User login
VTE, sepsis risk increased among COVID-19 patients with cancer
, according to data from a registry study.
Researchers analyzed data on 5,556 patients with COVID-19 who had an inpatient or emergency encounter at Mount Sinai Health System (MSHS) in New York between March 1 and May 27, 2020. Patients were included in an anonymous MSHS COVID-19 registry.
There were 421 patients who had cancer: 96 with a hematologic malignancy and 325 with solid tumors.
After adjustment for age, gender, and number of comorbidities, the odds ratios for acute VTE and sepsis for patients with cancer (versus those without cancer) were 1.77 and 1.34, respectively. The adjusted odds ratio for mortality in cancer patients was 1.02.
The results remained “relatively consistent” after stratification by solid and nonsolid cancer types, with no significant difference in outcomes between those two groups, and results remained consistent in a propensity-matched model, according to Naomi Alpert, a biostatistician at Icahn School of Medicine at Mount Sinai, New York.
Ms. Alpert reported these findings at the AACR virtual meeting: COVID-19 and Cancer.
She noted that the cancer patients were older than the noncancer patients (mean age, 69.2 years vs. 63.8 years), and cancer patients were more likely to have two or more comorbid conditions (48.2% vs. 30.4%). Cancer patients also had significantly lower hemoglobin levels and red blood cell, platelet, and white blood cell counts (P < .01 for all).
“Low white blood cell count may be one of the reasons for higher risk of sepsis in cancer patients, as it may lead to a higher risk of infection,” Ms. Alpert said. “However, it’s not clear what role cancer therapies play in the risks of COVID-19 morbidity and mortality, so there is still quite a bit to learn.”
In fact, the findings are limited by a lack of information about cancer treatment, as the registry was not designed for that purpose, she noted.
Another study limitation is the short follow-up of a month or less in most patients, due, in part, to the novelty of COVID-19, but also to the lack of information on patients after they left the hospital.
“However, we had a very large sample size, with more than 400 cancer patients included, and, to our knowledge, this is the largest analysis of its kind to be done so far,” Ms. Alpert said. “In the future, it’s going to be very important to assess the effect of cancer therapies on COVID-19 complications and to see if prior therapies had any effect on outcomes.”
Longer follow-up would also be helpful for assessing the chronic effects of COVID-19 on cancer patients over time, she said. “It would be important to see whether some of these elevated risks of venous thromboembolism and sepsis are associated with longer-term mortality risks than what we were able to measure here,” she added.
Asked about the discrepancy between mortality in this study and those of larger registries, such as the COVID-19 and Cancer Consortium (CCC19) and TERAVOLT, Ms. Alpert noted that the current study included only patients who required hospitalization or emergency care.
“Our mortality rate was actually a bit higher than what was reported in some of the other studies,” she said. “We had about a 30% mortality rate in the cancer patients and about 25% for the noncancer patients, so ... we’re sort of looking at a subset of patients who we know are the sickest of the sick, which may explain some of the higher mortality that we’re seeing.”
Ms. Alpert reported having no disclosures.
SOURCE: Alpert N et al. AACR COVID-19 and Cancer, Abstract S12-02.
, according to data from a registry study.
Researchers analyzed data on 5,556 patients with COVID-19 who had an inpatient or emergency encounter at Mount Sinai Health System (MSHS) in New York between March 1 and May 27, 2020. Patients were included in an anonymous MSHS COVID-19 registry.
There were 421 patients who had cancer: 96 with a hematologic malignancy and 325 with solid tumors.
After adjustment for age, gender, and number of comorbidities, the odds ratios for acute VTE and sepsis for patients with cancer (versus those without cancer) were 1.77 and 1.34, respectively. The adjusted odds ratio for mortality in cancer patients was 1.02.
The results remained “relatively consistent” after stratification by solid and nonsolid cancer types, with no significant difference in outcomes between those two groups, and results remained consistent in a propensity-matched model, according to Naomi Alpert, a biostatistician at Icahn School of Medicine at Mount Sinai, New York.
Ms. Alpert reported these findings at the AACR virtual meeting: COVID-19 and Cancer.
She noted that the cancer patients were older than the noncancer patients (mean age, 69.2 years vs. 63.8 years), and cancer patients were more likely to have two or more comorbid conditions (48.2% vs. 30.4%). Cancer patients also had significantly lower hemoglobin levels and red blood cell, platelet, and white blood cell counts (P < .01 for all).
“Low white blood cell count may be one of the reasons for higher risk of sepsis in cancer patients, as it may lead to a higher risk of infection,” Ms. Alpert said. “However, it’s not clear what role cancer therapies play in the risks of COVID-19 morbidity and mortality, so there is still quite a bit to learn.”
In fact, the findings are limited by a lack of information about cancer treatment, as the registry was not designed for that purpose, she noted.
Another study limitation is the short follow-up of a month or less in most patients, due, in part, to the novelty of COVID-19, but also to the lack of information on patients after they left the hospital.
“However, we had a very large sample size, with more than 400 cancer patients included, and, to our knowledge, this is the largest analysis of its kind to be done so far,” Ms. Alpert said. “In the future, it’s going to be very important to assess the effect of cancer therapies on COVID-19 complications and to see if prior therapies had any effect on outcomes.”
Longer follow-up would also be helpful for assessing the chronic effects of COVID-19 on cancer patients over time, she said. “It would be important to see whether some of these elevated risks of venous thromboembolism and sepsis are associated with longer-term mortality risks than what we were able to measure here,” she added.
Asked about the discrepancy between mortality in this study and those of larger registries, such as the COVID-19 and Cancer Consortium (CCC19) and TERAVOLT, Ms. Alpert noted that the current study included only patients who required hospitalization or emergency care.
“Our mortality rate was actually a bit higher than what was reported in some of the other studies,” she said. “We had about a 30% mortality rate in the cancer patients and about 25% for the noncancer patients, so ... we’re sort of looking at a subset of patients who we know are the sickest of the sick, which may explain some of the higher mortality that we’re seeing.”
Ms. Alpert reported having no disclosures.
SOURCE: Alpert N et al. AACR COVID-19 and Cancer, Abstract S12-02.
, according to data from a registry study.
Researchers analyzed data on 5,556 patients with COVID-19 who had an inpatient or emergency encounter at Mount Sinai Health System (MSHS) in New York between March 1 and May 27, 2020. Patients were included in an anonymous MSHS COVID-19 registry.
There were 421 patients who had cancer: 96 with a hematologic malignancy and 325 with solid tumors.
After adjustment for age, gender, and number of comorbidities, the odds ratios for acute VTE and sepsis for patients with cancer (versus those without cancer) were 1.77 and 1.34, respectively. The adjusted odds ratio for mortality in cancer patients was 1.02.
The results remained “relatively consistent” after stratification by solid and nonsolid cancer types, with no significant difference in outcomes between those two groups, and results remained consistent in a propensity-matched model, according to Naomi Alpert, a biostatistician at Icahn School of Medicine at Mount Sinai, New York.
Ms. Alpert reported these findings at the AACR virtual meeting: COVID-19 and Cancer.
She noted that the cancer patients were older than the noncancer patients (mean age, 69.2 years vs. 63.8 years), and cancer patients were more likely to have two or more comorbid conditions (48.2% vs. 30.4%). Cancer patients also had significantly lower hemoglobin levels and red blood cell, platelet, and white blood cell counts (P < .01 for all).
“Low white blood cell count may be one of the reasons for higher risk of sepsis in cancer patients, as it may lead to a higher risk of infection,” Ms. Alpert said. “However, it’s not clear what role cancer therapies play in the risks of COVID-19 morbidity and mortality, so there is still quite a bit to learn.”
In fact, the findings are limited by a lack of information about cancer treatment, as the registry was not designed for that purpose, she noted.
Another study limitation is the short follow-up of a month or less in most patients, due, in part, to the novelty of COVID-19, but also to the lack of information on patients after they left the hospital.
“However, we had a very large sample size, with more than 400 cancer patients included, and, to our knowledge, this is the largest analysis of its kind to be done so far,” Ms. Alpert said. “In the future, it’s going to be very important to assess the effect of cancer therapies on COVID-19 complications and to see if prior therapies had any effect on outcomes.”
Longer follow-up would also be helpful for assessing the chronic effects of COVID-19 on cancer patients over time, she said. “It would be important to see whether some of these elevated risks of venous thromboembolism and sepsis are associated with longer-term mortality risks than what we were able to measure here,” she added.
Asked about the discrepancy between mortality in this study and those of larger registries, such as the COVID-19 and Cancer Consortium (CCC19) and TERAVOLT, Ms. Alpert noted that the current study included only patients who required hospitalization or emergency care.
“Our mortality rate was actually a bit higher than what was reported in some of the other studies,” she said. “We had about a 30% mortality rate in the cancer patients and about 25% for the noncancer patients, so ... we’re sort of looking at a subset of patients who we know are the sickest of the sick, which may explain some of the higher mortality that we’re seeing.”
Ms. Alpert reported having no disclosures.
SOURCE: Alpert N et al. AACR COVID-19 and Cancer, Abstract S12-02.
FROM AACR: COVID-19 AND CANCER
First guideline on NGS testing in cancer, from ESMO
Recommendations on the use of next-generation sequencing (NGS) tests for patients with metastatic cancer have been issued by the European Society for Medical Oncology, the first recommendations of their kind to be published by any medical society.
“Until now, there were no recommendations from scientific societies on how to use this technique in daily clinical practice to profile metastatic cancers,” Fernanda Mosele, MD, medical oncologist, Gustave Roussy, Villejuif, France, said in a statement.
NGS testing is already used extensively in oncology, particularly in metastatic cancer, she noted. The technology is used to assess the sequence of DNA in genes from a tumor tissue sample. Numerous genes can be quickly sequenced at the same time at relatively low cost. The results provide information on mutations that are present, which, in turn, helps with deciding which treatments to use, including drugs targeting the identified mutations.
“Our intent is that they [the guidelines] will unify decision-making about how NGS should be used for patients with metastatic cancer,” Dr. Mosele said.
The recommendations were published online August 25 in Annals of Oncology.
Overall, ESMO recommends the use of tumor multigene NGS for non–small cell lung cancer (NSCLC), prostate cancer, ovarian cancer, and cholangiocarcinoma.
For other cancers, the authors said that NGS is not recommended in clinical practice but could be used for research purposes.
However, patients should be informed that it is unlikely that test results would benefit them much personally.
Physicians and patients may decide together to subject the tumor to mutational testing using a large panel of genes, provided testing doesn’t burden the health care system with additional costs.
“This recommendation acknowledges that a small number of patients could benefit from a drug because they have a rare mutation,” Joaquin Mateo, MD, chair of the ESMO working group, said in a statement.
“So beyond the cancers in which everyone should receive NGS, there is room for physicians and patients to discuss the pros and cons of ordering these tests,” he added.
ESMO also does not recommend the use of off-label drugs matched to any genomic alteration detected by NGS unless an access program and a decisional procedure have been developed, either regionally or nationally.
No need for NGS testing of other cancers
In contrast to NSCLC, “there is currently no need to perform tumor multigene NGS for patients with mBC [metastatic breast cancer] in the context of daily practice,” ESMO stated.
This is largely because somatic sequencing cannot fully substitute for germline testing for BRCA status, and other mutations, such as HER2, can be detected using immunohistochemistry (IHC).
The same can be said for patients with metastatic gastric cancer, inasmuch as detection of alterations can and should be done using cheaper testing methods, ESMO pointed out.
However, ESMO members still emphasized that it’s important to include patients with metastatic breast cancer in molecular screening programs as well as in clinical trials testing targeted agents.
Similarly, there is no need to test metastatic colorectal cancer (mCRC) using multigene NGS in daily practice, inasmuch as most level 1 alterations in mCRC can be determined by IHC or PCR.
However, NGS can be considered as an alternative to PCR-based tests in mCRC, provided NGS is not associated with additional cost.
ESMO again recommended that research centers include mCRC patients in molecular screening programs in order for them to have access to innovative clinical trial agents.
As for advanced prostate cancer, ESMO does recommend that clinicians perform NGS on tissue samples to assess the tumor’s mutational status, at least for the presence of BRCA1 and BRCA2 mutations, when patients have access to the poly (ADP-ribose) polymerase inhibitors for treatment.
The authors cautioned, however, that this strategy is unlikely to be cost-effective, so larger panels should be used only when there are specific agreements with payers.
Multigene NGS is also not recommended for patients with advanced pancreatic ductal adenocarcinoma (PDAC), although ESMO points out that it is the role of research centers to propose multigene sequencing for these patients in the context of molecular screening programs.
This is again to facilitate access to innovative drugs for these patients.
Similar to recommendations for patients with advanced PDAC, patients with advanced hepatocellular carcinoma (HCC) do not need to have tumor multigene NGS either.
Considering the high unmet needs of HCC patients, ESMO feels that research centers should propose multigene sequencing to patients with advanced HCC in the context of molecular screening programs.
In contrast, ESMO recommended that tumor multigene NGS be used to detect actionable alterations in patients with advanced cholangiocarcinoma.
Again, they predict that this strategy is unlikely to be cost-effective, so larger panels should only be used if a specific agreement is in place with payers.
ESMO also assessed the frequency of level 1 alterations in less frequent tumor types, including ovarian cancers. Because BRCA1 and BRCA2 somatic mutations in ovarian tumors have been associated with increased response to the PARP inhibitors, the use of multigene NGS is justified with this malignancy, ESMO states.
The authors also recommend that tumor mutational burden be determined in cervical cancer, moderately differentiated neuroendocrine tumors, salivary cancers, vulvar cancer, and thyroid cancers.
Dr. Mosele has disclosed no relevant financial relationships. Many coauthors have relationships with the pharmaceutical industry, as listed in the article.
This article first appeared on Medscape.com.
Recommendations on the use of next-generation sequencing (NGS) tests for patients with metastatic cancer have been issued by the European Society for Medical Oncology, the first recommendations of their kind to be published by any medical society.
“Until now, there were no recommendations from scientific societies on how to use this technique in daily clinical practice to profile metastatic cancers,” Fernanda Mosele, MD, medical oncologist, Gustave Roussy, Villejuif, France, said in a statement.
NGS testing is already used extensively in oncology, particularly in metastatic cancer, she noted. The technology is used to assess the sequence of DNA in genes from a tumor tissue sample. Numerous genes can be quickly sequenced at the same time at relatively low cost. The results provide information on mutations that are present, which, in turn, helps with deciding which treatments to use, including drugs targeting the identified mutations.
“Our intent is that they [the guidelines] will unify decision-making about how NGS should be used for patients with metastatic cancer,” Dr. Mosele said.
The recommendations were published online August 25 in Annals of Oncology.
Overall, ESMO recommends the use of tumor multigene NGS for non–small cell lung cancer (NSCLC), prostate cancer, ovarian cancer, and cholangiocarcinoma.
For other cancers, the authors said that NGS is not recommended in clinical practice but could be used for research purposes.
However, patients should be informed that it is unlikely that test results would benefit them much personally.
Physicians and patients may decide together to subject the tumor to mutational testing using a large panel of genes, provided testing doesn’t burden the health care system with additional costs.
“This recommendation acknowledges that a small number of patients could benefit from a drug because they have a rare mutation,” Joaquin Mateo, MD, chair of the ESMO working group, said in a statement.
“So beyond the cancers in which everyone should receive NGS, there is room for physicians and patients to discuss the pros and cons of ordering these tests,” he added.
ESMO also does not recommend the use of off-label drugs matched to any genomic alteration detected by NGS unless an access program and a decisional procedure have been developed, either regionally or nationally.
No need for NGS testing of other cancers
In contrast to NSCLC, “there is currently no need to perform tumor multigene NGS for patients with mBC [metastatic breast cancer] in the context of daily practice,” ESMO stated.
This is largely because somatic sequencing cannot fully substitute for germline testing for BRCA status, and other mutations, such as HER2, can be detected using immunohistochemistry (IHC).
The same can be said for patients with metastatic gastric cancer, inasmuch as detection of alterations can and should be done using cheaper testing methods, ESMO pointed out.
However, ESMO members still emphasized that it’s important to include patients with metastatic breast cancer in molecular screening programs as well as in clinical trials testing targeted agents.
Similarly, there is no need to test metastatic colorectal cancer (mCRC) using multigene NGS in daily practice, inasmuch as most level 1 alterations in mCRC can be determined by IHC or PCR.
However, NGS can be considered as an alternative to PCR-based tests in mCRC, provided NGS is not associated with additional cost.
ESMO again recommended that research centers include mCRC patients in molecular screening programs in order for them to have access to innovative clinical trial agents.
As for advanced prostate cancer, ESMO does recommend that clinicians perform NGS on tissue samples to assess the tumor’s mutational status, at least for the presence of BRCA1 and BRCA2 mutations, when patients have access to the poly (ADP-ribose) polymerase inhibitors for treatment.
The authors cautioned, however, that this strategy is unlikely to be cost-effective, so larger panels should be used only when there are specific agreements with payers.
Multigene NGS is also not recommended for patients with advanced pancreatic ductal adenocarcinoma (PDAC), although ESMO points out that it is the role of research centers to propose multigene sequencing for these patients in the context of molecular screening programs.
This is again to facilitate access to innovative drugs for these patients.
Similar to recommendations for patients with advanced PDAC, patients with advanced hepatocellular carcinoma (HCC) do not need to have tumor multigene NGS either.
Considering the high unmet needs of HCC patients, ESMO feels that research centers should propose multigene sequencing to patients with advanced HCC in the context of molecular screening programs.
In contrast, ESMO recommended that tumor multigene NGS be used to detect actionable alterations in patients with advanced cholangiocarcinoma.
Again, they predict that this strategy is unlikely to be cost-effective, so larger panels should only be used if a specific agreement is in place with payers.
ESMO also assessed the frequency of level 1 alterations in less frequent tumor types, including ovarian cancers. Because BRCA1 and BRCA2 somatic mutations in ovarian tumors have been associated with increased response to the PARP inhibitors, the use of multigene NGS is justified with this malignancy, ESMO states.
The authors also recommend that tumor mutational burden be determined in cervical cancer, moderately differentiated neuroendocrine tumors, salivary cancers, vulvar cancer, and thyroid cancers.
Dr. Mosele has disclosed no relevant financial relationships. Many coauthors have relationships with the pharmaceutical industry, as listed in the article.
This article first appeared on Medscape.com.
Recommendations on the use of next-generation sequencing (NGS) tests for patients with metastatic cancer have been issued by the European Society for Medical Oncology, the first recommendations of their kind to be published by any medical society.
“Until now, there were no recommendations from scientific societies on how to use this technique in daily clinical practice to profile metastatic cancers,” Fernanda Mosele, MD, medical oncologist, Gustave Roussy, Villejuif, France, said in a statement.
NGS testing is already used extensively in oncology, particularly in metastatic cancer, she noted. The technology is used to assess the sequence of DNA in genes from a tumor tissue sample. Numerous genes can be quickly sequenced at the same time at relatively low cost. The results provide information on mutations that are present, which, in turn, helps with deciding which treatments to use, including drugs targeting the identified mutations.
“Our intent is that they [the guidelines] will unify decision-making about how NGS should be used for patients with metastatic cancer,” Dr. Mosele said.
The recommendations were published online August 25 in Annals of Oncology.
Overall, ESMO recommends the use of tumor multigene NGS for non–small cell lung cancer (NSCLC), prostate cancer, ovarian cancer, and cholangiocarcinoma.
For other cancers, the authors said that NGS is not recommended in clinical practice but could be used for research purposes.
However, patients should be informed that it is unlikely that test results would benefit them much personally.
Physicians and patients may decide together to subject the tumor to mutational testing using a large panel of genes, provided testing doesn’t burden the health care system with additional costs.
“This recommendation acknowledges that a small number of patients could benefit from a drug because they have a rare mutation,” Joaquin Mateo, MD, chair of the ESMO working group, said in a statement.
“So beyond the cancers in which everyone should receive NGS, there is room for physicians and patients to discuss the pros and cons of ordering these tests,” he added.
ESMO also does not recommend the use of off-label drugs matched to any genomic alteration detected by NGS unless an access program and a decisional procedure have been developed, either regionally or nationally.
No need for NGS testing of other cancers
In contrast to NSCLC, “there is currently no need to perform tumor multigene NGS for patients with mBC [metastatic breast cancer] in the context of daily practice,” ESMO stated.
This is largely because somatic sequencing cannot fully substitute for germline testing for BRCA status, and other mutations, such as HER2, can be detected using immunohistochemistry (IHC).
The same can be said for patients with metastatic gastric cancer, inasmuch as detection of alterations can and should be done using cheaper testing methods, ESMO pointed out.
However, ESMO members still emphasized that it’s important to include patients with metastatic breast cancer in molecular screening programs as well as in clinical trials testing targeted agents.
Similarly, there is no need to test metastatic colorectal cancer (mCRC) using multigene NGS in daily practice, inasmuch as most level 1 alterations in mCRC can be determined by IHC or PCR.
However, NGS can be considered as an alternative to PCR-based tests in mCRC, provided NGS is not associated with additional cost.
ESMO again recommended that research centers include mCRC patients in molecular screening programs in order for them to have access to innovative clinical trial agents.
As for advanced prostate cancer, ESMO does recommend that clinicians perform NGS on tissue samples to assess the tumor’s mutational status, at least for the presence of BRCA1 and BRCA2 mutations, when patients have access to the poly (ADP-ribose) polymerase inhibitors for treatment.
The authors cautioned, however, that this strategy is unlikely to be cost-effective, so larger panels should be used only when there are specific agreements with payers.
Multigene NGS is also not recommended for patients with advanced pancreatic ductal adenocarcinoma (PDAC), although ESMO points out that it is the role of research centers to propose multigene sequencing for these patients in the context of molecular screening programs.
This is again to facilitate access to innovative drugs for these patients.
Similar to recommendations for patients with advanced PDAC, patients with advanced hepatocellular carcinoma (HCC) do not need to have tumor multigene NGS either.
Considering the high unmet needs of HCC patients, ESMO feels that research centers should propose multigene sequencing to patients with advanced HCC in the context of molecular screening programs.
In contrast, ESMO recommended that tumor multigene NGS be used to detect actionable alterations in patients with advanced cholangiocarcinoma.
Again, they predict that this strategy is unlikely to be cost-effective, so larger panels should only be used if a specific agreement is in place with payers.
ESMO also assessed the frequency of level 1 alterations in less frequent tumor types, including ovarian cancers. Because BRCA1 and BRCA2 somatic mutations in ovarian tumors have been associated with increased response to the PARP inhibitors, the use of multigene NGS is justified with this malignancy, ESMO states.
The authors also recommend that tumor mutational burden be determined in cervical cancer, moderately differentiated neuroendocrine tumors, salivary cancers, vulvar cancer, and thyroid cancers.
Dr. Mosele has disclosed no relevant financial relationships. Many coauthors have relationships with the pharmaceutical industry, as listed in the article.
This article first appeared on Medscape.com.
Immunotherapy should not be withheld because of sex, age, or PS
The improvement in survival in many cancer types that is seen with immune checkpoint inhibitors (ICIs), when compared to control therapies, is not affected by the patient’s sex, age, or Eastern Cooperative Oncology Group (ECOG) performance status (PS), according to a new meta-analysis.
Therefore, treatment with these immunotherapies should not be withheld on the basis of these factors, the authors concluded.
Asked whether there have been such instances of withholding ICIs, lead author Yucai Wang, MD, PhD, Mayo Clinic, Rochester, Minnesota, told Medscape Medical News: “We did this study solely based on scientific questions we had and not because we were seeing any bias at the moment in the use of ICIs.
“And we saw that the survival benefits were very similar across all of the categories [we analyzed], with a survival benefit of about 20% from immunotherapy across the board, which is clinically meaningful,” he added.
The study was published online August 7 in JAMA Network Open.
“The comparable survival advantage between patients of different sex, age, and ECOG PS may encourage more patients to receive ICI treatment regardless of cancer types, lines of therapy, agents of immunotherapy, and intervention therapies,” the authors commented.
Wang noted that there have been conflicting reports in the literature suggesting that male patients may benefit more from immunotherapy than female patients and that older patients may benefit more from the same treatment than younger patients.
However, there are also suggestions in the literature that women experience a stronger immune response than men and that, with aging, the immune system generally undergoes immunosenescence.
In addition, the PS of oncology patients has been implicated in how well patients respond to immunotherapy.
Wang noted that the findings of past studies have contradicted each other.
Findings of the Meta-Analysis
The meta-analysis included 37 randomized clinical trials that involved a total of 23,760 patients with a variety of advanced cancers. “Most of the trials were phase 3 (n = 34) and conduced for subsequent lines of therapy (n = 22),” the authors explained.
The most common cancers treated with an ICI were non–small cell lung cancer and melanoma.
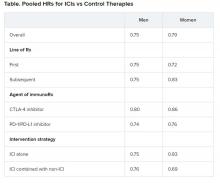
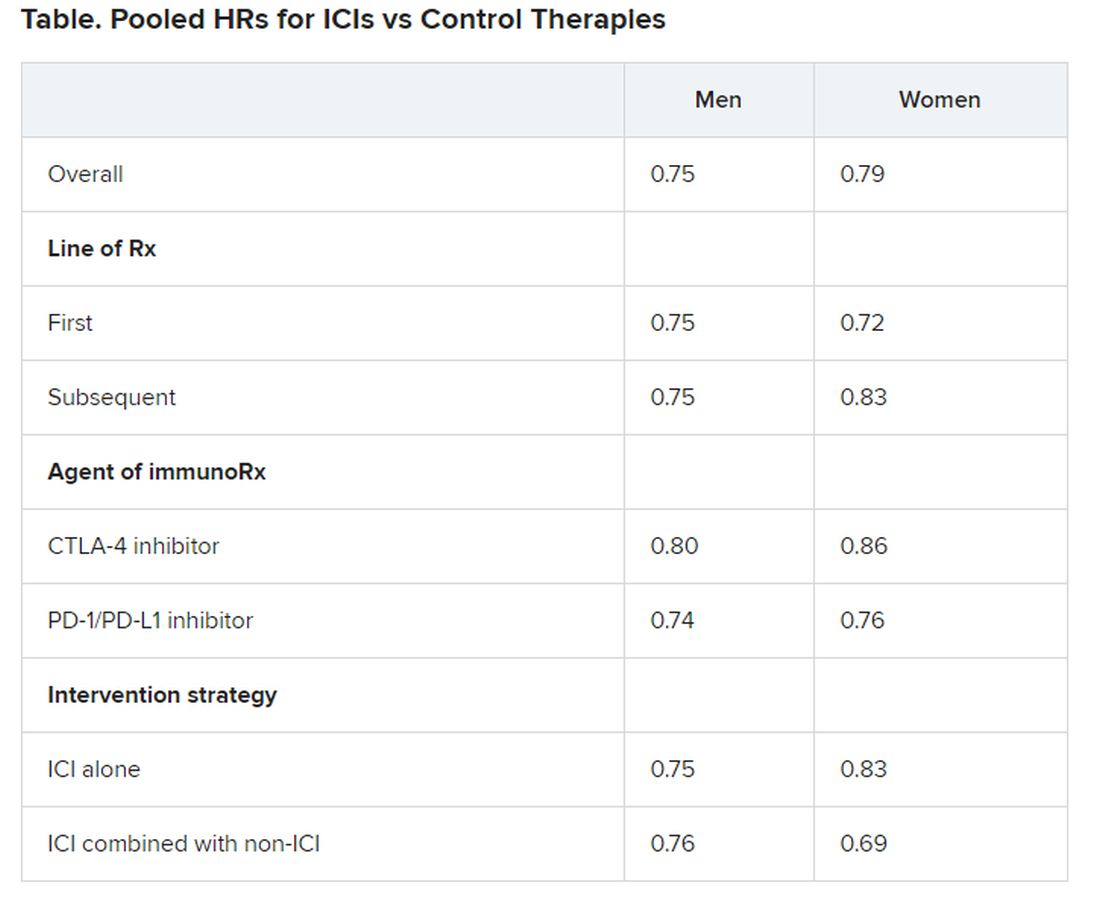
Pooled overall survival (OS) hazard ratios (HRs) were calculated on the basis of sex, age (younger than 65 years and 65 years and older), and an ECOG PS of 0 and 1 or higher.
Responses were stratified on the basis of cancer type, line of therapy, the ICI used, and the immunotherapy strategy used in the ICI arm.
Most of the drugs evaluated were PD-1 and PD-L1 inhibitors. The specific drugs assessed included ipilimumab, tremelimumab, nivolumab, pembrolizumab, atezolizumab, durvalumab, and avelumab.
A total of 32 trials that involved more than 20,000 patients reported HRs for death according to the patients’ sex. Thirty-four trials that involved more than 21,000 patients reported HRs for death according to patients’ age, and 30 trials that involved more than 19,000 patients reported HRs for death according to patients’ ECOG PS.
No significant differences in OS benefit were seen by cancer type, line of therapy, agent of immunotherapy, or intervention strategy, the investigators pointed out.
There were also no differences in survival benefit associated with immunotherapy vs control therapies for patients with an ECOG PS of 0 and an ECOG PS of 1 or greater. The OS benefit was 0.81 for those with an ECOG PS of 0 and 0.79 for those with an ECOG PS of 1 or greater.
Wang has disclosed no relevant financial relationships.
This article first appeared on Medscape.com .
The improvement in survival in many cancer types that is seen with immune checkpoint inhibitors (ICIs), when compared to control therapies, is not affected by the patient’s sex, age, or Eastern Cooperative Oncology Group (ECOG) performance status (PS), according to a new meta-analysis.
Therefore, treatment with these immunotherapies should not be withheld on the basis of these factors, the authors concluded.
Asked whether there have been such instances of withholding ICIs, lead author Yucai Wang, MD, PhD, Mayo Clinic, Rochester, Minnesota, told Medscape Medical News: “We did this study solely based on scientific questions we had and not because we were seeing any bias at the moment in the use of ICIs.
“And we saw that the survival benefits were very similar across all of the categories [we analyzed], with a survival benefit of about 20% from immunotherapy across the board, which is clinically meaningful,” he added.
The study was published online August 7 in JAMA Network Open.
“The comparable survival advantage between patients of different sex, age, and ECOG PS may encourage more patients to receive ICI treatment regardless of cancer types, lines of therapy, agents of immunotherapy, and intervention therapies,” the authors commented.
Wang noted that there have been conflicting reports in the literature suggesting that male patients may benefit more from immunotherapy than female patients and that older patients may benefit more from the same treatment than younger patients.
However, there are also suggestions in the literature that women experience a stronger immune response than men and that, with aging, the immune system generally undergoes immunosenescence.
In addition, the PS of oncology patients has been implicated in how well patients respond to immunotherapy.
Wang noted that the findings of past studies have contradicted each other.
Findings of the Meta-Analysis
The meta-analysis included 37 randomized clinical trials that involved a total of 23,760 patients with a variety of advanced cancers. “Most of the trials were phase 3 (n = 34) and conduced for subsequent lines of therapy (n = 22),” the authors explained.
The most common cancers treated with an ICI were non–small cell lung cancer and melanoma.
Pooled overall survival (OS) hazard ratios (HRs) were calculated on the basis of sex, age (younger than 65 years and 65 years and older), and an ECOG PS of 0 and 1 or higher.
Responses were stratified on the basis of cancer type, line of therapy, the ICI used, and the immunotherapy strategy used in the ICI arm.
Most of the drugs evaluated were PD-1 and PD-L1 inhibitors. The specific drugs assessed included ipilimumab, tremelimumab, nivolumab, pembrolizumab, atezolizumab, durvalumab, and avelumab.
A total of 32 trials that involved more than 20,000 patients reported HRs for death according to the patients’ sex. Thirty-four trials that involved more than 21,000 patients reported HRs for death according to patients’ age, and 30 trials that involved more than 19,000 patients reported HRs for death according to patients’ ECOG PS.
No significant differences in OS benefit were seen by cancer type, line of therapy, agent of immunotherapy, or intervention strategy, the investigators pointed out.
There were also no differences in survival benefit associated with immunotherapy vs control therapies for patients with an ECOG PS of 0 and an ECOG PS of 1 or greater. The OS benefit was 0.81 for those with an ECOG PS of 0 and 0.79 for those with an ECOG PS of 1 or greater.
Wang has disclosed no relevant financial relationships.
This article first appeared on Medscape.com .
The improvement in survival in many cancer types that is seen with immune checkpoint inhibitors (ICIs), when compared to control therapies, is not affected by the patient’s sex, age, or Eastern Cooperative Oncology Group (ECOG) performance status (PS), according to a new meta-analysis.
Therefore, treatment with these immunotherapies should not be withheld on the basis of these factors, the authors concluded.
Asked whether there have been such instances of withholding ICIs, lead author Yucai Wang, MD, PhD, Mayo Clinic, Rochester, Minnesota, told Medscape Medical News: “We did this study solely based on scientific questions we had and not because we were seeing any bias at the moment in the use of ICIs.
“And we saw that the survival benefits were very similar across all of the categories [we analyzed], with a survival benefit of about 20% from immunotherapy across the board, which is clinically meaningful,” he added.
The study was published online August 7 in JAMA Network Open.
“The comparable survival advantage between patients of different sex, age, and ECOG PS may encourage more patients to receive ICI treatment regardless of cancer types, lines of therapy, agents of immunotherapy, and intervention therapies,” the authors commented.
Wang noted that there have been conflicting reports in the literature suggesting that male patients may benefit more from immunotherapy than female patients and that older patients may benefit more from the same treatment than younger patients.
However, there are also suggestions in the literature that women experience a stronger immune response than men and that, with aging, the immune system generally undergoes immunosenescence.
In addition, the PS of oncology patients has been implicated in how well patients respond to immunotherapy.
Wang noted that the findings of past studies have contradicted each other.
Findings of the Meta-Analysis
The meta-analysis included 37 randomized clinical trials that involved a total of 23,760 patients with a variety of advanced cancers. “Most of the trials were phase 3 (n = 34) and conduced for subsequent lines of therapy (n = 22),” the authors explained.
The most common cancers treated with an ICI were non–small cell lung cancer and melanoma.
Pooled overall survival (OS) hazard ratios (HRs) were calculated on the basis of sex, age (younger than 65 years and 65 years and older), and an ECOG PS of 0 and 1 or higher.
Responses were stratified on the basis of cancer type, line of therapy, the ICI used, and the immunotherapy strategy used in the ICI arm.
Most of the drugs evaluated were PD-1 and PD-L1 inhibitors. The specific drugs assessed included ipilimumab, tremelimumab, nivolumab, pembrolizumab, atezolizumab, durvalumab, and avelumab.
A total of 32 trials that involved more than 20,000 patients reported HRs for death according to the patients’ sex. Thirty-four trials that involved more than 21,000 patients reported HRs for death according to patients’ age, and 30 trials that involved more than 19,000 patients reported HRs for death according to patients’ ECOG PS.
No significant differences in OS benefit were seen by cancer type, line of therapy, agent of immunotherapy, or intervention strategy, the investigators pointed out.
There were also no differences in survival benefit associated with immunotherapy vs control therapies for patients with an ECOG PS of 0 and an ECOG PS of 1 or greater. The OS benefit was 0.81 for those with an ECOG PS of 0 and 0.79 for those with an ECOG PS of 1 or greater.
Wang has disclosed no relevant financial relationships.
This article first appeared on Medscape.com .
Selpercatinib ‘poised to alter the landscape’ of RET+ cancers
Clinical data for the first-ever RET inhibitor, selpercatinib (Retevmo), show efficacy in two groups of patients with cancer – those with RET fusion–positive non–small cell lung cancer (NSCLC), and those with RET-mutant medullary thyroid cancer (MTC).
The drug showed “very good efficacy and also very good tolerability” in both groups, said lead author Lori J. Wirth, MD, medical director of head and neck cancers, Massachusetts General Hospital Cancer Center, Boston, in a statement.
“The response rates are high, responses are very durable, and overall, the drug does not cause a lot of toxicity,” she said.
“If you have a clean, RET-specific inhibitor such as selpercatinib, then you can really pound down RET very strongly and hit the driver alteration much harder, with a better side effect profile,” Dr. Wirth added.
Both groups of patients were part of the phase 1/2 LIBRETTO-001 study, which served as the basis for the recent accelerated approval of selpercatinib by the Food and Drug Administration.
Data from LIBRETTO-001 were published in the New England Journal of Medicine as two articles, one on NSCLC patients and one on MTC patients.
There has been a “remarkable increase” in the number of targeted agents that are effective in treating patients with advanced cancers that harbor specific genomic alterations, commented Razelle Kurzrock, MD, from the University of California, San Diego, in an accompanying editorial.
Selpercatinib, a potent RET inhibitor, “is now poised to alter the landscape of another genomic subgroup – RET-altered cancers,” she wrote.
Multikinase inhibitors such as vandetanib and cabozantinib have ancillary RET-inhibitor activity and are also active against RET-driven cancers. But these drugs are limited by off-target side effects, Dr. Krurzrock pointed out. “In contrast, next-generation, highly potent, and selective RET inhibitors such as selpercatinib offer the potential for improved efficacy and a more satisfactory side effect profile.”
In both parts of the study, selpercatinib produced durable responses in a majority of patients. Only about 3% of patients discontinued taking selpercatinib because of drug-related adverse events.
Taken together, these results show that selpercatinib “had marked and durable antitumor activity in most patients with RET-altered thyroid cancer or NSCLC,” wrote Dr. Krurzrock. “RET abnormalities now join other genomic alterations such as NTRK fusions, tumor mutational burden, and deficient mismatchrepair genes across cancers and ALK, BRAF, EGFR, MET, and ROS1 alterations in NSCLC that warrant molecular screening strategies.”
Results in patients with RET-mutated NSCLC
All patients enrolled in the LIBRETTO-001 trial received selpercatinib 160 mg orally twice daily until disease progression or unacceptable toxicity occurred.
Of 105 patients with NSCLC who had received at least one platinum-based chemotherapy regimen, the objective response rate was 64%. The median duration of response was 17.5 months.
At a median follow-up of 12.1 months, 63% of the responses were ongoing.
The cohort included 39 treatment-naive patients, among whom the response rate was even higher, at 85%; 90% of the responses were ongoing at 6 months. In addition, 11 patients had measurable central nervous system metastasis at study enrollment. Of this group, 91% achieved an intracranial response.
Common adverse events of grade 3 or higher included hypertension (in 14% of the patients), an increase in ALT level (in 12%), an increase in AST level (in 10%), hyponatremia (in 6%), and lymphopenia (in 6%). The drug was discontinued in 12 patients because of a drug-related adverse event.
Results in patients with RET-mutated MTC
Efficacy for MTC was evaluated in 55 patients with advanced or metastatic RET-mutant MTC who had previously been treated with cabozantinib, vandetanib, or both. The objective response rate was 69%. The 1-year progression-free survival rate was 82%.
For the 88 patients who had not previously received vandetanib or cabozantinib, the response rate was 73%. The 1-year progression-free survival rate was 92%.
In a subgroup of 19 patients with previously treated RET fusion–positive thyroid cancer, 79% responded to the therapy; 1-year progression-free survival was 64%.
The most common adverse events of grade 3 or higher were hypertension (in 21% of the patients), an increase in ALT level (in 11%), an increase in AST level (in 9%), hyponatremia (in 8%), and diarrhea (in 6%). Selpercatinib was discontinued by 12 patients because of drug-related adverse events.
The study was funded by Loxo Oncology (a wholly owned subsidiary of Eli Lilly) and by grants from the National Institutes of Health and the University of Texas MD Anderson Cancer Center. Kurzrock and Wirth report relationships with numerous pharmaceutical companies, as listed in the journal article.
This article first appeared on Medscape.com.
Clinical data for the first-ever RET inhibitor, selpercatinib (Retevmo), show efficacy in two groups of patients with cancer – those with RET fusion–positive non–small cell lung cancer (NSCLC), and those with RET-mutant medullary thyroid cancer (MTC).
The drug showed “very good efficacy and also very good tolerability” in both groups, said lead author Lori J. Wirth, MD, medical director of head and neck cancers, Massachusetts General Hospital Cancer Center, Boston, in a statement.
“The response rates are high, responses are very durable, and overall, the drug does not cause a lot of toxicity,” she said.
“If you have a clean, RET-specific inhibitor such as selpercatinib, then you can really pound down RET very strongly and hit the driver alteration much harder, with a better side effect profile,” Dr. Wirth added.
Both groups of patients were part of the phase 1/2 LIBRETTO-001 study, which served as the basis for the recent accelerated approval of selpercatinib by the Food and Drug Administration.
Data from LIBRETTO-001 were published in the New England Journal of Medicine as two articles, one on NSCLC patients and one on MTC patients.
There has been a “remarkable increase” in the number of targeted agents that are effective in treating patients with advanced cancers that harbor specific genomic alterations, commented Razelle Kurzrock, MD, from the University of California, San Diego, in an accompanying editorial.
Selpercatinib, a potent RET inhibitor, “is now poised to alter the landscape of another genomic subgroup – RET-altered cancers,” she wrote.
Multikinase inhibitors such as vandetanib and cabozantinib have ancillary RET-inhibitor activity and are also active against RET-driven cancers. But these drugs are limited by off-target side effects, Dr. Krurzrock pointed out. “In contrast, next-generation, highly potent, and selective RET inhibitors such as selpercatinib offer the potential for improved efficacy and a more satisfactory side effect profile.”
In both parts of the study, selpercatinib produced durable responses in a majority of patients. Only about 3% of patients discontinued taking selpercatinib because of drug-related adverse events.
Taken together, these results show that selpercatinib “had marked and durable antitumor activity in most patients with RET-altered thyroid cancer or NSCLC,” wrote Dr. Krurzrock. “RET abnormalities now join other genomic alterations such as NTRK fusions, tumor mutational burden, and deficient mismatchrepair genes across cancers and ALK, BRAF, EGFR, MET, and ROS1 alterations in NSCLC that warrant molecular screening strategies.”
Results in patients with RET-mutated NSCLC
All patients enrolled in the LIBRETTO-001 trial received selpercatinib 160 mg orally twice daily until disease progression or unacceptable toxicity occurred.
Of 105 patients with NSCLC who had received at least one platinum-based chemotherapy regimen, the objective response rate was 64%. The median duration of response was 17.5 months.
At a median follow-up of 12.1 months, 63% of the responses were ongoing.
The cohort included 39 treatment-naive patients, among whom the response rate was even higher, at 85%; 90% of the responses were ongoing at 6 months. In addition, 11 patients had measurable central nervous system metastasis at study enrollment. Of this group, 91% achieved an intracranial response.
Common adverse events of grade 3 or higher included hypertension (in 14% of the patients), an increase in ALT level (in 12%), an increase in AST level (in 10%), hyponatremia (in 6%), and lymphopenia (in 6%). The drug was discontinued in 12 patients because of a drug-related adverse event.
Results in patients with RET-mutated MTC
Efficacy for MTC was evaluated in 55 patients with advanced or metastatic RET-mutant MTC who had previously been treated with cabozantinib, vandetanib, or both. The objective response rate was 69%. The 1-year progression-free survival rate was 82%.
For the 88 patients who had not previously received vandetanib or cabozantinib, the response rate was 73%. The 1-year progression-free survival rate was 92%.
In a subgroup of 19 patients with previously treated RET fusion–positive thyroid cancer, 79% responded to the therapy; 1-year progression-free survival was 64%.
The most common adverse events of grade 3 or higher were hypertension (in 21% of the patients), an increase in ALT level (in 11%), an increase in AST level (in 9%), hyponatremia (in 8%), and diarrhea (in 6%). Selpercatinib was discontinued by 12 patients because of drug-related adverse events.
The study was funded by Loxo Oncology (a wholly owned subsidiary of Eli Lilly) and by grants from the National Institutes of Health and the University of Texas MD Anderson Cancer Center. Kurzrock and Wirth report relationships with numerous pharmaceutical companies, as listed in the journal article.
This article first appeared on Medscape.com.
Clinical data for the first-ever RET inhibitor, selpercatinib (Retevmo), show efficacy in two groups of patients with cancer – those with RET fusion–positive non–small cell lung cancer (NSCLC), and those with RET-mutant medullary thyroid cancer (MTC).
The drug showed “very good efficacy and also very good tolerability” in both groups, said lead author Lori J. Wirth, MD, medical director of head and neck cancers, Massachusetts General Hospital Cancer Center, Boston, in a statement.
“The response rates are high, responses are very durable, and overall, the drug does not cause a lot of toxicity,” she said.
“If you have a clean, RET-specific inhibitor such as selpercatinib, then you can really pound down RET very strongly and hit the driver alteration much harder, with a better side effect profile,” Dr. Wirth added.
Both groups of patients were part of the phase 1/2 LIBRETTO-001 study, which served as the basis for the recent accelerated approval of selpercatinib by the Food and Drug Administration.
Data from LIBRETTO-001 were published in the New England Journal of Medicine as two articles, one on NSCLC patients and one on MTC patients.
There has been a “remarkable increase” in the number of targeted agents that are effective in treating patients with advanced cancers that harbor specific genomic alterations, commented Razelle Kurzrock, MD, from the University of California, San Diego, in an accompanying editorial.
Selpercatinib, a potent RET inhibitor, “is now poised to alter the landscape of another genomic subgroup – RET-altered cancers,” she wrote.
Multikinase inhibitors such as vandetanib and cabozantinib have ancillary RET-inhibitor activity and are also active against RET-driven cancers. But these drugs are limited by off-target side effects, Dr. Krurzrock pointed out. “In contrast, next-generation, highly potent, and selective RET inhibitors such as selpercatinib offer the potential for improved efficacy and a more satisfactory side effect profile.”
In both parts of the study, selpercatinib produced durable responses in a majority of patients. Only about 3% of patients discontinued taking selpercatinib because of drug-related adverse events.
Taken together, these results show that selpercatinib “had marked and durable antitumor activity in most patients with RET-altered thyroid cancer or NSCLC,” wrote Dr. Krurzrock. “RET abnormalities now join other genomic alterations such as NTRK fusions, tumor mutational burden, and deficient mismatchrepair genes across cancers and ALK, BRAF, EGFR, MET, and ROS1 alterations in NSCLC that warrant molecular screening strategies.”
Results in patients with RET-mutated NSCLC
All patients enrolled in the LIBRETTO-001 trial received selpercatinib 160 mg orally twice daily until disease progression or unacceptable toxicity occurred.
Of 105 patients with NSCLC who had received at least one platinum-based chemotherapy regimen, the objective response rate was 64%. The median duration of response was 17.5 months.
At a median follow-up of 12.1 months, 63% of the responses were ongoing.
The cohort included 39 treatment-naive patients, among whom the response rate was even higher, at 85%; 90% of the responses were ongoing at 6 months. In addition, 11 patients had measurable central nervous system metastasis at study enrollment. Of this group, 91% achieved an intracranial response.
Common adverse events of grade 3 or higher included hypertension (in 14% of the patients), an increase in ALT level (in 12%), an increase in AST level (in 10%), hyponatremia (in 6%), and lymphopenia (in 6%). The drug was discontinued in 12 patients because of a drug-related adverse event.
Results in patients with RET-mutated MTC
Efficacy for MTC was evaluated in 55 patients with advanced or metastatic RET-mutant MTC who had previously been treated with cabozantinib, vandetanib, or both. The objective response rate was 69%. The 1-year progression-free survival rate was 82%.
For the 88 patients who had not previously received vandetanib or cabozantinib, the response rate was 73%. The 1-year progression-free survival rate was 92%.
In a subgroup of 19 patients with previously treated RET fusion–positive thyroid cancer, 79% responded to the therapy; 1-year progression-free survival was 64%.
The most common adverse events of grade 3 or higher were hypertension (in 21% of the patients), an increase in ALT level (in 11%), an increase in AST level (in 9%), hyponatremia (in 8%), and diarrhea (in 6%). Selpercatinib was discontinued by 12 patients because of drug-related adverse events.
The study was funded by Loxo Oncology (a wholly owned subsidiary of Eli Lilly) and by grants from the National Institutes of Health and the University of Texas MD Anderson Cancer Center. Kurzrock and Wirth report relationships with numerous pharmaceutical companies, as listed in the journal article.
This article first appeared on Medscape.com.
Aspirin may accelerate cancer progression in older adults
Aspirin may accelerate the progression of advanced cancers and lead to an earlier death as a result, new data from the ASPREE study suggest.
The results showed that patients 65 years and older who started taking daily low-dose aspirin had a 19% higher chance of being diagnosed with metastatic cancer, a 22% higher chance of being diagnosed with a stage 4 tumor, and a 31% increased risk of death from stage 4 cancer, when compared with patients who took a placebo.
John J. McNeil, MBBS, PhD, of Monash University in Melbourne, Australia, and colleagues detailed these findings in the Journal of the National Cancer Institute.
“If confirmed, the clinical implications of these findings could be important for the use of aspirin in an older population,” the authors wrote.
When results of the ASPREE study were first reported in 2018, they “raised important concerns,” Ernest Hawk, MD, and Karen Colbert Maresso wrote in an editorial related to the current publication.
“Unlike ARRIVE, ASCEND, and nearly all prior primary prevention CVD [cardiovascular disease] trials of aspirin, ASPREE surprisingly demonstrated increased all-cause mortality in the aspirin group, which appeared to be driven largely by an increase in cancer-related deaths,” wrote the editorialists, who are both from the University of Texas MD Anderson Cancer Center in Houston.
Even though the ASPREE investigators have now taken a deeper dive into their data, the findings “neither explain nor alleviate the concerns raised by the initial ASPREE report,” the editorialists noted.
ASPREE design and results
ASPREE is a multicenter, double-blind trial of 19,114 older adults living in Australia (n = 16,703) or the United States (n = 2,411). Most patients were 70 years or older at baseline. However, the U.S. group also included patients 65 years and older who were racial/ethnic minorities (n = 564).
Patients were randomized to receive 100 mg of enteric-coated aspirin daily (n = 9,525) or matching placebo (n = 9,589) from March 2010 through December 2014.
At inclusion, all participants were free from cardiovascular disease, dementia, or physical disability. A previous history of cancer was not used to exclude participants, and 19.1% of patients had cancer at randomization. Most patients (89%) had not used aspirin regularly before entering the trial.
At a median follow-up of 4.7 years, there were 981 incident cancer events in the aspirin-treated group and 952 in the placebo-treated group, with an overall incident cancer rate of 10.1%.
Of the 1,933 patients with newly diagnosed cancer, 65.7% had a localized cancer, 18.8% had a new metastatic cancer, 5.8% had metastatic disease from an existing cancer, and 9.7% had a new hematologic or lymphatic cancer.
A quarter of cancer patients (n = 495) died as a result of their malignancy, with 52 dying from a cancer they already had at randomization.
Aspirin was not associated with the risk of first incident cancer diagnosis or incident localized cancer diagnosis. The hazard ratios were 1.04 for all incident cancers (95% confidence interval, 0.95-1.14) and 0.99 for incident localized cancers (95% CI, 0.89-1.11).
However, aspirin was associated with an increased risk of metastatic cancer and cancer presenting at stage 4. The HR for metastatic cancer was 1.19 (95% CI, 1.00-1.43), and the HR for newly diagnosed stage 4 cancer was 1.22 (95% CI, 1.02-1.45).
Furthermore, “an increased progression to death was observed amongst those randomized to aspirin, regardless of whether the initial cancer presentation had been localized or metastatic,” the investigators wrote.
The HRs for death were 1.35 for all cancers (95% CI, 1.13-1.61), 1.47 for localized cancers (95% CI, 1.07-2.02), and 1.30 for metastatic cancers (95% CI, 1.03-1.63).
“Deaths were particularly high among those on aspirin who were diagnosed with advanced solid cancers,” study author Andrew Chan, MD, of Massachusetts General Hospital in Boston, said in a press statement.
Indeed, HRs for death in patients with solid tumors presenting at stage 3 and 4 were a respective 2.11 (95% CI, 1.03-4.33) and 1.31 (95% CI, 1.04-1.64). This suggests a possible adverse effect of aspirin on the growth of cancers once they have already developed in older adults, Dr. Chan said.
Where does that leave aspirin for cancer prevention?
“Although these results suggest that we should be cautious about starting aspirin therapy in otherwise healthy older adults, this does not mean that individuals who are already taking aspirin – particularly if they began taking it at a younger age – should stop their aspirin regimen,” Dr. Chan said.
There are decades of data supporting the use of daily aspirin to prevent multiple cancer types, particularly colorectal cancer, in individuals under the age of 70 years. In a recent meta-analysis, for example, regular aspirin use was linked to a 27% reduced risk for colorectal cancer, a 33% reduced risk for squamous cell esophageal cancer, a 39% decreased risk for adenocarcinoma of the esophagus and gastric cardia, a 36% decreased risk for stomach cancer, a 38% decreased risk for hepatobiliary tract cancer, and a 22% decreased risk for pancreatic cancer.
While these figures are mostly based on observational and case-control studies, it “reaffirms the fact that, overall, when you look at all of the ages, that there is still a benefit of aspirin for cancer,” John Cuzick, PhD, of Queen Mary University of London (England), said in an interview.
In fact, the meta-analysis goes as far as suggesting that perhaps the dose of aspirin being used is too low, with the authors noting that there was a 35% risk reduction in colorectal cancer with a dose of 325 mg daily. That’s a new finding, Dr. Cuzick said.
He noted that the ASPREE study largely consists of patients 70 years of age or older, and the authors “draw some conclusions which we can’t ignore about potential safety.”
One of the safety concerns is the increased risk for gastrointestinal bleeding, which is why Dr. Cuzick and colleagues previously recommended caution in the use of aspirin to prevent cancer in elderly patients. The group published a study in 2015 that suggested a benefit of taking aspirin daily for 5-10 years in patients aged 50-65 years, but the risk/benefit ratio was unclear for patients 70 years and older.
The ASPREE data now add to those uncertainties and suggest “there may be some side effects that we do not understand,” Dr. Cuzick said.
“I’m still optimistic that aspirin is going to be important for cancer prevention, but probably focusing on ages 50-70,” he added. “[The ASPREE data] reinforce the caution that we have to take in terms of trying to understand what the side effects are and what’s going on at these older ages.”
Dr. Cuzick is currently leading the AsCaP Project, an international effort to better understand why aspirin might work in preventing some cancer types but not others. AsCaP is supported by Cancer Research UK and also includes Dr. Chan among the researchers attempting to find out which patients may benefit the most from aspirin and which may be at greater risk of adverse effects.
The ASPREE trial was funded by grants from the National Institute on Aging, the National Cancer Institute, the National Health and Medical Research Council of Australia, Monash University, and the Victorian Cancer Agency. Several ASPREE investigators disclosed financial relationships with Bayer Pharma. The editorialists had no conflicts of interest. Dr. Cuzick has been an advisory board member for Bayer in the past.
SOURCE: McNeil J et al. J Natl Cancer Inst. 2020 Aug 11. doi: 10.1093/jnci/djaa114.
Aspirin may accelerate the progression of advanced cancers and lead to an earlier death as a result, new data from the ASPREE study suggest.
The results showed that patients 65 years and older who started taking daily low-dose aspirin had a 19% higher chance of being diagnosed with metastatic cancer, a 22% higher chance of being diagnosed with a stage 4 tumor, and a 31% increased risk of death from stage 4 cancer, when compared with patients who took a placebo.
John J. McNeil, MBBS, PhD, of Monash University in Melbourne, Australia, and colleagues detailed these findings in the Journal of the National Cancer Institute.
“If confirmed, the clinical implications of these findings could be important for the use of aspirin in an older population,” the authors wrote.
When results of the ASPREE study were first reported in 2018, they “raised important concerns,” Ernest Hawk, MD, and Karen Colbert Maresso wrote in an editorial related to the current publication.
“Unlike ARRIVE, ASCEND, and nearly all prior primary prevention CVD [cardiovascular disease] trials of aspirin, ASPREE surprisingly demonstrated increased all-cause mortality in the aspirin group, which appeared to be driven largely by an increase in cancer-related deaths,” wrote the editorialists, who are both from the University of Texas MD Anderson Cancer Center in Houston.
Even though the ASPREE investigators have now taken a deeper dive into their data, the findings “neither explain nor alleviate the concerns raised by the initial ASPREE report,” the editorialists noted.
ASPREE design and results
ASPREE is a multicenter, double-blind trial of 19,114 older adults living in Australia (n = 16,703) or the United States (n = 2,411). Most patients were 70 years or older at baseline. However, the U.S. group also included patients 65 years and older who were racial/ethnic minorities (n = 564).
Patients were randomized to receive 100 mg of enteric-coated aspirin daily (n = 9,525) or matching placebo (n = 9,589) from March 2010 through December 2014.
At inclusion, all participants were free from cardiovascular disease, dementia, or physical disability. A previous history of cancer was not used to exclude participants, and 19.1% of patients had cancer at randomization. Most patients (89%) had not used aspirin regularly before entering the trial.
At a median follow-up of 4.7 years, there were 981 incident cancer events in the aspirin-treated group and 952 in the placebo-treated group, with an overall incident cancer rate of 10.1%.
Of the 1,933 patients with newly diagnosed cancer, 65.7% had a localized cancer, 18.8% had a new metastatic cancer, 5.8% had metastatic disease from an existing cancer, and 9.7% had a new hematologic or lymphatic cancer.
A quarter of cancer patients (n = 495) died as a result of their malignancy, with 52 dying from a cancer they already had at randomization.
Aspirin was not associated with the risk of first incident cancer diagnosis or incident localized cancer diagnosis. The hazard ratios were 1.04 for all incident cancers (95% confidence interval, 0.95-1.14) and 0.99 for incident localized cancers (95% CI, 0.89-1.11).
However, aspirin was associated with an increased risk of metastatic cancer and cancer presenting at stage 4. The HR for metastatic cancer was 1.19 (95% CI, 1.00-1.43), and the HR for newly diagnosed stage 4 cancer was 1.22 (95% CI, 1.02-1.45).
Furthermore, “an increased progression to death was observed amongst those randomized to aspirin, regardless of whether the initial cancer presentation had been localized or metastatic,” the investigators wrote.
The HRs for death were 1.35 for all cancers (95% CI, 1.13-1.61), 1.47 for localized cancers (95% CI, 1.07-2.02), and 1.30 for metastatic cancers (95% CI, 1.03-1.63).
“Deaths were particularly high among those on aspirin who were diagnosed with advanced solid cancers,” study author Andrew Chan, MD, of Massachusetts General Hospital in Boston, said in a press statement.
Indeed, HRs for death in patients with solid tumors presenting at stage 3 and 4 were a respective 2.11 (95% CI, 1.03-4.33) and 1.31 (95% CI, 1.04-1.64). This suggests a possible adverse effect of aspirin on the growth of cancers once they have already developed in older adults, Dr. Chan said.
Where does that leave aspirin for cancer prevention?
“Although these results suggest that we should be cautious about starting aspirin therapy in otherwise healthy older adults, this does not mean that individuals who are already taking aspirin – particularly if they began taking it at a younger age – should stop their aspirin regimen,” Dr. Chan said.
There are decades of data supporting the use of daily aspirin to prevent multiple cancer types, particularly colorectal cancer, in individuals under the age of 70 years. In a recent meta-analysis, for example, regular aspirin use was linked to a 27% reduced risk for colorectal cancer, a 33% reduced risk for squamous cell esophageal cancer, a 39% decreased risk for adenocarcinoma of the esophagus and gastric cardia, a 36% decreased risk for stomach cancer, a 38% decreased risk for hepatobiliary tract cancer, and a 22% decreased risk for pancreatic cancer.
While these figures are mostly based on observational and case-control studies, it “reaffirms the fact that, overall, when you look at all of the ages, that there is still a benefit of aspirin for cancer,” John Cuzick, PhD, of Queen Mary University of London (England), said in an interview.
In fact, the meta-analysis goes as far as suggesting that perhaps the dose of aspirin being used is too low, with the authors noting that there was a 35% risk reduction in colorectal cancer with a dose of 325 mg daily. That’s a new finding, Dr. Cuzick said.
He noted that the ASPREE study largely consists of patients 70 years of age or older, and the authors “draw some conclusions which we can’t ignore about potential safety.”
One of the safety concerns is the increased risk for gastrointestinal bleeding, which is why Dr. Cuzick and colleagues previously recommended caution in the use of aspirin to prevent cancer in elderly patients. The group published a study in 2015 that suggested a benefit of taking aspirin daily for 5-10 years in patients aged 50-65 years, but the risk/benefit ratio was unclear for patients 70 years and older.
The ASPREE data now add to those uncertainties and suggest “there may be some side effects that we do not understand,” Dr. Cuzick said.
“I’m still optimistic that aspirin is going to be important for cancer prevention, but probably focusing on ages 50-70,” he added. “[The ASPREE data] reinforce the caution that we have to take in terms of trying to understand what the side effects are and what’s going on at these older ages.”
Dr. Cuzick is currently leading the AsCaP Project, an international effort to better understand why aspirin might work in preventing some cancer types but not others. AsCaP is supported by Cancer Research UK and also includes Dr. Chan among the researchers attempting to find out which patients may benefit the most from aspirin and which may be at greater risk of adverse effects.
The ASPREE trial was funded by grants from the National Institute on Aging, the National Cancer Institute, the National Health and Medical Research Council of Australia, Monash University, and the Victorian Cancer Agency. Several ASPREE investigators disclosed financial relationships with Bayer Pharma. The editorialists had no conflicts of interest. Dr. Cuzick has been an advisory board member for Bayer in the past.
SOURCE: McNeil J et al. J Natl Cancer Inst. 2020 Aug 11. doi: 10.1093/jnci/djaa114.
Aspirin may accelerate the progression of advanced cancers and lead to an earlier death as a result, new data from the ASPREE study suggest.
The results showed that patients 65 years and older who started taking daily low-dose aspirin had a 19% higher chance of being diagnosed with metastatic cancer, a 22% higher chance of being diagnosed with a stage 4 tumor, and a 31% increased risk of death from stage 4 cancer, when compared with patients who took a placebo.
John J. McNeil, MBBS, PhD, of Monash University in Melbourne, Australia, and colleagues detailed these findings in the Journal of the National Cancer Institute.
“If confirmed, the clinical implications of these findings could be important for the use of aspirin in an older population,” the authors wrote.
When results of the ASPREE study were first reported in 2018, they “raised important concerns,” Ernest Hawk, MD, and Karen Colbert Maresso wrote in an editorial related to the current publication.
“Unlike ARRIVE, ASCEND, and nearly all prior primary prevention CVD [cardiovascular disease] trials of aspirin, ASPREE surprisingly demonstrated increased all-cause mortality in the aspirin group, which appeared to be driven largely by an increase in cancer-related deaths,” wrote the editorialists, who are both from the University of Texas MD Anderson Cancer Center in Houston.
Even though the ASPREE investigators have now taken a deeper dive into their data, the findings “neither explain nor alleviate the concerns raised by the initial ASPREE report,” the editorialists noted.
ASPREE design and results
ASPREE is a multicenter, double-blind trial of 19,114 older adults living in Australia (n = 16,703) or the United States (n = 2,411). Most patients were 70 years or older at baseline. However, the U.S. group also included patients 65 years and older who were racial/ethnic minorities (n = 564).
Patients were randomized to receive 100 mg of enteric-coated aspirin daily (n = 9,525) or matching placebo (n = 9,589) from March 2010 through December 2014.
At inclusion, all participants were free from cardiovascular disease, dementia, or physical disability. A previous history of cancer was not used to exclude participants, and 19.1% of patients had cancer at randomization. Most patients (89%) had not used aspirin regularly before entering the trial.
At a median follow-up of 4.7 years, there were 981 incident cancer events in the aspirin-treated group and 952 in the placebo-treated group, with an overall incident cancer rate of 10.1%.
Of the 1,933 patients with newly diagnosed cancer, 65.7% had a localized cancer, 18.8% had a new metastatic cancer, 5.8% had metastatic disease from an existing cancer, and 9.7% had a new hematologic or lymphatic cancer.
A quarter of cancer patients (n = 495) died as a result of their malignancy, with 52 dying from a cancer they already had at randomization.
Aspirin was not associated with the risk of first incident cancer diagnosis or incident localized cancer diagnosis. The hazard ratios were 1.04 for all incident cancers (95% confidence interval, 0.95-1.14) and 0.99 for incident localized cancers (95% CI, 0.89-1.11).
However, aspirin was associated with an increased risk of metastatic cancer and cancer presenting at stage 4. The HR for metastatic cancer was 1.19 (95% CI, 1.00-1.43), and the HR for newly diagnosed stage 4 cancer was 1.22 (95% CI, 1.02-1.45).
Furthermore, “an increased progression to death was observed amongst those randomized to aspirin, regardless of whether the initial cancer presentation had been localized or metastatic,” the investigators wrote.
The HRs for death were 1.35 for all cancers (95% CI, 1.13-1.61), 1.47 for localized cancers (95% CI, 1.07-2.02), and 1.30 for metastatic cancers (95% CI, 1.03-1.63).
“Deaths were particularly high among those on aspirin who were diagnosed with advanced solid cancers,” study author Andrew Chan, MD, of Massachusetts General Hospital in Boston, said in a press statement.
Indeed, HRs for death in patients with solid tumors presenting at stage 3 and 4 were a respective 2.11 (95% CI, 1.03-4.33) and 1.31 (95% CI, 1.04-1.64). This suggests a possible adverse effect of aspirin on the growth of cancers once they have already developed in older adults, Dr. Chan said.
Where does that leave aspirin for cancer prevention?
“Although these results suggest that we should be cautious about starting aspirin therapy in otherwise healthy older adults, this does not mean that individuals who are already taking aspirin – particularly if they began taking it at a younger age – should stop their aspirin regimen,” Dr. Chan said.
There are decades of data supporting the use of daily aspirin to prevent multiple cancer types, particularly colorectal cancer, in individuals under the age of 70 years. In a recent meta-analysis, for example, regular aspirin use was linked to a 27% reduced risk for colorectal cancer, a 33% reduced risk for squamous cell esophageal cancer, a 39% decreased risk for adenocarcinoma of the esophagus and gastric cardia, a 36% decreased risk for stomach cancer, a 38% decreased risk for hepatobiliary tract cancer, and a 22% decreased risk for pancreatic cancer.
While these figures are mostly based on observational and case-control studies, it “reaffirms the fact that, overall, when you look at all of the ages, that there is still a benefit of aspirin for cancer,” John Cuzick, PhD, of Queen Mary University of London (England), said in an interview.
In fact, the meta-analysis goes as far as suggesting that perhaps the dose of aspirin being used is too low, with the authors noting that there was a 35% risk reduction in colorectal cancer with a dose of 325 mg daily. That’s a new finding, Dr. Cuzick said.
He noted that the ASPREE study largely consists of patients 70 years of age or older, and the authors “draw some conclusions which we can’t ignore about potential safety.”
One of the safety concerns is the increased risk for gastrointestinal bleeding, which is why Dr. Cuzick and colleagues previously recommended caution in the use of aspirin to prevent cancer in elderly patients. The group published a study in 2015 that suggested a benefit of taking aspirin daily for 5-10 years in patients aged 50-65 years, but the risk/benefit ratio was unclear for patients 70 years and older.
The ASPREE data now add to those uncertainties and suggest “there may be some side effects that we do not understand,” Dr. Cuzick said.
“I’m still optimistic that aspirin is going to be important for cancer prevention, but probably focusing on ages 50-70,” he added. “[The ASPREE data] reinforce the caution that we have to take in terms of trying to understand what the side effects are and what’s going on at these older ages.”
Dr. Cuzick is currently leading the AsCaP Project, an international effort to better understand why aspirin might work in preventing some cancer types but not others. AsCaP is supported by Cancer Research UK and also includes Dr. Chan among the researchers attempting to find out which patients may benefit the most from aspirin and which may be at greater risk of adverse effects.
The ASPREE trial was funded by grants from the National Institute on Aging, the National Cancer Institute, the National Health and Medical Research Council of Australia, Monash University, and the Victorian Cancer Agency. Several ASPREE investigators disclosed financial relationships with Bayer Pharma. The editorialists had no conflicts of interest. Dr. Cuzick has been an advisory board member for Bayer in the past.
SOURCE: McNeil J et al. J Natl Cancer Inst. 2020 Aug 11. doi: 10.1093/jnci/djaa114.
FROM JOURNAL OF THE NATIONAL CANCER INSTITUTE
Black/White gap gone: ‘The only cancer where this has happened’
The historically higher incidence rates of lung cancer among Black men, compared with White men, in the United States have all but been eliminated, at least among most men in the younger age groups, a new analysis from the American Cancer Society (ACS) indicates.
Among women, the trend is even more impressive, as the Black/White gap in lung cancer incidence rates has actually reversed in younger women. Black women in certain age groups are now less likely to develop lung cancer than White women, the same study indicates.
These trends reflect the steeper declines in smoking rates among Blacks in the US, compared with comparably-aged Whites, say the authors.
“This is the only cancer where this has happened,” lead author Ahmedin Jemal, DVM, PhD, senior vice president for Data Science at the American Cancer Society, told Medscape Medical News.
“If you look at cancers that are affected by access to screening and treatment, the disparity between the Blacks and the Whites has been increasing over the years because tests and treatment require access to insurance, so the Whites are getting more of them than the Blacks,” Jemal explained.
“But for smoking, all you have to do for prevention is just don’t smoke, so this is a success story that really should be highlighted,” he emphasized.
The study was published online Aug. 20 in JNCI Cancer: Spectrum.
Nationwide Incidence Data
For this study, Jemal and colleagues collected nationwide incidence data on individuals between ages 30 and 54 who had been diagnosed with lung cancer between 1997 and 2016.
“We categorized age at diagnosis by 5-year age intervals (from 30-34 to 50-54 years) and year of diagnosis by 5-year calendar period (from 1997-2001 to 2012-2016),” the investigators explain.
Analyses showed that lung cancer incidence rates generally decreased among both Black and White men during the study interval but the decline in incidence rates was steeper in Black men than in White men. As a consequence, the Black-to-White incidence rate ratios (IRRs) became similar in men born between 1967 and 1972 and reversed in women born since about 1967. For example, the Black-to-White IRRs in men between the ages of 40 and 44 who were born between 1957 and 1972 declined from 1.92 to 1.03.
Similarly, lung cancer incidence rates during the study interval declined among both Black and White women between the ages of 30 and 49 but, again, the decline was “considerably larger” among Black women. As a consequence, the Black-to-White IRR in women age 45 to 49 dropped from 1.25 during the period 1997-2001 down to 0.83 during the period 2012-2016.
This is in stark contrast to historical trends in lung cancer incidence rates, which were over 30% higher among similarly aged Black women born in the late 1950s. Now, lung cancer incidence rates are about 30% lower for similarly aged Black women born in 1972, compared with White women.
For Black and White women between age 50 and 54, lung cancer incidence rates either declined only slightly or remained stable during the study interval, the investigators reported.
The one exception to the diminishing gap in lung cancer incidence rates between Black and White men was an observed increase in IRRs in men born around the period 1977-1982
Among this group of men, who were between age 30 and 39 in the years 2012-2016, lung cancer incidence rates were higher in Black men than in White men.
As the authors point out, this increase in lung cancer rates among young Black men likely reflects a rapid rise in smoking seen among Black youth in the 1990s.
This trend coincided with an R.J. Reynolds tobacco ad campaign in which African Americans were targeted; between 1991 and 1997, the prevalence of smoking among Black high school students doubled from 14.1% to 28.2%, the investigators point out, citing a 2008 Centers for Disease Control and Prevention report on cigarette use among US high school students.
Smoking Prevalence Rates
Smoking prevalence rates were derived from National Health Interview Survey data from 1970 to 2016.
Mirroring findings in the racial patterns of lung cancer incidence rates, smoking prevalence rates declined in successive birth cohorts in both Black and White males and females, but the decline was again steeper in Black men and women than it was in White men and women.
As a result, the historically higher sex-specific smoking prevalence rates seen historically in Blacks disappeared in men born around 1960, and reversed in women born at the same time, Jemal and colleagues point out.
As the authors explain, the more rapid decline in smoking prevalence after 1960 is likely a reflection of the “precipitous” drop in smoking initiation rates among Black teenagers starting about the late 1970s through to the early 1990s.
For example, among 12th graders, smoking prevalence rates between 1977 and 1992 dropped from 36.7% to 8.1% among Black teens. In stark contrast, they hardly changed at all among White teens, dropping only from 38.3% in 1977 to 31.8% in 1992.
Jemal suggested that steeper decline in smoking initiation rates seen between the late 1970s and early 90s reflects the fact that Black teenagers were deterred from smoking because the cost of cigarettes kept going up.
He also suggested that smoking is less acceptable in the Black community than it is in the White community, especially among churchgoers, where smoking is severely frowned upon and nonsmoking is the community “norm.”
Additionally, Black youth may simply be heeding government antismoking messages to a greater extent than White youth, Jemal suggested.
He wondered if there are parallels now in the current pandemic. “When I go to a store here in Georgia, I would say almost all Blacks are wearing a mask [even though masks are not mandatory in Georgia] whereas it’s amazing the number of Whites who don’t wear a mask,” he recounts.
“So it would seem that Whites feel that government is simply interfering with their lives, while Blacks have a better perspective of the harms of smoking, so they are listening to the government’s antismoking campaigns,” he speculated.
Some Isolated Areas
Asked to comment on the study’s findings, Otis Brawley, MD, Bloomberg, distinguished professor of oncology and epidemiology at Johns Hopkins University in Baltimore, said that, while overall Black smoking rates are declining, there are some isolated areas where they are still very high.
For example, in his hometown of Baltimore, recent prevalence rates indicate that over 30% of Blacks are still smoking, “so these areas with high usage are still areas to focus on,” he told Medscape Medical News.
On the other hand, the study also supports the benefits of local, state, and federal government efforts to promote antismoking messages and tobacco-control activities over the past number of years.
“It proves that tactics used to control tobacco use have had some effect [even though] the study also shows that the tobacco industry’s advertising tactics such as the R.J. Reynolds targeted ads in the 90s can have deleterious effects,” Brawley noted.
Lung cancer has traditionally been one of the biggest drivers in the Black/White cancer mortality gap, Brawley said, adding that steeper declines in smoking initiation rates among Blacks compared with Whites are the main reason why this disparity is decreasing.
The study was supported by the Intramural Research Department of the American Cancer Society. The study authors have disclosed no relevant financial relationships. Brawley declares he does some consulting work for pharmaceutical company Genentech.
This article first appeared on Medscape.com.
The historically higher incidence rates of lung cancer among Black men, compared with White men, in the United States have all but been eliminated, at least among most men in the younger age groups, a new analysis from the American Cancer Society (ACS) indicates.
Among women, the trend is even more impressive, as the Black/White gap in lung cancer incidence rates has actually reversed in younger women. Black women in certain age groups are now less likely to develop lung cancer than White women, the same study indicates.
These trends reflect the steeper declines in smoking rates among Blacks in the US, compared with comparably-aged Whites, say the authors.
“This is the only cancer where this has happened,” lead author Ahmedin Jemal, DVM, PhD, senior vice president for Data Science at the American Cancer Society, told Medscape Medical News.
“If you look at cancers that are affected by access to screening and treatment, the disparity between the Blacks and the Whites has been increasing over the years because tests and treatment require access to insurance, so the Whites are getting more of them than the Blacks,” Jemal explained.
“But for smoking, all you have to do for prevention is just don’t smoke, so this is a success story that really should be highlighted,” he emphasized.
The study was published online Aug. 20 in JNCI Cancer: Spectrum.
Nationwide Incidence Data
For this study, Jemal and colleagues collected nationwide incidence data on individuals between ages 30 and 54 who had been diagnosed with lung cancer between 1997 and 2016.
“We categorized age at diagnosis by 5-year age intervals (from 30-34 to 50-54 years) and year of diagnosis by 5-year calendar period (from 1997-2001 to 2012-2016),” the investigators explain.
Analyses showed that lung cancer incidence rates generally decreased among both Black and White men during the study interval but the decline in incidence rates was steeper in Black men than in White men. As a consequence, the Black-to-White incidence rate ratios (IRRs) became similar in men born between 1967 and 1972 and reversed in women born since about 1967. For example, the Black-to-White IRRs in men between the ages of 40 and 44 who were born between 1957 and 1972 declined from 1.92 to 1.03.
Similarly, lung cancer incidence rates during the study interval declined among both Black and White women between the ages of 30 and 49 but, again, the decline was “considerably larger” among Black women. As a consequence, the Black-to-White IRR in women age 45 to 49 dropped from 1.25 during the period 1997-2001 down to 0.83 during the period 2012-2016.
This is in stark contrast to historical trends in lung cancer incidence rates, which were over 30% higher among similarly aged Black women born in the late 1950s. Now, lung cancer incidence rates are about 30% lower for similarly aged Black women born in 1972, compared with White women.
For Black and White women between age 50 and 54, lung cancer incidence rates either declined only slightly or remained stable during the study interval, the investigators reported.
The one exception to the diminishing gap in lung cancer incidence rates between Black and White men was an observed increase in IRRs in men born around the period 1977-1982
Among this group of men, who were between age 30 and 39 in the years 2012-2016, lung cancer incidence rates were higher in Black men than in White men.
As the authors point out, this increase in lung cancer rates among young Black men likely reflects a rapid rise in smoking seen among Black youth in the 1990s.
This trend coincided with an R.J. Reynolds tobacco ad campaign in which African Americans were targeted; between 1991 and 1997, the prevalence of smoking among Black high school students doubled from 14.1% to 28.2%, the investigators point out, citing a 2008 Centers for Disease Control and Prevention report on cigarette use among US high school students.
Smoking Prevalence Rates
Smoking prevalence rates were derived from National Health Interview Survey data from 1970 to 2016.
Mirroring findings in the racial patterns of lung cancer incidence rates, smoking prevalence rates declined in successive birth cohorts in both Black and White males and females, but the decline was again steeper in Black men and women than it was in White men and women.
As a result, the historically higher sex-specific smoking prevalence rates seen historically in Blacks disappeared in men born around 1960, and reversed in women born at the same time, Jemal and colleagues point out.
As the authors explain, the more rapid decline in smoking prevalence after 1960 is likely a reflection of the “precipitous” drop in smoking initiation rates among Black teenagers starting about the late 1970s through to the early 1990s.
For example, among 12th graders, smoking prevalence rates between 1977 and 1992 dropped from 36.7% to 8.1% among Black teens. In stark contrast, they hardly changed at all among White teens, dropping only from 38.3% in 1977 to 31.8% in 1992.
Jemal suggested that steeper decline in smoking initiation rates seen between the late 1970s and early 90s reflects the fact that Black teenagers were deterred from smoking because the cost of cigarettes kept going up.
He also suggested that smoking is less acceptable in the Black community than it is in the White community, especially among churchgoers, where smoking is severely frowned upon and nonsmoking is the community “norm.”
Additionally, Black youth may simply be heeding government antismoking messages to a greater extent than White youth, Jemal suggested.
He wondered if there are parallels now in the current pandemic. “When I go to a store here in Georgia, I would say almost all Blacks are wearing a mask [even though masks are not mandatory in Georgia] whereas it’s amazing the number of Whites who don’t wear a mask,” he recounts.
“So it would seem that Whites feel that government is simply interfering with their lives, while Blacks have a better perspective of the harms of smoking, so they are listening to the government’s antismoking campaigns,” he speculated.
Some Isolated Areas
Asked to comment on the study’s findings, Otis Brawley, MD, Bloomberg, distinguished professor of oncology and epidemiology at Johns Hopkins University in Baltimore, said that, while overall Black smoking rates are declining, there are some isolated areas where they are still very high.
For example, in his hometown of Baltimore, recent prevalence rates indicate that over 30% of Blacks are still smoking, “so these areas with high usage are still areas to focus on,” he told Medscape Medical News.
On the other hand, the study also supports the benefits of local, state, and federal government efforts to promote antismoking messages and tobacco-control activities over the past number of years.
“It proves that tactics used to control tobacco use have had some effect [even though] the study also shows that the tobacco industry’s advertising tactics such as the R.J. Reynolds targeted ads in the 90s can have deleterious effects,” Brawley noted.
Lung cancer has traditionally been one of the biggest drivers in the Black/White cancer mortality gap, Brawley said, adding that steeper declines in smoking initiation rates among Blacks compared with Whites are the main reason why this disparity is decreasing.
The study was supported by the Intramural Research Department of the American Cancer Society. The study authors have disclosed no relevant financial relationships. Brawley declares he does some consulting work for pharmaceutical company Genentech.
This article first appeared on Medscape.com.
The historically higher incidence rates of lung cancer among Black men, compared with White men, in the United States have all but been eliminated, at least among most men in the younger age groups, a new analysis from the American Cancer Society (ACS) indicates.
Among women, the trend is even more impressive, as the Black/White gap in lung cancer incidence rates has actually reversed in younger women. Black women in certain age groups are now less likely to develop lung cancer than White women, the same study indicates.
These trends reflect the steeper declines in smoking rates among Blacks in the US, compared with comparably-aged Whites, say the authors.
“This is the only cancer where this has happened,” lead author Ahmedin Jemal, DVM, PhD, senior vice president for Data Science at the American Cancer Society, told Medscape Medical News.
“If you look at cancers that are affected by access to screening and treatment, the disparity between the Blacks and the Whites has been increasing over the years because tests and treatment require access to insurance, so the Whites are getting more of them than the Blacks,” Jemal explained.
“But for smoking, all you have to do for prevention is just don’t smoke, so this is a success story that really should be highlighted,” he emphasized.
The study was published online Aug. 20 in JNCI Cancer: Spectrum.
Nationwide Incidence Data
For this study, Jemal and colleagues collected nationwide incidence data on individuals between ages 30 and 54 who had been diagnosed with lung cancer between 1997 and 2016.
“We categorized age at diagnosis by 5-year age intervals (from 30-34 to 50-54 years) and year of diagnosis by 5-year calendar period (from 1997-2001 to 2012-2016),” the investigators explain.
Analyses showed that lung cancer incidence rates generally decreased among both Black and White men during the study interval but the decline in incidence rates was steeper in Black men than in White men. As a consequence, the Black-to-White incidence rate ratios (IRRs) became similar in men born between 1967 and 1972 and reversed in women born since about 1967. For example, the Black-to-White IRRs in men between the ages of 40 and 44 who were born between 1957 and 1972 declined from 1.92 to 1.03.
Similarly, lung cancer incidence rates during the study interval declined among both Black and White women between the ages of 30 and 49 but, again, the decline was “considerably larger” among Black women. As a consequence, the Black-to-White IRR in women age 45 to 49 dropped from 1.25 during the period 1997-2001 down to 0.83 during the period 2012-2016.
This is in stark contrast to historical trends in lung cancer incidence rates, which were over 30% higher among similarly aged Black women born in the late 1950s. Now, lung cancer incidence rates are about 30% lower for similarly aged Black women born in 1972, compared with White women.
For Black and White women between age 50 and 54, lung cancer incidence rates either declined only slightly or remained stable during the study interval, the investigators reported.
The one exception to the diminishing gap in lung cancer incidence rates between Black and White men was an observed increase in IRRs in men born around the period 1977-1982
Among this group of men, who were between age 30 and 39 in the years 2012-2016, lung cancer incidence rates were higher in Black men than in White men.
As the authors point out, this increase in lung cancer rates among young Black men likely reflects a rapid rise in smoking seen among Black youth in the 1990s.
This trend coincided with an R.J. Reynolds tobacco ad campaign in which African Americans were targeted; between 1991 and 1997, the prevalence of smoking among Black high school students doubled from 14.1% to 28.2%, the investigators point out, citing a 2008 Centers for Disease Control and Prevention report on cigarette use among US high school students.
Smoking Prevalence Rates
Smoking prevalence rates were derived from National Health Interview Survey data from 1970 to 2016.
Mirroring findings in the racial patterns of lung cancer incidence rates, smoking prevalence rates declined in successive birth cohorts in both Black and White males and females, but the decline was again steeper in Black men and women than it was in White men and women.
As a result, the historically higher sex-specific smoking prevalence rates seen historically in Blacks disappeared in men born around 1960, and reversed in women born at the same time, Jemal and colleagues point out.
As the authors explain, the more rapid decline in smoking prevalence after 1960 is likely a reflection of the “precipitous” drop in smoking initiation rates among Black teenagers starting about the late 1970s through to the early 1990s.
For example, among 12th graders, smoking prevalence rates between 1977 and 1992 dropped from 36.7% to 8.1% among Black teens. In stark contrast, they hardly changed at all among White teens, dropping only from 38.3% in 1977 to 31.8% in 1992.
Jemal suggested that steeper decline in smoking initiation rates seen between the late 1970s and early 90s reflects the fact that Black teenagers were deterred from smoking because the cost of cigarettes kept going up.
He also suggested that smoking is less acceptable in the Black community than it is in the White community, especially among churchgoers, where smoking is severely frowned upon and nonsmoking is the community “norm.”
Additionally, Black youth may simply be heeding government antismoking messages to a greater extent than White youth, Jemal suggested.
He wondered if there are parallels now in the current pandemic. “When I go to a store here in Georgia, I would say almost all Blacks are wearing a mask [even though masks are not mandatory in Georgia] whereas it’s amazing the number of Whites who don’t wear a mask,” he recounts.
“So it would seem that Whites feel that government is simply interfering with their lives, while Blacks have a better perspective of the harms of smoking, so they are listening to the government’s antismoking campaigns,” he speculated.
Some Isolated Areas
Asked to comment on the study’s findings, Otis Brawley, MD, Bloomberg, distinguished professor of oncology and epidemiology at Johns Hopkins University in Baltimore, said that, while overall Black smoking rates are declining, there are some isolated areas where they are still very high.
For example, in his hometown of Baltimore, recent prevalence rates indicate that over 30% of Blacks are still smoking, “so these areas with high usage are still areas to focus on,” he told Medscape Medical News.
On the other hand, the study also supports the benefits of local, state, and federal government efforts to promote antismoking messages and tobacco-control activities over the past number of years.
“It proves that tactics used to control tobacco use have had some effect [even though] the study also shows that the tobacco industry’s advertising tactics such as the R.J. Reynolds targeted ads in the 90s can have deleterious effects,” Brawley noted.
Lung cancer has traditionally been one of the biggest drivers in the Black/White cancer mortality gap, Brawley said, adding that steeper declines in smoking initiation rates among Blacks compared with Whites are the main reason why this disparity is decreasing.
The study was supported by the Intramural Research Department of the American Cancer Society. The study authors have disclosed no relevant financial relationships. Brawley declares he does some consulting work for pharmaceutical company Genentech.
This article first appeared on Medscape.com.
Age, smoking among leading cancer risk factors for SLE patients
A new study has quantified cancer risk factors in patients with systemic lupus erythematosus, including smoking and the use of certain medications.
“As expected, older age was associated with cancer overall, as well as with the most common cancer subtypes,” wrote Sasha Bernatsky, MD, PhD, of McGill University, Montreal, and coauthors. The study was published in Arthritis Care & Research.
To determine the risk of cancer in people with clinically confirmed incident systemic lupus erythematosus (SLE), the researchers analyzed data from 1,668 newly diagnosed lupus patients with at least one follow-up visit. All patients were enrolled in the Systemic Lupus International Collaborating Clinics inception cohort from across 33 different centers in North America, Europe, and Asia. A total of 89% (n = 1,480) were women, and 49% (n = 824) were white. The average follow-up period was 9 years.
Of the 1,668 SLE patients, 65 developed some type of cancer. The cancers included 15 breast;, 10 nonmelanoma skin; 7 lung; 6 hematologic, 6 prostate; 5 melanoma; 3 cervical; 3 renal; 2 gastric; 2 head and neck; 2 thyroid; and 1 rectal, sarcoma, thymoma, or uterine. No patient had more than one type, and the mean age of the cancer patients at time of SLE diagnosis was 45.6 (standard deviation, 14.5).
Almost half of the 65 cancers occurred in past or current smokers, including all of the lung cancers, while only 33% of patients without cancers smoked prior to baseline. After univariate analysis, characteristics associated with a higher risk of all cancers included older age at SLE diagnosis (adjusted hazard ratio, 1.05; 95% confidence interval, 1.03-1.06), White race/ethnicity (aHR 1.34; 95% CI, 0.76-2.37), and smoking (aHR 1.21; 95% CI, 0.73-2.01).
After multivariate analysis, the two characteristics most associated with increased cancer risk were older age at SLE diagnosis and being male. The analyses also confirmed that older age was a risk factor for breast cancer (aHR 1.06; 95% CI, 1.02-1.10) and nonmelanoma skin cancer (aHR, 1.06; 95% CI, 1.02-1.11), while use of antimalarial drugs was associated with a lower risk of both breast (aHR, 0.28; 95% CI, 0.09-0.90) and nonmelanoma skin (aHR, 0.23; 95% CI, 0.05-0.95) cancers. For lung cancer, the highest risk factor was smoking 15 or more cigarettes a day (aHR, 6.64; 95% CI, 1.43-30.9); for hematologic cancers, it was being in the top quartile of SLE disease activity (aHR, 7.14; 95% CI, 1.13-45.3).
The authors acknowledged their study’s limitations, including the small number of cancers overall and purposefully not comparing cancer risk in SLE patients with risk in the general population. Although their methods – “physicians recording events at annual visits, confirmed by review of charts” – were recognized as very suitable for the current analysis, they noted that a broader comparison would “potentially be problematic due to differential misclassification error” in cancer registry data.
Two of the study’s authors reported potential conflicts of interest, including receiving grants and consulting and personal fees from various pharmaceutical companies. No other potential conflicts were reported.
SOURCE: Bernatsky S et al. Arthritis Care Res. 2020 Aug 19. doi: 10.1002/acr.24425.
A new study has quantified cancer risk factors in patients with systemic lupus erythematosus, including smoking and the use of certain medications.
“As expected, older age was associated with cancer overall, as well as with the most common cancer subtypes,” wrote Sasha Bernatsky, MD, PhD, of McGill University, Montreal, and coauthors. The study was published in Arthritis Care & Research.
To determine the risk of cancer in people with clinically confirmed incident systemic lupus erythematosus (SLE), the researchers analyzed data from 1,668 newly diagnosed lupus patients with at least one follow-up visit. All patients were enrolled in the Systemic Lupus International Collaborating Clinics inception cohort from across 33 different centers in North America, Europe, and Asia. A total of 89% (n = 1,480) were women, and 49% (n = 824) were white. The average follow-up period was 9 years.
Of the 1,668 SLE patients, 65 developed some type of cancer. The cancers included 15 breast;, 10 nonmelanoma skin; 7 lung; 6 hematologic, 6 prostate; 5 melanoma; 3 cervical; 3 renal; 2 gastric; 2 head and neck; 2 thyroid; and 1 rectal, sarcoma, thymoma, or uterine. No patient had more than one type, and the mean age of the cancer patients at time of SLE diagnosis was 45.6 (standard deviation, 14.5).
Almost half of the 65 cancers occurred in past or current smokers, including all of the lung cancers, while only 33% of patients without cancers smoked prior to baseline. After univariate analysis, characteristics associated with a higher risk of all cancers included older age at SLE diagnosis (adjusted hazard ratio, 1.05; 95% confidence interval, 1.03-1.06), White race/ethnicity (aHR 1.34; 95% CI, 0.76-2.37), and smoking (aHR 1.21; 95% CI, 0.73-2.01).
After multivariate analysis, the two characteristics most associated with increased cancer risk were older age at SLE diagnosis and being male. The analyses also confirmed that older age was a risk factor for breast cancer (aHR 1.06; 95% CI, 1.02-1.10) and nonmelanoma skin cancer (aHR, 1.06; 95% CI, 1.02-1.11), while use of antimalarial drugs was associated with a lower risk of both breast (aHR, 0.28; 95% CI, 0.09-0.90) and nonmelanoma skin (aHR, 0.23; 95% CI, 0.05-0.95) cancers. For lung cancer, the highest risk factor was smoking 15 or more cigarettes a day (aHR, 6.64; 95% CI, 1.43-30.9); for hematologic cancers, it was being in the top quartile of SLE disease activity (aHR, 7.14; 95% CI, 1.13-45.3).
The authors acknowledged their study’s limitations, including the small number of cancers overall and purposefully not comparing cancer risk in SLE patients with risk in the general population. Although their methods – “physicians recording events at annual visits, confirmed by review of charts” – were recognized as very suitable for the current analysis, they noted that a broader comparison would “potentially be problematic due to differential misclassification error” in cancer registry data.
Two of the study’s authors reported potential conflicts of interest, including receiving grants and consulting and personal fees from various pharmaceutical companies. No other potential conflicts were reported.
SOURCE: Bernatsky S et al. Arthritis Care Res. 2020 Aug 19. doi: 10.1002/acr.24425.
A new study has quantified cancer risk factors in patients with systemic lupus erythematosus, including smoking and the use of certain medications.
“As expected, older age was associated with cancer overall, as well as with the most common cancer subtypes,” wrote Sasha Bernatsky, MD, PhD, of McGill University, Montreal, and coauthors. The study was published in Arthritis Care & Research.
To determine the risk of cancer in people with clinically confirmed incident systemic lupus erythematosus (SLE), the researchers analyzed data from 1,668 newly diagnosed lupus patients with at least one follow-up visit. All patients were enrolled in the Systemic Lupus International Collaborating Clinics inception cohort from across 33 different centers in North America, Europe, and Asia. A total of 89% (n = 1,480) were women, and 49% (n = 824) were white. The average follow-up period was 9 years.
Of the 1,668 SLE patients, 65 developed some type of cancer. The cancers included 15 breast;, 10 nonmelanoma skin; 7 lung; 6 hematologic, 6 prostate; 5 melanoma; 3 cervical; 3 renal; 2 gastric; 2 head and neck; 2 thyroid; and 1 rectal, sarcoma, thymoma, or uterine. No patient had more than one type, and the mean age of the cancer patients at time of SLE diagnosis was 45.6 (standard deviation, 14.5).
Almost half of the 65 cancers occurred in past or current smokers, including all of the lung cancers, while only 33% of patients without cancers smoked prior to baseline. After univariate analysis, characteristics associated with a higher risk of all cancers included older age at SLE diagnosis (adjusted hazard ratio, 1.05; 95% confidence interval, 1.03-1.06), White race/ethnicity (aHR 1.34; 95% CI, 0.76-2.37), and smoking (aHR 1.21; 95% CI, 0.73-2.01).
After multivariate analysis, the two characteristics most associated with increased cancer risk were older age at SLE diagnosis and being male. The analyses also confirmed that older age was a risk factor for breast cancer (aHR 1.06; 95% CI, 1.02-1.10) and nonmelanoma skin cancer (aHR, 1.06; 95% CI, 1.02-1.11), while use of antimalarial drugs was associated with a lower risk of both breast (aHR, 0.28; 95% CI, 0.09-0.90) and nonmelanoma skin (aHR, 0.23; 95% CI, 0.05-0.95) cancers. For lung cancer, the highest risk factor was smoking 15 or more cigarettes a day (aHR, 6.64; 95% CI, 1.43-30.9); for hematologic cancers, it was being in the top quartile of SLE disease activity (aHR, 7.14; 95% CI, 1.13-45.3).
The authors acknowledged their study’s limitations, including the small number of cancers overall and purposefully not comparing cancer risk in SLE patients with risk in the general population. Although their methods – “physicians recording events at annual visits, confirmed by review of charts” – were recognized as very suitable for the current analysis, they noted that a broader comparison would “potentially be problematic due to differential misclassification error” in cancer registry data.
Two of the study’s authors reported potential conflicts of interest, including receiving grants and consulting and personal fees from various pharmaceutical companies. No other potential conflicts were reported.
SOURCE: Bernatsky S et al. Arthritis Care Res. 2020 Aug 19. doi: 10.1002/acr.24425.
FROM ARTHRITIS CARE & RESEARCH
Treatment for a tobacco-dependent adult
Applying American Thoracic Society’s new clinical practice guideline
Complications from tobacco use are the most common preventable cause of death, disability, and disease in the United States. Tobacco use causes 480,000 premature deaths every year. In pregnancy, tobacco use causes complications such as premature birth, intrauterine growth restriction, and placental abruption. In the perinatal period, it is associated with sudden infant death syndrome. While cigarette smoking is decreasing in adolescents, e-cigarette use in on the rise. Approximately 1,600 children aged 12-17 smoke their first cigarette every day and it is estimated that 5.6 million children and adolescents will die of a tobacco use–related death.1 For these reasons it is important to address tobacco use and cessation with patients whenever it is possible.
Case
A forty-five-year-old male who rarely comes to the office is here today for a physical exam at the urging of his partner. He has been smoking a pack a day since age 17. You have tried at past visits to discuss quitting, but he had been in the precontemplative stage and had been unwilling to consider any change. This visit, however, he is ready to try to quit. What can you offer him?
Core recommendations from ATS guidelines
This patient can be offered varenicline plus nicotine replacement therapy rather than nicotine replacement therapy, bupropion, e-cigarettes, or varenicline alone. His course of therapy should extend beyond 12 weeks instead of the standard 6- to 12-week therapy. Alternatively, he could be offered varenicline alone, rather than nicotine replacement.2
A change from previous guidelines
What makes this recommendation so interesting and new is the emphasis it places on varenicline. The United States Preventive Services Task Force released a recommendation statement in 2015 that stressed a combination of pharmacological and behavioral interventions. It discussed nicotine replacement therapy, bupropion, and varenicline, but did not recommend any one over any of the others.3 The new recommendation from the American Thoracic Society favors varenicline over other pharmacologic interventions. It is based on an independent systematic review of the literature that showed higher rates of tobacco use abstinence at the 6-month follow-up with varenicline alone versus nicotine replacement therapy alone, bupropion alone, or e-cigarette use only.
A review of 14 randomized controlled trials showed that varenicline improves abstinence rates during treatment by approximately 40% compared with nicotine replacement, and by 20% at the end of 6 months of treatment. The review found that varenicline plus nicotine replacement therapy is more effective than varenicline alone. In this comparison, based on three trials, there was a 36% higher abstinence rate at 6 months using varenicline plus nicotine replacement. When varenicline use was compared with use of a nicotine patch, bupropion, or e-cigarettes, there was a reduction in serious adverse events – changes in mood, suicidal ideation, and neurological side effects such as seizures.2 Clinicians may remember a black box warning on the varenicline label citing neuropsychiatric effects and it is important to note that the Food and Drug Administration removed this boxed warning in 2016.4
Opinion
This recommendation represents an important, evidence-based change from previous guidelines. It presents the opportunity for better outcomes, but will likely take a while to filter into practice, as clinicians need to become more comfortable with the use of varenicline and insurance supports the cost of varenicline.
The average cost of varenicline for 12 weeks is between $1,220 and $1,584. For comparison, nicotine replacement therapy costs $170 to $240 for the same number of weeks. To put those costs in perspective, the 12-week cost of cigarettes for a two-pack-a-day smoker is approximately $1,000.
For some patients, the motivation to quit smoking comes from the realization of how much they are spending on cigarettes each month. That said, if a patient does not have insurance or their insurance does not cover the cost of varenicline, nicotine replacement therapy might be more appealing. It should be noted that better abstinence rates have been seen in patients taking varenicline plus nicotine replacement therapy versus varenicline alone.
Suggested treatment
Based on a systematic review of randomized controlled trials, the American Thoracic Society’s guideline on pharmacological treatment in tobacco-dependent adults concludes that varenicline plus nicotine patch is the preferred pharmacological treatment for tobacco cessation when compared with varenicline alone, bupropion alone, nicotine replacement therapy alone, and e-cigarettes alone. If the patient does not want to start two medicines at once, then varenicline alone would be the preferred choice.
Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Hospital–Jefferson Health. Dr. Sprogell is a third-year resident in the family medicine residency program at Abington Jefferson Health. They have no conflicts related to the content of this piece. For questions or comments, feel free to contact Dr. Skolnik on Twitter @NeilSkolnik.
References
1. U.S. Preventive Services Task Force. Primary care interventions for prevention and cessation of tobacco use in children and adolescents: U.S. Preventive Services Task Force Recommendation Statement. JAMA.2020;323(16):1590-8. doi: 10.1001/jama.2020.4679.
2. Leone FT et al. Initiating pharmacologic treatment in tobacco-dependent adults: An official American Thoracic Society Clinical Practice Guideline. Am J Respir Crit Care Med. 2020;202(2):e5–e31.
3. Tobacco smoking cessation in adults, including pregnant women: Behavioral and pharmacotherapy interventions. U.S. Preventive Services Task Force 2015 Sep 21.
4. FDA Drug Safety Communication: FDA revises description of mental health side effects of the stop-smoking medicines Chantix (varenicline) and Zyban (bupropion) to reflect clinical trial findings. 2016 Dec. 16.
Applying American Thoracic Society’s new clinical practice guideline
Applying American Thoracic Society’s new clinical practice guideline
Complications from tobacco use are the most common preventable cause of death, disability, and disease in the United States. Tobacco use causes 480,000 premature deaths every year. In pregnancy, tobacco use causes complications such as premature birth, intrauterine growth restriction, and placental abruption. In the perinatal period, it is associated with sudden infant death syndrome. While cigarette smoking is decreasing in adolescents, e-cigarette use in on the rise. Approximately 1,600 children aged 12-17 smoke their first cigarette every day and it is estimated that 5.6 million children and adolescents will die of a tobacco use–related death.1 For these reasons it is important to address tobacco use and cessation with patients whenever it is possible.
Case
A forty-five-year-old male who rarely comes to the office is here today for a physical exam at the urging of his partner. He has been smoking a pack a day since age 17. You have tried at past visits to discuss quitting, but he had been in the precontemplative stage and had been unwilling to consider any change. This visit, however, he is ready to try to quit. What can you offer him?
Core recommendations from ATS guidelines
This patient can be offered varenicline plus nicotine replacement therapy rather than nicotine replacement therapy, bupropion, e-cigarettes, or varenicline alone. His course of therapy should extend beyond 12 weeks instead of the standard 6- to 12-week therapy. Alternatively, he could be offered varenicline alone, rather than nicotine replacement.2
A change from previous guidelines
What makes this recommendation so interesting and new is the emphasis it places on varenicline. The United States Preventive Services Task Force released a recommendation statement in 2015 that stressed a combination of pharmacological and behavioral interventions. It discussed nicotine replacement therapy, bupropion, and varenicline, but did not recommend any one over any of the others.3 The new recommendation from the American Thoracic Society favors varenicline over other pharmacologic interventions. It is based on an independent systematic review of the literature that showed higher rates of tobacco use abstinence at the 6-month follow-up with varenicline alone versus nicotine replacement therapy alone, bupropion alone, or e-cigarette use only.
A review of 14 randomized controlled trials showed that varenicline improves abstinence rates during treatment by approximately 40% compared with nicotine replacement, and by 20% at the end of 6 months of treatment. The review found that varenicline plus nicotine replacement therapy is more effective than varenicline alone. In this comparison, based on three trials, there was a 36% higher abstinence rate at 6 months using varenicline plus nicotine replacement. When varenicline use was compared with use of a nicotine patch, bupropion, or e-cigarettes, there was a reduction in serious adverse events – changes in mood, suicidal ideation, and neurological side effects such as seizures.2 Clinicians may remember a black box warning on the varenicline label citing neuropsychiatric effects and it is important to note that the Food and Drug Administration removed this boxed warning in 2016.4
Opinion
This recommendation represents an important, evidence-based change from previous guidelines. It presents the opportunity for better outcomes, but will likely take a while to filter into practice, as clinicians need to become more comfortable with the use of varenicline and insurance supports the cost of varenicline.
The average cost of varenicline for 12 weeks is between $1,220 and $1,584. For comparison, nicotine replacement therapy costs $170 to $240 for the same number of weeks. To put those costs in perspective, the 12-week cost of cigarettes for a two-pack-a-day smoker is approximately $1,000.
For some patients, the motivation to quit smoking comes from the realization of how much they are spending on cigarettes each month. That said, if a patient does not have insurance or their insurance does not cover the cost of varenicline, nicotine replacement therapy might be more appealing. It should be noted that better abstinence rates have been seen in patients taking varenicline plus nicotine replacement therapy versus varenicline alone.
Suggested treatment
Based on a systematic review of randomized controlled trials, the American Thoracic Society’s guideline on pharmacological treatment in tobacco-dependent adults concludes that varenicline plus nicotine patch is the preferred pharmacological treatment for tobacco cessation when compared with varenicline alone, bupropion alone, nicotine replacement therapy alone, and e-cigarettes alone. If the patient does not want to start two medicines at once, then varenicline alone would be the preferred choice.
Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Hospital–Jefferson Health. Dr. Sprogell is a third-year resident in the family medicine residency program at Abington Jefferson Health. They have no conflicts related to the content of this piece. For questions or comments, feel free to contact Dr. Skolnik on Twitter @NeilSkolnik.
References
1. U.S. Preventive Services Task Force. Primary care interventions for prevention and cessation of tobacco use in children and adolescents: U.S. Preventive Services Task Force Recommendation Statement. JAMA.2020;323(16):1590-8. doi: 10.1001/jama.2020.4679.
2. Leone FT et al. Initiating pharmacologic treatment in tobacco-dependent adults: An official American Thoracic Society Clinical Practice Guideline. Am J Respir Crit Care Med. 2020;202(2):e5–e31.
3. Tobacco smoking cessation in adults, including pregnant women: Behavioral and pharmacotherapy interventions. U.S. Preventive Services Task Force 2015 Sep 21.
4. FDA Drug Safety Communication: FDA revises description of mental health side effects of the stop-smoking medicines Chantix (varenicline) and Zyban (bupropion) to reflect clinical trial findings. 2016 Dec. 16.
Complications from tobacco use are the most common preventable cause of death, disability, and disease in the United States. Tobacco use causes 480,000 premature deaths every year. In pregnancy, tobacco use causes complications such as premature birth, intrauterine growth restriction, and placental abruption. In the perinatal period, it is associated with sudden infant death syndrome. While cigarette smoking is decreasing in adolescents, e-cigarette use in on the rise. Approximately 1,600 children aged 12-17 smoke their first cigarette every day and it is estimated that 5.6 million children and adolescents will die of a tobacco use–related death.1 For these reasons it is important to address tobacco use and cessation with patients whenever it is possible.
Case
A forty-five-year-old male who rarely comes to the office is here today for a physical exam at the urging of his partner. He has been smoking a pack a day since age 17. You have tried at past visits to discuss quitting, but he had been in the precontemplative stage and had been unwilling to consider any change. This visit, however, he is ready to try to quit. What can you offer him?
Core recommendations from ATS guidelines
This patient can be offered varenicline plus nicotine replacement therapy rather than nicotine replacement therapy, bupropion, e-cigarettes, or varenicline alone. His course of therapy should extend beyond 12 weeks instead of the standard 6- to 12-week therapy. Alternatively, he could be offered varenicline alone, rather than nicotine replacement.2
A change from previous guidelines
What makes this recommendation so interesting and new is the emphasis it places on varenicline. The United States Preventive Services Task Force released a recommendation statement in 2015 that stressed a combination of pharmacological and behavioral interventions. It discussed nicotine replacement therapy, bupropion, and varenicline, but did not recommend any one over any of the others.3 The new recommendation from the American Thoracic Society favors varenicline over other pharmacologic interventions. It is based on an independent systematic review of the literature that showed higher rates of tobacco use abstinence at the 6-month follow-up with varenicline alone versus nicotine replacement therapy alone, bupropion alone, or e-cigarette use only.
A review of 14 randomized controlled trials showed that varenicline improves abstinence rates during treatment by approximately 40% compared with nicotine replacement, and by 20% at the end of 6 months of treatment. The review found that varenicline plus nicotine replacement therapy is more effective than varenicline alone. In this comparison, based on three trials, there was a 36% higher abstinence rate at 6 months using varenicline plus nicotine replacement. When varenicline use was compared with use of a nicotine patch, bupropion, or e-cigarettes, there was a reduction in serious adverse events – changes in mood, suicidal ideation, and neurological side effects such as seizures.2 Clinicians may remember a black box warning on the varenicline label citing neuropsychiatric effects and it is important to note that the Food and Drug Administration removed this boxed warning in 2016.4
Opinion
This recommendation represents an important, evidence-based change from previous guidelines. It presents the opportunity for better outcomes, but will likely take a while to filter into practice, as clinicians need to become more comfortable with the use of varenicline and insurance supports the cost of varenicline.
The average cost of varenicline for 12 weeks is between $1,220 and $1,584. For comparison, nicotine replacement therapy costs $170 to $240 for the same number of weeks. To put those costs in perspective, the 12-week cost of cigarettes for a two-pack-a-day smoker is approximately $1,000.
For some patients, the motivation to quit smoking comes from the realization of how much they are spending on cigarettes each month. That said, if a patient does not have insurance or their insurance does not cover the cost of varenicline, nicotine replacement therapy might be more appealing. It should be noted that better abstinence rates have been seen in patients taking varenicline plus nicotine replacement therapy versus varenicline alone.
Suggested treatment
Based on a systematic review of randomized controlled trials, the American Thoracic Society’s guideline on pharmacological treatment in tobacco-dependent adults concludes that varenicline plus nicotine patch is the preferred pharmacological treatment for tobacco cessation when compared with varenicline alone, bupropion alone, nicotine replacement therapy alone, and e-cigarettes alone. If the patient does not want to start two medicines at once, then varenicline alone would be the preferred choice.
Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Hospital–Jefferson Health. Dr. Sprogell is a third-year resident in the family medicine residency program at Abington Jefferson Health. They have no conflicts related to the content of this piece. For questions or comments, feel free to contact Dr. Skolnik on Twitter @NeilSkolnik.
References
1. U.S. Preventive Services Task Force. Primary care interventions for prevention and cessation of tobacco use in children and adolescents: U.S. Preventive Services Task Force Recommendation Statement. JAMA.2020;323(16):1590-8. doi: 10.1001/jama.2020.4679.
2. Leone FT et al. Initiating pharmacologic treatment in tobacco-dependent adults: An official American Thoracic Society Clinical Practice Guideline. Am J Respir Crit Care Med. 2020;202(2):e5–e31.
3. Tobacco smoking cessation in adults, including pregnant women: Behavioral and pharmacotherapy interventions. U.S. Preventive Services Task Force 2015 Sep 21.
4. FDA Drug Safety Communication: FDA revises description of mental health side effects of the stop-smoking medicines Chantix (varenicline) and Zyban (bupropion) to reflect clinical trial findings. 2016 Dec. 16.
Performance status, molecular testing key to metastatic cancer prognosis
according to Sam Brondfield, MD, MA, an inpatient medical oncologist at the University of California, San Francisco.

Oncologists have at their fingertips a voluminous and ever-growing body of clinical trials data to draw on for prognostication. Yet many hospitalists will be surprised to learn that this wealth of information is of little value in the inpatient settings where they work, he said at HM20 Virtual, hosted by the Society of Hospital Medicine.
“The applicability of clinical trials data to hospitalized patients is generally poor. That’s an important caveat to keep in mind,” Dr. Brondfield said.
Enrollment in clinical trials is usually restricted to patients with a score of 0 or 1 on the Eastern Clinical Oncology Group Performance Status, meaning their cancer is causing minimal or no disruption to their life (see graphic). Sometimes trials will include patients with a performance status of 2 on the ECOG scale, a tool developed nearly 40 years ago, but clinical trials virtually never enroll those with an ECOG status of 3 or 4. Yet most hospitalized patients with metastatic cancer have an ECOG performance status of 3 or worse. Thus, the clinical trials outcome data are of little relevance.
“In oncology the distinction between ECOG 2 and 3 is very important,” Dr. Brondfield emphasized.
When he talks about treatment options with hospitalized patients who have metastatic cancer and poor performance status – that is, ECOG 3 or 4 – he’ll often say: “Assuming you feel better and can go home, that’s when these clinical trial data may apply better to you.”
Dr. Brondfield cautioned against quoting the National Cancer Institute’s Surveillance, Epidemiology and End Results (SEER) 5-year overall survival data when hospitalized patients with advanced cancer ask how long they have to live. For one thing, the national average 5-year overall survival figure is hardly an individualized assessment. Plus, oncology is a fast-moving field in which important treatment advances occur all the time, and the SEER data lag far behind. For example, when Dr. Brondfield recently looked up the current SEER 5-year survival for patients diagnosed with metastatic non–small cell lung cancer (NSCLC), the figure quoted was less than 6%, and it was drawn from data accrued in 2009-2015. That simply doesn’t reflect contemporary practice.
Indeed, it’s no longer true that the average survival of patients with metastatic NSCLC is less than a year. In the practice-changing KEYNOTE-189 randomized trial, which accrued participants in 2016-2017, the median overall survival of patients randomized to pembrolizumab (Keytruda) plus standard cytotoxic chemotherapy was 22 months, compared with 11 months with chemotherapy plus placebo (J Clin Oncol. 2020 May 10. doi: 10.1200/JCO.19.03136). As a result, immunotherapy with a programmed death–1 inhibitor such as pembrolizumab in combination with chemotherapy is now standard practice in patients with metastatic NSCLC without targetable mutations.
Performance status guides treatment decision-making
Hospitalists can help oncologists in decision-making regarding whether to offer palliative systemic therapy to patients with advanced metastatic cancer and poor performance status by determining whether that status is caused by the cancer itself or some other cause that’s not easily reversible, such as liver failure.
Take, for example, the inpatient with advanced SCLC. This is an aggressive and chemosensitive cancer. Dr. Brondfield said he is among many medical oncologists who are convinced that, if poor performance status in a patient with advanced SCLC is caused by the cancer itself, prompt initiation of inpatient chemotherapy should be recommended to elicit a response that improves quality of life and performance status in the short term. If, on the other hand, the poor performance status is caused by organ failure or some other issue that can’t easily be improved, hospice may be more appropriate.
“The contour of SCLC over time is that despite its treatment responsiveness it inevitably recurs. But with chemotherapy you can give people in this situation months of quality time, so we generally try to treat these sorts of patients,” Dr. Brondfield explained.
The National Comprehensive Cancer Network guidelines upon which oncologists rely leave lots of room for interpretation regarding the appropriateness of inpatient chemotherapy in patients with advanced cancer and poor patient performance status. Citing “knowledge that’s been passed down across oncology generations,” Dr. Brondfield said he and many of his colleagues believe early palliative supportive care rather than systemic cytotoxic cancer-directed therapy is appropriate for patients with poor performance status who have one of several specific relatively nonchemoresponsive types of metastatic cancer. These include esophageal, gastric, and head and neck cancers.
On the other hand, advanced SCLC isn’t the only type of metastatic cancer that’s so chemosensitive that he and many other oncologists believe aggressive chemotherapy should be offered even in the face of poor patient performance status attributable to the cancer itself.
Take, for example, colorectal cancer with no more than five metastases to the lung or liver, provided those metastases are treatable with resection or radiation. “Those patients are actually curable at a high rate. They have about a 30%-40% cure rate. So those patients, even if they have poor performance status, if we can get them up for surgery or radiation, we usually do try to treat them aggressively,” Dr. Brondfield said.
There are other often chemoresponsive metastatic cancers for which oncologists frequently recommend aggressive treatment to improve quality of life in patients with poor performance status. These cancers include aggressive lymphomas, which are actually often curable; multiple myeloma; testicular and germ cell cancers; NSCLC with a targetable mutation, which is often responsive to oral medications; and prostate and well-differentiated thyroid cancers, which can usually be treated with hormone- or iodine-based therapies rather than more toxic intravenous cytotoxic chemotherapy.
The impact of inpatient palliative chemotherapy in patients with poor performance status and advanced solid cancers not on the short list of highly chemosensitive cancers has not been well studied. A recent retrospective study of 228 such patients who received inpatient palliative chemotherapy at a large Brazilian academic medical center provided little reason for enthusiasm regarding the practice. Survival was short, with 30- and 60-day survival rates of 56% and 39%, respectively. Plus, 30% of patients were admitted to the ICU, where they received aggressive and costly end-of-life care. The investigators found these results suggestive of overprescribing of inpatient palliative chemotherapy (BMC Palliat Care. 2019 May 20;18[1]:42. doi: 10.1186/s12904-019-0427-4).
Of note, the investigators found in a multivariate analysis that an elevated bilirubin was associated with a 217% increased risk of 30-day mortality, and hypercalcemia was associated with a 119% increased risk.
“That’s something to take into account when these decisions are being made,” Dr. Brondfield advised.
In response to an audience comment that oncologists often seem overly optimistic about prognosis, Dr. Brondfield observed, “I think it’s very common for there to be a disagreement between the oncologist wanting to be aggressive for a sick inpatient and the hospitalist or generalist provider thinking: ‘This person looks way too sick for chemotherapy.’ ”
For this reason he is a firm believer in having multidisciplinary conversations regarding prognosis in challenging situations involving hospitalized patients with advanced cancer. An oncologist can bring to such discussions a detailed understanding of clinical trial and molecular data as well as information about the patient’s response to the first round of therapy. But lots of other factors are relevant to prognosis, including nutritional status, comorbidities, and the intuitive eyeball test of how a patient might do. The patient’s family, primary care provider, oncologist, the hospitalist, and the palliative care team will have perspectives of their own.
Molecular testing is now the norm in metastatic cancers
These days oncologists order molecular testing for most patients with metastatic carcinomas to determine eligibility for targeted therapy, suitability for participation in clinical trials, prognostication, and/or assistance in determining the site of origin if that’s unclear.
A single-pass fine needle aspiration biopsy doesn’t provide enough tissue for molecular testing. It’s therefore important to order initially a multipass fine needle aspiration to avoid the need for a repeat biopsy, which is uncomfortable for the patient and can delay diagnosis and treatment.
Dr. Brondfield advised waiting for molecular testing results to come in before trying to prognosticate in patients with a metastatic cancer for which targetable mutations might be present. Survival rates can vary substantially depending upon those test results. Take, for example, metastatic NSCLC: Just within the past year, clinical trials have been published reporting overall survival rates of 39 months in patients with treatable mutations in epidermal growth factor receptor, 42 months with anaplastic lymphoma kinase mutations, and 51 months in patients whose tumor signature features mutations in c-ros oncogene 1, as compared with 22 months with no targetable mutations in the KEYNOTE-189 trial.
“There’s a lot of heterogeneity around how metastatic tumors behave and respond to therapy. Not all metastatic cancers are the same,” the oncologist emphasized.
according to Sam Brondfield, MD, MA, an inpatient medical oncologist at the University of California, San Francisco.

Oncologists have at their fingertips a voluminous and ever-growing body of clinical trials data to draw on for prognostication. Yet many hospitalists will be surprised to learn that this wealth of information is of little value in the inpatient settings where they work, he said at HM20 Virtual, hosted by the Society of Hospital Medicine.
“The applicability of clinical trials data to hospitalized patients is generally poor. That’s an important caveat to keep in mind,” Dr. Brondfield said.
Enrollment in clinical trials is usually restricted to patients with a score of 0 or 1 on the Eastern Clinical Oncology Group Performance Status, meaning their cancer is causing minimal or no disruption to their life (see graphic). Sometimes trials will include patients with a performance status of 2 on the ECOG scale, a tool developed nearly 40 years ago, but clinical trials virtually never enroll those with an ECOG status of 3 or 4. Yet most hospitalized patients with metastatic cancer have an ECOG performance status of 3 or worse. Thus, the clinical trials outcome data are of little relevance.
“In oncology the distinction between ECOG 2 and 3 is very important,” Dr. Brondfield emphasized.
When he talks about treatment options with hospitalized patients who have metastatic cancer and poor performance status – that is, ECOG 3 or 4 – he’ll often say: “Assuming you feel better and can go home, that’s when these clinical trial data may apply better to you.”
Dr. Brondfield cautioned against quoting the National Cancer Institute’s Surveillance, Epidemiology and End Results (SEER) 5-year overall survival data when hospitalized patients with advanced cancer ask how long they have to live. For one thing, the national average 5-year overall survival figure is hardly an individualized assessment. Plus, oncology is a fast-moving field in which important treatment advances occur all the time, and the SEER data lag far behind. For example, when Dr. Brondfield recently looked up the current SEER 5-year survival for patients diagnosed with metastatic non–small cell lung cancer (NSCLC), the figure quoted was less than 6%, and it was drawn from data accrued in 2009-2015. That simply doesn’t reflect contemporary practice.
Indeed, it’s no longer true that the average survival of patients with metastatic NSCLC is less than a year. In the practice-changing KEYNOTE-189 randomized trial, which accrued participants in 2016-2017, the median overall survival of patients randomized to pembrolizumab (Keytruda) plus standard cytotoxic chemotherapy was 22 months, compared with 11 months with chemotherapy plus placebo (J Clin Oncol. 2020 May 10. doi: 10.1200/JCO.19.03136). As a result, immunotherapy with a programmed death–1 inhibitor such as pembrolizumab in combination with chemotherapy is now standard practice in patients with metastatic NSCLC without targetable mutations.
Performance status guides treatment decision-making
Hospitalists can help oncologists in decision-making regarding whether to offer palliative systemic therapy to patients with advanced metastatic cancer and poor performance status by determining whether that status is caused by the cancer itself or some other cause that’s not easily reversible, such as liver failure.
Take, for example, the inpatient with advanced SCLC. This is an aggressive and chemosensitive cancer. Dr. Brondfield said he is among many medical oncologists who are convinced that, if poor performance status in a patient with advanced SCLC is caused by the cancer itself, prompt initiation of inpatient chemotherapy should be recommended to elicit a response that improves quality of life and performance status in the short term. If, on the other hand, the poor performance status is caused by organ failure or some other issue that can’t easily be improved, hospice may be more appropriate.
“The contour of SCLC over time is that despite its treatment responsiveness it inevitably recurs. But with chemotherapy you can give people in this situation months of quality time, so we generally try to treat these sorts of patients,” Dr. Brondfield explained.
The National Comprehensive Cancer Network guidelines upon which oncologists rely leave lots of room for interpretation regarding the appropriateness of inpatient chemotherapy in patients with advanced cancer and poor patient performance status. Citing “knowledge that’s been passed down across oncology generations,” Dr. Brondfield said he and many of his colleagues believe early palliative supportive care rather than systemic cytotoxic cancer-directed therapy is appropriate for patients with poor performance status who have one of several specific relatively nonchemoresponsive types of metastatic cancer. These include esophageal, gastric, and head and neck cancers.
On the other hand, advanced SCLC isn’t the only type of metastatic cancer that’s so chemosensitive that he and many other oncologists believe aggressive chemotherapy should be offered even in the face of poor patient performance status attributable to the cancer itself.
Take, for example, colorectal cancer with no more than five metastases to the lung or liver, provided those metastases are treatable with resection or radiation. “Those patients are actually curable at a high rate. They have about a 30%-40% cure rate. So those patients, even if they have poor performance status, if we can get them up for surgery or radiation, we usually do try to treat them aggressively,” Dr. Brondfield said.
There are other often chemoresponsive metastatic cancers for which oncologists frequently recommend aggressive treatment to improve quality of life in patients with poor performance status. These cancers include aggressive lymphomas, which are actually often curable; multiple myeloma; testicular and germ cell cancers; NSCLC with a targetable mutation, which is often responsive to oral medications; and prostate and well-differentiated thyroid cancers, which can usually be treated with hormone- or iodine-based therapies rather than more toxic intravenous cytotoxic chemotherapy.
The impact of inpatient palliative chemotherapy in patients with poor performance status and advanced solid cancers not on the short list of highly chemosensitive cancers has not been well studied. A recent retrospective study of 228 such patients who received inpatient palliative chemotherapy at a large Brazilian academic medical center provided little reason for enthusiasm regarding the practice. Survival was short, with 30- and 60-day survival rates of 56% and 39%, respectively. Plus, 30% of patients were admitted to the ICU, where they received aggressive and costly end-of-life care. The investigators found these results suggestive of overprescribing of inpatient palliative chemotherapy (BMC Palliat Care. 2019 May 20;18[1]:42. doi: 10.1186/s12904-019-0427-4).
Of note, the investigators found in a multivariate analysis that an elevated bilirubin was associated with a 217% increased risk of 30-day mortality, and hypercalcemia was associated with a 119% increased risk.
“That’s something to take into account when these decisions are being made,” Dr. Brondfield advised.
In response to an audience comment that oncologists often seem overly optimistic about prognosis, Dr. Brondfield observed, “I think it’s very common for there to be a disagreement between the oncologist wanting to be aggressive for a sick inpatient and the hospitalist or generalist provider thinking: ‘This person looks way too sick for chemotherapy.’ ”
For this reason he is a firm believer in having multidisciplinary conversations regarding prognosis in challenging situations involving hospitalized patients with advanced cancer. An oncologist can bring to such discussions a detailed understanding of clinical trial and molecular data as well as information about the patient’s response to the first round of therapy. But lots of other factors are relevant to prognosis, including nutritional status, comorbidities, and the intuitive eyeball test of how a patient might do. The patient’s family, primary care provider, oncologist, the hospitalist, and the palliative care team will have perspectives of their own.
Molecular testing is now the norm in metastatic cancers
These days oncologists order molecular testing for most patients with metastatic carcinomas to determine eligibility for targeted therapy, suitability for participation in clinical trials, prognostication, and/or assistance in determining the site of origin if that’s unclear.
A single-pass fine needle aspiration biopsy doesn’t provide enough tissue for molecular testing. It’s therefore important to order initially a multipass fine needle aspiration to avoid the need for a repeat biopsy, which is uncomfortable for the patient and can delay diagnosis and treatment.
Dr. Brondfield advised waiting for molecular testing results to come in before trying to prognosticate in patients with a metastatic cancer for which targetable mutations might be present. Survival rates can vary substantially depending upon those test results. Take, for example, metastatic NSCLC: Just within the past year, clinical trials have been published reporting overall survival rates of 39 months in patients with treatable mutations in epidermal growth factor receptor, 42 months with anaplastic lymphoma kinase mutations, and 51 months in patients whose tumor signature features mutations in c-ros oncogene 1, as compared with 22 months with no targetable mutations in the KEYNOTE-189 trial.
“There’s a lot of heterogeneity around how metastatic tumors behave and respond to therapy. Not all metastatic cancers are the same,” the oncologist emphasized.
according to Sam Brondfield, MD, MA, an inpatient medical oncologist at the University of California, San Francisco.

Oncologists have at their fingertips a voluminous and ever-growing body of clinical trials data to draw on for prognostication. Yet many hospitalists will be surprised to learn that this wealth of information is of little value in the inpatient settings where they work, he said at HM20 Virtual, hosted by the Society of Hospital Medicine.
“The applicability of clinical trials data to hospitalized patients is generally poor. That’s an important caveat to keep in mind,” Dr. Brondfield said.
Enrollment in clinical trials is usually restricted to patients with a score of 0 or 1 on the Eastern Clinical Oncology Group Performance Status, meaning their cancer is causing minimal or no disruption to their life (see graphic). Sometimes trials will include patients with a performance status of 2 on the ECOG scale, a tool developed nearly 40 years ago, but clinical trials virtually never enroll those with an ECOG status of 3 or 4. Yet most hospitalized patients with metastatic cancer have an ECOG performance status of 3 or worse. Thus, the clinical trials outcome data are of little relevance.
“In oncology the distinction between ECOG 2 and 3 is very important,” Dr. Brondfield emphasized.
When he talks about treatment options with hospitalized patients who have metastatic cancer and poor performance status – that is, ECOG 3 or 4 – he’ll often say: “Assuming you feel better and can go home, that’s when these clinical trial data may apply better to you.”
Dr. Brondfield cautioned against quoting the National Cancer Institute’s Surveillance, Epidemiology and End Results (SEER) 5-year overall survival data when hospitalized patients with advanced cancer ask how long they have to live. For one thing, the national average 5-year overall survival figure is hardly an individualized assessment. Plus, oncology is a fast-moving field in which important treatment advances occur all the time, and the SEER data lag far behind. For example, when Dr. Brondfield recently looked up the current SEER 5-year survival for patients diagnosed with metastatic non–small cell lung cancer (NSCLC), the figure quoted was less than 6%, and it was drawn from data accrued in 2009-2015. That simply doesn’t reflect contemporary practice.
Indeed, it’s no longer true that the average survival of patients with metastatic NSCLC is less than a year. In the practice-changing KEYNOTE-189 randomized trial, which accrued participants in 2016-2017, the median overall survival of patients randomized to pembrolizumab (Keytruda) plus standard cytotoxic chemotherapy was 22 months, compared with 11 months with chemotherapy plus placebo (J Clin Oncol. 2020 May 10. doi: 10.1200/JCO.19.03136). As a result, immunotherapy with a programmed death–1 inhibitor such as pembrolizumab in combination with chemotherapy is now standard practice in patients with metastatic NSCLC without targetable mutations.
Performance status guides treatment decision-making
Hospitalists can help oncologists in decision-making regarding whether to offer palliative systemic therapy to patients with advanced metastatic cancer and poor performance status by determining whether that status is caused by the cancer itself or some other cause that’s not easily reversible, such as liver failure.
Take, for example, the inpatient with advanced SCLC. This is an aggressive and chemosensitive cancer. Dr. Brondfield said he is among many medical oncologists who are convinced that, if poor performance status in a patient with advanced SCLC is caused by the cancer itself, prompt initiation of inpatient chemotherapy should be recommended to elicit a response that improves quality of life and performance status in the short term. If, on the other hand, the poor performance status is caused by organ failure or some other issue that can’t easily be improved, hospice may be more appropriate.
“The contour of SCLC over time is that despite its treatment responsiveness it inevitably recurs. But with chemotherapy you can give people in this situation months of quality time, so we generally try to treat these sorts of patients,” Dr. Brondfield explained.
The National Comprehensive Cancer Network guidelines upon which oncologists rely leave lots of room for interpretation regarding the appropriateness of inpatient chemotherapy in patients with advanced cancer and poor patient performance status. Citing “knowledge that’s been passed down across oncology generations,” Dr. Brondfield said he and many of his colleagues believe early palliative supportive care rather than systemic cytotoxic cancer-directed therapy is appropriate for patients with poor performance status who have one of several specific relatively nonchemoresponsive types of metastatic cancer. These include esophageal, gastric, and head and neck cancers.
On the other hand, advanced SCLC isn’t the only type of metastatic cancer that’s so chemosensitive that he and many other oncologists believe aggressive chemotherapy should be offered even in the face of poor patient performance status attributable to the cancer itself.
Take, for example, colorectal cancer with no more than five metastases to the lung or liver, provided those metastases are treatable with resection or radiation. “Those patients are actually curable at a high rate. They have about a 30%-40% cure rate. So those patients, even if they have poor performance status, if we can get them up for surgery or radiation, we usually do try to treat them aggressively,” Dr. Brondfield said.
There are other often chemoresponsive metastatic cancers for which oncologists frequently recommend aggressive treatment to improve quality of life in patients with poor performance status. These cancers include aggressive lymphomas, which are actually often curable; multiple myeloma; testicular and germ cell cancers; NSCLC with a targetable mutation, which is often responsive to oral medications; and prostate and well-differentiated thyroid cancers, which can usually be treated with hormone- or iodine-based therapies rather than more toxic intravenous cytotoxic chemotherapy.
The impact of inpatient palliative chemotherapy in patients with poor performance status and advanced solid cancers not on the short list of highly chemosensitive cancers has not been well studied. A recent retrospective study of 228 such patients who received inpatient palliative chemotherapy at a large Brazilian academic medical center provided little reason for enthusiasm regarding the practice. Survival was short, with 30- and 60-day survival rates of 56% and 39%, respectively. Plus, 30% of patients were admitted to the ICU, where they received aggressive and costly end-of-life care. The investigators found these results suggestive of overprescribing of inpatient palliative chemotherapy (BMC Palliat Care. 2019 May 20;18[1]:42. doi: 10.1186/s12904-019-0427-4).
Of note, the investigators found in a multivariate analysis that an elevated bilirubin was associated with a 217% increased risk of 30-day mortality, and hypercalcemia was associated with a 119% increased risk.
“That’s something to take into account when these decisions are being made,” Dr. Brondfield advised.
In response to an audience comment that oncologists often seem overly optimistic about prognosis, Dr. Brondfield observed, “I think it’s very common for there to be a disagreement between the oncologist wanting to be aggressive for a sick inpatient and the hospitalist or generalist provider thinking: ‘This person looks way too sick for chemotherapy.’ ”
For this reason he is a firm believer in having multidisciplinary conversations regarding prognosis in challenging situations involving hospitalized patients with advanced cancer. An oncologist can bring to such discussions a detailed understanding of clinical trial and molecular data as well as information about the patient’s response to the first round of therapy. But lots of other factors are relevant to prognosis, including nutritional status, comorbidities, and the intuitive eyeball test of how a patient might do. The patient’s family, primary care provider, oncologist, the hospitalist, and the palliative care team will have perspectives of their own.
Molecular testing is now the norm in metastatic cancers
These days oncologists order molecular testing for most patients with metastatic carcinomas to determine eligibility for targeted therapy, suitability for participation in clinical trials, prognostication, and/or assistance in determining the site of origin if that’s unclear.
A single-pass fine needle aspiration biopsy doesn’t provide enough tissue for molecular testing. It’s therefore important to order initially a multipass fine needle aspiration to avoid the need for a repeat biopsy, which is uncomfortable for the patient and can delay diagnosis and treatment.
Dr. Brondfield advised waiting for molecular testing results to come in before trying to prognosticate in patients with a metastatic cancer for which targetable mutations might be present. Survival rates can vary substantially depending upon those test results. Take, for example, metastatic NSCLC: Just within the past year, clinical trials have been published reporting overall survival rates of 39 months in patients with treatable mutations in epidermal growth factor receptor, 42 months with anaplastic lymphoma kinase mutations, and 51 months in patients whose tumor signature features mutations in c-ros oncogene 1, as compared with 22 months with no targetable mutations in the KEYNOTE-189 trial.
“There’s a lot of heterogeneity around how metastatic tumors behave and respond to therapy. Not all metastatic cancers are the same,” the oncologist emphasized.
FROM HM20 VIRTUAL
Incidence, prognosis of second lung cancers support long-term surveillance
Second lung cancers occurring up to a decade after the first are on the rise, but their prognosis is similar – especially when detected early – which supports long-term surveillance in survivors, finds a large population-based study.
Although guidelines recommend continued annual low-dose CT scan surveillance extending beyond 4 years for this population based on expert consensus, long-term evidence of benefit is lacking.
Investigators led by John M. Varlotto, MD, a radiation oncologist at the University of Massachusetts Medical Center, Worcester, analyzed Surveillance, Epidemiology & End Results (SEER) data for more than 58,000 patients with first and sometimes second non–small cell lung cancers initially treated by surgical resection.
Study results reported in Lung Cancer showed that the age-adjusted incidence of second lung cancers occurring 4-10 years after the first lung cancer rose sharply during the 1985-2014 study period, driven by a large uptick in women patients.
Among all patients, second lung cancers had similar overall survival as first lung cancers, but poorer lung cancer–specific survival. However, among the subset of patients having early-stage resectable disease (tumors measuring less than 4 cm with negative nodes), both outcomes were statistically indistinguishable.
“Because our investigation noted that the overall survival of patients undergoing a second lung cancer operation was similar to those patients undergoing a first operation, and because there is a rising rate of second lung cancer in lung cancer survivors, we feel that continued surveillance beyond the 4-year interval as recommended by the American Association for Thoracic Surgery as well as the [National Comprehensive Cancer Network] guidelines would be beneficial to long-term survivors of early-stage lung cancer,” Dr. Varlotto and coinvestigators wrote.
“The recent results from recent lung cancer screening studies demonstrate that females may benefit preferentially from screening … and our study suggests that these preferential benefits of increased CT scan surveillance may extend to females who are long-term survivors of lung cancer as well,” they added.
Findings in context
“As this is an observational study, it is challenging to understand what is driving the rise in prevalence of second lung cancers,” Mara Antonoff, MD, of The University of Texas MD Anderson Cancer Center in Houston commented in an interview.
“Overall, the findings are very important, as they suggest that we should continue to perform surveillance imaging for patients beyond recommended guidelines, which may allow us to achieve better survival outcomes for those individuals who develop a second lung cancer years after the first lung cancer,” she agreed.
“Just as lung cancer screening is important to identifying lung cancers at an earlier stage when they are more easily treatable and more likely to be cured, surveillance after an initial treatment for lung cancer would allow a diagnosis of second lung cancers at an earlier stage, so the patients can again achieve durable cure,” Dr. Antonoff concluded.
Study details
For the study, Dr. Varlotto and coinvestigators used data from SEER-13 and SEER-18 to identify patients with a lung cancer diagnosis during 1998-2013, and data from SEER-9, covering the years 1985-2014, to calculate rates of second cancers occurring 4-10 years after a first lung cancer.
Analyses were based on 58,758 patients with a surgically resected first primary lung cancer (55.9% with early-stage disease) and 384 patients with a surgically resected second primary lung cancer (77.6% with early-stage disease). Median follow-up was 76 months for the former and 46 months for the latter.
Results showed that in the 4-10 years after a first lung cancer diagnosis, the age-adjusted incidence of second lung cancers rose by study year but remained less than that of all other second cancers combined until the mid-2000s. Among women, incidence started rising sharply in 2001 and significantly exceeded that of all other second cancers starting in 2005.
In the entire population of study patients, propensity-adjusted analyses showed that second lung cancers were similar to first lung cancers on overall survival (P = .1726) but had worse lung cancer–specific survival (P = .0143). However, in the subset of patients with early-stage resectable disease, second and first lung cancers were similar on both overall survival (P = .3872) and lung cancer–specific survival (P = .1276).
Dr. Varlotto disclosed that he had no conflicts of interest. The study was funded by the Department of Radiation Oncology, University of Massachusetts. Dr. Antonoff disclosed that she had no relevant conflicts of interest.
SOURCE: Varlotto JM et al. Lung Cancer. 2020;147:115-122.
Second lung cancers occurring up to a decade after the first are on the rise, but their prognosis is similar – especially when detected early – which supports long-term surveillance in survivors, finds a large population-based study.
Although guidelines recommend continued annual low-dose CT scan surveillance extending beyond 4 years for this population based on expert consensus, long-term evidence of benefit is lacking.
Investigators led by John M. Varlotto, MD, a radiation oncologist at the University of Massachusetts Medical Center, Worcester, analyzed Surveillance, Epidemiology & End Results (SEER) data for more than 58,000 patients with first and sometimes second non–small cell lung cancers initially treated by surgical resection.
Study results reported in Lung Cancer showed that the age-adjusted incidence of second lung cancers occurring 4-10 years after the first lung cancer rose sharply during the 1985-2014 study period, driven by a large uptick in women patients.
Among all patients, second lung cancers had similar overall survival as first lung cancers, but poorer lung cancer–specific survival. However, among the subset of patients having early-stage resectable disease (tumors measuring less than 4 cm with negative nodes), both outcomes were statistically indistinguishable.
“Because our investigation noted that the overall survival of patients undergoing a second lung cancer operation was similar to those patients undergoing a first operation, and because there is a rising rate of second lung cancer in lung cancer survivors, we feel that continued surveillance beyond the 4-year interval as recommended by the American Association for Thoracic Surgery as well as the [National Comprehensive Cancer Network] guidelines would be beneficial to long-term survivors of early-stage lung cancer,” Dr. Varlotto and coinvestigators wrote.
“The recent results from recent lung cancer screening studies demonstrate that females may benefit preferentially from screening … and our study suggests that these preferential benefits of increased CT scan surveillance may extend to females who are long-term survivors of lung cancer as well,” they added.
Findings in context
“As this is an observational study, it is challenging to understand what is driving the rise in prevalence of second lung cancers,” Mara Antonoff, MD, of The University of Texas MD Anderson Cancer Center in Houston commented in an interview.
“Overall, the findings are very important, as they suggest that we should continue to perform surveillance imaging for patients beyond recommended guidelines, which may allow us to achieve better survival outcomes for those individuals who develop a second lung cancer years after the first lung cancer,” she agreed.
“Just as lung cancer screening is important to identifying lung cancers at an earlier stage when they are more easily treatable and more likely to be cured, surveillance after an initial treatment for lung cancer would allow a diagnosis of second lung cancers at an earlier stage, so the patients can again achieve durable cure,” Dr. Antonoff concluded.
Study details
For the study, Dr. Varlotto and coinvestigators used data from SEER-13 and SEER-18 to identify patients with a lung cancer diagnosis during 1998-2013, and data from SEER-9, covering the years 1985-2014, to calculate rates of second cancers occurring 4-10 years after a first lung cancer.
Analyses were based on 58,758 patients with a surgically resected first primary lung cancer (55.9% with early-stage disease) and 384 patients with a surgically resected second primary lung cancer (77.6% with early-stage disease). Median follow-up was 76 months for the former and 46 months for the latter.
Results showed that in the 4-10 years after a first lung cancer diagnosis, the age-adjusted incidence of second lung cancers rose by study year but remained less than that of all other second cancers combined until the mid-2000s. Among women, incidence started rising sharply in 2001 and significantly exceeded that of all other second cancers starting in 2005.
In the entire population of study patients, propensity-adjusted analyses showed that second lung cancers were similar to first lung cancers on overall survival (P = .1726) but had worse lung cancer–specific survival (P = .0143). However, in the subset of patients with early-stage resectable disease, second and first lung cancers were similar on both overall survival (P = .3872) and lung cancer–specific survival (P = .1276).
Dr. Varlotto disclosed that he had no conflicts of interest. The study was funded by the Department of Radiation Oncology, University of Massachusetts. Dr. Antonoff disclosed that she had no relevant conflicts of interest.
SOURCE: Varlotto JM et al. Lung Cancer. 2020;147:115-122.
Second lung cancers occurring up to a decade after the first are on the rise, but their prognosis is similar – especially when detected early – which supports long-term surveillance in survivors, finds a large population-based study.
Although guidelines recommend continued annual low-dose CT scan surveillance extending beyond 4 years for this population based on expert consensus, long-term evidence of benefit is lacking.
Investigators led by John M. Varlotto, MD, a radiation oncologist at the University of Massachusetts Medical Center, Worcester, analyzed Surveillance, Epidemiology & End Results (SEER) data for more than 58,000 patients with first and sometimes second non–small cell lung cancers initially treated by surgical resection.
Study results reported in Lung Cancer showed that the age-adjusted incidence of second lung cancers occurring 4-10 years after the first lung cancer rose sharply during the 1985-2014 study period, driven by a large uptick in women patients.
Among all patients, second lung cancers had similar overall survival as first lung cancers, but poorer lung cancer–specific survival. However, among the subset of patients having early-stage resectable disease (tumors measuring less than 4 cm with negative nodes), both outcomes were statistically indistinguishable.
“Because our investigation noted that the overall survival of patients undergoing a second lung cancer operation was similar to those patients undergoing a first operation, and because there is a rising rate of second lung cancer in lung cancer survivors, we feel that continued surveillance beyond the 4-year interval as recommended by the American Association for Thoracic Surgery as well as the [National Comprehensive Cancer Network] guidelines would be beneficial to long-term survivors of early-stage lung cancer,” Dr. Varlotto and coinvestigators wrote.
“The recent results from recent lung cancer screening studies demonstrate that females may benefit preferentially from screening … and our study suggests that these preferential benefits of increased CT scan surveillance may extend to females who are long-term survivors of lung cancer as well,” they added.
Findings in context
“As this is an observational study, it is challenging to understand what is driving the rise in prevalence of second lung cancers,” Mara Antonoff, MD, of The University of Texas MD Anderson Cancer Center in Houston commented in an interview.
“Overall, the findings are very important, as they suggest that we should continue to perform surveillance imaging for patients beyond recommended guidelines, which may allow us to achieve better survival outcomes for those individuals who develop a second lung cancer years after the first lung cancer,” she agreed.
“Just as lung cancer screening is important to identifying lung cancers at an earlier stage when they are more easily treatable and more likely to be cured, surveillance after an initial treatment for lung cancer would allow a diagnosis of second lung cancers at an earlier stage, so the patients can again achieve durable cure,” Dr. Antonoff concluded.
Study details
For the study, Dr. Varlotto and coinvestigators used data from SEER-13 and SEER-18 to identify patients with a lung cancer diagnosis during 1998-2013, and data from SEER-9, covering the years 1985-2014, to calculate rates of second cancers occurring 4-10 years after a first lung cancer.
Analyses were based on 58,758 patients with a surgically resected first primary lung cancer (55.9% with early-stage disease) and 384 patients with a surgically resected second primary lung cancer (77.6% with early-stage disease). Median follow-up was 76 months for the former and 46 months for the latter.
Results showed that in the 4-10 years after a first lung cancer diagnosis, the age-adjusted incidence of second lung cancers rose by study year but remained less than that of all other second cancers combined until the mid-2000s. Among women, incidence started rising sharply in 2001 and significantly exceeded that of all other second cancers starting in 2005.
In the entire population of study patients, propensity-adjusted analyses showed that second lung cancers were similar to first lung cancers on overall survival (P = .1726) but had worse lung cancer–specific survival (P = .0143). However, in the subset of patients with early-stage resectable disease, second and first lung cancers were similar on both overall survival (P = .3872) and lung cancer–specific survival (P = .1276).
Dr. Varlotto disclosed that he had no conflicts of interest. The study was funded by the Department of Radiation Oncology, University of Massachusetts. Dr. Antonoff disclosed that she had no relevant conflicts of interest.
SOURCE: Varlotto JM et al. Lung Cancer. 2020;147:115-122.
FROM LUNG CANCER