User login
Cytokine release syndrome in severe COVID-19: Is tocilizumab effective?
A large amount of data suggest that mild or severe cytokine storms, accompanied by high expression of interleukin-6 (IL-6), occur in patients with severe coronavirus disease and can be an important cause of death. Blocking the signal transduction pathway of IL-6 is expected to become a new method for the treatment of patients with severe COVID-19, with the IL-6 inhibitor, tocilizumab (Actemra), poised to become an effective drug for these patients, according to the authors of a review published online in the International Journal of Antimicrobial Agents.
The reviewers from China detailed the metabolic pathways and regulation of cytokine release syndrome, especially with respect to what is known about severe COVID-19, and discussed the results of recent trials with tocilizumab, which is currently used for treatment of CRS in a variety of cancers and other metabolic disorders.
Tocilizumab is a recombinant humanized monoclonal antibody against human IL-6 receptor of immunoglobulin IgG1 subtype and has been approved for the treatment of rheumatoid arthritis and systemic juvenile idiopathic arthritis. The antibody specifically binds soluble- and membrane-bound IL-6 receptors (sIL-6R and mIL-6R) and inhibits sIL-6R– and mIL-6R–mediated signal transduction. It has been shown to be effective in the treatment of severe CRS patients. In 2017, the U.S. Food and Drug Administration approved tocilizumab for the treatment of CRS caused by CAR-T (chimeric antigen receptor T-cell immunotherapy) therapy.
A small clinical trial in China examined the effectiveness of tocilizumab in 21 patients who met the criteria for severe or critical COVID-19, including respiratory failure requiring mechanical ventilation, shock, or admission to the ICU with other organ failure. After a few days of tocilizumab treatment, the body temperatures returned to normal (initially, all 21 patients had fevers), and all other symptoms were significantly improved, according to the authors. A total of 75% (15/20) of the patients reduced their oxygen intake, and 1 patient did not need oxygen. CT scanning showed that 90.5% (19/21) of the patients had absorption of pulmonary lesions, and lab tests showed that the proportion of peripheral blood lymphocytes and C-reactive protein in the patients returned to normal.
The main deficiency of the study was that only the level of IL-6 in peripheral blood before treatment with tocilizumab was reported (mean value, 132.38 ± 278.54 pg/mL), but the level of IL-6 following treatment was not given, according to the reviewers. Serum levels of IL-6 in normal patients are undetectable or very low.
Based upon their analysis of COVID-19’s possible mechanism and the small samples of clinical data available, tocilizumab appeared effective, and “we suggest that it should be used in critically ill COVID-19 patients with significantly elevated IL-6,” the authors stated.
“CRS occurs in a large number of patients with severe COVID-19, which is also an important cause of death. IL-6 is the key molecule of CRS, so IL-6R antagonist tocilizumab may be an important drug to save patients’ lives,” the researchers concluded.
This study was supported by China Mega-Project for Infectious Diseases and the China Mega-Project for Innovative Drugs. The authors reported that they had no conflicts.
SOURCE: Zhang C et al. Int J Antimicrobial Agents. 2020. doi. org/10.1016/j.ijantimicag.2020.105954.
A large amount of data suggest that mild or severe cytokine storms, accompanied by high expression of interleukin-6 (IL-6), occur in patients with severe coronavirus disease and can be an important cause of death. Blocking the signal transduction pathway of IL-6 is expected to become a new method for the treatment of patients with severe COVID-19, with the IL-6 inhibitor, tocilizumab (Actemra), poised to become an effective drug for these patients, according to the authors of a review published online in the International Journal of Antimicrobial Agents.
The reviewers from China detailed the metabolic pathways and regulation of cytokine release syndrome, especially with respect to what is known about severe COVID-19, and discussed the results of recent trials with tocilizumab, which is currently used for treatment of CRS in a variety of cancers and other metabolic disorders.
Tocilizumab is a recombinant humanized monoclonal antibody against human IL-6 receptor of immunoglobulin IgG1 subtype and has been approved for the treatment of rheumatoid arthritis and systemic juvenile idiopathic arthritis. The antibody specifically binds soluble- and membrane-bound IL-6 receptors (sIL-6R and mIL-6R) and inhibits sIL-6R– and mIL-6R–mediated signal transduction. It has been shown to be effective in the treatment of severe CRS patients. In 2017, the U.S. Food and Drug Administration approved tocilizumab for the treatment of CRS caused by CAR-T (chimeric antigen receptor T-cell immunotherapy) therapy.
A small clinical trial in China examined the effectiveness of tocilizumab in 21 patients who met the criteria for severe or critical COVID-19, including respiratory failure requiring mechanical ventilation, shock, or admission to the ICU with other organ failure. After a few days of tocilizumab treatment, the body temperatures returned to normal (initially, all 21 patients had fevers), and all other symptoms were significantly improved, according to the authors. A total of 75% (15/20) of the patients reduced their oxygen intake, and 1 patient did not need oxygen. CT scanning showed that 90.5% (19/21) of the patients had absorption of pulmonary lesions, and lab tests showed that the proportion of peripheral blood lymphocytes and C-reactive protein in the patients returned to normal.
The main deficiency of the study was that only the level of IL-6 in peripheral blood before treatment with tocilizumab was reported (mean value, 132.38 ± 278.54 pg/mL), but the level of IL-6 following treatment was not given, according to the reviewers. Serum levels of IL-6 in normal patients are undetectable or very low.
Based upon their analysis of COVID-19’s possible mechanism and the small samples of clinical data available, tocilizumab appeared effective, and “we suggest that it should be used in critically ill COVID-19 patients with significantly elevated IL-6,” the authors stated.
“CRS occurs in a large number of patients with severe COVID-19, which is also an important cause of death. IL-6 is the key molecule of CRS, so IL-6R antagonist tocilizumab may be an important drug to save patients’ lives,” the researchers concluded.
This study was supported by China Mega-Project for Infectious Diseases and the China Mega-Project for Innovative Drugs. The authors reported that they had no conflicts.
SOURCE: Zhang C et al. Int J Antimicrobial Agents. 2020. doi. org/10.1016/j.ijantimicag.2020.105954.
A large amount of data suggest that mild or severe cytokine storms, accompanied by high expression of interleukin-6 (IL-6), occur in patients with severe coronavirus disease and can be an important cause of death. Blocking the signal transduction pathway of IL-6 is expected to become a new method for the treatment of patients with severe COVID-19, with the IL-6 inhibitor, tocilizumab (Actemra), poised to become an effective drug for these patients, according to the authors of a review published online in the International Journal of Antimicrobial Agents.
The reviewers from China detailed the metabolic pathways and regulation of cytokine release syndrome, especially with respect to what is known about severe COVID-19, and discussed the results of recent trials with tocilizumab, which is currently used for treatment of CRS in a variety of cancers and other metabolic disorders.
Tocilizumab is a recombinant humanized monoclonal antibody against human IL-6 receptor of immunoglobulin IgG1 subtype and has been approved for the treatment of rheumatoid arthritis and systemic juvenile idiopathic arthritis. The antibody specifically binds soluble- and membrane-bound IL-6 receptors (sIL-6R and mIL-6R) and inhibits sIL-6R– and mIL-6R–mediated signal transduction. It has been shown to be effective in the treatment of severe CRS patients. In 2017, the U.S. Food and Drug Administration approved tocilizumab for the treatment of CRS caused by CAR-T (chimeric antigen receptor T-cell immunotherapy) therapy.
A small clinical trial in China examined the effectiveness of tocilizumab in 21 patients who met the criteria for severe or critical COVID-19, including respiratory failure requiring mechanical ventilation, shock, or admission to the ICU with other organ failure. After a few days of tocilizumab treatment, the body temperatures returned to normal (initially, all 21 patients had fevers), and all other symptoms were significantly improved, according to the authors. A total of 75% (15/20) of the patients reduced their oxygen intake, and 1 patient did not need oxygen. CT scanning showed that 90.5% (19/21) of the patients had absorption of pulmonary lesions, and lab tests showed that the proportion of peripheral blood lymphocytes and C-reactive protein in the patients returned to normal.
The main deficiency of the study was that only the level of IL-6 in peripheral blood before treatment with tocilizumab was reported (mean value, 132.38 ± 278.54 pg/mL), but the level of IL-6 following treatment was not given, according to the reviewers. Serum levels of IL-6 in normal patients are undetectable or very low.
Based upon their analysis of COVID-19’s possible mechanism and the small samples of clinical data available, tocilizumab appeared effective, and “we suggest that it should be used in critically ill COVID-19 patients with significantly elevated IL-6,” the authors stated.
“CRS occurs in a large number of patients with severe COVID-19, which is also an important cause of death. IL-6 is the key molecule of CRS, so IL-6R antagonist tocilizumab may be an important drug to save patients’ lives,” the researchers concluded.
This study was supported by China Mega-Project for Infectious Diseases and the China Mega-Project for Innovative Drugs. The authors reported that they had no conflicts.
SOURCE: Zhang C et al. Int J Antimicrobial Agents. 2020. doi. org/10.1016/j.ijantimicag.2020.105954.
FROM THE INTERNATIONAL JOURNAL OF ANTIMICROBIAL AGENTS
U.S. hospitals facing severe challenges from COVID-19, HHS report says
Hospitals across the country encountered severe challenges as the first wave of the COVID-19 pandemic swept over them, and they anticipated much worse to come, according to a new report from the Office of Inspector General of the Department of Health and Human Services (HHS).
From March 23 to 27, the OIG interviewed 323 hospitals of several types in 46 states, the District of Columbia, and Puerto Rico. The report it pulled together from these interviews is intended to help HHS manage the crisis, rather than to review its response to the pandemic, the OIG said.
The most significant hospital challenges, the report states, were testing and caring for patients with known or suspected COVID-19 and protecting staff members. In addition, the hospitals faced challenges in maintaining or expanding their capacities to treat COVID-19 patients and ensuring the adequacy of basic supplies.
The critical shortages of ventilators, personal protective equipment (PPE), and test kits in hospitals have been widely reported by the media. But the OIG report also focused on some areas that have received less press attention.
To begin with, the shortage of tests has not only slowed the national response to the pandemic, but has had a major impact on inpatient care, according to the report’s authors. The limited number of test kits means that only symptomatic staff members and patients can be tested; in some hospitals, there aren’t even enough tests for that, and some facilities subdivided the test kits they had, the report states.
Moreover, the test results often took 7 days or more to come back from commercial or government labs, the report states. In the meantime, symptomatic patients were presumed to have the coronavirus. While awaiting the results, they had to stay in the hospital, using beds and requiring staff who could otherwise have been assigned to other patients.
The doctors and nurse who cared for these presumptive COVID-19 patients also had to take time suiting up in PPE before seeing them; much of that scarce PPE was wasted on those who were later found not to have the illness.
As one administrator explained to OIG, “Sitting with 60 patients with presumed positives in our hospital isn’t healthy for anybody.”
Delayed test results also reduced hospitals’ ability to provide care by sidelining clinicians who reported COVID-19 symptoms. In one hospital, 20% to 25% of staff were determined to be presumptively positive for COVID-19. As a result of their tests not being analyzed promptly, these doctors and nurses were prevented from providing clinical services for longer than necessary.
Supply Shortages
The report also described some factors contributing to mask shortages. Because of the fear factor, for example, all staff members in one hospital were wearing masks, instead of just those in designated areas. An administrator said the hospital was using 2,000 masks a day, 10 times the number before the COVID-19 crisis.
Another hospital received 2,300 N95 masks from a state reserve, but they were unusable because the elastic bands had dry-rotted.
Meanwhile, some vendors were profiteering. Masks that used to cost 50 cents now sold for $6 each, one administrator said.
To combat the supply chain disruptions, some facilities were buying PPE from nontraditional sources such as online retailers, home supply stores, paint stores, autobody supply shops, and beauty salons. Other hospitals were using non–medical-grade PPE such as construction masks and handmade masks and gowns.
Other hospitals reported they were conserving and reusing PPE to stretch their supplies. In some cases, they had even changed policies to reduce the extent and frequency of patient interactions with clinicians so the latter would have to change their gear less often.
Shortages of other critical supplies and materials were also reported. Hospitals were running out of supplies that supported patient rooms, such as IV poles, medical gas, linens, toilet paper, and food.
Hospitals across the country were also expecting or experiencing a shortage of ventilators, although none said any patients had been denied access to them. Some institutions were adapting anesthesia machines and single-use emergency transport ventilators.
Also concerning to hospitals was the shortage of intensive-care specialists and nurses to operate the ventilators and care for critically ill patients. Some facilities were training anesthesiologists, hospitalists, and other nonintensivists on how to use the lifesaving equipment.
Meanwhile, patients with COVID-19 symptoms were continuing to show up in droves at emergency departments. Hospitals were concerned about potential shortages of ICU beds, negative-pressure rooms, and isolation units. Given limited bed availability, some administrators said, it was getting hard to separate COVID-19 from non–COVID-19 patients.
What Hospitals Want
As the COVID-19 crisis continues to mount, many hospitals are facing financial emergencies as well, the report noted.
“Hospitals described increasing costs and decreasing revenues as a threat to their financial viability. Hospitals reported that ceasing elective procedures and other services decreased revenues at the same time that their costs have increased as they prepare for a potential surge of patients. Many hospitals reported that their cash reserves were quickly depleting, which could disrupt ongoing hospital operations,” the authors write.
This report was conducted a few days before the passage of the CURES Act, which earmarked $100 billion for hospitals on the frontline of the crisis. As a recent analysis of financial hospital data revealed, however, even with the 20% bump in Medicare payments for COVID-19 care that this cash infusion represents, many hospitals will face a cash-flow crunch within 60 to 90 days, as reported by Medscape Medical News.
Besides higher Medicare payments, the OIG report said, hospitals wanted the government to drop the 14-day waiting period for reimbursement and to offer them loans and grants.
Hospitals also want federal and state governments to relax regulations on professional licensing of, and business relationships with, doctors and other clinicians. They’d like the government to:
- Let them reassign licensed professionals within their hospitals and across healthcare networks
- Provide flexibility with respect to licensed professionals practicing across state lines
- Provide relief from regulations that may restrict using contracted staff or physicians based on business relationships
This article first appeared on Medscape.com.
Hospitals across the country encountered severe challenges as the first wave of the COVID-19 pandemic swept over them, and they anticipated much worse to come, according to a new report from the Office of Inspector General of the Department of Health and Human Services (HHS).
From March 23 to 27, the OIG interviewed 323 hospitals of several types in 46 states, the District of Columbia, and Puerto Rico. The report it pulled together from these interviews is intended to help HHS manage the crisis, rather than to review its response to the pandemic, the OIG said.
The most significant hospital challenges, the report states, were testing and caring for patients with known or suspected COVID-19 and protecting staff members. In addition, the hospitals faced challenges in maintaining or expanding their capacities to treat COVID-19 patients and ensuring the adequacy of basic supplies.
The critical shortages of ventilators, personal protective equipment (PPE), and test kits in hospitals have been widely reported by the media. But the OIG report also focused on some areas that have received less press attention.
To begin with, the shortage of tests has not only slowed the national response to the pandemic, but has had a major impact on inpatient care, according to the report’s authors. The limited number of test kits means that only symptomatic staff members and patients can be tested; in some hospitals, there aren’t even enough tests for that, and some facilities subdivided the test kits they had, the report states.
Moreover, the test results often took 7 days or more to come back from commercial or government labs, the report states. In the meantime, symptomatic patients were presumed to have the coronavirus. While awaiting the results, they had to stay in the hospital, using beds and requiring staff who could otherwise have been assigned to other patients.
The doctors and nurse who cared for these presumptive COVID-19 patients also had to take time suiting up in PPE before seeing them; much of that scarce PPE was wasted on those who were later found not to have the illness.
As one administrator explained to OIG, “Sitting with 60 patients with presumed positives in our hospital isn’t healthy for anybody.”
Delayed test results also reduced hospitals’ ability to provide care by sidelining clinicians who reported COVID-19 symptoms. In one hospital, 20% to 25% of staff were determined to be presumptively positive for COVID-19. As a result of their tests not being analyzed promptly, these doctors and nurses were prevented from providing clinical services for longer than necessary.
Supply Shortages
The report also described some factors contributing to mask shortages. Because of the fear factor, for example, all staff members in one hospital were wearing masks, instead of just those in designated areas. An administrator said the hospital was using 2,000 masks a day, 10 times the number before the COVID-19 crisis.
Another hospital received 2,300 N95 masks from a state reserve, but they were unusable because the elastic bands had dry-rotted.
Meanwhile, some vendors were profiteering. Masks that used to cost 50 cents now sold for $6 each, one administrator said.
To combat the supply chain disruptions, some facilities were buying PPE from nontraditional sources such as online retailers, home supply stores, paint stores, autobody supply shops, and beauty salons. Other hospitals were using non–medical-grade PPE such as construction masks and handmade masks and gowns.
Other hospitals reported they were conserving and reusing PPE to stretch their supplies. In some cases, they had even changed policies to reduce the extent and frequency of patient interactions with clinicians so the latter would have to change their gear less often.
Shortages of other critical supplies and materials were also reported. Hospitals were running out of supplies that supported patient rooms, such as IV poles, medical gas, linens, toilet paper, and food.
Hospitals across the country were also expecting or experiencing a shortage of ventilators, although none said any patients had been denied access to them. Some institutions were adapting anesthesia machines and single-use emergency transport ventilators.
Also concerning to hospitals was the shortage of intensive-care specialists and nurses to operate the ventilators and care for critically ill patients. Some facilities were training anesthesiologists, hospitalists, and other nonintensivists on how to use the lifesaving equipment.
Meanwhile, patients with COVID-19 symptoms were continuing to show up in droves at emergency departments. Hospitals were concerned about potential shortages of ICU beds, negative-pressure rooms, and isolation units. Given limited bed availability, some administrators said, it was getting hard to separate COVID-19 from non–COVID-19 patients.
What Hospitals Want
As the COVID-19 crisis continues to mount, many hospitals are facing financial emergencies as well, the report noted.
“Hospitals described increasing costs and decreasing revenues as a threat to their financial viability. Hospitals reported that ceasing elective procedures and other services decreased revenues at the same time that their costs have increased as they prepare for a potential surge of patients. Many hospitals reported that their cash reserves were quickly depleting, which could disrupt ongoing hospital operations,” the authors write.
This report was conducted a few days before the passage of the CURES Act, which earmarked $100 billion for hospitals on the frontline of the crisis. As a recent analysis of financial hospital data revealed, however, even with the 20% bump in Medicare payments for COVID-19 care that this cash infusion represents, many hospitals will face a cash-flow crunch within 60 to 90 days, as reported by Medscape Medical News.
Besides higher Medicare payments, the OIG report said, hospitals wanted the government to drop the 14-day waiting period for reimbursement and to offer them loans and grants.
Hospitals also want federal and state governments to relax regulations on professional licensing of, and business relationships with, doctors and other clinicians. They’d like the government to:
- Let them reassign licensed professionals within their hospitals and across healthcare networks
- Provide flexibility with respect to licensed professionals practicing across state lines
- Provide relief from regulations that may restrict using contracted staff or physicians based on business relationships
This article first appeared on Medscape.com.
Hospitals across the country encountered severe challenges as the first wave of the COVID-19 pandemic swept over them, and they anticipated much worse to come, according to a new report from the Office of Inspector General of the Department of Health and Human Services (HHS).
From March 23 to 27, the OIG interviewed 323 hospitals of several types in 46 states, the District of Columbia, and Puerto Rico. The report it pulled together from these interviews is intended to help HHS manage the crisis, rather than to review its response to the pandemic, the OIG said.
The most significant hospital challenges, the report states, were testing and caring for patients with known or suspected COVID-19 and protecting staff members. In addition, the hospitals faced challenges in maintaining or expanding their capacities to treat COVID-19 patients and ensuring the adequacy of basic supplies.
The critical shortages of ventilators, personal protective equipment (PPE), and test kits in hospitals have been widely reported by the media. But the OIG report also focused on some areas that have received less press attention.
To begin with, the shortage of tests has not only slowed the national response to the pandemic, but has had a major impact on inpatient care, according to the report’s authors. The limited number of test kits means that only symptomatic staff members and patients can be tested; in some hospitals, there aren’t even enough tests for that, and some facilities subdivided the test kits they had, the report states.
Moreover, the test results often took 7 days or more to come back from commercial or government labs, the report states. In the meantime, symptomatic patients were presumed to have the coronavirus. While awaiting the results, they had to stay in the hospital, using beds and requiring staff who could otherwise have been assigned to other patients.
The doctors and nurse who cared for these presumptive COVID-19 patients also had to take time suiting up in PPE before seeing them; much of that scarce PPE was wasted on those who were later found not to have the illness.
As one administrator explained to OIG, “Sitting with 60 patients with presumed positives in our hospital isn’t healthy for anybody.”
Delayed test results also reduced hospitals’ ability to provide care by sidelining clinicians who reported COVID-19 symptoms. In one hospital, 20% to 25% of staff were determined to be presumptively positive for COVID-19. As a result of their tests not being analyzed promptly, these doctors and nurses were prevented from providing clinical services for longer than necessary.
Supply Shortages
The report also described some factors contributing to mask shortages. Because of the fear factor, for example, all staff members in one hospital were wearing masks, instead of just those in designated areas. An administrator said the hospital was using 2,000 masks a day, 10 times the number before the COVID-19 crisis.
Another hospital received 2,300 N95 masks from a state reserve, but they were unusable because the elastic bands had dry-rotted.
Meanwhile, some vendors were profiteering. Masks that used to cost 50 cents now sold for $6 each, one administrator said.
To combat the supply chain disruptions, some facilities were buying PPE from nontraditional sources such as online retailers, home supply stores, paint stores, autobody supply shops, and beauty salons. Other hospitals were using non–medical-grade PPE such as construction masks and handmade masks and gowns.
Other hospitals reported they were conserving and reusing PPE to stretch their supplies. In some cases, they had even changed policies to reduce the extent and frequency of patient interactions with clinicians so the latter would have to change their gear less often.
Shortages of other critical supplies and materials were also reported. Hospitals were running out of supplies that supported patient rooms, such as IV poles, medical gas, linens, toilet paper, and food.
Hospitals across the country were also expecting or experiencing a shortage of ventilators, although none said any patients had been denied access to them. Some institutions were adapting anesthesia machines and single-use emergency transport ventilators.
Also concerning to hospitals was the shortage of intensive-care specialists and nurses to operate the ventilators and care for critically ill patients. Some facilities were training anesthesiologists, hospitalists, and other nonintensivists on how to use the lifesaving equipment.
Meanwhile, patients with COVID-19 symptoms were continuing to show up in droves at emergency departments. Hospitals were concerned about potential shortages of ICU beds, negative-pressure rooms, and isolation units. Given limited bed availability, some administrators said, it was getting hard to separate COVID-19 from non–COVID-19 patients.
What Hospitals Want
As the COVID-19 crisis continues to mount, many hospitals are facing financial emergencies as well, the report noted.
“Hospitals described increasing costs and decreasing revenues as a threat to their financial viability. Hospitals reported that ceasing elective procedures and other services decreased revenues at the same time that their costs have increased as they prepare for a potential surge of patients. Many hospitals reported that their cash reserves were quickly depleting, which could disrupt ongoing hospital operations,” the authors write.
This report was conducted a few days before the passage of the CURES Act, which earmarked $100 billion for hospitals on the frontline of the crisis. As a recent analysis of financial hospital data revealed, however, even with the 20% bump in Medicare payments for COVID-19 care that this cash infusion represents, many hospitals will face a cash-flow crunch within 60 to 90 days, as reported by Medscape Medical News.
Besides higher Medicare payments, the OIG report said, hospitals wanted the government to drop the 14-day waiting period for reimbursement and to offer them loans and grants.
Hospitals also want federal and state governments to relax regulations on professional licensing of, and business relationships with, doctors and other clinicians. They’d like the government to:
- Let them reassign licensed professionals within their hospitals and across healthcare networks
- Provide flexibility with respect to licensed professionals practicing across state lines
- Provide relief from regulations that may restrict using contracted staff or physicians based on business relationships
This article first appeared on Medscape.com.
Aerosolization of COVID-19 and Contamination Risks During Respiratory Treatments
Beyond asthma and chronic obstructive pulmonary disease (COPD), inhalation therapy is a mainstay in the management of bronchiectasis, cystic fibrosis, and pulmonary artery hypertension. Several US Food and Drug Administration off-label indications for inhalational medications include hypoxia secondary to acute respiratory distress syndrome (ARDS) and intraoperative and postoperative pulmonary hypertension during and following cardiac surgery, respectively.1-11 Therapeutic delivery of aerosols to the lung may be provided via nebulization, pressurized metered-dose inhalers (pMDI), and other devices (eg, dry powder inhalers, soft-mist inhalers, and smart inhalers).12 The most common aerosolized medications given in the clinical setting are bronchodilators.12
Product selection is often guided by practice guidelines (Table 1), consideration of the formulation’s advantages and disadvantages (Table 2), and/or formulary considerations. For example, current guidelines for COPD state that there is no evidence for superiority of nebulized bronchodilator therapy over handheld devices in patients who can use them properly.2 Due to equivalence, nebulized formulations are commonly used in hospitals, emergency departments (EDs) and ambulatory clinics based on the drug’s unit cost. In contrast, a pMDI is often more cost-effective for use in ambulatory patients who are administering multiple doses from the same canister.
The World Health Organization (WHO) and the Centers for Disease Control and Prevention (CDC) recommend droplet and contact precautions for all patients suspected or diagnosed with novel coronavirus-19 (COVID-19).13,14 Airborne precautions must be applied when performing aerosol-generating medical procedures (AGMPs), including but not limited to, open suctioning of the respiratory tract, intubation, bronchoscopy, and cardiopulmonary resuscitation (CPR). Data from the severe acute respiratory syndrome (SARS-CoV) epidemic suggest that nebulization of medication is also an AGMP.15-17
Institutions must ensure that their health care workers (HCWs) are wearing appropriate personal protective equipment (PPE) including gloves, long-sleeved gowns, eye protection, and fit-tested particulate respirators (N95 mask) for airborne procedures and are carefully discarding PPE after use.13,14 Due to severe shortages in available respirators in the US supply chain, the CDC has temporarily modified WHO recommendations. Face masks are now an acceptable alternative to protect HCWs from splashes and sprays from procedures not likely to generate aerosols and for cleaning of rooms, although there is no evidence to support this decision.
Internationally, HCWs are falling ill with COVID-19. Data from Italy and Spain show that about 9% to 13% of these countries’ cases are HCWs.18,19 Within the US, the Ohio health department reports approximately 16% of cases are HCWs.20 It is possible that 20% of frontline HCWs will become infected.21 Evolving laboratory research shows that COVID-19 remains viable in aerosols for up to 3 hours postaerosolization, thus making aerosol transmission plausible.22 Nebulizers convert liquids into aerosols and during dispersal may potentially cause secondary inhalation of fugitive emissions.23 Since interim CDC infection control guidance is to allow only essential personnel to enter the room of patients with COVID-19, many facilities will rely on their frontline nursing staff to clean and disinfect high-touch surfaces following routine care activities.24
Achieving adequate fomite disinfection following viral aerosolization may pose a significant problem for any patient receiving scheduled doses of nebulized medications. Additionally, for personnel who clean rooms following intermittent drug nebulization while wearing PPE that includes a face mask, protection from aerosolized virus may be inadequate. Subsequently, fugitive emissions from nebulized medications may potentially contribute to both nosocomial COVID-19 transmission and viral infections in the medical staff until proven otherwise by studies conducted outside of the laboratory. Prevention of infection in the medical staff is imperative since federal health care systems cannot sustain a significant loss of its workforce.
Recommendations
We recommend that health care systems stop business as usual and adopt public health recommendations issued by Canadian and Hong Kong health care authorities for the management of suspected or confirmed COVID-19 disease.25-28 We have further clarified and expanded on these interventions. During viral pandemics, prescribers and health care systems should:
- Deprescribe nebulized therapies on medical wards and intensive care units as an infection control measure. Also avoid use in any outpatient health care setting (eg, community-based clinics, EDs, triage).
- Avoid initiation of nebulized unproven therapies (eg, n-acetylcysteine, hypertonic saline).1
- Use alternative bronchodilator formulations as appropriate (eg, oral β-2 agonist, recognizing its slower onset) before prescribing nebulized agents to patients who are uncooperative or unable to follow directions needed to use a pMDI with a spacer or have experienced a prior poor response to a pMDI with spacer (eg, OptiChamber Diamond, Philips).25,27
- Limit nebulized drug utilization (eg, bronchodilators, epoprostenol) to patients who are on mechanical ventilation and will receive nebulized therapies via a closed system or to patients housed in negative pressure hospital rooms.22 Use a viral filter (eg, Salter Labs system) to decrease the spread of infection for those receiving epoprostenol via face mask.25
- Adjust procurement practices (eg, pharmacy, logistics) to address the transition from nebulized drugs to alternatives.
- Add a safety net to the drug-ordering process by restricting new orders for nebulized therapies to the prior authorization process.27 Apply the exclusion criterion of suspected or definite COVID-19.
- Add a safety net to environmental service practices. Nursing staff should track patients who received ≥ 1 nebulizations via open (before diagnosis) or closed systems so that staff wear suitable PPE to include a N-95 mask while cleaning the room.
Conclusions
To implement the aggressive infection control guidance promulgated here, we recommend collaboration with infection control, pharmacy service (eg, prior authorization team, clinical pharmacy team, and procurement team), respiratory therapy, pulmonary and other critical care physicians, EDs, CPR committee, and other stakeholders. When making significant transitions in clinical care during a viral pandemic, guidelines must be timely, use imperative wording, and consist of easily identifiable education and/or instructions for the affected frontline staff in order to change attitudes.29 Additionally, when transitioning from nebulized bronchodilators to pMDI, educational in-services should be provided to frontline staff to avoid misconceptions regarding pMDI treatment efficacy and patients’ ability to use their pMDI with spacer.30
Acknowledgments
This material is the result of work supported with resources and the use of facilities at the VA Tennessee Valley Healthcare System in Nashville.
1. Strickland SL, Rubin BK, Haas CF, Volsko TA, Drescher GS, O’Malley CA. AARC Clinical Practice Guideline: effectiveness of pharmacologic airway clearance therapies in hospitalized patients. Respir Care. 2015;60(7):1071-1077.
2. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease. 2020 GOLD Report. https://goldcopd.org/gold-reports/. Accessed March 26, 2020.
3. Van Geffen WH, Douma WR, Slebos DJ, Kerstjens HAM. Bronchodilators delivered by nebulizer versus pMDI with spacer or DPI for exacerbations of COPD (Review). Cochrane Database Syst Rev. 2016;8:CD011826.
4. Global Initiative for Asthma. https://ginasthma.org/wp-content/uploads/2019/06/GINA-2019-main-report-June-2019-wms.pdf. Accessed March 26, 2020.
5. Global Initiative for Asthma. Difficult-to-treat and severe asthma in adolescent and adult patients: diagnosis and management. https://ginasthma.org/wp-content/uploads/2019/04/GINA-Severe-asthma-Pocket-Guide-v2.0-wms-1.pdf. Accessed March 26, 2020.
6. Cates CJ, Welsh EJ, Rowe BH. Holding chambers (spacers) versus nebulizers for beta-agonist treatment of acute asthma. Cochrane Database Syst Rev. 2013;9:CD000052.
7. Welsh EJ, Evans DJ, Fowler SJ, Spencer S. Interventions for bronchiectasis: an overview of Cochrane systematic reviews. Cochrane Database Syst Rev. 2015;7:CD010337.
8. Taichman DB, Ornelas J, Chung L, et al. Pharmacologic therapy for pulmonary arterial hypertension in adults: CHEST Guideline and Expert Panel Report. CHEST. 2014;146(2):449-475.
9. Griffiths MJD, McAuley DF, Perkins GD, et al. Guidelines on the management of acute respiratory distress syndrome. BMJ Open Resp Res. 2019;6(1):e000420.
10. McGinn K, Reichert M. A comparison of inhaled nitric oxide versus inhaled epoprostenol for acute pulmonary hypertension following cardiac surgery. Ann Pharmacother. 2016;50(1):22-26.
11. Dzierba AL, Abel EE, Buckley MS, Lat I. A review of inhaled nitric oxide and aerosolized epoprostenol in acute lung injury or acute respiratory distress syndrome. Pharmacotherapy. 2014;34(3):279-290.
12. Pleasants RA, Hess DR. Aerosol delivery devices for obstructive lung diseases. Respir Care. 2018;63(6):708-733.
13. World Health Organization. Clinical management of severe acute respiratory infection when novel coronavirus (nCoV) infection is suspected. https://www.who.int/publications-detail/clinical-management-of-severe-acute-respiratory-infection-when-novel-coronavirus-(ncov)-infection-is-suspected Accessed March 26, 2020.
14. Centers for Disease Control and Prevention. Interim clinical guidance for management of patients with confirmed coronavirus disease (COVID-19). https://www.cdc.gov/coronavirus/2019-ncov/hcp/clinical-guidance-management-patients.html. Revised March 7, 2020. Accessed March 26, 2020.
15. Wong RSM, Hui DS. Index patient and SARS outbreak in Hong Kong. Emerg Infect Dis. 2004;10(2):339-341.
16. Wong T-W, Lee C-K, Tam W, et al; Outbreak Study Group. Emerg Infect Dis. 2004;10(2):269-276.
17. Seto WH, Tsang D, Yung RWH, et al; Advisors of Expert SARS group of Hospital Authority. Effectiveness of precautions against droplets and contact in prevention of nosocomial transmission of severe acute respiratory syndrome (SARS). Lancet. 2003;361(9368):1519-1520.
18. Livingston E, Bucher K. Coronavirus Disease 2019 (COVID-19) in Italy. https://jamanetwork.com/journals/jama/fullarticle/2763401?resultClick=1. Published March 17, 2020. Accessed March 26, 2020.
19. Jones S. Spain: doctors struggle to cope as 514 die from coronavirus in a day. The Guardian. March 24, 2020. https://www.theguardian.com/world/2020/mar/24/spain-doctors-lack-protection-coronavirus-covid-19. Accessed March 27, 2020.
20. 16% of Ohio’s diagnosed COVID-19 cases are healthcare workers. https://www.wlwt.com/article/16-of-ohio-s-diagnosed-covid-19-cases-are-healthcare-workers/31930566#. Updated March 25, 2020. Accessed March 27, 2020.
21. Remuzzi A, Remuzzi G. COVID-19 and Italy: what next? Lancet. http://www.thelancet.com/journals/lancet/article/PIIS0140-6736(20)30627-9/fulltext. Accessed March 27, 2020.
22. van Doremalen N, Bushmaker T, Morris DH, et al. Aerosol and surface stability of SARS-CoV-2 as Compared with SARS-CoV-1 [published online ahead of print, 2020 Mar 17]. N Engl J Med. 2020;10.1056/NEJMc2004973.
23. McGrath JA, O’Sullivan A, Bennett G, et al. Investigation of the quantity of exhaled aerosol released into the environment during nebulization. Pharmaceutics. 2019;11(2):75.
24. Centers for Disease Control and Prevention. Healthcare Infection prevention and control FAQs for COVID-19. https://www.cdc.gov/coronavirus/2019-ncov/infection-control/infection-prevention-control-faq.html. Revised March 24, 2020. Accessed March 26, 2020.
25. Practice standards of respiratory procedures: post SARS era. Use of aerosolized medications. December 2003. http://www.hkresp.com/hkts.php?page=page/hkts/detail&meid=93742. Accessed March 26, 2020.
26. Wax RS, Christian MD. Practical recommendations for critical care and anesthesiology teams caring for novel coronavirus (2019-nCoV) patients. Can J Anesth. 2020. [ePub ahead of print.]
27. Newhouse MT. RE: transmission of coronavirus by nebulizer- as serious, underappreciated risk! https://www.cmaj.ca/content/re-transmission-corona-virus-nebulizer-serious-underappreciated-risk. Accessed March 26, 2020. [ePub ahead of print.]
28. Moira C-Y. Severe acute respiratory syndrome (SARS) and healthcare workers. Int J Occup Environ Health. 2004;10(4):421-427.
29. Timen A, Hulscher MEJL, Rust L, et al. Barriers to implementing infection prevention and control guidelines during crises: experiences of health care professionals. Am J Infect Control. 2010;38(9):726-733.
30. Khoo SM, Tan LK, Said N, Lim TK. Metered-dose inhaler with spacer instead of nebulizer during the outbreak of severe acute respiratory syndrome in Singapore. Respir Care. 2009;54(7):855-860.
Beyond asthma and chronic obstructive pulmonary disease (COPD), inhalation therapy is a mainstay in the management of bronchiectasis, cystic fibrosis, and pulmonary artery hypertension. Several US Food and Drug Administration off-label indications for inhalational medications include hypoxia secondary to acute respiratory distress syndrome (ARDS) and intraoperative and postoperative pulmonary hypertension during and following cardiac surgery, respectively.1-11 Therapeutic delivery of aerosols to the lung may be provided via nebulization, pressurized metered-dose inhalers (pMDI), and other devices (eg, dry powder inhalers, soft-mist inhalers, and smart inhalers).12 The most common aerosolized medications given in the clinical setting are bronchodilators.12
Product selection is often guided by practice guidelines (Table 1), consideration of the formulation’s advantages and disadvantages (Table 2), and/or formulary considerations. For example, current guidelines for COPD state that there is no evidence for superiority of nebulized bronchodilator therapy over handheld devices in patients who can use them properly.2 Due to equivalence, nebulized formulations are commonly used in hospitals, emergency departments (EDs) and ambulatory clinics based on the drug’s unit cost. In contrast, a pMDI is often more cost-effective for use in ambulatory patients who are administering multiple doses from the same canister.
The World Health Organization (WHO) and the Centers for Disease Control and Prevention (CDC) recommend droplet and contact precautions for all patients suspected or diagnosed with novel coronavirus-19 (COVID-19).13,14 Airborne precautions must be applied when performing aerosol-generating medical procedures (AGMPs), including but not limited to, open suctioning of the respiratory tract, intubation, bronchoscopy, and cardiopulmonary resuscitation (CPR). Data from the severe acute respiratory syndrome (SARS-CoV) epidemic suggest that nebulization of medication is also an AGMP.15-17
Institutions must ensure that their health care workers (HCWs) are wearing appropriate personal protective equipment (PPE) including gloves, long-sleeved gowns, eye protection, and fit-tested particulate respirators (N95 mask) for airborne procedures and are carefully discarding PPE after use.13,14 Due to severe shortages in available respirators in the US supply chain, the CDC has temporarily modified WHO recommendations. Face masks are now an acceptable alternative to protect HCWs from splashes and sprays from procedures not likely to generate aerosols and for cleaning of rooms, although there is no evidence to support this decision.
Internationally, HCWs are falling ill with COVID-19. Data from Italy and Spain show that about 9% to 13% of these countries’ cases are HCWs.18,19 Within the US, the Ohio health department reports approximately 16% of cases are HCWs.20 It is possible that 20% of frontline HCWs will become infected.21 Evolving laboratory research shows that COVID-19 remains viable in aerosols for up to 3 hours postaerosolization, thus making aerosol transmission plausible.22 Nebulizers convert liquids into aerosols and during dispersal may potentially cause secondary inhalation of fugitive emissions.23 Since interim CDC infection control guidance is to allow only essential personnel to enter the room of patients with COVID-19, many facilities will rely on their frontline nursing staff to clean and disinfect high-touch surfaces following routine care activities.24
Achieving adequate fomite disinfection following viral aerosolization may pose a significant problem for any patient receiving scheduled doses of nebulized medications. Additionally, for personnel who clean rooms following intermittent drug nebulization while wearing PPE that includes a face mask, protection from aerosolized virus may be inadequate. Subsequently, fugitive emissions from nebulized medications may potentially contribute to both nosocomial COVID-19 transmission and viral infections in the medical staff until proven otherwise by studies conducted outside of the laboratory. Prevention of infection in the medical staff is imperative since federal health care systems cannot sustain a significant loss of its workforce.
Recommendations
We recommend that health care systems stop business as usual and adopt public health recommendations issued by Canadian and Hong Kong health care authorities for the management of suspected or confirmed COVID-19 disease.25-28 We have further clarified and expanded on these interventions. During viral pandemics, prescribers and health care systems should:
- Deprescribe nebulized therapies on medical wards and intensive care units as an infection control measure. Also avoid use in any outpatient health care setting (eg, community-based clinics, EDs, triage).
- Avoid initiation of nebulized unproven therapies (eg, n-acetylcysteine, hypertonic saline).1
- Use alternative bronchodilator formulations as appropriate (eg, oral β-2 agonist, recognizing its slower onset) before prescribing nebulized agents to patients who are uncooperative or unable to follow directions needed to use a pMDI with a spacer or have experienced a prior poor response to a pMDI with spacer (eg, OptiChamber Diamond, Philips).25,27
- Limit nebulized drug utilization (eg, bronchodilators, epoprostenol) to patients who are on mechanical ventilation and will receive nebulized therapies via a closed system or to patients housed in negative pressure hospital rooms.22 Use a viral filter (eg, Salter Labs system) to decrease the spread of infection for those receiving epoprostenol via face mask.25
- Adjust procurement practices (eg, pharmacy, logistics) to address the transition from nebulized drugs to alternatives.
- Add a safety net to the drug-ordering process by restricting new orders for nebulized therapies to the prior authorization process.27 Apply the exclusion criterion of suspected or definite COVID-19.
- Add a safety net to environmental service practices. Nursing staff should track patients who received ≥ 1 nebulizations via open (before diagnosis) or closed systems so that staff wear suitable PPE to include a N-95 mask while cleaning the room.
Conclusions
To implement the aggressive infection control guidance promulgated here, we recommend collaboration with infection control, pharmacy service (eg, prior authorization team, clinical pharmacy team, and procurement team), respiratory therapy, pulmonary and other critical care physicians, EDs, CPR committee, and other stakeholders. When making significant transitions in clinical care during a viral pandemic, guidelines must be timely, use imperative wording, and consist of easily identifiable education and/or instructions for the affected frontline staff in order to change attitudes.29 Additionally, when transitioning from nebulized bronchodilators to pMDI, educational in-services should be provided to frontline staff to avoid misconceptions regarding pMDI treatment efficacy and patients’ ability to use their pMDI with spacer.30
Acknowledgments
This material is the result of work supported with resources and the use of facilities at the VA Tennessee Valley Healthcare System in Nashville.
Beyond asthma and chronic obstructive pulmonary disease (COPD), inhalation therapy is a mainstay in the management of bronchiectasis, cystic fibrosis, and pulmonary artery hypertension. Several US Food and Drug Administration off-label indications for inhalational medications include hypoxia secondary to acute respiratory distress syndrome (ARDS) and intraoperative and postoperative pulmonary hypertension during and following cardiac surgery, respectively.1-11 Therapeutic delivery of aerosols to the lung may be provided via nebulization, pressurized metered-dose inhalers (pMDI), and other devices (eg, dry powder inhalers, soft-mist inhalers, and smart inhalers).12 The most common aerosolized medications given in the clinical setting are bronchodilators.12
Product selection is often guided by practice guidelines (Table 1), consideration of the formulation’s advantages and disadvantages (Table 2), and/or formulary considerations. For example, current guidelines for COPD state that there is no evidence for superiority of nebulized bronchodilator therapy over handheld devices in patients who can use them properly.2 Due to equivalence, nebulized formulations are commonly used in hospitals, emergency departments (EDs) and ambulatory clinics based on the drug’s unit cost. In contrast, a pMDI is often more cost-effective for use in ambulatory patients who are administering multiple doses from the same canister.
The World Health Organization (WHO) and the Centers for Disease Control and Prevention (CDC) recommend droplet and contact precautions for all patients suspected or diagnosed with novel coronavirus-19 (COVID-19).13,14 Airborne precautions must be applied when performing aerosol-generating medical procedures (AGMPs), including but not limited to, open suctioning of the respiratory tract, intubation, bronchoscopy, and cardiopulmonary resuscitation (CPR). Data from the severe acute respiratory syndrome (SARS-CoV) epidemic suggest that nebulization of medication is also an AGMP.15-17
Institutions must ensure that their health care workers (HCWs) are wearing appropriate personal protective equipment (PPE) including gloves, long-sleeved gowns, eye protection, and fit-tested particulate respirators (N95 mask) for airborne procedures and are carefully discarding PPE after use.13,14 Due to severe shortages in available respirators in the US supply chain, the CDC has temporarily modified WHO recommendations. Face masks are now an acceptable alternative to protect HCWs from splashes and sprays from procedures not likely to generate aerosols and for cleaning of rooms, although there is no evidence to support this decision.
Internationally, HCWs are falling ill with COVID-19. Data from Italy and Spain show that about 9% to 13% of these countries’ cases are HCWs.18,19 Within the US, the Ohio health department reports approximately 16% of cases are HCWs.20 It is possible that 20% of frontline HCWs will become infected.21 Evolving laboratory research shows that COVID-19 remains viable in aerosols for up to 3 hours postaerosolization, thus making aerosol transmission plausible.22 Nebulizers convert liquids into aerosols and during dispersal may potentially cause secondary inhalation of fugitive emissions.23 Since interim CDC infection control guidance is to allow only essential personnel to enter the room of patients with COVID-19, many facilities will rely on their frontline nursing staff to clean and disinfect high-touch surfaces following routine care activities.24
Achieving adequate fomite disinfection following viral aerosolization may pose a significant problem for any patient receiving scheduled doses of nebulized medications. Additionally, for personnel who clean rooms following intermittent drug nebulization while wearing PPE that includes a face mask, protection from aerosolized virus may be inadequate. Subsequently, fugitive emissions from nebulized medications may potentially contribute to both nosocomial COVID-19 transmission and viral infections in the medical staff until proven otherwise by studies conducted outside of the laboratory. Prevention of infection in the medical staff is imperative since federal health care systems cannot sustain a significant loss of its workforce.
Recommendations
We recommend that health care systems stop business as usual and adopt public health recommendations issued by Canadian and Hong Kong health care authorities for the management of suspected or confirmed COVID-19 disease.25-28 We have further clarified and expanded on these interventions. During viral pandemics, prescribers and health care systems should:
- Deprescribe nebulized therapies on medical wards and intensive care units as an infection control measure. Also avoid use in any outpatient health care setting (eg, community-based clinics, EDs, triage).
- Avoid initiation of nebulized unproven therapies (eg, n-acetylcysteine, hypertonic saline).1
- Use alternative bronchodilator formulations as appropriate (eg, oral β-2 agonist, recognizing its slower onset) before prescribing nebulized agents to patients who are uncooperative or unable to follow directions needed to use a pMDI with a spacer or have experienced a prior poor response to a pMDI with spacer (eg, OptiChamber Diamond, Philips).25,27
- Limit nebulized drug utilization (eg, bronchodilators, epoprostenol) to patients who are on mechanical ventilation and will receive nebulized therapies via a closed system or to patients housed in negative pressure hospital rooms.22 Use a viral filter (eg, Salter Labs system) to decrease the spread of infection for those receiving epoprostenol via face mask.25
- Adjust procurement practices (eg, pharmacy, logistics) to address the transition from nebulized drugs to alternatives.
- Add a safety net to the drug-ordering process by restricting new orders for nebulized therapies to the prior authorization process.27 Apply the exclusion criterion of suspected or definite COVID-19.
- Add a safety net to environmental service practices. Nursing staff should track patients who received ≥ 1 nebulizations via open (before diagnosis) or closed systems so that staff wear suitable PPE to include a N-95 mask while cleaning the room.
Conclusions
To implement the aggressive infection control guidance promulgated here, we recommend collaboration with infection control, pharmacy service (eg, prior authorization team, clinical pharmacy team, and procurement team), respiratory therapy, pulmonary and other critical care physicians, EDs, CPR committee, and other stakeholders. When making significant transitions in clinical care during a viral pandemic, guidelines must be timely, use imperative wording, and consist of easily identifiable education and/or instructions for the affected frontline staff in order to change attitudes.29 Additionally, when transitioning from nebulized bronchodilators to pMDI, educational in-services should be provided to frontline staff to avoid misconceptions regarding pMDI treatment efficacy and patients’ ability to use their pMDI with spacer.30
Acknowledgments
This material is the result of work supported with resources and the use of facilities at the VA Tennessee Valley Healthcare System in Nashville.
1. Strickland SL, Rubin BK, Haas CF, Volsko TA, Drescher GS, O’Malley CA. AARC Clinical Practice Guideline: effectiveness of pharmacologic airway clearance therapies in hospitalized patients. Respir Care. 2015;60(7):1071-1077.
2. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease. 2020 GOLD Report. https://goldcopd.org/gold-reports/. Accessed March 26, 2020.
3. Van Geffen WH, Douma WR, Slebos DJ, Kerstjens HAM. Bronchodilators delivered by nebulizer versus pMDI with spacer or DPI for exacerbations of COPD (Review). Cochrane Database Syst Rev. 2016;8:CD011826.
4. Global Initiative for Asthma. https://ginasthma.org/wp-content/uploads/2019/06/GINA-2019-main-report-June-2019-wms.pdf. Accessed March 26, 2020.
5. Global Initiative for Asthma. Difficult-to-treat and severe asthma in adolescent and adult patients: diagnosis and management. https://ginasthma.org/wp-content/uploads/2019/04/GINA-Severe-asthma-Pocket-Guide-v2.0-wms-1.pdf. Accessed March 26, 2020.
6. Cates CJ, Welsh EJ, Rowe BH. Holding chambers (spacers) versus nebulizers for beta-agonist treatment of acute asthma. Cochrane Database Syst Rev. 2013;9:CD000052.
7. Welsh EJ, Evans DJ, Fowler SJ, Spencer S. Interventions for bronchiectasis: an overview of Cochrane systematic reviews. Cochrane Database Syst Rev. 2015;7:CD010337.
8. Taichman DB, Ornelas J, Chung L, et al. Pharmacologic therapy for pulmonary arterial hypertension in adults: CHEST Guideline and Expert Panel Report. CHEST. 2014;146(2):449-475.
9. Griffiths MJD, McAuley DF, Perkins GD, et al. Guidelines on the management of acute respiratory distress syndrome. BMJ Open Resp Res. 2019;6(1):e000420.
10. McGinn K, Reichert M. A comparison of inhaled nitric oxide versus inhaled epoprostenol for acute pulmonary hypertension following cardiac surgery. Ann Pharmacother. 2016;50(1):22-26.
11. Dzierba AL, Abel EE, Buckley MS, Lat I. A review of inhaled nitric oxide and aerosolized epoprostenol in acute lung injury or acute respiratory distress syndrome. Pharmacotherapy. 2014;34(3):279-290.
12. Pleasants RA, Hess DR. Aerosol delivery devices for obstructive lung diseases. Respir Care. 2018;63(6):708-733.
13. World Health Organization. Clinical management of severe acute respiratory infection when novel coronavirus (nCoV) infection is suspected. https://www.who.int/publications-detail/clinical-management-of-severe-acute-respiratory-infection-when-novel-coronavirus-(ncov)-infection-is-suspected Accessed March 26, 2020.
14. Centers for Disease Control and Prevention. Interim clinical guidance for management of patients with confirmed coronavirus disease (COVID-19). https://www.cdc.gov/coronavirus/2019-ncov/hcp/clinical-guidance-management-patients.html. Revised March 7, 2020. Accessed March 26, 2020.
15. Wong RSM, Hui DS. Index patient and SARS outbreak in Hong Kong. Emerg Infect Dis. 2004;10(2):339-341.
16. Wong T-W, Lee C-K, Tam W, et al; Outbreak Study Group. Emerg Infect Dis. 2004;10(2):269-276.
17. Seto WH, Tsang D, Yung RWH, et al; Advisors of Expert SARS group of Hospital Authority. Effectiveness of precautions against droplets and contact in prevention of nosocomial transmission of severe acute respiratory syndrome (SARS). Lancet. 2003;361(9368):1519-1520.
18. Livingston E, Bucher K. Coronavirus Disease 2019 (COVID-19) in Italy. https://jamanetwork.com/journals/jama/fullarticle/2763401?resultClick=1. Published March 17, 2020. Accessed March 26, 2020.
19. Jones S. Spain: doctors struggle to cope as 514 die from coronavirus in a day. The Guardian. March 24, 2020. https://www.theguardian.com/world/2020/mar/24/spain-doctors-lack-protection-coronavirus-covid-19. Accessed March 27, 2020.
20. 16% of Ohio’s diagnosed COVID-19 cases are healthcare workers. https://www.wlwt.com/article/16-of-ohio-s-diagnosed-covid-19-cases-are-healthcare-workers/31930566#. Updated March 25, 2020. Accessed March 27, 2020.
21. Remuzzi A, Remuzzi G. COVID-19 and Italy: what next? Lancet. http://www.thelancet.com/journals/lancet/article/PIIS0140-6736(20)30627-9/fulltext. Accessed March 27, 2020.
22. van Doremalen N, Bushmaker T, Morris DH, et al. Aerosol and surface stability of SARS-CoV-2 as Compared with SARS-CoV-1 [published online ahead of print, 2020 Mar 17]. N Engl J Med. 2020;10.1056/NEJMc2004973.
23. McGrath JA, O’Sullivan A, Bennett G, et al. Investigation of the quantity of exhaled aerosol released into the environment during nebulization. Pharmaceutics. 2019;11(2):75.
24. Centers for Disease Control and Prevention. Healthcare Infection prevention and control FAQs for COVID-19. https://www.cdc.gov/coronavirus/2019-ncov/infection-control/infection-prevention-control-faq.html. Revised March 24, 2020. Accessed March 26, 2020.
25. Practice standards of respiratory procedures: post SARS era. Use of aerosolized medications. December 2003. http://www.hkresp.com/hkts.php?page=page/hkts/detail&meid=93742. Accessed March 26, 2020.
26. Wax RS, Christian MD. Practical recommendations for critical care and anesthesiology teams caring for novel coronavirus (2019-nCoV) patients. Can J Anesth. 2020. [ePub ahead of print.]
27. Newhouse MT. RE: transmission of coronavirus by nebulizer- as serious, underappreciated risk! https://www.cmaj.ca/content/re-transmission-corona-virus-nebulizer-serious-underappreciated-risk. Accessed March 26, 2020. [ePub ahead of print.]
28. Moira C-Y. Severe acute respiratory syndrome (SARS) and healthcare workers. Int J Occup Environ Health. 2004;10(4):421-427.
29. Timen A, Hulscher MEJL, Rust L, et al. Barriers to implementing infection prevention and control guidelines during crises: experiences of health care professionals. Am J Infect Control. 2010;38(9):726-733.
30. Khoo SM, Tan LK, Said N, Lim TK. Metered-dose inhaler with spacer instead of nebulizer during the outbreak of severe acute respiratory syndrome in Singapore. Respir Care. 2009;54(7):855-860.
1. Strickland SL, Rubin BK, Haas CF, Volsko TA, Drescher GS, O’Malley CA. AARC Clinical Practice Guideline: effectiveness of pharmacologic airway clearance therapies in hospitalized patients. Respir Care. 2015;60(7):1071-1077.
2. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease. 2020 GOLD Report. https://goldcopd.org/gold-reports/. Accessed March 26, 2020.
3. Van Geffen WH, Douma WR, Slebos DJ, Kerstjens HAM. Bronchodilators delivered by nebulizer versus pMDI with spacer or DPI for exacerbations of COPD (Review). Cochrane Database Syst Rev. 2016;8:CD011826.
4. Global Initiative for Asthma. https://ginasthma.org/wp-content/uploads/2019/06/GINA-2019-main-report-June-2019-wms.pdf. Accessed March 26, 2020.
5. Global Initiative for Asthma. Difficult-to-treat and severe asthma in adolescent and adult patients: diagnosis and management. https://ginasthma.org/wp-content/uploads/2019/04/GINA-Severe-asthma-Pocket-Guide-v2.0-wms-1.pdf. Accessed March 26, 2020.
6. Cates CJ, Welsh EJ, Rowe BH. Holding chambers (spacers) versus nebulizers for beta-agonist treatment of acute asthma. Cochrane Database Syst Rev. 2013;9:CD000052.
7. Welsh EJ, Evans DJ, Fowler SJ, Spencer S. Interventions for bronchiectasis: an overview of Cochrane systematic reviews. Cochrane Database Syst Rev. 2015;7:CD010337.
8. Taichman DB, Ornelas J, Chung L, et al. Pharmacologic therapy for pulmonary arterial hypertension in adults: CHEST Guideline and Expert Panel Report. CHEST. 2014;146(2):449-475.
9. Griffiths MJD, McAuley DF, Perkins GD, et al. Guidelines on the management of acute respiratory distress syndrome. BMJ Open Resp Res. 2019;6(1):e000420.
10. McGinn K, Reichert M. A comparison of inhaled nitric oxide versus inhaled epoprostenol for acute pulmonary hypertension following cardiac surgery. Ann Pharmacother. 2016;50(1):22-26.
11. Dzierba AL, Abel EE, Buckley MS, Lat I. A review of inhaled nitric oxide and aerosolized epoprostenol in acute lung injury or acute respiratory distress syndrome. Pharmacotherapy. 2014;34(3):279-290.
12. Pleasants RA, Hess DR. Aerosol delivery devices for obstructive lung diseases. Respir Care. 2018;63(6):708-733.
13. World Health Organization. Clinical management of severe acute respiratory infection when novel coronavirus (nCoV) infection is suspected. https://www.who.int/publications-detail/clinical-management-of-severe-acute-respiratory-infection-when-novel-coronavirus-(ncov)-infection-is-suspected Accessed March 26, 2020.
14. Centers for Disease Control and Prevention. Interim clinical guidance for management of patients with confirmed coronavirus disease (COVID-19). https://www.cdc.gov/coronavirus/2019-ncov/hcp/clinical-guidance-management-patients.html. Revised March 7, 2020. Accessed March 26, 2020.
15. Wong RSM, Hui DS. Index patient and SARS outbreak in Hong Kong. Emerg Infect Dis. 2004;10(2):339-341.
16. Wong T-W, Lee C-K, Tam W, et al; Outbreak Study Group. Emerg Infect Dis. 2004;10(2):269-276.
17. Seto WH, Tsang D, Yung RWH, et al; Advisors of Expert SARS group of Hospital Authority. Effectiveness of precautions against droplets and contact in prevention of nosocomial transmission of severe acute respiratory syndrome (SARS). Lancet. 2003;361(9368):1519-1520.
18. Livingston E, Bucher K. Coronavirus Disease 2019 (COVID-19) in Italy. https://jamanetwork.com/journals/jama/fullarticle/2763401?resultClick=1. Published March 17, 2020. Accessed March 26, 2020.
19. Jones S. Spain: doctors struggle to cope as 514 die from coronavirus in a day. The Guardian. March 24, 2020. https://www.theguardian.com/world/2020/mar/24/spain-doctors-lack-protection-coronavirus-covid-19. Accessed March 27, 2020.
20. 16% of Ohio’s diagnosed COVID-19 cases are healthcare workers. https://www.wlwt.com/article/16-of-ohio-s-diagnosed-covid-19-cases-are-healthcare-workers/31930566#. Updated March 25, 2020. Accessed March 27, 2020.
21. Remuzzi A, Remuzzi G. COVID-19 and Italy: what next? Lancet. http://www.thelancet.com/journals/lancet/article/PIIS0140-6736(20)30627-9/fulltext. Accessed March 27, 2020.
22. van Doremalen N, Bushmaker T, Morris DH, et al. Aerosol and surface stability of SARS-CoV-2 as Compared with SARS-CoV-1 [published online ahead of print, 2020 Mar 17]. N Engl J Med. 2020;10.1056/NEJMc2004973.
23. McGrath JA, O’Sullivan A, Bennett G, et al. Investigation of the quantity of exhaled aerosol released into the environment during nebulization. Pharmaceutics. 2019;11(2):75.
24. Centers for Disease Control and Prevention. Healthcare Infection prevention and control FAQs for COVID-19. https://www.cdc.gov/coronavirus/2019-ncov/infection-control/infection-prevention-control-faq.html. Revised March 24, 2020. Accessed March 26, 2020.
25. Practice standards of respiratory procedures: post SARS era. Use of aerosolized medications. December 2003. http://www.hkresp.com/hkts.php?page=page/hkts/detail&meid=93742. Accessed March 26, 2020.
26. Wax RS, Christian MD. Practical recommendations for critical care and anesthesiology teams caring for novel coronavirus (2019-nCoV) patients. Can J Anesth. 2020. [ePub ahead of print.]
27. Newhouse MT. RE: transmission of coronavirus by nebulizer- as serious, underappreciated risk! https://www.cmaj.ca/content/re-transmission-corona-virus-nebulizer-serious-underappreciated-risk. Accessed March 26, 2020. [ePub ahead of print.]
28. Moira C-Y. Severe acute respiratory syndrome (SARS) and healthcare workers. Int J Occup Environ Health. 2004;10(4):421-427.
29. Timen A, Hulscher MEJL, Rust L, et al. Barriers to implementing infection prevention and control guidelines during crises: experiences of health care professionals. Am J Infect Control. 2010;38(9):726-733.
30. Khoo SM, Tan LK, Said N, Lim TK. Metered-dose inhaler with spacer instead of nebulizer during the outbreak of severe acute respiratory syndrome in Singapore. Respir Care. 2009;54(7):855-860.
Many children with COVID-19 don’t have cough or fever
according to the Centers for Disease and Prevention Control.
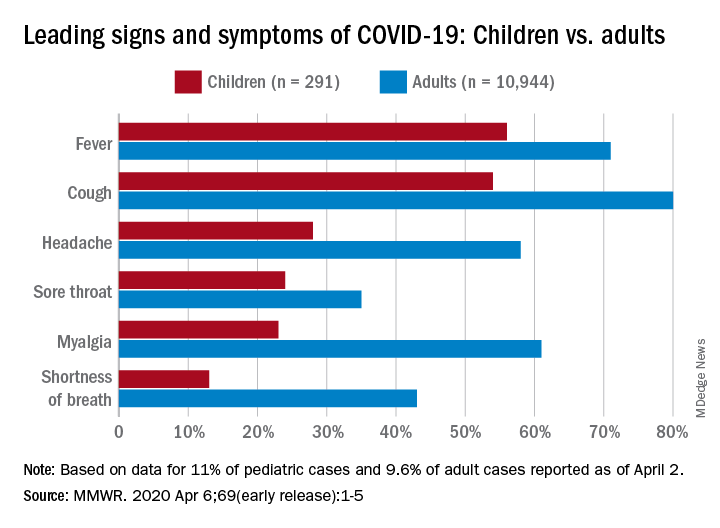
Among pediatric patients younger than 18 years in the United States, 73% had at least one of the trio of symptoms, compared with 93% of adults aged 18-64, noted Lucy A. McNamara, PhD, and the CDC’s COVID-19 response team, based on a preliminary analysis of the 149,082 cases reported as of April 2.
By a small margin, fever – present in 58% of pediatric patients – was the most common sign or symptom of COVID-19, compared with cough at 54% and shortness of breath in 13%. In adults, cough (81%) was seen most often, followed by fever (71%) and shortness of breath (43%), the investigators reported in the MMWR.
In both children and adults, headache and myalgia were more common than shortness of breath, as was sore throat in children, the team added.
“These findings are largely consistent with a report on pediatric COVID-19 patients aged <16 years in China, which found that only 41.5% of pediatric patients had fever [and] 48.5% had cough,” they wrote.
The CDC analysis of pediatric patients was limited by its small sample size, with data on signs and symptoms available for only 11% (291) of the 2,572 children known to have COVID-19 as of April 2. The adult population included 10,944 individuals, who represented 9.6% of the 113,985 U.S. patients aged 18-65, the response team said.
“As the number of COVID-19 cases continues to increase in many parts of the United States, it will be important to adapt COVID-19 surveillance strategies to maintain collection of critical case information without overburdening jurisdiction health departments,” they said.
SOURCE: McNamara LA et al. MMWR 2020 Apr 6;69(early release):1-5.
according to the Centers for Disease and Prevention Control.

Among pediatric patients younger than 18 years in the United States, 73% had at least one of the trio of symptoms, compared with 93% of adults aged 18-64, noted Lucy A. McNamara, PhD, and the CDC’s COVID-19 response team, based on a preliminary analysis of the 149,082 cases reported as of April 2.
By a small margin, fever – present in 58% of pediatric patients – was the most common sign or symptom of COVID-19, compared with cough at 54% and shortness of breath in 13%. In adults, cough (81%) was seen most often, followed by fever (71%) and shortness of breath (43%), the investigators reported in the MMWR.
In both children and adults, headache and myalgia were more common than shortness of breath, as was sore throat in children, the team added.
“These findings are largely consistent with a report on pediatric COVID-19 patients aged <16 years in China, which found that only 41.5% of pediatric patients had fever [and] 48.5% had cough,” they wrote.
The CDC analysis of pediatric patients was limited by its small sample size, with data on signs and symptoms available for only 11% (291) of the 2,572 children known to have COVID-19 as of April 2. The adult population included 10,944 individuals, who represented 9.6% of the 113,985 U.S. patients aged 18-65, the response team said.
“As the number of COVID-19 cases continues to increase in many parts of the United States, it will be important to adapt COVID-19 surveillance strategies to maintain collection of critical case information without overburdening jurisdiction health departments,” they said.
SOURCE: McNamara LA et al. MMWR 2020 Apr 6;69(early release):1-5.
according to the Centers for Disease and Prevention Control.

Among pediatric patients younger than 18 years in the United States, 73% had at least one of the trio of symptoms, compared with 93% of adults aged 18-64, noted Lucy A. McNamara, PhD, and the CDC’s COVID-19 response team, based on a preliminary analysis of the 149,082 cases reported as of April 2.
By a small margin, fever – present in 58% of pediatric patients – was the most common sign or symptom of COVID-19, compared with cough at 54% and shortness of breath in 13%. In adults, cough (81%) was seen most often, followed by fever (71%) and shortness of breath (43%), the investigators reported in the MMWR.
In both children and adults, headache and myalgia were more common than shortness of breath, as was sore throat in children, the team added.
“These findings are largely consistent with a report on pediatric COVID-19 patients aged <16 years in China, which found that only 41.5% of pediatric patients had fever [and] 48.5% had cough,” they wrote.
The CDC analysis of pediatric patients was limited by its small sample size, with data on signs and symptoms available for only 11% (291) of the 2,572 children known to have COVID-19 as of April 2. The adult population included 10,944 individuals, who represented 9.6% of the 113,985 U.S. patients aged 18-65, the response team said.
“As the number of COVID-19 cases continues to increase in many parts of the United States, it will be important to adapt COVID-19 surveillance strategies to maintain collection of critical case information without overburdening jurisdiction health departments,” they said.
SOURCE: McNamara LA et al. MMWR 2020 Apr 6;69(early release):1-5.
FROM MMWR
AAP issues guidance on managing infants born to mothers with COVID-19
“Pediatric cases of COVID-19 are so far reported as less severe than disease occurring among older individuals,” Karen M. Puopolo, MD, PhD, a neonatologist and chief of the section on newborn pediatrics at Pennsylvania Hospital, Philadelphia, and coauthors wrote in the 18-page document, which was released on April 2, 2020, along with an abbreviated “Frequently Asked Questions” summary. However, one study of children with COVID-19 in China found that 12% of confirmed cases occurred among 731 infants aged less than 1 year; 24% of those 86 infants “suffered severe or critical illness” (Pediatrics. 2020 March. doi: 10.1542/peds.2020-0702). There were no deaths reported among these infants. Other case reports have documented COVID-19 in children aged as young as 2 days.
The document, which was assembled by members of the AAP Committee on Fetus and Newborn, Section on Neonatal Perinatal Medicine, and Committee on Infectious Diseases, pointed out that “considerable uncertainty” exists about the possibility for vertical transmission of SARS-CoV-2 from infected pregnant women to their newborns. “Evidence-based guidelines for managing antenatal, intrapartum, and neonatal care around COVID-19 would require an understanding of whether the virus can be transmitted transplacentally; a determination of which maternal body fluids may be infectious; and data of adequate statistical power that describe which maternal, intrapartum, and neonatal factors influence perinatal transmission,” according to the document. “In the midst of the pandemic these data do not exist, with only limited information currently available to address these issues.”
Based on the best available evidence, the guidance authors recommend that clinicians temporarily separate newborns from affected mothers to minimize the risk of postnatal infant infection from maternal respiratory secretions. “Newborns should be bathed as soon as reasonably possible after birth to remove virus potentially present on skin surfaces,” they wrote. “Clinical staff should use airborne, droplet, and contact precautions until newborn virologic status is known to be negative by SARS-CoV-2 [polymerase chain reaction] testing.”
While SARS-CoV-2 has not been detected in breast milk to date, the authors noted that mothers with COVID-19 can express breast milk to be fed to their infants by uninfected caregivers until specific maternal criteria are met. In addition, infants born to mothers with COVID-19 should be tested for SARS-CoV-2 at 24 hours and, if still in the birth facility, at 48 hours after birth. Centers with limited resources for testing may make individual risk/benefit decisions regarding testing.
For infants infected with SARS-CoV-2 but have no symptoms of the disease, they “may be discharged home on a case-by-case basis with appropriate precautions and plans for frequent outpatient follow-up contacts (either by phone, telemedicine, or in office) through 14 days after birth,” according to the document.
If both infant and mother are discharged from the hospital and the mother still has COVID-19 symptoms, she should maintain at least 6 feet of distance from the baby; if she is in closer proximity she should use a mask and hand hygiene. The mother can stop such precautions until she is afebrile without the use of antipyretics for at least 72 hours, and it is at least 7 days since her symptoms first occurred.
In cases where infants require ongoing neonatal intensive care, mothers infected with COVID-19 should not visit their newborn until she is afebrile without the use of antipyretics for at least 72 hours, her respiratory symptoms are improved, and she has negative results of a molecular assay for detection of SARS-CoV-2 from at least two consecutive nasopharyngeal swab specimens collected at least 24 hours apart.
“Pediatric cases of COVID-19 are so far reported as less severe than disease occurring among older individuals,” Karen M. Puopolo, MD, PhD, a neonatologist and chief of the section on newborn pediatrics at Pennsylvania Hospital, Philadelphia, and coauthors wrote in the 18-page document, which was released on April 2, 2020, along with an abbreviated “Frequently Asked Questions” summary. However, one study of children with COVID-19 in China found that 12% of confirmed cases occurred among 731 infants aged less than 1 year; 24% of those 86 infants “suffered severe or critical illness” (Pediatrics. 2020 March. doi: 10.1542/peds.2020-0702). There were no deaths reported among these infants. Other case reports have documented COVID-19 in children aged as young as 2 days.
The document, which was assembled by members of the AAP Committee on Fetus and Newborn, Section on Neonatal Perinatal Medicine, and Committee on Infectious Diseases, pointed out that “considerable uncertainty” exists about the possibility for vertical transmission of SARS-CoV-2 from infected pregnant women to their newborns. “Evidence-based guidelines for managing antenatal, intrapartum, and neonatal care around COVID-19 would require an understanding of whether the virus can be transmitted transplacentally; a determination of which maternal body fluids may be infectious; and data of adequate statistical power that describe which maternal, intrapartum, and neonatal factors influence perinatal transmission,” according to the document. “In the midst of the pandemic these data do not exist, with only limited information currently available to address these issues.”
Based on the best available evidence, the guidance authors recommend that clinicians temporarily separate newborns from affected mothers to minimize the risk of postnatal infant infection from maternal respiratory secretions. “Newborns should be bathed as soon as reasonably possible after birth to remove virus potentially present on skin surfaces,” they wrote. “Clinical staff should use airborne, droplet, and contact precautions until newborn virologic status is known to be negative by SARS-CoV-2 [polymerase chain reaction] testing.”
While SARS-CoV-2 has not been detected in breast milk to date, the authors noted that mothers with COVID-19 can express breast milk to be fed to their infants by uninfected caregivers until specific maternal criteria are met. In addition, infants born to mothers with COVID-19 should be tested for SARS-CoV-2 at 24 hours and, if still in the birth facility, at 48 hours after birth. Centers with limited resources for testing may make individual risk/benefit decisions regarding testing.
For infants infected with SARS-CoV-2 but have no symptoms of the disease, they “may be discharged home on a case-by-case basis with appropriate precautions and plans for frequent outpatient follow-up contacts (either by phone, telemedicine, or in office) through 14 days after birth,” according to the document.
If both infant and mother are discharged from the hospital and the mother still has COVID-19 symptoms, she should maintain at least 6 feet of distance from the baby; if she is in closer proximity she should use a mask and hand hygiene. The mother can stop such precautions until she is afebrile without the use of antipyretics for at least 72 hours, and it is at least 7 days since her symptoms first occurred.
In cases where infants require ongoing neonatal intensive care, mothers infected with COVID-19 should not visit their newborn until she is afebrile without the use of antipyretics for at least 72 hours, her respiratory symptoms are improved, and she has negative results of a molecular assay for detection of SARS-CoV-2 from at least two consecutive nasopharyngeal swab specimens collected at least 24 hours apart.
“Pediatric cases of COVID-19 are so far reported as less severe than disease occurring among older individuals,” Karen M. Puopolo, MD, PhD, a neonatologist and chief of the section on newborn pediatrics at Pennsylvania Hospital, Philadelphia, and coauthors wrote in the 18-page document, which was released on April 2, 2020, along with an abbreviated “Frequently Asked Questions” summary. However, one study of children with COVID-19 in China found that 12% of confirmed cases occurred among 731 infants aged less than 1 year; 24% of those 86 infants “suffered severe or critical illness” (Pediatrics. 2020 March. doi: 10.1542/peds.2020-0702). There were no deaths reported among these infants. Other case reports have documented COVID-19 in children aged as young as 2 days.
The document, which was assembled by members of the AAP Committee on Fetus and Newborn, Section on Neonatal Perinatal Medicine, and Committee on Infectious Diseases, pointed out that “considerable uncertainty” exists about the possibility for vertical transmission of SARS-CoV-2 from infected pregnant women to their newborns. “Evidence-based guidelines for managing antenatal, intrapartum, and neonatal care around COVID-19 would require an understanding of whether the virus can be transmitted transplacentally; a determination of which maternal body fluids may be infectious; and data of adequate statistical power that describe which maternal, intrapartum, and neonatal factors influence perinatal transmission,” according to the document. “In the midst of the pandemic these data do not exist, with only limited information currently available to address these issues.”
Based on the best available evidence, the guidance authors recommend that clinicians temporarily separate newborns from affected mothers to minimize the risk of postnatal infant infection from maternal respiratory secretions. “Newborns should be bathed as soon as reasonably possible after birth to remove virus potentially present on skin surfaces,” they wrote. “Clinical staff should use airborne, droplet, and contact precautions until newborn virologic status is known to be negative by SARS-CoV-2 [polymerase chain reaction] testing.”
While SARS-CoV-2 has not been detected in breast milk to date, the authors noted that mothers with COVID-19 can express breast milk to be fed to their infants by uninfected caregivers until specific maternal criteria are met. In addition, infants born to mothers with COVID-19 should be tested for SARS-CoV-2 at 24 hours and, if still in the birth facility, at 48 hours after birth. Centers with limited resources for testing may make individual risk/benefit decisions regarding testing.
For infants infected with SARS-CoV-2 but have no symptoms of the disease, they “may be discharged home on a case-by-case basis with appropriate precautions and plans for frequent outpatient follow-up contacts (either by phone, telemedicine, or in office) through 14 days after birth,” according to the document.
If both infant and mother are discharged from the hospital and the mother still has COVID-19 symptoms, she should maintain at least 6 feet of distance from the baby; if she is in closer proximity she should use a mask and hand hygiene. The mother can stop such precautions until she is afebrile without the use of antipyretics for at least 72 hours, and it is at least 7 days since her symptoms first occurred.
In cases where infants require ongoing neonatal intensive care, mothers infected with COVID-19 should not visit their newborn until she is afebrile without the use of antipyretics for at least 72 hours, her respiratory symptoms are improved, and she has negative results of a molecular assay for detection of SARS-CoV-2 from at least two consecutive nasopharyngeal swab specimens collected at least 24 hours apart.
Practicing solo and feeling grateful – despite COVID-19
I know that the world has gone upside down. It’s a nightmare, and people are filled with fear, and death is everywhere. In my little bubble of a world, however, I’ve been doing well.
I can’t lose my job, because I am my job. I’m a solo practitioner and have been for more than a decade. The restrictions to stay at home have not affected me, because I have a home office. Besides, I’m an introvert and see myself as a bit of a recluse, so the social distancing hasn’t been stressful. Conducting appointments by phone rather than face to face hasn’t undermined my work, since I can do everything that I do in my office over the phone. But I do it now in sweats and at my desk in my bedroom more often than not. I am prepared for a decrease in income as people lose their jobs, but that hasn’t happened yet. There are still people out there who are very motivated to come off their medications holistically. No rest for the wicked, as the saying goes.
On an emotional level, I feel calm because I’m not attached to material things, though I like them when they’re here. My children and friends have remained healthy, so I am grateful for that. I feel grounded in my belief that life goes on one way or another, and I trust in God to direct me wherever I need to go. Socially, I’ve been forced to be less lazy and cook more at home. As a result: less salt, MSG, and greasy food. I’ve spent a lot less on restaurants this past month and am eating less since I have to eat whatever I cook.
Can a person be more pandemic proof? I was joking with a friend about how pandemic-friendly my lifestyle is: spiritually, mentally, emotionally, physically, and socially. Oh, did I forget to mention the year supply of supplements in my office closet? They were for my patients, but those whole food green and red powders may come in handy, just in case.
So, that is how things are going for me. Please don’t hate me for not freaking out. When I read the news, I feel very sad for people who are suffering. I get angry at the politicians who can’t get their egos out of the way. But, I look at the sunshine outside my window, and I feel grateful that, at least in my case, I am not adding to the burden of suffering in the world. Not yet, anyway. I will keep trying to do the little bit that I do to help others for as long as I can.
Dr. Lee specializes in integrative and holistic psychiatry and has a private practice in Gaithersburg, Md. She has no disclosures.
I know that the world has gone upside down. It’s a nightmare, and people are filled with fear, and death is everywhere. In my little bubble of a world, however, I’ve been doing well.
I can’t lose my job, because I am my job. I’m a solo practitioner and have been for more than a decade. The restrictions to stay at home have not affected me, because I have a home office. Besides, I’m an introvert and see myself as a bit of a recluse, so the social distancing hasn’t been stressful. Conducting appointments by phone rather than face to face hasn’t undermined my work, since I can do everything that I do in my office over the phone. But I do it now in sweats and at my desk in my bedroom more often than not. I am prepared for a decrease in income as people lose their jobs, but that hasn’t happened yet. There are still people out there who are very motivated to come off their medications holistically. No rest for the wicked, as the saying goes.
On an emotional level, I feel calm because I’m not attached to material things, though I like them when they’re here. My children and friends have remained healthy, so I am grateful for that. I feel grounded in my belief that life goes on one way or another, and I trust in God to direct me wherever I need to go. Socially, I’ve been forced to be less lazy and cook more at home. As a result: less salt, MSG, and greasy food. I’ve spent a lot less on restaurants this past month and am eating less since I have to eat whatever I cook.
Can a person be more pandemic proof? I was joking with a friend about how pandemic-friendly my lifestyle is: spiritually, mentally, emotionally, physically, and socially. Oh, did I forget to mention the year supply of supplements in my office closet? They were for my patients, but those whole food green and red powders may come in handy, just in case.
So, that is how things are going for me. Please don’t hate me for not freaking out. When I read the news, I feel very sad for people who are suffering. I get angry at the politicians who can’t get their egos out of the way. But, I look at the sunshine outside my window, and I feel grateful that, at least in my case, I am not adding to the burden of suffering in the world. Not yet, anyway. I will keep trying to do the little bit that I do to help others for as long as I can.
Dr. Lee specializes in integrative and holistic psychiatry and has a private practice in Gaithersburg, Md. She has no disclosures.
I know that the world has gone upside down. It’s a nightmare, and people are filled with fear, and death is everywhere. In my little bubble of a world, however, I’ve been doing well.
I can’t lose my job, because I am my job. I’m a solo practitioner and have been for more than a decade. The restrictions to stay at home have not affected me, because I have a home office. Besides, I’m an introvert and see myself as a bit of a recluse, so the social distancing hasn’t been stressful. Conducting appointments by phone rather than face to face hasn’t undermined my work, since I can do everything that I do in my office over the phone. But I do it now in sweats and at my desk in my bedroom more often than not. I am prepared for a decrease in income as people lose their jobs, but that hasn’t happened yet. There are still people out there who are very motivated to come off their medications holistically. No rest for the wicked, as the saying goes.
On an emotional level, I feel calm because I’m not attached to material things, though I like them when they’re here. My children and friends have remained healthy, so I am grateful for that. I feel grounded in my belief that life goes on one way or another, and I trust in God to direct me wherever I need to go. Socially, I’ve been forced to be less lazy and cook more at home. As a result: less salt, MSG, and greasy food. I’ve spent a lot less on restaurants this past month and am eating less since I have to eat whatever I cook.
Can a person be more pandemic proof? I was joking with a friend about how pandemic-friendly my lifestyle is: spiritually, mentally, emotionally, physically, and socially. Oh, did I forget to mention the year supply of supplements in my office closet? They were for my patients, but those whole food green and red powders may come in handy, just in case.
So, that is how things are going for me. Please don’t hate me for not freaking out. When I read the news, I feel very sad for people who are suffering. I get angry at the politicians who can’t get their egos out of the way. But, I look at the sunshine outside my window, and I feel grateful that, at least in my case, I am not adding to the burden of suffering in the world. Not yet, anyway. I will keep trying to do the little bit that I do to help others for as long as I can.
Dr. Lee specializes in integrative and holistic psychiatry and has a private practice in Gaithersburg, Md. She has no disclosures.
Which of the changes that coronavirus has forced upon us will remain?
Eventually this strange Twilight Zone world of coronavirus will end and life will return to normal.
But obviously it won’t be the same, and like everyone else I wonder what will be different.
Telemedicine is one obvious change in my world, though I don’t know how much yet (granted, no one else does, either). I’m seeing a handful of people that way, limited to established patients, where we’re discussing chronic issues or reviewing recent test results.
If I have to see a new patient or an established one with an urgent issue, I’m still willing to meet them at my office (wearing masks and washing hands frequently). In neurology, a lot still depends on a decent exam. It’s pretty hard to check reflexes, sensory modalities, and muscle tone over the phone. If you think a malpractice attorney is going to give you a pass because you missed something by not examining a patient because of coronavirus ... think again.
I’m not sure how the whole telemedicine thing will play out after the dust settles, at least not at my little practice. I’m currently seeing patients by FaceTime and Skype, neither of which is considered HIPAA compliant. The requirement has been waived during the crisis to make sure people can still see doctors, but I don’t see it lasting beyond that. Privacy will always be a central concern in medicine.
When they declare the pandemic over and say I can’t use FaceTime or Skype anymore, that will likely end my use of such. While there are HIPAA-compliant telemedicine services out there, in a small practice I don’t have the time or money to invest in them.
I also wonder how outcomes will change. I suspect the research-minded will be analyzing 2019 vs. 2020 data for years to come, trying to see if a sudden increase in telemedicine led to better or worse clinical outcomes. I’ll be curious to see what they find and how it breaks down by disease and specialty.
How will work change? Right now my staff of three (including me) are all working separately from home, handling phone calls as if it were another office day. In today’s era that’s easy to set up, and we’re used to the drill from when I’m out of town.
Maybe in the future, on lighter days, I’ll do this more often, and have my staff work from home (on typically busy days I’ll still need them to check patients in and out, fax things, file charts, and do all the other things they do to keep the practice running). The marked decrease in air pollution is certainly noticeable and good for all. When the year is over I’d like to see how non-coronavirus respiratory issues changed between 2019 and 2020.
Other businesses will be looking at that, too, with an increase in telecommuting. Why pay for a large office space when a lot can be done over the Internet? It saves rent, gas, and driving time. How it will affect us, as a socially-dependent species, I have no idea.
It’s the same with grocery delivery. While most of us will likely continue to shop at stores, many will stay with the ease of delivery services after this. It may cost more, but it certainly saves time.
There will be social changes, although how long they’ll last is anyone’s guess. Grocery baggers, stockers, and delivery staff, often seen as lower-level occupations, are now considered part of critical infrastructure in keeping people supplied with food and other necessities, as well as preventing fights from breaking out in the toilet paper and hand-sanitizer aisles.
I’d like to think that, in a country divided, the need to work together will help bring people of different opinions together again, but from the way things look I don’t see that happening, which is sad because viruses don’t discriminate, so we shouldn’t either in fighting them.
Like with other challenges that we face, big and little, I can only hope that we’ll learn something from this and have a better world after it’s over. Only time will tell.
Dr. Block has a solo neurology practice in Scottsdale, Ariz. He has no relevant disclosures.
Eventually this strange Twilight Zone world of coronavirus will end and life will return to normal.
But obviously it won’t be the same, and like everyone else I wonder what will be different.
Telemedicine is one obvious change in my world, though I don’t know how much yet (granted, no one else does, either). I’m seeing a handful of people that way, limited to established patients, where we’re discussing chronic issues or reviewing recent test results.
If I have to see a new patient or an established one with an urgent issue, I’m still willing to meet them at my office (wearing masks and washing hands frequently). In neurology, a lot still depends on a decent exam. It’s pretty hard to check reflexes, sensory modalities, and muscle tone over the phone. If you think a malpractice attorney is going to give you a pass because you missed something by not examining a patient because of coronavirus ... think again.
I’m not sure how the whole telemedicine thing will play out after the dust settles, at least not at my little practice. I’m currently seeing patients by FaceTime and Skype, neither of which is considered HIPAA compliant. The requirement has been waived during the crisis to make sure people can still see doctors, but I don’t see it lasting beyond that. Privacy will always be a central concern in medicine.
When they declare the pandemic over and say I can’t use FaceTime or Skype anymore, that will likely end my use of such. While there are HIPAA-compliant telemedicine services out there, in a small practice I don’t have the time or money to invest in them.
I also wonder how outcomes will change. I suspect the research-minded will be analyzing 2019 vs. 2020 data for years to come, trying to see if a sudden increase in telemedicine led to better or worse clinical outcomes. I’ll be curious to see what they find and how it breaks down by disease and specialty.
How will work change? Right now my staff of three (including me) are all working separately from home, handling phone calls as if it were another office day. In today’s era that’s easy to set up, and we’re used to the drill from when I’m out of town.
Maybe in the future, on lighter days, I’ll do this more often, and have my staff work from home (on typically busy days I’ll still need them to check patients in and out, fax things, file charts, and do all the other things they do to keep the practice running). The marked decrease in air pollution is certainly noticeable and good for all. When the year is over I’d like to see how non-coronavirus respiratory issues changed between 2019 and 2020.
Other businesses will be looking at that, too, with an increase in telecommuting. Why pay for a large office space when a lot can be done over the Internet? It saves rent, gas, and driving time. How it will affect us, as a socially-dependent species, I have no idea.
It’s the same with grocery delivery. While most of us will likely continue to shop at stores, many will stay with the ease of delivery services after this. It may cost more, but it certainly saves time.
There will be social changes, although how long they’ll last is anyone’s guess. Grocery baggers, stockers, and delivery staff, often seen as lower-level occupations, are now considered part of critical infrastructure in keeping people supplied with food and other necessities, as well as preventing fights from breaking out in the toilet paper and hand-sanitizer aisles.
I’d like to think that, in a country divided, the need to work together will help bring people of different opinions together again, but from the way things look I don’t see that happening, which is sad because viruses don’t discriminate, so we shouldn’t either in fighting them.
Like with other challenges that we face, big and little, I can only hope that we’ll learn something from this and have a better world after it’s over. Only time will tell.
Dr. Block has a solo neurology practice in Scottsdale, Ariz. He has no relevant disclosures.
Eventually this strange Twilight Zone world of coronavirus will end and life will return to normal.
But obviously it won’t be the same, and like everyone else I wonder what will be different.
Telemedicine is one obvious change in my world, though I don’t know how much yet (granted, no one else does, either). I’m seeing a handful of people that way, limited to established patients, where we’re discussing chronic issues or reviewing recent test results.
If I have to see a new patient or an established one with an urgent issue, I’m still willing to meet them at my office (wearing masks and washing hands frequently). In neurology, a lot still depends on a decent exam. It’s pretty hard to check reflexes, sensory modalities, and muscle tone over the phone. If you think a malpractice attorney is going to give you a pass because you missed something by not examining a patient because of coronavirus ... think again.
I’m not sure how the whole telemedicine thing will play out after the dust settles, at least not at my little practice. I’m currently seeing patients by FaceTime and Skype, neither of which is considered HIPAA compliant. The requirement has been waived during the crisis to make sure people can still see doctors, but I don’t see it lasting beyond that. Privacy will always be a central concern in medicine.
When they declare the pandemic over and say I can’t use FaceTime or Skype anymore, that will likely end my use of such. While there are HIPAA-compliant telemedicine services out there, in a small practice I don’t have the time or money to invest in them.
I also wonder how outcomes will change. I suspect the research-minded will be analyzing 2019 vs. 2020 data for years to come, trying to see if a sudden increase in telemedicine led to better or worse clinical outcomes. I’ll be curious to see what they find and how it breaks down by disease and specialty.
How will work change? Right now my staff of three (including me) are all working separately from home, handling phone calls as if it were another office day. In today’s era that’s easy to set up, and we’re used to the drill from when I’m out of town.
Maybe in the future, on lighter days, I’ll do this more often, and have my staff work from home (on typically busy days I’ll still need them to check patients in and out, fax things, file charts, and do all the other things they do to keep the practice running). The marked decrease in air pollution is certainly noticeable and good for all. When the year is over I’d like to see how non-coronavirus respiratory issues changed between 2019 and 2020.
Other businesses will be looking at that, too, with an increase in telecommuting. Why pay for a large office space when a lot can be done over the Internet? It saves rent, gas, and driving time. How it will affect us, as a socially-dependent species, I have no idea.
It’s the same with grocery delivery. While most of us will likely continue to shop at stores, many will stay with the ease of delivery services after this. It may cost more, but it certainly saves time.
There will be social changes, although how long they’ll last is anyone’s guess. Grocery baggers, stockers, and delivery staff, often seen as lower-level occupations, are now considered part of critical infrastructure in keeping people supplied with food and other necessities, as well as preventing fights from breaking out in the toilet paper and hand-sanitizer aisles.
I’d like to think that, in a country divided, the need to work together will help bring people of different opinions together again, but from the way things look I don’t see that happening, which is sad because viruses don’t discriminate, so we shouldn’t either in fighting them.
Like with other challenges that we face, big and little, I can only hope that we’ll learn something from this and have a better world after it’s over. Only time will tell.
Dr. Block has a solo neurology practice in Scottsdale, Ariz. He has no relevant disclosures.
COVID-19 CRISIS: We must care for ourselves as we care for others
“I do not shrink from this responsibility, I welcome it.” —John F. Kennedy, inaugural address
COVID-19 has changed our world. Social distancing is now the norm and flattening the curve is our motto. Family physicians’ place on the front line of medicine is more important now than it has ever been.
In the Pennsylvania community in which we work, the first person to don protective gear and sample patients for viral testing in a rapidly organized COVID-19 testing site was John Russell, MD, a family physician. When I asked him about his experience, Dr. Russell said, “No one became a fireman to get cats out of trees ... it was to fight fires. As doctors, this is the same idea ... this is a chance to help fight the fires in our community.”
And, of course, it is primary care providers—family physicians, internists, pediatricians, nurse practitioners, physician assistants, and nurses—who day in and day out are putting aside their own fears, while dealing with those of their family, to come to work with a sense of purpose and courage.
The military uses the term “operational tempo” to describe the speed and intensity of actions relative to the speed and intensity of unfolding events in the operational environment. Family physicians are being asked to work at an increased speed in unfamiliar terrain as our environments change by the hour. The challenge is to answer the call—and take care of ourselves—in unprecedented ways. We often use anticipatory guidance with our patients to help prepare them for the challenges they will face. So, too, must we anticipate the things we will need to be attentive to in the coming months in order to sustain the effort that will be required of us.
With this in mind, we would be wise to consider developing plans in 3 domains: physical, mental, and social.
Physical. With gyms closed and restaurants limiting their offerings to take-out, this is an opportune time to create an exercise regimen at home and experiment with healthy meal options. YouTube videos abound for workouts of every length. And of course, you can simply take a daily walk, go for a run, or take a bike ride. Similarly, good choices can be made with take-out and the foods we prepare at home.
Continue to: Mentally...
Mentally we need the discipline to take breaks, delegate when necessary, and use downtime to clear our minds. Need another option? Consider meditation. Google “best meditation apps” and take your pick.
Social distancing doesn’t have to mean emotional isolation; technology allows us to connect with others through messaging and face-to-face video. We need to remember to regularly check in with those we care about; few things in life are as affirming as the connections with those who are close to us: family, co-workers, and patients.
Out of crisis comes opportunity. Should we be quarantined, we can remind ourselves that Sir Isaac Newton, while in quarantine during the bubonic plague, laid the foundation for classical physics, composed theories on light and optics, and penned his first draft of the law of gravity.1
Life carries on, amidst the pandemic. Even though the current focus is on the COVID-19 crisis, our many needs, joys, and challenges as human beings remain. Today, someone will find out she is pregnant; someone else will be diagnosed with cancer, or plan a wedding, or attend the funeral of a loved one. We, as family physicians, have the training to lead with courage and empathy. We have the expertise gained through years of helping patients though diverse physical and emotional challenges.
We will continue to listen to our patients’ stories, diagnose and treat their diseases, and take steps to bring a sense of calm to the chaos around us. We need to be mindful of our own mindset, because we have a choice. As the psychologist Victor Frankl said in 1946, after being liberated from the concentration camps, “Everything can be taken from a man but one thing: the last of the human freedoms—to choose one’s attitude in any given set of circumstances, to choose one’s own way.”2
1. Brockell G. During a pandemic, Isaac Newton had to work from home, too. He used the time wisely. The Washington Post. March 12, 2020. 2. Frankl VE. Man’s Search for Meaning. Boston, MA: Beacon Press; 2006.
“I do not shrink from this responsibility, I welcome it.” —John F. Kennedy, inaugural address
COVID-19 has changed our world. Social distancing is now the norm and flattening the curve is our motto. Family physicians’ place on the front line of medicine is more important now than it has ever been.
In the Pennsylvania community in which we work, the first person to don protective gear and sample patients for viral testing in a rapidly organized COVID-19 testing site was John Russell, MD, a family physician. When I asked him about his experience, Dr. Russell said, “No one became a fireman to get cats out of trees ... it was to fight fires. As doctors, this is the same idea ... this is a chance to help fight the fires in our community.”
And, of course, it is primary care providers—family physicians, internists, pediatricians, nurse practitioners, physician assistants, and nurses—who day in and day out are putting aside their own fears, while dealing with those of their family, to come to work with a sense of purpose and courage.
The military uses the term “operational tempo” to describe the speed and intensity of actions relative to the speed and intensity of unfolding events in the operational environment. Family physicians are being asked to work at an increased speed in unfamiliar terrain as our environments change by the hour. The challenge is to answer the call—and take care of ourselves—in unprecedented ways. We often use anticipatory guidance with our patients to help prepare them for the challenges they will face. So, too, must we anticipate the things we will need to be attentive to in the coming months in order to sustain the effort that will be required of us.
With this in mind, we would be wise to consider developing plans in 3 domains: physical, mental, and social.
Physical. With gyms closed and restaurants limiting their offerings to take-out, this is an opportune time to create an exercise regimen at home and experiment with healthy meal options. YouTube videos abound for workouts of every length. And of course, you can simply take a daily walk, go for a run, or take a bike ride. Similarly, good choices can be made with take-out and the foods we prepare at home.
Continue to: Mentally...
Mentally we need the discipline to take breaks, delegate when necessary, and use downtime to clear our minds. Need another option? Consider meditation. Google “best meditation apps” and take your pick.
Social distancing doesn’t have to mean emotional isolation; technology allows us to connect with others through messaging and face-to-face video. We need to remember to regularly check in with those we care about; few things in life are as affirming as the connections with those who are close to us: family, co-workers, and patients.
Out of crisis comes opportunity. Should we be quarantined, we can remind ourselves that Sir Isaac Newton, while in quarantine during the bubonic plague, laid the foundation for classical physics, composed theories on light and optics, and penned his first draft of the law of gravity.1
Life carries on, amidst the pandemic. Even though the current focus is on the COVID-19 crisis, our many needs, joys, and challenges as human beings remain. Today, someone will find out she is pregnant; someone else will be diagnosed with cancer, or plan a wedding, or attend the funeral of a loved one. We, as family physicians, have the training to lead with courage and empathy. We have the expertise gained through years of helping patients though diverse physical and emotional challenges.
We will continue to listen to our patients’ stories, diagnose and treat their diseases, and take steps to bring a sense of calm to the chaos around us. We need to be mindful of our own mindset, because we have a choice. As the psychologist Victor Frankl said in 1946, after being liberated from the concentration camps, “Everything can be taken from a man but one thing: the last of the human freedoms—to choose one’s attitude in any given set of circumstances, to choose one’s own way.”2
“I do not shrink from this responsibility, I welcome it.” —John F. Kennedy, inaugural address
COVID-19 has changed our world. Social distancing is now the norm and flattening the curve is our motto. Family physicians’ place on the front line of medicine is more important now than it has ever been.
In the Pennsylvania community in which we work, the first person to don protective gear and sample patients for viral testing in a rapidly organized COVID-19 testing site was John Russell, MD, a family physician. When I asked him about his experience, Dr. Russell said, “No one became a fireman to get cats out of trees ... it was to fight fires. As doctors, this is the same idea ... this is a chance to help fight the fires in our community.”
And, of course, it is primary care providers—family physicians, internists, pediatricians, nurse practitioners, physician assistants, and nurses—who day in and day out are putting aside their own fears, while dealing with those of their family, to come to work with a sense of purpose and courage.
The military uses the term “operational tempo” to describe the speed and intensity of actions relative to the speed and intensity of unfolding events in the operational environment. Family physicians are being asked to work at an increased speed in unfamiliar terrain as our environments change by the hour. The challenge is to answer the call—and take care of ourselves—in unprecedented ways. We often use anticipatory guidance with our patients to help prepare them for the challenges they will face. So, too, must we anticipate the things we will need to be attentive to in the coming months in order to sustain the effort that will be required of us.
With this in mind, we would be wise to consider developing plans in 3 domains: physical, mental, and social.
Physical. With gyms closed and restaurants limiting their offerings to take-out, this is an opportune time to create an exercise regimen at home and experiment with healthy meal options. YouTube videos abound for workouts of every length. And of course, you can simply take a daily walk, go for a run, or take a bike ride. Similarly, good choices can be made with take-out and the foods we prepare at home.
Continue to: Mentally...
Mentally we need the discipline to take breaks, delegate when necessary, and use downtime to clear our minds. Need another option? Consider meditation. Google “best meditation apps” and take your pick.
Social distancing doesn’t have to mean emotional isolation; technology allows us to connect with others through messaging and face-to-face video. We need to remember to regularly check in with those we care about; few things in life are as affirming as the connections with those who are close to us: family, co-workers, and patients.
Out of crisis comes opportunity. Should we be quarantined, we can remind ourselves that Sir Isaac Newton, while in quarantine during the bubonic plague, laid the foundation for classical physics, composed theories on light and optics, and penned his first draft of the law of gravity.1
Life carries on, amidst the pandemic. Even though the current focus is on the COVID-19 crisis, our many needs, joys, and challenges as human beings remain. Today, someone will find out she is pregnant; someone else will be diagnosed with cancer, or plan a wedding, or attend the funeral of a loved one. We, as family physicians, have the training to lead with courage and empathy. We have the expertise gained through years of helping patients though diverse physical and emotional challenges.
We will continue to listen to our patients’ stories, diagnose and treat their diseases, and take steps to bring a sense of calm to the chaos around us. We need to be mindful of our own mindset, because we have a choice. As the psychologist Victor Frankl said in 1946, after being liberated from the concentration camps, “Everything can be taken from a man but one thing: the last of the human freedoms—to choose one’s attitude in any given set of circumstances, to choose one’s own way.”2
1. Brockell G. During a pandemic, Isaac Newton had to work from home, too. He used the time wisely. The Washington Post. March 12, 2020. 2. Frankl VE. Man’s Search for Meaning. Boston, MA: Beacon Press; 2006.
1. Brockell G. During a pandemic, Isaac Newton had to work from home, too. He used the time wisely. The Washington Post. March 12, 2020. 2. Frankl VE. Man’s Search for Meaning. Boston, MA: Beacon Press; 2006.
Is protocol-driven COVID-19 respiratory therapy doing more harm than good?
Physicians in the COVID-19 trenches are beginning to question whether standard respiratory therapy protocols for acute respiratory distress syndrome (ARDS) are the best approach for treating patients with COVID-19 pneumonia.
At issue is the standard use of ventilators for a virus whose presentation has not followed the standard for ARDS, but is looking more like high-altitude pulmonary edema (HAPE) in some patients.
In a letter to the editor published in the American Journal of Respiratory and Critical Care Medicine on March 30, and in an editorial accepted for publication in Intensive Care Medicine, Luciano Gattinoni, MD, of the Medical University of Göttingen in Germany and colleagues make the case that protocol-driven ventilator use for patients with COVID-19 could be doing more harm than good.
Dr. Gattinoni noted that COVID-19 patients in ICUs in northern Italy had an atypical ARDS presentation with severe hypoxemia and well-preserved lung gas volume. He and colleagues suggested that instead of high positive end-expiratory pressure (PEEP), physicians should consider the lowest possible PEEP and gentle ventilation–practicing patience to “buy time with minimum additional damage.”
Similar observations were made by Cameron Kyle-Sidell, MD, a critical care physician working in New York City, who has been speaking out about this issue on Twitter and who shared his own experiences in this video interview with WebMD chief medical officer John Whyte, MD.
The bottom line, as Dr. Kyle-Sidell and Dr. Gattinoni agree, is that protocol-driven ventilator use may be causing lung injury in COVID-19 patients.
Consider disease phenotype
In the editorial, Dr. Gattinoni and colleagues explained further that ventilator settings should be based on physiological findings – with different respiratory treatment based on disease phenotype rather than using standard protocols.
‘“This, of course, is a conceptual model, but based on the observations we have this far, I don’t know of any model which is better,” he said in an interview.
Anecdotal evidence has increasingly demonstrated that this proposed physiological approach is associated with much lower mortality rates among COVID-19 patients, he said.
While not willing to name the hospitals at this time, he said that one center in Europe has had a 0% mortality rate among COVID-19 patients in the ICU when using this approach, compared with a 60% mortality rate at a nearby hospital using a protocol-driven approach.
In his editorial, Dr. Gattinoni disputed the recently published recommendation from the Surviving Sepsis Campaign that “mechanically ventilated patients with COVID-19 should be managed similarly to other patients with acute respiratory failure in the ICU.”
“Yet, COVID-19 pneumonia, despite falling in most of the circumstances under the Berlin definition of ARDS, is a specific disease, whose distinctive features are severe hypoxemia often associated with near normal respiratory system compliance,” Dr. Gattinoni and colleagues wrote, noting that this was true for more than half of the 150 patients he and his colleagues had assessed, and that several other colleagues in northern Italy reported similar findings. “This remarkable combination is almost never seen in severe ARDS.”
Dr. Gattinoni and colleagues hypothesized that COVID-19 patterns at patient presentation depend on interaction between three sets of factors: 1) disease severity, host response, physiological reserve and comorbidities; 2) ventilatory responsiveness of the patient to hypoxemia; and 3) time elapsed between disease onset and hospitalization.
They identified two primary phenotypes based on the interaction of these factors: Type L, characterized by low elastance, low ventilator perfusion ratio, low lung weight, and low recruitability; and Type H, characterized by high elastance, high right-to-left shunt, high lung weight, and high recruitability.
“Given this conceptual model, it follows that the respiratory treatment offered to Type L and Type H patients must be different,” Dr. Gattinoni said.
Patients may transition between phenotypes as their disease evolves. “If you start with the wrong protocol, at the end they become similar,” he said.
Rather, it is important to identify the phenotype at presentation to understand the pathophysiology and treat accordingly, he advised.
The phenotypes are best identified by CT scan, but signs implicit in each of the phenotypes, including respiratory system elastance and recruitability, can be used as surrogates if CT is unavailable, he noted.
“This is a kind of disease in which you don’t have to follow the protocol – you have to follow the physiology,” he said. “Unfortunately, many, many doctors around the world cannot think outside the protocol.”
In his interview with Dr. Whyte, Dr. Kyle-Sidell stressed that doctors must begin to consider other approaches. “We are desperate now, in the sense that everything we are doing does not seem to be working,” Dr. Kyle-Sidell said, noting that the first step toward improving outcomes is admitting that “this is something new.”
“I think it all starts from there, and I think we have the kind of scientific technology and the human capital in this country to solve this or at least have a very good shot at it,” he said.
Proposed treatment model
Dr. Gattinoni and his colleagues offered a proposed treatment model based on their conceptualization:
- Reverse hypoxemia through an increase in FiO2 to a level at which the Type L patient responds well, particularly for Type L patients who are not experiencing dyspnea.
- In Type L patients with dyspnea, try noninvasive options such as high-flow nasal cannula, continuous positive airway pressure, or noninvasive ventilation, and be sure to measure inspiratory esophageal pressure using esophageal manometry or surrogate measures. In intubated patients, determine P0.1 and P occlusion. High PEEP may decrease pleural pressure swings “and stop the vicious cycle that exacerbates lung injury,” but may be associated with high failure rates and delayed intubation.
- Intubate as soon as possible for esophageal pressure swings that increase from 5-10 cm H2O to above 15 cm H2O, which marks a transition from Type L to Type H phenotype and represents the level at which lung injury risk increases.
- For intubated and deeply sedated Type L patients who are hypercapnic, ventilate with volumes greater than 6 mL/kg up to 8-9 mL/kg as this high compliance results in tolerable strain without risk of ventilator-associated lung injury. Prone positioning should be used only as a rescue maneuver. Reduce PEEP to 8-10 cm H2O, given that the recruitability is low and the risk of hemodynamic failure increases at higher levels. Early intubation may avert the transition to Type H phenotype.
- Treat Type H phenotype like severe ARDS, including with higher PEEP if compatible with hemodynamics, and with prone positioning and extracorporeal support.
Dr. Gattinoni reported having no financial disclosures.
sworcester@mdedge.com
Physicians in the COVID-19 trenches are beginning to question whether standard respiratory therapy protocols for acute respiratory distress syndrome (ARDS) are the best approach for treating patients with COVID-19 pneumonia.
At issue is the standard use of ventilators for a virus whose presentation has not followed the standard for ARDS, but is looking more like high-altitude pulmonary edema (HAPE) in some patients.
In a letter to the editor published in the American Journal of Respiratory and Critical Care Medicine on March 30, and in an editorial accepted for publication in Intensive Care Medicine, Luciano Gattinoni, MD, of the Medical University of Göttingen in Germany and colleagues make the case that protocol-driven ventilator use for patients with COVID-19 could be doing more harm than good.
Dr. Gattinoni noted that COVID-19 patients in ICUs in northern Italy had an atypical ARDS presentation with severe hypoxemia and well-preserved lung gas volume. He and colleagues suggested that instead of high positive end-expiratory pressure (PEEP), physicians should consider the lowest possible PEEP and gentle ventilation–practicing patience to “buy time with minimum additional damage.”
Similar observations were made by Cameron Kyle-Sidell, MD, a critical care physician working in New York City, who has been speaking out about this issue on Twitter and who shared his own experiences in this video interview with WebMD chief medical officer John Whyte, MD.
The bottom line, as Dr. Kyle-Sidell and Dr. Gattinoni agree, is that protocol-driven ventilator use may be causing lung injury in COVID-19 patients.
Consider disease phenotype
In the editorial, Dr. Gattinoni and colleagues explained further that ventilator settings should be based on physiological findings – with different respiratory treatment based on disease phenotype rather than using standard protocols.
‘“This, of course, is a conceptual model, but based on the observations we have this far, I don’t know of any model which is better,” he said in an interview.
Anecdotal evidence has increasingly demonstrated that this proposed physiological approach is associated with much lower mortality rates among COVID-19 patients, he said.
While not willing to name the hospitals at this time, he said that one center in Europe has had a 0% mortality rate among COVID-19 patients in the ICU when using this approach, compared with a 60% mortality rate at a nearby hospital using a protocol-driven approach.
In his editorial, Dr. Gattinoni disputed the recently published recommendation from the Surviving Sepsis Campaign that “mechanically ventilated patients with COVID-19 should be managed similarly to other patients with acute respiratory failure in the ICU.”
“Yet, COVID-19 pneumonia, despite falling in most of the circumstances under the Berlin definition of ARDS, is a specific disease, whose distinctive features are severe hypoxemia often associated with near normal respiratory system compliance,” Dr. Gattinoni and colleagues wrote, noting that this was true for more than half of the 150 patients he and his colleagues had assessed, and that several other colleagues in northern Italy reported similar findings. “This remarkable combination is almost never seen in severe ARDS.”
Dr. Gattinoni and colleagues hypothesized that COVID-19 patterns at patient presentation depend on interaction between three sets of factors: 1) disease severity, host response, physiological reserve and comorbidities; 2) ventilatory responsiveness of the patient to hypoxemia; and 3) time elapsed between disease onset and hospitalization.
They identified two primary phenotypes based on the interaction of these factors: Type L, characterized by low elastance, low ventilator perfusion ratio, low lung weight, and low recruitability; and Type H, characterized by high elastance, high right-to-left shunt, high lung weight, and high recruitability.
“Given this conceptual model, it follows that the respiratory treatment offered to Type L and Type H patients must be different,” Dr. Gattinoni said.
Patients may transition between phenotypes as their disease evolves. “If you start with the wrong protocol, at the end they become similar,” he said.
Rather, it is important to identify the phenotype at presentation to understand the pathophysiology and treat accordingly, he advised.
The phenotypes are best identified by CT scan, but signs implicit in each of the phenotypes, including respiratory system elastance and recruitability, can be used as surrogates if CT is unavailable, he noted.
“This is a kind of disease in which you don’t have to follow the protocol – you have to follow the physiology,” he said. “Unfortunately, many, many doctors around the world cannot think outside the protocol.”
In his interview with Dr. Whyte, Dr. Kyle-Sidell stressed that doctors must begin to consider other approaches. “We are desperate now, in the sense that everything we are doing does not seem to be working,” Dr. Kyle-Sidell said, noting that the first step toward improving outcomes is admitting that “this is something new.”
“I think it all starts from there, and I think we have the kind of scientific technology and the human capital in this country to solve this or at least have a very good shot at it,” he said.
Proposed treatment model
Dr. Gattinoni and his colleagues offered a proposed treatment model based on their conceptualization:
- Reverse hypoxemia through an increase in FiO2 to a level at which the Type L patient responds well, particularly for Type L patients who are not experiencing dyspnea.
- In Type L patients with dyspnea, try noninvasive options such as high-flow nasal cannula, continuous positive airway pressure, or noninvasive ventilation, and be sure to measure inspiratory esophageal pressure using esophageal manometry or surrogate measures. In intubated patients, determine P0.1 and P occlusion. High PEEP may decrease pleural pressure swings “and stop the vicious cycle that exacerbates lung injury,” but may be associated with high failure rates and delayed intubation.
- Intubate as soon as possible for esophageal pressure swings that increase from 5-10 cm H2O to above 15 cm H2O, which marks a transition from Type L to Type H phenotype and represents the level at which lung injury risk increases.
- For intubated and deeply sedated Type L patients who are hypercapnic, ventilate with volumes greater than 6 mL/kg up to 8-9 mL/kg as this high compliance results in tolerable strain without risk of ventilator-associated lung injury. Prone positioning should be used only as a rescue maneuver. Reduce PEEP to 8-10 cm H2O, given that the recruitability is low and the risk of hemodynamic failure increases at higher levels. Early intubation may avert the transition to Type H phenotype.
- Treat Type H phenotype like severe ARDS, including with higher PEEP if compatible with hemodynamics, and with prone positioning and extracorporeal support.
Dr. Gattinoni reported having no financial disclosures.
sworcester@mdedge.com
Physicians in the COVID-19 trenches are beginning to question whether standard respiratory therapy protocols for acute respiratory distress syndrome (ARDS) are the best approach for treating patients with COVID-19 pneumonia.
At issue is the standard use of ventilators for a virus whose presentation has not followed the standard for ARDS, but is looking more like high-altitude pulmonary edema (HAPE) in some patients.
In a letter to the editor published in the American Journal of Respiratory and Critical Care Medicine on March 30, and in an editorial accepted for publication in Intensive Care Medicine, Luciano Gattinoni, MD, of the Medical University of Göttingen in Germany and colleagues make the case that protocol-driven ventilator use for patients with COVID-19 could be doing more harm than good.
Dr. Gattinoni noted that COVID-19 patients in ICUs in northern Italy had an atypical ARDS presentation with severe hypoxemia and well-preserved lung gas volume. He and colleagues suggested that instead of high positive end-expiratory pressure (PEEP), physicians should consider the lowest possible PEEP and gentle ventilation–practicing patience to “buy time with minimum additional damage.”
Similar observations were made by Cameron Kyle-Sidell, MD, a critical care physician working in New York City, who has been speaking out about this issue on Twitter and who shared his own experiences in this video interview with WebMD chief medical officer John Whyte, MD.
The bottom line, as Dr. Kyle-Sidell and Dr. Gattinoni agree, is that protocol-driven ventilator use may be causing lung injury in COVID-19 patients.
Consider disease phenotype
In the editorial, Dr. Gattinoni and colleagues explained further that ventilator settings should be based on physiological findings – with different respiratory treatment based on disease phenotype rather than using standard protocols.
‘“This, of course, is a conceptual model, but based on the observations we have this far, I don’t know of any model which is better,” he said in an interview.
Anecdotal evidence has increasingly demonstrated that this proposed physiological approach is associated with much lower mortality rates among COVID-19 patients, he said.
While not willing to name the hospitals at this time, he said that one center in Europe has had a 0% mortality rate among COVID-19 patients in the ICU when using this approach, compared with a 60% mortality rate at a nearby hospital using a protocol-driven approach.
In his editorial, Dr. Gattinoni disputed the recently published recommendation from the Surviving Sepsis Campaign that “mechanically ventilated patients with COVID-19 should be managed similarly to other patients with acute respiratory failure in the ICU.”
“Yet, COVID-19 pneumonia, despite falling in most of the circumstances under the Berlin definition of ARDS, is a specific disease, whose distinctive features are severe hypoxemia often associated with near normal respiratory system compliance,” Dr. Gattinoni and colleagues wrote, noting that this was true for more than half of the 150 patients he and his colleagues had assessed, and that several other colleagues in northern Italy reported similar findings. “This remarkable combination is almost never seen in severe ARDS.”
Dr. Gattinoni and colleagues hypothesized that COVID-19 patterns at patient presentation depend on interaction between three sets of factors: 1) disease severity, host response, physiological reserve and comorbidities; 2) ventilatory responsiveness of the patient to hypoxemia; and 3) time elapsed between disease onset and hospitalization.
They identified two primary phenotypes based on the interaction of these factors: Type L, characterized by low elastance, low ventilator perfusion ratio, low lung weight, and low recruitability; and Type H, characterized by high elastance, high right-to-left shunt, high lung weight, and high recruitability.
“Given this conceptual model, it follows that the respiratory treatment offered to Type L and Type H patients must be different,” Dr. Gattinoni said.
Patients may transition between phenotypes as their disease evolves. “If you start with the wrong protocol, at the end they become similar,” he said.
Rather, it is important to identify the phenotype at presentation to understand the pathophysiology and treat accordingly, he advised.
The phenotypes are best identified by CT scan, but signs implicit in each of the phenotypes, including respiratory system elastance and recruitability, can be used as surrogates if CT is unavailable, he noted.
“This is a kind of disease in which you don’t have to follow the protocol – you have to follow the physiology,” he said. “Unfortunately, many, many doctors around the world cannot think outside the protocol.”
In his interview with Dr. Whyte, Dr. Kyle-Sidell stressed that doctors must begin to consider other approaches. “We are desperate now, in the sense that everything we are doing does not seem to be working,” Dr. Kyle-Sidell said, noting that the first step toward improving outcomes is admitting that “this is something new.”
“I think it all starts from there, and I think we have the kind of scientific technology and the human capital in this country to solve this or at least have a very good shot at it,” he said.
Proposed treatment model
Dr. Gattinoni and his colleagues offered a proposed treatment model based on their conceptualization:
- Reverse hypoxemia through an increase in FiO2 to a level at which the Type L patient responds well, particularly for Type L patients who are not experiencing dyspnea.
- In Type L patients with dyspnea, try noninvasive options such as high-flow nasal cannula, continuous positive airway pressure, or noninvasive ventilation, and be sure to measure inspiratory esophageal pressure using esophageal manometry or surrogate measures. In intubated patients, determine P0.1 and P occlusion. High PEEP may decrease pleural pressure swings “and stop the vicious cycle that exacerbates lung injury,” but may be associated with high failure rates and delayed intubation.
- Intubate as soon as possible for esophageal pressure swings that increase from 5-10 cm H2O to above 15 cm H2O, which marks a transition from Type L to Type H phenotype and represents the level at which lung injury risk increases.
- For intubated and deeply sedated Type L patients who are hypercapnic, ventilate with volumes greater than 6 mL/kg up to 8-9 mL/kg as this high compliance results in tolerable strain without risk of ventilator-associated lung injury. Prone positioning should be used only as a rescue maneuver. Reduce PEEP to 8-10 cm H2O, given that the recruitability is low and the risk of hemodynamic failure increases at higher levels. Early intubation may avert the transition to Type H phenotype.
- Treat Type H phenotype like severe ARDS, including with higher PEEP if compatible with hemodynamics, and with prone positioning and extracorporeal support.
Dr. Gattinoni reported having no financial disclosures.
sworcester@mdedge.com
A decade of telemedicine policy has advanced in just 2 weeks
The rapid spread of , which he’d never used.
But as soon as he learned that telehealth regulations had been relaxed by the Centers for Medicare & Medicaid Services and that reimbursement had been broadened, Dr. Desai, a dermatologist in private practice and his staff began to mobilize.
“Kaboom! We made the decision to start doing it,” he said in an interview. “We drafted a consent form, uploaded it to our website, called patients, changed our voice greeting, and got clarity on insurance coverage. We’ve been flying by the seat of our pants.”
“I’m doing it because I don’t have a choice at this point,” said Dr. Desai, who is a member of the American Academy of Dermatology board of directors and its coronavirus task force. “I’m very worried about continuing to be able to meet our payroll expenses for staff and overhead to keep the office open.”
“Flying by the seat of our pants” to see patients virtually
Dermatologists have long been considered pioneers in telemedicine. They have, since the 1990s, capitalized on the visual nature of the specialty to diagnose and treat skin diseases by incorporating photos, videos, and virtual-patient visits. But the pandemic has forced the hands of even holdouts like Dr. Desai, who clung to in-person consults because of confusion related to HIPAA compliance issues and the sense that teledermatology “really dehumanizes patient interaction” for him.
In fact, as of 2017, only 15% of the nation’s 11,000 or so dermatologists had implemented telehealth into their practices, according to an AAD practice survey. In the wake of COVID-19, however, that percentage has likely more than tripled, experts estimate.
Now, dermatologists are assuming the mantle of educators for other specialists who never considered telehealth before in-person visits became fraught with concerns about the spread of the virus. And some are publishing guidelines for colleagues on how to prioritize teledermatology to stem transmission and conserve personal protective equipment (PPE) and hospital beds.
User-friendly technology and the relaxed telehealth restrictions have made it fairly simple for patients and physicians to connect. Facetime and other once-prohibited platforms are all currently permissible, although physicians are encouraged to notify patients about potential privacy risks, according to an AAD teledermatology tool kit.
Teledermatology innovators
“We’ve moved 10 years in telemedicine policy in 2 weeks,” said Karen Edison, MD, of the University of Missouri, Columbia. “The federal government has really loosened the reins.”
At least half of all dermatologists in the United States have adopted telehealth since the pandemic emerged, she estimated. And most, like Dr. Desai, have done so in just the last several weeks.
“You can do about 90% of what you need to do as a dermatologist using the technology,” said Dr. Edison, who launched the first dermatology Extension for Community Healthcare Outcomes, or ECHO, program in the Midwest. That telehealth model was originally developed to connect rural general practitioners with specialists at academic medical centers or large health systems.
“People are used to taking pictures with their phones. In some ways, this crisis may change the face of our specialty,” she said in an interview.
“As we’re all practicing social distancing, I think physicians and patients are rethinking how we can access healthcare without pursuing traditional face-to-face interactions,” said Ivy Lee, MD, from the University of California, San Francisco, who is past chair of the AAD telemedicine task force and current chair of the teledermatology committee at the American Telemedicine Association. “Virtual health and telemedicine fit perfectly with that.”
Even before the pandemic, the innovative ways dermatologists were using telehealth were garnering increasing acclaim. All four clinical groups short-listed for dermatology team of the year at the BMJ Awards 2020 employed telehealth to improve patient services in the United Kingdom.
In the United States, dermatologists are joining forces to boost understanding of how telehealth can protect patients and clinicians from some of the ravages of the virus.
The Society of Dermatology Hospitalists has developed an algorithm – built on experiences its members have had caring for hospitalized patients with acute dermatologic conditions – to provide a “logical way” to triage telemedicine consults in multiple hospital settings during the coronavirus crisis, said President-Elect Daniela Kroshinsky, MD, from Massachusetts General Hospital in Boston.
Telemedicine consultation is prioritized and patients at high risk for COVID-19 exposure are identified so that exposure time and resource use are limited and patient and staff safety are maximized.
“We want to empower our colleagues in community hospitals to play a role in safely providing care for patients in need but to be mindful about preserving resources,” said Dr. Kroshinsky, who reported that the algorithm will be published imminently.
“If you don’t have to see a patient in person and can offer recommendations through telederm, you don’t need to put on a gown, gloves, mask, or goggles,” she said in an interview. “If you’re unable to assess photos, then of course you’ll use the appropriate protective wear, but it will be better if you can obtain the same result” without having to do so.
Sharing expertise
After the first week of tracking data to gauge the effectiveness of the algorithm at Massachusetts General, Dr. Kroshinsky said she is buoyed.
Of the 35 patients assessed electronically – all of whom would previously have been seen in person – only 4 ended up needing a subsequent in-person consult, she reported.
“It’s worked out great,” said Dr. Kroshinsky, who noted that the pandemic is a “nice opportunity” to test different telehealth platforms and improve quality down the line. “We never had to use any excessive PPE, beyond what was routine, and the majority of patients were able to be staffed remotely. All patients had successful outcomes.”
With telehealth more firmly established in dermatology than in most other specialties, dermatologists are now helping clinicians in other fields who are rapidly ramping up their own telemedicine offerings.
These might include obstetrics and gynecology or “any medical specialty where they need to do checkups with their patients and don’t want them coming in for nonemergent visits,” said Carrie L. Kovarik, MD, of the University of Pennsylvania, Philadelphia.
In addition to fielding many recent calls and emails from physicians seeking guidance on telehealth, Dr. Kovarik, Dr. Lee, and colleagues have published the steps required to integrate the technology into outpatient practices.
“Now that there’s a time for broad implementation, our colleagues are looking to us for help and troubleshooting advice,” said Dr. Kovarik, who is also a member of the AAD COVID-19 response task force.
Various specialties “lend themselves to telehealth, depending on how image- or data-dependent they are,” Dr. Lee said in an interview. “But all specialists thinking of limiting or shutting down their practices are thinking about how they can provide continuity of care without exposing patients or staff to the risk of contracting the coronavirus.”
After-COVID goals
In his first week of virtual patient consults, Dr. Desai said he saw about 50 patients, which is still far fewer than the 160-180 he sees in person during a normal week.
“The problem is that patients don’t really want to do telehealth. You’d think it would be a good option,” he said, “but patients hesitate because they don’t really know how to use their device.” Some have instead rescheduled in-person appointments for months down the line.
Although telehealth has enabled Dr. Desai to readily assess patients with acne, hair loss, psoriasis, rashes, warts, and eczema, he’s concerned that necessary procedures, such as biopsies and dermoscopies, could be dangerously delayed. It’s also hard to assess the texture and thickness of certain skin lesions in photos or videos, he said.
“I’m trying to stay optimistic that this will get better and we’re able to move back to taking care of patients the way we need to,” he said.
Like Dr. Desai, other dermatologists who’ve implemented telemedicine during the pandemic have largely been swayed by the relaxed CMS regulations. “It’s made all the difference,” Dr. Kovarik said. “It has brought down the anxiety level and decreased questions about platforms and concentrated them on how to code the visits.”
And although it’s difficult to envision post-COVID medical practice in the thick of the pandemic, dermatologists expect the current strides in telemedicine will stick.
“I’m hoping that telehealth use isn’t dialed back all the way to baseline” after the pandemic eases, Dr. Kovarik said. “The cat’s out of the bag, and now that it is, hopefully it won’t be put back in.”
“If there’s a silver lining to this,” Dr. Kroshinsky said, “I hope it’s that we’ll be able to innovate around health care in a fashion we wouldn’t have seen otherwise.”
A version of this article originally appeared on Medscape.com.
The rapid spread of , which he’d never used.
But as soon as he learned that telehealth regulations had been relaxed by the Centers for Medicare & Medicaid Services and that reimbursement had been broadened, Dr. Desai, a dermatologist in private practice and his staff began to mobilize.
“Kaboom! We made the decision to start doing it,” he said in an interview. “We drafted a consent form, uploaded it to our website, called patients, changed our voice greeting, and got clarity on insurance coverage. We’ve been flying by the seat of our pants.”
“I’m doing it because I don’t have a choice at this point,” said Dr. Desai, who is a member of the American Academy of Dermatology board of directors and its coronavirus task force. “I’m very worried about continuing to be able to meet our payroll expenses for staff and overhead to keep the office open.”
“Flying by the seat of our pants” to see patients virtually
Dermatologists have long been considered pioneers in telemedicine. They have, since the 1990s, capitalized on the visual nature of the specialty to diagnose and treat skin diseases by incorporating photos, videos, and virtual-patient visits. But the pandemic has forced the hands of even holdouts like Dr. Desai, who clung to in-person consults because of confusion related to HIPAA compliance issues and the sense that teledermatology “really dehumanizes patient interaction” for him.
In fact, as of 2017, only 15% of the nation’s 11,000 or so dermatologists had implemented telehealth into their practices, according to an AAD practice survey. In the wake of COVID-19, however, that percentage has likely more than tripled, experts estimate.
Now, dermatologists are assuming the mantle of educators for other specialists who never considered telehealth before in-person visits became fraught with concerns about the spread of the virus. And some are publishing guidelines for colleagues on how to prioritize teledermatology to stem transmission and conserve personal protective equipment (PPE) and hospital beds.
User-friendly technology and the relaxed telehealth restrictions have made it fairly simple for patients and physicians to connect. Facetime and other once-prohibited platforms are all currently permissible, although physicians are encouraged to notify patients about potential privacy risks, according to an AAD teledermatology tool kit.
Teledermatology innovators
“We’ve moved 10 years in telemedicine policy in 2 weeks,” said Karen Edison, MD, of the University of Missouri, Columbia. “The federal government has really loosened the reins.”
At least half of all dermatologists in the United States have adopted telehealth since the pandemic emerged, she estimated. And most, like Dr. Desai, have done so in just the last several weeks.
“You can do about 90% of what you need to do as a dermatologist using the technology,” said Dr. Edison, who launched the first dermatology Extension for Community Healthcare Outcomes, or ECHO, program in the Midwest. That telehealth model was originally developed to connect rural general practitioners with specialists at academic medical centers or large health systems.
“People are used to taking pictures with their phones. In some ways, this crisis may change the face of our specialty,” she said in an interview.
“As we’re all practicing social distancing, I think physicians and patients are rethinking how we can access healthcare without pursuing traditional face-to-face interactions,” said Ivy Lee, MD, from the University of California, San Francisco, who is past chair of the AAD telemedicine task force and current chair of the teledermatology committee at the American Telemedicine Association. “Virtual health and telemedicine fit perfectly with that.”
Even before the pandemic, the innovative ways dermatologists were using telehealth were garnering increasing acclaim. All four clinical groups short-listed for dermatology team of the year at the BMJ Awards 2020 employed telehealth to improve patient services in the United Kingdom.
In the United States, dermatologists are joining forces to boost understanding of how telehealth can protect patients and clinicians from some of the ravages of the virus.
The Society of Dermatology Hospitalists has developed an algorithm – built on experiences its members have had caring for hospitalized patients with acute dermatologic conditions – to provide a “logical way” to triage telemedicine consults in multiple hospital settings during the coronavirus crisis, said President-Elect Daniela Kroshinsky, MD, from Massachusetts General Hospital in Boston.
Telemedicine consultation is prioritized and patients at high risk for COVID-19 exposure are identified so that exposure time and resource use are limited and patient and staff safety are maximized.
“We want to empower our colleagues in community hospitals to play a role in safely providing care for patients in need but to be mindful about preserving resources,” said Dr. Kroshinsky, who reported that the algorithm will be published imminently.
“If you don’t have to see a patient in person and can offer recommendations through telederm, you don’t need to put on a gown, gloves, mask, or goggles,” she said in an interview. “If you’re unable to assess photos, then of course you’ll use the appropriate protective wear, but it will be better if you can obtain the same result” without having to do so.
Sharing expertise
After the first week of tracking data to gauge the effectiveness of the algorithm at Massachusetts General, Dr. Kroshinsky said she is buoyed.
Of the 35 patients assessed electronically – all of whom would previously have been seen in person – only 4 ended up needing a subsequent in-person consult, she reported.
“It’s worked out great,” said Dr. Kroshinsky, who noted that the pandemic is a “nice opportunity” to test different telehealth platforms and improve quality down the line. “We never had to use any excessive PPE, beyond what was routine, and the majority of patients were able to be staffed remotely. All patients had successful outcomes.”
With telehealth more firmly established in dermatology than in most other specialties, dermatologists are now helping clinicians in other fields who are rapidly ramping up their own telemedicine offerings.
These might include obstetrics and gynecology or “any medical specialty where they need to do checkups with their patients and don’t want them coming in for nonemergent visits,” said Carrie L. Kovarik, MD, of the University of Pennsylvania, Philadelphia.
In addition to fielding many recent calls and emails from physicians seeking guidance on telehealth, Dr. Kovarik, Dr. Lee, and colleagues have published the steps required to integrate the technology into outpatient practices.
“Now that there’s a time for broad implementation, our colleagues are looking to us for help and troubleshooting advice,” said Dr. Kovarik, who is also a member of the AAD COVID-19 response task force.
Various specialties “lend themselves to telehealth, depending on how image- or data-dependent they are,” Dr. Lee said in an interview. “But all specialists thinking of limiting or shutting down their practices are thinking about how they can provide continuity of care without exposing patients or staff to the risk of contracting the coronavirus.”
After-COVID goals
In his first week of virtual patient consults, Dr. Desai said he saw about 50 patients, which is still far fewer than the 160-180 he sees in person during a normal week.
“The problem is that patients don’t really want to do telehealth. You’d think it would be a good option,” he said, “but patients hesitate because they don’t really know how to use their device.” Some have instead rescheduled in-person appointments for months down the line.
Although telehealth has enabled Dr. Desai to readily assess patients with acne, hair loss, psoriasis, rashes, warts, and eczema, he’s concerned that necessary procedures, such as biopsies and dermoscopies, could be dangerously delayed. It’s also hard to assess the texture and thickness of certain skin lesions in photos or videos, he said.
“I’m trying to stay optimistic that this will get better and we’re able to move back to taking care of patients the way we need to,” he said.
Like Dr. Desai, other dermatologists who’ve implemented telemedicine during the pandemic have largely been swayed by the relaxed CMS regulations. “It’s made all the difference,” Dr. Kovarik said. “It has brought down the anxiety level and decreased questions about platforms and concentrated them on how to code the visits.”
And although it’s difficult to envision post-COVID medical practice in the thick of the pandemic, dermatologists expect the current strides in telemedicine will stick.
“I’m hoping that telehealth use isn’t dialed back all the way to baseline” after the pandemic eases, Dr. Kovarik said. “The cat’s out of the bag, and now that it is, hopefully it won’t be put back in.”
“If there’s a silver lining to this,” Dr. Kroshinsky said, “I hope it’s that we’ll be able to innovate around health care in a fashion we wouldn’t have seen otherwise.”
A version of this article originally appeared on Medscape.com.
The rapid spread of , which he’d never used.
But as soon as he learned that telehealth regulations had been relaxed by the Centers for Medicare & Medicaid Services and that reimbursement had been broadened, Dr. Desai, a dermatologist in private practice and his staff began to mobilize.
“Kaboom! We made the decision to start doing it,” he said in an interview. “We drafted a consent form, uploaded it to our website, called patients, changed our voice greeting, and got clarity on insurance coverage. We’ve been flying by the seat of our pants.”
“I’m doing it because I don’t have a choice at this point,” said Dr. Desai, who is a member of the American Academy of Dermatology board of directors and its coronavirus task force. “I’m very worried about continuing to be able to meet our payroll expenses for staff and overhead to keep the office open.”
“Flying by the seat of our pants” to see patients virtually
Dermatologists have long been considered pioneers in telemedicine. They have, since the 1990s, capitalized on the visual nature of the specialty to diagnose and treat skin diseases by incorporating photos, videos, and virtual-patient visits. But the pandemic has forced the hands of even holdouts like Dr. Desai, who clung to in-person consults because of confusion related to HIPAA compliance issues and the sense that teledermatology “really dehumanizes patient interaction” for him.
In fact, as of 2017, only 15% of the nation’s 11,000 or so dermatologists had implemented telehealth into their practices, according to an AAD practice survey. In the wake of COVID-19, however, that percentage has likely more than tripled, experts estimate.
Now, dermatologists are assuming the mantle of educators for other specialists who never considered telehealth before in-person visits became fraught with concerns about the spread of the virus. And some are publishing guidelines for colleagues on how to prioritize teledermatology to stem transmission and conserve personal protective equipment (PPE) and hospital beds.
User-friendly technology and the relaxed telehealth restrictions have made it fairly simple for patients and physicians to connect. Facetime and other once-prohibited platforms are all currently permissible, although physicians are encouraged to notify patients about potential privacy risks, according to an AAD teledermatology tool kit.
Teledermatology innovators
“We’ve moved 10 years in telemedicine policy in 2 weeks,” said Karen Edison, MD, of the University of Missouri, Columbia. “The federal government has really loosened the reins.”
At least half of all dermatologists in the United States have adopted telehealth since the pandemic emerged, she estimated. And most, like Dr. Desai, have done so in just the last several weeks.
“You can do about 90% of what you need to do as a dermatologist using the technology,” said Dr. Edison, who launched the first dermatology Extension for Community Healthcare Outcomes, or ECHO, program in the Midwest. That telehealth model was originally developed to connect rural general practitioners with specialists at academic medical centers or large health systems.
“People are used to taking pictures with their phones. In some ways, this crisis may change the face of our specialty,” she said in an interview.
“As we’re all practicing social distancing, I think physicians and patients are rethinking how we can access healthcare without pursuing traditional face-to-face interactions,” said Ivy Lee, MD, from the University of California, San Francisco, who is past chair of the AAD telemedicine task force and current chair of the teledermatology committee at the American Telemedicine Association. “Virtual health and telemedicine fit perfectly with that.”
Even before the pandemic, the innovative ways dermatologists were using telehealth were garnering increasing acclaim. All four clinical groups short-listed for dermatology team of the year at the BMJ Awards 2020 employed telehealth to improve patient services in the United Kingdom.
In the United States, dermatologists are joining forces to boost understanding of how telehealth can protect patients and clinicians from some of the ravages of the virus.
The Society of Dermatology Hospitalists has developed an algorithm – built on experiences its members have had caring for hospitalized patients with acute dermatologic conditions – to provide a “logical way” to triage telemedicine consults in multiple hospital settings during the coronavirus crisis, said President-Elect Daniela Kroshinsky, MD, from Massachusetts General Hospital in Boston.
Telemedicine consultation is prioritized and patients at high risk for COVID-19 exposure are identified so that exposure time and resource use are limited and patient and staff safety are maximized.
“We want to empower our colleagues in community hospitals to play a role in safely providing care for patients in need but to be mindful about preserving resources,” said Dr. Kroshinsky, who reported that the algorithm will be published imminently.
“If you don’t have to see a patient in person and can offer recommendations through telederm, you don’t need to put on a gown, gloves, mask, or goggles,” she said in an interview. “If you’re unable to assess photos, then of course you’ll use the appropriate protective wear, but it will be better if you can obtain the same result” without having to do so.
Sharing expertise
After the first week of tracking data to gauge the effectiveness of the algorithm at Massachusetts General, Dr. Kroshinsky said she is buoyed.
Of the 35 patients assessed electronically – all of whom would previously have been seen in person – only 4 ended up needing a subsequent in-person consult, she reported.
“It’s worked out great,” said Dr. Kroshinsky, who noted that the pandemic is a “nice opportunity” to test different telehealth platforms and improve quality down the line. “We never had to use any excessive PPE, beyond what was routine, and the majority of patients were able to be staffed remotely. All patients had successful outcomes.”
With telehealth more firmly established in dermatology than in most other specialties, dermatologists are now helping clinicians in other fields who are rapidly ramping up their own telemedicine offerings.
These might include obstetrics and gynecology or “any medical specialty where they need to do checkups with their patients and don’t want them coming in for nonemergent visits,” said Carrie L. Kovarik, MD, of the University of Pennsylvania, Philadelphia.
In addition to fielding many recent calls and emails from physicians seeking guidance on telehealth, Dr. Kovarik, Dr. Lee, and colleagues have published the steps required to integrate the technology into outpatient practices.
“Now that there’s a time for broad implementation, our colleagues are looking to us for help and troubleshooting advice,” said Dr. Kovarik, who is also a member of the AAD COVID-19 response task force.
Various specialties “lend themselves to telehealth, depending on how image- or data-dependent they are,” Dr. Lee said in an interview. “But all specialists thinking of limiting or shutting down their practices are thinking about how they can provide continuity of care without exposing patients or staff to the risk of contracting the coronavirus.”
After-COVID goals
In his first week of virtual patient consults, Dr. Desai said he saw about 50 patients, which is still far fewer than the 160-180 he sees in person during a normal week.
“The problem is that patients don’t really want to do telehealth. You’d think it would be a good option,” he said, “but patients hesitate because they don’t really know how to use their device.” Some have instead rescheduled in-person appointments for months down the line.
Although telehealth has enabled Dr. Desai to readily assess patients with acne, hair loss, psoriasis, rashes, warts, and eczema, he’s concerned that necessary procedures, such as biopsies and dermoscopies, could be dangerously delayed. It’s also hard to assess the texture and thickness of certain skin lesions in photos or videos, he said.
“I’m trying to stay optimistic that this will get better and we’re able to move back to taking care of patients the way we need to,” he said.
Like Dr. Desai, other dermatologists who’ve implemented telemedicine during the pandemic have largely been swayed by the relaxed CMS regulations. “It’s made all the difference,” Dr. Kovarik said. “It has brought down the anxiety level and decreased questions about platforms and concentrated them on how to code the visits.”
And although it’s difficult to envision post-COVID medical practice in the thick of the pandemic, dermatologists expect the current strides in telemedicine will stick.
“I’m hoping that telehealth use isn’t dialed back all the way to baseline” after the pandemic eases, Dr. Kovarik said. “The cat’s out of the bag, and now that it is, hopefully it won’t be put back in.”
“If there’s a silver lining to this,” Dr. Kroshinsky said, “I hope it’s that we’ll be able to innovate around health care in a fashion we wouldn’t have seen otherwise.”
A version of this article originally appeared on Medscape.com.