User login
Study questions value of Surgical Care Improvement Project quality measure
INDIANAPOLIS – Intraoperative temperature proved unrelated to the risk of surgical site infection following major colorectal surgery in a large patient series.
This finding undercuts the rationale for normothermia as a process measure that’s part of the Surgical Care Improvement Project (SCIP) sponsored by CMS in partnership with the American College of Surgeons and other organizations.
"Our study suggests that perioperative normothermia is not independently associated in and of itself with reduced surgical site infections after colorectal surgery, and this as a process measure may have limited utility in actually decreasing SSIs. We believe that efforts in other areas may be more efficacious," Dr. Genevieve B. Melton-Meaux said at the annual meeting of the American Surgical Association.
She hastened to add that she and her coinvestigators are by no means saying intraoperative warming is unimportant. Indeed, there is compelling evidence that warming has physiologic benefit. Also, it has been shown that intraoperative hypothermia boosts SSI risk by about three-fold (N. Engl. J. Med. 1996; 334:1209-15). But the investigators take issue with the SCIP quality measure mandating documentation of a temperature of exactly 36° C at the end of a surgical case, given that their study demonstrated that this metric had no correlation with SSI rate.
"Our message and belief is that warming is a good thing and hypothermia is not a good thing. Warming is indeed something that should be done," emphasized Dr. Melton-Meaux, a colorectal surgeon at the University of Minnesota, Minneapolis.
She presented an analysis of continuously measured intraoperative temperature data recorded via anesthesia information system in 1,008 adults who underwent major colorectal procedures at the Cleveland Clinic during a recent 1-year period. Roughly two-thirds of the patients had either a partial colectomy, a proctectomy, or total abdominal colectomy. The mean operating time was 173 minutes, and 22% of patients had a laparoscopic approach. The anesthesia information system, Dr. Melton-Meaux observed, is a hitherto largely untapped rich data source for research, since it records temperature and other physiologic data throughout the operation.
Active rewarming was performed in 92% of cases. A total of 91% of patients received an antibiotic within 1 hour prior to incision, in accord with another SCIP performance measure. The mean and median intraoperative temperature was 36.0° C, with an ending temperature of 36.3° C.
The 30-day SSI rate was 17.4%, including an organ/space infection rate of 8.5%. Neither maximum, minimum, median, nor ending temperature differed significantly among patients who developed an SSI and those who didn’t. In a multivariate analysis, the only factors significantly associated with SSI risk were preoperative diabetes, which carried a 1.9-fold increased risk; laparoscopic approach, which was associated with a 41% reduction in risk; and estimated blood loss.
Discussant Dr. Mary T. Hawn characterized the temperature study as an indictment of SCIP.
"Colorectal surgery, as we all know, has been a major focus of the Surgical Care Improvement Project. Yet despite rapid adoption and standardization of some aspects of perioperative care, there is little if any evidence that any meaningful improvements in outcomes have been realized. And the evidence to support many of the SCIP metrics is limited. For instance, the evidence to support the use of prophylactic antibiotics is based upon extensive Level 1 data, but that data is on whether or not the patient received the antibiotic, not whether it was given within 60 minutes prior to incision," said Dr. Hawn of the University of Alabama at Birmingham.
She added that it’s incumbent upon surgeons themselves to develop the evidence for alternative metrics that more meaningfully measure true surgical quality.
"If you Google ‘SCIP normothermia measure,’ the first three sites that come up are companies selling these devices, so I think we need to study them," the surgeon said.
Other audience members decried the fact that hospitals are spending millions of dollars to be compliant with quality scorecards based in large part upon SCIP process measures of unproven value.
"Are we ready to recommend to CMS that they modify their indirect attempts to alter the practice of medicine by telling us exactly what we ought to do with temperature?" commented Dr. Kenneth L. Mattox, professor and vice chairman of the department of surgery at Baylor College of Medicine, Houston.
Dr. Melton-Meaux commented, "I think the intention behind the process measures is the right one: that we should be implementing system-wide best practices. But I think what has happened inadvertently, especially because SCIP has become part of value-based purchasing, is we are all playing a game. We are playing to the measure rather than really focusing on delivering better care and better outcomes."
Reducing surgical site infections has been a major focus of SCIP because they constitute the most common and costly complication of colorectal surgery. Moreover, SSI is the most powerful risk factor for readmission within 30 days.
Dr. Melton-Meaux reported having no financial conflicts.
INDIANAPOLIS – Intraoperative temperature proved unrelated to the risk of surgical site infection following major colorectal surgery in a large patient series.
This finding undercuts the rationale for normothermia as a process measure that’s part of the Surgical Care Improvement Project (SCIP) sponsored by CMS in partnership with the American College of Surgeons and other organizations.
"Our study suggests that perioperative normothermia is not independently associated in and of itself with reduced surgical site infections after colorectal surgery, and this as a process measure may have limited utility in actually decreasing SSIs. We believe that efforts in other areas may be more efficacious," Dr. Genevieve B. Melton-Meaux said at the annual meeting of the American Surgical Association.
She hastened to add that she and her coinvestigators are by no means saying intraoperative warming is unimportant. Indeed, there is compelling evidence that warming has physiologic benefit. Also, it has been shown that intraoperative hypothermia boosts SSI risk by about three-fold (N. Engl. J. Med. 1996; 334:1209-15). But the investigators take issue with the SCIP quality measure mandating documentation of a temperature of exactly 36° C at the end of a surgical case, given that their study demonstrated that this metric had no correlation with SSI rate.
"Our message and belief is that warming is a good thing and hypothermia is not a good thing. Warming is indeed something that should be done," emphasized Dr. Melton-Meaux, a colorectal surgeon at the University of Minnesota, Minneapolis.
She presented an analysis of continuously measured intraoperative temperature data recorded via anesthesia information system in 1,008 adults who underwent major colorectal procedures at the Cleveland Clinic during a recent 1-year period. Roughly two-thirds of the patients had either a partial colectomy, a proctectomy, or total abdominal colectomy. The mean operating time was 173 minutes, and 22% of patients had a laparoscopic approach. The anesthesia information system, Dr. Melton-Meaux observed, is a hitherto largely untapped rich data source for research, since it records temperature and other physiologic data throughout the operation.
Active rewarming was performed in 92% of cases. A total of 91% of patients received an antibiotic within 1 hour prior to incision, in accord with another SCIP performance measure. The mean and median intraoperative temperature was 36.0° C, with an ending temperature of 36.3° C.
The 30-day SSI rate was 17.4%, including an organ/space infection rate of 8.5%. Neither maximum, minimum, median, nor ending temperature differed significantly among patients who developed an SSI and those who didn’t. In a multivariate analysis, the only factors significantly associated with SSI risk were preoperative diabetes, which carried a 1.9-fold increased risk; laparoscopic approach, which was associated with a 41% reduction in risk; and estimated blood loss.
Discussant Dr. Mary T. Hawn characterized the temperature study as an indictment of SCIP.
"Colorectal surgery, as we all know, has been a major focus of the Surgical Care Improvement Project. Yet despite rapid adoption and standardization of some aspects of perioperative care, there is little if any evidence that any meaningful improvements in outcomes have been realized. And the evidence to support many of the SCIP metrics is limited. For instance, the evidence to support the use of prophylactic antibiotics is based upon extensive Level 1 data, but that data is on whether or not the patient received the antibiotic, not whether it was given within 60 minutes prior to incision," said Dr. Hawn of the University of Alabama at Birmingham.
She added that it’s incumbent upon surgeons themselves to develop the evidence for alternative metrics that more meaningfully measure true surgical quality.
"If you Google ‘SCIP normothermia measure,’ the first three sites that come up are companies selling these devices, so I think we need to study them," the surgeon said.
Other audience members decried the fact that hospitals are spending millions of dollars to be compliant with quality scorecards based in large part upon SCIP process measures of unproven value.
"Are we ready to recommend to CMS that they modify their indirect attempts to alter the practice of medicine by telling us exactly what we ought to do with temperature?" commented Dr. Kenneth L. Mattox, professor and vice chairman of the department of surgery at Baylor College of Medicine, Houston.
Dr. Melton-Meaux commented, "I think the intention behind the process measures is the right one: that we should be implementing system-wide best practices. But I think what has happened inadvertently, especially because SCIP has become part of value-based purchasing, is we are all playing a game. We are playing to the measure rather than really focusing on delivering better care and better outcomes."
Reducing surgical site infections has been a major focus of SCIP because they constitute the most common and costly complication of colorectal surgery. Moreover, SSI is the most powerful risk factor for readmission within 30 days.
Dr. Melton-Meaux reported having no financial conflicts.
INDIANAPOLIS – Intraoperative temperature proved unrelated to the risk of surgical site infection following major colorectal surgery in a large patient series.
This finding undercuts the rationale for normothermia as a process measure that’s part of the Surgical Care Improvement Project (SCIP) sponsored by CMS in partnership with the American College of Surgeons and other organizations.
"Our study suggests that perioperative normothermia is not independently associated in and of itself with reduced surgical site infections after colorectal surgery, and this as a process measure may have limited utility in actually decreasing SSIs. We believe that efforts in other areas may be more efficacious," Dr. Genevieve B. Melton-Meaux said at the annual meeting of the American Surgical Association.
She hastened to add that she and her coinvestigators are by no means saying intraoperative warming is unimportant. Indeed, there is compelling evidence that warming has physiologic benefit. Also, it has been shown that intraoperative hypothermia boosts SSI risk by about three-fold (N. Engl. J. Med. 1996; 334:1209-15). But the investigators take issue with the SCIP quality measure mandating documentation of a temperature of exactly 36° C at the end of a surgical case, given that their study demonstrated that this metric had no correlation with SSI rate.
"Our message and belief is that warming is a good thing and hypothermia is not a good thing. Warming is indeed something that should be done," emphasized Dr. Melton-Meaux, a colorectal surgeon at the University of Minnesota, Minneapolis.
She presented an analysis of continuously measured intraoperative temperature data recorded via anesthesia information system in 1,008 adults who underwent major colorectal procedures at the Cleveland Clinic during a recent 1-year period. Roughly two-thirds of the patients had either a partial colectomy, a proctectomy, or total abdominal colectomy. The mean operating time was 173 minutes, and 22% of patients had a laparoscopic approach. The anesthesia information system, Dr. Melton-Meaux observed, is a hitherto largely untapped rich data source for research, since it records temperature and other physiologic data throughout the operation.
Active rewarming was performed in 92% of cases. A total of 91% of patients received an antibiotic within 1 hour prior to incision, in accord with another SCIP performance measure. The mean and median intraoperative temperature was 36.0° C, with an ending temperature of 36.3° C.
The 30-day SSI rate was 17.4%, including an organ/space infection rate of 8.5%. Neither maximum, minimum, median, nor ending temperature differed significantly among patients who developed an SSI and those who didn’t. In a multivariate analysis, the only factors significantly associated with SSI risk were preoperative diabetes, which carried a 1.9-fold increased risk; laparoscopic approach, which was associated with a 41% reduction in risk; and estimated blood loss.
Discussant Dr. Mary T. Hawn characterized the temperature study as an indictment of SCIP.
"Colorectal surgery, as we all know, has been a major focus of the Surgical Care Improvement Project. Yet despite rapid adoption and standardization of some aspects of perioperative care, there is little if any evidence that any meaningful improvements in outcomes have been realized. And the evidence to support many of the SCIP metrics is limited. For instance, the evidence to support the use of prophylactic antibiotics is based upon extensive Level 1 data, but that data is on whether or not the patient received the antibiotic, not whether it was given within 60 minutes prior to incision," said Dr. Hawn of the University of Alabama at Birmingham.
She added that it’s incumbent upon surgeons themselves to develop the evidence for alternative metrics that more meaningfully measure true surgical quality.
"If you Google ‘SCIP normothermia measure,’ the first three sites that come up are companies selling these devices, so I think we need to study them," the surgeon said.
Other audience members decried the fact that hospitals are spending millions of dollars to be compliant with quality scorecards based in large part upon SCIP process measures of unproven value.
"Are we ready to recommend to CMS that they modify their indirect attempts to alter the practice of medicine by telling us exactly what we ought to do with temperature?" commented Dr. Kenneth L. Mattox, professor and vice chairman of the department of surgery at Baylor College of Medicine, Houston.
Dr. Melton-Meaux commented, "I think the intention behind the process measures is the right one: that we should be implementing system-wide best practices. But I think what has happened inadvertently, especially because SCIP has become part of value-based purchasing, is we are all playing a game. We are playing to the measure rather than really focusing on delivering better care and better outcomes."
Reducing surgical site infections has been a major focus of SCIP because they constitute the most common and costly complication of colorectal surgery. Moreover, SSI is the most powerful risk factor for readmission within 30 days.
Dr. Melton-Meaux reported having no financial conflicts.
AT THE ASA ANNUAL MEETING
Major Finding: Intraoperative temperatures in patients undergoing major colorectal surgery proved unrelated to surgical site infection risk.
Data Source: A retrospective study of continuous intraoperative temperature data measured via an anesthesia information system in 1,008 adults undergoing major colorectal procedures.
Disclosures: The study presenter reported having no financial conflicts.
Radical resection trumps local excision in stage I CRC
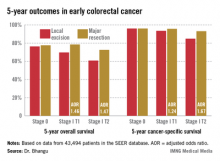
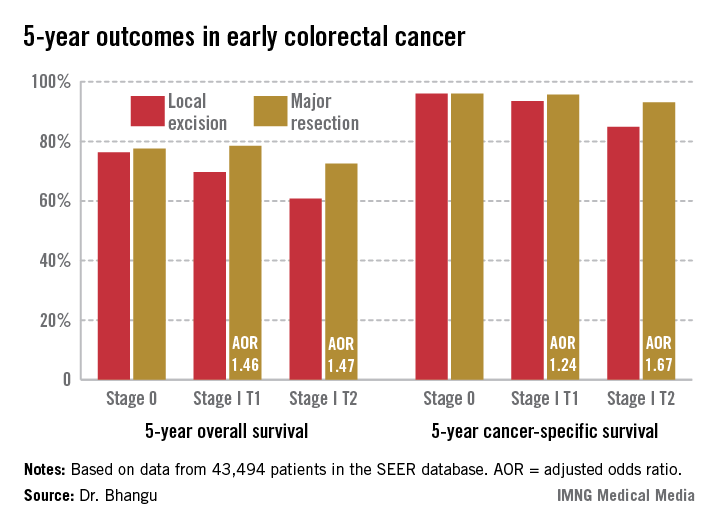
INDIANAPOLIS – Local excision of early invasive stage I colon or rectal carcinoma confers significantly worse 5-year overall and cancer-specific survival than does radical resection, according to analysis of a large national database.
This was true for stage I T1 and T2 disease; that is, for patients with tumor invading the submucosa as well as for those with tumor invading the muscularis propria, Dr. Aneel Bhangu reported at the annual meeting of the American Surgical Association.
In contrast, 5-year survival rates were equivalent with local excision compared with radical resection in patients with stage 0 disease, also known as carcinoma in situ, added Dr. Bhangu of Royal Marsden Hospital, London.
"We recommend that it is safe to perform local excision for stage 0 lesions – that is, carcinoma in situ or severely dysplastic polyps. Refined selection criteria for T1 cancers are required and should be the focus of further research. The use of local excision as a definitive treatment should be carefully considered for patients with T2 colorectal cancer, especially when treating younger, fit patients," he said.
The surgical oncologist presented an analysis of 43,494 patients with surgically treated stage 0 or I adenocarcinoma of the colon or rectum in the Surveillance, Epidemiology, and End Results (SEER) database for 1998-2009. He noted as an aside that the National Cancer Institute’s SEER database is "an open-access and free resource which is the envy of the worldwide oncological community and a shining example of how open-access data can be used by the global community to forward research."
Seventy percent of patients had colonic cancers, 30% rectal. Stage 0 cancer was present in 8.2%, while 91.8% of patients had stage I cancers, 51% of which were T1, 49% T2. Eighteen percent of subjects underwent local excision, while the rest had major resections.
Five-year overall survival was nearly an absolute 8% better in patients with stage I disease treated by radical resection, an advantage that grew even more dramatic in a multivariate analysis adjusted for age and other potential confounders.
Dr. Bhangu observed that these findings take on added import because the number of patients presenting with early colorectal cancer is climbing as a consequence of effective population-based colorectal cancer screening programs. The use of local excision as treatment for such cancers is growing as well. Yet the availability of high-tech tools and techniques for endoscopic local excision of these malignancies has created a dilemma: Such surgery spares the patient from a major operation with all its attendant hazards and morbidity, but when recurrences of these initially small cancers happen they may be inoperable in 10% of cases, and even when they are operable they require extensive visceral resection up to 50% of the time.
Discussant Dr. Genevieve Melton-Meaux of the University of Minnesota, Minneapolis, noted that a surprisingly large percentage of younger patients with stage I disease in this series – that is, patients under age 60 – were treated via local excision.
"We, too, were surprised by this," Dr. Bhangu replied. "If I were to speculate why, I’d say that it may be an issue of clinician equipoise. Some surgeons and endoscopists are true believers in this technology and they may be applying it to a wide scope of patients."
This same lack of equipoise explains the glacially slow recruitment rates into ongoing clinical trials badly needed to establish evidence-based therapy for early-stage colorectal cancer, he added.
He and his coworkers recommended directing future research in this field toward determining which patients with stage I disease are appropriate for local excision. Potentially relevant pathologic markers include depth of invasion and the degree of tumor differentiation. Biomarkers predictive of recurrence also are sorely needed.
Discussant Edward M. Copeland III noted that in light of Dr. Bhangu’s findings it makes sense to offer patients with T2 lesions neoadjuvant radiation and/or chemotherapy.
"You’d probably downstage a lot of them to T0 and you could then locally excise them, doing away with that 8% difference in survival between local excision and major resection you found in the SEER database," said Dr. Copeland, professor and chairman of the department of surgery at the University of Florida, Gainesville.
"I would venture to say if you had a patient with a T2 rectal lesion and offered neoadjuvant therapy followed by local excision, without a major operation, and with virtually zero chance of recurrence, I would take that, personally," he added.
Dr. Bhangu responded that it’s an intriguing notion, but the supporting evidence simply doesn’t exist. However, an ongoing U.K. randomized trial is evaluating just such an approach in patients with stage I T1 and T2 rectal cancer.
"I think this will provide the high-quality evidence that we require to treat these patients with evidence-based principles," he added.
Discussant Dr. Conor P. Delaney praised Dr. Bhangu for presenting "a great study." He placed the findings in perspective.
"It’s important to remember that the disadvantage that you’re showing with local therapy is very similar in size to the benefit that all of medical oncology gives us, with all of the effort that we invest in medical oncology. So this is actually a very significant result," declared Dr. Delaney, professor of surgery and chief of the division of colorectal surgery at Case Western Reserve University, Cleveland.
Dr. Bhangu reported having no financial conflicts.
INDIANAPOLIS – Local excision of early invasive stage I colon or rectal carcinoma confers significantly worse 5-year overall and cancer-specific survival than does radical resection, according to analysis of a large national database.
This was true for stage I T1 and T2 disease; that is, for patients with tumor invading the submucosa as well as for those with tumor invading the muscularis propria, Dr. Aneel Bhangu reported at the annual meeting of the American Surgical Association.
In contrast, 5-year survival rates were equivalent with local excision compared with radical resection in patients with stage 0 disease, also known as carcinoma in situ, added Dr. Bhangu of Royal Marsden Hospital, London.
"We recommend that it is safe to perform local excision for stage 0 lesions – that is, carcinoma in situ or severely dysplastic polyps. Refined selection criteria for T1 cancers are required and should be the focus of further research. The use of local excision as a definitive treatment should be carefully considered for patients with T2 colorectal cancer, especially when treating younger, fit patients," he said.
The surgical oncologist presented an analysis of 43,494 patients with surgically treated stage 0 or I adenocarcinoma of the colon or rectum in the Surveillance, Epidemiology, and End Results (SEER) database for 1998-2009. He noted as an aside that the National Cancer Institute’s SEER database is "an open-access and free resource which is the envy of the worldwide oncological community and a shining example of how open-access data can be used by the global community to forward research."
Seventy percent of patients had colonic cancers, 30% rectal. Stage 0 cancer was present in 8.2%, while 91.8% of patients had stage I cancers, 51% of which were T1, 49% T2. Eighteen percent of subjects underwent local excision, while the rest had major resections.
Five-year overall survival was nearly an absolute 8% better in patients with stage I disease treated by radical resection, an advantage that grew even more dramatic in a multivariate analysis adjusted for age and other potential confounders.
Dr. Bhangu observed that these findings take on added import because the number of patients presenting with early colorectal cancer is climbing as a consequence of effective population-based colorectal cancer screening programs. The use of local excision as treatment for such cancers is growing as well. Yet the availability of high-tech tools and techniques for endoscopic local excision of these malignancies has created a dilemma: Such surgery spares the patient from a major operation with all its attendant hazards and morbidity, but when recurrences of these initially small cancers happen they may be inoperable in 10% of cases, and even when they are operable they require extensive visceral resection up to 50% of the time.
Discussant Dr. Genevieve Melton-Meaux of the University of Minnesota, Minneapolis, noted that a surprisingly large percentage of younger patients with stage I disease in this series – that is, patients under age 60 – were treated via local excision.
"We, too, were surprised by this," Dr. Bhangu replied. "If I were to speculate why, I’d say that it may be an issue of clinician equipoise. Some surgeons and endoscopists are true believers in this technology and they may be applying it to a wide scope of patients."
This same lack of equipoise explains the glacially slow recruitment rates into ongoing clinical trials badly needed to establish evidence-based therapy for early-stage colorectal cancer, he added.
He and his coworkers recommended directing future research in this field toward determining which patients with stage I disease are appropriate for local excision. Potentially relevant pathologic markers include depth of invasion and the degree of tumor differentiation. Biomarkers predictive of recurrence also are sorely needed.
Discussant Edward M. Copeland III noted that in light of Dr. Bhangu’s findings it makes sense to offer patients with T2 lesions neoadjuvant radiation and/or chemotherapy.
"You’d probably downstage a lot of them to T0 and you could then locally excise them, doing away with that 8% difference in survival between local excision and major resection you found in the SEER database," said Dr. Copeland, professor and chairman of the department of surgery at the University of Florida, Gainesville.
"I would venture to say if you had a patient with a T2 rectal lesion and offered neoadjuvant therapy followed by local excision, without a major operation, and with virtually zero chance of recurrence, I would take that, personally," he added.
Dr. Bhangu responded that it’s an intriguing notion, but the supporting evidence simply doesn’t exist. However, an ongoing U.K. randomized trial is evaluating just such an approach in patients with stage I T1 and T2 rectal cancer.
"I think this will provide the high-quality evidence that we require to treat these patients with evidence-based principles," he added.
Discussant Dr. Conor P. Delaney praised Dr. Bhangu for presenting "a great study." He placed the findings in perspective.
"It’s important to remember that the disadvantage that you’re showing with local therapy is very similar in size to the benefit that all of medical oncology gives us, with all of the effort that we invest in medical oncology. So this is actually a very significant result," declared Dr. Delaney, professor of surgery and chief of the division of colorectal surgery at Case Western Reserve University, Cleveland.
Dr. Bhangu reported having no financial conflicts.
INDIANAPOLIS – Local excision of early invasive stage I colon or rectal carcinoma confers significantly worse 5-year overall and cancer-specific survival than does radical resection, according to analysis of a large national database.
This was true for stage I T1 and T2 disease; that is, for patients with tumor invading the submucosa as well as for those with tumor invading the muscularis propria, Dr. Aneel Bhangu reported at the annual meeting of the American Surgical Association.
In contrast, 5-year survival rates were equivalent with local excision compared with radical resection in patients with stage 0 disease, also known as carcinoma in situ, added Dr. Bhangu of Royal Marsden Hospital, London.
"We recommend that it is safe to perform local excision for stage 0 lesions – that is, carcinoma in situ or severely dysplastic polyps. Refined selection criteria for T1 cancers are required and should be the focus of further research. The use of local excision as a definitive treatment should be carefully considered for patients with T2 colorectal cancer, especially when treating younger, fit patients," he said.
The surgical oncologist presented an analysis of 43,494 patients with surgically treated stage 0 or I adenocarcinoma of the colon or rectum in the Surveillance, Epidemiology, and End Results (SEER) database for 1998-2009. He noted as an aside that the National Cancer Institute’s SEER database is "an open-access and free resource which is the envy of the worldwide oncological community and a shining example of how open-access data can be used by the global community to forward research."
Seventy percent of patients had colonic cancers, 30% rectal. Stage 0 cancer was present in 8.2%, while 91.8% of patients had stage I cancers, 51% of which were T1, 49% T2. Eighteen percent of subjects underwent local excision, while the rest had major resections.
Five-year overall survival was nearly an absolute 8% better in patients with stage I disease treated by radical resection, an advantage that grew even more dramatic in a multivariate analysis adjusted for age and other potential confounders.
Dr. Bhangu observed that these findings take on added import because the number of patients presenting with early colorectal cancer is climbing as a consequence of effective population-based colorectal cancer screening programs. The use of local excision as treatment for such cancers is growing as well. Yet the availability of high-tech tools and techniques for endoscopic local excision of these malignancies has created a dilemma: Such surgery spares the patient from a major operation with all its attendant hazards and morbidity, but when recurrences of these initially small cancers happen they may be inoperable in 10% of cases, and even when they are operable they require extensive visceral resection up to 50% of the time.
Discussant Dr. Genevieve Melton-Meaux of the University of Minnesota, Minneapolis, noted that a surprisingly large percentage of younger patients with stage I disease in this series – that is, patients under age 60 – were treated via local excision.
"We, too, were surprised by this," Dr. Bhangu replied. "If I were to speculate why, I’d say that it may be an issue of clinician equipoise. Some surgeons and endoscopists are true believers in this technology and they may be applying it to a wide scope of patients."
This same lack of equipoise explains the glacially slow recruitment rates into ongoing clinical trials badly needed to establish evidence-based therapy for early-stage colorectal cancer, he added.
He and his coworkers recommended directing future research in this field toward determining which patients with stage I disease are appropriate for local excision. Potentially relevant pathologic markers include depth of invasion and the degree of tumor differentiation. Biomarkers predictive of recurrence also are sorely needed.
Discussant Edward M. Copeland III noted that in light of Dr. Bhangu’s findings it makes sense to offer patients with T2 lesions neoadjuvant radiation and/or chemotherapy.
"You’d probably downstage a lot of them to T0 and you could then locally excise them, doing away with that 8% difference in survival between local excision and major resection you found in the SEER database," said Dr. Copeland, professor and chairman of the department of surgery at the University of Florida, Gainesville.
"I would venture to say if you had a patient with a T2 rectal lesion and offered neoadjuvant therapy followed by local excision, without a major operation, and with virtually zero chance of recurrence, I would take that, personally," he added.
Dr. Bhangu responded that it’s an intriguing notion, but the supporting evidence simply doesn’t exist. However, an ongoing U.K. randomized trial is evaluating just such an approach in patients with stage I T1 and T2 rectal cancer.
"I think this will provide the high-quality evidence that we require to treat these patients with evidence-based principles," he added.
Discussant Dr. Conor P. Delaney praised Dr. Bhangu for presenting "a great study." He placed the findings in perspective.
"It’s important to remember that the disadvantage that you’re showing with local therapy is very similar in size to the benefit that all of medical oncology gives us, with all of the effort that we invest in medical oncology. So this is actually a very significant result," declared Dr. Delaney, professor of surgery and chief of the division of colorectal surgery at Case Western Reserve University, Cleveland.
Dr. Bhangu reported having no financial conflicts.
AT THE ASA ANNUAL MEETING
Major Finding: Five-year overall survival in patients with stage I colorectal carcinoma treated via local excision was 68.4% compared to 75.2% with radical resection. Survival rates did not differ between the two surgical strategies in patients with stage 0 cancer.
Data Source: A retrospective analysis of data on nearly 44,000 patients with stage 0 or I colorectal cancer in the SEER database.
Disclosures: The SEER database is sponsored by the National Cancer Institute. The study presenter reported having no financial conflicts.
Adjuvant therapy selection in gastric cancer still more art than science
NATIONAL HARBOR, MD. – There are no hard and fast rules when it comes to selecting neoadjuvant or adjuvant therapy for patients with gastric cancer, according to Dr. Daniel G. Coit.
"Clinical trials of adjuvant therapy describe the impact of treatment on one group relative to another group. They do not predict the impact of treatment on an individual patient," said Dr. Coit, a surgical oncologist at Memorial Sloan-Kettering Cancer Center, New York.
Patient selection is a multi-step process that must incorporate an understanding of the risk of recurrence, the absolute benefit of treatment, strategies to improve the odds for achieving a benefit, and individual patient comorbidities and preferences, Dr. Coit said at the annual Society of Surgical Oncology Cancer Symposium.
Where oncologists often run into trouble is in the interpretation of data on benefits from clinical trials, and on the question of neoadjuvant vs. adjuvant therapy, both, or neither, he said.
Therapy delivered prior to surgery is likely to be better tolerated and improve the chance of R0 or "clean" resections. Neoadjuvant therapy can also help to identify, before surgery, those patients unlikely to respond metabolically or histologically to therapy, which can serve as both a prognostic factor for poor outcomes, and as a guide for additional pre- or postoperative therapy, Dr. Coit said.
Adjuvant therapy, on the other hand, allows treatment decisions to be based on an accurate estimate of recurrence risk using postoperative pathologic staging, he added.
"If we’re going to even consider preoperative therapy, neoadjuvant chemotherapy, we need to have some sense for how good our preoperative or pretreatment risk-assessment tools are. They’re not all that good," he said.
He noted that in a 2001 study (J. Clin. Gastroenterol. 2001;32:41-4) of 549 patients who were tested for carcinoembryonic antigen (CEA) and cancer antigen 19-9 (CA-19-9) before gastrectomy for gastric cancer, elevated levels of both markers were associated with "increased depth of invasion, more extensive nodal metastases, liver metastases, and less ‘curability.’ "
However, the authors found on multivariate analysis that a doubling of the normal threshold CEA was the only preoperative factor predictive of prognosis.
Endoscopic ultrasound (EUS), which is about 75% accurate at identifying node-positive vs. node-negative disease and at distinguishing early- from late-stage disease, is "very poorly predictive of outcome."
"The same thing happens with N stage: the endoscopic observation of node positivity is a terrible predictor of outcome," Dr. Coit said.
There are a few published nomograms that are fairly good at predicting disease-specific survival after R0 resections for gastric cancer, he noted, but these are all based on postoperative pathology findings, and "as yet we do not have a good nomogram for preoperative risk assessment."
Preventing recurrence
The goal of current therapy is to prevent recurrence at all costs, because few patients who experience recurrence will be salvageable, Dr. Coit said.
Studies of adjuvant therapy for gastric cancer with either postoperative 5-fluorauracil (5FU) and radiation (N. Engl. J. Med. 2001;345:725-30), postoperative epirubicin, cisplatin, and continuous 5FU infusion (ECF) (N. Engl. J. Med. 2006;355:11-20), or the oral fluoropyrimidine agent S-1, which is not available in the United States (N. Engl. J. Med. 2007:357:1810-20) all show about a 10% benefit, meaning that 10 patients would need to be treated for 1 to benefit.
More recently, in the CLASSIC trial (Lancet 2012;379:315-21) 1,035 patients with stage II-III gastric cancer who underwent curative resections with D2 extended lymph node dissection who were randomized to adjuvant capecitabine (Xeloda) and oxaliplatin had a 74% 3-year disease-free survival rate, compared with 59% for patients managed with postoperative observation only (hazard ratio 0.56, P less than .0001).
This trial showed that if patients are determined to have high-risk disease on postoperative pathology, "you have a very legitimate option available to you for postoperative treatment," Dr. Coit said.
In the ARTIST trial (J. Clin. Oncol. 2011;30:268-73), 458 patients underwent curative surgery with D2 lymph node dissection and were randomized to postoperative Xeloda/cisplatin (XP) or XP plus concurrent capecitabine radio therapy. The researchers found no statistically significant difference in 3-year disease-free survival among all patients, although there was some evidence of a benefit for XP plus radiation in patients with node-positive disease (P = .04).
Enriching the population
"It’s very clear that clinical trials do show a modest improvement in survival among the group of patients receiving adjuvant chemotherapy. The adjuvant therapy has no impact whatsoever in virtually 90% of the patients who are being treated, and we need to enrich this population that’s likely to benefit," he stated.
One strategy available for better selection of patients for adjuvant therapy is genetic tumor profiling to indentify targetable mutations such as HER-2 which is overexpressed in some gastric cancers.
A promising approach for identifying patients who might benefit from neoadjuvant therapy involves tumor uptake of fluorodeoxyglucose on positron-emission tomography (FDG-PET). In the MUNICON phase II trial (Lancet Oncol 2007;8:797-805), patients deemed to be responders, defined by decreases in tumor glucose standard uptake values (SUVs), had a median event-free survival of 29.7 months compared with 14.1 months in nonresponders (hazard ratio 2.18, P = .002), and 29 of 29 of 50 metabolic responders (58% ) had major histologic remissions, whereas none of the metabolic nonresponders did.
Dr. Coit reported having no financial disclosures.
NATIONAL HARBOR, MD. – There are no hard and fast rules when it comes to selecting neoadjuvant or adjuvant therapy for patients with gastric cancer, according to Dr. Daniel G. Coit.
"Clinical trials of adjuvant therapy describe the impact of treatment on one group relative to another group. They do not predict the impact of treatment on an individual patient," said Dr. Coit, a surgical oncologist at Memorial Sloan-Kettering Cancer Center, New York.
Patient selection is a multi-step process that must incorporate an understanding of the risk of recurrence, the absolute benefit of treatment, strategies to improve the odds for achieving a benefit, and individual patient comorbidities and preferences, Dr. Coit said at the annual Society of Surgical Oncology Cancer Symposium.
Where oncologists often run into trouble is in the interpretation of data on benefits from clinical trials, and on the question of neoadjuvant vs. adjuvant therapy, both, or neither, he said.
Therapy delivered prior to surgery is likely to be better tolerated and improve the chance of R0 or "clean" resections. Neoadjuvant therapy can also help to identify, before surgery, those patients unlikely to respond metabolically or histologically to therapy, which can serve as both a prognostic factor for poor outcomes, and as a guide for additional pre- or postoperative therapy, Dr. Coit said.
Adjuvant therapy, on the other hand, allows treatment decisions to be based on an accurate estimate of recurrence risk using postoperative pathologic staging, he added.
"If we’re going to even consider preoperative therapy, neoadjuvant chemotherapy, we need to have some sense for how good our preoperative or pretreatment risk-assessment tools are. They’re not all that good," he said.
He noted that in a 2001 study (J. Clin. Gastroenterol. 2001;32:41-4) of 549 patients who were tested for carcinoembryonic antigen (CEA) and cancer antigen 19-9 (CA-19-9) before gastrectomy for gastric cancer, elevated levels of both markers were associated with "increased depth of invasion, more extensive nodal metastases, liver metastases, and less ‘curability.’ "
However, the authors found on multivariate analysis that a doubling of the normal threshold CEA was the only preoperative factor predictive of prognosis.
Endoscopic ultrasound (EUS), which is about 75% accurate at identifying node-positive vs. node-negative disease and at distinguishing early- from late-stage disease, is "very poorly predictive of outcome."
"The same thing happens with N stage: the endoscopic observation of node positivity is a terrible predictor of outcome," Dr. Coit said.
There are a few published nomograms that are fairly good at predicting disease-specific survival after R0 resections for gastric cancer, he noted, but these are all based on postoperative pathology findings, and "as yet we do not have a good nomogram for preoperative risk assessment."
Preventing recurrence
The goal of current therapy is to prevent recurrence at all costs, because few patients who experience recurrence will be salvageable, Dr. Coit said.
Studies of adjuvant therapy for gastric cancer with either postoperative 5-fluorauracil (5FU) and radiation (N. Engl. J. Med. 2001;345:725-30), postoperative epirubicin, cisplatin, and continuous 5FU infusion (ECF) (N. Engl. J. Med. 2006;355:11-20), or the oral fluoropyrimidine agent S-1, which is not available in the United States (N. Engl. J. Med. 2007:357:1810-20) all show about a 10% benefit, meaning that 10 patients would need to be treated for 1 to benefit.
More recently, in the CLASSIC trial (Lancet 2012;379:315-21) 1,035 patients with stage II-III gastric cancer who underwent curative resections with D2 extended lymph node dissection who were randomized to adjuvant capecitabine (Xeloda) and oxaliplatin had a 74% 3-year disease-free survival rate, compared with 59% for patients managed with postoperative observation only (hazard ratio 0.56, P less than .0001).
This trial showed that if patients are determined to have high-risk disease on postoperative pathology, "you have a very legitimate option available to you for postoperative treatment," Dr. Coit said.
In the ARTIST trial (J. Clin. Oncol. 2011;30:268-73), 458 patients underwent curative surgery with D2 lymph node dissection and were randomized to postoperative Xeloda/cisplatin (XP) or XP plus concurrent capecitabine radio therapy. The researchers found no statistically significant difference in 3-year disease-free survival among all patients, although there was some evidence of a benefit for XP plus radiation in patients with node-positive disease (P = .04).
Enriching the population
"It’s very clear that clinical trials do show a modest improvement in survival among the group of patients receiving adjuvant chemotherapy. The adjuvant therapy has no impact whatsoever in virtually 90% of the patients who are being treated, and we need to enrich this population that’s likely to benefit," he stated.
One strategy available for better selection of patients for adjuvant therapy is genetic tumor profiling to indentify targetable mutations such as HER-2 which is overexpressed in some gastric cancers.
A promising approach for identifying patients who might benefit from neoadjuvant therapy involves tumor uptake of fluorodeoxyglucose on positron-emission tomography (FDG-PET). In the MUNICON phase II trial (Lancet Oncol 2007;8:797-805), patients deemed to be responders, defined by decreases in tumor glucose standard uptake values (SUVs), had a median event-free survival of 29.7 months compared with 14.1 months in nonresponders (hazard ratio 2.18, P = .002), and 29 of 29 of 50 metabolic responders (58% ) had major histologic remissions, whereas none of the metabolic nonresponders did.
Dr. Coit reported having no financial disclosures.
NATIONAL HARBOR, MD. – There are no hard and fast rules when it comes to selecting neoadjuvant or adjuvant therapy for patients with gastric cancer, according to Dr. Daniel G. Coit.
"Clinical trials of adjuvant therapy describe the impact of treatment on one group relative to another group. They do not predict the impact of treatment on an individual patient," said Dr. Coit, a surgical oncologist at Memorial Sloan-Kettering Cancer Center, New York.
Patient selection is a multi-step process that must incorporate an understanding of the risk of recurrence, the absolute benefit of treatment, strategies to improve the odds for achieving a benefit, and individual patient comorbidities and preferences, Dr. Coit said at the annual Society of Surgical Oncology Cancer Symposium.
Where oncologists often run into trouble is in the interpretation of data on benefits from clinical trials, and on the question of neoadjuvant vs. adjuvant therapy, both, or neither, he said.
Therapy delivered prior to surgery is likely to be better tolerated and improve the chance of R0 or "clean" resections. Neoadjuvant therapy can also help to identify, before surgery, those patients unlikely to respond metabolically or histologically to therapy, which can serve as both a prognostic factor for poor outcomes, and as a guide for additional pre- or postoperative therapy, Dr. Coit said.
Adjuvant therapy, on the other hand, allows treatment decisions to be based on an accurate estimate of recurrence risk using postoperative pathologic staging, he added.
"If we’re going to even consider preoperative therapy, neoadjuvant chemotherapy, we need to have some sense for how good our preoperative or pretreatment risk-assessment tools are. They’re not all that good," he said.
He noted that in a 2001 study (J. Clin. Gastroenterol. 2001;32:41-4) of 549 patients who were tested for carcinoembryonic antigen (CEA) and cancer antigen 19-9 (CA-19-9) before gastrectomy for gastric cancer, elevated levels of both markers were associated with "increased depth of invasion, more extensive nodal metastases, liver metastases, and less ‘curability.’ "
However, the authors found on multivariate analysis that a doubling of the normal threshold CEA was the only preoperative factor predictive of prognosis.
Endoscopic ultrasound (EUS), which is about 75% accurate at identifying node-positive vs. node-negative disease and at distinguishing early- from late-stage disease, is "very poorly predictive of outcome."
"The same thing happens with N stage: the endoscopic observation of node positivity is a terrible predictor of outcome," Dr. Coit said.
There are a few published nomograms that are fairly good at predicting disease-specific survival after R0 resections for gastric cancer, he noted, but these are all based on postoperative pathology findings, and "as yet we do not have a good nomogram for preoperative risk assessment."
Preventing recurrence
The goal of current therapy is to prevent recurrence at all costs, because few patients who experience recurrence will be salvageable, Dr. Coit said.
Studies of adjuvant therapy for gastric cancer with either postoperative 5-fluorauracil (5FU) and radiation (N. Engl. J. Med. 2001;345:725-30), postoperative epirubicin, cisplatin, and continuous 5FU infusion (ECF) (N. Engl. J. Med. 2006;355:11-20), or the oral fluoropyrimidine agent S-1, which is not available in the United States (N. Engl. J. Med. 2007:357:1810-20) all show about a 10% benefit, meaning that 10 patients would need to be treated for 1 to benefit.
More recently, in the CLASSIC trial (Lancet 2012;379:315-21) 1,035 patients with stage II-III gastric cancer who underwent curative resections with D2 extended lymph node dissection who were randomized to adjuvant capecitabine (Xeloda) and oxaliplatin had a 74% 3-year disease-free survival rate, compared with 59% for patients managed with postoperative observation only (hazard ratio 0.56, P less than .0001).
This trial showed that if patients are determined to have high-risk disease on postoperative pathology, "you have a very legitimate option available to you for postoperative treatment," Dr. Coit said.
In the ARTIST trial (J. Clin. Oncol. 2011;30:268-73), 458 patients underwent curative surgery with D2 lymph node dissection and were randomized to postoperative Xeloda/cisplatin (XP) or XP plus concurrent capecitabine radio therapy. The researchers found no statistically significant difference in 3-year disease-free survival among all patients, although there was some evidence of a benefit for XP plus radiation in patients with node-positive disease (P = .04).
Enriching the population
"It’s very clear that clinical trials do show a modest improvement in survival among the group of patients receiving adjuvant chemotherapy. The adjuvant therapy has no impact whatsoever in virtually 90% of the patients who are being treated, and we need to enrich this population that’s likely to benefit," he stated.
One strategy available for better selection of patients for adjuvant therapy is genetic tumor profiling to indentify targetable mutations such as HER-2 which is overexpressed in some gastric cancers.
A promising approach for identifying patients who might benefit from neoadjuvant therapy involves tumor uptake of fluorodeoxyglucose on positron-emission tomography (FDG-PET). In the MUNICON phase II trial (Lancet Oncol 2007;8:797-805), patients deemed to be responders, defined by decreases in tumor glucose standard uptake values (SUVs), had a median event-free survival of 29.7 months compared with 14.1 months in nonresponders (hazard ratio 2.18, P = .002), and 29 of 29 of 50 metabolic responders (58% ) had major histologic remissions, whereas none of the metabolic nonresponders did.
Dr. Coit reported having no financial disclosures.
AT SSO 2013
Surgery may be avoided in early rectal cancer
NATIONAL HARBOR, MD. – It may sound like heresy, but select patients with locally advanced rectal cancer may be spared surgery and its associated complications, a cancer surgeon suggested at the annual Society of Surgical Oncology Cancer Symposium.
Approximately 10%-25% of patients with locally advanced rectal cancer will have clinical complete responses (cCR) to neoadjuvant chemotherapy and radiation, said Dr. Philip B. Paty, an attending surgeon in the colorectal surgery service at Memorial Sloan-Kettering Cancer Center in New York City.
"The vast majority of these patients will avoid rectal resection, at least within the first 5 years," Dr. Paty added.
Although local failure occurs in 10%-25% of patients, most of the failures occur within the first 18 months, and most of these cases can be salvaged with R0 resections. Patients treated with nonoperative management appear to have rates of distant recurrence and survival similar to those of patients with pathologic complete responses (pCR) treated with total mesorectal resection, he said.
If surgery is required, local excision may be sufficient for some patients with stage T1 lesions and a select few with T2 lesions, said Dr. Heidi Nelson, professor of surgery in the department of colon and rectal surgery at the Mayo Clinic in Rochester, Minn.
If a patient has a favorable T1 lesion and would otherwise face a life-altering procedure such as abdominal perineal resection (APR) and colostomy, the surgeon should at least show the patient the data and discuss local excision as a safe and effective alternative with results comparable to more extensive resections, she said.
T2 lesions are more problematic, but a select few patients with this tumor type might be spared the morbidity of standard rectal resection, she added.
Hold the surgery?
Dr. Paty noted that, with standard management of stage T3 or T4 rectal cancers, the combination of neoadjuvant chemoradiotherapy, surgery, and adjuvant chemotherapy resulted in a 76% overall survival rate with less than 0.3% local recurrence after 5 years (Ann. Surg. 2005;241:829-36).
"What we have not dwelt on much is the morbidity of surgery, which is very significant. Having a rectal resection is a life-changing event for every patient that has one. Surgeons know that, and patients know that even better than surgeons," he said.
Rectal resections are associated with significant perioperative morbidity, colostomy, altered bowel function, sexual dysfunction, and infertility, he noted.
Pathologic complete responses to neoadjuvant therapy occur in 10%-44% of patients, and patients who have a pCR have markedly better oncologic outcomes than patients with less robust responses.
Of course, pCR can only be determined after surgery, raising the question of whether a clinical CR is sufficient for determining whether a patient might be spared rectal resection.
There are currently more data on pCR in rectal cancer than cCR, "probably because clinical CR criteria right now are quite stringent; we don’t want to not operate on patients who have disease," Dr. Paty said.
His criteria for clinical complete response include a flat mucosa with no nodularity or mass on digital rectal examination. Smooth induration or minor scarring without nodularity is acceptable, "but it has to have a benign feel to it," he said.
In addition, on proctoscopy the mucosa must appear normal and flat, and if a scar is present it should be pale or white in appearance. Alternatively, there can be telangiectasias, he said.
"What’s not clear is whether ulceration is an exclusion criterion [for nonoperative management]. For me it is. Any time I see ulceration I feel there is something ongoing in that tumor that is not resolved and I don’t feel comfortable calling it a complete response," Dr. Paty commented.
Take local route
When surgery is required, local excision rather than total mesorectal resection may suffice, Dr. Nelson said. Suitable patients may be those who are frail or elderly or have limited life expectancy or serious medical conditions that might preclude more extensive surgery.
Tumors that may be good candidates for local excision include smaller lesions (less than 2-3 cm) below the peritoneal reflection that are not amenable to lower anterior resection. The tumors should be subject to full thickness excision, and the team should be able to confirm negative margins, she noted.
Favorable pathologic findings include well-differentiated tumors with the absence of lymphovascular invasion, mucinous features, or signet ring features, she said.
"Local excision really just takes care of the primary, of course. It doesn’t deal with the lymphatics, which is always the hidden game," Dr. Nelson said.
She noted that a 1989 study showed that the likelihood of untreated lymph node disease in patients who had undergone local excision was 0% for patients with T1 tumors, 28% for those with T2 tumors, 36% for T3, and 53% for T4 lesions, showing a significant increase in risk associated with tumor depth (Cancer 1989;63:1421-9).
"If you start tackling anything above a T2 lesion, you’re probably going to be missing lymphatic disease. It’s of relevance because it will form the site of recurrent disease," she said.
For patients with T1 tumors, overall survival is the same, but disease-free survival and local recurrence rates favor standard resection over local excision. "Selection criteria must be much more restrictive when it comes to a T2 lesion," Dr. Nelson said. "I’m pretty reticent to use it in my own practice. I have to really choose the tumor well and choose the patient well to want to do that with some assurance that it’s the right decision."
She pointed to a study published in 2000 comparing patients who underwent either local excision or standard resection for rectal cancer (Dis. Colon Rectum 2000;43:1064-71). Over about 4.5 years of follow-up, local recurrence for patients with T2 lesions was 47% if they had received local excision, compared with 6% for those who had standard resections. Respective overall survival rates were 65% and 81%.
Dr. Paty and Dr. Nelson reported having no financial disclosures.
NATIONAL HARBOR, MD. – It may sound like heresy, but select patients with locally advanced rectal cancer may be spared surgery and its associated complications, a cancer surgeon suggested at the annual Society of Surgical Oncology Cancer Symposium.
Approximately 10%-25% of patients with locally advanced rectal cancer will have clinical complete responses (cCR) to neoadjuvant chemotherapy and radiation, said Dr. Philip B. Paty, an attending surgeon in the colorectal surgery service at Memorial Sloan-Kettering Cancer Center in New York City.
"The vast majority of these patients will avoid rectal resection, at least within the first 5 years," Dr. Paty added.
Although local failure occurs in 10%-25% of patients, most of the failures occur within the first 18 months, and most of these cases can be salvaged with R0 resections. Patients treated with nonoperative management appear to have rates of distant recurrence and survival similar to those of patients with pathologic complete responses (pCR) treated with total mesorectal resection, he said.
If surgery is required, local excision may be sufficient for some patients with stage T1 lesions and a select few with T2 lesions, said Dr. Heidi Nelson, professor of surgery in the department of colon and rectal surgery at the Mayo Clinic in Rochester, Minn.
If a patient has a favorable T1 lesion and would otherwise face a life-altering procedure such as abdominal perineal resection (APR) and colostomy, the surgeon should at least show the patient the data and discuss local excision as a safe and effective alternative with results comparable to more extensive resections, she said.
T2 lesions are more problematic, but a select few patients with this tumor type might be spared the morbidity of standard rectal resection, she added.
Hold the surgery?
Dr. Paty noted that, with standard management of stage T3 or T4 rectal cancers, the combination of neoadjuvant chemoradiotherapy, surgery, and adjuvant chemotherapy resulted in a 76% overall survival rate with less than 0.3% local recurrence after 5 years (Ann. Surg. 2005;241:829-36).
"What we have not dwelt on much is the morbidity of surgery, which is very significant. Having a rectal resection is a life-changing event for every patient that has one. Surgeons know that, and patients know that even better than surgeons," he said.
Rectal resections are associated with significant perioperative morbidity, colostomy, altered bowel function, sexual dysfunction, and infertility, he noted.
Pathologic complete responses to neoadjuvant therapy occur in 10%-44% of patients, and patients who have a pCR have markedly better oncologic outcomes than patients with less robust responses.
Of course, pCR can only be determined after surgery, raising the question of whether a clinical CR is sufficient for determining whether a patient might be spared rectal resection.
There are currently more data on pCR in rectal cancer than cCR, "probably because clinical CR criteria right now are quite stringent; we don’t want to not operate on patients who have disease," Dr. Paty said.
His criteria for clinical complete response include a flat mucosa with no nodularity or mass on digital rectal examination. Smooth induration or minor scarring without nodularity is acceptable, "but it has to have a benign feel to it," he said.
In addition, on proctoscopy the mucosa must appear normal and flat, and if a scar is present it should be pale or white in appearance. Alternatively, there can be telangiectasias, he said.
"What’s not clear is whether ulceration is an exclusion criterion [for nonoperative management]. For me it is. Any time I see ulceration I feel there is something ongoing in that tumor that is not resolved and I don’t feel comfortable calling it a complete response," Dr. Paty commented.
Take local route
When surgery is required, local excision rather than total mesorectal resection may suffice, Dr. Nelson said. Suitable patients may be those who are frail or elderly or have limited life expectancy or serious medical conditions that might preclude more extensive surgery.
Tumors that may be good candidates for local excision include smaller lesions (less than 2-3 cm) below the peritoneal reflection that are not amenable to lower anterior resection. The tumors should be subject to full thickness excision, and the team should be able to confirm negative margins, she noted.
Favorable pathologic findings include well-differentiated tumors with the absence of lymphovascular invasion, mucinous features, or signet ring features, she said.
"Local excision really just takes care of the primary, of course. It doesn’t deal with the lymphatics, which is always the hidden game," Dr. Nelson said.
She noted that a 1989 study showed that the likelihood of untreated lymph node disease in patients who had undergone local excision was 0% for patients with T1 tumors, 28% for those with T2 tumors, 36% for T3, and 53% for T4 lesions, showing a significant increase in risk associated with tumor depth (Cancer 1989;63:1421-9).
"If you start tackling anything above a T2 lesion, you’re probably going to be missing lymphatic disease. It’s of relevance because it will form the site of recurrent disease," she said.
For patients with T1 tumors, overall survival is the same, but disease-free survival and local recurrence rates favor standard resection over local excision. "Selection criteria must be much more restrictive when it comes to a T2 lesion," Dr. Nelson said. "I’m pretty reticent to use it in my own practice. I have to really choose the tumor well and choose the patient well to want to do that with some assurance that it’s the right decision."
She pointed to a study published in 2000 comparing patients who underwent either local excision or standard resection for rectal cancer (Dis. Colon Rectum 2000;43:1064-71). Over about 4.5 years of follow-up, local recurrence for patients with T2 lesions was 47% if they had received local excision, compared with 6% for those who had standard resections. Respective overall survival rates were 65% and 81%.
Dr. Paty and Dr. Nelson reported having no financial disclosures.
NATIONAL HARBOR, MD. – It may sound like heresy, but select patients with locally advanced rectal cancer may be spared surgery and its associated complications, a cancer surgeon suggested at the annual Society of Surgical Oncology Cancer Symposium.
Approximately 10%-25% of patients with locally advanced rectal cancer will have clinical complete responses (cCR) to neoadjuvant chemotherapy and radiation, said Dr. Philip B. Paty, an attending surgeon in the colorectal surgery service at Memorial Sloan-Kettering Cancer Center in New York City.
"The vast majority of these patients will avoid rectal resection, at least within the first 5 years," Dr. Paty added.
Although local failure occurs in 10%-25% of patients, most of the failures occur within the first 18 months, and most of these cases can be salvaged with R0 resections. Patients treated with nonoperative management appear to have rates of distant recurrence and survival similar to those of patients with pathologic complete responses (pCR) treated with total mesorectal resection, he said.
If surgery is required, local excision may be sufficient for some patients with stage T1 lesions and a select few with T2 lesions, said Dr. Heidi Nelson, professor of surgery in the department of colon and rectal surgery at the Mayo Clinic in Rochester, Minn.
If a patient has a favorable T1 lesion and would otherwise face a life-altering procedure such as abdominal perineal resection (APR) and colostomy, the surgeon should at least show the patient the data and discuss local excision as a safe and effective alternative with results comparable to more extensive resections, she said.
T2 lesions are more problematic, but a select few patients with this tumor type might be spared the morbidity of standard rectal resection, she added.
Hold the surgery?
Dr. Paty noted that, with standard management of stage T3 or T4 rectal cancers, the combination of neoadjuvant chemoradiotherapy, surgery, and adjuvant chemotherapy resulted in a 76% overall survival rate with less than 0.3% local recurrence after 5 years (Ann. Surg. 2005;241:829-36).
"What we have not dwelt on much is the morbidity of surgery, which is very significant. Having a rectal resection is a life-changing event for every patient that has one. Surgeons know that, and patients know that even better than surgeons," he said.
Rectal resections are associated with significant perioperative morbidity, colostomy, altered bowel function, sexual dysfunction, and infertility, he noted.
Pathologic complete responses to neoadjuvant therapy occur in 10%-44% of patients, and patients who have a pCR have markedly better oncologic outcomes than patients with less robust responses.
Of course, pCR can only be determined after surgery, raising the question of whether a clinical CR is sufficient for determining whether a patient might be spared rectal resection.
There are currently more data on pCR in rectal cancer than cCR, "probably because clinical CR criteria right now are quite stringent; we don’t want to not operate on patients who have disease," Dr. Paty said.
His criteria for clinical complete response include a flat mucosa with no nodularity or mass on digital rectal examination. Smooth induration or minor scarring without nodularity is acceptable, "but it has to have a benign feel to it," he said.
In addition, on proctoscopy the mucosa must appear normal and flat, and if a scar is present it should be pale or white in appearance. Alternatively, there can be telangiectasias, he said.
"What’s not clear is whether ulceration is an exclusion criterion [for nonoperative management]. For me it is. Any time I see ulceration I feel there is something ongoing in that tumor that is not resolved and I don’t feel comfortable calling it a complete response," Dr. Paty commented.
Take local route
When surgery is required, local excision rather than total mesorectal resection may suffice, Dr. Nelson said. Suitable patients may be those who are frail or elderly or have limited life expectancy or serious medical conditions that might preclude more extensive surgery.
Tumors that may be good candidates for local excision include smaller lesions (less than 2-3 cm) below the peritoneal reflection that are not amenable to lower anterior resection. The tumors should be subject to full thickness excision, and the team should be able to confirm negative margins, she noted.
Favorable pathologic findings include well-differentiated tumors with the absence of lymphovascular invasion, mucinous features, or signet ring features, she said.
"Local excision really just takes care of the primary, of course. It doesn’t deal with the lymphatics, which is always the hidden game," Dr. Nelson said.
She noted that a 1989 study showed that the likelihood of untreated lymph node disease in patients who had undergone local excision was 0% for patients with T1 tumors, 28% for those with T2 tumors, 36% for T3, and 53% for T4 lesions, showing a significant increase in risk associated with tumor depth (Cancer 1989;63:1421-9).
"If you start tackling anything above a T2 lesion, you’re probably going to be missing lymphatic disease. It’s of relevance because it will form the site of recurrent disease," she said.
For patients with T1 tumors, overall survival is the same, but disease-free survival and local recurrence rates favor standard resection over local excision. "Selection criteria must be much more restrictive when it comes to a T2 lesion," Dr. Nelson said. "I’m pretty reticent to use it in my own practice. I have to really choose the tumor well and choose the patient well to want to do that with some assurance that it’s the right decision."
She pointed to a study published in 2000 comparing patients who underwent either local excision or standard resection for rectal cancer (Dis. Colon Rectum 2000;43:1064-71). Over about 4.5 years of follow-up, local recurrence for patients with T2 lesions was 47% if they had received local excision, compared with 6% for those who had standard resections. Respective overall survival rates were 65% and 81%.
Dr. Paty and Dr. Nelson reported having no financial disclosures.
EXPERT ANALYSIS FROM SSO 2013
Bariatric surgery reduces mortality in obese diabetic patients
Bariatric surgery can significantly reduce the need for insulin and can even induce complete remission of type 2 diabetes mellitus, but some forms of the procedure pose too much risk and should be shunned, said a diabetes specialist at the annual advanced postgraduate course held by the American Diabetes Association.
Depending on the type of procedure, weight loss in the first year following bariatric surgery can range from about 30 kg to more than 50 kg, and the surgery can reduce the risk of mortality for formerly obese patients by about 25%, said Dr. John Bantle, professor of medicine and director of the division of endocrinology and diabetes at the University of Minnesota in Minneapolis.
Bariatric surgery should be considered in type 2 diabetic patients with a body mass index greater than 35 kg/m2, and may also benefit patients with a BMI of 30-35, although there is not enough evidence to support the latter assertion, Dr. Bantle said.
"I would choose laparoscopic gastric bypass as the procedure to recommend. I think laparoscopic gastric banding doesn’t have enough efficacy, and duodenal switch has too many complications," he said.
Looking back at Look AHEAD
The most recent follow-up data from the Action for Health in Diabetes (Look AHEAD) study compared cardiovascular event rates among patients randomized to either an intensive lifestyle intervention or a diabetes support and education program. Analysis showed that the lifestyle intervention was associated with significantly more weight loss (–4.7% vs. –1.1% at 4 years, P less than .001), and with a more favorable change in fitness levels, hemoglobin A1c, systolic blood pressure, high-density lipoprotein cholesterol levels, and triglyceride levels (P less than .001 for all comparisons) (Arch. Intern. Med. 2010;170:1566-75).
However, in September 2012, the National Institutes of Health, acting on the advice of the trial’s data and safety monitoring team, halted the lifestyle intervention because the primary endpoint of reduction in cardiovascular events had not been reached and would be unlikely to do so in the 2 years remaining in the study.
Although the trial failed to meet the cardiovascular endpoint, there was no evidence of harm, and patients who were randomized to the intervention had reduced need for diabetes medications, less sleep apnea, and increases in both physical mobility and quality of life.
"In data that are still under analysis and not yet published, I think we are going to see that there was some benefit on microvascular complications," Dr. Bantle said.
Choose procedures wisely
If lifestyle interventions are not sufficient, bariatric surgery may be helpful, Dr. Bantle said. Procedures commonly used in the United States include laparoscopic gastric banding, Roux-en-Y gastric bypass, and increasingly, sleeve gastrectomy and biliopancreatic diversion with duodenal switch.
The latter procedure is similar to a jejunoileal bypass, an early type of bariatric surgery that is no longer performed due to the high complication rate. Biliopancreatic diversion with duodenal switch involves division of the stomach and creation of a small gastric pouch that is drained with a loop of intestine attached downward to the ileum toward its head, so that biliary and pancreatic drainage occurs through the small intestine before the digestive juices meet up with food for digestion and absorption toward the end of the intestinal tract.
"I think this procedure is a really bad idea; it’s too much like a jejunoileal bypass with anastomosis, and I advise all my patients who are considering bariatric surgery to decline the option to pursue this procedure," he said.
Evidence favors surgery
Reviewing the evidence, Dr. Bantle pointed to a 2005 meta-analysis of 147 studies of bariatric surgery for obesity, which showed a mean 30.2-kg weight loss at 12 months and 34.8-kg loss after 36 months in patients who had a gastric banding procedure, compared with 43.5 kg and 41.5 kg, respectively, for gastric bypass, and 51.9 kg and 53.1 kg for those who had surgery with a duodenal switch procedure (Ann. Intern. Med. 2005;142:547-59).
The study also showed, however, that the rate of adverse events, including reflux, vomiting, dysphagia, and dumping syndrome, was markedly higher for patients who underwent a duodenal switch, at 37.7%, compared with 7.0% for gastric banding and 16.9% for gastric bypass.
A 2007 Swedish study also saw a reduction in mortality at a mean of 10.9 years of follow-up among 2,010 obese patients who underwent bariatric surgery compared with 2,037 patients who received standard medical management. The hazard ratio for death in the surgical patients was 0.76 (P = .04). The most common causes of death were myocardial infarction, which occurred in 13 surgical patients and 25 controls, and cancer, which occurred in 29 surgical patients and 47 controls (N. Engl. J. Med. 2007;357:741-52).
Similarly, a retrospective cohort study comparing 7,925 obese people who underwent gastric bypass surgery with the same number of obese controls matched by age, gender, and BMI showed that after a mean follow-up of 7.1 years, adjusted mortality for surgical patients was 37.6 per 10,000 person-years, compared with 57.1 per 10,000 for controls, a relative difference of 40%. Additionally, diabetes-specific mortality declined by 92% in the gastric bypass patients (N. Engl. J. Med. 2007;357:753-61).
Following surgery, it is important to ensure that patients have a protein intake of 60-120 g daily, and if they have undergone gastric bypass or duodenal switch procedures their diet should be supplemented with a multivitamin containing folate, thiamine, iron, vitamin B12, calcium, and vitamin D, Dr. Bantle said.
"I would suggest that the earlier the surgery is done, the better. Remission of diabetes is predicted by short duration of diabetes, need for few diabetes medications, and high postprandial C-peptide, so if a patient is considering [bariatric surgery], I typically get a postprandial C-peptide and use that to counsel them about whether or not they’re likely to have remission of diabetes after the procedure," he concluded.
Dr. Bantle reported having no financial disclosures.
Bariatric surgery can significantly reduce the need for insulin and can even induce complete remission of type 2 diabetes mellitus, but some forms of the procedure pose too much risk and should be shunned, said a diabetes specialist at the annual advanced postgraduate course held by the American Diabetes Association.
Depending on the type of procedure, weight loss in the first year following bariatric surgery can range from about 30 kg to more than 50 kg, and the surgery can reduce the risk of mortality for formerly obese patients by about 25%, said Dr. John Bantle, professor of medicine and director of the division of endocrinology and diabetes at the University of Minnesota in Minneapolis.
Bariatric surgery should be considered in type 2 diabetic patients with a body mass index greater than 35 kg/m2, and may also benefit patients with a BMI of 30-35, although there is not enough evidence to support the latter assertion, Dr. Bantle said.
"I would choose laparoscopic gastric bypass as the procedure to recommend. I think laparoscopic gastric banding doesn’t have enough efficacy, and duodenal switch has too many complications," he said.
Looking back at Look AHEAD
The most recent follow-up data from the Action for Health in Diabetes (Look AHEAD) study compared cardiovascular event rates among patients randomized to either an intensive lifestyle intervention or a diabetes support and education program. Analysis showed that the lifestyle intervention was associated with significantly more weight loss (–4.7% vs. –1.1% at 4 years, P less than .001), and with a more favorable change in fitness levels, hemoglobin A1c, systolic blood pressure, high-density lipoprotein cholesterol levels, and triglyceride levels (P less than .001 for all comparisons) (Arch. Intern. Med. 2010;170:1566-75).
However, in September 2012, the National Institutes of Health, acting on the advice of the trial’s data and safety monitoring team, halted the lifestyle intervention because the primary endpoint of reduction in cardiovascular events had not been reached and would be unlikely to do so in the 2 years remaining in the study.
Although the trial failed to meet the cardiovascular endpoint, there was no evidence of harm, and patients who were randomized to the intervention had reduced need for diabetes medications, less sleep apnea, and increases in both physical mobility and quality of life.
"In data that are still under analysis and not yet published, I think we are going to see that there was some benefit on microvascular complications," Dr. Bantle said.
Choose procedures wisely
If lifestyle interventions are not sufficient, bariatric surgery may be helpful, Dr. Bantle said. Procedures commonly used in the United States include laparoscopic gastric banding, Roux-en-Y gastric bypass, and increasingly, sleeve gastrectomy and biliopancreatic diversion with duodenal switch.
The latter procedure is similar to a jejunoileal bypass, an early type of bariatric surgery that is no longer performed due to the high complication rate. Biliopancreatic diversion with duodenal switch involves division of the stomach and creation of a small gastric pouch that is drained with a loop of intestine attached downward to the ileum toward its head, so that biliary and pancreatic drainage occurs through the small intestine before the digestive juices meet up with food for digestion and absorption toward the end of the intestinal tract.
"I think this procedure is a really bad idea; it’s too much like a jejunoileal bypass with anastomosis, and I advise all my patients who are considering bariatric surgery to decline the option to pursue this procedure," he said.
Evidence favors surgery
Reviewing the evidence, Dr. Bantle pointed to a 2005 meta-analysis of 147 studies of bariatric surgery for obesity, which showed a mean 30.2-kg weight loss at 12 months and 34.8-kg loss after 36 months in patients who had a gastric banding procedure, compared with 43.5 kg and 41.5 kg, respectively, for gastric bypass, and 51.9 kg and 53.1 kg for those who had surgery with a duodenal switch procedure (Ann. Intern. Med. 2005;142:547-59).
The study also showed, however, that the rate of adverse events, including reflux, vomiting, dysphagia, and dumping syndrome, was markedly higher for patients who underwent a duodenal switch, at 37.7%, compared with 7.0% for gastric banding and 16.9% for gastric bypass.
A 2007 Swedish study also saw a reduction in mortality at a mean of 10.9 years of follow-up among 2,010 obese patients who underwent bariatric surgery compared with 2,037 patients who received standard medical management. The hazard ratio for death in the surgical patients was 0.76 (P = .04). The most common causes of death were myocardial infarction, which occurred in 13 surgical patients and 25 controls, and cancer, which occurred in 29 surgical patients and 47 controls (N. Engl. J. Med. 2007;357:741-52).
Similarly, a retrospective cohort study comparing 7,925 obese people who underwent gastric bypass surgery with the same number of obese controls matched by age, gender, and BMI showed that after a mean follow-up of 7.1 years, adjusted mortality for surgical patients was 37.6 per 10,000 person-years, compared with 57.1 per 10,000 for controls, a relative difference of 40%. Additionally, diabetes-specific mortality declined by 92% in the gastric bypass patients (N. Engl. J. Med. 2007;357:753-61).
Following surgery, it is important to ensure that patients have a protein intake of 60-120 g daily, and if they have undergone gastric bypass or duodenal switch procedures their diet should be supplemented with a multivitamin containing folate, thiamine, iron, vitamin B12, calcium, and vitamin D, Dr. Bantle said.
"I would suggest that the earlier the surgery is done, the better. Remission of diabetes is predicted by short duration of diabetes, need for few diabetes medications, and high postprandial C-peptide, so if a patient is considering [bariatric surgery], I typically get a postprandial C-peptide and use that to counsel them about whether or not they’re likely to have remission of diabetes after the procedure," he concluded.
Dr. Bantle reported having no financial disclosures.
Bariatric surgery can significantly reduce the need for insulin and can even induce complete remission of type 2 diabetes mellitus, but some forms of the procedure pose too much risk and should be shunned, said a diabetes specialist at the annual advanced postgraduate course held by the American Diabetes Association.
Depending on the type of procedure, weight loss in the first year following bariatric surgery can range from about 30 kg to more than 50 kg, and the surgery can reduce the risk of mortality for formerly obese patients by about 25%, said Dr. John Bantle, professor of medicine and director of the division of endocrinology and diabetes at the University of Minnesota in Minneapolis.
Bariatric surgery should be considered in type 2 diabetic patients with a body mass index greater than 35 kg/m2, and may also benefit patients with a BMI of 30-35, although there is not enough evidence to support the latter assertion, Dr. Bantle said.
"I would choose laparoscopic gastric bypass as the procedure to recommend. I think laparoscopic gastric banding doesn’t have enough efficacy, and duodenal switch has too many complications," he said.
Looking back at Look AHEAD
The most recent follow-up data from the Action for Health in Diabetes (Look AHEAD) study compared cardiovascular event rates among patients randomized to either an intensive lifestyle intervention or a diabetes support and education program. Analysis showed that the lifestyle intervention was associated with significantly more weight loss (–4.7% vs. –1.1% at 4 years, P less than .001), and with a more favorable change in fitness levels, hemoglobin A1c, systolic blood pressure, high-density lipoprotein cholesterol levels, and triglyceride levels (P less than .001 for all comparisons) (Arch. Intern. Med. 2010;170:1566-75).
However, in September 2012, the National Institutes of Health, acting on the advice of the trial’s data and safety monitoring team, halted the lifestyle intervention because the primary endpoint of reduction in cardiovascular events had not been reached and would be unlikely to do so in the 2 years remaining in the study.
Although the trial failed to meet the cardiovascular endpoint, there was no evidence of harm, and patients who were randomized to the intervention had reduced need for diabetes medications, less sleep apnea, and increases in both physical mobility and quality of life.
"In data that are still under analysis and not yet published, I think we are going to see that there was some benefit on microvascular complications," Dr. Bantle said.
Choose procedures wisely
If lifestyle interventions are not sufficient, bariatric surgery may be helpful, Dr. Bantle said. Procedures commonly used in the United States include laparoscopic gastric banding, Roux-en-Y gastric bypass, and increasingly, sleeve gastrectomy and biliopancreatic diversion with duodenal switch.
The latter procedure is similar to a jejunoileal bypass, an early type of bariatric surgery that is no longer performed due to the high complication rate. Biliopancreatic diversion with duodenal switch involves division of the stomach and creation of a small gastric pouch that is drained with a loop of intestine attached downward to the ileum toward its head, so that biliary and pancreatic drainage occurs through the small intestine before the digestive juices meet up with food for digestion and absorption toward the end of the intestinal tract.
"I think this procedure is a really bad idea; it’s too much like a jejunoileal bypass with anastomosis, and I advise all my patients who are considering bariatric surgery to decline the option to pursue this procedure," he said.
Evidence favors surgery
Reviewing the evidence, Dr. Bantle pointed to a 2005 meta-analysis of 147 studies of bariatric surgery for obesity, which showed a mean 30.2-kg weight loss at 12 months and 34.8-kg loss after 36 months in patients who had a gastric banding procedure, compared with 43.5 kg and 41.5 kg, respectively, for gastric bypass, and 51.9 kg and 53.1 kg for those who had surgery with a duodenal switch procedure (Ann. Intern. Med. 2005;142:547-59).
The study also showed, however, that the rate of adverse events, including reflux, vomiting, dysphagia, and dumping syndrome, was markedly higher for patients who underwent a duodenal switch, at 37.7%, compared with 7.0% for gastric banding and 16.9% for gastric bypass.
A 2007 Swedish study also saw a reduction in mortality at a mean of 10.9 years of follow-up among 2,010 obese patients who underwent bariatric surgery compared with 2,037 patients who received standard medical management. The hazard ratio for death in the surgical patients was 0.76 (P = .04). The most common causes of death were myocardial infarction, which occurred in 13 surgical patients and 25 controls, and cancer, which occurred in 29 surgical patients and 47 controls (N. Engl. J. Med. 2007;357:741-52).
Similarly, a retrospective cohort study comparing 7,925 obese people who underwent gastric bypass surgery with the same number of obese controls matched by age, gender, and BMI showed that after a mean follow-up of 7.1 years, adjusted mortality for surgical patients was 37.6 per 10,000 person-years, compared with 57.1 per 10,000 for controls, a relative difference of 40%. Additionally, diabetes-specific mortality declined by 92% in the gastric bypass patients (N. Engl. J. Med. 2007;357:753-61).
Following surgery, it is important to ensure that patients have a protein intake of 60-120 g daily, and if they have undergone gastric bypass or duodenal switch procedures their diet should be supplemented with a multivitamin containing folate, thiamine, iron, vitamin B12, calcium, and vitamin D, Dr. Bantle said.
"I would suggest that the earlier the surgery is done, the better. Remission of diabetes is predicted by short duration of diabetes, need for few diabetes medications, and high postprandial C-peptide, so if a patient is considering [bariatric surgery], I typically get a postprandial C-peptide and use that to counsel them about whether or not they’re likely to have remission of diabetes after the procedure," he concluded.
Dr. Bantle reported having no financial disclosures.
AT THE ADA ADVANCED POSTGRADUATE COURSE
Major finding: Weight loss in the first year following bariatric surgery ranges from about 30 kg to more than 50 kg, and the surgery can reduce the risk of mortality for formerly obese patients by about 25%,
Data source: Review of medical literature on bariatric surgery for obese patients both with and without type 2 diabetes mellitus.
Disclosures: Dr. Bantle reported having no financial disclosures.
Robotic surgery called 'in the destiny of humanity'
The way Dr. Pier Cristoforo Giulianotti sees it, robots will play an increasingly prominent role in the future of surgery. He should know. Dr. Giulianotti, the Lloyd Nyhus Professor of Surgery and chief, division of minimally invasive, general, and robotic surgery at the University of Illinois at Chicago, pioneered robotic lung resection in 2002 and was the first surgeon to perform a robotic Whipple procedure in 2001.
"In my opinion there is no way back from robotic surgery," he said in an interview. "It’s in the destiny of humanity, for the simple reason: to perform more precisely and to overcome our limitations – not only manual limitations, the ability to control movements at a microscopic level, for example – but also diagnostic limitations. The computer and the artificial intelligence of the future will integrate our senses and our mechanical abilities, so we will be able to perform more precisely on selected targets."
Currently, most abdominal procedures performed at the University of Illinois Medical Center are done robotically, including the Whipple procedure, splenectomy, total gastrectomy, lung lobectomy, colorectal surgery, thyroidectomy, adrenalectomy, esophagectomy, major hepatectomies, and common bile duct procedures. The robot "has enormous benefits for the patients," Dr. Giulianotti said. "Open procedures involve a longer postoperative stay and more complications, and it takes more time to enter adjuvant oncological treatment after surgery, and there is less blood loss."
In Dr. Giulianotti’s published experience of 134 robotic-assisted laparoscopic pancreatic surgery procedures, morbidity was 26%, mortality was 2.2%, the conversion rate was 10.4%, and the fistula rate was 20.9%. The majority were grade A fistulas not requiring any treatment (Surg. Endosc. 2010; 24:1646-57). He said that robotic surgery "enables difficult technical maneuvers to be performed that facilitate the success of pancreatic minimally invasive surgery. The results in this series demonstrate feasibility and safety with clinical outcomes."
In a separate study, investigators who compared 32 open vs. 28 laparoscopic vs. 17 robotic distal pancreatectomies found that all three procedures resulted in a similar cost, while the robotic group had a shorter hospital stay, a higher rate of spleen-preserving cases, and an increased operative time, compared with patients in the other two groups (Surgery 2010;148:814-23).
Pelvic indications that are becoming the gold standard for robotic surgery include robotic prostatectomy and resection for rectal cancer. "When you have a very deep and complex surgical field like the pelvis, and you need to do a radical resection for cancer while at the same time sparing nerves that can be important for urinary function, the robot combines three-dimensional vision plus improved functionality of the instruments, allowing for a more precise dissection," Dr. Giulianotti said.
Dr. Leela M. Prasad, chief of the division of colon and rectal surgery at the University of Illinois at Chicago, described robotic-assisted surgery for rectal cancer as "far superior to anything we know right now," especially in overweight males. According to combined results from three published studies, circumferential margin positivity ranged from 0 to 7%, which, Dr. Prasad said, is significantly lower than the average number of positive margins reported following open and laparoscopic rectal surgery procedures (14% and 16%, respectively) (Ann. Surg. Oncol. 2007;14:3168-73; Surg. Oncol. 2009;16:1480-7; Dis. Colon Rectum 2009;52:1824-30).
At the present time, however, in his clinical experience there are no significant differences in outcomes between robotic-assisted and laparoscopically assisted open surgery for colon cancer. "We looked at our data, and it did not make any difference robotically or laparoscopically in terms of length of stay, lymph node harvest, or survival," he said. A recent study from Korean investigators supports those findings (Surg. Endosc. 2012 Dec. 13).
Dr. Giulianotti estimated that there are more than 1,600 robotic surgery systems in the United States. Some of them are devoted to urologic or gynecologic applications, "but in the majority of hospitals there is a growing tendency to use the robot as part of a multidisciplinary program," he said. "That means at the same institution different surgical teams on different days are using the robot for different indications. Of the 1,600 systems out there, I would estimate that about half are used for general surgery."
While hospital administrators have warmed up to the idea of initiating robotic surgery programs at their institutions in recent years, Dr. Giulianotti and Dr. Prasad both called for a culture change in terms of how surgeons and medical educators think about the robot’s place in clinical medicine. "There is a cultural resistance in accepting big changes in surgery," Dr. Giulianotti said. "Academic medical institutions can play a key role here. They need to start teaching robotics in a mandatory way by offering training opportunities in a lab. We also believe that simulation has a big future in this kind of training."
The economic investment required to launch a robotic surgery program is another challenge. "Some hospitals are only concerned about the cost," he said. "When you are doing the same procedure laparoscopically, the cost is probably about 30% less. In the future I hope we can better impact the overall economic aspect of surgical procedures. That means speeding up the postoperative and outpatient treatment of some pathologies. We are already doing Nissen fundoplication and gallbladder removal with robotic surgery, and patients are being discharged a few hours after the surgery."
Dr. Prasad said the field will be poised to further advance when makers of robotic surgery devices and instruments improve on existing technology. "I am using the same robot and instruments as I was using 3 years ago," he said. "I think we need new technologies – including a smaller robot to cut down the costs and speed up the operations. This will make it easier for patients."
To launch a successful robotic surgery program in this day and age, "you need a good team of nurses and surgeons who are dedicated to doing robotic surgery," Dr. Prasad concluded. "It is dangerous in the hands of a surgeon who is not qualified."
Dr. Giulianotti said that he had no relevant financial conflicts to disclose. Dr. Prasad disclosed that he has received honoraria from Intuitive Surgical, Ethicon Endo-Surgery, and Covidien.
I would like to congratulate Dr. Pier Cristoforo Giulianotti for his work to date demonstrating the effectiveness of robotic pancreatic surgery. Clearly the “robot,” as it is affectionately termed, is here to stay.
It has clear applications in all surgical subspecialties and is being embraced by surgeons, administrators, and patients as an important tool. It is a platform that will facilitate future developments that are certain to change the way we practice our craft.
Dr. John Sweeney |
Given the focus being placed on the value of health care deliveredin the United States, it is incumbent upon surgeons to continuously evaluate the quality and costs of each intervention that we offer to our patients. To many, this type of evaluative process might seem to come up short when it comes to robotic applications for many minimally invasive procedures. Quality in most studies has been equivalent, while the costs associated with this expensive technology can be very significant. Because this new piece of technology is the first step in a new direction, we must continue to place the time, effort and cost into refining, enhancing and improving it’s applications as the technology stands today. However unless we are able to accomplish this goal, I fear we fall into the trap of “using a Cadillac for a golf cart” which is something that the U.S. health care system cannot afford nor sustain.
Dr. John Sweeney, is an ACS Fellow and the W. Dean Warren Distinguished Chair in Surgery at Emory University, Atlanta.
I would like to congratulate Dr. Pier Cristoforo Giulianotti for his work to date demonstrating the effectiveness of robotic pancreatic surgery. Clearly the “robot,” as it is affectionately termed, is here to stay.
It has clear applications in all surgical subspecialties and is being embraced by surgeons, administrators, and patients as an important tool. It is a platform that will facilitate future developments that are certain to change the way we practice our craft.
Dr. John Sweeney |
Given the focus being placed on the value of health care deliveredin the United States, it is incumbent upon surgeons to continuously evaluate the quality and costs of each intervention that we offer to our patients. To many, this type of evaluative process might seem to come up short when it comes to robotic applications for many minimally invasive procedures. Quality in most studies has been equivalent, while the costs associated with this expensive technology can be very significant. Because this new piece of technology is the first step in a new direction, we must continue to place the time, effort and cost into refining, enhancing and improving it’s applications as the technology stands today. However unless we are able to accomplish this goal, I fear we fall into the trap of “using a Cadillac for a golf cart” which is something that the U.S. health care system cannot afford nor sustain.
Dr. John Sweeney, is an ACS Fellow and the W. Dean Warren Distinguished Chair in Surgery at Emory University, Atlanta.
I would like to congratulate Dr. Pier Cristoforo Giulianotti for his work to date demonstrating the effectiveness of robotic pancreatic surgery. Clearly the “robot,” as it is affectionately termed, is here to stay.
It has clear applications in all surgical subspecialties and is being embraced by surgeons, administrators, and patients as an important tool. It is a platform that will facilitate future developments that are certain to change the way we practice our craft.
Dr. John Sweeney |
Given the focus being placed on the value of health care deliveredin the United States, it is incumbent upon surgeons to continuously evaluate the quality and costs of each intervention that we offer to our patients. To many, this type of evaluative process might seem to come up short when it comes to robotic applications for many minimally invasive procedures. Quality in most studies has been equivalent, while the costs associated with this expensive technology can be very significant. Because this new piece of technology is the first step in a new direction, we must continue to place the time, effort and cost into refining, enhancing and improving it’s applications as the technology stands today. However unless we are able to accomplish this goal, I fear we fall into the trap of “using a Cadillac for a golf cart” which is something that the U.S. health care system cannot afford nor sustain.
Dr. John Sweeney, is an ACS Fellow and the W. Dean Warren Distinguished Chair in Surgery at Emory University, Atlanta.
The way Dr. Pier Cristoforo Giulianotti sees it, robots will play an increasingly prominent role in the future of surgery. He should know. Dr. Giulianotti, the Lloyd Nyhus Professor of Surgery and chief, division of minimally invasive, general, and robotic surgery at the University of Illinois at Chicago, pioneered robotic lung resection in 2002 and was the first surgeon to perform a robotic Whipple procedure in 2001.
"In my opinion there is no way back from robotic surgery," he said in an interview. "It’s in the destiny of humanity, for the simple reason: to perform more precisely and to overcome our limitations – not only manual limitations, the ability to control movements at a microscopic level, for example – but also diagnostic limitations. The computer and the artificial intelligence of the future will integrate our senses and our mechanical abilities, so we will be able to perform more precisely on selected targets."
Currently, most abdominal procedures performed at the University of Illinois Medical Center are done robotically, including the Whipple procedure, splenectomy, total gastrectomy, lung lobectomy, colorectal surgery, thyroidectomy, adrenalectomy, esophagectomy, major hepatectomies, and common bile duct procedures. The robot "has enormous benefits for the patients," Dr. Giulianotti said. "Open procedures involve a longer postoperative stay and more complications, and it takes more time to enter adjuvant oncological treatment after surgery, and there is less blood loss."
In Dr. Giulianotti’s published experience of 134 robotic-assisted laparoscopic pancreatic surgery procedures, morbidity was 26%, mortality was 2.2%, the conversion rate was 10.4%, and the fistula rate was 20.9%. The majority were grade A fistulas not requiring any treatment (Surg. Endosc. 2010; 24:1646-57). He said that robotic surgery "enables difficult technical maneuvers to be performed that facilitate the success of pancreatic minimally invasive surgery. The results in this series demonstrate feasibility and safety with clinical outcomes."
In a separate study, investigators who compared 32 open vs. 28 laparoscopic vs. 17 robotic distal pancreatectomies found that all three procedures resulted in a similar cost, while the robotic group had a shorter hospital stay, a higher rate of spleen-preserving cases, and an increased operative time, compared with patients in the other two groups (Surgery 2010;148:814-23).
Pelvic indications that are becoming the gold standard for robotic surgery include robotic prostatectomy and resection for rectal cancer. "When you have a very deep and complex surgical field like the pelvis, and you need to do a radical resection for cancer while at the same time sparing nerves that can be important for urinary function, the robot combines three-dimensional vision plus improved functionality of the instruments, allowing for a more precise dissection," Dr. Giulianotti said.
Dr. Leela M. Prasad, chief of the division of colon and rectal surgery at the University of Illinois at Chicago, described robotic-assisted surgery for rectal cancer as "far superior to anything we know right now," especially in overweight males. According to combined results from three published studies, circumferential margin positivity ranged from 0 to 7%, which, Dr. Prasad said, is significantly lower than the average number of positive margins reported following open and laparoscopic rectal surgery procedures (14% and 16%, respectively) (Ann. Surg. Oncol. 2007;14:3168-73; Surg. Oncol. 2009;16:1480-7; Dis. Colon Rectum 2009;52:1824-30).
At the present time, however, in his clinical experience there are no significant differences in outcomes between robotic-assisted and laparoscopically assisted open surgery for colon cancer. "We looked at our data, and it did not make any difference robotically or laparoscopically in terms of length of stay, lymph node harvest, or survival," he said. A recent study from Korean investigators supports those findings (Surg. Endosc. 2012 Dec. 13).
Dr. Giulianotti estimated that there are more than 1,600 robotic surgery systems in the United States. Some of them are devoted to urologic or gynecologic applications, "but in the majority of hospitals there is a growing tendency to use the robot as part of a multidisciplinary program," he said. "That means at the same institution different surgical teams on different days are using the robot for different indications. Of the 1,600 systems out there, I would estimate that about half are used for general surgery."
While hospital administrators have warmed up to the idea of initiating robotic surgery programs at their institutions in recent years, Dr. Giulianotti and Dr. Prasad both called for a culture change in terms of how surgeons and medical educators think about the robot’s place in clinical medicine. "There is a cultural resistance in accepting big changes in surgery," Dr. Giulianotti said. "Academic medical institutions can play a key role here. They need to start teaching robotics in a mandatory way by offering training opportunities in a lab. We also believe that simulation has a big future in this kind of training."
The economic investment required to launch a robotic surgery program is another challenge. "Some hospitals are only concerned about the cost," he said. "When you are doing the same procedure laparoscopically, the cost is probably about 30% less. In the future I hope we can better impact the overall economic aspect of surgical procedures. That means speeding up the postoperative and outpatient treatment of some pathologies. We are already doing Nissen fundoplication and gallbladder removal with robotic surgery, and patients are being discharged a few hours after the surgery."
Dr. Prasad said the field will be poised to further advance when makers of robotic surgery devices and instruments improve on existing technology. "I am using the same robot and instruments as I was using 3 years ago," he said. "I think we need new technologies – including a smaller robot to cut down the costs and speed up the operations. This will make it easier for patients."
To launch a successful robotic surgery program in this day and age, "you need a good team of nurses and surgeons who are dedicated to doing robotic surgery," Dr. Prasad concluded. "It is dangerous in the hands of a surgeon who is not qualified."
Dr. Giulianotti said that he had no relevant financial conflicts to disclose. Dr. Prasad disclosed that he has received honoraria from Intuitive Surgical, Ethicon Endo-Surgery, and Covidien.
The way Dr. Pier Cristoforo Giulianotti sees it, robots will play an increasingly prominent role in the future of surgery. He should know. Dr. Giulianotti, the Lloyd Nyhus Professor of Surgery and chief, division of minimally invasive, general, and robotic surgery at the University of Illinois at Chicago, pioneered robotic lung resection in 2002 and was the first surgeon to perform a robotic Whipple procedure in 2001.
"In my opinion there is no way back from robotic surgery," he said in an interview. "It’s in the destiny of humanity, for the simple reason: to perform more precisely and to overcome our limitations – not only manual limitations, the ability to control movements at a microscopic level, for example – but also diagnostic limitations. The computer and the artificial intelligence of the future will integrate our senses and our mechanical abilities, so we will be able to perform more precisely on selected targets."
Currently, most abdominal procedures performed at the University of Illinois Medical Center are done robotically, including the Whipple procedure, splenectomy, total gastrectomy, lung lobectomy, colorectal surgery, thyroidectomy, adrenalectomy, esophagectomy, major hepatectomies, and common bile duct procedures. The robot "has enormous benefits for the patients," Dr. Giulianotti said. "Open procedures involve a longer postoperative stay and more complications, and it takes more time to enter adjuvant oncological treatment after surgery, and there is less blood loss."
In Dr. Giulianotti’s published experience of 134 robotic-assisted laparoscopic pancreatic surgery procedures, morbidity was 26%, mortality was 2.2%, the conversion rate was 10.4%, and the fistula rate was 20.9%. The majority were grade A fistulas not requiring any treatment (Surg. Endosc. 2010; 24:1646-57). He said that robotic surgery "enables difficult technical maneuvers to be performed that facilitate the success of pancreatic minimally invasive surgery. The results in this series demonstrate feasibility and safety with clinical outcomes."
In a separate study, investigators who compared 32 open vs. 28 laparoscopic vs. 17 robotic distal pancreatectomies found that all three procedures resulted in a similar cost, while the robotic group had a shorter hospital stay, a higher rate of spleen-preserving cases, and an increased operative time, compared with patients in the other two groups (Surgery 2010;148:814-23).
Pelvic indications that are becoming the gold standard for robotic surgery include robotic prostatectomy and resection for rectal cancer. "When you have a very deep and complex surgical field like the pelvis, and you need to do a radical resection for cancer while at the same time sparing nerves that can be important for urinary function, the robot combines three-dimensional vision plus improved functionality of the instruments, allowing for a more precise dissection," Dr. Giulianotti said.
Dr. Leela M. Prasad, chief of the division of colon and rectal surgery at the University of Illinois at Chicago, described robotic-assisted surgery for rectal cancer as "far superior to anything we know right now," especially in overweight males. According to combined results from three published studies, circumferential margin positivity ranged from 0 to 7%, which, Dr. Prasad said, is significantly lower than the average number of positive margins reported following open and laparoscopic rectal surgery procedures (14% and 16%, respectively) (Ann. Surg. Oncol. 2007;14:3168-73; Surg. Oncol. 2009;16:1480-7; Dis. Colon Rectum 2009;52:1824-30).
At the present time, however, in his clinical experience there are no significant differences in outcomes between robotic-assisted and laparoscopically assisted open surgery for colon cancer. "We looked at our data, and it did not make any difference robotically or laparoscopically in terms of length of stay, lymph node harvest, or survival," he said. A recent study from Korean investigators supports those findings (Surg. Endosc. 2012 Dec. 13).
Dr. Giulianotti estimated that there are more than 1,600 robotic surgery systems in the United States. Some of them are devoted to urologic or gynecologic applications, "but in the majority of hospitals there is a growing tendency to use the robot as part of a multidisciplinary program," he said. "That means at the same institution different surgical teams on different days are using the robot for different indications. Of the 1,600 systems out there, I would estimate that about half are used for general surgery."
While hospital administrators have warmed up to the idea of initiating robotic surgery programs at their institutions in recent years, Dr. Giulianotti and Dr. Prasad both called for a culture change in terms of how surgeons and medical educators think about the robot’s place in clinical medicine. "There is a cultural resistance in accepting big changes in surgery," Dr. Giulianotti said. "Academic medical institutions can play a key role here. They need to start teaching robotics in a mandatory way by offering training opportunities in a lab. We also believe that simulation has a big future in this kind of training."
The economic investment required to launch a robotic surgery program is another challenge. "Some hospitals are only concerned about the cost," he said. "When you are doing the same procedure laparoscopically, the cost is probably about 30% less. In the future I hope we can better impact the overall economic aspect of surgical procedures. That means speeding up the postoperative and outpatient treatment of some pathologies. We are already doing Nissen fundoplication and gallbladder removal with robotic surgery, and patients are being discharged a few hours after the surgery."
Dr. Prasad said the field will be poised to further advance when makers of robotic surgery devices and instruments improve on existing technology. "I am using the same robot and instruments as I was using 3 years ago," he said. "I think we need new technologies – including a smaller robot to cut down the costs and speed up the operations. This will make it easier for patients."
To launch a successful robotic surgery program in this day and age, "you need a good team of nurses and surgeons who are dedicated to doing robotic surgery," Dr. Prasad concluded. "It is dangerous in the hands of a surgeon who is not qualified."
Dr. Giulianotti said that he had no relevant financial conflicts to disclose. Dr. Prasad disclosed that he has received honoraria from Intuitive Surgical, Ethicon Endo-Surgery, and Covidien.
New strain of norovirus wreaks havoc
A new strain of norovirus, identified last March in Australia, has swept through the United States, causing more than half of the 2012 outbreaks.
GII.4 Sydney is implicated in 53% of the outbreaks that occurred from September to December, Leslie Barclay and her colleagues reported in the Jan. 24 issue of the Morbidity and Mortality Weekly Report (2013;62:55).
The other outbreaks were caused by 10 different GI and GII genotypes, including GII.4 New Orleans, said Ms. Barclay of the National Calicivirus Laboratory at the Centers for Disease Control and Prevention, and her coauthors. However, she noted, the GII.4 Sydney virus appears to have replaced the New Orleans strain as the predominant pathogen.
GII.4 Sydney was implicated in 19% of the September norovirus outbreaks; 46% of the October outbreaks; and 58% of outbreaks in both November and December, Ms. Barclay and her coinvestigators said.
The report noted that 51% of the outbreaks were direct, person-to-person transmission; 20% were foodborne; and 1% waterborne. For the other outbreaks, the mode of transmission remains unknown.
The sudden appearance of a new norovirus strain isn’t unusual or unexpected, Dr. Aron J. Hall said in an interview.
"This group of viruses is constantly evolving. New strains develop all the time," said Dr. Hall, an epidemiologist with CDC’s Division of Viral Diseases. "This is following the same pattern we’ve seen for the past 10 years, with a new strain evolving every 2-3 years."
Since the norovirus season doesn’t normally peak until January, it’s too soon to say whether GII.4 Sydney is more virulent, either in the number or severity of cases it causes.
"Every time a new strain emerges, there’s a potential for increased disease activity, numbers of outbreaks, and severity. But sometimes they simply replace the predominant strain with no increases. We have to wait until we’ve tallied all the numbers before we can get an assessment of this season’s magnitude and impact," he said.
GII.4 Sydney was first identified in Australia, and rapidly moved to Japan, the Netherlands, and the United Kingdom, according to a Jan. 4 report (Eurosurv. 2013;18:1)
In early December and late November, the ProMed disease reporting system reported a 64% increase in confirmed norovirus lab reports in England and Wales, and increased deaths in elderly Japanese patients. Molecular assays pinpointed the cause as the new variant of norovirus, the Eurosurveillance researchers noted.
The virus is highly transmissible; exposure to as few as 18 viral particles can cause illness, according to a presentation by Jan Vinjé, Ph.D., head of the National Calicivirus Laboratory. And since about 30% of cases are asymptomatic, the transmission rate can be extremely high.
"These are the two characteristics that explain why norovirus is such a serious public health challenge," Dr. Hall said. "It takes a very tiny amount of exposure to make someone sick, and someone who is sick is shedding billions of viral particles. They can shed virus without any symptoms, so they don’t know they have it and there’s no way to advise them about how to avoid spreading it."
The illness is characterized by vomiting and/or diarrhea with abdominal cramps and low-grade fever. Most patients recover within 12-72 hours, but about 10% need hospitalization and fluid therapy. Severe illness and/or death are most common among the elderly and people with chronic illnesses.
The United States sees about 21 million cases annually; 800 will result in death. A norovirus outbreak can overwhelm local medical resources; the virus causes nearly 2 million outpatient visits and 400,000 emergency department visits each year. The combined annual cost reaches $2 billion, according to the CDC.
As federal employees, Dr. Hall and Ms. Barclay have no financial disclosures.
GII.4 Sydney, Leslie Barclay, Morbidity and Mortality Weekly Report, GII.4 New Orleans, National Calicivirus Laboratory at the Centers for Disease Control and Prevention, Dr. Aron J. Hall, CDC’s Division of Viral Diseases,
A new strain of norovirus, identified last March in Australia, has swept through the United States, causing more than half of the 2012 outbreaks.
GII.4 Sydney is implicated in 53% of the outbreaks that occurred from September to December, Leslie Barclay and her colleagues reported in the Jan. 24 issue of the Morbidity and Mortality Weekly Report (2013;62:55).
The other outbreaks were caused by 10 different GI and GII genotypes, including GII.4 New Orleans, said Ms. Barclay of the National Calicivirus Laboratory at the Centers for Disease Control and Prevention, and her coauthors. However, she noted, the GII.4 Sydney virus appears to have replaced the New Orleans strain as the predominant pathogen.
GII.4 Sydney was implicated in 19% of the September norovirus outbreaks; 46% of the October outbreaks; and 58% of outbreaks in both November and December, Ms. Barclay and her coinvestigators said.
The report noted that 51% of the outbreaks were direct, person-to-person transmission; 20% were foodborne; and 1% waterborne. For the other outbreaks, the mode of transmission remains unknown.
The sudden appearance of a new norovirus strain isn’t unusual or unexpected, Dr. Aron J. Hall said in an interview.
"This group of viruses is constantly evolving. New strains develop all the time," said Dr. Hall, an epidemiologist with CDC’s Division of Viral Diseases. "This is following the same pattern we’ve seen for the past 10 years, with a new strain evolving every 2-3 years."
Since the norovirus season doesn’t normally peak until January, it’s too soon to say whether GII.4 Sydney is more virulent, either in the number or severity of cases it causes.
"Every time a new strain emerges, there’s a potential for increased disease activity, numbers of outbreaks, and severity. But sometimes they simply replace the predominant strain with no increases. We have to wait until we’ve tallied all the numbers before we can get an assessment of this season’s magnitude and impact," he said.
GII.4 Sydney was first identified in Australia, and rapidly moved to Japan, the Netherlands, and the United Kingdom, according to a Jan. 4 report (Eurosurv. 2013;18:1)
In early December and late November, the ProMed disease reporting system reported a 64% increase in confirmed norovirus lab reports in England and Wales, and increased deaths in elderly Japanese patients. Molecular assays pinpointed the cause as the new variant of norovirus, the Eurosurveillance researchers noted.
The virus is highly transmissible; exposure to as few as 18 viral particles can cause illness, according to a presentation by Jan Vinjé, Ph.D., head of the National Calicivirus Laboratory. And since about 30% of cases are asymptomatic, the transmission rate can be extremely high.
"These are the two characteristics that explain why norovirus is such a serious public health challenge," Dr. Hall said. "It takes a very tiny amount of exposure to make someone sick, and someone who is sick is shedding billions of viral particles. They can shed virus without any symptoms, so they don’t know they have it and there’s no way to advise them about how to avoid spreading it."
The illness is characterized by vomiting and/or diarrhea with abdominal cramps and low-grade fever. Most patients recover within 12-72 hours, but about 10% need hospitalization and fluid therapy. Severe illness and/or death are most common among the elderly and people with chronic illnesses.
The United States sees about 21 million cases annually; 800 will result in death. A norovirus outbreak can overwhelm local medical resources; the virus causes nearly 2 million outpatient visits and 400,000 emergency department visits each year. The combined annual cost reaches $2 billion, according to the CDC.
As federal employees, Dr. Hall and Ms. Barclay have no financial disclosures.
A new strain of norovirus, identified last March in Australia, has swept through the United States, causing more than half of the 2012 outbreaks.
GII.4 Sydney is implicated in 53% of the outbreaks that occurred from September to December, Leslie Barclay and her colleagues reported in the Jan. 24 issue of the Morbidity and Mortality Weekly Report (2013;62:55).
The other outbreaks were caused by 10 different GI and GII genotypes, including GII.4 New Orleans, said Ms. Barclay of the National Calicivirus Laboratory at the Centers for Disease Control and Prevention, and her coauthors. However, she noted, the GII.4 Sydney virus appears to have replaced the New Orleans strain as the predominant pathogen.
GII.4 Sydney was implicated in 19% of the September norovirus outbreaks; 46% of the October outbreaks; and 58% of outbreaks in both November and December, Ms. Barclay and her coinvestigators said.
The report noted that 51% of the outbreaks were direct, person-to-person transmission; 20% were foodborne; and 1% waterborne. For the other outbreaks, the mode of transmission remains unknown.
The sudden appearance of a new norovirus strain isn’t unusual or unexpected, Dr. Aron J. Hall said in an interview.
"This group of viruses is constantly evolving. New strains develop all the time," said Dr. Hall, an epidemiologist with CDC’s Division of Viral Diseases. "This is following the same pattern we’ve seen for the past 10 years, with a new strain evolving every 2-3 years."
Since the norovirus season doesn’t normally peak until January, it’s too soon to say whether GII.4 Sydney is more virulent, either in the number or severity of cases it causes.
"Every time a new strain emerges, there’s a potential for increased disease activity, numbers of outbreaks, and severity. But sometimes they simply replace the predominant strain with no increases. We have to wait until we’ve tallied all the numbers before we can get an assessment of this season’s magnitude and impact," he said.
GII.4 Sydney was first identified in Australia, and rapidly moved to Japan, the Netherlands, and the United Kingdom, according to a Jan. 4 report (Eurosurv. 2013;18:1)
In early December and late November, the ProMed disease reporting system reported a 64% increase in confirmed norovirus lab reports in England and Wales, and increased deaths in elderly Japanese patients. Molecular assays pinpointed the cause as the new variant of norovirus, the Eurosurveillance researchers noted.
The virus is highly transmissible; exposure to as few as 18 viral particles can cause illness, according to a presentation by Jan Vinjé, Ph.D., head of the National Calicivirus Laboratory. And since about 30% of cases are asymptomatic, the transmission rate can be extremely high.
"These are the two characteristics that explain why norovirus is such a serious public health challenge," Dr. Hall said. "It takes a very tiny amount of exposure to make someone sick, and someone who is sick is shedding billions of viral particles. They can shed virus without any symptoms, so they don’t know they have it and there’s no way to advise them about how to avoid spreading it."
The illness is characterized by vomiting and/or diarrhea with abdominal cramps and low-grade fever. Most patients recover within 12-72 hours, but about 10% need hospitalization and fluid therapy. Severe illness and/or death are most common among the elderly and people with chronic illnesses.
The United States sees about 21 million cases annually; 800 will result in death. A norovirus outbreak can overwhelm local medical resources; the virus causes nearly 2 million outpatient visits and 400,000 emergency department visits each year. The combined annual cost reaches $2 billion, according to the CDC.
As federal employees, Dr. Hall and Ms. Barclay have no financial disclosures.
GII.4 Sydney, Leslie Barclay, Morbidity and Mortality Weekly Report, GII.4 New Orleans, National Calicivirus Laboratory at the Centers for Disease Control and Prevention, Dr. Aron J. Hall, CDC’s Division of Viral Diseases,
GII.4 Sydney, Leslie Barclay, Morbidity and Mortality Weekly Report, GII.4 New Orleans, National Calicivirus Laboratory at the Centers for Disease Control and Prevention, Dr. Aron J. Hall, CDC’s Division of Viral Diseases,
FROM THE MORBIDITY AND MORTALITY WEEKLY REPORT
Major Finding: A new strain of norovirus caused 53% of outbreaks in the United States from September-December 2012.
Data Source: These statistics were obtained from the Centers for Disease Control and Prevention’s National Calicivirus Laboratory
Disclosures: As federal employees, Dr. Hall and Ms. Barclay have no financial disclosures.
Lymph node count may not be best predictor of colon cancer survival
Lymph node counts of 12 or higher, widely considered a key marker of surgical quality in colon cancer resection, had no significant effect on 5-year survival rates in settings where surgeons are audited and credentialed and where surgical techniques are standardized.
Lymph node (LN) count is one of several measures used to determine the extent of surgical resection and an indicator of clear surgical margins, which have been assumed to result in better survival outcomes for colon cancer patients. A new data analysis, however, of the COST (Clinical Outcomes of Surgical Therapy) trial suggests reevaluating the use of surgical surrogates such as 12 LNs and margins.
The COST trial, a large, multicenter randomized trial of colon cancer procedures, compared outcomes for laparoscopic and open techniques in treating colon adenocarcinoma. The trial collected data on a number of surgical variables, including tumor location and LN count. A total of 787 patients were included: 267 with stage I disease, 284 with stage II, and 236 with stage III. Their median age was 70 years, and 50% were male.
In the current study, Dr. Kellie L. Mathis of the Mayo Clinic in Rochester, Minn., and her colleagues found that 5-year overall and disease-free survival were not influenced by LN count of above or below 12 (Ann. Surg. 2012;257:102-7 [doi:10.1097/SLA.0b013e318260a8e6]). When they adjusted for age and cancer stage, LN count was seen as not predictive of overall or disease-free survival (P = .60).
Other surgical surrogates, including total bowel length, margins, or mesenteric length, likewise did not have a significant effect on survival (P greater than .05 for all), nor did tumor location (right, left, or sigmoid), surgical technique (laparoscopic or open), and sex. Only patient age and cancer stage were found to be predictive of survival.
"On the basis of abundant literature and the acceptance of the 12 LN count as a surgical quality surrogate by National Quality Forum, most would expect the 12 LN count or other surgical variables to be predictive of survival," Dr. Mathis and her associates wrote. However, they hypothesized that procedural standardization, monitoring, and credentialing may provide a better strategy for quality control.
Overall 5-year survival results from the COST trial, they noted, were 77.2% – better than national rates for comparable patient groups in the same time period. All enrolling surgeons underwent pretrial credentialing and had performed a minimum of 20 laparoscopic colon resections, for which they had submitted operative and pathology reports. All laparoscopic resections were video recorded, and videos were randomly audited by an external review committee.
If the observations in the current study can be validated by others, Dr. Mathis and her colleagues said, "we submit that now is the time to invest in the development of technical quality control programs that directly measure and monitor surgical procedures."
Dr. Mathis and her colleagues stated that they had no conflicts of interest related to their findings.
Lymph node counts of 12 or higher, widely considered a key marker of surgical quality in colon cancer resection, had no significant effect on 5-year survival rates in settings where surgeons are audited and credentialed and where surgical techniques are standardized.
Lymph node (LN) count is one of several measures used to determine the extent of surgical resection and an indicator of clear surgical margins, which have been assumed to result in better survival outcomes for colon cancer patients. A new data analysis, however, of the COST (Clinical Outcomes of Surgical Therapy) trial suggests reevaluating the use of surgical surrogates such as 12 LNs and margins.
The COST trial, a large, multicenter randomized trial of colon cancer procedures, compared outcomes for laparoscopic and open techniques in treating colon adenocarcinoma. The trial collected data on a number of surgical variables, including tumor location and LN count. A total of 787 patients were included: 267 with stage I disease, 284 with stage II, and 236 with stage III. Their median age was 70 years, and 50% were male.
In the current study, Dr. Kellie L. Mathis of the Mayo Clinic in Rochester, Minn., and her colleagues found that 5-year overall and disease-free survival were not influenced by LN count of above or below 12 (Ann. Surg. 2012;257:102-7 [doi:10.1097/SLA.0b013e318260a8e6]). When they adjusted for age and cancer stage, LN count was seen as not predictive of overall or disease-free survival (P = .60).
Other surgical surrogates, including total bowel length, margins, or mesenteric length, likewise did not have a significant effect on survival (P greater than .05 for all), nor did tumor location (right, left, or sigmoid), surgical technique (laparoscopic or open), and sex. Only patient age and cancer stage were found to be predictive of survival.
"On the basis of abundant literature and the acceptance of the 12 LN count as a surgical quality surrogate by National Quality Forum, most would expect the 12 LN count or other surgical variables to be predictive of survival," Dr. Mathis and her associates wrote. However, they hypothesized that procedural standardization, monitoring, and credentialing may provide a better strategy for quality control.
Overall 5-year survival results from the COST trial, they noted, were 77.2% – better than national rates for comparable patient groups in the same time period. All enrolling surgeons underwent pretrial credentialing and had performed a minimum of 20 laparoscopic colon resections, for which they had submitted operative and pathology reports. All laparoscopic resections were video recorded, and videos were randomly audited by an external review committee.
If the observations in the current study can be validated by others, Dr. Mathis and her colleagues said, "we submit that now is the time to invest in the development of technical quality control programs that directly measure and monitor surgical procedures."
Dr. Mathis and her colleagues stated that they had no conflicts of interest related to their findings.
Lymph node counts of 12 or higher, widely considered a key marker of surgical quality in colon cancer resection, had no significant effect on 5-year survival rates in settings where surgeons are audited and credentialed and where surgical techniques are standardized.
Lymph node (LN) count is one of several measures used to determine the extent of surgical resection and an indicator of clear surgical margins, which have been assumed to result in better survival outcomes for colon cancer patients. A new data analysis, however, of the COST (Clinical Outcomes of Surgical Therapy) trial suggests reevaluating the use of surgical surrogates such as 12 LNs and margins.
The COST trial, a large, multicenter randomized trial of colon cancer procedures, compared outcomes for laparoscopic and open techniques in treating colon adenocarcinoma. The trial collected data on a number of surgical variables, including tumor location and LN count. A total of 787 patients were included: 267 with stage I disease, 284 with stage II, and 236 with stage III. Their median age was 70 years, and 50% were male.
In the current study, Dr. Kellie L. Mathis of the Mayo Clinic in Rochester, Minn., and her colleagues found that 5-year overall and disease-free survival were not influenced by LN count of above or below 12 (Ann. Surg. 2012;257:102-7 [doi:10.1097/SLA.0b013e318260a8e6]). When they adjusted for age and cancer stage, LN count was seen as not predictive of overall or disease-free survival (P = .60).
Other surgical surrogates, including total bowel length, margins, or mesenteric length, likewise did not have a significant effect on survival (P greater than .05 for all), nor did tumor location (right, left, or sigmoid), surgical technique (laparoscopic or open), and sex. Only patient age and cancer stage were found to be predictive of survival.
"On the basis of abundant literature and the acceptance of the 12 LN count as a surgical quality surrogate by National Quality Forum, most would expect the 12 LN count or other surgical variables to be predictive of survival," Dr. Mathis and her associates wrote. However, they hypothesized that procedural standardization, monitoring, and credentialing may provide a better strategy for quality control.
Overall 5-year survival results from the COST trial, they noted, were 77.2% – better than national rates for comparable patient groups in the same time period. All enrolling surgeons underwent pretrial credentialing and had performed a minimum of 20 laparoscopic colon resections, for which they had submitted operative and pathology reports. All laparoscopic resections were video recorded, and videos were randomly audited by an external review committee.
If the observations in the current study can be validated by others, Dr. Mathis and her colleagues said, "we submit that now is the time to invest in the development of technical quality control programs that directly measure and monitor surgical procedures."
Dr. Mathis and her colleagues stated that they had no conflicts of interest related to their findings.
FROM ANNALS OF SURGERY
Major Finding: Lymph node count of 12 or higher was not associated with better 5-year survival rates in patients undergoing surgery to treat colon cancer.
Data Source: A secondary analysis of data from the COST trial comparing laparoscopic vs. open colectomy in 787patients with stages I-III colon cancer.
Disclosures: Dr. Mathis and colleagues stated that they had no conflicts of interest related to their findings.
Staged surgery found unnecessary for colorectal tumors
PALM BEACH, FLA. – Simultaneous and staged removal of primary colorectal cancer and associated liver metastases worked equally well, a review of more than 1,000 cases at four international centers has shown.
"Long-term outcome of patients with synchronous colorectal liver metastases is dictated by biology, not by surgical strategy," Dr. Timothy M. Pawlik, FACS, said at the annual meeting of the Southern Surgical Association.
About a quarter of patients with advanced colorectal cancer present with synchronous liver metastases, and surgeons have used three different resection strategies. The classic approach has been staged removal of the primary, colorectal cancer first, followed by removal of the liver disease in a separate operation at a later time. In a much smaller number of cases, the staged approach is reversed, with initial excision of the liver disease followed later by removal of the primary tumor. The third option has been to do both excisions during a single operation. Until now, assessments that compared these approaches mostly have used data from a single center, and in several cases the reviews included relatively few patients, said Dr. Pawlik, a surgical oncologist at Johns Hopkins University in Baltimore.
The review done by Dr. Pawlik and his associates involved 1,004 patients treated between 1982 and 2011 at four major hepatobiliary centers in the United States and Europe. The series included 647 patients treated by removal of their primary colorectal tumor followed by a second surgery to remove their liver metastases, 329 patients who had both excisions done during a single operation, and 28 patients who had their liver metastases removed first and then had the primary tumor removed in a second operation.
The data showed no statistically significant difference for any outcome measure, including the incidence of complications, 90-day mortality, local recurrences, median survival, or rate of 5-year survival. For example, during a median follow-up of 34 months, the recurrence rate was 57% among patients treated with a classic, staged approach and 60% among patients who had a simultaneous excision, a difference that was not statistically significant. And in a multivariate regression analysis of factors associated with mortality during follow-up, simultaneous excision linked with an 8% higher mortality rate relative to a classic staged approach, but again the difference was not statistically significant.
"Long-term survival was associated with the location and extent of disease, but not with the surgical strategy," Dr. Pawlik said.
But he also qualified these findings with a couple of caveats. First, the findings "should only be extrapolated to academic centers, major hepatobiliary centers. The findings cannot be extrapolated to the community setting," he said.
Second, he agreed with the discussants of his report that a prospective, randomized trial should be done to definitively prove that surgical strategy has no impact on outcomes.
Dr. Pawlik said he had no relevant financial disclosures.
On Twitter @mitchelzoler
advanced colorectal cancer, synchronous liver metastases, resection strategies, removal of the primary, colorectal cancer, removal of the liver disease,
PALM BEACH, FLA. – Simultaneous and staged removal of primary colorectal cancer and associated liver metastases worked equally well, a review of more than 1,000 cases at four international centers has shown.
"Long-term outcome of patients with synchronous colorectal liver metastases is dictated by biology, not by surgical strategy," Dr. Timothy M. Pawlik, FACS, said at the annual meeting of the Southern Surgical Association.
About a quarter of patients with advanced colorectal cancer present with synchronous liver metastases, and surgeons have used three different resection strategies. The classic approach has been staged removal of the primary, colorectal cancer first, followed by removal of the liver disease in a separate operation at a later time. In a much smaller number of cases, the staged approach is reversed, with initial excision of the liver disease followed later by removal of the primary tumor. The third option has been to do both excisions during a single operation. Until now, assessments that compared these approaches mostly have used data from a single center, and in several cases the reviews included relatively few patients, said Dr. Pawlik, a surgical oncologist at Johns Hopkins University in Baltimore.
The review done by Dr. Pawlik and his associates involved 1,004 patients treated between 1982 and 2011 at four major hepatobiliary centers in the United States and Europe. The series included 647 patients treated by removal of their primary colorectal tumor followed by a second surgery to remove their liver metastases, 329 patients who had both excisions done during a single operation, and 28 patients who had their liver metastases removed first and then had the primary tumor removed in a second operation.
The data showed no statistically significant difference for any outcome measure, including the incidence of complications, 90-day mortality, local recurrences, median survival, or rate of 5-year survival. For example, during a median follow-up of 34 months, the recurrence rate was 57% among patients treated with a classic, staged approach and 60% among patients who had a simultaneous excision, a difference that was not statistically significant. And in a multivariate regression analysis of factors associated with mortality during follow-up, simultaneous excision linked with an 8% higher mortality rate relative to a classic staged approach, but again the difference was not statistically significant.
"Long-term survival was associated with the location and extent of disease, but not with the surgical strategy," Dr. Pawlik said.
But he also qualified these findings with a couple of caveats. First, the findings "should only be extrapolated to academic centers, major hepatobiliary centers. The findings cannot be extrapolated to the community setting," he said.
Second, he agreed with the discussants of his report that a prospective, randomized trial should be done to definitively prove that surgical strategy has no impact on outcomes.
Dr. Pawlik said he had no relevant financial disclosures.
On Twitter @mitchelzoler
PALM BEACH, FLA. – Simultaneous and staged removal of primary colorectal cancer and associated liver metastases worked equally well, a review of more than 1,000 cases at four international centers has shown.
"Long-term outcome of patients with synchronous colorectal liver metastases is dictated by biology, not by surgical strategy," Dr. Timothy M. Pawlik, FACS, said at the annual meeting of the Southern Surgical Association.
About a quarter of patients with advanced colorectal cancer present with synchronous liver metastases, and surgeons have used three different resection strategies. The classic approach has been staged removal of the primary, colorectal cancer first, followed by removal of the liver disease in a separate operation at a later time. In a much smaller number of cases, the staged approach is reversed, with initial excision of the liver disease followed later by removal of the primary tumor. The third option has been to do both excisions during a single operation. Until now, assessments that compared these approaches mostly have used data from a single center, and in several cases the reviews included relatively few patients, said Dr. Pawlik, a surgical oncologist at Johns Hopkins University in Baltimore.
The review done by Dr. Pawlik and his associates involved 1,004 patients treated between 1982 and 2011 at four major hepatobiliary centers in the United States and Europe. The series included 647 patients treated by removal of their primary colorectal tumor followed by a second surgery to remove their liver metastases, 329 patients who had both excisions done during a single operation, and 28 patients who had their liver metastases removed first and then had the primary tumor removed in a second operation.
The data showed no statistically significant difference for any outcome measure, including the incidence of complications, 90-day mortality, local recurrences, median survival, or rate of 5-year survival. For example, during a median follow-up of 34 months, the recurrence rate was 57% among patients treated with a classic, staged approach and 60% among patients who had a simultaneous excision, a difference that was not statistically significant. And in a multivariate regression analysis of factors associated with mortality during follow-up, simultaneous excision linked with an 8% higher mortality rate relative to a classic staged approach, but again the difference was not statistically significant.
"Long-term survival was associated with the location and extent of disease, but not with the surgical strategy," Dr. Pawlik said.
But he also qualified these findings with a couple of caveats. First, the findings "should only be extrapolated to academic centers, major hepatobiliary centers. The findings cannot be extrapolated to the community setting," he said.
Second, he agreed with the discussants of his report that a prospective, randomized trial should be done to definitively prove that surgical strategy has no impact on outcomes.
Dr. Pawlik said he had no relevant financial disclosures.
On Twitter @mitchelzoler
advanced colorectal cancer, synchronous liver metastases, resection strategies, removal of the primary, colorectal cancer, removal of the liver disease,
advanced colorectal cancer, synchronous liver metastases, resection strategies, removal of the primary, colorectal cancer, removal of the liver disease,
AT THE ANNUAL MEETING OF THE SOUTHERN SURGICAL ASSOCIATION
Major Finding: Removal of a colorectal tumor and synchronous liver metastases in a single operation or in two staged surgeries had equal outcomes.
Data Source: A review of 1,004 patients who underwent surgery to excise a primary colorectal tumor and synchronous liver metastases at four major hepatobiliary centers.
Disclosures: Dr. Pawlik said he had no relevant financial disclosures.
Restrictive transfusion strategy superior for severe acute GI bleeding
For patients with severe acute GI bleeding, outcomes are significantly better when a restrictive transfusion strategy is used – limiting the hemoglobin threshold to 7 g/dL – rather than a liberal transfusion strategy allowing a 9 g/dL threshold, according to a report published online Jan. 2 in the New England Journal of Medicine.
In a single-center randomized controlled trial involving 889 patients, the restrictive transfusion strategy resulted in significantly lower mortality, lower rates of rebleeding, less frequent need for rescue therapy, fewer complications, and shorter hospitalizations than did the liberal transfusion strategy. "Our results suggest that in patients with acute GI bleeding, a strategy of not performing transfusion until the hemoglobin concentration falls below 7 g/dL is a safe and effective approach," said Dr. Càndid Villanueva of Hospital de Sant Pau, Barcelona, and his associates.
"Current international guidelines recommend decreasing the hemoglobin threshold level for transfusion ... from 10g/dL to 7 g/dL" in such patients, but these recommendations are based on trials involving critically ill patients with normovolemic anemia that did not include subjects with acute bleeding. "Transfusion requirements may be different for patients with acute hemorrhage due to factors such as hemodynamic instability or rapid onset of anemia" resulting from extremely low hemoglobin levels.
In particular, results of animal studies suggest that transfusion can be especially harmful in patients with bleeding from portal hypertension sources, "since restitution of blood volume after hemorrhage can lead to a rebound increase in portal pressure, which is associated with a risk of rebleeding," the investigators noted.
To examine the effects of different transfusion strategies in this setting, Dr. Villanueva and his colleagues enrolled adults who presented with hematemesis, melena, or both, randomly assigning 444 to receive restrictive transfusion (with a target range for the posttransfusion hemoglobin level of 7-9 g/dL) and 445 to receive liberal transfusion (with a target range of 9-11 g/dL).
The study protocol permitted transfusions to be administered at the discretion of the attending physician any time symptoms or signs of anemia developed, massive bleeding occurred, or surgical intervention was needed, as well as when hemoglobin levels dipped below the assigned threshold.
All the study subjects underwent emergency gastroscopy within 6 hours of presentation, with appropriate treatment when the source of the bleeding was identified. Diagnoses included peptic ulcer, esophageal varices, cirrhosis, portal hypertension, and nonvariceal lesions.
The primary outcome measure, mortality from any cause at 45 days, was significantly lower in the restrictive-strategy group (5%) than the liberal-strategy group (9%). Death resulted from uncontrolled bleeding in 0.7% vs 3.1% of the 2 groups, respectively, the researchers said (N. Engl. J. Med. 2013 Jan. 2 [doi:10.1056/NEJMoa1211801]).
The rate of rebleeding also was significantly lower with the restrictive strategy (10% vs. 16%), and length of hospital stay was significantly shorter. In addition, rescue therapy for esophageal varices with balloon tamponade or a transjugular intrahepatic portosystemic shunt was required less often in the restrictive-strategy group than in the liberal-strategy group, as was emergency surgery to control further bleeding from peptic ulcer.
The rate of overall complications was significantly lower with the restrictive strategy (40%) than with the liberal strategy (48%), as was the rate of serious adverse events. In addition, transfusion reactions and cardiac events such as pulmonary edema were more frequent with the liberal strategy.
"Our results are consistent with those from previous observational studies and randomized trials performed in other settings, which have shown that a restrictive transfusion strategy did not increase, and even decreased, the mortality observed with a liberal transfusion strategy," Dr. Villanueva and his associates said.
This important study "provides long-awaited evidence to guide practice and justify current recommendations for the management of upper GI bleeding," said Dr. Loren Laine.
A restrictive transfusion strategy reduced the relative risk of mortality at 45 days by 45%. Extrapolating the study findings, only 25 patients would have to be treated with a restrictive rather than a liberal transfusion strategy to avert one additional death.
Dr. Laine is at Yale University, New Haven, Conn., and the Veterans Affairs Connecticut Healthcare System, West Haven. He reported no financial conflicts of interest. These remarks were taken from his editorial accompanying Dr. Villanueva’s report (N. Engl. J. Med. 2013 Jan. 2 [doi:10.1056/NEJMe1212009]).
This important study "provides long-awaited evidence to guide practice and justify current recommendations for the management of upper GI bleeding," said Dr. Loren Laine.
A restrictive transfusion strategy reduced the relative risk of mortality at 45 days by 45%. Extrapolating the study findings, only 25 patients would have to be treated with a restrictive rather than a liberal transfusion strategy to avert one additional death.
Dr. Laine is at Yale University, New Haven, Conn., and the Veterans Affairs Connecticut Healthcare System, West Haven. He reported no financial conflicts of interest. These remarks were taken from his editorial accompanying Dr. Villanueva’s report (N. Engl. J. Med. 2013 Jan. 2 [doi:10.1056/NEJMe1212009]).
This important study "provides long-awaited evidence to guide practice and justify current recommendations for the management of upper GI bleeding," said Dr. Loren Laine.
A restrictive transfusion strategy reduced the relative risk of mortality at 45 days by 45%. Extrapolating the study findings, only 25 patients would have to be treated with a restrictive rather than a liberal transfusion strategy to avert one additional death.
Dr. Laine is at Yale University, New Haven, Conn., and the Veterans Affairs Connecticut Healthcare System, West Haven. He reported no financial conflicts of interest. These remarks were taken from his editorial accompanying Dr. Villanueva’s report (N. Engl. J. Med. 2013 Jan. 2 [doi:10.1056/NEJMe1212009]).
For patients with severe acute GI bleeding, outcomes are significantly better when a restrictive transfusion strategy is used – limiting the hemoglobin threshold to 7 g/dL – rather than a liberal transfusion strategy allowing a 9 g/dL threshold, according to a report published online Jan. 2 in the New England Journal of Medicine.
In a single-center randomized controlled trial involving 889 patients, the restrictive transfusion strategy resulted in significantly lower mortality, lower rates of rebleeding, less frequent need for rescue therapy, fewer complications, and shorter hospitalizations than did the liberal transfusion strategy. "Our results suggest that in patients with acute GI bleeding, a strategy of not performing transfusion until the hemoglobin concentration falls below 7 g/dL is a safe and effective approach," said Dr. Càndid Villanueva of Hospital de Sant Pau, Barcelona, and his associates.
"Current international guidelines recommend decreasing the hemoglobin threshold level for transfusion ... from 10g/dL to 7 g/dL" in such patients, but these recommendations are based on trials involving critically ill patients with normovolemic anemia that did not include subjects with acute bleeding. "Transfusion requirements may be different for patients with acute hemorrhage due to factors such as hemodynamic instability or rapid onset of anemia" resulting from extremely low hemoglobin levels.
In particular, results of animal studies suggest that transfusion can be especially harmful in patients with bleeding from portal hypertension sources, "since restitution of blood volume after hemorrhage can lead to a rebound increase in portal pressure, which is associated with a risk of rebleeding," the investigators noted.
To examine the effects of different transfusion strategies in this setting, Dr. Villanueva and his colleagues enrolled adults who presented with hematemesis, melena, or both, randomly assigning 444 to receive restrictive transfusion (with a target range for the posttransfusion hemoglobin level of 7-9 g/dL) and 445 to receive liberal transfusion (with a target range of 9-11 g/dL).
The study protocol permitted transfusions to be administered at the discretion of the attending physician any time symptoms or signs of anemia developed, massive bleeding occurred, or surgical intervention was needed, as well as when hemoglobin levels dipped below the assigned threshold.
All the study subjects underwent emergency gastroscopy within 6 hours of presentation, with appropriate treatment when the source of the bleeding was identified. Diagnoses included peptic ulcer, esophageal varices, cirrhosis, portal hypertension, and nonvariceal lesions.
The primary outcome measure, mortality from any cause at 45 days, was significantly lower in the restrictive-strategy group (5%) than the liberal-strategy group (9%). Death resulted from uncontrolled bleeding in 0.7% vs 3.1% of the 2 groups, respectively, the researchers said (N. Engl. J. Med. 2013 Jan. 2 [doi:10.1056/NEJMoa1211801]).
The rate of rebleeding also was significantly lower with the restrictive strategy (10% vs. 16%), and length of hospital stay was significantly shorter. In addition, rescue therapy for esophageal varices with balloon tamponade or a transjugular intrahepatic portosystemic shunt was required less often in the restrictive-strategy group than in the liberal-strategy group, as was emergency surgery to control further bleeding from peptic ulcer.
The rate of overall complications was significantly lower with the restrictive strategy (40%) than with the liberal strategy (48%), as was the rate of serious adverse events. In addition, transfusion reactions and cardiac events such as pulmonary edema were more frequent with the liberal strategy.
"Our results are consistent with those from previous observational studies and randomized trials performed in other settings, which have shown that a restrictive transfusion strategy did not increase, and even decreased, the mortality observed with a liberal transfusion strategy," Dr. Villanueva and his associates said.
For patients with severe acute GI bleeding, outcomes are significantly better when a restrictive transfusion strategy is used – limiting the hemoglobin threshold to 7 g/dL – rather than a liberal transfusion strategy allowing a 9 g/dL threshold, according to a report published online Jan. 2 in the New England Journal of Medicine.
In a single-center randomized controlled trial involving 889 patients, the restrictive transfusion strategy resulted in significantly lower mortality, lower rates of rebleeding, less frequent need for rescue therapy, fewer complications, and shorter hospitalizations than did the liberal transfusion strategy. "Our results suggest that in patients with acute GI bleeding, a strategy of not performing transfusion until the hemoglobin concentration falls below 7 g/dL is a safe and effective approach," said Dr. Càndid Villanueva of Hospital de Sant Pau, Barcelona, and his associates.
"Current international guidelines recommend decreasing the hemoglobin threshold level for transfusion ... from 10g/dL to 7 g/dL" in such patients, but these recommendations are based on trials involving critically ill patients with normovolemic anemia that did not include subjects with acute bleeding. "Transfusion requirements may be different for patients with acute hemorrhage due to factors such as hemodynamic instability or rapid onset of anemia" resulting from extremely low hemoglobin levels.
In particular, results of animal studies suggest that transfusion can be especially harmful in patients with bleeding from portal hypertension sources, "since restitution of blood volume after hemorrhage can lead to a rebound increase in portal pressure, which is associated with a risk of rebleeding," the investigators noted.
To examine the effects of different transfusion strategies in this setting, Dr. Villanueva and his colleagues enrolled adults who presented with hematemesis, melena, or both, randomly assigning 444 to receive restrictive transfusion (with a target range for the posttransfusion hemoglobin level of 7-9 g/dL) and 445 to receive liberal transfusion (with a target range of 9-11 g/dL).
The study protocol permitted transfusions to be administered at the discretion of the attending physician any time symptoms or signs of anemia developed, massive bleeding occurred, or surgical intervention was needed, as well as when hemoglobin levels dipped below the assigned threshold.
All the study subjects underwent emergency gastroscopy within 6 hours of presentation, with appropriate treatment when the source of the bleeding was identified. Diagnoses included peptic ulcer, esophageal varices, cirrhosis, portal hypertension, and nonvariceal lesions.
The primary outcome measure, mortality from any cause at 45 days, was significantly lower in the restrictive-strategy group (5%) than the liberal-strategy group (9%). Death resulted from uncontrolled bleeding in 0.7% vs 3.1% of the 2 groups, respectively, the researchers said (N. Engl. J. Med. 2013 Jan. 2 [doi:10.1056/NEJMoa1211801]).
The rate of rebleeding also was significantly lower with the restrictive strategy (10% vs. 16%), and length of hospital stay was significantly shorter. In addition, rescue therapy for esophageal varices with balloon tamponade or a transjugular intrahepatic portosystemic shunt was required less often in the restrictive-strategy group than in the liberal-strategy group, as was emergency surgery to control further bleeding from peptic ulcer.
The rate of overall complications was significantly lower with the restrictive strategy (40%) than with the liberal strategy (48%), as was the rate of serious adverse events. In addition, transfusion reactions and cardiac events such as pulmonary edema were more frequent with the liberal strategy.
"Our results are consistent with those from previous observational studies and randomized trials performed in other settings, which have shown that a restrictive transfusion strategy did not increase, and even decreased, the mortality observed with a liberal transfusion strategy," Dr. Villanueva and his associates said.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: Mortality from any cause at 45 days was significantly lower in the restrictive-strategy group (5%) than in the liberal-strategy group (9%).
Data Source: A single-center randomized controlled trial of 889 patients.
Disclosures: This study was supported in part by the Fundacio Investigacio Sant Pau. Dr. Villanueva reported no financial conflicts of interest; one of his associates reported ties to Sequana Medical.