User login
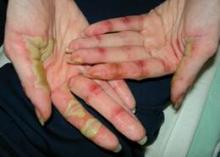
Metastatic Melanoma and Melanogenuria
What Is Your Diagnosis? Anal Mucosal Melanoma
New Melanoma Treatments: A Panacea or a Pandora's Box? [editorial]
Vemurafenib in melanoma with the BRAF V600E mutation
Vemurafenib, an oral inhibitor of some mutated forms of the BRAF serine threonine kinase, was recently approved for the treatment of patients with unresectable or metastatic melanoma with the BRAF V600E mutation as detected by an FDA- approved test. It is not recommended for use in patients with wild-type BRAF melanoma. The clinical trial supporting approval of vemurafenib (PLX4032) was performed in treatment-naïve patients with the V600E mutation as detected by the Cobas 4800 BRAF V600 Mutation Test. About 40%-60% of cutaneous melanomas have BRAF mutations that result in constitutive activation of downstream signaling through the MAPK pathway; about 90% of those carry the V600E mutation.
*For a PDF of the full article, click on the link to the left of this introduction.
Vemurafenib, an oral inhibitor of some mutated forms of the BRAF serine threonine kinase, was recently approved for the treatment of patients with unresectable or metastatic melanoma with the BRAF V600E mutation as detected by an FDA- approved test. It is not recommended for use in patients with wild-type BRAF melanoma. The clinical trial supporting approval of vemurafenib (PLX4032) was performed in treatment-naïve patients with the V600E mutation as detected by the Cobas 4800 BRAF V600 Mutation Test. About 40%-60% of cutaneous melanomas have BRAF mutations that result in constitutive activation of downstream signaling through the MAPK pathway; about 90% of those carry the V600E mutation.
*For a PDF of the full article, click on the link to the left of this introduction.
Vemurafenib, an oral inhibitor of some mutated forms of the BRAF serine threonine kinase, was recently approved for the treatment of patients with unresectable or metastatic melanoma with the BRAF V600E mutation as detected by an FDA- approved test. It is not recommended for use in patients with wild-type BRAF melanoma. The clinical trial supporting approval of vemurafenib (PLX4032) was performed in treatment-naïve patients with the V600E mutation as detected by the Cobas 4800 BRAF V600 Mutation Test. About 40%-60% of cutaneous melanomas have BRAF mutations that result in constitutive activation of downstream signaling through the MAPK pathway; about 90% of those carry the V600E mutation.
*For a PDF of the full article, click on the link to the left of this introduction.
'Early, Proactive' Management of Chemo Toxicites Improves QOL
Skin conditions in cancer patients can lead to a decreased quality of life because of the psychosocial impact, financial burden, poor health, and disruption of anticancer treatments, said Dr. Mario E. Lacouture at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Clinical trials are underway to address management, cost, and quality of life, but in the meantime, "an early and proactive approach toward toxicities is advisable," he said.
Dr. Lacouture, a dermatologist at Memorial Sloan-Kettering Cancer Center in New York, discussed common cancer-associated dermatology at the seminar.
• Rashes. A RAF inhibitor–induced rash can occur within 1-2 weeks of starting treatment, Dr. Lacouture noted. This rash is associated with sensations of burning or itching in photo-exposed areas. In most cases, the rash can be managed with topical steroids and oral GABA agonists. Vemurafenib use also has been associated with rashes, as well as with photosensitivity, itching, and alopecia, he said.
In addition, mTOR inhibitor–induced rashes involving erythematous pruritic papules and ulcerlike lesions have been observed in patients on everolimus and temsirolimus.
• Skin cancers. Data from several studies suggest that the incidence of squamous cell carcinomas in patients on RAF inhibitors is 7%, and the incidence of actinic keratoses in these patients is 4%, according to Dr. Lacouture. These conditions usually develop after approximately 6 months of treatment. They can be managed by surgery or destruction; and there have been no reports of metastases.
• Hand-foot syndrome. Hand-Foot Syndrome (HFSR) is a common side effect in cancer patients that usually occurs within 45 days of starting treatment, said Dr. Lacouture. HFSR is often associated with the use of sorafenib, sunitinib, and pazopanib, and the histology of these patients shows epidermal necrosis, he noted. Patients who develop HFSR may need to temporarily reduce their medications. Additional management of HFSR includes avoiding excess pressure on the affected areas and keeping them cool and moist to reduce patient discomfort.
• Hair and nail changes. Changes in hair can occur in patients being treated with epidermal growth factor receptor inhibitors (EGFRIs), including a slower hair growth rate and alopecia, Dr. Lacouture said. Changes in hair texture such as increased brittleness and hair curling also have been observed, he said. Other hair-related adverse events can include hirsutism on the face and eyelash trichomegaly. In addition, paronychia has been observed in patients on EGFRIs for more than 6 months.
• Radiation dermatitis. Approximately 50% of cancer patients develop radiation dermatitis, including 87% of breast cancer patients, reported Dr. Lacouture. However, the treatment of radiation dermatitis remains challenging, he said. In several studies, patients who used a nonsteroidal topical, including aloe vera and trolamine, showed no significant improvement in radiation dermatitis, compared with controls. However, studies are ongoing, and significant improvements in radiation dermatitis in breast cancer patients have been seen with topical corticosteroids including mometasone, beclomethasone, and betamethasone.
Dr. Lacouture disclosed financial relationships with multiple companies including Amgen, Bristol-Myers Squibb, Genentech, GlaxoSmithKline, Merck, and Roche. SDEF and this news organization are owned by Elsevier.
Skin conditions in cancer patients can lead to a decreased quality of life because of the psychosocial impact, financial burden, poor health, and disruption of anticancer treatments, said Dr. Mario E. Lacouture at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Clinical trials are underway to address management, cost, and quality of life, but in the meantime, "an early and proactive approach toward toxicities is advisable," he said.
Dr. Lacouture, a dermatologist at Memorial Sloan-Kettering Cancer Center in New York, discussed common cancer-associated dermatology at the seminar.
• Rashes. A RAF inhibitor–induced rash can occur within 1-2 weeks of starting treatment, Dr. Lacouture noted. This rash is associated with sensations of burning or itching in photo-exposed areas. In most cases, the rash can be managed with topical steroids and oral GABA agonists. Vemurafenib use also has been associated with rashes, as well as with photosensitivity, itching, and alopecia, he said.
In addition, mTOR inhibitor–induced rashes involving erythematous pruritic papules and ulcerlike lesions have been observed in patients on everolimus and temsirolimus.
• Skin cancers. Data from several studies suggest that the incidence of squamous cell carcinomas in patients on RAF inhibitors is 7%, and the incidence of actinic keratoses in these patients is 4%, according to Dr. Lacouture. These conditions usually develop after approximately 6 months of treatment. They can be managed by surgery or destruction; and there have been no reports of metastases.
• Hand-foot syndrome. Hand-Foot Syndrome (HFSR) is a common side effect in cancer patients that usually occurs within 45 days of starting treatment, said Dr. Lacouture. HFSR is often associated with the use of sorafenib, sunitinib, and pazopanib, and the histology of these patients shows epidermal necrosis, he noted. Patients who develop HFSR may need to temporarily reduce their medications. Additional management of HFSR includes avoiding excess pressure on the affected areas and keeping them cool and moist to reduce patient discomfort.
• Hair and nail changes. Changes in hair can occur in patients being treated with epidermal growth factor receptor inhibitors (EGFRIs), including a slower hair growth rate and alopecia, Dr. Lacouture said. Changes in hair texture such as increased brittleness and hair curling also have been observed, he said. Other hair-related adverse events can include hirsutism on the face and eyelash trichomegaly. In addition, paronychia has been observed in patients on EGFRIs for more than 6 months.
• Radiation dermatitis. Approximately 50% of cancer patients develop radiation dermatitis, including 87% of breast cancer patients, reported Dr. Lacouture. However, the treatment of radiation dermatitis remains challenging, he said. In several studies, patients who used a nonsteroidal topical, including aloe vera and trolamine, showed no significant improvement in radiation dermatitis, compared with controls. However, studies are ongoing, and significant improvements in radiation dermatitis in breast cancer patients have been seen with topical corticosteroids including mometasone, beclomethasone, and betamethasone.
Dr. Lacouture disclosed financial relationships with multiple companies including Amgen, Bristol-Myers Squibb, Genentech, GlaxoSmithKline, Merck, and Roche. SDEF and this news organization are owned by Elsevier.
Skin conditions in cancer patients can lead to a decreased quality of life because of the psychosocial impact, financial burden, poor health, and disruption of anticancer treatments, said Dr. Mario E. Lacouture at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Clinical trials are underway to address management, cost, and quality of life, but in the meantime, "an early and proactive approach toward toxicities is advisable," he said.
Dr. Lacouture, a dermatologist at Memorial Sloan-Kettering Cancer Center in New York, discussed common cancer-associated dermatology at the seminar.
• Rashes. A RAF inhibitor–induced rash can occur within 1-2 weeks of starting treatment, Dr. Lacouture noted. This rash is associated with sensations of burning or itching in photo-exposed areas. In most cases, the rash can be managed with topical steroids and oral GABA agonists. Vemurafenib use also has been associated with rashes, as well as with photosensitivity, itching, and alopecia, he said.
In addition, mTOR inhibitor–induced rashes involving erythematous pruritic papules and ulcerlike lesions have been observed in patients on everolimus and temsirolimus.
• Skin cancers. Data from several studies suggest that the incidence of squamous cell carcinomas in patients on RAF inhibitors is 7%, and the incidence of actinic keratoses in these patients is 4%, according to Dr. Lacouture. These conditions usually develop after approximately 6 months of treatment. They can be managed by surgery or destruction; and there have been no reports of metastases.
• Hand-foot syndrome. Hand-Foot Syndrome (HFSR) is a common side effect in cancer patients that usually occurs within 45 days of starting treatment, said Dr. Lacouture. HFSR is often associated with the use of sorafenib, sunitinib, and pazopanib, and the histology of these patients shows epidermal necrosis, he noted. Patients who develop HFSR may need to temporarily reduce their medications. Additional management of HFSR includes avoiding excess pressure on the affected areas and keeping them cool and moist to reduce patient discomfort.
• Hair and nail changes. Changes in hair can occur in patients being treated with epidermal growth factor receptor inhibitors (EGFRIs), including a slower hair growth rate and alopecia, Dr. Lacouture said. Changes in hair texture such as increased brittleness and hair curling also have been observed, he said. Other hair-related adverse events can include hirsutism on the face and eyelash trichomegaly. In addition, paronychia has been observed in patients on EGFRIs for more than 6 months.
• Radiation dermatitis. Approximately 50% of cancer patients develop radiation dermatitis, including 87% of breast cancer patients, reported Dr. Lacouture. However, the treatment of radiation dermatitis remains challenging, he said. In several studies, patients who used a nonsteroidal topical, including aloe vera and trolamine, showed no significant improvement in radiation dermatitis, compared with controls. However, studies are ongoing, and significant improvements in radiation dermatitis in breast cancer patients have been seen with topical corticosteroids including mometasone, beclomethasone, and betamethasone.
Dr. Lacouture disclosed financial relationships with multiple companies including Amgen, Bristol-Myers Squibb, Genentech, GlaxoSmithKline, Merck, and Roche. SDEF and this news organization are owned by Elsevier.
EXPERT ANALYSIS FROM THE SDEF HAWAII DERMATOLOGY SEMINAR
VEGF-Targeting Cancer Drugs Raise Risk of Fatal Side Effects
SAN FRANCISCO – Tyrosine kinase inhibitors that target the vascular endothelial growth factor receptor increase the risk of a fatal adverse event in cancer patients, but the absolute rate is still low, investigators concluded from a meta-analysis of 10 randomized trials with a total of 4,679 patients.
Trial participants given sorafenib (Nexavar), sunitinib (Sutent), or pazopanib (Votrient) were more than twice as likely to die from an adverse event as were peers given control treatment, said lead investigator Dr. Christopher J. Richards of the Beth Israel Deaconess Medical Center in Boston. Yet, the rate was still less than 2%.
"Our analysis may underestimate the true incidence of fatal adverse events, as study patients may be healthier than the general population exposed to these drugs," he noted at the Genitourinary Cancers Symposium. But that said, "all three of the study drugs have been shown in various randomized controlled trials to improve clinical outcomes compared to traditional therapies."
Discussant Dr. Thomas E. Hutson of the Baylor Sammons Cancer Center in Dallas commented, "I feel confident that everyone or almost everyone in the room today would agree with me that our current therapy benefit outweighs the risk associated with the treatment."
Moreover, even among the new targeted agents, the 1.5% rate seen with the vascular endothelial growth factor receptor (VEGFR) tyrosine kinase inhibitors (TKIs) in the meta-analysis "compares very favorably" with the rate of 2.9% seen previously with bevacizumab (Avastin), another VEGF-targeted therapy, in a similar analysis (JAMA 2011;305:487-94).
"Targeted therapies have real toxicities, and these toxicities include a low risk of death – a risk of death not much different than other therapies used for kidney and other cancer types," Dr. Hutson asserted. "If anything, this meta-analysis shows us that close monitoring is going to be essential for optimal use of these agents."
In the study, Dr. Richards and his colleagues identified 10 randomized controlled phase II or III trials in which patients received any of three VEGFR TKIs approved by the Food and Drug Administration for cancer treatment as of February 2011: sorafenib, sunitinib, or pazopanib. The control groups were usually treated with a placebo, but in some cases with chemotherapy or immunotherapy.
The enrolled patients had a variety of malignancies: renal cell carcinoma, hepatocellular carcinoma, melanoma, non–small cell lung cancer, breast cancer, and pancreatic neuroendocrine tumor. "All patients included in the studies had good performance status and had adequate baseline cardiac, hematologic, and renal function at the time of study entry," Dr. Richards pointed out.
In a pooled analysis, relative to control treatment, VEGFR TKI treatment was associated with a 2.23-fold increased risk of fatal adverse events (P = .02). But even so, the absolute rate of such events was just 1.5%, compared with 0.7% with the control treatment.
In stratified analyses, there was no significant difference in the relative risks across the three agents or the relative risks for renal cell carcinoma vs. other types of cancer.
The most common fatal adverse events in the trials overall (considering both control and VEGFR TKI arms) were hemorrhage (48% of the total), myocardial infarction (15%), and hepatic failure (10%). These events "occurred in both the placebo and control groups as well as study groups," Dr. Richards noted.
He acknowledged that limitations of the study included the fact that the analysis used study-level instead of patient-level data, which may have resulted in confounding, and that it did not have adequate power to detect differences between the three VEGFR TKIs, although they all have a similar mechanism of action.
The symposium was sponsored by the American Society of Clinical Oncology, the American Society for Radiation Oncology, and the Society of Urologic Oncology.
Dr. Richards disclosed that he had no relevant conflicts of interest. Dr. Hutson disclosed that he is a consultant to and receives honoraria from Bayer, Genentech, GlaxoSmithKline, Novartis, Onyx, Pfizer, and Wyeth, and that he receives research funding from GlaxoSmithKline, Pfizer, and Wyeth.
SAN FRANCISCO – Tyrosine kinase inhibitors that target the vascular endothelial growth factor receptor increase the risk of a fatal adverse event in cancer patients, but the absolute rate is still low, investigators concluded from a meta-analysis of 10 randomized trials with a total of 4,679 patients.
Trial participants given sorafenib (Nexavar), sunitinib (Sutent), or pazopanib (Votrient) were more than twice as likely to die from an adverse event as were peers given control treatment, said lead investigator Dr. Christopher J. Richards of the Beth Israel Deaconess Medical Center in Boston. Yet, the rate was still less than 2%.
"Our analysis may underestimate the true incidence of fatal adverse events, as study patients may be healthier than the general population exposed to these drugs," he noted at the Genitourinary Cancers Symposium. But that said, "all three of the study drugs have been shown in various randomized controlled trials to improve clinical outcomes compared to traditional therapies."
Discussant Dr. Thomas E. Hutson of the Baylor Sammons Cancer Center in Dallas commented, "I feel confident that everyone or almost everyone in the room today would agree with me that our current therapy benefit outweighs the risk associated with the treatment."
Moreover, even among the new targeted agents, the 1.5% rate seen with the vascular endothelial growth factor receptor (VEGFR) tyrosine kinase inhibitors (TKIs) in the meta-analysis "compares very favorably" with the rate of 2.9% seen previously with bevacizumab (Avastin), another VEGF-targeted therapy, in a similar analysis (JAMA 2011;305:487-94).
"Targeted therapies have real toxicities, and these toxicities include a low risk of death – a risk of death not much different than other therapies used for kidney and other cancer types," Dr. Hutson asserted. "If anything, this meta-analysis shows us that close monitoring is going to be essential for optimal use of these agents."
In the study, Dr. Richards and his colleagues identified 10 randomized controlled phase II or III trials in which patients received any of three VEGFR TKIs approved by the Food and Drug Administration for cancer treatment as of February 2011: sorafenib, sunitinib, or pazopanib. The control groups were usually treated with a placebo, but in some cases with chemotherapy or immunotherapy.
The enrolled patients had a variety of malignancies: renal cell carcinoma, hepatocellular carcinoma, melanoma, non–small cell lung cancer, breast cancer, and pancreatic neuroendocrine tumor. "All patients included in the studies had good performance status and had adequate baseline cardiac, hematologic, and renal function at the time of study entry," Dr. Richards pointed out.
In a pooled analysis, relative to control treatment, VEGFR TKI treatment was associated with a 2.23-fold increased risk of fatal adverse events (P = .02). But even so, the absolute rate of such events was just 1.5%, compared with 0.7% with the control treatment.
In stratified analyses, there was no significant difference in the relative risks across the three agents or the relative risks for renal cell carcinoma vs. other types of cancer.
The most common fatal adverse events in the trials overall (considering both control and VEGFR TKI arms) were hemorrhage (48% of the total), myocardial infarction (15%), and hepatic failure (10%). These events "occurred in both the placebo and control groups as well as study groups," Dr. Richards noted.
He acknowledged that limitations of the study included the fact that the analysis used study-level instead of patient-level data, which may have resulted in confounding, and that it did not have adequate power to detect differences between the three VEGFR TKIs, although they all have a similar mechanism of action.
The symposium was sponsored by the American Society of Clinical Oncology, the American Society for Radiation Oncology, and the Society of Urologic Oncology.
Dr. Richards disclosed that he had no relevant conflicts of interest. Dr. Hutson disclosed that he is a consultant to and receives honoraria from Bayer, Genentech, GlaxoSmithKline, Novartis, Onyx, Pfizer, and Wyeth, and that he receives research funding from GlaxoSmithKline, Pfizer, and Wyeth.
SAN FRANCISCO – Tyrosine kinase inhibitors that target the vascular endothelial growth factor receptor increase the risk of a fatal adverse event in cancer patients, but the absolute rate is still low, investigators concluded from a meta-analysis of 10 randomized trials with a total of 4,679 patients.
Trial participants given sorafenib (Nexavar), sunitinib (Sutent), or pazopanib (Votrient) were more than twice as likely to die from an adverse event as were peers given control treatment, said lead investigator Dr. Christopher J. Richards of the Beth Israel Deaconess Medical Center in Boston. Yet, the rate was still less than 2%.
"Our analysis may underestimate the true incidence of fatal adverse events, as study patients may be healthier than the general population exposed to these drugs," he noted at the Genitourinary Cancers Symposium. But that said, "all three of the study drugs have been shown in various randomized controlled trials to improve clinical outcomes compared to traditional therapies."
Discussant Dr. Thomas E. Hutson of the Baylor Sammons Cancer Center in Dallas commented, "I feel confident that everyone or almost everyone in the room today would agree with me that our current therapy benefit outweighs the risk associated with the treatment."
Moreover, even among the new targeted agents, the 1.5% rate seen with the vascular endothelial growth factor receptor (VEGFR) tyrosine kinase inhibitors (TKIs) in the meta-analysis "compares very favorably" with the rate of 2.9% seen previously with bevacizumab (Avastin), another VEGF-targeted therapy, in a similar analysis (JAMA 2011;305:487-94).
"Targeted therapies have real toxicities, and these toxicities include a low risk of death – a risk of death not much different than other therapies used for kidney and other cancer types," Dr. Hutson asserted. "If anything, this meta-analysis shows us that close monitoring is going to be essential for optimal use of these agents."
In the study, Dr. Richards and his colleagues identified 10 randomized controlled phase II or III trials in which patients received any of three VEGFR TKIs approved by the Food and Drug Administration for cancer treatment as of February 2011: sorafenib, sunitinib, or pazopanib. The control groups were usually treated with a placebo, but in some cases with chemotherapy or immunotherapy.
The enrolled patients had a variety of malignancies: renal cell carcinoma, hepatocellular carcinoma, melanoma, non–small cell lung cancer, breast cancer, and pancreatic neuroendocrine tumor. "All patients included in the studies had good performance status and had adequate baseline cardiac, hematologic, and renal function at the time of study entry," Dr. Richards pointed out.
In a pooled analysis, relative to control treatment, VEGFR TKI treatment was associated with a 2.23-fold increased risk of fatal adverse events (P = .02). But even so, the absolute rate of such events was just 1.5%, compared with 0.7% with the control treatment.
In stratified analyses, there was no significant difference in the relative risks across the three agents or the relative risks for renal cell carcinoma vs. other types of cancer.
The most common fatal adverse events in the trials overall (considering both control and VEGFR TKI arms) were hemorrhage (48% of the total), myocardial infarction (15%), and hepatic failure (10%). These events "occurred in both the placebo and control groups as well as study groups," Dr. Richards noted.
He acknowledged that limitations of the study included the fact that the analysis used study-level instead of patient-level data, which may have resulted in confounding, and that it did not have adequate power to detect differences between the three VEGFR TKIs, although they all have a similar mechanism of action.
The symposium was sponsored by the American Society of Clinical Oncology, the American Society for Radiation Oncology, and the Society of Urologic Oncology.
Dr. Richards disclosed that he had no relevant conflicts of interest. Dr. Hutson disclosed that he is a consultant to and receives honoraria from Bayer, Genentech, GlaxoSmithKline, Novartis, Onyx, Pfizer, and Wyeth, and that he receives research funding from GlaxoSmithKline, Pfizer, and Wyeth.
FROM THE GENITOURINARY CANCERS SYMPOSIUM
New Vemurafenib Data Highlight Long-Term Melanoma Survival
A phase-II clinical trial with the longest follow-up to date confirmed that vemurafenib induces a clinical response in more than half of patients with advanced melanoma expressing BRAFV600 mutations, according to a report in the Feb. 23 issue of the New England Journal of Medicine.
Median overall survival was 16 months in these patients, who had highly unfavorable characteristics at baseline. With follow-up as long as 20 months, this study "provides critical information on long-term survival" that was not available from previous studies of vemurafenib (Zelboraf) in metastatic melanoma, said Dr. Jeffrey A. Sosman of the Vanderbilt-Ingram Cancer Center, Nashville, Tenn., and his associates.
The Food and Drug Administration approved vemurafenib on Aug. 17, 2011 for patients with unresectable or metastatic melanoma with the BRAFV600E mutation as detected by an FDA-approved test. The FDA acted after a pivotal phase-III trial showed vemurafenib improved overall survival compared with dacarbazine. When the phase-III data were analyzed, median follow-up was just 6.2 months in the experimental arm, and median overall survival had not been reached for patients treated with vemurafenib.
The FDA also considered the currently reported phase-II trial, which included 132 patients with stage-IV melanoma that had progressed despite at least one systemic treatment. Tumor tissue from 56% of patients screened for the trial had a BRAFV600 mutation, which was required for study entry. Vemurafenib is a potent kinase inhibitor that was developed to target this mutation within cancer cells specifically.
Patients in the study were given oral vemurafenib (960 mg twice daily) and followed every 6 weeks for disease progression at 10 medical centers in the United States and 3 in Australia. Median follow-up for efficacy was 13 months (range, 0.6-20 months). Treatment response was assessed by both the investigative team and by a blinded independent review committee.
The committee found that 8 patients (6%) showed a complete response and 62 (47%) showed a partial response to vemurafenib, for an overall response rate of 53%. A total of 38 patients (29%) showed stable disease. Only 18 (14%) showed progressive disease; 6 patients could not be assessed.
Dr. Sosman and his colleagues found a 5% complete response rate and a 52% partial response rate, for an overall response rate of 57%, which generally accorded with the independent review committee’s findings.
Molecular sequencing showed 122 patients in the trial had BRAFV600E mutations and 10 patients had BRAFV600K mutations. Four of the 10 patients with the BRAFV600K mutation had a partial response and 3 had stable disease.
These response rates are higher than those associated with any previous treatments for advanced melanoma, the researchers noted. The lowest response occurred in patients with an LDL level more than 1.5 times the upper limit of normal.
The median duration of response was 6.7 months, and one-fourth of the subjects remained progression free for more than a year. Median overall survival was 16 months. At the time of the data cut-off (July 1, 2011), 62 patients (47% of the study population) were alive.
"Most objective responses were evident at the time of the first set of scans (week 6), but in some patients, responses did not appear until the patient had been receiving the drug for more than 6 months," the researchers said (N. Engl. J. Med. 2012;366:707-14).
The longer follow-up in this study thus permitted the investigators to observe these late responses.
Overall survival was 77% at 6 months and 58% at 12 months, and it was estimated to be 43% at 18 months, they noted.
Until now, median overall of metastatic melanoma has been 6-10 months, Dr. Sosman and his colleagues noted. In previously untreated patients who responded to ipilimumab (Yervoy) – an immunotherapy also approved last year – it reached 11 months.
Most patients had at least one adverse event related to vemurafenib, but most were not severe. Adverse effects chiefly were arthralgia, rash, photosensitivity, fatigue, and alopecia. Although many patients required dose interruptions (64%) or reductions (45%), "patients were able to receive most of their intended daily dose," the investigators wrote.
Four patients discontinued the study drug altogether, including one who developed a retinal-vein occlusion. Another patient died from rapidly progressive melanoma and renal failure that may have been related to the study drug.
Vemurafenib is known to have the paradoxical effect of promoting cutaneous squamous cell carcinomas or keratoacanthomas, so the study subjects were closely monitored for these. Thirty-four patients (26%) developed such neoplasms, usually at the start of therapy. All were managed with resection and vemurafenib was not discontinued. Another eight patients were found to have basal cell carcinomas.
"As with most targeted therapies that block a driver oncogene," vemurafenib will likely encounter resistance with long-term use, Dr. Sosman and his colleagues said. The mechanisms of resistance to vemurafenib are currently being investigated, so that strategies to prevent or overcome it can be devised.
This study was funded by Hoffman-LaRoche. Dr. Sosman and his associates reported ties to various companies, including Abraxis, Altor, Amgen, BMS, Celldex, Genentech, Genesis Biopharma, GlaxoSmithKline, Hoffman-La Roche, Merck, Novartis, Pfizer, Plexxikon, Prometheus, Roche, and Serametrix.
A phase-II clinical trial with the longest follow-up to date confirmed that vemurafenib induces a clinical response in more than half of patients with advanced melanoma expressing BRAFV600 mutations, according to a report in the Feb. 23 issue of the New England Journal of Medicine.
Median overall survival was 16 months in these patients, who had highly unfavorable characteristics at baseline. With follow-up as long as 20 months, this study "provides critical information on long-term survival" that was not available from previous studies of vemurafenib (Zelboraf) in metastatic melanoma, said Dr. Jeffrey A. Sosman of the Vanderbilt-Ingram Cancer Center, Nashville, Tenn., and his associates.
The Food and Drug Administration approved vemurafenib on Aug. 17, 2011 for patients with unresectable or metastatic melanoma with the BRAFV600E mutation as detected by an FDA-approved test. The FDA acted after a pivotal phase-III trial showed vemurafenib improved overall survival compared with dacarbazine. When the phase-III data were analyzed, median follow-up was just 6.2 months in the experimental arm, and median overall survival had not been reached for patients treated with vemurafenib.
The FDA also considered the currently reported phase-II trial, which included 132 patients with stage-IV melanoma that had progressed despite at least one systemic treatment. Tumor tissue from 56% of patients screened for the trial had a BRAFV600 mutation, which was required for study entry. Vemurafenib is a potent kinase inhibitor that was developed to target this mutation within cancer cells specifically.
Patients in the study were given oral vemurafenib (960 mg twice daily) and followed every 6 weeks for disease progression at 10 medical centers in the United States and 3 in Australia. Median follow-up for efficacy was 13 months (range, 0.6-20 months). Treatment response was assessed by both the investigative team and by a blinded independent review committee.
The committee found that 8 patients (6%) showed a complete response and 62 (47%) showed a partial response to vemurafenib, for an overall response rate of 53%. A total of 38 patients (29%) showed stable disease. Only 18 (14%) showed progressive disease; 6 patients could not be assessed.
Dr. Sosman and his colleagues found a 5% complete response rate and a 52% partial response rate, for an overall response rate of 57%, which generally accorded with the independent review committee’s findings.
Molecular sequencing showed 122 patients in the trial had BRAFV600E mutations and 10 patients had BRAFV600K mutations. Four of the 10 patients with the BRAFV600K mutation had a partial response and 3 had stable disease.
These response rates are higher than those associated with any previous treatments for advanced melanoma, the researchers noted. The lowest response occurred in patients with an LDL level more than 1.5 times the upper limit of normal.
The median duration of response was 6.7 months, and one-fourth of the subjects remained progression free for more than a year. Median overall survival was 16 months. At the time of the data cut-off (July 1, 2011), 62 patients (47% of the study population) were alive.
"Most objective responses were evident at the time of the first set of scans (week 6), but in some patients, responses did not appear until the patient had been receiving the drug for more than 6 months," the researchers said (N. Engl. J. Med. 2012;366:707-14).
The longer follow-up in this study thus permitted the investigators to observe these late responses.
Overall survival was 77% at 6 months and 58% at 12 months, and it was estimated to be 43% at 18 months, they noted.
Until now, median overall of metastatic melanoma has been 6-10 months, Dr. Sosman and his colleagues noted. In previously untreated patients who responded to ipilimumab (Yervoy) – an immunotherapy also approved last year – it reached 11 months.
Most patients had at least one adverse event related to vemurafenib, but most were not severe. Adverse effects chiefly were arthralgia, rash, photosensitivity, fatigue, and alopecia. Although many patients required dose interruptions (64%) or reductions (45%), "patients were able to receive most of their intended daily dose," the investigators wrote.
Four patients discontinued the study drug altogether, including one who developed a retinal-vein occlusion. Another patient died from rapidly progressive melanoma and renal failure that may have been related to the study drug.
Vemurafenib is known to have the paradoxical effect of promoting cutaneous squamous cell carcinomas or keratoacanthomas, so the study subjects were closely monitored for these. Thirty-four patients (26%) developed such neoplasms, usually at the start of therapy. All were managed with resection and vemurafenib was not discontinued. Another eight patients were found to have basal cell carcinomas.
"As with most targeted therapies that block a driver oncogene," vemurafenib will likely encounter resistance with long-term use, Dr. Sosman and his colleagues said. The mechanisms of resistance to vemurafenib are currently being investigated, so that strategies to prevent or overcome it can be devised.
This study was funded by Hoffman-LaRoche. Dr. Sosman and his associates reported ties to various companies, including Abraxis, Altor, Amgen, BMS, Celldex, Genentech, Genesis Biopharma, GlaxoSmithKline, Hoffman-La Roche, Merck, Novartis, Pfizer, Plexxikon, Prometheus, Roche, and Serametrix.
A phase-II clinical trial with the longest follow-up to date confirmed that vemurafenib induces a clinical response in more than half of patients with advanced melanoma expressing BRAFV600 mutations, according to a report in the Feb. 23 issue of the New England Journal of Medicine.
Median overall survival was 16 months in these patients, who had highly unfavorable characteristics at baseline. With follow-up as long as 20 months, this study "provides critical information on long-term survival" that was not available from previous studies of vemurafenib (Zelboraf) in metastatic melanoma, said Dr. Jeffrey A. Sosman of the Vanderbilt-Ingram Cancer Center, Nashville, Tenn., and his associates.
The Food and Drug Administration approved vemurafenib on Aug. 17, 2011 for patients with unresectable or metastatic melanoma with the BRAFV600E mutation as detected by an FDA-approved test. The FDA acted after a pivotal phase-III trial showed vemurafenib improved overall survival compared with dacarbazine. When the phase-III data were analyzed, median follow-up was just 6.2 months in the experimental arm, and median overall survival had not been reached for patients treated with vemurafenib.
The FDA also considered the currently reported phase-II trial, which included 132 patients with stage-IV melanoma that had progressed despite at least one systemic treatment. Tumor tissue from 56% of patients screened for the trial had a BRAFV600 mutation, which was required for study entry. Vemurafenib is a potent kinase inhibitor that was developed to target this mutation within cancer cells specifically.
Patients in the study were given oral vemurafenib (960 mg twice daily) and followed every 6 weeks for disease progression at 10 medical centers in the United States and 3 in Australia. Median follow-up for efficacy was 13 months (range, 0.6-20 months). Treatment response was assessed by both the investigative team and by a blinded independent review committee.
The committee found that 8 patients (6%) showed a complete response and 62 (47%) showed a partial response to vemurafenib, for an overall response rate of 53%. A total of 38 patients (29%) showed stable disease. Only 18 (14%) showed progressive disease; 6 patients could not be assessed.
Dr. Sosman and his colleagues found a 5% complete response rate and a 52% partial response rate, for an overall response rate of 57%, which generally accorded with the independent review committee’s findings.
Molecular sequencing showed 122 patients in the trial had BRAFV600E mutations and 10 patients had BRAFV600K mutations. Four of the 10 patients with the BRAFV600K mutation had a partial response and 3 had stable disease.
These response rates are higher than those associated with any previous treatments for advanced melanoma, the researchers noted. The lowest response occurred in patients with an LDL level more than 1.5 times the upper limit of normal.
The median duration of response was 6.7 months, and one-fourth of the subjects remained progression free for more than a year. Median overall survival was 16 months. At the time of the data cut-off (July 1, 2011), 62 patients (47% of the study population) were alive.
"Most objective responses were evident at the time of the first set of scans (week 6), but in some patients, responses did not appear until the patient had been receiving the drug for more than 6 months," the researchers said (N. Engl. J. Med. 2012;366:707-14).
The longer follow-up in this study thus permitted the investigators to observe these late responses.
Overall survival was 77% at 6 months and 58% at 12 months, and it was estimated to be 43% at 18 months, they noted.
Until now, median overall of metastatic melanoma has been 6-10 months, Dr. Sosman and his colleagues noted. In previously untreated patients who responded to ipilimumab (Yervoy) – an immunotherapy also approved last year – it reached 11 months.
Most patients had at least one adverse event related to vemurafenib, but most were not severe. Adverse effects chiefly were arthralgia, rash, photosensitivity, fatigue, and alopecia. Although many patients required dose interruptions (64%) or reductions (45%), "patients were able to receive most of their intended daily dose," the investigators wrote.
Four patients discontinued the study drug altogether, including one who developed a retinal-vein occlusion. Another patient died from rapidly progressive melanoma and renal failure that may have been related to the study drug.
Vemurafenib is known to have the paradoxical effect of promoting cutaneous squamous cell carcinomas or keratoacanthomas, so the study subjects were closely monitored for these. Thirty-four patients (26%) developed such neoplasms, usually at the start of therapy. All were managed with resection and vemurafenib was not discontinued. Another eight patients were found to have basal cell carcinomas.
"As with most targeted therapies that block a driver oncogene," vemurafenib will likely encounter resistance with long-term use, Dr. Sosman and his colleagues said. The mechanisms of resistance to vemurafenib are currently being investigated, so that strategies to prevent or overcome it can be devised.
This study was funded by Hoffman-LaRoche. Dr. Sosman and his associates reported ties to various companies, including Abraxis, Altor, Amgen, BMS, Celldex, Genentech, Genesis Biopharma, GlaxoSmithKline, Hoffman-La Roche, Merck, Novartis, Pfizer, Plexxikon, Prometheus, Roche, and Serametrix.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: The overall response rate to vemurafenib was 53%, and median overall survival was 16 months.
Data Source: The multicenter phase II clinical trial involved 132 patients who had advanced melanoma that expressed BRAFV600 mutations and were followed for as long as 20 months.
Disclosures: This study was funded by Hoffman-La Roche. Dr. Sosman and his associates reported ties to various companies, including Abraxis, Altor, Amgen, BMS, Celldex, Genentech, Genesis Biopharma, GlaxoSmithKline, Hoffman-La Roche, Merck, Novartis, Pfizer, Plexxikon, Prometheus, Roche, and Serametrix.
Identifying AK Variants Is Key to Management
The ability to distinguish between the two key variants of actinic keratosis is key to effective management, according to Dr. Theodore Rosen.
Pigmented actinic keratosis (AK) that spreads typically occurs in patients older than age 60 years and affects more women than men. The lesions are larger than 1.5 cm and have a history of lateral spread. "The lesions are flat, with a smooth, scaly surface, and they occur on sun-exposed skin, mostly the face, and near other manifestations of photodamage," noted Dr. Rosen at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Differential diagnosis includes solar lentigo, lentigo maligna, lentigo maligna melanoma, seborrheic keratosis, and pigmented squamous cell carcinoma in situ. "The presence of dots, globules, and asymmetry on dermatoscopy can help you distinguish spreading pigmented AKs from other lesions," said Dr. Rosen, professor of dermatology at Baylor College of Medicine, Houston.
The other main variant, proliferating AK, also tends to affect patients older than age 60 but it occurs more commonly in men than in women. This variant is greater than 1.5 cm in size with a history of rapid lateral spread, and it is marked by a confluent scaly surface with some palpability. Affected areas include sun-exposed skin, mostly the face, and areas near other manifestations of photodamage. "This variant is resistant to usual topical therapy, and the differential diagnosis may include eczema, psoriasis, squamous cell carcinoma in situ, and superficial basal cell carcinoma," said Dr. Rosen, who is also chief of the dermatology clinic at the Houston-based Michael E. DeBakey VA Medical Center. Histology reveals fingerlike invasive projections into the dermis of atypical squamous cells.
The appearance of multiple AKs in a cosmetic unit indicates that the clinician should consider the entire photodamaged area to be diseased. This concept, known as field-directed therapy, "not only addresses apparent clinical lesions, but also reveals and may treat subclinical lesions," he said. "Field therapy may also offer the advantage of reversing basic cellular aberrations leading to transition into invasive cutaneous malignancy."
Another trend in the treatment of AKs is the development of new imiquimod formulations. Results from two placebo-controlled studies of daily application of 2.5% and 3.75% imiquimod creams to the face and balding scalp for two 3-week cycles revealed that both formulations were more effective than placebo and had an acceptable safety profile (J. Am. Acad. Dermatol. 2010;62:573-81). The final evaluation occurred 8 weeks after the final treatment, said Dr. Rosen, who was one of the study investigators. Results from this and other studies led to Food and Drug Administration approval of imiquimod 2.5% (Zyclara) for AKs on July 19, 2011, but the formulation is not yet commercially available.
In a more recent analysis, investigators assessed complete clearance rates in AK patients from four phase III trials of imiquimod who were followed for 1 year (J. Drugs Dermatol. 2011;10:165-70). For imiquimod 3.75% and 2.5% cream, respectively, complete clearance was sustained for 1 year in 40.5% and 33.3% of patients from the 2-week cycle studies, and in 47.9% and 43.2% of patients from the 3-week cycle studies.
Combination therapies for AKs have become increasingly popular. Treatments include cryosurgery followed by diclofenac, 5-fluorouracil followed by cryosurgery, cryosurgery followed by imiquimod 5% or 3.75%, photodynamic therapy (PTD) followed by imiquimod 5%, and tazarotene followed by PDT. "For all studies where field therapy is combined with lesion-directed therapy or with another field therapy, the combination proved superior to monotherapy," Dr. Rosen said.
He went on to note that there is considerable user variation – such as time, the distance the device is held from the skin, and the type of unit used – in the delivery of cryosurgery to treat AKs. The variation "may have more to do with skin temperature than any other variable," said Dr. Rosen. In one study, investigators used an infrared detector in a liquid nitrogen container to deliver cryotherapy to a temperature of –5° C in 36 patients with 180 thin AKs (Dermatol. Surg. 2010;36:1956-61). The clearance rates were 67% at 1 week and 100% at 6 weeks.
Treatment of AKs with radiation is another area of study. In one recent trial, 12 patients with refractory AK and 4 patients with AKs near a nonmelanoma skin cancer underwent external beam radiation therapy five times a week for 4-6 weeks. The total dose of radiation ranged from 4 to 60 Gy (J. Clin. Aesthet. Dermatol. 2011;4:47-50). Of the 16 patients, 13 had complete resolution of AKs, 1 noted a prompt recurrence, and 90% said they were "very satisfied" with the treatment results.
Treating AKs with methyl aminolevulinate photodynamic therapy (MAL-PDT) is also being investigated. In a multicenter study conducted in Scandinavia, 120 patients with 1,572 AKs underwent MAL-PDT followed by immediate exposure to the sun for 1.5-2.5 hours (Br. J. Dermatol. 2011;164:1083-90). At 3 months, 75%-77% of the lesions had cleared. This approach "has an advantage for the health care provider in that no equipment is needed," Dr. Rosen said. "The advantage for the patient is that the therapy is nearly pain free."
Dr. Rosen disclosed that he is a consultant and a member of the speakers bureau for Medicis, which makes imiquimod. He is also a consultant for LEO Pharma.
SDEF and this news organization are owned by Elsevier.
The ability to distinguish between the two key variants of actinic keratosis is key to effective management, according to Dr. Theodore Rosen.
Pigmented actinic keratosis (AK) that spreads typically occurs in patients older than age 60 years and affects more women than men. The lesions are larger than 1.5 cm and have a history of lateral spread. "The lesions are flat, with a smooth, scaly surface, and they occur on sun-exposed skin, mostly the face, and near other manifestations of photodamage," noted Dr. Rosen at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Differential diagnosis includes solar lentigo, lentigo maligna, lentigo maligna melanoma, seborrheic keratosis, and pigmented squamous cell carcinoma in situ. "The presence of dots, globules, and asymmetry on dermatoscopy can help you distinguish spreading pigmented AKs from other lesions," said Dr. Rosen, professor of dermatology at Baylor College of Medicine, Houston.
The other main variant, proliferating AK, also tends to affect patients older than age 60 but it occurs more commonly in men than in women. This variant is greater than 1.5 cm in size with a history of rapid lateral spread, and it is marked by a confluent scaly surface with some palpability. Affected areas include sun-exposed skin, mostly the face, and areas near other manifestations of photodamage. "This variant is resistant to usual topical therapy, and the differential diagnosis may include eczema, psoriasis, squamous cell carcinoma in situ, and superficial basal cell carcinoma," said Dr. Rosen, who is also chief of the dermatology clinic at the Houston-based Michael E. DeBakey VA Medical Center. Histology reveals fingerlike invasive projections into the dermis of atypical squamous cells.
The appearance of multiple AKs in a cosmetic unit indicates that the clinician should consider the entire photodamaged area to be diseased. This concept, known as field-directed therapy, "not only addresses apparent clinical lesions, but also reveals and may treat subclinical lesions," he said. "Field therapy may also offer the advantage of reversing basic cellular aberrations leading to transition into invasive cutaneous malignancy."
Another trend in the treatment of AKs is the development of new imiquimod formulations. Results from two placebo-controlled studies of daily application of 2.5% and 3.75% imiquimod creams to the face and balding scalp for two 3-week cycles revealed that both formulations were more effective than placebo and had an acceptable safety profile (J. Am. Acad. Dermatol. 2010;62:573-81). The final evaluation occurred 8 weeks after the final treatment, said Dr. Rosen, who was one of the study investigators. Results from this and other studies led to Food and Drug Administration approval of imiquimod 2.5% (Zyclara) for AKs on July 19, 2011, but the formulation is not yet commercially available.
In a more recent analysis, investigators assessed complete clearance rates in AK patients from four phase III trials of imiquimod who were followed for 1 year (J. Drugs Dermatol. 2011;10:165-70). For imiquimod 3.75% and 2.5% cream, respectively, complete clearance was sustained for 1 year in 40.5% and 33.3% of patients from the 2-week cycle studies, and in 47.9% and 43.2% of patients from the 3-week cycle studies.
Combination therapies for AKs have become increasingly popular. Treatments include cryosurgery followed by diclofenac, 5-fluorouracil followed by cryosurgery, cryosurgery followed by imiquimod 5% or 3.75%, photodynamic therapy (PTD) followed by imiquimod 5%, and tazarotene followed by PDT. "For all studies where field therapy is combined with lesion-directed therapy or with another field therapy, the combination proved superior to monotherapy," Dr. Rosen said.
He went on to note that there is considerable user variation – such as time, the distance the device is held from the skin, and the type of unit used – in the delivery of cryosurgery to treat AKs. The variation "may have more to do with skin temperature than any other variable," said Dr. Rosen. In one study, investigators used an infrared detector in a liquid nitrogen container to deliver cryotherapy to a temperature of –5° C in 36 patients with 180 thin AKs (Dermatol. Surg. 2010;36:1956-61). The clearance rates were 67% at 1 week and 100% at 6 weeks.
Treatment of AKs with radiation is another area of study. In one recent trial, 12 patients with refractory AK and 4 patients with AKs near a nonmelanoma skin cancer underwent external beam radiation therapy five times a week for 4-6 weeks. The total dose of radiation ranged from 4 to 60 Gy (J. Clin. Aesthet. Dermatol. 2011;4:47-50). Of the 16 patients, 13 had complete resolution of AKs, 1 noted a prompt recurrence, and 90% said they were "very satisfied" with the treatment results.
Treating AKs with methyl aminolevulinate photodynamic therapy (MAL-PDT) is also being investigated. In a multicenter study conducted in Scandinavia, 120 patients with 1,572 AKs underwent MAL-PDT followed by immediate exposure to the sun for 1.5-2.5 hours (Br. J. Dermatol. 2011;164:1083-90). At 3 months, 75%-77% of the lesions had cleared. This approach "has an advantage for the health care provider in that no equipment is needed," Dr. Rosen said. "The advantage for the patient is that the therapy is nearly pain free."
Dr. Rosen disclosed that he is a consultant and a member of the speakers bureau for Medicis, which makes imiquimod. He is also a consultant for LEO Pharma.
SDEF and this news organization are owned by Elsevier.
The ability to distinguish between the two key variants of actinic keratosis is key to effective management, according to Dr. Theodore Rosen.
Pigmented actinic keratosis (AK) that spreads typically occurs in patients older than age 60 years and affects more women than men. The lesions are larger than 1.5 cm and have a history of lateral spread. "The lesions are flat, with a smooth, scaly surface, and they occur on sun-exposed skin, mostly the face, and near other manifestations of photodamage," noted Dr. Rosen at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Differential diagnosis includes solar lentigo, lentigo maligna, lentigo maligna melanoma, seborrheic keratosis, and pigmented squamous cell carcinoma in situ. "The presence of dots, globules, and asymmetry on dermatoscopy can help you distinguish spreading pigmented AKs from other lesions," said Dr. Rosen, professor of dermatology at Baylor College of Medicine, Houston.
The other main variant, proliferating AK, also tends to affect patients older than age 60 but it occurs more commonly in men than in women. This variant is greater than 1.5 cm in size with a history of rapid lateral spread, and it is marked by a confluent scaly surface with some palpability. Affected areas include sun-exposed skin, mostly the face, and areas near other manifestations of photodamage. "This variant is resistant to usual topical therapy, and the differential diagnosis may include eczema, psoriasis, squamous cell carcinoma in situ, and superficial basal cell carcinoma," said Dr. Rosen, who is also chief of the dermatology clinic at the Houston-based Michael E. DeBakey VA Medical Center. Histology reveals fingerlike invasive projections into the dermis of atypical squamous cells.
The appearance of multiple AKs in a cosmetic unit indicates that the clinician should consider the entire photodamaged area to be diseased. This concept, known as field-directed therapy, "not only addresses apparent clinical lesions, but also reveals and may treat subclinical lesions," he said. "Field therapy may also offer the advantage of reversing basic cellular aberrations leading to transition into invasive cutaneous malignancy."
Another trend in the treatment of AKs is the development of new imiquimod formulations. Results from two placebo-controlled studies of daily application of 2.5% and 3.75% imiquimod creams to the face and balding scalp for two 3-week cycles revealed that both formulations were more effective than placebo and had an acceptable safety profile (J. Am. Acad. Dermatol. 2010;62:573-81). The final evaluation occurred 8 weeks after the final treatment, said Dr. Rosen, who was one of the study investigators. Results from this and other studies led to Food and Drug Administration approval of imiquimod 2.5% (Zyclara) for AKs on July 19, 2011, but the formulation is not yet commercially available.
In a more recent analysis, investigators assessed complete clearance rates in AK patients from four phase III trials of imiquimod who were followed for 1 year (J. Drugs Dermatol. 2011;10:165-70). For imiquimod 3.75% and 2.5% cream, respectively, complete clearance was sustained for 1 year in 40.5% and 33.3% of patients from the 2-week cycle studies, and in 47.9% and 43.2% of patients from the 3-week cycle studies.
Combination therapies for AKs have become increasingly popular. Treatments include cryosurgery followed by diclofenac, 5-fluorouracil followed by cryosurgery, cryosurgery followed by imiquimod 5% or 3.75%, photodynamic therapy (PTD) followed by imiquimod 5%, and tazarotene followed by PDT. "For all studies where field therapy is combined with lesion-directed therapy or with another field therapy, the combination proved superior to monotherapy," Dr. Rosen said.
He went on to note that there is considerable user variation – such as time, the distance the device is held from the skin, and the type of unit used – in the delivery of cryosurgery to treat AKs. The variation "may have more to do with skin temperature than any other variable," said Dr. Rosen. In one study, investigators used an infrared detector in a liquid nitrogen container to deliver cryotherapy to a temperature of –5° C in 36 patients with 180 thin AKs (Dermatol. Surg. 2010;36:1956-61). The clearance rates were 67% at 1 week and 100% at 6 weeks.
Treatment of AKs with radiation is another area of study. In one recent trial, 12 patients with refractory AK and 4 patients with AKs near a nonmelanoma skin cancer underwent external beam radiation therapy five times a week for 4-6 weeks. The total dose of radiation ranged from 4 to 60 Gy (J. Clin. Aesthet. Dermatol. 2011;4:47-50). Of the 16 patients, 13 had complete resolution of AKs, 1 noted a prompt recurrence, and 90% said they were "very satisfied" with the treatment results.
Treating AKs with methyl aminolevulinate photodynamic therapy (MAL-PDT) is also being investigated. In a multicenter study conducted in Scandinavia, 120 patients with 1,572 AKs underwent MAL-PDT followed by immediate exposure to the sun for 1.5-2.5 hours (Br. J. Dermatol. 2011;164:1083-90). At 3 months, 75%-77% of the lesions had cleared. This approach "has an advantage for the health care provider in that no equipment is needed," Dr. Rosen said. "The advantage for the patient is that the therapy is nearly pain free."
Dr. Rosen disclosed that he is a consultant and a member of the speakers bureau for Medicis, which makes imiquimod. He is also a consultant for LEO Pharma.
SDEF and this news organization are owned by Elsevier.
EXPERT ANALYSIS FROM THE SDEF HAWAII DERMATOLOGY SEMINAR
Posttransplant Head and Neck Tumors Tallied
MIAMI BEACH – Patients who have undergone solid organ transplantation are at greater risk for subsequent tumor development, and head and neck cancers can be particularly aggressive, according to results of a single-institution study.
Dr. Robert H. Deeb and his associates at Henry Ford Hospital in Detroit studied 3,639 patients who underwent solid organ transplantation between January 1990 and December 2011. By retrospectively searching electronic medical records, they identified 95 people who developed cutaneous, salivary gland, or mucosal malignancies.
They found a 2.1% incidence of cutaneous cancers and a 0.5% incidence of noncutaneous head and neck cancers in this population. Despite a relatively low overall 2.6% incidence, there are reasons for concern, Dr. Deeb said at the Triological Society’s Combined Sections Meeting, which was jointly sponsored by the Triological Society and the American College of Surgeons. When cutaneous cancers did occur, they were more aggressive and were associated with shorter 1-year survival rates.
Henry Ford Transplant Institute maintains a noncutaneous tumor registry. This allowed the researchers to compare survival and other factors. Compared with the "nontransplant tumor registry in our institution, we found significantly fewer patients were alive at 1 year," Dr. Deeb said. Specifically, 55% of posttransplant patients with these cancers survived to 1 year, compared with 81% of nontransplant patients. There were no significant differences between groups in terms of age, sex, stage at diagnosis, or 5-year survival.
More than half (52%) of the patients who developed skin cancers had multiple head and neck cutaneous malignancies, pointing to the aggressive nature of these cancers, said Dr. Deeb of the department of otolaryngology–head and neck surgery at Henry Ford Hospital.
"We believe these patients require aggressive screening, treatment, and follow-up," Dr. Deeb said.
Of the 78 cutaneous head and neck cancers, 51% were squamous cell carcinomas, 36% were basal cell carcinomas, and the remaining 13% were other skin cancer types. The cheek and scalp were the most common sites. The patients’ average age was 61 years at the time of diagnosis, and 74% were men. Skin cancer developed a mean 4 years after transplant surgery.
Four patients had a salivary gland cancer and 13 had an upper aerodigestive tract mucosal malignancy. The average patient age in this group was 60 years, and 94% were men. Cancer was diagnosed a mean 66 months post transplantation. All 13 of the mucosal malignancies in the study were squamous cell carcinomas, Dr. Deeb said.
Kidney, liver, and heart were the most commonly transplanted organs among patients who ultimately developed these head and neck malignancies.
Although immunosuppressants are universally prescribed to prevent organ rejection following transplant surgery, the exact mechanism for subsequent cancer development remains unknown, Dr. Deeb said. Loss of immunosurveillance is one possibility; uncontrolled cell proliferation from chronic and low-level antigenic stimulation throughout the body is another. In addition, he said, immunosuppressants might somehow activate oncogenic viruses.
Despite the findings of this study, Dr. Deeb said that the benefits of solid organ transplantation still generally outweigh the risk of subsequent cancer development.
Henry Ford Hospital funded the study. Dr. Deeb said that he had no relevant disclosures.
MIAMI BEACH – Patients who have undergone solid organ transplantation are at greater risk for subsequent tumor development, and head and neck cancers can be particularly aggressive, according to results of a single-institution study.
Dr. Robert H. Deeb and his associates at Henry Ford Hospital in Detroit studied 3,639 patients who underwent solid organ transplantation between January 1990 and December 2011. By retrospectively searching electronic medical records, they identified 95 people who developed cutaneous, salivary gland, or mucosal malignancies.
They found a 2.1% incidence of cutaneous cancers and a 0.5% incidence of noncutaneous head and neck cancers in this population. Despite a relatively low overall 2.6% incidence, there are reasons for concern, Dr. Deeb said at the Triological Society’s Combined Sections Meeting, which was jointly sponsored by the Triological Society and the American College of Surgeons. When cutaneous cancers did occur, they were more aggressive and were associated with shorter 1-year survival rates.
Henry Ford Transplant Institute maintains a noncutaneous tumor registry. This allowed the researchers to compare survival and other factors. Compared with the "nontransplant tumor registry in our institution, we found significantly fewer patients were alive at 1 year," Dr. Deeb said. Specifically, 55% of posttransplant patients with these cancers survived to 1 year, compared with 81% of nontransplant patients. There were no significant differences between groups in terms of age, sex, stage at diagnosis, or 5-year survival.
More than half (52%) of the patients who developed skin cancers had multiple head and neck cutaneous malignancies, pointing to the aggressive nature of these cancers, said Dr. Deeb of the department of otolaryngology–head and neck surgery at Henry Ford Hospital.
"We believe these patients require aggressive screening, treatment, and follow-up," Dr. Deeb said.
Of the 78 cutaneous head and neck cancers, 51% were squamous cell carcinomas, 36% were basal cell carcinomas, and the remaining 13% were other skin cancer types. The cheek and scalp were the most common sites. The patients’ average age was 61 years at the time of diagnosis, and 74% were men. Skin cancer developed a mean 4 years after transplant surgery.
Four patients had a salivary gland cancer and 13 had an upper aerodigestive tract mucosal malignancy. The average patient age in this group was 60 years, and 94% were men. Cancer was diagnosed a mean 66 months post transplantation. All 13 of the mucosal malignancies in the study were squamous cell carcinomas, Dr. Deeb said.
Kidney, liver, and heart were the most commonly transplanted organs among patients who ultimately developed these head and neck malignancies.
Although immunosuppressants are universally prescribed to prevent organ rejection following transplant surgery, the exact mechanism for subsequent cancer development remains unknown, Dr. Deeb said. Loss of immunosurveillance is one possibility; uncontrolled cell proliferation from chronic and low-level antigenic stimulation throughout the body is another. In addition, he said, immunosuppressants might somehow activate oncogenic viruses.
Despite the findings of this study, Dr. Deeb said that the benefits of solid organ transplantation still generally outweigh the risk of subsequent cancer development.
Henry Ford Hospital funded the study. Dr. Deeb said that he had no relevant disclosures.
MIAMI BEACH – Patients who have undergone solid organ transplantation are at greater risk for subsequent tumor development, and head and neck cancers can be particularly aggressive, according to results of a single-institution study.
Dr. Robert H. Deeb and his associates at Henry Ford Hospital in Detroit studied 3,639 patients who underwent solid organ transplantation between January 1990 and December 2011. By retrospectively searching electronic medical records, they identified 95 people who developed cutaneous, salivary gland, or mucosal malignancies.
They found a 2.1% incidence of cutaneous cancers and a 0.5% incidence of noncutaneous head and neck cancers in this population. Despite a relatively low overall 2.6% incidence, there are reasons for concern, Dr. Deeb said at the Triological Society’s Combined Sections Meeting, which was jointly sponsored by the Triological Society and the American College of Surgeons. When cutaneous cancers did occur, they were more aggressive and were associated with shorter 1-year survival rates.
Henry Ford Transplant Institute maintains a noncutaneous tumor registry. This allowed the researchers to compare survival and other factors. Compared with the "nontransplant tumor registry in our institution, we found significantly fewer patients were alive at 1 year," Dr. Deeb said. Specifically, 55% of posttransplant patients with these cancers survived to 1 year, compared with 81% of nontransplant patients. There were no significant differences between groups in terms of age, sex, stage at diagnosis, or 5-year survival.
More than half (52%) of the patients who developed skin cancers had multiple head and neck cutaneous malignancies, pointing to the aggressive nature of these cancers, said Dr. Deeb of the department of otolaryngology–head and neck surgery at Henry Ford Hospital.
"We believe these patients require aggressive screening, treatment, and follow-up," Dr. Deeb said.
Of the 78 cutaneous head and neck cancers, 51% were squamous cell carcinomas, 36% were basal cell carcinomas, and the remaining 13% were other skin cancer types. The cheek and scalp were the most common sites. The patients’ average age was 61 years at the time of diagnosis, and 74% were men. Skin cancer developed a mean 4 years after transplant surgery.
Four patients had a salivary gland cancer and 13 had an upper aerodigestive tract mucosal malignancy. The average patient age in this group was 60 years, and 94% were men. Cancer was diagnosed a mean 66 months post transplantation. All 13 of the mucosal malignancies in the study were squamous cell carcinomas, Dr. Deeb said.
Kidney, liver, and heart were the most commonly transplanted organs among patients who ultimately developed these head and neck malignancies.
Although immunosuppressants are universally prescribed to prevent organ rejection following transplant surgery, the exact mechanism for subsequent cancer development remains unknown, Dr. Deeb said. Loss of immunosurveillance is one possibility; uncontrolled cell proliferation from chronic and low-level antigenic stimulation throughout the body is another. In addition, he said, immunosuppressants might somehow activate oncogenic viruses.
Despite the findings of this study, Dr. Deeb said that the benefits of solid organ transplantation still generally outweigh the risk of subsequent cancer development.
Henry Ford Hospital funded the study. Dr. Deeb said that he had no relevant disclosures.
FROM THE TRIOLOGICAL SOCIETY'S COMBINED SECTIONS MEETING
Major Finding: A total of 95 people who underwent solid organ transplantation over 21 years developed subsequent head and neck cancer, for an incidence of 2.6%.
Data Source: Data are taken from a retrospective study of 3,639 solid organ transplant patients treated at the Henry Ford Hospital since 1990.
Disclosures: Henry Ford Hospital sponsored the study. Dr. Deeb reported having no financial disclosures.
Observation Okayed as Option for Some Skin Cancers
MIAMI BEACH – Observation or a "watchful waiting" approach is an acceptable option vs. re-excision for patients with positive margins remaining after removal of head and neck skin cancers, according to a retrospective study.
"A lot of times we take these lesions off and they appear to be benign, not really knowing what the preoperative histologic diagnosis is. [Then we] find out the diagnosis is cancer, and not only that, but they have a positive margin." Dr. Justin Douglas said at the Triological Society Combined Sections meeting.
The question is, he said, "Do you have to rush them back in [and re-excise] or can you sit on them for a while?"
"Dr. Douglas addresses an interesting question," said session moderator Dr. Jeffrey H. Spiegel, chief of the division of facial plastic and reconstructive surgery at Boston University Medical Center and otolaryngology faculty member at Boston University. "I see a lot of people who had squamous cells and basal cells excised sent back for re-excision ... and he’ll help us understand if I even need to be doing that."
Dr. Douglas and his colleagues studied 492 patients at the Clarksburg Veterans Affairs Hospital in Morgantown, W.Va. All had skin cancers removed from their head or neck, including 387 basal cell carcinomas and 105 squamous cell carcinomas, over 5 years.
"Do you have to rush them back in [and re-excise] or can you sit on them for a while?"
About 40% or 197 lesions had a negative margin after wide local excision. The remaining 295 lesions featured a positive margin on permanent section analysis. The positive lesions included 232 basal cell carcinomas (BCCs) and 63 squamous cell carcinomas (SCCs).
"In the group of 232 positive basal cell lesions, everybody was offered re-excision. A total 105 chose re-excision and 127 decided to just watch it, despite our best judgment," said Dr. Douglas, an otolaryngologist at the Morgantown V.A.
"Even of the squamous cell patients, 26 still said they wanted to watch it," Dr. Douglas said at the meeting, which was cosponsored by the Triological Society and the American College of Surgeons.
Of the positive margin group, none of the BCC and four (3.8%) of the SCC lesions recurred within a median follow-up of 3.7 years. This finding suggests a role for clinical observation of low-risk, nonmelanomatous cancers, Dr. Douglas said.
The four SCC lesions that recurred included one lesion in the observation-only group and three in the re-excision group. Some of these patients likely had a more aggressive form of cancer, Dr. Douglas said.
"Essentially, we feel what we learned from this study is that ... given the fact that you can handle any recurrence of these lesions if they occur, you can watch the basal cells, even with positive margins," Dr. Douglas said. "We still feel all squamous cell lesions should be taken out until you get a clear margin."
Why the majority of positive margin patients who chose observation did not experience recurrence is poorly understood, Dr. Douglas said. However, it could be the patient’s immune system, secondary to the wound healing process, comes in and cleans up any residual cells. Another possibility is that patients die from other causes before recurrence can occur. "The truth of the matter is: We don’t know. We need more studies to figure this out."
The nose was the most common site for the T1, nonmelanoma lesions. The average size was 1.2 cm. Mean patient age was 61 years and 484 (98%) were men. Patients were excluded from the study if their lesion was previously treated or they chose an option other than re-excision or observation.
Although initial excision by a Mohs surgeon would be ideal, Dr. Douglas said, in his study dermatologists and otolaryngologists removed the lesions at one of the hospital ambulatory clinics. This strategy reflects some real-world limitations. "The problem is, at least in West Virginia, Mohs surgeons are very sparse. There are probably only a handful in the state." Operating room time and scheduling (particularly for patients with many lesions), availability of a pathologist to read frozen sections, and costs are additional challenges.
Dr. Douglas and Dr. Spiegel said that they had no relevant financial disclosures.
MIAMI BEACH – Observation or a "watchful waiting" approach is an acceptable option vs. re-excision for patients with positive margins remaining after removal of head and neck skin cancers, according to a retrospective study.
"A lot of times we take these lesions off and they appear to be benign, not really knowing what the preoperative histologic diagnosis is. [Then we] find out the diagnosis is cancer, and not only that, but they have a positive margin." Dr. Justin Douglas said at the Triological Society Combined Sections meeting.
The question is, he said, "Do you have to rush them back in [and re-excise] or can you sit on them for a while?"
"Dr. Douglas addresses an interesting question," said session moderator Dr. Jeffrey H. Spiegel, chief of the division of facial plastic and reconstructive surgery at Boston University Medical Center and otolaryngology faculty member at Boston University. "I see a lot of people who had squamous cells and basal cells excised sent back for re-excision ... and he’ll help us understand if I even need to be doing that."
Dr. Douglas and his colleagues studied 492 patients at the Clarksburg Veterans Affairs Hospital in Morgantown, W.Va. All had skin cancers removed from their head or neck, including 387 basal cell carcinomas and 105 squamous cell carcinomas, over 5 years.
"Do you have to rush them back in [and re-excise] or can you sit on them for a while?"
About 40% or 197 lesions had a negative margin after wide local excision. The remaining 295 lesions featured a positive margin on permanent section analysis. The positive lesions included 232 basal cell carcinomas (BCCs) and 63 squamous cell carcinomas (SCCs).
"In the group of 232 positive basal cell lesions, everybody was offered re-excision. A total 105 chose re-excision and 127 decided to just watch it, despite our best judgment," said Dr. Douglas, an otolaryngologist at the Morgantown V.A.
"Even of the squamous cell patients, 26 still said they wanted to watch it," Dr. Douglas said at the meeting, which was cosponsored by the Triological Society and the American College of Surgeons.
Of the positive margin group, none of the BCC and four (3.8%) of the SCC lesions recurred within a median follow-up of 3.7 years. This finding suggests a role for clinical observation of low-risk, nonmelanomatous cancers, Dr. Douglas said.
The four SCC lesions that recurred included one lesion in the observation-only group and three in the re-excision group. Some of these patients likely had a more aggressive form of cancer, Dr. Douglas said.
"Essentially, we feel what we learned from this study is that ... given the fact that you can handle any recurrence of these lesions if they occur, you can watch the basal cells, even with positive margins," Dr. Douglas said. "We still feel all squamous cell lesions should be taken out until you get a clear margin."
Why the majority of positive margin patients who chose observation did not experience recurrence is poorly understood, Dr. Douglas said. However, it could be the patient’s immune system, secondary to the wound healing process, comes in and cleans up any residual cells. Another possibility is that patients die from other causes before recurrence can occur. "The truth of the matter is: We don’t know. We need more studies to figure this out."
The nose was the most common site for the T1, nonmelanoma lesions. The average size was 1.2 cm. Mean patient age was 61 years and 484 (98%) were men. Patients were excluded from the study if their lesion was previously treated or they chose an option other than re-excision or observation.
Although initial excision by a Mohs surgeon would be ideal, Dr. Douglas said, in his study dermatologists and otolaryngologists removed the lesions at one of the hospital ambulatory clinics. This strategy reflects some real-world limitations. "The problem is, at least in West Virginia, Mohs surgeons are very sparse. There are probably only a handful in the state." Operating room time and scheduling (particularly for patients with many lesions), availability of a pathologist to read frozen sections, and costs are additional challenges.
Dr. Douglas and Dr. Spiegel said that they had no relevant financial disclosures.
MIAMI BEACH – Observation or a "watchful waiting" approach is an acceptable option vs. re-excision for patients with positive margins remaining after removal of head and neck skin cancers, according to a retrospective study.
"A lot of times we take these lesions off and they appear to be benign, not really knowing what the preoperative histologic diagnosis is. [Then we] find out the diagnosis is cancer, and not only that, but they have a positive margin." Dr. Justin Douglas said at the Triological Society Combined Sections meeting.
The question is, he said, "Do you have to rush them back in [and re-excise] or can you sit on them for a while?"
"Dr. Douglas addresses an interesting question," said session moderator Dr. Jeffrey H. Spiegel, chief of the division of facial plastic and reconstructive surgery at Boston University Medical Center and otolaryngology faculty member at Boston University. "I see a lot of people who had squamous cells and basal cells excised sent back for re-excision ... and he’ll help us understand if I even need to be doing that."
Dr. Douglas and his colleagues studied 492 patients at the Clarksburg Veterans Affairs Hospital in Morgantown, W.Va. All had skin cancers removed from their head or neck, including 387 basal cell carcinomas and 105 squamous cell carcinomas, over 5 years.
"Do you have to rush them back in [and re-excise] or can you sit on them for a while?"
About 40% or 197 lesions had a negative margin after wide local excision. The remaining 295 lesions featured a positive margin on permanent section analysis. The positive lesions included 232 basal cell carcinomas (BCCs) and 63 squamous cell carcinomas (SCCs).
"In the group of 232 positive basal cell lesions, everybody was offered re-excision. A total 105 chose re-excision and 127 decided to just watch it, despite our best judgment," said Dr. Douglas, an otolaryngologist at the Morgantown V.A.
"Even of the squamous cell patients, 26 still said they wanted to watch it," Dr. Douglas said at the meeting, which was cosponsored by the Triological Society and the American College of Surgeons.
Of the positive margin group, none of the BCC and four (3.8%) of the SCC lesions recurred within a median follow-up of 3.7 years. This finding suggests a role for clinical observation of low-risk, nonmelanomatous cancers, Dr. Douglas said.
The four SCC lesions that recurred included one lesion in the observation-only group and three in the re-excision group. Some of these patients likely had a more aggressive form of cancer, Dr. Douglas said.
"Essentially, we feel what we learned from this study is that ... given the fact that you can handle any recurrence of these lesions if they occur, you can watch the basal cells, even with positive margins," Dr. Douglas said. "We still feel all squamous cell lesions should be taken out until you get a clear margin."
Why the majority of positive margin patients who chose observation did not experience recurrence is poorly understood, Dr. Douglas said. However, it could be the patient’s immune system, secondary to the wound healing process, comes in and cleans up any residual cells. Another possibility is that patients die from other causes before recurrence can occur. "The truth of the matter is: We don’t know. We need more studies to figure this out."
The nose was the most common site for the T1, nonmelanoma lesions. The average size was 1.2 cm. Mean patient age was 61 years and 484 (98%) were men. Patients were excluded from the study if their lesion was previously treated or they chose an option other than re-excision or observation.
Although initial excision by a Mohs surgeon would be ideal, Dr. Douglas said, in his study dermatologists and otolaryngologists removed the lesions at one of the hospital ambulatory clinics. This strategy reflects some real-world limitations. "The problem is, at least in West Virginia, Mohs surgeons are very sparse. There are probably only a handful in the state." Operating room time and scheduling (particularly for patients with many lesions), availability of a pathologist to read frozen sections, and costs are additional challenges.
Dr. Douglas and Dr. Spiegel said that they had no relevant financial disclosures.
FROM THE TRIOLOGICAL SOCIETY COMBINED SECTIONS MEETING
Major Finding: None of 232 basal cell carcinoma lesions and 4 (3.8%) of 63 positive squamous cell carcinoma lesions with an initial positive margin recurred over a median follow-up of 3.7 years.
Data Source: Retrospective, single-center study of 492 patients diagnosed with head and neck skin cancers over 5 years.
Disclosures: Dr. Douglas reported having no financial disclosures.