User login
Mongersen induces 55%-65% remission rates in Crohn’s
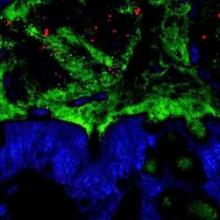
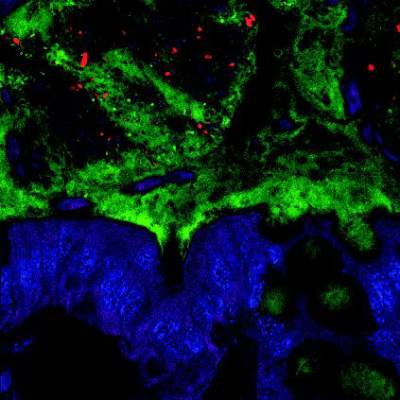
Mongersen, an oral SMAD7 antisense oligonucleotide formulated to deliver its active ingredient primarily into the lumen of the terminal ileum and right colon, induced remission rates as high as 55%-65% in a small, brief, manufacturer-sponsored, phase II clinical trial, according to a report published online March 19 in the New England Journal of Medicine.
In Crohn’s disease, gut inflammation is characterized by abnormal reductions in a particular immunosuppressive cytokine caused by increased levels of SMAD7. Mongersen (formerly GED0301) downregulates SMAD7 using a classic antisense mechanism, which in turn restores the proper cytokine function and suppresses inflammation, said Dr. Giovanni Monteleone of the department of systems medicine, University of Tor Vergata, Rome, and his associates.
They assessed a 2-week course of mongersen in 166 adults with active, moderate to severe Crohn’s disease who were treated and followed for approximately 3 months at 17 medical centers in Italy and Germany. The study participants were randomly assigned to receive one of three doses of the agent or a matching placebo in a double-blind fashion. The study’s primary endpoint was the percentage of patients in remission at day 15 who remained in remission for at least 2 more weeks. Remission was defined as a Crohn’s Disease Activity Index (CDAI) score of < 150.
Rates of remission were 65% in the 43 participants who received 160 mg of mongersen, 55% in the 40 who received 40 mg, 12% in the 41 who received 10 mg, and 10% in the 42 who received placebo. Thus, remission rates at the two highest doses of mongersen exceeded those achieved in other phase II trials for Crohn’s therapies, which ranged from 16% to 48%, the investigators said (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMoa1407250]). Rates of attaining the secondary endpoint of “clinical response,” defined as a decrease of 100 or more points in the CDAI score at day 28, also were significantly higher at the two highest doses of mongersen – 72% and 58% – than with the lowest dose (37%) or with placebo (17%).
No safety issues related to mongersen were identified in this study, but a 2-week course of treatment in such a small group of patients likely is not adequate to determine safety. Adverse events occurred in 65% of the active-treatment groups and 64% of the placebo group and were mostly mild. The nine serious adverse events that occurred were unrelated to study treatment, Dr. Monteleone and his associates said.
Further study is needed to assess longer durations of treatment, and to judge the effectiveness of the drug on the basis of endoscopic analyses of mucosal healing rather than on CDAI score. It also will be important to determine whether higher doses or longer treatment courses of mongersen raise the risk of fibrosis, given that the targeted cytokine plays a profibrogenic role in many organs, they added.
AGA Note
Review the AGA IBD Clinical Service Line to learn more about managing Crohn’s Disease.
The clinical response reported by Monteleone et al. is impressive, but it was not confirmed by endoscopic evidence of mucosal healing and it did not correlate with normalization of biomarkers such has fecal calprotectin or C-reactive protein. In short, there is a lack of congruence between clinical remission and biologic remission, an issue that must be addressed in future studies of this agent.
Also intriguing was the finding that clinical response was maintained for the duration of follow-up even though mongersen was only administered for 2 weeks and is thought not to linger in tissues. This is a stark contrast to the rapid recurrence of symptoms that characterizes withdrawal of existing anti-inflammatory drugs.
Severine Vermeire, M.D., Ph.D., is in the department of gastroenterology at Leuven (Belgium) University Hospital. She reported receiving grant support and personal fees from AbbVie, Merck Sharp & Dohme, Pfizer, Genentech/Roche, Takeda, and Mundipharma. Dr. Vermeire made these remarks in an editorial accompanying Dr. Monteleone’s report (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMe1415053]).
The clinical response reported by Monteleone et al. is impressive, but it was not confirmed by endoscopic evidence of mucosal healing and it did not correlate with normalization of biomarkers such has fecal calprotectin or C-reactive protein. In short, there is a lack of congruence between clinical remission and biologic remission, an issue that must be addressed in future studies of this agent.
Also intriguing was the finding that clinical response was maintained for the duration of follow-up even though mongersen was only administered for 2 weeks and is thought not to linger in tissues. This is a stark contrast to the rapid recurrence of symptoms that characterizes withdrawal of existing anti-inflammatory drugs.
Severine Vermeire, M.D., Ph.D., is in the department of gastroenterology at Leuven (Belgium) University Hospital. She reported receiving grant support and personal fees from AbbVie, Merck Sharp & Dohme, Pfizer, Genentech/Roche, Takeda, and Mundipharma. Dr. Vermeire made these remarks in an editorial accompanying Dr. Monteleone’s report (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMe1415053]).
The clinical response reported by Monteleone et al. is impressive, but it was not confirmed by endoscopic evidence of mucosal healing and it did not correlate with normalization of biomarkers such has fecal calprotectin or C-reactive protein. In short, there is a lack of congruence between clinical remission and biologic remission, an issue that must be addressed in future studies of this agent.
Also intriguing was the finding that clinical response was maintained for the duration of follow-up even though mongersen was only administered for 2 weeks and is thought not to linger in tissues. This is a stark contrast to the rapid recurrence of symptoms that characterizes withdrawal of existing anti-inflammatory drugs.
Severine Vermeire, M.D., Ph.D., is in the department of gastroenterology at Leuven (Belgium) University Hospital. She reported receiving grant support and personal fees from AbbVie, Merck Sharp & Dohme, Pfizer, Genentech/Roche, Takeda, and Mundipharma. Dr. Vermeire made these remarks in an editorial accompanying Dr. Monteleone’s report (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMe1415053]).
Mongersen, an oral SMAD7 antisense oligonucleotide formulated to deliver its active ingredient primarily into the lumen of the terminal ileum and right colon, induced remission rates as high as 55%-65% in a small, brief, manufacturer-sponsored, phase II clinical trial, according to a report published online March 19 in the New England Journal of Medicine.
In Crohn’s disease, gut inflammation is characterized by abnormal reductions in a particular immunosuppressive cytokine caused by increased levels of SMAD7. Mongersen (formerly GED0301) downregulates SMAD7 using a classic antisense mechanism, which in turn restores the proper cytokine function and suppresses inflammation, said Dr. Giovanni Monteleone of the department of systems medicine, University of Tor Vergata, Rome, and his associates.
They assessed a 2-week course of mongersen in 166 adults with active, moderate to severe Crohn’s disease who were treated and followed for approximately 3 months at 17 medical centers in Italy and Germany. The study participants were randomly assigned to receive one of three doses of the agent or a matching placebo in a double-blind fashion. The study’s primary endpoint was the percentage of patients in remission at day 15 who remained in remission for at least 2 more weeks. Remission was defined as a Crohn’s Disease Activity Index (CDAI) score of < 150.
Rates of remission were 65% in the 43 participants who received 160 mg of mongersen, 55% in the 40 who received 40 mg, 12% in the 41 who received 10 mg, and 10% in the 42 who received placebo. Thus, remission rates at the two highest doses of mongersen exceeded those achieved in other phase II trials for Crohn’s therapies, which ranged from 16% to 48%, the investigators said (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMoa1407250]). Rates of attaining the secondary endpoint of “clinical response,” defined as a decrease of 100 or more points in the CDAI score at day 28, also were significantly higher at the two highest doses of mongersen – 72% and 58% – than with the lowest dose (37%) or with placebo (17%).
No safety issues related to mongersen were identified in this study, but a 2-week course of treatment in such a small group of patients likely is not adequate to determine safety. Adverse events occurred in 65% of the active-treatment groups and 64% of the placebo group and were mostly mild. The nine serious adverse events that occurred were unrelated to study treatment, Dr. Monteleone and his associates said.
Further study is needed to assess longer durations of treatment, and to judge the effectiveness of the drug on the basis of endoscopic analyses of mucosal healing rather than on CDAI score. It also will be important to determine whether higher doses or longer treatment courses of mongersen raise the risk of fibrosis, given that the targeted cytokine plays a profibrogenic role in many organs, they added.
AGA Note
Review the AGA IBD Clinical Service Line to learn more about managing Crohn’s Disease.
Mongersen, an oral SMAD7 antisense oligonucleotide formulated to deliver its active ingredient primarily into the lumen of the terminal ileum and right colon, induced remission rates as high as 55%-65% in a small, brief, manufacturer-sponsored, phase II clinical trial, according to a report published online March 19 in the New England Journal of Medicine.
In Crohn’s disease, gut inflammation is characterized by abnormal reductions in a particular immunosuppressive cytokine caused by increased levels of SMAD7. Mongersen (formerly GED0301) downregulates SMAD7 using a classic antisense mechanism, which in turn restores the proper cytokine function and suppresses inflammation, said Dr. Giovanni Monteleone of the department of systems medicine, University of Tor Vergata, Rome, and his associates.
They assessed a 2-week course of mongersen in 166 adults with active, moderate to severe Crohn’s disease who were treated and followed for approximately 3 months at 17 medical centers in Italy and Germany. The study participants were randomly assigned to receive one of three doses of the agent or a matching placebo in a double-blind fashion. The study’s primary endpoint was the percentage of patients in remission at day 15 who remained in remission for at least 2 more weeks. Remission was defined as a Crohn’s Disease Activity Index (CDAI) score of < 150.
Rates of remission were 65% in the 43 participants who received 160 mg of mongersen, 55% in the 40 who received 40 mg, 12% in the 41 who received 10 mg, and 10% in the 42 who received placebo. Thus, remission rates at the two highest doses of mongersen exceeded those achieved in other phase II trials for Crohn’s therapies, which ranged from 16% to 48%, the investigators said (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMoa1407250]). Rates of attaining the secondary endpoint of “clinical response,” defined as a decrease of 100 or more points in the CDAI score at day 28, also were significantly higher at the two highest doses of mongersen – 72% and 58% – than with the lowest dose (37%) or with placebo (17%).
No safety issues related to mongersen were identified in this study, but a 2-week course of treatment in such a small group of patients likely is not adequate to determine safety. Adverse events occurred in 65% of the active-treatment groups and 64% of the placebo group and were mostly mild. The nine serious adverse events that occurred were unrelated to study treatment, Dr. Monteleone and his associates said.
Further study is needed to assess longer durations of treatment, and to judge the effectiveness of the drug on the basis of endoscopic analyses of mucosal healing rather than on CDAI score. It also will be important to determine whether higher doses or longer treatment courses of mongersen raise the risk of fibrosis, given that the targeted cytokine plays a profibrogenic role in many organs, they added.
AGA Note
Review the AGA IBD Clinical Service Line to learn more about managing Crohn’s Disease.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Mongersen, an oral SMAD7 antisense oligonucleotide, induced remission rates as high as 55%-65% in a small 2-week phase II clinical trial.
Major finding: Rates of remission were 65% in the 43 participants who received 160 mg of mongersen, 55% in the 40 who received 40 mg, 12% in the 41 who received 10 mg, and 10% in the 42 who received placebo.
Data source: A randomized placebo-controlled double-blind phase II clinical trial involving 166 adults at 17 medical centers in Italy and Germany.
Disclosures: This study was sponsored by Giuliani, acting under contract to Nogra Pharma. Dr. Monteleone reported ties to Giuliani, Novo Nordisk, Teva, Sitris, Lycera, Sofar, and Zambon, and holds a patent related to the use of SMAD7 antisense oligonucleotides in Crohn’s disease. His associates reported financial ties to numerous industry sources.
Biologics: Timing of use in IBD still variable
ORLANDO, FLA. – Biologics used to treat inflammatory bowel disease could be more effective if they were used earlier in patients with these conditions, according to an expert.
“The earlier the better in patients who need them,” said Dr. Stephen B. Hanauer, professor of medicine at Northwestern University (Chicago) and medical director of the school’s Digestive Health Center. “Biologics should be used with short-term combination therapies and with therapeutic drug monitoring.” Dr. Hanauer made his remarks at the most recent annual Advances in IBD meeting sponsored by the Crohn’s and Colitis Foundation of America.
Currently, most clinicians regard the use of immunomodulators in inflammatory bowel disease as a “last ditch effort” treatment, and turn to them only after patients fail first-line therapies. Similarly, guidelines in the field recommend immunomodulation for moderate to severe disease, the time when Dr. Hanauaer said “the disease has progressed to the point where there is already the structural damage we’re trying to prevent.”
Instead, he believes IBD sufferers would benefit from prospective trials that show earlier intervention yields higher remission rates, particularly in steroid-naive populations.
“We should learn from the rheumatoid arthritis people who figured out that administering highly effective agents earlier has the potential to modify long-term disease behavior,” Dr. Hanauer said.
Studies cited by Dr. Hanauer indicated that when biologics were introduced when patients were steroid- and immunosuppressive-naive, remission rates doubled, up to two-thirds of patients compared with only a third of patients who were given combination therapy. The results held as early as 26 weeks and at 1 year follow-up. “When we gave biologics on top of the other therapies, they offered no advantage.”
That the drugs are more widely used in rheumatoid conditions has also impacted dosing levels, said Dr. Hanauer, who said that much higher doses likely would yield better remission rates, particularly in Crohn’s disease. Not dosing at the correct levels, Dr. Harnauer said, can lead to serum troughs that diminish the patient’s response to treatment, and put them at risk of immunogenicity. “Trough levels can be used to predict clinical remission,” he said. “The problem is that we don’t have very good prospective data on it.”
Dr. Bruce E. Sands, chief of gastroenterology at Mount Sinai in (New York), and another presenter who spoke about biologics in IBD at the meeting, agreed. “When a patient fails the first anti-TNF, the question is whether it has been adequately dosed, particularly in the case of severe ulcerative colitis where we’re really starting to understand that we need higher doses to get the disease under control.”
As for earlier intervention in ulcerative colitis with biologics, Dr. Sands believed the timing is more fluid. “It really is your choice and the patient’s choice,” he said. The same was true for the recent addition of the biologic vedolizumab to the ulcerative colitis armementarium, he said, noting that there is a response both during induction and maintenance phases for all biologic therapies in ulcerative colitis.
Although infections and neoplasm are concerns with all biologic therapies, Dr. Harnauer downplayed the risks when compared to corticosteroid therapy, particularly in Crohn’s disease. “The risk of neoplasia in patients given anti-TNF (anti-tumor necrosis factor) therapy disappears after adjusting for the use of azathioprine,” he said.
Dr. Sands was less sanguine about the potential risks of anti-TNF treatment, reminding the audience that the drugs carry black box warnings from the U.S. Food and Drug Administration, and that patients must be screened for their candidacy before administering immunomodulation therapy. “It is possible to develop a variety of autoimmune disorders, and immunogenicity with these drugs,” said Dr. Sands, also adding that a risk of demylenating disorders must also be accounted for. “Make sure you monitor your patients, and that you explain the risks to them.”
Regardless of when biologics are used in treatment, both presenters agreed that their exorbitant cost is an issue. “We cannot use them for every patient who walks through the door,” said Dr. Sands.
For Dr. Harnauer, the cost stands in the way of the earlier positioning of the drugs in treatment algorithms. “If we were talking about a treatment that only cost a few dollars, we wouldn’t be worrying about any of this.” Prospective pharmaco-economic studies that go beyond measuring the in-patient and surgical costs, but account for the timing and use of biologics could help strengthen the case for earlier treatment, he said.
On Twitter @whitneymcknight
ORLANDO, FLA. – Biologics used to treat inflammatory bowel disease could be more effective if they were used earlier in patients with these conditions, according to an expert.
“The earlier the better in patients who need them,” said Dr. Stephen B. Hanauer, professor of medicine at Northwestern University (Chicago) and medical director of the school’s Digestive Health Center. “Biologics should be used with short-term combination therapies and with therapeutic drug monitoring.” Dr. Hanauer made his remarks at the most recent annual Advances in IBD meeting sponsored by the Crohn’s and Colitis Foundation of America.
Currently, most clinicians regard the use of immunomodulators in inflammatory bowel disease as a “last ditch effort” treatment, and turn to them only after patients fail first-line therapies. Similarly, guidelines in the field recommend immunomodulation for moderate to severe disease, the time when Dr. Hanauaer said “the disease has progressed to the point where there is already the structural damage we’re trying to prevent.”
Instead, he believes IBD sufferers would benefit from prospective trials that show earlier intervention yields higher remission rates, particularly in steroid-naive populations.
“We should learn from the rheumatoid arthritis people who figured out that administering highly effective agents earlier has the potential to modify long-term disease behavior,” Dr. Hanauer said.
Studies cited by Dr. Hanauer indicated that when biologics were introduced when patients were steroid- and immunosuppressive-naive, remission rates doubled, up to two-thirds of patients compared with only a third of patients who were given combination therapy. The results held as early as 26 weeks and at 1 year follow-up. “When we gave biologics on top of the other therapies, they offered no advantage.”
That the drugs are more widely used in rheumatoid conditions has also impacted dosing levels, said Dr. Hanauer, who said that much higher doses likely would yield better remission rates, particularly in Crohn’s disease. Not dosing at the correct levels, Dr. Harnauer said, can lead to serum troughs that diminish the patient’s response to treatment, and put them at risk of immunogenicity. “Trough levels can be used to predict clinical remission,” he said. “The problem is that we don’t have very good prospective data on it.”
Dr. Bruce E. Sands, chief of gastroenterology at Mount Sinai in (New York), and another presenter who spoke about biologics in IBD at the meeting, agreed. “When a patient fails the first anti-TNF, the question is whether it has been adequately dosed, particularly in the case of severe ulcerative colitis where we’re really starting to understand that we need higher doses to get the disease under control.”
As for earlier intervention in ulcerative colitis with biologics, Dr. Sands believed the timing is more fluid. “It really is your choice and the patient’s choice,” he said. The same was true for the recent addition of the biologic vedolizumab to the ulcerative colitis armementarium, he said, noting that there is a response both during induction and maintenance phases for all biologic therapies in ulcerative colitis.
Although infections and neoplasm are concerns with all biologic therapies, Dr. Harnauer downplayed the risks when compared to corticosteroid therapy, particularly in Crohn’s disease. “The risk of neoplasia in patients given anti-TNF (anti-tumor necrosis factor) therapy disappears after adjusting for the use of azathioprine,” he said.
Dr. Sands was less sanguine about the potential risks of anti-TNF treatment, reminding the audience that the drugs carry black box warnings from the U.S. Food and Drug Administration, and that patients must be screened for their candidacy before administering immunomodulation therapy. “It is possible to develop a variety of autoimmune disorders, and immunogenicity with these drugs,” said Dr. Sands, also adding that a risk of demylenating disorders must also be accounted for. “Make sure you monitor your patients, and that you explain the risks to them.”
Regardless of when biologics are used in treatment, both presenters agreed that their exorbitant cost is an issue. “We cannot use them for every patient who walks through the door,” said Dr. Sands.
For Dr. Harnauer, the cost stands in the way of the earlier positioning of the drugs in treatment algorithms. “If we were talking about a treatment that only cost a few dollars, we wouldn’t be worrying about any of this.” Prospective pharmaco-economic studies that go beyond measuring the in-patient and surgical costs, but account for the timing and use of biologics could help strengthen the case for earlier treatment, he said.
On Twitter @whitneymcknight
ORLANDO, FLA. – Biologics used to treat inflammatory bowel disease could be more effective if they were used earlier in patients with these conditions, according to an expert.
“The earlier the better in patients who need them,” said Dr. Stephen B. Hanauer, professor of medicine at Northwestern University (Chicago) and medical director of the school’s Digestive Health Center. “Biologics should be used with short-term combination therapies and with therapeutic drug monitoring.” Dr. Hanauer made his remarks at the most recent annual Advances in IBD meeting sponsored by the Crohn’s and Colitis Foundation of America.
Currently, most clinicians regard the use of immunomodulators in inflammatory bowel disease as a “last ditch effort” treatment, and turn to them only after patients fail first-line therapies. Similarly, guidelines in the field recommend immunomodulation for moderate to severe disease, the time when Dr. Hanauaer said “the disease has progressed to the point where there is already the structural damage we’re trying to prevent.”
Instead, he believes IBD sufferers would benefit from prospective trials that show earlier intervention yields higher remission rates, particularly in steroid-naive populations.
“We should learn from the rheumatoid arthritis people who figured out that administering highly effective agents earlier has the potential to modify long-term disease behavior,” Dr. Hanauer said.
Studies cited by Dr. Hanauer indicated that when biologics were introduced when patients were steroid- and immunosuppressive-naive, remission rates doubled, up to two-thirds of patients compared with only a third of patients who were given combination therapy. The results held as early as 26 weeks and at 1 year follow-up. “When we gave biologics on top of the other therapies, they offered no advantage.”
That the drugs are more widely used in rheumatoid conditions has also impacted dosing levels, said Dr. Hanauer, who said that much higher doses likely would yield better remission rates, particularly in Crohn’s disease. Not dosing at the correct levels, Dr. Harnauer said, can lead to serum troughs that diminish the patient’s response to treatment, and put them at risk of immunogenicity. “Trough levels can be used to predict clinical remission,” he said. “The problem is that we don’t have very good prospective data on it.”
Dr. Bruce E. Sands, chief of gastroenterology at Mount Sinai in (New York), and another presenter who spoke about biologics in IBD at the meeting, agreed. “When a patient fails the first anti-TNF, the question is whether it has been adequately dosed, particularly in the case of severe ulcerative colitis where we’re really starting to understand that we need higher doses to get the disease under control.”
As for earlier intervention in ulcerative colitis with biologics, Dr. Sands believed the timing is more fluid. “It really is your choice and the patient’s choice,” he said. The same was true for the recent addition of the biologic vedolizumab to the ulcerative colitis armementarium, he said, noting that there is a response both during induction and maintenance phases for all biologic therapies in ulcerative colitis.
Although infections and neoplasm are concerns with all biologic therapies, Dr. Harnauer downplayed the risks when compared to corticosteroid therapy, particularly in Crohn’s disease. “The risk of neoplasia in patients given anti-TNF (anti-tumor necrosis factor) therapy disappears after adjusting for the use of azathioprine,” he said.
Dr. Sands was less sanguine about the potential risks of anti-TNF treatment, reminding the audience that the drugs carry black box warnings from the U.S. Food and Drug Administration, and that patients must be screened for their candidacy before administering immunomodulation therapy. “It is possible to develop a variety of autoimmune disorders, and immunogenicity with these drugs,” said Dr. Sands, also adding that a risk of demylenating disorders must also be accounted for. “Make sure you monitor your patients, and that you explain the risks to them.”
Regardless of when biologics are used in treatment, both presenters agreed that their exorbitant cost is an issue. “We cannot use them for every patient who walks through the door,” said Dr. Sands.
For Dr. Harnauer, the cost stands in the way of the earlier positioning of the drugs in treatment algorithms. “If we were talking about a treatment that only cost a few dollars, we wouldn’t be worrying about any of this.” Prospective pharmaco-economic studies that go beyond measuring the in-patient and surgical costs, but account for the timing and use of biologics could help strengthen the case for earlier treatment, he said.
On Twitter @whitneymcknight
EXPERT ANALYSIS FROM CCFA/IBD
H. pylori might help regulate gastric immunity
Chronic Helicobacter pylori infection suppresses interleukin-33 cytokine in the stomach, which inhibits the CD4+ T helper cell 2 (or Th2) response and may set the stage for gastric carcinoma, investigators reported online in Cellular and Molecular Gastroenterology and Hepatology.
But long-term rises in IL-33 also can trigger precancerous changes in the stomach by causing the immune system to skew excessively toward Th2 instead of Th1 immunity, said Jon Buzzelli and his associates at the University of Melbourne in Parkville, Australia.
“Despite the immune response being vastly different ... the outcome appears to be similar, as preneoplastic changes occur in both settings,” the researchers added. “In keeping with recent findings, these data suggest that H. pylori may be a beneficial organism that under certain circumstances may help to ensure that gastric immunity is tightly regulated.” (Cellular and Molecular Gastroenterology and Hepatology 2015 [http://dx.doi.org/10.1016/j.jcmgh.2014.12.003 5])
IL-33 is a member of a diverse group of cytokines called alarmins, which quickly trigger an immune response to injury or infection when released by dying cells. Fasted mice in the study that were given oral aspirin to induce gastric injury had elevated IL-33 expression, compared with controls 4 hours later, the investigators reported. “This demonstrates that IL-33 responds immediately to gastric insult through relocalization and transcriptional changes, and may be involved in gastric wound healing,” they added.
The researchers also found that gastric IL-33 levels in mice rose fourfold just 1 day after they were infected with H. pylori. But 2 months later, the mice’s IL-33 levels had fallen below those of controls, resembling the researchers’ comparison of H. pylori–positive and uninfected human stomach specimens, they said.
Furthermore, the drop in IL-33 caused a tilt away from Th2 toward Th1 immunity, which appeared to trigger precancerous changes to the gastric mucosa, the researchers added. “The inhibition of gastric IL-33 in response to chronic H. pylori infection may be a key event in gastric cancer progression,” they concluded.
But eliminating H. pylori might also cause problems in some cases, the findings suggested. The declining prevalence of helicobacteriosis in humans has accompanied a rise in Barrett’s esophagus, which precedes esophageal cancer, they noted. When the researchers administered extra IL-33 to mice – mimicking the absence of H. pylori – the cardia of their stomachs expanded and became markedly metaplastic, which precedes Barrett’s esophagus in humans, they noted. H. pylori might help prevent Barrett’s esophagus by suppressing IL-33 and thereby regulating Th2 immunity, they concluded.
The researchers also found that mice born and reared in a pathogen-free environment had lower IL-33 levels, compared with those in conventional housing, they said. Gastric IL-33 expression appeared to rise as bacterial load and diversity increased, supporting the idea that IL-33 functions in this setting as an alarmin, they added.
The investigators also examined whether the TFF2 gene – which helps regulate homeostasis in mucus cells – promoted IL-33 expression in the stomach, as it does in the lungs. Indeed, TFF2 knockout mice had about 40% less gastric IL-33, compared with wild-type mice, they said. Humans with chronic helicobacteriosis also have low TFF2 expression, which continues to fall as gastric cancer grows, they noted.
The study was funded by the Victorian Government’s Operational Infrastructure Support Program and National Health & Medical Research Council, Australia. The researchers reported no conflicts of interest.
Gastric adenocarcinoma is the second-leading cause of cancer-related death worldwide, and chronic infection with Helicobacter pylori is the strongest known risk factor for the development of this malignancy. H. pylori colonization rates hover around 80%-90% in developing countries, but only a fraction of infected individuals ever develop disease. It is increasingly apparent that gastric carcinogenesis is multifactorial, influenced by host responses, H. pylori virulence, and environmental cofactors.
Parasitic helminth infections among H. pylori-infected individuals have been associated with a lower risk for the development of gastric cancer, and experimental data from animal models of Helicobacter infection have demonstrated that concurrent helminth infection attenuates the host immune response and reduces gastric atrophy. Infection with H. pylori typically induces a Th1-polarized immune response, while helminths drive Th2 responses.
Concurrent infections with helminths is endemic in regions of some developing countries that have a high prevalence of H. pylori infection, but a lower than expected rate of gastric cancer. Buzzelli et al. provide fresh insights into the role that IL-33 plays in polarizing Th2 immune responses by demonstrating that chronic, but not acute, H. pylori infection suppresses IL-33, which ultimately leads to a predominant Th1 response. These findings may represent a novel mechanism (e.g., manipulation of IL-33) explaining why populations harboring concurrent helminth and H. pylori infection have a reduced risk of gastric cancer.
Jennifer M. Noto, Ph.D., and Richard M. Peek Jr., M.D., AGAF, of the department of medicine, division of gastroenterology, hepatology, and department of nutrition and cancer biology, Vanderbilt University, Nashville, Tenn. Dr. Noto and Dr. Peek declared that no conflict of interest exists. They acknowledge the following funding sources: NIH R01CA077955, R01DK058587, P01CA116087, and P30DK058404.
Gastric adenocarcinoma is the second-leading cause of cancer-related death worldwide, and chronic infection with Helicobacter pylori is the strongest known risk factor for the development of this malignancy. H. pylori colonization rates hover around 80%-90% in developing countries, but only a fraction of infected individuals ever develop disease. It is increasingly apparent that gastric carcinogenesis is multifactorial, influenced by host responses, H. pylori virulence, and environmental cofactors.
Parasitic helminth infections among H. pylori-infected individuals have been associated with a lower risk for the development of gastric cancer, and experimental data from animal models of Helicobacter infection have demonstrated that concurrent helminth infection attenuates the host immune response and reduces gastric atrophy. Infection with H. pylori typically induces a Th1-polarized immune response, while helminths drive Th2 responses.
Concurrent infections with helminths is endemic in regions of some developing countries that have a high prevalence of H. pylori infection, but a lower than expected rate of gastric cancer. Buzzelli et al. provide fresh insights into the role that IL-33 plays in polarizing Th2 immune responses by demonstrating that chronic, but not acute, H. pylori infection suppresses IL-33, which ultimately leads to a predominant Th1 response. These findings may represent a novel mechanism (e.g., manipulation of IL-33) explaining why populations harboring concurrent helminth and H. pylori infection have a reduced risk of gastric cancer.
Jennifer M. Noto, Ph.D., and Richard M. Peek Jr., M.D., AGAF, of the department of medicine, division of gastroenterology, hepatology, and department of nutrition and cancer biology, Vanderbilt University, Nashville, Tenn. Dr. Noto and Dr. Peek declared that no conflict of interest exists. They acknowledge the following funding sources: NIH R01CA077955, R01DK058587, P01CA116087, and P30DK058404.
Gastric adenocarcinoma is the second-leading cause of cancer-related death worldwide, and chronic infection with Helicobacter pylori is the strongest known risk factor for the development of this malignancy. H. pylori colonization rates hover around 80%-90% in developing countries, but only a fraction of infected individuals ever develop disease. It is increasingly apparent that gastric carcinogenesis is multifactorial, influenced by host responses, H. pylori virulence, and environmental cofactors.
Parasitic helminth infections among H. pylori-infected individuals have been associated with a lower risk for the development of gastric cancer, and experimental data from animal models of Helicobacter infection have demonstrated that concurrent helminth infection attenuates the host immune response and reduces gastric atrophy. Infection with H. pylori typically induces a Th1-polarized immune response, while helminths drive Th2 responses.
Concurrent infections with helminths is endemic in regions of some developing countries that have a high prevalence of H. pylori infection, but a lower than expected rate of gastric cancer. Buzzelli et al. provide fresh insights into the role that IL-33 plays in polarizing Th2 immune responses by demonstrating that chronic, but not acute, H. pylori infection suppresses IL-33, which ultimately leads to a predominant Th1 response. These findings may represent a novel mechanism (e.g., manipulation of IL-33) explaining why populations harboring concurrent helminth and H. pylori infection have a reduced risk of gastric cancer.
Jennifer M. Noto, Ph.D., and Richard M. Peek Jr., M.D., AGAF, of the department of medicine, division of gastroenterology, hepatology, and department of nutrition and cancer biology, Vanderbilt University, Nashville, Tenn. Dr. Noto and Dr. Peek declared that no conflict of interest exists. They acknowledge the following funding sources: NIH R01CA077955, R01DK058587, P01CA116087, and P30DK058404.
Chronic Helicobacter pylori infection suppresses interleukin-33 cytokine in the stomach, which inhibits the CD4+ T helper cell 2 (or Th2) response and may set the stage for gastric carcinoma, investigators reported online in Cellular and Molecular Gastroenterology and Hepatology.
But long-term rises in IL-33 also can trigger precancerous changes in the stomach by causing the immune system to skew excessively toward Th2 instead of Th1 immunity, said Jon Buzzelli and his associates at the University of Melbourne in Parkville, Australia.
“Despite the immune response being vastly different ... the outcome appears to be similar, as preneoplastic changes occur in both settings,” the researchers added. “In keeping with recent findings, these data suggest that H. pylori may be a beneficial organism that under certain circumstances may help to ensure that gastric immunity is tightly regulated.” (Cellular and Molecular Gastroenterology and Hepatology 2015 [http://dx.doi.org/10.1016/j.jcmgh.2014.12.003 5])
IL-33 is a member of a diverse group of cytokines called alarmins, which quickly trigger an immune response to injury or infection when released by dying cells. Fasted mice in the study that were given oral aspirin to induce gastric injury had elevated IL-33 expression, compared with controls 4 hours later, the investigators reported. “This demonstrates that IL-33 responds immediately to gastric insult through relocalization and transcriptional changes, and may be involved in gastric wound healing,” they added.
The researchers also found that gastric IL-33 levels in mice rose fourfold just 1 day after they were infected with H. pylori. But 2 months later, the mice’s IL-33 levels had fallen below those of controls, resembling the researchers’ comparison of H. pylori–positive and uninfected human stomach specimens, they said.
Furthermore, the drop in IL-33 caused a tilt away from Th2 toward Th1 immunity, which appeared to trigger precancerous changes to the gastric mucosa, the researchers added. “The inhibition of gastric IL-33 in response to chronic H. pylori infection may be a key event in gastric cancer progression,” they concluded.
But eliminating H. pylori might also cause problems in some cases, the findings suggested. The declining prevalence of helicobacteriosis in humans has accompanied a rise in Barrett’s esophagus, which precedes esophageal cancer, they noted. When the researchers administered extra IL-33 to mice – mimicking the absence of H. pylori – the cardia of their stomachs expanded and became markedly metaplastic, which precedes Barrett’s esophagus in humans, they noted. H. pylori might help prevent Barrett’s esophagus by suppressing IL-33 and thereby regulating Th2 immunity, they concluded.
The researchers also found that mice born and reared in a pathogen-free environment had lower IL-33 levels, compared with those in conventional housing, they said. Gastric IL-33 expression appeared to rise as bacterial load and diversity increased, supporting the idea that IL-33 functions in this setting as an alarmin, they added.
The investigators also examined whether the TFF2 gene – which helps regulate homeostasis in mucus cells – promoted IL-33 expression in the stomach, as it does in the lungs. Indeed, TFF2 knockout mice had about 40% less gastric IL-33, compared with wild-type mice, they said. Humans with chronic helicobacteriosis also have low TFF2 expression, which continues to fall as gastric cancer grows, they noted.
The study was funded by the Victorian Government’s Operational Infrastructure Support Program and National Health & Medical Research Council, Australia. The researchers reported no conflicts of interest.
Chronic Helicobacter pylori infection suppresses interleukin-33 cytokine in the stomach, which inhibits the CD4+ T helper cell 2 (or Th2) response and may set the stage for gastric carcinoma, investigators reported online in Cellular and Molecular Gastroenterology and Hepatology.
But long-term rises in IL-33 also can trigger precancerous changes in the stomach by causing the immune system to skew excessively toward Th2 instead of Th1 immunity, said Jon Buzzelli and his associates at the University of Melbourne in Parkville, Australia.
“Despite the immune response being vastly different ... the outcome appears to be similar, as preneoplastic changes occur in both settings,” the researchers added. “In keeping with recent findings, these data suggest that H. pylori may be a beneficial organism that under certain circumstances may help to ensure that gastric immunity is tightly regulated.” (Cellular and Molecular Gastroenterology and Hepatology 2015 [http://dx.doi.org/10.1016/j.jcmgh.2014.12.003 5])
IL-33 is a member of a diverse group of cytokines called alarmins, which quickly trigger an immune response to injury or infection when released by dying cells. Fasted mice in the study that were given oral aspirin to induce gastric injury had elevated IL-33 expression, compared with controls 4 hours later, the investigators reported. “This demonstrates that IL-33 responds immediately to gastric insult through relocalization and transcriptional changes, and may be involved in gastric wound healing,” they added.
The researchers also found that gastric IL-33 levels in mice rose fourfold just 1 day after they were infected with H. pylori. But 2 months later, the mice’s IL-33 levels had fallen below those of controls, resembling the researchers’ comparison of H. pylori–positive and uninfected human stomach specimens, they said.
Furthermore, the drop in IL-33 caused a tilt away from Th2 toward Th1 immunity, which appeared to trigger precancerous changes to the gastric mucosa, the researchers added. “The inhibition of gastric IL-33 in response to chronic H. pylori infection may be a key event in gastric cancer progression,” they concluded.
But eliminating H. pylori might also cause problems in some cases, the findings suggested. The declining prevalence of helicobacteriosis in humans has accompanied a rise in Barrett’s esophagus, which precedes esophageal cancer, they noted. When the researchers administered extra IL-33 to mice – mimicking the absence of H. pylori – the cardia of their stomachs expanded and became markedly metaplastic, which precedes Barrett’s esophagus in humans, they noted. H. pylori might help prevent Barrett’s esophagus by suppressing IL-33 and thereby regulating Th2 immunity, they concluded.
The researchers also found that mice born and reared in a pathogen-free environment had lower IL-33 levels, compared with those in conventional housing, they said. Gastric IL-33 expression appeared to rise as bacterial load and diversity increased, supporting the idea that IL-33 functions in this setting as an alarmin, they added.
The investigators also examined whether the TFF2 gene – which helps regulate homeostasis in mucus cells – promoted IL-33 expression in the stomach, as it does in the lungs. Indeed, TFF2 knockout mice had about 40% less gastric IL-33, compared with wild-type mice, they said. Humans with chronic helicobacteriosis also have low TFF2 expression, which continues to fall as gastric cancer grows, they noted.
The study was funded by the Victorian Government’s Operational Infrastructure Support Program and National Health & Medical Research Council, Australia. The researchers reported no conflicts of interest.
FROM CELLULAR AND MOLECULAR GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: H. pylori may help regulate gastric immunity in some circumstances.
Major finding: Chronic gastric H. pylori infection lowered expression of IL-33, a cytokine that helps activate the CD4+ T helper cell 2 response.
Data source: Immunofluorescence, flow cytometry, and quantitative real-time polymerase chain reaction studies of tissue specimens from humans and mice.Disclosures: The study was funded by the Victorian Government’s Operational Infrastructure Support Program and NH&MRC Australia. The researchers reported no conflicts of interest.
C. difficile burden in U.S. documented in 2011 estimates of infections, deaths
The estimated 453,000 infections and 29,300 deaths caused by Clostridium difficile in the United States in 2011 underline the importance of appropriate use of antibiotics and rigorous infection control measures in health care settings, Dr. Michael Bell, an official at the Centers for Disease Control and Prevention, said during a CDC telebriefing.
“To reduce the majority of C. difficile infections, we will need to improve how antibiotics are being prescribed in hospitals and throughout health care,” said Dr. Bell, deputy director of the division of health care quality promotion, at the CDC’s National Center for Emerging and Zoonotic Infectious Diseases. Early diagnosis is also critical to prevent spread of C. difficile. Many infections are community acquired, and “it is essential that patients and their clinicians be aware that they need to take any diarrhea following antibiotic use very seriously,” he added.
The briefing was held to discuss the results and implications of the study published in the New England Journal of Medicine, which was supported by the CDC and the Emerging Infections Program (EIP) Cooperative Agreement between the 10 EIP sites and the CDC. In the study of 10 geographic regions in the United States in 2011, 15,461 cases were confirmed, with the estimated incidence of the infection being 453,000 (95% confidence interval, 397,100-508,500) after predictors of incidence were adjusted for, the investigators found. The estimated number of deaths from C. difficile was 29,300 (95% CI, 16,500-42,100).
Estimates for disease incidence were higher among women, whites, and patients 65 years of age or older, wrote Dr. Fernanda Lessa of the CDC, and her associates. Future efforts should focus on antibiotic use, the proper management of which may be effective in decreasing infection rates, the authors said in the report.
“Antibiotics clearly are driving this whole epidemic,” one of the study authors, Dr. Clifford McDonald of the CDC, said during the briefing. The epidemic strain in the United States, which emerged in 2000 in Pittsburgh and Montreal, is now spread globally, and accounted for about 30% of cases in this study, he added. It is transmitted more easily than other strains and causes more severe disease.
Dr. Bell said that to reduce the rate of these infections, antibiotics should be used only when needed and for as long as necessary, “and to ensure rigorous infection control in all health care settings.” The CDC’s National Strategy to Combat Antibiotic Resistant Bacteria has the potential to reduce C. difficile infections by 50%, he added.
“There’s no room for error” when infection control is considered, he added, pointing out that hand sanitizers do not kill C. difficile spores, which spread easily and are durable, “so that any breach in correct glove use, hand hygiene, or cleaning protocol can allow the spores to spread.”
The study also estimated that more than 150,000 infections were community acquired, with no documentation of inpatient exposure in the hospital. “Nonetheless, as we showed in another recent CDC study, 80% of patients with community-associated C. difficile infections did, in fact, have contact with a health care setting like a doctor’s office or a dental clinic,” generally during the 3-month period before being diagnosed, and most of the patients had also been treated with antibiotics, Dr. Bell said.
Future efforts should focus on antibiotic use, which may be effective in decreasing infection rates, Dr. Fernanda C. Lessa and her coauthors at the Centers for Disease Control and Prevention reported in The New England Journal of Medicine.
In a study of 10 geographic regions in the United States in 2011, 15,461 cases were confirmed, with the estimated incidence of the infection being 453,000 (95% confidence interval, 397,100-508,500) after predictors of incidence were adjusted for, the investigators found. The estimated number of deaths from C. difficile was 29,300 (95% CI, 16,500-42,100). Estimates for disease incidence were higher among women, whites, and patients 65 years of age or older, wrote Dr. Lessa and associates.
National efforts to address the increase in C. difficile infections include the requirement since 2013 that hospitals participating in the Centers for Medicare & Medicaid Services’ Hospital Inpatient Quality Reporting Program data on C. difficile infections to the CDC’s National Healthcare Safety Network, which has shown at least a 10% drop since 2013, Dr Bell said. Targets for reducing C. difficile infections in the United States by 2020 are being established in the National Action Plan to Prevent Health Care-Associated Infections: Road Map to Elimination.
Madhu Rajaraman contributed to this report.
The estimated 453,000 infections and 29,300 deaths caused by Clostridium difficile in the United States in 2011 underline the importance of appropriate use of antibiotics and rigorous infection control measures in health care settings, Dr. Michael Bell, an official at the Centers for Disease Control and Prevention, said during a CDC telebriefing.
“To reduce the majority of C. difficile infections, we will need to improve how antibiotics are being prescribed in hospitals and throughout health care,” said Dr. Bell, deputy director of the division of health care quality promotion, at the CDC’s National Center for Emerging and Zoonotic Infectious Diseases. Early diagnosis is also critical to prevent spread of C. difficile. Many infections are community acquired, and “it is essential that patients and their clinicians be aware that they need to take any diarrhea following antibiotic use very seriously,” he added.
The briefing was held to discuss the results and implications of the study published in the New England Journal of Medicine, which was supported by the CDC and the Emerging Infections Program (EIP) Cooperative Agreement between the 10 EIP sites and the CDC. In the study of 10 geographic regions in the United States in 2011, 15,461 cases were confirmed, with the estimated incidence of the infection being 453,000 (95% confidence interval, 397,100-508,500) after predictors of incidence were adjusted for, the investigators found. The estimated number of deaths from C. difficile was 29,300 (95% CI, 16,500-42,100).
Estimates for disease incidence were higher among women, whites, and patients 65 years of age or older, wrote Dr. Fernanda Lessa of the CDC, and her associates. Future efforts should focus on antibiotic use, the proper management of which may be effective in decreasing infection rates, the authors said in the report.
“Antibiotics clearly are driving this whole epidemic,” one of the study authors, Dr. Clifford McDonald of the CDC, said during the briefing. The epidemic strain in the United States, which emerged in 2000 in Pittsburgh and Montreal, is now spread globally, and accounted for about 30% of cases in this study, he added. It is transmitted more easily than other strains and causes more severe disease.
Dr. Bell said that to reduce the rate of these infections, antibiotics should be used only when needed and for as long as necessary, “and to ensure rigorous infection control in all health care settings.” The CDC’s National Strategy to Combat Antibiotic Resistant Bacteria has the potential to reduce C. difficile infections by 50%, he added.
“There’s no room for error” when infection control is considered, he added, pointing out that hand sanitizers do not kill C. difficile spores, which spread easily and are durable, “so that any breach in correct glove use, hand hygiene, or cleaning protocol can allow the spores to spread.”
The study also estimated that more than 150,000 infections were community acquired, with no documentation of inpatient exposure in the hospital. “Nonetheless, as we showed in another recent CDC study, 80% of patients with community-associated C. difficile infections did, in fact, have contact with a health care setting like a doctor’s office or a dental clinic,” generally during the 3-month period before being diagnosed, and most of the patients had also been treated with antibiotics, Dr. Bell said.
Future efforts should focus on antibiotic use, which may be effective in decreasing infection rates, Dr. Fernanda C. Lessa and her coauthors at the Centers for Disease Control and Prevention reported in The New England Journal of Medicine.
In a study of 10 geographic regions in the United States in 2011, 15,461 cases were confirmed, with the estimated incidence of the infection being 453,000 (95% confidence interval, 397,100-508,500) after predictors of incidence were adjusted for, the investigators found. The estimated number of deaths from C. difficile was 29,300 (95% CI, 16,500-42,100). Estimates for disease incidence were higher among women, whites, and patients 65 years of age or older, wrote Dr. Lessa and associates.
National efforts to address the increase in C. difficile infections include the requirement since 2013 that hospitals participating in the Centers for Medicare & Medicaid Services’ Hospital Inpatient Quality Reporting Program data on C. difficile infections to the CDC’s National Healthcare Safety Network, which has shown at least a 10% drop since 2013, Dr Bell said. Targets for reducing C. difficile infections in the United States by 2020 are being established in the National Action Plan to Prevent Health Care-Associated Infections: Road Map to Elimination.
Madhu Rajaraman contributed to this report.
The estimated 453,000 infections and 29,300 deaths caused by Clostridium difficile in the United States in 2011 underline the importance of appropriate use of antibiotics and rigorous infection control measures in health care settings, Dr. Michael Bell, an official at the Centers for Disease Control and Prevention, said during a CDC telebriefing.
“To reduce the majority of C. difficile infections, we will need to improve how antibiotics are being prescribed in hospitals and throughout health care,” said Dr. Bell, deputy director of the division of health care quality promotion, at the CDC’s National Center for Emerging and Zoonotic Infectious Diseases. Early diagnosis is also critical to prevent spread of C. difficile. Many infections are community acquired, and “it is essential that patients and their clinicians be aware that they need to take any diarrhea following antibiotic use very seriously,” he added.
The briefing was held to discuss the results and implications of the study published in the New England Journal of Medicine, which was supported by the CDC and the Emerging Infections Program (EIP) Cooperative Agreement between the 10 EIP sites and the CDC. In the study of 10 geographic regions in the United States in 2011, 15,461 cases were confirmed, with the estimated incidence of the infection being 453,000 (95% confidence interval, 397,100-508,500) after predictors of incidence were adjusted for, the investigators found. The estimated number of deaths from C. difficile was 29,300 (95% CI, 16,500-42,100).
Estimates for disease incidence were higher among women, whites, and patients 65 years of age or older, wrote Dr. Fernanda Lessa of the CDC, and her associates. Future efforts should focus on antibiotic use, the proper management of which may be effective in decreasing infection rates, the authors said in the report.
“Antibiotics clearly are driving this whole epidemic,” one of the study authors, Dr. Clifford McDonald of the CDC, said during the briefing. The epidemic strain in the United States, which emerged in 2000 in Pittsburgh and Montreal, is now spread globally, and accounted for about 30% of cases in this study, he added. It is transmitted more easily than other strains and causes more severe disease.
Dr. Bell said that to reduce the rate of these infections, antibiotics should be used only when needed and for as long as necessary, “and to ensure rigorous infection control in all health care settings.” The CDC’s National Strategy to Combat Antibiotic Resistant Bacteria has the potential to reduce C. difficile infections by 50%, he added.
“There’s no room for error” when infection control is considered, he added, pointing out that hand sanitizers do not kill C. difficile spores, which spread easily and are durable, “so that any breach in correct glove use, hand hygiene, or cleaning protocol can allow the spores to spread.”
The study also estimated that more than 150,000 infections were community acquired, with no documentation of inpatient exposure in the hospital. “Nonetheless, as we showed in another recent CDC study, 80% of patients with community-associated C. difficile infections did, in fact, have contact with a health care setting like a doctor’s office or a dental clinic,” generally during the 3-month period before being diagnosed, and most of the patients had also been treated with antibiotics, Dr. Bell said.
Future efforts should focus on antibiotic use, which may be effective in decreasing infection rates, Dr. Fernanda C. Lessa and her coauthors at the Centers for Disease Control and Prevention reported in The New England Journal of Medicine.
In a study of 10 geographic regions in the United States in 2011, 15,461 cases were confirmed, with the estimated incidence of the infection being 453,000 (95% confidence interval, 397,100-508,500) after predictors of incidence were adjusted for, the investigators found. The estimated number of deaths from C. difficile was 29,300 (95% CI, 16,500-42,100). Estimates for disease incidence were higher among women, whites, and patients 65 years of age or older, wrote Dr. Lessa and associates.
National efforts to address the increase in C. difficile infections include the requirement since 2013 that hospitals participating in the Centers for Medicare & Medicaid Services’ Hospital Inpatient Quality Reporting Program data on C. difficile infections to the CDC’s National Healthcare Safety Network, which has shown at least a 10% drop since 2013, Dr Bell said. Targets for reducing C. difficile infections in the United States by 2020 are being established in the National Action Plan to Prevent Health Care-Associated Infections: Road Map to Elimination.
Madhu Rajaraman contributed to this report.
FROM A CDC TELEBRIEFING
Emulsifiers alter gut bacteria in mice, set the stage for IBD and metabolic syndrome
The emulsifiers found in processed foods can alter the composition of gut microbiota and trigger intestinal inflammation that, in turn, may promote the development of inflammatory bowel disease and metabolic syndrome, Dr. Benoit Chassaing, PhD, of Georgia State University Institute for Biomedical Sciences, and his colleagues report Feb. 25 in Nature.
The research team fed mice polysorbate 80 and carboxymethylcellulose, two commonly used emulsifiers, at doses replicating those found in processed foods. Emulsifier consumption changed the composition of the gut microbiota in the mice, resulting in a proinflammatory state. The bacteria were able to digest and infiltrate the intestine’s mucosal layer and the altered bacterial species expressed more flagellin and lipopolysaccharide, which can activate proinflammatory gene expression by the immune system.
The bacterial changes triggered chronic colitis in mice genetically prone to the disorder. In mice with normal immune systems, low-grade intestinal inflammation resulted, and metabolic syndrome was noted.
Transplanting the microbiota from mice fed emulsifiers to germ-free mice also transferred some parameters of low-grade inflammation and metabolic syndrome, indicating a central role for the microbiota in mediating the adverse effect of emulsifiers.
The emulsifiers found in processed foods can alter the composition of gut microbiota and trigger intestinal inflammation that, in turn, may promote the development of inflammatory bowel disease and metabolic syndrome, Dr. Benoit Chassaing, PhD, of Georgia State University Institute for Biomedical Sciences, and his colleagues report Feb. 25 in Nature.
The research team fed mice polysorbate 80 and carboxymethylcellulose, two commonly used emulsifiers, at doses replicating those found in processed foods. Emulsifier consumption changed the composition of the gut microbiota in the mice, resulting in a proinflammatory state. The bacteria were able to digest and infiltrate the intestine’s mucosal layer and the altered bacterial species expressed more flagellin and lipopolysaccharide, which can activate proinflammatory gene expression by the immune system.
The bacterial changes triggered chronic colitis in mice genetically prone to the disorder. In mice with normal immune systems, low-grade intestinal inflammation resulted, and metabolic syndrome was noted.
Transplanting the microbiota from mice fed emulsifiers to germ-free mice also transferred some parameters of low-grade inflammation and metabolic syndrome, indicating a central role for the microbiota in mediating the adverse effect of emulsifiers.
The emulsifiers found in processed foods can alter the composition of gut microbiota and trigger intestinal inflammation that, in turn, may promote the development of inflammatory bowel disease and metabolic syndrome, Dr. Benoit Chassaing, PhD, of Georgia State University Institute for Biomedical Sciences, and his colleagues report Feb. 25 in Nature.
The research team fed mice polysorbate 80 and carboxymethylcellulose, two commonly used emulsifiers, at doses replicating those found in processed foods. Emulsifier consumption changed the composition of the gut microbiota in the mice, resulting in a proinflammatory state. The bacteria were able to digest and infiltrate the intestine’s mucosal layer and the altered bacterial species expressed more flagellin and lipopolysaccharide, which can activate proinflammatory gene expression by the immune system.
The bacterial changes triggered chronic colitis in mice genetically prone to the disorder. In mice with normal immune systems, low-grade intestinal inflammation resulted, and metabolic syndrome was noted.
Transplanting the microbiota from mice fed emulsifiers to germ-free mice also transferred some parameters of low-grade inflammation and metabolic syndrome, indicating a central role for the microbiota in mediating the adverse effect of emulsifiers.
FROM NATURE
New scoring system for small bowel–obstruction severity
LAKE BUENA VISTA, FLA. – A novel three-item scoring system reliably categorizes severity of small bowel obstruction and is more strongly associated with in-hospital mortality than the American Association for the Surgery of Trauma anatomic score alone.
The AAST developed a scoring system to standardize the severity of small-bowel obstruction (SBO) based on anatomic criteria. Its authors have subsequently recommended, however, that other parametersare needed that would take into consideration the entirety of the patient’s clinical situation (J. Trauma Acute Care Surg. 2014;77:705-8 and J. Trauma Acute Care Surg. 2014;76:884-7).
To that end, investigators at the Mayo Clinic in Rochester, Minn., created the Acute General Emergency Surgical Severity-Small Bowel Obstruction (AGESS-SBO) system that incorporates presenting physiology and pre-existing comorbidities with anatomic criteria.
“It’s evident that the complications and patient outcomes clearly depend on the extent of the involvement of the diseased organ, but also depend on the hosting environment, which means the patient’s physiology and pre-existing conditions,” Dr. Yaser Baghdadi explained at the annual scientific assembly of the Eastern Association for the Surgery of Trauma.
He reported a cohort study involving 377 patients who were treated for SBO at the Mayo Clinic between 2009 and 2012 and evaluated using anatomic criteria and the AGESS-SBO, which uses a 5-point scoring system for each of its three scales.
Most patients (57%) received a score of 1 on the AGESS-SBO anatomic involvement scale for a partial SBO without need of operation, while only 1% had a score of 5, indicating strangulation and perforation with diffuse peritoneal contamination.
On the physiology scale, 58.6% had no physiologic derangement or a score of 0, 36% had a score of 1 because of systemic inflammatory response syndrome, and only 1.1% had a score of 5 for multiple organ dysfunction syndrome.
A Charlson comorbid score of 1 or 2 earned 32% of patients 1 point on the comorbidity scale, while 4% had a score of 5 because of a Charlson score of 9 or more.
In all, 215 patients (57%) had nonoperative treatment and 162 patients (43%) underwent surgical exploration. The median overall AGESS-SBO score was 6 points (interquartile range [IQR], 3-13 points).
The median length of stay (LOS) was 5 days (IQR, 3-9.5 days), with 94 patients (25%) having a stay exceeding 9.5 days, Dr. Baghdadi said in the poster presentation. In-hospital complications occurred in 82 patients (22%) and eight patients (2%) died during their hospital stay.
Comparison of the areas under receiver operative characteristic curves revealed a statistically significant greater association between the AGESS-SBO score and in-hospital mortality than the AAST anatomic score (AUC, 0.79 vs. 0.55, P value = .015), reported Dr. Baghdadi, a research fellow in the Mayo Clinic’s trauma division.
The two scoring systems had comparable ability to predict in-hospital complications (AUC, 0.72 vs. 0.69; P = .42) and extended LOS (AUC, 0.72 vs. 0.74; P = .47). The lack of statistical significance favoring the AGESS-SBO may be because these outcomes would be more likely in patients requiring surgery and the analysis combined patients who did and did not require operative care, he said in an interview.
“The AGESS-SBO system is a useful tool to classify the disease severity among SBO patients compared to the AAST anatomic score alone. We are planning to run a prospective study to validate what we have found,” he added.
Dr. Baghdadi and his coauthors reported having no financial disclosures.
LAKE BUENA VISTA, FLA. – A novel three-item scoring system reliably categorizes severity of small bowel obstruction and is more strongly associated with in-hospital mortality than the American Association for the Surgery of Trauma anatomic score alone.
The AAST developed a scoring system to standardize the severity of small-bowel obstruction (SBO) based on anatomic criteria. Its authors have subsequently recommended, however, that other parametersare needed that would take into consideration the entirety of the patient’s clinical situation (J. Trauma Acute Care Surg. 2014;77:705-8 and J. Trauma Acute Care Surg. 2014;76:884-7).
To that end, investigators at the Mayo Clinic in Rochester, Minn., created the Acute General Emergency Surgical Severity-Small Bowel Obstruction (AGESS-SBO) system that incorporates presenting physiology and pre-existing comorbidities with anatomic criteria.
“It’s evident that the complications and patient outcomes clearly depend on the extent of the involvement of the diseased organ, but also depend on the hosting environment, which means the patient’s physiology and pre-existing conditions,” Dr. Yaser Baghdadi explained at the annual scientific assembly of the Eastern Association for the Surgery of Trauma.
He reported a cohort study involving 377 patients who were treated for SBO at the Mayo Clinic between 2009 and 2012 and evaluated using anatomic criteria and the AGESS-SBO, which uses a 5-point scoring system for each of its three scales.
Most patients (57%) received a score of 1 on the AGESS-SBO anatomic involvement scale for a partial SBO without need of operation, while only 1% had a score of 5, indicating strangulation and perforation with diffuse peritoneal contamination.
On the physiology scale, 58.6% had no physiologic derangement or a score of 0, 36% had a score of 1 because of systemic inflammatory response syndrome, and only 1.1% had a score of 5 for multiple organ dysfunction syndrome.
A Charlson comorbid score of 1 or 2 earned 32% of patients 1 point on the comorbidity scale, while 4% had a score of 5 because of a Charlson score of 9 or more.
In all, 215 patients (57%) had nonoperative treatment and 162 patients (43%) underwent surgical exploration. The median overall AGESS-SBO score was 6 points (interquartile range [IQR], 3-13 points).
The median length of stay (LOS) was 5 days (IQR, 3-9.5 days), with 94 patients (25%) having a stay exceeding 9.5 days, Dr. Baghdadi said in the poster presentation. In-hospital complications occurred in 82 patients (22%) and eight patients (2%) died during their hospital stay.
Comparison of the areas under receiver operative characteristic curves revealed a statistically significant greater association between the AGESS-SBO score and in-hospital mortality than the AAST anatomic score (AUC, 0.79 vs. 0.55, P value = .015), reported Dr. Baghdadi, a research fellow in the Mayo Clinic’s trauma division.
The two scoring systems had comparable ability to predict in-hospital complications (AUC, 0.72 vs. 0.69; P = .42) and extended LOS (AUC, 0.72 vs. 0.74; P = .47). The lack of statistical significance favoring the AGESS-SBO may be because these outcomes would be more likely in patients requiring surgery and the analysis combined patients who did and did not require operative care, he said in an interview.
“The AGESS-SBO system is a useful tool to classify the disease severity among SBO patients compared to the AAST anatomic score alone. We are planning to run a prospective study to validate what we have found,” he added.
Dr. Baghdadi and his coauthors reported having no financial disclosures.
LAKE BUENA VISTA, FLA. – A novel three-item scoring system reliably categorizes severity of small bowel obstruction and is more strongly associated with in-hospital mortality than the American Association for the Surgery of Trauma anatomic score alone.
The AAST developed a scoring system to standardize the severity of small-bowel obstruction (SBO) based on anatomic criteria. Its authors have subsequently recommended, however, that other parametersare needed that would take into consideration the entirety of the patient’s clinical situation (J. Trauma Acute Care Surg. 2014;77:705-8 and J. Trauma Acute Care Surg. 2014;76:884-7).
To that end, investigators at the Mayo Clinic in Rochester, Minn., created the Acute General Emergency Surgical Severity-Small Bowel Obstruction (AGESS-SBO) system that incorporates presenting physiology and pre-existing comorbidities with anatomic criteria.
“It’s evident that the complications and patient outcomes clearly depend on the extent of the involvement of the diseased organ, but also depend on the hosting environment, which means the patient’s physiology and pre-existing conditions,” Dr. Yaser Baghdadi explained at the annual scientific assembly of the Eastern Association for the Surgery of Trauma.
He reported a cohort study involving 377 patients who were treated for SBO at the Mayo Clinic between 2009 and 2012 and evaluated using anatomic criteria and the AGESS-SBO, which uses a 5-point scoring system for each of its three scales.
Most patients (57%) received a score of 1 on the AGESS-SBO anatomic involvement scale for a partial SBO without need of operation, while only 1% had a score of 5, indicating strangulation and perforation with diffuse peritoneal contamination.
On the physiology scale, 58.6% had no physiologic derangement or a score of 0, 36% had a score of 1 because of systemic inflammatory response syndrome, and only 1.1% had a score of 5 for multiple organ dysfunction syndrome.
A Charlson comorbid score of 1 or 2 earned 32% of patients 1 point on the comorbidity scale, while 4% had a score of 5 because of a Charlson score of 9 or more.
In all, 215 patients (57%) had nonoperative treatment and 162 patients (43%) underwent surgical exploration. The median overall AGESS-SBO score was 6 points (interquartile range [IQR], 3-13 points).
The median length of stay (LOS) was 5 days (IQR, 3-9.5 days), with 94 patients (25%) having a stay exceeding 9.5 days, Dr. Baghdadi said in the poster presentation. In-hospital complications occurred in 82 patients (22%) and eight patients (2%) died during their hospital stay.
Comparison of the areas under receiver operative characteristic curves revealed a statistically significant greater association between the AGESS-SBO score and in-hospital mortality than the AAST anatomic score (AUC, 0.79 vs. 0.55, P value = .015), reported Dr. Baghdadi, a research fellow in the Mayo Clinic’s trauma division.
The two scoring systems had comparable ability to predict in-hospital complications (AUC, 0.72 vs. 0.69; P = .42) and extended LOS (AUC, 0.72 vs. 0.74; P = .47). The lack of statistical significance favoring the AGESS-SBO may be because these outcomes would be more likely in patients requiring surgery and the analysis combined patients who did and did not require operative care, he said in an interview.
“The AGESS-SBO system is a useful tool to classify the disease severity among SBO patients compared to the AAST anatomic score alone. We are planning to run a prospective study to validate what we have found,” he added.
Dr. Baghdadi and his coauthors reported having no financial disclosures.
AT THE EAST SCIENTIFIC ASSEMBLY
Key clinical point: Adding presenting physiology and comorbidities to anatomic criteria provides a reliable tool to categorize severity of small-bowel obstruction.
Major finding: The AGESS-SBO score was significantly associated with in-hospital mortality, versus the AAST anatomic score (area under ROC curves: 0.79 vs. 0.55; P = .015).
Data source: A cohort study of 377 patients treated for small-bowel obstruction.
Disclosures: Dr. Baghdadi and his coauthors reported having no financial disclosures.
FDA allows marketing of vaginally inserted device for fecal incontinence
A vaginally inserted device that uses a balloon inflated by the patient to occlude the rectum has been cleared for marketing as a treatment for fecal incontinence in adult women, the Food and Drug Administration announced on Feb. 12.
The device is indicated for women aged 18-75 years who have had at least four episodes of fecal incontinence (FI) in a 2-week period, according to the FDA’s statement. The device includes a vaginal insert, placed in the same area as a tampon, and a pump that inflates and deflates a balloon on the insert. When inflated by the woman, the balloon “exerts pressure through the vaginal wall onto the rectal area, thereby reducing the number of FI episodes,” the statement said. The first time it is used, the clinician fits the device and inflates the balloon; subsequently, a patient deflates and inflates the device on her own, as needed. It will be marketed as the Eclipse System, by Pelvalon.
In the statement, Dr. William Maisel, deputy director for science and chief scientist in the FDA’s Center for Devices and Radiological Health, pointed out that treatment options for fecal incontinence currently include drugs, dietary changes, exercise, and surgery, and that the Eclipse System “provides an additional treatment option for women who suffer from this condition.”
Like many devices, this device did not go through the full approval process but was reviewed by the FDA through the “de novo classification process,” a pathway for making available some devices that have low to moderate risk and are not considered substantially equivalent to another available device.
The FDA reviewed nonclinical testing data and the main study that evaluated the device in 61 women with FI. One month after they started using the device, the number of FI episodes dropped by 50% in almost 80% of the women in the trial, according to the FDA statement. Pelvic cramping and discomfort; pelvic pain; vaginal abrasion, redness, or discharge; and urinary incontinence were among the adverse events associated with the device; all adverse events were mild or moderate. The LIFE study is scheduled to be published in the March 2015 issue of Obstetrics and Gynecology.
In a statement, the company said that it plans to release the Eclipse System later in 2015.
A vaginally inserted device that uses a balloon inflated by the patient to occlude the rectum has been cleared for marketing as a treatment for fecal incontinence in adult women, the Food and Drug Administration announced on Feb. 12.
The device is indicated for women aged 18-75 years who have had at least four episodes of fecal incontinence (FI) in a 2-week period, according to the FDA’s statement. The device includes a vaginal insert, placed in the same area as a tampon, and a pump that inflates and deflates a balloon on the insert. When inflated by the woman, the balloon “exerts pressure through the vaginal wall onto the rectal area, thereby reducing the number of FI episodes,” the statement said. The first time it is used, the clinician fits the device and inflates the balloon; subsequently, a patient deflates and inflates the device on her own, as needed. It will be marketed as the Eclipse System, by Pelvalon.
In the statement, Dr. William Maisel, deputy director for science and chief scientist in the FDA’s Center for Devices and Radiological Health, pointed out that treatment options for fecal incontinence currently include drugs, dietary changes, exercise, and surgery, and that the Eclipse System “provides an additional treatment option for women who suffer from this condition.”
Like many devices, this device did not go through the full approval process but was reviewed by the FDA through the “de novo classification process,” a pathway for making available some devices that have low to moderate risk and are not considered substantially equivalent to another available device.
The FDA reviewed nonclinical testing data and the main study that evaluated the device in 61 women with FI. One month after they started using the device, the number of FI episodes dropped by 50% in almost 80% of the women in the trial, according to the FDA statement. Pelvic cramping and discomfort; pelvic pain; vaginal abrasion, redness, or discharge; and urinary incontinence were among the adverse events associated with the device; all adverse events were mild or moderate. The LIFE study is scheduled to be published in the March 2015 issue of Obstetrics and Gynecology.
In a statement, the company said that it plans to release the Eclipse System later in 2015.
A vaginally inserted device that uses a balloon inflated by the patient to occlude the rectum has been cleared for marketing as a treatment for fecal incontinence in adult women, the Food and Drug Administration announced on Feb. 12.
The device is indicated for women aged 18-75 years who have had at least four episodes of fecal incontinence (FI) in a 2-week period, according to the FDA’s statement. The device includes a vaginal insert, placed in the same area as a tampon, and a pump that inflates and deflates a balloon on the insert. When inflated by the woman, the balloon “exerts pressure through the vaginal wall onto the rectal area, thereby reducing the number of FI episodes,” the statement said. The first time it is used, the clinician fits the device and inflates the balloon; subsequently, a patient deflates and inflates the device on her own, as needed. It will be marketed as the Eclipse System, by Pelvalon.
In the statement, Dr. William Maisel, deputy director for science and chief scientist in the FDA’s Center for Devices and Radiological Health, pointed out that treatment options for fecal incontinence currently include drugs, dietary changes, exercise, and surgery, and that the Eclipse System “provides an additional treatment option for women who suffer from this condition.”
Like many devices, this device did not go through the full approval process but was reviewed by the FDA through the “de novo classification process,” a pathway for making available some devices that have low to moderate risk and are not considered substantially equivalent to another available device.
The FDA reviewed nonclinical testing data and the main study that evaluated the device in 61 women with FI. One month after they started using the device, the number of FI episodes dropped by 50% in almost 80% of the women in the trial, according to the FDA statement. Pelvic cramping and discomfort; pelvic pain; vaginal abrasion, redness, or discharge; and urinary incontinence were among the adverse events associated with the device; all adverse events were mild or moderate. The LIFE study is scheduled to be published in the March 2015 issue of Obstetrics and Gynecology.
In a statement, the company said that it plans to release the Eclipse System later in 2015.
New drugs help but IBD questions remain
ORLANDO– Independent investigators eager to author studies that have notable impact on the field of inflammatory bowel disease have plenty of important paths to pursue, according to Dr. Jean-Frederic Colombel, a professor of gastroenterology at Mt. Sinai Hospital in New York.
“Over the next 3-5 years, we will have a lot of new drugs, which is nice, but we don’t yet know how to use them, or which ones to use,” Dr. Colombel said during a presentation on the future of the field at this year’s meeting of the annual Advances in Inflammatory Bowel Disease, sponsored by the Crohn’s and Colitis Foundation of America and endorsed by the American Gastroenterological Association.
A host of biologics including vedolizumab, recently indicated by the U.S. Food and Drug Administration for ulcerative colitis and Crohn’s disease, and ustekinumab, currently in phase III studies for Crohn’s disease, are set to revolutionize treatment at a time when the field is already undergoing great change, according to Dr. Colombel. The result, he says, is that despite “formidable” challenges in recruitment and funding, “huge opportunities” exist for investigators willing to collaborate and be creative.
Studies that elucidate the natural history of Crohn’s disease and colitis offer insight into the efficacy of various treatment strategies, help determine whether to target symptoms or biomarkers, and answer whether combination therapies are safe and effective in certain patients are what Dr. Colombel says he hopes will help improve the field as the drug pipeline continues to grow.
He stipulated a caveat, however, “I strongly believe we need some new study designs.” Although he noted that trials needn’t be complicated, they should be long enough to collect sorely needed prospective data. “These can only be done in investigator-initiated trials because of the time frames,” Dr. Colombel urged the audience of young investigators.
A fieldwide shift in thinking about ulcerative colitis and Crohn’s disease as chronic, progressive diseases, rather than intermittent afflictions has already helped generate new study endpoints such as the Lémann Score, an index of progressive bowel damage that allows researchers to better track the history of IBD in patients, and thus provide a window of opportunity for interventions, said Dr. Colombel.
To wit, the CURE study, conducted by the French IBD society GITAID, is a 5-year, prospective study of patients whose early Crohn’s disease is treated with the biologic adalimumab. The Lémann Score is used to screen patients at the end of each successive year, in order to adjust their treatment to reach the final endpoint of deep remission by year 5 when several indicators are measured, including bowel image, level of disability, and whether there was the need for surgery. Dr. Colombel said the novel design of the study, which has already recruited 60 patients, shows it is “feasible” to collect longitudinal data.
The field also has a responsibility to conduct randomly assigned controlled trials to demonstrate the comparative effectiveness, safety, immunogenicity, and cost benefits of the rapidly emerging spate of biosimilars, particularly since there is not global agreement about their use, according to Dr. Colombel. Their approval for IBD in Europe is “highly disputable.... I think this could be a very nice topic for an investigator-initiated trial, in Canada and the U.S.”
Head-to-head trials that are well designed will help answer “very important questions” about which treatment strategies have the best outcomes, but doing so requires fortitude, according to Dr. Colombel. “The Dutch have the guts to conduct the LIRIC trial,” he said. In it, patients with Crohn’s disease in the terminal ileum, who have failed steroids or immune therapy, will be assigned randomly to either laparoscopic ileocolic resection or infliximab, the first available biosimilar. Sixty participants have enrolled to date, he said.
These types of studies could also help delineate how best to employ combination therapies. “What I propose for this kind of study is an intensive therapy combining biologics very early in patients with bad prognoses, and looking at the long-term outcome using bowel image,” Dr. Colombel said. These data, and others indicating the most appropriate length of treatment in patients with varying states of disease, are Dr. Colombel’s personal “top choice” for investigation. The excitement that personalized medicine has engendered across the specialities has so far not resulted in specific, validated treatments for IBD patients. However, the question of how to use personal characteristics and serologic and genetic markers to create predictive models for which patients will need either step-up or top-down therapies, accounting for their individual risk of complication, is what he said is among the most “important [question] we need to answer because the choice of early therapy will depend on this predictor,” according to Dr. Colombel.
The question of which treatment targets are best remains unclear but is important to decipher, according to Dr. Colombel, who said simple studies comparing outcomes when patients are treated to symptoms vs. treated to biomarker measurements are needed, as well as the importance of mucosal healing vs. symptom improvement. “This is important because there is discordance between endoscopy and histology,” said Dr. Colombel. “Persistent histologic inflammation is frequently associated with bad outcomes.”
However, endoscopic scoring itself is another area Dr. Colombel said is worth investigating, especially when it comes to validating endpoints such as those in the Crohn’s Disease Index of Severity (CDEIS), and Simple Endoscopic Score for Crohn’s Disease (SES-CD), which will help determine remission cutoffs. Also, endoscopic scoring in comparison with live video and imaging still need standardized approaches. “When you see how it is done in the U.S., it is generally very poor,” Dr. Colombel said.
The biggest obstacles of all when it comes to independent investigation in the United States, according to Frenchman Dr. Colombel, is heavy regulation and cost. “In France, [conducting trials] was considered part of my job. I was not compensated. It is different here.”
Dr. Colombel has numerous financial ties to the pharmaceutical industry, including AB Science, Amgen, Baxter, Bristol-Meyers Squibb, and Merck, among several others.
On Twitter @whitneymcknight
ORLANDO– Independent investigators eager to author studies that have notable impact on the field of inflammatory bowel disease have plenty of important paths to pursue, according to Dr. Jean-Frederic Colombel, a professor of gastroenterology at Mt. Sinai Hospital in New York.
“Over the next 3-5 years, we will have a lot of new drugs, which is nice, but we don’t yet know how to use them, or which ones to use,” Dr. Colombel said during a presentation on the future of the field at this year’s meeting of the annual Advances in Inflammatory Bowel Disease, sponsored by the Crohn’s and Colitis Foundation of America and endorsed by the American Gastroenterological Association.
A host of biologics including vedolizumab, recently indicated by the U.S. Food and Drug Administration for ulcerative colitis and Crohn’s disease, and ustekinumab, currently in phase III studies for Crohn’s disease, are set to revolutionize treatment at a time when the field is already undergoing great change, according to Dr. Colombel. The result, he says, is that despite “formidable” challenges in recruitment and funding, “huge opportunities” exist for investigators willing to collaborate and be creative.
Studies that elucidate the natural history of Crohn’s disease and colitis offer insight into the efficacy of various treatment strategies, help determine whether to target symptoms or biomarkers, and answer whether combination therapies are safe and effective in certain patients are what Dr. Colombel says he hopes will help improve the field as the drug pipeline continues to grow.
He stipulated a caveat, however, “I strongly believe we need some new study designs.” Although he noted that trials needn’t be complicated, they should be long enough to collect sorely needed prospective data. “These can only be done in investigator-initiated trials because of the time frames,” Dr. Colombel urged the audience of young investigators.
A fieldwide shift in thinking about ulcerative colitis and Crohn’s disease as chronic, progressive diseases, rather than intermittent afflictions has already helped generate new study endpoints such as the Lémann Score, an index of progressive bowel damage that allows researchers to better track the history of IBD in patients, and thus provide a window of opportunity for interventions, said Dr. Colombel.
To wit, the CURE study, conducted by the French IBD society GITAID, is a 5-year, prospective study of patients whose early Crohn’s disease is treated with the biologic adalimumab. The Lémann Score is used to screen patients at the end of each successive year, in order to adjust their treatment to reach the final endpoint of deep remission by year 5 when several indicators are measured, including bowel image, level of disability, and whether there was the need for surgery. Dr. Colombel said the novel design of the study, which has already recruited 60 patients, shows it is “feasible” to collect longitudinal data.
The field also has a responsibility to conduct randomly assigned controlled trials to demonstrate the comparative effectiveness, safety, immunogenicity, and cost benefits of the rapidly emerging spate of biosimilars, particularly since there is not global agreement about their use, according to Dr. Colombel. Their approval for IBD in Europe is “highly disputable.... I think this could be a very nice topic for an investigator-initiated trial, in Canada and the U.S.”
Head-to-head trials that are well designed will help answer “very important questions” about which treatment strategies have the best outcomes, but doing so requires fortitude, according to Dr. Colombel. “The Dutch have the guts to conduct the LIRIC trial,” he said. In it, patients with Crohn’s disease in the terminal ileum, who have failed steroids or immune therapy, will be assigned randomly to either laparoscopic ileocolic resection or infliximab, the first available biosimilar. Sixty participants have enrolled to date, he said.
These types of studies could also help delineate how best to employ combination therapies. “What I propose for this kind of study is an intensive therapy combining biologics very early in patients with bad prognoses, and looking at the long-term outcome using bowel image,” Dr. Colombel said. These data, and others indicating the most appropriate length of treatment in patients with varying states of disease, are Dr. Colombel’s personal “top choice” for investigation. The excitement that personalized medicine has engendered across the specialities has so far not resulted in specific, validated treatments for IBD patients. However, the question of how to use personal characteristics and serologic and genetic markers to create predictive models for which patients will need either step-up or top-down therapies, accounting for their individual risk of complication, is what he said is among the most “important [question] we need to answer because the choice of early therapy will depend on this predictor,” according to Dr. Colombel.
The question of which treatment targets are best remains unclear but is important to decipher, according to Dr. Colombel, who said simple studies comparing outcomes when patients are treated to symptoms vs. treated to biomarker measurements are needed, as well as the importance of mucosal healing vs. symptom improvement. “This is important because there is discordance between endoscopy and histology,” said Dr. Colombel. “Persistent histologic inflammation is frequently associated with bad outcomes.”
However, endoscopic scoring itself is another area Dr. Colombel said is worth investigating, especially when it comes to validating endpoints such as those in the Crohn’s Disease Index of Severity (CDEIS), and Simple Endoscopic Score for Crohn’s Disease (SES-CD), which will help determine remission cutoffs. Also, endoscopic scoring in comparison with live video and imaging still need standardized approaches. “When you see how it is done in the U.S., it is generally very poor,” Dr. Colombel said.
The biggest obstacles of all when it comes to independent investigation in the United States, according to Frenchman Dr. Colombel, is heavy regulation and cost. “In France, [conducting trials] was considered part of my job. I was not compensated. It is different here.”
Dr. Colombel has numerous financial ties to the pharmaceutical industry, including AB Science, Amgen, Baxter, Bristol-Meyers Squibb, and Merck, among several others.
On Twitter @whitneymcknight
ORLANDO– Independent investigators eager to author studies that have notable impact on the field of inflammatory bowel disease have plenty of important paths to pursue, according to Dr. Jean-Frederic Colombel, a professor of gastroenterology at Mt. Sinai Hospital in New York.
“Over the next 3-5 years, we will have a lot of new drugs, which is nice, but we don’t yet know how to use them, or which ones to use,” Dr. Colombel said during a presentation on the future of the field at this year’s meeting of the annual Advances in Inflammatory Bowel Disease, sponsored by the Crohn’s and Colitis Foundation of America and endorsed by the American Gastroenterological Association.
A host of biologics including vedolizumab, recently indicated by the U.S. Food and Drug Administration for ulcerative colitis and Crohn’s disease, and ustekinumab, currently in phase III studies for Crohn’s disease, are set to revolutionize treatment at a time when the field is already undergoing great change, according to Dr. Colombel. The result, he says, is that despite “formidable” challenges in recruitment and funding, “huge opportunities” exist for investigators willing to collaborate and be creative.
Studies that elucidate the natural history of Crohn’s disease and colitis offer insight into the efficacy of various treatment strategies, help determine whether to target symptoms or biomarkers, and answer whether combination therapies are safe and effective in certain patients are what Dr. Colombel says he hopes will help improve the field as the drug pipeline continues to grow.
He stipulated a caveat, however, “I strongly believe we need some new study designs.” Although he noted that trials needn’t be complicated, they should be long enough to collect sorely needed prospective data. “These can only be done in investigator-initiated trials because of the time frames,” Dr. Colombel urged the audience of young investigators.
A fieldwide shift in thinking about ulcerative colitis and Crohn’s disease as chronic, progressive diseases, rather than intermittent afflictions has already helped generate new study endpoints such as the Lémann Score, an index of progressive bowel damage that allows researchers to better track the history of IBD in patients, and thus provide a window of opportunity for interventions, said Dr. Colombel.
To wit, the CURE study, conducted by the French IBD society GITAID, is a 5-year, prospective study of patients whose early Crohn’s disease is treated with the biologic adalimumab. The Lémann Score is used to screen patients at the end of each successive year, in order to adjust their treatment to reach the final endpoint of deep remission by year 5 when several indicators are measured, including bowel image, level of disability, and whether there was the need for surgery. Dr. Colombel said the novel design of the study, which has already recruited 60 patients, shows it is “feasible” to collect longitudinal data.
The field also has a responsibility to conduct randomly assigned controlled trials to demonstrate the comparative effectiveness, safety, immunogenicity, and cost benefits of the rapidly emerging spate of biosimilars, particularly since there is not global agreement about their use, according to Dr. Colombel. Their approval for IBD in Europe is “highly disputable.... I think this could be a very nice topic for an investigator-initiated trial, in Canada and the U.S.”
Head-to-head trials that are well designed will help answer “very important questions” about which treatment strategies have the best outcomes, but doing so requires fortitude, according to Dr. Colombel. “The Dutch have the guts to conduct the LIRIC trial,” he said. In it, patients with Crohn’s disease in the terminal ileum, who have failed steroids or immune therapy, will be assigned randomly to either laparoscopic ileocolic resection or infliximab, the first available biosimilar. Sixty participants have enrolled to date, he said.
These types of studies could also help delineate how best to employ combination therapies. “What I propose for this kind of study is an intensive therapy combining biologics very early in patients with bad prognoses, and looking at the long-term outcome using bowel image,” Dr. Colombel said. These data, and others indicating the most appropriate length of treatment in patients with varying states of disease, are Dr. Colombel’s personal “top choice” for investigation. The excitement that personalized medicine has engendered across the specialities has so far not resulted in specific, validated treatments for IBD patients. However, the question of how to use personal characteristics and serologic and genetic markers to create predictive models for which patients will need either step-up or top-down therapies, accounting for their individual risk of complication, is what he said is among the most “important [question] we need to answer because the choice of early therapy will depend on this predictor,” according to Dr. Colombel.
The question of which treatment targets are best remains unclear but is important to decipher, according to Dr. Colombel, who said simple studies comparing outcomes when patients are treated to symptoms vs. treated to biomarker measurements are needed, as well as the importance of mucosal healing vs. symptom improvement. “This is important because there is discordance between endoscopy and histology,” said Dr. Colombel. “Persistent histologic inflammation is frequently associated with bad outcomes.”
However, endoscopic scoring itself is another area Dr. Colombel said is worth investigating, especially when it comes to validating endpoints such as those in the Crohn’s Disease Index of Severity (CDEIS), and Simple Endoscopic Score for Crohn’s Disease (SES-CD), which will help determine remission cutoffs. Also, endoscopic scoring in comparison with live video and imaging still need standardized approaches. “When you see how it is done in the U.S., it is generally very poor,” Dr. Colombel said.
The biggest obstacles of all when it comes to independent investigation in the United States, according to Frenchman Dr. Colombel, is heavy regulation and cost. “In France, [conducting trials] was considered part of my job. I was not compensated. It is different here.”
Dr. Colombel has numerous financial ties to the pharmaceutical industry, including AB Science, Amgen, Baxter, Bristol-Meyers Squibb, and Merck, among several others.
On Twitter @whitneymcknight
Increased prevalence of chronic narcotic use in children with IBD
The prevalence of chronic narcotic use among pediatric patients with inflammatory bowel disease is abnormally high and should be curbed to prevent further adverse effects from taking hold, according to a new study published in the February issue of Clinical Gastroenterology and Hepatology.
“Although pain control is an important aspect of disease management in pediatric IBD [inflammatory bowel disease], little is known about chronic narcotic use in children,” wrote the investigators, led by Dr. Jessie P. Buckley of the University of North Carolina at Chapel Hill. They added that “although children with Crohn’s disease are at increased risk of anxiety and depression, the relationship between narcotic use and psychiatric conditions has not yet been assessed in the pediatric IBD population (Clin. Gastroenterol. Hepatol. 2015 February [doi:10.1016/j.cgh.2014.07.057].”
In a retrospective, cross-sectional study, Dr. Buckley and her associates counted all 4,911,286 patients aged 18 years or younger with continuous health plan enrollment and pharmacy benefits in the MarketScan Commercial Claims and Encounters database between January 1, 2010 and December 31, 2011. Children were defined as having IBD if they had either three or more health care visits for Crohn’s disease or ulcerative colitis during the study’s time frame, or “at least one health care contact for Crohn’s disease or ulcerative colitis and at least one pharmacy claim for any of the following medications: mesalamine, olsalazine, balsalazide, sulfasalazine, 6-mercaptopurine, azathioprine, methotrexate, enteral budesonide, or biologics (infliximab, adalimumab, certolizumab,or natalizumab).”
From this population, the authors used diagnosis codes based on data from the U.S. National Drug Code system, the European Pharmaceutical Market Research Association, and the Pharmaceutical Business Intelligence and Research Group Anatomical Classification System drug classes, along with information on dispensation of IBD medications, to select 4,344 IBD patients, each of whom was subsequently matched with 5 other patients without IBD based on age, sex, and geographic region within the United States (Northeast, North Central, South, or West), yielding 21,720 in this population. Subjects were defined as chronic users if they “had at least three narcotic drug claims during the 2-year study period.” Within the IBD population, 2,737 (63%) of subjects had Crohn’s disease and 1,607 (37%) had ulcerative colitis.
Investigators found that 5.6% (241 individuals) of the 4,344 subjects with IBD were chronic narcotic users, significantly higher than the 2.3% rate (489 subjects) of chronic narcotic use found in the non-IBD population (prevalence odds ratio, 2.59; 95% confidence interval, 2.21–3.04). Furthermore, chronic narcotic use was more prevalent in IBD patients with psychological impairment than those without: 15.7% (POR, 6.8; 95% CI, 4.3–10.6) versus 3.2% (POR, 2.3; 95% CI, 1.9–2.7), respectively. In children with IBD, older age and chronic narcotic use were also more highly associated with “increased health care utilization [and] fracture.”
“Children with IBD had more than twice the prevalence of chronic narcotic use as children without, and associations between IBD status and narcotic use indicate particularly high burden among those with concomitant anxiety or depression,” Dr. Buckley and her associates wrote, adding that “psychiatric diagnoses have also been associated with increased risk of narcotic use among adult patients with IBD. Greater attention to the complex relationships between pain and psychological impairment is warranted because children with IBD are at increased risk of anxiety and depression, compared with their peers, and other cofactors are not easily intervened on (e.g., age, region, fracture).”
The authors disclosed that financial support for this study was provided, at least in part, by GlaxoSmithKline (GSK). Dr. Buckley received funding through a research assistantship at GSK, coauthors Dr. Suzanne F. Cook and Dr. Jeffery K. Allen are employees of GSK, and coauthor Dr. Michael D. Kappelman is a consultant to GSK, among other companies.
The prevalence of chronic narcotic use among pediatric patients with inflammatory bowel disease is abnormally high and should be curbed to prevent further adverse effects from taking hold, according to a new study published in the February issue of Clinical Gastroenterology and Hepatology.
“Although pain control is an important aspect of disease management in pediatric IBD [inflammatory bowel disease], little is known about chronic narcotic use in children,” wrote the investigators, led by Dr. Jessie P. Buckley of the University of North Carolina at Chapel Hill. They added that “although children with Crohn’s disease are at increased risk of anxiety and depression, the relationship between narcotic use and psychiatric conditions has not yet been assessed in the pediatric IBD population (Clin. Gastroenterol. Hepatol. 2015 February [doi:10.1016/j.cgh.2014.07.057].”
In a retrospective, cross-sectional study, Dr. Buckley and her associates counted all 4,911,286 patients aged 18 years or younger with continuous health plan enrollment and pharmacy benefits in the MarketScan Commercial Claims and Encounters database between January 1, 2010 and December 31, 2011. Children were defined as having IBD if they had either three or more health care visits for Crohn’s disease or ulcerative colitis during the study’s time frame, or “at least one health care contact for Crohn’s disease or ulcerative colitis and at least one pharmacy claim for any of the following medications: mesalamine, olsalazine, balsalazide, sulfasalazine, 6-mercaptopurine, azathioprine, methotrexate, enteral budesonide, or biologics (infliximab, adalimumab, certolizumab,or natalizumab).”
From this population, the authors used diagnosis codes based on data from the U.S. National Drug Code system, the European Pharmaceutical Market Research Association, and the Pharmaceutical Business Intelligence and Research Group Anatomical Classification System drug classes, along with information on dispensation of IBD medications, to select 4,344 IBD patients, each of whom was subsequently matched with 5 other patients without IBD based on age, sex, and geographic region within the United States (Northeast, North Central, South, or West), yielding 21,720 in this population. Subjects were defined as chronic users if they “had at least three narcotic drug claims during the 2-year study period.” Within the IBD population, 2,737 (63%) of subjects had Crohn’s disease and 1,607 (37%) had ulcerative colitis.
Investigators found that 5.6% (241 individuals) of the 4,344 subjects with IBD were chronic narcotic users, significantly higher than the 2.3% rate (489 subjects) of chronic narcotic use found in the non-IBD population (prevalence odds ratio, 2.59; 95% confidence interval, 2.21–3.04). Furthermore, chronic narcotic use was more prevalent in IBD patients with psychological impairment than those without: 15.7% (POR, 6.8; 95% CI, 4.3–10.6) versus 3.2% (POR, 2.3; 95% CI, 1.9–2.7), respectively. In children with IBD, older age and chronic narcotic use were also more highly associated with “increased health care utilization [and] fracture.”
“Children with IBD had more than twice the prevalence of chronic narcotic use as children without, and associations between IBD status and narcotic use indicate particularly high burden among those with concomitant anxiety or depression,” Dr. Buckley and her associates wrote, adding that “psychiatric diagnoses have also been associated with increased risk of narcotic use among adult patients with IBD. Greater attention to the complex relationships between pain and psychological impairment is warranted because children with IBD are at increased risk of anxiety and depression, compared with their peers, and other cofactors are not easily intervened on (e.g., age, region, fracture).”
The authors disclosed that financial support for this study was provided, at least in part, by GlaxoSmithKline (GSK). Dr. Buckley received funding through a research assistantship at GSK, coauthors Dr. Suzanne F. Cook and Dr. Jeffery K. Allen are employees of GSK, and coauthor Dr. Michael D. Kappelman is a consultant to GSK, among other companies.
The prevalence of chronic narcotic use among pediatric patients with inflammatory bowel disease is abnormally high and should be curbed to prevent further adverse effects from taking hold, according to a new study published in the February issue of Clinical Gastroenterology and Hepatology.
“Although pain control is an important aspect of disease management in pediatric IBD [inflammatory bowel disease], little is known about chronic narcotic use in children,” wrote the investigators, led by Dr. Jessie P. Buckley of the University of North Carolina at Chapel Hill. They added that “although children with Crohn’s disease are at increased risk of anxiety and depression, the relationship between narcotic use and psychiatric conditions has not yet been assessed in the pediatric IBD population (Clin. Gastroenterol. Hepatol. 2015 February [doi:10.1016/j.cgh.2014.07.057].”
In a retrospective, cross-sectional study, Dr. Buckley and her associates counted all 4,911,286 patients aged 18 years or younger with continuous health plan enrollment and pharmacy benefits in the MarketScan Commercial Claims and Encounters database between January 1, 2010 and December 31, 2011. Children were defined as having IBD if they had either three or more health care visits for Crohn’s disease or ulcerative colitis during the study’s time frame, or “at least one health care contact for Crohn’s disease or ulcerative colitis and at least one pharmacy claim for any of the following medications: mesalamine, olsalazine, balsalazide, sulfasalazine, 6-mercaptopurine, azathioprine, methotrexate, enteral budesonide, or biologics (infliximab, adalimumab, certolizumab,or natalizumab).”
From this population, the authors used diagnosis codes based on data from the U.S. National Drug Code system, the European Pharmaceutical Market Research Association, and the Pharmaceutical Business Intelligence and Research Group Anatomical Classification System drug classes, along with information on dispensation of IBD medications, to select 4,344 IBD patients, each of whom was subsequently matched with 5 other patients without IBD based on age, sex, and geographic region within the United States (Northeast, North Central, South, or West), yielding 21,720 in this population. Subjects were defined as chronic users if they “had at least three narcotic drug claims during the 2-year study period.” Within the IBD population, 2,737 (63%) of subjects had Crohn’s disease and 1,607 (37%) had ulcerative colitis.
Investigators found that 5.6% (241 individuals) of the 4,344 subjects with IBD were chronic narcotic users, significantly higher than the 2.3% rate (489 subjects) of chronic narcotic use found in the non-IBD population (prevalence odds ratio, 2.59; 95% confidence interval, 2.21–3.04). Furthermore, chronic narcotic use was more prevalent in IBD patients with psychological impairment than those without: 15.7% (POR, 6.8; 95% CI, 4.3–10.6) versus 3.2% (POR, 2.3; 95% CI, 1.9–2.7), respectively. In children with IBD, older age and chronic narcotic use were also more highly associated with “increased health care utilization [and] fracture.”
“Children with IBD had more than twice the prevalence of chronic narcotic use as children without, and associations between IBD status and narcotic use indicate particularly high burden among those with concomitant anxiety or depression,” Dr. Buckley and her associates wrote, adding that “psychiatric diagnoses have also been associated with increased risk of narcotic use among adult patients with IBD. Greater attention to the complex relationships between pain and psychological impairment is warranted because children with IBD are at increased risk of anxiety and depression, compared with their peers, and other cofactors are not easily intervened on (e.g., age, region, fracture).”
The authors disclosed that financial support for this study was provided, at least in part, by GlaxoSmithKline (GSK). Dr. Buckley received funding through a research assistantship at GSK, coauthors Dr. Suzanne F. Cook and Dr. Jeffery K. Allen are employees of GSK, and coauthor Dr. Michael D. Kappelman is a consultant to GSK, among other companies.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: Chronic narcotic use in pediatric patients with inflammatory bowel disease has become increasingly prevalent.
Major finding: Prevalence of chronic narcotic use was 5.6% among children with IBD vs. 2.3% in the general population (OR, 2.6).
Data source: Retrospective, cross-sectional study.
Disclosures: Support for this study was provided by GlaxoSmithKline; all of the study’s coauthors are either employed by or otherwise affiliated with the company.
CDC: Hospital-acquired infections decreasing
Hospitals reduced significantly the number of surgical site and central-line associated bloodstream infections in 2013, according to a national analysis by the Centers for Disease Control and Prevention.
A review of data submitted by 14,500 health facilities found that central line–associated bloodstream infections (CLABSIs) fell by 46% between 2008 and 2013, and surgical site infections (SSIs) dropped by 19% over the same period. SSI data were derived from 10 select procedures, including hip arthroplasty, knee arthroplasty, colon surgery, rectal surgery, abdominal hysterectomy, vaginal hysterectomy, coronary artery bypass graft, other cardiac surgery, peripheral vascular bypass surgery, and abdominal aortic aneurysm repair.
Increased reporting by health care providers and quality measures imposed by the Centers for Medicare and Medicaid Services are likely contributors to the report’s findings, said Dr. Henry Pitt, chief quality officer for Temple University Health System, Philadelphia.
“Because of all the reporting that is being done, and the potential financial burdens that exist through CMS’s value-based purchasing program, and the work people are doing to improve these things, it’s not surprising that the data are looking better,” Dr. Pitt said in an interview.
The CDC’s annual National and State Healthcare-associated Infection Progress report summarizes data submitted to the CDC’s National Healthcare Safety Network (NHSN), a nationwide infection tracking system used by all 50 states, Washington, and Puerto Rico. In addition to SSIs and CLABSIs, findings of the report show also that methicillin-resistant Staphylococcus aureus (MRSA) bloodstream infections decreased by 8% from 2011 to 2013 and that Clostridium difficile infections dropped by 10% between 2011 and 2013. However, catheter-associated urinary tract infections have risen by 6% since 2009.
In a state-by-state comparison, 26 states performed better than the nation on at least two of the six infection types tracked by state. Sixteen states performed better than the nation on three or more infections and 19 states performed worse than the nation on two infections. Not all states reported or had enough data to calculate valid infection information on every infection in the report. Among the 2,543 U.S. hospitals with enough data to calculate a standardized infection ratio (SIR), 9% had an SIR significantly worse than the national SIR of 0.81.
Despite the progress, the report calls for more action to eliminate hospital infections and recommends its report be used by health departments, hospital associations, professional societies, health care systems and facilities, and quality improvement groups to identify infections that need additional prevention efforts.
Dr. Pitt added that CMS programs that use infection control and reduction as quality metrics will no doubt continue to impact infection reporting and outcomes. The three CMS programs associated with infection control and hospital payments include its hospital value-based purchasing, hospital readmissions reduction, and hospital-acquired condition reduction programs.
“Again, people are paying more attention, in addition to it’s the right thing to do, because more money is at risk,” Dr. Pitt said.
Data in the CDC report are from acute hospitals only.
On Twitter @legal_med
Hospitals reduced significantly the number of surgical site and central-line associated bloodstream infections in 2013, according to a national analysis by the Centers for Disease Control and Prevention.
A review of data submitted by 14,500 health facilities found that central line–associated bloodstream infections (CLABSIs) fell by 46% between 2008 and 2013, and surgical site infections (SSIs) dropped by 19% over the same period. SSI data were derived from 10 select procedures, including hip arthroplasty, knee arthroplasty, colon surgery, rectal surgery, abdominal hysterectomy, vaginal hysterectomy, coronary artery bypass graft, other cardiac surgery, peripheral vascular bypass surgery, and abdominal aortic aneurysm repair.
Increased reporting by health care providers and quality measures imposed by the Centers for Medicare and Medicaid Services are likely contributors to the report’s findings, said Dr. Henry Pitt, chief quality officer for Temple University Health System, Philadelphia.
“Because of all the reporting that is being done, and the potential financial burdens that exist through CMS’s value-based purchasing program, and the work people are doing to improve these things, it’s not surprising that the data are looking better,” Dr. Pitt said in an interview.
The CDC’s annual National and State Healthcare-associated Infection Progress report summarizes data submitted to the CDC’s National Healthcare Safety Network (NHSN), a nationwide infection tracking system used by all 50 states, Washington, and Puerto Rico. In addition to SSIs and CLABSIs, findings of the report show also that methicillin-resistant Staphylococcus aureus (MRSA) bloodstream infections decreased by 8% from 2011 to 2013 and that Clostridium difficile infections dropped by 10% between 2011 and 2013. However, catheter-associated urinary tract infections have risen by 6% since 2009.
In a state-by-state comparison, 26 states performed better than the nation on at least two of the six infection types tracked by state. Sixteen states performed better than the nation on three or more infections and 19 states performed worse than the nation on two infections. Not all states reported or had enough data to calculate valid infection information on every infection in the report. Among the 2,543 U.S. hospitals with enough data to calculate a standardized infection ratio (SIR), 9% had an SIR significantly worse than the national SIR of 0.81.
Despite the progress, the report calls for more action to eliminate hospital infections and recommends its report be used by health departments, hospital associations, professional societies, health care systems and facilities, and quality improvement groups to identify infections that need additional prevention efforts.
Dr. Pitt added that CMS programs that use infection control and reduction as quality metrics will no doubt continue to impact infection reporting and outcomes. The three CMS programs associated with infection control and hospital payments include its hospital value-based purchasing, hospital readmissions reduction, and hospital-acquired condition reduction programs.
“Again, people are paying more attention, in addition to it’s the right thing to do, because more money is at risk,” Dr. Pitt said.
Data in the CDC report are from acute hospitals only.
On Twitter @legal_med
Hospitals reduced significantly the number of surgical site and central-line associated bloodstream infections in 2013, according to a national analysis by the Centers for Disease Control and Prevention.
A review of data submitted by 14,500 health facilities found that central line–associated bloodstream infections (CLABSIs) fell by 46% between 2008 and 2013, and surgical site infections (SSIs) dropped by 19% over the same period. SSI data were derived from 10 select procedures, including hip arthroplasty, knee arthroplasty, colon surgery, rectal surgery, abdominal hysterectomy, vaginal hysterectomy, coronary artery bypass graft, other cardiac surgery, peripheral vascular bypass surgery, and abdominal aortic aneurysm repair.
Increased reporting by health care providers and quality measures imposed by the Centers for Medicare and Medicaid Services are likely contributors to the report’s findings, said Dr. Henry Pitt, chief quality officer for Temple University Health System, Philadelphia.
“Because of all the reporting that is being done, and the potential financial burdens that exist through CMS’s value-based purchasing program, and the work people are doing to improve these things, it’s not surprising that the data are looking better,” Dr. Pitt said in an interview.
The CDC’s annual National and State Healthcare-associated Infection Progress report summarizes data submitted to the CDC’s National Healthcare Safety Network (NHSN), a nationwide infection tracking system used by all 50 states, Washington, and Puerto Rico. In addition to SSIs and CLABSIs, findings of the report show also that methicillin-resistant Staphylococcus aureus (MRSA) bloodstream infections decreased by 8% from 2011 to 2013 and that Clostridium difficile infections dropped by 10% between 2011 and 2013. However, catheter-associated urinary tract infections have risen by 6% since 2009.
In a state-by-state comparison, 26 states performed better than the nation on at least two of the six infection types tracked by state. Sixteen states performed better than the nation on three or more infections and 19 states performed worse than the nation on two infections. Not all states reported or had enough data to calculate valid infection information on every infection in the report. Among the 2,543 U.S. hospitals with enough data to calculate a standardized infection ratio (SIR), 9% had an SIR significantly worse than the national SIR of 0.81.
Despite the progress, the report calls for more action to eliminate hospital infections and recommends its report be used by health departments, hospital associations, professional societies, health care systems and facilities, and quality improvement groups to identify infections that need additional prevention efforts.
Dr. Pitt added that CMS programs that use infection control and reduction as quality metrics will no doubt continue to impact infection reporting and outcomes. The three CMS programs associated with infection control and hospital payments include its hospital value-based purchasing, hospital readmissions reduction, and hospital-acquired condition reduction programs.
“Again, people are paying more attention, in addition to it’s the right thing to do, because more money is at risk,” Dr. Pitt said.
Data in the CDC report are from acute hospitals only.
On Twitter @legal_med