User login
Out-of-pocket (OOP) costs for Medicare enrollees receiving quadruple drug therapy for heart failure with reduced ejection fraction were “substantially higher than regimens limited to generically available medications,” according to a new analysis of prescription drug plans.
“Despite the clinical benefit of quadruple therapy” consisting of beta-blockers, angiotensin receptor-neprilysin inhibitors (ARNIs), mineralocorticoid receptor antagonists (MRAs), and sodium-glucose cotransporter-2 (SGLT2) inhibitors, “coverage was restricted primarily through cost sharing, and estimated annual OOP costs for beneficiaries were [over $2,000] per year under most plans,” wrote Kamil F. Faridi, MD, and associates. The findings were published in the Journal of the American College of Cardiology.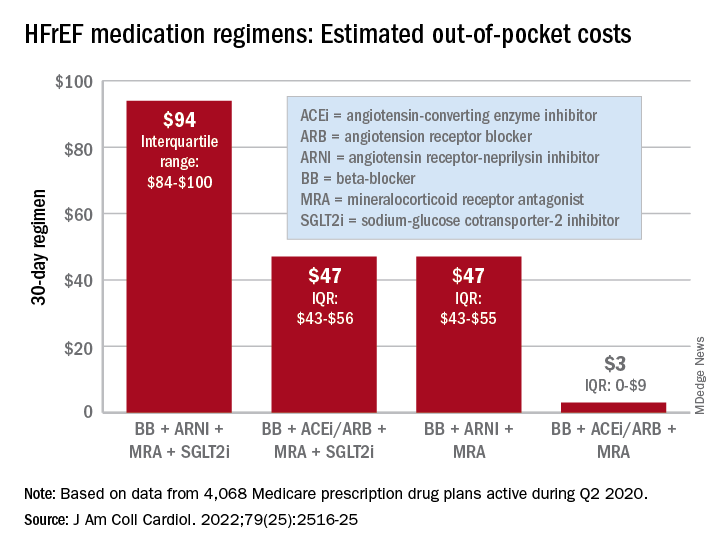
For just 1 month of quadruple drug therapy for heart failure with reduced ejection fraction (HFrEF), the estimated median OOP cost was $94 for individuals covered by a Medicare prescription drug plan during the second quarter of 2020, with the majority coming from the ARNI (median, $47) and the SGLT2 inhibitor (median, $45). Alternative HFrEF regimens were significantly less costly, ranging from $3 to $47 OOP, the investigators reported.
Almost all of the 4,068 plans participating in Medicare at that time covered quadruple therapy for HFrEF, but more than 99% restricted coverage by instituting cost sharing for medications at tier level 3 and above on the drug formularies. Such restrictions for ARNIs and SGLT2 inhibitors “might not be readily apparent to prescribing physicians,” wrote Dr. Faridi of Yale University, New Haven, Conn., and associates.
Other methods of regulating coverage were less common. Prior authorization of ARNIs was invoked by about a quarter of the plans, but none required authorization for any of the other drugs involved, and few plans used step therapy-requirements involving lower-cost alternatives, they noted.
“The use of cost sharing restricts access through high OOP costs for patients. Furthermore, these policies likely disadvantage relatively poorer patients (although the poorest Medicare patients will tend to be dual-enrolled in Medicaid and protected from cost sharing),” Jason H. Wasfy, MD, and Anna C. O’Kelly, MD, said in an accompanying editorial comment .
Since acceptable cost-effectiveness has been demonstrated for dapagliflozin, an SGLT1 inhibitor, and for the ARNIs, and because these medications have no generic equivalents, health plans should “use the discretion they have under Medicare Part D to reduce cost sharing for patients with HFrEF,” Dr. Wasfy and Dr. O’Kelly wrote, adding that the current study “demonstrates that without consensus on cost effectiveness from the societal perspective, costs can be imposed directly on patients in ways that slow uptake of cost-effective drugs.”
Data for all Medicare Advantage plans (n = 3,167) and standalone Part D plans (n = 901) came from the Medicare Prescription Drug Plan Formulary and Pricing Information Files. Annual OOP costs were estimated “using each phase of a 2020 Medicare part D standard benefit,” including deductible, standard coverage, coverage gap, and catastrophic coverage, the investigators explained.
Dr. Faridi and associates did not report any direct funding sources for their study. Dr Faridi received a grant from the National Institutes of Health outside the scope of the present work, and other investigators disclosed ties to the Food and Drug Administration, the Centers for Medicare and Medicaid Services, Johnson & Johnson, AstraZeneca, Boehringer Ingelheim, Amgen, Cytokinetics, and the Institute for Clinical and Economic Review.
Dr. Wasfy is supported by the American Heart Association and has received consulting fees from Pfizer and honoraria from the Institute for Clinical and Economic Review. Dr. O’Kelly has no relevant disclosures.
Out-of-pocket (OOP) costs for Medicare enrollees receiving quadruple drug therapy for heart failure with reduced ejection fraction were “substantially higher than regimens limited to generically available medications,” according to a new analysis of prescription drug plans.
“Despite the clinical benefit of quadruple therapy” consisting of beta-blockers, angiotensin receptor-neprilysin inhibitors (ARNIs), mineralocorticoid receptor antagonists (MRAs), and sodium-glucose cotransporter-2 (SGLT2) inhibitors, “coverage was restricted primarily through cost sharing, and estimated annual OOP costs for beneficiaries were [over $2,000] per year under most plans,” wrote Kamil F. Faridi, MD, and associates. The findings were published in the Journal of the American College of Cardiology.
For just 1 month of quadruple drug therapy for heart failure with reduced ejection fraction (HFrEF), the estimated median OOP cost was $94 for individuals covered by a Medicare prescription drug plan during the second quarter of 2020, with the majority coming from the ARNI (median, $47) and the SGLT2 inhibitor (median, $45). Alternative HFrEF regimens were significantly less costly, ranging from $3 to $47 OOP, the investigators reported.
Almost all of the 4,068 plans participating in Medicare at that time covered quadruple therapy for HFrEF, but more than 99% restricted coverage by instituting cost sharing for medications at tier level 3 and above on the drug formularies. Such restrictions for ARNIs and SGLT2 inhibitors “might not be readily apparent to prescribing physicians,” wrote Dr. Faridi of Yale University, New Haven, Conn., and associates.
Other methods of regulating coverage were less common. Prior authorization of ARNIs was invoked by about a quarter of the plans, but none required authorization for any of the other drugs involved, and few plans used step therapy-requirements involving lower-cost alternatives, they noted.
“The use of cost sharing restricts access through high OOP costs for patients. Furthermore, these policies likely disadvantage relatively poorer patients (although the poorest Medicare patients will tend to be dual-enrolled in Medicaid and protected from cost sharing),” Jason H. Wasfy, MD, and Anna C. O’Kelly, MD, said in an accompanying editorial comment .
Since acceptable cost-effectiveness has been demonstrated for dapagliflozin, an SGLT1 inhibitor, and for the ARNIs, and because these medications have no generic equivalents, health plans should “use the discretion they have under Medicare Part D to reduce cost sharing for patients with HFrEF,” Dr. Wasfy and Dr. O’Kelly wrote, adding that the current study “demonstrates that without consensus on cost effectiveness from the societal perspective, costs can be imposed directly on patients in ways that slow uptake of cost-effective drugs.”
Data for all Medicare Advantage plans (n = 3,167) and standalone Part D plans (n = 901) came from the Medicare Prescription Drug Plan Formulary and Pricing Information Files. Annual OOP costs were estimated “using each phase of a 2020 Medicare part D standard benefit,” including deductible, standard coverage, coverage gap, and catastrophic coverage, the investigators explained.
Dr. Faridi and associates did not report any direct funding sources for their study. Dr Faridi received a grant from the National Institutes of Health outside the scope of the present work, and other investigators disclosed ties to the Food and Drug Administration, the Centers for Medicare and Medicaid Services, Johnson & Johnson, AstraZeneca, Boehringer Ingelheim, Amgen, Cytokinetics, and the Institute for Clinical and Economic Review.
Dr. Wasfy is supported by the American Heart Association and has received consulting fees from Pfizer and honoraria from the Institute for Clinical and Economic Review. Dr. O’Kelly has no relevant disclosures.
Out-of-pocket (OOP) costs for Medicare enrollees receiving quadruple drug therapy for heart failure with reduced ejection fraction were “substantially higher than regimens limited to generically available medications,” according to a new analysis of prescription drug plans.
“Despite the clinical benefit of quadruple therapy” consisting of beta-blockers, angiotensin receptor-neprilysin inhibitors (ARNIs), mineralocorticoid receptor antagonists (MRAs), and sodium-glucose cotransporter-2 (SGLT2) inhibitors, “coverage was restricted primarily through cost sharing, and estimated annual OOP costs for beneficiaries were [over $2,000] per year under most plans,” wrote Kamil F. Faridi, MD, and associates. The findings were published in the Journal of the American College of Cardiology.
For just 1 month of quadruple drug therapy for heart failure with reduced ejection fraction (HFrEF), the estimated median OOP cost was $94 for individuals covered by a Medicare prescription drug plan during the second quarter of 2020, with the majority coming from the ARNI (median, $47) and the SGLT2 inhibitor (median, $45). Alternative HFrEF regimens were significantly less costly, ranging from $3 to $47 OOP, the investigators reported.
Almost all of the 4,068 plans participating in Medicare at that time covered quadruple therapy for HFrEF, but more than 99% restricted coverage by instituting cost sharing for medications at tier level 3 and above on the drug formularies. Such restrictions for ARNIs and SGLT2 inhibitors “might not be readily apparent to prescribing physicians,” wrote Dr. Faridi of Yale University, New Haven, Conn., and associates.
Other methods of regulating coverage were less common. Prior authorization of ARNIs was invoked by about a quarter of the plans, but none required authorization for any of the other drugs involved, and few plans used step therapy-requirements involving lower-cost alternatives, they noted.
“The use of cost sharing restricts access through high OOP costs for patients. Furthermore, these policies likely disadvantage relatively poorer patients (although the poorest Medicare patients will tend to be dual-enrolled in Medicaid and protected from cost sharing),” Jason H. Wasfy, MD, and Anna C. O’Kelly, MD, said in an accompanying editorial comment .
Since acceptable cost-effectiveness has been demonstrated for dapagliflozin, an SGLT1 inhibitor, and for the ARNIs, and because these medications have no generic equivalents, health plans should “use the discretion they have under Medicare Part D to reduce cost sharing for patients with HFrEF,” Dr. Wasfy and Dr. O’Kelly wrote, adding that the current study “demonstrates that without consensus on cost effectiveness from the societal perspective, costs can be imposed directly on patients in ways that slow uptake of cost-effective drugs.”
Data for all Medicare Advantage plans (n = 3,167) and standalone Part D plans (n = 901) came from the Medicare Prescription Drug Plan Formulary and Pricing Information Files. Annual OOP costs were estimated “using each phase of a 2020 Medicare part D standard benefit,” including deductible, standard coverage, coverage gap, and catastrophic coverage, the investigators explained.
Dr. Faridi and associates did not report any direct funding sources for their study. Dr Faridi received a grant from the National Institutes of Health outside the scope of the present work, and other investigators disclosed ties to the Food and Drug Administration, the Centers for Medicare and Medicaid Services, Johnson & Johnson, AstraZeneca, Boehringer Ingelheim, Amgen, Cytokinetics, and the Institute for Clinical and Economic Review.
Dr. Wasfy is supported by the American Heart Association and has received consulting fees from Pfizer and honoraria from the Institute for Clinical and Economic Review. Dr. O’Kelly has no relevant disclosures.
FROM THE JOURNAL Of the AMERICAN COLLEGE OF CARDIOLOGY