User login
Inside Mercy’s mission to care for non-COVID patients in Los Angeles
When the hospital ship USNS Mercy departed San Diego’s Naval Station North Island on March 23, 2020, to support the Department of Defense efforts in Los Angeles during the coronavirus outbreak, Commander Erin Blevins remembers the crew’s excitement was palpable.
“We normally do partnerships abroad and respond to tsunamis and earthquakes,” said Cdr. Blevins, MD, a pediatric hematologist-oncologist who served as director of medical services for the mission. “This was a slight change in situation, but still disaster relief in the form of a pandemic. We switched our mindset to putting together the best experts for an infectious disease pandemic versus an earthquake disaster relief.”
A new mission
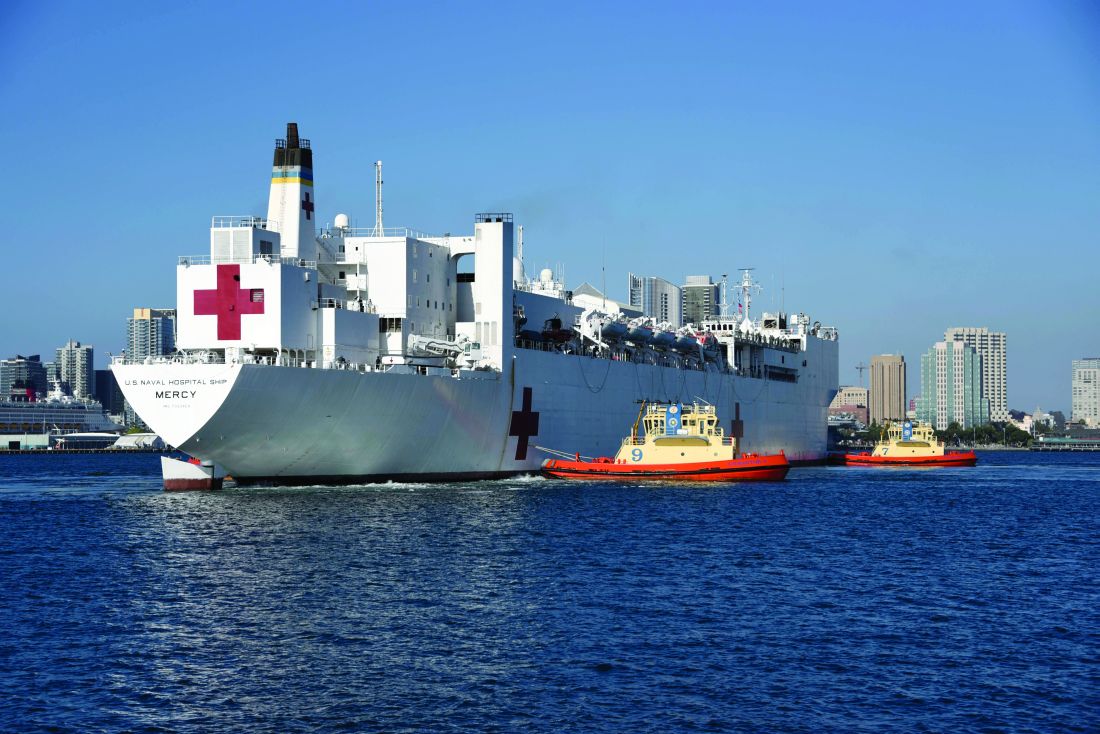
The 1,000-bed Mercy ship – a converted San Clemente–class oil tanker that was delivered in 1986 – spent nearly 50 days pier side in Los Angeles as a referral hospital for non–COVID-19 patients, so that clinicians at Los Angeles area hospitals could care for an anticipated surge of COVID-19 patients. “We went into it with expectations of, ‘We’ll treat as many patients as you need us to take,” Cdr. Blevins recalled. “I don’t even think Los Angeles [health officials] knew exactly where they were going to peak and what the need was going to be.”
Between March 29 and May 15, about 1,071 medical personnel aboard the Mercy cared for 77 patients with an average age of 53 years who were referred from 11 Los Angeles area hospitals. The physicians, nurses, and other medical support personnel were drawn from military treatment facilities across the country. “We had additional people join us as we scoped the mission to be more medically heavy and surgically light,” said Captain John Rotruck, MD, an anesthesiologist who is commanding officer of Mercy’s medical treatment facility. “We did adjust to make sure that we had the right staffing mix to meet the parameters that we were assigned. That was the crux of the change: a change in flavors of staffing to ensure that we focused on ICU and ward medical care as opposed to very heavy surgical care in support of a combat operation.”
About 10% of the team consisted of reservists who volunteered for the mission. “There’s no way you could have walked around the ship and known who was active duty and who was reservist,” said Capt. Rotruck, who was formerly chief of staff at Walter Reed National Military Medical Center, Bethesda, Md. “They worked together so well, and I think that marriage of active duty who are used to working in a military medical treatment facility – in our case, a Navy medical treatment facility – together with our reservist physician colleagues who work in civilian facilities around the country, was beneficial. It was a synergistic relationship. I think both sides walked away learning quite a bit from each other.”
Start with screening
All crew members underwent a temperature check and completed a health screening questionnaire: once before departing their home of record and again before boarding Mercy. Based on those results, crew members and medical staff were screened for COVID-19 and tested as needed in order to minimize the risk of an outbreak aboard the ship.
Fewer than 1% of crew members developed COVID-19 or tested positive for the virus during the mission, according to Capt. Rotruck. Affected individuals were isolated and quarantined. “All staff have recovered and are doing well,” he said.
Mercy personnel worked with local health officials to ensure that all patients transferred to the ship tested negative for COVID-19. Physicians aboard the Mercy then worked directly with the patients’ civilian physician to ensure a safe and thorough turnover process before the patients were transferred.
From basic medical to trauma care
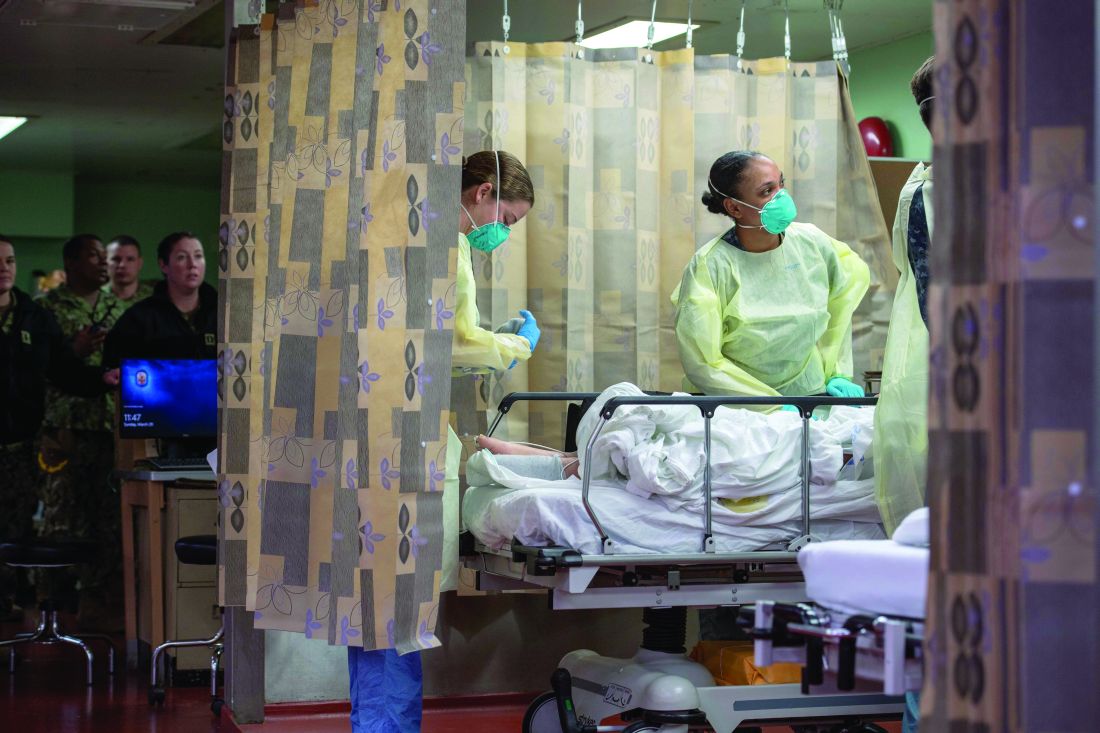
Care aboard the ship, which consists of open-bay medical wards, ranged from basic medical and surgical care to critical care and trauma. The most common procedures were cholecystectomies and orthopedic procedures, and the average length of stay was 4-5 days, according to Cdr. Blevins. Over the course of the mission, the medical professionals conducted 36 surgeries, 77 x-ray exams, 26 CT scans, and administered hundreds of ancillary studies ranging from routine labs to high-end x-rays and blood transfusion support.
“Within our ICU, we did have some end-of-life patients who ended up dying on our ship in comfort care,” Cdr. Blevins said. “Fortunately, we had a wonderful ICU team who had a great deal of experience with end-of-life care and were able to take care of these patients very comfortably and ensure good communication with family and loved ones during that time. In most instances we tried to make sure that people got to FaceTime or video chat with their loved one before they passed away.”
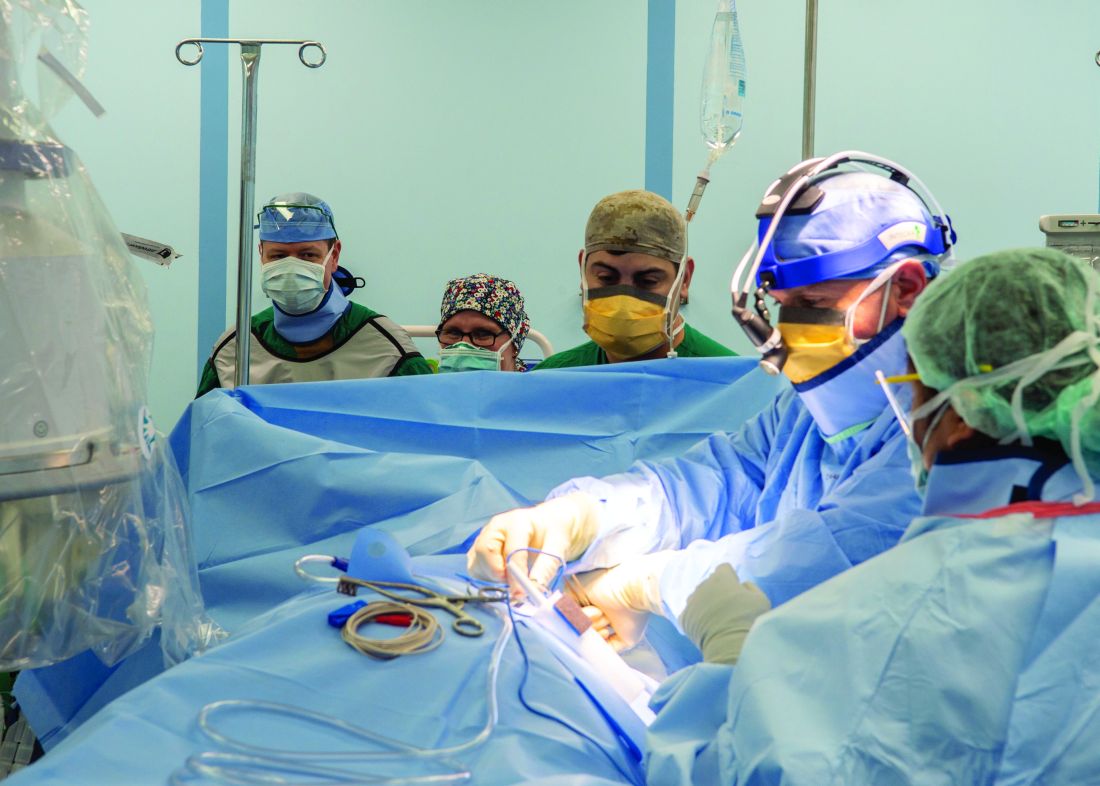
The Mercy, which includes 12 operating rooms, four x-ray units, and one CAT-scan unit, was not equipped to deliver pediatric or obstetrical care. Other unavailable services included psychiatry, oncology, cardiac and thoracic surgery, nuclear medicine, MRI, mammography, electrophysiology, cardiac catheterization, negative-pressure isolation, speech therapy, and occupational therapy.
Not your typical hospital experience
But for patients who did receive medical care aboard the Mercy – which made three 150-day deployments in recent years for the military-led humanitarian response known as Pacific Partnership in 2015, 2016, and 2018 – it was an experience that they are unlikely to forget.
“Every time a patient left the ship, our team on the ground surveyed them to see how their experience was and see what we could do to improve,” Cdr. Blevins said. “Across the board, they were all very appreciative of the medical care. We had a couple of veterans on board. They got [USNS Mercy] hats on their way out and seemed to very much enjoy a slightly different experience than they would get at a regular hospital.”
Capt. Rotruck added that the enthusiasm crew members had for supporting fellow Americans “really energized our team and really saturated that caring aspect of the people who interacted directly with patients,” he said. “It wasn’t just the physicians and nurses, but it was the staff delivering the food and coming to take blood samples and every other interaction that the patients had with our team. I think they really felt that enthusiasm for being there and supporting our neighbors in LA [Los Angeles].”
Crew life aboard the Mercy
Just as with any hospital on shore, personnel aboard the Mercy practiced preventive hygiene measures recommended by the Centers for Disease Control and Prevention to help prevent the spread of COVID-19, such as wearing cloth face masks, spacing out tables in the dining hall, closing indoor gyms, and devising creative ways to stay physically fit. Popular options included jogging around the perimeter of the ship and practicing yoga and calisthenics on the deck, “making sure you were physically distanced appropriately, and when you were done, putting your mask back on,” Cdr. Blevins said. Others supplemented their workouts with a pull-up bar on the deck. “In addition, we have a series of ramps that run on the starboard side of the ship that we can use for patient movement with litters on wheels or patient beds,” Capt. Rotruck said. “The uphill portion of those ramps represents a good workout opportunity as well.”
Downtime in an era of physical distancing also afforded crew members the opportunity to call or FaceTime with loved ones, watch streamed TV shows and movies, and work on their own professional development. Some continued with coursework for online degree programs offered by colleges and universities they were enrolled in, while some enlisted personnel used the time to complete the Navy Enlisted Warfare Qualification Programs Instruction, which issues the basic overarching requirements for the qualification and designation of all enlisted warfare programs.
“As you can imagine, people spend a lot of time learning how the ship works and how it integrates into larger naval forces and so forth,” Capt. Rotruck said. “Not just our ship but also other ships: their weapons systems and defense mechanisms and navigation systems. We had people spending a significant amount of time working on that. We had people complete their Enlisted Surface Warfare qualification while we were on the mission.”
End of the mission
Mercy returned to its home base in San Diego on May 15, but about 60 medical personnel stayed behind in Los Angeles to support Federal Emergency Management Agency (FEMA), state, and local health care professionals. Some worked at a site where clinicians provided care for COVID-19–positive patients who had been transferred from area skilled nursing facilities.
In addition, a team consisting of one nurse and five corpsmen “would go out to individual skilled nursing facilities and mainly conduct assessments and training, such as training in donning proper PPE [personal protective equipment] and determining what needs they had,” Capt. Rotruck said. “They met those needs if possible or [communicated with California officials] and let them know what the requirements were and what the needs were in that facility.” The assignment for those who stayed behind ended on May 31.
On the opposite coast, Mercy’s sister ship, USNS Comfort, arrived in New York Harbor from Norfolk, Va., on March 30 and spent 3½ weeks assisting area hospitals in the COVID-19 pandemic fight. A few days into the mission, Comfort’s internal spaces were reconfigured to create separate COVID-negative and COVID-positive sections. Medical teams aboard the ship cared for a total of 182 patients during the assignment.
Looking back on Mercy’s mission, Cdr. Blevins marveled at the sense of teamwork that unfolded. “We have quarterly training exercises with a core set of personnel, [and] we train getting ready for activation in 5 days,” she said. “All of that training kicks in and it comes to fruition in a mission like this. It was terrific to see a group of very disparate subject matter experts from all over the country come together with one purpose: which was to serve our own country during the pandemic.”
Capt. Rotruck pointed out that the experience enabled enlisted and nonenlisted physicians to maintain their skill sets during a time when military and civilian hospitals had stopped doing elective procedures and routine appointments. “The fact that those people were able to come on board the ship and continue to conduct their medical practice and maintain their skills and competencies in an environment that they weren’t quite used to is great,” he said. “Otherwise, some of those medical personnel would have been sitting idle, wherever they were from. This is the power of Navy medicine on behalf of our country.”
When the hospital ship USNS Mercy departed San Diego’s Naval Station North Island on March 23, 2020, to support the Department of Defense efforts in Los Angeles during the coronavirus outbreak, Commander Erin Blevins remembers the crew’s excitement was palpable.
“We normally do partnerships abroad and respond to tsunamis and earthquakes,” said Cdr. Blevins, MD, a pediatric hematologist-oncologist who served as director of medical services for the mission. “This was a slight change in situation, but still disaster relief in the form of a pandemic. We switched our mindset to putting together the best experts for an infectious disease pandemic versus an earthquake disaster relief.”
A new mission

The 1,000-bed Mercy ship – a converted San Clemente–class oil tanker that was delivered in 1986 – spent nearly 50 days pier side in Los Angeles as a referral hospital for non–COVID-19 patients, so that clinicians at Los Angeles area hospitals could care for an anticipated surge of COVID-19 patients. “We went into it with expectations of, ‘We’ll treat as many patients as you need us to take,” Cdr. Blevins recalled. “I don’t even think Los Angeles [health officials] knew exactly where they were going to peak and what the need was going to be.”
Between March 29 and May 15, about 1,071 medical personnel aboard the Mercy cared for 77 patients with an average age of 53 years who were referred from 11 Los Angeles area hospitals. The physicians, nurses, and other medical support personnel were drawn from military treatment facilities across the country. “We had additional people join us as we scoped the mission to be more medically heavy and surgically light,” said Captain John Rotruck, MD, an anesthesiologist who is commanding officer of Mercy’s medical treatment facility. “We did adjust to make sure that we had the right staffing mix to meet the parameters that we were assigned. That was the crux of the change: a change in flavors of staffing to ensure that we focused on ICU and ward medical care as opposed to very heavy surgical care in support of a combat operation.”
About 10% of the team consisted of reservists who volunteered for the mission. “There’s no way you could have walked around the ship and known who was active duty and who was reservist,” said Capt. Rotruck, who was formerly chief of staff at Walter Reed National Military Medical Center, Bethesda, Md. “They worked together so well, and I think that marriage of active duty who are used to working in a military medical treatment facility – in our case, a Navy medical treatment facility – together with our reservist physician colleagues who work in civilian facilities around the country, was beneficial. It was a synergistic relationship. I think both sides walked away learning quite a bit from each other.”
Start with screening
All crew members underwent a temperature check and completed a health screening questionnaire: once before departing their home of record and again before boarding Mercy. Based on those results, crew members and medical staff were screened for COVID-19 and tested as needed in order to minimize the risk of an outbreak aboard the ship.
Fewer than 1% of crew members developed COVID-19 or tested positive for the virus during the mission, according to Capt. Rotruck. Affected individuals were isolated and quarantined. “All staff have recovered and are doing well,” he said.
Mercy personnel worked with local health officials to ensure that all patients transferred to the ship tested negative for COVID-19. Physicians aboard the Mercy then worked directly with the patients’ civilian physician to ensure a safe and thorough turnover process before the patients were transferred.
From basic medical to trauma care

Care aboard the ship, which consists of open-bay medical wards, ranged from basic medical and surgical care to critical care and trauma. The most common procedures were cholecystectomies and orthopedic procedures, and the average length of stay was 4-5 days, according to Cdr. Blevins. Over the course of the mission, the medical professionals conducted 36 surgeries, 77 x-ray exams, 26 CT scans, and administered hundreds of ancillary studies ranging from routine labs to high-end x-rays and blood transfusion support.
“Within our ICU, we did have some end-of-life patients who ended up dying on our ship in comfort care,” Cdr. Blevins said. “Fortunately, we had a wonderful ICU team who had a great deal of experience with end-of-life care and were able to take care of these patients very comfortably and ensure good communication with family and loved ones during that time. In most instances we tried to make sure that people got to FaceTime or video chat with their loved one before they passed away.”

The Mercy, which includes 12 operating rooms, four x-ray units, and one CAT-scan unit, was not equipped to deliver pediatric or obstetrical care. Other unavailable services included psychiatry, oncology, cardiac and thoracic surgery, nuclear medicine, MRI, mammography, electrophysiology, cardiac catheterization, negative-pressure isolation, speech therapy, and occupational therapy.
Not your typical hospital experience
But for patients who did receive medical care aboard the Mercy – which made three 150-day deployments in recent years for the military-led humanitarian response known as Pacific Partnership in 2015, 2016, and 2018 – it was an experience that they are unlikely to forget.
“Every time a patient left the ship, our team on the ground surveyed them to see how their experience was and see what we could do to improve,” Cdr. Blevins said. “Across the board, they were all very appreciative of the medical care. We had a couple of veterans on board. They got [USNS Mercy] hats on their way out and seemed to very much enjoy a slightly different experience than they would get at a regular hospital.”
Capt. Rotruck added that the enthusiasm crew members had for supporting fellow Americans “really energized our team and really saturated that caring aspect of the people who interacted directly with patients,” he said. “It wasn’t just the physicians and nurses, but it was the staff delivering the food and coming to take blood samples and every other interaction that the patients had with our team. I think they really felt that enthusiasm for being there and supporting our neighbors in LA [Los Angeles].”
Crew life aboard the Mercy
Just as with any hospital on shore, personnel aboard the Mercy practiced preventive hygiene measures recommended by the Centers for Disease Control and Prevention to help prevent the spread of COVID-19, such as wearing cloth face masks, spacing out tables in the dining hall, closing indoor gyms, and devising creative ways to stay physically fit. Popular options included jogging around the perimeter of the ship and practicing yoga and calisthenics on the deck, “making sure you were physically distanced appropriately, and when you were done, putting your mask back on,” Cdr. Blevins said. Others supplemented their workouts with a pull-up bar on the deck. “In addition, we have a series of ramps that run on the starboard side of the ship that we can use for patient movement with litters on wheels or patient beds,” Capt. Rotruck said. “The uphill portion of those ramps represents a good workout opportunity as well.”
Downtime in an era of physical distancing also afforded crew members the opportunity to call or FaceTime with loved ones, watch streamed TV shows and movies, and work on their own professional development. Some continued with coursework for online degree programs offered by colleges and universities they were enrolled in, while some enlisted personnel used the time to complete the Navy Enlisted Warfare Qualification Programs Instruction, which issues the basic overarching requirements for the qualification and designation of all enlisted warfare programs.
“As you can imagine, people spend a lot of time learning how the ship works and how it integrates into larger naval forces and so forth,” Capt. Rotruck said. “Not just our ship but also other ships: their weapons systems and defense mechanisms and navigation systems. We had people spending a significant amount of time working on that. We had people complete their Enlisted Surface Warfare qualification while we were on the mission.”
End of the mission
Mercy returned to its home base in San Diego on May 15, but about 60 medical personnel stayed behind in Los Angeles to support Federal Emergency Management Agency (FEMA), state, and local health care professionals. Some worked at a site where clinicians provided care for COVID-19–positive patients who had been transferred from area skilled nursing facilities.
In addition, a team consisting of one nurse and five corpsmen “would go out to individual skilled nursing facilities and mainly conduct assessments and training, such as training in donning proper PPE [personal protective equipment] and determining what needs they had,” Capt. Rotruck said. “They met those needs if possible or [communicated with California officials] and let them know what the requirements were and what the needs were in that facility.” The assignment for those who stayed behind ended on May 31.
On the opposite coast, Mercy’s sister ship, USNS Comfort, arrived in New York Harbor from Norfolk, Va., on March 30 and spent 3½ weeks assisting area hospitals in the COVID-19 pandemic fight. A few days into the mission, Comfort’s internal spaces were reconfigured to create separate COVID-negative and COVID-positive sections. Medical teams aboard the ship cared for a total of 182 patients during the assignment.
Looking back on Mercy’s mission, Cdr. Blevins marveled at the sense of teamwork that unfolded. “We have quarterly training exercises with a core set of personnel, [and] we train getting ready for activation in 5 days,” she said. “All of that training kicks in and it comes to fruition in a mission like this. It was terrific to see a group of very disparate subject matter experts from all over the country come together with one purpose: which was to serve our own country during the pandemic.”
Capt. Rotruck pointed out that the experience enabled enlisted and nonenlisted physicians to maintain their skill sets during a time when military and civilian hospitals had stopped doing elective procedures and routine appointments. “The fact that those people were able to come on board the ship and continue to conduct their medical practice and maintain their skills and competencies in an environment that they weren’t quite used to is great,” he said. “Otherwise, some of those medical personnel would have been sitting idle, wherever they were from. This is the power of Navy medicine on behalf of our country.”
When the hospital ship USNS Mercy departed San Diego’s Naval Station North Island on March 23, 2020, to support the Department of Defense efforts in Los Angeles during the coronavirus outbreak, Commander Erin Blevins remembers the crew’s excitement was palpable.
“We normally do partnerships abroad and respond to tsunamis and earthquakes,” said Cdr. Blevins, MD, a pediatric hematologist-oncologist who served as director of medical services for the mission. “This was a slight change in situation, but still disaster relief in the form of a pandemic. We switched our mindset to putting together the best experts for an infectious disease pandemic versus an earthquake disaster relief.”
A new mission

The 1,000-bed Mercy ship – a converted San Clemente–class oil tanker that was delivered in 1986 – spent nearly 50 days pier side in Los Angeles as a referral hospital for non–COVID-19 patients, so that clinicians at Los Angeles area hospitals could care for an anticipated surge of COVID-19 patients. “We went into it with expectations of, ‘We’ll treat as many patients as you need us to take,” Cdr. Blevins recalled. “I don’t even think Los Angeles [health officials] knew exactly where they were going to peak and what the need was going to be.”
Between March 29 and May 15, about 1,071 medical personnel aboard the Mercy cared for 77 patients with an average age of 53 years who were referred from 11 Los Angeles area hospitals. The physicians, nurses, and other medical support personnel were drawn from military treatment facilities across the country. “We had additional people join us as we scoped the mission to be more medically heavy and surgically light,” said Captain John Rotruck, MD, an anesthesiologist who is commanding officer of Mercy’s medical treatment facility. “We did adjust to make sure that we had the right staffing mix to meet the parameters that we were assigned. That was the crux of the change: a change in flavors of staffing to ensure that we focused on ICU and ward medical care as opposed to very heavy surgical care in support of a combat operation.”
About 10% of the team consisted of reservists who volunteered for the mission. “There’s no way you could have walked around the ship and known who was active duty and who was reservist,” said Capt. Rotruck, who was formerly chief of staff at Walter Reed National Military Medical Center, Bethesda, Md. “They worked together so well, and I think that marriage of active duty who are used to working in a military medical treatment facility – in our case, a Navy medical treatment facility – together with our reservist physician colleagues who work in civilian facilities around the country, was beneficial. It was a synergistic relationship. I think both sides walked away learning quite a bit from each other.”
Start with screening
All crew members underwent a temperature check and completed a health screening questionnaire: once before departing their home of record and again before boarding Mercy. Based on those results, crew members and medical staff were screened for COVID-19 and tested as needed in order to minimize the risk of an outbreak aboard the ship.
Fewer than 1% of crew members developed COVID-19 or tested positive for the virus during the mission, according to Capt. Rotruck. Affected individuals were isolated and quarantined. “All staff have recovered and are doing well,” he said.
Mercy personnel worked with local health officials to ensure that all patients transferred to the ship tested negative for COVID-19. Physicians aboard the Mercy then worked directly with the patients’ civilian physician to ensure a safe and thorough turnover process before the patients were transferred.
From basic medical to trauma care

Care aboard the ship, which consists of open-bay medical wards, ranged from basic medical and surgical care to critical care and trauma. The most common procedures were cholecystectomies and orthopedic procedures, and the average length of stay was 4-5 days, according to Cdr. Blevins. Over the course of the mission, the medical professionals conducted 36 surgeries, 77 x-ray exams, 26 CT scans, and administered hundreds of ancillary studies ranging from routine labs to high-end x-rays and blood transfusion support.
“Within our ICU, we did have some end-of-life patients who ended up dying on our ship in comfort care,” Cdr. Blevins said. “Fortunately, we had a wonderful ICU team who had a great deal of experience with end-of-life care and were able to take care of these patients very comfortably and ensure good communication with family and loved ones during that time. In most instances we tried to make sure that people got to FaceTime or video chat with their loved one before they passed away.”

The Mercy, which includes 12 operating rooms, four x-ray units, and one CAT-scan unit, was not equipped to deliver pediatric or obstetrical care. Other unavailable services included psychiatry, oncology, cardiac and thoracic surgery, nuclear medicine, MRI, mammography, electrophysiology, cardiac catheterization, negative-pressure isolation, speech therapy, and occupational therapy.
Not your typical hospital experience
But for patients who did receive medical care aboard the Mercy – which made three 150-day deployments in recent years for the military-led humanitarian response known as Pacific Partnership in 2015, 2016, and 2018 – it was an experience that they are unlikely to forget.
“Every time a patient left the ship, our team on the ground surveyed them to see how their experience was and see what we could do to improve,” Cdr. Blevins said. “Across the board, they were all very appreciative of the medical care. We had a couple of veterans on board. They got [USNS Mercy] hats on their way out and seemed to very much enjoy a slightly different experience than they would get at a regular hospital.”
Capt. Rotruck added that the enthusiasm crew members had for supporting fellow Americans “really energized our team and really saturated that caring aspect of the people who interacted directly with patients,” he said. “It wasn’t just the physicians and nurses, but it was the staff delivering the food and coming to take blood samples and every other interaction that the patients had with our team. I think they really felt that enthusiasm for being there and supporting our neighbors in LA [Los Angeles].”
Crew life aboard the Mercy
Just as with any hospital on shore, personnel aboard the Mercy practiced preventive hygiene measures recommended by the Centers for Disease Control and Prevention to help prevent the spread of COVID-19, such as wearing cloth face masks, spacing out tables in the dining hall, closing indoor gyms, and devising creative ways to stay physically fit. Popular options included jogging around the perimeter of the ship and practicing yoga and calisthenics on the deck, “making sure you were physically distanced appropriately, and when you were done, putting your mask back on,” Cdr. Blevins said. Others supplemented their workouts with a pull-up bar on the deck. “In addition, we have a series of ramps that run on the starboard side of the ship that we can use for patient movement with litters on wheels or patient beds,” Capt. Rotruck said. “The uphill portion of those ramps represents a good workout opportunity as well.”
Downtime in an era of physical distancing also afforded crew members the opportunity to call or FaceTime with loved ones, watch streamed TV shows and movies, and work on their own professional development. Some continued with coursework for online degree programs offered by colleges and universities they were enrolled in, while some enlisted personnel used the time to complete the Navy Enlisted Warfare Qualification Programs Instruction, which issues the basic overarching requirements for the qualification and designation of all enlisted warfare programs.
“As you can imagine, people spend a lot of time learning how the ship works and how it integrates into larger naval forces and so forth,” Capt. Rotruck said. “Not just our ship but also other ships: their weapons systems and defense mechanisms and navigation systems. We had people spending a significant amount of time working on that. We had people complete their Enlisted Surface Warfare qualification while we were on the mission.”
End of the mission
Mercy returned to its home base in San Diego on May 15, but about 60 medical personnel stayed behind in Los Angeles to support Federal Emergency Management Agency (FEMA), state, and local health care professionals. Some worked at a site where clinicians provided care for COVID-19–positive patients who had been transferred from area skilled nursing facilities.
In addition, a team consisting of one nurse and five corpsmen “would go out to individual skilled nursing facilities and mainly conduct assessments and training, such as training in donning proper PPE [personal protective equipment] and determining what needs they had,” Capt. Rotruck said. “They met those needs if possible or [communicated with California officials] and let them know what the requirements were and what the needs were in that facility.” The assignment for those who stayed behind ended on May 31.
On the opposite coast, Mercy’s sister ship, USNS Comfort, arrived in New York Harbor from Norfolk, Va., on March 30 and spent 3½ weeks assisting area hospitals in the COVID-19 pandemic fight. A few days into the mission, Comfort’s internal spaces were reconfigured to create separate COVID-negative and COVID-positive sections. Medical teams aboard the ship cared for a total of 182 patients during the assignment.
Looking back on Mercy’s mission, Cdr. Blevins marveled at the sense of teamwork that unfolded. “We have quarterly training exercises with a core set of personnel, [and] we train getting ready for activation in 5 days,” she said. “All of that training kicks in and it comes to fruition in a mission like this. It was terrific to see a group of very disparate subject matter experts from all over the country come together with one purpose: which was to serve our own country during the pandemic.”
Capt. Rotruck pointed out that the experience enabled enlisted and nonenlisted physicians to maintain their skill sets during a time when military and civilian hospitals had stopped doing elective procedures and routine appointments. “The fact that those people were able to come on board the ship and continue to conduct their medical practice and maintain their skills and competencies in an environment that they weren’t quite used to is great,” he said. “Otherwise, some of those medical personnel would have been sitting idle, wherever they were from. This is the power of Navy medicine on behalf of our country.”
Daily Recap: ED visits for life-threatening conditions plummet; COVID-19 imaging strategies for kids
Here are the stories our MDedge editors across specialties think you need to know about today:
ED visits drop for life-threatening conditions
Emergency department visits for myocardial infarction, stroke, and hyperglycemic crisis dropped substantially in the 10 weeks after COVID-19 was declared a national emergency, according to new research from the Centers for Disease Control and Prevention.
Compared with the 10-week period from Jan. 5 to March 14, ED visits were down by 23% for MI, 20% for stroke, and 10% for hyperglycemic crisis from March 15 to May 23.
“A short-term decline of this magnitude … is biologically implausible for MI and stroke, especially for older adults, and unlikely for hyperglycemic crisis, and the finding suggests that patients with these conditions either could not access care or were delaying or avoiding seeking care during the early pandemic period,” the researchers wrote in the Morbidity and Mortality Weekly Report. Read more.
Expert recommendations for pediatric COVID-19 imaging
A team of pulmonologists has synthesized the clinical and imaging characteristics of COVID-19 in children, and has issued recommendations for ordering imaging studies in suspected cases of the infection.
Current recommendations from the American College of Radiology (ACR) do not include chest computed tomography (CT) or chest radiography (CXR) as an upfront test to diagnose pediatric COVID-19, but the tests may still have a role in clinical monitoring, especially in patients with a moderate to severe disease course. The potential benefits of utilizing radiologic evaluation – such as establishing a baseline for monitoring disease progression – must be balanced with potential drawbacks, including radiation exposure and reduced availability of imaging resources owing to necessary cleaning and air turnover time.
Based on the most recent international guidelines for pediatric COVID-19 patient management, the authors developed an algorithm for performing imaging studies in suspected cases of COVID-19 pneumonia. The purpose of the tool is to support clinical decision-making around the utilization of CXR and CT to evaluate pediatric COVID-19 pneumonia. “The step by step algorithm addresses the selection, sequence and timing of imaging studies with multiple images illustrating key findings of COVID-19 pneumonia in the pediatric age group,” said Mary Cataletto, MD, of NYU Langone Health in Mineola, N.Y. Read more.
Cortisol levels on COVID-19 admission may be a marker of severity
Patients with COVID-19 who have high levels of the steroid hormone cortisol on admission to the hospital have a substantially increased risk of dying, according to new study findings.
Researchers assessed 535 patients admitted to major London hospitals. Of these, 403 patients were diagnosed with COVID-19 based on a positive result on real-time polymerase chain reaction testing or a strong clinical and radiological suspicion, despite a negative test. Mean cortisol concentrations in patients with COVID-19 were significantly higher than those not diagnosed with the virus and as of May 8, significantly more patients with COVID-19 died than those without (27.8% vs 6.8%).
Measuring cortisol on admission is potentially “another simple marker to use alongside oxygen saturation levels to help us identify which patients need to be admitted immediately, and which may not,” said Waljit S. Dhillo, MBBS, PhD, head of the division of diabetes, endocrinology and metabolism at Imperial College London.
“Having an early indicator of which patients may deteriorate more quickly will help us with providing the best level of care as quickly as possible. In addition, we can also take cortisol levels into account when we are working out how best to treat our patients,” he said. Read more.
Normal-weight prediabetes patients can benefit from lifestyle changes
Adults of normal weight with prediabetes may derive at least as much benefit from lifestyle health coaching programs as adults who are overweight or obese, results of a recent nonrandomized, real-world study show.
Fasting plasma glucose (FPG) normalized in about 63% of prediabetic adults with normal body mass index (BMI) participating in a personalized coaching program that emphasized exercise, nutrition, and weight management. In contrast, FPG normalized in about 52% of overweight and 44% of obese prediabetic individuals participating in the program.
“It is interesting to note that, although the normal weight group lost the least amount of weight, they still benefited from the lifestyle health coaching program... having a resultant greatest decrease in fasting plasma glucose and normalization to a range of someone without prediabetes,” said researcher Mandy Salmon, MS, a medical student at the University of Pennsylvania, Philadelphia. She presented the findings at the virtual annual scientific sessions of the American Diabetes Association. Read more.
Diabetes-related amputations rise in older adults
The recent resurgence in diabetes-related lower-extremity amputations in the United States is not limited to younger adults, according to the author of a recent study that documents similar increases among an older population of Medicare beneficiaries.
While the rate of amputations fell among these older adults from 2000 to 2009, it increased significantly from 2009 to 2017, albeit at a “less severe rate” than recently reported in younger populations, according to study investigator Jessica Harding, PhD, an assistant professor in the department of surgery at Emory University, Atlanta. Dr. Harding reported the results at the virtual annual scientific sessions of the American Diabetes Association.
The rate of nontraumatic lower extremity amputation (NLEA) was ticking upward by more than 1% per year over the 2009-2017 period. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
ED visits drop for life-threatening conditions
Emergency department visits for myocardial infarction, stroke, and hyperglycemic crisis dropped substantially in the 10 weeks after COVID-19 was declared a national emergency, according to new research from the Centers for Disease Control and Prevention.
Compared with the 10-week period from Jan. 5 to March 14, ED visits were down by 23% for MI, 20% for stroke, and 10% for hyperglycemic crisis from March 15 to May 23.
“A short-term decline of this magnitude … is biologically implausible for MI and stroke, especially for older adults, and unlikely for hyperglycemic crisis, and the finding suggests that patients with these conditions either could not access care or were delaying or avoiding seeking care during the early pandemic period,” the researchers wrote in the Morbidity and Mortality Weekly Report. Read more.
Expert recommendations for pediatric COVID-19 imaging
A team of pulmonologists has synthesized the clinical and imaging characteristics of COVID-19 in children, and has issued recommendations for ordering imaging studies in suspected cases of the infection.
Current recommendations from the American College of Radiology (ACR) do not include chest computed tomography (CT) or chest radiography (CXR) as an upfront test to diagnose pediatric COVID-19, but the tests may still have a role in clinical monitoring, especially in patients with a moderate to severe disease course. The potential benefits of utilizing radiologic evaluation – such as establishing a baseline for monitoring disease progression – must be balanced with potential drawbacks, including radiation exposure and reduced availability of imaging resources owing to necessary cleaning and air turnover time.
Based on the most recent international guidelines for pediatric COVID-19 patient management, the authors developed an algorithm for performing imaging studies in suspected cases of COVID-19 pneumonia. The purpose of the tool is to support clinical decision-making around the utilization of CXR and CT to evaluate pediatric COVID-19 pneumonia. “The step by step algorithm addresses the selection, sequence and timing of imaging studies with multiple images illustrating key findings of COVID-19 pneumonia in the pediatric age group,” said Mary Cataletto, MD, of NYU Langone Health in Mineola, N.Y. Read more.
Cortisol levels on COVID-19 admission may be a marker of severity
Patients with COVID-19 who have high levels of the steroid hormone cortisol on admission to the hospital have a substantially increased risk of dying, according to new study findings.
Researchers assessed 535 patients admitted to major London hospitals. Of these, 403 patients were diagnosed with COVID-19 based on a positive result on real-time polymerase chain reaction testing or a strong clinical and radiological suspicion, despite a negative test. Mean cortisol concentrations in patients with COVID-19 were significantly higher than those not diagnosed with the virus and as of May 8, significantly more patients with COVID-19 died than those without (27.8% vs 6.8%).
Measuring cortisol on admission is potentially “another simple marker to use alongside oxygen saturation levels to help us identify which patients need to be admitted immediately, and which may not,” said Waljit S. Dhillo, MBBS, PhD, head of the division of diabetes, endocrinology and metabolism at Imperial College London.
“Having an early indicator of which patients may deteriorate more quickly will help us with providing the best level of care as quickly as possible. In addition, we can also take cortisol levels into account when we are working out how best to treat our patients,” he said. Read more.
Normal-weight prediabetes patients can benefit from lifestyle changes
Adults of normal weight with prediabetes may derive at least as much benefit from lifestyle health coaching programs as adults who are overweight or obese, results of a recent nonrandomized, real-world study show.
Fasting plasma glucose (FPG) normalized in about 63% of prediabetic adults with normal body mass index (BMI) participating in a personalized coaching program that emphasized exercise, nutrition, and weight management. In contrast, FPG normalized in about 52% of overweight and 44% of obese prediabetic individuals participating in the program.
“It is interesting to note that, although the normal weight group lost the least amount of weight, they still benefited from the lifestyle health coaching program... having a resultant greatest decrease in fasting plasma glucose and normalization to a range of someone without prediabetes,” said researcher Mandy Salmon, MS, a medical student at the University of Pennsylvania, Philadelphia. She presented the findings at the virtual annual scientific sessions of the American Diabetes Association. Read more.
Diabetes-related amputations rise in older adults
The recent resurgence in diabetes-related lower-extremity amputations in the United States is not limited to younger adults, according to the author of a recent study that documents similar increases among an older population of Medicare beneficiaries.
While the rate of amputations fell among these older adults from 2000 to 2009, it increased significantly from 2009 to 2017, albeit at a “less severe rate” than recently reported in younger populations, according to study investigator Jessica Harding, PhD, an assistant professor in the department of surgery at Emory University, Atlanta. Dr. Harding reported the results at the virtual annual scientific sessions of the American Diabetes Association.
The rate of nontraumatic lower extremity amputation (NLEA) was ticking upward by more than 1% per year over the 2009-2017 period. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
ED visits drop for life-threatening conditions
Emergency department visits for myocardial infarction, stroke, and hyperglycemic crisis dropped substantially in the 10 weeks after COVID-19 was declared a national emergency, according to new research from the Centers for Disease Control and Prevention.
Compared with the 10-week period from Jan. 5 to March 14, ED visits were down by 23% for MI, 20% for stroke, and 10% for hyperglycemic crisis from March 15 to May 23.
“A short-term decline of this magnitude … is biologically implausible for MI and stroke, especially for older adults, and unlikely for hyperglycemic crisis, and the finding suggests that patients with these conditions either could not access care or were delaying or avoiding seeking care during the early pandemic period,” the researchers wrote in the Morbidity and Mortality Weekly Report. Read more.
Expert recommendations for pediatric COVID-19 imaging
A team of pulmonologists has synthesized the clinical and imaging characteristics of COVID-19 in children, and has issued recommendations for ordering imaging studies in suspected cases of the infection.
Current recommendations from the American College of Radiology (ACR) do not include chest computed tomography (CT) or chest radiography (CXR) as an upfront test to diagnose pediatric COVID-19, but the tests may still have a role in clinical monitoring, especially in patients with a moderate to severe disease course. The potential benefits of utilizing radiologic evaluation – such as establishing a baseline for monitoring disease progression – must be balanced with potential drawbacks, including radiation exposure and reduced availability of imaging resources owing to necessary cleaning and air turnover time.
Based on the most recent international guidelines for pediatric COVID-19 patient management, the authors developed an algorithm for performing imaging studies in suspected cases of COVID-19 pneumonia. The purpose of the tool is to support clinical decision-making around the utilization of CXR and CT to evaluate pediatric COVID-19 pneumonia. “The step by step algorithm addresses the selection, sequence and timing of imaging studies with multiple images illustrating key findings of COVID-19 pneumonia in the pediatric age group,” said Mary Cataletto, MD, of NYU Langone Health in Mineola, N.Y. Read more.
Cortisol levels on COVID-19 admission may be a marker of severity
Patients with COVID-19 who have high levels of the steroid hormone cortisol on admission to the hospital have a substantially increased risk of dying, according to new study findings.
Researchers assessed 535 patients admitted to major London hospitals. Of these, 403 patients were diagnosed with COVID-19 based on a positive result on real-time polymerase chain reaction testing or a strong clinical and radiological suspicion, despite a negative test. Mean cortisol concentrations in patients with COVID-19 were significantly higher than those not diagnosed with the virus and as of May 8, significantly more patients with COVID-19 died than those without (27.8% vs 6.8%).
Measuring cortisol on admission is potentially “another simple marker to use alongside oxygen saturation levels to help us identify which patients need to be admitted immediately, and which may not,” said Waljit S. Dhillo, MBBS, PhD, head of the division of diabetes, endocrinology and metabolism at Imperial College London.
“Having an early indicator of which patients may deteriorate more quickly will help us with providing the best level of care as quickly as possible. In addition, we can also take cortisol levels into account when we are working out how best to treat our patients,” he said. Read more.
Normal-weight prediabetes patients can benefit from lifestyle changes
Adults of normal weight with prediabetes may derive at least as much benefit from lifestyle health coaching programs as adults who are overweight or obese, results of a recent nonrandomized, real-world study show.
Fasting plasma glucose (FPG) normalized in about 63% of prediabetic adults with normal body mass index (BMI) participating in a personalized coaching program that emphasized exercise, nutrition, and weight management. In contrast, FPG normalized in about 52% of overweight and 44% of obese prediabetic individuals participating in the program.
“It is interesting to note that, although the normal weight group lost the least amount of weight, they still benefited from the lifestyle health coaching program... having a resultant greatest decrease in fasting plasma glucose and normalization to a range of someone without prediabetes,” said researcher Mandy Salmon, MS, a medical student at the University of Pennsylvania, Philadelphia. She presented the findings at the virtual annual scientific sessions of the American Diabetes Association. Read more.
Diabetes-related amputations rise in older adults
The recent resurgence in diabetes-related lower-extremity amputations in the United States is not limited to younger adults, according to the author of a recent study that documents similar increases among an older population of Medicare beneficiaries.
While the rate of amputations fell among these older adults from 2000 to 2009, it increased significantly from 2009 to 2017, albeit at a “less severe rate” than recently reported in younger populations, according to study investigator Jessica Harding, PhD, an assistant professor in the department of surgery at Emory University, Atlanta. Dr. Harding reported the results at the virtual annual scientific sessions of the American Diabetes Association.
The rate of nontraumatic lower extremity amputation (NLEA) was ticking upward by more than 1% per year over the 2009-2017 period. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Guidance on infection prevention for health care personnel
As we reopen our offices we are faced with the challenge of determining the best way to do it safely – protecting ourselves, our staff, and our patients.
In this column we will focus on selected details of the recommendations from IDSA and the CDC that may be helpful in primary care offices.
Face masks
Many clinicians have asked whether a physician should use a mask while seeing patients without COVID-19 in the office, and if yes, which type. The IDSA guideline states that mask usage is imperative for reducing the risk of health care workers contracting COVID-19.1 The evidence is derived from a number of sources, including a retrospective study from Wuhan (China) University that examined two groups of health care workers during the outbreak. The first group wore N95 masks and washed their hands frequently, while the second group did not wear masks and washed their hands less frequently. In the group that took greater actions to protect themselves, none of the 493 staff members contracted COVID-19, compared with 10 of 213 staff members in the other group. The decrease in infection rate occurred in the group that wore masks despite the fact that this group had 733% more exposure to COVID-19 patients.2 Further evidence came from a case-control study done in hospitals in Hong Kong during the 2003 SARS-CoV outbreak.3 This study showed that mask wearing was the most significant intervention for reducing infection, followed by gowning, and then handwashing. These findings make it clear that mask usage is a must for all health care providers who may be caring for patients who could have COVID-19.
The guideline also reviews evidence about the use of surgical masks versus N95 masks. On reviewing indirect evidence from the SARS-CoV epidemic, IDSA found that wearing any mask – surgical or N95 – led to a large reduction in the risk of developing an infection. In this systematic review of five observational studies in health care personnel, for those wearing surgical masks, the odds ratio for developing an infection was 0.13 (95% CI, 0.03-0.62), and for those wearing N95 masks, the odds ratio was 0.12 (95% CI, 0.06-0.26). There was not a significant difference between risk reductions for those who wore surgical masks and N95 masks, respectively.1,4 The IDSA guideline panel recommended “that health care personnel caring for patients with suspected or known COVID-19 use either a surgical mask or N95 respirator ... as part of appropriate PPE.” Since there is not a significant difference in outcomes between those who use surgical masks and those who use N95 respirators, and the IDSA guideline states either type of mask is considered appropriate when taking care of patients with suspected or known COVID-19, in our opinion, use of surgical masks rather than N95s is sufficient when performing low-risk activities. Such activities include seeing patients who do not have a high likelihood of COVID-19 in the office setting.
The IDSA recommendation also discusses universal masking, defined as both patients and clinicians wearing masks. The recommendation is supported by the findings of a study in which universal mask usage was used to prevent the spread of H1N 1 during the 2009 outbreak. In this study of staff members and patients exposed to H1N1 who all wore masks, only 0.48% of 836 acquired infection. In the same study, not wearing a mask by either the provider or patient increased the risk of infection.5 Also, in a prospective study of hematopoietic stem cell transplant patients, universal masking caused infection rates to drop from 10.3% to 4.4%.6
The IDSA guideline states the following: “There may be some, albeit uncertain, benefit to universal masking in the absence of resource constraints. However, the benefits of universal masking with surgical masks should be weighed against the risk of increasing the PPE burn rate and contextualized to the background COVID-19 prevalence rate for asymptomatic or minimally symptomatic HCPs [health care providers] and visitors.”1
The CDC’s guidance statement says the following: “Continued community transmission has increased the number of individuals potentially exposed to and infectious with SARS-CoV-2. Fever and symptom screening have proven to be relatively ineffective in identifying all infected individuals, including HCPs. Symptom screening also will not identify individuals who are infected but otherwise asymptomatic or pre-symptomatic; additional interventions are needed to limit the unrecognized introduction of SARS-CoV-2 into healthcare settings by these individuals. As part of aggressive source control measures, healthcare facilities should consider implementing policies requiring everyone entering the facility to wear a cloth face covering (if tolerated) while in the building, regardless of symptoms.”7
It is our opinion, based on the CDC and IDSA recommendations, that both clinicians and patients should be required to wear masks when patients are seen in the office if possible. Many offices have instituted a policy that says, if a patient refuses to wear a mask during an office visit, then the patient will not be seen.
Eye protection
Many clinicians are uncertain about whether eye protection needs to be used when seeing asymptomatic patients. The IDSA acknowledges that there are not studies that have looked critically at eye protection, but the society also acknowledges “appropriate personal protective equipment includes, in addition to a mask or respirator, eye protection, gown and gloves.”1 In addition, the CDC recommends that, for healthcare workers located in areas with moderate or higher prevalence of COVID-19, HCPs should wear eye protection in addition to facemasks since they may encounter asymptomatic individuals with COVID-19.
Gowns and gloves
Gowns and gloves are recommended as a part of personal protective gear when caring for patients who have COVID-19. The IDSA guideline is clear in its recommendations, but does not cite evidence for having no gloves versus having gloves. Furthermore, they state that the evidence is insufficient to recommend double gloves, with the top glove used to take off a personal protective gown, and the inner glove discarded after the gown is removed. The CDC do not make recommendations for routine use of gloves in the care of patients who do not have COVID-19, even in areas where there may be asymptomatic COVID-19, and recommends standard precautions, specifically practicing hand hygiene before and after patient contact.8
The Bottom Line
When seeing patients with COVID-19, N-95 masks, goggles or face shields, gowns, and gloves should be used, with hand hygiene routinely practiced before and after seeing patients. For offices seeing patients not suspected of having COVID-19, the IDSA guideline clarifies that there is not a statistical difference in acquisition of infection with the use of surgical face masks vs N95 respirators. According to the CDC recommendations, eye protection in addition to facemasks should be used by the health care provider, and masks should be worn by patients. Hand hygiene should be used routinely before and after all patient contact. With use of these approaches, it should be safe for offices to reopen and see patients.
Neil Skolnik, MD, is professor of family and community medicine at the Thomas Jefferson University, Philadelphia, and associate director of the Family Medicine Residency Program at Abington (Pa.) Jefferson Health. Jeffrey Matthews, DO, is a second-year resident in the Family Medicine Residency at Abington Jefferson Health. For questions or comments, feel free to contact Dr. Skolnik on Twitter @NeilSkolnik.
References
1. Lynch JB, Davitkov P, Anderson DJ, et al. COVID-19 Guideline, Part 2: Infection Prevention. IDSA Home. https://www.idsociety.org/practice-guideline/covid-19-guideline-infection-prevention/. April 27, 2020. Accessed June 10, 2020.
2. J Hosp Infect. 2020 May;105(1):104-5.
3. Lancet. 2003;361(9368):1519-20.
4. Influenza Other Respir Viruses. 2020 Apr 4. doi: 2020;10.1111/irv.12745.
5. J Hosp Infect. 2010;74(3):271-7.
6. Clin Infect Dis. 2016;63(8):999-1006.
7. Centers for Disease Control and Prevention. Interim Infection Prevention and Control Recommendations for Patients with Suspected or Confirmed Coronavirus Disease 2019 (COVID-19) in Healthcare Settings. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-recommendations.html. Accessed Jun 16, 2020.
8. Centers for Disease Control and Prevention. Healthcare Infection Prevention and Control FAQs for COVID-19. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-faq.html. Accessed June 15, 2020.
As we reopen our offices we are faced with the challenge of determining the best way to do it safely – protecting ourselves, our staff, and our patients.
In this column we will focus on selected details of the recommendations from IDSA and the CDC that may be helpful in primary care offices.
Face masks
Many clinicians have asked whether a physician should use a mask while seeing patients without COVID-19 in the office, and if yes, which type. The IDSA guideline states that mask usage is imperative for reducing the risk of health care workers contracting COVID-19.1 The evidence is derived from a number of sources, including a retrospective study from Wuhan (China) University that examined two groups of health care workers during the outbreak. The first group wore N95 masks and washed their hands frequently, while the second group did not wear masks and washed their hands less frequently. In the group that took greater actions to protect themselves, none of the 493 staff members contracted COVID-19, compared with 10 of 213 staff members in the other group. The decrease in infection rate occurred in the group that wore masks despite the fact that this group had 733% more exposure to COVID-19 patients.2 Further evidence came from a case-control study done in hospitals in Hong Kong during the 2003 SARS-CoV outbreak.3 This study showed that mask wearing was the most significant intervention for reducing infection, followed by gowning, and then handwashing. These findings make it clear that mask usage is a must for all health care providers who may be caring for patients who could have COVID-19.
The guideline also reviews evidence about the use of surgical masks versus N95 masks. On reviewing indirect evidence from the SARS-CoV epidemic, IDSA found that wearing any mask – surgical or N95 – led to a large reduction in the risk of developing an infection. In this systematic review of five observational studies in health care personnel, for those wearing surgical masks, the odds ratio for developing an infection was 0.13 (95% CI, 0.03-0.62), and for those wearing N95 masks, the odds ratio was 0.12 (95% CI, 0.06-0.26). There was not a significant difference between risk reductions for those who wore surgical masks and N95 masks, respectively.1,4 The IDSA guideline panel recommended “that health care personnel caring for patients with suspected or known COVID-19 use either a surgical mask or N95 respirator ... as part of appropriate PPE.” Since there is not a significant difference in outcomes between those who use surgical masks and those who use N95 respirators, and the IDSA guideline states either type of mask is considered appropriate when taking care of patients with suspected or known COVID-19, in our opinion, use of surgical masks rather than N95s is sufficient when performing low-risk activities. Such activities include seeing patients who do not have a high likelihood of COVID-19 in the office setting.
The IDSA recommendation also discusses universal masking, defined as both patients and clinicians wearing masks. The recommendation is supported by the findings of a study in which universal mask usage was used to prevent the spread of H1N 1 during the 2009 outbreak. In this study of staff members and patients exposed to H1N1 who all wore masks, only 0.48% of 836 acquired infection. In the same study, not wearing a mask by either the provider or patient increased the risk of infection.5 Also, in a prospective study of hematopoietic stem cell transplant patients, universal masking caused infection rates to drop from 10.3% to 4.4%.6
The IDSA guideline states the following: “There may be some, albeit uncertain, benefit to universal masking in the absence of resource constraints. However, the benefits of universal masking with surgical masks should be weighed against the risk of increasing the PPE burn rate and contextualized to the background COVID-19 prevalence rate for asymptomatic or minimally symptomatic HCPs [health care providers] and visitors.”1
The CDC’s guidance statement says the following: “Continued community transmission has increased the number of individuals potentially exposed to and infectious with SARS-CoV-2. Fever and symptom screening have proven to be relatively ineffective in identifying all infected individuals, including HCPs. Symptom screening also will not identify individuals who are infected but otherwise asymptomatic or pre-symptomatic; additional interventions are needed to limit the unrecognized introduction of SARS-CoV-2 into healthcare settings by these individuals. As part of aggressive source control measures, healthcare facilities should consider implementing policies requiring everyone entering the facility to wear a cloth face covering (if tolerated) while in the building, regardless of symptoms.”7
It is our opinion, based on the CDC and IDSA recommendations, that both clinicians and patients should be required to wear masks when patients are seen in the office if possible. Many offices have instituted a policy that says, if a patient refuses to wear a mask during an office visit, then the patient will not be seen.
Eye protection
Many clinicians are uncertain about whether eye protection needs to be used when seeing asymptomatic patients. The IDSA acknowledges that there are not studies that have looked critically at eye protection, but the society also acknowledges “appropriate personal protective equipment includes, in addition to a mask or respirator, eye protection, gown and gloves.”1 In addition, the CDC recommends that, for healthcare workers located in areas with moderate or higher prevalence of COVID-19, HCPs should wear eye protection in addition to facemasks since they may encounter asymptomatic individuals with COVID-19.
Gowns and gloves
Gowns and gloves are recommended as a part of personal protective gear when caring for patients who have COVID-19. The IDSA guideline is clear in its recommendations, but does not cite evidence for having no gloves versus having gloves. Furthermore, they state that the evidence is insufficient to recommend double gloves, with the top glove used to take off a personal protective gown, and the inner glove discarded after the gown is removed. The CDC do not make recommendations for routine use of gloves in the care of patients who do not have COVID-19, even in areas where there may be asymptomatic COVID-19, and recommends standard precautions, specifically practicing hand hygiene before and after patient contact.8
The Bottom Line
When seeing patients with COVID-19, N-95 masks, goggles or face shields, gowns, and gloves should be used, with hand hygiene routinely practiced before and after seeing patients. For offices seeing patients not suspected of having COVID-19, the IDSA guideline clarifies that there is not a statistical difference in acquisition of infection with the use of surgical face masks vs N95 respirators. According to the CDC recommendations, eye protection in addition to facemasks should be used by the health care provider, and masks should be worn by patients. Hand hygiene should be used routinely before and after all patient contact. With use of these approaches, it should be safe for offices to reopen and see patients.
Neil Skolnik, MD, is professor of family and community medicine at the Thomas Jefferson University, Philadelphia, and associate director of the Family Medicine Residency Program at Abington (Pa.) Jefferson Health. Jeffrey Matthews, DO, is a second-year resident in the Family Medicine Residency at Abington Jefferson Health. For questions or comments, feel free to contact Dr. Skolnik on Twitter @NeilSkolnik.
References
1. Lynch JB, Davitkov P, Anderson DJ, et al. COVID-19 Guideline, Part 2: Infection Prevention. IDSA Home. https://www.idsociety.org/practice-guideline/covid-19-guideline-infection-prevention/. April 27, 2020. Accessed June 10, 2020.
2. J Hosp Infect. 2020 May;105(1):104-5.
3. Lancet. 2003;361(9368):1519-20.
4. Influenza Other Respir Viruses. 2020 Apr 4. doi: 2020;10.1111/irv.12745.
5. J Hosp Infect. 2010;74(3):271-7.
6. Clin Infect Dis. 2016;63(8):999-1006.
7. Centers for Disease Control and Prevention. Interim Infection Prevention and Control Recommendations for Patients with Suspected or Confirmed Coronavirus Disease 2019 (COVID-19) in Healthcare Settings. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-recommendations.html. Accessed Jun 16, 2020.
8. Centers for Disease Control and Prevention. Healthcare Infection Prevention and Control FAQs for COVID-19. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-faq.html. Accessed June 15, 2020.
As we reopen our offices we are faced with the challenge of determining the best way to do it safely – protecting ourselves, our staff, and our patients.
In this column we will focus on selected details of the recommendations from IDSA and the CDC that may be helpful in primary care offices.
Face masks
Many clinicians have asked whether a physician should use a mask while seeing patients without COVID-19 in the office, and if yes, which type. The IDSA guideline states that mask usage is imperative for reducing the risk of health care workers contracting COVID-19.1 The evidence is derived from a number of sources, including a retrospective study from Wuhan (China) University that examined two groups of health care workers during the outbreak. The first group wore N95 masks and washed their hands frequently, while the second group did not wear masks and washed their hands less frequently. In the group that took greater actions to protect themselves, none of the 493 staff members contracted COVID-19, compared with 10 of 213 staff members in the other group. The decrease in infection rate occurred in the group that wore masks despite the fact that this group had 733% more exposure to COVID-19 patients.2 Further evidence came from a case-control study done in hospitals in Hong Kong during the 2003 SARS-CoV outbreak.3 This study showed that mask wearing was the most significant intervention for reducing infection, followed by gowning, and then handwashing. These findings make it clear that mask usage is a must for all health care providers who may be caring for patients who could have COVID-19.
The guideline also reviews evidence about the use of surgical masks versus N95 masks. On reviewing indirect evidence from the SARS-CoV epidemic, IDSA found that wearing any mask – surgical or N95 – led to a large reduction in the risk of developing an infection. In this systematic review of five observational studies in health care personnel, for those wearing surgical masks, the odds ratio for developing an infection was 0.13 (95% CI, 0.03-0.62), and for those wearing N95 masks, the odds ratio was 0.12 (95% CI, 0.06-0.26). There was not a significant difference between risk reductions for those who wore surgical masks and N95 masks, respectively.1,4 The IDSA guideline panel recommended “that health care personnel caring for patients with suspected or known COVID-19 use either a surgical mask or N95 respirator ... as part of appropriate PPE.” Since there is not a significant difference in outcomes between those who use surgical masks and those who use N95 respirators, and the IDSA guideline states either type of mask is considered appropriate when taking care of patients with suspected or known COVID-19, in our opinion, use of surgical masks rather than N95s is sufficient when performing low-risk activities. Such activities include seeing patients who do not have a high likelihood of COVID-19 in the office setting.
The IDSA recommendation also discusses universal masking, defined as both patients and clinicians wearing masks. The recommendation is supported by the findings of a study in which universal mask usage was used to prevent the spread of H1N 1 during the 2009 outbreak. In this study of staff members and patients exposed to H1N1 who all wore masks, only 0.48% of 836 acquired infection. In the same study, not wearing a mask by either the provider or patient increased the risk of infection.5 Also, in a prospective study of hematopoietic stem cell transplant patients, universal masking caused infection rates to drop from 10.3% to 4.4%.6
The IDSA guideline states the following: “There may be some, albeit uncertain, benefit to universal masking in the absence of resource constraints. However, the benefits of universal masking with surgical masks should be weighed against the risk of increasing the PPE burn rate and contextualized to the background COVID-19 prevalence rate for asymptomatic or minimally symptomatic HCPs [health care providers] and visitors.”1
The CDC’s guidance statement says the following: “Continued community transmission has increased the number of individuals potentially exposed to and infectious with SARS-CoV-2. Fever and symptom screening have proven to be relatively ineffective in identifying all infected individuals, including HCPs. Symptom screening also will not identify individuals who are infected but otherwise asymptomatic or pre-symptomatic; additional interventions are needed to limit the unrecognized introduction of SARS-CoV-2 into healthcare settings by these individuals. As part of aggressive source control measures, healthcare facilities should consider implementing policies requiring everyone entering the facility to wear a cloth face covering (if tolerated) while in the building, regardless of symptoms.”7
It is our opinion, based on the CDC and IDSA recommendations, that both clinicians and patients should be required to wear masks when patients are seen in the office if possible. Many offices have instituted a policy that says, if a patient refuses to wear a mask during an office visit, then the patient will not be seen.
Eye protection
Many clinicians are uncertain about whether eye protection needs to be used when seeing asymptomatic patients. The IDSA acknowledges that there are not studies that have looked critically at eye protection, but the society also acknowledges “appropriate personal protective equipment includes, in addition to a mask or respirator, eye protection, gown and gloves.”1 In addition, the CDC recommends that, for healthcare workers located in areas with moderate or higher prevalence of COVID-19, HCPs should wear eye protection in addition to facemasks since they may encounter asymptomatic individuals with COVID-19.
Gowns and gloves
Gowns and gloves are recommended as a part of personal protective gear when caring for patients who have COVID-19. The IDSA guideline is clear in its recommendations, but does not cite evidence for having no gloves versus having gloves. Furthermore, they state that the evidence is insufficient to recommend double gloves, with the top glove used to take off a personal protective gown, and the inner glove discarded after the gown is removed. The CDC do not make recommendations for routine use of gloves in the care of patients who do not have COVID-19, even in areas where there may be asymptomatic COVID-19, and recommends standard precautions, specifically practicing hand hygiene before and after patient contact.8
The Bottom Line
When seeing patients with COVID-19, N-95 masks, goggles or face shields, gowns, and gloves should be used, with hand hygiene routinely practiced before and after seeing patients. For offices seeing patients not suspected of having COVID-19, the IDSA guideline clarifies that there is not a statistical difference in acquisition of infection with the use of surgical face masks vs N95 respirators. According to the CDC recommendations, eye protection in addition to facemasks should be used by the health care provider, and masks should be worn by patients. Hand hygiene should be used routinely before and after all patient contact. With use of these approaches, it should be safe for offices to reopen and see patients.
Neil Skolnik, MD, is professor of family and community medicine at the Thomas Jefferson University, Philadelphia, and associate director of the Family Medicine Residency Program at Abington (Pa.) Jefferson Health. Jeffrey Matthews, DO, is a second-year resident in the Family Medicine Residency at Abington Jefferson Health. For questions or comments, feel free to contact Dr. Skolnik on Twitter @NeilSkolnik.
References
1. Lynch JB, Davitkov P, Anderson DJ, et al. COVID-19 Guideline, Part 2: Infection Prevention. IDSA Home. https://www.idsociety.org/practice-guideline/covid-19-guideline-infection-prevention/. April 27, 2020. Accessed June 10, 2020.
2. J Hosp Infect. 2020 May;105(1):104-5.
3. Lancet. 2003;361(9368):1519-20.
4. Influenza Other Respir Viruses. 2020 Apr 4. doi: 2020;10.1111/irv.12745.
5. J Hosp Infect. 2010;74(3):271-7.
6. Clin Infect Dis. 2016;63(8):999-1006.
7. Centers for Disease Control and Prevention. Interim Infection Prevention and Control Recommendations for Patients with Suspected or Confirmed Coronavirus Disease 2019 (COVID-19) in Healthcare Settings. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-recommendations.html. Accessed Jun 16, 2020.
8. Centers for Disease Control and Prevention. Healthcare Infection Prevention and Control FAQs for COVID-19. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-faq.html. Accessed June 15, 2020.
ACR issues guidances for MIS-C and pediatric rheumatic disease during pandemic
Two new clinical guidance documents from the American College of Rheumatology provide evidence-based recommendations for managing pediatric rheumatic disease during the COVID-19 pandemic as well as diagnostic and treatment recommendations for multisystem inflammatory syndrome in children (MIS-C) associated with COVID-19 infection.
Although several children’s hospitals have published their treatment protocols for MIS-C since the condition’s initial discovery, the ACR appears to be the first medical organization to review all the most current evidence to issue interim guidance with the expectations that it will change as more data become available.
“It is challenging having to make recommendations not having a lot of scientific evidence, but we still felt we had to use whatever’s out there to the best of our ability and use our experience to put together these recommendations,” Dawn M. Wahezi, MD, chief of pediatric rheumatology at Children’s Hospital at Montefiore and an associate professor of pediatrics at Albert Einstein College of Medicine, New York, said in an interview.
“We wanted to be mindful of the fact that there are things we know and things we don’t know, and we have to be careful about what we’re recommending,” said Dr. Wahezi, a member of the ACR working group that assembled the recommendations for pediatric rheumatic disease management during the pandemic. “We’re recommending the best we can at this moment, but if there are new studies that come out and suggest otherwise, we will definitely have to go back and amend the document.”
The foremost priority of the pediatric rheumatic disease guidance focuses on maintaining control of the disease and avoiding flares that may put children at greater risk of infection. Dr. Wahezi said the ACR has received many calls from patients and clinicians asking whether patients should continue their immunosuppressant medications. Fear of the coronavirus infection, medication shortages, difficulty getting to the pharmacy, uneasiness about going to the clinic or hospital for infusions, and other barriers may have led to gaps in medication.
“We didn’t want people to be too quick to hold patients’ medications just because they were scared of COVID,” Dr. Wahezi said. “If they did have medication stopped for one reason or another and their disease flared, having active disease, regardless of which disease it is, actually puts you at higher risk for infection. By controlling their disease, that would be the way to protect them the most.”
A key takeaway in the guidance on MIS-C, meanwhile, is an emphasis on its rarity lest physicians be too quick to diagnose it and miss another serious condition with overlapping symptoms, explained Lauren Henderson, MD, an attending rheumatologist at Boston Children’s Hospital and assistant professor of pediatrics at Harvard Medical School, Boston. Dr. Henderson participated in the ACR group that wrote the MIS-C guidance.
“The first thing we want to be thoughtful about clinically is to recognize that children in general with the acute infectious phase of SARS-CoV-2 have mild symptoms and generally do well,” Dr. Henderson said. “From what we can tell from all the data, MIS-C is rare. That really needs to be considered when clinicians on the ground are doing the diagnostic evaluation” because of concerns that clinicians “could rush to diagnose and treat patients with MIS-C and miss important diagnoses like malignancies and infections.”
Management of pediatric rheumatic disease during the pandemic
The COVID-19 clinical guidance for managing pediatric rheumatic disease grew from the work of the North American Pediatric Rheumatology Clinical Guidance Task Force, which included seven pediatric rheumatologists, two pediatric infectious disease physicians, one adult rheumatologist, and one pediatric nurse practitioner. The general guidance covers usual preventive measures for reducing risk for COVID-19 infection, the recommendation that children continue to receive recommended vaccines unless contraindicated by medication, and routine in-person visits for ophthalmologic surveillance of those with a history of uveitis or at high risk for chronic uveitis. The guidance also notes the risk of mental health concerns, such as depression and anxiety, related to quarantine and the pandemic.
The top recommendation is initiation or continuation of all medications necessary to control underlying disease, including NSAIDs, hydroxychloroquine, ACE inhibitors/angiotensin II receptor blockers, colchicine, conventional disease-modifying antirheumatic drugs (cDMARDs), biologic DMARDs, and targeted synthetic DMARDs. Even patients who may have had exposure to COVID-19 or who have an asymptomatic COVID-19 infection should continue to take these medications with the exception of ACEi/ARBs.
In those with pediatric rheumatic disease who have a symptomatic COVID-19 infection, “NSAIDs, HCQ, and colchicine may be continued, if necessary, to control underlying disease,” as can interleukin (IL)-1 and IL-6 inhibitors, but “cDMARDs, bDMARDs [except IL-1 and IL-6 inhibitors] and tsDMARDs should be temporarily delayed or withheld,” according to the guidance. Glucocorticoids can be continued at the lowest possible dose to control disease.
“There’s nothing in the literature that suggests people who have rheumatic disease, especially children, and people who are on these medications, really are at increased risk for COVID-19,” Dr. Wahezi said. “That’s why we didn’t want people to be overcautious in stopping medications when the main priority is to control their disease.”
She noted some experts’ speculations that these medications may actually benefit patients with rheumatic disease who develop a COVID-19 infection because the medications keep the immune response in check. “If you allow them to have this dysregulated immune response and have active disease, you’re potentially putting them at greater risk,” Dr. Wahezi said, although she stressed that inadequate evidence exists to support these speculations right now.
Lack of evidence has been the biggest challenge all around with developing this guidance, she said.
“Because this is such an unprecedented situation and because people are so desperate to find treatments both for the illness and to protect those at risk for it, there are lots of people trying to put evidence out there, but it may not be the best-quality evidence,” Dr. Wahezi said.
Insufficient evidence also drove the group’s determination that “SARS-CoV-2 antibody testing is not useful in informing on the history of infection or risk of reinfection,” as the guidance states. Too much variability in the assays exist, Dr. Wahezi said, and, further, it’s unclear what the clinical significance of a positive test would be.
“We didn’t want anyone to feel they had to make clinical decisions based on the results of that antibody testing,” she said. “Even if the test is accurate, we don’t know how to interpret it because it’s so new.”
The guidance also notes that patients with stable disease and previously stable lab markers on stable doses of their medication may be able to extend the interval for medication toxicity lab testing a few months if there is concern about exposure to COVID-19 to get the blood work.
“If you’re just starting a medicine or there’s someone who’s had abnormalities with the medicine in the past or you’re making medication adjustments, you wouldn’t do it in those scenarios, but if there’s someone who’s been on the drug for a long time and are nervous to get [blood] drawn, it’s probably okay to delay it,” Dr. Wahezi said. Lab work for disease activity measures, on the other hand, remain particularly important, especially since telemedicine visits may require clinicians to rely on lab results more than previously.
Management of MIS-C associated with COVID-19
The task force that developed guidance for the new inflammatory condition recently linked to SARS-CoV-2 infections in children included nine pediatric rheumatologists, two adult rheumatologists, two pediatric cardiologists, two pediatric infectious disease specialists, and one pediatric critical care physician.
The guidance includes a figure for the diagnostic pathway in evaluating children suspected of having MIS-C and extensive detail on diagnostic work-up, but the task force intentionally avoided providing a case definition for the condition. Existing case definitions from the Centers for Disease Control and Prevention, World Health Organization, and the United Kingdom’s Royal College of Paediatrics and Child Health differ from one another and are based on unclear evidence, Dr. Henderson noted. “We really don’t have enough data to know the sensitivity and specificity of each parameter, and until that’s available, we didn’t want to add to the confusion,” she said.
The guidance also stresses that MIS-C is a rare complication, so patients suspected of having the condition who do not have “life-threatening manifestations should undergo diagnostic evaluation for MIS-C as well as other possible infectious and noninfectious etiologies before immunomodulatory treatment is initiated,” the guidance states.
Unless a child is in shock or otherwise requires urgent care, physicians should take the time to complete the diagnostic work-up while monitoring the child, Dr. Henderson said. If the child does have MIS-C, the guidance currently recommends intravenous immunoglobulin (IVIG) and/or glucocorticoids to prevent coronary artery aneurysms, the same treatment other institutions have been recommending.
“We don’t have rigorous comparative studies looking at different types of treatments,” Dr. Henderson said, noting that the vast majority of children in the literature received IVIG and/or glucocorticoid treatment. “Often children really responded quite forcefully to those treatments, but we don’t have high-quality data yet to know that this treatment is better than supportive care or another medication.”
Dr. Henderson also stressed the importance of children receiving care at a facility with the necessary expertise to manage MIS-C and receiving long-term follow-up care from a multidisciplinary clinical team that includes a rheumatologist, an infectious disease doctor, a cardiologist, and possibly a hematologist.
“Making sure children are admitted to a hospital that has the resources and are followed by physicians with expertise or understanding of the intricacies of MIS-C is really important,” she said, particularly for children with cardiac involvement. “We don’t know if all the kids presenting with left ventricular dysfunction and shock are at risk for having myocardial fibrosis down the line,” she noted. “There is so much we do not understand and very little data to guide us on what to do, so these children really need to be under the care of a cardiologist and rheumatologist to make sure that their care is tailored to them.”
Although MIS-C shares overlapping symptoms with Kawasaki disease, it’s still unclear how similar or different the two conditions are, Dr. Henderson said.
“We can definitely say that when we look at MIS-C and compare it to historical groups of Kawasaki disease before the pandemic, there are definitely different features in the MIS-C group,” she said. Kawasaki disease generally only affects children under age 5, whereas MIS-C patients run the gamut from age 1-17. Racial demographics are also different, with a higher proportion of black children affected by MIS-C.
It’s possible that the pathophysiology of both conditions will turn out to be similar, particularly given the hypothesis that Kawasaki disease is triggered by infections in genetically predisposed people. However, the severity of symptoms and risk of aneurysms appear greater with MIS-C so far.
“The degree to which these patients are presenting with left ventricular dysfunction and shock is much higher than what we’ve seen previously,” Dr. Henderson said. “Children can have aneurysms even if they don’t meet all the Kawasaki disease features, which makes it feel that this is somehow clinically different from what we’ve seen before. It’s not just the kids who have the rash and the conjunctivitis and the extremity changes and oral changes who have the aneurysms.”
The reason for including both IVIG and glucocorticoids as possible first-line drugs to prevent aneurysms is that some evidence suggests children with MIS-C may have higher levels of IVIG resistance, she said.
Like Dr. Wahezi, Dr. Henderson emphasized the necessarily transient nature of these recommendations.
“These recommendations will almost certainly change based on evolving understanding of MIS-C and the data,” Dr. Henderson said, adding that this new, unique condition highlights the importance of including children in allocating funding for research and in clinical trials.
“Children are not always identical to adults, and it’s really important that we have high-quality data to inform our decisions about how to care for them,” she said.
Dr. Wahezi had no disclosures. Dr. Henderson has consulted for Sobi and Adaptive Technologies. The guidelines did not note other disclosures for members of the ACR groups.
SOURCES: COVID-19 Clinical Guidance for Pediatric Patients with Rheumatic Disease and Clinical Guidance for Pediatric Patients with Multisystem Inflammatory Syndrome in Children (MIS-C) Associated with SARS-CoV-2 and Hyperinflammation in COVID-19
Two new clinical guidance documents from the American College of Rheumatology provide evidence-based recommendations for managing pediatric rheumatic disease during the COVID-19 pandemic as well as diagnostic and treatment recommendations for multisystem inflammatory syndrome in children (MIS-C) associated with COVID-19 infection.
Although several children’s hospitals have published their treatment protocols for MIS-C since the condition’s initial discovery, the ACR appears to be the first medical organization to review all the most current evidence to issue interim guidance with the expectations that it will change as more data become available.
“It is challenging having to make recommendations not having a lot of scientific evidence, but we still felt we had to use whatever’s out there to the best of our ability and use our experience to put together these recommendations,” Dawn M. Wahezi, MD, chief of pediatric rheumatology at Children’s Hospital at Montefiore and an associate professor of pediatrics at Albert Einstein College of Medicine, New York, said in an interview.
“We wanted to be mindful of the fact that there are things we know and things we don’t know, and we have to be careful about what we’re recommending,” said Dr. Wahezi, a member of the ACR working group that assembled the recommendations for pediatric rheumatic disease management during the pandemic. “We’re recommending the best we can at this moment, but if there are new studies that come out and suggest otherwise, we will definitely have to go back and amend the document.”
The foremost priority of the pediatric rheumatic disease guidance focuses on maintaining control of the disease and avoiding flares that may put children at greater risk of infection. Dr. Wahezi said the ACR has received many calls from patients and clinicians asking whether patients should continue their immunosuppressant medications. Fear of the coronavirus infection, medication shortages, difficulty getting to the pharmacy, uneasiness about going to the clinic or hospital for infusions, and other barriers may have led to gaps in medication.
“We didn’t want people to be too quick to hold patients’ medications just because they were scared of COVID,” Dr. Wahezi said. “If they did have medication stopped for one reason or another and their disease flared, having active disease, regardless of which disease it is, actually puts you at higher risk for infection. By controlling their disease, that would be the way to protect them the most.”
A key takeaway in the guidance on MIS-C, meanwhile, is an emphasis on its rarity lest physicians be too quick to diagnose it and miss another serious condition with overlapping symptoms, explained Lauren Henderson, MD, an attending rheumatologist at Boston Children’s Hospital and assistant professor of pediatrics at Harvard Medical School, Boston. Dr. Henderson participated in the ACR group that wrote the MIS-C guidance.
“The first thing we want to be thoughtful about clinically is to recognize that children in general with the acute infectious phase of SARS-CoV-2 have mild symptoms and generally do well,” Dr. Henderson said. “From what we can tell from all the data, MIS-C is rare. That really needs to be considered when clinicians on the ground are doing the diagnostic evaluation” because of concerns that clinicians “could rush to diagnose and treat patients with MIS-C and miss important diagnoses like malignancies and infections.”
Management of pediatric rheumatic disease during the pandemic
The COVID-19 clinical guidance for managing pediatric rheumatic disease grew from the work of the North American Pediatric Rheumatology Clinical Guidance Task Force, which included seven pediatric rheumatologists, two pediatric infectious disease physicians, one adult rheumatologist, and one pediatric nurse practitioner. The general guidance covers usual preventive measures for reducing risk for COVID-19 infection, the recommendation that children continue to receive recommended vaccines unless contraindicated by medication, and routine in-person visits for ophthalmologic surveillance of those with a history of uveitis or at high risk for chronic uveitis. The guidance also notes the risk of mental health concerns, such as depression and anxiety, related to quarantine and the pandemic.
The top recommendation is initiation or continuation of all medications necessary to control underlying disease, including NSAIDs, hydroxychloroquine, ACE inhibitors/angiotensin II receptor blockers, colchicine, conventional disease-modifying antirheumatic drugs (cDMARDs), biologic DMARDs, and targeted synthetic DMARDs. Even patients who may have had exposure to COVID-19 or who have an asymptomatic COVID-19 infection should continue to take these medications with the exception of ACEi/ARBs.
In those with pediatric rheumatic disease who have a symptomatic COVID-19 infection, “NSAIDs, HCQ, and colchicine may be continued, if necessary, to control underlying disease,” as can interleukin (IL)-1 and IL-6 inhibitors, but “cDMARDs, bDMARDs [except IL-1 and IL-6 inhibitors] and tsDMARDs should be temporarily delayed or withheld,” according to the guidance. Glucocorticoids can be continued at the lowest possible dose to control disease.
“There’s nothing in the literature that suggests people who have rheumatic disease, especially children, and people who are on these medications, really are at increased risk for COVID-19,” Dr. Wahezi said. “That’s why we didn’t want people to be overcautious in stopping medications when the main priority is to control their disease.”
She noted some experts’ speculations that these medications may actually benefit patients with rheumatic disease who develop a COVID-19 infection because the medications keep the immune response in check. “If you allow them to have this dysregulated immune response and have active disease, you’re potentially putting them at greater risk,” Dr. Wahezi said, although she stressed that inadequate evidence exists to support these speculations right now.
Lack of evidence has been the biggest challenge all around with developing this guidance, she said.
“Because this is such an unprecedented situation and because people are so desperate to find treatments both for the illness and to protect those at risk for it, there are lots of people trying to put evidence out there, but it may not be the best-quality evidence,” Dr. Wahezi said.
Insufficient evidence also drove the group’s determination that “SARS-CoV-2 antibody testing is not useful in informing on the history of infection or risk of reinfection,” as the guidance states. Too much variability in the assays exist, Dr. Wahezi said, and, further, it’s unclear what the clinical significance of a positive test would be.
“We didn’t want anyone to feel they had to make clinical decisions based on the results of that antibody testing,” she said. “Even if the test is accurate, we don’t know how to interpret it because it’s so new.”
The guidance also notes that patients with stable disease and previously stable lab markers on stable doses of their medication may be able to extend the interval for medication toxicity lab testing a few months if there is concern about exposure to COVID-19 to get the blood work.
“If you’re just starting a medicine or there’s someone who’s had abnormalities with the medicine in the past or you’re making medication adjustments, you wouldn’t do it in those scenarios, but if there’s someone who’s been on the drug for a long time and are nervous to get [blood] drawn, it’s probably okay to delay it,” Dr. Wahezi said. Lab work for disease activity measures, on the other hand, remain particularly important, especially since telemedicine visits may require clinicians to rely on lab results more than previously.
Management of MIS-C associated with COVID-19
The task force that developed guidance for the new inflammatory condition recently linked to SARS-CoV-2 infections in children included nine pediatric rheumatologists, two adult rheumatologists, two pediatric cardiologists, two pediatric infectious disease specialists, and one pediatric critical care physician.
The guidance includes a figure for the diagnostic pathway in evaluating children suspected of having MIS-C and extensive detail on diagnostic work-up, but the task force intentionally avoided providing a case definition for the condition. Existing case definitions from the Centers for Disease Control and Prevention, World Health Organization, and the United Kingdom’s Royal College of Paediatrics and Child Health differ from one another and are based on unclear evidence, Dr. Henderson noted. “We really don’t have enough data to know the sensitivity and specificity of each parameter, and until that’s available, we didn’t want to add to the confusion,” she said.
The guidance also stresses that MIS-C is a rare complication, so patients suspected of having the condition who do not have “life-threatening manifestations should undergo diagnostic evaluation for MIS-C as well as other possible infectious and noninfectious etiologies before immunomodulatory treatment is initiated,” the guidance states.
Unless a child is in shock or otherwise requires urgent care, physicians should take the time to complete the diagnostic work-up while monitoring the child, Dr. Henderson said. If the child does have MIS-C, the guidance currently recommends intravenous immunoglobulin (IVIG) and/or glucocorticoids to prevent coronary artery aneurysms, the same treatment other institutions have been recommending.
“We don’t have rigorous comparative studies looking at different types of treatments,” Dr. Henderson said, noting that the vast majority of children in the literature received IVIG and/or glucocorticoid treatment. “Often children really responded quite forcefully to those treatments, but we don’t have high-quality data yet to know that this treatment is better than supportive care or another medication.”
Dr. Henderson also stressed the importance of children receiving care at a facility with the necessary expertise to manage MIS-C and receiving long-term follow-up care from a multidisciplinary clinical team that includes a rheumatologist, an infectious disease doctor, a cardiologist, and possibly a hematologist.
“Making sure children are admitted to a hospital that has the resources and are followed by physicians with expertise or understanding of the intricacies of MIS-C is really important,” she said, particularly for children with cardiac involvement. “We don’t know if all the kids presenting with left ventricular dysfunction and shock are at risk for having myocardial fibrosis down the line,” she noted. “There is so much we do not understand and very little data to guide us on what to do, so these children really need to be under the care of a cardiologist and rheumatologist to make sure that their care is tailored to them.”
Although MIS-C shares overlapping symptoms with Kawasaki disease, it’s still unclear how similar or different the two conditions are, Dr. Henderson said.
“We can definitely say that when we look at MIS-C and compare it to historical groups of Kawasaki disease before the pandemic, there are definitely different features in the MIS-C group,” she said. Kawasaki disease generally only affects children under age 5, whereas MIS-C patients run the gamut from age 1-17. Racial demographics are also different, with a higher proportion of black children affected by MIS-C.
It’s possible that the pathophysiology of both conditions will turn out to be similar, particularly given the hypothesis that Kawasaki disease is triggered by infections in genetically predisposed people. However, the severity of symptoms and risk of aneurysms appear greater with MIS-C so far.
“The degree to which these patients are presenting with left ventricular dysfunction and shock is much higher than what we’ve seen previously,” Dr. Henderson said. “Children can have aneurysms even if they don’t meet all the Kawasaki disease features, which makes it feel that this is somehow clinically different from what we’ve seen before. It’s not just the kids who have the rash and the conjunctivitis and the extremity changes and oral changes who have the aneurysms.”
The reason for including both IVIG and glucocorticoids as possible first-line drugs to prevent aneurysms is that some evidence suggests children with MIS-C may have higher levels of IVIG resistance, she said.
Like Dr. Wahezi, Dr. Henderson emphasized the necessarily transient nature of these recommendations.
“These recommendations will almost certainly change based on evolving understanding of MIS-C and the data,” Dr. Henderson said, adding that this new, unique condition highlights the importance of including children in allocating funding for research and in clinical trials.
“Children are not always identical to adults, and it’s really important that we have high-quality data to inform our decisions about how to care for them,” she said.
Dr. Wahezi had no disclosures. Dr. Henderson has consulted for Sobi and Adaptive Technologies. The guidelines did not note other disclosures for members of the ACR groups.
SOURCES: COVID-19 Clinical Guidance for Pediatric Patients with Rheumatic Disease and Clinical Guidance for Pediatric Patients with Multisystem Inflammatory Syndrome in Children (MIS-C) Associated with SARS-CoV-2 and Hyperinflammation in COVID-19
Two new clinical guidance documents from the American College of Rheumatology provide evidence-based recommendations for managing pediatric rheumatic disease during the COVID-19 pandemic as well as diagnostic and treatment recommendations for multisystem inflammatory syndrome in children (MIS-C) associated with COVID-19 infection.
Although several children’s hospitals have published their treatment protocols for MIS-C since the condition’s initial discovery, the ACR appears to be the first medical organization to review all the most current evidence to issue interim guidance with the expectations that it will change as more data become available.
“It is challenging having to make recommendations not having a lot of scientific evidence, but we still felt we had to use whatever’s out there to the best of our ability and use our experience to put together these recommendations,” Dawn M. Wahezi, MD, chief of pediatric rheumatology at Children’s Hospital at Montefiore and an associate professor of pediatrics at Albert Einstein College of Medicine, New York, said in an interview.
“We wanted to be mindful of the fact that there are things we know and things we don’t know, and we have to be careful about what we’re recommending,” said Dr. Wahezi, a member of the ACR working group that assembled the recommendations for pediatric rheumatic disease management during the pandemic. “We’re recommending the best we can at this moment, but if there are new studies that come out and suggest otherwise, we will definitely have to go back and amend the document.”
The foremost priority of the pediatric rheumatic disease guidance focuses on maintaining control of the disease and avoiding flares that may put children at greater risk of infection. Dr. Wahezi said the ACR has received many calls from patients and clinicians asking whether patients should continue their immunosuppressant medications. Fear of the coronavirus infection, medication shortages, difficulty getting to the pharmacy, uneasiness about going to the clinic or hospital for infusions, and other barriers may have led to gaps in medication.
“We didn’t want people to be too quick to hold patients’ medications just because they were scared of COVID,” Dr. Wahezi said. “If they did have medication stopped for one reason or another and their disease flared, having active disease, regardless of which disease it is, actually puts you at higher risk for infection. By controlling their disease, that would be the way to protect them the most.”
A key takeaway in the guidance on MIS-C, meanwhile, is an emphasis on its rarity lest physicians be too quick to diagnose it and miss another serious condition with overlapping symptoms, explained Lauren Henderson, MD, an attending rheumatologist at Boston Children’s Hospital and assistant professor of pediatrics at Harvard Medical School, Boston. Dr. Henderson participated in the ACR group that wrote the MIS-C guidance.
“The first thing we want to be thoughtful about clinically is to recognize that children in general with the acute infectious phase of SARS-CoV-2 have mild symptoms and generally do well,” Dr. Henderson said. “From what we can tell from all the data, MIS-C is rare. That really needs to be considered when clinicians on the ground are doing the diagnostic evaluation” because of concerns that clinicians “could rush to diagnose and treat patients with MIS-C and miss important diagnoses like malignancies and infections.”
Management of pediatric rheumatic disease during the pandemic
The COVID-19 clinical guidance for managing pediatric rheumatic disease grew from the work of the North American Pediatric Rheumatology Clinical Guidance Task Force, which included seven pediatric rheumatologists, two pediatric infectious disease physicians, one adult rheumatologist, and one pediatric nurse practitioner. The general guidance covers usual preventive measures for reducing risk for COVID-19 infection, the recommendation that children continue to receive recommended vaccines unless contraindicated by medication, and routine in-person visits for ophthalmologic surveillance of those with a history of uveitis or at high risk for chronic uveitis. The guidance also notes the risk of mental health concerns, such as depression and anxiety, related to quarantine and the pandemic.
The top recommendation is initiation or continuation of all medications necessary to control underlying disease, including NSAIDs, hydroxychloroquine, ACE inhibitors/angiotensin II receptor blockers, colchicine, conventional disease-modifying antirheumatic drugs (cDMARDs), biologic DMARDs, and targeted synthetic DMARDs. Even patients who may have had exposure to COVID-19 or who have an asymptomatic COVID-19 infection should continue to take these medications with the exception of ACEi/ARBs.
In those with pediatric rheumatic disease who have a symptomatic COVID-19 infection, “NSAIDs, HCQ, and colchicine may be continued, if necessary, to control underlying disease,” as can interleukin (IL)-1 and IL-6 inhibitors, but “cDMARDs, bDMARDs [except IL-1 and IL-6 inhibitors] and tsDMARDs should be temporarily delayed or withheld,” according to the guidance. Glucocorticoids can be continued at the lowest possible dose to control disease.
“There’s nothing in the literature that suggests people who have rheumatic disease, especially children, and people who are on these medications, really are at increased risk for COVID-19,” Dr. Wahezi said. “That’s why we didn’t want people to be overcautious in stopping medications when the main priority is to control their disease.”
She noted some experts’ speculations that these medications may actually benefit patients with rheumatic disease who develop a COVID-19 infection because the medications keep the immune response in check. “If you allow them to have this dysregulated immune response and have active disease, you’re potentially putting them at greater risk,” Dr. Wahezi said, although she stressed that inadequate evidence exists to support these speculations right now.
Lack of evidence has been the biggest challenge all around with developing this guidance, she said.
“Because this is such an unprecedented situation and because people are so desperate to find treatments both for the illness and to protect those at risk for it, there are lots of people trying to put evidence out there, but it may not be the best-quality evidence,” Dr. Wahezi said.
Insufficient evidence also drove the group’s determination that “SARS-CoV-2 antibody testing is not useful in informing on the history of infection or risk of reinfection,” as the guidance states. Too much variability in the assays exist, Dr. Wahezi said, and, further, it’s unclear what the clinical significance of a positive test would be.
“We didn’t want anyone to feel they had to make clinical decisions based on the results of that antibody testing,” she said. “Even if the test is accurate, we don’t know how to interpret it because it’s so new.”
The guidance also notes that patients with stable disease and previously stable lab markers on stable doses of their medication may be able to extend the interval for medication toxicity lab testing a few months if there is concern about exposure to COVID-19 to get the blood work.
“If you’re just starting a medicine or there’s someone who’s had abnormalities with the medicine in the past or you’re making medication adjustments, you wouldn’t do it in those scenarios, but if there’s someone who’s been on the drug for a long time and are nervous to get [blood] drawn, it’s probably okay to delay it,” Dr. Wahezi said. Lab work for disease activity measures, on the other hand, remain particularly important, especially since telemedicine visits may require clinicians to rely on lab results more than previously.
Management of MIS-C associated with COVID-19
The task force that developed guidance for the new inflammatory condition recently linked to SARS-CoV-2 infections in children included nine pediatric rheumatologists, two adult rheumatologists, two pediatric cardiologists, two pediatric infectious disease specialists, and one pediatric critical care physician.
The guidance includes a figure for the diagnostic pathway in evaluating children suspected of having MIS-C and extensive detail on diagnostic work-up, but the task force intentionally avoided providing a case definition for the condition. Existing case definitions from the Centers for Disease Control and Prevention, World Health Organization, and the United Kingdom’s Royal College of Paediatrics and Child Health differ from one another and are based on unclear evidence, Dr. Henderson noted. “We really don’t have enough data to know the sensitivity and specificity of each parameter, and until that’s available, we didn’t want to add to the confusion,” she said.
The guidance also stresses that MIS-C is a rare complication, so patients suspected of having the condition who do not have “life-threatening manifestations should undergo diagnostic evaluation for MIS-C as well as other possible infectious and noninfectious etiologies before immunomodulatory treatment is initiated,” the guidance states.
Unless a child is in shock or otherwise requires urgent care, physicians should take the time to complete the diagnostic work-up while monitoring the child, Dr. Henderson said. If the child does have MIS-C, the guidance currently recommends intravenous immunoglobulin (IVIG) and/or glucocorticoids to prevent coronary artery aneurysms, the same treatment other institutions have been recommending.
“We don’t have rigorous comparative studies looking at different types of treatments,” Dr. Henderson said, noting that the vast majority of children in the literature received IVIG and/or glucocorticoid treatment. “Often children really responded quite forcefully to those treatments, but we don’t have high-quality data yet to know that this treatment is better than supportive care or another medication.”
Dr. Henderson also stressed the importance of children receiving care at a facility with the necessary expertise to manage MIS-C and receiving long-term follow-up care from a multidisciplinary clinical team that includes a rheumatologist, an infectious disease doctor, a cardiologist, and possibly a hematologist.
“Making sure children are admitted to a hospital that has the resources and are followed by physicians with expertise or understanding of the intricacies of MIS-C is really important,” she said, particularly for children with cardiac involvement. “We don’t know if all the kids presenting with left ventricular dysfunction and shock are at risk for having myocardial fibrosis down the line,” she noted. “There is so much we do not understand and very little data to guide us on what to do, so these children really need to be under the care of a cardiologist and rheumatologist to make sure that their care is tailored to them.”
Although MIS-C shares overlapping symptoms with Kawasaki disease, it’s still unclear how similar or different the two conditions are, Dr. Henderson said.
“We can definitely say that when we look at MIS-C and compare it to historical groups of Kawasaki disease before the pandemic, there are definitely different features in the MIS-C group,” she said. Kawasaki disease generally only affects children under age 5, whereas MIS-C patients run the gamut from age 1-17. Racial demographics are also different, with a higher proportion of black children affected by MIS-C.
It’s possible that the pathophysiology of both conditions will turn out to be similar, particularly given the hypothesis that Kawasaki disease is triggered by infections in genetically predisposed people. However, the severity of symptoms and risk of aneurysms appear greater with MIS-C so far.
“The degree to which these patients are presenting with left ventricular dysfunction and shock is much higher than what we’ve seen previously,” Dr. Henderson said. “Children can have aneurysms even if they don’t meet all the Kawasaki disease features, which makes it feel that this is somehow clinically different from what we’ve seen before. It’s not just the kids who have the rash and the conjunctivitis and the extremity changes and oral changes who have the aneurysms.”
The reason for including both IVIG and glucocorticoids as possible first-line drugs to prevent aneurysms is that some evidence suggests children with MIS-C may have higher levels of IVIG resistance, she said.
Like Dr. Wahezi, Dr. Henderson emphasized the necessarily transient nature of these recommendations.
“These recommendations will almost certainly change based on evolving understanding of MIS-C and the data,” Dr. Henderson said, adding that this new, unique condition highlights the importance of including children in allocating funding for research and in clinical trials.
“Children are not always identical to adults, and it’s really important that we have high-quality data to inform our decisions about how to care for them,” she said.
Dr. Wahezi had no disclosures. Dr. Henderson has consulted for Sobi and Adaptive Technologies. The guidelines did not note other disclosures for members of the ACR groups.
SOURCES: COVID-19 Clinical Guidance for Pediatric Patients with Rheumatic Disease and Clinical Guidance for Pediatric Patients with Multisystem Inflammatory Syndrome in Children (MIS-C) Associated with SARS-CoV-2 and Hyperinflammation in COVID-19
Cortisol levels on COVID-19 admission may be a marker of severity
Patients with COVID-19 who have high levels of the steroid hormone cortisol on admission to hospital have a substantially increased risk of dying, U.K. researchers have discovered.
Waljit S. Dhillo, MBBS, PhD, head of the division of diabetes, endocrinology and metabolism at Imperial College London, and colleagues studied 535 patients admitted to major London hospitals. Their article was published online June 18 in Lancet Diabetes & Endocrinology.
“Our analyses show for the first time that patients with COVID-19 mount a marked and appropriate acute cortisol stress response,” said Dr. Dhillo and colleagues.
Moreover, “high cortisol concentrations were associated with increased mortality and a reduced median survival, probably because this is a marker of the severity of illness.”
So measuring cortisol on admission is potentially “another simple marker to use alongside oxygen saturation levels to help us identify which patients need to be admitted immediately, and which may not,” Dr. Dhillo noted in a statement from his institution.
“Having an early indicator of which patients may deteriorate more quickly will help us with providing the best level of care as quickly as possible. In addition, we can also take cortisol levels into account when we are working out how best to treat our patients,” he said.
However, it’s important to note that this means – particularly in the wake of the RECOVERY trial reported last week – that “in the early part of the disease you don’t need steroids,” he said.
In contrast to SARS, no adrenal insufficiency with COVID-19
Cortisol levels when healthy and resting are 100-200 nmol/L and nearly zero when sleeping, the researchers explained.
They decided to examine cortisol levels because, although physiological stress from critical illness normally increases levels of the hormone, the prior coronavirus, severe acute respiratory syndrome coronavirus (SARS-CoV), had the opposite effect and induced cortisol insufficiency in some patients.
“We would have said we’re not quite sure” what effect SARS-CoV-2 is having on cortisol levels, “so that’s why we collected the data,” Dr. Dhillo said in an interview.
The researchers studied patients admitted to three large London teaching hospitals between March 9 and April 22 with a clinical suspicion of SARS-CoV-2 infection. All patients had a standard set of blood tests, including full blood count, creatinine, C-reactive protein, D-dimer, and serum cortisol.
After exclusions, the team assessed 535 patients admitted over the study period who had baseline cortisol measured within 48 hours of admission.
Of these, 403 patients were diagnosed with COVID-19 based on a positive result on real-time polymerase chain reaction testing (88%) or a strong clinical and radiological suspicion, despite a negative test (12%).
In total, 132 (25%) individuals were not diagnosed with COVID-19.
Patients with COVID-19 were a mean age of 66.3 years, and 59.6% were men.
Mean cortisol concentrations in patients with COVID-19 were significantly higher than those not diagnosed with the virus (619 vs 519 nmol/L; P < .0001).
And by May 8, significantly more patients with COVID-19 died than those without (27.8% vs 6.8%; P < .0001).
Doubling of cortisol levels associated with 40% higher mortality
Multivariate analysis taking into account age, presence of comorbidities, and laboratory tests revealed that a doubling of cortisol concentrations among those with COVID-19 was associated with a significant increase in mortality, at a hazard ratio of 1.42 (P = .014).
And patients with COVID-19 whose baseline cortisol level was >744 nmol/L had a median survival of just 15 days, compared with those with a level ≤744 nmol/L, who had a median survival of 36 days (P < .0001).
The team notes that the cortisol stress responses in their patients with COVID-19 ranged up to 3,241 nmol/L, which is “a marked cortisol stress response, perhaps higher than is observed in patients undergoing major surgery.”
Of interest, there was no interaction between cortisol levels and ethnicity in their study; a subsequent analysis of the data stratified by black, Asian, and other minority ethnicities revealed no significant differences.
The team note that their results will need to be reproduced in other populations.
“Any potential role for cortisol measurement at baseline and later during an inpatient stay with COVID-19 as a prognostic biomarker, either by itself or in combination with other biomarkers, will require validation in a prospective study.”
Implications for treatment: Reserve dexamethasone for critically ill
Dr. Dhillo explained that, because their findings indicate that people initially infected with COVID-19 do mount an appropriate stress (cortisol) response, it is important that people properly understand this in the wake of the RECOVERY trial, reported last week.
The trial showed that the widely available steroid dexamethasone significantly reduced mortality among severely ill COVID-19 patients in the intensive care unit when given at a supraphysiologic dose of 6 mg.
But it would be hazardous for anyone to self-medicate with steroids at an early stage of COVID-19 because that would further increase cortisol levels and could suppress the immune system.
“For the average person on the street with COVID-19,” excess steroids will make their symptoms worse, Dr. Dhillo explained, adding this is important to emphasize because dexamethasone, and similar steroids, “are cheap and likely available on the Internet, and so misunderstanding of the RECOVERY trial could have serious implications.”
But once patients are very sick, with “inflammation in their lungs” and are in the intensive care unit, and often on ventilators – which is a very small subgroup of those with COVID-19 – it becomes a very different story, he stressed.
“RECOVERY shows clearly there seems to be a benefit once you need oxygen or are on a ventilator, and that makes sense because [dexamethasone] is going to be an anti-inflammatory,” in this instance when the “lungs are full of water.”
“But in the early days you definitely don’t need it and it could be harmful,” he reiterated.
The study is funded by the U.K. National Institute for Health Research and Medical Research Council. The authors have reported no relevant financial relationships.
This article first appeared on Medscape.com.
Patients with COVID-19 who have high levels of the steroid hormone cortisol on admission to hospital have a substantially increased risk of dying, U.K. researchers have discovered.
Waljit S. Dhillo, MBBS, PhD, head of the division of diabetes, endocrinology and metabolism at Imperial College London, and colleagues studied 535 patients admitted to major London hospitals. Their article was published online June 18 in Lancet Diabetes & Endocrinology.
“Our analyses show for the first time that patients with COVID-19 mount a marked and appropriate acute cortisol stress response,” said Dr. Dhillo and colleagues.
Moreover, “high cortisol concentrations were associated with increased mortality and a reduced median survival, probably because this is a marker of the severity of illness.”
So measuring cortisol on admission is potentially “another simple marker to use alongside oxygen saturation levels to help us identify which patients need to be admitted immediately, and which may not,” Dr. Dhillo noted in a statement from his institution.
“Having an early indicator of which patients may deteriorate more quickly will help us with providing the best level of care as quickly as possible. In addition, we can also take cortisol levels into account when we are working out how best to treat our patients,” he said.
However, it’s important to note that this means – particularly in the wake of the RECOVERY trial reported last week – that “in the early part of the disease you don’t need steroids,” he said.
In contrast to SARS, no adrenal insufficiency with COVID-19
Cortisol levels when healthy and resting are 100-200 nmol/L and nearly zero when sleeping, the researchers explained.
They decided to examine cortisol levels because, although physiological stress from critical illness normally increases levels of the hormone, the prior coronavirus, severe acute respiratory syndrome coronavirus (SARS-CoV), had the opposite effect and induced cortisol insufficiency in some patients.
“We would have said we’re not quite sure” what effect SARS-CoV-2 is having on cortisol levels, “so that’s why we collected the data,” Dr. Dhillo said in an interview.
The researchers studied patients admitted to three large London teaching hospitals between March 9 and April 22 with a clinical suspicion of SARS-CoV-2 infection. All patients had a standard set of blood tests, including full blood count, creatinine, C-reactive protein, D-dimer, and serum cortisol.
After exclusions, the team assessed 535 patients admitted over the study period who had baseline cortisol measured within 48 hours of admission.
Of these, 403 patients were diagnosed with COVID-19 based on a positive result on real-time polymerase chain reaction testing (88%) or a strong clinical and radiological suspicion, despite a negative test (12%).
In total, 132 (25%) individuals were not diagnosed with COVID-19.
Patients with COVID-19 were a mean age of 66.3 years, and 59.6% were men.
Mean cortisol concentrations in patients with COVID-19 were significantly higher than those not diagnosed with the virus (619 vs 519 nmol/L; P < .0001).
And by May 8, significantly more patients with COVID-19 died than those without (27.8% vs 6.8%; P < .0001).
Doubling of cortisol levels associated with 40% higher mortality
Multivariate analysis taking into account age, presence of comorbidities, and laboratory tests revealed that a doubling of cortisol concentrations among those with COVID-19 was associated with a significant increase in mortality, at a hazard ratio of 1.42 (P = .014).
And patients with COVID-19 whose baseline cortisol level was >744 nmol/L had a median survival of just 15 days, compared with those with a level ≤744 nmol/L, who had a median survival of 36 days (P < .0001).
The team notes that the cortisol stress responses in their patients with COVID-19 ranged up to 3,241 nmol/L, which is “a marked cortisol stress response, perhaps higher than is observed in patients undergoing major surgery.”
Of interest, there was no interaction between cortisol levels and ethnicity in their study; a subsequent analysis of the data stratified by black, Asian, and other minority ethnicities revealed no significant differences.
The team note that their results will need to be reproduced in other populations.
“Any potential role for cortisol measurement at baseline and later during an inpatient stay with COVID-19 as a prognostic biomarker, either by itself or in combination with other biomarkers, will require validation in a prospective study.”
Implications for treatment: Reserve dexamethasone for critically ill
Dr. Dhillo explained that, because their findings indicate that people initially infected with COVID-19 do mount an appropriate stress (cortisol) response, it is important that people properly understand this in the wake of the RECOVERY trial, reported last week.
The trial showed that the widely available steroid dexamethasone significantly reduced mortality among severely ill COVID-19 patients in the intensive care unit when given at a supraphysiologic dose of 6 mg.
But it would be hazardous for anyone to self-medicate with steroids at an early stage of COVID-19 because that would further increase cortisol levels and could suppress the immune system.
“For the average person on the street with COVID-19,” excess steroids will make their symptoms worse, Dr. Dhillo explained, adding this is important to emphasize because dexamethasone, and similar steroids, “are cheap and likely available on the Internet, and so misunderstanding of the RECOVERY trial could have serious implications.”
But once patients are very sick, with “inflammation in their lungs” and are in the intensive care unit, and often on ventilators – which is a very small subgroup of those with COVID-19 – it becomes a very different story, he stressed.
“RECOVERY shows clearly there seems to be a benefit once you need oxygen or are on a ventilator, and that makes sense because [dexamethasone] is going to be an anti-inflammatory,” in this instance when the “lungs are full of water.”
“But in the early days you definitely don’t need it and it could be harmful,” he reiterated.
The study is funded by the U.K. National Institute for Health Research and Medical Research Council. The authors have reported no relevant financial relationships.
This article first appeared on Medscape.com.
Patients with COVID-19 who have high levels of the steroid hormone cortisol on admission to hospital have a substantially increased risk of dying, U.K. researchers have discovered.
Waljit S. Dhillo, MBBS, PhD, head of the division of diabetes, endocrinology and metabolism at Imperial College London, and colleagues studied 535 patients admitted to major London hospitals. Their article was published online June 18 in Lancet Diabetes & Endocrinology.
“Our analyses show for the first time that patients with COVID-19 mount a marked and appropriate acute cortisol stress response,” said Dr. Dhillo and colleagues.
Moreover, “high cortisol concentrations were associated with increased mortality and a reduced median survival, probably because this is a marker of the severity of illness.”
So measuring cortisol on admission is potentially “another simple marker to use alongside oxygen saturation levels to help us identify which patients need to be admitted immediately, and which may not,” Dr. Dhillo noted in a statement from his institution.
“Having an early indicator of which patients may deteriorate more quickly will help us with providing the best level of care as quickly as possible. In addition, we can also take cortisol levels into account when we are working out how best to treat our patients,” he said.
However, it’s important to note that this means – particularly in the wake of the RECOVERY trial reported last week – that “in the early part of the disease you don’t need steroids,” he said.
In contrast to SARS, no adrenal insufficiency with COVID-19
Cortisol levels when healthy and resting are 100-200 nmol/L and nearly zero when sleeping, the researchers explained.
They decided to examine cortisol levels because, although physiological stress from critical illness normally increases levels of the hormone, the prior coronavirus, severe acute respiratory syndrome coronavirus (SARS-CoV), had the opposite effect and induced cortisol insufficiency in some patients.
“We would have said we’re not quite sure” what effect SARS-CoV-2 is having on cortisol levels, “so that’s why we collected the data,” Dr. Dhillo said in an interview.
The researchers studied patients admitted to three large London teaching hospitals between March 9 and April 22 with a clinical suspicion of SARS-CoV-2 infection. All patients had a standard set of blood tests, including full blood count, creatinine, C-reactive protein, D-dimer, and serum cortisol.
After exclusions, the team assessed 535 patients admitted over the study period who had baseline cortisol measured within 48 hours of admission.
Of these, 403 patients were diagnosed with COVID-19 based on a positive result on real-time polymerase chain reaction testing (88%) or a strong clinical and radiological suspicion, despite a negative test (12%).
In total, 132 (25%) individuals were not diagnosed with COVID-19.
Patients with COVID-19 were a mean age of 66.3 years, and 59.6% were men.
Mean cortisol concentrations in patients with COVID-19 were significantly higher than those not diagnosed with the virus (619 vs 519 nmol/L; P < .0001).
And by May 8, significantly more patients with COVID-19 died than those without (27.8% vs 6.8%; P < .0001).
Doubling of cortisol levels associated with 40% higher mortality
Multivariate analysis taking into account age, presence of comorbidities, and laboratory tests revealed that a doubling of cortisol concentrations among those with COVID-19 was associated with a significant increase in mortality, at a hazard ratio of 1.42 (P = .014).
And patients with COVID-19 whose baseline cortisol level was >744 nmol/L had a median survival of just 15 days, compared with those with a level ≤744 nmol/L, who had a median survival of 36 days (P < .0001).
The team notes that the cortisol stress responses in their patients with COVID-19 ranged up to 3,241 nmol/L, which is “a marked cortisol stress response, perhaps higher than is observed in patients undergoing major surgery.”
Of interest, there was no interaction between cortisol levels and ethnicity in their study; a subsequent analysis of the data stratified by black, Asian, and other minority ethnicities revealed no significant differences.
The team note that their results will need to be reproduced in other populations.
“Any potential role for cortisol measurement at baseline and later during an inpatient stay with COVID-19 as a prognostic biomarker, either by itself or in combination with other biomarkers, will require validation in a prospective study.”
Implications for treatment: Reserve dexamethasone for critically ill
Dr. Dhillo explained that, because their findings indicate that people initially infected with COVID-19 do mount an appropriate stress (cortisol) response, it is important that people properly understand this in the wake of the RECOVERY trial, reported last week.
The trial showed that the widely available steroid dexamethasone significantly reduced mortality among severely ill COVID-19 patients in the intensive care unit when given at a supraphysiologic dose of 6 mg.
But it would be hazardous for anyone to self-medicate with steroids at an early stage of COVID-19 because that would further increase cortisol levels and could suppress the immune system.
“For the average person on the street with COVID-19,” excess steroids will make their symptoms worse, Dr. Dhillo explained, adding this is important to emphasize because dexamethasone, and similar steroids, “are cheap and likely available on the Internet, and so misunderstanding of the RECOVERY trial could have serious implications.”
But once patients are very sick, with “inflammation in their lungs” and are in the intensive care unit, and often on ventilators – which is a very small subgroup of those with COVID-19 – it becomes a very different story, he stressed.
“RECOVERY shows clearly there seems to be a benefit once you need oxygen or are on a ventilator, and that makes sense because [dexamethasone] is going to be an anti-inflammatory,” in this instance when the “lungs are full of water.”
“But in the early days you definitely don’t need it and it could be harmful,” he reiterated.
The study is funded by the U.K. National Institute for Health Research and Medical Research Council. The authors have reported no relevant financial relationships.
This article first appeared on Medscape.com.
Experts publish imaging recommendations for pediatric COVID-19
A team of pulmonologists has synthesized the clinical and imaging characteristics of COVID-19 in children, and has devised recommendations for ordering imaging studies in suspected cases of the infection.
The review also included useful radiographic findings to help in the differential diagnosis of COVID-19 pneumonia from other respiratory infections. Alexandra M. Foust, DO, of Boston Children’s Hospital, and colleagues reported the summary of findings and recommendations in Pediatric Pulmonology.
“Pediatricians face numerous challenges created by increasing reports of severe COVID-19 related findings in affected children,” said Mary Cataletto, MD, of NYU Langone Health in Mineola, N.Y. “[The current review] represents a multinational collaboration to provide up to date information and key imaging findings to guide chest physicians caring for children with pneumonia symptoms during the COVID-19 pandemic.”
Clinical presentation in children
In general, pediatric patients infected with the virus show milder symptoms compared with adults, and based on the limited evidence reported to date, the most common clinical symptoms of COVID-19 in children are rhinorrhea and/or nasal congestion, fever and cough with sore throat, fatigue or dyspnea, and diarrhea.
As with other viral pneumonias in children, the laboratory parameters are usually nonspecific; however, while the complete blood count (CBC) is often normal, lymphopenia, thrombocytopenia, and neutropenia have been reported in some cases of pediatric COVID-19, the authors noted.
The current Centers for Disease Control and Prevention (CDC) recommendation for initial diagnosis of SARS-CoV-2 is obtaining a nasopharyngeal swab, followed by reverse transcription polymerase chain reaction (RT-PCR) testing, they explained.
Role of imaging in diagnosis
The researchers reported that current recommendations from the American College of Radiology (ACR) do not include chest computed tomography (CT) or chest radiography (CXR) as a upfront test to diagnose pediatric COVID-19, but they may still have a role in clinical monitoring, especially in patients with a moderate to severe disease course.
The potential benefits of utilizing radiologic evaluation, such as establishing a baseline for monitoring disease progression, must be balanced with potential drawbacks, which include radiation exposure, and reduced availability of imaging resources owing to necessary cleaning and air turnover time.
Recommendations for ordering imaging studies
Based on the most recent international guidelines for pediatric COVID-19 patient management, the authors developed an algorithm for performing imaging studies in suspected cases of COVID-19 pneumonia.
The purpose of the tool is to support clinical decision-making around the utilization of CXR and CT to evaluate pediatric COVID-19 pneumonia.
“The step by step algorithm addresses the selection, sequence and timing of imaging studies with multiple images illustrating key findings of COVID-19 pneumonia in the pediatric age group,” said Dr. Cataletto. “By synthesizing the available imaging case series and guidelines, this primer provides a useful tool for the practicing pulmonologist,” she explained.
Key recommendations: CXR
“For pediatric patients with suspected or known COVID-19 infection with moderate to severe clinical symptoms requiring hospitalization (i.e., hypoxia, moderate or severe dyspnea, signs of sepsis, shock, cardiovascular compromise, altered mentation), CXR is usually indicated to establish an imaging baseline and to assess for an alternative diagnosis,” they recommended.
“Sequential CXRs may be helpful to assess pediatric patients with COVID-19 who demonstrate worsening clinical symptoms or to assess response to supportive therapy,” they wrote.
Key recommendations: CT
“Due to the increased radiation sensitivity of pediatric patients, chest CT is not recommended as an initial diagnostic test for pediatric patients with known or suspected COVID-19 pneumonia,” they explained.
The guide also included several considerations around the differential diagnosis of COVID-19 pneumonia from other pediatric lung disorders, including immune-related conditions, infectious etiologies, hematological dyscrasias, and inhalation-related lung injury.
As best practice recommendations for COVID-19 continue to evolve, the availability of practical clinical decision-making tools becomes essential to ensure optimal patient care.
No funding sources or financial disclosures were reported in the manuscript.
SOURCE: Foust AM et al. Pediatr Pulmonol. 2020 May 28. doi: 10.1002/ppul.24870.
A team of pulmonologists has synthesized the clinical and imaging characteristics of COVID-19 in children, and has devised recommendations for ordering imaging studies in suspected cases of the infection.
The review also included useful radiographic findings to help in the differential diagnosis of COVID-19 pneumonia from other respiratory infections. Alexandra M. Foust, DO, of Boston Children’s Hospital, and colleagues reported the summary of findings and recommendations in Pediatric Pulmonology.
“Pediatricians face numerous challenges created by increasing reports of severe COVID-19 related findings in affected children,” said Mary Cataletto, MD, of NYU Langone Health in Mineola, N.Y. “[The current review] represents a multinational collaboration to provide up to date information and key imaging findings to guide chest physicians caring for children with pneumonia symptoms during the COVID-19 pandemic.”
Clinical presentation in children
In general, pediatric patients infected with the virus show milder symptoms compared with adults, and based on the limited evidence reported to date, the most common clinical symptoms of COVID-19 in children are rhinorrhea and/or nasal congestion, fever and cough with sore throat, fatigue or dyspnea, and diarrhea.
As with other viral pneumonias in children, the laboratory parameters are usually nonspecific; however, while the complete blood count (CBC) is often normal, lymphopenia, thrombocytopenia, and neutropenia have been reported in some cases of pediatric COVID-19, the authors noted.
The current Centers for Disease Control and Prevention (CDC) recommendation for initial diagnosis of SARS-CoV-2 is obtaining a nasopharyngeal swab, followed by reverse transcription polymerase chain reaction (RT-PCR) testing, they explained.
Role of imaging in diagnosis
The researchers reported that current recommendations from the American College of Radiology (ACR) do not include chest computed tomography (CT) or chest radiography (CXR) as a upfront test to diagnose pediatric COVID-19, but they may still have a role in clinical monitoring, especially in patients with a moderate to severe disease course.
The potential benefits of utilizing radiologic evaluation, such as establishing a baseline for monitoring disease progression, must be balanced with potential drawbacks, which include radiation exposure, and reduced availability of imaging resources owing to necessary cleaning and air turnover time.
Recommendations for ordering imaging studies
Based on the most recent international guidelines for pediatric COVID-19 patient management, the authors developed an algorithm for performing imaging studies in suspected cases of COVID-19 pneumonia.
The purpose of the tool is to support clinical decision-making around the utilization of CXR and CT to evaluate pediatric COVID-19 pneumonia.
“The step by step algorithm addresses the selection, sequence and timing of imaging studies with multiple images illustrating key findings of COVID-19 pneumonia in the pediatric age group,” said Dr. Cataletto. “By synthesizing the available imaging case series and guidelines, this primer provides a useful tool for the practicing pulmonologist,” she explained.
Key recommendations: CXR
“For pediatric patients with suspected or known COVID-19 infection with moderate to severe clinical symptoms requiring hospitalization (i.e., hypoxia, moderate or severe dyspnea, signs of sepsis, shock, cardiovascular compromise, altered mentation), CXR is usually indicated to establish an imaging baseline and to assess for an alternative diagnosis,” they recommended.
“Sequential CXRs may be helpful to assess pediatric patients with COVID-19 who demonstrate worsening clinical symptoms or to assess response to supportive therapy,” they wrote.
Key recommendations: CT
“Due to the increased radiation sensitivity of pediatric patients, chest CT is not recommended as an initial diagnostic test for pediatric patients with known or suspected COVID-19 pneumonia,” they explained.
The guide also included several considerations around the differential diagnosis of COVID-19 pneumonia from other pediatric lung disorders, including immune-related conditions, infectious etiologies, hematological dyscrasias, and inhalation-related lung injury.
As best practice recommendations for COVID-19 continue to evolve, the availability of practical clinical decision-making tools becomes essential to ensure optimal patient care.
No funding sources or financial disclosures were reported in the manuscript.
SOURCE: Foust AM et al. Pediatr Pulmonol. 2020 May 28. doi: 10.1002/ppul.24870.
A team of pulmonologists has synthesized the clinical and imaging characteristics of COVID-19 in children, and has devised recommendations for ordering imaging studies in suspected cases of the infection.
The review also included useful radiographic findings to help in the differential diagnosis of COVID-19 pneumonia from other respiratory infections. Alexandra M. Foust, DO, of Boston Children’s Hospital, and colleagues reported the summary of findings and recommendations in Pediatric Pulmonology.
“Pediatricians face numerous challenges created by increasing reports of severe COVID-19 related findings in affected children,” said Mary Cataletto, MD, of NYU Langone Health in Mineola, N.Y. “[The current review] represents a multinational collaboration to provide up to date information and key imaging findings to guide chest physicians caring for children with pneumonia symptoms during the COVID-19 pandemic.”
Clinical presentation in children
In general, pediatric patients infected with the virus show milder symptoms compared with adults, and based on the limited evidence reported to date, the most common clinical symptoms of COVID-19 in children are rhinorrhea and/or nasal congestion, fever and cough with sore throat, fatigue or dyspnea, and diarrhea.
As with other viral pneumonias in children, the laboratory parameters are usually nonspecific; however, while the complete blood count (CBC) is often normal, lymphopenia, thrombocytopenia, and neutropenia have been reported in some cases of pediatric COVID-19, the authors noted.
The current Centers for Disease Control and Prevention (CDC) recommendation for initial diagnosis of SARS-CoV-2 is obtaining a nasopharyngeal swab, followed by reverse transcription polymerase chain reaction (RT-PCR) testing, they explained.
Role of imaging in diagnosis
The researchers reported that current recommendations from the American College of Radiology (ACR) do not include chest computed tomography (CT) or chest radiography (CXR) as a upfront test to diagnose pediatric COVID-19, but they may still have a role in clinical monitoring, especially in patients with a moderate to severe disease course.
The potential benefits of utilizing radiologic evaluation, such as establishing a baseline for monitoring disease progression, must be balanced with potential drawbacks, which include radiation exposure, and reduced availability of imaging resources owing to necessary cleaning and air turnover time.
Recommendations for ordering imaging studies
Based on the most recent international guidelines for pediatric COVID-19 patient management, the authors developed an algorithm for performing imaging studies in suspected cases of COVID-19 pneumonia.
The purpose of the tool is to support clinical decision-making around the utilization of CXR and CT to evaluate pediatric COVID-19 pneumonia.
“The step by step algorithm addresses the selection, sequence and timing of imaging studies with multiple images illustrating key findings of COVID-19 pneumonia in the pediatric age group,” said Dr. Cataletto. “By synthesizing the available imaging case series and guidelines, this primer provides a useful tool for the practicing pulmonologist,” she explained.
Key recommendations: CXR
“For pediatric patients with suspected or known COVID-19 infection with moderate to severe clinical symptoms requiring hospitalization (i.e., hypoxia, moderate or severe dyspnea, signs of sepsis, shock, cardiovascular compromise, altered mentation), CXR is usually indicated to establish an imaging baseline and to assess for an alternative diagnosis,” they recommended.
“Sequential CXRs may be helpful to assess pediatric patients with COVID-19 who demonstrate worsening clinical symptoms or to assess response to supportive therapy,” they wrote.
Key recommendations: CT
“Due to the increased radiation sensitivity of pediatric patients, chest CT is not recommended as an initial diagnostic test for pediatric patients with known or suspected COVID-19 pneumonia,” they explained.
The guide also included several considerations around the differential diagnosis of COVID-19 pneumonia from other pediatric lung disorders, including immune-related conditions, infectious etiologies, hematological dyscrasias, and inhalation-related lung injury.
As best practice recommendations for COVID-19 continue to evolve, the availability of practical clinical decision-making tools becomes essential to ensure optimal patient care.
No funding sources or financial disclosures were reported in the manuscript.
SOURCE: Foust AM et al. Pediatr Pulmonol. 2020 May 28. doi: 10.1002/ppul.24870.
FROM PEDIATRIC PULMONOLOGY
ED visits for life-threatening conditions declined early in COVID-19 pandemic
ED visits for myocardial infarction, stroke, and hyperglycemic crisis dropped substantially in the 10 weeks after COVID-19 was declared a national emergency on March 13, according to the Centers for Disease Control and Prevention.
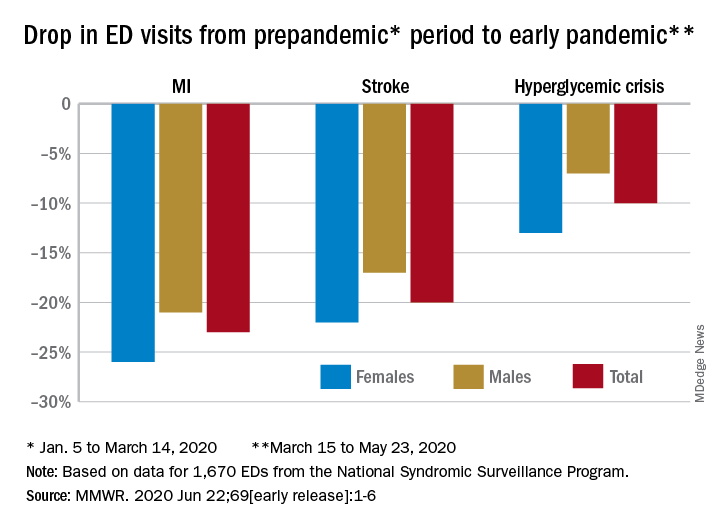
Compared with the 10-week period from Jan. 5 to March 14, ED visits were down by 23% for MI, 20% for stroke, and 10% for hyperglycemic crisis from March 15 to May 23, Samantha J. Lange, MPH, and associates at the CDC reported June 22 in the Morbidity and Mortality Weekly Report.
“A short-term decline of this magnitude … is biologically implausible for MI and stroke, especially for older adults, and unlikely for hyperglycemic crisis, and the finding suggests that patients with these conditions either could not access care or were delaying or avoiding seeking care during the early pandemic period,” they wrote.
The largest decreases in the actual number of visits for MI occurred among both men (down by 2,114, –24%) and women (down by 1,459, –25%) aged 65-74 years. For stroke, men aged 65-74 years had 1,406 (–19%) fewer visits to the ED and women 75-84 years had 1,642 (–23%) fewer visits, the CDC researchers said.
For hypoglycemic crisis, the largest declines during the early pandemic period occurred among younger adults: ED visits for men and women aged 18-44 years were down, respectively, by 419 (–8%) and 775 (–16%), they reported based on data from the National Syndromic Surveillance Program.
“Decreases in ED visits for hyperglycemic crisis might be less striking because patient recognition of this crisis is typically augmented by home glucose monitoring and not reliant upon symptoms alone, as is the case for MI and stroke,” Ms. Lange and her associates noted.
Charting weekly visit numbers showed that the drop for all three conditions actually started the week before the emergency was declared and reached its nadir the week after (March 22) for MI and 2 weeks later (March 29) for stroke and hypoglycemic crisis.
Visits for hypoglycemic crisis have largely returned to normal since those low points, but MI and stroke visits “remain below prepandemic levels” despite gradual increases through April and May, they said.
It has been reported that “deaths not associated with confirmed or probable COVID-19 might have been directly or indirectly attributed to the pandemic. The striking decline in ED visits for acute life-threatening conditions might partially explain observed excess mortality not associated with COVID-19,” the investigators wrote.
ED visits for myocardial infarction, stroke, and hyperglycemic crisis dropped substantially in the 10 weeks after COVID-19 was declared a national emergency on March 13, according to the Centers for Disease Control and Prevention.

Compared with the 10-week period from Jan. 5 to March 14, ED visits were down by 23% for MI, 20% for stroke, and 10% for hyperglycemic crisis from March 15 to May 23, Samantha J. Lange, MPH, and associates at the CDC reported June 22 in the Morbidity and Mortality Weekly Report.
“A short-term decline of this magnitude … is biologically implausible for MI and stroke, especially for older adults, and unlikely for hyperglycemic crisis, and the finding suggests that patients with these conditions either could not access care or were delaying or avoiding seeking care during the early pandemic period,” they wrote.
The largest decreases in the actual number of visits for MI occurred among both men (down by 2,114, –24%) and women (down by 1,459, –25%) aged 65-74 years. For stroke, men aged 65-74 years had 1,406 (–19%) fewer visits to the ED and women 75-84 years had 1,642 (–23%) fewer visits, the CDC researchers said.
For hypoglycemic crisis, the largest declines during the early pandemic period occurred among younger adults: ED visits for men and women aged 18-44 years were down, respectively, by 419 (–8%) and 775 (–16%), they reported based on data from the National Syndromic Surveillance Program.
“Decreases in ED visits for hyperglycemic crisis might be less striking because patient recognition of this crisis is typically augmented by home glucose monitoring and not reliant upon symptoms alone, as is the case for MI and stroke,” Ms. Lange and her associates noted.
Charting weekly visit numbers showed that the drop for all three conditions actually started the week before the emergency was declared and reached its nadir the week after (March 22) for MI and 2 weeks later (March 29) for stroke and hypoglycemic crisis.
Visits for hypoglycemic crisis have largely returned to normal since those low points, but MI and stroke visits “remain below prepandemic levels” despite gradual increases through April and May, they said.
It has been reported that “deaths not associated with confirmed or probable COVID-19 might have been directly or indirectly attributed to the pandemic. The striking decline in ED visits for acute life-threatening conditions might partially explain observed excess mortality not associated with COVID-19,” the investigators wrote.
ED visits for myocardial infarction, stroke, and hyperglycemic crisis dropped substantially in the 10 weeks after COVID-19 was declared a national emergency on March 13, according to the Centers for Disease Control and Prevention.

Compared with the 10-week period from Jan. 5 to March 14, ED visits were down by 23% for MI, 20% for stroke, and 10% for hyperglycemic crisis from March 15 to May 23, Samantha J. Lange, MPH, and associates at the CDC reported June 22 in the Morbidity and Mortality Weekly Report.
“A short-term decline of this magnitude … is biologically implausible for MI and stroke, especially for older adults, and unlikely for hyperglycemic crisis, and the finding suggests that patients with these conditions either could not access care or were delaying or avoiding seeking care during the early pandemic period,” they wrote.
The largest decreases in the actual number of visits for MI occurred among both men (down by 2,114, –24%) and women (down by 1,459, –25%) aged 65-74 years. For stroke, men aged 65-74 years had 1,406 (–19%) fewer visits to the ED and women 75-84 years had 1,642 (–23%) fewer visits, the CDC researchers said.
For hypoglycemic crisis, the largest declines during the early pandemic period occurred among younger adults: ED visits for men and women aged 18-44 years were down, respectively, by 419 (–8%) and 775 (–16%), they reported based on data from the National Syndromic Surveillance Program.
“Decreases in ED visits for hyperglycemic crisis might be less striking because patient recognition of this crisis is typically augmented by home glucose monitoring and not reliant upon symptoms alone, as is the case for MI and stroke,” Ms. Lange and her associates noted.
Charting weekly visit numbers showed that the drop for all three conditions actually started the week before the emergency was declared and reached its nadir the week after (March 22) for MI and 2 weeks later (March 29) for stroke and hypoglycemic crisis.
Visits for hypoglycemic crisis have largely returned to normal since those low points, but MI and stroke visits “remain below prepandemic levels” despite gradual increases through April and May, they said.
It has been reported that “deaths not associated with confirmed or probable COVID-19 might have been directly or indirectly attributed to the pandemic. The striking decline in ED visits for acute life-threatening conditions might partially explain observed excess mortality not associated with COVID-19,” the investigators wrote.
FROM MMWR
Daily Recap: Headache as COVID evolution predictor; psoriasis drug treats canker sores
Here are the stories our MDedge editors across specialties think you need to know about today:
Headache may predict clinical evolution of COVID-19
Headache may be a key symptom of COVID-19 that predicts the disease’s clinical evolution, new research suggests. An observational study of more than 100 patients showed that headache onset could occur during the presymptomatic or symptomatic phase of COVID-19.
Headache itself was associated with a shorter symptomatic period, while headache and anosmia were associated with a shorter hospitalization period.
It seems that those patients who start early on, during the asymptomatic or early symptomatic period of COVID-19, with headache have a more localized inflammatory response that may reflect the ability of the body to better control and respond to the infection,” lead investigator Patricia Pozo-Rosich, MD, PhD, said at the virtual annual meeting of the American Headache Society. Read more.
More tops news from the AHS meeting is available on our website.
Pilot study shows apremilast effective for severe recurrent canker sores
Apremilast was highly effective in treating patients with severe recurrent aphthous stomatitis, with rapid response and an excellent safety profile, results from a small pilot study showed.
Apremilast is approved by the FDA for psoriasis and was shown in a recent phase 2 trial to be effective for Behçet’s disease aphthosis.
Dr. Alison Bruce and colleagues found that, within 4 weeks of therapy, complete clearance of RAS lesions occurred in all patients except one in whom ulcers were reported to be less severe. Remission in all patients was sustained during 16 weeks of treatment, Dr. Bruce noted at the virtual annual meeting of the American Academy of Dermatology. Read more.
For more top news from the AAD virtual conference, visit our website.
Where does dexamethasone fit in with diabetic ketoacidosis in COVID-19?
A new article in the Journal of Clinical Endocrinology & Metabolism (JCEM) addresses unique concerns and considerations regarding diabetic ketoacidosis (DKA) in the setting of COVID-19.
“Hospitals and clinicians need to be able to quickly identify and manage DKA in COVID patients to save lives. This involves determining the options for management, including when less intensive subcutaneous insulin is indicated, and understanding how to guide patients on avoiding this serious complication,” corresponding author Marie E. McDonnell, MD, said in an Endocrine Society statement.
The new article briefly touches on the fact that upward adjustments to intensive intravenous insulin therapy for DKA may be necessary in patients with COVID-19 who are receiving concomitant corticosteroids or vasopressors. But it was written prior to the June 16 announcement of the “RECOVERY” trial results with dexamethasone. The UK National Health Service immediately approved the drug’s use in the COVID-19 setting, despite the fact that there has been no published article on the findings yet.
“The peer review will be critical. It looks as if it only benefits people who need respiratory support, but I want to understand that in much more detail,” said Dr. McDonnell. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
Headache may predict clinical evolution of COVID-19
Headache may be a key symptom of COVID-19 that predicts the disease’s clinical evolution, new research suggests. An observational study of more than 100 patients showed that headache onset could occur during the presymptomatic or symptomatic phase of COVID-19.
Headache itself was associated with a shorter symptomatic period, while headache and anosmia were associated with a shorter hospitalization period.
It seems that those patients who start early on, during the asymptomatic or early symptomatic period of COVID-19, with headache have a more localized inflammatory response that may reflect the ability of the body to better control and respond to the infection,” lead investigator Patricia Pozo-Rosich, MD, PhD, said at the virtual annual meeting of the American Headache Society. Read more.
More tops news from the AHS meeting is available on our website.
Pilot study shows apremilast effective for severe recurrent canker sores
Apremilast was highly effective in treating patients with severe recurrent aphthous stomatitis, with rapid response and an excellent safety profile, results from a small pilot study showed.
Apremilast is approved by the FDA for psoriasis and was shown in a recent phase 2 trial to be effective for Behçet’s disease aphthosis.
Dr. Alison Bruce and colleagues found that, within 4 weeks of therapy, complete clearance of RAS lesions occurred in all patients except one in whom ulcers were reported to be less severe. Remission in all patients was sustained during 16 weeks of treatment, Dr. Bruce noted at the virtual annual meeting of the American Academy of Dermatology. Read more.
For more top news from the AAD virtual conference, visit our website.
Where does dexamethasone fit in with diabetic ketoacidosis in COVID-19?
A new article in the Journal of Clinical Endocrinology & Metabolism (JCEM) addresses unique concerns and considerations regarding diabetic ketoacidosis (DKA) in the setting of COVID-19.
“Hospitals and clinicians need to be able to quickly identify and manage DKA in COVID patients to save lives. This involves determining the options for management, including when less intensive subcutaneous insulin is indicated, and understanding how to guide patients on avoiding this serious complication,” corresponding author Marie E. McDonnell, MD, said in an Endocrine Society statement.
The new article briefly touches on the fact that upward adjustments to intensive intravenous insulin therapy for DKA may be necessary in patients with COVID-19 who are receiving concomitant corticosteroids or vasopressors. But it was written prior to the June 16 announcement of the “RECOVERY” trial results with dexamethasone. The UK National Health Service immediately approved the drug’s use in the COVID-19 setting, despite the fact that there has been no published article on the findings yet.
“The peer review will be critical. It looks as if it only benefits people who need respiratory support, but I want to understand that in much more detail,” said Dr. McDonnell. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
Headache may predict clinical evolution of COVID-19
Headache may be a key symptom of COVID-19 that predicts the disease’s clinical evolution, new research suggests. An observational study of more than 100 patients showed that headache onset could occur during the presymptomatic or symptomatic phase of COVID-19.
Headache itself was associated with a shorter symptomatic period, while headache and anosmia were associated with a shorter hospitalization period.
It seems that those patients who start early on, during the asymptomatic or early symptomatic period of COVID-19, with headache have a more localized inflammatory response that may reflect the ability of the body to better control and respond to the infection,” lead investigator Patricia Pozo-Rosich, MD, PhD, said at the virtual annual meeting of the American Headache Society. Read more.
More tops news from the AHS meeting is available on our website.
Pilot study shows apremilast effective for severe recurrent canker sores
Apremilast was highly effective in treating patients with severe recurrent aphthous stomatitis, with rapid response and an excellent safety profile, results from a small pilot study showed.
Apremilast is approved by the FDA for psoriasis and was shown in a recent phase 2 trial to be effective for Behçet’s disease aphthosis.
Dr. Alison Bruce and colleagues found that, within 4 weeks of therapy, complete clearance of RAS lesions occurred in all patients except one in whom ulcers were reported to be less severe. Remission in all patients was sustained during 16 weeks of treatment, Dr. Bruce noted at the virtual annual meeting of the American Academy of Dermatology. Read more.
For more top news from the AAD virtual conference, visit our website.
Where does dexamethasone fit in with diabetic ketoacidosis in COVID-19?
A new article in the Journal of Clinical Endocrinology & Metabolism (JCEM) addresses unique concerns and considerations regarding diabetic ketoacidosis (DKA) in the setting of COVID-19.
“Hospitals and clinicians need to be able to quickly identify and manage DKA in COVID patients to save lives. This involves determining the options for management, including when less intensive subcutaneous insulin is indicated, and understanding how to guide patients on avoiding this serious complication,” corresponding author Marie E. McDonnell, MD, said in an Endocrine Society statement.
The new article briefly touches on the fact that upward adjustments to intensive intravenous insulin therapy for DKA may be necessary in patients with COVID-19 who are receiving concomitant corticosteroids or vasopressors. But it was written prior to the June 16 announcement of the “RECOVERY” trial results with dexamethasone. The UK National Health Service immediately approved the drug’s use in the COVID-19 setting, despite the fact that there has been no published article on the findings yet.
“The peer review will be critical. It looks as if it only benefits people who need respiratory support, but I want to understand that in much more detail,” said Dr. McDonnell. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
‘COVID-sorting’: How we decide whom to get close to and whom to avoid
I was recently interviewed, as a gay psychiatrist treating gay patients who lived through the AIDS epidemic, about my perspectives on living through a COVID pandemic: Were there parallels and contrasts between the two? A month later, listening to patients remotely via teletherapy, I’m experiencing an unsettling similarity to serosorting, a phenomenon that emerged during the AIDS epidemic.
Serosorting is the practice of choosing a sexual partner based on their HIV serostatus. Sorting out who was positive from who was negative allowed people to give themselves permission to have unprotected sex without risk of getting HIV. However, it was not uncommon to make those decisions without really knowing a potential partner’s actual serostatus. In fact, a lot of people serosorted by guessing.
Why not just ask a potential partner, “What’s your serostatus?” Apparently, for some, introducing the subject of HIV was deemed a sexual buzzkill. Instead, assumptions were made based on outer appearances.
Did someone look healthy? Were they well built? Were they overweight, meaning not emaciated from AIDS? If so, they were presumed negative and safe to have risky, unprotected sex with them.
Some imagined age correlated with serostatus. Since anyone older than some arbitrary age – like 30, to pull a number out of a hat – was expected to be more likely to have HIV than someone under 30, they would use that guideline in choosing sexual partners. However, these decisions were made without factual knowledge, like a blood test, but using some internal reasoning process.
Which brings us to what might be called “COVID-sorting.”
Some of my patients believe they had COVID-19, although they’d not been tested to either confirm or disprove that belief. Others had positive COVID-19 antibody tests, which they believe provides immunity. Among that group, some had symptoms, others did not.
Yet regardless of what they actually know or don’t know, patients are making calculations about managing physical distancing using their own internal formulas. They make risk calculations having little to do with actual knowledge of public health precautions on preventing COVID’s spread.
For example, one patient was planning a Memorial Day weekend in a shared Fire Island house with five friends and acquaintances. All six live alone and, as far as he knows, all are physically distancing. Consequently, my patient doesn’t think house-sharing is anything to worry about, even though he doesn’t know how scrupulously others have followed distancing guidelines.
Another patient, recovering at home after being ill with COVID-19, felt safe inviting someone over for sex who had also been ill and recovered. He didn’t think they could infect each other, presuming, not altogether unreasonably, they were both immune.
Finally, there are those who don’t know whether they had COVID-19, but think they did because they experienced influenza-like symptoms. They are giving themselves permission to meet up with others who feel the same.
Yet a Mount Sinai study, which has not yet been peer-reviewed, raises fascinating issues about immunity. The study included 719 people who suspected they had COVID-19 based on some respiratory symptoms. The majority, 62%, had no antibodies. Researchers believe they mistook influenza, another viral infection, or allergies for COVID-19 (medRxiv. 2020 May 5. doi: 10.1101/2020.04.04.2008516).
The study also included 624 people who tested positive for the virus and recovered. All but three developed antibodies. Many assume those who are antibody-positive are now immune. They may be right. However, we don’t know definitively that they are, and if they are, we do not yet know how long immunity may last. Further, as reported in the New York Times, just because you test positive for antibodies, doesn’t mean you have them.
It should be underscored that COVID-sorting is not limited to gay men or psychiatric patients. And as many states have begun opening up restrictions on social gatherings, we are seeing an all-too-human psychological mindset with wider implications – rising numbers of cases. As we move forward, all of us will have to decide for ourselves, and not only in sexual situations, how to get on with our lives in a post–COVID-19 era.
Given how much is still unknown, it is likely each of us will come up with our own algorithm of risk assessment. It is likely that the formulas used will not necessarily be based on scientific facts, although that would be ideal. If past epidemic and recent pandemic behaviors are any indicators, people’s actions will reflect some combination of their own needs and desires, their own comfort level with risk-taking, and their relative understanding of complex subjects like virology, immunology, epidemiology, and public health. The challenge faced by public health officials today is to translate complex scientific and medical issues into messages average people can understand.
What exactly can be done? I’m not exactly sure, but I hope that improved education and communication can help. In the first 2 decades of the AIDS epidemic, efforts were made to change and tailor HIV-prevention messages to specific, at-risk demographic groups. Today, public health messages aimed at preventing COVID-19’s spread that resonate with older people can fall on a younger person’s deaf ears. One message size does not fit all. Hopefully, public health officials and government leaders will act on this sooner rather than later.
Dr. Drescher, a psychoanalyst, is clinical professor of psychiatry at Columbia University, and training and supervising analyst at the William A. White Institute, both in New York. He also is emeritus editor of the Journal of Gay & Lesbian Mental Health. Dr. Drescher has no other disclosures.
I was recently interviewed, as a gay psychiatrist treating gay patients who lived through the AIDS epidemic, about my perspectives on living through a COVID pandemic: Were there parallels and contrasts between the two? A month later, listening to patients remotely via teletherapy, I’m experiencing an unsettling similarity to serosorting, a phenomenon that emerged during the AIDS epidemic.
Serosorting is the practice of choosing a sexual partner based on their HIV serostatus. Sorting out who was positive from who was negative allowed people to give themselves permission to have unprotected sex without risk of getting HIV. However, it was not uncommon to make those decisions without really knowing a potential partner’s actual serostatus. In fact, a lot of people serosorted by guessing.
Why not just ask a potential partner, “What’s your serostatus?” Apparently, for some, introducing the subject of HIV was deemed a sexual buzzkill. Instead, assumptions were made based on outer appearances.
Did someone look healthy? Were they well built? Were they overweight, meaning not emaciated from AIDS? If so, they were presumed negative and safe to have risky, unprotected sex with them.
Some imagined age correlated with serostatus. Since anyone older than some arbitrary age – like 30, to pull a number out of a hat – was expected to be more likely to have HIV than someone under 30, they would use that guideline in choosing sexual partners. However, these decisions were made without factual knowledge, like a blood test, but using some internal reasoning process.
Which brings us to what might be called “COVID-sorting.”
Some of my patients believe they had COVID-19, although they’d not been tested to either confirm or disprove that belief. Others had positive COVID-19 antibody tests, which they believe provides immunity. Among that group, some had symptoms, others did not.
Yet regardless of what they actually know or don’t know, patients are making calculations about managing physical distancing using their own internal formulas. They make risk calculations having little to do with actual knowledge of public health precautions on preventing COVID’s spread.
For example, one patient was planning a Memorial Day weekend in a shared Fire Island house with five friends and acquaintances. All six live alone and, as far as he knows, all are physically distancing. Consequently, my patient doesn’t think house-sharing is anything to worry about, even though he doesn’t know how scrupulously others have followed distancing guidelines.
Another patient, recovering at home after being ill with COVID-19, felt safe inviting someone over for sex who had also been ill and recovered. He didn’t think they could infect each other, presuming, not altogether unreasonably, they were both immune.
Finally, there are those who don’t know whether they had COVID-19, but think they did because they experienced influenza-like symptoms. They are giving themselves permission to meet up with others who feel the same.
Yet a Mount Sinai study, which has not yet been peer-reviewed, raises fascinating issues about immunity. The study included 719 people who suspected they had COVID-19 based on some respiratory symptoms. The majority, 62%, had no antibodies. Researchers believe they mistook influenza, another viral infection, or allergies for COVID-19 (medRxiv. 2020 May 5. doi: 10.1101/2020.04.04.2008516).
The study also included 624 people who tested positive for the virus and recovered. All but three developed antibodies. Many assume those who are antibody-positive are now immune. They may be right. However, we don’t know definitively that they are, and if they are, we do not yet know how long immunity may last. Further, as reported in the New York Times, just because you test positive for antibodies, doesn’t mean you have them.
It should be underscored that COVID-sorting is not limited to gay men or psychiatric patients. And as many states have begun opening up restrictions on social gatherings, we are seeing an all-too-human psychological mindset with wider implications – rising numbers of cases. As we move forward, all of us will have to decide for ourselves, and not only in sexual situations, how to get on with our lives in a post–COVID-19 era.
Given how much is still unknown, it is likely each of us will come up with our own algorithm of risk assessment. It is likely that the formulas used will not necessarily be based on scientific facts, although that would be ideal. If past epidemic and recent pandemic behaviors are any indicators, people’s actions will reflect some combination of their own needs and desires, their own comfort level with risk-taking, and their relative understanding of complex subjects like virology, immunology, epidemiology, and public health. The challenge faced by public health officials today is to translate complex scientific and medical issues into messages average people can understand.
What exactly can be done? I’m not exactly sure, but I hope that improved education and communication can help. In the first 2 decades of the AIDS epidemic, efforts were made to change and tailor HIV-prevention messages to specific, at-risk demographic groups. Today, public health messages aimed at preventing COVID-19’s spread that resonate with older people can fall on a younger person’s deaf ears. One message size does not fit all. Hopefully, public health officials and government leaders will act on this sooner rather than later.
Dr. Drescher, a psychoanalyst, is clinical professor of psychiatry at Columbia University, and training and supervising analyst at the William A. White Institute, both in New York. He also is emeritus editor of the Journal of Gay & Lesbian Mental Health. Dr. Drescher has no other disclosures.
I was recently interviewed, as a gay psychiatrist treating gay patients who lived through the AIDS epidemic, about my perspectives on living through a COVID pandemic: Were there parallels and contrasts between the two? A month later, listening to patients remotely via teletherapy, I’m experiencing an unsettling similarity to serosorting, a phenomenon that emerged during the AIDS epidemic.
Serosorting is the practice of choosing a sexual partner based on their HIV serostatus. Sorting out who was positive from who was negative allowed people to give themselves permission to have unprotected sex without risk of getting HIV. However, it was not uncommon to make those decisions without really knowing a potential partner’s actual serostatus. In fact, a lot of people serosorted by guessing.
Why not just ask a potential partner, “What’s your serostatus?” Apparently, for some, introducing the subject of HIV was deemed a sexual buzzkill. Instead, assumptions were made based on outer appearances.
Did someone look healthy? Were they well built? Were they overweight, meaning not emaciated from AIDS? If so, they were presumed negative and safe to have risky, unprotected sex with them.
Some imagined age correlated with serostatus. Since anyone older than some arbitrary age – like 30, to pull a number out of a hat – was expected to be more likely to have HIV than someone under 30, they would use that guideline in choosing sexual partners. However, these decisions were made without factual knowledge, like a blood test, but using some internal reasoning process.
Which brings us to what might be called “COVID-sorting.”
Some of my patients believe they had COVID-19, although they’d not been tested to either confirm or disprove that belief. Others had positive COVID-19 antibody tests, which they believe provides immunity. Among that group, some had symptoms, others did not.
Yet regardless of what they actually know or don’t know, patients are making calculations about managing physical distancing using their own internal formulas. They make risk calculations having little to do with actual knowledge of public health precautions on preventing COVID’s spread.
For example, one patient was planning a Memorial Day weekend in a shared Fire Island house with five friends and acquaintances. All six live alone and, as far as he knows, all are physically distancing. Consequently, my patient doesn’t think house-sharing is anything to worry about, even though he doesn’t know how scrupulously others have followed distancing guidelines.
Another patient, recovering at home after being ill with COVID-19, felt safe inviting someone over for sex who had also been ill and recovered. He didn’t think they could infect each other, presuming, not altogether unreasonably, they were both immune.
Finally, there are those who don’t know whether they had COVID-19, but think they did because they experienced influenza-like symptoms. They are giving themselves permission to meet up with others who feel the same.
Yet a Mount Sinai study, which has not yet been peer-reviewed, raises fascinating issues about immunity. The study included 719 people who suspected they had COVID-19 based on some respiratory symptoms. The majority, 62%, had no antibodies. Researchers believe they mistook influenza, another viral infection, or allergies for COVID-19 (medRxiv. 2020 May 5. doi: 10.1101/2020.04.04.2008516).
The study also included 624 people who tested positive for the virus and recovered. All but three developed antibodies. Many assume those who are antibody-positive are now immune. They may be right. However, we don’t know definitively that they are, and if they are, we do not yet know how long immunity may last. Further, as reported in the New York Times, just because you test positive for antibodies, doesn’t mean you have them.
It should be underscored that COVID-sorting is not limited to gay men or psychiatric patients. And as many states have begun opening up restrictions on social gatherings, we are seeing an all-too-human psychological mindset with wider implications – rising numbers of cases. As we move forward, all of us will have to decide for ourselves, and not only in sexual situations, how to get on with our lives in a post–COVID-19 era.
Given how much is still unknown, it is likely each of us will come up with our own algorithm of risk assessment. It is likely that the formulas used will not necessarily be based on scientific facts, although that would be ideal. If past epidemic and recent pandemic behaviors are any indicators, people’s actions will reflect some combination of their own needs and desires, their own comfort level with risk-taking, and their relative understanding of complex subjects like virology, immunology, epidemiology, and public health. The challenge faced by public health officials today is to translate complex scientific and medical issues into messages average people can understand.
What exactly can be done? I’m not exactly sure, but I hope that improved education and communication can help. In the first 2 decades of the AIDS epidemic, efforts were made to change and tailor HIV-prevention messages to specific, at-risk demographic groups. Today, public health messages aimed at preventing COVID-19’s spread that resonate with older people can fall on a younger person’s deaf ears. One message size does not fit all. Hopefully, public health officials and government leaders will act on this sooner rather than later.
Dr. Drescher, a psychoanalyst, is clinical professor of psychiatry at Columbia University, and training and supervising analyst at the William A. White Institute, both in New York. He also is emeritus editor of the Journal of Gay & Lesbian Mental Health. Dr. Drescher has no other disclosures.
‘Collateral damage’: COVID-19 threatens patients with COPD
according to a commentary published in CHEST (2020 May 28. doi: 10.1016/j.chest.2020.05.549) by a group of physicians who study COPD.
Not only is COPD among the most prevalent underlying diseases among hospitalized COVID-19 patients (Clin Microbiol Infect. 2020 Jun 8. doi: 10.1016/j.cmi.2020.05.041), but other unanticipated factors of treatment put these patients at extra risk. Valerie Press, MD, assistant professor of medicine and pediatrics at the University of Chicago, and colleagues aimed to alert physicians to be aware of potential negative effects, or collateral damage, that the pandemic can have on their patients with COPD, even those without a COVID-19 diagnosis.
These concerns include that patients may delay presenting to the ED with acute exacerbations of COPD and once they present they may be at later stages of the exacerbation. Further, evaluation for COVID-19 as a possible trigger of acute exacerbations of COPD (AECOPD) is essential; however, implementing proven AECOPD therapies remains challenging. For instance, routine therapy with corticosteroids for AECOPD may be delayed due to diagnostic uncertainty and hesitation to treat COVID-19 with steroids while COVID-19 testing is pending,” Dr. Press and her colleagues stated.
Shortages and scarcity of medications such as albuterol inhalers to treat COPD have been reported. In addition, patients with COPD are currently less likely to access their health care providers because of fear of COVID-19 infection. This barrier to care and the current higher threshold for presenting to the hospital may to lead to more cases of AECOPD and worsening health in these patients, according to the authors.
Dr. Press said in an interview: “Access to medications delivered through inhalers is challenging even without the pandemic due to high cost of medications. Generic medications are key to improving access for patients with chronic lung disease, so once the generic albuterol becomes available, this should help with access. In the meantime, some companies help provide medications at reduced cost, but usually only on a short time basis. In addition, some pharmacies have lower-cost albuterol inhalers, but these are often not supplied with a full month of dosing.”
In addition to all these concerns is the economic toll this pandemic is taking on patients. The association between COPD and socioeconomic status has been studied in depth (Am J Respir Crit Care Med. 2019; 199[8]:961-69) and would indicate that low-income patients with COPD would face an increased burden during an economic downturn. The authors noted, “Historic rapid job loss and unemployment in the U.S., coupled with a health system of employment-integrated health insurance coverage, makes it more likely that people with COPD will not be able to afford their medication.”
Dr. Press stressed that the COVID pandemic has highlighted critically important disparities in access to health care and disparities in health. “Many of the recommendations regarding stay-at-home and other safety mechanisms to prevent contracting and spreading COVID-19 have not been feasible for all sub-populations in the United States. Those that were essential workers did not have the ability to stay home. Further, those that rely on public transportation had less opportunities to social distance. Finally, while telemedicine opportunities have advanced for clinical care, not all patients have equal access to these capabilities and health disparities could widen in this regard as well. Clinicians have a responsibility to identify social determinants of health that increase risks to our patients’ health and limit their safety.”*
The authors offer some concrete suggestions of how physicians can address some of these concerns, including the following:
- Be alert to potential barriers to accessing medication and be aware of generic albuterol inhaler recently approved by the FDA in response to COVID-19–related shortages.
- Use telemedicine to monitor patients and improvement of home self-management. Clinicians should help patients “seek care with worsening symptoms and have clear management guidelines regarding seeking phone/video visits; implementing therapy with corticosteroids, antibiotics, or inhalers and nebulizers; COVID-19 testing recommendations; and thresholds for seeking emergent, urgent, or outpatient care in person,” Dr. Press added, “Building on the work of nurse advice lines and case management and other support services for high-risk patients with COPD may continue via telehealth and telephone visits.”
- Ensure that untried therapy for COVID-19 “does not displace proven and necessary treatments for patients with COPD, hence placing them at increased risk for poor outcomes.”
Dr. Press is also concerned about the post–COVID-19 period for patients with COPD. “It is too early to know if there are specific after effects of the COVID infection on patients with COPD, but given the damage the virus does to even healthy lungs, there is reason to have concern that COVID could cause worsening damage to the lungs of individuals with COPD.”
She noted, “Post-ICU [PICU] syndrome has been recognized in patients with ARDS generally, and patients who recover from critical illness may have long-lasting (and permanent) effects on strength, cognition, disability, and pulmonary function. Whether the PICU syndrome in patients with ARDS due to COVID-19 specifically is different from the PICU syndrome due to other causes remains unknown. But clinicians whose patients with COPD survive COVID-19 may expect long-lasting effects and slow recovery in cases where COVID-19 led to severe ARDS and a prolonged ICU stay. Assessment of overall patient recovery and functional capacity (beyond lung function and dyspnea symptoms) including deconditioning, anxiety, PTSD, weakness, and malnutrition will need to be addressed. Additionally, clinicians may help patients and their families understand the expected recovery and help facilitate family conversations about residual effects of COVID-19.”
The authors had no disclosures.
SOURCE: Press V et al. Chest. 2020 May 28. doi:10.1016/j.chest.2020.05.549.
CORRECTION: *This story was updated with further comments and clarifications from Dr. Press. 6/23/2020
according to a commentary published in CHEST (2020 May 28. doi: 10.1016/j.chest.2020.05.549) by a group of physicians who study COPD.
Not only is COPD among the most prevalent underlying diseases among hospitalized COVID-19 patients (Clin Microbiol Infect. 2020 Jun 8. doi: 10.1016/j.cmi.2020.05.041), but other unanticipated factors of treatment put these patients at extra risk. Valerie Press, MD, assistant professor of medicine and pediatrics at the University of Chicago, and colleagues aimed to alert physicians to be aware of potential negative effects, or collateral damage, that the pandemic can have on their patients with COPD, even those without a COVID-19 diagnosis.
These concerns include that patients may delay presenting to the ED with acute exacerbations of COPD and once they present they may be at later stages of the exacerbation. Further, evaluation for COVID-19 as a possible trigger of acute exacerbations of COPD (AECOPD) is essential; however, implementing proven AECOPD therapies remains challenging. For instance, routine therapy with corticosteroids for AECOPD may be delayed due to diagnostic uncertainty and hesitation to treat COVID-19 with steroids while COVID-19 testing is pending,” Dr. Press and her colleagues stated.
Shortages and scarcity of medications such as albuterol inhalers to treat COPD have been reported. In addition, patients with COPD are currently less likely to access their health care providers because of fear of COVID-19 infection. This barrier to care and the current higher threshold for presenting to the hospital may to lead to more cases of AECOPD and worsening health in these patients, according to the authors.
Dr. Press said in an interview: “Access to medications delivered through inhalers is challenging even without the pandemic due to high cost of medications. Generic medications are key to improving access for patients with chronic lung disease, so once the generic albuterol becomes available, this should help with access. In the meantime, some companies help provide medications at reduced cost, but usually only on a short time basis. In addition, some pharmacies have lower-cost albuterol inhalers, but these are often not supplied with a full month of dosing.”
In addition to all these concerns is the economic toll this pandemic is taking on patients. The association between COPD and socioeconomic status has been studied in depth (Am J Respir Crit Care Med. 2019; 199[8]:961-69) and would indicate that low-income patients with COPD would face an increased burden during an economic downturn. The authors noted, “Historic rapid job loss and unemployment in the U.S., coupled with a health system of employment-integrated health insurance coverage, makes it more likely that people with COPD will not be able to afford their medication.”
Dr. Press stressed that the COVID pandemic has highlighted critically important disparities in access to health care and disparities in health. “Many of the recommendations regarding stay-at-home and other safety mechanisms to prevent contracting and spreading COVID-19 have not been feasible for all sub-populations in the United States. Those that were essential workers did not have the ability to stay home. Further, those that rely on public transportation had less opportunities to social distance. Finally, while telemedicine opportunities have advanced for clinical care, not all patients have equal access to these capabilities and health disparities could widen in this regard as well. Clinicians have a responsibility to identify social determinants of health that increase risks to our patients’ health and limit their safety.”*
The authors offer some concrete suggestions of how physicians can address some of these concerns, including the following:
- Be alert to potential barriers to accessing medication and be aware of generic albuterol inhaler recently approved by the FDA in response to COVID-19–related shortages.
- Use telemedicine to monitor patients and improvement of home self-management. Clinicians should help patients “seek care with worsening symptoms and have clear management guidelines regarding seeking phone/video visits; implementing therapy with corticosteroids, antibiotics, or inhalers and nebulizers; COVID-19 testing recommendations; and thresholds for seeking emergent, urgent, or outpatient care in person,” Dr. Press added, “Building on the work of nurse advice lines and case management and other support services for high-risk patients with COPD may continue via telehealth and telephone visits.”
- Ensure that untried therapy for COVID-19 “does not displace proven and necessary treatments for patients with COPD, hence placing them at increased risk for poor outcomes.”
Dr. Press is also concerned about the post–COVID-19 period for patients with COPD. “It is too early to know if there are specific after effects of the COVID infection on patients with COPD, but given the damage the virus does to even healthy lungs, there is reason to have concern that COVID could cause worsening damage to the lungs of individuals with COPD.”
She noted, “Post-ICU [PICU] syndrome has been recognized in patients with ARDS generally, and patients who recover from critical illness may have long-lasting (and permanent) effects on strength, cognition, disability, and pulmonary function. Whether the PICU syndrome in patients with ARDS due to COVID-19 specifically is different from the PICU syndrome due to other causes remains unknown. But clinicians whose patients with COPD survive COVID-19 may expect long-lasting effects and slow recovery in cases where COVID-19 led to severe ARDS and a prolonged ICU stay. Assessment of overall patient recovery and functional capacity (beyond lung function and dyspnea symptoms) including deconditioning, anxiety, PTSD, weakness, and malnutrition will need to be addressed. Additionally, clinicians may help patients and their families understand the expected recovery and help facilitate family conversations about residual effects of COVID-19.”
The authors had no disclosures.
SOURCE: Press V et al. Chest. 2020 May 28. doi:10.1016/j.chest.2020.05.549.
CORRECTION: *This story was updated with further comments and clarifications from Dr. Press. 6/23/2020
according to a commentary published in CHEST (2020 May 28. doi: 10.1016/j.chest.2020.05.549) by a group of physicians who study COPD.
Not only is COPD among the most prevalent underlying diseases among hospitalized COVID-19 patients (Clin Microbiol Infect. 2020 Jun 8. doi: 10.1016/j.cmi.2020.05.041), but other unanticipated factors of treatment put these patients at extra risk. Valerie Press, MD, assistant professor of medicine and pediatrics at the University of Chicago, and colleagues aimed to alert physicians to be aware of potential negative effects, or collateral damage, that the pandemic can have on their patients with COPD, even those without a COVID-19 diagnosis.
These concerns include that patients may delay presenting to the ED with acute exacerbations of COPD and once they present they may be at later stages of the exacerbation. Further, evaluation for COVID-19 as a possible trigger of acute exacerbations of COPD (AECOPD) is essential; however, implementing proven AECOPD therapies remains challenging. For instance, routine therapy with corticosteroids for AECOPD may be delayed due to diagnostic uncertainty and hesitation to treat COVID-19 with steroids while COVID-19 testing is pending,” Dr. Press and her colleagues stated.
Shortages and scarcity of medications such as albuterol inhalers to treat COPD have been reported. In addition, patients with COPD are currently less likely to access their health care providers because of fear of COVID-19 infection. This barrier to care and the current higher threshold for presenting to the hospital may to lead to more cases of AECOPD and worsening health in these patients, according to the authors.
Dr. Press said in an interview: “Access to medications delivered through inhalers is challenging even without the pandemic due to high cost of medications. Generic medications are key to improving access for patients with chronic lung disease, so once the generic albuterol becomes available, this should help with access. In the meantime, some companies help provide medications at reduced cost, but usually only on a short time basis. In addition, some pharmacies have lower-cost albuterol inhalers, but these are often not supplied with a full month of dosing.”
In addition to all these concerns is the economic toll this pandemic is taking on patients. The association between COPD and socioeconomic status has been studied in depth (Am J Respir Crit Care Med. 2019; 199[8]:961-69) and would indicate that low-income patients with COPD would face an increased burden during an economic downturn. The authors noted, “Historic rapid job loss and unemployment in the U.S., coupled with a health system of employment-integrated health insurance coverage, makes it more likely that people with COPD will not be able to afford their medication.”
Dr. Press stressed that the COVID pandemic has highlighted critically important disparities in access to health care and disparities in health. “Many of the recommendations regarding stay-at-home and other safety mechanisms to prevent contracting and spreading COVID-19 have not been feasible for all sub-populations in the United States. Those that were essential workers did not have the ability to stay home. Further, those that rely on public transportation had less opportunities to social distance. Finally, while telemedicine opportunities have advanced for clinical care, not all patients have equal access to these capabilities and health disparities could widen in this regard as well. Clinicians have a responsibility to identify social determinants of health that increase risks to our patients’ health and limit their safety.”*
The authors offer some concrete suggestions of how physicians can address some of these concerns, including the following:
- Be alert to potential barriers to accessing medication and be aware of generic albuterol inhaler recently approved by the FDA in response to COVID-19–related shortages.
- Use telemedicine to monitor patients and improvement of home self-management. Clinicians should help patients “seek care with worsening symptoms and have clear management guidelines regarding seeking phone/video visits; implementing therapy with corticosteroids, antibiotics, or inhalers and nebulizers; COVID-19 testing recommendations; and thresholds for seeking emergent, urgent, or outpatient care in person,” Dr. Press added, “Building on the work of nurse advice lines and case management and other support services for high-risk patients with COPD may continue via telehealth and telephone visits.”
- Ensure that untried therapy for COVID-19 “does not displace proven and necessary treatments for patients with COPD, hence placing them at increased risk for poor outcomes.”
Dr. Press is also concerned about the post–COVID-19 period for patients with COPD. “It is too early to know if there are specific after effects of the COVID infection on patients with COPD, but given the damage the virus does to even healthy lungs, there is reason to have concern that COVID could cause worsening damage to the lungs of individuals with COPD.”
She noted, “Post-ICU [PICU] syndrome has been recognized in patients with ARDS generally, and patients who recover from critical illness may have long-lasting (and permanent) effects on strength, cognition, disability, and pulmonary function. Whether the PICU syndrome in patients with ARDS due to COVID-19 specifically is different from the PICU syndrome due to other causes remains unknown. But clinicians whose patients with COPD survive COVID-19 may expect long-lasting effects and slow recovery in cases where COVID-19 led to severe ARDS and a prolonged ICU stay. Assessment of overall patient recovery and functional capacity (beyond lung function and dyspnea symptoms) including deconditioning, anxiety, PTSD, weakness, and malnutrition will need to be addressed. Additionally, clinicians may help patients and their families understand the expected recovery and help facilitate family conversations about residual effects of COVID-19.”
The authors had no disclosures.
SOURCE: Press V et al. Chest. 2020 May 28. doi:10.1016/j.chest.2020.05.549.
CORRECTION: *This story was updated with further comments and clarifications from Dr. Press. 6/23/2020
FROM CHEST












