User login
FDA: MelaFind Device Is Approvable
The Food and Drug Administration has deemed MelaFind, a noninvasive melanoma detection device, approvable, according to the manufacturer.
The FDA still has a list of requirements the manufacturer, Mela Sciences, must fulfill before MelaFind is given final approval. But company CEO, Joseph V. Gulfo, M.D., said in a conference call with analysts that much of the work is already done and that he expects no significant delays to market.
The road to approval has been somewhat rocky. In November 2010, an FDA advisory panel split 8-7, with one abstention over whether MelaFind should be approved.
Then in May 2011, the company submitted a Citizen Petition to the FDA seeking action on its application for approval.
The company will have to work with the FDA on physician and patient labeling, a package insert and user’s guide, a training program for dermatologists, and the protocol for a postmarketing study.
Dr. Gulfo said that he expects MelaFind to be commercially available in the spring of 2012. Initially, it will only be sold to a handful of dermatologists in Connecticut, New Jersey, and New York. Mela Sciences will be working with dermatologists to fine tune the device before rolling it out to a larger number of practices, he said.
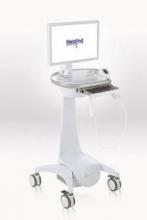
MelaFind is a multispectral computer vision system with a handheld imager that captures the image of a lesion; software uses algorithms to analyze the image, indicating within 2 minutes whether a biopsy should be done.
At the FDA panel meeting in November, some advisers expressed concern that MelaFind might be used by nondermatologists or that it would be used in place of clinical evaluation.
Mela Sciences and the FDA worked on clarifying the indications for use. Those indications now cover multiple paragraphs. According to a Mela Sciences press release, the device "is intended for use on clinically atypical cutaneous pigmented lesions with one or more clinical or historical characteristics of melanoma, excluding those with a clinical diagnosis of melanoma or likely melanoma."
Labeling will also state that the device is "only for use by physicians trained in the clinical diagnosis and management of skin cancer (i.e., dermatologists) who have also successfully completed a training program in the appropriate use of MelaFind."
Dr. Darrell S. Rigel, a consultant for Mela Sciences, said in the company statement that the device can help dermatologists decide which lesions to biopsy and will be an aid to clinical evaluation.* "While there have been incremental improvements in imaging tools for melanoma detection, we still primarily rely on our judgment based on a visual examination to select the lesions to biopsy; data show that this is often not enough," said Dr. Rigel, a clinical professor of dermatology at New York University.
The pivotal study of the device included 1,383 patients. According to the company, MelaFind had a sensitivity of 98%. The device’s sensitivity rating for malignant melanoma was significantly better than that of dermatologists, who showed a wide range of variability about which lesions would have been recommended for biopsy and which relegated to observation.
MelaFind will also soon be available in Germany, which has some of the highest rates of melanoma in Europe, said Dr. Gulfo. The device received European Union approval in September. The company aims to have about 75 systems in Germany by next September, Dr. Gulfo said.
* Correction, 9/30/2011: The original version of this article listed an incorrect title for Dr. Darrell S. Rigel. He is a consultant for Mela Sciences. This version has been corrected.
The Food and Drug Administration has deemed MelaFind, a noninvasive melanoma detection device, approvable, according to the manufacturer.
The FDA still has a list of requirements the manufacturer, Mela Sciences, must fulfill before MelaFind is given final approval. But company CEO, Joseph V. Gulfo, M.D., said in a conference call with analysts that much of the work is already done and that he expects no significant delays to market.
The road to approval has been somewhat rocky. In November 2010, an FDA advisory panel split 8-7, with one abstention over whether MelaFind should be approved.
Then in May 2011, the company submitted a Citizen Petition to the FDA seeking action on its application for approval.
The company will have to work with the FDA on physician and patient labeling, a package insert and user’s guide, a training program for dermatologists, and the protocol for a postmarketing study.
Dr. Gulfo said that he expects MelaFind to be commercially available in the spring of 2012. Initially, it will only be sold to a handful of dermatologists in Connecticut, New Jersey, and New York. Mela Sciences will be working with dermatologists to fine tune the device before rolling it out to a larger number of practices, he said.
MelaFind is a multispectral computer vision system with a handheld imager that captures the image of a lesion; software uses algorithms to analyze the image, indicating within 2 minutes whether a biopsy should be done.
At the FDA panel meeting in November, some advisers expressed concern that MelaFind might be used by nondermatologists or that it would be used in place of clinical evaluation.
Mela Sciences and the FDA worked on clarifying the indications for use. Those indications now cover multiple paragraphs. According to a Mela Sciences press release, the device "is intended for use on clinically atypical cutaneous pigmented lesions with one or more clinical or historical characteristics of melanoma, excluding those with a clinical diagnosis of melanoma or likely melanoma."
Labeling will also state that the device is "only for use by physicians trained in the clinical diagnosis and management of skin cancer (i.e., dermatologists) who have also successfully completed a training program in the appropriate use of MelaFind."
Dr. Darrell S. Rigel, a consultant for Mela Sciences, said in the company statement that the device can help dermatologists decide which lesions to biopsy and will be an aid to clinical evaluation.* "While there have been incremental improvements in imaging tools for melanoma detection, we still primarily rely on our judgment based on a visual examination to select the lesions to biopsy; data show that this is often not enough," said Dr. Rigel, a clinical professor of dermatology at New York University.
The pivotal study of the device included 1,383 patients. According to the company, MelaFind had a sensitivity of 98%. The device’s sensitivity rating for malignant melanoma was significantly better than that of dermatologists, who showed a wide range of variability about which lesions would have been recommended for biopsy and which relegated to observation.
MelaFind will also soon be available in Germany, which has some of the highest rates of melanoma in Europe, said Dr. Gulfo. The device received European Union approval in September. The company aims to have about 75 systems in Germany by next September, Dr. Gulfo said.
* Correction, 9/30/2011: The original version of this article listed an incorrect title for Dr. Darrell S. Rigel. He is a consultant for Mela Sciences. This version has been corrected.
The Food and Drug Administration has deemed MelaFind, a noninvasive melanoma detection device, approvable, according to the manufacturer.
The FDA still has a list of requirements the manufacturer, Mela Sciences, must fulfill before MelaFind is given final approval. But company CEO, Joseph V. Gulfo, M.D., said in a conference call with analysts that much of the work is already done and that he expects no significant delays to market.
The road to approval has been somewhat rocky. In November 2010, an FDA advisory panel split 8-7, with one abstention over whether MelaFind should be approved.
Then in May 2011, the company submitted a Citizen Petition to the FDA seeking action on its application for approval.
The company will have to work with the FDA on physician and patient labeling, a package insert and user’s guide, a training program for dermatologists, and the protocol for a postmarketing study.
Dr. Gulfo said that he expects MelaFind to be commercially available in the spring of 2012. Initially, it will only be sold to a handful of dermatologists in Connecticut, New Jersey, and New York. Mela Sciences will be working with dermatologists to fine tune the device before rolling it out to a larger number of practices, he said.
MelaFind is a multispectral computer vision system with a handheld imager that captures the image of a lesion; software uses algorithms to analyze the image, indicating within 2 minutes whether a biopsy should be done.
At the FDA panel meeting in November, some advisers expressed concern that MelaFind might be used by nondermatologists or that it would be used in place of clinical evaluation.
Mela Sciences and the FDA worked on clarifying the indications for use. Those indications now cover multiple paragraphs. According to a Mela Sciences press release, the device "is intended for use on clinically atypical cutaneous pigmented lesions with one or more clinical or historical characteristics of melanoma, excluding those with a clinical diagnosis of melanoma or likely melanoma."
Labeling will also state that the device is "only for use by physicians trained in the clinical diagnosis and management of skin cancer (i.e., dermatologists) who have also successfully completed a training program in the appropriate use of MelaFind."
Dr. Darrell S. Rigel, a consultant for Mela Sciences, said in the company statement that the device can help dermatologists decide which lesions to biopsy and will be an aid to clinical evaluation.* "While there have been incremental improvements in imaging tools for melanoma detection, we still primarily rely on our judgment based on a visual examination to select the lesions to biopsy; data show that this is often not enough," said Dr. Rigel, a clinical professor of dermatology at New York University.
The pivotal study of the device included 1,383 patients. According to the company, MelaFind had a sensitivity of 98%. The device’s sensitivity rating for malignant melanoma was significantly better than that of dermatologists, who showed a wide range of variability about which lesions would have been recommended for biopsy and which relegated to observation.
MelaFind will also soon be available in Germany, which has some of the highest rates of melanoma in Europe, said Dr. Gulfo. The device received European Union approval in September. The company aims to have about 75 systems in Germany by next September, Dr. Gulfo said.
* Correction, 9/30/2011: The original version of this article listed an incorrect title for Dr. Darrell S. Rigel. He is a consultant for Mela Sciences. This version has been corrected.
AAD Unveils Updated Melanoma Treatment Guidelines
In patients diagnosed with invasive primary cutaneous melanoma, a detailed patient history is crucial, and a thorough examination of the skin and lymph nodes should be performed to help determine the extent of clinical spread of the disease. However, for patients with melanomas of any thickness, baseline blood tests and imaging studies are not recommended because clinical research does not support their use unless suspicious signs and symptoms are present.
Those are two recommendations contained in the American Academy of Dermatology’s updated guidelines for the treatment of primary cutaneous melanoma, which were published online in the Journal of the American Academy of Dermatology. Last updated in 2001, the new guidelines focus on biopsy techniques, pathology, surgical treatment, and long-term follow-up care.
"There are many factors that must be considered when diagnosing and treating melanoma, and these new guidelines offer physicians clinically sound recommendations on how to treat melanoma patients and potentially increase their chance of survival from this deadly disease," AAD President Dr. Ronald L. Moy said in a statement.
A 15-member work group of melanoma experts convened by the AAD and chaired by Dr. Allan C. Halpern of Memorial Sloan-Kettering Cancer Center, New York, and Timothy M. Johnson of the University of Michigan and its Comprehensive Cancer Center, Ann Arbor, reviewed clinical studies and guidelines related to melanoma treatment that appeared in English-language publications between 2000 and 2010. Members of the work group used a 3-point scale to grade evidence, and then developed clinical recommendations on the best available evidence (J. Am. Acad. Dermatol. 2011 Aug. 26 doi: 10.1016/j.jaad.2011.04.031]).
The recommendations were ranked as "A" (recommendation based on consistent and good-quality patient-oriented evidence); "B" (recommendation based on inconsistent or limited-quality patient-oriented evidence); and "C" (recommendation based on consensus, opinion, case studies, or disease-oriented evidence).
The Increased Value of Mitotic Rate
According to Dr. David E. Kent, the most significant change from the 2001 guidelines relates to staging, with mitotic rate replacing Clark level of invasion as the second factor predicting melanoma survival in addition to tumor (Breslow) thickness for tumors 1 mm or smaller in thickness.
Included as a prognostic value in the 2010 American Joint Committee on Cancer staging system to upstage patients with melanoma, the mitotic rate is defined as the number of mitoses in the dermis per mm2.
"The placement of mitotic rate has substantial value for dermatologists because the question we get asked often is, ‘What is the risk of this superficial melanoma spreading to other parts of the body, either regionally to the lymph nodes or distantly to other sites?’" said Dr. Kent, a clinical instructor in the division of dermatology at the Medical College of Georgia, Augusta, who was not involved in assembling the guidelines. "With the mitotic rate less than 1 mm2, it gives us a little bit more comfort in recommending to the patient that there’s less risk of spread. However, if there were to be a substantial mitotic rate, such as 2 mm2, 3 mm2, 4 mm2, or even higher, that would lead one to consider referring the patient for evaluation for a staging and lymph node biopsy, even though the depth of the lesion is thin."
The guidelines note that there is "strong evidence" to support mitotic rate and two other histologic features as the most important characteristics of the primary tumor to predict outcome. The other features are maximum tumor (Breslow) thickness, which "is measured from the granular layer of the overlying epidermis or base of a superficial ulceration to the deepest malignant cells invading dermis to the nearest 0.1 mm, not including deeper adventitial extension," and the presence or absence of microscopic ulceration, "defined as tumor-induced full thickness loss of epidermis with subjacent dermal tumor and reactive dermal changes."
A complete physical exam is recommended by the work group and "includes looking through the scalp, looking in the mouth, and a genital exam as well," Dr. Kent said. "In our practice we refer women to their gynecologist for that, because 1% of all melanomas are genital. We also recommend that they see their medical ophthalmologist every 2 years because you can get melanomas in different areas of the eye."
Screening blood tests, including serum lactate dehydrogenase, are "insensitive for the detection of metastatic disease," the guidelines state, while the use of routine imaging studies "is limited by a very low yield and the frequent occurrence of false positive findings. Ample evidence exists that a routine chest X-ray is a cost inefficient test for the detection of metastatic disease with a consistent relatively high false positive rate." The guidelines also note that advanced imaging studies such as positron emission tomography and computed tomography have lower sensitivity, compared with sentinel lymph node biopsy (SLNB).
As for follow-up of patients with newly diagnosed cutaneous melanoma, the guidelines recommend an annual visit with a dermatologist at the least, but note that visits may range from every 3-12 months based on a patient’s history and risk factors. "The goal of follow-up is to detect any evidence of local recurrence and to detect any additional primary melanomas that may have developed," Dr. Kent said. "I have several patients that have personally had over three melanomas. One patient has had five. Some high risk patients we see every 3 or 4 months for follow-up."
Sentinel Lymph Node Biopsy and Lentigo Maligna
The work group also addressed non-surgical treatments for lentigo maligna, a topic that was not contained in the older version of the guidelines. For example, while off-label use of topical imiquimod has been proposed for lentigo maligna, "studies are limited by highly variable treatment regimens and lack of long-term follow-up with an average of approximately 18 months," the work group wrote. "Histologic verification following treatment has shown persistent disease in approximately 25% of treated patients and progression to invasive melanoma has been noted. As an adjunctive modality following surgical excision, the efficacy of topical imiquimod has not been established. High cost of treatment, an appropriate low threshold for subsequent biopsy to exclude residual or recurrent disease, and the risk of a severe inflammatory reaction should be taken into account when considering imiquimod."
SLNB is another topic contained in the guidelines for the first time. While the work group acknowledged that SLNB is "not without controversy," it described sentinel lymph node status as "the most important prognostic factor for disease-specific survival of patients with melanoma greater than 1 mm in thickness. Whether early detection of occult nodal disease provides greater regional control has not been definitively shown, but available evidence suggests a lower rate of post-operative complications in patients who underwent completion lymph node dissection for micrometastatic disease detected by SLNB, compared to those who underwent therapeutic lymph node dissection for clinically palpable disease."
The work group acknowledged that "significant gaps" exist in research related to the management of primary cutaneous melanoma, including "standardization of the interpretation of mitotic rate; placebo-controlled trials for the treatment of lentigo maligna; the use and value of dermoscopy and other imaging modalities; the clinical and prognostic significance of the use of biomarkers and mutational analysis; and the use of sentinel lymph node biopsy."
For his part, Dr. Kent predicted that major advances in the treatment of cutaneous melanoma should occur in the coming years. "Melanoma is a very unpredictable disease," he said. "Hopefully sometime in the future, we’ll have biomarkers that will assist physicians in identifying patients who are a greater risk of more aggressive disease, of spread to both regional and distant sites. That area of research is very exciting."
Dr. Kent said that he had no relevant financial disclosures.
In patients diagnosed with invasive primary cutaneous melanoma, a detailed patient history is crucial, and a thorough examination of the skin and lymph nodes should be performed to help determine the extent of clinical spread of the disease. However, for patients with melanomas of any thickness, baseline blood tests and imaging studies are not recommended because clinical research does not support their use unless suspicious signs and symptoms are present.
Those are two recommendations contained in the American Academy of Dermatology’s updated guidelines for the treatment of primary cutaneous melanoma, which were published online in the Journal of the American Academy of Dermatology. Last updated in 2001, the new guidelines focus on biopsy techniques, pathology, surgical treatment, and long-term follow-up care.
"There are many factors that must be considered when diagnosing and treating melanoma, and these new guidelines offer physicians clinically sound recommendations on how to treat melanoma patients and potentially increase their chance of survival from this deadly disease," AAD President Dr. Ronald L. Moy said in a statement.
A 15-member work group of melanoma experts convened by the AAD and chaired by Dr. Allan C. Halpern of Memorial Sloan-Kettering Cancer Center, New York, and Timothy M. Johnson of the University of Michigan and its Comprehensive Cancer Center, Ann Arbor, reviewed clinical studies and guidelines related to melanoma treatment that appeared in English-language publications between 2000 and 2010. Members of the work group used a 3-point scale to grade evidence, and then developed clinical recommendations on the best available evidence (J. Am. Acad. Dermatol. 2011 Aug. 26 doi: 10.1016/j.jaad.2011.04.031]).
The recommendations were ranked as "A" (recommendation based on consistent and good-quality patient-oriented evidence); "B" (recommendation based on inconsistent or limited-quality patient-oriented evidence); and "C" (recommendation based on consensus, opinion, case studies, or disease-oriented evidence).
The Increased Value of Mitotic Rate
According to Dr. David E. Kent, the most significant change from the 2001 guidelines relates to staging, with mitotic rate replacing Clark level of invasion as the second factor predicting melanoma survival in addition to tumor (Breslow) thickness for tumors 1 mm or smaller in thickness.
Included as a prognostic value in the 2010 American Joint Committee on Cancer staging system to upstage patients with melanoma, the mitotic rate is defined as the number of mitoses in the dermis per mm2.
"The placement of mitotic rate has substantial value for dermatologists because the question we get asked often is, ‘What is the risk of this superficial melanoma spreading to other parts of the body, either regionally to the lymph nodes or distantly to other sites?’" said Dr. Kent, a clinical instructor in the division of dermatology at the Medical College of Georgia, Augusta, who was not involved in assembling the guidelines. "With the mitotic rate less than 1 mm2, it gives us a little bit more comfort in recommending to the patient that there’s less risk of spread. However, if there were to be a substantial mitotic rate, such as 2 mm2, 3 mm2, 4 mm2, or even higher, that would lead one to consider referring the patient for evaluation for a staging and lymph node biopsy, even though the depth of the lesion is thin."
The guidelines note that there is "strong evidence" to support mitotic rate and two other histologic features as the most important characteristics of the primary tumor to predict outcome. The other features are maximum tumor (Breslow) thickness, which "is measured from the granular layer of the overlying epidermis or base of a superficial ulceration to the deepest malignant cells invading dermis to the nearest 0.1 mm, not including deeper adventitial extension," and the presence or absence of microscopic ulceration, "defined as tumor-induced full thickness loss of epidermis with subjacent dermal tumor and reactive dermal changes."
A complete physical exam is recommended by the work group and "includes looking through the scalp, looking in the mouth, and a genital exam as well," Dr. Kent said. "In our practice we refer women to their gynecologist for that, because 1% of all melanomas are genital. We also recommend that they see their medical ophthalmologist every 2 years because you can get melanomas in different areas of the eye."
Screening blood tests, including serum lactate dehydrogenase, are "insensitive for the detection of metastatic disease," the guidelines state, while the use of routine imaging studies "is limited by a very low yield and the frequent occurrence of false positive findings. Ample evidence exists that a routine chest X-ray is a cost inefficient test for the detection of metastatic disease with a consistent relatively high false positive rate." The guidelines also note that advanced imaging studies such as positron emission tomography and computed tomography have lower sensitivity, compared with sentinel lymph node biopsy (SLNB).
As for follow-up of patients with newly diagnosed cutaneous melanoma, the guidelines recommend an annual visit with a dermatologist at the least, but note that visits may range from every 3-12 months based on a patient’s history and risk factors. "The goal of follow-up is to detect any evidence of local recurrence and to detect any additional primary melanomas that may have developed," Dr. Kent said. "I have several patients that have personally had over three melanomas. One patient has had five. Some high risk patients we see every 3 or 4 months for follow-up."
Sentinel Lymph Node Biopsy and Lentigo Maligna
The work group also addressed non-surgical treatments for lentigo maligna, a topic that was not contained in the older version of the guidelines. For example, while off-label use of topical imiquimod has been proposed for lentigo maligna, "studies are limited by highly variable treatment regimens and lack of long-term follow-up with an average of approximately 18 months," the work group wrote. "Histologic verification following treatment has shown persistent disease in approximately 25% of treated patients and progression to invasive melanoma has been noted. As an adjunctive modality following surgical excision, the efficacy of topical imiquimod has not been established. High cost of treatment, an appropriate low threshold for subsequent biopsy to exclude residual or recurrent disease, and the risk of a severe inflammatory reaction should be taken into account when considering imiquimod."
SLNB is another topic contained in the guidelines for the first time. While the work group acknowledged that SLNB is "not without controversy," it described sentinel lymph node status as "the most important prognostic factor for disease-specific survival of patients with melanoma greater than 1 mm in thickness. Whether early detection of occult nodal disease provides greater regional control has not been definitively shown, but available evidence suggests a lower rate of post-operative complications in patients who underwent completion lymph node dissection for micrometastatic disease detected by SLNB, compared to those who underwent therapeutic lymph node dissection for clinically palpable disease."
The work group acknowledged that "significant gaps" exist in research related to the management of primary cutaneous melanoma, including "standardization of the interpretation of mitotic rate; placebo-controlled trials for the treatment of lentigo maligna; the use and value of dermoscopy and other imaging modalities; the clinical and prognostic significance of the use of biomarkers and mutational analysis; and the use of sentinel lymph node biopsy."
For his part, Dr. Kent predicted that major advances in the treatment of cutaneous melanoma should occur in the coming years. "Melanoma is a very unpredictable disease," he said. "Hopefully sometime in the future, we’ll have biomarkers that will assist physicians in identifying patients who are a greater risk of more aggressive disease, of spread to both regional and distant sites. That area of research is very exciting."
Dr. Kent said that he had no relevant financial disclosures.
In patients diagnosed with invasive primary cutaneous melanoma, a detailed patient history is crucial, and a thorough examination of the skin and lymph nodes should be performed to help determine the extent of clinical spread of the disease. However, for patients with melanomas of any thickness, baseline blood tests and imaging studies are not recommended because clinical research does not support their use unless suspicious signs and symptoms are present.
Those are two recommendations contained in the American Academy of Dermatology’s updated guidelines for the treatment of primary cutaneous melanoma, which were published online in the Journal of the American Academy of Dermatology. Last updated in 2001, the new guidelines focus on biopsy techniques, pathology, surgical treatment, and long-term follow-up care.
"There are many factors that must be considered when diagnosing and treating melanoma, and these new guidelines offer physicians clinically sound recommendations on how to treat melanoma patients and potentially increase their chance of survival from this deadly disease," AAD President Dr. Ronald L. Moy said in a statement.
A 15-member work group of melanoma experts convened by the AAD and chaired by Dr. Allan C. Halpern of Memorial Sloan-Kettering Cancer Center, New York, and Timothy M. Johnson of the University of Michigan and its Comprehensive Cancer Center, Ann Arbor, reviewed clinical studies and guidelines related to melanoma treatment that appeared in English-language publications between 2000 and 2010. Members of the work group used a 3-point scale to grade evidence, and then developed clinical recommendations on the best available evidence (J. Am. Acad. Dermatol. 2011 Aug. 26 doi: 10.1016/j.jaad.2011.04.031]).
The recommendations were ranked as "A" (recommendation based on consistent and good-quality patient-oriented evidence); "B" (recommendation based on inconsistent or limited-quality patient-oriented evidence); and "C" (recommendation based on consensus, opinion, case studies, or disease-oriented evidence).
The Increased Value of Mitotic Rate
According to Dr. David E. Kent, the most significant change from the 2001 guidelines relates to staging, with mitotic rate replacing Clark level of invasion as the second factor predicting melanoma survival in addition to tumor (Breslow) thickness for tumors 1 mm or smaller in thickness.
Included as a prognostic value in the 2010 American Joint Committee on Cancer staging system to upstage patients with melanoma, the mitotic rate is defined as the number of mitoses in the dermis per mm2.
"The placement of mitotic rate has substantial value for dermatologists because the question we get asked often is, ‘What is the risk of this superficial melanoma spreading to other parts of the body, either regionally to the lymph nodes or distantly to other sites?’" said Dr. Kent, a clinical instructor in the division of dermatology at the Medical College of Georgia, Augusta, who was not involved in assembling the guidelines. "With the mitotic rate less than 1 mm2, it gives us a little bit more comfort in recommending to the patient that there’s less risk of spread. However, if there were to be a substantial mitotic rate, such as 2 mm2, 3 mm2, 4 mm2, or even higher, that would lead one to consider referring the patient for evaluation for a staging and lymph node biopsy, even though the depth of the lesion is thin."
The guidelines note that there is "strong evidence" to support mitotic rate and two other histologic features as the most important characteristics of the primary tumor to predict outcome. The other features are maximum tumor (Breslow) thickness, which "is measured from the granular layer of the overlying epidermis or base of a superficial ulceration to the deepest malignant cells invading dermis to the nearest 0.1 mm, not including deeper adventitial extension," and the presence or absence of microscopic ulceration, "defined as tumor-induced full thickness loss of epidermis with subjacent dermal tumor and reactive dermal changes."
A complete physical exam is recommended by the work group and "includes looking through the scalp, looking in the mouth, and a genital exam as well," Dr. Kent said. "In our practice we refer women to their gynecologist for that, because 1% of all melanomas are genital. We also recommend that they see their medical ophthalmologist every 2 years because you can get melanomas in different areas of the eye."
Screening blood tests, including serum lactate dehydrogenase, are "insensitive for the detection of metastatic disease," the guidelines state, while the use of routine imaging studies "is limited by a very low yield and the frequent occurrence of false positive findings. Ample evidence exists that a routine chest X-ray is a cost inefficient test for the detection of metastatic disease with a consistent relatively high false positive rate." The guidelines also note that advanced imaging studies such as positron emission tomography and computed tomography have lower sensitivity, compared with sentinel lymph node biopsy (SLNB).
As for follow-up of patients with newly diagnosed cutaneous melanoma, the guidelines recommend an annual visit with a dermatologist at the least, but note that visits may range from every 3-12 months based on a patient’s history and risk factors. "The goal of follow-up is to detect any evidence of local recurrence and to detect any additional primary melanomas that may have developed," Dr. Kent said. "I have several patients that have personally had over three melanomas. One patient has had five. Some high risk patients we see every 3 or 4 months for follow-up."
Sentinel Lymph Node Biopsy and Lentigo Maligna
The work group also addressed non-surgical treatments for lentigo maligna, a topic that was not contained in the older version of the guidelines. For example, while off-label use of topical imiquimod has been proposed for lentigo maligna, "studies are limited by highly variable treatment regimens and lack of long-term follow-up with an average of approximately 18 months," the work group wrote. "Histologic verification following treatment has shown persistent disease in approximately 25% of treated patients and progression to invasive melanoma has been noted. As an adjunctive modality following surgical excision, the efficacy of topical imiquimod has not been established. High cost of treatment, an appropriate low threshold for subsequent biopsy to exclude residual or recurrent disease, and the risk of a severe inflammatory reaction should be taken into account when considering imiquimod."
SLNB is another topic contained in the guidelines for the first time. While the work group acknowledged that SLNB is "not without controversy," it described sentinel lymph node status as "the most important prognostic factor for disease-specific survival of patients with melanoma greater than 1 mm in thickness. Whether early detection of occult nodal disease provides greater regional control has not been definitively shown, but available evidence suggests a lower rate of post-operative complications in patients who underwent completion lymph node dissection for micrometastatic disease detected by SLNB, compared to those who underwent therapeutic lymph node dissection for clinically palpable disease."
The work group acknowledged that "significant gaps" exist in research related to the management of primary cutaneous melanoma, including "standardization of the interpretation of mitotic rate; placebo-controlled trials for the treatment of lentigo maligna; the use and value of dermoscopy and other imaging modalities; the clinical and prognostic significance of the use of biomarkers and mutational analysis; and the use of sentinel lymph node biopsy."
For his part, Dr. Kent predicted that major advances in the treatment of cutaneous melanoma should occur in the coming years. "Melanoma is a very unpredictable disease," he said. "Hopefully sometime in the future, we’ll have biomarkers that will assist physicians in identifying patients who are a greater risk of more aggressive disease, of spread to both regional and distant sites. That area of research is very exciting."
Dr. Kent said that he had no relevant financial disclosures.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Oncovirus Expert: Cattle May Transmit Human Cancers
Current estimates hold that 21% of all human cancers are linked to infections. But some experts believe this figure is too low and is headed substantially higher.
Fueled by the success of vaccines for the hepatitis B virus and high-risk human papillomaviruses, which offer the first-ever means of preventing specific widespread cancers by vaccination, investigators are hunting for additional infectious causes of common malignancies. The search is focused on viruses, since they are already implicated in 64% of the known infection-related cancer burden, with bacteria and parasites accounting for the remainder.
One of the world’s most celebrated oncovirus hunters is Dr. Harald zur Hausen, winner of the 2008 Nobel Prize in Medicine for his work on human papillomaviruses as the major cause of cervical cancer. He sees a number of other malignancies as being prime candidates for potential linkage to infections, including childhood lymphoblastic leukemias, basal cell carcinomas, Epstein-Barr virus–negative Hodgkin’s lymphomas, colorectal and breast cancers, and lung cancers in nonsmokers.
The TTV (torque teno virus) family shows particular promise in this regard. The TTVs, first described by Japanese investigators in 1997 (Rev. Med. Virol. 2007;17:45-57), are an extremely heterogeneous family of single-strand DNA viruses. There are well over 100 genotypes. The torque teno viruses are widespread in all human populations. They have been found in umbilical cord blood and are vertically transmitted from mother to child, even prenatally. They frequently rearrange their genomes; and transmissibility and replication have been demonstrated to occur even for small portions of the TTV genome, according to Dr. zur Hausen, professor emeritus at the German Cancer Research Center, Heidelberg.
The TTVs are known to replicate in lymphatic and bone marrow cells – the precursor cells of leukemia. In every cell line of acute lymphoblastic leukemia and Epstein-Barr virus–negative Hodgkin’s lymphoma analyzed by Dr. zur Hausen and his coworkers, they have found the same highly conserved region of TTV DNA.
Of note, TTV load is known to be reduced by interferon. This finding is consistent with the epidemiologic observation that infants who experience multiple upper respiratory infections and other infections during their first year of life seem to be protected against childhood leukemias. The hypothesis is that bursts of increased interferon production in response to multiple infections prevents levels of an interferon-sensitive putative leukemogenic agent – perhaps a TTV – from reaching critical mass.
Dr. zur Hausen is particularly attracted to the possibility that cattle may play a role in some human cancers. He hypothesizes that some as-yet unidentified bovine virus, which is nononcogenic in its normal host, can become carcinogenic when transmitted to humans. Multiple lines of circumstantial evidence support this notion. For example, basal cell carcinomas are known to have a predisposition to arise in smallpox vaccination scars. Smallpox vaccines were prepared by inoculating vaccinia virus into the skin of calves and then harvesting the crusted skin, which could in theory contain contaminating bovine viruses.
Also, colorectal cancer, and to a lesser extent breast cancer and lung cancer in nonsmokers, have repeatedly been associated with beef consumption in epidemiologic studies. Countries with high consumption of goat or pork have relatively low rates of these malignancies.
The observed link between a red meat–rich diet and increased rates of colorectal and other cancers is often attributed to the formation of aromatic hydrocarbons and other known carcinogens during cooking or meat curing. But Dr. zur Hausen believes this interpretation might be inadequate. He noted that grilled, fried, and smoked poultry contain similar concentrations of these carcinogens, yet heavy consumption of poultry hasn’t been associated with an increased cancer risk.
It is of interest that the temperature achieved in the center of a piece of beef cooked rare is only 40 -50° C, yet torque teno viruses, papillomaviruses, and polyomaviruses are able to survive in a protein environment at temperatures of 80° C for 30 minutes or more.
"These are just suggestions. They do not prove anything, of course. But, for now, we can speculate that beef consumption plays a significant role in the development of colorectal cancer," Dr. zur Hausen said.
If even a small portion of these speculations turn out to be true, the implications for cancer prevention could be huge. It is estimated that if both the hepatitis B vaccine and the HPV vaccine were to be applied globally, the overall cancer burden in women could be reduced by 12%-14% and in men by 4%-5%, he noted.
He presented his theory at the World Congress of Dermatology in Seoul, South Korea.
Dr. zur Hausen declared having no financial conflicts.
Current estimates hold that 21% of all human cancers are linked to infections. But some experts believe this figure is too low and is headed substantially higher.
Fueled by the success of vaccines for the hepatitis B virus and high-risk human papillomaviruses, which offer the first-ever means of preventing specific widespread cancers by vaccination, investigators are hunting for additional infectious causes of common malignancies. The search is focused on viruses, since they are already implicated in 64% of the known infection-related cancer burden, with bacteria and parasites accounting for the remainder.
One of the world’s most celebrated oncovirus hunters is Dr. Harald zur Hausen, winner of the 2008 Nobel Prize in Medicine for his work on human papillomaviruses as the major cause of cervical cancer. He sees a number of other malignancies as being prime candidates for potential linkage to infections, including childhood lymphoblastic leukemias, basal cell carcinomas, Epstein-Barr virus–negative Hodgkin’s lymphomas, colorectal and breast cancers, and lung cancers in nonsmokers.
The TTV (torque teno virus) family shows particular promise in this regard. The TTVs, first described by Japanese investigators in 1997 (Rev. Med. Virol. 2007;17:45-57), are an extremely heterogeneous family of single-strand DNA viruses. There are well over 100 genotypes. The torque teno viruses are widespread in all human populations. They have been found in umbilical cord blood and are vertically transmitted from mother to child, even prenatally. They frequently rearrange their genomes; and transmissibility and replication have been demonstrated to occur even for small portions of the TTV genome, according to Dr. zur Hausen, professor emeritus at the German Cancer Research Center, Heidelberg.
The TTVs are known to replicate in lymphatic and bone marrow cells – the precursor cells of leukemia. In every cell line of acute lymphoblastic leukemia and Epstein-Barr virus–negative Hodgkin’s lymphoma analyzed by Dr. zur Hausen and his coworkers, they have found the same highly conserved region of TTV DNA.
Of note, TTV load is known to be reduced by interferon. This finding is consistent with the epidemiologic observation that infants who experience multiple upper respiratory infections and other infections during their first year of life seem to be protected against childhood leukemias. The hypothesis is that bursts of increased interferon production in response to multiple infections prevents levels of an interferon-sensitive putative leukemogenic agent – perhaps a TTV – from reaching critical mass.
Dr. zur Hausen is particularly attracted to the possibility that cattle may play a role in some human cancers. He hypothesizes that some as-yet unidentified bovine virus, which is nononcogenic in its normal host, can become carcinogenic when transmitted to humans. Multiple lines of circumstantial evidence support this notion. For example, basal cell carcinomas are known to have a predisposition to arise in smallpox vaccination scars. Smallpox vaccines were prepared by inoculating vaccinia virus into the skin of calves and then harvesting the crusted skin, which could in theory contain contaminating bovine viruses.
Also, colorectal cancer, and to a lesser extent breast cancer and lung cancer in nonsmokers, have repeatedly been associated with beef consumption in epidemiologic studies. Countries with high consumption of goat or pork have relatively low rates of these malignancies.
The observed link between a red meat–rich diet and increased rates of colorectal and other cancers is often attributed to the formation of aromatic hydrocarbons and other known carcinogens during cooking or meat curing. But Dr. zur Hausen believes this interpretation might be inadequate. He noted that grilled, fried, and smoked poultry contain similar concentrations of these carcinogens, yet heavy consumption of poultry hasn’t been associated with an increased cancer risk.
It is of interest that the temperature achieved in the center of a piece of beef cooked rare is only 40 -50° C, yet torque teno viruses, papillomaviruses, and polyomaviruses are able to survive in a protein environment at temperatures of 80° C for 30 minutes or more.
"These are just suggestions. They do not prove anything, of course. But, for now, we can speculate that beef consumption plays a significant role in the development of colorectal cancer," Dr. zur Hausen said.
If even a small portion of these speculations turn out to be true, the implications for cancer prevention could be huge. It is estimated that if both the hepatitis B vaccine and the HPV vaccine were to be applied globally, the overall cancer burden in women could be reduced by 12%-14% and in men by 4%-5%, he noted.
He presented his theory at the World Congress of Dermatology in Seoul, South Korea.
Dr. zur Hausen declared having no financial conflicts.
Current estimates hold that 21% of all human cancers are linked to infections. But some experts believe this figure is too low and is headed substantially higher.
Fueled by the success of vaccines for the hepatitis B virus and high-risk human papillomaviruses, which offer the first-ever means of preventing specific widespread cancers by vaccination, investigators are hunting for additional infectious causes of common malignancies. The search is focused on viruses, since they are already implicated in 64% of the known infection-related cancer burden, with bacteria and parasites accounting for the remainder.
One of the world’s most celebrated oncovirus hunters is Dr. Harald zur Hausen, winner of the 2008 Nobel Prize in Medicine for his work on human papillomaviruses as the major cause of cervical cancer. He sees a number of other malignancies as being prime candidates for potential linkage to infections, including childhood lymphoblastic leukemias, basal cell carcinomas, Epstein-Barr virus–negative Hodgkin’s lymphomas, colorectal and breast cancers, and lung cancers in nonsmokers.
The TTV (torque teno virus) family shows particular promise in this regard. The TTVs, first described by Japanese investigators in 1997 (Rev. Med. Virol. 2007;17:45-57), are an extremely heterogeneous family of single-strand DNA viruses. There are well over 100 genotypes. The torque teno viruses are widespread in all human populations. They have been found in umbilical cord blood and are vertically transmitted from mother to child, even prenatally. They frequently rearrange their genomes; and transmissibility and replication have been demonstrated to occur even for small portions of the TTV genome, according to Dr. zur Hausen, professor emeritus at the German Cancer Research Center, Heidelberg.
The TTVs are known to replicate in lymphatic and bone marrow cells – the precursor cells of leukemia. In every cell line of acute lymphoblastic leukemia and Epstein-Barr virus–negative Hodgkin’s lymphoma analyzed by Dr. zur Hausen and his coworkers, they have found the same highly conserved region of TTV DNA.
Of note, TTV load is known to be reduced by interferon. This finding is consistent with the epidemiologic observation that infants who experience multiple upper respiratory infections and other infections during their first year of life seem to be protected against childhood leukemias. The hypothesis is that bursts of increased interferon production in response to multiple infections prevents levels of an interferon-sensitive putative leukemogenic agent – perhaps a TTV – from reaching critical mass.
Dr. zur Hausen is particularly attracted to the possibility that cattle may play a role in some human cancers. He hypothesizes that some as-yet unidentified bovine virus, which is nononcogenic in its normal host, can become carcinogenic when transmitted to humans. Multiple lines of circumstantial evidence support this notion. For example, basal cell carcinomas are known to have a predisposition to arise in smallpox vaccination scars. Smallpox vaccines were prepared by inoculating vaccinia virus into the skin of calves and then harvesting the crusted skin, which could in theory contain contaminating bovine viruses.
Also, colorectal cancer, and to a lesser extent breast cancer and lung cancer in nonsmokers, have repeatedly been associated with beef consumption in epidemiologic studies. Countries with high consumption of goat or pork have relatively low rates of these malignancies.
The observed link between a red meat–rich diet and increased rates of colorectal and other cancers is often attributed to the formation of aromatic hydrocarbons and other known carcinogens during cooking or meat curing. But Dr. zur Hausen believes this interpretation might be inadequate. He noted that grilled, fried, and smoked poultry contain similar concentrations of these carcinogens, yet heavy consumption of poultry hasn’t been associated with an increased cancer risk.
It is of interest that the temperature achieved in the center of a piece of beef cooked rare is only 40 -50° C, yet torque teno viruses, papillomaviruses, and polyomaviruses are able to survive in a protein environment at temperatures of 80° C for 30 minutes or more.
"These are just suggestions. They do not prove anything, of course. But, for now, we can speculate that beef consumption plays a significant role in the development of colorectal cancer," Dr. zur Hausen said.
If even a small portion of these speculations turn out to be true, the implications for cancer prevention could be huge. It is estimated that if both the hepatitis B vaccine and the HPV vaccine were to be applied globally, the overall cancer burden in women could be reduced by 12%-14% and in men by 4%-5%, he noted.
He presented his theory at the World Congress of Dermatology in Seoul, South Korea.
Dr. zur Hausen declared having no financial conflicts.
Trial Assesses SLNB's Future in Melanoma Management
NEW YORK – Sentinel lymph node biopsy continues to aid the management of about 30% of melanoma patients, though its role in the future will depend on ongoing follow-up of a major trial that started nearly 20 years ago to assess the effect of SLNB on patient survival.
But SLNB’s role could soon be upstaged by molecular profiling and the identification of specific genetic mutations that drive various melanoma subtypes, Dr. Timothy M. Johnson said at the American Academy of Dermatology’s Summer Academy Meeting.
"In the future, I hope we can do molecular profiling on melanoma and [based on that] tell patients who will benefit [from various targeted treatments] and who won’t. We are already doing this [on an investigational basis] with several new molecular profiling tests such as BRAF mutations" and treatment with vemurafenib, said Dr. Johnson, a professor of dermatology at the University of Michigan in Ann Arbor.
For the time being, melanoma patients should be counseled regarding SLNB if their tumors are at least 0.75 mm in Breslow depth and if they meet certain other criteria.
Even patients with thinner tumors should be counseled on the pros and cons of SLNB, because they will likely hear or know about the technique and may have questions. That is the approach taken at the University of Michigan and other centers, he said.
"SLN status is a powerful predictor of survival in the relatively small subset of patients where SLNB is indicated," he said. Reasons not to do SLNB include thin primary melanomas generally less than 0.75 mm with a low probability of a positive SLNB, high comorbidities, and maybe prior wide local excision, certainly in areas of ambiguous lymphatic drainage or following a local flap. This group includes about 70% of all U.S. melanoma patients. Albeit rarely, these thin lesions may be at higher risk when they have greater degrees of adverse features or positive deep margins on shave biopsy.
Melanoma patients with primary tumors that are 0.75-0.99 mm generally need an additional risk factor, such as ulceration, young age – up to about 40 years old, an increased mitotic rate, or angiolymphatic invasion to justify SLNB. When patients have additional risk factors, "we have a discussion with the patient to help them decide," he said. "We tell them the likely rate of positive lymph nodes based on our best information."
A clearer role for SLNB exists for patients with tumors with a Breslow thickness of 1 mm or greater. In these patients, exclusions from SLNB include "highly significant" medical comorbidities, a prior wide excision of the tumor, and tumors in "ambiguous" lymphatic drainage areas, such as the central back or central chest. Even in these cases, exclusion from SLNB "usually occurs but not always," Dr. Johnson noted.
Patients whose tumors have a Breslow thickness of more than 4 mm are also candidates for SLNB. In these cases, SLNB can help refine the patient’s prognosis, and may also aid the choice of adjuvant therapy.
Historically, patients with tumors 4 mm or larger were considered likely to have distant metastatic disease and a poor prognosis, and, hence, no benefit from SLNB. But Dr. Johnson and his colleagues showed a substantial difference in survival rate between patients with positive and negative SLNs even when they had thicker primary tumors.
In a series of 227 patients with melanomas at least 4 mm thick and no clinically or radiologically-apparent distant disease, patients with a positive SLNB had a 5-year overall survival rate of 47%, while patients with negative SLNs had a 5-year survival rate of 80%, a statistically significant difference (P less than .0001). The difference in 5-year distant disease-free survival rates ran slightly larger, 85% in the node-negative patients and 48% in the node-positive patients (P less than .0001) (Cancer 2009;115:5752-60). Following this study, supporting evidence has come from several other research groups.
The ongoing Multicenter Selective Lymphadenectomy Trial (MSLT) should provide more definitive evidence on the survival effects of SLNB. The interim, 5-year follow-up data from 1,327 patients at 18 worldwide centers reported in 2006 included patients with primary-tumor Breslow thicknesses of 1.2-3.5 mm. Although the study’s primary endpoint of the 5-year melanoma-specific survival rate was similar in the patients randomized to SLNB or wide local excision (87%), the prespecified secondary endpoint of 5-year disease-free survival ran 78% in patients who underwent SLNB and 73% in the wide excision group, a statistically-significant difference (P = .009) (N. Engl. J. Med. 2006;355:1307-17).
"The question is, can we identify a subset of patients with occult nodal disease with SLNB, intervene early, and improve outcomes? At face value, the third interim analysis data [the 2006 report] suggests potentially yes. However, this is a subset analysis and its validity can be debated. The data certainly support the potential for a subset benefit in node-positive patients," said Dr. Johnson. Updated survival results from a longer-term, fourth interim analysis of the MSLT survival outcomes are expected within the next year, and will help answer this question.
In addition to the disease-free survival benefit shown in the MSLT, the 30% of melanoma patients who are candidates for SLNB stand to gain useful prognostic information from the procedure. On average, about 20% of patients who are eligible for SLNB have a positive SLN, and this subgroup of patients also stand to benefit from more durable regional control of their disease with better quality of life. The survival benefit most likely occurs in a subset, perhaps 20%, of the node-positive patients, he said.
In summary, Dr. Johnson said the main reasons to consider SLNB are prognosis, durable regional control, and potential survival benefit in a select subset. These factors should be presented in "an honest discussion with the patient and family about the risks and benefits of SLNB, to help them make a best informed decision," he said.
Although many cancer centers currently use SLNB for melanoma in the way outlined by Dr. Johnson, with "not much controversy" among the "mainstream" of physicians and surgeons who treat melanoma, he noted that "there is posturing and emotion with this procedure," with controversy among some clinicians about the proper role.
"Some dermatologists don’t believe that SLNB should be used for anybody, and some surgeons overuse it on everybody. The answer [on when to use SLNB] is somewhere in between," he said in an interview. The forthcoming, longer-term follow-up data from MSLT may help clinicians how best to use SLNB most appropriately, he added.
Dr. Johnson said that he had no disclosures.
NEW YORK – Sentinel lymph node biopsy continues to aid the management of about 30% of melanoma patients, though its role in the future will depend on ongoing follow-up of a major trial that started nearly 20 years ago to assess the effect of SLNB on patient survival.
But SLNB’s role could soon be upstaged by molecular profiling and the identification of specific genetic mutations that drive various melanoma subtypes, Dr. Timothy M. Johnson said at the American Academy of Dermatology’s Summer Academy Meeting.
"In the future, I hope we can do molecular profiling on melanoma and [based on that] tell patients who will benefit [from various targeted treatments] and who won’t. We are already doing this [on an investigational basis] with several new molecular profiling tests such as BRAF mutations" and treatment with vemurafenib, said Dr. Johnson, a professor of dermatology at the University of Michigan in Ann Arbor.
For the time being, melanoma patients should be counseled regarding SLNB if their tumors are at least 0.75 mm in Breslow depth and if they meet certain other criteria.
Even patients with thinner tumors should be counseled on the pros and cons of SLNB, because they will likely hear or know about the technique and may have questions. That is the approach taken at the University of Michigan and other centers, he said.
"SLN status is a powerful predictor of survival in the relatively small subset of patients where SLNB is indicated," he said. Reasons not to do SLNB include thin primary melanomas generally less than 0.75 mm with a low probability of a positive SLNB, high comorbidities, and maybe prior wide local excision, certainly in areas of ambiguous lymphatic drainage or following a local flap. This group includes about 70% of all U.S. melanoma patients. Albeit rarely, these thin lesions may be at higher risk when they have greater degrees of adverse features or positive deep margins on shave biopsy.
Melanoma patients with primary tumors that are 0.75-0.99 mm generally need an additional risk factor, such as ulceration, young age – up to about 40 years old, an increased mitotic rate, or angiolymphatic invasion to justify SLNB. When patients have additional risk factors, "we have a discussion with the patient to help them decide," he said. "We tell them the likely rate of positive lymph nodes based on our best information."
A clearer role for SLNB exists for patients with tumors with a Breslow thickness of 1 mm or greater. In these patients, exclusions from SLNB include "highly significant" medical comorbidities, a prior wide excision of the tumor, and tumors in "ambiguous" lymphatic drainage areas, such as the central back or central chest. Even in these cases, exclusion from SLNB "usually occurs but not always," Dr. Johnson noted.
Patients whose tumors have a Breslow thickness of more than 4 mm are also candidates for SLNB. In these cases, SLNB can help refine the patient’s prognosis, and may also aid the choice of adjuvant therapy.
Historically, patients with tumors 4 mm or larger were considered likely to have distant metastatic disease and a poor prognosis, and, hence, no benefit from SLNB. But Dr. Johnson and his colleagues showed a substantial difference in survival rate between patients with positive and negative SLNs even when they had thicker primary tumors.
In a series of 227 patients with melanomas at least 4 mm thick and no clinically or radiologically-apparent distant disease, patients with a positive SLNB had a 5-year overall survival rate of 47%, while patients with negative SLNs had a 5-year survival rate of 80%, a statistically significant difference (P less than .0001). The difference in 5-year distant disease-free survival rates ran slightly larger, 85% in the node-negative patients and 48% in the node-positive patients (P less than .0001) (Cancer 2009;115:5752-60). Following this study, supporting evidence has come from several other research groups.
The ongoing Multicenter Selective Lymphadenectomy Trial (MSLT) should provide more definitive evidence on the survival effects of SLNB. The interim, 5-year follow-up data from 1,327 patients at 18 worldwide centers reported in 2006 included patients with primary-tumor Breslow thicknesses of 1.2-3.5 mm. Although the study’s primary endpoint of the 5-year melanoma-specific survival rate was similar in the patients randomized to SLNB or wide local excision (87%), the prespecified secondary endpoint of 5-year disease-free survival ran 78% in patients who underwent SLNB and 73% in the wide excision group, a statistically-significant difference (P = .009) (N. Engl. J. Med. 2006;355:1307-17).
"The question is, can we identify a subset of patients with occult nodal disease with SLNB, intervene early, and improve outcomes? At face value, the third interim analysis data [the 2006 report] suggests potentially yes. However, this is a subset analysis and its validity can be debated. The data certainly support the potential for a subset benefit in node-positive patients," said Dr. Johnson. Updated survival results from a longer-term, fourth interim analysis of the MSLT survival outcomes are expected within the next year, and will help answer this question.
In addition to the disease-free survival benefit shown in the MSLT, the 30% of melanoma patients who are candidates for SLNB stand to gain useful prognostic information from the procedure. On average, about 20% of patients who are eligible for SLNB have a positive SLN, and this subgroup of patients also stand to benefit from more durable regional control of their disease with better quality of life. The survival benefit most likely occurs in a subset, perhaps 20%, of the node-positive patients, he said.
In summary, Dr. Johnson said the main reasons to consider SLNB are prognosis, durable regional control, and potential survival benefit in a select subset. These factors should be presented in "an honest discussion with the patient and family about the risks and benefits of SLNB, to help them make a best informed decision," he said.
Although many cancer centers currently use SLNB for melanoma in the way outlined by Dr. Johnson, with "not much controversy" among the "mainstream" of physicians and surgeons who treat melanoma, he noted that "there is posturing and emotion with this procedure," with controversy among some clinicians about the proper role.
"Some dermatologists don’t believe that SLNB should be used for anybody, and some surgeons overuse it on everybody. The answer [on when to use SLNB] is somewhere in between," he said in an interview. The forthcoming, longer-term follow-up data from MSLT may help clinicians how best to use SLNB most appropriately, he added.
Dr. Johnson said that he had no disclosures.
NEW YORK – Sentinel lymph node biopsy continues to aid the management of about 30% of melanoma patients, though its role in the future will depend on ongoing follow-up of a major trial that started nearly 20 years ago to assess the effect of SLNB on patient survival.
But SLNB’s role could soon be upstaged by molecular profiling and the identification of specific genetic mutations that drive various melanoma subtypes, Dr. Timothy M. Johnson said at the American Academy of Dermatology’s Summer Academy Meeting.
"In the future, I hope we can do molecular profiling on melanoma and [based on that] tell patients who will benefit [from various targeted treatments] and who won’t. We are already doing this [on an investigational basis] with several new molecular profiling tests such as BRAF mutations" and treatment with vemurafenib, said Dr. Johnson, a professor of dermatology at the University of Michigan in Ann Arbor.
For the time being, melanoma patients should be counseled regarding SLNB if their tumors are at least 0.75 mm in Breslow depth and if they meet certain other criteria.
Even patients with thinner tumors should be counseled on the pros and cons of SLNB, because they will likely hear or know about the technique and may have questions. That is the approach taken at the University of Michigan and other centers, he said.
"SLN status is a powerful predictor of survival in the relatively small subset of patients where SLNB is indicated," he said. Reasons not to do SLNB include thin primary melanomas generally less than 0.75 mm with a low probability of a positive SLNB, high comorbidities, and maybe prior wide local excision, certainly in areas of ambiguous lymphatic drainage or following a local flap. This group includes about 70% of all U.S. melanoma patients. Albeit rarely, these thin lesions may be at higher risk when they have greater degrees of adverse features or positive deep margins on shave biopsy.
Melanoma patients with primary tumors that are 0.75-0.99 mm generally need an additional risk factor, such as ulceration, young age – up to about 40 years old, an increased mitotic rate, or angiolymphatic invasion to justify SLNB. When patients have additional risk factors, "we have a discussion with the patient to help them decide," he said. "We tell them the likely rate of positive lymph nodes based on our best information."
A clearer role for SLNB exists for patients with tumors with a Breslow thickness of 1 mm or greater. In these patients, exclusions from SLNB include "highly significant" medical comorbidities, a prior wide excision of the tumor, and tumors in "ambiguous" lymphatic drainage areas, such as the central back or central chest. Even in these cases, exclusion from SLNB "usually occurs but not always," Dr. Johnson noted.
Patients whose tumors have a Breslow thickness of more than 4 mm are also candidates for SLNB. In these cases, SLNB can help refine the patient’s prognosis, and may also aid the choice of adjuvant therapy.
Historically, patients with tumors 4 mm or larger were considered likely to have distant metastatic disease and a poor prognosis, and, hence, no benefit from SLNB. But Dr. Johnson and his colleagues showed a substantial difference in survival rate between patients with positive and negative SLNs even when they had thicker primary tumors.
In a series of 227 patients with melanomas at least 4 mm thick and no clinically or radiologically-apparent distant disease, patients with a positive SLNB had a 5-year overall survival rate of 47%, while patients with negative SLNs had a 5-year survival rate of 80%, a statistically significant difference (P less than .0001). The difference in 5-year distant disease-free survival rates ran slightly larger, 85% in the node-negative patients and 48% in the node-positive patients (P less than .0001) (Cancer 2009;115:5752-60). Following this study, supporting evidence has come from several other research groups.
The ongoing Multicenter Selective Lymphadenectomy Trial (MSLT) should provide more definitive evidence on the survival effects of SLNB. The interim, 5-year follow-up data from 1,327 patients at 18 worldwide centers reported in 2006 included patients with primary-tumor Breslow thicknesses of 1.2-3.5 mm. Although the study’s primary endpoint of the 5-year melanoma-specific survival rate was similar in the patients randomized to SLNB or wide local excision (87%), the prespecified secondary endpoint of 5-year disease-free survival ran 78% in patients who underwent SLNB and 73% in the wide excision group, a statistically-significant difference (P = .009) (N. Engl. J. Med. 2006;355:1307-17).
"The question is, can we identify a subset of patients with occult nodal disease with SLNB, intervene early, and improve outcomes? At face value, the third interim analysis data [the 2006 report] suggests potentially yes. However, this is a subset analysis and its validity can be debated. The data certainly support the potential for a subset benefit in node-positive patients," said Dr. Johnson. Updated survival results from a longer-term, fourth interim analysis of the MSLT survival outcomes are expected within the next year, and will help answer this question.
In addition to the disease-free survival benefit shown in the MSLT, the 30% of melanoma patients who are candidates for SLNB stand to gain useful prognostic information from the procedure. On average, about 20% of patients who are eligible for SLNB have a positive SLN, and this subgroup of patients also stand to benefit from more durable regional control of their disease with better quality of life. The survival benefit most likely occurs in a subset, perhaps 20%, of the node-positive patients, he said.
In summary, Dr. Johnson said the main reasons to consider SLNB are prognosis, durable regional control, and potential survival benefit in a select subset. These factors should be presented in "an honest discussion with the patient and family about the risks and benefits of SLNB, to help them make a best informed decision," he said.
Although many cancer centers currently use SLNB for melanoma in the way outlined by Dr. Johnson, with "not much controversy" among the "mainstream" of physicians and surgeons who treat melanoma, he noted that "there is posturing and emotion with this procedure," with controversy among some clinicians about the proper role.
"Some dermatologists don’t believe that SLNB should be used for anybody, and some surgeons overuse it on everybody. The answer [on when to use SLNB] is somewhere in between," he said in an interview. The forthcoming, longer-term follow-up data from MSLT may help clinicians how best to use SLNB most appropriately, he added.
Dr. Johnson said that he had no disclosures.
EXPERT ANALYSIS FROM THE AMERICAN ACADEMY OF DERMATOLOGY'S SUMMER ACADEMY MEETING
The Fire Ant's Northward March: The Skinny Podcast
In this month's Skinny Podcast, we catch up with Dr. Richard Carvajal for the latest on the FDA's recent approval of vemurafenib for advanced-stage melanoma.
We also talk to Dr. Henry Lim about why the thinks adults and children (starting at age 1) should be taking regular vitamin D supplements.
Dr. Ronald Rapini discusses the northward march of the fire ant. His stories are sure to make your skin crawl.
In this month's Cosmetic Counter segment, Dr. Lily Talakoub lays out the facts about cleansers and offers tips that you can share with your patients.
And last but not least, Dr. Alan Rockoff entertains us with a story about a bath tub and an angry mother-in-law.
In this month's Skinny Podcast, we catch up with Dr. Richard Carvajal for the latest on the FDA's recent approval of vemurafenib for advanced-stage melanoma.
We also talk to Dr. Henry Lim about why the thinks adults and children (starting at age 1) should be taking regular vitamin D supplements.
Dr. Ronald Rapini discusses the northward march of the fire ant. His stories are sure to make your skin crawl.
In this month's Cosmetic Counter segment, Dr. Lily Talakoub lays out the facts about cleansers and offers tips that you can share with your patients.
And last but not least, Dr. Alan Rockoff entertains us with a story about a bath tub and an angry mother-in-law.
In this month's Skinny Podcast, we catch up with Dr. Richard Carvajal for the latest on the FDA's recent approval of vemurafenib for advanced-stage melanoma.
We also talk to Dr. Henry Lim about why the thinks adults and children (starting at age 1) should be taking regular vitamin D supplements.
Dr. Ronald Rapini discusses the northward march of the fire ant. His stories are sure to make your skin crawl.
In this month's Cosmetic Counter segment, Dr. Lily Talakoub lays out the facts about cleansers and offers tips that you can share with your patients.
And last but not least, Dr. Alan Rockoff entertains us with a story about a bath tub and an angry mother-in-law.
Personalized medicine: myth to reality
Ever since the International Human GenomeSequencing Consortium unveiled its “working draft” of the human genome sequence in 2000, the scientific community has eagerly discussed and speculated on the potential of genomic medicine. Patients, clinicians, and scientists were—and still are—excited about the possibilities of targeted therapies such as imatinib (Gleevec) for chronic myeloid leukemias and gastrointestinal stromal tumors and trastuzumab (Herceptin) for HER2/neu-positive breast cancer. There has been significant progress in the development of these and other targeted therapies, and they remain part of an intriguing and promising work in progress. However, for patients with lung cancer and melanoma, both of which are highly refractory diseases, the therapeutic choices other than chemotherapy have been limited and the commensurate outcomes discouraging.
In the early 2000s, we were excited about targeted therapies such as gefitinib (Iressa), which blocks epidermal growth factor receptor (EGFR)- tyrosine kinase activity in non-small cell lung cancer (NSCLC), but its clinical benefit was still limited. We have since gained a better understanding of lung cancer as a molecularly heterogeneous disease and have adjusted our approach to its treatment, based on new data showing that all lung cancer patients cannot be treated with the same drug regimen and achieve the same outcomes. With those insights, the implications of targeted therapies came into sharper focus in 2004 with the US Food and Drug Administration (FDA) approval of erlotinib (Tarceva) for the 10%–15% of patients with NSCLC (adenocarcinoma) who have the EGFR gene mutation. At around the same time, scientists identified BRAF gene mutations in about 40%– 60% of patients with melanoma, and the quest for therapies for that disease was redirected to the cellular level as well. ...
* For a PDF of the full article, click in the link to the left of this introduction.
Ever since the International Human GenomeSequencing Consortium unveiled its “working draft” of the human genome sequence in 2000, the scientific community has eagerly discussed and speculated on the potential of genomic medicine. Patients, clinicians, and scientists were—and still are—excited about the possibilities of targeted therapies such as imatinib (Gleevec) for chronic myeloid leukemias and gastrointestinal stromal tumors and trastuzumab (Herceptin) for HER2/neu-positive breast cancer. There has been significant progress in the development of these and other targeted therapies, and they remain part of an intriguing and promising work in progress. However, for patients with lung cancer and melanoma, both of which are highly refractory diseases, the therapeutic choices other than chemotherapy have been limited and the commensurate outcomes discouraging.
In the early 2000s, we were excited about targeted therapies such as gefitinib (Iressa), which blocks epidermal growth factor receptor (EGFR)- tyrosine kinase activity in non-small cell lung cancer (NSCLC), but its clinical benefit was still limited. We have since gained a better understanding of lung cancer as a molecularly heterogeneous disease and have adjusted our approach to its treatment, based on new data showing that all lung cancer patients cannot be treated with the same drug regimen and achieve the same outcomes. With those insights, the implications of targeted therapies came into sharper focus in 2004 with the US Food and Drug Administration (FDA) approval of erlotinib (Tarceva) for the 10%–15% of patients with NSCLC (adenocarcinoma) who have the EGFR gene mutation. At around the same time, scientists identified BRAF gene mutations in about 40%– 60% of patients with melanoma, and the quest for therapies for that disease was redirected to the cellular level as well. ...
* For a PDF of the full article, click in the link to the left of this introduction.
Ever since the International Human GenomeSequencing Consortium unveiled its “working draft” of the human genome sequence in 2000, the scientific community has eagerly discussed and speculated on the potential of genomic medicine. Patients, clinicians, and scientists were—and still are—excited about the possibilities of targeted therapies such as imatinib (Gleevec) for chronic myeloid leukemias and gastrointestinal stromal tumors and trastuzumab (Herceptin) for HER2/neu-positive breast cancer. There has been significant progress in the development of these and other targeted therapies, and they remain part of an intriguing and promising work in progress. However, for patients with lung cancer and melanoma, both of which are highly refractory diseases, the therapeutic choices other than chemotherapy have been limited and the commensurate outcomes discouraging.
In the early 2000s, we were excited about targeted therapies such as gefitinib (Iressa), which blocks epidermal growth factor receptor (EGFR)- tyrosine kinase activity in non-small cell lung cancer (NSCLC), but its clinical benefit was still limited. We have since gained a better understanding of lung cancer as a molecularly heterogeneous disease and have adjusted our approach to its treatment, based on new data showing that all lung cancer patients cannot be treated with the same drug regimen and achieve the same outcomes. With those insights, the implications of targeted therapies came into sharper focus in 2004 with the US Food and Drug Administration (FDA) approval of erlotinib (Tarceva) for the 10%–15% of patients with NSCLC (adenocarcinoma) who have the EGFR gene mutation. At around the same time, scientists identified BRAF gene mutations in about 40%– 60% of patients with melanoma, and the quest for therapies for that disease was redirected to the cellular level as well. ...
* For a PDF of the full article, click in the link to the left of this introduction.
Prom Tanning in '90s Behind Current Spike in Melanoma
NEW YORK – Teenage girls who used tanning beds in the 1990s are behind the sharp increase in melanoma in young women, according to Dr. Darrell S. Rigel.
The rise in melanoma from this often prom-driven surge in tanning has shown up as increased cases in women aged 25-34 years, Dr. Rigel said at the American Academy of Dermatology’s Summer Academy meeting.
"All the melanoma I see in women in their 20s and 30s, virtually every one of them has gone to a tanning salon, and we’re seeing a lot more" melanoma in women in this age group, said Dr. Rigel, a dermatologist at New York University. In addition, the primary melanomas seen in young women "often occur where the sun doesn’t shine but the tanning beds do," on breasts and near the genitalia, he said. "There definitely is a causal relationship."
Documentation of the rising rate of melanoma in young women includes data collected by the National Cancer Institute’s SEER (Surveillance, Epidemiology, and End Results) program. Data collected by SEER for U.S. melanoma incidence rates showed that during 2004-2006, white women aged 25-29 had a 14.3 per 100,000 incidence of melanoma, a 4% absolute and 42% relative jump from the rate in similarly aged women in 1994-1996. Among white women aged 30-34, the 2004-2006 SEER rate was 16.4 per 100,000 cases, also a 4% absolute and 32% relative jump from the rate in 1994-1996.
Recent increases in melanoma incidence among young white women have been "especially rapid," according to comments published earlier this year (J. Natl. Cancer Inst. 2011;103:171). Since 1995, the annual incidence of melanoma among young, white U.S. women rose by an average of 3.8%, compared with each preceding year.
Increased melanoma incidence since the 1990s links with the greater popularity of tanning beds among teens that first reached high levels in the 1990s. The first commercial U.S. tanning salon opened in 1978, and today there are about 60,000, with about 1 million Americans using a tanning bed daily and about 28 million using a tanning bed at least once each year, Dr. Rigel said. Women patronize tanning salons about sevenfold times more often than men, and other statistics show that more than a third of white, U.S. teens aged 17-18 have had a tanning session at least once in the prior year, he said.
"Major marketing is targeted at these women prior to proms," Dr. Rigel said in an interview. Young women aged 17-18 are "particularly susceptible," to the effects of artificial UV tanning radiation in the development of skin cancer. "This will take a broad sea change. We need models who are not tan in magazines aimed at young women. The pale look has to be in. It’s still cool to be tan. We’re working against that, and against their not being worried about skin cancer, and about how they’ll look at age 50. It’s just not there."
The spike in cases appearing in women once they have reached 25 years or older "is exactly when you’d expect to see the melanomas" based on high-level exposure at age 17-18, he added. The latency period from the time of intensified exposure to the appearance of melanoma is 5-20 years, he said.
"Twenty years ago, it was rare to see a woman in her 20s with melanoma, and we also did not see a lot among women in their 30s. Now, we commonly see cases in women in their 20s, and every one of them has a tanning history. The foremost issue for melanoma in women is tanning beds. For the first time, we’re seeing an increased incidence of melanoma in young women in their 20s and 30s, and the only thing they appear to do differently than young men is go to tanning salons."
Primary care physicians who see women in their 20s and 30s should examine their skin for signs of melanoma when they have the chance during physical examination, and should ask about a history of tanning-bed use. Results from several studies show that even a single tanning episode can significantly increase the risk for melanoma, so all a physician has to ask is, "Have you ever gone to a tanning salon?" to know whether a patient has an increased melanoma risk.
In addition to increased awareness, physicians need to educate teenage girls and the general public about the danger from tanning beds. "It’s very analogous to cigarette smoking. All we can do is get the word out, and hope that people do less harm to themselves."
Dr. Rigel said that he had no relevant disclosures.
NEW YORK – Teenage girls who used tanning beds in the 1990s are behind the sharp increase in melanoma in young women, according to Dr. Darrell S. Rigel.
The rise in melanoma from this often prom-driven surge in tanning has shown up as increased cases in women aged 25-34 years, Dr. Rigel said at the American Academy of Dermatology’s Summer Academy meeting.
"All the melanoma I see in women in their 20s and 30s, virtually every one of them has gone to a tanning salon, and we’re seeing a lot more" melanoma in women in this age group, said Dr. Rigel, a dermatologist at New York University. In addition, the primary melanomas seen in young women "often occur where the sun doesn’t shine but the tanning beds do," on breasts and near the genitalia, he said. "There definitely is a causal relationship."
Documentation of the rising rate of melanoma in young women includes data collected by the National Cancer Institute’s SEER (Surveillance, Epidemiology, and End Results) program. Data collected by SEER for U.S. melanoma incidence rates showed that during 2004-2006, white women aged 25-29 had a 14.3 per 100,000 incidence of melanoma, a 4% absolute and 42% relative jump from the rate in similarly aged women in 1994-1996. Among white women aged 30-34, the 2004-2006 SEER rate was 16.4 per 100,000 cases, also a 4% absolute and 32% relative jump from the rate in 1994-1996.
Recent increases in melanoma incidence among young white women have been "especially rapid," according to comments published earlier this year (J. Natl. Cancer Inst. 2011;103:171). Since 1995, the annual incidence of melanoma among young, white U.S. women rose by an average of 3.8%, compared with each preceding year.
Increased melanoma incidence since the 1990s links with the greater popularity of tanning beds among teens that first reached high levels in the 1990s. The first commercial U.S. tanning salon opened in 1978, and today there are about 60,000, with about 1 million Americans using a tanning bed daily and about 28 million using a tanning bed at least once each year, Dr. Rigel said. Women patronize tanning salons about sevenfold times more often than men, and other statistics show that more than a third of white, U.S. teens aged 17-18 have had a tanning session at least once in the prior year, he said.
"Major marketing is targeted at these women prior to proms," Dr. Rigel said in an interview. Young women aged 17-18 are "particularly susceptible," to the effects of artificial UV tanning radiation in the development of skin cancer. "This will take a broad sea change. We need models who are not tan in magazines aimed at young women. The pale look has to be in. It’s still cool to be tan. We’re working against that, and against their not being worried about skin cancer, and about how they’ll look at age 50. It’s just not there."
The spike in cases appearing in women once they have reached 25 years or older "is exactly when you’d expect to see the melanomas" based on high-level exposure at age 17-18, he added. The latency period from the time of intensified exposure to the appearance of melanoma is 5-20 years, he said.
"Twenty years ago, it was rare to see a woman in her 20s with melanoma, and we also did not see a lot among women in their 30s. Now, we commonly see cases in women in their 20s, and every one of them has a tanning history. The foremost issue for melanoma in women is tanning beds. For the first time, we’re seeing an increased incidence of melanoma in young women in their 20s and 30s, and the only thing they appear to do differently than young men is go to tanning salons."
Primary care physicians who see women in their 20s and 30s should examine their skin for signs of melanoma when they have the chance during physical examination, and should ask about a history of tanning-bed use. Results from several studies show that even a single tanning episode can significantly increase the risk for melanoma, so all a physician has to ask is, "Have you ever gone to a tanning salon?" to know whether a patient has an increased melanoma risk.
In addition to increased awareness, physicians need to educate teenage girls and the general public about the danger from tanning beds. "It’s very analogous to cigarette smoking. All we can do is get the word out, and hope that people do less harm to themselves."
Dr. Rigel said that he had no relevant disclosures.
NEW YORK – Teenage girls who used tanning beds in the 1990s are behind the sharp increase in melanoma in young women, according to Dr. Darrell S. Rigel.
The rise in melanoma from this often prom-driven surge in tanning has shown up as increased cases in women aged 25-34 years, Dr. Rigel said at the American Academy of Dermatology’s Summer Academy meeting.
"All the melanoma I see in women in their 20s and 30s, virtually every one of them has gone to a tanning salon, and we’re seeing a lot more" melanoma in women in this age group, said Dr. Rigel, a dermatologist at New York University. In addition, the primary melanomas seen in young women "often occur where the sun doesn’t shine but the tanning beds do," on breasts and near the genitalia, he said. "There definitely is a causal relationship."
Documentation of the rising rate of melanoma in young women includes data collected by the National Cancer Institute’s SEER (Surveillance, Epidemiology, and End Results) program. Data collected by SEER for U.S. melanoma incidence rates showed that during 2004-2006, white women aged 25-29 had a 14.3 per 100,000 incidence of melanoma, a 4% absolute and 42% relative jump from the rate in similarly aged women in 1994-1996. Among white women aged 30-34, the 2004-2006 SEER rate was 16.4 per 100,000 cases, also a 4% absolute and 32% relative jump from the rate in 1994-1996.
Recent increases in melanoma incidence among young white women have been "especially rapid," according to comments published earlier this year (J. Natl. Cancer Inst. 2011;103:171). Since 1995, the annual incidence of melanoma among young, white U.S. women rose by an average of 3.8%, compared with each preceding year.
Increased melanoma incidence since the 1990s links with the greater popularity of tanning beds among teens that first reached high levels in the 1990s. The first commercial U.S. tanning salon opened in 1978, and today there are about 60,000, with about 1 million Americans using a tanning bed daily and about 28 million using a tanning bed at least once each year, Dr. Rigel said. Women patronize tanning salons about sevenfold times more often than men, and other statistics show that more than a third of white, U.S. teens aged 17-18 have had a tanning session at least once in the prior year, he said.
"Major marketing is targeted at these women prior to proms," Dr. Rigel said in an interview. Young women aged 17-18 are "particularly susceptible," to the effects of artificial UV tanning radiation in the development of skin cancer. "This will take a broad sea change. We need models who are not tan in magazines aimed at young women. The pale look has to be in. It’s still cool to be tan. We’re working against that, and against their not being worried about skin cancer, and about how they’ll look at age 50. It’s just not there."
The spike in cases appearing in women once they have reached 25 years or older "is exactly when you’d expect to see the melanomas" based on high-level exposure at age 17-18, he added. The latency period from the time of intensified exposure to the appearance of melanoma is 5-20 years, he said.
"Twenty years ago, it was rare to see a woman in her 20s with melanoma, and we also did not see a lot among women in their 30s. Now, we commonly see cases in women in their 20s, and every one of them has a tanning history. The foremost issue for melanoma in women is tanning beds. For the first time, we’re seeing an increased incidence of melanoma in young women in their 20s and 30s, and the only thing they appear to do differently than young men is go to tanning salons."
Primary care physicians who see women in their 20s and 30s should examine their skin for signs of melanoma when they have the chance during physical examination, and should ask about a history of tanning-bed use. Results from several studies show that even a single tanning episode can significantly increase the risk for melanoma, so all a physician has to ask is, "Have you ever gone to a tanning salon?" to know whether a patient has an increased melanoma risk.
In addition to increased awareness, physicians need to educate teenage girls and the general public about the danger from tanning beds. "It’s very analogous to cigarette smoking. All we can do is get the word out, and hope that people do less harm to themselves."
Dr. Rigel said that he had no relevant disclosures.
EXPERT ANALYSIS FROM THE AMERICAN ACADEMY OF DERMATOLOGY'S SUMMER ACADEMY MEETING
FDA Approves Vemurafenib for Advanced Melanoma
The Food and Drug Administration announced on August 17 the approval of vemurafenib, a highly anticipated metastatic melanoma therapy that targets the BRAF V600E mutation found in 40%-60% of patients.
The agency also approved the cobas 4800 BRAF V600 Mutation Test, a companion diagnostic test designed to help determine if a patient’s melanoma cells carry the BRAF V600E mutation.
Vemurafenib is the second therapy to prolong the lives of patients with metastatic melanoma. The first, a slow-acting immunotherapy called ipilimumab (Yervoy), was approved in March 2011.
Clinical trials have shown that vemurafenib (better known as PLX4032) can produce rapid remission in patients at risk of death from metastatic melanoma.
Vemurafenib is not a panacea, as only about half of patients with BRAF V600E mutations have responded, and in the early studies almost all eventually relapsed. However, the drug has produced delays in recurrence and prolongations of overall survival that are clinically and statistically significant.
"This is a bright day for many melanoma patients," Dr. Jeffrey A. Sosman, a professor of medicine at Vanderbilt-Ingram Cancer Center in Nashville said in response to the FDA announcement.
"But it is only the start, and we have to continue to enroll patients onto clinical trials in order to build upon this exciting advance. It is only the beginning," added Dr. Sosman, an investigator in the BRIM-3 trial that led to approval of vemurafenib.
Early results of the pivotal phase III BRIM-3 trial, presented at the American Society of Clinical Oncology (ASCO) in June, and published simultaneously in the New England Journal of Medicine (2011;364:2507-16) , showed that vemurafenib reduced the relative risk of death by 63% in comparison with dacarbazine (DTIC), a standard but ineffective therapy, at a median follow-up of 3 months.
The randomized, open-label study screened 2,107 patients with unresectable stage III or IV melanoma for BRAF mutations, which were found in 47% of patients. It accrued 675 patients from January 2010 though December 2010, but some had not been in the study long enough to be included in the reports, as the trial was stopped based on early positive results.
Among the findings were estimates that 84% of patients treated with oral vemurafenib but only 64% of those given DTIC would be alive at 6 months. Vemurafenib’s hazard ratio for death was 0.37 (P less than .0001). An analysis of progression-free survival showed a 74% reduction in the risk of progression (HR 0.26, P less than .0001).
Median time to progression was 5.3 months with vemurafenib versus 1.6 months with DTIC, Dr. Paul Chapman of Memorial Sloan-Kettering Cancer Center in New York City and colleagues reported. Median overall survival had not been reached in the vemurafenib arm of BRIM-3, but Dr. Chapman expressed optimism that the advantage would hold up over time, as the Kaplan Meier survival curve mirrored results from the precursor BRIM-2 trial.
In that phase II study, median overall survival also had not yet been reached with 77% of patients alive at 6 months and 58% at 12 months, Dr. Antoni Ribas of the Jonsson Comprehensive Cancer Center in Los Angeles reported at the ASCO meeting. His presentation was based on a median follow-up of 7 months with an overall response rate of 53%.
The investigators also looked at patients who had BRAF mutations other than V600E, and were encouraged to find about half responded to vemurafenib, Dr. Chapman said in an interview.
In yet another presentation at ASCO, Dr. Alexander M. Menzies of the Melanoma Institute Australia reported that BRAF mutations are more common in younger melanoma patients. In all, 46% of 311 advanced melanoma patients had a BRAF mutation; 73% of these were V600E mutations, 19% V600K, and the remainder other forms.
The mutation rate declined from more than 80% of melanoma patients aged 20-40 years to 50% of those aged 41-70 years, and to less than 25% of those older than 70 years, Dr. Menzies said. The V600E mutation predominated in younger patients, occurring in 86% of those aged 20-30 years, whereas the V600K mutation emerged in older age.
Vemurafenib - approved at a recommended dose of 960 mg orally twice daily - has a manageable side effect profile, according to the BRIM-3 investigators. Although the safety data were not formally pooled, investigators said less than 10% of patients on active therapy had a grade 3 or higher toxicity. Joint pain, photosensitivity, and skin rashes were the most common. About 20% of patients developed cutaneous squamous cell carcinoma, but it was easily removed by dermatologists, and there were no instances of metastases. Only 6% of patients on vemurafenib discontinued because of adverse events.
Vemurafenib will be marketed as Zelboraf by Roche, which announced submission of applications for approval in the United States and Europe on May 11. Roche’s Genentech has a comarketing agreement with vemurafenib developer Plexxikon, a member of Daiichi Sankyo.
In June, Roche and Bristol-Meyers Squibb (maker of ipilimumab), announced that they would collaborate on a phase I/II study to determine whether combining the two drugs is safe and effective in patients with BRAF mutations.
The cobas 4800 BRAF V600 Mutation Test is manufactured by Roche Molecular Systems in Pleasanton, Calif.
Genentech sponsored the BRIM-2 and BRIM-3 trials. Dr. Chapman reported consultant/advisory roles and research funding from its parent company Roche. Some coauthors were Roche employees, and others reported similar relationships with Genentech. Dr. Ribas and several of his coauthors reported similar consulting and advisory relationships with Roche/Genentech. Dr. Menzies had no relevant financial conflicts of interest.
The Food and Drug Administration announced on August 17 the approval of vemurafenib, a highly anticipated metastatic melanoma therapy that targets the BRAF V600E mutation found in 40%-60% of patients.
The agency also approved the cobas 4800 BRAF V600 Mutation Test, a companion diagnostic test designed to help determine if a patient’s melanoma cells carry the BRAF V600E mutation.
Vemurafenib is the second therapy to prolong the lives of patients with metastatic melanoma. The first, a slow-acting immunotherapy called ipilimumab (Yervoy), was approved in March 2011.
Clinical trials have shown that vemurafenib (better known as PLX4032) can produce rapid remission in patients at risk of death from metastatic melanoma.
Vemurafenib is not a panacea, as only about half of patients with BRAF V600E mutations have responded, and in the early studies almost all eventually relapsed. However, the drug has produced delays in recurrence and prolongations of overall survival that are clinically and statistically significant.
"This is a bright day for many melanoma patients," Dr. Jeffrey A. Sosman, a professor of medicine at Vanderbilt-Ingram Cancer Center in Nashville said in response to the FDA announcement.
"But it is only the start, and we have to continue to enroll patients onto clinical trials in order to build upon this exciting advance. It is only the beginning," added Dr. Sosman, an investigator in the BRIM-3 trial that led to approval of vemurafenib.
Early results of the pivotal phase III BRIM-3 trial, presented at the American Society of Clinical Oncology (ASCO) in June, and published simultaneously in the New England Journal of Medicine (2011;364:2507-16) , showed that vemurafenib reduced the relative risk of death by 63% in comparison with dacarbazine (DTIC), a standard but ineffective therapy, at a median follow-up of 3 months.
The randomized, open-label study screened 2,107 patients with unresectable stage III or IV melanoma for BRAF mutations, which were found in 47% of patients. It accrued 675 patients from January 2010 though December 2010, but some had not been in the study long enough to be included in the reports, as the trial was stopped based on early positive results.
Among the findings were estimates that 84% of patients treated with oral vemurafenib but only 64% of those given DTIC would be alive at 6 months. Vemurafenib’s hazard ratio for death was 0.37 (P less than .0001). An analysis of progression-free survival showed a 74% reduction in the risk of progression (HR 0.26, P less than .0001).
Median time to progression was 5.3 months with vemurafenib versus 1.6 months with DTIC, Dr. Paul Chapman of Memorial Sloan-Kettering Cancer Center in New York City and colleagues reported. Median overall survival had not been reached in the vemurafenib arm of BRIM-3, but Dr. Chapman expressed optimism that the advantage would hold up over time, as the Kaplan Meier survival curve mirrored results from the precursor BRIM-2 trial.
In that phase II study, median overall survival also had not yet been reached with 77% of patients alive at 6 months and 58% at 12 months, Dr. Antoni Ribas of the Jonsson Comprehensive Cancer Center in Los Angeles reported at the ASCO meeting. His presentation was based on a median follow-up of 7 months with an overall response rate of 53%.
The investigators also looked at patients who had BRAF mutations other than V600E, and were encouraged to find about half responded to vemurafenib, Dr. Chapman said in an interview.
In yet another presentation at ASCO, Dr. Alexander M. Menzies of the Melanoma Institute Australia reported that BRAF mutations are more common in younger melanoma patients. In all, 46% of 311 advanced melanoma patients had a BRAF mutation; 73% of these were V600E mutations, 19% V600K, and the remainder other forms.
The mutation rate declined from more than 80% of melanoma patients aged 20-40 years to 50% of those aged 41-70 years, and to less than 25% of those older than 70 years, Dr. Menzies said. The V600E mutation predominated in younger patients, occurring in 86% of those aged 20-30 years, whereas the V600K mutation emerged in older age.
Vemurafenib - approved at a recommended dose of 960 mg orally twice daily - has a manageable side effect profile, according to the BRIM-3 investigators. Although the safety data were not formally pooled, investigators said less than 10% of patients on active therapy had a grade 3 or higher toxicity. Joint pain, photosensitivity, and skin rashes were the most common. About 20% of patients developed cutaneous squamous cell carcinoma, but it was easily removed by dermatologists, and there were no instances of metastases. Only 6% of patients on vemurafenib discontinued because of adverse events.
Vemurafenib will be marketed as Zelboraf by Roche, which announced submission of applications for approval in the United States and Europe on May 11. Roche’s Genentech has a comarketing agreement with vemurafenib developer Plexxikon, a member of Daiichi Sankyo.
In June, Roche and Bristol-Meyers Squibb (maker of ipilimumab), announced that they would collaborate on a phase I/II study to determine whether combining the two drugs is safe and effective in patients with BRAF mutations.
The cobas 4800 BRAF V600 Mutation Test is manufactured by Roche Molecular Systems in Pleasanton, Calif.
Genentech sponsored the BRIM-2 and BRIM-3 trials. Dr. Chapman reported consultant/advisory roles and research funding from its parent company Roche. Some coauthors were Roche employees, and others reported similar relationships with Genentech. Dr. Ribas and several of his coauthors reported similar consulting and advisory relationships with Roche/Genentech. Dr. Menzies had no relevant financial conflicts of interest.
The Food and Drug Administration announced on August 17 the approval of vemurafenib, a highly anticipated metastatic melanoma therapy that targets the BRAF V600E mutation found in 40%-60% of patients.
The agency also approved the cobas 4800 BRAF V600 Mutation Test, a companion diagnostic test designed to help determine if a patient’s melanoma cells carry the BRAF V600E mutation.
Vemurafenib is the second therapy to prolong the lives of patients with metastatic melanoma. The first, a slow-acting immunotherapy called ipilimumab (Yervoy), was approved in March 2011.
Clinical trials have shown that vemurafenib (better known as PLX4032) can produce rapid remission in patients at risk of death from metastatic melanoma.
Vemurafenib is not a panacea, as only about half of patients with BRAF V600E mutations have responded, and in the early studies almost all eventually relapsed. However, the drug has produced delays in recurrence and prolongations of overall survival that are clinically and statistically significant.
"This is a bright day for many melanoma patients," Dr. Jeffrey A. Sosman, a professor of medicine at Vanderbilt-Ingram Cancer Center in Nashville said in response to the FDA announcement.
"But it is only the start, and we have to continue to enroll patients onto clinical trials in order to build upon this exciting advance. It is only the beginning," added Dr. Sosman, an investigator in the BRIM-3 trial that led to approval of vemurafenib.
Early results of the pivotal phase III BRIM-3 trial, presented at the American Society of Clinical Oncology (ASCO) in June, and published simultaneously in the New England Journal of Medicine (2011;364:2507-16) , showed that vemurafenib reduced the relative risk of death by 63% in comparison with dacarbazine (DTIC), a standard but ineffective therapy, at a median follow-up of 3 months.
The randomized, open-label study screened 2,107 patients with unresectable stage III or IV melanoma for BRAF mutations, which were found in 47% of patients. It accrued 675 patients from January 2010 though December 2010, but some had not been in the study long enough to be included in the reports, as the trial was stopped based on early positive results.
Among the findings were estimates that 84% of patients treated with oral vemurafenib but only 64% of those given DTIC would be alive at 6 months. Vemurafenib’s hazard ratio for death was 0.37 (P less than .0001). An analysis of progression-free survival showed a 74% reduction in the risk of progression (HR 0.26, P less than .0001).
Median time to progression was 5.3 months with vemurafenib versus 1.6 months with DTIC, Dr. Paul Chapman of Memorial Sloan-Kettering Cancer Center in New York City and colleagues reported. Median overall survival had not been reached in the vemurafenib arm of BRIM-3, but Dr. Chapman expressed optimism that the advantage would hold up over time, as the Kaplan Meier survival curve mirrored results from the precursor BRIM-2 trial.
In that phase II study, median overall survival also had not yet been reached with 77% of patients alive at 6 months and 58% at 12 months, Dr. Antoni Ribas of the Jonsson Comprehensive Cancer Center in Los Angeles reported at the ASCO meeting. His presentation was based on a median follow-up of 7 months with an overall response rate of 53%.
The investigators also looked at patients who had BRAF mutations other than V600E, and were encouraged to find about half responded to vemurafenib, Dr. Chapman said in an interview.
In yet another presentation at ASCO, Dr. Alexander M. Menzies of the Melanoma Institute Australia reported that BRAF mutations are more common in younger melanoma patients. In all, 46% of 311 advanced melanoma patients had a BRAF mutation; 73% of these were V600E mutations, 19% V600K, and the remainder other forms.
The mutation rate declined from more than 80% of melanoma patients aged 20-40 years to 50% of those aged 41-70 years, and to less than 25% of those older than 70 years, Dr. Menzies said. The V600E mutation predominated in younger patients, occurring in 86% of those aged 20-30 years, whereas the V600K mutation emerged in older age.
Vemurafenib - approved at a recommended dose of 960 mg orally twice daily - has a manageable side effect profile, according to the BRIM-3 investigators. Although the safety data were not formally pooled, investigators said less than 10% of patients on active therapy had a grade 3 or higher toxicity. Joint pain, photosensitivity, and skin rashes were the most common. About 20% of patients developed cutaneous squamous cell carcinoma, but it was easily removed by dermatologists, and there were no instances of metastases. Only 6% of patients on vemurafenib discontinued because of adverse events.
Vemurafenib will be marketed as Zelboraf by Roche, which announced submission of applications for approval in the United States and Europe on May 11. Roche’s Genentech has a comarketing agreement with vemurafenib developer Plexxikon, a member of Daiichi Sankyo.
In June, Roche and Bristol-Meyers Squibb (maker of ipilimumab), announced that they would collaborate on a phase I/II study to determine whether combining the two drugs is safe and effective in patients with BRAF mutations.
The cobas 4800 BRAF V600 Mutation Test is manufactured by Roche Molecular Systems in Pleasanton, Calif.
Genentech sponsored the BRIM-2 and BRIM-3 trials. Dr. Chapman reported consultant/advisory roles and research funding from its parent company Roche. Some coauthors were Roche employees, and others reported similar relationships with Genentech. Dr. Ribas and several of his coauthors reported similar consulting and advisory relationships with Roche/Genentech. Dr. Menzies had no relevant financial conflicts of interest.
FDA Approves Vemurafenib for Advanced Melanoma
The Food and Drug Administration announced on August 17 the approval of vemurafenib, a highly anticipated metastatic melanoma therapy that targets the BRAF V600E mutation found in 40%-60% of patients.
The agency also approved the cobas 4800 BRAF V600 Mutation Test, a companion diagnostic test designed to help determine if a patient’s melanoma cells carry the BRAF V600E mutation.
Vemurafenib is the second therapy to prolong the lives of patients with metastatic melanoma. The first, a slow-acting immunotherapy called ipilimumab (Yervoy), was approved in March 2011.
Clinical trials have shown that vemurafenib (better known as PLX4032) can produce rapid remission in patients at risk of death from metastatic melanoma.
Vemurafenib is not a panacea, as only about half of patients with BRAF V600E mutations have responded, and in the early studies almost all eventually relapsed. However, the drug has produced delays in recurrence and prolongations of overall survival that are clinically and statistically significant.
"This is a bright day for many melanoma patients," Dr. Jeffrey A. Sosman, a professor of medicine at Vanderbilt-Ingram Cancer Center in Nashville said in response to the FDA announcement.
"But it is only the start, and we have to continue to enroll patients onto clinical trials in order to build upon this exciting advance. It is only the beginning," added Dr. Sosman, an investigator in the BRIM-3 trial that led to approval of vemurafenib.
Early results of the pivotal phase III BRIM-3 trial, presented at the American Society of Clinical Oncology (ASCO) in June, and published simultaneously in the New England Journal of Medicine (2011;364:2507-16) , showed that vemurafenib reduced the relative risk of death by 63% in comparison with dacarbazine (DTIC), a standard but ineffective therapy, at a median follow-up of 3 months.
The randomized, open-label study screened 2,107 patients with unresectable stage III or IV melanoma for BRAF mutations, which were found in 47% of patients. It accrued 675 patients from January 2010 though December 2010, but some had not been in the study long enough to be included in the reports, as the trial was stopped based on early positive results.
Among the findings were estimates that 84% of patients treated with oral vemurafenib but only 64% of those given DTIC would be alive at 6 months. Vemurafenib’s hazard ratio for death was 0.37 (P less than .0001). An analysis of progression-free survival showed a 74% reduction in the risk of progression (HR 0.26, P less than .0001).
Median time to progression was 5.3 months with vemurafenib versus 1.6 months with DTIC, Dr. Paul Chapman of Memorial Sloan-Kettering Cancer Center in New York City and colleagues reported. Median overall survival had not been reached in the vemurafenib arm of BRIM-3, but Dr. Chapman expressed optimism that the advantage would hold up over time, as the Kaplan Meier survival curve mirrored results from the precursor BRIM-2 trial.
In that phase II study, median overall survival also had not yet been reached with 77% of patients alive at 6 months and 58% at 12 months, Dr. Antoni Ribas of the Jonsson Comprehensive Cancer Center in Los Angeles reported at the ASCO meeting. His presentation was based on a median follow-up of 7 months with an overall response rate of 53%.
The investigators also looked at patients who had BRAF mutations other than V600E, and were encouraged to find about half responded to vemurafenib, Dr. Chapman said in an interview.
In yet another presentation at ASCO, Dr. Alexander M. Menzies of the Melanoma Institute Australia reported that BRAF mutations are more common in younger melanoma patients. In all, 46% of 311 advanced melanoma patients had a BRAF mutation; 73% of these were V600E mutations, 19% V600K, and the remainder other forms.
The mutation rate declined from more than 80% of melanoma patients aged 20-40 years to 50% of those aged 41-70 years, and to less than 25% of those older than 70 years, Dr. Menzies said. The V600E mutation predominated in younger patients, occurring in 86% of those aged 20-30 years, whereas the V600K mutation emerged in older age.
Vemurafenib - approved at a recommended dose of 960 mg orally twice daily - has a manageable side effect profile, according to the BRIM-3 investigators. Although the safety data were not formally pooled, investigators said less than 10% of patients on active therapy had a grade 3 or higher toxicity. Joint pain, photosensitivity, and skin rashes were the most common. About 20% of patients developed cutaneous squamous cell carcinoma, but it was easily removed by dermatologists, and there were no instances of metastases. Only 6% of patients on vemurafenib discontinued because of adverse events.
Vemurafenib will be marketed as Zelboraf by Roche, which announced submission of applications for approval in the United States and Europe on May 11. Roche’s Genentech has a comarketing agreement with vemurafenib developer Plexxikon, a member of Daiichi Sankyo.
In June, Roche and Bristol-Meyers Squibb (maker of ipilimumab), announced that they would collaborate on a phase I/II study to determine whether combining the two drugs is safe and effective in patients with BRAF mutations.
The cobas 4800 BRAF V600 Mutation Test is manufactured by Roche Molecular Systems in Pleasanton, Calif.
Genentech sponsored the BRIM-2 and BRIM-3 trials. Dr. Chapman reported consultant/advisory roles and research funding from its parent company Roche. Some coauthors were Roche employees, and others reported similar relationships with Genentech. Dr. Ribas and several of his coauthors reported similar consulting and advisory relationships with Roche/Genentech. Dr. Menzies had no relevant financial conflicts of interest.
The Food and Drug Administration announced on August 17 the approval of vemurafenib, a highly anticipated metastatic melanoma therapy that targets the BRAF V600E mutation found in 40%-60% of patients.
The agency also approved the cobas 4800 BRAF V600 Mutation Test, a companion diagnostic test designed to help determine if a patient’s melanoma cells carry the BRAF V600E mutation.
Vemurafenib is the second therapy to prolong the lives of patients with metastatic melanoma. The first, a slow-acting immunotherapy called ipilimumab (Yervoy), was approved in March 2011.
Clinical trials have shown that vemurafenib (better known as PLX4032) can produce rapid remission in patients at risk of death from metastatic melanoma.
Vemurafenib is not a panacea, as only about half of patients with BRAF V600E mutations have responded, and in the early studies almost all eventually relapsed. However, the drug has produced delays in recurrence and prolongations of overall survival that are clinically and statistically significant.
"This is a bright day for many melanoma patients," Dr. Jeffrey A. Sosman, a professor of medicine at Vanderbilt-Ingram Cancer Center in Nashville said in response to the FDA announcement.
"But it is only the start, and we have to continue to enroll patients onto clinical trials in order to build upon this exciting advance. It is only the beginning," added Dr. Sosman, an investigator in the BRIM-3 trial that led to approval of vemurafenib.
Early results of the pivotal phase III BRIM-3 trial, presented at the American Society of Clinical Oncology (ASCO) in June, and published simultaneously in the New England Journal of Medicine (2011;364:2507-16) , showed that vemurafenib reduced the relative risk of death by 63% in comparison with dacarbazine (DTIC), a standard but ineffective therapy, at a median follow-up of 3 months.
The randomized, open-label study screened 2,107 patients with unresectable stage III or IV melanoma for BRAF mutations, which were found in 47% of patients. It accrued 675 patients from January 2010 though December 2010, but some had not been in the study long enough to be included in the reports, as the trial was stopped based on early positive results.
Among the findings were estimates that 84% of patients treated with oral vemurafenib but only 64% of those given DTIC would be alive at 6 months. Vemurafenib’s hazard ratio for death was 0.37 (P less than .0001). An analysis of progression-free survival showed a 74% reduction in the risk of progression (HR 0.26, P less than .0001).
Median time to progression was 5.3 months with vemurafenib versus 1.6 months with DTIC, Dr. Paul Chapman of Memorial Sloan-Kettering Cancer Center in New York City and colleagues reported. Median overall survival had not been reached in the vemurafenib arm of BRIM-3, but Dr. Chapman expressed optimism that the advantage would hold up over time, as the Kaplan Meier survival curve mirrored results from the precursor BRIM-2 trial.
In that phase II study, median overall survival also had not yet been reached with 77% of patients alive at 6 months and 58% at 12 months, Dr. Antoni Ribas of the Jonsson Comprehensive Cancer Center in Los Angeles reported at the ASCO meeting. His presentation was based on a median follow-up of 7 months with an overall response rate of 53%.
The investigators also looked at patients who had BRAF mutations other than V600E, and were encouraged to find about half responded to vemurafenib, Dr. Chapman said in an interview.
In yet another presentation at ASCO, Dr. Alexander M. Menzies of the Melanoma Institute Australia reported that BRAF mutations are more common in younger melanoma patients. In all, 46% of 311 advanced melanoma patients had a BRAF mutation; 73% of these were V600E mutations, 19% V600K, and the remainder other forms.
The mutation rate declined from more than 80% of melanoma patients aged 20-40 years to 50% of those aged 41-70 years, and to less than 25% of those older than 70 years, Dr. Menzies said. The V600E mutation predominated in younger patients, occurring in 86% of those aged 20-30 years, whereas the V600K mutation emerged in older age.
Vemurafenib - approved at a recommended dose of 960 mg orally twice daily - has a manageable side effect profile, according to the BRIM-3 investigators. Although the safety data were not formally pooled, investigators said less than 10% of patients on active therapy had a grade 3 or higher toxicity. Joint pain, photosensitivity, and skin rashes were the most common. About 20% of patients developed cutaneous squamous cell carcinoma, but it was easily removed by dermatologists, and there were no instances of metastases. Only 6% of patients on vemurafenib discontinued because of adverse events.
Vemurafenib will be marketed as Zelboraf by Roche, which announced submission of applications for approval in the United States and Europe on May 11. Roche’s Genentech has a comarketing agreement with vemurafenib developer Plexxikon, a member of Daiichi Sankyo.
In June, Roche and Bristol-Meyers Squibb (maker of ipilimumab), announced that they would collaborate on a phase I/II study to determine whether combining the two drugs is safe and effective in patients with BRAF mutations.
The cobas 4800 BRAF V600 Mutation Test is manufactured by Roche Molecular Systems in Pleasanton, Calif.
Genentech sponsored the BRIM-2 and BRIM-3 trials. Dr. Chapman reported consultant/advisory roles and research funding from its parent company Roche. Some coauthors were Roche employees, and others reported similar relationships with Genentech. Dr. Ribas and several of his coauthors reported similar consulting and advisory relationships with Roche/Genentech. Dr. Menzies had no relevant financial conflicts of interest.
The Food and Drug Administration announced on August 17 the approval of vemurafenib, a highly anticipated metastatic melanoma therapy that targets the BRAF V600E mutation found in 40%-60% of patients.
The agency also approved the cobas 4800 BRAF V600 Mutation Test, a companion diagnostic test designed to help determine if a patient’s melanoma cells carry the BRAF V600E mutation.
Vemurafenib is the second therapy to prolong the lives of patients with metastatic melanoma. The first, a slow-acting immunotherapy called ipilimumab (Yervoy), was approved in March 2011.
Clinical trials have shown that vemurafenib (better known as PLX4032) can produce rapid remission in patients at risk of death from metastatic melanoma.
Vemurafenib is not a panacea, as only about half of patients with BRAF V600E mutations have responded, and in the early studies almost all eventually relapsed. However, the drug has produced delays in recurrence and prolongations of overall survival that are clinically and statistically significant.
"This is a bright day for many melanoma patients," Dr. Jeffrey A. Sosman, a professor of medicine at Vanderbilt-Ingram Cancer Center in Nashville said in response to the FDA announcement.
"But it is only the start, and we have to continue to enroll patients onto clinical trials in order to build upon this exciting advance. It is only the beginning," added Dr. Sosman, an investigator in the BRIM-3 trial that led to approval of vemurafenib.
Early results of the pivotal phase III BRIM-3 trial, presented at the American Society of Clinical Oncology (ASCO) in June, and published simultaneously in the New England Journal of Medicine (2011;364:2507-16) , showed that vemurafenib reduced the relative risk of death by 63% in comparison with dacarbazine (DTIC), a standard but ineffective therapy, at a median follow-up of 3 months.
The randomized, open-label study screened 2,107 patients with unresectable stage III or IV melanoma for BRAF mutations, which were found in 47% of patients. It accrued 675 patients from January 2010 though December 2010, but some had not been in the study long enough to be included in the reports, as the trial was stopped based on early positive results.
Among the findings were estimates that 84% of patients treated with oral vemurafenib but only 64% of those given DTIC would be alive at 6 months. Vemurafenib’s hazard ratio for death was 0.37 (P less than .0001). An analysis of progression-free survival showed a 74% reduction in the risk of progression (HR 0.26, P less than .0001).
Median time to progression was 5.3 months with vemurafenib versus 1.6 months with DTIC, Dr. Paul Chapman of Memorial Sloan-Kettering Cancer Center in New York City and colleagues reported. Median overall survival had not been reached in the vemurafenib arm of BRIM-3, but Dr. Chapman expressed optimism that the advantage would hold up over time, as the Kaplan Meier survival curve mirrored results from the precursor BRIM-2 trial.
In that phase II study, median overall survival also had not yet been reached with 77% of patients alive at 6 months and 58% at 12 months, Dr. Antoni Ribas of the Jonsson Comprehensive Cancer Center in Los Angeles reported at the ASCO meeting. His presentation was based on a median follow-up of 7 months with an overall response rate of 53%.
The investigators also looked at patients who had BRAF mutations other than V600E, and were encouraged to find about half responded to vemurafenib, Dr. Chapman said in an interview.
In yet another presentation at ASCO, Dr. Alexander M. Menzies of the Melanoma Institute Australia reported that BRAF mutations are more common in younger melanoma patients. In all, 46% of 311 advanced melanoma patients had a BRAF mutation; 73% of these were V600E mutations, 19% V600K, and the remainder other forms.
The mutation rate declined from more than 80% of melanoma patients aged 20-40 years to 50% of those aged 41-70 years, and to less than 25% of those older than 70 years, Dr. Menzies said. The V600E mutation predominated in younger patients, occurring in 86% of those aged 20-30 years, whereas the V600K mutation emerged in older age.
Vemurafenib - approved at a recommended dose of 960 mg orally twice daily - has a manageable side effect profile, according to the BRIM-3 investigators. Although the safety data were not formally pooled, investigators said less than 10% of patients on active therapy had a grade 3 or higher toxicity. Joint pain, photosensitivity, and skin rashes were the most common. About 20% of patients developed cutaneous squamous cell carcinoma, but it was easily removed by dermatologists, and there were no instances of metastases. Only 6% of patients on vemurafenib discontinued because of adverse events.
Vemurafenib will be marketed as Zelboraf by Roche, which announced submission of applications for approval in the United States and Europe on May 11. Roche’s Genentech has a comarketing agreement with vemurafenib developer Plexxikon, a member of Daiichi Sankyo.
In June, Roche and Bristol-Meyers Squibb (maker of ipilimumab), announced that they would collaborate on a phase I/II study to determine whether combining the two drugs is safe and effective in patients with BRAF mutations.
The cobas 4800 BRAF V600 Mutation Test is manufactured by Roche Molecular Systems in Pleasanton, Calif.
Genentech sponsored the BRIM-2 and BRIM-3 trials. Dr. Chapman reported consultant/advisory roles and research funding from its parent company Roche. Some coauthors were Roche employees, and others reported similar relationships with Genentech. Dr. Ribas and several of his coauthors reported similar consulting and advisory relationships with Roche/Genentech. Dr. Menzies had no relevant financial conflicts of interest.
Q&A: Putting the New Melanoma Drugs to Work
For the first time, physicians have two new drugs that can prolong the lives of patients with advanced melanoma. They also have a host of questions as to how to bring ipilimumab and vemurafenib into practice. We asked four experts for their thoughts on what comes next:
• Dr. Paul Chapman of Memorial Sloan-Kettering Cancer Center in New York; lead author of the BRIM-3 study of vemurafenib vs. dacarbazine in newly diagnosed patients.
• Dr. Jedd Wolchok of Memorial Sloan-Kettering Cancer Center; lead author of the phase III trial showing the efficacy of ipilimumab plus dacarbazine vs. placebo plus dacarbazine in newly diagnosed patients.
• Dr. Vernon K. Sondak, chair of cutaneous oncology and director of surgical education, H. Lee Moffitt Cancer Center & Research Institute in Tampa.
• Dr. Alexander M.M. Eggermont, general director of the Institut de Cancérologie Gustave Roussy in Villejuif, France.
Question: Should ipilimumab and vemurafenib be used together?
Dr. Chapman: We are about to start a phase III trial combining these to see if it is safe and making sure that vemurafenib does not inhibit the effect of ipilimumab. We all hope that this will result in sustained complete responses. That is what we all are looking for.
Dr. Wolchok: The natural next step that many of us are considering is how to integrate these two forms of therapy together. Vemurafenib directly targets the tumor by inhibiting BRAF kinase; ipilimumab really treats the patient by starting an immune reaction that hopefully will control the disease. These are very different approaches to cancer therapy. They are in no way mutually exclusive, and I believe them to be quite complementary.
I think it is important that the combination be explored in a clinical trial because I could make a list of reasons why these two drugs would work together; I could also create a list of reasons why they might not work well together and may even be antagonists. So, I think before physicians begin to combine potentially approved medications outside of clinical trials, we [need to] have some idea that this is a safe and effective way to go. Until then, I think monotherapy is the best path forward outside of a clinical trial.
Dr. Sondak: If you combine ipilimumab with something else, sometimes the side effects aren’t what you expect. Don’t just take a patient and give them both and see what happens. That’s not safe. You have to do this in a controlled way a research study as quickly as possible because it is the obvious next question.
Dr. Eggermont: I think at this point they should wait at least for the safety data on the combination.
Question: So which agent would you use first in a patient who has a BRAF mutation?
Dr. Chapman: I think that is a question is that is still open. Where I am on this question right now is that for a patient who is relatively well, who has a fairly good performance status, I would think about using ipilimumab first because that person may have time to respond to ipilimumab since it does take 3-6 months to have the full effect. On the other hand, a person who has a poor performance status and is sick and does not have time to respond to ipilimumab I would treat that person up-front with vemurafenib.
Dr. Sondak: I think there is going to be uniform agreement throughout the melanoma community that if you have a BRAF-mutant patient with widespread disease, symptomatic disease, high tumor burden that person is going to go immediately on to vemurafenib because nothing else is going to get a rapid response, a rapid resolution of symptoms.
The question is going to be [what to do] for 80% of patients who are BRAF mutant but have much less acute symptomatic disease. Maybe they have some lung metastases, some subcutaneous nodules, [and] they can’t have it all removed surgically but they are still not in dire straits. And I think at our institution we are going to be very motivated to continue to use the immunologic therapies - the IL-2 for some patients, and ipilimumab for many patients because of the possibility that they will get a big benefit at the other end, not an immediate benefit but a long-term benefit. And then if that doesn’t work, I think vemurafenib.
Dr. Eggermont: BRAF-positive patients especially the ones that have rapidly progressive disease or bulky disease that have little time – they will all be treated up front with the BRAF inhibitor. The BRAF-positive patients that have very small disease that is not rapidly progressing could actually also be managed by ipilimumab on first line and only on progression be managed by the BRAF inhibitor because ipilimumab needs more time to work with patients, to kick into activity and into an antitumor effect. ... All BRAF-negative patients would see ipilimumab in first line. That is for sure.
Question: The ipilimumab trial paired ipilimumab with dacarbazine. Is DTIC still an option?
Dr. Wolchok: Looking at the result as someone who has been taking care of patients with melanoma using ipilimumab for several years, I think it is still unknown whether dacarbazine should be added. My own opinion would be to oncologists in the community that if they are going to give ipilimumab at 3 mg/kg to stick with the monotherapy label because we really don’t have data comparing ipilimumab with dacarbazine alone at that dose. And so I think we stick with what we know produces an overall survival benefit.
There are reasons to imagine the addition of dacarbazine could make results better by killing some cancer cells and releasing some antigenic debris that would serve as a target for the immune system. Chemotherapy can also be thought of as changing the tumor microenvironment, which might be advantageous, but chemotherapy could also be immunosuppressive. So we could really make a case either way that dacarbazine added to or detracted from or left things just the same.
Question: Will interleukin-2 still have a role in melanoma treatment?
Dr. Sondak: I am still going to use IL-2, and it makes the most sense if you have someone who uses IL-2 most of time, you will want to use that first before you use anything else. Why? Ipilimumab is tough on the patient. We want them in as good shape as possible. ... We know if they get IL-2 first and then get ipilimumab; the results are just as good, if not better. We don’t know the opposite way. And we also know that a lot of people on ipilimumab get side effects. They wind up on steroids and other immunosuppressive drugs that would make it very difficult to put them on IL-2.
Vemurafenib is going to change a lot of things. A lot of people are too far gone to have surgery, too far gone to have IL-2. Now we have BRAF mutant melanoma [patients] who will get vemurafenib, and shrink down... If we could keep a close eye and figure out when is right time, we are going to do more surgery and maybe IL-2 in those patients because we are going to restore them to place where IL-2 is going to have more room to work. But it’s not going to be 80% of people getting IL-2. It is going to be a very restricted group of people treated by a very specialized group of doctors.
Dr. Eggermont: I think IL-2 will move down in the options list. IL-2 will remain as an option in second or third line, and IL-2 will also be an option in combination with ipilimumab because [Dr.] Steve Rosenberg already did a trial in 36 patients ... and the majority of the complete responders are alive 6 years and longer and are still in complete remission (ASCO 2010, Abstract 8544). The combination of IL-2 and ipilimumab will be relaunched, I am sure.
Question: And interferon – Where will it fit in?
Dr. Sondak: We don’t know how long we should treat people, how intensively, and how we should combine the new and the old, and the data that we have just doesn’t sort it out. On the other hand, if you look at the whole landscape, it actually makes adjuvant treatment with interferon more important now than it used to be.
It’s very clear that interferon treatment does delay recurrences in a substantial fraction of patients. It may not cure many people, but it delays recurrence in quite a few people, and right now today we are so much better off than we were in melanoma 2 years ago and I have no doubt that 2 years from now just with stuff we heard about at this meeting we will be much more clear on how do we use how do we take care of side effects not to mention other new exciting drugs that we hope 2 years from now will start to be available.
In the past we debated, does it even matter that we delay recurrence. The ASCO answer should be, "It absolutely matters to our patients." So interferon and any form of adjuvant therapy is suddenly a bridge. It is a shaky bridge – made with some ropes and a few planks – it isn’t very sturdy to walk on, but we’re trying to get from here, one place where it was safe to another place where hopefully we are better off and not at the bottom of ravine. It’s real exciting – not just the data we already have but for the future and how many patients this will affect positively.
Dr. Eggermont: I think you need to separate the new drugs from the question, what are we going to do in adjuvant? In adjuvant right now, we will still have to wait for about 2.5-3 years for the outcome of the EORTC trial that randomized in double-blind ipilimumab vs. observation in patients with high-risk lymph node positive disease. That answer about ipilimumab in the adjuvant situation will not be there before 2.5 years from now. That means that until then there is only one kid on the block in the adjuvant setting, which is regular interferon, and the currently novel approach of pegylated interferon.
For the first time, physicians have two new drugs that can prolong the lives of patients with advanced melanoma. They also have a host of questions as to how to bring ipilimumab and vemurafenib into practice. We asked four experts for their thoughts on what comes next:
• Dr. Paul Chapman of Memorial Sloan-Kettering Cancer Center in New York; lead author of the BRIM-3 study of vemurafenib vs. dacarbazine in newly diagnosed patients.
• Dr. Jedd Wolchok of Memorial Sloan-Kettering Cancer Center; lead author of the phase III trial showing the efficacy of ipilimumab plus dacarbazine vs. placebo plus dacarbazine in newly diagnosed patients.
• Dr. Vernon K. Sondak, chair of cutaneous oncology and director of surgical education, H. Lee Moffitt Cancer Center & Research Institute in Tampa.
• Dr. Alexander M.M. Eggermont, general director of the Institut de Cancérologie Gustave Roussy in Villejuif, France.
Question: Should ipilimumab and vemurafenib be used together?
Dr. Chapman: We are about to start a phase III trial combining these to see if it is safe and making sure that vemurafenib does not inhibit the effect of ipilimumab. We all hope that this will result in sustained complete responses. That is what we all are looking for.
Dr. Wolchok: The natural next step that many of us are considering is how to integrate these two forms of therapy together. Vemurafenib directly targets the tumor by inhibiting BRAF kinase; ipilimumab really treats the patient by starting an immune reaction that hopefully will control the disease. These are very different approaches to cancer therapy. They are in no way mutually exclusive, and I believe them to be quite complementary.
I think it is important that the combination be explored in a clinical trial because I could make a list of reasons why these two drugs would work together; I could also create a list of reasons why they might not work well together and may even be antagonists. So, I think before physicians begin to combine potentially approved medications outside of clinical trials, we [need to] have some idea that this is a safe and effective way to go. Until then, I think monotherapy is the best path forward outside of a clinical trial.
Dr. Sondak: If you combine ipilimumab with something else, sometimes the side effects aren’t what you expect. Don’t just take a patient and give them both and see what happens. That’s not safe. You have to do this in a controlled way a research study as quickly as possible because it is the obvious next question.
Dr. Eggermont: I think at this point they should wait at least for the safety data on the combination.
Question: So which agent would you use first in a patient who has a BRAF mutation?
Dr. Chapman: I think that is a question is that is still open. Where I am on this question right now is that for a patient who is relatively well, who has a fairly good performance status, I would think about using ipilimumab first because that person may have time to respond to ipilimumab since it does take 3-6 months to have the full effect. On the other hand, a person who has a poor performance status and is sick and does not have time to respond to ipilimumab I would treat that person up-front with vemurafenib.
Dr. Sondak: I think there is going to be uniform agreement throughout the melanoma community that if you have a BRAF-mutant patient with widespread disease, symptomatic disease, high tumor burden that person is going to go immediately on to vemurafenib because nothing else is going to get a rapid response, a rapid resolution of symptoms.
The question is going to be [what to do] for 80% of patients who are BRAF mutant but have much less acute symptomatic disease. Maybe they have some lung metastases, some subcutaneous nodules, [and] they can’t have it all removed surgically but they are still not in dire straits. And I think at our institution we are going to be very motivated to continue to use the immunologic therapies - the IL-2 for some patients, and ipilimumab for many patients because of the possibility that they will get a big benefit at the other end, not an immediate benefit but a long-term benefit. And then if that doesn’t work, I think vemurafenib.
Dr. Eggermont: BRAF-positive patients especially the ones that have rapidly progressive disease or bulky disease that have little time – they will all be treated up front with the BRAF inhibitor. The BRAF-positive patients that have very small disease that is not rapidly progressing could actually also be managed by ipilimumab on first line and only on progression be managed by the BRAF inhibitor because ipilimumab needs more time to work with patients, to kick into activity and into an antitumor effect. ... All BRAF-negative patients would see ipilimumab in first line. That is for sure.
Question: The ipilimumab trial paired ipilimumab with dacarbazine. Is DTIC still an option?
Dr. Wolchok: Looking at the result as someone who has been taking care of patients with melanoma using ipilimumab for several years, I think it is still unknown whether dacarbazine should be added. My own opinion would be to oncologists in the community that if they are going to give ipilimumab at 3 mg/kg to stick with the monotherapy label because we really don’t have data comparing ipilimumab with dacarbazine alone at that dose. And so I think we stick with what we know produces an overall survival benefit.
There are reasons to imagine the addition of dacarbazine could make results better by killing some cancer cells and releasing some antigenic debris that would serve as a target for the immune system. Chemotherapy can also be thought of as changing the tumor microenvironment, which might be advantageous, but chemotherapy could also be immunosuppressive. So we could really make a case either way that dacarbazine added to or detracted from or left things just the same.
Question: Will interleukin-2 still have a role in melanoma treatment?
Dr. Sondak: I am still going to use IL-2, and it makes the most sense if you have someone who uses IL-2 most of time, you will want to use that first before you use anything else. Why? Ipilimumab is tough on the patient. We want them in as good shape as possible. ... We know if they get IL-2 first and then get ipilimumab; the results are just as good, if not better. We don’t know the opposite way. And we also know that a lot of people on ipilimumab get side effects. They wind up on steroids and other immunosuppressive drugs that would make it very difficult to put them on IL-2.
Vemurafenib is going to change a lot of things. A lot of people are too far gone to have surgery, too far gone to have IL-2. Now we have BRAF mutant melanoma [patients] who will get vemurafenib, and shrink down... If we could keep a close eye and figure out when is right time, we are going to do more surgery and maybe IL-2 in those patients because we are going to restore them to place where IL-2 is going to have more room to work. But it’s not going to be 80% of people getting IL-2. It is going to be a very restricted group of people treated by a very specialized group of doctors.
Dr. Eggermont: I think IL-2 will move down in the options list. IL-2 will remain as an option in second or third line, and IL-2 will also be an option in combination with ipilimumab because [Dr.] Steve Rosenberg already did a trial in 36 patients ... and the majority of the complete responders are alive 6 years and longer and are still in complete remission (ASCO 2010, Abstract 8544). The combination of IL-2 and ipilimumab will be relaunched, I am sure.
Question: And interferon – Where will it fit in?
Dr. Sondak: We don’t know how long we should treat people, how intensively, and how we should combine the new and the old, and the data that we have just doesn’t sort it out. On the other hand, if you look at the whole landscape, it actually makes adjuvant treatment with interferon more important now than it used to be.
It’s very clear that interferon treatment does delay recurrences in a substantial fraction of patients. It may not cure many people, but it delays recurrence in quite a few people, and right now today we are so much better off than we were in melanoma 2 years ago and I have no doubt that 2 years from now just with stuff we heard about at this meeting we will be much more clear on how do we use how do we take care of side effects not to mention other new exciting drugs that we hope 2 years from now will start to be available.
In the past we debated, does it even matter that we delay recurrence. The ASCO answer should be, "It absolutely matters to our patients." So interferon and any form of adjuvant therapy is suddenly a bridge. It is a shaky bridge – made with some ropes and a few planks – it isn’t very sturdy to walk on, but we’re trying to get from here, one place where it was safe to another place where hopefully we are better off and not at the bottom of ravine. It’s real exciting – not just the data we already have but for the future and how many patients this will affect positively.
Dr. Eggermont: I think you need to separate the new drugs from the question, what are we going to do in adjuvant? In adjuvant right now, we will still have to wait for about 2.5-3 years for the outcome of the EORTC trial that randomized in double-blind ipilimumab vs. observation in patients with high-risk lymph node positive disease. That answer about ipilimumab in the adjuvant situation will not be there before 2.5 years from now. That means that until then there is only one kid on the block in the adjuvant setting, which is regular interferon, and the currently novel approach of pegylated interferon.
For the first time, physicians have two new drugs that can prolong the lives of patients with advanced melanoma. They also have a host of questions as to how to bring ipilimumab and vemurafenib into practice. We asked four experts for their thoughts on what comes next:
• Dr. Paul Chapman of Memorial Sloan-Kettering Cancer Center in New York; lead author of the BRIM-3 study of vemurafenib vs. dacarbazine in newly diagnosed patients.
• Dr. Jedd Wolchok of Memorial Sloan-Kettering Cancer Center; lead author of the phase III trial showing the efficacy of ipilimumab plus dacarbazine vs. placebo plus dacarbazine in newly diagnosed patients.
• Dr. Vernon K. Sondak, chair of cutaneous oncology and director of surgical education, H. Lee Moffitt Cancer Center & Research Institute in Tampa.
• Dr. Alexander M.M. Eggermont, general director of the Institut de Cancérologie Gustave Roussy in Villejuif, France.
Question: Should ipilimumab and vemurafenib be used together?
Dr. Chapman: We are about to start a phase III trial combining these to see if it is safe and making sure that vemurafenib does not inhibit the effect of ipilimumab. We all hope that this will result in sustained complete responses. That is what we all are looking for.
Dr. Wolchok: The natural next step that many of us are considering is how to integrate these two forms of therapy together. Vemurafenib directly targets the tumor by inhibiting BRAF kinase; ipilimumab really treats the patient by starting an immune reaction that hopefully will control the disease. These are very different approaches to cancer therapy. They are in no way mutually exclusive, and I believe them to be quite complementary.
I think it is important that the combination be explored in a clinical trial because I could make a list of reasons why these two drugs would work together; I could also create a list of reasons why they might not work well together and may even be antagonists. So, I think before physicians begin to combine potentially approved medications outside of clinical trials, we [need to] have some idea that this is a safe and effective way to go. Until then, I think monotherapy is the best path forward outside of a clinical trial.
Dr. Sondak: If you combine ipilimumab with something else, sometimes the side effects aren’t what you expect. Don’t just take a patient and give them both and see what happens. That’s not safe. You have to do this in a controlled way a research study as quickly as possible because it is the obvious next question.
Dr. Eggermont: I think at this point they should wait at least for the safety data on the combination.
Question: So which agent would you use first in a patient who has a BRAF mutation?
Dr. Chapman: I think that is a question is that is still open. Where I am on this question right now is that for a patient who is relatively well, who has a fairly good performance status, I would think about using ipilimumab first because that person may have time to respond to ipilimumab since it does take 3-6 months to have the full effect. On the other hand, a person who has a poor performance status and is sick and does not have time to respond to ipilimumab I would treat that person up-front with vemurafenib.
Dr. Sondak: I think there is going to be uniform agreement throughout the melanoma community that if you have a BRAF-mutant patient with widespread disease, symptomatic disease, high tumor burden that person is going to go immediately on to vemurafenib because nothing else is going to get a rapid response, a rapid resolution of symptoms.
The question is going to be [what to do] for 80% of patients who are BRAF mutant but have much less acute symptomatic disease. Maybe they have some lung metastases, some subcutaneous nodules, [and] they can’t have it all removed surgically but they are still not in dire straits. And I think at our institution we are going to be very motivated to continue to use the immunologic therapies - the IL-2 for some patients, and ipilimumab for many patients because of the possibility that they will get a big benefit at the other end, not an immediate benefit but a long-term benefit. And then if that doesn’t work, I think vemurafenib.
Dr. Eggermont: BRAF-positive patients especially the ones that have rapidly progressive disease or bulky disease that have little time – they will all be treated up front with the BRAF inhibitor. The BRAF-positive patients that have very small disease that is not rapidly progressing could actually also be managed by ipilimumab on first line and only on progression be managed by the BRAF inhibitor because ipilimumab needs more time to work with patients, to kick into activity and into an antitumor effect. ... All BRAF-negative patients would see ipilimumab in first line. That is for sure.
Question: The ipilimumab trial paired ipilimumab with dacarbazine. Is DTIC still an option?
Dr. Wolchok: Looking at the result as someone who has been taking care of patients with melanoma using ipilimumab for several years, I think it is still unknown whether dacarbazine should be added. My own opinion would be to oncologists in the community that if they are going to give ipilimumab at 3 mg/kg to stick with the monotherapy label because we really don’t have data comparing ipilimumab with dacarbazine alone at that dose. And so I think we stick with what we know produces an overall survival benefit.
There are reasons to imagine the addition of dacarbazine could make results better by killing some cancer cells and releasing some antigenic debris that would serve as a target for the immune system. Chemotherapy can also be thought of as changing the tumor microenvironment, which might be advantageous, but chemotherapy could also be immunosuppressive. So we could really make a case either way that dacarbazine added to or detracted from or left things just the same.
Question: Will interleukin-2 still have a role in melanoma treatment?
Dr. Sondak: I am still going to use IL-2, and it makes the most sense if you have someone who uses IL-2 most of time, you will want to use that first before you use anything else. Why? Ipilimumab is tough on the patient. We want them in as good shape as possible. ... We know if they get IL-2 first and then get ipilimumab; the results are just as good, if not better. We don’t know the opposite way. And we also know that a lot of people on ipilimumab get side effects. They wind up on steroids and other immunosuppressive drugs that would make it very difficult to put them on IL-2.
Vemurafenib is going to change a lot of things. A lot of people are too far gone to have surgery, too far gone to have IL-2. Now we have BRAF mutant melanoma [patients] who will get vemurafenib, and shrink down... If we could keep a close eye and figure out when is right time, we are going to do more surgery and maybe IL-2 in those patients because we are going to restore them to place where IL-2 is going to have more room to work. But it’s not going to be 80% of people getting IL-2. It is going to be a very restricted group of people treated by a very specialized group of doctors.
Dr. Eggermont: I think IL-2 will move down in the options list. IL-2 will remain as an option in second or third line, and IL-2 will also be an option in combination with ipilimumab because [Dr.] Steve Rosenberg already did a trial in 36 patients ... and the majority of the complete responders are alive 6 years and longer and are still in complete remission (ASCO 2010, Abstract 8544). The combination of IL-2 and ipilimumab will be relaunched, I am sure.
Question: And interferon – Where will it fit in?
Dr. Sondak: We don’t know how long we should treat people, how intensively, and how we should combine the new and the old, and the data that we have just doesn’t sort it out. On the other hand, if you look at the whole landscape, it actually makes adjuvant treatment with interferon more important now than it used to be.
It’s very clear that interferon treatment does delay recurrences in a substantial fraction of patients. It may not cure many people, but it delays recurrence in quite a few people, and right now today we are so much better off than we were in melanoma 2 years ago and I have no doubt that 2 years from now just with stuff we heard about at this meeting we will be much more clear on how do we use how do we take care of side effects not to mention other new exciting drugs that we hope 2 years from now will start to be available.
In the past we debated, does it even matter that we delay recurrence. The ASCO answer should be, "It absolutely matters to our patients." So interferon and any form of adjuvant therapy is suddenly a bridge. It is a shaky bridge – made with some ropes and a few planks – it isn’t very sturdy to walk on, but we’re trying to get from here, one place where it was safe to another place where hopefully we are better off and not at the bottom of ravine. It’s real exciting – not just the data we already have but for the future and how many patients this will affect positively.
Dr. Eggermont: I think you need to separate the new drugs from the question, what are we going to do in adjuvant? In adjuvant right now, we will still have to wait for about 2.5-3 years for the outcome of the EORTC trial that randomized in double-blind ipilimumab vs. observation in patients with high-risk lymph node positive disease. That answer about ipilimumab in the adjuvant situation will not be there before 2.5 years from now. That means that until then there is only one kid on the block in the adjuvant setting, which is regular interferon, and the currently novel approach of pegylated interferon.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF CLINICAL ONCOLOGY