User login
N.Y. Palliative Care Law May Not Change Practice
A new law requiring New York physicians to discuss palliative care and end-of-life options with terminally ill patients is well intentioned, but may not do much to change clinical practice or institutional culture, according to some observers in the state.
The New York Palliative Care Information Act was signed into law by Gov. David Paterson (D) in August. Perhaps as a sign that palliative care is being embraced more readily and becoming better understood, it took just 14 months from the bill’s introduction in the state Senate (S. 4498 and A. 7617) to its signing.
Even so, “whether or not it will change behavior is a bit of a black box,” said Dr. Bradley Flansbaum, director of hospitalist services at Lenox Hill Hospital in New York. “It’s a nice thought, but I don’t know how they’re going to put it into effect.”
Under the law, physicians and nurse practitioners are required to provide a patient who has less than 6 months to live with information and counseling on palliative care and end-of-life options, including, “the range of options appropriate to the patient, the prognosis, risks and benefits of the various options, and the patient’s legal rights to comprehensive pain and symptom management at the end of life.”
The physician or nurse practitioner can refer the patient to another provider who is willing to meet the legal statute or who is “professionally qualified” to offer the services.
There is no reimbursement offered for the required services.
Because it is an amendment to the state’s public health law, violations of the new law could result in penalties or fines. It’s not clear how it will be enforced or what might trigger the penalties; the health department has until the law’s effective date (February 2011) to devise regulations, said David Leven, executive director of Compassion and Choices of New York.
That advocacy group helped devise the proposal and then shepherded it though the legislature, said Mr. Leven. California has a similar statute, but is not as strong because it does not put the onus on physicians, he said.
The organization sought the legislation because even with increased training on end-of-life issues, too few physicians are having conversations with their dying patients, Mr. Leven said. That means patients’ wishes aren’t being respected, to the detriment of both patients and the practice of medicine.
The organization also hoped that the law would be a catalyst to improving end-of-life education in medical school and at the professional level, he said.
Dr. Wendy Edwards, director of the palliative medicine program at Lenox Hill, said that education would be a key component, but there appeared to be no such formal requirements in the law. About 15 years ago, she was part of a group that attempted to get a bill passed to mandate the teaching of palliative care in medical schools, but it did not get anywhere.
She said she wasn’t sure that the new law was the way to increase attention to palliative care, but that it had likely come about as a result of frustration and impatience on the part of palliative specialists.
The law will be positive, however, she said. Palliative care won’t just be the standard of care, but will be the law, which gives some backing to hospitals that seek to implement and strengthen their quality of care, and end-of-life care in particular.
But it still will not make it easier for physicians who do not have experience in palliative care, Dr. Edwards said. “It’s a very hard discussion to have; it’s not something doctors are trained to do.”
A recent study in non–small cell lung cancer patients found that those who were given palliative care at the time of diagnosis had a better quality of life than did those in standard care (N. Engl. J. Med. 2010;363:733-42). This study may do more to advance the field than does the New York law, Dr. Edwards noted.
Although the Hospice and Palliative Care Association of New York State supported the law, the Medical Society of the State of New York did not. The medical society, which represents 25,000 physicians, opposed the law because of concerns that it would interfere with the way each and every doctor navigates through end-of-life situations with each individual patient, said Elizabeth C. Dears, the society’s senior vice president for legislative and regulatory affairs.
Mandating that information be given on palliative care “may undermine the patient’s belief and conviction in prevailing against their disease and undercut the confidence in their treating physician,” said Ms. Dears.
The medical society also said that physicians are not licensed to provide legal advice in areas such as pain or symptom management, and that they may not know what they are supposed to be communicating to patients under certain provisions, while still being subject to penalties.
Although the medical society might object to requiring any such talk, both Dr. Flansbaum and Dr. Edwards said that, realistically, the law should be requiring palliative care to be offered sooner in the disease process and to a broader group of patients, such as those who have chronic life-limiting conditions such as heart failure.
“By the time you’re invoking palliative care in terminal patients, you’re behind the curve,” said Dr. Flansbaum.
A new law requiring New York physicians to discuss palliative care and end-of-life options with terminally ill patients is well intentioned, but may not do much to change clinical practice or institutional culture, according to some observers in the state.
The New York Palliative Care Information Act was signed into law by Gov. David Paterson (D) in August. Perhaps as a sign that palliative care is being embraced more readily and becoming better understood, it took just 14 months from the bill’s introduction in the state Senate (S. 4498 and A. 7617) to its signing.
Even so, “whether or not it will change behavior is a bit of a black box,” said Dr. Bradley Flansbaum, director of hospitalist services at Lenox Hill Hospital in New York. “It’s a nice thought, but I don’t know how they’re going to put it into effect.”
Under the law, physicians and nurse practitioners are required to provide a patient who has less than 6 months to live with information and counseling on palliative care and end-of-life options, including, “the range of options appropriate to the patient, the prognosis, risks and benefits of the various options, and the patient’s legal rights to comprehensive pain and symptom management at the end of life.”
The physician or nurse practitioner can refer the patient to another provider who is willing to meet the legal statute or who is “professionally qualified” to offer the services.
There is no reimbursement offered for the required services.
Because it is an amendment to the state’s public health law, violations of the new law could result in penalties or fines. It’s not clear how it will be enforced or what might trigger the penalties; the health department has until the law’s effective date (February 2011) to devise regulations, said David Leven, executive director of Compassion and Choices of New York.
That advocacy group helped devise the proposal and then shepherded it though the legislature, said Mr. Leven. California has a similar statute, but is not as strong because it does not put the onus on physicians, he said.
The organization sought the legislation because even with increased training on end-of-life issues, too few physicians are having conversations with their dying patients, Mr. Leven said. That means patients’ wishes aren’t being respected, to the detriment of both patients and the practice of medicine.
The organization also hoped that the law would be a catalyst to improving end-of-life education in medical school and at the professional level, he said.
Dr. Wendy Edwards, director of the palliative medicine program at Lenox Hill, said that education would be a key component, but there appeared to be no such formal requirements in the law. About 15 years ago, she was part of a group that attempted to get a bill passed to mandate the teaching of palliative care in medical schools, but it did not get anywhere.
She said she wasn’t sure that the new law was the way to increase attention to palliative care, but that it had likely come about as a result of frustration and impatience on the part of palliative specialists.
The law will be positive, however, she said. Palliative care won’t just be the standard of care, but will be the law, which gives some backing to hospitals that seek to implement and strengthen their quality of care, and end-of-life care in particular.
But it still will not make it easier for physicians who do not have experience in palliative care, Dr. Edwards said. “It’s a very hard discussion to have; it’s not something doctors are trained to do.”
A recent study in non–small cell lung cancer patients found that those who were given palliative care at the time of diagnosis had a better quality of life than did those in standard care (N. Engl. J. Med. 2010;363:733-42). This study may do more to advance the field than does the New York law, Dr. Edwards noted.
Although the Hospice and Palliative Care Association of New York State supported the law, the Medical Society of the State of New York did not. The medical society, which represents 25,000 physicians, opposed the law because of concerns that it would interfere with the way each and every doctor navigates through end-of-life situations with each individual patient, said Elizabeth C. Dears, the society’s senior vice president for legislative and regulatory affairs.
Mandating that information be given on palliative care “may undermine the patient’s belief and conviction in prevailing against their disease and undercut the confidence in their treating physician,” said Ms. Dears.
The medical society also said that physicians are not licensed to provide legal advice in areas such as pain or symptom management, and that they may not know what they are supposed to be communicating to patients under certain provisions, while still being subject to penalties.
Although the medical society might object to requiring any such talk, both Dr. Flansbaum and Dr. Edwards said that, realistically, the law should be requiring palliative care to be offered sooner in the disease process and to a broader group of patients, such as those who have chronic life-limiting conditions such as heart failure.
“By the time you’re invoking palliative care in terminal patients, you’re behind the curve,” said Dr. Flansbaum.
A new law requiring New York physicians to discuss palliative care and end-of-life options with terminally ill patients is well intentioned, but may not do much to change clinical practice or institutional culture, according to some observers in the state.
The New York Palliative Care Information Act was signed into law by Gov. David Paterson (D) in August. Perhaps as a sign that palliative care is being embraced more readily and becoming better understood, it took just 14 months from the bill’s introduction in the state Senate (S. 4498 and A. 7617) to its signing.
Even so, “whether or not it will change behavior is a bit of a black box,” said Dr. Bradley Flansbaum, director of hospitalist services at Lenox Hill Hospital in New York. “It’s a nice thought, but I don’t know how they’re going to put it into effect.”
Under the law, physicians and nurse practitioners are required to provide a patient who has less than 6 months to live with information and counseling on palliative care and end-of-life options, including, “the range of options appropriate to the patient, the prognosis, risks and benefits of the various options, and the patient’s legal rights to comprehensive pain and symptom management at the end of life.”
The physician or nurse practitioner can refer the patient to another provider who is willing to meet the legal statute or who is “professionally qualified” to offer the services.
There is no reimbursement offered for the required services.
Because it is an amendment to the state’s public health law, violations of the new law could result in penalties or fines. It’s not clear how it will be enforced or what might trigger the penalties; the health department has until the law’s effective date (February 2011) to devise regulations, said David Leven, executive director of Compassion and Choices of New York.
That advocacy group helped devise the proposal and then shepherded it though the legislature, said Mr. Leven. California has a similar statute, but is not as strong because it does not put the onus on physicians, he said.
The organization sought the legislation because even with increased training on end-of-life issues, too few physicians are having conversations with their dying patients, Mr. Leven said. That means patients’ wishes aren’t being respected, to the detriment of both patients and the practice of medicine.
The organization also hoped that the law would be a catalyst to improving end-of-life education in medical school and at the professional level, he said.
Dr. Wendy Edwards, director of the palliative medicine program at Lenox Hill, said that education would be a key component, but there appeared to be no such formal requirements in the law. About 15 years ago, she was part of a group that attempted to get a bill passed to mandate the teaching of palliative care in medical schools, but it did not get anywhere.
She said she wasn’t sure that the new law was the way to increase attention to palliative care, but that it had likely come about as a result of frustration and impatience on the part of palliative specialists.
The law will be positive, however, she said. Palliative care won’t just be the standard of care, but will be the law, which gives some backing to hospitals that seek to implement and strengthen their quality of care, and end-of-life care in particular.
But it still will not make it easier for physicians who do not have experience in palliative care, Dr. Edwards said. “It’s a very hard discussion to have; it’s not something doctors are trained to do.”
A recent study in non–small cell lung cancer patients found that those who were given palliative care at the time of diagnosis had a better quality of life than did those in standard care (N. Engl. J. Med. 2010;363:733-42). This study may do more to advance the field than does the New York law, Dr. Edwards noted.
Although the Hospice and Palliative Care Association of New York State supported the law, the Medical Society of the State of New York did not. The medical society, which represents 25,000 physicians, opposed the law because of concerns that it would interfere with the way each and every doctor navigates through end-of-life situations with each individual patient, said Elizabeth C. Dears, the society’s senior vice president for legislative and regulatory affairs.
Mandating that information be given on palliative care “may undermine the patient’s belief and conviction in prevailing against their disease and undercut the confidence in their treating physician,” said Ms. Dears.
The medical society also said that physicians are not licensed to provide legal advice in areas such as pain or symptom management, and that they may not know what they are supposed to be communicating to patients under certain provisions, while still being subject to penalties.
Although the medical society might object to requiring any such talk, both Dr. Flansbaum and Dr. Edwards said that, realistically, the law should be requiring palliative care to be offered sooner in the disease process and to a broader group of patients, such as those who have chronic life-limiting conditions such as heart failure.
“By the time you’re invoking palliative care in terminal patients, you’re behind the curve,” said Dr. Flansbaum.
ONLINE EXCLUSIVE: Palliative Care Documentation Key to Core Measures
A growing number of publicly reported hospital quality initiatives include severity-adjusted hospital mortality rates. Although individual hospitalists are unlikely to be rated based on their patients’ mortality, this is an important component of how hospitals are evaluated—and thus a natural target for the hospital’s quality-improvement (QI) efforts and for hospitalists’ participation in them.
The challenge is that some hospital-connected deaths are unavoidable, predictable, and even appropriate when care plans focused on maximizing comfort and quality of life replace medical efforts to stave off death. Referring seriously ill patients to the hospital’s palliative-care service or to a community hospice can influence a hospital’s mortality rate, but not always in the same ways.
Where hospice care and palliative care fit in hospital mortality rates, how they are defined and counted, and how predictable deaths are either included or excluded from hospitals’ risk-adjusted mortality tallies vary between the reporting programs, according to J. Brian Cassel, PhD, senior analyst at Virginia Commonwealth University (VCU), a presenter at the National Hospice and Palliative Care Organization’s Management and Leadership Conference in April 2010 in Washington, D.C.
“Hospitals are naturally concerned about mortality rates because they want to be seen as quality health providers,” Dr. Cassel says. “How hospital mortality rates are determined can be quite complex,” with varied data sources and various methods of adjusting for severity and balancing mortality with other quality metrics. Dr. Cassel says he began digging into mortality data when concerns were raised that VCU’s acute-palliative-care unit might be causing the medical center’s overall mortality rates to spike. His research found that the unit’s operation was probably neutral relative to VCU’s overall mortality rates.
Typically, the risk-adjusted mortality rate is for selected diagnoses but counts deaths from all causes, either during the index hospitalization or within 30 days of that admission, Dr. Cassel says. Three examples of QI programs that use mortality data: CMS’ Hospital Compare, which publicly reports data on patient satisfaction and hospital processes and outcomes, including mortality; U.S. News & World Report’s “Best Hospitals” list, for which one-third of total scores are derived from its mortality index; and HealthGrades, a Golden, Colo.-based company that ranks hospitals and other health providers within a region, one condition or procedure at a time.
An ICD-9 billing code, V66.7 for “palliative care encounter,” can flag the involvement of palliative-care consultants on a hospital case, although this code often goes unused and should be among the top nine listed diagnoses in order to turn up in most quality calculations. Palliative-care consultants can help promote the use and higher positioning of this code in hospital billing, along with more complete documentation of comorbidities and symptoms. It also is possible that involving hospice and palliative-care teams with seriously ill patients earlier in their disease progression could help manage their care in community settings, avoiding hospitalizations when death is likely in the next few months.
Some hospitals might choose to refer patients thought to be close to death to contracted hospice programs—and some hospice and palliative-care advocates are using the rates as conversation starters with hospital administrators. Dr. Cassel’s advice for those advocates: Know which quality-measurement systems the hospital’s leadership follows, where adjusted mortality rates fit in those systems, and how hospice and palliative care affect them.
Regardless of mortality metrics, Dr. Cassell says, a clinician’s primary responsibility is to provide the best possible care to patients and families, reflecting their values, hopes, and treatment goals.
A growing number of publicly reported hospital quality initiatives include severity-adjusted hospital mortality rates. Although individual hospitalists are unlikely to be rated based on their patients’ mortality, this is an important component of how hospitals are evaluated—and thus a natural target for the hospital’s quality-improvement (QI) efforts and for hospitalists’ participation in them.
The challenge is that some hospital-connected deaths are unavoidable, predictable, and even appropriate when care plans focused on maximizing comfort and quality of life replace medical efforts to stave off death. Referring seriously ill patients to the hospital’s palliative-care service or to a community hospice can influence a hospital’s mortality rate, but not always in the same ways.
Where hospice care and palliative care fit in hospital mortality rates, how they are defined and counted, and how predictable deaths are either included or excluded from hospitals’ risk-adjusted mortality tallies vary between the reporting programs, according to J. Brian Cassel, PhD, senior analyst at Virginia Commonwealth University (VCU), a presenter at the National Hospice and Palliative Care Organization’s Management and Leadership Conference in April 2010 in Washington, D.C.
“Hospitals are naturally concerned about mortality rates because they want to be seen as quality health providers,” Dr. Cassel says. “How hospital mortality rates are determined can be quite complex,” with varied data sources and various methods of adjusting for severity and balancing mortality with other quality metrics. Dr. Cassel says he began digging into mortality data when concerns were raised that VCU’s acute-palliative-care unit might be causing the medical center’s overall mortality rates to spike. His research found that the unit’s operation was probably neutral relative to VCU’s overall mortality rates.
Typically, the risk-adjusted mortality rate is for selected diagnoses but counts deaths from all causes, either during the index hospitalization or within 30 days of that admission, Dr. Cassel says. Three examples of QI programs that use mortality data: CMS’ Hospital Compare, which publicly reports data on patient satisfaction and hospital processes and outcomes, including mortality; U.S. News & World Report’s “Best Hospitals” list, for which one-third of total scores are derived from its mortality index; and HealthGrades, a Golden, Colo.-based company that ranks hospitals and other health providers within a region, one condition or procedure at a time.
An ICD-9 billing code, V66.7 for “palliative care encounter,” can flag the involvement of palliative-care consultants on a hospital case, although this code often goes unused and should be among the top nine listed diagnoses in order to turn up in most quality calculations. Palliative-care consultants can help promote the use and higher positioning of this code in hospital billing, along with more complete documentation of comorbidities and symptoms. It also is possible that involving hospice and palliative-care teams with seriously ill patients earlier in their disease progression could help manage their care in community settings, avoiding hospitalizations when death is likely in the next few months.
Some hospitals might choose to refer patients thought to be close to death to contracted hospice programs—and some hospice and palliative-care advocates are using the rates as conversation starters with hospital administrators. Dr. Cassel’s advice for those advocates: Know which quality-measurement systems the hospital’s leadership follows, where adjusted mortality rates fit in those systems, and how hospice and palliative care affect them.
Regardless of mortality metrics, Dr. Cassell says, a clinician’s primary responsibility is to provide the best possible care to patients and families, reflecting their values, hopes, and treatment goals.
A growing number of publicly reported hospital quality initiatives include severity-adjusted hospital mortality rates. Although individual hospitalists are unlikely to be rated based on their patients’ mortality, this is an important component of how hospitals are evaluated—and thus a natural target for the hospital’s quality-improvement (QI) efforts and for hospitalists’ participation in them.
The challenge is that some hospital-connected deaths are unavoidable, predictable, and even appropriate when care plans focused on maximizing comfort and quality of life replace medical efforts to stave off death. Referring seriously ill patients to the hospital’s palliative-care service or to a community hospice can influence a hospital’s mortality rate, but not always in the same ways.
Where hospice care and palliative care fit in hospital mortality rates, how they are defined and counted, and how predictable deaths are either included or excluded from hospitals’ risk-adjusted mortality tallies vary between the reporting programs, according to J. Brian Cassel, PhD, senior analyst at Virginia Commonwealth University (VCU), a presenter at the National Hospice and Palliative Care Organization’s Management and Leadership Conference in April 2010 in Washington, D.C.
“Hospitals are naturally concerned about mortality rates because they want to be seen as quality health providers,” Dr. Cassel says. “How hospital mortality rates are determined can be quite complex,” with varied data sources and various methods of adjusting for severity and balancing mortality with other quality metrics. Dr. Cassel says he began digging into mortality data when concerns were raised that VCU’s acute-palliative-care unit might be causing the medical center’s overall mortality rates to spike. His research found that the unit’s operation was probably neutral relative to VCU’s overall mortality rates.
Typically, the risk-adjusted mortality rate is for selected diagnoses but counts deaths from all causes, either during the index hospitalization or within 30 days of that admission, Dr. Cassel says. Three examples of QI programs that use mortality data: CMS’ Hospital Compare, which publicly reports data on patient satisfaction and hospital processes and outcomes, including mortality; U.S. News & World Report’s “Best Hospitals” list, for which one-third of total scores are derived from its mortality index; and HealthGrades, a Golden, Colo.-based company that ranks hospitals and other health providers within a region, one condition or procedure at a time.
An ICD-9 billing code, V66.7 for “palliative care encounter,” can flag the involvement of palliative-care consultants on a hospital case, although this code often goes unused and should be among the top nine listed diagnoses in order to turn up in most quality calculations. Palliative-care consultants can help promote the use and higher positioning of this code in hospital billing, along with more complete documentation of comorbidities and symptoms. It also is possible that involving hospice and palliative-care teams with seriously ill patients earlier in their disease progression could help manage their care in community settings, avoiding hospitalizations when death is likely in the next few months.
Some hospitals might choose to refer patients thought to be close to death to contracted hospice programs—and some hospice and palliative-care advocates are using the rates as conversation starters with hospital administrators. Dr. Cassel’s advice for those advocates: Know which quality-measurement systems the hospital’s leadership follows, where adjusted mortality rates fit in those systems, and how hospice and palliative care affect them.
Regardless of mortality metrics, Dr. Cassell says, a clinician’s primary responsibility is to provide the best possible care to patients and families, reflecting their values, hopes, and treatment goals.
ONLINE EXCLUSIVE: Audio interview with a pediatric hospitalist who is starting a palliative care team
In Search of a "Good Death"
An elderly patient, Mr. Jones, presents for his routine primary care office visit. His wife, who cares for him at home, reports that his dementia is growing worse. He is nonverbal, is having difficulty swallowing, and has lost 10% of his body weight in the past six months. He was recently hospitalized for treatment of aspiration pneumonia and experienced a marked decline during his hospital stay.
What action on the provider’s part would be most helpful for Mr. Jones and his family during this visit?
a) Order a swallow study and a chest x-ray.
b) Evaluate Mr. Jones for depression.
c) Help his family to plan for a comfortable death in the setting of their choosing.
The answer is, of course, “c.” Broaching this topic can be uncomfortable and time-consuming. But for a number of compelling reasons, providers should be communicating with their patients regarding their wishes in the final months of life.
Mr. Jones is at very high risk for dying in the coming six months; he qualifies for hospice care today. Timely hospice care will include physical and emotional support for his family and will allow Mr. Jones to die comfortably in the location that he and his family prefer.
Dementia and End-of-Life Care
Dementia was first identified as a terminal illness in 2000, when the American Medical Association (AMA) issued the Practical Guide for the Primary Care Physician on the Diagnosis, Management and Treatment of Dementia.1 This guide, in addition to more recently published literature,2,3,4 can assist primary care providers in diagnosing, managing, and treating patients with dementia from diagnosis to end of life. The AMA guide recommends that patients with dementia be offered comfort-focused care early in the course of their disease.1
Among recently admitted nursing home residents, it has been shown, about 48% have dementia.5,6 Even in this setting, patients with dementia are often not considered terminally ill.7 In one study of patients with advanced dementia who were admitted to a nursing home, only 1% were perceived by the facility staff to have a life expectancy of six months or less. In reality, 71% of those patients died within six months of admission.8 Alzheimer’s dementia, specifically, is the fifth leading cause of death among persons older than 65,9 yet even medical professionals often fail to recognize this condition as a terminal illness.
Although some 80% of Americans say they want to die at home, more than 70% die in a facility. Additionally, nearly 30% of Medicare enrollees are admitted to intensive care during their terminal hospital stay.10 Families of terminally ill patients, especially those with dementia, often make poor end-of-life decisions for several reasons: They do not understand the natural progression of the patient’s illness; they are unaware of the benefits and burdens of available treatments; and their decisions are often colored by the burden of guilt.11
Without essential conversations with the health care provider and an individualized plan in place, patients and their families will continue to seek help in emergency departments (EDs) and hospitals for treatment of possibly manageable symptoms: pain, fever, dyspnea, constipation.8 Many members of this vulnerable population will submit to aggressive medical interventions (eg, respirators, tube feeding, IV hydration, CPR12), when what they really require is high-touch comfort care.1
The Choice of Hospice
Hospice is a viable option for patients with dementia. Among family members of hospice care patients, 75% are very satisfied with that care.13 Yet according to 2008 statistics from the Hospice Association of America (HAA),14 only one in 10 patients who qualify for hospice care actually choose it. In one study, 10% of a group of recently admitted nursing home residents with dementia were perceived to have less than six months to live, but only 5.7% were referred to hospice.15 The HAA recommends that health care providers who care for terminally ill patients in clinical facilities open a dialogue with patients and family caregivers about the option of hospice.14
Once a patient is referred and accepted, the hospice team can educate the family about the benefits and burdens of end-of-life treatments. Together, they can formulate a plan and put into place resources that may be needed if the patient begins to deteriorate rapidly. Thanks to the Medicare Hospice Benefit16 (which provides specialized services in addition to members’ regular Medicare Part A benefits), oxygen can be ordered without the patient’s meeting oximetry specifications, and emergency medications may be kept in the patient’s home for future need. Patients who require inpatient hospice care will be transferred to a facility owned by or under contract with the Medicare-approved hospice program.
A Role in Providing End-of-Life Care
Primary care providers can play a significant role in improving quality of life for their end-stage patients. Optimal end-of-life care begins with information for patients and their caregivers about the expected progression of their illness. Family members may not know, for example, that dementia is considered a terminal illness.
During office visits with patients who have end-stage dementia and their family members, the provider should7,17-20:
• Review the expected course of dementia. Providers can help the family understand what to expect in the coming months. Hearing that the patient is in the late stage of the disease can be helpful to family members. Increased functional dependency (eg, dressing, bathing, toileting) and recurrent infections (with risk increased by dysphagia, apraxia, and reduced mobility) are likely.7,17
• Offer caregivers sources of emotional support. Families who receive such support are better able to provide care in the home, putting off the need for institutionalization. In addition to caregiver support groups, the Alzheimer’s Association offers a 24-hour helpline number: (800) 272-3900. The hospice team will also provide emotional and spiritual support.
• Remind caregivers that weight loss is expected in patients with end-stage dementia. Difficulty swallowing and other eating problems are common in patients with end-stage dementia. Tube feeding appears to offer neither survival advantage, nor improved nutritional status, nor improvement in quality of life in dementia patients, compared with hand feeding.18 Tube feeding has also been linked with increased risk of aspiration pneumonia. Yet surveyed hospital physicians often express the belief that feeding tubes have benefits not supported in evidence-based literature; they have also been shown to underestimate the 30-day mortality rate in dementia patients with feeding tubes.19 In some states, up to 44% of nursing home residents with dementia reportedly die with a feeding tube in place.7
Because the decision to implement enteral tube feeding is often based on emotional rather than factual data, it is important to discuss this practice with the family in advance of a crisis. Once hospitalized, patients with dementia are likely to be offered feeding tube insertion. The Alzheimer’s Association recommends a conscientious program of hand feeding rather than tube feeding.12 A brochure addressing this and other end-of-life decisions can be downloaded from the association’s Web site and shared with patients’ decision makers (www.alz.org/national/documents/brochure_endoflifedecisions.pdf).12
• Describe the burdens associated with hospitalization for patients with dementia. Adjusting to new routines and new caregivers who do not understand patients’ needs can trigger significant anxiety and accelerate their decline. Treatments that cause discomfort may agitate them. Patients with dementia who are hospitalized have been found to lose weight and experience loss of function in the activities of daily living—developments that are not reversed after hospital discharge.21
Nursing home residents may include a “do not hospitalize” order in their advance directives. For patients who would ordinarily be admitted for treatment of an infection, clinicians can consider less invasive therapy without moving the patient from where he or she lives. Oral antibiotics have been shown as effective as parenteral agents for treating infections in patients with dementia.21
• Explain why a do-not-resuscitate (DNR) order is advisable. The absence of a DNR directive is one of the strongest predictors of high utilization of medical care near the end of life.20 Contrary to perceptions families may have developed from watching television, the CPR survival rate for non–community-dwelling elderly persons is only 1% to 2%, and those who survive do so only briefly, if not with severe disability.22 Nor does CPR begin and end in the field; it is important for the provider to clarify that the CPR process starts at home but is concluded in the ED—or possibly in the ICU, with the patient on a ventilator. Thus, the choice of CPR should be portrayed as an option that is likely to be futile and that may actually increase a dying patient’s distress.
Many families struggle with the notion of withholding any intervention, no matter how small the potential benefit; they fear that the patient with a durable DNR order will receive limited care—or no care. It is important for the provider to clarify that “do not resuscitate” does not mean “do not treat.” In fact, families can still activate emergency medical services if they need help. But instead of performing CPR, the EMT will administer aggressive comfort measures (eg, pain management, hydration). Being with patients as natural death occurs may be the most important assistance family members can provide.
Conclusion
The health care provider plays an invaluable role in educating and supporting families who seek a good death for a loved one. Most elderly patients do not want aggressive end-of-life care; they and their families welcome discussions and strategies that will help them maintain control during the course of a terminal illness.
Many patients are often candidates for hospice care months sooner than clinicians realize. Early referral to hospice care will ensure that their physical needs—and the family’s emotional needs—are met in the optimal setting.
1. American Medical Association, Program on Aging and Community Health. Practical Guide for the Primary Care Physician on the Diagnosis, Management and Treatment of Dementia. Chicago, IL: American Medical Association; 2000.
2. Farias ST, Mungas D, Reed BR, et al. The measurement of everyday cognition (ECog): scale development and psychometric properties. Neuropsychology. 2008;22(4):531-544.
3. Boustani M, Peterson B, Hanson L, et al. Screening for dementia in primary care: a summary of the evidence for the US Preventive Services Task Force. Ann Intern Med. 2003;138(11): 927-937.
4. US Preventive Services Task Force. Screening for dementia: recommendation and rationale. Ann Intern Med. 2003;138(11):925-926.
5. Magaziner J, German P, Zimmerman SI, et al; Epidemiology of Dementia in Nursing Homes Research Group. The prevalence of dementia in a statewide sample of new nursing home admissions aged 65 or older: diagnosis by expert panel. Gerontologist. 2000;40(6):663-672.
6. Jakob A, Busse A, Riedel-Heller SG, et al. Prevalence and incidence of dementia among nursing home residents and residents in homes for the aged in comparison to private homes [in German]. Z Gerontol Geriatr. 2002;35(5):474-481.
7. Sachs GA, Shega JW, Cox-Hayley D. Barriers to excellent end-of-life care for patients with dementia. J Gen Intern Med. 2004;19(10):1057-1063.
8. Mitchell SL, Kiely DK, Hamel MB. Dying with advanced dementia in the nursing home. Arch Intern Med. 2004;164(3):321-326.
9. Alzheimer’s Association. 2009 Alzheimer’s Disease Facts and Figures. www.alz.org/national/documents/report_alzfactsfigures2009.pdf. Accessed September 24, 2009.
10. Wennberg JE, Cooper MM. Dartmouth Atlas of Health Care. Washington, DC: AHA Press; 1999.
11. Forbes S, Bern-Klug M, Gessert C. End-of-life decision making for nursing home residents with dementia. J Nurs Scholarsh. 2000;32(3):251-258.
12. Alzheimer’s Association. End-of-Life Decisions: Honoring the Wishes of the Person With Alzheimer’s Disease. www.alz.org/national/docu ments/brochure_endoflifedecisions.pdf. Accessed September 23, 2009.
13. Mitchell SL, Kiely DK, Miller SC, et al. Hospice care for patients with dementia. J Pain Symptom Manage. 2007;34(1):7-16.
14. Hospice Association of America. Hospice facts and statistics (2008). www.nahc.org/facts/Hospice Stats08.pdf. Accessed October 26, 2009.
15. Mitchell SL, Morris JN, Park PS, Fried BE. Terminal care for persons with advanced dementia in the nursing home and home care settings. J Palliat Med. 2004;7(6):808-816.
16. Centers for Medicare and Medicaid Services, US Department of Health and Human Services. Medicare Hospice Benefits (2008). CMS Publication No. 02154. www.medicare.gov/publications/Pubs/pdf/02154.pdf. Accessed October 26, 2009.
17. Mitchell SL, Kiely DK, Hamel MB, et al. Estimating prognosis for nursing home residents with advanced dementia. JAMA. 2004;291(22):2734-2740.
18. Sampson EL, Candy B, Jones L. Enteral tube feeding for older people with advanced dementia. Cochrane Database Syst Rev. 2009;(2): CD007209.
19. Shega JW, Hougham GW, Stocking CB, et al. Barriers to limiting the practice of feeding tube placement in advanced dementia. J Palliat Med. 2003;6(6):885-893.
20. Haller IV, Gessert CE. Utilization of medical services at the end of life in older adults with cognitive impairment: focus on outliers. J Palliat Med. 2007;10(2):400-407.
21. Volicer L, McKee A, Hewitt S. Dementia. Neurol Clin. 2001;19(4):867-885.
22. Mohr M, Bömelburg K, Bahr J. Attempted CPR in nursing homes: life-saving at the end of life? [in German] Anasthesiol Intensivmed Notfallmed Schmerzther. 2001;36(9):566-572.
An elderly patient, Mr. Jones, presents for his routine primary care office visit. His wife, who cares for him at home, reports that his dementia is growing worse. He is nonverbal, is having difficulty swallowing, and has lost 10% of his body weight in the past six months. He was recently hospitalized for treatment of aspiration pneumonia and experienced a marked decline during his hospital stay.
What action on the provider’s part would be most helpful for Mr. Jones and his family during this visit?
a) Order a swallow study and a chest x-ray.
b) Evaluate Mr. Jones for depression.
c) Help his family to plan for a comfortable death in the setting of their choosing.
The answer is, of course, “c.” Broaching this topic can be uncomfortable and time-consuming. But for a number of compelling reasons, providers should be communicating with their patients regarding their wishes in the final months of life.
Mr. Jones is at very high risk for dying in the coming six months; he qualifies for hospice care today. Timely hospice care will include physical and emotional support for his family and will allow Mr. Jones to die comfortably in the location that he and his family prefer.
Dementia and End-of-Life Care
Dementia was first identified as a terminal illness in 2000, when the American Medical Association (AMA) issued the Practical Guide for the Primary Care Physician on the Diagnosis, Management and Treatment of Dementia.1 This guide, in addition to more recently published literature,2,3,4 can assist primary care providers in diagnosing, managing, and treating patients with dementia from diagnosis to end of life. The AMA guide recommends that patients with dementia be offered comfort-focused care early in the course of their disease.1
Among recently admitted nursing home residents, it has been shown, about 48% have dementia.5,6 Even in this setting, patients with dementia are often not considered terminally ill.7 In one study of patients with advanced dementia who were admitted to a nursing home, only 1% were perceived by the facility staff to have a life expectancy of six months or less. In reality, 71% of those patients died within six months of admission.8 Alzheimer’s dementia, specifically, is the fifth leading cause of death among persons older than 65,9 yet even medical professionals often fail to recognize this condition as a terminal illness.
Although some 80% of Americans say they want to die at home, more than 70% die in a facility. Additionally, nearly 30% of Medicare enrollees are admitted to intensive care during their terminal hospital stay.10 Families of terminally ill patients, especially those with dementia, often make poor end-of-life decisions for several reasons: They do not understand the natural progression of the patient’s illness; they are unaware of the benefits and burdens of available treatments; and their decisions are often colored by the burden of guilt.11
Without essential conversations with the health care provider and an individualized plan in place, patients and their families will continue to seek help in emergency departments (EDs) and hospitals for treatment of possibly manageable symptoms: pain, fever, dyspnea, constipation.8 Many members of this vulnerable population will submit to aggressive medical interventions (eg, respirators, tube feeding, IV hydration, CPR12), when what they really require is high-touch comfort care.1
The Choice of Hospice
Hospice is a viable option for patients with dementia. Among family members of hospice care patients, 75% are very satisfied with that care.13 Yet according to 2008 statistics from the Hospice Association of America (HAA),14 only one in 10 patients who qualify for hospice care actually choose it. In one study, 10% of a group of recently admitted nursing home residents with dementia were perceived to have less than six months to live, but only 5.7% were referred to hospice.15 The HAA recommends that health care providers who care for terminally ill patients in clinical facilities open a dialogue with patients and family caregivers about the option of hospice.14
Once a patient is referred and accepted, the hospice team can educate the family about the benefits and burdens of end-of-life treatments. Together, they can formulate a plan and put into place resources that may be needed if the patient begins to deteriorate rapidly. Thanks to the Medicare Hospice Benefit16 (which provides specialized services in addition to members’ regular Medicare Part A benefits), oxygen can be ordered without the patient’s meeting oximetry specifications, and emergency medications may be kept in the patient’s home for future need. Patients who require inpatient hospice care will be transferred to a facility owned by or under contract with the Medicare-approved hospice program.
A Role in Providing End-of-Life Care
Primary care providers can play a significant role in improving quality of life for their end-stage patients. Optimal end-of-life care begins with information for patients and their caregivers about the expected progression of their illness. Family members may not know, for example, that dementia is considered a terminal illness.
During office visits with patients who have end-stage dementia and their family members, the provider should7,17-20:
• Review the expected course of dementia. Providers can help the family understand what to expect in the coming months. Hearing that the patient is in the late stage of the disease can be helpful to family members. Increased functional dependency (eg, dressing, bathing, toileting) and recurrent infections (with risk increased by dysphagia, apraxia, and reduced mobility) are likely.7,17
• Offer caregivers sources of emotional support. Families who receive such support are better able to provide care in the home, putting off the need for institutionalization. In addition to caregiver support groups, the Alzheimer’s Association offers a 24-hour helpline number: (800) 272-3900. The hospice team will also provide emotional and spiritual support.
• Remind caregivers that weight loss is expected in patients with end-stage dementia. Difficulty swallowing and other eating problems are common in patients with end-stage dementia. Tube feeding appears to offer neither survival advantage, nor improved nutritional status, nor improvement in quality of life in dementia patients, compared with hand feeding.18 Tube feeding has also been linked with increased risk of aspiration pneumonia. Yet surveyed hospital physicians often express the belief that feeding tubes have benefits not supported in evidence-based literature; they have also been shown to underestimate the 30-day mortality rate in dementia patients with feeding tubes.19 In some states, up to 44% of nursing home residents with dementia reportedly die with a feeding tube in place.7
Because the decision to implement enteral tube feeding is often based on emotional rather than factual data, it is important to discuss this practice with the family in advance of a crisis. Once hospitalized, patients with dementia are likely to be offered feeding tube insertion. The Alzheimer’s Association recommends a conscientious program of hand feeding rather than tube feeding.12 A brochure addressing this and other end-of-life decisions can be downloaded from the association’s Web site and shared with patients’ decision makers (www.alz.org/national/documents/brochure_endoflifedecisions.pdf).12
• Describe the burdens associated with hospitalization for patients with dementia. Adjusting to new routines and new caregivers who do not understand patients’ needs can trigger significant anxiety and accelerate their decline. Treatments that cause discomfort may agitate them. Patients with dementia who are hospitalized have been found to lose weight and experience loss of function in the activities of daily living—developments that are not reversed after hospital discharge.21
Nursing home residents may include a “do not hospitalize” order in their advance directives. For patients who would ordinarily be admitted for treatment of an infection, clinicians can consider less invasive therapy without moving the patient from where he or she lives. Oral antibiotics have been shown as effective as parenteral agents for treating infections in patients with dementia.21
• Explain why a do-not-resuscitate (DNR) order is advisable. The absence of a DNR directive is one of the strongest predictors of high utilization of medical care near the end of life.20 Contrary to perceptions families may have developed from watching television, the CPR survival rate for non–community-dwelling elderly persons is only 1% to 2%, and those who survive do so only briefly, if not with severe disability.22 Nor does CPR begin and end in the field; it is important for the provider to clarify that the CPR process starts at home but is concluded in the ED—or possibly in the ICU, with the patient on a ventilator. Thus, the choice of CPR should be portrayed as an option that is likely to be futile and that may actually increase a dying patient’s distress.
Many families struggle with the notion of withholding any intervention, no matter how small the potential benefit; they fear that the patient with a durable DNR order will receive limited care—or no care. It is important for the provider to clarify that “do not resuscitate” does not mean “do not treat.” In fact, families can still activate emergency medical services if they need help. But instead of performing CPR, the EMT will administer aggressive comfort measures (eg, pain management, hydration). Being with patients as natural death occurs may be the most important assistance family members can provide.
Conclusion
The health care provider plays an invaluable role in educating and supporting families who seek a good death for a loved one. Most elderly patients do not want aggressive end-of-life care; they and their families welcome discussions and strategies that will help them maintain control during the course of a terminal illness.
Many patients are often candidates for hospice care months sooner than clinicians realize. Early referral to hospice care will ensure that their physical needs—and the family’s emotional needs—are met in the optimal setting.
An elderly patient, Mr. Jones, presents for his routine primary care office visit. His wife, who cares for him at home, reports that his dementia is growing worse. He is nonverbal, is having difficulty swallowing, and has lost 10% of his body weight in the past six months. He was recently hospitalized for treatment of aspiration pneumonia and experienced a marked decline during his hospital stay.
What action on the provider’s part would be most helpful for Mr. Jones and his family during this visit?
a) Order a swallow study and a chest x-ray.
b) Evaluate Mr. Jones for depression.
c) Help his family to plan for a comfortable death in the setting of their choosing.
The answer is, of course, “c.” Broaching this topic can be uncomfortable and time-consuming. But for a number of compelling reasons, providers should be communicating with their patients regarding their wishes in the final months of life.
Mr. Jones is at very high risk for dying in the coming six months; he qualifies for hospice care today. Timely hospice care will include physical and emotional support for his family and will allow Mr. Jones to die comfortably in the location that he and his family prefer.
Dementia and End-of-Life Care
Dementia was first identified as a terminal illness in 2000, when the American Medical Association (AMA) issued the Practical Guide for the Primary Care Physician on the Diagnosis, Management and Treatment of Dementia.1 This guide, in addition to more recently published literature,2,3,4 can assist primary care providers in diagnosing, managing, and treating patients with dementia from diagnosis to end of life. The AMA guide recommends that patients with dementia be offered comfort-focused care early in the course of their disease.1
Among recently admitted nursing home residents, it has been shown, about 48% have dementia.5,6 Even in this setting, patients with dementia are often not considered terminally ill.7 In one study of patients with advanced dementia who were admitted to a nursing home, only 1% were perceived by the facility staff to have a life expectancy of six months or less. In reality, 71% of those patients died within six months of admission.8 Alzheimer’s dementia, specifically, is the fifth leading cause of death among persons older than 65,9 yet even medical professionals often fail to recognize this condition as a terminal illness.
Although some 80% of Americans say they want to die at home, more than 70% die in a facility. Additionally, nearly 30% of Medicare enrollees are admitted to intensive care during their terminal hospital stay.10 Families of terminally ill patients, especially those with dementia, often make poor end-of-life decisions for several reasons: They do not understand the natural progression of the patient’s illness; they are unaware of the benefits and burdens of available treatments; and their decisions are often colored by the burden of guilt.11
Without essential conversations with the health care provider and an individualized plan in place, patients and their families will continue to seek help in emergency departments (EDs) and hospitals for treatment of possibly manageable symptoms: pain, fever, dyspnea, constipation.8 Many members of this vulnerable population will submit to aggressive medical interventions (eg, respirators, tube feeding, IV hydration, CPR12), when what they really require is high-touch comfort care.1
The Choice of Hospice
Hospice is a viable option for patients with dementia. Among family members of hospice care patients, 75% are very satisfied with that care.13 Yet according to 2008 statistics from the Hospice Association of America (HAA),14 only one in 10 patients who qualify for hospice care actually choose it. In one study, 10% of a group of recently admitted nursing home residents with dementia were perceived to have less than six months to live, but only 5.7% were referred to hospice.15 The HAA recommends that health care providers who care for terminally ill patients in clinical facilities open a dialogue with patients and family caregivers about the option of hospice.14
Once a patient is referred and accepted, the hospice team can educate the family about the benefits and burdens of end-of-life treatments. Together, they can formulate a plan and put into place resources that may be needed if the patient begins to deteriorate rapidly. Thanks to the Medicare Hospice Benefit16 (which provides specialized services in addition to members’ regular Medicare Part A benefits), oxygen can be ordered without the patient’s meeting oximetry specifications, and emergency medications may be kept in the patient’s home for future need. Patients who require inpatient hospice care will be transferred to a facility owned by or under contract with the Medicare-approved hospice program.
A Role in Providing End-of-Life Care
Primary care providers can play a significant role in improving quality of life for their end-stage patients. Optimal end-of-life care begins with information for patients and their caregivers about the expected progression of their illness. Family members may not know, for example, that dementia is considered a terminal illness.
During office visits with patients who have end-stage dementia and their family members, the provider should7,17-20:
• Review the expected course of dementia. Providers can help the family understand what to expect in the coming months. Hearing that the patient is in the late stage of the disease can be helpful to family members. Increased functional dependency (eg, dressing, bathing, toileting) and recurrent infections (with risk increased by dysphagia, apraxia, and reduced mobility) are likely.7,17
• Offer caregivers sources of emotional support. Families who receive such support are better able to provide care in the home, putting off the need for institutionalization. In addition to caregiver support groups, the Alzheimer’s Association offers a 24-hour helpline number: (800) 272-3900. The hospice team will also provide emotional and spiritual support.
• Remind caregivers that weight loss is expected in patients with end-stage dementia. Difficulty swallowing and other eating problems are common in patients with end-stage dementia. Tube feeding appears to offer neither survival advantage, nor improved nutritional status, nor improvement in quality of life in dementia patients, compared with hand feeding.18 Tube feeding has also been linked with increased risk of aspiration pneumonia. Yet surveyed hospital physicians often express the belief that feeding tubes have benefits not supported in evidence-based literature; they have also been shown to underestimate the 30-day mortality rate in dementia patients with feeding tubes.19 In some states, up to 44% of nursing home residents with dementia reportedly die with a feeding tube in place.7
Because the decision to implement enteral tube feeding is often based on emotional rather than factual data, it is important to discuss this practice with the family in advance of a crisis. Once hospitalized, patients with dementia are likely to be offered feeding tube insertion. The Alzheimer’s Association recommends a conscientious program of hand feeding rather than tube feeding.12 A brochure addressing this and other end-of-life decisions can be downloaded from the association’s Web site and shared with patients’ decision makers (www.alz.org/national/documents/brochure_endoflifedecisions.pdf).12
• Describe the burdens associated with hospitalization for patients with dementia. Adjusting to new routines and new caregivers who do not understand patients’ needs can trigger significant anxiety and accelerate their decline. Treatments that cause discomfort may agitate them. Patients with dementia who are hospitalized have been found to lose weight and experience loss of function in the activities of daily living—developments that are not reversed after hospital discharge.21
Nursing home residents may include a “do not hospitalize” order in their advance directives. For patients who would ordinarily be admitted for treatment of an infection, clinicians can consider less invasive therapy without moving the patient from where he or she lives. Oral antibiotics have been shown as effective as parenteral agents for treating infections in patients with dementia.21
• Explain why a do-not-resuscitate (DNR) order is advisable. The absence of a DNR directive is one of the strongest predictors of high utilization of medical care near the end of life.20 Contrary to perceptions families may have developed from watching television, the CPR survival rate for non–community-dwelling elderly persons is only 1% to 2%, and those who survive do so only briefly, if not with severe disability.22 Nor does CPR begin and end in the field; it is important for the provider to clarify that the CPR process starts at home but is concluded in the ED—or possibly in the ICU, with the patient on a ventilator. Thus, the choice of CPR should be portrayed as an option that is likely to be futile and that may actually increase a dying patient’s distress.
Many families struggle with the notion of withholding any intervention, no matter how small the potential benefit; they fear that the patient with a durable DNR order will receive limited care—or no care. It is important for the provider to clarify that “do not resuscitate” does not mean “do not treat.” In fact, families can still activate emergency medical services if they need help. But instead of performing CPR, the EMT will administer aggressive comfort measures (eg, pain management, hydration). Being with patients as natural death occurs may be the most important assistance family members can provide.
Conclusion
The health care provider plays an invaluable role in educating and supporting families who seek a good death for a loved one. Most elderly patients do not want aggressive end-of-life care; they and their families welcome discussions and strategies that will help them maintain control during the course of a terminal illness.
Many patients are often candidates for hospice care months sooner than clinicians realize. Early referral to hospice care will ensure that their physical needs—and the family’s emotional needs—are met in the optimal setting.
1. American Medical Association, Program on Aging and Community Health. Practical Guide for the Primary Care Physician on the Diagnosis, Management and Treatment of Dementia. Chicago, IL: American Medical Association; 2000.
2. Farias ST, Mungas D, Reed BR, et al. The measurement of everyday cognition (ECog): scale development and psychometric properties. Neuropsychology. 2008;22(4):531-544.
3. Boustani M, Peterson B, Hanson L, et al. Screening for dementia in primary care: a summary of the evidence for the US Preventive Services Task Force. Ann Intern Med. 2003;138(11): 927-937.
4. US Preventive Services Task Force. Screening for dementia: recommendation and rationale. Ann Intern Med. 2003;138(11):925-926.
5. Magaziner J, German P, Zimmerman SI, et al; Epidemiology of Dementia in Nursing Homes Research Group. The prevalence of dementia in a statewide sample of new nursing home admissions aged 65 or older: diagnosis by expert panel. Gerontologist. 2000;40(6):663-672.
6. Jakob A, Busse A, Riedel-Heller SG, et al. Prevalence and incidence of dementia among nursing home residents and residents in homes for the aged in comparison to private homes [in German]. Z Gerontol Geriatr. 2002;35(5):474-481.
7. Sachs GA, Shega JW, Cox-Hayley D. Barriers to excellent end-of-life care for patients with dementia. J Gen Intern Med. 2004;19(10):1057-1063.
8. Mitchell SL, Kiely DK, Hamel MB. Dying with advanced dementia in the nursing home. Arch Intern Med. 2004;164(3):321-326.
9. Alzheimer’s Association. 2009 Alzheimer’s Disease Facts and Figures. www.alz.org/national/documents/report_alzfactsfigures2009.pdf. Accessed September 24, 2009.
10. Wennberg JE, Cooper MM. Dartmouth Atlas of Health Care. Washington, DC: AHA Press; 1999.
11. Forbes S, Bern-Klug M, Gessert C. End-of-life decision making for nursing home residents with dementia. J Nurs Scholarsh. 2000;32(3):251-258.
12. Alzheimer’s Association. End-of-Life Decisions: Honoring the Wishes of the Person With Alzheimer’s Disease. www.alz.org/national/docu ments/brochure_endoflifedecisions.pdf. Accessed September 23, 2009.
13. Mitchell SL, Kiely DK, Miller SC, et al. Hospice care for patients with dementia. J Pain Symptom Manage. 2007;34(1):7-16.
14. Hospice Association of America. Hospice facts and statistics (2008). www.nahc.org/facts/Hospice Stats08.pdf. Accessed October 26, 2009.
15. Mitchell SL, Morris JN, Park PS, Fried BE. Terminal care for persons with advanced dementia in the nursing home and home care settings. J Palliat Med. 2004;7(6):808-816.
16. Centers for Medicare and Medicaid Services, US Department of Health and Human Services. Medicare Hospice Benefits (2008). CMS Publication No. 02154. www.medicare.gov/publications/Pubs/pdf/02154.pdf. Accessed October 26, 2009.
17. Mitchell SL, Kiely DK, Hamel MB, et al. Estimating prognosis for nursing home residents with advanced dementia. JAMA. 2004;291(22):2734-2740.
18. Sampson EL, Candy B, Jones L. Enteral tube feeding for older people with advanced dementia. Cochrane Database Syst Rev. 2009;(2): CD007209.
19. Shega JW, Hougham GW, Stocking CB, et al. Barriers to limiting the practice of feeding tube placement in advanced dementia. J Palliat Med. 2003;6(6):885-893.
20. Haller IV, Gessert CE. Utilization of medical services at the end of life in older adults with cognitive impairment: focus on outliers. J Palliat Med. 2007;10(2):400-407.
21. Volicer L, McKee A, Hewitt S. Dementia. Neurol Clin. 2001;19(4):867-885.
22. Mohr M, Bömelburg K, Bahr J. Attempted CPR in nursing homes: life-saving at the end of life? [in German] Anasthesiol Intensivmed Notfallmed Schmerzther. 2001;36(9):566-572.
1. American Medical Association, Program on Aging and Community Health. Practical Guide for the Primary Care Physician on the Diagnosis, Management and Treatment of Dementia. Chicago, IL: American Medical Association; 2000.
2. Farias ST, Mungas D, Reed BR, et al. The measurement of everyday cognition (ECog): scale development and psychometric properties. Neuropsychology. 2008;22(4):531-544.
3. Boustani M, Peterson B, Hanson L, et al. Screening for dementia in primary care: a summary of the evidence for the US Preventive Services Task Force. Ann Intern Med. 2003;138(11): 927-937.
4. US Preventive Services Task Force. Screening for dementia: recommendation and rationale. Ann Intern Med. 2003;138(11):925-926.
5. Magaziner J, German P, Zimmerman SI, et al; Epidemiology of Dementia in Nursing Homes Research Group. The prevalence of dementia in a statewide sample of new nursing home admissions aged 65 or older: diagnosis by expert panel. Gerontologist. 2000;40(6):663-672.
6. Jakob A, Busse A, Riedel-Heller SG, et al. Prevalence and incidence of dementia among nursing home residents and residents in homes for the aged in comparison to private homes [in German]. Z Gerontol Geriatr. 2002;35(5):474-481.
7. Sachs GA, Shega JW, Cox-Hayley D. Barriers to excellent end-of-life care for patients with dementia. J Gen Intern Med. 2004;19(10):1057-1063.
8. Mitchell SL, Kiely DK, Hamel MB. Dying with advanced dementia in the nursing home. Arch Intern Med. 2004;164(3):321-326.
9. Alzheimer’s Association. 2009 Alzheimer’s Disease Facts and Figures. www.alz.org/national/documents/report_alzfactsfigures2009.pdf. Accessed September 24, 2009.
10. Wennberg JE, Cooper MM. Dartmouth Atlas of Health Care. Washington, DC: AHA Press; 1999.
11. Forbes S, Bern-Klug M, Gessert C. End-of-life decision making for nursing home residents with dementia. J Nurs Scholarsh. 2000;32(3):251-258.
12. Alzheimer’s Association. End-of-Life Decisions: Honoring the Wishes of the Person With Alzheimer’s Disease. www.alz.org/national/docu ments/brochure_endoflifedecisions.pdf. Accessed September 23, 2009.
13. Mitchell SL, Kiely DK, Miller SC, et al. Hospice care for patients with dementia. J Pain Symptom Manage. 2007;34(1):7-16.
14. Hospice Association of America. Hospice facts and statistics (2008). www.nahc.org/facts/Hospice Stats08.pdf. Accessed October 26, 2009.
15. Mitchell SL, Morris JN, Park PS, Fried BE. Terminal care for persons with advanced dementia in the nursing home and home care settings. J Palliat Med. 2004;7(6):808-816.
16. Centers for Medicare and Medicaid Services, US Department of Health and Human Services. Medicare Hospice Benefits (2008). CMS Publication No. 02154. www.medicare.gov/publications/Pubs/pdf/02154.pdf. Accessed October 26, 2009.
17. Mitchell SL, Kiely DK, Hamel MB, et al. Estimating prognosis for nursing home residents with advanced dementia. JAMA. 2004;291(22):2734-2740.
18. Sampson EL, Candy B, Jones L. Enteral tube feeding for older people with advanced dementia. Cochrane Database Syst Rev. 2009;(2): CD007209.
19. Shega JW, Hougham GW, Stocking CB, et al. Barriers to limiting the practice of feeding tube placement in advanced dementia. J Palliat Med. 2003;6(6):885-893.
20. Haller IV, Gessert CE. Utilization of medical services at the end of life in older adults with cognitive impairment: focus on outliers. J Palliat Med. 2007;10(2):400-407.
21. Volicer L, McKee A, Hewitt S. Dementia. Neurol Clin. 2001;19(4):867-885.
22. Mohr M, Bömelburg K, Bahr J. Attempted CPR in nursing homes: life-saving at the end of life? [in German] Anasthesiol Intensivmed Notfallmed Schmerzther. 2001;36(9):566-572.
Advance care planning: Beyond the living will
Mr. B., an 82-year-old retired accountant with hypertension, was diagnosed with early Alzheimer disease 6 years ago. He now needs supervision with bathing and dressing and no longer consistently recognizes family members. You are seeing him in the office today after a hospitalization for aspiration pneumonia, his second in the past 6 months.
In the hospital, a brain scan showed that atrophy had progressed and white-matter disease was more extensive than 3 years earlier. A barium swallow study showed esophageal dysmotility and aspiration. He was prescribed a “dysphagia diet,”1 which he dislikes.
Since returning home, he has been disoriented, he has been wandering about the house, and he has fallen several times. He has lost 10 pounds in 6 months. Because of his confusion, his wife cannot take him out, and she is exhausted caring for him.
Reviewing his medical record, you note that 10 years ago, Mr. B. completed a living will and designated his wife as his proxy decision-maker via a medical power of attorney document.
PLANNING IS OFTEN NEGLECTED
Many clinicians and older patients feel a strong need to document, in advance, the patient’s wishes regarding medical care in the event the patient becomes seriously ill and unable to participate in treatment decisions. Professional societies such as the American Geriatrics Society promote advance care planning,2 and some indices of the quality of medical care include whether advance directives have been discussed and completed.3
Yet, despite the high profile of advance care planning,4 few patients actually fill out advance directives,5 with completion rates that vary widely,6–8 sometimes by ethnicity and sex.9,10 Furthermore, in a crisis, these directives are seldom followed.11
In this paper, we recommend an approach to advance care planning for older adults that redirects the focus from “signing away” interventions such as dialysis, mechanical ventilation, and tube feeding. Instead, the focus is on the goals of care. We also advocate naming a surrogate decision-maker, since the medical power of attorney is more flexible and more widely applicable than the living will.
START BY LISTENING
A change in function resulting from disease progression, hospitalization, trauma, or other reasons is an ideal opportunity to introduce the process of advance care planning.
The first step is to find out how well the patient and family understand the patient’s relevant medical conditions, and what their expectations, hopes, and concerns are. This listening phase can provide insight into the patient’s values and goals and how much the patient and family want to engage in these discussions.
In matters of health behavior (such as advance care planning), people change only when they are ready to change.12,13 Thus, we advise physicians to defer extensive discussions of values and goals of care until patients and families are ready to listen, hear, and talk about these topics (often, after a change for the worse in prognosis).
And it is a process. Advance care directives are most likely to be set up and followed if the patient and doctor discuss this issue during multiple visits, rather than if the physician merely hands the patient a packet of forms and information.14–16
CASE CONTINUED: A PEG TUBE REFUSED
Mrs. B. says that Mr. B. is in good health except for his memory: he does not have a serious condition such as diabetes, heart failure, or cancer. While Mr. B. was in the hospital, the hospitalist recommended placing a percutaneous endoscopic gastrostomy (PEG) tube, but Mrs. B. declined the recommendation because her husband had a living will that specified “no artificially or technologically supplied nutrition or hydration.”
At this point, Mrs. B. begins to cry. She has slept poorly because of his wandering. Also, her two daughters do not support her refusal of the PEG tube.
Comment. This brief conversation illuminates knowledge deficits in Mrs. B.’s understanding of Alzheimer disease and the circumstances in which the living will applies. Although one could argue that Mr. B.’s Alzheimer disease has advanced to the point that he is likely to die of a complication of that condition, he is not likely to die in the near future. If he is not considered by law and his physician to be terminally ill or permanently unconscious, a living will likely does not offer guidance about artificial feeding.
LIMITATIONS OF A LIVING WILL
A living will, a commonly used advance directive, states that the patient does not wish to receive life-sustaining treatment in the event that he or she suffers an incurable, irreversible disease and cannot give informed consent, and it often lists specific treatments that the patient does not want. However, we believe that approaching the patient with a list of life-sustaining measures to accept or reject, before discussing goals of care and prognosis, puts the cart before the horse. This approach threatens to distract from the need to ascertain values and offer appropriate care. Additionally, a living will is active only within a very limited scenario and does not address relatively routine but important decisions in a person’s care.
All ‘terminal illness’ isn’t the same
A living will goes into effect only if the patient either enters a permanent vegetative state following an event such as cardiac arrest or severe brain trauma, or is diagnosed with a terminal illness such as metastatic cancer, and lacks decision-making capacity.
But what is terminal illness? The definition differs from state to state, but it is generally defined as an irreversible condition leading to death in a relatively short time. The time may not be specified, as in Florida statute 765. In contrast, Ohio Revised Code 2133.01(AA) uses the phrase “relatively short,” while other states specify a time, such as within 6 months (Texas Health and Safety Code 166.002). The Medicare hospice benefit also carries a short time limit, usually less than 6 months.
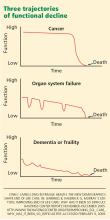
The middle panel in Figure 1 depicts a more typical decline from serial organ insults such as stroke followed by infection from aspiration or followed by falls. Older adults with dementia or with multiple progressive diseases such as heart failure, diabetes, hypertension, or cancer decline in a trajectory such as in the bottom panel of Figure 1.
A living will might not be activated in the latter two scenarios until years into the condition because the patient would not be considered terminally ill—by state law, by the health care provider, or even by the patient.
The living will does not address routine interventions
In most states, living wills address only life-sustaining treatments such as dialysis, mechanical ventilation, and medically supplied nutrition and hydration. Living wills do not address relatively common diseases in older adults that could cause severe debility, such as a major stroke or advanced dementia.
If an older patient has dementia, a living will is unlikely to provide guidance about interventions such as intubation to get through an episode of potentially reversible respiratory failure, a feeding tube to correct weight loss, or cardiac catheterization or bypass surgery to treat angina. Yet these important decisions often arise as function declines and comorbidities progress.
Patients may change their minds
Many older adults are reluctant to sign documents to “micromanage” their future care if they should become ill.19 Many people change their mind as the situation changes.11,20,21 Although few claim they would want burdensome interventions if they had dementia22 or if their prognosis were poor,23 patients may tolerate more burdensome interventions if they are already receiving treatments for chronic illnesses such as end-stage renal disease.24
Thus, a living will may help if unexpected trauma occurs in a healthy person, but not so much if chronic illness progresses over a period of years.
Advance directives may not be honored
Even if completed, written advance directives may not be followed, for a variety of reasons. Physicians may not know the patient has a living will, and fewer than one-third of people who actually complete an advance directive have discussed its content with a physician.25 The people named as surrogate decision-makers may not know the patient’s wishes. Family members may disagree with the goals and plan of care and may interfere with implementation of the advance care plan. A patient may see multiple physicians at different institutions who may not communicate with each other about the patient’s wishes. Also, physicians and patients may interpret terms such as “terminally ill” differently, making it difficult to translate the documents into an action plan.
CASE CONTINUED: RELIEVING CAREGIVER STRESS
Returning to Mr. B., your first goal is to address care issues, including caregiver stress. Skilled services in the home are appropriate for him at this time (and Medicare will pay for them) because he is still homebound. These services could include physical therapy, occupational therapy, and speech (swallowing) therapy. A home care agency may also provide an aide for a few weeks to assist with bathing and other personal needs.
You strongly recommend that the family (including both daughters) participate in the Alzheimer’s Association educational programs. You recommend that Mrs. B. locate an adult day care program now so that when Mr. B. completes his home therapy course and is no longer homebound, he may attend. Day care would provide a therapeutic environment for him and respite for her.
You request that the home care agency provide a social worker to advise her on community resources. Meta-analysis suggests that structured, multicomponent interventions with caregivers of demented patients reduce several types of caregiver burden and delay institutionalization.26
He improves with conservative measures
Three weeks later, Mr. B. is sleeping better and has stopped wandering. However, he dislikes the thickened liquids required by the dysphagia diet and has lost another 2 pounds. If his beverages are not thickened, he coughs profusely when he swallows. His daughters are still pressuring Mrs. B. for a PEG tube; one of them has angrily asserted that the doctors are going to allow her father to die.
You explain the burdens of PEG tubes: surgical risks, continued aspiration, disrupted bowel habits, the risk of the tube being accidentally or intentionally dislodged by the patient, and special binders (which may be uncomfortable) or restraints (which may cause further functional decline) that may be necessary to prevent this complication.
You request that the speech therapist work with the patient more aggressively in the use of swallowing techniques such as the chin tuck, which may be at least as effective as thickeners in preventing both aspiration pneumonia and dehydration.27 The therapist will need to include Mrs. B. in these sessions, since she will be Mr. B.’s coach at mealtime.
With more aggressive speech therapy, the patient’s weight stabilizes over the next 4 weeks. He is in day care 3 days a week, and Mrs. B. is more rested and relaxed.
Cardiopulmonary resuscitation
You continue the advance care planning discussion and suggest that if Mr. B. aspirates, is hospitalized again, and declines further care, it would be helpful to delineate instructions for resuscitation. Right now, although his Alzheimer disease is advanced, he is not clearly terminal. Thus, his living will does not strictly apply and provides limited guidance about intubation, cardiopulmonary resuscitation (CPR), or medically supplied nutrition and hydration. However, because Mrs. B. is his agent in the medical power of attorney, this document enables her to make a wide spectrum of treatment decisions on his behalf.
Mrs. B. asks about her husband’s prognosis and why CPR would not be helpful.
Comment. Further discussion with her could be guided by an estimate of Mr. B.’s prognosis. Function-based tools28,29 may also be useful. For example,28,30 an 80-year-old man with high functional status might have a life expectancy of more than 10 years. Mr. B., with multiple medical problems and declining function, would have an estimated life expectancy of approximately 3 years. Even without specifically categorizing function, impaired cognition by itself predicted a shorter life expectancy in population-based studies.31,32
Regarding CPR, patients and families may overestimate successful outcomes. A recent study33 of 10 years of outcomes of in-hospital cardiac arrest found that only 6.6% of patients survived to discharge. The average age of the survivors was 59 years, and fewer than half of them survived 3 years after cardiac arrest. In eight studies of CPR outcomes in nursing homes,34 three studies had no survivors, and all but one study had a survival rate below 5%.
PROVIDING APPROPRIATE CARE, NOT LIMITING TREATMENTS
In the case of Mr. B., as in many situations encountered with older patients, written advance directives provide little help or guidance. Instead, we recommend a model of advance care planning that takes place during multiple office visits over time, and that maintains a focus on providing appropriate care rather than on limiting life-sustaining treatments. We recommend providing estimates of prognosis and CPR outcomes when the family appeares ready to hear them. This approach should result in a care-oriented process while moving the family towards decisions regarding artificial feeding and CPR.
All patients, particularly those unwilling or unable to participate in advance care planning, are encouraged to identify one or more surrogate decision-makers and articulate how much flexibility that person should be given in important health care decisions. The medical power of attorney can be activated any time the patient lacks decision-making capacity and deactivated when decision-making capacity returns.38,39
As in the case of Mr. B., a tailored approach to advance care planning requires clinicians to estimate life expectancy (more than 5 years, less than 5 years, or less than 1 year) and to determine the patient’s and the family’s readiness to focus on a values-oriented and goal-oriented care plan. Some patients are not receptive to advance care planning, and clinical time and effort are optimized by providing the right amount of information to patients when they are ready to receive it.
For relatively healthy older adults
Figure 2 is the algorithm for older patients who are expected to live at least 5 years, ie, who are relatively healthy and functional. Patients with little or no interest in advance care planning can be asked about it annually, or sooner if their medical condition changes. Patients with limited interest can be given written information, specifically living will and medical power of attorney documents recognized in their state. Patients more open to advance care planning can be offered a values history form (Table 1), Web sites, and educational materials, with a plan to discuss them at future appointments.
Periodic reevaluation of values and goals of care is important. Patients may assert that particular interventions (eg, a PEG tube or dialysis) are “worse than death” when they are healthy, but they may change their views over time.21 Additionally, although a recent hospitalization or a decline in function may predispose patients to want to limit life-sustaining treatments, they may return to their earlier values and wishes a few months later, particularly if their medical condition stabilizes. 20 Values and decisions should be reassessed not only when medical conditions deteriorate, but also when they improve.
For chronically ill patients
Caregiver stress is important to identify and address, since caregivers often neglect themselves.40–42
For terminally ill patients
In this situation, patients and families need information about community resources that can assist them in the home. Some older adults with cognitive impairment may be exploited or neglect themselves, and referral to an adult protective services agency may be needed.
Treatment burden, particularly due to multiple prescribed medications, may be high and should be reassessed in light of the goals of treatment. Polypharmacy reduction is especially important at this stage in the illness, since the goals of care may be different than when the medications were prescribed.
Physical or psychosocial symptoms may be the cue to bring up the topic of palliative care. If the patient is expected to live less than 6 months, hospice referral is appropriate. With either palliative care or hospice, the focus of attention shifts explicitly from curing the disease to managing symptoms, and from the patient to the patient-family dyad. Interventions such as CPR and ventilatory support should be discussed and information from Table 2 provided to the patient and family.
Complete advance care planning incorporates taking a values history, estimating life expectancy, determining physical, psychosocial and spiritual needs, clarifying treatment goals, and estimating manageable treatment burden. Offering statistics on CPR and providing state-specific living will and medical power of attorney documents are important but are only one facet of effective advance care planning. In fact, shifting the emphasis of advance care planning from statistics and forms to values and goals of care may help in developing a more comprehensive care plan.
Goals of care range from curing the disease (with aggressive therapy, which may be burdensome) to simply improving function or decreasing pain. In the latter case, one may be able to discontinue some of the patient’s drugs, utilize medical and community resources more effectively, and better meet the patient’s needs.
Woven through all these discussions should be reassurance that the plan can be revisited and possibly revised, and that the physician will be there to help with those decisions.
For acutely ill patients in the hospital
Episodic, staged advance care planning is appropriate not only in the office but also in other settings such as assisted living and nursing facilities.
In the hospital, however, a different approach is needed, since patients are usually admitted because of an acute illness or sudden functional decline, or both. Decisions about technological interventions such as CPR, mechanical ventilation, or dialysis may be needed urgently. Often, patients are unable to provide guidance to physicians during acute illness because of delirium and other impediments. Developing a plan for care in the hospital may require urgent family meetings. However, if a surrogate decision-maker is in place, and if the patient has already participated in some form of advance care planning as an outpatient, the values and goals of care previously identified can contribute to decision-making during hospitalization.
As mentioned above, fragmentation of health care across providers and health care systems may limit the effectiveness of office-based advance care planning. It may be reasonable to train office staff to place advance care planning documentation in easily accessible sections of the patient’s medical record and to forward these to specialists involved in a patient’s care.
The patient and family should be encouraged and empowered to help with this process and should have updated advance care planning documentation readily available. In some states, comprehensive medical order sets, especially for end-of-life care, are portable across care settings and address CPR, medically supplied nutrition, hospital transfer, and antibiotic treatment.43
Research suggests that health care systems are more likely to comply with patients’ end-of-life preferences when portable medical order forms are developed and disseminated.44–44 Ultimately, major changes in health care delivery, including universal electronic health records, may be needed to implement and communicate patients’ advance care planning preferences across settings.
- National Dysphagia Diet Task Force. National Dysphagia Diet: Standardization for Optimal Care. Chicago, IL: American Dietetic Association, 2002.
- Nusbaum N, Goldstein M. American Geriatrics Society. The Patient Education Forum. Advance Directives, 2008. www.americangeriatrics.org/education/forum/advance_dir.shtml. Accessed March 9, 2009.
- Wenger NS, Roth CP, Shekelle PA; COVE Investigators. Introduction to the assessing care of vulnerable elders–3 quality indicator measurement set. J Am Geriatr Soc 2007; 55(suppl 2):S247–S252.
- Emanuel LL, Danis M, Pearlman RA, Singer PA. Advance care planning as a process: structuring the discussions in practice. J Am Geriatr Soc 1995; 43:440–446.
- Teno J, Lynn J, Wenger N, et al. Advance directives for seriously ill hospitalized patients: effectiveness with the patient self-determination act and the SUPPORT intervention. SUPPORT Investigators. Study to Understand Prognoses and P for Outcomes and Risks of Treatment. J Am Geriatr Soc 1997; 45:500–507.
- Hammes BJ, Rooney BL. Death and end-of-life planning in one midwestern community. Arch Intern Med 1998; 158:383–390.
- Gordon NP, Shade SB. Advance directives are more likely among seniors asked about end-of-life care p. Arch Intern Med 1999; 159:701–704.
- Morrison RS, Meier DE. High rates of advance care planning in New York City’s elderly population. Arch Intern Med 2004; 164:2421–2426.
- Perkins HS, Geppert CMA, Gonzales A, Cortez JD, Hazuda HP. Cross-cultural similarities and differences in attitudes about advance care planning. J Gen Intern Med 2002; 17:48–57.
- Perkins HS, Cortez JD, Hazuda HP. Advance care planning: does patient gender make a difference? Am J Med Sci 2004; 327:25–32.
- The SUPPORT Principal Investigators. A controlled trial to improve care for seriously ill hospitalized patients. Study to Understand Prognoses and P for Outcomes and Risks of Treatments (SUPPORT). JAMA 1995; 274:1591–1598.
- Prochaska JO, DiClemente CC, Norcross JC. In search of how people change. Applications to addictive behaviors. Am Psychol 1992; 47:1102–1114.
- Nigg CR, Burbank PM, Padula C, et al. Stages of change across ten health risk behaviors for older adults. Gerontologist 1999; 39:473–482.
- Patel RV, Sinuff T, Cook DJ. Influencing advance directive completion rates in non-terminally ill patients: a systematic review. J Crit Care 2004; 19:1–9.
- Hanson LC, Earp JA, Garrett J, Menon M, Danis M. Community physicians who provide terminal care. Arch Intern Med 1999; 159:1133–1138.
- Ramsaroop SD, Reid MC, Adelman RD. Completing an advance directive in the primary care setting: what do we need for success? J Am Geriatr Soc 2007; 55:277–283.
- Lynn J. Living long in fragile health: the new demographics shape end of life care. In:Jennings B, Kaebnick G, Murray T, editors. Improving End of Life Care: Why Has It Been So Difficult. Hastings Center Report November–December 2005: Special No:S14–S18.
- Beaumont JG, Kenealy PM. Incidence and prevalence of the vegetative and minimally conscious states. Neuropsychol Rehabil 2005; 15:184–189.
- Hawkins NA, Ditto PH, Danks JH, Smucker WD. Micromanaging death: process p, values, and goals in end-of-life medical decision making. Gerontologist 2005; 45:107–117.
- Ditto PH, Jacobson JA, Smucker WD, Danks JH, Fagerlin A. Context changes choices: a prospective study of the effects of hospitalization on life-sustaining treatment p. Med Decis Making 2006; 26:313–322.
- Lockhart LK, Ditto PH, Danks JH, Coppola KM, Smucker WD. The stability of older adults’ judgments of fates better and worse than death. Death Stud 2001; 25:299–317.
- Gjerdingen DK, Neff JA, Wang M, Chaloner K. Older persons’ opinions about life-sustaining procedures in the face of dementia. Arch Fam Med 1999; 8:421–425.
- Heap MJ, Munglani R, Klinck JR, Males AG. Elderly patients’ p concerning life-support treatment. Anaesthesia 1993; 48:1027–1033.
- Singer PA, Thiel EC, Naylor CD, et al. Life-sustaining treatment p of hemodialysis patients: implications for advance directives. J Am Soc Nephrol 1995; 6:1410–1417.
- Hofmann JC, Wenger NS, Davis RB, et al. Patient p for communication with physicians about end-of-life decisions. SUPPORT Investigators. Study to Understand Prognoses and Preference for Outcomes and Risks of Treatment. Ann Intern Med 1997; 127:1–12.
- Pinquart M, Sorensen S. Helping caregivers of persons with dementia: which interventions work and how large are their effects? Int Psychogeriatr 2006; 18:577–595.
- Robbins J, Gensler G, Hind J, et al. Comparison of 2 Interventions for liquid aspiration on pneumonia incidence: a randomized trial. Ann Intern Med 2008; 148:509–518.
- Walter LC, Covinsky KE. Cancer screening in elderly patients: a framework for individualized decision making. JAMA 2001; 285:2750–2756.
- Lee SJ, Lindquist K, Segal MR, Covinsky KE. Development and validation of a prognostic index for 4-year mortality in older adults. JAMA 2006; 295:801–808.
- Losey R, Messinger-Rapport BJ. At what age should we discontinue colon cancer screening in the elderly? Cleve Clin J Med 2007; 74:269–272.
- Larson EB, Shadlen MF, Wang L, et al. Survival after initial diagnosis of Alzheimer disease. Ann Intern Med 2004; 140:501–509.
- Suthers K, Kim JK, Crimmins E. Life expectancy with cognitive impairment in the older population of the United States. J Gerontol B Psychol Sci Soc Sci 2003; 58:S179–S186.
- Bloom HL, Shukrullah I, Cuellar JR, Lloyd MS, Dudley SC, Zafari AM. Long-term survival after successful inhospital cardiac arrest resuscitation. Am Heart J 2007; 153:831–836.
- Finucane TE, Harper GM. Attempting resuscitation in nursing homes: policy considerations. J Am Geriatr Soc 1999; 47:1261–1264.
- Pearlman R, Startks H, Cain K, Cole W, Rosengren D, Patrick D. Your Life, Your Choices. 2nd ed. Department of Veterans Affairs, National Center for Ethics in Health Care, 2007.
- Molloy DW. Let Me Decide. Hamilton, Ontario: Newgrange Press, 1996.
- Dunn PM, Schmidt TA, Carley MM, Donius M, Weinstein MA, Dull VT. A method to communicate patient p about medically indicated life-sustaining treatment in the out-of-hospital setting. J Am Geriatr Soc 1996; 44:785–791.
- Fried TR, O’Leary J, Van Ness P, Fraenkel L. Inconsistency over time in the p of older persons with advanced illness for life-sustaining treatment. J Am Geriatr Soc 2007; 55:1007–1014.
- Fried TR, Van Ness PH, Byers AL, Towle VR, O’Leary JR, Dubin JA. Changes in p for life-sustaining treatment among older persons with advanced illness. J Gen Intern Med 2007; 22:495–501.
- Diwan S, Hougham GW, Sachs GA. Strain experienced by caregivers of dementia patients receiving palliative care: findings from the Palliative Excellence in Alzheimer Care Efforts (PEACE) Program. J Palliat Med 2004; 7:797–807.
- Covinsky KE, Yaffe K. Dementia, prognosis, and the needs of patients and caregivers. Ann Intern Med 2004; 140:573–574.
- Shega JW, Levin A, Hougham GW, et al. Palliative Excellence in Alzheimer Care Efforts (PEACE): a program description. J Palliat Med 2003; 6:315–320.
- Center for Ethics in Health Care. Physician orders for life-sustaining treatment paradigm. www.ohsu.edu/ethics/polst/. Accessed March 9, 2009.
- Lee MA, Brummel-Smith K, Meyer J, Drew N, London MR. Physician orders for life-sustaining treatment (POLST): outcomes in a PACE program. Program of All-Inclusive Care for the Elderly. J Am Geriatr Soc 2000; 48:1219–1225.
- Meyers JL, Moore C, McGrory A, Sparr J, Ahern M. Physician orders for life-sustaining treatment form: honoring end-of-life directives for nursing home residents. J Gerontol Nurs 2004; 30:37–46.
- Tolle SW, Tilden VP, Nelson CA, Dunn PM. A prospective study of the efficacy of the physician order form for life-sustaining treatment. J Am Geriatr Soc 1998; 46:1097–1102.
- Cantor MD, Pearlman RA. Advance care planning in long-term care facilities. J Am Med Dir Assoc 2004; 5(suppl 2):S72–S80.
Mr. B., an 82-year-old retired accountant with hypertension, was diagnosed with early Alzheimer disease 6 years ago. He now needs supervision with bathing and dressing and no longer consistently recognizes family members. You are seeing him in the office today after a hospitalization for aspiration pneumonia, his second in the past 6 months.
In the hospital, a brain scan showed that atrophy had progressed and white-matter disease was more extensive than 3 years earlier. A barium swallow study showed esophageal dysmotility and aspiration. He was prescribed a “dysphagia diet,”1 which he dislikes.
Since returning home, he has been disoriented, he has been wandering about the house, and he has fallen several times. He has lost 10 pounds in 6 months. Because of his confusion, his wife cannot take him out, and she is exhausted caring for him.
Reviewing his medical record, you note that 10 years ago, Mr. B. completed a living will and designated his wife as his proxy decision-maker via a medical power of attorney document.
PLANNING IS OFTEN NEGLECTED
Many clinicians and older patients feel a strong need to document, in advance, the patient’s wishes regarding medical care in the event the patient becomes seriously ill and unable to participate in treatment decisions. Professional societies such as the American Geriatrics Society promote advance care planning,2 and some indices of the quality of medical care include whether advance directives have been discussed and completed.3
Yet, despite the high profile of advance care planning,4 few patients actually fill out advance directives,5 with completion rates that vary widely,6–8 sometimes by ethnicity and sex.9,10 Furthermore, in a crisis, these directives are seldom followed.11
In this paper, we recommend an approach to advance care planning for older adults that redirects the focus from “signing away” interventions such as dialysis, mechanical ventilation, and tube feeding. Instead, the focus is on the goals of care. We also advocate naming a surrogate decision-maker, since the medical power of attorney is more flexible and more widely applicable than the living will.
START BY LISTENING
A change in function resulting from disease progression, hospitalization, trauma, or other reasons is an ideal opportunity to introduce the process of advance care planning.
The first step is to find out how well the patient and family understand the patient’s relevant medical conditions, and what their expectations, hopes, and concerns are. This listening phase can provide insight into the patient’s values and goals and how much the patient and family want to engage in these discussions.
In matters of health behavior (such as advance care planning), people change only when they are ready to change.12,13 Thus, we advise physicians to defer extensive discussions of values and goals of care until patients and families are ready to listen, hear, and talk about these topics (often, after a change for the worse in prognosis).
And it is a process. Advance care directives are most likely to be set up and followed if the patient and doctor discuss this issue during multiple visits, rather than if the physician merely hands the patient a packet of forms and information.14–16
CASE CONTINUED: A PEG TUBE REFUSED
Mrs. B. says that Mr. B. is in good health except for his memory: he does not have a serious condition such as diabetes, heart failure, or cancer. While Mr. B. was in the hospital, the hospitalist recommended placing a percutaneous endoscopic gastrostomy (PEG) tube, but Mrs. B. declined the recommendation because her husband had a living will that specified “no artificially or technologically supplied nutrition or hydration.”
At this point, Mrs. B. begins to cry. She has slept poorly because of his wandering. Also, her two daughters do not support her refusal of the PEG tube.
Comment. This brief conversation illuminates knowledge deficits in Mrs. B.’s understanding of Alzheimer disease and the circumstances in which the living will applies. Although one could argue that Mr. B.’s Alzheimer disease has advanced to the point that he is likely to die of a complication of that condition, he is not likely to die in the near future. If he is not considered by law and his physician to be terminally ill or permanently unconscious, a living will likely does not offer guidance about artificial feeding.
LIMITATIONS OF A LIVING WILL
A living will, a commonly used advance directive, states that the patient does not wish to receive life-sustaining treatment in the event that he or she suffers an incurable, irreversible disease and cannot give informed consent, and it often lists specific treatments that the patient does not want. However, we believe that approaching the patient with a list of life-sustaining measures to accept or reject, before discussing goals of care and prognosis, puts the cart before the horse. This approach threatens to distract from the need to ascertain values and offer appropriate care. Additionally, a living will is active only within a very limited scenario and does not address relatively routine but important decisions in a person’s care.
All ‘terminal illness’ isn’t the same
A living will goes into effect only if the patient either enters a permanent vegetative state following an event such as cardiac arrest or severe brain trauma, or is diagnosed with a terminal illness such as metastatic cancer, and lacks decision-making capacity.
But what is terminal illness? The definition differs from state to state, but it is generally defined as an irreversible condition leading to death in a relatively short time. The time may not be specified, as in Florida statute 765. In contrast, Ohio Revised Code 2133.01(AA) uses the phrase “relatively short,” while other states specify a time, such as within 6 months (Texas Health and Safety Code 166.002). The Medicare hospice benefit also carries a short time limit, usually less than 6 months.
The middle panel in Figure 1 depicts a more typical decline from serial organ insults such as stroke followed by infection from aspiration or followed by falls. Older adults with dementia or with multiple progressive diseases such as heart failure, diabetes, hypertension, or cancer decline in a trajectory such as in the bottom panel of Figure 1.
A living will might not be activated in the latter two scenarios until years into the condition because the patient would not be considered terminally ill—by state law, by the health care provider, or even by the patient.
The living will does not address routine interventions
In most states, living wills address only life-sustaining treatments such as dialysis, mechanical ventilation, and medically supplied nutrition and hydration. Living wills do not address relatively common diseases in older adults that could cause severe debility, such as a major stroke or advanced dementia.
If an older patient has dementia, a living will is unlikely to provide guidance about interventions such as intubation to get through an episode of potentially reversible respiratory failure, a feeding tube to correct weight loss, or cardiac catheterization or bypass surgery to treat angina. Yet these important decisions often arise as function declines and comorbidities progress.
Patients may change their minds
Many older adults are reluctant to sign documents to “micromanage” their future care if they should become ill.19 Many people change their mind as the situation changes.11,20,21 Although few claim they would want burdensome interventions if they had dementia22 or if their prognosis were poor,23 patients may tolerate more burdensome interventions if they are already receiving treatments for chronic illnesses such as end-stage renal disease.24
Thus, a living will may help if unexpected trauma occurs in a healthy person, but not so much if chronic illness progresses over a period of years.
Advance directives may not be honored
Even if completed, written advance directives may not be followed, for a variety of reasons. Physicians may not know the patient has a living will, and fewer than one-third of people who actually complete an advance directive have discussed its content with a physician.25 The people named as surrogate decision-makers may not know the patient’s wishes. Family members may disagree with the goals and plan of care and may interfere with implementation of the advance care plan. A patient may see multiple physicians at different institutions who may not communicate with each other about the patient’s wishes. Also, physicians and patients may interpret terms such as “terminally ill” differently, making it difficult to translate the documents into an action plan.
CASE CONTINUED: RELIEVING CAREGIVER STRESS
Returning to Mr. B., your first goal is to address care issues, including caregiver stress. Skilled services in the home are appropriate for him at this time (and Medicare will pay for them) because he is still homebound. These services could include physical therapy, occupational therapy, and speech (swallowing) therapy. A home care agency may also provide an aide for a few weeks to assist with bathing and other personal needs.
You strongly recommend that the family (including both daughters) participate in the Alzheimer’s Association educational programs. You recommend that Mrs. B. locate an adult day care program now so that when Mr. B. completes his home therapy course and is no longer homebound, he may attend. Day care would provide a therapeutic environment for him and respite for her.
You request that the home care agency provide a social worker to advise her on community resources. Meta-analysis suggests that structured, multicomponent interventions with caregivers of demented patients reduce several types of caregiver burden and delay institutionalization.26
He improves with conservative measures
Three weeks later, Mr. B. is sleeping better and has stopped wandering. However, he dislikes the thickened liquids required by the dysphagia diet and has lost another 2 pounds. If his beverages are not thickened, he coughs profusely when he swallows. His daughters are still pressuring Mrs. B. for a PEG tube; one of them has angrily asserted that the doctors are going to allow her father to die.
You explain the burdens of PEG tubes: surgical risks, continued aspiration, disrupted bowel habits, the risk of the tube being accidentally or intentionally dislodged by the patient, and special binders (which may be uncomfortable) or restraints (which may cause further functional decline) that may be necessary to prevent this complication.
You request that the speech therapist work with the patient more aggressively in the use of swallowing techniques such as the chin tuck, which may be at least as effective as thickeners in preventing both aspiration pneumonia and dehydration.27 The therapist will need to include Mrs. B. in these sessions, since she will be Mr. B.’s coach at mealtime.
With more aggressive speech therapy, the patient’s weight stabilizes over the next 4 weeks. He is in day care 3 days a week, and Mrs. B. is more rested and relaxed.
Cardiopulmonary resuscitation
You continue the advance care planning discussion and suggest that if Mr. B. aspirates, is hospitalized again, and declines further care, it would be helpful to delineate instructions for resuscitation. Right now, although his Alzheimer disease is advanced, he is not clearly terminal. Thus, his living will does not strictly apply and provides limited guidance about intubation, cardiopulmonary resuscitation (CPR), or medically supplied nutrition and hydration. However, because Mrs. B. is his agent in the medical power of attorney, this document enables her to make a wide spectrum of treatment decisions on his behalf.
Mrs. B. asks about her husband’s prognosis and why CPR would not be helpful.
Comment. Further discussion with her could be guided by an estimate of Mr. B.’s prognosis. Function-based tools28,29 may also be useful. For example,28,30 an 80-year-old man with high functional status might have a life expectancy of more than 10 years. Mr. B., with multiple medical problems and declining function, would have an estimated life expectancy of approximately 3 years. Even without specifically categorizing function, impaired cognition by itself predicted a shorter life expectancy in population-based studies.31,32
Regarding CPR, patients and families may overestimate successful outcomes. A recent study33 of 10 years of outcomes of in-hospital cardiac arrest found that only 6.6% of patients survived to discharge. The average age of the survivors was 59 years, and fewer than half of them survived 3 years after cardiac arrest. In eight studies of CPR outcomes in nursing homes,34 three studies had no survivors, and all but one study had a survival rate below 5%.
PROVIDING APPROPRIATE CARE, NOT LIMITING TREATMENTS
In the case of Mr. B., as in many situations encountered with older patients, written advance directives provide little help or guidance. Instead, we recommend a model of advance care planning that takes place during multiple office visits over time, and that maintains a focus on providing appropriate care rather than on limiting life-sustaining treatments. We recommend providing estimates of prognosis and CPR outcomes when the family appeares ready to hear them. This approach should result in a care-oriented process while moving the family towards decisions regarding artificial feeding and CPR.
All patients, particularly those unwilling or unable to participate in advance care planning, are encouraged to identify one or more surrogate decision-makers and articulate how much flexibility that person should be given in important health care decisions. The medical power of attorney can be activated any time the patient lacks decision-making capacity and deactivated when decision-making capacity returns.38,39
As in the case of Mr. B., a tailored approach to advance care planning requires clinicians to estimate life expectancy (more than 5 years, less than 5 years, or less than 1 year) and to determine the patient’s and the family’s readiness to focus on a values-oriented and goal-oriented care plan. Some patients are not receptive to advance care planning, and clinical time and effort are optimized by providing the right amount of information to patients when they are ready to receive it.
For relatively healthy older adults
Figure 2 is the algorithm for older patients who are expected to live at least 5 years, ie, who are relatively healthy and functional. Patients with little or no interest in advance care planning can be asked about it annually, or sooner if their medical condition changes. Patients with limited interest can be given written information, specifically living will and medical power of attorney documents recognized in their state. Patients more open to advance care planning can be offered a values history form (Table 1), Web sites, and educational materials, with a plan to discuss them at future appointments.
Periodic reevaluation of values and goals of care is important. Patients may assert that particular interventions (eg, a PEG tube or dialysis) are “worse than death” when they are healthy, but they may change their views over time.21 Additionally, although a recent hospitalization or a decline in function may predispose patients to want to limit life-sustaining treatments, they may return to their earlier values and wishes a few months later, particularly if their medical condition stabilizes. 20 Values and decisions should be reassessed not only when medical conditions deteriorate, but also when they improve.
For chronically ill patients
Caregiver stress is important to identify and address, since caregivers often neglect themselves.40–42
For terminally ill patients
In this situation, patients and families need information about community resources that can assist them in the home. Some older adults with cognitive impairment may be exploited or neglect themselves, and referral to an adult protective services agency may be needed.
Treatment burden, particularly due to multiple prescribed medications, may be high and should be reassessed in light of the goals of treatment. Polypharmacy reduction is especially important at this stage in the illness, since the goals of care may be different than when the medications were prescribed.
Physical or psychosocial symptoms may be the cue to bring up the topic of palliative care. If the patient is expected to live less than 6 months, hospice referral is appropriate. With either palliative care or hospice, the focus of attention shifts explicitly from curing the disease to managing symptoms, and from the patient to the patient-family dyad. Interventions such as CPR and ventilatory support should be discussed and information from Table 2 provided to the patient and family.
Complete advance care planning incorporates taking a values history, estimating life expectancy, determining physical, psychosocial and spiritual needs, clarifying treatment goals, and estimating manageable treatment burden. Offering statistics on CPR and providing state-specific living will and medical power of attorney documents are important but are only one facet of effective advance care planning. In fact, shifting the emphasis of advance care planning from statistics and forms to values and goals of care may help in developing a more comprehensive care plan.
Goals of care range from curing the disease (with aggressive therapy, which may be burdensome) to simply improving function or decreasing pain. In the latter case, one may be able to discontinue some of the patient’s drugs, utilize medical and community resources more effectively, and better meet the patient’s needs.
Woven through all these discussions should be reassurance that the plan can be revisited and possibly revised, and that the physician will be there to help with those decisions.
For acutely ill patients in the hospital
Episodic, staged advance care planning is appropriate not only in the office but also in other settings such as assisted living and nursing facilities.
In the hospital, however, a different approach is needed, since patients are usually admitted because of an acute illness or sudden functional decline, or both. Decisions about technological interventions such as CPR, mechanical ventilation, or dialysis may be needed urgently. Often, patients are unable to provide guidance to physicians during acute illness because of delirium and other impediments. Developing a plan for care in the hospital may require urgent family meetings. However, if a surrogate decision-maker is in place, and if the patient has already participated in some form of advance care planning as an outpatient, the values and goals of care previously identified can contribute to decision-making during hospitalization.
As mentioned above, fragmentation of health care across providers and health care systems may limit the effectiveness of office-based advance care planning. It may be reasonable to train office staff to place advance care planning documentation in easily accessible sections of the patient’s medical record and to forward these to specialists involved in a patient’s care.
The patient and family should be encouraged and empowered to help with this process and should have updated advance care planning documentation readily available. In some states, comprehensive medical order sets, especially for end-of-life care, are portable across care settings and address CPR, medically supplied nutrition, hospital transfer, and antibiotic treatment.43
Research suggests that health care systems are more likely to comply with patients’ end-of-life preferences when portable medical order forms are developed and disseminated.44–44 Ultimately, major changes in health care delivery, including universal electronic health records, may be needed to implement and communicate patients’ advance care planning preferences across settings.
Mr. B., an 82-year-old retired accountant with hypertension, was diagnosed with early Alzheimer disease 6 years ago. He now needs supervision with bathing and dressing and no longer consistently recognizes family members. You are seeing him in the office today after a hospitalization for aspiration pneumonia, his second in the past 6 months.
In the hospital, a brain scan showed that atrophy had progressed and white-matter disease was more extensive than 3 years earlier. A barium swallow study showed esophageal dysmotility and aspiration. He was prescribed a “dysphagia diet,”1 which he dislikes.
Since returning home, he has been disoriented, he has been wandering about the house, and he has fallen several times. He has lost 10 pounds in 6 months. Because of his confusion, his wife cannot take him out, and she is exhausted caring for him.
Reviewing his medical record, you note that 10 years ago, Mr. B. completed a living will and designated his wife as his proxy decision-maker via a medical power of attorney document.
PLANNING IS OFTEN NEGLECTED
Many clinicians and older patients feel a strong need to document, in advance, the patient’s wishes regarding medical care in the event the patient becomes seriously ill and unable to participate in treatment decisions. Professional societies such as the American Geriatrics Society promote advance care planning,2 and some indices of the quality of medical care include whether advance directives have been discussed and completed.3
Yet, despite the high profile of advance care planning,4 few patients actually fill out advance directives,5 with completion rates that vary widely,6–8 sometimes by ethnicity and sex.9,10 Furthermore, in a crisis, these directives are seldom followed.11
In this paper, we recommend an approach to advance care planning for older adults that redirects the focus from “signing away” interventions such as dialysis, mechanical ventilation, and tube feeding. Instead, the focus is on the goals of care. We also advocate naming a surrogate decision-maker, since the medical power of attorney is more flexible and more widely applicable than the living will.
START BY LISTENING
A change in function resulting from disease progression, hospitalization, trauma, or other reasons is an ideal opportunity to introduce the process of advance care planning.
The first step is to find out how well the patient and family understand the patient’s relevant medical conditions, and what their expectations, hopes, and concerns are. This listening phase can provide insight into the patient’s values and goals and how much the patient and family want to engage in these discussions.
In matters of health behavior (such as advance care planning), people change only when they are ready to change.12,13 Thus, we advise physicians to defer extensive discussions of values and goals of care until patients and families are ready to listen, hear, and talk about these topics (often, after a change for the worse in prognosis).
And it is a process. Advance care directives are most likely to be set up and followed if the patient and doctor discuss this issue during multiple visits, rather than if the physician merely hands the patient a packet of forms and information.14–16
CASE CONTINUED: A PEG TUBE REFUSED
Mrs. B. says that Mr. B. is in good health except for his memory: he does not have a serious condition such as diabetes, heart failure, or cancer. While Mr. B. was in the hospital, the hospitalist recommended placing a percutaneous endoscopic gastrostomy (PEG) tube, but Mrs. B. declined the recommendation because her husband had a living will that specified “no artificially or technologically supplied nutrition or hydration.”
At this point, Mrs. B. begins to cry. She has slept poorly because of his wandering. Also, her two daughters do not support her refusal of the PEG tube.
Comment. This brief conversation illuminates knowledge deficits in Mrs. B.’s understanding of Alzheimer disease and the circumstances in which the living will applies. Although one could argue that Mr. B.’s Alzheimer disease has advanced to the point that he is likely to die of a complication of that condition, he is not likely to die in the near future. If he is not considered by law and his physician to be terminally ill or permanently unconscious, a living will likely does not offer guidance about artificial feeding.
LIMITATIONS OF A LIVING WILL
A living will, a commonly used advance directive, states that the patient does not wish to receive life-sustaining treatment in the event that he or she suffers an incurable, irreversible disease and cannot give informed consent, and it often lists specific treatments that the patient does not want. However, we believe that approaching the patient with a list of life-sustaining measures to accept or reject, before discussing goals of care and prognosis, puts the cart before the horse. This approach threatens to distract from the need to ascertain values and offer appropriate care. Additionally, a living will is active only within a very limited scenario and does not address relatively routine but important decisions in a person’s care.
All ‘terminal illness’ isn’t the same
A living will goes into effect only if the patient either enters a permanent vegetative state following an event such as cardiac arrest or severe brain trauma, or is diagnosed with a terminal illness such as metastatic cancer, and lacks decision-making capacity.
But what is terminal illness? The definition differs from state to state, but it is generally defined as an irreversible condition leading to death in a relatively short time. The time may not be specified, as in Florida statute 765. In contrast, Ohio Revised Code 2133.01(AA) uses the phrase “relatively short,” while other states specify a time, such as within 6 months (Texas Health and Safety Code 166.002). The Medicare hospice benefit also carries a short time limit, usually less than 6 months.
The middle panel in Figure 1 depicts a more typical decline from serial organ insults such as stroke followed by infection from aspiration or followed by falls. Older adults with dementia or with multiple progressive diseases such as heart failure, diabetes, hypertension, or cancer decline in a trajectory such as in the bottom panel of Figure 1.
A living will might not be activated in the latter two scenarios until years into the condition because the patient would not be considered terminally ill—by state law, by the health care provider, or even by the patient.
The living will does not address routine interventions
In most states, living wills address only life-sustaining treatments such as dialysis, mechanical ventilation, and medically supplied nutrition and hydration. Living wills do not address relatively common diseases in older adults that could cause severe debility, such as a major stroke or advanced dementia.
If an older patient has dementia, a living will is unlikely to provide guidance about interventions such as intubation to get through an episode of potentially reversible respiratory failure, a feeding tube to correct weight loss, or cardiac catheterization or bypass surgery to treat angina. Yet these important decisions often arise as function declines and comorbidities progress.
Patients may change their minds
Many older adults are reluctant to sign documents to “micromanage” their future care if they should become ill.19 Many people change their mind as the situation changes.11,20,21 Although few claim they would want burdensome interventions if they had dementia22 or if their prognosis were poor,23 patients may tolerate more burdensome interventions if they are already receiving treatments for chronic illnesses such as end-stage renal disease.24
Thus, a living will may help if unexpected trauma occurs in a healthy person, but not so much if chronic illness progresses over a period of years.
Advance directives may not be honored
Even if completed, written advance directives may not be followed, for a variety of reasons. Physicians may not know the patient has a living will, and fewer than one-third of people who actually complete an advance directive have discussed its content with a physician.25 The people named as surrogate decision-makers may not know the patient’s wishes. Family members may disagree with the goals and plan of care and may interfere with implementation of the advance care plan. A patient may see multiple physicians at different institutions who may not communicate with each other about the patient’s wishes. Also, physicians and patients may interpret terms such as “terminally ill” differently, making it difficult to translate the documents into an action plan.
CASE CONTINUED: RELIEVING CAREGIVER STRESS
Returning to Mr. B., your first goal is to address care issues, including caregiver stress. Skilled services in the home are appropriate for him at this time (and Medicare will pay for them) because he is still homebound. These services could include physical therapy, occupational therapy, and speech (swallowing) therapy. A home care agency may also provide an aide for a few weeks to assist with bathing and other personal needs.
You strongly recommend that the family (including both daughters) participate in the Alzheimer’s Association educational programs. You recommend that Mrs. B. locate an adult day care program now so that when Mr. B. completes his home therapy course and is no longer homebound, he may attend. Day care would provide a therapeutic environment for him and respite for her.
You request that the home care agency provide a social worker to advise her on community resources. Meta-analysis suggests that structured, multicomponent interventions with caregivers of demented patients reduce several types of caregiver burden and delay institutionalization.26
He improves with conservative measures
Three weeks later, Mr. B. is sleeping better and has stopped wandering. However, he dislikes the thickened liquids required by the dysphagia diet and has lost another 2 pounds. If his beverages are not thickened, he coughs profusely when he swallows. His daughters are still pressuring Mrs. B. for a PEG tube; one of them has angrily asserted that the doctors are going to allow her father to die.
You explain the burdens of PEG tubes: surgical risks, continued aspiration, disrupted bowel habits, the risk of the tube being accidentally or intentionally dislodged by the patient, and special binders (which may be uncomfortable) or restraints (which may cause further functional decline) that may be necessary to prevent this complication.
You request that the speech therapist work with the patient more aggressively in the use of swallowing techniques such as the chin tuck, which may be at least as effective as thickeners in preventing both aspiration pneumonia and dehydration.27 The therapist will need to include Mrs. B. in these sessions, since she will be Mr. B.’s coach at mealtime.
With more aggressive speech therapy, the patient’s weight stabilizes over the next 4 weeks. He is in day care 3 days a week, and Mrs. B. is more rested and relaxed.
Cardiopulmonary resuscitation
You continue the advance care planning discussion and suggest that if Mr. B. aspirates, is hospitalized again, and declines further care, it would be helpful to delineate instructions for resuscitation. Right now, although his Alzheimer disease is advanced, he is not clearly terminal. Thus, his living will does not strictly apply and provides limited guidance about intubation, cardiopulmonary resuscitation (CPR), or medically supplied nutrition and hydration. However, because Mrs. B. is his agent in the medical power of attorney, this document enables her to make a wide spectrum of treatment decisions on his behalf.
Mrs. B. asks about her husband’s prognosis and why CPR would not be helpful.
Comment. Further discussion with her could be guided by an estimate of Mr. B.’s prognosis. Function-based tools28,29 may also be useful. For example,28,30 an 80-year-old man with high functional status might have a life expectancy of more than 10 years. Mr. B., with multiple medical problems and declining function, would have an estimated life expectancy of approximately 3 years. Even without specifically categorizing function, impaired cognition by itself predicted a shorter life expectancy in population-based studies.31,32
Regarding CPR, patients and families may overestimate successful outcomes. A recent study33 of 10 years of outcomes of in-hospital cardiac arrest found that only 6.6% of patients survived to discharge. The average age of the survivors was 59 years, and fewer than half of them survived 3 years after cardiac arrest. In eight studies of CPR outcomes in nursing homes,34 three studies had no survivors, and all but one study had a survival rate below 5%.
PROVIDING APPROPRIATE CARE, NOT LIMITING TREATMENTS
In the case of Mr. B., as in many situations encountered with older patients, written advance directives provide little help or guidance. Instead, we recommend a model of advance care planning that takes place during multiple office visits over time, and that maintains a focus on providing appropriate care rather than on limiting life-sustaining treatments. We recommend providing estimates of prognosis and CPR outcomes when the family appeares ready to hear them. This approach should result in a care-oriented process while moving the family towards decisions regarding artificial feeding and CPR.
All patients, particularly those unwilling or unable to participate in advance care planning, are encouraged to identify one or more surrogate decision-makers and articulate how much flexibility that person should be given in important health care decisions. The medical power of attorney can be activated any time the patient lacks decision-making capacity and deactivated when decision-making capacity returns.38,39
As in the case of Mr. B., a tailored approach to advance care planning requires clinicians to estimate life expectancy (more than 5 years, less than 5 years, or less than 1 year) and to determine the patient’s and the family’s readiness to focus on a values-oriented and goal-oriented care plan. Some patients are not receptive to advance care planning, and clinical time and effort are optimized by providing the right amount of information to patients when they are ready to receive it.
For relatively healthy older adults
Figure 2 is the algorithm for older patients who are expected to live at least 5 years, ie, who are relatively healthy and functional. Patients with little or no interest in advance care planning can be asked about it annually, or sooner if their medical condition changes. Patients with limited interest can be given written information, specifically living will and medical power of attorney documents recognized in their state. Patients more open to advance care planning can be offered a values history form (Table 1), Web sites, and educational materials, with a plan to discuss them at future appointments.
Periodic reevaluation of values and goals of care is important. Patients may assert that particular interventions (eg, a PEG tube or dialysis) are “worse than death” when they are healthy, but they may change their views over time.21 Additionally, although a recent hospitalization or a decline in function may predispose patients to want to limit life-sustaining treatments, they may return to their earlier values and wishes a few months later, particularly if their medical condition stabilizes. 20 Values and decisions should be reassessed not only when medical conditions deteriorate, but also when they improve.
For chronically ill patients
Caregiver stress is important to identify and address, since caregivers often neglect themselves.40–42
For terminally ill patients
In this situation, patients and families need information about community resources that can assist them in the home. Some older adults with cognitive impairment may be exploited or neglect themselves, and referral to an adult protective services agency may be needed.
Treatment burden, particularly due to multiple prescribed medications, may be high and should be reassessed in light of the goals of treatment. Polypharmacy reduction is especially important at this stage in the illness, since the goals of care may be different than when the medications were prescribed.
Physical or psychosocial symptoms may be the cue to bring up the topic of palliative care. If the patient is expected to live less than 6 months, hospice referral is appropriate. With either palliative care or hospice, the focus of attention shifts explicitly from curing the disease to managing symptoms, and from the patient to the patient-family dyad. Interventions such as CPR and ventilatory support should be discussed and information from Table 2 provided to the patient and family.
Complete advance care planning incorporates taking a values history, estimating life expectancy, determining physical, psychosocial and spiritual needs, clarifying treatment goals, and estimating manageable treatment burden. Offering statistics on CPR and providing state-specific living will and medical power of attorney documents are important but are only one facet of effective advance care planning. In fact, shifting the emphasis of advance care planning from statistics and forms to values and goals of care may help in developing a more comprehensive care plan.
Goals of care range from curing the disease (with aggressive therapy, which may be burdensome) to simply improving function or decreasing pain. In the latter case, one may be able to discontinue some of the patient’s drugs, utilize medical and community resources more effectively, and better meet the patient’s needs.
Woven through all these discussions should be reassurance that the plan can be revisited and possibly revised, and that the physician will be there to help with those decisions.
For acutely ill patients in the hospital
Episodic, staged advance care planning is appropriate not only in the office but also in other settings such as assisted living and nursing facilities.
In the hospital, however, a different approach is needed, since patients are usually admitted because of an acute illness or sudden functional decline, or both. Decisions about technological interventions such as CPR, mechanical ventilation, or dialysis may be needed urgently. Often, patients are unable to provide guidance to physicians during acute illness because of delirium and other impediments. Developing a plan for care in the hospital may require urgent family meetings. However, if a surrogate decision-maker is in place, and if the patient has already participated in some form of advance care planning as an outpatient, the values and goals of care previously identified can contribute to decision-making during hospitalization.
As mentioned above, fragmentation of health care across providers and health care systems may limit the effectiveness of office-based advance care planning. It may be reasonable to train office staff to place advance care planning documentation in easily accessible sections of the patient’s medical record and to forward these to specialists involved in a patient’s care.
The patient and family should be encouraged and empowered to help with this process and should have updated advance care planning documentation readily available. In some states, comprehensive medical order sets, especially for end-of-life care, are portable across care settings and address CPR, medically supplied nutrition, hospital transfer, and antibiotic treatment.43
Research suggests that health care systems are more likely to comply with patients’ end-of-life preferences when portable medical order forms are developed and disseminated.44–44 Ultimately, major changes in health care delivery, including universal electronic health records, may be needed to implement and communicate patients’ advance care planning preferences across settings.
- National Dysphagia Diet Task Force. National Dysphagia Diet: Standardization for Optimal Care. Chicago, IL: American Dietetic Association, 2002.
- Nusbaum N, Goldstein M. American Geriatrics Society. The Patient Education Forum. Advance Directives, 2008. www.americangeriatrics.org/education/forum/advance_dir.shtml. Accessed March 9, 2009.
- Wenger NS, Roth CP, Shekelle PA; COVE Investigators. Introduction to the assessing care of vulnerable elders–3 quality indicator measurement set. J Am Geriatr Soc 2007; 55(suppl 2):S247–S252.
- Emanuel LL, Danis M, Pearlman RA, Singer PA. Advance care planning as a process: structuring the discussions in practice. J Am Geriatr Soc 1995; 43:440–446.
- Teno J, Lynn J, Wenger N, et al. Advance directives for seriously ill hospitalized patients: effectiveness with the patient self-determination act and the SUPPORT intervention. SUPPORT Investigators. Study to Understand Prognoses and P for Outcomes and Risks of Treatment. J Am Geriatr Soc 1997; 45:500–507.
- Hammes BJ, Rooney BL. Death and end-of-life planning in one midwestern community. Arch Intern Med 1998; 158:383–390.
- Gordon NP, Shade SB. Advance directives are more likely among seniors asked about end-of-life care p. Arch Intern Med 1999; 159:701–704.
- Morrison RS, Meier DE. High rates of advance care planning in New York City’s elderly population. Arch Intern Med 2004; 164:2421–2426.
- Perkins HS, Geppert CMA, Gonzales A, Cortez JD, Hazuda HP. Cross-cultural similarities and differences in attitudes about advance care planning. J Gen Intern Med 2002; 17:48–57.
- Perkins HS, Cortez JD, Hazuda HP. Advance care planning: does patient gender make a difference? Am J Med Sci 2004; 327:25–32.
- The SUPPORT Principal Investigators. A controlled trial to improve care for seriously ill hospitalized patients. Study to Understand Prognoses and P for Outcomes and Risks of Treatments (SUPPORT). JAMA 1995; 274:1591–1598.
- Prochaska JO, DiClemente CC, Norcross JC. In search of how people change. Applications to addictive behaviors. Am Psychol 1992; 47:1102–1114.
- Nigg CR, Burbank PM, Padula C, et al. Stages of change across ten health risk behaviors for older adults. Gerontologist 1999; 39:473–482.
- Patel RV, Sinuff T, Cook DJ. Influencing advance directive completion rates in non-terminally ill patients: a systematic review. J Crit Care 2004; 19:1–9.
- Hanson LC, Earp JA, Garrett J, Menon M, Danis M. Community physicians who provide terminal care. Arch Intern Med 1999; 159:1133–1138.
- Ramsaroop SD, Reid MC, Adelman RD. Completing an advance directive in the primary care setting: what do we need for success? J Am Geriatr Soc 2007; 55:277–283.
- Lynn J. Living long in fragile health: the new demographics shape end of life care. In:Jennings B, Kaebnick G, Murray T, editors. Improving End of Life Care: Why Has It Been So Difficult. Hastings Center Report November–December 2005: Special No:S14–S18.
- Beaumont JG, Kenealy PM. Incidence and prevalence of the vegetative and minimally conscious states. Neuropsychol Rehabil 2005; 15:184–189.
- Hawkins NA, Ditto PH, Danks JH, Smucker WD. Micromanaging death: process p, values, and goals in end-of-life medical decision making. Gerontologist 2005; 45:107–117.
- Ditto PH, Jacobson JA, Smucker WD, Danks JH, Fagerlin A. Context changes choices: a prospective study of the effects of hospitalization on life-sustaining treatment p. Med Decis Making 2006; 26:313–322.
- Lockhart LK, Ditto PH, Danks JH, Coppola KM, Smucker WD. The stability of older adults’ judgments of fates better and worse than death. Death Stud 2001; 25:299–317.
- Gjerdingen DK, Neff JA, Wang M, Chaloner K. Older persons’ opinions about life-sustaining procedures in the face of dementia. Arch Fam Med 1999; 8:421–425.
- Heap MJ, Munglani R, Klinck JR, Males AG. Elderly patients’ p concerning life-support treatment. Anaesthesia 1993; 48:1027–1033.
- Singer PA, Thiel EC, Naylor CD, et al. Life-sustaining treatment p of hemodialysis patients: implications for advance directives. J Am Soc Nephrol 1995; 6:1410–1417.
- Hofmann JC, Wenger NS, Davis RB, et al. Patient p for communication with physicians about end-of-life decisions. SUPPORT Investigators. Study to Understand Prognoses and Preference for Outcomes and Risks of Treatment. Ann Intern Med 1997; 127:1–12.
- Pinquart M, Sorensen S. Helping caregivers of persons with dementia: which interventions work and how large are their effects? Int Psychogeriatr 2006; 18:577–595.
- Robbins J, Gensler G, Hind J, et al. Comparison of 2 Interventions for liquid aspiration on pneumonia incidence: a randomized trial. Ann Intern Med 2008; 148:509–518.
- Walter LC, Covinsky KE. Cancer screening in elderly patients: a framework for individualized decision making. JAMA 2001; 285:2750–2756.
- Lee SJ, Lindquist K, Segal MR, Covinsky KE. Development and validation of a prognostic index for 4-year mortality in older adults. JAMA 2006; 295:801–808.
- Losey R, Messinger-Rapport BJ. At what age should we discontinue colon cancer screening in the elderly? Cleve Clin J Med 2007; 74:269–272.
- Larson EB, Shadlen MF, Wang L, et al. Survival after initial diagnosis of Alzheimer disease. Ann Intern Med 2004; 140:501–509.
- Suthers K, Kim JK, Crimmins E. Life expectancy with cognitive impairment in the older population of the United States. J Gerontol B Psychol Sci Soc Sci 2003; 58:S179–S186.
- Bloom HL, Shukrullah I, Cuellar JR, Lloyd MS, Dudley SC, Zafari AM. Long-term survival after successful inhospital cardiac arrest resuscitation. Am Heart J 2007; 153:831–836.
- Finucane TE, Harper GM. Attempting resuscitation in nursing homes: policy considerations. J Am Geriatr Soc 1999; 47:1261–1264.
- Pearlman R, Startks H, Cain K, Cole W, Rosengren D, Patrick D. Your Life, Your Choices. 2nd ed. Department of Veterans Affairs, National Center for Ethics in Health Care, 2007.
- Molloy DW. Let Me Decide. Hamilton, Ontario: Newgrange Press, 1996.
- Dunn PM, Schmidt TA, Carley MM, Donius M, Weinstein MA, Dull VT. A method to communicate patient p about medically indicated life-sustaining treatment in the out-of-hospital setting. J Am Geriatr Soc 1996; 44:785–791.
- Fried TR, O’Leary J, Van Ness P, Fraenkel L. Inconsistency over time in the p of older persons with advanced illness for life-sustaining treatment. J Am Geriatr Soc 2007; 55:1007–1014.
- Fried TR, Van Ness PH, Byers AL, Towle VR, O’Leary JR, Dubin JA. Changes in p for life-sustaining treatment among older persons with advanced illness. J Gen Intern Med 2007; 22:495–501.
- Diwan S, Hougham GW, Sachs GA. Strain experienced by caregivers of dementia patients receiving palliative care: findings from the Palliative Excellence in Alzheimer Care Efforts (PEACE) Program. J Palliat Med 2004; 7:797–807.
- Covinsky KE, Yaffe K. Dementia, prognosis, and the needs of patients and caregivers. Ann Intern Med 2004; 140:573–574.
- Shega JW, Levin A, Hougham GW, et al. Palliative Excellence in Alzheimer Care Efforts (PEACE): a program description. J Palliat Med 2003; 6:315–320.
- Center for Ethics in Health Care. Physician orders for life-sustaining treatment paradigm. www.ohsu.edu/ethics/polst/. Accessed March 9, 2009.
- Lee MA, Brummel-Smith K, Meyer J, Drew N, London MR. Physician orders for life-sustaining treatment (POLST): outcomes in a PACE program. Program of All-Inclusive Care for the Elderly. J Am Geriatr Soc 2000; 48:1219–1225.
- Meyers JL, Moore C, McGrory A, Sparr J, Ahern M. Physician orders for life-sustaining treatment form: honoring end-of-life directives for nursing home residents. J Gerontol Nurs 2004; 30:37–46.
- Tolle SW, Tilden VP, Nelson CA, Dunn PM. A prospective study of the efficacy of the physician order form for life-sustaining treatment. J Am Geriatr Soc 1998; 46:1097–1102.
- Cantor MD, Pearlman RA. Advance care planning in long-term care facilities. J Am Med Dir Assoc 2004; 5(suppl 2):S72–S80.
- National Dysphagia Diet Task Force. National Dysphagia Diet: Standardization for Optimal Care. Chicago, IL: American Dietetic Association, 2002.
- Nusbaum N, Goldstein M. American Geriatrics Society. The Patient Education Forum. Advance Directives, 2008. www.americangeriatrics.org/education/forum/advance_dir.shtml. Accessed March 9, 2009.
- Wenger NS, Roth CP, Shekelle PA; COVE Investigators. Introduction to the assessing care of vulnerable elders–3 quality indicator measurement set. J Am Geriatr Soc 2007; 55(suppl 2):S247–S252.
- Emanuel LL, Danis M, Pearlman RA, Singer PA. Advance care planning as a process: structuring the discussions in practice. J Am Geriatr Soc 1995; 43:440–446.
- Teno J, Lynn J, Wenger N, et al. Advance directives for seriously ill hospitalized patients: effectiveness with the patient self-determination act and the SUPPORT intervention. SUPPORT Investigators. Study to Understand Prognoses and P for Outcomes and Risks of Treatment. J Am Geriatr Soc 1997; 45:500–507.
- Hammes BJ, Rooney BL. Death and end-of-life planning in one midwestern community. Arch Intern Med 1998; 158:383–390.
- Gordon NP, Shade SB. Advance directives are more likely among seniors asked about end-of-life care p. Arch Intern Med 1999; 159:701–704.
- Morrison RS, Meier DE. High rates of advance care planning in New York City’s elderly population. Arch Intern Med 2004; 164:2421–2426.
- Perkins HS, Geppert CMA, Gonzales A, Cortez JD, Hazuda HP. Cross-cultural similarities and differences in attitudes about advance care planning. J Gen Intern Med 2002; 17:48–57.
- Perkins HS, Cortez JD, Hazuda HP. Advance care planning: does patient gender make a difference? Am J Med Sci 2004; 327:25–32.
- The SUPPORT Principal Investigators. A controlled trial to improve care for seriously ill hospitalized patients. Study to Understand Prognoses and P for Outcomes and Risks of Treatments (SUPPORT). JAMA 1995; 274:1591–1598.
- Prochaska JO, DiClemente CC, Norcross JC. In search of how people change. Applications to addictive behaviors. Am Psychol 1992; 47:1102–1114.
- Nigg CR, Burbank PM, Padula C, et al. Stages of change across ten health risk behaviors for older adults. Gerontologist 1999; 39:473–482.
- Patel RV, Sinuff T, Cook DJ. Influencing advance directive completion rates in non-terminally ill patients: a systematic review. J Crit Care 2004; 19:1–9.
- Hanson LC, Earp JA, Garrett J, Menon M, Danis M. Community physicians who provide terminal care. Arch Intern Med 1999; 159:1133–1138.
- Ramsaroop SD, Reid MC, Adelman RD. Completing an advance directive in the primary care setting: what do we need for success? J Am Geriatr Soc 2007; 55:277–283.
- Lynn J. Living long in fragile health: the new demographics shape end of life care. In:Jennings B, Kaebnick G, Murray T, editors. Improving End of Life Care: Why Has It Been So Difficult. Hastings Center Report November–December 2005: Special No:S14–S18.
- Beaumont JG, Kenealy PM. Incidence and prevalence of the vegetative and minimally conscious states. Neuropsychol Rehabil 2005; 15:184–189.
- Hawkins NA, Ditto PH, Danks JH, Smucker WD. Micromanaging death: process p, values, and goals in end-of-life medical decision making. Gerontologist 2005; 45:107–117.
- Ditto PH, Jacobson JA, Smucker WD, Danks JH, Fagerlin A. Context changes choices: a prospective study of the effects of hospitalization on life-sustaining treatment p. Med Decis Making 2006; 26:313–322.
- Lockhart LK, Ditto PH, Danks JH, Coppola KM, Smucker WD. The stability of older adults’ judgments of fates better and worse than death. Death Stud 2001; 25:299–317.
- Gjerdingen DK, Neff JA, Wang M, Chaloner K. Older persons’ opinions about life-sustaining procedures in the face of dementia. Arch Fam Med 1999; 8:421–425.
- Heap MJ, Munglani R, Klinck JR, Males AG. Elderly patients’ p concerning life-support treatment. Anaesthesia 1993; 48:1027–1033.
- Singer PA, Thiel EC, Naylor CD, et al. Life-sustaining treatment p of hemodialysis patients: implications for advance directives. J Am Soc Nephrol 1995; 6:1410–1417.
- Hofmann JC, Wenger NS, Davis RB, et al. Patient p for communication with physicians about end-of-life decisions. SUPPORT Investigators. Study to Understand Prognoses and Preference for Outcomes and Risks of Treatment. Ann Intern Med 1997; 127:1–12.
- Pinquart M, Sorensen S. Helping caregivers of persons with dementia: which interventions work and how large are their effects? Int Psychogeriatr 2006; 18:577–595.
- Robbins J, Gensler G, Hind J, et al. Comparison of 2 Interventions for liquid aspiration on pneumonia incidence: a randomized trial. Ann Intern Med 2008; 148:509–518.
- Walter LC, Covinsky KE. Cancer screening in elderly patients: a framework for individualized decision making. JAMA 2001; 285:2750–2756.
- Lee SJ, Lindquist K, Segal MR, Covinsky KE. Development and validation of a prognostic index for 4-year mortality in older adults. JAMA 2006; 295:801–808.
- Losey R, Messinger-Rapport BJ. At what age should we discontinue colon cancer screening in the elderly? Cleve Clin J Med 2007; 74:269–272.
- Larson EB, Shadlen MF, Wang L, et al. Survival after initial diagnosis of Alzheimer disease. Ann Intern Med 2004; 140:501–509.
- Suthers K, Kim JK, Crimmins E. Life expectancy with cognitive impairment in the older population of the United States. J Gerontol B Psychol Sci Soc Sci 2003; 58:S179–S186.
- Bloom HL, Shukrullah I, Cuellar JR, Lloyd MS, Dudley SC, Zafari AM. Long-term survival after successful inhospital cardiac arrest resuscitation. Am Heart J 2007; 153:831–836.
- Finucane TE, Harper GM. Attempting resuscitation in nursing homes: policy considerations. J Am Geriatr Soc 1999; 47:1261–1264.
- Pearlman R, Startks H, Cain K, Cole W, Rosengren D, Patrick D. Your Life, Your Choices. 2nd ed. Department of Veterans Affairs, National Center for Ethics in Health Care, 2007.
- Molloy DW. Let Me Decide. Hamilton, Ontario: Newgrange Press, 1996.
- Dunn PM, Schmidt TA, Carley MM, Donius M, Weinstein MA, Dull VT. A method to communicate patient p about medically indicated life-sustaining treatment in the out-of-hospital setting. J Am Geriatr Soc 1996; 44:785–791.
- Fried TR, O’Leary J, Van Ness P, Fraenkel L. Inconsistency over time in the p of older persons with advanced illness for life-sustaining treatment. J Am Geriatr Soc 2007; 55:1007–1014.
- Fried TR, Van Ness PH, Byers AL, Towle VR, O’Leary JR, Dubin JA. Changes in p for life-sustaining treatment among older persons with advanced illness. J Gen Intern Med 2007; 22:495–501.
- Diwan S, Hougham GW, Sachs GA. Strain experienced by caregivers of dementia patients receiving palliative care: findings from the Palliative Excellence in Alzheimer Care Efforts (PEACE) Program. J Palliat Med 2004; 7:797–807.
- Covinsky KE, Yaffe K. Dementia, prognosis, and the needs of patients and caregivers. Ann Intern Med 2004; 140:573–574.
- Shega JW, Levin A, Hougham GW, et al. Palliative Excellence in Alzheimer Care Efforts (PEACE): a program description. J Palliat Med 2003; 6:315–320.
- Center for Ethics in Health Care. Physician orders for life-sustaining treatment paradigm. www.ohsu.edu/ethics/polst/. Accessed March 9, 2009.
- Lee MA, Brummel-Smith K, Meyer J, Drew N, London MR. Physician orders for life-sustaining treatment (POLST): outcomes in a PACE program. Program of All-Inclusive Care for the Elderly. J Am Geriatr Soc 2000; 48:1219–1225.
- Meyers JL, Moore C, McGrory A, Sparr J, Ahern M. Physician orders for life-sustaining treatment form: honoring end-of-life directives for nursing home residents. J Gerontol Nurs 2004; 30:37–46.
- Tolle SW, Tilden VP, Nelson CA, Dunn PM. A prospective study of the efficacy of the physician order form for life-sustaining treatment. J Am Geriatr Soc 1998; 46:1097–1102.
- Cantor MD, Pearlman RA. Advance care planning in long-term care facilities. J Am Med Dir Assoc 2004; 5(suppl 2):S72–S80.
KEY POINTS
- In the ambulatory setting, start by assessing the patient’s prognosis and his or her receptiveness to advance care planning.
- For a patient in declining health who is willing to participate in the care planning process, it may be useful to take a full values history and to review the goals of care.
- For a patient with advanced disease who is unable or unwilling to participate in advance care planning, a limited approach may be appropriate, ie, identifying a surrogate decision-maker and ascertaining how much flexibility the surrogate should have with health care decisions.
- Whatever the patient’s life expectancy and level of receptivity, brief, episodic discussions are more useful than a one-time description of available written advance directives.
Advance care planning is an art, not an algorithm
The article by Drs. Messinger-Rapport et al in this issue of the Cleveland Clinic Journal of Medicine makes several assertions about advance care planning with which I disagree. As a practicing oncologist and hospice medical director in a community setting for almost 20 years, I believe the authors’ attempt to reduce a complex physician-patient interaction to a practice algorithm oversimplifies the issues.
INTUITION, EXPERIENCE, AND TRAINING PLAY A VITAL ROLE
I agree with the authors that advance care planning is an oft-mentioned but underperformed function of being a physician in current American society. Our nation has become a people who deny physical vulnerability, frailty, and death. This area is not well taught in our medical schools and residency training programs.
However, the algorithms in the paper do not effectively address the timing and process of advance care planning. It is clearly not effective to have an end-of-life discussion at the bedside of a critically ill patient about to be intubated. It also seems futile to have a superficial discussion with patients about advance care planning at a time when they are healthy and in the setting of a routine and brief office visit. In this healthy population, the episodic discussions recommended by the authors could become background noise and could seem irrelevant to a patient. Physicians are already overburdened with responsibilities. Re-educating society that lives are not infinite is a social issue that needs to begin early in our lives.
In any setting, a social history, properly taken, should identify family structure, responsible next of kin, and family issues of discord that could affect treatment decisions and patient care. This is all that is needed as a minimal discussion about advance care planning.
Once patients develop significant illness, speaking to them about advance care planning becomes more relevant to their lives. As patients struggle with their illnesses and physical decline, the opportunity for further discussion grows and the impact of these interventions becomes greater. The energy and time it takes to have such discussions are better spent in these settings.
Discussing long-term planning with a patient is where intuition, experience, and training play a vital role. The balance between the pragmatic need for advance care planning and the need for allowing the patient to have hope of wellness is difficult to achieve and different for every patient, every family, and every care situation. It is for this reason that I find the flowcharts outlined in the paper difficult to follow and not very useful for the care of patients. Given the diversity of patients for whom we care, it seems impossible to me to condense the subject down to a care-flow matrix. This is an area in which the art of patient care and the art of being a physician come fully into play and cannot be replaced by an algorithm. Individual and small-group training with mentors, at all levels of medical education, would allow a physician to grow in comfort and skill in dealing with advance care planning.
LIVING WILLS CAN BE USEFUL
Another point on which I disagree with Dr. Messinger-Rapport et al is their assessment of the utility of the living will. They state that a living will applies only to patients who are terminally ill or in a persistent vegetative state. However, I find that it can also supply important information at all stages of illness. While it may lie “dormant” in a legal sense, it can give important information for a family by providing a window into the patient’s state of mind as it relates to the patient’s willingness to limit care in certain settings. Once a patient is able to articulate situations that warrant limiting care, a surrogate decision-maker (or the patient) can try to broaden those limits. It is up to the physician to articulate prognosis so the patient and family can decide how much they are willing to do to maintain that limited level of function. Any treatment can be declined at any time during a patient’s life or illness.
The living will also provides a framework in which to discuss end-of-life issues with a patient. It can open the discussion about current quality of life as perceived by the patient and what level of medical treatment the patient is willing to pursue. As the authors note in their article, those desires are fluid and can change over time. This does not render the living will useless. It shows that the living will needs to be adapted over time to suit the patient’s current situation.
The authors describe a patient with Alzheimer disease for whom a percutaneous endoscopic gastrostomy tube was recommended by a physician but declined by his wife. They assert that a living will does not offer guidance in this situation, since the patient was not, strictly speaking, terminally ill. I disagree. Medical care can delay death for years. If the patient’s quality of life is poor, that delay may violate the implied wishes of the patient and should be discussed. Before he became severely demented, the patient may not have wanted to have his life prolonged if the end result was a continued decline in his already compromised quality of life. The family should have been given that option to consider.
RE-EDUCATING PEOPLE ABOUT LIFE, ILLNESS, AND DEATH
Advance care planning is an essential component of being a physician and taking care of patients. A broad movement needs to be undertaken to re-educate people about the realities of life, illness, and death. The training of our physicians about advance care planning should begin early and should be continued throughout their medical education and careers. One-on-one or small-group mentoring would be an ideal method of training. The attempt to develop an algorithm to guide those discussions tries to simplify a process that is extraordinarily complex. Each situation is different and requires well-developed skills and practiced and mentored intuition. Experience and the art of being a physician cannot be reduced to a “model approach” or a flowchart.
The article by Drs. Messinger-Rapport et al in this issue of the Cleveland Clinic Journal of Medicine makes several assertions about advance care planning with which I disagree. As a practicing oncologist and hospice medical director in a community setting for almost 20 years, I believe the authors’ attempt to reduce a complex physician-patient interaction to a practice algorithm oversimplifies the issues.
INTUITION, EXPERIENCE, AND TRAINING PLAY A VITAL ROLE
I agree with the authors that advance care planning is an oft-mentioned but underperformed function of being a physician in current American society. Our nation has become a people who deny physical vulnerability, frailty, and death. This area is not well taught in our medical schools and residency training programs.
However, the algorithms in the paper do not effectively address the timing and process of advance care planning. It is clearly not effective to have an end-of-life discussion at the bedside of a critically ill patient about to be intubated. It also seems futile to have a superficial discussion with patients about advance care planning at a time when they are healthy and in the setting of a routine and brief office visit. In this healthy population, the episodic discussions recommended by the authors could become background noise and could seem irrelevant to a patient. Physicians are already overburdened with responsibilities. Re-educating society that lives are not infinite is a social issue that needs to begin early in our lives.
In any setting, a social history, properly taken, should identify family structure, responsible next of kin, and family issues of discord that could affect treatment decisions and patient care. This is all that is needed as a minimal discussion about advance care planning.
Once patients develop significant illness, speaking to them about advance care planning becomes more relevant to their lives. As patients struggle with their illnesses and physical decline, the opportunity for further discussion grows and the impact of these interventions becomes greater. The energy and time it takes to have such discussions are better spent in these settings.
Discussing long-term planning with a patient is where intuition, experience, and training play a vital role. The balance between the pragmatic need for advance care planning and the need for allowing the patient to have hope of wellness is difficult to achieve and different for every patient, every family, and every care situation. It is for this reason that I find the flowcharts outlined in the paper difficult to follow and not very useful for the care of patients. Given the diversity of patients for whom we care, it seems impossible to me to condense the subject down to a care-flow matrix. This is an area in which the art of patient care and the art of being a physician come fully into play and cannot be replaced by an algorithm. Individual and small-group training with mentors, at all levels of medical education, would allow a physician to grow in comfort and skill in dealing with advance care planning.
LIVING WILLS CAN BE USEFUL
Another point on which I disagree with Dr. Messinger-Rapport et al is their assessment of the utility of the living will. They state that a living will applies only to patients who are terminally ill or in a persistent vegetative state. However, I find that it can also supply important information at all stages of illness. While it may lie “dormant” in a legal sense, it can give important information for a family by providing a window into the patient’s state of mind as it relates to the patient’s willingness to limit care in certain settings. Once a patient is able to articulate situations that warrant limiting care, a surrogate decision-maker (or the patient) can try to broaden those limits. It is up to the physician to articulate prognosis so the patient and family can decide how much they are willing to do to maintain that limited level of function. Any treatment can be declined at any time during a patient’s life or illness.
The living will also provides a framework in which to discuss end-of-life issues with a patient. It can open the discussion about current quality of life as perceived by the patient and what level of medical treatment the patient is willing to pursue. As the authors note in their article, those desires are fluid and can change over time. This does not render the living will useless. It shows that the living will needs to be adapted over time to suit the patient’s current situation.
The authors describe a patient with Alzheimer disease for whom a percutaneous endoscopic gastrostomy tube was recommended by a physician but declined by his wife. They assert that a living will does not offer guidance in this situation, since the patient was not, strictly speaking, terminally ill. I disagree. Medical care can delay death for years. If the patient’s quality of life is poor, that delay may violate the implied wishes of the patient and should be discussed. Before he became severely demented, the patient may not have wanted to have his life prolonged if the end result was a continued decline in his already compromised quality of life. The family should have been given that option to consider.
RE-EDUCATING PEOPLE ABOUT LIFE, ILLNESS, AND DEATH
Advance care planning is an essential component of being a physician and taking care of patients. A broad movement needs to be undertaken to re-educate people about the realities of life, illness, and death. The training of our physicians about advance care planning should begin early and should be continued throughout their medical education and careers. One-on-one or small-group mentoring would be an ideal method of training. The attempt to develop an algorithm to guide those discussions tries to simplify a process that is extraordinarily complex. Each situation is different and requires well-developed skills and practiced and mentored intuition. Experience and the art of being a physician cannot be reduced to a “model approach” or a flowchart.
The article by Drs. Messinger-Rapport et al in this issue of the Cleveland Clinic Journal of Medicine makes several assertions about advance care planning with which I disagree. As a practicing oncologist and hospice medical director in a community setting for almost 20 years, I believe the authors’ attempt to reduce a complex physician-patient interaction to a practice algorithm oversimplifies the issues.
INTUITION, EXPERIENCE, AND TRAINING PLAY A VITAL ROLE
I agree with the authors that advance care planning is an oft-mentioned but underperformed function of being a physician in current American society. Our nation has become a people who deny physical vulnerability, frailty, and death. This area is not well taught in our medical schools and residency training programs.
However, the algorithms in the paper do not effectively address the timing and process of advance care planning. It is clearly not effective to have an end-of-life discussion at the bedside of a critically ill patient about to be intubated. It also seems futile to have a superficial discussion with patients about advance care planning at a time when they are healthy and in the setting of a routine and brief office visit. In this healthy population, the episodic discussions recommended by the authors could become background noise and could seem irrelevant to a patient. Physicians are already overburdened with responsibilities. Re-educating society that lives are not infinite is a social issue that needs to begin early in our lives.
In any setting, a social history, properly taken, should identify family structure, responsible next of kin, and family issues of discord that could affect treatment decisions and patient care. This is all that is needed as a minimal discussion about advance care planning.
Once patients develop significant illness, speaking to them about advance care planning becomes more relevant to their lives. As patients struggle with their illnesses and physical decline, the opportunity for further discussion grows and the impact of these interventions becomes greater. The energy and time it takes to have such discussions are better spent in these settings.
Discussing long-term planning with a patient is where intuition, experience, and training play a vital role. The balance between the pragmatic need for advance care planning and the need for allowing the patient to have hope of wellness is difficult to achieve and different for every patient, every family, and every care situation. It is for this reason that I find the flowcharts outlined in the paper difficult to follow and not very useful for the care of patients. Given the diversity of patients for whom we care, it seems impossible to me to condense the subject down to a care-flow matrix. This is an area in which the art of patient care and the art of being a physician come fully into play and cannot be replaced by an algorithm. Individual and small-group training with mentors, at all levels of medical education, would allow a physician to grow in comfort and skill in dealing with advance care planning.
LIVING WILLS CAN BE USEFUL
Another point on which I disagree with Dr. Messinger-Rapport et al is their assessment of the utility of the living will. They state that a living will applies only to patients who are terminally ill or in a persistent vegetative state. However, I find that it can also supply important information at all stages of illness. While it may lie “dormant” in a legal sense, it can give important information for a family by providing a window into the patient’s state of mind as it relates to the patient’s willingness to limit care in certain settings. Once a patient is able to articulate situations that warrant limiting care, a surrogate decision-maker (or the patient) can try to broaden those limits. It is up to the physician to articulate prognosis so the patient and family can decide how much they are willing to do to maintain that limited level of function. Any treatment can be declined at any time during a patient’s life or illness.
The living will also provides a framework in which to discuss end-of-life issues with a patient. It can open the discussion about current quality of life as perceived by the patient and what level of medical treatment the patient is willing to pursue. As the authors note in their article, those desires are fluid and can change over time. This does not render the living will useless. It shows that the living will needs to be adapted over time to suit the patient’s current situation.
The authors describe a patient with Alzheimer disease for whom a percutaneous endoscopic gastrostomy tube was recommended by a physician but declined by his wife. They assert that a living will does not offer guidance in this situation, since the patient was not, strictly speaking, terminally ill. I disagree. Medical care can delay death for years. If the patient’s quality of life is poor, that delay may violate the implied wishes of the patient and should be discussed. Before he became severely demented, the patient may not have wanted to have his life prolonged if the end result was a continued decline in his already compromised quality of life. The family should have been given that option to consider.
RE-EDUCATING PEOPLE ABOUT LIFE, ILLNESS, AND DEATH
Advance care planning is an essential component of being a physician and taking care of patients. A broad movement needs to be undertaken to re-educate people about the realities of life, illness, and death. The training of our physicians about advance care planning should begin early and should be continued throughout their medical education and careers. One-on-one or small-group mentoring would be an ideal method of training. The attempt to develop an algorithm to guide those discussions tries to simplify a process that is extraordinarily complex. Each situation is different and requires well-developed skills and practiced and mentored intuition. Experience and the art of being a physician cannot be reduced to a “model approach” or a flowchart.
Discuss Hospice and Palliative Care With Cancer Patients
AUSTIN, TEX. — Some palliative chemotherapy regimens can cost up to $100,000 a year for end-of-life care. Yet oncologists and their patients often do not discuss less costly, alternative advanced-care options.
“This is going to come to the fore over the next year or two, as fewer and fewer people have insurance,” Dr. Thomas Smith said at the annual meeting of the American Academy of Hospice and Palliative Care Medicine. “We spend twice as much as any other country for the same cancer results.”
He noted that some insurance companies may soon be asking patients to pay more for third-line treatments because of their reduced possibility of benefit. If hospice is introduced early in treatment as an end-of-life option, moreover, patients tend to switch earlier and spend more time in hospice, thereby reducing patient and hospital costs. Currently, one-third of patients with cancer spend fewer than 7 days in hospice, he said.
A sea change may already be underway. Kaiser Permanente has put hospice and palliative care teams in all of its major markets, and many insurers (such as UnitedHealthcare) are expected to roll out their plans for concurrent oncology and palliative care later this year, said Dr. Smith, professor of palliative care research at Virginia Commonwealth University in Richmond.
Part of the problem is that neither oncologists nor patients want to talk about death. A recent study showed that oncologists discussed prognosis 39% of the time and impending death only 37% of the time (JAMA 2008;300:1665–73). Of 111 inpatients with cancer, only 23 said they wished to discuss their advanced-care preferences with their oncologists, and 64 said they would prefer to do so with an admitting doctor (J. Palliat. Med. 2000;3:27–35).
Reimbursement is also a thorny issue. The Medicare reimbursement for hospice and palliative care hasn't kept pace with inflation or current oncology practice trends, even though patients with cancer account for about 40% of Medicare drug costs, Dr. Smith said. Oncologists are reimbursed far more for administering chemotherapeutic agents than for having discussions about prognosis and palliative care options.
It's also hard to find good “bad” news, Dr. Smith said. He noted that treatment options for recurrent pancreatic cancer on the National Cancer Institute's Web site (www.cancer.gov
“How about putting on there [that] in fact 95% of people are going to be dead within a year … and suggest hospice and palliative care?” he asked. “I've been beating on the NCI for 15 years on this, and will probably die before it happens.”
For those who say patients can't handle the truth, Dr. Smith said it is nearly impossible to take away hope. Most cancer patients are overly optimistic about their prognosis and are willing to take a phase I drug, even if it has a 10% chance of killing them.
New data suggest that one of the biggest fears of the terminally ill is abandonment by their physician or nurse when disease-modifying therapy is no longer an option (Arch. Intern. Med. 2009;169:474–9).
Written treatment plans offer patients truthful information about prognosis and treatment effectiveness, Dr. Smith said. He has been using them in his practice for years, and noted that the American Society of Clinical Oncology now makes these plans available online (www.asco.org
During the same presentation, Dr. Sarah E. Harrington offered suggestions for what oncologists should say about illness and patient options. They include being realistic about the goals of therapy; defining cure, remission, response, and what is likely to happen; and being negative, if appropriate.
“Language is important. Patients can easily mistake a 20% chance of response for a 20% chance of cure,” said Dr. Harrington, also at Virginia Commonwealth.
The subject of hospice should be brought up early as part of routine oncologic care, rather than delayed until death is imminent. Oncologists should be especially realistic about nth-line chemotherapy. If no proof of benefit is available, don't offer it, she said. To avoid feelings of abandonment, oncologists should tell their patients they will not abandon them if they enroll in hospice.
Dr. Harrington and Dr. Smith referred the audience to a recent article in which they discussed questions patients should consider when asking about palliative chemotherapy, and what oncologists should or should not do or say about chemotherapy for advanced cancer (JAMA 2008;299:2667–78).
They reported no financial disclosures.
AUSTIN, TEX. — Some palliative chemotherapy regimens can cost up to $100,000 a year for end-of-life care. Yet oncologists and their patients often do not discuss less costly, alternative advanced-care options.
“This is going to come to the fore over the next year or two, as fewer and fewer people have insurance,” Dr. Thomas Smith said at the annual meeting of the American Academy of Hospice and Palliative Care Medicine. “We spend twice as much as any other country for the same cancer results.”
He noted that some insurance companies may soon be asking patients to pay more for third-line treatments because of their reduced possibility of benefit. If hospice is introduced early in treatment as an end-of-life option, moreover, patients tend to switch earlier and spend more time in hospice, thereby reducing patient and hospital costs. Currently, one-third of patients with cancer spend fewer than 7 days in hospice, he said.
A sea change may already be underway. Kaiser Permanente has put hospice and palliative care teams in all of its major markets, and many insurers (such as UnitedHealthcare) are expected to roll out their plans for concurrent oncology and palliative care later this year, said Dr. Smith, professor of palliative care research at Virginia Commonwealth University in Richmond.
Part of the problem is that neither oncologists nor patients want to talk about death. A recent study showed that oncologists discussed prognosis 39% of the time and impending death only 37% of the time (JAMA 2008;300:1665–73). Of 111 inpatients with cancer, only 23 said they wished to discuss their advanced-care preferences with their oncologists, and 64 said they would prefer to do so with an admitting doctor (J. Palliat. Med. 2000;3:27–35).
Reimbursement is also a thorny issue. The Medicare reimbursement for hospice and palliative care hasn't kept pace with inflation or current oncology practice trends, even though patients with cancer account for about 40% of Medicare drug costs, Dr. Smith said. Oncologists are reimbursed far more for administering chemotherapeutic agents than for having discussions about prognosis and palliative care options.
It's also hard to find good “bad” news, Dr. Smith said. He noted that treatment options for recurrent pancreatic cancer on the National Cancer Institute's Web site (www.cancer.gov
“How about putting on there [that] in fact 95% of people are going to be dead within a year … and suggest hospice and palliative care?” he asked. “I've been beating on the NCI for 15 years on this, and will probably die before it happens.”
For those who say patients can't handle the truth, Dr. Smith said it is nearly impossible to take away hope. Most cancer patients are overly optimistic about their prognosis and are willing to take a phase I drug, even if it has a 10% chance of killing them.
New data suggest that one of the biggest fears of the terminally ill is abandonment by their physician or nurse when disease-modifying therapy is no longer an option (Arch. Intern. Med. 2009;169:474–9).
Written treatment plans offer patients truthful information about prognosis and treatment effectiveness, Dr. Smith said. He has been using them in his practice for years, and noted that the American Society of Clinical Oncology now makes these plans available online (www.asco.org
During the same presentation, Dr. Sarah E. Harrington offered suggestions for what oncologists should say about illness and patient options. They include being realistic about the goals of therapy; defining cure, remission, response, and what is likely to happen; and being negative, if appropriate.
“Language is important. Patients can easily mistake a 20% chance of response for a 20% chance of cure,” said Dr. Harrington, also at Virginia Commonwealth.
The subject of hospice should be brought up early as part of routine oncologic care, rather than delayed until death is imminent. Oncologists should be especially realistic about nth-line chemotherapy. If no proof of benefit is available, don't offer it, she said. To avoid feelings of abandonment, oncologists should tell their patients they will not abandon them if they enroll in hospice.
Dr. Harrington and Dr. Smith referred the audience to a recent article in which they discussed questions patients should consider when asking about palliative chemotherapy, and what oncologists should or should not do or say about chemotherapy for advanced cancer (JAMA 2008;299:2667–78).
They reported no financial disclosures.
AUSTIN, TEX. — Some palliative chemotherapy regimens can cost up to $100,000 a year for end-of-life care. Yet oncologists and their patients often do not discuss less costly, alternative advanced-care options.
“This is going to come to the fore over the next year or two, as fewer and fewer people have insurance,” Dr. Thomas Smith said at the annual meeting of the American Academy of Hospice and Palliative Care Medicine. “We spend twice as much as any other country for the same cancer results.”
He noted that some insurance companies may soon be asking patients to pay more for third-line treatments because of their reduced possibility of benefit. If hospice is introduced early in treatment as an end-of-life option, moreover, patients tend to switch earlier and spend more time in hospice, thereby reducing patient and hospital costs. Currently, one-third of patients with cancer spend fewer than 7 days in hospice, he said.
A sea change may already be underway. Kaiser Permanente has put hospice and palliative care teams in all of its major markets, and many insurers (such as UnitedHealthcare) are expected to roll out their plans for concurrent oncology and palliative care later this year, said Dr. Smith, professor of palliative care research at Virginia Commonwealth University in Richmond.
Part of the problem is that neither oncologists nor patients want to talk about death. A recent study showed that oncologists discussed prognosis 39% of the time and impending death only 37% of the time (JAMA 2008;300:1665–73). Of 111 inpatients with cancer, only 23 said they wished to discuss their advanced-care preferences with their oncologists, and 64 said they would prefer to do so with an admitting doctor (J. Palliat. Med. 2000;3:27–35).
Reimbursement is also a thorny issue. The Medicare reimbursement for hospice and palliative care hasn't kept pace with inflation or current oncology practice trends, even though patients with cancer account for about 40% of Medicare drug costs, Dr. Smith said. Oncologists are reimbursed far more for administering chemotherapeutic agents than for having discussions about prognosis and palliative care options.
It's also hard to find good “bad” news, Dr. Smith said. He noted that treatment options for recurrent pancreatic cancer on the National Cancer Institute's Web site (www.cancer.gov
“How about putting on there [that] in fact 95% of people are going to be dead within a year … and suggest hospice and palliative care?” he asked. “I've been beating on the NCI for 15 years on this, and will probably die before it happens.”
For those who say patients can't handle the truth, Dr. Smith said it is nearly impossible to take away hope. Most cancer patients are overly optimistic about their prognosis and are willing to take a phase I drug, even if it has a 10% chance of killing them.
New data suggest that one of the biggest fears of the terminally ill is abandonment by their physician or nurse when disease-modifying therapy is no longer an option (Arch. Intern. Med. 2009;169:474–9).
Written treatment plans offer patients truthful information about prognosis and treatment effectiveness, Dr. Smith said. He has been using them in his practice for years, and noted that the American Society of Clinical Oncology now makes these plans available online (www.asco.org
During the same presentation, Dr. Sarah E. Harrington offered suggestions for what oncologists should say about illness and patient options. They include being realistic about the goals of therapy; defining cure, remission, response, and what is likely to happen; and being negative, if appropriate.
“Language is important. Patients can easily mistake a 20% chance of response for a 20% chance of cure,” said Dr. Harrington, also at Virginia Commonwealth.
The subject of hospice should be brought up early as part of routine oncologic care, rather than delayed until death is imminent. Oncologists should be especially realistic about nth-line chemotherapy. If no proof of benefit is available, don't offer it, she said. To avoid feelings of abandonment, oncologists should tell their patients they will not abandon them if they enroll in hospice.
Dr. Harrington and Dr. Smith referred the audience to a recent article in which they discussed questions patients should consider when asking about palliative chemotherapy, and what oncologists should or should not do or say about chemotherapy for advanced cancer (JAMA 2008;299:2667–78).
They reported no financial disclosures.
Expert Sizes Up Palliative Care Medications
AUSTIN, TEX. — Many new medications relevant to palliative care have come on the market recently or are about to, hospital pharmacist Mary Lynn McPherson, Pharm.D., said at the American Academy of Hospice and Palliative Medicine annual meeting.
Dr. McPherson described several new prescription drugs and over-the-counter therapies that may often be given to patients at the end of life.
Dr. McPherson commented on the following products:
▸ Dexlansoprazole (Kapidex) delayed-release capsules were approved in late January for the treatment of heartburn associated with gastroesophageal reflux disease. This R-isomer of lansoprazole (Prevacid) comes to market just as Prevacid is expected to go generic. Dexlansoprazole is the first proton pump inhibitor (PPI) with a dual delayed-release formulation, allowing doses of 30–60 mg a day, versus 15–30 mg a day for Prevacid. Costs per month are $150 for dexlansoprazole and $168 for Prevacid.
“We get the patient frequently in my practice in a hospice on a PPI they don't even need that's been advertised to death,” said Dr. McPherson, a professor of pharmacy at the University of Maryland, Baltimore. “You know, the purple pill, [but] it doesn't work any better than the 80 cents a day over-the-counter omeprazole [Prilosec]. We only provide a PPI if the patient is on a steroid or nonsteroidal that we are also providing.”
▸ Sancuso is a transdermal patch designed to deliver 3.1 mg of granisetron over 24 hours to prevent emesis caused by moderate- and high-risk emetogenic drugs. Approved by the Food and Drug Administration last fall, the patch is applied to the upper arm at least 24 hours before the first chemotherapy session and can be worn for up to 7 days. In clinical trials, it has been shown to have equal efficacy to 2 mg oral granisetron per day, Dr. McPherson said. Cost is $287 per patch.
The patch may be a better option for inpatient palliative care than for home-based hospice, where Haldol (haloperidol) is the mainstay for nausea, she said.
▸ Zolpidem (ZolpiMist) 5-mg and 10-mg oral spray was approved in late December 2008 for the short-term treatment of difficulties getting to sleep. The spray acts quickly, reaching therapeutic levels within the body in 15 minutes. Patients should be prepared to spend 7–8 hours in bed after receiving the drug.
▸ Metoclopramide products, which include Reglan (metoclopramide) tablets and injections, received a black box warning in February because chronic use has been linked to tardive dyskinesia. Patients at the end of life typically are treated with up to 40 mg a day of metoclopramide for less than 3 months, but caution should be used in elderly patients, especially women, and in those receiving both Reglan and Haldol because the combination doubles the risk of tardive dyskinesia, Dr. McPherson said.
▸ Tapentadol, a centrally acting analgesic with potency between those of morphine and tramadol, was approved at the end of 2008 for relief of moderate to severe acute pain in adults. Although not approved for chronic pain, it may be of use in hospice and palliative care, Dr. McPherson said. Tapentadol is under review by the Drug Enforcement Administration and is expected to be a scheduled drug.
P Tramadol is not a controlled substance at the federal level, but it may be heading that way, Dr. McPherson said. Arkansas and Kentucky have made it a schedule IV drug, and authorities in North Dakota, Ohio, and Wyoming are tracking tramadol usage through their prescription drug monitoring program as if it were controlled.
▸ Propoxyphene may be on the chopping block after two FDA advisory committees narrowly voted on Jan. 30 to recommend discontinued marketing of Darvon and Darvocet for mild to moderate pain. Propoxyphene is banned in the United Kingdom and is rarely used in Canada, but it is among the top 25 most prescribed drugs in the United States, Dr. McPherson said. She added that physicians like it because it causes less stomach upset than other opioids and can be taken by patients allergic to morphine and hydrocodone.
Propoxyphene is far from benign, Dr. McPherson said, noting that both the drug and its metabolite are cardiotoxic. Propoxyphene was a factor in 5.6% of drug-related deaths in the United States from 1981 to 1999, she said.
▸ OTC products. Emuprofen is a topically administered analgesic that contains ibuprofen and oil from the fat of the emu. It's being marketed as an anti-inflammatory and an alternative to systemic NSAID therapy for various painful conditions. Cost is about $35 for a small jar. The cream is about 10% ibuprofen. Rash and itching at the application site have been reported in up to 15% of patients.
Rain Dry Mouth Spray may be another therapeutic option for xerostomia, which affects 20% of the U.S. population and is common in people with head and neck cancer. The active ingredient is xylitol, a sugar alcohol, which can raise blood glucose if overused. Cost is about $14 for 4.5 ounces.
Tums QuickPak is a calcium carbonate powder that dissolves instantly on the tongue without the need for water and is the equivalent of two regular-strength Tums. It can be used as a daily calcium source, and for patients who can no longer swallow, she said.
Dr. McPherson disclosed that she is a consultant for Alpharma Inc.
AUSTIN, TEX. — Many new medications relevant to palliative care have come on the market recently or are about to, hospital pharmacist Mary Lynn McPherson, Pharm.D., said at the American Academy of Hospice and Palliative Medicine annual meeting.
Dr. McPherson described several new prescription drugs and over-the-counter therapies that may often be given to patients at the end of life.
Dr. McPherson commented on the following products:
▸ Dexlansoprazole (Kapidex) delayed-release capsules were approved in late January for the treatment of heartburn associated with gastroesophageal reflux disease. This R-isomer of lansoprazole (Prevacid) comes to market just as Prevacid is expected to go generic. Dexlansoprazole is the first proton pump inhibitor (PPI) with a dual delayed-release formulation, allowing doses of 30–60 mg a day, versus 15–30 mg a day for Prevacid. Costs per month are $150 for dexlansoprazole and $168 for Prevacid.
“We get the patient frequently in my practice in a hospice on a PPI they don't even need that's been advertised to death,” said Dr. McPherson, a professor of pharmacy at the University of Maryland, Baltimore. “You know, the purple pill, [but] it doesn't work any better than the 80 cents a day over-the-counter omeprazole [Prilosec]. We only provide a PPI if the patient is on a steroid or nonsteroidal that we are also providing.”
▸ Sancuso is a transdermal patch designed to deliver 3.1 mg of granisetron over 24 hours to prevent emesis caused by moderate- and high-risk emetogenic drugs. Approved by the Food and Drug Administration last fall, the patch is applied to the upper arm at least 24 hours before the first chemotherapy session and can be worn for up to 7 days. In clinical trials, it has been shown to have equal efficacy to 2 mg oral granisetron per day, Dr. McPherson said. Cost is $287 per patch.
The patch may be a better option for inpatient palliative care than for home-based hospice, where Haldol (haloperidol) is the mainstay for nausea, she said.
▸ Zolpidem (ZolpiMist) 5-mg and 10-mg oral spray was approved in late December 2008 for the short-term treatment of difficulties getting to sleep. The spray acts quickly, reaching therapeutic levels within the body in 15 minutes. Patients should be prepared to spend 7–8 hours in bed after receiving the drug.
▸ Metoclopramide products, which include Reglan (metoclopramide) tablets and injections, received a black box warning in February because chronic use has been linked to tardive dyskinesia. Patients at the end of life typically are treated with up to 40 mg a day of metoclopramide for less than 3 months, but caution should be used in elderly patients, especially women, and in those receiving both Reglan and Haldol because the combination doubles the risk of tardive dyskinesia, Dr. McPherson said.
▸ Tapentadol, a centrally acting analgesic with potency between those of morphine and tramadol, was approved at the end of 2008 for relief of moderate to severe acute pain in adults. Although not approved for chronic pain, it may be of use in hospice and palliative care, Dr. McPherson said. Tapentadol is under review by the Drug Enforcement Administration and is expected to be a scheduled drug.
P Tramadol is not a controlled substance at the federal level, but it may be heading that way, Dr. McPherson said. Arkansas and Kentucky have made it a schedule IV drug, and authorities in North Dakota, Ohio, and Wyoming are tracking tramadol usage through their prescription drug monitoring program as if it were controlled.
▸ Propoxyphene may be on the chopping block after two FDA advisory committees narrowly voted on Jan. 30 to recommend discontinued marketing of Darvon and Darvocet for mild to moderate pain. Propoxyphene is banned in the United Kingdom and is rarely used in Canada, but it is among the top 25 most prescribed drugs in the United States, Dr. McPherson said. She added that physicians like it because it causes less stomach upset than other opioids and can be taken by patients allergic to morphine and hydrocodone.
Propoxyphene is far from benign, Dr. McPherson said, noting that both the drug and its metabolite are cardiotoxic. Propoxyphene was a factor in 5.6% of drug-related deaths in the United States from 1981 to 1999, she said.
▸ OTC products. Emuprofen is a topically administered analgesic that contains ibuprofen and oil from the fat of the emu. It's being marketed as an anti-inflammatory and an alternative to systemic NSAID therapy for various painful conditions. Cost is about $35 for a small jar. The cream is about 10% ibuprofen. Rash and itching at the application site have been reported in up to 15% of patients.
Rain Dry Mouth Spray may be another therapeutic option for xerostomia, which affects 20% of the U.S. population and is common in people with head and neck cancer. The active ingredient is xylitol, a sugar alcohol, which can raise blood glucose if overused. Cost is about $14 for 4.5 ounces.
Tums QuickPak is a calcium carbonate powder that dissolves instantly on the tongue without the need for water and is the equivalent of two regular-strength Tums. It can be used as a daily calcium source, and for patients who can no longer swallow, she said.
Dr. McPherson disclosed that she is a consultant for Alpharma Inc.
AUSTIN, TEX. — Many new medications relevant to palliative care have come on the market recently or are about to, hospital pharmacist Mary Lynn McPherson, Pharm.D., said at the American Academy of Hospice and Palliative Medicine annual meeting.
Dr. McPherson described several new prescription drugs and over-the-counter therapies that may often be given to patients at the end of life.
Dr. McPherson commented on the following products:
▸ Dexlansoprazole (Kapidex) delayed-release capsules were approved in late January for the treatment of heartburn associated with gastroesophageal reflux disease. This R-isomer of lansoprazole (Prevacid) comes to market just as Prevacid is expected to go generic. Dexlansoprazole is the first proton pump inhibitor (PPI) with a dual delayed-release formulation, allowing doses of 30–60 mg a day, versus 15–30 mg a day for Prevacid. Costs per month are $150 for dexlansoprazole and $168 for Prevacid.
“We get the patient frequently in my practice in a hospice on a PPI they don't even need that's been advertised to death,” said Dr. McPherson, a professor of pharmacy at the University of Maryland, Baltimore. “You know, the purple pill, [but] it doesn't work any better than the 80 cents a day over-the-counter omeprazole [Prilosec]. We only provide a PPI if the patient is on a steroid or nonsteroidal that we are also providing.”
▸ Sancuso is a transdermal patch designed to deliver 3.1 mg of granisetron over 24 hours to prevent emesis caused by moderate- and high-risk emetogenic drugs. Approved by the Food and Drug Administration last fall, the patch is applied to the upper arm at least 24 hours before the first chemotherapy session and can be worn for up to 7 days. In clinical trials, it has been shown to have equal efficacy to 2 mg oral granisetron per day, Dr. McPherson said. Cost is $287 per patch.
The patch may be a better option for inpatient palliative care than for home-based hospice, where Haldol (haloperidol) is the mainstay for nausea, she said.
▸ Zolpidem (ZolpiMist) 5-mg and 10-mg oral spray was approved in late December 2008 for the short-term treatment of difficulties getting to sleep. The spray acts quickly, reaching therapeutic levels within the body in 15 minutes. Patients should be prepared to spend 7–8 hours in bed after receiving the drug.
▸ Metoclopramide products, which include Reglan (metoclopramide) tablets and injections, received a black box warning in February because chronic use has been linked to tardive dyskinesia. Patients at the end of life typically are treated with up to 40 mg a day of metoclopramide for less than 3 months, but caution should be used in elderly patients, especially women, and in those receiving both Reglan and Haldol because the combination doubles the risk of tardive dyskinesia, Dr. McPherson said.
▸ Tapentadol, a centrally acting analgesic with potency between those of morphine and tramadol, was approved at the end of 2008 for relief of moderate to severe acute pain in adults. Although not approved for chronic pain, it may be of use in hospice and palliative care, Dr. McPherson said. Tapentadol is under review by the Drug Enforcement Administration and is expected to be a scheduled drug.
P Tramadol is not a controlled substance at the federal level, but it may be heading that way, Dr. McPherson said. Arkansas and Kentucky have made it a schedule IV drug, and authorities in North Dakota, Ohio, and Wyoming are tracking tramadol usage through their prescription drug monitoring program as if it were controlled.
▸ Propoxyphene may be on the chopping block after two FDA advisory committees narrowly voted on Jan. 30 to recommend discontinued marketing of Darvon and Darvocet for mild to moderate pain. Propoxyphene is banned in the United Kingdom and is rarely used in Canada, but it is among the top 25 most prescribed drugs in the United States, Dr. McPherson said. She added that physicians like it because it causes less stomach upset than other opioids and can be taken by patients allergic to morphine and hydrocodone.
Propoxyphene is far from benign, Dr. McPherson said, noting that both the drug and its metabolite are cardiotoxic. Propoxyphene was a factor in 5.6% of drug-related deaths in the United States from 1981 to 1999, she said.
▸ OTC products. Emuprofen is a topically administered analgesic that contains ibuprofen and oil from the fat of the emu. It's being marketed as an anti-inflammatory and an alternative to systemic NSAID therapy for various painful conditions. Cost is about $35 for a small jar. The cream is about 10% ibuprofen. Rash and itching at the application site have been reported in up to 15% of patients.
Rain Dry Mouth Spray may be another therapeutic option for xerostomia, which affects 20% of the U.S. population and is common in people with head and neck cancer. The active ingredient is xylitol, a sugar alcohol, which can raise blood glucose if overused. Cost is about $14 for 4.5 ounces.
Tums QuickPak is a calcium carbonate powder that dissolves instantly on the tongue without the need for water and is the equivalent of two regular-strength Tums. It can be used as a daily calcium source, and for patients who can no longer swallow, she said.
Dr. McPherson disclosed that she is a consultant for Alpharma Inc.
Palliative-Care Payment
Many hospitalists provide palliative-care services to patients at the request of physicians within their own groups or from other specialists. Varying factors affect how hospitalists report these services—namely, the nature of the request and the type of service provided. Palliative-care programs can be quite costly as they involve several team members and a substantial amount of time delivering these services. Capturing services appropriately and obtaining reimbursement to help continue program initiatives is pertinent.
Nature of the Request
Members of a palliative-care team often are called on to provide management options to assist in reducing pain and suffering associated with both terminal and nonterminal disease, thereby improving a patient’s quality of life. When a palliative-care specialist is asked to provide an opinion or advice, the initial service could qualify as a consultation. However, all requirements must be met in order to report the service as an inpatient consultation (codes 99251-99255).
There must be a written request from a qualified healthcare provider involved in the patient’s care (e.g., a physician, resident, or nurse practitioner). In the inpatient setting, this request can be documented as a physician order or in the assessment of the requesting provider’s progress note. Standing orders for consultation are not permitted. Ideally, the requesting provider should identify the reason for a consult to support the medical necessity of the service.
Additionally, the palliative-care physician renders and documents the service, then reports findings to the requesting physician. The consultant’s required written report does not have to be sent separately to the requesting physician. Because the requesting physician and the consultant share a common medical record in an inpatient setting, the consultant’s inpatient progress note suffices the “written report” requirement.
One concern about billing consultations involves the nature of the request. If the requesting physician documents the need for an opinion or advice from the palliative-care specialist, the service can be reported as a consultation. If, however, the request states consult for “medical management” or “palliative management,” it’s less likely that payors will consider the service a consultation. In the latter situation, it appears as if the requesting physician is not seeking an opinion or advice from the consultant to incorporate into his own plan of care for the patient and would rather the consultant take over that portion of patient care.
Recently revised billing policies prevent the consultant from billing consults under these circumstances. Without a sufficient request for consultation, the palliative-care specialist can only report “subsequent” hospital care services.1 Language that better supports the consultative nature of the request is:
- Consult for an opinion or advice on palliative measures;
- Consult for evaluation of palliative options; and
- Consult palliative care for treatment options.
Proper Documentation
The requesting physician can be in the same or different provider group as the consultant. The consultant must possess expertise in an area beyond that of the requesting provider. Because the specialty designation for most hospitalists is internal medicine, palliative-care claims could be scrutinized more closely. This does not necessarily occur when the requesting provider has a different two-digit specialty designation (e.g., internal medicine and gastroenterology).2 Scrutiny is more likely to occur when the requesting provider has the same internal-medicine designation as the palliative-care consultant, even if they are in different provider groups.
Payor concern escalates when physicians of the same designated specialty submit claims for the same patient on the same date. Having different primary diagnosis codes attached to each visit level does not necessarily help. The payor is likely to deny the second claim received, pending a review of documentation. If this happens, the provider who received the denial should submit a copy of both progress notes for the date in question. Hopefully, the distinction between the services is demonstrated in the documentation.
Service Type
Palliative services might involve obtaining and documenting the standard key components for visit-level selection: history, exam, and medical decision-making.3 However, the palliative-care specialist might spend more time providing counseling or coordination of care for a patient and family. When this occurs, the palliative-care specialist should not forget about the guidelines for reporting time-based services.4 Inpatient services may be reported on the basis of time, as long as a face-to-face service between the provider and the patient occurs. Consider the total time spent face to face with the patient, and the time spent obtaining, discussing, and coordinating patient care, while you are in the patient’s unit or floor.
As a reminder, document the total time, the amount of time spent counseling, and the details of discussion and coordination. The physician may count the time spent counseling the patient’s family regarding the treatment and care, as long as the focus is not emotional support for the family, the meeting takes place in the patient’s unit or floor, and the patient is present, unless there is medically supported reason for which the patient is unable to participate (e.g., cognitive impairment). The palliative-care specialist can then select the visit level based on time.5 (See Table 1, above.) TH
Carol Pohlig is a billing and coding expert with the University of Pennsylvania Medical Center, Philadelphia. She is on the faculty of SHM’s inpatient coding course.
References
1. Centers for Medicare and Medicaid Services. Medicare Claims Processing Manual: Chapter 12, Section 30.6.10. CMS Web site. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed Jan. 30, 2009.
2. Centers for Medicare and Medicaid Services. Medicare Claims Processing Manual: Chapter 26, Section 10.8. CMS Web site. Available at: www.cms.hhs.gov/manuals/downloads/clm104c26.pdf. Accessed Jan. 30, 2009.
3. Centers for Medicare and Medicaid Services. Documentation Guidelines for Evaluation and Management Services. CMS Web site. Available at: www.cms.hhs.gov/MLNEdWebGuide/25_EMDOC.asp. Accessed Jan. 30, 2009.
4. Centers for Medicare and Medicaid Services. Medicare Claims Processing Manual: Chapter 12, Section 30.6.1B. CMS Web site. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed Jan. 30, 2009.
5. Beebe M, Dalton J, Espronceda M, Evans D, Glenn R. Current Procedural Terminology Professional Edition. Chicago: American Medical Association Press; 2008.
Many hospitalists provide palliative-care services to patients at the request of physicians within their own groups or from other specialists. Varying factors affect how hospitalists report these services—namely, the nature of the request and the type of service provided. Palliative-care programs can be quite costly as they involve several team members and a substantial amount of time delivering these services. Capturing services appropriately and obtaining reimbursement to help continue program initiatives is pertinent.
Nature of the Request
Members of a palliative-care team often are called on to provide management options to assist in reducing pain and suffering associated with both terminal and nonterminal disease, thereby improving a patient’s quality of life. When a palliative-care specialist is asked to provide an opinion or advice, the initial service could qualify as a consultation. However, all requirements must be met in order to report the service as an inpatient consultation (codes 99251-99255).
There must be a written request from a qualified healthcare provider involved in the patient’s care (e.g., a physician, resident, or nurse practitioner). In the inpatient setting, this request can be documented as a physician order or in the assessment of the requesting provider’s progress note. Standing orders for consultation are not permitted. Ideally, the requesting provider should identify the reason for a consult to support the medical necessity of the service.
Additionally, the palliative-care physician renders and documents the service, then reports findings to the requesting physician. The consultant’s required written report does not have to be sent separately to the requesting physician. Because the requesting physician and the consultant share a common medical record in an inpatient setting, the consultant’s inpatient progress note suffices the “written report” requirement.
One concern about billing consultations involves the nature of the request. If the requesting physician documents the need for an opinion or advice from the palliative-care specialist, the service can be reported as a consultation. If, however, the request states consult for “medical management” or “palliative management,” it’s less likely that payors will consider the service a consultation. In the latter situation, it appears as if the requesting physician is not seeking an opinion or advice from the consultant to incorporate into his own plan of care for the patient and would rather the consultant take over that portion of patient care.
Recently revised billing policies prevent the consultant from billing consults under these circumstances. Without a sufficient request for consultation, the palliative-care specialist can only report “subsequent” hospital care services.1 Language that better supports the consultative nature of the request is:
- Consult for an opinion or advice on palliative measures;
- Consult for evaluation of palliative options; and
- Consult palliative care for treatment options.
Proper Documentation
The requesting physician can be in the same or different provider group as the consultant. The consultant must possess expertise in an area beyond that of the requesting provider. Because the specialty designation for most hospitalists is internal medicine, palliative-care claims could be scrutinized more closely. This does not necessarily occur when the requesting provider has a different two-digit specialty designation (e.g., internal medicine and gastroenterology).2 Scrutiny is more likely to occur when the requesting provider has the same internal-medicine designation as the palliative-care consultant, even if they are in different provider groups.
Payor concern escalates when physicians of the same designated specialty submit claims for the same patient on the same date. Having different primary diagnosis codes attached to each visit level does not necessarily help. The payor is likely to deny the second claim received, pending a review of documentation. If this happens, the provider who received the denial should submit a copy of both progress notes for the date in question. Hopefully, the distinction between the services is demonstrated in the documentation.
Service Type
Palliative services might involve obtaining and documenting the standard key components for visit-level selection: history, exam, and medical decision-making.3 However, the palliative-care specialist might spend more time providing counseling or coordination of care for a patient and family. When this occurs, the palliative-care specialist should not forget about the guidelines for reporting time-based services.4 Inpatient services may be reported on the basis of time, as long as a face-to-face service between the provider and the patient occurs. Consider the total time spent face to face with the patient, and the time spent obtaining, discussing, and coordinating patient care, while you are in the patient’s unit or floor.
As a reminder, document the total time, the amount of time spent counseling, and the details of discussion and coordination. The physician may count the time spent counseling the patient’s family regarding the treatment and care, as long as the focus is not emotional support for the family, the meeting takes place in the patient’s unit or floor, and the patient is present, unless there is medically supported reason for which the patient is unable to participate (e.g., cognitive impairment). The palliative-care specialist can then select the visit level based on time.5 (See Table 1, above.) TH
Carol Pohlig is a billing and coding expert with the University of Pennsylvania Medical Center, Philadelphia. She is on the faculty of SHM’s inpatient coding course.
References
1. Centers for Medicare and Medicaid Services. Medicare Claims Processing Manual: Chapter 12, Section 30.6.10. CMS Web site. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed Jan. 30, 2009.
2. Centers for Medicare and Medicaid Services. Medicare Claims Processing Manual: Chapter 26, Section 10.8. CMS Web site. Available at: www.cms.hhs.gov/manuals/downloads/clm104c26.pdf. Accessed Jan. 30, 2009.
3. Centers for Medicare and Medicaid Services. Documentation Guidelines for Evaluation and Management Services. CMS Web site. Available at: www.cms.hhs.gov/MLNEdWebGuide/25_EMDOC.asp. Accessed Jan. 30, 2009.
4. Centers for Medicare and Medicaid Services. Medicare Claims Processing Manual: Chapter 12, Section 30.6.1B. CMS Web site. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed Jan. 30, 2009.
5. Beebe M, Dalton J, Espronceda M, Evans D, Glenn R. Current Procedural Terminology Professional Edition. Chicago: American Medical Association Press; 2008.
Many hospitalists provide palliative-care services to patients at the request of physicians within their own groups or from other specialists. Varying factors affect how hospitalists report these services—namely, the nature of the request and the type of service provided. Palliative-care programs can be quite costly as they involve several team members and a substantial amount of time delivering these services. Capturing services appropriately and obtaining reimbursement to help continue program initiatives is pertinent.
Nature of the Request
Members of a palliative-care team often are called on to provide management options to assist in reducing pain and suffering associated with both terminal and nonterminal disease, thereby improving a patient’s quality of life. When a palliative-care specialist is asked to provide an opinion or advice, the initial service could qualify as a consultation. However, all requirements must be met in order to report the service as an inpatient consultation (codes 99251-99255).
There must be a written request from a qualified healthcare provider involved in the patient’s care (e.g., a physician, resident, or nurse practitioner). In the inpatient setting, this request can be documented as a physician order or in the assessment of the requesting provider’s progress note. Standing orders for consultation are not permitted. Ideally, the requesting provider should identify the reason for a consult to support the medical necessity of the service.
Additionally, the palliative-care physician renders and documents the service, then reports findings to the requesting physician. The consultant’s required written report does not have to be sent separately to the requesting physician. Because the requesting physician and the consultant share a common medical record in an inpatient setting, the consultant’s inpatient progress note suffices the “written report” requirement.
One concern about billing consultations involves the nature of the request. If the requesting physician documents the need for an opinion or advice from the palliative-care specialist, the service can be reported as a consultation. If, however, the request states consult for “medical management” or “palliative management,” it’s less likely that payors will consider the service a consultation. In the latter situation, it appears as if the requesting physician is not seeking an opinion or advice from the consultant to incorporate into his own plan of care for the patient and would rather the consultant take over that portion of patient care.
Recently revised billing policies prevent the consultant from billing consults under these circumstances. Without a sufficient request for consultation, the palliative-care specialist can only report “subsequent” hospital care services.1 Language that better supports the consultative nature of the request is:
- Consult for an opinion or advice on palliative measures;
- Consult for evaluation of palliative options; and
- Consult palliative care for treatment options.
Proper Documentation
The requesting physician can be in the same or different provider group as the consultant. The consultant must possess expertise in an area beyond that of the requesting provider. Because the specialty designation for most hospitalists is internal medicine, palliative-care claims could be scrutinized more closely. This does not necessarily occur when the requesting provider has a different two-digit specialty designation (e.g., internal medicine and gastroenterology).2 Scrutiny is more likely to occur when the requesting provider has the same internal-medicine designation as the palliative-care consultant, even if they are in different provider groups.
Payor concern escalates when physicians of the same designated specialty submit claims for the same patient on the same date. Having different primary diagnosis codes attached to each visit level does not necessarily help. The payor is likely to deny the second claim received, pending a review of documentation. If this happens, the provider who received the denial should submit a copy of both progress notes for the date in question. Hopefully, the distinction between the services is demonstrated in the documentation.
Service Type
Palliative services might involve obtaining and documenting the standard key components for visit-level selection: history, exam, and medical decision-making.3 However, the palliative-care specialist might spend more time providing counseling or coordination of care for a patient and family. When this occurs, the palliative-care specialist should not forget about the guidelines for reporting time-based services.4 Inpatient services may be reported on the basis of time, as long as a face-to-face service between the provider and the patient occurs. Consider the total time spent face to face with the patient, and the time spent obtaining, discussing, and coordinating patient care, while you are in the patient’s unit or floor.
As a reminder, document the total time, the amount of time spent counseling, and the details of discussion and coordination. The physician may count the time spent counseling the patient’s family regarding the treatment and care, as long as the focus is not emotional support for the family, the meeting takes place in the patient’s unit or floor, and the patient is present, unless there is medically supported reason for which the patient is unable to participate (e.g., cognitive impairment). The palliative-care specialist can then select the visit level based on time.5 (See Table 1, above.) TH
Carol Pohlig is a billing and coding expert with the University of Pennsylvania Medical Center, Philadelphia. She is on the faculty of SHM’s inpatient coding course.
References
1. Centers for Medicare and Medicaid Services. Medicare Claims Processing Manual: Chapter 12, Section 30.6.10. CMS Web site. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed Jan. 30, 2009.
2. Centers for Medicare and Medicaid Services. Medicare Claims Processing Manual: Chapter 26, Section 10.8. CMS Web site. Available at: www.cms.hhs.gov/manuals/downloads/clm104c26.pdf. Accessed Jan. 30, 2009.
3. Centers for Medicare and Medicaid Services. Documentation Guidelines for Evaluation and Management Services. CMS Web site. Available at: www.cms.hhs.gov/MLNEdWebGuide/25_EMDOC.asp. Accessed Jan. 30, 2009.
4. Centers for Medicare and Medicaid Services. Medicare Claims Processing Manual: Chapter 12, Section 30.6.1B. CMS Web site. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed Jan. 30, 2009.
5. Beebe M, Dalton J, Espronceda M, Evans D, Glenn R. Current Procedural Terminology Professional Edition. Chicago: American Medical Association Press; 2008.
“Caregiver Culture” and End-of-Life Discussions
When it comes to discussing a patient’s wishes for code status care, practices at the institution play a more important role than almost any other factor, according to a new study, “Factors Associated with Discussion of Care Plans and Code Status at the Time of Hospital Admission: Results from the Multicenter Hospitalist Study,” in the Journal of Hospital Medicine.
“What was most surprising to me was how variable the discussion rate was,” lead author Andrew Auerbach, MD, tells The Hospitalist. “It had little or nothing to do with how sick the patient was, or with the type of institution in which the discussion took place.”
Hospitalists are no more or less likely to document such discussions than doctors in any other specialty, says Dr. Auerbach, a hospitalist and associate professor of medicine at the University of California San Francisco. He and his colleagues analyzed data from patients admitted to the general medicine services at six academic medical centers as part of the Multicenter Hospitalist Study. Each site complied with requirements established by the Patient Self-Determination Act (PSDA), which says patients must be informed of their right to create an advance directive.
None of the hospitals in the study had established guidelines or formal policies regarding physician-patient discussions about code status or end-of-life care. Patients were interviewed immediately after informed consent was obtained, usually within 24 hours of admission. In each case, the authors determined whether or not the patient had had a care discussion, defined as a documented discussion “between patients (or family) and at least one physician … during the first 24 hours of hospitalization,” the authors write. “Care discussions needed to specify that the person who wrote the note had actually spoken with the patient or their family for the purposes of determining preferences for care, and that this discussion resulted in a specific care plan.”
A notation, such as “the patient continues to want full efforts,” qualified as a care discussion. Less-specific comments, such as “DNR/DNI” or “spoke with family, questions answered,” did not qualify. “These were all really, really sick people, and it was important to assess their wishes for care,” Dr. Auerbach says. Individually, PSDA or durable power of attorney may not adequately convey a patient’s true wishes, because often times the measures involve nothing more than having the patient or family complete a form. “The true marker is a conversation,” he says.
—Andrew Auerbach, MD, University of California San Francisco
By the Numbers
Of 17,097 patients interviewed, only 1,776 (10.4%) had a documented care discussion within 24 hours of hospital admission. The frequency of discussions varied from a low of 2.8% at one institution, to a high of 24.9% at another.
On unadjusted analysis, patients with documented care discussions were more likely to have living wills, durable powers of attorney, or the names of surrogate decision-makers in their charts (P<0.0001 for all categories). These patients were older, more likely to be white, and more likely to be on Medicare, compared to patients without documented care discussions.
The unadjusted analysis also showed patients with care discussions were more likely to be married, but less likely to be living in their own home or apartment, and, not surprisingly, more likely to have been hospitalized at least once within the previous 12 months. Overall, the general health of patients with care discussions was poorer than those without. Patients with documented discussions were more likely to report needing help within the past month with chores or bathing or dressing themselves, than were patients who did not have care discussions. Cancer, depression, and a history of stroke were common among patients with care discussions. Compared to patients without documented discussions, those who did have the discussion appear to want more of a say in their care: they were less likely to agree with the study statements, “I prefer my doctor give me choices regarding my care,” and “I prefer to leave care decisions to my physician.”
The authors found multivariate analysis showed many of these factors turn out to have only a moderate association with a documented care discussion, with adjusted odds ratios of less than 2.0. The strongest predictors were the existence of informal notations describing pre-hospital care wishes, with odds ratios ranging from 3.22 to 11.32, compared to people with no such documentation, and site of enrollment, with odds ratios of 1.74 to 5.14.
The Caregiver Culture
These findings suggest the “caregiver culture” at any given institution is a stronger determinant of a patient participating in a documented care discussion than other, more intuitive factors, such as medical condition or socioeconomic characteristics, or even whether or not the patient has a pre-existing advance directive or durable power of attorney, Dr. Auerbach explains. “It may just be a part of what some hospitals do. It’s driven by what your peers are doing and by local practices.”
Based on the results of the study, Dr. Auerbach and his co-authors suggest simply establishing mandates to document code status on admission probably will not encourage more conversations of this nature, “unless sites also develop an approach to using this newly documented information as a prompt for subsequent discussions.” The Patient Self-Determination Act went into effect in 1991, but its effect on improving communication around end-of-life care remains uncertain, the authors note. It might be why California passed a new law, effective as of January 2009, requiring physicians and health care organizations in the state to provide terminally ill patients who ask about their end-of-life care options with comprehensive information and counseling. Such discussions must cover advance directives, hospice care, and the right to receive palliative care. The new law is the first of its kind in the nation, but proponents of the legislation hope it will serve as a model for other states to adopt.1
“Documentation has an impact on quality of care. Lots of errors are driven by code status,” Dr. Auerbach points out. “If a patient is admitted in the middle of the night, when the doctor checks in on him the next day, he can look at the chart and see that his partner documented the care discussion the night before. That is incredibly helpful.” TH
Norra MacReady is a medical writer based in California.
Reference
1. O’Reilly KB. California law mandates discussing end-of-life options. Am Med News Web site. Available at amaassn.org/amednews/2008/11/10/prsc1110.htm. Last accessed November 6, 2008.
When it comes to discussing a patient’s wishes for code status care, practices at the institution play a more important role than almost any other factor, according to a new study, “Factors Associated with Discussion of Care Plans and Code Status at the Time of Hospital Admission: Results from the Multicenter Hospitalist Study,” in the Journal of Hospital Medicine.
“What was most surprising to me was how variable the discussion rate was,” lead author Andrew Auerbach, MD, tells The Hospitalist. “It had little or nothing to do with how sick the patient was, or with the type of institution in which the discussion took place.”
Hospitalists are no more or less likely to document such discussions than doctors in any other specialty, says Dr. Auerbach, a hospitalist and associate professor of medicine at the University of California San Francisco. He and his colleagues analyzed data from patients admitted to the general medicine services at six academic medical centers as part of the Multicenter Hospitalist Study. Each site complied with requirements established by the Patient Self-Determination Act (PSDA), which says patients must be informed of their right to create an advance directive.
None of the hospitals in the study had established guidelines or formal policies regarding physician-patient discussions about code status or end-of-life care. Patients were interviewed immediately after informed consent was obtained, usually within 24 hours of admission. In each case, the authors determined whether or not the patient had had a care discussion, defined as a documented discussion “between patients (or family) and at least one physician … during the first 24 hours of hospitalization,” the authors write. “Care discussions needed to specify that the person who wrote the note had actually spoken with the patient or their family for the purposes of determining preferences for care, and that this discussion resulted in a specific care plan.”
A notation, such as “the patient continues to want full efforts,” qualified as a care discussion. Less-specific comments, such as “DNR/DNI” or “spoke with family, questions answered,” did not qualify. “These were all really, really sick people, and it was important to assess their wishes for care,” Dr. Auerbach says. Individually, PSDA or durable power of attorney may not adequately convey a patient’s true wishes, because often times the measures involve nothing more than having the patient or family complete a form. “The true marker is a conversation,” he says.
—Andrew Auerbach, MD, University of California San Francisco
By the Numbers
Of 17,097 patients interviewed, only 1,776 (10.4%) had a documented care discussion within 24 hours of hospital admission. The frequency of discussions varied from a low of 2.8% at one institution, to a high of 24.9% at another.
On unadjusted analysis, patients with documented care discussions were more likely to have living wills, durable powers of attorney, or the names of surrogate decision-makers in their charts (P<0.0001 for all categories). These patients were older, more likely to be white, and more likely to be on Medicare, compared to patients without documented care discussions.
The unadjusted analysis also showed patients with care discussions were more likely to be married, but less likely to be living in their own home or apartment, and, not surprisingly, more likely to have been hospitalized at least once within the previous 12 months. Overall, the general health of patients with care discussions was poorer than those without. Patients with documented discussions were more likely to report needing help within the past month with chores or bathing or dressing themselves, than were patients who did not have care discussions. Cancer, depression, and a history of stroke were common among patients with care discussions. Compared to patients without documented discussions, those who did have the discussion appear to want more of a say in their care: they were less likely to agree with the study statements, “I prefer my doctor give me choices regarding my care,” and “I prefer to leave care decisions to my physician.”
The authors found multivariate analysis showed many of these factors turn out to have only a moderate association with a documented care discussion, with adjusted odds ratios of less than 2.0. The strongest predictors were the existence of informal notations describing pre-hospital care wishes, with odds ratios ranging from 3.22 to 11.32, compared to people with no such documentation, and site of enrollment, with odds ratios of 1.74 to 5.14.
The Caregiver Culture
These findings suggest the “caregiver culture” at any given institution is a stronger determinant of a patient participating in a documented care discussion than other, more intuitive factors, such as medical condition or socioeconomic characteristics, or even whether or not the patient has a pre-existing advance directive or durable power of attorney, Dr. Auerbach explains. “It may just be a part of what some hospitals do. It’s driven by what your peers are doing and by local practices.”
Based on the results of the study, Dr. Auerbach and his co-authors suggest simply establishing mandates to document code status on admission probably will not encourage more conversations of this nature, “unless sites also develop an approach to using this newly documented information as a prompt for subsequent discussions.” The Patient Self-Determination Act went into effect in 1991, but its effect on improving communication around end-of-life care remains uncertain, the authors note. It might be why California passed a new law, effective as of January 2009, requiring physicians and health care organizations in the state to provide terminally ill patients who ask about their end-of-life care options with comprehensive information and counseling. Such discussions must cover advance directives, hospice care, and the right to receive palliative care. The new law is the first of its kind in the nation, but proponents of the legislation hope it will serve as a model for other states to adopt.1
“Documentation has an impact on quality of care. Lots of errors are driven by code status,” Dr. Auerbach points out. “If a patient is admitted in the middle of the night, when the doctor checks in on him the next day, he can look at the chart and see that his partner documented the care discussion the night before. That is incredibly helpful.” TH
Norra MacReady is a medical writer based in California.
Reference
1. O’Reilly KB. California law mandates discussing end-of-life options. Am Med News Web site. Available at amaassn.org/amednews/2008/11/10/prsc1110.htm. Last accessed November 6, 2008.
When it comes to discussing a patient’s wishes for code status care, practices at the institution play a more important role than almost any other factor, according to a new study, “Factors Associated with Discussion of Care Plans and Code Status at the Time of Hospital Admission: Results from the Multicenter Hospitalist Study,” in the Journal of Hospital Medicine.
“What was most surprising to me was how variable the discussion rate was,” lead author Andrew Auerbach, MD, tells The Hospitalist. “It had little or nothing to do with how sick the patient was, or with the type of institution in which the discussion took place.”
Hospitalists are no more or less likely to document such discussions than doctors in any other specialty, says Dr. Auerbach, a hospitalist and associate professor of medicine at the University of California San Francisco. He and his colleagues analyzed data from patients admitted to the general medicine services at six academic medical centers as part of the Multicenter Hospitalist Study. Each site complied with requirements established by the Patient Self-Determination Act (PSDA), which says patients must be informed of their right to create an advance directive.
None of the hospitals in the study had established guidelines or formal policies regarding physician-patient discussions about code status or end-of-life care. Patients were interviewed immediately after informed consent was obtained, usually within 24 hours of admission. In each case, the authors determined whether or not the patient had had a care discussion, defined as a documented discussion “between patients (or family) and at least one physician … during the first 24 hours of hospitalization,” the authors write. “Care discussions needed to specify that the person who wrote the note had actually spoken with the patient or their family for the purposes of determining preferences for care, and that this discussion resulted in a specific care plan.”
A notation, such as “the patient continues to want full efforts,” qualified as a care discussion. Less-specific comments, such as “DNR/DNI” or “spoke with family, questions answered,” did not qualify. “These were all really, really sick people, and it was important to assess their wishes for care,” Dr. Auerbach says. Individually, PSDA or durable power of attorney may not adequately convey a patient’s true wishes, because often times the measures involve nothing more than having the patient or family complete a form. “The true marker is a conversation,” he says.
—Andrew Auerbach, MD, University of California San Francisco
By the Numbers
Of 17,097 patients interviewed, only 1,776 (10.4%) had a documented care discussion within 24 hours of hospital admission. The frequency of discussions varied from a low of 2.8% at one institution, to a high of 24.9% at another.
On unadjusted analysis, patients with documented care discussions were more likely to have living wills, durable powers of attorney, or the names of surrogate decision-makers in their charts (P<0.0001 for all categories). These patients were older, more likely to be white, and more likely to be on Medicare, compared to patients without documented care discussions.
The unadjusted analysis also showed patients with care discussions were more likely to be married, but less likely to be living in their own home or apartment, and, not surprisingly, more likely to have been hospitalized at least once within the previous 12 months. Overall, the general health of patients with care discussions was poorer than those without. Patients with documented discussions were more likely to report needing help within the past month with chores or bathing or dressing themselves, than were patients who did not have care discussions. Cancer, depression, and a history of stroke were common among patients with care discussions. Compared to patients without documented discussions, those who did have the discussion appear to want more of a say in their care: they were less likely to agree with the study statements, “I prefer my doctor give me choices regarding my care,” and “I prefer to leave care decisions to my physician.”
The authors found multivariate analysis showed many of these factors turn out to have only a moderate association with a documented care discussion, with adjusted odds ratios of less than 2.0. The strongest predictors were the existence of informal notations describing pre-hospital care wishes, with odds ratios ranging from 3.22 to 11.32, compared to people with no such documentation, and site of enrollment, with odds ratios of 1.74 to 5.14.
The Caregiver Culture
These findings suggest the “caregiver culture” at any given institution is a stronger determinant of a patient participating in a documented care discussion than other, more intuitive factors, such as medical condition or socioeconomic characteristics, or even whether or not the patient has a pre-existing advance directive or durable power of attorney, Dr. Auerbach explains. “It may just be a part of what some hospitals do. It’s driven by what your peers are doing and by local practices.”
Based on the results of the study, Dr. Auerbach and his co-authors suggest simply establishing mandates to document code status on admission probably will not encourage more conversations of this nature, “unless sites also develop an approach to using this newly documented information as a prompt for subsequent discussions.” The Patient Self-Determination Act went into effect in 1991, but its effect on improving communication around end-of-life care remains uncertain, the authors note. It might be why California passed a new law, effective as of January 2009, requiring physicians and health care organizations in the state to provide terminally ill patients who ask about their end-of-life care options with comprehensive information and counseling. Such discussions must cover advance directives, hospice care, and the right to receive palliative care. The new law is the first of its kind in the nation, but proponents of the legislation hope it will serve as a model for other states to adopt.1
“Documentation has an impact on quality of care. Lots of errors are driven by code status,” Dr. Auerbach points out. “If a patient is admitted in the middle of the night, when the doctor checks in on him the next day, he can look at the chart and see that his partner documented the care discussion the night before. That is incredibly helpful.” TH
Norra MacReady is a medical writer based in California.
Reference
1. O’Reilly KB. California law mandates discussing end-of-life options. Am Med News Web site. Available at amaassn.org/amednews/2008/11/10/prsc1110.htm. Last accessed November 6, 2008.