User login
To better manage cancer symptoms
A striking change in cancer medicine over the past several decades has been the rising amplitude of the voices of cancer patients and survivors and their loved ones. Increasingly, they have organized as advocates for better cancer treatment, better research, and better attention to the experience of those directly affected by cancer.
At the same time, despite a somewhat delirious period in the 1970s when it was expected that cancer could be cured, or at least that the mortality rate could be cut in half, it has become clear that progress in eliminating the disease is a long, slow slog with no guarantee of success. Nowhere is this lack of major progress clearer than in the US Food and Drug Administration’s decision a few years ago to approve a drug for pancreatic cancer based on a 10-day increase in median survival.1
These two factors, the rising voices of those affected by cancer and the failure of cancer research to deliver a cure, have helped fuel a dramatic increase in the attention paid to the symptoms caused by cancer and its treatments. Improving quality of life has become recognized as an additional important goal worthy of rigorous study. Alleviating symptoms is often the most we have to offer patients with advanced cancer, and palliative medicine services are now found at many if not all major cancer centers. To help ensure a supply of well-trained palliative medicine doctors, accredited palliative medicine fellowships have been started at institutions around the country. These programs produce physicians with a higher level of expertise in managing pain, nausea, constipation, fatigue, psychosocial distress, dyspnea, and a wide variety of other symptoms. And while the needs of cancer patients have helped accelerate the growth of palliative medicine, the specialty has a role to play with almost any patient with intractable symptoms, regardless of the nature of the underlying disease.
With the growing recognition of a need to better manage cancer patients’ symptoms, research in palliative care has grown rapidly, and an evidence-based approach to symptom management has become possible. Meanwhile, a variety of substantial advances has occurred. For instance, modern antiemetics have dramatically reduced chemotherapy-related vomiting, and long-acting narcotics have allowed patients to achieve better pain control with milder side effects.
In other areas such as cancer-related fatigue or chemotherapy-induced neuropathy, there is very limited evidence that our interventions are effective at alleviating symptoms or improving quality of life. In these and a number of other areas, more research and better treatments are urgently needed.
In order to keep our readers up to date on the progress that is being made in palliative medicine for cancer patients, the Cleveland Clinic Journal of Medicine is presenting a series of articles on the topic. The series begins this issue with an article on the principles of symptom management and a review of the current state of knowledge about alleviating fatigue, nausea, constipation, and dyspnea. Future articles will focus on pain and bowel obstruction.
Our goal is to give our readers practical information that will help them provide better symptom management for their patients, particularly their cancer patients. This series will deal with one of the most important problems of cancer medicine.
- Moore MJ, Goldstein D, Hamm J, et al. Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: a phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol 2007; 25:1960–1966.
A striking change in cancer medicine over the past several decades has been the rising amplitude of the voices of cancer patients and survivors and their loved ones. Increasingly, they have organized as advocates for better cancer treatment, better research, and better attention to the experience of those directly affected by cancer.
At the same time, despite a somewhat delirious period in the 1970s when it was expected that cancer could be cured, or at least that the mortality rate could be cut in half, it has become clear that progress in eliminating the disease is a long, slow slog with no guarantee of success. Nowhere is this lack of major progress clearer than in the US Food and Drug Administration’s decision a few years ago to approve a drug for pancreatic cancer based on a 10-day increase in median survival.1
These two factors, the rising voices of those affected by cancer and the failure of cancer research to deliver a cure, have helped fuel a dramatic increase in the attention paid to the symptoms caused by cancer and its treatments. Improving quality of life has become recognized as an additional important goal worthy of rigorous study. Alleviating symptoms is often the most we have to offer patients with advanced cancer, and palliative medicine services are now found at many if not all major cancer centers. To help ensure a supply of well-trained palliative medicine doctors, accredited palliative medicine fellowships have been started at institutions around the country. These programs produce physicians with a higher level of expertise in managing pain, nausea, constipation, fatigue, psychosocial distress, dyspnea, and a wide variety of other symptoms. And while the needs of cancer patients have helped accelerate the growth of palliative medicine, the specialty has a role to play with almost any patient with intractable symptoms, regardless of the nature of the underlying disease.
With the growing recognition of a need to better manage cancer patients’ symptoms, research in palliative care has grown rapidly, and an evidence-based approach to symptom management has become possible. Meanwhile, a variety of substantial advances has occurred. For instance, modern antiemetics have dramatically reduced chemotherapy-related vomiting, and long-acting narcotics have allowed patients to achieve better pain control with milder side effects.
In other areas such as cancer-related fatigue or chemotherapy-induced neuropathy, there is very limited evidence that our interventions are effective at alleviating symptoms or improving quality of life. In these and a number of other areas, more research and better treatments are urgently needed.
In order to keep our readers up to date on the progress that is being made in palliative medicine for cancer patients, the Cleveland Clinic Journal of Medicine is presenting a series of articles on the topic. The series begins this issue with an article on the principles of symptom management and a review of the current state of knowledge about alleviating fatigue, nausea, constipation, and dyspnea. Future articles will focus on pain and bowel obstruction.
Our goal is to give our readers practical information that will help them provide better symptom management for their patients, particularly their cancer patients. This series will deal with one of the most important problems of cancer medicine.
A striking change in cancer medicine over the past several decades has been the rising amplitude of the voices of cancer patients and survivors and their loved ones. Increasingly, they have organized as advocates for better cancer treatment, better research, and better attention to the experience of those directly affected by cancer.
At the same time, despite a somewhat delirious period in the 1970s when it was expected that cancer could be cured, or at least that the mortality rate could be cut in half, it has become clear that progress in eliminating the disease is a long, slow slog with no guarantee of success. Nowhere is this lack of major progress clearer than in the US Food and Drug Administration’s decision a few years ago to approve a drug for pancreatic cancer based on a 10-day increase in median survival.1
These two factors, the rising voices of those affected by cancer and the failure of cancer research to deliver a cure, have helped fuel a dramatic increase in the attention paid to the symptoms caused by cancer and its treatments. Improving quality of life has become recognized as an additional important goal worthy of rigorous study. Alleviating symptoms is often the most we have to offer patients with advanced cancer, and palliative medicine services are now found at many if not all major cancer centers. To help ensure a supply of well-trained palliative medicine doctors, accredited palliative medicine fellowships have been started at institutions around the country. These programs produce physicians with a higher level of expertise in managing pain, nausea, constipation, fatigue, psychosocial distress, dyspnea, and a wide variety of other symptoms. And while the needs of cancer patients have helped accelerate the growth of palliative medicine, the specialty has a role to play with almost any patient with intractable symptoms, regardless of the nature of the underlying disease.
With the growing recognition of a need to better manage cancer patients’ symptoms, research in palliative care has grown rapidly, and an evidence-based approach to symptom management has become possible. Meanwhile, a variety of substantial advances has occurred. For instance, modern antiemetics have dramatically reduced chemotherapy-related vomiting, and long-acting narcotics have allowed patients to achieve better pain control with milder side effects.
In other areas such as cancer-related fatigue or chemotherapy-induced neuropathy, there is very limited evidence that our interventions are effective at alleviating symptoms or improving quality of life. In these and a number of other areas, more research and better treatments are urgently needed.
In order to keep our readers up to date on the progress that is being made in palliative medicine for cancer patients, the Cleveland Clinic Journal of Medicine is presenting a series of articles on the topic. The series begins this issue with an article on the principles of symptom management and a review of the current state of knowledge about alleviating fatigue, nausea, constipation, and dyspnea. Future articles will focus on pain and bowel obstruction.
Our goal is to give our readers practical information that will help them provide better symptom management for their patients, particularly their cancer patients. This series will deal with one of the most important problems of cancer medicine.
- Moore MJ, Goldstein D, Hamm J, et al. Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: a phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol 2007; 25:1960–1966.
- Moore MJ, Goldstein D, Hamm J, et al. Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: a phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol 2007; 25:1960–1966.
Symptom management: An important part of cancer care
Cancer patients experience many distressing symptoms during the course of their illness. In addition to pain, they commonly suffer from fatigue, anorexia, constipation, dyspnea, nausea, and vomiting.1
Although it is important to diagnose and manage the cancer itself, it is also the physician’s duty to recognize and effectively treat associated symptoms, regardless of the outcome of the underlying disease.
Some of the symptoms are due to the underlying disease, but some are iatrogenic, as many medical interventions have predictable adverse effects, such as nausea and vomiting with chemotherapy or constipation with opioids.
Symptoms of advanced cancer become chronic, and patients usually rate them as moderate or severe.1 Unrelieved suffering causes demoralization and may quickly impair quality of life.2
Understanding the principles of symptom management may help optimize palliation and improve quality of life. In this paper, we outline an approach to the management of cancer-related symptoms.
A HEAVY BURDEN OF SYMPTOMS
In patients with advanced cancer, the prevalence rates of various symptoms are approximately as follows1,3:
- Pain 89%
- Fatigue 69%
- Weakness 66%
- Anorexia 66%
- Lack of energy 61%
- Nausea 60%
- Dry mouth 57%
- Constipation 52%
- Early satiety 51%
- Dyspnea 50%
- Vomiting 30%.
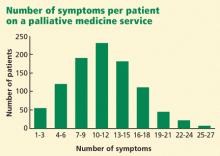
Furthermore, patients with advanced cancer typically have multiple concurrent symptoms. In a survey of patients in a palliative medicine service at our hospital,4 we found that the median number of symptoms per patient was 10 (range 0–25) (Figure 1).
PRINCIPLES OF SYMPTOM MANAGEMENT
Show an interest in the patient’s symptoms. Many patients with advanced cancer believe that suffering is an inevitable part of the disease or of its treatment.
Ask patients about their symptoms in a positive and detailed fashion, starting with open-ended questions and following up with specific questions. Patients may underreport their symptoms or may not mention them if not asked directly. In the survey of palliative care patients at our hospital mentioned above,4 the median number of volunteered symptoms was only 1 (range 0–6), whereas a median of 10 were found by systematic assessment.
The examiner should clarify when necessary and recognize that a layperson’s language may not directly translate to medical language. For example, a patient may not understand the term “anorexia.” Furthermore, “loss of appetite” may mean nausea, vomiting, constipation, or early satiety. “Numbness” may mean a loss of sensation or a pins-and-needles sensation. Symptoms should also be quantified using a consistent measure (ie, numerical or categorical) to facilitate monitoring.
Prioritize the symptoms. Advanced cancer is accompanied by multiple symptoms. Assess which ones are most bothersome, and where therapy should be directed first.
Try to understand the pathophysiology behind the symptom. When possible, choose a drug treatment that targets the likely underlying cause. Nausea and vomiting, for example, can be secondary to gastric outlet obstruction, hypercalcemia, increased intracranial pressure, esophagitis, opioid use, or constipation.
Be specific about the drug, dosing, timing, and route, and keep it simple. If a regimen is cumbersome, compliance suffers. It is better to start one medication for the most bothersome symptom or symptoms and make some progress than it is to overwhelm the patient with a complex list of drugs. Sustained-release formulations are often useful. It is unrealistic to expect most patients to take a medication every 4 hours around the clock. Try the most cost-effective remedies first, and attempt to use one drug that may address multiple symptoms. For example, dexamethasone may have positive effects on energy, pain, and appetite.
Use ‘rescue dosing.’ Rescue drugs are important for expected symptom exacerbations in those on sustained medication. This approach increases efficacy and minimizes adverse effects. In most cases, the rescue medication should be the same as the regularly scheduled one. For example, a prescription to treat nausea may read “metoclopramide (Reglan) 10 mg by mouth before meals and at bedtime and every 4 hours as needed to treat nausea or vomiting.”
Consider the patient’s age and fragility, the cost of the drug, and anticipated adverse effects. Oral or transdermal preparations are preferable to parenteral ones with regard to convenience and compliance, although many transdermal preparations are costly. If parenteral dosing is necessary, the subcutaneous route is an alternative to the intravenous route.
Discontinue drugs that are ineffective or unnecessary. This may help compliance and diminish adverse effects.
Make one change at a time so the response to that change is clear. Titrate one drug to its effective dose, to its maximum dose, or to a level of intolerability before considering another. If one drug of a class is ineffective, another drug in the same class may work.
Reassess often. A follow-up phone call or office visit in 1 to 2 weeks is appropriate. The symptoms of advanced cancer are often progressive, so regular evaluation is important, even if symptoms are controlled on stable drug regimens. Instructions should be both verbal and written and should be communicated to patients and any involved caregiver to ensure compliance. Have a “plan B” if the first plan is ineffective.
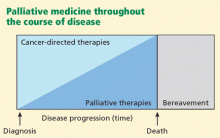
A challenging and important part of symptom management is to assess the goals of care. Every intervention is not appropriate for every patient. Which therapies are used depends on the stage of the disease, the available disease-modifying treatments, and the patient’s condition and preferences. Patients and their loved ones should be engaged in discussions about goals of care early in the disease and should be included in medical decision-making. Both curative treatment and palliative treatment are important, but palliation plays a bigger role towards the later stages of advanced cancer (Figure 2).
CANCER-RELATED FATIGUE: COMMON BUT NOT INEVITABLE
Most cancer patients report fatigue. Although it is one of the most common symptoms in advanced cancer,5 it is not necessarily inevitable or untreatable.6
Cancer-related fatigue is multidimensional and develops over time, diminishing energy, mental capacity, and psychological condition.7 Patients may report feeling tired or being unable to complete their activities of daily living. People who were previously very active may be frustrated by their inability to participate in favorite leisure activities, which has a big impact on quality of life. Fatigue can be physical, emotional, or mental. It is important to distinguish physical weakness from dyspnea on exertion, which is commonly reported as fatigue. Depression may also cause or exacerbate fatigue.
Unlike fatigue in the general population, cancer-related fatigue does not improve with rest, and patients often report large amounts of unrestorative sleep.
Look for reversible causes of fatigue
First, conduct a thorough assessment to identify any reversible causes, such as:
- Anemia
- Insomnia, sleep disturbance
- Malnutrition
- Pain
- Depression
- Medical comorbidities: renal, cardiac, or pulmonary disease
- Hypothyroidism
- Hypogonadism.
In many cases, however, a reversible cause cannot be found.
Treating cancer-related fatigue
Nonpharmacologic interventions have been evaluated for this application, but evidence of efficacy is limited and mixed. The National Cancer Comprehensive Network guidelines8 suggest that energy conservation and education about cancer-related fatigue are central to management. Patients should be advised that fatigue has a fluctuating course and that they have a limited pool of energy, which they should conserve and use judiciously.
In a meta-analysis by Schmitz et al,9 physical activity interventions were found to be beneficial. Sixty-three percent of those studied were undergoing active treatment, so whether this population reflects advanced cancer is unclear. A small pilot study in advanced cancer found a trend toward benefit with exercise.10
Comment. The strategies of rest and exercise are complementary. The key point is to plan them per personal preference.
Psychostimulants include methylphenidate (Ritalin). A randomized placebo-controlled trial in patients with acquired immunodeficiency syndrome (AIDS) found methylphenidate 15 to 60 mg/day to have a positive effect.11 Prospective studies have shown similar results in cancer patients,12 and a Cochrane review in 2008 showed a small but significant benefit in cancer-related fatigue.13
Methylphenidate is usually started at a dose of 5 mg given at 8:00 am and at noon, and then titrated. Benefit, when experienced, is typically noted within 24 to 48 hours. Possible adverse effects include anorexia, insomnia, anxiety, confusion, tremor, and tachycardia.
Stimulants should be used with caution in patients with cardiac disease or delirium.
Modafinil (Provigil), a nonstimulant agent, has been less studied, but it may also help.14,15 The usual dosage is 50 to 200 mg daily.
Corticosteroids may have a role in advanced cancer, as suggested by anecdotal reports.16 They should be used judiciously, as their adverse effects (insomnia, muscle wasting, edema) are themselves burdensome and may outweigh their benefits.
ANOREXIA CAN BE DISTRESSING TO THE FAMILY AND THE PATIENT
Most patients with advanced cancer experience anorexia, which is a marker of poor prognosis.1
Appetite loss may occur in isolation or as a part of the anorexia-cachexia syndrome. This syndrome is a wasting state seen in chronic, advanced diseases including cancer, AIDS, chronic obstructive pulmonary disease, chronic renal insufficiency, and congestive heart failure.17 The associated weight loss is involuntary and includes both muscle and fat.
Appetite loss alone is usually not bothersome. In fact, anorexia frequently causes more distress to the family than to the patient.18 The ramifications of decreased appetite, on the other hand, can be devastating. Decreased caloric intake coupled with the hypermetabolic state of malignancy leads to rapid, dramatic changes in body habitus. This outward sign of the ravages of cancer can be psychologically damaging to patients and their loved ones as they contemplate advanced disease and limited life expectancy. They may be concerned about starvation, in which case education about and attempts to normalize the anorexia-cachexia syndrome are essential.
Look for reversible causes of anorexia
The first step in the management of anorexia is to identify any reversible causes, such as:
- Stomatitis
- Constipation
- Uncontrolled severe symptoms such as pain or dyspnea
- Delirium
- Nausea, vomiting
- Depression
- Gastroparesis.
Managing cancer-related anorexia
Nonpharmacologic measures include nutritional counseling and increased physical activity. Patients may be counseled to eat calorie-dense foods and supplemental high-calorie, high-protein, high-fat drinks. Some may be able to take advantage of a diurnal variation in appetite, usually an increased appetite in the morning.
Megestrol acetate (Megace) improved appetite and induced weight gain when used in a dosage of 800 mg daily in a randomized controlled trial in AIDS patients.19 Case studies have shown doses as low as 80 to 160 mg daily to be beneficial.20 Most of the added weight is fat, not lean muscle mass. Unfortunately, the addition of testosterone to megestrol did not increase the accumulation of lean muscle mass in another randomized trial.21 But the addition of olanzapine (Zyprexa) to megestrol was associated with improved appetite and weight gain in a significant percentage of advanced cancer patients.22 Rates of adverse effects with megestrol are low; the most significant adverse effect is thromboembolism.
Corticosteroids. While much of the support for corticosteroids is anecdotal, a prospective study of dexamethasone 4 to 16 mg daily showed improvement in several symptoms, including appetite.23 Because of the multiple adverse effects of corticosteroids, careful attention to dose, duration, and tolerability is essential. Corticosteroids should be discontinued if the desired positive effects are not observed within 3 to 5 days. If prolonged survival is expected, wean to the lowest effective dose.
Cannabinoids. Dronabinol (Marinol), a synthetic formulation of delta-9-tetrahydrocannabinol (THC), the active agent of marijuana, has been beneficial in AIDS anorexia. Fewer studies have been done in advanced cancer.
In a small, open-label case series, doses of 7.5 to 15 mg of dronabinol daily improved appetite and were well tolerated.24 On the other hand, in a multicenter, randomized, double-blind, placebo-controlled trial, neither cannabis extract nor THC (5 mg daily) significantly improved appetite over a 6-week period.25
A large randomized study found megestrol acetate 800 mg to be superior to dronabinol 5 mg daily for treating anorexia.26
Neurotoxicity, anxiety, nervousness, dizziness, euphoria, and somnolence from dronabinol can be severe and intolerable for some.
Enteral tube feeding and parenteral nutrition do not improve survival or comfort in terminally ill patients.27 On the contrary, they are associated with complications, including aspiration pneumonia, sepsis, abdominal pain, vomiting, and diarrhea. Nevertheless, in some patients with mechanical impediments to nutrition (eg, esophageal fistula, obstruction, or proximal small bowel obstruction) or in those who are hungry and unable to take food by mouth, tube feeding may be appropriate.
CONSTIPATION SHOULD BE ANTICIPATED, AND PREVENTED IF POSSIBLE
Constipation is variably defined by patients and health care professionals, but it usually includes components of the Rome II criteria, ie, two or more of the following symptoms28:
- Straining at least 25% of the time
- Hard stools at least 25% of the time
- Incomplete evacuation at least 25% of the time
- Two or fewer bowel movements per week.
These criteria were intended to describe functional constipation in a healthy population.27
More than 50% of patients with advanced cancer report constipation,1 and in those on opioids, the scope of the problem is larger. In addition to binding central nervous system receptors to mediate pain perception, opioids bind systemic receptors including those in the gut. As a result, opioids interfere with smooth muscle tone and contractility, lengthen transit time, promote dry stools, and increase anal sphincter tone.29 A nursing study found that when patients taking opioids were screened for constipation, 95% identified it as the major adverse effect of their pain regimen.30
Multiple causes of constipation
Factors that can cause or contribute to constipation include:
- Dietary factors such as a generally low intake of food, and specifically of fiber
- Inactivity
- Confusion
- Dehydration
- Intestinal obstruction
- Cormorbidities such as diabetes mellitus, hypothyroidism, hypercalcemia
- Uncomfortable toilet arrangements
- Drugs such as opioids (as noted above), anticholinergics, antihypertensives, antacids, diuretics, and iron supplements.
Take a proactive approach to constipation
Constipation is expected in a number of clinical scenarios, such as with the use of opioids or with limited mobility. Patients often attribute constipation to diminished oral intake. But despite low oral intake, regular, smaller-caliber bowel movements are important to ensure that sloughed bowel endothelium and bacteria are eliminated.
Although little evidence supports the use of one standard bowel regimen, prevention is essential. The goal is a soft bowel movement every 1 to 2 days. Constipation prophylaxis should be started at the initiation of any regular opioid regimen. Encouraging physical activity and oral fluid intake and creating a favorable environment for elimination may also help manage constipation.
In our practice, we use a softening agent such as docusate sodium (Colace) 100 mg twice daily, and add a laxative agent such as senna (Senokot) or a magnesium-based osmotic agent as needed. Bulking agents such as over-the-counter fiber supplements should be used with caution in opioid-related constipation. If there has been no bowel movement for 48 hours, a rectal suppository or enema is used. Suppositories or enemas can be scheduled regularly for bedbound patients with chronic constipation.
Methylnaltrexone (Relistor), a mu-opioid antagonist, is a new agent that blocks peripheral opioid receptors in the gut. In a randomized study of 133 patients, methylnaltrexone produced laxation within 4 hours of administration in 48%.31 This methylated, charged compound does not significantly cross the blood-brain barrier and therefore does not interfere with analgesia or cause opioid withdrawal. The dose is 8 mg or 12 mg subcutaneously (based on weight), which can be repeated in 48 hours. If laxation does not occur after one to three doses, other causes of constipation should be explored.
Methylnaltrexone is contraindicated in patients with bowel obstruction, even if the obstruction is thought to be secondary to opioids. Adverse effects include abdominal pain, flatulence, and nausea.
NAUSEA AND VOMITING: NOT ALWAYS DUE TO CHEMOTHERAPY
Nausea (the sensation of the need to vomit) and vomiting (the forceful expulsion of gastric contents) are common symptoms in advanced cancer and are not necessarily related to chemotherapy or radiation therapy. About 60% of cancer patients have nausea, and about 30% vomit.32 Both symptoms are very distressing and diminish quality of life.
Look for potentially reversible causes of nausea and vomiting
Identifying the cause, which is sometimes reversible, may help direct treatment. Potentially reversible causes include:
- Drugs
- Uremia
- Infection
- Anxiety
- Constipation
- Gastric irritation
- Proximal gastrointestinal obstruction.
In a prospective study of 121 patients with advanced cancer, the most common causes of nausea and vomiting were impaired gastric emptying, chemical and metabolic factors (drugs, organ failure, electrolyte disturbance, infection), and bowel obstruction.33–35
Management of nausea and vomiting
Management of nausea and vomiting may require multiple antiemetics, which may need to be given intravenously or subcutaneously.33
The choice of drugs depends on the cause of the nausea
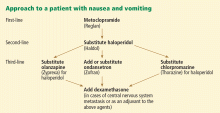
The evidence-based choice of drugs for nausea depends on the cause33–35:
- Nausea due to chemical or metabolic factors: haloperidol (Haldol), levomepromazine (another antipsychotic drug, not available in the United States), cyclizine (Marezine)
- Nausea due to gastric stasis, outlet obstruction: metoclopramide, domperidone (a similar drug, not available in the United States), levomepromazine
- Nausea due to regurgitation: metoclopramide, cyclizine, haloperidol, levomepromazine
- Nausea due to bowel obstruction: metoclopramide (if obstruction is not complete), domperidone, cyclizine, levomepromazine, octreotide (Sandostatin), hyoscyamine (Levsin)
- Nausea due to cranial disease: cyclizine, levomepromazine
- Movement-related nausea: cyclizine, levomepromazine, hyoscyamine
- Cause unclear or multiple causes: cyclizine, haloperidol, levomepromazine
- Cortical nausea: lorazepam (Ativan).
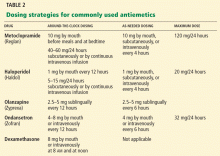
Metoclopramide. If complete bowel obstruction is not suspected, oral metoclopramide, a dopamine antagonist, is our choice for first-line drug therapy.32 Adverse effects include abdominal pain, diarrhea, and sedation.
Haloperidol, another dopamine antagonist, can also be used.32 Haloperidol may cause sedation and is associated with a prolonged QTc interval. Care should be taken in those at risk for dysrhythmia or arrhythmia.
Olanzapine (Zyprexa) is an alternative antipsychotic for patients who cannot tolerate or do not respond to metoclopramide and haloperidol.
Ondansetron (Zofran), a serotonin 5-HT3 receptor antagonist, is usually reserved for nausea and vomiting associated with chemotherapy or radiation, but it can be used in advanced cancer if the above agents fail.37
DYSPNEA IS COMMON, EVEN WITHOUT LUNG DISEASE
Dyspnea is the subjective perception of impaired breathing, which may include the sensation of breathlessness, chest tightness, air hunger, suffocation, or increased work of breathing.
At least half of patients with advanced cancer complain of dyspnea.1 Most have primary pulmonary malignancies or metastatic lung disease, but almost 25% have no documented lung involvement or underlying cardiopulmonary diagnosis to which to attribute it.38
Dyspnea is often very distressing. Palliative sedation is used more frequently for the relief of intractable dyspnea than for pain.39
Opioids are effective but underutilized for dyspnea
Although opioids are effective in both oral and parenteral formulations for the symptomatic management of dyspnea,40 the exact mechanism by which they improve dyspnea is unknown. Central control of respiration occurs in the medulla, and perception of dyspnea is mediated by the sensory cortex.
Opioids are underutilized by physicians other than palliative medicine specialists because of concern about respiratory depression. Appropriately titrated, opioids are safe and do not cause clinically significant respiratory depression.41
Allen et al42 showed that an opioid in low doses (diamorphine 2.5 mg subcutaneously) was effective and well tolerated in elderly patients with advanced pulmonary fibrosis who had not received opioids before.
Start low and go slow. An appropriate starting dose for a patient who has not been on opioids before may be morphine sulfate 2 mg intravenously (or a 5-mg immediate-release tablet by mouth) every 2 hours as needed for dyspnea. After 24 to 48 hours of an as-needed regimen, one can evaluate the patient’s response, tolerance, and dose requirement. If needed, parenteral infusion or a long-acting opioid preparation can be started with continued as-needed bolus dosing for breakthrough dyspnea.
We do not recommend writing opioid infusion orders with a “titrate to comfort” clause in the terminally ill. Increasing the rate of a continuous infusion does not provide the prompt symptomatic relief a bolus dose delivers. Dose accumulation and adverse effects are more likely when opioids are titrated in this fashion.
A Cochrane review showed that nebulized opioids are ineffective for dyspnea.43
Oxygen paradoxically does not improve dyspnea
Oxygen is commonly prescribed, although the literature does not indicate that it improves the sensation of breathlessness.44
A study by Clemens et al45 showed no correlation between dyspnea and oxygen saturation. It also found morphine to be superior to oxygen in subjective dyspnea, even in hypoxia.
A double-blind crossover study showed that ambient air delivered via nasal cannula was as effective as oxygen for dyspnea.46 The inexpensive and simple practice of a fan to blow ambient air on the patient’s face may help relieve dyspnea.
- Donnelly S, Walsh D. The symptoms of advanced cancer. Semin Oncol 1995; 22(2 suppl 3):67–72.
- Walsh D, Rybicki L, Nelson KA, Donnelly S. Symptoms and prognosis in advanced cancer. Support Care Cancer 2002; 10:385–388.
- Komurcu S, Nelson KA, Walsh D, Donnelly SM, Homsi J, Abdullah O. Common symptoms in advanced cancer. Semin Oncol 2000; 27:24–33.
- Homsi J, Walsh D, Rivera N, et al. Symptom evaluation in palliative medicine: patient report vs systematic assessment. Support Care Cancer 2006; 14:444–453.
- Donnelly S. Quality-of-life assessment in advanced cancer. Curr Oncol Rep 2000; 2:338–342.
- Stone P, Richardson A, Ream E, Smith AG, Kerr DJ, Kearney N. Cancer-related fatigue: inevitable, unimportant and untreatable? Results of a multi-centre patient survey. Cancer Fatigue Forum. Ann Oncol 2000; 11:971–975.
- Portenoy RK, Itri LM. Cancer-related fatigue: guidelines for evaluation and management. Oncologist 1999; 4:1–10.
- Berger AM, Abernethy AP, Atkinson A, et al. NCCN Clinical Practice Guidelines in Oncology Cancer-related fatigue—v.1.2010. www.nccn.org/professionals/physician_gls/PDF/fatigue.pdf. Accessed November 15, 2010.
- Schmitz KH, Holtzman J, Courneya KS, Mâsse LC, Duval S, Kane R. Controlled physical activity trials in cancer survivors: a systematic review and meta-analysis. Cancer Epidemiol Biomarkers Prev 2005; 14:1588–1595.
- Porock D, Kristjanson LJ, Tinnelly K, Duke T, Blight J. An exercise intervention for advanced cancer patients experiencing fatigue: a pilot study. J Palliat Care 2000 Autumn; 16:30–36.
- Breitbart W, Rosenfeld B, Kaim M, Funesti-Esch J. A randomized, double-blind, placebo-controlled trial of psychostimulants for the treatment of fatigue in ambulatory patients with human immunodeficiency virus disease. Arch Intern Med 2001; 161:411–420.
- Sarhill N, Walsh D, Nelson KA, Homsi J, LeGrand S, Davis MP. Methylphenidate for fatigue in advanced cancer: a prospective open-label pilot study. Am J Hosp Palliat Care 2001; 18:187–192.
- Minton O, Richardson A, Sharpe M, Hotopf M, Stone P. A systematic review and meta-analysis of the pharmacological treatment of cancer-related fatigue. J Natl Cancer Inst 2008; 100:1155–1166.
- Kaleita TA, Wellisch DK, Graham CA, et al. Pilot study of modafinil for treatment of neurobehavioral dysfunction and fatigue in adult patients with brain tumors (abstract). J Clin Oncol 2006; 24(suppl):58s.
- Morrow GR, Jean-Pierre P, Roscoe JA, et al. A phase III randomized, placebo-controlled, double-blind trial of a eugeroic agent in 642 cancer patients reporting fatigue during chemotherapy: a URCC CCOP study (abstract). J Clin Oncol 2008; 26(suppl):504s.
- Tannock I, Gospodarowicz M, Meakin W, Panzarella T, Stewart L, Rider W. Treatment of metastatic prostatic cancer with low-dose prednisone: evaluation of pain and quality of life as pragmatic indices of response. J Clin Oncol 1989; 7:590–597.
- Morley JE, Thomas DR, Wilson MM. Cachexia: pathophysiology and clinical relevance. Am J Clin Nutr 2006; 83:735–743.
- Poole K, Froggatt K. Loss of weight and loss of appetite in advanced cancer: a problem for the patient, the carer, or the health professional? Palliat Med 2002; 16:499–506.
- Von Roenn JH. Randomized trials of megestrol acetate for AIDS-associated anorexia and cachexia. Oncology 1994; 51(suppl 1):19–24.
- Donnelly S, Walsh TD. Low-dose megestrol acetate for appetite stimulation in advanced cancer. J Pain Symptom Manage 1995; 10:182–183.
- Mulligan K, Zackin R, Von Roenn JH, et al; ACTG 313 Study Team. Testosterone supplementation of megestrol therapy does not enhance lean tissue accrual in men with human immunodeficiency virus-associated weight loss: a randomized, double-blind, placebo-controlled, multicenter trial. J Clin Endocrinol Metab 2007; 92:563–570.
- Navari RM, Brenner MC. Treatment of cancer-related anorexia with olanzapine and megestrol acetate: a randomized trial. Support Care Cancer 2010; 18:951–956.
- Mercadante S, Fulfaro F, Casuccio A. The use of corticosteroids in home palliative care. Support Care Cancer 2001; 9:386–389.
- Walsh D, Kirkova J, Davis MP. The efficacy and tolerability of long-term use of dronabinol in cancer-related anorexia: a case series. J Pain Symptom Manage 2005; 30:493–495.
- Cannabis-In-Cachexia-Study-Group; Strasser F, Luftner D, Possinger K, et al. Comparison of orally administered cannabis extract and delta-9-tetrahydrocannabinol in treating patients with cancer-related anorexia-cachexia syndrome: a multicenter, phase III, randomized, double-blind, placebo-controlled clinical trial from the Cannabis-In-Cachexia-Study-Group. J Clin Oncol 2006; 24:3394–3400.
- Jatoi A, Windschitl HE, Loprinzi CL, et al. Dronabinol versus megestrol acetate versus combination therapy for cancer-associated anorexia: a North Central Cancer Treatment Group study. J Clin Oncol 2002; 20:567–573.
- Winter SM. Terminal nutrition: framing the debate for the withdrawal of nutritional support in terminally ill patients. Am J Med 2000; 109:723–726.
- Drossman DA, Sandler RS, McKee DC, Lovitz AJ. Bowel patterns among subjects not seeking health care. Use of a questionnaire to identify a population with bowel dysfunction. Gastroenterology 1982; 83:529–534.
- McMillan SC. Assessing and managing opiate-induced constipation in adults with cancer. Cancer Control 2004; 11(suppl 3):3–9.
- Robinson CB, Fritch M, Hullett L, et al. Development of a protocol to prevent opioid-induced constipation in patients with cancer: a research utilization project. Clin J Oncol Nurs 2000; 4:79–84.
- Thomas J, Karver S, Cooney GA, et al. Methylnaltrexone for opioid-induced constipation in advanced illness. N Engl J Med 2008; 358:2332–2343.
- Davis MP, Walsh D. Treatment of nausea and vomiting in advanced cancer. Support Care Cancer 2000; 8:444–452.
- Stephenson J, Davies A. An assessment of aetiology-based guidelines for the management of nausea and vomiting in patients with advanced cancer. Support Care Cancer 2006; 14:348–353.
- Lichter I. Results of antiemetic management in terminal illness. J Palliat Care 1993; 9:19–21.
- Bentley A, Boyd K. Use of clinical pictures in the management of nausea and vomiting: a prospective audit. Palliat Med 2001; 15:247–253.
- Glare P, Pereira G, Kristjanson LJ, Stockler M, Tattersall M. Systematic review of the efficacy of antiemetics in the treatment of nausea in patients with far-advanced cancer. Support Care Cancer 2004; 12:432–440.
- Currow DC, Coughlan M, Fardell B, Cooney NJ. Use of ondansetron in palliative medicine. J Pain Symptom Manage 1997; 13:302–307.
- Reuben DB, Mor V. Dyspnea in terminally ill cancer patients. Chest 1986; 89:234–236.
- Fainsinger RL, Waller A, Bercovici M, et al. A multicentre international study of sedation for uncontrolled symptoms in terminally ill patients. Palliat Med 2000; 14:257–265.
- Jennings AL, Davies AN, Higgins JP, Broadley K. Opioids for the palliation of breathlessness in terminal illness. Cochrane Database Syst Rev 2001;CD002066.
- Estfan B, Mahmoud F, Shaheen P, et al. Respiratory function during parenteral opioid titration for cancer pain. Palliat Med 2007; 21:81–86.
- Allen S, Raut S, Woollard J, Vassallo M. Low dose diamorphine reduces breathlessness without causing a fall in oxygen saturation in elderly patients with end-stage idiopathic pulmonary fibrosis. Palliat Med 2005; 19:128–130.
- Polosa R, Simidchiev A, Walters EH. Nebulised morphine for severe interstitial lung disease. Cochrane Database Syst Rev 2002;CD002872.
- Currow DC, Agar M, Smith J, Abernethy AP. Does palliative home oxygen improve dyspnoea? A consecutive cohort study. Palliat Med 2009; 23:309–316.
- Clemens KE, Quednau I, Klaschik E. Use of oxygen and opioids in the palliation of dyspnoea in hypoxic and non-hypoxic palliative care patients: a prospective study. Support Care Cancer 2009; 17:367–377.
- Philip J, Gold M, Milner A, Di Iulio J, Miller B, Spruyt O. A randomized, double-blind, crossover trial of the effect of oxygen on dyspnea in patients with advanced cancer. J Pain Symptom Manage 2006; 32:541–550.
Cancer patients experience many distressing symptoms during the course of their illness. In addition to pain, they commonly suffer from fatigue, anorexia, constipation, dyspnea, nausea, and vomiting.1
Although it is important to diagnose and manage the cancer itself, it is also the physician’s duty to recognize and effectively treat associated symptoms, regardless of the outcome of the underlying disease.
Some of the symptoms are due to the underlying disease, but some are iatrogenic, as many medical interventions have predictable adverse effects, such as nausea and vomiting with chemotherapy or constipation with opioids.
Symptoms of advanced cancer become chronic, and patients usually rate them as moderate or severe.1 Unrelieved suffering causes demoralization and may quickly impair quality of life.2
Understanding the principles of symptom management may help optimize palliation and improve quality of life. In this paper, we outline an approach to the management of cancer-related symptoms.
A HEAVY BURDEN OF SYMPTOMS
In patients with advanced cancer, the prevalence rates of various symptoms are approximately as follows1,3:
- Pain 89%
- Fatigue 69%
- Weakness 66%
- Anorexia 66%
- Lack of energy 61%
- Nausea 60%
- Dry mouth 57%
- Constipation 52%
- Early satiety 51%
- Dyspnea 50%
- Vomiting 30%.
Furthermore, patients with advanced cancer typically have multiple concurrent symptoms. In a survey of patients in a palliative medicine service at our hospital,4 we found that the median number of symptoms per patient was 10 (range 0–25) (Figure 1).
PRINCIPLES OF SYMPTOM MANAGEMENT
Show an interest in the patient’s symptoms. Many patients with advanced cancer believe that suffering is an inevitable part of the disease or of its treatment.
Ask patients about their symptoms in a positive and detailed fashion, starting with open-ended questions and following up with specific questions. Patients may underreport their symptoms or may not mention them if not asked directly. In the survey of palliative care patients at our hospital mentioned above,4 the median number of volunteered symptoms was only 1 (range 0–6), whereas a median of 10 were found by systematic assessment.
The examiner should clarify when necessary and recognize that a layperson’s language may not directly translate to medical language. For example, a patient may not understand the term “anorexia.” Furthermore, “loss of appetite” may mean nausea, vomiting, constipation, or early satiety. “Numbness” may mean a loss of sensation or a pins-and-needles sensation. Symptoms should also be quantified using a consistent measure (ie, numerical or categorical) to facilitate monitoring.
Prioritize the symptoms. Advanced cancer is accompanied by multiple symptoms. Assess which ones are most bothersome, and where therapy should be directed first.
Try to understand the pathophysiology behind the symptom. When possible, choose a drug treatment that targets the likely underlying cause. Nausea and vomiting, for example, can be secondary to gastric outlet obstruction, hypercalcemia, increased intracranial pressure, esophagitis, opioid use, or constipation.
Be specific about the drug, dosing, timing, and route, and keep it simple. If a regimen is cumbersome, compliance suffers. It is better to start one medication for the most bothersome symptom or symptoms and make some progress than it is to overwhelm the patient with a complex list of drugs. Sustained-release formulations are often useful. It is unrealistic to expect most patients to take a medication every 4 hours around the clock. Try the most cost-effective remedies first, and attempt to use one drug that may address multiple symptoms. For example, dexamethasone may have positive effects on energy, pain, and appetite.
Use ‘rescue dosing.’ Rescue drugs are important for expected symptom exacerbations in those on sustained medication. This approach increases efficacy and minimizes adverse effects. In most cases, the rescue medication should be the same as the regularly scheduled one. For example, a prescription to treat nausea may read “metoclopramide (Reglan) 10 mg by mouth before meals and at bedtime and every 4 hours as needed to treat nausea or vomiting.”
Consider the patient’s age and fragility, the cost of the drug, and anticipated adverse effects. Oral or transdermal preparations are preferable to parenteral ones with regard to convenience and compliance, although many transdermal preparations are costly. If parenteral dosing is necessary, the subcutaneous route is an alternative to the intravenous route.
Discontinue drugs that are ineffective or unnecessary. This may help compliance and diminish adverse effects.
Make one change at a time so the response to that change is clear. Titrate one drug to its effective dose, to its maximum dose, or to a level of intolerability before considering another. If one drug of a class is ineffective, another drug in the same class may work.
Reassess often. A follow-up phone call or office visit in 1 to 2 weeks is appropriate. The symptoms of advanced cancer are often progressive, so regular evaluation is important, even if symptoms are controlled on stable drug regimens. Instructions should be both verbal and written and should be communicated to patients and any involved caregiver to ensure compliance. Have a “plan B” if the first plan is ineffective.
A challenging and important part of symptom management is to assess the goals of care. Every intervention is not appropriate for every patient. Which therapies are used depends on the stage of the disease, the available disease-modifying treatments, and the patient’s condition and preferences. Patients and their loved ones should be engaged in discussions about goals of care early in the disease and should be included in medical decision-making. Both curative treatment and palliative treatment are important, but palliation plays a bigger role towards the later stages of advanced cancer (Figure 2).
CANCER-RELATED FATIGUE: COMMON BUT NOT INEVITABLE
Most cancer patients report fatigue. Although it is one of the most common symptoms in advanced cancer,5 it is not necessarily inevitable or untreatable.6
Cancer-related fatigue is multidimensional and develops over time, diminishing energy, mental capacity, and psychological condition.7 Patients may report feeling tired or being unable to complete their activities of daily living. People who were previously very active may be frustrated by their inability to participate in favorite leisure activities, which has a big impact on quality of life. Fatigue can be physical, emotional, or mental. It is important to distinguish physical weakness from dyspnea on exertion, which is commonly reported as fatigue. Depression may also cause or exacerbate fatigue.
Unlike fatigue in the general population, cancer-related fatigue does not improve with rest, and patients often report large amounts of unrestorative sleep.
Look for reversible causes of fatigue
First, conduct a thorough assessment to identify any reversible causes, such as:
- Anemia
- Insomnia, sleep disturbance
- Malnutrition
- Pain
- Depression
- Medical comorbidities: renal, cardiac, or pulmonary disease
- Hypothyroidism
- Hypogonadism.
In many cases, however, a reversible cause cannot be found.
Treating cancer-related fatigue
Nonpharmacologic interventions have been evaluated for this application, but evidence of efficacy is limited and mixed. The National Cancer Comprehensive Network guidelines8 suggest that energy conservation and education about cancer-related fatigue are central to management. Patients should be advised that fatigue has a fluctuating course and that they have a limited pool of energy, which they should conserve and use judiciously.
In a meta-analysis by Schmitz et al,9 physical activity interventions were found to be beneficial. Sixty-three percent of those studied were undergoing active treatment, so whether this population reflects advanced cancer is unclear. A small pilot study in advanced cancer found a trend toward benefit with exercise.10
Comment. The strategies of rest and exercise are complementary. The key point is to plan them per personal preference.
Psychostimulants include methylphenidate (Ritalin). A randomized placebo-controlled trial in patients with acquired immunodeficiency syndrome (AIDS) found methylphenidate 15 to 60 mg/day to have a positive effect.11 Prospective studies have shown similar results in cancer patients,12 and a Cochrane review in 2008 showed a small but significant benefit in cancer-related fatigue.13
Methylphenidate is usually started at a dose of 5 mg given at 8:00 am and at noon, and then titrated. Benefit, when experienced, is typically noted within 24 to 48 hours. Possible adverse effects include anorexia, insomnia, anxiety, confusion, tremor, and tachycardia.
Stimulants should be used with caution in patients with cardiac disease or delirium.
Modafinil (Provigil), a nonstimulant agent, has been less studied, but it may also help.14,15 The usual dosage is 50 to 200 mg daily.
Corticosteroids may have a role in advanced cancer, as suggested by anecdotal reports.16 They should be used judiciously, as their adverse effects (insomnia, muscle wasting, edema) are themselves burdensome and may outweigh their benefits.
ANOREXIA CAN BE DISTRESSING TO THE FAMILY AND THE PATIENT
Most patients with advanced cancer experience anorexia, which is a marker of poor prognosis.1
Appetite loss may occur in isolation or as a part of the anorexia-cachexia syndrome. This syndrome is a wasting state seen in chronic, advanced diseases including cancer, AIDS, chronic obstructive pulmonary disease, chronic renal insufficiency, and congestive heart failure.17 The associated weight loss is involuntary and includes both muscle and fat.
Appetite loss alone is usually not bothersome. In fact, anorexia frequently causes more distress to the family than to the patient.18 The ramifications of decreased appetite, on the other hand, can be devastating. Decreased caloric intake coupled with the hypermetabolic state of malignancy leads to rapid, dramatic changes in body habitus. This outward sign of the ravages of cancer can be psychologically damaging to patients and their loved ones as they contemplate advanced disease and limited life expectancy. They may be concerned about starvation, in which case education about and attempts to normalize the anorexia-cachexia syndrome are essential.
Look for reversible causes of anorexia
The first step in the management of anorexia is to identify any reversible causes, such as:
- Stomatitis
- Constipation
- Uncontrolled severe symptoms such as pain or dyspnea
- Delirium
- Nausea, vomiting
- Depression
- Gastroparesis.
Managing cancer-related anorexia
Nonpharmacologic measures include nutritional counseling and increased physical activity. Patients may be counseled to eat calorie-dense foods and supplemental high-calorie, high-protein, high-fat drinks. Some may be able to take advantage of a diurnal variation in appetite, usually an increased appetite in the morning.
Megestrol acetate (Megace) improved appetite and induced weight gain when used in a dosage of 800 mg daily in a randomized controlled trial in AIDS patients.19 Case studies have shown doses as low as 80 to 160 mg daily to be beneficial.20 Most of the added weight is fat, not lean muscle mass. Unfortunately, the addition of testosterone to megestrol did not increase the accumulation of lean muscle mass in another randomized trial.21 But the addition of olanzapine (Zyprexa) to megestrol was associated with improved appetite and weight gain in a significant percentage of advanced cancer patients.22 Rates of adverse effects with megestrol are low; the most significant adverse effect is thromboembolism.
Corticosteroids. While much of the support for corticosteroids is anecdotal, a prospective study of dexamethasone 4 to 16 mg daily showed improvement in several symptoms, including appetite.23 Because of the multiple adverse effects of corticosteroids, careful attention to dose, duration, and tolerability is essential. Corticosteroids should be discontinued if the desired positive effects are not observed within 3 to 5 days. If prolonged survival is expected, wean to the lowest effective dose.
Cannabinoids. Dronabinol (Marinol), a synthetic formulation of delta-9-tetrahydrocannabinol (THC), the active agent of marijuana, has been beneficial in AIDS anorexia. Fewer studies have been done in advanced cancer.
In a small, open-label case series, doses of 7.5 to 15 mg of dronabinol daily improved appetite and were well tolerated.24 On the other hand, in a multicenter, randomized, double-blind, placebo-controlled trial, neither cannabis extract nor THC (5 mg daily) significantly improved appetite over a 6-week period.25
A large randomized study found megestrol acetate 800 mg to be superior to dronabinol 5 mg daily for treating anorexia.26
Neurotoxicity, anxiety, nervousness, dizziness, euphoria, and somnolence from dronabinol can be severe and intolerable for some.
Enteral tube feeding and parenteral nutrition do not improve survival or comfort in terminally ill patients.27 On the contrary, they are associated with complications, including aspiration pneumonia, sepsis, abdominal pain, vomiting, and diarrhea. Nevertheless, in some patients with mechanical impediments to nutrition (eg, esophageal fistula, obstruction, or proximal small bowel obstruction) or in those who are hungry and unable to take food by mouth, tube feeding may be appropriate.
CONSTIPATION SHOULD BE ANTICIPATED, AND PREVENTED IF POSSIBLE
Constipation is variably defined by patients and health care professionals, but it usually includes components of the Rome II criteria, ie, two or more of the following symptoms28:
- Straining at least 25% of the time
- Hard stools at least 25% of the time
- Incomplete evacuation at least 25% of the time
- Two or fewer bowel movements per week.
These criteria were intended to describe functional constipation in a healthy population.27
More than 50% of patients with advanced cancer report constipation,1 and in those on opioids, the scope of the problem is larger. In addition to binding central nervous system receptors to mediate pain perception, opioids bind systemic receptors including those in the gut. As a result, opioids interfere with smooth muscle tone and contractility, lengthen transit time, promote dry stools, and increase anal sphincter tone.29 A nursing study found that when patients taking opioids were screened for constipation, 95% identified it as the major adverse effect of their pain regimen.30
Multiple causes of constipation
Factors that can cause or contribute to constipation include:
- Dietary factors such as a generally low intake of food, and specifically of fiber
- Inactivity
- Confusion
- Dehydration
- Intestinal obstruction
- Cormorbidities such as diabetes mellitus, hypothyroidism, hypercalcemia
- Uncomfortable toilet arrangements
- Drugs such as opioids (as noted above), anticholinergics, antihypertensives, antacids, diuretics, and iron supplements.
Take a proactive approach to constipation
Constipation is expected in a number of clinical scenarios, such as with the use of opioids or with limited mobility. Patients often attribute constipation to diminished oral intake. But despite low oral intake, regular, smaller-caliber bowel movements are important to ensure that sloughed bowel endothelium and bacteria are eliminated.
Although little evidence supports the use of one standard bowel regimen, prevention is essential. The goal is a soft bowel movement every 1 to 2 days. Constipation prophylaxis should be started at the initiation of any regular opioid regimen. Encouraging physical activity and oral fluid intake and creating a favorable environment for elimination may also help manage constipation.
In our practice, we use a softening agent such as docusate sodium (Colace) 100 mg twice daily, and add a laxative agent such as senna (Senokot) or a magnesium-based osmotic agent as needed. Bulking agents such as over-the-counter fiber supplements should be used with caution in opioid-related constipation. If there has been no bowel movement for 48 hours, a rectal suppository or enema is used. Suppositories or enemas can be scheduled regularly for bedbound patients with chronic constipation.
Methylnaltrexone (Relistor), a mu-opioid antagonist, is a new agent that blocks peripheral opioid receptors in the gut. In a randomized study of 133 patients, methylnaltrexone produced laxation within 4 hours of administration in 48%.31 This methylated, charged compound does not significantly cross the blood-brain barrier and therefore does not interfere with analgesia or cause opioid withdrawal. The dose is 8 mg or 12 mg subcutaneously (based on weight), which can be repeated in 48 hours. If laxation does not occur after one to three doses, other causes of constipation should be explored.
Methylnaltrexone is contraindicated in patients with bowel obstruction, even if the obstruction is thought to be secondary to opioids. Adverse effects include abdominal pain, flatulence, and nausea.
NAUSEA AND VOMITING: NOT ALWAYS DUE TO CHEMOTHERAPY
Nausea (the sensation of the need to vomit) and vomiting (the forceful expulsion of gastric contents) are common symptoms in advanced cancer and are not necessarily related to chemotherapy or radiation therapy. About 60% of cancer patients have nausea, and about 30% vomit.32 Both symptoms are very distressing and diminish quality of life.
Look for potentially reversible causes of nausea and vomiting
Identifying the cause, which is sometimes reversible, may help direct treatment. Potentially reversible causes include:
- Drugs
- Uremia
- Infection
- Anxiety
- Constipation
- Gastric irritation
- Proximal gastrointestinal obstruction.
In a prospective study of 121 patients with advanced cancer, the most common causes of nausea and vomiting were impaired gastric emptying, chemical and metabolic factors (drugs, organ failure, electrolyte disturbance, infection), and bowel obstruction.33–35
Management of nausea and vomiting
Management of nausea and vomiting may require multiple antiemetics, which may need to be given intravenously or subcutaneously.33
The choice of drugs depends on the cause of the nausea
The evidence-based choice of drugs for nausea depends on the cause33–35:
- Nausea due to chemical or metabolic factors: haloperidol (Haldol), levomepromazine (another antipsychotic drug, not available in the United States), cyclizine (Marezine)
- Nausea due to gastric stasis, outlet obstruction: metoclopramide, domperidone (a similar drug, not available in the United States), levomepromazine
- Nausea due to regurgitation: metoclopramide, cyclizine, haloperidol, levomepromazine
- Nausea due to bowel obstruction: metoclopramide (if obstruction is not complete), domperidone, cyclizine, levomepromazine, octreotide (Sandostatin), hyoscyamine (Levsin)
- Nausea due to cranial disease: cyclizine, levomepromazine
- Movement-related nausea: cyclizine, levomepromazine, hyoscyamine
- Cause unclear or multiple causes: cyclizine, haloperidol, levomepromazine
- Cortical nausea: lorazepam (Ativan).
Metoclopramide. If complete bowel obstruction is not suspected, oral metoclopramide, a dopamine antagonist, is our choice for first-line drug therapy.32 Adverse effects include abdominal pain, diarrhea, and sedation.
Haloperidol, another dopamine antagonist, can also be used.32 Haloperidol may cause sedation and is associated with a prolonged QTc interval. Care should be taken in those at risk for dysrhythmia or arrhythmia.
Olanzapine (Zyprexa) is an alternative antipsychotic for patients who cannot tolerate or do not respond to metoclopramide and haloperidol.
Ondansetron (Zofran), a serotonin 5-HT3 receptor antagonist, is usually reserved for nausea and vomiting associated with chemotherapy or radiation, but it can be used in advanced cancer if the above agents fail.37
DYSPNEA IS COMMON, EVEN WITHOUT LUNG DISEASE
Dyspnea is the subjective perception of impaired breathing, which may include the sensation of breathlessness, chest tightness, air hunger, suffocation, or increased work of breathing.
At least half of patients with advanced cancer complain of dyspnea.1 Most have primary pulmonary malignancies or metastatic lung disease, but almost 25% have no documented lung involvement or underlying cardiopulmonary diagnosis to which to attribute it.38
Dyspnea is often very distressing. Palliative sedation is used more frequently for the relief of intractable dyspnea than for pain.39
Opioids are effective but underutilized for dyspnea
Although opioids are effective in both oral and parenteral formulations for the symptomatic management of dyspnea,40 the exact mechanism by which they improve dyspnea is unknown. Central control of respiration occurs in the medulla, and perception of dyspnea is mediated by the sensory cortex.
Opioids are underutilized by physicians other than palliative medicine specialists because of concern about respiratory depression. Appropriately titrated, opioids are safe and do not cause clinically significant respiratory depression.41
Allen et al42 showed that an opioid in low doses (diamorphine 2.5 mg subcutaneously) was effective and well tolerated in elderly patients with advanced pulmonary fibrosis who had not received opioids before.
Start low and go slow. An appropriate starting dose for a patient who has not been on opioids before may be morphine sulfate 2 mg intravenously (or a 5-mg immediate-release tablet by mouth) every 2 hours as needed for dyspnea. After 24 to 48 hours of an as-needed regimen, one can evaluate the patient’s response, tolerance, and dose requirement. If needed, parenteral infusion or a long-acting opioid preparation can be started with continued as-needed bolus dosing for breakthrough dyspnea.
We do not recommend writing opioid infusion orders with a “titrate to comfort” clause in the terminally ill. Increasing the rate of a continuous infusion does not provide the prompt symptomatic relief a bolus dose delivers. Dose accumulation and adverse effects are more likely when opioids are titrated in this fashion.
A Cochrane review showed that nebulized opioids are ineffective for dyspnea.43
Oxygen paradoxically does not improve dyspnea
Oxygen is commonly prescribed, although the literature does not indicate that it improves the sensation of breathlessness.44
A study by Clemens et al45 showed no correlation between dyspnea and oxygen saturation. It also found morphine to be superior to oxygen in subjective dyspnea, even in hypoxia.
A double-blind crossover study showed that ambient air delivered via nasal cannula was as effective as oxygen for dyspnea.46 The inexpensive and simple practice of a fan to blow ambient air on the patient’s face may help relieve dyspnea.
Cancer patients experience many distressing symptoms during the course of their illness. In addition to pain, they commonly suffer from fatigue, anorexia, constipation, dyspnea, nausea, and vomiting.1
Although it is important to diagnose and manage the cancer itself, it is also the physician’s duty to recognize and effectively treat associated symptoms, regardless of the outcome of the underlying disease.
Some of the symptoms are due to the underlying disease, but some are iatrogenic, as many medical interventions have predictable adverse effects, such as nausea and vomiting with chemotherapy or constipation with opioids.
Symptoms of advanced cancer become chronic, and patients usually rate them as moderate or severe.1 Unrelieved suffering causes demoralization and may quickly impair quality of life.2
Understanding the principles of symptom management may help optimize palliation and improve quality of life. In this paper, we outline an approach to the management of cancer-related symptoms.
A HEAVY BURDEN OF SYMPTOMS
In patients with advanced cancer, the prevalence rates of various symptoms are approximately as follows1,3:
- Pain 89%
- Fatigue 69%
- Weakness 66%
- Anorexia 66%
- Lack of energy 61%
- Nausea 60%
- Dry mouth 57%
- Constipation 52%
- Early satiety 51%
- Dyspnea 50%
- Vomiting 30%.
Furthermore, patients with advanced cancer typically have multiple concurrent symptoms. In a survey of patients in a palliative medicine service at our hospital,4 we found that the median number of symptoms per patient was 10 (range 0–25) (Figure 1).
PRINCIPLES OF SYMPTOM MANAGEMENT
Show an interest in the patient’s symptoms. Many patients with advanced cancer believe that suffering is an inevitable part of the disease or of its treatment.
Ask patients about their symptoms in a positive and detailed fashion, starting with open-ended questions and following up with specific questions. Patients may underreport their symptoms or may not mention them if not asked directly. In the survey of palliative care patients at our hospital mentioned above,4 the median number of volunteered symptoms was only 1 (range 0–6), whereas a median of 10 were found by systematic assessment.
The examiner should clarify when necessary and recognize that a layperson’s language may not directly translate to medical language. For example, a patient may not understand the term “anorexia.” Furthermore, “loss of appetite” may mean nausea, vomiting, constipation, or early satiety. “Numbness” may mean a loss of sensation or a pins-and-needles sensation. Symptoms should also be quantified using a consistent measure (ie, numerical or categorical) to facilitate monitoring.
Prioritize the symptoms. Advanced cancer is accompanied by multiple symptoms. Assess which ones are most bothersome, and where therapy should be directed first.
Try to understand the pathophysiology behind the symptom. When possible, choose a drug treatment that targets the likely underlying cause. Nausea and vomiting, for example, can be secondary to gastric outlet obstruction, hypercalcemia, increased intracranial pressure, esophagitis, opioid use, or constipation.
Be specific about the drug, dosing, timing, and route, and keep it simple. If a regimen is cumbersome, compliance suffers. It is better to start one medication for the most bothersome symptom or symptoms and make some progress than it is to overwhelm the patient with a complex list of drugs. Sustained-release formulations are often useful. It is unrealistic to expect most patients to take a medication every 4 hours around the clock. Try the most cost-effective remedies first, and attempt to use one drug that may address multiple symptoms. For example, dexamethasone may have positive effects on energy, pain, and appetite.
Use ‘rescue dosing.’ Rescue drugs are important for expected symptom exacerbations in those on sustained medication. This approach increases efficacy and minimizes adverse effects. In most cases, the rescue medication should be the same as the regularly scheduled one. For example, a prescription to treat nausea may read “metoclopramide (Reglan) 10 mg by mouth before meals and at bedtime and every 4 hours as needed to treat nausea or vomiting.”
Consider the patient’s age and fragility, the cost of the drug, and anticipated adverse effects. Oral or transdermal preparations are preferable to parenteral ones with regard to convenience and compliance, although many transdermal preparations are costly. If parenteral dosing is necessary, the subcutaneous route is an alternative to the intravenous route.
Discontinue drugs that are ineffective or unnecessary. This may help compliance and diminish adverse effects.
Make one change at a time so the response to that change is clear. Titrate one drug to its effective dose, to its maximum dose, or to a level of intolerability before considering another. If one drug of a class is ineffective, another drug in the same class may work.
Reassess often. A follow-up phone call or office visit in 1 to 2 weeks is appropriate. The symptoms of advanced cancer are often progressive, so regular evaluation is important, even if symptoms are controlled on stable drug regimens. Instructions should be both verbal and written and should be communicated to patients and any involved caregiver to ensure compliance. Have a “plan B” if the first plan is ineffective.
A challenging and important part of symptom management is to assess the goals of care. Every intervention is not appropriate for every patient. Which therapies are used depends on the stage of the disease, the available disease-modifying treatments, and the patient’s condition and preferences. Patients and their loved ones should be engaged in discussions about goals of care early in the disease and should be included in medical decision-making. Both curative treatment and palliative treatment are important, but palliation plays a bigger role towards the later stages of advanced cancer (Figure 2).
CANCER-RELATED FATIGUE: COMMON BUT NOT INEVITABLE
Most cancer patients report fatigue. Although it is one of the most common symptoms in advanced cancer,5 it is not necessarily inevitable or untreatable.6
Cancer-related fatigue is multidimensional and develops over time, diminishing energy, mental capacity, and psychological condition.7 Patients may report feeling tired or being unable to complete their activities of daily living. People who were previously very active may be frustrated by their inability to participate in favorite leisure activities, which has a big impact on quality of life. Fatigue can be physical, emotional, or mental. It is important to distinguish physical weakness from dyspnea on exertion, which is commonly reported as fatigue. Depression may also cause or exacerbate fatigue.
Unlike fatigue in the general population, cancer-related fatigue does not improve with rest, and patients often report large amounts of unrestorative sleep.
Look for reversible causes of fatigue
First, conduct a thorough assessment to identify any reversible causes, such as:
- Anemia
- Insomnia, sleep disturbance
- Malnutrition
- Pain
- Depression
- Medical comorbidities: renal, cardiac, or pulmonary disease
- Hypothyroidism
- Hypogonadism.
In many cases, however, a reversible cause cannot be found.
Treating cancer-related fatigue
Nonpharmacologic interventions have been evaluated for this application, but evidence of efficacy is limited and mixed. The National Cancer Comprehensive Network guidelines8 suggest that energy conservation and education about cancer-related fatigue are central to management. Patients should be advised that fatigue has a fluctuating course and that they have a limited pool of energy, which they should conserve and use judiciously.
In a meta-analysis by Schmitz et al,9 physical activity interventions were found to be beneficial. Sixty-three percent of those studied were undergoing active treatment, so whether this population reflects advanced cancer is unclear. A small pilot study in advanced cancer found a trend toward benefit with exercise.10
Comment. The strategies of rest and exercise are complementary. The key point is to plan them per personal preference.
Psychostimulants include methylphenidate (Ritalin). A randomized placebo-controlled trial in patients with acquired immunodeficiency syndrome (AIDS) found methylphenidate 15 to 60 mg/day to have a positive effect.11 Prospective studies have shown similar results in cancer patients,12 and a Cochrane review in 2008 showed a small but significant benefit in cancer-related fatigue.13
Methylphenidate is usually started at a dose of 5 mg given at 8:00 am and at noon, and then titrated. Benefit, when experienced, is typically noted within 24 to 48 hours. Possible adverse effects include anorexia, insomnia, anxiety, confusion, tremor, and tachycardia.
Stimulants should be used with caution in patients with cardiac disease or delirium.
Modafinil (Provigil), a nonstimulant agent, has been less studied, but it may also help.14,15 The usual dosage is 50 to 200 mg daily.
Corticosteroids may have a role in advanced cancer, as suggested by anecdotal reports.16 They should be used judiciously, as their adverse effects (insomnia, muscle wasting, edema) are themselves burdensome and may outweigh their benefits.
ANOREXIA CAN BE DISTRESSING TO THE FAMILY AND THE PATIENT
Most patients with advanced cancer experience anorexia, which is a marker of poor prognosis.1
Appetite loss may occur in isolation or as a part of the anorexia-cachexia syndrome. This syndrome is a wasting state seen in chronic, advanced diseases including cancer, AIDS, chronic obstructive pulmonary disease, chronic renal insufficiency, and congestive heart failure.17 The associated weight loss is involuntary and includes both muscle and fat.
Appetite loss alone is usually not bothersome. In fact, anorexia frequently causes more distress to the family than to the patient.18 The ramifications of decreased appetite, on the other hand, can be devastating. Decreased caloric intake coupled with the hypermetabolic state of malignancy leads to rapid, dramatic changes in body habitus. This outward sign of the ravages of cancer can be psychologically damaging to patients and their loved ones as they contemplate advanced disease and limited life expectancy. They may be concerned about starvation, in which case education about and attempts to normalize the anorexia-cachexia syndrome are essential.
Look for reversible causes of anorexia
The first step in the management of anorexia is to identify any reversible causes, such as:
- Stomatitis
- Constipation
- Uncontrolled severe symptoms such as pain or dyspnea
- Delirium
- Nausea, vomiting
- Depression
- Gastroparesis.
Managing cancer-related anorexia
Nonpharmacologic measures include nutritional counseling and increased physical activity. Patients may be counseled to eat calorie-dense foods and supplemental high-calorie, high-protein, high-fat drinks. Some may be able to take advantage of a diurnal variation in appetite, usually an increased appetite in the morning.
Megestrol acetate (Megace) improved appetite and induced weight gain when used in a dosage of 800 mg daily in a randomized controlled trial in AIDS patients.19 Case studies have shown doses as low as 80 to 160 mg daily to be beneficial.20 Most of the added weight is fat, not lean muscle mass. Unfortunately, the addition of testosterone to megestrol did not increase the accumulation of lean muscle mass in another randomized trial.21 But the addition of olanzapine (Zyprexa) to megestrol was associated with improved appetite and weight gain in a significant percentage of advanced cancer patients.22 Rates of adverse effects with megestrol are low; the most significant adverse effect is thromboembolism.
Corticosteroids. While much of the support for corticosteroids is anecdotal, a prospective study of dexamethasone 4 to 16 mg daily showed improvement in several symptoms, including appetite.23 Because of the multiple adverse effects of corticosteroids, careful attention to dose, duration, and tolerability is essential. Corticosteroids should be discontinued if the desired positive effects are not observed within 3 to 5 days. If prolonged survival is expected, wean to the lowest effective dose.
Cannabinoids. Dronabinol (Marinol), a synthetic formulation of delta-9-tetrahydrocannabinol (THC), the active agent of marijuana, has been beneficial in AIDS anorexia. Fewer studies have been done in advanced cancer.
In a small, open-label case series, doses of 7.5 to 15 mg of dronabinol daily improved appetite and were well tolerated.24 On the other hand, in a multicenter, randomized, double-blind, placebo-controlled trial, neither cannabis extract nor THC (5 mg daily) significantly improved appetite over a 6-week period.25
A large randomized study found megestrol acetate 800 mg to be superior to dronabinol 5 mg daily for treating anorexia.26
Neurotoxicity, anxiety, nervousness, dizziness, euphoria, and somnolence from dronabinol can be severe and intolerable for some.
Enteral tube feeding and parenteral nutrition do not improve survival or comfort in terminally ill patients.27 On the contrary, they are associated with complications, including aspiration pneumonia, sepsis, abdominal pain, vomiting, and diarrhea. Nevertheless, in some patients with mechanical impediments to nutrition (eg, esophageal fistula, obstruction, or proximal small bowel obstruction) or in those who are hungry and unable to take food by mouth, tube feeding may be appropriate.
CONSTIPATION SHOULD BE ANTICIPATED, AND PREVENTED IF POSSIBLE
Constipation is variably defined by patients and health care professionals, but it usually includes components of the Rome II criteria, ie, two or more of the following symptoms28:
- Straining at least 25% of the time
- Hard stools at least 25% of the time
- Incomplete evacuation at least 25% of the time
- Two or fewer bowel movements per week.
These criteria were intended to describe functional constipation in a healthy population.27
More than 50% of patients with advanced cancer report constipation,1 and in those on opioids, the scope of the problem is larger. In addition to binding central nervous system receptors to mediate pain perception, opioids bind systemic receptors including those in the gut. As a result, opioids interfere with smooth muscle tone and contractility, lengthen transit time, promote dry stools, and increase anal sphincter tone.29 A nursing study found that when patients taking opioids were screened for constipation, 95% identified it as the major adverse effect of their pain regimen.30
Multiple causes of constipation
Factors that can cause or contribute to constipation include:
- Dietary factors such as a generally low intake of food, and specifically of fiber
- Inactivity
- Confusion
- Dehydration
- Intestinal obstruction
- Cormorbidities such as diabetes mellitus, hypothyroidism, hypercalcemia
- Uncomfortable toilet arrangements
- Drugs such as opioids (as noted above), anticholinergics, antihypertensives, antacids, diuretics, and iron supplements.
Take a proactive approach to constipation
Constipation is expected in a number of clinical scenarios, such as with the use of opioids or with limited mobility. Patients often attribute constipation to diminished oral intake. But despite low oral intake, regular, smaller-caliber bowel movements are important to ensure that sloughed bowel endothelium and bacteria are eliminated.
Although little evidence supports the use of one standard bowel regimen, prevention is essential. The goal is a soft bowel movement every 1 to 2 days. Constipation prophylaxis should be started at the initiation of any regular opioid regimen. Encouraging physical activity and oral fluid intake and creating a favorable environment for elimination may also help manage constipation.
In our practice, we use a softening agent such as docusate sodium (Colace) 100 mg twice daily, and add a laxative agent such as senna (Senokot) or a magnesium-based osmotic agent as needed. Bulking agents such as over-the-counter fiber supplements should be used with caution in opioid-related constipation. If there has been no bowel movement for 48 hours, a rectal suppository or enema is used. Suppositories or enemas can be scheduled regularly for bedbound patients with chronic constipation.
Methylnaltrexone (Relistor), a mu-opioid antagonist, is a new agent that blocks peripheral opioid receptors in the gut. In a randomized study of 133 patients, methylnaltrexone produced laxation within 4 hours of administration in 48%.31 This methylated, charged compound does not significantly cross the blood-brain barrier and therefore does not interfere with analgesia or cause opioid withdrawal. The dose is 8 mg or 12 mg subcutaneously (based on weight), which can be repeated in 48 hours. If laxation does not occur after one to three doses, other causes of constipation should be explored.
Methylnaltrexone is contraindicated in patients with bowel obstruction, even if the obstruction is thought to be secondary to opioids. Adverse effects include abdominal pain, flatulence, and nausea.
NAUSEA AND VOMITING: NOT ALWAYS DUE TO CHEMOTHERAPY
Nausea (the sensation of the need to vomit) and vomiting (the forceful expulsion of gastric contents) are common symptoms in advanced cancer and are not necessarily related to chemotherapy or radiation therapy. About 60% of cancer patients have nausea, and about 30% vomit.32 Both symptoms are very distressing and diminish quality of life.
Look for potentially reversible causes of nausea and vomiting
Identifying the cause, which is sometimes reversible, may help direct treatment. Potentially reversible causes include:
- Drugs
- Uremia
- Infection
- Anxiety
- Constipation
- Gastric irritation
- Proximal gastrointestinal obstruction.
In a prospective study of 121 patients with advanced cancer, the most common causes of nausea and vomiting were impaired gastric emptying, chemical and metabolic factors (drugs, organ failure, electrolyte disturbance, infection), and bowel obstruction.33–35
Management of nausea and vomiting
Management of nausea and vomiting may require multiple antiemetics, which may need to be given intravenously or subcutaneously.33
The choice of drugs depends on the cause of the nausea
The evidence-based choice of drugs for nausea depends on the cause33–35:
- Nausea due to chemical or metabolic factors: haloperidol (Haldol), levomepromazine (another antipsychotic drug, not available in the United States), cyclizine (Marezine)
- Nausea due to gastric stasis, outlet obstruction: metoclopramide, domperidone (a similar drug, not available in the United States), levomepromazine
- Nausea due to regurgitation: metoclopramide, cyclizine, haloperidol, levomepromazine
- Nausea due to bowel obstruction: metoclopramide (if obstruction is not complete), domperidone, cyclizine, levomepromazine, octreotide (Sandostatin), hyoscyamine (Levsin)
- Nausea due to cranial disease: cyclizine, levomepromazine
- Movement-related nausea: cyclizine, levomepromazine, hyoscyamine
- Cause unclear or multiple causes: cyclizine, haloperidol, levomepromazine
- Cortical nausea: lorazepam (Ativan).
Metoclopramide. If complete bowel obstruction is not suspected, oral metoclopramide, a dopamine antagonist, is our choice for first-line drug therapy.32 Adverse effects include abdominal pain, diarrhea, and sedation.
Haloperidol, another dopamine antagonist, can also be used.32 Haloperidol may cause sedation and is associated with a prolonged QTc interval. Care should be taken in those at risk for dysrhythmia or arrhythmia.
Olanzapine (Zyprexa) is an alternative antipsychotic for patients who cannot tolerate or do not respond to metoclopramide and haloperidol.
Ondansetron (Zofran), a serotonin 5-HT3 receptor antagonist, is usually reserved for nausea and vomiting associated with chemotherapy or radiation, but it can be used in advanced cancer if the above agents fail.37
DYSPNEA IS COMMON, EVEN WITHOUT LUNG DISEASE
Dyspnea is the subjective perception of impaired breathing, which may include the sensation of breathlessness, chest tightness, air hunger, suffocation, or increased work of breathing.
At least half of patients with advanced cancer complain of dyspnea.1 Most have primary pulmonary malignancies or metastatic lung disease, but almost 25% have no documented lung involvement or underlying cardiopulmonary diagnosis to which to attribute it.38
Dyspnea is often very distressing. Palliative sedation is used more frequently for the relief of intractable dyspnea than for pain.39
Opioids are effective but underutilized for dyspnea
Although opioids are effective in both oral and parenteral formulations for the symptomatic management of dyspnea,40 the exact mechanism by which they improve dyspnea is unknown. Central control of respiration occurs in the medulla, and perception of dyspnea is mediated by the sensory cortex.
Opioids are underutilized by physicians other than palliative medicine specialists because of concern about respiratory depression. Appropriately titrated, opioids are safe and do not cause clinically significant respiratory depression.41
Allen et al42 showed that an opioid in low doses (diamorphine 2.5 mg subcutaneously) was effective and well tolerated in elderly patients with advanced pulmonary fibrosis who had not received opioids before.
Start low and go slow. An appropriate starting dose for a patient who has not been on opioids before may be morphine sulfate 2 mg intravenously (or a 5-mg immediate-release tablet by mouth) every 2 hours as needed for dyspnea. After 24 to 48 hours of an as-needed regimen, one can evaluate the patient’s response, tolerance, and dose requirement. If needed, parenteral infusion or a long-acting opioid preparation can be started with continued as-needed bolus dosing for breakthrough dyspnea.
We do not recommend writing opioid infusion orders with a “titrate to comfort” clause in the terminally ill. Increasing the rate of a continuous infusion does not provide the prompt symptomatic relief a bolus dose delivers. Dose accumulation and adverse effects are more likely when opioids are titrated in this fashion.
A Cochrane review showed that nebulized opioids are ineffective for dyspnea.43
Oxygen paradoxically does not improve dyspnea
Oxygen is commonly prescribed, although the literature does not indicate that it improves the sensation of breathlessness.44
A study by Clemens et al45 showed no correlation between dyspnea and oxygen saturation. It also found morphine to be superior to oxygen in subjective dyspnea, even in hypoxia.
A double-blind crossover study showed that ambient air delivered via nasal cannula was as effective as oxygen for dyspnea.46 The inexpensive and simple practice of a fan to blow ambient air on the patient’s face may help relieve dyspnea.
- Donnelly S, Walsh D. The symptoms of advanced cancer. Semin Oncol 1995; 22(2 suppl 3):67–72.
- Walsh D, Rybicki L, Nelson KA, Donnelly S. Symptoms and prognosis in advanced cancer. Support Care Cancer 2002; 10:385–388.
- Komurcu S, Nelson KA, Walsh D, Donnelly SM, Homsi J, Abdullah O. Common symptoms in advanced cancer. Semin Oncol 2000; 27:24–33.
- Homsi J, Walsh D, Rivera N, et al. Symptom evaluation in palliative medicine: patient report vs systematic assessment. Support Care Cancer 2006; 14:444–453.
- Donnelly S. Quality-of-life assessment in advanced cancer. Curr Oncol Rep 2000; 2:338–342.
- Stone P, Richardson A, Ream E, Smith AG, Kerr DJ, Kearney N. Cancer-related fatigue: inevitable, unimportant and untreatable? Results of a multi-centre patient survey. Cancer Fatigue Forum. Ann Oncol 2000; 11:971–975.
- Portenoy RK, Itri LM. Cancer-related fatigue: guidelines for evaluation and management. Oncologist 1999; 4:1–10.
- Berger AM, Abernethy AP, Atkinson A, et al. NCCN Clinical Practice Guidelines in Oncology Cancer-related fatigue—v.1.2010. www.nccn.org/professionals/physician_gls/PDF/fatigue.pdf. Accessed November 15, 2010.
- Schmitz KH, Holtzman J, Courneya KS, Mâsse LC, Duval S, Kane R. Controlled physical activity trials in cancer survivors: a systematic review and meta-analysis. Cancer Epidemiol Biomarkers Prev 2005; 14:1588–1595.
- Porock D, Kristjanson LJ, Tinnelly K, Duke T, Blight J. An exercise intervention for advanced cancer patients experiencing fatigue: a pilot study. J Palliat Care 2000 Autumn; 16:30–36.
- Breitbart W, Rosenfeld B, Kaim M, Funesti-Esch J. A randomized, double-blind, placebo-controlled trial of psychostimulants for the treatment of fatigue in ambulatory patients with human immunodeficiency virus disease. Arch Intern Med 2001; 161:411–420.
- Sarhill N, Walsh D, Nelson KA, Homsi J, LeGrand S, Davis MP. Methylphenidate for fatigue in advanced cancer: a prospective open-label pilot study. Am J Hosp Palliat Care 2001; 18:187–192.
- Minton O, Richardson A, Sharpe M, Hotopf M, Stone P. A systematic review and meta-analysis of the pharmacological treatment of cancer-related fatigue. J Natl Cancer Inst 2008; 100:1155–1166.
- Kaleita TA, Wellisch DK, Graham CA, et al. Pilot study of modafinil for treatment of neurobehavioral dysfunction and fatigue in adult patients with brain tumors (abstract). J Clin Oncol 2006; 24(suppl):58s.
- Morrow GR, Jean-Pierre P, Roscoe JA, et al. A phase III randomized, placebo-controlled, double-blind trial of a eugeroic agent in 642 cancer patients reporting fatigue during chemotherapy: a URCC CCOP study (abstract). J Clin Oncol 2008; 26(suppl):504s.
- Tannock I, Gospodarowicz M, Meakin W, Panzarella T, Stewart L, Rider W. Treatment of metastatic prostatic cancer with low-dose prednisone: evaluation of pain and quality of life as pragmatic indices of response. J Clin Oncol 1989; 7:590–597.
- Morley JE, Thomas DR, Wilson MM. Cachexia: pathophysiology and clinical relevance. Am J Clin Nutr 2006; 83:735–743.
- Poole K, Froggatt K. Loss of weight and loss of appetite in advanced cancer: a problem for the patient, the carer, or the health professional? Palliat Med 2002; 16:499–506.
- Von Roenn JH. Randomized trials of megestrol acetate for AIDS-associated anorexia and cachexia. Oncology 1994; 51(suppl 1):19–24.
- Donnelly S, Walsh TD. Low-dose megestrol acetate for appetite stimulation in advanced cancer. J Pain Symptom Manage 1995; 10:182–183.
- Mulligan K, Zackin R, Von Roenn JH, et al; ACTG 313 Study Team. Testosterone supplementation of megestrol therapy does not enhance lean tissue accrual in men with human immunodeficiency virus-associated weight loss: a randomized, double-blind, placebo-controlled, multicenter trial. J Clin Endocrinol Metab 2007; 92:563–570.
- Navari RM, Brenner MC. Treatment of cancer-related anorexia with olanzapine and megestrol acetate: a randomized trial. Support Care Cancer 2010; 18:951–956.
- Mercadante S, Fulfaro F, Casuccio A. The use of corticosteroids in home palliative care. Support Care Cancer 2001; 9:386–389.
- Walsh D, Kirkova J, Davis MP. The efficacy and tolerability of long-term use of dronabinol in cancer-related anorexia: a case series. J Pain Symptom Manage 2005; 30:493–495.
- Cannabis-In-Cachexia-Study-Group; Strasser F, Luftner D, Possinger K, et al. Comparison of orally administered cannabis extract and delta-9-tetrahydrocannabinol in treating patients with cancer-related anorexia-cachexia syndrome: a multicenter, phase III, randomized, double-blind, placebo-controlled clinical trial from the Cannabis-In-Cachexia-Study-Group. J Clin Oncol 2006; 24:3394–3400.
- Jatoi A, Windschitl HE, Loprinzi CL, et al. Dronabinol versus megestrol acetate versus combination therapy for cancer-associated anorexia: a North Central Cancer Treatment Group study. J Clin Oncol 2002; 20:567–573.
- Winter SM. Terminal nutrition: framing the debate for the withdrawal of nutritional support in terminally ill patients. Am J Med 2000; 109:723–726.
- Drossman DA, Sandler RS, McKee DC, Lovitz AJ. Bowel patterns among subjects not seeking health care. Use of a questionnaire to identify a population with bowel dysfunction. Gastroenterology 1982; 83:529–534.
- McMillan SC. Assessing and managing opiate-induced constipation in adults with cancer. Cancer Control 2004; 11(suppl 3):3–9.
- Robinson CB, Fritch M, Hullett L, et al. Development of a protocol to prevent opioid-induced constipation in patients with cancer: a research utilization project. Clin J Oncol Nurs 2000; 4:79–84.
- Thomas J, Karver S, Cooney GA, et al. Methylnaltrexone for opioid-induced constipation in advanced illness. N Engl J Med 2008; 358:2332–2343.
- Davis MP, Walsh D. Treatment of nausea and vomiting in advanced cancer. Support Care Cancer 2000; 8:444–452.
- Stephenson J, Davies A. An assessment of aetiology-based guidelines for the management of nausea and vomiting in patients with advanced cancer. Support Care Cancer 2006; 14:348–353.
- Lichter I. Results of antiemetic management in terminal illness. J Palliat Care 1993; 9:19–21.
- Bentley A, Boyd K. Use of clinical pictures in the management of nausea and vomiting: a prospective audit. Palliat Med 2001; 15:247–253.
- Glare P, Pereira G, Kristjanson LJ, Stockler M, Tattersall M. Systematic review of the efficacy of antiemetics in the treatment of nausea in patients with far-advanced cancer. Support Care Cancer 2004; 12:432–440.
- Currow DC, Coughlan M, Fardell B, Cooney NJ. Use of ondansetron in palliative medicine. J Pain Symptom Manage 1997; 13:302–307.
- Reuben DB, Mor V. Dyspnea in terminally ill cancer patients. Chest 1986; 89:234–236.
- Fainsinger RL, Waller A, Bercovici M, et al. A multicentre international study of sedation for uncontrolled symptoms in terminally ill patients. Palliat Med 2000; 14:257–265.
- Jennings AL, Davies AN, Higgins JP, Broadley K. Opioids for the palliation of breathlessness in terminal illness. Cochrane Database Syst Rev 2001;CD002066.
- Estfan B, Mahmoud F, Shaheen P, et al. Respiratory function during parenteral opioid titration for cancer pain. Palliat Med 2007; 21:81–86.
- Allen S, Raut S, Woollard J, Vassallo M. Low dose diamorphine reduces breathlessness without causing a fall in oxygen saturation in elderly patients with end-stage idiopathic pulmonary fibrosis. Palliat Med 2005; 19:128–130.
- Polosa R, Simidchiev A, Walters EH. Nebulised morphine for severe interstitial lung disease. Cochrane Database Syst Rev 2002;CD002872.
- Currow DC, Agar M, Smith J, Abernethy AP. Does palliative home oxygen improve dyspnoea? A consecutive cohort study. Palliat Med 2009; 23:309–316.
- Clemens KE, Quednau I, Klaschik E. Use of oxygen and opioids in the palliation of dyspnoea in hypoxic and non-hypoxic palliative care patients: a prospective study. Support Care Cancer 2009; 17:367–377.
- Philip J, Gold M, Milner A, Di Iulio J, Miller B, Spruyt O. A randomized, double-blind, crossover trial of the effect of oxygen on dyspnea in patients with advanced cancer. J Pain Symptom Manage 2006; 32:541–550.
- Donnelly S, Walsh D. The symptoms of advanced cancer. Semin Oncol 1995; 22(2 suppl 3):67–72.
- Walsh D, Rybicki L, Nelson KA, Donnelly S. Symptoms and prognosis in advanced cancer. Support Care Cancer 2002; 10:385–388.
- Komurcu S, Nelson KA, Walsh D, Donnelly SM, Homsi J, Abdullah O. Common symptoms in advanced cancer. Semin Oncol 2000; 27:24–33.
- Homsi J, Walsh D, Rivera N, et al. Symptom evaluation in palliative medicine: patient report vs systematic assessment. Support Care Cancer 2006; 14:444–453.
- Donnelly S. Quality-of-life assessment in advanced cancer. Curr Oncol Rep 2000; 2:338–342.
- Stone P, Richardson A, Ream E, Smith AG, Kerr DJ, Kearney N. Cancer-related fatigue: inevitable, unimportant and untreatable? Results of a multi-centre patient survey. Cancer Fatigue Forum. Ann Oncol 2000; 11:971–975.
- Portenoy RK, Itri LM. Cancer-related fatigue: guidelines for evaluation and management. Oncologist 1999; 4:1–10.
- Berger AM, Abernethy AP, Atkinson A, et al. NCCN Clinical Practice Guidelines in Oncology Cancer-related fatigue—v.1.2010. www.nccn.org/professionals/physician_gls/PDF/fatigue.pdf. Accessed November 15, 2010.
- Schmitz KH, Holtzman J, Courneya KS, Mâsse LC, Duval S, Kane R. Controlled physical activity trials in cancer survivors: a systematic review and meta-analysis. Cancer Epidemiol Biomarkers Prev 2005; 14:1588–1595.
- Porock D, Kristjanson LJ, Tinnelly K, Duke T, Blight J. An exercise intervention for advanced cancer patients experiencing fatigue: a pilot study. J Palliat Care 2000 Autumn; 16:30–36.
- Breitbart W, Rosenfeld B, Kaim M, Funesti-Esch J. A randomized, double-blind, placebo-controlled trial of psychostimulants for the treatment of fatigue in ambulatory patients with human immunodeficiency virus disease. Arch Intern Med 2001; 161:411–420.
- Sarhill N, Walsh D, Nelson KA, Homsi J, LeGrand S, Davis MP. Methylphenidate for fatigue in advanced cancer: a prospective open-label pilot study. Am J Hosp Palliat Care 2001; 18:187–192.
- Minton O, Richardson A, Sharpe M, Hotopf M, Stone P. A systematic review and meta-analysis of the pharmacological treatment of cancer-related fatigue. J Natl Cancer Inst 2008; 100:1155–1166.
- Kaleita TA, Wellisch DK, Graham CA, et al. Pilot study of modafinil for treatment of neurobehavioral dysfunction and fatigue in adult patients with brain tumors (abstract). J Clin Oncol 2006; 24(suppl):58s.
- Morrow GR, Jean-Pierre P, Roscoe JA, et al. A phase III randomized, placebo-controlled, double-blind trial of a eugeroic agent in 642 cancer patients reporting fatigue during chemotherapy: a URCC CCOP study (abstract). J Clin Oncol 2008; 26(suppl):504s.
- Tannock I, Gospodarowicz M, Meakin W, Panzarella T, Stewart L, Rider W. Treatment of metastatic prostatic cancer with low-dose prednisone: evaluation of pain and quality of life as pragmatic indices of response. J Clin Oncol 1989; 7:590–597.
- Morley JE, Thomas DR, Wilson MM. Cachexia: pathophysiology and clinical relevance. Am J Clin Nutr 2006; 83:735–743.
- Poole K, Froggatt K. Loss of weight and loss of appetite in advanced cancer: a problem for the patient, the carer, or the health professional? Palliat Med 2002; 16:499–506.
- Von Roenn JH. Randomized trials of megestrol acetate for AIDS-associated anorexia and cachexia. Oncology 1994; 51(suppl 1):19–24.
- Donnelly S, Walsh TD. Low-dose megestrol acetate for appetite stimulation in advanced cancer. J Pain Symptom Manage 1995; 10:182–183.
- Mulligan K, Zackin R, Von Roenn JH, et al; ACTG 313 Study Team. Testosterone supplementation of megestrol therapy does not enhance lean tissue accrual in men with human immunodeficiency virus-associated weight loss: a randomized, double-blind, placebo-controlled, multicenter trial. J Clin Endocrinol Metab 2007; 92:563–570.
- Navari RM, Brenner MC. Treatment of cancer-related anorexia with olanzapine and megestrol acetate: a randomized trial. Support Care Cancer 2010; 18:951–956.
- Mercadante S, Fulfaro F, Casuccio A. The use of corticosteroids in home palliative care. Support Care Cancer 2001; 9:386–389.
- Walsh D, Kirkova J, Davis MP. The efficacy and tolerability of long-term use of dronabinol in cancer-related anorexia: a case series. J Pain Symptom Manage 2005; 30:493–495.
- Cannabis-In-Cachexia-Study-Group; Strasser F, Luftner D, Possinger K, et al. Comparison of orally administered cannabis extract and delta-9-tetrahydrocannabinol in treating patients with cancer-related anorexia-cachexia syndrome: a multicenter, phase III, randomized, double-blind, placebo-controlled clinical trial from the Cannabis-In-Cachexia-Study-Group. J Clin Oncol 2006; 24:3394–3400.
- Jatoi A, Windschitl HE, Loprinzi CL, et al. Dronabinol versus megestrol acetate versus combination therapy for cancer-associated anorexia: a North Central Cancer Treatment Group study. J Clin Oncol 2002; 20:567–573.
- Winter SM. Terminal nutrition: framing the debate for the withdrawal of nutritional support in terminally ill patients. Am J Med 2000; 109:723–726.
- Drossman DA, Sandler RS, McKee DC, Lovitz AJ. Bowel patterns among subjects not seeking health care. Use of a questionnaire to identify a population with bowel dysfunction. Gastroenterology 1982; 83:529–534.
- McMillan SC. Assessing and managing opiate-induced constipation in adults with cancer. Cancer Control 2004; 11(suppl 3):3–9.
- Robinson CB, Fritch M, Hullett L, et al. Development of a protocol to prevent opioid-induced constipation in patients with cancer: a research utilization project. Clin J Oncol Nurs 2000; 4:79–84.
- Thomas J, Karver S, Cooney GA, et al. Methylnaltrexone for opioid-induced constipation in advanced illness. N Engl J Med 2008; 358:2332–2343.
- Davis MP, Walsh D. Treatment of nausea and vomiting in advanced cancer. Support Care Cancer 2000; 8:444–452.
- Stephenson J, Davies A. An assessment of aetiology-based guidelines for the management of nausea and vomiting in patients with advanced cancer. Support Care Cancer 2006; 14:348–353.
- Lichter I. Results of antiemetic management in terminal illness. J Palliat Care 1993; 9:19–21.
- Bentley A, Boyd K. Use of clinical pictures in the management of nausea and vomiting: a prospective audit. Palliat Med 2001; 15:247–253.
- Glare P, Pereira G, Kristjanson LJ, Stockler M, Tattersall M. Systematic review of the efficacy of antiemetics in the treatment of nausea in patients with far-advanced cancer. Support Care Cancer 2004; 12:432–440.
- Currow DC, Coughlan M, Fardell B, Cooney NJ. Use of ondansetron in palliative medicine. J Pain Symptom Manage 1997; 13:302–307.
- Reuben DB, Mor V. Dyspnea in terminally ill cancer patients. Chest 1986; 89:234–236.
- Fainsinger RL, Waller A, Bercovici M, et al. A multicentre international study of sedation for uncontrolled symptoms in terminally ill patients. Palliat Med 2000; 14:257–265.
- Jennings AL, Davies AN, Higgins JP, Broadley K. Opioids for the palliation of breathlessness in terminal illness. Cochrane Database Syst Rev 2001;CD002066.
- Estfan B, Mahmoud F, Shaheen P, et al. Respiratory function during parenteral opioid titration for cancer pain. Palliat Med 2007; 21:81–86.
- Allen S, Raut S, Woollard J, Vassallo M. Low dose diamorphine reduces breathlessness without causing a fall in oxygen saturation in elderly patients with end-stage idiopathic pulmonary fibrosis. Palliat Med 2005; 19:128–130.
- Polosa R, Simidchiev A, Walters EH. Nebulised morphine for severe interstitial lung disease. Cochrane Database Syst Rev 2002;CD002872.
- Currow DC, Agar M, Smith J, Abernethy AP. Does palliative home oxygen improve dyspnoea? A consecutive cohort study. Palliat Med 2009; 23:309–316.
- Clemens KE, Quednau I, Klaschik E. Use of oxygen and opioids in the palliation of dyspnoea in hypoxic and non-hypoxic palliative care patients: a prospective study. Support Care Cancer 2009; 17:367–377.
- Philip J, Gold M, Milner A, Di Iulio J, Miller B, Spruyt O. A randomized, double-blind, crossover trial of the effect of oxygen on dyspnea in patients with advanced cancer. J Pain Symptom Manage 2006; 32:541–550.
KEY POINTS
- Patients with advanced cancer typically suffer from multiple concurrent symptoms, which they rate as moderate or severe.
- The principles of symptom management include taking an aggressive detailed approach, prioritizing, and identifying symptom pathophysiology.
- Prescribed regimens should be specific and simple; physicians should consider the patient’s age and fragility, the cost of the treatment, and anticipated drug side effects.
- To ensure optimal palliation with the fewest possible adverse effects, reassess frequently, make one change at a time, and use rescue doses.
Some Opioids Safer Than Others in Elderly With Nonmalignant Pain
The risks of opioid use for elderly patients with nonmalignant pain vary considerably by different agents and by different durations of use, according to a report in the Dec. 13/27 issue of Archives of Internal Medicine.
Patients taking codeine for more than 180 days are at increased risk for cardiovascular events, and those taking oxycodone or codeine for only 30 days are at increased risk for mortality due to any cause, said Dr. Daniel H. Solomon and his associates in the rheumatology department and the pharmacoepidemiology division at Brigham and Women’s Hospital, Boston.
"This study’s findings do not agree with a commonly held belief that all opioids are associated with similar risk," the investigators noted.
They compared the safety profiles of different opioids for the treatment of nonmalignant pain in elderly patients because "relatively little attention has been paid" to this issue even though the use of these drugs has increased by 50%-100% in recent years. In contrast, patients and physicians are relatively well informed about the toxicities of NSAIDs used for the same indications.
Dr. Solomon and his colleagues analyzed information in two states’ Medicare databases of pharmaceutical coverage for low-income patients (mean age, 79 years) between 1995 and 2005. They were able to match 6,275 subjects who took five of the most commonly prescribed opioids for a variety of baseline factors using propensity scores.
Hydrocodone was used as the reference exposure, to which codeine, oxycodone, propoxyphene, and tramadol were compared.
The risk of cardiovascular events including MI, stroke, heart failure, revascularization, and cardiac death was similar across the five opioid groups after 30 days of use. However, by 180 days the cardiovascular risk with codeine was significantly elevated (risk ratio, 1.62), compared with the other four opioids.
This finding is surprising and requires validation in other study populations, the investigators said (Arch. Intern. Med. 2010;170:1979-86).
All-cause mortality was elevated after only 30 days of use for patients taking oxycodone (RR, 2.43) or codeine (RR, 2.05), but not for those taking other opioids.
In contrast, the risk of fracture of the hip, pelvis, wrist, or humerus was significantly reduced after 30 days of treatment for patients taking tramadol (RR, 0.21) or propoxyphene (RR, 0.54).
The risk of gastrointestinal adverse events – which included upper GI bleeding, lower GI bleeding, and bowel obstruction – did not differ across opioid groups at either 30 days or 180 days.
These risks remained consistent through a range of sensitivity analyses of the data. Importantly, the risks also were consistent regardless of whether patients were taking low, medium, high, or very high doses of the drugs. "The risks were substantial and translated into numbers needed to treat that would be considered clinically significant," Dr. Solomon and his associates wrote.
This study was supported by the Agency for Healthcare Research and Quality. Dr. Solomon reported being an unpaid member of a celecoxib trial executive committee sponsored by Pfizer and an unpaid member of the data safety monitoring board for an analgesic trial sponsored by Pfizer.
The findings by Dr. Solomon and his associates "challenge the conventional notion that the safety profiles of opioids are generally interchangeable" and carry two important clinical implications, said Dr. William C. Becker and Dr. Patrick G. O’Connor.
The first and most crucial implication is that the frequent use of codeine must be reexamined. "The untested but widespread assumption that codeine is safer from an addiction standpoint because of its lower potency may need to give way to these data demonstrating increased risk of cardiovascular events and all-cause mortality. If codeine is of middling efficacy for pain and is more risky than other opioids, there would be little reason to use it," they noted.
Secondly, the elevated risk of fractures with opioid use "has solid biological plausibility" by two mechanisms of action: Opioids raise the rate of falls, and they suppress the production of androgen and estradiol, imperiling bone health. The study findings suggest that basic safety measures to counterbalance these effects are not being implemented. "Efforts to improve patients’ understanding of safe medication-taking practices, providers’ understanding of safe prescribing practices, and standardization of safety-oriented follow-up are sorely needed," Dr. Becker and Dr. O’Connor said.
Dr. Becker and Dr. O’Connor are internists at Yale University, New Haven, Conn. They reported no relevant financial disclosures. These comments were taken from their invited commentary accompanying Dr. Solomon’s report (Arch. Intern. Med. 2010;170:1986-8).
The findings by Dr. Solomon and his associates "challenge the conventional notion that the safety profiles of opioids are generally interchangeable" and carry two important clinical implications, said Dr. William C. Becker and Dr. Patrick G. O’Connor.
The first and most crucial implication is that the frequent use of codeine must be reexamined. "The untested but widespread assumption that codeine is safer from an addiction standpoint because of its lower potency may need to give way to these data demonstrating increased risk of cardiovascular events and all-cause mortality. If codeine is of middling efficacy for pain and is more risky than other opioids, there would be little reason to use it," they noted.
Secondly, the elevated risk of fractures with opioid use "has solid biological plausibility" by two mechanisms of action: Opioids raise the rate of falls, and they suppress the production of androgen and estradiol, imperiling bone health. The study findings suggest that basic safety measures to counterbalance these effects are not being implemented. "Efforts to improve patients’ understanding of safe medication-taking practices, providers’ understanding of safe prescribing practices, and standardization of safety-oriented follow-up are sorely needed," Dr. Becker and Dr. O’Connor said.
Dr. Becker and Dr. O’Connor are internists at Yale University, New Haven, Conn. They reported no relevant financial disclosures. These comments were taken from their invited commentary accompanying Dr. Solomon’s report (Arch. Intern. Med. 2010;170:1986-8).
The findings by Dr. Solomon and his associates "challenge the conventional notion that the safety profiles of opioids are generally interchangeable" and carry two important clinical implications, said Dr. William C. Becker and Dr. Patrick G. O’Connor.
The first and most crucial implication is that the frequent use of codeine must be reexamined. "The untested but widespread assumption that codeine is safer from an addiction standpoint because of its lower potency may need to give way to these data demonstrating increased risk of cardiovascular events and all-cause mortality. If codeine is of middling efficacy for pain and is more risky than other opioids, there would be little reason to use it," they noted.
Secondly, the elevated risk of fractures with opioid use "has solid biological plausibility" by two mechanisms of action: Opioids raise the rate of falls, and they suppress the production of androgen and estradiol, imperiling bone health. The study findings suggest that basic safety measures to counterbalance these effects are not being implemented. "Efforts to improve patients’ understanding of safe medication-taking practices, providers’ understanding of safe prescribing practices, and standardization of safety-oriented follow-up are sorely needed," Dr. Becker and Dr. O’Connor said.
Dr. Becker and Dr. O’Connor are internists at Yale University, New Haven, Conn. They reported no relevant financial disclosures. These comments were taken from their invited commentary accompanying Dr. Solomon’s report (Arch. Intern. Med. 2010;170:1986-8).
The risks of opioid use for elderly patients with nonmalignant pain vary considerably by different agents and by different durations of use, according to a report in the Dec. 13/27 issue of Archives of Internal Medicine.
Patients taking codeine for more than 180 days are at increased risk for cardiovascular events, and those taking oxycodone or codeine for only 30 days are at increased risk for mortality due to any cause, said Dr. Daniel H. Solomon and his associates in the rheumatology department and the pharmacoepidemiology division at Brigham and Women’s Hospital, Boston.
"This study’s findings do not agree with a commonly held belief that all opioids are associated with similar risk," the investigators noted.
They compared the safety profiles of different opioids for the treatment of nonmalignant pain in elderly patients because "relatively little attention has been paid" to this issue even though the use of these drugs has increased by 50%-100% in recent years. In contrast, patients and physicians are relatively well informed about the toxicities of NSAIDs used for the same indications.
Dr. Solomon and his colleagues analyzed information in two states’ Medicare databases of pharmaceutical coverage for low-income patients (mean age, 79 years) between 1995 and 2005. They were able to match 6,275 subjects who took five of the most commonly prescribed opioids for a variety of baseline factors using propensity scores.
Hydrocodone was used as the reference exposure, to which codeine, oxycodone, propoxyphene, and tramadol were compared.
The risk of cardiovascular events including MI, stroke, heart failure, revascularization, and cardiac death was similar across the five opioid groups after 30 days of use. However, by 180 days the cardiovascular risk with codeine was significantly elevated (risk ratio, 1.62), compared with the other four opioids.
This finding is surprising and requires validation in other study populations, the investigators said (Arch. Intern. Med. 2010;170:1979-86).
All-cause mortality was elevated after only 30 days of use for patients taking oxycodone (RR, 2.43) or codeine (RR, 2.05), but not for those taking other opioids.
In contrast, the risk of fracture of the hip, pelvis, wrist, or humerus was significantly reduced after 30 days of treatment for patients taking tramadol (RR, 0.21) or propoxyphene (RR, 0.54).
The risk of gastrointestinal adverse events – which included upper GI bleeding, lower GI bleeding, and bowel obstruction – did not differ across opioid groups at either 30 days or 180 days.
These risks remained consistent through a range of sensitivity analyses of the data. Importantly, the risks also were consistent regardless of whether patients were taking low, medium, high, or very high doses of the drugs. "The risks were substantial and translated into numbers needed to treat that would be considered clinically significant," Dr. Solomon and his associates wrote.
This study was supported by the Agency for Healthcare Research and Quality. Dr. Solomon reported being an unpaid member of a celecoxib trial executive committee sponsored by Pfizer and an unpaid member of the data safety monitoring board for an analgesic trial sponsored by Pfizer.
The risks of opioid use for elderly patients with nonmalignant pain vary considerably by different agents and by different durations of use, according to a report in the Dec. 13/27 issue of Archives of Internal Medicine.
Patients taking codeine for more than 180 days are at increased risk for cardiovascular events, and those taking oxycodone or codeine for only 30 days are at increased risk for mortality due to any cause, said Dr. Daniel H. Solomon and his associates in the rheumatology department and the pharmacoepidemiology division at Brigham and Women’s Hospital, Boston.
"This study’s findings do not agree with a commonly held belief that all opioids are associated with similar risk," the investigators noted.
They compared the safety profiles of different opioids for the treatment of nonmalignant pain in elderly patients because "relatively little attention has been paid" to this issue even though the use of these drugs has increased by 50%-100% in recent years. In contrast, patients and physicians are relatively well informed about the toxicities of NSAIDs used for the same indications.
Dr. Solomon and his colleagues analyzed information in two states’ Medicare databases of pharmaceutical coverage for low-income patients (mean age, 79 years) between 1995 and 2005. They were able to match 6,275 subjects who took five of the most commonly prescribed opioids for a variety of baseline factors using propensity scores.
Hydrocodone was used as the reference exposure, to which codeine, oxycodone, propoxyphene, and tramadol were compared.
The risk of cardiovascular events including MI, stroke, heart failure, revascularization, and cardiac death was similar across the five opioid groups after 30 days of use. However, by 180 days the cardiovascular risk with codeine was significantly elevated (risk ratio, 1.62), compared with the other four opioids.
This finding is surprising and requires validation in other study populations, the investigators said (Arch. Intern. Med. 2010;170:1979-86).
All-cause mortality was elevated after only 30 days of use for patients taking oxycodone (RR, 2.43) or codeine (RR, 2.05), but not for those taking other opioids.
In contrast, the risk of fracture of the hip, pelvis, wrist, or humerus was significantly reduced after 30 days of treatment for patients taking tramadol (RR, 0.21) or propoxyphene (RR, 0.54).
The risk of gastrointestinal adverse events – which included upper GI bleeding, lower GI bleeding, and bowel obstruction – did not differ across opioid groups at either 30 days or 180 days.
These risks remained consistent through a range of sensitivity analyses of the data. Importantly, the risks also were consistent regardless of whether patients were taking low, medium, high, or very high doses of the drugs. "The risks were substantial and translated into numbers needed to treat that would be considered clinically significant," Dr. Solomon and his associates wrote.
This study was supported by the Agency for Healthcare Research and Quality. Dr. Solomon reported being an unpaid member of a celecoxib trial executive committee sponsored by Pfizer and an unpaid member of the data safety monitoring board for an analgesic trial sponsored by Pfizer.
FROM ARCHIVES OF INTERNAL MEDICINE
Survival Improved After Colorectal Liver/Lung Mets Resection
CHICAGO – Resection of liver and lung colorectal metastases produced superior survival, compared with resection of liver metastases alone, in a retrospective analysis of 1,260 consecutive patients.
After a median follow-up of 49 months, 5-year overall survival was 40% in patients with colorectal cancer who underwent resection of liver metastases alone, compared with 50% in those who underwent resection of both liver and lung metastases (P = .01), Dr. Eddie Abdalla reported at the annual meeting of the Western Surgical Association.
"Survival following resection of liver and lung metastases is excellent, and may even be better than [survival] for the general population that undergoes resection for colorectal lung metastases alone," he said.
Overall survival at 5 years is about 40% in patients with resected colorectal lung metastases, and ranges from 11% to 31% in the limited studies involving patients with colorectal liver-plus-lung metastases.
The presumed explanation for the improved survival is that patients with liver and lung metastases had less-extensive recurrence (27%) than did the rest of the cohort (69%), said Dr. Abdalla of the University of Texas M.D. Anderson Cancer Center in Houston. Recurrence also was seen in 33% of patients with lung-only metastases and in 22% with liver-only metastases.
Although recurrence in the study was substantial, it can often be retreated, he observed. Among 78 patients who developed recurrence, 33 (42%) were retreated with resection, radiofrequency ablation, or chemotherapy, resulting in a 5-year disease-free survival of 25%.
Extrahepatic disease, long associated with poor outcome, has been considered a contraindication to resection. More recent studies are challenging this belief, citing advances in systemic chemotherapy and surgical technique as well as improved multidisciplinary management and patient selection.
In multivariate analysis, the only significant predictors of worse survival in patients with resection of both liver and lung metastases were a rectal primary tumor (P = .004; hazard ratio, 2.9) and a carcinoembryonic antigen (CEA) level before resection of liver metastases of more than 5 ng/mL (P = .04; HR, 2.1), Dr. Abdalla said. Interestingly, liver metastasis greater than 5 cm, positive margins at resection of liver metastases, synchronous lung metastases, and even disease-free intervals were not significant.
"Occurrence of lung metastases that are subsequently resected is not a poor prognostic factor or a contraindication to resection of colorectal liver metastases," he said.
In all, 32 of the 112 patients with liver-plus-lung metastases had a rectal primary tumor, compared with 254 of the 1,148 patients with liver-only metastases. Their median preoperative CEA levels were 3.2 and 3.7, respectively. Preoperative chemotherapy was used in 69% of patients before resection of liver metastases and in 50% of patients before resection of lung metastases.
Invited discussant Dr. John Brems of Loyola University Medical Center in Maywood, Ill., asked whether the survival outcomes were influenced by selecting patients with a higher rate of chemotherapy utilization in the liver-plus-lung group, and by considering patients for surgery if they had no increase in tumor size on chemotherapy.
"There’s no question that we are selecting patients, and the objective of selection is to deliver treatment to patients who will benefit," Dr. Abdalla said. "Response to chemotherapy appears to be a good way to select patients for resection of colorectal liver metastases."
Dr. Abdalla referenced a recent study he coauthored that reported a significantly longer overall survival among patients with colorectal liver metastases who had an optimal morphological response on imaging to bevacizumab-containing chemotherapy prior to hepatic resection, compared with those who had an incomplete or no response (median, 31 months vs. 19 months) (JAMA. 2009;302:2338-44).
During the discussion, Dr. Anton Bilchik of the University of California, Los Angeles, asked whether the researchers treat disappearing lung and liver lesions in the same manner.
Dr. Abdalla said that they tend to follow the disappearing lung lesion, but resect the disappearing liver lesion. "At this point, if you have a patient with a tiny liver lesion that disappears, it’s difficult to propose a major hepatectomy to take out a central disappearing lesion," he said. "However, we know from follow-up and resection studies that most of those – certainly over 90% – will recur. We don’t have that kind of data in the lung."
None of the speakers disclosed any conflicts of interest.
CHICAGO – Resection of liver and lung colorectal metastases produced superior survival, compared with resection of liver metastases alone, in a retrospective analysis of 1,260 consecutive patients.
After a median follow-up of 49 months, 5-year overall survival was 40% in patients with colorectal cancer who underwent resection of liver metastases alone, compared with 50% in those who underwent resection of both liver and lung metastases (P = .01), Dr. Eddie Abdalla reported at the annual meeting of the Western Surgical Association.
"Survival following resection of liver and lung metastases is excellent, and may even be better than [survival] for the general population that undergoes resection for colorectal lung metastases alone," he said.
Overall survival at 5 years is about 40% in patients with resected colorectal lung metastases, and ranges from 11% to 31% in the limited studies involving patients with colorectal liver-plus-lung metastases.
The presumed explanation for the improved survival is that patients with liver and lung metastases had less-extensive recurrence (27%) than did the rest of the cohort (69%), said Dr. Abdalla of the University of Texas M.D. Anderson Cancer Center in Houston. Recurrence also was seen in 33% of patients with lung-only metastases and in 22% with liver-only metastases.
Although recurrence in the study was substantial, it can often be retreated, he observed. Among 78 patients who developed recurrence, 33 (42%) were retreated with resection, radiofrequency ablation, or chemotherapy, resulting in a 5-year disease-free survival of 25%.
Extrahepatic disease, long associated with poor outcome, has been considered a contraindication to resection. More recent studies are challenging this belief, citing advances in systemic chemotherapy and surgical technique as well as improved multidisciplinary management and patient selection.
In multivariate analysis, the only significant predictors of worse survival in patients with resection of both liver and lung metastases were a rectal primary tumor (P = .004; hazard ratio, 2.9) and a carcinoembryonic antigen (CEA) level before resection of liver metastases of more than 5 ng/mL (P = .04; HR, 2.1), Dr. Abdalla said. Interestingly, liver metastasis greater than 5 cm, positive margins at resection of liver metastases, synchronous lung metastases, and even disease-free intervals were not significant.
"Occurrence of lung metastases that are subsequently resected is not a poor prognostic factor or a contraindication to resection of colorectal liver metastases," he said.
In all, 32 of the 112 patients with liver-plus-lung metastases had a rectal primary tumor, compared with 254 of the 1,148 patients with liver-only metastases. Their median preoperative CEA levels were 3.2 and 3.7, respectively. Preoperative chemotherapy was used in 69% of patients before resection of liver metastases and in 50% of patients before resection of lung metastases.
Invited discussant Dr. John Brems of Loyola University Medical Center in Maywood, Ill., asked whether the survival outcomes were influenced by selecting patients with a higher rate of chemotherapy utilization in the liver-plus-lung group, and by considering patients for surgery if they had no increase in tumor size on chemotherapy.
"There’s no question that we are selecting patients, and the objective of selection is to deliver treatment to patients who will benefit," Dr. Abdalla said. "Response to chemotherapy appears to be a good way to select patients for resection of colorectal liver metastases."
Dr. Abdalla referenced a recent study he coauthored that reported a significantly longer overall survival among patients with colorectal liver metastases who had an optimal morphological response on imaging to bevacizumab-containing chemotherapy prior to hepatic resection, compared with those who had an incomplete or no response (median, 31 months vs. 19 months) (JAMA. 2009;302:2338-44).
During the discussion, Dr. Anton Bilchik of the University of California, Los Angeles, asked whether the researchers treat disappearing lung and liver lesions in the same manner.
Dr. Abdalla said that they tend to follow the disappearing lung lesion, but resect the disappearing liver lesion. "At this point, if you have a patient with a tiny liver lesion that disappears, it’s difficult to propose a major hepatectomy to take out a central disappearing lesion," he said. "However, we know from follow-up and resection studies that most of those – certainly over 90% – will recur. We don’t have that kind of data in the lung."
None of the speakers disclosed any conflicts of interest.
CHICAGO – Resection of liver and lung colorectal metastases produced superior survival, compared with resection of liver metastases alone, in a retrospective analysis of 1,260 consecutive patients.
After a median follow-up of 49 months, 5-year overall survival was 40% in patients with colorectal cancer who underwent resection of liver metastases alone, compared with 50% in those who underwent resection of both liver and lung metastases (P = .01), Dr. Eddie Abdalla reported at the annual meeting of the Western Surgical Association.
"Survival following resection of liver and lung metastases is excellent, and may even be better than [survival] for the general population that undergoes resection for colorectal lung metastases alone," he said.
Overall survival at 5 years is about 40% in patients with resected colorectal lung metastases, and ranges from 11% to 31% in the limited studies involving patients with colorectal liver-plus-lung metastases.
The presumed explanation for the improved survival is that patients with liver and lung metastases had less-extensive recurrence (27%) than did the rest of the cohort (69%), said Dr. Abdalla of the University of Texas M.D. Anderson Cancer Center in Houston. Recurrence also was seen in 33% of patients with lung-only metastases and in 22% with liver-only metastases.
Although recurrence in the study was substantial, it can often be retreated, he observed. Among 78 patients who developed recurrence, 33 (42%) were retreated with resection, radiofrequency ablation, or chemotherapy, resulting in a 5-year disease-free survival of 25%.
Extrahepatic disease, long associated with poor outcome, has been considered a contraindication to resection. More recent studies are challenging this belief, citing advances in systemic chemotherapy and surgical technique as well as improved multidisciplinary management and patient selection.
In multivariate analysis, the only significant predictors of worse survival in patients with resection of both liver and lung metastases were a rectal primary tumor (P = .004; hazard ratio, 2.9) and a carcinoembryonic antigen (CEA) level before resection of liver metastases of more than 5 ng/mL (P = .04; HR, 2.1), Dr. Abdalla said. Interestingly, liver metastasis greater than 5 cm, positive margins at resection of liver metastases, synchronous lung metastases, and even disease-free intervals were not significant.
"Occurrence of lung metastases that are subsequently resected is not a poor prognostic factor or a contraindication to resection of colorectal liver metastases," he said.
In all, 32 of the 112 patients with liver-plus-lung metastases had a rectal primary tumor, compared with 254 of the 1,148 patients with liver-only metastases. Their median preoperative CEA levels were 3.2 and 3.7, respectively. Preoperative chemotherapy was used in 69% of patients before resection of liver metastases and in 50% of patients before resection of lung metastases.
Invited discussant Dr. John Brems of Loyola University Medical Center in Maywood, Ill., asked whether the survival outcomes were influenced by selecting patients with a higher rate of chemotherapy utilization in the liver-plus-lung group, and by considering patients for surgery if they had no increase in tumor size on chemotherapy.
"There’s no question that we are selecting patients, and the objective of selection is to deliver treatment to patients who will benefit," Dr. Abdalla said. "Response to chemotherapy appears to be a good way to select patients for resection of colorectal liver metastases."
Dr. Abdalla referenced a recent study he coauthored that reported a significantly longer overall survival among patients with colorectal liver metastases who had an optimal morphological response on imaging to bevacizumab-containing chemotherapy prior to hepatic resection, compared with those who had an incomplete or no response (median, 31 months vs. 19 months) (JAMA. 2009;302:2338-44).
During the discussion, Dr. Anton Bilchik of the University of California, Los Angeles, asked whether the researchers treat disappearing lung and liver lesions in the same manner.
Dr. Abdalla said that they tend to follow the disappearing lung lesion, but resect the disappearing liver lesion. "At this point, if you have a patient with a tiny liver lesion that disappears, it’s difficult to propose a major hepatectomy to take out a central disappearing lesion," he said. "However, we know from follow-up and resection studies that most of those – certainly over 90% – will recur. We don’t have that kind of data in the lung."
None of the speakers disclosed any conflicts of interest.
New Drugs Target Recurrent Meningioma
MONTREAL – The tyrosine kinase inhibitors vatalanib and sunitinib may offer alternative approaches to treating patients with recurrent, high-grade meningioma who have failed surgery and radiation therapy, according to two separate phase II studies.
Both drugs significantly improved survival compared with what is generally expected in this patient population, most of whom had grade II or III tumors. Progression-free survival at 6 months occurred in 57% of 25 patients with meningioma who received the investigational drug vatalanib in one study, and in 36% of 36 patients who received sunitinib in another study. Both drugs are inhibitors of receptors for vascular endothelial growth factor and platelet-derived growth factor.
Sunitinib is marketed by the brand name Sutent and has been approved by the Food and Drug Administration for the treatment of advanced renal cell carcinoma and gastrointestinal stromal tumor after disease progression on – or intolerance to – imatinib mesylate (Gleevec).
Progression-free survival (PFS) for patients with recurrent grade II and III tumors is usually considered to occur in fewer than 5%, said Dr. Jeffrey Raizer, who presented the vatalanib study at the annual meeting of the Society for Neuro-Oncology.
"For recurrence of meningioma, treatment options are limited once patients have failed surgery and radiation, and chemotherapies have been very limited to date," said Dr. Raizer, director of medical neuro-oncology at the cancer center of Northwestern University and Northwestern Memorial Hospital, both in Chicago.
Despite the encouraging findings of the two studies, a lack of information on the natural history of these tumors makes it difficult to interpret the studies, said Dr. Michael Vogelbaum, who chaired the session in which the studies were presented.
"While the studies suggest there may be some promise, we don’t really know what to expect from that mix of grade II and III tumors on their own," Dr. Vogelbaum said in an interview. He is chair of neuro-oncology and associate director of the brain tumor and neuro-oncology center at the Cleveland Clinic Neurological Institute.
Dr. Fred Barker of the department of neurosurgery at Massachusetts General Hospital, Boston, echoed Dr. Vogelbaum’s comments. "There’s been very little organized effort to do chemotherapy trials for meningiomas, so these studies are important because they are the first. The results are interesting, but we don’t know exactly what the natural history would be without treatment, so whether there’s a benefit to giving these drugs will require further study."
In Dr. Raizer’s Pfizer-sponsored study of vatalanib in meningioma patients, the median PFS was 5.9 months, and the median overall survival (OS) was 22.3 months. Of the 25 patients in the study, 22 had grade II and III tumors and 14 were men. The PFS at 6 months was comparable between patients with grade II (39%) or grade III disease only (43%).
Dosing of vatalanib began at 250 mg twice daily and increased by 250 mg/day every 7 days until the dosage reached 500 mg twice daily, he said.
For 21 patients with available imaging data, tumors partially responded to treatment in 1, remained stable in 15, and progressed in 5 patients.
The most common adverse events observed were fatigue, rash, and elevated transaminases.
The sunitinib study, sponsored by Novartis, included 36 patients (median age, 62 years) with recurrent grade II and III meningiomas who had exhausted all surgical and radiation options. The patients had a median of five recurrences.
The primary end point of PFS at 6 months was 36% and the median PFS was 5.1 months. The overall survival rate is not known, as only eight patients have died, reported Dr. Thomas Kaley, codirector of the neuro-oncology fellowship program at Memorial Sloan-Kettering Cancer Center in New York.
Imaging was available for 34 patients and showed stable disease in 26, a partial response in 1, and progressive disease in 7 patients, he said.
The dose of sunitinib was 50 mg orally per day on a 4-week-on, 2-week-off cycle.
"The toxicity was high with this drug," Dr. Kaley commented.
A total of 18 patients required dose reductions, and 6 patients withdrew from the study because of toxicity. There was one fatal grade 5 intratumoral hemorrhage and two nonfatal intratumoral hemorrhages (grades 3 and 4). The most common toxicities included myelosuppression, fatigue, headache, and GI side effects.
The investigators reported having no relevant conflicts of interest.*
* CORRECTION, 1/17/2011: An earlier version of this story included incorrect information about the investigators' disclosures. The error has been corrected.
MONTREAL – The tyrosine kinase inhibitors vatalanib and sunitinib may offer alternative approaches to treating patients with recurrent, high-grade meningioma who have failed surgery and radiation therapy, according to two separate phase II studies.
Both drugs significantly improved survival compared with what is generally expected in this patient population, most of whom had grade II or III tumors. Progression-free survival at 6 months occurred in 57% of 25 patients with meningioma who received the investigational drug vatalanib in one study, and in 36% of 36 patients who received sunitinib in another study. Both drugs are inhibitors of receptors for vascular endothelial growth factor and platelet-derived growth factor.
Sunitinib is marketed by the brand name Sutent and has been approved by the Food and Drug Administration for the treatment of advanced renal cell carcinoma and gastrointestinal stromal tumor after disease progression on – or intolerance to – imatinib mesylate (Gleevec).
Progression-free survival (PFS) for patients with recurrent grade II and III tumors is usually considered to occur in fewer than 5%, said Dr. Jeffrey Raizer, who presented the vatalanib study at the annual meeting of the Society for Neuro-Oncology.
"For recurrence of meningioma, treatment options are limited once patients have failed surgery and radiation, and chemotherapies have been very limited to date," said Dr. Raizer, director of medical neuro-oncology at the cancer center of Northwestern University and Northwestern Memorial Hospital, both in Chicago.
Despite the encouraging findings of the two studies, a lack of information on the natural history of these tumors makes it difficult to interpret the studies, said Dr. Michael Vogelbaum, who chaired the session in which the studies were presented.
"While the studies suggest there may be some promise, we don’t really know what to expect from that mix of grade II and III tumors on their own," Dr. Vogelbaum said in an interview. He is chair of neuro-oncology and associate director of the brain tumor and neuro-oncology center at the Cleveland Clinic Neurological Institute.
Dr. Fred Barker of the department of neurosurgery at Massachusetts General Hospital, Boston, echoed Dr. Vogelbaum’s comments. "There’s been very little organized effort to do chemotherapy trials for meningiomas, so these studies are important because they are the first. The results are interesting, but we don’t know exactly what the natural history would be without treatment, so whether there’s a benefit to giving these drugs will require further study."
In Dr. Raizer’s Pfizer-sponsored study of vatalanib in meningioma patients, the median PFS was 5.9 months, and the median overall survival (OS) was 22.3 months. Of the 25 patients in the study, 22 had grade II and III tumors and 14 were men. The PFS at 6 months was comparable between patients with grade II (39%) or grade III disease only (43%).
Dosing of vatalanib began at 250 mg twice daily and increased by 250 mg/day every 7 days until the dosage reached 500 mg twice daily, he said.
For 21 patients with available imaging data, tumors partially responded to treatment in 1, remained stable in 15, and progressed in 5 patients.
The most common adverse events observed were fatigue, rash, and elevated transaminases.
The sunitinib study, sponsored by Novartis, included 36 patients (median age, 62 years) with recurrent grade II and III meningiomas who had exhausted all surgical and radiation options. The patients had a median of five recurrences.
The primary end point of PFS at 6 months was 36% and the median PFS was 5.1 months. The overall survival rate is not known, as only eight patients have died, reported Dr. Thomas Kaley, codirector of the neuro-oncology fellowship program at Memorial Sloan-Kettering Cancer Center in New York.
Imaging was available for 34 patients and showed stable disease in 26, a partial response in 1, and progressive disease in 7 patients, he said.
The dose of sunitinib was 50 mg orally per day on a 4-week-on, 2-week-off cycle.
"The toxicity was high with this drug," Dr. Kaley commented.
A total of 18 patients required dose reductions, and 6 patients withdrew from the study because of toxicity. There was one fatal grade 5 intratumoral hemorrhage and two nonfatal intratumoral hemorrhages (grades 3 and 4). The most common toxicities included myelosuppression, fatigue, headache, and GI side effects.
The investigators reported having no relevant conflicts of interest.*
* CORRECTION, 1/17/2011: An earlier version of this story included incorrect information about the investigators' disclosures. The error has been corrected.
MONTREAL – The tyrosine kinase inhibitors vatalanib and sunitinib may offer alternative approaches to treating patients with recurrent, high-grade meningioma who have failed surgery and radiation therapy, according to two separate phase II studies.
Both drugs significantly improved survival compared with what is generally expected in this patient population, most of whom had grade II or III tumors. Progression-free survival at 6 months occurred in 57% of 25 patients with meningioma who received the investigational drug vatalanib in one study, and in 36% of 36 patients who received sunitinib in another study. Both drugs are inhibitors of receptors for vascular endothelial growth factor and platelet-derived growth factor.
Sunitinib is marketed by the brand name Sutent and has been approved by the Food and Drug Administration for the treatment of advanced renal cell carcinoma and gastrointestinal stromal tumor after disease progression on – or intolerance to – imatinib mesylate (Gleevec).
Progression-free survival (PFS) for patients with recurrent grade II and III tumors is usually considered to occur in fewer than 5%, said Dr. Jeffrey Raizer, who presented the vatalanib study at the annual meeting of the Society for Neuro-Oncology.
"For recurrence of meningioma, treatment options are limited once patients have failed surgery and radiation, and chemotherapies have been very limited to date," said Dr. Raizer, director of medical neuro-oncology at the cancer center of Northwestern University and Northwestern Memorial Hospital, both in Chicago.
Despite the encouraging findings of the two studies, a lack of information on the natural history of these tumors makes it difficult to interpret the studies, said Dr. Michael Vogelbaum, who chaired the session in which the studies were presented.
"While the studies suggest there may be some promise, we don’t really know what to expect from that mix of grade II and III tumors on their own," Dr. Vogelbaum said in an interview. He is chair of neuro-oncology and associate director of the brain tumor and neuro-oncology center at the Cleveland Clinic Neurological Institute.
Dr. Fred Barker of the department of neurosurgery at Massachusetts General Hospital, Boston, echoed Dr. Vogelbaum’s comments. "There’s been very little organized effort to do chemotherapy trials for meningiomas, so these studies are important because they are the first. The results are interesting, but we don’t know exactly what the natural history would be without treatment, so whether there’s a benefit to giving these drugs will require further study."
In Dr. Raizer’s Pfizer-sponsored study of vatalanib in meningioma patients, the median PFS was 5.9 months, and the median overall survival (OS) was 22.3 months. Of the 25 patients in the study, 22 had grade II and III tumors and 14 were men. The PFS at 6 months was comparable between patients with grade II (39%) or grade III disease only (43%).
Dosing of vatalanib began at 250 mg twice daily and increased by 250 mg/day every 7 days until the dosage reached 500 mg twice daily, he said.
For 21 patients with available imaging data, tumors partially responded to treatment in 1, remained stable in 15, and progressed in 5 patients.
The most common adverse events observed were fatigue, rash, and elevated transaminases.
The sunitinib study, sponsored by Novartis, included 36 patients (median age, 62 years) with recurrent grade II and III meningiomas who had exhausted all surgical and radiation options. The patients had a median of five recurrences.
The primary end point of PFS at 6 months was 36% and the median PFS was 5.1 months. The overall survival rate is not known, as only eight patients have died, reported Dr. Thomas Kaley, codirector of the neuro-oncology fellowship program at Memorial Sloan-Kettering Cancer Center in New York.
Imaging was available for 34 patients and showed stable disease in 26, a partial response in 1, and progressive disease in 7 patients, he said.
The dose of sunitinib was 50 mg orally per day on a 4-week-on, 2-week-off cycle.
"The toxicity was high with this drug," Dr. Kaley commented.
A total of 18 patients required dose reductions, and 6 patients withdrew from the study because of toxicity. There was one fatal grade 5 intratumoral hemorrhage and two nonfatal intratumoral hemorrhages (grades 3 and 4). The most common toxicities included myelosuppression, fatigue, headache, and GI side effects.
The investigators reported having no relevant conflicts of interest.*
* CORRECTION, 1/17/2011: An earlier version of this story included incorrect information about the investigators' disclosures. The error has been corrected.
One-Third of Medicare Patients With Cancer Die in Hospitals
Approximately 29% of Medicare patients with cancer died in a hospital, and hospice care was inconsistent among medical centers in the United States, according to a report published Nov. 16 by the Dartmouth Institute for Health Policy & Clinical Practice.
The rate of hospital deaths varied widely by region, ranging from 47% in Manhattan (New York City) to 7% in Mason City, Iowa.
"The bottom line is that the care patients receive has less to do with what they want and more to do with the hospital they seek care from," Dr. David Goodman of Dartmouth University, Hanover, N.H., said in a press briefing. "We as physicians often make assumptions about what patients and families want for their lives. What patients really want is for a physician to be honest with them and to share the full range of options with them."
The report, "Quality of End-of-Life Cancer Care for Medicare Beneficiaries: Regional and Hospital-Specific Analyses," is the first to focus on cancer care at the end of life for the Medicare population in particular. The report was produced by the Dartmouth Atlas Project, which has been documenting the use of medical resources in the United States for more than 2 decades.
Researchers reviewed data from 235,821 Medicare patients (aged 65 years and older) with metastatic or aggressive cancer who died in 2003-2007. They identified regional trends as well as information about specific academic medical centers.
Nationwide, approximately 61% of cancer patients were hospitalized at least once during their last month of life. Hospital admissions varied from approximately 70% of cancer patients in Detroit to 46% of cancer patients in San Angelo, Tex.
The researchers identified the highest rates of hospitalization during the last month of life (more than 75%) among cancer patients at two hospitals in Detroit: St. John Hospital and Medical Center and Sinai-Grace Hospital. The lowest rate of hospitalization during the last month of life was 45% of cancer patients at the University of Washington Medical Center in Seattle.
Chemotherapy was part of care during the last 2 weeks of life in approximately 6% of patients nationwide, but the rate exceeded 10% in eight areas, including Olympia, Wash. (12%), and San Antonio (11%).
The researchers also found that fewer than 50% of cancer patients with a poor prognosis received hospice care, and some centers provided hospice care only in the last few days of life.
The degree of variation in the findings is too great to be explained by racial or ethnic preferences for aggressive end-of-life care, and adjustment for socioeconomic status does not change the results, said Dr. Goodman said.
But the data show that investment in beds, ICU facilities, and palliative care services is quite different from one health care system to another, he said.
The report highlights the need for doctors and health systems "to take a look at themselves and examine where they need to make their investments," said Dr. Goodman, who added that clinicians "need to look at ourselves in terms of how we communicate with patients" about cancer care options.
In a commentary accompanying the report, Dr. Joan Teno of Brown University in Providence, R.I., emphasized the need for health care providers to educate patients about their prognoses and to provide hospice options.
"If opportunities to improve are identified, hospitals should work with the local hospice or existing palliative care consult service, and/or start a palliative care consult service," to ensure that the institution is delivering high-quality care, Dr. Teno wrote.
Conversations about end-of-life care need to be started early in the course of treatment, Dr. Goodman said. "Our preferences can certainly change, but opening up the conversation early allows the conversation to evolve over time. The tragedy is when those conversations start near the end."
In a statement, Dr. George W. Sledge Jr., president of the American Society of Clinical Oncology said, "While saving lives is the oncologist’s goal, helping individuals live their final days in comfort and dignity is one of the most valuable and important responsibilities of our profession.
"Options to enhance a patient’s quality of life should be discussed throughout the course of their treatment. It is critical that we increase medical training in end-of-life patient communication and planning, and that Medicare and other insurers cover the costs of these essential conversations," Dr. Sledge noted.
The study was supported in part by the Robert Wood Johnson Foundation.
Approximately 29% of Medicare patients with cancer died in a hospital, and hospice care was inconsistent among medical centers in the United States, according to a report published Nov. 16 by the Dartmouth Institute for Health Policy & Clinical Practice.
The rate of hospital deaths varied widely by region, ranging from 47% in Manhattan (New York City) to 7% in Mason City, Iowa.
"The bottom line is that the care patients receive has less to do with what they want and more to do with the hospital they seek care from," Dr. David Goodman of Dartmouth University, Hanover, N.H., said in a press briefing. "We as physicians often make assumptions about what patients and families want for their lives. What patients really want is for a physician to be honest with them and to share the full range of options with them."
The report, "Quality of End-of-Life Cancer Care for Medicare Beneficiaries: Regional and Hospital-Specific Analyses," is the first to focus on cancer care at the end of life for the Medicare population in particular. The report was produced by the Dartmouth Atlas Project, which has been documenting the use of medical resources in the United States for more than 2 decades.
Researchers reviewed data from 235,821 Medicare patients (aged 65 years and older) with metastatic or aggressive cancer who died in 2003-2007. They identified regional trends as well as information about specific academic medical centers.
Nationwide, approximately 61% of cancer patients were hospitalized at least once during their last month of life. Hospital admissions varied from approximately 70% of cancer patients in Detroit to 46% of cancer patients in San Angelo, Tex.
The researchers identified the highest rates of hospitalization during the last month of life (more than 75%) among cancer patients at two hospitals in Detroit: St. John Hospital and Medical Center and Sinai-Grace Hospital. The lowest rate of hospitalization during the last month of life was 45% of cancer patients at the University of Washington Medical Center in Seattle.
Chemotherapy was part of care during the last 2 weeks of life in approximately 6% of patients nationwide, but the rate exceeded 10% in eight areas, including Olympia, Wash. (12%), and San Antonio (11%).
The researchers also found that fewer than 50% of cancer patients with a poor prognosis received hospice care, and some centers provided hospice care only in the last few days of life.
The degree of variation in the findings is too great to be explained by racial or ethnic preferences for aggressive end-of-life care, and adjustment for socioeconomic status does not change the results, said Dr. Goodman said.
But the data show that investment in beds, ICU facilities, and palliative care services is quite different from one health care system to another, he said.
The report highlights the need for doctors and health systems "to take a look at themselves and examine where they need to make their investments," said Dr. Goodman, who added that clinicians "need to look at ourselves in terms of how we communicate with patients" about cancer care options.
In a commentary accompanying the report, Dr. Joan Teno of Brown University in Providence, R.I., emphasized the need for health care providers to educate patients about their prognoses and to provide hospice options.
"If opportunities to improve are identified, hospitals should work with the local hospice or existing palliative care consult service, and/or start a palliative care consult service," to ensure that the institution is delivering high-quality care, Dr. Teno wrote.
Conversations about end-of-life care need to be started early in the course of treatment, Dr. Goodman said. "Our preferences can certainly change, but opening up the conversation early allows the conversation to evolve over time. The tragedy is when those conversations start near the end."
In a statement, Dr. George W. Sledge Jr., president of the American Society of Clinical Oncology said, "While saving lives is the oncologist’s goal, helping individuals live their final days in comfort and dignity is one of the most valuable and important responsibilities of our profession.
"Options to enhance a patient’s quality of life should be discussed throughout the course of their treatment. It is critical that we increase medical training in end-of-life patient communication and planning, and that Medicare and other insurers cover the costs of these essential conversations," Dr. Sledge noted.
The study was supported in part by the Robert Wood Johnson Foundation.
Approximately 29% of Medicare patients with cancer died in a hospital, and hospice care was inconsistent among medical centers in the United States, according to a report published Nov. 16 by the Dartmouth Institute for Health Policy & Clinical Practice.
The rate of hospital deaths varied widely by region, ranging from 47% in Manhattan (New York City) to 7% in Mason City, Iowa.
"The bottom line is that the care patients receive has less to do with what they want and more to do with the hospital they seek care from," Dr. David Goodman of Dartmouth University, Hanover, N.H., said in a press briefing. "We as physicians often make assumptions about what patients and families want for their lives. What patients really want is for a physician to be honest with them and to share the full range of options with them."
The report, "Quality of End-of-Life Cancer Care for Medicare Beneficiaries: Regional and Hospital-Specific Analyses," is the first to focus on cancer care at the end of life for the Medicare population in particular. The report was produced by the Dartmouth Atlas Project, which has been documenting the use of medical resources in the United States for more than 2 decades.
Researchers reviewed data from 235,821 Medicare patients (aged 65 years and older) with metastatic or aggressive cancer who died in 2003-2007. They identified regional trends as well as information about specific academic medical centers.
Nationwide, approximately 61% of cancer patients were hospitalized at least once during their last month of life. Hospital admissions varied from approximately 70% of cancer patients in Detroit to 46% of cancer patients in San Angelo, Tex.
The researchers identified the highest rates of hospitalization during the last month of life (more than 75%) among cancer patients at two hospitals in Detroit: St. John Hospital and Medical Center and Sinai-Grace Hospital. The lowest rate of hospitalization during the last month of life was 45% of cancer patients at the University of Washington Medical Center in Seattle.
Chemotherapy was part of care during the last 2 weeks of life in approximately 6% of patients nationwide, but the rate exceeded 10% in eight areas, including Olympia, Wash. (12%), and San Antonio (11%).
The researchers also found that fewer than 50% of cancer patients with a poor prognosis received hospice care, and some centers provided hospice care only in the last few days of life.
The degree of variation in the findings is too great to be explained by racial or ethnic preferences for aggressive end-of-life care, and adjustment for socioeconomic status does not change the results, said Dr. Goodman said.
But the data show that investment in beds, ICU facilities, and palliative care services is quite different from one health care system to another, he said.
The report highlights the need for doctors and health systems "to take a look at themselves and examine where they need to make their investments," said Dr. Goodman, who added that clinicians "need to look at ourselves in terms of how we communicate with patients" about cancer care options.
In a commentary accompanying the report, Dr. Joan Teno of Brown University in Providence, R.I., emphasized the need for health care providers to educate patients about their prognoses and to provide hospice options.
"If opportunities to improve are identified, hospitals should work with the local hospice or existing palliative care consult service, and/or start a palliative care consult service," to ensure that the institution is delivering high-quality care, Dr. Teno wrote.
Conversations about end-of-life care need to be started early in the course of treatment, Dr. Goodman said. "Our preferences can certainly change, but opening up the conversation early allows the conversation to evolve over time. The tragedy is when those conversations start near the end."
In a statement, Dr. George W. Sledge Jr., president of the American Society of Clinical Oncology said, "While saving lives is the oncologist’s goal, helping individuals live their final days in comfort and dignity is one of the most valuable and important responsibilities of our profession.
"Options to enhance a patient’s quality of life should be discussed throughout the course of their treatment. It is critical that we increase medical training in end-of-life patient communication and planning, and that Medicare and other insurers cover the costs of these essential conversations," Dr. Sledge noted.
The study was supported in part by the Robert Wood Johnson Foundation.
Wish Statements Help Doctors Deliver Painful News to Patients
CHICAGO - Oncologists can navigate the sensitive and arduous communication of painful news to patients with greater honesty and compassion by expressing wishes in response to their patients’ sense of loss, futility, or unrealistic hope, Dr. Timothy E. Quill, told attendees at the annual Chicago Supportive Oncology Conference.
Communicating with statements such as "I wish there was more we could do for you," "I wish I had better news to give you," "I wish things had turned out better," or "I wish our palliative care was more effective" communicates empathy and caring without distorting the truth, fostering a false sense of hope, or emotionally bludgeoning the patient, said Dr. Quill, director of the center for ethics, humanities, and palliative care at the University of Rochester (N.Y.).
Wishes are effective because they acknowledge the emotional impact of the patient’s illness and allow the physician to respond as a human being to the patient’s emotional distress, while communicating with gentleness and compassion that the hoped-for outcome is unlikely, explained Dr. Quill, a professor of medicine, psychiatry, and medical humanities at the university. Simultaneously – and paradoxically – wishes enable the physician to identify with the patient’s hope while casting that hope in a realistic light.
Dr. Quill stressed, however, that wish statements should be used only as the beginning of an important dialogue. Wishes acknowledge that a desired result probably will not happen, and can elicit anger, hopelessness, terror, and despair. It is essential to talk with patients about their feelings and experiences, he said. The failure to do so could cause emotional harm.
Questions following wish statements – such as "What has been the hardest part for you?" "What are your biggest concerns?" and "How is your family managing this?" – can help to guide meaningful communication. Also critical are statements that validate the patient’s experience ("Anyone going through this would have such feelings") and express empathy ("I can imagine that must have been frightening"), as well as efforts to facilitate opportunities for growth and closure in addressing short-term medical decisions, financial and legal affairs, unfinished personal and family business, and, possibly, spiritual, religious, and existential issues.
He emphasized that this communication must be accompanied by the search for new treatment direction and hope through the appropriate use of standard treatment options, experimental treatment, palliative care, or hospice care.
In responding to an expression of loss, grief, and hopelessness ("I do not see how I can go on"), Dr. Quill noted that the clinician does not have to have all the answers. A statement such as "I wish I had a simple formula" allows the physician to take the same emotional stance as the patient, whereas expressions of empathy ("Tell me about the scariest part," "We can work on this together") can help patients deal with intense emotions, such as fear of abandonment.
In responding to a patient’s expression of unrealistic hope ("I want to live to see the birth of my grandchild"), Dr. Quill suggested that responding with a wish ("I wish I could promise that") and statements of empathy ("It must be hard to contemplate missing that; in case you cannot, perhaps you could ...") can provide support without creating unrealistic expectations.
Dr. Quill had no disclosures. The conference was sponsored by Elsevier Oncology, a company that is affiliated with this news organization.
CHICAGO - Oncologists can navigate the sensitive and arduous communication of painful news to patients with greater honesty and compassion by expressing wishes in response to their patients’ sense of loss, futility, or unrealistic hope, Dr. Timothy E. Quill, told attendees at the annual Chicago Supportive Oncology Conference.
Communicating with statements such as "I wish there was more we could do for you," "I wish I had better news to give you," "I wish things had turned out better," or "I wish our palliative care was more effective" communicates empathy and caring without distorting the truth, fostering a false sense of hope, or emotionally bludgeoning the patient, said Dr. Quill, director of the center for ethics, humanities, and palliative care at the University of Rochester (N.Y.).
Wishes are effective because they acknowledge the emotional impact of the patient’s illness and allow the physician to respond as a human being to the patient’s emotional distress, while communicating with gentleness and compassion that the hoped-for outcome is unlikely, explained Dr. Quill, a professor of medicine, psychiatry, and medical humanities at the university. Simultaneously – and paradoxically – wishes enable the physician to identify with the patient’s hope while casting that hope in a realistic light.
Dr. Quill stressed, however, that wish statements should be used only as the beginning of an important dialogue. Wishes acknowledge that a desired result probably will not happen, and can elicit anger, hopelessness, terror, and despair. It is essential to talk with patients about their feelings and experiences, he said. The failure to do so could cause emotional harm.
Questions following wish statements – such as "What has been the hardest part for you?" "What are your biggest concerns?" and "How is your family managing this?" – can help to guide meaningful communication. Also critical are statements that validate the patient’s experience ("Anyone going through this would have such feelings") and express empathy ("I can imagine that must have been frightening"), as well as efforts to facilitate opportunities for growth and closure in addressing short-term medical decisions, financial and legal affairs, unfinished personal and family business, and, possibly, spiritual, religious, and existential issues.
He emphasized that this communication must be accompanied by the search for new treatment direction and hope through the appropriate use of standard treatment options, experimental treatment, palliative care, or hospice care.
In responding to an expression of loss, grief, and hopelessness ("I do not see how I can go on"), Dr. Quill noted that the clinician does not have to have all the answers. A statement such as "I wish I had a simple formula" allows the physician to take the same emotional stance as the patient, whereas expressions of empathy ("Tell me about the scariest part," "We can work on this together") can help patients deal with intense emotions, such as fear of abandonment.
In responding to a patient’s expression of unrealistic hope ("I want to live to see the birth of my grandchild"), Dr. Quill suggested that responding with a wish ("I wish I could promise that") and statements of empathy ("It must be hard to contemplate missing that; in case you cannot, perhaps you could ...") can provide support without creating unrealistic expectations.
Dr. Quill had no disclosures. The conference was sponsored by Elsevier Oncology, a company that is affiliated with this news organization.
CHICAGO - Oncologists can navigate the sensitive and arduous communication of painful news to patients with greater honesty and compassion by expressing wishes in response to their patients’ sense of loss, futility, or unrealistic hope, Dr. Timothy E. Quill, told attendees at the annual Chicago Supportive Oncology Conference.
Communicating with statements such as "I wish there was more we could do for you," "I wish I had better news to give you," "I wish things had turned out better," or "I wish our palliative care was more effective" communicates empathy and caring without distorting the truth, fostering a false sense of hope, or emotionally bludgeoning the patient, said Dr. Quill, director of the center for ethics, humanities, and palliative care at the University of Rochester (N.Y.).
Wishes are effective because they acknowledge the emotional impact of the patient’s illness and allow the physician to respond as a human being to the patient’s emotional distress, while communicating with gentleness and compassion that the hoped-for outcome is unlikely, explained Dr. Quill, a professor of medicine, psychiatry, and medical humanities at the university. Simultaneously – and paradoxically – wishes enable the physician to identify with the patient’s hope while casting that hope in a realistic light.
Dr. Quill stressed, however, that wish statements should be used only as the beginning of an important dialogue. Wishes acknowledge that a desired result probably will not happen, and can elicit anger, hopelessness, terror, and despair. It is essential to talk with patients about their feelings and experiences, he said. The failure to do so could cause emotional harm.
Questions following wish statements – such as "What has been the hardest part for you?" "What are your biggest concerns?" and "How is your family managing this?" – can help to guide meaningful communication. Also critical are statements that validate the patient’s experience ("Anyone going through this would have such feelings") and express empathy ("I can imagine that must have been frightening"), as well as efforts to facilitate opportunities for growth and closure in addressing short-term medical decisions, financial and legal affairs, unfinished personal and family business, and, possibly, spiritual, religious, and existential issues.
He emphasized that this communication must be accompanied by the search for new treatment direction and hope through the appropriate use of standard treatment options, experimental treatment, palliative care, or hospice care.
In responding to an expression of loss, grief, and hopelessness ("I do not see how I can go on"), Dr. Quill noted that the clinician does not have to have all the answers. A statement such as "I wish I had a simple formula" allows the physician to take the same emotional stance as the patient, whereas expressions of empathy ("Tell me about the scariest part," "We can work on this together") can help patients deal with intense emotions, such as fear of abandonment.
In responding to a patient’s expression of unrealistic hope ("I want to live to see the birth of my grandchild"), Dr. Quill suggested that responding with a wish ("I wish I could promise that") and statements of empathy ("It must be hard to contemplate missing that; in case you cannot, perhaps you could ...") can provide support without creating unrealistic expectations.
Dr. Quill had no disclosures. The conference was sponsored by Elsevier Oncology, a company that is affiliated with this news organization.
Malpractice Chronicle
Reprinted with permission from Medical Malpractice Verdicts, Settlements and Experts, Lewis Laska, Editor, (800) 298-6288.
Hemorrhoid or Cancerous Mass?
In May 2000, a 35-year-old woman gave birth to her second child at a Massachusetts hospital. She sustained a second-degree vaginal tear. During repair of the tear, a large hemorrhoid was visualized by the physician, who instructed a nurse-midwife to have the hemorrhoid evaluated, with a possible gastroenterology consult to rule out a mass.
The next day, the patient was examined by another doctor and another nurse-midwife. It was agreed by the clinicians and the patient that she would defer a gastroenterology consult and follow up with her primary care provider in a few weeks. When she saw her primary care physician three weeks after the delivery, her examination was negative for hemorrhoids, and the patient was instructed to call back if she had a recurrence. Since she experienced no recurrence, she did not follow up with the primary care provider. Over the next four years, the woman received medical care from her gynecologist but at no time underwent a rectal examination.
In February 2005, the plaintiff went to her primary care physician with complaints of rectal bleeding during bowel movements. No external hemorrhoids were found, but a rectal mass was present.
The woman was referred for a gastroenterology consult and biopsy, through which an intramucosal adenocarcinoma was identified. Chest CT revealed a nodule in the patient’s right lower lung lobe, which was suspicious for metastasis. Abdominal CT and positron emission tomography showed likely liver metastases. A liver biopsy performed in mid-March 2005 confirmed adenocarcinoma of the liver.
The patient received chemotherapy and chemoradiation. In September 2005, she underwent an abdominal perineal resection, left lateral segmentectomy of the liver, cholecystectomy, and appendectomy. By the time of settlement, she was doing well and was no longer receiving treatment for cancer.
The plaintiff claimed that her primary care provider should have followed up on the initial rectal finding, which would have led to an earlier diagnosis and treatment of her cancer.
The defendant argued that the lesion noted at the time of the delivery was a simple hemorrhoid, which resolved after delivery. The defendant also contended that the absence of any symptoms for nearly five years indicated that the cancer could not have been present in 2000. The defendant further claimed that the cancer found at diagnosis and the hemorrhoid that was originally noted were in different locations.
A $1 million settlement was reached.
Diagnosis, Treatment Delayed by Suspicion of Abuse
A seven-week-old boy was taken to the pediatrician by his parents. They said he had been crying inconsolably all day, with decreased food intake, limited urinary output, and bruising. The pediatrician suspected meningitis and sent them to the emergency department (ED) at a children’s hospital in Georgia. The intake staff and emergency physician, Dr. C., were informed that the pediatrician suspected meningitis.
A blood culture, chest x-ray, and ultrasound were performed. The infant’s white blood cell count was normal, and his condition improved during the ED stay. He was afebrile, and the defendants maintained that he had no symptoms that would indicate meningitis.
Bruising was found on the child’s rib cage and one knee, and an ultrasound indicated that the bruising was due to trauma; thus, a mandatory report was filed with the authorities, and the physician made a diagnosis of abuse. The parents were forced to leave the child at the hospital and were told not to return because the child was in the hospital’s custody. The child was transferred out of the ED and the care of Dr. C.
Subsequently, the blood culture was reported as positive and showed gram-positive cocci. This report was returned about 26 hours after the child’s symptoms had begun at home. The report on the blood culture was relayed to one of the doctors, but no orders were given to evaluate the child. The child was not given antibiotics to treat the meningitis until the following day. The police dropped the report of abuse shortly after the investigation into the allegations began.
The child experienced a severe seizure with catastrophic brain damage, which the plaintiffs attributed to a delay in diagnosis and treatment for meningitis. The child was unable to roll over by age 2 years and would require extensive care and treatment for the remainder of his life.
The defendants claimed that the actions taken were proper and that there was no reason to suspect infection because the child did not have a fever and his condition improved while he was in the ED.
According to a published report, a defense verdict was returned.
Failure to Recognize Adrenal Crisis
A 26-year-old Massachusetts woman had a history of type 2 diabetes and newly diagnosed adrenal insufficiency, for which she was taking hydrocortisone. She presented to an emergency department (ED) complaining of cold symptoms and abdominal pain.
She was evaluated by an emergency physician, who noted moderate pain and tachycardia, a heart rate of 100 beats/min, and tenderness in the woman’s sinuses, neck, and left lower quadrant of the abdomen. Laboratory test results included a positive pregnancy test and an abnormal potassium level. Subsequent cardiac monitoring revealed atrial tachycardia. ECG revealed a rapid heart rate and diffuse, nonspecific abnormalities.
A second physician assumed care of the patient and treated her with potassium chloride, IV fluids of dextrose and sodium chloride, and ondansetron for nausea and vomiting. Neither physician had noted the medications being taken by the patient.
She returned to the ED two days after her initial admission with complaints of vomiting, chest pain, and abdominal pain. Her vital signs were abnormal. A different emergency physician evaluated the patient and noted tachycardia and vitiligo (consistent with adrenal insufficiency). Laboratory study results revealed metabolic abnormalities, and the woman was diagnosed with pregnancy-related nausea and vomiting and poorly controlled diabetes.
Over the following week, five additional physicians examined and/or treated the woman, one of whom was told that she had been taking hydrocortisone but stopped when she learned she was pregnant. The woman’s condition worsened, involving blindness, severe metabolic acidosis, and respiratory arrest. The family agreed to a transfer to a tertiary care facility.
At this time, during conversations between the patient’s family and her physicians, the physicians were made aware that she was taking hydrocortisone for adrenal insufficiency. The physicians immediately began treatment with methylprednisolone. The patient’s neurologic status continued to decline, however, and CT revealed findings consistent with a diffuse anoxic injury. She was placed on comfort measures only, and she died nine days after her original ED admission.
The plaintiffs alleged negligence in the ED staff’s failure to take a thorough medical history. The defendant claimed that the decedent’s symptoms were not consistent with adrenal crisis and that she had not fully disclosed her use of hydrocortisone.
According to a published account, a $3 million settlement was reached.
High-Dose Morphine After DNR Order
At age 79, a woman with chronic obstructive pulmonary disease was admitted to a hospital in Georgia with breathing difficulties. During her hospitalization, the patient experienced respiratory arrest. A code was called and the defendant, the critical care pulmonologist on duty, responded.
Once bag ventilation was implemented, the patient started to breathe and the code was stopped. After the incident, the attending physician, who had also responded to the code, initiated a discussion with the patient’s daughter about the plan of treatment and the patient’s prognosis. At the conclusion of this conversation, the patient’s daughter agreed to a “do-not-resuscitate” order. The attending physician ordered 2 mg morphine as needed to keep the patient comfortable.
Five minutes later, the pulmonologist overrode this order and ordered 20 mg morphine pushed. Shortly after the medication was administered, the patient, who was talking to her daughter and granddaughter, lost consciousness. She died about three hours later without regaining consciousness.
The plaintiff claimed that the decedent’s condition improved during her hospitalization until the night before her arrest, when she was not given her scheduled breathing treatments. The plaintiff also alleged that the defendant pulmonologist was negligent in ordering the 20-mg dose of morphine and that the hospital nurse was negligent in administering such a high dose.
The defendants claimed that no negligence occurred and that the woman would have died sooner than three hours after the morphine was administered, if that indeed was the cause of her death.
According to a published account, a $3 million verdict was returned.
Reprinted with permission from Medical Malpractice Verdicts, Settlements and Experts, Lewis Laska, Editor, (800) 298-6288.
Hemorrhoid or Cancerous Mass?
In May 2000, a 35-year-old woman gave birth to her second child at a Massachusetts hospital. She sustained a second-degree vaginal tear. During repair of the tear, a large hemorrhoid was visualized by the physician, who instructed a nurse-midwife to have the hemorrhoid evaluated, with a possible gastroenterology consult to rule out a mass.
The next day, the patient was examined by another doctor and another nurse-midwife. It was agreed by the clinicians and the patient that she would defer a gastroenterology consult and follow up with her primary care provider in a few weeks. When she saw her primary care physician three weeks after the delivery, her examination was negative for hemorrhoids, and the patient was instructed to call back if she had a recurrence. Since she experienced no recurrence, she did not follow up with the primary care provider. Over the next four years, the woman received medical care from her gynecologist but at no time underwent a rectal examination.
In February 2005, the plaintiff went to her primary care physician with complaints of rectal bleeding during bowel movements. No external hemorrhoids were found, but a rectal mass was present.
The woman was referred for a gastroenterology consult and biopsy, through which an intramucosal adenocarcinoma was identified. Chest CT revealed a nodule in the patient’s right lower lung lobe, which was suspicious for metastasis. Abdominal CT and positron emission tomography showed likely liver metastases. A liver biopsy performed in mid-March 2005 confirmed adenocarcinoma of the liver.
The patient received chemotherapy and chemoradiation. In September 2005, she underwent an abdominal perineal resection, left lateral segmentectomy of the liver, cholecystectomy, and appendectomy. By the time of settlement, she was doing well and was no longer receiving treatment for cancer.
The plaintiff claimed that her primary care provider should have followed up on the initial rectal finding, which would have led to an earlier diagnosis and treatment of her cancer.
The defendant argued that the lesion noted at the time of the delivery was a simple hemorrhoid, which resolved after delivery. The defendant also contended that the absence of any symptoms for nearly five years indicated that the cancer could not have been present in 2000. The defendant further claimed that the cancer found at diagnosis and the hemorrhoid that was originally noted were in different locations.
A $1 million settlement was reached.
Diagnosis, Treatment Delayed by Suspicion of Abuse
A seven-week-old boy was taken to the pediatrician by his parents. They said he had been crying inconsolably all day, with decreased food intake, limited urinary output, and bruising. The pediatrician suspected meningitis and sent them to the emergency department (ED) at a children’s hospital in Georgia. The intake staff and emergency physician, Dr. C., were informed that the pediatrician suspected meningitis.
A blood culture, chest x-ray, and ultrasound were performed. The infant’s white blood cell count was normal, and his condition improved during the ED stay. He was afebrile, and the defendants maintained that he had no symptoms that would indicate meningitis.
Bruising was found on the child’s rib cage and one knee, and an ultrasound indicated that the bruising was due to trauma; thus, a mandatory report was filed with the authorities, and the physician made a diagnosis of abuse. The parents were forced to leave the child at the hospital and were told not to return because the child was in the hospital’s custody. The child was transferred out of the ED and the care of Dr. C.
Subsequently, the blood culture was reported as positive and showed gram-positive cocci. This report was returned about 26 hours after the child’s symptoms had begun at home. The report on the blood culture was relayed to one of the doctors, but no orders were given to evaluate the child. The child was not given antibiotics to treat the meningitis until the following day. The police dropped the report of abuse shortly after the investigation into the allegations began.
The child experienced a severe seizure with catastrophic brain damage, which the plaintiffs attributed to a delay in diagnosis and treatment for meningitis. The child was unable to roll over by age 2 years and would require extensive care and treatment for the remainder of his life.
The defendants claimed that the actions taken were proper and that there was no reason to suspect infection because the child did not have a fever and his condition improved while he was in the ED.
According to a published report, a defense verdict was returned.
Failure to Recognize Adrenal Crisis
A 26-year-old Massachusetts woman had a history of type 2 diabetes and newly diagnosed adrenal insufficiency, for which she was taking hydrocortisone. She presented to an emergency department (ED) complaining of cold symptoms and abdominal pain.
She was evaluated by an emergency physician, who noted moderate pain and tachycardia, a heart rate of 100 beats/min, and tenderness in the woman’s sinuses, neck, and left lower quadrant of the abdomen. Laboratory test results included a positive pregnancy test and an abnormal potassium level. Subsequent cardiac monitoring revealed atrial tachycardia. ECG revealed a rapid heart rate and diffuse, nonspecific abnormalities.
A second physician assumed care of the patient and treated her with potassium chloride, IV fluids of dextrose and sodium chloride, and ondansetron for nausea and vomiting. Neither physician had noted the medications being taken by the patient.
She returned to the ED two days after her initial admission with complaints of vomiting, chest pain, and abdominal pain. Her vital signs were abnormal. A different emergency physician evaluated the patient and noted tachycardia and vitiligo (consistent with adrenal insufficiency). Laboratory study results revealed metabolic abnormalities, and the woman was diagnosed with pregnancy-related nausea and vomiting and poorly controlled diabetes.
Over the following week, five additional physicians examined and/or treated the woman, one of whom was told that she had been taking hydrocortisone but stopped when she learned she was pregnant. The woman’s condition worsened, involving blindness, severe metabolic acidosis, and respiratory arrest. The family agreed to a transfer to a tertiary care facility.
At this time, during conversations between the patient’s family and her physicians, the physicians were made aware that she was taking hydrocortisone for adrenal insufficiency. The physicians immediately began treatment with methylprednisolone. The patient’s neurologic status continued to decline, however, and CT revealed findings consistent with a diffuse anoxic injury. She was placed on comfort measures only, and she died nine days after her original ED admission.
The plaintiffs alleged negligence in the ED staff’s failure to take a thorough medical history. The defendant claimed that the decedent’s symptoms were not consistent with adrenal crisis and that she had not fully disclosed her use of hydrocortisone.
According to a published account, a $3 million settlement was reached.
High-Dose Morphine After DNR Order
At age 79, a woman with chronic obstructive pulmonary disease was admitted to a hospital in Georgia with breathing difficulties. During her hospitalization, the patient experienced respiratory arrest. A code was called and the defendant, the critical care pulmonologist on duty, responded.
Once bag ventilation was implemented, the patient started to breathe and the code was stopped. After the incident, the attending physician, who had also responded to the code, initiated a discussion with the patient’s daughter about the plan of treatment and the patient’s prognosis. At the conclusion of this conversation, the patient’s daughter agreed to a “do-not-resuscitate” order. The attending physician ordered 2 mg morphine as needed to keep the patient comfortable.
Five minutes later, the pulmonologist overrode this order and ordered 20 mg morphine pushed. Shortly after the medication was administered, the patient, who was talking to her daughter and granddaughter, lost consciousness. She died about three hours later without regaining consciousness.
The plaintiff claimed that the decedent’s condition improved during her hospitalization until the night before her arrest, when she was not given her scheduled breathing treatments. The plaintiff also alleged that the defendant pulmonologist was negligent in ordering the 20-mg dose of morphine and that the hospital nurse was negligent in administering such a high dose.
The defendants claimed that no negligence occurred and that the woman would have died sooner than three hours after the morphine was administered, if that indeed was the cause of her death.
According to a published account, a $3 million verdict was returned.
Reprinted with permission from Medical Malpractice Verdicts, Settlements and Experts, Lewis Laska, Editor, (800) 298-6288.
Hemorrhoid or Cancerous Mass?
In May 2000, a 35-year-old woman gave birth to her second child at a Massachusetts hospital. She sustained a second-degree vaginal tear. During repair of the tear, a large hemorrhoid was visualized by the physician, who instructed a nurse-midwife to have the hemorrhoid evaluated, with a possible gastroenterology consult to rule out a mass.
The next day, the patient was examined by another doctor and another nurse-midwife. It was agreed by the clinicians and the patient that she would defer a gastroenterology consult and follow up with her primary care provider in a few weeks. When she saw her primary care physician three weeks after the delivery, her examination was negative for hemorrhoids, and the patient was instructed to call back if she had a recurrence. Since she experienced no recurrence, she did not follow up with the primary care provider. Over the next four years, the woman received medical care from her gynecologist but at no time underwent a rectal examination.
In February 2005, the plaintiff went to her primary care physician with complaints of rectal bleeding during bowel movements. No external hemorrhoids were found, but a rectal mass was present.
The woman was referred for a gastroenterology consult and biopsy, through which an intramucosal adenocarcinoma was identified. Chest CT revealed a nodule in the patient’s right lower lung lobe, which was suspicious for metastasis. Abdominal CT and positron emission tomography showed likely liver metastases. A liver biopsy performed in mid-March 2005 confirmed adenocarcinoma of the liver.
The patient received chemotherapy and chemoradiation. In September 2005, she underwent an abdominal perineal resection, left lateral segmentectomy of the liver, cholecystectomy, and appendectomy. By the time of settlement, she was doing well and was no longer receiving treatment for cancer.
The plaintiff claimed that her primary care provider should have followed up on the initial rectal finding, which would have led to an earlier diagnosis and treatment of her cancer.
The defendant argued that the lesion noted at the time of the delivery was a simple hemorrhoid, which resolved after delivery. The defendant also contended that the absence of any symptoms for nearly five years indicated that the cancer could not have been present in 2000. The defendant further claimed that the cancer found at diagnosis and the hemorrhoid that was originally noted were in different locations.
A $1 million settlement was reached.
Diagnosis, Treatment Delayed by Suspicion of Abuse
A seven-week-old boy was taken to the pediatrician by his parents. They said he had been crying inconsolably all day, with decreased food intake, limited urinary output, and bruising. The pediatrician suspected meningitis and sent them to the emergency department (ED) at a children’s hospital in Georgia. The intake staff and emergency physician, Dr. C., were informed that the pediatrician suspected meningitis.
A blood culture, chest x-ray, and ultrasound were performed. The infant’s white blood cell count was normal, and his condition improved during the ED stay. He was afebrile, and the defendants maintained that he had no symptoms that would indicate meningitis.
Bruising was found on the child’s rib cage and one knee, and an ultrasound indicated that the bruising was due to trauma; thus, a mandatory report was filed with the authorities, and the physician made a diagnosis of abuse. The parents were forced to leave the child at the hospital and were told not to return because the child was in the hospital’s custody. The child was transferred out of the ED and the care of Dr. C.
Subsequently, the blood culture was reported as positive and showed gram-positive cocci. This report was returned about 26 hours after the child’s symptoms had begun at home. The report on the blood culture was relayed to one of the doctors, but no orders were given to evaluate the child. The child was not given antibiotics to treat the meningitis until the following day. The police dropped the report of abuse shortly after the investigation into the allegations began.
The child experienced a severe seizure with catastrophic brain damage, which the plaintiffs attributed to a delay in diagnosis and treatment for meningitis. The child was unable to roll over by age 2 years and would require extensive care and treatment for the remainder of his life.
The defendants claimed that the actions taken were proper and that there was no reason to suspect infection because the child did not have a fever and his condition improved while he was in the ED.
According to a published report, a defense verdict was returned.
Failure to Recognize Adrenal Crisis
A 26-year-old Massachusetts woman had a history of type 2 diabetes and newly diagnosed adrenal insufficiency, for which she was taking hydrocortisone. She presented to an emergency department (ED) complaining of cold symptoms and abdominal pain.
She was evaluated by an emergency physician, who noted moderate pain and tachycardia, a heart rate of 100 beats/min, and tenderness in the woman’s sinuses, neck, and left lower quadrant of the abdomen. Laboratory test results included a positive pregnancy test and an abnormal potassium level. Subsequent cardiac monitoring revealed atrial tachycardia. ECG revealed a rapid heart rate and diffuse, nonspecific abnormalities.
A second physician assumed care of the patient and treated her with potassium chloride, IV fluids of dextrose and sodium chloride, and ondansetron for nausea and vomiting. Neither physician had noted the medications being taken by the patient.
She returned to the ED two days after her initial admission with complaints of vomiting, chest pain, and abdominal pain. Her vital signs were abnormal. A different emergency physician evaluated the patient and noted tachycardia and vitiligo (consistent with adrenal insufficiency). Laboratory study results revealed metabolic abnormalities, and the woman was diagnosed with pregnancy-related nausea and vomiting and poorly controlled diabetes.
Over the following week, five additional physicians examined and/or treated the woman, one of whom was told that she had been taking hydrocortisone but stopped when she learned she was pregnant. The woman’s condition worsened, involving blindness, severe metabolic acidosis, and respiratory arrest. The family agreed to a transfer to a tertiary care facility.
At this time, during conversations between the patient’s family and her physicians, the physicians were made aware that she was taking hydrocortisone for adrenal insufficiency. The physicians immediately began treatment with methylprednisolone. The patient’s neurologic status continued to decline, however, and CT revealed findings consistent with a diffuse anoxic injury. She was placed on comfort measures only, and she died nine days after her original ED admission.
The plaintiffs alleged negligence in the ED staff’s failure to take a thorough medical history. The defendant claimed that the decedent’s symptoms were not consistent with adrenal crisis and that she had not fully disclosed her use of hydrocortisone.
According to a published account, a $3 million settlement was reached.
High-Dose Morphine After DNR Order
At age 79, a woman with chronic obstructive pulmonary disease was admitted to a hospital in Georgia with breathing difficulties. During her hospitalization, the patient experienced respiratory arrest. A code was called and the defendant, the critical care pulmonologist on duty, responded.
Once bag ventilation was implemented, the patient started to breathe and the code was stopped. After the incident, the attending physician, who had also responded to the code, initiated a discussion with the patient’s daughter about the plan of treatment and the patient’s prognosis. At the conclusion of this conversation, the patient’s daughter agreed to a “do-not-resuscitate” order. The attending physician ordered 2 mg morphine as needed to keep the patient comfortable.
Five minutes later, the pulmonologist overrode this order and ordered 20 mg morphine pushed. Shortly after the medication was administered, the patient, who was talking to her daughter and granddaughter, lost consciousness. She died about three hours later without regaining consciousness.
The plaintiff claimed that the decedent’s condition improved during her hospitalization until the night before her arrest, when she was not given her scheduled breathing treatments. The plaintiff also alleged that the defendant pulmonologist was negligent in ordering the 20-mg dose of morphine and that the hospital nurse was negligent in administering such a high dose.
The defendants claimed that no negligence occurred and that the woman would have died sooner than three hours after the morphine was administered, if that indeed was the cause of her death.
According to a published account, a $3 million verdict was returned.
Many Elderly Terminally Ill Undergo Unnecessary 'Autopilot' Cancer Screening
A “meaningful proportion” of patients aged 65 years and older who have advanced cancer and a limited life expectancy undergo unnecessary screening for other cancers, needlessly exposing them to physical risk and psychological distress, according to a report in the Oct. 13 issue of JAMA.
Dr. Camelia S. Sima of the department of epidemiology and biostatistics at Memorial Sloan-Kettering Cancer Center, New York, and her associates said the terminally ill patients “have essentially no chance of benefiting from these procedures,” which they said were the result of a “culture of screening on ‘autopilot.’?”
They noted, however, that current guidelines “do not directly address the appropriateness of screening for individuals with terminal illnesses.”
The screening rates were one-third to one-half the rates in a matched control population of healthy adults who were followed over the same time period. Dr. Sima and her colleagues noted that rates of unnecessary cancer screening are likely to be even higher in the general population of patients with advanced cancer, which would include younger patients and those who are commercially insured (JAMA 2010;304:1584-91).
The investigators analyzed 1998-2005 data from the Surveillance, Epidemiology, and End Results (SEER) cancer registry, which is linked with Medicare claims. They identified 87,736 Medicare patients (median age, 77 years) who survived at least 2 months after a diagnosis of advanced lung, colorectal, breast, gastroesophageal, or pancreatic cancer, and were followed until death or 2008.
Although such cancers have a median survival of less than 2 years, 15% of the men had PSA testing. About 9% of the women underwent screening mammography and 6% underwent Pap testing. Two percent of men and women underwent lower GI endoscopy screening for colorectal cancer.
The data did not indicate whether this screening was ordered by oncologists or primary care physicians, nor whether it was driven by patient demand. But “whatever the impetus, screening utilization by patients with advanced cancer adds to the mounting concern about overdiagnosis,” the researchers said.
“The most plausible interpretation of our data is that efforts to foster adherence to screening have led to deeply ingrained habits. Patients and their health care practitioners accustomed to obtaining screening tests at regular intervals continue to do so even when the benefits have been rendered futile in the face of competing risk from advanced cancer,” Dr. Sima and her associates said.
As an example of this “autopilot” screening, they noted that 20% of the study population continued to undergo regular testing of cholesterol levels even though their life expectancy was drastically limited.
“Furthermore, we hypothesize that neither primary care physicians nor oncologists routinely engage in the difficult discussions that require explanation of why continuation of procedures to which patients have become accustomed is no longer necessary.
“There is substantial evidence that even when physicians recognize that life expectancy is limited, they do not consistently communicate prognosis, and patients may use denial as a coping strategy to face impending loss. Our findings represent one manifestation of this communication deficit.”
Any attempt to limit patient care “is routinely met with vocal opposition,” but this overuse of cancer screening “is likely to be relatively uncontroversial,” the investigators said.
“Each medical specialty needs to engage in thoughtful self-scrutiny to identify episodes of unnecessary care. We suggest that the road to a high-performing, high-value health care system will be paved with small stones such as the example we have identified.”
This study was supported in part by a grant from the National Cancer Institute. Dr. Sima and her colleagues did not report any financial disclosures.
A “meaningful proportion” of patients aged 65 years and older who have advanced cancer and a limited life expectancy undergo unnecessary screening for other cancers, needlessly exposing them to physical risk and psychological distress, according to a report in the Oct. 13 issue of JAMA.
Dr. Camelia S. Sima of the department of epidemiology and biostatistics at Memorial Sloan-Kettering Cancer Center, New York, and her associates said the terminally ill patients “have essentially no chance of benefiting from these procedures,” which they said were the result of a “culture of screening on ‘autopilot.’?”
They noted, however, that current guidelines “do not directly address the appropriateness of screening for individuals with terminal illnesses.”
The screening rates were one-third to one-half the rates in a matched control population of healthy adults who were followed over the same time period. Dr. Sima and her colleagues noted that rates of unnecessary cancer screening are likely to be even higher in the general population of patients with advanced cancer, which would include younger patients and those who are commercially insured (JAMA 2010;304:1584-91).
The investigators analyzed 1998-2005 data from the Surveillance, Epidemiology, and End Results (SEER) cancer registry, which is linked with Medicare claims. They identified 87,736 Medicare patients (median age, 77 years) who survived at least 2 months after a diagnosis of advanced lung, colorectal, breast, gastroesophageal, or pancreatic cancer, and were followed until death or 2008.
Although such cancers have a median survival of less than 2 years, 15% of the men had PSA testing. About 9% of the women underwent screening mammography and 6% underwent Pap testing. Two percent of men and women underwent lower GI endoscopy screening for colorectal cancer.
The data did not indicate whether this screening was ordered by oncologists or primary care physicians, nor whether it was driven by patient demand. But “whatever the impetus, screening utilization by patients with advanced cancer adds to the mounting concern about overdiagnosis,” the researchers said.
“The most plausible interpretation of our data is that efforts to foster adherence to screening have led to deeply ingrained habits. Patients and their health care practitioners accustomed to obtaining screening tests at regular intervals continue to do so even when the benefits have been rendered futile in the face of competing risk from advanced cancer,” Dr. Sima and her associates said.
As an example of this “autopilot” screening, they noted that 20% of the study population continued to undergo regular testing of cholesterol levels even though their life expectancy was drastically limited.
“Furthermore, we hypothesize that neither primary care physicians nor oncologists routinely engage in the difficult discussions that require explanation of why continuation of procedures to which patients have become accustomed is no longer necessary.
“There is substantial evidence that even when physicians recognize that life expectancy is limited, they do not consistently communicate prognosis, and patients may use denial as a coping strategy to face impending loss. Our findings represent one manifestation of this communication deficit.”
Any attempt to limit patient care “is routinely met with vocal opposition,” but this overuse of cancer screening “is likely to be relatively uncontroversial,” the investigators said.
“Each medical specialty needs to engage in thoughtful self-scrutiny to identify episodes of unnecessary care. We suggest that the road to a high-performing, high-value health care system will be paved with small stones such as the example we have identified.”
This study was supported in part by a grant from the National Cancer Institute. Dr. Sima and her colleagues did not report any financial disclosures.
A “meaningful proportion” of patients aged 65 years and older who have advanced cancer and a limited life expectancy undergo unnecessary screening for other cancers, needlessly exposing them to physical risk and psychological distress, according to a report in the Oct. 13 issue of JAMA.
Dr. Camelia S. Sima of the department of epidemiology and biostatistics at Memorial Sloan-Kettering Cancer Center, New York, and her associates said the terminally ill patients “have essentially no chance of benefiting from these procedures,” which they said were the result of a “culture of screening on ‘autopilot.’?”
They noted, however, that current guidelines “do not directly address the appropriateness of screening for individuals with terminal illnesses.”
The screening rates were one-third to one-half the rates in a matched control population of healthy adults who were followed over the same time period. Dr. Sima and her colleagues noted that rates of unnecessary cancer screening are likely to be even higher in the general population of patients with advanced cancer, which would include younger patients and those who are commercially insured (JAMA 2010;304:1584-91).
The investigators analyzed 1998-2005 data from the Surveillance, Epidemiology, and End Results (SEER) cancer registry, which is linked with Medicare claims. They identified 87,736 Medicare patients (median age, 77 years) who survived at least 2 months after a diagnosis of advanced lung, colorectal, breast, gastroesophageal, or pancreatic cancer, and were followed until death or 2008.
Although such cancers have a median survival of less than 2 years, 15% of the men had PSA testing. About 9% of the women underwent screening mammography and 6% underwent Pap testing. Two percent of men and women underwent lower GI endoscopy screening for colorectal cancer.
The data did not indicate whether this screening was ordered by oncologists or primary care physicians, nor whether it was driven by patient demand. But “whatever the impetus, screening utilization by patients with advanced cancer adds to the mounting concern about overdiagnosis,” the researchers said.
“The most plausible interpretation of our data is that efforts to foster adherence to screening have led to deeply ingrained habits. Patients and their health care practitioners accustomed to obtaining screening tests at regular intervals continue to do so even when the benefits have been rendered futile in the face of competing risk from advanced cancer,” Dr. Sima and her associates said.
As an example of this “autopilot” screening, they noted that 20% of the study population continued to undergo regular testing of cholesterol levels even though their life expectancy was drastically limited.
“Furthermore, we hypothesize that neither primary care physicians nor oncologists routinely engage in the difficult discussions that require explanation of why continuation of procedures to which patients have become accustomed is no longer necessary.
“There is substantial evidence that even when physicians recognize that life expectancy is limited, they do not consistently communicate prognosis, and patients may use denial as a coping strategy to face impending loss. Our findings represent one manifestation of this communication deficit.”
Any attempt to limit patient care “is routinely met with vocal opposition,” but this overuse of cancer screening “is likely to be relatively uncontroversial,” the investigators said.
“Each medical specialty needs to engage in thoughtful self-scrutiny to identify episodes of unnecessary care. We suggest that the road to a high-performing, high-value health care system will be paved with small stones such as the example we have identified.”
This study was supported in part by a grant from the National Cancer Institute. Dr. Sima and her colleagues did not report any financial disclosures.
ONLINE EXCLUSIVE: HM is a perfect fit for a palliative care service
John Harney, COO at University of Colorado Hospital, moved west in 2008 after working at New York University Hospitals Center. The East Coast hospital had used a grant to establish a palliative-care program and witnessed immediate results.
“We truly believed it resulted in reductions in length of stay, as well as humanistic benefits,” Harney says. “When I came out to Colorado, I was pleasantly surprised at the breadth and depth of the programs here.”
Harney says he believes HM is a logical place to advance palliative care to the next level, as most HM groups already possess an in-house presence and commitment to efficient throughput. Hospital administrators will be concerned with consistency, routines, and protocols, he says, as well as the palliative-care service’s commitment to quality improvement. Those same administrators appreciate the need for program and salary support, although he advises palliative-care advocates to do their homework and develop a viable business plan.
“Hospital administrators will quickly figure out the math,” Harney says. “If you’re coming to speak to us, you need to have your numbers in order. You also need some monitoring in place.”
The initial salvo should include confirmation that HM group leaders have done their homework: surveyed their HM staff and discussed the idea with oncologists and other specialists. “It’s also helpful to have real champions in nursing and social work,” Harney says. “It’s never easy to get financial support for a new program, but if you have those ducks lined up, it goes better.”
John Harney, COO at University of Colorado Hospital, moved west in 2008 after working at New York University Hospitals Center. The East Coast hospital had used a grant to establish a palliative-care program and witnessed immediate results.
“We truly believed it resulted in reductions in length of stay, as well as humanistic benefits,” Harney says. “When I came out to Colorado, I was pleasantly surprised at the breadth and depth of the programs here.”
Harney says he believes HM is a logical place to advance palliative care to the next level, as most HM groups already possess an in-house presence and commitment to efficient throughput. Hospital administrators will be concerned with consistency, routines, and protocols, he says, as well as the palliative-care service’s commitment to quality improvement. Those same administrators appreciate the need for program and salary support, although he advises palliative-care advocates to do their homework and develop a viable business plan.
“Hospital administrators will quickly figure out the math,” Harney says. “If you’re coming to speak to us, you need to have your numbers in order. You also need some monitoring in place.”
The initial salvo should include confirmation that HM group leaders have done their homework: surveyed their HM staff and discussed the idea with oncologists and other specialists. “It’s also helpful to have real champions in nursing and social work,” Harney says. “It’s never easy to get financial support for a new program, but if you have those ducks lined up, it goes better.”
John Harney, COO at University of Colorado Hospital, moved west in 2008 after working at New York University Hospitals Center. The East Coast hospital had used a grant to establish a palliative-care program and witnessed immediate results.
“We truly believed it resulted in reductions in length of stay, as well as humanistic benefits,” Harney says. “When I came out to Colorado, I was pleasantly surprised at the breadth and depth of the programs here.”
Harney says he believes HM is a logical place to advance palliative care to the next level, as most HM groups already possess an in-house presence and commitment to efficient throughput. Hospital administrators will be concerned with consistency, routines, and protocols, he says, as well as the palliative-care service’s commitment to quality improvement. Those same administrators appreciate the need for program and salary support, although he advises palliative-care advocates to do their homework and develop a viable business plan.
“Hospital administrators will quickly figure out the math,” Harney says. “If you’re coming to speak to us, you need to have your numbers in order. You also need some monitoring in place.”
The initial salvo should include confirmation that HM group leaders have done their homework: surveyed their HM staff and discussed the idea with oncologists and other specialists. “It’s also helpful to have real champions in nursing and social work,” Harney says. “It’s never easy to get financial support for a new program, but if you have those ducks lined up, it goes better.”