User login
'Clinical equipoise' seen for surgical approaches to early rectal cancer
PHOENIX – Two differing surgical approaches for early rectal cancer – radical resection and local resection—appear to be essentially equivalent treatment options, finds a systematic review and meta-analysis.
The conclusion is based on an analysis of morbidity and mortality data from 13 studies with a total of 2,855 patients with stage T1N0M0 rectal adenocarcinoma. All of the studies were published after 1979, when total mesorectal excision and modern local resection techniques were being used.
There are "improved results with the newer techniques of TEMS [transanal endoscopic microsurgery] and TAMIS [transanal minimally invasive surgery], as well as comparable outcomes when we adjust for the fact that there is a bit of a selection bias in the literature for lower-third lesions to be performed by local resection," lead investigator Dr. Sami A. Chadi reported at the annual meeting of the American Society of Colon and Rectal Surgeons.
"We know that quality of life data is better with local resection," he commented. Relative to radical resection, local resection was associated with an 87% lower risk of postoperative complications, a 69% lower risk of perioperative mortality, and an 83% lower risk of permanent ostomy.
On the other hand, local resection also was associated with a 46% higher likelihood of death at 5 years. Survival no longer differed significantly, however, when analyses took into account the greater use of local resection for cancers located in the lower third of the rectum.
"The implication is that we have established clinical equipoise between groups with T1N0M0 adenocarcinoma of the rectum, thus prompting the need for a prospective randomized, controlled trial on these two procedures," he maintained. "We do need further data to assess whether or not there is a role for neoadjuvant or adjuvant therapy in these groups." Ongoing studies are assessing the role of therapy, as well as the potential for local resection to be performed for T2 lesions.
Dr. Chadi, a surgeon with the University of Western Ontario in London, and his colleagues analyzed data from 12 observational studies and one randomized, controlled trial among patients with T1N0M0 cancer.
The 5-year rate of overall survival was poorer with local resection (relative risk, 1.46), with the difference between groups corresponding to 72 more deaths per 1,000 patients in the local resection group, according to Dr. Chadi, who disclosed no conflicts of interest related to the research.
However, this difference was largely driven by transanal excision (TAE) local procedures. There was no significant difference in this outcome for TEMS local procedures as compared with radical resection.
The researchers also repeated the survival analysis with an adjustment for cancers in the lower third of the rectum. In these patients, the surgical choice is more often local resection, potentially leading to selection bias.
When the ratio of lower-third cancers was equal in both the radical and local resection groups, there was no longer a significant difference in 5-year overall survival.
Compared with radical resection, local resection yielded a lower risk of postoperative complications (rate ratio, 0.13), with the difference corresponding to 129 fewer complications per 1,000 patients in the local resection group. The difference was significant for both TAE and TEMS individually as compared with radical resection.
Local resection also was associated with a lower risk of perioperative mortality (rate ratio, 0.31), with the difference corresponding to 11 fewer deaths per 1,000 patients in the local resection group, and a lower risk of permanent ostomy (risk ratio, 0.17), with the difference corresponding to 225 fewer permanent ostomies per 1,000 patients in the local resection group.
Local excision is an ideal management strategy for early-stage rectal cancer patients, T1 and/or T2 with no nodal involvement and no metastases. It would be great if we could pursue local excision as a primary management strategy. At this point, however, the data in the literature suggest that radical resection is associated with superior oncological outcomes.
Robust data from a randomized controlled trial are needed to decide management. Dr. Chadi was said that such a trial would have to include patients who are candidates for either procedure and are diagnosed using a standard procedure such as magnetic resonance imaging or endorectal ultrasound. Comorbidities and receipt of adjuvant therapy would be among the main confounders to consider in trial design. Study subgroups would include patients with lesions in either the lower one-third or the upper two-thirds of the rectum, and the sample size would need to be powered to account for the selection bias for lower third lesions to have local resection.
Since the initiation of local resection, newer techniques have become available that allow for better visualization and more precise dissections. This has given rise to the hope that oncologic outcomes—historically superior with radical resection—might now be similar with local resection.
Dr. Allyson H. Stone, of the Dartmouth-Hitchcock Medical Center in Lebanon, N.H., was the invited discussant of the study. She had no relevant financial disclosures.
Local excision is an ideal management strategy for early-stage rectal cancer patients, T1 and/or T2 with no nodal involvement and no metastases. It would be great if we could pursue local excision as a primary management strategy. At this point, however, the data in the literature suggest that radical resection is associated with superior oncological outcomes.
Robust data from a randomized controlled trial are needed to decide management. Dr. Chadi was said that such a trial would have to include patients who are candidates for either procedure and are diagnosed using a standard procedure such as magnetic resonance imaging or endorectal ultrasound. Comorbidities and receipt of adjuvant therapy would be among the main confounders to consider in trial design. Study subgroups would include patients with lesions in either the lower one-third or the upper two-thirds of the rectum, and the sample size would need to be powered to account for the selection bias for lower third lesions to have local resection.
Since the initiation of local resection, newer techniques have become available that allow for better visualization and more precise dissections. This has given rise to the hope that oncologic outcomes—historically superior with radical resection—might now be similar with local resection.
Dr. Allyson H. Stone, of the Dartmouth-Hitchcock Medical Center in Lebanon, N.H., was the invited discussant of the study. She had no relevant financial disclosures.
Local excision is an ideal management strategy for early-stage rectal cancer patients, T1 and/or T2 with no nodal involvement and no metastases. It would be great if we could pursue local excision as a primary management strategy. At this point, however, the data in the literature suggest that radical resection is associated with superior oncological outcomes.
Robust data from a randomized controlled trial are needed to decide management. Dr. Chadi was said that such a trial would have to include patients who are candidates for either procedure and are diagnosed using a standard procedure such as magnetic resonance imaging or endorectal ultrasound. Comorbidities and receipt of adjuvant therapy would be among the main confounders to consider in trial design. Study subgroups would include patients with lesions in either the lower one-third or the upper two-thirds of the rectum, and the sample size would need to be powered to account for the selection bias for lower third lesions to have local resection.
Since the initiation of local resection, newer techniques have become available that allow for better visualization and more precise dissections. This has given rise to the hope that oncologic outcomes—historically superior with radical resection—might now be similar with local resection.
Dr. Allyson H. Stone, of the Dartmouth-Hitchcock Medical Center in Lebanon, N.H., was the invited discussant of the study. She had no relevant financial disclosures.
PHOENIX – Two differing surgical approaches for early rectal cancer – radical resection and local resection—appear to be essentially equivalent treatment options, finds a systematic review and meta-analysis.
The conclusion is based on an analysis of morbidity and mortality data from 13 studies with a total of 2,855 patients with stage T1N0M0 rectal adenocarcinoma. All of the studies were published after 1979, when total mesorectal excision and modern local resection techniques were being used.
There are "improved results with the newer techniques of TEMS [transanal endoscopic microsurgery] and TAMIS [transanal minimally invasive surgery], as well as comparable outcomes when we adjust for the fact that there is a bit of a selection bias in the literature for lower-third lesions to be performed by local resection," lead investigator Dr. Sami A. Chadi reported at the annual meeting of the American Society of Colon and Rectal Surgeons.
"We know that quality of life data is better with local resection," he commented. Relative to radical resection, local resection was associated with an 87% lower risk of postoperative complications, a 69% lower risk of perioperative mortality, and an 83% lower risk of permanent ostomy.
On the other hand, local resection also was associated with a 46% higher likelihood of death at 5 years. Survival no longer differed significantly, however, when analyses took into account the greater use of local resection for cancers located in the lower third of the rectum.
"The implication is that we have established clinical equipoise between groups with T1N0M0 adenocarcinoma of the rectum, thus prompting the need for a prospective randomized, controlled trial on these two procedures," he maintained. "We do need further data to assess whether or not there is a role for neoadjuvant or adjuvant therapy in these groups." Ongoing studies are assessing the role of therapy, as well as the potential for local resection to be performed for T2 lesions.
Dr. Chadi, a surgeon with the University of Western Ontario in London, and his colleagues analyzed data from 12 observational studies and one randomized, controlled trial among patients with T1N0M0 cancer.
The 5-year rate of overall survival was poorer with local resection (relative risk, 1.46), with the difference between groups corresponding to 72 more deaths per 1,000 patients in the local resection group, according to Dr. Chadi, who disclosed no conflicts of interest related to the research.
However, this difference was largely driven by transanal excision (TAE) local procedures. There was no significant difference in this outcome for TEMS local procedures as compared with radical resection.
The researchers also repeated the survival analysis with an adjustment for cancers in the lower third of the rectum. In these patients, the surgical choice is more often local resection, potentially leading to selection bias.
When the ratio of lower-third cancers was equal in both the radical and local resection groups, there was no longer a significant difference in 5-year overall survival.
Compared with radical resection, local resection yielded a lower risk of postoperative complications (rate ratio, 0.13), with the difference corresponding to 129 fewer complications per 1,000 patients in the local resection group. The difference was significant for both TAE and TEMS individually as compared with radical resection.
Local resection also was associated with a lower risk of perioperative mortality (rate ratio, 0.31), with the difference corresponding to 11 fewer deaths per 1,000 patients in the local resection group, and a lower risk of permanent ostomy (risk ratio, 0.17), with the difference corresponding to 225 fewer permanent ostomies per 1,000 patients in the local resection group.
PHOENIX – Two differing surgical approaches for early rectal cancer – radical resection and local resection—appear to be essentially equivalent treatment options, finds a systematic review and meta-analysis.
The conclusion is based on an analysis of morbidity and mortality data from 13 studies with a total of 2,855 patients with stage T1N0M0 rectal adenocarcinoma. All of the studies were published after 1979, when total mesorectal excision and modern local resection techniques were being used.
There are "improved results with the newer techniques of TEMS [transanal endoscopic microsurgery] and TAMIS [transanal minimally invasive surgery], as well as comparable outcomes when we adjust for the fact that there is a bit of a selection bias in the literature for lower-third lesions to be performed by local resection," lead investigator Dr. Sami A. Chadi reported at the annual meeting of the American Society of Colon and Rectal Surgeons.
"We know that quality of life data is better with local resection," he commented. Relative to radical resection, local resection was associated with an 87% lower risk of postoperative complications, a 69% lower risk of perioperative mortality, and an 83% lower risk of permanent ostomy.
On the other hand, local resection also was associated with a 46% higher likelihood of death at 5 years. Survival no longer differed significantly, however, when analyses took into account the greater use of local resection for cancers located in the lower third of the rectum.
"The implication is that we have established clinical equipoise between groups with T1N0M0 adenocarcinoma of the rectum, thus prompting the need for a prospective randomized, controlled trial on these two procedures," he maintained. "We do need further data to assess whether or not there is a role for neoadjuvant or adjuvant therapy in these groups." Ongoing studies are assessing the role of therapy, as well as the potential for local resection to be performed for T2 lesions.
Dr. Chadi, a surgeon with the University of Western Ontario in London, and his colleagues analyzed data from 12 observational studies and one randomized, controlled trial among patients with T1N0M0 cancer.
The 5-year rate of overall survival was poorer with local resection (relative risk, 1.46), with the difference between groups corresponding to 72 more deaths per 1,000 patients in the local resection group, according to Dr. Chadi, who disclosed no conflicts of interest related to the research.
However, this difference was largely driven by transanal excision (TAE) local procedures. There was no significant difference in this outcome for TEMS local procedures as compared with radical resection.
The researchers also repeated the survival analysis with an adjustment for cancers in the lower third of the rectum. In these patients, the surgical choice is more often local resection, potentially leading to selection bias.
When the ratio of lower-third cancers was equal in both the radical and local resection groups, there was no longer a significant difference in 5-year overall survival.
Compared with radical resection, local resection yielded a lower risk of postoperative complications (rate ratio, 0.13), with the difference corresponding to 129 fewer complications per 1,000 patients in the local resection group. The difference was significant for both TAE and TEMS individually as compared with radical resection.
Local resection also was associated with a lower risk of perioperative mortality (rate ratio, 0.31), with the difference corresponding to 11 fewer deaths per 1,000 patients in the local resection group, and a lower risk of permanent ostomy (risk ratio, 0.17), with the difference corresponding to 225 fewer permanent ostomies per 1,000 patients in the local resection group.
AT THE ASCRS ANNUAL MEETING
Major finding: Compared with radical resection, local resection was associated with a lower risk of perioperative mortality (rate ratio, 0.31), with the difference corresponding to 11 fewer deaths per 1,000 patients and a lower risk of permanent ostomy (risk ratio, 0.17), with the difference corresponding to 225 fewer permanent ostomies per 1,000 patients.
Data source: A systematic review and meta-analysis of 12 observational studies and one randomized controlled trial with a total of 2,855 patients with T1N0M0 rectal cancer.
Disclosures: Dr. Chadi disclosed no relevant conflicts of interest.
Less may be more in postop colorectal cancer follow-up
CHICAGO – Long-term intensive blood and computed tomography follow-up offered no significant advantage over minimal follow-up for detecting colorectal cancer recurrences in the phase III FACS trial.
The proportion of patients with recurrence treated surgically with curative intent was 6.7% with intensive carcinoembryonic antigen (CEA) testing, 8% with monthly computed tomography imaging and 6.6% with combination CEA plus CT, compared with 2.3% with minimal follow-up involving a single CT scan.
After adjustment, it was about three times more likely that patients would have a recurrence with CEA (adjusted odds ratio, 2.70; P = .035), monthly CT imaging (OR, 3.45; P = .007) and CEA plus CT (OR, 2.95; P = .021) than with minimal follow-up.
"This result appeared to be quite robust" and was independent of cancer stage, co-primary investigator Dr. David Mant said at the annual meeting of the American Society of Clinical Oncology.
There was virtually no deviation in protocol by patients, however, the desire for unscheduled CT scans by surgeons meant that up to 30% in the minimal follow-up arm had 1 or more additional CTs, he noted. Still, in a per protocol analysis, the absolute differences in recurrence were 3.8- to 5.4-times higher with intensive follow-up. Again, there was no evidence of an additive effect of CEA plus CT.
Guidelines in the United States and Europe stress intensive follow-up including routine history and physical examination, CEA monitoring, yearly colonoscopy and repeated CT scans in those at high risk of recurrence.
The FACS (Follow-Up After Colorectal Surgery) trial looked at whether long-term intensive follow-up was worthwhile. Though commonplace after curative surgery, economic modeling suggests intensive followup may not be cost-effective and claims of a substantial overall survival benefit are inconsistent with the reported frequency and effectiveness of treatment for recurrence, explained Dr. David Mant, emeritus professor of general practice, Oxford University.
"As a long-term family physician, I’d seen many patients being followed-up for cancer and watched their cycle of deep anxiety, followed usually by relief, but sometimes with misery, as they waited for their follow-up appointments," he added. "It seemed to me that it was a very bad idea to carry on with this practice, unless it was to their benefit."
FACS involved 1,202 patients who were disease free on colonoscopy and CT imaging and had a blood CEA level of 10 mcg/L or less (Dukes’ stages A-C) after treatment for primary colorectal cancer. Their median age was 70 years.
Patients were randomly assigned to one of four follow-up regimens: "minimal" follow-up based mainly on symptoms and a single CT scan at 12-18 months; "CEA," which included minimal follow-up, plus 3 monthly blood CEA tests for 2 years then 6 monthly tests in years 3-5; "CT," which included minimal follow-up plus intensive CT imaging involving 6 monthly scans of the chest, abdomen, and pelvis for 2 years, and then annually for another 3 years; and "CEA plus CT," which included the CEA regimen in group 2 and the CT measures in group 3.
If during monitoring, a patient’s CEA level was 7 mcg/L or more above their baseline level at trial entry, the test was repeated as soon as possible. If the second test was also above the threshold, the patient’s physician was asked to refer the patient urgently to the local hospital.
Overall, 6% of patients (71/1,202) developed a recurrence treated surgically with curative intent. There was little difference between patients according to Duke’s staging (stage A, 5.1%; stage B, 6.1%; stage C, 6.2%), reported Dr. Mant and coprincipal investigator Dr. John Primrose of University of Southhampton, U.K.
"You actually do need rigorous initial staging to detect residual disease before you embark on any follow-up," Dr. Mant stressed. "I think both John and I are convinced that the reason why we have a 6% overall recurrence rate is because the people in this trial were appropriately staged. The survival curves don’t suggest they were any less sick than patients in other trials."
At the time of the analysis, 59% of patients with a recurrence treated with curative intent were still alive, but there was no statistical difference in colorectal cancer deaths (P = .66) or total deaths (P = .45) between the minimum and intensive follow-up arms, he said.
In a meta-analysis that integrated the FACS data with that from three previous trials, the overall effect of intensive follow-up was not significant (OR, 0.96; P = .56), Dr. Mant said. The number needed to treat to detect potentially curable recurrence with intensive follow-up is about 20 to 25 and that predicts a number-needed-to-treat of about 40 to 50 for 5-year post-recurrence survival.
Dr. Maughan said the disparity between the current meta-analysis and the Cochrane review may be because the latter included six studies, not three, and that the survival data from FACS may not be fully mature. A further analysis of the FACS data is planned in about 18 months.
Dr. Mant and his coauthors reported having no financial disclosures.
Mant, D., et al. "Effect of 3-5 years of scheduled CED and CT follow-up to detect recurrence of colorectal cancer: FACS randomized controlled trial." Ab. 3500.
Current guidelines are based on older data indicating that follow-up for patients with resected colorectal cancer adds as much benefit to overall survival as does adjuvant chemotherapy (hazard ratio 0.73) (Cochrane Database Syst Rev. 2007;CD002200). This conclusion may not reflect the outcomes associated with more current treatments.
Although FACS was of insufficient statistical power to estimate survival advantage, the lack of difference between the three intensive follow-up protocols challenges our current practice of repeated CT scans with their extra radiation exposure.
Clinicians also may want to rethink the emphasis on history and physical exam in the follow-up of these patients. FACS shows that symptomatic follow-up and clinical appointments add nothing to the detection of resectable recurrence. The main effect of monitoring was seen with a single CT scan at 12-18 months.
Dr. Tim Maughan is professor of clinical oncology at Oxford University and was the invited discussant of the paper at the meeting. He reported a consultant/advisory role with Sanofi and research funding from Merck Serono.
Current guidelines are based on older data indicating that follow-up for patients with resected colorectal cancer adds as much benefit to overall survival as does adjuvant chemotherapy (hazard ratio 0.73) (Cochrane Database Syst Rev. 2007;CD002200). This conclusion may not reflect the outcomes associated with more current treatments.
Although FACS was of insufficient statistical power to estimate survival advantage, the lack of difference between the three intensive follow-up protocols challenges our current practice of repeated CT scans with their extra radiation exposure.
Clinicians also may want to rethink the emphasis on history and physical exam in the follow-up of these patients. FACS shows that symptomatic follow-up and clinical appointments add nothing to the detection of resectable recurrence. The main effect of monitoring was seen with a single CT scan at 12-18 months.
Dr. Tim Maughan is professor of clinical oncology at Oxford University and was the invited discussant of the paper at the meeting. He reported a consultant/advisory role with Sanofi and research funding from Merck Serono.
Current guidelines are based on older data indicating that follow-up for patients with resected colorectal cancer adds as much benefit to overall survival as does adjuvant chemotherapy (hazard ratio 0.73) (Cochrane Database Syst Rev. 2007;CD002200). This conclusion may not reflect the outcomes associated with more current treatments.
Although FACS was of insufficient statistical power to estimate survival advantage, the lack of difference between the three intensive follow-up protocols challenges our current practice of repeated CT scans with their extra radiation exposure.
Clinicians also may want to rethink the emphasis on history and physical exam in the follow-up of these patients. FACS shows that symptomatic follow-up and clinical appointments add nothing to the detection of resectable recurrence. The main effect of monitoring was seen with a single CT scan at 12-18 months.
Dr. Tim Maughan is professor of clinical oncology at Oxford University and was the invited discussant of the paper at the meeting. He reported a consultant/advisory role with Sanofi and research funding from Merck Serono.
CHICAGO – Long-term intensive blood and computed tomography follow-up offered no significant advantage over minimal follow-up for detecting colorectal cancer recurrences in the phase III FACS trial.
The proportion of patients with recurrence treated surgically with curative intent was 6.7% with intensive carcinoembryonic antigen (CEA) testing, 8% with monthly computed tomography imaging and 6.6% with combination CEA plus CT, compared with 2.3% with minimal follow-up involving a single CT scan.
After adjustment, it was about three times more likely that patients would have a recurrence with CEA (adjusted odds ratio, 2.70; P = .035), monthly CT imaging (OR, 3.45; P = .007) and CEA plus CT (OR, 2.95; P = .021) than with minimal follow-up.
"This result appeared to be quite robust" and was independent of cancer stage, co-primary investigator Dr. David Mant said at the annual meeting of the American Society of Clinical Oncology.
There was virtually no deviation in protocol by patients, however, the desire for unscheduled CT scans by surgeons meant that up to 30% in the minimal follow-up arm had 1 or more additional CTs, he noted. Still, in a per protocol analysis, the absolute differences in recurrence were 3.8- to 5.4-times higher with intensive follow-up. Again, there was no evidence of an additive effect of CEA plus CT.
Guidelines in the United States and Europe stress intensive follow-up including routine history and physical examination, CEA monitoring, yearly colonoscopy and repeated CT scans in those at high risk of recurrence.
The FACS (Follow-Up After Colorectal Surgery) trial looked at whether long-term intensive follow-up was worthwhile. Though commonplace after curative surgery, economic modeling suggests intensive followup may not be cost-effective and claims of a substantial overall survival benefit are inconsistent with the reported frequency and effectiveness of treatment for recurrence, explained Dr. David Mant, emeritus professor of general practice, Oxford University.
"As a long-term family physician, I’d seen many patients being followed-up for cancer and watched their cycle of deep anxiety, followed usually by relief, but sometimes with misery, as they waited for their follow-up appointments," he added. "It seemed to me that it was a very bad idea to carry on with this practice, unless it was to their benefit."
FACS involved 1,202 patients who were disease free on colonoscopy and CT imaging and had a blood CEA level of 10 mcg/L or less (Dukes’ stages A-C) after treatment for primary colorectal cancer. Their median age was 70 years.
Patients were randomly assigned to one of four follow-up regimens: "minimal" follow-up based mainly on symptoms and a single CT scan at 12-18 months; "CEA," which included minimal follow-up, plus 3 monthly blood CEA tests for 2 years then 6 monthly tests in years 3-5; "CT," which included minimal follow-up plus intensive CT imaging involving 6 monthly scans of the chest, abdomen, and pelvis for 2 years, and then annually for another 3 years; and "CEA plus CT," which included the CEA regimen in group 2 and the CT measures in group 3.
If during monitoring, a patient’s CEA level was 7 mcg/L or more above their baseline level at trial entry, the test was repeated as soon as possible. If the second test was also above the threshold, the patient’s physician was asked to refer the patient urgently to the local hospital.
Overall, 6% of patients (71/1,202) developed a recurrence treated surgically with curative intent. There was little difference between patients according to Duke’s staging (stage A, 5.1%; stage B, 6.1%; stage C, 6.2%), reported Dr. Mant and coprincipal investigator Dr. John Primrose of University of Southhampton, U.K.
"You actually do need rigorous initial staging to detect residual disease before you embark on any follow-up," Dr. Mant stressed. "I think both John and I are convinced that the reason why we have a 6% overall recurrence rate is because the people in this trial were appropriately staged. The survival curves don’t suggest they were any less sick than patients in other trials."
At the time of the analysis, 59% of patients with a recurrence treated with curative intent were still alive, but there was no statistical difference in colorectal cancer deaths (P = .66) or total deaths (P = .45) between the minimum and intensive follow-up arms, he said.
In a meta-analysis that integrated the FACS data with that from three previous trials, the overall effect of intensive follow-up was not significant (OR, 0.96; P = .56), Dr. Mant said. The number needed to treat to detect potentially curable recurrence with intensive follow-up is about 20 to 25 and that predicts a number-needed-to-treat of about 40 to 50 for 5-year post-recurrence survival.
Dr. Maughan said the disparity between the current meta-analysis and the Cochrane review may be because the latter included six studies, not three, and that the survival data from FACS may not be fully mature. A further analysis of the FACS data is planned in about 18 months.
Dr. Mant and his coauthors reported having no financial disclosures.
Mant, D., et al. "Effect of 3-5 years of scheduled CED and CT follow-up to detect recurrence of colorectal cancer: FACS randomized controlled trial." Ab. 3500.
CHICAGO – Long-term intensive blood and computed tomography follow-up offered no significant advantage over minimal follow-up for detecting colorectal cancer recurrences in the phase III FACS trial.
The proportion of patients with recurrence treated surgically with curative intent was 6.7% with intensive carcinoembryonic antigen (CEA) testing, 8% with monthly computed tomography imaging and 6.6% with combination CEA plus CT, compared with 2.3% with minimal follow-up involving a single CT scan.
After adjustment, it was about three times more likely that patients would have a recurrence with CEA (adjusted odds ratio, 2.70; P = .035), monthly CT imaging (OR, 3.45; P = .007) and CEA plus CT (OR, 2.95; P = .021) than with minimal follow-up.
"This result appeared to be quite robust" and was independent of cancer stage, co-primary investigator Dr. David Mant said at the annual meeting of the American Society of Clinical Oncology.
There was virtually no deviation in protocol by patients, however, the desire for unscheduled CT scans by surgeons meant that up to 30% in the minimal follow-up arm had 1 or more additional CTs, he noted. Still, in a per protocol analysis, the absolute differences in recurrence were 3.8- to 5.4-times higher with intensive follow-up. Again, there was no evidence of an additive effect of CEA plus CT.
Guidelines in the United States and Europe stress intensive follow-up including routine history and physical examination, CEA monitoring, yearly colonoscopy and repeated CT scans in those at high risk of recurrence.
The FACS (Follow-Up After Colorectal Surgery) trial looked at whether long-term intensive follow-up was worthwhile. Though commonplace after curative surgery, economic modeling suggests intensive followup may not be cost-effective and claims of a substantial overall survival benefit are inconsistent with the reported frequency and effectiveness of treatment for recurrence, explained Dr. David Mant, emeritus professor of general practice, Oxford University.
"As a long-term family physician, I’d seen many patients being followed-up for cancer and watched their cycle of deep anxiety, followed usually by relief, but sometimes with misery, as they waited for their follow-up appointments," he added. "It seemed to me that it was a very bad idea to carry on with this practice, unless it was to their benefit."
FACS involved 1,202 patients who were disease free on colonoscopy and CT imaging and had a blood CEA level of 10 mcg/L or less (Dukes’ stages A-C) after treatment for primary colorectal cancer. Their median age was 70 years.
Patients were randomly assigned to one of four follow-up regimens: "minimal" follow-up based mainly on symptoms and a single CT scan at 12-18 months; "CEA," which included minimal follow-up, plus 3 monthly blood CEA tests for 2 years then 6 monthly tests in years 3-5; "CT," which included minimal follow-up plus intensive CT imaging involving 6 monthly scans of the chest, abdomen, and pelvis for 2 years, and then annually for another 3 years; and "CEA plus CT," which included the CEA regimen in group 2 and the CT measures in group 3.
If during monitoring, a patient’s CEA level was 7 mcg/L or more above their baseline level at trial entry, the test was repeated as soon as possible. If the second test was also above the threshold, the patient’s physician was asked to refer the patient urgently to the local hospital.
Overall, 6% of patients (71/1,202) developed a recurrence treated surgically with curative intent. There was little difference between patients according to Duke’s staging (stage A, 5.1%; stage B, 6.1%; stage C, 6.2%), reported Dr. Mant and coprincipal investigator Dr. John Primrose of University of Southhampton, U.K.
"You actually do need rigorous initial staging to detect residual disease before you embark on any follow-up," Dr. Mant stressed. "I think both John and I are convinced that the reason why we have a 6% overall recurrence rate is because the people in this trial were appropriately staged. The survival curves don’t suggest they were any less sick than patients in other trials."
At the time of the analysis, 59% of patients with a recurrence treated with curative intent were still alive, but there was no statistical difference in colorectal cancer deaths (P = .66) or total deaths (P = .45) between the minimum and intensive follow-up arms, he said.
In a meta-analysis that integrated the FACS data with that from three previous trials, the overall effect of intensive follow-up was not significant (OR, 0.96; P = .56), Dr. Mant said. The number needed to treat to detect potentially curable recurrence with intensive follow-up is about 20 to 25 and that predicts a number-needed-to-treat of about 40 to 50 for 5-year post-recurrence survival.
Dr. Maughan said the disparity between the current meta-analysis and the Cochrane review may be because the latter included six studies, not three, and that the survival data from FACS may not be fully mature. A further analysis of the FACS data is planned in about 18 months.
Dr. Mant and his coauthors reported having no financial disclosures.
Mant, D., et al. "Effect of 3-5 years of scheduled CED and CT follow-up to detect recurrence of colorectal cancer: FACS randomized controlled trial." Ab. 3500.
AT ASCO ANNUAL MEETING 2013
Major finding: The risk of recurrence after surgical treatment with curative intent was about three times higher with intensive carcinoembryonic antigen monitoring (odds ratio, 2.70; P = .035), computed tomography (OR, 3.45; P = .007) and CEA plus CT follow-up (OR, 2.95; P = .021) than with minimal follow-up. At analysis, 59% of patients with a recurrence treated with curative intent were still alive, but there was no statistical difference in colorectal cancer deaths (P = .66) or total deaths (P = .45) between the minimum and intensive follow-up arms.
Data source: Phase III trial of 1,202 patients from 39 U.K. hospitals who had completed curative treatment for colorectal cancer.
Disclosures: Dr. Mant and his coauthors reported having no financial disclosures.
Benefits of laparoscopic over open colectomy decrease with operative time
PHOENIX – The longer a laparoscopic colectomy for cancer takes, the less its advantages over an open colectomy in terms of morbidity and mortality, researchers reported at the annual meeting of the American Society of Colon and Rectal Surgeons.
A team led by Dr. Matthew Bailey, a surgery resident at the University of Kentucky in Lexington, queried the National Surgical Quality Improvement Program (NSQIP) database to identify more than 4,000 patients undergoing right colectomy because of colorectal malignancy.
Results showed that compared with their peers having an open operation lasting 3 hours or less, patients having a laparoscopic operation of this duration were less likely to develop complications and to die. However, when the operations lasted more than 3 hours, there was no longer any significant difference.
Within the laparoscopic group, patients were more likely to have a procedure lasting longer than 3 hours if they had recently received radiation therapy, were morbidly obese, or had peripheral vascular disease.
"We recommend that surgeons consider an open approach if the patient has risk factors for an operative duration greater than 3 hours," Dr. Bailey said. "We also recommend surgeons consider conversion to an open approach when it is anticipated that a laparoscopic right colectomy will exceed 3 hours."
Dr. Walter Peters of Columbia (Mo.) Surgical Associates, who comoderated the session, asked, "Were the laparoscopic procedures lasting more than 3 hours concentrated in a few institutions, or were they spread across the entire NSQIP database?"
The investigators did not assess the institutional distribution, and it may not be possible to tease that information out of NSQIP, Dr. Bailey replied.
Session attendee Dr. Eric Haas, of Colorectal Surgical Associates in Houston asked what percentage of patients had a conversion from laparoscopic to open procedures and whether analyses were conducted according to intention to treat.
"NSQIP unfortunately does not allow you to discern that, there’s no CPT [Current Procedural Terminology] code for conversion," Dr. Bailey said. "We can only assume that cases that were converted laparoscopic to open were most likely ultimately coded as open. So there was no way to perform an intent-to-treat analysis."
Dr. Haas also noted that surgeon experience with laparoscopy may have played a role. "In my own experience, certainly at the beginning of the learning curve phase, I would take 3, maybe 4 hours. And the one risk factor that you can’t put [in analyses] is surgeon," he said. "So were these 3-hour cases because of the learning curve, or were they true 3-hour cases because of the patient factors?"
The NSQIP database captures the surgeon who dictates the operative report and the level of training, the highest-level resident involved, and the specialty of the surgeon (although colorectal surgery is not among the options), according to Dr. Bailey. Still, it is generally not possible to determine who did all or most of the operation.
"We did look at operative time, and it was around 138 minutes, plus-minus, for a laparoscopic right colectomy, with the reported literature being around 187 minutes. So I’m not sure if the reported literature is dated or if, in these over 200 hospitals across the nation, this is realistic of current practice trends," he said.
"The study shows that patients are probably going to have increased complications with longer surgery, whether it’s laparoscopic or open," Dr. Timothy Geiger of Vanderbilt University Medical Center, Nashville, Tenn., the session’s other comoderator, said in an interview. "It was a good study, but it needs a little bit more in-depth look, especially at things like redosing of antibiotics, whether that is done in an appropriate manner. But otherwise, it’s a great kind of intro for us to look at this."
Giving some background to the research, Dr. Bailey noted, "The use of laparoscopy in colon cancer has been shown to be equivalent to open surgery in survival and oncologic outcomes. The question of whether operative time negatively impacts laparoscopic outcomes compared to open surgery outcomes requires further investigation if we are to endorse a laparoscopic approach regardless of procedure length. We postulated that there is an operative duration where the benefits of a laparoscopic approach are negated."
The investigators analyzed data from the NSQIP database for the years 2005 through 2010, identifying patients who had a right colectomy for cancer and excluding those treated on an emergent basis, or having secondary procedures other than enterolysis or mobilization of the splenic flexure.
Analyses were based on 2,141 patients in the laparoscopic group and 2,132 patients in the open group. Procedures lasted longer than 3 hours in 18% of the former and 11% of the latter.
Compared with the open group, the laparoscopic group was younger and had lower American Society of Anesthesiologists scores, higher serum albumin levels, and lower prothrombin time and platelet count.
In unadjusted analysis among patients whose operations lasted 3 hours or less, the laparoscopic group fared better than did the open group in terms of 30-day mortality (1.1% vs 2.6%, P less than .001), cardiopulmonary and cerebrovascular complications (5.4% vs. 8.4%, P less than .001), and infectious complications (9.2% vs. 13.3%, P less than .001).
In contrast, among patients whose operations lasted more than 3 hours, differences in these outcomes were not significant. If anything, there was a trend toward a higher rate of infectious complications with laparoscopy.
To assess the role of preoperative risk factors, the investigators looked at NSQIP estimates of morbidity and mortality for all gastrointestinal and abdominal surgery by operative duration and including 37 preoperative risk factors, and found patterns differing from those in the study cohort.
Specifically, these estimates suggested mortality was consistently lower for procedures lasting more then 3 hours, whether laparoscopic or open. And the risk of infectious complications was constant for each type and less for laparoscopy, regardless of the operative duration.
Thus, "there is something other than preoperative risk factors causing an increase in mortality" with longer operative time for laparoscopic procedures in the study cohort, said Dr. Bailey, who disclosed no conflicts of interest related to the research. Also, "the increased infectious complications that we demonstrated are not related to the morbidity risk."
Patients in the laparoscopic group had a significantly higher risk of surgery lasting more than 3 hours if they had recently received radiation therapy (odds ratio, 6.5), were morbidly obese (3.0), or had peripheral vascular disease (2.5).
Hospital length of stay was consistently shorter with the laparoscopic approach, regardless of how long the operation lasted, according to Dr. Bailey.
PHOENIX – The longer a laparoscopic colectomy for cancer takes, the less its advantages over an open colectomy in terms of morbidity and mortality, researchers reported at the annual meeting of the American Society of Colon and Rectal Surgeons.
A team led by Dr. Matthew Bailey, a surgery resident at the University of Kentucky in Lexington, queried the National Surgical Quality Improvement Program (NSQIP) database to identify more than 4,000 patients undergoing right colectomy because of colorectal malignancy.
Results showed that compared with their peers having an open operation lasting 3 hours or less, patients having a laparoscopic operation of this duration were less likely to develop complications and to die. However, when the operations lasted more than 3 hours, there was no longer any significant difference.
Within the laparoscopic group, patients were more likely to have a procedure lasting longer than 3 hours if they had recently received radiation therapy, were morbidly obese, or had peripheral vascular disease.
"We recommend that surgeons consider an open approach if the patient has risk factors for an operative duration greater than 3 hours," Dr. Bailey said. "We also recommend surgeons consider conversion to an open approach when it is anticipated that a laparoscopic right colectomy will exceed 3 hours."
Dr. Walter Peters of Columbia (Mo.) Surgical Associates, who comoderated the session, asked, "Were the laparoscopic procedures lasting more than 3 hours concentrated in a few institutions, or were they spread across the entire NSQIP database?"
The investigators did not assess the institutional distribution, and it may not be possible to tease that information out of NSQIP, Dr. Bailey replied.
Session attendee Dr. Eric Haas, of Colorectal Surgical Associates in Houston asked what percentage of patients had a conversion from laparoscopic to open procedures and whether analyses were conducted according to intention to treat.
"NSQIP unfortunately does not allow you to discern that, there’s no CPT [Current Procedural Terminology] code for conversion," Dr. Bailey said. "We can only assume that cases that were converted laparoscopic to open were most likely ultimately coded as open. So there was no way to perform an intent-to-treat analysis."
Dr. Haas also noted that surgeon experience with laparoscopy may have played a role. "In my own experience, certainly at the beginning of the learning curve phase, I would take 3, maybe 4 hours. And the one risk factor that you can’t put [in analyses] is surgeon," he said. "So were these 3-hour cases because of the learning curve, or were they true 3-hour cases because of the patient factors?"
The NSQIP database captures the surgeon who dictates the operative report and the level of training, the highest-level resident involved, and the specialty of the surgeon (although colorectal surgery is not among the options), according to Dr. Bailey. Still, it is generally not possible to determine who did all or most of the operation.
"We did look at operative time, and it was around 138 minutes, plus-minus, for a laparoscopic right colectomy, with the reported literature being around 187 minutes. So I’m not sure if the reported literature is dated or if, in these over 200 hospitals across the nation, this is realistic of current practice trends," he said.
"The study shows that patients are probably going to have increased complications with longer surgery, whether it’s laparoscopic or open," Dr. Timothy Geiger of Vanderbilt University Medical Center, Nashville, Tenn., the session’s other comoderator, said in an interview. "It was a good study, but it needs a little bit more in-depth look, especially at things like redosing of antibiotics, whether that is done in an appropriate manner. But otherwise, it’s a great kind of intro for us to look at this."
Giving some background to the research, Dr. Bailey noted, "The use of laparoscopy in colon cancer has been shown to be equivalent to open surgery in survival and oncologic outcomes. The question of whether operative time negatively impacts laparoscopic outcomes compared to open surgery outcomes requires further investigation if we are to endorse a laparoscopic approach regardless of procedure length. We postulated that there is an operative duration where the benefits of a laparoscopic approach are negated."
The investigators analyzed data from the NSQIP database for the years 2005 through 2010, identifying patients who had a right colectomy for cancer and excluding those treated on an emergent basis, or having secondary procedures other than enterolysis or mobilization of the splenic flexure.
Analyses were based on 2,141 patients in the laparoscopic group and 2,132 patients in the open group. Procedures lasted longer than 3 hours in 18% of the former and 11% of the latter.
Compared with the open group, the laparoscopic group was younger and had lower American Society of Anesthesiologists scores, higher serum albumin levels, and lower prothrombin time and platelet count.
In unadjusted analysis among patients whose operations lasted 3 hours or less, the laparoscopic group fared better than did the open group in terms of 30-day mortality (1.1% vs 2.6%, P less than .001), cardiopulmonary and cerebrovascular complications (5.4% vs. 8.4%, P less than .001), and infectious complications (9.2% vs. 13.3%, P less than .001).
In contrast, among patients whose operations lasted more than 3 hours, differences in these outcomes were not significant. If anything, there was a trend toward a higher rate of infectious complications with laparoscopy.
To assess the role of preoperative risk factors, the investigators looked at NSQIP estimates of morbidity and mortality for all gastrointestinal and abdominal surgery by operative duration and including 37 preoperative risk factors, and found patterns differing from those in the study cohort.
Specifically, these estimates suggested mortality was consistently lower for procedures lasting more then 3 hours, whether laparoscopic or open. And the risk of infectious complications was constant for each type and less for laparoscopy, regardless of the operative duration.
Thus, "there is something other than preoperative risk factors causing an increase in mortality" with longer operative time for laparoscopic procedures in the study cohort, said Dr. Bailey, who disclosed no conflicts of interest related to the research. Also, "the increased infectious complications that we demonstrated are not related to the morbidity risk."
Patients in the laparoscopic group had a significantly higher risk of surgery lasting more than 3 hours if they had recently received radiation therapy (odds ratio, 6.5), were morbidly obese (3.0), or had peripheral vascular disease (2.5).
Hospital length of stay was consistently shorter with the laparoscopic approach, regardless of how long the operation lasted, according to Dr. Bailey.
PHOENIX – The longer a laparoscopic colectomy for cancer takes, the less its advantages over an open colectomy in terms of morbidity and mortality, researchers reported at the annual meeting of the American Society of Colon and Rectal Surgeons.
A team led by Dr. Matthew Bailey, a surgery resident at the University of Kentucky in Lexington, queried the National Surgical Quality Improvement Program (NSQIP) database to identify more than 4,000 patients undergoing right colectomy because of colorectal malignancy.
Results showed that compared with their peers having an open operation lasting 3 hours or less, patients having a laparoscopic operation of this duration were less likely to develop complications and to die. However, when the operations lasted more than 3 hours, there was no longer any significant difference.
Within the laparoscopic group, patients were more likely to have a procedure lasting longer than 3 hours if they had recently received radiation therapy, were morbidly obese, or had peripheral vascular disease.
"We recommend that surgeons consider an open approach if the patient has risk factors for an operative duration greater than 3 hours," Dr. Bailey said. "We also recommend surgeons consider conversion to an open approach when it is anticipated that a laparoscopic right colectomy will exceed 3 hours."
Dr. Walter Peters of Columbia (Mo.) Surgical Associates, who comoderated the session, asked, "Were the laparoscopic procedures lasting more than 3 hours concentrated in a few institutions, or were they spread across the entire NSQIP database?"
The investigators did not assess the institutional distribution, and it may not be possible to tease that information out of NSQIP, Dr. Bailey replied.
Session attendee Dr. Eric Haas, of Colorectal Surgical Associates in Houston asked what percentage of patients had a conversion from laparoscopic to open procedures and whether analyses were conducted according to intention to treat.
"NSQIP unfortunately does not allow you to discern that, there’s no CPT [Current Procedural Terminology] code for conversion," Dr. Bailey said. "We can only assume that cases that were converted laparoscopic to open were most likely ultimately coded as open. So there was no way to perform an intent-to-treat analysis."
Dr. Haas also noted that surgeon experience with laparoscopy may have played a role. "In my own experience, certainly at the beginning of the learning curve phase, I would take 3, maybe 4 hours. And the one risk factor that you can’t put [in analyses] is surgeon," he said. "So were these 3-hour cases because of the learning curve, or were they true 3-hour cases because of the patient factors?"
The NSQIP database captures the surgeon who dictates the operative report and the level of training, the highest-level resident involved, and the specialty of the surgeon (although colorectal surgery is not among the options), according to Dr. Bailey. Still, it is generally not possible to determine who did all or most of the operation.
"We did look at operative time, and it was around 138 minutes, plus-minus, for a laparoscopic right colectomy, with the reported literature being around 187 minutes. So I’m not sure if the reported literature is dated or if, in these over 200 hospitals across the nation, this is realistic of current practice trends," he said.
"The study shows that patients are probably going to have increased complications with longer surgery, whether it’s laparoscopic or open," Dr. Timothy Geiger of Vanderbilt University Medical Center, Nashville, Tenn., the session’s other comoderator, said in an interview. "It was a good study, but it needs a little bit more in-depth look, especially at things like redosing of antibiotics, whether that is done in an appropriate manner. But otherwise, it’s a great kind of intro for us to look at this."
Giving some background to the research, Dr. Bailey noted, "The use of laparoscopy in colon cancer has been shown to be equivalent to open surgery in survival and oncologic outcomes. The question of whether operative time negatively impacts laparoscopic outcomes compared to open surgery outcomes requires further investigation if we are to endorse a laparoscopic approach regardless of procedure length. We postulated that there is an operative duration where the benefits of a laparoscopic approach are negated."
The investigators analyzed data from the NSQIP database for the years 2005 through 2010, identifying patients who had a right colectomy for cancer and excluding those treated on an emergent basis, or having secondary procedures other than enterolysis or mobilization of the splenic flexure.
Analyses were based on 2,141 patients in the laparoscopic group and 2,132 patients in the open group. Procedures lasted longer than 3 hours in 18% of the former and 11% of the latter.
Compared with the open group, the laparoscopic group was younger and had lower American Society of Anesthesiologists scores, higher serum albumin levels, and lower prothrombin time and platelet count.
In unadjusted analysis among patients whose operations lasted 3 hours or less, the laparoscopic group fared better than did the open group in terms of 30-day mortality (1.1% vs 2.6%, P less than .001), cardiopulmonary and cerebrovascular complications (5.4% vs. 8.4%, P less than .001), and infectious complications (9.2% vs. 13.3%, P less than .001).
In contrast, among patients whose operations lasted more than 3 hours, differences in these outcomes were not significant. If anything, there was a trend toward a higher rate of infectious complications with laparoscopy.
To assess the role of preoperative risk factors, the investigators looked at NSQIP estimates of morbidity and mortality for all gastrointestinal and abdominal surgery by operative duration and including 37 preoperative risk factors, and found patterns differing from those in the study cohort.
Specifically, these estimates suggested mortality was consistently lower for procedures lasting more then 3 hours, whether laparoscopic or open. And the risk of infectious complications was constant for each type and less for laparoscopy, regardless of the operative duration.
Thus, "there is something other than preoperative risk factors causing an increase in mortality" with longer operative time for laparoscopic procedures in the study cohort, said Dr. Bailey, who disclosed no conflicts of interest related to the research. Also, "the increased infectious complications that we demonstrated are not related to the morbidity risk."
Patients in the laparoscopic group had a significantly higher risk of surgery lasting more than 3 hours if they had recently received radiation therapy (odds ratio, 6.5), were morbidly obese (3.0), or had peripheral vascular disease (2.5).
Hospital length of stay was consistently shorter with the laparoscopic approach, regardless of how long the operation lasted, according to Dr. Bailey.
AT THE ASCRS ANNUAL MEETING
Major Finding: The rates of complications and death postoperatively were lower with the laparoscopic approach if the operation lasted 3 hours or less, but not if it lasted longer.
Data Source: A retrospective cohort study of 4,273 cases of laparoscopic or open right colectomy for cancer.
Disclosures: Dr. Bailey disclosed no relevant conflicts of interest.
Risk-based strategy bests drug therapy after Crohn's surgery
ORLANDO – Risk-based drug treatment with colonoscopy at 6 months – and treatment step-up for recurrence – was significantly more effective in preventing postoperative recurrence of Crohn’s disease, compared with standard drug therapy alone, based on data from the POCER (Post-Operative Crohn’s Endoscopic Recurrence) study.
At an 18-month follow-up colonoscopy, 49% of patients randomized to a risk-based intervention had endoscopic recurrence, compared with 67% of those in a standard drug therapy arm, Dr. Peter De Cruz reported during a late-breaking abstract session at the annual Digestive Disease Week.
Researchers compared the two recurrence-prevention strategies in 174 subjects who were stratified as high risk or low risk based on smoking status, presence of perforating disease, and previous surgeries. The participants were randomized 2:1 to either active care or standard drug therapy. Approximately one-third of patients in the active care arm showed signs of early recurrence at a 6-month colonoscopy and thus received step-up treatment.
Complete mucosal normality was observed in 22% of those in the risk-based treatment arm, compared with 8% in the standard therapy arm; no significant differences were noted with respect to severe endoscopic recurrence or clinical recurrence, said Dr. De Cruz of St. Vincent’s Hospital, Melbourne, Australia.
Patients in the multicenter POCER study were adults undergoing intestinal resection of all macroscopic disease. The treatment arms were similar in terms of demographics, disease characteristics, and prior drug therapy. Of 122 patients in the risk-based treatment arm, 101 were high-risk, and 21 were low-risk. Of 52 patients in the standard drug therapy arm, 44 were high-risk, and 8 were low-risk.
All patients were treated beginning postoperatively with 3 months of 400 mg metronidazole twice daily. High-risk patients also received 2 mg/kg of azathioprine daily, or 1.5 mg/kg of 6 mercaptopurine. Those intolerant of thiopurine were treated with adalimumab induction, followed by 40 mg every 2 weeks. Low-risk patients received no additional treatment following the 3 months of metronidazole.
For recurrence at 6 months, low-risk patients were stepped up to thiopurine; high-risk patients on thiopurine were stepped up to 40 mg adalimumab every 2 weeks, and high-risk patients on adalimumab every 2 weeks stepped up to weekly adalimumab.
The endoscopic remission rate at 18 months was 25% for the low-risk patients who stepped up to thiopurine 41% for high-risk patients who stepped up to thiopurine, and 50% for those who stepped up to weekly adalimumab.
Overall, 39% of patients who stepped up at 6 months were in remission at 1 year. Notably, 31% of the low-risk patients with endoscopic remission at the 6-month colonoscopy, 46% of the high-risk patients on thiopurine, and 41% of the high-risk patients on adalimumab with endoscopic remission at 6 months had endoscopic recurrence 1 year later.
"We can’t afford to relax," Dr. De Cruz said regarding those patients with remission 6 months postoperatively.
This study also allowed for comparison of the efficacy of adalimumab and thiopurine given immediately postoperatively in the high-risk patients. At 6 months, adalimumab was associated with a nearly 80% endoscopic remission rate, compared with 55% for thiopurine, Dr. De Cruz noted.
At 18 months, there was no significant difference in the recurrence rates between patients treated with adalimumab immediately postoperatively and those stepped up to adalimumab in combination with thiopurine. There was, however, a trend toward reduced recurrence rates among those on immediate postoperative adalimumab.
A multivariate analysis showed that active smoking was significantly associated with an increased endoscopic recurrence rate at 18 months, whereas the intervention of endoscopy at 6 months was significantly associated with a reduced recurrence risk at 18 months.
Most prior studies of postoperative recurrence prevention in Crohn’s surgery patients have focused on drug therapy compared with placebo; few have focused on risk-based strategies, Dr. De Cruz noted.
The findings indicate that step-up therapy based on endoscopy is a viable postoperative strategy in patients at high risk of recurrence, he said. No unexpected serious adverse events occurred in this study.
Dr. De Cruz warned, however, that remission at 6 months is no guarantee of remission 1 year later.
"Intensifying treatment at 6 months brings about 40% of patients with recurrence into remission, and we found that a small group of patients had recurrent disease despite endoscopic monitoring and intense treatment," he said.
Dr. De Cruz reported having no disclosures. Several study coauthors disclosed relationships with multiple pharmaceutical companies.
ORLANDO – Risk-based drug treatment with colonoscopy at 6 months – and treatment step-up for recurrence – was significantly more effective in preventing postoperative recurrence of Crohn’s disease, compared with standard drug therapy alone, based on data from the POCER (Post-Operative Crohn’s Endoscopic Recurrence) study.
At an 18-month follow-up colonoscopy, 49% of patients randomized to a risk-based intervention had endoscopic recurrence, compared with 67% of those in a standard drug therapy arm, Dr. Peter De Cruz reported during a late-breaking abstract session at the annual Digestive Disease Week.
Researchers compared the two recurrence-prevention strategies in 174 subjects who were stratified as high risk or low risk based on smoking status, presence of perforating disease, and previous surgeries. The participants were randomized 2:1 to either active care or standard drug therapy. Approximately one-third of patients in the active care arm showed signs of early recurrence at a 6-month colonoscopy and thus received step-up treatment.
Complete mucosal normality was observed in 22% of those in the risk-based treatment arm, compared with 8% in the standard therapy arm; no significant differences were noted with respect to severe endoscopic recurrence or clinical recurrence, said Dr. De Cruz of St. Vincent’s Hospital, Melbourne, Australia.
Patients in the multicenter POCER study were adults undergoing intestinal resection of all macroscopic disease. The treatment arms were similar in terms of demographics, disease characteristics, and prior drug therapy. Of 122 patients in the risk-based treatment arm, 101 were high-risk, and 21 were low-risk. Of 52 patients in the standard drug therapy arm, 44 were high-risk, and 8 were low-risk.
All patients were treated beginning postoperatively with 3 months of 400 mg metronidazole twice daily. High-risk patients also received 2 mg/kg of azathioprine daily, or 1.5 mg/kg of 6 mercaptopurine. Those intolerant of thiopurine were treated with adalimumab induction, followed by 40 mg every 2 weeks. Low-risk patients received no additional treatment following the 3 months of metronidazole.
For recurrence at 6 months, low-risk patients were stepped up to thiopurine; high-risk patients on thiopurine were stepped up to 40 mg adalimumab every 2 weeks, and high-risk patients on adalimumab every 2 weeks stepped up to weekly adalimumab.
The endoscopic remission rate at 18 months was 25% for the low-risk patients who stepped up to thiopurine 41% for high-risk patients who stepped up to thiopurine, and 50% for those who stepped up to weekly adalimumab.
Overall, 39% of patients who stepped up at 6 months were in remission at 1 year. Notably, 31% of the low-risk patients with endoscopic remission at the 6-month colonoscopy, 46% of the high-risk patients on thiopurine, and 41% of the high-risk patients on adalimumab with endoscopic remission at 6 months had endoscopic recurrence 1 year later.
"We can’t afford to relax," Dr. De Cruz said regarding those patients with remission 6 months postoperatively.
This study also allowed for comparison of the efficacy of adalimumab and thiopurine given immediately postoperatively in the high-risk patients. At 6 months, adalimumab was associated with a nearly 80% endoscopic remission rate, compared with 55% for thiopurine, Dr. De Cruz noted.
At 18 months, there was no significant difference in the recurrence rates between patients treated with adalimumab immediately postoperatively and those stepped up to adalimumab in combination with thiopurine. There was, however, a trend toward reduced recurrence rates among those on immediate postoperative adalimumab.
A multivariate analysis showed that active smoking was significantly associated with an increased endoscopic recurrence rate at 18 months, whereas the intervention of endoscopy at 6 months was significantly associated with a reduced recurrence risk at 18 months.
Most prior studies of postoperative recurrence prevention in Crohn’s surgery patients have focused on drug therapy compared with placebo; few have focused on risk-based strategies, Dr. De Cruz noted.
The findings indicate that step-up therapy based on endoscopy is a viable postoperative strategy in patients at high risk of recurrence, he said. No unexpected serious adverse events occurred in this study.
Dr. De Cruz warned, however, that remission at 6 months is no guarantee of remission 1 year later.
"Intensifying treatment at 6 months brings about 40% of patients with recurrence into remission, and we found that a small group of patients had recurrent disease despite endoscopic monitoring and intense treatment," he said.
Dr. De Cruz reported having no disclosures. Several study coauthors disclosed relationships with multiple pharmaceutical companies.
ORLANDO – Risk-based drug treatment with colonoscopy at 6 months – and treatment step-up for recurrence – was significantly more effective in preventing postoperative recurrence of Crohn’s disease, compared with standard drug therapy alone, based on data from the POCER (Post-Operative Crohn’s Endoscopic Recurrence) study.
At an 18-month follow-up colonoscopy, 49% of patients randomized to a risk-based intervention had endoscopic recurrence, compared with 67% of those in a standard drug therapy arm, Dr. Peter De Cruz reported during a late-breaking abstract session at the annual Digestive Disease Week.
Researchers compared the two recurrence-prevention strategies in 174 subjects who were stratified as high risk or low risk based on smoking status, presence of perforating disease, and previous surgeries. The participants were randomized 2:1 to either active care or standard drug therapy. Approximately one-third of patients in the active care arm showed signs of early recurrence at a 6-month colonoscopy and thus received step-up treatment.
Complete mucosal normality was observed in 22% of those in the risk-based treatment arm, compared with 8% in the standard therapy arm; no significant differences were noted with respect to severe endoscopic recurrence or clinical recurrence, said Dr. De Cruz of St. Vincent’s Hospital, Melbourne, Australia.
Patients in the multicenter POCER study were adults undergoing intestinal resection of all macroscopic disease. The treatment arms were similar in terms of demographics, disease characteristics, and prior drug therapy. Of 122 patients in the risk-based treatment arm, 101 were high-risk, and 21 were low-risk. Of 52 patients in the standard drug therapy arm, 44 were high-risk, and 8 were low-risk.
All patients were treated beginning postoperatively with 3 months of 400 mg metronidazole twice daily. High-risk patients also received 2 mg/kg of azathioprine daily, or 1.5 mg/kg of 6 mercaptopurine. Those intolerant of thiopurine were treated with adalimumab induction, followed by 40 mg every 2 weeks. Low-risk patients received no additional treatment following the 3 months of metronidazole.
For recurrence at 6 months, low-risk patients were stepped up to thiopurine; high-risk patients on thiopurine were stepped up to 40 mg adalimumab every 2 weeks, and high-risk patients on adalimumab every 2 weeks stepped up to weekly adalimumab.
The endoscopic remission rate at 18 months was 25% for the low-risk patients who stepped up to thiopurine 41% for high-risk patients who stepped up to thiopurine, and 50% for those who stepped up to weekly adalimumab.
Overall, 39% of patients who stepped up at 6 months were in remission at 1 year. Notably, 31% of the low-risk patients with endoscopic remission at the 6-month colonoscopy, 46% of the high-risk patients on thiopurine, and 41% of the high-risk patients on adalimumab with endoscopic remission at 6 months had endoscopic recurrence 1 year later.
"We can’t afford to relax," Dr. De Cruz said regarding those patients with remission 6 months postoperatively.
This study also allowed for comparison of the efficacy of adalimumab and thiopurine given immediately postoperatively in the high-risk patients. At 6 months, adalimumab was associated with a nearly 80% endoscopic remission rate, compared with 55% for thiopurine, Dr. De Cruz noted.
At 18 months, there was no significant difference in the recurrence rates between patients treated with adalimumab immediately postoperatively and those stepped up to adalimumab in combination with thiopurine. There was, however, a trend toward reduced recurrence rates among those on immediate postoperative adalimumab.
A multivariate analysis showed that active smoking was significantly associated with an increased endoscopic recurrence rate at 18 months, whereas the intervention of endoscopy at 6 months was significantly associated with a reduced recurrence risk at 18 months.
Most prior studies of postoperative recurrence prevention in Crohn’s surgery patients have focused on drug therapy compared with placebo; few have focused on risk-based strategies, Dr. De Cruz noted.
The findings indicate that step-up therapy based on endoscopy is a viable postoperative strategy in patients at high risk of recurrence, he said. No unexpected serious adverse events occurred in this study.
Dr. De Cruz warned, however, that remission at 6 months is no guarantee of remission 1 year later.
"Intensifying treatment at 6 months brings about 40% of patients with recurrence into remission, and we found that a small group of patients had recurrent disease despite endoscopic monitoring and intense treatment," he said.
Dr. De Cruz reported having no disclosures. Several study coauthors disclosed relationships with multiple pharmaceutical companies.
AT DDW 2013
Major finding: 49% vs. 67% 18-month recurrence rates for risk-based vs. standard drug therapy.
Data source: Data from 174 adults in the randomized, controlled POCER study.
Disclosures: Dr. De Cruz reported having no disclosures. Several study co-authors disclosed relationships with multiple pharmaceutical companies.
Mechanical bowel prep may up cancer-specific survival after CRC resection
PHOENIX – Improving cancer-specific survival after resection for colorectal cancer may be as simple as ordering preoperative mechanical bowel preparation, according to an analysis of data from a randomized trial conducted in Sweden and Germany.
Roughly half of the 841 patients studied had such preparation before their operation. The actuarial 5-year rate of cancer-specific survival was about 8% higher in this group, first author Dr. Åsa Collin of Uppsala University reported at the annual meeting of the American Society of Colon and Rectal Surgeons.
An analysis of cancer stage at the time of surgery suggested that the two groups were well matched on this measure. Preparation did not significantly decrease the rate of recurrence or increase the rate of overall survival.
Session attendee Dr. J. Daniel Stanley of University Surgical Associates in Chattanooga, Tenn., questioned whether the patients might have differed on other factors that influenced whether they underwent mechanical bowel preparation.
"Were some of them obstructed, which might indicate a different tumor biology than was reflected in the staging?" he asked.
"These were all elective surgeries, and there was no patient who we actually decided that they shouldn’t have preparation," Dr. Collin replied.
In an interview, session comoderator Dr. David Maron, a colorectal surgeon at the Cleveland Clinic in Weston, Fla., said, "What was interesting was that they didn’t show a significant difference in cancer recurrence, although it was close to significance. So the question remains, is it in fact the mechanical bowel prep, or could there be differences in the postoperative follow-up and even the postoperative treatment of those patients who developed recurrence?"
Guidelines leave preparation up to the treating surgeon, and several studies have found no benefit at least in the short term, he noted. "This is one of the first studies that have shown that perhaps from a long-term standpoint in patients with cancer, that there may be some benefit to preoperative preparation of the bowel, although again, it’s preliminary and there’s a lot of unknowns out there."
Dr. Collin and her colleagues analyzed data from a trial among patients who underwent an elective resection for cancer, adenoma, or diverticular disease of the colon at 21 hospitals in Sweden and Germany between 1999 and 2005.
Earlier results for the entire trial population, previously reported, showed no significant reduction in 30-day rates of complications with mechanical bowel preparation (Br. J. Surg. 2007;94:689-95).
The new, long-term results for just the 841 patients with colorectal cancer – 53% of whom had a mechanical bowel preparation before their surgery – indicated those undergoing this preparation had a higher actuarial 5-year rate of cancer-specific survival (90% vs. 82%; P = .03).
The two groups were similar in terms of the 5-year rate of recurrence (80% vs. 89%, P = .08) and overall survival, Dr. Collin said.
"We looked at tumor stage to see if the explanation for patients having no mechanical bowel preparation having poorer cancer-specific survival was that they had more advanced tumors, and there was no difference in the stages" between groups, she said, with the majority of patients in both groups having stage II or III disease.
Dr. Collin disclosed no conflicts of interest related to the research.
PHOENIX – Improving cancer-specific survival after resection for colorectal cancer may be as simple as ordering preoperative mechanical bowel preparation, according to an analysis of data from a randomized trial conducted in Sweden and Germany.
Roughly half of the 841 patients studied had such preparation before their operation. The actuarial 5-year rate of cancer-specific survival was about 8% higher in this group, first author Dr. Åsa Collin of Uppsala University reported at the annual meeting of the American Society of Colon and Rectal Surgeons.
An analysis of cancer stage at the time of surgery suggested that the two groups were well matched on this measure. Preparation did not significantly decrease the rate of recurrence or increase the rate of overall survival.
Session attendee Dr. J. Daniel Stanley of University Surgical Associates in Chattanooga, Tenn., questioned whether the patients might have differed on other factors that influenced whether they underwent mechanical bowel preparation.
"Were some of them obstructed, which might indicate a different tumor biology than was reflected in the staging?" he asked.
"These were all elective surgeries, and there was no patient who we actually decided that they shouldn’t have preparation," Dr. Collin replied.
In an interview, session comoderator Dr. David Maron, a colorectal surgeon at the Cleveland Clinic in Weston, Fla., said, "What was interesting was that they didn’t show a significant difference in cancer recurrence, although it was close to significance. So the question remains, is it in fact the mechanical bowel prep, or could there be differences in the postoperative follow-up and even the postoperative treatment of those patients who developed recurrence?"
Guidelines leave preparation up to the treating surgeon, and several studies have found no benefit at least in the short term, he noted. "This is one of the first studies that have shown that perhaps from a long-term standpoint in patients with cancer, that there may be some benefit to preoperative preparation of the bowel, although again, it’s preliminary and there’s a lot of unknowns out there."
Dr. Collin and her colleagues analyzed data from a trial among patients who underwent an elective resection for cancer, adenoma, or diverticular disease of the colon at 21 hospitals in Sweden and Germany between 1999 and 2005.
Earlier results for the entire trial population, previously reported, showed no significant reduction in 30-day rates of complications with mechanical bowel preparation (Br. J. Surg. 2007;94:689-95).
The new, long-term results for just the 841 patients with colorectal cancer – 53% of whom had a mechanical bowel preparation before their surgery – indicated those undergoing this preparation had a higher actuarial 5-year rate of cancer-specific survival (90% vs. 82%; P = .03).
The two groups were similar in terms of the 5-year rate of recurrence (80% vs. 89%, P = .08) and overall survival, Dr. Collin said.
"We looked at tumor stage to see if the explanation for patients having no mechanical bowel preparation having poorer cancer-specific survival was that they had more advanced tumors, and there was no difference in the stages" between groups, she said, with the majority of patients in both groups having stage II or III disease.
Dr. Collin disclosed no conflicts of interest related to the research.
PHOENIX – Improving cancer-specific survival after resection for colorectal cancer may be as simple as ordering preoperative mechanical bowel preparation, according to an analysis of data from a randomized trial conducted in Sweden and Germany.
Roughly half of the 841 patients studied had such preparation before their operation. The actuarial 5-year rate of cancer-specific survival was about 8% higher in this group, first author Dr. Åsa Collin of Uppsala University reported at the annual meeting of the American Society of Colon and Rectal Surgeons.
An analysis of cancer stage at the time of surgery suggested that the two groups were well matched on this measure. Preparation did not significantly decrease the rate of recurrence or increase the rate of overall survival.
Session attendee Dr. J. Daniel Stanley of University Surgical Associates in Chattanooga, Tenn., questioned whether the patients might have differed on other factors that influenced whether they underwent mechanical bowel preparation.
"Were some of them obstructed, which might indicate a different tumor biology than was reflected in the staging?" he asked.
"These were all elective surgeries, and there was no patient who we actually decided that they shouldn’t have preparation," Dr. Collin replied.
In an interview, session comoderator Dr. David Maron, a colorectal surgeon at the Cleveland Clinic in Weston, Fla., said, "What was interesting was that they didn’t show a significant difference in cancer recurrence, although it was close to significance. So the question remains, is it in fact the mechanical bowel prep, or could there be differences in the postoperative follow-up and even the postoperative treatment of those patients who developed recurrence?"
Guidelines leave preparation up to the treating surgeon, and several studies have found no benefit at least in the short term, he noted. "This is one of the first studies that have shown that perhaps from a long-term standpoint in patients with cancer, that there may be some benefit to preoperative preparation of the bowel, although again, it’s preliminary and there’s a lot of unknowns out there."
Dr. Collin and her colleagues analyzed data from a trial among patients who underwent an elective resection for cancer, adenoma, or diverticular disease of the colon at 21 hospitals in Sweden and Germany between 1999 and 2005.
Earlier results for the entire trial population, previously reported, showed no significant reduction in 30-day rates of complications with mechanical bowel preparation (Br. J. Surg. 2007;94:689-95).
The new, long-term results for just the 841 patients with colorectal cancer – 53% of whom had a mechanical bowel preparation before their surgery – indicated those undergoing this preparation had a higher actuarial 5-year rate of cancer-specific survival (90% vs. 82%; P = .03).
The two groups were similar in terms of the 5-year rate of recurrence (80% vs. 89%, P = .08) and overall survival, Dr. Collin said.
"We looked at tumor stage to see if the explanation for patients having no mechanical bowel preparation having poorer cancer-specific survival was that they had more advanced tumors, and there was no difference in the stages" between groups, she said, with the majority of patients in both groups having stage II or III disease.
Dr. Collin disclosed no conflicts of interest related to the research.
AT THE ASCRS ANNUAL MEETING
Major finding: Patients who had mechanical bowel preparation before surgery had a higher actuarial 5-year rate of cancer-specific survival (90% vs. 82%).
Data source: An analysis of 841 patients undergoing elective resection for colorectal cancer in a randomized trial.
Disclosures: Dr. Collin disclosed no relevant conflicts of interest.
Anti-TNFs for ulcerative colitis up sepsis risk after some proctocolectomies
PHOENIX – Among patients with ulcerative colitis, preoperative therapy targeting tumor necrosis factor increases the risk of postoperative complications after two-stage restorative proctocolectomy procedures but not after three-stage ones, a study has shown.
A team at the Cleveland Clinic retrospectively assessed outcomes in more than 500 patients who underwent a restorative proctocolectomy for medically refractory ulcerative colitis during a recent 5-year period. Overall, 28% were receiving an agent that targets tumor necrosis factor (TNF) before their surgery.
The main results, reported at the annual meeting of the American Society of Colon and Rectal Surgeons, showed that among patients having an initial total proctocolectomy (TPC) with ileoanal pouch–anal anastomosis, preoperative anti-TNF therapy more than doubled the risk of pelvic sepsis in the subsequent year.
In contrast, among patients having an initial subtotal colectomy (STC) with end ileostomy, preoperative anti-TNF therapy did not significantly affect the risk of complications overall, or in the subset who went on to have completion proctectomy and ileoanal pouch–anal anastomosis.
"Considering the lack of [a randomized controlled trial], our conclusion is that preoperative exposure to biologics is associated with an increased risk of pelvic sepsis after TPC with ileoanal pouch–anal anastomosis," commented lead investigator Dr. Jinyu Gu, a colorectal surgeon at the Cleveland Clinic. "This risk can be mitigated by the performance of initial STC."
The study’s senior investigator, Dr. P. Ravi Kiran, noted the importance of studying late complications in this population.
"We consciously decided to include patients who developed complications up to 1 year after surgery because quite often, these patients will not manifest their pelvic sepsis until the stoma is closed," he explained. "Some of the previous data from our institution have also shown that if someone were to get pelvic complications or septic complications that resemble Crohn’s disease, it is unlikely that it is really the disease that does it within 1 year after surgery; it is usually septic complications that manifest in a delayed fashion. ... A problem with any study that does not include patients for a prolonged follow-up is that you cannot really know what are the long-term complications because the presence of a stoma sometimes keeps the pelvic infection hidden."
"We, as colorectal surgeons, have been questioning what to do with patients who are on anti-TNF therapy when we operate on them," session comoderator Dr. Janice Rafferty, chief of the division of colon and rectal surgery at the University of Cincinnati, commented in an interview.
"I think this tells us that if we do a pouch procedure on them, and they are on anti-TNF therapy, their risk for pelvic sepsis is higher than if they had a three-stage procedure and we get them off of anti-TNF therapy," she said. "That probably supports what most colorectal surgeons suspect and want to do, but I think the gastroenterologists are currently pushing us, saying there is not a lot of evidence to say that they have a worse outcome."
Session comoderator Dr. Bruce Robb of Indiana University in Indianapolis agreed and noted that the findings support a recent shift toward multistage procedures in this population.
"For a long time, we were talking about two- versus one-stage procedures, and now we are going back, I think, especially with the advent of laparoscopy; people are much happier to do a three-stage procedure than they were even 10 years ago," he said. "This study sort of validates a change in practice pattern that’s already in place."
Dr. Gu’s team retrospectively assessed outcomes in 588 patients who underwent a restorative proctocolectomy for medically refractory ulcerative colitis between 2006 and 2010. Patients with complicated colitis, colitis-associated neoplasia, and Crohn’s disease were excluded.
The investigators assessed the rates of a variety of postoperative complications: pelvic sepsis, leaking of the colorectal stump, wound infection, postoperative hemorrhage, thromboembolism, urinary tract infection, and pneumonia.
Patients were defined as receiving anti-TNF therapy preoperatively if they had received at least 12 weeks of infliximab (Remicade) or at least 4 weeks of adalimumab (Humira) or certolizumab (Cimzia).
Of the 181 patients whose initial surgery was TPC with ileoanal pouch–anal anastomosis, 14% were receiving anti-TNF therapy preoperatively.
Within this group, the 30-day rate of complications did not differ significantly between patients who were and were not receiving preoperative anti-TNF therapy.
But the cumulative 1-year rate of pelvic sepsis was twice as high in patients receiving anti-TNF therapy (32% vs. 16%, P = .012). In adjusted analyses, these patients still had a more than doubling of the risk of pelvic sepsis (hazard ratio, 2.62; P = .027).
Of the 407 patients whose initial surgery was STC with end ileostomy, 35% were receiving anti-TNF agents preoperatively.
Within this group, patients taking anti-TNF agents preoperatively did not have an elevated risk of any of the complications studied at either 30 days or 1 year. The findings were similar among the subset who went on to have a completion proctectomy and ileoanal pouch–anal anastomosis.
Dr. Gu and Dr. Kiran both disclosed no relevant financial conflicts.
PHOENIX – Among patients with ulcerative colitis, preoperative therapy targeting tumor necrosis factor increases the risk of postoperative complications after two-stage restorative proctocolectomy procedures but not after three-stage ones, a study has shown.
A team at the Cleveland Clinic retrospectively assessed outcomes in more than 500 patients who underwent a restorative proctocolectomy for medically refractory ulcerative colitis during a recent 5-year period. Overall, 28% were receiving an agent that targets tumor necrosis factor (TNF) before their surgery.
The main results, reported at the annual meeting of the American Society of Colon and Rectal Surgeons, showed that among patients having an initial total proctocolectomy (TPC) with ileoanal pouch–anal anastomosis, preoperative anti-TNF therapy more than doubled the risk of pelvic sepsis in the subsequent year.
In contrast, among patients having an initial subtotal colectomy (STC) with end ileostomy, preoperative anti-TNF therapy did not significantly affect the risk of complications overall, or in the subset who went on to have completion proctectomy and ileoanal pouch–anal anastomosis.
"Considering the lack of [a randomized controlled trial], our conclusion is that preoperative exposure to biologics is associated with an increased risk of pelvic sepsis after TPC with ileoanal pouch–anal anastomosis," commented lead investigator Dr. Jinyu Gu, a colorectal surgeon at the Cleveland Clinic. "This risk can be mitigated by the performance of initial STC."
The study’s senior investigator, Dr. P. Ravi Kiran, noted the importance of studying late complications in this population.
"We consciously decided to include patients who developed complications up to 1 year after surgery because quite often, these patients will not manifest their pelvic sepsis until the stoma is closed," he explained. "Some of the previous data from our institution have also shown that if someone were to get pelvic complications or septic complications that resemble Crohn’s disease, it is unlikely that it is really the disease that does it within 1 year after surgery; it is usually septic complications that manifest in a delayed fashion. ... A problem with any study that does not include patients for a prolonged follow-up is that you cannot really know what are the long-term complications because the presence of a stoma sometimes keeps the pelvic infection hidden."
"We, as colorectal surgeons, have been questioning what to do with patients who are on anti-TNF therapy when we operate on them," session comoderator Dr. Janice Rafferty, chief of the division of colon and rectal surgery at the University of Cincinnati, commented in an interview.
"I think this tells us that if we do a pouch procedure on them, and they are on anti-TNF therapy, their risk for pelvic sepsis is higher than if they had a three-stage procedure and we get them off of anti-TNF therapy," she said. "That probably supports what most colorectal surgeons suspect and want to do, but I think the gastroenterologists are currently pushing us, saying there is not a lot of evidence to say that they have a worse outcome."
Session comoderator Dr. Bruce Robb of Indiana University in Indianapolis agreed and noted that the findings support a recent shift toward multistage procedures in this population.
"For a long time, we were talking about two- versus one-stage procedures, and now we are going back, I think, especially with the advent of laparoscopy; people are much happier to do a three-stage procedure than they were even 10 years ago," he said. "This study sort of validates a change in practice pattern that’s already in place."
Dr. Gu’s team retrospectively assessed outcomes in 588 patients who underwent a restorative proctocolectomy for medically refractory ulcerative colitis between 2006 and 2010. Patients with complicated colitis, colitis-associated neoplasia, and Crohn’s disease were excluded.
The investigators assessed the rates of a variety of postoperative complications: pelvic sepsis, leaking of the colorectal stump, wound infection, postoperative hemorrhage, thromboembolism, urinary tract infection, and pneumonia.
Patients were defined as receiving anti-TNF therapy preoperatively if they had received at least 12 weeks of infliximab (Remicade) or at least 4 weeks of adalimumab (Humira) or certolizumab (Cimzia).
Of the 181 patients whose initial surgery was TPC with ileoanal pouch–anal anastomosis, 14% were receiving anti-TNF therapy preoperatively.
Within this group, the 30-day rate of complications did not differ significantly between patients who were and were not receiving preoperative anti-TNF therapy.
But the cumulative 1-year rate of pelvic sepsis was twice as high in patients receiving anti-TNF therapy (32% vs. 16%, P = .012). In adjusted analyses, these patients still had a more than doubling of the risk of pelvic sepsis (hazard ratio, 2.62; P = .027).
Of the 407 patients whose initial surgery was STC with end ileostomy, 35% were receiving anti-TNF agents preoperatively.
Within this group, patients taking anti-TNF agents preoperatively did not have an elevated risk of any of the complications studied at either 30 days or 1 year. The findings were similar among the subset who went on to have a completion proctectomy and ileoanal pouch–anal anastomosis.
Dr. Gu and Dr. Kiran both disclosed no relevant financial conflicts.
PHOENIX – Among patients with ulcerative colitis, preoperative therapy targeting tumor necrosis factor increases the risk of postoperative complications after two-stage restorative proctocolectomy procedures but not after three-stage ones, a study has shown.
A team at the Cleveland Clinic retrospectively assessed outcomes in more than 500 patients who underwent a restorative proctocolectomy for medically refractory ulcerative colitis during a recent 5-year period. Overall, 28% were receiving an agent that targets tumor necrosis factor (TNF) before their surgery.
The main results, reported at the annual meeting of the American Society of Colon and Rectal Surgeons, showed that among patients having an initial total proctocolectomy (TPC) with ileoanal pouch–anal anastomosis, preoperative anti-TNF therapy more than doubled the risk of pelvic sepsis in the subsequent year.
In contrast, among patients having an initial subtotal colectomy (STC) with end ileostomy, preoperative anti-TNF therapy did not significantly affect the risk of complications overall, or in the subset who went on to have completion proctectomy and ileoanal pouch–anal anastomosis.
"Considering the lack of [a randomized controlled trial], our conclusion is that preoperative exposure to biologics is associated with an increased risk of pelvic sepsis after TPC with ileoanal pouch–anal anastomosis," commented lead investigator Dr. Jinyu Gu, a colorectal surgeon at the Cleveland Clinic. "This risk can be mitigated by the performance of initial STC."
The study’s senior investigator, Dr. P. Ravi Kiran, noted the importance of studying late complications in this population.
"We consciously decided to include patients who developed complications up to 1 year after surgery because quite often, these patients will not manifest their pelvic sepsis until the stoma is closed," he explained. "Some of the previous data from our institution have also shown that if someone were to get pelvic complications or septic complications that resemble Crohn’s disease, it is unlikely that it is really the disease that does it within 1 year after surgery; it is usually septic complications that manifest in a delayed fashion. ... A problem with any study that does not include patients for a prolonged follow-up is that you cannot really know what are the long-term complications because the presence of a stoma sometimes keeps the pelvic infection hidden."
"We, as colorectal surgeons, have been questioning what to do with patients who are on anti-TNF therapy when we operate on them," session comoderator Dr. Janice Rafferty, chief of the division of colon and rectal surgery at the University of Cincinnati, commented in an interview.
"I think this tells us that if we do a pouch procedure on them, and they are on anti-TNF therapy, their risk for pelvic sepsis is higher than if they had a three-stage procedure and we get them off of anti-TNF therapy," she said. "That probably supports what most colorectal surgeons suspect and want to do, but I think the gastroenterologists are currently pushing us, saying there is not a lot of evidence to say that they have a worse outcome."
Session comoderator Dr. Bruce Robb of Indiana University in Indianapolis agreed and noted that the findings support a recent shift toward multistage procedures in this population.
"For a long time, we were talking about two- versus one-stage procedures, and now we are going back, I think, especially with the advent of laparoscopy; people are much happier to do a three-stage procedure than they were even 10 years ago," he said. "This study sort of validates a change in practice pattern that’s already in place."
Dr. Gu’s team retrospectively assessed outcomes in 588 patients who underwent a restorative proctocolectomy for medically refractory ulcerative colitis between 2006 and 2010. Patients with complicated colitis, colitis-associated neoplasia, and Crohn’s disease were excluded.
The investigators assessed the rates of a variety of postoperative complications: pelvic sepsis, leaking of the colorectal stump, wound infection, postoperative hemorrhage, thromboembolism, urinary tract infection, and pneumonia.
Patients were defined as receiving anti-TNF therapy preoperatively if they had received at least 12 weeks of infliximab (Remicade) or at least 4 weeks of adalimumab (Humira) or certolizumab (Cimzia).
Of the 181 patients whose initial surgery was TPC with ileoanal pouch–anal anastomosis, 14% were receiving anti-TNF therapy preoperatively.
Within this group, the 30-day rate of complications did not differ significantly between patients who were and were not receiving preoperative anti-TNF therapy.
But the cumulative 1-year rate of pelvic sepsis was twice as high in patients receiving anti-TNF therapy (32% vs. 16%, P = .012). In adjusted analyses, these patients still had a more than doubling of the risk of pelvic sepsis (hazard ratio, 2.62; P = .027).
Of the 407 patients whose initial surgery was STC with end ileostomy, 35% were receiving anti-TNF agents preoperatively.
Within this group, patients taking anti-TNF agents preoperatively did not have an elevated risk of any of the complications studied at either 30 days or 1 year. The findings were similar among the subset who went on to have a completion proctectomy and ileoanal pouch–anal anastomosis.
Dr. Gu and Dr. Kiran both disclosed no relevant financial conflicts.
AT THE ASCRS ANNUAL MEETING
Major finding: Preoperative anti-TNF therapy increased the risk of sepsis after initial total proctocolectomy with ileoanal pouch–anal anastomosis (HR, 2.62). In contrast, it did not increase the risk of any complications after initial subtotal colectomy with end ileostomy.
Data source: A retrospective cohort study of 588 patients with ulcerative colitis
Disclosures: Dr. Gu and Dr. Kiran disclosed no relevant financial conflicts.
Colon cancer screening in African Americans
Data show that African Americans present with colon cancer at an earlier age than whites. But there's debate about the appropriate screening age. Should all African American patients be screened at age 40? Dr. Byron Cryer, professor of medicine at the University of Texas Southwestern Medical Center, discusses the pros and cons of earlier screening and shares his opinion on the issue.
Data show that African Americans present with colon cancer at an earlier age than whites. But there's debate about the appropriate screening age. Should all African American patients be screened at age 40? Dr. Byron Cryer, professor of medicine at the University of Texas Southwestern Medical Center, discusses the pros and cons of earlier screening and shares his opinion on the issue.
Data show that African Americans present with colon cancer at an earlier age than whites. But there's debate about the appropriate screening age. Should all African American patients be screened at age 40? Dr. Byron Cryer, professor of medicine at the University of Texas Southwestern Medical Center, discusses the pros and cons of earlier screening and shares his opinion on the issue.
Completion study key after incomplete colonoscopy
PHOENIX – When the entire colon cannot be examined during a colonoscopy, physicians should persevere in obtaining a full colonic evaluation because a sizable share of these patients will have an advanced polyp or cancer, a new study shows.
In the study, which used a prospective institutional database, about 1 in every 8 patients having an incomplete initial colonoscopy were subsequently found to have a clinically significant lesion after undergoing some other type of completion exam, lead investigator Dr. Timothy J. Ridolfi reported at the annual meeting of the American Society of Colon and Rectal Surgeons.
Patients with a personal history of polyps or cancer accounted for two-thirds of all patients who were found to have lesions on completion studies.
"Complete colonic evaluation in patients with an incomplete colonoscopy is important, and repeat colonoscopy is the most efficient way of achieving that," he commented. "Those with a personal history of polyps or cancer have the highest likelihood of a positive finding on repeat colonoscopy."
Session attendee Dr. Joshua Bleier, of the Hospital of the University of Pennsylvania, Philadelphia, asked, "Since you have this prospective database, was any chart review done to follow up on the patients who didn’t have completion evaluations?"
The investigators have not yet assessed outcomes among patients who were offered a completion study but skipped it, according to Dr. Ridolfi, who is a colorectal surgery fellow at the Cleveland Clinic. He noted that some patients may have had such studies done at outside institutions.
Commenting on the research in an interview, comoderator Dr. H. David Vargas, of the Ochsner Medical Center in New Orleans, said that although the study’s findings are not surprising, they are a good reminder to physicians of the need to follow through in cases where a colonoscopy cannot be completed.
Examining the entire colon "is really incumbent upon us. The onus is on us to complete it," he said. The study "confirms why we need to be thorough, if you will."
The investigators analyzed data from a prospective database that captured information on 25,645 colonoscopies performed by colorectal surgeons at the Cleveland Clinic between 1982 and 2009.
A total of 242 patients were identified as having an incomplete initial colonoscopy. The group had a mean age of 59 years, and 69% were women.
The leading reasons for an incomplete procedure were presence of stool, pain, and tortuosity, with the reason varying according to the anatomic extent of the procedure. Fully 71% of procedures were terminated at the splenic flexure or more distally, he reported.
Overall, 90% of the patients were offered some type of completion study, and 82% of this group complied and underwent the study.
The completion study was most commonly a barium enema (41%) or repeat colonoscopy (40%), and less commonly CT colonography (9%), colonoscopy under general anesthesia (5%), or indicated resection with intraoperative or perioperative colonoscopy (5%).
Some 12% of the patients undergoing a barium enema or CT colonography were found to have new abnormalities, according to Dr. Ridolfi.
In addition, 24% of the patients undergoing repeat colonoscopy were found to have polyps. Of the 32 polyps identified (21 located beyond the extent of the initial incomplete colonoscopy), 9 were advanced adenomas.
And 38% of the patients undergoing indicated resection were found to have some additional lesions on their intraoperative or perioperative colonoscopy.
Patients whose indication for the initial colonoscopy was a personal history of polyps or cancer were more likely than those with other indications – a family history, symptoms, or screening – to have lesions on their completion study. In fact, these patients made up 67% of all patients with positive findings.
Ultimately, 12% of the patients undergoing completion studies were determined to have clinically significant lesions that led to polypectomy or partial colectomy.
Dr. Ridolfi disclosed no relevant conflicts of interest.
PHOENIX – When the entire colon cannot be examined during a colonoscopy, physicians should persevere in obtaining a full colonic evaluation because a sizable share of these patients will have an advanced polyp or cancer, a new study shows.
In the study, which used a prospective institutional database, about 1 in every 8 patients having an incomplete initial colonoscopy were subsequently found to have a clinically significant lesion after undergoing some other type of completion exam, lead investigator Dr. Timothy J. Ridolfi reported at the annual meeting of the American Society of Colon and Rectal Surgeons.
Patients with a personal history of polyps or cancer accounted for two-thirds of all patients who were found to have lesions on completion studies.
"Complete colonic evaluation in patients with an incomplete colonoscopy is important, and repeat colonoscopy is the most efficient way of achieving that," he commented. "Those with a personal history of polyps or cancer have the highest likelihood of a positive finding on repeat colonoscopy."
Session attendee Dr. Joshua Bleier, of the Hospital of the University of Pennsylvania, Philadelphia, asked, "Since you have this prospective database, was any chart review done to follow up on the patients who didn’t have completion evaluations?"
The investigators have not yet assessed outcomes among patients who were offered a completion study but skipped it, according to Dr. Ridolfi, who is a colorectal surgery fellow at the Cleveland Clinic. He noted that some patients may have had such studies done at outside institutions.
Commenting on the research in an interview, comoderator Dr. H. David Vargas, of the Ochsner Medical Center in New Orleans, said that although the study’s findings are not surprising, they are a good reminder to physicians of the need to follow through in cases where a colonoscopy cannot be completed.
Examining the entire colon "is really incumbent upon us. The onus is on us to complete it," he said. The study "confirms why we need to be thorough, if you will."
The investigators analyzed data from a prospective database that captured information on 25,645 colonoscopies performed by colorectal surgeons at the Cleveland Clinic between 1982 and 2009.
A total of 242 patients were identified as having an incomplete initial colonoscopy. The group had a mean age of 59 years, and 69% were women.
The leading reasons for an incomplete procedure were presence of stool, pain, and tortuosity, with the reason varying according to the anatomic extent of the procedure. Fully 71% of procedures were terminated at the splenic flexure or more distally, he reported.
Overall, 90% of the patients were offered some type of completion study, and 82% of this group complied and underwent the study.
The completion study was most commonly a barium enema (41%) or repeat colonoscopy (40%), and less commonly CT colonography (9%), colonoscopy under general anesthesia (5%), or indicated resection with intraoperative or perioperative colonoscopy (5%).
Some 12% of the patients undergoing a barium enema or CT colonography were found to have new abnormalities, according to Dr. Ridolfi.
In addition, 24% of the patients undergoing repeat colonoscopy were found to have polyps. Of the 32 polyps identified (21 located beyond the extent of the initial incomplete colonoscopy), 9 were advanced adenomas.
And 38% of the patients undergoing indicated resection were found to have some additional lesions on their intraoperative or perioperative colonoscopy.
Patients whose indication for the initial colonoscopy was a personal history of polyps or cancer were more likely than those with other indications – a family history, symptoms, or screening – to have lesions on their completion study. In fact, these patients made up 67% of all patients with positive findings.
Ultimately, 12% of the patients undergoing completion studies were determined to have clinically significant lesions that led to polypectomy or partial colectomy.
Dr. Ridolfi disclosed no relevant conflicts of interest.
PHOENIX – When the entire colon cannot be examined during a colonoscopy, physicians should persevere in obtaining a full colonic evaluation because a sizable share of these patients will have an advanced polyp or cancer, a new study shows.
In the study, which used a prospective institutional database, about 1 in every 8 patients having an incomplete initial colonoscopy were subsequently found to have a clinically significant lesion after undergoing some other type of completion exam, lead investigator Dr. Timothy J. Ridolfi reported at the annual meeting of the American Society of Colon and Rectal Surgeons.
Patients with a personal history of polyps or cancer accounted for two-thirds of all patients who were found to have lesions on completion studies.
"Complete colonic evaluation in patients with an incomplete colonoscopy is important, and repeat colonoscopy is the most efficient way of achieving that," he commented. "Those with a personal history of polyps or cancer have the highest likelihood of a positive finding on repeat colonoscopy."
Session attendee Dr. Joshua Bleier, of the Hospital of the University of Pennsylvania, Philadelphia, asked, "Since you have this prospective database, was any chart review done to follow up on the patients who didn’t have completion evaluations?"
The investigators have not yet assessed outcomes among patients who were offered a completion study but skipped it, according to Dr. Ridolfi, who is a colorectal surgery fellow at the Cleveland Clinic. He noted that some patients may have had such studies done at outside institutions.
Commenting on the research in an interview, comoderator Dr. H. David Vargas, of the Ochsner Medical Center in New Orleans, said that although the study’s findings are not surprising, they are a good reminder to physicians of the need to follow through in cases where a colonoscopy cannot be completed.
Examining the entire colon "is really incumbent upon us. The onus is on us to complete it," he said. The study "confirms why we need to be thorough, if you will."
The investigators analyzed data from a prospective database that captured information on 25,645 colonoscopies performed by colorectal surgeons at the Cleveland Clinic between 1982 and 2009.
A total of 242 patients were identified as having an incomplete initial colonoscopy. The group had a mean age of 59 years, and 69% were women.
The leading reasons for an incomplete procedure were presence of stool, pain, and tortuosity, with the reason varying according to the anatomic extent of the procedure. Fully 71% of procedures were terminated at the splenic flexure or more distally, he reported.
Overall, 90% of the patients were offered some type of completion study, and 82% of this group complied and underwent the study.
The completion study was most commonly a barium enema (41%) or repeat colonoscopy (40%), and less commonly CT colonography (9%), colonoscopy under general anesthesia (5%), or indicated resection with intraoperative or perioperative colonoscopy (5%).
Some 12% of the patients undergoing a barium enema or CT colonography were found to have new abnormalities, according to Dr. Ridolfi.
In addition, 24% of the patients undergoing repeat colonoscopy were found to have polyps. Of the 32 polyps identified (21 located beyond the extent of the initial incomplete colonoscopy), 9 were advanced adenomas.
And 38% of the patients undergoing indicated resection were found to have some additional lesions on their intraoperative or perioperative colonoscopy.
Patients whose indication for the initial colonoscopy was a personal history of polyps or cancer were more likely than those with other indications – a family history, symptoms, or screening – to have lesions on their completion study. In fact, these patients made up 67% of all patients with positive findings.
Ultimately, 12% of the patients undergoing completion studies were determined to have clinically significant lesions that led to polypectomy or partial colectomy.
Dr. Ridolfi disclosed no relevant conflicts of interest.
AT THE ASCRS ANNUAL MEETING
Major finding: Overall, 12% of the patients were found to have clinically significant lesions when they underwent completion studies.
Data source: A cohort analysis of 242 patients with an incomplete initial colonoscopy.
Disclosures: Dr. Ridolfi disclosed no relevant conflicts of interest.
Mobile app guides patients through precolonoscopy bowel prep
A smartphone application that provides step-by-step instructions for precolonoscopy bowel preparation appears to improve users’ bowel preparation quality, according to early findings from an ongoing study.
The Boston Bowel Preparation Scale score in 16 patients who used the doctor-designed Arizona Digestive Health App was 8.19, which was significantly better than the scores in 162 smartphone users who did not use the app (6.92) and 164 patients without smartphones (6.76). The difference in scores between the smartphone users who didn’t use the app and those without a smartphone was not statistically significant. The findings are being reported during the annual Digestive Disease Week in Orlando.
In an initial phase of the study, the 362 non–app users who underwent screening colonoscopy were simply asked whether they owned a smartphone and about their likelihood of using an app. Bowel preparation quality in those patients was evaluated by a blinded endoscopist, Dr. Nilay H. Kavathia reported during a press conference prior to the meeting
In a second phase, patients were instructed on how to download the app, which includes timed alerts with step-by-step instructions regarding dietary restrictions and use of the prep, as well as photos and tips to ensure patients’ understanding of the process, said Dr. Kavathia of the Carl T. Hayden Veterans Affairs Medical Center in Phoenix.
The information was the same as that provided in written form to those not using the app, he noted.
Bowel preparation quality in these patients was also assessed by a blinded endoscopist.
Though early, the findings are encouraging, given that the success of colonoscopy is largely based on the quality of bowel preparation achieved, Dr. Kavathia said during the press briefing.
"Basically, good colon prep is difficult to do. Inadequate preparation can result in cancellation or a compromised exam ... We believe this app provides an important step in improving patient satisfaction and harnessing technology for better health care outcomes," he added.
Dr. Larry Friedman, Digestive Disease Week Council chair and chair of the department of medicine at Newton-Wellesley Hospital in Newton, Mass., agreed, calling the app "ingenious."
"There are a lot of barriers to success of colorectal cancer screening, and one of the main ones is ensuring that the preparation is adequate ... and everyone knows that the worst part of colonoscopy is taking the prep. Anything you can do to encourage patients to complete the prep, and perhaps enjoy doing it a little more than they generally do with the use of an app, I think that’s an ingenious approach that will improve the quality of colorectal cancer screening," he said.
The Arizona Digestive Health App is available for free for both Apple iPhone and Android devices, and is adaptable to multiple bowel preparations.
Dr. Kavathia reported having no relevant financial disclosures.
A smartphone application that provides step-by-step instructions for precolonoscopy bowel preparation appears to improve users’ bowel preparation quality, according to early findings from an ongoing study.
The Boston Bowel Preparation Scale score in 16 patients who used the doctor-designed Arizona Digestive Health App was 8.19, which was significantly better than the scores in 162 smartphone users who did not use the app (6.92) and 164 patients without smartphones (6.76). The difference in scores between the smartphone users who didn’t use the app and those without a smartphone was not statistically significant. The findings are being reported during the annual Digestive Disease Week in Orlando.
In an initial phase of the study, the 362 non–app users who underwent screening colonoscopy were simply asked whether they owned a smartphone and about their likelihood of using an app. Bowel preparation quality in those patients was evaluated by a blinded endoscopist, Dr. Nilay H. Kavathia reported during a press conference prior to the meeting
In a second phase, patients were instructed on how to download the app, which includes timed alerts with step-by-step instructions regarding dietary restrictions and use of the prep, as well as photos and tips to ensure patients’ understanding of the process, said Dr. Kavathia of the Carl T. Hayden Veterans Affairs Medical Center in Phoenix.
The information was the same as that provided in written form to those not using the app, he noted.
Bowel preparation quality in these patients was also assessed by a blinded endoscopist.
Though early, the findings are encouraging, given that the success of colonoscopy is largely based on the quality of bowel preparation achieved, Dr. Kavathia said during the press briefing.
"Basically, good colon prep is difficult to do. Inadequate preparation can result in cancellation or a compromised exam ... We believe this app provides an important step in improving patient satisfaction and harnessing technology for better health care outcomes," he added.
Dr. Larry Friedman, Digestive Disease Week Council chair and chair of the department of medicine at Newton-Wellesley Hospital in Newton, Mass., agreed, calling the app "ingenious."
"There are a lot of barriers to success of colorectal cancer screening, and one of the main ones is ensuring that the preparation is adequate ... and everyone knows that the worst part of colonoscopy is taking the prep. Anything you can do to encourage patients to complete the prep, and perhaps enjoy doing it a little more than they generally do with the use of an app, I think that’s an ingenious approach that will improve the quality of colorectal cancer screening," he said.
The Arizona Digestive Health App is available for free for both Apple iPhone and Android devices, and is adaptable to multiple bowel preparations.
Dr. Kavathia reported having no relevant financial disclosures.
A smartphone application that provides step-by-step instructions for precolonoscopy bowel preparation appears to improve users’ bowel preparation quality, according to early findings from an ongoing study.
The Boston Bowel Preparation Scale score in 16 patients who used the doctor-designed Arizona Digestive Health App was 8.19, which was significantly better than the scores in 162 smartphone users who did not use the app (6.92) and 164 patients without smartphones (6.76). The difference in scores between the smartphone users who didn’t use the app and those without a smartphone was not statistically significant. The findings are being reported during the annual Digestive Disease Week in Orlando.
In an initial phase of the study, the 362 non–app users who underwent screening colonoscopy were simply asked whether they owned a smartphone and about their likelihood of using an app. Bowel preparation quality in those patients was evaluated by a blinded endoscopist, Dr. Nilay H. Kavathia reported during a press conference prior to the meeting
In a second phase, patients were instructed on how to download the app, which includes timed alerts with step-by-step instructions regarding dietary restrictions and use of the prep, as well as photos and tips to ensure patients’ understanding of the process, said Dr. Kavathia of the Carl T. Hayden Veterans Affairs Medical Center in Phoenix.
The information was the same as that provided in written form to those not using the app, he noted.
Bowel preparation quality in these patients was also assessed by a blinded endoscopist.
Though early, the findings are encouraging, given that the success of colonoscopy is largely based on the quality of bowel preparation achieved, Dr. Kavathia said during the press briefing.
"Basically, good colon prep is difficult to do. Inadequate preparation can result in cancellation or a compromised exam ... We believe this app provides an important step in improving patient satisfaction and harnessing technology for better health care outcomes," he added.
Dr. Larry Friedman, Digestive Disease Week Council chair and chair of the department of medicine at Newton-Wellesley Hospital in Newton, Mass., agreed, calling the app "ingenious."
"There are a lot of barriers to success of colorectal cancer screening, and one of the main ones is ensuring that the preparation is adequate ... and everyone knows that the worst part of colonoscopy is taking the prep. Anything you can do to encourage patients to complete the prep, and perhaps enjoy doing it a little more than they generally do with the use of an app, I think that’s an ingenious approach that will improve the quality of colorectal cancer screening," he said.
The Arizona Digestive Health App is available for free for both Apple iPhone and Android devices, and is adaptable to multiple bowel preparations.
Dr. Kavathia reported having no relevant financial disclosures.
Major finding: Bowel prep scores were significantly better for app users (8.19) than for those who had smartphones (6.92) but did not use the app and those who did not have smartphones (6.76).
Data source: A prospective, assessor-blinded comparative study.
Disclosures: Dr. Kavathia reported having no relevant financial disclosures.
Elevated glucose after colorectal surgery spells trouble
INDIANAPOLIS – Even a single brief episode of postoperative hyperglycemia after colorectal resection in nondiabetic patients was independently associated with increased morbidity and mortality in a large consecutive patient series.
The risks of a variety of complications, both infectious and noninfectious, in nondiabetic patients with postoperative hyperglycemia were similar in magnitude to those seen in diabetic patients experiencing postoperative hyperglycemia.
"A take-home point from our study would be that since it’s known that in diabetic patients it’s absolutely paramount to control hyperglycemia, perhaps nondiabetic patients undergoing major operations – especially colorectal surgery – need to be carefully monitored and have their glucose managed," Dr. P. Ravi Kiran said at the annual meeting of the American Surgical Association.
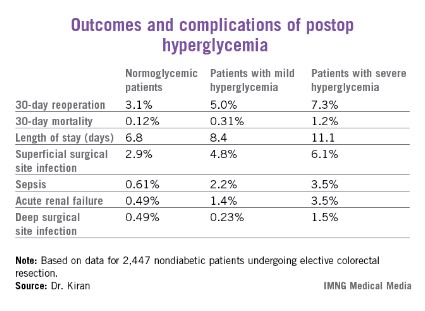
He and his coinvestigators evaluated the significance of hyperglycemia occurring within 48 hours postoperatively in 2,628 consecutive patients undergoing elective colorectal resection at the Cleveland Clinic in a recent 2-year period.
A total of 2,447 of these patients were nondiabetic. They collectively had 16,404 randomly obtained postoperative blood glucose measurements. One-third of them remained normoglycemic, with all their blood glucose values remaining at 125 mg/dL or less. Another 52.7% had one or more episodes of mild hyperglycemia as defined by a blood glucose value of 126-200 mg/dL. And 14% of nondiabetic subjects experienced postoperative severe hyperglycemia, with a level in excess of 200 mg/dL.
Those rates were similar to those of the known-diabetic patients, 35% of whom remained normoglycemic postoperatively, while 54% became mildly hyperglycemic and 11% severely hyperglycemic.
Postoperative hyperglycemia in nondiabetic patients was associated with greater intraoperative estimated blood loss. The transfusion rate was 4.8% in normoglycemic patients, 10.3% in mildly hyperglycemic ones, and 18.1% in those with severe hyperglycemia. The length of surgery averaged 137 minutes in normoglycemic patients, 166 minutes in patients with postoperative mild hyperglycemia, and 181 minutes in patients with severe hyperglycemia.
Any episode of postoperative severe hyperglycemia in the nondiabetic patients was associated with significantly higher rates of both superficial and deep surgical site infections, greater length of stay, and higher 30-day mortality, compared with normoglycemic patients (see chart). Mild hyperglycemia wasn’t associated with as many complications; however, it was linked to a significantly increased rate of sepsis and a greater length of stay.
Moreover, in a multivariate analysis mild hyperglycemia was independently associated with a 2.1-fold increased risk of reoperation within 30 days, while severe hyperglycemia carried a 3.8-fold increased risk, compared with normoglycemia, continued Dr. Kiran of the Cleveland Clinic.
The investigators identified two major independent risk factors for sepsis in nondiabetic patients: an American Society of Anesthesiologists Physical Status class of 3 or more (odds ratio, 4.2), and postoperative hyperglycemia, which was associated with roughly an 8-fold risk regardless of whether the hyperglycemia was mild or severe.
Studies from the cardiovascular and trauma surgery literature suggest postoperative uncontrolled blood glucose may lead to adverse outcomes. Dr. Kiran and his coworkers performed this study because the impact of elevated blood glucose after elective major abdominal surgery had not been well defined, although colorectal surgery entails bacterial contamination, so background rates of surgical site and other infectious complications already tend to run relatively high.
Dr. Kiran said he believes postoperative hyperglycemia is probably a surrogate marker, perhaps for a distressed physiologic state or for looming complications. The next major question he and his coinvestigators want to tackle is this: Do prompt recognition and management of postoperative hyperglycemia in nondiabetic colorectal resection patients improve outcomes?
A cautionary note was sounded by discussant Dr. Hiram C. Polk Jr.,who emphasized that management strategies involving tight glucose control entail the risk of potentially disastrous postsurgical hypoglycemia.
"As many of you know, there are more than half a dozen patients in the midwestern U.S. who’ve been rendered badly hurt with hypoglycemia and cerebral damage. They’re working their way through the legal system at this point. It’s a fine balance: Sometimes perfection is the enemy of good in this situation," warned Dr. Polk, professor and chairman of the surgery department at the University of Louisville (Ky.).
He noted as an aside that just as Dr. Kiran found that even a single episode of postoperative hyperglycemia has adverse consequences, he and his Louisville coworkers have found the same is true for hypothermia.
"A single brief episode of hypothermia seems to throw the wheels off the track. It disrupts something in host defenses and makes everything very difficult," Dr. Polk observed.
The study was sponsored by the Cleveland Clinic Foundation. Dr. Kiran reported no financial conflicts.
INDIANAPOLIS – Even a single brief episode of postoperative hyperglycemia after colorectal resection in nondiabetic patients was independently associated with increased morbidity and mortality in a large consecutive patient series.
The risks of a variety of complications, both infectious and noninfectious, in nondiabetic patients with postoperative hyperglycemia were similar in magnitude to those seen in diabetic patients experiencing postoperative hyperglycemia.
"A take-home point from our study would be that since it’s known that in diabetic patients it’s absolutely paramount to control hyperglycemia, perhaps nondiabetic patients undergoing major operations – especially colorectal surgery – need to be carefully monitored and have their glucose managed," Dr. P. Ravi Kiran said at the annual meeting of the American Surgical Association.

He and his coinvestigators evaluated the significance of hyperglycemia occurring within 48 hours postoperatively in 2,628 consecutive patients undergoing elective colorectal resection at the Cleveland Clinic in a recent 2-year period.
A total of 2,447 of these patients were nondiabetic. They collectively had 16,404 randomly obtained postoperative blood glucose measurements. One-third of them remained normoglycemic, with all their blood glucose values remaining at 125 mg/dL or less. Another 52.7% had one or more episodes of mild hyperglycemia as defined by a blood glucose value of 126-200 mg/dL. And 14% of nondiabetic subjects experienced postoperative severe hyperglycemia, with a level in excess of 200 mg/dL.
Those rates were similar to those of the known-diabetic patients, 35% of whom remained normoglycemic postoperatively, while 54% became mildly hyperglycemic and 11% severely hyperglycemic.
Postoperative hyperglycemia in nondiabetic patients was associated with greater intraoperative estimated blood loss. The transfusion rate was 4.8% in normoglycemic patients, 10.3% in mildly hyperglycemic ones, and 18.1% in those with severe hyperglycemia. The length of surgery averaged 137 minutes in normoglycemic patients, 166 minutes in patients with postoperative mild hyperglycemia, and 181 minutes in patients with severe hyperglycemia.
Any episode of postoperative severe hyperglycemia in the nondiabetic patients was associated with significantly higher rates of both superficial and deep surgical site infections, greater length of stay, and higher 30-day mortality, compared with normoglycemic patients (see chart). Mild hyperglycemia wasn’t associated with as many complications; however, it was linked to a significantly increased rate of sepsis and a greater length of stay.
Moreover, in a multivariate analysis mild hyperglycemia was independently associated with a 2.1-fold increased risk of reoperation within 30 days, while severe hyperglycemia carried a 3.8-fold increased risk, compared with normoglycemia, continued Dr. Kiran of the Cleveland Clinic.
The investigators identified two major independent risk factors for sepsis in nondiabetic patients: an American Society of Anesthesiologists Physical Status class of 3 or more (odds ratio, 4.2), and postoperative hyperglycemia, which was associated with roughly an 8-fold risk regardless of whether the hyperglycemia was mild or severe.
Studies from the cardiovascular and trauma surgery literature suggest postoperative uncontrolled blood glucose may lead to adverse outcomes. Dr. Kiran and his coworkers performed this study because the impact of elevated blood glucose after elective major abdominal surgery had not been well defined, although colorectal surgery entails bacterial contamination, so background rates of surgical site and other infectious complications already tend to run relatively high.
Dr. Kiran said he believes postoperative hyperglycemia is probably a surrogate marker, perhaps for a distressed physiologic state or for looming complications. The next major question he and his coinvestigators want to tackle is this: Do prompt recognition and management of postoperative hyperglycemia in nondiabetic colorectal resection patients improve outcomes?
A cautionary note was sounded by discussant Dr. Hiram C. Polk Jr.,who emphasized that management strategies involving tight glucose control entail the risk of potentially disastrous postsurgical hypoglycemia.
"As many of you know, there are more than half a dozen patients in the midwestern U.S. who’ve been rendered badly hurt with hypoglycemia and cerebral damage. They’re working their way through the legal system at this point. It’s a fine balance: Sometimes perfection is the enemy of good in this situation," warned Dr. Polk, professor and chairman of the surgery department at the University of Louisville (Ky.).
He noted as an aside that just as Dr. Kiran found that even a single episode of postoperative hyperglycemia has adverse consequences, he and his Louisville coworkers have found the same is true for hypothermia.
"A single brief episode of hypothermia seems to throw the wheels off the track. It disrupts something in host defenses and makes everything very difficult," Dr. Polk observed.
The study was sponsored by the Cleveland Clinic Foundation. Dr. Kiran reported no financial conflicts.
INDIANAPOLIS – Even a single brief episode of postoperative hyperglycemia after colorectal resection in nondiabetic patients was independently associated with increased morbidity and mortality in a large consecutive patient series.
The risks of a variety of complications, both infectious and noninfectious, in nondiabetic patients with postoperative hyperglycemia were similar in magnitude to those seen in diabetic patients experiencing postoperative hyperglycemia.
"A take-home point from our study would be that since it’s known that in diabetic patients it’s absolutely paramount to control hyperglycemia, perhaps nondiabetic patients undergoing major operations – especially colorectal surgery – need to be carefully monitored and have their glucose managed," Dr. P. Ravi Kiran said at the annual meeting of the American Surgical Association.

He and his coinvestigators evaluated the significance of hyperglycemia occurring within 48 hours postoperatively in 2,628 consecutive patients undergoing elective colorectal resection at the Cleveland Clinic in a recent 2-year period.
A total of 2,447 of these patients were nondiabetic. They collectively had 16,404 randomly obtained postoperative blood glucose measurements. One-third of them remained normoglycemic, with all their blood glucose values remaining at 125 mg/dL or less. Another 52.7% had one or more episodes of mild hyperglycemia as defined by a blood glucose value of 126-200 mg/dL. And 14% of nondiabetic subjects experienced postoperative severe hyperglycemia, with a level in excess of 200 mg/dL.
Those rates were similar to those of the known-diabetic patients, 35% of whom remained normoglycemic postoperatively, while 54% became mildly hyperglycemic and 11% severely hyperglycemic.
Postoperative hyperglycemia in nondiabetic patients was associated with greater intraoperative estimated blood loss. The transfusion rate was 4.8% in normoglycemic patients, 10.3% in mildly hyperglycemic ones, and 18.1% in those with severe hyperglycemia. The length of surgery averaged 137 minutes in normoglycemic patients, 166 minutes in patients with postoperative mild hyperglycemia, and 181 minutes in patients with severe hyperglycemia.
Any episode of postoperative severe hyperglycemia in the nondiabetic patients was associated with significantly higher rates of both superficial and deep surgical site infections, greater length of stay, and higher 30-day mortality, compared with normoglycemic patients (see chart). Mild hyperglycemia wasn’t associated with as many complications; however, it was linked to a significantly increased rate of sepsis and a greater length of stay.
Moreover, in a multivariate analysis mild hyperglycemia was independently associated with a 2.1-fold increased risk of reoperation within 30 days, while severe hyperglycemia carried a 3.8-fold increased risk, compared with normoglycemia, continued Dr. Kiran of the Cleveland Clinic.
The investigators identified two major independent risk factors for sepsis in nondiabetic patients: an American Society of Anesthesiologists Physical Status class of 3 or more (odds ratio, 4.2), and postoperative hyperglycemia, which was associated with roughly an 8-fold risk regardless of whether the hyperglycemia was mild or severe.
Studies from the cardiovascular and trauma surgery literature suggest postoperative uncontrolled blood glucose may lead to adverse outcomes. Dr. Kiran and his coworkers performed this study because the impact of elevated blood glucose after elective major abdominal surgery had not been well defined, although colorectal surgery entails bacterial contamination, so background rates of surgical site and other infectious complications already tend to run relatively high.
Dr. Kiran said he believes postoperative hyperglycemia is probably a surrogate marker, perhaps for a distressed physiologic state or for looming complications. The next major question he and his coinvestigators want to tackle is this: Do prompt recognition and management of postoperative hyperglycemia in nondiabetic colorectal resection patients improve outcomes?
A cautionary note was sounded by discussant Dr. Hiram C. Polk Jr.,who emphasized that management strategies involving tight glucose control entail the risk of potentially disastrous postsurgical hypoglycemia.
"As many of you know, there are more than half a dozen patients in the midwestern U.S. who’ve been rendered badly hurt with hypoglycemia and cerebral damage. They’re working their way through the legal system at this point. It’s a fine balance: Sometimes perfection is the enemy of good in this situation," warned Dr. Polk, professor and chairman of the surgery department at the University of Louisville (Ky.).
He noted as an aside that just as Dr. Kiran found that even a single episode of postoperative hyperglycemia has adverse consequences, he and his Louisville coworkers have found the same is true for hypothermia.
"A single brief episode of hypothermia seems to throw the wheels off the track. It disrupts something in host defenses and makes everything very difficult," Dr. Polk observed.
The study was sponsored by the Cleveland Clinic Foundation. Dr. Kiran reported no financial conflicts.
AT THE ASA ANNUAL MEETING
Major finding: Mild hyperglycemia occurring within 48 hours after major colorectal surgery in nondiabetic patients was independently associated with a doubled risk of reoperation within 30 days and a 7.9-fold increase in sepsis, compared with patients who remained normoglycemic. Severe hyperglycemia – a blood glucose measurement in excess of 200 mg/dL – was associated with even worse outcomes.
Data source: A single-center study of more than 16,000 postoperative blood glucose measurements in 2,628 consecutive patients who underwent colorectal resection.
Disclosures: The study was sponsored by the Cleveland Clinic Foundation. Dr. Kiran reported no financial conflicts.