User login
Examining Interventions and Adverse Events After Nonfatal Opioid Overdoses in Veterans
The number of opioid-related overdose deaths in the United States is estimated to have increased 6-fold over the past 2 decades.1 In 2017, more than two-thirds of drug overdose deaths involved opioids, yielding a mortality rate of 14.9 per 100,000.2 Not only does the opioid epidemic currently pose a significant public health crisis characterized by high morbidity and mortality, but it is also projected to worsen in coming years. According to Chen and colleagues, opioid overdose deaths are estimated to increase by 147% from 2015 to 2025.3 That projects almost 82,000 US deaths annually and > 700,000 deaths in this period—even before accounting for surges in opioid overdoses and opioid-related mortality coinciding with the COVID-19 pandemic.3,4
As health systems and communities globally struggle with unprecedented losses and stressors introduced by the pandemic, emerging data warrants escalating concerns with regard to increased vulnerability to relapse and overdose among those with mental health and substance use disorders (SUDs). In a recent report, the American Medical Association estimates that opioid-related deaths have increased in more than 40 states with the COVID-19 pandemic.4
Veterans are twice as likely to experience a fatal opioid overdose compared with their civilian counterparts.5 While several risk mitigation strategies have been employed in recent years to improve opioid prescribing and safety within the US Department of Veterans Affairs (VA), veterans continue to overdose on opioids, both prescribed and obtained illicitly.6 Variables shown to be strongly associated with opioid overdose risk include presence of mental health disorders, SUDs, medical conditions involving impaired drug metabolism or excretion, respiratory disorders, higher doses of opioids, concomitant use of sedative medications, and history of overdose.6-8 Many veterans struggle with chronic pain and those prescribed high doses of opioids were more likely to have comorbid pain diagnoses, mental health disorders, and SUDs.9 Dashboards and predictive models, such as the Stratification Tool for Opioid Risk Mitigation (STORM) and the Risk Index for Overdose or Serious Opioid-induced Respiratory Depression (RIOSORD), incorporate such factors to stratify overdose risk among veterans, in an effort to prioritize high-risk individuals for review and provision of care.6,10,11 Despite recent recognition that overdose prevention likely requires a holistic approach that addresses the biopsychosocial factors contributing to opioid-related morbidity and mortality, it is unclear whether veterans are receiving adequate and appropriate treatment for contributing conditions.
There are currently no existing studies that describe health service utilization (HSU), medication interventions, and rates of opioid-related adverse events (ORAEs) among veterans after survival of a nonfatal opioid overdose (NFO). Clinical characteristics of veterans treated for opioid overdose at a VA emergency department (ED) have previously been described by Clement and Stock.12 Despite improvements that have been made in VA opioid prescribing and safety, knowledge gaps remain with regard to best practices for opioid overdose prevention. The aim of this study was to characterize HSU and medication interventions in veterans following NFO, as well as the frequency of ORAEs after overdose. The findings of this study may aid in the identification of areas for targeted improvement in the prevention and reduction of opioid overdoses and adverse opioid-related sequelae.
Methods
This retrospective descriptive study was conducted at VA San Diego Healthcare System (VASDHCS) in California. Subjects included were veterans administered naloxone in the ED for suspected opioid overdose between July 1, 2013 and April 1, 2017. The study population was identified through data retrieved from automated drug dispensing systems, which was then confirmed through manual chart review of notes associated with the index ED visit. Inclusion criteria included documented increased respiration or responsiveness following naloxone administration. Subjects were excluded if they demonstrated lack of response to naloxone, overdosed secondary to inpatient administration of opioids, received palliative or hospice care during the study period, or were lost to follow-up.
Data were collected via retrospective chart review and included date of index ED visit, demographics, active prescriptions, urine drug screen (UDS) results, benzodiazepine (BZD) use corroborated by positive UDS or mention of BZD in index visit chart notes, whether overdose was determined to be a suicide attempt, and naloxone kit dispensing. Patient data was collected for 2 years following overdose, including: ORAEs; ED visits; hospitalizations; repeat overdoses; fatal overdose; whether subjects were still alive; follow-up visits for pain management, mental health, and addiction treatment services; and visits to the psychiatric emergency clinic. Clinical characteristics, such as mental health disorder diagnoses, SUDs, and relevant medical conditions also were collected. Statistical analysis was performed using Microsoft Excel and included only descriptive statistics.
Results
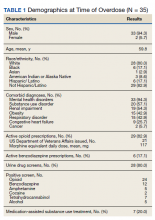
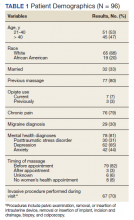
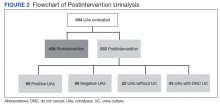
Ninety-three patients received naloxone in the VASDHCS ED. Thirty-five met inclusion criteria and were included in the primary analysis. All subjects received IV naloxone with a mean 0.8 mg IV boluses (range, 0.1-4.4 mg).
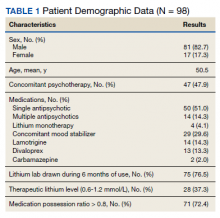
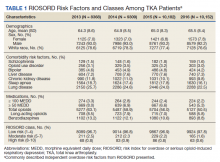
Most patients were male with a mean age of 59.8 years (Table 1). Almost all overdoses were nonintentional except for 3 suicide attempts that were reviewed by the Suicide Prevention Committee. Three patients had previously been treated for opioid overdose at the VA with a documented positive clinical response to naloxone administration.
At the time of overdose, 29 patients (82.9%) had an active opioid prescription. Of these, the majority were issued through the VA with a mean 117 mg morphine equivalent daily dose (MEDD). Interestingly, only 24 of the 28 patients with a UDS collected at time of overdose tested positive for opioids, which may be attributable to the use of synthetic opioids, which are not reliably detected by traditional UDS. Concomitant BZD use was involved in 13 of the 35 index overdoses (37.1%), although only 6 patients (17.1%) had an active BZD prescription at time of overdose. Seven patients (20.0%) were prescribed medication-assisted treatment (MAT) for opioid use disorder (OUD), with all 7 using methadone. According to VA records, only 1 patient had previously been dispensed a naloxone kit at any point prior to overdosing. Mental health and SUD diagnoses frequently co-occurred, with 20 patients (57.1%) having at least 1 mental health condition and at least 1 SUD.
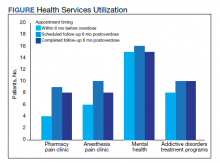
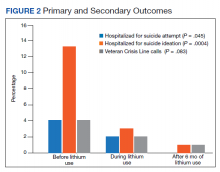
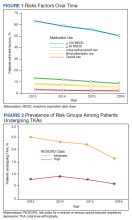
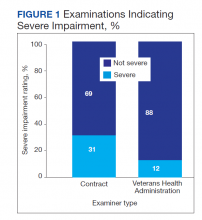
Rates of follow-up varied by clinician type in the 6 months after NFO (Figure). Of those with mental health disorders, 15 patients (45.5%) received mental health services before and after overdose, while 8 (40.0%) and 10 (50.0%) of those with SUDs received addiction treatment services before and after overdose, respectively. Seven patients presented to the psychiatric emergency clinic within 6 months prior to overdose and 5 patients within the 6 months following overdose.
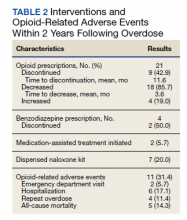
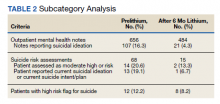
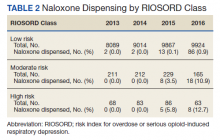
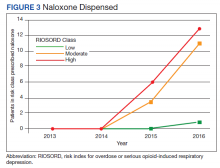
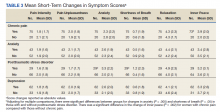
Of patients with VA opioid prescriptions, within 2 years of NFO, 9 (42.9%) had their opioids discontinued, and 18 (85.7%) had MEDD reductions ranging from 10 mg to 150 mg (12.5-71.4% reduction) with a mean of 63 mg. Two of the 4 patients with active BZD prescriptions at the time of the overdose event had their prescriptions continued. Seven patients (20.0%) were dispensed naloxone kits following overdose (Table 2).
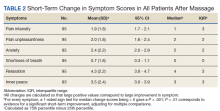
Rates of ORAEs ranged from 0% to 17% with no documented overdose fatalities. Examples of AEs observed in this study included ED visits or hospitalizations involving opioid withdrawal, opioid-related personality changes, and opioid overdose. Five patients died during the study period, yielding an all-cause mortality rate of 14.3% with a mean time to death of 10.8 months. The causes of death were largely unknown except for 1 patient, whose death was reportedly investigated as an accidental medication overdose without additional information.
Repeat overdose verified by hospital records occurred in 4 patients (11.4%) within 2 years. Patients who experienced a subsequent overdose were prescribed higher doses of opioids with a mean MEDD among VA prescriptions of 130 mg vs 114 mg for those without repeat overdose. In this group, 3 patients (75.0%) also had concomitant BZD use, which was proportionally higher than the 10 patients (32.3%) without a subsequent overdose. Of note, 2 of the 4 patients with a repeat overdose had their opioid doses increased above the MEDD prescribed at the time of index overdose. None of the 4 subjects who experienced a repeat overdose were initiated on MAT within 2 years according to VA records.
Discussions
This retrospective study is representative of many veterans receiving VA care, despite the small sample size. Clinical characteristics observed in the study population were generally consistent with those published by Clement and Stock, including high rates of medical and psychiatric comorbidities.12 Subjects in both studies were prescribed comparable dosages of opioids; among those prescribed opioids but not BZDs through the VA, the mean MEDD was 117 mg in our study compared with 126 mg in the Clement and Stock study. Since implementation of the Opioid Safety Initiative (OSI) in 2013, opioid prescribing practices have improved nationwide across VA facilities, including successful reduction in the numbers of patients prescribed high-dose opioids and concurrent BZDs.13
Despite the tools and resources available to clinicians, discontinuing opioid therapy remains a difficult process. Concerns related to mental health and/or substance-use related decompensations often exist in the setting of rapid dose reductions or abrupt discontinuation of opioids.6 Although less than half of patients in the present study with an active opioid prescription at time of index overdose had their opioids discontinued within 2 years, it is reassuring to note the much higher rate of those with subsequent decreases in their prescribed doses, as well as the 50% reduction in BZD coprescribing. Ultimately, these findings remain consistent with the VA goals of mitigating harm, improving opioid prescribing, and ensuring the safe use of opioid medications when clinically appropriate.
Moreover, recent evidence suggests that interventions focused solely on opioid prescribing practices are becoming increasingly limited in their impact on reducing opioid-related deaths and will likely be insufficient for addressing the opioid epidemic as it continues to evolve. According to Chen and colleagues, opioid overdose deaths are projected to increase over the next several years, while further reduction in the incidence of prescription opioid misuse is estimated to decrease overdose deaths by only 3% to 5.3%. In the context of recent surges in synthetic opioid use, it is projected that 80% of overdose deaths between 2016 and 2025 will be attributable to illicit opioids.3 Such predictions underscore the urgent need to adopt alternative approaches to risk-reducing measures and policy change.
The increased risk of mortality associated with opioid misuse and overdose is well established in the current literature. However, less is known regarding the rate of ORAEs after survival of an NFO. Olfson and colleagues sought to address this knowledge gap by characterizing mortality risks in 76,325 US adults within 1 year following NFO.14 Among their studied population, all-cause mortality occurred at a rate of 778.3 per 10,000 person-years, which was 24 times greater than that of the general population. This emphasizes the need for the optimization of mental health services, addiction treatment, and medical care for these individuals at higher risk.
Limitations
Certain factors and limitations should be considered when interpreting the results of this study. Given that the study included only veterans, factors such as the demographic and clinical characteristics more commonly observed among these patients should be taken into account and may in turn limit the generalizability of these findings to nonveteran populations. Another major limitation is the small sample size; the study period and by extension, the number of patients able to be included in the present study were restricted by the availability of retrievable data from automated drug dispensing systems. Patients without documented response to naloxone were excluded from the study due to low clinical suspicion for opioid overdose, although the possibility that the dose administered was too low to produce a robust clinical response cannot be definitively ruled out. The lack of reliable methods to capture events and overdoses treated outside of the VA may have resulted in underestimations of the true occurrence of ORAEs following NFO. Information regarding naloxone administration outside VA facilities, such as in transport to the hospital, self-reported, or bystander administration, was similarly limited by lack of reliable methods for retrieving such data and absence of documentation in VA records. Although all interventions and outcomes reported in the present study occurred within 2 years following NFO, further conclusions pertaining to the relative timing of specific interventions and ORAEs cannot be made. Lastly, this study did not investigate the direct impact of opioid risk mitigation initiatives implemented by the VA in the years coinciding with the study period.
Future Directions
Despite these limitations, an important strength of this study is its ability to identify potential areas for targeted improvement and to guide further efforts relating to the prevention of opioid overdose and opioid-related mortality among veterans. Identification of individuals at high risk for opioid overdose and misuse is an imperative first step that allows for the implementation of downstream risk-mitigating interventions. Within the VA, several tools have been developed in recent years to provide clinicians with additional resources and support in this regard.6,15
No more than half of those diagnosed with mental health disorders and SUDs in the present study received outpatient follow-up care for these conditions within 6 months following NFO, which may suggest high rates of inadequate treatment. Given the strong association between mental health disorders, SUDs, and increased risk of overdose, increasing engagement with mental health and addiction treatment services may be paramount to preventing subsequent ORAEs, including repeat overdose.6-9,11
Naloxone kit dispensing represents another area for targeted improvement. Interventions may include clinician education and systematic changes, such as implementing protocols that boost the likelihood of high-risk individuals being provided with naloxone at the earliest opportunity. Bystander-administered naloxone programs can also be considered for increasing naloxone access and reducing opioid-related mortality.16
Finally, despite evidence supporting the benefit of MAT in OUD treatment and reducing all-cause and opioid-related mortality after NFO, the low rates of MAT observed in this study are consistent with previous reports that these medications remain underutilized.17 Screening for OUD, in conjunction with increasing access to and utilization of OUD treatment modalities, is an established and integral component of overdose prevention efforts. For VA clinicians, the Psychotropic Drug Safety Initiative (PDSI) dashboard can be used to identify patients diagnosed with OUD who are not yet on MAT.18 Initiatives to expand MAT access through the ED have the potential to provide life-saving interventions and bridge care in the interim until patients are able to become established with a long-term health care practitioner.19
Conclusions
This is the first study to describe HSU, medication interventions, and ORAEs among veterans who survive NFO. Studies have shown that veterans with a history of NFO are at increased risk of subsequent AEs and premature death.6,7,10,14 As such, NFOs represent crucial opportunities to identify high-risk individuals and ensure provision of adequate care. Recent data supports the development of a holistic, multimodal approach focused on adequate treatment of conditions that contribute to opioid-related risks, including mental health disorders, SUDs, pain diagnoses, and medical comorbidities.3,14 Interventions designed to improve access, engagement, and retention in such care therefore play a pivotal role in overdose prevention and reducing mortality.
Although existing risk mitigation initiatives have improved opioid prescribing and safety within the VA, the findings of this study suggest that there remains room for improvement, and the need for well-coordinated efforts to reduce risks associated with both prescribed and illicit opioid use cannot be overstated. Rates of overdose deaths not only remain high but are projected to continue increasing in coming years, despite advances in clinical practice aimed at reducing harms associated with opioid use. The present findings aim to help identify processes with the potential to reduce rates of overdose, death, and adverse sequelae in high-risk populations. However, future studies are warranted to expand on these findings and contribute to ongoing efforts in reducing opioid-related harms and overdose deaths. This study may provide critical insight to inform further investigations to guide such interventions and highlight tools that health care facilities even outside the VA can consider implementing.
Acknowledgments
The authors would like to thank Jonathan Lacro, PharmD, BCPP, for his guidance with this important clinical topic and navigating IRB submissions.
1. Centers for Disease Control and Prevention. Data overview: the drug overdose epidemic: behind the numbers. Updated March 25, 2021. Accessed February 9, 2022. www.cdc.gov/drugoverdose/data/index.html
2. Scholl L, Seth P, Kariisa M, Wilson N, Baldwin G. Drug and Opioid-Involved Overdose Deaths - United States, 2013-2017. MMWR Morb Mortal Wkly Rep. 2018;67(5152):1419-1427. Published 2018 Jan 4. doi:10.15585/mmwr.mm675152e1 3. Chen Q, Larochelle MR, Weaver DT, et al. Prevention of prescription opioid misuse and projected overdose deaths in the United States. JAMA Netw Open. 2019;2(2):e187621. Published 2019 Feb 1. doi:10.1001/jamanetworkopen.2018.7621
4. American Medical Association. Issue brief: nation’s drug-related overdose and death epidemic continues to worsen. Updated November 12, 2021. Accessed February 11, 2022. https://www.ama-assn.org/system/files/issue-brief-increases-in-opioid-related-overdose.pdf
5. Bohnert AS, Ilgen MA, Galea S, McCarthy JF, Blow FC. Accidental poisoning mortality among patients in the Department of Veterans Affairs Health System. Med Care. 2011;49(4):393-396. doi:10.1097/MLR.0b013e318202aa27
6. Lewis ET, Trafton J, Oliva E. Data-based case reviews of patients with opioid related risk factors as a tool to prevent overdose and suicide. Accessed February 9, 2022. www.hsrd.research.va.gov/for_researchers/cyber_seminars/archives/2488-notes.pdf
7. Zedler B, Xie L, Wang L, et al. Risk factors for serious prescription opioid-related toxicity or overdose among Veterans Health Administration patients. Pain Med. 2014;15(11):1911-1929. doi:10.1111/pme.12480
8. Webster LR. Risk Factors for Opioid-Use Disorder and Overdose. Anesth Analg. 2017;125(5):1741-1748. doi:10.1213/ANE.0000000000002496
9. Morasco BJ, Duckart JP, Carr TP, Deyo RA, Dobscha SK. Clinical characteristics of veterans prescribed high doses of opioid medications for chronic non-cancer pain. Pain. 2010;151(3):625-632. doi:10.1016/j.pain.2010.08.002
10. Oliva EM, Bowe T, Tavakoli S, et al. Development and applications of the Veterans Health Administration’s Stratification Tool for Opioid Risk Mitigation (STORM) to improve opioid safety and prevent overdose and suicide. Psychol Serv. 2017;14(1):34-49. doi:10.1037/ser0000099
11. Zedler B, Xie L, Wang L, et al. Development of a risk index for serious prescription opioid-induced respiratory depression or overdose in Veterans’ Health Administration patients. Pain Med. 2015;16(8):1566-1579. doi:10.1111/pme.12777
12. Clement C, Stock C. Who Overdoses at a VA Emergency Department? Fed Pract. 2016;33(11):14-20.
13. Lin LA, Bohnert ASB, Kerns RD, Clay MA, Ganoczy D, Ilgen MA. Impact of the Opioid Safety Initiative on opioid-related prescribing in veterans. Pain. 2017;158(5):833-839. doi:10.1097/j.pain.0000000000000837
14. Olfson M, Crystal S, Wall M, Wang S, Liu SM, Blanco C. Causes of death after nonfatal opioid overdose [published correction appears in JAMA Psychiatry. 2018 Aug 1;75(8):867]. JAMA Psychiatry. 2018;75(8):820-827. doi:10.1001/jamapsychiatry.2018.1471
15. US Department of Veterans Affairs, Veterans Health Administration. VHA pain management – opioid safety – clinical tools. Updated November 14, 2019. Accessed February 9, 2022. https://www.va.gov/PAINMANAGEMENT/Opioid_Safety/Clinical_Tools.asp
16. Doe-Simkins M, Walley AY, Epstein A, Moyer P. Saved by the nose: bystander-administered intranasal naloxone hydrochloride for opioid overdose. Am J Public Health. 2009;99(5):788-791. doi:10.2105/AJPH.2008.146647
17. Larochelle MR, Bernson D, Land T, et al. Medication for opioid use disorder after nonfatal opioid overdose and association with mortality: a cohort study. Ann Intern Med. 2018;169(3):137-145. doi:10.7326/M17-3107
18. Wiechers I. Program focuses on safe psychiatric medication. Published April 21, 2016. Accessed February 9, 2022. https://blogs.va.gov/VAntage/27099/program-focuses-safe-psychiatric-medication/
19. Newman S; California Health Care Foundation. How to pay for it – MAT in the emergency department: FAQ. Published March 2019. Accessed February 9, 2022. https://www.chcf.org/wp-content/uploads/2019/03/HowToPayForMATinED.pdf
The number of opioid-related overdose deaths in the United States is estimated to have increased 6-fold over the past 2 decades.1 In 2017, more than two-thirds of drug overdose deaths involved opioids, yielding a mortality rate of 14.9 per 100,000.2 Not only does the opioid epidemic currently pose a significant public health crisis characterized by high morbidity and mortality, but it is also projected to worsen in coming years. According to Chen and colleagues, opioid overdose deaths are estimated to increase by 147% from 2015 to 2025.3 That projects almost 82,000 US deaths annually and > 700,000 deaths in this period—even before accounting for surges in opioid overdoses and opioid-related mortality coinciding with the COVID-19 pandemic.3,4
As health systems and communities globally struggle with unprecedented losses and stressors introduced by the pandemic, emerging data warrants escalating concerns with regard to increased vulnerability to relapse and overdose among those with mental health and substance use disorders (SUDs). In a recent report, the American Medical Association estimates that opioid-related deaths have increased in more than 40 states with the COVID-19 pandemic.4
Veterans are twice as likely to experience a fatal opioid overdose compared with their civilian counterparts.5 While several risk mitigation strategies have been employed in recent years to improve opioid prescribing and safety within the US Department of Veterans Affairs (VA), veterans continue to overdose on opioids, both prescribed and obtained illicitly.6 Variables shown to be strongly associated with opioid overdose risk include presence of mental health disorders, SUDs, medical conditions involving impaired drug metabolism or excretion, respiratory disorders, higher doses of opioids, concomitant use of sedative medications, and history of overdose.6-8 Many veterans struggle with chronic pain and those prescribed high doses of opioids were more likely to have comorbid pain diagnoses, mental health disorders, and SUDs.9 Dashboards and predictive models, such as the Stratification Tool for Opioid Risk Mitigation (STORM) and the Risk Index for Overdose or Serious Opioid-induced Respiratory Depression (RIOSORD), incorporate such factors to stratify overdose risk among veterans, in an effort to prioritize high-risk individuals for review and provision of care.6,10,11 Despite recent recognition that overdose prevention likely requires a holistic approach that addresses the biopsychosocial factors contributing to opioid-related morbidity and mortality, it is unclear whether veterans are receiving adequate and appropriate treatment for contributing conditions.
There are currently no existing studies that describe health service utilization (HSU), medication interventions, and rates of opioid-related adverse events (ORAEs) among veterans after survival of a nonfatal opioid overdose (NFO). Clinical characteristics of veterans treated for opioid overdose at a VA emergency department (ED) have previously been described by Clement and Stock.12 Despite improvements that have been made in VA opioid prescribing and safety, knowledge gaps remain with regard to best practices for opioid overdose prevention. The aim of this study was to characterize HSU and medication interventions in veterans following NFO, as well as the frequency of ORAEs after overdose. The findings of this study may aid in the identification of areas for targeted improvement in the prevention and reduction of opioid overdoses and adverse opioid-related sequelae.
Methods
This retrospective descriptive study was conducted at VA San Diego Healthcare System (VASDHCS) in California. Subjects included were veterans administered naloxone in the ED for suspected opioid overdose between July 1, 2013 and April 1, 2017. The study population was identified through data retrieved from automated drug dispensing systems, which was then confirmed through manual chart review of notes associated with the index ED visit. Inclusion criteria included documented increased respiration or responsiveness following naloxone administration. Subjects were excluded if they demonstrated lack of response to naloxone, overdosed secondary to inpatient administration of opioids, received palliative or hospice care during the study period, or were lost to follow-up.
Data were collected via retrospective chart review and included date of index ED visit, demographics, active prescriptions, urine drug screen (UDS) results, benzodiazepine (BZD) use corroborated by positive UDS or mention of BZD in index visit chart notes, whether overdose was determined to be a suicide attempt, and naloxone kit dispensing. Patient data was collected for 2 years following overdose, including: ORAEs; ED visits; hospitalizations; repeat overdoses; fatal overdose; whether subjects were still alive; follow-up visits for pain management, mental health, and addiction treatment services; and visits to the psychiatric emergency clinic. Clinical characteristics, such as mental health disorder diagnoses, SUDs, and relevant medical conditions also were collected. Statistical analysis was performed using Microsoft Excel and included only descriptive statistics.
Results
Ninety-three patients received naloxone in the VASDHCS ED. Thirty-five met inclusion criteria and were included in the primary analysis. All subjects received IV naloxone with a mean 0.8 mg IV boluses (range, 0.1-4.4 mg).
Most patients were male with a mean age of 59.8 years (Table 1). Almost all overdoses were nonintentional except for 3 suicide attempts that were reviewed by the Suicide Prevention Committee. Three patients had previously been treated for opioid overdose at the VA with a documented positive clinical response to naloxone administration.
At the time of overdose, 29 patients (82.9%) had an active opioid prescription. Of these, the majority were issued through the VA with a mean 117 mg morphine equivalent daily dose (MEDD). Interestingly, only 24 of the 28 patients with a UDS collected at time of overdose tested positive for opioids, which may be attributable to the use of synthetic opioids, which are not reliably detected by traditional UDS. Concomitant BZD use was involved in 13 of the 35 index overdoses (37.1%), although only 6 patients (17.1%) had an active BZD prescription at time of overdose. Seven patients (20.0%) were prescribed medication-assisted treatment (MAT) for opioid use disorder (OUD), with all 7 using methadone. According to VA records, only 1 patient had previously been dispensed a naloxone kit at any point prior to overdosing. Mental health and SUD diagnoses frequently co-occurred, with 20 patients (57.1%) having at least 1 mental health condition and at least 1 SUD.
Rates of follow-up varied by clinician type in the 6 months after NFO (Figure). Of those with mental health disorders, 15 patients (45.5%) received mental health services before and after overdose, while 8 (40.0%) and 10 (50.0%) of those with SUDs received addiction treatment services before and after overdose, respectively. Seven patients presented to the psychiatric emergency clinic within 6 months prior to overdose and 5 patients within the 6 months following overdose.
Of patients with VA opioid prescriptions, within 2 years of NFO, 9 (42.9%) had their opioids discontinued, and 18 (85.7%) had MEDD reductions ranging from 10 mg to 150 mg (12.5-71.4% reduction) with a mean of 63 mg. Two of the 4 patients with active BZD prescriptions at the time of the overdose event had their prescriptions continued. Seven patients (20.0%) were dispensed naloxone kits following overdose (Table 2).
Rates of ORAEs ranged from 0% to 17% with no documented overdose fatalities. Examples of AEs observed in this study included ED visits or hospitalizations involving opioid withdrawal, opioid-related personality changes, and opioid overdose. Five patients died during the study period, yielding an all-cause mortality rate of 14.3% with a mean time to death of 10.8 months. The causes of death were largely unknown except for 1 patient, whose death was reportedly investigated as an accidental medication overdose without additional information.
Repeat overdose verified by hospital records occurred in 4 patients (11.4%) within 2 years. Patients who experienced a subsequent overdose were prescribed higher doses of opioids with a mean MEDD among VA prescriptions of 130 mg vs 114 mg for those without repeat overdose. In this group, 3 patients (75.0%) also had concomitant BZD use, which was proportionally higher than the 10 patients (32.3%) without a subsequent overdose. Of note, 2 of the 4 patients with a repeat overdose had their opioid doses increased above the MEDD prescribed at the time of index overdose. None of the 4 subjects who experienced a repeat overdose were initiated on MAT within 2 years according to VA records.
Discussions
This retrospective study is representative of many veterans receiving VA care, despite the small sample size. Clinical characteristics observed in the study population were generally consistent with those published by Clement and Stock, including high rates of medical and psychiatric comorbidities.12 Subjects in both studies were prescribed comparable dosages of opioids; among those prescribed opioids but not BZDs through the VA, the mean MEDD was 117 mg in our study compared with 126 mg in the Clement and Stock study. Since implementation of the Opioid Safety Initiative (OSI) in 2013, opioid prescribing practices have improved nationwide across VA facilities, including successful reduction in the numbers of patients prescribed high-dose opioids and concurrent BZDs.13
Despite the tools and resources available to clinicians, discontinuing opioid therapy remains a difficult process. Concerns related to mental health and/or substance-use related decompensations often exist in the setting of rapid dose reductions or abrupt discontinuation of opioids.6 Although less than half of patients in the present study with an active opioid prescription at time of index overdose had their opioids discontinued within 2 years, it is reassuring to note the much higher rate of those with subsequent decreases in their prescribed doses, as well as the 50% reduction in BZD coprescribing. Ultimately, these findings remain consistent with the VA goals of mitigating harm, improving opioid prescribing, and ensuring the safe use of opioid medications when clinically appropriate.
Moreover, recent evidence suggests that interventions focused solely on opioid prescribing practices are becoming increasingly limited in their impact on reducing opioid-related deaths and will likely be insufficient for addressing the opioid epidemic as it continues to evolve. According to Chen and colleagues, opioid overdose deaths are projected to increase over the next several years, while further reduction in the incidence of prescription opioid misuse is estimated to decrease overdose deaths by only 3% to 5.3%. In the context of recent surges in synthetic opioid use, it is projected that 80% of overdose deaths between 2016 and 2025 will be attributable to illicit opioids.3 Such predictions underscore the urgent need to adopt alternative approaches to risk-reducing measures and policy change.
The increased risk of mortality associated with opioid misuse and overdose is well established in the current literature. However, less is known regarding the rate of ORAEs after survival of an NFO. Olfson and colleagues sought to address this knowledge gap by characterizing mortality risks in 76,325 US adults within 1 year following NFO.14 Among their studied population, all-cause mortality occurred at a rate of 778.3 per 10,000 person-years, which was 24 times greater than that of the general population. This emphasizes the need for the optimization of mental health services, addiction treatment, and medical care for these individuals at higher risk.
Limitations
Certain factors and limitations should be considered when interpreting the results of this study. Given that the study included only veterans, factors such as the demographic and clinical characteristics more commonly observed among these patients should be taken into account and may in turn limit the generalizability of these findings to nonveteran populations. Another major limitation is the small sample size; the study period and by extension, the number of patients able to be included in the present study were restricted by the availability of retrievable data from automated drug dispensing systems. Patients without documented response to naloxone were excluded from the study due to low clinical suspicion for opioid overdose, although the possibility that the dose administered was too low to produce a robust clinical response cannot be definitively ruled out. The lack of reliable methods to capture events and overdoses treated outside of the VA may have resulted in underestimations of the true occurrence of ORAEs following NFO. Information regarding naloxone administration outside VA facilities, such as in transport to the hospital, self-reported, or bystander administration, was similarly limited by lack of reliable methods for retrieving such data and absence of documentation in VA records. Although all interventions and outcomes reported in the present study occurred within 2 years following NFO, further conclusions pertaining to the relative timing of specific interventions and ORAEs cannot be made. Lastly, this study did not investigate the direct impact of opioid risk mitigation initiatives implemented by the VA in the years coinciding with the study period.
Future Directions
Despite these limitations, an important strength of this study is its ability to identify potential areas for targeted improvement and to guide further efforts relating to the prevention of opioid overdose and opioid-related mortality among veterans. Identification of individuals at high risk for opioid overdose and misuse is an imperative first step that allows for the implementation of downstream risk-mitigating interventions. Within the VA, several tools have been developed in recent years to provide clinicians with additional resources and support in this regard.6,15
No more than half of those diagnosed with mental health disorders and SUDs in the present study received outpatient follow-up care for these conditions within 6 months following NFO, which may suggest high rates of inadequate treatment. Given the strong association between mental health disorders, SUDs, and increased risk of overdose, increasing engagement with mental health and addiction treatment services may be paramount to preventing subsequent ORAEs, including repeat overdose.6-9,11
Naloxone kit dispensing represents another area for targeted improvement. Interventions may include clinician education and systematic changes, such as implementing protocols that boost the likelihood of high-risk individuals being provided with naloxone at the earliest opportunity. Bystander-administered naloxone programs can also be considered for increasing naloxone access and reducing opioid-related mortality.16
Finally, despite evidence supporting the benefit of MAT in OUD treatment and reducing all-cause and opioid-related mortality after NFO, the low rates of MAT observed in this study are consistent with previous reports that these medications remain underutilized.17 Screening for OUD, in conjunction with increasing access to and utilization of OUD treatment modalities, is an established and integral component of overdose prevention efforts. For VA clinicians, the Psychotropic Drug Safety Initiative (PDSI) dashboard can be used to identify patients diagnosed with OUD who are not yet on MAT.18 Initiatives to expand MAT access through the ED have the potential to provide life-saving interventions and bridge care in the interim until patients are able to become established with a long-term health care practitioner.19
Conclusions
This is the first study to describe HSU, medication interventions, and ORAEs among veterans who survive NFO. Studies have shown that veterans with a history of NFO are at increased risk of subsequent AEs and premature death.6,7,10,14 As such, NFOs represent crucial opportunities to identify high-risk individuals and ensure provision of adequate care. Recent data supports the development of a holistic, multimodal approach focused on adequate treatment of conditions that contribute to opioid-related risks, including mental health disorders, SUDs, pain diagnoses, and medical comorbidities.3,14 Interventions designed to improve access, engagement, and retention in such care therefore play a pivotal role in overdose prevention and reducing mortality.
Although existing risk mitigation initiatives have improved opioid prescribing and safety within the VA, the findings of this study suggest that there remains room for improvement, and the need for well-coordinated efforts to reduce risks associated with both prescribed and illicit opioid use cannot be overstated. Rates of overdose deaths not only remain high but are projected to continue increasing in coming years, despite advances in clinical practice aimed at reducing harms associated with opioid use. The present findings aim to help identify processes with the potential to reduce rates of overdose, death, and adverse sequelae in high-risk populations. However, future studies are warranted to expand on these findings and contribute to ongoing efforts in reducing opioid-related harms and overdose deaths. This study may provide critical insight to inform further investigations to guide such interventions and highlight tools that health care facilities even outside the VA can consider implementing.
Acknowledgments
The authors would like to thank Jonathan Lacro, PharmD, BCPP, for his guidance with this important clinical topic and navigating IRB submissions.
The number of opioid-related overdose deaths in the United States is estimated to have increased 6-fold over the past 2 decades.1 In 2017, more than two-thirds of drug overdose deaths involved opioids, yielding a mortality rate of 14.9 per 100,000.2 Not only does the opioid epidemic currently pose a significant public health crisis characterized by high morbidity and mortality, but it is also projected to worsen in coming years. According to Chen and colleagues, opioid overdose deaths are estimated to increase by 147% from 2015 to 2025.3 That projects almost 82,000 US deaths annually and > 700,000 deaths in this period—even before accounting for surges in opioid overdoses and opioid-related mortality coinciding with the COVID-19 pandemic.3,4
As health systems and communities globally struggle with unprecedented losses and stressors introduced by the pandemic, emerging data warrants escalating concerns with regard to increased vulnerability to relapse and overdose among those with mental health and substance use disorders (SUDs). In a recent report, the American Medical Association estimates that opioid-related deaths have increased in more than 40 states with the COVID-19 pandemic.4
Veterans are twice as likely to experience a fatal opioid overdose compared with their civilian counterparts.5 While several risk mitigation strategies have been employed in recent years to improve opioid prescribing and safety within the US Department of Veterans Affairs (VA), veterans continue to overdose on opioids, both prescribed and obtained illicitly.6 Variables shown to be strongly associated with opioid overdose risk include presence of mental health disorders, SUDs, medical conditions involving impaired drug metabolism or excretion, respiratory disorders, higher doses of opioids, concomitant use of sedative medications, and history of overdose.6-8 Many veterans struggle with chronic pain and those prescribed high doses of opioids were more likely to have comorbid pain diagnoses, mental health disorders, and SUDs.9 Dashboards and predictive models, such as the Stratification Tool for Opioid Risk Mitigation (STORM) and the Risk Index for Overdose or Serious Opioid-induced Respiratory Depression (RIOSORD), incorporate such factors to stratify overdose risk among veterans, in an effort to prioritize high-risk individuals for review and provision of care.6,10,11 Despite recent recognition that overdose prevention likely requires a holistic approach that addresses the biopsychosocial factors contributing to opioid-related morbidity and mortality, it is unclear whether veterans are receiving adequate and appropriate treatment for contributing conditions.
There are currently no existing studies that describe health service utilization (HSU), medication interventions, and rates of opioid-related adverse events (ORAEs) among veterans after survival of a nonfatal opioid overdose (NFO). Clinical characteristics of veterans treated for opioid overdose at a VA emergency department (ED) have previously been described by Clement and Stock.12 Despite improvements that have been made in VA opioid prescribing and safety, knowledge gaps remain with regard to best practices for opioid overdose prevention. The aim of this study was to characterize HSU and medication interventions in veterans following NFO, as well as the frequency of ORAEs after overdose. The findings of this study may aid in the identification of areas for targeted improvement in the prevention and reduction of opioid overdoses and adverse opioid-related sequelae.
Methods
This retrospective descriptive study was conducted at VA San Diego Healthcare System (VASDHCS) in California. Subjects included were veterans administered naloxone in the ED for suspected opioid overdose between July 1, 2013 and April 1, 2017. The study population was identified through data retrieved from automated drug dispensing systems, which was then confirmed through manual chart review of notes associated with the index ED visit. Inclusion criteria included documented increased respiration or responsiveness following naloxone administration. Subjects were excluded if they demonstrated lack of response to naloxone, overdosed secondary to inpatient administration of opioids, received palliative or hospice care during the study period, or were lost to follow-up.
Data were collected via retrospective chart review and included date of index ED visit, demographics, active prescriptions, urine drug screen (UDS) results, benzodiazepine (BZD) use corroborated by positive UDS or mention of BZD in index visit chart notes, whether overdose was determined to be a suicide attempt, and naloxone kit dispensing. Patient data was collected for 2 years following overdose, including: ORAEs; ED visits; hospitalizations; repeat overdoses; fatal overdose; whether subjects were still alive; follow-up visits for pain management, mental health, and addiction treatment services; and visits to the psychiatric emergency clinic. Clinical characteristics, such as mental health disorder diagnoses, SUDs, and relevant medical conditions also were collected. Statistical analysis was performed using Microsoft Excel and included only descriptive statistics.
Results
Ninety-three patients received naloxone in the VASDHCS ED. Thirty-five met inclusion criteria and were included in the primary analysis. All subjects received IV naloxone with a mean 0.8 mg IV boluses (range, 0.1-4.4 mg).
Most patients were male with a mean age of 59.8 years (Table 1). Almost all overdoses were nonintentional except for 3 suicide attempts that were reviewed by the Suicide Prevention Committee. Three patients had previously been treated for opioid overdose at the VA with a documented positive clinical response to naloxone administration.
At the time of overdose, 29 patients (82.9%) had an active opioid prescription. Of these, the majority were issued through the VA with a mean 117 mg morphine equivalent daily dose (MEDD). Interestingly, only 24 of the 28 patients with a UDS collected at time of overdose tested positive for opioids, which may be attributable to the use of synthetic opioids, which are not reliably detected by traditional UDS. Concomitant BZD use was involved in 13 of the 35 index overdoses (37.1%), although only 6 patients (17.1%) had an active BZD prescription at time of overdose. Seven patients (20.0%) were prescribed medication-assisted treatment (MAT) for opioid use disorder (OUD), with all 7 using methadone. According to VA records, only 1 patient had previously been dispensed a naloxone kit at any point prior to overdosing. Mental health and SUD diagnoses frequently co-occurred, with 20 patients (57.1%) having at least 1 mental health condition and at least 1 SUD.
Rates of follow-up varied by clinician type in the 6 months after NFO (Figure). Of those with mental health disorders, 15 patients (45.5%) received mental health services before and after overdose, while 8 (40.0%) and 10 (50.0%) of those with SUDs received addiction treatment services before and after overdose, respectively. Seven patients presented to the psychiatric emergency clinic within 6 months prior to overdose and 5 patients within the 6 months following overdose.
Of patients with VA opioid prescriptions, within 2 years of NFO, 9 (42.9%) had their opioids discontinued, and 18 (85.7%) had MEDD reductions ranging from 10 mg to 150 mg (12.5-71.4% reduction) with a mean of 63 mg. Two of the 4 patients with active BZD prescriptions at the time of the overdose event had their prescriptions continued. Seven patients (20.0%) were dispensed naloxone kits following overdose (Table 2).
Rates of ORAEs ranged from 0% to 17% with no documented overdose fatalities. Examples of AEs observed in this study included ED visits or hospitalizations involving opioid withdrawal, opioid-related personality changes, and opioid overdose. Five patients died during the study period, yielding an all-cause mortality rate of 14.3% with a mean time to death of 10.8 months. The causes of death were largely unknown except for 1 patient, whose death was reportedly investigated as an accidental medication overdose without additional information.
Repeat overdose verified by hospital records occurred in 4 patients (11.4%) within 2 years. Patients who experienced a subsequent overdose were prescribed higher doses of opioids with a mean MEDD among VA prescriptions of 130 mg vs 114 mg for those without repeat overdose. In this group, 3 patients (75.0%) also had concomitant BZD use, which was proportionally higher than the 10 patients (32.3%) without a subsequent overdose. Of note, 2 of the 4 patients with a repeat overdose had their opioid doses increased above the MEDD prescribed at the time of index overdose. None of the 4 subjects who experienced a repeat overdose were initiated on MAT within 2 years according to VA records.
Discussions
This retrospective study is representative of many veterans receiving VA care, despite the small sample size. Clinical characteristics observed in the study population were generally consistent with those published by Clement and Stock, including high rates of medical and psychiatric comorbidities.12 Subjects in both studies were prescribed comparable dosages of opioids; among those prescribed opioids but not BZDs through the VA, the mean MEDD was 117 mg in our study compared with 126 mg in the Clement and Stock study. Since implementation of the Opioid Safety Initiative (OSI) in 2013, opioid prescribing practices have improved nationwide across VA facilities, including successful reduction in the numbers of patients prescribed high-dose opioids and concurrent BZDs.13
Despite the tools and resources available to clinicians, discontinuing opioid therapy remains a difficult process. Concerns related to mental health and/or substance-use related decompensations often exist in the setting of rapid dose reductions or abrupt discontinuation of opioids.6 Although less than half of patients in the present study with an active opioid prescription at time of index overdose had their opioids discontinued within 2 years, it is reassuring to note the much higher rate of those with subsequent decreases in their prescribed doses, as well as the 50% reduction in BZD coprescribing. Ultimately, these findings remain consistent with the VA goals of mitigating harm, improving opioid prescribing, and ensuring the safe use of opioid medications when clinically appropriate.
Moreover, recent evidence suggests that interventions focused solely on opioid prescribing practices are becoming increasingly limited in their impact on reducing opioid-related deaths and will likely be insufficient for addressing the opioid epidemic as it continues to evolve. According to Chen and colleagues, opioid overdose deaths are projected to increase over the next several years, while further reduction in the incidence of prescription opioid misuse is estimated to decrease overdose deaths by only 3% to 5.3%. In the context of recent surges in synthetic opioid use, it is projected that 80% of overdose deaths between 2016 and 2025 will be attributable to illicit opioids.3 Such predictions underscore the urgent need to adopt alternative approaches to risk-reducing measures and policy change.
The increased risk of mortality associated with opioid misuse and overdose is well established in the current literature. However, less is known regarding the rate of ORAEs after survival of an NFO. Olfson and colleagues sought to address this knowledge gap by characterizing mortality risks in 76,325 US adults within 1 year following NFO.14 Among their studied population, all-cause mortality occurred at a rate of 778.3 per 10,000 person-years, which was 24 times greater than that of the general population. This emphasizes the need for the optimization of mental health services, addiction treatment, and medical care for these individuals at higher risk.
Limitations
Certain factors and limitations should be considered when interpreting the results of this study. Given that the study included only veterans, factors such as the demographic and clinical characteristics more commonly observed among these patients should be taken into account and may in turn limit the generalizability of these findings to nonveteran populations. Another major limitation is the small sample size; the study period and by extension, the number of patients able to be included in the present study were restricted by the availability of retrievable data from automated drug dispensing systems. Patients without documented response to naloxone were excluded from the study due to low clinical suspicion for opioid overdose, although the possibility that the dose administered was too low to produce a robust clinical response cannot be definitively ruled out. The lack of reliable methods to capture events and overdoses treated outside of the VA may have resulted in underestimations of the true occurrence of ORAEs following NFO. Information regarding naloxone administration outside VA facilities, such as in transport to the hospital, self-reported, or bystander administration, was similarly limited by lack of reliable methods for retrieving such data and absence of documentation in VA records. Although all interventions and outcomes reported in the present study occurred within 2 years following NFO, further conclusions pertaining to the relative timing of specific interventions and ORAEs cannot be made. Lastly, this study did not investigate the direct impact of opioid risk mitigation initiatives implemented by the VA in the years coinciding with the study period.
Future Directions
Despite these limitations, an important strength of this study is its ability to identify potential areas for targeted improvement and to guide further efforts relating to the prevention of opioid overdose and opioid-related mortality among veterans. Identification of individuals at high risk for opioid overdose and misuse is an imperative first step that allows for the implementation of downstream risk-mitigating interventions. Within the VA, several tools have been developed in recent years to provide clinicians with additional resources and support in this regard.6,15
No more than half of those diagnosed with mental health disorders and SUDs in the present study received outpatient follow-up care for these conditions within 6 months following NFO, which may suggest high rates of inadequate treatment. Given the strong association between mental health disorders, SUDs, and increased risk of overdose, increasing engagement with mental health and addiction treatment services may be paramount to preventing subsequent ORAEs, including repeat overdose.6-9,11
Naloxone kit dispensing represents another area for targeted improvement. Interventions may include clinician education and systematic changes, such as implementing protocols that boost the likelihood of high-risk individuals being provided with naloxone at the earliest opportunity. Bystander-administered naloxone programs can also be considered for increasing naloxone access and reducing opioid-related mortality.16
Finally, despite evidence supporting the benefit of MAT in OUD treatment and reducing all-cause and opioid-related mortality after NFO, the low rates of MAT observed in this study are consistent with previous reports that these medications remain underutilized.17 Screening for OUD, in conjunction with increasing access to and utilization of OUD treatment modalities, is an established and integral component of overdose prevention efforts. For VA clinicians, the Psychotropic Drug Safety Initiative (PDSI) dashboard can be used to identify patients diagnosed with OUD who are not yet on MAT.18 Initiatives to expand MAT access through the ED have the potential to provide life-saving interventions and bridge care in the interim until patients are able to become established with a long-term health care practitioner.19
Conclusions
This is the first study to describe HSU, medication interventions, and ORAEs among veterans who survive NFO. Studies have shown that veterans with a history of NFO are at increased risk of subsequent AEs and premature death.6,7,10,14 As such, NFOs represent crucial opportunities to identify high-risk individuals and ensure provision of adequate care. Recent data supports the development of a holistic, multimodal approach focused on adequate treatment of conditions that contribute to opioid-related risks, including mental health disorders, SUDs, pain diagnoses, and medical comorbidities.3,14 Interventions designed to improve access, engagement, and retention in such care therefore play a pivotal role in overdose prevention and reducing mortality.
Although existing risk mitigation initiatives have improved opioid prescribing and safety within the VA, the findings of this study suggest that there remains room for improvement, and the need for well-coordinated efforts to reduce risks associated with both prescribed and illicit opioid use cannot be overstated. Rates of overdose deaths not only remain high but are projected to continue increasing in coming years, despite advances in clinical practice aimed at reducing harms associated with opioid use. The present findings aim to help identify processes with the potential to reduce rates of overdose, death, and adverse sequelae in high-risk populations. However, future studies are warranted to expand on these findings and contribute to ongoing efforts in reducing opioid-related harms and overdose deaths. This study may provide critical insight to inform further investigations to guide such interventions and highlight tools that health care facilities even outside the VA can consider implementing.
Acknowledgments
The authors would like to thank Jonathan Lacro, PharmD, BCPP, for his guidance with this important clinical topic and navigating IRB submissions.
1. Centers for Disease Control and Prevention. Data overview: the drug overdose epidemic: behind the numbers. Updated March 25, 2021. Accessed February 9, 2022. www.cdc.gov/drugoverdose/data/index.html
2. Scholl L, Seth P, Kariisa M, Wilson N, Baldwin G. Drug and Opioid-Involved Overdose Deaths - United States, 2013-2017. MMWR Morb Mortal Wkly Rep. 2018;67(5152):1419-1427. Published 2018 Jan 4. doi:10.15585/mmwr.mm675152e1 3. Chen Q, Larochelle MR, Weaver DT, et al. Prevention of prescription opioid misuse and projected overdose deaths in the United States. JAMA Netw Open. 2019;2(2):e187621. Published 2019 Feb 1. doi:10.1001/jamanetworkopen.2018.7621
4. American Medical Association. Issue brief: nation’s drug-related overdose and death epidemic continues to worsen. Updated November 12, 2021. Accessed February 11, 2022. https://www.ama-assn.org/system/files/issue-brief-increases-in-opioid-related-overdose.pdf
5. Bohnert AS, Ilgen MA, Galea S, McCarthy JF, Blow FC. Accidental poisoning mortality among patients in the Department of Veterans Affairs Health System. Med Care. 2011;49(4):393-396. doi:10.1097/MLR.0b013e318202aa27
6. Lewis ET, Trafton J, Oliva E. Data-based case reviews of patients with opioid related risk factors as a tool to prevent overdose and suicide. Accessed February 9, 2022. www.hsrd.research.va.gov/for_researchers/cyber_seminars/archives/2488-notes.pdf
7. Zedler B, Xie L, Wang L, et al. Risk factors for serious prescription opioid-related toxicity or overdose among Veterans Health Administration patients. Pain Med. 2014;15(11):1911-1929. doi:10.1111/pme.12480
8. Webster LR. Risk Factors for Opioid-Use Disorder and Overdose. Anesth Analg. 2017;125(5):1741-1748. doi:10.1213/ANE.0000000000002496
9. Morasco BJ, Duckart JP, Carr TP, Deyo RA, Dobscha SK. Clinical characteristics of veterans prescribed high doses of opioid medications for chronic non-cancer pain. Pain. 2010;151(3):625-632. doi:10.1016/j.pain.2010.08.002
10. Oliva EM, Bowe T, Tavakoli S, et al. Development and applications of the Veterans Health Administration’s Stratification Tool for Opioid Risk Mitigation (STORM) to improve opioid safety and prevent overdose and suicide. Psychol Serv. 2017;14(1):34-49. doi:10.1037/ser0000099
11. Zedler B, Xie L, Wang L, et al. Development of a risk index for serious prescription opioid-induced respiratory depression or overdose in Veterans’ Health Administration patients. Pain Med. 2015;16(8):1566-1579. doi:10.1111/pme.12777
12. Clement C, Stock C. Who Overdoses at a VA Emergency Department? Fed Pract. 2016;33(11):14-20.
13. Lin LA, Bohnert ASB, Kerns RD, Clay MA, Ganoczy D, Ilgen MA. Impact of the Opioid Safety Initiative on opioid-related prescribing in veterans. Pain. 2017;158(5):833-839. doi:10.1097/j.pain.0000000000000837
14. Olfson M, Crystal S, Wall M, Wang S, Liu SM, Blanco C. Causes of death after nonfatal opioid overdose [published correction appears in JAMA Psychiatry. 2018 Aug 1;75(8):867]. JAMA Psychiatry. 2018;75(8):820-827. doi:10.1001/jamapsychiatry.2018.1471
15. US Department of Veterans Affairs, Veterans Health Administration. VHA pain management – opioid safety – clinical tools. Updated November 14, 2019. Accessed February 9, 2022. https://www.va.gov/PAINMANAGEMENT/Opioid_Safety/Clinical_Tools.asp
16. Doe-Simkins M, Walley AY, Epstein A, Moyer P. Saved by the nose: bystander-administered intranasal naloxone hydrochloride for opioid overdose. Am J Public Health. 2009;99(5):788-791. doi:10.2105/AJPH.2008.146647
17. Larochelle MR, Bernson D, Land T, et al. Medication for opioid use disorder after nonfatal opioid overdose and association with mortality: a cohort study. Ann Intern Med. 2018;169(3):137-145. doi:10.7326/M17-3107
18. Wiechers I. Program focuses on safe psychiatric medication. Published April 21, 2016. Accessed February 9, 2022. https://blogs.va.gov/VAntage/27099/program-focuses-safe-psychiatric-medication/
19. Newman S; California Health Care Foundation. How to pay for it – MAT in the emergency department: FAQ. Published March 2019. Accessed February 9, 2022. https://www.chcf.org/wp-content/uploads/2019/03/HowToPayForMATinED.pdf
1. Centers for Disease Control and Prevention. Data overview: the drug overdose epidemic: behind the numbers. Updated March 25, 2021. Accessed February 9, 2022. www.cdc.gov/drugoverdose/data/index.html
2. Scholl L, Seth P, Kariisa M, Wilson N, Baldwin G. Drug and Opioid-Involved Overdose Deaths - United States, 2013-2017. MMWR Morb Mortal Wkly Rep. 2018;67(5152):1419-1427. Published 2018 Jan 4. doi:10.15585/mmwr.mm675152e1 3. Chen Q, Larochelle MR, Weaver DT, et al. Prevention of prescription opioid misuse and projected overdose deaths in the United States. JAMA Netw Open. 2019;2(2):e187621. Published 2019 Feb 1. doi:10.1001/jamanetworkopen.2018.7621
4. American Medical Association. Issue brief: nation’s drug-related overdose and death epidemic continues to worsen. Updated November 12, 2021. Accessed February 11, 2022. https://www.ama-assn.org/system/files/issue-brief-increases-in-opioid-related-overdose.pdf
5. Bohnert AS, Ilgen MA, Galea S, McCarthy JF, Blow FC. Accidental poisoning mortality among patients in the Department of Veterans Affairs Health System. Med Care. 2011;49(4):393-396. doi:10.1097/MLR.0b013e318202aa27
6. Lewis ET, Trafton J, Oliva E. Data-based case reviews of patients with opioid related risk factors as a tool to prevent overdose and suicide. Accessed February 9, 2022. www.hsrd.research.va.gov/for_researchers/cyber_seminars/archives/2488-notes.pdf
7. Zedler B, Xie L, Wang L, et al. Risk factors for serious prescription opioid-related toxicity or overdose among Veterans Health Administration patients. Pain Med. 2014;15(11):1911-1929. doi:10.1111/pme.12480
8. Webster LR. Risk Factors for Opioid-Use Disorder and Overdose. Anesth Analg. 2017;125(5):1741-1748. doi:10.1213/ANE.0000000000002496
9. Morasco BJ, Duckart JP, Carr TP, Deyo RA, Dobscha SK. Clinical characteristics of veterans prescribed high doses of opioid medications for chronic non-cancer pain. Pain. 2010;151(3):625-632. doi:10.1016/j.pain.2010.08.002
10. Oliva EM, Bowe T, Tavakoli S, et al. Development and applications of the Veterans Health Administration’s Stratification Tool for Opioid Risk Mitigation (STORM) to improve opioid safety and prevent overdose and suicide. Psychol Serv. 2017;14(1):34-49. doi:10.1037/ser0000099
11. Zedler B, Xie L, Wang L, et al. Development of a risk index for serious prescription opioid-induced respiratory depression or overdose in Veterans’ Health Administration patients. Pain Med. 2015;16(8):1566-1579. doi:10.1111/pme.12777
12. Clement C, Stock C. Who Overdoses at a VA Emergency Department? Fed Pract. 2016;33(11):14-20.
13. Lin LA, Bohnert ASB, Kerns RD, Clay MA, Ganoczy D, Ilgen MA. Impact of the Opioid Safety Initiative on opioid-related prescribing in veterans. Pain. 2017;158(5):833-839. doi:10.1097/j.pain.0000000000000837
14. Olfson M, Crystal S, Wall M, Wang S, Liu SM, Blanco C. Causes of death after nonfatal opioid overdose [published correction appears in JAMA Psychiatry. 2018 Aug 1;75(8):867]. JAMA Psychiatry. 2018;75(8):820-827. doi:10.1001/jamapsychiatry.2018.1471
15. US Department of Veterans Affairs, Veterans Health Administration. VHA pain management – opioid safety – clinical tools. Updated November 14, 2019. Accessed February 9, 2022. https://www.va.gov/PAINMANAGEMENT/Opioid_Safety/Clinical_Tools.asp
16. Doe-Simkins M, Walley AY, Epstein A, Moyer P. Saved by the nose: bystander-administered intranasal naloxone hydrochloride for opioid overdose. Am J Public Health. 2009;99(5):788-791. doi:10.2105/AJPH.2008.146647
17. Larochelle MR, Bernson D, Land T, et al. Medication for opioid use disorder after nonfatal opioid overdose and association with mortality: a cohort study. Ann Intern Med. 2018;169(3):137-145. doi:10.7326/M17-3107
18. Wiechers I. Program focuses on safe psychiatric medication. Published April 21, 2016. Accessed February 9, 2022. https://blogs.va.gov/VAntage/27099/program-focuses-safe-psychiatric-medication/
19. Newman S; California Health Care Foundation. How to pay for it – MAT in the emergency department: FAQ. Published March 2019. Accessed February 9, 2022. https://www.chcf.org/wp-content/uploads/2019/03/HowToPayForMATinED.pdf
Preliminary Observations of Veterans Without HIV Who Have Mycobacterium avium Complex Pulmonary Disease
Nontuberculous Mycobacterium (NTM) is a ubiquitous organism known to cause a variety of infections in susceptible hosts; however, pulmonary infection is the most common. Mycobacterium avium complex (MAC) is the most prevalent cause of NTM-related pulmonary disease (NTM-PD) and is associated with underlying structural lung disease, such as chronic obstructive pulmonary disease (COPD) and noncystic fibrosis bronchiectasis.1-3
Diagnosis of NTM-PD requires (1) symptoms or radiographic abnormality; and (2) at least 2 sputum cultures positive with the same organism or at least 1 positive culture result on bronchoscopy (wash, lavage, or biopsy).1 Notably, the natural history of untreated NTM-PD varies, though even mild disease may progress substantially.4-6 Progressive disease is more likely to occur in those with a positive smear or more extensive radiographic findings at the initial diagnosis.7 A nationwide Medicare-based study showed that patients with NTM-PD had a higher rate of all-cause mortality than did patients without NTM-PD.8 In a study of 123 patients from Taiwan with MAC-PD, lack of treatment was an independent predictor of mortality.9 Given the risk of progressive morbidity and mortality, recent guidelines recommend initiation of a susceptibility driven, macrolide-based, 3-drug treatment regimen over watchful waiting.10
MAC-PD is increasingly recognized among US veterans.11,12 The Jesse Brown Veterans Affairs Medical Center (JBVAMC) in south/west Chicago serves a large, predominantly Black male population of veterans many of whom are socioeconomically underresourced, and half are aged ≥ 65 years. We observed that initiation of guideline-directed therapy in veterans with MAC-PD at JBVAMC varied among health care professionals (HCPs) in the pulmonary clinic. Therefore, the purpose of this retrospective study was to describe and compare the characteristics of veterans without HIV were diagnosed with MAC-PD and managed at JBVAMC.
Methods
The hospital microbiology department identified veterans diagnosed with NTM at JBVAMC between October 2008 and July 2019. Veterans included in the study were considered to have MAC-PD per American Thoracic Society (ATS)/Infectious Diseases Society of America (ISDA) guidelines and those diagnosed with HIV were excluded from analysis. The electronic health record (EHR) was queried for pertinent demographics, smoking history, comorbidities, and symptoms at the time of a positive mycobacterial culture. Computed tomography (CT) and pulmonary function tests (PFTs) performed within 1 year of diagnosis were included. PFTs were assessed in accordance with Global Initiative for Obstructive Lung Disease (GOLD) criteria, with normal forced expiratory volume in 1 second (FEV1) and forced vital capacity (FVC) values defined as ≥ 80% and a normal FEV1/FVC ratio defined as ≥ 70. The diffusion capacity of lung for carbon monoxide (DLCO) was assessed per 2017 European Respiratory Society (ERS) technical standards and was considered reduced if below the lower limit of normal.13 Information regarding treatment decisions, initiation, and cessation were collected. All-cause mortality was recorded if available in the EHR at the time of data collection.
Statistical analysis was performed using Mann-Whitney U and Fisher exact tests where appropriate. P < .05 was considered statistically significant. The study was approved by the JBVAMC Institutional Review Board.
Results
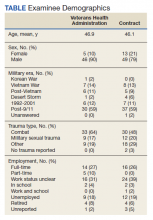
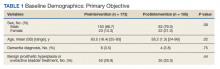
We identified 43 veterans who had a positive culture for MAC; however, only 19 veterans met the diagnostic criteria for MAC-PD and were included in the study (Table). The cohort included predominantly Black and male veterans with a median age of 74 years at time of diagnosis (range, 45-92). Sixteen veterans had underlying lung disease (84.2%), and 16 (84.2%) were current or former smokers. Common comorbidities included COPD, obstructive sleep apnea, gastroesophageal reflux disease, and lung cancer. Respiratory symptoms were reported in 17 veterans (89.5%), 15 (78.9%) had a chronic cough, and 10 (52.6%) had dyspnea. Fifteen veterans had a chest CT scan within 1 year of diagnosis: A nodular and tree-in-bud pattern was most commonly found in 13 (86.7%) of veterans. Thirteen veterans had PFTs within 1 year of MAC-PD diagnosis, of whom 6 had a restrictive pattern with percent predicted FVC < 80%, and 9 had evidence of obstruction with FEV1/FVC < 70. DLCO was below the lower limit of normal in 18 veterans. Finally, 6 veterans were deceased at the time of the study.
Of the 19 veterans, guideline-directed, combination antimycobacterial therapy for MAC-PD was initiated in only 10 (52.6%) patients due to presence of symptoms and/or imaging abnormalities. Treatment was deferred due to improved symptoms, concern for adverse events (AEs), or lost to follow-up. Five veterans stopped treatment prematurely due to AEs, lost to follow-up, or all-cause mortality. Assessment of differences between treated and untreated groups revealed no significant difference in race, sex, age, body mass index (BMI), symptom presence, or chest CT abnormalities. There was no statistically significant difference in all-cause mortality (40% and 22.2% in treated and untreated group, respectively).
To further understand the differences of this cohort, the 13 veterans alive at time of the study were compared with the 6 who had since died of all-cause mortality. No statistically significant differences were found.
Discussion
Consistent with previous reports in the literature, veterans in our cohort were predominantly current or former smoking males with underlying COPD and bronchiectasis.1-3,11,12 Chest CT findings varied: Most veterans presented not only with nodules and tree-in-bud opacities, but also a high frequency of fibrosis and emphysema. PFTs revealed a variety of obstruction and restrictive patterns, and most veterans had a reduced DLCO, though it is unclear whether this is reflective of underlying emphysema, fibrosis, or an alternative cardiopulmonary disease.13,14
While underlying structural lung disease may have been a risk factor for MAC-PD in this cohort, the contribution of environmental and domiciliary factors in metropolitan Chicago neighborhoods is unknown. JBVAMC serves an underresourced population who live in the west and south Chicago neighborhoods. Household factors, ambient and indoor air pollution, and potential contamination of the water supply and surface soil may contribute to the prevalence of MAC-PD in this group.15-19 Further studies are warranted to characterize MAC-PD and its treatment in veterans without HIV who reside in underresourced urban communities in the US.
Recent ATS, European Society of Clinical Microbiology and Infectious Diseases, and IDSA guidelines recommend combination antimycobacterial therapy for patients who meet clinical, radiographic, and microbiologic criteria for the diagnosis of MAC-PD.10 Patients who meet these diagnostic criteria, particularly patients with smear positivity or fibrocavitary disease, should be treated because of risk of unfavorable outcomes.15,20-22 However, we found that the initiation of guideline-recommended antimycobacterial therapy in veterans without HIV with MAC-PD were inconsistent among HCPs. The reasons underlying this phenomenon were not apparent beyond cited reasons for treatment initiation or deference. Despite this inconsistency, there was no clear difference in age, BMI, symptom burden, radiographic abnormality, or all-cause mortality between treatment groups. Existing studies support slow but substantial progression of untreated MAC-PD, and while treatment prevents deterioration of the disease, it does not prevent progression of bronchiectasis.6 The natural history of MAC-PD in this veteran cohort has yet to be fully elucidated. Furthermore, the 50% treatment dropout rate was higher than previously reported rates (11-33%).5 However, the small number of veterans in this study precludes meaningful comparison with similar reports in the literature.
Limitations
The limitations of this small, single-center, retrospective study prevent a robust, generalizable comparison between groups. Further studies are warranted to characterize MAC-PD and its treatment in veterans without HIV who reside in underresourced urban communities in the US.24-26
Conclusions
These data suggest that clinical, imaging, and treatment attributes of MAC-PD in veterans without HIV who reside in metropolitan Chicago are heterogeneous and are associated with a relatively high mortality rate. Although there was no difference in the attributes or outcomes of veterans who did and did not initiate treatment despite current recommendations, further studies are needed to better explore these relationships.
1. Griffith DE, Aksamit T, Brown-Elliott BA, et al. An official ATS/ IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases [published correction appears in Am J Respir Crit Care Med. 2007 Apr 1;175(7):744-5. Dosage error in article text]. Am J Respir Crit Care Med. 2007;175(4):367-416. doi:10.1164/rccm.200604-571ST
2. Prevots DR, Shaw PA, Strickland D, et al. Nontuberculous mycobacterial lung disease prevalence at four integrated health care delivery systems. Am J Respir Crit Care Med. 2010;182(7):970-976. doi:10.1164/rccm.201002-0310OC
3. Winthrop KL, Marras TK, Adjemian J, Zhang H, Wang P, Zhang Q. Incidence and prevalence of nontuberculous mycobacterial lung disease in a large U.S. managed care health plan, 2008-2015. Ann Am Thorac Soc. 2020;17(2):178-185. doi:10.1513/AnnalsATS.201804-236OC
4. Field SK, Fisher D, Cowie RL. Mycobacterium avium complex pulmonary disease in patients without HIV infection. Chest. 2004;126(2):566-581. doi:10.1378/chest.126.2.566
5. Kimizuka Y, Hoshino Y, Nishimura T, et al. Retrospective evaluation of natural course in mild cases of Mycobacterium avium complex pulmonary disease. PLoS One. 2019;14(4):e0216034. Published 2019 Apr 25. doi:10.1371/journal.pone.0216034
6. Kotilainen H, Valtonen V, Tukiainen P, Poussa T, Eskola J, Järvinen A. Clinical findings in relation to mortality in nontuberculous mycobacterial infections: patients with Mycobacterium avium complex have better survival than patients with other mycobacteria. Eur J Clin Microbiol Infect Dis. 2015;34(9):1909-1918. doi:10.1007/s10096-015-2432-8.
7. Hwang JA, Kim S, Jo KW, Shim TS. Natural history of Mycobacterium avium complex lung disease in untreated patients with stable course. Eur Respir J. 2017;49(3):1600537. Published 2017 Mar 8. doi:10.1183/13993003.00537-2016
8. Adjemian J, Olivier KN, Seitz AE, Holland SM, Prevots DR. Prevalence of nontuberculous mycobacterial lung disease in U.S. Medicare beneficiaries. Am J Respir Crit Care Med. 2012;185(8):881-886. doi:10.1164/rccm.201111-2016OC
9. Wang PH, Pan SW, Shu CC, et al. Clinical course and risk factors of mortality in Mycobacterium avium complex lung disease without initial treatment. Respir Med. 2020;171:106070. doi:10.1016/j.rmed.2020.106070
10. Daley CL, Iaccarino JM, Lange C, et al. Treatment of nontuberculous mycobacterial pulmonary disease: an official ATS/ ERS/ESCMID/IDSA Clinical Practice Guideline [published correction appears in Clin Infect Dis. 2020 Dec 31;71(11):3023]. Clin Infect Dis. 2020;71(4):e1-e36. doi:10.1093/cid/ciaa241
11. Mirsaeidi M, Hadid W, Ericsoussi B, Rodgers D, Sadikot RT. Non-tuberculous mycobacterial disease is common in patients with non-cystic fibrosis bronchiectasis. Int J Infect Dis. 2013;17(11):e1000-e1004. doi:10.1016/j.ijid.2013.03.018
12. Oda G, Winters MA, Pacheco SM, et al. Clusters of nontuberculous mycobacteria linked to water sources at three Veterans Affairs medical centers. Infect Control Hosp Epidemiol. 2020;41(3):320-330. doi:10.1017/ice.2019.342
13. Stanojevic S, Graham BL, Cooper BG, et al. Official ERS technical standards: Global Lung Function Initiative reference values for the carbon monoxide transfer factor for Caucasians [published correction appears in Eur Respir J. 2020 Oct 15;56(4):]. Eur Respir J. 2017;50(3):1700010. Published 2017 Sep 11. doi:10.1183/13993003.00010-2017
14. Macintyre N, Crapo RO, Viegi G, et al. Standardisation of the single-breath determination of carbon monoxide uptake in the lung. Eur Respir J. 2005;26(4):720-735. doi:10.1183/09031936.05.00034905
15. Chalmers JD, Balavoine C, Castellotti PF, et al. European Respiratory Society International Congress, Madrid, 2019: nontuberculous mycobacterial pulmonary disease highlights. ERJ Open Res. 2020;6(4):00317-2020. Published 2020 Oct 19. doi:10.1183/23120541.00317-2020
16. Hamilton LA, Falkinham JO. Aerosolization of Mycobacterium avium and Mycobacterium abscessus from a household ultrasonic humidifier. J Med Microbiol. 2018;67(10):1491-1495. doi:10.1099/jmm.0.000822
17. Hannah CE, Ford BA, Chung J, Ince D, Wanat KA. Characteristics of nontuberculous mycobacterial infections at a midwestern tertiary hospital: a retrospective study of 365 patients. Open Forum Infect Dis. 2020;7(6):ofaa173. Published 2020 May 25. doi:10.1093/ofid/ofaa173
18. Rautiala S, Torvinen E, Torkko P, et al. Potentially pathogenic, slow-growing mycobacteria released into workplace air during the remediation of buildings. J Occup Environ Hyg. 2004;1(1):1-6. doi:10.1080/15459620490250008
19. Tzou CL, Dirac MA, Becker AL, et al. Association between Mycobacterium avium complex pulmonary disease and mycobacteria in home water and soil. Ann Am Thorac Soc. 2020;17(1):57-62. doi:10.1513/AnnalsATS.201812-915OC
20. Daley CL, Winthrop KL. Mycobacterium avium complex: addressing gaps in diagnosis and management. J Infect Dis. 2020;222(suppl 4):S199-S211. doi:10.1093/infdis/jiaa354 21. Kwon BS, Lee JH, Koh Y, et al. The natural history of noncavitary nodular bronchiectatic Mycobacterium avium complex lung disease. Respir Med. 2019;150:45-50. doi:10.1016/j.rmed.2019.02.007
22. Nasiri MJ, Ebrahimi G, Arefzadeh S, Zamani S, Nikpor Z, Mirsaeidi M. Antibiotic therapy success rate in pulmonary Mycobacterium avium complex: a systematic review and meta-analysis. Expert Rev Anti Infect Ther. 2020;18(3):263- 273. doi:10.1080/14787210.2020.1720650
23. Diel R, Lipman M, Hoefsloot W. High mortality in patients with Mycobacterium avium complex lung disease: a systematic review. BMC Infect Dis. 2018;18(1):206. Published 2018 May 3. doi:10.1186/s12879-018-3113-x
24. Marras TK, Prevots DR, Jamieson FB, Winthrop KL; Pulmonary MAC Outcomes Group. Opinions differ by expertise in Mycobacterium avium complex disease. Ann Am Thorac Soc. 2014;11(1):17-22. doi:10.1513/AnnalsATS.201305-136OC
25. Plotinsky RN, Talbot EA, von Reyn CF. Proposed definitions for epidemiologic and clinical studies of Mycobacterium avium complex pulmonary disease. PLoS One. 2013;8(11):e77385. Published 2013 Nov 12. doi:10.1371/journal.pone.0077385
26. Swenson C, Zerbe CS, Fennelly K. Host variability in NTM disease: implications for research needs. Front Microbiol. 2018;9:2901. Published 2018 Dec 3. doi:10.3389/fmicb.2018.02901
Nontuberculous Mycobacterium (NTM) is a ubiquitous organism known to cause a variety of infections in susceptible hosts; however, pulmonary infection is the most common. Mycobacterium avium complex (MAC) is the most prevalent cause of NTM-related pulmonary disease (NTM-PD) and is associated with underlying structural lung disease, such as chronic obstructive pulmonary disease (COPD) and noncystic fibrosis bronchiectasis.1-3
Diagnosis of NTM-PD requires (1) symptoms or radiographic abnormality; and (2) at least 2 sputum cultures positive with the same organism or at least 1 positive culture result on bronchoscopy (wash, lavage, or biopsy).1 Notably, the natural history of untreated NTM-PD varies, though even mild disease may progress substantially.4-6 Progressive disease is more likely to occur in those with a positive smear or more extensive radiographic findings at the initial diagnosis.7 A nationwide Medicare-based study showed that patients with NTM-PD had a higher rate of all-cause mortality than did patients without NTM-PD.8 In a study of 123 patients from Taiwan with MAC-PD, lack of treatment was an independent predictor of mortality.9 Given the risk of progressive morbidity and mortality, recent guidelines recommend initiation of a susceptibility driven, macrolide-based, 3-drug treatment regimen over watchful waiting.10
MAC-PD is increasingly recognized among US veterans.11,12 The Jesse Brown Veterans Affairs Medical Center (JBVAMC) in south/west Chicago serves a large, predominantly Black male population of veterans many of whom are socioeconomically underresourced, and half are aged ≥ 65 years. We observed that initiation of guideline-directed therapy in veterans with MAC-PD at JBVAMC varied among health care professionals (HCPs) in the pulmonary clinic. Therefore, the purpose of this retrospective study was to describe and compare the characteristics of veterans without HIV were diagnosed with MAC-PD and managed at JBVAMC.
Methods
The hospital microbiology department identified veterans diagnosed with NTM at JBVAMC between October 2008 and July 2019. Veterans included in the study were considered to have MAC-PD per American Thoracic Society (ATS)/Infectious Diseases Society of America (ISDA) guidelines and those diagnosed with HIV were excluded from analysis. The electronic health record (EHR) was queried for pertinent demographics, smoking history, comorbidities, and symptoms at the time of a positive mycobacterial culture. Computed tomography (CT) and pulmonary function tests (PFTs) performed within 1 year of diagnosis were included. PFTs were assessed in accordance with Global Initiative for Obstructive Lung Disease (GOLD) criteria, with normal forced expiratory volume in 1 second (FEV1) and forced vital capacity (FVC) values defined as ≥ 80% and a normal FEV1/FVC ratio defined as ≥ 70. The diffusion capacity of lung for carbon monoxide (DLCO) was assessed per 2017 European Respiratory Society (ERS) technical standards and was considered reduced if below the lower limit of normal.13 Information regarding treatment decisions, initiation, and cessation were collected. All-cause mortality was recorded if available in the EHR at the time of data collection.
Statistical analysis was performed using Mann-Whitney U and Fisher exact tests where appropriate. P < .05 was considered statistically significant. The study was approved by the JBVAMC Institutional Review Board.
Results
We identified 43 veterans who had a positive culture for MAC; however, only 19 veterans met the diagnostic criteria for MAC-PD and were included in the study (Table). The cohort included predominantly Black and male veterans with a median age of 74 years at time of diagnosis (range, 45-92). Sixteen veterans had underlying lung disease (84.2%), and 16 (84.2%) were current or former smokers. Common comorbidities included COPD, obstructive sleep apnea, gastroesophageal reflux disease, and lung cancer. Respiratory symptoms were reported in 17 veterans (89.5%), 15 (78.9%) had a chronic cough, and 10 (52.6%) had dyspnea. Fifteen veterans had a chest CT scan within 1 year of diagnosis: A nodular and tree-in-bud pattern was most commonly found in 13 (86.7%) of veterans. Thirteen veterans had PFTs within 1 year of MAC-PD diagnosis, of whom 6 had a restrictive pattern with percent predicted FVC < 80%, and 9 had evidence of obstruction with FEV1/FVC < 70. DLCO was below the lower limit of normal in 18 veterans. Finally, 6 veterans were deceased at the time of the study.
Of the 19 veterans, guideline-directed, combination antimycobacterial therapy for MAC-PD was initiated in only 10 (52.6%) patients due to presence of symptoms and/or imaging abnormalities. Treatment was deferred due to improved symptoms, concern for adverse events (AEs), or lost to follow-up. Five veterans stopped treatment prematurely due to AEs, lost to follow-up, or all-cause mortality. Assessment of differences between treated and untreated groups revealed no significant difference in race, sex, age, body mass index (BMI), symptom presence, or chest CT abnormalities. There was no statistically significant difference in all-cause mortality (40% and 22.2% in treated and untreated group, respectively).
To further understand the differences of this cohort, the 13 veterans alive at time of the study were compared with the 6 who had since died of all-cause mortality. No statistically significant differences were found.
Discussion
Consistent with previous reports in the literature, veterans in our cohort were predominantly current or former smoking males with underlying COPD and bronchiectasis.1-3,11,12 Chest CT findings varied: Most veterans presented not only with nodules and tree-in-bud opacities, but also a high frequency of fibrosis and emphysema. PFTs revealed a variety of obstruction and restrictive patterns, and most veterans had a reduced DLCO, though it is unclear whether this is reflective of underlying emphysema, fibrosis, or an alternative cardiopulmonary disease.13,14
While underlying structural lung disease may have been a risk factor for MAC-PD in this cohort, the contribution of environmental and domiciliary factors in metropolitan Chicago neighborhoods is unknown. JBVAMC serves an underresourced population who live in the west and south Chicago neighborhoods. Household factors, ambient and indoor air pollution, and potential contamination of the water supply and surface soil may contribute to the prevalence of MAC-PD in this group.15-19 Further studies are warranted to characterize MAC-PD and its treatment in veterans without HIV who reside in underresourced urban communities in the US.
Recent ATS, European Society of Clinical Microbiology and Infectious Diseases, and IDSA guidelines recommend combination antimycobacterial therapy for patients who meet clinical, radiographic, and microbiologic criteria for the diagnosis of MAC-PD.10 Patients who meet these diagnostic criteria, particularly patients with smear positivity or fibrocavitary disease, should be treated because of risk of unfavorable outcomes.15,20-22 However, we found that the initiation of guideline-recommended antimycobacterial therapy in veterans without HIV with MAC-PD were inconsistent among HCPs. The reasons underlying this phenomenon were not apparent beyond cited reasons for treatment initiation or deference. Despite this inconsistency, there was no clear difference in age, BMI, symptom burden, radiographic abnormality, or all-cause mortality between treatment groups. Existing studies support slow but substantial progression of untreated MAC-PD, and while treatment prevents deterioration of the disease, it does not prevent progression of bronchiectasis.6 The natural history of MAC-PD in this veteran cohort has yet to be fully elucidated. Furthermore, the 50% treatment dropout rate was higher than previously reported rates (11-33%).5 However, the small number of veterans in this study precludes meaningful comparison with similar reports in the literature.
Limitations
The limitations of this small, single-center, retrospective study prevent a robust, generalizable comparison between groups. Further studies are warranted to characterize MAC-PD and its treatment in veterans without HIV who reside in underresourced urban communities in the US.24-26
Conclusions
These data suggest that clinical, imaging, and treatment attributes of MAC-PD in veterans without HIV who reside in metropolitan Chicago are heterogeneous and are associated with a relatively high mortality rate. Although there was no difference in the attributes or outcomes of veterans who did and did not initiate treatment despite current recommendations, further studies are needed to better explore these relationships.
Nontuberculous Mycobacterium (NTM) is a ubiquitous organism known to cause a variety of infections in susceptible hosts; however, pulmonary infection is the most common. Mycobacterium avium complex (MAC) is the most prevalent cause of NTM-related pulmonary disease (NTM-PD) and is associated with underlying structural lung disease, such as chronic obstructive pulmonary disease (COPD) and noncystic fibrosis bronchiectasis.1-3
Diagnosis of NTM-PD requires (1) symptoms or radiographic abnormality; and (2) at least 2 sputum cultures positive with the same organism or at least 1 positive culture result on bronchoscopy (wash, lavage, or biopsy).1 Notably, the natural history of untreated NTM-PD varies, though even mild disease may progress substantially.4-6 Progressive disease is more likely to occur in those with a positive smear or more extensive radiographic findings at the initial diagnosis.7 A nationwide Medicare-based study showed that patients with NTM-PD had a higher rate of all-cause mortality than did patients without NTM-PD.8 In a study of 123 patients from Taiwan with MAC-PD, lack of treatment was an independent predictor of mortality.9 Given the risk of progressive morbidity and mortality, recent guidelines recommend initiation of a susceptibility driven, macrolide-based, 3-drug treatment regimen over watchful waiting.10
MAC-PD is increasingly recognized among US veterans.11,12 The Jesse Brown Veterans Affairs Medical Center (JBVAMC) in south/west Chicago serves a large, predominantly Black male population of veterans many of whom are socioeconomically underresourced, and half are aged ≥ 65 years. We observed that initiation of guideline-directed therapy in veterans with MAC-PD at JBVAMC varied among health care professionals (HCPs) in the pulmonary clinic. Therefore, the purpose of this retrospective study was to describe and compare the characteristics of veterans without HIV were diagnosed with MAC-PD and managed at JBVAMC.
Methods
The hospital microbiology department identified veterans diagnosed with NTM at JBVAMC between October 2008 and July 2019. Veterans included in the study were considered to have MAC-PD per American Thoracic Society (ATS)/Infectious Diseases Society of America (ISDA) guidelines and those diagnosed with HIV were excluded from analysis. The electronic health record (EHR) was queried for pertinent demographics, smoking history, comorbidities, and symptoms at the time of a positive mycobacterial culture. Computed tomography (CT) and pulmonary function tests (PFTs) performed within 1 year of diagnosis were included. PFTs were assessed in accordance with Global Initiative for Obstructive Lung Disease (GOLD) criteria, with normal forced expiratory volume in 1 second (FEV1) and forced vital capacity (FVC) values defined as ≥ 80% and a normal FEV1/FVC ratio defined as ≥ 70. The diffusion capacity of lung for carbon monoxide (DLCO) was assessed per 2017 European Respiratory Society (ERS) technical standards and was considered reduced if below the lower limit of normal.13 Information regarding treatment decisions, initiation, and cessation were collected. All-cause mortality was recorded if available in the EHR at the time of data collection.
Statistical analysis was performed using Mann-Whitney U and Fisher exact tests where appropriate. P < .05 was considered statistically significant. The study was approved by the JBVAMC Institutional Review Board.
Results
We identified 43 veterans who had a positive culture for MAC; however, only 19 veterans met the diagnostic criteria for MAC-PD and were included in the study (Table). The cohort included predominantly Black and male veterans with a median age of 74 years at time of diagnosis (range, 45-92). Sixteen veterans had underlying lung disease (84.2%), and 16 (84.2%) were current or former smokers. Common comorbidities included COPD, obstructive sleep apnea, gastroesophageal reflux disease, and lung cancer. Respiratory symptoms were reported in 17 veterans (89.5%), 15 (78.9%) had a chronic cough, and 10 (52.6%) had dyspnea. Fifteen veterans had a chest CT scan within 1 year of diagnosis: A nodular and tree-in-bud pattern was most commonly found in 13 (86.7%) of veterans. Thirteen veterans had PFTs within 1 year of MAC-PD diagnosis, of whom 6 had a restrictive pattern with percent predicted FVC < 80%, and 9 had evidence of obstruction with FEV1/FVC < 70. DLCO was below the lower limit of normal in 18 veterans. Finally, 6 veterans were deceased at the time of the study.
Of the 19 veterans, guideline-directed, combination antimycobacterial therapy for MAC-PD was initiated in only 10 (52.6%) patients due to presence of symptoms and/or imaging abnormalities. Treatment was deferred due to improved symptoms, concern for adverse events (AEs), or lost to follow-up. Five veterans stopped treatment prematurely due to AEs, lost to follow-up, or all-cause mortality. Assessment of differences between treated and untreated groups revealed no significant difference in race, sex, age, body mass index (BMI), symptom presence, or chest CT abnormalities. There was no statistically significant difference in all-cause mortality (40% and 22.2% in treated and untreated group, respectively).
To further understand the differences of this cohort, the 13 veterans alive at time of the study were compared with the 6 who had since died of all-cause mortality. No statistically significant differences were found.
Discussion
Consistent with previous reports in the literature, veterans in our cohort were predominantly current or former smoking males with underlying COPD and bronchiectasis.1-3,11,12 Chest CT findings varied: Most veterans presented not only with nodules and tree-in-bud opacities, but also a high frequency of fibrosis and emphysema. PFTs revealed a variety of obstruction and restrictive patterns, and most veterans had a reduced DLCO, though it is unclear whether this is reflective of underlying emphysema, fibrosis, or an alternative cardiopulmonary disease.13,14
While underlying structural lung disease may have been a risk factor for MAC-PD in this cohort, the contribution of environmental and domiciliary factors in metropolitan Chicago neighborhoods is unknown. JBVAMC serves an underresourced population who live in the west and south Chicago neighborhoods. Household factors, ambient and indoor air pollution, and potential contamination of the water supply and surface soil may contribute to the prevalence of MAC-PD in this group.15-19 Further studies are warranted to characterize MAC-PD and its treatment in veterans without HIV who reside in underresourced urban communities in the US.
Recent ATS, European Society of Clinical Microbiology and Infectious Diseases, and IDSA guidelines recommend combination antimycobacterial therapy for patients who meet clinical, radiographic, and microbiologic criteria for the diagnosis of MAC-PD.10 Patients who meet these diagnostic criteria, particularly patients with smear positivity or fibrocavitary disease, should be treated because of risk of unfavorable outcomes.15,20-22 However, we found that the initiation of guideline-recommended antimycobacterial therapy in veterans without HIV with MAC-PD were inconsistent among HCPs. The reasons underlying this phenomenon were not apparent beyond cited reasons for treatment initiation or deference. Despite this inconsistency, there was no clear difference in age, BMI, symptom burden, radiographic abnormality, or all-cause mortality between treatment groups. Existing studies support slow but substantial progression of untreated MAC-PD, and while treatment prevents deterioration of the disease, it does not prevent progression of bronchiectasis.6 The natural history of MAC-PD in this veteran cohort has yet to be fully elucidated. Furthermore, the 50% treatment dropout rate was higher than previously reported rates (11-33%).5 However, the small number of veterans in this study precludes meaningful comparison with similar reports in the literature.
Limitations
The limitations of this small, single-center, retrospective study prevent a robust, generalizable comparison between groups. Further studies are warranted to characterize MAC-PD and its treatment in veterans without HIV who reside in underresourced urban communities in the US.24-26
Conclusions
These data suggest that clinical, imaging, and treatment attributes of MAC-PD in veterans without HIV who reside in metropolitan Chicago are heterogeneous and are associated with a relatively high mortality rate. Although there was no difference in the attributes or outcomes of veterans who did and did not initiate treatment despite current recommendations, further studies are needed to better explore these relationships.
1. Griffith DE, Aksamit T, Brown-Elliott BA, et al. An official ATS/ IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases [published correction appears in Am J Respir Crit Care Med. 2007 Apr 1;175(7):744-5. Dosage error in article text]. Am J Respir Crit Care Med. 2007;175(4):367-416. doi:10.1164/rccm.200604-571ST
2. Prevots DR, Shaw PA, Strickland D, et al. Nontuberculous mycobacterial lung disease prevalence at four integrated health care delivery systems. Am J Respir Crit Care Med. 2010;182(7):970-976. doi:10.1164/rccm.201002-0310OC
3. Winthrop KL, Marras TK, Adjemian J, Zhang H, Wang P, Zhang Q. Incidence and prevalence of nontuberculous mycobacterial lung disease in a large U.S. managed care health plan, 2008-2015. Ann Am Thorac Soc. 2020;17(2):178-185. doi:10.1513/AnnalsATS.201804-236OC
4. Field SK, Fisher D, Cowie RL. Mycobacterium avium complex pulmonary disease in patients without HIV infection. Chest. 2004;126(2):566-581. doi:10.1378/chest.126.2.566
5. Kimizuka Y, Hoshino Y, Nishimura T, et al. Retrospective evaluation of natural course in mild cases of Mycobacterium avium complex pulmonary disease. PLoS One. 2019;14(4):e0216034. Published 2019 Apr 25. doi:10.1371/journal.pone.0216034
6. Kotilainen H, Valtonen V, Tukiainen P, Poussa T, Eskola J, Järvinen A. Clinical findings in relation to mortality in nontuberculous mycobacterial infections: patients with Mycobacterium avium complex have better survival than patients with other mycobacteria. Eur J Clin Microbiol Infect Dis. 2015;34(9):1909-1918. doi:10.1007/s10096-015-2432-8.
7. Hwang JA, Kim S, Jo KW, Shim TS. Natural history of Mycobacterium avium complex lung disease in untreated patients with stable course. Eur Respir J. 2017;49(3):1600537. Published 2017 Mar 8. doi:10.1183/13993003.00537-2016
8. Adjemian J, Olivier KN, Seitz AE, Holland SM, Prevots DR. Prevalence of nontuberculous mycobacterial lung disease in U.S. Medicare beneficiaries. Am J Respir Crit Care Med. 2012;185(8):881-886. doi:10.1164/rccm.201111-2016OC
9. Wang PH, Pan SW, Shu CC, et al. Clinical course and risk factors of mortality in Mycobacterium avium complex lung disease without initial treatment. Respir Med. 2020;171:106070. doi:10.1016/j.rmed.2020.106070
10. Daley CL, Iaccarino JM, Lange C, et al. Treatment of nontuberculous mycobacterial pulmonary disease: an official ATS/ ERS/ESCMID/IDSA Clinical Practice Guideline [published correction appears in Clin Infect Dis. 2020 Dec 31;71(11):3023]. Clin Infect Dis. 2020;71(4):e1-e36. doi:10.1093/cid/ciaa241
11. Mirsaeidi M, Hadid W, Ericsoussi B, Rodgers D, Sadikot RT. Non-tuberculous mycobacterial disease is common in patients with non-cystic fibrosis bronchiectasis. Int J Infect Dis. 2013;17(11):e1000-e1004. doi:10.1016/j.ijid.2013.03.018
12. Oda G, Winters MA, Pacheco SM, et al. Clusters of nontuberculous mycobacteria linked to water sources at three Veterans Affairs medical centers. Infect Control Hosp Epidemiol. 2020;41(3):320-330. doi:10.1017/ice.2019.342
13. Stanojevic S, Graham BL, Cooper BG, et al. Official ERS technical standards: Global Lung Function Initiative reference values for the carbon monoxide transfer factor for Caucasians [published correction appears in Eur Respir J. 2020 Oct 15;56(4):]. Eur Respir J. 2017;50(3):1700010. Published 2017 Sep 11. doi:10.1183/13993003.00010-2017
14. Macintyre N, Crapo RO, Viegi G, et al. Standardisation of the single-breath determination of carbon monoxide uptake in the lung. Eur Respir J. 2005;26(4):720-735. doi:10.1183/09031936.05.00034905
15. Chalmers JD, Balavoine C, Castellotti PF, et al. European Respiratory Society International Congress, Madrid, 2019: nontuberculous mycobacterial pulmonary disease highlights. ERJ Open Res. 2020;6(4):00317-2020. Published 2020 Oct 19. doi:10.1183/23120541.00317-2020
16. Hamilton LA, Falkinham JO. Aerosolization of Mycobacterium avium and Mycobacterium abscessus from a household ultrasonic humidifier. J Med Microbiol. 2018;67(10):1491-1495. doi:10.1099/jmm.0.000822
17. Hannah CE, Ford BA, Chung J, Ince D, Wanat KA. Characteristics of nontuberculous mycobacterial infections at a midwestern tertiary hospital: a retrospective study of 365 patients. Open Forum Infect Dis. 2020;7(6):ofaa173. Published 2020 May 25. doi:10.1093/ofid/ofaa173
18. Rautiala S, Torvinen E, Torkko P, et al. Potentially pathogenic, slow-growing mycobacteria released into workplace air during the remediation of buildings. J Occup Environ Hyg. 2004;1(1):1-6. doi:10.1080/15459620490250008
19. Tzou CL, Dirac MA, Becker AL, et al. Association between Mycobacterium avium complex pulmonary disease and mycobacteria in home water and soil. Ann Am Thorac Soc. 2020;17(1):57-62. doi:10.1513/AnnalsATS.201812-915OC
20. Daley CL, Winthrop KL. Mycobacterium avium complex: addressing gaps in diagnosis and management. J Infect Dis. 2020;222(suppl 4):S199-S211. doi:10.1093/infdis/jiaa354 21. Kwon BS, Lee JH, Koh Y, et al. The natural history of noncavitary nodular bronchiectatic Mycobacterium avium complex lung disease. Respir Med. 2019;150:45-50. doi:10.1016/j.rmed.2019.02.007
22. Nasiri MJ, Ebrahimi G, Arefzadeh S, Zamani S, Nikpor Z, Mirsaeidi M. Antibiotic therapy success rate in pulmonary Mycobacterium avium complex: a systematic review and meta-analysis. Expert Rev Anti Infect Ther. 2020;18(3):263- 273. doi:10.1080/14787210.2020.1720650
23. Diel R, Lipman M, Hoefsloot W. High mortality in patients with Mycobacterium avium complex lung disease: a systematic review. BMC Infect Dis. 2018;18(1):206. Published 2018 May 3. doi:10.1186/s12879-018-3113-x
24. Marras TK, Prevots DR, Jamieson FB, Winthrop KL; Pulmonary MAC Outcomes Group. Opinions differ by expertise in Mycobacterium avium complex disease. Ann Am Thorac Soc. 2014;11(1):17-22. doi:10.1513/AnnalsATS.201305-136OC
25. Plotinsky RN, Talbot EA, von Reyn CF. Proposed definitions for epidemiologic and clinical studies of Mycobacterium avium complex pulmonary disease. PLoS One. 2013;8(11):e77385. Published 2013 Nov 12. doi:10.1371/journal.pone.0077385
26. Swenson C, Zerbe CS, Fennelly K. Host variability in NTM disease: implications for research needs. Front Microbiol. 2018;9:2901. Published 2018 Dec 3. doi:10.3389/fmicb.2018.02901
1. Griffith DE, Aksamit T, Brown-Elliott BA, et al. An official ATS/ IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases [published correction appears in Am J Respir Crit Care Med. 2007 Apr 1;175(7):744-5. Dosage error in article text]. Am J Respir Crit Care Med. 2007;175(4):367-416. doi:10.1164/rccm.200604-571ST
2. Prevots DR, Shaw PA, Strickland D, et al. Nontuberculous mycobacterial lung disease prevalence at four integrated health care delivery systems. Am J Respir Crit Care Med. 2010;182(7):970-976. doi:10.1164/rccm.201002-0310OC
3. Winthrop KL, Marras TK, Adjemian J, Zhang H, Wang P, Zhang Q. Incidence and prevalence of nontuberculous mycobacterial lung disease in a large U.S. managed care health plan, 2008-2015. Ann Am Thorac Soc. 2020;17(2):178-185. doi:10.1513/AnnalsATS.201804-236OC
4. Field SK, Fisher D, Cowie RL. Mycobacterium avium complex pulmonary disease in patients without HIV infection. Chest. 2004;126(2):566-581. doi:10.1378/chest.126.2.566
5. Kimizuka Y, Hoshino Y, Nishimura T, et al. Retrospective evaluation of natural course in mild cases of Mycobacterium avium complex pulmonary disease. PLoS One. 2019;14(4):e0216034. Published 2019 Apr 25. doi:10.1371/journal.pone.0216034
6. Kotilainen H, Valtonen V, Tukiainen P, Poussa T, Eskola J, Järvinen A. Clinical findings in relation to mortality in nontuberculous mycobacterial infections: patients with Mycobacterium avium complex have better survival than patients with other mycobacteria. Eur J Clin Microbiol Infect Dis. 2015;34(9):1909-1918. doi:10.1007/s10096-015-2432-8.
7. Hwang JA, Kim S, Jo KW, Shim TS. Natural history of Mycobacterium avium complex lung disease in untreated patients with stable course. Eur Respir J. 2017;49(3):1600537. Published 2017 Mar 8. doi:10.1183/13993003.00537-2016
8. Adjemian J, Olivier KN, Seitz AE, Holland SM, Prevots DR. Prevalence of nontuberculous mycobacterial lung disease in U.S. Medicare beneficiaries. Am J Respir Crit Care Med. 2012;185(8):881-886. doi:10.1164/rccm.201111-2016OC
9. Wang PH, Pan SW, Shu CC, et al. Clinical course and risk factors of mortality in Mycobacterium avium complex lung disease without initial treatment. Respir Med. 2020;171:106070. doi:10.1016/j.rmed.2020.106070
10. Daley CL, Iaccarino JM, Lange C, et al. Treatment of nontuberculous mycobacterial pulmonary disease: an official ATS/ ERS/ESCMID/IDSA Clinical Practice Guideline [published correction appears in Clin Infect Dis. 2020 Dec 31;71(11):3023]. Clin Infect Dis. 2020;71(4):e1-e36. doi:10.1093/cid/ciaa241
11. Mirsaeidi M, Hadid W, Ericsoussi B, Rodgers D, Sadikot RT. Non-tuberculous mycobacterial disease is common in patients with non-cystic fibrosis bronchiectasis. Int J Infect Dis. 2013;17(11):e1000-e1004. doi:10.1016/j.ijid.2013.03.018
12. Oda G, Winters MA, Pacheco SM, et al. Clusters of nontuberculous mycobacteria linked to water sources at three Veterans Affairs medical centers. Infect Control Hosp Epidemiol. 2020;41(3):320-330. doi:10.1017/ice.2019.342
13. Stanojevic S, Graham BL, Cooper BG, et al. Official ERS technical standards: Global Lung Function Initiative reference values for the carbon monoxide transfer factor for Caucasians [published correction appears in Eur Respir J. 2020 Oct 15;56(4):]. Eur Respir J. 2017;50(3):1700010. Published 2017 Sep 11. doi:10.1183/13993003.00010-2017
14. Macintyre N, Crapo RO, Viegi G, et al. Standardisation of the single-breath determination of carbon monoxide uptake in the lung. Eur Respir J. 2005;26(4):720-735. doi:10.1183/09031936.05.00034905
15. Chalmers JD, Balavoine C, Castellotti PF, et al. European Respiratory Society International Congress, Madrid, 2019: nontuberculous mycobacterial pulmonary disease highlights. ERJ Open Res. 2020;6(4):00317-2020. Published 2020 Oct 19. doi:10.1183/23120541.00317-2020
16. Hamilton LA, Falkinham JO. Aerosolization of Mycobacterium avium and Mycobacterium abscessus from a household ultrasonic humidifier. J Med Microbiol. 2018;67(10):1491-1495. doi:10.1099/jmm.0.000822
17. Hannah CE, Ford BA, Chung J, Ince D, Wanat KA. Characteristics of nontuberculous mycobacterial infections at a midwestern tertiary hospital: a retrospective study of 365 patients. Open Forum Infect Dis. 2020;7(6):ofaa173. Published 2020 May 25. doi:10.1093/ofid/ofaa173
18. Rautiala S, Torvinen E, Torkko P, et al. Potentially pathogenic, slow-growing mycobacteria released into workplace air during the remediation of buildings. J Occup Environ Hyg. 2004;1(1):1-6. doi:10.1080/15459620490250008
19. Tzou CL, Dirac MA, Becker AL, et al. Association between Mycobacterium avium complex pulmonary disease and mycobacteria in home water and soil. Ann Am Thorac Soc. 2020;17(1):57-62. doi:10.1513/AnnalsATS.201812-915OC
20. Daley CL, Winthrop KL. Mycobacterium avium complex: addressing gaps in diagnosis and management. J Infect Dis. 2020;222(suppl 4):S199-S211. doi:10.1093/infdis/jiaa354 21. Kwon BS, Lee JH, Koh Y, et al. The natural history of noncavitary nodular bronchiectatic Mycobacterium avium complex lung disease. Respir Med. 2019;150:45-50. doi:10.1016/j.rmed.2019.02.007
22. Nasiri MJ, Ebrahimi G, Arefzadeh S, Zamani S, Nikpor Z, Mirsaeidi M. Antibiotic therapy success rate in pulmonary Mycobacterium avium complex: a systematic review and meta-analysis. Expert Rev Anti Infect Ther. 2020;18(3):263- 273. doi:10.1080/14787210.2020.1720650
23. Diel R, Lipman M, Hoefsloot W. High mortality in patients with Mycobacterium avium complex lung disease: a systematic review. BMC Infect Dis. 2018;18(1):206. Published 2018 May 3. doi:10.1186/s12879-018-3113-x
24. Marras TK, Prevots DR, Jamieson FB, Winthrop KL; Pulmonary MAC Outcomes Group. Opinions differ by expertise in Mycobacterium avium complex disease. Ann Am Thorac Soc. 2014;11(1):17-22. doi:10.1513/AnnalsATS.201305-136OC
25. Plotinsky RN, Talbot EA, von Reyn CF. Proposed definitions for epidemiologic and clinical studies of Mycobacterium avium complex pulmonary disease. PLoS One. 2013;8(11):e77385. Published 2013 Nov 12. doi:10.1371/journal.pone.0077385
26. Swenson C, Zerbe CS, Fennelly K. Host variability in NTM disease: implications for research needs. Front Microbiol. 2018;9:2901. Published 2018 Dec 3. doi:10.3389/fmicb.2018.02901
Impact of Lithium on Suicidality in the Veteran Population
Suicide is the tenth leading cause of death in the United States claiming nearly 48,000 individuals in 2019 and is the second leading cause of death among individuals aged 10 to 34 years.1 From 1999 to 2019, the suicide rate increased by 33%.1 In a retrospective study evaluating suicide risk in > 29,000 men, veterans had a greater risk for suicide in all age groups except for the oldest when compared with nonveterans.2 Another study of > 800,000 veterans found that younger veterans were most at risk for suicide.3 Veterans with completed suicides have a high incidence of affective disorders comorbid with substance use disorders, and therefore it is imperative to optimally treat these conditions to address suicidality.4 Additionally, a retrospective case-control study of veterans who died by suicide matched to controls identified that the cases had significantly higher rates of mental health conditions and suicidal ideation. Given that the veteran population is at higher risk of suicide, research of treatments to address suicidal ideation in veterans is needed.5
Lithium and Antisuicidal Properties
Lithium is the oldest treatment for bipolar disorder and is a long-standing first-line option due to its well-established efficacy as a mood stabilizer.6 Lithium’s antisuicidal properties separate it from the other pharmacologic options for bipolar disorder. A possible explanation for lithium’s unique antisuicidal properties is that these effects are mediated by its impact on aggression and impulsivity, which are both linked to an increased suicide risk.7,8 A meta-analysis by Baldessarini and colleagues demonstrated that patients with mood disorders who were prescribed lithium had a 5 times lower risk of suicide and attempts than did those not treated with lithium.9 Lithium’s current place in therapy is in the treatment of bipolar disorder and major depressive disorder augmentation.10-12Smith and colleagues found that in a cohort study of 21,194 veterans diagnosed with mental health conditions and initiated on lithium or valproate, there were no significant differences in associations with suicide observed between these agents over 365 days; however, there was a significant increased risk of suicide among patients discontinuing or modifying lithium within the first 180 days of treatment.13
Currently, lithium is thought to be underutilized at the US Department of Veterans Affairs (VA) Michael E. DeBakey VA Medical Center (MEDVAMC) in Houston, Texas, based on the number of prescriptions of lithium in the large population of veterans seen by mental health clinicians. MEDVAMC is a 538-bed academic teaching hospital serving approximately 130,000 veterans in southeast Texas. The Mental Health Care Line has 73 inpatient beds and an outpatient clinic serving > 12,000 patients annually. By retrospectively evaluating changes in suicidality in a sample of veterans prescribed lithium, we may be able to better understand the role that lithium plays in a population that has a higher suicide rate than does the general population. The primary objective of this study was to evaluate the change in number of suicide attempts from 3 months prior to lithium initiation to 3 months following a 6-month duration of lithium use. The secondary objective was to determine the change in suicidal ideation from the period prior to lithium use to the period following 6 months of lithium use.
Methods
This was a single-site, retrospective chart review conducted between October 2017 and April 2018. Prior to data collection, the MEDVAMC Research and Development committee approved the study as quality assurance research. Patients with an active lithium prescription were identified using the VA Lithium Lab Monitoring Dashboard, which includes all patients on lithium, their lithium level, and other data such as upcoming appointments.
Inclusion criteria consisted of adults who were aged ≥ 18 years, had an active lithium prescription on the date of data extraction, and had an active lithium prescription for at least 6 months. Patients were excluded if they had < 3 months of data before and/or after lithium was used for 6 months, and if they were initiated on lithium outside MEDVAMC. Cumulatively, patients had to have at least 12 months of data: 3 months prior to lithium use, at least 6 months of lithium use, and 9 months after lithium initiation.
Suicide Attempt and Suicidal Ideation Identification
When determining the number of suicide attempts, we recorded 4 data points: Veterans Crisis Line notes documenting suicide attempts, hospital admissions for suicide attempts, suicide behavior reports within the indicated time frame, and mental health progress notes documenting suicide attempts. Suicidal ideation was measured in 4 ways. First, we looked at the percentage of outpatient mental health progress notes documenting suicidal ideation. Second, using the Patient Health Questionnaire-9 (PHQ-9) depression assessments, we looked at the percentage of patients that indicated several days, more than half the days, or nearly every day to the question, “Thoughts that you would be better off dead or of hurting yourself in some way.”14 Third, we recorded the percentage of suicide risk assessments that patients responded yes to both questions on current preoccupation with suicidal thoughts and serious intent and plan to commit suicide with access to guns, stashed pills, or other means. Finally, we noted the percentage of suicide risk assessments where the assessment of risk was moderate or high.
A retrospective electronic health record (EHR) review was performed and the following information was obtained: patient demographics, lithium refill history, concomitant psychotropic medications and psychotherapy, lithium levels, comorbidities at lithium initiation, presence of a high-risk suicide flag in the EHR, suicide risk assessments, suicide behavior reports, Veteran Crisis Line notes, PHQ-9 assessments, and hospital admission and mental health outpatient notes. The lithium therapeutic range of 0.6-1.2 mmol/L is indicated for bipolar disorder and not other indications where the dose is typically titrated to effect rather than level. Medication possession ratio (MPR) was also calculated for lithium (sum of days’ supply for all fills in period ÷ number of days in period). A high-risk suicide flag alerts clinicians and staff that a mental health professional considers the veteran at risk for suicide.15 Statistical analysis was performed using the paired t test for means to assess proportional differences between variables for the primary and secondary outcomes. Descriptive statistics were used to describe the baseline characteristics.
Results
A total of 214 patients with an active prescription for lithium were identified on the Lithium Lab Monitoring Dashboard on October 31, 2017. After exclusion criteria were applied, 98 patients were included in the study (Figure 1). The 2 most common reasons for exclusion were due to patients not being on lithium for at least 6 months and being initiated on lithium at an outside facility. One patient was enrolled in a lithium research study (the medication ordered was lithium/placebo) and another patient refused all psychotropic medications according to the progress notes.
Most of the 98 patients (82.7%) were male with average age 50.5 years (Table 1). Almost half the patients (n = 47) were concomitantly participating in psychotherapy, and 50 (51.0%) patients received at least 1 antipsychotic medication. Twenty-nine patients had an active prescription for an additional mood stabilizer, and only 4 (4.1%) patients received lithium as monotherapy. Only 75 (76.5%) patients had a lithium level drawn during the 6 months of therapy, with 28 (37.3%) patients having a therapeutic lithium level (0.6 - 1.2 mmol/L). Seventy-one patients (72.4% ) were adherent to lithium therapy with a MPR > 0.8.16 Participants had 13 different psychiatric diagnoses at the time of lithium initiation; the most common were bipolar spectrum disorder (n = 38; 38.8%), depressive disorder (n = 27; 27.6%), and posttraumatic stress disorder (PTSD) (n = 26; 26.5%). Of note, 5 patients had a diagnosis of only PTSD without a concomitant mood disorder.
For the primary outcome, hospitalization for a suspected suicide attempt decreased from 4 (4.1%) before lithium use with a mean (SD) 0.04 (0.20) attempts per person to none within 3 months after lithium use for 6 months (t(97) = 2.03, P = .045) (Figure 2). The secondary outcome of hospitalization for suicidal ideations also decreased from 13 (13.3%) before lithium use with a mean (SD) 0.1 (0.3) ideations per person to 1 (1.0%) within 3 months after lithium use for 6 months with a mean (SD) 0.01 (0.1) ideations per person (t(97) = 3.68, P = .0004). Veteran Crisis Line calls also decreased from 4 (4.1%) with a mean (SD) 0.04 (0.2) calls per person to 1 (1.0%) within 3 months after 6 months of lithium with a mean (SD) 0.01 (0.1) calls per person (t(97) = 1.75, P = .08). The comparison of metrics from 3 months before lithium initiation and within 3 months after use saw decreases in all categories (Table 2). Outpatient notes documenting suicidal ideation decreased, as did the number of patients with a high-risk suicide flag.
Discussion
The results of this study suggest lithium may have a role in reducing suicidality in a veteran population. There was a statistically significant reduction in hospitalizations for suicide attempt and suicidal ideation after at least 6 months of lithium use. These results are comparable with a previously published study that observed significant decreases in suicidal behavior and/or hospitalization risks among veterans taking lithium compared with those not taking lithium.17 Our study was similar in respect to the reduced hospitalizations among a veteran population; however, the previous study did not report a difference in suicide attempts and lithium use. This could be related to the longer follow-up time in the previous study (3 years) vs our study (9 months).
Our study identified a significant reduction in Veteran Crisis Line calls after at least 6 months of lithium use. While a reduction in suicidal ideations could be implicated in the decrease in crisis line calls, there may be a confounding variable. It is possible that after lithium initiation, veterans had more frequent contact with health care practitioners due to laboratory test monitoring and follow-up visits and thus had concerns/crises addressed during these interactions ultimately leading to a decreased utilization of the crisis line. Interestingly, there was a reduction in mental health outpatient notes from the prelithium period to the 3-month period that followed 6 months of lithium therapy. However, our study did not report on the number of mental health outpatient notes or visits during the 6-month lithium duration. Additionally, time of year/season could have an impact on suicidality, but this relationship was not evaluated in this study.
The presence of high-risk suicide flags also decreased from the prelithium period to the period 3 months following 6 months of lithium use. High-risk flags are reviewed by the suicide prevention coordinators and mental health professionals every 90 days; therefore, the patients with flags had multiple opportunities for review and thus renewal or discontinuation during the study period. A similar rationale can be applied to the high-risk flag as with the Veteran Crisis Line reduction, although this change could also be representative of a decrease in suicidality. Our study is different from other lithium studies because it included patients with a multitude of psychiatric diagnoses rather than just mood disorders. Five of the patients had a diagnosis of only PTSD and no documented mood disorder at the time of lithium initiation. Additional research is needed on the impact of lithium on suicidality in veterans with PTSD and psychiatric conditions other than mood disorders.
Underutilization of Lithium
Despite widespread knowledge of lithium’s antisuicidal effects, it is underutilized as a mood stabilizer in the US.18 There are various modifiable barriers that impact the prescribing as well as use of lithium. Clinicians may not be fully aware of lithium’s antisuicidal properties and may also have a low level of confidence in patients’ likelihood of adherence to laboratory monitoring.18,19 Due to the narrow therapeutic index of lithium, the consequences of nonadherence to monitoring can be dangerous, which may deter mental health professionals from prescribing this antisuicidal agent. At MEDVAMC, only 72.4% of patients with a lithium prescription had a lithium level drawn within a 6-month period. This could be attributed to patient nonadherence (eg, the test was ordered but the patient did not go) or clinician nonadherence (eg, test was not ordered).
With increased clinician education as well as clinics dedicated to lithium management that allow for closer follow-up, facilities may see an increased level of comfort with lithium use. Lithium management clinics that provide close follow-up may also help address patient-related concerns about adverse effects and allow for close monitoring. To facilitate lithium monitoring at MEDVAMC, mental health practitioners and pharmacists developed a lithium test monitoring menu that serves as a “one-stop shop” for lithium baseline and ongoing test results.
In the future, we may study the impact of this test monitoring menu on lithium prescribing. One may also consider whether lithium levels need to be monitored at different frequencies (eg, less frequently for depression than bipolar disorder) depending on the diagnoses. A better understanding of the necessity for therapeutic monitoring may potentially reduce barriers to prescribing for patients who do not have indications that have a recommended therapeutic range (eg, bipolar disorder).
Lithium Adherence
A primary patient-related concern for low lithium utilization is poor adherence. In this sample, 71 patients (72.4%) were considered fully adherent. This was higher than the rate of 54.1% reported by Sajatovic and colleagues in a study evaluating adherence to lithium and other anticonvulsants in veterans with bipolar disorder.20 Patients’ beliefs about medications and overall health as well as knowledge of the illness and treatment may impact adherence.21 The literature indicates that strategies such as cognitive behavioral therapy (CBT) and didactic lectures positively impact patients’ attitudes about lithium, which ultimately influences adherence.21-23 Involving a family member or significant other in psychotherapy may also improve lithium adherence.21 Specifically in the VA, to address knowledge deficits and improve overall adherence, the Lithium Lab Monitoring Dashboard could be used to identify and invite new lithium starts to educational groups about lithium. These groups could also serve as lithium management clinics.
Limitations
There were several limitations to this study. This was a single-site, retrospective chart review with a small sample size. We studied a cross-section of veterans with only active prescriptions, which limited the sample size. The results should be interpreted cautiously because < 40% of patients who had a level drawn were in the therapeutic range. Patients whose lithium levels were outside of the therapeutic range may have not been fully adherent to the medication. Further analysis based on reason for lithium prescription (eg, bipolar disorder vs depression vs aggression/impulsivity in PTSD) may be helpful in better understanding the results.
Additionally, while we collected data on concomitant mood stabilizers and antipsychotics, we did not collect data on concurrent antidepressant therapy and only 4% of patients were on lithium monotherapy. Data regarding veterans undergoing concurrent CBT during their lithium trial were not assessed in this study and could be considered a confounding factor for future studies. We included any Veteran Crisis Line call in our results regardless of the reason for the call, which could have led to overreporting of this suicidality marker.
Given its small sample size, this study should be considered as hypothesis-generating. Further studies are needed to address lithium’s antisuicidal effects in specific diagnoses (eg, PTSD, anxiety, schizoaffective disorder) to better understand its place in therapy. Studies evaluating the relationship between dosing and suicidality may help provide insight into whether the antisuicidal effect of lithium is dose-dependent and whether a specific dose range rather than a therapeutic level should be targeted for antisuicidal purposes.
Conclusions
People treated for an affective disorder have a 30-times greater risk of suicide than do those in the general population; however, as lithium can reduce the risk of suicide and self-harm, it should continue to have an important role in clinical practice.24 At MEDVAMC, we observed a statistically significant reduction in hospitalization for suicide attempts and suicidal ideation in veterans prescribed lithium following nonfatal suicide behavior and suicidal ideation. Prospective randomized placebo-controlled studies are needed to better understand lithium’s antisuicidal effects.
1. Centers for Disease Control and Prevention. Preventing Suicide Fact Sheet. Updated April 2021. Accessed February 16, 2022. https://www.cdc.gov/suicide/pdf/preventing-suicide-factsheet-2021-508.pdf
2. Kaplan MS, McFarland BH, Huguet N, Valenstein M. Suicide risk and precipitating circumstances among young, middle-aged, and older male veterans. Am J Public Health. 2012;102 Suppl 1(Suppl 1):S131-S137. doi:10.2105/AJPH.2011.300445
3. Zivin K, Kim HM, McCarthy JF, et al. Suicide mortality among individuals receiving treatment for depression in the Veterans Affairs health system: associations with patient and treatment setting characteristics. Am J Public Health. 2007;97(12):2193-2198. doi:10.2105/AJPH.2007.115477
4. Lehmann L, McCormick RA, McCracken L. Suicidal behavior among patients in the VA health care system. Psychiatr Serv. 1995;46(10):1069-1071. doi:10.1176/ps.46.10.1069
5. Dobscha SK, Denneson LM, Kovas AE, et al. Correlates of suicide among veterans treated in primary care: case-control study of a nationally representative sample. J Gen Intern Med. 2014;29(suppl 4):853-860. doi:10.1007/s11606-014-3028-1
6. Malhi GS, Tanious M, Das P, Coulston CM, Berk M. Potential mechanisms of action of lithium in bipolar disorder. Current understanding. CNS Drugs. 2013;27(2):135-153. doi:10.1007/s40263-013-0039-0
7. Kovacsics CE, Gottesman II, Gould TD. Lithium’s antisuicidal efficacy: elucidation of neurobiological targets using endophenotype strategies. Annu Rev Pharmacol Toxicol. 2009;49:175-198. doi:10.1146/annurev.pharmtox.011008.145557
8. Mann JJ, Waternaux C, Haas GL, Malone KM. Toward a clinical model of suicidal behavior in psychiatric patients. Am J Psychiatry. 1999;156(2):181-189. doi:10.1176/ajp.156.2.181
9. Baldessarini RJ, Tondo L, Davis P, Pompili M, Goodwin FK, Hennen J. Decreased risk of suicides and attempts during long-term lithium treatment: a meta-analytic review [published correction appears in Bipolar Disord. 2007 May;9(3):314]. Bipolar Disord. 2006;8(5 Pt 2):625-639. doi:10.1111/j.1399-5618.2006.00344.x
10. Yatham LN, Kennedy SH, Parikh SV, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) 2018 guidelines for the management of patients with bipolar disorder. Bipolar Disord. 2018;20(2):97-170. doi:10.1111/bdi.12609
11. Stein G, Bernadt M. Lithium augmentation therapy in tricyclic-resistant depression. A controlled trial using lithium in low and normal doses. Br J Psychiatry. 1993;162:634-640. doi:10.1192/bjp.162.5.634
12. Bauer M, Bschor T, Kunz D, Berghöfer A, Ströhle A, Müller-Oerlinghausen B. Double-blind, placebo-controlled trial of the use of lithium to augment antidepressant medication in continuation treatment of unipolar major depression. Am J Psychiatry. 2000;157(9):1429-1435. doi:10.1176/appi.ajp.157.9.1429
13. Smith EG, Austin KL, Kim HM, et al. Suicide risk in Veterans Health Administration patients with mental health diagnoses initiating lithium or valproate: a historical prospective cohort study. BMC Psychiatry. 2014;14:357. Published 2014 Dec 17. doi:10.1186/s12888-014-0357-x
14. Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606-613. doi:10.1046/j.1525-1497.2001.016009606.x
15. US Department of Veterans Affairs, Veterans Health Administration. Use of patient record flags to identify patients at high risk for suicide. VHA Directive 2008-036. Published July 18, 2008. Accessed February 7, 2022. www.va.gov/vhapublications/ViewPublication.asp?pub_ID=1719
16. Sylvia LG, Reilly-Harrington NA, Leon AC, et al. Medication adherence in a comparative effectiveness trial for bipolar disorder. Acta Psychiatr Scand. 2014;129(5):359-365. doi:10.1111/acps.12202
17. Yerevanian BI, Koek RJ, Mintz J. Bipolar pharmacotherapy and suicidal behavior. Part I: Lithium, divalproex and carbamazepine. J Affect Disord. 2007;103(1-3):5-11. doi:10.1016/j.jad.2007.05.019
18. Post RM. The New News about Lithium: An Underutilized Treatment in the United States. Neuropsychopharmacology. 2018;43(5):1174-1179. doi:10.1038/npp.2017.238
19. Öhlund L, Ott M, Oja S, et al. Reasons for lithium discontinuation in men and women with bipolar disorder: a retrospective cohort study [published correction appears in BMC Psychiatry. 2018 Oct 3;18(1):322]. BMC Psychiatry. 2018;18(1):37. Published 2018 Feb 7. doi:10.1186/s12888-018-1622-1
20. Sajatovic M, Valenstein M, Blow F, Ganoczy D, Ignacio R. Treatment adherence with lithium and anticonvulsant medications among patients with bipolar disorder. Psychiatr Serv. 2007;58(6):855-863. doi:10.1176/ps.2007.58.6.855
21. Chakrabarti S. Treatment-adherence in bipolar disorder: A patient-centred approach. World J Psychiatry. 2016;6(4):399-409. Published 2016 Dec 22. doi:10.5498/wjp.v6.i4.399
22. Gaudiano BA, Weinstock LM, Miller IW. Improving treatment adherence in bipolar disorder: a review of current psychosocial treatment efficacy and recommendations for future treatment development. Behav Modif. 2008;32(3):267-301. doi:10.1177/0145445507309023
23. Peet M, Harvey NS. Lithium maintenance: 1. A standard education programme for patients. Br J Psychiatry. 1991;158:197-200. doi:10.1192/bjp.158.2.197
24. Cipriani A, Hawton K, Stockton S, Geddes JR. Lithium in the prevention of suicide in mood disorders: updated systematic review and meta-analysis. BMJ. 2013;346:f3646. doi:10.1136/bmj.f3646
Suicide is the tenth leading cause of death in the United States claiming nearly 48,000 individuals in 2019 and is the second leading cause of death among individuals aged 10 to 34 years.1 From 1999 to 2019, the suicide rate increased by 33%.1 In a retrospective study evaluating suicide risk in > 29,000 men, veterans had a greater risk for suicide in all age groups except for the oldest when compared with nonveterans.2 Another study of > 800,000 veterans found that younger veterans were most at risk for suicide.3 Veterans with completed suicides have a high incidence of affective disorders comorbid with substance use disorders, and therefore it is imperative to optimally treat these conditions to address suicidality.4 Additionally, a retrospective case-control study of veterans who died by suicide matched to controls identified that the cases had significantly higher rates of mental health conditions and suicidal ideation. Given that the veteran population is at higher risk of suicide, research of treatments to address suicidal ideation in veterans is needed.5
Lithium and Antisuicidal Properties
Lithium is the oldest treatment for bipolar disorder and is a long-standing first-line option due to its well-established efficacy as a mood stabilizer.6 Lithium’s antisuicidal properties separate it from the other pharmacologic options for bipolar disorder. A possible explanation for lithium’s unique antisuicidal properties is that these effects are mediated by its impact on aggression and impulsivity, which are both linked to an increased suicide risk.7,8 A meta-analysis by Baldessarini and colleagues demonstrated that patients with mood disorders who were prescribed lithium had a 5 times lower risk of suicide and attempts than did those not treated with lithium.9 Lithium’s current place in therapy is in the treatment of bipolar disorder and major depressive disorder augmentation.10-12Smith and colleagues found that in a cohort study of 21,194 veterans diagnosed with mental health conditions and initiated on lithium or valproate, there were no significant differences in associations with suicide observed between these agents over 365 days; however, there was a significant increased risk of suicide among patients discontinuing or modifying lithium within the first 180 days of treatment.13
Currently, lithium is thought to be underutilized at the US Department of Veterans Affairs (VA) Michael E. DeBakey VA Medical Center (MEDVAMC) in Houston, Texas, based on the number of prescriptions of lithium in the large population of veterans seen by mental health clinicians. MEDVAMC is a 538-bed academic teaching hospital serving approximately 130,000 veterans in southeast Texas. The Mental Health Care Line has 73 inpatient beds and an outpatient clinic serving > 12,000 patients annually. By retrospectively evaluating changes in suicidality in a sample of veterans prescribed lithium, we may be able to better understand the role that lithium plays in a population that has a higher suicide rate than does the general population. The primary objective of this study was to evaluate the change in number of suicide attempts from 3 months prior to lithium initiation to 3 months following a 6-month duration of lithium use. The secondary objective was to determine the change in suicidal ideation from the period prior to lithium use to the period following 6 months of lithium use.
Methods
This was a single-site, retrospective chart review conducted between October 2017 and April 2018. Prior to data collection, the MEDVAMC Research and Development committee approved the study as quality assurance research. Patients with an active lithium prescription were identified using the VA Lithium Lab Monitoring Dashboard, which includes all patients on lithium, their lithium level, and other data such as upcoming appointments.
Inclusion criteria consisted of adults who were aged ≥ 18 years, had an active lithium prescription on the date of data extraction, and had an active lithium prescription for at least 6 months. Patients were excluded if they had < 3 months of data before and/or after lithium was used for 6 months, and if they were initiated on lithium outside MEDVAMC. Cumulatively, patients had to have at least 12 months of data: 3 months prior to lithium use, at least 6 months of lithium use, and 9 months after lithium initiation.
Suicide Attempt and Suicidal Ideation Identification
When determining the number of suicide attempts, we recorded 4 data points: Veterans Crisis Line notes documenting suicide attempts, hospital admissions for suicide attempts, suicide behavior reports within the indicated time frame, and mental health progress notes documenting suicide attempts. Suicidal ideation was measured in 4 ways. First, we looked at the percentage of outpatient mental health progress notes documenting suicidal ideation. Second, using the Patient Health Questionnaire-9 (PHQ-9) depression assessments, we looked at the percentage of patients that indicated several days, more than half the days, or nearly every day to the question, “Thoughts that you would be better off dead or of hurting yourself in some way.”14 Third, we recorded the percentage of suicide risk assessments that patients responded yes to both questions on current preoccupation with suicidal thoughts and serious intent and plan to commit suicide with access to guns, stashed pills, or other means. Finally, we noted the percentage of suicide risk assessments where the assessment of risk was moderate or high.
A retrospective electronic health record (EHR) review was performed and the following information was obtained: patient demographics, lithium refill history, concomitant psychotropic medications and psychotherapy, lithium levels, comorbidities at lithium initiation, presence of a high-risk suicide flag in the EHR, suicide risk assessments, suicide behavior reports, Veteran Crisis Line notes, PHQ-9 assessments, and hospital admission and mental health outpatient notes. The lithium therapeutic range of 0.6-1.2 mmol/L is indicated for bipolar disorder and not other indications where the dose is typically titrated to effect rather than level. Medication possession ratio (MPR) was also calculated for lithium (sum of days’ supply for all fills in period ÷ number of days in period). A high-risk suicide flag alerts clinicians and staff that a mental health professional considers the veteran at risk for suicide.15 Statistical analysis was performed using the paired t test for means to assess proportional differences between variables for the primary and secondary outcomes. Descriptive statistics were used to describe the baseline characteristics.
Results
A total of 214 patients with an active prescription for lithium were identified on the Lithium Lab Monitoring Dashboard on October 31, 2017. After exclusion criteria were applied, 98 patients were included in the study (Figure 1). The 2 most common reasons for exclusion were due to patients not being on lithium for at least 6 months and being initiated on lithium at an outside facility. One patient was enrolled in a lithium research study (the medication ordered was lithium/placebo) and another patient refused all psychotropic medications according to the progress notes.
Most of the 98 patients (82.7%) were male with average age 50.5 years (Table 1). Almost half the patients (n = 47) were concomitantly participating in psychotherapy, and 50 (51.0%) patients received at least 1 antipsychotic medication. Twenty-nine patients had an active prescription for an additional mood stabilizer, and only 4 (4.1%) patients received lithium as monotherapy. Only 75 (76.5%) patients had a lithium level drawn during the 6 months of therapy, with 28 (37.3%) patients having a therapeutic lithium level (0.6 - 1.2 mmol/L). Seventy-one patients (72.4% ) were adherent to lithium therapy with a MPR > 0.8.16 Participants had 13 different psychiatric diagnoses at the time of lithium initiation; the most common were bipolar spectrum disorder (n = 38; 38.8%), depressive disorder (n = 27; 27.6%), and posttraumatic stress disorder (PTSD) (n = 26; 26.5%). Of note, 5 patients had a diagnosis of only PTSD without a concomitant mood disorder.
For the primary outcome, hospitalization for a suspected suicide attempt decreased from 4 (4.1%) before lithium use with a mean (SD) 0.04 (0.20) attempts per person to none within 3 months after lithium use for 6 months (t(97) = 2.03, P = .045) (Figure 2). The secondary outcome of hospitalization for suicidal ideations also decreased from 13 (13.3%) before lithium use with a mean (SD) 0.1 (0.3) ideations per person to 1 (1.0%) within 3 months after lithium use for 6 months with a mean (SD) 0.01 (0.1) ideations per person (t(97) = 3.68, P = .0004). Veteran Crisis Line calls also decreased from 4 (4.1%) with a mean (SD) 0.04 (0.2) calls per person to 1 (1.0%) within 3 months after 6 months of lithium with a mean (SD) 0.01 (0.1) calls per person (t(97) = 1.75, P = .08). The comparison of metrics from 3 months before lithium initiation and within 3 months after use saw decreases in all categories (Table 2). Outpatient notes documenting suicidal ideation decreased, as did the number of patients with a high-risk suicide flag.
Discussion
The results of this study suggest lithium may have a role in reducing suicidality in a veteran population. There was a statistically significant reduction in hospitalizations for suicide attempt and suicidal ideation after at least 6 months of lithium use. These results are comparable with a previously published study that observed significant decreases in suicidal behavior and/or hospitalization risks among veterans taking lithium compared with those not taking lithium.17 Our study was similar in respect to the reduced hospitalizations among a veteran population; however, the previous study did not report a difference in suicide attempts and lithium use. This could be related to the longer follow-up time in the previous study (3 years) vs our study (9 months).
Our study identified a significant reduction in Veteran Crisis Line calls after at least 6 months of lithium use. While a reduction in suicidal ideations could be implicated in the decrease in crisis line calls, there may be a confounding variable. It is possible that after lithium initiation, veterans had more frequent contact with health care practitioners due to laboratory test monitoring and follow-up visits and thus had concerns/crises addressed during these interactions ultimately leading to a decreased utilization of the crisis line. Interestingly, there was a reduction in mental health outpatient notes from the prelithium period to the 3-month period that followed 6 months of lithium therapy. However, our study did not report on the number of mental health outpatient notes or visits during the 6-month lithium duration. Additionally, time of year/season could have an impact on suicidality, but this relationship was not evaluated in this study.
The presence of high-risk suicide flags also decreased from the prelithium period to the period 3 months following 6 months of lithium use. High-risk flags are reviewed by the suicide prevention coordinators and mental health professionals every 90 days; therefore, the patients with flags had multiple opportunities for review and thus renewal or discontinuation during the study period. A similar rationale can be applied to the high-risk flag as with the Veteran Crisis Line reduction, although this change could also be representative of a decrease in suicidality. Our study is different from other lithium studies because it included patients with a multitude of psychiatric diagnoses rather than just mood disorders. Five of the patients had a diagnosis of only PTSD and no documented mood disorder at the time of lithium initiation. Additional research is needed on the impact of lithium on suicidality in veterans with PTSD and psychiatric conditions other than mood disorders.
Underutilization of Lithium
Despite widespread knowledge of lithium’s antisuicidal effects, it is underutilized as a mood stabilizer in the US.18 There are various modifiable barriers that impact the prescribing as well as use of lithium. Clinicians may not be fully aware of lithium’s antisuicidal properties and may also have a low level of confidence in patients’ likelihood of adherence to laboratory monitoring.18,19 Due to the narrow therapeutic index of lithium, the consequences of nonadherence to monitoring can be dangerous, which may deter mental health professionals from prescribing this antisuicidal agent. At MEDVAMC, only 72.4% of patients with a lithium prescription had a lithium level drawn within a 6-month period. This could be attributed to patient nonadherence (eg, the test was ordered but the patient did not go) or clinician nonadherence (eg, test was not ordered).
With increased clinician education as well as clinics dedicated to lithium management that allow for closer follow-up, facilities may see an increased level of comfort with lithium use. Lithium management clinics that provide close follow-up may also help address patient-related concerns about adverse effects and allow for close monitoring. To facilitate lithium monitoring at MEDVAMC, mental health practitioners and pharmacists developed a lithium test monitoring menu that serves as a “one-stop shop” for lithium baseline and ongoing test results.
In the future, we may study the impact of this test monitoring menu on lithium prescribing. One may also consider whether lithium levels need to be monitored at different frequencies (eg, less frequently for depression than bipolar disorder) depending on the diagnoses. A better understanding of the necessity for therapeutic monitoring may potentially reduce barriers to prescribing for patients who do not have indications that have a recommended therapeutic range (eg, bipolar disorder).
Lithium Adherence
A primary patient-related concern for low lithium utilization is poor adherence. In this sample, 71 patients (72.4%) were considered fully adherent. This was higher than the rate of 54.1% reported by Sajatovic and colleagues in a study evaluating adherence to lithium and other anticonvulsants in veterans with bipolar disorder.20 Patients’ beliefs about medications and overall health as well as knowledge of the illness and treatment may impact adherence.21 The literature indicates that strategies such as cognitive behavioral therapy (CBT) and didactic lectures positively impact patients’ attitudes about lithium, which ultimately influences adherence.21-23 Involving a family member or significant other in psychotherapy may also improve lithium adherence.21 Specifically in the VA, to address knowledge deficits and improve overall adherence, the Lithium Lab Monitoring Dashboard could be used to identify and invite new lithium starts to educational groups about lithium. These groups could also serve as lithium management clinics.
Limitations
There were several limitations to this study. This was a single-site, retrospective chart review with a small sample size. We studied a cross-section of veterans with only active prescriptions, which limited the sample size. The results should be interpreted cautiously because < 40% of patients who had a level drawn were in the therapeutic range. Patients whose lithium levels were outside of the therapeutic range may have not been fully adherent to the medication. Further analysis based on reason for lithium prescription (eg, bipolar disorder vs depression vs aggression/impulsivity in PTSD) may be helpful in better understanding the results.
Additionally, while we collected data on concomitant mood stabilizers and antipsychotics, we did not collect data on concurrent antidepressant therapy and only 4% of patients were on lithium monotherapy. Data regarding veterans undergoing concurrent CBT during their lithium trial were not assessed in this study and could be considered a confounding factor for future studies. We included any Veteran Crisis Line call in our results regardless of the reason for the call, which could have led to overreporting of this suicidality marker.
Given its small sample size, this study should be considered as hypothesis-generating. Further studies are needed to address lithium’s antisuicidal effects in specific diagnoses (eg, PTSD, anxiety, schizoaffective disorder) to better understand its place in therapy. Studies evaluating the relationship between dosing and suicidality may help provide insight into whether the antisuicidal effect of lithium is dose-dependent and whether a specific dose range rather than a therapeutic level should be targeted for antisuicidal purposes.
Conclusions
People treated for an affective disorder have a 30-times greater risk of suicide than do those in the general population; however, as lithium can reduce the risk of suicide and self-harm, it should continue to have an important role in clinical practice.24 At MEDVAMC, we observed a statistically significant reduction in hospitalization for suicide attempts and suicidal ideation in veterans prescribed lithium following nonfatal suicide behavior and suicidal ideation. Prospective randomized placebo-controlled studies are needed to better understand lithium’s antisuicidal effects.
Suicide is the tenth leading cause of death in the United States claiming nearly 48,000 individuals in 2019 and is the second leading cause of death among individuals aged 10 to 34 years.1 From 1999 to 2019, the suicide rate increased by 33%.1 In a retrospective study evaluating suicide risk in > 29,000 men, veterans had a greater risk for suicide in all age groups except for the oldest when compared with nonveterans.2 Another study of > 800,000 veterans found that younger veterans were most at risk for suicide.3 Veterans with completed suicides have a high incidence of affective disorders comorbid with substance use disorders, and therefore it is imperative to optimally treat these conditions to address suicidality.4 Additionally, a retrospective case-control study of veterans who died by suicide matched to controls identified that the cases had significantly higher rates of mental health conditions and suicidal ideation. Given that the veteran population is at higher risk of suicide, research of treatments to address suicidal ideation in veterans is needed.5
Lithium and Antisuicidal Properties
Lithium is the oldest treatment for bipolar disorder and is a long-standing first-line option due to its well-established efficacy as a mood stabilizer.6 Lithium’s antisuicidal properties separate it from the other pharmacologic options for bipolar disorder. A possible explanation for lithium’s unique antisuicidal properties is that these effects are mediated by its impact on aggression and impulsivity, which are both linked to an increased suicide risk.7,8 A meta-analysis by Baldessarini and colleagues demonstrated that patients with mood disorders who were prescribed lithium had a 5 times lower risk of suicide and attempts than did those not treated with lithium.9 Lithium’s current place in therapy is in the treatment of bipolar disorder and major depressive disorder augmentation.10-12Smith and colleagues found that in a cohort study of 21,194 veterans diagnosed with mental health conditions and initiated on lithium or valproate, there were no significant differences in associations with suicide observed between these agents over 365 days; however, there was a significant increased risk of suicide among patients discontinuing or modifying lithium within the first 180 days of treatment.13
Currently, lithium is thought to be underutilized at the US Department of Veterans Affairs (VA) Michael E. DeBakey VA Medical Center (MEDVAMC) in Houston, Texas, based on the number of prescriptions of lithium in the large population of veterans seen by mental health clinicians. MEDVAMC is a 538-bed academic teaching hospital serving approximately 130,000 veterans in southeast Texas. The Mental Health Care Line has 73 inpatient beds and an outpatient clinic serving > 12,000 patients annually. By retrospectively evaluating changes in suicidality in a sample of veterans prescribed lithium, we may be able to better understand the role that lithium plays in a population that has a higher suicide rate than does the general population. The primary objective of this study was to evaluate the change in number of suicide attempts from 3 months prior to lithium initiation to 3 months following a 6-month duration of lithium use. The secondary objective was to determine the change in suicidal ideation from the period prior to lithium use to the period following 6 months of lithium use.
Methods
This was a single-site, retrospective chart review conducted between October 2017 and April 2018. Prior to data collection, the MEDVAMC Research and Development committee approved the study as quality assurance research. Patients with an active lithium prescription were identified using the VA Lithium Lab Monitoring Dashboard, which includes all patients on lithium, their lithium level, and other data such as upcoming appointments.
Inclusion criteria consisted of adults who were aged ≥ 18 years, had an active lithium prescription on the date of data extraction, and had an active lithium prescription for at least 6 months. Patients were excluded if they had < 3 months of data before and/or after lithium was used for 6 months, and if they were initiated on lithium outside MEDVAMC. Cumulatively, patients had to have at least 12 months of data: 3 months prior to lithium use, at least 6 months of lithium use, and 9 months after lithium initiation.
Suicide Attempt and Suicidal Ideation Identification
When determining the number of suicide attempts, we recorded 4 data points: Veterans Crisis Line notes documenting suicide attempts, hospital admissions for suicide attempts, suicide behavior reports within the indicated time frame, and mental health progress notes documenting suicide attempts. Suicidal ideation was measured in 4 ways. First, we looked at the percentage of outpatient mental health progress notes documenting suicidal ideation. Second, using the Patient Health Questionnaire-9 (PHQ-9) depression assessments, we looked at the percentage of patients that indicated several days, more than half the days, or nearly every day to the question, “Thoughts that you would be better off dead or of hurting yourself in some way.”14 Third, we recorded the percentage of suicide risk assessments that patients responded yes to both questions on current preoccupation with suicidal thoughts and serious intent and plan to commit suicide with access to guns, stashed pills, or other means. Finally, we noted the percentage of suicide risk assessments where the assessment of risk was moderate or high.
A retrospective electronic health record (EHR) review was performed and the following information was obtained: patient demographics, lithium refill history, concomitant psychotropic medications and psychotherapy, lithium levels, comorbidities at lithium initiation, presence of a high-risk suicide flag in the EHR, suicide risk assessments, suicide behavior reports, Veteran Crisis Line notes, PHQ-9 assessments, and hospital admission and mental health outpatient notes. The lithium therapeutic range of 0.6-1.2 mmol/L is indicated for bipolar disorder and not other indications where the dose is typically titrated to effect rather than level. Medication possession ratio (MPR) was also calculated for lithium (sum of days’ supply for all fills in period ÷ number of days in period). A high-risk suicide flag alerts clinicians and staff that a mental health professional considers the veteran at risk for suicide.15 Statistical analysis was performed using the paired t test for means to assess proportional differences between variables for the primary and secondary outcomes. Descriptive statistics were used to describe the baseline characteristics.
Results
A total of 214 patients with an active prescription for lithium were identified on the Lithium Lab Monitoring Dashboard on October 31, 2017. After exclusion criteria were applied, 98 patients were included in the study (Figure 1). The 2 most common reasons for exclusion were due to patients not being on lithium for at least 6 months and being initiated on lithium at an outside facility. One patient was enrolled in a lithium research study (the medication ordered was lithium/placebo) and another patient refused all psychotropic medications according to the progress notes.
Most of the 98 patients (82.7%) were male with average age 50.5 years (Table 1). Almost half the patients (n = 47) were concomitantly participating in psychotherapy, and 50 (51.0%) patients received at least 1 antipsychotic medication. Twenty-nine patients had an active prescription for an additional mood stabilizer, and only 4 (4.1%) patients received lithium as monotherapy. Only 75 (76.5%) patients had a lithium level drawn during the 6 months of therapy, with 28 (37.3%) patients having a therapeutic lithium level (0.6 - 1.2 mmol/L). Seventy-one patients (72.4% ) were adherent to lithium therapy with a MPR > 0.8.16 Participants had 13 different psychiatric diagnoses at the time of lithium initiation; the most common were bipolar spectrum disorder (n = 38; 38.8%), depressive disorder (n = 27; 27.6%), and posttraumatic stress disorder (PTSD) (n = 26; 26.5%). Of note, 5 patients had a diagnosis of only PTSD without a concomitant mood disorder.
For the primary outcome, hospitalization for a suspected suicide attempt decreased from 4 (4.1%) before lithium use with a mean (SD) 0.04 (0.20) attempts per person to none within 3 months after lithium use for 6 months (t(97) = 2.03, P = .045) (Figure 2). The secondary outcome of hospitalization for suicidal ideations also decreased from 13 (13.3%) before lithium use with a mean (SD) 0.1 (0.3) ideations per person to 1 (1.0%) within 3 months after lithium use for 6 months with a mean (SD) 0.01 (0.1) ideations per person (t(97) = 3.68, P = .0004). Veteran Crisis Line calls also decreased from 4 (4.1%) with a mean (SD) 0.04 (0.2) calls per person to 1 (1.0%) within 3 months after 6 months of lithium with a mean (SD) 0.01 (0.1) calls per person (t(97) = 1.75, P = .08). The comparison of metrics from 3 months before lithium initiation and within 3 months after use saw decreases in all categories (Table 2). Outpatient notes documenting suicidal ideation decreased, as did the number of patients with a high-risk suicide flag.
Discussion
The results of this study suggest lithium may have a role in reducing suicidality in a veteran population. There was a statistically significant reduction in hospitalizations for suicide attempt and suicidal ideation after at least 6 months of lithium use. These results are comparable with a previously published study that observed significant decreases in suicidal behavior and/or hospitalization risks among veterans taking lithium compared with those not taking lithium.17 Our study was similar in respect to the reduced hospitalizations among a veteran population; however, the previous study did not report a difference in suicide attempts and lithium use. This could be related to the longer follow-up time in the previous study (3 years) vs our study (9 months).
Our study identified a significant reduction in Veteran Crisis Line calls after at least 6 months of lithium use. While a reduction in suicidal ideations could be implicated in the decrease in crisis line calls, there may be a confounding variable. It is possible that after lithium initiation, veterans had more frequent contact with health care practitioners due to laboratory test monitoring and follow-up visits and thus had concerns/crises addressed during these interactions ultimately leading to a decreased utilization of the crisis line. Interestingly, there was a reduction in mental health outpatient notes from the prelithium period to the 3-month period that followed 6 months of lithium therapy. However, our study did not report on the number of mental health outpatient notes or visits during the 6-month lithium duration. Additionally, time of year/season could have an impact on suicidality, but this relationship was not evaluated in this study.
The presence of high-risk suicide flags also decreased from the prelithium period to the period 3 months following 6 months of lithium use. High-risk flags are reviewed by the suicide prevention coordinators and mental health professionals every 90 days; therefore, the patients with flags had multiple opportunities for review and thus renewal or discontinuation during the study period. A similar rationale can be applied to the high-risk flag as with the Veteran Crisis Line reduction, although this change could also be representative of a decrease in suicidality. Our study is different from other lithium studies because it included patients with a multitude of psychiatric diagnoses rather than just mood disorders. Five of the patients had a diagnosis of only PTSD and no documented mood disorder at the time of lithium initiation. Additional research is needed on the impact of lithium on suicidality in veterans with PTSD and psychiatric conditions other than mood disorders.
Underutilization of Lithium
Despite widespread knowledge of lithium’s antisuicidal effects, it is underutilized as a mood stabilizer in the US.18 There are various modifiable barriers that impact the prescribing as well as use of lithium. Clinicians may not be fully aware of lithium’s antisuicidal properties and may also have a low level of confidence in patients’ likelihood of adherence to laboratory monitoring.18,19 Due to the narrow therapeutic index of lithium, the consequences of nonadherence to monitoring can be dangerous, which may deter mental health professionals from prescribing this antisuicidal agent. At MEDVAMC, only 72.4% of patients with a lithium prescription had a lithium level drawn within a 6-month period. This could be attributed to patient nonadherence (eg, the test was ordered but the patient did not go) or clinician nonadherence (eg, test was not ordered).
With increased clinician education as well as clinics dedicated to lithium management that allow for closer follow-up, facilities may see an increased level of comfort with lithium use. Lithium management clinics that provide close follow-up may also help address patient-related concerns about adverse effects and allow for close monitoring. To facilitate lithium monitoring at MEDVAMC, mental health practitioners and pharmacists developed a lithium test monitoring menu that serves as a “one-stop shop” for lithium baseline and ongoing test results.
In the future, we may study the impact of this test monitoring menu on lithium prescribing. One may also consider whether lithium levels need to be monitored at different frequencies (eg, less frequently for depression than bipolar disorder) depending on the diagnoses. A better understanding of the necessity for therapeutic monitoring may potentially reduce barriers to prescribing for patients who do not have indications that have a recommended therapeutic range (eg, bipolar disorder).
Lithium Adherence
A primary patient-related concern for low lithium utilization is poor adherence. In this sample, 71 patients (72.4%) were considered fully adherent. This was higher than the rate of 54.1% reported by Sajatovic and colleagues in a study evaluating adherence to lithium and other anticonvulsants in veterans with bipolar disorder.20 Patients’ beliefs about medications and overall health as well as knowledge of the illness and treatment may impact adherence.21 The literature indicates that strategies such as cognitive behavioral therapy (CBT) and didactic lectures positively impact patients’ attitudes about lithium, which ultimately influences adherence.21-23 Involving a family member or significant other in psychotherapy may also improve lithium adherence.21 Specifically in the VA, to address knowledge deficits and improve overall adherence, the Lithium Lab Monitoring Dashboard could be used to identify and invite new lithium starts to educational groups about lithium. These groups could also serve as lithium management clinics.
Limitations
There were several limitations to this study. This was a single-site, retrospective chart review with a small sample size. We studied a cross-section of veterans with only active prescriptions, which limited the sample size. The results should be interpreted cautiously because < 40% of patients who had a level drawn were in the therapeutic range. Patients whose lithium levels were outside of the therapeutic range may have not been fully adherent to the medication. Further analysis based on reason for lithium prescription (eg, bipolar disorder vs depression vs aggression/impulsivity in PTSD) may be helpful in better understanding the results.
Additionally, while we collected data on concomitant mood stabilizers and antipsychotics, we did not collect data on concurrent antidepressant therapy and only 4% of patients were on lithium monotherapy. Data regarding veterans undergoing concurrent CBT during their lithium trial were not assessed in this study and could be considered a confounding factor for future studies. We included any Veteran Crisis Line call in our results regardless of the reason for the call, which could have led to overreporting of this suicidality marker.
Given its small sample size, this study should be considered as hypothesis-generating. Further studies are needed to address lithium’s antisuicidal effects in specific diagnoses (eg, PTSD, anxiety, schizoaffective disorder) to better understand its place in therapy. Studies evaluating the relationship between dosing and suicidality may help provide insight into whether the antisuicidal effect of lithium is dose-dependent and whether a specific dose range rather than a therapeutic level should be targeted for antisuicidal purposes.
Conclusions
People treated for an affective disorder have a 30-times greater risk of suicide than do those in the general population; however, as lithium can reduce the risk of suicide and self-harm, it should continue to have an important role in clinical practice.24 At MEDVAMC, we observed a statistically significant reduction in hospitalization for suicide attempts and suicidal ideation in veterans prescribed lithium following nonfatal suicide behavior and suicidal ideation. Prospective randomized placebo-controlled studies are needed to better understand lithium’s antisuicidal effects.
1. Centers for Disease Control and Prevention. Preventing Suicide Fact Sheet. Updated April 2021. Accessed February 16, 2022. https://www.cdc.gov/suicide/pdf/preventing-suicide-factsheet-2021-508.pdf
2. Kaplan MS, McFarland BH, Huguet N, Valenstein M. Suicide risk and precipitating circumstances among young, middle-aged, and older male veterans. Am J Public Health. 2012;102 Suppl 1(Suppl 1):S131-S137. doi:10.2105/AJPH.2011.300445
3. Zivin K, Kim HM, McCarthy JF, et al. Suicide mortality among individuals receiving treatment for depression in the Veterans Affairs health system: associations with patient and treatment setting characteristics. Am J Public Health. 2007;97(12):2193-2198. doi:10.2105/AJPH.2007.115477
4. Lehmann L, McCormick RA, McCracken L. Suicidal behavior among patients in the VA health care system. Psychiatr Serv. 1995;46(10):1069-1071. doi:10.1176/ps.46.10.1069
5. Dobscha SK, Denneson LM, Kovas AE, et al. Correlates of suicide among veterans treated in primary care: case-control study of a nationally representative sample. J Gen Intern Med. 2014;29(suppl 4):853-860. doi:10.1007/s11606-014-3028-1
6. Malhi GS, Tanious M, Das P, Coulston CM, Berk M. Potential mechanisms of action of lithium in bipolar disorder. Current understanding. CNS Drugs. 2013;27(2):135-153. doi:10.1007/s40263-013-0039-0
7. Kovacsics CE, Gottesman II, Gould TD. Lithium’s antisuicidal efficacy: elucidation of neurobiological targets using endophenotype strategies. Annu Rev Pharmacol Toxicol. 2009;49:175-198. doi:10.1146/annurev.pharmtox.011008.145557
8. Mann JJ, Waternaux C, Haas GL, Malone KM. Toward a clinical model of suicidal behavior in psychiatric patients. Am J Psychiatry. 1999;156(2):181-189. doi:10.1176/ajp.156.2.181
9. Baldessarini RJ, Tondo L, Davis P, Pompili M, Goodwin FK, Hennen J. Decreased risk of suicides and attempts during long-term lithium treatment: a meta-analytic review [published correction appears in Bipolar Disord. 2007 May;9(3):314]. Bipolar Disord. 2006;8(5 Pt 2):625-639. doi:10.1111/j.1399-5618.2006.00344.x
10. Yatham LN, Kennedy SH, Parikh SV, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) 2018 guidelines for the management of patients with bipolar disorder. Bipolar Disord. 2018;20(2):97-170. doi:10.1111/bdi.12609
11. Stein G, Bernadt M. Lithium augmentation therapy in tricyclic-resistant depression. A controlled trial using lithium in low and normal doses. Br J Psychiatry. 1993;162:634-640. doi:10.1192/bjp.162.5.634
12. Bauer M, Bschor T, Kunz D, Berghöfer A, Ströhle A, Müller-Oerlinghausen B. Double-blind, placebo-controlled trial of the use of lithium to augment antidepressant medication in continuation treatment of unipolar major depression. Am J Psychiatry. 2000;157(9):1429-1435. doi:10.1176/appi.ajp.157.9.1429
13. Smith EG, Austin KL, Kim HM, et al. Suicide risk in Veterans Health Administration patients with mental health diagnoses initiating lithium or valproate: a historical prospective cohort study. BMC Psychiatry. 2014;14:357. Published 2014 Dec 17. doi:10.1186/s12888-014-0357-x
14. Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606-613. doi:10.1046/j.1525-1497.2001.016009606.x
15. US Department of Veterans Affairs, Veterans Health Administration. Use of patient record flags to identify patients at high risk for suicide. VHA Directive 2008-036. Published July 18, 2008. Accessed February 7, 2022. www.va.gov/vhapublications/ViewPublication.asp?pub_ID=1719
16. Sylvia LG, Reilly-Harrington NA, Leon AC, et al. Medication adherence in a comparative effectiveness trial for bipolar disorder. Acta Psychiatr Scand. 2014;129(5):359-365. doi:10.1111/acps.12202
17. Yerevanian BI, Koek RJ, Mintz J. Bipolar pharmacotherapy and suicidal behavior. Part I: Lithium, divalproex and carbamazepine. J Affect Disord. 2007;103(1-3):5-11. doi:10.1016/j.jad.2007.05.019
18. Post RM. The New News about Lithium: An Underutilized Treatment in the United States. Neuropsychopharmacology. 2018;43(5):1174-1179. doi:10.1038/npp.2017.238
19. Öhlund L, Ott M, Oja S, et al. Reasons for lithium discontinuation in men and women with bipolar disorder: a retrospective cohort study [published correction appears in BMC Psychiatry. 2018 Oct 3;18(1):322]. BMC Psychiatry. 2018;18(1):37. Published 2018 Feb 7. doi:10.1186/s12888-018-1622-1
20. Sajatovic M, Valenstein M, Blow F, Ganoczy D, Ignacio R. Treatment adherence with lithium and anticonvulsant medications among patients with bipolar disorder. Psychiatr Serv. 2007;58(6):855-863. doi:10.1176/ps.2007.58.6.855
21. Chakrabarti S. Treatment-adherence in bipolar disorder: A patient-centred approach. World J Psychiatry. 2016;6(4):399-409. Published 2016 Dec 22. doi:10.5498/wjp.v6.i4.399
22. Gaudiano BA, Weinstock LM, Miller IW. Improving treatment adherence in bipolar disorder: a review of current psychosocial treatment efficacy and recommendations for future treatment development. Behav Modif. 2008;32(3):267-301. doi:10.1177/0145445507309023
23. Peet M, Harvey NS. Lithium maintenance: 1. A standard education programme for patients. Br J Psychiatry. 1991;158:197-200. doi:10.1192/bjp.158.2.197
24. Cipriani A, Hawton K, Stockton S, Geddes JR. Lithium in the prevention of suicide in mood disorders: updated systematic review and meta-analysis. BMJ. 2013;346:f3646. doi:10.1136/bmj.f3646
1. Centers for Disease Control and Prevention. Preventing Suicide Fact Sheet. Updated April 2021. Accessed February 16, 2022. https://www.cdc.gov/suicide/pdf/preventing-suicide-factsheet-2021-508.pdf
2. Kaplan MS, McFarland BH, Huguet N, Valenstein M. Suicide risk and precipitating circumstances among young, middle-aged, and older male veterans. Am J Public Health. 2012;102 Suppl 1(Suppl 1):S131-S137. doi:10.2105/AJPH.2011.300445
3. Zivin K, Kim HM, McCarthy JF, et al. Suicide mortality among individuals receiving treatment for depression in the Veterans Affairs health system: associations with patient and treatment setting characteristics. Am J Public Health. 2007;97(12):2193-2198. doi:10.2105/AJPH.2007.115477
4. Lehmann L, McCormick RA, McCracken L. Suicidal behavior among patients in the VA health care system. Psychiatr Serv. 1995;46(10):1069-1071. doi:10.1176/ps.46.10.1069
5. Dobscha SK, Denneson LM, Kovas AE, et al. Correlates of suicide among veterans treated in primary care: case-control study of a nationally representative sample. J Gen Intern Med. 2014;29(suppl 4):853-860. doi:10.1007/s11606-014-3028-1
6. Malhi GS, Tanious M, Das P, Coulston CM, Berk M. Potential mechanisms of action of lithium in bipolar disorder. Current understanding. CNS Drugs. 2013;27(2):135-153. doi:10.1007/s40263-013-0039-0
7. Kovacsics CE, Gottesman II, Gould TD. Lithium’s antisuicidal efficacy: elucidation of neurobiological targets using endophenotype strategies. Annu Rev Pharmacol Toxicol. 2009;49:175-198. doi:10.1146/annurev.pharmtox.011008.145557
8. Mann JJ, Waternaux C, Haas GL, Malone KM. Toward a clinical model of suicidal behavior in psychiatric patients. Am J Psychiatry. 1999;156(2):181-189. doi:10.1176/ajp.156.2.181
9. Baldessarini RJ, Tondo L, Davis P, Pompili M, Goodwin FK, Hennen J. Decreased risk of suicides and attempts during long-term lithium treatment: a meta-analytic review [published correction appears in Bipolar Disord. 2007 May;9(3):314]. Bipolar Disord. 2006;8(5 Pt 2):625-639. doi:10.1111/j.1399-5618.2006.00344.x
10. Yatham LN, Kennedy SH, Parikh SV, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) 2018 guidelines for the management of patients with bipolar disorder. Bipolar Disord. 2018;20(2):97-170. doi:10.1111/bdi.12609
11. Stein G, Bernadt M. Lithium augmentation therapy in tricyclic-resistant depression. A controlled trial using lithium in low and normal doses. Br J Psychiatry. 1993;162:634-640. doi:10.1192/bjp.162.5.634
12. Bauer M, Bschor T, Kunz D, Berghöfer A, Ströhle A, Müller-Oerlinghausen B. Double-blind, placebo-controlled trial of the use of lithium to augment antidepressant medication in continuation treatment of unipolar major depression. Am J Psychiatry. 2000;157(9):1429-1435. doi:10.1176/appi.ajp.157.9.1429
13. Smith EG, Austin KL, Kim HM, et al. Suicide risk in Veterans Health Administration patients with mental health diagnoses initiating lithium or valproate: a historical prospective cohort study. BMC Psychiatry. 2014;14:357. Published 2014 Dec 17. doi:10.1186/s12888-014-0357-x
14. Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606-613. doi:10.1046/j.1525-1497.2001.016009606.x
15. US Department of Veterans Affairs, Veterans Health Administration. Use of patient record flags to identify patients at high risk for suicide. VHA Directive 2008-036. Published July 18, 2008. Accessed February 7, 2022. www.va.gov/vhapublications/ViewPublication.asp?pub_ID=1719
16. Sylvia LG, Reilly-Harrington NA, Leon AC, et al. Medication adherence in a comparative effectiveness trial for bipolar disorder. Acta Psychiatr Scand. 2014;129(5):359-365. doi:10.1111/acps.12202
17. Yerevanian BI, Koek RJ, Mintz J. Bipolar pharmacotherapy and suicidal behavior. Part I: Lithium, divalproex and carbamazepine. J Affect Disord. 2007;103(1-3):5-11. doi:10.1016/j.jad.2007.05.019
18. Post RM. The New News about Lithium: An Underutilized Treatment in the United States. Neuropsychopharmacology. 2018;43(5):1174-1179. doi:10.1038/npp.2017.238
19. Öhlund L, Ott M, Oja S, et al. Reasons for lithium discontinuation in men and women with bipolar disorder: a retrospective cohort study [published correction appears in BMC Psychiatry. 2018 Oct 3;18(1):322]. BMC Psychiatry. 2018;18(1):37. Published 2018 Feb 7. doi:10.1186/s12888-018-1622-1
20. Sajatovic M, Valenstein M, Blow F, Ganoczy D, Ignacio R. Treatment adherence with lithium and anticonvulsant medications among patients with bipolar disorder. Psychiatr Serv. 2007;58(6):855-863. doi:10.1176/ps.2007.58.6.855
21. Chakrabarti S. Treatment-adherence in bipolar disorder: A patient-centred approach. World J Psychiatry. 2016;6(4):399-409. Published 2016 Dec 22. doi:10.5498/wjp.v6.i4.399
22. Gaudiano BA, Weinstock LM, Miller IW. Improving treatment adherence in bipolar disorder: a review of current psychosocial treatment efficacy and recommendations for future treatment development. Behav Modif. 2008;32(3):267-301. doi:10.1177/0145445507309023
23. Peet M, Harvey NS. Lithium maintenance: 1. A standard education programme for patients. Br J Psychiatry. 1991;158:197-200. doi:10.1192/bjp.158.2.197
24. Cipriani A, Hawton K, Stockton S, Geddes JR. Lithium in the prevention of suicide in mood disorders: updated systematic review and meta-analysis. BMJ. 2013;346:f3646. doi:10.1136/bmj.f3646
Oral Lichen Planus Treated With Plasma Rich in Growth Factors
Lichen planus is a chronic inflammatory mucocutaneous disease that usually affects the skin and/or the genital and oral mucosae.1,2 This disease classically presents with clinical relapses or outbreaks that alternate with periods of remission or latency. Oral lichen planus (OLP) can present with or without extraoral manifestation. It sometimes is difficult to differentiate OLP from oral lichenoid reactions, which can be related to dental materials, some drugs, and systemic conditions or can be idiopathic.1,2
Oral lichen planus is one of the most common noninfectious diseases of the oral cavity, with a reported prevalence of 1% worldwide and marked geographical differences. In Europe, the prevalence of OLP ranges from 1% to 2%.3,4 It is more frequent in women (1.5:1 to 2:1) and usually appears in the fourth and fifth decades of life.1-4
The causes of OLP have not been entirely elucidated, but it is broadly accepted that there is a deregulation on different T lymphocytes that in turn causes effects on CD8 lymphocytes in response to an external noxa. This unknown “trigger” or starting factor also produces an impact on basal keratinocytes. Therefore, the pathogenesis of lichen planus is influenced by a series of cellular events mediated by different cytokines.2,5,6 Among these, tumor necrosis factor α and IL-1 are known to have important roles in the disease. More recently, other cytokines, such as IL-4, secreted by type 2 helper T cells, also have been related to the development and progression of the oral lesions.5,6 In addition to the factors that generate the onset of the disease, there are others that may precipitate clinical outbreaks. Different factors have been related to the progression of the disease, influencing the initiation, perpetuation, and/or worsening of OLP lesions.1,2 Exactly how these factors affect disease progression is another challenging question. The list of possible or potential factors related to disease progression is long; nonetheless, in the vast majority, a clear explanation at a molecular level has not been clearly demonstrated.2,5
Conventionally, 6 clinical presentations of OLP lesions divided into 2 main groups have been described in the oral cavity: white forms (reticular, papular, and plaquelike) and red forms (erythematous, atrophic-erosive, and bullous).1,7-9
Oral lichen planus mainly is treated with topically or systemically administered steroids based on the presence of symptoms such as pain and inability to perform daily activities (eg, eating, talking).5,10 The treatment of choice often is based on the professional’s experience, as there are no broadly accepted national or international clinical practice guidelines on steroid type, administration route, dose, vehicle for administration, or maintenance.11 Despite this lack of unified criteria, different topical and systemic steroid administration protocols allow a reduction in the symptoms or even the disappearance of the red lesions to be achieved in many cases. Unfortunately, there are many patients with lesions refractory to standard treatments for OLP.12 Several alternatives for these patients have been described in the literature, though on many occasions these alternatives present substantial side effects for the patient.13 The search for an effective treatment without side effects is still challenging. One of the treatments tested under this premise has been the application of plasma rich in growth factors (PRGF) by means of infiltration or topical application, in both cases obtaining good results without side effects.14
We sought to analyze the information from a case series of patients treated at the Eduardo Anitua Clinic (Vitoria-Gasteiz, Spain) and describe the results and follow-up of patients with erosive OLP refractory to standard therapy who have been successfully treated by local infiltration of PRGF as the only treatment.
Material and Methods
Patients—We included data from the database of the clinical center with de-identified information of patients with erosive OLP diagnosed clinically and histopathologically who did not respond to conventional treatment (ie, topical and/or systemic corticosteroids [depending on the case]) as well as patients who presented with extensive erosive OLP with systemic involvement and whose systemic treatment was not effective in resolving oral manifestations.
Therapies Administered and Evaluations—Lesions refractory to conventional corticosteroid protocols had been previously treated for 30 days with 0.5% triamcinolone acetonide mouth rinse followed by a cycle of 1% triamcinolone acetonide mouth rinse. Subsequently, a cycle of oral corticosteroids (prednisone for 30 days: 1 mg/kg/d in a single morning dose with staged reduction after the first week) had been administered. One dayafter the corticosteroid treatment was suspended, the patients were treated by PRGF-Endoret (BTI Biotechnology Institute) infiltration following the protocol described by Anitua et al.15,16
Before starting the infiltrations with PRGF, the patient had been asked to rate the pain level on a visual analog scale (VAS) of 1 to 10, with 10 being the most intense imaginable pain. Pain score was subsequently rated and registered during every visit. An initial photograph of the lesion also was obtained to establish a starting point for further comparisons of clinical evolution of the lesions.
Prior to each infiltration, the plasma was separated into 2 fractions. The second fraction was the one that corresponded to the highest number of platelets and included the 2 mL of plasma just above the white series (or buffy coat). This fraction of plasma was the one used to infiltrate the lesions.
Plasma rich in growth factors was activated just before infiltration. The activation was done by adding 10% calcium chloride. Once activated, it was infiltrated into the active lesion using a 31-G × 1/6-in hypodermic needle and a 2-mL Luer-lock syringe. Infiltrations were performed without anesthesia. Four punctures were made for each ulcerative lesion, dividing the lesion into 4 points: upper, lower, right, and left. Plasma rich in growth factors was infiltrated until a slight blanching was observed in the surrounding tissue. At that moment, the infiltration was stopped and was carried out in the next infiltration site.
One treatment session was performed per week, with follow-up 1 week after treatment. In the control visit, the state of the lesions was re-evaluated, and it was decided whether new infiltrations were needed. The treatment was finished when complete epithelialization of the lesion was visualized or the associated symptoms disappeared. At each visit, photographs were taken, and the patient assessed the severity of pain on the VAS.
Statistical Analysis—A Shapiro-Wilk test was carried out with the obtained data to check the normal distribution of the sample. The evolution of pain during the study was compared by paired t test. The qualitative variables were described by means of a frequency analysis. Quantitative variables were described by the mean and the SD. The data were analyzed with SPSS V15.0 for Windows (SPSS Inc). P<.05 showed statistical significance.
Results
A total of 15 patients were included in the study, all with atrophic-erosive lichen planus. Two patients were male, and 13 were female. The mean age (SD) of the patients included in the study was 55.27 (14.19) years. The mean number of outbreaks per year (SD) was 3.2 (1.7), with a range of 1 to 8 outbreaks.
Healing of OLP Lesions—The number of treatment sessions to achieve complete healing varied among the patients (Figures 1 and 2). Ten patients (66.7%) required a single session, 2 patients (13.3%) required 2 sessions, and 3 patients (20%) required 3 sessions. The mean time (SD) without lesions for the patients who required a single session was 10.9 (5.2) months (range, 6–24 months).
Pain Assessment—The mean (SD) score obtained on the VAS before treatment with PRGF was 8.27 (1.16); this score dropped to 1.27 (1.53) after the first treatment session and was a statistically significant difference (P=.006).
For those patients requiring more than 1 session, the mean (SD) pain scores decreased by 0.75 (0.97) points and 0 points after the first and second sessions of treatment, respectively. The mean (SD) amount of PRGF infiltrated in each patient in the first session was 2.60 (0.63) mL. In the second session, the mean (SD) amount was 1.2 (0.33) mL; these differences were statistically significant (P=.008). In the last session, the mean (SD) amount was 1.1 (0.22) mL.
Follow-up and Adverse Effects—The mean (SD) follow-up time was 47.16 (15.78) months. The patients were free of symptoms, and there were no adverse effects derived from the treatment during follow-up.
Comment
The primary goal of OLP treatment is to stop the outbreaks.1,9,13 The lack of potency of corticosteroids in some patients with OLP could be due in part to the inadequate selection of the vehicle (ointment/oral rinse) for the extension and characteristics of the lesion or because of an inappropriate prescription dose, time, and/or frequency, as described by González-Moles.17 However, even when using an appropriate protocol, some lesions are resistant to topical treatment and require other therapeutic modalities.1,9,13 Previously proposed topical treatments include different immunosuppressants, such as the mammalian target of rapamycin, tacrolimus ointment 0.1%, pimecrolimus cream 1%, or cyclosporine A (50–100 mg/mL) formulations.18 Nevertheless, these drugs seem to have a greater number of side effects than topical steroids, and tacrolimus has been associated with cases of oral malignancy after continuing treatment.15
Severe and/or recalcitrant lesions and extraoral involvement have been successfully treated with systemic prednisone (40–80 mg/d).1,9,13 Nevertheless, systemic corticosteroid toxicity requires that these treatments should be used only when necessary at the lowest possible dose and for the shortest possible duration.19 Other nonpharmacologic options for treatment are photodynamic, UV, and low-level laser therapy.20,21 They have been accepted as supplementary modalities in different inflammatory skin conditions but present important technical requirements. Their effectiveness in corticosteroid-resistant cases have not been definitively assessed. Interestingly, promising results recently have been reported by Bennardo et al22 when comparing the efficacy of autologous platelet concentrates with triamcinolone injection.
In our study, the use of PRGF stopped the lesions’ evolution since the first treatment session, reducing them by 6.5-fold. The positive effects observed may have been promoted by the activity of different proteins present in PRGF (eg, platelet-derived growth factor, vascular endothelial growth factor, transforming growth factor, epidermal growth factor, fibroblast growth factor, fibronectin). These molecules contribute to collagen synthesis; angiogenesis; endothelial cell migration and proliferation; or keratinocyte cell migration, proliferation, differentiation, growth, and migration—phenomena that are essential for healing and re-epithelialization.23-25
Different studies also have supported an anti-inflammatory effect of PRGF mediated by an inhibition of the transcription of nuclear factor–κB and the expression of cyclooxygenase-2 and chemokine receptor type 4 produced by its high content of hepatocyte growth factor or the reduction of inflammatory marker expression, such as intercellular adhesion molecule 1. The development of an efficient 3-dimensional fibrin scaffold formation that occurs after PRGF administration also could facilitate healing, helping some cell populations to guide their position and function.23-25
Limitations of our study include the small number of patients and the absence of a control group. The higher number of female patients in the study did not seem to affect the results, as differences related to gender have not been reported when treating patients with OLP with autologous platelet concentrates or other modalities of treatment.
Conclusion
Results from our study indicate that the use of PRGF could be a new treatment option for OLP cases refractory to conventional therapy. No complications were observed during the treatment procedure or during the complete follow-up period. Nonetheless, new prospective studies with a greater number of patients and longer follow-up periods are needed to confirm these preliminary results.
- Al-Hashimi I, Schifter M, Lockhart PB, et al. Oral lichen planus and oral lichenoid lesions: diagnostic and therapeutic considerations. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;103:1-12.
- Kurago ZB. Etiology and pathogenesis of oral lichen planus: an overview. Oral Surg Oral Med Oral Pathol Oral Radiol. 2016;122:72-80.
- McCartan BE, Healy CM. The reported prevalence of oral lichen planus: a review and critique. J Oral Pathol Med. 2008;37:447-453.
- González-Moles MÁ, Warnakulasuriya S, González-Ruiz I, et al. Worldwide prevalence of oral lichen planus: a systematic review and meta-analysis. Oral Dis. 2021;27:813-828.
- Nosratzehi T. Oral lichen planus: an overview of potential risk factors, biomarkers and treatments. Asian Pac J Cancer Prev. 2018;19:1161-1167.
- Mehrbani SP, Motahari P, Azar FP, et al. Role of interleukin-4 in pathogenesis of oral lichen planus: a systematic review. Med Oral Patol Oral Cir Bucal. 2020;25:E410-E415.
- Edwards PC, Kelsch R. Oral lichen planus: clinical presentation and management. J Can Dent Assoc. 2002;68:494-499.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826.
- Babu A, Chellaswamy S, Muthukumar S, et al. Bullous lichen planus: case report and review. J Pharm Bioallied Sci. 2019;11(suppl 2):S499-S506.
- Thongprasom K, Carrozzo M, Furness S, et al. Interventions for treating oral lichen planus. Cochrane Database Syst Rev. 2011;7:CD001168.
- López-Jornet P, Martínez-Beneyto Y, Nicolás AV, et al. Professional attitudes toward oral lichen planus: need for national and international guidelines. J Eval Clin Pract. 2009;15:541-542.
- Yang H, Wu Y, Jiang L, et al. Possible alternative therapies for oral lichen planus cases refractory to steroid therapies. Oral Surg Oral Med Oral Pathol Oral Radiol. 2016;121:496-509.
- Ribero S, Borradori L. Re: risk of malignancy and systemic absorption after application of topical tacrolimus in oral lichen planus. J Eur Acad Dermatol Venereol. 2017;31:E85-E86.
- Piñas L, Alkhraisat MH, Fernández RS, et al. Biological therapy of refractory ulcerative oral lichen planus with plasma rich in growth factors. Am J Clin Dermatol. 2017;18:429-433.
- Anitua E, Zalduendo MM, Prado R, et al. Morphogen and proinflammatory cytokine release kinetics from PRGF-Endoret fibrin scaffolds: evaluation of the effect of leukocyte inclusion. J Biomed Mater Res A. 2015;103:1011-1020.
- Anitua E, Prado R, Sánchez M, et al. Platelet-rich plasma: preparation and formulation. Oper Tech Orthop. 2012;22:25-32.
- González-Moles MA. The use of topical corticoids in oral pathology. Med Oral Pathol Oral Cir Bucal. 2010;15:E827-E831.
- Siponen M, Huuskonen L, Kallio-Pulkkinen S, et al. Topical tacrolimus, triamcinolone acetonide, and placebo in oral lichen planus: a pilot randomized controlled trial. Oral Dis. 2017;23:660-668.
- Adami G, Saag KG. Glucocorticoid-induced osteoporosis update. Curr Opin Rheumatol. 2019;31:388-393.
- Lavaee F, Shadmanpour M. Comparison of the effect of photodynamic therapy and topical corticosteroid on oral lichen planus lesions. Oral Dis. 2019;25:1954-1963.
- Derikvand N, Ghasemi SS, Moharami M, et al. Management of oral lichen planus by 980 nm diode laser. J Lasers Med Sci. 2017;8:150-154.
- Bennardo F, Liborio F, Barone S, et al. Efficacy of platelet-rich fibrin compared with triamcinolone acetonide as injective therapy in the treatment of symptomatic oral lichen planus: a pilot study. Clin Oral Investig. 2021;25:3747-3755.
- Anitua E, Andia I, Ardanza B, et al. Autologous platelets as a source of proteins for healing and tissue regeneration. Thromb Haemost. 2004;91:4-15.
- Barrientos S, Brem H, Stojadinovic O, et al. Clinical application of growth factors and cytokines in wound healing. Wound Repair Regen. 2014;22:569-578.
- Anitua E. Plasma rich in growth factors: preliminary results of use in the preparation of future sites for implants. Int J Oral Maxillofac Implants. 1999;14:529-535.
Lichen planus is a chronic inflammatory mucocutaneous disease that usually affects the skin and/or the genital and oral mucosae.1,2 This disease classically presents with clinical relapses or outbreaks that alternate with periods of remission or latency. Oral lichen planus (OLP) can present with or without extraoral manifestation. It sometimes is difficult to differentiate OLP from oral lichenoid reactions, which can be related to dental materials, some drugs, and systemic conditions or can be idiopathic.1,2
Oral lichen planus is one of the most common noninfectious diseases of the oral cavity, with a reported prevalence of 1% worldwide and marked geographical differences. In Europe, the prevalence of OLP ranges from 1% to 2%.3,4 It is more frequent in women (1.5:1 to 2:1) and usually appears in the fourth and fifth decades of life.1-4
The causes of OLP have not been entirely elucidated, but it is broadly accepted that there is a deregulation on different T lymphocytes that in turn causes effects on CD8 lymphocytes in response to an external noxa. This unknown “trigger” or starting factor also produces an impact on basal keratinocytes. Therefore, the pathogenesis of lichen planus is influenced by a series of cellular events mediated by different cytokines.2,5,6 Among these, tumor necrosis factor α and IL-1 are known to have important roles in the disease. More recently, other cytokines, such as IL-4, secreted by type 2 helper T cells, also have been related to the development and progression of the oral lesions.5,6 In addition to the factors that generate the onset of the disease, there are others that may precipitate clinical outbreaks. Different factors have been related to the progression of the disease, influencing the initiation, perpetuation, and/or worsening of OLP lesions.1,2 Exactly how these factors affect disease progression is another challenging question. The list of possible or potential factors related to disease progression is long; nonetheless, in the vast majority, a clear explanation at a molecular level has not been clearly demonstrated.2,5
Conventionally, 6 clinical presentations of OLP lesions divided into 2 main groups have been described in the oral cavity: white forms (reticular, papular, and plaquelike) and red forms (erythematous, atrophic-erosive, and bullous).1,7-9
Oral lichen planus mainly is treated with topically or systemically administered steroids based on the presence of symptoms such as pain and inability to perform daily activities (eg, eating, talking).5,10 The treatment of choice often is based on the professional’s experience, as there are no broadly accepted national or international clinical practice guidelines on steroid type, administration route, dose, vehicle for administration, or maintenance.11 Despite this lack of unified criteria, different topical and systemic steroid administration protocols allow a reduction in the symptoms or even the disappearance of the red lesions to be achieved in many cases. Unfortunately, there are many patients with lesions refractory to standard treatments for OLP.12 Several alternatives for these patients have been described in the literature, though on many occasions these alternatives present substantial side effects for the patient.13 The search for an effective treatment without side effects is still challenging. One of the treatments tested under this premise has been the application of plasma rich in growth factors (PRGF) by means of infiltration or topical application, in both cases obtaining good results without side effects.14
We sought to analyze the information from a case series of patients treated at the Eduardo Anitua Clinic (Vitoria-Gasteiz, Spain) and describe the results and follow-up of patients with erosive OLP refractory to standard therapy who have been successfully treated by local infiltration of PRGF as the only treatment.
Material and Methods
Patients—We included data from the database of the clinical center with de-identified information of patients with erosive OLP diagnosed clinically and histopathologically who did not respond to conventional treatment (ie, topical and/or systemic corticosteroids [depending on the case]) as well as patients who presented with extensive erosive OLP with systemic involvement and whose systemic treatment was not effective in resolving oral manifestations.
Therapies Administered and Evaluations—Lesions refractory to conventional corticosteroid protocols had been previously treated for 30 days with 0.5% triamcinolone acetonide mouth rinse followed by a cycle of 1% triamcinolone acetonide mouth rinse. Subsequently, a cycle of oral corticosteroids (prednisone for 30 days: 1 mg/kg/d in a single morning dose with staged reduction after the first week) had been administered. One dayafter the corticosteroid treatment was suspended, the patients were treated by PRGF-Endoret (BTI Biotechnology Institute) infiltration following the protocol described by Anitua et al.15,16
Before starting the infiltrations with PRGF, the patient had been asked to rate the pain level on a visual analog scale (VAS) of 1 to 10, with 10 being the most intense imaginable pain. Pain score was subsequently rated and registered during every visit. An initial photograph of the lesion also was obtained to establish a starting point for further comparisons of clinical evolution of the lesions.
Prior to each infiltration, the plasma was separated into 2 fractions. The second fraction was the one that corresponded to the highest number of platelets and included the 2 mL of plasma just above the white series (or buffy coat). This fraction of plasma was the one used to infiltrate the lesions.
Plasma rich in growth factors was activated just before infiltration. The activation was done by adding 10% calcium chloride. Once activated, it was infiltrated into the active lesion using a 31-G × 1/6-in hypodermic needle and a 2-mL Luer-lock syringe. Infiltrations were performed without anesthesia. Four punctures were made for each ulcerative lesion, dividing the lesion into 4 points: upper, lower, right, and left. Plasma rich in growth factors was infiltrated until a slight blanching was observed in the surrounding tissue. At that moment, the infiltration was stopped and was carried out in the next infiltration site.
One treatment session was performed per week, with follow-up 1 week after treatment. In the control visit, the state of the lesions was re-evaluated, and it was decided whether new infiltrations were needed. The treatment was finished when complete epithelialization of the lesion was visualized or the associated symptoms disappeared. At each visit, photographs were taken, and the patient assessed the severity of pain on the VAS.
Statistical Analysis—A Shapiro-Wilk test was carried out with the obtained data to check the normal distribution of the sample. The evolution of pain during the study was compared by paired t test. The qualitative variables were described by means of a frequency analysis. Quantitative variables were described by the mean and the SD. The data were analyzed with SPSS V15.0 for Windows (SPSS Inc). P<.05 showed statistical significance.
Results
A total of 15 patients were included in the study, all with atrophic-erosive lichen planus. Two patients were male, and 13 were female. The mean age (SD) of the patients included in the study was 55.27 (14.19) years. The mean number of outbreaks per year (SD) was 3.2 (1.7), with a range of 1 to 8 outbreaks.
Healing of OLP Lesions—The number of treatment sessions to achieve complete healing varied among the patients (Figures 1 and 2). Ten patients (66.7%) required a single session, 2 patients (13.3%) required 2 sessions, and 3 patients (20%) required 3 sessions. The mean time (SD) without lesions for the patients who required a single session was 10.9 (5.2) months (range, 6–24 months).

Pain Assessment—The mean (SD) score obtained on the VAS before treatment with PRGF was 8.27 (1.16); this score dropped to 1.27 (1.53) after the first treatment session and was a statistically significant difference (P=.006).

For those patients requiring more than 1 session, the mean (SD) pain scores decreased by 0.75 (0.97) points and 0 points after the first and second sessions of treatment, respectively. The mean (SD) amount of PRGF infiltrated in each patient in the first session was 2.60 (0.63) mL. In the second session, the mean (SD) amount was 1.2 (0.33) mL; these differences were statistically significant (P=.008). In the last session, the mean (SD) amount was 1.1 (0.22) mL.
Follow-up and Adverse Effects—The mean (SD) follow-up time was 47.16 (15.78) months. The patients were free of symptoms, and there were no adverse effects derived from the treatment during follow-up.
Comment
The primary goal of OLP treatment is to stop the outbreaks.1,9,13 The lack of potency of corticosteroids in some patients with OLP could be due in part to the inadequate selection of the vehicle (ointment/oral rinse) for the extension and characteristics of the lesion or because of an inappropriate prescription dose, time, and/or frequency, as described by González-Moles.17 However, even when using an appropriate protocol, some lesions are resistant to topical treatment and require other therapeutic modalities.1,9,13 Previously proposed topical treatments include different immunosuppressants, such as the mammalian target of rapamycin, tacrolimus ointment 0.1%, pimecrolimus cream 1%, or cyclosporine A (50–100 mg/mL) formulations.18 Nevertheless, these drugs seem to have a greater number of side effects than topical steroids, and tacrolimus has been associated with cases of oral malignancy after continuing treatment.15
Severe and/or recalcitrant lesions and extraoral involvement have been successfully treated with systemic prednisone (40–80 mg/d).1,9,13 Nevertheless, systemic corticosteroid toxicity requires that these treatments should be used only when necessary at the lowest possible dose and for the shortest possible duration.19 Other nonpharmacologic options for treatment are photodynamic, UV, and low-level laser therapy.20,21 They have been accepted as supplementary modalities in different inflammatory skin conditions but present important technical requirements. Their effectiveness in corticosteroid-resistant cases have not been definitively assessed. Interestingly, promising results recently have been reported by Bennardo et al22 when comparing the efficacy of autologous platelet concentrates with triamcinolone injection.
In our study, the use of PRGF stopped the lesions’ evolution since the first treatment session, reducing them by 6.5-fold. The positive effects observed may have been promoted by the activity of different proteins present in PRGF (eg, platelet-derived growth factor, vascular endothelial growth factor, transforming growth factor, epidermal growth factor, fibroblast growth factor, fibronectin). These molecules contribute to collagen synthesis; angiogenesis; endothelial cell migration and proliferation; or keratinocyte cell migration, proliferation, differentiation, growth, and migration—phenomena that are essential for healing and re-epithelialization.23-25
Different studies also have supported an anti-inflammatory effect of PRGF mediated by an inhibition of the transcription of nuclear factor–κB and the expression of cyclooxygenase-2 and chemokine receptor type 4 produced by its high content of hepatocyte growth factor or the reduction of inflammatory marker expression, such as intercellular adhesion molecule 1. The development of an efficient 3-dimensional fibrin scaffold formation that occurs after PRGF administration also could facilitate healing, helping some cell populations to guide their position and function.23-25
Limitations of our study include the small number of patients and the absence of a control group. The higher number of female patients in the study did not seem to affect the results, as differences related to gender have not been reported when treating patients with OLP with autologous platelet concentrates or other modalities of treatment.
Conclusion
Results from our study indicate that the use of PRGF could be a new treatment option for OLP cases refractory to conventional therapy. No complications were observed during the treatment procedure or during the complete follow-up period. Nonetheless, new prospective studies with a greater number of patients and longer follow-up periods are needed to confirm these preliminary results.
Lichen planus is a chronic inflammatory mucocutaneous disease that usually affects the skin and/or the genital and oral mucosae.1,2 This disease classically presents with clinical relapses or outbreaks that alternate with periods of remission or latency. Oral lichen planus (OLP) can present with or without extraoral manifestation. It sometimes is difficult to differentiate OLP from oral lichenoid reactions, which can be related to dental materials, some drugs, and systemic conditions or can be idiopathic.1,2
Oral lichen planus is one of the most common noninfectious diseases of the oral cavity, with a reported prevalence of 1% worldwide and marked geographical differences. In Europe, the prevalence of OLP ranges from 1% to 2%.3,4 It is more frequent in women (1.5:1 to 2:1) and usually appears in the fourth and fifth decades of life.1-4
The causes of OLP have not been entirely elucidated, but it is broadly accepted that there is a deregulation on different T lymphocytes that in turn causes effects on CD8 lymphocytes in response to an external noxa. This unknown “trigger” or starting factor also produces an impact on basal keratinocytes. Therefore, the pathogenesis of lichen planus is influenced by a series of cellular events mediated by different cytokines.2,5,6 Among these, tumor necrosis factor α and IL-1 are known to have important roles in the disease. More recently, other cytokines, such as IL-4, secreted by type 2 helper T cells, also have been related to the development and progression of the oral lesions.5,6 In addition to the factors that generate the onset of the disease, there are others that may precipitate clinical outbreaks. Different factors have been related to the progression of the disease, influencing the initiation, perpetuation, and/or worsening of OLP lesions.1,2 Exactly how these factors affect disease progression is another challenging question. The list of possible or potential factors related to disease progression is long; nonetheless, in the vast majority, a clear explanation at a molecular level has not been clearly demonstrated.2,5
Conventionally, 6 clinical presentations of OLP lesions divided into 2 main groups have been described in the oral cavity: white forms (reticular, papular, and plaquelike) and red forms (erythematous, atrophic-erosive, and bullous).1,7-9
Oral lichen planus mainly is treated with topically or systemically administered steroids based on the presence of symptoms such as pain and inability to perform daily activities (eg, eating, talking).5,10 The treatment of choice often is based on the professional’s experience, as there are no broadly accepted national or international clinical practice guidelines on steroid type, administration route, dose, vehicle for administration, or maintenance.11 Despite this lack of unified criteria, different topical and systemic steroid administration protocols allow a reduction in the symptoms or even the disappearance of the red lesions to be achieved in many cases. Unfortunately, there are many patients with lesions refractory to standard treatments for OLP.12 Several alternatives for these patients have been described in the literature, though on many occasions these alternatives present substantial side effects for the patient.13 The search for an effective treatment without side effects is still challenging. One of the treatments tested under this premise has been the application of plasma rich in growth factors (PRGF) by means of infiltration or topical application, in both cases obtaining good results without side effects.14
We sought to analyze the information from a case series of patients treated at the Eduardo Anitua Clinic (Vitoria-Gasteiz, Spain) and describe the results and follow-up of patients with erosive OLP refractory to standard therapy who have been successfully treated by local infiltration of PRGF as the only treatment.
Material and Methods
Patients—We included data from the database of the clinical center with de-identified information of patients with erosive OLP diagnosed clinically and histopathologically who did not respond to conventional treatment (ie, topical and/or systemic corticosteroids [depending on the case]) as well as patients who presented with extensive erosive OLP with systemic involvement and whose systemic treatment was not effective in resolving oral manifestations.
Therapies Administered and Evaluations—Lesions refractory to conventional corticosteroid protocols had been previously treated for 30 days with 0.5% triamcinolone acetonide mouth rinse followed by a cycle of 1% triamcinolone acetonide mouth rinse. Subsequently, a cycle of oral corticosteroids (prednisone for 30 days: 1 mg/kg/d in a single morning dose with staged reduction after the first week) had been administered. One dayafter the corticosteroid treatment was suspended, the patients were treated by PRGF-Endoret (BTI Biotechnology Institute) infiltration following the protocol described by Anitua et al.15,16
Before starting the infiltrations with PRGF, the patient had been asked to rate the pain level on a visual analog scale (VAS) of 1 to 10, with 10 being the most intense imaginable pain. Pain score was subsequently rated and registered during every visit. An initial photograph of the lesion also was obtained to establish a starting point for further comparisons of clinical evolution of the lesions.
Prior to each infiltration, the plasma was separated into 2 fractions. The second fraction was the one that corresponded to the highest number of platelets and included the 2 mL of plasma just above the white series (or buffy coat). This fraction of plasma was the one used to infiltrate the lesions.
Plasma rich in growth factors was activated just before infiltration. The activation was done by adding 10% calcium chloride. Once activated, it was infiltrated into the active lesion using a 31-G × 1/6-in hypodermic needle and a 2-mL Luer-lock syringe. Infiltrations were performed without anesthesia. Four punctures were made for each ulcerative lesion, dividing the lesion into 4 points: upper, lower, right, and left. Plasma rich in growth factors was infiltrated until a slight blanching was observed in the surrounding tissue. At that moment, the infiltration was stopped and was carried out in the next infiltration site.
One treatment session was performed per week, with follow-up 1 week after treatment. In the control visit, the state of the lesions was re-evaluated, and it was decided whether new infiltrations were needed. The treatment was finished when complete epithelialization of the lesion was visualized or the associated symptoms disappeared. At each visit, photographs were taken, and the patient assessed the severity of pain on the VAS.
Statistical Analysis—A Shapiro-Wilk test was carried out with the obtained data to check the normal distribution of the sample. The evolution of pain during the study was compared by paired t test. The qualitative variables were described by means of a frequency analysis. Quantitative variables were described by the mean and the SD. The data were analyzed with SPSS V15.0 for Windows (SPSS Inc). P<.05 showed statistical significance.
Results
A total of 15 patients were included in the study, all with atrophic-erosive lichen planus. Two patients were male, and 13 were female. The mean age (SD) of the patients included in the study was 55.27 (14.19) years. The mean number of outbreaks per year (SD) was 3.2 (1.7), with a range of 1 to 8 outbreaks.
Healing of OLP Lesions—The number of treatment sessions to achieve complete healing varied among the patients (Figures 1 and 2). Ten patients (66.7%) required a single session, 2 patients (13.3%) required 2 sessions, and 3 patients (20%) required 3 sessions. The mean time (SD) without lesions for the patients who required a single session was 10.9 (5.2) months (range, 6–24 months).

Pain Assessment—The mean (SD) score obtained on the VAS before treatment with PRGF was 8.27 (1.16); this score dropped to 1.27 (1.53) after the first treatment session and was a statistically significant difference (P=.006).

For those patients requiring more than 1 session, the mean (SD) pain scores decreased by 0.75 (0.97) points and 0 points after the first and second sessions of treatment, respectively. The mean (SD) amount of PRGF infiltrated in each patient in the first session was 2.60 (0.63) mL. In the second session, the mean (SD) amount was 1.2 (0.33) mL; these differences were statistically significant (P=.008). In the last session, the mean (SD) amount was 1.1 (0.22) mL.
Follow-up and Adverse Effects—The mean (SD) follow-up time was 47.16 (15.78) months. The patients were free of symptoms, and there were no adverse effects derived from the treatment during follow-up.
Comment
The primary goal of OLP treatment is to stop the outbreaks.1,9,13 The lack of potency of corticosteroids in some patients with OLP could be due in part to the inadequate selection of the vehicle (ointment/oral rinse) for the extension and characteristics of the lesion or because of an inappropriate prescription dose, time, and/or frequency, as described by González-Moles.17 However, even when using an appropriate protocol, some lesions are resistant to topical treatment and require other therapeutic modalities.1,9,13 Previously proposed topical treatments include different immunosuppressants, such as the mammalian target of rapamycin, tacrolimus ointment 0.1%, pimecrolimus cream 1%, or cyclosporine A (50–100 mg/mL) formulations.18 Nevertheless, these drugs seem to have a greater number of side effects than topical steroids, and tacrolimus has been associated with cases of oral malignancy after continuing treatment.15
Severe and/or recalcitrant lesions and extraoral involvement have been successfully treated with systemic prednisone (40–80 mg/d).1,9,13 Nevertheless, systemic corticosteroid toxicity requires that these treatments should be used only when necessary at the lowest possible dose and for the shortest possible duration.19 Other nonpharmacologic options for treatment are photodynamic, UV, and low-level laser therapy.20,21 They have been accepted as supplementary modalities in different inflammatory skin conditions but present important technical requirements. Their effectiveness in corticosteroid-resistant cases have not been definitively assessed. Interestingly, promising results recently have been reported by Bennardo et al22 when comparing the efficacy of autologous platelet concentrates with triamcinolone injection.
In our study, the use of PRGF stopped the lesions’ evolution since the first treatment session, reducing them by 6.5-fold. The positive effects observed may have been promoted by the activity of different proteins present in PRGF (eg, platelet-derived growth factor, vascular endothelial growth factor, transforming growth factor, epidermal growth factor, fibroblast growth factor, fibronectin). These molecules contribute to collagen synthesis; angiogenesis; endothelial cell migration and proliferation; or keratinocyte cell migration, proliferation, differentiation, growth, and migration—phenomena that are essential for healing and re-epithelialization.23-25
Different studies also have supported an anti-inflammatory effect of PRGF mediated by an inhibition of the transcription of nuclear factor–κB and the expression of cyclooxygenase-2 and chemokine receptor type 4 produced by its high content of hepatocyte growth factor or the reduction of inflammatory marker expression, such as intercellular adhesion molecule 1. The development of an efficient 3-dimensional fibrin scaffold formation that occurs after PRGF administration also could facilitate healing, helping some cell populations to guide their position and function.23-25
Limitations of our study include the small number of patients and the absence of a control group. The higher number of female patients in the study did not seem to affect the results, as differences related to gender have not been reported when treating patients with OLP with autologous platelet concentrates or other modalities of treatment.
Conclusion
Results from our study indicate that the use of PRGF could be a new treatment option for OLP cases refractory to conventional therapy. No complications were observed during the treatment procedure or during the complete follow-up period. Nonetheless, new prospective studies with a greater number of patients and longer follow-up periods are needed to confirm these preliminary results.
- Al-Hashimi I, Schifter M, Lockhart PB, et al. Oral lichen planus and oral lichenoid lesions: diagnostic and therapeutic considerations. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;103:1-12.
- Kurago ZB. Etiology and pathogenesis of oral lichen planus: an overview. Oral Surg Oral Med Oral Pathol Oral Radiol. 2016;122:72-80.
- McCartan BE, Healy CM. The reported prevalence of oral lichen planus: a review and critique. J Oral Pathol Med. 2008;37:447-453.
- González-Moles MÁ, Warnakulasuriya S, González-Ruiz I, et al. Worldwide prevalence of oral lichen planus: a systematic review and meta-analysis. Oral Dis. 2021;27:813-828.
- Nosratzehi T. Oral lichen planus: an overview of potential risk factors, biomarkers and treatments. Asian Pac J Cancer Prev. 2018;19:1161-1167.
- Mehrbani SP, Motahari P, Azar FP, et al. Role of interleukin-4 in pathogenesis of oral lichen planus: a systematic review. Med Oral Patol Oral Cir Bucal. 2020;25:E410-E415.
- Edwards PC, Kelsch R. Oral lichen planus: clinical presentation and management. J Can Dent Assoc. 2002;68:494-499.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826.
- Babu A, Chellaswamy S, Muthukumar S, et al. Bullous lichen planus: case report and review. J Pharm Bioallied Sci. 2019;11(suppl 2):S499-S506.
- Thongprasom K, Carrozzo M, Furness S, et al. Interventions for treating oral lichen planus. Cochrane Database Syst Rev. 2011;7:CD001168.
- López-Jornet P, Martínez-Beneyto Y, Nicolás AV, et al. Professional attitudes toward oral lichen planus: need for national and international guidelines. J Eval Clin Pract. 2009;15:541-542.
- Yang H, Wu Y, Jiang L, et al. Possible alternative therapies for oral lichen planus cases refractory to steroid therapies. Oral Surg Oral Med Oral Pathol Oral Radiol. 2016;121:496-509.
- Ribero S, Borradori L. Re: risk of malignancy and systemic absorption after application of topical tacrolimus in oral lichen planus. J Eur Acad Dermatol Venereol. 2017;31:E85-E86.
- Piñas L, Alkhraisat MH, Fernández RS, et al. Biological therapy of refractory ulcerative oral lichen planus with plasma rich in growth factors. Am J Clin Dermatol. 2017;18:429-433.
- Anitua E, Zalduendo MM, Prado R, et al. Morphogen and proinflammatory cytokine release kinetics from PRGF-Endoret fibrin scaffolds: evaluation of the effect of leukocyte inclusion. J Biomed Mater Res A. 2015;103:1011-1020.
- Anitua E, Prado R, Sánchez M, et al. Platelet-rich plasma: preparation and formulation. Oper Tech Orthop. 2012;22:25-32.
- González-Moles MA. The use of topical corticoids in oral pathology. Med Oral Pathol Oral Cir Bucal. 2010;15:E827-E831.
- Siponen M, Huuskonen L, Kallio-Pulkkinen S, et al. Topical tacrolimus, triamcinolone acetonide, and placebo in oral lichen planus: a pilot randomized controlled trial. Oral Dis. 2017;23:660-668.
- Adami G, Saag KG. Glucocorticoid-induced osteoporosis update. Curr Opin Rheumatol. 2019;31:388-393.
- Lavaee F, Shadmanpour M. Comparison of the effect of photodynamic therapy and topical corticosteroid on oral lichen planus lesions. Oral Dis. 2019;25:1954-1963.
- Derikvand N, Ghasemi SS, Moharami M, et al. Management of oral lichen planus by 980 nm diode laser. J Lasers Med Sci. 2017;8:150-154.
- Bennardo F, Liborio F, Barone S, et al. Efficacy of platelet-rich fibrin compared with triamcinolone acetonide as injective therapy in the treatment of symptomatic oral lichen planus: a pilot study. Clin Oral Investig. 2021;25:3747-3755.
- Anitua E, Andia I, Ardanza B, et al. Autologous platelets as a source of proteins for healing and tissue regeneration. Thromb Haemost. 2004;91:4-15.
- Barrientos S, Brem H, Stojadinovic O, et al. Clinical application of growth factors and cytokines in wound healing. Wound Repair Regen. 2014;22:569-578.
- Anitua E. Plasma rich in growth factors: preliminary results of use in the preparation of future sites for implants. Int J Oral Maxillofac Implants. 1999;14:529-535.
- Al-Hashimi I, Schifter M, Lockhart PB, et al. Oral lichen planus and oral lichenoid lesions: diagnostic and therapeutic considerations. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;103:1-12.
- Kurago ZB. Etiology and pathogenesis of oral lichen planus: an overview. Oral Surg Oral Med Oral Pathol Oral Radiol. 2016;122:72-80.
- McCartan BE, Healy CM. The reported prevalence of oral lichen planus: a review and critique. J Oral Pathol Med. 2008;37:447-453.
- González-Moles MÁ, Warnakulasuriya S, González-Ruiz I, et al. Worldwide prevalence of oral lichen planus: a systematic review and meta-analysis. Oral Dis. 2021;27:813-828.
- Nosratzehi T. Oral lichen planus: an overview of potential risk factors, biomarkers and treatments. Asian Pac J Cancer Prev. 2018;19:1161-1167.
- Mehrbani SP, Motahari P, Azar FP, et al. Role of interleukin-4 in pathogenesis of oral lichen planus: a systematic review. Med Oral Patol Oral Cir Bucal. 2020;25:E410-E415.
- Edwards PC, Kelsch R. Oral lichen planus: clinical presentation and management. J Can Dent Assoc. 2002;68:494-499.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826.
- Babu A, Chellaswamy S, Muthukumar S, et al. Bullous lichen planus: case report and review. J Pharm Bioallied Sci. 2019;11(suppl 2):S499-S506.
- Thongprasom K, Carrozzo M, Furness S, et al. Interventions for treating oral lichen planus. Cochrane Database Syst Rev. 2011;7:CD001168.
- López-Jornet P, Martínez-Beneyto Y, Nicolás AV, et al. Professional attitudes toward oral lichen planus: need for national and international guidelines. J Eval Clin Pract. 2009;15:541-542.
- Yang H, Wu Y, Jiang L, et al. Possible alternative therapies for oral lichen planus cases refractory to steroid therapies. Oral Surg Oral Med Oral Pathol Oral Radiol. 2016;121:496-509.
- Ribero S, Borradori L. Re: risk of malignancy and systemic absorption after application of topical tacrolimus in oral lichen planus. J Eur Acad Dermatol Venereol. 2017;31:E85-E86.
- Piñas L, Alkhraisat MH, Fernández RS, et al. Biological therapy of refractory ulcerative oral lichen planus with plasma rich in growth factors. Am J Clin Dermatol. 2017;18:429-433.
- Anitua E, Zalduendo MM, Prado R, et al. Morphogen and proinflammatory cytokine release kinetics from PRGF-Endoret fibrin scaffolds: evaluation of the effect of leukocyte inclusion. J Biomed Mater Res A. 2015;103:1011-1020.
- Anitua E, Prado R, Sánchez M, et al. Platelet-rich plasma: preparation and formulation. Oper Tech Orthop. 2012;22:25-32.
- González-Moles MA. The use of topical corticoids in oral pathology. Med Oral Pathol Oral Cir Bucal. 2010;15:E827-E831.
- Siponen M, Huuskonen L, Kallio-Pulkkinen S, et al. Topical tacrolimus, triamcinolone acetonide, and placebo in oral lichen planus: a pilot randomized controlled trial. Oral Dis. 2017;23:660-668.
- Adami G, Saag KG. Glucocorticoid-induced osteoporosis update. Curr Opin Rheumatol. 2019;31:388-393.
- Lavaee F, Shadmanpour M. Comparison of the effect of photodynamic therapy and topical corticosteroid on oral lichen planus lesions. Oral Dis. 2019;25:1954-1963.
- Derikvand N, Ghasemi SS, Moharami M, et al. Management of oral lichen planus by 980 nm diode laser. J Lasers Med Sci. 2017;8:150-154.
- Bennardo F, Liborio F, Barone S, et al. Efficacy of platelet-rich fibrin compared with triamcinolone acetonide as injective therapy in the treatment of symptomatic oral lichen planus: a pilot study. Clin Oral Investig. 2021;25:3747-3755.
- Anitua E, Andia I, Ardanza B, et al. Autologous platelets as a source of proteins for healing and tissue regeneration. Thromb Haemost. 2004;91:4-15.
- Barrientos S, Brem H, Stojadinovic O, et al. Clinical application of growth factors and cytokines in wound healing. Wound Repair Regen. 2014;22:569-578.
- Anitua E. Plasma rich in growth factors: preliminary results of use in the preparation of future sites for implants. Int J Oral Maxillofac Implants. 1999;14:529-535.
Practice Points
- Treating erosive oral lichen planus lesions refractory to conventional steroid treatments can be challenging for clinicians.
- Complete re-epithelialization and total pain relief could be observed after 1 to 3 weekly perilesional infiltrations with plasma rich in growth factors.
- No relapse of the lesions in the same area or other complications could be observed during the follow-up time.
Teaching Evidence-Based Dermatology Using a Web-Based Journal Club: A Pilot Study and Survey
To the Editor:
With a steady increase in dermatology publications over recent decades, there is an expanding pool of evidence to address clinical questions.1 Residency training is the time when appraising the medical literature and practicing evidence-based medicine is most honed. Evidence-based medicine is an essential component of Practice-based Learning and Improvement, a required core competency of the Accreditation Council for Graduate Medical Education.2 Assimilation of new research evidence is traditionally taught through didactics and journal club discussions in residency.
However, at a time when the demand for information overwhelms safeguards that exist to evaluate its quality, it is more important than ever to be equipped with the proper tools to critically appraise novel literature. Beyond accepting a scientific article at face value, physicians must learn to ask targeted questions of the study design, results, and clinical relevance. These questions change based on the type of study, and organizations such as the Oxford Centre for Evidence-Based Medicine provide guidance through critical appraisal worksheets.3
To investigate the utility of using guided questions to evaluate the reliability, significance, and applicability of clinical evidence, we beta tested a novel web-based application in an academic dermatology setting to design and run a journal club for residents. Six dermatology residents participated in this institutional review board–approved study comprised of 3 phases: (1) independent article appraisal through the web-based application, (2) group discussion, and (3) anonymous postsurvey.
Using this platform, we uploaded a recent article into the interactive reader, which contained an integrated tool for appraisal based on specific questions. Because the article described the results of a randomized clinical trial, we used questions from the Centre for Evidence-Based Medicine’s Randomised Controlled Trials Critical Appraisal Worksheet, which has a series of questions to evaluate internal validity, results, and external validity and applicability.3
Residents used the platform to independently read the article, highlight areas of the text that corresponded to 8 critical appraisal questions, and answer yes or no to these questions. Based on residents’ answers, a final appraisal score (on a scale of 1% to 100%) was generated. Simultaneously, the attending dermatologist leading the journal club (C.W.) also completed the assignment to establish an expert score.
Scores from the residents’ independent appraisal ranged from 75% to 100% (mean, 85.4%). Upon discussing the article in a group setting, the residents established a consensus score of 75%. This consensus score matched the expert score, which suggested to us that both independently reviewing the article using guided questions and conducting a group debriefing were necessary to match the expert level of critical appraisal.
Of note, the residents’ average independent appraisal score was higher than both the consensus and expert scores, indicating that the residents evaluated the article less critically on their own. With more practice using this method, it is possible that the precision and accuracy of the residents’ critical appraisal of scientific articles will improve.
In the postsurvey, we asked residents about the critical appraisal of the medical literature. All residents agreed that evaluating the quality of evidence when reading a scientific article was somewhat important or very important to them; however, only 2 of 6 evaluated the quality of evidence all the time, and the other 4 did so half of the time or less than half of the time.
When critically appraising articles, 2 of 6 residents used specific rubrics half of the time; 4 of 6 less than half of the time. Most important, 5 of 6 residents agreed that the quality of evidence affected their management decisions more than half of the time or all of the time. Although it is clear that residents value evidence-based medicine and understand the importance of evaluating the quality of evidence, doing so currently might not be simple or practical.
An organized framework for appraising articles would streamline the process. Five of 6 residents agreed that the use of specific questions as a guide made it easier to appraise an article for the quality of its evidence. Four of 6 residents found that juxtaposing specific questions with the interactive reader was helpful; 5 of 6 agreed that they would use a web-based journal club platform if given the option.
Lastly, 5 of 6 residents agreed that if such a tool were available, a platform containing all major dermatology publications in an interactive reader format, along with relevant appraisal questions on the side, would be useful.
This pilot study augmented the typical journal club experience by emphasizing goal-directed reading and the importance of analyzing the quality of evidence. The combination of independent appraisal of an article using targeted questions and a group debrief led to better understanding of the evidence and its clinical applicability. The COVID-19 pandemic may be a better time than ever to explore innovative ways to teach evidence-based medicine in residency training.
- Mimouni D, Pavlovsky L, Akerman L, et al. Trends in dermatology publications over the past 15 years. Am J Clin Dermatol. 2010;11:55-58. doi:10.2165/11530190-000000000-00000.
- NEJM Knowledge+ Team. Exploring the ACGME Core Competencies: Practice-Based Learning and Improvement (part 2 of 7). Massachusetts Medical Society. NEJM Knowledge+ website. Published July 28, 2016. Accessed January 15, 2022. https://knowledgeplus.nejm.org/blog/practice-based-learning-and-improvement/
- University of Oxford. Critical appraisal tools. Centre for Evidence-Based Medicine website. Accessed January 2, 2022. www.cebm.ox.ac.uk/resources/ebm-tools/critical-appraisal-tools
To the Editor:
With a steady increase in dermatology publications over recent decades, there is an expanding pool of evidence to address clinical questions.1 Residency training is the time when appraising the medical literature and practicing evidence-based medicine is most honed. Evidence-based medicine is an essential component of Practice-based Learning and Improvement, a required core competency of the Accreditation Council for Graduate Medical Education.2 Assimilation of new research evidence is traditionally taught through didactics and journal club discussions in residency.
However, at a time when the demand for information overwhelms safeguards that exist to evaluate its quality, it is more important than ever to be equipped with the proper tools to critically appraise novel literature. Beyond accepting a scientific article at face value, physicians must learn to ask targeted questions of the study design, results, and clinical relevance. These questions change based on the type of study, and organizations such as the Oxford Centre for Evidence-Based Medicine provide guidance through critical appraisal worksheets.3
To investigate the utility of using guided questions to evaluate the reliability, significance, and applicability of clinical evidence, we beta tested a novel web-based application in an academic dermatology setting to design and run a journal club for residents. Six dermatology residents participated in this institutional review board–approved study comprised of 3 phases: (1) independent article appraisal through the web-based application, (2) group discussion, and (3) anonymous postsurvey.
Using this platform, we uploaded a recent article into the interactive reader, which contained an integrated tool for appraisal based on specific questions. Because the article described the results of a randomized clinical trial, we used questions from the Centre for Evidence-Based Medicine’s Randomised Controlled Trials Critical Appraisal Worksheet, which has a series of questions to evaluate internal validity, results, and external validity and applicability.3
Residents used the platform to independently read the article, highlight areas of the text that corresponded to 8 critical appraisal questions, and answer yes or no to these questions. Based on residents’ answers, a final appraisal score (on a scale of 1% to 100%) was generated. Simultaneously, the attending dermatologist leading the journal club (C.W.) also completed the assignment to establish an expert score.
Scores from the residents’ independent appraisal ranged from 75% to 100% (mean, 85.4%). Upon discussing the article in a group setting, the residents established a consensus score of 75%. This consensus score matched the expert score, which suggested to us that both independently reviewing the article using guided questions and conducting a group debriefing were necessary to match the expert level of critical appraisal.
Of note, the residents’ average independent appraisal score was higher than both the consensus and expert scores, indicating that the residents evaluated the article less critically on their own. With more practice using this method, it is possible that the precision and accuracy of the residents’ critical appraisal of scientific articles will improve.
In the postsurvey, we asked residents about the critical appraisal of the medical literature. All residents agreed that evaluating the quality of evidence when reading a scientific article was somewhat important or very important to them; however, only 2 of 6 evaluated the quality of evidence all the time, and the other 4 did so half of the time or less than half of the time.
When critically appraising articles, 2 of 6 residents used specific rubrics half of the time; 4 of 6 less than half of the time. Most important, 5 of 6 residents agreed that the quality of evidence affected their management decisions more than half of the time or all of the time. Although it is clear that residents value evidence-based medicine and understand the importance of evaluating the quality of evidence, doing so currently might not be simple or practical.
An organized framework for appraising articles would streamline the process. Five of 6 residents agreed that the use of specific questions as a guide made it easier to appraise an article for the quality of its evidence. Four of 6 residents found that juxtaposing specific questions with the interactive reader was helpful; 5 of 6 agreed that they would use a web-based journal club platform if given the option.
Lastly, 5 of 6 residents agreed that if such a tool were available, a platform containing all major dermatology publications in an interactive reader format, along with relevant appraisal questions on the side, would be useful.
This pilot study augmented the typical journal club experience by emphasizing goal-directed reading and the importance of analyzing the quality of evidence. The combination of independent appraisal of an article using targeted questions and a group debrief led to better understanding of the evidence and its clinical applicability. The COVID-19 pandemic may be a better time than ever to explore innovative ways to teach evidence-based medicine in residency training.
To the Editor:
With a steady increase in dermatology publications over recent decades, there is an expanding pool of evidence to address clinical questions.1 Residency training is the time when appraising the medical literature and practicing evidence-based medicine is most honed. Evidence-based medicine is an essential component of Practice-based Learning and Improvement, a required core competency of the Accreditation Council for Graduate Medical Education.2 Assimilation of new research evidence is traditionally taught through didactics and journal club discussions in residency.
However, at a time when the demand for information overwhelms safeguards that exist to evaluate its quality, it is more important than ever to be equipped with the proper tools to critically appraise novel literature. Beyond accepting a scientific article at face value, physicians must learn to ask targeted questions of the study design, results, and clinical relevance. These questions change based on the type of study, and organizations such as the Oxford Centre for Evidence-Based Medicine provide guidance through critical appraisal worksheets.3
To investigate the utility of using guided questions to evaluate the reliability, significance, and applicability of clinical evidence, we beta tested a novel web-based application in an academic dermatology setting to design and run a journal club for residents. Six dermatology residents participated in this institutional review board–approved study comprised of 3 phases: (1) independent article appraisal through the web-based application, (2) group discussion, and (3) anonymous postsurvey.
Using this platform, we uploaded a recent article into the interactive reader, which contained an integrated tool for appraisal based on specific questions. Because the article described the results of a randomized clinical trial, we used questions from the Centre for Evidence-Based Medicine’s Randomised Controlled Trials Critical Appraisal Worksheet, which has a series of questions to evaluate internal validity, results, and external validity and applicability.3
Residents used the platform to independently read the article, highlight areas of the text that corresponded to 8 critical appraisal questions, and answer yes or no to these questions. Based on residents’ answers, a final appraisal score (on a scale of 1% to 100%) was generated. Simultaneously, the attending dermatologist leading the journal club (C.W.) also completed the assignment to establish an expert score.
Scores from the residents’ independent appraisal ranged from 75% to 100% (mean, 85.4%). Upon discussing the article in a group setting, the residents established a consensus score of 75%. This consensus score matched the expert score, which suggested to us that both independently reviewing the article using guided questions and conducting a group debriefing were necessary to match the expert level of critical appraisal.
Of note, the residents’ average independent appraisal score was higher than both the consensus and expert scores, indicating that the residents evaluated the article less critically on their own. With more practice using this method, it is possible that the precision and accuracy of the residents’ critical appraisal of scientific articles will improve.
In the postsurvey, we asked residents about the critical appraisal of the medical literature. All residents agreed that evaluating the quality of evidence when reading a scientific article was somewhat important or very important to them; however, only 2 of 6 evaluated the quality of evidence all the time, and the other 4 did so half of the time or less than half of the time.
When critically appraising articles, 2 of 6 residents used specific rubrics half of the time; 4 of 6 less than half of the time. Most important, 5 of 6 residents agreed that the quality of evidence affected their management decisions more than half of the time or all of the time. Although it is clear that residents value evidence-based medicine and understand the importance of evaluating the quality of evidence, doing so currently might not be simple or practical.
An organized framework for appraising articles would streamline the process. Five of 6 residents agreed that the use of specific questions as a guide made it easier to appraise an article for the quality of its evidence. Four of 6 residents found that juxtaposing specific questions with the interactive reader was helpful; 5 of 6 agreed that they would use a web-based journal club platform if given the option.
Lastly, 5 of 6 residents agreed that if such a tool were available, a platform containing all major dermatology publications in an interactive reader format, along with relevant appraisal questions on the side, would be useful.
This pilot study augmented the typical journal club experience by emphasizing goal-directed reading and the importance of analyzing the quality of evidence. The combination of independent appraisal of an article using targeted questions and a group debrief led to better understanding of the evidence and its clinical applicability. The COVID-19 pandemic may be a better time than ever to explore innovative ways to teach evidence-based medicine in residency training.
- Mimouni D, Pavlovsky L, Akerman L, et al. Trends in dermatology publications over the past 15 years. Am J Clin Dermatol. 2010;11:55-58. doi:10.2165/11530190-000000000-00000.
- NEJM Knowledge+ Team. Exploring the ACGME Core Competencies: Practice-Based Learning and Improvement (part 2 of 7). Massachusetts Medical Society. NEJM Knowledge+ website. Published July 28, 2016. Accessed January 15, 2022. https://knowledgeplus.nejm.org/blog/practice-based-learning-and-improvement/
- University of Oxford. Critical appraisal tools. Centre for Evidence-Based Medicine website. Accessed January 2, 2022. www.cebm.ox.ac.uk/resources/ebm-tools/critical-appraisal-tools
- Mimouni D, Pavlovsky L, Akerman L, et al. Trends in dermatology publications over the past 15 years. Am J Clin Dermatol. 2010;11:55-58. doi:10.2165/11530190-000000000-00000.
- NEJM Knowledge+ Team. Exploring the ACGME Core Competencies: Practice-Based Learning and Improvement (part 2 of 7). Massachusetts Medical Society. NEJM Knowledge+ website. Published July 28, 2016. Accessed January 15, 2022. https://knowledgeplus.nejm.org/blog/practice-based-learning-and-improvement/
- University of Oxford. Critical appraisal tools. Centre for Evidence-Based Medicine website. Accessed January 2, 2022. www.cebm.ox.ac.uk/resources/ebm-tools/critical-appraisal-tools
Practice Points
- A novel web-based application was beta tested in an academic dermatology setting to design and run a journal club for residents.
- Goal-directed reading was emphasized by using guided questions to critically appraise literature based on reliability, significance, and applicability.
- The combination of independent appraisal of an article using targeted questions and a group debrief led to better understanding of the evidence and its clinical applicability.
Naloxone Dispensing in Patients at Risk for Opioid Overdose After Total Knee Arthroplasty Within the Veterans Health Administration
Opioid overdose is a major public health challenge, with recent reports estimating 41 deaths per day in the United States from prescription opioid overdose.1,2 Prescribing naloxone has increasingly been advocated to reduce the risk of opioid overdose for patients identified as high risk. Naloxone distribution has been shown to decrease the incidence of opioid overdoses in the general population.3,4 The Centers for Disease Control and Prevention (CDC) Guideline for Prescribing Opioids for Chronic Pain recommends considering naloxone prescription for patients with a history of overdose or substance use disorder, opioid dosages ≥ 50 morphine equivalent daily dose (MEDD), and concurrent use of benzodiazepines.5
Although the CDC guidelines are intended for primary care clinicians in outpatient settings, naloxone prescribing is also relevant in the postsurgical setting.5 Many surgical patients are at risk for opioid overdose and data from the Veterans Health Administration (VHA) has shown that risk of opioid overdose is 11-fold higher in the 30 days following discharge from a surgical admission, when compared with the subsequent calendar year.6,7 This likely occurs due to new prescriptions or escalated doses of opioids following surgery. Overdose risk may be particularly relevant to orthopedic surgery as postoperative opioids are commonly prescribed.8 Patients undergoing total knee arthroplasty (TKA) may represent a vulnerable population to overdose as it is one of the most commonly performed surgeries for the treatment of chronic pain, and is frequently performed in older adults with medical comorbidities.9,10
Identifying patients at high risk for opioid overdose is important for targeted naloxone dispensing.5 A risk index for overdose or serious opioid-induced respiratory depression (RIOSORD) tool has been developed and validated in veteran and other populations to identify such patients.11 The RIOSORD tool classifies patients by risk level (1-10) and predicts probability of overdose or serious opioid-induced respiratory depression (OSORD). A patient’s level of risk is based on a weighted combination of the 15 independent risk factors most highly associated with OSORD, including comorbid conditions, prescription drug use, and health care utilization.12 Using the RIOSORD tool, the VHA Opioid Education and Naloxone Distribution (OEND) program is a risk mitigation initiative that aims to decrease opioid-related overdose morbidity and mortality. This is achieved via opioid overdose education for prevention, recognition, and response and includes outpatient naloxone prescription.13,14
Despite the comprehensive OEND program, there exists very little data to guide postsurgical naloxone prescribing. The prevalence of known risk factors for overdose in surgical patients remains unknown, as does the prevalence of perioperative naloxone distribution. Understanding overdose risk factors and naloxone prescribing patterns in surgical patients may identify potential targets for OEND efforts. This study retrospectively estimated RIOSORD scores for TKA patients between 2013 to 2016 and described naloxone distribution based on RIOSORD scores and risk factors.
Methods
We identified patients who had undergone primary TKA at VHA hospitals using Current Procedural Terminology (CPT), International Classification of Diseases, Ninth Revision (ICD-9) procedure codes, and data extracted from the VHA Corporate Data Warehouse (CDW) of electronic health records (EHRs). Our study was granted approval with exemption from informed consent by the Durham Veteran Affairs Healthcare System Institutional Review Board.
This retrospective cohort study included all veterans who underwent elective primary TKA from January 1, 2013 through December 31, 2016. We excluded patients who died before discharge.
Outcomes
Our primary outcome was being dispensed an outpatient naloxone prescription following TKA. Naloxone dispensing was identified by examining CDW outpatient pharmacy records with a final dispense date from 1 year before surgery through 7 days after discharge following TKA. To exclude naloxone administration that may have been given in a clinic, prescription data included only records with an outpatient prescription copay. Naloxone dispensing in the year before surgery was chosen to estimate likely preoperative possession of naloxone which could be available in the postoperative period. Naloxone dispensing until 7 days after discharge was chosen to identify any new dispensing that would be available in the postoperative period. These outcomes were examined over the study time frame on an annual basis.
Patient Factors
Demographic variables included age, sex, and race/ethnicity. Independent risk factors for overdose from RIOSORD were identified for each patient.15 These risk factors included comorbidities (opioid use disorder, schizophrenia, bipolar disorder, liver disease, chronic kidney disease, sleep apnea, or lung disease) and prescription drug use (use of opioids, benzodiazepines, long-acting opioids, ≥ 50 MEDD or ≥ 100 MEDD). ICD-9 and ICD-10 diagnosis codes were used to identify comorbidities. Risk classes on day of surgery were identified using a RIOSORD algorithm code. Consistent with the display of RIOSORD risk classes on the VHA Academic Detailing Service OEND risk report, patients were grouped into 3 groups based on their RIOSORD score: classes 1 to 4 (low risk), 5 to 7 (moderate risk), and 8 to 10 (high risk).
Descriptive statistics were used to summarize data on patient demographics, RIOSORD risk factors, overdose events, and naloxone dispensing over time.
Results
The study cohort included 38,011 veterans who underwent primary TKA in the VHA between January 1, 2013 and December 30, 2016. In this cohort, the mean age was 65 years, 93% were male, and 77% were White patients (Table 1). The most common comorbidities were lung disease in 9170 (24.1%) patients, sleep apnea in 6630 (17.4%) patients, chronic kidney disease in 4036 (10.6%) patients, liver disease in 2822 (7.4%) patients, and bipolar disorder in 1748 (4.6%) patients.
In 2013, 63.1% of patients presenting for surgery were actively prescribed opioids. By 2016, this decreased to 50.5%. Benzodiazepine use decreased from 13.2 to 8.8% and long-acting opioid use decreased from 8.5 to 5.8% over the same period. Patients taking ≥ 50 MEDD decreased from 8.0 to 5.3% and patients taking ≥ 100 MEDD decreased from 3.3 to 2.2%. The prevalence of moderate-risk patients decreased from 2.5 to 1.6% and high-risk patients decreased from 0.8 to 0.6% (Figure 1). Cumulatively, the prevalence of presenting with either moderate or high risk of overdose decreased from 3.3 to 2.2% between 2013 to 2016.
Naloxone Dispensing
In 2013, naloxone was not dispensed to any patients at moderate or high risk for overdose between 365 days prior to surgery until 7 days after discharge (Table 2 and Figure 2). Low-risk group naloxone dispensing increased to 2 (0.0%) in 2014, to 13 (0.1%), in 2015, and to 86 (0.9%) in 2016. Moderate-risk group naloxone dispensing remained at 0 (0.0%) in 2014, but increased to 8 (3.5%) in 2015, and to 18 (10.9%) in 2016. High-risk group naloxone dispensing remained at 0 (0.0%) in 2014, but increased to 5 (5.8%) in 2015, and to 8 (12.7%) in 2016 (Figure 3).
Discussion
Our data demonstrate that patients presenting for TKA between 2013 and 2016 routinely had individual risk factors for overdose related to either prescription drug use or comorbidities. We also show that, although the number of patients at moderate and high risk for opioid overdose is decreasing, 2.2% of TKA patients remain at moderate or high risk for opioid overdose based on a weighted combination of these individual risk factors using RIOSORD. As demand for primary TKA is projected to grow to 3.5 million procedures by 2030, using prevalence from 2016, we estimate that 76,560 patients may present for TKA across the US with moderate or high risk for opioid overdose.9 Following discharge, this risk may be even higher as this estimate does not yet account for postoperative opioid use. We demonstrate that through a VHA OEND initiative, naloxone distribution increased and appeared to be targeted to those most at risk using a simple validated tool like RIOSORD.
Presence of an individual risk factor for overdose was present in as many as 63.1% of patients presenting for TKA, as was seen in 2013 with preoperative opioid use. The 3 highest scoring prescription use–related risk factors in RIOSORD are use of opioids ≥ 100 MEDD (16 points), ≥ 50 MEDD (9 points), and long-acting formulations (9 points). All 3 decreased in prevalence over the study period but by 2016 were still seen in 2.2% for ≥ 100 MEDD, 5.3% for ≥ 50 MEDD, and 5.8% for long-acting opioids. This decrease was not surprising given implementation of a VHA-wide opioid safety initiative and the OEND program, but this could also be related to changes in patient selection for surgery in the context of increased awareness of the opioid epidemic. Despite the trend toward safer opioid prescribing, by 2016 over half of patients (50.5%) who presented for TKA were already taking opioids, with 10.6% (543 of 5127) on doses ≥ 50 MEDD.
We observed a decrease in RIOSORD risk each year, consistent with decreasing prescription-related risk factors over time. This was most obvious in the moderate-risk group. It is unclear why a similar decrease was not as obvious in the high-risk group, but this in part may be due to the already low numbers of patients in the high-risk group. This may also represent the high-risk group being somewhat resistant to the initiatives that shifted moderate-risk patients to the low-risk group. There were proportionately more patients in the moderate- and high-risk groups in the original RIOSORD population than in our surgical population, which may be attributed to the fewer comorbidities seen in our surgical population, as well as the higher opioid-prescribing patterns seen prior to the VA OEND initiative.12
Naloxone prescribing was rare prior to the OEND initiative and increased from 2013 to 2016. Increases were most marked in those in moderate- and high-risk groups, although naloxone prescribing also increased among the low-risk group. Integration of RIOSORD stratification into the OEND initiative likely played a role in targeting increased access to naloxone among those at highest risk of overdose. Naloxone dispensing increased for every group, although a significant proportion of moderate- and high-risk patients, 89.1% and 87.3%, respectively, were still not dispensed naloxone by 2016. Moreover, our estimates of perioperative naloxone access were likely an overestimate by including patients dispensed naloxone up to 1 year before surgery until 7 days after surgery. The aim was to include patients who may not have been prescribed naloxone postoperatively because of an existing naloxone prescription at home. Perioperative naloxone access estimates would have been even lower if a narrower window had been used to approximate perioperative access. This identifies an important gap between those who may benefit from naloxone dispensing and those who received naloxone. This in part may be because OEND has not been implemented as routinely in surgical settings as other settings (eg, primary care). OEND efforts may more effectively increase naloxone prescribing among surgical patients if these efforts were targeted at surgical and anesthesia departments. Given that the Comprehensive Addiction and Recovery Act of 2016 requires an assessment of patient risk prior to opioid prescribing and VHA efforts to increase utilization of tools like the Stratification Tool for Opioid Risk Mitigation (STORM), which estimates patient risk when initiating an opioid prescription and includes naloxone as one of many risk mitigation strategies, we anticipate that rates of naloxone prescribing will increase over time.
Limitations
Our study captures a large number of patients across VHA hospitals of varying size nationwide, including a mix of those with and without academic medical center affiliations. This veteran population may not represent the US commercially insured population (CIP). Zedler and colleagues highlighted the differences in prevalence of individual risk factors: notably, the CIP had a substantially higher proportion of females and younger patients.11 VHA had a greater prevalence of common chronic conditions associated with older age. The frequency of opioid dependence was similar among CIP and VHA. However, substance abuse and nonopioid substance dependence diagnoses were 4-fold more frequent among VHA controls as CIP controls. Prescribing of all opioids, except morphine and methadone, was substantially greater in CIP than in VHA.11 Despite a difference in individual risk factors, a CIP-specific RIOSORD has been validated and can be used outside of the VHA to obviate the limitations of the VHA-specific RIOSORD.11
Other limitations include our estimation of naloxone access. We do not know whether naloxone was administered or have a reliable estimate of overdose incidence in this postoperative TKA population. Also, it is important to note that RIOSORD was not developed for surgical patients. The use of RIOSORD in a postoperative population likely underestimates risk of opioid overdose due to the frequent prescriptions of new opioids or escalation of existing MEDD to the postoperative patient. Our study was also retrospective in nature and reliant on accurate coding of patient risk factors. It is possible that comorbidities were not accurately identified by EHR and therefore subject to inconsistency.
Conclusions
Veterans presenting for TKA routinely have risk factors for opioid overdose. We observed a trend toward decreasing overdose risk which coincided with the Opioid Safety and OEND initiatives within the VHA. We also observed an increase in naloxone prescription for moderate- and high-risk patients undergoing TKA, although most of these patients still did not receive naloxone as of 2016. More research is needed to refine and validate the RIOSORD score for surgical populations. Expanding initiatives such as OEND to include surgical patients presents an opportunity to improve access to naloxone for postoperative patients that may help reduce opioid overdose in this population.
1. Rudd RA, Seth P, David F, Scholl L. Increases in drug and opioid-involved overdose deaths - United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2016;65(50-51):1445-1452. Published 2016 Dec 30. doi:10.15585/mmwr.mm655051e1
2. Wilson N, Kariisa M, Seth P, Smith H, Davis NL. Drug and opioid-involved overdose deaths - United States, 2017-2018. MMWR Morb Mortal Wkly Rep. 2020;69(11):290-297. doi:10.15585/mmwr.mm6911a4
3. Walley AY, Xuan Z, Hackman HH, et al. Opioid overdose rates and implementation of overdose education and nasal naloxone distribution in Massachusetts: interrupted time series analysis. BMJ. Jan 30 2013;346:f174. doi:10.1136/bmj.f174
4. McClellan C, Lambdin BH, Ali MM, et al. Opioid-overdose laws association with opioid use and overdose mortality. Addict Behav. 2018;86:90-95. doi:10.1016/j.addbeh.2018.03.014
5. Dowell D, Haegerich TM, Chou R. CDC Guideline for prescribing opioids for chronic pain--United States, 2016. JAMA. 2016;315(15):1624-1645. doi:10.1001/jama.2016.1464
6. Brat GA, Agniel D, Beam A, et al. Postsurgical prescriptions for opioid naive patients and association with overdose and misuse: retrospective cohort study. BMJ. 2018;360:j5790. Published 2018 Jan 17. doi:10.1136/bmj.j5790
7. Mudumbai SC, Lewis ET, Oliva EM, et al. Overdose risk associated with opioid use upon hospital discharge in Veterans Health Administration surgical patients. Pain Med. 2019;20(5):1020-1031. doi:10.1093/pm/pny150
8. Hsia HL, Takemoto S, van de Ven T, et al. Acute pain is associated with chronic opioid use after total knee arthroplasty. Reg Anesth Pain Med. 2018;43(7):705-711. doi:10.1097/AAP.0000000000000831
9. Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785. doi:10.2106/JBJS.F.00222
10. Kurtz SM, Ong KL, Lau E, Bozic KJ. Impact of the economic downturn on total joint replacement demand in the United States: updated projections to 2021. J Bone Joint Surg Am. 2014;96(8):624-630. doi:10.2106/JBJS.M.00285
11. Zedler BK, Saunders WB, Joyce AR, Vick CC, Murrelle EL. Validation of a screening risk index for serious prescription opioid-induced respiratory depression or overdose in a US commercial health plan claims database. Pain Med. 2018;19(1):68-78. doi:10.1093/pm/pnx009
12. Zedler B, Xie L, Wang L, et al. Development of a risk index for serious prescription opioid-induced respiratory depression or overdose in Veterans Health Administration patients. Pain Med. 2015;16(8):1566-79. doi:10.1111/pme.12777
13. Oliva EM, Bowe T, Tavakoli S, et al. Development and applications of the Veterans Health Administration’s Stratification Tool for Opioid Risk Mitigation (STORM) to improve opioid safety and prevent overdose and suicide. Psychol Serv. 2017;14(1):34-49. doi:10.1037/ser0000099
14. Oliva EM, Christopher MLD, Wells D, et al. Opioid overdose education and naloxone distribution: development of the Veterans Health Administration’s national program. J Am Pharm Assoc (2003). 2017;57(2S):S168-S179.e4. doi:10.1016/j.japh.2017.01.022
15. Noël PH, Copeland LA, Perrin RA, et al. VHA Corporate Data Warehouse height and weight data: opportunities and challenges for health services research. J Rehabil Res Dev. 2010;47(8):739-750. doi:10.1682/jrrd.2009.08.0110
Opioid overdose is a major public health challenge, with recent reports estimating 41 deaths per day in the United States from prescription opioid overdose.1,2 Prescribing naloxone has increasingly been advocated to reduce the risk of opioid overdose for patients identified as high risk. Naloxone distribution has been shown to decrease the incidence of opioid overdoses in the general population.3,4 The Centers for Disease Control and Prevention (CDC) Guideline for Prescribing Opioids for Chronic Pain recommends considering naloxone prescription for patients with a history of overdose or substance use disorder, opioid dosages ≥ 50 morphine equivalent daily dose (MEDD), and concurrent use of benzodiazepines.5
Although the CDC guidelines are intended for primary care clinicians in outpatient settings, naloxone prescribing is also relevant in the postsurgical setting.5 Many surgical patients are at risk for opioid overdose and data from the Veterans Health Administration (VHA) has shown that risk of opioid overdose is 11-fold higher in the 30 days following discharge from a surgical admission, when compared with the subsequent calendar year.6,7 This likely occurs due to new prescriptions or escalated doses of opioids following surgery. Overdose risk may be particularly relevant to orthopedic surgery as postoperative opioids are commonly prescribed.8 Patients undergoing total knee arthroplasty (TKA) may represent a vulnerable population to overdose as it is one of the most commonly performed surgeries for the treatment of chronic pain, and is frequently performed in older adults with medical comorbidities.9,10
Identifying patients at high risk for opioid overdose is important for targeted naloxone dispensing.5 A risk index for overdose or serious opioid-induced respiratory depression (RIOSORD) tool has been developed and validated in veteran and other populations to identify such patients.11 The RIOSORD tool classifies patients by risk level (1-10) and predicts probability of overdose or serious opioid-induced respiratory depression (OSORD). A patient’s level of risk is based on a weighted combination of the 15 independent risk factors most highly associated with OSORD, including comorbid conditions, prescription drug use, and health care utilization.12 Using the RIOSORD tool, the VHA Opioid Education and Naloxone Distribution (OEND) program is a risk mitigation initiative that aims to decrease opioid-related overdose morbidity and mortality. This is achieved via opioid overdose education for prevention, recognition, and response and includes outpatient naloxone prescription.13,14
Despite the comprehensive OEND program, there exists very little data to guide postsurgical naloxone prescribing. The prevalence of known risk factors for overdose in surgical patients remains unknown, as does the prevalence of perioperative naloxone distribution. Understanding overdose risk factors and naloxone prescribing patterns in surgical patients may identify potential targets for OEND efforts. This study retrospectively estimated RIOSORD scores for TKA patients between 2013 to 2016 and described naloxone distribution based on RIOSORD scores and risk factors.
Methods
We identified patients who had undergone primary TKA at VHA hospitals using Current Procedural Terminology (CPT), International Classification of Diseases, Ninth Revision (ICD-9) procedure codes, and data extracted from the VHA Corporate Data Warehouse (CDW) of electronic health records (EHRs). Our study was granted approval with exemption from informed consent by the Durham Veteran Affairs Healthcare System Institutional Review Board.
This retrospective cohort study included all veterans who underwent elective primary TKA from January 1, 2013 through December 31, 2016. We excluded patients who died before discharge.
Outcomes
Our primary outcome was being dispensed an outpatient naloxone prescription following TKA. Naloxone dispensing was identified by examining CDW outpatient pharmacy records with a final dispense date from 1 year before surgery through 7 days after discharge following TKA. To exclude naloxone administration that may have been given in a clinic, prescription data included only records with an outpatient prescription copay. Naloxone dispensing in the year before surgery was chosen to estimate likely preoperative possession of naloxone which could be available in the postoperative period. Naloxone dispensing until 7 days after discharge was chosen to identify any new dispensing that would be available in the postoperative period. These outcomes were examined over the study time frame on an annual basis.
Patient Factors
Demographic variables included age, sex, and race/ethnicity. Independent risk factors for overdose from RIOSORD were identified for each patient.15 These risk factors included comorbidities (opioid use disorder, schizophrenia, bipolar disorder, liver disease, chronic kidney disease, sleep apnea, or lung disease) and prescription drug use (use of opioids, benzodiazepines, long-acting opioids, ≥ 50 MEDD or ≥ 100 MEDD). ICD-9 and ICD-10 diagnosis codes were used to identify comorbidities. Risk classes on day of surgery were identified using a RIOSORD algorithm code. Consistent with the display of RIOSORD risk classes on the VHA Academic Detailing Service OEND risk report, patients were grouped into 3 groups based on their RIOSORD score: classes 1 to 4 (low risk), 5 to 7 (moderate risk), and 8 to 10 (high risk).
Descriptive statistics were used to summarize data on patient demographics, RIOSORD risk factors, overdose events, and naloxone dispensing over time.
Results
The study cohort included 38,011 veterans who underwent primary TKA in the VHA between January 1, 2013 and December 30, 2016. In this cohort, the mean age was 65 years, 93% were male, and 77% were White patients (Table 1). The most common comorbidities were lung disease in 9170 (24.1%) patients, sleep apnea in 6630 (17.4%) patients, chronic kidney disease in 4036 (10.6%) patients, liver disease in 2822 (7.4%) patients, and bipolar disorder in 1748 (4.6%) patients.
In 2013, 63.1% of patients presenting for surgery were actively prescribed opioids. By 2016, this decreased to 50.5%. Benzodiazepine use decreased from 13.2 to 8.8% and long-acting opioid use decreased from 8.5 to 5.8% over the same period. Patients taking ≥ 50 MEDD decreased from 8.0 to 5.3% and patients taking ≥ 100 MEDD decreased from 3.3 to 2.2%. The prevalence of moderate-risk patients decreased from 2.5 to 1.6% and high-risk patients decreased from 0.8 to 0.6% (Figure 1). Cumulatively, the prevalence of presenting with either moderate or high risk of overdose decreased from 3.3 to 2.2% between 2013 to 2016.
Naloxone Dispensing
In 2013, naloxone was not dispensed to any patients at moderate or high risk for overdose between 365 days prior to surgery until 7 days after discharge (Table 2 and Figure 2). Low-risk group naloxone dispensing increased to 2 (0.0%) in 2014, to 13 (0.1%), in 2015, and to 86 (0.9%) in 2016. Moderate-risk group naloxone dispensing remained at 0 (0.0%) in 2014, but increased to 8 (3.5%) in 2015, and to 18 (10.9%) in 2016. High-risk group naloxone dispensing remained at 0 (0.0%) in 2014, but increased to 5 (5.8%) in 2015, and to 8 (12.7%) in 2016 (Figure 3).
Discussion
Our data demonstrate that patients presenting for TKA between 2013 and 2016 routinely had individual risk factors for overdose related to either prescription drug use or comorbidities. We also show that, although the number of patients at moderate and high risk for opioid overdose is decreasing, 2.2% of TKA patients remain at moderate or high risk for opioid overdose based on a weighted combination of these individual risk factors using RIOSORD. As demand for primary TKA is projected to grow to 3.5 million procedures by 2030, using prevalence from 2016, we estimate that 76,560 patients may present for TKA across the US with moderate or high risk for opioid overdose.9 Following discharge, this risk may be even higher as this estimate does not yet account for postoperative opioid use. We demonstrate that through a VHA OEND initiative, naloxone distribution increased and appeared to be targeted to those most at risk using a simple validated tool like RIOSORD.
Presence of an individual risk factor for overdose was present in as many as 63.1% of patients presenting for TKA, as was seen in 2013 with preoperative opioid use. The 3 highest scoring prescription use–related risk factors in RIOSORD are use of opioids ≥ 100 MEDD (16 points), ≥ 50 MEDD (9 points), and long-acting formulations (9 points). All 3 decreased in prevalence over the study period but by 2016 were still seen in 2.2% for ≥ 100 MEDD, 5.3% for ≥ 50 MEDD, and 5.8% for long-acting opioids. This decrease was not surprising given implementation of a VHA-wide opioid safety initiative and the OEND program, but this could also be related to changes in patient selection for surgery in the context of increased awareness of the opioid epidemic. Despite the trend toward safer opioid prescribing, by 2016 over half of patients (50.5%) who presented for TKA were already taking opioids, with 10.6% (543 of 5127) on doses ≥ 50 MEDD.
We observed a decrease in RIOSORD risk each year, consistent with decreasing prescription-related risk factors over time. This was most obvious in the moderate-risk group. It is unclear why a similar decrease was not as obvious in the high-risk group, but this in part may be due to the already low numbers of patients in the high-risk group. This may also represent the high-risk group being somewhat resistant to the initiatives that shifted moderate-risk patients to the low-risk group. There were proportionately more patients in the moderate- and high-risk groups in the original RIOSORD population than in our surgical population, which may be attributed to the fewer comorbidities seen in our surgical population, as well as the higher opioid-prescribing patterns seen prior to the VA OEND initiative.12
Naloxone prescribing was rare prior to the OEND initiative and increased from 2013 to 2016. Increases were most marked in those in moderate- and high-risk groups, although naloxone prescribing also increased among the low-risk group. Integration of RIOSORD stratification into the OEND initiative likely played a role in targeting increased access to naloxone among those at highest risk of overdose. Naloxone dispensing increased for every group, although a significant proportion of moderate- and high-risk patients, 89.1% and 87.3%, respectively, were still not dispensed naloxone by 2016. Moreover, our estimates of perioperative naloxone access were likely an overestimate by including patients dispensed naloxone up to 1 year before surgery until 7 days after surgery. The aim was to include patients who may not have been prescribed naloxone postoperatively because of an existing naloxone prescription at home. Perioperative naloxone access estimates would have been even lower if a narrower window had been used to approximate perioperative access. This identifies an important gap between those who may benefit from naloxone dispensing and those who received naloxone. This in part may be because OEND has not been implemented as routinely in surgical settings as other settings (eg, primary care). OEND efforts may more effectively increase naloxone prescribing among surgical patients if these efforts were targeted at surgical and anesthesia departments. Given that the Comprehensive Addiction and Recovery Act of 2016 requires an assessment of patient risk prior to opioid prescribing and VHA efforts to increase utilization of tools like the Stratification Tool for Opioid Risk Mitigation (STORM), which estimates patient risk when initiating an opioid prescription and includes naloxone as one of many risk mitigation strategies, we anticipate that rates of naloxone prescribing will increase over time.
Limitations
Our study captures a large number of patients across VHA hospitals of varying size nationwide, including a mix of those with and without academic medical center affiliations. This veteran population may not represent the US commercially insured population (CIP). Zedler and colleagues highlighted the differences in prevalence of individual risk factors: notably, the CIP had a substantially higher proportion of females and younger patients.11 VHA had a greater prevalence of common chronic conditions associated with older age. The frequency of opioid dependence was similar among CIP and VHA. However, substance abuse and nonopioid substance dependence diagnoses were 4-fold more frequent among VHA controls as CIP controls. Prescribing of all opioids, except morphine and methadone, was substantially greater in CIP than in VHA.11 Despite a difference in individual risk factors, a CIP-specific RIOSORD has been validated and can be used outside of the VHA to obviate the limitations of the VHA-specific RIOSORD.11
Other limitations include our estimation of naloxone access. We do not know whether naloxone was administered or have a reliable estimate of overdose incidence in this postoperative TKA population. Also, it is important to note that RIOSORD was not developed for surgical patients. The use of RIOSORD in a postoperative population likely underestimates risk of opioid overdose due to the frequent prescriptions of new opioids or escalation of existing MEDD to the postoperative patient. Our study was also retrospective in nature and reliant on accurate coding of patient risk factors. It is possible that comorbidities were not accurately identified by EHR and therefore subject to inconsistency.
Conclusions
Veterans presenting for TKA routinely have risk factors for opioid overdose. We observed a trend toward decreasing overdose risk which coincided with the Opioid Safety and OEND initiatives within the VHA. We also observed an increase in naloxone prescription for moderate- and high-risk patients undergoing TKA, although most of these patients still did not receive naloxone as of 2016. More research is needed to refine and validate the RIOSORD score for surgical populations. Expanding initiatives such as OEND to include surgical patients presents an opportunity to improve access to naloxone for postoperative patients that may help reduce opioid overdose in this population.
Opioid overdose is a major public health challenge, with recent reports estimating 41 deaths per day in the United States from prescription opioid overdose.1,2 Prescribing naloxone has increasingly been advocated to reduce the risk of opioid overdose for patients identified as high risk. Naloxone distribution has been shown to decrease the incidence of opioid overdoses in the general population.3,4 The Centers for Disease Control and Prevention (CDC) Guideline for Prescribing Opioids for Chronic Pain recommends considering naloxone prescription for patients with a history of overdose or substance use disorder, opioid dosages ≥ 50 morphine equivalent daily dose (MEDD), and concurrent use of benzodiazepines.5
Although the CDC guidelines are intended for primary care clinicians in outpatient settings, naloxone prescribing is also relevant in the postsurgical setting.5 Many surgical patients are at risk for opioid overdose and data from the Veterans Health Administration (VHA) has shown that risk of opioid overdose is 11-fold higher in the 30 days following discharge from a surgical admission, when compared with the subsequent calendar year.6,7 This likely occurs due to new prescriptions or escalated doses of opioids following surgery. Overdose risk may be particularly relevant to orthopedic surgery as postoperative opioids are commonly prescribed.8 Patients undergoing total knee arthroplasty (TKA) may represent a vulnerable population to overdose as it is one of the most commonly performed surgeries for the treatment of chronic pain, and is frequently performed in older adults with medical comorbidities.9,10
Identifying patients at high risk for opioid overdose is important for targeted naloxone dispensing.5 A risk index for overdose or serious opioid-induced respiratory depression (RIOSORD) tool has been developed and validated in veteran and other populations to identify such patients.11 The RIOSORD tool classifies patients by risk level (1-10) and predicts probability of overdose or serious opioid-induced respiratory depression (OSORD). A patient’s level of risk is based on a weighted combination of the 15 independent risk factors most highly associated with OSORD, including comorbid conditions, prescription drug use, and health care utilization.12 Using the RIOSORD tool, the VHA Opioid Education and Naloxone Distribution (OEND) program is a risk mitigation initiative that aims to decrease opioid-related overdose morbidity and mortality. This is achieved via opioid overdose education for prevention, recognition, and response and includes outpatient naloxone prescription.13,14
Despite the comprehensive OEND program, there exists very little data to guide postsurgical naloxone prescribing. The prevalence of known risk factors for overdose in surgical patients remains unknown, as does the prevalence of perioperative naloxone distribution. Understanding overdose risk factors and naloxone prescribing patterns in surgical patients may identify potential targets for OEND efforts. This study retrospectively estimated RIOSORD scores for TKA patients between 2013 to 2016 and described naloxone distribution based on RIOSORD scores and risk factors.
Methods
We identified patients who had undergone primary TKA at VHA hospitals using Current Procedural Terminology (CPT), International Classification of Diseases, Ninth Revision (ICD-9) procedure codes, and data extracted from the VHA Corporate Data Warehouse (CDW) of electronic health records (EHRs). Our study was granted approval with exemption from informed consent by the Durham Veteran Affairs Healthcare System Institutional Review Board.
This retrospective cohort study included all veterans who underwent elective primary TKA from January 1, 2013 through December 31, 2016. We excluded patients who died before discharge.
Outcomes
Our primary outcome was being dispensed an outpatient naloxone prescription following TKA. Naloxone dispensing was identified by examining CDW outpatient pharmacy records with a final dispense date from 1 year before surgery through 7 days after discharge following TKA. To exclude naloxone administration that may have been given in a clinic, prescription data included only records with an outpatient prescription copay. Naloxone dispensing in the year before surgery was chosen to estimate likely preoperative possession of naloxone which could be available in the postoperative period. Naloxone dispensing until 7 days after discharge was chosen to identify any new dispensing that would be available in the postoperative period. These outcomes were examined over the study time frame on an annual basis.
Patient Factors
Demographic variables included age, sex, and race/ethnicity. Independent risk factors for overdose from RIOSORD were identified for each patient.15 These risk factors included comorbidities (opioid use disorder, schizophrenia, bipolar disorder, liver disease, chronic kidney disease, sleep apnea, or lung disease) and prescription drug use (use of opioids, benzodiazepines, long-acting opioids, ≥ 50 MEDD or ≥ 100 MEDD). ICD-9 and ICD-10 diagnosis codes were used to identify comorbidities. Risk classes on day of surgery were identified using a RIOSORD algorithm code. Consistent with the display of RIOSORD risk classes on the VHA Academic Detailing Service OEND risk report, patients were grouped into 3 groups based on their RIOSORD score: classes 1 to 4 (low risk), 5 to 7 (moderate risk), and 8 to 10 (high risk).
Descriptive statistics were used to summarize data on patient demographics, RIOSORD risk factors, overdose events, and naloxone dispensing over time.
Results
The study cohort included 38,011 veterans who underwent primary TKA in the VHA between January 1, 2013 and December 30, 2016. In this cohort, the mean age was 65 years, 93% were male, and 77% were White patients (Table 1). The most common comorbidities were lung disease in 9170 (24.1%) patients, sleep apnea in 6630 (17.4%) patients, chronic kidney disease in 4036 (10.6%) patients, liver disease in 2822 (7.4%) patients, and bipolar disorder in 1748 (4.6%) patients.
In 2013, 63.1% of patients presenting for surgery were actively prescribed opioids. By 2016, this decreased to 50.5%. Benzodiazepine use decreased from 13.2 to 8.8% and long-acting opioid use decreased from 8.5 to 5.8% over the same period. Patients taking ≥ 50 MEDD decreased from 8.0 to 5.3% and patients taking ≥ 100 MEDD decreased from 3.3 to 2.2%. The prevalence of moderate-risk patients decreased from 2.5 to 1.6% and high-risk patients decreased from 0.8 to 0.6% (Figure 1). Cumulatively, the prevalence of presenting with either moderate or high risk of overdose decreased from 3.3 to 2.2% between 2013 to 2016.
Naloxone Dispensing
In 2013, naloxone was not dispensed to any patients at moderate or high risk for overdose between 365 days prior to surgery until 7 days after discharge (Table 2 and Figure 2). Low-risk group naloxone dispensing increased to 2 (0.0%) in 2014, to 13 (0.1%), in 2015, and to 86 (0.9%) in 2016. Moderate-risk group naloxone dispensing remained at 0 (0.0%) in 2014, but increased to 8 (3.5%) in 2015, and to 18 (10.9%) in 2016. High-risk group naloxone dispensing remained at 0 (0.0%) in 2014, but increased to 5 (5.8%) in 2015, and to 8 (12.7%) in 2016 (Figure 3).
Discussion
Our data demonstrate that patients presenting for TKA between 2013 and 2016 routinely had individual risk factors for overdose related to either prescription drug use or comorbidities. We also show that, although the number of patients at moderate and high risk for opioid overdose is decreasing, 2.2% of TKA patients remain at moderate or high risk for opioid overdose based on a weighted combination of these individual risk factors using RIOSORD. As demand for primary TKA is projected to grow to 3.5 million procedures by 2030, using prevalence from 2016, we estimate that 76,560 patients may present for TKA across the US with moderate or high risk for opioid overdose.9 Following discharge, this risk may be even higher as this estimate does not yet account for postoperative opioid use. We demonstrate that through a VHA OEND initiative, naloxone distribution increased and appeared to be targeted to those most at risk using a simple validated tool like RIOSORD.
Presence of an individual risk factor for overdose was present in as many as 63.1% of patients presenting for TKA, as was seen in 2013 with preoperative opioid use. The 3 highest scoring prescription use–related risk factors in RIOSORD are use of opioids ≥ 100 MEDD (16 points), ≥ 50 MEDD (9 points), and long-acting formulations (9 points). All 3 decreased in prevalence over the study period but by 2016 were still seen in 2.2% for ≥ 100 MEDD, 5.3% for ≥ 50 MEDD, and 5.8% for long-acting opioids. This decrease was not surprising given implementation of a VHA-wide opioid safety initiative and the OEND program, but this could also be related to changes in patient selection for surgery in the context of increased awareness of the opioid epidemic. Despite the trend toward safer opioid prescribing, by 2016 over half of patients (50.5%) who presented for TKA were already taking opioids, with 10.6% (543 of 5127) on doses ≥ 50 MEDD.
We observed a decrease in RIOSORD risk each year, consistent with decreasing prescription-related risk factors over time. This was most obvious in the moderate-risk group. It is unclear why a similar decrease was not as obvious in the high-risk group, but this in part may be due to the already low numbers of patients in the high-risk group. This may also represent the high-risk group being somewhat resistant to the initiatives that shifted moderate-risk patients to the low-risk group. There were proportionately more patients in the moderate- and high-risk groups in the original RIOSORD population than in our surgical population, which may be attributed to the fewer comorbidities seen in our surgical population, as well as the higher opioid-prescribing patterns seen prior to the VA OEND initiative.12
Naloxone prescribing was rare prior to the OEND initiative and increased from 2013 to 2016. Increases were most marked in those in moderate- and high-risk groups, although naloxone prescribing also increased among the low-risk group. Integration of RIOSORD stratification into the OEND initiative likely played a role in targeting increased access to naloxone among those at highest risk of overdose. Naloxone dispensing increased for every group, although a significant proportion of moderate- and high-risk patients, 89.1% and 87.3%, respectively, were still not dispensed naloxone by 2016. Moreover, our estimates of perioperative naloxone access were likely an overestimate by including patients dispensed naloxone up to 1 year before surgery until 7 days after surgery. The aim was to include patients who may not have been prescribed naloxone postoperatively because of an existing naloxone prescription at home. Perioperative naloxone access estimates would have been even lower if a narrower window had been used to approximate perioperative access. This identifies an important gap between those who may benefit from naloxone dispensing and those who received naloxone. This in part may be because OEND has not been implemented as routinely in surgical settings as other settings (eg, primary care). OEND efforts may more effectively increase naloxone prescribing among surgical patients if these efforts were targeted at surgical and anesthesia departments. Given that the Comprehensive Addiction and Recovery Act of 2016 requires an assessment of patient risk prior to opioid prescribing and VHA efforts to increase utilization of tools like the Stratification Tool for Opioid Risk Mitigation (STORM), which estimates patient risk when initiating an opioid prescription and includes naloxone as one of many risk mitigation strategies, we anticipate that rates of naloxone prescribing will increase over time.
Limitations
Our study captures a large number of patients across VHA hospitals of varying size nationwide, including a mix of those with and without academic medical center affiliations. This veteran population may not represent the US commercially insured population (CIP). Zedler and colleagues highlighted the differences in prevalence of individual risk factors: notably, the CIP had a substantially higher proportion of females and younger patients.11 VHA had a greater prevalence of common chronic conditions associated with older age. The frequency of opioid dependence was similar among CIP and VHA. However, substance abuse and nonopioid substance dependence diagnoses were 4-fold more frequent among VHA controls as CIP controls. Prescribing of all opioids, except morphine and methadone, was substantially greater in CIP than in VHA.11 Despite a difference in individual risk factors, a CIP-specific RIOSORD has been validated and can be used outside of the VHA to obviate the limitations of the VHA-specific RIOSORD.11
Other limitations include our estimation of naloxone access. We do not know whether naloxone was administered or have a reliable estimate of overdose incidence in this postoperative TKA population. Also, it is important to note that RIOSORD was not developed for surgical patients. The use of RIOSORD in a postoperative population likely underestimates risk of opioid overdose due to the frequent prescriptions of new opioids or escalation of existing MEDD to the postoperative patient. Our study was also retrospective in nature and reliant on accurate coding of patient risk factors. It is possible that comorbidities were not accurately identified by EHR and therefore subject to inconsistency.
Conclusions
Veterans presenting for TKA routinely have risk factors for opioid overdose. We observed a trend toward decreasing overdose risk which coincided with the Opioid Safety and OEND initiatives within the VHA. We also observed an increase in naloxone prescription for moderate- and high-risk patients undergoing TKA, although most of these patients still did not receive naloxone as of 2016. More research is needed to refine and validate the RIOSORD score for surgical populations. Expanding initiatives such as OEND to include surgical patients presents an opportunity to improve access to naloxone for postoperative patients that may help reduce opioid overdose in this population.
1. Rudd RA, Seth P, David F, Scholl L. Increases in drug and opioid-involved overdose deaths - United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2016;65(50-51):1445-1452. Published 2016 Dec 30. doi:10.15585/mmwr.mm655051e1
2. Wilson N, Kariisa M, Seth P, Smith H, Davis NL. Drug and opioid-involved overdose deaths - United States, 2017-2018. MMWR Morb Mortal Wkly Rep. 2020;69(11):290-297. doi:10.15585/mmwr.mm6911a4
3. Walley AY, Xuan Z, Hackman HH, et al. Opioid overdose rates and implementation of overdose education and nasal naloxone distribution in Massachusetts: interrupted time series analysis. BMJ. Jan 30 2013;346:f174. doi:10.1136/bmj.f174
4. McClellan C, Lambdin BH, Ali MM, et al. Opioid-overdose laws association with opioid use and overdose mortality. Addict Behav. 2018;86:90-95. doi:10.1016/j.addbeh.2018.03.014
5. Dowell D, Haegerich TM, Chou R. CDC Guideline for prescribing opioids for chronic pain--United States, 2016. JAMA. 2016;315(15):1624-1645. doi:10.1001/jama.2016.1464
6. Brat GA, Agniel D, Beam A, et al. Postsurgical prescriptions for opioid naive patients and association with overdose and misuse: retrospective cohort study. BMJ. 2018;360:j5790. Published 2018 Jan 17. doi:10.1136/bmj.j5790
7. Mudumbai SC, Lewis ET, Oliva EM, et al. Overdose risk associated with opioid use upon hospital discharge in Veterans Health Administration surgical patients. Pain Med. 2019;20(5):1020-1031. doi:10.1093/pm/pny150
8. Hsia HL, Takemoto S, van de Ven T, et al. Acute pain is associated with chronic opioid use after total knee arthroplasty. Reg Anesth Pain Med. 2018;43(7):705-711. doi:10.1097/AAP.0000000000000831
9. Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785. doi:10.2106/JBJS.F.00222
10. Kurtz SM, Ong KL, Lau E, Bozic KJ. Impact of the economic downturn on total joint replacement demand in the United States: updated projections to 2021. J Bone Joint Surg Am. 2014;96(8):624-630. doi:10.2106/JBJS.M.00285
11. Zedler BK, Saunders WB, Joyce AR, Vick CC, Murrelle EL. Validation of a screening risk index for serious prescription opioid-induced respiratory depression or overdose in a US commercial health plan claims database. Pain Med. 2018;19(1):68-78. doi:10.1093/pm/pnx009
12. Zedler B, Xie L, Wang L, et al. Development of a risk index for serious prescription opioid-induced respiratory depression or overdose in Veterans Health Administration patients. Pain Med. 2015;16(8):1566-79. doi:10.1111/pme.12777
13. Oliva EM, Bowe T, Tavakoli S, et al. Development and applications of the Veterans Health Administration’s Stratification Tool for Opioid Risk Mitigation (STORM) to improve opioid safety and prevent overdose and suicide. Psychol Serv. 2017;14(1):34-49. doi:10.1037/ser0000099
14. Oliva EM, Christopher MLD, Wells D, et al. Opioid overdose education and naloxone distribution: development of the Veterans Health Administration’s national program. J Am Pharm Assoc (2003). 2017;57(2S):S168-S179.e4. doi:10.1016/j.japh.2017.01.022
15. Noël PH, Copeland LA, Perrin RA, et al. VHA Corporate Data Warehouse height and weight data: opportunities and challenges for health services research. J Rehabil Res Dev. 2010;47(8):739-750. doi:10.1682/jrrd.2009.08.0110
1. Rudd RA, Seth P, David F, Scholl L. Increases in drug and opioid-involved overdose deaths - United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2016;65(50-51):1445-1452. Published 2016 Dec 30. doi:10.15585/mmwr.mm655051e1
2. Wilson N, Kariisa M, Seth P, Smith H, Davis NL. Drug and opioid-involved overdose deaths - United States, 2017-2018. MMWR Morb Mortal Wkly Rep. 2020;69(11):290-297. doi:10.15585/mmwr.mm6911a4
3. Walley AY, Xuan Z, Hackman HH, et al. Opioid overdose rates and implementation of overdose education and nasal naloxone distribution in Massachusetts: interrupted time series analysis. BMJ. Jan 30 2013;346:f174. doi:10.1136/bmj.f174
4. McClellan C, Lambdin BH, Ali MM, et al. Opioid-overdose laws association with opioid use and overdose mortality. Addict Behav. 2018;86:90-95. doi:10.1016/j.addbeh.2018.03.014
5. Dowell D, Haegerich TM, Chou R. CDC Guideline for prescribing opioids for chronic pain--United States, 2016. JAMA. 2016;315(15):1624-1645. doi:10.1001/jama.2016.1464
6. Brat GA, Agniel D, Beam A, et al. Postsurgical prescriptions for opioid naive patients and association with overdose and misuse: retrospective cohort study. BMJ. 2018;360:j5790. Published 2018 Jan 17. doi:10.1136/bmj.j5790
7. Mudumbai SC, Lewis ET, Oliva EM, et al. Overdose risk associated with opioid use upon hospital discharge in Veterans Health Administration surgical patients. Pain Med. 2019;20(5):1020-1031. doi:10.1093/pm/pny150
8. Hsia HL, Takemoto S, van de Ven T, et al. Acute pain is associated with chronic opioid use after total knee arthroplasty. Reg Anesth Pain Med. 2018;43(7):705-711. doi:10.1097/AAP.0000000000000831
9. Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785. doi:10.2106/JBJS.F.00222
10. Kurtz SM, Ong KL, Lau E, Bozic KJ. Impact of the economic downturn on total joint replacement demand in the United States: updated projections to 2021. J Bone Joint Surg Am. 2014;96(8):624-630. doi:10.2106/JBJS.M.00285
11. Zedler BK, Saunders WB, Joyce AR, Vick CC, Murrelle EL. Validation of a screening risk index for serious prescription opioid-induced respiratory depression or overdose in a US commercial health plan claims database. Pain Med. 2018;19(1):68-78. doi:10.1093/pm/pnx009
12. Zedler B, Xie L, Wang L, et al. Development of a risk index for serious prescription opioid-induced respiratory depression or overdose in Veterans Health Administration patients. Pain Med. 2015;16(8):1566-79. doi:10.1111/pme.12777
13. Oliva EM, Bowe T, Tavakoli S, et al. Development and applications of the Veterans Health Administration’s Stratification Tool for Opioid Risk Mitigation (STORM) to improve opioid safety and prevent overdose and suicide. Psychol Serv. 2017;14(1):34-49. doi:10.1037/ser0000099
14. Oliva EM, Christopher MLD, Wells D, et al. Opioid overdose education and naloxone distribution: development of the Veterans Health Administration’s national program. J Am Pharm Assoc (2003). 2017;57(2S):S168-S179.e4. doi:10.1016/j.japh.2017.01.022
15. Noël PH, Copeland LA, Perrin RA, et al. VHA Corporate Data Warehouse height and weight data: opportunities and challenges for health services research. J Rehabil Res Dev. 2010;47(8):739-750. doi:10.1682/jrrd.2009.08.0110
Integrating Massage Therapy Into the Health Care of Female Veterans
There are approximately 2 million female veterans in the United States, representing about 10% of the veteran population.1 In 2015, 456,000 female veterans used the US Department of Veterans Affairs (VA) health care services. The VA predicts an increase in utilization over the next 20 years.2
Female veterans are more likely to have musculoskeletal disorder multimorbidity compared with male veterans and have higher rates of depressive and bipolar disorders, anxiety, and posttraumatic stress disorder (PTSD).3,4 Compared with male veterans, female veterans are younger, more likely to be unmarried and to have served during the wars in Iraq and Afghanistan.3 Fifty-five percent of women veterans vs 41% of men veterans have a service-connected disability, and a greater percentage of women veterans have a service connection rating > 50%.5 The top service-connected disabilities for women veterans are PTSD, major depressive disorder, migraines, and lumbosacral or cervical strain.2 In addition, one-third of women veterans using VA health care report experiencing military sexual trauma (MST).6 Military service may impact the health of female veterans both physically and mentally. Providing treatments and programs to improve their health and their health care experience are current VA priorities.
The VA is changing the way health care is conceptualized and delivered by implementing a holistic model of care known as Whole Health, which seeks to empower and equip patients to take charge of their health, blending conventional medicine with self-care and complementary and integrative health (CIH) approaches, such as massage therapy, yoga, acupuncture, and meditation.7 CIH therapies can help improve physical and mental health with little to no adverse effects.8-10
As part of the Whole Health initiative at the VA Ann Arbor Healthcare System (VAAAHS) in Michigan, the massage program was expanded in 2017 to offer relaxation massages to female veterans attending the women’s health clinic, which provides gynecologic care. Patients visiting a gynecology clinic often experience anxiety and pain related to invasive procedures and examinations. This is especially true for female veterans who experienced MST.11
VAAAHS has 1 staff massage therapist (MT). To expand the program to the women’s health clinic, volunteer licensed MTs were recruited and trained in specific procedures by the staff MT.
Several studies have demonstrated the effect of therapeutic massage on pain and anxiety in predominantly male veteran study populations, including veterans needing postsurgical and palliative care as well as those experiencing chronic pain and knee osteoarthritis.12-16 Little is known about the effects of massage therapy on female veterans. The purpose of this pilot study was to examine the effects of massage therapy among female veterans participating in the women’s health massage program.
Methods
The setting for this pre-post intervention study was VAAAHS. Veterans were called in advance by clinic staff and scheduled for 60-minute appointments either before or after their clinic appointment, depending on availability. MTs were instructed to provide relaxation massage using Swedish massage techniques with moderate pressure, avoiding deep pressure techniques.
The volunteer MTs gave the participants a survey to provide comments and to rate baseline pain and other symptoms prior to and following the massage. The MT left the room to provide privacy while completing the survey. The staff included the symptom data in the massage note as clinical outcomes and entered them into the electronic health record. Massages were given from October 1, 2017 to June 30, 2018. Data including symptom scores, demographics, the presence of chronic pain, mental health diagnoses, patient comments, and opioid use were abstracted from the electronic health record by 2 members of the study team and entered into an Excel database. This study was approved by the VAAAHS Institutional Review Board.
Study Measures
Pain intensity, pain unpleasantness (the affective component of pain), anxiety, shortness of breath, relaxation, and inner peace were rated pre- and postmassage on a 0 to 10 scale. Shortness of breath was included due to the relationship between breathing and anxiety. Inner peace was assessed to measure the calming effects of massage therapy. Beck and colleagues found the concept of inner peace was an important outcome of massage therapy.17 The scale anchors for pain intensity were “no pain” and “severe pain”; and “not at all unpleasant” and “as unpleasant as it can be” for pain unpleasantness. For anxiety, the anchors were “no anxiety” and “as anxious as I can be.” Anchors for relaxation and inner peace were reversed so that a 0 indicated low relaxation and inner peace while a 10 indicated the highest state of relaxation and inner peace.
Chronic pain was defined as pain existing for > 3 months. A history of chronic pain was determined from a review and synthesis of primary care and specialty care recorded diagnoses, patient concerns, and service-connected disabilities. The diagnoses included lumbosacral or cervical strain, chronic low back, joint (knee, shoulder, hip, ankle), neck, or pelvic pain, fibromyalgia, headache, migraine, osteoarthritis, and myofascial pain syndrome. The presence of mental health conditions, including depression, anxiety, bipolar disorders, and PTSD, were similarly determined by a review of mental health clinical notes. Sex was determined from the gynecology note.
Statistical Analysis
Means and medians were calculated for short-term changes in symptom scores. Due to skewness in the short-term changes, significance was tested using a nonparametric sign test. Significance was adjusted using the Bonferroni correction to protect the overall type I error level at 5% from multiple testing. We also assessed for differences in symptom changes in 4 subgroups, using an unadjusted general linear model: those with (1) chronic pain vs without; (2) an anxiety diagnosis vs without; (3) depression vs without; and (4) a PTSD diagnosis vs without. Data were analyzed using SPSS 25 and SAS 9.4.
Results
Results are based on the first massage received by 96 unique individuals (Table 1). Fifty-one (53%) patients were aged 21 to 40 years, and 45 (47%) were aged ≥ 41 years. Most participants (80%) had had a previous massage. Seven (7%) participants were currently on prescription opioids; 76 (79%) participants had a history of one or more chronic pain diagnoses (eg, back pain, migraine headaches, fibromyalgia) and 78 (81%) had a history of a mental health diagnosis (eg, depression, anxiety, PTSD). Massage sessions ranged from 30 to 60 minutes; most patients received massage therapy for 50 minutes.
Prior to massage, mean scores were 3.9 pain intensity, 3.7 pain unpleasantness, 3.8 anxiety, 1.0 shortness of breath, 4.0 relaxation, and 4.2 inner peace. Short-term changes in symptom scores are shown in Table 2. The mean score for pain intensity decreased by 1.9 points, pain unpleasantness by 2.0 points, anxiety by 2.4 points. The greatest change occurred for relaxation, which increased by 4.3 points. All changes in symptoms were statistically significant (P < .001). For subgroup comparisons
Verbal feedback and written comments about the massage experience were all favorable: No adverse events were reported.
Discussion
Massage therapy may be a useful treatment for female veterans experiencing chronic pain, anxiety disorders, depression, or situational anxiety related to gynecologic procedures. After receiving a relaxation massage, female veterans reported decreased pain intensity, pain unpleasantness, and anxiety while reporting increased relaxation and feelings of inner peace. The effects of massage were consistent for all the symptoms or characteristics assessed, suggesting that massage may act on the body in multiple ways.
These changes parallel those seen in a palliative care population primarily composed of male veterans.14 However, the female veterans in this cohort experienced greater changes in relaxation and feelings of inner peace, which may be partly due to relief of tension related to an upcoming stressful appointment. The large mean decrease in anxiety level among female veterans with PTSD is notable as well as the larger increase in inner peace in those with chronic pain.
Many patients expressed their gratitude for the massage and interest in having access to more massage therapy. Female patients who have experienced sexual trauma or other trauma may especially benefit from massage prior to painful, invasive gynecologic procedures. Anecdotally, 2 nurse chaperones in the clinic mentioned separately to the massage program supervisor that the massages helped some very anxious women better tolerate an invasive procedure that would have been otherwise extremely difficult.
Female veterans are more likely to have musculoskeletal issues after deployment and have higher rates of anxiety, PTSD, and depression compared with those of male veterans.3,4,18,19 Determining relationships between and causes of chronic pain, depression, and PTSD is very challenging but the increased prevalence of chronic pain and comorbid mental health conditions in female veterans may be partially related to MST or other trauma experiences.20-22 Female veterans are most likely to have more than one source of chronic pain.23-25 Female patients with chronic musculoskeletal pain report more pain-related disability.26 Furthermore, greater disability in the context of depression is reported by women with pain compared with those of men.27 Most (78%) female veterans in a primary care population reported chronic pain.23 Similarly, 79% of the female veterans in this study population had chronic pain and 81% had a history of mental health disorders, including depression, anxiety, and PTSD.
Studies have shown that massage therapy improves pain in populations experiencing chronic low back, neck, and knee pain.28-32 A 2020 Agency for Healthcare Research and Quality review determined there is some evidence that massage therapy is helpful for chronic low back and neck pain and fibromyalgia.33 Research also has demonstrated that massage reduces anxiety and depression in several different population types.13,34,35 Li and colleagues showed that foot massage increased oxytocin levels in healthy males.36 Although further research is needed to determine the mechanisms of massage therapy, there are important physiologic effects. Unlike most medications, massage therapy is unique in that it can impact health and well-being through multiple mechanisms; for example, by reducing pain, improving mood, providing a sense of social connection and/or improving mobility.
Patients using CIH therapies report greater awareness of the need for ongoing engagement in their own care and health behavior changes.37,38
Driscoll and colleagues reported that women veterans are interested in conservative treatment for their chronic musculoskeletal pain and are open to using CIH therapies.39 Research suggests that veterans are interested in and, in some cases, already using massage therapy.23,40-43 Access to massage therapy and other CIH therapies offers patients choice and control over the types and timing of therapy they receive, exemplified by the 80% of patients in our study who previously received a massage and sought another before a potentially stressful situation.
Access to massage therapy or other CIH therapies may reduce the need for more expensive procedures. Although research on the cost-effectiveness of massage therapy is limited, Herman and colleagues did an economic evaluation of CIH therapies in a veteran population, finding that CIH users had lower annual health care costs and lower pain in the year after CIH started. Sensitivity analyses indicated similar results for acupuncture, chiropractic care, and massage but higher costs for those with 8 or more visits.44
The prevalence of comorbid mental health conditions with MSD suggests that female veterans may benefit from multidisciplinary treatment of pain and depression.3,26 Women-centered programs would be both encouraging and validating to women.39 Massage therapy can be combined with physical therapy, yoga, tai chi, and meditation programs to improve pain, anxiety, strength, and flexibility and can be incorporated into a multimodal treatment plan. Likewise, other subpopulations of female veterans with chronic pain, mental health conditions, or cancer could be targeted with multidisciplinary programs that include massage therapy.
Limitations
This study has several limitations including lack of a control group, a self-selected population, the lack of objective biochemical measurements, and possible respondent bias to please the MTs. Eighty percent had previously experienced massage therapy and may have been biased toward the effects of massage before receiving the intervention. The first report of the effects of massage therapy in an exclusively female veteran population is a major strength of this study.
Further research including randomized controlled trials is needed, especially in populations with coexisting chronic pain and mental health disorders, as is exploring the acceptability of massage therapy for female veterans with MST. Finding viable alternatives to medications has become even more important as the nation addresses the challenge of the opioid crisis.45,46
Conclusions
Female veterans are increasingly seeking VA health care.
Acknowledgments
The authors express our gratitude to the Women Veteran Program Manager, Cheryl Allen, RN; Massage Therapists Denise McGee and Kimberly Morro; Dara Ganoczy, MPH, for help with statistical analysis; and Mark Hausman, MD, for leadership support.
1. US Department of Veteran Affairs, National Center for Veterans Analysis and Statistics. Veteran population. Updated April 14, 2021. Accessed January 6, 2022. https://www.va.gov/vetdata/veteran_population.asp
2. US Department of Veteran Affairs. Women veterans report: the past, present, and future of women veterans. Published February 2017. Accessed January 6, 2022. https://www.va.gov/vetdata/docs/specialreports/women_veterans_2015_final.pdf
3. Higgins DM, Fenton BT, Driscoll MA, et al. Gender differences in demographic and clinical correlates among veterans with musculoskeletal disorders. Womens Health Issues. 2017;27(4):463-470. doi:10.1016/j.whi.2017.01.008
4. Lehavot K, Goldberg SB, Chen JA, et al. Do trauma type, stressful life events, and social support explain women veterans’ high prevalence of PTSD?. Soc Psychiatry Psychiatr Epidemiol. 2018;53(9):943-953. doi:10.1007/s00127-018-1550-x
5. Levander XA, Overland MK. Care of women veterans. Med Clin North Am. 2015;99(3):651-662. doi:10.1016/j.mcna.2015.01.013
6. US Department of Veteran Affairs. Facts and statistics about women veterans. Updated May 28. 2020. Accessed January 6, 2022. https://www.womenshealth.va.gov/womenshealth/latestinformation/facts.asp
7. Krejci LP, Carter K, Gaudet T. Whole health: the vision and implementation of personalized, proactive, patient-driven health care for veterans. Med Care. 2014;52(12)(suppl 5):S5-S8. doi:10.1097/MLR.0000000000000226
8. Elwy AR, Taylor SL, Zhao S, et al. Participating in complementary and integrative health approaches is associated with veterans’ patient-reported outcomes over time. Med Care. 2020;58:S125-S132. doi:10.1097/MLR.0000000000001357
9. Smeeding SJ, Bradshaw DH, Kumpfer K, Trevithick S, Stoddard GJ. Outcome evaluation of the Veterans Affairs Salt Lake City Integrative Health Clinic for chronic pain and stress-related depression, anxiety, and post-traumatic stress disorder. J Altern Complement Med. 2010;16(8):823-835. doi:10.1089/acm.2009.0510
10. Hull A, Brooks Holliday S, Eickhoff C, et al. Veteran participation in the integrative health and wellness program: impact on self-reported mental and physical health outcomes. Psychol Serv. 2019;16(3):475-483. doi:10.1037/ser0000192
11. Zephyrin LC. Reproductive health management for the care of women veterans [published correction appears in Obstet Gynecol. 2016 Mar;127(3):605]. Obstet Gynecol. 2016;127(2):383-392. doi:10.1097/AOG.0000000000001252
12. Piotrowski MM, Paterson C, Mitchinson A, Kim HM, Kirsh M, Hinshaw DB. Massage as adjuvant therapy in the management of acute postoperative pain: a preliminary study in men. J Am Coll Surg. 2003;197(6):1037-1046. doi:10.1016/j.jamcollsurg.2003.07.020
13. Mitchinson AR, Kim HM, Rosenberg JM, et al. Acute postoperative pain management using massage as an adjuvant therapy: a randomized trial. Arch Surg. 2007;142(12):1158-1167. doi:10.1001/archsurg.142.12.1158
14. Mitchinson A, Fletcher CE, Kim HM, Montagnini M, Hinshaw DB. Integrating massage therapy within the palliative care of veterans with advanced illnesses: an outcome study. Am J Hosp Palliat Care. 2014;31(1):6-12. doi:10.1177/1049909113476568
15. Fletcher CE, Mitchinson AR, Trumble EL, Hinshaw DB, Dusek JA. Perceptions of other integrative health therapies by veterans with pain who are receiving massage. J Rehabil Res Dev. 2016;53(1):117-126. doi:10.1682/JRRD.2015.01.0015
16. Juberg M, Jerger KK, Allen KD, Dmitrieva NO, Keever T, Perlman AI. Pilot study of massage in veterans with knee osteoarthritis. J Altern Complement Med. 2015;21(6):333-338. doi:10.1089/acm.2014.0254
17. Beck I, Runeson I, Blomqvist K. To find inner peace: soft massage as an established and integrated part of palliative care. Int J Palliate Nurse. 2009;15(11):541-545. doi: 10.12968/ijpn.2009.15.11.45493
18. Haskell SG, Ning Y, Krebs E, et al. Prevalence of painful musculoskeletal conditions in female and male veterans in 7 years after return from deployment in Operation Enduring Freedom/Operation Iraqi Freedom. Clin J Pain. 2012;28(2):163-167. doi:10.1097/AJP.0b013e318223d951
19. Maguen S, Ren L, Bosch JO, Marmar CR, Seal KH. Gender differences in mental health diagnoses among Iraq and Afghanistan veterans enrolled in veterans affairs health care. Am J Public Health. 2010;100(12):2450-2456. doi:10.2105/AJPH.2009.166165
20. Outcalt SD, Kroenke K, Krebs EE, et al. Chronic pain and comorbid mental health conditions: independent associations of posttraumatic stress disorder and depression with pain, disability, and quality of life. J Behav Med. 2015;38(3):535-543. doi:10.1007/s10865-015-9628-3
21. Gibson CJ, Maguen S, Xia F, Barnes DE, Peltz CB, Yaffe K. Military sexual trauma in older women veterans: prevalence and comorbidities. J Gen Intern Med. 2020;35(1):207-213. doi:10.1007/s11606-019-05342-7
22. Tan G, Teo I, Srivastava D, et al. Improving access to care for women veterans suffering from chronic pain and depression associated with trauma. Pain Med. 2013;14(7):1010-1020. doi:10.1111/pme.12131
23. Haskell SG, Heapy A, Reid MC, Papas RK, Kerns RD. The prevalence and age-related characteristics of pain in a sample of women veterans receiving primary care. J Womens Health (Larchmt). 2006;15(7):862-869. doi:10.1089/jwh.2006.15.862
24. Driscoll MA, Higgins D, Shamaskin-Garroway A, et al. Examining gender as a correlate of self-reported pain treatment use among recent service veterans with deployment-related musculoskeletal disorders. Pain Med. 2017;18(9):1767-1777. doi:10.1093/pm/pnx023
25. Weimer MB, Macey TA, Nicolaidis C, Dobscha SK, Duckart JP, Morasco BJ. Sex differences in the medical care of VA patients with chronic non-cancer pain. Pain Med. 2013;14(12):1839-1847. doi:10.1111/pme.12177
26. Stubbs D, Krebs E, Bair M, et al. Sex differences in pain and pain-related disability among primary care patients with chronic musculoskeletal pain. Pain Med. 2010;11(2):232-239. doi:10.1111/j.1526-4637.2009.00760.x
27. Keogh E, McCracken LM, Eccleston C. Gender moderates the association between depression and disability in chronic pain patients. Eur J Pain. 2006;10(5):413-422. doi:10.1016/j.ejpain.2005.05.007
28. Miake-Lye IM, Mak S, Lee J, et al. Massage for pain: an evidence map. J Altern Complement Med. 2019;25(5):475-502. doi:10.1089/acm.2018.0282
29. Cherkin DC, Sherman KJ, Kahn J, et al. A comparison of the effects of 2 types of massage and usual care on chronic low back pain: a randomized, controlled trial. Ann Intern Med. 2011;155(1):1-9. doi:10.7326/0003-4819-155-1-201107050-00002
30. Sherman KJ, Cook AJ, Wellman RD, et al. Five-week outcomes from a dosing trial of therapeutic massage for chronic neck pain. Ann Fam Med. 2014;12(2):112-120. doi:10.1370/afm.1602
31. Perlman AI, Sabina A, Williams AL, Njike VY, Katz DL. Massage therapy for osteoarthritis of the knee: a randomized controlled trial. Arch Intern Med. 2006;166(22):2533-2538. doi:10.1001/archinte.166.22.2533
32. Perlman A, Fogerite SG, Glass O, et al. Efficacy and safety of massage for osteoarthritis of the knee: a randomized clinical trial. J Gen Intern Med. 2019;34(3):379-386. doi:10.1007/s11606-018-4763-5
33. Skelly AC, Chou R, Dettori JR, et al. Noninvasive Nonpharmacological Treatment for Chronic Pain: A Systematic Review Update. Comparative Effectiveness Review. No. 227. Agency for Healthcare Research and Quality; 2020. doi:10.23970/AHRQEPCCER227
34. Moyer CA, Rounds J, Hannum JW. A meta-analysis of massage therapy research. Psychol Bull. 2004;130(1):3-18. doi:10.1037/0033-2909.130.1.3
35. Field T, Hernandez-Reif M, Diego M, Schanberg S, Kuhn C. Cortisol decreases and serotonin and dopamine increase following massage therapy. Int J Neurosci. 2005;115(10):1397-1413. doi:10.1080/ 00207450590956459
36. Li Q, Becker B, Wernicke J, et al. Foot massage evokes oxytocin release and activation of orbitofrontal cortex and superior temporal sulcus. Psychoneuroendocrinology. 2019;101:193-203. doi:10.1016/j.psyneuen.2018.11.016
37. Eaves ER, Sherman KJ, Ritenbaugh C, et al. A qualitative study of changes in expectations over time among patients with chronic low back pain seeking four CAM therapies. BMC Complement Altern Med. 2015;15:12. Published 2015 Feb 5. doi:10.1186/s12906-015-0531-9
38. Bishop FL, Lauche R, Cramer H, et al. Health behavior change and complementary medicine use: National Health Interview Survey 2012. Medicina (Kaunas). 2019;55(10):632. Published 2019 Sep 24. doi:10.3390/medicina55100632
39. Driscoll MA, Knobf MT, Higgins DM, Heapy A, Lee A, Haskell S. Patient experiences navigating chronic pain management in an integrated health care system: a qualitative investigation of women and men. Pain Med. 2018;19(suppl 1):S19-S29. doi:10.1093/pm/pny139
40. Denneson LM, Corson K, Dobscha SK. Complementary and alternative medicine use among veterans with chronic noncancer pain. J Rehabil Res Dev. 2011;48(9):1119-1128. doi:10.1682/jrrd.2010.12.0243
41. Taylor SL, Herman PM, Marshall NJ, et al. Use of complementary and integrated health: a retrospective analysis of U.S. veterans with chronic musculoskeletal pain nationally. J Altern Complement Med. 2019;25(1):32-39. doi:10.1089/acm.2018.0276
42. Evans EA, Herman PM, Washington DL, et al. Gender differences in use of complementary and integrative health by U.S. military veterans with chronic musculoskeletal pain. Womens Health Issues. 2018;28(5):379-386. doi:10.1016/j.whi.2018.07.003
43. Reinhard MJ, Nassif TH, Bloeser K, et al. CAM utilization among OEF/OIF veterans: findings from the National Health Study for a New Generation of US Veterans. Med Care. 2014;52(12)(suppl 5):S45-S49. doi:10.1097/MLR.0000000000000229
44. Herman PM, Yuan AH, Cefalu MS, et al. The use of complementary and integrative health approaches for chronic musculoskeletal pain in younger US Veterans: An economic evaluation. PLoS One. 2019;14(6):e0217831. Published 2019 Jun 5. doi:10.1371/journal.pone.0217831
45. Jonas WB, Schoomaker EB. Pain and opioids in the military: we must do better. JAMA Intern Med. 2014;174(8):1402-1403. doi:10.1001/jamainternmed.2014.2114
46. Han B, Compton WM, Blanco C, Crane E, Lee J, Jones CM. Prescription opioid use, misuse, and use disorders in U.S. adults: 2015 National Survey on Drug Use and Health. Ann Intern Med. 2017;167(5):293-301. doi:10.7326/M17-0865
There are approximately 2 million female veterans in the United States, representing about 10% of the veteran population.1 In 2015, 456,000 female veterans used the US Department of Veterans Affairs (VA) health care services. The VA predicts an increase in utilization over the next 20 years.2
Female veterans are more likely to have musculoskeletal disorder multimorbidity compared with male veterans and have higher rates of depressive and bipolar disorders, anxiety, and posttraumatic stress disorder (PTSD).3,4 Compared with male veterans, female veterans are younger, more likely to be unmarried and to have served during the wars in Iraq and Afghanistan.3 Fifty-five percent of women veterans vs 41% of men veterans have a service-connected disability, and a greater percentage of women veterans have a service connection rating > 50%.5 The top service-connected disabilities for women veterans are PTSD, major depressive disorder, migraines, and lumbosacral or cervical strain.2 In addition, one-third of women veterans using VA health care report experiencing military sexual trauma (MST).6 Military service may impact the health of female veterans both physically and mentally. Providing treatments and programs to improve their health and their health care experience are current VA priorities.
The VA is changing the way health care is conceptualized and delivered by implementing a holistic model of care known as Whole Health, which seeks to empower and equip patients to take charge of their health, blending conventional medicine with self-care and complementary and integrative health (CIH) approaches, such as massage therapy, yoga, acupuncture, and meditation.7 CIH therapies can help improve physical and mental health with little to no adverse effects.8-10
As part of the Whole Health initiative at the VA Ann Arbor Healthcare System (VAAAHS) in Michigan, the massage program was expanded in 2017 to offer relaxation massages to female veterans attending the women’s health clinic, which provides gynecologic care. Patients visiting a gynecology clinic often experience anxiety and pain related to invasive procedures and examinations. This is especially true for female veterans who experienced MST.11
VAAAHS has 1 staff massage therapist (MT). To expand the program to the women’s health clinic, volunteer licensed MTs were recruited and trained in specific procedures by the staff MT.
Several studies have demonstrated the effect of therapeutic massage on pain and anxiety in predominantly male veteran study populations, including veterans needing postsurgical and palliative care as well as those experiencing chronic pain and knee osteoarthritis.12-16 Little is known about the effects of massage therapy on female veterans. The purpose of this pilot study was to examine the effects of massage therapy among female veterans participating in the women’s health massage program.
Methods
The setting for this pre-post intervention study was VAAAHS. Veterans were called in advance by clinic staff and scheduled for 60-minute appointments either before or after their clinic appointment, depending on availability. MTs were instructed to provide relaxation massage using Swedish massage techniques with moderate pressure, avoiding deep pressure techniques.
The volunteer MTs gave the participants a survey to provide comments and to rate baseline pain and other symptoms prior to and following the massage. The MT left the room to provide privacy while completing the survey. The staff included the symptom data in the massage note as clinical outcomes and entered them into the electronic health record. Massages were given from October 1, 2017 to June 30, 2018. Data including symptom scores, demographics, the presence of chronic pain, mental health diagnoses, patient comments, and opioid use were abstracted from the electronic health record by 2 members of the study team and entered into an Excel database. This study was approved by the VAAAHS Institutional Review Board.
Study Measures
Pain intensity, pain unpleasantness (the affective component of pain), anxiety, shortness of breath, relaxation, and inner peace were rated pre- and postmassage on a 0 to 10 scale. Shortness of breath was included due to the relationship between breathing and anxiety. Inner peace was assessed to measure the calming effects of massage therapy. Beck and colleagues found the concept of inner peace was an important outcome of massage therapy.17 The scale anchors for pain intensity were “no pain” and “severe pain”; and “not at all unpleasant” and “as unpleasant as it can be” for pain unpleasantness. For anxiety, the anchors were “no anxiety” and “as anxious as I can be.” Anchors for relaxation and inner peace were reversed so that a 0 indicated low relaxation and inner peace while a 10 indicated the highest state of relaxation and inner peace.
Chronic pain was defined as pain existing for > 3 months. A history of chronic pain was determined from a review and synthesis of primary care and specialty care recorded diagnoses, patient concerns, and service-connected disabilities. The diagnoses included lumbosacral or cervical strain, chronic low back, joint (knee, shoulder, hip, ankle), neck, or pelvic pain, fibromyalgia, headache, migraine, osteoarthritis, and myofascial pain syndrome. The presence of mental health conditions, including depression, anxiety, bipolar disorders, and PTSD, were similarly determined by a review of mental health clinical notes. Sex was determined from the gynecology note.
Statistical Analysis
Means and medians were calculated for short-term changes in symptom scores. Due to skewness in the short-term changes, significance was tested using a nonparametric sign test. Significance was adjusted using the Bonferroni correction to protect the overall type I error level at 5% from multiple testing. We also assessed for differences in symptom changes in 4 subgroups, using an unadjusted general linear model: those with (1) chronic pain vs without; (2) an anxiety diagnosis vs without; (3) depression vs without; and (4) a PTSD diagnosis vs without. Data were analyzed using SPSS 25 and SAS 9.4.
Results
Results are based on the first massage received by 96 unique individuals (Table 1). Fifty-one (53%) patients were aged 21 to 40 years, and 45 (47%) were aged ≥ 41 years. Most participants (80%) had had a previous massage. Seven (7%) participants were currently on prescription opioids; 76 (79%) participants had a history of one or more chronic pain diagnoses (eg, back pain, migraine headaches, fibromyalgia) and 78 (81%) had a history of a mental health diagnosis (eg, depression, anxiety, PTSD). Massage sessions ranged from 30 to 60 minutes; most patients received massage therapy for 50 minutes.
Prior to massage, mean scores were 3.9 pain intensity, 3.7 pain unpleasantness, 3.8 anxiety, 1.0 shortness of breath, 4.0 relaxation, and 4.2 inner peace. Short-term changes in symptom scores are shown in Table 2. The mean score for pain intensity decreased by 1.9 points, pain unpleasantness by 2.0 points, anxiety by 2.4 points. The greatest change occurred for relaxation, which increased by 4.3 points. All changes in symptoms were statistically significant (P < .001). For subgroup comparisons
Verbal feedback and written comments about the massage experience were all favorable: No adverse events were reported.
Discussion
Massage therapy may be a useful treatment for female veterans experiencing chronic pain, anxiety disorders, depression, or situational anxiety related to gynecologic procedures. After receiving a relaxation massage, female veterans reported decreased pain intensity, pain unpleasantness, and anxiety while reporting increased relaxation and feelings of inner peace. The effects of massage were consistent for all the symptoms or characteristics assessed, suggesting that massage may act on the body in multiple ways.
These changes parallel those seen in a palliative care population primarily composed of male veterans.14 However, the female veterans in this cohort experienced greater changes in relaxation and feelings of inner peace, which may be partly due to relief of tension related to an upcoming stressful appointment. The large mean decrease in anxiety level among female veterans with PTSD is notable as well as the larger increase in inner peace in those with chronic pain.
Many patients expressed their gratitude for the massage and interest in having access to more massage therapy. Female patients who have experienced sexual trauma or other trauma may especially benefit from massage prior to painful, invasive gynecologic procedures. Anecdotally, 2 nurse chaperones in the clinic mentioned separately to the massage program supervisor that the massages helped some very anxious women better tolerate an invasive procedure that would have been otherwise extremely difficult.
Female veterans are more likely to have musculoskeletal issues after deployment and have higher rates of anxiety, PTSD, and depression compared with those of male veterans.3,4,18,19 Determining relationships between and causes of chronic pain, depression, and PTSD is very challenging but the increased prevalence of chronic pain and comorbid mental health conditions in female veterans may be partially related to MST or other trauma experiences.20-22 Female veterans are most likely to have more than one source of chronic pain.23-25 Female patients with chronic musculoskeletal pain report more pain-related disability.26 Furthermore, greater disability in the context of depression is reported by women with pain compared with those of men.27 Most (78%) female veterans in a primary care population reported chronic pain.23 Similarly, 79% of the female veterans in this study population had chronic pain and 81% had a history of mental health disorders, including depression, anxiety, and PTSD.
Studies have shown that massage therapy improves pain in populations experiencing chronic low back, neck, and knee pain.28-32 A 2020 Agency for Healthcare Research and Quality review determined there is some evidence that massage therapy is helpful for chronic low back and neck pain and fibromyalgia.33 Research also has demonstrated that massage reduces anxiety and depression in several different population types.13,34,35 Li and colleagues showed that foot massage increased oxytocin levels in healthy males.36 Although further research is needed to determine the mechanisms of massage therapy, there are important physiologic effects. Unlike most medications, massage therapy is unique in that it can impact health and well-being through multiple mechanisms; for example, by reducing pain, improving mood, providing a sense of social connection and/or improving mobility.
Patients using CIH therapies report greater awareness of the need for ongoing engagement in their own care and health behavior changes.37,38
Driscoll and colleagues reported that women veterans are interested in conservative treatment for their chronic musculoskeletal pain and are open to using CIH therapies.39 Research suggests that veterans are interested in and, in some cases, already using massage therapy.23,40-43 Access to massage therapy and other CIH therapies offers patients choice and control over the types and timing of therapy they receive, exemplified by the 80% of patients in our study who previously received a massage and sought another before a potentially stressful situation.
Access to massage therapy or other CIH therapies may reduce the need for more expensive procedures. Although research on the cost-effectiveness of massage therapy is limited, Herman and colleagues did an economic evaluation of CIH therapies in a veteran population, finding that CIH users had lower annual health care costs and lower pain in the year after CIH started. Sensitivity analyses indicated similar results for acupuncture, chiropractic care, and massage but higher costs for those with 8 or more visits.44
The prevalence of comorbid mental health conditions with MSD suggests that female veterans may benefit from multidisciplinary treatment of pain and depression.3,26 Women-centered programs would be both encouraging and validating to women.39 Massage therapy can be combined with physical therapy, yoga, tai chi, and meditation programs to improve pain, anxiety, strength, and flexibility and can be incorporated into a multimodal treatment plan. Likewise, other subpopulations of female veterans with chronic pain, mental health conditions, or cancer could be targeted with multidisciplinary programs that include massage therapy.
Limitations
This study has several limitations including lack of a control group, a self-selected population, the lack of objective biochemical measurements, and possible respondent bias to please the MTs. Eighty percent had previously experienced massage therapy and may have been biased toward the effects of massage before receiving the intervention. The first report of the effects of massage therapy in an exclusively female veteran population is a major strength of this study.
Further research including randomized controlled trials is needed, especially in populations with coexisting chronic pain and mental health disorders, as is exploring the acceptability of massage therapy for female veterans with MST. Finding viable alternatives to medications has become even more important as the nation addresses the challenge of the opioid crisis.45,46
Conclusions
Female veterans are increasingly seeking VA health care.
Acknowledgments
The authors express our gratitude to the Women Veteran Program Manager, Cheryl Allen, RN; Massage Therapists Denise McGee and Kimberly Morro; Dara Ganoczy, MPH, for help with statistical analysis; and Mark Hausman, MD, for leadership support.
There are approximately 2 million female veterans in the United States, representing about 10% of the veteran population.1 In 2015, 456,000 female veterans used the US Department of Veterans Affairs (VA) health care services. The VA predicts an increase in utilization over the next 20 years.2
Female veterans are more likely to have musculoskeletal disorder multimorbidity compared with male veterans and have higher rates of depressive and bipolar disorders, anxiety, and posttraumatic stress disorder (PTSD).3,4 Compared with male veterans, female veterans are younger, more likely to be unmarried and to have served during the wars in Iraq and Afghanistan.3 Fifty-five percent of women veterans vs 41% of men veterans have a service-connected disability, and a greater percentage of women veterans have a service connection rating > 50%.5 The top service-connected disabilities for women veterans are PTSD, major depressive disorder, migraines, and lumbosacral or cervical strain.2 In addition, one-third of women veterans using VA health care report experiencing military sexual trauma (MST).6 Military service may impact the health of female veterans both physically and mentally. Providing treatments and programs to improve their health and their health care experience are current VA priorities.
The VA is changing the way health care is conceptualized and delivered by implementing a holistic model of care known as Whole Health, which seeks to empower and equip patients to take charge of their health, blending conventional medicine with self-care and complementary and integrative health (CIH) approaches, such as massage therapy, yoga, acupuncture, and meditation.7 CIH therapies can help improve physical and mental health with little to no adverse effects.8-10
As part of the Whole Health initiative at the VA Ann Arbor Healthcare System (VAAAHS) in Michigan, the massage program was expanded in 2017 to offer relaxation massages to female veterans attending the women’s health clinic, which provides gynecologic care. Patients visiting a gynecology clinic often experience anxiety and pain related to invasive procedures and examinations. This is especially true for female veterans who experienced MST.11
VAAAHS has 1 staff massage therapist (MT). To expand the program to the women’s health clinic, volunteer licensed MTs were recruited and trained in specific procedures by the staff MT.
Several studies have demonstrated the effect of therapeutic massage on pain and anxiety in predominantly male veteran study populations, including veterans needing postsurgical and palliative care as well as those experiencing chronic pain and knee osteoarthritis.12-16 Little is known about the effects of massage therapy on female veterans. The purpose of this pilot study was to examine the effects of massage therapy among female veterans participating in the women’s health massage program.
Methods
The setting for this pre-post intervention study was VAAAHS. Veterans were called in advance by clinic staff and scheduled for 60-minute appointments either before or after their clinic appointment, depending on availability. MTs were instructed to provide relaxation massage using Swedish massage techniques with moderate pressure, avoiding deep pressure techniques.
The volunteer MTs gave the participants a survey to provide comments and to rate baseline pain and other symptoms prior to and following the massage. The MT left the room to provide privacy while completing the survey. The staff included the symptom data in the massage note as clinical outcomes and entered them into the electronic health record. Massages were given from October 1, 2017 to June 30, 2018. Data including symptom scores, demographics, the presence of chronic pain, mental health diagnoses, patient comments, and opioid use were abstracted from the electronic health record by 2 members of the study team and entered into an Excel database. This study was approved by the VAAAHS Institutional Review Board.
Study Measures
Pain intensity, pain unpleasantness (the affective component of pain), anxiety, shortness of breath, relaxation, and inner peace were rated pre- and postmassage on a 0 to 10 scale. Shortness of breath was included due to the relationship between breathing and anxiety. Inner peace was assessed to measure the calming effects of massage therapy. Beck and colleagues found the concept of inner peace was an important outcome of massage therapy.17 The scale anchors for pain intensity were “no pain” and “severe pain”; and “not at all unpleasant” and “as unpleasant as it can be” for pain unpleasantness. For anxiety, the anchors were “no anxiety” and “as anxious as I can be.” Anchors for relaxation and inner peace were reversed so that a 0 indicated low relaxation and inner peace while a 10 indicated the highest state of relaxation and inner peace.
Chronic pain was defined as pain existing for > 3 months. A history of chronic pain was determined from a review and synthesis of primary care and specialty care recorded diagnoses, patient concerns, and service-connected disabilities. The diagnoses included lumbosacral or cervical strain, chronic low back, joint (knee, shoulder, hip, ankle), neck, or pelvic pain, fibromyalgia, headache, migraine, osteoarthritis, and myofascial pain syndrome. The presence of mental health conditions, including depression, anxiety, bipolar disorders, and PTSD, were similarly determined by a review of mental health clinical notes. Sex was determined from the gynecology note.
Statistical Analysis
Means and medians were calculated for short-term changes in symptom scores. Due to skewness in the short-term changes, significance was tested using a nonparametric sign test. Significance was adjusted using the Bonferroni correction to protect the overall type I error level at 5% from multiple testing. We also assessed for differences in symptom changes in 4 subgroups, using an unadjusted general linear model: those with (1) chronic pain vs without; (2) an anxiety diagnosis vs without; (3) depression vs without; and (4) a PTSD diagnosis vs without. Data were analyzed using SPSS 25 and SAS 9.4.
Results
Results are based on the first massage received by 96 unique individuals (Table 1). Fifty-one (53%) patients were aged 21 to 40 years, and 45 (47%) were aged ≥ 41 years. Most participants (80%) had had a previous massage. Seven (7%) participants were currently on prescription opioids; 76 (79%) participants had a history of one or more chronic pain diagnoses (eg, back pain, migraine headaches, fibromyalgia) and 78 (81%) had a history of a mental health diagnosis (eg, depression, anxiety, PTSD). Massage sessions ranged from 30 to 60 minutes; most patients received massage therapy for 50 minutes.
Prior to massage, mean scores were 3.9 pain intensity, 3.7 pain unpleasantness, 3.8 anxiety, 1.0 shortness of breath, 4.0 relaxation, and 4.2 inner peace. Short-term changes in symptom scores are shown in Table 2. The mean score for pain intensity decreased by 1.9 points, pain unpleasantness by 2.0 points, anxiety by 2.4 points. The greatest change occurred for relaxation, which increased by 4.3 points. All changes in symptoms were statistically significant (P < .001). For subgroup comparisons
Verbal feedback and written comments about the massage experience were all favorable: No adverse events were reported.
Discussion
Massage therapy may be a useful treatment for female veterans experiencing chronic pain, anxiety disorders, depression, or situational anxiety related to gynecologic procedures. After receiving a relaxation massage, female veterans reported decreased pain intensity, pain unpleasantness, and anxiety while reporting increased relaxation and feelings of inner peace. The effects of massage were consistent for all the symptoms or characteristics assessed, suggesting that massage may act on the body in multiple ways.
These changes parallel those seen in a palliative care population primarily composed of male veterans.14 However, the female veterans in this cohort experienced greater changes in relaxation and feelings of inner peace, which may be partly due to relief of tension related to an upcoming stressful appointment. The large mean decrease in anxiety level among female veterans with PTSD is notable as well as the larger increase in inner peace in those with chronic pain.
Many patients expressed their gratitude for the massage and interest in having access to more massage therapy. Female patients who have experienced sexual trauma or other trauma may especially benefit from massage prior to painful, invasive gynecologic procedures. Anecdotally, 2 nurse chaperones in the clinic mentioned separately to the massage program supervisor that the massages helped some very anxious women better tolerate an invasive procedure that would have been otherwise extremely difficult.
Female veterans are more likely to have musculoskeletal issues after deployment and have higher rates of anxiety, PTSD, and depression compared with those of male veterans.3,4,18,19 Determining relationships between and causes of chronic pain, depression, and PTSD is very challenging but the increased prevalence of chronic pain and comorbid mental health conditions in female veterans may be partially related to MST or other trauma experiences.20-22 Female veterans are most likely to have more than one source of chronic pain.23-25 Female patients with chronic musculoskeletal pain report more pain-related disability.26 Furthermore, greater disability in the context of depression is reported by women with pain compared with those of men.27 Most (78%) female veterans in a primary care population reported chronic pain.23 Similarly, 79% of the female veterans in this study population had chronic pain and 81% had a history of mental health disorders, including depression, anxiety, and PTSD.
Studies have shown that massage therapy improves pain in populations experiencing chronic low back, neck, and knee pain.28-32 A 2020 Agency for Healthcare Research and Quality review determined there is some evidence that massage therapy is helpful for chronic low back and neck pain and fibromyalgia.33 Research also has demonstrated that massage reduces anxiety and depression in several different population types.13,34,35 Li and colleagues showed that foot massage increased oxytocin levels in healthy males.36 Although further research is needed to determine the mechanisms of massage therapy, there are important physiologic effects. Unlike most medications, massage therapy is unique in that it can impact health and well-being through multiple mechanisms; for example, by reducing pain, improving mood, providing a sense of social connection and/or improving mobility.
Patients using CIH therapies report greater awareness of the need for ongoing engagement in their own care and health behavior changes.37,38
Driscoll and colleagues reported that women veterans are interested in conservative treatment for their chronic musculoskeletal pain and are open to using CIH therapies.39 Research suggests that veterans are interested in and, in some cases, already using massage therapy.23,40-43 Access to massage therapy and other CIH therapies offers patients choice and control over the types and timing of therapy they receive, exemplified by the 80% of patients in our study who previously received a massage and sought another before a potentially stressful situation.
Access to massage therapy or other CIH therapies may reduce the need for more expensive procedures. Although research on the cost-effectiveness of massage therapy is limited, Herman and colleagues did an economic evaluation of CIH therapies in a veteran population, finding that CIH users had lower annual health care costs and lower pain in the year after CIH started. Sensitivity analyses indicated similar results for acupuncture, chiropractic care, and massage but higher costs for those with 8 or more visits.44
The prevalence of comorbid mental health conditions with MSD suggests that female veterans may benefit from multidisciplinary treatment of pain and depression.3,26 Women-centered programs would be both encouraging and validating to women.39 Massage therapy can be combined with physical therapy, yoga, tai chi, and meditation programs to improve pain, anxiety, strength, and flexibility and can be incorporated into a multimodal treatment plan. Likewise, other subpopulations of female veterans with chronic pain, mental health conditions, or cancer could be targeted with multidisciplinary programs that include massage therapy.
Limitations
This study has several limitations including lack of a control group, a self-selected population, the lack of objective biochemical measurements, and possible respondent bias to please the MTs. Eighty percent had previously experienced massage therapy and may have been biased toward the effects of massage before receiving the intervention. The first report of the effects of massage therapy in an exclusively female veteran population is a major strength of this study.
Further research including randomized controlled trials is needed, especially in populations with coexisting chronic pain and mental health disorders, as is exploring the acceptability of massage therapy for female veterans with MST. Finding viable alternatives to medications has become even more important as the nation addresses the challenge of the opioid crisis.45,46
Conclusions
Female veterans are increasingly seeking VA health care.
Acknowledgments
The authors express our gratitude to the Women Veteran Program Manager, Cheryl Allen, RN; Massage Therapists Denise McGee and Kimberly Morro; Dara Ganoczy, MPH, for help with statistical analysis; and Mark Hausman, MD, for leadership support.
1. US Department of Veteran Affairs, National Center for Veterans Analysis and Statistics. Veteran population. Updated April 14, 2021. Accessed January 6, 2022. https://www.va.gov/vetdata/veteran_population.asp
2. US Department of Veteran Affairs. Women veterans report: the past, present, and future of women veterans. Published February 2017. Accessed January 6, 2022. https://www.va.gov/vetdata/docs/specialreports/women_veterans_2015_final.pdf
3. Higgins DM, Fenton BT, Driscoll MA, et al. Gender differences in demographic and clinical correlates among veterans with musculoskeletal disorders. Womens Health Issues. 2017;27(4):463-470. doi:10.1016/j.whi.2017.01.008
4. Lehavot K, Goldberg SB, Chen JA, et al. Do trauma type, stressful life events, and social support explain women veterans’ high prevalence of PTSD?. Soc Psychiatry Psychiatr Epidemiol. 2018;53(9):943-953. doi:10.1007/s00127-018-1550-x
5. Levander XA, Overland MK. Care of women veterans. Med Clin North Am. 2015;99(3):651-662. doi:10.1016/j.mcna.2015.01.013
6. US Department of Veteran Affairs. Facts and statistics about women veterans. Updated May 28. 2020. Accessed January 6, 2022. https://www.womenshealth.va.gov/womenshealth/latestinformation/facts.asp
7. Krejci LP, Carter K, Gaudet T. Whole health: the vision and implementation of personalized, proactive, patient-driven health care for veterans. Med Care. 2014;52(12)(suppl 5):S5-S8. doi:10.1097/MLR.0000000000000226
8. Elwy AR, Taylor SL, Zhao S, et al. Participating in complementary and integrative health approaches is associated with veterans’ patient-reported outcomes over time. Med Care. 2020;58:S125-S132. doi:10.1097/MLR.0000000000001357
9. Smeeding SJ, Bradshaw DH, Kumpfer K, Trevithick S, Stoddard GJ. Outcome evaluation of the Veterans Affairs Salt Lake City Integrative Health Clinic for chronic pain and stress-related depression, anxiety, and post-traumatic stress disorder. J Altern Complement Med. 2010;16(8):823-835. doi:10.1089/acm.2009.0510
10. Hull A, Brooks Holliday S, Eickhoff C, et al. Veteran participation in the integrative health and wellness program: impact on self-reported mental and physical health outcomes. Psychol Serv. 2019;16(3):475-483. doi:10.1037/ser0000192
11. Zephyrin LC. Reproductive health management for the care of women veterans [published correction appears in Obstet Gynecol. 2016 Mar;127(3):605]. Obstet Gynecol. 2016;127(2):383-392. doi:10.1097/AOG.0000000000001252
12. Piotrowski MM, Paterson C, Mitchinson A, Kim HM, Kirsh M, Hinshaw DB. Massage as adjuvant therapy in the management of acute postoperative pain: a preliminary study in men. J Am Coll Surg. 2003;197(6):1037-1046. doi:10.1016/j.jamcollsurg.2003.07.020
13. Mitchinson AR, Kim HM, Rosenberg JM, et al. Acute postoperative pain management using massage as an adjuvant therapy: a randomized trial. Arch Surg. 2007;142(12):1158-1167. doi:10.1001/archsurg.142.12.1158
14. Mitchinson A, Fletcher CE, Kim HM, Montagnini M, Hinshaw DB. Integrating massage therapy within the palliative care of veterans with advanced illnesses: an outcome study. Am J Hosp Palliat Care. 2014;31(1):6-12. doi:10.1177/1049909113476568
15. Fletcher CE, Mitchinson AR, Trumble EL, Hinshaw DB, Dusek JA. Perceptions of other integrative health therapies by veterans with pain who are receiving massage. J Rehabil Res Dev. 2016;53(1):117-126. doi:10.1682/JRRD.2015.01.0015
16. Juberg M, Jerger KK, Allen KD, Dmitrieva NO, Keever T, Perlman AI. Pilot study of massage in veterans with knee osteoarthritis. J Altern Complement Med. 2015;21(6):333-338. doi:10.1089/acm.2014.0254
17. Beck I, Runeson I, Blomqvist K. To find inner peace: soft massage as an established and integrated part of palliative care. Int J Palliate Nurse. 2009;15(11):541-545. doi: 10.12968/ijpn.2009.15.11.45493
18. Haskell SG, Ning Y, Krebs E, et al. Prevalence of painful musculoskeletal conditions in female and male veterans in 7 years after return from deployment in Operation Enduring Freedom/Operation Iraqi Freedom. Clin J Pain. 2012;28(2):163-167. doi:10.1097/AJP.0b013e318223d951
19. Maguen S, Ren L, Bosch JO, Marmar CR, Seal KH. Gender differences in mental health diagnoses among Iraq and Afghanistan veterans enrolled in veterans affairs health care. Am J Public Health. 2010;100(12):2450-2456. doi:10.2105/AJPH.2009.166165
20. Outcalt SD, Kroenke K, Krebs EE, et al. Chronic pain and comorbid mental health conditions: independent associations of posttraumatic stress disorder and depression with pain, disability, and quality of life. J Behav Med. 2015;38(3):535-543. doi:10.1007/s10865-015-9628-3
21. Gibson CJ, Maguen S, Xia F, Barnes DE, Peltz CB, Yaffe K. Military sexual trauma in older women veterans: prevalence and comorbidities. J Gen Intern Med. 2020;35(1):207-213. doi:10.1007/s11606-019-05342-7
22. Tan G, Teo I, Srivastava D, et al. Improving access to care for women veterans suffering from chronic pain and depression associated with trauma. Pain Med. 2013;14(7):1010-1020. doi:10.1111/pme.12131
23. Haskell SG, Heapy A, Reid MC, Papas RK, Kerns RD. The prevalence and age-related characteristics of pain in a sample of women veterans receiving primary care. J Womens Health (Larchmt). 2006;15(7):862-869. doi:10.1089/jwh.2006.15.862
24. Driscoll MA, Higgins D, Shamaskin-Garroway A, et al. Examining gender as a correlate of self-reported pain treatment use among recent service veterans with deployment-related musculoskeletal disorders. Pain Med. 2017;18(9):1767-1777. doi:10.1093/pm/pnx023
25. Weimer MB, Macey TA, Nicolaidis C, Dobscha SK, Duckart JP, Morasco BJ. Sex differences in the medical care of VA patients with chronic non-cancer pain. Pain Med. 2013;14(12):1839-1847. doi:10.1111/pme.12177
26. Stubbs D, Krebs E, Bair M, et al. Sex differences in pain and pain-related disability among primary care patients with chronic musculoskeletal pain. Pain Med. 2010;11(2):232-239. doi:10.1111/j.1526-4637.2009.00760.x
27. Keogh E, McCracken LM, Eccleston C. Gender moderates the association between depression and disability in chronic pain patients. Eur J Pain. 2006;10(5):413-422. doi:10.1016/j.ejpain.2005.05.007
28. Miake-Lye IM, Mak S, Lee J, et al. Massage for pain: an evidence map. J Altern Complement Med. 2019;25(5):475-502. doi:10.1089/acm.2018.0282
29. Cherkin DC, Sherman KJ, Kahn J, et al. A comparison of the effects of 2 types of massage and usual care on chronic low back pain: a randomized, controlled trial. Ann Intern Med. 2011;155(1):1-9. doi:10.7326/0003-4819-155-1-201107050-00002
30. Sherman KJ, Cook AJ, Wellman RD, et al. Five-week outcomes from a dosing trial of therapeutic massage for chronic neck pain. Ann Fam Med. 2014;12(2):112-120. doi:10.1370/afm.1602
31. Perlman AI, Sabina A, Williams AL, Njike VY, Katz DL. Massage therapy for osteoarthritis of the knee: a randomized controlled trial. Arch Intern Med. 2006;166(22):2533-2538. doi:10.1001/archinte.166.22.2533
32. Perlman A, Fogerite SG, Glass O, et al. Efficacy and safety of massage for osteoarthritis of the knee: a randomized clinical trial. J Gen Intern Med. 2019;34(3):379-386. doi:10.1007/s11606-018-4763-5
33. Skelly AC, Chou R, Dettori JR, et al. Noninvasive Nonpharmacological Treatment for Chronic Pain: A Systematic Review Update. Comparative Effectiveness Review. No. 227. Agency for Healthcare Research and Quality; 2020. doi:10.23970/AHRQEPCCER227
34. Moyer CA, Rounds J, Hannum JW. A meta-analysis of massage therapy research. Psychol Bull. 2004;130(1):3-18. doi:10.1037/0033-2909.130.1.3
35. Field T, Hernandez-Reif M, Diego M, Schanberg S, Kuhn C. Cortisol decreases and serotonin and dopamine increase following massage therapy. Int J Neurosci. 2005;115(10):1397-1413. doi:10.1080/ 00207450590956459
36. Li Q, Becker B, Wernicke J, et al. Foot massage evokes oxytocin release and activation of orbitofrontal cortex and superior temporal sulcus. Psychoneuroendocrinology. 2019;101:193-203. doi:10.1016/j.psyneuen.2018.11.016
37. Eaves ER, Sherman KJ, Ritenbaugh C, et al. A qualitative study of changes in expectations over time among patients with chronic low back pain seeking four CAM therapies. BMC Complement Altern Med. 2015;15:12. Published 2015 Feb 5. doi:10.1186/s12906-015-0531-9
38. Bishop FL, Lauche R, Cramer H, et al. Health behavior change and complementary medicine use: National Health Interview Survey 2012. Medicina (Kaunas). 2019;55(10):632. Published 2019 Sep 24. doi:10.3390/medicina55100632
39. Driscoll MA, Knobf MT, Higgins DM, Heapy A, Lee A, Haskell S. Patient experiences navigating chronic pain management in an integrated health care system: a qualitative investigation of women and men. Pain Med. 2018;19(suppl 1):S19-S29. doi:10.1093/pm/pny139
40. Denneson LM, Corson K, Dobscha SK. Complementary and alternative medicine use among veterans with chronic noncancer pain. J Rehabil Res Dev. 2011;48(9):1119-1128. doi:10.1682/jrrd.2010.12.0243
41. Taylor SL, Herman PM, Marshall NJ, et al. Use of complementary and integrated health: a retrospective analysis of U.S. veterans with chronic musculoskeletal pain nationally. J Altern Complement Med. 2019;25(1):32-39. doi:10.1089/acm.2018.0276
42. Evans EA, Herman PM, Washington DL, et al. Gender differences in use of complementary and integrative health by U.S. military veterans with chronic musculoskeletal pain. Womens Health Issues. 2018;28(5):379-386. doi:10.1016/j.whi.2018.07.003
43. Reinhard MJ, Nassif TH, Bloeser K, et al. CAM utilization among OEF/OIF veterans: findings from the National Health Study for a New Generation of US Veterans. Med Care. 2014;52(12)(suppl 5):S45-S49. doi:10.1097/MLR.0000000000000229
44. Herman PM, Yuan AH, Cefalu MS, et al. The use of complementary and integrative health approaches for chronic musculoskeletal pain in younger US Veterans: An economic evaluation. PLoS One. 2019;14(6):e0217831. Published 2019 Jun 5. doi:10.1371/journal.pone.0217831
45. Jonas WB, Schoomaker EB. Pain and opioids in the military: we must do better. JAMA Intern Med. 2014;174(8):1402-1403. doi:10.1001/jamainternmed.2014.2114
46. Han B, Compton WM, Blanco C, Crane E, Lee J, Jones CM. Prescription opioid use, misuse, and use disorders in U.S. adults: 2015 National Survey on Drug Use and Health. Ann Intern Med. 2017;167(5):293-301. doi:10.7326/M17-0865
1. US Department of Veteran Affairs, National Center for Veterans Analysis and Statistics. Veteran population. Updated April 14, 2021. Accessed January 6, 2022. https://www.va.gov/vetdata/veteran_population.asp
2. US Department of Veteran Affairs. Women veterans report: the past, present, and future of women veterans. Published February 2017. Accessed January 6, 2022. https://www.va.gov/vetdata/docs/specialreports/women_veterans_2015_final.pdf
3. Higgins DM, Fenton BT, Driscoll MA, et al. Gender differences in demographic and clinical correlates among veterans with musculoskeletal disorders. Womens Health Issues. 2017;27(4):463-470. doi:10.1016/j.whi.2017.01.008
4. Lehavot K, Goldberg SB, Chen JA, et al. Do trauma type, stressful life events, and social support explain women veterans’ high prevalence of PTSD?. Soc Psychiatry Psychiatr Epidemiol. 2018;53(9):943-953. doi:10.1007/s00127-018-1550-x
5. Levander XA, Overland MK. Care of women veterans. Med Clin North Am. 2015;99(3):651-662. doi:10.1016/j.mcna.2015.01.013
6. US Department of Veteran Affairs. Facts and statistics about women veterans. Updated May 28. 2020. Accessed January 6, 2022. https://www.womenshealth.va.gov/womenshealth/latestinformation/facts.asp
7. Krejci LP, Carter K, Gaudet T. Whole health: the vision and implementation of personalized, proactive, patient-driven health care for veterans. Med Care. 2014;52(12)(suppl 5):S5-S8. doi:10.1097/MLR.0000000000000226
8. Elwy AR, Taylor SL, Zhao S, et al. Participating in complementary and integrative health approaches is associated with veterans’ patient-reported outcomes over time. Med Care. 2020;58:S125-S132. doi:10.1097/MLR.0000000000001357
9. Smeeding SJ, Bradshaw DH, Kumpfer K, Trevithick S, Stoddard GJ. Outcome evaluation of the Veterans Affairs Salt Lake City Integrative Health Clinic for chronic pain and stress-related depression, anxiety, and post-traumatic stress disorder. J Altern Complement Med. 2010;16(8):823-835. doi:10.1089/acm.2009.0510
10. Hull A, Brooks Holliday S, Eickhoff C, et al. Veteran participation in the integrative health and wellness program: impact on self-reported mental and physical health outcomes. Psychol Serv. 2019;16(3):475-483. doi:10.1037/ser0000192
11. Zephyrin LC. Reproductive health management for the care of women veterans [published correction appears in Obstet Gynecol. 2016 Mar;127(3):605]. Obstet Gynecol. 2016;127(2):383-392. doi:10.1097/AOG.0000000000001252
12. Piotrowski MM, Paterson C, Mitchinson A, Kim HM, Kirsh M, Hinshaw DB. Massage as adjuvant therapy in the management of acute postoperative pain: a preliminary study in men. J Am Coll Surg. 2003;197(6):1037-1046. doi:10.1016/j.jamcollsurg.2003.07.020
13. Mitchinson AR, Kim HM, Rosenberg JM, et al. Acute postoperative pain management using massage as an adjuvant therapy: a randomized trial. Arch Surg. 2007;142(12):1158-1167. doi:10.1001/archsurg.142.12.1158
14. Mitchinson A, Fletcher CE, Kim HM, Montagnini M, Hinshaw DB. Integrating massage therapy within the palliative care of veterans with advanced illnesses: an outcome study. Am J Hosp Palliat Care. 2014;31(1):6-12. doi:10.1177/1049909113476568
15. Fletcher CE, Mitchinson AR, Trumble EL, Hinshaw DB, Dusek JA. Perceptions of other integrative health therapies by veterans with pain who are receiving massage. J Rehabil Res Dev. 2016;53(1):117-126. doi:10.1682/JRRD.2015.01.0015
16. Juberg M, Jerger KK, Allen KD, Dmitrieva NO, Keever T, Perlman AI. Pilot study of massage in veterans with knee osteoarthritis. J Altern Complement Med. 2015;21(6):333-338. doi:10.1089/acm.2014.0254
17. Beck I, Runeson I, Blomqvist K. To find inner peace: soft massage as an established and integrated part of palliative care. Int J Palliate Nurse. 2009;15(11):541-545. doi: 10.12968/ijpn.2009.15.11.45493
18. Haskell SG, Ning Y, Krebs E, et al. Prevalence of painful musculoskeletal conditions in female and male veterans in 7 years after return from deployment in Operation Enduring Freedom/Operation Iraqi Freedom. Clin J Pain. 2012;28(2):163-167. doi:10.1097/AJP.0b013e318223d951
19. Maguen S, Ren L, Bosch JO, Marmar CR, Seal KH. Gender differences in mental health diagnoses among Iraq and Afghanistan veterans enrolled in veterans affairs health care. Am J Public Health. 2010;100(12):2450-2456. doi:10.2105/AJPH.2009.166165
20. Outcalt SD, Kroenke K, Krebs EE, et al. Chronic pain and comorbid mental health conditions: independent associations of posttraumatic stress disorder and depression with pain, disability, and quality of life. J Behav Med. 2015;38(3):535-543. doi:10.1007/s10865-015-9628-3
21. Gibson CJ, Maguen S, Xia F, Barnes DE, Peltz CB, Yaffe K. Military sexual trauma in older women veterans: prevalence and comorbidities. J Gen Intern Med. 2020;35(1):207-213. doi:10.1007/s11606-019-05342-7
22. Tan G, Teo I, Srivastava D, et al. Improving access to care for women veterans suffering from chronic pain and depression associated with trauma. Pain Med. 2013;14(7):1010-1020. doi:10.1111/pme.12131
23. Haskell SG, Heapy A, Reid MC, Papas RK, Kerns RD. The prevalence and age-related characteristics of pain in a sample of women veterans receiving primary care. J Womens Health (Larchmt). 2006;15(7):862-869. doi:10.1089/jwh.2006.15.862
24. Driscoll MA, Higgins D, Shamaskin-Garroway A, et al. Examining gender as a correlate of self-reported pain treatment use among recent service veterans with deployment-related musculoskeletal disorders. Pain Med. 2017;18(9):1767-1777. doi:10.1093/pm/pnx023
25. Weimer MB, Macey TA, Nicolaidis C, Dobscha SK, Duckart JP, Morasco BJ. Sex differences in the medical care of VA patients with chronic non-cancer pain. Pain Med. 2013;14(12):1839-1847. doi:10.1111/pme.12177
26. Stubbs D, Krebs E, Bair M, et al. Sex differences in pain and pain-related disability among primary care patients with chronic musculoskeletal pain. Pain Med. 2010;11(2):232-239. doi:10.1111/j.1526-4637.2009.00760.x
27. Keogh E, McCracken LM, Eccleston C. Gender moderates the association between depression and disability in chronic pain patients. Eur J Pain. 2006;10(5):413-422. doi:10.1016/j.ejpain.2005.05.007
28. Miake-Lye IM, Mak S, Lee J, et al. Massage for pain: an evidence map. J Altern Complement Med. 2019;25(5):475-502. doi:10.1089/acm.2018.0282
29. Cherkin DC, Sherman KJ, Kahn J, et al. A comparison of the effects of 2 types of massage and usual care on chronic low back pain: a randomized, controlled trial. Ann Intern Med. 2011;155(1):1-9. doi:10.7326/0003-4819-155-1-201107050-00002
30. Sherman KJ, Cook AJ, Wellman RD, et al. Five-week outcomes from a dosing trial of therapeutic massage for chronic neck pain. Ann Fam Med. 2014;12(2):112-120. doi:10.1370/afm.1602
31. Perlman AI, Sabina A, Williams AL, Njike VY, Katz DL. Massage therapy for osteoarthritis of the knee: a randomized controlled trial. Arch Intern Med. 2006;166(22):2533-2538. doi:10.1001/archinte.166.22.2533
32. Perlman A, Fogerite SG, Glass O, et al. Efficacy and safety of massage for osteoarthritis of the knee: a randomized clinical trial. J Gen Intern Med. 2019;34(3):379-386. doi:10.1007/s11606-018-4763-5
33. Skelly AC, Chou R, Dettori JR, et al. Noninvasive Nonpharmacological Treatment for Chronic Pain: A Systematic Review Update. Comparative Effectiveness Review. No. 227. Agency for Healthcare Research and Quality; 2020. doi:10.23970/AHRQEPCCER227
34. Moyer CA, Rounds J, Hannum JW. A meta-analysis of massage therapy research. Psychol Bull. 2004;130(1):3-18. doi:10.1037/0033-2909.130.1.3
35. Field T, Hernandez-Reif M, Diego M, Schanberg S, Kuhn C. Cortisol decreases and serotonin and dopamine increase following massage therapy. Int J Neurosci. 2005;115(10):1397-1413. doi:10.1080/ 00207450590956459
36. Li Q, Becker B, Wernicke J, et al. Foot massage evokes oxytocin release and activation of orbitofrontal cortex and superior temporal sulcus. Psychoneuroendocrinology. 2019;101:193-203. doi:10.1016/j.psyneuen.2018.11.016
37. Eaves ER, Sherman KJ, Ritenbaugh C, et al. A qualitative study of changes in expectations over time among patients with chronic low back pain seeking four CAM therapies. BMC Complement Altern Med. 2015;15:12. Published 2015 Feb 5. doi:10.1186/s12906-015-0531-9
38. Bishop FL, Lauche R, Cramer H, et al. Health behavior change and complementary medicine use: National Health Interview Survey 2012. Medicina (Kaunas). 2019;55(10):632. Published 2019 Sep 24. doi:10.3390/medicina55100632
39. Driscoll MA, Knobf MT, Higgins DM, Heapy A, Lee A, Haskell S. Patient experiences navigating chronic pain management in an integrated health care system: a qualitative investigation of women and men. Pain Med. 2018;19(suppl 1):S19-S29. doi:10.1093/pm/pny139
40. Denneson LM, Corson K, Dobscha SK. Complementary and alternative medicine use among veterans with chronic noncancer pain. J Rehabil Res Dev. 2011;48(9):1119-1128. doi:10.1682/jrrd.2010.12.0243
41. Taylor SL, Herman PM, Marshall NJ, et al. Use of complementary and integrated health: a retrospective analysis of U.S. veterans with chronic musculoskeletal pain nationally. J Altern Complement Med. 2019;25(1):32-39. doi:10.1089/acm.2018.0276
42. Evans EA, Herman PM, Washington DL, et al. Gender differences in use of complementary and integrative health by U.S. military veterans with chronic musculoskeletal pain. Womens Health Issues. 2018;28(5):379-386. doi:10.1016/j.whi.2018.07.003
43. Reinhard MJ, Nassif TH, Bloeser K, et al. CAM utilization among OEF/OIF veterans: findings from the National Health Study for a New Generation of US Veterans. Med Care. 2014;52(12)(suppl 5):S45-S49. doi:10.1097/MLR.0000000000000229
44. Herman PM, Yuan AH, Cefalu MS, et al. The use of complementary and integrative health approaches for chronic musculoskeletal pain in younger US Veterans: An economic evaluation. PLoS One. 2019;14(6):e0217831. Published 2019 Jun 5. doi:10.1371/journal.pone.0217831
45. Jonas WB, Schoomaker EB. Pain and opioids in the military: we must do better. JAMA Intern Med. 2014;174(8):1402-1403. doi:10.1001/jamainternmed.2014.2114
46. Han B, Compton WM, Blanco C, Crane E, Lee J, Jones CM. Prescription opioid use, misuse, and use disorders in U.S. adults: 2015 National Survey on Drug Use and Health. Ann Intern Med. 2017;167(5):293-301. doi:10.7326/M17-0865
PTSD Disability Examination Reports: A Comparison of Veterans Health Administration and Contract Examiners
Updated June 17, 2022
The US Department of Veterans Affairs (VA) provides health care for > 9 million military veterans, nearly half of all former service members.1 Over the past 15 years, there has been a steady and substantial increase in the frequency of disability awards for veterans with post-9/11 military service. Recent data from the Bureau of Labor Statistics indicate that 41% of veterans who served after 9/11 receive service-connected disability benefits compared with 28% of veterans overall.2 More than 5 million veterans receive VA service-related disability benefits.2,3 More than half of the VA $243 billion budget for fiscal year (FY) 2021 ($135.5 billion) was allocated to the Veterans Benefits Administration (VBA), of which $115.7 billion (85%) was allocated specifically for service-related compensation claims payments.4
The VA predicted that VBA will have completed 1.4 million ratings for disability claims in 2021.5 A substantial percentage of these claims will be for mental disorders, specifically posttraumatic stress disorder (PTSD). VA officials testifying before Congress in 2017 noted that the number of PTSD claims had nearly tripled in the previous 10 years.6 As far back as 2013, McNally and Frueh analyzed “the skyrocketing of disability claims,” particularly for PTSD, among veterans who served in Iraq and Afghanistan.7
This large increase has placed an unprecedented burden on the VBA to expand its capacity to conduct initial PTSD disability evaluations that by regulations are completed by psychologists or psychiatrists. This need has led the VBA to make significant changes in the compensation and pension (C&P) process, including a reduced role for Veterans Health Administration (VHA) examiners and increased reliance on non-VA (contract) examiners through the Contract Medical Disability Examination (MDE) program. In 2019, the MDE budget was $1.23 billion; in 2020, it was increased to $1.79 billion, and for 2021, it was $2.23 billion, reflecting the increasing investment of resources in non-VA examiners, ostensibly to both increase capacity and save costs.5
Anecdotally, concerns have been raised regarding inadequate training of contract examiners as well as inadequate reports by these examiners. A 2018 Government Accountability Office (GAO) report concluded that VA lacked the data to determine whether contract examiners were meeting standards for quality, timeliness, and accuracy.8 The GAO report noted that VA required 92% of contractor reports contained no obvious errors, a relatively low target; however, in the first half of 2017, only 1 contractor group met that target. The report noted further that “VBA does not verify if examiners have completed training nor does it collect information to assess training effectiveness in preparing examiners.”8 A subsequent analysis of contract examinations completed by the VA Office of the Inspector General (OIG) in 2019 concluded that the MDE program was “hampered in their ability to provide oversight because of limitations with VBA’s electronic examination management systems, the lack of reliable data, and inadequate staffing of the program.”9
These reports have focused almost exclusively on simple performance metrics, such as timeliness of examination completion. However, the 2018 GAO report referenced isolated “focused reviews” of complaints about the quality of examinations by contract examiners and gave as an example an isolated “review of one contracted examiner who had high rates of diagnosing severe posttraumatic stress disorder.”8 After review indicated the examiner’s reports were of poor quality, the VBA discontinued the examiner’s contract.
Unfortunately, despite such anecdotal reports and isolated actions, to date there are no published reports examining and comparing the quality of PTSD examination reports completed by VHA and contract examiners or the subsequent disability determinations made by the VBA as a result of these evaluations. In a November 2020 letter to the VA Secretary, 11 US Senators expressed “grave concerns” regarding the VA decision to privatize C&P programs noting, among other concerns, that there were “no clinical quality measurement for, or evaluation of, contractor examinations.”10 The letter cited anecdotal evidence of contract examiners not reviewing veteran’s medical records and diagnosing conditions “without supporting evidence.”10
The purpose of the present evaluation was to provide a systematic comparison of the content and quality of initial PTSD disability examinations conducted by VHA and non-VA contract examiners. In addition, this study compared the disability rating decisions resulting from VHA and contract examinations.
Methods
A random sample of 100 Initial PTSD Disability Benefits Questionnaires (DBQs)—structured forms completed by all examiners—were obtained from a list supplied by the VA Office of Performance Analysis and Integrity. All examinations were from the Veterans Integrated Service Network (VISN) 1, encompassing the New England region and were conducted in 2019 and 2020. Two of the 100 cases were excluded for technical reasons, resulting in 98 examination reports. However, the final pool yielded 62 contract examinations and only 36 VHA examinations. To make the sample sizes more comparable, an additional 15 examinations were randomly selected from the local examination database (also VISN 1) to complement the original examination pool.
Once DBQs were retrieved, all identifying information was deleted, and cases were analyzed using assigned record numbers. All coding was completed by the 2 principal investigators, both VA psychologists with extensive training and experience in C&P evaluation and treatment of veterans with PTSD. Due to inherent structural differences between the forms used for VA and contract examinations, raters could not be masked/blinded to the source of the report.
A number of measures were taken to reduce bias and enhance objectivity of rating. First, objectively coded variables (eg, age and sex of veteran, period of service, trauma type, diagnoses rendered by the examiner, impairment category endorsed, number and type of symptoms) were transcribed directly from the DBQ as recorded by the examiner. Second, to rate report quality, an initial categorical rating scale was developed based on predetermined elements of examination quality that were considered essential. After refinement and preliminary analysis of interrater reliabilities, 3 quality-related indices were identified: (1) level of detail in description of key content areas (history before service, service trauma, after service social and vocational history, mental health history, substance use); (2) synthesis of history and findings in explaining opinion rendered; and (3) clarity of opinion regarding causation required “at least as likely as not” degree of confidence. The first 2 quality ratings were based on a 3-point scale (poor, fair, good), and the third variable was coded as yes or no. (eAppendix available at doi:10.12788/fp.0225). Interrater reliabilities calculated based on a subsample of 18 cases, randomly selected and rated by both raters, yielded Cohen κ in the acceptable range (.61, .72, and .89 for detail, synthesis, and clarity, respectively). Finally, for information regarding VBA decision making, rating decision documents contained in the Veterans Benefit Management System database were reviewed to determine whether the veteran was granted service connection for PTSD or another mental disorder based on the examination report in question and, if so, the disability rating percentage awarded. These were recorded independently after all other coding had been completed.
Results
Comparison of VHA and contract examinations revealed no significant differences between groups on relevant sociodemographic and other measures (Table). Missing data were not obtained from other records or sources, and for this study, reflect only what is recorded in the examination reports except for age, which was calculated using veteran’s date of birth and the date of examination.
To examine differences between VHA and contract examinations, the groups were first compared on a set of predetermined objectively coded variables taken directly from the DBQ. The frequency of PTSD diagnoses by VHA (57%) and contract (71%) examiners was not significantly different nor were rates of non-PTSD diagnoses by VHA (51%) and contract (73%) examiners. There also was no difference in the mean number of PTSD symptoms endorsed across PTSD diagnostic criteria B, C, D, and E (maximum of 20) recorded by VHA (9.4) and contract (10.9) examiners.
Contract examiners recorded a significantly greater mean number of “other symptoms” on a checklist of 31 possible symptoms as compared to VHA examiners: 7.3 vs 5.8, respectively (t[104] = 2.27, P < .05). An initial analysis of overall social/vocational impairment ratings coded by examiners did not reveal significant differences between examiner groups. However, when the 2 most severe impairment categories were combined to create a pooled “severe” category, 31% of contract examiners rated veterans as severely impaired compared with only 12% of VHA examiners (χ2 = 5.79, 1 df, P < .05) (Figure 1).
VHA and contract examinations were compared on 3 measures of report quality. Significant differences were found for both level of detail (χ2 = 16.44, 2 df, P < .01) and synthesis (χ2 = 6.68, 2 df, P < .05). Contract examinations were more likely to be rated as poor and less likely to be rated good, with a similar proportion of fair ratings for the 2 examination types (Figures 2 and 3). There was no significant difference in the proportion of VHA and contract examinations providing clear statement of opinion regarding causation (ie, whether or not the diagnosed condition was service related), with the majority rendering an adequate opinion in both examiner groups (VHA, 78%; contract, 79%).
Qualitative review revealed examples of markedly deficient examinations among contract examinations, including several reports that contained no review of records, no report of relevant background, and no mention or assessment of social and vocational function needed to inform opinions about diagnosis and impairment.
Finally, the VBA database was used to compare the resulting disability award decisions made by VBA based on the examination reports in question. Examination by contractors resulted in significantly higher mean service-connected disability ratings for examinees compared with VHA examiners (46.8 vs 33.5, respectively; t[108] = 2.3, P < .05).
Discussion
The present study provides the first reported systematic comparison of VA disability examinations for PTSD completed by examiners employed by the VHA and those hired as contract examiners through the MDE program. Although the frequency of PTSD diagnoses by contract examiners was higher than that of VHA examiners (71% vs 57%, respectively), the difference was not statistically significant. However, contract examiners recorded significantly more symptoms for examinees and rated them as severely impaired more frequently than did their VHA counterparts. In keeping with rating guidelines used by the VBA, these differences in examination content resulted in higher disability ratings for veterans seen by contract examiners.
Along with these elevated reports in symptom and severity ratings, contract examiners were less likely to provide adequate detail in the narrative sections of their reports and less frequently provided a satisfactory explanation and synthesis of relevant history and findings in support of their conclusions. Although not reflected in the statistical analysis, case-by-case review revealed some startlingly inadequate examination reports by contract examiners, several of which contained no review of records, no report or discussion of relevant background, and no discussion or analysis of social and vocational function to inform and support their opinion about level of impairment. None of the VHA examination reports reviewed lacked information to that degree.
Such deficiencies in detail and synthesis run counter to accepted guidelines for the adequate assessment of psychological injury in general and in VA disability claims specifically.11,12 For example, Watson and colleagues proposed that a minimum of 3 hours was required to conduct an initial PTSD examination, with more complex cases possibly taking longer.11 There is no information available about how long contract examiners take to complete their examinations and how that compares with the time taken by VA examiners. The VBA failure to monitor whether or not examiners follow accepted guidelines for PTSD examination has not previously been evaluated. Historically, a large number of clinicians, researchers, and policy critics have raised concerns about the potential for exaggeration or malingering among VA PTSD disability claimants and have urged the need to adequately assess for unreliable reporting and presentation.13,14 However, the possibility of systematic examiner deficiency and/or bias increasing the frequency of false or inflated claims being approved has received little empirical attention.
Although contract examiners did not diagnose PTSD significantly more frequently than VHA examiners (71% vs 57%, respectively), the overall frequency of PTSD diagnosis across both groups (65%) was substantially higher than previous figures that have, on average, estimated the lifetime prevalence of PTSD in trauma-exposed veterans to be about 31%.15 A re-analysis of the same National Vietnam Veterans Readjustment Survey data, but applying more conservative diagnostic criteria, reduced the lifetime prevalence to just under 19%, with point prevalence estimates even lower.15,16
In a study of concordance rates between service connection for PTSD and both current and lifetime diagnosis by independent, structured assessment, Marx and colleagues found that a “significant minority” of veterans who were already receiving service-connected disability for PTSD did not meet lifetime and/or current diagnostic criteria.17 Although it is possible that the group of veterans who were applying for disability benefits in our study had a higher rate of PTSD, it also is possible if not likely that the PTSD examination process overall yields inflated rates of diagnosis and levels of impairment. This speaks to the concern raised by Marx and colleagues who found that veterans with service connection for PTSD who received related benefits “may not have the disorder.”17
Limitations
A methodological limitation of the present study was that, due to structural differences in the DBQ forms used for VHA and contract examinations, the reports could not be de-identified as to examiner type and thus raters could not be masked/blinded. To mitigate bias, a predetermined, piloted, and refined coding and rating plan for report quality metrics was adhered to strictly, and interrater reliabilities were acceptable. Future study is suggested in which all report content is standardized for coding using the same format, which at present would require a complete rewriting of the entire report; this problem could be resolved by having the VBA adopt a more coherent system in which all reports, regardless of examiner type, use a single, standardized template. Further study using larger data sets and expanding to other VA regions also is needed.
Conclusions
The present study suggests that poor examination and report quality—by contract examiners and to a lesser degree VHA examiners—are not uncommon. The findings confirm and extend previous anecdotal reports of deficiencies in PTSD examinations performed by contract examiners and provide empirical support for concerns raised of global deficiencies in the VBA oversight of the MDE program. Such deficiencies have significant implications for the quality and integrity of the VA disability determination process for veterans claiming PTSD related to military service.
The current findings support and strengthen the call for development and management of a structured and enforced training and quality assurance/improvement program for VA PTSD disability examinations. Such training and oversight will be critical to improve the quality and integrity of these examinations, reduce error and waste in VBA’s Compensation and Pension process, and in doing so optimize VA financial resources to best serve veterans’ benefits and health care needs.
1. US Department of Veterans Affairs, Veterans Health Adminstration. About VHA. Updated April 23, 2021. Accessed January 6, 2022. www.va.gov/health/aboutvha.asp
2. US Department of Labor, Bureau of Labor Statistics. News release. Employment situation of veterans—2020. Published March 18, 2020. Accessed January 6, 2022. https://www.bls.gov/news.release/pdf/vet.pdf
3. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Department of Veterans Affairs statistics at a glance. Updated December 31, 2020. Accessed January 6, 2022. https://www.va.gov/vetdata/docs/Quickfacts/Stats_at_a_glance_12_31_20.PDF
4. US Department of Veterans Affairs. FY 2021 Budget submission: budget in brief. Published February 2020. Accessed January 6, 2022. https://www.va.gov/budget/docs/summary/archive/FY-2021-VA-BudgetSubmission.zip
5. US Department of Veterans Affairs. FY 2021 budget submission: benefits and burial programs and Departmental Administration volume 3 of 4:178. Published February 2020. Accessed January 6, 2022. https://www.va.gov/budget/docs/summary/archive/FY-2021-VA-BudgetSubmission.zip
6. Statement of Ronald Burke, assistant deputy under secretary, office of field operations Veterans Benefits Administration before the Subcommittee on Disability And Memorial Affairs of the House Committee on Veterans’ Affairs. Published July 25, 2017. Accessed January 6, 2022. https://www.congress.gov/115/meeting/house/106322/witnesses/HHRG-115-VR09-Wstate-BurkeR-20170725.pdf
7. McNally RJ, Frueh BC. Why are Iraq and Afghanistan War veterans seeking PTSD disability compensation at unprecedented rates? J Anxiety Disord. 2013;27(5):520-526. doi:10.1016/j.janxdis.2013.07.002
8. US Government Accountability Office. VA disability exams: improved performance analysis and training oversight needed for contracted exams. GAO-19-13. Published October 2018. Accessed January 6, 2022. https://www.gao.gov/assets/gao-19-13.pdf
9. US Department of Veterans Affairs, Office of Inspector General. Inadequate oversight of contracted disability exam cancellations. Report #18-04266-115. Published June 10, 2019. Accessed January 6, 2022. https://www.va.gov/oig/pubs/VAOIG-18-04266-115.pdf
10. Letter to VA Secretary Wilkie. Published November 11, 2020. Accessed January 6, 2022. https://www.veterans.senate.gov/download/candp-exam-va-letter
11. Watson PW, McFall M, McBrine C, Schnurr PP, Friedman MJ, Keane TM, Hamblen JL (2005). Best practice manual for posttraumatic stress disorder (PTSD) compensation and pension examinations. Portland, OR: Northwest Network Mental Illness Research, Education, and Clinical Center, VA Puget Sound Healthcare System.
12. Worthen MD, Moering RG. A practical guide to conducting VA compensation and pension exams for PTSD and other mental disorders. Psychol Inj and Law. 2011;4:187-216. doi:10.1007/s12207-011-9115-2
13. DeViva JC, Bloem WD. Symptom exaggeration and compensation seeking among combat veterans with posttraumatic stress disorder. J Trauma Stress. 2003;16(5):503-507. doi:10.1023/A:1025766713188
14. Ray CL. Feigning screeners in VA PTSD compensation and pension examinations. Psychol Inj and Law. 2014;7:370-387. doi:10.1007/s12207-014-9210-2
15. Kulka RA, Schlenger WE, Fairbank JA, et al. Trauma and the Vietnam War Generation: Report of Findings From the National Vietnam Veterans Readjustment Study. Brunner Mazel Publishers; 1990.
16. Dohrenwend BP, Turner JB, Turse NA, Adams BG, Koenen KC, Marshall R. The psychological risks of Vietnam for U.S. veterans: a revisit with new data and methods. Science. 2006;313(5789):979-982. doi:10.1126/science.1128944
17. Marx BP, Bovin MJ, Szafranski DD, et al. Validity of posttraumatic stress disorder service connection status in Veterans Affairs electronic records of Iraq and Afghanistan Veterans. J Clin Psychiatry. 2016;77(4):517-522. doi:10.4088/JCP.14m09666
Updated June 17, 2022
The US Department of Veterans Affairs (VA) provides health care for > 9 million military veterans, nearly half of all former service members.1 Over the past 15 years, there has been a steady and substantial increase in the frequency of disability awards for veterans with post-9/11 military service. Recent data from the Bureau of Labor Statistics indicate that 41% of veterans who served after 9/11 receive service-connected disability benefits compared with 28% of veterans overall.2 More than 5 million veterans receive VA service-related disability benefits.2,3 More than half of the VA $243 billion budget for fiscal year (FY) 2021 ($135.5 billion) was allocated to the Veterans Benefits Administration (VBA), of which $115.7 billion (85%) was allocated specifically for service-related compensation claims payments.4
The VA predicted that VBA will have completed 1.4 million ratings for disability claims in 2021.5 A substantial percentage of these claims will be for mental disorders, specifically posttraumatic stress disorder (PTSD). VA officials testifying before Congress in 2017 noted that the number of PTSD claims had nearly tripled in the previous 10 years.6 As far back as 2013, McNally and Frueh analyzed “the skyrocketing of disability claims,” particularly for PTSD, among veterans who served in Iraq and Afghanistan.7
This large increase has placed an unprecedented burden on the VBA to expand its capacity to conduct initial PTSD disability evaluations that by regulations are completed by psychologists or psychiatrists. This need has led the VBA to make significant changes in the compensation and pension (C&P) process, including a reduced role for Veterans Health Administration (VHA) examiners and increased reliance on non-VA (contract) examiners through the Contract Medical Disability Examination (MDE) program. In 2019, the MDE budget was $1.23 billion; in 2020, it was increased to $1.79 billion, and for 2021, it was $2.23 billion, reflecting the increasing investment of resources in non-VA examiners, ostensibly to both increase capacity and save costs.5
Anecdotally, concerns have been raised regarding inadequate training of contract examiners as well as inadequate reports by these examiners. A 2018 Government Accountability Office (GAO) report concluded that VA lacked the data to determine whether contract examiners were meeting standards for quality, timeliness, and accuracy.8 The GAO report noted that VA required 92% of contractor reports contained no obvious errors, a relatively low target; however, in the first half of 2017, only 1 contractor group met that target. The report noted further that “VBA does not verify if examiners have completed training nor does it collect information to assess training effectiveness in preparing examiners.”8 A subsequent analysis of contract examinations completed by the VA Office of the Inspector General (OIG) in 2019 concluded that the MDE program was “hampered in their ability to provide oversight because of limitations with VBA’s electronic examination management systems, the lack of reliable data, and inadequate staffing of the program.”9
These reports have focused almost exclusively on simple performance metrics, such as timeliness of examination completion. However, the 2018 GAO report referenced isolated “focused reviews” of complaints about the quality of examinations by contract examiners and gave as an example an isolated “review of one contracted examiner who had high rates of diagnosing severe posttraumatic stress disorder.”8 After review indicated the examiner’s reports were of poor quality, the VBA discontinued the examiner’s contract.
Unfortunately, despite such anecdotal reports and isolated actions, to date there are no published reports examining and comparing the quality of PTSD examination reports completed by VHA and contract examiners or the subsequent disability determinations made by the VBA as a result of these evaluations. In a November 2020 letter to the VA Secretary, 11 US Senators expressed “grave concerns” regarding the VA decision to privatize C&P programs noting, among other concerns, that there were “no clinical quality measurement for, or evaluation of, contractor examinations.”10 The letter cited anecdotal evidence of contract examiners not reviewing veteran’s medical records and diagnosing conditions “without supporting evidence.”10
The purpose of the present evaluation was to provide a systematic comparison of the content and quality of initial PTSD disability examinations conducted by VHA and non-VA contract examiners. In addition, this study compared the disability rating decisions resulting from VHA and contract examinations.
Methods
A random sample of 100 Initial PTSD Disability Benefits Questionnaires (DBQs)—structured forms completed by all examiners—were obtained from a list supplied by the VA Office of Performance Analysis and Integrity. All examinations were from the Veterans Integrated Service Network (VISN) 1, encompassing the New England region and were conducted in 2019 and 2020. Two of the 100 cases were excluded for technical reasons, resulting in 98 examination reports. However, the final pool yielded 62 contract examinations and only 36 VHA examinations. To make the sample sizes more comparable, an additional 15 examinations were randomly selected from the local examination database (also VISN 1) to complement the original examination pool.
Once DBQs were retrieved, all identifying information was deleted, and cases were analyzed using assigned record numbers. All coding was completed by the 2 principal investigators, both VA psychologists with extensive training and experience in C&P evaluation and treatment of veterans with PTSD. Due to inherent structural differences between the forms used for VA and contract examinations, raters could not be masked/blinded to the source of the report.
A number of measures were taken to reduce bias and enhance objectivity of rating. First, objectively coded variables (eg, age and sex of veteran, period of service, trauma type, diagnoses rendered by the examiner, impairment category endorsed, number and type of symptoms) were transcribed directly from the DBQ as recorded by the examiner. Second, to rate report quality, an initial categorical rating scale was developed based on predetermined elements of examination quality that were considered essential. After refinement and preliminary analysis of interrater reliabilities, 3 quality-related indices were identified: (1) level of detail in description of key content areas (history before service, service trauma, after service social and vocational history, mental health history, substance use); (2) synthesis of history and findings in explaining opinion rendered; and (3) clarity of opinion regarding causation required “at least as likely as not” degree of confidence. The first 2 quality ratings were based on a 3-point scale (poor, fair, good), and the third variable was coded as yes or no. (eAppendix available at doi:10.12788/fp.0225). Interrater reliabilities calculated based on a subsample of 18 cases, randomly selected and rated by both raters, yielded Cohen κ in the acceptable range (.61, .72, and .89 for detail, synthesis, and clarity, respectively). Finally, for information regarding VBA decision making, rating decision documents contained in the Veterans Benefit Management System database were reviewed to determine whether the veteran was granted service connection for PTSD or another mental disorder based on the examination report in question and, if so, the disability rating percentage awarded. These were recorded independently after all other coding had been completed.
Results
Comparison of VHA and contract examinations revealed no significant differences between groups on relevant sociodemographic and other measures (Table). Missing data were not obtained from other records or sources, and for this study, reflect only what is recorded in the examination reports except for age, which was calculated using veteran’s date of birth and the date of examination.
To examine differences between VHA and contract examinations, the groups were first compared on a set of predetermined objectively coded variables taken directly from the DBQ. The frequency of PTSD diagnoses by VHA (57%) and contract (71%) examiners was not significantly different nor were rates of non-PTSD diagnoses by VHA (51%) and contract (73%) examiners. There also was no difference in the mean number of PTSD symptoms endorsed across PTSD diagnostic criteria B, C, D, and E (maximum of 20) recorded by VHA (9.4) and contract (10.9) examiners.
Contract examiners recorded a significantly greater mean number of “other symptoms” on a checklist of 31 possible symptoms as compared to VHA examiners: 7.3 vs 5.8, respectively (t[104] = 2.27, P < .05). An initial analysis of overall social/vocational impairment ratings coded by examiners did not reveal significant differences between examiner groups. However, when the 2 most severe impairment categories were combined to create a pooled “severe” category, 31% of contract examiners rated veterans as severely impaired compared with only 12% of VHA examiners (χ2 = 5.79, 1 df, P < .05) (Figure 1).
VHA and contract examinations were compared on 3 measures of report quality. Significant differences were found for both level of detail (χ2 = 16.44, 2 df, P < .01) and synthesis (χ2 = 6.68, 2 df, P < .05). Contract examinations were more likely to be rated as poor and less likely to be rated good, with a similar proportion of fair ratings for the 2 examination types (Figures 2 and 3). There was no significant difference in the proportion of VHA and contract examinations providing clear statement of opinion regarding causation (ie, whether or not the diagnosed condition was service related), with the majority rendering an adequate opinion in both examiner groups (VHA, 78%; contract, 79%).
Qualitative review revealed examples of markedly deficient examinations among contract examinations, including several reports that contained no review of records, no report of relevant background, and no mention or assessment of social and vocational function needed to inform opinions about diagnosis and impairment.
Finally, the VBA database was used to compare the resulting disability award decisions made by VBA based on the examination reports in question. Examination by contractors resulted in significantly higher mean service-connected disability ratings for examinees compared with VHA examiners (46.8 vs 33.5, respectively; t[108] = 2.3, P < .05).
Discussion
The present study provides the first reported systematic comparison of VA disability examinations for PTSD completed by examiners employed by the VHA and those hired as contract examiners through the MDE program. Although the frequency of PTSD diagnoses by contract examiners was higher than that of VHA examiners (71% vs 57%, respectively), the difference was not statistically significant. However, contract examiners recorded significantly more symptoms for examinees and rated them as severely impaired more frequently than did their VHA counterparts. In keeping with rating guidelines used by the VBA, these differences in examination content resulted in higher disability ratings for veterans seen by contract examiners.
Along with these elevated reports in symptom and severity ratings, contract examiners were less likely to provide adequate detail in the narrative sections of their reports and less frequently provided a satisfactory explanation and synthesis of relevant history and findings in support of their conclusions. Although not reflected in the statistical analysis, case-by-case review revealed some startlingly inadequate examination reports by contract examiners, several of which contained no review of records, no report or discussion of relevant background, and no discussion or analysis of social and vocational function to inform and support their opinion about level of impairment. None of the VHA examination reports reviewed lacked information to that degree.
Such deficiencies in detail and synthesis run counter to accepted guidelines for the adequate assessment of psychological injury in general and in VA disability claims specifically.11,12 For example, Watson and colleagues proposed that a minimum of 3 hours was required to conduct an initial PTSD examination, with more complex cases possibly taking longer.11 There is no information available about how long contract examiners take to complete their examinations and how that compares with the time taken by VA examiners. The VBA failure to monitor whether or not examiners follow accepted guidelines for PTSD examination has not previously been evaluated. Historically, a large number of clinicians, researchers, and policy critics have raised concerns about the potential for exaggeration or malingering among VA PTSD disability claimants and have urged the need to adequately assess for unreliable reporting and presentation.13,14 However, the possibility of systematic examiner deficiency and/or bias increasing the frequency of false or inflated claims being approved has received little empirical attention.
Although contract examiners did not diagnose PTSD significantly more frequently than VHA examiners (71% vs 57%, respectively), the overall frequency of PTSD diagnosis across both groups (65%) was substantially higher than previous figures that have, on average, estimated the lifetime prevalence of PTSD in trauma-exposed veterans to be about 31%.15 A re-analysis of the same National Vietnam Veterans Readjustment Survey data, but applying more conservative diagnostic criteria, reduced the lifetime prevalence to just under 19%, with point prevalence estimates even lower.15,16
In a study of concordance rates between service connection for PTSD and both current and lifetime diagnosis by independent, structured assessment, Marx and colleagues found that a “significant minority” of veterans who were already receiving service-connected disability for PTSD did not meet lifetime and/or current diagnostic criteria.17 Although it is possible that the group of veterans who were applying for disability benefits in our study had a higher rate of PTSD, it also is possible if not likely that the PTSD examination process overall yields inflated rates of diagnosis and levels of impairment. This speaks to the concern raised by Marx and colleagues who found that veterans with service connection for PTSD who received related benefits “may not have the disorder.”17
Limitations
A methodological limitation of the present study was that, due to structural differences in the DBQ forms used for VHA and contract examinations, the reports could not be de-identified as to examiner type and thus raters could not be masked/blinded. To mitigate bias, a predetermined, piloted, and refined coding and rating plan for report quality metrics was adhered to strictly, and interrater reliabilities were acceptable. Future study is suggested in which all report content is standardized for coding using the same format, which at present would require a complete rewriting of the entire report; this problem could be resolved by having the VBA adopt a more coherent system in which all reports, regardless of examiner type, use a single, standardized template. Further study using larger data sets and expanding to other VA regions also is needed.
Conclusions
The present study suggests that poor examination and report quality—by contract examiners and to a lesser degree VHA examiners—are not uncommon. The findings confirm and extend previous anecdotal reports of deficiencies in PTSD examinations performed by contract examiners and provide empirical support for concerns raised of global deficiencies in the VBA oversight of the MDE program. Such deficiencies have significant implications for the quality and integrity of the VA disability determination process for veterans claiming PTSD related to military service.
The current findings support and strengthen the call for development and management of a structured and enforced training and quality assurance/improvement program for VA PTSD disability examinations. Such training and oversight will be critical to improve the quality and integrity of these examinations, reduce error and waste in VBA’s Compensation and Pension process, and in doing so optimize VA financial resources to best serve veterans’ benefits and health care needs.
Updated June 17, 2022
The US Department of Veterans Affairs (VA) provides health care for > 9 million military veterans, nearly half of all former service members.1 Over the past 15 years, there has been a steady and substantial increase in the frequency of disability awards for veterans with post-9/11 military service. Recent data from the Bureau of Labor Statistics indicate that 41% of veterans who served after 9/11 receive service-connected disability benefits compared with 28% of veterans overall.2 More than 5 million veterans receive VA service-related disability benefits.2,3 More than half of the VA $243 billion budget for fiscal year (FY) 2021 ($135.5 billion) was allocated to the Veterans Benefits Administration (VBA), of which $115.7 billion (85%) was allocated specifically for service-related compensation claims payments.4
The VA predicted that VBA will have completed 1.4 million ratings for disability claims in 2021.5 A substantial percentage of these claims will be for mental disorders, specifically posttraumatic stress disorder (PTSD). VA officials testifying before Congress in 2017 noted that the number of PTSD claims had nearly tripled in the previous 10 years.6 As far back as 2013, McNally and Frueh analyzed “the skyrocketing of disability claims,” particularly for PTSD, among veterans who served in Iraq and Afghanistan.7
This large increase has placed an unprecedented burden on the VBA to expand its capacity to conduct initial PTSD disability evaluations that by regulations are completed by psychologists or psychiatrists. This need has led the VBA to make significant changes in the compensation and pension (C&P) process, including a reduced role for Veterans Health Administration (VHA) examiners and increased reliance on non-VA (contract) examiners through the Contract Medical Disability Examination (MDE) program. In 2019, the MDE budget was $1.23 billion; in 2020, it was increased to $1.79 billion, and for 2021, it was $2.23 billion, reflecting the increasing investment of resources in non-VA examiners, ostensibly to both increase capacity and save costs.5
Anecdotally, concerns have been raised regarding inadequate training of contract examiners as well as inadequate reports by these examiners. A 2018 Government Accountability Office (GAO) report concluded that VA lacked the data to determine whether contract examiners were meeting standards for quality, timeliness, and accuracy.8 The GAO report noted that VA required 92% of contractor reports contained no obvious errors, a relatively low target; however, in the first half of 2017, only 1 contractor group met that target. The report noted further that “VBA does not verify if examiners have completed training nor does it collect information to assess training effectiveness in preparing examiners.”8 A subsequent analysis of contract examinations completed by the VA Office of the Inspector General (OIG) in 2019 concluded that the MDE program was “hampered in their ability to provide oversight because of limitations with VBA’s electronic examination management systems, the lack of reliable data, and inadequate staffing of the program.”9
These reports have focused almost exclusively on simple performance metrics, such as timeliness of examination completion. However, the 2018 GAO report referenced isolated “focused reviews” of complaints about the quality of examinations by contract examiners and gave as an example an isolated “review of one contracted examiner who had high rates of diagnosing severe posttraumatic stress disorder.”8 After review indicated the examiner’s reports were of poor quality, the VBA discontinued the examiner’s contract.
Unfortunately, despite such anecdotal reports and isolated actions, to date there are no published reports examining and comparing the quality of PTSD examination reports completed by VHA and contract examiners or the subsequent disability determinations made by the VBA as a result of these evaluations. In a November 2020 letter to the VA Secretary, 11 US Senators expressed “grave concerns” regarding the VA decision to privatize C&P programs noting, among other concerns, that there were “no clinical quality measurement for, or evaluation of, contractor examinations.”10 The letter cited anecdotal evidence of contract examiners not reviewing veteran’s medical records and diagnosing conditions “without supporting evidence.”10
The purpose of the present evaluation was to provide a systematic comparison of the content and quality of initial PTSD disability examinations conducted by VHA and non-VA contract examiners. In addition, this study compared the disability rating decisions resulting from VHA and contract examinations.
Methods
A random sample of 100 Initial PTSD Disability Benefits Questionnaires (DBQs)—structured forms completed by all examiners—were obtained from a list supplied by the VA Office of Performance Analysis and Integrity. All examinations were from the Veterans Integrated Service Network (VISN) 1, encompassing the New England region and were conducted in 2019 and 2020. Two of the 100 cases were excluded for technical reasons, resulting in 98 examination reports. However, the final pool yielded 62 contract examinations and only 36 VHA examinations. To make the sample sizes more comparable, an additional 15 examinations were randomly selected from the local examination database (also VISN 1) to complement the original examination pool.
Once DBQs were retrieved, all identifying information was deleted, and cases were analyzed using assigned record numbers. All coding was completed by the 2 principal investigators, both VA psychologists with extensive training and experience in C&P evaluation and treatment of veterans with PTSD. Due to inherent structural differences between the forms used for VA and contract examinations, raters could not be masked/blinded to the source of the report.
A number of measures were taken to reduce bias and enhance objectivity of rating. First, objectively coded variables (eg, age and sex of veteran, period of service, trauma type, diagnoses rendered by the examiner, impairment category endorsed, number and type of symptoms) were transcribed directly from the DBQ as recorded by the examiner. Second, to rate report quality, an initial categorical rating scale was developed based on predetermined elements of examination quality that were considered essential. After refinement and preliminary analysis of interrater reliabilities, 3 quality-related indices were identified: (1) level of detail in description of key content areas (history before service, service trauma, after service social and vocational history, mental health history, substance use); (2) synthesis of history and findings in explaining opinion rendered; and (3) clarity of opinion regarding causation required “at least as likely as not” degree of confidence. The first 2 quality ratings were based on a 3-point scale (poor, fair, good), and the third variable was coded as yes or no. (eAppendix available at doi:10.12788/fp.0225). Interrater reliabilities calculated based on a subsample of 18 cases, randomly selected and rated by both raters, yielded Cohen κ in the acceptable range (.61, .72, and .89 for detail, synthesis, and clarity, respectively). Finally, for information regarding VBA decision making, rating decision documents contained in the Veterans Benefit Management System database were reviewed to determine whether the veteran was granted service connection for PTSD or another mental disorder based on the examination report in question and, if so, the disability rating percentage awarded. These were recorded independently after all other coding had been completed.
Results
Comparison of VHA and contract examinations revealed no significant differences between groups on relevant sociodemographic and other measures (Table). Missing data were not obtained from other records or sources, and for this study, reflect only what is recorded in the examination reports except for age, which was calculated using veteran’s date of birth and the date of examination.
To examine differences between VHA and contract examinations, the groups were first compared on a set of predetermined objectively coded variables taken directly from the DBQ. The frequency of PTSD diagnoses by VHA (57%) and contract (71%) examiners was not significantly different nor were rates of non-PTSD diagnoses by VHA (51%) and contract (73%) examiners. There also was no difference in the mean number of PTSD symptoms endorsed across PTSD diagnostic criteria B, C, D, and E (maximum of 20) recorded by VHA (9.4) and contract (10.9) examiners.
Contract examiners recorded a significantly greater mean number of “other symptoms” on a checklist of 31 possible symptoms as compared to VHA examiners: 7.3 vs 5.8, respectively (t[104] = 2.27, P < .05). An initial analysis of overall social/vocational impairment ratings coded by examiners did not reveal significant differences between examiner groups. However, when the 2 most severe impairment categories were combined to create a pooled “severe” category, 31% of contract examiners rated veterans as severely impaired compared with only 12% of VHA examiners (χ2 = 5.79, 1 df, P < .05) (Figure 1).
VHA and contract examinations were compared on 3 measures of report quality. Significant differences were found for both level of detail (χ2 = 16.44, 2 df, P < .01) and synthesis (χ2 = 6.68, 2 df, P < .05). Contract examinations were more likely to be rated as poor and less likely to be rated good, with a similar proportion of fair ratings for the 2 examination types (Figures 2 and 3). There was no significant difference in the proportion of VHA and contract examinations providing clear statement of opinion regarding causation (ie, whether or not the diagnosed condition was service related), with the majority rendering an adequate opinion in both examiner groups (VHA, 78%; contract, 79%).
Qualitative review revealed examples of markedly deficient examinations among contract examinations, including several reports that contained no review of records, no report of relevant background, and no mention or assessment of social and vocational function needed to inform opinions about diagnosis and impairment.
Finally, the VBA database was used to compare the resulting disability award decisions made by VBA based on the examination reports in question. Examination by contractors resulted in significantly higher mean service-connected disability ratings for examinees compared with VHA examiners (46.8 vs 33.5, respectively; t[108] = 2.3, P < .05).
Discussion
The present study provides the first reported systematic comparison of VA disability examinations for PTSD completed by examiners employed by the VHA and those hired as contract examiners through the MDE program. Although the frequency of PTSD diagnoses by contract examiners was higher than that of VHA examiners (71% vs 57%, respectively), the difference was not statistically significant. However, contract examiners recorded significantly more symptoms for examinees and rated them as severely impaired more frequently than did their VHA counterparts. In keeping with rating guidelines used by the VBA, these differences in examination content resulted in higher disability ratings for veterans seen by contract examiners.
Along with these elevated reports in symptom and severity ratings, contract examiners were less likely to provide adequate detail in the narrative sections of their reports and less frequently provided a satisfactory explanation and synthesis of relevant history and findings in support of their conclusions. Although not reflected in the statistical analysis, case-by-case review revealed some startlingly inadequate examination reports by contract examiners, several of which contained no review of records, no report or discussion of relevant background, and no discussion or analysis of social and vocational function to inform and support their opinion about level of impairment. None of the VHA examination reports reviewed lacked information to that degree.
Such deficiencies in detail and synthesis run counter to accepted guidelines for the adequate assessment of psychological injury in general and in VA disability claims specifically.11,12 For example, Watson and colleagues proposed that a minimum of 3 hours was required to conduct an initial PTSD examination, with more complex cases possibly taking longer.11 There is no information available about how long contract examiners take to complete their examinations and how that compares with the time taken by VA examiners. The VBA failure to monitor whether or not examiners follow accepted guidelines for PTSD examination has not previously been evaluated. Historically, a large number of clinicians, researchers, and policy critics have raised concerns about the potential for exaggeration or malingering among VA PTSD disability claimants and have urged the need to adequately assess for unreliable reporting and presentation.13,14 However, the possibility of systematic examiner deficiency and/or bias increasing the frequency of false or inflated claims being approved has received little empirical attention.
Although contract examiners did not diagnose PTSD significantly more frequently than VHA examiners (71% vs 57%, respectively), the overall frequency of PTSD diagnosis across both groups (65%) was substantially higher than previous figures that have, on average, estimated the lifetime prevalence of PTSD in trauma-exposed veterans to be about 31%.15 A re-analysis of the same National Vietnam Veterans Readjustment Survey data, but applying more conservative diagnostic criteria, reduced the lifetime prevalence to just under 19%, with point prevalence estimates even lower.15,16
In a study of concordance rates between service connection for PTSD and both current and lifetime diagnosis by independent, structured assessment, Marx and colleagues found that a “significant minority” of veterans who were already receiving service-connected disability for PTSD did not meet lifetime and/or current diagnostic criteria.17 Although it is possible that the group of veterans who were applying for disability benefits in our study had a higher rate of PTSD, it also is possible if not likely that the PTSD examination process overall yields inflated rates of diagnosis and levels of impairment. This speaks to the concern raised by Marx and colleagues who found that veterans with service connection for PTSD who received related benefits “may not have the disorder.”17
Limitations
A methodological limitation of the present study was that, due to structural differences in the DBQ forms used for VHA and contract examinations, the reports could not be de-identified as to examiner type and thus raters could not be masked/blinded. To mitigate bias, a predetermined, piloted, and refined coding and rating plan for report quality metrics was adhered to strictly, and interrater reliabilities were acceptable. Future study is suggested in which all report content is standardized for coding using the same format, which at present would require a complete rewriting of the entire report; this problem could be resolved by having the VBA adopt a more coherent system in which all reports, regardless of examiner type, use a single, standardized template. Further study using larger data sets and expanding to other VA regions also is needed.
Conclusions
The present study suggests that poor examination and report quality—by contract examiners and to a lesser degree VHA examiners—are not uncommon. The findings confirm and extend previous anecdotal reports of deficiencies in PTSD examinations performed by contract examiners and provide empirical support for concerns raised of global deficiencies in the VBA oversight of the MDE program. Such deficiencies have significant implications for the quality and integrity of the VA disability determination process for veterans claiming PTSD related to military service.
The current findings support and strengthen the call for development and management of a structured and enforced training and quality assurance/improvement program for VA PTSD disability examinations. Such training and oversight will be critical to improve the quality and integrity of these examinations, reduce error and waste in VBA’s Compensation and Pension process, and in doing so optimize VA financial resources to best serve veterans’ benefits and health care needs.
1. US Department of Veterans Affairs, Veterans Health Adminstration. About VHA. Updated April 23, 2021. Accessed January 6, 2022. www.va.gov/health/aboutvha.asp
2. US Department of Labor, Bureau of Labor Statistics. News release. Employment situation of veterans—2020. Published March 18, 2020. Accessed January 6, 2022. https://www.bls.gov/news.release/pdf/vet.pdf
3. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Department of Veterans Affairs statistics at a glance. Updated December 31, 2020. Accessed January 6, 2022. https://www.va.gov/vetdata/docs/Quickfacts/Stats_at_a_glance_12_31_20.PDF
4. US Department of Veterans Affairs. FY 2021 Budget submission: budget in brief. Published February 2020. Accessed January 6, 2022. https://www.va.gov/budget/docs/summary/archive/FY-2021-VA-BudgetSubmission.zip
5. US Department of Veterans Affairs. FY 2021 budget submission: benefits and burial programs and Departmental Administration volume 3 of 4:178. Published February 2020. Accessed January 6, 2022. https://www.va.gov/budget/docs/summary/archive/FY-2021-VA-BudgetSubmission.zip
6. Statement of Ronald Burke, assistant deputy under secretary, office of field operations Veterans Benefits Administration before the Subcommittee on Disability And Memorial Affairs of the House Committee on Veterans’ Affairs. Published July 25, 2017. Accessed January 6, 2022. https://www.congress.gov/115/meeting/house/106322/witnesses/HHRG-115-VR09-Wstate-BurkeR-20170725.pdf
7. McNally RJ, Frueh BC. Why are Iraq and Afghanistan War veterans seeking PTSD disability compensation at unprecedented rates? J Anxiety Disord. 2013;27(5):520-526. doi:10.1016/j.janxdis.2013.07.002
8. US Government Accountability Office. VA disability exams: improved performance analysis and training oversight needed for contracted exams. GAO-19-13. Published October 2018. Accessed January 6, 2022. https://www.gao.gov/assets/gao-19-13.pdf
9. US Department of Veterans Affairs, Office of Inspector General. Inadequate oversight of contracted disability exam cancellations. Report #18-04266-115. Published June 10, 2019. Accessed January 6, 2022. https://www.va.gov/oig/pubs/VAOIG-18-04266-115.pdf
10. Letter to VA Secretary Wilkie. Published November 11, 2020. Accessed January 6, 2022. https://www.veterans.senate.gov/download/candp-exam-va-letter
11. Watson PW, McFall M, McBrine C, Schnurr PP, Friedman MJ, Keane TM, Hamblen JL (2005). Best practice manual for posttraumatic stress disorder (PTSD) compensation and pension examinations. Portland, OR: Northwest Network Mental Illness Research, Education, and Clinical Center, VA Puget Sound Healthcare System.
12. Worthen MD, Moering RG. A practical guide to conducting VA compensation and pension exams for PTSD and other mental disorders. Psychol Inj and Law. 2011;4:187-216. doi:10.1007/s12207-011-9115-2
13. DeViva JC, Bloem WD. Symptom exaggeration and compensation seeking among combat veterans with posttraumatic stress disorder. J Trauma Stress. 2003;16(5):503-507. doi:10.1023/A:1025766713188
14. Ray CL. Feigning screeners in VA PTSD compensation and pension examinations. Psychol Inj and Law. 2014;7:370-387. doi:10.1007/s12207-014-9210-2
15. Kulka RA, Schlenger WE, Fairbank JA, et al. Trauma and the Vietnam War Generation: Report of Findings From the National Vietnam Veterans Readjustment Study. Brunner Mazel Publishers; 1990.
16. Dohrenwend BP, Turner JB, Turse NA, Adams BG, Koenen KC, Marshall R. The psychological risks of Vietnam for U.S. veterans: a revisit with new data and methods. Science. 2006;313(5789):979-982. doi:10.1126/science.1128944
17. Marx BP, Bovin MJ, Szafranski DD, et al. Validity of posttraumatic stress disorder service connection status in Veterans Affairs electronic records of Iraq and Afghanistan Veterans. J Clin Psychiatry. 2016;77(4):517-522. doi:10.4088/JCP.14m09666
1. US Department of Veterans Affairs, Veterans Health Adminstration. About VHA. Updated April 23, 2021. Accessed January 6, 2022. www.va.gov/health/aboutvha.asp
2. US Department of Labor, Bureau of Labor Statistics. News release. Employment situation of veterans—2020. Published March 18, 2020. Accessed January 6, 2022. https://www.bls.gov/news.release/pdf/vet.pdf
3. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Department of Veterans Affairs statistics at a glance. Updated December 31, 2020. Accessed January 6, 2022. https://www.va.gov/vetdata/docs/Quickfacts/Stats_at_a_glance_12_31_20.PDF
4. US Department of Veterans Affairs. FY 2021 Budget submission: budget in brief. Published February 2020. Accessed January 6, 2022. https://www.va.gov/budget/docs/summary/archive/FY-2021-VA-BudgetSubmission.zip
5. US Department of Veterans Affairs. FY 2021 budget submission: benefits and burial programs and Departmental Administration volume 3 of 4:178. Published February 2020. Accessed January 6, 2022. https://www.va.gov/budget/docs/summary/archive/FY-2021-VA-BudgetSubmission.zip
6. Statement of Ronald Burke, assistant deputy under secretary, office of field operations Veterans Benefits Administration before the Subcommittee on Disability And Memorial Affairs of the House Committee on Veterans’ Affairs. Published July 25, 2017. Accessed January 6, 2022. https://www.congress.gov/115/meeting/house/106322/witnesses/HHRG-115-VR09-Wstate-BurkeR-20170725.pdf
7. McNally RJ, Frueh BC. Why are Iraq and Afghanistan War veterans seeking PTSD disability compensation at unprecedented rates? J Anxiety Disord. 2013;27(5):520-526. doi:10.1016/j.janxdis.2013.07.002
8. US Government Accountability Office. VA disability exams: improved performance analysis and training oversight needed for contracted exams. GAO-19-13. Published October 2018. Accessed January 6, 2022. https://www.gao.gov/assets/gao-19-13.pdf
9. US Department of Veterans Affairs, Office of Inspector General. Inadequate oversight of contracted disability exam cancellations. Report #18-04266-115. Published June 10, 2019. Accessed January 6, 2022. https://www.va.gov/oig/pubs/VAOIG-18-04266-115.pdf
10. Letter to VA Secretary Wilkie. Published November 11, 2020. Accessed January 6, 2022. https://www.veterans.senate.gov/download/candp-exam-va-letter
11. Watson PW, McFall M, McBrine C, Schnurr PP, Friedman MJ, Keane TM, Hamblen JL (2005). Best practice manual for posttraumatic stress disorder (PTSD) compensation and pension examinations. Portland, OR: Northwest Network Mental Illness Research, Education, and Clinical Center, VA Puget Sound Healthcare System.
12. Worthen MD, Moering RG. A practical guide to conducting VA compensation and pension exams for PTSD and other mental disorders. Psychol Inj and Law. 2011;4:187-216. doi:10.1007/s12207-011-9115-2
13. DeViva JC, Bloem WD. Symptom exaggeration and compensation seeking among combat veterans with posttraumatic stress disorder. J Trauma Stress. 2003;16(5):503-507. doi:10.1023/A:1025766713188
14. Ray CL. Feigning screeners in VA PTSD compensation and pension examinations. Psychol Inj and Law. 2014;7:370-387. doi:10.1007/s12207-014-9210-2
15. Kulka RA, Schlenger WE, Fairbank JA, et al. Trauma and the Vietnam War Generation: Report of Findings From the National Vietnam Veterans Readjustment Study. Brunner Mazel Publishers; 1990.
16. Dohrenwend BP, Turner JB, Turse NA, Adams BG, Koenen KC, Marshall R. The psychological risks of Vietnam for U.S. veterans: a revisit with new data and methods. Science. 2006;313(5789):979-982. doi:10.1126/science.1128944
17. Marx BP, Bovin MJ, Szafranski DD, et al. Validity of posttraumatic stress disorder service connection status in Veterans Affairs electronic records of Iraq and Afghanistan Veterans. J Clin Psychiatry. 2016;77(4):517-522. doi:10.4088/JCP.14m09666
Evaluating the Impact of a Urinalysis to Reflex Culture Process Change in the Emergency Department at a Veterans Affairs Hospital
Automated urine cultures (UCs) following urinalysis (UA) are often used in emergency departments (EDs) to identify urinary tract infections (UTIs). The fast-paced environment of the ED makes this method of proactive collection and facilitation of UC favorable. However, results are often reported as no organism growth or the growth of clinically insignificant organisms, leading to the overdetection and overtreatment of asymptomatic bacteriuria (ASB).1-3 An estimated 30 to 60% of patients with ASB receive unwarranted antibiotic treatment, which is associated with an increased risk of developing Clostridioides difficile infection and contributes to the development of antimicrobial resistance.4-10 The costs associated with UC are an important consideration given the use of resources, the time and effort required to collect and process large numbers of negative cultures, and further efforts devoted to the follow-up of ED culture results.
Changes in traditional testing involving testing of both a UA and UC to reflex testing where urine specimens undergo culture only if they meet certain criteria have been described.11-14 This change in traditional testing aims to reduce the number of potentially unnecessary cultures performed without compromising clinical care. Leukocyte quantity in the UA has been shown to be a reliable predictor of true infection.11,15 Fok and colleagues demonstrated that reflex urine testing in ambulatory male urology patients in which cultures were done on only urine specimens with > 5 white blood cells per high-power field (WBC/HPF) would have missed only 7% of positive UCs, while avoiding 69% of cultures.11
At the Edward Hines, Jr Veterans Affairs Hospital (Hines VA), inappropriate UC ordering and treatment for ASB has been identified as an area needing improvement. An evaluation was conducted at the facility to determine the population of inpatient veterans with a positive UC who were appropriately managed. Of the 113 study patients with a positive UC included in this review, 77 (68%) had a diagnosis of ASB, with > 80% of patients with ASB (and no other suspected infections) receiving antimicrobial therapy.8 A subsequent evaluation was conducted at the Hines VA ED to evaluate UTI treatment and follow-up. Of the 173 ED patients included, 23% received antibiotic therapy for an ASB and 60% had a UA and UC collected but did not report symptoms.9 Finally, a review by the Hines VA laboratory showed that in May 2017, of 359 UCs sent from various locations of the hospital, 38% were obtained in the setting of a negative UA.
A multidisciplinary group with representation from primary care, infectious diseases, pharmacy, nursing, laboratory, and informatics was created with a goal to improve the workup and management of UTIs. In addition to periodic education for the clinicians regarding appropriate use and interpretation of UA and UC along with judicious use of antimicrobials especially in the setting of ASB, a UA to reflex culture process change was implemented. This allowed for automatic cancellation of a UC in the setting of a negative UA, which was designed to help facilitate appropriate UC ordering.
Methods
The primary objective of this study was to compare the frequency of inappropriate UC use and inappropriate antibiotic prescribing pre- and postimplementation of this UA to reflex culture process change. An inappropriate UC was defined as a UC ordered despite a negative UA in asymptomatic patients. Inappropriate antibiotic prescribing was defined as treatment of patients with ASB. The secondary objective evaluated postintervention data to assess the frequency of outpatient, ED, and hospital visits for UTI-related symptoms in the group of patients that had a UC cancelled as a result of the new process change (within a 7-day period of the initial UA) to determine whether patients with true infections were missed due to the process change.
Study Design and Setting
This pre-post quality improvement (QI) study analyzed the UC-ordering practices for UTIs sent from the ED at the Hines VA. This VA is a 483-bed tertiary care hospital in Chicago, Illinois, and serves > 57,000 veterans and about 23,000 ED visits annually. This study was approved by the Edward Hines, Jr VA Institutional Review Board as a quality assurance/QI proposal prior to data collection.
Patient Selection
All patients who
When comparing postintervention data with preintervention data for the primary study objective, the same exclusion criteria from the 2015 study were applied to the present study, which excluded ED patients who were admitted for inpatient care, concurrent antibiotic therapy for a non-UTI indication, duplicate cultures, and use of chronic bladder management devices. All patients identified as receiving a UA during the specified postintervention study period were included for evaluation of the secondary study objective.
Interventions
After physician education, an ED process change was implemented on October 3, 2017. This process change involved the creation of new order sets in the EHR that allowed clinicians to order a UA only, a UA with culture that would be cancelled by laboratory personnel if the UA did not result in > 5 WBC/HPF, and a UA with culture designated as do not cancel, where the UC was processed regardless of the UA results. The scenarios in which the latter option was considered appropriate were listed on the ordering screen and included pregnancy, a genitourinary procedure with necessary preoperative culture, and neutropenia.
Measurements
Postimplementation, all UAs were reviewed and grouped as follows: (1) positive UA with subsequent UC; (2) negative UA, culture cancelled; (3) only UA ordered (no culture); or (4) do not cancel UC ordered. Of the UAs that were analyzed, the following data were collected: demographics, comorbidities, concurrent medications for benign prostatic hyperplasia (BPH) and/or overactive bladder (OAB), documented allergies/adverse drug reactions to antibiotics, date of ED visit, documented UTI signs/symptoms (defined as frequency, urgency, dysuria, fever, suprapubic pain, or altered mental status in patients unable to verbalize urinary symptoms), UC results and susceptibilities, number of UCs repeated within 7 days after initial UA, requirement of antibiotic for UTI within 7 days of initial UA, antibiotic prescribed, duration of antibiotic therapy, and outpatient visits, ED visits, or need for hospital admission within 7 days of the initial UA for UTI-related symptoms. Other relevant UA and UC data that could not be obtained from the EHR were collected by generating a report using the Veterans Information Systems and Technology Architecture (VistA).
Analysis
Statistical analysis was performed using SAS v9.4. Independent t tests and Fisher exact tests were used to describe difference pre- and postintervention. Statistical significance was considered for P < .05. Based on results from the previous study conducted at this facility in addition to a literature review, it was determined that 92 patients in each group (pre- and postintervention) would be necessary to detect a 15% increase in percentage of patients appropriately treated for a UTI.
Results
There were 684 UAs evaluated from ED visits, 429 preintervention and 255 postintervention. The 255 patients were evaluated for the secondary objective of the study. Of the 255 patients with UAs identified postintervention, 150 were excluded based on the predefined exclusion criteria, and the remaining 105 were compared with the 173 patients from the preintervention group and were included in the analysis for the primary objective (Figure 1).
Patients in the postintervention group were younger than those in the preintervention group (P < .02): otherwise the groups were similar (Table 1). Inappropriate antibiotics for ASB decreased from 10.2% preintervention to 1.9% postintervention (odds ratio, 0.17; P = .01) (Table 2). UC processing despite a negative UA significantly decreased from 100% preintervention to 38.6% postintervention (P < .001) (Table 3). In patients with a negative UA, antibiotic prescribing decreased by 25.3% postintervention, but this difference was not statistically significant.
Postintervention, of 255 UAs evaluated, 95 (37.3%) were positive with a processed UC and 95 (37.3%) were negative with UC cancelled, 43 (16.9%) were ordered as DNC, and 22 (8.6%) were ordered without a UC (Figure 2). Twenty-eight of the 95 (29.5%) UAs with processed UCs did not meet the criteria for a positive UA and were not designated as DNC. When the UCs of this subgroup of patients were further analyzed, we found that 2 of the cultures were positive of which 1 patient was symptomatic and required antibiotic therapy.
Of the 95 patients with a negative UA, 69 (72.6%) presented without any UTI-related symptoms. In this group, there were no reports of outpatient visits, ED visits, or hospital admissions within 7 days of initial UA for UTI-related symptoms. None of the UCs ordered as DNC had a supporting reason identified. Nonetheless, the UC results from this patient subgroup also were analyzed further and resulted in 4 patients with negative UA and positive subsequent UC, 1 was symptomatic and required antibiotic therapy.
Discussion
A simple process change at the Hines VA resulted in benefits related to antimicrobial stewardship without conferring adverse outcomes on patient safety. Both UC processing despite a negative UA and inappropriate antibiotic prescribing for ASB were reduced significantly postintervention. This process change was piloted in the ED where UCs are often included as part of the initial diagnostic testing in patients who may not report UTI-related symptoms but for whom a UC is often bundled with other infectious workup, depending on the patient presentation.
Reflex testing of urine specimens has been described in the literature, both in an exploratory nature where impact of a reflex UC cancellation protocol based on certain UA criteria is measured by percent reduction of UCs processed as well as results of such interventions implemented into clinical practice.11-13 A retrospective study performed at the University of North Carolina Medical Center evaluated patients who presented to the ED during a 6-month period and had both an automated UA and UC collected. UC processing was restricted to UA that was positive for nitrites, leukocyte esterase, bacteria, or > 10 WBC/HPF. Use of this reflex culture cancellation protocol could have eliminated 604 of the 1546 (39.1%) cultures processed. However, 11 of the 314 (3.5%) positive cultures could have been missed.13 This same protocol was externally validated at another large academic ED setting, where similar results were found.14
In clinical practice, there is a natural tendency to reflexively prescribe antibiotics based on the results of a positive UC due to the hesitancy in ignoring these results, despite lack of a suspicion for a true infection. Leis and colleagues explored this in a proof-of-concept study evaluating the impact of discontinuing the routine reporting of positive UC results from noncatheterized inpatients and requesting clinicians to call the laboratory for results if a UTI was suspected.16 This intervention resulted in a statistically significant reduction in treatment of ASB in noncatheterized patients from 48 to 12% pre- and postintervention. Clinicians requested culture results only 14% of the time, and there were no adverse outcomes among untreated noncatheterized patients. More recently, a QI study conducted at a large community hospital in Toronto, Ontario, Canada, implemented a 2-step model of care for urine collection.17 UC was collected but only processed by the microbiology laboratory if the ED physicians deemed it necessary after clinical assessment.
After implementation, there was a decrease in the proportion of ED visits associated with processed UC (from 6.0% to 4.7% of visits per week; P < .001), ED visits associated with callbacks for processing UC (1.8% to 1.1% of visits per month; P < .001), and antimicrobial prescriptions for urinary symptoms among hospitalized patients (from 20.6% to 10.9%; P < .001). Equally important, despite the 937 cases in which urine was collected but cultures were not processed, no evidence of untreated UTIs was identified.17
The results from the present study similarly demonstrate minimal concern for potentially undertreating these patients. As seen in the subgroup of patients included in the positive UA group, which did not meet criteria for positive UA per protocol (n = 29), only 2 of the subsequent cultures were positive, of which only 1 patient required antibiotic therapy based on the clinical presentation. In addition, in the group of negative UAs with subsequent cancellation of the UC, there were no found reports of outpatient visits, ED visits, or hospital admissions within 7 days of the initial UA for UTI-related symptoms.
Limitations
This single-center, pre-post QI study was not without limitations. Manual chart reviews were required, and accuracy of information was dependent on clinician documentation and assessment of UTI-related symptoms. The population studied was predominately older males; thus, results may not be applicable to females or young adults. Additionally, recognition of a negative UA and subsequent cancellation of the UC was dependent on laboratory personnel. As noted in the patient group with a positive UA, some of these UAs were negative and may have been overlooked; therefore, subsequent UCs were inappropriately processed. However, this occurred infrequently and confirmed the low probability of true UTI in the setting of a negative UA. Follow-up for UTI-related symptoms may not have been captured if a patient had presented to an outside facility. Last, definitions of a positive UA differed slightly between the pre- and postintervention groups. The preintervention study defined a positive UA as a WBC count > 5 WBC/HPF and positive leukocyte esterase, whereas the present study defined a positive UA with a WBC count > 5. This may have resulted in an overestimation of positive UA in the postintervention group.
Conclusions
Better selective use of UC testing may improve stewardship resources and reduce costs impacting both ED and clinical laboratories. Furthermore, benefits can include a reduction in the use of time and resources required to collect samples for culture, use of test supplies, the time and effort required to process the large number of negative cultures, and resources devoted to the follow-up of these ED culture results. The described UA to reflex culture process change demonstrated a significant reduction in the processing of inappropriate UC and unnecessary antibiotics for ASB. There were no missed UTIs or other adverse patient outcomes noted. This process change has been implemented in all departments at the Hines VA and additional data will be collected to ensure consistent outcomes.
1. Chironda B, Clancy S, Powis JE. Optimizing urine culture collection in the emergency department using frontline ownership interventions. Clin Infect Dis. 2014;59(7):1038-1039. doi:10.1093/cid/ciu412
2. Nagurney JT, Brown DF, Chang Y, Sane S, Wang AC, Weiner JB. Use of diagnostic testing in the emergency department for patients presenting with non-traumatic abdominal pain. J Emerg Med. 2003;25(4):363-371. doi:10.1016/s0736-4679(03)00237-3
3. Lammers RL, Gibson S, Kovacs D, Sears W, Strachan G. Comparison of test characteristics of urine dipstick and urinalysis at various test cutoff points. Ann Emerg Med. 2001;38(5):505-512. doi:10.1067/mem.2001.119427
4. Nicolle LE, Gupta K, Bradley SF, et al. Clinical practice guideline for the management of asymptomatic bacteriuria: 2019 update by the Infectious Diseases Society of America. Clin Infect Dis. 2019;68(10):1611-1615. doi:10.1093/cid/ciy1121
5. Trautner BW, Grigoryan L, Petersen NJ, et al. Effectiveness of an antimicrobial stewardship approach for urinary catheter-associated asymptomatic bacteriuria. JAMA Intern Med. 2015;175(7):1120-1127. doi:10.1001/jamainternmed.2015.1878
6. Hartley S, Valley S, Kuhn L, et al. Overtreatment of asymptomatic bacteriuria: identifying targets for improvement. Infect Control Hosp Epidemiol. 2015;36(4):470-473. doi:10.1017/ice.2014.73
7. Bader MS, Loeb M, Brooks AA. An update on the management of urinary tract infections in the era of antimicrobial resistance. Postgrad Med. 2017;129(2):242-258. doi:10.1080/00325481.2017.1246055
8. Spivak ES, Burk M, Zhang R, et al. Management of bacteriuria in Veterans Affairs hospitals. Clin Infect Dis. 2017;65(6):910-917. doi:10.1093/cid/cix474
9. Kim EY, Patel U, Patel B, Suda KJ. Evaluation of bacteriuria treatment and follow-up initiated in the emergency department at a Veterans Affairs hospital. J Pharm Technol. 2017;33(5):183-188. doi:10.1177/8755122517718214
10. Brown E, Talbot GH, Axelrod P, Provencher M, Hoegg C. Risk factors for Clostridium difficile toxin-associated diarrhea. Infect Control Hosp Epidemiol. 1990;11(6):283-290. doi:10.1086/646173
11. Fok C, Fitzgerald MP, Turk T, Mueller E, Dalaza L, Schreckenberger P. Reflex testing of male urine specimens misses few positive cultures may reduce unnecessary testing of normal specimens. Urology. 2010;75(1):74-76. doi:10.1016/j.urology.2009.08.071
12. Munigala S, Jackups RR Jr, Poirier RF, et al. Impact of order set design on urine culturing practices at an academic medical centre emergency department. BMJ Qual Saf. 2018;27(8):587-592. doi:10.1136/bmjqs-2017-006899
13. Jones CW, Culbreath KD, Mehrotra A, Gilligan PH. Reflect urine culture cancellation in the emergency department. J Emerg Med. 2014;46(1):71-76. doi:10.1016/j.jemermed.2013.08.042
14. Hertz JT, Lescallette RD, Barrett TW, Ward MJ, Self WH. External validation of an ED protocol for reflex urine culture cancelation. Am J Emerg Med. 2015;33(12):1838-1839. doi:10.1016/j.ajem.2015.09.026
15. Stamm WE. Measurement of pyuria and its relation to bacteriuria. Am J Med. 1983;75(1B):53-58. doi:10.1016/0002-9343(83)90073-6
16. Leis JA, Rebick GW, Daneman N, et al. Reducing antimicrobial therapy for asymptomatic bacteriuria among noncatheterized inpatients: a proof-of-concept study. Clin Infect Dis. 2014;58(7):980-983. doi:10.1093/cid/ciu010
17. Stagg A, Lutz H, Kirpalaney S, et al. Impact of two-step urine culture ordering in the emergency department: a time series analysis. BMJ Qual Saf. 2017;27:140-147. doi:10.1136/bmjqs-2016-006250
Automated urine cultures (UCs) following urinalysis (UA) are often used in emergency departments (EDs) to identify urinary tract infections (UTIs). The fast-paced environment of the ED makes this method of proactive collection and facilitation of UC favorable. However, results are often reported as no organism growth or the growth of clinically insignificant organisms, leading to the overdetection and overtreatment of asymptomatic bacteriuria (ASB).1-3 An estimated 30 to 60% of patients with ASB receive unwarranted antibiotic treatment, which is associated with an increased risk of developing Clostridioides difficile infection and contributes to the development of antimicrobial resistance.4-10 The costs associated with UC are an important consideration given the use of resources, the time and effort required to collect and process large numbers of negative cultures, and further efforts devoted to the follow-up of ED culture results.
Changes in traditional testing involving testing of both a UA and UC to reflex testing where urine specimens undergo culture only if they meet certain criteria have been described.11-14 This change in traditional testing aims to reduce the number of potentially unnecessary cultures performed without compromising clinical care. Leukocyte quantity in the UA has been shown to be a reliable predictor of true infection.11,15 Fok and colleagues demonstrated that reflex urine testing in ambulatory male urology patients in which cultures were done on only urine specimens with > 5 white blood cells per high-power field (WBC/HPF) would have missed only 7% of positive UCs, while avoiding 69% of cultures.11
At the Edward Hines, Jr Veterans Affairs Hospital (Hines VA), inappropriate UC ordering and treatment for ASB has been identified as an area needing improvement. An evaluation was conducted at the facility to determine the population of inpatient veterans with a positive UC who were appropriately managed. Of the 113 study patients with a positive UC included in this review, 77 (68%) had a diagnosis of ASB, with > 80% of patients with ASB (and no other suspected infections) receiving antimicrobial therapy.8 A subsequent evaluation was conducted at the Hines VA ED to evaluate UTI treatment and follow-up. Of the 173 ED patients included, 23% received antibiotic therapy for an ASB and 60% had a UA and UC collected but did not report symptoms.9 Finally, a review by the Hines VA laboratory showed that in May 2017, of 359 UCs sent from various locations of the hospital, 38% were obtained in the setting of a negative UA.
A multidisciplinary group with representation from primary care, infectious diseases, pharmacy, nursing, laboratory, and informatics was created with a goal to improve the workup and management of UTIs. In addition to periodic education for the clinicians regarding appropriate use and interpretation of UA and UC along with judicious use of antimicrobials especially in the setting of ASB, a UA to reflex culture process change was implemented. This allowed for automatic cancellation of a UC in the setting of a negative UA, which was designed to help facilitate appropriate UC ordering.
Methods
The primary objective of this study was to compare the frequency of inappropriate UC use and inappropriate antibiotic prescribing pre- and postimplementation of this UA to reflex culture process change. An inappropriate UC was defined as a UC ordered despite a negative UA in asymptomatic patients. Inappropriate antibiotic prescribing was defined as treatment of patients with ASB. The secondary objective evaluated postintervention data to assess the frequency of outpatient, ED, and hospital visits for UTI-related symptoms in the group of patients that had a UC cancelled as a result of the new process change (within a 7-day period of the initial UA) to determine whether patients with true infections were missed due to the process change.
Study Design and Setting
This pre-post quality improvement (QI) study analyzed the UC-ordering practices for UTIs sent from the ED at the Hines VA. This VA is a 483-bed tertiary care hospital in Chicago, Illinois, and serves > 57,000 veterans and about 23,000 ED visits annually. This study was approved by the Edward Hines, Jr VA Institutional Review Board as a quality assurance/QI proposal prior to data collection.
Patient Selection
All patients who
When comparing postintervention data with preintervention data for the primary study objective, the same exclusion criteria from the 2015 study were applied to the present study, which excluded ED patients who were admitted for inpatient care, concurrent antibiotic therapy for a non-UTI indication, duplicate cultures, and use of chronic bladder management devices. All patients identified as receiving a UA during the specified postintervention study period were included for evaluation of the secondary study objective.
Interventions
After physician education, an ED process change was implemented on October 3, 2017. This process change involved the creation of new order sets in the EHR that allowed clinicians to order a UA only, a UA with culture that would be cancelled by laboratory personnel if the UA did not result in > 5 WBC/HPF, and a UA with culture designated as do not cancel, where the UC was processed regardless of the UA results. The scenarios in which the latter option was considered appropriate were listed on the ordering screen and included pregnancy, a genitourinary procedure with necessary preoperative culture, and neutropenia.
Measurements
Postimplementation, all UAs were reviewed and grouped as follows: (1) positive UA with subsequent UC; (2) negative UA, culture cancelled; (3) only UA ordered (no culture); or (4) do not cancel UC ordered. Of the UAs that were analyzed, the following data were collected: demographics, comorbidities, concurrent medications for benign prostatic hyperplasia (BPH) and/or overactive bladder (OAB), documented allergies/adverse drug reactions to antibiotics, date of ED visit, documented UTI signs/symptoms (defined as frequency, urgency, dysuria, fever, suprapubic pain, or altered mental status in patients unable to verbalize urinary symptoms), UC results and susceptibilities, number of UCs repeated within 7 days after initial UA, requirement of antibiotic for UTI within 7 days of initial UA, antibiotic prescribed, duration of antibiotic therapy, and outpatient visits, ED visits, or need for hospital admission within 7 days of the initial UA for UTI-related symptoms. Other relevant UA and UC data that could not be obtained from the EHR were collected by generating a report using the Veterans Information Systems and Technology Architecture (VistA).
Analysis
Statistical analysis was performed using SAS v9.4. Independent t tests and Fisher exact tests were used to describe difference pre- and postintervention. Statistical significance was considered for P < .05. Based on results from the previous study conducted at this facility in addition to a literature review, it was determined that 92 patients in each group (pre- and postintervention) would be necessary to detect a 15% increase in percentage of patients appropriately treated for a UTI.
Results
There were 684 UAs evaluated from ED visits, 429 preintervention and 255 postintervention. The 255 patients were evaluated for the secondary objective of the study. Of the 255 patients with UAs identified postintervention, 150 were excluded based on the predefined exclusion criteria, and the remaining 105 were compared with the 173 patients from the preintervention group and were included in the analysis for the primary objective (Figure 1).
Patients in the postintervention group were younger than those in the preintervention group (P < .02): otherwise the groups were similar (Table 1). Inappropriate antibiotics for ASB decreased from 10.2% preintervention to 1.9% postintervention (odds ratio, 0.17; P = .01) (Table 2). UC processing despite a negative UA significantly decreased from 100% preintervention to 38.6% postintervention (P < .001) (Table 3). In patients with a negative UA, antibiotic prescribing decreased by 25.3% postintervention, but this difference was not statistically significant.
Postintervention, of 255 UAs evaluated, 95 (37.3%) were positive with a processed UC and 95 (37.3%) were negative with UC cancelled, 43 (16.9%) were ordered as DNC, and 22 (8.6%) were ordered without a UC (Figure 2). Twenty-eight of the 95 (29.5%) UAs with processed UCs did not meet the criteria for a positive UA and were not designated as DNC. When the UCs of this subgroup of patients were further analyzed, we found that 2 of the cultures were positive of which 1 patient was symptomatic and required antibiotic therapy.
Of the 95 patients with a negative UA, 69 (72.6%) presented without any UTI-related symptoms. In this group, there were no reports of outpatient visits, ED visits, or hospital admissions within 7 days of initial UA for UTI-related symptoms. None of the UCs ordered as DNC had a supporting reason identified. Nonetheless, the UC results from this patient subgroup also were analyzed further and resulted in 4 patients with negative UA and positive subsequent UC, 1 was symptomatic and required antibiotic therapy.
Discussion
A simple process change at the Hines VA resulted in benefits related to antimicrobial stewardship without conferring adverse outcomes on patient safety. Both UC processing despite a negative UA and inappropriate antibiotic prescribing for ASB were reduced significantly postintervention. This process change was piloted in the ED where UCs are often included as part of the initial diagnostic testing in patients who may not report UTI-related symptoms but for whom a UC is often bundled with other infectious workup, depending on the patient presentation.
Reflex testing of urine specimens has been described in the literature, both in an exploratory nature where impact of a reflex UC cancellation protocol based on certain UA criteria is measured by percent reduction of UCs processed as well as results of such interventions implemented into clinical practice.11-13 A retrospective study performed at the University of North Carolina Medical Center evaluated patients who presented to the ED during a 6-month period and had both an automated UA and UC collected. UC processing was restricted to UA that was positive for nitrites, leukocyte esterase, bacteria, or > 10 WBC/HPF. Use of this reflex culture cancellation protocol could have eliminated 604 of the 1546 (39.1%) cultures processed. However, 11 of the 314 (3.5%) positive cultures could have been missed.13 This same protocol was externally validated at another large academic ED setting, where similar results were found.14
In clinical practice, there is a natural tendency to reflexively prescribe antibiotics based on the results of a positive UC due to the hesitancy in ignoring these results, despite lack of a suspicion for a true infection. Leis and colleagues explored this in a proof-of-concept study evaluating the impact of discontinuing the routine reporting of positive UC results from noncatheterized inpatients and requesting clinicians to call the laboratory for results if a UTI was suspected.16 This intervention resulted in a statistically significant reduction in treatment of ASB in noncatheterized patients from 48 to 12% pre- and postintervention. Clinicians requested culture results only 14% of the time, and there were no adverse outcomes among untreated noncatheterized patients. More recently, a QI study conducted at a large community hospital in Toronto, Ontario, Canada, implemented a 2-step model of care for urine collection.17 UC was collected but only processed by the microbiology laboratory if the ED physicians deemed it necessary after clinical assessment.
After implementation, there was a decrease in the proportion of ED visits associated with processed UC (from 6.0% to 4.7% of visits per week; P < .001), ED visits associated with callbacks for processing UC (1.8% to 1.1% of visits per month; P < .001), and antimicrobial prescriptions for urinary symptoms among hospitalized patients (from 20.6% to 10.9%; P < .001). Equally important, despite the 937 cases in which urine was collected but cultures were not processed, no evidence of untreated UTIs was identified.17
The results from the present study similarly demonstrate minimal concern for potentially undertreating these patients. As seen in the subgroup of patients included in the positive UA group, which did not meet criteria for positive UA per protocol (n = 29), only 2 of the subsequent cultures were positive, of which only 1 patient required antibiotic therapy based on the clinical presentation. In addition, in the group of negative UAs with subsequent cancellation of the UC, there were no found reports of outpatient visits, ED visits, or hospital admissions within 7 days of the initial UA for UTI-related symptoms.
Limitations
This single-center, pre-post QI study was not without limitations. Manual chart reviews were required, and accuracy of information was dependent on clinician documentation and assessment of UTI-related symptoms. The population studied was predominately older males; thus, results may not be applicable to females or young adults. Additionally, recognition of a negative UA and subsequent cancellation of the UC was dependent on laboratory personnel. As noted in the patient group with a positive UA, some of these UAs were negative and may have been overlooked; therefore, subsequent UCs were inappropriately processed. However, this occurred infrequently and confirmed the low probability of true UTI in the setting of a negative UA. Follow-up for UTI-related symptoms may not have been captured if a patient had presented to an outside facility. Last, definitions of a positive UA differed slightly between the pre- and postintervention groups. The preintervention study defined a positive UA as a WBC count > 5 WBC/HPF and positive leukocyte esterase, whereas the present study defined a positive UA with a WBC count > 5. This may have resulted in an overestimation of positive UA in the postintervention group.
Conclusions
Better selective use of UC testing may improve stewardship resources and reduce costs impacting both ED and clinical laboratories. Furthermore, benefits can include a reduction in the use of time and resources required to collect samples for culture, use of test supplies, the time and effort required to process the large number of negative cultures, and resources devoted to the follow-up of these ED culture results. The described UA to reflex culture process change demonstrated a significant reduction in the processing of inappropriate UC and unnecessary antibiotics for ASB. There were no missed UTIs or other adverse patient outcomes noted. This process change has been implemented in all departments at the Hines VA and additional data will be collected to ensure consistent outcomes.
Automated urine cultures (UCs) following urinalysis (UA) are often used in emergency departments (EDs) to identify urinary tract infections (UTIs). The fast-paced environment of the ED makes this method of proactive collection and facilitation of UC favorable. However, results are often reported as no organism growth or the growth of clinically insignificant organisms, leading to the overdetection and overtreatment of asymptomatic bacteriuria (ASB).1-3 An estimated 30 to 60% of patients with ASB receive unwarranted antibiotic treatment, which is associated with an increased risk of developing Clostridioides difficile infection and contributes to the development of antimicrobial resistance.4-10 The costs associated with UC are an important consideration given the use of resources, the time and effort required to collect and process large numbers of negative cultures, and further efforts devoted to the follow-up of ED culture results.
Changes in traditional testing involving testing of both a UA and UC to reflex testing where urine specimens undergo culture only if they meet certain criteria have been described.11-14 This change in traditional testing aims to reduce the number of potentially unnecessary cultures performed without compromising clinical care. Leukocyte quantity in the UA has been shown to be a reliable predictor of true infection.11,15 Fok and colleagues demonstrated that reflex urine testing in ambulatory male urology patients in which cultures were done on only urine specimens with > 5 white blood cells per high-power field (WBC/HPF) would have missed only 7% of positive UCs, while avoiding 69% of cultures.11
At the Edward Hines, Jr Veterans Affairs Hospital (Hines VA), inappropriate UC ordering and treatment for ASB has been identified as an area needing improvement. An evaluation was conducted at the facility to determine the population of inpatient veterans with a positive UC who were appropriately managed. Of the 113 study patients with a positive UC included in this review, 77 (68%) had a diagnosis of ASB, with > 80% of patients with ASB (and no other suspected infections) receiving antimicrobial therapy.8 A subsequent evaluation was conducted at the Hines VA ED to evaluate UTI treatment and follow-up. Of the 173 ED patients included, 23% received antibiotic therapy for an ASB and 60% had a UA and UC collected but did not report symptoms.9 Finally, a review by the Hines VA laboratory showed that in May 2017, of 359 UCs sent from various locations of the hospital, 38% were obtained in the setting of a negative UA.
A multidisciplinary group with representation from primary care, infectious diseases, pharmacy, nursing, laboratory, and informatics was created with a goal to improve the workup and management of UTIs. In addition to periodic education for the clinicians regarding appropriate use and interpretation of UA and UC along with judicious use of antimicrobials especially in the setting of ASB, a UA to reflex culture process change was implemented. This allowed for automatic cancellation of a UC in the setting of a negative UA, which was designed to help facilitate appropriate UC ordering.
Methods
The primary objective of this study was to compare the frequency of inappropriate UC use and inappropriate antibiotic prescribing pre- and postimplementation of this UA to reflex culture process change. An inappropriate UC was defined as a UC ordered despite a negative UA in asymptomatic patients. Inappropriate antibiotic prescribing was defined as treatment of patients with ASB. The secondary objective evaluated postintervention data to assess the frequency of outpatient, ED, and hospital visits for UTI-related symptoms in the group of patients that had a UC cancelled as a result of the new process change (within a 7-day period of the initial UA) to determine whether patients with true infections were missed due to the process change.
Study Design and Setting
This pre-post quality improvement (QI) study analyzed the UC-ordering practices for UTIs sent from the ED at the Hines VA. This VA is a 483-bed tertiary care hospital in Chicago, Illinois, and serves > 57,000 veterans and about 23,000 ED visits annually. This study was approved by the Edward Hines, Jr VA Institutional Review Board as a quality assurance/QI proposal prior to data collection.
Patient Selection
All patients who
When comparing postintervention data with preintervention data for the primary study objective, the same exclusion criteria from the 2015 study were applied to the present study, which excluded ED patients who were admitted for inpatient care, concurrent antibiotic therapy for a non-UTI indication, duplicate cultures, and use of chronic bladder management devices. All patients identified as receiving a UA during the specified postintervention study period were included for evaluation of the secondary study objective.
Interventions
After physician education, an ED process change was implemented on October 3, 2017. This process change involved the creation of new order sets in the EHR that allowed clinicians to order a UA only, a UA with culture that would be cancelled by laboratory personnel if the UA did not result in > 5 WBC/HPF, and a UA with culture designated as do not cancel, where the UC was processed regardless of the UA results. The scenarios in which the latter option was considered appropriate were listed on the ordering screen and included pregnancy, a genitourinary procedure with necessary preoperative culture, and neutropenia.
Measurements
Postimplementation, all UAs were reviewed and grouped as follows: (1) positive UA with subsequent UC; (2) negative UA, culture cancelled; (3) only UA ordered (no culture); or (4) do not cancel UC ordered. Of the UAs that were analyzed, the following data were collected: demographics, comorbidities, concurrent medications for benign prostatic hyperplasia (BPH) and/or overactive bladder (OAB), documented allergies/adverse drug reactions to antibiotics, date of ED visit, documented UTI signs/symptoms (defined as frequency, urgency, dysuria, fever, suprapubic pain, or altered mental status in patients unable to verbalize urinary symptoms), UC results and susceptibilities, number of UCs repeated within 7 days after initial UA, requirement of antibiotic for UTI within 7 days of initial UA, antibiotic prescribed, duration of antibiotic therapy, and outpatient visits, ED visits, or need for hospital admission within 7 days of the initial UA for UTI-related symptoms. Other relevant UA and UC data that could not be obtained from the EHR were collected by generating a report using the Veterans Information Systems and Technology Architecture (VistA).
Analysis
Statistical analysis was performed using SAS v9.4. Independent t tests and Fisher exact tests were used to describe difference pre- and postintervention. Statistical significance was considered for P < .05. Based on results from the previous study conducted at this facility in addition to a literature review, it was determined that 92 patients in each group (pre- and postintervention) would be necessary to detect a 15% increase in percentage of patients appropriately treated for a UTI.
Results
There were 684 UAs evaluated from ED visits, 429 preintervention and 255 postintervention. The 255 patients were evaluated for the secondary objective of the study. Of the 255 patients with UAs identified postintervention, 150 were excluded based on the predefined exclusion criteria, and the remaining 105 were compared with the 173 patients from the preintervention group and were included in the analysis for the primary objective (Figure 1).
Patients in the postintervention group were younger than those in the preintervention group (P < .02): otherwise the groups were similar (Table 1). Inappropriate antibiotics for ASB decreased from 10.2% preintervention to 1.9% postintervention (odds ratio, 0.17; P = .01) (Table 2). UC processing despite a negative UA significantly decreased from 100% preintervention to 38.6% postintervention (P < .001) (Table 3). In patients with a negative UA, antibiotic prescribing decreased by 25.3% postintervention, but this difference was not statistically significant.
Postintervention, of 255 UAs evaluated, 95 (37.3%) were positive with a processed UC and 95 (37.3%) were negative with UC cancelled, 43 (16.9%) were ordered as DNC, and 22 (8.6%) were ordered without a UC (Figure 2). Twenty-eight of the 95 (29.5%) UAs with processed UCs did not meet the criteria for a positive UA and were not designated as DNC. When the UCs of this subgroup of patients were further analyzed, we found that 2 of the cultures were positive of which 1 patient was symptomatic and required antibiotic therapy.
Of the 95 patients with a negative UA, 69 (72.6%) presented without any UTI-related symptoms. In this group, there were no reports of outpatient visits, ED visits, or hospital admissions within 7 days of initial UA for UTI-related symptoms. None of the UCs ordered as DNC had a supporting reason identified. Nonetheless, the UC results from this patient subgroup also were analyzed further and resulted in 4 patients with negative UA and positive subsequent UC, 1 was symptomatic and required antibiotic therapy.
Discussion
A simple process change at the Hines VA resulted in benefits related to antimicrobial stewardship without conferring adverse outcomes on patient safety. Both UC processing despite a negative UA and inappropriate antibiotic prescribing for ASB were reduced significantly postintervention. This process change was piloted in the ED where UCs are often included as part of the initial diagnostic testing in patients who may not report UTI-related symptoms but for whom a UC is often bundled with other infectious workup, depending on the patient presentation.
Reflex testing of urine specimens has been described in the literature, both in an exploratory nature where impact of a reflex UC cancellation protocol based on certain UA criteria is measured by percent reduction of UCs processed as well as results of such interventions implemented into clinical practice.11-13 A retrospective study performed at the University of North Carolina Medical Center evaluated patients who presented to the ED during a 6-month period and had both an automated UA and UC collected. UC processing was restricted to UA that was positive for nitrites, leukocyte esterase, bacteria, or > 10 WBC/HPF. Use of this reflex culture cancellation protocol could have eliminated 604 of the 1546 (39.1%) cultures processed. However, 11 of the 314 (3.5%) positive cultures could have been missed.13 This same protocol was externally validated at another large academic ED setting, where similar results were found.14
In clinical practice, there is a natural tendency to reflexively prescribe antibiotics based on the results of a positive UC due to the hesitancy in ignoring these results, despite lack of a suspicion for a true infection. Leis and colleagues explored this in a proof-of-concept study evaluating the impact of discontinuing the routine reporting of positive UC results from noncatheterized inpatients and requesting clinicians to call the laboratory for results if a UTI was suspected.16 This intervention resulted in a statistically significant reduction in treatment of ASB in noncatheterized patients from 48 to 12% pre- and postintervention. Clinicians requested culture results only 14% of the time, and there were no adverse outcomes among untreated noncatheterized patients. More recently, a QI study conducted at a large community hospital in Toronto, Ontario, Canada, implemented a 2-step model of care for urine collection.17 UC was collected but only processed by the microbiology laboratory if the ED physicians deemed it necessary after clinical assessment.
After implementation, there was a decrease in the proportion of ED visits associated with processed UC (from 6.0% to 4.7% of visits per week; P < .001), ED visits associated with callbacks for processing UC (1.8% to 1.1% of visits per month; P < .001), and antimicrobial prescriptions for urinary symptoms among hospitalized patients (from 20.6% to 10.9%; P < .001). Equally important, despite the 937 cases in which urine was collected but cultures were not processed, no evidence of untreated UTIs was identified.17
The results from the present study similarly demonstrate minimal concern for potentially undertreating these patients. As seen in the subgroup of patients included in the positive UA group, which did not meet criteria for positive UA per protocol (n = 29), only 2 of the subsequent cultures were positive, of which only 1 patient required antibiotic therapy based on the clinical presentation. In addition, in the group of negative UAs with subsequent cancellation of the UC, there were no found reports of outpatient visits, ED visits, or hospital admissions within 7 days of the initial UA for UTI-related symptoms.
Limitations
This single-center, pre-post QI study was not without limitations. Manual chart reviews were required, and accuracy of information was dependent on clinician documentation and assessment of UTI-related symptoms. The population studied was predominately older males; thus, results may not be applicable to females or young adults. Additionally, recognition of a negative UA and subsequent cancellation of the UC was dependent on laboratory personnel. As noted in the patient group with a positive UA, some of these UAs were negative and may have been overlooked; therefore, subsequent UCs were inappropriately processed. However, this occurred infrequently and confirmed the low probability of true UTI in the setting of a negative UA. Follow-up for UTI-related symptoms may not have been captured if a patient had presented to an outside facility. Last, definitions of a positive UA differed slightly between the pre- and postintervention groups. The preintervention study defined a positive UA as a WBC count > 5 WBC/HPF and positive leukocyte esterase, whereas the present study defined a positive UA with a WBC count > 5. This may have resulted in an overestimation of positive UA in the postintervention group.
Conclusions
Better selective use of UC testing may improve stewardship resources and reduce costs impacting both ED and clinical laboratories. Furthermore, benefits can include a reduction in the use of time and resources required to collect samples for culture, use of test supplies, the time and effort required to process the large number of negative cultures, and resources devoted to the follow-up of these ED culture results. The described UA to reflex culture process change demonstrated a significant reduction in the processing of inappropriate UC and unnecessary antibiotics for ASB. There were no missed UTIs or other adverse patient outcomes noted. This process change has been implemented in all departments at the Hines VA and additional data will be collected to ensure consistent outcomes.
1. Chironda B, Clancy S, Powis JE. Optimizing urine culture collection in the emergency department using frontline ownership interventions. Clin Infect Dis. 2014;59(7):1038-1039. doi:10.1093/cid/ciu412
2. Nagurney JT, Brown DF, Chang Y, Sane S, Wang AC, Weiner JB. Use of diagnostic testing in the emergency department for patients presenting with non-traumatic abdominal pain. J Emerg Med. 2003;25(4):363-371. doi:10.1016/s0736-4679(03)00237-3
3. Lammers RL, Gibson S, Kovacs D, Sears W, Strachan G. Comparison of test characteristics of urine dipstick and urinalysis at various test cutoff points. Ann Emerg Med. 2001;38(5):505-512. doi:10.1067/mem.2001.119427
4. Nicolle LE, Gupta K, Bradley SF, et al. Clinical practice guideline for the management of asymptomatic bacteriuria: 2019 update by the Infectious Diseases Society of America. Clin Infect Dis. 2019;68(10):1611-1615. doi:10.1093/cid/ciy1121
5. Trautner BW, Grigoryan L, Petersen NJ, et al. Effectiveness of an antimicrobial stewardship approach for urinary catheter-associated asymptomatic bacteriuria. JAMA Intern Med. 2015;175(7):1120-1127. doi:10.1001/jamainternmed.2015.1878
6. Hartley S, Valley S, Kuhn L, et al. Overtreatment of asymptomatic bacteriuria: identifying targets for improvement. Infect Control Hosp Epidemiol. 2015;36(4):470-473. doi:10.1017/ice.2014.73
7. Bader MS, Loeb M, Brooks AA. An update on the management of urinary tract infections in the era of antimicrobial resistance. Postgrad Med. 2017;129(2):242-258. doi:10.1080/00325481.2017.1246055
8. Spivak ES, Burk M, Zhang R, et al. Management of bacteriuria in Veterans Affairs hospitals. Clin Infect Dis. 2017;65(6):910-917. doi:10.1093/cid/cix474
9. Kim EY, Patel U, Patel B, Suda KJ. Evaluation of bacteriuria treatment and follow-up initiated in the emergency department at a Veterans Affairs hospital. J Pharm Technol. 2017;33(5):183-188. doi:10.1177/8755122517718214
10. Brown E, Talbot GH, Axelrod P, Provencher M, Hoegg C. Risk factors for Clostridium difficile toxin-associated diarrhea. Infect Control Hosp Epidemiol. 1990;11(6):283-290. doi:10.1086/646173
11. Fok C, Fitzgerald MP, Turk T, Mueller E, Dalaza L, Schreckenberger P. Reflex testing of male urine specimens misses few positive cultures may reduce unnecessary testing of normal specimens. Urology. 2010;75(1):74-76. doi:10.1016/j.urology.2009.08.071
12. Munigala S, Jackups RR Jr, Poirier RF, et al. Impact of order set design on urine culturing practices at an academic medical centre emergency department. BMJ Qual Saf. 2018;27(8):587-592. doi:10.1136/bmjqs-2017-006899
13. Jones CW, Culbreath KD, Mehrotra A, Gilligan PH. Reflect urine culture cancellation in the emergency department. J Emerg Med. 2014;46(1):71-76. doi:10.1016/j.jemermed.2013.08.042
14. Hertz JT, Lescallette RD, Barrett TW, Ward MJ, Self WH. External validation of an ED protocol for reflex urine culture cancelation. Am J Emerg Med. 2015;33(12):1838-1839. doi:10.1016/j.ajem.2015.09.026
15. Stamm WE. Measurement of pyuria and its relation to bacteriuria. Am J Med. 1983;75(1B):53-58. doi:10.1016/0002-9343(83)90073-6
16. Leis JA, Rebick GW, Daneman N, et al. Reducing antimicrobial therapy for asymptomatic bacteriuria among noncatheterized inpatients: a proof-of-concept study. Clin Infect Dis. 2014;58(7):980-983. doi:10.1093/cid/ciu010
17. Stagg A, Lutz H, Kirpalaney S, et al. Impact of two-step urine culture ordering in the emergency department: a time series analysis. BMJ Qual Saf. 2017;27:140-147. doi:10.1136/bmjqs-2016-006250
1. Chironda B, Clancy S, Powis JE. Optimizing urine culture collection in the emergency department using frontline ownership interventions. Clin Infect Dis. 2014;59(7):1038-1039. doi:10.1093/cid/ciu412
2. Nagurney JT, Brown DF, Chang Y, Sane S, Wang AC, Weiner JB. Use of diagnostic testing in the emergency department for patients presenting with non-traumatic abdominal pain. J Emerg Med. 2003;25(4):363-371. doi:10.1016/s0736-4679(03)00237-3
3. Lammers RL, Gibson S, Kovacs D, Sears W, Strachan G. Comparison of test characteristics of urine dipstick and urinalysis at various test cutoff points. Ann Emerg Med. 2001;38(5):505-512. doi:10.1067/mem.2001.119427
4. Nicolle LE, Gupta K, Bradley SF, et al. Clinical practice guideline for the management of asymptomatic bacteriuria: 2019 update by the Infectious Diseases Society of America. Clin Infect Dis. 2019;68(10):1611-1615. doi:10.1093/cid/ciy1121
5. Trautner BW, Grigoryan L, Petersen NJ, et al. Effectiveness of an antimicrobial stewardship approach for urinary catheter-associated asymptomatic bacteriuria. JAMA Intern Med. 2015;175(7):1120-1127. doi:10.1001/jamainternmed.2015.1878
6. Hartley S, Valley S, Kuhn L, et al. Overtreatment of asymptomatic bacteriuria: identifying targets for improvement. Infect Control Hosp Epidemiol. 2015;36(4):470-473. doi:10.1017/ice.2014.73
7. Bader MS, Loeb M, Brooks AA. An update on the management of urinary tract infections in the era of antimicrobial resistance. Postgrad Med. 2017;129(2):242-258. doi:10.1080/00325481.2017.1246055
8. Spivak ES, Burk M, Zhang R, et al. Management of bacteriuria in Veterans Affairs hospitals. Clin Infect Dis. 2017;65(6):910-917. doi:10.1093/cid/cix474
9. Kim EY, Patel U, Patel B, Suda KJ. Evaluation of bacteriuria treatment and follow-up initiated in the emergency department at a Veterans Affairs hospital. J Pharm Technol. 2017;33(5):183-188. doi:10.1177/8755122517718214
10. Brown E, Talbot GH, Axelrod P, Provencher M, Hoegg C. Risk factors for Clostridium difficile toxin-associated diarrhea. Infect Control Hosp Epidemiol. 1990;11(6):283-290. doi:10.1086/646173
11. Fok C, Fitzgerald MP, Turk T, Mueller E, Dalaza L, Schreckenberger P. Reflex testing of male urine specimens misses few positive cultures may reduce unnecessary testing of normal specimens. Urology. 2010;75(1):74-76. doi:10.1016/j.urology.2009.08.071
12. Munigala S, Jackups RR Jr, Poirier RF, et al. Impact of order set design on urine culturing practices at an academic medical centre emergency department. BMJ Qual Saf. 2018;27(8):587-592. doi:10.1136/bmjqs-2017-006899
13. Jones CW, Culbreath KD, Mehrotra A, Gilligan PH. Reflect urine culture cancellation in the emergency department. J Emerg Med. 2014;46(1):71-76. doi:10.1016/j.jemermed.2013.08.042
14. Hertz JT, Lescallette RD, Barrett TW, Ward MJ, Self WH. External validation of an ED protocol for reflex urine culture cancelation. Am J Emerg Med. 2015;33(12):1838-1839. doi:10.1016/j.ajem.2015.09.026
15. Stamm WE. Measurement of pyuria and its relation to bacteriuria. Am J Med. 1983;75(1B):53-58. doi:10.1016/0002-9343(83)90073-6
16. Leis JA, Rebick GW, Daneman N, et al. Reducing antimicrobial therapy for asymptomatic bacteriuria among noncatheterized inpatients: a proof-of-concept study. Clin Infect Dis. 2014;58(7):980-983. doi:10.1093/cid/ciu010
17. Stagg A, Lutz H, Kirpalaney S, et al. Impact of two-step urine culture ordering in the emergency department: a time series analysis. BMJ Qual Saf. 2017;27:140-147. doi:10.1136/bmjqs-2016-006250
Adjunctive Use of Halobetasol Propionate–Tazarotene in Biologic-Experienced Patients With Psoriasis
Psoriasis is a common chronic immunologic skin disease that affects approximately 7.4 million adults in the United States1 and more than 100 million individuals worldwide.2 Patients with psoriasis have a potentially heightened risk for cardiometabolic diseases, psychiatric disorders, and psoriatic arthritis,3 as well as impaired quality of life (QOL).4 Psoriasis also is associated with increased health care costs5 and may result in substantial socioeconomic repercussions for affected patients.6,7
Psoriasis treatments focus on relieving symptoms and improving patient QOL. Systemic therapy has been the mainstay of treatment for moderate to severe psoriasis.8 Although topical therapy usually is applied to treat mild symptoms, it also can be used as an adjunct to enhance efficacy of other treatment approaches.9 The National Psoriasis Foundation (NPF) recommends a treat-to-target (TTT) strategy for plaque psoriasis, the most common form of psoriasis, with a target response of attaining affected body surface area (BSA) of 1% or lower at 3 months after treatment initiation, allowing for regular assessments of treatment responses.10
Not all patients with moderate to severe psoriasis can achieve a satisfactory response with systemic biologic monotherapy.11 Switching to a new biologic improves responses in some but not all cases12 and could be associated with new safety issues and additional costs. Combinations of biologics with phototherapy, nonbiologic systemic agents, or topical medications were found to be more effective than biologics alone,9,11 though long-term safety studies are needed for biologics combined with other systemic inverventions.11
A lotion containing a fixed combination of halobetasol propionate (HP) 0.01%, a corticosteroid, and tazarotene (TAZ) 0.045%, a retinoid, is indicated for plaque psoriasis in adults.13 Two randomized, controlled, phase 3 trials demonstrated the rapid and sustained efficacy of HP-TAZ in treating moderate to severe plaque psoriasis without any safety concerns.14,15 However, combining HP-TAZ lotion with biologics has not been examined yet, to our knowledge.
This open-label study evaluated the effectiveness and safety of adjunctive HP-TAZ lotion in adult patients with moderate to severe plaque psoriasis who were being treated with biologics in a real-world setting. Potential cost savings with the addition of topical HP-TAZ to ongoing biologics vs switching to a new biologic also were assessed.
Methods
Study Design and Participants—A single-center, institutional review board–approved, open-label study evaluated adjunctive therapy with HP 0.01%–TAZ 0.045% lotion in patients with psoriasis being treated with biologic agents. The study was conducted in accordance with the ethical principles of the Declaration of Helsinki and in compliance with Good Clinical Practices. All patients provided written informed consent before enrollment.
Male and nonpregnant female patients (aged ≥18 years)with moderate to severe chronic plaque psoriasis and a BSA of 2% to 10% who were being treated with biologics for at least 24 weeks at baseline were enrolled. Patients were excluded if they had used oral systemic medications for psoriasis (≤4 weeks), other topical antipsoriatic therapies (≤14 days), UVB phototherapy (≤2 weeks), and psoralen plus UVA phototherapy (≤4 weeks) prior to study initiation. Concomitant use of steroid-free topical emollients or low-potency topical steroids and appropriate interventions deemed necessary by the investigator were allowed.
Although participants maintained their prescribed biologics for the duration of the study, HP-TAZ lotion also was applied once daily for 8 weeks, followed by once every other day for an additional 4 weeks. Participants then continued with biologics only for the last 4 weeks of the study.
Study Outcome Measures—Disease severity and treatment efficacy were assessed by affected BSA, Physician Global Assessment (PGA) score, composite BSA×PGA score, and participant-reported Dermatology Life Quality Index (DLQI). The primary end point was the proportion of participants achieving a BSA of 0% to 1% (NPF TTT status) at week 8. Secondary end points included the proportions of participants with BSA of 0% to 1% at weeks 12 and 16; BSA×PGA score at weeks 8, 12, and 16; and improvements in BSA, PGA, and DLQI at weeks 8, 12, and 16.
Adverse events (AEs) that occurred after the signing of the informed consent and for the duration of the participant’s participation were recorded, regardless of causality. Physical examinations were performed at screening; baseline; and weeks 8, 12, and 16 to document any clinically significant abnormalities. Localized skin reactions were assessed for tolerability of the study drug, with any reaction requiring concomitant therapy recorded as an AE.
The likelihood of switching to a new biologic regimen was assessed by the investigator for each participant at baseline and weeks 8, 12, and 16. Participants with unacceptable responses to their treatments (BSA >3%) were reported as likely to be considered for switching biologics by the investigator.
Pharmacoeconomic Evaluation—Potential cost savings were evaluated for the addition of HP-TAZ lotion to ongoing biologics vs switching to a new biologic. Cost comparisons were made in participants for whom the investigator would likely have switched biologics at baseline but not at the end of the study. For maintaining the same biologic with adjunctive topical HP-TAZ, total cost was estimated by adding the cost for 12 weeks (once daily for 8 weeks and once every other day for 4 weeks) of the HP-TAZ lotion to that of 16-week maintenance dosing with the biologic. The projected cost for switching to a new biologic for 16 weeks of treatment was based on both induction and maintenance dosing as recommended in its product label. Prices were obtained from the 2020 average wholesale price specialty pharmacy reports (BioPlus Specialty Pharmacy Services [https://www.bioplusrx.com]).
Data Handling—Enrollment of approximately 25 participants was desired for the study. Data on disease severity and participant-reported outcomes were assessed using descriptive statistics. Adverse events were summarized descriptively by incidence, severity, and relationship to the study drug. All participants with data available at a measured time point were included in the analyses for that time point.
Results
Participant Disposition and Demographics—Twenty-five participants (15 male and 10 female) were included in the study (Table 1). Seven participants discontinued the study for the following reasons: AEs (n=4), patient choice (n=2), and noncompliance (n=1).

The average age of the participants was 50 years, the majority were White (76.0% [19/25]) andnon-Hispanic (88.0% [22/25]), and the mean duration of chronic plaque psoriasis was 18.9 years (Table 1). Participants had been receiving biologic monotherapy for at least 24 weeks prior to enrollment, most commonly ustekinumab (32.0% [8/25])(Table 1). None had achieved the NPF TTT status with their biologics. At baseline, mean (SD) affected BSA, PGA, BSA×PGA, and participant-reported DLQI were 4.16% (2.04%), 2.84 (0.55), 11.88 (6.39), and 4.00 (4.74), respectively.
Efficacy Assessment—Application of HP-TAZ lotion in addition to the participants’ existing biologic therapy reduced severity of the disease, as evidenced by the reductions in mean BSA, PGA, and BSA×PGA. After 8 weeks of once-daily concomitant HP-TAZ use with biologic, mean BSA and PGA dropped by approximately 40% and 37%, respectively (Figures 1A and 1B). A greater reduction (54%) was found for mean BSA×PGA (Figure 1C). Disease severity continued to improve when the application schedule for HP-TAZ was changed to once every other day for 4 weeks, as mean BSA, PGA, and BSA×PGA decreased further at week 12. These beneficial effects were sustained during the last 4 weeks of the study after HP-TAZ was discontinued, with reductions of 57%, 43%, and 70% from baseline for mean BSA, PGA, and BSA×PGA, respectively (Figure 1).
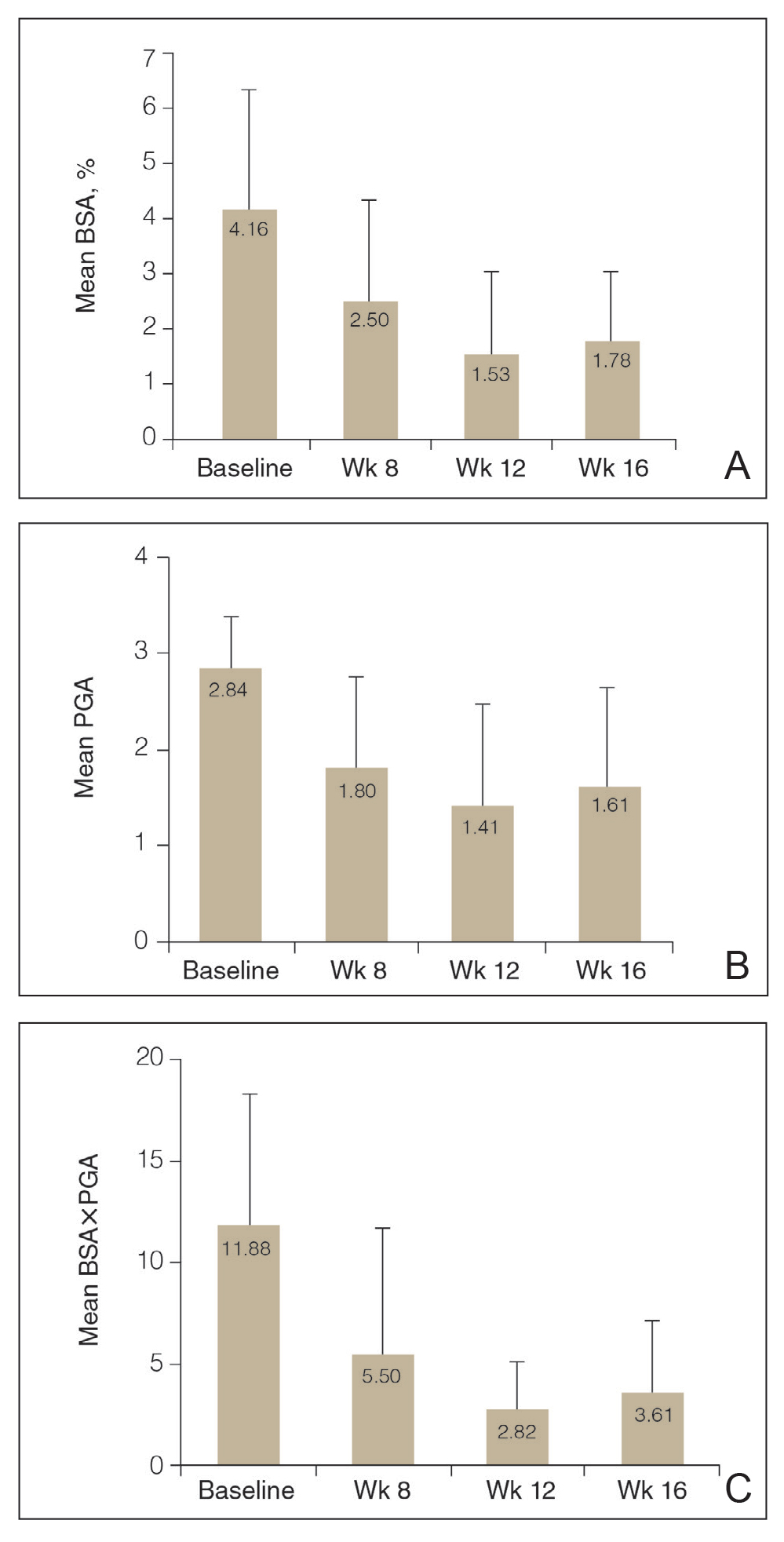
The proportion of participants who achieved NPF TTT status increased from 0% at baseline to 20.0% (5/20) at week 8 with once-daily use of HP-TAZ plus biologic for 8 weeks (Figure 2). At week 12, more participants (64.7% [11/17]) achieved the treatment goal after application of HP-TAZ once every other day with biologic for 4 weeks. Most participants maintained NPF TTT status after HP-TAZ was discontinued; at week 16, 50.0% (9/18) attained the NPF treatment goal (Figure 2).
![Proportion of participants achieving National Psoriasis Foundation target-to-treat status (body surface area [BSA] ≤1%) at baseline and weeks 8, 12, and 16 Proportion of participants achieving National Psoriasis Foundation target-to-treat status (body surface area [BSA] ≤1%) at baseline and weeks 8, 12, and 16](https://cdn.mdedge.com/files/s3fs-public/Bagel_0222_2.JPG)
The mean DLQI score decreased from 4.00 at baseline to 2.45 after 8 weeks of concomitant use of once-daily HP-TAZ with biologic, reflecting a 39% score reduction. An additional 4 weeks of adjunctive HP-TAZ applied once every other day with biologic further decreased the DLQI score to 2.18 at week 12. Mean DLQI remained similar (2.33) after another 4 weeks of biologics alone. The proportion of participants reporting a DLQI score of 0 to 1 increased from 40% (10/25) at baseline to 60% (12/20) at week 8 and 76.5% (13/17) at week 12 with adjunctive HP-TAZ lotion use with biologic. At week 16, a DLQI score of 0 to 1 was reported in 61.1% (11/18) of participants after receiving only biologics for 4 weeks.
Safety Assessment—A total of 19 AEs were reported in 11 participants during the study; 16 AEs were considered treatment related in 8 participants (Table 2). The most common AEs were retinoid dermatitis (28% [7/25]), burning at the application site (8% [2/25]), and pruritus at the application site (8% [2/25]), all of which were considered related to the treatment. Among all AEs, 12 were mild in severity, and the remaining 7 were moderate. Adverse events led to early study termination in 4 participants, all with retinoid dermatitis as the primary reason.
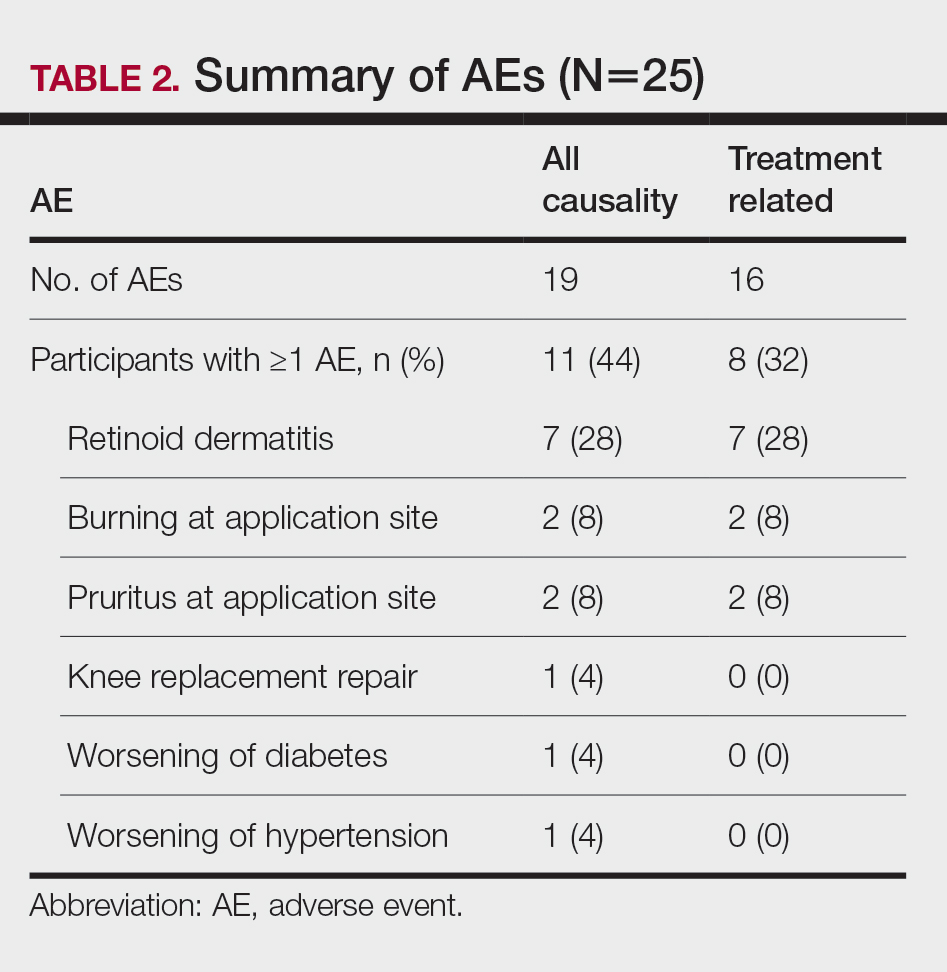
Likelihood of Switching Biologics—At baseline, almost 90% (22/25) of participants were rated as likely to switch biologics by the investigator due to unacceptable responses to their currently prescribed biologics (BSA >3%)(Figure 3). The likelihood was greatly reduced by concomitant HP-TAZ, as the proportion of participants defined as nonresponders to their biologic decreased to 35% (7/20) with 8-week adjunctive application of once-daily HP-TAZ with biologic and further decreased to 23.5% (4/17) with another 4 weeks of adjunctive HP-TAZ applied every other day plus biologic. At week 16, after 4 weeks of biologics alone, the proportion was maintained at 33.3% (6/18).
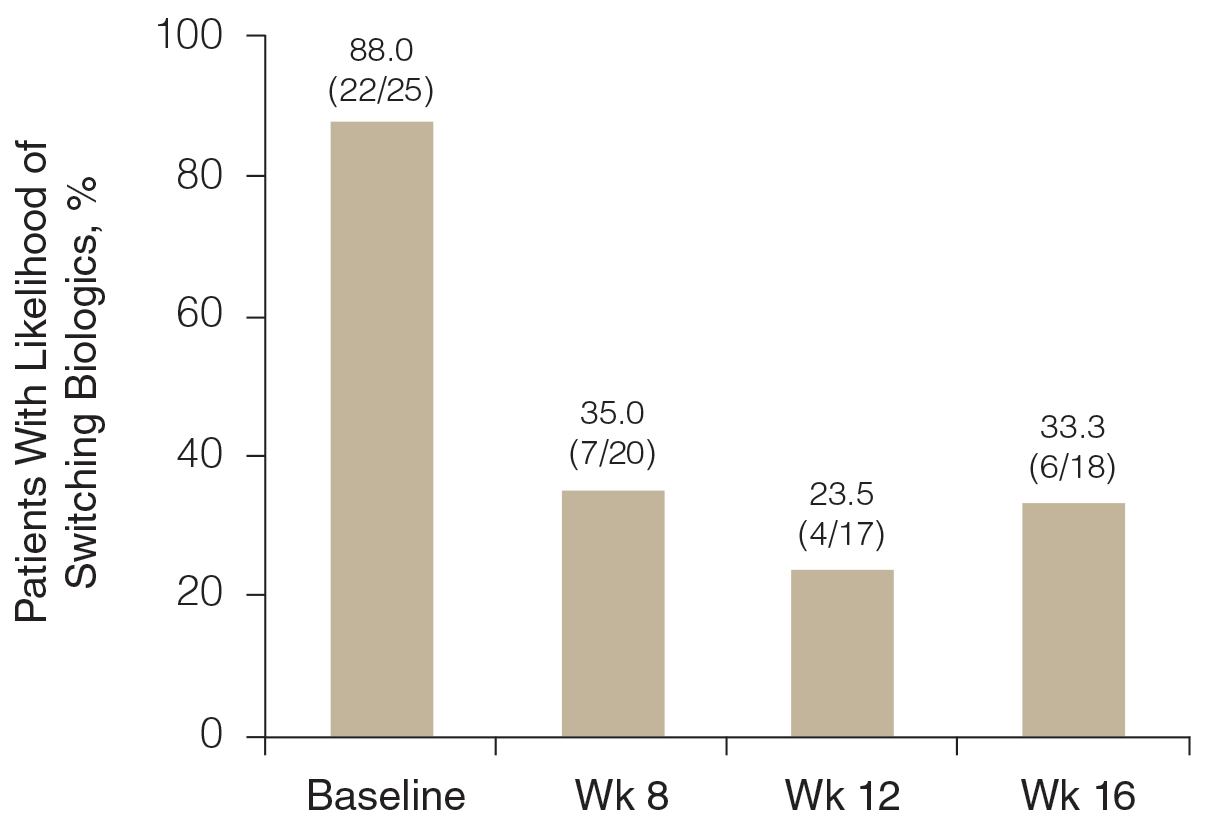
Pharmacoeconomics of Adding Topical HP-TAZ vs Switching Biologics—In the participants whom the investigator reported as likely to switch biologics at baseline, 9 had improvements in disease control such that switching biologics was no longer considered necessary for them at week 16. Potential cost savings with adjunctive therapy of HP-TAZ plus biologic vs switching biologics were therefore evaluated in these 9 participants, who were receiving ustekinumab, adalimumab, guselkumab, ixekizumab, and secukinumab during the study (Table 3). The estimated total cost of 16-week maintenance dosing of biologics plus adjunct HP-TAZ lotion ranged from $14,675 (ustekinumab 45 mg) to $54,025 (secukinumab 300 mg), while switching to other most commonly prescribed biologics for 16 weeks would cost an estimated $33,340 to $106,400 (induction and subsequent maintenance phases)(Table 3). Most biologic plus HP-TAZ combinations were estimated to cost less than $30,000, potentially saving $4816 to $91,725 compared with switching to any of the other 7 biologics (Table 3). The relatively more expensive maintenance combination containing secukinumab plus HP-TAZ ($54,025) appeared to be a less expensive option when compared with switching to ustekinumab (90 mg)($83,097), ixekizumab (80 mg)($61,452), or risankizumab (150 mg)($57,030) as an alternative biologic.
Comment
Adjunctive Use of HP-TAZ Lotion—In the present study, we showed that adjunctive HP-TAZ lotion improved biologic treatment response and reduced disease severity in participants with moderate to severe psoriasis whose symptoms could not be adequately controlled by 24 weeks or more of biologic monotherapy in a real-world setting. Disease activity decreased as evidenced by reductions in all assessed effectiveness variables, including BSA involvement, PGA score, composite BSA×PGA score, and participant-reported DLQI score. Half of the participants achieved NPF TTT status at the end of the study. The treatment was well tolerated with no unexpected safety concerns. Compared with switching to a new biologic, adding topical HP-TAZ to ongoing biologics appeared to be a more cost-effective approach to enhance treatment effects. Our results suggest that adjunctive use of HP-TAZ lotion may be a safe, effective, and economical option for patients with psoriasis who are failing their ongoing biologic monotherapy.
Treat-to-Target Status—The NPF-recommended target response to a treatment for plaque psoriasis is BSA of 1% or lower at 3 months postinitiation.10 Patients in the current study had major psoriasis activity at study entry despite being treated with a biologic for at least 24 weeks, as none had attained NPF TTT status at baseline. Because the time period of prior biologic treatment (at least 24 weeks) is much longer than the 3 months suggested by NPF, we believe that we were observing a true failure of the biologic rather than a slow onset of treatment effects in these patients at the time of enrollment. By week 12, with the addition of HP-TAZ lotion to the biologic, a high rate of participants achieved NPF TTT status (64.7%), with most participants being able to maintain this TTT status at study end after 4 weeks of biologic alone. Most participants also reported no impact of psoriasis on their QOL (DLQI, 0–116; 76.5%) at week 12. Improvements we found in disease control with adjunctive HP-TAZ lotion plus biologic support prior reports showing enhanced responses when a topical medication was added to a biologic.17,18 Reductions in psoriasis activity after 8 weeks of combined biologics plus once-daily HP-TAZ also are consistent with 2 phase 3 RCTs in which a monotherapy of HP-TAZ lotion used once daily for 8 weeks reduced BSA and DLQI.15 Notably, in the current study, disease severity continued to decrease when dosing of HP-TAZ was reduced to once every other day for 4 weeks, and the improvements were maintained even after the adjunct topical therapy was discontinued.
Safety Profile of HP-TAZ Lotion—The overall safety profile in our study also was consistent with that previously reported for HP-TAZ lotion,15,19-21 with no new safety signals observed. The combination treatment was well tolerated, with most reported AEs being mild in severity. Adverse events were mostly related to application-site reactions, the most common being dermatitis (28%), which was likely attributable to the TAZ component of the topical regimen.15
Likelihood of Switching Biologics—Reduced disease activity was reflected by a decrease in the percentage of participants the investigator considered likely to change biologics, which was 88.0% at baseline but only 33.3% at the end of the study. Although switching to a different biologic agent can improve treatment effect,22 it could lead to a substantial increase in health care costs and use of resources compared with no switch.5 We found that switching to one of the other most commonly prescribed biologics could incur $4816 to $91,725 in additional costs in most cases when compared with the combination strategy we evaluated over the 16-week treatment period. Therefore, concomitant use of HP-TAZ lotion with the ongoing biologics would be a potentially more economical alternative for patients to achieve acceptable responses or the NPF TTT goal. Moreover, combination with an adjunctive topical medication could avoid potential risks associated with switching, such as new AEs with new biologic regimens or disease flare during any washout period sometimes adopted for switching biologics.
Study Limitations—Our estimated costs were based on average wholesale prices and did not reflect net prices paid by patients or health plans due to the lack of known discount rates.
Conclusion
In this real-world study, patients with psoriasis that failed to respond to biologic monotherapy had improved disease control and QOL and reported no new safety concerns with adjunctive use of HP-TAZ lotion. Adding HP-TAZ to the ongoing biologics could be a more cost-effective option vs switching biologics for patients whose psoriasis symptoms could not be controlled with biologic monotherapy. Taken together, our results support the use of HP-TAZ lotion as an effective and safe adjunctive topical therapy in combination with biologics for psoriasis treatment.
Acknowledgments—We acknowledge the medical writing assistance provided by Hui Zhang, PhD, and Kathleen Ohleth, PhD, from Precise Publications LLC (Far Hills, New Jersey), which was funded by Ortho Dermatologics.
- Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014;70:512-516.
- Global Report on Psoriasis. World Health Organization; 2016. Accessed January 11, 2022. https://apps.who.int/iris/handle/10665/204417
- Takeshita J, Grewal S, Langan SM, et al. Psoriasis and comorbid diseases: epidemiology. J Am Acad Dermatol. 2017;76:377-390.
- Moller AH, Erntoft S, Vinding GR, et al. A systematic literature review to compare quality of life in psoriasis with other chronic diseases using EQ-5D-derived utility values. Patient Relat Outcome Meas. 2015;6:167-177.
- Feldman SR, Tian H, Wang X, et al. Health care utilization and cost associated with biologic treatment patterns among patients with moderate to severe psoriasis: analyses from a large U.S. claims database. J Manag Care Spec Pharm. 2019;25:479-488.
- Thomsen SF, Skov L, Dodge R, et al. Socioeconomic costs and health inequalities from psoriasis: a cohort study. Dermatology. 2019;235:372-379.
- Fowler JF, Duh MS, Rovba L, et al. The impact of psoriasis on health care costs and patient work loss. J Am Acad Dermatol. 2008;59:772-780.
- Menter A, Gottlieb A, Feldman SR, et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: section 1. overview of psoriasis and guidelines of care for the treatment of psoriasis with biologics. J Am Acad Dermatol. 2008;58:826-850.
- Bagel J, Gold LS. Combining topical psoriasis treatment to enhance systemic and phototherapy: a review of the literature. J Drugs Dermatol. 2017;16:1209-1222.
- Armstrong AW, Siegel MP, Bagel J, et al. From the Medical Board of the National Psoriasis Foundation: treatment targets for plaque psoriasis. J Am Acad Dermatol. 2017;76:290-298.
- Armstrong AW, Bagel J, Van Voorhees AS, et al. Combining biologic therapies with other systemic treatments in psoriasis: evidence-based, best-practice recommendations from the Medical Board of the National Psoriasis Foundation. JAMA Dermatol. 2015;151:432-438.
- Menter A, Strober BE, Kaplan DH, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with biologics. J Am Acad Dermatol. 2019;80:1029-1072.
- Duobrii. Prescribing information. Bausch Health Companies Inc; 2019.
- Sugarman JL, Weiss J, Tanghetti EA, et al. Safety and efficacy of a fixed combination halobetasol and tazarotene lotion in the treatment of moderate-to-severe plaque psoriasis: a pooled analysis of two phase 3 studies. J Drugs Dermatol. 2018;17:855-861.
- Gold LS, Lebwohl MG, Sugarman JL, et al. Safety and efficacy of a fixed combination of halobetasol and tazarotene in the treatment of moderate-to-severe plaque psoriasis: results of 2 phase 3 randomized controlled trials. J Am Acad Dermatol. 2018;79:287-293.
- Finlay AY. Current severe psoriasis and the rule of tens. Br J Dermatol. 2005;152:861-867.
- Campione E, Mazzotta A, Paterno EJ, et al. Effect of calcipotriol on etanercept partial responder psoriasis vulgaris and psoriatic arthritis patients. Acta Derm Venereol. 2009;89:288-291.
- Bagel J, Zapata J, Nelson E. A prospective, open-label study evaluating adjunctive calcipotriene 0.005%/betamethasone dipropionate 0.064% foam in psoriasis patients with inadequate response to biologic therapy. J Drugs Dermatol. 2018;17:845-850.
- Sugarman JL, Gold LS, Lebwohl MG, et al. A phase 2, multicenter, double-blind, randomized, vehicle controlled clinical study to assess the safety and efficacy of a halobetasol/tazarotene fixed combination in the treatment of plaque psoriasis. J Drugs Dermatol. 2017;16:197-204.
- Lebwohl MG, Sugarman JL, Gold LS, et al. Long-term safety results from a phase 3 open-label study of a fixed combination halobetasol propionate 0.01% and tazarotene 0.045% lotion in moderate-to-severe plaque psoriasis. J Am Acad Dermatol. 2019;80:282-285.
- Bhatia ND, Pariser DM, Kircik L, et al. Safety and efficacy of a halobetasol 0.01%/tazarotene 0.045% fixed combination lotion in the treatment of moderate-to-severe plaque psoriasis: a comparison with halobetasol propionate 0.05% cream. J Clin Aesthet Dermatol. 2018;11:15-19.
- Wang TS, Tsai TF. Biologics switch in psoriasis. Immunotherapy. 2019;11:531-541.
Psoriasis is a common chronic immunologic skin disease that affects approximately 7.4 million adults in the United States1 and more than 100 million individuals worldwide.2 Patients with psoriasis have a potentially heightened risk for cardiometabolic diseases, psychiatric disorders, and psoriatic arthritis,3 as well as impaired quality of life (QOL).4 Psoriasis also is associated with increased health care costs5 and may result in substantial socioeconomic repercussions for affected patients.6,7
Psoriasis treatments focus on relieving symptoms and improving patient QOL. Systemic therapy has been the mainstay of treatment for moderate to severe psoriasis.8 Although topical therapy usually is applied to treat mild symptoms, it also can be used as an adjunct to enhance efficacy of other treatment approaches.9 The National Psoriasis Foundation (NPF) recommends a treat-to-target (TTT) strategy for plaque psoriasis, the most common form of psoriasis, with a target response of attaining affected body surface area (BSA) of 1% or lower at 3 months after treatment initiation, allowing for regular assessments of treatment responses.10
Not all patients with moderate to severe psoriasis can achieve a satisfactory response with systemic biologic monotherapy.11 Switching to a new biologic improves responses in some but not all cases12 and could be associated with new safety issues and additional costs. Combinations of biologics with phototherapy, nonbiologic systemic agents, or topical medications were found to be more effective than biologics alone,9,11 though long-term safety studies are needed for biologics combined with other systemic inverventions.11
A lotion containing a fixed combination of halobetasol propionate (HP) 0.01%, a corticosteroid, and tazarotene (TAZ) 0.045%, a retinoid, is indicated for plaque psoriasis in adults.13 Two randomized, controlled, phase 3 trials demonstrated the rapid and sustained efficacy of HP-TAZ in treating moderate to severe plaque psoriasis without any safety concerns.14,15 However, combining HP-TAZ lotion with biologics has not been examined yet, to our knowledge.
This open-label study evaluated the effectiveness and safety of adjunctive HP-TAZ lotion in adult patients with moderate to severe plaque psoriasis who were being treated with biologics in a real-world setting. Potential cost savings with the addition of topical HP-TAZ to ongoing biologics vs switching to a new biologic also were assessed.
Methods
Study Design and Participants—A single-center, institutional review board–approved, open-label study evaluated adjunctive therapy with HP 0.01%–TAZ 0.045% lotion in patients with psoriasis being treated with biologic agents. The study was conducted in accordance with the ethical principles of the Declaration of Helsinki and in compliance with Good Clinical Practices. All patients provided written informed consent before enrollment.
Male and nonpregnant female patients (aged ≥18 years)with moderate to severe chronic plaque psoriasis and a BSA of 2% to 10% who were being treated with biologics for at least 24 weeks at baseline were enrolled. Patients were excluded if they had used oral systemic medications for psoriasis (≤4 weeks), other topical antipsoriatic therapies (≤14 days), UVB phototherapy (≤2 weeks), and psoralen plus UVA phototherapy (≤4 weeks) prior to study initiation. Concomitant use of steroid-free topical emollients or low-potency topical steroids and appropriate interventions deemed necessary by the investigator were allowed.
Although participants maintained their prescribed biologics for the duration of the study, HP-TAZ lotion also was applied once daily for 8 weeks, followed by once every other day for an additional 4 weeks. Participants then continued with biologics only for the last 4 weeks of the study.
Study Outcome Measures—Disease severity and treatment efficacy were assessed by affected BSA, Physician Global Assessment (PGA) score, composite BSA×PGA score, and participant-reported Dermatology Life Quality Index (DLQI). The primary end point was the proportion of participants achieving a BSA of 0% to 1% (NPF TTT status) at week 8. Secondary end points included the proportions of participants with BSA of 0% to 1% at weeks 12 and 16; BSA×PGA score at weeks 8, 12, and 16; and improvements in BSA, PGA, and DLQI at weeks 8, 12, and 16.
Adverse events (AEs) that occurred after the signing of the informed consent and for the duration of the participant’s participation were recorded, regardless of causality. Physical examinations were performed at screening; baseline; and weeks 8, 12, and 16 to document any clinically significant abnormalities. Localized skin reactions were assessed for tolerability of the study drug, with any reaction requiring concomitant therapy recorded as an AE.
The likelihood of switching to a new biologic regimen was assessed by the investigator for each participant at baseline and weeks 8, 12, and 16. Participants with unacceptable responses to their treatments (BSA >3%) were reported as likely to be considered for switching biologics by the investigator.
Pharmacoeconomic Evaluation—Potential cost savings were evaluated for the addition of HP-TAZ lotion to ongoing biologics vs switching to a new biologic. Cost comparisons were made in participants for whom the investigator would likely have switched biologics at baseline but not at the end of the study. For maintaining the same biologic with adjunctive topical HP-TAZ, total cost was estimated by adding the cost for 12 weeks (once daily for 8 weeks and once every other day for 4 weeks) of the HP-TAZ lotion to that of 16-week maintenance dosing with the biologic. The projected cost for switching to a new biologic for 16 weeks of treatment was based on both induction and maintenance dosing as recommended in its product label. Prices were obtained from the 2020 average wholesale price specialty pharmacy reports (BioPlus Specialty Pharmacy Services [https://www.bioplusrx.com]).
Data Handling—Enrollment of approximately 25 participants was desired for the study. Data on disease severity and participant-reported outcomes were assessed using descriptive statistics. Adverse events were summarized descriptively by incidence, severity, and relationship to the study drug. All participants with data available at a measured time point were included in the analyses for that time point.
Results
Participant Disposition and Demographics—Twenty-five participants (15 male and 10 female) were included in the study (Table 1). Seven participants discontinued the study for the following reasons: AEs (n=4), patient choice (n=2), and noncompliance (n=1).

The average age of the participants was 50 years, the majority were White (76.0% [19/25]) andnon-Hispanic (88.0% [22/25]), and the mean duration of chronic plaque psoriasis was 18.9 years (Table 1). Participants had been receiving biologic monotherapy for at least 24 weeks prior to enrollment, most commonly ustekinumab (32.0% [8/25])(Table 1). None had achieved the NPF TTT status with their biologics. At baseline, mean (SD) affected BSA, PGA, BSA×PGA, and participant-reported DLQI were 4.16% (2.04%), 2.84 (0.55), 11.88 (6.39), and 4.00 (4.74), respectively.
Efficacy Assessment—Application of HP-TAZ lotion in addition to the participants’ existing biologic therapy reduced severity of the disease, as evidenced by the reductions in mean BSA, PGA, and BSA×PGA. After 8 weeks of once-daily concomitant HP-TAZ use with biologic, mean BSA and PGA dropped by approximately 40% and 37%, respectively (Figures 1A and 1B). A greater reduction (54%) was found for mean BSA×PGA (Figure 1C). Disease severity continued to improve when the application schedule for HP-TAZ was changed to once every other day for 4 weeks, as mean BSA, PGA, and BSA×PGA decreased further at week 12. These beneficial effects were sustained during the last 4 weeks of the study after HP-TAZ was discontinued, with reductions of 57%, 43%, and 70% from baseline for mean BSA, PGA, and BSA×PGA, respectively (Figure 1).

The proportion of participants who achieved NPF TTT status increased from 0% at baseline to 20.0% (5/20) at week 8 with once-daily use of HP-TAZ plus biologic for 8 weeks (Figure 2). At week 12, more participants (64.7% [11/17]) achieved the treatment goal after application of HP-TAZ once every other day with biologic for 4 weeks. Most participants maintained NPF TTT status after HP-TAZ was discontinued; at week 16, 50.0% (9/18) attained the NPF treatment goal (Figure 2).
![Proportion of participants achieving National Psoriasis Foundation target-to-treat status (body surface area [BSA] ≤1%) at baseline and weeks 8, 12, and 16 Proportion of participants achieving National Psoriasis Foundation target-to-treat status (body surface area [BSA] ≤1%) at baseline and weeks 8, 12, and 16](https://cdn.mdedge.com/files/s3fs-public/Bagel_0222_2.JPG)
The mean DLQI score decreased from 4.00 at baseline to 2.45 after 8 weeks of concomitant use of once-daily HP-TAZ with biologic, reflecting a 39% score reduction. An additional 4 weeks of adjunctive HP-TAZ applied once every other day with biologic further decreased the DLQI score to 2.18 at week 12. Mean DLQI remained similar (2.33) after another 4 weeks of biologics alone. The proportion of participants reporting a DLQI score of 0 to 1 increased from 40% (10/25) at baseline to 60% (12/20) at week 8 and 76.5% (13/17) at week 12 with adjunctive HP-TAZ lotion use with biologic. At week 16, a DLQI score of 0 to 1 was reported in 61.1% (11/18) of participants after receiving only biologics for 4 weeks.
Safety Assessment—A total of 19 AEs were reported in 11 participants during the study; 16 AEs were considered treatment related in 8 participants (Table 2). The most common AEs were retinoid dermatitis (28% [7/25]), burning at the application site (8% [2/25]), and pruritus at the application site (8% [2/25]), all of which were considered related to the treatment. Among all AEs, 12 were mild in severity, and the remaining 7 were moderate. Adverse events led to early study termination in 4 participants, all with retinoid dermatitis as the primary reason.

Likelihood of Switching Biologics—At baseline, almost 90% (22/25) of participants were rated as likely to switch biologics by the investigator due to unacceptable responses to their currently prescribed biologics (BSA >3%)(Figure 3). The likelihood was greatly reduced by concomitant HP-TAZ, as the proportion of participants defined as nonresponders to their biologic decreased to 35% (7/20) with 8-week adjunctive application of once-daily HP-TAZ with biologic and further decreased to 23.5% (4/17) with another 4 weeks of adjunctive HP-TAZ applied every other day plus biologic. At week 16, after 4 weeks of biologics alone, the proportion was maintained at 33.3% (6/18).

Pharmacoeconomics of Adding Topical HP-TAZ vs Switching Biologics—In the participants whom the investigator reported as likely to switch biologics at baseline, 9 had improvements in disease control such that switching biologics was no longer considered necessary for them at week 16. Potential cost savings with adjunctive therapy of HP-TAZ plus biologic vs switching biologics were therefore evaluated in these 9 participants, who were receiving ustekinumab, adalimumab, guselkumab, ixekizumab, and secukinumab during the study (Table 3). The estimated total cost of 16-week maintenance dosing of biologics plus adjunct HP-TAZ lotion ranged from $14,675 (ustekinumab 45 mg) to $54,025 (secukinumab 300 mg), while switching to other most commonly prescribed biologics for 16 weeks would cost an estimated $33,340 to $106,400 (induction and subsequent maintenance phases)(Table 3). Most biologic plus HP-TAZ combinations were estimated to cost less than $30,000, potentially saving $4816 to $91,725 compared with switching to any of the other 7 biologics (Table 3). The relatively more expensive maintenance combination containing secukinumab plus HP-TAZ ($54,025) appeared to be a less expensive option when compared with switching to ustekinumab (90 mg)($83,097), ixekizumab (80 mg)($61,452), or risankizumab (150 mg)($57,030) as an alternative biologic.

Comment
Adjunctive Use of HP-TAZ Lotion—In the present study, we showed that adjunctive HP-TAZ lotion improved biologic treatment response and reduced disease severity in participants with moderate to severe psoriasis whose symptoms could not be adequately controlled by 24 weeks or more of biologic monotherapy in a real-world setting. Disease activity decreased as evidenced by reductions in all assessed effectiveness variables, including BSA involvement, PGA score, composite BSA×PGA score, and participant-reported DLQI score. Half of the participants achieved NPF TTT status at the end of the study. The treatment was well tolerated with no unexpected safety concerns. Compared with switching to a new biologic, adding topical HP-TAZ to ongoing biologics appeared to be a more cost-effective approach to enhance treatment effects. Our results suggest that adjunctive use of HP-TAZ lotion may be a safe, effective, and economical option for patients with psoriasis who are failing their ongoing biologic monotherapy.
Treat-to-Target Status—The NPF-recommended target response to a treatment for plaque psoriasis is BSA of 1% or lower at 3 months postinitiation.10 Patients in the current study had major psoriasis activity at study entry despite being treated with a biologic for at least 24 weeks, as none had attained NPF TTT status at baseline. Because the time period of prior biologic treatment (at least 24 weeks) is much longer than the 3 months suggested by NPF, we believe that we were observing a true failure of the biologic rather than a slow onset of treatment effects in these patients at the time of enrollment. By week 12, with the addition of HP-TAZ lotion to the biologic, a high rate of participants achieved NPF TTT status (64.7%), with most participants being able to maintain this TTT status at study end after 4 weeks of biologic alone. Most participants also reported no impact of psoriasis on their QOL (DLQI, 0–116; 76.5%) at week 12. Improvements we found in disease control with adjunctive HP-TAZ lotion plus biologic support prior reports showing enhanced responses when a topical medication was added to a biologic.17,18 Reductions in psoriasis activity after 8 weeks of combined biologics plus once-daily HP-TAZ also are consistent with 2 phase 3 RCTs in which a monotherapy of HP-TAZ lotion used once daily for 8 weeks reduced BSA and DLQI.15 Notably, in the current study, disease severity continued to decrease when dosing of HP-TAZ was reduced to once every other day for 4 weeks, and the improvements were maintained even after the adjunct topical therapy was discontinued.
Safety Profile of HP-TAZ Lotion—The overall safety profile in our study also was consistent with that previously reported for HP-TAZ lotion,15,19-21 with no new safety signals observed. The combination treatment was well tolerated, with most reported AEs being mild in severity. Adverse events were mostly related to application-site reactions, the most common being dermatitis (28%), which was likely attributable to the TAZ component of the topical regimen.15
Likelihood of Switching Biologics—Reduced disease activity was reflected by a decrease in the percentage of participants the investigator considered likely to change biologics, which was 88.0% at baseline but only 33.3% at the end of the study. Although switching to a different biologic agent can improve treatment effect,22 it could lead to a substantial increase in health care costs and use of resources compared with no switch.5 We found that switching to one of the other most commonly prescribed biologics could incur $4816 to $91,725 in additional costs in most cases when compared with the combination strategy we evaluated over the 16-week treatment period. Therefore, concomitant use of HP-TAZ lotion with the ongoing biologics would be a potentially more economical alternative for patients to achieve acceptable responses or the NPF TTT goal. Moreover, combination with an adjunctive topical medication could avoid potential risks associated with switching, such as new AEs with new biologic regimens or disease flare during any washout period sometimes adopted for switching biologics.
Study Limitations—Our estimated costs were based on average wholesale prices and did not reflect net prices paid by patients or health plans due to the lack of known discount rates.
Conclusion
In this real-world study, patients with psoriasis that failed to respond to biologic monotherapy had improved disease control and QOL and reported no new safety concerns with adjunctive use of HP-TAZ lotion. Adding HP-TAZ to the ongoing biologics could be a more cost-effective option vs switching biologics for patients whose psoriasis symptoms could not be controlled with biologic monotherapy. Taken together, our results support the use of HP-TAZ lotion as an effective and safe adjunctive topical therapy in combination with biologics for psoriasis treatment.
Acknowledgments—We acknowledge the medical writing assistance provided by Hui Zhang, PhD, and Kathleen Ohleth, PhD, from Precise Publications LLC (Far Hills, New Jersey), which was funded by Ortho Dermatologics.
Psoriasis is a common chronic immunologic skin disease that affects approximately 7.4 million adults in the United States1 and more than 100 million individuals worldwide.2 Patients with psoriasis have a potentially heightened risk for cardiometabolic diseases, psychiatric disorders, and psoriatic arthritis,3 as well as impaired quality of life (QOL).4 Psoriasis also is associated with increased health care costs5 and may result in substantial socioeconomic repercussions for affected patients.6,7
Psoriasis treatments focus on relieving symptoms and improving patient QOL. Systemic therapy has been the mainstay of treatment for moderate to severe psoriasis.8 Although topical therapy usually is applied to treat mild symptoms, it also can be used as an adjunct to enhance efficacy of other treatment approaches.9 The National Psoriasis Foundation (NPF) recommends a treat-to-target (TTT) strategy for plaque psoriasis, the most common form of psoriasis, with a target response of attaining affected body surface area (BSA) of 1% or lower at 3 months after treatment initiation, allowing for regular assessments of treatment responses.10
Not all patients with moderate to severe psoriasis can achieve a satisfactory response with systemic biologic monotherapy.11 Switching to a new biologic improves responses in some but not all cases12 and could be associated with new safety issues and additional costs. Combinations of biologics with phototherapy, nonbiologic systemic agents, or topical medications were found to be more effective than biologics alone,9,11 though long-term safety studies are needed for biologics combined with other systemic inverventions.11
A lotion containing a fixed combination of halobetasol propionate (HP) 0.01%, a corticosteroid, and tazarotene (TAZ) 0.045%, a retinoid, is indicated for plaque psoriasis in adults.13 Two randomized, controlled, phase 3 trials demonstrated the rapid and sustained efficacy of HP-TAZ in treating moderate to severe plaque psoriasis without any safety concerns.14,15 However, combining HP-TAZ lotion with biologics has not been examined yet, to our knowledge.
This open-label study evaluated the effectiveness and safety of adjunctive HP-TAZ lotion in adult patients with moderate to severe plaque psoriasis who were being treated with biologics in a real-world setting. Potential cost savings with the addition of topical HP-TAZ to ongoing biologics vs switching to a new biologic also were assessed.
Methods
Study Design and Participants—A single-center, institutional review board–approved, open-label study evaluated adjunctive therapy with HP 0.01%–TAZ 0.045% lotion in patients with psoriasis being treated with biologic agents. The study was conducted in accordance with the ethical principles of the Declaration of Helsinki and in compliance with Good Clinical Practices. All patients provided written informed consent before enrollment.
Male and nonpregnant female patients (aged ≥18 years)with moderate to severe chronic plaque psoriasis and a BSA of 2% to 10% who were being treated with biologics for at least 24 weeks at baseline were enrolled. Patients were excluded if they had used oral systemic medications for psoriasis (≤4 weeks), other topical antipsoriatic therapies (≤14 days), UVB phototherapy (≤2 weeks), and psoralen plus UVA phototherapy (≤4 weeks) prior to study initiation. Concomitant use of steroid-free topical emollients or low-potency topical steroids and appropriate interventions deemed necessary by the investigator were allowed.
Although participants maintained their prescribed biologics for the duration of the study, HP-TAZ lotion also was applied once daily for 8 weeks, followed by once every other day for an additional 4 weeks. Participants then continued with biologics only for the last 4 weeks of the study.
Study Outcome Measures—Disease severity and treatment efficacy were assessed by affected BSA, Physician Global Assessment (PGA) score, composite BSA×PGA score, and participant-reported Dermatology Life Quality Index (DLQI). The primary end point was the proportion of participants achieving a BSA of 0% to 1% (NPF TTT status) at week 8. Secondary end points included the proportions of participants with BSA of 0% to 1% at weeks 12 and 16; BSA×PGA score at weeks 8, 12, and 16; and improvements in BSA, PGA, and DLQI at weeks 8, 12, and 16.
Adverse events (AEs) that occurred after the signing of the informed consent and for the duration of the participant’s participation were recorded, regardless of causality. Physical examinations were performed at screening; baseline; and weeks 8, 12, and 16 to document any clinically significant abnormalities. Localized skin reactions were assessed for tolerability of the study drug, with any reaction requiring concomitant therapy recorded as an AE.
The likelihood of switching to a new biologic regimen was assessed by the investigator for each participant at baseline and weeks 8, 12, and 16. Participants with unacceptable responses to their treatments (BSA >3%) were reported as likely to be considered for switching biologics by the investigator.
Pharmacoeconomic Evaluation—Potential cost savings were evaluated for the addition of HP-TAZ lotion to ongoing biologics vs switching to a new biologic. Cost comparisons were made in participants for whom the investigator would likely have switched biologics at baseline but not at the end of the study. For maintaining the same biologic with adjunctive topical HP-TAZ, total cost was estimated by adding the cost for 12 weeks (once daily for 8 weeks and once every other day for 4 weeks) of the HP-TAZ lotion to that of 16-week maintenance dosing with the biologic. The projected cost for switching to a new biologic for 16 weeks of treatment was based on both induction and maintenance dosing as recommended in its product label. Prices were obtained from the 2020 average wholesale price specialty pharmacy reports (BioPlus Specialty Pharmacy Services [https://www.bioplusrx.com]).
Data Handling—Enrollment of approximately 25 participants was desired for the study. Data on disease severity and participant-reported outcomes were assessed using descriptive statistics. Adverse events were summarized descriptively by incidence, severity, and relationship to the study drug. All participants with data available at a measured time point were included in the analyses for that time point.
Results
Participant Disposition and Demographics—Twenty-five participants (15 male and 10 female) were included in the study (Table 1). Seven participants discontinued the study for the following reasons: AEs (n=4), patient choice (n=2), and noncompliance (n=1).

The average age of the participants was 50 years, the majority were White (76.0% [19/25]) andnon-Hispanic (88.0% [22/25]), and the mean duration of chronic plaque psoriasis was 18.9 years (Table 1). Participants had been receiving biologic monotherapy for at least 24 weeks prior to enrollment, most commonly ustekinumab (32.0% [8/25])(Table 1). None had achieved the NPF TTT status with their biologics. At baseline, mean (SD) affected BSA, PGA, BSA×PGA, and participant-reported DLQI were 4.16% (2.04%), 2.84 (0.55), 11.88 (6.39), and 4.00 (4.74), respectively.
Efficacy Assessment—Application of HP-TAZ lotion in addition to the participants’ existing biologic therapy reduced severity of the disease, as evidenced by the reductions in mean BSA, PGA, and BSA×PGA. After 8 weeks of once-daily concomitant HP-TAZ use with biologic, mean BSA and PGA dropped by approximately 40% and 37%, respectively (Figures 1A and 1B). A greater reduction (54%) was found for mean BSA×PGA (Figure 1C). Disease severity continued to improve when the application schedule for HP-TAZ was changed to once every other day for 4 weeks, as mean BSA, PGA, and BSA×PGA decreased further at week 12. These beneficial effects were sustained during the last 4 weeks of the study after HP-TAZ was discontinued, with reductions of 57%, 43%, and 70% from baseline for mean BSA, PGA, and BSA×PGA, respectively (Figure 1).

The proportion of participants who achieved NPF TTT status increased from 0% at baseline to 20.0% (5/20) at week 8 with once-daily use of HP-TAZ plus biologic for 8 weeks (Figure 2). At week 12, more participants (64.7% [11/17]) achieved the treatment goal after application of HP-TAZ once every other day with biologic for 4 weeks. Most participants maintained NPF TTT status after HP-TAZ was discontinued; at week 16, 50.0% (9/18) attained the NPF treatment goal (Figure 2).
![Proportion of participants achieving National Psoriasis Foundation target-to-treat status (body surface area [BSA] ≤1%) at baseline and weeks 8, 12, and 16 Proportion of participants achieving National Psoriasis Foundation target-to-treat status (body surface area [BSA] ≤1%) at baseline and weeks 8, 12, and 16](https://cdn.mdedge.com/files/s3fs-public/Bagel_0222_2.JPG)
The mean DLQI score decreased from 4.00 at baseline to 2.45 after 8 weeks of concomitant use of once-daily HP-TAZ with biologic, reflecting a 39% score reduction. An additional 4 weeks of adjunctive HP-TAZ applied once every other day with biologic further decreased the DLQI score to 2.18 at week 12. Mean DLQI remained similar (2.33) after another 4 weeks of biologics alone. The proportion of participants reporting a DLQI score of 0 to 1 increased from 40% (10/25) at baseline to 60% (12/20) at week 8 and 76.5% (13/17) at week 12 with adjunctive HP-TAZ lotion use with biologic. At week 16, a DLQI score of 0 to 1 was reported in 61.1% (11/18) of participants after receiving only biologics for 4 weeks.
Safety Assessment—A total of 19 AEs were reported in 11 participants during the study; 16 AEs were considered treatment related in 8 participants (Table 2). The most common AEs were retinoid dermatitis (28% [7/25]), burning at the application site (8% [2/25]), and pruritus at the application site (8% [2/25]), all of which were considered related to the treatment. Among all AEs, 12 were mild in severity, and the remaining 7 were moderate. Adverse events led to early study termination in 4 participants, all with retinoid dermatitis as the primary reason.

Likelihood of Switching Biologics—At baseline, almost 90% (22/25) of participants were rated as likely to switch biologics by the investigator due to unacceptable responses to their currently prescribed biologics (BSA >3%)(Figure 3). The likelihood was greatly reduced by concomitant HP-TAZ, as the proportion of participants defined as nonresponders to their biologic decreased to 35% (7/20) with 8-week adjunctive application of once-daily HP-TAZ with biologic and further decreased to 23.5% (4/17) with another 4 weeks of adjunctive HP-TAZ applied every other day plus biologic. At week 16, after 4 weeks of biologics alone, the proportion was maintained at 33.3% (6/18).

Pharmacoeconomics of Adding Topical HP-TAZ vs Switching Biologics—In the participants whom the investigator reported as likely to switch biologics at baseline, 9 had improvements in disease control such that switching biologics was no longer considered necessary for them at week 16. Potential cost savings with adjunctive therapy of HP-TAZ plus biologic vs switching biologics were therefore evaluated in these 9 participants, who were receiving ustekinumab, adalimumab, guselkumab, ixekizumab, and secukinumab during the study (Table 3). The estimated total cost of 16-week maintenance dosing of biologics plus adjunct HP-TAZ lotion ranged from $14,675 (ustekinumab 45 mg) to $54,025 (secukinumab 300 mg), while switching to other most commonly prescribed biologics for 16 weeks would cost an estimated $33,340 to $106,400 (induction and subsequent maintenance phases)(Table 3). Most biologic plus HP-TAZ combinations were estimated to cost less than $30,000, potentially saving $4816 to $91,725 compared with switching to any of the other 7 biologics (Table 3). The relatively more expensive maintenance combination containing secukinumab plus HP-TAZ ($54,025) appeared to be a less expensive option when compared with switching to ustekinumab (90 mg)($83,097), ixekizumab (80 mg)($61,452), or risankizumab (150 mg)($57,030) as an alternative biologic.

Comment
Adjunctive Use of HP-TAZ Lotion—In the present study, we showed that adjunctive HP-TAZ lotion improved biologic treatment response and reduced disease severity in participants with moderate to severe psoriasis whose symptoms could not be adequately controlled by 24 weeks or more of biologic monotherapy in a real-world setting. Disease activity decreased as evidenced by reductions in all assessed effectiveness variables, including BSA involvement, PGA score, composite BSA×PGA score, and participant-reported DLQI score. Half of the participants achieved NPF TTT status at the end of the study. The treatment was well tolerated with no unexpected safety concerns. Compared with switching to a new biologic, adding topical HP-TAZ to ongoing biologics appeared to be a more cost-effective approach to enhance treatment effects. Our results suggest that adjunctive use of HP-TAZ lotion may be a safe, effective, and economical option for patients with psoriasis who are failing their ongoing biologic monotherapy.
Treat-to-Target Status—The NPF-recommended target response to a treatment for plaque psoriasis is BSA of 1% or lower at 3 months postinitiation.10 Patients in the current study had major psoriasis activity at study entry despite being treated with a biologic for at least 24 weeks, as none had attained NPF TTT status at baseline. Because the time period of prior biologic treatment (at least 24 weeks) is much longer than the 3 months suggested by NPF, we believe that we were observing a true failure of the biologic rather than a slow onset of treatment effects in these patients at the time of enrollment. By week 12, with the addition of HP-TAZ lotion to the biologic, a high rate of participants achieved NPF TTT status (64.7%), with most participants being able to maintain this TTT status at study end after 4 weeks of biologic alone. Most participants also reported no impact of psoriasis on their QOL (DLQI, 0–116; 76.5%) at week 12. Improvements we found in disease control with adjunctive HP-TAZ lotion plus biologic support prior reports showing enhanced responses when a topical medication was added to a biologic.17,18 Reductions in psoriasis activity after 8 weeks of combined biologics plus once-daily HP-TAZ also are consistent with 2 phase 3 RCTs in which a monotherapy of HP-TAZ lotion used once daily for 8 weeks reduced BSA and DLQI.15 Notably, in the current study, disease severity continued to decrease when dosing of HP-TAZ was reduced to once every other day for 4 weeks, and the improvements were maintained even after the adjunct topical therapy was discontinued.
Safety Profile of HP-TAZ Lotion—The overall safety profile in our study also was consistent with that previously reported for HP-TAZ lotion,15,19-21 with no new safety signals observed. The combination treatment was well tolerated, with most reported AEs being mild in severity. Adverse events were mostly related to application-site reactions, the most common being dermatitis (28%), which was likely attributable to the TAZ component of the topical regimen.15
Likelihood of Switching Biologics—Reduced disease activity was reflected by a decrease in the percentage of participants the investigator considered likely to change biologics, which was 88.0% at baseline but only 33.3% at the end of the study. Although switching to a different biologic agent can improve treatment effect,22 it could lead to a substantial increase in health care costs and use of resources compared with no switch.5 We found that switching to one of the other most commonly prescribed biologics could incur $4816 to $91,725 in additional costs in most cases when compared with the combination strategy we evaluated over the 16-week treatment period. Therefore, concomitant use of HP-TAZ lotion with the ongoing biologics would be a potentially more economical alternative for patients to achieve acceptable responses or the NPF TTT goal. Moreover, combination with an adjunctive topical medication could avoid potential risks associated with switching, such as new AEs with new biologic regimens or disease flare during any washout period sometimes adopted for switching biologics.
Study Limitations—Our estimated costs were based on average wholesale prices and did not reflect net prices paid by patients or health plans due to the lack of known discount rates.
Conclusion
In this real-world study, patients with psoriasis that failed to respond to biologic monotherapy had improved disease control and QOL and reported no new safety concerns with adjunctive use of HP-TAZ lotion. Adding HP-TAZ to the ongoing biologics could be a more cost-effective option vs switching biologics for patients whose psoriasis symptoms could not be controlled with biologic monotherapy. Taken together, our results support the use of HP-TAZ lotion as an effective and safe adjunctive topical therapy in combination with biologics for psoriasis treatment.
Acknowledgments—We acknowledge the medical writing assistance provided by Hui Zhang, PhD, and Kathleen Ohleth, PhD, from Precise Publications LLC (Far Hills, New Jersey), which was funded by Ortho Dermatologics.
- Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014;70:512-516.
- Global Report on Psoriasis. World Health Organization; 2016. Accessed January 11, 2022. https://apps.who.int/iris/handle/10665/204417
- Takeshita J, Grewal S, Langan SM, et al. Psoriasis and comorbid diseases: epidemiology. J Am Acad Dermatol. 2017;76:377-390.
- Moller AH, Erntoft S, Vinding GR, et al. A systematic literature review to compare quality of life in psoriasis with other chronic diseases using EQ-5D-derived utility values. Patient Relat Outcome Meas. 2015;6:167-177.
- Feldman SR, Tian H, Wang X, et al. Health care utilization and cost associated with biologic treatment patterns among patients with moderate to severe psoriasis: analyses from a large U.S. claims database. J Manag Care Spec Pharm. 2019;25:479-488.
- Thomsen SF, Skov L, Dodge R, et al. Socioeconomic costs and health inequalities from psoriasis: a cohort study. Dermatology. 2019;235:372-379.
- Fowler JF, Duh MS, Rovba L, et al. The impact of psoriasis on health care costs and patient work loss. J Am Acad Dermatol. 2008;59:772-780.
- Menter A, Gottlieb A, Feldman SR, et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: section 1. overview of psoriasis and guidelines of care for the treatment of psoriasis with biologics. J Am Acad Dermatol. 2008;58:826-850.
- Bagel J, Gold LS. Combining topical psoriasis treatment to enhance systemic and phototherapy: a review of the literature. J Drugs Dermatol. 2017;16:1209-1222.
- Armstrong AW, Siegel MP, Bagel J, et al. From the Medical Board of the National Psoriasis Foundation: treatment targets for plaque psoriasis. J Am Acad Dermatol. 2017;76:290-298.
- Armstrong AW, Bagel J, Van Voorhees AS, et al. Combining biologic therapies with other systemic treatments in psoriasis: evidence-based, best-practice recommendations from the Medical Board of the National Psoriasis Foundation. JAMA Dermatol. 2015;151:432-438.
- Menter A, Strober BE, Kaplan DH, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with biologics. J Am Acad Dermatol. 2019;80:1029-1072.
- Duobrii. Prescribing information. Bausch Health Companies Inc; 2019.
- Sugarman JL, Weiss J, Tanghetti EA, et al. Safety and efficacy of a fixed combination halobetasol and tazarotene lotion in the treatment of moderate-to-severe plaque psoriasis: a pooled analysis of two phase 3 studies. J Drugs Dermatol. 2018;17:855-861.
- Gold LS, Lebwohl MG, Sugarman JL, et al. Safety and efficacy of a fixed combination of halobetasol and tazarotene in the treatment of moderate-to-severe plaque psoriasis: results of 2 phase 3 randomized controlled trials. J Am Acad Dermatol. 2018;79:287-293.
- Finlay AY. Current severe psoriasis and the rule of tens. Br J Dermatol. 2005;152:861-867.
- Campione E, Mazzotta A, Paterno EJ, et al. Effect of calcipotriol on etanercept partial responder psoriasis vulgaris and psoriatic arthritis patients. Acta Derm Venereol. 2009;89:288-291.
- Bagel J, Zapata J, Nelson E. A prospective, open-label study evaluating adjunctive calcipotriene 0.005%/betamethasone dipropionate 0.064% foam in psoriasis patients with inadequate response to biologic therapy. J Drugs Dermatol. 2018;17:845-850.
- Sugarman JL, Gold LS, Lebwohl MG, et al. A phase 2, multicenter, double-blind, randomized, vehicle controlled clinical study to assess the safety and efficacy of a halobetasol/tazarotene fixed combination in the treatment of plaque psoriasis. J Drugs Dermatol. 2017;16:197-204.
- Lebwohl MG, Sugarman JL, Gold LS, et al. Long-term safety results from a phase 3 open-label study of a fixed combination halobetasol propionate 0.01% and tazarotene 0.045% lotion in moderate-to-severe plaque psoriasis. J Am Acad Dermatol. 2019;80:282-285.
- Bhatia ND, Pariser DM, Kircik L, et al. Safety and efficacy of a halobetasol 0.01%/tazarotene 0.045% fixed combination lotion in the treatment of moderate-to-severe plaque psoriasis: a comparison with halobetasol propionate 0.05% cream. J Clin Aesthet Dermatol. 2018;11:15-19.
- Wang TS, Tsai TF. Biologics switch in psoriasis. Immunotherapy. 2019;11:531-541.
- Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014;70:512-516.
- Global Report on Psoriasis. World Health Organization; 2016. Accessed January 11, 2022. https://apps.who.int/iris/handle/10665/204417
- Takeshita J, Grewal S, Langan SM, et al. Psoriasis and comorbid diseases: epidemiology. J Am Acad Dermatol. 2017;76:377-390.
- Moller AH, Erntoft S, Vinding GR, et al. A systematic literature review to compare quality of life in psoriasis with other chronic diseases using EQ-5D-derived utility values. Patient Relat Outcome Meas. 2015;6:167-177.
- Feldman SR, Tian H, Wang X, et al. Health care utilization and cost associated with biologic treatment patterns among patients with moderate to severe psoriasis: analyses from a large U.S. claims database. J Manag Care Spec Pharm. 2019;25:479-488.
- Thomsen SF, Skov L, Dodge R, et al. Socioeconomic costs and health inequalities from psoriasis: a cohort study. Dermatology. 2019;235:372-379.
- Fowler JF, Duh MS, Rovba L, et al. The impact of psoriasis on health care costs and patient work loss. J Am Acad Dermatol. 2008;59:772-780.
- Menter A, Gottlieb A, Feldman SR, et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: section 1. overview of psoriasis and guidelines of care for the treatment of psoriasis with biologics. J Am Acad Dermatol. 2008;58:826-850.
- Bagel J, Gold LS. Combining topical psoriasis treatment to enhance systemic and phototherapy: a review of the literature. J Drugs Dermatol. 2017;16:1209-1222.
- Armstrong AW, Siegel MP, Bagel J, et al. From the Medical Board of the National Psoriasis Foundation: treatment targets for plaque psoriasis. J Am Acad Dermatol. 2017;76:290-298.
- Armstrong AW, Bagel J, Van Voorhees AS, et al. Combining biologic therapies with other systemic treatments in psoriasis: evidence-based, best-practice recommendations from the Medical Board of the National Psoriasis Foundation. JAMA Dermatol. 2015;151:432-438.
- Menter A, Strober BE, Kaplan DH, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with biologics. J Am Acad Dermatol. 2019;80:1029-1072.
- Duobrii. Prescribing information. Bausch Health Companies Inc; 2019.
- Sugarman JL, Weiss J, Tanghetti EA, et al. Safety and efficacy of a fixed combination halobetasol and tazarotene lotion in the treatment of moderate-to-severe plaque psoriasis: a pooled analysis of two phase 3 studies. J Drugs Dermatol. 2018;17:855-861.
- Gold LS, Lebwohl MG, Sugarman JL, et al. Safety and efficacy of a fixed combination of halobetasol and tazarotene in the treatment of moderate-to-severe plaque psoriasis: results of 2 phase 3 randomized controlled trials. J Am Acad Dermatol. 2018;79:287-293.
- Finlay AY. Current severe psoriasis and the rule of tens. Br J Dermatol. 2005;152:861-867.
- Campione E, Mazzotta A, Paterno EJ, et al. Effect of calcipotriol on etanercept partial responder psoriasis vulgaris and psoriatic arthritis patients. Acta Derm Venereol. 2009;89:288-291.
- Bagel J, Zapata J, Nelson E. A prospective, open-label study evaluating adjunctive calcipotriene 0.005%/betamethasone dipropionate 0.064% foam in psoriasis patients with inadequate response to biologic therapy. J Drugs Dermatol. 2018;17:845-850.
- Sugarman JL, Gold LS, Lebwohl MG, et al. A phase 2, multicenter, double-blind, randomized, vehicle controlled clinical study to assess the safety and efficacy of a halobetasol/tazarotene fixed combination in the treatment of plaque psoriasis. J Drugs Dermatol. 2017;16:197-204.
- Lebwohl MG, Sugarman JL, Gold LS, et al. Long-term safety results from a phase 3 open-label study of a fixed combination halobetasol propionate 0.01% and tazarotene 0.045% lotion in moderate-to-severe plaque psoriasis. J Am Acad Dermatol. 2019;80:282-285.
- Bhatia ND, Pariser DM, Kircik L, et al. Safety and efficacy of a halobetasol 0.01%/tazarotene 0.045% fixed combination lotion in the treatment of moderate-to-severe plaque psoriasis: a comparison with halobetasol propionate 0.05% cream. J Clin Aesthet Dermatol. 2018;11:15-19.
- Wang TS, Tsai TF. Biologics switch in psoriasis. Immunotherapy. 2019;11:531-541.
Practice Points
- Although monotherapy with biologic agents is effective to treat psoriasis, some patients do not achieve a satisfactory response.
- Adjunctive therapy with halobetasol propionate (HP) 0.01%–tazarotene (TAZ) 0.045% lotion can improve responses to biologic treatment in patients whose psoriasis symptoms could not be adequately controlled by biologic monotherapy.
- Adjunctive use of HP-TAZ lotion in addition to biologics was well tolerated.
- Compared with switching to a new biologic regimen, adding a topical regimen of HP-TAZ lotion to ongoing biologics may be a more cost-effective approach to enhance treatment effects.