User login
Cathelicidins might help prevent, treat colonic fibrosis
Peptides known as cathelicidins directly inhibited collagen synthesis in human colonic fibroblasts and in mice with colitis, authors of a controlled, prospective study reported. The findings appeared Nov. 11 in Cellular and Molecular Gastroenterology and Hepatology.
“Our results strongly suggest that cathelicidin administration may be a novel approach to prevent or treat inflammatory bowel disease and IBD-related colonic fibrosis,” said Dr. Jun Hwan Yoo at the University of California, Los Angeles, and his associates.
Cathelicidins are endogenous antimicrobial peptides that exhibit “potent” anti-inflammatory effects against acute colitis, and inhibit colonic fibrosis in mice with chronic or infectious colitis, the investigators said. In past studies, cathelicidin-deficient mice were more susceptible to infections, had poorer wound healing, and developed worse colitis, compared with mice that were not cathelicidin deficient, they added. Cathelicidins also inhibit collagen synthesis in human dermal fibroblasts, they said (Cellular and Molecular Gastroenterology and Hepatology 2014 Nov. 11 10.1016/j.jcmgh.2014.08.001 [doi:10.1016/j.jcmgh.2014.08.001]).
To further explore the role of cathelicidins in intestinal fibrosis, Dr. Yoo and associates created two murine models of intestinal inflammation by infecting mice with Salmonella or by administering trinitrobenzene sulfonic acid enemas. Then they administered either intracolonic mCRAMP peptide at a dose of 5 mg/kg every 3 days, or intravenous injections of a lentivirus that overexpressed the cathelicidin gene. The researchers also exposed human intestinal fibroblasts and human colonic CCD-18Co fibroblasts to transforming growth factor beta1 (TGF-beta1) or to insulinlike growth factor 1, which induced collagen protein and mRNA expression that mimicked intestinal fibrosis. Then they exposed these cells to 3-5 mcm of the human cathelicidin LL-37.
The groups of mice with colitis had significantly higher colonic expression of collagen mRNA and significantly more colon tissue damage than did the normal controls, said the researchers. Mice with colitis that received mCRAMP or lentivirus-overexpressing cathelicidin gene had significantly lower collagen mRNA levels and less cecal and colonic collagen deposition, compared with mice that received only the vehicle control, they added (all P values less than .05). Intracolonic mCRAMP also restored body weight (P = .0178) in mice with colitis, compared with untreated controls, they added. Furthermore, LL-37 inhibited collagen synthesis in human intestinal and colonic fibroblasts (P = .0001), they said.
The research was supported by the UCLA CURE Center, the Crohn’s and Colitis Foundation of America, the National Institutes of Health, the Blinder Research Foundation for Crohn’s Disease, the Eli and Edythe Broad Chair, and the U.S. Public Health Service. The authors declared no conflicts of interest.
Fibrosis is a major complication of Crohn’s disease that can lead to strictures and intestinal obstruction. While biologic therapies have revolutionized medical treatment of Crohn’s disease and may reduce the incidence of recurrent stricturing disease, as many as 20% of Crohn’s disease patients treated with these agents still develop strictures.
One attractive therapeutic target could be TGF-beta, which induces fibroblasts to synthesize collagen and is upregulated in Crohn’s disease strictures. Unfortunately, anti-TGF-beta1 antibody therapy failed to work in other fibrotic diseases. In addition, toxicity might be expected in Crohn’s disease, as TGF-beta has pleiotropic functions in the gut, some of which are critical to homeostasis.
Recent work in extraintestinal organs has elucidated the involvement of cathelicidins, antimicrobial cationic peptides, in fibrosis. For example, LL-37, the cleaved form of human cathelicidin, can reduce TGF-beta–induced collagen synthesis by human keloid fibroblasts. However, the ability of cathelicidins to limit intestinal fibrosis has not been explored.
The study by Yoo et al. is important because it demonstrates that cathelicidins may be a powerful therapy for intestinal fibrosis. Importantly, cathelicidin therapy does not appear to affect TGF-beta signaling, as LL-37 therapy did not affect TGF-beta1 expression in vivo, but did inhibit TGF-beta–induced collagen production by primary Crohn’s disease intestinal fibroblasts in vitro.
It is also important to recognize that cathelicidins are already being developed as therapy to limit fibrosis in extra-intestinal diseases. Thus, much in the same way Crohn’s disease patients benefited from the development of anti-TNF-beta biologics for rheumatoid arthritis, it may be possible to take advantage of ongoing studies to reduce the cost and time involved in delivering a new therapeutic agent to patients.
Dr. Stefania Vetrano is a research associate at the IBD Center, Humanitas Clinical and Research Center, Rozzano, Milan. She has no conflicts of interest.
Fibrosis is a major complication of Crohn’s disease that can lead to strictures and intestinal obstruction. While biologic therapies have revolutionized medical treatment of Crohn’s disease and may reduce the incidence of recurrent stricturing disease, as many as 20% of Crohn’s disease patients treated with these agents still develop strictures.
One attractive therapeutic target could be TGF-beta, which induces fibroblasts to synthesize collagen and is upregulated in Crohn’s disease strictures. Unfortunately, anti-TGF-beta1 antibody therapy failed to work in other fibrotic diseases. In addition, toxicity might be expected in Crohn’s disease, as TGF-beta has pleiotropic functions in the gut, some of which are critical to homeostasis.
Recent work in extraintestinal organs has elucidated the involvement of cathelicidins, antimicrobial cationic peptides, in fibrosis. For example, LL-37, the cleaved form of human cathelicidin, can reduce TGF-beta–induced collagen synthesis by human keloid fibroblasts. However, the ability of cathelicidins to limit intestinal fibrosis has not been explored.
The study by Yoo et al. is important because it demonstrates that cathelicidins may be a powerful therapy for intestinal fibrosis. Importantly, cathelicidin therapy does not appear to affect TGF-beta signaling, as LL-37 therapy did not affect TGF-beta1 expression in vivo, but did inhibit TGF-beta–induced collagen production by primary Crohn’s disease intestinal fibroblasts in vitro.
It is also important to recognize that cathelicidins are already being developed as therapy to limit fibrosis in extra-intestinal diseases. Thus, much in the same way Crohn’s disease patients benefited from the development of anti-TNF-beta biologics for rheumatoid arthritis, it may be possible to take advantage of ongoing studies to reduce the cost and time involved in delivering a new therapeutic agent to patients.
Dr. Stefania Vetrano is a research associate at the IBD Center, Humanitas Clinical and Research Center, Rozzano, Milan. She has no conflicts of interest.
Fibrosis is a major complication of Crohn’s disease that can lead to strictures and intestinal obstruction. While biologic therapies have revolutionized medical treatment of Crohn’s disease and may reduce the incidence of recurrent stricturing disease, as many as 20% of Crohn’s disease patients treated with these agents still develop strictures.
One attractive therapeutic target could be TGF-beta, which induces fibroblasts to synthesize collagen and is upregulated in Crohn’s disease strictures. Unfortunately, anti-TGF-beta1 antibody therapy failed to work in other fibrotic diseases. In addition, toxicity might be expected in Crohn’s disease, as TGF-beta has pleiotropic functions in the gut, some of which are critical to homeostasis.
Recent work in extraintestinal organs has elucidated the involvement of cathelicidins, antimicrobial cationic peptides, in fibrosis. For example, LL-37, the cleaved form of human cathelicidin, can reduce TGF-beta–induced collagen synthesis by human keloid fibroblasts. However, the ability of cathelicidins to limit intestinal fibrosis has not been explored.
The study by Yoo et al. is important because it demonstrates that cathelicidins may be a powerful therapy for intestinal fibrosis. Importantly, cathelicidin therapy does not appear to affect TGF-beta signaling, as LL-37 therapy did not affect TGF-beta1 expression in vivo, but did inhibit TGF-beta–induced collagen production by primary Crohn’s disease intestinal fibroblasts in vitro.
It is also important to recognize that cathelicidins are already being developed as therapy to limit fibrosis in extra-intestinal diseases. Thus, much in the same way Crohn’s disease patients benefited from the development of anti-TNF-beta biologics for rheumatoid arthritis, it may be possible to take advantage of ongoing studies to reduce the cost and time involved in delivering a new therapeutic agent to patients.
Dr. Stefania Vetrano is a research associate at the IBD Center, Humanitas Clinical and Research Center, Rozzano, Milan. She has no conflicts of interest.
Peptides known as cathelicidins directly inhibited collagen synthesis in human colonic fibroblasts and in mice with colitis, authors of a controlled, prospective study reported. The findings appeared Nov. 11 in Cellular and Molecular Gastroenterology and Hepatology.
“Our results strongly suggest that cathelicidin administration may be a novel approach to prevent or treat inflammatory bowel disease and IBD-related colonic fibrosis,” said Dr. Jun Hwan Yoo at the University of California, Los Angeles, and his associates.
Cathelicidins are endogenous antimicrobial peptides that exhibit “potent” anti-inflammatory effects against acute colitis, and inhibit colonic fibrosis in mice with chronic or infectious colitis, the investigators said. In past studies, cathelicidin-deficient mice were more susceptible to infections, had poorer wound healing, and developed worse colitis, compared with mice that were not cathelicidin deficient, they added. Cathelicidins also inhibit collagen synthesis in human dermal fibroblasts, they said (Cellular and Molecular Gastroenterology and Hepatology 2014 Nov. 11 10.1016/j.jcmgh.2014.08.001 [doi:10.1016/j.jcmgh.2014.08.001]).
To further explore the role of cathelicidins in intestinal fibrosis, Dr. Yoo and associates created two murine models of intestinal inflammation by infecting mice with Salmonella or by administering trinitrobenzene sulfonic acid enemas. Then they administered either intracolonic mCRAMP peptide at a dose of 5 mg/kg every 3 days, or intravenous injections of a lentivirus that overexpressed the cathelicidin gene. The researchers also exposed human intestinal fibroblasts and human colonic CCD-18Co fibroblasts to transforming growth factor beta1 (TGF-beta1) or to insulinlike growth factor 1, which induced collagen protein and mRNA expression that mimicked intestinal fibrosis. Then they exposed these cells to 3-5 mcm of the human cathelicidin LL-37.
The groups of mice with colitis had significantly higher colonic expression of collagen mRNA and significantly more colon tissue damage than did the normal controls, said the researchers. Mice with colitis that received mCRAMP or lentivirus-overexpressing cathelicidin gene had significantly lower collagen mRNA levels and less cecal and colonic collagen deposition, compared with mice that received only the vehicle control, they added (all P values less than .05). Intracolonic mCRAMP also restored body weight (P = .0178) in mice with colitis, compared with untreated controls, they added. Furthermore, LL-37 inhibited collagen synthesis in human intestinal and colonic fibroblasts (P = .0001), they said.
The research was supported by the UCLA CURE Center, the Crohn’s and Colitis Foundation of America, the National Institutes of Health, the Blinder Research Foundation for Crohn’s Disease, the Eli and Edythe Broad Chair, and the U.S. Public Health Service. The authors declared no conflicts of interest.
Peptides known as cathelicidins directly inhibited collagen synthesis in human colonic fibroblasts and in mice with colitis, authors of a controlled, prospective study reported. The findings appeared Nov. 11 in Cellular and Molecular Gastroenterology and Hepatology.
“Our results strongly suggest that cathelicidin administration may be a novel approach to prevent or treat inflammatory bowel disease and IBD-related colonic fibrosis,” said Dr. Jun Hwan Yoo at the University of California, Los Angeles, and his associates.
Cathelicidins are endogenous antimicrobial peptides that exhibit “potent” anti-inflammatory effects against acute colitis, and inhibit colonic fibrosis in mice with chronic or infectious colitis, the investigators said. In past studies, cathelicidin-deficient mice were more susceptible to infections, had poorer wound healing, and developed worse colitis, compared with mice that were not cathelicidin deficient, they added. Cathelicidins also inhibit collagen synthesis in human dermal fibroblasts, they said (Cellular and Molecular Gastroenterology and Hepatology 2014 Nov. 11 10.1016/j.jcmgh.2014.08.001 [doi:10.1016/j.jcmgh.2014.08.001]).
To further explore the role of cathelicidins in intestinal fibrosis, Dr. Yoo and associates created two murine models of intestinal inflammation by infecting mice with Salmonella or by administering trinitrobenzene sulfonic acid enemas. Then they administered either intracolonic mCRAMP peptide at a dose of 5 mg/kg every 3 days, or intravenous injections of a lentivirus that overexpressed the cathelicidin gene. The researchers also exposed human intestinal fibroblasts and human colonic CCD-18Co fibroblasts to transforming growth factor beta1 (TGF-beta1) or to insulinlike growth factor 1, which induced collagen protein and mRNA expression that mimicked intestinal fibrosis. Then they exposed these cells to 3-5 mcm of the human cathelicidin LL-37.
The groups of mice with colitis had significantly higher colonic expression of collagen mRNA and significantly more colon tissue damage than did the normal controls, said the researchers. Mice with colitis that received mCRAMP or lentivirus-overexpressing cathelicidin gene had significantly lower collagen mRNA levels and less cecal and colonic collagen deposition, compared with mice that received only the vehicle control, they added (all P values less than .05). Intracolonic mCRAMP also restored body weight (P = .0178) in mice with colitis, compared with untreated controls, they added. Furthermore, LL-37 inhibited collagen synthesis in human intestinal and colonic fibroblasts (P = .0001), they said.
The research was supported by the UCLA CURE Center, the Crohn’s and Colitis Foundation of America, the National Institutes of Health, the Blinder Research Foundation for Crohn’s Disease, the Eli and Edythe Broad Chair, and the U.S. Public Health Service. The authors declared no conflicts of interest.
Key clinical point: Cathelicidins might help prevent or reverse intestinal fibrosis in patients with inflammatory bowel disease.
Major finding: Cathelicidins inhibited colonic fibrosis in mice with colitis and in human intestinal fibroblasts (P = .0001).
Data source: Controlled prospective study of the effects of cathelicidins in laboratory mice with colitis and in human intestinal fibroblasts.
Disclosures: The research was supported by the UCLA CURE Center, the Crohn’s and Colitis Foundation of America, the National Institutes of Health, the Blinder Research Foundation for Crohn’s Disease, the Eli and Edythe Broad Chair, and the U.S. Public Health Service. The authors declared no conflicts of interest.
FDA approves IV antibacterial for complicated UTIs, abdominal infections
A combination of a cephalosporin and a beta-lactamase inhibitor in an intravenous formulation has been approved for treating complicated intra-abdominal infections and complicated urinary tract infections in adults, the Food and Drug Administration announced on Dec. 19.
The cephalosporin is ceftolozane and the beta-lactamase inhibitor is tazobactam; it will be marketed as Zerbaxa by Cubist Pharmaceuticals.
This is the fourth antibacterial drug product approved by the FDA in 2014 and, like the other three, it was designated as a Qualified Infectious Disease Product (QIDP) and was given priority review, according to the FDA statement. Zerbaxa was granted a QIDP designation under the Generating Antibiotic Incentives Now (GAIN) Act of the FDA Safety and Innovation Act, “because it is an antibacterial or antifungal human drug intended to treat a serious or life-threatening infection,” the statement said.
As part of the QIDP program, the manufacturer has also been granted an extra 5 years of “exclusivity” – exclusive marketing rights – by the FDA.
Ceftolozane-tazobactam was approved for treating complicated intra-abdominal infections in combination with metronidazole; approval for this indication was based on a study of 979 adults, randomized to the combination or to meropenem.
Approval for complicated urinary tract infections, including pyelonephritis, was based on a study of 1,068 adults, randomized to treatment with ceftolozane-tazobactam or levofloxacin.
The prescribing information includes a warning about decreased efficacy in patients with renal impairment (a baseline creatinine clearance of 30-50 mL/min or less, and the recommendation to monitor creatinine clearance “at least daily in patients with changing renal function,” and to adjust dose accordingly. Nausea, diarrhea, headache, and fever were the most common adverse events in studies, according to the FDA statement.
The other antibacterials approved by the FDA in 2014 were approved for treating acute bacterial skin and skin structure infections caused by certain susceptible bacteria. They were dalbavancin (Dalvance), approved in May; tedizolid (Sivextro), approved in June; and oritavancin (Orbactiv), approved in August.
Serious adverse events associated with Zerbaxa should be reported to the FDA’s MedWatch program at 800-332-1088 or http://www.fda.gov/Safety/MedWatch/HowToReport/default.htm.
A combination of a cephalosporin and a beta-lactamase inhibitor in an intravenous formulation has been approved for treating complicated intra-abdominal infections and complicated urinary tract infections in adults, the Food and Drug Administration announced on Dec. 19.
The cephalosporin is ceftolozane and the beta-lactamase inhibitor is tazobactam; it will be marketed as Zerbaxa by Cubist Pharmaceuticals.
This is the fourth antibacterial drug product approved by the FDA in 2014 and, like the other three, it was designated as a Qualified Infectious Disease Product (QIDP) and was given priority review, according to the FDA statement. Zerbaxa was granted a QIDP designation under the Generating Antibiotic Incentives Now (GAIN) Act of the FDA Safety and Innovation Act, “because it is an antibacterial or antifungal human drug intended to treat a serious or life-threatening infection,” the statement said.
As part of the QIDP program, the manufacturer has also been granted an extra 5 years of “exclusivity” – exclusive marketing rights – by the FDA.
Ceftolozane-tazobactam was approved for treating complicated intra-abdominal infections in combination with metronidazole; approval for this indication was based on a study of 979 adults, randomized to the combination or to meropenem.
Approval for complicated urinary tract infections, including pyelonephritis, was based on a study of 1,068 adults, randomized to treatment with ceftolozane-tazobactam or levofloxacin.
The prescribing information includes a warning about decreased efficacy in patients with renal impairment (a baseline creatinine clearance of 30-50 mL/min or less, and the recommendation to monitor creatinine clearance “at least daily in patients with changing renal function,” and to adjust dose accordingly. Nausea, diarrhea, headache, and fever were the most common adverse events in studies, according to the FDA statement.
The other antibacterials approved by the FDA in 2014 were approved for treating acute bacterial skin and skin structure infections caused by certain susceptible bacteria. They were dalbavancin (Dalvance), approved in May; tedizolid (Sivextro), approved in June; and oritavancin (Orbactiv), approved in August.
Serious adverse events associated with Zerbaxa should be reported to the FDA’s MedWatch program at 800-332-1088 or http://www.fda.gov/Safety/MedWatch/HowToReport/default.htm.
A combination of a cephalosporin and a beta-lactamase inhibitor in an intravenous formulation has been approved for treating complicated intra-abdominal infections and complicated urinary tract infections in adults, the Food and Drug Administration announced on Dec. 19.
The cephalosporin is ceftolozane and the beta-lactamase inhibitor is tazobactam; it will be marketed as Zerbaxa by Cubist Pharmaceuticals.
This is the fourth antibacterial drug product approved by the FDA in 2014 and, like the other three, it was designated as a Qualified Infectious Disease Product (QIDP) and was given priority review, according to the FDA statement. Zerbaxa was granted a QIDP designation under the Generating Antibiotic Incentives Now (GAIN) Act of the FDA Safety and Innovation Act, “because it is an antibacterial or antifungal human drug intended to treat a serious or life-threatening infection,” the statement said.
As part of the QIDP program, the manufacturer has also been granted an extra 5 years of “exclusivity” – exclusive marketing rights – by the FDA.
Ceftolozane-tazobactam was approved for treating complicated intra-abdominal infections in combination with metronidazole; approval for this indication was based on a study of 979 adults, randomized to the combination or to meropenem.
Approval for complicated urinary tract infections, including pyelonephritis, was based on a study of 1,068 adults, randomized to treatment with ceftolozane-tazobactam or levofloxacin.
The prescribing information includes a warning about decreased efficacy in patients with renal impairment (a baseline creatinine clearance of 30-50 mL/min or less, and the recommendation to monitor creatinine clearance “at least daily in patients with changing renal function,” and to adjust dose accordingly. Nausea, diarrhea, headache, and fever were the most common adverse events in studies, according to the FDA statement.
The other antibacterials approved by the FDA in 2014 were approved for treating acute bacterial skin and skin structure infections caused by certain susceptible bacteria. They were dalbavancin (Dalvance), approved in May; tedizolid (Sivextro), approved in June; and oritavancin (Orbactiv), approved in August.
Serious adverse events associated with Zerbaxa should be reported to the FDA’s MedWatch program at 800-332-1088 or http://www.fda.gov/Safety/MedWatch/HowToReport/default.htm.
FDA approves four-drug combination for HCV
The Food and Drug Administration has approved the four-drug combination package Viekira Pak for the treatment of hepatitis C virus genotype 1 infection with or without cirrhosis, the agency announced December 19.
Viekira Pak is a combination of three new drugs, ombitasvir, paritaprevir, and dasabuvir; and the previously approved drug ritonavir. It can be taken with or without ribavirin but is not recommended for those with decompensated cirrhosis, the FDA said in a statement.
The FDA studied the drug combination’s efficacy in six clinical trials with 2,308 HCV patients, with or without cirrhosis. Patients were randomized to receive either Viekira Pak or placebo; Viekira Pak with or without ribavirin; or Viekira Pak with ribavirin for 12-24 weeks. Results showed that 91%-100% of participants who received the drug combination achieved sustained virologic response at least 12 weeks after treatment, the FDA said.
The most common side effects in trial participants were tiredness, itching, lack of energy, weakness, nausea, and trouble sleeping.
Viekira Pak is the fourth treatment approved for chronic hepatitis C virus in the past year. Simeprevir (Olysio); sofosbuvir (Sovaldi); and ledipasvir and sofosbuvir (Harvoni) were approved in November 2013, December 2013, and October 2014, respectively.
The recommended dosage for Viekira Pak is two ombitasvir, paritaprevir, and ritonavir tablets once per day at 12.5 mg, 75 mg, and 50 mg, respectively; and one 250-mg dasabuvir tablet twice per day.
The treatment was reviewed via FDA’s priority review program, which allows for expedited review of especially effective treatments for serious conditions.
Viekira Pak is marketed by AbbVie.
The Food and Drug Administration has approved the four-drug combination package Viekira Pak for the treatment of hepatitis C virus genotype 1 infection with or without cirrhosis, the agency announced December 19.
Viekira Pak is a combination of three new drugs, ombitasvir, paritaprevir, and dasabuvir; and the previously approved drug ritonavir. It can be taken with or without ribavirin but is not recommended for those with decompensated cirrhosis, the FDA said in a statement.
The FDA studied the drug combination’s efficacy in six clinical trials with 2,308 HCV patients, with or without cirrhosis. Patients were randomized to receive either Viekira Pak or placebo; Viekira Pak with or without ribavirin; or Viekira Pak with ribavirin for 12-24 weeks. Results showed that 91%-100% of participants who received the drug combination achieved sustained virologic response at least 12 weeks after treatment, the FDA said.
The most common side effects in trial participants were tiredness, itching, lack of energy, weakness, nausea, and trouble sleeping.
Viekira Pak is the fourth treatment approved for chronic hepatitis C virus in the past year. Simeprevir (Olysio); sofosbuvir (Sovaldi); and ledipasvir and sofosbuvir (Harvoni) were approved in November 2013, December 2013, and October 2014, respectively.
The recommended dosage for Viekira Pak is two ombitasvir, paritaprevir, and ritonavir tablets once per day at 12.5 mg, 75 mg, and 50 mg, respectively; and one 250-mg dasabuvir tablet twice per day.
The treatment was reviewed via FDA’s priority review program, which allows for expedited review of especially effective treatments for serious conditions.
Viekira Pak is marketed by AbbVie.
The Food and Drug Administration has approved the four-drug combination package Viekira Pak for the treatment of hepatitis C virus genotype 1 infection with or without cirrhosis, the agency announced December 19.
Viekira Pak is a combination of three new drugs, ombitasvir, paritaprevir, and dasabuvir; and the previously approved drug ritonavir. It can be taken with or without ribavirin but is not recommended for those with decompensated cirrhosis, the FDA said in a statement.
The FDA studied the drug combination’s efficacy in six clinical trials with 2,308 HCV patients, with or without cirrhosis. Patients were randomized to receive either Viekira Pak or placebo; Viekira Pak with or without ribavirin; or Viekira Pak with ribavirin for 12-24 weeks. Results showed that 91%-100% of participants who received the drug combination achieved sustained virologic response at least 12 weeks after treatment, the FDA said.
The most common side effects in trial participants were tiredness, itching, lack of energy, weakness, nausea, and trouble sleeping.
Viekira Pak is the fourth treatment approved for chronic hepatitis C virus in the past year. Simeprevir (Olysio); sofosbuvir (Sovaldi); and ledipasvir and sofosbuvir (Harvoni) were approved in November 2013, December 2013, and October 2014, respectively.
The recommended dosage for Viekira Pak is two ombitasvir, paritaprevir, and ritonavir tablets once per day at 12.5 mg, 75 mg, and 50 mg, respectively; and one 250-mg dasabuvir tablet twice per day.
The treatment was reviewed via FDA’s priority review program, which allows for expedited review of especially effective treatments for serious conditions.
Viekira Pak is marketed by AbbVie.
Faster clearance of vedolizumab associated with less mucosal healing in UC
ORLANDO – Ulcerative colitis sufferers with higher vedolizumab trough scores at 6 weeks in the GEMINI-1 study were found to have higher rates of mucosal healing, a post hoc, population pharmacokinetics analysis has shown.
The findings’ significance, however, is still a matter of debate, according to Maria Rosario, Ph.D., a director at Takeda Pharmaceuticals, and the data’s presenter at this year’s annual Advances in Inflammatory Bowel Disease meeting sponsored by the Crohn’s and Colitis Foundation of America.
“We have established a relationship between higher endoscopic scores and faster clearance, but we need to be careful how we interpret the data,” she concluded in her presentation, citing a lack of an established causal relationship between the two.
Results from the GEMINI-1 study lead to the Food and Drug Administration’s 2013 indication of vedolizumab, a disease-modifying monoclonal antibody, for the treatment of refractory ulcerative colitis (UC) and Crohn’s disease.
In the phase III study, two cohorts of UC patients were either double-blinded to vedolizumab 300 mg or placebo; or, to open-label vedolizumab 300 mg at weeks 0 and 2 during induction.
At week 6, responders to the medication in each cohort were re-randomized to either placebo or the study drug every 4 or 8 weeks during maintenance, up to week 52. Induction placebo patients and week 6 nonresponders continued their respective regimens.
For the post hoc analysis, serum levels of the drug in both cohorts were determined at weeks 6 and 46 according to each person’s Mayo Clinic endoscopic subscore at weeks 6 and 52. Trough concentration levels were divided into quartiles at weeks 6 and 46, as were the associated rates of mucosal healing at weeks 6 and 52. Dr. Rosario and her colleagues then used population pharmacokinetic modeling to estimate individual clearance values.
Patients who had higher levels of drug serum concentrations at week 6 were also found to have more mucosal healing. In the 55 patients who had an endoscopic subscore of 0, median trough concentrations were 34.5 mcg/mL; 30.4 mcg/mL in the 223 patients with subscores of 1; 24.0 mcg/mL in the 224 patients with a subscore of 2; and 19.6 mcg/mL in the 188 patients who had a subscore of 3.
Dr. Rosario also noted that in median week 6, trough concentrations in patients with the highest subscores lagged behind the overall week 6 median GEMINI-1 results, which were 25.6 mcg/mL.
Because this study did not measure fecal levels of the drug, these preliminary findings should encourage further investigation, said Dr. Rosario, who said the role of disease severity would be key to more precise interpretation of the data. Dr. Rosario is a director at Takeda Pharmaceuticals, manufacturer of Entyvio, the brand name for vedolizumab.
On Twitter @whitneymcknight
ORLANDO – Ulcerative colitis sufferers with higher vedolizumab trough scores at 6 weeks in the GEMINI-1 study were found to have higher rates of mucosal healing, a post hoc, population pharmacokinetics analysis has shown.
The findings’ significance, however, is still a matter of debate, according to Maria Rosario, Ph.D., a director at Takeda Pharmaceuticals, and the data’s presenter at this year’s annual Advances in Inflammatory Bowel Disease meeting sponsored by the Crohn’s and Colitis Foundation of America.
“We have established a relationship between higher endoscopic scores and faster clearance, but we need to be careful how we interpret the data,” she concluded in her presentation, citing a lack of an established causal relationship between the two.
Results from the GEMINI-1 study lead to the Food and Drug Administration’s 2013 indication of vedolizumab, a disease-modifying monoclonal antibody, for the treatment of refractory ulcerative colitis (UC) and Crohn’s disease.
In the phase III study, two cohorts of UC patients were either double-blinded to vedolizumab 300 mg or placebo; or, to open-label vedolizumab 300 mg at weeks 0 and 2 during induction.
At week 6, responders to the medication in each cohort were re-randomized to either placebo or the study drug every 4 or 8 weeks during maintenance, up to week 52. Induction placebo patients and week 6 nonresponders continued their respective regimens.
For the post hoc analysis, serum levels of the drug in both cohorts were determined at weeks 6 and 46 according to each person’s Mayo Clinic endoscopic subscore at weeks 6 and 52. Trough concentration levels were divided into quartiles at weeks 6 and 46, as were the associated rates of mucosal healing at weeks 6 and 52. Dr. Rosario and her colleagues then used population pharmacokinetic modeling to estimate individual clearance values.
Patients who had higher levels of drug serum concentrations at week 6 were also found to have more mucosal healing. In the 55 patients who had an endoscopic subscore of 0, median trough concentrations were 34.5 mcg/mL; 30.4 mcg/mL in the 223 patients with subscores of 1; 24.0 mcg/mL in the 224 patients with a subscore of 2; and 19.6 mcg/mL in the 188 patients who had a subscore of 3.
Dr. Rosario also noted that in median week 6, trough concentrations in patients with the highest subscores lagged behind the overall week 6 median GEMINI-1 results, which were 25.6 mcg/mL.
Because this study did not measure fecal levels of the drug, these preliminary findings should encourage further investigation, said Dr. Rosario, who said the role of disease severity would be key to more precise interpretation of the data. Dr. Rosario is a director at Takeda Pharmaceuticals, manufacturer of Entyvio, the brand name for vedolizumab.
On Twitter @whitneymcknight
ORLANDO – Ulcerative colitis sufferers with higher vedolizumab trough scores at 6 weeks in the GEMINI-1 study were found to have higher rates of mucosal healing, a post hoc, population pharmacokinetics analysis has shown.
The findings’ significance, however, is still a matter of debate, according to Maria Rosario, Ph.D., a director at Takeda Pharmaceuticals, and the data’s presenter at this year’s annual Advances in Inflammatory Bowel Disease meeting sponsored by the Crohn’s and Colitis Foundation of America.
“We have established a relationship between higher endoscopic scores and faster clearance, but we need to be careful how we interpret the data,” she concluded in her presentation, citing a lack of an established causal relationship between the two.
Results from the GEMINI-1 study lead to the Food and Drug Administration’s 2013 indication of vedolizumab, a disease-modifying monoclonal antibody, for the treatment of refractory ulcerative colitis (UC) and Crohn’s disease.
In the phase III study, two cohorts of UC patients were either double-blinded to vedolizumab 300 mg or placebo; or, to open-label vedolizumab 300 mg at weeks 0 and 2 during induction.
At week 6, responders to the medication in each cohort were re-randomized to either placebo or the study drug every 4 or 8 weeks during maintenance, up to week 52. Induction placebo patients and week 6 nonresponders continued their respective regimens.
For the post hoc analysis, serum levels of the drug in both cohorts were determined at weeks 6 and 46 according to each person’s Mayo Clinic endoscopic subscore at weeks 6 and 52. Trough concentration levels were divided into quartiles at weeks 6 and 46, as were the associated rates of mucosal healing at weeks 6 and 52. Dr. Rosario and her colleagues then used population pharmacokinetic modeling to estimate individual clearance values.
Patients who had higher levels of drug serum concentrations at week 6 were also found to have more mucosal healing. In the 55 patients who had an endoscopic subscore of 0, median trough concentrations were 34.5 mcg/mL; 30.4 mcg/mL in the 223 patients with subscores of 1; 24.0 mcg/mL in the 224 patients with a subscore of 2; and 19.6 mcg/mL in the 188 patients who had a subscore of 3.
Dr. Rosario also noted that in median week 6, trough concentrations in patients with the highest subscores lagged behind the overall week 6 median GEMINI-1 results, which were 25.6 mcg/mL.
Because this study did not measure fecal levels of the drug, these preliminary findings should encourage further investigation, said Dr. Rosario, who said the role of disease severity would be key to more precise interpretation of the data. Dr. Rosario is a director at Takeda Pharmaceuticals, manufacturer of Entyvio, the brand name for vedolizumab.
On Twitter @whitneymcknight
AT ADVANCES IN IBD 2014
Key clinical point: Patients with higher endoscopic subscores may be clearing vedolizumab faster than are those who show more mucosal healing.
Major finding: At 6 weeks, patients with higher trough concentrations of vedolizumab had lower endoscopic subscores.
Data source: Post hoc analysis of 693 ulcerative colitis patients from phase III, randomized GEMINI-1 study of vedolizumab’s efficacy in UC.
Disclosures: Dr. Rosario is a director at Takeda Pharmaceuticals, manufacturer of Entyvio, the brand name for vedolizumab.
Infliximab serum concentrations, efficacy linked in ulcerative colitis
Higher serum concentrations of infliximab are associated with clinical response, mucosal healing, and clinical remission in adults with moderate to severe ulcerative colitis, according to post hoc analyses of data from the Active Ulcerative Colitis Trials, ACT-1 and ACT-2.
In the 728 patients from the two randomized, controlled phase III pivotal trials, median serum concentrations of infliximab at week 8 were higher among patients with clinical response or mucosal healing during induction than in those who did not achieve these endpoints. For example, the median concentration among those who received 5-mg/kg doses of infliximab was 35.0 mcg/mL in responders, compared with 25.8 mcg/mL in nonresponders, Omoniyi J. Adedokun of Janssen Research and Development, Spring House, Pa., and colleagues reported in the December issue of Gastroenterology [doi:10.1053/j.gastro.2014.08.035].
“Similar results were observed for clinical response and mucosal healing during maintenance at week 30 and week 54,” the investigators wrote, noting that in the 5-mg/kg group, the median trough serum infliximab concentration was several-fold higher in responders than in nonresponders (3.9 vs. 1.2 mcg/mL at week 30 and 5.0 vs. 0.7 mcg/mL at week 54).
Concentrations did not differ significantly at 8 weeks in remitters and nonremitters in the 5-mg/kg group, but they did in patients who received 10 mg/kg and in patients in both dose groups at weeks 30 and 54, the investigators reported.
When assessed by infliximab concentration quartiles, treatment efficacy – defined by clinical response, mucosal healing, and/or clinical remission – generally improved with increasing concentrations in patients in both the 5- and 10-mg/kg groups; those with concentrations in the lowest quartile consistently were less likely to show clinical response, clinical remission, or mucosal healing, and had rates of success approaching those observed in the placebo groups.
Optimal infliximab concentration target thresholds associated with clinical improvement in ulcerative colitis patients in these analyses were 41 mcg/mL at week 8 (sensitivity, specificity, and positive predictive values of 63%, 62%, and 80%, respectively) and 3.7 mcg/mL at week 30 for maintenance of clinical response (sensitivity, specificity, and positive predictive values of 65%, 71%, and 82%, respectively). The data at week 54 suggested a range for serum infliximab concentrations of similar sensitivity, specificity, and positive predictive value, but those data represented only a subset of patients assessed, the investigators said.
Patients who achieved an efficacy outcome, but who failed to maintain that outcome, had lower serum infliximab concentrations earlier in the course of therapy than did those who maintained the outcome, the investigators said.
“In general, the lower the infliximab concentration at a given time point, the more likely the patients were to fail to maintain remission,” they wrote.
The findings demonstrate a strong association between serum infliximab concentration and efficacy outcomes in patients with ulcerative colitis, and highlight the possibility of infliximab dose optimization – particularly in patients who are likely to lose efficacy while receiving a standard dose of infliximab, the investigators said.
Target threshold concentrations identified by this analysis could help clinicians understand why an individual patient fails to achieve the expected efficacy, but a prospective study designed to confirm the importance of optimizing infliximab concentrations is needed before it can be determined whether these results can be exploited to achieve better outcomes for patients with ulcerative colitis, Mr. Adedokun and coinvestigators said.
The ACT trials were funded by Janssen Research and Development, which also employs Mr. Adedokun.
Higher serum concentrations of infliximab are associated with clinical response, mucosal healing, and clinical remission in adults with moderate to severe ulcerative colitis, according to post hoc analyses of data from the Active Ulcerative Colitis Trials, ACT-1 and ACT-2.
In the 728 patients from the two randomized, controlled phase III pivotal trials, median serum concentrations of infliximab at week 8 were higher among patients with clinical response or mucosal healing during induction than in those who did not achieve these endpoints. For example, the median concentration among those who received 5-mg/kg doses of infliximab was 35.0 mcg/mL in responders, compared with 25.8 mcg/mL in nonresponders, Omoniyi J. Adedokun of Janssen Research and Development, Spring House, Pa., and colleagues reported in the December issue of Gastroenterology [doi:10.1053/j.gastro.2014.08.035].
“Similar results were observed for clinical response and mucosal healing during maintenance at week 30 and week 54,” the investigators wrote, noting that in the 5-mg/kg group, the median trough serum infliximab concentration was several-fold higher in responders than in nonresponders (3.9 vs. 1.2 mcg/mL at week 30 and 5.0 vs. 0.7 mcg/mL at week 54).
Concentrations did not differ significantly at 8 weeks in remitters and nonremitters in the 5-mg/kg group, but they did in patients who received 10 mg/kg and in patients in both dose groups at weeks 30 and 54, the investigators reported.
When assessed by infliximab concentration quartiles, treatment efficacy – defined by clinical response, mucosal healing, and/or clinical remission – generally improved with increasing concentrations in patients in both the 5- and 10-mg/kg groups; those with concentrations in the lowest quartile consistently were less likely to show clinical response, clinical remission, or mucosal healing, and had rates of success approaching those observed in the placebo groups.
Optimal infliximab concentration target thresholds associated with clinical improvement in ulcerative colitis patients in these analyses were 41 mcg/mL at week 8 (sensitivity, specificity, and positive predictive values of 63%, 62%, and 80%, respectively) and 3.7 mcg/mL at week 30 for maintenance of clinical response (sensitivity, specificity, and positive predictive values of 65%, 71%, and 82%, respectively). The data at week 54 suggested a range for serum infliximab concentrations of similar sensitivity, specificity, and positive predictive value, but those data represented only a subset of patients assessed, the investigators said.
Patients who achieved an efficacy outcome, but who failed to maintain that outcome, had lower serum infliximab concentrations earlier in the course of therapy than did those who maintained the outcome, the investigators said.
“In general, the lower the infliximab concentration at a given time point, the more likely the patients were to fail to maintain remission,” they wrote.
The findings demonstrate a strong association between serum infliximab concentration and efficacy outcomes in patients with ulcerative colitis, and highlight the possibility of infliximab dose optimization – particularly in patients who are likely to lose efficacy while receiving a standard dose of infliximab, the investigators said.
Target threshold concentrations identified by this analysis could help clinicians understand why an individual patient fails to achieve the expected efficacy, but a prospective study designed to confirm the importance of optimizing infliximab concentrations is needed before it can be determined whether these results can be exploited to achieve better outcomes for patients with ulcerative colitis, Mr. Adedokun and coinvestigators said.
The ACT trials were funded by Janssen Research and Development, which also employs Mr. Adedokun.
Higher serum concentrations of infliximab are associated with clinical response, mucosal healing, and clinical remission in adults with moderate to severe ulcerative colitis, according to post hoc analyses of data from the Active Ulcerative Colitis Trials, ACT-1 and ACT-2.
In the 728 patients from the two randomized, controlled phase III pivotal trials, median serum concentrations of infliximab at week 8 were higher among patients with clinical response or mucosal healing during induction than in those who did not achieve these endpoints. For example, the median concentration among those who received 5-mg/kg doses of infliximab was 35.0 mcg/mL in responders, compared with 25.8 mcg/mL in nonresponders, Omoniyi J. Adedokun of Janssen Research and Development, Spring House, Pa., and colleagues reported in the December issue of Gastroenterology [doi:10.1053/j.gastro.2014.08.035].
“Similar results were observed for clinical response and mucosal healing during maintenance at week 30 and week 54,” the investigators wrote, noting that in the 5-mg/kg group, the median trough serum infliximab concentration was several-fold higher in responders than in nonresponders (3.9 vs. 1.2 mcg/mL at week 30 and 5.0 vs. 0.7 mcg/mL at week 54).
Concentrations did not differ significantly at 8 weeks in remitters and nonremitters in the 5-mg/kg group, but they did in patients who received 10 mg/kg and in patients in both dose groups at weeks 30 and 54, the investigators reported.
When assessed by infliximab concentration quartiles, treatment efficacy – defined by clinical response, mucosal healing, and/or clinical remission – generally improved with increasing concentrations in patients in both the 5- and 10-mg/kg groups; those with concentrations in the lowest quartile consistently were less likely to show clinical response, clinical remission, or mucosal healing, and had rates of success approaching those observed in the placebo groups.
Optimal infliximab concentration target thresholds associated with clinical improvement in ulcerative colitis patients in these analyses were 41 mcg/mL at week 8 (sensitivity, specificity, and positive predictive values of 63%, 62%, and 80%, respectively) and 3.7 mcg/mL at week 30 for maintenance of clinical response (sensitivity, specificity, and positive predictive values of 65%, 71%, and 82%, respectively). The data at week 54 suggested a range for serum infliximab concentrations of similar sensitivity, specificity, and positive predictive value, but those data represented only a subset of patients assessed, the investigators said.
Patients who achieved an efficacy outcome, but who failed to maintain that outcome, had lower serum infliximab concentrations earlier in the course of therapy than did those who maintained the outcome, the investigators said.
“In general, the lower the infliximab concentration at a given time point, the more likely the patients were to fail to maintain remission,” they wrote.
The findings demonstrate a strong association between serum infliximab concentration and efficacy outcomes in patients with ulcerative colitis, and highlight the possibility of infliximab dose optimization – particularly in patients who are likely to lose efficacy while receiving a standard dose of infliximab, the investigators said.
Target threshold concentrations identified by this analysis could help clinicians understand why an individual patient fails to achieve the expected efficacy, but a prospective study designed to confirm the importance of optimizing infliximab concentrations is needed before it can be determined whether these results can be exploited to achieve better outcomes for patients with ulcerative colitis, Mr. Adedokun and coinvestigators said.
The ACT trials were funded by Janssen Research and Development, which also employs Mr. Adedokun.
Key clinical point: Serum concentrations of infliximab are associated with response, and could allow for dose optimization.
Major finding: Median concentrations of infliximab at 8 weeks in those receiving 5 mg/kg were 35.0 mcg/mL in responders vs. 25.8 mcg/mL in nonresponders.
Data source: Post hoc analyses of the ACT trials involving 728 patients.
Disclosures: The ACT trials were funded by Janssen Research and Development, which also employs Mr. Adedokun.
Large study shows no link between celiac disease and fertility problems
With the exception of those diagnosed between the age of 25 and 29 years, women with celiac disease are no more likely than are women without celiac disease to have fertility problems, according to findings from a large population-based study in the United Kingdom.
Of more than 2.4 million women with prospective primary care records available during their childbearing years (ages 15-49 years) between 1990 and 2013, 6,506 were diagnosed with celiac disease. The women with celiac disease had a similar rate of recorded fertility problems as did those without celiac disease (4.4% vs. 4.1%), Nafeesa N. Dhalwani of the University of Nottingham and City Hospital Nottingham, U.K., and colleagues reported in the December issue of Gastroenterology (doi:10.1053/j.gastro.2014.08.025).
Source: American Gastroenterological Association
Further, the rates of infertility in those with celiac disease were similar to those without celiac disease both before and after diagnosis except in those aged 25-29 years at the time of diagnosis; the rates in those women were 41% higher, compared with those without celiac disease who were the same age (incidence rate ratio, 1.41), the investigators said.
“However, the absolute excess risk [for those diagnosed at age 25-29 years] was only 0.5% (5.2/1,000 person-years), they said.
Women included in the analysis were identified from the Health Improvement Network database. Rates of new clinically recorded fertility problems among those with and without diagnosed celiac disease were stratified by timing of celiac disease diagnosis after adjustment for sociodemographics, comorbidities, and calendar year.
The findings contrast with those from a number of smaller studies that demonstrated an association between infertility and celiac disease, but those studies included small numbers of women, including many who were receiving infertility specialist services, the investigators said, explaining that the women may not have been representative of the general population, and that other small studies found no link between celiac disease and fertility problems.
Celiac disease affects about 1% of the population in North America and Western Europe, and between 60% and 70% of those who are diagnosed are women. Several mechanisms through which celiac disease might affect a woman’s fertility have been described in the literature, but no conclusive evidence exists to support them, they noted.
Despite this lack of evidence and the inconsistent findings from small studies, a number of reviews include infertility as a key nongastrointestinal manifestation of celiac disease. The current findings suggest that most women with celiac disease – whether diagnosed or undiagnosed – do not have a substantially greater likelihood of having clinically recorded fertility problems than do those without celiac disease.
“Therefore, screening when women initially present with fertility problems may not identify a significant number of women with celiac disease, beyond the general population prevalence. This may not always apply to subgroups of women with severe celiac disease. However, in terms of the clinical burden of fertility problems at a population level, these findings should be reassuring for women with celiac disease and all stakeholders involved in their care,” the investigators concluded.
This study was supported by CORE/Coeliac UK, and by a University of Nottingham/Nottingham University Hospitals National Health Service Trust Senior Clinical Research Fellowship. The authors reported having no disclosures.
With the exception of those diagnosed between the age of 25 and 29 years, women with celiac disease are no more likely than are women without celiac disease to have fertility problems, according to findings from a large population-based study in the United Kingdom.
Of more than 2.4 million women with prospective primary care records available during their childbearing years (ages 15-49 years) between 1990 and 2013, 6,506 were diagnosed with celiac disease. The women with celiac disease had a similar rate of recorded fertility problems as did those without celiac disease (4.4% vs. 4.1%), Nafeesa N. Dhalwani of the University of Nottingham and City Hospital Nottingham, U.K., and colleagues reported in the December issue of Gastroenterology (doi:10.1053/j.gastro.2014.08.025).
Source: American Gastroenterological Association
Further, the rates of infertility in those with celiac disease were similar to those without celiac disease both before and after diagnosis except in those aged 25-29 years at the time of diagnosis; the rates in those women were 41% higher, compared with those without celiac disease who were the same age (incidence rate ratio, 1.41), the investigators said.
“However, the absolute excess risk [for those diagnosed at age 25-29 years] was only 0.5% (5.2/1,000 person-years), they said.
Women included in the analysis were identified from the Health Improvement Network database. Rates of new clinically recorded fertility problems among those with and without diagnosed celiac disease were stratified by timing of celiac disease diagnosis after adjustment for sociodemographics, comorbidities, and calendar year.
The findings contrast with those from a number of smaller studies that demonstrated an association between infertility and celiac disease, but those studies included small numbers of women, including many who were receiving infertility specialist services, the investigators said, explaining that the women may not have been representative of the general population, and that other small studies found no link between celiac disease and fertility problems.
Celiac disease affects about 1% of the population in North America and Western Europe, and between 60% and 70% of those who are diagnosed are women. Several mechanisms through which celiac disease might affect a woman’s fertility have been described in the literature, but no conclusive evidence exists to support them, they noted.
Despite this lack of evidence and the inconsistent findings from small studies, a number of reviews include infertility as a key nongastrointestinal manifestation of celiac disease. The current findings suggest that most women with celiac disease – whether diagnosed or undiagnosed – do not have a substantially greater likelihood of having clinically recorded fertility problems than do those without celiac disease.
“Therefore, screening when women initially present with fertility problems may not identify a significant number of women with celiac disease, beyond the general population prevalence. This may not always apply to subgroups of women with severe celiac disease. However, in terms of the clinical burden of fertility problems at a population level, these findings should be reassuring for women with celiac disease and all stakeholders involved in their care,” the investigators concluded.
This study was supported by CORE/Coeliac UK, and by a University of Nottingham/Nottingham University Hospitals National Health Service Trust Senior Clinical Research Fellowship. The authors reported having no disclosures.
With the exception of those diagnosed between the age of 25 and 29 years, women with celiac disease are no more likely than are women without celiac disease to have fertility problems, according to findings from a large population-based study in the United Kingdom.
Of more than 2.4 million women with prospective primary care records available during their childbearing years (ages 15-49 years) between 1990 and 2013, 6,506 were diagnosed with celiac disease. The women with celiac disease had a similar rate of recorded fertility problems as did those without celiac disease (4.4% vs. 4.1%), Nafeesa N. Dhalwani of the University of Nottingham and City Hospital Nottingham, U.K., and colleagues reported in the December issue of Gastroenterology (doi:10.1053/j.gastro.2014.08.025).
Source: American Gastroenterological Association
Further, the rates of infertility in those with celiac disease were similar to those without celiac disease both before and after diagnosis except in those aged 25-29 years at the time of diagnosis; the rates in those women were 41% higher, compared with those without celiac disease who were the same age (incidence rate ratio, 1.41), the investigators said.
“However, the absolute excess risk [for those diagnosed at age 25-29 years] was only 0.5% (5.2/1,000 person-years), they said.
Women included in the analysis were identified from the Health Improvement Network database. Rates of new clinically recorded fertility problems among those with and without diagnosed celiac disease were stratified by timing of celiac disease diagnosis after adjustment for sociodemographics, comorbidities, and calendar year.
The findings contrast with those from a number of smaller studies that demonstrated an association between infertility and celiac disease, but those studies included small numbers of women, including many who were receiving infertility specialist services, the investigators said, explaining that the women may not have been representative of the general population, and that other small studies found no link between celiac disease and fertility problems.
Celiac disease affects about 1% of the population in North America and Western Europe, and between 60% and 70% of those who are diagnosed are women. Several mechanisms through which celiac disease might affect a woman’s fertility have been described in the literature, but no conclusive evidence exists to support them, they noted.
Despite this lack of evidence and the inconsistent findings from small studies, a number of reviews include infertility as a key nongastrointestinal manifestation of celiac disease. The current findings suggest that most women with celiac disease – whether diagnosed or undiagnosed – do not have a substantially greater likelihood of having clinically recorded fertility problems than do those without celiac disease.
“Therefore, screening when women initially present with fertility problems may not identify a significant number of women with celiac disease, beyond the general population prevalence. This may not always apply to subgroups of women with severe celiac disease. However, in terms of the clinical burden of fertility problems at a population level, these findings should be reassuring for women with celiac disease and all stakeholders involved in their care,” the investigators concluded.
This study was supported by CORE/Coeliac UK, and by a University of Nottingham/Nottingham University Hospitals National Health Service Trust Senior Clinical Research Fellowship. The authors reported having no disclosures.
Key clinical point: Women with celiac disease are not at increased risk of fertility problems.
Major finding: Women with celiac disease had a similar rate of recorded fertility problems as did those without celiac disease (4.4% vs. 4.1%).
Data source: A population-based study of more than 2.4 million women.
Disclosures: This study was supported by CORE/Coeliac UK, and by a University of Nottingham/Nottingham University Hospitals National Health Service Trust Senior Clinical Research Fellowship. The authors reported having no disclosures.
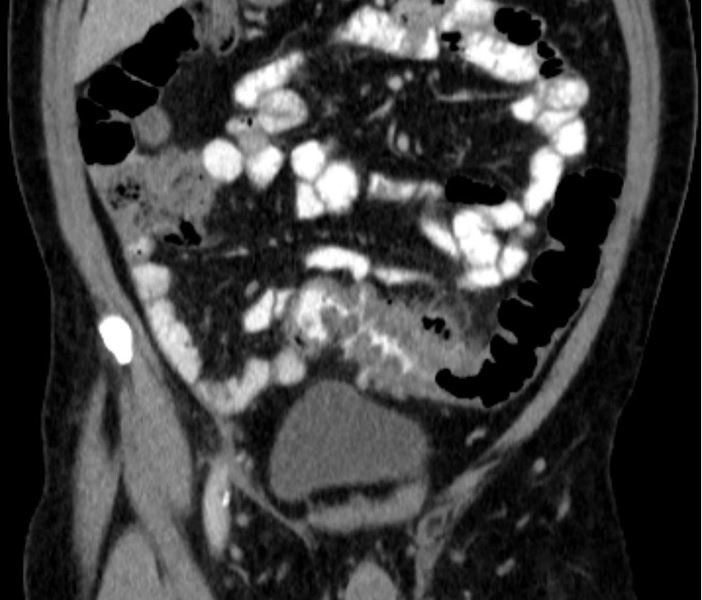
CT overutilized to diagnose appendicitis
SAN FRANCISCO – At least 25% of CT scans to diagnose appendicitis were unnecessary, potentially resulting in $1.8 million in costs at one institution and up to four new cancers from the radiation exposure, a retrospective study suggests.
The review of 1,054 patients who underwent appendectomy at the University of California, Davis, in 2005-2010 focused on costs for the patients who had high Alvarado scores, a clinical scoring system used to diagnose appendicitis, before they underwent appendectomy. CT scans to help diagnose appendicitis were performed on 77% of all patients.
Records showed that 26% of patients had an Alvarado score of 8-10, meaning that appendicitis was highly likely. CT was performed on 70% of patients with an Alvarado score of 8 and 77% of patients with a score of 9-10, comprising nearly 25% of all CT scans. That resulted in an estimated $1,813,399 in unnecessary costs for imaging, Dr. Adam Dougherty and his associates reported at the annual clinical congress of the American College of Surgeons.
This “overutilization” of CT scans delivered more than 4,009 mSv in unnecessary radiation exposure, averaging 19.75 mSV per scan, which is 20 times the annual limit suggested for safety, said Dr. Dougherty of the university. That excess radiation could be expected to produce up to four new cancers down the line, resulting in additional costs, he said.
The investigators also looked at the 9% of patients with low Alvarado scores, meaning that appendicitis was unlikely. CT scans were performed in 75% of patients with a score of 0-3 and 80% of patients with an Alvarado score of 4. In this subgroup, 24% showed normal/early pathology on appendectomy, which “argues against imaging and surgical treatment,” Dr. Dougherty said. The 44 CT scans in this subgroup resulted in an estimated $393,052 in unnecessary costs, he said.
That doesn’t include additional costs that could be expected from imaging, such as wait time, appendectomy and its sequelae, and potential workups of incidentalomas in the low-risk group, he added.
Previous studies have shown that a comprehensive clinical exam is as accurate as CT in diagnosing appendicitis, and that clinical assessment unaided by CT can reliably diagnose acute appendicitis, Dr. Dougherty said.
With a 72% increase in abdominal CT scans documented in other U.S. data from 2000 to 2005, he called for a “necessary, fundamental culture change” to restrain resource utilization “in order to maximize the value of the health care dollar while doing what is best for the patient.”
The investigators proposed a clinical pathway for the workup of suspected appendicitis that places greater emphasis on ultrasound imaging and conservative pathways, such as 23-hour admission for observation and next-day follow-up.
In the study, ultrasonography was underutilized across all subgroups as a viable alternative to CT scans, he said.
Dr. Dougherty reported having no financial disclosures.
On Twitter @sherryboschert
SAN FRANCISCO – At least 25% of CT scans to diagnose appendicitis were unnecessary, potentially resulting in $1.8 million in costs at one institution and up to four new cancers from the radiation exposure, a retrospective study suggests.
The review of 1,054 patients who underwent appendectomy at the University of California, Davis, in 2005-2010 focused on costs for the patients who had high Alvarado scores, a clinical scoring system used to diagnose appendicitis, before they underwent appendectomy. CT scans to help diagnose appendicitis were performed on 77% of all patients.
Records showed that 26% of patients had an Alvarado score of 8-10, meaning that appendicitis was highly likely. CT was performed on 70% of patients with an Alvarado score of 8 and 77% of patients with a score of 9-10, comprising nearly 25% of all CT scans. That resulted in an estimated $1,813,399 in unnecessary costs for imaging, Dr. Adam Dougherty and his associates reported at the annual clinical congress of the American College of Surgeons.
This “overutilization” of CT scans delivered more than 4,009 mSv in unnecessary radiation exposure, averaging 19.75 mSV per scan, which is 20 times the annual limit suggested for safety, said Dr. Dougherty of the university. That excess radiation could be expected to produce up to four new cancers down the line, resulting in additional costs, he said.
The investigators also looked at the 9% of patients with low Alvarado scores, meaning that appendicitis was unlikely. CT scans were performed in 75% of patients with a score of 0-3 and 80% of patients with an Alvarado score of 4. In this subgroup, 24% showed normal/early pathology on appendectomy, which “argues against imaging and surgical treatment,” Dr. Dougherty said. The 44 CT scans in this subgroup resulted in an estimated $393,052 in unnecessary costs, he said.
That doesn’t include additional costs that could be expected from imaging, such as wait time, appendectomy and its sequelae, and potential workups of incidentalomas in the low-risk group, he added.
Previous studies have shown that a comprehensive clinical exam is as accurate as CT in diagnosing appendicitis, and that clinical assessment unaided by CT can reliably diagnose acute appendicitis, Dr. Dougherty said.
With a 72% increase in abdominal CT scans documented in other U.S. data from 2000 to 2005, he called for a “necessary, fundamental culture change” to restrain resource utilization “in order to maximize the value of the health care dollar while doing what is best for the patient.”
The investigators proposed a clinical pathway for the workup of suspected appendicitis that places greater emphasis on ultrasound imaging and conservative pathways, such as 23-hour admission for observation and next-day follow-up.
In the study, ultrasonography was underutilized across all subgroups as a viable alternative to CT scans, he said.
Dr. Dougherty reported having no financial disclosures.
On Twitter @sherryboschert
SAN FRANCISCO – At least 25% of CT scans to diagnose appendicitis were unnecessary, potentially resulting in $1.8 million in costs at one institution and up to four new cancers from the radiation exposure, a retrospective study suggests.
The review of 1,054 patients who underwent appendectomy at the University of California, Davis, in 2005-2010 focused on costs for the patients who had high Alvarado scores, a clinical scoring system used to diagnose appendicitis, before they underwent appendectomy. CT scans to help diagnose appendicitis were performed on 77% of all patients.
Records showed that 26% of patients had an Alvarado score of 8-10, meaning that appendicitis was highly likely. CT was performed on 70% of patients with an Alvarado score of 8 and 77% of patients with a score of 9-10, comprising nearly 25% of all CT scans. That resulted in an estimated $1,813,399 in unnecessary costs for imaging, Dr. Adam Dougherty and his associates reported at the annual clinical congress of the American College of Surgeons.
This “overutilization” of CT scans delivered more than 4,009 mSv in unnecessary radiation exposure, averaging 19.75 mSV per scan, which is 20 times the annual limit suggested for safety, said Dr. Dougherty of the university. That excess radiation could be expected to produce up to four new cancers down the line, resulting in additional costs, he said.
The investigators also looked at the 9% of patients with low Alvarado scores, meaning that appendicitis was unlikely. CT scans were performed in 75% of patients with a score of 0-3 and 80% of patients with an Alvarado score of 4. In this subgroup, 24% showed normal/early pathology on appendectomy, which “argues against imaging and surgical treatment,” Dr. Dougherty said. The 44 CT scans in this subgroup resulted in an estimated $393,052 in unnecessary costs, he said.
That doesn’t include additional costs that could be expected from imaging, such as wait time, appendectomy and its sequelae, and potential workups of incidentalomas in the low-risk group, he added.
Previous studies have shown that a comprehensive clinical exam is as accurate as CT in diagnosing appendicitis, and that clinical assessment unaided by CT can reliably diagnose acute appendicitis, Dr. Dougherty said.
With a 72% increase in abdominal CT scans documented in other U.S. data from 2000 to 2005, he called for a “necessary, fundamental culture change” to restrain resource utilization “in order to maximize the value of the health care dollar while doing what is best for the patient.”
The investigators proposed a clinical pathway for the workup of suspected appendicitis that places greater emphasis on ultrasound imaging and conservative pathways, such as 23-hour admission for observation and next-day follow-up.
In the study, ultrasonography was underutilized across all subgroups as a viable alternative to CT scans, he said.
Dr. Dougherty reported having no financial disclosures.
On Twitter @sherryboschert
AT THE ACS CLINICAL CONGRESS
Key clinical point: Records showed that 26% of patients who had CT scans for suspect appendicitis had an Alvarado score of 8-10.
Major finding: A quarter of CT scans were on patients with likely appendicitis by Alvarado score, producing $1.8 million in unnecessary costs.
Data source: A retrospective study of 1,054 patients undergoing appendectomy in 2005-2010 at one institution.
Disclosures: Dr. Dougherty reported having no financial disclosures.
VIDEO: Mortality rate after elective colorectal surgery hits 2%
SAN FRANCISCO– Patients undergoing elective colorectal surgery had an overall mortality rate of 1.7% after 30 days, an analysis of data from 65,716 patients showed.
Patients with significant preoperative morbidity had a significantly higher risk of dying after the surgery, Dr. Alodia Gabre-Kidan and her associates reported at the annual clinical congress of the American College of Surgeons.
In a video interview, Dr. Gabre-Kidan discusses the results of the retrospective study, including the especially high risk for patients with preoperative renal failure or heart failure. The findings should help clinicians better counsel patients who are considering elective colorectal surgery, said Dr. Gabre-Kidan of Columbia University, New York.
Dr. Gabre-Kidan reporting having no financial disclosures.
On Twitter @sherryboschert
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN FRANCISCO– Patients undergoing elective colorectal surgery had an overall mortality rate of 1.7% after 30 days, an analysis of data from 65,716 patients showed.
Patients with significant preoperative morbidity had a significantly higher risk of dying after the surgery, Dr. Alodia Gabre-Kidan and her associates reported at the annual clinical congress of the American College of Surgeons.
In a video interview, Dr. Gabre-Kidan discusses the results of the retrospective study, including the especially high risk for patients with preoperative renal failure or heart failure. The findings should help clinicians better counsel patients who are considering elective colorectal surgery, said Dr. Gabre-Kidan of Columbia University, New York.
Dr. Gabre-Kidan reporting having no financial disclosures.
On Twitter @sherryboschert
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN FRANCISCO– Patients undergoing elective colorectal surgery had an overall mortality rate of 1.7% after 30 days, an analysis of data from 65,716 patients showed.
Patients with significant preoperative morbidity had a significantly higher risk of dying after the surgery, Dr. Alodia Gabre-Kidan and her associates reported at the annual clinical congress of the American College of Surgeons.
In a video interview, Dr. Gabre-Kidan discusses the results of the retrospective study, including the especially high risk for patients with preoperative renal failure or heart failure. The findings should help clinicians better counsel patients who are considering elective colorectal surgery, said Dr. Gabre-Kidan of Columbia University, New York.
Dr. Gabre-Kidan reporting having no financial disclosures.
On Twitter @sherryboschert
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE ACS CLINICAL CONGRESS
Mismatch seen between trends in elective, emergent colectomies for diverticulitis
SAN FRANCISCO – Performance of more elective colectomies for diverticulitis in recent years does not seem to be preventing the need for emergent or urgent colectomies, investigators reported at the annual clinical congress of the American College of Surgeons.
The investigators analyzed data from the Comprehensive Hospital Abstract Reporting System (CHARS), a Washington State inpatient database, for the years 1987-2012.
Results showed that the age- and sex-adjusted rate of elective colectomies for diverticulitis increased from about 7 per 100,000 people to more than 20 per 100,000 people. But the rate of nonelective (emergent or urgent) colectomies for this indication remained fairly stable, if anything, increasing slightly.
“Despite 15 years of evolving guidelines to operate less, we are actually seeing a threefold increase in elective colectomy for diverticulitis. We did not see a decrease in emergency surgery during the same time,” commented lead investigator Dr. Vlad V. Simianu, a research fellow in the department of surgery, University of Washington, Seattle.
Several hypotheses might explain this disconnect, he proposed: a rising incidence of diverticulitis, increasing severity of disease (although data do not support this), or surgeons’ adoption of a lower threshold for elective colectomy. “In fact, if you look at where the biggest climb in elective surgery was, it was sort of in the early 2000s, which was the time contemporaneous to when the first laparoscopic colorectal randomized trials were being published, and training programs were incorporating laparoscopy into their training. That’s really where we see the biggest jump. So it may be that what we are seeing is an increase in this procedure [laparoscopic colectomy], the same as what we saw with laparoscopic cholecystectomy.”
“Whatever you choose ... to believe about what’s driving this, what remains certain going forward is that routine elective colectomy is not supported by the most recent guidelines [from the American Society of Colon and Rectal Surgeons], and actually these guidelines are for the first time recommending that the decision to offer elective surgery be individualized,” Dr. Simianu noted.
“Our group is actually doing some neat work in this space trying to identify which metrics should be used to individualize surgery, so we look forward to sharing that with you in the future,” he added.
Session comoderator Dr. James J. Mezhir of the University of Iowa, Iowa City, asked, “It may be difficult, but are you able to look at general surgeons versus those who are colorectal trained?”
The database does not specify surgeon training, according to Dr. Simianu, who disclosed that he had no relevant conflicts of interest. “What is interesting is that in Washington State, about half the surgery is done laparoscopically, in recent years at least. Whether laparoscopy is lowering the threshold, we know that colorectal surgeons are trained to do more laparoscopic colectomy maybe than their general surgeon counterparts.”
Another state database has provided some additional relevant information, he noted. “When laparoscopy first started being adopted, we saw a spike in right-sided colon surgery, which was previously a pretty rare colectomy for diverticulitis. So it is hard to know who is sort of driving this.”
Dr. Mezhir also wanted to know, “How generalizable is this across the United States? Have you looked at something like the nationwide inpatient sample or some other data set to say whether this is something you are seeing in Washington or is this something across the U.S.?”
Similar national trends have been reported in studies using that database, according to Dr. Simianu. “One of the things they point out is that there is a spike in diverticulitis in the younger population, which is something we are seeing in Washington State as well,” he noted.
Another session attendee noted that patients with acute diverticulitis sometimes have immediate procedures, such as drainage, but then go on to have colectomy not long afterward. Thus, he wondered about misclassification, asking, “Are you capturing it as elective although it’s sorted out as an acute episode?”
“That’s a great question, said another way, are we actually just delaying things into the elective category, as opposed to operating on them emergently?” Dr. Simianu replied. “It’s a little hard to tell. We do know that during this time, rates of percutaneous drainage have gone up. So it’s sort of the same conclusion that we are doing more elective surgery but we are not really preventing complications. But I think the crux of that is when these patients have complications and have emergency surgery, they have it on their first episode, 80%-90% of them. So offering a delayed operation ... to prevent [complications] where the likelihood is highest of having a complication, doesn’t seem to bear out in the data.”
SAN FRANCISCO – Performance of more elective colectomies for diverticulitis in recent years does not seem to be preventing the need for emergent or urgent colectomies, investigators reported at the annual clinical congress of the American College of Surgeons.
The investigators analyzed data from the Comprehensive Hospital Abstract Reporting System (CHARS), a Washington State inpatient database, for the years 1987-2012.
Results showed that the age- and sex-adjusted rate of elective colectomies for diverticulitis increased from about 7 per 100,000 people to more than 20 per 100,000 people. But the rate of nonelective (emergent or urgent) colectomies for this indication remained fairly stable, if anything, increasing slightly.
“Despite 15 years of evolving guidelines to operate less, we are actually seeing a threefold increase in elective colectomy for diverticulitis. We did not see a decrease in emergency surgery during the same time,” commented lead investigator Dr. Vlad V. Simianu, a research fellow in the department of surgery, University of Washington, Seattle.
Several hypotheses might explain this disconnect, he proposed: a rising incidence of diverticulitis, increasing severity of disease (although data do not support this), or surgeons’ adoption of a lower threshold for elective colectomy. “In fact, if you look at where the biggest climb in elective surgery was, it was sort of in the early 2000s, which was the time contemporaneous to when the first laparoscopic colorectal randomized trials were being published, and training programs were incorporating laparoscopy into their training. That’s really where we see the biggest jump. So it may be that what we are seeing is an increase in this procedure [laparoscopic colectomy], the same as what we saw with laparoscopic cholecystectomy.”
“Whatever you choose ... to believe about what’s driving this, what remains certain going forward is that routine elective colectomy is not supported by the most recent guidelines [from the American Society of Colon and Rectal Surgeons], and actually these guidelines are for the first time recommending that the decision to offer elective surgery be individualized,” Dr. Simianu noted.
“Our group is actually doing some neat work in this space trying to identify which metrics should be used to individualize surgery, so we look forward to sharing that with you in the future,” he added.
Session comoderator Dr. James J. Mezhir of the University of Iowa, Iowa City, asked, “It may be difficult, but are you able to look at general surgeons versus those who are colorectal trained?”
The database does not specify surgeon training, according to Dr. Simianu, who disclosed that he had no relevant conflicts of interest. “What is interesting is that in Washington State, about half the surgery is done laparoscopically, in recent years at least. Whether laparoscopy is lowering the threshold, we know that colorectal surgeons are trained to do more laparoscopic colectomy maybe than their general surgeon counterparts.”
Another state database has provided some additional relevant information, he noted. “When laparoscopy first started being adopted, we saw a spike in right-sided colon surgery, which was previously a pretty rare colectomy for diverticulitis. So it is hard to know who is sort of driving this.”
Dr. Mezhir also wanted to know, “How generalizable is this across the United States? Have you looked at something like the nationwide inpatient sample or some other data set to say whether this is something you are seeing in Washington or is this something across the U.S.?”
Similar national trends have been reported in studies using that database, according to Dr. Simianu. “One of the things they point out is that there is a spike in diverticulitis in the younger population, which is something we are seeing in Washington State as well,” he noted.
Another session attendee noted that patients with acute diverticulitis sometimes have immediate procedures, such as drainage, but then go on to have colectomy not long afterward. Thus, he wondered about misclassification, asking, “Are you capturing it as elective although it’s sorted out as an acute episode?”
“That’s a great question, said another way, are we actually just delaying things into the elective category, as opposed to operating on them emergently?” Dr. Simianu replied. “It’s a little hard to tell. We do know that during this time, rates of percutaneous drainage have gone up. So it’s sort of the same conclusion that we are doing more elective surgery but we are not really preventing complications. But I think the crux of that is when these patients have complications and have emergency surgery, they have it on their first episode, 80%-90% of them. So offering a delayed operation ... to prevent [complications] where the likelihood is highest of having a complication, doesn’t seem to bear out in the data.”
SAN FRANCISCO – Performance of more elective colectomies for diverticulitis in recent years does not seem to be preventing the need for emergent or urgent colectomies, investigators reported at the annual clinical congress of the American College of Surgeons.
The investigators analyzed data from the Comprehensive Hospital Abstract Reporting System (CHARS), a Washington State inpatient database, for the years 1987-2012.
Results showed that the age- and sex-adjusted rate of elective colectomies for diverticulitis increased from about 7 per 100,000 people to more than 20 per 100,000 people. But the rate of nonelective (emergent or urgent) colectomies for this indication remained fairly stable, if anything, increasing slightly.
“Despite 15 years of evolving guidelines to operate less, we are actually seeing a threefold increase in elective colectomy for diverticulitis. We did not see a decrease in emergency surgery during the same time,” commented lead investigator Dr. Vlad V. Simianu, a research fellow in the department of surgery, University of Washington, Seattle.
Several hypotheses might explain this disconnect, he proposed: a rising incidence of diverticulitis, increasing severity of disease (although data do not support this), or surgeons’ adoption of a lower threshold for elective colectomy. “In fact, if you look at where the biggest climb in elective surgery was, it was sort of in the early 2000s, which was the time contemporaneous to when the first laparoscopic colorectal randomized trials were being published, and training programs were incorporating laparoscopy into their training. That’s really where we see the biggest jump. So it may be that what we are seeing is an increase in this procedure [laparoscopic colectomy], the same as what we saw with laparoscopic cholecystectomy.”
“Whatever you choose ... to believe about what’s driving this, what remains certain going forward is that routine elective colectomy is not supported by the most recent guidelines [from the American Society of Colon and Rectal Surgeons], and actually these guidelines are for the first time recommending that the decision to offer elective surgery be individualized,” Dr. Simianu noted.
“Our group is actually doing some neat work in this space trying to identify which metrics should be used to individualize surgery, so we look forward to sharing that with you in the future,” he added.
Session comoderator Dr. James J. Mezhir of the University of Iowa, Iowa City, asked, “It may be difficult, but are you able to look at general surgeons versus those who are colorectal trained?”
The database does not specify surgeon training, according to Dr. Simianu, who disclosed that he had no relevant conflicts of interest. “What is interesting is that in Washington State, about half the surgery is done laparoscopically, in recent years at least. Whether laparoscopy is lowering the threshold, we know that colorectal surgeons are trained to do more laparoscopic colectomy maybe than their general surgeon counterparts.”
Another state database has provided some additional relevant information, he noted. “When laparoscopy first started being adopted, we saw a spike in right-sided colon surgery, which was previously a pretty rare colectomy for diverticulitis. So it is hard to know who is sort of driving this.”
Dr. Mezhir also wanted to know, “How generalizable is this across the United States? Have you looked at something like the nationwide inpatient sample or some other data set to say whether this is something you are seeing in Washington or is this something across the U.S.?”
Similar national trends have been reported in studies using that database, according to Dr. Simianu. “One of the things they point out is that there is a spike in diverticulitis in the younger population, which is something we are seeing in Washington State as well,” he noted.
Another session attendee noted that patients with acute diverticulitis sometimes have immediate procedures, such as drainage, but then go on to have colectomy not long afterward. Thus, he wondered about misclassification, asking, “Are you capturing it as elective although it’s sorted out as an acute episode?”
“That’s a great question, said another way, are we actually just delaying things into the elective category, as opposed to operating on them emergently?” Dr. Simianu replied. “It’s a little hard to tell. We do know that during this time, rates of percutaneous drainage have gone up. So it’s sort of the same conclusion that we are doing more elective surgery but we are not really preventing complications. But I think the crux of that is when these patients have complications and have emergency surgery, they have it on their first episode, 80%-90% of them. So offering a delayed operation ... to prevent [complications] where the likelihood is highest of having a complication, doesn’t seem to bear out in the data.”
AT THE ACS CLINICAL CONGRESS
Key clinical point: An increase in elective colectomies has not been accompanied by a decrease in emergent/urgent colectomies.
Major finding: The adjusted rate of elective colectomies tripled, but the rate of emergent/urgent colectomies remained stable.
Data source: A cohort study of colectomies for diverticulitis in Washington State between 1987 and 2012.
Disclosures: Dr. Simianu disclosed that he had no relevant conflicts of interest.
Antibiotic bowel prep significantly reduces colon surgery infections
SAN FRANCISCO – Oral antibiotics with mechanical bowel preparation significantly reduce surgical site infections, length of hospital stay, and readmissions in both open and laparoscopic elective colorectal surgery, according to a review of 8,415 cases in the National Surgery Quality Improvement Program.
It “should be adopted for elective colorectal surgery,” concluded the investigators, led by colorectal surgeon Melanie Morris of the University of Alabama at Birmingham.
A quarter of the patients had no bowel prep, 45% had mechanical prep alone – GoLytely or another laxative, and 30% received both oral antibiotics and mechanical prep. Partial colectomy and sigmoid resections were the most common procedures, generally for neoplasms or diverticulitis.
Overall, 15% of the no-prep group, 12% of the mechanical prep group, and 6.5% of the oral antibiotic plus mechanical prep group developed subsequent surgical site infections (SSIs), a statistically significant difference. Results were similar when broken down into superficial, deep wound, and organ space infections (J. Am. Coll. Surg. 2014:219;S18-19).
On multivariate analysis and after adjusting for diabetes, smoking, age, and other potential confounders, the antibiotic group was 54% less likely than the no-prep group to develop an SSI (odds ratio 0.46), 26% less likely to be readmitted (OR 0.74), and more likely to leave the hospital earlier, at about 4 days instead of 5 (OR 0.90). SSIs and readmissions were only slightly less likely in the mechanical prep–only group, compared with the no-prep group, and there was no difference in length of stay.
There were fewer anastomotic leaks, fewer cases of sepsis, less significant bleeding, and other benefits for oral antibiotics with mechanical preparation. In addition, there was a protective effect against 30-day mortality in open procedures.
“Even in the minimally invasive group,” about two-thirds of patients, “oral antibiotic prep … was protective for surgical site infections,” Dr. Morris said at the annual clinical congress of the American College of Surgeons.
Still, bowel prep remains “a very controversial topic.” Nationwide some surgeons prep, some don’t. “People have very strong beliefs that may or may not be rooted in the data,” she said.
There are concerns about fluid and electrolyte disturbances, Clostridium difficile overgrowth, and other potential problems. Plus, mechanical preparation hasn’t worked any better than placebo in recent studies, but many of those studies didn’t include oral antibiotics. Effective bowel prep includes both, Dr. Morris said.
It’s possible that confounders might have been at work in the Alabama study. Perhaps surgeons shied away from bowel prep in older, sicker patients, but the rate of acute renal injury was the same in all three study groups at about 0.9%, suggesting similar background comorbidities.
“I don’t think confounding issues” explain the findings. Previously, “we’ve shown the benefit of oral antibiotic bowel prep in a [Veterans Affairs] cohort, and now we’ve shown it in this national cohort,” Dr. Morris said (Dis. Colon. Rectum 2012;55:1160-6).
After a marked reduction in surgical site infections in the VA study, “we changed our practices. We now do mechanical and oral antibiotic preps and have seen a 50% reduction in our SSI rate. We don’t feel like we are doing anything else differently,” she said.
Dr. Morris has no disclosures.
SAN FRANCISCO – Oral antibiotics with mechanical bowel preparation significantly reduce surgical site infections, length of hospital stay, and readmissions in both open and laparoscopic elective colorectal surgery, according to a review of 8,415 cases in the National Surgery Quality Improvement Program.
It “should be adopted for elective colorectal surgery,” concluded the investigators, led by colorectal surgeon Melanie Morris of the University of Alabama at Birmingham.
A quarter of the patients had no bowel prep, 45% had mechanical prep alone – GoLytely or another laxative, and 30% received both oral antibiotics and mechanical prep. Partial colectomy and sigmoid resections were the most common procedures, generally for neoplasms or diverticulitis.
Overall, 15% of the no-prep group, 12% of the mechanical prep group, and 6.5% of the oral antibiotic plus mechanical prep group developed subsequent surgical site infections (SSIs), a statistically significant difference. Results were similar when broken down into superficial, deep wound, and organ space infections (J. Am. Coll. Surg. 2014:219;S18-19).
On multivariate analysis and after adjusting for diabetes, smoking, age, and other potential confounders, the antibiotic group was 54% less likely than the no-prep group to develop an SSI (odds ratio 0.46), 26% less likely to be readmitted (OR 0.74), and more likely to leave the hospital earlier, at about 4 days instead of 5 (OR 0.90). SSIs and readmissions were only slightly less likely in the mechanical prep–only group, compared with the no-prep group, and there was no difference in length of stay.
There were fewer anastomotic leaks, fewer cases of sepsis, less significant bleeding, and other benefits for oral antibiotics with mechanical preparation. In addition, there was a protective effect against 30-day mortality in open procedures.
“Even in the minimally invasive group,” about two-thirds of patients, “oral antibiotic prep … was protective for surgical site infections,” Dr. Morris said at the annual clinical congress of the American College of Surgeons.
Still, bowel prep remains “a very controversial topic.” Nationwide some surgeons prep, some don’t. “People have very strong beliefs that may or may not be rooted in the data,” she said.
There are concerns about fluid and electrolyte disturbances, Clostridium difficile overgrowth, and other potential problems. Plus, mechanical preparation hasn’t worked any better than placebo in recent studies, but many of those studies didn’t include oral antibiotics. Effective bowel prep includes both, Dr. Morris said.
It’s possible that confounders might have been at work in the Alabama study. Perhaps surgeons shied away from bowel prep in older, sicker patients, but the rate of acute renal injury was the same in all three study groups at about 0.9%, suggesting similar background comorbidities.
“I don’t think confounding issues” explain the findings. Previously, “we’ve shown the benefit of oral antibiotic bowel prep in a [Veterans Affairs] cohort, and now we’ve shown it in this national cohort,” Dr. Morris said (Dis. Colon. Rectum 2012;55:1160-6).
After a marked reduction in surgical site infections in the VA study, “we changed our practices. We now do mechanical and oral antibiotic preps and have seen a 50% reduction in our SSI rate. We don’t feel like we are doing anything else differently,” she said.
Dr. Morris has no disclosures.
SAN FRANCISCO – Oral antibiotics with mechanical bowel preparation significantly reduce surgical site infections, length of hospital stay, and readmissions in both open and laparoscopic elective colorectal surgery, according to a review of 8,415 cases in the National Surgery Quality Improvement Program.
It “should be adopted for elective colorectal surgery,” concluded the investigators, led by colorectal surgeon Melanie Morris of the University of Alabama at Birmingham.
A quarter of the patients had no bowel prep, 45% had mechanical prep alone – GoLytely or another laxative, and 30% received both oral antibiotics and mechanical prep. Partial colectomy and sigmoid resections were the most common procedures, generally for neoplasms or diverticulitis.
Overall, 15% of the no-prep group, 12% of the mechanical prep group, and 6.5% of the oral antibiotic plus mechanical prep group developed subsequent surgical site infections (SSIs), a statistically significant difference. Results were similar when broken down into superficial, deep wound, and organ space infections (J. Am. Coll. Surg. 2014:219;S18-19).
On multivariate analysis and after adjusting for diabetes, smoking, age, and other potential confounders, the antibiotic group was 54% less likely than the no-prep group to develop an SSI (odds ratio 0.46), 26% less likely to be readmitted (OR 0.74), and more likely to leave the hospital earlier, at about 4 days instead of 5 (OR 0.90). SSIs and readmissions were only slightly less likely in the mechanical prep–only group, compared with the no-prep group, and there was no difference in length of stay.
There were fewer anastomotic leaks, fewer cases of sepsis, less significant bleeding, and other benefits for oral antibiotics with mechanical preparation. In addition, there was a protective effect against 30-day mortality in open procedures.
“Even in the minimally invasive group,” about two-thirds of patients, “oral antibiotic prep … was protective for surgical site infections,” Dr. Morris said at the annual clinical congress of the American College of Surgeons.
Still, bowel prep remains “a very controversial topic.” Nationwide some surgeons prep, some don’t. “People have very strong beliefs that may or may not be rooted in the data,” she said.
There are concerns about fluid and electrolyte disturbances, Clostridium difficile overgrowth, and other potential problems. Plus, mechanical preparation hasn’t worked any better than placebo in recent studies, but many of those studies didn’t include oral antibiotics. Effective bowel prep includes both, Dr. Morris said.
It’s possible that confounders might have been at work in the Alabama study. Perhaps surgeons shied away from bowel prep in older, sicker patients, but the rate of acute renal injury was the same in all three study groups at about 0.9%, suggesting similar background comorbidities.
“I don’t think confounding issues” explain the findings. Previously, “we’ve shown the benefit of oral antibiotic bowel prep in a [Veterans Affairs] cohort, and now we’ve shown it in this national cohort,” Dr. Morris said (Dis. Colon. Rectum 2012;55:1160-6).
After a marked reduction in surgical site infections in the VA study, “we changed our practices. We now do mechanical and oral antibiotic preps and have seen a 50% reduction in our SSI rate. We don’t feel like we are doing anything else differently,” she said.
Dr. Morris has no disclosures.
AT THE ACS CLINICAL CONGRESS
Key clinical point: Effective bowel prep must include oral antibiotics as well as laxatives.
Major finding: Prepping colorectal surgery patients with both oral antibiotics and laxatives halves the risk of surgery site infections (OR 0.46, 95% CI 0.36-0.59, P < .001),
Data source: Review of 8,415 cases in the National Surgery Quality Improvement Program
Disclosures: Dr. Morris has no relevant financial disclosures.