User login
Social distancing impacts other infectious diseases
Diagnoses of 12 common pediatric infectious diseases in a large pediatric primary care network declined significantly in the weeks after COVID-19 social distancing (SD) was enacted in Massachusetts, compared with the same time period in 2019, an analysis of EHR data has shown.
While declines in infectious disease transmission with SD are not surprising, “these data demonstrate the extent to which transmission of common pediatric infections can be altered when close contact with other children is eliminated,” Jonathan Hatoun, MD, MPH of the Pediatric Physicians’ Organization at Children’s in Brookline, Mass., and coauthors wrote in Pediatrics . “Notably, three of the studied diseases, namely, influenza, croup, and bronchiolitis, essentially disappeared with [social distancing].”
The researchers analyzed the weekly incidence of each diagnosis for similar calendar periods in 2019 and 2020. A pre-SD period was defined as week 1-9, starting on Jan. 1, and a post-SD period was defined as week 13-18. (The several-week gap represented an implementation period as social distancing was enacted in the state earlier in 2020, from a declared statewide state of emergency through school closures and stay-at-home advisories.)
To isolate the effect of widespread SD, they performed a “difference-in-differences regression analysis, with diagnosis count as a function of calendar year, time period (pre-SD versus post-SD) and the interaction between the two.” The Massachusetts pediatric network provides care for approximately 375,000 children in 100 locations around the state.
In their research brief, Dr. Hatoun and coauthors presented weekly rates expressed as diagnoses per 100,000 patients per day. The rate of bronchiolitis, for instance, was 18 and 8 in the pre- and post-SD–equivalent weeks of 2019, respectively, and 20 and 0.6 in the pre- and post-SD weeks of 2020. Their analysis showed the rate in the 2020 post-SD period to be 10 diagnoses per 100,000 patients per day lower than they would have expected based on the 2019 trend.
Rates of pneumonia, acute otitis media, and streptococcal pharyngitis were similarly 14, 85, and 31 diagnoses per 100,000 patients per day lower, respectively. The prevalence of each of the other conditions analyzed – the common cold, croup, gastroenteritis, nonstreptococcal pharyngitis, sinusitis, skin and soft tissue infections, and urinary tract infection (UTI) – also was significantly lower in the 2020 post-SD period than would be expected based on 2019 data (P < .001 for all diagnoses).
Putting things in perspective
“This study puts numbers to the sense that we have all had in pediatrics – that social distancing appears to have had a dramatic impact on the transmission of common childhood infectious diseases, especially other respiratory viral pathogens,” Audrey R. John, MD, PhD, chief of the division of pediatric infectious disease at Children’s Hospital of Philadelphia, said in an interview.
The authors acknowledged the possible role of families not seeking care, but said that a smaller decrease in diagnoses of UTI – generally not a contagious disease – “suggests that changes in care-seeking behavior had a relatively modest effect on the other observed declines.” (The rate of UTI for the pre- and post-SD periods was 3.3 and 3.7 per 100,000 patients per day in 2019, and 3.4 and 2.4 in 2020, for a difference in differences of –1.5).
In an accompanying editorial, David W. Kimberlin, MD and Erica C. Bjornstad, MD, PhD, MPH, of the University of Alabama at Birmingham, called the report “provocative” and wrote that similar observations of infections dropping during periods of isolation – namely, dramatic declines in influenza and other respiratory viruses in Seattle after a record snowstorm in 2019 – combined with findings from other modeling studies “suggest that the decline [reported in Boston] is indeed real” (Pediatrics 2020. doi: 10.1542/peds.2020-019232).
However, “we also now know that immunization rates for American children have plummeted since the onset of the SARS-CoV-2 pandemic [because of a] ... dramatic decrease in the use of health care during the first months of the pandemic,” they wrote. “Viewed through this lens,” the declines reported in Boston may reflect inflections going “undiagnosed and untreated.”
Ultimately, Dr. Kimberlin and Dr. Bjornstad said, “the verdict remains out.”
Dr. John said that she and others are “concerned about children not seeking care in a timely manner, and [concerned] that reductions in reported infections might be due to a lack of recognition rather than a lack of transmission.”
In Philadelphia, however, declines in admissions for asthma exacerbations, “which are often caused by respiratory viral infections, suggests that this may not be the case,” said Dr. John, who was asked to comment on the study.
In addition, she said, the Massachusetts data showing that UTI diagnoses “are nearly as common this year as in 2019” are “reassuring.”
Are there lessons for the future?
Coauthor Louis Vernacchio, MD, MSc, chief medical officer of the Pediatric Physicians’ Organization at Children’s network, said in an interview that beyond the pandemic, it’s likely that “more careful attention to proven infection control practices in daycares and schools could reduce the burden of common infectious diseases in children.”
Dr. John similarly sees a long-term value of quantifying the impact of social distancing. “We’ve always known [for instance] that bronchiolitis is the result of viral infection.” Findings like the Massachusetts data “will help us advise families who might be trying to protect their premature infants (at risk for severe bronchiolitis) through social distancing.”
The analysis covered both in-person and telemedicine encounters occurring on weekdays.
The authors of the research brief indicated they have no relevant financial disclosures and there was no external funding. The authors of the commentary also reported they have no relevant financial disclosures, and Dr. John said she had no relevant financial disclosures.
SOURCE: Hatoun J et al. Pediatrics. 2020. doi: 10.1542/peds.2020-006460.
Diagnoses of 12 common pediatric infectious diseases in a large pediatric primary care network declined significantly in the weeks after COVID-19 social distancing (SD) was enacted in Massachusetts, compared with the same time period in 2019, an analysis of EHR data has shown.
While declines in infectious disease transmission with SD are not surprising, “these data demonstrate the extent to which transmission of common pediatric infections can be altered when close contact with other children is eliminated,” Jonathan Hatoun, MD, MPH of the Pediatric Physicians’ Organization at Children’s in Brookline, Mass., and coauthors wrote in Pediatrics . “Notably, three of the studied diseases, namely, influenza, croup, and bronchiolitis, essentially disappeared with [social distancing].”
The researchers analyzed the weekly incidence of each diagnosis for similar calendar periods in 2019 and 2020. A pre-SD period was defined as week 1-9, starting on Jan. 1, and a post-SD period was defined as week 13-18. (The several-week gap represented an implementation period as social distancing was enacted in the state earlier in 2020, from a declared statewide state of emergency through school closures and stay-at-home advisories.)
To isolate the effect of widespread SD, they performed a “difference-in-differences regression analysis, with diagnosis count as a function of calendar year, time period (pre-SD versus post-SD) and the interaction between the two.” The Massachusetts pediatric network provides care for approximately 375,000 children in 100 locations around the state.
In their research brief, Dr. Hatoun and coauthors presented weekly rates expressed as diagnoses per 100,000 patients per day. The rate of bronchiolitis, for instance, was 18 and 8 in the pre- and post-SD–equivalent weeks of 2019, respectively, and 20 and 0.6 in the pre- and post-SD weeks of 2020. Their analysis showed the rate in the 2020 post-SD period to be 10 diagnoses per 100,000 patients per day lower than they would have expected based on the 2019 trend.
Rates of pneumonia, acute otitis media, and streptococcal pharyngitis were similarly 14, 85, and 31 diagnoses per 100,000 patients per day lower, respectively. The prevalence of each of the other conditions analyzed – the common cold, croup, gastroenteritis, nonstreptococcal pharyngitis, sinusitis, skin and soft tissue infections, and urinary tract infection (UTI) – also was significantly lower in the 2020 post-SD period than would be expected based on 2019 data (P < .001 for all diagnoses).
Putting things in perspective
“This study puts numbers to the sense that we have all had in pediatrics – that social distancing appears to have had a dramatic impact on the transmission of common childhood infectious diseases, especially other respiratory viral pathogens,” Audrey R. John, MD, PhD, chief of the division of pediatric infectious disease at Children’s Hospital of Philadelphia, said in an interview.
The authors acknowledged the possible role of families not seeking care, but said that a smaller decrease in diagnoses of UTI – generally not a contagious disease – “suggests that changes in care-seeking behavior had a relatively modest effect on the other observed declines.” (The rate of UTI for the pre- and post-SD periods was 3.3 and 3.7 per 100,000 patients per day in 2019, and 3.4 and 2.4 in 2020, for a difference in differences of –1.5).
In an accompanying editorial, David W. Kimberlin, MD and Erica C. Bjornstad, MD, PhD, MPH, of the University of Alabama at Birmingham, called the report “provocative” and wrote that similar observations of infections dropping during periods of isolation – namely, dramatic declines in influenza and other respiratory viruses in Seattle after a record snowstorm in 2019 – combined with findings from other modeling studies “suggest that the decline [reported in Boston] is indeed real” (Pediatrics 2020. doi: 10.1542/peds.2020-019232).
However, “we also now know that immunization rates for American children have plummeted since the onset of the SARS-CoV-2 pandemic [because of a] ... dramatic decrease in the use of health care during the first months of the pandemic,” they wrote. “Viewed through this lens,” the declines reported in Boston may reflect inflections going “undiagnosed and untreated.”
Ultimately, Dr. Kimberlin and Dr. Bjornstad said, “the verdict remains out.”
Dr. John said that she and others are “concerned about children not seeking care in a timely manner, and [concerned] that reductions in reported infections might be due to a lack of recognition rather than a lack of transmission.”
In Philadelphia, however, declines in admissions for asthma exacerbations, “which are often caused by respiratory viral infections, suggests that this may not be the case,” said Dr. John, who was asked to comment on the study.
In addition, she said, the Massachusetts data showing that UTI diagnoses “are nearly as common this year as in 2019” are “reassuring.”
Are there lessons for the future?
Coauthor Louis Vernacchio, MD, MSc, chief medical officer of the Pediatric Physicians’ Organization at Children’s network, said in an interview that beyond the pandemic, it’s likely that “more careful attention to proven infection control practices in daycares and schools could reduce the burden of common infectious diseases in children.”
Dr. John similarly sees a long-term value of quantifying the impact of social distancing. “We’ve always known [for instance] that bronchiolitis is the result of viral infection.” Findings like the Massachusetts data “will help us advise families who might be trying to protect their premature infants (at risk for severe bronchiolitis) through social distancing.”
The analysis covered both in-person and telemedicine encounters occurring on weekdays.
The authors of the research brief indicated they have no relevant financial disclosures and there was no external funding. The authors of the commentary also reported they have no relevant financial disclosures, and Dr. John said she had no relevant financial disclosures.
SOURCE: Hatoun J et al. Pediatrics. 2020. doi: 10.1542/peds.2020-006460.
Diagnoses of 12 common pediatric infectious diseases in a large pediatric primary care network declined significantly in the weeks after COVID-19 social distancing (SD) was enacted in Massachusetts, compared with the same time period in 2019, an analysis of EHR data has shown.
While declines in infectious disease transmission with SD are not surprising, “these data demonstrate the extent to which transmission of common pediatric infections can be altered when close contact with other children is eliminated,” Jonathan Hatoun, MD, MPH of the Pediatric Physicians’ Organization at Children’s in Brookline, Mass., and coauthors wrote in Pediatrics . “Notably, three of the studied diseases, namely, influenza, croup, and bronchiolitis, essentially disappeared with [social distancing].”
The researchers analyzed the weekly incidence of each diagnosis for similar calendar periods in 2019 and 2020. A pre-SD period was defined as week 1-9, starting on Jan. 1, and a post-SD period was defined as week 13-18. (The several-week gap represented an implementation period as social distancing was enacted in the state earlier in 2020, from a declared statewide state of emergency through school closures and stay-at-home advisories.)
To isolate the effect of widespread SD, they performed a “difference-in-differences regression analysis, with diagnosis count as a function of calendar year, time period (pre-SD versus post-SD) and the interaction between the two.” The Massachusetts pediatric network provides care for approximately 375,000 children in 100 locations around the state.
In their research brief, Dr. Hatoun and coauthors presented weekly rates expressed as diagnoses per 100,000 patients per day. The rate of bronchiolitis, for instance, was 18 and 8 in the pre- and post-SD–equivalent weeks of 2019, respectively, and 20 and 0.6 in the pre- and post-SD weeks of 2020. Their analysis showed the rate in the 2020 post-SD period to be 10 diagnoses per 100,000 patients per day lower than they would have expected based on the 2019 trend.
Rates of pneumonia, acute otitis media, and streptococcal pharyngitis were similarly 14, 85, and 31 diagnoses per 100,000 patients per day lower, respectively. The prevalence of each of the other conditions analyzed – the common cold, croup, gastroenteritis, nonstreptococcal pharyngitis, sinusitis, skin and soft tissue infections, and urinary tract infection (UTI) – also was significantly lower in the 2020 post-SD period than would be expected based on 2019 data (P < .001 for all diagnoses).
Putting things in perspective
“This study puts numbers to the sense that we have all had in pediatrics – that social distancing appears to have had a dramatic impact on the transmission of common childhood infectious diseases, especially other respiratory viral pathogens,” Audrey R. John, MD, PhD, chief of the division of pediatric infectious disease at Children’s Hospital of Philadelphia, said in an interview.
The authors acknowledged the possible role of families not seeking care, but said that a smaller decrease in diagnoses of UTI – generally not a contagious disease – “suggests that changes in care-seeking behavior had a relatively modest effect on the other observed declines.” (The rate of UTI for the pre- and post-SD periods was 3.3 and 3.7 per 100,000 patients per day in 2019, and 3.4 and 2.4 in 2020, for a difference in differences of –1.5).
In an accompanying editorial, David W. Kimberlin, MD and Erica C. Bjornstad, MD, PhD, MPH, of the University of Alabama at Birmingham, called the report “provocative” and wrote that similar observations of infections dropping during periods of isolation – namely, dramatic declines in influenza and other respiratory viruses in Seattle after a record snowstorm in 2019 – combined with findings from other modeling studies “suggest that the decline [reported in Boston] is indeed real” (Pediatrics 2020. doi: 10.1542/peds.2020-019232).
However, “we also now know that immunization rates for American children have plummeted since the onset of the SARS-CoV-2 pandemic [because of a] ... dramatic decrease in the use of health care during the first months of the pandemic,” they wrote. “Viewed through this lens,” the declines reported in Boston may reflect inflections going “undiagnosed and untreated.”
Ultimately, Dr. Kimberlin and Dr. Bjornstad said, “the verdict remains out.”
Dr. John said that she and others are “concerned about children not seeking care in a timely manner, and [concerned] that reductions in reported infections might be due to a lack of recognition rather than a lack of transmission.”
In Philadelphia, however, declines in admissions for asthma exacerbations, “which are often caused by respiratory viral infections, suggests that this may not be the case,” said Dr. John, who was asked to comment on the study.
In addition, she said, the Massachusetts data showing that UTI diagnoses “are nearly as common this year as in 2019” are “reassuring.”
Are there lessons for the future?
Coauthor Louis Vernacchio, MD, MSc, chief medical officer of the Pediatric Physicians’ Organization at Children’s network, said in an interview that beyond the pandemic, it’s likely that “more careful attention to proven infection control practices in daycares and schools could reduce the burden of common infectious diseases in children.”
Dr. John similarly sees a long-term value of quantifying the impact of social distancing. “We’ve always known [for instance] that bronchiolitis is the result of viral infection.” Findings like the Massachusetts data “will help us advise families who might be trying to protect their premature infants (at risk for severe bronchiolitis) through social distancing.”
The analysis covered both in-person and telemedicine encounters occurring on weekdays.
The authors of the research brief indicated they have no relevant financial disclosures and there was no external funding. The authors of the commentary also reported they have no relevant financial disclosures, and Dr. John said she had no relevant financial disclosures.
SOURCE: Hatoun J et al. Pediatrics. 2020. doi: 10.1542/peds.2020-006460.
FROM PEDIATRICS
COVID-SAFE: Strategies for safeguarding your outpatient clinical practice against COVID-19
No question, the COVID-19 pandemic has been a challenging time for medical practices across the United States. Uncertainty remains regarding bringing patients and services back into our offices. One factor that distinguishes many ObGyn practices from other specialties is that our practices have remained open—in some form—since the beginning of the pandemic. In various parts of the country, gynecologic surgeries and routine office visits have been significantly reduced; however, deliveries and gynecologic emergencies have continued.
In this article, I suggest a framework of strategies and resources to provide insight for outpatient operations. Individual practices will vary across the nation depending on local conditions. Full practice capacity may take on a different look than it had prior to the pandemic, and there is opportunity to change the way we operate.
Strategy 1: Consult regulatory requirements frequently
As the local status of COVID-19 evolves quickly, it is essential to examine the frequently updated recommendations from regulatory agencies at the federal, state, and local levels. Clinical practices that function within health systems need to demonstrate alignment with hospital or university policies and procedures. The Centers for Disease Control and Prevention (CDC), Occupational Safety and Health Administration (OSHA), Centers for Medicare and Medicaid Services (CMS), and individual state departments of health provide dynamic resources that are easily accessible online.1-3
The American College of Obstetricians and Gynecologists (ACOG) continues to be an excellent medical society resource.4 Subspecialty organizations that provide up-to-date guidance include the Society for Maternal-Fetal Medicine (SMFM), Society of Gynecologic Surgeons (SGS), AAGL (American Association of Gynecologic Laparoscopists), American Society for Reproductive Medicine (ASRM), and Society of Gynecologic Oncology (SGO).5-9 These resources are updated as more information about COVID-19 emerges, and they may be modified to different local-regional conditions.
The professional liability insurance carrier is an important source of insight for a number of circumstances, including modifications to your office practice, such as returning to full-scope or part-time practice; operating outside normal clinical service arrangements (for example, assisting with emergency care); offering telehealth services; and adding extra hours or employees to accommodate the patient backlog. Business insurance coverage is a separate issue to consider. Reviewing the practice policy may protect your business from COVID-related liabilities.
Consulting with legal counselors can be helpful. They can assist with navigating various practice and personnel COVID-related changes, as well as developing a viable plan for patients who were previously insured pre–COVID-19 who are currently uninsured.
Continue to: Strategy 2: Reimagine schedule capacity...
Strategy 2: Reimagine schedule capacity
The waxing and waning of the COVID-19 crisis presents an opportunity to evaluate our office practices and make necessary and positive changes. The question becomes, do we operate our practices as usual or do we rethink our strategy for seeing patients and integrate lessons learned from the pandemic? Patients are deciding when they are comfortable to schedule elective surgeries and routine office encounters. This gives us the chance to break from the tradition of 100% in-person visits and change the way we care for women.
The coronavirus has accelerated the rise of telehealth/telemedicine and is, perhaps, a silver lining of the pandemic. Telehealth is a valuable tool for accessing health services when in-person visits are not possible. Evaluating and triaging patients for in-person versus telehealth visits is now a viable option for clinical practice and reduces exposure to COVID-19 infection.
Telemedicine is convenient, and clinicians can use it to counsel and screen for various health issues as well as to extend their reach to rural communities. Appropriate consent should be documented in the patient chart. As some areas continue to be without adequate access to WiFi, telephone contact also is currently acceptable. Telehealth does not replace the in-person visit but can be viewed as a complementary and supplementary service.
Consider a balance between telehealth and in-person visits by evaluating which visits can continue remotely and which can alternate with in-person visits. This offers tremendous flexibility and will expand delivery of essential health care to patients.10 Integrating telemedicine into clinical practice provides an additional benefit: It minimizes the exposure and transmission of COVID-19 to health care workers and patients and preserves supplies, including personal protective equipment (PPE).
Prioritize the backlog of patients who require follow-up testing, procedures, and surgeries. Communicate with patients that it is safe to be seen and important to not avoid routine and preventative visits that might reveal concerns or conditions that require treatment.
Strategy 3: Institute infection prevention and control measures
The importance of instituting and ensuring safety measures for office personnel and patients cannot be underestimated. Recently, a study from King’s College in London found that frontline health care workers with PPE still have 3 to 4 times the risk of contracting coronavirus compared with the general public.11 Health care systems should ensure adequate PPE availability and develop additional strategies to protect health care workers from COVID-19. We have to be fanatical about cleanliness and PPE. We have to be diligent about how we space ourselves and our patients. Consider adjusting workflows to ensure that visits can be conducted as quickly and safely as possible.
Communicating updated safety plans and processes are invaluable for both patients and health care workers. Patients want to be reassured that safety precautions are in place to keep the environment safe and clean. Additionally, privacy and confidentiality concerns should be addressed.
Consider a modified office schedule that can reduce the number of people in the office, person-to-person contact, and COVID-19 transmission. Social distancing is improved and PPE and other supplies are preserved.
Continue to: Employees can work on alternating days...
- Employees can work on alternating days or during different parts of the day.
- Administrative staff who do not need to be physically present in the office might work remotely.
- Expanding office hours (early morning, evening, and weekends) spreads patient visits throughout the day and minimizes high-volume in-person visits.
Institute a daily COVID-19 symptom attestation and temperature check for employees on arrival at work.
Health care personnel with symptoms of COVID-19 should be prioritized for SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2) RNA testing with an approved nucleic acid or antigen detection assay. A negative result indicates that the person most likely did not have an active SARS-CoV-2 infection at the time the sample was collected. A second test for SARS-CoV-2 RNA may be performed at the discretion of the evaluating health care provider, particularly when a higher level of clinical suspicion for infection exists.
The return to work decision should be determined by an agreed on symptom-based approach to clearance. If needed on a case-by-case basis, a review can be performed with the individual’s health care provider.12
Require universal masking and appropriate protective equipment.
- All staff members, patients, and visitors must wear masks correctly in the facilities (except children under age 2).
- All clinical staff members must wear masks correctly and eye protection during every patient encounter.
Reconfigure the waiting room and patient flow.
- Configure waiting room furniture to reinforce 6 feet of physical distancing.
- Remove all books, magazines, and toys from all waiting areas.
- Laminate signage for display.
- Install plexiglass at the check-in desk to minimize virus transmission.
- If possible, ask patients to wait in their car until their appointment time or to go directly to their exam room on arrival if it is available.
- Implement virtual check-in and check-out so that patients reduce unnecessary contact with surfaces and staff.
- Limit a high volume of patients to maintain social distancing etiquette, avoid delays, and allow adequate cleaning time between patients.
- Permit visitors to accompany adult patients to their ambulatory appointments only if special assistance is required.
- Limit the number of staff members in the exam and treatment rooms and maintain at least 6 feet between people except during medical care activities.
- Consider patient flow in a one-way traffic pattern.
Focus on keeping the clinical practice clean. (Follow the instructions and disinfect with a registered disinfectant product that meets the US Environmental Protection Agency criteria for use against COVID-19.13)
- Clean waiting rooms and restrooms frequently.
- Coordinate patient appointments to allow for infection control measures.
- Frequently clean high-touch surfaces, including tables, doorknobs, light switches, countertops, handles, desks, phones, keyboards, toilets, faucets, and sinks.
- Clinicians and all medical staff members should wash their hands before and after interacting with patients.
- Clean and disinfect the exam and treatment rooms before and after each patient.
- Use products that are effective against a range of organisms and viruses, including the coronavirus that causes COVID-19.
- Place signs indicating that rooms have been cleaned; this will assure and comfort patients. Take credit for your infection control processes.
Keep abreast of isolation and precaution guidelines. Based on data available at the time of this article’s publication, the CDC recommends ending isolation and transmission-based precautions for most people with COVID-19 using a symptom-based strategy.14 This limits unnecessary prolonged isolation and use of laboratory testing resources.
Generally, repeat SARS-CoV-2 polymerase chain reaction (PCR) testing is not recommended for “COVID-19 recovered” patients. Specifically, those patients with a prior positive SARS-CoV-2 PCR test result and who have met criteria for isolation discontinuation do not need a follow-up PCR test. A test-based strategy to discontinue isolation and transmission-based precautions is required only for severely immunocompromised patients.15
Prepare for a future COVID-19 surge and review your emergency plan and responses and revise as needed. Review handling of the current pandemic and best practices plus areas of improvement.
Symptom-based criteria for discontinuing transmission-based precautions include the following:
Patients with mild to moderate illness, not severely immunocompromised:
- at least 10 days have passed since symptoms first appeared and
- at least 24 hours have passed since last fever without fever-reducing medications and
- symptoms (cough, shortness of breath) have improved.
Note: For patients who are not severely immunocompromised and are asymptomatic throughout their infection, transmission-based precautions may be discontinued when at least 10 days have passed since the date of their first positive viral diagnostic test.
Patients with severe to critical illness, severely immunocompromised:
- at least 20 days have passed since symptoms first appeared and
- at least 24 hours have passed since last fever without fever-reducing medications and
- symptoms (cough, shortness of breath) have improved.
Note: For patients who are severely immunocompromised and are asymptomatic throughout their infection, transmission-based precautions may be discontinued when at least 20 days have passed since the date of their first positive viral diagnostic test.
Continue to: Strategy 4: Implement frequent employee communication and care...
Strategy 4: Implement frequent employee communication and care
The safety and well-being of our health care workers and patients in our clinical practices is paramount. Continuing to communicate this message and developing and sharing a plan may ameliorate the obvious toll on mental and emotional well-being. Frequent and effective communication with your clinical team is vital to reinforce policies and protocols, eliminate silos, and reduce errors.
Practice communication and care with these approaches:
- Offer regular employee COVID-19 testing.
- Re-educate staff about infection control protocols to ensure buy-in.
- Communicate with staff about the plan to address staffing shortages.
- Implement regular employee team huddles that can address accomplishments, challenges, areas for improvement, and top priorities.
- Perform regular celebrations for staff appreciation.
- Address mental health and chronic stress and offer empathy and coping resources and services to staff and clinicians. This will have a valuable, long-term benefit.
Patient communication. As the COVID-19 pandemic continues and stay-at-home policies are in place, patients should be encouraged to seek medical care if they are ill or have acute or chronic conditions. Communicate regularly with patients and let them know that their safety and well-being is the top priority. Prior to in-person visits, inform them of the safety processes that are in place to protect them.
Fostering an honest clinician-patient relationship enhances communication. Despite these efforts, some patients may not be forthcoming about their COVID-19 symptoms, illness, exposure, or travel. Health care staff can be encouraged to set a tone of tolerance and compassion and treat everyone with universal precautions.
Rising to the challenges
During the coronavirus pandemic, ObGyns continue to safely care for pregnant women and also triage and treat women who require timely office care as well as emergency and cancer-related surgeries.
The COVID-19 environment rapidly changes depending on the practice location. The strategies described represent a compilation of resources from key organizations that hopefully will prove useful and can be shaped to fit your practice. Local and regional recommendations vary, and no one can predict the course of the virus.
Consider reviewing your contingency plans regularly. As we have learned over the last several months, there is a science to maintaining a COVID-SAFE environment.
Practice operations likely will change to adapt to new conditions. The pandemic has challenged us to evolve, and we have responded with new capabilities and resilience while we continue to deliver superior and compassionate care for women.
For additional strategies on how to safeguard your practice against COVID-19, see the box below. ●
Continue to: Additional strategies on how to safeguard your practice against COVID-19...
Strategy 5: Develop a resource plan for practice operations
Assess financial solvency. Because of the mitigation measures taken during the pandemic, physician practices of all sizes are facing financial hardships and instability. As the pandemic progresses, physicians in private practice and those employed by health systems may benefit from existing resources and pandemic relief to help navigate COVID-related challenges.
Frequent revision of your financial plan may safeguard cash flow in the event of fewer patient visits and elective surgeries. Many medical organizations, including ACOG, are advocating for financial relief, fair reimbursement for telehealth and in-person visits, and access to adequate PPE. ACOG provides updated information on practice management.1
The American Medical Association (AMA) has created resources for physician practices to assist in staying focused on business and financial operations. The AMA has provided a summary of the Health, Economic Assistance, Liability Protection and Schools Act (HEALS Act).2 This is the next proposed coronavirus relief fund package, which includes provisions that benefit physicians and physician practices.
Create a plan. Review available resources and establish processes to optimize your practice capacity during the ongoing COVID-19 pandemic. Develop a game plan for patient care with a phased approach to identify and address challenges. This planning will allow your practice to pivot in response to changing local COVID-19 conditions to help you anticipate and prepare for a future surge. Maintain and revise plans as the pandemic shifts. Thinking ahead avoids the need to navigate issues in real time. Communicating clearly and often with all members of the office staff and patients lets everyone know that their safety is the main priority.
Assess staffing for flexible coverage. Frequent needs assessment helps to determine the number of staff needed to maintain a safe work environment for the patient volume.
Staff shortages may occur because of COVID-19 exposure, personal or family member illness, or childcare constraints due to daycare or school availability. Staff readiness includes evaluating individual availability and willingness.
Staff members with health issues, including comorbidities and chronic medical conditions, may not be comfortable working. Nonclinical staff members with health concerns could work remotely, although some may not be able to work from home due to technology-related issues such as WiFi deficiencies.
The CDC has interim guidelines to assist employers with providing a safe workplace and employees with making the best health decisions for themselves and their families.3,4 The US Office of Personnel Management provides guidance for COVID-19–related leave and benefits for federal employees.5
To mitigate staff shortages, approaches include adjusting schedules, cross-training to perform the tasks of other positions, and hiring additional personnel. A needs assessment can help determine if existing personnel could be cross-trained for other purposes or if additional staff should be hired. Understanding the minimum number of staff required for safe and effective patient care will assist in planning for shortages as the pandemic progresses. Understanding the availability of external resources could be a critical part of an office contingency plan.6
Proactively manage your supply chain. The pandemic has caused global supply shortages. Solid supply chain management is crucial for practice operations. Take inventory of your PPE and various supplies and place orders in advance. Analyze cash flow and connect with vendors as well as local and state health agencies to understand available resources. Given ongoing PPE shortages, practices should consider preserving PPE and employ appropriate strategies for optimizing supplies of face masks.7
Certain medications and vaccines administered in the office setting may be outdated and need to be replaced. Office equipment that has not been used for several months will need to be tested. For equipment used in office electrosurgery procedures, certain safety measures can be taken to reduce the transmission of aerosolized viral particles to the health care team. While currently the risk is theoretical and more research is needed, this potential risk should be mitigated.8 Assessing availability of hospital and ambulance or transport services also is recommended as these may change depending on the local COVID-19 status.
Strategy 6: Establish and refine the patient screening process
Patients want reassurance that the health care environment is safe and that their well-being is a priority. In advance of a patient’s visit, relieve any anxiety by explaining the COVID-SAFE measures that your practice has taken.
For employee use, consider telephone and in-person scripting to ensure consistent messaging for patients.
Prescreening. At the time of appointment scheduling and on the day prior to the scheduled appointment, all patients should be screened for symptoms of COVID-19,9 fever, exposure within 14 days to someone newly diagnosed with COVID-19, and travel within 14 days from a foreign country or from a US state with a quarantine requirement.
Patients who screen positive for symptoms, exposure, or travel should be referred to a clinician. If possible, asymptomatic patients who report exposure or travel should have their in-person visit deferred until after the required 14-day quarantine.
Consider restricting visitors from accompanying adult patients to their appointment unless they are required for special assistance.
Arrival screening. At the time of presentation for the appointment, all patients and any accompanying visitors should be rescreened. The optimal location for arrival screening should be determined by the local operations team and the infection prevention and control program.
At presentation, all patients and visitors should appropriately don a surgical mask or other face covering. Patients and visitors should have their temperature checked on arrival. Patients who screen positive for symptoms, exposure to COVID-19, and/or travel should be referred to a clinician or the visit deferred and a telehealth visit considered.
Visitors who screen positive for symptoms, fever, or exposure to COVID-19 are not permitted to accompany the patient. Asymptomatic parents or guardians of pediatric patients may serve as support persons.
References
- American College of Obstetricians and Gynecologists. Financial support for physicians and practices during the COVID-19 pandemic. https://www.acog.org/practice -management/coding/coding-library/financial-support-for-physicians-and-practices-during-the-covid-19-pandemic. Accessed July 23, 2020.
- American Medical Association. HEALS Act: What physicians and medical students need to know. https://www.ama-assn.org/delivering-care/public-health/heals-act -what-physicians-and-medical-students-need-know. Accessed July 29, 2020.
- Centers for Disease Control and Prevention. Interim guidance for businesses and employers responding to coronavirus disease 2019 (COVID-19). https://www.cdc.gov /coronavirus/2019-ncov/community/guidance-business-response.html. Accessed July 12, 2020.
- Centers for Disease Control and Prevention. Criteria for return to work for healthcare personnel with SARS-CoV-2 infection (interim guidance). https://www.cdc.gov /coronavirus/2019-ncov/hcp/return-to-work.html. Accessed July 20, 2020.
- US Office of Personnel Management. Questions and answers on human resources flexibilities and authorities for coronavirus disease 2019 (COVID-19). https://www .opm.gov/policy-data-oversight/covid-19/questions-and-answers-on-human-resources-flexibilities-and-authorities-for-coronavirus-disease-2019-covid-19.pdf. Accessed July 3, 2020.
- Centers for Disease Control and Prevention. Strategies to mitigate healthcare personnel staffing shortages. https://www.cdc.gov/coronavirus/2019-ncov/hcp /mitigating-staff-shortages.html. Accessed July 17, 2020.
- Centers for Disease Control and Prevention. Strategies for optimizing the supply of facemasks. https://www.cdc.gov/coronavirus/2019-ncov/hcp/ppe-strategy/face -masks.html. Accessed July 3, 2020.
- Rahman S, Klebanoff J, Moawad G. Smoke evacuation in the age of COVID-19. Contemporary OB/GYN. July 2, 2020. https://www.contemporaryobgyn.net/view/smoke -evacuation-in-the-age-of-covid-19. Accessed July 3, 2020.
- Centers for Disease Control and Prevention. Symptoms of coronavirus. https://www.cdc.gov/coronavirus/2019-ncov/symptoms-testing/symptoms.html. Accessed July 25, 2020.
- Centers for Disease Control and Prevention. Information for healthcare professionals about coronavirus (COVID-19). https://www.cdc.gov/coronavirus/2019-nCoV/hcp/index.html. Accessed July 22, 2020.
- US Department of Labor Occupational Safety and Health Administration. COVID-19 control and prevention: healthcare workers and employers. https://www.osha.gov/SLTC/covid-19/healthcare-workers.html. Accessed August 5, 2020.
- Centers for Medicare and Medicaid Services. COVID-19 resources for health care professionals and researchers. https://www.cms.gov/About-CMS/Agency-Information/OMH/resource-center/hcps-and-researchers. Accessed August 7, 2020.
- American College of Obstetricians and Gynecologists. COVID-19. https://www.acog.org/topics/covid-19. Accessed August 7, 2020.
- Society for Maternal-Fetal Medicine. Coronavirus (COVID-19). https://www.smfm.org/covid19. Accessed August 7, 2020.
- Society of Gynecologic Surgeons. Joint statement on re-introduction of hospital and office-based procedures in the COVID-19 climate for the practicing gynecologist. https://www.sgsonline.org/joint-statement-on-re-introduction-of-hospital-and-office-based-procedures-in-the-covid-19-climate. Accessed August 7, 2020.
- AAGL. COVID-19 articles, resources and webinars. https://www.aagl.org/covid-19/. Accessed August 7, 2020.
- American Society for Reproductive Medicine. COVID-19 updates and resources. https://www.asrm.org/news-and-publications/covid-19/. Accessed August 7, 2020.
- Society of Gynecologic Oncology. COVID-19 resources for health care practitioners. https://www.sgo.org/practice-management/covid-19/. Accessed August 7, 2020.
- Centers for Disease Control and Prevention. Using telehealth to expand access to essential health services during the COVID-19 pandemic. https://www.cdc.gov/coronavirus/2019-ncov/hcp/telehealth.html. Accessed July 3, 2020.
- Nguyen LH, Drew DA, Graham MS, et al; on behalf of the COronavirus Pandemic Epidemiology Consortium. Risk of COVID-19 among front-line health-care workers and the general community: a prospective cohort study. Lancet. July 31, 2020. http://www.thelancet-press.com/embargo/hcwcovid.pdf. Accessed August 25, 2020.
- Centers for Disease Control and Prevention. Criteria for return to work for healthcare personnel with SARS-CoV-2 infection (interim guidance). https://www.cdc.gov/coronavirus/2019-ncov/hcp/return-to-work.html. Accessed July 20, 2020.
- US Environmental Protection Agency. List N: Disinfectants for use against SARS-CoV-2 (COVID-19). https://www.epa.gov/pesticide-registration/list-n-disinfectants-use-against-sars-cov-2-covid-19. Accessed August 24, 2020.
- Centers for Disease Control and Prevention. Duration of isolation and precautions for adults with COVID-19. https://www.cdc.gov/coronavirus/2019-ncov/hcp/duration-isolation.html. Accessed July 23, 2020.
- Centers for Disease Control and Prevention. Discontinuation of transmission-based precautions and disposition of patients with COVID-19 in healthcare settings (interim guidance). https://www.cdc.gov/coronavirus/2019-ncov/hcp/disposition-hospitalized-patients.html#fn1. Accessed July 23, 2020.
No question, the COVID-19 pandemic has been a challenging time for medical practices across the United States. Uncertainty remains regarding bringing patients and services back into our offices. One factor that distinguishes many ObGyn practices from other specialties is that our practices have remained open—in some form—since the beginning of the pandemic. In various parts of the country, gynecologic surgeries and routine office visits have been significantly reduced; however, deliveries and gynecologic emergencies have continued.
In this article, I suggest a framework of strategies and resources to provide insight for outpatient operations. Individual practices will vary across the nation depending on local conditions. Full practice capacity may take on a different look than it had prior to the pandemic, and there is opportunity to change the way we operate.
Strategy 1: Consult regulatory requirements frequently
As the local status of COVID-19 evolves quickly, it is essential to examine the frequently updated recommendations from regulatory agencies at the federal, state, and local levels. Clinical practices that function within health systems need to demonstrate alignment with hospital or university policies and procedures. The Centers for Disease Control and Prevention (CDC), Occupational Safety and Health Administration (OSHA), Centers for Medicare and Medicaid Services (CMS), and individual state departments of health provide dynamic resources that are easily accessible online.1-3
The American College of Obstetricians and Gynecologists (ACOG) continues to be an excellent medical society resource.4 Subspecialty organizations that provide up-to-date guidance include the Society for Maternal-Fetal Medicine (SMFM), Society of Gynecologic Surgeons (SGS), AAGL (American Association of Gynecologic Laparoscopists), American Society for Reproductive Medicine (ASRM), and Society of Gynecologic Oncology (SGO).5-9 These resources are updated as more information about COVID-19 emerges, and they may be modified to different local-regional conditions.
The professional liability insurance carrier is an important source of insight for a number of circumstances, including modifications to your office practice, such as returning to full-scope or part-time practice; operating outside normal clinical service arrangements (for example, assisting with emergency care); offering telehealth services; and adding extra hours or employees to accommodate the patient backlog. Business insurance coverage is a separate issue to consider. Reviewing the practice policy may protect your business from COVID-related liabilities.
Consulting with legal counselors can be helpful. They can assist with navigating various practice and personnel COVID-related changes, as well as developing a viable plan for patients who were previously insured pre–COVID-19 who are currently uninsured.
Continue to: Strategy 2: Reimagine schedule capacity...
Strategy 2: Reimagine schedule capacity
The waxing and waning of the COVID-19 crisis presents an opportunity to evaluate our office practices and make necessary and positive changes. The question becomes, do we operate our practices as usual or do we rethink our strategy for seeing patients and integrate lessons learned from the pandemic? Patients are deciding when they are comfortable to schedule elective surgeries and routine office encounters. This gives us the chance to break from the tradition of 100% in-person visits and change the way we care for women.
The coronavirus has accelerated the rise of telehealth/telemedicine and is, perhaps, a silver lining of the pandemic. Telehealth is a valuable tool for accessing health services when in-person visits are not possible. Evaluating and triaging patients for in-person versus telehealth visits is now a viable option for clinical practice and reduces exposure to COVID-19 infection.
Telemedicine is convenient, and clinicians can use it to counsel and screen for various health issues as well as to extend their reach to rural communities. Appropriate consent should be documented in the patient chart. As some areas continue to be without adequate access to WiFi, telephone contact also is currently acceptable. Telehealth does not replace the in-person visit but can be viewed as a complementary and supplementary service.
Consider a balance between telehealth and in-person visits by evaluating which visits can continue remotely and which can alternate with in-person visits. This offers tremendous flexibility and will expand delivery of essential health care to patients.10 Integrating telemedicine into clinical practice provides an additional benefit: It minimizes the exposure and transmission of COVID-19 to health care workers and patients and preserves supplies, including personal protective equipment (PPE).
Prioritize the backlog of patients who require follow-up testing, procedures, and surgeries. Communicate with patients that it is safe to be seen and important to not avoid routine and preventative visits that might reveal concerns or conditions that require treatment.
Strategy 3: Institute infection prevention and control measures
The importance of instituting and ensuring safety measures for office personnel and patients cannot be underestimated. Recently, a study from King’s College in London found that frontline health care workers with PPE still have 3 to 4 times the risk of contracting coronavirus compared with the general public.11 Health care systems should ensure adequate PPE availability and develop additional strategies to protect health care workers from COVID-19. We have to be fanatical about cleanliness and PPE. We have to be diligent about how we space ourselves and our patients. Consider adjusting workflows to ensure that visits can be conducted as quickly and safely as possible.
Communicating updated safety plans and processes are invaluable for both patients and health care workers. Patients want to be reassured that safety precautions are in place to keep the environment safe and clean. Additionally, privacy and confidentiality concerns should be addressed.
Consider a modified office schedule that can reduce the number of people in the office, person-to-person contact, and COVID-19 transmission. Social distancing is improved and PPE and other supplies are preserved.
Continue to: Employees can work on alternating days...
- Employees can work on alternating days or during different parts of the day.
- Administrative staff who do not need to be physically present in the office might work remotely.
- Expanding office hours (early morning, evening, and weekends) spreads patient visits throughout the day and minimizes high-volume in-person visits.
Institute a daily COVID-19 symptom attestation and temperature check for employees on arrival at work.
Health care personnel with symptoms of COVID-19 should be prioritized for SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2) RNA testing with an approved nucleic acid or antigen detection assay. A negative result indicates that the person most likely did not have an active SARS-CoV-2 infection at the time the sample was collected. A second test for SARS-CoV-2 RNA may be performed at the discretion of the evaluating health care provider, particularly when a higher level of clinical suspicion for infection exists.
The return to work decision should be determined by an agreed on symptom-based approach to clearance. If needed on a case-by-case basis, a review can be performed with the individual’s health care provider.12
Require universal masking and appropriate protective equipment.
- All staff members, patients, and visitors must wear masks correctly in the facilities (except children under age 2).
- All clinical staff members must wear masks correctly and eye protection during every patient encounter.
Reconfigure the waiting room and patient flow.
- Configure waiting room furniture to reinforce 6 feet of physical distancing.
- Remove all books, magazines, and toys from all waiting areas.
- Laminate signage for display.
- Install plexiglass at the check-in desk to minimize virus transmission.
- If possible, ask patients to wait in their car until their appointment time or to go directly to their exam room on arrival if it is available.
- Implement virtual check-in and check-out so that patients reduce unnecessary contact with surfaces and staff.
- Limit a high volume of patients to maintain social distancing etiquette, avoid delays, and allow adequate cleaning time between patients.
- Permit visitors to accompany adult patients to their ambulatory appointments only if special assistance is required.
- Limit the number of staff members in the exam and treatment rooms and maintain at least 6 feet between people except during medical care activities.
- Consider patient flow in a one-way traffic pattern.
Focus on keeping the clinical practice clean. (Follow the instructions and disinfect with a registered disinfectant product that meets the US Environmental Protection Agency criteria for use against COVID-19.13)
- Clean waiting rooms and restrooms frequently.
- Coordinate patient appointments to allow for infection control measures.
- Frequently clean high-touch surfaces, including tables, doorknobs, light switches, countertops, handles, desks, phones, keyboards, toilets, faucets, and sinks.
- Clinicians and all medical staff members should wash their hands before and after interacting with patients.
- Clean and disinfect the exam and treatment rooms before and after each patient.
- Use products that are effective against a range of organisms and viruses, including the coronavirus that causes COVID-19.
- Place signs indicating that rooms have been cleaned; this will assure and comfort patients. Take credit for your infection control processes.
Keep abreast of isolation and precaution guidelines. Based on data available at the time of this article’s publication, the CDC recommends ending isolation and transmission-based precautions for most people with COVID-19 using a symptom-based strategy.14 This limits unnecessary prolonged isolation and use of laboratory testing resources.
Generally, repeat SARS-CoV-2 polymerase chain reaction (PCR) testing is not recommended for “COVID-19 recovered” patients. Specifically, those patients with a prior positive SARS-CoV-2 PCR test result and who have met criteria for isolation discontinuation do not need a follow-up PCR test. A test-based strategy to discontinue isolation and transmission-based precautions is required only for severely immunocompromised patients.15
Prepare for a future COVID-19 surge and review your emergency plan and responses and revise as needed. Review handling of the current pandemic and best practices plus areas of improvement.
Symptom-based criteria for discontinuing transmission-based precautions include the following:
Patients with mild to moderate illness, not severely immunocompromised:
- at least 10 days have passed since symptoms first appeared and
- at least 24 hours have passed since last fever without fever-reducing medications and
- symptoms (cough, shortness of breath) have improved.
Note: For patients who are not severely immunocompromised and are asymptomatic throughout their infection, transmission-based precautions may be discontinued when at least 10 days have passed since the date of their first positive viral diagnostic test.
Patients with severe to critical illness, severely immunocompromised:
- at least 20 days have passed since symptoms first appeared and
- at least 24 hours have passed since last fever without fever-reducing medications and
- symptoms (cough, shortness of breath) have improved.
Note: For patients who are severely immunocompromised and are asymptomatic throughout their infection, transmission-based precautions may be discontinued when at least 20 days have passed since the date of their first positive viral diagnostic test.
Continue to: Strategy 4: Implement frequent employee communication and care...
Strategy 4: Implement frequent employee communication and care
The safety and well-being of our health care workers and patients in our clinical practices is paramount. Continuing to communicate this message and developing and sharing a plan may ameliorate the obvious toll on mental and emotional well-being. Frequent and effective communication with your clinical team is vital to reinforce policies and protocols, eliminate silos, and reduce errors.
Practice communication and care with these approaches:
- Offer regular employee COVID-19 testing.
- Re-educate staff about infection control protocols to ensure buy-in.
- Communicate with staff about the plan to address staffing shortages.
- Implement regular employee team huddles that can address accomplishments, challenges, areas for improvement, and top priorities.
- Perform regular celebrations for staff appreciation.
- Address mental health and chronic stress and offer empathy and coping resources and services to staff and clinicians. This will have a valuable, long-term benefit.
Patient communication. As the COVID-19 pandemic continues and stay-at-home policies are in place, patients should be encouraged to seek medical care if they are ill or have acute or chronic conditions. Communicate regularly with patients and let them know that their safety and well-being is the top priority. Prior to in-person visits, inform them of the safety processes that are in place to protect them.
Fostering an honest clinician-patient relationship enhances communication. Despite these efforts, some patients may not be forthcoming about their COVID-19 symptoms, illness, exposure, or travel. Health care staff can be encouraged to set a tone of tolerance and compassion and treat everyone with universal precautions.
Rising to the challenges
During the coronavirus pandemic, ObGyns continue to safely care for pregnant women and also triage and treat women who require timely office care as well as emergency and cancer-related surgeries.
The COVID-19 environment rapidly changes depending on the practice location. The strategies described represent a compilation of resources from key organizations that hopefully will prove useful and can be shaped to fit your practice. Local and regional recommendations vary, and no one can predict the course of the virus.
Consider reviewing your contingency plans regularly. As we have learned over the last several months, there is a science to maintaining a COVID-SAFE environment.
Practice operations likely will change to adapt to new conditions. The pandemic has challenged us to evolve, and we have responded with new capabilities and resilience while we continue to deliver superior and compassionate care for women.
For additional strategies on how to safeguard your practice against COVID-19, see the box below. ●
Continue to: Additional strategies on how to safeguard your practice against COVID-19...
Strategy 5: Develop a resource plan for practice operations
Assess financial solvency. Because of the mitigation measures taken during the pandemic, physician practices of all sizes are facing financial hardships and instability. As the pandemic progresses, physicians in private practice and those employed by health systems may benefit from existing resources and pandemic relief to help navigate COVID-related challenges.
Frequent revision of your financial plan may safeguard cash flow in the event of fewer patient visits and elective surgeries. Many medical organizations, including ACOG, are advocating for financial relief, fair reimbursement for telehealth and in-person visits, and access to adequate PPE. ACOG provides updated information on practice management.1
The American Medical Association (AMA) has created resources for physician practices to assist in staying focused on business and financial operations. The AMA has provided a summary of the Health, Economic Assistance, Liability Protection and Schools Act (HEALS Act).2 This is the next proposed coronavirus relief fund package, which includes provisions that benefit physicians and physician practices.
Create a plan. Review available resources and establish processes to optimize your practice capacity during the ongoing COVID-19 pandemic. Develop a game plan for patient care with a phased approach to identify and address challenges. This planning will allow your practice to pivot in response to changing local COVID-19 conditions to help you anticipate and prepare for a future surge. Maintain and revise plans as the pandemic shifts. Thinking ahead avoids the need to navigate issues in real time. Communicating clearly and often with all members of the office staff and patients lets everyone know that their safety is the main priority.
Assess staffing for flexible coverage. Frequent needs assessment helps to determine the number of staff needed to maintain a safe work environment for the patient volume.
Staff shortages may occur because of COVID-19 exposure, personal or family member illness, or childcare constraints due to daycare or school availability. Staff readiness includes evaluating individual availability and willingness.
Staff members with health issues, including comorbidities and chronic medical conditions, may not be comfortable working. Nonclinical staff members with health concerns could work remotely, although some may not be able to work from home due to technology-related issues such as WiFi deficiencies.
The CDC has interim guidelines to assist employers with providing a safe workplace and employees with making the best health decisions for themselves and their families.3,4 The US Office of Personnel Management provides guidance for COVID-19–related leave and benefits for federal employees.5
To mitigate staff shortages, approaches include adjusting schedules, cross-training to perform the tasks of other positions, and hiring additional personnel. A needs assessment can help determine if existing personnel could be cross-trained for other purposes or if additional staff should be hired. Understanding the minimum number of staff required for safe and effective patient care will assist in planning for shortages as the pandemic progresses. Understanding the availability of external resources could be a critical part of an office contingency plan.6
Proactively manage your supply chain. The pandemic has caused global supply shortages. Solid supply chain management is crucial for practice operations. Take inventory of your PPE and various supplies and place orders in advance. Analyze cash flow and connect with vendors as well as local and state health agencies to understand available resources. Given ongoing PPE shortages, practices should consider preserving PPE and employ appropriate strategies for optimizing supplies of face masks.7
Certain medications and vaccines administered in the office setting may be outdated and need to be replaced. Office equipment that has not been used for several months will need to be tested. For equipment used in office electrosurgery procedures, certain safety measures can be taken to reduce the transmission of aerosolized viral particles to the health care team. While currently the risk is theoretical and more research is needed, this potential risk should be mitigated.8 Assessing availability of hospital and ambulance or transport services also is recommended as these may change depending on the local COVID-19 status.
Strategy 6: Establish and refine the patient screening process
Patients want reassurance that the health care environment is safe and that their well-being is a priority. In advance of a patient’s visit, relieve any anxiety by explaining the COVID-SAFE measures that your practice has taken.
For employee use, consider telephone and in-person scripting to ensure consistent messaging for patients.
Prescreening. At the time of appointment scheduling and on the day prior to the scheduled appointment, all patients should be screened for symptoms of COVID-19,9 fever, exposure within 14 days to someone newly diagnosed with COVID-19, and travel within 14 days from a foreign country or from a US state with a quarantine requirement.
Patients who screen positive for symptoms, exposure, or travel should be referred to a clinician. If possible, asymptomatic patients who report exposure or travel should have their in-person visit deferred until after the required 14-day quarantine.
Consider restricting visitors from accompanying adult patients to their appointment unless they are required for special assistance.
Arrival screening. At the time of presentation for the appointment, all patients and any accompanying visitors should be rescreened. The optimal location for arrival screening should be determined by the local operations team and the infection prevention and control program.
At presentation, all patients and visitors should appropriately don a surgical mask or other face covering. Patients and visitors should have their temperature checked on arrival. Patients who screen positive for symptoms, exposure to COVID-19, and/or travel should be referred to a clinician or the visit deferred and a telehealth visit considered.
Visitors who screen positive for symptoms, fever, or exposure to COVID-19 are not permitted to accompany the patient. Asymptomatic parents or guardians of pediatric patients may serve as support persons.
References
- American College of Obstetricians and Gynecologists. Financial support for physicians and practices during the COVID-19 pandemic. https://www.acog.org/practice -management/coding/coding-library/financial-support-for-physicians-and-practices-during-the-covid-19-pandemic. Accessed July 23, 2020.
- American Medical Association. HEALS Act: What physicians and medical students need to know. https://www.ama-assn.org/delivering-care/public-health/heals-act -what-physicians-and-medical-students-need-know. Accessed July 29, 2020.
- Centers for Disease Control and Prevention. Interim guidance for businesses and employers responding to coronavirus disease 2019 (COVID-19). https://www.cdc.gov /coronavirus/2019-ncov/community/guidance-business-response.html. Accessed July 12, 2020.
- Centers for Disease Control and Prevention. Criteria for return to work for healthcare personnel with SARS-CoV-2 infection (interim guidance). https://www.cdc.gov /coronavirus/2019-ncov/hcp/return-to-work.html. Accessed July 20, 2020.
- US Office of Personnel Management. Questions and answers on human resources flexibilities and authorities for coronavirus disease 2019 (COVID-19). https://www .opm.gov/policy-data-oversight/covid-19/questions-and-answers-on-human-resources-flexibilities-and-authorities-for-coronavirus-disease-2019-covid-19.pdf. Accessed July 3, 2020.
- Centers for Disease Control and Prevention. Strategies to mitigate healthcare personnel staffing shortages. https://www.cdc.gov/coronavirus/2019-ncov/hcp /mitigating-staff-shortages.html. Accessed July 17, 2020.
- Centers for Disease Control and Prevention. Strategies for optimizing the supply of facemasks. https://www.cdc.gov/coronavirus/2019-ncov/hcp/ppe-strategy/face -masks.html. Accessed July 3, 2020.
- Rahman S, Klebanoff J, Moawad G. Smoke evacuation in the age of COVID-19. Contemporary OB/GYN. July 2, 2020. https://www.contemporaryobgyn.net/view/smoke -evacuation-in-the-age-of-covid-19. Accessed July 3, 2020.
- Centers for Disease Control and Prevention. Symptoms of coronavirus. https://www.cdc.gov/coronavirus/2019-ncov/symptoms-testing/symptoms.html. Accessed July 25, 2020.
No question, the COVID-19 pandemic has been a challenging time for medical practices across the United States. Uncertainty remains regarding bringing patients and services back into our offices. One factor that distinguishes many ObGyn practices from other specialties is that our practices have remained open—in some form—since the beginning of the pandemic. In various parts of the country, gynecologic surgeries and routine office visits have been significantly reduced; however, deliveries and gynecologic emergencies have continued.
In this article, I suggest a framework of strategies and resources to provide insight for outpatient operations. Individual practices will vary across the nation depending on local conditions. Full practice capacity may take on a different look than it had prior to the pandemic, and there is opportunity to change the way we operate.
Strategy 1: Consult regulatory requirements frequently
As the local status of COVID-19 evolves quickly, it is essential to examine the frequently updated recommendations from regulatory agencies at the federal, state, and local levels. Clinical practices that function within health systems need to demonstrate alignment with hospital or university policies and procedures. The Centers for Disease Control and Prevention (CDC), Occupational Safety and Health Administration (OSHA), Centers for Medicare and Medicaid Services (CMS), and individual state departments of health provide dynamic resources that are easily accessible online.1-3
The American College of Obstetricians and Gynecologists (ACOG) continues to be an excellent medical society resource.4 Subspecialty organizations that provide up-to-date guidance include the Society for Maternal-Fetal Medicine (SMFM), Society of Gynecologic Surgeons (SGS), AAGL (American Association of Gynecologic Laparoscopists), American Society for Reproductive Medicine (ASRM), and Society of Gynecologic Oncology (SGO).5-9 These resources are updated as more information about COVID-19 emerges, and they may be modified to different local-regional conditions.
The professional liability insurance carrier is an important source of insight for a number of circumstances, including modifications to your office practice, such as returning to full-scope or part-time practice; operating outside normal clinical service arrangements (for example, assisting with emergency care); offering telehealth services; and adding extra hours or employees to accommodate the patient backlog. Business insurance coverage is a separate issue to consider. Reviewing the practice policy may protect your business from COVID-related liabilities.
Consulting with legal counselors can be helpful. They can assist with navigating various practice and personnel COVID-related changes, as well as developing a viable plan for patients who were previously insured pre–COVID-19 who are currently uninsured.
Continue to: Strategy 2: Reimagine schedule capacity...
Strategy 2: Reimagine schedule capacity
The waxing and waning of the COVID-19 crisis presents an opportunity to evaluate our office practices and make necessary and positive changes. The question becomes, do we operate our practices as usual or do we rethink our strategy for seeing patients and integrate lessons learned from the pandemic? Patients are deciding when they are comfortable to schedule elective surgeries and routine office encounters. This gives us the chance to break from the tradition of 100% in-person visits and change the way we care for women.
The coronavirus has accelerated the rise of telehealth/telemedicine and is, perhaps, a silver lining of the pandemic. Telehealth is a valuable tool for accessing health services when in-person visits are not possible. Evaluating and triaging patients for in-person versus telehealth visits is now a viable option for clinical practice and reduces exposure to COVID-19 infection.
Telemedicine is convenient, and clinicians can use it to counsel and screen for various health issues as well as to extend their reach to rural communities. Appropriate consent should be documented in the patient chart. As some areas continue to be without adequate access to WiFi, telephone contact also is currently acceptable. Telehealth does not replace the in-person visit but can be viewed as a complementary and supplementary service.
Consider a balance between telehealth and in-person visits by evaluating which visits can continue remotely and which can alternate with in-person visits. This offers tremendous flexibility and will expand delivery of essential health care to patients.10 Integrating telemedicine into clinical practice provides an additional benefit: It minimizes the exposure and transmission of COVID-19 to health care workers and patients and preserves supplies, including personal protective equipment (PPE).
Prioritize the backlog of patients who require follow-up testing, procedures, and surgeries. Communicate with patients that it is safe to be seen and important to not avoid routine and preventative visits that might reveal concerns or conditions that require treatment.
Strategy 3: Institute infection prevention and control measures
The importance of instituting and ensuring safety measures for office personnel and patients cannot be underestimated. Recently, a study from King’s College in London found that frontline health care workers with PPE still have 3 to 4 times the risk of contracting coronavirus compared with the general public.11 Health care systems should ensure adequate PPE availability and develop additional strategies to protect health care workers from COVID-19. We have to be fanatical about cleanliness and PPE. We have to be diligent about how we space ourselves and our patients. Consider adjusting workflows to ensure that visits can be conducted as quickly and safely as possible.
Communicating updated safety plans and processes are invaluable for both patients and health care workers. Patients want to be reassured that safety precautions are in place to keep the environment safe and clean. Additionally, privacy and confidentiality concerns should be addressed.
Consider a modified office schedule that can reduce the number of people in the office, person-to-person contact, and COVID-19 transmission. Social distancing is improved and PPE and other supplies are preserved.
Continue to: Employees can work on alternating days...
- Employees can work on alternating days or during different parts of the day.
- Administrative staff who do not need to be physically present in the office might work remotely.
- Expanding office hours (early morning, evening, and weekends) spreads patient visits throughout the day and minimizes high-volume in-person visits.
Institute a daily COVID-19 symptom attestation and temperature check for employees on arrival at work.
Health care personnel with symptoms of COVID-19 should be prioritized for SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2) RNA testing with an approved nucleic acid or antigen detection assay. A negative result indicates that the person most likely did not have an active SARS-CoV-2 infection at the time the sample was collected. A second test for SARS-CoV-2 RNA may be performed at the discretion of the evaluating health care provider, particularly when a higher level of clinical suspicion for infection exists.
The return to work decision should be determined by an agreed on symptom-based approach to clearance. If needed on a case-by-case basis, a review can be performed with the individual’s health care provider.12
Require universal masking and appropriate protective equipment.
- All staff members, patients, and visitors must wear masks correctly in the facilities (except children under age 2).
- All clinical staff members must wear masks correctly and eye protection during every patient encounter.
Reconfigure the waiting room and patient flow.
- Configure waiting room furniture to reinforce 6 feet of physical distancing.
- Remove all books, magazines, and toys from all waiting areas.
- Laminate signage for display.
- Install plexiglass at the check-in desk to minimize virus transmission.
- If possible, ask patients to wait in their car until their appointment time or to go directly to their exam room on arrival if it is available.
- Implement virtual check-in and check-out so that patients reduce unnecessary contact with surfaces and staff.
- Limit a high volume of patients to maintain social distancing etiquette, avoid delays, and allow adequate cleaning time between patients.
- Permit visitors to accompany adult patients to their ambulatory appointments only if special assistance is required.
- Limit the number of staff members in the exam and treatment rooms and maintain at least 6 feet between people except during medical care activities.
- Consider patient flow in a one-way traffic pattern.
Focus on keeping the clinical practice clean. (Follow the instructions and disinfect with a registered disinfectant product that meets the US Environmental Protection Agency criteria for use against COVID-19.13)
- Clean waiting rooms and restrooms frequently.
- Coordinate patient appointments to allow for infection control measures.
- Frequently clean high-touch surfaces, including tables, doorknobs, light switches, countertops, handles, desks, phones, keyboards, toilets, faucets, and sinks.
- Clinicians and all medical staff members should wash their hands before and after interacting with patients.
- Clean and disinfect the exam and treatment rooms before and after each patient.
- Use products that are effective against a range of organisms and viruses, including the coronavirus that causes COVID-19.
- Place signs indicating that rooms have been cleaned; this will assure and comfort patients. Take credit for your infection control processes.
Keep abreast of isolation and precaution guidelines. Based on data available at the time of this article’s publication, the CDC recommends ending isolation and transmission-based precautions for most people with COVID-19 using a symptom-based strategy.14 This limits unnecessary prolonged isolation and use of laboratory testing resources.
Generally, repeat SARS-CoV-2 polymerase chain reaction (PCR) testing is not recommended for “COVID-19 recovered” patients. Specifically, those patients with a prior positive SARS-CoV-2 PCR test result and who have met criteria for isolation discontinuation do not need a follow-up PCR test. A test-based strategy to discontinue isolation and transmission-based precautions is required only for severely immunocompromised patients.15
Prepare for a future COVID-19 surge and review your emergency plan and responses and revise as needed. Review handling of the current pandemic and best practices plus areas of improvement.
Symptom-based criteria for discontinuing transmission-based precautions include the following:
Patients with mild to moderate illness, not severely immunocompromised:
- at least 10 days have passed since symptoms first appeared and
- at least 24 hours have passed since last fever without fever-reducing medications and
- symptoms (cough, shortness of breath) have improved.
Note: For patients who are not severely immunocompromised and are asymptomatic throughout their infection, transmission-based precautions may be discontinued when at least 10 days have passed since the date of their first positive viral diagnostic test.
Patients with severe to critical illness, severely immunocompromised:
- at least 20 days have passed since symptoms first appeared and
- at least 24 hours have passed since last fever without fever-reducing medications and
- symptoms (cough, shortness of breath) have improved.
Note: For patients who are severely immunocompromised and are asymptomatic throughout their infection, transmission-based precautions may be discontinued when at least 20 days have passed since the date of their first positive viral diagnostic test.
Continue to: Strategy 4: Implement frequent employee communication and care...
Strategy 4: Implement frequent employee communication and care
The safety and well-being of our health care workers and patients in our clinical practices is paramount. Continuing to communicate this message and developing and sharing a plan may ameliorate the obvious toll on mental and emotional well-being. Frequent and effective communication with your clinical team is vital to reinforce policies and protocols, eliminate silos, and reduce errors.
Practice communication and care with these approaches:
- Offer regular employee COVID-19 testing.
- Re-educate staff about infection control protocols to ensure buy-in.
- Communicate with staff about the plan to address staffing shortages.
- Implement regular employee team huddles that can address accomplishments, challenges, areas for improvement, and top priorities.
- Perform regular celebrations for staff appreciation.
- Address mental health and chronic stress and offer empathy and coping resources and services to staff and clinicians. This will have a valuable, long-term benefit.
Patient communication. As the COVID-19 pandemic continues and stay-at-home policies are in place, patients should be encouraged to seek medical care if they are ill or have acute or chronic conditions. Communicate regularly with patients and let them know that their safety and well-being is the top priority. Prior to in-person visits, inform them of the safety processes that are in place to protect them.
Fostering an honest clinician-patient relationship enhances communication. Despite these efforts, some patients may not be forthcoming about their COVID-19 symptoms, illness, exposure, or travel. Health care staff can be encouraged to set a tone of tolerance and compassion and treat everyone with universal precautions.
Rising to the challenges
During the coronavirus pandemic, ObGyns continue to safely care for pregnant women and also triage and treat women who require timely office care as well as emergency and cancer-related surgeries.
The COVID-19 environment rapidly changes depending on the practice location. The strategies described represent a compilation of resources from key organizations that hopefully will prove useful and can be shaped to fit your practice. Local and regional recommendations vary, and no one can predict the course of the virus.
Consider reviewing your contingency plans regularly. As we have learned over the last several months, there is a science to maintaining a COVID-SAFE environment.
Practice operations likely will change to adapt to new conditions. The pandemic has challenged us to evolve, and we have responded with new capabilities and resilience while we continue to deliver superior and compassionate care for women.
For additional strategies on how to safeguard your practice against COVID-19, see the box below. ●
Continue to: Additional strategies on how to safeguard your practice against COVID-19...
Strategy 5: Develop a resource plan for practice operations
Assess financial solvency. Because of the mitigation measures taken during the pandemic, physician practices of all sizes are facing financial hardships and instability. As the pandemic progresses, physicians in private practice and those employed by health systems may benefit from existing resources and pandemic relief to help navigate COVID-related challenges.
Frequent revision of your financial plan may safeguard cash flow in the event of fewer patient visits and elective surgeries. Many medical organizations, including ACOG, are advocating for financial relief, fair reimbursement for telehealth and in-person visits, and access to adequate PPE. ACOG provides updated information on practice management.1
The American Medical Association (AMA) has created resources for physician practices to assist in staying focused on business and financial operations. The AMA has provided a summary of the Health, Economic Assistance, Liability Protection and Schools Act (HEALS Act).2 This is the next proposed coronavirus relief fund package, which includes provisions that benefit physicians and physician practices.
Create a plan. Review available resources and establish processes to optimize your practice capacity during the ongoing COVID-19 pandemic. Develop a game plan for patient care with a phased approach to identify and address challenges. This planning will allow your practice to pivot in response to changing local COVID-19 conditions to help you anticipate and prepare for a future surge. Maintain and revise plans as the pandemic shifts. Thinking ahead avoids the need to navigate issues in real time. Communicating clearly and often with all members of the office staff and patients lets everyone know that their safety is the main priority.
Assess staffing for flexible coverage. Frequent needs assessment helps to determine the number of staff needed to maintain a safe work environment for the patient volume.
Staff shortages may occur because of COVID-19 exposure, personal or family member illness, or childcare constraints due to daycare or school availability. Staff readiness includes evaluating individual availability and willingness.
Staff members with health issues, including comorbidities and chronic medical conditions, may not be comfortable working. Nonclinical staff members with health concerns could work remotely, although some may not be able to work from home due to technology-related issues such as WiFi deficiencies.
The CDC has interim guidelines to assist employers with providing a safe workplace and employees with making the best health decisions for themselves and their families.3,4 The US Office of Personnel Management provides guidance for COVID-19–related leave and benefits for federal employees.5
To mitigate staff shortages, approaches include adjusting schedules, cross-training to perform the tasks of other positions, and hiring additional personnel. A needs assessment can help determine if existing personnel could be cross-trained for other purposes or if additional staff should be hired. Understanding the minimum number of staff required for safe and effective patient care will assist in planning for shortages as the pandemic progresses. Understanding the availability of external resources could be a critical part of an office contingency plan.6
Proactively manage your supply chain. The pandemic has caused global supply shortages. Solid supply chain management is crucial for practice operations. Take inventory of your PPE and various supplies and place orders in advance. Analyze cash flow and connect with vendors as well as local and state health agencies to understand available resources. Given ongoing PPE shortages, practices should consider preserving PPE and employ appropriate strategies for optimizing supplies of face masks.7
Certain medications and vaccines administered in the office setting may be outdated and need to be replaced. Office equipment that has not been used for several months will need to be tested. For equipment used in office electrosurgery procedures, certain safety measures can be taken to reduce the transmission of aerosolized viral particles to the health care team. While currently the risk is theoretical and more research is needed, this potential risk should be mitigated.8 Assessing availability of hospital and ambulance or transport services also is recommended as these may change depending on the local COVID-19 status.
Strategy 6: Establish and refine the patient screening process
Patients want reassurance that the health care environment is safe and that their well-being is a priority. In advance of a patient’s visit, relieve any anxiety by explaining the COVID-SAFE measures that your practice has taken.
For employee use, consider telephone and in-person scripting to ensure consistent messaging for patients.
Prescreening. At the time of appointment scheduling and on the day prior to the scheduled appointment, all patients should be screened for symptoms of COVID-19,9 fever, exposure within 14 days to someone newly diagnosed with COVID-19, and travel within 14 days from a foreign country or from a US state with a quarantine requirement.
Patients who screen positive for symptoms, exposure, or travel should be referred to a clinician. If possible, asymptomatic patients who report exposure or travel should have their in-person visit deferred until after the required 14-day quarantine.
Consider restricting visitors from accompanying adult patients to their appointment unless they are required for special assistance.
Arrival screening. At the time of presentation for the appointment, all patients and any accompanying visitors should be rescreened. The optimal location for arrival screening should be determined by the local operations team and the infection prevention and control program.
At presentation, all patients and visitors should appropriately don a surgical mask or other face covering. Patients and visitors should have their temperature checked on arrival. Patients who screen positive for symptoms, exposure to COVID-19, and/or travel should be referred to a clinician or the visit deferred and a telehealth visit considered.
Visitors who screen positive for symptoms, fever, or exposure to COVID-19 are not permitted to accompany the patient. Asymptomatic parents or guardians of pediatric patients may serve as support persons.
References
- American College of Obstetricians and Gynecologists. Financial support for physicians and practices during the COVID-19 pandemic. https://www.acog.org/practice -management/coding/coding-library/financial-support-for-physicians-and-practices-during-the-covid-19-pandemic. Accessed July 23, 2020.
- American Medical Association. HEALS Act: What physicians and medical students need to know. https://www.ama-assn.org/delivering-care/public-health/heals-act -what-physicians-and-medical-students-need-know. Accessed July 29, 2020.
- Centers for Disease Control and Prevention. Interim guidance for businesses and employers responding to coronavirus disease 2019 (COVID-19). https://www.cdc.gov /coronavirus/2019-ncov/community/guidance-business-response.html. Accessed July 12, 2020.
- Centers for Disease Control and Prevention. Criteria for return to work for healthcare personnel with SARS-CoV-2 infection (interim guidance). https://www.cdc.gov /coronavirus/2019-ncov/hcp/return-to-work.html. Accessed July 20, 2020.
- US Office of Personnel Management. Questions and answers on human resources flexibilities and authorities for coronavirus disease 2019 (COVID-19). https://www .opm.gov/policy-data-oversight/covid-19/questions-and-answers-on-human-resources-flexibilities-and-authorities-for-coronavirus-disease-2019-covid-19.pdf. Accessed July 3, 2020.
- Centers for Disease Control and Prevention. Strategies to mitigate healthcare personnel staffing shortages. https://www.cdc.gov/coronavirus/2019-ncov/hcp /mitigating-staff-shortages.html. Accessed July 17, 2020.
- Centers for Disease Control and Prevention. Strategies for optimizing the supply of facemasks. https://www.cdc.gov/coronavirus/2019-ncov/hcp/ppe-strategy/face -masks.html. Accessed July 3, 2020.
- Rahman S, Klebanoff J, Moawad G. Smoke evacuation in the age of COVID-19. Contemporary OB/GYN. July 2, 2020. https://www.contemporaryobgyn.net/view/smoke -evacuation-in-the-age-of-covid-19. Accessed July 3, 2020.
- Centers for Disease Control and Prevention. Symptoms of coronavirus. https://www.cdc.gov/coronavirus/2019-ncov/symptoms-testing/symptoms.html. Accessed July 25, 2020.
- Centers for Disease Control and Prevention. Information for healthcare professionals about coronavirus (COVID-19). https://www.cdc.gov/coronavirus/2019-nCoV/hcp/index.html. Accessed July 22, 2020.
- US Department of Labor Occupational Safety and Health Administration. COVID-19 control and prevention: healthcare workers and employers. https://www.osha.gov/SLTC/covid-19/healthcare-workers.html. Accessed August 5, 2020.
- Centers for Medicare and Medicaid Services. COVID-19 resources for health care professionals and researchers. https://www.cms.gov/About-CMS/Agency-Information/OMH/resource-center/hcps-and-researchers. Accessed August 7, 2020.
- American College of Obstetricians and Gynecologists. COVID-19. https://www.acog.org/topics/covid-19. Accessed August 7, 2020.
- Society for Maternal-Fetal Medicine. Coronavirus (COVID-19). https://www.smfm.org/covid19. Accessed August 7, 2020.
- Society of Gynecologic Surgeons. Joint statement on re-introduction of hospital and office-based procedures in the COVID-19 climate for the practicing gynecologist. https://www.sgsonline.org/joint-statement-on-re-introduction-of-hospital-and-office-based-procedures-in-the-covid-19-climate. Accessed August 7, 2020.
- AAGL. COVID-19 articles, resources and webinars. https://www.aagl.org/covid-19/. Accessed August 7, 2020.
- American Society for Reproductive Medicine. COVID-19 updates and resources. https://www.asrm.org/news-and-publications/covid-19/. Accessed August 7, 2020.
- Society of Gynecologic Oncology. COVID-19 resources for health care practitioners. https://www.sgo.org/practice-management/covid-19/. Accessed August 7, 2020.
- Centers for Disease Control and Prevention. Using telehealth to expand access to essential health services during the COVID-19 pandemic. https://www.cdc.gov/coronavirus/2019-ncov/hcp/telehealth.html. Accessed July 3, 2020.
- Nguyen LH, Drew DA, Graham MS, et al; on behalf of the COronavirus Pandemic Epidemiology Consortium. Risk of COVID-19 among front-line health-care workers and the general community: a prospective cohort study. Lancet. July 31, 2020. http://www.thelancet-press.com/embargo/hcwcovid.pdf. Accessed August 25, 2020.
- Centers for Disease Control and Prevention. Criteria for return to work for healthcare personnel with SARS-CoV-2 infection (interim guidance). https://www.cdc.gov/coronavirus/2019-ncov/hcp/return-to-work.html. Accessed July 20, 2020.
- US Environmental Protection Agency. List N: Disinfectants for use against SARS-CoV-2 (COVID-19). https://www.epa.gov/pesticide-registration/list-n-disinfectants-use-against-sars-cov-2-covid-19. Accessed August 24, 2020.
- Centers for Disease Control and Prevention. Duration of isolation and precautions for adults with COVID-19. https://www.cdc.gov/coronavirus/2019-ncov/hcp/duration-isolation.html. Accessed July 23, 2020.
- Centers for Disease Control and Prevention. Discontinuation of transmission-based precautions and disposition of patients with COVID-19 in healthcare settings (interim guidance). https://www.cdc.gov/coronavirus/2019-ncov/hcp/disposition-hospitalized-patients.html#fn1. Accessed July 23, 2020.
- Centers for Disease Control and Prevention. Information for healthcare professionals about coronavirus (COVID-19). https://www.cdc.gov/coronavirus/2019-nCoV/hcp/index.html. Accessed July 22, 2020.
- US Department of Labor Occupational Safety and Health Administration. COVID-19 control and prevention: healthcare workers and employers. https://www.osha.gov/SLTC/covid-19/healthcare-workers.html. Accessed August 5, 2020.
- Centers for Medicare and Medicaid Services. COVID-19 resources for health care professionals and researchers. https://www.cms.gov/About-CMS/Agency-Information/OMH/resource-center/hcps-and-researchers. Accessed August 7, 2020.
- American College of Obstetricians and Gynecologists. COVID-19. https://www.acog.org/topics/covid-19. Accessed August 7, 2020.
- Society for Maternal-Fetal Medicine. Coronavirus (COVID-19). https://www.smfm.org/covid19. Accessed August 7, 2020.
- Society of Gynecologic Surgeons. Joint statement on re-introduction of hospital and office-based procedures in the COVID-19 climate for the practicing gynecologist. https://www.sgsonline.org/joint-statement-on-re-introduction-of-hospital-and-office-based-procedures-in-the-covid-19-climate. Accessed August 7, 2020.
- AAGL. COVID-19 articles, resources and webinars. https://www.aagl.org/covid-19/. Accessed August 7, 2020.
- American Society for Reproductive Medicine. COVID-19 updates and resources. https://www.asrm.org/news-and-publications/covid-19/. Accessed August 7, 2020.
- Society of Gynecologic Oncology. COVID-19 resources for health care practitioners. https://www.sgo.org/practice-management/covid-19/. Accessed August 7, 2020.
- Centers for Disease Control and Prevention. Using telehealth to expand access to essential health services during the COVID-19 pandemic. https://www.cdc.gov/coronavirus/2019-ncov/hcp/telehealth.html. Accessed July 3, 2020.
- Nguyen LH, Drew DA, Graham MS, et al; on behalf of the COronavirus Pandemic Epidemiology Consortium. Risk of COVID-19 among front-line health-care workers and the general community: a prospective cohort study. Lancet. July 31, 2020. http://www.thelancet-press.com/embargo/hcwcovid.pdf. Accessed August 25, 2020.
- Centers for Disease Control and Prevention. Criteria for return to work for healthcare personnel with SARS-CoV-2 infection (interim guidance). https://www.cdc.gov/coronavirus/2019-ncov/hcp/return-to-work.html. Accessed July 20, 2020.
- US Environmental Protection Agency. List N: Disinfectants for use against SARS-CoV-2 (COVID-19). https://www.epa.gov/pesticide-registration/list-n-disinfectants-use-against-sars-cov-2-covid-19. Accessed August 24, 2020.
- Centers for Disease Control and Prevention. Duration of isolation and precautions for adults with COVID-19. https://www.cdc.gov/coronavirus/2019-ncov/hcp/duration-isolation.html. Accessed July 23, 2020.
- Centers for Disease Control and Prevention. Discontinuation of transmission-based precautions and disposition of patients with COVID-19 in healthcare settings (interim guidance). https://www.cdc.gov/coronavirus/2019-ncov/hcp/disposition-hospitalized-patients.html#fn1. Accessed July 23, 2020.
Lifting the restrictions on mifepristone during COVID-19: A step in the right direction
Mifepristone is a safe, effective, and well-tolerated medication for managing miscarriage and for medical abortion when combined with misoprostol.1,2 Since the US Food and Drug Administration (FDA) approved its use in 2000, more than 4 million women have used this medication.3 The combination of mifepristone with misoprostol was used for 39% of all US abortions in 2017.4 Approximately 10% of all clinically recognized pregnancies end in miscarriages, and many are safely managed with either misoprostol alone or with the combination of mifepristone and misoprostol.5
The issue
The prescription and distribution of mifepristone is highly regulated by the FDA via requirements outlined in the Risk Evaluation and Mitigation Strategies (REMS) drug safety program. The FDA may determine a REMS is necessary for a specific drug to ensure the benefits of a drug outweigh the potential risks. A REMS may include an informative package insert for patients, follow-up communication to prescribers—including letters, safety protocols or recommended laboratory tests, or Elements to Assure Safe Use (ETASU). ETASU are types of REMS that are placed on medications that have significant potential for serious adverse effects, and without such restrictions FDA approval would be rescinded.
Are mifepristone requirements fairly applied?
The 3 ETASU restrictions on the distribution of mifepristone are in-person dispensation, prescriber certification, and patient signatures on special forms.6 The in-person dispensing requirement is applied to only 16 other medications (one of which is Mifeprex, the brand version of mifepristone), and Mifeprex/mifepristone are the only ones deemed safe for self-administration—meaning that patients receive the drug from a clinic but then may take it at a site of their choosing. The prescriber certification requirement places expectations on providers to account for distribution of doses and keep records of serial numbers (in effect, having clinicians act as both physician and pharmacist, as most medications are distributed and recorded in pharmacies). The patient form was recommended for elimination in 2016 due to its duplicative information and burden on patients—a recommendation that was then overruled by the FDA commissioner.7
These 3 requirements placed on mifepristone specifically target dosages for use related to abortions and miscarriages. Mifepristone is used to treat other medical conditions, with much higher doses, without the same restrictions—in fact, the FDA has allowed much higher doses of mifepristone to be mailed directly to a patient when prescribed for different disorders. The American College of Obstetricians and Gynecologists (ACOG) has long opposed the burdensome REMS requirements on mifepristone for reproductive health indications.8
Arguments regarding the safety of mifepristone must be understood in the context of how the medication is taken, and the unique difference with other medications that must be administered by physicians or in health care facilities. Mifepristone is self-administered, and the desired effect—evacuation of uterine contents—typically occurs after a patient takes the accompanying medication misoprostol, which is some 24 to 72 hours later. This timeframe makes it highly unlikely that any patient would be in the presence of their provider at the time of medication effect, thus an in-person dispensing requirement has no medical bearing on the outcome of the health of the patient.
REMS changes during the COVID-19 pandemic
The coronavirus disease 2019 (COVID-19) pandemic has necessarily changed the structure of REMS and ETASU requirements for many medications, with changes made in order to mitigate viral transmission through the limitation of unnecessary visits to clinics or hospitals. The FDA announced in March of 2020 that it would not enforce pre-prescription requirements, such as laboratory or magnetic resonance imaging results, for many medications (including those more toxic than mifepristone), and that it would lift the requirement for in-person dispensation of several medications.9 Also in March 2020 the Department of Health and Human Services Secretary (HHS) and the Drug Enforcement Agency (DEA) activated a “telemedicine exception” to allow physicians to use telemedicine to satisfy mandatory requirements for prescribing controlled substances, including opioids.10
Despite repeated pleas from organizations, individuals, and physician groups, the FDA continued to enforce the REMS/ETASU for mifepristone as the pandemic decimated communities. Importantly, the pandemic has not had an equal effect on all communities, and the disparities highlighted in outcomes as related to COVID-19 are also reflected in disparities to access to reproductive choices.11 By enforcing REMS/ETASU for mifepristone during a global pandemic, the FDA has placed additional burden on women and people who menstruate. As offices and clinics have closed, and as many jobs have evaporated, additional barriers have emerged, such as lack of childcare, fewer transportation options, and decreased clinic appointments.
As the pandemic continues to affect communities in the United States, ACOG has issued guidance recommending assessment for eligibility for medical abortion remotely, and has encouraged the use of telemedicine and other remote interactions for its members and patients to limit transmission of the virus.
The lawsuit
On May 27, 2020, the American Civil Liberties Union (ACLU) (on behalf of ACOG, the Council of University Chairs of Obstetrics and Gynecology, New York State Academy of Family Physicians, SisterSong, and Honor MacNaughton, MD) filed a civil action against the FDA and HHS challenging the requirement for in-person dispensing of mifepristone and associated ETASU requirements during the COVID-19 pandemic. The plaintiffs sought this injunction based on the claim that these restrictions during the pandemic infringe on the constitutional rights to patients’ privacy and liberty and to equal protection of the law as protected by the Due Process Clause of the Fifth Amendment. Additionally, the ACLU and other organizations said these unnecessary restrictions place patients, providers, and staff at unnecessary risk of viral exposure amidst a global pandemic.
The verdict
On July 13, 2020, a federal court granted the preliminary injunction to suspend FDA’s enforcement of the in-person requirements of mifepristone for abortion during the COVID-19 pandemic. The court denied the motion for suspension of in-person restrictions as applied to miscarriage management. The preliminary injunction applies nationwide without geographic limitation. It will remain in effect until the end of the litigation or for 30 days following the expiration of the public health emergency.
What the outcome means
This injunction is a step in the right direction for patients and providers to allow for autonomy and clinical practice guided by clinician expertise. However, this ruling remains narrow. Patients must be counseled about mifepristone via telemedicine and sign a Patient Agreement Form, which must be returned electronically or by mail. Patients must receive a copy of the mifepristone medication guide, and dispensing of mifepristone must still be conducted by or under the supervision of a certified provider. The medication may not be dispensed by retail pharmacies, thus requiring providers to arrange for mailing of prescriptions to patients. Given state-based legal statutes regarding mailing of medications, this injunction may not lead to an immediate increase in access to care. In addition, patients seeking management for miscarriage must go to clinic to have mifepristone dispensed and thus risk exposure to viral transmission.
What now?
The regulation of mifepristone—in spite of excellent safety and specifically for the narrow purpose of administration in the setting of abortion and miscarriage care—is by definition a discriminatory practice against patients and providers. As clinicians, we are duty-bound to speak out against injustices to our practices and our patients. At a local level, we can work to implement safe practices in the setting of this injunction and continue to work on a national level to ensure this injunction becomes permanent and with more broad scope to eliminate all of the REMS requirements for mifepristone.
ACTION ITEMS
- Act locally! Are you an abortion provider? Contact your local ACLU (find them here) or lawyer in your area for assistance navigating the legal landscape to prescribe after this injunction.
- Act statewide! Press candidates in your state to stand up for science and data. Support legislative acts and bills that address combating discriminatory regulations.
- Act nationally! The President is responsible for appointing the Commissioner of the FDA and the Secretary of Health and Human Services (with Senate advice and consent). Who we elect matters. Seek out opportunities to become involved in increasing access to and awareness of voter registration and Election Day, and speak out against voter suppression. Make sure you are registered to vote here and check your area to review new recommendations amidst the pandemic.
- American College of Obstetricians and Gynecologists. Practice bulletin number 143: medical management of first trimester abortion. Obstet Gynecol. 2014;123:676-692.
- Schreiber CA, Crenin MD, Atrio J, et al. Mifepristone pretreatment for the medical management of early pregnancy loss. N Engl J Med. 2018;378:2161-2170.
- Danco Laboratories. Mifeprex effectiveness and advantages. https://www.earlyoptionpill.com/is-mifeprex-right-for-me/effectiveness-advantages/ Accessed August 2, 2020.
- Jones RK, Witwer E, Jerman J. Abortion incidence and service availability in the United States, 2017. September 2019. https://www.guttmacher.org/report/abortion-incidence-service-availability-us-2017. Accessed September 10, 2020.
- American College of Obstetricians and Gynecologists. Practice bulletin no. 150: early pregnancy loss. Obstet Gynecol. 2015;125:1258-1267.
- US Food and Drug Administration. Risk evaluation and mitigation strategy (REMS) single shared system for mifepristone 200 mg. April 2019. https://www.accessdata.fda.gov/drugsatfda_docs/rems/Mifepristone_2019_04_11_REMS_Full.pdf. Accessed September 10, 2020.
- US Food and Drug Administration; Center for Drug Evaluation and Research. 2016 REMS Review, Summary Review 25. March 29, 2016. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2016/020687Orig1s020SumR.pdf. Accessed September 10, 2020.
- American College of Obstetricians and Gynecologists. Improving access to mifepristone for reproductive health indications. June 2018. https://www.acog.org/clinical-information/policy-and-position-statements/position-statements/2018/improving-access-to-mifepristone-for-reproductive-health-indications. Accessed August 2, 2020.
- US Food and Drug Administration. Policy for certain REMS requirements during the COVID-19 public health emergency: guidance for industry and health care professionals. March 2020. https://www.fda.gov/media/136317/download. Accessed September 10, 2020.
- US Department of Justice. US Drug Enforcement Administration. COVID-19 Information Page, Telemedicine. https://www.deadiversion.usdoj.gov/coronavirus.html#TELE. Accessed May 25, 2020.
- Centers for Disease Control and Prevention. Coronavirus disease 2019: health equity considerations and racial and ethnic minority groups. https://www.cdc.gov/coronavirus/2019-ncov/community/health-equity/race-ethnicity.html. Updated July 24, 2020. Accessed September 10, 2020.
Mifepristone is a safe, effective, and well-tolerated medication for managing miscarriage and for medical abortion when combined with misoprostol.1,2 Since the US Food and Drug Administration (FDA) approved its use in 2000, more than 4 million women have used this medication.3 The combination of mifepristone with misoprostol was used for 39% of all US abortions in 2017.4 Approximately 10% of all clinically recognized pregnancies end in miscarriages, and many are safely managed with either misoprostol alone or with the combination of mifepristone and misoprostol.5
The issue
The prescription and distribution of mifepristone is highly regulated by the FDA via requirements outlined in the Risk Evaluation and Mitigation Strategies (REMS) drug safety program. The FDA may determine a REMS is necessary for a specific drug to ensure the benefits of a drug outweigh the potential risks. A REMS may include an informative package insert for patients, follow-up communication to prescribers—including letters, safety protocols or recommended laboratory tests, or Elements to Assure Safe Use (ETASU). ETASU are types of REMS that are placed on medications that have significant potential for serious adverse effects, and without such restrictions FDA approval would be rescinded.
Are mifepristone requirements fairly applied?
The 3 ETASU restrictions on the distribution of mifepristone are in-person dispensation, prescriber certification, and patient signatures on special forms.6 The in-person dispensing requirement is applied to only 16 other medications (one of which is Mifeprex, the brand version of mifepristone), and Mifeprex/mifepristone are the only ones deemed safe for self-administration—meaning that patients receive the drug from a clinic but then may take it at a site of their choosing. The prescriber certification requirement places expectations on providers to account for distribution of doses and keep records of serial numbers (in effect, having clinicians act as both physician and pharmacist, as most medications are distributed and recorded in pharmacies). The patient form was recommended for elimination in 2016 due to its duplicative information and burden on patients—a recommendation that was then overruled by the FDA commissioner.7
These 3 requirements placed on mifepristone specifically target dosages for use related to abortions and miscarriages. Mifepristone is used to treat other medical conditions, with much higher doses, without the same restrictions—in fact, the FDA has allowed much higher doses of mifepristone to be mailed directly to a patient when prescribed for different disorders. The American College of Obstetricians and Gynecologists (ACOG) has long opposed the burdensome REMS requirements on mifepristone for reproductive health indications.8
Arguments regarding the safety of mifepristone must be understood in the context of how the medication is taken, and the unique difference with other medications that must be administered by physicians or in health care facilities. Mifepristone is self-administered, and the desired effect—evacuation of uterine contents—typically occurs after a patient takes the accompanying medication misoprostol, which is some 24 to 72 hours later. This timeframe makes it highly unlikely that any patient would be in the presence of their provider at the time of medication effect, thus an in-person dispensing requirement has no medical bearing on the outcome of the health of the patient.
REMS changes during the COVID-19 pandemic
The coronavirus disease 2019 (COVID-19) pandemic has necessarily changed the structure of REMS and ETASU requirements for many medications, with changes made in order to mitigate viral transmission through the limitation of unnecessary visits to clinics or hospitals. The FDA announced in March of 2020 that it would not enforce pre-prescription requirements, such as laboratory or magnetic resonance imaging results, for many medications (including those more toxic than mifepristone), and that it would lift the requirement for in-person dispensation of several medications.9 Also in March 2020 the Department of Health and Human Services Secretary (HHS) and the Drug Enforcement Agency (DEA) activated a “telemedicine exception” to allow physicians to use telemedicine to satisfy mandatory requirements for prescribing controlled substances, including opioids.10
Despite repeated pleas from organizations, individuals, and physician groups, the FDA continued to enforce the REMS/ETASU for mifepristone as the pandemic decimated communities. Importantly, the pandemic has not had an equal effect on all communities, and the disparities highlighted in outcomes as related to COVID-19 are also reflected in disparities to access to reproductive choices.11 By enforcing REMS/ETASU for mifepristone during a global pandemic, the FDA has placed additional burden on women and people who menstruate. As offices and clinics have closed, and as many jobs have evaporated, additional barriers have emerged, such as lack of childcare, fewer transportation options, and decreased clinic appointments.
As the pandemic continues to affect communities in the United States, ACOG has issued guidance recommending assessment for eligibility for medical abortion remotely, and has encouraged the use of telemedicine and other remote interactions for its members and patients to limit transmission of the virus.
The lawsuit
On May 27, 2020, the American Civil Liberties Union (ACLU) (on behalf of ACOG, the Council of University Chairs of Obstetrics and Gynecology, New York State Academy of Family Physicians, SisterSong, and Honor MacNaughton, MD) filed a civil action against the FDA and HHS challenging the requirement for in-person dispensing of mifepristone and associated ETASU requirements during the COVID-19 pandemic. The plaintiffs sought this injunction based on the claim that these restrictions during the pandemic infringe on the constitutional rights to patients’ privacy and liberty and to equal protection of the law as protected by the Due Process Clause of the Fifth Amendment. Additionally, the ACLU and other organizations said these unnecessary restrictions place patients, providers, and staff at unnecessary risk of viral exposure amidst a global pandemic.
The verdict
On July 13, 2020, a federal court granted the preliminary injunction to suspend FDA’s enforcement of the in-person requirements of mifepristone for abortion during the COVID-19 pandemic. The court denied the motion for suspension of in-person restrictions as applied to miscarriage management. The preliminary injunction applies nationwide without geographic limitation. It will remain in effect until the end of the litigation or for 30 days following the expiration of the public health emergency.
What the outcome means
This injunction is a step in the right direction for patients and providers to allow for autonomy and clinical practice guided by clinician expertise. However, this ruling remains narrow. Patients must be counseled about mifepristone via telemedicine and sign a Patient Agreement Form, which must be returned electronically or by mail. Patients must receive a copy of the mifepristone medication guide, and dispensing of mifepristone must still be conducted by or under the supervision of a certified provider. The medication may not be dispensed by retail pharmacies, thus requiring providers to arrange for mailing of prescriptions to patients. Given state-based legal statutes regarding mailing of medications, this injunction may not lead to an immediate increase in access to care. In addition, patients seeking management for miscarriage must go to clinic to have mifepristone dispensed and thus risk exposure to viral transmission.
What now?
The regulation of mifepristone—in spite of excellent safety and specifically for the narrow purpose of administration in the setting of abortion and miscarriage care—is by definition a discriminatory practice against patients and providers. As clinicians, we are duty-bound to speak out against injustices to our practices and our patients. At a local level, we can work to implement safe practices in the setting of this injunction and continue to work on a national level to ensure this injunction becomes permanent and with more broad scope to eliminate all of the REMS requirements for mifepristone.
ACTION ITEMS
- Act locally! Are you an abortion provider? Contact your local ACLU (find them here) or lawyer in your area for assistance navigating the legal landscape to prescribe after this injunction.
- Act statewide! Press candidates in your state to stand up for science and data. Support legislative acts and bills that address combating discriminatory regulations.
- Act nationally! The President is responsible for appointing the Commissioner of the FDA and the Secretary of Health and Human Services (with Senate advice and consent). Who we elect matters. Seek out opportunities to become involved in increasing access to and awareness of voter registration and Election Day, and speak out against voter suppression. Make sure you are registered to vote here and check your area to review new recommendations amidst the pandemic.
Mifepristone is a safe, effective, and well-tolerated medication for managing miscarriage and for medical abortion when combined with misoprostol.1,2 Since the US Food and Drug Administration (FDA) approved its use in 2000, more than 4 million women have used this medication.3 The combination of mifepristone with misoprostol was used for 39% of all US abortions in 2017.4 Approximately 10% of all clinically recognized pregnancies end in miscarriages, and many are safely managed with either misoprostol alone or with the combination of mifepristone and misoprostol.5
The issue
The prescription and distribution of mifepristone is highly regulated by the FDA via requirements outlined in the Risk Evaluation and Mitigation Strategies (REMS) drug safety program. The FDA may determine a REMS is necessary for a specific drug to ensure the benefits of a drug outweigh the potential risks. A REMS may include an informative package insert for patients, follow-up communication to prescribers—including letters, safety protocols or recommended laboratory tests, or Elements to Assure Safe Use (ETASU). ETASU are types of REMS that are placed on medications that have significant potential for serious adverse effects, and without such restrictions FDA approval would be rescinded.
Are mifepristone requirements fairly applied?
The 3 ETASU restrictions on the distribution of mifepristone are in-person dispensation, prescriber certification, and patient signatures on special forms.6 The in-person dispensing requirement is applied to only 16 other medications (one of which is Mifeprex, the brand version of mifepristone), and Mifeprex/mifepristone are the only ones deemed safe for self-administration—meaning that patients receive the drug from a clinic but then may take it at a site of their choosing. The prescriber certification requirement places expectations on providers to account for distribution of doses and keep records of serial numbers (in effect, having clinicians act as both physician and pharmacist, as most medications are distributed and recorded in pharmacies). The patient form was recommended for elimination in 2016 due to its duplicative information and burden on patients—a recommendation that was then overruled by the FDA commissioner.7
These 3 requirements placed on mifepristone specifically target dosages for use related to abortions and miscarriages. Mifepristone is used to treat other medical conditions, with much higher doses, without the same restrictions—in fact, the FDA has allowed much higher doses of mifepristone to be mailed directly to a patient when prescribed for different disorders. The American College of Obstetricians and Gynecologists (ACOG) has long opposed the burdensome REMS requirements on mifepristone for reproductive health indications.8
Arguments regarding the safety of mifepristone must be understood in the context of how the medication is taken, and the unique difference with other medications that must be administered by physicians or in health care facilities. Mifepristone is self-administered, and the desired effect—evacuation of uterine contents—typically occurs after a patient takes the accompanying medication misoprostol, which is some 24 to 72 hours later. This timeframe makes it highly unlikely that any patient would be in the presence of their provider at the time of medication effect, thus an in-person dispensing requirement has no medical bearing on the outcome of the health of the patient.
REMS changes during the COVID-19 pandemic
The coronavirus disease 2019 (COVID-19) pandemic has necessarily changed the structure of REMS and ETASU requirements for many medications, with changes made in order to mitigate viral transmission through the limitation of unnecessary visits to clinics or hospitals. The FDA announced in March of 2020 that it would not enforce pre-prescription requirements, such as laboratory or magnetic resonance imaging results, for many medications (including those more toxic than mifepristone), and that it would lift the requirement for in-person dispensation of several medications.9 Also in March 2020 the Department of Health and Human Services Secretary (HHS) and the Drug Enforcement Agency (DEA) activated a “telemedicine exception” to allow physicians to use telemedicine to satisfy mandatory requirements for prescribing controlled substances, including opioids.10
Despite repeated pleas from organizations, individuals, and physician groups, the FDA continued to enforce the REMS/ETASU for mifepristone as the pandemic decimated communities. Importantly, the pandemic has not had an equal effect on all communities, and the disparities highlighted in outcomes as related to COVID-19 are also reflected in disparities to access to reproductive choices.11 By enforcing REMS/ETASU for mifepristone during a global pandemic, the FDA has placed additional burden on women and people who menstruate. As offices and clinics have closed, and as many jobs have evaporated, additional barriers have emerged, such as lack of childcare, fewer transportation options, and decreased clinic appointments.
As the pandemic continues to affect communities in the United States, ACOG has issued guidance recommending assessment for eligibility for medical abortion remotely, and has encouraged the use of telemedicine and other remote interactions for its members and patients to limit transmission of the virus.
The lawsuit
On May 27, 2020, the American Civil Liberties Union (ACLU) (on behalf of ACOG, the Council of University Chairs of Obstetrics and Gynecology, New York State Academy of Family Physicians, SisterSong, and Honor MacNaughton, MD) filed a civil action against the FDA and HHS challenging the requirement for in-person dispensing of mifepristone and associated ETASU requirements during the COVID-19 pandemic. The plaintiffs sought this injunction based on the claim that these restrictions during the pandemic infringe on the constitutional rights to patients’ privacy and liberty and to equal protection of the law as protected by the Due Process Clause of the Fifth Amendment. Additionally, the ACLU and other organizations said these unnecessary restrictions place patients, providers, and staff at unnecessary risk of viral exposure amidst a global pandemic.
The verdict
On July 13, 2020, a federal court granted the preliminary injunction to suspend FDA’s enforcement of the in-person requirements of mifepristone for abortion during the COVID-19 pandemic. The court denied the motion for suspension of in-person restrictions as applied to miscarriage management. The preliminary injunction applies nationwide without geographic limitation. It will remain in effect until the end of the litigation or for 30 days following the expiration of the public health emergency.
What the outcome means
This injunction is a step in the right direction for patients and providers to allow for autonomy and clinical practice guided by clinician expertise. However, this ruling remains narrow. Patients must be counseled about mifepristone via telemedicine and sign a Patient Agreement Form, which must be returned electronically or by mail. Patients must receive a copy of the mifepristone medication guide, and dispensing of mifepristone must still be conducted by or under the supervision of a certified provider. The medication may not be dispensed by retail pharmacies, thus requiring providers to arrange for mailing of prescriptions to patients. Given state-based legal statutes regarding mailing of medications, this injunction may not lead to an immediate increase in access to care. In addition, patients seeking management for miscarriage must go to clinic to have mifepristone dispensed and thus risk exposure to viral transmission.
What now?
The regulation of mifepristone—in spite of excellent safety and specifically for the narrow purpose of administration in the setting of abortion and miscarriage care—is by definition a discriminatory practice against patients and providers. As clinicians, we are duty-bound to speak out against injustices to our practices and our patients. At a local level, we can work to implement safe practices in the setting of this injunction and continue to work on a national level to ensure this injunction becomes permanent and with more broad scope to eliminate all of the REMS requirements for mifepristone.
ACTION ITEMS
- Act locally! Are you an abortion provider? Contact your local ACLU (find them here) or lawyer in your area for assistance navigating the legal landscape to prescribe after this injunction.
- Act statewide! Press candidates in your state to stand up for science and data. Support legislative acts and bills that address combating discriminatory regulations.
- Act nationally! The President is responsible for appointing the Commissioner of the FDA and the Secretary of Health and Human Services (with Senate advice and consent). Who we elect matters. Seek out opportunities to become involved in increasing access to and awareness of voter registration and Election Day, and speak out against voter suppression. Make sure you are registered to vote here and check your area to review new recommendations amidst the pandemic.
- American College of Obstetricians and Gynecologists. Practice bulletin number 143: medical management of first trimester abortion. Obstet Gynecol. 2014;123:676-692.
- Schreiber CA, Crenin MD, Atrio J, et al. Mifepristone pretreatment for the medical management of early pregnancy loss. N Engl J Med. 2018;378:2161-2170.
- Danco Laboratories. Mifeprex effectiveness and advantages. https://www.earlyoptionpill.com/is-mifeprex-right-for-me/effectiveness-advantages/ Accessed August 2, 2020.
- Jones RK, Witwer E, Jerman J. Abortion incidence and service availability in the United States, 2017. September 2019. https://www.guttmacher.org/report/abortion-incidence-service-availability-us-2017. Accessed September 10, 2020.
- American College of Obstetricians and Gynecologists. Practice bulletin no. 150: early pregnancy loss. Obstet Gynecol. 2015;125:1258-1267.
- US Food and Drug Administration. Risk evaluation and mitigation strategy (REMS) single shared system for mifepristone 200 mg. April 2019. https://www.accessdata.fda.gov/drugsatfda_docs/rems/Mifepristone_2019_04_11_REMS_Full.pdf. Accessed September 10, 2020.
- US Food and Drug Administration; Center for Drug Evaluation and Research. 2016 REMS Review, Summary Review 25. March 29, 2016. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2016/020687Orig1s020SumR.pdf. Accessed September 10, 2020.
- American College of Obstetricians and Gynecologists. Improving access to mifepristone for reproductive health indications. June 2018. https://www.acog.org/clinical-information/policy-and-position-statements/position-statements/2018/improving-access-to-mifepristone-for-reproductive-health-indications. Accessed August 2, 2020.
- US Food and Drug Administration. Policy for certain REMS requirements during the COVID-19 public health emergency: guidance for industry and health care professionals. March 2020. https://www.fda.gov/media/136317/download. Accessed September 10, 2020.
- US Department of Justice. US Drug Enforcement Administration. COVID-19 Information Page, Telemedicine. https://www.deadiversion.usdoj.gov/coronavirus.html#TELE. Accessed May 25, 2020.
- Centers for Disease Control and Prevention. Coronavirus disease 2019: health equity considerations and racial and ethnic minority groups. https://www.cdc.gov/coronavirus/2019-ncov/community/health-equity/race-ethnicity.html. Updated July 24, 2020. Accessed September 10, 2020.
- American College of Obstetricians and Gynecologists. Practice bulletin number 143: medical management of first trimester abortion. Obstet Gynecol. 2014;123:676-692.
- Schreiber CA, Crenin MD, Atrio J, et al. Mifepristone pretreatment for the medical management of early pregnancy loss. N Engl J Med. 2018;378:2161-2170.
- Danco Laboratories. Mifeprex effectiveness and advantages. https://www.earlyoptionpill.com/is-mifeprex-right-for-me/effectiveness-advantages/ Accessed August 2, 2020.
- Jones RK, Witwer E, Jerman J. Abortion incidence and service availability in the United States, 2017. September 2019. https://www.guttmacher.org/report/abortion-incidence-service-availability-us-2017. Accessed September 10, 2020.
- American College of Obstetricians and Gynecologists. Practice bulletin no. 150: early pregnancy loss. Obstet Gynecol. 2015;125:1258-1267.
- US Food and Drug Administration. Risk evaluation and mitigation strategy (REMS) single shared system for mifepristone 200 mg. April 2019. https://www.accessdata.fda.gov/drugsatfda_docs/rems/Mifepristone_2019_04_11_REMS_Full.pdf. Accessed September 10, 2020.
- US Food and Drug Administration; Center for Drug Evaluation and Research. 2016 REMS Review, Summary Review 25. March 29, 2016. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2016/020687Orig1s020SumR.pdf. Accessed September 10, 2020.
- American College of Obstetricians and Gynecologists. Improving access to mifepristone for reproductive health indications. June 2018. https://www.acog.org/clinical-information/policy-and-position-statements/position-statements/2018/improving-access-to-mifepristone-for-reproductive-health-indications. Accessed August 2, 2020.
- US Food and Drug Administration. Policy for certain REMS requirements during the COVID-19 public health emergency: guidance for industry and health care professionals. March 2020. https://www.fda.gov/media/136317/download. Accessed September 10, 2020.
- US Department of Justice. US Drug Enforcement Administration. COVID-19 Information Page, Telemedicine. https://www.deadiversion.usdoj.gov/coronavirus.html#TELE. Accessed May 25, 2020.
- Centers for Disease Control and Prevention. Coronavirus disease 2019: health equity considerations and racial and ethnic minority groups. https://www.cdc.gov/coronavirus/2019-ncov/community/health-equity/race-ethnicity.html. Updated July 24, 2020. Accessed September 10, 2020.
COVID-19: New guidance to stem mental health crisis in frontline HCPs
A new review offers fresh guidance to help stem the mental health toll of the COVID-19 pandemic on frontline clinicians.
Investigators gathered practice guidelines and resources from a wide range of health care organizations and professional societies to develop a conceptual framework of mental health support for health care professionals (HCPs) caring for COVID-19 patients.
“Support needs to be deployed in multiple dimensions – including individual, organizational, and societal levels – and include training in resilience, stress reduction, emotional awareness, and self-care strategies,” lead author Rachel Schwartz, PhD, health services researcher, Stanford (Calif.) University, said in an interview.
The review was published Aug. 21 in the Annals of Internal Medicine.
An opportune moment
Coauthor Rebecca Margolis, DO, director of well-being in the division of medical education and faculty development, Children’s Hospital of Los Angeles, said that this is “an opportune moment to look at how we treat frontline providers in this country.”
Studies of previous pandemics have shown heightened distress in HCPs, even years after the pandemic, and the unique challenges posed by the COVID-19 pandemic surpass those of previous pandemics, Dr. Margolis said in an interview.
Dr. Schwartz, Dr. Margolis, and coauthors Uma Anand, PhD, LP, and Jina Sinskey, MD, met through the Collaborative for Healing and Renewal in Medicine network, a group of medical educators, leaders in academic medicine, experts in burnout research and interventions, and trainees working together to promote well-being among trainees and practicing physicians.
“We were brought together on a conference call in March, when things were particularly bad in New York, and started looking to see what resources we could get to frontline providers who were suffering. It was great to lean on each other and stand on the shoulders of colleagues in New York, who were the ones we learned from on these calls,” said Dr. Margolis.
The authors recommended addressing clinicians’ basic practical needs, including ensuring essentials like meals and transportation, establishing a “well-being area” within hospitals for staff to rest, and providing well-stocked living quarters so clinicians can safely quarantine from family, as well as personal protective equipment and child care.
Clinicians are often asked to “assume new professional roles to meet evolving needs” during a pandemic, which can increase stress. The authors recommended targeted training, assessment of clinician skills before redeployment to a new clinical role, and clear communication practices around redeployment.
Recognition from hospital and government leaders improves morale and supports clinicians’ ability to continue delivering care. Leadership should “leverage communication strategies to provide clinicians with up-to-date information and reassurance,” they wrote.
‘Uniquely isolated’
Dr. Margolis noted that
“My colleagues feel a sense of moral injury, putting their lives on the line at work, performing the most perilous job, and their kids can’t hang out with other kids, which just puts salt on the wound,” she said.
Additional sources of moral injury are deciding which patients should receive life support in the event of inadequate resources and bearing witness to, or enforcing, policies that lead to patients dying alone.
Leaders should encourage clinicians to “seek informal support from colleagues, managers, or chaplains” and to “provide rapid access to professional help,” the authors noted.
Furthermore, they contended that leaders should “proactively and routinely monitor the psychological well-being of their teams,” since guilt and shame often prevent clinicians from disclosing feelings of moral injury.
“Being provided with routine mental health support should be normalized and it should be part of the job – not only during COVID-19 but in general,” Dr. Schwartz said.
‘Battle buddies’
Dr. Margolis recommended the “battle buddy” model for mutual peer support.
Dr. Anand, a mental health clinician at Mayo Medical School, Rochester, Minn., elaborated.
“We connect residents with each other, and they form pairs to support each other and watch for warning signs such as withdrawal from colleagues, being frequently tearful, not showing up at work or showing up late, missing assignments, making mistakes at work, increased use of alcohol, or verbalizing serious concerns,” Dr. Anand said.
If the buddy shows any of these warning signs, he or she can be directed to appropriate resources to get help.
Since the pandemic has interfered with the ability to connect with colleagues and family members, attention should be paid to addressing the social support needs of clinicians.
Dr. Anand suggested that clinicians maintain contact with counselors, friends, and family, even if they cannot be together in person and must connect “virtually.”
Resilience and strength training are “key” components of reducing clinician distress, but trainings as well as processing groups and support workshops should be offered during protected time, Dr. Margolis advised, since it can be burdensome for clinicians to wake up early or stay late to attend these sessions.
Leaders and administrators should “model self-care and well-being,” she noted. For example, sending emails to clinicians late at night or on weekends creates an expectation of a rapid reply, which leads to additional pressure for the clinician.
“This is of the most powerful unspoken curricula we can develop,” Dr. Margolis emphasized.
Self-care critical
Marcus S. Shaker, MD, MSc, associate professor of pediatrics, medicine, and community and family medicine, Children’s Hospital at Dartmouth-Hitchcock in Lebanon, N.H., and Geisel School of Medicine at Dartmouth, Hanover, N.H., said the study was “a much appreciated, timely reminder of the importance of clinician wellness.”
Moreover, “without self-care, our ability to help our patients withers. This article provides a useful conceptual framework for individuals and organizations to provide the right care at the right time in these unprecedented times,” said Dr. Shaker, who was not involved with the study.
The authors agreed, stating that clinicians “require proactive psychological protection specifically because they are a population known for putting others’ needs before their own.”
They recommended several resources for HCPs, including the Physician Support Line; Headspace, a mindfulness Web-based app for reducing stress and anxiety; the National Suicide Prevention Lifeline; and the Crisis Text Line.
The authors and Dr. Shaker disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
A new review offers fresh guidance to help stem the mental health toll of the COVID-19 pandemic on frontline clinicians.
Investigators gathered practice guidelines and resources from a wide range of health care organizations and professional societies to develop a conceptual framework of mental health support for health care professionals (HCPs) caring for COVID-19 patients.
“Support needs to be deployed in multiple dimensions – including individual, organizational, and societal levels – and include training in resilience, stress reduction, emotional awareness, and self-care strategies,” lead author Rachel Schwartz, PhD, health services researcher, Stanford (Calif.) University, said in an interview.
The review was published Aug. 21 in the Annals of Internal Medicine.
An opportune moment
Coauthor Rebecca Margolis, DO, director of well-being in the division of medical education and faculty development, Children’s Hospital of Los Angeles, said that this is “an opportune moment to look at how we treat frontline providers in this country.”
Studies of previous pandemics have shown heightened distress in HCPs, even years after the pandemic, and the unique challenges posed by the COVID-19 pandemic surpass those of previous pandemics, Dr. Margolis said in an interview.
Dr. Schwartz, Dr. Margolis, and coauthors Uma Anand, PhD, LP, and Jina Sinskey, MD, met through the Collaborative for Healing and Renewal in Medicine network, a group of medical educators, leaders in academic medicine, experts in burnout research and interventions, and trainees working together to promote well-being among trainees and practicing physicians.
“We were brought together on a conference call in March, when things were particularly bad in New York, and started looking to see what resources we could get to frontline providers who were suffering. It was great to lean on each other and stand on the shoulders of colleagues in New York, who were the ones we learned from on these calls,” said Dr. Margolis.
The authors recommended addressing clinicians’ basic practical needs, including ensuring essentials like meals and transportation, establishing a “well-being area” within hospitals for staff to rest, and providing well-stocked living quarters so clinicians can safely quarantine from family, as well as personal protective equipment and child care.
Clinicians are often asked to “assume new professional roles to meet evolving needs” during a pandemic, which can increase stress. The authors recommended targeted training, assessment of clinician skills before redeployment to a new clinical role, and clear communication practices around redeployment.
Recognition from hospital and government leaders improves morale and supports clinicians’ ability to continue delivering care. Leadership should “leverage communication strategies to provide clinicians with up-to-date information and reassurance,” they wrote.
‘Uniquely isolated’
Dr. Margolis noted that
“My colleagues feel a sense of moral injury, putting their lives on the line at work, performing the most perilous job, and their kids can’t hang out with other kids, which just puts salt on the wound,” she said.
Additional sources of moral injury are deciding which patients should receive life support in the event of inadequate resources and bearing witness to, or enforcing, policies that lead to patients dying alone.
Leaders should encourage clinicians to “seek informal support from colleagues, managers, or chaplains” and to “provide rapid access to professional help,” the authors noted.
Furthermore, they contended that leaders should “proactively and routinely monitor the psychological well-being of their teams,” since guilt and shame often prevent clinicians from disclosing feelings of moral injury.
“Being provided with routine mental health support should be normalized and it should be part of the job – not only during COVID-19 but in general,” Dr. Schwartz said.
‘Battle buddies’
Dr. Margolis recommended the “battle buddy” model for mutual peer support.
Dr. Anand, a mental health clinician at Mayo Medical School, Rochester, Minn., elaborated.
“We connect residents with each other, and they form pairs to support each other and watch for warning signs such as withdrawal from colleagues, being frequently tearful, not showing up at work or showing up late, missing assignments, making mistakes at work, increased use of alcohol, or verbalizing serious concerns,” Dr. Anand said.
If the buddy shows any of these warning signs, he or she can be directed to appropriate resources to get help.
Since the pandemic has interfered with the ability to connect with colleagues and family members, attention should be paid to addressing the social support needs of clinicians.
Dr. Anand suggested that clinicians maintain contact with counselors, friends, and family, even if they cannot be together in person and must connect “virtually.”
Resilience and strength training are “key” components of reducing clinician distress, but trainings as well as processing groups and support workshops should be offered during protected time, Dr. Margolis advised, since it can be burdensome for clinicians to wake up early or stay late to attend these sessions.
Leaders and administrators should “model self-care and well-being,” she noted. For example, sending emails to clinicians late at night or on weekends creates an expectation of a rapid reply, which leads to additional pressure for the clinician.
“This is of the most powerful unspoken curricula we can develop,” Dr. Margolis emphasized.
Self-care critical
Marcus S. Shaker, MD, MSc, associate professor of pediatrics, medicine, and community and family medicine, Children’s Hospital at Dartmouth-Hitchcock in Lebanon, N.H., and Geisel School of Medicine at Dartmouth, Hanover, N.H., said the study was “a much appreciated, timely reminder of the importance of clinician wellness.”
Moreover, “without self-care, our ability to help our patients withers. This article provides a useful conceptual framework for individuals and organizations to provide the right care at the right time in these unprecedented times,” said Dr. Shaker, who was not involved with the study.
The authors agreed, stating that clinicians “require proactive psychological protection specifically because they are a population known for putting others’ needs before their own.”
They recommended several resources for HCPs, including the Physician Support Line; Headspace, a mindfulness Web-based app for reducing stress and anxiety; the National Suicide Prevention Lifeline; and the Crisis Text Line.
The authors and Dr. Shaker disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
A new review offers fresh guidance to help stem the mental health toll of the COVID-19 pandemic on frontline clinicians.
Investigators gathered practice guidelines and resources from a wide range of health care organizations and professional societies to develop a conceptual framework of mental health support for health care professionals (HCPs) caring for COVID-19 patients.
“Support needs to be deployed in multiple dimensions – including individual, organizational, and societal levels – and include training in resilience, stress reduction, emotional awareness, and self-care strategies,” lead author Rachel Schwartz, PhD, health services researcher, Stanford (Calif.) University, said in an interview.
The review was published Aug. 21 in the Annals of Internal Medicine.
An opportune moment
Coauthor Rebecca Margolis, DO, director of well-being in the division of medical education and faculty development, Children’s Hospital of Los Angeles, said that this is “an opportune moment to look at how we treat frontline providers in this country.”
Studies of previous pandemics have shown heightened distress in HCPs, even years after the pandemic, and the unique challenges posed by the COVID-19 pandemic surpass those of previous pandemics, Dr. Margolis said in an interview.
Dr. Schwartz, Dr. Margolis, and coauthors Uma Anand, PhD, LP, and Jina Sinskey, MD, met through the Collaborative for Healing and Renewal in Medicine network, a group of medical educators, leaders in academic medicine, experts in burnout research and interventions, and trainees working together to promote well-being among trainees and practicing physicians.
“We were brought together on a conference call in March, when things were particularly bad in New York, and started looking to see what resources we could get to frontline providers who were suffering. It was great to lean on each other and stand on the shoulders of colleagues in New York, who were the ones we learned from on these calls,” said Dr. Margolis.
The authors recommended addressing clinicians’ basic practical needs, including ensuring essentials like meals and transportation, establishing a “well-being area” within hospitals for staff to rest, and providing well-stocked living quarters so clinicians can safely quarantine from family, as well as personal protective equipment and child care.
Clinicians are often asked to “assume new professional roles to meet evolving needs” during a pandemic, which can increase stress. The authors recommended targeted training, assessment of clinician skills before redeployment to a new clinical role, and clear communication practices around redeployment.
Recognition from hospital and government leaders improves morale and supports clinicians’ ability to continue delivering care. Leadership should “leverage communication strategies to provide clinicians with up-to-date information and reassurance,” they wrote.
‘Uniquely isolated’
Dr. Margolis noted that
“My colleagues feel a sense of moral injury, putting their lives on the line at work, performing the most perilous job, and their kids can’t hang out with other kids, which just puts salt on the wound,” she said.
Additional sources of moral injury are deciding which patients should receive life support in the event of inadequate resources and bearing witness to, or enforcing, policies that lead to patients dying alone.
Leaders should encourage clinicians to “seek informal support from colleagues, managers, or chaplains” and to “provide rapid access to professional help,” the authors noted.
Furthermore, they contended that leaders should “proactively and routinely monitor the psychological well-being of their teams,” since guilt and shame often prevent clinicians from disclosing feelings of moral injury.
“Being provided with routine mental health support should be normalized and it should be part of the job – not only during COVID-19 but in general,” Dr. Schwartz said.
‘Battle buddies’
Dr. Margolis recommended the “battle buddy” model for mutual peer support.
Dr. Anand, a mental health clinician at Mayo Medical School, Rochester, Minn., elaborated.
“We connect residents with each other, and they form pairs to support each other and watch for warning signs such as withdrawal from colleagues, being frequently tearful, not showing up at work or showing up late, missing assignments, making mistakes at work, increased use of alcohol, or verbalizing serious concerns,” Dr. Anand said.
If the buddy shows any of these warning signs, he or she can be directed to appropriate resources to get help.
Since the pandemic has interfered with the ability to connect with colleagues and family members, attention should be paid to addressing the social support needs of clinicians.
Dr. Anand suggested that clinicians maintain contact with counselors, friends, and family, even if they cannot be together in person and must connect “virtually.”
Resilience and strength training are “key” components of reducing clinician distress, but trainings as well as processing groups and support workshops should be offered during protected time, Dr. Margolis advised, since it can be burdensome for clinicians to wake up early or stay late to attend these sessions.
Leaders and administrators should “model self-care and well-being,” she noted. For example, sending emails to clinicians late at night or on weekends creates an expectation of a rapid reply, which leads to additional pressure for the clinician.
“This is of the most powerful unspoken curricula we can develop,” Dr. Margolis emphasized.
Self-care critical
Marcus S. Shaker, MD, MSc, associate professor of pediatrics, medicine, and community and family medicine, Children’s Hospital at Dartmouth-Hitchcock in Lebanon, N.H., and Geisel School of Medicine at Dartmouth, Hanover, N.H., said the study was “a much appreciated, timely reminder of the importance of clinician wellness.”
Moreover, “without self-care, our ability to help our patients withers. This article provides a useful conceptual framework for individuals and organizations to provide the right care at the right time in these unprecedented times,” said Dr. Shaker, who was not involved with the study.
The authors agreed, stating that clinicians “require proactive psychological protection specifically because they are a population known for putting others’ needs before their own.”
They recommended several resources for HCPs, including the Physician Support Line; Headspace, a mindfulness Web-based app for reducing stress and anxiety; the National Suicide Prevention Lifeline; and the Crisis Text Line.
The authors and Dr. Shaker disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Hysteroscopy and COVID-19: Have recommended techniques changed due to the pandemic?
The emergence of the coronavirus severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection (COVID-19) in December 2019, has resulted in a global pandemic that has challenged the medical community and will continue to represent a public health emergency for the next several months.1 It has rapidly spread globally, infecting many individuals in an unprecedented rate of infection and worldwide reach. On March 11, 2020, the World Health Organization designated COVID-19 as a pandemic. While the majority of infected individuals are asymptomatic or develop only mild symptoms, some have an unfortunate clinical course resulting in multi-organ failure and death.2
It is accepted that the virus mainly spreads during close contact and via respiratory droplets.3 The average time from infection to onset of symptoms ranges from 2 to 14 days, with an average of 5 days.4 Recommended measures to prevent the spread of the infection include social distancing (at least 6 feet from others), meticulous hand hygiene, and wearing a mask covering the mouth and nose when in public.5 Aiming to mitigate the risk of viral dissemination for patients and health care providers, and to preserve hospital resources, all nonessential medical interventions were initially suspended. Recently, the American College of Surgeons in a joint statement with 9 women’s health care societies have provided recommendations on how to resume clinical activities as we recover from the pandemic.6
As we reinitiate clinical activities, gynecologists have been alerted of the potential risk of viral dissemination during gynecologic minimally invasive surgical procedures due to the presence of the virus in blood, stool, and the potential risk of aerosolization of the virus, especially when using smoke-generating devices.7,8 This risk is not limited to intubation and extubation of the airway during anesthesia; the risk also presents itself during other aerosol-generating procedures, such as laparoscopy or robotic surgery.9,10
Hysteroscopy is considered the gold standard procedure for the diagnosis and management of intrauterine pathologies.11 It is frequently performed in an office setting without the use of anesthesia.11,12 It is usually well tolerated, with only a few patients reporting discomfort.12 It allows for immediate treatment (using the “see and treat” approach) while avoiding not only the risk of anesthesia, as stated, but also the need for intubation—which has a high risk of droplet contamination in COVID-19–infected individuals.13
Is there risk of viral dissemination during hysteroscopic procedures?
The novel and rapidly changing nature of the COVID-19 pandemic present many challenges to the gynecologist. Significant concerns have been raised regarding potential risk of viral dissemination during laparoscopic surgery due to aerosolization of viral particles and the presence of the virus in blood and the gastrointestinal tract of infected patients.7 Diagnostic, and some simple, hysteroscopic procedures are commonly performed in an outpatient setting, with the patient awake. Complex hysteroscopic interventions, however, are generally performed in the operating room, typically with the use of general anesthesia. Hysteroscopy has the theoretical risks of viral dissemination when performed in COVID-19–positive patients. Two important questions must be addressed to better understand the potential risk of COVID-19 viral dissemination during hysteroscopic procedures.
Continue to: 1. Is the virus present in the vaginal fluid of women infected with COVID-19?...
1. Is the virus present in the vaginal fluid of women infected with COVID-19?
Recent studies have confirmed the presence of viral particles in urine, feces, blood, and tears in addition to the respiratory tract in patients infected with COVID-19.3,14,15 The presence of the SARS-CoV-2 virus in the female genital system is currently unknown. Previous studies, of other epidemic viral infections, have demonstrated the presence of the virus in the female genital tract in affected patients of Zika virus and Ebola.16,17 However, 2 recent studies have failed to demonstrate the presence of the SARS-CoV-2 virus in the vaginal fluid of pregnant14 and not pregnant18 women with severe COVID-19 infection.
2. Is there risk of viral dissemination during hysteroscopy if using electrosurgery?
There are significant concerns with possible risk of COVID-19 transmission to health care providers in direct contact with infected patients during minimally invasive gynecologic procedures due to direct contamination and aerosolization of the virus.10,19 Current data on COVID-19 transmission during surgery are limited. However, it is important to recognize that viral aerosolization has been documented with other viral diseases, such as human papillomavirus and hepatitis B.20 A recent report called for awareness in the surgical community about the potential risks of COVID-19 viral dissemination during laparoscopic surgery. Among other recommendations, international experts advised minimizing the use of electrosurgery to reduce the creation of surgical plume, decreasing the pneumoperitoneum pressure to minimum levels, and using suction devices in a closed system.21 Although these preventive measures apply to laparoscopic surgery, it is important to consider that hysteroscopy is performed in a unique environment.
During hysteroscopy the uterine cavity is distended with a liquid medium (normal saline or electrolyte-free solutions); this is opposed to gynecologic laparoscopy, in which the peritoneal cavity is distended with carbon dioxide.22 The smoke produced with the use of hysteroscopic electrosurgical instruments generates bubbles that are immediately cooled down to the temperature of the distention media and subsequently dissolve into it. Therefore, there are no bubbles generated during hysteroscopic surgery that are subsequently released into the air. This results in a low risk for viral dissemination during hysteroscopic procedures. Nevertheless, the necessary precautions to minimize the risk of COVID-19 transmission during hysteroscopic intervention are extremely important.
Recommendations for hysteroscopic procedures during the COVID-19 pandemic
We provide our overall recommendations for hysteroscopy, as well as those specific to the office and hospital setting.
Recommendations: General
Limit hysteroscopic procedures to COVID-19–negative patients and to those patients in whom delaying the procedure could result in adverse clinical outcomes.23
Universally screen for potential COVID-19 infection. When possible, a phone interview to triage patients based on their symptoms and infection exposure status should take place before the patient arrives to the health care center. Patients with suspected or confirmed COVID-19 infection who require immediate evaluation should be directed to COVID-19–designated emergency areas.
Universally test for SARS-CoV-2 before procedures performed in the operating room (OR). Using nasopharyngeal swabs for the detection of viral RNA, employing molecular methods such as polymerase chain reaction (PCR), within 48 to 72 hours prior to all OR hysteroscopic procedures is strongly recommended. Adopting this testing strategy will aid to identify asymptomatic SARS-CoV-2‒infected patients, allowing to defer the procedure, if possible, among patients testing positive. If tests are limited, testing only patients scheduled for hysteroscopic procedures in which general or regional anesthesia will be required is acceptable.
Universal SARS-CoV-2 testing of patients undergoing in-office hysteroscopic diagnostic or minor operative procedures without the use of anesthesia is not required.
Limit the presence of a companion. It is understood that visitor policies may vary at the discretion of each institution’s guidelines. Children and individuals over the age of 60 years should not be granted access to the center. Companions will be subjected to the same screening criteria as patients.
Provide for social distancing and other precautionary measures. If more than one patient is scheduled to be at the facility at the same time, ensure that the facility provides adequate space to allow the appropriate social distancing recommendations between patients. Hand sanitizers and facemasks should be available for patients and companions.
Provide PPE for clinicians. All health care providers in close contact with the patient must wear personal protective equipment (PPE), which includes an apron and gown, a surgical mask, eye protection, and gloves. Health care providers should wear PPE deemed appropriate by their regulatory institutions following their local and national guidelines during clinical patient interactions.
Restrict surgical attendees to vital personnel. The participation of learners by physical presence in the office or operating room should be restricted.
Continue to: Recommendations: Office setting...
Recommendations: Office setting
Preprocedural recommendations
- Advise patients to come to the office alone. If the patient requires a companion, a maximum of one adult companion under the age of 60 should be accepted.
- Limit the number of health care team members present in the procedure room.
Intraprocedural recommendations
- Choose the appropriate device(s) that will allow for an effective and fast procedure.
- Use the recommended PPE for all clinicians.
- Limit the movement of staff members in and out of the procedure room.
Postprocedure recommendations
- When more than one case is scheduled to be performed in the same procedure room, allow enough time in between cases to grant a thorough OR decontamination.
- Allow for patients to recover from the procedure in the same room as the procedure took place in order to avoid potential contamination of multiple rooms.
- Expedite patient discharge.
- Follow up after the procedure by phone or telemedicine.
- Use standard endoscope disinfection procedures, as they are effective and should not be modified.
Continue to: Recommendations: Operating room setting...
Recommendations: Operating room setting
Preprocedural recommendations
- Perform adequate patient screening for potential COVID-19 infection. (Screening should be independent of symptoms and not be limited to those with clinical symptoms.)
- Limit the number of health care team members in the operating procedure room.
- To minimize unnecessary staff exposure, have surgeons and staff not needed for intubation remain outside the OR until intubation is completed and leave the OR before extubation.
Intraprocedure recommendations
- Limit personnel in the OR to a minimum.
- Staff should not enter or leave the room during the procedure.
- When possible, use conscious sedation or regional anesthesia to avoid the risk of viral dissemination at the time of intubation/extubation.
- Choose the device that will allow an effective and fast procedure.
- Favor non–smoke-generating devices, such as hysteroscopic scissors, graspers, and tissue retrieval systems.
- Connect active suction to the outflow, especially when using smoke-generating instruments, to facilitate the extraction of surgical smoke.
Postprocedure recommendations
- When more than one case is scheduled to be performed in the same room, allow enough time in between cases to grant a thorough OR decontamination.
- Expedite postprocedure recovery and patient discharge.
- After completion of the procedure, staff should remove scrubs and change into clean clothing.
- Use standard endoscope disinfection procedures, as they are effective and should not be modified.
Conclusions
The COVID-19 pandemic has caused a global health emergency. Our knowledge of this devastating virus is constantly evolving as we continue to fight this overwhelming disease. Theoretical risk of “viral” dissemination is considered extremely low, or negligible, during hysterosocopy. Hysteroscopic procedures in COVID-19–positive patients with life-threatening conditions or in patients in whom delaying the procedure could worsen outcomes should be performed taking appropriate measures. Patients who test negative for COVID-19 (confirmed by PCR) and require hysteroscopic procedures, should be treated using universal precautions. ●
- Al-Shamsi HO, Alhazzani W, Alhuraiji A, et al. A practical approach to the management of cancer patients during the novel coronavirus disease 2019 (COVID-19) pandemic: an international collaborative group. Oncologist. 2020;25:e936-e945.
- Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention. JAMA. February 24, 2020. doi:10.1001/jama.2020.2648.
- Wang W, Xu Y, Gao R, et al. Detection of SARS-CoV-2 in different types of clinical specimens. JAMA. 2020;323:1843-1844.
- Yu F, Yan L, Wang N, et al. Quantitative detection and viral load analysis of SARS-CoV-2 in infected patients. Clin Infect Dis. 2020;71:793-798.
- Prem K, Liu Y, Russell TW, et al; Centre for the Mathematical Modelling of Infectious Diseases COVID-19 Working Group. The effect of control strategies to reduce social mixing on outcomes of the COVID-19 epidemic in Wuhan, China: a modelling study. Lancet Public Health. 2020;5:e261-e270.
- American College of Surgeons, American Society of Aesthesiologists, Association of periOperative Registered Nurses, American Hospital Association. Joint Statement: Roadmap for resuming elective surgery after COVID-19 pandemic. April 16, 2020. https://www.aorn.org/guidelines/aorn-support/roadmap-for-resuming-elective-surgery-after-covid-19. Accessed August 27, 2020.
- Zhang W, Du RH, Li B, et al. Molecular and serological investigation of 2019-nCoV infected patients: implication of multiple shedding routes. Emerg Microbes Infect. 2020;9:386-389.
- Mowbray NG, Ansell J, Horwood J, et al. Safe management of surgical smoke in the age of COVID-19. Br J Surg. May 3, 2020. doi: 10.1002/bjs.11679.
- Cohen SL, Liu G, Abrao M, et al. Perspectives on surgery in the time of COVID-19: safety first. J Minim Invasive Gynecol. 2020;27:792-793.
- COVID-19: protecting health-care workers. Lancet. 2020;395:922.
- Salazar CA, Isaacson KB. Office operative hysteroscopy: an update. J Minim Invasive Gynecol. 2018;25:199-208.
- Cicinelli E. Hysteroscopy without anesthesia: review of recent literature. J Minim Invasive Gynecol. 2010;17:703-708.
- Wax RS, Christian MD. Practical recommendations for critical care and anesthesiology teams caring for novel coronavirus (2019-nCoV) patients. Can J Anaesth. 2020;67:568-576.
- Aslan MM, Yuvaci HU, Köse O, et al. SARS-CoV-2 is not present in the vaginal fluid of pregnant women with COVID-19. J Matern Fetal Neonatal Med. 2020:1-3. doi: 10.1080/14767058.2020.1793318.
- Chen Y, Chen L, Deng Q, et al. The presence of SARS-CoV-2 RNA in the feces of COVID-19 patients. J Med Virol. 2020;92:833-840.
- Prisant N, Bujan L, Benichou H, et al. Zika virus in the female genital tract. Lancet Infect Dis. 2016;16:1000-1001.
- Rodriguez LL, De Roo A, Guimard Y, et al. Persistence and genetic stability of Ebola virus during the outbreak in Kikwit, Democratic Republic of the Congo, 1995. J Infect Dis. 1999;179 Suppl 1:S170-S176.
- Qiu L, Liu X, Xiao M, et al. SARS-CoV-2 is not detectable in the vaginal fluid of women with severe COVID-19 infection. Clin Infect Dis. 2020;71:813-817.
- Brat GA, Hersey S, Chhabra K, et al. Protecting surgical teams during the COVID-19 outbreak: a narrative review and clinical considerations. Ann Surg. April 17, 2020. doi: 10.1097/SLA.0000000000003926.
- Kwak HD, Kim SH, Seo YS, et al. Detecting hepatitis B virus in surgical smoke emitted during laparoscopic surgery. Occup Environ Med. 2016;73:857-863.
- Zheng MH, Boni L, Fingerhut A. Minimally invasive surgery and the novel coronavirus outbreak: lessons learned in China and Italy. Ann Surg. 2020;272:e5-e6.
- Catena U. Surgical smoke in hysteroscopic surgery: does it really matter in COVID-19 times? Facts Views Vis Obgyn. 2020;12:67-68.
- Carugno J, Di Spiezio Sardo A, Alonso L, et al. COVID-19 pandemic. Impact on hysteroscopic procedures: a consensus statement from the Global Congress of Hysteroscopy Scientific Committee. J Minim Invasive Gynecol. 2020;27:988-992.
The emergence of the coronavirus severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection (COVID-19) in December 2019, has resulted in a global pandemic that has challenged the medical community and will continue to represent a public health emergency for the next several months.1 It has rapidly spread globally, infecting many individuals in an unprecedented rate of infection and worldwide reach. On March 11, 2020, the World Health Organization designated COVID-19 as a pandemic. While the majority of infected individuals are asymptomatic or develop only mild symptoms, some have an unfortunate clinical course resulting in multi-organ failure and death.2
It is accepted that the virus mainly spreads during close contact and via respiratory droplets.3 The average time from infection to onset of symptoms ranges from 2 to 14 days, with an average of 5 days.4 Recommended measures to prevent the spread of the infection include social distancing (at least 6 feet from others), meticulous hand hygiene, and wearing a mask covering the mouth and nose when in public.5 Aiming to mitigate the risk of viral dissemination for patients and health care providers, and to preserve hospital resources, all nonessential medical interventions were initially suspended. Recently, the American College of Surgeons in a joint statement with 9 women’s health care societies have provided recommendations on how to resume clinical activities as we recover from the pandemic.6
As we reinitiate clinical activities, gynecologists have been alerted of the potential risk of viral dissemination during gynecologic minimally invasive surgical procedures due to the presence of the virus in blood, stool, and the potential risk of aerosolization of the virus, especially when using smoke-generating devices.7,8 This risk is not limited to intubation and extubation of the airway during anesthesia; the risk also presents itself during other aerosol-generating procedures, such as laparoscopy or robotic surgery.9,10
Hysteroscopy is considered the gold standard procedure for the diagnosis and management of intrauterine pathologies.11 It is frequently performed in an office setting without the use of anesthesia.11,12 It is usually well tolerated, with only a few patients reporting discomfort.12 It allows for immediate treatment (using the “see and treat” approach) while avoiding not only the risk of anesthesia, as stated, but also the need for intubation—which has a high risk of droplet contamination in COVID-19–infected individuals.13

Is there risk of viral dissemination during hysteroscopic procedures?
The novel and rapidly changing nature of the COVID-19 pandemic present many challenges to the gynecologist. Significant concerns have been raised regarding potential risk of viral dissemination during laparoscopic surgery due to aerosolization of viral particles and the presence of the virus in blood and the gastrointestinal tract of infected patients.7 Diagnostic, and some simple, hysteroscopic procedures are commonly performed in an outpatient setting, with the patient awake. Complex hysteroscopic interventions, however, are generally performed in the operating room, typically with the use of general anesthesia. Hysteroscopy has the theoretical risks of viral dissemination when performed in COVID-19–positive patients. Two important questions must be addressed to better understand the potential risk of COVID-19 viral dissemination during hysteroscopic procedures.
Continue to: 1. Is the virus present in the vaginal fluid of women infected with COVID-19?...
1. Is the virus present in the vaginal fluid of women infected with COVID-19?
Recent studies have confirmed the presence of viral particles in urine, feces, blood, and tears in addition to the respiratory tract in patients infected with COVID-19.3,14,15 The presence of the SARS-CoV-2 virus in the female genital system is currently unknown. Previous studies, of other epidemic viral infections, have demonstrated the presence of the virus in the female genital tract in affected patients of Zika virus and Ebola.16,17 However, 2 recent studies have failed to demonstrate the presence of the SARS-CoV-2 virus in the vaginal fluid of pregnant14 and not pregnant18 women with severe COVID-19 infection.
2. Is there risk of viral dissemination during hysteroscopy if using electrosurgery?
There are significant concerns with possible risk of COVID-19 transmission to health care providers in direct contact with infected patients during minimally invasive gynecologic procedures due to direct contamination and aerosolization of the virus.10,19 Current data on COVID-19 transmission during surgery are limited. However, it is important to recognize that viral aerosolization has been documented with other viral diseases, such as human papillomavirus and hepatitis B.20 A recent report called for awareness in the surgical community about the potential risks of COVID-19 viral dissemination during laparoscopic surgery. Among other recommendations, international experts advised minimizing the use of electrosurgery to reduce the creation of surgical plume, decreasing the pneumoperitoneum pressure to minimum levels, and using suction devices in a closed system.21 Although these preventive measures apply to laparoscopic surgery, it is important to consider that hysteroscopy is performed in a unique environment.
During hysteroscopy the uterine cavity is distended with a liquid medium (normal saline or electrolyte-free solutions); this is opposed to gynecologic laparoscopy, in which the peritoneal cavity is distended with carbon dioxide.22 The smoke produced with the use of hysteroscopic electrosurgical instruments generates bubbles that are immediately cooled down to the temperature of the distention media and subsequently dissolve into it. Therefore, there are no bubbles generated during hysteroscopic surgery that are subsequently released into the air. This results in a low risk for viral dissemination during hysteroscopic procedures. Nevertheless, the necessary precautions to minimize the risk of COVID-19 transmission during hysteroscopic intervention are extremely important.
Recommendations for hysteroscopic procedures during the COVID-19 pandemic
We provide our overall recommendations for hysteroscopy, as well as those specific to the office and hospital setting.
Recommendations: General
Limit hysteroscopic procedures to COVID-19–negative patients and to those patients in whom delaying the procedure could result in adverse clinical outcomes.23
Universally screen for potential COVID-19 infection. When possible, a phone interview to triage patients based on their symptoms and infection exposure status should take place before the patient arrives to the health care center. Patients with suspected or confirmed COVID-19 infection who require immediate evaluation should be directed to COVID-19–designated emergency areas.
Universally test for SARS-CoV-2 before procedures performed in the operating room (OR). Using nasopharyngeal swabs for the detection of viral RNA, employing molecular methods such as polymerase chain reaction (PCR), within 48 to 72 hours prior to all OR hysteroscopic procedures is strongly recommended. Adopting this testing strategy will aid to identify asymptomatic SARS-CoV-2‒infected patients, allowing to defer the procedure, if possible, among patients testing positive. If tests are limited, testing only patients scheduled for hysteroscopic procedures in which general or regional anesthesia will be required is acceptable.
Universal SARS-CoV-2 testing of patients undergoing in-office hysteroscopic diagnostic or minor operative procedures without the use of anesthesia is not required.
Limit the presence of a companion. It is understood that visitor policies may vary at the discretion of each institution’s guidelines. Children and individuals over the age of 60 years should not be granted access to the center. Companions will be subjected to the same screening criteria as patients.
Provide for social distancing and other precautionary measures. If more than one patient is scheduled to be at the facility at the same time, ensure that the facility provides adequate space to allow the appropriate social distancing recommendations between patients. Hand sanitizers and facemasks should be available for patients and companions.
Provide PPE for clinicians. All health care providers in close contact with the patient must wear personal protective equipment (PPE), which includes an apron and gown, a surgical mask, eye protection, and gloves. Health care providers should wear PPE deemed appropriate by their regulatory institutions following their local and national guidelines during clinical patient interactions.
Restrict surgical attendees to vital personnel. The participation of learners by physical presence in the office or operating room should be restricted.
Continue to: Recommendations: Office setting...
Recommendations: Office setting
Preprocedural recommendations
- Advise patients to come to the office alone. If the patient requires a companion, a maximum of one adult companion under the age of 60 should be accepted.
- Limit the number of health care team members present in the procedure room.
Intraprocedural recommendations
- Choose the appropriate device(s) that will allow for an effective and fast procedure.
- Use the recommended PPE for all clinicians.
- Limit the movement of staff members in and out of the procedure room.
Postprocedure recommendations
- When more than one case is scheduled to be performed in the same procedure room, allow enough time in between cases to grant a thorough OR decontamination.
- Allow for patients to recover from the procedure in the same room as the procedure took place in order to avoid potential contamination of multiple rooms.
- Expedite patient discharge.
- Follow up after the procedure by phone or telemedicine.
- Use standard endoscope disinfection procedures, as they are effective and should not be modified.
Continue to: Recommendations: Operating room setting...
Recommendations: Operating room setting
Preprocedural recommendations
- Perform adequate patient screening for potential COVID-19 infection. (Screening should be independent of symptoms and not be limited to those with clinical symptoms.)
- Limit the number of health care team members in the operating procedure room.
- To minimize unnecessary staff exposure, have surgeons and staff not needed for intubation remain outside the OR until intubation is completed and leave the OR before extubation.
Intraprocedure recommendations
- Limit personnel in the OR to a minimum.
- Staff should not enter or leave the room during the procedure.
- When possible, use conscious sedation or regional anesthesia to avoid the risk of viral dissemination at the time of intubation/extubation.
- Choose the device that will allow an effective and fast procedure.
- Favor non–smoke-generating devices, such as hysteroscopic scissors, graspers, and tissue retrieval systems.
- Connect active suction to the outflow, especially when using smoke-generating instruments, to facilitate the extraction of surgical smoke.
Postprocedure recommendations
- When more than one case is scheduled to be performed in the same room, allow enough time in between cases to grant a thorough OR decontamination.
- Expedite postprocedure recovery and patient discharge.
- After completion of the procedure, staff should remove scrubs and change into clean clothing.
- Use standard endoscope disinfection procedures, as they are effective and should not be modified.
Conclusions
The COVID-19 pandemic has caused a global health emergency. Our knowledge of this devastating virus is constantly evolving as we continue to fight this overwhelming disease. Theoretical risk of “viral” dissemination is considered extremely low, or negligible, during hysterosocopy. Hysteroscopic procedures in COVID-19–positive patients with life-threatening conditions or in patients in whom delaying the procedure could worsen outcomes should be performed taking appropriate measures. Patients who test negative for COVID-19 (confirmed by PCR) and require hysteroscopic procedures, should be treated using universal precautions. ●
The emergence of the coronavirus severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection (COVID-19) in December 2019, has resulted in a global pandemic that has challenged the medical community and will continue to represent a public health emergency for the next several months.1 It has rapidly spread globally, infecting many individuals in an unprecedented rate of infection and worldwide reach. On March 11, 2020, the World Health Organization designated COVID-19 as a pandemic. While the majority of infected individuals are asymptomatic or develop only mild symptoms, some have an unfortunate clinical course resulting in multi-organ failure and death.2
It is accepted that the virus mainly spreads during close contact and via respiratory droplets.3 The average time from infection to onset of symptoms ranges from 2 to 14 days, with an average of 5 days.4 Recommended measures to prevent the spread of the infection include social distancing (at least 6 feet from others), meticulous hand hygiene, and wearing a mask covering the mouth and nose when in public.5 Aiming to mitigate the risk of viral dissemination for patients and health care providers, and to preserve hospital resources, all nonessential medical interventions were initially suspended. Recently, the American College of Surgeons in a joint statement with 9 women’s health care societies have provided recommendations on how to resume clinical activities as we recover from the pandemic.6
As we reinitiate clinical activities, gynecologists have been alerted of the potential risk of viral dissemination during gynecologic minimally invasive surgical procedures due to the presence of the virus in blood, stool, and the potential risk of aerosolization of the virus, especially when using smoke-generating devices.7,8 This risk is not limited to intubation and extubation of the airway during anesthesia; the risk also presents itself during other aerosol-generating procedures, such as laparoscopy or robotic surgery.9,10
Hysteroscopy is considered the gold standard procedure for the diagnosis and management of intrauterine pathologies.11 It is frequently performed in an office setting without the use of anesthesia.11,12 It is usually well tolerated, with only a few patients reporting discomfort.12 It allows for immediate treatment (using the “see and treat” approach) while avoiding not only the risk of anesthesia, as stated, but also the need for intubation—which has a high risk of droplet contamination in COVID-19–infected individuals.13

Is there risk of viral dissemination during hysteroscopic procedures?
The novel and rapidly changing nature of the COVID-19 pandemic present many challenges to the gynecologist. Significant concerns have been raised regarding potential risk of viral dissemination during laparoscopic surgery due to aerosolization of viral particles and the presence of the virus in blood and the gastrointestinal tract of infected patients.7 Diagnostic, and some simple, hysteroscopic procedures are commonly performed in an outpatient setting, with the patient awake. Complex hysteroscopic interventions, however, are generally performed in the operating room, typically with the use of general anesthesia. Hysteroscopy has the theoretical risks of viral dissemination when performed in COVID-19–positive patients. Two important questions must be addressed to better understand the potential risk of COVID-19 viral dissemination during hysteroscopic procedures.
Continue to: 1. Is the virus present in the vaginal fluid of women infected with COVID-19?...
1. Is the virus present in the vaginal fluid of women infected with COVID-19?
Recent studies have confirmed the presence of viral particles in urine, feces, blood, and tears in addition to the respiratory tract in patients infected with COVID-19.3,14,15 The presence of the SARS-CoV-2 virus in the female genital system is currently unknown. Previous studies, of other epidemic viral infections, have demonstrated the presence of the virus in the female genital tract in affected patients of Zika virus and Ebola.16,17 However, 2 recent studies have failed to demonstrate the presence of the SARS-CoV-2 virus in the vaginal fluid of pregnant14 and not pregnant18 women with severe COVID-19 infection.
2. Is there risk of viral dissemination during hysteroscopy if using electrosurgery?
There are significant concerns with possible risk of COVID-19 transmission to health care providers in direct contact with infected patients during minimally invasive gynecologic procedures due to direct contamination and aerosolization of the virus.10,19 Current data on COVID-19 transmission during surgery are limited. However, it is important to recognize that viral aerosolization has been documented with other viral diseases, such as human papillomavirus and hepatitis B.20 A recent report called for awareness in the surgical community about the potential risks of COVID-19 viral dissemination during laparoscopic surgery. Among other recommendations, international experts advised minimizing the use of electrosurgery to reduce the creation of surgical plume, decreasing the pneumoperitoneum pressure to minimum levels, and using suction devices in a closed system.21 Although these preventive measures apply to laparoscopic surgery, it is important to consider that hysteroscopy is performed in a unique environment.
During hysteroscopy the uterine cavity is distended with a liquid medium (normal saline or electrolyte-free solutions); this is opposed to gynecologic laparoscopy, in which the peritoneal cavity is distended with carbon dioxide.22 The smoke produced with the use of hysteroscopic electrosurgical instruments generates bubbles that are immediately cooled down to the temperature of the distention media and subsequently dissolve into it. Therefore, there are no bubbles generated during hysteroscopic surgery that are subsequently released into the air. This results in a low risk for viral dissemination during hysteroscopic procedures. Nevertheless, the necessary precautions to minimize the risk of COVID-19 transmission during hysteroscopic intervention are extremely important.
Recommendations for hysteroscopic procedures during the COVID-19 pandemic
We provide our overall recommendations for hysteroscopy, as well as those specific to the office and hospital setting.
Recommendations: General
Limit hysteroscopic procedures to COVID-19–negative patients and to those patients in whom delaying the procedure could result in adverse clinical outcomes.23
Universally screen for potential COVID-19 infection. When possible, a phone interview to triage patients based on their symptoms and infection exposure status should take place before the patient arrives to the health care center. Patients with suspected or confirmed COVID-19 infection who require immediate evaluation should be directed to COVID-19–designated emergency areas.
Universally test for SARS-CoV-2 before procedures performed in the operating room (OR). Using nasopharyngeal swabs for the detection of viral RNA, employing molecular methods such as polymerase chain reaction (PCR), within 48 to 72 hours prior to all OR hysteroscopic procedures is strongly recommended. Adopting this testing strategy will aid to identify asymptomatic SARS-CoV-2‒infected patients, allowing to defer the procedure, if possible, among patients testing positive. If tests are limited, testing only patients scheduled for hysteroscopic procedures in which general or regional anesthesia will be required is acceptable.
Universal SARS-CoV-2 testing of patients undergoing in-office hysteroscopic diagnostic or minor operative procedures without the use of anesthesia is not required.
Limit the presence of a companion. It is understood that visitor policies may vary at the discretion of each institution’s guidelines. Children and individuals over the age of 60 years should not be granted access to the center. Companions will be subjected to the same screening criteria as patients.
Provide for social distancing and other precautionary measures. If more than one patient is scheduled to be at the facility at the same time, ensure that the facility provides adequate space to allow the appropriate social distancing recommendations between patients. Hand sanitizers and facemasks should be available for patients and companions.
Provide PPE for clinicians. All health care providers in close contact with the patient must wear personal protective equipment (PPE), which includes an apron and gown, a surgical mask, eye protection, and gloves. Health care providers should wear PPE deemed appropriate by their regulatory institutions following their local and national guidelines during clinical patient interactions.
Restrict surgical attendees to vital personnel. The participation of learners by physical presence in the office or operating room should be restricted.
Continue to: Recommendations: Office setting...
Recommendations: Office setting
Preprocedural recommendations
- Advise patients to come to the office alone. If the patient requires a companion, a maximum of one adult companion under the age of 60 should be accepted.
- Limit the number of health care team members present in the procedure room.
Intraprocedural recommendations
- Choose the appropriate device(s) that will allow for an effective and fast procedure.
- Use the recommended PPE for all clinicians.
- Limit the movement of staff members in and out of the procedure room.
Postprocedure recommendations
- When more than one case is scheduled to be performed in the same procedure room, allow enough time in between cases to grant a thorough OR decontamination.
- Allow for patients to recover from the procedure in the same room as the procedure took place in order to avoid potential contamination of multiple rooms.
- Expedite patient discharge.
- Follow up after the procedure by phone or telemedicine.
- Use standard endoscope disinfection procedures, as they are effective and should not be modified.
Continue to: Recommendations: Operating room setting...
Recommendations: Operating room setting
Preprocedural recommendations
- Perform adequate patient screening for potential COVID-19 infection. (Screening should be independent of symptoms and not be limited to those with clinical symptoms.)
- Limit the number of health care team members in the operating procedure room.
- To minimize unnecessary staff exposure, have surgeons and staff not needed for intubation remain outside the OR until intubation is completed and leave the OR before extubation.
Intraprocedure recommendations
- Limit personnel in the OR to a minimum.
- Staff should not enter or leave the room during the procedure.
- When possible, use conscious sedation or regional anesthesia to avoid the risk of viral dissemination at the time of intubation/extubation.
- Choose the device that will allow an effective and fast procedure.
- Favor non–smoke-generating devices, such as hysteroscopic scissors, graspers, and tissue retrieval systems.
- Connect active suction to the outflow, especially when using smoke-generating instruments, to facilitate the extraction of surgical smoke.
Postprocedure recommendations
- When more than one case is scheduled to be performed in the same room, allow enough time in between cases to grant a thorough OR decontamination.
- Expedite postprocedure recovery and patient discharge.
- After completion of the procedure, staff should remove scrubs and change into clean clothing.
- Use standard endoscope disinfection procedures, as they are effective and should not be modified.
Conclusions
The COVID-19 pandemic has caused a global health emergency. Our knowledge of this devastating virus is constantly evolving as we continue to fight this overwhelming disease. Theoretical risk of “viral” dissemination is considered extremely low, or negligible, during hysterosocopy. Hysteroscopic procedures in COVID-19–positive patients with life-threatening conditions or in patients in whom delaying the procedure could worsen outcomes should be performed taking appropriate measures. Patients who test negative for COVID-19 (confirmed by PCR) and require hysteroscopic procedures, should be treated using universal precautions. ●
- Al-Shamsi HO, Alhazzani W, Alhuraiji A, et al. A practical approach to the management of cancer patients during the novel coronavirus disease 2019 (COVID-19) pandemic: an international collaborative group. Oncologist. 2020;25:e936-e945.
- Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention. JAMA. February 24, 2020. doi:10.1001/jama.2020.2648.
- Wang W, Xu Y, Gao R, et al. Detection of SARS-CoV-2 in different types of clinical specimens. JAMA. 2020;323:1843-1844.
- Yu F, Yan L, Wang N, et al. Quantitative detection and viral load analysis of SARS-CoV-2 in infected patients. Clin Infect Dis. 2020;71:793-798.
- Prem K, Liu Y, Russell TW, et al; Centre for the Mathematical Modelling of Infectious Diseases COVID-19 Working Group. The effect of control strategies to reduce social mixing on outcomes of the COVID-19 epidemic in Wuhan, China: a modelling study. Lancet Public Health. 2020;5:e261-e270.
- American College of Surgeons, American Society of Aesthesiologists, Association of periOperative Registered Nurses, American Hospital Association. Joint Statement: Roadmap for resuming elective surgery after COVID-19 pandemic. April 16, 2020. https://www.aorn.org/guidelines/aorn-support/roadmap-for-resuming-elective-surgery-after-covid-19. Accessed August 27, 2020.
- Zhang W, Du RH, Li B, et al. Molecular and serological investigation of 2019-nCoV infected patients: implication of multiple shedding routes. Emerg Microbes Infect. 2020;9:386-389.
- Mowbray NG, Ansell J, Horwood J, et al. Safe management of surgical smoke in the age of COVID-19. Br J Surg. May 3, 2020. doi: 10.1002/bjs.11679.
- Cohen SL, Liu G, Abrao M, et al. Perspectives on surgery in the time of COVID-19: safety first. J Minim Invasive Gynecol. 2020;27:792-793.
- COVID-19: protecting health-care workers. Lancet. 2020;395:922.
- Salazar CA, Isaacson KB. Office operative hysteroscopy: an update. J Minim Invasive Gynecol. 2018;25:199-208.
- Cicinelli E. Hysteroscopy without anesthesia: review of recent literature. J Minim Invasive Gynecol. 2010;17:703-708.
- Wax RS, Christian MD. Practical recommendations for critical care and anesthesiology teams caring for novel coronavirus (2019-nCoV) patients. Can J Anaesth. 2020;67:568-576.
- Aslan MM, Yuvaci HU, Köse O, et al. SARS-CoV-2 is not present in the vaginal fluid of pregnant women with COVID-19. J Matern Fetal Neonatal Med. 2020:1-3. doi: 10.1080/14767058.2020.1793318.
- Chen Y, Chen L, Deng Q, et al. The presence of SARS-CoV-2 RNA in the feces of COVID-19 patients. J Med Virol. 2020;92:833-840.
- Prisant N, Bujan L, Benichou H, et al. Zika virus in the female genital tract. Lancet Infect Dis. 2016;16:1000-1001.
- Rodriguez LL, De Roo A, Guimard Y, et al. Persistence and genetic stability of Ebola virus during the outbreak in Kikwit, Democratic Republic of the Congo, 1995. J Infect Dis. 1999;179 Suppl 1:S170-S176.
- Qiu L, Liu X, Xiao M, et al. SARS-CoV-2 is not detectable in the vaginal fluid of women with severe COVID-19 infection. Clin Infect Dis. 2020;71:813-817.
- Brat GA, Hersey S, Chhabra K, et al. Protecting surgical teams during the COVID-19 outbreak: a narrative review and clinical considerations. Ann Surg. April 17, 2020. doi: 10.1097/SLA.0000000000003926.
- Kwak HD, Kim SH, Seo YS, et al. Detecting hepatitis B virus in surgical smoke emitted during laparoscopic surgery. Occup Environ Med. 2016;73:857-863.
- Zheng MH, Boni L, Fingerhut A. Minimally invasive surgery and the novel coronavirus outbreak: lessons learned in China and Italy. Ann Surg. 2020;272:e5-e6.
- Catena U. Surgical smoke in hysteroscopic surgery: does it really matter in COVID-19 times? Facts Views Vis Obgyn. 2020;12:67-68.
- Carugno J, Di Spiezio Sardo A, Alonso L, et al. COVID-19 pandemic. Impact on hysteroscopic procedures: a consensus statement from the Global Congress of Hysteroscopy Scientific Committee. J Minim Invasive Gynecol. 2020;27:988-992.
- Al-Shamsi HO, Alhazzani W, Alhuraiji A, et al. A practical approach to the management of cancer patients during the novel coronavirus disease 2019 (COVID-19) pandemic: an international collaborative group. Oncologist. 2020;25:e936-e945.
- Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention. JAMA. February 24, 2020. doi:10.1001/jama.2020.2648.
- Wang W, Xu Y, Gao R, et al. Detection of SARS-CoV-2 in different types of clinical specimens. JAMA. 2020;323:1843-1844.
- Yu F, Yan L, Wang N, et al. Quantitative detection and viral load analysis of SARS-CoV-2 in infected patients. Clin Infect Dis. 2020;71:793-798.
- Prem K, Liu Y, Russell TW, et al; Centre for the Mathematical Modelling of Infectious Diseases COVID-19 Working Group. The effect of control strategies to reduce social mixing on outcomes of the COVID-19 epidemic in Wuhan, China: a modelling study. Lancet Public Health. 2020;5:e261-e270.
- American College of Surgeons, American Society of Aesthesiologists, Association of periOperative Registered Nurses, American Hospital Association. Joint Statement: Roadmap for resuming elective surgery after COVID-19 pandemic. April 16, 2020. https://www.aorn.org/guidelines/aorn-support/roadmap-for-resuming-elective-surgery-after-covid-19. Accessed August 27, 2020.
- Zhang W, Du RH, Li B, et al. Molecular and serological investigation of 2019-nCoV infected patients: implication of multiple shedding routes. Emerg Microbes Infect. 2020;9:386-389.
- Mowbray NG, Ansell J, Horwood J, et al. Safe management of surgical smoke in the age of COVID-19. Br J Surg. May 3, 2020. doi: 10.1002/bjs.11679.
- Cohen SL, Liu G, Abrao M, et al. Perspectives on surgery in the time of COVID-19: safety first. J Minim Invasive Gynecol. 2020;27:792-793.
- COVID-19: protecting health-care workers. Lancet. 2020;395:922.
- Salazar CA, Isaacson KB. Office operative hysteroscopy: an update. J Minim Invasive Gynecol. 2018;25:199-208.
- Cicinelli E. Hysteroscopy without anesthesia: review of recent literature. J Minim Invasive Gynecol. 2010;17:703-708.
- Wax RS, Christian MD. Practical recommendations for critical care and anesthesiology teams caring for novel coronavirus (2019-nCoV) patients. Can J Anaesth. 2020;67:568-576.
- Aslan MM, Yuvaci HU, Köse O, et al. SARS-CoV-2 is not present in the vaginal fluid of pregnant women with COVID-19. J Matern Fetal Neonatal Med. 2020:1-3. doi: 10.1080/14767058.2020.1793318.
- Chen Y, Chen L, Deng Q, et al. The presence of SARS-CoV-2 RNA in the feces of COVID-19 patients. J Med Virol. 2020;92:833-840.
- Prisant N, Bujan L, Benichou H, et al. Zika virus in the female genital tract. Lancet Infect Dis. 2016;16:1000-1001.
- Rodriguez LL, De Roo A, Guimard Y, et al. Persistence and genetic stability of Ebola virus during the outbreak in Kikwit, Democratic Republic of the Congo, 1995. J Infect Dis. 1999;179 Suppl 1:S170-S176.
- Qiu L, Liu X, Xiao M, et al. SARS-CoV-2 is not detectable in the vaginal fluid of women with severe COVID-19 infection. Clin Infect Dis. 2020;71:813-817.
- Brat GA, Hersey S, Chhabra K, et al. Protecting surgical teams during the COVID-19 outbreak: a narrative review and clinical considerations. Ann Surg. April 17, 2020. doi: 10.1097/SLA.0000000000003926.
- Kwak HD, Kim SH, Seo YS, et al. Detecting hepatitis B virus in surgical smoke emitted during laparoscopic surgery. Occup Environ Med. 2016;73:857-863.
- Zheng MH, Boni L, Fingerhut A. Minimally invasive surgery and the novel coronavirus outbreak: lessons learned in China and Italy. Ann Surg. 2020;272:e5-e6.
- Catena U. Surgical smoke in hysteroscopic surgery: does it really matter in COVID-19 times? Facts Views Vis Obgyn. 2020;12:67-68.
- Carugno J, Di Spiezio Sardo A, Alonso L, et al. COVID-19 pandemic. Impact on hysteroscopic procedures: a consensus statement from the Global Congress of Hysteroscopy Scientific Committee. J Minim Invasive Gynecol. 2020;27:988-992.
Dangers behind antimaskers and antivaxxers: How to combat both
Niket Sonpal, MD, thought he’d heard most of the myths about wearing masks during the pandemic, but the recent claim from a patient was a new one for the New York City gastroenterologist.
The patient refused to wear a mask because she heard inhaling bad breath through a mask could be toxic. The woman said the rumor was circulating on Facebook. Sonpal calmly explained that breathing your own breath is not going to cause health problems, he said.
“There’s a lot of controversy on masks,” he said. “Unfortunately, it’s really just a lack of education and buy-in. Social media is the primary source of all this misinformation. These kinds of over-the-top hyperbole has basically led to a disbelief that masks are effective. The disbelief is hard to break up.”
As mask requirements have tightened amid the ongoing pandemic, debates about face coverings have emerged front and center, with a growing number of people opposing mask usage. So-called antimaskers dispute the benefits of wearing masks and many contend that face coverings decrease oxygen flow and can lead to illness. Sentiment against masks have led to protests nationwide, ignited public conflicts in some areas, and even generated lawsuits over mask mandates.
The issue presents an ongoing challenge for physicians as they strive to educate patients about the significance of masking against the flood of antimask messages on social media and beyond. Opposition to masks is particularly frustrating for health professionals who have witnessed patients, family, or friends become ill or die from the virus. Refusing to mask and failing to social distance have been linked to the rapid spread of the coronavirus and subsequent deaths.
“I have had colleagues pass away, and it’s extremely disheartening and frustrating to see science so easily disregarded,” Sonpal said. “Masks save lives and protect people and not wearing them is simply a lack of respect, not just for your fellow colleagues, but for a member of your species.”
Michael Rebresh, who helped create the antimask group Million Unmasked Patriots, says his group’s objections to masks are rational and reasonable. The group, which has more than 8,000 members, formed in response to guidance by Illinois state officials that children would only be allowed to return to school wearing a mask.
“Our objections are to the fact that masks on children in school have a greater propensity to make children sick from breathing in bacteria that forms on the inner layer of a mask worn for hours on end,” Rebresh said. “We have an objection to the increase of CO2 intake and a decrease in oxygen flow for kids who need all the oxygen they can get during a learning environment. We recognized the masking of ourselves and kids for what it is: A political move to separate the two parties in our November election and define and create division between the two.”
Million Unmasked Patriots is one of dozens of antimask groups on social media platforms such as Facebook, Instagram, and TikTok. In July, Facebook suspended one such group, Unmasking America, which boasts 9,600 members, for posting repeated claims that face masks obstruct oxygen flow and have negative mental health effects.
Experts say the antiscience rhetoric is far from new. The antimask movement in many ways, shares similarities with that of the anti-vaccine movement, says Todd Wolynn, MD, a Pittsburgh pediatrician and cofounder of Shots Heard Round the World, an organization that defends vaccine advocates against coordinated online attacks by antivaxxers.
“A lot of it is conspiracy-laden,” said Wolynn of the disinformation. “That Dr. [Anthony] Fauci somehow helped construct the pandemic and that it’s not real. That Bill Gates is funding the vaccine so he can inject people with microchips. All sorts of really out-there, ungrounded conspiracy theories. If you had Venn diagram of antimask and antivaxx, I would say there’s clearly overlap.”
Parallels between antimaskers, antivaxxers
Opponents to masks fall on a spectrum, explains Vineet Arora, MD, a hospitalist and associate chief medical officer–clinical learning environment at University of Chicago Medicine. People who believe conspiracy theories and push misinformation are on one end, she said. There are also those who generally don’t believe the seriousness of the pandemic, feel their risk is minimal, or doubt the benefits of masks.
The two trains of thought resemble the distinction among parents who are antivaccine and those who are simply “vaccine hesitant,” says Arora, who co-authored a recent article about masking and misinformation that addresses antivaccine attitudes.
“While the antimask sentiment gets a lot of attention, I think it’s important to highlight there’s a lot of vocal anti-mask sentiment since most people are supportive of masks,” she said. “There might be people sitting on the fence who are just unsure about wearing a mask. That’s understandable because the science and the communication has evolved. There was a lot of early mixed messages about masking. Anytime you have confusion about the science or the science is evolving, it’s easy to have misinformation and then have that take off as myth.”
Just as antivaxxers work to swing the opinion of the vaccine hesitant, antimaskers are vying with public health advocates for the support of the mask hesitant, she said. Creating doubt in public health authorities is one way they are gaining followers. Anti-maskers often question and scrutinize past messaging about masks by public health officials, claiming that because guidance on masks has changed over time, the science behind masks and current guidance can’t be trusted, Wolynn said. Similarly, antivaxxers frequently question past actions by public health officials, such as the Tuskegee Experiment (which began in 1932), to try to poke holes in the credibility of public health officials and their advice.
Both the antimask and antivaccine movements also tend to base their resistance on a personal liberties argument, adds Jacqueline Winfield Fincher, MD, president for the American College of Physicians and an internist based in Thomson, Georgia. Antimaskers contend they should be free to decide whether to wear face coverings and that rules requiring masks infringe upon their civil liberties. Similarly, antivaxxers argue they should be free to decide whether to vaccinate their children and contend vaccine mandates violate their personal liberties.
Taking a deeper look, fear and control are two likely drivers of antimasking and antivaccine attitudes, Fincher said. Those refusing to wear masks may feel they have no control over the pandemic or its impacts, but they can control how they respond to mask-wearing requirements, she said.
Antivaccine parents often want more control over their children’s healthcare and falsely believe that vaccines are injecting something harmful into their children or may lead to harmful reactions.
“It’s a control issue and a defense mechanism,” she said. “Some people may feel helpless to deal with the pandemic or believe since it is not affecting them or their family, that it is not real. ‘If I just deny it and I don’t acknowledge facts, I don’t have to worry about it or do anything about it, and therefore I will have more control over my day-to-day life.’”
Groups fueling each other
In some cases, antimask and antivaxx groups are joining forces or adopting dual causes.
In California for instance, longtime opponents to vaccines are now objecting to mask policies as similar infringement to their bodily autonomy. Demonstrations in Texas, Idaho, and Michigan against mask mandates and other COVID-19 requirements have drawn support from anti-vaccine activists and incorporated antivaccine propaganda.
In Illinois, Million Unmasked Patriots, formally the Million Unmasked March, has received widespread attention for protesting both masks for returning schoolchildren and a future COVID-19 vaccine requirement.
A July protest planned by the antimask group triggered a letter by Arora and 500 other healthcare professionals to Illinois lawmakers decrying the group’s views and urging the state to move forward with universal masking in schools.
“What’s happening is those who are distrustful of government and public health and science are joining together,” said Arora, who coauthored a piece about the problem on KevinMD.com. “It’s important to address both movements together because they can quickly feed off each other and build in momentum. At the heart of both is really this deep skepticism of science.”
Rebresh of Million Unmasked Patriots said most of his members are not opposed to all vaccines, but rather they are opposed to “untested vaccines.” The primary concern is the inability to research long-term effects of a COVID-19 vaccine before its approval, he said.
Rebresh disagrees with the antimask movement being compared with the antivaccine movement. The two groups are “motivated by different things and a different set of circumstances drive their opinions,” he said. However, Rebresh believes that potential harm resulting from “mass vaccinations” is a valid concern. For this reason, he and his wife chose for their children to receive their vaccinations individually over a series of weeks, rather than the “kiddie cocktail of vaccines,” at a single visit, he said.
Vaccine scientist Peter Hotez, MD, PhD, said the antivaccine movement appears to have grown stronger from the pandemic fueled by fresh conspiracies and new alliances. Antivaccine sentiment has been gaining steam over the last several years and collecting more allies from the far-right, said Hotez, dean for the National School of Tropical Medicine and codirector for the Texas Children’s Hospital Center for Vaccine Development.
“Now what you’re seeing is yet another expansion this year, with antivaccine groups, under the banner of ‘health freedom,’ campaigning against social distancing and wearing masks and contact tracing,” he said. “What was an antivaccine movement has now become a full-blown antiscience movement and an anti-public health movement. It’s causing a lot of damage and I believe costing a lot of American lives.”
Neil F. Johnson, PhD, who has studied the antivaccine movement and its social media proliferation during the pandemic, said online comments by antivaxxers frequently condemn mask usage and showcase memes making fun of masks.
“In those same narratives about opposing vaccines for COVID, we see a lot of discussion against masks,” said Johnson, a physics professor at George Washington University in Washington, D.C. “If you don’t believe in the official picture of COVID, you don’t believe the policies or the advice that’s given about COVID.”
An analysis by Johnson that examined 1,300 Facebook pages found that, while antivaxxers have fewer followers than provaccine pages, antivaccine pages are more numerous, faster growing, and are more often connected to unrelated, undecided pages. Conversely, pages that advocate the benefits of vaccinations and explain the science behind immunizations are largely disconnected from such undecided communities, according to the study, published May 13 in Nature.
The study suggests the antivaccine movement is making influential strides during the pandemic and connecting with people who are undecided, while public health advocates are not building the same bridges, Johnson said.
“I think it’s hugely dangerous, because I don’t know any other moment in science or in public health when there was so much uncertainty in something affecting everybody,” he said. “Every policy that will be coming, everything depends on people buying into the official message. Once you have the seeds of doubt, that’s a very difficult thing to overcome. It’s an unprecedented challenge.”
How physicians and clinicians can help
A more aggressive approach is necessary when it comes to taking down antiscience content on social media, says Hotez. Too often, misinformation and antiscience rhetoric is allowed to linger on popular sites such as Facebook and Amazon.
Wolynn agrees. On personal or business platforms, it’s crucial to ban, hide, and delete such comments as quickly as possible, he said. On public sites, purposeful disinformation should be immediately reported to the platform.
At the same time, Wolynn said it’s essential to support those who make sound, science-based comments in social media forums.
“If you see someone who is pushing accurate, evidence-based information, and they come under attack, they should be supported and defended and empowered,” Wolynn said. “Shots Heard Round the World is doing all of those things, including galvanizing and recruiting more people to help get their voices out there.”
Expanded visibility by physicians and scientists would greatly help counter the spread of antiscience sentiment, adds Hotez.
“Too often, antiscience movements are able to flourish because scientists and physicians are invisible,” he said. “They’re too focused on either clinical practices or in the case of physician scientists, on grants and papers and not enough attention to public engagement. We’re going to have to change that around. We need to hear more from scientists directly.”
To that end, Wolynn said health care professionals, including medical students and residents, need to have formal training in communications, media, and social media as part of their education – and more support from employers to engage through social media.
“That’s where the fight is,” Wolynn said. “You can be the best diagnostician, the best clinician. You can make the right diagnosis and prescribe the right medication, but if families don’t hear what you’re saying, you’re not going to be effective. If you can’t be on the platform where they’re being influenced, we’re losing the battle.”
Speaking to your mask-hesitant patients
Concentrating on those who are uncertain about masks is particularly key for physicians and public health advocates as the pandemic continues, says Arora.
“It’s important for us to focus on the mask-hesitant who often don’t get the attention they need,” she said.
She suggests bringing up the subject of masks with patients during visits, asking about mask usage, discussing rumors they’ve heard, and emphasizing why masks are important. Be a role model by wearing a mask in your community and on social media, she added.
Some patients have real concerns about not being able to breathe through masks or anxiety disorders that can be aggravated even by the thought of wearing a mask, noted Susan R. Bailey, MD, president for the American Medical Association. Bailey, an immunologist, recently counseled a patient with a deviated nasal septum in addition to a panic disorder who was worried about wearing a mask, she said. Bailey listened to the patient’s concerns, discussed his health conditions, and proposed an alternative face covering that might make him more comfortable.
“Every patient is different,” Bailey said. “It’s important for us to remember that each person who is reluctant to wear a mask has their own reasons. It’s important for us to express some empathy – to agree with them, yes, masks are hot and inconvenient – and help understand their questions, which you may be able to answer to their satisfaction. There are patients that have legitimate questions and a physician caring about how they feel, can make all the difference.”
Physicians can also get involved with the AMA’s #MaskUp campaign, an effort to normalize mask wearing and debunk myths associated with masks. The campaign includes social media materials, slogans doctors can tweet, and profile pictures they can use on social media. The campaign’s toolkit includes images, videos, and information that physicians can share with patients and the public.
Enforcing strong mask policies at your practice and ensuring all staff are modeling appropriate mask behavior is also important, adds Fincher of the ACP. The college recently issued a policy supporting mask usage in community settings.
If a patient conveys an antimask belief, Fincher suggests not directly challenging the person’s views, but listening to them and offering objective data, discussing the science behind masks, and directing them to credible sources.
“Doctors are used to this. We recommend a lot of things to patients that they don’t want to do,” Fincher said. “If a patient feels attacked, they act defensively. But if you base your explanation in more objective terms with data, numbers, and personalize the risks and benefits of a vaccine, a healthy change in behavior, or a medication, then patients are more likely to hear your concerns and do the right thing. Having a long-term relationship with a trusted physician makes all of these issues much easier to discuss and to implement the best plan for the individual patient.”
This article first appeared on Medscape.com.
Niket Sonpal, MD, thought he’d heard most of the myths about wearing masks during the pandemic, but the recent claim from a patient was a new one for the New York City gastroenterologist.
The patient refused to wear a mask because she heard inhaling bad breath through a mask could be toxic. The woman said the rumor was circulating on Facebook. Sonpal calmly explained that breathing your own breath is not going to cause health problems, he said.
“There’s a lot of controversy on masks,” he said. “Unfortunately, it’s really just a lack of education and buy-in. Social media is the primary source of all this misinformation. These kinds of over-the-top hyperbole has basically led to a disbelief that masks are effective. The disbelief is hard to break up.”
As mask requirements have tightened amid the ongoing pandemic, debates about face coverings have emerged front and center, with a growing number of people opposing mask usage. So-called antimaskers dispute the benefits of wearing masks and many contend that face coverings decrease oxygen flow and can lead to illness. Sentiment against masks have led to protests nationwide, ignited public conflicts in some areas, and even generated lawsuits over mask mandates.
The issue presents an ongoing challenge for physicians as they strive to educate patients about the significance of masking against the flood of antimask messages on social media and beyond. Opposition to masks is particularly frustrating for health professionals who have witnessed patients, family, or friends become ill or die from the virus. Refusing to mask and failing to social distance have been linked to the rapid spread of the coronavirus and subsequent deaths.
“I have had colleagues pass away, and it’s extremely disheartening and frustrating to see science so easily disregarded,” Sonpal said. “Masks save lives and protect people and not wearing them is simply a lack of respect, not just for your fellow colleagues, but for a member of your species.”
Michael Rebresh, who helped create the antimask group Million Unmasked Patriots, says his group’s objections to masks are rational and reasonable. The group, which has more than 8,000 members, formed in response to guidance by Illinois state officials that children would only be allowed to return to school wearing a mask.
“Our objections are to the fact that masks on children in school have a greater propensity to make children sick from breathing in bacteria that forms on the inner layer of a mask worn for hours on end,” Rebresh said. “We have an objection to the increase of CO2 intake and a decrease in oxygen flow for kids who need all the oxygen they can get during a learning environment. We recognized the masking of ourselves and kids for what it is: A political move to separate the two parties in our November election and define and create division between the two.”
Million Unmasked Patriots is one of dozens of antimask groups on social media platforms such as Facebook, Instagram, and TikTok. In July, Facebook suspended one such group, Unmasking America, which boasts 9,600 members, for posting repeated claims that face masks obstruct oxygen flow and have negative mental health effects.
Experts say the antiscience rhetoric is far from new. The antimask movement in many ways, shares similarities with that of the anti-vaccine movement, says Todd Wolynn, MD, a Pittsburgh pediatrician and cofounder of Shots Heard Round the World, an organization that defends vaccine advocates against coordinated online attacks by antivaxxers.
“A lot of it is conspiracy-laden,” said Wolynn of the disinformation. “That Dr. [Anthony] Fauci somehow helped construct the pandemic and that it’s not real. That Bill Gates is funding the vaccine so he can inject people with microchips. All sorts of really out-there, ungrounded conspiracy theories. If you had Venn diagram of antimask and antivaxx, I would say there’s clearly overlap.”
Parallels between antimaskers, antivaxxers
Opponents to masks fall on a spectrum, explains Vineet Arora, MD, a hospitalist and associate chief medical officer–clinical learning environment at University of Chicago Medicine. People who believe conspiracy theories and push misinformation are on one end, she said. There are also those who generally don’t believe the seriousness of the pandemic, feel their risk is minimal, or doubt the benefits of masks.
The two trains of thought resemble the distinction among parents who are antivaccine and those who are simply “vaccine hesitant,” says Arora, who co-authored a recent article about masking and misinformation that addresses antivaccine attitudes.
“While the antimask sentiment gets a lot of attention, I think it’s important to highlight there’s a lot of vocal anti-mask sentiment since most people are supportive of masks,” she said. “There might be people sitting on the fence who are just unsure about wearing a mask. That’s understandable because the science and the communication has evolved. There was a lot of early mixed messages about masking. Anytime you have confusion about the science or the science is evolving, it’s easy to have misinformation and then have that take off as myth.”
Just as antivaxxers work to swing the opinion of the vaccine hesitant, antimaskers are vying with public health advocates for the support of the mask hesitant, she said. Creating doubt in public health authorities is one way they are gaining followers. Anti-maskers often question and scrutinize past messaging about masks by public health officials, claiming that because guidance on masks has changed over time, the science behind masks and current guidance can’t be trusted, Wolynn said. Similarly, antivaxxers frequently question past actions by public health officials, such as the Tuskegee Experiment (which began in 1932), to try to poke holes in the credibility of public health officials and their advice.
Both the antimask and antivaccine movements also tend to base their resistance on a personal liberties argument, adds Jacqueline Winfield Fincher, MD, president for the American College of Physicians and an internist based in Thomson, Georgia. Antimaskers contend they should be free to decide whether to wear face coverings and that rules requiring masks infringe upon their civil liberties. Similarly, antivaxxers argue they should be free to decide whether to vaccinate their children and contend vaccine mandates violate their personal liberties.
Taking a deeper look, fear and control are two likely drivers of antimasking and antivaccine attitudes, Fincher said. Those refusing to wear masks may feel they have no control over the pandemic or its impacts, but they can control how they respond to mask-wearing requirements, she said.
Antivaccine parents often want more control over their children’s healthcare and falsely believe that vaccines are injecting something harmful into their children or may lead to harmful reactions.
“It’s a control issue and a defense mechanism,” she said. “Some people may feel helpless to deal with the pandemic or believe since it is not affecting them or their family, that it is not real. ‘If I just deny it and I don’t acknowledge facts, I don’t have to worry about it or do anything about it, and therefore I will have more control over my day-to-day life.’”
Groups fueling each other
In some cases, antimask and antivaxx groups are joining forces or adopting dual causes.
In California for instance, longtime opponents to vaccines are now objecting to mask policies as similar infringement to their bodily autonomy. Demonstrations in Texas, Idaho, and Michigan against mask mandates and other COVID-19 requirements have drawn support from anti-vaccine activists and incorporated antivaccine propaganda.
In Illinois, Million Unmasked Patriots, formally the Million Unmasked March, has received widespread attention for protesting both masks for returning schoolchildren and a future COVID-19 vaccine requirement.
A July protest planned by the antimask group triggered a letter by Arora and 500 other healthcare professionals to Illinois lawmakers decrying the group’s views and urging the state to move forward with universal masking in schools.
“What’s happening is those who are distrustful of government and public health and science are joining together,” said Arora, who coauthored a piece about the problem on KevinMD.com. “It’s important to address both movements together because they can quickly feed off each other and build in momentum. At the heart of both is really this deep skepticism of science.”
Rebresh of Million Unmasked Patriots said most of his members are not opposed to all vaccines, but rather they are opposed to “untested vaccines.” The primary concern is the inability to research long-term effects of a COVID-19 vaccine before its approval, he said.
Rebresh disagrees with the antimask movement being compared with the antivaccine movement. The two groups are “motivated by different things and a different set of circumstances drive their opinions,” he said. However, Rebresh believes that potential harm resulting from “mass vaccinations” is a valid concern. For this reason, he and his wife chose for their children to receive their vaccinations individually over a series of weeks, rather than the “kiddie cocktail of vaccines,” at a single visit, he said.
Vaccine scientist Peter Hotez, MD, PhD, said the antivaccine movement appears to have grown stronger from the pandemic fueled by fresh conspiracies and new alliances. Antivaccine sentiment has been gaining steam over the last several years and collecting more allies from the far-right, said Hotez, dean for the National School of Tropical Medicine and codirector for the Texas Children’s Hospital Center for Vaccine Development.
“Now what you’re seeing is yet another expansion this year, with antivaccine groups, under the banner of ‘health freedom,’ campaigning against social distancing and wearing masks and contact tracing,” he said. “What was an antivaccine movement has now become a full-blown antiscience movement and an anti-public health movement. It’s causing a lot of damage and I believe costing a lot of American lives.”
Neil F. Johnson, PhD, who has studied the antivaccine movement and its social media proliferation during the pandemic, said online comments by antivaxxers frequently condemn mask usage and showcase memes making fun of masks.
“In those same narratives about opposing vaccines for COVID, we see a lot of discussion against masks,” said Johnson, a physics professor at George Washington University in Washington, D.C. “If you don’t believe in the official picture of COVID, you don’t believe the policies or the advice that’s given about COVID.”
An analysis by Johnson that examined 1,300 Facebook pages found that, while antivaxxers have fewer followers than provaccine pages, antivaccine pages are more numerous, faster growing, and are more often connected to unrelated, undecided pages. Conversely, pages that advocate the benefits of vaccinations and explain the science behind immunizations are largely disconnected from such undecided communities, according to the study, published May 13 in Nature.
The study suggests the antivaccine movement is making influential strides during the pandemic and connecting with people who are undecided, while public health advocates are not building the same bridges, Johnson said.
“I think it’s hugely dangerous, because I don’t know any other moment in science or in public health when there was so much uncertainty in something affecting everybody,” he said. “Every policy that will be coming, everything depends on people buying into the official message. Once you have the seeds of doubt, that’s a very difficult thing to overcome. It’s an unprecedented challenge.”
How physicians and clinicians can help
A more aggressive approach is necessary when it comes to taking down antiscience content on social media, says Hotez. Too often, misinformation and antiscience rhetoric is allowed to linger on popular sites such as Facebook and Amazon.
Wolynn agrees. On personal or business platforms, it’s crucial to ban, hide, and delete such comments as quickly as possible, he said. On public sites, purposeful disinformation should be immediately reported to the platform.
At the same time, Wolynn said it’s essential to support those who make sound, science-based comments in social media forums.
“If you see someone who is pushing accurate, evidence-based information, and they come under attack, they should be supported and defended and empowered,” Wolynn said. “Shots Heard Round the World is doing all of those things, including galvanizing and recruiting more people to help get their voices out there.”
Expanded visibility by physicians and scientists would greatly help counter the spread of antiscience sentiment, adds Hotez.
“Too often, antiscience movements are able to flourish because scientists and physicians are invisible,” he said. “They’re too focused on either clinical practices or in the case of physician scientists, on grants and papers and not enough attention to public engagement. We’re going to have to change that around. We need to hear more from scientists directly.”
To that end, Wolynn said health care professionals, including medical students and residents, need to have formal training in communications, media, and social media as part of their education – and more support from employers to engage through social media.
“That’s where the fight is,” Wolynn said. “You can be the best diagnostician, the best clinician. You can make the right diagnosis and prescribe the right medication, but if families don’t hear what you’re saying, you’re not going to be effective. If you can’t be on the platform where they’re being influenced, we’re losing the battle.”
Speaking to your mask-hesitant patients
Concentrating on those who are uncertain about masks is particularly key for physicians and public health advocates as the pandemic continues, says Arora.
“It’s important for us to focus on the mask-hesitant who often don’t get the attention they need,” she said.
She suggests bringing up the subject of masks with patients during visits, asking about mask usage, discussing rumors they’ve heard, and emphasizing why masks are important. Be a role model by wearing a mask in your community and on social media, she added.
Some patients have real concerns about not being able to breathe through masks or anxiety disorders that can be aggravated even by the thought of wearing a mask, noted Susan R. Bailey, MD, president for the American Medical Association. Bailey, an immunologist, recently counseled a patient with a deviated nasal septum in addition to a panic disorder who was worried about wearing a mask, she said. Bailey listened to the patient’s concerns, discussed his health conditions, and proposed an alternative face covering that might make him more comfortable.
“Every patient is different,” Bailey said. “It’s important for us to remember that each person who is reluctant to wear a mask has their own reasons. It’s important for us to express some empathy – to agree with them, yes, masks are hot and inconvenient – and help understand their questions, which you may be able to answer to their satisfaction. There are patients that have legitimate questions and a physician caring about how they feel, can make all the difference.”
Physicians can also get involved with the AMA’s #MaskUp campaign, an effort to normalize mask wearing and debunk myths associated with masks. The campaign includes social media materials, slogans doctors can tweet, and profile pictures they can use on social media. The campaign’s toolkit includes images, videos, and information that physicians can share with patients and the public.
Enforcing strong mask policies at your practice and ensuring all staff are modeling appropriate mask behavior is also important, adds Fincher of the ACP. The college recently issued a policy supporting mask usage in community settings.
If a patient conveys an antimask belief, Fincher suggests not directly challenging the person’s views, but listening to them and offering objective data, discussing the science behind masks, and directing them to credible sources.
“Doctors are used to this. We recommend a lot of things to patients that they don’t want to do,” Fincher said. “If a patient feels attacked, they act defensively. But if you base your explanation in more objective terms with data, numbers, and personalize the risks and benefits of a vaccine, a healthy change in behavior, or a medication, then patients are more likely to hear your concerns and do the right thing. Having a long-term relationship with a trusted physician makes all of these issues much easier to discuss and to implement the best plan for the individual patient.”
This article first appeared on Medscape.com.
Niket Sonpal, MD, thought he’d heard most of the myths about wearing masks during the pandemic, but the recent claim from a patient was a new one for the New York City gastroenterologist.
The patient refused to wear a mask because she heard inhaling bad breath through a mask could be toxic. The woman said the rumor was circulating on Facebook. Sonpal calmly explained that breathing your own breath is not going to cause health problems, he said.
“There’s a lot of controversy on masks,” he said. “Unfortunately, it’s really just a lack of education and buy-in. Social media is the primary source of all this misinformation. These kinds of over-the-top hyperbole has basically led to a disbelief that masks are effective. The disbelief is hard to break up.”
As mask requirements have tightened amid the ongoing pandemic, debates about face coverings have emerged front and center, with a growing number of people opposing mask usage. So-called antimaskers dispute the benefits of wearing masks and many contend that face coverings decrease oxygen flow and can lead to illness. Sentiment against masks have led to protests nationwide, ignited public conflicts in some areas, and even generated lawsuits over mask mandates.
The issue presents an ongoing challenge for physicians as they strive to educate patients about the significance of masking against the flood of antimask messages on social media and beyond. Opposition to masks is particularly frustrating for health professionals who have witnessed patients, family, or friends become ill or die from the virus. Refusing to mask and failing to social distance have been linked to the rapid spread of the coronavirus and subsequent deaths.
“I have had colleagues pass away, and it’s extremely disheartening and frustrating to see science so easily disregarded,” Sonpal said. “Masks save lives and protect people and not wearing them is simply a lack of respect, not just for your fellow colleagues, but for a member of your species.”
Michael Rebresh, who helped create the antimask group Million Unmasked Patriots, says his group’s objections to masks are rational and reasonable. The group, which has more than 8,000 members, formed in response to guidance by Illinois state officials that children would only be allowed to return to school wearing a mask.
“Our objections are to the fact that masks on children in school have a greater propensity to make children sick from breathing in bacteria that forms on the inner layer of a mask worn for hours on end,” Rebresh said. “We have an objection to the increase of CO2 intake and a decrease in oxygen flow for kids who need all the oxygen they can get during a learning environment. We recognized the masking of ourselves and kids for what it is: A political move to separate the two parties in our November election and define and create division between the two.”
Million Unmasked Patriots is one of dozens of antimask groups on social media platforms such as Facebook, Instagram, and TikTok. In July, Facebook suspended one such group, Unmasking America, which boasts 9,600 members, for posting repeated claims that face masks obstruct oxygen flow and have negative mental health effects.
Experts say the antiscience rhetoric is far from new. The antimask movement in many ways, shares similarities with that of the anti-vaccine movement, says Todd Wolynn, MD, a Pittsburgh pediatrician and cofounder of Shots Heard Round the World, an organization that defends vaccine advocates against coordinated online attacks by antivaxxers.
“A lot of it is conspiracy-laden,” said Wolynn of the disinformation. “That Dr. [Anthony] Fauci somehow helped construct the pandemic and that it’s not real. That Bill Gates is funding the vaccine so he can inject people with microchips. All sorts of really out-there, ungrounded conspiracy theories. If you had Venn diagram of antimask and antivaxx, I would say there’s clearly overlap.”
Parallels between antimaskers, antivaxxers
Opponents to masks fall on a spectrum, explains Vineet Arora, MD, a hospitalist and associate chief medical officer–clinical learning environment at University of Chicago Medicine. People who believe conspiracy theories and push misinformation are on one end, she said. There are also those who generally don’t believe the seriousness of the pandemic, feel their risk is minimal, or doubt the benefits of masks.
The two trains of thought resemble the distinction among parents who are antivaccine and those who are simply “vaccine hesitant,” says Arora, who co-authored a recent article about masking and misinformation that addresses antivaccine attitudes.
“While the antimask sentiment gets a lot of attention, I think it’s important to highlight there’s a lot of vocal anti-mask sentiment since most people are supportive of masks,” she said. “There might be people sitting on the fence who are just unsure about wearing a mask. That’s understandable because the science and the communication has evolved. There was a lot of early mixed messages about masking. Anytime you have confusion about the science or the science is evolving, it’s easy to have misinformation and then have that take off as myth.”
Just as antivaxxers work to swing the opinion of the vaccine hesitant, antimaskers are vying with public health advocates for the support of the mask hesitant, she said. Creating doubt in public health authorities is one way they are gaining followers. Anti-maskers often question and scrutinize past messaging about masks by public health officials, claiming that because guidance on masks has changed over time, the science behind masks and current guidance can’t be trusted, Wolynn said. Similarly, antivaxxers frequently question past actions by public health officials, such as the Tuskegee Experiment (which began in 1932), to try to poke holes in the credibility of public health officials and their advice.
Both the antimask and antivaccine movements also tend to base their resistance on a personal liberties argument, adds Jacqueline Winfield Fincher, MD, president for the American College of Physicians and an internist based in Thomson, Georgia. Antimaskers contend they should be free to decide whether to wear face coverings and that rules requiring masks infringe upon their civil liberties. Similarly, antivaxxers argue they should be free to decide whether to vaccinate their children and contend vaccine mandates violate their personal liberties.
Taking a deeper look, fear and control are two likely drivers of antimasking and antivaccine attitudes, Fincher said. Those refusing to wear masks may feel they have no control over the pandemic or its impacts, but they can control how they respond to mask-wearing requirements, she said.
Antivaccine parents often want more control over their children’s healthcare and falsely believe that vaccines are injecting something harmful into their children or may lead to harmful reactions.
“It’s a control issue and a defense mechanism,” she said. “Some people may feel helpless to deal with the pandemic or believe since it is not affecting them or their family, that it is not real. ‘If I just deny it and I don’t acknowledge facts, I don’t have to worry about it or do anything about it, and therefore I will have more control over my day-to-day life.’”
Groups fueling each other
In some cases, antimask and antivaxx groups are joining forces or adopting dual causes.
In California for instance, longtime opponents to vaccines are now objecting to mask policies as similar infringement to their bodily autonomy. Demonstrations in Texas, Idaho, and Michigan against mask mandates and other COVID-19 requirements have drawn support from anti-vaccine activists and incorporated antivaccine propaganda.
In Illinois, Million Unmasked Patriots, formally the Million Unmasked March, has received widespread attention for protesting both masks for returning schoolchildren and a future COVID-19 vaccine requirement.
A July protest planned by the antimask group triggered a letter by Arora and 500 other healthcare professionals to Illinois lawmakers decrying the group’s views and urging the state to move forward with universal masking in schools.
“What’s happening is those who are distrustful of government and public health and science are joining together,” said Arora, who coauthored a piece about the problem on KevinMD.com. “It’s important to address both movements together because they can quickly feed off each other and build in momentum. At the heart of both is really this deep skepticism of science.”
Rebresh of Million Unmasked Patriots said most of his members are not opposed to all vaccines, but rather they are opposed to “untested vaccines.” The primary concern is the inability to research long-term effects of a COVID-19 vaccine before its approval, he said.
Rebresh disagrees with the antimask movement being compared with the antivaccine movement. The two groups are “motivated by different things and a different set of circumstances drive their opinions,” he said. However, Rebresh believes that potential harm resulting from “mass vaccinations” is a valid concern. For this reason, he and his wife chose for their children to receive their vaccinations individually over a series of weeks, rather than the “kiddie cocktail of vaccines,” at a single visit, he said.
Vaccine scientist Peter Hotez, MD, PhD, said the antivaccine movement appears to have grown stronger from the pandemic fueled by fresh conspiracies and new alliances. Antivaccine sentiment has been gaining steam over the last several years and collecting more allies from the far-right, said Hotez, dean for the National School of Tropical Medicine and codirector for the Texas Children’s Hospital Center for Vaccine Development.
“Now what you’re seeing is yet another expansion this year, with antivaccine groups, under the banner of ‘health freedom,’ campaigning against social distancing and wearing masks and contact tracing,” he said. “What was an antivaccine movement has now become a full-blown antiscience movement and an anti-public health movement. It’s causing a lot of damage and I believe costing a lot of American lives.”
Neil F. Johnson, PhD, who has studied the antivaccine movement and its social media proliferation during the pandemic, said online comments by antivaxxers frequently condemn mask usage and showcase memes making fun of masks.
“In those same narratives about opposing vaccines for COVID, we see a lot of discussion against masks,” said Johnson, a physics professor at George Washington University in Washington, D.C. “If you don’t believe in the official picture of COVID, you don’t believe the policies or the advice that’s given about COVID.”
An analysis by Johnson that examined 1,300 Facebook pages found that, while antivaxxers have fewer followers than provaccine pages, antivaccine pages are more numerous, faster growing, and are more often connected to unrelated, undecided pages. Conversely, pages that advocate the benefits of vaccinations and explain the science behind immunizations are largely disconnected from such undecided communities, according to the study, published May 13 in Nature.
The study suggests the antivaccine movement is making influential strides during the pandemic and connecting with people who are undecided, while public health advocates are not building the same bridges, Johnson said.
“I think it’s hugely dangerous, because I don’t know any other moment in science or in public health when there was so much uncertainty in something affecting everybody,” he said. “Every policy that will be coming, everything depends on people buying into the official message. Once you have the seeds of doubt, that’s a very difficult thing to overcome. It’s an unprecedented challenge.”
How physicians and clinicians can help
A more aggressive approach is necessary when it comes to taking down antiscience content on social media, says Hotez. Too often, misinformation and antiscience rhetoric is allowed to linger on popular sites such as Facebook and Amazon.
Wolynn agrees. On personal or business platforms, it’s crucial to ban, hide, and delete such comments as quickly as possible, he said. On public sites, purposeful disinformation should be immediately reported to the platform.
At the same time, Wolynn said it’s essential to support those who make sound, science-based comments in social media forums.
“If you see someone who is pushing accurate, evidence-based information, and they come under attack, they should be supported and defended and empowered,” Wolynn said. “Shots Heard Round the World is doing all of those things, including galvanizing and recruiting more people to help get their voices out there.”
Expanded visibility by physicians and scientists would greatly help counter the spread of antiscience sentiment, adds Hotez.
“Too often, antiscience movements are able to flourish because scientists and physicians are invisible,” he said. “They’re too focused on either clinical practices or in the case of physician scientists, on grants and papers and not enough attention to public engagement. We’re going to have to change that around. We need to hear more from scientists directly.”
To that end, Wolynn said health care professionals, including medical students and residents, need to have formal training in communications, media, and social media as part of their education – and more support from employers to engage through social media.
“That’s where the fight is,” Wolynn said. “You can be the best diagnostician, the best clinician. You can make the right diagnosis and prescribe the right medication, but if families don’t hear what you’re saying, you’re not going to be effective. If you can’t be on the platform where they’re being influenced, we’re losing the battle.”
Speaking to your mask-hesitant patients
Concentrating on those who are uncertain about masks is particularly key for physicians and public health advocates as the pandemic continues, says Arora.
“It’s important for us to focus on the mask-hesitant who often don’t get the attention they need,” she said.
She suggests bringing up the subject of masks with patients during visits, asking about mask usage, discussing rumors they’ve heard, and emphasizing why masks are important. Be a role model by wearing a mask in your community and on social media, she added.
Some patients have real concerns about not being able to breathe through masks or anxiety disorders that can be aggravated even by the thought of wearing a mask, noted Susan R. Bailey, MD, president for the American Medical Association. Bailey, an immunologist, recently counseled a patient with a deviated nasal septum in addition to a panic disorder who was worried about wearing a mask, she said. Bailey listened to the patient’s concerns, discussed his health conditions, and proposed an alternative face covering that might make him more comfortable.
“Every patient is different,” Bailey said. “It’s important for us to remember that each person who is reluctant to wear a mask has their own reasons. It’s important for us to express some empathy – to agree with them, yes, masks are hot and inconvenient – and help understand their questions, which you may be able to answer to their satisfaction. There are patients that have legitimate questions and a physician caring about how they feel, can make all the difference.”
Physicians can also get involved with the AMA’s #MaskUp campaign, an effort to normalize mask wearing and debunk myths associated with masks. The campaign includes social media materials, slogans doctors can tweet, and profile pictures they can use on social media. The campaign’s toolkit includes images, videos, and information that physicians can share with patients and the public.
Enforcing strong mask policies at your practice and ensuring all staff are modeling appropriate mask behavior is also important, adds Fincher of the ACP. The college recently issued a policy supporting mask usage in community settings.
If a patient conveys an antimask belief, Fincher suggests not directly challenging the person’s views, but listening to them and offering objective data, discussing the science behind masks, and directing them to credible sources.
“Doctors are used to this. We recommend a lot of things to patients that they don’t want to do,” Fincher said. “If a patient feels attacked, they act defensively. But if you base your explanation in more objective terms with data, numbers, and personalize the risks and benefits of a vaccine, a healthy change in behavior, or a medication, then patients are more likely to hear your concerns and do the right thing. Having a long-term relationship with a trusted physician makes all of these issues much easier to discuss and to implement the best plan for the individual patient.”
This article first appeared on Medscape.com.
Mounting data support COVID-19 acute pancreatitis
Mounting data support acute pancreatitis as one possible GI manifestation of COVID-19, according to investigators.
While previous case reports suggested that infection with SARS-CoV2 may lead to pancreatitis, this retrospective analysis, which is the largest to date, is the first to offer substantial evidence for this claim, reported lead author Sumant Inamdar, MBBS, of the University of Arkansas, Little Rock, and colleagues.
“It has become increasingly clear that COVID-19 has systemic effects that also includes the gastrointestinal and pancreaticobiliary systems,” the investigators wrote in Gastroenterology. “As islet cells of the pancreas contain ACE2 receptor proteins, SARS-CoV2 can bind to these receptors and cause pancreatic injury.”
For the present analysis, Dr. Inamdar and colleagues reviewed charts from 48,012 patients who were hospitalized in New York between March and June of this year. While pancreatitis is usually diagnosed based on two out of three criteria, disease classification in the study required all three: characteristic upper abdominal pain upon admission, lipase greater than three times the upper limit of normal, and evidence of pancreatitis on cross-sectional imaging.
“[B]y including all three criteria for pancreatitis in our definition, we may be underestimating the rate of pancreatitis,” the investigators wrote. “However, we felt including diagnostic lipase levels and imaging was important for the accuracy of the diagnosis.”
Primary outcomes included mechanical ventilation, length of stay, development of pancreatic necrosis, and mortality. Outcomes were compared between patients with and without COVID-19.
Out of 48,012 hospitalized patients, 11,883 (24.75%) tested positive for SARS-CoV2. Across the entire population, 189 patients had pancreatitis (0.39%), and of these, 32 (17%) also had COVID-19. This translates to a point prevalence for pancreatitis of 0.27% for patients hospitalized with COVID-19.
Among patients with pancreatitis who did not have COVID-19, the most common etiologies for pancreatitis were gallstones (34%) and alcohol (37%), compared with just 16% and 6% of SARS-CoV2-positive cases of pancreatitis, respectively. Idiopathic pancreatitis was significantly more common among patients with COVID-19 than those without (69% vs 21%; P less than .0001).
Black or Hispanic patients with pancreatitis were 4-5 times more likely to have COVID-19 than patients with pancreatitis who were white. Across all races/ethnicities, patients with pancreatitis and COVID-19 more often required mechanical ventilation (odds ratio [OR], 5.65) and longer hospital stays (OR, 3.22), compared with those who had pancreatitis alone. While rates of mortality and pancreatic necrosis showed similar trends, associations with COVID-19 were not statistically significant.
“These findings support the notion that pancreatitis should be included in the list of GI manifestations of COVID-19,” the investigators wrote.
When caring for patients with COVID-19, Dr. Inamdar and colleagues recommended that clinicians pay close attention to any history of abdominal pain, and consider testing serum lipase levels.
“Further large studies are needed to confirm our findings,” they concluded.
Avinash Ketwaroo, MD, of Baylor College of Medicine in Houston, agreed that more work is needed; in the meantime, he suggested that evidence is now strong enough for clinicians to take notice.
“Overall, this study adds further weight to COVID-19 acute pancreatitis,” he said. “Larger studies, and convincing pathophysiologic data, will be needed to confirm COVID-19 as a cause of acute pancreatitis. However, there appears to be enough circumstantial evidence to consider a COVID-19 diagnosis in patients presenting with acute pancreatitis.”
He noted that the new clinical evidence also stands on a solid theoretical foundation.
“Viruses, especially mumps and coxsackie, have long been known to cause acute pancreatitis,” he said. “Additionally, the ACE2 receptor is present on pancreatic beta-cells and may mediate COVID-19 induced pancreatitis.”
Along with larger observational studies, Dr. Ketwaroo suggested that a number of interventional questions remain unanswered.
“While most acute pancreatitis is treated with supportive care, could proven therapies for COVID-19, such as steroids, also mitigate COVID-19 acute pancreatitis?” he asked. “Is COVID-19 a cofactor for acute pancreatitis caused by alcohol or endoscopic retrograde cholangiopancreatography? We await further information from an active area of research.”
The investigators disclosed relationships with Boston Scientific, Olympus, Fujifilm, and others.
SOURCE: Inamdar S et al. Gastroenterology. 2020 Aug 26. doi: 10.1053/j.gastro.2020.08.044.
Share AGA GI Patient Center content to help your patients understand the symptoms and complications of pancreatitis at http://ow.ly/j1AN30r8ZDa.
This story was updated on 9/14/2020.
Mounting data support acute pancreatitis as one possible GI manifestation of COVID-19, according to investigators.
While previous case reports suggested that infection with SARS-CoV2 may lead to pancreatitis, this retrospective analysis, which is the largest to date, is the first to offer substantial evidence for this claim, reported lead author Sumant Inamdar, MBBS, of the University of Arkansas, Little Rock, and colleagues.
“It has become increasingly clear that COVID-19 has systemic effects that also includes the gastrointestinal and pancreaticobiliary systems,” the investigators wrote in Gastroenterology. “As islet cells of the pancreas contain ACE2 receptor proteins, SARS-CoV2 can bind to these receptors and cause pancreatic injury.”
For the present analysis, Dr. Inamdar and colleagues reviewed charts from 48,012 patients who were hospitalized in New York between March and June of this year. While pancreatitis is usually diagnosed based on two out of three criteria, disease classification in the study required all three: characteristic upper abdominal pain upon admission, lipase greater than three times the upper limit of normal, and evidence of pancreatitis on cross-sectional imaging.
“[B]y including all three criteria for pancreatitis in our definition, we may be underestimating the rate of pancreatitis,” the investigators wrote. “However, we felt including diagnostic lipase levels and imaging was important for the accuracy of the diagnosis.”
Primary outcomes included mechanical ventilation, length of stay, development of pancreatic necrosis, and mortality. Outcomes were compared between patients with and without COVID-19.
Out of 48,012 hospitalized patients, 11,883 (24.75%) tested positive for SARS-CoV2. Across the entire population, 189 patients had pancreatitis (0.39%), and of these, 32 (17%) also had COVID-19. This translates to a point prevalence for pancreatitis of 0.27% for patients hospitalized with COVID-19.
Among patients with pancreatitis who did not have COVID-19, the most common etiologies for pancreatitis were gallstones (34%) and alcohol (37%), compared with just 16% and 6% of SARS-CoV2-positive cases of pancreatitis, respectively. Idiopathic pancreatitis was significantly more common among patients with COVID-19 than those without (69% vs 21%; P less than .0001).
Black or Hispanic patients with pancreatitis were 4-5 times more likely to have COVID-19 than patients with pancreatitis who were white. Across all races/ethnicities, patients with pancreatitis and COVID-19 more often required mechanical ventilation (odds ratio [OR], 5.65) and longer hospital stays (OR, 3.22), compared with those who had pancreatitis alone. While rates of mortality and pancreatic necrosis showed similar trends, associations with COVID-19 were not statistically significant.
“These findings support the notion that pancreatitis should be included in the list of GI manifestations of COVID-19,” the investigators wrote.
When caring for patients with COVID-19, Dr. Inamdar and colleagues recommended that clinicians pay close attention to any history of abdominal pain, and consider testing serum lipase levels.
“Further large studies are needed to confirm our findings,” they concluded.
Avinash Ketwaroo, MD, of Baylor College of Medicine in Houston, agreed that more work is needed; in the meantime, he suggested that evidence is now strong enough for clinicians to take notice.
“Overall, this study adds further weight to COVID-19 acute pancreatitis,” he said. “Larger studies, and convincing pathophysiologic data, will be needed to confirm COVID-19 as a cause of acute pancreatitis. However, there appears to be enough circumstantial evidence to consider a COVID-19 diagnosis in patients presenting with acute pancreatitis.”
He noted that the new clinical evidence also stands on a solid theoretical foundation.
“Viruses, especially mumps and coxsackie, have long been known to cause acute pancreatitis,” he said. “Additionally, the ACE2 receptor is present on pancreatic beta-cells and may mediate COVID-19 induced pancreatitis.”
Along with larger observational studies, Dr. Ketwaroo suggested that a number of interventional questions remain unanswered.
“While most acute pancreatitis is treated with supportive care, could proven therapies for COVID-19, such as steroids, also mitigate COVID-19 acute pancreatitis?” he asked. “Is COVID-19 a cofactor for acute pancreatitis caused by alcohol or endoscopic retrograde cholangiopancreatography? We await further information from an active area of research.”
The investigators disclosed relationships with Boston Scientific, Olympus, Fujifilm, and others.
SOURCE: Inamdar S et al. Gastroenterology. 2020 Aug 26. doi: 10.1053/j.gastro.2020.08.044.
Share AGA GI Patient Center content to help your patients understand the symptoms and complications of pancreatitis at http://ow.ly/j1AN30r8ZDa.
This story was updated on 9/14/2020.
Mounting data support acute pancreatitis as one possible GI manifestation of COVID-19, according to investigators.
While previous case reports suggested that infection with SARS-CoV2 may lead to pancreatitis, this retrospective analysis, which is the largest to date, is the first to offer substantial evidence for this claim, reported lead author Sumant Inamdar, MBBS, of the University of Arkansas, Little Rock, and colleagues.
“It has become increasingly clear that COVID-19 has systemic effects that also includes the gastrointestinal and pancreaticobiliary systems,” the investigators wrote in Gastroenterology. “As islet cells of the pancreas contain ACE2 receptor proteins, SARS-CoV2 can bind to these receptors and cause pancreatic injury.”
For the present analysis, Dr. Inamdar and colleagues reviewed charts from 48,012 patients who were hospitalized in New York between March and June of this year. While pancreatitis is usually diagnosed based on two out of three criteria, disease classification in the study required all three: characteristic upper abdominal pain upon admission, lipase greater than three times the upper limit of normal, and evidence of pancreatitis on cross-sectional imaging.
“[B]y including all three criteria for pancreatitis in our definition, we may be underestimating the rate of pancreatitis,” the investigators wrote. “However, we felt including diagnostic lipase levels and imaging was important for the accuracy of the diagnosis.”
Primary outcomes included mechanical ventilation, length of stay, development of pancreatic necrosis, and mortality. Outcomes were compared between patients with and without COVID-19.
Out of 48,012 hospitalized patients, 11,883 (24.75%) tested positive for SARS-CoV2. Across the entire population, 189 patients had pancreatitis (0.39%), and of these, 32 (17%) also had COVID-19. This translates to a point prevalence for pancreatitis of 0.27% for patients hospitalized with COVID-19.
Among patients with pancreatitis who did not have COVID-19, the most common etiologies for pancreatitis were gallstones (34%) and alcohol (37%), compared with just 16% and 6% of SARS-CoV2-positive cases of pancreatitis, respectively. Idiopathic pancreatitis was significantly more common among patients with COVID-19 than those without (69% vs 21%; P less than .0001).
Black or Hispanic patients with pancreatitis were 4-5 times more likely to have COVID-19 than patients with pancreatitis who were white. Across all races/ethnicities, patients with pancreatitis and COVID-19 more often required mechanical ventilation (odds ratio [OR], 5.65) and longer hospital stays (OR, 3.22), compared with those who had pancreatitis alone. While rates of mortality and pancreatic necrosis showed similar trends, associations with COVID-19 were not statistically significant.
“These findings support the notion that pancreatitis should be included in the list of GI manifestations of COVID-19,” the investigators wrote.
When caring for patients with COVID-19, Dr. Inamdar and colleagues recommended that clinicians pay close attention to any history of abdominal pain, and consider testing serum lipase levels.
“Further large studies are needed to confirm our findings,” they concluded.
Avinash Ketwaroo, MD, of Baylor College of Medicine in Houston, agreed that more work is needed; in the meantime, he suggested that evidence is now strong enough for clinicians to take notice.
“Overall, this study adds further weight to COVID-19 acute pancreatitis,” he said. “Larger studies, and convincing pathophysiologic data, will be needed to confirm COVID-19 as a cause of acute pancreatitis. However, there appears to be enough circumstantial evidence to consider a COVID-19 diagnosis in patients presenting with acute pancreatitis.”
He noted that the new clinical evidence also stands on a solid theoretical foundation.
“Viruses, especially mumps and coxsackie, have long been known to cause acute pancreatitis,” he said. “Additionally, the ACE2 receptor is present on pancreatic beta-cells and may mediate COVID-19 induced pancreatitis.”
Along with larger observational studies, Dr. Ketwaroo suggested that a number of interventional questions remain unanswered.
“While most acute pancreatitis is treated with supportive care, could proven therapies for COVID-19, such as steroids, also mitigate COVID-19 acute pancreatitis?” he asked. “Is COVID-19 a cofactor for acute pancreatitis caused by alcohol or endoscopic retrograde cholangiopancreatography? We await further information from an active area of research.”
The investigators disclosed relationships with Boston Scientific, Olympus, Fujifilm, and others.
SOURCE: Inamdar S et al. Gastroenterology. 2020 Aug 26. doi: 10.1053/j.gastro.2020.08.044.
Share AGA GI Patient Center content to help your patients understand the symptoms and complications of pancreatitis at http://ow.ly/j1AN30r8ZDa.
This story was updated on 9/14/2020.
FROM GASTROENTEROLOGY
U.S. tops 500,000 COVID-19 cases in children
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
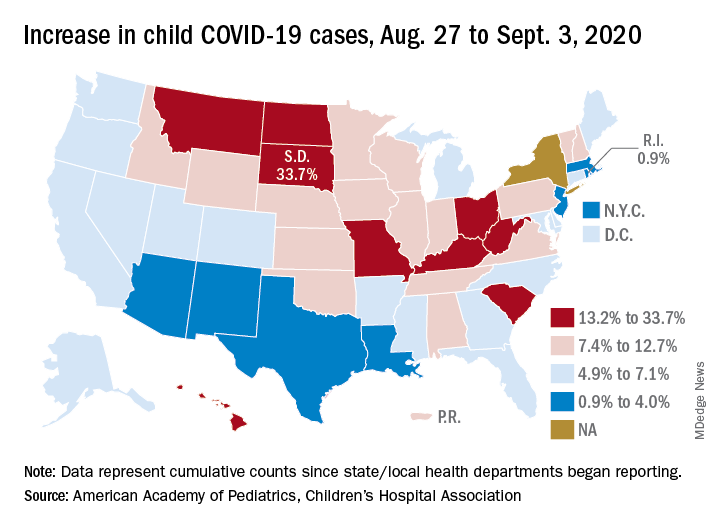
States have reported 513,415 cases of COVID-19 in children since the beginning of the pandemic, with almost 37,000 coming in the last week, the AAP and the CHA said Sept. 8 in the weekly report. That figure includes New York City – the rest of New York State is not reporting ages for COVID-19 patients – as well as Puerto Rico, the District of Columbia, and Guam.
“These numbers are a chilling reminder of why we need to take this virus seriously,” AAP President Sara Goza, MD, said in a written statement.
Children now represent 9.8% of the almost 5.3 million cases that have been reported in Americans of all ages. The proportion of child cases has continued to increase as the pandemic has progressed – it was 8.0% as of mid-July and 5.2% in early June, the data show.
“Throughout the summer, surges in the virus have occurred in Southern, Western, and Midwestern states,” the AAP statement said.
The latest AAP/CHA report shows that, from Aug. 27 to Sept. 3, the total number of child cases jumped by 33.7% in South Dakota, more than any other state. North Dakota was next at 22.7%, followed by Hawaii (18.1%), Missouri (16.8%), and Kentucky (16.4%).
“This rapid rise in positive cases occurred over the summer, and as the weather cools, we know people will spend more time indoors,” said Sean O’Leary, MD, MPH, vice chair of the AAP Committee on Infectious Diseases. “The goal is to get children back into schools for in-person learning, but in many communities, this is not possible as the virus spreads unchecked.”
The smallest increase over the last week, just 0.9%, came in Rhode Island, with Massachusetts just a bit higher at 1.0%. Also at the low end of the increase scale are Arizona (3.3%) and Louisiana (4.0%), two states that have very high rates of cumulative cases: 1,380 per 100,000 children for Arizona and 1,234 per 100,000 for Louisiana, the report said.
To give those figures some context, Tennessee has the highest cumulative count of any state at 1,553 cases per 100,000 children and Vermont has the lowest at 151, based on the data gathered by the AAP and CHA.
“While much remains unknown about COVID-19, we do know that the spread among children reflects what is happening in the broader communities. A disproportionate number of cases are reported in Black and Hispanic children and in places where there is high poverty. We must work harder to address societal inequities that contribute to these disparities,” Dr. Goza said.
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

States have reported 513,415 cases of COVID-19 in children since the beginning of the pandemic, with almost 37,000 coming in the last week, the AAP and the CHA said Sept. 8 in the weekly report. That figure includes New York City – the rest of New York State is not reporting ages for COVID-19 patients – as well as Puerto Rico, the District of Columbia, and Guam.
“These numbers are a chilling reminder of why we need to take this virus seriously,” AAP President Sara Goza, MD, said in a written statement.
Children now represent 9.8% of the almost 5.3 million cases that have been reported in Americans of all ages. The proportion of child cases has continued to increase as the pandemic has progressed – it was 8.0% as of mid-July and 5.2% in early June, the data show.
“Throughout the summer, surges in the virus have occurred in Southern, Western, and Midwestern states,” the AAP statement said.
The latest AAP/CHA report shows that, from Aug. 27 to Sept. 3, the total number of child cases jumped by 33.7% in South Dakota, more than any other state. North Dakota was next at 22.7%, followed by Hawaii (18.1%), Missouri (16.8%), and Kentucky (16.4%).
“This rapid rise in positive cases occurred over the summer, and as the weather cools, we know people will spend more time indoors,” said Sean O’Leary, MD, MPH, vice chair of the AAP Committee on Infectious Diseases. “The goal is to get children back into schools for in-person learning, but in many communities, this is not possible as the virus spreads unchecked.”
The smallest increase over the last week, just 0.9%, came in Rhode Island, with Massachusetts just a bit higher at 1.0%. Also at the low end of the increase scale are Arizona (3.3%) and Louisiana (4.0%), two states that have very high rates of cumulative cases: 1,380 per 100,000 children for Arizona and 1,234 per 100,000 for Louisiana, the report said.
To give those figures some context, Tennessee has the highest cumulative count of any state at 1,553 cases per 100,000 children and Vermont has the lowest at 151, based on the data gathered by the AAP and CHA.
“While much remains unknown about COVID-19, we do know that the spread among children reflects what is happening in the broader communities. A disproportionate number of cases are reported in Black and Hispanic children and in places where there is high poverty. We must work harder to address societal inequities that contribute to these disparities,” Dr. Goza said.
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

States have reported 513,415 cases of COVID-19 in children since the beginning of the pandemic, with almost 37,000 coming in the last week, the AAP and the CHA said Sept. 8 in the weekly report. That figure includes New York City – the rest of New York State is not reporting ages for COVID-19 patients – as well as Puerto Rico, the District of Columbia, and Guam.
“These numbers are a chilling reminder of why we need to take this virus seriously,” AAP President Sara Goza, MD, said in a written statement.
Children now represent 9.8% of the almost 5.3 million cases that have been reported in Americans of all ages. The proportion of child cases has continued to increase as the pandemic has progressed – it was 8.0% as of mid-July and 5.2% in early June, the data show.
“Throughout the summer, surges in the virus have occurred in Southern, Western, and Midwestern states,” the AAP statement said.
The latest AAP/CHA report shows that, from Aug. 27 to Sept. 3, the total number of child cases jumped by 33.7% in South Dakota, more than any other state. North Dakota was next at 22.7%, followed by Hawaii (18.1%), Missouri (16.8%), and Kentucky (16.4%).
“This rapid rise in positive cases occurred over the summer, and as the weather cools, we know people will spend more time indoors,” said Sean O’Leary, MD, MPH, vice chair of the AAP Committee on Infectious Diseases. “The goal is to get children back into schools for in-person learning, but in many communities, this is not possible as the virus spreads unchecked.”
The smallest increase over the last week, just 0.9%, came in Rhode Island, with Massachusetts just a bit higher at 1.0%. Also at the low end of the increase scale are Arizona (3.3%) and Louisiana (4.0%), two states that have very high rates of cumulative cases: 1,380 per 100,000 children for Arizona and 1,234 per 100,000 for Louisiana, the report said.
To give those figures some context, Tennessee has the highest cumulative count of any state at 1,553 cases per 100,000 children and Vermont has the lowest at 151, based on the data gathered by the AAP and CHA.
“While much remains unknown about COVID-19, we do know that the spread among children reflects what is happening in the broader communities. A disproportionate number of cases are reported in Black and Hispanic children and in places where there is high poverty. We must work harder to address societal inequities that contribute to these disparities,” Dr. Goza said.
Mounting data support COVID-19 acute pancreatitis
Mounting data support acute pancreatitis as one possible GI manifestation of COVID-19, according to investigators.
While previous case reports suggested that infection with SARS-CoV2 may lead to pancreatitis, this retrospective analysis, which is the largest to date, is the first to offer substantial evidence for this claim, reported lead author Sumant Inamdar, MBBS, of the University of Arkansas, Little Rock, and colleagues.
“It has become increasingly clear that COVID-19 has systemic effects that also includes the gastrointestinal and pancreaticobiliary systems,” the investigators wrote in Gastroenterology. “As islet cells of the pancreas contain ACE2 receptor proteins, SARS-CoV2 can bind to these receptors and cause pancreatic injury.”
For the present analysis, Dr. Inamdar and colleagues reviewed charts from 48,012 patients who were hospitalized in New York between March and June of this year. While pancreatitis is usually diagnosed based on two out of three criteria, disease classification in the study required all three: characteristic upper abdominal pain upon admission, lipase greater than three times the upper limit of normal, and evidence of pancreatitis on cross-sectional imaging.
“[B]y including all three criteria for pancreatitis in our definition, we may be underestimating the rate of pancreatitis,” the investigators wrote. “However, we felt including diagnostic lipase levels and imaging was important for the accuracy of the diagnosis.”
Primary outcomes included mechanical ventilation, length of stay, development of pancreatic necrosis, and mortality. Outcomes were compared between patients with and without COVID-19.
Out of 48,012 hospitalized patients, 11,883 (24.75%) tested positive for SARS-CoV2. Across the entire population, 189 patients had pancreatitis (0.39%), and of these, 32 (17%) also had COVID-19. This translates to a point prevalence for pancreatitis of 0.27% for patients hospitalized with COVID-19.
Among patients with pancreatitis who did not have COVID-19, the most common etiologies for pancreatitis were gallstones (34%) and alcohol (37%), compared with just 16% and 6% of SARS-CoV2-positive cases of pancreatitis, respectively. Idiopathic pancreatitis was significantly more common among patients with COVID-19 than those without (69% vs 21%; P less than .0001).
Black or Hispanic patients with pancreatitis were 4-5 times more likely to have COVID-19 than patients with pancreatitis who were white. Across all races/ethnicities, patients with pancreatitis and COVID-19 more often required mechanical ventilation (odds ratio [OR], 5.65) and longer hospital stays (OR, 3.22), compared with those who had pancreatitis alone. While rates of mortality and pancreatic necrosis showed similar trends, associations with COVID-19 were not statistically significant.
“These findings support the notion that pancreatitis should be included in the list of GI manifestations of COVID-19,” the investigators wrote.
When caring for patients with COVID-19, Dr. Inamdar and colleagues recommended that clinicians pay close attention to any history of abdominal pain, and consider testing serum lipase levels.
“Further large studies are needed to confirm our findings,” they concluded.
Gyanprakash Avinash Ketwaroo, MD, of Baylor College of Medicine in Houston, agreed that more work is needed; in the meantime, he suggested that evidence is now strong enough for clinicians to take notice.
“Overall, this study adds further weight to COVID-19 acute pancreatitis,” he said. “Larger studies, and convincing pathophysiologic data, will be needed to confirm COVID-19 as a cause of acute pancreatitis. However, there appears to be enough circumstantial evidence to consider a COVID-19 diagnosis in patients presenting with acute pancreatitis.”
He noted that the new clinical evidence also stands on a solid theoretical foundation.
“Viruses, especially mumps and measles, have long been known to cause acute pancreatitis,” he said. “Additionally, the ACE2 receptor is present on pancreatic beta-cells and may mediate COVID-19 induced pancreatitis.”
Along with larger observational studies, Dr. Ketwaroo suggested that a number of interventional questions remain unanswered.
“While most acute pancreatitis is treated with supportive care, could proven therapies for COVID-19, such as steroids, also mitigate COVID-19 acute pancreatitis?” he asked. “Is COVID-19 a cofactor for acute pancreatitis caused by alcohol or endoscopic retrograde cholangiopancreatography? We await further information from an active area of research.”
The investigators disclosed relationships with Boston Scientific, Olympus, Fujifilm, and others.
SOURCE: Inamdar S et al. Gastroenterology. 2020 Aug 26. doi: 10.1053/j.gastro.2020.08.044.
Mounting data support acute pancreatitis as one possible GI manifestation of COVID-19, according to investigators.
While previous case reports suggested that infection with SARS-CoV2 may lead to pancreatitis, this retrospective analysis, which is the largest to date, is the first to offer substantial evidence for this claim, reported lead author Sumant Inamdar, MBBS, of the University of Arkansas, Little Rock, and colleagues.
“It has become increasingly clear that COVID-19 has systemic effects that also includes the gastrointestinal and pancreaticobiliary systems,” the investigators wrote in Gastroenterology. “As islet cells of the pancreas contain ACE2 receptor proteins, SARS-CoV2 can bind to these receptors and cause pancreatic injury.”
For the present analysis, Dr. Inamdar and colleagues reviewed charts from 48,012 patients who were hospitalized in New York between March and June of this year. While pancreatitis is usually diagnosed based on two out of three criteria, disease classification in the study required all three: characteristic upper abdominal pain upon admission, lipase greater than three times the upper limit of normal, and evidence of pancreatitis on cross-sectional imaging.
“[B]y including all three criteria for pancreatitis in our definition, we may be underestimating the rate of pancreatitis,” the investigators wrote. “However, we felt including diagnostic lipase levels and imaging was important for the accuracy of the diagnosis.”
Primary outcomes included mechanical ventilation, length of stay, development of pancreatic necrosis, and mortality. Outcomes were compared between patients with and without COVID-19.
Out of 48,012 hospitalized patients, 11,883 (24.75%) tested positive for SARS-CoV2. Across the entire population, 189 patients had pancreatitis (0.39%), and of these, 32 (17%) also had COVID-19. This translates to a point prevalence for pancreatitis of 0.27% for patients hospitalized with COVID-19.
Among patients with pancreatitis who did not have COVID-19, the most common etiologies for pancreatitis were gallstones (34%) and alcohol (37%), compared with just 16% and 6% of SARS-CoV2-positive cases of pancreatitis, respectively. Idiopathic pancreatitis was significantly more common among patients with COVID-19 than those without (69% vs 21%; P less than .0001).
Black or Hispanic patients with pancreatitis were 4-5 times more likely to have COVID-19 than patients with pancreatitis who were white. Across all races/ethnicities, patients with pancreatitis and COVID-19 more often required mechanical ventilation (odds ratio [OR], 5.65) and longer hospital stays (OR, 3.22), compared with those who had pancreatitis alone. While rates of mortality and pancreatic necrosis showed similar trends, associations with COVID-19 were not statistically significant.
“These findings support the notion that pancreatitis should be included in the list of GI manifestations of COVID-19,” the investigators wrote.
When caring for patients with COVID-19, Dr. Inamdar and colleagues recommended that clinicians pay close attention to any history of abdominal pain, and consider testing serum lipase levels.
“Further large studies are needed to confirm our findings,” they concluded.
Gyanprakash Avinash Ketwaroo, MD, of Baylor College of Medicine in Houston, agreed that more work is needed; in the meantime, he suggested that evidence is now strong enough for clinicians to take notice.
“Overall, this study adds further weight to COVID-19 acute pancreatitis,” he said. “Larger studies, and convincing pathophysiologic data, will be needed to confirm COVID-19 as a cause of acute pancreatitis. However, there appears to be enough circumstantial evidence to consider a COVID-19 diagnosis in patients presenting with acute pancreatitis.”
He noted that the new clinical evidence also stands on a solid theoretical foundation.
“Viruses, especially mumps and measles, have long been known to cause acute pancreatitis,” he said. “Additionally, the ACE2 receptor is present on pancreatic beta-cells and may mediate COVID-19 induced pancreatitis.”
Along with larger observational studies, Dr. Ketwaroo suggested that a number of interventional questions remain unanswered.
“While most acute pancreatitis is treated with supportive care, could proven therapies for COVID-19, such as steroids, also mitigate COVID-19 acute pancreatitis?” he asked. “Is COVID-19 a cofactor for acute pancreatitis caused by alcohol or endoscopic retrograde cholangiopancreatography? We await further information from an active area of research.”
The investigators disclosed relationships with Boston Scientific, Olympus, Fujifilm, and others.
SOURCE: Inamdar S et al. Gastroenterology. 2020 Aug 26. doi: 10.1053/j.gastro.2020.08.044.
Mounting data support acute pancreatitis as one possible GI manifestation of COVID-19, according to investigators.
While previous case reports suggested that infection with SARS-CoV2 may lead to pancreatitis, this retrospective analysis, which is the largest to date, is the first to offer substantial evidence for this claim, reported lead author Sumant Inamdar, MBBS, of the University of Arkansas, Little Rock, and colleagues.
“It has become increasingly clear that COVID-19 has systemic effects that also includes the gastrointestinal and pancreaticobiliary systems,” the investigators wrote in Gastroenterology. “As islet cells of the pancreas contain ACE2 receptor proteins, SARS-CoV2 can bind to these receptors and cause pancreatic injury.”
For the present analysis, Dr. Inamdar and colleagues reviewed charts from 48,012 patients who were hospitalized in New York between March and June of this year. While pancreatitis is usually diagnosed based on two out of three criteria, disease classification in the study required all three: characteristic upper abdominal pain upon admission, lipase greater than three times the upper limit of normal, and evidence of pancreatitis on cross-sectional imaging.
“[B]y including all three criteria for pancreatitis in our definition, we may be underestimating the rate of pancreatitis,” the investigators wrote. “However, we felt including diagnostic lipase levels and imaging was important for the accuracy of the diagnosis.”
Primary outcomes included mechanical ventilation, length of stay, development of pancreatic necrosis, and mortality. Outcomes were compared between patients with and without COVID-19.
Out of 48,012 hospitalized patients, 11,883 (24.75%) tested positive for SARS-CoV2. Across the entire population, 189 patients had pancreatitis (0.39%), and of these, 32 (17%) also had COVID-19. This translates to a point prevalence for pancreatitis of 0.27% for patients hospitalized with COVID-19.
Among patients with pancreatitis who did not have COVID-19, the most common etiologies for pancreatitis were gallstones (34%) and alcohol (37%), compared with just 16% and 6% of SARS-CoV2-positive cases of pancreatitis, respectively. Idiopathic pancreatitis was significantly more common among patients with COVID-19 than those without (69% vs 21%; P less than .0001).
Black or Hispanic patients with pancreatitis were 4-5 times more likely to have COVID-19 than patients with pancreatitis who were white. Across all races/ethnicities, patients with pancreatitis and COVID-19 more often required mechanical ventilation (odds ratio [OR], 5.65) and longer hospital stays (OR, 3.22), compared with those who had pancreatitis alone. While rates of mortality and pancreatic necrosis showed similar trends, associations with COVID-19 were not statistically significant.
“These findings support the notion that pancreatitis should be included in the list of GI manifestations of COVID-19,” the investigators wrote.
When caring for patients with COVID-19, Dr. Inamdar and colleagues recommended that clinicians pay close attention to any history of abdominal pain, and consider testing serum lipase levels.
“Further large studies are needed to confirm our findings,” they concluded.
Gyanprakash Avinash Ketwaroo, MD, of Baylor College of Medicine in Houston, agreed that more work is needed; in the meantime, he suggested that evidence is now strong enough for clinicians to take notice.
“Overall, this study adds further weight to COVID-19 acute pancreatitis,” he said. “Larger studies, and convincing pathophysiologic data, will be needed to confirm COVID-19 as a cause of acute pancreatitis. However, there appears to be enough circumstantial evidence to consider a COVID-19 diagnosis in patients presenting with acute pancreatitis.”
He noted that the new clinical evidence also stands on a solid theoretical foundation.
“Viruses, especially mumps and measles, have long been known to cause acute pancreatitis,” he said. “Additionally, the ACE2 receptor is present on pancreatic beta-cells and may mediate COVID-19 induced pancreatitis.”
Along with larger observational studies, Dr. Ketwaroo suggested that a number of interventional questions remain unanswered.
“While most acute pancreatitis is treated with supportive care, could proven therapies for COVID-19, such as steroids, also mitigate COVID-19 acute pancreatitis?” he asked. “Is COVID-19 a cofactor for acute pancreatitis caused by alcohol or endoscopic retrograde cholangiopancreatography? We await further information from an active area of research.”
The investigators disclosed relationships with Boston Scientific, Olympus, Fujifilm, and others.
SOURCE: Inamdar S et al. Gastroenterology. 2020 Aug 26. doi: 10.1053/j.gastro.2020.08.044.
FROM GASTROENTEROLOGY
Deaths sky high in hospitalized COVID patients with kidney injury
More evidence indicates that the development of acute kidney injury
“This ... is the first study in the United States to report the persistence of kidney dysfunction (lack of recovery) in survivors of COVID-19–associated AKI [and] this is in marked contrast to other forms of AKI where over 80% of patients recover their renal function by 10 days,” Lili Chan, MD, of the Icahn School of Medicine at Mount Sinai, New York, and colleagues observed.
The research is a retrospective, observational cohort study published online Sept. 3 in the Journal of the American Society of Nephrology
“We may be facing an epidemic of post–COVID-19 kidney disease and that, in turn, could mean much greater numbers of patients who require kidney dialysis and even transplants,” said senior author Girish Nadkarni, MD, a nephrologist, in a statement from Mount Sinai.
Nephrologists will need to prepare for a significant uptick in patients with chronic kidney disease as a result of exposure to the SARS-CoV-2 virus that causes COVID-19, the researchers warned.
“These findings may help centers with resource planning and preparing for the increased load resulting from survivors of COVID-19–associated AKI who do not experience recovery of kidney function,” they added.
Analysis of patients from February to end of May 2020
“AKI among hospitalized patients with COVID-19 in the United States is not well described,” they noted in their article.
And so they analyzed data from five major hospitals in the Mount Sinai Health System between Feb. 27 and May 30 of this year, during which 3,993 patients were hospitalized within the system for COVID-19. The MSHS has a patient population of racially and ethnically diverse citizens from New York.
AKI was defined using Kidney Disease: Improving Global Outcomes (KDIGO) criteria. AKI occurred in 46% of the overall cohort of patients, 19% of whom required dialysis.
However, among those patients who required admission to the ICU, over three-quarters (76%) developed AKI and almost one-third of ICU patients required dialysis, the investigators said.
“The median time from hospital admission until AKI diagnoses was 1 day and the median time from AKI diagnosis to dialysis was 3 days,” they explain.
The proportion of patients with stages 1, 2, or 3 AKI among those admitted to hospital were 39%, 19%, and 42%, respectively. In patients requiring admission to ICU, 28% had stage 1 AKI, 17% had stage 2, and 56% had stage 3.
And among those who required dialysis for AKI, the median peak serum creatinine was 8.2 mg/dL, compared with 2.2 mg/dL for those who did not require dialysis.
Predictors of AKI: male sex, potassium levels, and preexisting CKD
Almost two thirds of patients (65%) had recovered from their kidney injury by the time they left hospital but 35% had acute kidney disease. Of this latter group, on follow-up, 36% had recovered from it, the investigators noted.
Conversely, of those patients who had recovered from AKI by hospital discharge, 14% went on to develop acute kidney disease at the time of follow-up.
And 30% of patients who had required dialysis at some point during their hospital care required dialysis again within 72 hours of being discharged, the investigators noted.
Predictors of severe AKI included male sex (adjusted odds ratio, 1.46), potassium levels on admission (aOR, 1.7), and preexisting chronic kidney disease (CKD) (aOR, 2.8).
Most compellingly, “in-hospital mortality in patients who experienced AKI was 50% [versus] 8% in patients without AKI (P < .001),” Dr. Nadkarni and colleagues reported.
Among those who required ICU care, 42% of patients with AKI died, compared with 7% of those in ICU who did not develop AKI, while in patients cared for outside of ICU, 62% with AKI died compared with only 13% of those who did not develop AKI.
And after adjusting for demographics, comorbidities, and laboratory values, the aOR for death was 11.4 times higher for ICU patients with AKI, compared with ICU patients without AKI, the authors emphasize.
In all patients who developed AKI, the aOR for mortality was 9.2, compared with patients who did not develop AKI, they added.
Perhaps predictably, the risk of death rose with increasing stage of AKI, and patients with stage 3 AKI who required dialysis were at highest risk of death, the authors observe.
Sheer number of AKI cases, need for dialysis unprecedented
“The sheer number of AKI cases and the overwhelming need for dialysis that we are seeing in the context of COVID-19 is unprecedented,” Dr. Nadkarni said.
“These findings bring clinical evidence to the hypothesis of lingering organ dysfunction among patients recovering from COVID-19 and serve as a reminder to hospitals around the country to be very strategic in the allocation of resources to care for patients who experience AKI,” he cautioned.
“We are grappling with a great deal of uncertainty as to how the virus will impact the kidneys in the long haul,” Dr. Nadkarni added. “We may be facing an epidemic of post–COVID-19 kidney disease, and that, in turn, could mean much greater numbers of patients who require kidney dialysis and even transplants.”
Dr. Nadkarni reported serving as a consultant and advisory board member for RenalytixAI and owns equity in the company.
This article first appeared on Medscape.com.
More evidence indicates that the development of acute kidney injury
“This ... is the first study in the United States to report the persistence of kidney dysfunction (lack of recovery) in survivors of COVID-19–associated AKI [and] this is in marked contrast to other forms of AKI where over 80% of patients recover their renal function by 10 days,” Lili Chan, MD, of the Icahn School of Medicine at Mount Sinai, New York, and colleagues observed.
The research is a retrospective, observational cohort study published online Sept. 3 in the Journal of the American Society of Nephrology
“We may be facing an epidemic of post–COVID-19 kidney disease and that, in turn, could mean much greater numbers of patients who require kidney dialysis and even transplants,” said senior author Girish Nadkarni, MD, a nephrologist, in a statement from Mount Sinai.
Nephrologists will need to prepare for a significant uptick in patients with chronic kidney disease as a result of exposure to the SARS-CoV-2 virus that causes COVID-19, the researchers warned.
“These findings may help centers with resource planning and preparing for the increased load resulting from survivors of COVID-19–associated AKI who do not experience recovery of kidney function,” they added.
Analysis of patients from February to end of May 2020
“AKI among hospitalized patients with COVID-19 in the United States is not well described,” they noted in their article.
And so they analyzed data from five major hospitals in the Mount Sinai Health System between Feb. 27 and May 30 of this year, during which 3,993 patients were hospitalized within the system for COVID-19. The MSHS has a patient population of racially and ethnically diverse citizens from New York.
AKI was defined using Kidney Disease: Improving Global Outcomes (KDIGO) criteria. AKI occurred in 46% of the overall cohort of patients, 19% of whom required dialysis.
However, among those patients who required admission to the ICU, over three-quarters (76%) developed AKI and almost one-third of ICU patients required dialysis, the investigators said.
“The median time from hospital admission until AKI diagnoses was 1 day and the median time from AKI diagnosis to dialysis was 3 days,” they explain.
The proportion of patients with stages 1, 2, or 3 AKI among those admitted to hospital were 39%, 19%, and 42%, respectively. In patients requiring admission to ICU, 28% had stage 1 AKI, 17% had stage 2, and 56% had stage 3.
And among those who required dialysis for AKI, the median peak serum creatinine was 8.2 mg/dL, compared with 2.2 mg/dL for those who did not require dialysis.
Predictors of AKI: male sex, potassium levels, and preexisting CKD
Almost two thirds of patients (65%) had recovered from their kidney injury by the time they left hospital but 35% had acute kidney disease. Of this latter group, on follow-up, 36% had recovered from it, the investigators noted.
Conversely, of those patients who had recovered from AKI by hospital discharge, 14% went on to develop acute kidney disease at the time of follow-up.
And 30% of patients who had required dialysis at some point during their hospital care required dialysis again within 72 hours of being discharged, the investigators noted.
Predictors of severe AKI included male sex (adjusted odds ratio, 1.46), potassium levels on admission (aOR, 1.7), and preexisting chronic kidney disease (CKD) (aOR, 2.8).
Most compellingly, “in-hospital mortality in patients who experienced AKI was 50% [versus] 8% in patients without AKI (P < .001),” Dr. Nadkarni and colleagues reported.
Among those who required ICU care, 42% of patients with AKI died, compared with 7% of those in ICU who did not develop AKI, while in patients cared for outside of ICU, 62% with AKI died compared with only 13% of those who did not develop AKI.
And after adjusting for demographics, comorbidities, and laboratory values, the aOR for death was 11.4 times higher for ICU patients with AKI, compared with ICU patients without AKI, the authors emphasize.
In all patients who developed AKI, the aOR for mortality was 9.2, compared with patients who did not develop AKI, they added.
Perhaps predictably, the risk of death rose with increasing stage of AKI, and patients with stage 3 AKI who required dialysis were at highest risk of death, the authors observe.
Sheer number of AKI cases, need for dialysis unprecedented
“The sheer number of AKI cases and the overwhelming need for dialysis that we are seeing in the context of COVID-19 is unprecedented,” Dr. Nadkarni said.
“These findings bring clinical evidence to the hypothesis of lingering organ dysfunction among patients recovering from COVID-19 and serve as a reminder to hospitals around the country to be very strategic in the allocation of resources to care for patients who experience AKI,” he cautioned.
“We are grappling with a great deal of uncertainty as to how the virus will impact the kidneys in the long haul,” Dr. Nadkarni added. “We may be facing an epidemic of post–COVID-19 kidney disease, and that, in turn, could mean much greater numbers of patients who require kidney dialysis and even transplants.”
Dr. Nadkarni reported serving as a consultant and advisory board member for RenalytixAI and owns equity in the company.
This article first appeared on Medscape.com.
More evidence indicates that the development of acute kidney injury
“This ... is the first study in the United States to report the persistence of kidney dysfunction (lack of recovery) in survivors of COVID-19–associated AKI [and] this is in marked contrast to other forms of AKI where over 80% of patients recover their renal function by 10 days,” Lili Chan, MD, of the Icahn School of Medicine at Mount Sinai, New York, and colleagues observed.
The research is a retrospective, observational cohort study published online Sept. 3 in the Journal of the American Society of Nephrology
“We may be facing an epidemic of post–COVID-19 kidney disease and that, in turn, could mean much greater numbers of patients who require kidney dialysis and even transplants,” said senior author Girish Nadkarni, MD, a nephrologist, in a statement from Mount Sinai.
Nephrologists will need to prepare for a significant uptick in patients with chronic kidney disease as a result of exposure to the SARS-CoV-2 virus that causes COVID-19, the researchers warned.
“These findings may help centers with resource planning and preparing for the increased load resulting from survivors of COVID-19–associated AKI who do not experience recovery of kidney function,” they added.
Analysis of patients from February to end of May 2020
“AKI among hospitalized patients with COVID-19 in the United States is not well described,” they noted in their article.
And so they analyzed data from five major hospitals in the Mount Sinai Health System between Feb. 27 and May 30 of this year, during which 3,993 patients were hospitalized within the system for COVID-19. The MSHS has a patient population of racially and ethnically diverse citizens from New York.
AKI was defined using Kidney Disease: Improving Global Outcomes (KDIGO) criteria. AKI occurred in 46% of the overall cohort of patients, 19% of whom required dialysis.
However, among those patients who required admission to the ICU, over three-quarters (76%) developed AKI and almost one-third of ICU patients required dialysis, the investigators said.
“The median time from hospital admission until AKI diagnoses was 1 day and the median time from AKI diagnosis to dialysis was 3 days,” they explain.
The proportion of patients with stages 1, 2, or 3 AKI among those admitted to hospital were 39%, 19%, and 42%, respectively. In patients requiring admission to ICU, 28% had stage 1 AKI, 17% had stage 2, and 56% had stage 3.
And among those who required dialysis for AKI, the median peak serum creatinine was 8.2 mg/dL, compared with 2.2 mg/dL for those who did not require dialysis.
Predictors of AKI: male sex, potassium levels, and preexisting CKD
Almost two thirds of patients (65%) had recovered from their kidney injury by the time they left hospital but 35% had acute kidney disease. Of this latter group, on follow-up, 36% had recovered from it, the investigators noted.
Conversely, of those patients who had recovered from AKI by hospital discharge, 14% went on to develop acute kidney disease at the time of follow-up.
And 30% of patients who had required dialysis at some point during their hospital care required dialysis again within 72 hours of being discharged, the investigators noted.
Predictors of severe AKI included male sex (adjusted odds ratio, 1.46), potassium levels on admission (aOR, 1.7), and preexisting chronic kidney disease (CKD) (aOR, 2.8).
Most compellingly, “in-hospital mortality in patients who experienced AKI was 50% [versus] 8% in patients without AKI (P < .001),” Dr. Nadkarni and colleagues reported.
Among those who required ICU care, 42% of patients with AKI died, compared with 7% of those in ICU who did not develop AKI, while in patients cared for outside of ICU, 62% with AKI died compared with only 13% of those who did not develop AKI.
And after adjusting for demographics, comorbidities, and laboratory values, the aOR for death was 11.4 times higher for ICU patients with AKI, compared with ICU patients without AKI, the authors emphasize.
In all patients who developed AKI, the aOR for mortality was 9.2, compared with patients who did not develop AKI, they added.
Perhaps predictably, the risk of death rose with increasing stage of AKI, and patients with stage 3 AKI who required dialysis were at highest risk of death, the authors observe.
Sheer number of AKI cases, need for dialysis unprecedented
“The sheer number of AKI cases and the overwhelming need for dialysis that we are seeing in the context of COVID-19 is unprecedented,” Dr. Nadkarni said.
“These findings bring clinical evidence to the hypothesis of lingering organ dysfunction among patients recovering from COVID-19 and serve as a reminder to hospitals around the country to be very strategic in the allocation of resources to care for patients who experience AKI,” he cautioned.
“We are grappling with a great deal of uncertainty as to how the virus will impact the kidneys in the long haul,” Dr. Nadkarni added. “We may be facing an epidemic of post–COVID-19 kidney disease, and that, in turn, could mean much greater numbers of patients who require kidney dialysis and even transplants.”
Dr. Nadkarni reported serving as a consultant and advisory board member for RenalytixAI and owns equity in the company.
This article first appeared on Medscape.com.