User login
Zoom Zoom Zoom: An end-of-year update from a virtual psychiatrist
In mid-April, a month into pandemic life with a stay-at-home order, I wrote about my experiences as a virtual outpatient psychiatrist in private practice. It’s been 10 months now and with this tragic year drawing to a close, it seems like a good time for an update.
In that April column, I describe how I created a makeshift home office. This entailed pushing my son’s baseball card collection and dusty sports trophies to the side of the room, bringing in a desk and a rug, a house plant, and a statue of a Buddha. I enjoyed watching out the window behind my computer screen as the neighbors and their dogs walked by, and I loved seeing the tree out the window blossom into gorgeous flowers.
With time, my physical space has changed. The remnants of my son’s childhood have all been moved to a closet, artwork has been added to the wall behind me, and the space is now clearly an office, though my laptop remains propped on a pile of books so that no one is looking up my nose. The room, with four large windows facing north and west, has issues with temperature control. In an old house, the heat works all too well in the adjacent bedroom (while the rest of the occupants in other rooms freeze), but the office itself has no heat: I have added both a fan and a space heater, and there are some very cold days where I’ve propped open one of the windows. And with the shortened days, large windows on two walls have presented a challenge as the sun changes positions throughout the day – there are times when the sun’s rays streak across my face in such a way that I look rather ethereal, and between sessions I have lowered, raised, and adjusted the blinds to avoid this. I finally pulled off the thin metal venetian blinds and took them to Lowe’s, where a partially masked young woman cut me new blinds with larger slats. An ergonomic office chair has replaced the wicker Ikea chair I was using, and between all these machinations, I am now physically comfortable most of the time. I believe I am still a bit too pixelated on the screen, but my patients are not complaining, and when the natural lighting fades at 4:30 p.m., the overhead lighting is all wrong again. These all are things I never considered – or long ago addressed – in my real-life practice of psychiatry in a office I have loved for years.
With time, I’ve grown more comfortable working from home on a screen and there are things about this life I’ve grown to like. My husband no longer travels, my daughter – my gift of the pandemic – returned home from New York City where she was in her final months of graduate school, and these unexpected months with her (and her cat) have been a pleasure. There is something nice about being trapped at home with people I love, even if we are all in our respective places, in front of our separate screens. There has been time for long walks, trips to the beach, and long bike rides. And as my daughter now prepares to move to Denver, I have been heartened by the hope of vaccines, and the knowledge that I will likely be able to see her again in the coming months. The people are not the only ones who have benefited from this time at home together – I have no idea how we would have managed with our elderly dog if we were not home to care for him.
My life has become more efficient. I used to find myself aggravated when patients forgot their appointments, a not-infrequent occurrence. People no longer get caught in traffic, they come on time, and they don’t complain about my crowded parking lot. When there is down time, I use it more efficiently at home – a load of laundry gets done, I get a chance to turn on the news or exercise, or make dinner early. And because I have two other family members working from home, I am not the only one mixing work with chores or exercise.
While my medical colleagues who work in settings where they must see patients in person have struggled or functioned in some state of denial, I have felt safe and protected, a bit cocooned with my family in a house big enough to give us all space, in a neighborhood with sidewalks and places to walk, and to protect my sanity, I am lucky to have a patio that has now been equipped with lights, patio heaters, a fire pit, and socially distanced tables so that I can still see friends outside.
Telemedicine has added a new dimension to treatment. I’ve had family sessions with multiple people joining a zoom link from different locations – so much easier than coordinating a time when everyone can travel to my office. I’ve had patients call in from cars and from closets in search of privacy, and from their gardens and poolsides. I’ve met spouses, children, many a dog and cat, plus the more unusual of pets and farm animals, including a goat, ferret, lizard, African grey parrot, and guinea pigs.
These are the good things, and while I wish I could say it was all good, so much of what remains is laden with anxiety. My son lives nearby, but he has shared a house with a hospital worker for much of the past year and there were COVID scares, months at a time without so much as a hug, and my husband has not seen his parents or brother for a year now. There are the awkward waves or salutes with friends I once gave carefree hugs, the constant thoughts of how far away is that person standing, and each person’s “beliefs” about what is safe when we still don’t fully understand how this virus spreads. I worry for myself, I worry for my family and friends, and I worry for my patients when they tell me about behaviors that clearly are not safe.
At first, I found my work as a telepsychiatrist to be exhausting, and I assumed it was because my patients were now just faces, inches from my own eyes, and no longer diffused by a visual field that included my whole office and the opportunity to break eye contact while I still listened with full attention. This has gotten much better – I’ve adjusted to my on-screen relationships, but what has not gotten better is both the acuity, and sometimes the boredom.
Patients are struggling; they are sad, lonely, and missing the richness of their former lives. They miss friends, meeting new people, cultural experiences, diversity in how they spend their time, and travel. They have all the same human experiences of loss, illness, and grief, but with the added burden of struggling alone or within the confines of pandemic life that has destroyed our ability to mark events with social and religious customs that guide healing. People who had done well for years are now needing more, and those who were not doing well are doing worse. It makes for long days.
I mentioned boredom: With less time spent with other people, so many sessions are about COVID – who has it, who might have it, what people are doing to avoid it, and still, how they get their groceries. The second most popular psychotherapy topic includes what they are watching on Netflix, and as human beings trudging through this together, I have appreciated my patients’ suggestions as much as they have appreciated mine.* Life for all of us has come to be more about survival, and less about self-discovery and striving. Many sessions have started to feel the same from 1 hour to the next, in ways they never did before.
There are other aspects to telepsychiatry that I have found difficult. The site I have used most – Doxy.me – works well with some patients, but with others there are technical problems. Sessions freeze, the sound goes in or out, and we end up switching to another platform, which may or may not work better. Sometimes patients have the camera at odd angles, or they bounce a laptop on their knees to the point that I get seasick. One of my family members has said that I can sometimes be overheard, so I now have a radio playing classical music outside my door, and I often use earbuds so that the patient can’t be overheard and I speak more softly with them – this has all been good in terms of improving privacy, but after a while I find that it’s stressful to have people talking to me inside my own ears! These are little kinks, but when you do it for hours a day, they add up to a sense of being stressed in ways that in-person psychiatry does not lend itself to.
Finally, three seasons into my work-at-home life, I still have not found a new rhythm for some of the logistical aspects of private practice that came so easily in my office. My mail still goes to the office, the plants there still need water, my files and computer are there, but tasks that were once a seamless part of my work day now spill into my time off and I go into the office each week to file, log medications, and attend to the business of my practice. My smartphone, with its ability to e-prescribe, invoice, and fax, has made it possible for me to manage and certainly, outpatient psychiatrists are very lucky that we have the option to continue our work with patients remotely during such difficult times.
I have sent people for virtual intensive substance treatment, and to virtual couples’ counseling, and these remote treatments have been useful. The one treatment that has been very difficult for patients to negotiate has been outpatient electroconvulsive therapy – this requires coordination with another person to drive the patient to treatments (and to wait outside in the parking lot), and also for separate weekly COVID testing. Transcranial magnetic stimulation, which also is still being done in person, has not been any different – patients can drive themselves and the one center I referred to has not required preprocedure COVID testing.
What does the future hold? Will we ever go back to practicing the way we did? While some of my patients miss real-life therapy, most do not; they too like the added efficiency, getting treatment from the comfort of their home without the stress of finding the time to travel. I’ve taken on new patients during this time, and while I anticipated that it would be difficult, it has gone surprisingly well – people I have never met in real life talk to me with ease, and both psychotherapy and medication management have gone well. The one area that I have found most difficult is assessing tremors and dyskinesias, and one patient mentioned she has gained nearly 50 pounds over the past year – something I certainly would have noticed and attended to sooner in real life. I have mixed feelings about returning to a completely live practice. I think I would like a combination where I see all my patients in person once in a while, but would like to be able to offer some times where I see people virtually from home at least one day a week.
Time will tell how that plays out with insurers. My best guess is that, with the lowered no-show rates that everyone is seeing and the higher levels of depression and anxiety that people are having, this may have been a costly time for mental health care. At the same time, inpatient psychiatric units have decreased their capacity, and perhaps more efficient delivery of outpatient care has lowered the overall cost. I suppose we will wait to hear, but for many, the transition to virtual care has allowed many people to get treatment who would have otherwise gone without care.
In my April article, I mentioned that I was having daily Facetime check-in visits with a distressed patient who was on a COVID unit with pneumonia. Since then, I have had several more patients contract COVID, and many of my patients have had family members who have tested positive or become symptomatic with COVID. It has been nice to have sessions with people during this time, and thankfully, I have not had any more patients who have required hospitalization for the virus.
I still catch myself thinking that, of all the things I have worried about over the years, “pandemic” was never on my list. It seems so strange that I left my office on a Friday with no idea that I would not be returning to work the following Monday, or that life would change in such a radical way. As we leave this awful year behind and greet the new one with the hope that vaccines and a new administration might offer solutions, I’d like to wish my readers the best for a healthy, safe, and gentle New Year.
*My top viewing picks for now are “The Queen’s Gambit” (Netflix), and “A Place to Call Home” (Acorn).
Dr. Miller is coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins, both in Baltimore.
In mid-April, a month into pandemic life with a stay-at-home order, I wrote about my experiences as a virtual outpatient psychiatrist in private practice. It’s been 10 months now and with this tragic year drawing to a close, it seems like a good time for an update.
In that April column, I describe how I created a makeshift home office. This entailed pushing my son’s baseball card collection and dusty sports trophies to the side of the room, bringing in a desk and a rug, a house plant, and a statue of a Buddha. I enjoyed watching out the window behind my computer screen as the neighbors and their dogs walked by, and I loved seeing the tree out the window blossom into gorgeous flowers.
With time, my physical space has changed. The remnants of my son’s childhood have all been moved to a closet, artwork has been added to the wall behind me, and the space is now clearly an office, though my laptop remains propped on a pile of books so that no one is looking up my nose. The room, with four large windows facing north and west, has issues with temperature control. In an old house, the heat works all too well in the adjacent bedroom (while the rest of the occupants in other rooms freeze), but the office itself has no heat: I have added both a fan and a space heater, and there are some very cold days where I’ve propped open one of the windows. And with the shortened days, large windows on two walls have presented a challenge as the sun changes positions throughout the day – there are times when the sun’s rays streak across my face in such a way that I look rather ethereal, and between sessions I have lowered, raised, and adjusted the blinds to avoid this. I finally pulled off the thin metal venetian blinds and took them to Lowe’s, where a partially masked young woman cut me new blinds with larger slats. An ergonomic office chair has replaced the wicker Ikea chair I was using, and between all these machinations, I am now physically comfortable most of the time. I believe I am still a bit too pixelated on the screen, but my patients are not complaining, and when the natural lighting fades at 4:30 p.m., the overhead lighting is all wrong again. These all are things I never considered – or long ago addressed – in my real-life practice of psychiatry in a office I have loved for years.
With time, I’ve grown more comfortable working from home on a screen and there are things about this life I’ve grown to like. My husband no longer travels, my daughter – my gift of the pandemic – returned home from New York City where she was in her final months of graduate school, and these unexpected months with her (and her cat) have been a pleasure. There is something nice about being trapped at home with people I love, even if we are all in our respective places, in front of our separate screens. There has been time for long walks, trips to the beach, and long bike rides. And as my daughter now prepares to move to Denver, I have been heartened by the hope of vaccines, and the knowledge that I will likely be able to see her again in the coming months. The people are not the only ones who have benefited from this time at home together – I have no idea how we would have managed with our elderly dog if we were not home to care for him.
My life has become more efficient. I used to find myself aggravated when patients forgot their appointments, a not-infrequent occurrence. People no longer get caught in traffic, they come on time, and they don’t complain about my crowded parking lot. When there is down time, I use it more efficiently at home – a load of laundry gets done, I get a chance to turn on the news or exercise, or make dinner early. And because I have two other family members working from home, I am not the only one mixing work with chores or exercise.
While my medical colleagues who work in settings where they must see patients in person have struggled or functioned in some state of denial, I have felt safe and protected, a bit cocooned with my family in a house big enough to give us all space, in a neighborhood with sidewalks and places to walk, and to protect my sanity, I am lucky to have a patio that has now been equipped with lights, patio heaters, a fire pit, and socially distanced tables so that I can still see friends outside.
Telemedicine has added a new dimension to treatment. I’ve had family sessions with multiple people joining a zoom link from different locations – so much easier than coordinating a time when everyone can travel to my office. I’ve had patients call in from cars and from closets in search of privacy, and from their gardens and poolsides. I’ve met spouses, children, many a dog and cat, plus the more unusual of pets and farm animals, including a goat, ferret, lizard, African grey parrot, and guinea pigs.
These are the good things, and while I wish I could say it was all good, so much of what remains is laden with anxiety. My son lives nearby, but he has shared a house with a hospital worker for much of the past year and there were COVID scares, months at a time without so much as a hug, and my husband has not seen his parents or brother for a year now. There are the awkward waves or salutes with friends I once gave carefree hugs, the constant thoughts of how far away is that person standing, and each person’s “beliefs” about what is safe when we still don’t fully understand how this virus spreads. I worry for myself, I worry for my family and friends, and I worry for my patients when they tell me about behaviors that clearly are not safe.
At first, I found my work as a telepsychiatrist to be exhausting, and I assumed it was because my patients were now just faces, inches from my own eyes, and no longer diffused by a visual field that included my whole office and the opportunity to break eye contact while I still listened with full attention. This has gotten much better – I’ve adjusted to my on-screen relationships, but what has not gotten better is both the acuity, and sometimes the boredom.
Patients are struggling; they are sad, lonely, and missing the richness of their former lives. They miss friends, meeting new people, cultural experiences, diversity in how they spend their time, and travel. They have all the same human experiences of loss, illness, and grief, but with the added burden of struggling alone or within the confines of pandemic life that has destroyed our ability to mark events with social and religious customs that guide healing. People who had done well for years are now needing more, and those who were not doing well are doing worse. It makes for long days.
I mentioned boredom: With less time spent with other people, so many sessions are about COVID – who has it, who might have it, what people are doing to avoid it, and still, how they get their groceries. The second most popular psychotherapy topic includes what they are watching on Netflix, and as human beings trudging through this together, I have appreciated my patients’ suggestions as much as they have appreciated mine.* Life for all of us has come to be more about survival, and less about self-discovery and striving. Many sessions have started to feel the same from 1 hour to the next, in ways they never did before.
There are other aspects to telepsychiatry that I have found difficult. The site I have used most – Doxy.me – works well with some patients, but with others there are technical problems. Sessions freeze, the sound goes in or out, and we end up switching to another platform, which may or may not work better. Sometimes patients have the camera at odd angles, or they bounce a laptop on their knees to the point that I get seasick. One of my family members has said that I can sometimes be overheard, so I now have a radio playing classical music outside my door, and I often use earbuds so that the patient can’t be overheard and I speak more softly with them – this has all been good in terms of improving privacy, but after a while I find that it’s stressful to have people talking to me inside my own ears! These are little kinks, but when you do it for hours a day, they add up to a sense of being stressed in ways that in-person psychiatry does not lend itself to.
Finally, three seasons into my work-at-home life, I still have not found a new rhythm for some of the logistical aspects of private practice that came so easily in my office. My mail still goes to the office, the plants there still need water, my files and computer are there, but tasks that were once a seamless part of my work day now spill into my time off and I go into the office each week to file, log medications, and attend to the business of my practice. My smartphone, with its ability to e-prescribe, invoice, and fax, has made it possible for me to manage and certainly, outpatient psychiatrists are very lucky that we have the option to continue our work with patients remotely during such difficult times.
I have sent people for virtual intensive substance treatment, and to virtual couples’ counseling, and these remote treatments have been useful. The one treatment that has been very difficult for patients to negotiate has been outpatient electroconvulsive therapy – this requires coordination with another person to drive the patient to treatments (and to wait outside in the parking lot), and also for separate weekly COVID testing. Transcranial magnetic stimulation, which also is still being done in person, has not been any different – patients can drive themselves and the one center I referred to has not required preprocedure COVID testing.
What does the future hold? Will we ever go back to practicing the way we did? While some of my patients miss real-life therapy, most do not; they too like the added efficiency, getting treatment from the comfort of their home without the stress of finding the time to travel. I’ve taken on new patients during this time, and while I anticipated that it would be difficult, it has gone surprisingly well – people I have never met in real life talk to me with ease, and both psychotherapy and medication management have gone well. The one area that I have found most difficult is assessing tremors and dyskinesias, and one patient mentioned she has gained nearly 50 pounds over the past year – something I certainly would have noticed and attended to sooner in real life. I have mixed feelings about returning to a completely live practice. I think I would like a combination where I see all my patients in person once in a while, but would like to be able to offer some times where I see people virtually from home at least one day a week.
Time will tell how that plays out with insurers. My best guess is that, with the lowered no-show rates that everyone is seeing and the higher levels of depression and anxiety that people are having, this may have been a costly time for mental health care. At the same time, inpatient psychiatric units have decreased their capacity, and perhaps more efficient delivery of outpatient care has lowered the overall cost. I suppose we will wait to hear, but for many, the transition to virtual care has allowed many people to get treatment who would have otherwise gone without care.
In my April article, I mentioned that I was having daily Facetime check-in visits with a distressed patient who was on a COVID unit with pneumonia. Since then, I have had several more patients contract COVID, and many of my patients have had family members who have tested positive or become symptomatic with COVID. It has been nice to have sessions with people during this time, and thankfully, I have not had any more patients who have required hospitalization for the virus.
I still catch myself thinking that, of all the things I have worried about over the years, “pandemic” was never on my list. It seems so strange that I left my office on a Friday with no idea that I would not be returning to work the following Monday, or that life would change in such a radical way. As we leave this awful year behind and greet the new one with the hope that vaccines and a new administration might offer solutions, I’d like to wish my readers the best for a healthy, safe, and gentle New Year.
*My top viewing picks for now are “The Queen’s Gambit” (Netflix), and “A Place to Call Home” (Acorn).
Dr. Miller is coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins, both in Baltimore.
In mid-April, a month into pandemic life with a stay-at-home order, I wrote about my experiences as a virtual outpatient psychiatrist in private practice. It’s been 10 months now and with this tragic year drawing to a close, it seems like a good time for an update.
In that April column, I describe how I created a makeshift home office. This entailed pushing my son’s baseball card collection and dusty sports trophies to the side of the room, bringing in a desk and a rug, a house plant, and a statue of a Buddha. I enjoyed watching out the window behind my computer screen as the neighbors and their dogs walked by, and I loved seeing the tree out the window blossom into gorgeous flowers.
With time, my physical space has changed. The remnants of my son’s childhood have all been moved to a closet, artwork has been added to the wall behind me, and the space is now clearly an office, though my laptop remains propped on a pile of books so that no one is looking up my nose. The room, with four large windows facing north and west, has issues with temperature control. In an old house, the heat works all too well in the adjacent bedroom (while the rest of the occupants in other rooms freeze), but the office itself has no heat: I have added both a fan and a space heater, and there are some very cold days where I’ve propped open one of the windows. And with the shortened days, large windows on two walls have presented a challenge as the sun changes positions throughout the day – there are times when the sun’s rays streak across my face in such a way that I look rather ethereal, and between sessions I have lowered, raised, and adjusted the blinds to avoid this. I finally pulled off the thin metal venetian blinds and took them to Lowe’s, where a partially masked young woman cut me new blinds with larger slats. An ergonomic office chair has replaced the wicker Ikea chair I was using, and between all these machinations, I am now physically comfortable most of the time. I believe I am still a bit too pixelated on the screen, but my patients are not complaining, and when the natural lighting fades at 4:30 p.m., the overhead lighting is all wrong again. These all are things I never considered – or long ago addressed – in my real-life practice of psychiatry in a office I have loved for years.
With time, I’ve grown more comfortable working from home on a screen and there are things about this life I’ve grown to like. My husband no longer travels, my daughter – my gift of the pandemic – returned home from New York City where she was in her final months of graduate school, and these unexpected months with her (and her cat) have been a pleasure. There is something nice about being trapped at home with people I love, even if we are all in our respective places, in front of our separate screens. There has been time for long walks, trips to the beach, and long bike rides. And as my daughter now prepares to move to Denver, I have been heartened by the hope of vaccines, and the knowledge that I will likely be able to see her again in the coming months. The people are not the only ones who have benefited from this time at home together – I have no idea how we would have managed with our elderly dog if we were not home to care for him.
My life has become more efficient. I used to find myself aggravated when patients forgot their appointments, a not-infrequent occurrence. People no longer get caught in traffic, they come on time, and they don’t complain about my crowded parking lot. When there is down time, I use it more efficiently at home – a load of laundry gets done, I get a chance to turn on the news or exercise, or make dinner early. And because I have two other family members working from home, I am not the only one mixing work with chores or exercise.
While my medical colleagues who work in settings where they must see patients in person have struggled or functioned in some state of denial, I have felt safe and protected, a bit cocooned with my family in a house big enough to give us all space, in a neighborhood with sidewalks and places to walk, and to protect my sanity, I am lucky to have a patio that has now been equipped with lights, patio heaters, a fire pit, and socially distanced tables so that I can still see friends outside.
Telemedicine has added a new dimension to treatment. I’ve had family sessions with multiple people joining a zoom link from different locations – so much easier than coordinating a time when everyone can travel to my office. I’ve had patients call in from cars and from closets in search of privacy, and from their gardens and poolsides. I’ve met spouses, children, many a dog and cat, plus the more unusual of pets and farm animals, including a goat, ferret, lizard, African grey parrot, and guinea pigs.
These are the good things, and while I wish I could say it was all good, so much of what remains is laden with anxiety. My son lives nearby, but he has shared a house with a hospital worker for much of the past year and there were COVID scares, months at a time without so much as a hug, and my husband has not seen his parents or brother for a year now. There are the awkward waves or salutes with friends I once gave carefree hugs, the constant thoughts of how far away is that person standing, and each person’s “beliefs” about what is safe when we still don’t fully understand how this virus spreads. I worry for myself, I worry for my family and friends, and I worry for my patients when they tell me about behaviors that clearly are not safe.
At first, I found my work as a telepsychiatrist to be exhausting, and I assumed it was because my patients were now just faces, inches from my own eyes, and no longer diffused by a visual field that included my whole office and the opportunity to break eye contact while I still listened with full attention. This has gotten much better – I’ve adjusted to my on-screen relationships, but what has not gotten better is both the acuity, and sometimes the boredom.
Patients are struggling; they are sad, lonely, and missing the richness of their former lives. They miss friends, meeting new people, cultural experiences, diversity in how they spend their time, and travel. They have all the same human experiences of loss, illness, and grief, but with the added burden of struggling alone or within the confines of pandemic life that has destroyed our ability to mark events with social and religious customs that guide healing. People who had done well for years are now needing more, and those who were not doing well are doing worse. It makes for long days.
I mentioned boredom: With less time spent with other people, so many sessions are about COVID – who has it, who might have it, what people are doing to avoid it, and still, how they get their groceries. The second most popular psychotherapy topic includes what they are watching on Netflix, and as human beings trudging through this together, I have appreciated my patients’ suggestions as much as they have appreciated mine.* Life for all of us has come to be more about survival, and less about self-discovery and striving. Many sessions have started to feel the same from 1 hour to the next, in ways they never did before.
There are other aspects to telepsychiatry that I have found difficult. The site I have used most – Doxy.me – works well with some patients, but with others there are technical problems. Sessions freeze, the sound goes in or out, and we end up switching to another platform, which may or may not work better. Sometimes patients have the camera at odd angles, or they bounce a laptop on their knees to the point that I get seasick. One of my family members has said that I can sometimes be overheard, so I now have a radio playing classical music outside my door, and I often use earbuds so that the patient can’t be overheard and I speak more softly with them – this has all been good in terms of improving privacy, but after a while I find that it’s stressful to have people talking to me inside my own ears! These are little kinks, but when you do it for hours a day, they add up to a sense of being stressed in ways that in-person psychiatry does not lend itself to.
Finally, three seasons into my work-at-home life, I still have not found a new rhythm for some of the logistical aspects of private practice that came so easily in my office. My mail still goes to the office, the plants there still need water, my files and computer are there, but tasks that were once a seamless part of my work day now spill into my time off and I go into the office each week to file, log medications, and attend to the business of my practice. My smartphone, with its ability to e-prescribe, invoice, and fax, has made it possible for me to manage and certainly, outpatient psychiatrists are very lucky that we have the option to continue our work with patients remotely during such difficult times.
I have sent people for virtual intensive substance treatment, and to virtual couples’ counseling, and these remote treatments have been useful. The one treatment that has been very difficult for patients to negotiate has been outpatient electroconvulsive therapy – this requires coordination with another person to drive the patient to treatments (and to wait outside in the parking lot), and also for separate weekly COVID testing. Transcranial magnetic stimulation, which also is still being done in person, has not been any different – patients can drive themselves and the one center I referred to has not required preprocedure COVID testing.
What does the future hold? Will we ever go back to practicing the way we did? While some of my patients miss real-life therapy, most do not; they too like the added efficiency, getting treatment from the comfort of their home without the stress of finding the time to travel. I’ve taken on new patients during this time, and while I anticipated that it would be difficult, it has gone surprisingly well – people I have never met in real life talk to me with ease, and both psychotherapy and medication management have gone well. The one area that I have found most difficult is assessing tremors and dyskinesias, and one patient mentioned she has gained nearly 50 pounds over the past year – something I certainly would have noticed and attended to sooner in real life. I have mixed feelings about returning to a completely live practice. I think I would like a combination where I see all my patients in person once in a while, but would like to be able to offer some times where I see people virtually from home at least one day a week.
Time will tell how that plays out with insurers. My best guess is that, with the lowered no-show rates that everyone is seeing and the higher levels of depression and anxiety that people are having, this may have been a costly time for mental health care. At the same time, inpatient psychiatric units have decreased their capacity, and perhaps more efficient delivery of outpatient care has lowered the overall cost. I suppose we will wait to hear, but for many, the transition to virtual care has allowed many people to get treatment who would have otherwise gone without care.
In my April article, I mentioned that I was having daily Facetime check-in visits with a distressed patient who was on a COVID unit with pneumonia. Since then, I have had several more patients contract COVID, and many of my patients have had family members who have tested positive or become symptomatic with COVID. It has been nice to have sessions with people during this time, and thankfully, I have not had any more patients who have required hospitalization for the virus.
I still catch myself thinking that, of all the things I have worried about over the years, “pandemic” was never on my list. It seems so strange that I left my office on a Friday with no idea that I would not be returning to work the following Monday, or that life would change in such a radical way. As we leave this awful year behind and greet the new one with the hope that vaccines and a new administration might offer solutions, I’d like to wish my readers the best for a healthy, safe, and gentle New Year.
*My top viewing picks for now are “The Queen’s Gambit” (Netflix), and “A Place to Call Home” (Acorn).
Dr. Miller is coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins, both in Baltimore.
COVID-19 vaccine rollout faces delays
If the current pace of vaccination continues, “it’s going to take years, not months, to vaccinate the American people,” President-elect Joe Biden said during a briefing Dec. 29.
In fact, at the current rate, it would take nearly 10 years to vaccinate enough Americans to bring the pandemic under control, according to NBC News. To reach 80% of the country by late June, 3 million people would need to receive a COVID-19 vaccine each day.
“As I long feared and warned, the effort to distribute and administer the vaccine is not progressing as it should,” Mr. Biden said, reemphasizing his pledge to get 100 million doses to Americans during his first 100 days as president.
So far, 11.4 million doses have been distributed and 2.1 million people have received a vaccine, according to the Centers for Disease Control and Prevention. Most states have administered a fraction of the doses they’ve received, according to data compiled by The New York Times.
Federal officials have said there’s an “expected lag” between delivery of doses, shots going into arms, and the data being reported to the CDC, according to CNN. The Food and Drug Administration must assess each shipment for quality control, which has slowed down distribution, and the CDC data are just now beginning to include the Moderna vaccine, which the FDA authorized for emergency use on Dec. 18.
The 2.1 million number is “an underestimate,” Brett Giroir, MD, the assistant secretary of the U.S. Department of Health & Human Services, told NBC News Dec. 29. At the same time, the U.S. won’t meet the goal of vaccinating 20 million people in the next few days, he said.
Another 30 million doses will go out in January, Dr. Giroir said, followed by 50 million in February.
Some vaccine experts have said they’re not surprised by the speed of vaccine distribution.
“It had to go this way,” Paul Offit, MD, a professor of pediatrics at Children’s Hospital of Philadelphia, told STAT. “We had to trip and fall and stumble and figure this out.”
To speed up distribution in 2021, the federal government will need to help states, Mr. Biden said Dec. 29. He plans to use the Defense Authorization Act to ramp up production of vaccine supplies. Even still, the process will take months, he said.
A version of this article first appeared on WebMD.com .
If the current pace of vaccination continues, “it’s going to take years, not months, to vaccinate the American people,” President-elect Joe Biden said during a briefing Dec. 29.
In fact, at the current rate, it would take nearly 10 years to vaccinate enough Americans to bring the pandemic under control, according to NBC News. To reach 80% of the country by late June, 3 million people would need to receive a COVID-19 vaccine each day.
“As I long feared and warned, the effort to distribute and administer the vaccine is not progressing as it should,” Mr. Biden said, reemphasizing his pledge to get 100 million doses to Americans during his first 100 days as president.
So far, 11.4 million doses have been distributed and 2.1 million people have received a vaccine, according to the Centers for Disease Control and Prevention. Most states have administered a fraction of the doses they’ve received, according to data compiled by The New York Times.
Federal officials have said there’s an “expected lag” between delivery of doses, shots going into arms, and the data being reported to the CDC, according to CNN. The Food and Drug Administration must assess each shipment for quality control, which has slowed down distribution, and the CDC data are just now beginning to include the Moderna vaccine, which the FDA authorized for emergency use on Dec. 18.
The 2.1 million number is “an underestimate,” Brett Giroir, MD, the assistant secretary of the U.S. Department of Health & Human Services, told NBC News Dec. 29. At the same time, the U.S. won’t meet the goal of vaccinating 20 million people in the next few days, he said.
Another 30 million doses will go out in January, Dr. Giroir said, followed by 50 million in February.
Some vaccine experts have said they’re not surprised by the speed of vaccine distribution.
“It had to go this way,” Paul Offit, MD, a professor of pediatrics at Children’s Hospital of Philadelphia, told STAT. “We had to trip and fall and stumble and figure this out.”
To speed up distribution in 2021, the federal government will need to help states, Mr. Biden said Dec. 29. He plans to use the Defense Authorization Act to ramp up production of vaccine supplies. Even still, the process will take months, he said.
A version of this article first appeared on WebMD.com .
If the current pace of vaccination continues, “it’s going to take years, not months, to vaccinate the American people,” President-elect Joe Biden said during a briefing Dec. 29.
In fact, at the current rate, it would take nearly 10 years to vaccinate enough Americans to bring the pandemic under control, according to NBC News. To reach 80% of the country by late June, 3 million people would need to receive a COVID-19 vaccine each day.
“As I long feared and warned, the effort to distribute and administer the vaccine is not progressing as it should,” Mr. Biden said, reemphasizing his pledge to get 100 million doses to Americans during his first 100 days as president.
So far, 11.4 million doses have been distributed and 2.1 million people have received a vaccine, according to the Centers for Disease Control and Prevention. Most states have administered a fraction of the doses they’ve received, according to data compiled by The New York Times.
Federal officials have said there’s an “expected lag” between delivery of doses, shots going into arms, and the data being reported to the CDC, according to CNN. The Food and Drug Administration must assess each shipment for quality control, which has slowed down distribution, and the CDC data are just now beginning to include the Moderna vaccine, which the FDA authorized for emergency use on Dec. 18.
The 2.1 million number is “an underestimate,” Brett Giroir, MD, the assistant secretary of the U.S. Department of Health & Human Services, told NBC News Dec. 29. At the same time, the U.S. won’t meet the goal of vaccinating 20 million people in the next few days, he said.
Another 30 million doses will go out in January, Dr. Giroir said, followed by 50 million in February.
Some vaccine experts have said they’re not surprised by the speed of vaccine distribution.
“It had to go this way,” Paul Offit, MD, a professor of pediatrics at Children’s Hospital of Philadelphia, told STAT. “We had to trip and fall and stumble and figure this out.”
To speed up distribution in 2021, the federal government will need to help states, Mr. Biden said Dec. 29. He plans to use the Defense Authorization Act to ramp up production of vaccine supplies. Even still, the process will take months, he said.
A version of this article first appeared on WebMD.com .
Anorexia and diarrhea top list of GI symptoms in COVID-19 patients
Patients with severe COVID-19 were significantly more likely than those with milder cases to have GI symptoms of anorexia and diarrhea, as well as abnormal liver function, based on data from a meta-analysis of more than 4,500 patients.
Previous studies have shown that liver damage “was more likely to be observed in severe patients during the process of disease,” and other studies have shown varying degrees of liver insufficiency in COVID-19 patients, but gastrointestinal symptoms have not been well studied, wrote Zi-yuan Dong, MD, of China Medical University, Shenyang City, and colleagues.
In a study published in the Journal of Clinical Gastroenterology, the researchers identified 31 studies including 4,682 COVID-19 patients. Case collection was from Dec. 11, 2019, to Feb. 28, 2020. Median age among studies ranged from 36 to 62 years, and 55% of patients were male.
A total of 26 studies were analyzed for the prevalence of GI symptoms, specifically nausea, vomiting, diarrhea, abdominal pain, and anorexia. Of these, anorexia and diarrhea were significantly more common in COVID-19 patients, with prevalence of 17% and 8% respectively, (P < .0001 for both).
In addition, 14 of the studies included in the analysis assessed the prevalence of abnormal liver function based on increased levels of aspartate aminotransferase, alanine aminotransferase, and total bilirubin. Of these, increased alanine aminotransferase was the most common, occurring in 25% of patients, compared with increased AST (in 24%) and total bilirubin (in 13%).
When assessed by disease severity, patients with severe disease and those in the ICU were significantly more likely than general/non-ICU patients to have anorexia (odds ratio, 2.19), diarrhea (OR, 1.65), and abdominal pain (OR, 6.38). The severely ill patients were significantly more likely to have increased AST and ALT (OR, 2.98 and 2.66, respectively).
“However, there were no significant differences between severe/ICU group and general/non-ICU group for the prevalence of nausea and vomiting and liver disease,” the researchers said.
The study findings were limited by several factors including the unclear classification of digestive system disease and liver disease in many of the studies, the small sample sizes, and the lack of data on pathology of the liver or colon in COVID-19 patients, the researchers noted.
More research is needed, but the findings suggest that COVID-19 could contribute to liver damage because the most significant abnormal liver function was increased ALT, they said.
Check liver function in cases with GI symptoms
“COVID patients can present asymptomatically or with nonspecific symptoms, including GI symptoms,” said Ziad F. Gellad, MD, of Duke University Medical Center, Durham, N.C., in an interview. “While the focus of management naturally is directed to the pulmonary consequences of the disease, it is important to evaluate the patient holistically,” he said.
“I do not think these findings have profound clinical implications because they identify relatively nonspecific symptoms that are commonly seen in patients in a number of other conditions,” noted Dr. Gellad. “The management of COVID should not change, with the exception of perhaps making sure to check for abnormal liver function tests in patients that present with more typical COVID symptoms,” he said.
“Additional research is needed to understand the biologic mechanism by which COVID impacts systems outside of the lungs,” Dr. Gellad emphasized. “For example, there has been some very interesting work understanding the impact of COVID on the pancreas and risk for pancreatitis. That work is similarly needed to understand how COVID, outside of causing a general illness, specifically impacts the rest of the GI tract,” he said.
The study was supported by the Liaoning Science and Technology Foundation. The researchers had no financial conflicts to disclose. Dr. Gellad had no financial conflicts to disclose.
SOURCE: Dong Z-Y et al. J Clin Gastroenterol. 2021 Jan. doi: 10.1097/MCG.0000000000001424.
Patients with severe COVID-19 were significantly more likely than those with milder cases to have GI symptoms of anorexia and diarrhea, as well as abnormal liver function, based on data from a meta-analysis of more than 4,500 patients.
Previous studies have shown that liver damage “was more likely to be observed in severe patients during the process of disease,” and other studies have shown varying degrees of liver insufficiency in COVID-19 patients, but gastrointestinal symptoms have not been well studied, wrote Zi-yuan Dong, MD, of China Medical University, Shenyang City, and colleagues.
In a study published in the Journal of Clinical Gastroenterology, the researchers identified 31 studies including 4,682 COVID-19 patients. Case collection was from Dec. 11, 2019, to Feb. 28, 2020. Median age among studies ranged from 36 to 62 years, and 55% of patients were male.
A total of 26 studies were analyzed for the prevalence of GI symptoms, specifically nausea, vomiting, diarrhea, abdominal pain, and anorexia. Of these, anorexia and diarrhea were significantly more common in COVID-19 patients, with prevalence of 17% and 8% respectively, (P < .0001 for both).
In addition, 14 of the studies included in the analysis assessed the prevalence of abnormal liver function based on increased levels of aspartate aminotransferase, alanine aminotransferase, and total bilirubin. Of these, increased alanine aminotransferase was the most common, occurring in 25% of patients, compared with increased AST (in 24%) and total bilirubin (in 13%).
When assessed by disease severity, patients with severe disease and those in the ICU were significantly more likely than general/non-ICU patients to have anorexia (odds ratio, 2.19), diarrhea (OR, 1.65), and abdominal pain (OR, 6.38). The severely ill patients were significantly more likely to have increased AST and ALT (OR, 2.98 and 2.66, respectively).
“However, there were no significant differences between severe/ICU group and general/non-ICU group for the prevalence of nausea and vomiting and liver disease,” the researchers said.
The study findings were limited by several factors including the unclear classification of digestive system disease and liver disease in many of the studies, the small sample sizes, and the lack of data on pathology of the liver or colon in COVID-19 patients, the researchers noted.
More research is needed, but the findings suggest that COVID-19 could contribute to liver damage because the most significant abnormal liver function was increased ALT, they said.
Check liver function in cases with GI symptoms
“COVID patients can present asymptomatically or with nonspecific symptoms, including GI symptoms,” said Ziad F. Gellad, MD, of Duke University Medical Center, Durham, N.C., in an interview. “While the focus of management naturally is directed to the pulmonary consequences of the disease, it is important to evaluate the patient holistically,” he said.
“I do not think these findings have profound clinical implications because they identify relatively nonspecific symptoms that are commonly seen in patients in a number of other conditions,” noted Dr. Gellad. “The management of COVID should not change, with the exception of perhaps making sure to check for abnormal liver function tests in patients that present with more typical COVID symptoms,” he said.
“Additional research is needed to understand the biologic mechanism by which COVID impacts systems outside of the lungs,” Dr. Gellad emphasized. “For example, there has been some very interesting work understanding the impact of COVID on the pancreas and risk for pancreatitis. That work is similarly needed to understand how COVID, outside of causing a general illness, specifically impacts the rest of the GI tract,” he said.
The study was supported by the Liaoning Science and Technology Foundation. The researchers had no financial conflicts to disclose. Dr. Gellad had no financial conflicts to disclose.
SOURCE: Dong Z-Y et al. J Clin Gastroenterol. 2021 Jan. doi: 10.1097/MCG.0000000000001424.
Patients with severe COVID-19 were significantly more likely than those with milder cases to have GI symptoms of anorexia and diarrhea, as well as abnormal liver function, based on data from a meta-analysis of more than 4,500 patients.
Previous studies have shown that liver damage “was more likely to be observed in severe patients during the process of disease,” and other studies have shown varying degrees of liver insufficiency in COVID-19 patients, but gastrointestinal symptoms have not been well studied, wrote Zi-yuan Dong, MD, of China Medical University, Shenyang City, and colleagues.
In a study published in the Journal of Clinical Gastroenterology, the researchers identified 31 studies including 4,682 COVID-19 patients. Case collection was from Dec. 11, 2019, to Feb. 28, 2020. Median age among studies ranged from 36 to 62 years, and 55% of patients were male.
A total of 26 studies were analyzed for the prevalence of GI symptoms, specifically nausea, vomiting, diarrhea, abdominal pain, and anorexia. Of these, anorexia and diarrhea were significantly more common in COVID-19 patients, with prevalence of 17% and 8% respectively, (P < .0001 for both).
In addition, 14 of the studies included in the analysis assessed the prevalence of abnormal liver function based on increased levels of aspartate aminotransferase, alanine aminotransferase, and total bilirubin. Of these, increased alanine aminotransferase was the most common, occurring in 25% of patients, compared with increased AST (in 24%) and total bilirubin (in 13%).
When assessed by disease severity, patients with severe disease and those in the ICU were significantly more likely than general/non-ICU patients to have anorexia (odds ratio, 2.19), diarrhea (OR, 1.65), and abdominal pain (OR, 6.38). The severely ill patients were significantly more likely to have increased AST and ALT (OR, 2.98 and 2.66, respectively).
“However, there were no significant differences between severe/ICU group and general/non-ICU group for the prevalence of nausea and vomiting and liver disease,” the researchers said.
The study findings were limited by several factors including the unclear classification of digestive system disease and liver disease in many of the studies, the small sample sizes, and the lack of data on pathology of the liver or colon in COVID-19 patients, the researchers noted.
More research is needed, but the findings suggest that COVID-19 could contribute to liver damage because the most significant abnormal liver function was increased ALT, they said.
Check liver function in cases with GI symptoms
“COVID patients can present asymptomatically or with nonspecific symptoms, including GI symptoms,” said Ziad F. Gellad, MD, of Duke University Medical Center, Durham, N.C., in an interview. “While the focus of management naturally is directed to the pulmonary consequences of the disease, it is important to evaluate the patient holistically,” he said.
“I do not think these findings have profound clinical implications because they identify relatively nonspecific symptoms that are commonly seen in patients in a number of other conditions,” noted Dr. Gellad. “The management of COVID should not change, with the exception of perhaps making sure to check for abnormal liver function tests in patients that present with more typical COVID symptoms,” he said.
“Additional research is needed to understand the biologic mechanism by which COVID impacts systems outside of the lungs,” Dr. Gellad emphasized. “For example, there has been some very interesting work understanding the impact of COVID on the pancreas and risk for pancreatitis. That work is similarly needed to understand how COVID, outside of causing a general illness, specifically impacts the rest of the GI tract,” he said.
The study was supported by the Liaoning Science and Technology Foundation. The researchers had no financial conflicts to disclose. Dr. Gellad had no financial conflicts to disclose.
SOURCE: Dong Z-Y et al. J Clin Gastroenterol. 2021 Jan. doi: 10.1097/MCG.0000000000001424.
FROM THE JOURNAL OF CLINICAL GASTROENTEROLOGY
Complete blood count scoring can predict COVID-19 severity
A scoring system based on 10 parameters in a complete blood count with differential within 3 days of hospital presentation predict those with COVID-19 who are most likely to progress to critical illness, new evidence shows.
Advantages include prognosis based on a common and inexpensive clinical measure, as well as automatic generation of the score along with CBC results, noted investigators in the observational study conducted throughout 11 European hospitals.
“COVID-19 comes along with specific alterations in circulating blood cells that can be detected by a routine hematology analyzer, especially when that hematology analyzer is also capable to recognize activated immune cells and early circulating blood cells, such as erythroblast and immature granulocytes,” senior author Andre van der Ven, MD, PhD, infectious diseases specialist and professor of international health at Radboud University Medical Center’s Center for Infectious Diseases in Nijmegen, the Netherlands, said in an interview.
Furthermore, Dr. van der Ven said, “these specific changes are also seen in the early course of COVID-19 disease, and more in those that will develop serious disease compared to those with mild disease.”
The study was published online Dec. 21 in the journal eLife.
The study is “almost instinctively correct. It’s basically what clinicians do informally with complete blood count … looking at a combination of results to get the gestalt of what patients are going through,” Samuel Reichberg, MD, PhD, associate medical director of the Northwell Health Core Laboratory in Lake Success, N.Y., said in an interview.
“This is something that begs to be done for COVID-19. I’m surprised no one has done this before,” he added.
Dr. Van der Ven and colleagues created an algorithm based on 1,587 CBC assays from 923 adults. They also validated the scoring system in a second cohort of 217 CBC measurements in 202 people. The findings were concordant – the score accurately predicted the need for critical care within 14 days in 70.5% of the development cohort and 72% of the validation group.
The scoring system was superior to any of the 10 parameters alone. Over 14 days, the majority of those classified as noncritical within the first 3 days remained clinically stable, whereas the “clinical illness” group progressed. Clinical severity peaked on day 6.
Most previous COVID-19 prognosis research was geographically limited, carried a high risk for bias and/or did not validate the findings, Dr. Van der Ven and colleagues noted.
Early identification, early intervention
The aim of the score is “to assist with objective risk stratification to support patient management decision-making early on, and thus facilitate timely interventions, such as need for ICU or not, before symptoms of severe illness become clinically overt, with the intention to improve patient outcomes, and not to predict mortality,” the investigators noted.
Dr. Van der Ven and colleagues developed the score based on adults presenting from Feb. 21 to April 6, with outcomes followed until June 9. Median age of the 982 patients was 71 years and approximately two-thirds were men. They used a Sysmex Europe XN-1000 (Hamburg, Germany) hemocytometric analyzer in the study.
Only 7% of this cohort was not admitted to a hospital. Another 74% were admitted to a general ward and the remaining 19% were transferred directly to the ICU.
The scoring system includes parameters for neutrophils, monocytes, red blood cells and immature granulocytes, and when available, reticulocyte and iron bioavailability measures.
The researchers report significant differences over time in the neutrophil-to-lymphocyte ratio between the critical illness and noncritical groups (P < .001), for example. They also found significant differences in hemoglobin levels between cohorts after day 5.
The system generates a score from 0 to 28. Sensitivity for correctly predicting the need for critical care increased from 62% on day 1 to 93% on day 6.
A more objective assessment of risk
The study demonstrated that SARS-CoV-2 infection is characterized by hemocytometric changes over time. These changes, reflected together in the prognostic score, could aid in the early identification of patients whose clinical course is more likely to deteriorate over time.
The findings also support other work that shows men are more likely to present to the hospital with COVID-19, and that older age and presence of comorbidities add to overall risk. “However,” the researchers noted, “not all young patients had a mild course, and not all old patients with comorbidities were critical.”
Therefore, the prognostic score can help identify patients at risk for severe progression outside other risk factors and “support individualized treatment decisions with objective data,” they added.
Dr. Reichberg called the concept of combining CBC parameters into one score “very valuable.” However, he added that incorporating an index into clinical practice “has historically been tricky.”
The results “probably have to be replicated,” Dr. Reichberg said.
He added that it is likely a CBC-based score will be combined with other measures. “I would like to see an index that combines all the tests we do [for COVID-19], including complete blood count.”
Dr. Van der Ven shared the next step in his research. “The algorithm should be installed on the hematology analyzers so the prognostic score will be automatically generated if a full blood count is asked for in a COVID-19 patient,” he said. “So implementation of score is the main focus now.”
Dr. van der Ven disclosed an ad hoc consultancy agreement with Sysmex Europe. Sysmex Europe provided the reagents in the study free of charge; no other funders were involved. Dr. Reichberg has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A scoring system based on 10 parameters in a complete blood count with differential within 3 days of hospital presentation predict those with COVID-19 who are most likely to progress to critical illness, new evidence shows.
Advantages include prognosis based on a common and inexpensive clinical measure, as well as automatic generation of the score along with CBC results, noted investigators in the observational study conducted throughout 11 European hospitals.
“COVID-19 comes along with specific alterations in circulating blood cells that can be detected by a routine hematology analyzer, especially when that hematology analyzer is also capable to recognize activated immune cells and early circulating blood cells, such as erythroblast and immature granulocytes,” senior author Andre van der Ven, MD, PhD, infectious diseases specialist and professor of international health at Radboud University Medical Center’s Center for Infectious Diseases in Nijmegen, the Netherlands, said in an interview.
Furthermore, Dr. van der Ven said, “these specific changes are also seen in the early course of COVID-19 disease, and more in those that will develop serious disease compared to those with mild disease.”
The study was published online Dec. 21 in the journal eLife.
The study is “almost instinctively correct. It’s basically what clinicians do informally with complete blood count … looking at a combination of results to get the gestalt of what patients are going through,” Samuel Reichberg, MD, PhD, associate medical director of the Northwell Health Core Laboratory in Lake Success, N.Y., said in an interview.
“This is something that begs to be done for COVID-19. I’m surprised no one has done this before,” he added.
Dr. Van der Ven and colleagues created an algorithm based on 1,587 CBC assays from 923 adults. They also validated the scoring system in a second cohort of 217 CBC measurements in 202 people. The findings were concordant – the score accurately predicted the need for critical care within 14 days in 70.5% of the development cohort and 72% of the validation group.
The scoring system was superior to any of the 10 parameters alone. Over 14 days, the majority of those classified as noncritical within the first 3 days remained clinically stable, whereas the “clinical illness” group progressed. Clinical severity peaked on day 6.
Most previous COVID-19 prognosis research was geographically limited, carried a high risk for bias and/or did not validate the findings, Dr. Van der Ven and colleagues noted.
Early identification, early intervention
The aim of the score is “to assist with objective risk stratification to support patient management decision-making early on, and thus facilitate timely interventions, such as need for ICU or not, before symptoms of severe illness become clinically overt, with the intention to improve patient outcomes, and not to predict mortality,” the investigators noted.
Dr. Van der Ven and colleagues developed the score based on adults presenting from Feb. 21 to April 6, with outcomes followed until June 9. Median age of the 982 patients was 71 years and approximately two-thirds were men. They used a Sysmex Europe XN-1000 (Hamburg, Germany) hemocytometric analyzer in the study.
Only 7% of this cohort was not admitted to a hospital. Another 74% were admitted to a general ward and the remaining 19% were transferred directly to the ICU.
The scoring system includes parameters for neutrophils, monocytes, red blood cells and immature granulocytes, and when available, reticulocyte and iron bioavailability measures.
The researchers report significant differences over time in the neutrophil-to-lymphocyte ratio between the critical illness and noncritical groups (P < .001), for example. They also found significant differences in hemoglobin levels between cohorts after day 5.
The system generates a score from 0 to 28. Sensitivity for correctly predicting the need for critical care increased from 62% on day 1 to 93% on day 6.
A more objective assessment of risk
The study demonstrated that SARS-CoV-2 infection is characterized by hemocytometric changes over time. These changes, reflected together in the prognostic score, could aid in the early identification of patients whose clinical course is more likely to deteriorate over time.
The findings also support other work that shows men are more likely to present to the hospital with COVID-19, and that older age and presence of comorbidities add to overall risk. “However,” the researchers noted, “not all young patients had a mild course, and not all old patients with comorbidities were critical.”
Therefore, the prognostic score can help identify patients at risk for severe progression outside other risk factors and “support individualized treatment decisions with objective data,” they added.
Dr. Reichberg called the concept of combining CBC parameters into one score “very valuable.” However, he added that incorporating an index into clinical practice “has historically been tricky.”
The results “probably have to be replicated,” Dr. Reichberg said.
He added that it is likely a CBC-based score will be combined with other measures. “I would like to see an index that combines all the tests we do [for COVID-19], including complete blood count.”
Dr. Van der Ven shared the next step in his research. “The algorithm should be installed on the hematology analyzers so the prognostic score will be automatically generated if a full blood count is asked for in a COVID-19 patient,” he said. “So implementation of score is the main focus now.”
Dr. van der Ven disclosed an ad hoc consultancy agreement with Sysmex Europe. Sysmex Europe provided the reagents in the study free of charge; no other funders were involved. Dr. Reichberg has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A scoring system based on 10 parameters in a complete blood count with differential within 3 days of hospital presentation predict those with COVID-19 who are most likely to progress to critical illness, new evidence shows.
Advantages include prognosis based on a common and inexpensive clinical measure, as well as automatic generation of the score along with CBC results, noted investigators in the observational study conducted throughout 11 European hospitals.
“COVID-19 comes along with specific alterations in circulating blood cells that can be detected by a routine hematology analyzer, especially when that hematology analyzer is also capable to recognize activated immune cells and early circulating blood cells, such as erythroblast and immature granulocytes,” senior author Andre van der Ven, MD, PhD, infectious diseases specialist and professor of international health at Radboud University Medical Center’s Center for Infectious Diseases in Nijmegen, the Netherlands, said in an interview.
Furthermore, Dr. van der Ven said, “these specific changes are also seen in the early course of COVID-19 disease, and more in those that will develop serious disease compared to those with mild disease.”
The study was published online Dec. 21 in the journal eLife.
The study is “almost instinctively correct. It’s basically what clinicians do informally with complete blood count … looking at a combination of results to get the gestalt of what patients are going through,” Samuel Reichberg, MD, PhD, associate medical director of the Northwell Health Core Laboratory in Lake Success, N.Y., said in an interview.
“This is something that begs to be done for COVID-19. I’m surprised no one has done this before,” he added.
Dr. Van der Ven and colleagues created an algorithm based on 1,587 CBC assays from 923 adults. They also validated the scoring system in a second cohort of 217 CBC measurements in 202 people. The findings were concordant – the score accurately predicted the need for critical care within 14 days in 70.5% of the development cohort and 72% of the validation group.
The scoring system was superior to any of the 10 parameters alone. Over 14 days, the majority of those classified as noncritical within the first 3 days remained clinically stable, whereas the “clinical illness” group progressed. Clinical severity peaked on day 6.
Most previous COVID-19 prognosis research was geographically limited, carried a high risk for bias and/or did not validate the findings, Dr. Van der Ven and colleagues noted.
Early identification, early intervention
The aim of the score is “to assist with objective risk stratification to support patient management decision-making early on, and thus facilitate timely interventions, such as need for ICU or not, before symptoms of severe illness become clinically overt, with the intention to improve patient outcomes, and not to predict mortality,” the investigators noted.
Dr. Van der Ven and colleagues developed the score based on adults presenting from Feb. 21 to April 6, with outcomes followed until June 9. Median age of the 982 patients was 71 years and approximately two-thirds were men. They used a Sysmex Europe XN-1000 (Hamburg, Germany) hemocytometric analyzer in the study.
Only 7% of this cohort was not admitted to a hospital. Another 74% were admitted to a general ward and the remaining 19% were transferred directly to the ICU.
The scoring system includes parameters for neutrophils, monocytes, red blood cells and immature granulocytes, and when available, reticulocyte and iron bioavailability measures.
The researchers report significant differences over time in the neutrophil-to-lymphocyte ratio between the critical illness and noncritical groups (P < .001), for example. They also found significant differences in hemoglobin levels between cohorts after day 5.
The system generates a score from 0 to 28. Sensitivity for correctly predicting the need for critical care increased from 62% on day 1 to 93% on day 6.
A more objective assessment of risk
The study demonstrated that SARS-CoV-2 infection is characterized by hemocytometric changes over time. These changes, reflected together in the prognostic score, could aid in the early identification of patients whose clinical course is more likely to deteriorate over time.
The findings also support other work that shows men are more likely to present to the hospital with COVID-19, and that older age and presence of comorbidities add to overall risk. “However,” the researchers noted, “not all young patients had a mild course, and not all old patients with comorbidities were critical.”
Therefore, the prognostic score can help identify patients at risk for severe progression outside other risk factors and “support individualized treatment decisions with objective data,” they added.
Dr. Reichberg called the concept of combining CBC parameters into one score “very valuable.” However, he added that incorporating an index into clinical practice “has historically been tricky.”
The results “probably have to be replicated,” Dr. Reichberg said.
He added that it is likely a CBC-based score will be combined with other measures. “I would like to see an index that combines all the tests we do [for COVID-19], including complete blood count.”
Dr. Van der Ven shared the next step in his research. “The algorithm should be installed on the hematology analyzers so the prognostic score will be automatically generated if a full blood count is asked for in a COVID-19 patient,” he said. “So implementation of score is the main focus now.”
Dr. van der Ven disclosed an ad hoc consultancy agreement with Sysmex Europe. Sysmex Europe provided the reagents in the study free of charge; no other funders were involved. Dr. Reichberg has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New pediatric cases down as U.S. tops 2 million children with COVID-19
The United States exceeded 2 million reported cases of COVID-19 in children just 6 weeks after recording its 1 millionth case, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
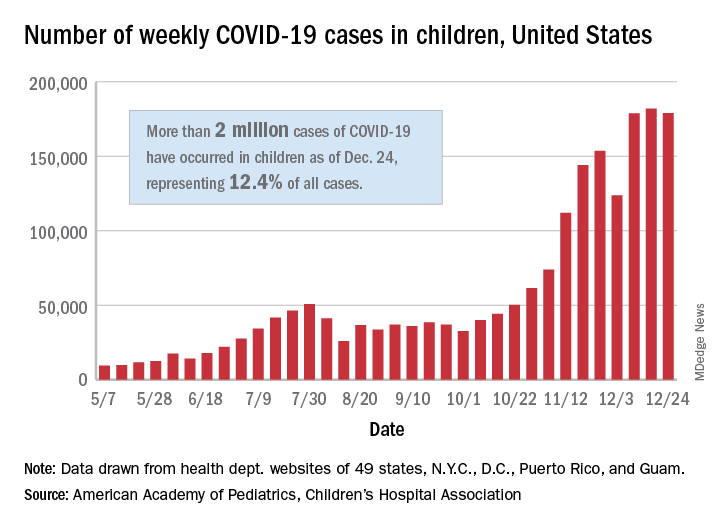
The total number of cases in children was 2,000,681 as of Dec. 24, which represents 12.4% of all cases reported by the health departments of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam, the AAP and CHA stated Dec. 29.
The case count for just the latest week, 178,935, was actually down 1.7% from the 182,018 reported the week before, marking the second drop since the beginning of December. The first came during the week ending Dec. 3, when the number of cases dropped more than 19% from the previous week, based on data from the AAP/CHA report.
The cumulative national rate of coronavirus infection is now 2,658 cases per 100,000 children, and “13 states have reported more than 4,000 cases per 100,000,” the two groups said.
The highest rate for any state can be found in North Dakota, which has had 7,722 cases of COVID-19 per 100,000 children. Wyoming has the highest proportion of cases in children at 20.5%, and California has reported the most cases overall, 234,174, the report shows.
Data on testing, hospitalization, and mortality were not included in the Dec. 29 report because of the holiday but will be available in the next edition, scheduled for release on Jan. 5, 2021.
The United States exceeded 2 million reported cases of COVID-19 in children just 6 weeks after recording its 1 millionth case, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

The total number of cases in children was 2,000,681 as of Dec. 24, which represents 12.4% of all cases reported by the health departments of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam, the AAP and CHA stated Dec. 29.
The case count for just the latest week, 178,935, was actually down 1.7% from the 182,018 reported the week before, marking the second drop since the beginning of December. The first came during the week ending Dec. 3, when the number of cases dropped more than 19% from the previous week, based on data from the AAP/CHA report.
The cumulative national rate of coronavirus infection is now 2,658 cases per 100,000 children, and “13 states have reported more than 4,000 cases per 100,000,” the two groups said.
The highest rate for any state can be found in North Dakota, which has had 7,722 cases of COVID-19 per 100,000 children. Wyoming has the highest proportion of cases in children at 20.5%, and California has reported the most cases overall, 234,174, the report shows.
Data on testing, hospitalization, and mortality were not included in the Dec. 29 report because of the holiday but will be available in the next edition, scheduled for release on Jan. 5, 2021.
The United States exceeded 2 million reported cases of COVID-19 in children just 6 weeks after recording its 1 millionth case, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

The total number of cases in children was 2,000,681 as of Dec. 24, which represents 12.4% of all cases reported by the health departments of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam, the AAP and CHA stated Dec. 29.
The case count for just the latest week, 178,935, was actually down 1.7% from the 182,018 reported the week before, marking the second drop since the beginning of December. The first came during the week ending Dec. 3, when the number of cases dropped more than 19% from the previous week, based on data from the AAP/CHA report.
The cumulative national rate of coronavirus infection is now 2,658 cases per 100,000 children, and “13 states have reported more than 4,000 cases per 100,000,” the two groups said.
The highest rate for any state can be found in North Dakota, which has had 7,722 cases of COVID-19 per 100,000 children. Wyoming has the highest proportion of cases in children at 20.5%, and California has reported the most cases overall, 234,174, the report shows.
Data on testing, hospitalization, and mortality were not included in the Dec. 29 report because of the holiday but will be available in the next edition, scheduled for release on Jan. 5, 2021.
COVID-19 vaccines: Safe for immunocompromised patients?
Coronavirus vaccines have become a reality, as they are now being approved and authorized for use in a growing number of countries including the United States. The U.S. Food and Drug Administration has just issued emergency authorization for the use of the COVID-19 vaccine produced by Pfizer and BioNTech. Close behind is the vaccine developed by Moderna, which has also applied to the FDA for emergency authorization.
The efficacy of a two-dose administration of the vaccine has been pegged at 95.0%, and the FDA has said that the 95% credible interval for the vaccine efficacy was 90.3%-97.6%. But as with many initial clinical trials, whether for drugs or vaccines, not all populations were represented in the trial cohort, including individuals who are immunocompromised. At the current time, it is largely unknown how safe or effective the vaccine may be in this large population, many of whom are at high risk for serious COVID-19 complications.
At a special session held during the recent annual meeting of the American Society of Hematology, Anthony Fauci, MD, the nation’s leading infectious disease expert, said that individuals with compromised immune systems, whether because of chemotherapy or a bone marrow transplant, should plan to be vaccinated when the opportunity arises.
In response to a question from ASH President Stephanie J. Lee, MD, of the Fred Hutchinson Cancer Center, Seattle, Dr. Fauci emphasized that, despite being excluded from clinical trials, this population should get vaccinated. “I think we should recommend that they get vaccinated,” he said. “I mean, it is clear that, if you are on immunosuppressive agents, history tells us that you’re not going to have as robust a response as if you had an intact immune system that was not being compromised. But some degree of immunity is better than no degree of immunity.”
That does seem to be the consensus among experts who spoke in interviews: that as long as these are not live attenuated vaccines, they hold no specific risk to an immunocompromised patient, other than any factors specific to the individual that could be a contraindication.
“Patients, family members, friends, and work contacts should be encouraged to receive the vaccine,” said William Stohl, MD, PhD, chief of the division of rheumatology at the University of Southern California, Los Angeles. “Clinicians should advise patients to obtain the vaccine sooner rather than later.”
Kevin C. Wang, MD, PhD, of the department of dermatology at Stanford (Calif.) University, agreed. “I am 100% with Dr. Fauci. Everyone should get the vaccine, even if it may not be as effective,” he said. “I would treat it exactly like the flu vaccines that we recommend folks get every year.”
Dr. Wang noted that he couldn’t think of any contraindications unless the immunosuppressed patients have a history of severe allergic reactions to prior vaccinations. “But I would even say patients with history of cancer, upon recommendation of their oncologists, are likely to be suitable candidates for the vaccine,” he added. “I would say clinicians should approach counseling the same way they counsel patients for the flu vaccine, and as far as I know, there are no concerns for systemic drugs commonly used in dermatology patients.”
However, guidance has not yet been issued from either the FDA or the Centers for Disease Control and Prevention regarding the use of the vaccine in immunocompromised individuals. Given the lack of data, the FDA has said that “it will be something that providers will need to consider on an individual basis,” and that individuals should consult with physicians to weigh the potential benefits and potential risks.
The CDC’s Advisory Committee on Immunization Practices has said that clinicians need more guidance on whether to use the vaccine in pregnant or breastfeeding women, the immunocompromised, or those who have a history of allergies. The CDC itself has not yet released its formal guidance on vaccine use.
COVID-19 vaccines
Vaccines typically require years of research and testing before reaching the clinic, but this year researchers embarked on a global effort to develop safe and effective coronavirus vaccines in record time. Both the Pfizer/BioNTech and Moderna vaccines have only a few months of phase 3 clinical trial data, so much remains unknown about them, including their duration of effect and any long-term safety signals. In addition to excluding immunocompromised individuals, the clinical trials did not include children or pregnant women, so data are lacking for several population subgroups.
But these will not be the only vaccines available, as the pipeline is already becoming crowded. U.S. clinical trial data from a vaccine jointly being developed by Oxford-AstraZeneca, could potentially be ready, along with a request for FDA emergency use authorization, by late January 2021.
In addition, China and Russia have released vaccines, and there are currently 61 vaccines being investigated in clinical trials and at least 85 preclinical products under active investigation.
The vaccine candidates are using both conventional and novel mechanisms of action to elicit an immune response in patients. Conventional methods include attenuated inactivated (killed) virus and recombinant viral protein vaccines to develop immunity. Novel approaches include replication-deficient, adenovirus vector-based vaccines that contain the viral protein, and mRNA-based vaccines, such as the Pfizer and Moderna vaccines, that encode for a SARS-CoV-2 spike protein.
“The special vaccine concern for immunocompromised individuals is introduction of a live virus,” Dr. Stohl said. “Neither the Moderna nor Pfizer vaccines are live viruses, so there should be no special contraindication for such individuals.”
Live vaccine should be avoided in immunocompromised patients, and currently, live SARS-CoV-2 vaccines are only being developed in India and Turkey.
It is not unusual for vaccine trials to begin with cohorts that exclude participants with various health conditions, including those who are immunocompromised. These groups are generally then evaluated in phase 4 trials, or postmarketing surveillance. While the precise number of immunosuppressed adults in the United States is not known, the numbers are believed to be rising because of increased life expectancy among immunosuppressed adults as a result of advances in treatment and new and wider indications for therapies that can affect the immune system.
According to data from the 2013 National Health Interview Survey, an estimated 2.7% of U.S. adults are immunosuppressed. This population covers a broad array of health conditions and medical specialties; people living with inflammatory or autoimmune conditions, such as inflammatory rheumatic diseases (rheumatoid arthritis, axial spondyloarthritis, lupus); inflammatory bowel disease (Crohn’s disease and ulcerative colitis); psoriasis; multiple sclerosis; organ transplant recipients; patients undergoing chemotherapy; and life-long immunosuppression attributable to HIV infection.
As the vaccines begin to roll out and become available, how should clinicians advise their patients, in the absence of any clinical trial data?
Risk vs. benefit
Gilaad Kaplan, MD, MPH, a gastroenterologist and professor of medicine at the University of Calgary (Alta.), noted that the inflammatory bowel disease (IBD) community has dealt with tremendous anxiety during the pandemic because many are immunocompromised because of the medications they use to treat their disease.
“For example, many patients with IBD are on biologics like anti-TNF [tumor necrosis factor] therapies, which are also used in other immune-mediated inflammatory diseases such as rheumatoid arthritis,” he said. “Understandably, individuals with IBD on immunosuppressive medications are concerned about the risk of severe complications due to COVID-19.”
The entire IBD community, along with the world, celebrated the announcement that multiple vaccines are protective against SARS-CoV-2, he noted. “Vaccines offer the potential to reduce the spread of COVID-19, allowing society to revert back to normalcy,” Dr. Kaplan said. “Moreover, for vulnerable populations, including those who are immunocompromised, vaccines offer the potential to directly protect them from the morbidity and mortality associated with COVID-19.”
That said, even though the news of vaccines are extremely promising, some cautions must be raised regarding their use in immunocompromised populations, such as persons with IBD. “The current trials, to my knowledge, did not include immunocompromised individuals and thus, we can only extrapolate from what we know from other trials of different vaccines,” he explained. “We know from prior vaccines studies that the immune response following vaccination is less robust in those who are immunocompromised as compared to a healthy control population.”
Dr. Kaplan also pointed to recent reports of allergic reactions that have been reported in healthy individuals. “We don’t know whether side effects, like allergic reactions, may be different in unstudied populations,” he said. “Thus, the medical and scientific community should prioritize clinical studies of safety and effectiveness of COVID-19 vaccines in immunocompromised populations.”
So, what does this mean for an individual with an immune-mediated inflammatory disease like Crohn’s disease or ulcerative colitis who is immunocompromised? Dr. Kaplan explained that it is a balance between the potential harm of being infected with COVID-19 and the uncertainty of receiving a vaccine in an understudied population. For those who are highly susceptible to dying from COVID-19, such as an older adult with IBD, or someone who faces high exposure, such as a health care worker, the potential protection of the vaccine greatly outweighs the uncertainty.
“However, for individuals who are at otherwise lower risk – for example, young and able to work from home – then waiting a few extra months for postmarketing surveillance studies in immunocompromised populations may be a reasonable approach, as long as these individuals are taking great care to avoid infection,” he said.
No waiting needed
Joel M. Gelfand, MD, MSCE, professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, feels that the newly approved vaccine should be safe for most of his patients.
“Patients with psoriatic disease should get the mRNA-based COVID-19 vaccine as soon as possible based on eligibility as determined by the CDC and local public health officials,” he said. “It is not a live vaccine, and therefore patients on biologics or other immune-modulating or immune-suppressing treatment can receive it.”
However, the impact of psoriasis treatment on immune response to the mRNA-based vaccines is not known. Dr. Gelfand noted that, extrapolating from the vaccine literature, there is some evidence that methotrexate reduces response to the influenza vaccine. “However, the clinical significance of this finding is not clear,” he said. “Since the mRNA vaccine needs to be taken twice, a few weeks apart, I do not recommend interrupting or delaying treatment for psoriatic disease while undergoing vaccination for COVID-19.”
Given the reports of allergic reactions, he added that it is advisable for patients with a history of life-threatening allergic reactions such as anaphylaxis or who have been advised to carry an epinephrine autoinjector, to talk with their health care provider to determine if COVID-19 vaccination is medically appropriate.
The National Psoriasis Foundation has issued guidance on COVID-19, explained Steven R. Feldman, MD, PhD, professor of dermatology, pathology, and social sciences & health policy at Wake Forest University, Winston-Salem, N.C., who is also a member of the committee that is working on those guidelines and keeping them up to date. “We are in the process of updating the guidelines with information on COVID vaccines,” he said.
He agreed that there are no contraindications for psoriasis patients to receive the vaccine, regardless of whether they are on immunosuppressive treatment, even though definitive data are lacking. “Fortunately, there’s a lot of good data coming out of Italy that patients with psoriasis on biologics do not appear to be at increased risk of getting COVID or of having worse outcomes from COVID,” he said.
Patients are going to ask about the vaccines, and when counseling them, clinicians should discuss the available data, the residual uncertainty, and patients’ concerns should be considered, Dr. Feldman explained. “There may be some concern that steroids and cyclosporine would reduce the effectiveness of vaccines, but there is no concern that any of the drugs would cause increased risk from nonlive vaccines.”
He added that there is evidence that “patients on biologics who receive nonlive vaccines do develop antibody responses and are immunized.”
Boosting efficacy
Even prior to making their announcement, the American College of Rheumatology had said that they would endorse the vaccine for all patients, explained rheumatologist Brett Smith, DO, from Blount Memorial Physicians Group and East Tennessee Children’s Hospital, Alcoa. “The vaccine is safe for all patients, but the problem may be that it’s not as effective,” he said. “But we don’t know that because it hasn’t been tested.”
With other vaccines, biologic medicines are held for 2 weeks before and afterwards, to get the best response. “But some patients don’t want to stop the medication,” Dr. Smith said. “They are afraid that their symptoms will return.”
As for counseling patients as to whether they should receive this vaccine, he explained that he typically doesn’t try to sway patients one way or another until they are really high risk. “When I counsel, it really depends on the individual situation. And for this vaccine, we have to be open to the fact that many people have already made up their mind.”
There are a lot of questions regarding the vaccine. One is the short time frame of development. “Vaccines typically take 6-10 years to come on the market, and this one is now available after a 3-month study,” Dr. Smith said. “Some have already decided that it’s too new for them.”
The process is also new, and patients need to understand that it doesn’t contain an active virus and “you can’t catch coronavirus from it.”
Dr. Smith also explained that, because the vaccine may be less effective in a person using biologic therapies, there is currently no information available on repeat vaccination. “These are all unanswered questions,” he said. “If the antibodies wane in a short time, can we be revaccinated and in what time frame? We just don’t know that yet.”
Marcelo Bonomi, MD, a medical oncologist from The Ohio State University Comprehensive Cancer Center, Columbus, explained that one way to ensure a more optimal response to the vaccine would be to wait until the patient has finished chemotherapy.* “The vaccine can be offered at that time, and in the meantime, they can take other steps to avoid infection,” he said. “If they are very immunosuppressed, it isn’t worth trying to give the vaccine.”
Cancer patients should be encouraged to stay as healthy as possible, and to wear masks and social distance. “It’s a comprehensive approach. Eat healthy, avoid alcohol and tobacco, and exercise. [These things] will help boost the immune system,” Dr. Bonomi said. “Family members should be encouraged to get vaccinated, which will help them avoid infection and exposing the patient.”
Jim Boonyaratanakornkit, MD, PhD, an infectious disease specialist who cares for cancer patients at the Fred Hutchinson Cancer Research Center, agreed. “Giving a vaccine right after a transplant is a futile endeavor,” he said. “We need to wait 6 months to have an immune response.”
He pointed out there may be a continuing higher number of cases, with high levels peaking in Washington in February and March. “Close friends and family should be vaccinated if possible,” he said, “which will help interrupt transmission.”
The vaccines are using new platforms that are totally different, and there is no clear data as to how long the antibodies will persist. “We know that they last for at least 4 months,” said Dr. Boonyaratanakornkit. “We don’t know what level of antibody will protect them from COVID-19 infection. Current studies are being conducted, but we don’t have that information for anyone yet.”
*Correction, 1/7/21: An earlier version of this article misattributed quotes from Dr. Marcelo Bonomi.
For the latest clinical guidance, education, research and physician resources about coronavirus, visit the AGA COVID-19 Resource Center at www.gastro.org/COVID.
Coronavirus vaccines have become a reality, as they are now being approved and authorized for use in a growing number of countries including the United States. The U.S. Food and Drug Administration has just issued emergency authorization for the use of the COVID-19 vaccine produced by Pfizer and BioNTech. Close behind is the vaccine developed by Moderna, which has also applied to the FDA for emergency authorization.
The efficacy of a two-dose administration of the vaccine has been pegged at 95.0%, and the FDA has said that the 95% credible interval for the vaccine efficacy was 90.3%-97.6%. But as with many initial clinical trials, whether for drugs or vaccines, not all populations were represented in the trial cohort, including individuals who are immunocompromised. At the current time, it is largely unknown how safe or effective the vaccine may be in this large population, many of whom are at high risk for serious COVID-19 complications.
At a special session held during the recent annual meeting of the American Society of Hematology, Anthony Fauci, MD, the nation’s leading infectious disease expert, said that individuals with compromised immune systems, whether because of chemotherapy or a bone marrow transplant, should plan to be vaccinated when the opportunity arises.
In response to a question from ASH President Stephanie J. Lee, MD, of the Fred Hutchinson Cancer Center, Seattle, Dr. Fauci emphasized that, despite being excluded from clinical trials, this population should get vaccinated. “I think we should recommend that they get vaccinated,” he said. “I mean, it is clear that, if you are on immunosuppressive agents, history tells us that you’re not going to have as robust a response as if you had an intact immune system that was not being compromised. But some degree of immunity is better than no degree of immunity.”
That does seem to be the consensus among experts who spoke in interviews: that as long as these are not live attenuated vaccines, they hold no specific risk to an immunocompromised patient, other than any factors specific to the individual that could be a contraindication.
“Patients, family members, friends, and work contacts should be encouraged to receive the vaccine,” said William Stohl, MD, PhD, chief of the division of rheumatology at the University of Southern California, Los Angeles. “Clinicians should advise patients to obtain the vaccine sooner rather than later.”
Kevin C. Wang, MD, PhD, of the department of dermatology at Stanford (Calif.) University, agreed. “I am 100% with Dr. Fauci. Everyone should get the vaccine, even if it may not be as effective,” he said. “I would treat it exactly like the flu vaccines that we recommend folks get every year.”
Dr. Wang noted that he couldn’t think of any contraindications unless the immunosuppressed patients have a history of severe allergic reactions to prior vaccinations. “But I would even say patients with history of cancer, upon recommendation of their oncologists, are likely to be suitable candidates for the vaccine,” he added. “I would say clinicians should approach counseling the same way they counsel patients for the flu vaccine, and as far as I know, there are no concerns for systemic drugs commonly used in dermatology patients.”
However, guidance has not yet been issued from either the FDA or the Centers for Disease Control and Prevention regarding the use of the vaccine in immunocompromised individuals. Given the lack of data, the FDA has said that “it will be something that providers will need to consider on an individual basis,” and that individuals should consult with physicians to weigh the potential benefits and potential risks.
The CDC’s Advisory Committee on Immunization Practices has said that clinicians need more guidance on whether to use the vaccine in pregnant or breastfeeding women, the immunocompromised, or those who have a history of allergies. The CDC itself has not yet released its formal guidance on vaccine use.
COVID-19 vaccines
Vaccines typically require years of research and testing before reaching the clinic, but this year researchers embarked on a global effort to develop safe and effective coronavirus vaccines in record time. Both the Pfizer/BioNTech and Moderna vaccines have only a few months of phase 3 clinical trial data, so much remains unknown about them, including their duration of effect and any long-term safety signals. In addition to excluding immunocompromised individuals, the clinical trials did not include children or pregnant women, so data are lacking for several population subgroups.
But these will not be the only vaccines available, as the pipeline is already becoming crowded. U.S. clinical trial data from a vaccine jointly being developed by Oxford-AstraZeneca, could potentially be ready, along with a request for FDA emergency use authorization, by late January 2021.
In addition, China and Russia have released vaccines, and there are currently 61 vaccines being investigated in clinical trials and at least 85 preclinical products under active investigation.
The vaccine candidates are using both conventional and novel mechanisms of action to elicit an immune response in patients. Conventional methods include attenuated inactivated (killed) virus and recombinant viral protein vaccines to develop immunity. Novel approaches include replication-deficient, adenovirus vector-based vaccines that contain the viral protein, and mRNA-based vaccines, such as the Pfizer and Moderna vaccines, that encode for a SARS-CoV-2 spike protein.
“The special vaccine concern for immunocompromised individuals is introduction of a live virus,” Dr. Stohl said. “Neither the Moderna nor Pfizer vaccines are live viruses, so there should be no special contraindication for such individuals.”
Live vaccine should be avoided in immunocompromised patients, and currently, live SARS-CoV-2 vaccines are only being developed in India and Turkey.
It is not unusual for vaccine trials to begin with cohorts that exclude participants with various health conditions, including those who are immunocompromised. These groups are generally then evaluated in phase 4 trials, or postmarketing surveillance. While the precise number of immunosuppressed adults in the United States is not known, the numbers are believed to be rising because of increased life expectancy among immunosuppressed adults as a result of advances in treatment and new and wider indications for therapies that can affect the immune system.
According to data from the 2013 National Health Interview Survey, an estimated 2.7% of U.S. adults are immunosuppressed. This population covers a broad array of health conditions and medical specialties; people living with inflammatory or autoimmune conditions, such as inflammatory rheumatic diseases (rheumatoid arthritis, axial spondyloarthritis, lupus); inflammatory bowel disease (Crohn’s disease and ulcerative colitis); psoriasis; multiple sclerosis; organ transplant recipients; patients undergoing chemotherapy; and life-long immunosuppression attributable to HIV infection.
As the vaccines begin to roll out and become available, how should clinicians advise their patients, in the absence of any clinical trial data?
Risk vs. benefit
Gilaad Kaplan, MD, MPH, a gastroenterologist and professor of medicine at the University of Calgary (Alta.), noted that the inflammatory bowel disease (IBD) community has dealt with tremendous anxiety during the pandemic because many are immunocompromised because of the medications they use to treat their disease.
“For example, many patients with IBD are on biologics like anti-TNF [tumor necrosis factor] therapies, which are also used in other immune-mediated inflammatory diseases such as rheumatoid arthritis,” he said. “Understandably, individuals with IBD on immunosuppressive medications are concerned about the risk of severe complications due to COVID-19.”
The entire IBD community, along with the world, celebrated the announcement that multiple vaccines are protective against SARS-CoV-2, he noted. “Vaccines offer the potential to reduce the spread of COVID-19, allowing society to revert back to normalcy,” Dr. Kaplan said. “Moreover, for vulnerable populations, including those who are immunocompromised, vaccines offer the potential to directly protect them from the morbidity and mortality associated with COVID-19.”
That said, even though the news of vaccines are extremely promising, some cautions must be raised regarding their use in immunocompromised populations, such as persons with IBD. “The current trials, to my knowledge, did not include immunocompromised individuals and thus, we can only extrapolate from what we know from other trials of different vaccines,” he explained. “We know from prior vaccines studies that the immune response following vaccination is less robust in those who are immunocompromised as compared to a healthy control population.”
Dr. Kaplan also pointed to recent reports of allergic reactions that have been reported in healthy individuals. “We don’t know whether side effects, like allergic reactions, may be different in unstudied populations,” he said. “Thus, the medical and scientific community should prioritize clinical studies of safety and effectiveness of COVID-19 vaccines in immunocompromised populations.”
So, what does this mean for an individual with an immune-mediated inflammatory disease like Crohn’s disease or ulcerative colitis who is immunocompromised? Dr. Kaplan explained that it is a balance between the potential harm of being infected with COVID-19 and the uncertainty of receiving a vaccine in an understudied population. For those who are highly susceptible to dying from COVID-19, such as an older adult with IBD, or someone who faces high exposure, such as a health care worker, the potential protection of the vaccine greatly outweighs the uncertainty.
“However, for individuals who are at otherwise lower risk – for example, young and able to work from home – then waiting a few extra months for postmarketing surveillance studies in immunocompromised populations may be a reasonable approach, as long as these individuals are taking great care to avoid infection,” he said.
No waiting needed
Joel M. Gelfand, MD, MSCE, professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, feels that the newly approved vaccine should be safe for most of his patients.
“Patients with psoriatic disease should get the mRNA-based COVID-19 vaccine as soon as possible based on eligibility as determined by the CDC and local public health officials,” he said. “It is not a live vaccine, and therefore patients on biologics or other immune-modulating or immune-suppressing treatment can receive it.”
However, the impact of psoriasis treatment on immune response to the mRNA-based vaccines is not known. Dr. Gelfand noted that, extrapolating from the vaccine literature, there is some evidence that methotrexate reduces response to the influenza vaccine. “However, the clinical significance of this finding is not clear,” he said. “Since the mRNA vaccine needs to be taken twice, a few weeks apart, I do not recommend interrupting or delaying treatment for psoriatic disease while undergoing vaccination for COVID-19.”
Given the reports of allergic reactions, he added that it is advisable for patients with a history of life-threatening allergic reactions such as anaphylaxis or who have been advised to carry an epinephrine autoinjector, to talk with their health care provider to determine if COVID-19 vaccination is medically appropriate.
The National Psoriasis Foundation has issued guidance on COVID-19, explained Steven R. Feldman, MD, PhD, professor of dermatology, pathology, and social sciences & health policy at Wake Forest University, Winston-Salem, N.C., who is also a member of the committee that is working on those guidelines and keeping them up to date. “We are in the process of updating the guidelines with information on COVID vaccines,” he said.
He agreed that there are no contraindications for psoriasis patients to receive the vaccine, regardless of whether they are on immunosuppressive treatment, even though definitive data are lacking. “Fortunately, there’s a lot of good data coming out of Italy that patients with psoriasis on biologics do not appear to be at increased risk of getting COVID or of having worse outcomes from COVID,” he said.
Patients are going to ask about the vaccines, and when counseling them, clinicians should discuss the available data, the residual uncertainty, and patients’ concerns should be considered, Dr. Feldman explained. “There may be some concern that steroids and cyclosporine would reduce the effectiveness of vaccines, but there is no concern that any of the drugs would cause increased risk from nonlive vaccines.”
He added that there is evidence that “patients on biologics who receive nonlive vaccines do develop antibody responses and are immunized.”
Boosting efficacy
Even prior to making their announcement, the American College of Rheumatology had said that they would endorse the vaccine for all patients, explained rheumatologist Brett Smith, DO, from Blount Memorial Physicians Group and East Tennessee Children’s Hospital, Alcoa. “The vaccine is safe for all patients, but the problem may be that it’s not as effective,” he said. “But we don’t know that because it hasn’t been tested.”
With other vaccines, biologic medicines are held for 2 weeks before and afterwards, to get the best response. “But some patients don’t want to stop the medication,” Dr. Smith said. “They are afraid that their symptoms will return.”
As for counseling patients as to whether they should receive this vaccine, he explained that he typically doesn’t try to sway patients one way or another until they are really high risk. “When I counsel, it really depends on the individual situation. And for this vaccine, we have to be open to the fact that many people have already made up their mind.”
There are a lot of questions regarding the vaccine. One is the short time frame of development. “Vaccines typically take 6-10 years to come on the market, and this one is now available after a 3-month study,” Dr. Smith said. “Some have already decided that it’s too new for them.”
The process is also new, and patients need to understand that it doesn’t contain an active virus and “you can’t catch coronavirus from it.”
Dr. Smith also explained that, because the vaccine may be less effective in a person using biologic therapies, there is currently no information available on repeat vaccination. “These are all unanswered questions,” he said. “If the antibodies wane in a short time, can we be revaccinated and in what time frame? We just don’t know that yet.”
Marcelo Bonomi, MD, a medical oncologist from The Ohio State University Comprehensive Cancer Center, Columbus, explained that one way to ensure a more optimal response to the vaccine would be to wait until the patient has finished chemotherapy.* “The vaccine can be offered at that time, and in the meantime, they can take other steps to avoid infection,” he said. “If they are very immunosuppressed, it isn’t worth trying to give the vaccine.”
Cancer patients should be encouraged to stay as healthy as possible, and to wear masks and social distance. “It’s a comprehensive approach. Eat healthy, avoid alcohol and tobacco, and exercise. [These things] will help boost the immune system,” Dr. Bonomi said. “Family members should be encouraged to get vaccinated, which will help them avoid infection and exposing the patient.”
Jim Boonyaratanakornkit, MD, PhD, an infectious disease specialist who cares for cancer patients at the Fred Hutchinson Cancer Research Center, agreed. “Giving a vaccine right after a transplant is a futile endeavor,” he said. “We need to wait 6 months to have an immune response.”
He pointed out there may be a continuing higher number of cases, with high levels peaking in Washington in February and March. “Close friends and family should be vaccinated if possible,” he said, “which will help interrupt transmission.”
The vaccines are using new platforms that are totally different, and there is no clear data as to how long the antibodies will persist. “We know that they last for at least 4 months,” said Dr. Boonyaratanakornkit. “We don’t know what level of antibody will protect them from COVID-19 infection. Current studies are being conducted, but we don’t have that information for anyone yet.”
*Correction, 1/7/21: An earlier version of this article misattributed quotes from Dr. Marcelo Bonomi.
For the latest clinical guidance, education, research and physician resources about coronavirus, visit the AGA COVID-19 Resource Center at www.gastro.org/COVID.
Coronavirus vaccines have become a reality, as they are now being approved and authorized for use in a growing number of countries including the United States. The U.S. Food and Drug Administration has just issued emergency authorization for the use of the COVID-19 vaccine produced by Pfizer and BioNTech. Close behind is the vaccine developed by Moderna, which has also applied to the FDA for emergency authorization.
The efficacy of a two-dose administration of the vaccine has been pegged at 95.0%, and the FDA has said that the 95% credible interval for the vaccine efficacy was 90.3%-97.6%. But as with many initial clinical trials, whether for drugs or vaccines, not all populations were represented in the trial cohort, including individuals who are immunocompromised. At the current time, it is largely unknown how safe or effective the vaccine may be in this large population, many of whom are at high risk for serious COVID-19 complications.
At a special session held during the recent annual meeting of the American Society of Hematology, Anthony Fauci, MD, the nation’s leading infectious disease expert, said that individuals with compromised immune systems, whether because of chemotherapy or a bone marrow transplant, should plan to be vaccinated when the opportunity arises.
In response to a question from ASH President Stephanie J. Lee, MD, of the Fred Hutchinson Cancer Center, Seattle, Dr. Fauci emphasized that, despite being excluded from clinical trials, this population should get vaccinated. “I think we should recommend that they get vaccinated,” he said. “I mean, it is clear that, if you are on immunosuppressive agents, history tells us that you’re not going to have as robust a response as if you had an intact immune system that was not being compromised. But some degree of immunity is better than no degree of immunity.”
That does seem to be the consensus among experts who spoke in interviews: that as long as these are not live attenuated vaccines, they hold no specific risk to an immunocompromised patient, other than any factors specific to the individual that could be a contraindication.
“Patients, family members, friends, and work contacts should be encouraged to receive the vaccine,” said William Stohl, MD, PhD, chief of the division of rheumatology at the University of Southern California, Los Angeles. “Clinicians should advise patients to obtain the vaccine sooner rather than later.”
Kevin C. Wang, MD, PhD, of the department of dermatology at Stanford (Calif.) University, agreed. “I am 100% with Dr. Fauci. Everyone should get the vaccine, even if it may not be as effective,” he said. “I would treat it exactly like the flu vaccines that we recommend folks get every year.”
Dr. Wang noted that he couldn’t think of any contraindications unless the immunosuppressed patients have a history of severe allergic reactions to prior vaccinations. “But I would even say patients with history of cancer, upon recommendation of their oncologists, are likely to be suitable candidates for the vaccine,” he added. “I would say clinicians should approach counseling the same way they counsel patients for the flu vaccine, and as far as I know, there are no concerns for systemic drugs commonly used in dermatology patients.”
However, guidance has not yet been issued from either the FDA or the Centers for Disease Control and Prevention regarding the use of the vaccine in immunocompromised individuals. Given the lack of data, the FDA has said that “it will be something that providers will need to consider on an individual basis,” and that individuals should consult with physicians to weigh the potential benefits and potential risks.
The CDC’s Advisory Committee on Immunization Practices has said that clinicians need more guidance on whether to use the vaccine in pregnant or breastfeeding women, the immunocompromised, or those who have a history of allergies. The CDC itself has not yet released its formal guidance on vaccine use.
COVID-19 vaccines
Vaccines typically require years of research and testing before reaching the clinic, but this year researchers embarked on a global effort to develop safe and effective coronavirus vaccines in record time. Both the Pfizer/BioNTech and Moderna vaccines have only a few months of phase 3 clinical trial data, so much remains unknown about them, including their duration of effect and any long-term safety signals. In addition to excluding immunocompromised individuals, the clinical trials did not include children or pregnant women, so data are lacking for several population subgroups.
But these will not be the only vaccines available, as the pipeline is already becoming crowded. U.S. clinical trial data from a vaccine jointly being developed by Oxford-AstraZeneca, could potentially be ready, along with a request for FDA emergency use authorization, by late January 2021.
In addition, China and Russia have released vaccines, and there are currently 61 vaccines being investigated in clinical trials and at least 85 preclinical products under active investigation.
The vaccine candidates are using both conventional and novel mechanisms of action to elicit an immune response in patients. Conventional methods include attenuated inactivated (killed) virus and recombinant viral protein vaccines to develop immunity. Novel approaches include replication-deficient, adenovirus vector-based vaccines that contain the viral protein, and mRNA-based vaccines, such as the Pfizer and Moderna vaccines, that encode for a SARS-CoV-2 spike protein.
“The special vaccine concern for immunocompromised individuals is introduction of a live virus,” Dr. Stohl said. “Neither the Moderna nor Pfizer vaccines are live viruses, so there should be no special contraindication for such individuals.”
Live vaccine should be avoided in immunocompromised patients, and currently, live SARS-CoV-2 vaccines are only being developed in India and Turkey.
It is not unusual for vaccine trials to begin with cohorts that exclude participants with various health conditions, including those who are immunocompromised. These groups are generally then evaluated in phase 4 trials, or postmarketing surveillance. While the precise number of immunosuppressed adults in the United States is not known, the numbers are believed to be rising because of increased life expectancy among immunosuppressed adults as a result of advances in treatment and new and wider indications for therapies that can affect the immune system.
According to data from the 2013 National Health Interview Survey, an estimated 2.7% of U.S. adults are immunosuppressed. This population covers a broad array of health conditions and medical specialties; people living with inflammatory or autoimmune conditions, such as inflammatory rheumatic diseases (rheumatoid arthritis, axial spondyloarthritis, lupus); inflammatory bowel disease (Crohn’s disease and ulcerative colitis); psoriasis; multiple sclerosis; organ transplant recipients; patients undergoing chemotherapy; and life-long immunosuppression attributable to HIV infection.
As the vaccines begin to roll out and become available, how should clinicians advise their patients, in the absence of any clinical trial data?
Risk vs. benefit
Gilaad Kaplan, MD, MPH, a gastroenterologist and professor of medicine at the University of Calgary (Alta.), noted that the inflammatory bowel disease (IBD) community has dealt with tremendous anxiety during the pandemic because many are immunocompromised because of the medications they use to treat their disease.
“For example, many patients with IBD are on biologics like anti-TNF [tumor necrosis factor] therapies, which are also used in other immune-mediated inflammatory diseases such as rheumatoid arthritis,” he said. “Understandably, individuals with IBD on immunosuppressive medications are concerned about the risk of severe complications due to COVID-19.”
The entire IBD community, along with the world, celebrated the announcement that multiple vaccines are protective against SARS-CoV-2, he noted. “Vaccines offer the potential to reduce the spread of COVID-19, allowing society to revert back to normalcy,” Dr. Kaplan said. “Moreover, for vulnerable populations, including those who are immunocompromised, vaccines offer the potential to directly protect them from the morbidity and mortality associated with COVID-19.”
That said, even though the news of vaccines are extremely promising, some cautions must be raised regarding their use in immunocompromised populations, such as persons with IBD. “The current trials, to my knowledge, did not include immunocompromised individuals and thus, we can only extrapolate from what we know from other trials of different vaccines,” he explained. “We know from prior vaccines studies that the immune response following vaccination is less robust in those who are immunocompromised as compared to a healthy control population.”
Dr. Kaplan also pointed to recent reports of allergic reactions that have been reported in healthy individuals. “We don’t know whether side effects, like allergic reactions, may be different in unstudied populations,” he said. “Thus, the medical and scientific community should prioritize clinical studies of safety and effectiveness of COVID-19 vaccines in immunocompromised populations.”
So, what does this mean for an individual with an immune-mediated inflammatory disease like Crohn’s disease or ulcerative colitis who is immunocompromised? Dr. Kaplan explained that it is a balance between the potential harm of being infected with COVID-19 and the uncertainty of receiving a vaccine in an understudied population. For those who are highly susceptible to dying from COVID-19, such as an older adult with IBD, or someone who faces high exposure, such as a health care worker, the potential protection of the vaccine greatly outweighs the uncertainty.
“However, for individuals who are at otherwise lower risk – for example, young and able to work from home – then waiting a few extra months for postmarketing surveillance studies in immunocompromised populations may be a reasonable approach, as long as these individuals are taking great care to avoid infection,” he said.
No waiting needed
Joel M. Gelfand, MD, MSCE, professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, feels that the newly approved vaccine should be safe for most of his patients.
“Patients with psoriatic disease should get the mRNA-based COVID-19 vaccine as soon as possible based on eligibility as determined by the CDC and local public health officials,” he said. “It is not a live vaccine, and therefore patients on biologics or other immune-modulating or immune-suppressing treatment can receive it.”
However, the impact of psoriasis treatment on immune response to the mRNA-based vaccines is not known. Dr. Gelfand noted that, extrapolating from the vaccine literature, there is some evidence that methotrexate reduces response to the influenza vaccine. “However, the clinical significance of this finding is not clear,” he said. “Since the mRNA vaccine needs to be taken twice, a few weeks apart, I do not recommend interrupting or delaying treatment for psoriatic disease while undergoing vaccination for COVID-19.”
Given the reports of allergic reactions, he added that it is advisable for patients with a history of life-threatening allergic reactions such as anaphylaxis or who have been advised to carry an epinephrine autoinjector, to talk with their health care provider to determine if COVID-19 vaccination is medically appropriate.
The National Psoriasis Foundation has issued guidance on COVID-19, explained Steven R. Feldman, MD, PhD, professor of dermatology, pathology, and social sciences & health policy at Wake Forest University, Winston-Salem, N.C., who is also a member of the committee that is working on those guidelines and keeping them up to date. “We are in the process of updating the guidelines with information on COVID vaccines,” he said.
He agreed that there are no contraindications for psoriasis patients to receive the vaccine, regardless of whether they are on immunosuppressive treatment, even though definitive data are lacking. “Fortunately, there’s a lot of good data coming out of Italy that patients with psoriasis on biologics do not appear to be at increased risk of getting COVID or of having worse outcomes from COVID,” he said.
Patients are going to ask about the vaccines, and when counseling them, clinicians should discuss the available data, the residual uncertainty, and patients’ concerns should be considered, Dr. Feldman explained. “There may be some concern that steroids and cyclosporine would reduce the effectiveness of vaccines, but there is no concern that any of the drugs would cause increased risk from nonlive vaccines.”
He added that there is evidence that “patients on biologics who receive nonlive vaccines do develop antibody responses and are immunized.”
Boosting efficacy
Even prior to making their announcement, the American College of Rheumatology had said that they would endorse the vaccine for all patients, explained rheumatologist Brett Smith, DO, from Blount Memorial Physicians Group and East Tennessee Children’s Hospital, Alcoa. “The vaccine is safe for all patients, but the problem may be that it’s not as effective,” he said. “But we don’t know that because it hasn’t been tested.”
With other vaccines, biologic medicines are held for 2 weeks before and afterwards, to get the best response. “But some patients don’t want to stop the medication,” Dr. Smith said. “They are afraid that their symptoms will return.”
As for counseling patients as to whether they should receive this vaccine, he explained that he typically doesn’t try to sway patients one way or another until they are really high risk. “When I counsel, it really depends on the individual situation. And for this vaccine, we have to be open to the fact that many people have already made up their mind.”
There are a lot of questions regarding the vaccine. One is the short time frame of development. “Vaccines typically take 6-10 years to come on the market, and this one is now available after a 3-month study,” Dr. Smith said. “Some have already decided that it’s too new for them.”
The process is also new, and patients need to understand that it doesn’t contain an active virus and “you can’t catch coronavirus from it.”
Dr. Smith also explained that, because the vaccine may be less effective in a person using biologic therapies, there is currently no information available on repeat vaccination. “These are all unanswered questions,” he said. “If the antibodies wane in a short time, can we be revaccinated and in what time frame? We just don’t know that yet.”
Marcelo Bonomi, MD, a medical oncologist from The Ohio State University Comprehensive Cancer Center, Columbus, explained that one way to ensure a more optimal response to the vaccine would be to wait until the patient has finished chemotherapy.* “The vaccine can be offered at that time, and in the meantime, they can take other steps to avoid infection,” he said. “If they are very immunosuppressed, it isn’t worth trying to give the vaccine.”
Cancer patients should be encouraged to stay as healthy as possible, and to wear masks and social distance. “It’s a comprehensive approach. Eat healthy, avoid alcohol and tobacco, and exercise. [These things] will help boost the immune system,” Dr. Bonomi said. “Family members should be encouraged to get vaccinated, which will help them avoid infection and exposing the patient.”
Jim Boonyaratanakornkit, MD, PhD, an infectious disease specialist who cares for cancer patients at the Fred Hutchinson Cancer Research Center, agreed. “Giving a vaccine right after a transplant is a futile endeavor,” he said. “We need to wait 6 months to have an immune response.”
He pointed out there may be a continuing higher number of cases, with high levels peaking in Washington in February and March. “Close friends and family should be vaccinated if possible,” he said, “which will help interrupt transmission.”
The vaccines are using new platforms that are totally different, and there is no clear data as to how long the antibodies will persist. “We know that they last for at least 4 months,” said Dr. Boonyaratanakornkit. “We don’t know what level of antibody will protect them from COVID-19 infection. Current studies are being conducted, but we don’t have that information for anyone yet.”
*Correction, 1/7/21: An earlier version of this article misattributed quotes from Dr. Marcelo Bonomi.
For the latest clinical guidance, education, research and physician resources about coronavirus, visit the AGA COVID-19 Resource Center at www.gastro.org/COVID.
Patients with cancer a ‘high priority’ for COVID-19 vaccine, says AACR task force
“The available evidence supports the conclusion that patients with cancer, in particular with hematologic malignancies, should be considered among the high-risk groups for priority COVID-19 vaccination,” according to the AACR’s COVID-19 and Cancer Task Force.
A review of literature suggested that COVID-19 fatality rates for patients with cancer were double that of individuals without cancer, the team noted. The higher mortality rates still trended upward, even after adjusting for confounders including age, sex, and comorbidities, indicating that there is a greater risk for severe disease and COVID-19–related mortality.
The new AACR position paper was published online Dec. 19 in Cancer Discovery.
“We conclude that patients with an active cancer should be considered for priority access to COVID-19 vaccination, along other particularly vulnerable populations with risk factors for adverse outcomes with COVID-19,” the team wrote.
However, the authors noted that “it is unclear whether this recommendation should be applicable to patients with a past diagnosis of cancer, as cancer survivors can be considered having the same risk as other persons with matched age and other risk factors.
“Given that there are nearly 17 million people living with a history of cancer in the United States alone, it is critical to understand whether these individuals are at a higher risk to contract SARS-COV-2 and to experience severe outcomes from COVID-19,” they added.
Allocation of initial doses
There has already been much discussion on the allocation of the initial doses of COVID-19 vaccines that have become available in the United States. The Advisory Committee on Immunization Practices of the Centers for Disease Control and Prevention recommended that the first wave of vaccinations, described as phase 1a, should be administered to health care workers (about 21 million people) and residents of long-term care facilities (about 3 million).
The next priority group, phase 1b, should consist of frontline essential workers, a group of about 30 million, and adults aged 75 years or older, a group of about 21 million. When overlap between the groups is taken into account, phase 1b covers about 49 million people, according to the CDC.
Finally, phase 1c, the third priority group, would include adults aged 65-74 years (a group of about 32 million), adults aged 16-64 years with high-risk medical conditions (a group of about 110 million), and essential workers who did not qualify for inclusion in phase 1b (a group of about 57 million). With the overlap, Phase 1c would cover about 129 million people.
The AACR task force, led by Antoni Ribas, MD, PhD, of the Jonsson Comprehensive Cancer Center at the University of California, Los Angeles, noted in their position paper that their recommendation is consistent with ACIP’s guidelines. Those guidelines concluded that patients with cancer are at a higher risk for severe COVID-19, and should be one of the groups considered for early COVID-19 vaccination.
Questions remain
Approached for independent comment, Cardinale Smith, MD, PhD, chief quality officer for cancer services for the Mount Sinai Health System in New York, agreed with the AACR task force. “I share that they should be high priority,” she said, “But we don’t know that the efficacy will the same.”
Dr. Smith noted that the impact of cancer therapy on patient immune systems is more related to the type of treatment they’re receiving, and B- and T-cell responses. “But regardless, they should be getting the vaccine, and we just need to follow the guidelines.”
The AACR task force noted that information thus far is quite limited as to the effects of COVID-19 vaccination in patients with cancer. In the Pfizer-BioNTech BNT162b2 COVID vaccine trial, of 43,540 participants, only 3.7% were reported to have cancer. Other large COVID-19 vaccine trials will provide further follow-up information on the effectiveness of the vaccines in patients receiving different cancer treatments, they wrote, but for now, there is “currently not enough data to evaluate the interactions between active oncologic therapy with the ability to induce protective immunity” to COVID-19 with vaccination.
In a recent interview, Nora Disis, MD, a medical oncologist and director of both the Institute of Translational Health Sciences and the Cancer Vaccine Institute, University of Washington, Seattle, also discussed vaccinating cancer patients.
She pointed out that even though there are data suggesting that cancer patients are at higher risk, “they are a bit murky, in part because cancer patients are a heterogeneous group.”
“For example, there are data suggesting that lung and blood cancer patients fare worse,” said Dr. Disis, who is also editor in chief of JAMA Oncology. “There is also a suggestion that, like in the general population, COVID risk in cancer patients remains driven by comorbidities.”
She also pointed out the likelihood that individualized risk factors, including the type of cancer therapy, site of disease, and comorbidities, “will shape individual choices about vaccination among cancer patients.”
It is also reasonable to expect that patients with cancer will respond to the vaccines, even though historically some believed that they would be unable to mount an immune response. “Data on other viral vaccines have shown otherwise,” said Dr. Disis. “For example, there has been a long history of studies of flu vaccination in cancer patients, and in general, those vaccines confer protection.”
Several of the authors of the AACR position paper, including Dr. Ribas, reported relationships with industry as detailed in the paper. Dr. Smith has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
“The available evidence supports the conclusion that patients with cancer, in particular with hematologic malignancies, should be considered among the high-risk groups for priority COVID-19 vaccination,” according to the AACR’s COVID-19 and Cancer Task Force.
A review of literature suggested that COVID-19 fatality rates for patients with cancer were double that of individuals without cancer, the team noted. The higher mortality rates still trended upward, even after adjusting for confounders including age, sex, and comorbidities, indicating that there is a greater risk for severe disease and COVID-19–related mortality.
The new AACR position paper was published online Dec. 19 in Cancer Discovery.
“We conclude that patients with an active cancer should be considered for priority access to COVID-19 vaccination, along other particularly vulnerable populations with risk factors for adverse outcomes with COVID-19,” the team wrote.
However, the authors noted that “it is unclear whether this recommendation should be applicable to patients with a past diagnosis of cancer, as cancer survivors can be considered having the same risk as other persons with matched age and other risk factors.
“Given that there are nearly 17 million people living with a history of cancer in the United States alone, it is critical to understand whether these individuals are at a higher risk to contract SARS-COV-2 and to experience severe outcomes from COVID-19,” they added.
Allocation of initial doses
There has already been much discussion on the allocation of the initial doses of COVID-19 vaccines that have become available in the United States. The Advisory Committee on Immunization Practices of the Centers for Disease Control and Prevention recommended that the first wave of vaccinations, described as phase 1a, should be administered to health care workers (about 21 million people) and residents of long-term care facilities (about 3 million).
The next priority group, phase 1b, should consist of frontline essential workers, a group of about 30 million, and adults aged 75 years or older, a group of about 21 million. When overlap between the groups is taken into account, phase 1b covers about 49 million people, according to the CDC.
Finally, phase 1c, the third priority group, would include adults aged 65-74 years (a group of about 32 million), adults aged 16-64 years with high-risk medical conditions (a group of about 110 million), and essential workers who did not qualify for inclusion in phase 1b (a group of about 57 million). With the overlap, Phase 1c would cover about 129 million people.
The AACR task force, led by Antoni Ribas, MD, PhD, of the Jonsson Comprehensive Cancer Center at the University of California, Los Angeles, noted in their position paper that their recommendation is consistent with ACIP’s guidelines. Those guidelines concluded that patients with cancer are at a higher risk for severe COVID-19, and should be one of the groups considered for early COVID-19 vaccination.
Questions remain
Approached for independent comment, Cardinale Smith, MD, PhD, chief quality officer for cancer services for the Mount Sinai Health System in New York, agreed with the AACR task force. “I share that they should be high priority,” she said, “But we don’t know that the efficacy will the same.”
Dr. Smith noted that the impact of cancer therapy on patient immune systems is more related to the type of treatment they’re receiving, and B- and T-cell responses. “But regardless, they should be getting the vaccine, and we just need to follow the guidelines.”
The AACR task force noted that information thus far is quite limited as to the effects of COVID-19 vaccination in patients with cancer. In the Pfizer-BioNTech BNT162b2 COVID vaccine trial, of 43,540 participants, only 3.7% were reported to have cancer. Other large COVID-19 vaccine trials will provide further follow-up information on the effectiveness of the vaccines in patients receiving different cancer treatments, they wrote, but for now, there is “currently not enough data to evaluate the interactions between active oncologic therapy with the ability to induce protective immunity” to COVID-19 with vaccination.
In a recent interview, Nora Disis, MD, a medical oncologist and director of both the Institute of Translational Health Sciences and the Cancer Vaccine Institute, University of Washington, Seattle, also discussed vaccinating cancer patients.
She pointed out that even though there are data suggesting that cancer patients are at higher risk, “they are a bit murky, in part because cancer patients are a heterogeneous group.”
“For example, there are data suggesting that lung and blood cancer patients fare worse,” said Dr. Disis, who is also editor in chief of JAMA Oncology. “There is also a suggestion that, like in the general population, COVID risk in cancer patients remains driven by comorbidities.”
She also pointed out the likelihood that individualized risk factors, including the type of cancer therapy, site of disease, and comorbidities, “will shape individual choices about vaccination among cancer patients.”
It is also reasonable to expect that patients with cancer will respond to the vaccines, even though historically some believed that they would be unable to mount an immune response. “Data on other viral vaccines have shown otherwise,” said Dr. Disis. “For example, there has been a long history of studies of flu vaccination in cancer patients, and in general, those vaccines confer protection.”
Several of the authors of the AACR position paper, including Dr. Ribas, reported relationships with industry as detailed in the paper. Dr. Smith has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
“The available evidence supports the conclusion that patients with cancer, in particular with hematologic malignancies, should be considered among the high-risk groups for priority COVID-19 vaccination,” according to the AACR’s COVID-19 and Cancer Task Force.
A review of literature suggested that COVID-19 fatality rates for patients with cancer were double that of individuals without cancer, the team noted. The higher mortality rates still trended upward, even after adjusting for confounders including age, sex, and comorbidities, indicating that there is a greater risk for severe disease and COVID-19–related mortality.
The new AACR position paper was published online Dec. 19 in Cancer Discovery.
“We conclude that patients with an active cancer should be considered for priority access to COVID-19 vaccination, along other particularly vulnerable populations with risk factors for adverse outcomes with COVID-19,” the team wrote.
However, the authors noted that “it is unclear whether this recommendation should be applicable to patients with a past diagnosis of cancer, as cancer survivors can be considered having the same risk as other persons with matched age and other risk factors.
“Given that there are nearly 17 million people living with a history of cancer in the United States alone, it is critical to understand whether these individuals are at a higher risk to contract SARS-COV-2 and to experience severe outcomes from COVID-19,” they added.
Allocation of initial doses
There has already been much discussion on the allocation of the initial doses of COVID-19 vaccines that have become available in the United States. The Advisory Committee on Immunization Practices of the Centers for Disease Control and Prevention recommended that the first wave of vaccinations, described as phase 1a, should be administered to health care workers (about 21 million people) and residents of long-term care facilities (about 3 million).
The next priority group, phase 1b, should consist of frontline essential workers, a group of about 30 million, and adults aged 75 years or older, a group of about 21 million. When overlap between the groups is taken into account, phase 1b covers about 49 million people, according to the CDC.
Finally, phase 1c, the third priority group, would include adults aged 65-74 years (a group of about 32 million), adults aged 16-64 years with high-risk medical conditions (a group of about 110 million), and essential workers who did not qualify for inclusion in phase 1b (a group of about 57 million). With the overlap, Phase 1c would cover about 129 million people.
The AACR task force, led by Antoni Ribas, MD, PhD, of the Jonsson Comprehensive Cancer Center at the University of California, Los Angeles, noted in their position paper that their recommendation is consistent with ACIP’s guidelines. Those guidelines concluded that patients with cancer are at a higher risk for severe COVID-19, and should be one of the groups considered for early COVID-19 vaccination.
Questions remain
Approached for independent comment, Cardinale Smith, MD, PhD, chief quality officer for cancer services for the Mount Sinai Health System in New York, agreed with the AACR task force. “I share that they should be high priority,” she said, “But we don’t know that the efficacy will the same.”
Dr. Smith noted that the impact of cancer therapy on patient immune systems is more related to the type of treatment they’re receiving, and B- and T-cell responses. “But regardless, they should be getting the vaccine, and we just need to follow the guidelines.”
The AACR task force noted that information thus far is quite limited as to the effects of COVID-19 vaccination in patients with cancer. In the Pfizer-BioNTech BNT162b2 COVID vaccine trial, of 43,540 participants, only 3.7% were reported to have cancer. Other large COVID-19 vaccine trials will provide further follow-up information on the effectiveness of the vaccines in patients receiving different cancer treatments, they wrote, but for now, there is “currently not enough data to evaluate the interactions between active oncologic therapy with the ability to induce protective immunity” to COVID-19 with vaccination.
In a recent interview, Nora Disis, MD, a medical oncologist and director of both the Institute of Translational Health Sciences and the Cancer Vaccine Institute, University of Washington, Seattle, also discussed vaccinating cancer patients.
She pointed out that even though there are data suggesting that cancer patients are at higher risk, “they are a bit murky, in part because cancer patients are a heterogeneous group.”
“For example, there are data suggesting that lung and blood cancer patients fare worse,” said Dr. Disis, who is also editor in chief of JAMA Oncology. “There is also a suggestion that, like in the general population, COVID risk in cancer patients remains driven by comorbidities.”
She also pointed out the likelihood that individualized risk factors, including the type of cancer therapy, site of disease, and comorbidities, “will shape individual choices about vaccination among cancer patients.”
It is also reasonable to expect that patients with cancer will respond to the vaccines, even though historically some believed that they would be unable to mount an immune response. “Data on other viral vaccines have shown otherwise,” said Dr. Disis. “For example, there has been a long history of studies of flu vaccination in cancer patients, and in general, those vaccines confer protection.”
Several of the authors of the AACR position paper, including Dr. Ribas, reported relationships with industry as detailed in the paper. Dr. Smith has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
2.1 Million COVID Vaccine Doses Given in U.S.
The U.S. has distributed more than 11.4 million doses of the Pfizer and Moderna COVID-19 vaccines, and more than 2.1 million of those had been given to people as of December 28, according to the CDC.
The CDC’s COVID Data Tracker showed the updated numbers as of 9 a.m. on that day. The distribution total is based on the CDC’s Vaccine Tracking System, and the administered total is based on reports from state and local public health departments, as well as updates from five federal agencies: the Bureau of Prisons, Veterans Administration, Department of Defense, Department of State, and Indian Health Services.
Health care providers report to public health agencies up to 72 hours after the vaccine is given, and public health agencies report to the CDC after that, so there may be a lag in the data. The CDC’s numbers will be updated on Mondays, Wednesdays, and Fridays.
“A large difference between the number of doses distributed and the number of doses administered is expected at this point in the COVID vaccination program due to several factors,” the CDC says.
Delays could occur due to the reporting of doses given, how states and local vaccine sites are managing vaccines, and the pending launch of vaccination through the federal Pharmacy Partnership for Long-Term Care Program.
“Numbers reported on other websites may differ from what is posted on CDC’s website because CDC’s overall numbers are validated through a data submission process with each jurisdiction,” the CDC says.
On Dec. 26, the agency’s tally showed that 9.5 million doses had been distributed and 1.9 million had been given, according to Reuters.
Public health officials and health care workers have begun to voice their concerns about the delay in giving the vaccines.
“We certainly are not at the numbers that we wanted to be at the end of December,” Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, told CNNDec. 29.
Operation Warp Speed had planned for 20 million people to be vaccinated by the end of the year. Fauci said he hopes that number will be achieved next month.
“I believe that as we get into January, we are going to see an increase in the momentum,” he said.
Shipment delays have affected other priority groups as well. The New York Police Department anticipated a rollout Dec. 29, but it’s now been delayed since the department hasn’t received enough Moderna doses to start giving the shots, according to the New York Daily News.
“We’ve made numerous attempts to get updated information, and when we get further word on its availability, we will immediately keep our members appraised of the new date and the method of distribution,” Paul DiGiacomo, president of the Detectives’ Endowment Association, wrote in a memo to members on Dec. 28.
“Every detective squad has been crushed with [COVID-19],” he told the newspaper. “Within the last couple of weeks, we’ve had at least two detectives hospitalized.”
President-elect Joe Biden will receive a briefing from his COVID-19 advisory team, provide a general update on the pandemic, and describe his own plan for vaccinating people quickly during an address Dec. 29, a transition official told Axios. Biden has pledged to administer 100 million vaccine doses in his first 100 days in office.
A version of this article originally appeared on WebMd.
The U.S. has distributed more than 11.4 million doses of the Pfizer and Moderna COVID-19 vaccines, and more than 2.1 million of those had been given to people as of December 28, according to the CDC.
The CDC’s COVID Data Tracker showed the updated numbers as of 9 a.m. on that day. The distribution total is based on the CDC’s Vaccine Tracking System, and the administered total is based on reports from state and local public health departments, as well as updates from five federal agencies: the Bureau of Prisons, Veterans Administration, Department of Defense, Department of State, and Indian Health Services.
Health care providers report to public health agencies up to 72 hours after the vaccine is given, and public health agencies report to the CDC after that, so there may be a lag in the data. The CDC’s numbers will be updated on Mondays, Wednesdays, and Fridays.
“A large difference between the number of doses distributed and the number of doses administered is expected at this point in the COVID vaccination program due to several factors,” the CDC says.
Delays could occur due to the reporting of doses given, how states and local vaccine sites are managing vaccines, and the pending launch of vaccination through the federal Pharmacy Partnership for Long-Term Care Program.
“Numbers reported on other websites may differ from what is posted on CDC’s website because CDC’s overall numbers are validated through a data submission process with each jurisdiction,” the CDC says.
On Dec. 26, the agency’s tally showed that 9.5 million doses had been distributed and 1.9 million had been given, according to Reuters.
Public health officials and health care workers have begun to voice their concerns about the delay in giving the vaccines.
“We certainly are not at the numbers that we wanted to be at the end of December,” Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, told CNNDec. 29.
Operation Warp Speed had planned for 20 million people to be vaccinated by the end of the year. Fauci said he hopes that number will be achieved next month.
“I believe that as we get into January, we are going to see an increase in the momentum,” he said.
Shipment delays have affected other priority groups as well. The New York Police Department anticipated a rollout Dec. 29, but it’s now been delayed since the department hasn’t received enough Moderna doses to start giving the shots, according to the New York Daily News.
“We’ve made numerous attempts to get updated information, and when we get further word on its availability, we will immediately keep our members appraised of the new date and the method of distribution,” Paul DiGiacomo, president of the Detectives’ Endowment Association, wrote in a memo to members on Dec. 28.
“Every detective squad has been crushed with [COVID-19],” he told the newspaper. “Within the last couple of weeks, we’ve had at least two detectives hospitalized.”
President-elect Joe Biden will receive a briefing from his COVID-19 advisory team, provide a general update on the pandemic, and describe his own plan for vaccinating people quickly during an address Dec. 29, a transition official told Axios. Biden has pledged to administer 100 million vaccine doses in his first 100 days in office.
A version of this article originally appeared on WebMd.
The U.S. has distributed more than 11.4 million doses of the Pfizer and Moderna COVID-19 vaccines, and more than 2.1 million of those had been given to people as of December 28, according to the CDC.
The CDC’s COVID Data Tracker showed the updated numbers as of 9 a.m. on that day. The distribution total is based on the CDC’s Vaccine Tracking System, and the administered total is based on reports from state and local public health departments, as well as updates from five federal agencies: the Bureau of Prisons, Veterans Administration, Department of Defense, Department of State, and Indian Health Services.
Health care providers report to public health agencies up to 72 hours after the vaccine is given, and public health agencies report to the CDC after that, so there may be a lag in the data. The CDC’s numbers will be updated on Mondays, Wednesdays, and Fridays.
“A large difference between the number of doses distributed and the number of doses administered is expected at this point in the COVID vaccination program due to several factors,” the CDC says.
Delays could occur due to the reporting of doses given, how states and local vaccine sites are managing vaccines, and the pending launch of vaccination through the federal Pharmacy Partnership for Long-Term Care Program.
“Numbers reported on other websites may differ from what is posted on CDC’s website because CDC’s overall numbers are validated through a data submission process with each jurisdiction,” the CDC says.
On Dec. 26, the agency’s tally showed that 9.5 million doses had been distributed and 1.9 million had been given, according to Reuters.
Public health officials and health care workers have begun to voice their concerns about the delay in giving the vaccines.
“We certainly are not at the numbers that we wanted to be at the end of December,” Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, told CNNDec. 29.
Operation Warp Speed had planned for 20 million people to be vaccinated by the end of the year. Fauci said he hopes that number will be achieved next month.
“I believe that as we get into January, we are going to see an increase in the momentum,” he said.
Shipment delays have affected other priority groups as well. The New York Police Department anticipated a rollout Dec. 29, but it’s now been delayed since the department hasn’t received enough Moderna doses to start giving the shots, according to the New York Daily News.
“We’ve made numerous attempts to get updated information, and when we get further word on its availability, we will immediately keep our members appraised of the new date and the method of distribution,” Paul DiGiacomo, president of the Detectives’ Endowment Association, wrote in a memo to members on Dec. 28.
“Every detective squad has been crushed with [COVID-19],” he told the newspaper. “Within the last couple of weeks, we’ve had at least two detectives hospitalized.”
President-elect Joe Biden will receive a briefing from his COVID-19 advisory team, provide a general update on the pandemic, and describe his own plan for vaccinating people quickly during an address Dec. 29, a transition official told Axios. Biden has pledged to administer 100 million vaccine doses in his first 100 days in office.
A version of this article originally appeared on WebMd.
CDC issues COVID-19 vaccine guidance for underlying conditions
The Centers for Disease Control and Prevention has issued updated guidance for people with underlying medical conditions who are considering getting the coronavirus vaccine.
“Adults of any age with certain underlying medical conditions are at increased risk for severe illness from the virus that causes COVID-19,” the CDC said in the guidance, posted on Dec. 26. “mRNA COVID-19 vaccines may be administered to people with underlying medical conditions provided they have not had a severe allergic reaction to any of the ingredients in the vaccine.”
Both the Pfizer and Moderna vaccines use mRNA, or messenger RNA.
The CDC guidance had specific information for people with HIV, weakened immune systems, and autoimmune conditions such as Guillain-Barré syndrome (GBS) and Bell’s palsy who are thinking of getting the vaccine.
People with HIV and weakened immune systems “may receive a COVID-19 vaccine. However, they should be aware of the limited safety data,” the CDC said.
There’s no information available yet about the safety of the vaccines for people with weakened immune systems. People with HIV were included in clinical trials, but “safety data specific to this group are not yet available at this time,” the CDC said.
Cases of Bell’s palsy, a temporary facial paralysis, were reported in people receiving the Pfizer and Moderna vaccines in clinical trials, the Food and Drug Administration said Dec. 17.
But the new CDC guidance said that the FDA “does not consider these to be above the rate expected in the general population. They have not concluded these cases were caused by vaccination. Therefore, persons who have previously had Bell’s palsy may receive an mRNA COVID-19 vaccine.”
Researchers have determined the vaccines are safe for people with GBS, a rare autoimmune disorder in which the body’s immune system attacks nerves just as they leave the spinal cord, the CDC said.
“To date, no cases of GBS have been reported following vaccination among participants in the mRNA COVID-19 vaccine clinical trials,” the CDC guidance said. “With few exceptions, the independent Advisory Committee on Immunization Practices general best practice guidelines for immunization do not include a history of GBS as a precaution to vaccination with other vaccines.”
For months, the CDC and other health authorities have said that people with certain medical conditions are at an increased risk of developing severe cases of COVID-19.
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention has issued updated guidance for people with underlying medical conditions who are considering getting the coronavirus vaccine.
“Adults of any age with certain underlying medical conditions are at increased risk for severe illness from the virus that causes COVID-19,” the CDC said in the guidance, posted on Dec. 26. “mRNA COVID-19 vaccines may be administered to people with underlying medical conditions provided they have not had a severe allergic reaction to any of the ingredients in the vaccine.”
Both the Pfizer and Moderna vaccines use mRNA, or messenger RNA.
The CDC guidance had specific information for people with HIV, weakened immune systems, and autoimmune conditions such as Guillain-Barré syndrome (GBS) and Bell’s palsy who are thinking of getting the vaccine.
People with HIV and weakened immune systems “may receive a COVID-19 vaccine. However, they should be aware of the limited safety data,” the CDC said.
There’s no information available yet about the safety of the vaccines for people with weakened immune systems. People with HIV were included in clinical trials, but “safety data specific to this group are not yet available at this time,” the CDC said.
Cases of Bell’s palsy, a temporary facial paralysis, were reported in people receiving the Pfizer and Moderna vaccines in clinical trials, the Food and Drug Administration said Dec. 17.
But the new CDC guidance said that the FDA “does not consider these to be above the rate expected in the general population. They have not concluded these cases were caused by vaccination. Therefore, persons who have previously had Bell’s palsy may receive an mRNA COVID-19 vaccine.”
Researchers have determined the vaccines are safe for people with GBS, a rare autoimmune disorder in which the body’s immune system attacks nerves just as they leave the spinal cord, the CDC said.
“To date, no cases of GBS have been reported following vaccination among participants in the mRNA COVID-19 vaccine clinical trials,” the CDC guidance said. “With few exceptions, the independent Advisory Committee on Immunization Practices general best practice guidelines for immunization do not include a history of GBS as a precaution to vaccination with other vaccines.”
For months, the CDC and other health authorities have said that people with certain medical conditions are at an increased risk of developing severe cases of COVID-19.
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention has issued updated guidance for people with underlying medical conditions who are considering getting the coronavirus vaccine.
“Adults of any age with certain underlying medical conditions are at increased risk for severe illness from the virus that causes COVID-19,” the CDC said in the guidance, posted on Dec. 26. “mRNA COVID-19 vaccines may be administered to people with underlying medical conditions provided they have not had a severe allergic reaction to any of the ingredients in the vaccine.”
Both the Pfizer and Moderna vaccines use mRNA, or messenger RNA.
The CDC guidance had specific information for people with HIV, weakened immune systems, and autoimmune conditions such as Guillain-Barré syndrome (GBS) and Bell’s palsy who are thinking of getting the vaccine.
People with HIV and weakened immune systems “may receive a COVID-19 vaccine. However, they should be aware of the limited safety data,” the CDC said.
There’s no information available yet about the safety of the vaccines for people with weakened immune systems. People with HIV were included in clinical trials, but “safety data specific to this group are not yet available at this time,” the CDC said.
Cases of Bell’s palsy, a temporary facial paralysis, were reported in people receiving the Pfizer and Moderna vaccines in clinical trials, the Food and Drug Administration said Dec. 17.
But the new CDC guidance said that the FDA “does not consider these to be above the rate expected in the general population. They have not concluded these cases were caused by vaccination. Therefore, persons who have previously had Bell’s palsy may receive an mRNA COVID-19 vaccine.”
Researchers have determined the vaccines are safe for people with GBS, a rare autoimmune disorder in which the body’s immune system attacks nerves just as they leave the spinal cord, the CDC said.
“To date, no cases of GBS have been reported following vaccination among participants in the mRNA COVID-19 vaccine clinical trials,” the CDC guidance said. “With few exceptions, the independent Advisory Committee on Immunization Practices general best practice guidelines for immunization do not include a history of GBS as a precaution to vaccination with other vaccines.”
For months, the CDC and other health authorities have said that people with certain medical conditions are at an increased risk of developing severe cases of COVID-19.
A version of this article first appeared on Medscape.com.
Reducing COVID-19 opioid deaths
Editor's Note: Due to updated statistics from the CDC, the online version of this article has been modified from the version that appears in the printed edition of the January 2021 issue of Current Psychiatry.
Individuals with mental health and substance use disorders (SUDs) are particularly susceptible to negative effects of the coronavirus disease 2019 (COVID-19) pandemic. The collision of the COVID-19 pandemic and the drug overdose epidemic has highlighted the urgent need for physicians, policymakers, and health care professionals to optimize care for individuals with SUDs because they may be particularly vulnerable to the effects of the virus due to compromised respiratory and immune function, and poor social support.1 In this commentary, we highlight the challenges of the drug overdose epidemic, and recommend strategies to mitigate the impact of the COVID-19 pandemic among patients with SUDs.
A crisis exacerbated by COVID-19
The current drug overdose epidemic has become an American public health nightmare. According to preliminary data released by the CDC on December 17, 2020, there were more than 81,000 drug overdose deaths in the United States in the 12 months ending May 2020.2,3 This is the highest number of overdose deaths ever recorded in a 12-month period. The CDC also noted that while overdose deaths were already increasing in the months preceding the COVID-19 pandemic, the latest numbers suggest an acceleration of overdose deaths during the pandemic.
What is causing this significant loss of life? Prescription opioids and illegal opioids such as heroin and illicitly manufactured fentanyl are the main agents associated with overdose deaths. These opioids were responsible for 61% (28,647) of drug overdose deaths in the United States in 2014.4 In 2015, the opioid overdose death rate increased by 15.6%.5
The increase in the number of opioid overdose deaths in part coincides with a sharp increase in the availability and use of heroin. Heroin overdose deaths have more than tripled since 2010, but heroin is not the only opiate involved. Fentanyl, a synthetic, short-acting opioid that is approved for managing pain in patients with advanced cancers, is 50 times more potent than heroin. The abuse of prescribed fentanyl has been accelerating over the past decade, as is the use of illicitly produced fentanyl. Evidence from US Drug Enforcement Administration (DEA) seizure records shows heroin is being adulterated with illicit fentanyl to enhance the potency of the heroin.6,7 Mixing illicit fentanyl with heroin may be contributing to the recent increase in heroin overdose fatalities. According to the CDC, overdose deaths related to synthetic opioids increased 38.4% from the 12-month period leading up to June 2019 compared with the 12-month period leading up to May 2020.2,3 Postmortem studies of individuals who died from a heroin overdose have frequently found the presence of fentanyl along with heroin.8 Overdose deaths involving heroin may be occurring because individuals may be unknowingly using heroin adulterated with fentanyl.9 In addition, carfentanil, a powerful new synthetic fentanyl, has been recently identified in heroin mixtures. Carfentanil is 10,000 times stronger than morphine. Even in miniscule amounts, carfentanil can suppress breathing to the degree that multiple doses of naloxone are needed to restore respirations.
Initial studies indicate that the COVID-19 pandemic has been exacerbating this situation. Wainwright et al10 conducted an analysis of urine drug test results of patients with SUDs from 4 months before and 4 months after COVID-19 was declared a national emergency on March 13, 2020. Compared with before COVID-19, the proportion of specimens testing positive since COVID-19 increased from 3.80% to 7.32% for fentanyl and from 1.29% to 2.09% for heroin.10
A similar drug testing study found that during the pandemic, the proportion of positive results (positivity) increased by 35% for non-prescribed fentanyl and 44% for heroin.11 Positivity for non-prescribed fentanyl increased significantly among patients who tested positive for other drugs, including by 89% for amphetamines; 48% for benzodiazepines; 34% for cocaine; and 39% for opiates (P < .1 for all).11
In a review of electronic medical records, Ochalek et al12 found that the number of nonfatal opioid overdoses in an emergency department in Virginia increased from 102 in March-June 2019 to 227 in March-June 2020. In an issue brief published on October 31, 2020, the American Medical Association reported increase in opioid and other drug-related overdoses in more than 40 states during the COVID-19 pandemic.13
Continue to: Strategies for intervention...
Strategies for intervention
A multi-dimensional approach is needed to protect the public from this growing opioid overdose epidemic. To address this challenging task, we recommend several strategies:
Enhance access to virtual treatment
Even when in-person treatment cannot take place due to COVID-19-related restrictions, it is vital that services are accessible to patients with SUDs during this pandemic. Examples of virtual treatment include:
- Telehealth for medication-assisted treatment (MAT) using buprenorphine (recently updated guidance from the US DEA and Substance Abuse and Mental Health Services Administration [SAMHSA] allows this method of prescribing)
- Teletherapy to prevent relapse
- Remote drug screens by sending saliva or urine kits to patients' homes, visiting patients to collect fluid samples, or asking patients to come to a "drive-through" facility to provide samples
- Virtual (online) Alcoholics Anonymous, Narcotics Anonymous, SMART Recovery, and similar meetings to provide support in the absence of in-person meetings.
The American Society of Addiction Medicine (ASAM) offers guidance to treatment programs to focus on infection control and mitigation. The Table14 summarizes the ASAM recommendations for office-based opioid treatment during COVID-19.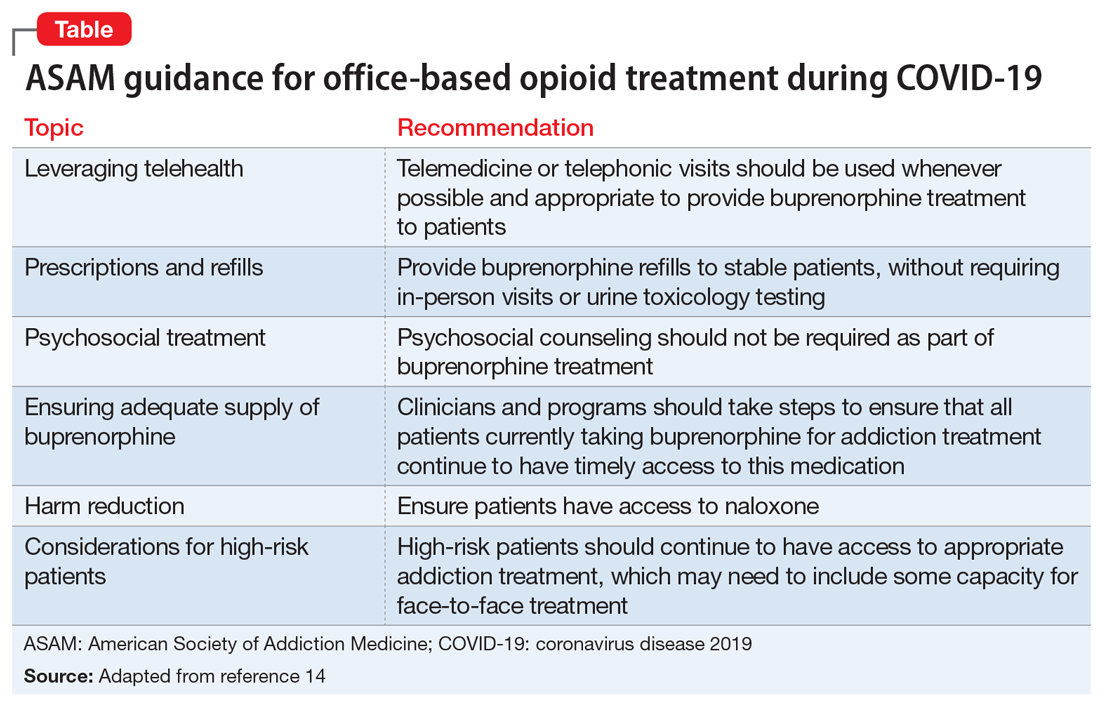
Expand access to treatment
This includes access to MAT (such as buprenorphine/naloxone, methadone, naltrexone, and depot naltrexone) and, equally important, to psychosocial treatment, counseling, and/or recovery services. Recent legislative changes have increased the number of patients that a qualified physician can treat with buprenorphine/naloxone from 100 to 275, and allowed physician extenders to prescribe buprenorphine/naloxone in office-based settings. A recent population-based, retrospective Canadian study showed that opioid agonist treatment decreased the risk of mortality among opioid users, and the protective effects of this treatment increased as fentanyl and other synthetic opioids became common in the illicit drug supply.15 However, because of the shortage of psychiatrists and addiction medicine specialists in several regions of the United States, access to treatment is extremely limited and often inadequate. This constitutes a major public health crisis and contributes to our inability to intervene effectively in the opioid epidemic. Telepsychiatry programs can bring needed services to underserved areas, but they need additional support and development. Further, involving other specialties is paramount for treating this epidemic. Integrating MAT in primary care settings can improve access to treatment. Harm-reduction approaches, such as syringe exchange programs, can play an important role in reducing the adverse consequences associated with heroin use and establish health care relationships with at-risk individuals. Syringe exchange programs can also reduce the rate of infections associated with IV drug use, such as human immunodeficiency virus and hepatitis C virus.
Continue to: Increase education on naloxone...
Increase education on naloxone
Naloxone is a safe and effective opioid antagonist used to treat opioid overdoses. Timely access to naloxone is of the essence when treating opioid-related overdoses. Many states have enacted laws allowing health care professionals, law enforcement officers, and patients and relatives to obtain naloxone without a physician's prescription. It appears this approach may be yielding results. For example, the North Carolina Harm Reduction Coalition distributed >101,000 free overdose rescue kits that included naloxone and recorded 13,392 confirmed cases of overdose rescue with naloxone from 2013 to 2019.16
Divert patients with SUDs from the criminal justice system to treatment
We need to develop programs to divert patients with SUDs from the criminal justice system, which is focused on punishment, to interventions that focus on treatment. Data indicates high recidivism rates for incarcerated individuals with SUDs who do not have access to treatment after they are released. Recognizing this, communities are developing programs that divert low-level offenders from the criminal justice system into treatment. For instance, in Seattle, the Law Enforcement Assisted Diversion is a pilot program developed to divert low-level drug and prostitution offenders into community-based treatment and support services. This helps provide housing, health care, job training, treatment, and mental health support. Innovative programs are needed to provide SUD treatment in the rehabilitation programs of correctional facilities and ensure case managers and discharge planners can transition participants to community treatment programs upon their release.
Develop early identification and prevention programs
These programs should focus on individuals at high risk, such as patients with comorbid SUDs and psychiatric disorders, those with chronic pain, and at-risk children whose parents abuse opiates. Traditional addiction treatment programs typically do not address patients with complex conditions or special populations, such as adolescents or pregnant women with substance use issues. Evidence-based approaches such as Screening, Brief Intervention, and Referral to Treatment (SBIRT), Integrated Dual Diagnosis Treatment (IDDT), and prevention approaches that target students in middle schools and high schools need to be more widely available.
Improve education on opioid prescribing
Responsible opioid prescribing for clinicians should include education about the regular use of prescription drug monitoring programs, urine drug screening, avoiding co-prescription of opioids with sedative-hypnotic medications, and better linkage with addiction treatment.
Treat comorbid psychiatric conditions
It is critical to both identify and effectively treat underlying affective, anxiety, and psychotic disorders in patients with SUDs. Anxiety, depression, and emotional dysregulation often contribute to worsening substance abuse, abuse of prescription drugs, diversion of prescribed drugs, and an increased risk of overdoses and suicides. Effective treatment of comorbid psychiatric conditions also may reduce relapses.
Increase research on causes and treatments
Through research, we must expand our knowledge to better understand the factors that contribute to this epidemic and develop better treatments. These efforts may allow for the development of prevention mechanisms. For example, a recent study found that the continued use of opioid medications after an overdose was associated with a high risk of a repeated overdosecall out material?.17 At the end of a 2-year observation, 17% (confidence interval [CI]: 14% to 20%) of patients receiving a high daily dosage of a prescribed opioid had a repeat overdose compared with 15% (CI: 10% to 21%) of those receiving a moderate dosage, 9% (CI: 6% to 14%) of those receiving a low dosage, and 8% (CI: 6% to 11%) of those receiving no opioids.17 Of the patients who overdosed on prescribed opiates, 30% switched to a new prescriber after their overdose, many of whom may not have been aware of the previous overdose. From a public health perspective, it would make sense for prescribers to know of prior opioid and/or benzodiazepine overdoses. This could be reported by emergency department clinicians, law enforcement, and hospitals into a prescription drug monitoring program, which is readily available to prescribers in most states.
Acknowledgment
The authors thank Scott Proescholdbell, MPH, Injury and Violence Prevention Branch, Chronic Disease and Injury Section, Division of Public Health, North Carolina Department of Health and Human Services, for his assistance.
Bottom Line
The collision of the coronavirus disease 2019 pandemic and the drug overdose epidemic has highlighted the urgent need for health care professionals to optimize care for individuals with substance use disorders. Suggested interventions include enhancing access to medication-assisted treatment and virtual treatment, improving education about naloxone and safe opioid prescribing practices, and diverting at-risk patients from the criminal justice system to interventions that focus on treatment.
1. Volkow ND. Collision of the COVID-19 and addiction epidemics. Ann Intern Med. 2020;173(1):61-62.
2.Centers for Disease Control and Prevention. Overdose deaths accelerating during COVID-19. Accessed December 23, 2020. https://www.cdc.gov/media/releases/2020/p1218-overdose-deaths-covid-19.html
3.Centers for Disease Control and Prevention. National Center for Health Statistics Vital Statistics Rapid Release. Provisional drug overdose death counts. Accessed December 30, 2020. https://www.cdc.gov/nchs/nvss/vsrr/drug-overdose-data.htm
4.Rudd RA, Aleshire N, Zibbell JE, et al. Increases in drug and opioid overdose deaths -- United States, 2000-2014. MMWR Morb Mortal Wkly Rep. 2016;64(50-51):1378-1382.
5.Rudd RA, Seth P, David F, et al. Increases in drug and opioid-involved overdose deaths -- United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2016;65(50-51):1445-1452.
6.US Drug Enforcement Administration. DEA issues nationwide alert on fentanyl as threat to health and public safety. Published March 19, 2015. Accessed October 28, 2020. http://www.dea.gov/divisions/hq/2015/hq031815.shtml
7.Gladden RM, Martinez P, Seth P. Fentanyl law enforcement submissions and increases in synthetic opioid-involved overdose deaths - 27 states, 2013-2014. MMWR Morb Mortal Wkly Rep. 2016;65(33):837-843.
8.Algren DA, Monteilh CP, Punja M, et al. Fentanyl-associated fatalities among illicit drug users in Wayne County, Michigan (July 2005-May 2006). J Med Toxicol. 2013;9(1):106-115.
9.Centers for Disease Control and Prevention. Increases in fentanyl drug confiscations and fentanyl-related overdose fatalities. HAN Health Advisory. Published October 26, 2015. Accessed October 28, 2020. http://emergency.cdc.gov/han/han00384.asp
10.Wainwright JJ, Mikre M, Whitley P, et al. Analysis of drug test results before and after the us declaration of a national emergency concerning the COVID-19 outbreak. JAMA. 2020;324(16):1674-1677.
11.Niles JK, Gudin J, Radliff J, et al. The opioid epidemic within the COVID-19 pandemic: drug testing in 2020 [published online October 8, 2020]. Population Health Management. doi: 10.1089/pop.2020.0230
12.Ochalek TA, Cumpston KL, Wills BK, et al. Nonfatal opioid overdoses at an urban emergency department during the COVID-19 pandemic. JAMA. 2020;324(16):1673-1674.
13.American Medical Association. Issue brief: reports of increases in opioid- and other drug-related overdose and other concerns during COVID pandemic. Published October 31, 2020. Accessed November 9, 2020. https://www.ama-assn.org/system/files/2020-11/issue-brief-increases-in-opioid-related-overdose.pdf
14.American Society of Addiction Medicine. Caring for patients during the COVID-19 pandemic: ASAM COVID-19 Task Force recommendations. Accessed October 30, 2020. https://www.asam.org/docs/default-source/covid-19/medication-formulation-and-dosage-guidance-(1).pdf
15.Pearce LA, Min JE, Piske M, et al. Opioid agonist treatment and risk of mortality during opioid overdose public health emergency: population based retrospective cohort study. BMJ. 2020;368:m772. doi: 10.1136/bmj.m772
16.North Carolina Harm Reduction Coalition. NCHRC'S community-based overdose prevention project. Accessed March 29, 2020. http://www.nchrc.org/programs-and-services
17.Larochelle MR, Liebschutz JM, Zhang F, et al. Opioid prescribing after nonfatal overdose and association with repeated overdose: a cohort study. Ann Intern Med. 2016;164(1):1-9.
Editor's Note: Due to updated statistics from the CDC, the online version of this article has been modified from the version that appears in the printed edition of the January 2021 issue of Current Psychiatry.
Individuals with mental health and substance use disorders (SUDs) are particularly susceptible to negative effects of the coronavirus disease 2019 (COVID-19) pandemic. The collision of the COVID-19 pandemic and the drug overdose epidemic has highlighted the urgent need for physicians, policymakers, and health care professionals to optimize care for individuals with SUDs because they may be particularly vulnerable to the effects of the virus due to compromised respiratory and immune function, and poor social support.1 In this commentary, we highlight the challenges of the drug overdose epidemic, and recommend strategies to mitigate the impact of the COVID-19 pandemic among patients with SUDs.
A crisis exacerbated by COVID-19
The current drug overdose epidemic has become an American public health nightmare. According to preliminary data released by the CDC on December 17, 2020, there were more than 81,000 drug overdose deaths in the United States in the 12 months ending May 2020.2,3 This is the highest number of overdose deaths ever recorded in a 12-month period. The CDC also noted that while overdose deaths were already increasing in the months preceding the COVID-19 pandemic, the latest numbers suggest an acceleration of overdose deaths during the pandemic.
What is causing this significant loss of life? Prescription opioids and illegal opioids such as heroin and illicitly manufactured fentanyl are the main agents associated with overdose deaths. These opioids were responsible for 61% (28,647) of drug overdose deaths in the United States in 2014.4 In 2015, the opioid overdose death rate increased by 15.6%.5
The increase in the number of opioid overdose deaths in part coincides with a sharp increase in the availability and use of heroin. Heroin overdose deaths have more than tripled since 2010, but heroin is not the only opiate involved. Fentanyl, a synthetic, short-acting opioid that is approved for managing pain in patients with advanced cancers, is 50 times more potent than heroin. The abuse of prescribed fentanyl has been accelerating over the past decade, as is the use of illicitly produced fentanyl. Evidence from US Drug Enforcement Administration (DEA) seizure records shows heroin is being adulterated with illicit fentanyl to enhance the potency of the heroin.6,7 Mixing illicit fentanyl with heroin may be contributing to the recent increase in heroin overdose fatalities. According to the CDC, overdose deaths related to synthetic opioids increased 38.4% from the 12-month period leading up to June 2019 compared with the 12-month period leading up to May 2020.2,3 Postmortem studies of individuals who died from a heroin overdose have frequently found the presence of fentanyl along with heroin.8 Overdose deaths involving heroin may be occurring because individuals may be unknowingly using heroin adulterated with fentanyl.9 In addition, carfentanil, a powerful new synthetic fentanyl, has been recently identified in heroin mixtures. Carfentanil is 10,000 times stronger than morphine. Even in miniscule amounts, carfentanil can suppress breathing to the degree that multiple doses of naloxone are needed to restore respirations.
Initial studies indicate that the COVID-19 pandemic has been exacerbating this situation. Wainwright et al10 conducted an analysis of urine drug test results of patients with SUDs from 4 months before and 4 months after COVID-19 was declared a national emergency on March 13, 2020. Compared with before COVID-19, the proportion of specimens testing positive since COVID-19 increased from 3.80% to 7.32% for fentanyl and from 1.29% to 2.09% for heroin.10
A similar drug testing study found that during the pandemic, the proportion of positive results (positivity) increased by 35% for non-prescribed fentanyl and 44% for heroin.11 Positivity for non-prescribed fentanyl increased significantly among patients who tested positive for other drugs, including by 89% for amphetamines; 48% for benzodiazepines; 34% for cocaine; and 39% for opiates (P < .1 for all).11
In a review of electronic medical records, Ochalek et al12 found that the number of nonfatal opioid overdoses in an emergency department in Virginia increased from 102 in March-June 2019 to 227 in March-June 2020. In an issue brief published on October 31, 2020, the American Medical Association reported increase in opioid and other drug-related overdoses in more than 40 states during the COVID-19 pandemic.13
Continue to: Strategies for intervention...
Strategies for intervention
A multi-dimensional approach is needed to protect the public from this growing opioid overdose epidemic. To address this challenging task, we recommend several strategies:
Enhance access to virtual treatment
Even when in-person treatment cannot take place due to COVID-19-related restrictions, it is vital that services are accessible to patients with SUDs during this pandemic. Examples of virtual treatment include:
- Telehealth for medication-assisted treatment (MAT) using buprenorphine (recently updated guidance from the US DEA and Substance Abuse and Mental Health Services Administration [SAMHSA] allows this method of prescribing)
- Teletherapy to prevent relapse
- Remote drug screens by sending saliva or urine kits to patients' homes, visiting patients to collect fluid samples, or asking patients to come to a "drive-through" facility to provide samples
- Virtual (online) Alcoholics Anonymous, Narcotics Anonymous, SMART Recovery, and similar meetings to provide support in the absence of in-person meetings.
The American Society of Addiction Medicine (ASAM) offers guidance to treatment programs to focus on infection control and mitigation. The Table14 summarizes the ASAM recommendations for office-based opioid treatment during COVID-19.
Expand access to treatment
This includes access to MAT (such as buprenorphine/naloxone, methadone, naltrexone, and depot naltrexone) and, equally important, to psychosocial treatment, counseling, and/or recovery services. Recent legislative changes have increased the number of patients that a qualified physician can treat with buprenorphine/naloxone from 100 to 275, and allowed physician extenders to prescribe buprenorphine/naloxone in office-based settings. A recent population-based, retrospective Canadian study showed that opioid agonist treatment decreased the risk of mortality among opioid users, and the protective effects of this treatment increased as fentanyl and other synthetic opioids became common in the illicit drug supply.15 However, because of the shortage of psychiatrists and addiction medicine specialists in several regions of the United States, access to treatment is extremely limited and often inadequate. This constitutes a major public health crisis and contributes to our inability to intervene effectively in the opioid epidemic. Telepsychiatry programs can bring needed services to underserved areas, but they need additional support and development. Further, involving other specialties is paramount for treating this epidemic. Integrating MAT in primary care settings can improve access to treatment. Harm-reduction approaches, such as syringe exchange programs, can play an important role in reducing the adverse consequences associated with heroin use and establish health care relationships with at-risk individuals. Syringe exchange programs can also reduce the rate of infections associated with IV drug use, such as human immunodeficiency virus and hepatitis C virus.
Continue to: Increase education on naloxone...
Increase education on naloxone
Naloxone is a safe and effective opioid antagonist used to treat opioid overdoses. Timely access to naloxone is of the essence when treating opioid-related overdoses. Many states have enacted laws allowing health care professionals, law enforcement officers, and patients and relatives to obtain naloxone without a physician's prescription. It appears this approach may be yielding results. For example, the North Carolina Harm Reduction Coalition distributed >101,000 free overdose rescue kits that included naloxone and recorded 13,392 confirmed cases of overdose rescue with naloxone from 2013 to 2019.16
Divert patients with SUDs from the criminal justice system to treatment
We need to develop programs to divert patients with SUDs from the criminal justice system, which is focused on punishment, to interventions that focus on treatment. Data indicates high recidivism rates for incarcerated individuals with SUDs who do not have access to treatment after they are released. Recognizing this, communities are developing programs that divert low-level offenders from the criminal justice system into treatment. For instance, in Seattle, the Law Enforcement Assisted Diversion is a pilot program developed to divert low-level drug and prostitution offenders into community-based treatment and support services. This helps provide housing, health care, job training, treatment, and mental health support. Innovative programs are needed to provide SUD treatment in the rehabilitation programs of correctional facilities and ensure case managers and discharge planners can transition participants to community treatment programs upon their release.
Develop early identification and prevention programs
These programs should focus on individuals at high risk, such as patients with comorbid SUDs and psychiatric disorders, those with chronic pain, and at-risk children whose parents abuse opiates. Traditional addiction treatment programs typically do not address patients with complex conditions or special populations, such as adolescents or pregnant women with substance use issues. Evidence-based approaches such as Screening, Brief Intervention, and Referral to Treatment (SBIRT), Integrated Dual Diagnosis Treatment (IDDT), and prevention approaches that target students in middle schools and high schools need to be more widely available.
Improve education on opioid prescribing
Responsible opioid prescribing for clinicians should include education about the regular use of prescription drug monitoring programs, urine drug screening, avoiding co-prescription of opioids with sedative-hypnotic medications, and better linkage with addiction treatment.
Treat comorbid psychiatric conditions
It is critical to both identify and effectively treat underlying affective, anxiety, and psychotic disorders in patients with SUDs. Anxiety, depression, and emotional dysregulation often contribute to worsening substance abuse, abuse of prescription drugs, diversion of prescribed drugs, and an increased risk of overdoses and suicides. Effective treatment of comorbid psychiatric conditions also may reduce relapses.
Increase research on causes and treatments
Through research, we must expand our knowledge to better understand the factors that contribute to this epidemic and develop better treatments. These efforts may allow for the development of prevention mechanisms. For example, a recent study found that the continued use of opioid medications after an overdose was associated with a high risk of a repeated overdosecall out material?.17 At the end of a 2-year observation, 17% (confidence interval [CI]: 14% to 20%) of patients receiving a high daily dosage of a prescribed opioid had a repeat overdose compared with 15% (CI: 10% to 21%) of those receiving a moderate dosage, 9% (CI: 6% to 14%) of those receiving a low dosage, and 8% (CI: 6% to 11%) of those receiving no opioids.17 Of the patients who overdosed on prescribed opiates, 30% switched to a new prescriber after their overdose, many of whom may not have been aware of the previous overdose. From a public health perspective, it would make sense for prescribers to know of prior opioid and/or benzodiazepine overdoses. This could be reported by emergency department clinicians, law enforcement, and hospitals into a prescription drug monitoring program, which is readily available to prescribers in most states.
Acknowledgment
The authors thank Scott Proescholdbell, MPH, Injury and Violence Prevention Branch, Chronic Disease and Injury Section, Division of Public Health, North Carolina Department of Health and Human Services, for his assistance.
Bottom Line
The collision of the coronavirus disease 2019 pandemic and the drug overdose epidemic has highlighted the urgent need for health care professionals to optimize care for individuals with substance use disorders. Suggested interventions include enhancing access to medication-assisted treatment and virtual treatment, improving education about naloxone and safe opioid prescribing practices, and diverting at-risk patients from the criminal justice system to interventions that focus on treatment.
Editor's Note: Due to updated statistics from the CDC, the online version of this article has been modified from the version that appears in the printed edition of the January 2021 issue of Current Psychiatry.
Individuals with mental health and substance use disorders (SUDs) are particularly susceptible to negative effects of the coronavirus disease 2019 (COVID-19) pandemic. The collision of the COVID-19 pandemic and the drug overdose epidemic has highlighted the urgent need for physicians, policymakers, and health care professionals to optimize care for individuals with SUDs because they may be particularly vulnerable to the effects of the virus due to compromised respiratory and immune function, and poor social support.1 In this commentary, we highlight the challenges of the drug overdose epidemic, and recommend strategies to mitigate the impact of the COVID-19 pandemic among patients with SUDs.
A crisis exacerbated by COVID-19
The current drug overdose epidemic has become an American public health nightmare. According to preliminary data released by the CDC on December 17, 2020, there were more than 81,000 drug overdose deaths in the United States in the 12 months ending May 2020.2,3 This is the highest number of overdose deaths ever recorded in a 12-month period. The CDC also noted that while overdose deaths were already increasing in the months preceding the COVID-19 pandemic, the latest numbers suggest an acceleration of overdose deaths during the pandemic.
What is causing this significant loss of life? Prescription opioids and illegal opioids such as heroin and illicitly manufactured fentanyl are the main agents associated with overdose deaths. These opioids were responsible for 61% (28,647) of drug overdose deaths in the United States in 2014.4 In 2015, the opioid overdose death rate increased by 15.6%.5
The increase in the number of opioid overdose deaths in part coincides with a sharp increase in the availability and use of heroin. Heroin overdose deaths have more than tripled since 2010, but heroin is not the only opiate involved. Fentanyl, a synthetic, short-acting opioid that is approved for managing pain in patients with advanced cancers, is 50 times more potent than heroin. The abuse of prescribed fentanyl has been accelerating over the past decade, as is the use of illicitly produced fentanyl. Evidence from US Drug Enforcement Administration (DEA) seizure records shows heroin is being adulterated with illicit fentanyl to enhance the potency of the heroin.6,7 Mixing illicit fentanyl with heroin may be contributing to the recent increase in heroin overdose fatalities. According to the CDC, overdose deaths related to synthetic opioids increased 38.4% from the 12-month period leading up to June 2019 compared with the 12-month period leading up to May 2020.2,3 Postmortem studies of individuals who died from a heroin overdose have frequently found the presence of fentanyl along with heroin.8 Overdose deaths involving heroin may be occurring because individuals may be unknowingly using heroin adulterated with fentanyl.9 In addition, carfentanil, a powerful new synthetic fentanyl, has been recently identified in heroin mixtures. Carfentanil is 10,000 times stronger than morphine. Even in miniscule amounts, carfentanil can suppress breathing to the degree that multiple doses of naloxone are needed to restore respirations.
Initial studies indicate that the COVID-19 pandemic has been exacerbating this situation. Wainwright et al10 conducted an analysis of urine drug test results of patients with SUDs from 4 months before and 4 months after COVID-19 was declared a national emergency on March 13, 2020. Compared with before COVID-19, the proportion of specimens testing positive since COVID-19 increased from 3.80% to 7.32% for fentanyl and from 1.29% to 2.09% for heroin.10
A similar drug testing study found that during the pandemic, the proportion of positive results (positivity) increased by 35% for non-prescribed fentanyl and 44% for heroin.11 Positivity for non-prescribed fentanyl increased significantly among patients who tested positive for other drugs, including by 89% for amphetamines; 48% for benzodiazepines; 34% for cocaine; and 39% for opiates (P < .1 for all).11
In a review of electronic medical records, Ochalek et al12 found that the number of nonfatal opioid overdoses in an emergency department in Virginia increased from 102 in March-June 2019 to 227 in March-June 2020. In an issue brief published on October 31, 2020, the American Medical Association reported increase in opioid and other drug-related overdoses in more than 40 states during the COVID-19 pandemic.13
Continue to: Strategies for intervention...
Strategies for intervention
A multi-dimensional approach is needed to protect the public from this growing opioid overdose epidemic. To address this challenging task, we recommend several strategies:
Enhance access to virtual treatment
Even when in-person treatment cannot take place due to COVID-19-related restrictions, it is vital that services are accessible to patients with SUDs during this pandemic. Examples of virtual treatment include:
- Telehealth for medication-assisted treatment (MAT) using buprenorphine (recently updated guidance from the US DEA and Substance Abuse and Mental Health Services Administration [SAMHSA] allows this method of prescribing)
- Teletherapy to prevent relapse
- Remote drug screens by sending saliva or urine kits to patients' homes, visiting patients to collect fluid samples, or asking patients to come to a "drive-through" facility to provide samples
- Virtual (online) Alcoholics Anonymous, Narcotics Anonymous, SMART Recovery, and similar meetings to provide support in the absence of in-person meetings.
The American Society of Addiction Medicine (ASAM) offers guidance to treatment programs to focus on infection control and mitigation. The Table14 summarizes the ASAM recommendations for office-based opioid treatment during COVID-19.
Expand access to treatment
This includes access to MAT (such as buprenorphine/naloxone, methadone, naltrexone, and depot naltrexone) and, equally important, to psychosocial treatment, counseling, and/or recovery services. Recent legislative changes have increased the number of patients that a qualified physician can treat with buprenorphine/naloxone from 100 to 275, and allowed physician extenders to prescribe buprenorphine/naloxone in office-based settings. A recent population-based, retrospective Canadian study showed that opioid agonist treatment decreased the risk of mortality among opioid users, and the protective effects of this treatment increased as fentanyl and other synthetic opioids became common in the illicit drug supply.15 However, because of the shortage of psychiatrists and addiction medicine specialists in several regions of the United States, access to treatment is extremely limited and often inadequate. This constitutes a major public health crisis and contributes to our inability to intervene effectively in the opioid epidemic. Telepsychiatry programs can bring needed services to underserved areas, but they need additional support and development. Further, involving other specialties is paramount for treating this epidemic. Integrating MAT in primary care settings can improve access to treatment. Harm-reduction approaches, such as syringe exchange programs, can play an important role in reducing the adverse consequences associated with heroin use and establish health care relationships with at-risk individuals. Syringe exchange programs can also reduce the rate of infections associated with IV drug use, such as human immunodeficiency virus and hepatitis C virus.
Continue to: Increase education on naloxone...
Increase education on naloxone
Naloxone is a safe and effective opioid antagonist used to treat opioid overdoses. Timely access to naloxone is of the essence when treating opioid-related overdoses. Many states have enacted laws allowing health care professionals, law enforcement officers, and patients and relatives to obtain naloxone without a physician's prescription. It appears this approach may be yielding results. For example, the North Carolina Harm Reduction Coalition distributed >101,000 free overdose rescue kits that included naloxone and recorded 13,392 confirmed cases of overdose rescue with naloxone from 2013 to 2019.16
Divert patients with SUDs from the criminal justice system to treatment
We need to develop programs to divert patients with SUDs from the criminal justice system, which is focused on punishment, to interventions that focus on treatment. Data indicates high recidivism rates for incarcerated individuals with SUDs who do not have access to treatment after they are released. Recognizing this, communities are developing programs that divert low-level offenders from the criminal justice system into treatment. For instance, in Seattle, the Law Enforcement Assisted Diversion is a pilot program developed to divert low-level drug and prostitution offenders into community-based treatment and support services. This helps provide housing, health care, job training, treatment, and mental health support. Innovative programs are needed to provide SUD treatment in the rehabilitation programs of correctional facilities and ensure case managers and discharge planners can transition participants to community treatment programs upon their release.
Develop early identification and prevention programs
These programs should focus on individuals at high risk, such as patients with comorbid SUDs and psychiatric disorders, those with chronic pain, and at-risk children whose parents abuse opiates. Traditional addiction treatment programs typically do not address patients with complex conditions or special populations, such as adolescents or pregnant women with substance use issues. Evidence-based approaches such as Screening, Brief Intervention, and Referral to Treatment (SBIRT), Integrated Dual Diagnosis Treatment (IDDT), and prevention approaches that target students in middle schools and high schools need to be more widely available.
Improve education on opioid prescribing
Responsible opioid prescribing for clinicians should include education about the regular use of prescription drug monitoring programs, urine drug screening, avoiding co-prescription of opioids with sedative-hypnotic medications, and better linkage with addiction treatment.
Treat comorbid psychiatric conditions
It is critical to both identify and effectively treat underlying affective, anxiety, and psychotic disorders in patients with SUDs. Anxiety, depression, and emotional dysregulation often contribute to worsening substance abuse, abuse of prescription drugs, diversion of prescribed drugs, and an increased risk of overdoses and suicides. Effective treatment of comorbid psychiatric conditions also may reduce relapses.
Increase research on causes and treatments
Through research, we must expand our knowledge to better understand the factors that contribute to this epidemic and develop better treatments. These efforts may allow for the development of prevention mechanisms. For example, a recent study found that the continued use of opioid medications after an overdose was associated with a high risk of a repeated overdosecall out material?.17 At the end of a 2-year observation, 17% (confidence interval [CI]: 14% to 20%) of patients receiving a high daily dosage of a prescribed opioid had a repeat overdose compared with 15% (CI: 10% to 21%) of those receiving a moderate dosage, 9% (CI: 6% to 14%) of those receiving a low dosage, and 8% (CI: 6% to 11%) of those receiving no opioids.17 Of the patients who overdosed on prescribed opiates, 30% switched to a new prescriber after their overdose, many of whom may not have been aware of the previous overdose. From a public health perspective, it would make sense for prescribers to know of prior opioid and/or benzodiazepine overdoses. This could be reported by emergency department clinicians, law enforcement, and hospitals into a prescription drug monitoring program, which is readily available to prescribers in most states.
Acknowledgment
The authors thank Scott Proescholdbell, MPH, Injury and Violence Prevention Branch, Chronic Disease and Injury Section, Division of Public Health, North Carolina Department of Health and Human Services, for his assistance.
Bottom Line
The collision of the coronavirus disease 2019 pandemic and the drug overdose epidemic has highlighted the urgent need for health care professionals to optimize care for individuals with substance use disorders. Suggested interventions include enhancing access to medication-assisted treatment and virtual treatment, improving education about naloxone and safe opioid prescribing practices, and diverting at-risk patients from the criminal justice system to interventions that focus on treatment.
1. Volkow ND. Collision of the COVID-19 and addiction epidemics. Ann Intern Med. 2020;173(1):61-62.
2.Centers for Disease Control and Prevention. Overdose deaths accelerating during COVID-19. Accessed December 23, 2020. https://www.cdc.gov/media/releases/2020/p1218-overdose-deaths-covid-19.html
3.Centers for Disease Control and Prevention. National Center for Health Statistics Vital Statistics Rapid Release. Provisional drug overdose death counts. Accessed December 30, 2020. https://www.cdc.gov/nchs/nvss/vsrr/drug-overdose-data.htm
4.Rudd RA, Aleshire N, Zibbell JE, et al. Increases in drug and opioid overdose deaths -- United States, 2000-2014. MMWR Morb Mortal Wkly Rep. 2016;64(50-51):1378-1382.
5.Rudd RA, Seth P, David F, et al. Increases in drug and opioid-involved overdose deaths -- United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2016;65(50-51):1445-1452.
6.US Drug Enforcement Administration. DEA issues nationwide alert on fentanyl as threat to health and public safety. Published March 19, 2015. Accessed October 28, 2020. http://www.dea.gov/divisions/hq/2015/hq031815.shtml
7.Gladden RM, Martinez P, Seth P. Fentanyl law enforcement submissions and increases in synthetic opioid-involved overdose deaths - 27 states, 2013-2014. MMWR Morb Mortal Wkly Rep. 2016;65(33):837-843.
8.Algren DA, Monteilh CP, Punja M, et al. Fentanyl-associated fatalities among illicit drug users in Wayne County, Michigan (July 2005-May 2006). J Med Toxicol. 2013;9(1):106-115.
9.Centers for Disease Control and Prevention. Increases in fentanyl drug confiscations and fentanyl-related overdose fatalities. HAN Health Advisory. Published October 26, 2015. Accessed October 28, 2020. http://emergency.cdc.gov/han/han00384.asp
10.Wainwright JJ, Mikre M, Whitley P, et al. Analysis of drug test results before and after the us declaration of a national emergency concerning the COVID-19 outbreak. JAMA. 2020;324(16):1674-1677.
11.Niles JK, Gudin J, Radliff J, et al. The opioid epidemic within the COVID-19 pandemic: drug testing in 2020 [published online October 8, 2020]. Population Health Management. doi: 10.1089/pop.2020.0230
12.Ochalek TA, Cumpston KL, Wills BK, et al. Nonfatal opioid overdoses at an urban emergency department during the COVID-19 pandemic. JAMA. 2020;324(16):1673-1674.
13.American Medical Association. Issue brief: reports of increases in opioid- and other drug-related overdose and other concerns during COVID pandemic. Published October 31, 2020. Accessed November 9, 2020. https://www.ama-assn.org/system/files/2020-11/issue-brief-increases-in-opioid-related-overdose.pdf
14.American Society of Addiction Medicine. Caring for patients during the COVID-19 pandemic: ASAM COVID-19 Task Force recommendations. Accessed October 30, 2020. https://www.asam.org/docs/default-source/covid-19/medication-formulation-and-dosage-guidance-(1).pdf
15.Pearce LA, Min JE, Piske M, et al. Opioid agonist treatment and risk of mortality during opioid overdose public health emergency: population based retrospective cohort study. BMJ. 2020;368:m772. doi: 10.1136/bmj.m772
16.North Carolina Harm Reduction Coalition. NCHRC'S community-based overdose prevention project. Accessed March 29, 2020. http://www.nchrc.org/programs-and-services
17.Larochelle MR, Liebschutz JM, Zhang F, et al. Opioid prescribing after nonfatal overdose and association with repeated overdose: a cohort study. Ann Intern Med. 2016;164(1):1-9.
1. Volkow ND. Collision of the COVID-19 and addiction epidemics. Ann Intern Med. 2020;173(1):61-62.
2.Centers for Disease Control and Prevention. Overdose deaths accelerating during COVID-19. Accessed December 23, 2020. https://www.cdc.gov/media/releases/2020/p1218-overdose-deaths-covid-19.html
3.Centers for Disease Control and Prevention. National Center for Health Statistics Vital Statistics Rapid Release. Provisional drug overdose death counts. Accessed December 30, 2020. https://www.cdc.gov/nchs/nvss/vsrr/drug-overdose-data.htm
4.Rudd RA, Aleshire N, Zibbell JE, et al. Increases in drug and opioid overdose deaths -- United States, 2000-2014. MMWR Morb Mortal Wkly Rep. 2016;64(50-51):1378-1382.
5.Rudd RA, Seth P, David F, et al. Increases in drug and opioid-involved overdose deaths -- United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2016;65(50-51):1445-1452.
6.US Drug Enforcement Administration. DEA issues nationwide alert on fentanyl as threat to health and public safety. Published March 19, 2015. Accessed October 28, 2020. http://www.dea.gov/divisions/hq/2015/hq031815.shtml
7.Gladden RM, Martinez P, Seth P. Fentanyl law enforcement submissions and increases in synthetic opioid-involved overdose deaths - 27 states, 2013-2014. MMWR Morb Mortal Wkly Rep. 2016;65(33):837-843.
8.Algren DA, Monteilh CP, Punja M, et al. Fentanyl-associated fatalities among illicit drug users in Wayne County, Michigan (July 2005-May 2006). J Med Toxicol. 2013;9(1):106-115.
9.Centers for Disease Control and Prevention. Increases in fentanyl drug confiscations and fentanyl-related overdose fatalities. HAN Health Advisory. Published October 26, 2015. Accessed October 28, 2020. http://emergency.cdc.gov/han/han00384.asp
10.Wainwright JJ, Mikre M, Whitley P, et al. Analysis of drug test results before and after the us declaration of a national emergency concerning the COVID-19 outbreak. JAMA. 2020;324(16):1674-1677.
11.Niles JK, Gudin J, Radliff J, et al. The opioid epidemic within the COVID-19 pandemic: drug testing in 2020 [published online October 8, 2020]. Population Health Management. doi: 10.1089/pop.2020.0230
12.Ochalek TA, Cumpston KL, Wills BK, et al. Nonfatal opioid overdoses at an urban emergency department during the COVID-19 pandemic. JAMA. 2020;324(16):1673-1674.
13.American Medical Association. Issue brief: reports of increases in opioid- and other drug-related overdose and other concerns during COVID pandemic. Published October 31, 2020. Accessed November 9, 2020. https://www.ama-assn.org/system/files/2020-11/issue-brief-increases-in-opioid-related-overdose.pdf
14.American Society of Addiction Medicine. Caring for patients during the COVID-19 pandemic: ASAM COVID-19 Task Force recommendations. Accessed October 30, 2020. https://www.asam.org/docs/default-source/covid-19/medication-formulation-and-dosage-guidance-(1).pdf
15.Pearce LA, Min JE, Piske M, et al. Opioid agonist treatment and risk of mortality during opioid overdose public health emergency: population based retrospective cohort study. BMJ. 2020;368:m772. doi: 10.1136/bmj.m772
16.North Carolina Harm Reduction Coalition. NCHRC'S community-based overdose prevention project. Accessed March 29, 2020. http://www.nchrc.org/programs-and-services
17.Larochelle MR, Liebschutz JM, Zhang F, et al. Opioid prescribing after nonfatal overdose and association with repeated overdose: a cohort study. Ann Intern Med. 2016;164(1):1-9.