User login
Hair Stylists Report Looking for Suspicious Lesions
Hair stylists are on the lookout for suspicious lesions on their customers’ scalps, necks, and faces, and are "very" interested in receiving formal skin cancer education, according to a study published Oct. 17 in the Archives of Dermatology.
Although only 28% of hair professionals have received any formal skin cancer education, many reported routinely looking for problematic spots or changing moles, according to Dr. Elizabeth E. Bailey of the department of medicine at Brigham and Women’s Hospital in Boston.
Dr. Bailey and her colleagues received 203 completed surveys from hair professionals in January 2010 on skin cancer practice and knowledge. The hair professionals were from a chain of 17 salons in the greater Houston area.
Hair stylists who frequently talked with their customers about health issues, including personal skin protection practices, were more likely to scan for suspicious lesions. However, it didn’t seem to matter whether or not the stylists had basic skin cancer knowledge, possibly because most already knew the basics, the investigators reported (Arch. Dermatol. 2011;147:1159-65).
According to the study, about 90% agreed or strongly agreed that a customer should see a health professional for a mole that is changing in size or frequently bleeds. A total of 89% said customers should see a health professional if they have a mole that is changing in color, and 78% said moles that itch frequently should be checked out.
A total of 37% of the hair professionals surveyed had scanned more than half of their customers’ scalps, 29% had scanned more than half of their customers’ necks, and 15% had scanned more than half of their customers’ faces for suspicious lesions in the past month.
Survey participants who knew the ABCD rule for melanoma were more likely to look at customers’ skin, and "understanding the difference between melanoma and ordinary skin growths and disagreeing that skin cancer was more difficult to detect than other types of cancer was also associated with a higher likelihood of customer observation," the investigators wrote.
Hair stylists who were confident looking at their own moles also tended to look at their customers’ skin, and stylists who had a personal history of skin cancer or experience with a friend or family member’s skin cancer also looked for potential problem spots on customers’ skin more often, the study found.
About half (49%) of survey participants said they were "very" or "extremely" interested in participating in a skin cancer education program, indicating that dermatologists should consider investigating hair stylists’ potential role in skin cancer prevention and detection.
"Hair professionals are currently acting as lay health advisors for skin cancer detection and prevention and are willing to become more involved in skin cancer education in the salon," said the investigators. "As professionals who have a natural view of difficult-to-see areas and who develop a close rapport with their customers, hair professionals are ideally suited to this role."
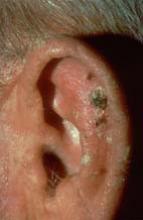
Melanoma of the scalp and neck accounted for 6% of all melanoma and for 10% of melanoma deaths in the United States between 1973 and 2003, likely because it’s difficult to find suspicious lesions in these locations during self-examinations by patients and routine exams by physicians, the investigators noted.
Therefore, hair stylists – who typically see areas of the head and scalp that patients and physicians might miss – are in a unique position to detect skin cancers that might otherwise go unnoticed.
"Through the many active professional education venues within the hair industry, the infrastructure exists to educate them," wrote Dr. Bailey and her colleagues. "Future research should focus on creating a program that provides hair professionals with expert training and effective health communication tools to become confident and skilled lay skin cancer educators."
The investigators did not report having any conflicts of interest.
Hair stylists are on the lookout for suspicious lesions on their customers’ scalps, necks, and faces, and are "very" interested in receiving formal skin cancer education, according to a study published Oct. 17 in the Archives of Dermatology.
Although only 28% of hair professionals have received any formal skin cancer education, many reported routinely looking for problematic spots or changing moles, according to Dr. Elizabeth E. Bailey of the department of medicine at Brigham and Women’s Hospital in Boston.
Dr. Bailey and her colleagues received 203 completed surveys from hair professionals in January 2010 on skin cancer practice and knowledge. The hair professionals were from a chain of 17 salons in the greater Houston area.
Hair stylists who frequently talked with their customers about health issues, including personal skin protection practices, were more likely to scan for suspicious lesions. However, it didn’t seem to matter whether or not the stylists had basic skin cancer knowledge, possibly because most already knew the basics, the investigators reported (Arch. Dermatol. 2011;147:1159-65).
According to the study, about 90% agreed or strongly agreed that a customer should see a health professional for a mole that is changing in size or frequently bleeds. A total of 89% said customers should see a health professional if they have a mole that is changing in color, and 78% said moles that itch frequently should be checked out.
A total of 37% of the hair professionals surveyed had scanned more than half of their customers’ scalps, 29% had scanned more than half of their customers’ necks, and 15% had scanned more than half of their customers’ faces for suspicious lesions in the past month.
Survey participants who knew the ABCD rule for melanoma were more likely to look at customers’ skin, and "understanding the difference between melanoma and ordinary skin growths and disagreeing that skin cancer was more difficult to detect than other types of cancer was also associated with a higher likelihood of customer observation," the investigators wrote.
Hair stylists who were confident looking at their own moles also tended to look at their customers’ skin, and stylists who had a personal history of skin cancer or experience with a friend or family member’s skin cancer also looked for potential problem spots on customers’ skin more often, the study found.
About half (49%) of survey participants said they were "very" or "extremely" interested in participating in a skin cancer education program, indicating that dermatologists should consider investigating hair stylists’ potential role in skin cancer prevention and detection.
"Hair professionals are currently acting as lay health advisors for skin cancer detection and prevention and are willing to become more involved in skin cancer education in the salon," said the investigators. "As professionals who have a natural view of difficult-to-see areas and who develop a close rapport with their customers, hair professionals are ideally suited to this role."
Melanoma of the scalp and neck accounted for 6% of all melanoma and for 10% of melanoma deaths in the United States between 1973 and 2003, likely because it’s difficult to find suspicious lesions in these locations during self-examinations by patients and routine exams by physicians, the investigators noted.
Therefore, hair stylists – who typically see areas of the head and scalp that patients and physicians might miss – are in a unique position to detect skin cancers that might otherwise go unnoticed.
"Through the many active professional education venues within the hair industry, the infrastructure exists to educate them," wrote Dr. Bailey and her colleagues. "Future research should focus on creating a program that provides hair professionals with expert training and effective health communication tools to become confident and skilled lay skin cancer educators."
The investigators did not report having any conflicts of interest.
Hair stylists are on the lookout for suspicious lesions on their customers’ scalps, necks, and faces, and are "very" interested in receiving formal skin cancer education, according to a study published Oct. 17 in the Archives of Dermatology.
Although only 28% of hair professionals have received any formal skin cancer education, many reported routinely looking for problematic spots or changing moles, according to Dr. Elizabeth E. Bailey of the department of medicine at Brigham and Women’s Hospital in Boston.
Dr. Bailey and her colleagues received 203 completed surveys from hair professionals in January 2010 on skin cancer practice and knowledge. The hair professionals were from a chain of 17 salons in the greater Houston area.
Hair stylists who frequently talked with their customers about health issues, including personal skin protection practices, were more likely to scan for suspicious lesions. However, it didn’t seem to matter whether or not the stylists had basic skin cancer knowledge, possibly because most already knew the basics, the investigators reported (Arch. Dermatol. 2011;147:1159-65).
According to the study, about 90% agreed or strongly agreed that a customer should see a health professional for a mole that is changing in size or frequently bleeds. A total of 89% said customers should see a health professional if they have a mole that is changing in color, and 78% said moles that itch frequently should be checked out.
A total of 37% of the hair professionals surveyed had scanned more than half of their customers’ scalps, 29% had scanned more than half of their customers’ necks, and 15% had scanned more than half of their customers’ faces for suspicious lesions in the past month.
Survey participants who knew the ABCD rule for melanoma were more likely to look at customers’ skin, and "understanding the difference between melanoma and ordinary skin growths and disagreeing that skin cancer was more difficult to detect than other types of cancer was also associated with a higher likelihood of customer observation," the investigators wrote.
Hair stylists who were confident looking at their own moles also tended to look at their customers’ skin, and stylists who had a personal history of skin cancer or experience with a friend or family member’s skin cancer also looked for potential problem spots on customers’ skin more often, the study found.
About half (49%) of survey participants said they were "very" or "extremely" interested in participating in a skin cancer education program, indicating that dermatologists should consider investigating hair stylists’ potential role in skin cancer prevention and detection.
"Hair professionals are currently acting as lay health advisors for skin cancer detection and prevention and are willing to become more involved in skin cancer education in the salon," said the investigators. "As professionals who have a natural view of difficult-to-see areas and who develop a close rapport with their customers, hair professionals are ideally suited to this role."
Melanoma of the scalp and neck accounted for 6% of all melanoma and for 10% of melanoma deaths in the United States between 1973 and 2003, likely because it’s difficult to find suspicious lesions in these locations during self-examinations by patients and routine exams by physicians, the investigators noted.
Therefore, hair stylists – who typically see areas of the head and scalp that patients and physicians might miss – are in a unique position to detect skin cancers that might otherwise go unnoticed.
"Through the many active professional education venues within the hair industry, the infrastructure exists to educate them," wrote Dr. Bailey and her colleagues. "Future research should focus on creating a program that provides hair professionals with expert training and effective health communication tools to become confident and skilled lay skin cancer educators."
The investigators did not report having any conflicts of interest.
FROM ARCHIVES OF DERMATOLOGY
Major Finding: A total of 37% of the hair professionals surveyed had scanned more than half of their customers’ scalps, 29% had scanned more than half of their customers’ necks, and 15% had scanned more than half of their customers’ faces for suspicious lesions in the past month.
Data Source: Survey of hair professionals (n = 203) from a chain of 17 salons in the greater Houston area, conducted in January 2010.
Disclosures: The investigators did not report having any conflicts of interest.
NICE Rejects Ipilimumab
A drug considered a breakthrough treatment for advanced melanoma has been turned down by England’s clinical and cost-effectiveness agency.
In first draft guidance issued Oct. 14, the National Institute for Health and Clinical Excellence, which makes recommendations to the National Health Service in England and Wales, said that it was not likely to recommend ipilimumab (Bristol-Myers Squibb’s Yervoy).
The agency cited cost concerns and what it called insufficient follow-up results from a manufacturer-sponsored, phase III, randomized, placebo-controlled trial of ipilimumab (n = 676). The drug, a monoclonal antibody, was shown to significantly improve overall survival in previously treated patients with unresectable stage II or IV melanomas (J. Clin. Oncol. 28:18s, 2010 [suppl; abstr 4]).
Subjects who received ipilimumab alone or with a peptide vaccine saw a median survival of about 10 months, compared with 6.4 months for those receiving only the vaccine. At 1 year follow up, between 44% and 46% of subjects in the ipilimumab arms had survived, compared with 25% in the vaccine-only arm, and at 2 years, between 22% and 24% in the ipilimumab arms had survived, compared with 14% in the vaccine-only arm.
"Ipilimumab is not a cost-effective use of NHS resources."
On the strength of these results, in patient groups for whom few effective treatments exist, the Food and Drug Administration fast-tracked its review of ipilimumab, approving it in March; the European Medicines Agency recommended it in May, and it has been available Europe-wide since July.
But NICE chief executive Andrew Dillon criticized the same results in a news release, saying that ipilimumab had not been compared to the drugs currently used to treat stage III or IV melanoma. (In U.K. practice, this is carboplatin-based chemotherapy, dacarbazine, or supportive care.)
Although the results, Mr. Dillon continued, "did show the drug could potentially be very effective for a small percentage of patients," the follow up from the trial "was too short to determine how long this effect would last."
Clinical specialists have advised NICE that about 30% of people treated with ipilimumab would have improved survival, with an estimated 10% potentially experiencing long-term benefits.
Currently no biomarkers have been established to identify patients who will benefit, Mr. Dillon said, and "ipilimumab currently costs around £80k per patient whether the treatment is effective for them or not." On the basis of evidence submitted so far, he said, "ipilimumab is not a cost-effective use of NHS resources."
NICE’s decision on ipilimumab is likely to be met with fierce criticism and appeals in the United Kingdom, where about 10,000 new cases of malignant melanoma are registered each year, a fifth of them in young adults between the ages of 15 and 39. Some 400-500 Britons with advanced melanoma have disease that has progressed on second-line treatment, NICE said. The 5-year survival rates are between 40% and 50% for stage III disease and 5%-15% for stage IV disease, where median survival is 6-9 months.
Ipilimumab is administered intravenously. It works by blocking the activity of CTLA-4, boosting and prolonging the body’s T-cell response against cancer cells. At £20k per dose, and four doses per course of treatment, NICE estimates the incremental cost effectiveness ratio for ipilimumab at between £54,000 and £70,000 per quality-adjusted life-year gained, based on current evidence.
NICE’s draft guidance on ipilimumab is open for comment until Nov. 4.
A drug considered a breakthrough treatment for advanced melanoma has been turned down by England’s clinical and cost-effectiveness agency.
In first draft guidance issued Oct. 14, the National Institute for Health and Clinical Excellence, which makes recommendations to the National Health Service in England and Wales, said that it was not likely to recommend ipilimumab (Bristol-Myers Squibb’s Yervoy).
The agency cited cost concerns and what it called insufficient follow-up results from a manufacturer-sponsored, phase III, randomized, placebo-controlled trial of ipilimumab (n = 676). The drug, a monoclonal antibody, was shown to significantly improve overall survival in previously treated patients with unresectable stage II or IV melanomas (J. Clin. Oncol. 28:18s, 2010 [suppl; abstr 4]).
Subjects who received ipilimumab alone or with a peptide vaccine saw a median survival of about 10 months, compared with 6.4 months for those receiving only the vaccine. At 1 year follow up, between 44% and 46% of subjects in the ipilimumab arms had survived, compared with 25% in the vaccine-only arm, and at 2 years, between 22% and 24% in the ipilimumab arms had survived, compared with 14% in the vaccine-only arm.
"Ipilimumab is not a cost-effective use of NHS resources."
On the strength of these results, in patient groups for whom few effective treatments exist, the Food and Drug Administration fast-tracked its review of ipilimumab, approving it in March; the European Medicines Agency recommended it in May, and it has been available Europe-wide since July.
But NICE chief executive Andrew Dillon criticized the same results in a news release, saying that ipilimumab had not been compared to the drugs currently used to treat stage III or IV melanoma. (In U.K. practice, this is carboplatin-based chemotherapy, dacarbazine, or supportive care.)
Although the results, Mr. Dillon continued, "did show the drug could potentially be very effective for a small percentage of patients," the follow up from the trial "was too short to determine how long this effect would last."
Clinical specialists have advised NICE that about 30% of people treated with ipilimumab would have improved survival, with an estimated 10% potentially experiencing long-term benefits.
Currently no biomarkers have been established to identify patients who will benefit, Mr. Dillon said, and "ipilimumab currently costs around £80k per patient whether the treatment is effective for them or not." On the basis of evidence submitted so far, he said, "ipilimumab is not a cost-effective use of NHS resources."
NICE’s decision on ipilimumab is likely to be met with fierce criticism and appeals in the United Kingdom, where about 10,000 new cases of malignant melanoma are registered each year, a fifth of them in young adults between the ages of 15 and 39. Some 400-500 Britons with advanced melanoma have disease that has progressed on second-line treatment, NICE said. The 5-year survival rates are between 40% and 50% for stage III disease and 5%-15% for stage IV disease, where median survival is 6-9 months.
Ipilimumab is administered intravenously. It works by blocking the activity of CTLA-4, boosting and prolonging the body’s T-cell response against cancer cells. At £20k per dose, and four doses per course of treatment, NICE estimates the incremental cost effectiveness ratio for ipilimumab at between £54,000 and £70,000 per quality-adjusted life-year gained, based on current evidence.
NICE’s draft guidance on ipilimumab is open for comment until Nov. 4.
A drug considered a breakthrough treatment for advanced melanoma has been turned down by England’s clinical and cost-effectiveness agency.
In first draft guidance issued Oct. 14, the National Institute for Health and Clinical Excellence, which makes recommendations to the National Health Service in England and Wales, said that it was not likely to recommend ipilimumab (Bristol-Myers Squibb’s Yervoy).
The agency cited cost concerns and what it called insufficient follow-up results from a manufacturer-sponsored, phase III, randomized, placebo-controlled trial of ipilimumab (n = 676). The drug, a monoclonal antibody, was shown to significantly improve overall survival in previously treated patients with unresectable stage II or IV melanomas (J. Clin. Oncol. 28:18s, 2010 [suppl; abstr 4]).
Subjects who received ipilimumab alone or with a peptide vaccine saw a median survival of about 10 months, compared with 6.4 months for those receiving only the vaccine. At 1 year follow up, between 44% and 46% of subjects in the ipilimumab arms had survived, compared with 25% in the vaccine-only arm, and at 2 years, between 22% and 24% in the ipilimumab arms had survived, compared with 14% in the vaccine-only arm.
"Ipilimumab is not a cost-effective use of NHS resources."
On the strength of these results, in patient groups for whom few effective treatments exist, the Food and Drug Administration fast-tracked its review of ipilimumab, approving it in March; the European Medicines Agency recommended it in May, and it has been available Europe-wide since July.
But NICE chief executive Andrew Dillon criticized the same results in a news release, saying that ipilimumab had not been compared to the drugs currently used to treat stage III or IV melanoma. (In U.K. practice, this is carboplatin-based chemotherapy, dacarbazine, or supportive care.)
Although the results, Mr. Dillon continued, "did show the drug could potentially be very effective for a small percentage of patients," the follow up from the trial "was too short to determine how long this effect would last."
Clinical specialists have advised NICE that about 30% of people treated with ipilimumab would have improved survival, with an estimated 10% potentially experiencing long-term benefits.
Currently no biomarkers have been established to identify patients who will benefit, Mr. Dillon said, and "ipilimumab currently costs around £80k per patient whether the treatment is effective for them or not." On the basis of evidence submitted so far, he said, "ipilimumab is not a cost-effective use of NHS resources."
NICE’s decision on ipilimumab is likely to be met with fierce criticism and appeals in the United Kingdom, where about 10,000 new cases of malignant melanoma are registered each year, a fifth of them in young adults between the ages of 15 and 39. Some 400-500 Britons with advanced melanoma have disease that has progressed on second-line treatment, NICE said. The 5-year survival rates are between 40% and 50% for stage III disease and 5%-15% for stage IV disease, where median survival is 6-9 months.
Ipilimumab is administered intravenously. It works by blocking the activity of CTLA-4, boosting and prolonging the body’s T-cell response against cancer cells. At £20k per dose, and four doses per course of treatment, NICE estimates the incremental cost effectiveness ratio for ipilimumab at between £54,000 and £70,000 per quality-adjusted life-year gained, based on current evidence.
NICE’s draft guidance on ipilimumab is open for comment until Nov. 4.
California Teen Tanning Ban Signed Into Law
On Oct. 9, California became the first state in the nation to enact a comprehensive ban on the use of indoor tanning beds by minors.
California Gov. Jerry Brown signed a bill sponsored by state Sen. Ted W. Lieu, a democrat. The bill, SB 746, was brought to fruition by the California Society of Dermatology and Dermatologic Surgery and the Aim at Melanoma Foundation, and is supported by the American Academy of Dermatology, the California Medical Association, Anthem Blue Cross, Kaiser Permanente, and the American Academy of Pediatrics, among other groups.
The law, which goes into effect on Jan. 1, 2012, prohibits tanning bed use by anyone under the age of 18, but makes an exception for physicians to prescribe use of the devices for phototherapy.
"Indoor tanning is especially harmful because of the intense and dangerous type of UV rays emitted from the tanning beds," said Sen. Lieu in a statement. "Moreover the skin damage is cumulative, so the more exposure one gets younger in life, the worse the harmful effects will be."
According to the American Academy of Dermatology, exposure to ultraviolet radiation from indoor tanning is associated with an increased risk of melanoma and nonmelanoma skin cancer. There are more than 3.5 million skin cancers in more than 2 million Americans diagnosed annually.
"In 2011, California is expected to have 8,250 new cases of melanoma, which is approximately 12% of the national number of new cases, which is 70,230," said Dr. Ann F. Haas, past president of the California Society of Dermatology and Dermatologic Surgery in a statement. "Melanoma incidence rates have been increasing for the last 30 years, with the most rapid increases occurring among young, white women, 3% per year since 1992 in those ages 15 to 39," she added. "We pushed for this legislation in the hopes of stemming that rise and encouraging other states to follow California’s lead and prohibit the use of tanning devices by minors to reduce the incidence of skin cancer in the U.S."
Dr. Ronald Moy, president of the American Academy of Dermatology, said that the organization supported the legislation and was pleased that it had been made into law. "We commend Gov. Brown, Sen. Ted Lieu, and the other members of the California legislature for their efforts to help reduce the future incidence of skin cancer by protecting youth from the dangers of indoor tanning," said Dr. Moy, in a statement.
Valerie Guild, president and founder of Aim at Melanoma, called the law a "major victory in the fight against melanoma." Added, Ms. Guild: "It is alarming that so many young women are unnecessarily developing melanoma because of a recreational activity. We hope other states will follow California’s lead."
The Indoor Tanning Association issued a statement saying that it was "disappointed" by the Governor’s decision to sign the bill into law. "In making this decision, they ignored the fact that there is no consensus among researchers that normal non-burning exposure to ultraviolet light, whether from the sun or a sun bed, has any effect on the development of melanoma skin cancer," said the association.
The group also said that the law will probably cause more tanning salons to close, reporting that 25% of the state’s salons have shut down since 2009. And, said the Indoor Tanning Association, teens would likely continue to tan outdoors without supervision.
On Oct. 9, California became the first state in the nation to enact a comprehensive ban on the use of indoor tanning beds by minors.
California Gov. Jerry Brown signed a bill sponsored by state Sen. Ted W. Lieu, a democrat. The bill, SB 746, was brought to fruition by the California Society of Dermatology and Dermatologic Surgery and the Aim at Melanoma Foundation, and is supported by the American Academy of Dermatology, the California Medical Association, Anthem Blue Cross, Kaiser Permanente, and the American Academy of Pediatrics, among other groups.
The law, which goes into effect on Jan. 1, 2012, prohibits tanning bed use by anyone under the age of 18, but makes an exception for physicians to prescribe use of the devices for phototherapy.
"Indoor tanning is especially harmful because of the intense and dangerous type of UV rays emitted from the tanning beds," said Sen. Lieu in a statement. "Moreover the skin damage is cumulative, so the more exposure one gets younger in life, the worse the harmful effects will be."
According to the American Academy of Dermatology, exposure to ultraviolet radiation from indoor tanning is associated with an increased risk of melanoma and nonmelanoma skin cancer. There are more than 3.5 million skin cancers in more than 2 million Americans diagnosed annually.
"In 2011, California is expected to have 8,250 new cases of melanoma, which is approximately 12% of the national number of new cases, which is 70,230," said Dr. Ann F. Haas, past president of the California Society of Dermatology and Dermatologic Surgery in a statement. "Melanoma incidence rates have been increasing for the last 30 years, with the most rapid increases occurring among young, white women, 3% per year since 1992 in those ages 15 to 39," she added. "We pushed for this legislation in the hopes of stemming that rise and encouraging other states to follow California’s lead and prohibit the use of tanning devices by minors to reduce the incidence of skin cancer in the U.S."
Dr. Ronald Moy, president of the American Academy of Dermatology, said that the organization supported the legislation and was pleased that it had been made into law. "We commend Gov. Brown, Sen. Ted Lieu, and the other members of the California legislature for their efforts to help reduce the future incidence of skin cancer by protecting youth from the dangers of indoor tanning," said Dr. Moy, in a statement.
Valerie Guild, president and founder of Aim at Melanoma, called the law a "major victory in the fight against melanoma." Added, Ms. Guild: "It is alarming that so many young women are unnecessarily developing melanoma because of a recreational activity. We hope other states will follow California’s lead."
The Indoor Tanning Association issued a statement saying that it was "disappointed" by the Governor’s decision to sign the bill into law. "In making this decision, they ignored the fact that there is no consensus among researchers that normal non-burning exposure to ultraviolet light, whether from the sun or a sun bed, has any effect on the development of melanoma skin cancer," said the association.
The group also said that the law will probably cause more tanning salons to close, reporting that 25% of the state’s salons have shut down since 2009. And, said the Indoor Tanning Association, teens would likely continue to tan outdoors without supervision.
On Oct. 9, California became the first state in the nation to enact a comprehensive ban on the use of indoor tanning beds by minors.
California Gov. Jerry Brown signed a bill sponsored by state Sen. Ted W. Lieu, a democrat. The bill, SB 746, was brought to fruition by the California Society of Dermatology and Dermatologic Surgery and the Aim at Melanoma Foundation, and is supported by the American Academy of Dermatology, the California Medical Association, Anthem Blue Cross, Kaiser Permanente, and the American Academy of Pediatrics, among other groups.
The law, which goes into effect on Jan. 1, 2012, prohibits tanning bed use by anyone under the age of 18, but makes an exception for physicians to prescribe use of the devices for phototherapy.
"Indoor tanning is especially harmful because of the intense and dangerous type of UV rays emitted from the tanning beds," said Sen. Lieu in a statement. "Moreover the skin damage is cumulative, so the more exposure one gets younger in life, the worse the harmful effects will be."
According to the American Academy of Dermatology, exposure to ultraviolet radiation from indoor tanning is associated with an increased risk of melanoma and nonmelanoma skin cancer. There are more than 3.5 million skin cancers in more than 2 million Americans diagnosed annually.
"In 2011, California is expected to have 8,250 new cases of melanoma, which is approximately 12% of the national number of new cases, which is 70,230," said Dr. Ann F. Haas, past president of the California Society of Dermatology and Dermatologic Surgery in a statement. "Melanoma incidence rates have been increasing for the last 30 years, with the most rapid increases occurring among young, white women, 3% per year since 1992 in those ages 15 to 39," she added. "We pushed for this legislation in the hopes of stemming that rise and encouraging other states to follow California’s lead and prohibit the use of tanning devices by minors to reduce the incidence of skin cancer in the U.S."
Dr. Ronald Moy, president of the American Academy of Dermatology, said that the organization supported the legislation and was pleased that it had been made into law. "We commend Gov. Brown, Sen. Ted Lieu, and the other members of the California legislature for their efforts to help reduce the future incidence of skin cancer by protecting youth from the dangers of indoor tanning," said Dr. Moy, in a statement.
Valerie Guild, president and founder of Aim at Melanoma, called the law a "major victory in the fight against melanoma." Added, Ms. Guild: "It is alarming that so many young women are unnecessarily developing melanoma because of a recreational activity. We hope other states will follow California’s lead."
The Indoor Tanning Association issued a statement saying that it was "disappointed" by the Governor’s decision to sign the bill into law. "In making this decision, they ignored the fact that there is no consensus among researchers that normal non-burning exposure to ultraviolet light, whether from the sun or a sun bed, has any effect on the development of melanoma skin cancer," said the association.
The group also said that the law will probably cause more tanning salons to close, reporting that 25% of the state’s salons have shut down since 2009. And, said the Indoor Tanning Association, teens would likely continue to tan outdoors without supervision.
The Skinny Vodcast: Episode 2
Skin & Allergy News editor Amy Pfeiffer and reporter Naseem Miller review hot news in dermatology with the experts. And, guest dermatologist Lily Talakoub tackles the sea of cleansers.
Skin & Allergy News editor Amy Pfeiffer and reporter Naseem Miller review hot news in dermatology with the experts. And, guest dermatologist Lily Talakoub tackles the sea of cleansers.
Skin & Allergy News editor Amy Pfeiffer and reporter Naseem Miller review hot news in dermatology with the experts. And, guest dermatologist Lily Talakoub tackles the sea of cleansers.
Vismodegib Shrinks Advanced Basal Cell Carcinoma
STOCKHOLM – The experimental oral drug vismodegib provided a "substantial clinical benefit" for patients with locally advanced and metastatic basal cell carcinoma in a phase II trial involving 104 patients.
"Nearly all patients did have some tumor shrinkage," Dr. Luc Dirix said at the European Multidisciplinary Cancer Congress. The overall majority of those with locally advanced disease "had huge responses, with major decreases in tumor size."
The primary end point of overall response rate by independent review was reached in 43% of patients with locally advanced basal cell carcinoma (P less than .0001) and in 30% of those with metastatic disease (P = .0011). Response rates per investigator reached 60% and 45.5%, respectively.
Only a small minority of basal cell carcinomas (BCCs) become locally advanced or metastatic, but the consequences can be disfiguring and ultimately life threatening. For these patients, there is no clear standard of care, and thus this is an unmet medical need, explained Dr. Dirix of the Iridium Kankernetwerk at Sint-Augustinus Hospital in Wilrijk, Belgium.
Vismodegib is a first-in-class small-molecule inhibitor of the hedgehog signaling pathway, which is activated in more than 90% of BCC. Genentech, a member of the Roche group, is studying the drug in a variety of cancers. Based on the current phase II data, it filed a New Drug Application, announced on Sept. 12, 2011, with the Food and Drug Administration for vismodegib in the treatment of advanced, inoperable BCC.
"Vismodegib is really a breakthrough for the treatment of advanced basal cell carcinoma, but I think that long-term tolerance is really an issue," the invited discussant, Dr. Caroline Robert, said at the meeting, which was a joint congress of the European Cancer Organization (ECCO), the European Society for Medical Oncology (ESMO), and the European Society for Radiotherapy and Oncology (ESTRO).
"It’s not a very serious adverse event, but [rather] the chronic effect [of] fatigue, loss of appetite, muscular pain that will impact the patients," said Dr. Robert, chief of dermatology at the Institut Cancérologie Gustave Roussy in Villejuif, France.
"Vismodegib is really a breakthrough for the treatment of advanced basal cell carcinoma."
Adverse events occurring in more than 30% of patients were muscle spasms, alopecia, taste loss, weight loss, and fatigue, but they were mainly grade 1 and 2, Dr. Dirix said.
Serious adverse events were reported in 26 patients (25%), of which four were possibly related to vismodegib. They included cholestasis; dehydration with syncope; pneumonia and cardiac failure; and pulmonary embolism. No fatal events were linked to the drug, he said.
The efficacy analysis was based on 63 patients who had locally advanced BCC that was inoperable or for whom surgery would be significantly disfiguring, and 33 patients with histologically confirmed, RECIST (Response Evaluation Criteria in Solid Tumors)–measurable metastatic BCC.
Patients received 150-mg oral vismodegib daily until disease progression or intolerable toxicity. Their mean age was 61 years.
Three-fourths of patients in both cohorts had a clinical benefit (defined as a response at any time plus stable disease), Dr. Dirix said. The response often occurred before week 8, and lasted a median of 7.6 months in both cohorts, based on independent review.
Median progression-free survival by independent review was 9.5 months in both cohorts.
The current phase II trial, called ERIVANCE BCC, was prompted by a phase I trial reporting a 55% response rate in 33 patients with advanced BCC, including 2 complete responses and 16 partial responses (N. Engl. J. Med. 2009;361:1164-72). Only one patient withdrew because of adverse events.
In a recent unpublished phase II study, however, 28% of 41 patients taking vismodegib for Gorlin's syndrome dropped out because of an adverse event, Dr. Robert said during the presidential session. She called for other treatment modalities for advanced BCC, and suggested that vismodegib may be optimal at lower doses, when used sequentially, or as a 3-month course prior to surgery.
"This is very important, I think, because it may lead us to the use of this drug in the neoadjuvant setting, where we can do surgery after the tumor has already shrunk," she added.
Genentech supported the trial. Dr. Dirix reported that a coauthor is a Genentech employee.
STOCKHOLM – The experimental oral drug vismodegib provided a "substantial clinical benefit" for patients with locally advanced and metastatic basal cell carcinoma in a phase II trial involving 104 patients.
"Nearly all patients did have some tumor shrinkage," Dr. Luc Dirix said at the European Multidisciplinary Cancer Congress. The overall majority of those with locally advanced disease "had huge responses, with major decreases in tumor size."
The primary end point of overall response rate by independent review was reached in 43% of patients with locally advanced basal cell carcinoma (P less than .0001) and in 30% of those with metastatic disease (P = .0011). Response rates per investigator reached 60% and 45.5%, respectively.
Only a small minority of basal cell carcinomas (BCCs) become locally advanced or metastatic, but the consequences can be disfiguring and ultimately life threatening. For these patients, there is no clear standard of care, and thus this is an unmet medical need, explained Dr. Dirix of the Iridium Kankernetwerk at Sint-Augustinus Hospital in Wilrijk, Belgium.
Vismodegib is a first-in-class small-molecule inhibitor of the hedgehog signaling pathway, which is activated in more than 90% of BCC. Genentech, a member of the Roche group, is studying the drug in a variety of cancers. Based on the current phase II data, it filed a New Drug Application, announced on Sept. 12, 2011, with the Food and Drug Administration for vismodegib in the treatment of advanced, inoperable BCC.
"Vismodegib is really a breakthrough for the treatment of advanced basal cell carcinoma, but I think that long-term tolerance is really an issue," the invited discussant, Dr. Caroline Robert, said at the meeting, which was a joint congress of the European Cancer Organization (ECCO), the European Society for Medical Oncology (ESMO), and the European Society for Radiotherapy and Oncology (ESTRO).
"It’s not a very serious adverse event, but [rather] the chronic effect [of] fatigue, loss of appetite, muscular pain that will impact the patients," said Dr. Robert, chief of dermatology at the Institut Cancérologie Gustave Roussy in Villejuif, France.
"Vismodegib is really a breakthrough for the treatment of advanced basal cell carcinoma."
Adverse events occurring in more than 30% of patients were muscle spasms, alopecia, taste loss, weight loss, and fatigue, but they were mainly grade 1 and 2, Dr. Dirix said.
Serious adverse events were reported in 26 patients (25%), of which four were possibly related to vismodegib. They included cholestasis; dehydration with syncope; pneumonia and cardiac failure; and pulmonary embolism. No fatal events were linked to the drug, he said.
The efficacy analysis was based on 63 patients who had locally advanced BCC that was inoperable or for whom surgery would be significantly disfiguring, and 33 patients with histologically confirmed, RECIST (Response Evaluation Criteria in Solid Tumors)–measurable metastatic BCC.
Patients received 150-mg oral vismodegib daily until disease progression or intolerable toxicity. Their mean age was 61 years.
Three-fourths of patients in both cohorts had a clinical benefit (defined as a response at any time plus stable disease), Dr. Dirix said. The response often occurred before week 8, and lasted a median of 7.6 months in both cohorts, based on independent review.
Median progression-free survival by independent review was 9.5 months in both cohorts.
The current phase II trial, called ERIVANCE BCC, was prompted by a phase I trial reporting a 55% response rate in 33 patients with advanced BCC, including 2 complete responses and 16 partial responses (N. Engl. J. Med. 2009;361:1164-72). Only one patient withdrew because of adverse events.
In a recent unpublished phase II study, however, 28% of 41 patients taking vismodegib for Gorlin's syndrome dropped out because of an adverse event, Dr. Robert said during the presidential session. She called for other treatment modalities for advanced BCC, and suggested that vismodegib may be optimal at lower doses, when used sequentially, or as a 3-month course prior to surgery.
"This is very important, I think, because it may lead us to the use of this drug in the neoadjuvant setting, where we can do surgery after the tumor has already shrunk," she added.
Genentech supported the trial. Dr. Dirix reported that a coauthor is a Genentech employee.
STOCKHOLM – The experimental oral drug vismodegib provided a "substantial clinical benefit" for patients with locally advanced and metastatic basal cell carcinoma in a phase II trial involving 104 patients.
"Nearly all patients did have some tumor shrinkage," Dr. Luc Dirix said at the European Multidisciplinary Cancer Congress. The overall majority of those with locally advanced disease "had huge responses, with major decreases in tumor size."
The primary end point of overall response rate by independent review was reached in 43% of patients with locally advanced basal cell carcinoma (P less than .0001) and in 30% of those with metastatic disease (P = .0011). Response rates per investigator reached 60% and 45.5%, respectively.
Only a small minority of basal cell carcinomas (BCCs) become locally advanced or metastatic, but the consequences can be disfiguring and ultimately life threatening. For these patients, there is no clear standard of care, and thus this is an unmet medical need, explained Dr. Dirix of the Iridium Kankernetwerk at Sint-Augustinus Hospital in Wilrijk, Belgium.
Vismodegib is a first-in-class small-molecule inhibitor of the hedgehog signaling pathway, which is activated in more than 90% of BCC. Genentech, a member of the Roche group, is studying the drug in a variety of cancers. Based on the current phase II data, it filed a New Drug Application, announced on Sept. 12, 2011, with the Food and Drug Administration for vismodegib in the treatment of advanced, inoperable BCC.
"Vismodegib is really a breakthrough for the treatment of advanced basal cell carcinoma, but I think that long-term tolerance is really an issue," the invited discussant, Dr. Caroline Robert, said at the meeting, which was a joint congress of the European Cancer Organization (ECCO), the European Society for Medical Oncology (ESMO), and the European Society for Radiotherapy and Oncology (ESTRO).
"It’s not a very serious adverse event, but [rather] the chronic effect [of] fatigue, loss of appetite, muscular pain that will impact the patients," said Dr. Robert, chief of dermatology at the Institut Cancérologie Gustave Roussy in Villejuif, France.
"Vismodegib is really a breakthrough for the treatment of advanced basal cell carcinoma."
Adverse events occurring in more than 30% of patients were muscle spasms, alopecia, taste loss, weight loss, and fatigue, but they were mainly grade 1 and 2, Dr. Dirix said.
Serious adverse events were reported in 26 patients (25%), of which four were possibly related to vismodegib. They included cholestasis; dehydration with syncope; pneumonia and cardiac failure; and pulmonary embolism. No fatal events were linked to the drug, he said.
The efficacy analysis was based on 63 patients who had locally advanced BCC that was inoperable or for whom surgery would be significantly disfiguring, and 33 patients with histologically confirmed, RECIST (Response Evaluation Criteria in Solid Tumors)–measurable metastatic BCC.
Patients received 150-mg oral vismodegib daily until disease progression or intolerable toxicity. Their mean age was 61 years.
Three-fourths of patients in both cohorts had a clinical benefit (defined as a response at any time plus stable disease), Dr. Dirix said. The response often occurred before week 8, and lasted a median of 7.6 months in both cohorts, based on independent review.
Median progression-free survival by independent review was 9.5 months in both cohorts.
The current phase II trial, called ERIVANCE BCC, was prompted by a phase I trial reporting a 55% response rate in 33 patients with advanced BCC, including 2 complete responses and 16 partial responses (N. Engl. J. Med. 2009;361:1164-72). Only one patient withdrew because of adverse events.
In a recent unpublished phase II study, however, 28% of 41 patients taking vismodegib for Gorlin's syndrome dropped out because of an adverse event, Dr. Robert said during the presidential session. She called for other treatment modalities for advanced BCC, and suggested that vismodegib may be optimal at lower doses, when used sequentially, or as a 3-month course prior to surgery.
"This is very important, I think, because it may lead us to the use of this drug in the neoadjuvant setting, where we can do surgery after the tumor has already shrunk," she added.
Genentech supported the trial. Dr. Dirix reported that a coauthor is a Genentech employee.
FROM THE EUROPEAN MULTIDISCIPLINARY CANCER CONGRESS
Major Finding: The overall response rate by independent review was 43% for locally advanced BCC (P less than .0001) and 30% for metastatic BCC (P = .0011).
Data Source: Phase II multicenter trial in 104 patients with locally advanced or metastatic BCC.
Disclosures: Genentech supported the trial. Dr. Dirix reported that a coauthor is a Genentech employee.
Unopened Scotch and Genital Warts: The Skinny Podcast
In this month's podcast, we catch up with Dr. Darrel S. Rigel to discuss the spike in melanoma in women in their 20s and 30s.
Contributor Gina Henderson discusses news stats on how dermatology practices are faring in today's tough economy.
And, in "Hats on for Albinism," reporter Naseem Miller interviews the secretary general of the International League of Dermatological Societies, Dr. David McLean, who is leading the effort to help albinos in Tanzania.
In this month's Cosmetic Counter segment, Dr. Lily Talakoub offers tips on how to navigate the overwhelming sea of moisturizers.
And last but not least, Dr. Alan Rockoff relays the story of a how an unopened bottle of scotch eased the nerves of a man facing cryosurgery for his genital warts.
Don't miss another episode of The Skinny Podcast; subscribe on iTunes!
In this month's podcast, we catch up with Dr. Darrel S. Rigel to discuss the spike in melanoma in women in their 20s and 30s.
Contributor Gina Henderson discusses news stats on how dermatology practices are faring in today's tough economy.
And, in "Hats on for Albinism," reporter Naseem Miller interviews the secretary general of the International League of Dermatological Societies, Dr. David McLean, who is leading the effort to help albinos in Tanzania.
In this month's Cosmetic Counter segment, Dr. Lily Talakoub offers tips on how to navigate the overwhelming sea of moisturizers.
And last but not least, Dr. Alan Rockoff relays the story of a how an unopened bottle of scotch eased the nerves of a man facing cryosurgery for his genital warts.
Don't miss another episode of The Skinny Podcast; subscribe on iTunes!
In this month's podcast, we catch up with Dr. Darrel S. Rigel to discuss the spike in melanoma in women in their 20s and 30s.
Contributor Gina Henderson discusses news stats on how dermatology practices are faring in today's tough economy.
And, in "Hats on for Albinism," reporter Naseem Miller interviews the secretary general of the International League of Dermatological Societies, Dr. David McLean, who is leading the effort to help albinos in Tanzania.
In this month's Cosmetic Counter segment, Dr. Lily Talakoub offers tips on how to navigate the overwhelming sea of moisturizers.
And last but not least, Dr. Alan Rockoff relays the story of a how an unopened bottle of scotch eased the nerves of a man facing cryosurgery for his genital warts.
Don't miss another episode of The Skinny Podcast; subscribe on iTunes!
Expert: Risk of AK Progression to SCC Hard to Predict
NAPA, CALIF. – Because actinic keratoses are the most common lesions with premalignant potential in the skin, knowing how to predict their malignancy can be life saving, according to Dr. Miriam S. Bettencourt.
Most AKs do not progress to squamous cell carcinoma (SCC), but most invasive SCCs have evidence of preexisting AK, she said. Of the AKs that progress, 10% will metastasize, giving patients a 5-year survival rate, Dr. Bettencourt of the University of Nevada, Las Vegas.
"Major risk factors for AK malignancy include induration and inflammation, a diameter of over 1 cm, rapid enlargement, bleeding, erythema, and ulceration. Minor risk factors include pigmentation changes, palpability, pain, pruritus, and hyperkeratosis.
Hyperkeratotic AKs greater than 1 cm on the hands, wrists, or forearms have a 50% or greater risk for malignancy, she noted.
Other risk factors include age at diagnosis, human papillomavirus status, skin type, sunburns during childhood, proximity to the Equator, amount of outdoor activity, and a history of skin cancer.
Organ-transplant recipients have a 250-fold greater risk for developing AKs, and a 100-fold risk for SCC; 40% of organ-transplant patients with an AK will show progression to SCC, compared with 10% of the average population, she said.
However, the wide range of rates for the risk of AK progression to SCC found in the literature highlights the difficulty of putting risk into perspective, she noted.
In one study, of 169 patients with AKs, 65% developed an SCC from an AK; 97% of the patients had a contiguous AK near the site of the SCC (Dermatol. Surg. 1995;21:184). But a review of 875 studies of AKs found a much lower rate; 11 studies were identified that predicted the risk of malignancy. The review found the rate to be 0.025% to 10% per lesion, per year (Eur. J. Dermatol. 2006;16:335-9).
In another study, 10% of AKs were found to transform into SCC in 2 years (Dermatol. Surg. 2007;33:1099-101).
A more recent report suggests that AKs also may progress to basal cell carcinomas (BCCs), she reported. In a study of 7,784 lesions on the face and ears of 169 patients, 65% of SCCs and 36% of BCCs arose in lesions that were previously diagnosed as AKs (Cancer 2009;115:2523-30). The study also found the risk of progression of AK to SCC to be 0.6% at year 1 and 2.6% at year 4.
It is important to remember that risk applies to each individual AK and that the fate of any one AK is impossible to predict, said Dr. Bettencourt. "Although the risk [of progression] is small, since most [patients] have multiple AKs, the risk of transformation is much greater than the risk of any individual lesion."
She recommended treating all AKs because some may be difficult to distinguish from SCC and lentigo maligna.
Dr. Bettencourt reported being on the speakers bureaus of PharmaDerm and Graceway, and conducting clinical trials for 3M Pharmaceuticals.
NAPA, CALIF. – Because actinic keratoses are the most common lesions with premalignant potential in the skin, knowing how to predict their malignancy can be life saving, according to Dr. Miriam S. Bettencourt.
Most AKs do not progress to squamous cell carcinoma (SCC), but most invasive SCCs have evidence of preexisting AK, she said. Of the AKs that progress, 10% will metastasize, giving patients a 5-year survival rate, Dr. Bettencourt of the University of Nevada, Las Vegas.
"Major risk factors for AK malignancy include induration and inflammation, a diameter of over 1 cm, rapid enlargement, bleeding, erythema, and ulceration. Minor risk factors include pigmentation changes, palpability, pain, pruritus, and hyperkeratosis.
Hyperkeratotic AKs greater than 1 cm on the hands, wrists, or forearms have a 50% or greater risk for malignancy, she noted.
Other risk factors include age at diagnosis, human papillomavirus status, skin type, sunburns during childhood, proximity to the Equator, amount of outdoor activity, and a history of skin cancer.
Organ-transplant recipients have a 250-fold greater risk for developing AKs, and a 100-fold risk for SCC; 40% of organ-transplant patients with an AK will show progression to SCC, compared with 10% of the average population, she said.
However, the wide range of rates for the risk of AK progression to SCC found in the literature highlights the difficulty of putting risk into perspective, she noted.
In one study, of 169 patients with AKs, 65% developed an SCC from an AK; 97% of the patients had a contiguous AK near the site of the SCC (Dermatol. Surg. 1995;21:184). But a review of 875 studies of AKs found a much lower rate; 11 studies were identified that predicted the risk of malignancy. The review found the rate to be 0.025% to 10% per lesion, per year (Eur. J. Dermatol. 2006;16:335-9).
In another study, 10% of AKs were found to transform into SCC in 2 years (Dermatol. Surg. 2007;33:1099-101).
A more recent report suggests that AKs also may progress to basal cell carcinomas (BCCs), she reported. In a study of 7,784 lesions on the face and ears of 169 patients, 65% of SCCs and 36% of BCCs arose in lesions that were previously diagnosed as AKs (Cancer 2009;115:2523-30). The study also found the risk of progression of AK to SCC to be 0.6% at year 1 and 2.6% at year 4.
It is important to remember that risk applies to each individual AK and that the fate of any one AK is impossible to predict, said Dr. Bettencourt. "Although the risk [of progression] is small, since most [patients] have multiple AKs, the risk of transformation is much greater than the risk of any individual lesion."
She recommended treating all AKs because some may be difficult to distinguish from SCC and lentigo maligna.
Dr. Bettencourt reported being on the speakers bureaus of PharmaDerm and Graceway, and conducting clinical trials for 3M Pharmaceuticals.
NAPA, CALIF. – Because actinic keratoses are the most common lesions with premalignant potential in the skin, knowing how to predict their malignancy can be life saving, according to Dr. Miriam S. Bettencourt.
Most AKs do not progress to squamous cell carcinoma (SCC), but most invasive SCCs have evidence of preexisting AK, she said. Of the AKs that progress, 10% will metastasize, giving patients a 5-year survival rate, Dr. Bettencourt of the University of Nevada, Las Vegas.
"Major risk factors for AK malignancy include induration and inflammation, a diameter of over 1 cm, rapid enlargement, bleeding, erythema, and ulceration. Minor risk factors include pigmentation changes, palpability, pain, pruritus, and hyperkeratosis.
Hyperkeratotic AKs greater than 1 cm on the hands, wrists, or forearms have a 50% or greater risk for malignancy, she noted.
Other risk factors include age at diagnosis, human papillomavirus status, skin type, sunburns during childhood, proximity to the Equator, amount of outdoor activity, and a history of skin cancer.
Organ-transplant recipients have a 250-fold greater risk for developing AKs, and a 100-fold risk for SCC; 40% of organ-transplant patients with an AK will show progression to SCC, compared with 10% of the average population, she said.
However, the wide range of rates for the risk of AK progression to SCC found in the literature highlights the difficulty of putting risk into perspective, she noted.
In one study, of 169 patients with AKs, 65% developed an SCC from an AK; 97% of the patients had a contiguous AK near the site of the SCC (Dermatol. Surg. 1995;21:184). But a review of 875 studies of AKs found a much lower rate; 11 studies were identified that predicted the risk of malignancy. The review found the rate to be 0.025% to 10% per lesion, per year (Eur. J. Dermatol. 2006;16:335-9).
In another study, 10% of AKs were found to transform into SCC in 2 years (Dermatol. Surg. 2007;33:1099-101).
A more recent report suggests that AKs also may progress to basal cell carcinomas (BCCs), she reported. In a study of 7,784 lesions on the face and ears of 169 patients, 65% of SCCs and 36% of BCCs arose in lesions that were previously diagnosed as AKs (Cancer 2009;115:2523-30). The study also found the risk of progression of AK to SCC to be 0.6% at year 1 and 2.6% at year 4.
It is important to remember that risk applies to each individual AK and that the fate of any one AK is impossible to predict, said Dr. Bettencourt. "Although the risk [of progression] is small, since most [patients] have multiple AKs, the risk of transformation is much greater than the risk of any individual lesion."
She recommended treating all AKs because some may be difficult to distinguish from SCC and lentigo maligna.
Dr. Bettencourt reported being on the speakers bureaus of PharmaDerm and Graceway, and conducting clinical trials for 3M Pharmaceuticals.
EXPERT ANALYSIS FROM THE COASTAL DERMATOLOGY SYMPOSISUM
Treat All AKs to Prevent Skin Cancer
NAPA, CALIF. – A preventive approach is needed in the treatment of actinic keratoses to curtail the potential emergence of nonmelanoma skin cancer, according to Dr. Miriam S. Bettencourt.
"In addition to the visible AKs that we see, there’s a lot more in the field area of the AK below the skin surface," she said. "AKs are just the tip of the iceberg."
Because nonmelanoma skin cancer can be easily missed in AK patients, Dr. Bettencourt, of the University of Nevada in Las Vegas, recommended treating all AK lesions.
Many factors should be taken into consideration when determining the best AK treatment for patients, she said, including the number of lesions and location, patient compliance, cosmetic implications, age of patient, history of skin cancer, and insurance coverage.
The treatment of AK involves lesion-directed destructive procedures, topical field-directed therapies, or combination treatment.
Lesion Destruction Therapies
For treating nonspecific lesions, Dr. Bettencourt recommended considering cryosurgery, noting an efficacy rate of 67%-88% and a 1-year recurrence rate of 1.2%-12%. Side effects are mild, the most common being pain, redness, edema, and hypo- or hyperpigmentation.
Chemical peels are an option for extensive AKs, she said, noting an efficacy rate of 75% and a recurrence rate of 25%-35% at 1 year, depending on the agent. Side effects with peels are mild, the most common being pain, inflammation, and pigment alterations.
For single lesions on the face, scalp, or neck, or for full-face resurfacing, CO2 and YAG lasers can be used. While device dependent, 90% of patients achieve lesion remission; however, the recurrence rate is 10%-15% at 3-6 months.
Photodynamic therapy (PDT) offers selective destruction of atypical cells through light activation of a photosensitizer. She noted an efficacy rate of 70%-78% after one treatment session, and a rate of 90% after two sessions. However, there is a risk of photosensitivity and pain from treatment, and it tends to cost more than other therapies, she noted.
Topical Treatment
Topical 5-fluorouracil "inhibits thymidylate synthetase and causes cell death in actively proliferating cells," reported Dr. Bettencourt. A 5% cream or solution is the most popular form of 5-FU, but it is also available in 2%, 1%, and 0.5% formulas.
The 5% cream should be applied twice a day for a month. Common side effects include temporary ulcers, erythema, crusting, and scarring. She highly recommended reviewing the adverse effects with patients before treatment. She noted that she has patients sign the package insert before she begins treatment with 5-FU.
Imiquimod is another topical treatment available for AKs that upregulates a variety of cytokines, she said. "Experimental evidence suggests that patients may develop T-cell memory after treatment, and thus develop less AKs in the future." She recommended Aldara 5%, which is applied 2-3 times per week for up to 4 months, or Zyclara 3.75%, which is applied once a day for two cycles of 2 weeks (with a 2-week free interval in the middle).
The last topical agent she recommended was diclofenac sodium 3% gel (Solaraze), which is a nonsteroidal anti-inflammatory drug that is well tolerated. "The treatment inhibits cyclooxygenase, which is essential for biosynthesis of prostaglandins," she said. Hyaluronic acid delays uptake of the drug, which leads to higher concentrations in the skin. Patients should be treated with the gel twice a day for 3 months. Little to no inflammation may occur during treatment; however, the drug has been known to cause hepatotoxicity.
She noted that diclofenac is her treatment of choice for AKs on the lips.
Combination Therapy
Consider combination, sequential therapy with 0.5% micronized 5-FU before cryotherapy for better results than cryotherapy alone, said Dr. Bettencourt. In a study of 144 patients, 5-FU plus cryotherapy resulted in partial remission in 62% of patients, and complete remission in 17% of patients after 4 weeks of treatment (Arch. Dermatol. 2004;140:813-6).
Low-dose 5-FU plus salicylic acid has also been found to be an effective treatment for AKs, she said. A 2009 study found that 5-FU combined with Jessner’s solution or a 70% glycolic acid peel every other week had an 80% clearance rate and offered photodamage improvement (Int. J. Dermatol. 2009;48:902-7).
PDT followed by treatment with imiquimod 5% was found to reduce lesions by 89.9%, compared with 74.5% with placebo at 12 months (J. Drugs Dermatol. 2009;8:35-9). The randomized vehicle-controlled study enrolled 25 patients (24 completed the study), with a at least 10 AKs. The entire face of each patient was treated with PDT and aminolevulinic acid 20% at baseline and at 1 month; then, at month 2, imiquimod 5% was applied to one-half of each patient’s face and a vehicle to the other half two times a week for 16 weeks. Erythema and flaking were the most serious skin reactions reported during the study.
Positive results have also been seen when imiquimod is combined with cryosurgery, noted Dr. Bettencourt. She also recommended considering diclofenac gel after cryosurgery.
Whatever treatment is deemed best for the AK patient, he or she should be strongly encouraged to avoid the sun whenever possible, to wear protective clothing, to use sunscreen, and to limit outdoor activity between 10 a.m. and 3 p.m., she concluded. A low-fat diet may also lead to a greater resolution of existent AKs and may limit the development of future lesions.
Dr. Bettencourt reported being on the speakers bureaus of PharmaDerm and Graceway, and conducting clinical trials for 3M Pharmaceuticals.
NAPA, CALIF. – A preventive approach is needed in the treatment of actinic keratoses to curtail the potential emergence of nonmelanoma skin cancer, according to Dr. Miriam S. Bettencourt.
"In addition to the visible AKs that we see, there’s a lot more in the field area of the AK below the skin surface," she said. "AKs are just the tip of the iceberg."
Because nonmelanoma skin cancer can be easily missed in AK patients, Dr. Bettencourt, of the University of Nevada in Las Vegas, recommended treating all AK lesions.
Many factors should be taken into consideration when determining the best AK treatment for patients, she said, including the number of lesions and location, patient compliance, cosmetic implications, age of patient, history of skin cancer, and insurance coverage.
The treatment of AK involves lesion-directed destructive procedures, topical field-directed therapies, or combination treatment.
Lesion Destruction Therapies
For treating nonspecific lesions, Dr. Bettencourt recommended considering cryosurgery, noting an efficacy rate of 67%-88% and a 1-year recurrence rate of 1.2%-12%. Side effects are mild, the most common being pain, redness, edema, and hypo- or hyperpigmentation.
Chemical peels are an option for extensive AKs, she said, noting an efficacy rate of 75% and a recurrence rate of 25%-35% at 1 year, depending on the agent. Side effects with peels are mild, the most common being pain, inflammation, and pigment alterations.
For single lesions on the face, scalp, or neck, or for full-face resurfacing, CO2 and YAG lasers can be used. While device dependent, 90% of patients achieve lesion remission; however, the recurrence rate is 10%-15% at 3-6 months.
Photodynamic therapy (PDT) offers selective destruction of atypical cells through light activation of a photosensitizer. She noted an efficacy rate of 70%-78% after one treatment session, and a rate of 90% after two sessions. However, there is a risk of photosensitivity and pain from treatment, and it tends to cost more than other therapies, she noted.
Topical Treatment
Topical 5-fluorouracil "inhibits thymidylate synthetase and causes cell death in actively proliferating cells," reported Dr. Bettencourt. A 5% cream or solution is the most popular form of 5-FU, but it is also available in 2%, 1%, and 0.5% formulas.
The 5% cream should be applied twice a day for a month. Common side effects include temporary ulcers, erythema, crusting, and scarring. She highly recommended reviewing the adverse effects with patients before treatment. She noted that she has patients sign the package insert before she begins treatment with 5-FU.
Imiquimod is another topical treatment available for AKs that upregulates a variety of cytokines, she said. "Experimental evidence suggests that patients may develop T-cell memory after treatment, and thus develop less AKs in the future." She recommended Aldara 5%, which is applied 2-3 times per week for up to 4 months, or Zyclara 3.75%, which is applied once a day for two cycles of 2 weeks (with a 2-week free interval in the middle).
The last topical agent she recommended was diclofenac sodium 3% gel (Solaraze), which is a nonsteroidal anti-inflammatory drug that is well tolerated. "The treatment inhibits cyclooxygenase, which is essential for biosynthesis of prostaglandins," she said. Hyaluronic acid delays uptake of the drug, which leads to higher concentrations in the skin. Patients should be treated with the gel twice a day for 3 months. Little to no inflammation may occur during treatment; however, the drug has been known to cause hepatotoxicity.
She noted that diclofenac is her treatment of choice for AKs on the lips.
Combination Therapy
Consider combination, sequential therapy with 0.5% micronized 5-FU before cryotherapy for better results than cryotherapy alone, said Dr. Bettencourt. In a study of 144 patients, 5-FU plus cryotherapy resulted in partial remission in 62% of patients, and complete remission in 17% of patients after 4 weeks of treatment (Arch. Dermatol. 2004;140:813-6).
Low-dose 5-FU plus salicylic acid has also been found to be an effective treatment for AKs, she said. A 2009 study found that 5-FU combined with Jessner’s solution or a 70% glycolic acid peel every other week had an 80% clearance rate and offered photodamage improvement (Int. J. Dermatol. 2009;48:902-7).
PDT followed by treatment with imiquimod 5% was found to reduce lesions by 89.9%, compared with 74.5% with placebo at 12 months (J. Drugs Dermatol. 2009;8:35-9). The randomized vehicle-controlled study enrolled 25 patients (24 completed the study), with a at least 10 AKs. The entire face of each patient was treated with PDT and aminolevulinic acid 20% at baseline and at 1 month; then, at month 2, imiquimod 5% was applied to one-half of each patient’s face and a vehicle to the other half two times a week for 16 weeks. Erythema and flaking were the most serious skin reactions reported during the study.
Positive results have also been seen when imiquimod is combined with cryosurgery, noted Dr. Bettencourt. She also recommended considering diclofenac gel after cryosurgery.
Whatever treatment is deemed best for the AK patient, he or she should be strongly encouraged to avoid the sun whenever possible, to wear protective clothing, to use sunscreen, and to limit outdoor activity between 10 a.m. and 3 p.m., she concluded. A low-fat diet may also lead to a greater resolution of existent AKs and may limit the development of future lesions.
Dr. Bettencourt reported being on the speakers bureaus of PharmaDerm and Graceway, and conducting clinical trials for 3M Pharmaceuticals.
NAPA, CALIF. – A preventive approach is needed in the treatment of actinic keratoses to curtail the potential emergence of nonmelanoma skin cancer, according to Dr. Miriam S. Bettencourt.
"In addition to the visible AKs that we see, there’s a lot more in the field area of the AK below the skin surface," she said. "AKs are just the tip of the iceberg."
Because nonmelanoma skin cancer can be easily missed in AK patients, Dr. Bettencourt, of the University of Nevada in Las Vegas, recommended treating all AK lesions.
Many factors should be taken into consideration when determining the best AK treatment for patients, she said, including the number of lesions and location, patient compliance, cosmetic implications, age of patient, history of skin cancer, and insurance coverage.
The treatment of AK involves lesion-directed destructive procedures, topical field-directed therapies, or combination treatment.
Lesion Destruction Therapies
For treating nonspecific lesions, Dr. Bettencourt recommended considering cryosurgery, noting an efficacy rate of 67%-88% and a 1-year recurrence rate of 1.2%-12%. Side effects are mild, the most common being pain, redness, edema, and hypo- or hyperpigmentation.
Chemical peels are an option for extensive AKs, she said, noting an efficacy rate of 75% and a recurrence rate of 25%-35% at 1 year, depending on the agent. Side effects with peels are mild, the most common being pain, inflammation, and pigment alterations.
For single lesions on the face, scalp, or neck, or for full-face resurfacing, CO2 and YAG lasers can be used. While device dependent, 90% of patients achieve lesion remission; however, the recurrence rate is 10%-15% at 3-6 months.
Photodynamic therapy (PDT) offers selective destruction of atypical cells through light activation of a photosensitizer. She noted an efficacy rate of 70%-78% after one treatment session, and a rate of 90% after two sessions. However, there is a risk of photosensitivity and pain from treatment, and it tends to cost more than other therapies, she noted.
Topical Treatment
Topical 5-fluorouracil "inhibits thymidylate synthetase and causes cell death in actively proliferating cells," reported Dr. Bettencourt. A 5% cream or solution is the most popular form of 5-FU, but it is also available in 2%, 1%, and 0.5% formulas.
The 5% cream should be applied twice a day for a month. Common side effects include temporary ulcers, erythema, crusting, and scarring. She highly recommended reviewing the adverse effects with patients before treatment. She noted that she has patients sign the package insert before she begins treatment with 5-FU.
Imiquimod is another topical treatment available for AKs that upregulates a variety of cytokines, she said. "Experimental evidence suggests that patients may develop T-cell memory after treatment, and thus develop less AKs in the future." She recommended Aldara 5%, which is applied 2-3 times per week for up to 4 months, or Zyclara 3.75%, which is applied once a day for two cycles of 2 weeks (with a 2-week free interval in the middle).
The last topical agent she recommended was diclofenac sodium 3% gel (Solaraze), which is a nonsteroidal anti-inflammatory drug that is well tolerated. "The treatment inhibits cyclooxygenase, which is essential for biosynthesis of prostaglandins," she said. Hyaluronic acid delays uptake of the drug, which leads to higher concentrations in the skin. Patients should be treated with the gel twice a day for 3 months. Little to no inflammation may occur during treatment; however, the drug has been known to cause hepatotoxicity.
She noted that diclofenac is her treatment of choice for AKs on the lips.
Combination Therapy
Consider combination, sequential therapy with 0.5% micronized 5-FU before cryotherapy for better results than cryotherapy alone, said Dr. Bettencourt. In a study of 144 patients, 5-FU plus cryotherapy resulted in partial remission in 62% of patients, and complete remission in 17% of patients after 4 weeks of treatment (Arch. Dermatol. 2004;140:813-6).
Low-dose 5-FU plus salicylic acid has also been found to be an effective treatment for AKs, she said. A 2009 study found that 5-FU combined with Jessner’s solution or a 70% glycolic acid peel every other week had an 80% clearance rate and offered photodamage improvement (Int. J. Dermatol. 2009;48:902-7).
PDT followed by treatment with imiquimod 5% was found to reduce lesions by 89.9%, compared with 74.5% with placebo at 12 months (J. Drugs Dermatol. 2009;8:35-9). The randomized vehicle-controlled study enrolled 25 patients (24 completed the study), with a at least 10 AKs. The entire face of each patient was treated with PDT and aminolevulinic acid 20% at baseline and at 1 month; then, at month 2, imiquimod 5% was applied to one-half of each patient’s face and a vehicle to the other half two times a week for 16 weeks. Erythema and flaking were the most serious skin reactions reported during the study.
Positive results have also been seen when imiquimod is combined with cryosurgery, noted Dr. Bettencourt. She also recommended considering diclofenac gel after cryosurgery.
Whatever treatment is deemed best for the AK patient, he or she should be strongly encouraged to avoid the sun whenever possible, to wear protective clothing, to use sunscreen, and to limit outdoor activity between 10 a.m. and 3 p.m., she concluded. A low-fat diet may also lead to a greater resolution of existent AKs and may limit the development of future lesions.
Dr. Bettencourt reported being on the speakers bureaus of PharmaDerm and Graceway, and conducting clinical trials for 3M Pharmaceuticals.
EXPERT ANALYSIS FROM THE COASTAL DERMATOLOGY SYMPOSIUM
Shot of Sunscreen a Day Helps Keep Skin Cancer Away
NAPA, CALIF. – When counseling patients to use sunscreens, instructions should also be given on how much to use, recommended Dr. Vincent DeLeo.
Sunscreens should prevent sunburn, photoimmune suppression, photoaging, photocarcinogenicity, and photosensitive disorders – but considering studies have found that people only apply 25%-75% of the testing quantity, sunscreens can’t effectively do their job, noted Dr. DeLeo, chairman of the department of dermatology at St. Luke’s-Roosevelt Hospital Center and Beth Israel Medical Center in New York.
Two tablespoons of sunscreen should cover an adult’s skin surface, he said. And an 8-oz bottle of sunscreen contains 16 tablespoons. Therefore, a family of four at the beach should use one bottle of sunscreen every 2 days, assuming they spend 4 hours a day in the sun (based on American Academy of Dermatology recommendations that sunscreen be applied at least every 2 hours).
Dr. DeLeo noted that a shot glass is 2.5 tablespoons, so this is a good guide for patients. Spray-on sunscreens are a good choice, because they allow patients to self apply to their backs and are a quicker way to coat impatient children. However, 2 tablespoons still need to be used with the sprays.
A recent sunscreen cost analysis found that during a 1-week beach vacation, a family of four should be spending $178-$238 a week on sunscreen, he said. This cost can be brought down by 33% if sun protective clothing is worn, and by 44% if large volumes of sunscreen are purchased. The same cost analysis also found that a transplant patient should be spending $249-$292 per year on sunscreen (J. Am. Acad. Dermatol. 2011;65:e73-9).
It is also important to remind patients that mixing an SPF 10 sunscreen with an SPF 20 will only give them an SPF of 15, he said. And while an SPF 30 may allow patients to stay in the sun twice as long as an SPF 15 with the same amount of protection, it does not offer 100% protection from burning rays.
Sunscreens with an SPF 10 block 90% of damaging rays, SPF 15 blocks 92.5%, SPF 20 blocks 95%, and SPF 40 blocks 97.5%, he noted. Always recommend a high SPF sunscreen to patients, because chances are they are not using enough of whatever they are putting on, he said.
Dr. DeLeo reported relationships with La Roche-Posay, Estée Lauder, Mary Kay, Galderma, Johnson & Johnson, and Pfizer.
NAPA, CALIF. – When counseling patients to use sunscreens, instructions should also be given on how much to use, recommended Dr. Vincent DeLeo.
Sunscreens should prevent sunburn, photoimmune suppression, photoaging, photocarcinogenicity, and photosensitive disorders – but considering studies have found that people only apply 25%-75% of the testing quantity, sunscreens can’t effectively do their job, noted Dr. DeLeo, chairman of the department of dermatology at St. Luke’s-Roosevelt Hospital Center and Beth Israel Medical Center in New York.
Two tablespoons of sunscreen should cover an adult’s skin surface, he said. And an 8-oz bottle of sunscreen contains 16 tablespoons. Therefore, a family of four at the beach should use one bottle of sunscreen every 2 days, assuming they spend 4 hours a day in the sun (based on American Academy of Dermatology recommendations that sunscreen be applied at least every 2 hours).
Dr. DeLeo noted that a shot glass is 2.5 tablespoons, so this is a good guide for patients. Spray-on sunscreens are a good choice, because they allow patients to self apply to their backs and are a quicker way to coat impatient children. However, 2 tablespoons still need to be used with the sprays.
A recent sunscreen cost analysis found that during a 1-week beach vacation, a family of four should be spending $178-$238 a week on sunscreen, he said. This cost can be brought down by 33% if sun protective clothing is worn, and by 44% if large volumes of sunscreen are purchased. The same cost analysis also found that a transplant patient should be spending $249-$292 per year on sunscreen (J. Am. Acad. Dermatol. 2011;65:e73-9).
It is also important to remind patients that mixing an SPF 10 sunscreen with an SPF 20 will only give them an SPF of 15, he said. And while an SPF 30 may allow patients to stay in the sun twice as long as an SPF 15 with the same amount of protection, it does not offer 100% protection from burning rays.
Sunscreens with an SPF 10 block 90% of damaging rays, SPF 15 blocks 92.5%, SPF 20 blocks 95%, and SPF 40 blocks 97.5%, he noted. Always recommend a high SPF sunscreen to patients, because chances are they are not using enough of whatever they are putting on, he said.
Dr. DeLeo reported relationships with La Roche-Posay, Estée Lauder, Mary Kay, Galderma, Johnson & Johnson, and Pfizer.
NAPA, CALIF. – When counseling patients to use sunscreens, instructions should also be given on how much to use, recommended Dr. Vincent DeLeo.
Sunscreens should prevent sunburn, photoimmune suppression, photoaging, photocarcinogenicity, and photosensitive disorders – but considering studies have found that people only apply 25%-75% of the testing quantity, sunscreens can’t effectively do their job, noted Dr. DeLeo, chairman of the department of dermatology at St. Luke’s-Roosevelt Hospital Center and Beth Israel Medical Center in New York.
Two tablespoons of sunscreen should cover an adult’s skin surface, he said. And an 8-oz bottle of sunscreen contains 16 tablespoons. Therefore, a family of four at the beach should use one bottle of sunscreen every 2 days, assuming they spend 4 hours a day in the sun (based on American Academy of Dermatology recommendations that sunscreen be applied at least every 2 hours).
Dr. DeLeo noted that a shot glass is 2.5 tablespoons, so this is a good guide for patients. Spray-on sunscreens are a good choice, because they allow patients to self apply to their backs and are a quicker way to coat impatient children. However, 2 tablespoons still need to be used with the sprays.
A recent sunscreen cost analysis found that during a 1-week beach vacation, a family of four should be spending $178-$238 a week on sunscreen, he said. This cost can be brought down by 33% if sun protective clothing is worn, and by 44% if large volumes of sunscreen are purchased. The same cost analysis also found that a transplant patient should be spending $249-$292 per year on sunscreen (J. Am. Acad. Dermatol. 2011;65:e73-9).
It is also important to remind patients that mixing an SPF 10 sunscreen with an SPF 20 will only give them an SPF of 15, he said. And while an SPF 30 may allow patients to stay in the sun twice as long as an SPF 15 with the same amount of protection, it does not offer 100% protection from burning rays.
Sunscreens with an SPF 10 block 90% of damaging rays, SPF 15 blocks 92.5%, SPF 20 blocks 95%, and SPF 40 blocks 97.5%, he noted. Always recommend a high SPF sunscreen to patients, because chances are they are not using enough of whatever they are putting on, he said.
Dr. DeLeo reported relationships with La Roche-Posay, Estée Lauder, Mary Kay, Galderma, Johnson & Johnson, and Pfizer.
EXPERT ANALYSIS FROM THE COASTAL DERMATOLOGY SYMPOSIUM
FDA: MelaFind Device Is Approvable
The Food and Drug Administration has deemed MelaFind, a noninvasive melanoma detection device, approvable, according to the manufacturer.
The FDA still has a list of requirements the manufacturer, Mela Sciences, must fulfill before MelaFind is given final approval. But company CEO, Joseph V. Gulfo, M.D., said in a conference call with analysts that much of the work is already done and that he expects no significant delays to market.
The road to approval has been somewhat rocky. In November 2010, an FDA advisory panel split 8-7, with one abstention over whether MelaFind should be approved.
Then in May 2011, the company submitted a Citizen Petition to the FDA seeking action on its application for approval.
The company will have to work with the FDA on physician and patient labeling, a package insert and user’s guide, a training program for dermatologists, and the protocol for a postmarketing study.
Dr. Gulfo said that he expects MelaFind to be commercially available in the spring of 2012. Initially, it will only be sold to a handful of dermatologists in Connecticut, New Jersey, and New York. Mela Sciences will be working with dermatologists to fine tune the device before rolling it out to a larger number of practices, he said.
MelaFind is a multispectral computer vision system with a handheld imager that captures the image of a lesion; software uses algorithms to analyze the image, indicating within 2 minutes whether a biopsy should be done.
At the FDA panel meeting in November, some advisers expressed concern that MelaFind might be used by nondermatologists or that it would be used in place of clinical evaluation.
Mela Sciences and the FDA worked on clarifying the indications for use. Those indications now cover multiple paragraphs. According to a Mela Sciences press release, the device "is intended for use on clinically atypical cutaneous pigmented lesions with one or more clinical or historical characteristics of melanoma, excluding those with a clinical diagnosis of melanoma or likely melanoma."
Labeling will also state that the device is "only for use by physicians trained in the clinical diagnosis and management of skin cancer (i.e., dermatologists) who have also successfully completed a training program in the appropriate use of MelaFind."
Dr. Darrell S. Rigel, a consultant for Mela Sciences, said in the company statement that the device can help dermatologists decide which lesions to biopsy and will be an aid to clinical evaluation. "While there have been incremental improvements in imaging tools for melanoma detection, we still primarily rely on our judgment based on a visual examination to select the lesions to biopsy; data show that this is often not enough," said Dr. Rigel, a clinical professor of dermatology at New York University.
The pivotal study of the device included 1,383 patients. According to the company, MelaFind had a sensitivity of 98%. The device’s sensitivity rating for malignant melanoma was significantly better than that of dermatologists, who showed a wide range of variability about which lesions would have been recommended for biopsy and which relegated to observation.
MelaFind will also soon be available in Germany, which has some of the highest rates of melanoma in Europe, said Dr. Gulfo. The device received European Union approval in September. The company aims to have about 75 systems in Germany by next September, Dr. Gulfo said.
The Food and Drug Administration has deemed MelaFind, a noninvasive melanoma detection device, approvable, according to the manufacturer.
The FDA still has a list of requirements the manufacturer, Mela Sciences, must fulfill before MelaFind is given final approval. But company CEO, Joseph V. Gulfo, M.D., said in a conference call with analysts that much of the work is already done and that he expects no significant delays to market.
The road to approval has been somewhat rocky. In November 2010, an FDA advisory panel split 8-7, with one abstention over whether MelaFind should be approved.
Then in May 2011, the company submitted a Citizen Petition to the FDA seeking action on its application for approval.
The company will have to work with the FDA on physician and patient labeling, a package insert and user’s guide, a training program for dermatologists, and the protocol for a postmarketing study.
Dr. Gulfo said that he expects MelaFind to be commercially available in the spring of 2012. Initially, it will only be sold to a handful of dermatologists in Connecticut, New Jersey, and New York. Mela Sciences will be working with dermatologists to fine tune the device before rolling it out to a larger number of practices, he said.
MelaFind is a multispectral computer vision system with a handheld imager that captures the image of a lesion; software uses algorithms to analyze the image, indicating within 2 minutes whether a biopsy should be done.
At the FDA panel meeting in November, some advisers expressed concern that MelaFind might be used by nondermatologists or that it would be used in place of clinical evaluation.
Mela Sciences and the FDA worked on clarifying the indications for use. Those indications now cover multiple paragraphs. According to a Mela Sciences press release, the device "is intended for use on clinically atypical cutaneous pigmented lesions with one or more clinical or historical characteristics of melanoma, excluding those with a clinical diagnosis of melanoma or likely melanoma."
Labeling will also state that the device is "only for use by physicians trained in the clinical diagnosis and management of skin cancer (i.e., dermatologists) who have also successfully completed a training program in the appropriate use of MelaFind."
Dr. Darrell S. Rigel, a consultant for Mela Sciences, said in the company statement that the device can help dermatologists decide which lesions to biopsy and will be an aid to clinical evaluation. "While there have been incremental improvements in imaging tools for melanoma detection, we still primarily rely on our judgment based on a visual examination to select the lesions to biopsy; data show that this is often not enough," said Dr. Rigel, a clinical professor of dermatology at New York University.
The pivotal study of the device included 1,383 patients. According to the company, MelaFind had a sensitivity of 98%. The device’s sensitivity rating for malignant melanoma was significantly better than that of dermatologists, who showed a wide range of variability about which lesions would have been recommended for biopsy and which relegated to observation.
MelaFind will also soon be available in Germany, which has some of the highest rates of melanoma in Europe, said Dr. Gulfo. The device received European Union approval in September. The company aims to have about 75 systems in Germany by next September, Dr. Gulfo said.
The Food and Drug Administration has deemed MelaFind, a noninvasive melanoma detection device, approvable, according to the manufacturer.
The FDA still has a list of requirements the manufacturer, Mela Sciences, must fulfill before MelaFind is given final approval. But company CEO, Joseph V. Gulfo, M.D., said in a conference call with analysts that much of the work is already done and that he expects no significant delays to market.
The road to approval has been somewhat rocky. In November 2010, an FDA advisory panel split 8-7, with one abstention over whether MelaFind should be approved.
Then in May 2011, the company submitted a Citizen Petition to the FDA seeking action on its application for approval.
The company will have to work with the FDA on physician and patient labeling, a package insert and user’s guide, a training program for dermatologists, and the protocol for a postmarketing study.
Dr. Gulfo said that he expects MelaFind to be commercially available in the spring of 2012. Initially, it will only be sold to a handful of dermatologists in Connecticut, New Jersey, and New York. Mela Sciences will be working with dermatologists to fine tune the device before rolling it out to a larger number of practices, he said.
MelaFind is a multispectral computer vision system with a handheld imager that captures the image of a lesion; software uses algorithms to analyze the image, indicating within 2 minutes whether a biopsy should be done.
At the FDA panel meeting in November, some advisers expressed concern that MelaFind might be used by nondermatologists or that it would be used in place of clinical evaluation.
Mela Sciences and the FDA worked on clarifying the indications for use. Those indications now cover multiple paragraphs. According to a Mela Sciences press release, the device "is intended for use on clinically atypical cutaneous pigmented lesions with one or more clinical or historical characteristics of melanoma, excluding those with a clinical diagnosis of melanoma or likely melanoma."
Labeling will also state that the device is "only for use by physicians trained in the clinical diagnosis and management of skin cancer (i.e., dermatologists) who have also successfully completed a training program in the appropriate use of MelaFind."
Dr. Darrell S. Rigel, a consultant for Mela Sciences, said in the company statement that the device can help dermatologists decide which lesions to biopsy and will be an aid to clinical evaluation. "While there have been incremental improvements in imaging tools for melanoma detection, we still primarily rely on our judgment based on a visual examination to select the lesions to biopsy; data show that this is often not enough," said Dr. Rigel, a clinical professor of dermatology at New York University.
The pivotal study of the device included 1,383 patients. According to the company, MelaFind had a sensitivity of 98%. The device’s sensitivity rating for malignant melanoma was significantly better than that of dermatologists, who showed a wide range of variability about which lesions would have been recommended for biopsy and which relegated to observation.
MelaFind will also soon be available in Germany, which has some of the highest rates of melanoma in Europe, said Dr. Gulfo. The device received European Union approval in September. The company aims to have about 75 systems in Germany by next September, Dr. Gulfo said.