User login
Pandemic public health measures may have mitigated Kawasaki disease
The social behavior associated with the COVID-19 pandemic may have reduced the incidence of Kawasaki disease, according to results of a cohort study of nearly 4,000 children.
The incidence of Kawasaki disease in the United States declined by 28.2% between 2018 and 2020, possibly as a result of factors including school closures, mask mandates, and reduced ambient pollution that might reduce exposure to Kawasaki disease (KD) in the environment, but a potential association has not been explored, wrote Jennifer A. Burney, PhD, of the University of California, San Diego, and colleagues.
KD received greater attention in the public and medical communities because of the emergence of multisystem inflammatory syndrome in children (MIS-C), which is similar to, but distinct from, KD, and because of the noticeable drop in KD cases during the pandemic, the researchers said.
In a multicenter cohort study published in JAMA Network Open , the researchers reviewed data from 2,461 consecutive patients with KD who were diagnosed between Jan. 1, 2018, and Dec. 31, 2020. They conducted a detailed analysis of analysis of 1,461 children with KD who were diagnosed between Jan. 1, 2002, and Nov. 15, 2021, at Rady Children’s Hospital San Diego (RCHSD), using data from before, during, and after the height of the pandemic. The median age of the children in the RCHSD analysis was 2.8 years, 62% were male, and 35% were Hispanic.
Overall, the prevalence of KD declined from 894 in 2018 to 646 in 2020, across the United States, but the decline was uneven, the researchers noted.
In the RCHSD cohort in San Diego, KD cases in children aged 1-5 years decreased significantly from 2020 to 2021 compared to the mean number of cases in previous years (22 vs. 44.9, P = .02). KD cases also decreased significantly among males and Asian children.
Notably, the occurrence of the KD clinical features of strawberry tongue, enlarged cervical lymph node, and subacute periungual desquamation decreased during 2020 compared with the baseline period, although only strawberry tongue reached statistical significance (39% vs. 63%, P = .04). The prevalence of patients with an enlarged lymph node was 21% in 2020 vs. 32% prior to the pandemic (P = .09); the prevalence of periungual desquamation during these periods was 47% vs. 58%, P = .16).
The researchers also used data from Census Block Groups (CBGs) to assess the impact of mobility metrics and environmental exposures on KD during the pandemic for the San Diego patient cohort. They found that KD cases during the pandemic were more likely to occur in neighborhoods of higher socioeconomic status, and that neighborhoods with lower levels of nitrous oxides had fewer KD cases.
Overall, “The reduction in KD case numbers coincided with masking, school closures, reduced circulation of respiratory viruses, and reduced air pollution,” the researchers wrote in their discussion of the findings. “A rebound in KD case numbers to prepandemic levels coincided with the lifting of mask mandates and, subsequently, the return to in-person schooling,” they wrote.
The study findings were limited by several factors including the small sample sizes, which also limit the interpretation of mobility and pollution data, the researchers noted. Other limitations include the high interannual variability of KD and the inclusion of 2021 rebound data from the San Diego region only.
“Although our original hypothesis was that shelter-in-place measures would track with reduced KD cases, this was not borne out by the San Diego region data. Instead, the San Diego case occurrence data suggest that exposures that triggered KD were more likely to occur in the home, with a shift toward households with higher SES during the pandemic,” the researchers noted. However, “The results presented here are consistent with a respiratory portal of entry for the trigger(s) of KD,” they said.
Study fails to validate its conclusions
“This study attempts to test the hypothesis that various social restrictions were associated with a decrease in rate of diagnosed Kawasaki disease cases during portions of the SARS-CoV-2 pandemic,” Mark Gorelik, MD, assistant professor of pediatrics at Columbia University, New York, said in an interview.
“However, it appears that it fails to achieve this conclusion and I disagree with the findings,” said Dr. Gorelik, who was not involved in the study but served as first author on an updated Kawasaki disease treatment guideline published earlier this spring in Arthritis & Rheumatology.
“The study does not find statistically significant associations either with shelter in place orders or with cell phone mobility data, as stated in the conclusion, directly contradicting its own claim,” Dr. Gorelik said. “Secondly, the study makes an assumption that various methods, especially the wearing of masks by children and school closures, had a significant effect on the spread of respiratory viruses. There are no prospective, population based, controlled real world studies that validate this claim, and two prospective controlled real-world studies that dispute this,” he emphasized. “Cloth masks and surgical masks, which were the types of masks worn by school students, are also known to have a nonsignificant and paltry – in the latter, certainly less than 50%, and perhaps as little as 10% – effect on the reduction of respiratory viral spread,” he added.
“Mechanistic studies on mask wearing may suggest some mask efficacy, but these studies are as valid as mechanistic studies showing the effect of various antifungal pharmaceuticals on the replication of SARS-CoV-2 virus in culture, meaning only valid as hypothesis generating, and ultimately the latter hypothesis failed to bear out,” Dr. Gorelik explained. “We do not know the reason why other respiratory viruses and non-SARS-CoV-2 coronaviruses declined during the pandemic, but we do know that despite this, the SARS-CoV-2 coronavirus itself did not appear to suffer the same fate. Thus, it is very possible that another factor was at work, and we know that during other viral pandemics, typically circulating viruses decline, potentially due to induction of interferon responses in hosts, in a general effect known as ‘viral interference,’ ” he said.
“Overall, we must have robust evidence to support benefits of hypotheses that have demonstrated clear damage to children during this pandemic (such as school closures), and this study fails to live up to that requirement,” Dr. Gorelik said.
The study was supported by the Gordon and Marilyn Macklin Foundation and the Patient-Centered Outcomes Research Institute. Dr. Burney and Dr. Gorelik had no financial conflicts to disclose.
The social behavior associated with the COVID-19 pandemic may have reduced the incidence of Kawasaki disease, according to results of a cohort study of nearly 4,000 children.
The incidence of Kawasaki disease in the United States declined by 28.2% between 2018 and 2020, possibly as a result of factors including school closures, mask mandates, and reduced ambient pollution that might reduce exposure to Kawasaki disease (KD) in the environment, but a potential association has not been explored, wrote Jennifer A. Burney, PhD, of the University of California, San Diego, and colleagues.
KD received greater attention in the public and medical communities because of the emergence of multisystem inflammatory syndrome in children (MIS-C), which is similar to, but distinct from, KD, and because of the noticeable drop in KD cases during the pandemic, the researchers said.
In a multicenter cohort study published in JAMA Network Open , the researchers reviewed data from 2,461 consecutive patients with KD who were diagnosed between Jan. 1, 2018, and Dec. 31, 2020. They conducted a detailed analysis of analysis of 1,461 children with KD who were diagnosed between Jan. 1, 2002, and Nov. 15, 2021, at Rady Children’s Hospital San Diego (RCHSD), using data from before, during, and after the height of the pandemic. The median age of the children in the RCHSD analysis was 2.8 years, 62% were male, and 35% were Hispanic.
Overall, the prevalence of KD declined from 894 in 2018 to 646 in 2020, across the United States, but the decline was uneven, the researchers noted.
In the RCHSD cohort in San Diego, KD cases in children aged 1-5 years decreased significantly from 2020 to 2021 compared to the mean number of cases in previous years (22 vs. 44.9, P = .02). KD cases also decreased significantly among males and Asian children.
Notably, the occurrence of the KD clinical features of strawberry tongue, enlarged cervical lymph node, and subacute periungual desquamation decreased during 2020 compared with the baseline period, although only strawberry tongue reached statistical significance (39% vs. 63%, P = .04). The prevalence of patients with an enlarged lymph node was 21% in 2020 vs. 32% prior to the pandemic (P = .09); the prevalence of periungual desquamation during these periods was 47% vs. 58%, P = .16).
The researchers also used data from Census Block Groups (CBGs) to assess the impact of mobility metrics and environmental exposures on KD during the pandemic for the San Diego patient cohort. They found that KD cases during the pandemic were more likely to occur in neighborhoods of higher socioeconomic status, and that neighborhoods with lower levels of nitrous oxides had fewer KD cases.
Overall, “The reduction in KD case numbers coincided with masking, school closures, reduced circulation of respiratory viruses, and reduced air pollution,” the researchers wrote in their discussion of the findings. “A rebound in KD case numbers to prepandemic levels coincided with the lifting of mask mandates and, subsequently, the return to in-person schooling,” they wrote.
The study findings were limited by several factors including the small sample sizes, which also limit the interpretation of mobility and pollution data, the researchers noted. Other limitations include the high interannual variability of KD and the inclusion of 2021 rebound data from the San Diego region only.
“Although our original hypothesis was that shelter-in-place measures would track with reduced KD cases, this was not borne out by the San Diego region data. Instead, the San Diego case occurrence data suggest that exposures that triggered KD were more likely to occur in the home, with a shift toward households with higher SES during the pandemic,” the researchers noted. However, “The results presented here are consistent with a respiratory portal of entry for the trigger(s) of KD,” they said.
Study fails to validate its conclusions
“This study attempts to test the hypothesis that various social restrictions were associated with a decrease in rate of diagnosed Kawasaki disease cases during portions of the SARS-CoV-2 pandemic,” Mark Gorelik, MD, assistant professor of pediatrics at Columbia University, New York, said in an interview.
“However, it appears that it fails to achieve this conclusion and I disagree with the findings,” said Dr. Gorelik, who was not involved in the study but served as first author on an updated Kawasaki disease treatment guideline published earlier this spring in Arthritis & Rheumatology.
“The study does not find statistically significant associations either with shelter in place orders or with cell phone mobility data, as stated in the conclusion, directly contradicting its own claim,” Dr. Gorelik said. “Secondly, the study makes an assumption that various methods, especially the wearing of masks by children and school closures, had a significant effect on the spread of respiratory viruses. There are no prospective, population based, controlled real world studies that validate this claim, and two prospective controlled real-world studies that dispute this,” he emphasized. “Cloth masks and surgical masks, which were the types of masks worn by school students, are also known to have a nonsignificant and paltry – in the latter, certainly less than 50%, and perhaps as little as 10% – effect on the reduction of respiratory viral spread,” he added.
“Mechanistic studies on mask wearing may suggest some mask efficacy, but these studies are as valid as mechanistic studies showing the effect of various antifungal pharmaceuticals on the replication of SARS-CoV-2 virus in culture, meaning only valid as hypothesis generating, and ultimately the latter hypothesis failed to bear out,” Dr. Gorelik explained. “We do not know the reason why other respiratory viruses and non-SARS-CoV-2 coronaviruses declined during the pandemic, but we do know that despite this, the SARS-CoV-2 coronavirus itself did not appear to suffer the same fate. Thus, it is very possible that another factor was at work, and we know that during other viral pandemics, typically circulating viruses decline, potentially due to induction of interferon responses in hosts, in a general effect known as ‘viral interference,’ ” he said.
“Overall, we must have robust evidence to support benefits of hypotheses that have demonstrated clear damage to children during this pandemic (such as school closures), and this study fails to live up to that requirement,” Dr. Gorelik said.
The study was supported by the Gordon and Marilyn Macklin Foundation and the Patient-Centered Outcomes Research Institute. Dr. Burney and Dr. Gorelik had no financial conflicts to disclose.
The social behavior associated with the COVID-19 pandemic may have reduced the incidence of Kawasaki disease, according to results of a cohort study of nearly 4,000 children.
The incidence of Kawasaki disease in the United States declined by 28.2% between 2018 and 2020, possibly as a result of factors including school closures, mask mandates, and reduced ambient pollution that might reduce exposure to Kawasaki disease (KD) in the environment, but a potential association has not been explored, wrote Jennifer A. Burney, PhD, of the University of California, San Diego, and colleagues.
KD received greater attention in the public and medical communities because of the emergence of multisystem inflammatory syndrome in children (MIS-C), which is similar to, but distinct from, KD, and because of the noticeable drop in KD cases during the pandemic, the researchers said.
In a multicenter cohort study published in JAMA Network Open , the researchers reviewed data from 2,461 consecutive patients with KD who were diagnosed between Jan. 1, 2018, and Dec. 31, 2020. They conducted a detailed analysis of analysis of 1,461 children with KD who were diagnosed between Jan. 1, 2002, and Nov. 15, 2021, at Rady Children’s Hospital San Diego (RCHSD), using data from before, during, and after the height of the pandemic. The median age of the children in the RCHSD analysis was 2.8 years, 62% were male, and 35% were Hispanic.
Overall, the prevalence of KD declined from 894 in 2018 to 646 in 2020, across the United States, but the decline was uneven, the researchers noted.
In the RCHSD cohort in San Diego, KD cases in children aged 1-5 years decreased significantly from 2020 to 2021 compared to the mean number of cases in previous years (22 vs. 44.9, P = .02). KD cases also decreased significantly among males and Asian children.
Notably, the occurrence of the KD clinical features of strawberry tongue, enlarged cervical lymph node, and subacute periungual desquamation decreased during 2020 compared with the baseline period, although only strawberry tongue reached statistical significance (39% vs. 63%, P = .04). The prevalence of patients with an enlarged lymph node was 21% in 2020 vs. 32% prior to the pandemic (P = .09); the prevalence of periungual desquamation during these periods was 47% vs. 58%, P = .16).
The researchers also used data from Census Block Groups (CBGs) to assess the impact of mobility metrics and environmental exposures on KD during the pandemic for the San Diego patient cohort. They found that KD cases during the pandemic were more likely to occur in neighborhoods of higher socioeconomic status, and that neighborhoods with lower levels of nitrous oxides had fewer KD cases.
Overall, “The reduction in KD case numbers coincided with masking, school closures, reduced circulation of respiratory viruses, and reduced air pollution,” the researchers wrote in their discussion of the findings. “A rebound in KD case numbers to prepandemic levels coincided with the lifting of mask mandates and, subsequently, the return to in-person schooling,” they wrote.
The study findings were limited by several factors including the small sample sizes, which also limit the interpretation of mobility and pollution data, the researchers noted. Other limitations include the high interannual variability of KD and the inclusion of 2021 rebound data from the San Diego region only.
“Although our original hypothesis was that shelter-in-place measures would track with reduced KD cases, this was not borne out by the San Diego region data. Instead, the San Diego case occurrence data suggest that exposures that triggered KD were more likely to occur in the home, with a shift toward households with higher SES during the pandemic,” the researchers noted. However, “The results presented here are consistent with a respiratory portal of entry for the trigger(s) of KD,” they said.
Study fails to validate its conclusions
“This study attempts to test the hypothesis that various social restrictions were associated with a decrease in rate of diagnosed Kawasaki disease cases during portions of the SARS-CoV-2 pandemic,” Mark Gorelik, MD, assistant professor of pediatrics at Columbia University, New York, said in an interview.
“However, it appears that it fails to achieve this conclusion and I disagree with the findings,” said Dr. Gorelik, who was not involved in the study but served as first author on an updated Kawasaki disease treatment guideline published earlier this spring in Arthritis & Rheumatology.
“The study does not find statistically significant associations either with shelter in place orders or with cell phone mobility data, as stated in the conclusion, directly contradicting its own claim,” Dr. Gorelik said. “Secondly, the study makes an assumption that various methods, especially the wearing of masks by children and school closures, had a significant effect on the spread of respiratory viruses. There are no prospective, population based, controlled real world studies that validate this claim, and two prospective controlled real-world studies that dispute this,” he emphasized. “Cloth masks and surgical masks, which were the types of masks worn by school students, are also known to have a nonsignificant and paltry – in the latter, certainly less than 50%, and perhaps as little as 10% – effect on the reduction of respiratory viral spread,” he added.
“Mechanistic studies on mask wearing may suggest some mask efficacy, but these studies are as valid as mechanistic studies showing the effect of various antifungal pharmaceuticals on the replication of SARS-CoV-2 virus in culture, meaning only valid as hypothesis generating, and ultimately the latter hypothesis failed to bear out,” Dr. Gorelik explained. “We do not know the reason why other respiratory viruses and non-SARS-CoV-2 coronaviruses declined during the pandemic, but we do know that despite this, the SARS-CoV-2 coronavirus itself did not appear to suffer the same fate. Thus, it is very possible that another factor was at work, and we know that during other viral pandemics, typically circulating viruses decline, potentially due to induction of interferon responses in hosts, in a general effect known as ‘viral interference,’ ” he said.
“Overall, we must have robust evidence to support benefits of hypotheses that have demonstrated clear damage to children during this pandemic (such as school closures), and this study fails to live up to that requirement,” Dr. Gorelik said.
The study was supported by the Gordon and Marilyn Macklin Foundation and the Patient-Centered Outcomes Research Institute. Dr. Burney and Dr. Gorelik had no financial conflicts to disclose.
FROM JAMA NETWORK OPEN
Updates in aspirin use, aducanumab, and CKD diagnostic criteria in geriatric medicine
I selected these topics as they were among the most discussed by my colleagues in geriatric medicine and inquired about by my primary care patients in geriatric medicine clinic. I hope that these updates provide primary care clinicians who care for older adults with more context and background information regarding new Alzheimer’s disease therapy to better answer patient inquiries, and to feel empowered to deprescribe aspirin and reframe the diagnostic criteria of chronic kidney disease (CKD).
Aspirin for primary prevention
It was welcome news in the geriatrics community when the United States Preventive Services Task Force updated their guidelines in April 2022 to recommend against the initiation of aspirin for primary prevention in adults aged 60 or older. This recommendation was based on studies that found that net benefits of CVD prevention in older adults are outweighed by risk of bleeding.1
The risk of bleeding increases with age and can occur in individuals without common risk factors for bleeding, such as prior gastrointestinal bleeding, peptic ulcer disease, concurrent NSAID use, or corticosteroid use.
While it may be easier to not initiate aspirin for primary prevention, deprescribing aspirin for patients who have been on aspirin long term for primary prevention presents more of a challenge. Modeling data from the USPTSF suggest stopping aspirin at age 75 for those taking aspirin for primary prevention.2
Behavioral change, particularly for patients who have been on aspirin for decades, can be difficult. A 2021 study by Green et al. found that language that resonates the most with older adults when deprescribing emphasized the side effects rather than statements such as “this will not help you” or “do not need anymore.”3
Aducanumab for mild cognitive impairment and mild Alzheimer’s dementia
One of the most discussed topics this past year is the Food and Drug Administration approval of aducanumab (brand name Aduhelm) in June 2021. Aducanumab is the first approved disease-modifying therapy for Alzheimer’s disease and the first drug approved for the treatment of Alzheimer’s disease since 2003. Aducanumab is an antiamyloid monoclonal antibody that was developed to reduce amyloid plaque in the brain, one of the features of Alzheimer’s disease pathology.
Uptake of aducanumab by dementia providers has been limited for several reasons. Firstly, the clinical significance of the drug remains in question. ENGAGE and EMERGE were the two main randomized clinical trials that studied the effect of aducanumab on amyloid burden and clinical stages of dementia over 18 months. While both studies demonstrated that aducanumab reduced amyloid burden based on neuroimaging and in cerebrospinal fluid, the ENGAGE trial found no difference in the stage of dementia. The EMERGE trial did note a small, statistically significant difference in stage of dementia, however the participants of the EMERGE trial had a faster rate of progression of dementia than the placebo participants in the ENGAGE trial, which could have contributed to the difference detected.4
Additionally, exclusion criteria for both trials call into question the generalizability of this study. Participants over age 85, with CKD, prior stroke, or transient ischemic attacks, or on anticoagulation were excluded. One of the drivers for the exclusion criteria is the increased risk of macro and microhemorrhages.
Thirty-five percent of research participants were incidentally noted to have brain edema, an abnormality called amyloid-related imaging abnormality or ARIA-E, that necessitated serial monitoring with brain MRIs. It is also important to highlight that inclusion of African American, Hispanic, and Latinx participants in these studies was less than 5%, despite a higher incidence of Alzheimer’s disease in these populations.5
Lastly, economic implications for the U.S. health care system with increased uptake of aducanumab could be enormous. Originally quoted at $56,000 yearly, Biogen, the maker of aducanumab, recently reduced annual costs to $28,200 per patient.
In April 2022, CMS released a statement that antiamyloid monoclonal antibodies and related services, including PET scans, would be covered under Medicare for those with mild cognitive impairment and mild Alzheimer’s dementia with confirmed presence of amyloid. A study by Mafi et al. estimated that aducanumab could cost Medicare between $7 billion and $37.4 billion annually based on lower and upper bound estimates of eligible Medicare beneficiaries.6
Overdiagnosis of CKD in older adults
The current diagnostic criteria of CKD, which is based on an estimated glomerular filtration rate (eGFR) of less than 60, has been up for debate, as glomerular filtration rate (GFR) physiologically decreases with age. Fixed thresholds can lead to underdiagnosis of CKD in younger adults and overdiagnosis of CKD in older adults. Age-adapted thresholds for the diagnosis of CKD have been proposed, with the suggestion of an eGFR threshold of 45mL/min/1.73 m2 for adults aged 65 and older.7
The clinical implication of using an age-adapted eGFR threshold definition was investigated in a 2021 cohort study by Liu et al.8 In this study, outcomes of adults diagnosed with CKD using a fixed threshold versus age-adapted threshold were compared with a healthy cohort.
A fixed threshold led to a 60% higher incidence of CKD diagnosis. However, incidence of renal failure and all-cause mortality in older adults with an eGFR between 45-59 /min/1.73 m2 with normal or mild albuminuria was of similar magnitude to the healthy cohort at 5 years of follow-up.
These findings support the use of age-adapted thresholds for the diagnosis of CKD in older adults, as an earlier diagnosis of mild CKD does not equate to clinical benefits, but could lead to harms of unnecessary interventions and patient anxiety.
Dr. Mengru “Ruru” Wang is a geriatrician and internist at the University of Washington, Seattle. She practices full-spectrum medicine, seeing patients in primary care, nursing homes, and acute care. Dr. Wang has no disclosures related to this piece.
References
1. Selak Vet al. Predicting bleeding risk to guide aspirin use for the primary prevention of cardiovascular disease: A cohort study. Ann Intern Med. 2019;170(6):357-68. doi: 10.7326/M18-2808.
2. US Preventive Services Task Force. Aspirin Use to Prevent Cardiovascular Disease: US Preventive Services Task Force Recommendation Statement. JAMA. 2022;327(16):1577-84. doi: 10.1001/jama.2022.4983.
3. Green AR et al. Assessment of patient-preferred language to achieve goal-aligned deprescribing in older adults. JAMA Netw Open. 2021;4(4):e212633. doi: 10.1001/jamanetworkopen.2021.2633.
4. Oh ES. Use of anti-amyloid therapy for Alzheimer’s disease in clinical practice. An update on Alzheimer’s disease diagnosis and therapeutics. Presentation at American Geriatrics Society Meeting, 2022. Orlando.
5. Amjad H. Issues of Access and Marginalization. An update on Alzheimer’s disease diagnosis and therapeutics. Presentation at: American Geriatrics Society Meeting, 2022. Orlando.
6. Mafi JN et al. Estimated annual spending on aducanumab in the U.S. Medicare program. JAMA Health Forum. 2022;3(1):e214495. doi: 10.1001/jamahealthforum.2021.4495.
7. Delanaye P et al. CKD: A call for an age-adapted definition. J Am Soc Nephrol. 2019;30(10):1785-1805. doi: 10.1681/ASN.2019030238.
8. Liu Pet al. Accounting for age in the definition of chronic kidney disease. JAMA Intern Med. 2021;181(10):1359-66. doi: 10.1001/jamainternmed.2021.4813.
I selected these topics as they were among the most discussed by my colleagues in geriatric medicine and inquired about by my primary care patients in geriatric medicine clinic. I hope that these updates provide primary care clinicians who care for older adults with more context and background information regarding new Alzheimer’s disease therapy to better answer patient inquiries, and to feel empowered to deprescribe aspirin and reframe the diagnostic criteria of chronic kidney disease (CKD).
Aspirin for primary prevention
It was welcome news in the geriatrics community when the United States Preventive Services Task Force updated their guidelines in April 2022 to recommend against the initiation of aspirin for primary prevention in adults aged 60 or older. This recommendation was based on studies that found that net benefits of CVD prevention in older adults are outweighed by risk of bleeding.1
The risk of bleeding increases with age and can occur in individuals without common risk factors for bleeding, such as prior gastrointestinal bleeding, peptic ulcer disease, concurrent NSAID use, or corticosteroid use.
While it may be easier to not initiate aspirin for primary prevention, deprescribing aspirin for patients who have been on aspirin long term for primary prevention presents more of a challenge. Modeling data from the USPTSF suggest stopping aspirin at age 75 for those taking aspirin for primary prevention.2
Behavioral change, particularly for patients who have been on aspirin for decades, can be difficult. A 2021 study by Green et al. found that language that resonates the most with older adults when deprescribing emphasized the side effects rather than statements such as “this will not help you” or “do not need anymore.”3
Aducanumab for mild cognitive impairment and mild Alzheimer’s dementia
One of the most discussed topics this past year is the Food and Drug Administration approval of aducanumab (brand name Aduhelm) in June 2021. Aducanumab is the first approved disease-modifying therapy for Alzheimer’s disease and the first drug approved for the treatment of Alzheimer’s disease since 2003. Aducanumab is an antiamyloid monoclonal antibody that was developed to reduce amyloid plaque in the brain, one of the features of Alzheimer’s disease pathology.
Uptake of aducanumab by dementia providers has been limited for several reasons. Firstly, the clinical significance of the drug remains in question. ENGAGE and EMERGE were the two main randomized clinical trials that studied the effect of aducanumab on amyloid burden and clinical stages of dementia over 18 months. While both studies demonstrated that aducanumab reduced amyloid burden based on neuroimaging and in cerebrospinal fluid, the ENGAGE trial found no difference in the stage of dementia. The EMERGE trial did note a small, statistically significant difference in stage of dementia, however the participants of the EMERGE trial had a faster rate of progression of dementia than the placebo participants in the ENGAGE trial, which could have contributed to the difference detected.4
Additionally, exclusion criteria for both trials call into question the generalizability of this study. Participants over age 85, with CKD, prior stroke, or transient ischemic attacks, or on anticoagulation were excluded. One of the drivers for the exclusion criteria is the increased risk of macro and microhemorrhages.
Thirty-five percent of research participants were incidentally noted to have brain edema, an abnormality called amyloid-related imaging abnormality or ARIA-E, that necessitated serial monitoring with brain MRIs. It is also important to highlight that inclusion of African American, Hispanic, and Latinx participants in these studies was less than 5%, despite a higher incidence of Alzheimer’s disease in these populations.5
Lastly, economic implications for the U.S. health care system with increased uptake of aducanumab could be enormous. Originally quoted at $56,000 yearly, Biogen, the maker of aducanumab, recently reduced annual costs to $28,200 per patient.
In April 2022, CMS released a statement that antiamyloid monoclonal antibodies and related services, including PET scans, would be covered under Medicare for those with mild cognitive impairment and mild Alzheimer’s dementia with confirmed presence of amyloid. A study by Mafi et al. estimated that aducanumab could cost Medicare between $7 billion and $37.4 billion annually based on lower and upper bound estimates of eligible Medicare beneficiaries.6
Overdiagnosis of CKD in older adults
The current diagnostic criteria of CKD, which is based on an estimated glomerular filtration rate (eGFR) of less than 60, has been up for debate, as glomerular filtration rate (GFR) physiologically decreases with age. Fixed thresholds can lead to underdiagnosis of CKD in younger adults and overdiagnosis of CKD in older adults. Age-adapted thresholds for the diagnosis of CKD have been proposed, with the suggestion of an eGFR threshold of 45mL/min/1.73 m2 for adults aged 65 and older.7
The clinical implication of using an age-adapted eGFR threshold definition was investigated in a 2021 cohort study by Liu et al.8 In this study, outcomes of adults diagnosed with CKD using a fixed threshold versus age-adapted threshold were compared with a healthy cohort.
A fixed threshold led to a 60% higher incidence of CKD diagnosis. However, incidence of renal failure and all-cause mortality in older adults with an eGFR between 45-59 /min/1.73 m2 with normal or mild albuminuria was of similar magnitude to the healthy cohort at 5 years of follow-up.
These findings support the use of age-adapted thresholds for the diagnosis of CKD in older adults, as an earlier diagnosis of mild CKD does not equate to clinical benefits, but could lead to harms of unnecessary interventions and patient anxiety.
Dr. Mengru “Ruru” Wang is a geriatrician and internist at the University of Washington, Seattle. She practices full-spectrum medicine, seeing patients in primary care, nursing homes, and acute care. Dr. Wang has no disclosures related to this piece.
References
1. Selak Vet al. Predicting bleeding risk to guide aspirin use for the primary prevention of cardiovascular disease: A cohort study. Ann Intern Med. 2019;170(6):357-68. doi: 10.7326/M18-2808.
2. US Preventive Services Task Force. Aspirin Use to Prevent Cardiovascular Disease: US Preventive Services Task Force Recommendation Statement. JAMA. 2022;327(16):1577-84. doi: 10.1001/jama.2022.4983.
3. Green AR et al. Assessment of patient-preferred language to achieve goal-aligned deprescribing in older adults. JAMA Netw Open. 2021;4(4):e212633. doi: 10.1001/jamanetworkopen.2021.2633.
4. Oh ES. Use of anti-amyloid therapy for Alzheimer’s disease in clinical practice. An update on Alzheimer’s disease diagnosis and therapeutics. Presentation at American Geriatrics Society Meeting, 2022. Orlando.
5. Amjad H. Issues of Access and Marginalization. An update on Alzheimer’s disease diagnosis and therapeutics. Presentation at: American Geriatrics Society Meeting, 2022. Orlando.
6. Mafi JN et al. Estimated annual spending on aducanumab in the U.S. Medicare program. JAMA Health Forum. 2022;3(1):e214495. doi: 10.1001/jamahealthforum.2021.4495.
7. Delanaye P et al. CKD: A call for an age-adapted definition. J Am Soc Nephrol. 2019;30(10):1785-1805. doi: 10.1681/ASN.2019030238.
8. Liu Pet al. Accounting for age in the definition of chronic kidney disease. JAMA Intern Med. 2021;181(10):1359-66. doi: 10.1001/jamainternmed.2021.4813.
I selected these topics as they were among the most discussed by my colleagues in geriatric medicine and inquired about by my primary care patients in geriatric medicine clinic. I hope that these updates provide primary care clinicians who care for older adults with more context and background information regarding new Alzheimer’s disease therapy to better answer patient inquiries, and to feel empowered to deprescribe aspirin and reframe the diagnostic criteria of chronic kidney disease (CKD).
Aspirin for primary prevention
It was welcome news in the geriatrics community when the United States Preventive Services Task Force updated their guidelines in April 2022 to recommend against the initiation of aspirin for primary prevention in adults aged 60 or older. This recommendation was based on studies that found that net benefits of CVD prevention in older adults are outweighed by risk of bleeding.1
The risk of bleeding increases with age and can occur in individuals without common risk factors for bleeding, such as prior gastrointestinal bleeding, peptic ulcer disease, concurrent NSAID use, or corticosteroid use.
While it may be easier to not initiate aspirin for primary prevention, deprescribing aspirin for patients who have been on aspirin long term for primary prevention presents more of a challenge. Modeling data from the USPTSF suggest stopping aspirin at age 75 for those taking aspirin for primary prevention.2
Behavioral change, particularly for patients who have been on aspirin for decades, can be difficult. A 2021 study by Green et al. found that language that resonates the most with older adults when deprescribing emphasized the side effects rather than statements such as “this will not help you” or “do not need anymore.”3
Aducanumab for mild cognitive impairment and mild Alzheimer’s dementia
One of the most discussed topics this past year is the Food and Drug Administration approval of aducanumab (brand name Aduhelm) in June 2021. Aducanumab is the first approved disease-modifying therapy for Alzheimer’s disease and the first drug approved for the treatment of Alzheimer’s disease since 2003. Aducanumab is an antiamyloid monoclonal antibody that was developed to reduce amyloid plaque in the brain, one of the features of Alzheimer’s disease pathology.
Uptake of aducanumab by dementia providers has been limited for several reasons. Firstly, the clinical significance of the drug remains in question. ENGAGE and EMERGE were the two main randomized clinical trials that studied the effect of aducanumab on amyloid burden and clinical stages of dementia over 18 months. While both studies demonstrated that aducanumab reduced amyloid burden based on neuroimaging and in cerebrospinal fluid, the ENGAGE trial found no difference in the stage of dementia. The EMERGE trial did note a small, statistically significant difference in stage of dementia, however the participants of the EMERGE trial had a faster rate of progression of dementia than the placebo participants in the ENGAGE trial, which could have contributed to the difference detected.4
Additionally, exclusion criteria for both trials call into question the generalizability of this study. Participants over age 85, with CKD, prior stroke, or transient ischemic attacks, or on anticoagulation were excluded. One of the drivers for the exclusion criteria is the increased risk of macro and microhemorrhages.
Thirty-five percent of research participants were incidentally noted to have brain edema, an abnormality called amyloid-related imaging abnormality or ARIA-E, that necessitated serial monitoring with brain MRIs. It is also important to highlight that inclusion of African American, Hispanic, and Latinx participants in these studies was less than 5%, despite a higher incidence of Alzheimer’s disease in these populations.5
Lastly, economic implications for the U.S. health care system with increased uptake of aducanumab could be enormous. Originally quoted at $56,000 yearly, Biogen, the maker of aducanumab, recently reduced annual costs to $28,200 per patient.
In April 2022, CMS released a statement that antiamyloid monoclonal antibodies and related services, including PET scans, would be covered under Medicare for those with mild cognitive impairment and mild Alzheimer’s dementia with confirmed presence of amyloid. A study by Mafi et al. estimated that aducanumab could cost Medicare between $7 billion and $37.4 billion annually based on lower and upper bound estimates of eligible Medicare beneficiaries.6
Overdiagnosis of CKD in older adults
The current diagnostic criteria of CKD, which is based on an estimated glomerular filtration rate (eGFR) of less than 60, has been up for debate, as glomerular filtration rate (GFR) physiologically decreases with age. Fixed thresholds can lead to underdiagnosis of CKD in younger adults and overdiagnosis of CKD in older adults. Age-adapted thresholds for the diagnosis of CKD have been proposed, with the suggestion of an eGFR threshold of 45mL/min/1.73 m2 for adults aged 65 and older.7
The clinical implication of using an age-adapted eGFR threshold definition was investigated in a 2021 cohort study by Liu et al.8 In this study, outcomes of adults diagnosed with CKD using a fixed threshold versus age-adapted threshold were compared with a healthy cohort.
A fixed threshold led to a 60% higher incidence of CKD diagnosis. However, incidence of renal failure and all-cause mortality in older adults with an eGFR between 45-59 /min/1.73 m2 with normal or mild albuminuria was of similar magnitude to the healthy cohort at 5 years of follow-up.
These findings support the use of age-adapted thresholds for the diagnosis of CKD in older adults, as an earlier diagnosis of mild CKD does not equate to clinical benefits, but could lead to harms of unnecessary interventions and patient anxiety.
Dr. Mengru “Ruru” Wang is a geriatrician and internist at the University of Washington, Seattle. She practices full-spectrum medicine, seeing patients in primary care, nursing homes, and acute care. Dr. Wang has no disclosures related to this piece.
References
1. Selak Vet al. Predicting bleeding risk to guide aspirin use for the primary prevention of cardiovascular disease: A cohort study. Ann Intern Med. 2019;170(6):357-68. doi: 10.7326/M18-2808.
2. US Preventive Services Task Force. Aspirin Use to Prevent Cardiovascular Disease: US Preventive Services Task Force Recommendation Statement. JAMA. 2022;327(16):1577-84. doi: 10.1001/jama.2022.4983.
3. Green AR et al. Assessment of patient-preferred language to achieve goal-aligned deprescribing in older adults. JAMA Netw Open. 2021;4(4):e212633. doi: 10.1001/jamanetworkopen.2021.2633.
4. Oh ES. Use of anti-amyloid therapy for Alzheimer’s disease in clinical practice. An update on Alzheimer’s disease diagnosis and therapeutics. Presentation at American Geriatrics Society Meeting, 2022. Orlando.
5. Amjad H. Issues of Access and Marginalization. An update on Alzheimer’s disease diagnosis and therapeutics. Presentation at: American Geriatrics Society Meeting, 2022. Orlando.
6. Mafi JN et al. Estimated annual spending on aducanumab in the U.S. Medicare program. JAMA Health Forum. 2022;3(1):e214495. doi: 10.1001/jamahealthforum.2021.4495.
7. Delanaye P et al. CKD: A call for an age-adapted definition. J Am Soc Nephrol. 2019;30(10):1785-1805. doi: 10.1681/ASN.2019030238.
8. Liu Pet al. Accounting for age in the definition of chronic kidney disease. JAMA Intern Med. 2021;181(10):1359-66. doi: 10.1001/jamainternmed.2021.4813.
Remnant cholesterol improves CV risk prediction
, a new study suggests.
The study, which followed almost 42,000 Danish individuals without a history of ischemic cardiovascular disease, diabetes, or statin use for more than 10 years, found that elevated remnant cholesterol appropriately reclassified up to 40% of those who later experienced myocardial infarction and ischemic heart disease.
“The clinical implications of our study include that doctors and patients should be aware of remnant cholesterol levels to prevent future risk of MI and ischemic heart disease,” the authors conclude.
They suggest that the development of a cardiovascular risk algorithm, including remnant cholesterol together with LDL cholesterol, would help to better identify high-risk individuals who could be candidates for statins in a primary prevention setting.
They note that physicians are encouraged to evaluate non-HDL cholesterol and/or apolipoprotein B rather than LDL cholesterol and certainly not yet remnant cholesterol, possibly because of the limited availability of remnant cholesterol values in some parts of the world.
However, they point out that remnant cholesterol can be calculated with a standard lipid profile without additional cost, which is currently already the standard procedure in the greater Copenhagen area.
“This means that the use of remnant cholesterol is easy to introduce into daily clinical practice,” they say.
The study was published online in the Journal of the American College of Cardiology.
The authors, Takahito Doi, MD, Anne Langsted, MD, and Børge Nordestgaard, from Copenhagen University Hospital, Denmark, explain that remnant cholesterol is total cholesterol minus LDL-cholesterol minus HDL-cholesterol and includes the cholesterol content of the triglyceride-rich very-low-density lipoproteins, intermediate-density lipoproteins, and chylomicron remnants in the nonfasting state.
“When these particles enter the arterial wall, they are taken up by macrophages to produce foam cells, and therefore elevated remnant cholesterol likely enhance accumulation of cholesterol in the arterial wall, leading to progression of atherosclerosis and in consequence ischemic heart disease,” they note.
They point out that most guidelines for assessment of the 10-year risk of ischemic heart and atherosclerotic cardiovascular disease include levels of total and HDL cholesterol, but remnant cholesterol levels are not included.
They conducted the current study to investigate whether elevated remnant cholesterol would lead to appropriate reclassification of individuals who later experienced MI or ischemic heart disease.
The researchers analyzed data from the Copenhagen General Population Study, which recruited individuals from the White Danish general population from 2003-2015 and followed them until 2018. Information on lifestyle, health, and medication, including statin therapy, was obtained through a questionnaire, and participants underwent physical examinations and had nonfasting blood samples drawn for biochemical measurements.
For the current study, they included 41,928 individuals aged 40-100 years enrolled before 2009 without a history of ischemic cardiovascular disease, diabetes, and statin use at baseline. The median follow-up time was 12 years. Information on diagnoses of MI and ischemic heart disease was collected from the national Danish Causes of Death Registry and all hospital admissions and diagnoses entered in the national Danish Patient Registry.
During the first 10 years of follow-up there were 1,063 MIs and 1,460 ischemic heart disease events (death of ischemic heart disease, nonfatal MI, and coronary revascularization).
Results showed that in models based on conventional risk factors estimating risk of heart disease of above or below 5% in 10 years, adding remnant cholesterol at levels above the 95th percentile, appropriately reclassified 23% of individuals who had an MI and 21% of individuals who had an ischemic heart disease event.
Using remnant cholesterol levels above the 75th percentile appropriately reclassified 10% of those who had an MI and 8% of those who had an ischemic heart disease event. No events were reclassified incorrectly.
Using measurements of remnant cholesterol also improved reclassification of individuals with heart disease risk above or below 7.5% or 10% in 10 years.
When reclassifications were combined from below to above 5%, 7.5%, and 10% risk of events, 42% of individuals with MI and 41% with ischemic heart disease events were reclassified appropriately.
In an editorial accompanying publication of the study in JACC, Peter Wilson, MD, Emory University School of Medicine, Atlanta, and Alan Remaley, MD, National Heart, Lung, and Blood Institute, say these findings rekindle interest in atherogenic nonfasting lipid measurements and emphasize an important role for elevated nonfasting remnant cholesterol as a value-added predictor of ischemic events.
The editorialists note that both fasting and nonfasting lipid values provide useful information for atherosclerotic cardiovascular disease (ASCVD) risk estimation, and elevated nonfasting remnant cholesterol appears to help identify persons at greater risk for an initial cardiovascular ischemic event.
They add that very elevated levels (above the 75th percentile) of nonfasting remnant cholesterol deserve further evaluation as a potentially valuable “modifier of ASCVD risk,” and replication of the results could move these findings forward to potentially improve prognostication and care for patients at risk for ischemic heart disease events.
An indirect measure of triglycerides
Dr. Wilson explained that remnant cholesterol is an indirect measure of triglycerides beyond LDL levels, and it is thus including a new lipid measurement in risk prediction.
“We are completely focused on LDL cholesterol,” he said. “This opens it up a bit by adding in another measure that takes into account triglycerides as well as LDL.”
He also pointed out that use of a nonfasting sample is another advantage of measuring remnant cholesterol.
“An accurate measure of LDL needs a fasting sample, which is a nuisance, whereas remnant cholesterol can be measured in a nonfasting blood sample, so it is more convenient,” Dr. Wilson said.
While this study shows this measure is helpful for risk prediction in the primary prevention population, Dr. Wilson believes remnant cholesterol could be most useful in helping to guide further medication choice in patients who are already taking statins.
“Statins mainly target LDL, but if we can also measure nonfasting triglycerides this will be helpful. It may help us select some patients who may need a different type of drug to use in addition to statins that lowers triglycerides,” he said.
This work was supported by the Global Excellence Programme, the Research Fund for the Capital Region of Denmark, the Japanese College of Cardiology Overseas Research Fellowship, and the Scandinavia Japan Sasakawa Foundation. Mr. Nordestgaard has reported consultancies or talks sponsored by AstraZeneca, Sanofi, Regeneron, Akcea, Amgen, Amarin, Kowa, Denka, Novartis, Novo Nordisk, Esperion, and Silence Therapeutics. Dr. Doi has reported talks sponsored by MSD.
A version of this article first appeared on Medscape.com.
, a new study suggests.
The study, which followed almost 42,000 Danish individuals without a history of ischemic cardiovascular disease, diabetes, or statin use for more than 10 years, found that elevated remnant cholesterol appropriately reclassified up to 40% of those who later experienced myocardial infarction and ischemic heart disease.
“The clinical implications of our study include that doctors and patients should be aware of remnant cholesterol levels to prevent future risk of MI and ischemic heart disease,” the authors conclude.
They suggest that the development of a cardiovascular risk algorithm, including remnant cholesterol together with LDL cholesterol, would help to better identify high-risk individuals who could be candidates for statins in a primary prevention setting.
They note that physicians are encouraged to evaluate non-HDL cholesterol and/or apolipoprotein B rather than LDL cholesterol and certainly not yet remnant cholesterol, possibly because of the limited availability of remnant cholesterol values in some parts of the world.
However, they point out that remnant cholesterol can be calculated with a standard lipid profile without additional cost, which is currently already the standard procedure in the greater Copenhagen area.
“This means that the use of remnant cholesterol is easy to introduce into daily clinical practice,” they say.
The study was published online in the Journal of the American College of Cardiology.
The authors, Takahito Doi, MD, Anne Langsted, MD, and Børge Nordestgaard, from Copenhagen University Hospital, Denmark, explain that remnant cholesterol is total cholesterol minus LDL-cholesterol minus HDL-cholesterol and includes the cholesterol content of the triglyceride-rich very-low-density lipoproteins, intermediate-density lipoproteins, and chylomicron remnants in the nonfasting state.
“When these particles enter the arterial wall, they are taken up by macrophages to produce foam cells, and therefore elevated remnant cholesterol likely enhance accumulation of cholesterol in the arterial wall, leading to progression of atherosclerosis and in consequence ischemic heart disease,” they note.
They point out that most guidelines for assessment of the 10-year risk of ischemic heart and atherosclerotic cardiovascular disease include levels of total and HDL cholesterol, but remnant cholesterol levels are not included.
They conducted the current study to investigate whether elevated remnant cholesterol would lead to appropriate reclassification of individuals who later experienced MI or ischemic heart disease.
The researchers analyzed data from the Copenhagen General Population Study, which recruited individuals from the White Danish general population from 2003-2015 and followed them until 2018. Information on lifestyle, health, and medication, including statin therapy, was obtained through a questionnaire, and participants underwent physical examinations and had nonfasting blood samples drawn for biochemical measurements.
For the current study, they included 41,928 individuals aged 40-100 years enrolled before 2009 without a history of ischemic cardiovascular disease, diabetes, and statin use at baseline. The median follow-up time was 12 years. Information on diagnoses of MI and ischemic heart disease was collected from the national Danish Causes of Death Registry and all hospital admissions and diagnoses entered in the national Danish Patient Registry.
During the first 10 years of follow-up there were 1,063 MIs and 1,460 ischemic heart disease events (death of ischemic heart disease, nonfatal MI, and coronary revascularization).
Results showed that in models based on conventional risk factors estimating risk of heart disease of above or below 5% in 10 years, adding remnant cholesterol at levels above the 95th percentile, appropriately reclassified 23% of individuals who had an MI and 21% of individuals who had an ischemic heart disease event.
Using remnant cholesterol levels above the 75th percentile appropriately reclassified 10% of those who had an MI and 8% of those who had an ischemic heart disease event. No events were reclassified incorrectly.
Using measurements of remnant cholesterol also improved reclassification of individuals with heart disease risk above or below 7.5% or 10% in 10 years.
When reclassifications were combined from below to above 5%, 7.5%, and 10% risk of events, 42% of individuals with MI and 41% with ischemic heart disease events were reclassified appropriately.
In an editorial accompanying publication of the study in JACC, Peter Wilson, MD, Emory University School of Medicine, Atlanta, and Alan Remaley, MD, National Heart, Lung, and Blood Institute, say these findings rekindle interest in atherogenic nonfasting lipid measurements and emphasize an important role for elevated nonfasting remnant cholesterol as a value-added predictor of ischemic events.
The editorialists note that both fasting and nonfasting lipid values provide useful information for atherosclerotic cardiovascular disease (ASCVD) risk estimation, and elevated nonfasting remnant cholesterol appears to help identify persons at greater risk for an initial cardiovascular ischemic event.
They add that very elevated levels (above the 75th percentile) of nonfasting remnant cholesterol deserve further evaluation as a potentially valuable “modifier of ASCVD risk,” and replication of the results could move these findings forward to potentially improve prognostication and care for patients at risk for ischemic heart disease events.
An indirect measure of triglycerides
Dr. Wilson explained that remnant cholesterol is an indirect measure of triglycerides beyond LDL levels, and it is thus including a new lipid measurement in risk prediction.
“We are completely focused on LDL cholesterol,” he said. “This opens it up a bit by adding in another measure that takes into account triglycerides as well as LDL.”
He also pointed out that use of a nonfasting sample is another advantage of measuring remnant cholesterol.
“An accurate measure of LDL needs a fasting sample, which is a nuisance, whereas remnant cholesterol can be measured in a nonfasting blood sample, so it is more convenient,” Dr. Wilson said.
While this study shows this measure is helpful for risk prediction in the primary prevention population, Dr. Wilson believes remnant cholesterol could be most useful in helping to guide further medication choice in patients who are already taking statins.
“Statins mainly target LDL, but if we can also measure nonfasting triglycerides this will be helpful. It may help us select some patients who may need a different type of drug to use in addition to statins that lowers triglycerides,” he said.
This work was supported by the Global Excellence Programme, the Research Fund for the Capital Region of Denmark, the Japanese College of Cardiology Overseas Research Fellowship, and the Scandinavia Japan Sasakawa Foundation. Mr. Nordestgaard has reported consultancies or talks sponsored by AstraZeneca, Sanofi, Regeneron, Akcea, Amgen, Amarin, Kowa, Denka, Novartis, Novo Nordisk, Esperion, and Silence Therapeutics. Dr. Doi has reported talks sponsored by MSD.
A version of this article first appeared on Medscape.com.
, a new study suggests.
The study, which followed almost 42,000 Danish individuals without a history of ischemic cardiovascular disease, diabetes, or statin use for more than 10 years, found that elevated remnant cholesterol appropriately reclassified up to 40% of those who later experienced myocardial infarction and ischemic heart disease.
“The clinical implications of our study include that doctors and patients should be aware of remnant cholesterol levels to prevent future risk of MI and ischemic heart disease,” the authors conclude.
They suggest that the development of a cardiovascular risk algorithm, including remnant cholesterol together with LDL cholesterol, would help to better identify high-risk individuals who could be candidates for statins in a primary prevention setting.
They note that physicians are encouraged to evaluate non-HDL cholesterol and/or apolipoprotein B rather than LDL cholesterol and certainly not yet remnant cholesterol, possibly because of the limited availability of remnant cholesterol values in some parts of the world.
However, they point out that remnant cholesterol can be calculated with a standard lipid profile without additional cost, which is currently already the standard procedure in the greater Copenhagen area.
“This means that the use of remnant cholesterol is easy to introduce into daily clinical practice,” they say.
The study was published online in the Journal of the American College of Cardiology.
The authors, Takahito Doi, MD, Anne Langsted, MD, and Børge Nordestgaard, from Copenhagen University Hospital, Denmark, explain that remnant cholesterol is total cholesterol minus LDL-cholesterol minus HDL-cholesterol and includes the cholesterol content of the triglyceride-rich very-low-density lipoproteins, intermediate-density lipoproteins, and chylomicron remnants in the nonfasting state.
“When these particles enter the arterial wall, they are taken up by macrophages to produce foam cells, and therefore elevated remnant cholesterol likely enhance accumulation of cholesterol in the arterial wall, leading to progression of atherosclerosis and in consequence ischemic heart disease,” they note.
They point out that most guidelines for assessment of the 10-year risk of ischemic heart and atherosclerotic cardiovascular disease include levels of total and HDL cholesterol, but remnant cholesterol levels are not included.
They conducted the current study to investigate whether elevated remnant cholesterol would lead to appropriate reclassification of individuals who later experienced MI or ischemic heart disease.
The researchers analyzed data from the Copenhagen General Population Study, which recruited individuals from the White Danish general population from 2003-2015 and followed them until 2018. Information on lifestyle, health, and medication, including statin therapy, was obtained through a questionnaire, and participants underwent physical examinations and had nonfasting blood samples drawn for biochemical measurements.
For the current study, they included 41,928 individuals aged 40-100 years enrolled before 2009 without a history of ischemic cardiovascular disease, diabetes, and statin use at baseline. The median follow-up time was 12 years. Information on diagnoses of MI and ischemic heart disease was collected from the national Danish Causes of Death Registry and all hospital admissions and diagnoses entered in the national Danish Patient Registry.
During the first 10 years of follow-up there were 1,063 MIs and 1,460 ischemic heart disease events (death of ischemic heart disease, nonfatal MI, and coronary revascularization).
Results showed that in models based on conventional risk factors estimating risk of heart disease of above or below 5% in 10 years, adding remnant cholesterol at levels above the 95th percentile, appropriately reclassified 23% of individuals who had an MI and 21% of individuals who had an ischemic heart disease event.
Using remnant cholesterol levels above the 75th percentile appropriately reclassified 10% of those who had an MI and 8% of those who had an ischemic heart disease event. No events were reclassified incorrectly.
Using measurements of remnant cholesterol also improved reclassification of individuals with heart disease risk above or below 7.5% or 10% in 10 years.
When reclassifications were combined from below to above 5%, 7.5%, and 10% risk of events, 42% of individuals with MI and 41% with ischemic heart disease events were reclassified appropriately.
In an editorial accompanying publication of the study in JACC, Peter Wilson, MD, Emory University School of Medicine, Atlanta, and Alan Remaley, MD, National Heart, Lung, and Blood Institute, say these findings rekindle interest in atherogenic nonfasting lipid measurements and emphasize an important role for elevated nonfasting remnant cholesterol as a value-added predictor of ischemic events.
The editorialists note that both fasting and nonfasting lipid values provide useful information for atherosclerotic cardiovascular disease (ASCVD) risk estimation, and elevated nonfasting remnant cholesterol appears to help identify persons at greater risk for an initial cardiovascular ischemic event.
They add that very elevated levels (above the 75th percentile) of nonfasting remnant cholesterol deserve further evaluation as a potentially valuable “modifier of ASCVD risk,” and replication of the results could move these findings forward to potentially improve prognostication and care for patients at risk for ischemic heart disease events.
An indirect measure of triglycerides
Dr. Wilson explained that remnant cholesterol is an indirect measure of triglycerides beyond LDL levels, and it is thus including a new lipid measurement in risk prediction.
“We are completely focused on LDL cholesterol,” he said. “This opens it up a bit by adding in another measure that takes into account triglycerides as well as LDL.”
He also pointed out that use of a nonfasting sample is another advantage of measuring remnant cholesterol.
“An accurate measure of LDL needs a fasting sample, which is a nuisance, whereas remnant cholesterol can be measured in a nonfasting blood sample, so it is more convenient,” Dr. Wilson said.
While this study shows this measure is helpful for risk prediction in the primary prevention population, Dr. Wilson believes remnant cholesterol could be most useful in helping to guide further medication choice in patients who are already taking statins.
“Statins mainly target LDL, but if we can also measure nonfasting triglycerides this will be helpful. It may help us select some patients who may need a different type of drug to use in addition to statins that lowers triglycerides,” he said.
This work was supported by the Global Excellence Programme, the Research Fund for the Capital Region of Denmark, the Japanese College of Cardiology Overseas Research Fellowship, and the Scandinavia Japan Sasakawa Foundation. Mr. Nordestgaard has reported consultancies or talks sponsored by AstraZeneca, Sanofi, Regeneron, Akcea, Amgen, Amarin, Kowa, Denka, Novartis, Novo Nordisk, Esperion, and Silence Therapeutics. Dr. Doi has reported talks sponsored by MSD.
A version of this article first appeared on Medscape.com.
Meta-analysis points to safety of acetylcholine coronary testing
Provocation testing with intracoronary acetylcholine is safe, particularly among Western patients, suggests a large systematic review that underscores the importance of functional coronary angiography to diagnose epicardial or microvascular spasm.
The results, derived from more than 12,000 patients in 16 studies, showed a 0.5% risk of major complications, defined as death, ventricular tachycardia/ventricular fibrillation, myocardial infarction, and shock requiring resuscitation.
Ventricular tachycardia/fibrillation were the most common events and mainly reported from two Japanese studies. There were no deaths.
Exploratory subgroup analyses revealed significantly fewer major complications in Western populations (0.0%; P for heterogeneity = .938), compared with Asian populations (2.3%; P for heterogeneity < .001).
The pooled positive vasospasm rate was also lower in Western versus Asian studies (37.9% vs. 50.7%; P for between-group heterogeneity = .010), as reported by the Microvascular Network in the Journal of the American College of Cardiology.
“If you look at the data between Asian studies versus others, mainly European or U.S. studies, primarily in Caucasian populations, it’s like zero percent history of major complications. So, it sounds extremely safe to do this testing in Caucasian populations,” Yuhei Kobayashi, MD, NewYork-Presbyterian Brooklyn Methodist Hospital, Weill Cornell Medicine, said.
Safety will need to be assessed in African Americans and other racial/ethnic groups, but “it makes us think we should end up testing more in the United States,” he told this news organization.
Intracoronary acetylcholine testing is daily practice in Japan but is limited in the United States and Europe to a few specialized centers due to safety concerns. Three deaths were reported in 1980 with intravenous ergonovine testing, whereas the safety of acetylcholine protocols has been studied largely in single-center retrospective studies, typically in Asian populations.
Growing recognition of myocardial infarction with nonobstructive coronary arteries (MINOCA) and ischemia with no obstructive coronary arteries (INOCA), however, is changing the landscape. In recent U.S. and European guidelines, intracoronary acetylcholine testing is indicated as a class 2a recommendation in MINOCA/INOCA.
“More and more institutions in Europe and the United States are starting to do acetylcholine testing, because now we know that chest pain isn’t necessarily coming from the blocked arteries,” Dr. Kobayashi said. “There are functional abnormalities, including coronary spasm, and if we diagnose it, we have appropriate medical regimens for this kind of disease.”
First safety meta-analysis
The present review and meta-analysis included 12,585 participants in 16 studies through November 2021. Of these, 63% were conducted in Western countries, and most were prospective studies published over the past decade in patients with MINOCA or INOCA.
Ten studies used the contemporary diagnostic criteria for epicardial spasm of at least 90% reduction in coronary diameter. Acetylcholine was administered into the left coronary artery at up to 100 mcg and 200 mcg in seven and six studies, respectively, and was used in the other three studies to assess endothelial function with a slower infusion of up to 36.4 mcg.
Major complications were significantly higher in studies following the contemporary diagnostic cutoff than in those using a lower cutoff of at least 75% diameter reduction (1.0% vs. 0.0%; P for between-group heterogeneity < .001).
The incidence of major complications was 0.2% with the slower infusion of up to 36.4 mcg, 0.8% with a maximum dose of 100 mcg, and 0.3% with a maximum dose of 200 mcg. The positive vasospasm rate was similar with the latter two protocols, at 46.3% and 41.4%, respectively.
Minor complications occurred in 3.3% of patients but were not detailed. They can include paroxysmal atrial fibrillation, ventricular ectopic beats, transient hypotension, and bradycardia requiring intervention.
As with major complications, minor complications were lower in studies using noncontemporary versus contemporary diagnostic cutoffs for epicardial spasm (1.8% vs. 4.7%) and in Western versus Asian populations (2.6% vs. 9.4%). Minor complications were similar between protocols with maximum doses of 100 mcg and 200 mcg (3.6% vs. 3.8%).
Dr. Kobayashi suggested that several factors may explain the racial differences, including previously reported smooth muscle hyperresponsiveness to provocation stimuli in Japanese patients and the inclusion of a wide range of patients in Japanese studies, such as those with obstructive coronary disease.
Japanese studies also used sequential acetylcholine injection into both the right and left coronaries, a faster injection speed of 20 seconds, and upfront placement of a temporary pacing catheter in case of acetylcholine-induced bradycardia, particularly with right coronary injection.
Although the protocol is largely settled in Japan, he said, provocation protocols need to be standardized because “depending on the country and depending on the institution, people are doing totally different things.”
A big step forward
Commenting on the study, C. Noel Bairey Merz, MD, from Cedars Sinai, Los Angeles, said it has “widespread relevance” because half of all coronary angiograms done invasively in the United States for suspected ischemia find no obstructive coronary disease. Left untreated, however, MINOCA has a 2.5% annual event rate, and a quarter of that is death.
“This is a big step forward with likely equal opportunity to improve women and men’s ischemic heart disease,” she said.
On the other hand, all studies were conducted at centers of excellence, so safety will need to be carefully watched as testing rolls out to more community care, Dr. Merz said. “And it always needs to be underscored that this is done by an interventional cardiologist because they’re familiar with wires that can dissect arteries, and they’re familiar with minor complications that could turn into major, if someone didn’t act appropriately.”
Dr. Merz also called for unifying protocols and the need to raise awareness within the general cardiology community to ask interventionalists for acetylcholine spasm testing. Randomized controlled data from within the WISE study and the CorMica study showed that diagnostic certainty leads to greater therapeutic certainty. “You do a much better job about who and how to treat,” she said.
There are also three ongoing randomized controlled trials – WARRIOR, MINOCA-BAT, and iCorMica – in the INOCA and MINOCA populations testing different treatment strategies for hard clinical outcomes like death and myocardial infarction.
“So in addition to this publication being guideline-forming for diagnosis, we anticipate in the next several years to have clinical trial evidence about therapeutics, again, for formulation of class 1 guidelines,” Dr. Merz said.
John Beltrame, BMBS, PhD, University of Adelaide, Australia, said the meta-analysis shows that intracoronary acetylcholine spasm testing is safe and should prompt greater adoption of invasive functional angiography.
Interventionalists are quite happy to do fractional flow reserve using intravenous adenosine to assess coronary microvascular dysfunction, he said, and “what we think is that functional angiography should test both – both the spasm as well as the microvasculature – and that will give us a clear direction because the treatments are slightly different when you’re treating the large arteries as compared to the microscopic arteries. It’s an important thing.”
Dr. Beltrame and colleagues further detail the benefits of comprehensive invasive functional angiography over structural angiography in a related editorial.
He also noted that the Coronary Vasomotion Disorders International Study Group published international diagnostic criteria for microvascular angina and that several protocols for acetylcholine spasm testing are in the works, including one from Australia. Australian investigators are also organizing an accreditation program for those performing the test.
“The protocol itself is relatively straightforward, but it’s not merely picking up a manual and following the instructions,” Dr. Beltrame said. “Just the same as when you train someone in angioplasty, you don’t just go out and do it. You need to develop some experience in it and so should be proctored.”
Dr. Kobayashi reported consulting agreements with Abbott Vascular. Coauthor disclosures are listed in the paper. Dr. Beltrame and colleagues have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Provocation testing with intracoronary acetylcholine is safe, particularly among Western patients, suggests a large systematic review that underscores the importance of functional coronary angiography to diagnose epicardial or microvascular spasm.
The results, derived from more than 12,000 patients in 16 studies, showed a 0.5% risk of major complications, defined as death, ventricular tachycardia/ventricular fibrillation, myocardial infarction, and shock requiring resuscitation.
Ventricular tachycardia/fibrillation were the most common events and mainly reported from two Japanese studies. There were no deaths.
Exploratory subgroup analyses revealed significantly fewer major complications in Western populations (0.0%; P for heterogeneity = .938), compared with Asian populations (2.3%; P for heterogeneity < .001).
The pooled positive vasospasm rate was also lower in Western versus Asian studies (37.9% vs. 50.7%; P for between-group heterogeneity = .010), as reported by the Microvascular Network in the Journal of the American College of Cardiology.
“If you look at the data between Asian studies versus others, mainly European or U.S. studies, primarily in Caucasian populations, it’s like zero percent history of major complications. So, it sounds extremely safe to do this testing in Caucasian populations,” Yuhei Kobayashi, MD, NewYork-Presbyterian Brooklyn Methodist Hospital, Weill Cornell Medicine, said.
Safety will need to be assessed in African Americans and other racial/ethnic groups, but “it makes us think we should end up testing more in the United States,” he told this news organization.
Intracoronary acetylcholine testing is daily practice in Japan but is limited in the United States and Europe to a few specialized centers due to safety concerns. Three deaths were reported in 1980 with intravenous ergonovine testing, whereas the safety of acetylcholine protocols has been studied largely in single-center retrospective studies, typically in Asian populations.
Growing recognition of myocardial infarction with nonobstructive coronary arteries (MINOCA) and ischemia with no obstructive coronary arteries (INOCA), however, is changing the landscape. In recent U.S. and European guidelines, intracoronary acetylcholine testing is indicated as a class 2a recommendation in MINOCA/INOCA.
“More and more institutions in Europe and the United States are starting to do acetylcholine testing, because now we know that chest pain isn’t necessarily coming from the blocked arteries,” Dr. Kobayashi said. “There are functional abnormalities, including coronary spasm, and if we diagnose it, we have appropriate medical regimens for this kind of disease.”
First safety meta-analysis
The present review and meta-analysis included 12,585 participants in 16 studies through November 2021. Of these, 63% were conducted in Western countries, and most were prospective studies published over the past decade in patients with MINOCA or INOCA.
Ten studies used the contemporary diagnostic criteria for epicardial spasm of at least 90% reduction in coronary diameter. Acetylcholine was administered into the left coronary artery at up to 100 mcg and 200 mcg in seven and six studies, respectively, and was used in the other three studies to assess endothelial function with a slower infusion of up to 36.4 mcg.
Major complications were significantly higher in studies following the contemporary diagnostic cutoff than in those using a lower cutoff of at least 75% diameter reduction (1.0% vs. 0.0%; P for between-group heterogeneity < .001).
The incidence of major complications was 0.2% with the slower infusion of up to 36.4 mcg, 0.8% with a maximum dose of 100 mcg, and 0.3% with a maximum dose of 200 mcg. The positive vasospasm rate was similar with the latter two protocols, at 46.3% and 41.4%, respectively.
Minor complications occurred in 3.3% of patients but were not detailed. They can include paroxysmal atrial fibrillation, ventricular ectopic beats, transient hypotension, and bradycardia requiring intervention.
As with major complications, minor complications were lower in studies using noncontemporary versus contemporary diagnostic cutoffs for epicardial spasm (1.8% vs. 4.7%) and in Western versus Asian populations (2.6% vs. 9.4%). Minor complications were similar between protocols with maximum doses of 100 mcg and 200 mcg (3.6% vs. 3.8%).
Dr. Kobayashi suggested that several factors may explain the racial differences, including previously reported smooth muscle hyperresponsiveness to provocation stimuli in Japanese patients and the inclusion of a wide range of patients in Japanese studies, such as those with obstructive coronary disease.
Japanese studies also used sequential acetylcholine injection into both the right and left coronaries, a faster injection speed of 20 seconds, and upfront placement of a temporary pacing catheter in case of acetylcholine-induced bradycardia, particularly with right coronary injection.
Although the protocol is largely settled in Japan, he said, provocation protocols need to be standardized because “depending on the country and depending on the institution, people are doing totally different things.”
A big step forward
Commenting on the study, C. Noel Bairey Merz, MD, from Cedars Sinai, Los Angeles, said it has “widespread relevance” because half of all coronary angiograms done invasively in the United States for suspected ischemia find no obstructive coronary disease. Left untreated, however, MINOCA has a 2.5% annual event rate, and a quarter of that is death.
“This is a big step forward with likely equal opportunity to improve women and men’s ischemic heart disease,” she said.
On the other hand, all studies were conducted at centers of excellence, so safety will need to be carefully watched as testing rolls out to more community care, Dr. Merz said. “And it always needs to be underscored that this is done by an interventional cardiologist because they’re familiar with wires that can dissect arteries, and they’re familiar with minor complications that could turn into major, if someone didn’t act appropriately.”
Dr. Merz also called for unifying protocols and the need to raise awareness within the general cardiology community to ask interventionalists for acetylcholine spasm testing. Randomized controlled data from within the WISE study and the CorMica study showed that diagnostic certainty leads to greater therapeutic certainty. “You do a much better job about who and how to treat,” she said.
There are also three ongoing randomized controlled trials – WARRIOR, MINOCA-BAT, and iCorMica – in the INOCA and MINOCA populations testing different treatment strategies for hard clinical outcomes like death and myocardial infarction.
“So in addition to this publication being guideline-forming for diagnosis, we anticipate in the next several years to have clinical trial evidence about therapeutics, again, for formulation of class 1 guidelines,” Dr. Merz said.
John Beltrame, BMBS, PhD, University of Adelaide, Australia, said the meta-analysis shows that intracoronary acetylcholine spasm testing is safe and should prompt greater adoption of invasive functional angiography.
Interventionalists are quite happy to do fractional flow reserve using intravenous adenosine to assess coronary microvascular dysfunction, he said, and “what we think is that functional angiography should test both – both the spasm as well as the microvasculature – and that will give us a clear direction because the treatments are slightly different when you’re treating the large arteries as compared to the microscopic arteries. It’s an important thing.”
Dr. Beltrame and colleagues further detail the benefits of comprehensive invasive functional angiography over structural angiography in a related editorial.
He also noted that the Coronary Vasomotion Disorders International Study Group published international diagnostic criteria for microvascular angina and that several protocols for acetylcholine spasm testing are in the works, including one from Australia. Australian investigators are also organizing an accreditation program for those performing the test.
“The protocol itself is relatively straightforward, but it’s not merely picking up a manual and following the instructions,” Dr. Beltrame said. “Just the same as when you train someone in angioplasty, you don’t just go out and do it. You need to develop some experience in it and so should be proctored.”
Dr. Kobayashi reported consulting agreements with Abbott Vascular. Coauthor disclosures are listed in the paper. Dr. Beltrame and colleagues have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Provocation testing with intracoronary acetylcholine is safe, particularly among Western patients, suggests a large systematic review that underscores the importance of functional coronary angiography to diagnose epicardial or microvascular spasm.
The results, derived from more than 12,000 patients in 16 studies, showed a 0.5% risk of major complications, defined as death, ventricular tachycardia/ventricular fibrillation, myocardial infarction, and shock requiring resuscitation.
Ventricular tachycardia/fibrillation were the most common events and mainly reported from two Japanese studies. There were no deaths.
Exploratory subgroup analyses revealed significantly fewer major complications in Western populations (0.0%; P for heterogeneity = .938), compared with Asian populations (2.3%; P for heterogeneity < .001).
The pooled positive vasospasm rate was also lower in Western versus Asian studies (37.9% vs. 50.7%; P for between-group heterogeneity = .010), as reported by the Microvascular Network in the Journal of the American College of Cardiology.
“If you look at the data between Asian studies versus others, mainly European or U.S. studies, primarily in Caucasian populations, it’s like zero percent history of major complications. So, it sounds extremely safe to do this testing in Caucasian populations,” Yuhei Kobayashi, MD, NewYork-Presbyterian Brooklyn Methodist Hospital, Weill Cornell Medicine, said.
Safety will need to be assessed in African Americans and other racial/ethnic groups, but “it makes us think we should end up testing more in the United States,” he told this news organization.
Intracoronary acetylcholine testing is daily practice in Japan but is limited in the United States and Europe to a few specialized centers due to safety concerns. Three deaths were reported in 1980 with intravenous ergonovine testing, whereas the safety of acetylcholine protocols has been studied largely in single-center retrospective studies, typically in Asian populations.
Growing recognition of myocardial infarction with nonobstructive coronary arteries (MINOCA) and ischemia with no obstructive coronary arteries (INOCA), however, is changing the landscape. In recent U.S. and European guidelines, intracoronary acetylcholine testing is indicated as a class 2a recommendation in MINOCA/INOCA.
“More and more institutions in Europe and the United States are starting to do acetylcholine testing, because now we know that chest pain isn’t necessarily coming from the blocked arteries,” Dr. Kobayashi said. “There are functional abnormalities, including coronary spasm, and if we diagnose it, we have appropriate medical regimens for this kind of disease.”
First safety meta-analysis
The present review and meta-analysis included 12,585 participants in 16 studies through November 2021. Of these, 63% were conducted in Western countries, and most were prospective studies published over the past decade in patients with MINOCA or INOCA.
Ten studies used the contemporary diagnostic criteria for epicardial spasm of at least 90% reduction in coronary diameter. Acetylcholine was administered into the left coronary artery at up to 100 mcg and 200 mcg in seven and six studies, respectively, and was used in the other three studies to assess endothelial function with a slower infusion of up to 36.4 mcg.
Major complications were significantly higher in studies following the contemporary diagnostic cutoff than in those using a lower cutoff of at least 75% diameter reduction (1.0% vs. 0.0%; P for between-group heterogeneity < .001).
The incidence of major complications was 0.2% with the slower infusion of up to 36.4 mcg, 0.8% with a maximum dose of 100 mcg, and 0.3% with a maximum dose of 200 mcg. The positive vasospasm rate was similar with the latter two protocols, at 46.3% and 41.4%, respectively.
Minor complications occurred in 3.3% of patients but were not detailed. They can include paroxysmal atrial fibrillation, ventricular ectopic beats, transient hypotension, and bradycardia requiring intervention.
As with major complications, minor complications were lower in studies using noncontemporary versus contemporary diagnostic cutoffs for epicardial spasm (1.8% vs. 4.7%) and in Western versus Asian populations (2.6% vs. 9.4%). Minor complications were similar between protocols with maximum doses of 100 mcg and 200 mcg (3.6% vs. 3.8%).
Dr. Kobayashi suggested that several factors may explain the racial differences, including previously reported smooth muscle hyperresponsiveness to provocation stimuli in Japanese patients and the inclusion of a wide range of patients in Japanese studies, such as those with obstructive coronary disease.
Japanese studies also used sequential acetylcholine injection into both the right and left coronaries, a faster injection speed of 20 seconds, and upfront placement of a temporary pacing catheter in case of acetylcholine-induced bradycardia, particularly with right coronary injection.
Although the protocol is largely settled in Japan, he said, provocation protocols need to be standardized because “depending on the country and depending on the institution, people are doing totally different things.”
A big step forward
Commenting on the study, C. Noel Bairey Merz, MD, from Cedars Sinai, Los Angeles, said it has “widespread relevance” because half of all coronary angiograms done invasively in the United States for suspected ischemia find no obstructive coronary disease. Left untreated, however, MINOCA has a 2.5% annual event rate, and a quarter of that is death.
“This is a big step forward with likely equal opportunity to improve women and men’s ischemic heart disease,” she said.
On the other hand, all studies were conducted at centers of excellence, so safety will need to be carefully watched as testing rolls out to more community care, Dr. Merz said. “And it always needs to be underscored that this is done by an interventional cardiologist because they’re familiar with wires that can dissect arteries, and they’re familiar with minor complications that could turn into major, if someone didn’t act appropriately.”
Dr. Merz also called for unifying protocols and the need to raise awareness within the general cardiology community to ask interventionalists for acetylcholine spasm testing. Randomized controlled data from within the WISE study and the CorMica study showed that diagnostic certainty leads to greater therapeutic certainty. “You do a much better job about who and how to treat,” she said.
There are also three ongoing randomized controlled trials – WARRIOR, MINOCA-BAT, and iCorMica – in the INOCA and MINOCA populations testing different treatment strategies for hard clinical outcomes like death and myocardial infarction.
“So in addition to this publication being guideline-forming for diagnosis, we anticipate in the next several years to have clinical trial evidence about therapeutics, again, for formulation of class 1 guidelines,” Dr. Merz said.
John Beltrame, BMBS, PhD, University of Adelaide, Australia, said the meta-analysis shows that intracoronary acetylcholine spasm testing is safe and should prompt greater adoption of invasive functional angiography.
Interventionalists are quite happy to do fractional flow reserve using intravenous adenosine to assess coronary microvascular dysfunction, he said, and “what we think is that functional angiography should test both – both the spasm as well as the microvasculature – and that will give us a clear direction because the treatments are slightly different when you’re treating the large arteries as compared to the microscopic arteries. It’s an important thing.”
Dr. Beltrame and colleagues further detail the benefits of comprehensive invasive functional angiography over structural angiography in a related editorial.
He also noted that the Coronary Vasomotion Disorders International Study Group published international diagnostic criteria for microvascular angina and that several protocols for acetylcholine spasm testing are in the works, including one from Australia. Australian investigators are also organizing an accreditation program for those performing the test.
“The protocol itself is relatively straightforward, but it’s not merely picking up a manual and following the instructions,” Dr. Beltrame said. “Just the same as when you train someone in angioplasty, you don’t just go out and do it. You need to develop some experience in it and so should be proctored.”
Dr. Kobayashi reported consulting agreements with Abbott Vascular. Coauthor disclosures are listed in the paper. Dr. Beltrame and colleagues have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Heart failure: Medicare cost sharing may put quadruple therapy out of reach
Out-of-pocket (OOP) costs for Medicare enrollees receiving quadruple drug therapy for heart failure with reduced ejection fraction were “substantially higher than regimens limited to generically available medications,” according to a new analysis of prescription drug plans.
“Despite the clinical benefit of quadruple therapy” consisting of beta-blockers, angiotensin receptor-neprilysin inhibitors (ARNIs), mineralocorticoid receptor antagonists (MRAs), and sodium-glucose cotransporter-2 (SGLT2) inhibitors, “coverage was restricted primarily through cost sharing, and estimated annual OOP costs for beneficiaries were [over $2,000] per year under most plans,” wrote Kamil F. Faridi, MD, and associates. The findings were published in the Journal of the American College of Cardiology.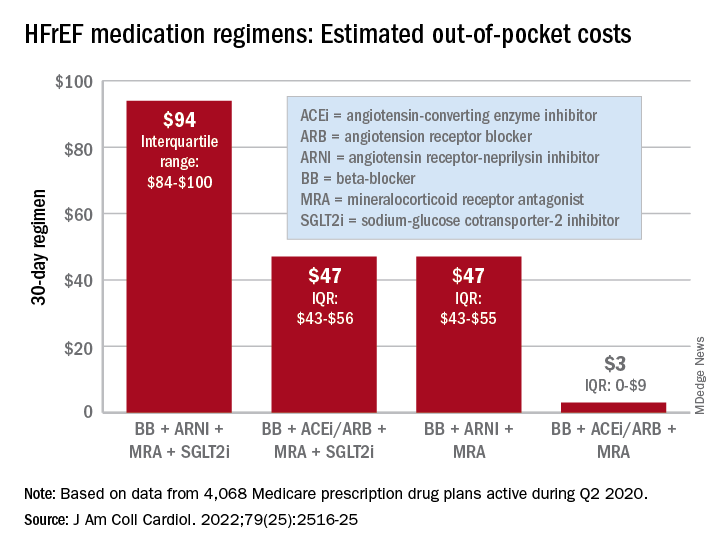
For just 1 month of quadruple drug therapy for heart failure with reduced ejection fraction (HFrEF), the estimated median OOP cost was $94 for individuals covered by a Medicare prescription drug plan during the second quarter of 2020, with the majority coming from the ARNI (median, $47) and the SGLT2 inhibitor (median, $45). Alternative HFrEF regimens were significantly less costly, ranging from $3 to $47 OOP, the investigators reported.
Almost all of the 4,068 plans participating in Medicare at that time covered quadruple therapy for HFrEF, but more than 99% restricted coverage by instituting cost sharing for medications at tier level 3 and above on the drug formularies. Such restrictions for ARNIs and SGLT2 inhibitors “might not be readily apparent to prescribing physicians,” wrote Dr. Faridi of Yale University, New Haven, Conn., and associates.
Other methods of regulating coverage were less common. Prior authorization of ARNIs was invoked by about a quarter of the plans, but none required authorization for any of the other drugs involved, and few plans used step therapy-requirements involving lower-cost alternatives, they noted.
“The use of cost sharing restricts access through high OOP costs for patients. Furthermore, these policies likely disadvantage relatively poorer patients (although the poorest Medicare patients will tend to be dual-enrolled in Medicaid and protected from cost sharing),” Jason H. Wasfy, MD, and Anna C. O’Kelly, MD, said in an accompanying editorial comment .
Since acceptable cost-effectiveness has been demonstrated for dapagliflozin, an SGLT1 inhibitor, and for the ARNIs, and because these medications have no generic equivalents, health plans should “use the discretion they have under Medicare Part D to reduce cost sharing for patients with HFrEF,” Dr. Wasfy and Dr. O’Kelly wrote, adding that the current study “demonstrates that without consensus on cost effectiveness from the societal perspective, costs can be imposed directly on patients in ways that slow uptake of cost-effective drugs.”
Data for all Medicare Advantage plans (n = 3,167) and standalone Part D plans (n = 901) came from the Medicare Prescription Drug Plan Formulary and Pricing Information Files. Annual OOP costs were estimated “using each phase of a 2020 Medicare part D standard benefit,” including deductible, standard coverage, coverage gap, and catastrophic coverage, the investigators explained.
Dr. Faridi and associates did not report any direct funding sources for their study. Dr Faridi received a grant from the National Institutes of Health outside the scope of the present work, and other investigators disclosed ties to the Food and Drug Administration, the Centers for Medicare and Medicaid Services, Johnson & Johnson, AstraZeneca, Boehringer Ingelheim, Amgen, Cytokinetics, and the Institute for Clinical and Economic Review.
Dr. Wasfy is supported by the American Heart Association and has received consulting fees from Pfizer and honoraria from the Institute for Clinical and Economic Review. Dr. O’Kelly has no relevant disclosures.
Out-of-pocket (OOP) costs for Medicare enrollees receiving quadruple drug therapy for heart failure with reduced ejection fraction were “substantially higher than regimens limited to generically available medications,” according to a new analysis of prescription drug plans.
“Despite the clinical benefit of quadruple therapy” consisting of beta-blockers, angiotensin receptor-neprilysin inhibitors (ARNIs), mineralocorticoid receptor antagonists (MRAs), and sodium-glucose cotransporter-2 (SGLT2) inhibitors, “coverage was restricted primarily through cost sharing, and estimated annual OOP costs for beneficiaries were [over $2,000] per year under most plans,” wrote Kamil F. Faridi, MD, and associates. The findings were published in the Journal of the American College of Cardiology.
For just 1 month of quadruple drug therapy for heart failure with reduced ejection fraction (HFrEF), the estimated median OOP cost was $94 for individuals covered by a Medicare prescription drug plan during the second quarter of 2020, with the majority coming from the ARNI (median, $47) and the SGLT2 inhibitor (median, $45). Alternative HFrEF regimens were significantly less costly, ranging from $3 to $47 OOP, the investigators reported.
Almost all of the 4,068 plans participating in Medicare at that time covered quadruple therapy for HFrEF, but more than 99% restricted coverage by instituting cost sharing for medications at tier level 3 and above on the drug formularies. Such restrictions for ARNIs and SGLT2 inhibitors “might not be readily apparent to prescribing physicians,” wrote Dr. Faridi of Yale University, New Haven, Conn., and associates.
Other methods of regulating coverage were less common. Prior authorization of ARNIs was invoked by about a quarter of the plans, but none required authorization for any of the other drugs involved, and few plans used step therapy-requirements involving lower-cost alternatives, they noted.
“The use of cost sharing restricts access through high OOP costs for patients. Furthermore, these policies likely disadvantage relatively poorer patients (although the poorest Medicare patients will tend to be dual-enrolled in Medicaid and protected from cost sharing),” Jason H. Wasfy, MD, and Anna C. O’Kelly, MD, said in an accompanying editorial comment .
Since acceptable cost-effectiveness has been demonstrated for dapagliflozin, an SGLT1 inhibitor, and for the ARNIs, and because these medications have no generic equivalents, health plans should “use the discretion they have under Medicare Part D to reduce cost sharing for patients with HFrEF,” Dr. Wasfy and Dr. O’Kelly wrote, adding that the current study “demonstrates that without consensus on cost effectiveness from the societal perspective, costs can be imposed directly on patients in ways that slow uptake of cost-effective drugs.”
Data for all Medicare Advantage plans (n = 3,167) and standalone Part D plans (n = 901) came from the Medicare Prescription Drug Plan Formulary and Pricing Information Files. Annual OOP costs were estimated “using each phase of a 2020 Medicare part D standard benefit,” including deductible, standard coverage, coverage gap, and catastrophic coverage, the investigators explained.
Dr. Faridi and associates did not report any direct funding sources for their study. Dr Faridi received a grant from the National Institutes of Health outside the scope of the present work, and other investigators disclosed ties to the Food and Drug Administration, the Centers for Medicare and Medicaid Services, Johnson & Johnson, AstraZeneca, Boehringer Ingelheim, Amgen, Cytokinetics, and the Institute for Clinical and Economic Review.
Dr. Wasfy is supported by the American Heart Association and has received consulting fees from Pfizer and honoraria from the Institute for Clinical and Economic Review. Dr. O’Kelly has no relevant disclosures.
Out-of-pocket (OOP) costs for Medicare enrollees receiving quadruple drug therapy for heart failure with reduced ejection fraction were “substantially higher than regimens limited to generically available medications,” according to a new analysis of prescription drug plans.
“Despite the clinical benefit of quadruple therapy” consisting of beta-blockers, angiotensin receptor-neprilysin inhibitors (ARNIs), mineralocorticoid receptor antagonists (MRAs), and sodium-glucose cotransporter-2 (SGLT2) inhibitors, “coverage was restricted primarily through cost sharing, and estimated annual OOP costs for beneficiaries were [over $2,000] per year under most plans,” wrote Kamil F. Faridi, MD, and associates. The findings were published in the Journal of the American College of Cardiology.
For just 1 month of quadruple drug therapy for heart failure with reduced ejection fraction (HFrEF), the estimated median OOP cost was $94 for individuals covered by a Medicare prescription drug plan during the second quarter of 2020, with the majority coming from the ARNI (median, $47) and the SGLT2 inhibitor (median, $45). Alternative HFrEF regimens were significantly less costly, ranging from $3 to $47 OOP, the investigators reported.
Almost all of the 4,068 plans participating in Medicare at that time covered quadruple therapy for HFrEF, but more than 99% restricted coverage by instituting cost sharing for medications at tier level 3 and above on the drug formularies. Such restrictions for ARNIs and SGLT2 inhibitors “might not be readily apparent to prescribing physicians,” wrote Dr. Faridi of Yale University, New Haven, Conn., and associates.
Other methods of regulating coverage were less common. Prior authorization of ARNIs was invoked by about a quarter of the plans, but none required authorization for any of the other drugs involved, and few plans used step therapy-requirements involving lower-cost alternatives, they noted.
“The use of cost sharing restricts access through high OOP costs for patients. Furthermore, these policies likely disadvantage relatively poorer patients (although the poorest Medicare patients will tend to be dual-enrolled in Medicaid and protected from cost sharing),” Jason H. Wasfy, MD, and Anna C. O’Kelly, MD, said in an accompanying editorial comment .
Since acceptable cost-effectiveness has been demonstrated for dapagliflozin, an SGLT1 inhibitor, and for the ARNIs, and because these medications have no generic equivalents, health plans should “use the discretion they have under Medicare Part D to reduce cost sharing for patients with HFrEF,” Dr. Wasfy and Dr. O’Kelly wrote, adding that the current study “demonstrates that without consensus on cost effectiveness from the societal perspective, costs can be imposed directly on patients in ways that slow uptake of cost-effective drugs.”
Data for all Medicare Advantage plans (n = 3,167) and standalone Part D plans (n = 901) came from the Medicare Prescription Drug Plan Formulary and Pricing Information Files. Annual OOP costs were estimated “using each phase of a 2020 Medicare part D standard benefit,” including deductible, standard coverage, coverage gap, and catastrophic coverage, the investigators explained.
Dr. Faridi and associates did not report any direct funding sources for their study. Dr Faridi received a grant from the National Institutes of Health outside the scope of the present work, and other investigators disclosed ties to the Food and Drug Administration, the Centers for Medicare and Medicaid Services, Johnson & Johnson, AstraZeneca, Boehringer Ingelheim, Amgen, Cytokinetics, and the Institute for Clinical and Economic Review.
Dr. Wasfy is supported by the American Heart Association and has received consulting fees from Pfizer and honoraria from the Institute for Clinical and Economic Review. Dr. O’Kelly has no relevant disclosures.
FROM THE JOURNAL Of the AMERICAN COLLEGE OF CARDIOLOGY
Ultra-processed: Doctors debate whether putting this label on foods is useful
The NOVA system divides foods into “fresh or minimally processed,” such as strawberries or steel-cut oats; “processed culinary ingredients,” such as olive oil; “processed foods,” such as cheeses; and “ultra-processed foods.” UPFs are defined as “industrial formulations made by deconstructing natural food into its chemical constituents, modifying them and recombining them with additives into products liable to displace all other NOVA food groups.”
According to doctors who presented during the meeting, ultra-processed foods are drawing increased attention, because researchers have been examining them in National Institutes of Health–funded studies and journalists have been writing about them.
During the debate session at the meeting, some experts said that, with obesity and poor health skyrocketing, increased awareness and labeling of UPFs can only be a good thing. In contrast others noted at the meeting that the classification system that has come to be used for identifying UPFs – the NOVA Food Classification system – is too mushy, confusing, and, ultimately unhelpful.
Carlos Monteiro, MD, PhD, professor of nutrition and public health at the University of Sao Paolo, was part of the group favoring the NOVA system’s classifying certain foods as UPFs, during the debate. He drew attention to the extent to which the world’s population is getting its calories from UPFs.
Mexico and France get about 30% of calories from these foods. In Canada, it’s 48%. And in the United States, it’s 57%, Dr. Monteiro said.
Studies have found that UPFs, many of which are designed to be exceedingly flavorful and intended to replace consumption of unprocessed whole foods, lead to more overall energy intake, more added sugar in the diet, and less fiber and protein intake, he said.
To further support his arguments, Dr. Monteiro pointed to studies suggesting that it is not just the resulting change in the nutritional intake that is unhealthy, but the UPF manufacturing process itself. When adjusting for fat, sugar, and sodium intake, for example, health outcomes associated with UPFs remain poor, he explained.
“I’m sorry,” he said in the debate. “If you don’t reduce this, you don’t reduce your obesity, your diabetes prevalence.”
A study presented by Jacqueline Vernarelli, PhD, during a different session at the meeting suggested there may be other downsides to consuming UPFs. This research, which was based on the U.S. National Youth Fitness Survey, found that poorer locomotor skills among children aged 3-5 and poorer cardiovascular fitness among those aged 12-15 were associated with getting more calories from UPFs.
Those with lower cardiovascular fitness consumed 1,234 calories a day from UPFs, and those with higher cardiovascular fitness consumed 1,007 calories a day from UPFs (P = .002), according to the new research.
“It’s notable here that, although these differences are significant, both groups are consuming a pretty high proportion of their diet from ultra-processed foods,” said Dr. Vernarelli, associate professor of public health at Sacred Heart University, Fairfield, Conn., during her presentation.
In the debate session, Arne Astrup, MD, PhD, senior project director at the Healthy Weight Center at the Novo Nordisk Foundation, Hellerup, Denmark, presented an opposing view.
He said the definition of UPFs makes it too difficult to categorize many foods, pointing to a study from this year in which about 150 nutrition experts, doctors, and dietitians classified 120 foods. Only three marketed foods and one generic food were classified the same by all the evaluators.
Referring to the study Dr. Astrup cited, Dr. Monteiro said it was a mere “exercise,” and the experts involved in it had conflicts of interest.
Dr. Astrup touted this study’s size and its appearance in the peer-reviewed journal the European Journal of Clinical Nutrition.
Defending his point of view, Dr. Astrup said, “The definition and classification is so ambiguous, and the risk of misclassification is so extremely high, I think we really miss the basic requirement of science, namely that we know what we are talking about,” he said.
If you take an unprocessed food, and insert a “little additive … suddenly it’s an ultra-processed food,” he added.
UPF definition doesn’t flag some unhealthy foods
Susan Roberts, PhD, professor of nutrition at Tufts University, Boston, was a discussant at the debate and touched on the merits of both sides. She noted that the UPF definition doesn’t flag some “clearly unhealthy foods,” such as table sugar, but does flag some healthy ones, such as plant-based burgers – to which Dr. Monteiro said that the system was not a system meant to divide foods into healthy and unhealthy groups, during the debate session.
The inclusion of both healthy and unhealthy foods in NOVA’s definition of a UPF is a serious problem, Dr. Roberts said.
“It’s almost like it’s an emotional classification designed to get at the food industry rather than focusing on health – and I think that’s asking for trouble because it’s just going to be such a mess to tell consumers, ‘Well, this ultra-processed food is healthy and this one isn’t,’ ” she said. What’s happening is the term ultra-processed is being used interchangeably with unhealthy.
The discussion that the UPF classification has generated is useful, Dr. Roberts continued. “This definition grew out of that recognition that we’re engaged in an unprecedented experiment of how unhealthy can you make the world without having a major catastrophe.”
She added that the UPF concept deserves a more formalized and rigorous evaluation.
“This is an important topic for the future of public health, and I think it needs big committees to address it seriously,” she said. “I think we should not be dealing with this individually in different labs.”
Doctor’s take on usefulness of discussing UPF concept with patients
Mark Corkins, MD, who did not participate in the debate at the meeting, said he talks to parents and children about nutrition at every office visit in which he sees a child with an unhealthy weight.
“Persistence wears down resistance,” said the chair of the American Academy of Pediatrics nutrition committee, in an interview.“A consistent message – you say the same thing and you say it multiple times.”
The idea of “ultra-processed foods” plays a role in those conversations, but largely in the background. It’s a topic that’s important for pediatric health, Dr. Corkins said – but he doesn’t make it the focal point.
“It’s not a direct attack on ultra-processed foods that usually I take as my direction,” said Dr. Corkins, who is also chief of pediatric gastroenterology at Le Bonheur Children’s Hospital in Memphis, Tenn.. “What I try to focus on, and what I think the American Academy of Pediatrics would focus on, is that we need to focus on making the diet better.”
He added, “Parents are aware – they don’t call it ultra-processed food, they call it junk food.”
Dr. Corkins continued that he is reluctant to directly challenge parents on feeding their children unhealthy foods – ultra-processed or not – lest he shame them and harm the relationship.
“Guilt as a motivator isn’t really highly successful,” he said, in an interview.
Dr. Astrup reported advisory committee or board member involvement with Green Leaf Medical and RNPC, France. Dr. Roberts reported advisory committee or board member involvement with Danone, and an ownership interest in Instinct Health Science. Dr. Monteiro and Dr. Corkins reported no relevant disclosures.
The NOVA system divides foods into “fresh or minimally processed,” such as strawberries or steel-cut oats; “processed culinary ingredients,” such as olive oil; “processed foods,” such as cheeses; and “ultra-processed foods.” UPFs are defined as “industrial formulations made by deconstructing natural food into its chemical constituents, modifying them and recombining them with additives into products liable to displace all other NOVA food groups.”
According to doctors who presented during the meeting, ultra-processed foods are drawing increased attention, because researchers have been examining them in National Institutes of Health–funded studies and journalists have been writing about them.
During the debate session at the meeting, some experts said that, with obesity and poor health skyrocketing, increased awareness and labeling of UPFs can only be a good thing. In contrast others noted at the meeting that the classification system that has come to be used for identifying UPFs – the NOVA Food Classification system – is too mushy, confusing, and, ultimately unhelpful.
Carlos Monteiro, MD, PhD, professor of nutrition and public health at the University of Sao Paolo, was part of the group favoring the NOVA system’s classifying certain foods as UPFs, during the debate. He drew attention to the extent to which the world’s population is getting its calories from UPFs.
Mexico and France get about 30% of calories from these foods. In Canada, it’s 48%. And in the United States, it’s 57%, Dr. Monteiro said.
Studies have found that UPFs, many of which are designed to be exceedingly flavorful and intended to replace consumption of unprocessed whole foods, lead to more overall energy intake, more added sugar in the diet, and less fiber and protein intake, he said.
To further support his arguments, Dr. Monteiro pointed to studies suggesting that it is not just the resulting change in the nutritional intake that is unhealthy, but the UPF manufacturing process itself. When adjusting for fat, sugar, and sodium intake, for example, health outcomes associated with UPFs remain poor, he explained.
“I’m sorry,” he said in the debate. “If you don’t reduce this, you don’t reduce your obesity, your diabetes prevalence.”
A study presented by Jacqueline Vernarelli, PhD, during a different session at the meeting suggested there may be other downsides to consuming UPFs. This research, which was based on the U.S. National Youth Fitness Survey, found that poorer locomotor skills among children aged 3-5 and poorer cardiovascular fitness among those aged 12-15 were associated with getting more calories from UPFs.
Those with lower cardiovascular fitness consumed 1,234 calories a day from UPFs, and those with higher cardiovascular fitness consumed 1,007 calories a day from UPFs (P = .002), according to the new research.
“It’s notable here that, although these differences are significant, both groups are consuming a pretty high proportion of their diet from ultra-processed foods,” said Dr. Vernarelli, associate professor of public health at Sacred Heart University, Fairfield, Conn., during her presentation.
In the debate session, Arne Astrup, MD, PhD, senior project director at the Healthy Weight Center at the Novo Nordisk Foundation, Hellerup, Denmark, presented an opposing view.
He said the definition of UPFs makes it too difficult to categorize many foods, pointing to a study from this year in which about 150 nutrition experts, doctors, and dietitians classified 120 foods. Only three marketed foods and one generic food were classified the same by all the evaluators.
Referring to the study Dr. Astrup cited, Dr. Monteiro said it was a mere “exercise,” and the experts involved in it had conflicts of interest.
Dr. Astrup touted this study’s size and its appearance in the peer-reviewed journal the European Journal of Clinical Nutrition.
Defending his point of view, Dr. Astrup said, “The definition and classification is so ambiguous, and the risk of misclassification is so extremely high, I think we really miss the basic requirement of science, namely that we know what we are talking about,” he said.
If you take an unprocessed food, and insert a “little additive … suddenly it’s an ultra-processed food,” he added.
UPF definition doesn’t flag some unhealthy foods
Susan Roberts, PhD, professor of nutrition at Tufts University, Boston, was a discussant at the debate and touched on the merits of both sides. She noted that the UPF definition doesn’t flag some “clearly unhealthy foods,” such as table sugar, but does flag some healthy ones, such as plant-based burgers – to which Dr. Monteiro said that the system was not a system meant to divide foods into healthy and unhealthy groups, during the debate session.
The inclusion of both healthy and unhealthy foods in NOVA’s definition of a UPF is a serious problem, Dr. Roberts said.
“It’s almost like it’s an emotional classification designed to get at the food industry rather than focusing on health – and I think that’s asking for trouble because it’s just going to be such a mess to tell consumers, ‘Well, this ultra-processed food is healthy and this one isn’t,’ ” she said. What’s happening is the term ultra-processed is being used interchangeably with unhealthy.
The discussion that the UPF classification has generated is useful, Dr. Roberts continued. “This definition grew out of that recognition that we’re engaged in an unprecedented experiment of how unhealthy can you make the world without having a major catastrophe.”
She added that the UPF concept deserves a more formalized and rigorous evaluation.
“This is an important topic for the future of public health, and I think it needs big committees to address it seriously,” she said. “I think we should not be dealing with this individually in different labs.”
Doctor’s take on usefulness of discussing UPF concept with patients
Mark Corkins, MD, who did not participate in the debate at the meeting, said he talks to parents and children about nutrition at every office visit in which he sees a child with an unhealthy weight.
“Persistence wears down resistance,” said the chair of the American Academy of Pediatrics nutrition committee, in an interview.“A consistent message – you say the same thing and you say it multiple times.”
The idea of “ultra-processed foods” plays a role in those conversations, but largely in the background. It’s a topic that’s important for pediatric health, Dr. Corkins said – but he doesn’t make it the focal point.
“It’s not a direct attack on ultra-processed foods that usually I take as my direction,” said Dr. Corkins, who is also chief of pediatric gastroenterology at Le Bonheur Children’s Hospital in Memphis, Tenn.. “What I try to focus on, and what I think the American Academy of Pediatrics would focus on, is that we need to focus on making the diet better.”
He added, “Parents are aware – they don’t call it ultra-processed food, they call it junk food.”
Dr. Corkins continued that he is reluctant to directly challenge parents on feeding their children unhealthy foods – ultra-processed or not – lest he shame them and harm the relationship.
“Guilt as a motivator isn’t really highly successful,” he said, in an interview.
Dr. Astrup reported advisory committee or board member involvement with Green Leaf Medical and RNPC, France. Dr. Roberts reported advisory committee or board member involvement with Danone, and an ownership interest in Instinct Health Science. Dr. Monteiro and Dr. Corkins reported no relevant disclosures.
The NOVA system divides foods into “fresh or minimally processed,” such as strawberries or steel-cut oats; “processed culinary ingredients,” such as olive oil; “processed foods,” such as cheeses; and “ultra-processed foods.” UPFs are defined as “industrial formulations made by deconstructing natural food into its chemical constituents, modifying them and recombining them with additives into products liable to displace all other NOVA food groups.”
According to doctors who presented during the meeting, ultra-processed foods are drawing increased attention, because researchers have been examining them in National Institutes of Health–funded studies and journalists have been writing about them.
During the debate session at the meeting, some experts said that, with obesity and poor health skyrocketing, increased awareness and labeling of UPFs can only be a good thing. In contrast others noted at the meeting that the classification system that has come to be used for identifying UPFs – the NOVA Food Classification system – is too mushy, confusing, and, ultimately unhelpful.
Carlos Monteiro, MD, PhD, professor of nutrition and public health at the University of Sao Paolo, was part of the group favoring the NOVA system’s classifying certain foods as UPFs, during the debate. He drew attention to the extent to which the world’s population is getting its calories from UPFs.
Mexico and France get about 30% of calories from these foods. In Canada, it’s 48%. And in the United States, it’s 57%, Dr. Monteiro said.
Studies have found that UPFs, many of which are designed to be exceedingly flavorful and intended to replace consumption of unprocessed whole foods, lead to more overall energy intake, more added sugar in the diet, and less fiber and protein intake, he said.
To further support his arguments, Dr. Monteiro pointed to studies suggesting that it is not just the resulting change in the nutritional intake that is unhealthy, but the UPF manufacturing process itself. When adjusting for fat, sugar, and sodium intake, for example, health outcomes associated with UPFs remain poor, he explained.
“I’m sorry,” he said in the debate. “If you don’t reduce this, you don’t reduce your obesity, your diabetes prevalence.”
A study presented by Jacqueline Vernarelli, PhD, during a different session at the meeting suggested there may be other downsides to consuming UPFs. This research, which was based on the U.S. National Youth Fitness Survey, found that poorer locomotor skills among children aged 3-5 and poorer cardiovascular fitness among those aged 12-15 were associated with getting more calories from UPFs.
Those with lower cardiovascular fitness consumed 1,234 calories a day from UPFs, and those with higher cardiovascular fitness consumed 1,007 calories a day from UPFs (P = .002), according to the new research.
“It’s notable here that, although these differences are significant, both groups are consuming a pretty high proportion of their diet from ultra-processed foods,” said Dr. Vernarelli, associate professor of public health at Sacred Heart University, Fairfield, Conn., during her presentation.
In the debate session, Arne Astrup, MD, PhD, senior project director at the Healthy Weight Center at the Novo Nordisk Foundation, Hellerup, Denmark, presented an opposing view.
He said the definition of UPFs makes it too difficult to categorize many foods, pointing to a study from this year in which about 150 nutrition experts, doctors, and dietitians classified 120 foods. Only three marketed foods and one generic food were classified the same by all the evaluators.
Referring to the study Dr. Astrup cited, Dr. Monteiro said it was a mere “exercise,” and the experts involved in it had conflicts of interest.
Dr. Astrup touted this study’s size and its appearance in the peer-reviewed journal the European Journal of Clinical Nutrition.
Defending his point of view, Dr. Astrup said, “The definition and classification is so ambiguous, and the risk of misclassification is so extremely high, I think we really miss the basic requirement of science, namely that we know what we are talking about,” he said.
If you take an unprocessed food, and insert a “little additive … suddenly it’s an ultra-processed food,” he added.
UPF definition doesn’t flag some unhealthy foods
Susan Roberts, PhD, professor of nutrition at Tufts University, Boston, was a discussant at the debate and touched on the merits of both sides. She noted that the UPF definition doesn’t flag some “clearly unhealthy foods,” such as table sugar, but does flag some healthy ones, such as plant-based burgers – to which Dr. Monteiro said that the system was not a system meant to divide foods into healthy and unhealthy groups, during the debate session.
The inclusion of both healthy and unhealthy foods in NOVA’s definition of a UPF is a serious problem, Dr. Roberts said.
“It’s almost like it’s an emotional classification designed to get at the food industry rather than focusing on health – and I think that’s asking for trouble because it’s just going to be such a mess to tell consumers, ‘Well, this ultra-processed food is healthy and this one isn’t,’ ” she said. What’s happening is the term ultra-processed is being used interchangeably with unhealthy.
The discussion that the UPF classification has generated is useful, Dr. Roberts continued. “This definition grew out of that recognition that we’re engaged in an unprecedented experiment of how unhealthy can you make the world without having a major catastrophe.”
She added that the UPF concept deserves a more formalized and rigorous evaluation.
“This is an important topic for the future of public health, and I think it needs big committees to address it seriously,” she said. “I think we should not be dealing with this individually in different labs.”
Doctor’s take on usefulness of discussing UPF concept with patients
Mark Corkins, MD, who did not participate in the debate at the meeting, said he talks to parents and children about nutrition at every office visit in which he sees a child with an unhealthy weight.
“Persistence wears down resistance,” said the chair of the American Academy of Pediatrics nutrition committee, in an interview.“A consistent message – you say the same thing and you say it multiple times.”
The idea of “ultra-processed foods” plays a role in those conversations, but largely in the background. It’s a topic that’s important for pediatric health, Dr. Corkins said – but he doesn’t make it the focal point.
“It’s not a direct attack on ultra-processed foods that usually I take as my direction,” said Dr. Corkins, who is also chief of pediatric gastroenterology at Le Bonheur Children’s Hospital in Memphis, Tenn.. “What I try to focus on, and what I think the American Academy of Pediatrics would focus on, is that we need to focus on making the diet better.”
He added, “Parents are aware – they don’t call it ultra-processed food, they call it junk food.”
Dr. Corkins continued that he is reluctant to directly challenge parents on feeding their children unhealthy foods – ultra-processed or not – lest he shame them and harm the relationship.
“Guilt as a motivator isn’t really highly successful,” he said, in an interview.
Dr. Astrup reported advisory committee or board member involvement with Green Leaf Medical and RNPC, France. Dr. Roberts reported advisory committee or board member involvement with Danone, and an ownership interest in Instinct Health Science. Dr. Monteiro and Dr. Corkins reported no relevant disclosures.
FROM NUTRITION 2022
Air pollution tied to ventricular arrhythmias in those with ICDs
Ventricular arrhythmias more commonly occur on days when there are higher levels of air pollution, especially with fine particulate matter (PM), a new study suggests.
The investigators studied the relationship between air pollution and ventricular arrhythmias in Piacenza, Italy by examining 5-year data on patients who received an implantable cardioverter defibrillator (ICD).
They found a significant association between PM2.5 levels and ventricular arrhythmias, especially those treated with direct current shock. Moreover, higher levels of PM2.5 and PM10 were associated with increased risk of all ventricular arrhythmias.
“These data confirm that environmental pollution is not only a climate emergency but also a public health problem,” lead author Alessia Zanni, currently at Maggiore Hospital, Bologna, Italy, and previously at Piacenza Hospital, said in an interview.
“The study suggests that the survival of patients with heart disease is affected not only by pharmacological therapies and advances in cardiology, but also by the air that they breathe,” she said.
The results were presented at European Society of Cardiology Heart Failure 2022.
More ED visits
The World Health Organization estimates around 7 million people die every year from exposure to polluted air, “as 91% of the world’s population lives in areas where air contaminants exceed safety levels,” Dr. Zanni said. Furthermore, “air pollution has been defined as the fourth-highest ranking risk factor for mortality – more important than LDL cholesterol, obesity, physical activity, or alcohol use.”
She noted that Piacenza has “historically been very attentive to the issues of early defibrillation and cardiac arrest.” Her group had previously found a correlation between out-of-hospital cardiac arrests and air pollution in the general population.
Moreover, her group recently observed that ED visits for patients with ICDs “tended to cluster; on some special days, many patients with ICDs had cardiac arrhythmias, and during those days, air pollution levels were particularly high.”
Her group therefore decided to compare the concentration of air pollutants on days when patients suffered from an arrhythmia event versus pollution levels on days without an arrhythmia, she said.
Further piece in a complex puzzle
The researchers studied 146 patients with ICDs between January 2013 and December 2017, assigning exposures (short, mid, and long term) to these patients based on their residential addresses.
They extracted day-by-day urban PM10, PM2.5, CO, NO2, and O3 levels from the Environmental Protection Agency monitoring stations and then, using time-stratified case-crossover analysis methodology, they calculated the association of ventricular arrhythmia onset with 0- to 7-day moving averages of the various air pollutants prior to the event.
Patients had received their ICD to control cardiac dysfunction brought on by previous myocardial infarction (n = 93), genetic or inflammatory conditions (n = 53), secondary prevention after a lethal arrhythmia (n = 67), and primary prevention (n = 79).
Of the 440 ventricular arrhythmias recorded, 322 were treated with antitachycardia pacing, while the remaining 118 were treated with direct current shock.
The researchers found a significant association between PM2.5 levels and ventricular arrhythmia treated with shock, corresponding to a 15% increased risk or every additional 10mg/m3 (P < .019).
They also found that, when PM2.5 concentrations were elevated by 1 mg/m3 for an entire week, compared with average levels, there was a 2.4% higher likelihood of ventricular arrhythmias, regardless of the temperature, and when PM10 was 1 mg/m3 above average for a week, there was a 2.1% increased risk for arrhythmias (odds ratio, 1,024; 95% confidence interval, 1,009-1,040] and OR, 1,021; 95% CI, 1,009-1,033, respectively), Dr. Zanni reported.
“Since the majority of out-of-hospital cardiac arrest causes still remain unclear, our data add a further piece to the complex puzzle of cardiac arrest triggers,” Dr. Zanni commented. “We think that particulate matter can cause acute inflammation of the heart muscle and potentially act as a trigger for lethal cardiac arrhythmias.
“As these toxic particles are emitted from power plants, industries, and cars, we think that cardiovascular research should highlight these new findings to promote green projects among the general population, clarifying the risks to the health of the human being, and we think strategies to prevent air pollutant exposure in high-risk patients [with previous cardiac disease] should be developed,” she added.
Further, “we advise patients at risk, during days with high PM2.5 (> 35 mg/m3) and PM10 (> 50 mg/m3) to use a mask of the N95 type outdoors, to reduce time spent outdoors – particularly in traffic – and to improve home air filtration,” Dr. Zanni said.
Entering the mainstream
In a comment, Joel Kaufman, MD, MPH, professor of internal medicine and environmental health, University of Washington, Seattle, said the study “adds to a fairly substantial literature already on this topic of short-term exposure to air pollution.”
The evidence that air pollutants “can be a trigger of worsening of cardiovascular disease is fairly consistent at this time, and although the effect sizes are small, they are consistent,” said Dr. Kaufman, who was the chair of the writing group for the American Heart Association’s 2020 policy statement, “Guidance to Reduce Cardiovascular Burden of Ambient Air Pollutants.”
“The research into this issue has become clearer during the past 10 years but still is not in the mainstream of most cardiologists’ awareness. They tend to focus more on controlling cholesterol and performing procedures, etc., but there are modifiable risk factors like air pollution that are increasingly recognized as being part of the picture,” said Dr. Kaufman, who was not involved with the current study.
Dr. Zanni added: “It is important that politics work hand in hand with the scientific community in order to win the battle against global warming, which will reduce the number of cardiovascular deaths – the leading cause of death worldwide – as well as environmental integrity.”
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. Dr. Zanni and coauthors and Dr. Kaufman reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Ventricular arrhythmias more commonly occur on days when there are higher levels of air pollution, especially with fine particulate matter (PM), a new study suggests.
The investigators studied the relationship between air pollution and ventricular arrhythmias in Piacenza, Italy by examining 5-year data on patients who received an implantable cardioverter defibrillator (ICD).
They found a significant association between PM2.5 levels and ventricular arrhythmias, especially those treated with direct current shock. Moreover, higher levels of PM2.5 and PM10 were associated with increased risk of all ventricular arrhythmias.
“These data confirm that environmental pollution is not only a climate emergency but also a public health problem,” lead author Alessia Zanni, currently at Maggiore Hospital, Bologna, Italy, and previously at Piacenza Hospital, said in an interview.
“The study suggests that the survival of patients with heart disease is affected not only by pharmacological therapies and advances in cardiology, but also by the air that they breathe,” she said.
The results were presented at European Society of Cardiology Heart Failure 2022.
More ED visits
The World Health Organization estimates around 7 million people die every year from exposure to polluted air, “as 91% of the world’s population lives in areas where air contaminants exceed safety levels,” Dr. Zanni said. Furthermore, “air pollution has been defined as the fourth-highest ranking risk factor for mortality – more important than LDL cholesterol, obesity, physical activity, or alcohol use.”
She noted that Piacenza has “historically been very attentive to the issues of early defibrillation and cardiac arrest.” Her group had previously found a correlation between out-of-hospital cardiac arrests and air pollution in the general population.
Moreover, her group recently observed that ED visits for patients with ICDs “tended to cluster; on some special days, many patients with ICDs had cardiac arrhythmias, and during those days, air pollution levels were particularly high.”
Her group therefore decided to compare the concentration of air pollutants on days when patients suffered from an arrhythmia event versus pollution levels on days without an arrhythmia, she said.
Further piece in a complex puzzle
The researchers studied 146 patients with ICDs between January 2013 and December 2017, assigning exposures (short, mid, and long term) to these patients based on their residential addresses.
They extracted day-by-day urban PM10, PM2.5, CO, NO2, and O3 levels from the Environmental Protection Agency monitoring stations and then, using time-stratified case-crossover analysis methodology, they calculated the association of ventricular arrhythmia onset with 0- to 7-day moving averages of the various air pollutants prior to the event.
Patients had received their ICD to control cardiac dysfunction brought on by previous myocardial infarction (n = 93), genetic or inflammatory conditions (n = 53), secondary prevention after a lethal arrhythmia (n = 67), and primary prevention (n = 79).
Of the 440 ventricular arrhythmias recorded, 322 were treated with antitachycardia pacing, while the remaining 118 were treated with direct current shock.
The researchers found a significant association between PM2.5 levels and ventricular arrhythmia treated with shock, corresponding to a 15% increased risk or every additional 10mg/m3 (P < .019).
They also found that, when PM2.5 concentrations were elevated by 1 mg/m3 for an entire week, compared with average levels, there was a 2.4% higher likelihood of ventricular arrhythmias, regardless of the temperature, and when PM10 was 1 mg/m3 above average for a week, there was a 2.1% increased risk for arrhythmias (odds ratio, 1,024; 95% confidence interval, 1,009-1,040] and OR, 1,021; 95% CI, 1,009-1,033, respectively), Dr. Zanni reported.
“Since the majority of out-of-hospital cardiac arrest causes still remain unclear, our data add a further piece to the complex puzzle of cardiac arrest triggers,” Dr. Zanni commented. “We think that particulate matter can cause acute inflammation of the heart muscle and potentially act as a trigger for lethal cardiac arrhythmias.
“As these toxic particles are emitted from power plants, industries, and cars, we think that cardiovascular research should highlight these new findings to promote green projects among the general population, clarifying the risks to the health of the human being, and we think strategies to prevent air pollutant exposure in high-risk patients [with previous cardiac disease] should be developed,” she added.
Further, “we advise patients at risk, during days with high PM2.5 (> 35 mg/m3) and PM10 (> 50 mg/m3) to use a mask of the N95 type outdoors, to reduce time spent outdoors – particularly in traffic – and to improve home air filtration,” Dr. Zanni said.
Entering the mainstream
In a comment, Joel Kaufman, MD, MPH, professor of internal medicine and environmental health, University of Washington, Seattle, said the study “adds to a fairly substantial literature already on this topic of short-term exposure to air pollution.”
The evidence that air pollutants “can be a trigger of worsening of cardiovascular disease is fairly consistent at this time, and although the effect sizes are small, they are consistent,” said Dr. Kaufman, who was the chair of the writing group for the American Heart Association’s 2020 policy statement, “Guidance to Reduce Cardiovascular Burden of Ambient Air Pollutants.”
“The research into this issue has become clearer during the past 10 years but still is not in the mainstream of most cardiologists’ awareness. They tend to focus more on controlling cholesterol and performing procedures, etc., but there are modifiable risk factors like air pollution that are increasingly recognized as being part of the picture,” said Dr. Kaufman, who was not involved with the current study.
Dr. Zanni added: “It is important that politics work hand in hand with the scientific community in order to win the battle against global warming, which will reduce the number of cardiovascular deaths – the leading cause of death worldwide – as well as environmental integrity.”
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. Dr. Zanni and coauthors and Dr. Kaufman reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Ventricular arrhythmias more commonly occur on days when there are higher levels of air pollution, especially with fine particulate matter (PM), a new study suggests.
The investigators studied the relationship between air pollution and ventricular arrhythmias in Piacenza, Italy by examining 5-year data on patients who received an implantable cardioverter defibrillator (ICD).
They found a significant association between PM2.5 levels and ventricular arrhythmias, especially those treated with direct current shock. Moreover, higher levels of PM2.5 and PM10 were associated with increased risk of all ventricular arrhythmias.
“These data confirm that environmental pollution is not only a climate emergency but also a public health problem,” lead author Alessia Zanni, currently at Maggiore Hospital, Bologna, Italy, and previously at Piacenza Hospital, said in an interview.
“The study suggests that the survival of patients with heart disease is affected not only by pharmacological therapies and advances in cardiology, but also by the air that they breathe,” she said.
The results were presented at European Society of Cardiology Heart Failure 2022.
More ED visits
The World Health Organization estimates around 7 million people die every year from exposure to polluted air, “as 91% of the world’s population lives in areas where air contaminants exceed safety levels,” Dr. Zanni said. Furthermore, “air pollution has been defined as the fourth-highest ranking risk factor for mortality – more important than LDL cholesterol, obesity, physical activity, or alcohol use.”
She noted that Piacenza has “historically been very attentive to the issues of early defibrillation and cardiac arrest.” Her group had previously found a correlation between out-of-hospital cardiac arrests and air pollution in the general population.
Moreover, her group recently observed that ED visits for patients with ICDs “tended to cluster; on some special days, many patients with ICDs had cardiac arrhythmias, and during those days, air pollution levels were particularly high.”
Her group therefore decided to compare the concentration of air pollutants on days when patients suffered from an arrhythmia event versus pollution levels on days without an arrhythmia, she said.
Further piece in a complex puzzle
The researchers studied 146 patients with ICDs between January 2013 and December 2017, assigning exposures (short, mid, and long term) to these patients based on their residential addresses.
They extracted day-by-day urban PM10, PM2.5, CO, NO2, and O3 levels from the Environmental Protection Agency monitoring stations and then, using time-stratified case-crossover analysis methodology, they calculated the association of ventricular arrhythmia onset with 0- to 7-day moving averages of the various air pollutants prior to the event.
Patients had received their ICD to control cardiac dysfunction brought on by previous myocardial infarction (n = 93), genetic or inflammatory conditions (n = 53), secondary prevention after a lethal arrhythmia (n = 67), and primary prevention (n = 79).
Of the 440 ventricular arrhythmias recorded, 322 were treated with antitachycardia pacing, while the remaining 118 were treated with direct current shock.
The researchers found a significant association between PM2.5 levels and ventricular arrhythmia treated with shock, corresponding to a 15% increased risk or every additional 10mg/m3 (P < .019).
They also found that, when PM2.5 concentrations were elevated by 1 mg/m3 for an entire week, compared with average levels, there was a 2.4% higher likelihood of ventricular arrhythmias, regardless of the temperature, and when PM10 was 1 mg/m3 above average for a week, there was a 2.1% increased risk for arrhythmias (odds ratio, 1,024; 95% confidence interval, 1,009-1,040] and OR, 1,021; 95% CI, 1,009-1,033, respectively), Dr. Zanni reported.
“Since the majority of out-of-hospital cardiac arrest causes still remain unclear, our data add a further piece to the complex puzzle of cardiac arrest triggers,” Dr. Zanni commented. “We think that particulate matter can cause acute inflammation of the heart muscle and potentially act as a trigger for lethal cardiac arrhythmias.
“As these toxic particles are emitted from power plants, industries, and cars, we think that cardiovascular research should highlight these new findings to promote green projects among the general population, clarifying the risks to the health of the human being, and we think strategies to prevent air pollutant exposure in high-risk patients [with previous cardiac disease] should be developed,” she added.
Further, “we advise patients at risk, during days with high PM2.5 (> 35 mg/m3) and PM10 (> 50 mg/m3) to use a mask of the N95 type outdoors, to reduce time spent outdoors – particularly in traffic – and to improve home air filtration,” Dr. Zanni said.
Entering the mainstream
In a comment, Joel Kaufman, MD, MPH, professor of internal medicine and environmental health, University of Washington, Seattle, said the study “adds to a fairly substantial literature already on this topic of short-term exposure to air pollution.”
The evidence that air pollutants “can be a trigger of worsening of cardiovascular disease is fairly consistent at this time, and although the effect sizes are small, they are consistent,” said Dr. Kaufman, who was the chair of the writing group for the American Heart Association’s 2020 policy statement, “Guidance to Reduce Cardiovascular Burden of Ambient Air Pollutants.”
“The research into this issue has become clearer during the past 10 years but still is not in the mainstream of most cardiologists’ awareness. They tend to focus more on controlling cholesterol and performing procedures, etc., but there are modifiable risk factors like air pollution that are increasingly recognized as being part of the picture,” said Dr. Kaufman, who was not involved with the current study.
Dr. Zanni added: “It is important that politics work hand in hand with the scientific community in order to win the battle against global warming, which will reduce the number of cardiovascular deaths – the leading cause of death worldwide – as well as environmental integrity.”
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. Dr. Zanni and coauthors and Dr. Kaufman reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ESC HEART FAILURE 2022
New National Lipid Association statement on statin intolerance
The U.S. National Lipid Association has issued a new scientific statement on the management of patients with statin intolerance, which recommends different strategies to help patients stay on statin medications, and also suggests alternatives that can be used in patients who really cannot tolerate statin drugs.
The statement was published online in the Journal of Clinical Lipidology.
It notes that, although statins are generally well tolerated, statin intolerance is reported in 5%-30% of patients and contributes to reduced statin adherence and persistence, as well as higher risk for adverse cardiovascular outcomes.
The statement acknowledges the importance of identifying modifiable risk factors for statin intolerance and recognizes the possibility of a “nocebo” effect, basically the patient expectation of harm resulting in perceived side effects.
To identify a tolerable statin regimen, it recommends that clinicians consider using several different strategies (different statin, dose, and/or dosing frequency), and to classify a patient as having statin intolerance, a minimum of two statins should have been attempted, including at least one at the lowest-approved daily dosage.
The statement says that nonstatin therapy may be required for patients who cannot reach therapeutic objectives with lifestyle and maximal tolerated statin therapy, and in these cases, therapies with outcomes data from randomized trials showing reduced cardiovascular events are favored.
In high and very high-risk patients who are statin intolerant, clinicians should consider initiating nonstatin therapy while additional attempts are made to identify a tolerable statin in order to limit the time of exposure to elevated levels of atherogenic lipoproteins, it suggests.
“There is strong evidence that statins reduce risk of cardiovascular events particularly in patients with atherosclerotic cardiovascular disease, but recent research shows that only about half of patients with ASCVD are on a statin,” Kevin C. Maki, PhD, coauthor of the statement and current president of the National Lipid Association, said in an interview.
“There is an urgent problem with underutilization of statins and undertreatment of ASCVD. And we know that perceived side effects associated with statins are a common reason for discontinuation of these drugs and the consequent failure to manage ASCVD adequately,” he said.
Dr. Maki noted that the NLA’s first message is that, when experiencing symptoms taking statins, a large majority of patients can still tolerate a statin. “They can try a different agent or a different dose. But for those who still can’t tolerate a statin, we then recommend nonstatin therapies and we favor those therapies with evidence from randomized trials.”
He pointed out that many patients who believe they are experiencing side effects from taking statins still experience the same effects on a placebo, a condition known as the nocebo effect.
“Several studies have shown that the nocebo effect is very common and accounts for more than half of perceived statin side effects. It is therefore estimated that many of the complaints of statin intolerance are probably not directly related to the pharmacodynamic actions of the drugs,” Dr. Maki said.
One recent study on the nocebo effect, the SAMSON study, suggested that 90% of symptoms attributed to statins were elicited by placebo tablets too.
But Dr. Maki added that it can be a losing battle for the clinician if patients think their symptoms are related to taking a statin.
“We suggest that clinicians inform patients that most people can tolerate a statin – maybe with a different agent or an alternative dose – and it is really important to lower LDL cholesterol as that will lower the risk of MI and stroke, so we need to find a regimen that works for each individual,” he said. “Most people can find a regimen that works. If this means taking a lower dose of a statin, they can take some additional therapy as well. This is a better situation than stopping taking statins altogether and allowing ASCVD to progress.”
Dr. Maki stressed that statins should still be the first choice as they are effective, taken orally, and inexpensive.
“Other medications do not have all these advantages. For example, PCSK9 inhibitors are very effective but they are expensive and injectable,” he noted. “And while ezetimibe [Zetia] is now generic so inexpensive, it has a more modest effect on LDL-lowering compared to statins, so by itself it is not normally enough for most patients to get to their target LDL, but it is an option for use in combination with a statin.”
He added that the NLA message is to do everything possible to keep patients on a statin, especially patients with preexisting ASCVD.
“We would like these patients to be on high-intensity statins. If they really can’t tolerate this, then they could be on a low-intensity statin plus an additional agent.”
Commenting on the NLA statement, SAMSON study coauthor James Howard, MB BChir, PhD, Imperial College London, said he had reservations about some of the recommendations.
“Whilst I think it is great news that the existence and importance of the nocebo effect is increasingly recognized in international guidelines and statements, I think we need to be very careful about recommending reduced doses and frequencies of statins,” Dr. Howard said.
“Studies such as SAMSON and StatinWISE indicate the vast majority of side effects reported by patients taking statins are not caused by the statin molecule, but instead are caused by either the nocebo effect, or ever-present background symptoms that are wrongly attributed to the statins,” he commented. “Therefore, to recommend that the correct approach in a patient with a history of MI suffering symptoms on 80 mg of atorvastatin is to reduce the dose or try alternate daily dosing. This reinforces the view that these drugs are side-effect prone and need to be carefully titrated.”
Dr. Howard suggested that patients should be educated on the possibility of the nocebo effect or background symptoms and encouraged to retrial statins at the same dose. “If that doesn’t work, then formal recording with a symptom diary might help patients recognize background symptoms,” he added.
Dr. Howard noted that, if symptoms still persist, an “n-of-1” trial could be conducted, in which the patient rotates between multiple periods of taking a statin and a placebo, but he acknowledged that this is expensive and time consuming.
Also commenting, Steve Nissen, MD, Cleveland Clinic, said he thought the NLA statement was “reasonable and thoughtful.”
“Regardless of whether the symptoms are due to the nocebo effect or not, some patients will just not take a statin no matter how hard you try to convince them to persevere, so we do need alternatives,” Dr. Nissen said.
He noted that current alternatives would include the PCSK9 inhibitors and ezetimibe, but a future candidate could be the oral bempedoic acid (Nexletol), which is currently being evaluated in a large outcomes trial (CLEAR Outcomes).
A version of this article first appeared on Medscape.com.
The U.S. National Lipid Association has issued a new scientific statement on the management of patients with statin intolerance, which recommends different strategies to help patients stay on statin medications, and also suggests alternatives that can be used in patients who really cannot tolerate statin drugs.
The statement was published online in the Journal of Clinical Lipidology.
It notes that, although statins are generally well tolerated, statin intolerance is reported in 5%-30% of patients and contributes to reduced statin adherence and persistence, as well as higher risk for adverse cardiovascular outcomes.
The statement acknowledges the importance of identifying modifiable risk factors for statin intolerance and recognizes the possibility of a “nocebo” effect, basically the patient expectation of harm resulting in perceived side effects.
To identify a tolerable statin regimen, it recommends that clinicians consider using several different strategies (different statin, dose, and/or dosing frequency), and to classify a patient as having statin intolerance, a minimum of two statins should have been attempted, including at least one at the lowest-approved daily dosage.
The statement says that nonstatin therapy may be required for patients who cannot reach therapeutic objectives with lifestyle and maximal tolerated statin therapy, and in these cases, therapies with outcomes data from randomized trials showing reduced cardiovascular events are favored.
In high and very high-risk patients who are statin intolerant, clinicians should consider initiating nonstatin therapy while additional attempts are made to identify a tolerable statin in order to limit the time of exposure to elevated levels of atherogenic lipoproteins, it suggests.
“There is strong evidence that statins reduce risk of cardiovascular events particularly in patients with atherosclerotic cardiovascular disease, but recent research shows that only about half of patients with ASCVD are on a statin,” Kevin C. Maki, PhD, coauthor of the statement and current president of the National Lipid Association, said in an interview.
“There is an urgent problem with underutilization of statins and undertreatment of ASCVD. And we know that perceived side effects associated with statins are a common reason for discontinuation of these drugs and the consequent failure to manage ASCVD adequately,” he said.
Dr. Maki noted that the NLA’s first message is that, when experiencing symptoms taking statins, a large majority of patients can still tolerate a statin. “They can try a different agent or a different dose. But for those who still can’t tolerate a statin, we then recommend nonstatin therapies and we favor those therapies with evidence from randomized trials.”
He pointed out that many patients who believe they are experiencing side effects from taking statins still experience the same effects on a placebo, a condition known as the nocebo effect.
“Several studies have shown that the nocebo effect is very common and accounts for more than half of perceived statin side effects. It is therefore estimated that many of the complaints of statin intolerance are probably not directly related to the pharmacodynamic actions of the drugs,” Dr. Maki said.
One recent study on the nocebo effect, the SAMSON study, suggested that 90% of symptoms attributed to statins were elicited by placebo tablets too.
But Dr. Maki added that it can be a losing battle for the clinician if patients think their symptoms are related to taking a statin.
“We suggest that clinicians inform patients that most people can tolerate a statin – maybe with a different agent or an alternative dose – and it is really important to lower LDL cholesterol as that will lower the risk of MI and stroke, so we need to find a regimen that works for each individual,” he said. “Most people can find a regimen that works. If this means taking a lower dose of a statin, they can take some additional therapy as well. This is a better situation than stopping taking statins altogether and allowing ASCVD to progress.”
Dr. Maki stressed that statins should still be the first choice as they are effective, taken orally, and inexpensive.
“Other medications do not have all these advantages. For example, PCSK9 inhibitors are very effective but they are expensive and injectable,” he noted. “And while ezetimibe [Zetia] is now generic so inexpensive, it has a more modest effect on LDL-lowering compared to statins, so by itself it is not normally enough for most patients to get to their target LDL, but it is an option for use in combination with a statin.”
He added that the NLA message is to do everything possible to keep patients on a statin, especially patients with preexisting ASCVD.
“We would like these patients to be on high-intensity statins. If they really can’t tolerate this, then they could be on a low-intensity statin plus an additional agent.”
Commenting on the NLA statement, SAMSON study coauthor James Howard, MB BChir, PhD, Imperial College London, said he had reservations about some of the recommendations.
“Whilst I think it is great news that the existence and importance of the nocebo effect is increasingly recognized in international guidelines and statements, I think we need to be very careful about recommending reduced doses and frequencies of statins,” Dr. Howard said.
“Studies such as SAMSON and StatinWISE indicate the vast majority of side effects reported by patients taking statins are not caused by the statin molecule, but instead are caused by either the nocebo effect, or ever-present background symptoms that are wrongly attributed to the statins,” he commented. “Therefore, to recommend that the correct approach in a patient with a history of MI suffering symptoms on 80 mg of atorvastatin is to reduce the dose or try alternate daily dosing. This reinforces the view that these drugs are side-effect prone and need to be carefully titrated.”
Dr. Howard suggested that patients should be educated on the possibility of the nocebo effect or background symptoms and encouraged to retrial statins at the same dose. “If that doesn’t work, then formal recording with a symptom diary might help patients recognize background symptoms,” he added.
Dr. Howard noted that, if symptoms still persist, an “n-of-1” trial could be conducted, in which the patient rotates between multiple periods of taking a statin and a placebo, but he acknowledged that this is expensive and time consuming.
Also commenting, Steve Nissen, MD, Cleveland Clinic, said he thought the NLA statement was “reasonable and thoughtful.”
“Regardless of whether the symptoms are due to the nocebo effect or not, some patients will just not take a statin no matter how hard you try to convince them to persevere, so we do need alternatives,” Dr. Nissen said.
He noted that current alternatives would include the PCSK9 inhibitors and ezetimibe, but a future candidate could be the oral bempedoic acid (Nexletol), which is currently being evaluated in a large outcomes trial (CLEAR Outcomes).
A version of this article first appeared on Medscape.com.
The U.S. National Lipid Association has issued a new scientific statement on the management of patients with statin intolerance, which recommends different strategies to help patients stay on statin medications, and also suggests alternatives that can be used in patients who really cannot tolerate statin drugs.
The statement was published online in the Journal of Clinical Lipidology.
It notes that, although statins are generally well tolerated, statin intolerance is reported in 5%-30% of patients and contributes to reduced statin adherence and persistence, as well as higher risk for adverse cardiovascular outcomes.
The statement acknowledges the importance of identifying modifiable risk factors for statin intolerance and recognizes the possibility of a “nocebo” effect, basically the patient expectation of harm resulting in perceived side effects.
To identify a tolerable statin regimen, it recommends that clinicians consider using several different strategies (different statin, dose, and/or dosing frequency), and to classify a patient as having statin intolerance, a minimum of two statins should have been attempted, including at least one at the lowest-approved daily dosage.
The statement says that nonstatin therapy may be required for patients who cannot reach therapeutic objectives with lifestyle and maximal tolerated statin therapy, and in these cases, therapies with outcomes data from randomized trials showing reduced cardiovascular events are favored.
In high and very high-risk patients who are statin intolerant, clinicians should consider initiating nonstatin therapy while additional attempts are made to identify a tolerable statin in order to limit the time of exposure to elevated levels of atherogenic lipoproteins, it suggests.
“There is strong evidence that statins reduce risk of cardiovascular events particularly in patients with atherosclerotic cardiovascular disease, but recent research shows that only about half of patients with ASCVD are on a statin,” Kevin C. Maki, PhD, coauthor of the statement and current president of the National Lipid Association, said in an interview.
“There is an urgent problem with underutilization of statins and undertreatment of ASCVD. And we know that perceived side effects associated with statins are a common reason for discontinuation of these drugs and the consequent failure to manage ASCVD adequately,” he said.
Dr. Maki noted that the NLA’s first message is that, when experiencing symptoms taking statins, a large majority of patients can still tolerate a statin. “They can try a different agent or a different dose. But for those who still can’t tolerate a statin, we then recommend nonstatin therapies and we favor those therapies with evidence from randomized trials.”
He pointed out that many patients who believe they are experiencing side effects from taking statins still experience the same effects on a placebo, a condition known as the nocebo effect.
“Several studies have shown that the nocebo effect is very common and accounts for more than half of perceived statin side effects. It is therefore estimated that many of the complaints of statin intolerance are probably not directly related to the pharmacodynamic actions of the drugs,” Dr. Maki said.
One recent study on the nocebo effect, the SAMSON study, suggested that 90% of symptoms attributed to statins were elicited by placebo tablets too.
But Dr. Maki added that it can be a losing battle for the clinician if patients think their symptoms are related to taking a statin.
“We suggest that clinicians inform patients that most people can tolerate a statin – maybe with a different agent or an alternative dose – and it is really important to lower LDL cholesterol as that will lower the risk of MI and stroke, so we need to find a regimen that works for each individual,” he said. “Most people can find a regimen that works. If this means taking a lower dose of a statin, they can take some additional therapy as well. This is a better situation than stopping taking statins altogether and allowing ASCVD to progress.”
Dr. Maki stressed that statins should still be the first choice as they are effective, taken orally, and inexpensive.
“Other medications do not have all these advantages. For example, PCSK9 inhibitors are very effective but they are expensive and injectable,” he noted. “And while ezetimibe [Zetia] is now generic so inexpensive, it has a more modest effect on LDL-lowering compared to statins, so by itself it is not normally enough for most patients to get to their target LDL, but it is an option for use in combination with a statin.”
He added that the NLA message is to do everything possible to keep patients on a statin, especially patients with preexisting ASCVD.
“We would like these patients to be on high-intensity statins. If they really can’t tolerate this, then they could be on a low-intensity statin plus an additional agent.”
Commenting on the NLA statement, SAMSON study coauthor James Howard, MB BChir, PhD, Imperial College London, said he had reservations about some of the recommendations.
“Whilst I think it is great news that the existence and importance of the nocebo effect is increasingly recognized in international guidelines and statements, I think we need to be very careful about recommending reduced doses and frequencies of statins,” Dr. Howard said.
“Studies such as SAMSON and StatinWISE indicate the vast majority of side effects reported by patients taking statins are not caused by the statin molecule, but instead are caused by either the nocebo effect, or ever-present background symptoms that are wrongly attributed to the statins,” he commented. “Therefore, to recommend that the correct approach in a patient with a history of MI suffering symptoms on 80 mg of atorvastatin is to reduce the dose or try alternate daily dosing. This reinforces the view that these drugs are side-effect prone and need to be carefully titrated.”
Dr. Howard suggested that patients should be educated on the possibility of the nocebo effect or background symptoms and encouraged to retrial statins at the same dose. “If that doesn’t work, then formal recording with a symptom diary might help patients recognize background symptoms,” he added.
Dr. Howard noted that, if symptoms still persist, an “n-of-1” trial could be conducted, in which the patient rotates between multiple periods of taking a statin and a placebo, but he acknowledged that this is expensive and time consuming.
Also commenting, Steve Nissen, MD, Cleveland Clinic, said he thought the NLA statement was “reasonable and thoughtful.”
“Regardless of whether the symptoms are due to the nocebo effect or not, some patients will just not take a statin no matter how hard you try to convince them to persevere, so we do need alternatives,” Dr. Nissen said.
He noted that current alternatives would include the PCSK9 inhibitors and ezetimibe, but a future candidate could be the oral bempedoic acid (Nexletol), which is currently being evaluated in a large outcomes trial (CLEAR Outcomes).
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF CLINICAL LIPIDOLOGY
‘Forever chemicals’ linked to hypertension in middle-aged women
In a large, prospective study, researchers found an association between higher blood levels of PFAS and increased risk of hypertension in middle-aged women. Women in the highest tertile of overall PFAS concentrations had a 71% increased risk of developing hypertension.
“Our findings suggest that long-term cumulative exposure, even before midlife, may increase the risk of high blood pressure, and therefore, the benefit of reducing the population exposure to PFAS and potential prevention of high blood pressure and other health conditions would be enormous,” Sung Kyun Park, ScD, MPH, University of Michigan School of Public Health, Ann Arbor, said in an interview.
The study was published online in Hypertension.
Everywhere and forever
“PFAS are forever chemicals as well as everywhere chemicals,” Dr. Park noted.
Possible sources of PFAS exposure run the gamut from nonstick cookware, food wrappers, and waterproof fabrics to cosmetics and drinking water. They have been detected in the blood of most people and have been linked to a variety of health concerns.
“A few studies showed an association between PFAS and hypertension, but those were cross-sectional and examined prevalence of hypertension. It was unclear whether PFAS are associated with the development (incidence) of hypertension,” Dr. Park explained.
For their study, the researchers examined the association between serum concentrations of PFAS and risks of incident hypertension in 1,058 initially normotensive women participating in the Study of Women’s Health Across the Nation-Multi-Pollutant Study (SWAN-MPS). They were followed annually between 1999 and 2017.
During 11,722 person-years of follow-up, 470 of the women developed hypertension, at a rate of 40.1 cases per 1,000 person-years. Hypertension was defined as blood pressure of at least 140 mm Hg systolic or at least 90 mm Hg diastolic or receiving antihypertensive treatment.
Women in the highest tertile of baseline serum concentration of perfluorooctane sulfonate (PFOS) had a 42% higher risk of developing hypertension, compared with peers in the lowest tertile (adjusted hazard ratio, 1.42; 95% confidence interval, 1.19-1.68; P trend = .01).
Similar results were found for perfluorooctanoate (PFOA) and 2-N-ethyl-perfluorooctane sulfonamido acetate (EtFOSAA), with 47% (aHR, 1.47; 95% CI, 1.24-1.75; P trend = .01) and 42% (aHR, 1.42; 95% CI, 1.19-1.70; P trend = .01) higher risks of incident hypertension, comparing the highest to the lowest tertiles.
The risks persisted after adjusting for various factors, including race, study site, education, financial strain, smoking status, alcohol use, total calorie intake, and menopausal status.
In the PFAS “mixture” analysis, women in the highest tertile of overall PFAS concentrations were 71% more likely to develop hypertension during follow-up, compared with women in the lowest tertile (aHR, 1.71; 95% CI, 1.15-2.54; P trend = .008).
“These findings suggest that PFAS might be an underappreciated contributing factor to women’s cardiovascular disease risk,” the researchers write.
They caution that the study only included middle-aged women and that it is unclear whether the findings hold for middle-aged men.
“This is an important question, but the answer is that we do not know,” Dr. Park told this news organization.
“Women become more susceptible to metabolic changes and hypertension risk during the menopausal transition. Our findings suggest that PFAS may play a role in the development of hypertension in women during this critical life stage,” Dr. Park said.
The researchers say more research is needed to confirm and expand the findings and to find ways to reduce PFAS exposure.
“If confirmed in future studies, these findings suggest that understanding human exposure to PFAS and developing effective strategies to reduce PFAS exposure may help prevent the development of hypertension and thereby reduce the global burden of CVD,” the researchers write.
‘The more we learn, the worse it gets’
This is an “interesting” study and shows that “the more we learn about PFAS, the worse it seems to get,” Ankur Shah, MD, division of kidney disease and hypertension, Warren Alpert Medical School of Brown University, Providence, R.I., said in an interview.
“This multisite, multiracial and multiethnic, community-based longitudinal study establishes an association between PFAS and hypertension,” said Dr. Shah, who wasn’t involved in the study.
“This adds to a growing literature base of associations of PFAS with illnesses, including malignancy, thyroid disorders, diabetes, ulcerative colitis, hyperlipidemia, and pregnancy-induced hypertension,” he noted.
Dr. Shah also noted that the authors adjusted for race and ethnicity, study site, education, financial strain, smoking status, environmental tobacco smoke, alcohol consumption, total calorie intake, and menopausal status “and still found a strong association.”
“Still to be determined are both whether PFAS are the causative agent or if there is an unmeasured/unadjusted for entity which has resulted in both increased PFAS exposure and hypertension, as well as if PFAS are causative, if reduction in PFAS exposure would be result in blood pressure reduction,” Dr. Shah added.
The study had no sources of funding. Dr. Park and Dr. Shah have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a large, prospective study, researchers found an association between higher blood levels of PFAS and increased risk of hypertension in middle-aged women. Women in the highest tertile of overall PFAS concentrations had a 71% increased risk of developing hypertension.
“Our findings suggest that long-term cumulative exposure, even before midlife, may increase the risk of high blood pressure, and therefore, the benefit of reducing the population exposure to PFAS and potential prevention of high blood pressure and other health conditions would be enormous,” Sung Kyun Park, ScD, MPH, University of Michigan School of Public Health, Ann Arbor, said in an interview.
The study was published online in Hypertension.
Everywhere and forever
“PFAS are forever chemicals as well as everywhere chemicals,” Dr. Park noted.
Possible sources of PFAS exposure run the gamut from nonstick cookware, food wrappers, and waterproof fabrics to cosmetics and drinking water. They have been detected in the blood of most people and have been linked to a variety of health concerns.
“A few studies showed an association between PFAS and hypertension, but those were cross-sectional and examined prevalence of hypertension. It was unclear whether PFAS are associated with the development (incidence) of hypertension,” Dr. Park explained.
For their study, the researchers examined the association between serum concentrations of PFAS and risks of incident hypertension in 1,058 initially normotensive women participating in the Study of Women’s Health Across the Nation-Multi-Pollutant Study (SWAN-MPS). They were followed annually between 1999 and 2017.
During 11,722 person-years of follow-up, 470 of the women developed hypertension, at a rate of 40.1 cases per 1,000 person-years. Hypertension was defined as blood pressure of at least 140 mm Hg systolic or at least 90 mm Hg diastolic or receiving antihypertensive treatment.
Women in the highest tertile of baseline serum concentration of perfluorooctane sulfonate (PFOS) had a 42% higher risk of developing hypertension, compared with peers in the lowest tertile (adjusted hazard ratio, 1.42; 95% confidence interval, 1.19-1.68; P trend = .01).
Similar results were found for perfluorooctanoate (PFOA) and 2-N-ethyl-perfluorooctane sulfonamido acetate (EtFOSAA), with 47% (aHR, 1.47; 95% CI, 1.24-1.75; P trend = .01) and 42% (aHR, 1.42; 95% CI, 1.19-1.70; P trend = .01) higher risks of incident hypertension, comparing the highest to the lowest tertiles.
The risks persisted after adjusting for various factors, including race, study site, education, financial strain, smoking status, alcohol use, total calorie intake, and menopausal status.
In the PFAS “mixture” analysis, women in the highest tertile of overall PFAS concentrations were 71% more likely to develop hypertension during follow-up, compared with women in the lowest tertile (aHR, 1.71; 95% CI, 1.15-2.54; P trend = .008).
“These findings suggest that PFAS might be an underappreciated contributing factor to women’s cardiovascular disease risk,” the researchers write.
They caution that the study only included middle-aged women and that it is unclear whether the findings hold for middle-aged men.
“This is an important question, but the answer is that we do not know,” Dr. Park told this news organization.
“Women become more susceptible to metabolic changes and hypertension risk during the menopausal transition. Our findings suggest that PFAS may play a role in the development of hypertension in women during this critical life stage,” Dr. Park said.
The researchers say more research is needed to confirm and expand the findings and to find ways to reduce PFAS exposure.
“If confirmed in future studies, these findings suggest that understanding human exposure to PFAS and developing effective strategies to reduce PFAS exposure may help prevent the development of hypertension and thereby reduce the global burden of CVD,” the researchers write.
‘The more we learn, the worse it gets’
This is an “interesting” study and shows that “the more we learn about PFAS, the worse it seems to get,” Ankur Shah, MD, division of kidney disease and hypertension, Warren Alpert Medical School of Brown University, Providence, R.I., said in an interview.
“This multisite, multiracial and multiethnic, community-based longitudinal study establishes an association between PFAS and hypertension,” said Dr. Shah, who wasn’t involved in the study.
“This adds to a growing literature base of associations of PFAS with illnesses, including malignancy, thyroid disorders, diabetes, ulcerative colitis, hyperlipidemia, and pregnancy-induced hypertension,” he noted.
Dr. Shah also noted that the authors adjusted for race and ethnicity, study site, education, financial strain, smoking status, environmental tobacco smoke, alcohol consumption, total calorie intake, and menopausal status “and still found a strong association.”
“Still to be determined are both whether PFAS are the causative agent or if there is an unmeasured/unadjusted for entity which has resulted in both increased PFAS exposure and hypertension, as well as if PFAS are causative, if reduction in PFAS exposure would be result in blood pressure reduction,” Dr. Shah added.
The study had no sources of funding. Dr. Park and Dr. Shah have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a large, prospective study, researchers found an association between higher blood levels of PFAS and increased risk of hypertension in middle-aged women. Women in the highest tertile of overall PFAS concentrations had a 71% increased risk of developing hypertension.
“Our findings suggest that long-term cumulative exposure, even before midlife, may increase the risk of high blood pressure, and therefore, the benefit of reducing the population exposure to PFAS and potential prevention of high blood pressure and other health conditions would be enormous,” Sung Kyun Park, ScD, MPH, University of Michigan School of Public Health, Ann Arbor, said in an interview.
The study was published online in Hypertension.
Everywhere and forever
“PFAS are forever chemicals as well as everywhere chemicals,” Dr. Park noted.
Possible sources of PFAS exposure run the gamut from nonstick cookware, food wrappers, and waterproof fabrics to cosmetics and drinking water. They have been detected in the blood of most people and have been linked to a variety of health concerns.
“A few studies showed an association between PFAS and hypertension, but those were cross-sectional and examined prevalence of hypertension. It was unclear whether PFAS are associated with the development (incidence) of hypertension,” Dr. Park explained.
For their study, the researchers examined the association between serum concentrations of PFAS and risks of incident hypertension in 1,058 initially normotensive women participating in the Study of Women’s Health Across the Nation-Multi-Pollutant Study (SWAN-MPS). They were followed annually between 1999 and 2017.
During 11,722 person-years of follow-up, 470 of the women developed hypertension, at a rate of 40.1 cases per 1,000 person-years. Hypertension was defined as blood pressure of at least 140 mm Hg systolic or at least 90 mm Hg diastolic or receiving antihypertensive treatment.
Women in the highest tertile of baseline serum concentration of perfluorooctane sulfonate (PFOS) had a 42% higher risk of developing hypertension, compared with peers in the lowest tertile (adjusted hazard ratio, 1.42; 95% confidence interval, 1.19-1.68; P trend = .01).
Similar results were found for perfluorooctanoate (PFOA) and 2-N-ethyl-perfluorooctane sulfonamido acetate (EtFOSAA), with 47% (aHR, 1.47; 95% CI, 1.24-1.75; P trend = .01) and 42% (aHR, 1.42; 95% CI, 1.19-1.70; P trend = .01) higher risks of incident hypertension, comparing the highest to the lowest tertiles.
The risks persisted after adjusting for various factors, including race, study site, education, financial strain, smoking status, alcohol use, total calorie intake, and menopausal status.
In the PFAS “mixture” analysis, women in the highest tertile of overall PFAS concentrations were 71% more likely to develop hypertension during follow-up, compared with women in the lowest tertile (aHR, 1.71; 95% CI, 1.15-2.54; P trend = .008).
“These findings suggest that PFAS might be an underappreciated contributing factor to women’s cardiovascular disease risk,” the researchers write.
They caution that the study only included middle-aged women and that it is unclear whether the findings hold for middle-aged men.
“This is an important question, but the answer is that we do not know,” Dr. Park told this news organization.
“Women become more susceptible to metabolic changes and hypertension risk during the menopausal transition. Our findings suggest that PFAS may play a role in the development of hypertension in women during this critical life stage,” Dr. Park said.
The researchers say more research is needed to confirm and expand the findings and to find ways to reduce PFAS exposure.
“If confirmed in future studies, these findings suggest that understanding human exposure to PFAS and developing effective strategies to reduce PFAS exposure may help prevent the development of hypertension and thereby reduce the global burden of CVD,” the researchers write.
‘The more we learn, the worse it gets’
This is an “interesting” study and shows that “the more we learn about PFAS, the worse it seems to get,” Ankur Shah, MD, division of kidney disease and hypertension, Warren Alpert Medical School of Brown University, Providence, R.I., said in an interview.
“This multisite, multiracial and multiethnic, community-based longitudinal study establishes an association between PFAS and hypertension,” said Dr. Shah, who wasn’t involved in the study.
“This adds to a growing literature base of associations of PFAS with illnesses, including malignancy, thyroid disorders, diabetes, ulcerative colitis, hyperlipidemia, and pregnancy-induced hypertension,” he noted.
Dr. Shah also noted that the authors adjusted for race and ethnicity, study site, education, financial strain, smoking status, environmental tobacco smoke, alcohol consumption, total calorie intake, and menopausal status “and still found a strong association.”
“Still to be determined are both whether PFAS are the causative agent or if there is an unmeasured/unadjusted for entity which has resulted in both increased PFAS exposure and hypertension, as well as if PFAS are causative, if reduction in PFAS exposure would be result in blood pressure reduction,” Dr. Shah added.
The study had no sources of funding. Dr. Park and Dr. Shah have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM HYPERTENSION
Cause of death in pig heart recipient: New clues
The underlying cause of David Bennett’s death on March 8, two months after he received the heart of a genetically altered pig, remains unknown and is only slightly less mysterious for what can likely be ruled out, suggests a progress report on the case from the director of the cardiac xenotransplantation program where the pioneering surgery took place.
Mr. Bennett died in “diastolic heart failure,” reported Muhammad M. Mohiuddin, MBBS, University of Maryland School of Medicine, Baltimore, “but the mechanism is still under investigation.”
Although the immediate cause could have been single or multiple, evidence so far does not point to immune rejection nor does it support a role for a recently proposed suspect, infection by porcine cytomegalovirus (PCMV), Dr. Mohiuddin observed in front of a standing-room-only audience June 6 at the American Transplant Congress (ATC) in Boston. The congress is a joint meeting of the American Society of Transplant Surgeons (ASTS) and the American Society of Transplantation (AST).
Rocky clinical course
Early characterizations of the patient’s death focused more on his diminished, end-stage clinical condition at the time of the surgery than on immune rejection or other direct effects of the xenograft or on the first-of-its-kind procedure itself.
The 57-year-old Mr. Bennett had presented to the University of Maryland team with nonischemic cardiomyopathy, on multiple inotropes, and requiring an intra-aortic balloon pump, Dr. Mohiuddin said in his ATC presentation. The patient had suffered multiple arrests and resuscitations, and by the time of surgery had been hospitalized for almost 2 months, including 40 days on veno-arterial extracorporeal membrane oxygenation (ECMO).
The transplant procedure itself went as planned until removal of the aortic cross clamp, which triggered a type-A aortic dissection. “We put a graft in the ascending aorta and a stent in the descending aorta. Even after 2 days, we found the dissection extending to the renal artery, so we had to go back and also put a stent in the renal artery,” Dr. Mohiuddin said.
Mr. Bennett also underwent two exploratory laparotomies in the first 10 days after transplantation, after CT imaging revealed signs of possible bowel inflammation and ischemia.
Further, he had to fight back a series of infections that led to major changes to his experimental drug regimen, which included immunosuppressants methylprednisolone and mycophenolate mofetil (MMF), the investigational anti-CD40 antibody KPL-404 (Kiniksa Pharmaceuticals), and the anti-inflammatories etanercept (Enbrel) and tocilizumab (Actemra).
One episode of sepsis, in particular, forced temporary withdrawal of MMF and a reduction in methylprednisolone dosage. It’s unknown whether the 30-day MMF suspension played a role in Mr. Bennett’s ultimate clinical deterioration and death, but it’s “highly possible,” Dr. Mohiuddin said in an interview.
Realistically, Mr. Bennett’s death was likely “multifactorial,” Dr. Mohiuddin said. He was in such poor clinical condition going into the procedure, and afterward confronted so many clinical challenges, that “it’s very difficult to say that one thing caused it.”
That hasn’t lessened speculation that the patient’s heart failed secondary to immunologic rejection or PCMV infection, either in Mr. Bennett or the donor pig.
A role for PCMV?
Weeks after Mr. Bennett’s death, as previously reported, his surgeon announced at a public forum that PCMV had been identified in the transplanted heart and in tissues of the donor pig. Mr. Bennett’s circulation showed traces of the viral DNA but not of the virus itself.
The presence of PCMV in transplanted porcine hearts is a well-recognized potential hazard in animal models but is considered avoidable with proper screening. In Mr. Bennett’s case, preoperative screening of the pig donor missed signs of the virus.
Still, PCMV could potentially have contributed to Mr. Bennett’s death, acknowledged Bartley P. Griffith, MD, University of Maryland School of Medicine, who had announced the PCMV finding in an AST-sponsored April 20 webcast.
Preclinical evidence does suggest that PCMV can harm a xenograft organ, observed David H. Sachs, MD, Columbia University Medical Center, New York, from the audience during the comment period after Dr. Mohiuddin’s presentation.
“Each species has a CMV, and they’re quite species-specific,” observed the renowned surgeon and xenotransplantation immunologist. “We showed almost 10 years ago that if PCMV was in a pig kidney, it led to a much shortened survival of the pig kidney in a baboon. There was never any evidence, however, that the CMV infected the baboon or any baboon cells.”
Dr. Sachs asked Dr. Mohiuddin for confirmation that Mr. Bennett displayed no more than DNAemia, circulating cell-free PCMV DNA presumably shed from the porcine heart, but no sign of the virus itself outside of the heart’s porcine cells.
Cell-free DNA had shown up in Mr. Bennett’s circulation about 20 days after the surgery, with concentrations rising until at least day 50. Post-hoc polymerase chain reaction (PCR) testing disclosed PCMV only in the pig’s spleen and porcine cells of the transplanted heart, Dr. Mohiuddin noted.
“We have not found any evidence that the patient was infected by PCMV,” nor was there evidence of any disease related to PCMV, Dr. Mohiuddin replied.
Nor of ongoing rejection
Mr. Bennett’s new heart passed a critical test in the first post-implantation hours by avoiding acute rejection, a potentially disastrous outcome that three of the pig’s 10 gene edits had been designed to prevent.
Although chronic immune rejection was always a concern despite Mr. Bennett’s novel immunosuppressant regimen, myocardial biopsy on postoperative days 34, 50, and 56 and necropsy showed “no signs of typical xenograft rejection,” Dr. Mohiuddin said at the ATC presentation. But “there’s a chance of atypical rejection which we were not accustomed to.”
By day 50, his diastolic function showed echocardiographic signs of deterioration, and “we started seeing interstitial edema with some extravasation of red blood cells, which we thought would resolve over a period of time,” he said. Eventually, however, “we saw that turn into fibroblasts and scar tissue.”
Mr. Bennett once again went on veno-arterial ECMO but died 10 days later. Once they had seen histologic evidence of fibrosis, Dr. Mohiuddin told this news organization, the team believed the myocardial injury was irreversible. “That was the reason we gave up on recovery.”
Mr. Bennett’s xenotransplantation journey has taught the field a lot, he said. “By no means was this a failure; we consider this a huge success. You can do all the experiments in animal models, but you won’t find out the true mechanism of rejection unless you do these kinds of human experiments.”
Looking ahead to clinical trials
Research involving humans is always subject to vagaries of human nature, including degree of adherence to prescribed therapy and – in xenotransplantation – precautions in place to mitigate any risks to public health. Such risks theoretically include transfer of porcine viruses or other pathogens to the patient and subsequent release into the general population.
Looking ahead to the possibility of clinical trials after this successful xenotransplantation experience, transplant nephrologist and epidemiologist Peter P. Reese, MD, PhD, University of Pennsylvania, Philadelphia, raised the potentially controversial issue in discussion following Dr. Mohiuddin’s presentation.
It’s known that Mr. Bennett had been repeatedly turned down for a conventional allograft transplant primarily because of his history of treatment noncompliance. Should such a record, Dr. Reese asked, be a relative contraindication to enrollment in any future xenotransplantation trials? Or does the field need a standardized gauge of a patient’s readiness, once discharged, to adhere not only to all medications – including those that fight infection – but also with rules established for public safety, such as routine contact reporting?
“It makes me wonder about choosing a noncompliant patient for these trials,” Dr. Reese said. “If we discharge a patient from the hospital who is at risk for a zoonotic infection that could spread if they basically refuse to cooperate with us or with public health authorities, it really could have negative consequences for the reputation of the field.”
Dr. Mohiuddin agreed such concerns are valid. Mr. Bennett “and all his immediate contacts” signed consent forms acknowledging their willingness to be followed should he be discharged. Mr. Bennett himself “signed a consent to inform us if he has any other intimate contact with someone,” he said in an interview.
“But those are only on paper.” Had Mr. Bennett survived to be discharged, Dr. Mohuiddin said, “no one knows how he would have behaved.”
Dr. Mohiuddin said the research staff had prepared to monitor Mr. Bennett at his home if that’s what it took. “We were ready to follow him as long as we could. There was a surveillance plan in place.”
A version of this article first appeared on Medscape.com.
The underlying cause of David Bennett’s death on March 8, two months after he received the heart of a genetically altered pig, remains unknown and is only slightly less mysterious for what can likely be ruled out, suggests a progress report on the case from the director of the cardiac xenotransplantation program where the pioneering surgery took place.
Mr. Bennett died in “diastolic heart failure,” reported Muhammad M. Mohiuddin, MBBS, University of Maryland School of Medicine, Baltimore, “but the mechanism is still under investigation.”
Although the immediate cause could have been single or multiple, evidence so far does not point to immune rejection nor does it support a role for a recently proposed suspect, infection by porcine cytomegalovirus (PCMV), Dr. Mohiuddin observed in front of a standing-room-only audience June 6 at the American Transplant Congress (ATC) in Boston. The congress is a joint meeting of the American Society of Transplant Surgeons (ASTS) and the American Society of Transplantation (AST).
Rocky clinical course
Early characterizations of the patient’s death focused more on his diminished, end-stage clinical condition at the time of the surgery than on immune rejection or other direct effects of the xenograft or on the first-of-its-kind procedure itself.
The 57-year-old Mr. Bennett had presented to the University of Maryland team with nonischemic cardiomyopathy, on multiple inotropes, and requiring an intra-aortic balloon pump, Dr. Mohiuddin said in his ATC presentation. The patient had suffered multiple arrests and resuscitations, and by the time of surgery had been hospitalized for almost 2 months, including 40 days on veno-arterial extracorporeal membrane oxygenation (ECMO).
The transplant procedure itself went as planned until removal of the aortic cross clamp, which triggered a type-A aortic dissection. “We put a graft in the ascending aorta and a stent in the descending aorta. Even after 2 days, we found the dissection extending to the renal artery, so we had to go back and also put a stent in the renal artery,” Dr. Mohiuddin said.
Mr. Bennett also underwent two exploratory laparotomies in the first 10 days after transplantation, after CT imaging revealed signs of possible bowel inflammation and ischemia.
Further, he had to fight back a series of infections that led to major changes to his experimental drug regimen, which included immunosuppressants methylprednisolone and mycophenolate mofetil (MMF), the investigational anti-CD40 antibody KPL-404 (Kiniksa Pharmaceuticals), and the anti-inflammatories etanercept (Enbrel) and tocilizumab (Actemra).
One episode of sepsis, in particular, forced temporary withdrawal of MMF and a reduction in methylprednisolone dosage. It’s unknown whether the 30-day MMF suspension played a role in Mr. Bennett’s ultimate clinical deterioration and death, but it’s “highly possible,” Dr. Mohiuddin said in an interview.
Realistically, Mr. Bennett’s death was likely “multifactorial,” Dr. Mohiuddin said. He was in such poor clinical condition going into the procedure, and afterward confronted so many clinical challenges, that “it’s very difficult to say that one thing caused it.”
That hasn’t lessened speculation that the patient’s heart failed secondary to immunologic rejection or PCMV infection, either in Mr. Bennett or the donor pig.
A role for PCMV?
Weeks after Mr. Bennett’s death, as previously reported, his surgeon announced at a public forum that PCMV had been identified in the transplanted heart and in tissues of the donor pig. Mr. Bennett’s circulation showed traces of the viral DNA but not of the virus itself.
The presence of PCMV in transplanted porcine hearts is a well-recognized potential hazard in animal models but is considered avoidable with proper screening. In Mr. Bennett’s case, preoperative screening of the pig donor missed signs of the virus.
Still, PCMV could potentially have contributed to Mr. Bennett’s death, acknowledged Bartley P. Griffith, MD, University of Maryland School of Medicine, who had announced the PCMV finding in an AST-sponsored April 20 webcast.
Preclinical evidence does suggest that PCMV can harm a xenograft organ, observed David H. Sachs, MD, Columbia University Medical Center, New York, from the audience during the comment period after Dr. Mohiuddin’s presentation.
“Each species has a CMV, and they’re quite species-specific,” observed the renowned surgeon and xenotransplantation immunologist. “We showed almost 10 years ago that if PCMV was in a pig kidney, it led to a much shortened survival of the pig kidney in a baboon. There was never any evidence, however, that the CMV infected the baboon or any baboon cells.”
Dr. Sachs asked Dr. Mohiuddin for confirmation that Mr. Bennett displayed no more than DNAemia, circulating cell-free PCMV DNA presumably shed from the porcine heart, but no sign of the virus itself outside of the heart’s porcine cells.
Cell-free DNA had shown up in Mr. Bennett’s circulation about 20 days after the surgery, with concentrations rising until at least day 50. Post-hoc polymerase chain reaction (PCR) testing disclosed PCMV only in the pig’s spleen and porcine cells of the transplanted heart, Dr. Mohiuddin noted.
“We have not found any evidence that the patient was infected by PCMV,” nor was there evidence of any disease related to PCMV, Dr. Mohiuddin replied.
Nor of ongoing rejection
Mr. Bennett’s new heart passed a critical test in the first post-implantation hours by avoiding acute rejection, a potentially disastrous outcome that three of the pig’s 10 gene edits had been designed to prevent.
Although chronic immune rejection was always a concern despite Mr. Bennett’s novel immunosuppressant regimen, myocardial biopsy on postoperative days 34, 50, and 56 and necropsy showed “no signs of typical xenograft rejection,” Dr. Mohiuddin said at the ATC presentation. But “there’s a chance of atypical rejection which we were not accustomed to.”
By day 50, his diastolic function showed echocardiographic signs of deterioration, and “we started seeing interstitial edema with some extravasation of red blood cells, which we thought would resolve over a period of time,” he said. Eventually, however, “we saw that turn into fibroblasts and scar tissue.”
Mr. Bennett once again went on veno-arterial ECMO but died 10 days later. Once they had seen histologic evidence of fibrosis, Dr. Mohiuddin told this news organization, the team believed the myocardial injury was irreversible. “That was the reason we gave up on recovery.”
Mr. Bennett’s xenotransplantation journey has taught the field a lot, he said. “By no means was this a failure; we consider this a huge success. You can do all the experiments in animal models, but you won’t find out the true mechanism of rejection unless you do these kinds of human experiments.”
Looking ahead to clinical trials
Research involving humans is always subject to vagaries of human nature, including degree of adherence to prescribed therapy and – in xenotransplantation – precautions in place to mitigate any risks to public health. Such risks theoretically include transfer of porcine viruses or other pathogens to the patient and subsequent release into the general population.
Looking ahead to the possibility of clinical trials after this successful xenotransplantation experience, transplant nephrologist and epidemiologist Peter P. Reese, MD, PhD, University of Pennsylvania, Philadelphia, raised the potentially controversial issue in discussion following Dr. Mohiuddin’s presentation.
It’s known that Mr. Bennett had been repeatedly turned down for a conventional allograft transplant primarily because of his history of treatment noncompliance. Should such a record, Dr. Reese asked, be a relative contraindication to enrollment in any future xenotransplantation trials? Or does the field need a standardized gauge of a patient’s readiness, once discharged, to adhere not only to all medications – including those that fight infection – but also with rules established for public safety, such as routine contact reporting?
“It makes me wonder about choosing a noncompliant patient for these trials,” Dr. Reese said. “If we discharge a patient from the hospital who is at risk for a zoonotic infection that could spread if they basically refuse to cooperate with us or with public health authorities, it really could have negative consequences for the reputation of the field.”
Dr. Mohiuddin agreed such concerns are valid. Mr. Bennett “and all his immediate contacts” signed consent forms acknowledging their willingness to be followed should he be discharged. Mr. Bennett himself “signed a consent to inform us if he has any other intimate contact with someone,” he said in an interview.
“But those are only on paper.” Had Mr. Bennett survived to be discharged, Dr. Mohuiddin said, “no one knows how he would have behaved.”
Dr. Mohiuddin said the research staff had prepared to monitor Mr. Bennett at his home if that’s what it took. “We were ready to follow him as long as we could. There was a surveillance plan in place.”
A version of this article first appeared on Medscape.com.
The underlying cause of David Bennett’s death on March 8, two months after he received the heart of a genetically altered pig, remains unknown and is only slightly less mysterious for what can likely be ruled out, suggests a progress report on the case from the director of the cardiac xenotransplantation program where the pioneering surgery took place.
Mr. Bennett died in “diastolic heart failure,” reported Muhammad M. Mohiuddin, MBBS, University of Maryland School of Medicine, Baltimore, “but the mechanism is still under investigation.”
Although the immediate cause could have been single or multiple, evidence so far does not point to immune rejection nor does it support a role for a recently proposed suspect, infection by porcine cytomegalovirus (PCMV), Dr. Mohiuddin observed in front of a standing-room-only audience June 6 at the American Transplant Congress (ATC) in Boston. The congress is a joint meeting of the American Society of Transplant Surgeons (ASTS) and the American Society of Transplantation (AST).
Rocky clinical course
Early characterizations of the patient’s death focused more on his diminished, end-stage clinical condition at the time of the surgery than on immune rejection or other direct effects of the xenograft or on the first-of-its-kind procedure itself.
The 57-year-old Mr. Bennett had presented to the University of Maryland team with nonischemic cardiomyopathy, on multiple inotropes, and requiring an intra-aortic balloon pump, Dr. Mohiuddin said in his ATC presentation. The patient had suffered multiple arrests and resuscitations, and by the time of surgery had been hospitalized for almost 2 months, including 40 days on veno-arterial extracorporeal membrane oxygenation (ECMO).
The transplant procedure itself went as planned until removal of the aortic cross clamp, which triggered a type-A aortic dissection. “We put a graft in the ascending aorta and a stent in the descending aorta. Even after 2 days, we found the dissection extending to the renal artery, so we had to go back and also put a stent in the renal artery,” Dr. Mohiuddin said.
Mr. Bennett also underwent two exploratory laparotomies in the first 10 days after transplantation, after CT imaging revealed signs of possible bowel inflammation and ischemia.
Further, he had to fight back a series of infections that led to major changes to his experimental drug regimen, which included immunosuppressants methylprednisolone and mycophenolate mofetil (MMF), the investigational anti-CD40 antibody KPL-404 (Kiniksa Pharmaceuticals), and the anti-inflammatories etanercept (Enbrel) and tocilizumab (Actemra).
One episode of sepsis, in particular, forced temporary withdrawal of MMF and a reduction in methylprednisolone dosage. It’s unknown whether the 30-day MMF suspension played a role in Mr. Bennett’s ultimate clinical deterioration and death, but it’s “highly possible,” Dr. Mohiuddin said in an interview.
Realistically, Mr. Bennett’s death was likely “multifactorial,” Dr. Mohiuddin said. He was in such poor clinical condition going into the procedure, and afterward confronted so many clinical challenges, that “it’s very difficult to say that one thing caused it.”
That hasn’t lessened speculation that the patient’s heart failed secondary to immunologic rejection or PCMV infection, either in Mr. Bennett or the donor pig.
A role for PCMV?
Weeks after Mr. Bennett’s death, as previously reported, his surgeon announced at a public forum that PCMV had been identified in the transplanted heart and in tissues of the donor pig. Mr. Bennett’s circulation showed traces of the viral DNA but not of the virus itself.
The presence of PCMV in transplanted porcine hearts is a well-recognized potential hazard in animal models but is considered avoidable with proper screening. In Mr. Bennett’s case, preoperative screening of the pig donor missed signs of the virus.
Still, PCMV could potentially have contributed to Mr. Bennett’s death, acknowledged Bartley P. Griffith, MD, University of Maryland School of Medicine, who had announced the PCMV finding in an AST-sponsored April 20 webcast.
Preclinical evidence does suggest that PCMV can harm a xenograft organ, observed David H. Sachs, MD, Columbia University Medical Center, New York, from the audience during the comment period after Dr. Mohiuddin’s presentation.
“Each species has a CMV, and they’re quite species-specific,” observed the renowned surgeon and xenotransplantation immunologist. “We showed almost 10 years ago that if PCMV was in a pig kidney, it led to a much shortened survival of the pig kidney in a baboon. There was never any evidence, however, that the CMV infected the baboon or any baboon cells.”
Dr. Sachs asked Dr. Mohiuddin for confirmation that Mr. Bennett displayed no more than DNAemia, circulating cell-free PCMV DNA presumably shed from the porcine heart, but no sign of the virus itself outside of the heart’s porcine cells.
Cell-free DNA had shown up in Mr. Bennett’s circulation about 20 days after the surgery, with concentrations rising until at least day 50. Post-hoc polymerase chain reaction (PCR) testing disclosed PCMV only in the pig’s spleen and porcine cells of the transplanted heart, Dr. Mohiuddin noted.
“We have not found any evidence that the patient was infected by PCMV,” nor was there evidence of any disease related to PCMV, Dr. Mohiuddin replied.
Nor of ongoing rejection
Mr. Bennett’s new heart passed a critical test in the first post-implantation hours by avoiding acute rejection, a potentially disastrous outcome that three of the pig’s 10 gene edits had been designed to prevent.
Although chronic immune rejection was always a concern despite Mr. Bennett’s novel immunosuppressant regimen, myocardial biopsy on postoperative days 34, 50, and 56 and necropsy showed “no signs of typical xenograft rejection,” Dr. Mohiuddin said at the ATC presentation. But “there’s a chance of atypical rejection which we were not accustomed to.”
By day 50, his diastolic function showed echocardiographic signs of deterioration, and “we started seeing interstitial edema with some extravasation of red blood cells, which we thought would resolve over a period of time,” he said. Eventually, however, “we saw that turn into fibroblasts and scar tissue.”
Mr. Bennett once again went on veno-arterial ECMO but died 10 days later. Once they had seen histologic evidence of fibrosis, Dr. Mohiuddin told this news organization, the team believed the myocardial injury was irreversible. “That was the reason we gave up on recovery.”
Mr. Bennett’s xenotransplantation journey has taught the field a lot, he said. “By no means was this a failure; we consider this a huge success. You can do all the experiments in animal models, but you won’t find out the true mechanism of rejection unless you do these kinds of human experiments.”
Looking ahead to clinical trials
Research involving humans is always subject to vagaries of human nature, including degree of adherence to prescribed therapy and – in xenotransplantation – precautions in place to mitigate any risks to public health. Such risks theoretically include transfer of porcine viruses or other pathogens to the patient and subsequent release into the general population.
Looking ahead to the possibility of clinical trials after this successful xenotransplantation experience, transplant nephrologist and epidemiologist Peter P. Reese, MD, PhD, University of Pennsylvania, Philadelphia, raised the potentially controversial issue in discussion following Dr. Mohiuddin’s presentation.
It’s known that Mr. Bennett had been repeatedly turned down for a conventional allograft transplant primarily because of his history of treatment noncompliance. Should such a record, Dr. Reese asked, be a relative contraindication to enrollment in any future xenotransplantation trials? Or does the field need a standardized gauge of a patient’s readiness, once discharged, to adhere not only to all medications – including those that fight infection – but also with rules established for public safety, such as routine contact reporting?
“It makes me wonder about choosing a noncompliant patient for these trials,” Dr. Reese said. “If we discharge a patient from the hospital who is at risk for a zoonotic infection that could spread if they basically refuse to cooperate with us or with public health authorities, it really could have negative consequences for the reputation of the field.”
Dr. Mohiuddin agreed such concerns are valid. Mr. Bennett “and all his immediate contacts” signed consent forms acknowledging their willingness to be followed should he be discharged. Mr. Bennett himself “signed a consent to inform us if he has any other intimate contact with someone,” he said in an interview.
“But those are only on paper.” Had Mr. Bennett survived to be discharged, Dr. Mohuiddin said, “no one knows how he would have behaved.”
Dr. Mohiuddin said the research staff had prepared to monitor Mr. Bennett at his home if that’s what it took. “We were ready to follow him as long as we could. There was a surveillance plan in place.”
A version of this article first appeared on Medscape.com.