User login
Digestive Disease Week® – always the biggest GI meeting in the world
GI & Hepatology News will be in Chicago this week at the McCormick Place Convention Center reporting the latest news and perspective across gastroenterology. Studies at this year’s meeting have a decidedly genetic slant as the genetic bases for many GI and liver diseases are being determined and studied for their use in treatments.
Our reporters will cover comparative effectiveness studies and controversies in inflammatory bowel disease; Clostridium difficile colitis; and prevention and treatment of clinical hepatitis, among many other topics, as well as an NIH Consortium presentation on chronic pancreatitis, diabetes, and pancreatic cancer.
Highly anticipated presentations include:
- Genetic markers could improve treatment of hepatitis C.
- Nonsurgical weight-loss treatment could help patients with limited options.
- First-ever autonomously controlled “capsule robot” explores colon.
- Enzyme could be a game-changer for gluten-sensitive patients.
Our team will provide daily coverage, starting Saturday, May 6.
GI & Hepatology News will be in Chicago this week at the McCormick Place Convention Center reporting the latest news and perspective across gastroenterology. Studies at this year’s meeting have a decidedly genetic slant as the genetic bases for many GI and liver diseases are being determined and studied for their use in treatments.
Our reporters will cover comparative effectiveness studies and controversies in inflammatory bowel disease; Clostridium difficile colitis; and prevention and treatment of clinical hepatitis, among many other topics, as well as an NIH Consortium presentation on chronic pancreatitis, diabetes, and pancreatic cancer.
Highly anticipated presentations include:
- Genetic markers could improve treatment of hepatitis C.
- Nonsurgical weight-loss treatment could help patients with limited options.
- First-ever autonomously controlled “capsule robot” explores colon.
- Enzyme could be a game-changer for gluten-sensitive patients.
Our team will provide daily coverage, starting Saturday, May 6.
GI & Hepatology News will be in Chicago this week at the McCormick Place Convention Center reporting the latest news and perspective across gastroenterology. Studies at this year’s meeting have a decidedly genetic slant as the genetic bases for many GI and liver diseases are being determined and studied for their use in treatments.
Our reporters will cover comparative effectiveness studies and controversies in inflammatory bowel disease; Clostridium difficile colitis; and prevention and treatment of clinical hepatitis, among many other topics, as well as an NIH Consortium presentation on chronic pancreatitis, diabetes, and pancreatic cancer.
Highly anticipated presentations include:
- Genetic markers could improve treatment of hepatitis C.
- Nonsurgical weight-loss treatment could help patients with limited options.
- First-ever autonomously controlled “capsule robot” explores colon.
- Enzyme could be a game-changer for gluten-sensitive patients.
Our team will provide daily coverage, starting Saturday, May 6.
FROM DDW
Project Sonar brings gastroenterologists one step closer to a specialty-specific APM
Gastroenterologists wanting to participate in the advanced alternative payment model track of the new Quality Payment Program developed under the Medicare Access and CHIP Reauthorization Act (MACRA) may soon have an option as early as 2018.
Project Sonar, a program that helps gastroenterologists and their patients help manage inflammatory bowel diseases such as Crohn’s disease and ulcerative colitis was the first physician-focused gastroenterology-specific payment model to receive a recommendation from the Physician-Focused Payment Model Technical Advisory Committee (PTAC). The proposal was supported by public comments from AGA and the American Medical Association. It now heads to the U.S. Department of Health & Human Services for final approval.
“The intensive medical home Blue Cross initiated with us because of the success of the pilot pays us a supplemental management fee for every patient that is enrolled,” Lawrence Kosinski, MD, MBA, managing partner at IGG, and a Practice Councillor for the AGA Institute Governing Board, said in an interview. “That management fee is adjusted based on how much savings we produce for the payer. It ultimately winds up being a shared savings program based on our ability to control the cost of care, but we get a management fee upfront in order to make it happen.”
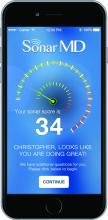
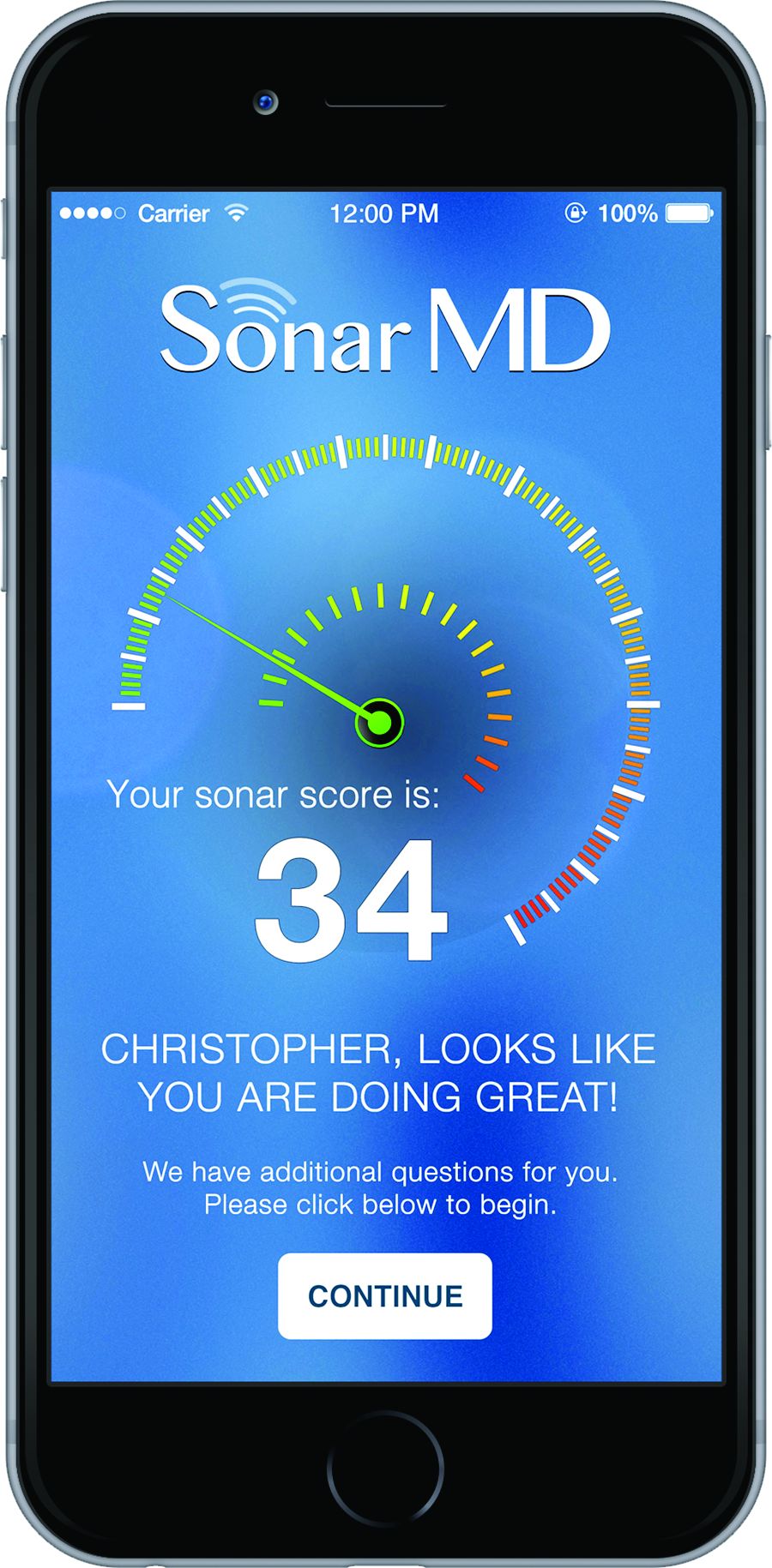
And getting those savings was simply a matter of getting patients engaged in their care. The way Project Sonar works is patients are “pinged” once a month to report on their symptoms. They generally receive a text alert with a link to a secure website where they answer five questions that are derived from the Crohn’s Disease Activity Index. The first one asks how many bowel movements per day over the last 7 days. The second one asks for a rating of abdominal pain. The third one is a rating of the patient’s general state of well-being. Then they are asked whether they are taking drugs for diarrhea. Finally, there are some check boxes for whether they have any eye symptoms or skin rashes or joint pains.
The answers are then weighted and a score is given both to the patient and the doctor. Doctors are alerted to which patients might require more focused attention over those who have their symptoms well managed.
But what is making this program work is that IGG has been able to get an 80% sustained response rate from patients using the program.
“The entire savings that we are generating is coming solely from the patients who participate in the platform,” Dr. Kosinski said.
From an analysis of BCBS of Illinois’ claims data, Dr. Kosinski and his colleagues determined that “ it costs about $24,000 a year to take care of a Crohn’s patient, and over half the money that is spent is spent for inpatient care, complications, bad things happening to these patients.”
He continued: “The difference in annual costs between a pinger and a nonpinger is $6,300. That is a 25% drop in the cost of care.” He added that Project Sonar is leading to a 10% savings in the overall cost of care for these patients when total costs are factored in.
One surprise aspect that Dr. Kosinski found was that patients needed no additional incentive to respond to their pings.
“We had thought we were going to have to get into some gameification and we actually ran a few lotteries to reward people,” he said. “We haven’t had to do it. The patients have participated because I think they feel like they are getting excellent care this way, and I’m very impressed by the fact that the patients have not had to be stimulated to do it.”
At the end of the day, it’s all about improving patients’ lives.
“Patients are doing better,” he noted. “They are staying out of the hospital. They are staying out of the operating room. They are staying at work. They are staying with their families. We have to realize, we look at these as patients with diseases. These are human beings that just happen to have a disease, and they have a life, they have jobs, family, responsibility, and if our Sonar system is allowing them to have a more normal life, there is a lot of traction there.”
Dr. Kosinski is also an Associate Editor for GI & Hepatology News.
Gastroenterologists wanting to participate in the advanced alternative payment model track of the new Quality Payment Program developed under the Medicare Access and CHIP Reauthorization Act (MACRA) may soon have an option as early as 2018.
Project Sonar, a program that helps gastroenterologists and their patients help manage inflammatory bowel diseases such as Crohn’s disease and ulcerative colitis was the first physician-focused gastroenterology-specific payment model to receive a recommendation from the Physician-Focused Payment Model Technical Advisory Committee (PTAC). The proposal was supported by public comments from AGA and the American Medical Association. It now heads to the U.S. Department of Health & Human Services for final approval.
“The intensive medical home Blue Cross initiated with us because of the success of the pilot pays us a supplemental management fee for every patient that is enrolled,” Lawrence Kosinski, MD, MBA, managing partner at IGG, and a Practice Councillor for the AGA Institute Governing Board, said in an interview. “That management fee is adjusted based on how much savings we produce for the payer. It ultimately winds up being a shared savings program based on our ability to control the cost of care, but we get a management fee upfront in order to make it happen.”
And getting those savings was simply a matter of getting patients engaged in their care. The way Project Sonar works is patients are “pinged” once a month to report on their symptoms. They generally receive a text alert with a link to a secure website where they answer five questions that are derived from the Crohn’s Disease Activity Index. The first one asks how many bowel movements per day over the last 7 days. The second one asks for a rating of abdominal pain. The third one is a rating of the patient’s general state of well-being. Then they are asked whether they are taking drugs for diarrhea. Finally, there are some check boxes for whether they have any eye symptoms or skin rashes or joint pains.
The answers are then weighted and a score is given both to the patient and the doctor. Doctors are alerted to which patients might require more focused attention over those who have their symptoms well managed.
But what is making this program work is that IGG has been able to get an 80% sustained response rate from patients using the program.
“The entire savings that we are generating is coming solely from the patients who participate in the platform,” Dr. Kosinski said.
From an analysis of BCBS of Illinois’ claims data, Dr. Kosinski and his colleagues determined that “ it costs about $24,000 a year to take care of a Crohn’s patient, and over half the money that is spent is spent for inpatient care, complications, bad things happening to these patients.”
He continued: “The difference in annual costs between a pinger and a nonpinger is $6,300. That is a 25% drop in the cost of care.” He added that Project Sonar is leading to a 10% savings in the overall cost of care for these patients when total costs are factored in.
One surprise aspect that Dr. Kosinski found was that patients needed no additional incentive to respond to their pings.
“We had thought we were going to have to get into some gameification and we actually ran a few lotteries to reward people,” he said. “We haven’t had to do it. The patients have participated because I think they feel like they are getting excellent care this way, and I’m very impressed by the fact that the patients have not had to be stimulated to do it.”
At the end of the day, it’s all about improving patients’ lives.
“Patients are doing better,” he noted. “They are staying out of the hospital. They are staying out of the operating room. They are staying at work. They are staying with their families. We have to realize, we look at these as patients with diseases. These are human beings that just happen to have a disease, and they have a life, they have jobs, family, responsibility, and if our Sonar system is allowing them to have a more normal life, there is a lot of traction there.”
Dr. Kosinski is also an Associate Editor for GI & Hepatology News.
Gastroenterologists wanting to participate in the advanced alternative payment model track of the new Quality Payment Program developed under the Medicare Access and CHIP Reauthorization Act (MACRA) may soon have an option as early as 2018.
Project Sonar, a program that helps gastroenterologists and their patients help manage inflammatory bowel diseases such as Crohn’s disease and ulcerative colitis was the first physician-focused gastroenterology-specific payment model to receive a recommendation from the Physician-Focused Payment Model Technical Advisory Committee (PTAC). The proposal was supported by public comments from AGA and the American Medical Association. It now heads to the U.S. Department of Health & Human Services for final approval.
“The intensive medical home Blue Cross initiated with us because of the success of the pilot pays us a supplemental management fee for every patient that is enrolled,” Lawrence Kosinski, MD, MBA, managing partner at IGG, and a Practice Councillor for the AGA Institute Governing Board, said in an interview. “That management fee is adjusted based on how much savings we produce for the payer. It ultimately winds up being a shared savings program based on our ability to control the cost of care, but we get a management fee upfront in order to make it happen.”
And getting those savings was simply a matter of getting patients engaged in their care. The way Project Sonar works is patients are “pinged” once a month to report on their symptoms. They generally receive a text alert with a link to a secure website where they answer five questions that are derived from the Crohn’s Disease Activity Index. The first one asks how many bowel movements per day over the last 7 days. The second one asks for a rating of abdominal pain. The third one is a rating of the patient’s general state of well-being. Then they are asked whether they are taking drugs for diarrhea. Finally, there are some check boxes for whether they have any eye symptoms or skin rashes or joint pains.
The answers are then weighted and a score is given both to the patient and the doctor. Doctors are alerted to which patients might require more focused attention over those who have their symptoms well managed.
But what is making this program work is that IGG has been able to get an 80% sustained response rate from patients using the program.
“The entire savings that we are generating is coming solely from the patients who participate in the platform,” Dr. Kosinski said.
From an analysis of BCBS of Illinois’ claims data, Dr. Kosinski and his colleagues determined that “ it costs about $24,000 a year to take care of a Crohn’s patient, and over half the money that is spent is spent for inpatient care, complications, bad things happening to these patients.”
He continued: “The difference in annual costs between a pinger and a nonpinger is $6,300. That is a 25% drop in the cost of care.” He added that Project Sonar is leading to a 10% savings in the overall cost of care for these patients when total costs are factored in.
One surprise aspect that Dr. Kosinski found was that patients needed no additional incentive to respond to their pings.
“We had thought we were going to have to get into some gameification and we actually ran a few lotteries to reward people,” he said. “We haven’t had to do it. The patients have participated because I think they feel like they are getting excellent care this way, and I’m very impressed by the fact that the patients have not had to be stimulated to do it.”
At the end of the day, it’s all about improving patients’ lives.
“Patients are doing better,” he noted. “They are staying out of the hospital. They are staying out of the operating room. They are staying at work. They are staying with their families. We have to realize, we look at these as patients with diseases. These are human beings that just happen to have a disease, and they have a life, they have jobs, family, responsibility, and if our Sonar system is allowing them to have a more normal life, there is a lot of traction there.”
Dr. Kosinski is also an Associate Editor for GI & Hepatology News.
Renflexis approved as second infliximab biosimilar
Infliximab-abda is the second infliximab biosimilar approved by the Food and Drug Administration, the agency announced April 21.
Infliximab-abda, to be marketed as Renflexis, is approved for all indications as the reference product, including Crohn’s diseases in adults and children, ulcerative colitis, rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis, and plaque psoriasis, according to the product label.
Like Remicade, Renflexis will come with a boxed warning and a Medication Guide that describes important information about its uses and risks, which include serious infections, lymphoma and other malignancies, liver injury, blood problems, lupuslike syndrome, psoriasis, and in rare cases, nervous system disorders.
Renflexis will be marketed by Merck Sharp & Dohme and is manufactured by Samsung Bioepis.
dfulton@frontlinemedcom.com
On Twitter @denisefulton
Infliximab-abda is the second infliximab biosimilar approved by the Food and Drug Administration, the agency announced April 21.
Infliximab-abda, to be marketed as Renflexis, is approved for all indications as the reference product, including Crohn’s diseases in adults and children, ulcerative colitis, rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis, and plaque psoriasis, according to the product label.
Like Remicade, Renflexis will come with a boxed warning and a Medication Guide that describes important information about its uses and risks, which include serious infections, lymphoma and other malignancies, liver injury, blood problems, lupuslike syndrome, psoriasis, and in rare cases, nervous system disorders.
Renflexis will be marketed by Merck Sharp & Dohme and is manufactured by Samsung Bioepis.
dfulton@frontlinemedcom.com
On Twitter @denisefulton
Infliximab-abda is the second infliximab biosimilar approved by the Food and Drug Administration, the agency announced April 21.
Infliximab-abda, to be marketed as Renflexis, is approved for all indications as the reference product, including Crohn’s diseases in adults and children, ulcerative colitis, rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis, and plaque psoriasis, according to the product label.
Like Remicade, Renflexis will come with a boxed warning and a Medication Guide that describes important information about its uses and risks, which include serious infections, lymphoma and other malignancies, liver injury, blood problems, lupuslike syndrome, psoriasis, and in rare cases, nervous system disorders.
Renflexis will be marketed by Merck Sharp & Dohme and is manufactured by Samsung Bioepis.
dfulton@frontlinemedcom.com
On Twitter @denisefulton
CDC: Some Shigella strains show reduced ciprofloxacin susceptibility
The Centers for Disease Control and Prevention has identified an increase in Shigella isolates with reduced susceptibility to ciprofloxacin, and has released an official health advisory outlining new recommendations for clinical diagnosis, management, and reporting, as well as for laboratories and public health officials.
The Shigella isolates of concern in the United States have minimum inhibitory concentration (MIC) values of 0.12-1 mcg/mL for ciprofloxacin, which is within the range considered susceptible. These strains, however, “often have a quinolone resistance gene that may lead to clinically significant reduced susceptibility to fluoroquinolone antibiotics,” such as ciprofloxacin, according to the CDC advisory.
It is possible that strains with MIC in the 0.12-1 mcg/mL range may have worse clinical outcome or increased risk of transmission, so the CDC made the following recommendations to clinicians:
• Order a stool culture to obtain isolates for antimicrobial susceptibility testing in suspected cases.
• Order antimicrobial susceptibility testing when ordering a stool culture for Shigella.
• Avoid routine prescribing of antibiotic therapy for Shigella infection, instead reserving antibiotics for patients with a clinical indication or when advised by public health officials in an outbreak setting.
• Tailor antibiotic choice (when antibiotics are indicated) to susceptibility results as soon as possible – with special attention given to the MIC for fluoroquinolone antibiotics.
• Obtain follow-up stool cultures in shigellosis patients who have continued or worsening symptoms despite antibiotic therapy.
• Consult local or state health departments for guidance regarding when patients may return to child care, school, or work.
• Counsel patients with active diarrhea on how they can prevent spreading the infection to others, regardless of whether antibiotic treatment is prescribed.
Additionally, the CDC noted that shigellosis is a nationally notifiable condition; all cases should be reported to the local health department. If a patient with shigellosis and a ciprofloxacin MIC of 0.12-1 mcg/mL is identified, this information should be included in the report to facilitate further testing of the isolate.
The CDC reported that it is working with state and local public health departments and clinical partners to determine if outcomes are indeed worse for patients treated with ciprofloxacin for Shigella strains harboring a quinolone resistance gene, and it will continue to monitor trends in susceptibility of Shigella isolates and to perform genetic testing on select strains to confirm the presence and type of resistance genes.
The Centers for Disease Control and Prevention has identified an increase in Shigella isolates with reduced susceptibility to ciprofloxacin, and has released an official health advisory outlining new recommendations for clinical diagnosis, management, and reporting, as well as for laboratories and public health officials.
The Shigella isolates of concern in the United States have minimum inhibitory concentration (MIC) values of 0.12-1 mcg/mL for ciprofloxacin, which is within the range considered susceptible. These strains, however, “often have a quinolone resistance gene that may lead to clinically significant reduced susceptibility to fluoroquinolone antibiotics,” such as ciprofloxacin, according to the CDC advisory.
It is possible that strains with MIC in the 0.12-1 mcg/mL range may have worse clinical outcome or increased risk of transmission, so the CDC made the following recommendations to clinicians:
• Order a stool culture to obtain isolates for antimicrobial susceptibility testing in suspected cases.
• Order antimicrobial susceptibility testing when ordering a stool culture for Shigella.
• Avoid routine prescribing of antibiotic therapy for Shigella infection, instead reserving antibiotics for patients with a clinical indication or when advised by public health officials in an outbreak setting.
• Tailor antibiotic choice (when antibiotics are indicated) to susceptibility results as soon as possible – with special attention given to the MIC for fluoroquinolone antibiotics.
• Obtain follow-up stool cultures in shigellosis patients who have continued or worsening symptoms despite antibiotic therapy.
• Consult local or state health departments for guidance regarding when patients may return to child care, school, or work.
• Counsel patients with active diarrhea on how they can prevent spreading the infection to others, regardless of whether antibiotic treatment is prescribed.
Additionally, the CDC noted that shigellosis is a nationally notifiable condition; all cases should be reported to the local health department. If a patient with shigellosis and a ciprofloxacin MIC of 0.12-1 mcg/mL is identified, this information should be included in the report to facilitate further testing of the isolate.
The CDC reported that it is working with state and local public health departments and clinical partners to determine if outcomes are indeed worse for patients treated with ciprofloxacin for Shigella strains harboring a quinolone resistance gene, and it will continue to monitor trends in susceptibility of Shigella isolates and to perform genetic testing on select strains to confirm the presence and type of resistance genes.
The Centers for Disease Control and Prevention has identified an increase in Shigella isolates with reduced susceptibility to ciprofloxacin, and has released an official health advisory outlining new recommendations for clinical diagnosis, management, and reporting, as well as for laboratories and public health officials.
The Shigella isolates of concern in the United States have minimum inhibitory concentration (MIC) values of 0.12-1 mcg/mL for ciprofloxacin, which is within the range considered susceptible. These strains, however, “often have a quinolone resistance gene that may lead to clinically significant reduced susceptibility to fluoroquinolone antibiotics,” such as ciprofloxacin, according to the CDC advisory.
It is possible that strains with MIC in the 0.12-1 mcg/mL range may have worse clinical outcome or increased risk of transmission, so the CDC made the following recommendations to clinicians:
• Order a stool culture to obtain isolates for antimicrobial susceptibility testing in suspected cases.
• Order antimicrobial susceptibility testing when ordering a stool culture for Shigella.
• Avoid routine prescribing of antibiotic therapy for Shigella infection, instead reserving antibiotics for patients with a clinical indication or when advised by public health officials in an outbreak setting.
• Tailor antibiotic choice (when antibiotics are indicated) to susceptibility results as soon as possible – with special attention given to the MIC for fluoroquinolone antibiotics.
• Obtain follow-up stool cultures in shigellosis patients who have continued or worsening symptoms despite antibiotic therapy.
• Consult local or state health departments for guidance regarding when patients may return to child care, school, or work.
• Counsel patients with active diarrhea on how they can prevent spreading the infection to others, regardless of whether antibiotic treatment is prescribed.
Additionally, the CDC noted that shigellosis is a nationally notifiable condition; all cases should be reported to the local health department. If a patient with shigellosis and a ciprofloxacin MIC of 0.12-1 mcg/mL is identified, this information should be included in the report to facilitate further testing of the isolate.
The CDC reported that it is working with state and local public health departments and clinical partners to determine if outcomes are indeed worse for patients treated with ciprofloxacin for Shigella strains harboring a quinolone resistance gene, and it will continue to monitor trends in susceptibility of Shigella isolates and to perform genetic testing on select strains to confirm the presence and type of resistance genes.
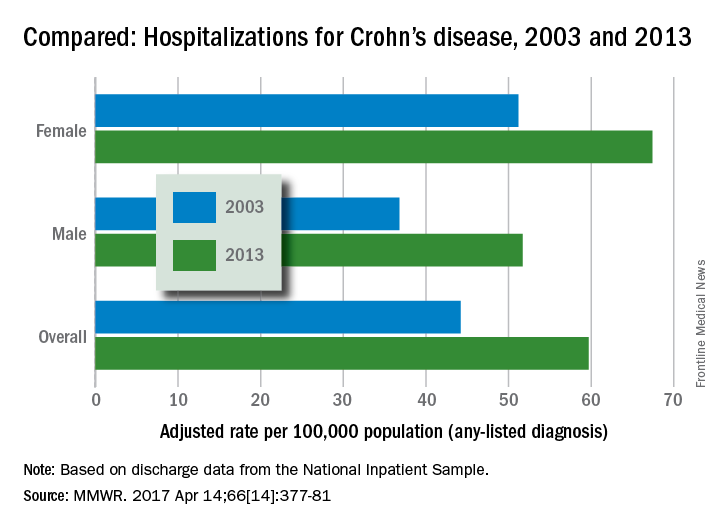
Crohn’s hospitalizations up significantly … or not
Hospitalizations for Crohn’s disease were up by a statistically significant 35% from 2003 to 2013 … or they were up just 5%, according to the Centers for Disease Control and Prevention.
It depends on how you look at it. In 2013, age-adjusted hospitalization was 59.7 stays per 100,000 population for Crohn’s as any-listed diagnosis – indicating that patients had Crohn’s disease but that it was not necessarily the main reason they were being hospitalized – compared with 44.2 per 100,000 in 2003. That’s an increase of 35%, the CDC said (MMWR. 2017 Apr 14;66[14]:377-81).
Hospitalizations with Crohn’s as the first-listed diagnosis – making it the main reason for the admission – were up 5% over the same period: 18.2 stays per 100,000 pop. in 2003 and 19.1 per 100,000 in 2013. That increase is not statistically significant, the CDC noted.
The increase in any-listed diagnosis “might represent greater physician awareness and diagnosis of Crohn’s disease or more complete coding of secondary diagnoses by physicians,” CDC investigators suggested.
Increases in hospitalizations for any-listed Crohn’s were significant for both men (40.5%) and women (31.6%). For first-listed Crohn’s, women saw a slight but not significant decrease of 1% while men had a significant 14.5% increase, according to the report, which was based on data from the National Inpatient Sample.
Hospitalizations for Crohn’s disease were up by a statistically significant 35% from 2003 to 2013 … or they were up just 5%, according to the Centers for Disease Control and Prevention.
It depends on how you look at it. In 2013, age-adjusted hospitalization was 59.7 stays per 100,000 population for Crohn’s as any-listed diagnosis – indicating that patients had Crohn’s disease but that it was not necessarily the main reason they were being hospitalized – compared with 44.2 per 100,000 in 2003. That’s an increase of 35%, the CDC said (MMWR. 2017 Apr 14;66[14]:377-81).
Hospitalizations with Crohn’s as the first-listed diagnosis – making it the main reason for the admission – were up 5% over the same period: 18.2 stays per 100,000 pop. in 2003 and 19.1 per 100,000 in 2013. That increase is not statistically significant, the CDC noted.
The increase in any-listed diagnosis “might represent greater physician awareness and diagnosis of Crohn’s disease or more complete coding of secondary diagnoses by physicians,” CDC investigators suggested.
Increases in hospitalizations for any-listed Crohn’s were significant for both men (40.5%) and women (31.6%). For first-listed Crohn’s, women saw a slight but not significant decrease of 1% while men had a significant 14.5% increase, according to the report, which was based on data from the National Inpatient Sample.
Hospitalizations for Crohn’s disease were up by a statistically significant 35% from 2003 to 2013 … or they were up just 5%, according to the Centers for Disease Control and Prevention.
It depends on how you look at it. In 2013, age-adjusted hospitalization was 59.7 stays per 100,000 population for Crohn’s as any-listed diagnosis – indicating that patients had Crohn’s disease but that it was not necessarily the main reason they were being hospitalized – compared with 44.2 per 100,000 in 2003. That’s an increase of 35%, the CDC said (MMWR. 2017 Apr 14;66[14]:377-81).
Hospitalizations with Crohn’s as the first-listed diagnosis – making it the main reason for the admission – were up 5% over the same period: 18.2 stays per 100,000 pop. in 2003 and 19.1 per 100,000 in 2013. That increase is not statistically significant, the CDC noted.
The increase in any-listed diagnosis “might represent greater physician awareness and diagnosis of Crohn’s disease or more complete coding of secondary diagnoses by physicians,” CDC investigators suggested.
Increases in hospitalizations for any-listed Crohn’s were significant for both men (40.5%) and women (31.6%). For first-listed Crohn’s, women saw a slight but not significant decrease of 1% while men had a significant 14.5% increase, according to the report, which was based on data from the National Inpatient Sample.
FROM MORBIDITY AND MORTALITY WEEKLY REPORT
A viral inducer of celiac disease?
A viral infection may be the culprit behind celiac disease, which is caused by an autoimmune response to dietary gluten. The findings are based on an engineered reovirus, which is normally benign. The researchers believe that a reovirus may disrupt intestinal immune homeostasis in susceptible individuals as a result of infection during childhood.
According to in vitro and mouse studies carried out by the researchers, one strain of reovirus suppresses peripheral regulatory T-cell conversion and promotes T helper 1 immune response at sites that normally induce tolerance to dietary antigens. The work appeared in the April issue of Science (2017;356:44-50).
The researchers decided to investigate reoviruses. They often infect humans, commonly in early childhood when gluten usually is first introduced. They also infect humans and mice similarly, allowing a more straightforward comparison between human and mouse studies than would be possible in other virus types.
The researchers created an engineered virus made from two reovirus strains, T1L and T3D, which naturally reassort in human hosts. T1L infects the intestine, while T3D does not. The new strain, T3D-RV, retains most of the characteristics of T3D but can also infect the intestine.
The researchers then conducted mouse studies and showed that both T1L and T3D-RV affect immune responses to dietary antigens at the inductive and effector sites of oral tolerance. However, the original T1L strain caused more changes in gene transcription, both in the number of genes and the intensity of transcription level. This suggested that T1L might uniquely alter immunogenic responses to dietary antigens.
A further test in mice showed that T1L also prompted a proinflammatory response in dendritic cells that take up ovalbumin, but T3D-RV did not. Furthermore, T1L interfered with induction of peripheral tolerance to oral ovalbumin, and T3D-RV did not.
With this data in hand, the researchers turned to human subjects. They compared 73 healthy controls to 160 patients with celiac disease who were on a gluten-free diet. Celiac disease patients had higher mean antireovirus antibody titers, though the result fell short of statistical significance (P = .06), and subjects with celiac disease were over-represented among subjects who had antireovirus titers above the median value.
“You can have two viruses of the same family infecting the intestine in the same way, inducing protective immunity, and being cleared, but only one sets the stage for disease. Finally, using these two viruses allows [us] to dissociate protective immunity from immunopathology. Only the virus that has the capacity to enter the site where dietary proteins are seen by the immune system can trigger disease,” said Bana Jabri, MD, PhD, professor of medicine at the University of Chicago.
Reovirus is unlikely to be the only, otherwise harmless, virus that could prompt wayward immune responses. The research points the way to the identification of viruses linked to celiac disease and other autoimmune diseases and could inform vaccine strategies to prevent such conditions.
The study received funding from the National Institutes of Health and the University of Chicago. No conflict of interest information was disclosed in the article.
A viral infection may be the culprit behind celiac disease, which is caused by an autoimmune response to dietary gluten. The findings are based on an engineered reovirus, which is normally benign. The researchers believe that a reovirus may disrupt intestinal immune homeostasis in susceptible individuals as a result of infection during childhood.
According to in vitro and mouse studies carried out by the researchers, one strain of reovirus suppresses peripheral regulatory T-cell conversion and promotes T helper 1 immune response at sites that normally induce tolerance to dietary antigens. The work appeared in the April issue of Science (2017;356:44-50).
The researchers decided to investigate reoviruses. They often infect humans, commonly in early childhood when gluten usually is first introduced. They also infect humans and mice similarly, allowing a more straightforward comparison between human and mouse studies than would be possible in other virus types.
The researchers created an engineered virus made from two reovirus strains, T1L and T3D, which naturally reassort in human hosts. T1L infects the intestine, while T3D does not. The new strain, T3D-RV, retains most of the characteristics of T3D but can also infect the intestine.
The researchers then conducted mouse studies and showed that both T1L and T3D-RV affect immune responses to dietary antigens at the inductive and effector sites of oral tolerance. However, the original T1L strain caused more changes in gene transcription, both in the number of genes and the intensity of transcription level. This suggested that T1L might uniquely alter immunogenic responses to dietary antigens.
A further test in mice showed that T1L also prompted a proinflammatory response in dendritic cells that take up ovalbumin, but T3D-RV did not. Furthermore, T1L interfered with induction of peripheral tolerance to oral ovalbumin, and T3D-RV did not.
With this data in hand, the researchers turned to human subjects. They compared 73 healthy controls to 160 patients with celiac disease who were on a gluten-free diet. Celiac disease patients had higher mean antireovirus antibody titers, though the result fell short of statistical significance (P = .06), and subjects with celiac disease were over-represented among subjects who had antireovirus titers above the median value.
“You can have two viruses of the same family infecting the intestine in the same way, inducing protective immunity, and being cleared, but only one sets the stage for disease. Finally, using these two viruses allows [us] to dissociate protective immunity from immunopathology. Only the virus that has the capacity to enter the site where dietary proteins are seen by the immune system can trigger disease,” said Bana Jabri, MD, PhD, professor of medicine at the University of Chicago.
Reovirus is unlikely to be the only, otherwise harmless, virus that could prompt wayward immune responses. The research points the way to the identification of viruses linked to celiac disease and other autoimmune diseases and could inform vaccine strategies to prevent such conditions.
The study received funding from the National Institutes of Health and the University of Chicago. No conflict of interest information was disclosed in the article.
A viral infection may be the culprit behind celiac disease, which is caused by an autoimmune response to dietary gluten. The findings are based on an engineered reovirus, which is normally benign. The researchers believe that a reovirus may disrupt intestinal immune homeostasis in susceptible individuals as a result of infection during childhood.
According to in vitro and mouse studies carried out by the researchers, one strain of reovirus suppresses peripheral regulatory T-cell conversion and promotes T helper 1 immune response at sites that normally induce tolerance to dietary antigens. The work appeared in the April issue of Science (2017;356:44-50).
The researchers decided to investigate reoviruses. They often infect humans, commonly in early childhood when gluten usually is first introduced. They also infect humans and mice similarly, allowing a more straightforward comparison between human and mouse studies than would be possible in other virus types.
The researchers created an engineered virus made from two reovirus strains, T1L and T3D, which naturally reassort in human hosts. T1L infects the intestine, while T3D does not. The new strain, T3D-RV, retains most of the characteristics of T3D but can also infect the intestine.
The researchers then conducted mouse studies and showed that both T1L and T3D-RV affect immune responses to dietary antigens at the inductive and effector sites of oral tolerance. However, the original T1L strain caused more changes in gene transcription, both in the number of genes and the intensity of transcription level. This suggested that T1L might uniquely alter immunogenic responses to dietary antigens.
A further test in mice showed that T1L also prompted a proinflammatory response in dendritic cells that take up ovalbumin, but T3D-RV did not. Furthermore, T1L interfered with induction of peripheral tolerance to oral ovalbumin, and T3D-RV did not.
With this data in hand, the researchers turned to human subjects. They compared 73 healthy controls to 160 patients with celiac disease who were on a gluten-free diet. Celiac disease patients had higher mean antireovirus antibody titers, though the result fell short of statistical significance (P = .06), and subjects with celiac disease were over-represented among subjects who had antireovirus titers above the median value.
“You can have two viruses of the same family infecting the intestine in the same way, inducing protective immunity, and being cleared, but only one sets the stage for disease. Finally, using these two viruses allows [us] to dissociate protective immunity from immunopathology. Only the virus that has the capacity to enter the site where dietary proteins are seen by the immune system can trigger disease,” said Bana Jabri, MD, PhD, professor of medicine at the University of Chicago.
Reovirus is unlikely to be the only, otherwise harmless, virus that could prompt wayward immune responses. The research points the way to the identification of viruses linked to celiac disease and other autoimmune diseases and could inform vaccine strategies to prevent such conditions.
The study received funding from the National Institutes of Health and the University of Chicago. No conflict of interest information was disclosed in the article.
FROM SCIENCE
Key clinical point: Celiac disease patients have high reovirus antibody titers.
Major finding: Researchers detail mechanistic pathway that could explain a viral link.
Data source: In vitro, human, and mouse observational studies.
Disclosures: The study received funding from the National Institutes of Health and the University of Chicago. No conflict of interest information was disclosed in the article.
VIDEO: Study estimates prevalence of pediatric celiac disease, autoimmunity
By age 15 years, 3.1% of adolescents in Denver developed celiac disease, and another 2% developed a lesser degree of celiac disease autoimmunity, according to a 20-year prospective longitudinal study.
“Although more than 5% of children may experience a period of celiac disease autoimmunity [CDA], not all develop celiac disease [CD] or require gluten-free diets,” Edwin Liu, MD, of University of Colorado School of Medicine and Children’s Hospital Colorado (Aurora, Colo.), wrote with his associates in the May issue of Gastroenterology (doi: 10.1053/j.gastro.2017.02.002). Most celiac autoimmunity probably develops before age 10, “which informs future efforts for universal screening,” they added.
Source: American Gastroenterological Association
About 40% of the general population has the HLA-DQ2 or DQ8 risk genotypes for celiac disease [CD], but little is known about rates of celiac disease among children in the United States, the researchers said. To help fill this gap, they analyzed celiac-risk HLA genotypes for 31,766 infants born between 1993 and 2004 from the Diabetes Autoimmunity Study in the Young. The 1,339 children with HLA risk genotypes were followed for up to 20 years.
By age 15 years, 66 of these children (4.9%) had developed tissue transglutaminase autoantibodies (tTGA) consistent with CDA, and also met criteria for CD, the researchers said. Another 46 (3.4%) children developed only CDA, of whom 46% experienced spontaneous resolution of tTGA seropositivity without treatment. By using genotype-specific risk weighting for population frequencies of HLA, the researchers estimated that 2.4% of the general population of Denver had CDA by age 5 years, 4.3% had CDA by age 10 years, and 5.1% had CDA by age 15 years. Estimated rates of CD were 1.6%, 2.8%, and 3.1%, respectively.
These findings suggest a significant rise in the incidence of CD compared with historical estimates in the United States, and reflect recent studies “using different approaches in North America,” the researchers said. Reasons for the “dramatic increase” are unknown, but environmental causes seem likely, especially given the absence of identified genetic differences and marked changes in the prevalence of CD during the past 2 decades, they added.
Several other reports have documented fluctuating and transient tTGA antibodies in children, the researchers noted. Awareness of transient CD autoantibodies might limit public acceptance of universal screening programs for CD, they said. “Continued long-term follow-up will identify whether the autoimmunity in these subjects truly abates and tolerance develops, or if CDA will recur in time, possibly in response to additional stimulating events,” they added. “At present, low positive tTGA results should be interpreted with caution, and do not necessarily indicate need for biopsy or for treatment.”
The study did not include the DR5/DR7 risk genotype, which accounts for less than 5% of CD cases. The study also did not account for the estimated 2.5% of the general population that has DR3/DR7, which can be considered high risk, the researchers said. Thus, the study is conservative and might underestimate the real incidence of CD or CDA, they added.
The National Institutes of Health provided funding. The investigators reported having no conflicts of interest.
This study calls into question the incidence of celiac disease in the modern pediatric population and, by extension, future prevalence in adults. This is a unique prospective cohort study that followed children over a decade and a half and estimated a cumulative incidence of celiac disease of 3.1% by age 15. In sharp contrast, previous retrospective population-based studies estimated a prevalence of approximately 0.75%-1% in adult and pediatric populations. A recent publication by the United States Preventive Services Task Force used the previously accepted prevalence estimates to recommend against routine screening for celiac disease in the asymptomatic general population as well as targeted screening in those at higher risk. Increases in disease incidence as reported by the current study may call these recommendations into question, particularly in young children where cumulative incidence was high and potential for treatment benefit is substantial.
Dawn Wiese Adams, MD, MS, is assistant professor, director of celiac clinic, in the department of gastroenterology, hepatology, and nutrition, Vanderbilt University Medical Center, Nashville, Tenn. She has no conflicts of interest.
This study calls into question the incidence of celiac disease in the modern pediatric population and, by extension, future prevalence in adults. This is a unique prospective cohort study that followed children over a decade and a half and estimated a cumulative incidence of celiac disease of 3.1% by age 15. In sharp contrast, previous retrospective population-based studies estimated a prevalence of approximately 0.75%-1% in adult and pediatric populations. A recent publication by the United States Preventive Services Task Force used the previously accepted prevalence estimates to recommend against routine screening for celiac disease in the asymptomatic general population as well as targeted screening in those at higher risk. Increases in disease incidence as reported by the current study may call these recommendations into question, particularly in young children where cumulative incidence was high and potential for treatment benefit is substantial.
Dawn Wiese Adams, MD, MS, is assistant professor, director of celiac clinic, in the department of gastroenterology, hepatology, and nutrition, Vanderbilt University Medical Center, Nashville, Tenn. She has no conflicts of interest.
This study calls into question the incidence of celiac disease in the modern pediatric population and, by extension, future prevalence in adults. This is a unique prospective cohort study that followed children over a decade and a half and estimated a cumulative incidence of celiac disease of 3.1% by age 15. In sharp contrast, previous retrospective population-based studies estimated a prevalence of approximately 0.75%-1% in adult and pediatric populations. A recent publication by the United States Preventive Services Task Force used the previously accepted prevalence estimates to recommend against routine screening for celiac disease in the asymptomatic general population as well as targeted screening in those at higher risk. Increases in disease incidence as reported by the current study may call these recommendations into question, particularly in young children where cumulative incidence was high and potential for treatment benefit is substantial.
Dawn Wiese Adams, MD, MS, is assistant professor, director of celiac clinic, in the department of gastroenterology, hepatology, and nutrition, Vanderbilt University Medical Center, Nashville, Tenn. She has no conflicts of interest.
By age 15 years, 3.1% of adolescents in Denver developed celiac disease, and another 2% developed a lesser degree of celiac disease autoimmunity, according to a 20-year prospective longitudinal study.
“Although more than 5% of children may experience a period of celiac disease autoimmunity [CDA], not all develop celiac disease [CD] or require gluten-free diets,” Edwin Liu, MD, of University of Colorado School of Medicine and Children’s Hospital Colorado (Aurora, Colo.), wrote with his associates in the May issue of Gastroenterology (doi: 10.1053/j.gastro.2017.02.002). Most celiac autoimmunity probably develops before age 10, “which informs future efforts for universal screening,” they added.
Source: American Gastroenterological Association
About 40% of the general population has the HLA-DQ2 or DQ8 risk genotypes for celiac disease [CD], but little is known about rates of celiac disease among children in the United States, the researchers said. To help fill this gap, they analyzed celiac-risk HLA genotypes for 31,766 infants born between 1993 and 2004 from the Diabetes Autoimmunity Study in the Young. The 1,339 children with HLA risk genotypes were followed for up to 20 years.
By age 15 years, 66 of these children (4.9%) had developed tissue transglutaminase autoantibodies (tTGA) consistent with CDA, and also met criteria for CD, the researchers said. Another 46 (3.4%) children developed only CDA, of whom 46% experienced spontaneous resolution of tTGA seropositivity without treatment. By using genotype-specific risk weighting for population frequencies of HLA, the researchers estimated that 2.4% of the general population of Denver had CDA by age 5 years, 4.3% had CDA by age 10 years, and 5.1% had CDA by age 15 years. Estimated rates of CD were 1.6%, 2.8%, and 3.1%, respectively.
These findings suggest a significant rise in the incidence of CD compared with historical estimates in the United States, and reflect recent studies “using different approaches in North America,” the researchers said. Reasons for the “dramatic increase” are unknown, but environmental causes seem likely, especially given the absence of identified genetic differences and marked changes in the prevalence of CD during the past 2 decades, they added.
Several other reports have documented fluctuating and transient tTGA antibodies in children, the researchers noted. Awareness of transient CD autoantibodies might limit public acceptance of universal screening programs for CD, they said. “Continued long-term follow-up will identify whether the autoimmunity in these subjects truly abates and tolerance develops, or if CDA will recur in time, possibly in response to additional stimulating events,” they added. “At present, low positive tTGA results should be interpreted with caution, and do not necessarily indicate need for biopsy or for treatment.”
The study did not include the DR5/DR7 risk genotype, which accounts for less than 5% of CD cases. The study also did not account for the estimated 2.5% of the general population that has DR3/DR7, which can be considered high risk, the researchers said. Thus, the study is conservative and might underestimate the real incidence of CD or CDA, they added.
The National Institutes of Health provided funding. The investigators reported having no conflicts of interest.
By age 15 years, 3.1% of adolescents in Denver developed celiac disease, and another 2% developed a lesser degree of celiac disease autoimmunity, according to a 20-year prospective longitudinal study.
“Although more than 5% of children may experience a period of celiac disease autoimmunity [CDA], not all develop celiac disease [CD] or require gluten-free diets,” Edwin Liu, MD, of University of Colorado School of Medicine and Children’s Hospital Colorado (Aurora, Colo.), wrote with his associates in the May issue of Gastroenterology (doi: 10.1053/j.gastro.2017.02.002). Most celiac autoimmunity probably develops before age 10, “which informs future efforts for universal screening,” they added.
Source: American Gastroenterological Association
About 40% of the general population has the HLA-DQ2 or DQ8 risk genotypes for celiac disease [CD], but little is known about rates of celiac disease among children in the United States, the researchers said. To help fill this gap, they analyzed celiac-risk HLA genotypes for 31,766 infants born between 1993 and 2004 from the Diabetes Autoimmunity Study in the Young. The 1,339 children with HLA risk genotypes were followed for up to 20 years.
By age 15 years, 66 of these children (4.9%) had developed tissue transglutaminase autoantibodies (tTGA) consistent with CDA, and also met criteria for CD, the researchers said. Another 46 (3.4%) children developed only CDA, of whom 46% experienced spontaneous resolution of tTGA seropositivity without treatment. By using genotype-specific risk weighting for population frequencies of HLA, the researchers estimated that 2.4% of the general population of Denver had CDA by age 5 years, 4.3% had CDA by age 10 years, and 5.1% had CDA by age 15 years. Estimated rates of CD were 1.6%, 2.8%, and 3.1%, respectively.
These findings suggest a significant rise in the incidence of CD compared with historical estimates in the United States, and reflect recent studies “using different approaches in North America,” the researchers said. Reasons for the “dramatic increase” are unknown, but environmental causes seem likely, especially given the absence of identified genetic differences and marked changes in the prevalence of CD during the past 2 decades, they added.
Several other reports have documented fluctuating and transient tTGA antibodies in children, the researchers noted. Awareness of transient CD autoantibodies might limit public acceptance of universal screening programs for CD, they said. “Continued long-term follow-up will identify whether the autoimmunity in these subjects truly abates and tolerance develops, or if CDA will recur in time, possibly in response to additional stimulating events,” they added. “At present, low positive tTGA results should be interpreted with caution, and do not necessarily indicate need for biopsy or for treatment.”
The study did not include the DR5/DR7 risk genotype, which accounts for less than 5% of CD cases. The study also did not account for the estimated 2.5% of the general population that has DR3/DR7, which can be considered high risk, the researchers said. Thus, the study is conservative and might underestimate the real incidence of CD or CDA, they added.
The National Institutes of Health provided funding. The investigators reported having no conflicts of interest.
Key clinical point: The presence of celiac disease autoimmunity does not predict universal progression to celiac disease.
Major finding: By age 15 years, an estimated 3.1% of children in Denver developed celiac disease, and another 2% developed a lesser degree of celiac disease autoimmunity that often resolved spontaneously without treatment.
Data source: A 20-year prospective study of 1,339 children with genetic risk factors for celiac disease, with extrapolation based on the prevalence of human leukocyte antigen genotypes in the general population.
Disclosures: The National Institutes of Health provided funding. The investigators reported having no conflicts of interest.
Psyllium cut frequency of abdominal pain in pediatric IBS trial
Consuming psyllium fiber significantly reduced the frequency, but not the severity, of abdominal pain in children with irritable bowel syndrome in a randomized, double-blind, placebo-controlled trial reported in the May issue of Clinical Gastroenterology and Hepatology (2016 Nov;14[11]:1667).
Psyllium therapy did not reduce the self-reported severity of abdominal pain, Robert J. Shulman, MD, of Baylor College of Medicine in Houston reported with his associates in Clinical Gastroenterology and Hepatology. Psyllium was associated with shifts in intestinal microbiota, compared with baseline, although the changes did not reach statistical significance when compared with placebo, the researchers added. “Further studies are needed to investigate the potential mechanism whereby psyllium decreases abdominal pain frequency in children with irritable bowel syndrome [IBS],” they wrote.
IBS affects up to 20% of school-aged children. Consuming psyllium is thought to improve abdominal pain and stooling symptoms in adults with IBS, but data are inconclusive, and few randomized trials have evaluated fiber in childhood IBS. Therefore, the investigators randomly assigned 103 children (average age, 13 years; standard deviation, 3 years) with IBS who had responded inadequately to an 8-day carbohydrate elimination diet to receive a single daily dose of either psyllium or placebo maltodextrin for 6 weeks. Children aged 7-11 years received 6 g of fiber, while those aged 12-18 years received 12 g of fiber. Patients filled out a daily pain and stool diary during a 2-week baseline assessment period and again during the final 2 weeks of the trial. They also underwent breath hydrogen and methane testing, gut permeability testing, and a stool microbiota assessment during the final weekend of treatment.
At baseline, the trial arms resembled each other in terms of frequency and severity of abdominal pain, psychological characteristics, percentage of normal stools, baseline hydrogen production, and gastrointestinal permeability, the researchers said. During the final 2 weeks of treatment, the psyllium arm reported an average of 8.2 (standard deviation, 1.2) fewer episodes of abdominal pain, compared with baseline, while the control arm reported a mean reduction of 4.1 (SD, 1.3) episodes of abdominal pain (P = .03). At the end of treatment, the arms did not significantly differ in percentage of breath hydrogen or methane production, gastrointestinal permeability, or percentage of normal stools or diarrhea. However, controls had a significantly greater reduction in constipation compared with the psyllium group (P = .048).
Stool microbiome assessments of 33 children revealed a trend toward a greater increase in Bacteroidetes and a greater decrease in Firmicutes bacteria in the fiber group, compared with the control group (P = .068). The fiber group was also “marginally enriched” in bacteria of class Bacteroidia, while the placebo group was enriched in bacteria of class Clostridia (P = .094). However, the groups did not differ at narrower taxonomic levels, the researchers said. A larger sample size might have facilitated better detection of differences between groups, such as in breath hydrogen production or interactions between abdominal pain and psychological symptoms, they added.
The study was supported in part by the National Institutes of Health, the Daffy’s Foundation, and the USDA/ARS. The investigators reported having no conflicts of interest.
Consuming psyllium fiber significantly reduced the frequency, but not the severity, of abdominal pain in children with irritable bowel syndrome in a randomized, double-blind, placebo-controlled trial reported in the May issue of Clinical Gastroenterology and Hepatology (2016 Nov;14[11]:1667).
Psyllium therapy did not reduce the self-reported severity of abdominal pain, Robert J. Shulman, MD, of Baylor College of Medicine in Houston reported with his associates in Clinical Gastroenterology and Hepatology. Psyllium was associated with shifts in intestinal microbiota, compared with baseline, although the changes did not reach statistical significance when compared with placebo, the researchers added. “Further studies are needed to investigate the potential mechanism whereby psyllium decreases abdominal pain frequency in children with irritable bowel syndrome [IBS],” they wrote.
IBS affects up to 20% of school-aged children. Consuming psyllium is thought to improve abdominal pain and stooling symptoms in adults with IBS, but data are inconclusive, and few randomized trials have evaluated fiber in childhood IBS. Therefore, the investigators randomly assigned 103 children (average age, 13 years; standard deviation, 3 years) with IBS who had responded inadequately to an 8-day carbohydrate elimination diet to receive a single daily dose of either psyllium or placebo maltodextrin for 6 weeks. Children aged 7-11 years received 6 g of fiber, while those aged 12-18 years received 12 g of fiber. Patients filled out a daily pain and stool diary during a 2-week baseline assessment period and again during the final 2 weeks of the trial. They also underwent breath hydrogen and methane testing, gut permeability testing, and a stool microbiota assessment during the final weekend of treatment.
At baseline, the trial arms resembled each other in terms of frequency and severity of abdominal pain, psychological characteristics, percentage of normal stools, baseline hydrogen production, and gastrointestinal permeability, the researchers said. During the final 2 weeks of treatment, the psyllium arm reported an average of 8.2 (standard deviation, 1.2) fewer episodes of abdominal pain, compared with baseline, while the control arm reported a mean reduction of 4.1 (SD, 1.3) episodes of abdominal pain (P = .03). At the end of treatment, the arms did not significantly differ in percentage of breath hydrogen or methane production, gastrointestinal permeability, or percentage of normal stools or diarrhea. However, controls had a significantly greater reduction in constipation compared with the psyllium group (P = .048).
Stool microbiome assessments of 33 children revealed a trend toward a greater increase in Bacteroidetes and a greater decrease in Firmicutes bacteria in the fiber group, compared with the control group (P = .068). The fiber group was also “marginally enriched” in bacteria of class Bacteroidia, while the placebo group was enriched in bacteria of class Clostridia (P = .094). However, the groups did not differ at narrower taxonomic levels, the researchers said. A larger sample size might have facilitated better detection of differences between groups, such as in breath hydrogen production or interactions between abdominal pain and psychological symptoms, they added.
The study was supported in part by the National Institutes of Health, the Daffy’s Foundation, and the USDA/ARS. The investigators reported having no conflicts of interest.
Consuming psyllium fiber significantly reduced the frequency, but not the severity, of abdominal pain in children with irritable bowel syndrome in a randomized, double-blind, placebo-controlled trial reported in the May issue of Clinical Gastroenterology and Hepatology (2016 Nov;14[11]:1667).
Psyllium therapy did not reduce the self-reported severity of abdominal pain, Robert J. Shulman, MD, of Baylor College of Medicine in Houston reported with his associates in Clinical Gastroenterology and Hepatology. Psyllium was associated with shifts in intestinal microbiota, compared with baseline, although the changes did not reach statistical significance when compared with placebo, the researchers added. “Further studies are needed to investigate the potential mechanism whereby psyllium decreases abdominal pain frequency in children with irritable bowel syndrome [IBS],” they wrote.
IBS affects up to 20% of school-aged children. Consuming psyllium is thought to improve abdominal pain and stooling symptoms in adults with IBS, but data are inconclusive, and few randomized trials have evaluated fiber in childhood IBS. Therefore, the investigators randomly assigned 103 children (average age, 13 years; standard deviation, 3 years) with IBS who had responded inadequately to an 8-day carbohydrate elimination diet to receive a single daily dose of either psyllium or placebo maltodextrin for 6 weeks. Children aged 7-11 years received 6 g of fiber, while those aged 12-18 years received 12 g of fiber. Patients filled out a daily pain and stool diary during a 2-week baseline assessment period and again during the final 2 weeks of the trial. They also underwent breath hydrogen and methane testing, gut permeability testing, and a stool microbiota assessment during the final weekend of treatment.
At baseline, the trial arms resembled each other in terms of frequency and severity of abdominal pain, psychological characteristics, percentage of normal stools, baseline hydrogen production, and gastrointestinal permeability, the researchers said. During the final 2 weeks of treatment, the psyllium arm reported an average of 8.2 (standard deviation, 1.2) fewer episodes of abdominal pain, compared with baseline, while the control arm reported a mean reduction of 4.1 (SD, 1.3) episodes of abdominal pain (P = .03). At the end of treatment, the arms did not significantly differ in percentage of breath hydrogen or methane production, gastrointestinal permeability, or percentage of normal stools or diarrhea. However, controls had a significantly greater reduction in constipation compared with the psyllium group (P = .048).
Stool microbiome assessments of 33 children revealed a trend toward a greater increase in Bacteroidetes and a greater decrease in Firmicutes bacteria in the fiber group, compared with the control group (P = .068). The fiber group was also “marginally enriched” in bacteria of class Bacteroidia, while the placebo group was enriched in bacteria of class Clostridia (P = .094). However, the groups did not differ at narrower taxonomic levels, the researchers said. A larger sample size might have facilitated better detection of differences between groups, such as in breath hydrogen production or interactions between abdominal pain and psychological symptoms, they added.
The study was supported in part by the National Institutes of Health, the Daffy’s Foundation, and the USDA/ARS. The investigators reported having no conflicts of interest.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: Compared with placebo maltodextrin, consuming psyllium fiber significantly reduced the self-reported frequency of abdominal pain in children with irritable bowel syndrome.
Major finding: Children who received psyllium reported an average of 8.2 fewer pain episodes, compared with baseline, while controls reported a mean reduction of 4.1 pain episodes (P = .03).
Data source: A randomized, double-blind trial of 103 children aged 12-18 years of age with irritable bowel syndrome.
Disclosures: The study was supported in part by the National Institutes of Health, the Daffy’s Foundation, and the USDA/ARS. The investigators reported having no conflicts of interest.
USPSTF: No recommendation on screening for celiac disease
The current evidence is insufficient for the U.S. Preventive Services Task Force to recommend either for or against routine screening of asymptomatic people for celiac disease, according to a Recommendation Statement published online March 28 in JAMA.
The USPSTF is tasked with making recommendations regarding the effectiveness of specific preventive health care services for patients who have no related signs or symptoms. In this case, the group commissioned a systematic review of the literature on celiac disease from 1946 through June 2016, which became the basis for the evidence report informing their Recommendation Statement, said Kirsten Bibbins-Domingo, PhD, MD, chair of the USPSTF and lead author of the statement, and her associates.
However, only 4 studies out of the 3,036 that were examined addressed the question of screening adequately, and they offered few data of use. According to Roger Chou, MD, lead author of the Evidence Report, and his associates, no trials assessed the screening of asymptomatic children, adolescents, or adults for celiac disease with regard to morbidity, mortality, or quality of life. The evidence also was inadequate concerning targeted screening of at-risk individuals, and there were no studies of the effectiveness of treatment after screen-detected celiac disease was found.
There was some evidence supporting the diagnostic accuracy of the tissue transglutaminase-IgA test to detect celiac disease, but “little or no evidence ... to inform most of the key questions related to benefits and harms of screening for celiac disease in asymptomatic individuals,” said Dr. Chou, of the Pacific Northwest Evidence-Based Practice Center and Oregon Health & Science University, both in Portland, and his associates (JAMA. 2017 Mar 28. doi: 10.1001/jama.2016.10395).
Dr. Bibbins-Domingo noted that the American Academy of Family Physicians also has concluded that the evidence is insufficient to assess the balance of screening’s benefits and harms. In contrast, the American College of Gastroenterology recommends that screening be considered in asymptomatic people who have a first-degree relative with a confirmed diagnosis of celiac disease (JAMA. 2017 Mar 28. doi: 10.1001/jama.2017.1462).
In addition, the North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition recommends screening at age 3 years for asymptomatic children who have conditions associated with celiac disease, including type 1 diabetes, autoimmune thyroiditis, Down syndrome, Turner syndrome, William’s syndrome, or selective IgA deficiency, said Dr. Bobbins-Domingo, who is also professor of medicine at the University of California, San Francisco.
Copies of the Recommendation Statement, the Evidence Report, the authors’ disclosures, and other materials are available at www.uspreventiveservicestaskforce.org.
AGA Resource
The AGA celiac disease patient education materials will help you discuss the disorder and management with your patients. Review and download the materials, in English and Spanish, at http://www.gastro.org/patient-care/conditions-diseases/celiac-disease.
Even though the current evidence on the effectiveness of screening for celiac disease is scarce or absent, it remains reasonable for clinicians to have a low threshold for testing patients, especially in high-risk populations such as those with an affected family member or a related autoimmune disorder.
This is because most celiac disease is unrecognized, and patients can present with diverse symptoms rather than the classic triad of abdominal pain, diarrhea, and weight loss.
Rok Seon Choung, MD, and Joseph A. Murray, MD , are in the division of gastroenterology and hepatology at Mayo Clinic, Rochester, Minn. Dr. Murray reported ties to Alvine Pharmaceuticals, Alba Therapeutics, Celimmune, BioLineRx, and numerous others. Dr. Choung and Dr. Murray made these remarks in an editorial accompanying the USPSTF reports (JAMA. 2017 Mar 28;317:1221-3).
Even though the current evidence on the effectiveness of screening for celiac disease is scarce or absent, it remains reasonable for clinicians to have a low threshold for testing patients, especially in high-risk populations such as those with an affected family member or a related autoimmune disorder.
This is because most celiac disease is unrecognized, and patients can present with diverse symptoms rather than the classic triad of abdominal pain, diarrhea, and weight loss.
Rok Seon Choung, MD, and Joseph A. Murray, MD , are in the division of gastroenterology and hepatology at Mayo Clinic, Rochester, Minn. Dr. Murray reported ties to Alvine Pharmaceuticals, Alba Therapeutics, Celimmune, BioLineRx, and numerous others. Dr. Choung and Dr. Murray made these remarks in an editorial accompanying the USPSTF reports (JAMA. 2017 Mar 28;317:1221-3).
Even though the current evidence on the effectiveness of screening for celiac disease is scarce or absent, it remains reasonable for clinicians to have a low threshold for testing patients, especially in high-risk populations such as those with an affected family member or a related autoimmune disorder.
This is because most celiac disease is unrecognized, and patients can present with diverse symptoms rather than the classic triad of abdominal pain, diarrhea, and weight loss.
Rok Seon Choung, MD, and Joseph A. Murray, MD , are in the division of gastroenterology and hepatology at Mayo Clinic, Rochester, Minn. Dr. Murray reported ties to Alvine Pharmaceuticals, Alba Therapeutics, Celimmune, BioLineRx, and numerous others. Dr. Choung and Dr. Murray made these remarks in an editorial accompanying the USPSTF reports (JAMA. 2017 Mar 28;317:1221-3).
The current evidence is insufficient for the U.S. Preventive Services Task Force to recommend either for or against routine screening of asymptomatic people for celiac disease, according to a Recommendation Statement published online March 28 in JAMA.
The USPSTF is tasked with making recommendations regarding the effectiveness of specific preventive health care services for patients who have no related signs or symptoms. In this case, the group commissioned a systematic review of the literature on celiac disease from 1946 through June 2016, which became the basis for the evidence report informing their Recommendation Statement, said Kirsten Bibbins-Domingo, PhD, MD, chair of the USPSTF and lead author of the statement, and her associates.
However, only 4 studies out of the 3,036 that were examined addressed the question of screening adequately, and they offered few data of use. According to Roger Chou, MD, lead author of the Evidence Report, and his associates, no trials assessed the screening of asymptomatic children, adolescents, or adults for celiac disease with regard to morbidity, mortality, or quality of life. The evidence also was inadequate concerning targeted screening of at-risk individuals, and there were no studies of the effectiveness of treatment after screen-detected celiac disease was found.
There was some evidence supporting the diagnostic accuracy of the tissue transglutaminase-IgA test to detect celiac disease, but “little or no evidence ... to inform most of the key questions related to benefits and harms of screening for celiac disease in asymptomatic individuals,” said Dr. Chou, of the Pacific Northwest Evidence-Based Practice Center and Oregon Health & Science University, both in Portland, and his associates (JAMA. 2017 Mar 28. doi: 10.1001/jama.2016.10395).
Dr. Bibbins-Domingo noted that the American Academy of Family Physicians also has concluded that the evidence is insufficient to assess the balance of screening’s benefits and harms. In contrast, the American College of Gastroenterology recommends that screening be considered in asymptomatic people who have a first-degree relative with a confirmed diagnosis of celiac disease (JAMA. 2017 Mar 28. doi: 10.1001/jama.2017.1462).
In addition, the North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition recommends screening at age 3 years for asymptomatic children who have conditions associated with celiac disease, including type 1 diabetes, autoimmune thyroiditis, Down syndrome, Turner syndrome, William’s syndrome, or selective IgA deficiency, said Dr. Bobbins-Domingo, who is also professor of medicine at the University of California, San Francisco.
Copies of the Recommendation Statement, the Evidence Report, the authors’ disclosures, and other materials are available at www.uspreventiveservicestaskforce.org.
AGA Resource
The AGA celiac disease patient education materials will help you discuss the disorder and management with your patients. Review and download the materials, in English and Spanish, at http://www.gastro.org/patient-care/conditions-diseases/celiac-disease.
The current evidence is insufficient for the U.S. Preventive Services Task Force to recommend either for or against routine screening of asymptomatic people for celiac disease, according to a Recommendation Statement published online March 28 in JAMA.
The USPSTF is tasked with making recommendations regarding the effectiveness of specific preventive health care services for patients who have no related signs or symptoms. In this case, the group commissioned a systematic review of the literature on celiac disease from 1946 through June 2016, which became the basis for the evidence report informing their Recommendation Statement, said Kirsten Bibbins-Domingo, PhD, MD, chair of the USPSTF and lead author of the statement, and her associates.
However, only 4 studies out of the 3,036 that were examined addressed the question of screening adequately, and they offered few data of use. According to Roger Chou, MD, lead author of the Evidence Report, and his associates, no trials assessed the screening of asymptomatic children, adolescents, or adults for celiac disease with regard to morbidity, mortality, or quality of life. The evidence also was inadequate concerning targeted screening of at-risk individuals, and there were no studies of the effectiveness of treatment after screen-detected celiac disease was found.
There was some evidence supporting the diagnostic accuracy of the tissue transglutaminase-IgA test to detect celiac disease, but “little or no evidence ... to inform most of the key questions related to benefits and harms of screening for celiac disease in asymptomatic individuals,” said Dr. Chou, of the Pacific Northwest Evidence-Based Practice Center and Oregon Health & Science University, both in Portland, and his associates (JAMA. 2017 Mar 28. doi: 10.1001/jama.2016.10395).
Dr. Bibbins-Domingo noted that the American Academy of Family Physicians also has concluded that the evidence is insufficient to assess the balance of screening’s benefits and harms. In contrast, the American College of Gastroenterology recommends that screening be considered in asymptomatic people who have a first-degree relative with a confirmed diagnosis of celiac disease (JAMA. 2017 Mar 28. doi: 10.1001/jama.2017.1462).
In addition, the North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition recommends screening at age 3 years for asymptomatic children who have conditions associated with celiac disease, including type 1 diabetes, autoimmune thyroiditis, Down syndrome, Turner syndrome, William’s syndrome, or selective IgA deficiency, said Dr. Bobbins-Domingo, who is also professor of medicine at the University of California, San Francisco.
Copies of the Recommendation Statement, the Evidence Report, the authors’ disclosures, and other materials are available at www.uspreventiveservicestaskforce.org.
AGA Resource
The AGA celiac disease patient education materials will help you discuss the disorder and management with your patients. Review and download the materials, in English and Spanish, at http://www.gastro.org/patient-care/conditions-diseases/celiac-disease.
FROM JAMA
Key clinical point: The current evidence is insufficient for the USPSTF to recommend either for or against routine screening of asymptomatic people for celiac disease.
Major finding: Only 4 studies out of the 3,036 that were examined addressed the question of screening adequately.
Data source: An assessment of the benefits and harms of screening based on a review of four studies.
Disclosures: The USPSTF’s work is supported by the U.S. Agency for Healthcare Research and Quality. The authors’ financial disclosures are available at www.uspreventiveservicestaskforce.org.
Preoperative opioid use linked to worse outcomes following abdominal surgery
Surgeons need to do more to identify patients who are taking opioids preoperatively, because this is a population that appears to be at a higher risk of worse surgical outcomes, according to a large retrospective investigation.
“Opioid users represent a potentially high-risk surgical population and may require tailored perioperative care [and] the prevalence and clinical impact of this problem in the general surgery population remain unclear,” wrote the authors of a study, led by David C. Cron, a medical student of the University of Michigan, Ann Arbor. “Given the impact of pain control and gastrointestinal function on hospital stay, it is intuitive that opioid users may have increased hospital length of stay (LOS) and may incur more costs [and] also be at higher risk for surgical complications.”
The study was published in the Annals of Surgery (2017;465[4]696-701).
Mr. Cron and his coauthors made a study cohort retrospectively from abdominopelvic surgery patients from the Michigan Surgical Quality Collaborative (MSQC) database who had surgery between 2008 and 2014. All patients were treated at the University of Michigan, and were admitted within 2 days of undergoing their operation. Any patient with data indicating use of buprenorphine prior to surgery, or those were who opioid naive before admission but received opioids after being admitted, were excluded.
Investigators found a total of 3,107 patients in the MSQC database that underwent abdominopelvic surgery at the University of Michigan during the designated time frame; from that group, 2,413 were ultimately found to match the inclusion criteria for the study. The primary outcomes were 90-day total hospital costs, along with patient LOS, and 30-day rates of complications and readmissions. Data underwent covariate risk adjustment to account for age, race, gender, body mass index, insurance class, and other factors.
“Major complications are recorded by the MSQC and include: surgical site infection, deep venous thrombosis, acute renal failure, postoperative bleeding requiring transfusion, stroke, unplanned intubation, fascial dehiscence, prolonged mechanical ventilation longer than 48 hours, myocardial infarction, pneumonia, pulmonary embolism, sepsis, vascular graft loss, and renal insufficiency,” the authors noted.
Of the 2,413 subjects overall, 502 (20.8%) were found to use opioids before surgery. Differences between opioid users and nonusers were not significant in terms of age (P = .10), gender (P = .76), and race (P = .78). After adjustment, data indicated that preoperative opioid users had 9.2% higher hospital costs than nonusers (95% confidence interval, 2.8%-15.6%, P = .005) and 12.4% longer hospital stay (95% CI, 2.3%-23.5%; P = .015). Complications and readmission rates were quantified by odds ratios, which were also found to be significantly higher in subjects who were preoperative opioid users: OR = 1.36 (95% CI, 1.04-1.78; P = .023) and OR = 1.57 (95% CI, 1.08-2.29; P = .018), respectively.
The study had some limitations, including being conducted at a single center and potentially not generalizable to other types of health care facilities and population types. Additionally, information on opioid dosage and duration of use was lacking, making it that “possible that some opioid users in our study were using opioids over a shorter time period for pain related to their surgical disease,” according to the investigators.
“These results argue the potential cost-effectiveness of intervention in this unique patient population,” Mr. Cron and his colleagues concluded. “Opioid use is a potentially modifiable risk factor, and major surgery can provide powerful leverage to improve health behavior [and] our institution has implemented a preoperative program to optimize high-risk patients for surgery.”
Mr. Cron received funding from the 2015 AOA Carolyn L. Kuckein Student Research Fellowship and the Blue Cross Blue Shield of Michigan Foundation Student Research Award for this study. He and his coauthors reported no relevant financial disclosures.
The opioid epidemic is probably the biggest health care problem in the United States, and it’s getting worse. Opioids are absolutely affecting surgical outcomes on a large scale. Unfortunately, there’s no great solution in terms of how to better manage these patients – in a perfect world, we’d take the time to wean them off opioids before operating, but that isn’t very pragmatic. With such a large and expanding proportion of our patients presenting to the operating room on long-term opioids, we have developed a new cohort of high-risk surgical patients over the past several years.
Michael J. Englesbe, MD , is a professor of surgery at the University of Michigan, Ann Arbor, and a coauthor on this study.
The opioid epidemic is probably the biggest health care problem in the United States, and it’s getting worse. Opioids are absolutely affecting surgical outcomes on a large scale. Unfortunately, there’s no great solution in terms of how to better manage these patients – in a perfect world, we’d take the time to wean them off opioids before operating, but that isn’t very pragmatic. With such a large and expanding proportion of our patients presenting to the operating room on long-term opioids, we have developed a new cohort of high-risk surgical patients over the past several years.
Michael J. Englesbe, MD , is a professor of surgery at the University of Michigan, Ann Arbor, and a coauthor on this study.
The opioid epidemic is probably the biggest health care problem in the United States, and it’s getting worse. Opioids are absolutely affecting surgical outcomes on a large scale. Unfortunately, there’s no great solution in terms of how to better manage these patients – in a perfect world, we’d take the time to wean them off opioids before operating, but that isn’t very pragmatic. With such a large and expanding proportion of our patients presenting to the operating room on long-term opioids, we have developed a new cohort of high-risk surgical patients over the past several years.
Michael J. Englesbe, MD , is a professor of surgery at the University of Michigan, Ann Arbor, and a coauthor on this study.
Surgeons need to do more to identify patients who are taking opioids preoperatively, because this is a population that appears to be at a higher risk of worse surgical outcomes, according to a large retrospective investigation.
“Opioid users represent a potentially high-risk surgical population and may require tailored perioperative care [and] the prevalence and clinical impact of this problem in the general surgery population remain unclear,” wrote the authors of a study, led by David C. Cron, a medical student of the University of Michigan, Ann Arbor. “Given the impact of pain control and gastrointestinal function on hospital stay, it is intuitive that opioid users may have increased hospital length of stay (LOS) and may incur more costs [and] also be at higher risk for surgical complications.”
The study was published in the Annals of Surgery (2017;465[4]696-701).
Mr. Cron and his coauthors made a study cohort retrospectively from abdominopelvic surgery patients from the Michigan Surgical Quality Collaborative (MSQC) database who had surgery between 2008 and 2014. All patients were treated at the University of Michigan, and were admitted within 2 days of undergoing their operation. Any patient with data indicating use of buprenorphine prior to surgery, or those were who opioid naive before admission but received opioids after being admitted, were excluded.
Investigators found a total of 3,107 patients in the MSQC database that underwent abdominopelvic surgery at the University of Michigan during the designated time frame; from that group, 2,413 were ultimately found to match the inclusion criteria for the study. The primary outcomes were 90-day total hospital costs, along with patient LOS, and 30-day rates of complications and readmissions. Data underwent covariate risk adjustment to account for age, race, gender, body mass index, insurance class, and other factors.
“Major complications are recorded by the MSQC and include: surgical site infection, deep venous thrombosis, acute renal failure, postoperative bleeding requiring transfusion, stroke, unplanned intubation, fascial dehiscence, prolonged mechanical ventilation longer than 48 hours, myocardial infarction, pneumonia, pulmonary embolism, sepsis, vascular graft loss, and renal insufficiency,” the authors noted.
Of the 2,413 subjects overall, 502 (20.8%) were found to use opioids before surgery. Differences between opioid users and nonusers were not significant in terms of age (P = .10), gender (P = .76), and race (P = .78). After adjustment, data indicated that preoperative opioid users had 9.2% higher hospital costs than nonusers (95% confidence interval, 2.8%-15.6%, P = .005) and 12.4% longer hospital stay (95% CI, 2.3%-23.5%; P = .015). Complications and readmission rates were quantified by odds ratios, which were also found to be significantly higher in subjects who were preoperative opioid users: OR = 1.36 (95% CI, 1.04-1.78; P = .023) and OR = 1.57 (95% CI, 1.08-2.29; P = .018), respectively.
The study had some limitations, including being conducted at a single center and potentially not generalizable to other types of health care facilities and population types. Additionally, information on opioid dosage and duration of use was lacking, making it that “possible that some opioid users in our study were using opioids over a shorter time period for pain related to their surgical disease,” according to the investigators.
“These results argue the potential cost-effectiveness of intervention in this unique patient population,” Mr. Cron and his colleagues concluded. “Opioid use is a potentially modifiable risk factor, and major surgery can provide powerful leverage to improve health behavior [and] our institution has implemented a preoperative program to optimize high-risk patients for surgery.”
Mr. Cron received funding from the 2015 AOA Carolyn L. Kuckein Student Research Fellowship and the Blue Cross Blue Shield of Michigan Foundation Student Research Award for this study. He and his coauthors reported no relevant financial disclosures.
Surgeons need to do more to identify patients who are taking opioids preoperatively, because this is a population that appears to be at a higher risk of worse surgical outcomes, according to a large retrospective investigation.
“Opioid users represent a potentially high-risk surgical population and may require tailored perioperative care [and] the prevalence and clinical impact of this problem in the general surgery population remain unclear,” wrote the authors of a study, led by David C. Cron, a medical student of the University of Michigan, Ann Arbor. “Given the impact of pain control and gastrointestinal function on hospital stay, it is intuitive that opioid users may have increased hospital length of stay (LOS) and may incur more costs [and] also be at higher risk for surgical complications.”
The study was published in the Annals of Surgery (2017;465[4]696-701).
Mr. Cron and his coauthors made a study cohort retrospectively from abdominopelvic surgery patients from the Michigan Surgical Quality Collaborative (MSQC) database who had surgery between 2008 and 2014. All patients were treated at the University of Michigan, and were admitted within 2 days of undergoing their operation. Any patient with data indicating use of buprenorphine prior to surgery, or those were who opioid naive before admission but received opioids after being admitted, were excluded.
Investigators found a total of 3,107 patients in the MSQC database that underwent abdominopelvic surgery at the University of Michigan during the designated time frame; from that group, 2,413 were ultimately found to match the inclusion criteria for the study. The primary outcomes were 90-day total hospital costs, along with patient LOS, and 30-day rates of complications and readmissions. Data underwent covariate risk adjustment to account for age, race, gender, body mass index, insurance class, and other factors.
“Major complications are recorded by the MSQC and include: surgical site infection, deep venous thrombosis, acute renal failure, postoperative bleeding requiring transfusion, stroke, unplanned intubation, fascial dehiscence, prolonged mechanical ventilation longer than 48 hours, myocardial infarction, pneumonia, pulmonary embolism, sepsis, vascular graft loss, and renal insufficiency,” the authors noted.
Of the 2,413 subjects overall, 502 (20.8%) were found to use opioids before surgery. Differences between opioid users and nonusers were not significant in terms of age (P = .10), gender (P = .76), and race (P = .78). After adjustment, data indicated that preoperative opioid users had 9.2% higher hospital costs than nonusers (95% confidence interval, 2.8%-15.6%, P = .005) and 12.4% longer hospital stay (95% CI, 2.3%-23.5%; P = .015). Complications and readmission rates were quantified by odds ratios, which were also found to be significantly higher in subjects who were preoperative opioid users: OR = 1.36 (95% CI, 1.04-1.78; P = .023) and OR = 1.57 (95% CI, 1.08-2.29; P = .018), respectively.
The study had some limitations, including being conducted at a single center and potentially not generalizable to other types of health care facilities and population types. Additionally, information on opioid dosage and duration of use was lacking, making it that “possible that some opioid users in our study were using opioids over a shorter time period for pain related to their surgical disease,” according to the investigators.
“These results argue the potential cost-effectiveness of intervention in this unique patient population,” Mr. Cron and his colleagues concluded. “Opioid use is a potentially modifiable risk factor, and major surgery can provide powerful leverage to improve health behavior [and] our institution has implemented a preoperative program to optimize high-risk patients for surgery.”
Mr. Cron received funding from the 2015 AOA Carolyn L. Kuckein Student Research Fellowship and the Blue Cross Blue Shield of Michigan Foundation Student Research Award for this study. He and his coauthors reported no relevant financial disclosures.
FROM THE ANNALS OF SURGERY
Key clinical point:
Major finding: Preoperative opioid users had 9.2% higher costs and 12.4% longer LOS, with higher ORs for complications and readmissions, than patients who did not take opioids.
Data source: Retrospective analysis of data on 2,413 abdominopelvic surgery patients during 2008-2014.
Disclosures: Study funded by grants awarded to Mr. Cron from the 2015 AOA Carolyn L. Kuckein Student Research Fellowship and the Blue Cross Blue Shield of Michigan Foundation Student Research Award. Authors reported no relevant financial disclosures.