define our peer groups within dermatology, and a list must be developed of acceptable and unacceptable nominees to the Independent Payment Advisory Board - the panel appointed by Congress to determine payment structure, which is perhaps the biggest threat to the specialty.
What else can you do? Join the American Medical Association (AMA), not because of their politics but because our representation is dependent on your membership. Also, donate to the SkinPAC. This is critical to get politicians elected who understand us. And elect strong, decisive people - not necessarily the nicest people - to your leadership and board of directors because there are some challenges and tough decisions ahead.
Dr. Coldiron is president of the American College of Mohs Surgery and a clinical assistant professor of dermatology at the University of Cincinnati. He has a private practice in Cincinnati.
Increases in the volume of dermatologic procedures over the past 15 years are the result of dermatology's reinvention of itself as a surgical specialty, as well as a result of what is a very real skin cancer epidemic.
But that, coupled with the disproportionate percentage of the total reimbursed relative value unit (RVU) pool going to dermatologists, has raised red flags for those charged with cutting costs, and has left the specialty particularly vulnerable to cuts in reimbursement.
Between 1995 and 2008, skin biopsies increased by 76%, destructions by 64%, excisions by 17%, actinic keratoses (AKs) of 15 or greater by 17%, and use of pathology code 88305 by 81%. Even more striking - and potentially at the expense of cuts of up to 25%-30% in reimbursement for these procedures - Mohs surgery is up about 400%, according to the most recent data.
The powers that be aren't looking too favorably upon these increases, especially considering that from 1992 to 2002, the total reimbursed RVU pool percentage for dermatologists increased from 2.5% to 2.9%, while dermatologists accounted for only 1% of all physicians.
This increase is appropriate, given the overhead required for the surgical components of dermatology practice and the increased demand, but it has led to a view of dermatologists as greedy and overpaid.
I don't think we're the greedy dermatologists that many make us out to be; we're office-based, frontline specialists responding to an unrecognized epidemic. But those charged with cutting costs don't see it that way, because they either don't understand or refuse to acknowledge that there is a very real skin cancer epidemic in this country.
A review of multiple Medicare and Ambulatory Medical Care Service databases showed that the number of procedures for skin cancer increased 77% from nearly 1.2 million in 1992 to nearly 2.1 million in 2006. The most recent estimate is that the total number of nonmelanoma skin cancers in the U.S. population is about 4 million.
There are a number of misconceptions about dermatology that are contributing to the specialty's being under siege, and some of them are from dermatologists themselves.
Among the fundamental misconceptions by MedPAC, Congress, and the Centers for Medicare and Medicaid Services are the following:
- What dermatologists do is not important and is mostly cosmetic.
- The increase in RVUs comes from waste and abuse, and from unimportant minor-procedure codes that pay too much.
- There is no skin cancer epidemic.
- New money is not needed in the payment pool for health care.
Dermatologists themselves are also guilty of misconceptions about the specialty. For example, there is a misconception among dermatologists that training more dermatologists and physician extenders will benefit dermatology, when in actuality they will increase utilization and trigger more scrutiny and punishment in the form of cuts to reimbursement.
Similarly, promoting dermatology services to the public won't help until the utilization constraints on the payer side have been solved.
In the next 5 years, it is likely that physicians - and particularly specialists - will take the brunt of any cost-savings attempts by Congress. Dermatology, in particular, will be targeted for cuts because of increased utilization.
I also predict that in the next 5 years the skin cancer epidemic will continue as baby boomers age; that Mohs surgery will not regain its multiple surgery reduction exemption; that appropriateness criteria for Mohs, AK destruction, and perhaps even shaves, skin biopsies, and pathology will emerge; that existing ambulatory surgery centers will become more valuable because new centers will become difficult to license; that Mohs surgeons will band together in groups; and that cosmetic procedures will continue to increase as baby boomers age.
In the face of so many misconceptions and some dire predictions about the future of the specialty, it is imperative that dermatologists work together to educate Congress, MedPAC, and the CMS about the skin cancer epidemic, and prove that dermatologists are the solution, not the problem.
We have to show them that as their constituents live longer, they get more diseases per unit of time, and that we aren't making this up to make more money.
We also have to convince them that epidemics require new money to allow them to be addressed adequately.
This will require a coordinated agenda at the highest levels. Quality must be defined and measured on our terms, and utilization must be controlled or we will risk losing our specialty. We must define our peer groups within dermatology, and a list must be developed of acceptable and unacceptable nominees to the Independent Payment Advisory Board - the panel appointed by Congress to determine payment structure, which is perhaps the biggest threat to the specialty.
What else can you do? Join the American Medical Association (AMA), not because of their politics but because our representation is dependent on your membership. Also, donate to the SkinPAC. This is critical to get politicians elected who understand us. And elect strong, decisive people - not necessarily the nicest people - to your leadership and board of directors because there are some challenges and tough decisions ahead.
Dr. Coldiron is president of the American College of Mohs Surgery and a clinical assistant professor of dermatology at the University of Cincinnati. He has a private practice in Cincinnati.
ASCO: Melanoma Patients Respond to Combo BRAF, MEK Inhibitors
CHICAGO – The combination of two oral drugs that target selected molecules in the mitogen-activated protein kinase signaling pathway appears to be safe and active in patients with melanoma, a phase I trial found. The finding was reported at the annual meeting of the American Society of Clinical Oncology.
Among patients with BRAF-mutated melanoma who had not previously received an agent targeting that molecule, the combination of an investigational BRAF inhibitor (GSK436) and an investigational MEK inhibitor (GSK212) achieved a response rate of 50%-77%.
Both drugs could be used in their full monotherapy doses when combined. And the combination was well tolerated; indeed, certain adverse effects that were seen with each drug when used alone (such as a rash with MEK inhibitor monotherapy) were less common when they were used together.
"The clinical activity in BRAF V600–mutant melanoma is definitely compelling," commented lead investigator Dr. Jeffrey R. Infante, a medical oncologist and director of drug development at the Sarah Cannon Research Institute in Nashville, Tenn. "But like everyone in the audience, I think, we are anxiously awaiting the long-term durability" data. "This combination could have potential implications in multiple tumor types outside of melanoma and have broader impacts in oncology," he added.
"I am really impressed by these data," said discussant Dr. Reinhard Dummer of the University Hospital Zurich. In particular, although the trial was not designed to assess responses and the follow-up is still short, some of the patients had complete remissions, he noted.
"I have to admit, I never thought that we could have complete remissions with kinase inhibitors, so I hope that they will continue," he said. "With this in mind, I think we can reset our goals for melanoma research: We don't want to go for progression-free survival in the future, but we should go for cure."
The reduced rate of the inflammatory rash with the combination was "very surprising," given that the rash is likely a stress response resulting from the MEK inhibitor's effect on the MAP [mitogen-activated protein] kinase pathway in the skin, Dr. Dummer said. "How can it happen that this rash is reduced in the combination with the BRAF inhibitor? This is completely not understandable for me. Cutaneous biologists will have a lot of fun to study this model."
Both drugs, which are manufactured by GlaxoSmithKline, have previously shown activity as single agents in patients who have melanoma with BRAF V600 gene mutations, which are found in about half of cases. "The goal was to ... give [the drugs] together and really hope to get a profound synergy in combination in pathway inhibition," explained Dr. Infante.
Patients were eligible for the trial if they had melanoma or another cancer with a V600 mutation in the BRAF gene. Those who had stable brain metastases after surgery or radiosurgery could enroll.
Patients having certain eye conditions were excluded because of previously identified, related issues associated with MEK inhibitor therapy. But there was no limit on the number of prior therapies.
In part A of the study, the investigators tested for drug-drug interactions. In part B, they performed dose escalation, assessed safety and activity, and enrolled expansion cohorts of patients, such as those who had previously received a BRAF inhibitor.
Among the eight patients in part A, there was no effect of the MEK inhibitor when given daily on the pharmacokinetics of single doses of the BRAF inhibitor, Dr. Infante reported.
Of the 109 patients in part B, fully 93% had melanoma and 91% specifically had the V600E BRAF mutation. The majority (54%) had received at least two prior lines of systemic anticancer therapy.
Dose-escalation results showed that both drugs could be given in combination at their full monotherapy doses (150 mg twice daily of the BRAF inhibitor and 2 mg once daily of the MEK inhibitor), largely without dose-limiting toxicities.
Only one dose-limiting toxicity was seen with the full dose of each drug. The patient developed a neutrophilic panniculitis, manifesting as painful red nodules on the thorax and extremities, in association with fever and chills. It resolved with discontinuation of the drugs and steroid therapy.
Across the dose combinations, 79% of patients experienced an adverse event and 19% experienced a grade 3 or higher adverse event, most often neutropenia, leukopenia, diarrhea, and fever. There was no clear association with doses, according to Dr. Infante.
In a finding as yet without explanation, certain adverse events that had been seen previously with each drug when they were given alone were actually less common when they were combined. Specifically, rash occurred in 25% of patients, whereas historically it has been seen in 75%-80% of those given the MEK inhibitor alone. And squamous cell carcinoma and other hyperproliferative skin lesions were seen in roughly 1%, whereas they have been seen in 7%-15% of those given the BRAF inhibitor alone.
Among the 71 patients with melanoma who had not previously received a BRAF inhibitor, the rate of unconfirmed objective response (complete or partial response) ranged from 50% to 77%, depending on the dose combination. Five patients had complete responses. For comparison, the response rate in BRAF-mutated melanoma has been 63% with the BRAF inhibitor alone and 40% with the MEK inhibitor alone.
Additionally, the rate of disease control (complete response, partial response, or stable disease) ranged from 95% to 100%, Dr. Infante reported. And the large majority of patients (83%) are still receiving combination therapy.
Analyses among the 24 patients with melanoma who had previously received a BRAF inhibitor showed that about half had a reduction in tumor size. There was no clear difference according to the duration of the earlier BRAF inhibitor therapy.
The investigators are now conducting a phase II trial in which 150 patients with V600E- or V600K-mutant melanoma are being randomly assigned to therapy with the BRAF inhibitor alone or in combination with the MEK inhibitor at two dose levels.
Dr. Dummer, the discussant, noted that obtaining biopsies is "very crucial" to understanding how such novel antimelanoma therapies are working. "Our patients, when you explain this to them, they will agree to sequential biopsies," he commented. "And I encourage all of you to participate in trials with a very strong translational research" component.
Dr. Infante reported that he is an unpaid consultant to GlaxoSmithKline, and that several coauthors are GSK employees. Dr. Dummer reported that he is a consultant to Bristol-Myers Squibb, Merck, Novartis, and Roche, and receives honoraria from Bristol-Myers Squibb.
CHICAGO – The combination of two oral drugs that target selected molecules in the mitogen-activated protein kinase signaling pathway appears to be safe and active in patients with melanoma, a phase I trial found. The finding was reported at the annual meeting of the American Society of Clinical Oncology.
Among patients with BRAF-mutated melanoma who had not previously received an agent targeting that molecule, the combination of an investigational BRAF inhibitor (GSK436) and an investigational MEK inhibitor (GSK212) achieved a response rate of 50%-77%.
Both drugs could be used in their full monotherapy doses when combined. And the combination was well tolerated; indeed, certain adverse effects that were seen with each drug when used alone (such as a rash with MEK inhibitor monotherapy) were less common when they were used together.
"The clinical activity in BRAF V600–mutant melanoma is definitely compelling," commented lead investigator Dr. Jeffrey R. Infante, a medical oncologist and director of drug development at the Sarah Cannon Research Institute in Nashville, Tenn. "But like everyone in the audience, I think, we are anxiously awaiting the long-term durability" data. "This combination could have potential implications in multiple tumor types outside of melanoma and have broader impacts in oncology," he added.
"I am really impressed by these data," said discussant Dr. Reinhard Dummer of the University Hospital Zurich. In particular, although the trial was not designed to assess responses and the follow-up is still short, some of the patients had complete remissions, he noted.
"I have to admit, I never thought that we could have complete remissions with kinase inhibitors, so I hope that they will continue," he said. "With this in mind, I think we can reset our goals for melanoma research: We don't want to go for progression-free survival in the future, but we should go for cure."
The reduced rate of the inflammatory rash with the combination was "very surprising," given that the rash is likely a stress response resulting from the MEK inhibitor's effect on the MAP [mitogen-activated protein] kinase pathway in the skin, Dr. Dummer said. "How can it happen that this rash is reduced in the combination with the BRAF inhibitor? This is completely not understandable for me. Cutaneous biologists will have a lot of fun to study this model."
Both drugs, which are manufactured by GlaxoSmithKline, have previously shown activity as single agents in patients who have melanoma with BRAF V600 gene mutations, which are found in about half of cases. "The goal was to ... give [the drugs] together and really hope to get a profound synergy in combination in pathway inhibition," explained Dr. Infante.
Patients were eligible for the trial if they had melanoma or another cancer with a V600 mutation in the BRAF gene. Those who had stable brain metastases after surgery or radiosurgery could enroll.
Patients having certain eye conditions were excluded because of previously identified, related issues associated with MEK inhibitor therapy. But there was no limit on the number of prior therapies.
In part A of the study, the investigators tested for drug-drug interactions. In part B, they performed dose escalation, assessed safety and activity, and enrolled expansion cohorts of patients, such as those who had previously received a BRAF inhibitor.
Among the eight patients in part A, there was no effect of the MEK inhibitor when given daily on the pharmacokinetics of single doses of the BRAF inhibitor, Dr. Infante reported.
Of the 109 patients in part B, fully 93% had melanoma and 91% specifically had the V600E BRAF mutation. The majority (54%) had received at least two prior lines of systemic anticancer therapy.
Dose-escalation results showed that both drugs could be given in combination at their full monotherapy doses (150 mg twice daily of the BRAF inhibitor and 2 mg once daily of the MEK inhibitor), largely without dose-limiting toxicities.
Only one dose-limiting toxicity was seen with the full dose of each drug. The patient developed a neutrophilic panniculitis, manifesting as painful red nodules on the thorax and extremities, in association with fever and chills. It resolved with discontinuation of the drugs and steroid therapy.
Across the dose combinations, 79% of patients experienced an adverse event and 19% experienced a grade 3 or higher adverse event, most often neutropenia, leukopenia, diarrhea, and fever. There was no clear association with doses, according to Dr. Infante.
In a finding as yet without explanation, certain adverse events that had been seen previously with each drug when they were given alone were actually less common when they were combined. Specifically, rash occurred in 25% of patients, whereas historically it has been seen in 75%-80% of those given the MEK inhibitor alone. And squamous cell carcinoma and other hyperproliferative skin lesions were seen in roughly 1%, whereas they have been seen in 7%-15% of those given the BRAF inhibitor alone.
Among the 71 patients with melanoma who had not previously received a BRAF inhibitor, the rate of unconfirmed objective response (complete or partial response) ranged from 50% to 77%, depending on the dose combination. Five patients had complete responses. For comparison, the response rate in BRAF-mutated melanoma has been 63% with the BRAF inhibitor alone and 40% with the MEK inhibitor alone.
Additionally, the rate of disease control (complete response, partial response, or stable disease) ranged from 95% to 100%, Dr. Infante reported. And the large majority of patients (83%) are still receiving combination therapy.
Analyses among the 24 patients with melanoma who had previously received a BRAF inhibitor showed that about half had a reduction in tumor size. There was no clear difference according to the duration of the earlier BRAF inhibitor therapy.
The investigators are now conducting a phase II trial in which 150 patients with V600E- or V600K-mutant melanoma are being randomly assigned to therapy with the BRAF inhibitor alone or in combination with the MEK inhibitor at two dose levels.
Dr. Dummer, the discussant, noted that obtaining biopsies is "very crucial" to understanding how such novel antimelanoma therapies are working. "Our patients, when you explain this to them, they will agree to sequential biopsies," he commented. "And I encourage all of you to participate in trials with a very strong translational research" component.
Dr. Infante reported that he is an unpaid consultant to GlaxoSmithKline, and that several coauthors are GSK employees. Dr. Dummer reported that he is a consultant to Bristol-Myers Squibb, Merck, Novartis, and Roche, and receives honoraria from Bristol-Myers Squibb.
CHICAGO – The combination of two oral drugs that target selected molecules in the mitogen-activated protein kinase signaling pathway appears to be safe and active in patients with melanoma, a phase I trial found. The finding was reported at the annual meeting of the American Society of Clinical Oncology.
Among patients with BRAF-mutated melanoma who had not previously received an agent targeting that molecule, the combination of an investigational BRAF inhibitor (GSK436) and an investigational MEK inhibitor (GSK212) achieved a response rate of 50%-77%.
Both drugs could be used in their full monotherapy doses when combined. And the combination was well tolerated; indeed, certain adverse effects that were seen with each drug when used alone (such as a rash with MEK inhibitor monotherapy) were less common when they were used together.
"The clinical activity in BRAF V600–mutant melanoma is definitely compelling," commented lead investigator Dr. Jeffrey R. Infante, a medical oncologist and director of drug development at the Sarah Cannon Research Institute in Nashville, Tenn. "But like everyone in the audience, I think, we are anxiously awaiting the long-term durability" data. "This combination could have potential implications in multiple tumor types outside of melanoma and have broader impacts in oncology," he added.
"I am really impressed by these data," said discussant Dr. Reinhard Dummer of the University Hospital Zurich. In particular, although the trial was not designed to assess responses and the follow-up is still short, some of the patients had complete remissions, he noted.
"I have to admit, I never thought that we could have complete remissions with kinase inhibitors, so I hope that they will continue," he said. "With this in mind, I think we can reset our goals for melanoma research: We don't want to go for progression-free survival in the future, but we should go for cure."
The reduced rate of the inflammatory rash with the combination was "very surprising," given that the rash is likely a stress response resulting from the MEK inhibitor's effect on the MAP [mitogen-activated protein] kinase pathway in the skin, Dr. Dummer said. "How can it happen that this rash is reduced in the combination with the BRAF inhibitor? This is completely not understandable for me. Cutaneous biologists will have a lot of fun to study this model."
Both drugs, which are manufactured by GlaxoSmithKline, have previously shown activity as single agents in patients who have melanoma with BRAF V600 gene mutations, which are found in about half of cases. "The goal was to ... give [the drugs] together and really hope to get a profound synergy in combination in pathway inhibition," explained Dr. Infante.
Patients were eligible for the trial if they had melanoma or another cancer with a V600 mutation in the BRAF gene. Those who had stable brain metastases after surgery or radiosurgery could enroll.
Patients having certain eye conditions were excluded because of previously identified, related issues associated with MEK inhibitor therapy. But there was no limit on the number of prior therapies.
In part A of the study, the investigators tested for drug-drug interactions. In part B, they performed dose escalation, assessed safety and activity, and enrolled expansion cohorts of patients, such as those who had previously received a BRAF inhibitor.
Among the eight patients in part A, there was no effect of the MEK inhibitor when given daily on the pharmacokinetics of single doses of the BRAF inhibitor, Dr. Infante reported.
Of the 109 patients in part B, fully 93% had melanoma and 91% specifically had the V600E BRAF mutation. The majority (54%) had received at least two prior lines of systemic anticancer therapy.
Dose-escalation results showed that both drugs could be given in combination at their full monotherapy doses (150 mg twice daily of the BRAF inhibitor and 2 mg once daily of the MEK inhibitor), largely without dose-limiting toxicities.
Only one dose-limiting toxicity was seen with the full dose of each drug. The patient developed a neutrophilic panniculitis, manifesting as painful red nodules on the thorax and extremities, in association with fever and chills. It resolved with discontinuation of the drugs and steroid therapy.
Across the dose combinations, 79% of patients experienced an adverse event and 19% experienced a grade 3 or higher adverse event, most often neutropenia, leukopenia, diarrhea, and fever. There was no clear association with doses, according to Dr. Infante.
In a finding as yet without explanation, certain adverse events that had been seen previously with each drug when they were given alone were actually less common when they were combined. Specifically, rash occurred in 25% of patients, whereas historically it has been seen in 75%-80% of those given the MEK inhibitor alone. And squamous cell carcinoma and other hyperproliferative skin lesions were seen in roughly 1%, whereas they have been seen in 7%-15% of those given the BRAF inhibitor alone.
Among the 71 patients with melanoma who had not previously received a BRAF inhibitor, the rate of unconfirmed objective response (complete or partial response) ranged from 50% to 77%, depending on the dose combination. Five patients had complete responses. For comparison, the response rate in BRAF-mutated melanoma has been 63% with the BRAF inhibitor alone and 40% with the MEK inhibitor alone.
Additionally, the rate of disease control (complete response, partial response, or stable disease) ranged from 95% to 100%, Dr. Infante reported. And the large majority of patients (83%) are still receiving combination therapy.
Analyses among the 24 patients with melanoma who had previously received a BRAF inhibitor showed that about half had a reduction in tumor size. There was no clear difference according to the duration of the earlier BRAF inhibitor therapy.
The investigators are now conducting a phase II trial in which 150 patients with V600E- or V600K-mutant melanoma are being randomly assigned to therapy with the BRAF inhibitor alone or in combination with the MEK inhibitor at two dose levels.
Dr. Dummer, the discussant, noted that obtaining biopsies is "very crucial" to understanding how such novel antimelanoma therapies are working. "Our patients, when you explain this to them, they will agree to sequential biopsies," he commented. "And I encourage all of you to participate in trials with a very strong translational research" component.
Dr. Infante reported that he is an unpaid consultant to GlaxoSmithKline, and that several coauthors are GSK employees. Dr. Dummer reported that he is a consultant to Bristol-Myers Squibb, Merck, Novartis, and Roche, and receives honoraria from Bristol-Myers Squibb.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF CLINICAL ONCOLOGY
Major Finding: The objective response rate ranged from 50% to 77%, depending on doses, among patients who had not previously received any BRAF inhibitor therapy.
Data Source: A phase I trial of an oral MEK inhibitor plus an oral BRAF inhibitor that enrolled 101 patients with melanoma and the BRAF V600 mutation
Disclosures: Dr. Infante reported that he is an unpaid consultant to GlaxoSmithKline, and that several of his coauthors are GSK employees. Dr. Dummer reported that he is a consultant to Bristol-Myers Squibb, Merck, Novartis, and Roche, and receives honoraria from Bristol-Myers Squibb.
Melanoma Patients Respond to Combo of BRAF and MEK Inhibitors
CHICAGO – The combination of two oral drugs that target selected molecules in the mitogen-activated protein kinase signaling pathway appears to be safe and active in patients with melanoma, a phase I trial found. The finding was reported at the annual meeting of the American Society of Clinical Oncology.
Among patients with BRAF-mutated melanoma who had not previously received an agent targeting that molecule, the combination of an investigational BRAF inhibitor (GSK436) and an investigational MEK inhibitor (GSK212) achieved a response rate of 50%-77%.
Both drugs could be used in their full monotherapy doses when combined. And the combination was well tolerated; indeed, certain adverse effects that were seen with each drug when used alone (such as a rash with MEK inhibitor monotherapy) were less common when they were used together.
"The clinical activity in BRAF V600–mutant melanoma is definitely compelling," commented lead investigator Dr. Jeffrey R. Infante, a medical oncologist and director of drug development at the Sarah Cannon Research Institute in Nashville, Tenn. "But like everyone in the audience, I think, we are anxiously awaiting the long-term durability" data. "This combination could have potential implications in multiple tumor types outside of melanoma and have broader impacts in oncology," he added.
"I am really impressed by these data," said discussant Dr. Reinhard Dummer of the University Hospital Zurich. In particular, although the trial was not designed to assess responses and the follow-up is still short, some of the patients had complete remissions, he noted.
"I have to admit, I never thought that we could have complete remissions with kinase inhibitors, so I hope that they will continue," he said. "With this in mind, I think we can reset our goals for melanoma research: We don’t want to go for progression-free survival in the future, but we should go for cure."
The reduced rate of the inflammatory rash with the combination was "very surprising," given that the rash is likely a stress response resulting from the MEK inhibitor’s effect on the MAP [mitogen-activated protein] kinase pathway in the skin, Dr. Dummer said. "How can it happen that this rash is reduced in the combination with the BRAF inhibitor? This is completely not understandable for me. Cutaneous biologists will have a lot of fun to study this model."
Both drugs, which are manufactured by GlaxoSmithKline, have previously shown activity as single agents in patients who have melanoma with BRAF V600 gene mutations, which are found in about half of cases. "The goal was to ... give [the drugs] together and really hope to get a profound synergy in combination in pathway inhibition," explained Dr. Infante.
Patients were eligible for the trial if they had melanoma or another cancer with a V600 mutation in the BRAF gene. Those who had stable brain metastases after surgery or radiosurgery could enroll.
Patients having certain eye conditions were excluded because of previously identified, related issues associated with MEK inhibitor therapy. But there was no limit on the number of prior therapies.
In part A of the study, the investigators tested for drug-drug interactions. In part B, they performed dose escalation, assessed safety and activity, and enrolled expansion cohorts of patients, such as those who had previously received a BRAF inhibitor.
Among the eight patients in part A, there was no effect of the MEK inhibitor when given daily on the pharmacokinetics of single doses of the BRAF inhibitor, Dr. Infante reported.
Of the 109 patients in part B, fully 93% had melanoma and 91% specifically had the V600E BRAF mutation. The majority (54%) had received at least two prior lines of systemic anticancer therapy.
Dose-escalation results showed that both drugs could be given in combination at their full monotherapy doses (150 mg twice daily of the BRAF inhibitor and 2 mg once daily of the MEK inhibitor), largely without dose-limiting toxicities.
Only one dose-limiting toxicity was seen with the full dose of each drug. The patient developed a neutrophilic panniculitis, manifesting as painful red nodules on the thorax and extremities, in association with fever and chills. It resolved with discontinuation of the drugs and steroid therapy.
Across the dose combinations, 79% of patients experienced an adverse event and 19% experienced a grade 3 or higher adverse event, most often neutropenia, leukopenia, diarrhea, and fever. There was no clear association with doses, according to Dr. Infante.
In a finding as yet without explanation, certain adverse events that had been seen previously with each drug when they were given alone were actually less common when they were combined. Specifically, rash occurred in 25% of patients, whereas historically it has been seen in 75%-80% of those given the MEK inhibitor alone. And squamous cell carcinoma and other hyperproliferative skin lesions were seen in roughly 1%, whereas they have been seen in 7%-15% of those given the BRAF inhibitor alone.
Among the 71 patients with melanoma who had not previously received a BRAF inhibitor, the rate of unconfirmed objective response (complete or partial response) ranged from 50% to 77%, depending on the dose combination. Five patients had complete responses. For comparison, the response rate in BRAF-mutated melanoma has been 63% with the BRAF inhibitor alone and 40% with the MEK inhibitor alone.
Additionally, the rate of disease control (complete response, partial response, or stable disease) ranged from 95% to 100%, Dr. Infante reported. And the large majority of patients (83%) are still receiving combination therapy.
Analyses among the 24 patients with melanoma who had previously received a BRAF inhibitor showed that about half had a reduction in tumor size. There was no clear difference according to the duration of the earlier BRAF inhibitor therapy.
The investigators are now conducting a phase II trial in which 150 patients with V600E- or V600K-mutant melanoma are being randomly assigned to therapy with the BRAF inhibitor alone or in combination with the MEK inhibitor at two dose levels.
Dr. Dummer, the discussant, noted that obtaining biopsies is "very crucial" to understanding how such novel antimelanoma therapies are working. "Our patients, when you explain this to them, they will agree to sequential biopsies," he commented. "And I encourage all of you to participate in trials with a very strong translational research" component.
Dr. Infante reported that he is an unpaid consultant to GlaxoSmithKline, and that several coauthors are GSK employees. Dr. Dummer reported that he is a consultant to Bristol-Myers Squibb, Merck, Novartis, and Roche, and receives honoraria from Bristol-Myers Squibb.
CHICAGO – The combination of two oral drugs that target selected molecules in the mitogen-activated protein kinase signaling pathway appears to be safe and active in patients with melanoma, a phase I trial found. The finding was reported at the annual meeting of the American Society of Clinical Oncology.
Among patients with BRAF-mutated melanoma who had not previously received an agent targeting that molecule, the combination of an investigational BRAF inhibitor (GSK436) and an investigational MEK inhibitor (GSK212) achieved a response rate of 50%-77%.
Both drugs could be used in their full monotherapy doses when combined. And the combination was well tolerated; indeed, certain adverse effects that were seen with each drug when used alone (such as a rash with MEK inhibitor monotherapy) were less common when they were used together.
"The clinical activity in BRAF V600–mutant melanoma is definitely compelling," commented lead investigator Dr. Jeffrey R. Infante, a medical oncologist and director of drug development at the Sarah Cannon Research Institute in Nashville, Tenn. "But like everyone in the audience, I think, we are anxiously awaiting the long-term durability" data. "This combination could have potential implications in multiple tumor types outside of melanoma and have broader impacts in oncology," he added.
"I am really impressed by these data," said discussant Dr. Reinhard Dummer of the University Hospital Zurich. In particular, although the trial was not designed to assess responses and the follow-up is still short, some of the patients had complete remissions, he noted.
"I have to admit, I never thought that we could have complete remissions with kinase inhibitors, so I hope that they will continue," he said. "With this in mind, I think we can reset our goals for melanoma research: We don’t want to go for progression-free survival in the future, but we should go for cure."
The reduced rate of the inflammatory rash with the combination was "very surprising," given that the rash is likely a stress response resulting from the MEK inhibitor’s effect on the MAP [mitogen-activated protein] kinase pathway in the skin, Dr. Dummer said. "How can it happen that this rash is reduced in the combination with the BRAF inhibitor? This is completely not understandable for me. Cutaneous biologists will have a lot of fun to study this model."
Both drugs, which are manufactured by GlaxoSmithKline, have previously shown activity as single agents in patients who have melanoma with BRAF V600 gene mutations, which are found in about half of cases. "The goal was to ... give [the drugs] together and really hope to get a profound synergy in combination in pathway inhibition," explained Dr. Infante.
Patients were eligible for the trial if they had melanoma or another cancer with a V600 mutation in the BRAF gene. Those who had stable brain metastases after surgery or radiosurgery could enroll.
Patients having certain eye conditions were excluded because of previously identified, related issues associated with MEK inhibitor therapy. But there was no limit on the number of prior therapies.
In part A of the study, the investigators tested for drug-drug interactions. In part B, they performed dose escalation, assessed safety and activity, and enrolled expansion cohorts of patients, such as those who had previously received a BRAF inhibitor.
Among the eight patients in part A, there was no effect of the MEK inhibitor when given daily on the pharmacokinetics of single doses of the BRAF inhibitor, Dr. Infante reported.
Of the 109 patients in part B, fully 93% had melanoma and 91% specifically had the V600E BRAF mutation. The majority (54%) had received at least two prior lines of systemic anticancer therapy.
Dose-escalation results showed that both drugs could be given in combination at their full monotherapy doses (150 mg twice daily of the BRAF inhibitor and 2 mg once daily of the MEK inhibitor), largely without dose-limiting toxicities.
Only one dose-limiting toxicity was seen with the full dose of each drug. The patient developed a neutrophilic panniculitis, manifesting as painful red nodules on the thorax and extremities, in association with fever and chills. It resolved with discontinuation of the drugs and steroid therapy.
Across the dose combinations, 79% of patients experienced an adverse event and 19% experienced a grade 3 or higher adverse event, most often neutropenia, leukopenia, diarrhea, and fever. There was no clear association with doses, according to Dr. Infante.
In a finding as yet without explanation, certain adverse events that had been seen previously with each drug when they were given alone were actually less common when they were combined. Specifically, rash occurred in 25% of patients, whereas historically it has been seen in 75%-80% of those given the MEK inhibitor alone. And squamous cell carcinoma and other hyperproliferative skin lesions were seen in roughly 1%, whereas they have been seen in 7%-15% of those given the BRAF inhibitor alone.
Among the 71 patients with melanoma who had not previously received a BRAF inhibitor, the rate of unconfirmed objective response (complete or partial response) ranged from 50% to 77%, depending on the dose combination. Five patients had complete responses. For comparison, the response rate in BRAF-mutated melanoma has been 63% with the BRAF inhibitor alone and 40% with the MEK inhibitor alone.
Additionally, the rate of disease control (complete response, partial response, or stable disease) ranged from 95% to 100%, Dr. Infante reported. And the large majority of patients (83%) are still receiving combination therapy.
Analyses among the 24 patients with melanoma who had previously received a BRAF inhibitor showed that about half had a reduction in tumor size. There was no clear difference according to the duration of the earlier BRAF inhibitor therapy.
The investigators are now conducting a phase II trial in which 150 patients with V600E- or V600K-mutant melanoma are being randomly assigned to therapy with the BRAF inhibitor alone or in combination with the MEK inhibitor at two dose levels.
Dr. Dummer, the discussant, noted that obtaining biopsies is "very crucial" to understanding how such novel antimelanoma therapies are working. "Our patients, when you explain this to them, they will agree to sequential biopsies," he commented. "And I encourage all of you to participate in trials with a very strong translational research" component.
Dr. Infante reported that he is an unpaid consultant to GlaxoSmithKline, and that several coauthors are GSK employees. Dr. Dummer reported that he is a consultant to Bristol-Myers Squibb, Merck, Novartis, and Roche, and receives honoraria from Bristol-Myers Squibb.
CHICAGO – The combination of two oral drugs that target selected molecules in the mitogen-activated protein kinase signaling pathway appears to be safe and active in patients with melanoma, a phase I trial found. The finding was reported at the annual meeting of the American Society of Clinical Oncology.
Among patients with BRAF-mutated melanoma who had not previously received an agent targeting that molecule, the combination of an investigational BRAF inhibitor (GSK436) and an investigational MEK inhibitor (GSK212) achieved a response rate of 50%-77%.
Both drugs could be used in their full monotherapy doses when combined. And the combination was well tolerated; indeed, certain adverse effects that were seen with each drug when used alone (such as a rash with MEK inhibitor monotherapy) were less common when they were used together.
"The clinical activity in BRAF V600–mutant melanoma is definitely compelling," commented lead investigator Dr. Jeffrey R. Infante, a medical oncologist and director of drug development at the Sarah Cannon Research Institute in Nashville, Tenn. "But like everyone in the audience, I think, we are anxiously awaiting the long-term durability" data. "This combination could have potential implications in multiple tumor types outside of melanoma and have broader impacts in oncology," he added.
"I am really impressed by these data," said discussant Dr. Reinhard Dummer of the University Hospital Zurich. In particular, although the trial was not designed to assess responses and the follow-up is still short, some of the patients had complete remissions, he noted.
"I have to admit, I never thought that we could have complete remissions with kinase inhibitors, so I hope that they will continue," he said. "With this in mind, I think we can reset our goals for melanoma research: We don’t want to go for progression-free survival in the future, but we should go for cure."
The reduced rate of the inflammatory rash with the combination was "very surprising," given that the rash is likely a stress response resulting from the MEK inhibitor’s effect on the MAP [mitogen-activated protein] kinase pathway in the skin, Dr. Dummer said. "How can it happen that this rash is reduced in the combination with the BRAF inhibitor? This is completely not understandable for me. Cutaneous biologists will have a lot of fun to study this model."
Both drugs, which are manufactured by GlaxoSmithKline, have previously shown activity as single agents in patients who have melanoma with BRAF V600 gene mutations, which are found in about half of cases. "The goal was to ... give [the drugs] together and really hope to get a profound synergy in combination in pathway inhibition," explained Dr. Infante.
Patients were eligible for the trial if they had melanoma or another cancer with a V600 mutation in the BRAF gene. Those who had stable brain metastases after surgery or radiosurgery could enroll.
Patients having certain eye conditions were excluded because of previously identified, related issues associated with MEK inhibitor therapy. But there was no limit on the number of prior therapies.
In part A of the study, the investigators tested for drug-drug interactions. In part B, they performed dose escalation, assessed safety and activity, and enrolled expansion cohorts of patients, such as those who had previously received a BRAF inhibitor.
Among the eight patients in part A, there was no effect of the MEK inhibitor when given daily on the pharmacokinetics of single doses of the BRAF inhibitor, Dr. Infante reported.
Of the 109 patients in part B, fully 93% had melanoma and 91% specifically had the V600E BRAF mutation. The majority (54%) had received at least two prior lines of systemic anticancer therapy.
Dose-escalation results showed that both drugs could be given in combination at their full monotherapy doses (150 mg twice daily of the BRAF inhibitor and 2 mg once daily of the MEK inhibitor), largely without dose-limiting toxicities.
Only one dose-limiting toxicity was seen with the full dose of each drug. The patient developed a neutrophilic panniculitis, manifesting as painful red nodules on the thorax and extremities, in association with fever and chills. It resolved with discontinuation of the drugs and steroid therapy.
Across the dose combinations, 79% of patients experienced an adverse event and 19% experienced a grade 3 or higher adverse event, most often neutropenia, leukopenia, diarrhea, and fever. There was no clear association with doses, according to Dr. Infante.
In a finding as yet without explanation, certain adverse events that had been seen previously with each drug when they were given alone were actually less common when they were combined. Specifically, rash occurred in 25% of patients, whereas historically it has been seen in 75%-80% of those given the MEK inhibitor alone. And squamous cell carcinoma and other hyperproliferative skin lesions were seen in roughly 1%, whereas they have been seen in 7%-15% of those given the BRAF inhibitor alone.
Among the 71 patients with melanoma who had not previously received a BRAF inhibitor, the rate of unconfirmed objective response (complete or partial response) ranged from 50% to 77%, depending on the dose combination. Five patients had complete responses. For comparison, the response rate in BRAF-mutated melanoma has been 63% with the BRAF inhibitor alone and 40% with the MEK inhibitor alone.
Additionally, the rate of disease control (complete response, partial response, or stable disease) ranged from 95% to 100%, Dr. Infante reported. And the large majority of patients (83%) are still receiving combination therapy.
Analyses among the 24 patients with melanoma who had previously received a BRAF inhibitor showed that about half had a reduction in tumor size. There was no clear difference according to the duration of the earlier BRAF inhibitor therapy.
The investigators are now conducting a phase II trial in which 150 patients with V600E- or V600K-mutant melanoma are being randomly assigned to therapy with the BRAF inhibitor alone or in combination with the MEK inhibitor at two dose levels.
Dr. Dummer, the discussant, noted that obtaining biopsies is "very crucial" to understanding how such novel antimelanoma therapies are working. "Our patients, when you explain this to them, they will agree to sequential biopsies," he commented. "And I encourage all of you to participate in trials with a very strong translational research" component.
Dr. Infante reported that he is an unpaid consultant to GlaxoSmithKline, and that several coauthors are GSK employees. Dr. Dummer reported that he is a consultant to Bristol-Myers Squibb, Merck, Novartis, and Roche, and receives honoraria from Bristol-Myers Squibb.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF CLINICAL ONCOLOGY
Major Finding: The objective response rate ranged from 50% to 77%, depending on doses, among patients who had not previously received any BRAF inhibitor therapy.
Data Source: A phase I trial of an oral MEK inhibitor plus an oral BRAF inhibitor that enrolled 101 patients with melanoma and the BRAF V600 mutation
Disclosures: Dr. Infante reported that he is an unpaid consultant to GlaxoSmithKline, and that several of his coauthors are GSK employees. Dr. Dummer reported that he is a consultant to Bristol-Myers Squibb, Merck, Novartis, and Roche, and receives honoraria from Bristol-Myers Squibb.