User login
Commentary: Functional assessment developed for older adults with sickle cell disease
As individuals with sickle cell disease (SCD) are living longer than ever before there is a greater need to focus on maintaining and improving function and independence in this growing population. In the general population, impairments in functional measures such as usual gait speed, grip strength, Timed Up and Go, and cognition are associated with adverse health outcomes such as falls, fractures, loss of independence, and death.
Adults with SCD experience multiple complications such as avascular necrosis of the joints, retinopathy, and strokes that lead to functional limitations similar to those experienced by geriatric populations. However, functional assessments are not routinely performed during clinic visits with older adults with SCD.
In order to address this gap in care, my colleagues and I developed the first functional assessment for older adults with SCD, called the Sickle Cell Disease Functional Assessment (SCD-FA). This assessment will allow providers to evaluate the capabilities and vulnerabilities of older adults with SCD.
We assessed the feasibility of administering the SCD-FA in a prospective cohort pilot study. We enrolled 40 adults with SCD (20 older adults aged at least 50 years and 20 younger adults aged 18-49 years as a comparison group). All participants were assessed at steady-state.
For the SCD-FA, we selected geriatric assessment measures across seven domains: functional status, comorbid medical conditions, psychological state, social support, nutritional status, cognition, and medications. Several of these measures were previously validated in an oncology geriatric assessment and enriched with additional physical and cognitive measures to evaluate conditions at the intersection of SCD and geriatrics.
In September 2020, we published a protocol describing the methods and rationale for selecting measures for the SCD-FA in Pilot and Feasibility Studies.1 The preliminary data was presented at the annual meeting of the American Society of Hematology in December 2020 and was included in the annual Hematology and Aging Poster Walk.
The results of this pilot study showed that the SCD-FA is feasible (91% of participants who consented completed the SCD-FA), acceptable (95% reported the length as appropriate and had no difficulty understanding the measures), and safe with no adverse events.2 On physical performance testing, both younger and older participants had results consistent with accelerated aging with a functional age at least 20-30 years older than their chronological age.2
The majority of the participants (63%) had a usual gait speed slower than the speed required to safely cross the street at an intersection, and 25% had a gait speed slower than 1 m/s, which has been associated with increased mortality in the general population.3,4
Benefits to management
The SCD-FA can improve management of adults with SCD by:
- Characterizing their capabilities and physiological age, identifying individuals at high risk for functional decline and death early identifying targets for interventions that have been successful in geriatrics,5 assessing risk of toxicity from curative therapies, and evaluating functional response to SCD-specific therapies.
The SCD-FA provides a framework for developing exercise interventions to target functional impairments. This work supports our goal of improving the quality of life and longevity for people with SCD.
Dr. Oyedeji is a senior hematology Fellow at the department of medicine, division of hematology, Duke University, Durham, N.C. She reported that she has no conflicts of interest.
References
1. Pilot Feasibility Stud. 2020;6:131.
2. Blood. 2020;136(Supplement 1):26-7.
3. J Rehabil Res Dev. 2005;42(4):535-46.
4. JAMA. 2011;305(1):50-8.
5. South Med J. 1994;87(5):S83-7.
As individuals with sickle cell disease (SCD) are living longer than ever before there is a greater need to focus on maintaining and improving function and independence in this growing population. In the general population, impairments in functional measures such as usual gait speed, grip strength, Timed Up and Go, and cognition are associated with adverse health outcomes such as falls, fractures, loss of independence, and death.
Adults with SCD experience multiple complications such as avascular necrosis of the joints, retinopathy, and strokes that lead to functional limitations similar to those experienced by geriatric populations. However, functional assessments are not routinely performed during clinic visits with older adults with SCD.
In order to address this gap in care, my colleagues and I developed the first functional assessment for older adults with SCD, called the Sickle Cell Disease Functional Assessment (SCD-FA). This assessment will allow providers to evaluate the capabilities and vulnerabilities of older adults with SCD.
We assessed the feasibility of administering the SCD-FA in a prospective cohort pilot study. We enrolled 40 adults with SCD (20 older adults aged at least 50 years and 20 younger adults aged 18-49 years as a comparison group). All participants were assessed at steady-state.
For the SCD-FA, we selected geriatric assessment measures across seven domains: functional status, comorbid medical conditions, psychological state, social support, nutritional status, cognition, and medications. Several of these measures were previously validated in an oncology geriatric assessment and enriched with additional physical and cognitive measures to evaluate conditions at the intersection of SCD and geriatrics.
In September 2020, we published a protocol describing the methods and rationale for selecting measures for the SCD-FA in Pilot and Feasibility Studies.1 The preliminary data was presented at the annual meeting of the American Society of Hematology in December 2020 and was included in the annual Hematology and Aging Poster Walk.
The results of this pilot study showed that the SCD-FA is feasible (91% of participants who consented completed the SCD-FA), acceptable (95% reported the length as appropriate and had no difficulty understanding the measures), and safe with no adverse events.2 On physical performance testing, both younger and older participants had results consistent with accelerated aging with a functional age at least 20-30 years older than their chronological age.2
The majority of the participants (63%) had a usual gait speed slower than the speed required to safely cross the street at an intersection, and 25% had a gait speed slower than 1 m/s, which has been associated with increased mortality in the general population.3,4
Benefits to management
The SCD-FA can improve management of adults with SCD by:
- Characterizing their capabilities and physiological age, identifying individuals at high risk for functional decline and death early identifying targets for interventions that have been successful in geriatrics,5 assessing risk of toxicity from curative therapies, and evaluating functional response to SCD-specific therapies.
The SCD-FA provides a framework for developing exercise interventions to target functional impairments. This work supports our goal of improving the quality of life and longevity for people with SCD.
Dr. Oyedeji is a senior hematology Fellow at the department of medicine, division of hematology, Duke University, Durham, N.C. She reported that she has no conflicts of interest.
References
1. Pilot Feasibility Stud. 2020;6:131.
2. Blood. 2020;136(Supplement 1):26-7.
3. J Rehabil Res Dev. 2005;42(4):535-46.
4. JAMA. 2011;305(1):50-8.
5. South Med J. 1994;87(5):S83-7.
As individuals with sickle cell disease (SCD) are living longer than ever before there is a greater need to focus on maintaining and improving function and independence in this growing population. In the general population, impairments in functional measures such as usual gait speed, grip strength, Timed Up and Go, and cognition are associated with adverse health outcomes such as falls, fractures, loss of independence, and death.
Adults with SCD experience multiple complications such as avascular necrosis of the joints, retinopathy, and strokes that lead to functional limitations similar to those experienced by geriatric populations. However, functional assessments are not routinely performed during clinic visits with older adults with SCD.
In order to address this gap in care, my colleagues and I developed the first functional assessment for older adults with SCD, called the Sickle Cell Disease Functional Assessment (SCD-FA). This assessment will allow providers to evaluate the capabilities and vulnerabilities of older adults with SCD.
We assessed the feasibility of administering the SCD-FA in a prospective cohort pilot study. We enrolled 40 adults with SCD (20 older adults aged at least 50 years and 20 younger adults aged 18-49 years as a comparison group). All participants were assessed at steady-state.
For the SCD-FA, we selected geriatric assessment measures across seven domains: functional status, comorbid medical conditions, psychological state, social support, nutritional status, cognition, and medications. Several of these measures were previously validated in an oncology geriatric assessment and enriched with additional physical and cognitive measures to evaluate conditions at the intersection of SCD and geriatrics.
In September 2020, we published a protocol describing the methods and rationale for selecting measures for the SCD-FA in Pilot and Feasibility Studies.1 The preliminary data was presented at the annual meeting of the American Society of Hematology in December 2020 and was included in the annual Hematology and Aging Poster Walk.
The results of this pilot study showed that the SCD-FA is feasible (91% of participants who consented completed the SCD-FA), acceptable (95% reported the length as appropriate and had no difficulty understanding the measures), and safe with no adverse events.2 On physical performance testing, both younger and older participants had results consistent with accelerated aging with a functional age at least 20-30 years older than their chronological age.2
The majority of the participants (63%) had a usual gait speed slower than the speed required to safely cross the street at an intersection, and 25% had a gait speed slower than 1 m/s, which has been associated with increased mortality in the general population.3,4
Benefits to management
The SCD-FA can improve management of adults with SCD by:
- Characterizing their capabilities and physiological age, identifying individuals at high risk for functional decline and death early identifying targets for interventions that have been successful in geriatrics,5 assessing risk of toxicity from curative therapies, and evaluating functional response to SCD-specific therapies.
The SCD-FA provides a framework for developing exercise interventions to target functional impairments. This work supports our goal of improving the quality of life and longevity for people with SCD.
Dr. Oyedeji is a senior hematology Fellow at the department of medicine, division of hematology, Duke University, Durham, N.C. She reported that she has no conflicts of interest.
References
1. Pilot Feasibility Stud. 2020;6:131.
2. Blood. 2020;136(Supplement 1):26-7.
3. J Rehabil Res Dev. 2005;42(4):535-46.
4. JAMA. 2011;305(1):50-8.
5. South Med J. 1994;87(5):S83-7.
IMvigor130: A treasure trove of data for urothelial carcinoma
A second interim overall survival (OS) analysis suggested that atezolizumab monotherapy provides a clinical benefit as first-line treatment for mUC patients with PD-L1–expressing immune cells representing at least 5% of the tumor area (IC2/3), including patients who are cisplatin ineligible.
The analysis also suggested that atezolizumab plus chemotherapy produces similar OS results as chemotherapy plus placebo, but patients receiving atezolizumab may do better with cisplatin-based chemotherapy than with carboplatin-based chemotherapy.
These results were reported in two presentations at the American Association for Cancer Research Annual Meeting 2021: Week 1.
Current guidelines from the National Comprehensive Cancer Network and the European Society for Medical Oncology recommend atezolizumab monotherapy for cisplatin-ineligible patients with mUC and PD-L1 IC2/3.
The ongoing phase 3 IMvigor130 trial was designed to compare atezolizumab plus gemcitabine/platinum chemotherapy, atezolizumab monotherapy, and placebo plus chemotherapy. Platinum-based chemotherapy included either cisplatin or carboplatin, per investigator choice.
Coprimary endpoints for IMvigor130 were progression-free survival (PFS) and OS for atezolizumab plus chemotherapy versus placebo plus chemotherapy. The hierarchical study design dictated that OS would only be assessed for the comparison of atezolizumab monotherapy versus placebo-chemotherapy in the overall and PD-L1 IC2/3 populations if there was statistical improvement in OS for the atezolizumab-chemotherapy arm over the placebo-chemotherapy arm.
Secondary endpoints were overall response rate (ORR; per RECIST 1.1), duration of response (DOR) for all patients, and PFS for the comparison between atezolizumab monotherapy and placebo-chemotherapy. Exploratory analyses were performed on cisplatin-ineligible patients by PD-L1 status.
At the time of the primary analysis, an OS benefit for atezolizumab-chemotherapy over placebo-chemotherapy was not observed. Therefore, the OS benefit of atezolizumab monotherapy versus placebo-chemotherapy was not assessed. However, a trend toward improved OS was noted with atezolizumab for PD-L1 IC2/3 patients, including cisplatin-ineligible patients.
Atezolizumab vs. placebo-chemo
Ian D. Davis, MBBS, PhD, of Monash University in Melbourne, presented the second interim analysis of OS with atezolizumab monotherapy versus placebo plus chemotherapy (Abstract CT040).
The median follow-up was 14.9 months for atezolizumab monotherapy (n = 360) and 11.8 months for placebo-chemotherapy (n = 359). The median OS was 15.2 months and 13.1 months, respectively (hazard ratio, 0.99; 95% confidence interval, 0.83-1.19). There was no apparent OS benefit of atezolizumab for any clinically selected subgroup.
The ORR was 23.4% for atezolizumab monotherapy and 44.1% for placebo-chemotherapy. The median DOR was more than 3.5 times longer for atezolizumab monotherapy than for placebo-chemotherapy – 29.6 months and 8.1 months, respectively.
Although there was no formal statistical comparison, exploratory subgroup analyses demonstrated that the median OS for the PD-L1 IC2/3 patients appeared higher in the atezolizumab monotherapy arm than in the placebo-chemotherapy arm – 27.5 months and 16.7 months, respectively.
Similarly, the median OS for cisplatin-ineligible PD-L1 IC2/3 patients appeared higher for atezolizumab monotherapy than for placebo-chemotherapy – 18.6 months and 10.0 months, respectively.
In terms of safety, atezolizumab monotherapy compared favorably with placebo plus chemotherapy. There were similar numbers of grade 3/4 adverse events and comparable adverse events leading to discontinuation of treatment in both arms.
The atezolizumab monotherapy arm had fewer adverse events leading to withdrawal from any treatment, when compared with the placebo-chemotherapy arm – 7% and 34%, respectively. Two patients in the atezolizumab arm and one in the placebo-chemotherapy died of treatment-related causes.
Atezolizumab-chemo vs. placebo-chemo
Matthew D. Galsky, MD, of Mount Sinai Health System and Icahn School of Medicine at Mount Sinai in New York, presented the second interim OS comparison of atezolizumab plus chemotherapy with placebo plus chemotherapy (Abstract CT042).
The primary analysis had shown a statistically significant improvement in PFS for patients on atezolizumab-chemotherapy, in comparison with placebo-chemotherapy, with encouraging OS improvement, but the boundary for declaring significance for the OS endpoint was not crossed (Lancet. 2020 May 16;395[10236]:1547-1557).
Because IMvigor130 included both patients who received cisplatin and patients who investigators deemed cisplatin ineligible, the second interim analysis included an exploratory analysis of whether there was a difference in outcome between patients who received or did not receive cisplatin.
At a median follow-up of 13.3 months, the median OS was not significantly different in the atezolizumab-chemotherapy arm (n = 451) and the placebo-chemotherapy arm (n = 400) – 16.1 months and 13.4 months, respectively (HR, 0.84; 95% CI, 0.71-1.00; P = .026).
There were no clinically or pathologically defined subgroups that experienced an OS benefit from atezolizumab-chemotherapy over placebo-chemotherapy.
As for subsequent nonprotocol therapy, 24% of the placebo-chemotherapy arm received an immune checkpoint inhibitor at progression, as did 7% of the atezolizumab-chemotherapy arm. There was no difference in receipt of an immune checkpoint inhibitor post progression among patients treated with cisplatin versus carboplatin.
The benefit of combining atezolizumab with chemotherapy appeared more substantial with cisplatin-based chemotherapy than with carboplatin-based treatment. With cisplatin, the median OS was 21.6 months for the atezolizumab-chemotherapy arm and 14.6 months for the placebo-chemotherapy arm. With carboplatin, the median OS was 14.3 months and 13.0 months, respectively.
PD-L1 status was prognostic for patients who received cisplatin, with lower OS being observed for patients with PD-L1 IC0/1 status and higher OS observed for patients with PD-L1 IC2/3 status. Atezolizumab plus cisplatin-based chemotherapy appeared superior to cisplatin-based chemotherapy alone in both PD-L1–low and –high groups.
Atezolizumab did not seem to benefit patients who were treated with carboplatin, and PD-L1 status did not seem to influence OS among the carboplatin-treated patients.
Although similar ORR results were seen with cisplatin and carboplatin, there appeared to be a longer median DOR among cisplatin-treated patients who received atezolizumab than among those who did not – 13.2 months and 8.3 months, respectively.
No such benefit from atezolizumab was seen in carboplatin-treated patients. The median DOR was 8.1 months among patients who received atezolizumab and 7.1 months among those who did not.
The overall safety profile for atezolizumab plus chemotherapy was consistent with prior reports of the combination. Treatment-related grade 3-5 adverse events were similar on the atezolizumab-chemotherapy arm and the placebo-chemotherapy arm.
The present and future
The investigators who presented the second interim analysis for OS of the IMvigor130 trial were appropriately modest in their conclusions. After all, the prespecified boundary for significant improvement in OS for the addition of atezolizumab to chemotherapy was not crossed. No change in guideline-based clinical practice would be appropriate at the present time.
The various exploratory analyses are hypothesis generating and invite potential mechanistic explanations. However, given the nonrandom allocation of patients to cisplatin- or carboplatin-based chemotherapy, unrecognized variables may have influenced any appearance of a difference in OS between the regimens.
In IMvigor130, treatment was given until unacceptable toxicity or disease progression. It is uncertain whether the current National Comprehensive Cancer Network category 1 recommendation of chemotherapy induction followed by immune checkpoint inhibitor maintenance therapy will prove superior to the IMvigor130 strategy.
Clearly – and concordant with current treatment guidelines – atezolizumab monotherapy can benefit some patients, though the response rate for atezolizumab monotherapy was lower than for chemotherapy (23.4% vs. 44.1%).
As noted by the session chair, Marina Chiara Garassino, MD, of the University of Chicago, the OS curves were initially superior for chemotherapy over atezolizumab. However, the apparent early OS benefit for chemotherapy dissipated over time and, among responders to atezolizumab, response duration was considerably longer than for chemotherapy.
IMvigor130 will ultimately have a final OS analysis to clarify the relative benefits of the various treatment strategies. Fortunately, this large phase 3 study will yield a treasure trove of data to inform future research and build on the advances of recent years for patients with advanced urothelial cancer.
IMvigor130 is sponsored by Hoffmann-La Roche. Dr. Davis, Dr. Galsky, and Dr. Garassino disclosed relationships with Hoffmann-La Roche and many other companies.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
A second interim overall survival (OS) analysis suggested that atezolizumab monotherapy provides a clinical benefit as first-line treatment for mUC patients with PD-L1–expressing immune cells representing at least 5% of the tumor area (IC2/3), including patients who are cisplatin ineligible.
The analysis also suggested that atezolizumab plus chemotherapy produces similar OS results as chemotherapy plus placebo, but patients receiving atezolizumab may do better with cisplatin-based chemotherapy than with carboplatin-based chemotherapy.
These results were reported in two presentations at the American Association for Cancer Research Annual Meeting 2021: Week 1.
Current guidelines from the National Comprehensive Cancer Network and the European Society for Medical Oncology recommend atezolizumab monotherapy for cisplatin-ineligible patients with mUC and PD-L1 IC2/3.
The ongoing phase 3 IMvigor130 trial was designed to compare atezolizumab plus gemcitabine/platinum chemotherapy, atezolizumab monotherapy, and placebo plus chemotherapy. Platinum-based chemotherapy included either cisplatin or carboplatin, per investigator choice.
Coprimary endpoints for IMvigor130 were progression-free survival (PFS) and OS for atezolizumab plus chemotherapy versus placebo plus chemotherapy. The hierarchical study design dictated that OS would only be assessed for the comparison of atezolizumab monotherapy versus placebo-chemotherapy in the overall and PD-L1 IC2/3 populations if there was statistical improvement in OS for the atezolizumab-chemotherapy arm over the placebo-chemotherapy arm.
Secondary endpoints were overall response rate (ORR; per RECIST 1.1), duration of response (DOR) for all patients, and PFS for the comparison between atezolizumab monotherapy and placebo-chemotherapy. Exploratory analyses were performed on cisplatin-ineligible patients by PD-L1 status.
At the time of the primary analysis, an OS benefit for atezolizumab-chemotherapy over placebo-chemotherapy was not observed. Therefore, the OS benefit of atezolizumab monotherapy versus placebo-chemotherapy was not assessed. However, a trend toward improved OS was noted with atezolizumab for PD-L1 IC2/3 patients, including cisplatin-ineligible patients.
Atezolizumab vs. placebo-chemo
Ian D. Davis, MBBS, PhD, of Monash University in Melbourne, presented the second interim analysis of OS with atezolizumab monotherapy versus placebo plus chemotherapy (Abstract CT040).
The median follow-up was 14.9 months for atezolizumab monotherapy (n = 360) and 11.8 months for placebo-chemotherapy (n = 359). The median OS was 15.2 months and 13.1 months, respectively (hazard ratio, 0.99; 95% confidence interval, 0.83-1.19). There was no apparent OS benefit of atezolizumab for any clinically selected subgroup.
The ORR was 23.4% for atezolizumab monotherapy and 44.1% for placebo-chemotherapy. The median DOR was more than 3.5 times longer for atezolizumab monotherapy than for placebo-chemotherapy – 29.6 months and 8.1 months, respectively.
Although there was no formal statistical comparison, exploratory subgroup analyses demonstrated that the median OS for the PD-L1 IC2/3 patients appeared higher in the atezolizumab monotherapy arm than in the placebo-chemotherapy arm – 27.5 months and 16.7 months, respectively.
Similarly, the median OS for cisplatin-ineligible PD-L1 IC2/3 patients appeared higher for atezolizumab monotherapy than for placebo-chemotherapy – 18.6 months and 10.0 months, respectively.
In terms of safety, atezolizumab monotherapy compared favorably with placebo plus chemotherapy. There were similar numbers of grade 3/4 adverse events and comparable adverse events leading to discontinuation of treatment in both arms.
The atezolizumab monotherapy arm had fewer adverse events leading to withdrawal from any treatment, when compared with the placebo-chemotherapy arm – 7% and 34%, respectively. Two patients in the atezolizumab arm and one in the placebo-chemotherapy died of treatment-related causes.
Atezolizumab-chemo vs. placebo-chemo
Matthew D. Galsky, MD, of Mount Sinai Health System and Icahn School of Medicine at Mount Sinai in New York, presented the second interim OS comparison of atezolizumab plus chemotherapy with placebo plus chemotherapy (Abstract CT042).
The primary analysis had shown a statistically significant improvement in PFS for patients on atezolizumab-chemotherapy, in comparison with placebo-chemotherapy, with encouraging OS improvement, but the boundary for declaring significance for the OS endpoint was not crossed (Lancet. 2020 May 16;395[10236]:1547-1557).
Because IMvigor130 included both patients who received cisplatin and patients who investigators deemed cisplatin ineligible, the second interim analysis included an exploratory analysis of whether there was a difference in outcome between patients who received or did not receive cisplatin.
At a median follow-up of 13.3 months, the median OS was not significantly different in the atezolizumab-chemotherapy arm (n = 451) and the placebo-chemotherapy arm (n = 400) – 16.1 months and 13.4 months, respectively (HR, 0.84; 95% CI, 0.71-1.00; P = .026).
There were no clinically or pathologically defined subgroups that experienced an OS benefit from atezolizumab-chemotherapy over placebo-chemotherapy.
As for subsequent nonprotocol therapy, 24% of the placebo-chemotherapy arm received an immune checkpoint inhibitor at progression, as did 7% of the atezolizumab-chemotherapy arm. There was no difference in receipt of an immune checkpoint inhibitor post progression among patients treated with cisplatin versus carboplatin.
The benefit of combining atezolizumab with chemotherapy appeared more substantial with cisplatin-based chemotherapy than with carboplatin-based treatment. With cisplatin, the median OS was 21.6 months for the atezolizumab-chemotherapy arm and 14.6 months for the placebo-chemotherapy arm. With carboplatin, the median OS was 14.3 months and 13.0 months, respectively.
PD-L1 status was prognostic for patients who received cisplatin, with lower OS being observed for patients with PD-L1 IC0/1 status and higher OS observed for patients with PD-L1 IC2/3 status. Atezolizumab plus cisplatin-based chemotherapy appeared superior to cisplatin-based chemotherapy alone in both PD-L1–low and –high groups.
Atezolizumab did not seem to benefit patients who were treated with carboplatin, and PD-L1 status did not seem to influence OS among the carboplatin-treated patients.
Although similar ORR results were seen with cisplatin and carboplatin, there appeared to be a longer median DOR among cisplatin-treated patients who received atezolizumab than among those who did not – 13.2 months and 8.3 months, respectively.
No such benefit from atezolizumab was seen in carboplatin-treated patients. The median DOR was 8.1 months among patients who received atezolizumab and 7.1 months among those who did not.
The overall safety profile for atezolizumab plus chemotherapy was consistent with prior reports of the combination. Treatment-related grade 3-5 adverse events were similar on the atezolizumab-chemotherapy arm and the placebo-chemotherapy arm.
The present and future
The investigators who presented the second interim analysis for OS of the IMvigor130 trial were appropriately modest in their conclusions. After all, the prespecified boundary for significant improvement in OS for the addition of atezolizumab to chemotherapy was not crossed. No change in guideline-based clinical practice would be appropriate at the present time.
The various exploratory analyses are hypothesis generating and invite potential mechanistic explanations. However, given the nonrandom allocation of patients to cisplatin- or carboplatin-based chemotherapy, unrecognized variables may have influenced any appearance of a difference in OS between the regimens.
In IMvigor130, treatment was given until unacceptable toxicity or disease progression. It is uncertain whether the current National Comprehensive Cancer Network category 1 recommendation of chemotherapy induction followed by immune checkpoint inhibitor maintenance therapy will prove superior to the IMvigor130 strategy.
Clearly – and concordant with current treatment guidelines – atezolizumab monotherapy can benefit some patients, though the response rate for atezolizumab monotherapy was lower than for chemotherapy (23.4% vs. 44.1%).
As noted by the session chair, Marina Chiara Garassino, MD, of the University of Chicago, the OS curves were initially superior for chemotherapy over atezolizumab. However, the apparent early OS benefit for chemotherapy dissipated over time and, among responders to atezolizumab, response duration was considerably longer than for chemotherapy.
IMvigor130 will ultimately have a final OS analysis to clarify the relative benefits of the various treatment strategies. Fortunately, this large phase 3 study will yield a treasure trove of data to inform future research and build on the advances of recent years for patients with advanced urothelial cancer.
IMvigor130 is sponsored by Hoffmann-La Roche. Dr. Davis, Dr. Galsky, and Dr. Garassino disclosed relationships with Hoffmann-La Roche and many other companies.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
A second interim overall survival (OS) analysis suggested that atezolizumab monotherapy provides a clinical benefit as first-line treatment for mUC patients with PD-L1–expressing immune cells representing at least 5% of the tumor area (IC2/3), including patients who are cisplatin ineligible.
The analysis also suggested that atezolizumab plus chemotherapy produces similar OS results as chemotherapy plus placebo, but patients receiving atezolizumab may do better with cisplatin-based chemotherapy than with carboplatin-based chemotherapy.
These results were reported in two presentations at the American Association for Cancer Research Annual Meeting 2021: Week 1.
Current guidelines from the National Comprehensive Cancer Network and the European Society for Medical Oncology recommend atezolizumab monotherapy for cisplatin-ineligible patients with mUC and PD-L1 IC2/3.
The ongoing phase 3 IMvigor130 trial was designed to compare atezolizumab plus gemcitabine/platinum chemotherapy, atezolizumab monotherapy, and placebo plus chemotherapy. Platinum-based chemotherapy included either cisplatin or carboplatin, per investigator choice.
Coprimary endpoints for IMvigor130 were progression-free survival (PFS) and OS for atezolizumab plus chemotherapy versus placebo plus chemotherapy. The hierarchical study design dictated that OS would only be assessed for the comparison of atezolizumab monotherapy versus placebo-chemotherapy in the overall and PD-L1 IC2/3 populations if there was statistical improvement in OS for the atezolizumab-chemotherapy arm over the placebo-chemotherapy arm.
Secondary endpoints were overall response rate (ORR; per RECIST 1.1), duration of response (DOR) for all patients, and PFS for the comparison between atezolizumab monotherapy and placebo-chemotherapy. Exploratory analyses were performed on cisplatin-ineligible patients by PD-L1 status.
At the time of the primary analysis, an OS benefit for atezolizumab-chemotherapy over placebo-chemotherapy was not observed. Therefore, the OS benefit of atezolizumab monotherapy versus placebo-chemotherapy was not assessed. However, a trend toward improved OS was noted with atezolizumab for PD-L1 IC2/3 patients, including cisplatin-ineligible patients.
Atezolizumab vs. placebo-chemo
Ian D. Davis, MBBS, PhD, of Monash University in Melbourne, presented the second interim analysis of OS with atezolizumab monotherapy versus placebo plus chemotherapy (Abstract CT040).
The median follow-up was 14.9 months for atezolizumab monotherapy (n = 360) and 11.8 months for placebo-chemotherapy (n = 359). The median OS was 15.2 months and 13.1 months, respectively (hazard ratio, 0.99; 95% confidence interval, 0.83-1.19). There was no apparent OS benefit of atezolizumab for any clinically selected subgroup.
The ORR was 23.4% for atezolizumab monotherapy and 44.1% for placebo-chemotherapy. The median DOR was more than 3.5 times longer for atezolizumab monotherapy than for placebo-chemotherapy – 29.6 months and 8.1 months, respectively.
Although there was no formal statistical comparison, exploratory subgroup analyses demonstrated that the median OS for the PD-L1 IC2/3 patients appeared higher in the atezolizumab monotherapy arm than in the placebo-chemotherapy arm – 27.5 months and 16.7 months, respectively.
Similarly, the median OS for cisplatin-ineligible PD-L1 IC2/3 patients appeared higher for atezolizumab monotherapy than for placebo-chemotherapy – 18.6 months and 10.0 months, respectively.
In terms of safety, atezolizumab monotherapy compared favorably with placebo plus chemotherapy. There were similar numbers of grade 3/4 adverse events and comparable adverse events leading to discontinuation of treatment in both arms.
The atezolizumab monotherapy arm had fewer adverse events leading to withdrawal from any treatment, when compared with the placebo-chemotherapy arm – 7% and 34%, respectively. Two patients in the atezolizumab arm and one in the placebo-chemotherapy died of treatment-related causes.
Atezolizumab-chemo vs. placebo-chemo
Matthew D. Galsky, MD, of Mount Sinai Health System and Icahn School of Medicine at Mount Sinai in New York, presented the second interim OS comparison of atezolizumab plus chemotherapy with placebo plus chemotherapy (Abstract CT042).
The primary analysis had shown a statistically significant improvement in PFS for patients on atezolizumab-chemotherapy, in comparison with placebo-chemotherapy, with encouraging OS improvement, but the boundary for declaring significance for the OS endpoint was not crossed (Lancet. 2020 May 16;395[10236]:1547-1557).
Because IMvigor130 included both patients who received cisplatin and patients who investigators deemed cisplatin ineligible, the second interim analysis included an exploratory analysis of whether there was a difference in outcome between patients who received or did not receive cisplatin.
At a median follow-up of 13.3 months, the median OS was not significantly different in the atezolizumab-chemotherapy arm (n = 451) and the placebo-chemotherapy arm (n = 400) – 16.1 months and 13.4 months, respectively (HR, 0.84; 95% CI, 0.71-1.00; P = .026).
There were no clinically or pathologically defined subgroups that experienced an OS benefit from atezolizumab-chemotherapy over placebo-chemotherapy.
As for subsequent nonprotocol therapy, 24% of the placebo-chemotherapy arm received an immune checkpoint inhibitor at progression, as did 7% of the atezolizumab-chemotherapy arm. There was no difference in receipt of an immune checkpoint inhibitor post progression among patients treated with cisplatin versus carboplatin.
The benefit of combining atezolizumab with chemotherapy appeared more substantial with cisplatin-based chemotherapy than with carboplatin-based treatment. With cisplatin, the median OS was 21.6 months for the atezolizumab-chemotherapy arm and 14.6 months for the placebo-chemotherapy arm. With carboplatin, the median OS was 14.3 months and 13.0 months, respectively.
PD-L1 status was prognostic for patients who received cisplatin, with lower OS being observed for patients with PD-L1 IC0/1 status and higher OS observed for patients with PD-L1 IC2/3 status. Atezolizumab plus cisplatin-based chemotherapy appeared superior to cisplatin-based chemotherapy alone in both PD-L1–low and –high groups.
Atezolizumab did not seem to benefit patients who were treated with carboplatin, and PD-L1 status did not seem to influence OS among the carboplatin-treated patients.
Although similar ORR results were seen with cisplatin and carboplatin, there appeared to be a longer median DOR among cisplatin-treated patients who received atezolizumab than among those who did not – 13.2 months and 8.3 months, respectively.
No such benefit from atezolizumab was seen in carboplatin-treated patients. The median DOR was 8.1 months among patients who received atezolizumab and 7.1 months among those who did not.
The overall safety profile for atezolizumab plus chemotherapy was consistent with prior reports of the combination. Treatment-related grade 3-5 adverse events were similar on the atezolizumab-chemotherapy arm and the placebo-chemotherapy arm.
The present and future
The investigators who presented the second interim analysis for OS of the IMvigor130 trial were appropriately modest in their conclusions. After all, the prespecified boundary for significant improvement in OS for the addition of atezolizumab to chemotherapy was not crossed. No change in guideline-based clinical practice would be appropriate at the present time.
The various exploratory analyses are hypothesis generating and invite potential mechanistic explanations. However, given the nonrandom allocation of patients to cisplatin- or carboplatin-based chemotherapy, unrecognized variables may have influenced any appearance of a difference in OS between the regimens.
In IMvigor130, treatment was given until unacceptable toxicity or disease progression. It is uncertain whether the current National Comprehensive Cancer Network category 1 recommendation of chemotherapy induction followed by immune checkpoint inhibitor maintenance therapy will prove superior to the IMvigor130 strategy.
Clearly – and concordant with current treatment guidelines – atezolizumab monotherapy can benefit some patients, though the response rate for atezolizumab monotherapy was lower than for chemotherapy (23.4% vs. 44.1%).
As noted by the session chair, Marina Chiara Garassino, MD, of the University of Chicago, the OS curves were initially superior for chemotherapy over atezolizumab. However, the apparent early OS benefit for chemotherapy dissipated over time and, among responders to atezolizumab, response duration was considerably longer than for chemotherapy.
IMvigor130 will ultimately have a final OS analysis to clarify the relative benefits of the various treatment strategies. Fortunately, this large phase 3 study will yield a treasure trove of data to inform future research and build on the advances of recent years for patients with advanced urothelial cancer.
IMvigor130 is sponsored by Hoffmann-La Roche. Dr. Davis, Dr. Galsky, and Dr. Garassino disclosed relationships with Hoffmann-La Roche and many other companies.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
AACR 2021
How about contraceptives for men?
With the introduction of new technology to vaccinate the world with the Pfizer and Moderna mRNA vaccines, I considered other health conditions that could benefit from new modalities. Unplanned pregnancies are a public health crisis, yet the burden falls solely on women to solve, burdening them with contraceptive practices to prevent unplanned pregnancy. With the insurrection of Row v. Wade and the new bills being pushed through states that are limiting abortion, perhaps the time has come for males to accept the responsibility for contraception to prevent unplanned pregnancy. The methods that currently exist for males are condoms and vasectomy. Other options are being explored – both nonhormonal and reversible contraception including daily pills, gels, and long-acting injections.
The pill for men has been under preliminary trials with promising results. This contraceptive pill contains dimethandrolone undecanoate, which is an androgen anabolic steroid progesterone once-daily pill that suppresses FSH and LH, causing a decrease in the production of testosterone and consequently sperm production.1 (Long, Lee, & Blithe, 2019). This pill is in long-term trials to determine the efficacy and side effects, including the impact on libido, liver, and kidney disease.
The injectable male contraceptive in trials now includes two different options. The first was a long-acting progestin, testosterone, and androgen combination. The male participants received an intramuscular injection every 8 weeks. Although the results of the study were promising – sperm production was effectively reduced, the side effects were too severe for participants to continue use. Side effects much like those of the female Depo-Provera injections included acne and mood disorders. Men experienced erectile dysfunction while at the same time having an increase in sex drive.2 (Em, 2018).
Recently, researchers in India have studied a nonhormonal injectable with promising outcomes. It prevented pregnancy in more than 97% of participants. This injectable polymer gel is placed into the male’s vas deferens to block sperm from leaving the body. This product inactivates sperm, essentially creating temporary sterilization for men. The benefit of this product, called RISUG (reversible inhibition of sperm under guidance), is a single injection that can be effective for 13 years. It can be reversed earlier if needed by injecting a dissolving gel into the male’s vas deferens.1,2 In the United States, there is an identical product called Vasalgel – a polymer injected into the vas deferens – also being studied for temporary infertility.
Another synthetic implanted androgen product being studied is 7 alpha-methyl-19-nortestosterone (MENT), a synthetic steroid that resembles testosterone but does not convert into testosterone and, consequently, does not stimulate prostate growth. It is administered via two subdermal implants and is effective for 12 months. The first subdermal implant releases the synthetic androgen, which is more potent than testosterone, and the other emits LH-releasing hormone.3 Studies demonstrate that MENT suppresses sperm production.1
Finally, studies are underway using transdermal gel applications to suppress sperm concentrations. The daily gel is absorbed through the skin after application to two different areas of the man’s body: the shoulders and upper arms. The daily application of the progestin product, Nestorone, and testosterone gel has been found to reduce sperm concentrations to < 1 x 106/mL. Studies measured gonadal concentrations after 4 weeks.1 Users were happy with the use of a topical gel, with minimal side effects such as lower libido, weight gain, and changes in cholesterol, yet inconsistent use of the product resulted in lower than anticipated results.4
Male contraceptive options are long overdue to dramatically reduce the rate of unplanned pregnancies and the burden of contraception placed on women. Getting these products to market will be half the battle – getting men to commit to using these options and women to trust male compliance may further impede acceptance. Men have not had to carry the burden and economics of single parenting. Men interested in casual sex may now need to accept more responsibility for unplanned pregnancy and be proactive with prevention, particularly as abortion laws are being challenged.
Ms. Thew is medical director of the department of pediatrics division of adolescent medicine at the Medical College of Wisconsin in Milwaukee. She is a member of the editorial board for Pediatric News and has no relevant disclosures.
References
1. Long J E et al. Clin Chem. 2019;65(1):153-60.
2. Male birth control: Current options and new breakthroughs, SingleCare: Health Education. Aug. 6, 2018.
3. Sundaram K et al. Ann Med. 1993;25(2):199-205.
4. Anawalt BD et al. Andrology. 2019;7(6):878-87.
With the introduction of new technology to vaccinate the world with the Pfizer and Moderna mRNA vaccines, I considered other health conditions that could benefit from new modalities. Unplanned pregnancies are a public health crisis, yet the burden falls solely on women to solve, burdening them with contraceptive practices to prevent unplanned pregnancy. With the insurrection of Row v. Wade and the new bills being pushed through states that are limiting abortion, perhaps the time has come for males to accept the responsibility for contraception to prevent unplanned pregnancy. The methods that currently exist for males are condoms and vasectomy. Other options are being explored – both nonhormonal and reversible contraception including daily pills, gels, and long-acting injections.
The pill for men has been under preliminary trials with promising results. This contraceptive pill contains dimethandrolone undecanoate, which is an androgen anabolic steroid progesterone once-daily pill that suppresses FSH and LH, causing a decrease in the production of testosterone and consequently sperm production.1 (Long, Lee, & Blithe, 2019). This pill is in long-term trials to determine the efficacy and side effects, including the impact on libido, liver, and kidney disease.
The injectable male contraceptive in trials now includes two different options. The first was a long-acting progestin, testosterone, and androgen combination. The male participants received an intramuscular injection every 8 weeks. Although the results of the study were promising – sperm production was effectively reduced, the side effects were too severe for participants to continue use. Side effects much like those of the female Depo-Provera injections included acne and mood disorders. Men experienced erectile dysfunction while at the same time having an increase in sex drive.2 (Em, 2018).
Recently, researchers in India have studied a nonhormonal injectable with promising outcomes. It prevented pregnancy in more than 97% of participants. This injectable polymer gel is placed into the male’s vas deferens to block sperm from leaving the body. This product inactivates sperm, essentially creating temporary sterilization for men. The benefit of this product, called RISUG (reversible inhibition of sperm under guidance), is a single injection that can be effective for 13 years. It can be reversed earlier if needed by injecting a dissolving gel into the male’s vas deferens.1,2 In the United States, there is an identical product called Vasalgel – a polymer injected into the vas deferens – also being studied for temporary infertility.
Another synthetic implanted androgen product being studied is 7 alpha-methyl-19-nortestosterone (MENT), a synthetic steroid that resembles testosterone but does not convert into testosterone and, consequently, does not stimulate prostate growth. It is administered via two subdermal implants and is effective for 12 months. The first subdermal implant releases the synthetic androgen, which is more potent than testosterone, and the other emits LH-releasing hormone.3 Studies demonstrate that MENT suppresses sperm production.1
Finally, studies are underway using transdermal gel applications to suppress sperm concentrations. The daily gel is absorbed through the skin after application to two different areas of the man’s body: the shoulders and upper arms. The daily application of the progestin product, Nestorone, and testosterone gel has been found to reduce sperm concentrations to < 1 x 106/mL. Studies measured gonadal concentrations after 4 weeks.1 Users were happy with the use of a topical gel, with minimal side effects such as lower libido, weight gain, and changes in cholesterol, yet inconsistent use of the product resulted in lower than anticipated results.4
Male contraceptive options are long overdue to dramatically reduce the rate of unplanned pregnancies and the burden of contraception placed on women. Getting these products to market will be half the battle – getting men to commit to using these options and women to trust male compliance may further impede acceptance. Men have not had to carry the burden and economics of single parenting. Men interested in casual sex may now need to accept more responsibility for unplanned pregnancy and be proactive with prevention, particularly as abortion laws are being challenged.
Ms. Thew is medical director of the department of pediatrics division of adolescent medicine at the Medical College of Wisconsin in Milwaukee. She is a member of the editorial board for Pediatric News and has no relevant disclosures.
References
1. Long J E et al. Clin Chem. 2019;65(1):153-60.
2. Male birth control: Current options and new breakthroughs, SingleCare: Health Education. Aug. 6, 2018.
3. Sundaram K et al. Ann Med. 1993;25(2):199-205.
4. Anawalt BD et al. Andrology. 2019;7(6):878-87.
With the introduction of new technology to vaccinate the world with the Pfizer and Moderna mRNA vaccines, I considered other health conditions that could benefit from new modalities. Unplanned pregnancies are a public health crisis, yet the burden falls solely on women to solve, burdening them with contraceptive practices to prevent unplanned pregnancy. With the insurrection of Row v. Wade and the new bills being pushed through states that are limiting abortion, perhaps the time has come for males to accept the responsibility for contraception to prevent unplanned pregnancy. The methods that currently exist for males are condoms and vasectomy. Other options are being explored – both nonhormonal and reversible contraception including daily pills, gels, and long-acting injections.
The pill for men has been under preliminary trials with promising results. This contraceptive pill contains dimethandrolone undecanoate, which is an androgen anabolic steroid progesterone once-daily pill that suppresses FSH and LH, causing a decrease in the production of testosterone and consequently sperm production.1 (Long, Lee, & Blithe, 2019). This pill is in long-term trials to determine the efficacy and side effects, including the impact on libido, liver, and kidney disease.
The injectable male contraceptive in trials now includes two different options. The first was a long-acting progestin, testosterone, and androgen combination. The male participants received an intramuscular injection every 8 weeks. Although the results of the study were promising – sperm production was effectively reduced, the side effects were too severe for participants to continue use. Side effects much like those of the female Depo-Provera injections included acne and mood disorders. Men experienced erectile dysfunction while at the same time having an increase in sex drive.2 (Em, 2018).
Recently, researchers in India have studied a nonhormonal injectable with promising outcomes. It prevented pregnancy in more than 97% of participants. This injectable polymer gel is placed into the male’s vas deferens to block sperm from leaving the body. This product inactivates sperm, essentially creating temporary sterilization for men. The benefit of this product, called RISUG (reversible inhibition of sperm under guidance), is a single injection that can be effective for 13 years. It can be reversed earlier if needed by injecting a dissolving gel into the male’s vas deferens.1,2 In the United States, there is an identical product called Vasalgel – a polymer injected into the vas deferens – also being studied for temporary infertility.
Another synthetic implanted androgen product being studied is 7 alpha-methyl-19-nortestosterone (MENT), a synthetic steroid that resembles testosterone but does not convert into testosterone and, consequently, does not stimulate prostate growth. It is administered via two subdermal implants and is effective for 12 months. The first subdermal implant releases the synthetic androgen, which is more potent than testosterone, and the other emits LH-releasing hormone.3 Studies demonstrate that MENT suppresses sperm production.1
Finally, studies are underway using transdermal gel applications to suppress sperm concentrations. The daily gel is absorbed through the skin after application to two different areas of the man’s body: the shoulders and upper arms. The daily application of the progestin product, Nestorone, and testosterone gel has been found to reduce sperm concentrations to < 1 x 106/mL. Studies measured gonadal concentrations after 4 weeks.1 Users were happy with the use of a topical gel, with minimal side effects such as lower libido, weight gain, and changes in cholesterol, yet inconsistent use of the product resulted in lower than anticipated results.4
Male contraceptive options are long overdue to dramatically reduce the rate of unplanned pregnancies and the burden of contraception placed on women. Getting these products to market will be half the battle – getting men to commit to using these options and women to trust male compliance may further impede acceptance. Men have not had to carry the burden and economics of single parenting. Men interested in casual sex may now need to accept more responsibility for unplanned pregnancy and be proactive with prevention, particularly as abortion laws are being challenged.
Ms. Thew is medical director of the department of pediatrics division of adolescent medicine at the Medical College of Wisconsin in Milwaukee. She is a member of the editorial board for Pediatric News and has no relevant disclosures.
References
1. Long J E et al. Clin Chem. 2019;65(1):153-60.
2. Male birth control: Current options and new breakthroughs, SingleCare: Health Education. Aug. 6, 2018.
3. Sundaram K et al. Ann Med. 1993;25(2):199-205.
4. Anawalt BD et al. Andrology. 2019;7(6):878-87.
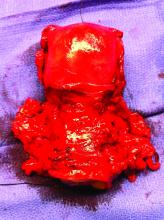
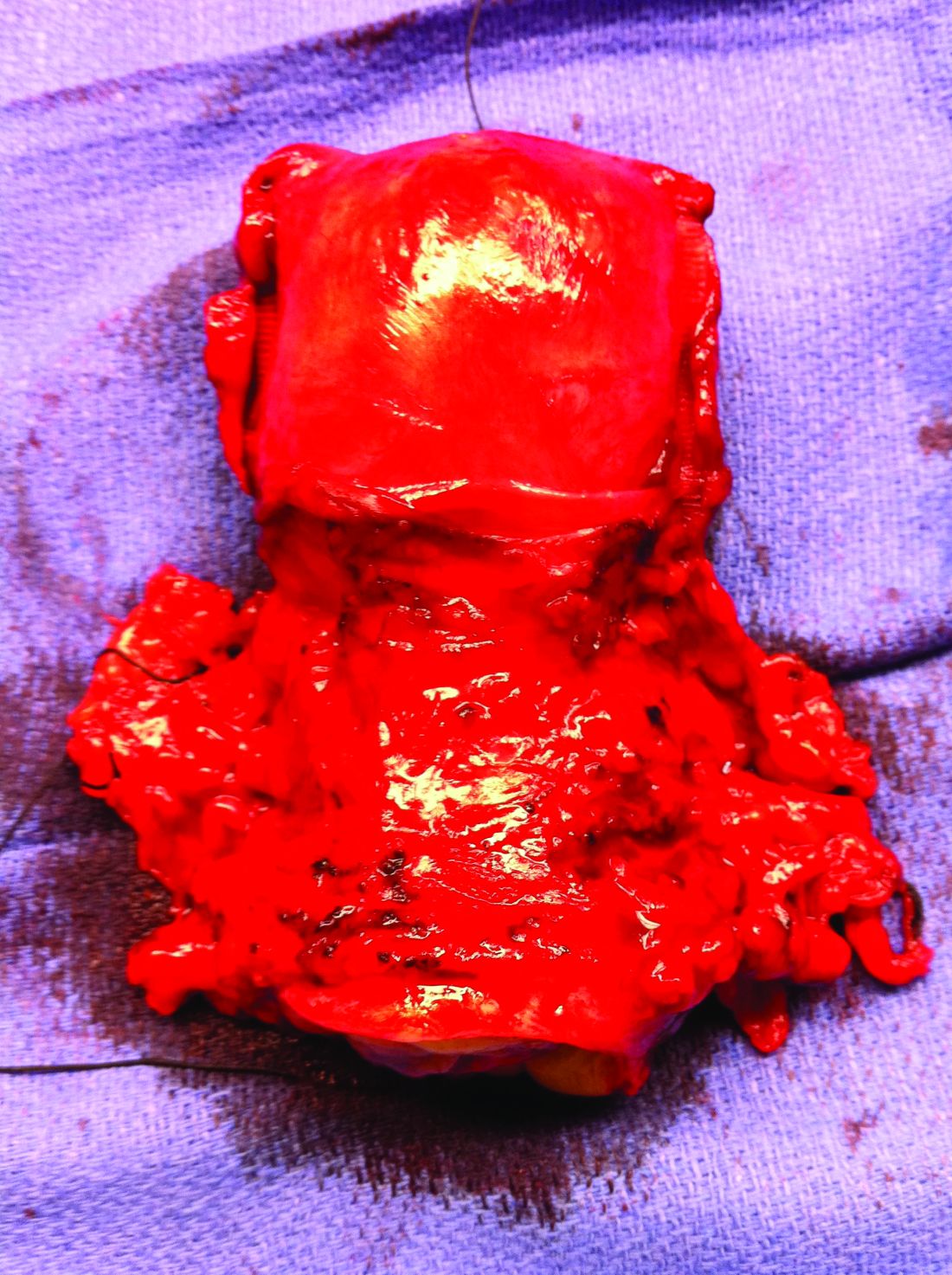
Surgery for early-stage cervical cancer: Are we still too radical?
It has been more than 120 years since Ernst Wertheim, a Viennese surgeon, performed and described what is considered to have been the first radical total hysterectomy with lymphadenectomy for early-stage cervical cancer, yet this morbid procedure remains the standard of care for most early-stage cervical cancers. The rationale for this procedure, which included removal of the parametrial tissue, uterosacral and cardinal ligaments, and upper vagina en bloc with the cervix and uterus, was to obtain margins around a cancer that has a dominant radial growth pattern. The morbidity associated with this procedure is substantial. The parametrium houses important vascular, neural, and urologic structures. Unlike extrafascial hysterectomy, often referred to as “simple” hysterectomy, in which surgeons follow a fascial plane, and therefore a relatively avascular dissection, surgeons performing radical hysterectomy must venture outside of these embryologic fusion planes into less well–defined anatomy. Therefore, surgical complications are relatively common including hemorrhage, ureteral and bladder injury, as well as late-onset devastating complications such as fistula, urinary retention, or incontinence, and sexual dysfunction.1 More recently, variations of the Wertheim-Meigs radical hysterectomy have been described, and objective classifications created, which include modified radical procedures (removing less parametria) and nerve-sparing procedures to facilitate standardized nomenclature for tailoring the most appropriate procedure for any given tumor.2
The trend, and a positive one at that, over the course of the past century, has been a move away from routine radical surgical procedures for most clinical stage 1 cancers. No better example exists than breast cancer, in which the Halsted radical mastectomy has been largely replaced by less morbid breast-conserving or nonradical procedures with adjunct medical and radiation therapies offered to achieve high rates of cure with far more acceptable patient-centered outcomes.3 And so why is it that radical hysterectomy is still considered the standard of care for all but the smallest of microscopic cervical cancers?
The risk of lymph node metastases or recurrence is exceptionally low for women with microscopic (stage IA1) cervical cancers that are less than 3 mm in depth. Therefore, the National Comprehensive Cancer Network guidelines recommend nonradical surgical remedies (such as extrafascial hysterectomy, or cone biopsy or trachelectomy if fertility preservation is desired) for this earlier stage of disease.4 If there is lymphovascular space invasion (an indicator of poor prognosis and potential lymphatic involvement), a lymphadenectomy or sentinel lymph node biopsy is also recommended. For women with stage IA2 or IB lesions, radical excisions (either trachelectomy or hysterectomy) are considered the standard of care. However, this “gold standard” was achieved largely through legacy, and not a result of randomized trials comparing its outcomes with nonradical procedures.
Initial strides away from radical cervical cancer surgery focused on the goal of fertility preservation via radical trachelectomy which allowed women to preserve an intact uterine fundus. This was initially met with skepticism and concern that surgeons could be sacrificing oncologic outcomes in order to preserve a woman’s fertility. Thanks to pioneering work, including prospective research studies by surgeon innovators it has been shown that, in appropriately selected candidates with tumors less than 2 cm, it is an accepted standard of care.4 Radical vaginal or abdominal trachelectomy is associated with cancer recurrence rates of less than 5% and successful pregnancy in approximately three-quarters of patients in whom this is desired.5,6 However, full-term pregnancy is achieved in 50%-75% of cases, reflecting increased obstetric risk, and radical trachelectomy still subjects patients to the morbidity of a radical parametrial resection, despite the fact that many of them will have no residual carcinoma in their final pathological specimens.
Therefore, can we be even more conservative in our surgery for these patients? Are simple hysterectomy or conization potentially adequate treatments for small (<2 cm) stage IA2 and IB1 lesions that have favorable histology (<10 mm stromal invasion, low-risk histology, no lymphovascular space involvement, negative margins on conization and no lymph node metastases)? In patients whose tumor exhibits these histologic features, the likelihood of parametrial involvement is approximately 1%, calling into question the virtue of parametrial resection.7 Observational studies have identified mixed results on the safety of conservative surgical techniques in early-stage cervical cancer. In a study of the National Cancer Database, the outcomes of 2,543 radical hysterectomies and 1,388 extrafascial hysterectomies for women with stage IB1 disease were evaluated and observed a difference in 5-year survival (92.4% vs. 95.3%) favoring the radical procedure.8 Unfortunately, database analyses such as these are limited by potential confounders and discordance between the groups such as rates of lymphadenectomy, known involvement of oncologic surgeon specialists, and margin status. An alternative evaluation of the Surveillance, Epidemiology, and End Results database including 2,571 patients with stage IB1 disease, all of whom had lymphadenectomy performed, showed no difference in 10-year disease-specific survival between the two surgical approaches.9
Ultimately, whether conservative procedures (such as conization or extrafascial hysterectomy) can be offered to women with small, low-risk IB1 or IA2 cervical cancers will be best determined by prospective single-arm or randomized trials. Fortunately, these are underway. Preliminary results from the ConCerv trial in which 100 women with early-stage, low-risk stage IA2 and IB1 cervical cancer were treated with either repeat conization or extrafascial hysterectomy with sentinel lymph node biopsy showed acceptably low rates of recurrence (3%) with this approach.10 If the mature data supports this finding, it seems that, for appropriately selected and well-counseled patients, conservative surgery may become more broadly accepted as a reasonable option for treatment that spares women not only loss of fertility, but also the early and late surgical morbidity from radical procedures.
In the meantime, until more is known about the oncologic safety of nonradical procedures for stage IA2 and IB1 cervical cancer, this option should not be considered standard of care, and only offered to patients with favorable tumor factors who are well counseled regarding the uncertainty of this approach. It is critical that patients with early-stage cervical cancer be evaluated by a gynecologic cancer specialist prior to definitive surgical treatment as they are best equipped to evaluate risk profiles and counsel about her options for surgery, its known and unknown consequences, and the appropriateness of fertility preservation or radicality of surgery. We eagerly await the results of trials evaluating the safety of conservative cervical cancer surgery, which promise to advance us from 19th-century practices, preserving not only fertility, but also quality of life.
Dr. Rossi is assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill. She has no disclosures and can be contacted at obnews@mdedge.com.
References
1. Trimbos JB et al. Eur J Cancer. 2004;40(3):375-8.
2. Querleu D and Morrow CP. Lancet Oncol. 2008;9:297-303.
3. Sakorafas GH and Safioleas M. Eur J Cancer Care. 2010 Mar;19(2):145-66.
4. National Comprehensive Cancer Network. Cervical Cancer (Version 1.2021). https://www.nccn.org/professionals/physician_gls/pdf/cervical.pdf. Accessed 2021 Apr 21.
5. Plante M et al. Gynecol Oncol. 2011;121:290-7.
6. Wethington SL et al. Int J Gynecol Cancer. 2012;22:1251-7.
7. Domgue J and Schmeler K. Best Pract Res Clin Obstet Gynaecol. 2019 Feb;55:79-92.
8. Sia TY et al. Obstet Gyenecol. 2019;134(6):1132.
9. Tseng J et al. Gynecol Oncol. 2018;150(1):44.
10. Schmeler K et al. Int J Gynecol Cancer. 2019;29:A14-5.
It has been more than 120 years since Ernst Wertheim, a Viennese surgeon, performed and described what is considered to have been the first radical total hysterectomy with lymphadenectomy for early-stage cervical cancer, yet this morbid procedure remains the standard of care for most early-stage cervical cancers. The rationale for this procedure, which included removal of the parametrial tissue, uterosacral and cardinal ligaments, and upper vagina en bloc with the cervix and uterus, was to obtain margins around a cancer that has a dominant radial growth pattern. The morbidity associated with this procedure is substantial. The parametrium houses important vascular, neural, and urologic structures. Unlike extrafascial hysterectomy, often referred to as “simple” hysterectomy, in which surgeons follow a fascial plane, and therefore a relatively avascular dissection, surgeons performing radical hysterectomy must venture outside of these embryologic fusion planes into less well–defined anatomy. Therefore, surgical complications are relatively common including hemorrhage, ureteral and bladder injury, as well as late-onset devastating complications such as fistula, urinary retention, or incontinence, and sexual dysfunction.1 More recently, variations of the Wertheim-Meigs radical hysterectomy have been described, and objective classifications created, which include modified radical procedures (removing less parametria) and nerve-sparing procedures to facilitate standardized nomenclature for tailoring the most appropriate procedure for any given tumor.2
The trend, and a positive one at that, over the course of the past century, has been a move away from routine radical surgical procedures for most clinical stage 1 cancers. No better example exists than breast cancer, in which the Halsted radical mastectomy has been largely replaced by less morbid breast-conserving or nonradical procedures with adjunct medical and radiation therapies offered to achieve high rates of cure with far more acceptable patient-centered outcomes.3 And so why is it that radical hysterectomy is still considered the standard of care for all but the smallest of microscopic cervical cancers?
The risk of lymph node metastases or recurrence is exceptionally low for women with microscopic (stage IA1) cervical cancers that are less than 3 mm in depth. Therefore, the National Comprehensive Cancer Network guidelines recommend nonradical surgical remedies (such as extrafascial hysterectomy, or cone biopsy or trachelectomy if fertility preservation is desired) for this earlier stage of disease.4 If there is lymphovascular space invasion (an indicator of poor prognosis and potential lymphatic involvement), a lymphadenectomy or sentinel lymph node biopsy is also recommended. For women with stage IA2 or IB lesions, radical excisions (either trachelectomy or hysterectomy) are considered the standard of care. However, this “gold standard” was achieved largely through legacy, and not a result of randomized trials comparing its outcomes with nonradical procedures.
Initial strides away from radical cervical cancer surgery focused on the goal of fertility preservation via radical trachelectomy which allowed women to preserve an intact uterine fundus. This was initially met with skepticism and concern that surgeons could be sacrificing oncologic outcomes in order to preserve a woman’s fertility. Thanks to pioneering work, including prospective research studies by surgeon innovators it has been shown that, in appropriately selected candidates with tumors less than 2 cm, it is an accepted standard of care.4 Radical vaginal or abdominal trachelectomy is associated with cancer recurrence rates of less than 5% and successful pregnancy in approximately three-quarters of patients in whom this is desired.5,6 However, full-term pregnancy is achieved in 50%-75% of cases, reflecting increased obstetric risk, and radical trachelectomy still subjects patients to the morbidity of a radical parametrial resection, despite the fact that many of them will have no residual carcinoma in their final pathological specimens.
Therefore, can we be even more conservative in our surgery for these patients? Are simple hysterectomy or conization potentially adequate treatments for small (<2 cm) stage IA2 and IB1 lesions that have favorable histology (<10 mm stromal invasion, low-risk histology, no lymphovascular space involvement, negative margins on conization and no lymph node metastases)? In patients whose tumor exhibits these histologic features, the likelihood of parametrial involvement is approximately 1%, calling into question the virtue of parametrial resection.7 Observational studies have identified mixed results on the safety of conservative surgical techniques in early-stage cervical cancer. In a study of the National Cancer Database, the outcomes of 2,543 radical hysterectomies and 1,388 extrafascial hysterectomies for women with stage IB1 disease were evaluated and observed a difference in 5-year survival (92.4% vs. 95.3%) favoring the radical procedure.8 Unfortunately, database analyses such as these are limited by potential confounders and discordance between the groups such as rates of lymphadenectomy, known involvement of oncologic surgeon specialists, and margin status. An alternative evaluation of the Surveillance, Epidemiology, and End Results database including 2,571 patients with stage IB1 disease, all of whom had lymphadenectomy performed, showed no difference in 10-year disease-specific survival between the two surgical approaches.9
Ultimately, whether conservative procedures (such as conization or extrafascial hysterectomy) can be offered to women with small, low-risk IB1 or IA2 cervical cancers will be best determined by prospective single-arm or randomized trials. Fortunately, these are underway. Preliminary results from the ConCerv trial in which 100 women with early-stage, low-risk stage IA2 and IB1 cervical cancer were treated with either repeat conization or extrafascial hysterectomy with sentinel lymph node biopsy showed acceptably low rates of recurrence (3%) with this approach.10 If the mature data supports this finding, it seems that, for appropriately selected and well-counseled patients, conservative surgery may become more broadly accepted as a reasonable option for treatment that spares women not only loss of fertility, but also the early and late surgical morbidity from radical procedures.
In the meantime, until more is known about the oncologic safety of nonradical procedures for stage IA2 and IB1 cervical cancer, this option should not be considered standard of care, and only offered to patients with favorable tumor factors who are well counseled regarding the uncertainty of this approach. It is critical that patients with early-stage cervical cancer be evaluated by a gynecologic cancer specialist prior to definitive surgical treatment as they are best equipped to evaluate risk profiles and counsel about her options for surgery, its known and unknown consequences, and the appropriateness of fertility preservation or radicality of surgery. We eagerly await the results of trials evaluating the safety of conservative cervical cancer surgery, which promise to advance us from 19th-century practices, preserving not only fertility, but also quality of life.
Dr. Rossi is assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill. She has no disclosures and can be contacted at obnews@mdedge.com.
References
1. Trimbos JB et al. Eur J Cancer. 2004;40(3):375-8.
2. Querleu D and Morrow CP. Lancet Oncol. 2008;9:297-303.
3. Sakorafas GH and Safioleas M. Eur J Cancer Care. 2010 Mar;19(2):145-66.
4. National Comprehensive Cancer Network. Cervical Cancer (Version 1.2021). https://www.nccn.org/professionals/physician_gls/pdf/cervical.pdf. Accessed 2021 Apr 21.
5. Plante M et al. Gynecol Oncol. 2011;121:290-7.
6. Wethington SL et al. Int J Gynecol Cancer. 2012;22:1251-7.
7. Domgue J and Schmeler K. Best Pract Res Clin Obstet Gynaecol. 2019 Feb;55:79-92.
8. Sia TY et al. Obstet Gyenecol. 2019;134(6):1132.
9. Tseng J et al. Gynecol Oncol. 2018;150(1):44.
10. Schmeler K et al. Int J Gynecol Cancer. 2019;29:A14-5.
It has been more than 120 years since Ernst Wertheim, a Viennese surgeon, performed and described what is considered to have been the first radical total hysterectomy with lymphadenectomy for early-stage cervical cancer, yet this morbid procedure remains the standard of care for most early-stage cervical cancers. The rationale for this procedure, which included removal of the parametrial tissue, uterosacral and cardinal ligaments, and upper vagina en bloc with the cervix and uterus, was to obtain margins around a cancer that has a dominant radial growth pattern. The morbidity associated with this procedure is substantial. The parametrium houses important vascular, neural, and urologic structures. Unlike extrafascial hysterectomy, often referred to as “simple” hysterectomy, in which surgeons follow a fascial plane, and therefore a relatively avascular dissection, surgeons performing radical hysterectomy must venture outside of these embryologic fusion planes into less well–defined anatomy. Therefore, surgical complications are relatively common including hemorrhage, ureteral and bladder injury, as well as late-onset devastating complications such as fistula, urinary retention, or incontinence, and sexual dysfunction.1 More recently, variations of the Wertheim-Meigs radical hysterectomy have been described, and objective classifications created, which include modified radical procedures (removing less parametria) and nerve-sparing procedures to facilitate standardized nomenclature for tailoring the most appropriate procedure for any given tumor.2
The trend, and a positive one at that, over the course of the past century, has been a move away from routine radical surgical procedures for most clinical stage 1 cancers. No better example exists than breast cancer, in which the Halsted radical mastectomy has been largely replaced by less morbid breast-conserving or nonradical procedures with adjunct medical and radiation therapies offered to achieve high rates of cure with far more acceptable patient-centered outcomes.3 And so why is it that radical hysterectomy is still considered the standard of care for all but the smallest of microscopic cervical cancers?
The risk of lymph node metastases or recurrence is exceptionally low for women with microscopic (stage IA1) cervical cancers that are less than 3 mm in depth. Therefore, the National Comprehensive Cancer Network guidelines recommend nonradical surgical remedies (such as extrafascial hysterectomy, or cone biopsy or trachelectomy if fertility preservation is desired) for this earlier stage of disease.4 If there is lymphovascular space invasion (an indicator of poor prognosis and potential lymphatic involvement), a lymphadenectomy or sentinel lymph node biopsy is also recommended. For women with stage IA2 or IB lesions, radical excisions (either trachelectomy or hysterectomy) are considered the standard of care. However, this “gold standard” was achieved largely through legacy, and not a result of randomized trials comparing its outcomes with nonradical procedures.
Initial strides away from radical cervical cancer surgery focused on the goal of fertility preservation via radical trachelectomy which allowed women to preserve an intact uterine fundus. This was initially met with skepticism and concern that surgeons could be sacrificing oncologic outcomes in order to preserve a woman’s fertility. Thanks to pioneering work, including prospective research studies by surgeon innovators it has been shown that, in appropriately selected candidates with tumors less than 2 cm, it is an accepted standard of care.4 Radical vaginal or abdominal trachelectomy is associated with cancer recurrence rates of less than 5% and successful pregnancy in approximately three-quarters of patients in whom this is desired.5,6 However, full-term pregnancy is achieved in 50%-75% of cases, reflecting increased obstetric risk, and radical trachelectomy still subjects patients to the morbidity of a radical parametrial resection, despite the fact that many of them will have no residual carcinoma in their final pathological specimens.
Therefore, can we be even more conservative in our surgery for these patients? Are simple hysterectomy or conization potentially adequate treatments for small (<2 cm) stage IA2 and IB1 lesions that have favorable histology (<10 mm stromal invasion, low-risk histology, no lymphovascular space involvement, negative margins on conization and no lymph node metastases)? In patients whose tumor exhibits these histologic features, the likelihood of parametrial involvement is approximately 1%, calling into question the virtue of parametrial resection.7 Observational studies have identified mixed results on the safety of conservative surgical techniques in early-stage cervical cancer. In a study of the National Cancer Database, the outcomes of 2,543 radical hysterectomies and 1,388 extrafascial hysterectomies for women with stage IB1 disease were evaluated and observed a difference in 5-year survival (92.4% vs. 95.3%) favoring the radical procedure.8 Unfortunately, database analyses such as these are limited by potential confounders and discordance between the groups such as rates of lymphadenectomy, known involvement of oncologic surgeon specialists, and margin status. An alternative evaluation of the Surveillance, Epidemiology, and End Results database including 2,571 patients with stage IB1 disease, all of whom had lymphadenectomy performed, showed no difference in 10-year disease-specific survival between the two surgical approaches.9
Ultimately, whether conservative procedures (such as conization or extrafascial hysterectomy) can be offered to women with small, low-risk IB1 or IA2 cervical cancers will be best determined by prospective single-arm or randomized trials. Fortunately, these are underway. Preliminary results from the ConCerv trial in which 100 women with early-stage, low-risk stage IA2 and IB1 cervical cancer were treated with either repeat conization or extrafascial hysterectomy with sentinel lymph node biopsy showed acceptably low rates of recurrence (3%) with this approach.10 If the mature data supports this finding, it seems that, for appropriately selected and well-counseled patients, conservative surgery may become more broadly accepted as a reasonable option for treatment that spares women not only loss of fertility, but also the early and late surgical morbidity from radical procedures.
In the meantime, until more is known about the oncologic safety of nonradical procedures for stage IA2 and IB1 cervical cancer, this option should not be considered standard of care, and only offered to patients with favorable tumor factors who are well counseled regarding the uncertainty of this approach. It is critical that patients with early-stage cervical cancer be evaluated by a gynecologic cancer specialist prior to definitive surgical treatment as they are best equipped to evaluate risk profiles and counsel about her options for surgery, its known and unknown consequences, and the appropriateness of fertility preservation or radicality of surgery. We eagerly await the results of trials evaluating the safety of conservative cervical cancer surgery, which promise to advance us from 19th-century practices, preserving not only fertility, but also quality of life.
Dr. Rossi is assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill. She has no disclosures and can be contacted at obnews@mdedge.com.
References
1. Trimbos JB et al. Eur J Cancer. 2004;40(3):375-8.
2. Querleu D and Morrow CP. Lancet Oncol. 2008;9:297-303.
3. Sakorafas GH and Safioleas M. Eur J Cancer Care. 2010 Mar;19(2):145-66.
4. National Comprehensive Cancer Network. Cervical Cancer (Version 1.2021). https://www.nccn.org/professionals/physician_gls/pdf/cervical.pdf. Accessed 2021 Apr 21.
5. Plante M et al. Gynecol Oncol. 2011;121:290-7.
6. Wethington SL et al. Int J Gynecol Cancer. 2012;22:1251-7.
7. Domgue J and Schmeler K. Best Pract Res Clin Obstet Gynaecol. 2019 Feb;55:79-92.
8. Sia TY et al. Obstet Gyenecol. 2019;134(6):1132.
9. Tseng J et al. Gynecol Oncol. 2018;150(1):44.
10. Schmeler K et al. Int J Gynecol Cancer. 2019;29:A14-5.
Pros and cons of proposed recommendation for prediabetes and T2D screening
. If accepted as written, the new recommendation will be to “screen all asymptomatic adults ages 35 to 70 years who are overweight or obese.” Upon diagnosis of prediabetes, the recommendation is to offer or refer patients to preventive interventions.
This new recommendation would replace the one from 2015, which recommended screening adults aged 40-70 who are overweight or obese, lowering the age at which screening begins by 5 years. It would also replace the recommendation of referral to intensive behavioral counseling to promote a healthy diet and exercise.1
The American Diabetes Association (ADA) identifies A1c, fasting plasma glucose, or oral glucose tolerance tests as appropriate tests for the diagnosis of prediabetes and type 2 DM, and the new draft recommendation does not provide a preference for method of screening.2
The USPSTF’s draft recommendation could expand screening with the hope of identifying patients with prediabetes, or those with diabetes who are asymptomatic, with the intent of beginning treatment before there are serious complications.
Unknown diabetes or prediabetes diagnosis common
It has been estimated by the Centers for Disease Control and Prevention that 12% of U.S. adults had DM as of 2015, though nearly 24% were not aware that they had it. Also, according to the CDC, the prevalence of DM increases with age and is higher in those with less than a high school education. The same report indicates that more than 30% of U.S. adults have prediabetes, and with less than 12% of those individuals are aware of it.3 A possible explanation for a patient’s being unaware of a diagnosis could be that it has been documented in a chart but the patient does not know such information is in his or her health record. According to the evidence provided for the updated recommendation, earlier diagnosis may have an important benefit in preventing serious complications.
A modeling study compared simulated screening strategies and found that the most optimal screening strategy from a cost-effectiveness perspective begins between the ages of 30 and 45, with rescreening every 3-5 years. Further models have led researchers to conclude that early diagnosis can lead to decreased cardiovascular events as well as an opportunity for multifactorial treatment.1 For this reason, it makes sense to expand the ages of screening for obese and overweight individuals.
Treatment recommendations are more flexible
The change in treatment recommendations for a new diagnosis of prediabetes is potentially more useful. It may not be feasible or reasonable for physicians to always provide or refer their patients for intensive behavior interventions. The updated recommendation would allow for the inclusion of not only behavioral counseling and health education, but also potential medication options that are currently available but not approved, or that may be available in the future. The evidence review seemed to be mixed in outcome in this area, so the increased flexibility will likely allow for future opportunities.
Screening criteria may be too narrow
This recommendation, does not, however, provide any guidance on screening of individuals who have other risk factors besides a body mass index consistent with overweight or obesity. It seems that this may be a missed opportunity.
The draft statement clearly indicates that there are other factors associated with increased risk of developing DM, but does not consider these factors in determining which patients should be screened. Both the ADA and the American Association of Clinical Endocrinology (AACE) have recommendations for universal screening for all adults 45 and older, acknowledging that incidence of DM increases with age. The ADA also recommends screening individuals who are overweight or obese and have an additional risk factor regardless of age. The AACE recommends screening all individuals for risk factors regardless of age.
The current and draft recommendations by the USPSTF do not address other risk factors and indicate only that further research is needed to understand the risk associated with DM and the natural history of pre-DM and who may progress to DM or revert to normoglycemia. Without comment on other risk factors or universal screening with age, the USPSTF recommendation potentially would not be sensitive enough to capture all those who may meet criteria for prediabetes or DM.2,4
In addition to not addressing other risk factors and screening for those of normal and underweight BMI, the USPSTF recommendation does not address frequency of screening. The recommendations from both the ADA and the AACE indicate screening at 3-year intervals for those who are eligible – for any reason. The supporting evidence review did not seem to address this aspect, and so it is understandable that there was no comment. However, I feel this will lead physicians to turn to the other guidelines for guidance where there is disagreement in other aspects.
Ultimately, the draft updated recommendation will provide physicians with the opportunity to identify more patients with prediabetes and DM. This will be wonderful in terms of being able to offer treatments and lifestyle interventions to decrease the morbidity patients would face were these conditions not diagnosed. I hope that future recommendations will also address risk factors in addition to BMI as well as frequency of screening for those who remain at increased risk but initially screen negative.
Dr. Wheat is a family physician at Erie Family Health Center in Chicago. She is program director of Northwestern’s McGaw Family Medicine residency program at Humboldt Park, Chicago. Dr. Wheat serves on the editorial advisory board of Family Practice News. You can contact her at fpnews@mdedge.com.
References
1. Screening for prediabetes and type 2 diabetes mellitus. U.S. Preventive Services Task Force. 2021 Mar 16.
2. Classification and diagnosis of diabetes: Standards of medical care in diabetes – 2020. American Diabetes Association. Diabetes Care. 2020 Jan. doi: 10.2337/dc20-S002.
3. National Diabetes Statistics Report, 2020. Centers for Disease Control and Prevention.
4. American Association of Clinical Endocrinologists and American College of Endocrinology – clinical practice guidelines for developing a diabetes mellitus comprehensive care plan. Hadelsman Y et al. Endocr Pract. 2015 Apr. 1-87. doi: 10.4158/EP15672.GL.
. If accepted as written, the new recommendation will be to “screen all asymptomatic adults ages 35 to 70 years who are overweight or obese.” Upon diagnosis of prediabetes, the recommendation is to offer or refer patients to preventive interventions.
This new recommendation would replace the one from 2015, which recommended screening adults aged 40-70 who are overweight or obese, lowering the age at which screening begins by 5 years. It would also replace the recommendation of referral to intensive behavioral counseling to promote a healthy diet and exercise.1
The American Diabetes Association (ADA) identifies A1c, fasting plasma glucose, or oral glucose tolerance tests as appropriate tests for the diagnosis of prediabetes and type 2 DM, and the new draft recommendation does not provide a preference for method of screening.2
The USPSTF’s draft recommendation could expand screening with the hope of identifying patients with prediabetes, or those with diabetes who are asymptomatic, with the intent of beginning treatment before there are serious complications.
Unknown diabetes or prediabetes diagnosis common
It has been estimated by the Centers for Disease Control and Prevention that 12% of U.S. adults had DM as of 2015, though nearly 24% were not aware that they had it. Also, according to the CDC, the prevalence of DM increases with age and is higher in those with less than a high school education. The same report indicates that more than 30% of U.S. adults have prediabetes, and with less than 12% of those individuals are aware of it.3 A possible explanation for a patient’s being unaware of a diagnosis could be that it has been documented in a chart but the patient does not know such information is in his or her health record. According to the evidence provided for the updated recommendation, earlier diagnosis may have an important benefit in preventing serious complications.
A modeling study compared simulated screening strategies and found that the most optimal screening strategy from a cost-effectiveness perspective begins between the ages of 30 and 45, with rescreening every 3-5 years. Further models have led researchers to conclude that early diagnosis can lead to decreased cardiovascular events as well as an opportunity for multifactorial treatment.1 For this reason, it makes sense to expand the ages of screening for obese and overweight individuals.
Treatment recommendations are more flexible
The change in treatment recommendations for a new diagnosis of prediabetes is potentially more useful. It may not be feasible or reasonable for physicians to always provide or refer their patients for intensive behavior interventions. The updated recommendation would allow for the inclusion of not only behavioral counseling and health education, but also potential medication options that are currently available but not approved, or that may be available in the future. The evidence review seemed to be mixed in outcome in this area, so the increased flexibility will likely allow for future opportunities.
Screening criteria may be too narrow
This recommendation, does not, however, provide any guidance on screening of individuals who have other risk factors besides a body mass index consistent with overweight or obesity. It seems that this may be a missed opportunity.
The draft statement clearly indicates that there are other factors associated with increased risk of developing DM, but does not consider these factors in determining which patients should be screened. Both the ADA and the American Association of Clinical Endocrinology (AACE) have recommendations for universal screening for all adults 45 and older, acknowledging that incidence of DM increases with age. The ADA also recommends screening individuals who are overweight or obese and have an additional risk factor regardless of age. The AACE recommends screening all individuals for risk factors regardless of age.
The current and draft recommendations by the USPSTF do not address other risk factors and indicate only that further research is needed to understand the risk associated with DM and the natural history of pre-DM and who may progress to DM or revert to normoglycemia. Without comment on other risk factors or universal screening with age, the USPSTF recommendation potentially would not be sensitive enough to capture all those who may meet criteria for prediabetes or DM.2,4
In addition to not addressing other risk factors and screening for those of normal and underweight BMI, the USPSTF recommendation does not address frequency of screening. The recommendations from both the ADA and the AACE indicate screening at 3-year intervals for those who are eligible – for any reason. The supporting evidence review did not seem to address this aspect, and so it is understandable that there was no comment. However, I feel this will lead physicians to turn to the other guidelines for guidance where there is disagreement in other aspects.
Ultimately, the draft updated recommendation will provide physicians with the opportunity to identify more patients with prediabetes and DM. This will be wonderful in terms of being able to offer treatments and lifestyle interventions to decrease the morbidity patients would face were these conditions not diagnosed. I hope that future recommendations will also address risk factors in addition to BMI as well as frequency of screening for those who remain at increased risk but initially screen negative.
Dr. Wheat is a family physician at Erie Family Health Center in Chicago. She is program director of Northwestern’s McGaw Family Medicine residency program at Humboldt Park, Chicago. Dr. Wheat serves on the editorial advisory board of Family Practice News. You can contact her at fpnews@mdedge.com.
References
1. Screening for prediabetes and type 2 diabetes mellitus. U.S. Preventive Services Task Force. 2021 Mar 16.
2. Classification and diagnosis of diabetes: Standards of medical care in diabetes – 2020. American Diabetes Association. Diabetes Care. 2020 Jan. doi: 10.2337/dc20-S002.
3. National Diabetes Statistics Report, 2020. Centers for Disease Control and Prevention.
4. American Association of Clinical Endocrinologists and American College of Endocrinology – clinical practice guidelines for developing a diabetes mellitus comprehensive care plan. Hadelsman Y et al. Endocr Pract. 2015 Apr. 1-87. doi: 10.4158/EP15672.GL.
. If accepted as written, the new recommendation will be to “screen all asymptomatic adults ages 35 to 70 years who are overweight or obese.” Upon diagnosis of prediabetes, the recommendation is to offer or refer patients to preventive interventions.
This new recommendation would replace the one from 2015, which recommended screening adults aged 40-70 who are overweight or obese, lowering the age at which screening begins by 5 years. It would also replace the recommendation of referral to intensive behavioral counseling to promote a healthy diet and exercise.1
The American Diabetes Association (ADA) identifies A1c, fasting plasma glucose, or oral glucose tolerance tests as appropriate tests for the diagnosis of prediabetes and type 2 DM, and the new draft recommendation does not provide a preference for method of screening.2
The USPSTF’s draft recommendation could expand screening with the hope of identifying patients with prediabetes, or those with diabetes who are asymptomatic, with the intent of beginning treatment before there are serious complications.
Unknown diabetes or prediabetes diagnosis common
It has been estimated by the Centers for Disease Control and Prevention that 12% of U.S. adults had DM as of 2015, though nearly 24% were not aware that they had it. Also, according to the CDC, the prevalence of DM increases with age and is higher in those with less than a high school education. The same report indicates that more than 30% of U.S. adults have prediabetes, and with less than 12% of those individuals are aware of it.3 A possible explanation for a patient’s being unaware of a diagnosis could be that it has been documented in a chart but the patient does not know such information is in his or her health record. According to the evidence provided for the updated recommendation, earlier diagnosis may have an important benefit in preventing serious complications.
A modeling study compared simulated screening strategies and found that the most optimal screening strategy from a cost-effectiveness perspective begins between the ages of 30 and 45, with rescreening every 3-5 years. Further models have led researchers to conclude that early diagnosis can lead to decreased cardiovascular events as well as an opportunity for multifactorial treatment.1 For this reason, it makes sense to expand the ages of screening for obese and overweight individuals.
Treatment recommendations are more flexible
The change in treatment recommendations for a new diagnosis of prediabetes is potentially more useful. It may not be feasible or reasonable for physicians to always provide or refer their patients for intensive behavior interventions. The updated recommendation would allow for the inclusion of not only behavioral counseling and health education, but also potential medication options that are currently available but not approved, or that may be available in the future. The evidence review seemed to be mixed in outcome in this area, so the increased flexibility will likely allow for future opportunities.
Screening criteria may be too narrow
This recommendation, does not, however, provide any guidance on screening of individuals who have other risk factors besides a body mass index consistent with overweight or obesity. It seems that this may be a missed opportunity.
The draft statement clearly indicates that there are other factors associated with increased risk of developing DM, but does not consider these factors in determining which patients should be screened. Both the ADA and the American Association of Clinical Endocrinology (AACE) have recommendations for universal screening for all adults 45 and older, acknowledging that incidence of DM increases with age. The ADA also recommends screening individuals who are overweight or obese and have an additional risk factor regardless of age. The AACE recommends screening all individuals for risk factors regardless of age.
The current and draft recommendations by the USPSTF do not address other risk factors and indicate only that further research is needed to understand the risk associated with DM and the natural history of pre-DM and who may progress to DM or revert to normoglycemia. Without comment on other risk factors or universal screening with age, the USPSTF recommendation potentially would not be sensitive enough to capture all those who may meet criteria for prediabetes or DM.2,4
In addition to not addressing other risk factors and screening for those of normal and underweight BMI, the USPSTF recommendation does not address frequency of screening. The recommendations from both the ADA and the AACE indicate screening at 3-year intervals for those who are eligible – for any reason. The supporting evidence review did not seem to address this aspect, and so it is understandable that there was no comment. However, I feel this will lead physicians to turn to the other guidelines for guidance where there is disagreement in other aspects.
Ultimately, the draft updated recommendation will provide physicians with the opportunity to identify more patients with prediabetes and DM. This will be wonderful in terms of being able to offer treatments and lifestyle interventions to decrease the morbidity patients would face were these conditions not diagnosed. I hope that future recommendations will also address risk factors in addition to BMI as well as frequency of screening for those who remain at increased risk but initially screen negative.
Dr. Wheat is a family physician at Erie Family Health Center in Chicago. She is program director of Northwestern’s McGaw Family Medicine residency program at Humboldt Park, Chicago. Dr. Wheat serves on the editorial advisory board of Family Practice News. You can contact her at fpnews@mdedge.com.
References
1. Screening for prediabetes and type 2 diabetes mellitus. U.S. Preventive Services Task Force. 2021 Mar 16.
2. Classification and diagnosis of diabetes: Standards of medical care in diabetes – 2020. American Diabetes Association. Diabetes Care. 2020 Jan. doi: 10.2337/dc20-S002.
3. National Diabetes Statistics Report, 2020. Centers for Disease Control and Prevention.
4. American Association of Clinical Endocrinologists and American College of Endocrinology – clinical practice guidelines for developing a diabetes mellitus comprehensive care plan. Hadelsman Y et al. Endocr Pract. 2015 Apr. 1-87. doi: 10.4158/EP15672.GL.
Chauvin guilty verdict: Now it’s time to get to work
On Tuesday, April 20, the country braced for the impact of the trial verdict in death of George Floyd. Despite the case having what many would consider an overwhelming amount of evidence pointing toward conviction, if we’re completely honest, the country – and particularly the African American community – had significant doubts that the jury would render a guilty verdict.
In the hour leading up to the announcement, people and images dominated my thoughts; Tamir Rice, Breonna Taylor, Eric Garner, Rashard Brooks, and most recently, Daunte Wright. With the deaths of these Black Americans and many others as historical context, I took a stoic stance and held my breath as the verdict was read. Former Minneapolis Police Officer Derek Chauvin was found guilty of second-degree murder, third-degree murder, and second-degree manslaughter.
As Mr. Chauvin was remanded to custody and led away in handcuffs, it was clear there were no “winners” in this verdict. Mr. Floyd is still dead, and violent encounters experienced by Black Americans continue at a vastly disproportionate rate. The result is far from true justice, but what we as a country do have is a moment of accountability – and perhaps an opportunity for true system-level reform.
The final report of the President’s Task Force on 21st Century Policing, released in May 2015, recommended major policy changes at the federal level and developed key pillars aimed at promoting effective crime reduction while building public trust. Based on this report, four key takeaways are relevant to any discussion of police reform. All are vitally important, but two stand out as particularly relevant in the aftermath of the verdict. One of the key recommendations was “embracing a guardian – rather than a warrior mindset” in an effort to build trust and legitimacy. Another was ensuring that “peace officer and standards training (POST) boards include mandatory Crisis Intervention Training.”
As health professionals, we know that the ultimate effectiveness of any intervention is based upon the amount of shared trust and collaboration in the patient-physician relationship. As a consultation-liaison psychiatrist, I’ve been trained to recognize that, when requested to consult on a case, I’m frequently not making a medical diagnosis or delivering an intervention; I’m helping the team and patient reestablish trust in each other. Communication skills and techniques help start a dialogue, but you will ultimately fall short of shared understanding without trust. The underpinning of trust could begin with a commitment to procedural justice. Procedural justice, as described in The Justice Collaboratory of Yale Law School, “speaks to the idea of fair processes and how the quality of their experiences strongly impacts people’s perception of fairness.” There are four central tenets of procedural justice:
- Whether they were treated with dignity and respect.
- Whether they were given voice.
- Whether the decision-maker was neutral and transparent.
- Whether the decision-maker conveyed trustworthy motives.
These four tenets have been researched and shown to improve the trust and confidence a community has in police, and lay the foundation for creating a standard set of shared interests and values.
As health professionals, there are many aspects of procedural justice that we can and should embrace, particularly as we come to our reckoning with the use of restraints in medical settings.
Building on the work of the Task Force on 21st Century Policing, the National Initiative for Building Community Trust and Justice, from January 2015 through December 2018, implemented a six-city intervention aimed at generating measurable improvements in officer behavior, public safety, and community trust in police. The National Initiative was organized around three principal ideas: procedural justice, implicit bias training, and reconciliation and candid conversations about law enforcement’s historic role in racial tensions.
In addition to the recommendations of the federal government and independent institutions, national-level health policy organizations have made clear statements regarding police brutality and the need for systemic reform to address police brutality and systemic racism. In 2018, the American Psychiatric Association released a position statement on Police Brutality and Black Males. This was then followed in 2020 with a joint statement from the National Medical Association and the APA condemning systemic racism and police violence against Black Americans. Other health policy associations, including the American Medical Association and the American Association of Medical Colleges, have made clear statements condemning systemic racism and police brutality.
In the aftermath of the verdict, we also saw something very different. In our partisan country, there appeared to be uniform common ground. Statements were made acknowledging the importance of this historic moment, from police unions, and both political parties, and various invested grassroots organizations. In short, we may have true agreement and motivation to take the next hard steps in police reform for this country. There will be policy discussions and new mandates for training, and certainly a push to ban the use of lethal techniques, such as choke holds. While helpful, these will ultimately fall short unless we hold ourselves accountable for a true culture change.
The challenge of implementing procedural justice shouldn’t be just a law enforcement challenge, and it shouldn’t fall on the shoulders of communities with high crime areas. In other words, no single racial group should own it. Ultimately, procedural justice will need to be embraced by all of us.
On April 20, as I watched the verdict, my oldest daughter watched with me, and she asked, “What do you think, Dad?” I responded: “It’s accountability and an opportunity.” She nodded her head with resolve. She then grabbed her smartphone and jumped into social media and proclaimed in her very knowledgeable teenage voice, “See Dad, one voice is cool, but many voices in unison is better; time to get to work!” To Darnella Frazier, who captured the crime on video at age 17, and all in your generation who dare to hold us accountable, I salute you. I thank you for forcing us to look even when it was painful and not ignore the humanity of our fellow man. It is indeed time to get to work.
Dr. Norris is associate dean of student affairs and administration at George Washington University, Washington. He has no disclosures.
On Tuesday, April 20, the country braced for the impact of the trial verdict in death of George Floyd. Despite the case having what many would consider an overwhelming amount of evidence pointing toward conviction, if we’re completely honest, the country – and particularly the African American community – had significant doubts that the jury would render a guilty verdict.
In the hour leading up to the announcement, people and images dominated my thoughts; Tamir Rice, Breonna Taylor, Eric Garner, Rashard Brooks, and most recently, Daunte Wright. With the deaths of these Black Americans and many others as historical context, I took a stoic stance and held my breath as the verdict was read. Former Minneapolis Police Officer Derek Chauvin was found guilty of second-degree murder, third-degree murder, and second-degree manslaughter.
As Mr. Chauvin was remanded to custody and led away in handcuffs, it was clear there were no “winners” in this verdict. Mr. Floyd is still dead, and violent encounters experienced by Black Americans continue at a vastly disproportionate rate. The result is far from true justice, but what we as a country do have is a moment of accountability – and perhaps an opportunity for true system-level reform.
The final report of the President’s Task Force on 21st Century Policing, released in May 2015, recommended major policy changes at the federal level and developed key pillars aimed at promoting effective crime reduction while building public trust. Based on this report, four key takeaways are relevant to any discussion of police reform. All are vitally important, but two stand out as particularly relevant in the aftermath of the verdict. One of the key recommendations was “embracing a guardian – rather than a warrior mindset” in an effort to build trust and legitimacy. Another was ensuring that “peace officer and standards training (POST) boards include mandatory Crisis Intervention Training.”
As health professionals, we know that the ultimate effectiveness of any intervention is based upon the amount of shared trust and collaboration in the patient-physician relationship. As a consultation-liaison psychiatrist, I’ve been trained to recognize that, when requested to consult on a case, I’m frequently not making a medical diagnosis or delivering an intervention; I’m helping the team and patient reestablish trust in each other. Communication skills and techniques help start a dialogue, but you will ultimately fall short of shared understanding without trust. The underpinning of trust could begin with a commitment to procedural justice. Procedural justice, as described in The Justice Collaboratory of Yale Law School, “speaks to the idea of fair processes and how the quality of their experiences strongly impacts people’s perception of fairness.” There are four central tenets of procedural justice:
- Whether they were treated with dignity and respect.
- Whether they were given voice.
- Whether the decision-maker was neutral and transparent.
- Whether the decision-maker conveyed trustworthy motives.
These four tenets have been researched and shown to improve the trust and confidence a community has in police, and lay the foundation for creating a standard set of shared interests and values.
As health professionals, there are many aspects of procedural justice that we can and should embrace, particularly as we come to our reckoning with the use of restraints in medical settings.
Building on the work of the Task Force on 21st Century Policing, the National Initiative for Building Community Trust and Justice, from January 2015 through December 2018, implemented a six-city intervention aimed at generating measurable improvements in officer behavior, public safety, and community trust in police. The National Initiative was organized around three principal ideas: procedural justice, implicit bias training, and reconciliation and candid conversations about law enforcement’s historic role in racial tensions.
In addition to the recommendations of the federal government and independent institutions, national-level health policy organizations have made clear statements regarding police brutality and the need for systemic reform to address police brutality and systemic racism. In 2018, the American Psychiatric Association released a position statement on Police Brutality and Black Males. This was then followed in 2020 with a joint statement from the National Medical Association and the APA condemning systemic racism and police violence against Black Americans. Other health policy associations, including the American Medical Association and the American Association of Medical Colleges, have made clear statements condemning systemic racism and police brutality.
In the aftermath of the verdict, we also saw something very different. In our partisan country, there appeared to be uniform common ground. Statements were made acknowledging the importance of this historic moment, from police unions, and both political parties, and various invested grassroots organizations. In short, we may have true agreement and motivation to take the next hard steps in police reform for this country. There will be policy discussions and new mandates for training, and certainly a push to ban the use of lethal techniques, such as choke holds. While helpful, these will ultimately fall short unless we hold ourselves accountable for a true culture change.
The challenge of implementing procedural justice shouldn’t be just a law enforcement challenge, and it shouldn’t fall on the shoulders of communities with high crime areas. In other words, no single racial group should own it. Ultimately, procedural justice will need to be embraced by all of us.
On April 20, as I watched the verdict, my oldest daughter watched with me, and she asked, “What do you think, Dad?” I responded: “It’s accountability and an opportunity.” She nodded her head with resolve. She then grabbed her smartphone and jumped into social media and proclaimed in her very knowledgeable teenage voice, “See Dad, one voice is cool, but many voices in unison is better; time to get to work!” To Darnella Frazier, who captured the crime on video at age 17, and all in your generation who dare to hold us accountable, I salute you. I thank you for forcing us to look even when it was painful and not ignore the humanity of our fellow man. It is indeed time to get to work.
Dr. Norris is associate dean of student affairs and administration at George Washington University, Washington. He has no disclosures.
On Tuesday, April 20, the country braced for the impact of the trial verdict in death of George Floyd. Despite the case having what many would consider an overwhelming amount of evidence pointing toward conviction, if we’re completely honest, the country – and particularly the African American community – had significant doubts that the jury would render a guilty verdict.
In the hour leading up to the announcement, people and images dominated my thoughts; Tamir Rice, Breonna Taylor, Eric Garner, Rashard Brooks, and most recently, Daunte Wright. With the deaths of these Black Americans and many others as historical context, I took a stoic stance and held my breath as the verdict was read. Former Minneapolis Police Officer Derek Chauvin was found guilty of second-degree murder, third-degree murder, and second-degree manslaughter.
As Mr. Chauvin was remanded to custody and led away in handcuffs, it was clear there were no “winners” in this verdict. Mr. Floyd is still dead, and violent encounters experienced by Black Americans continue at a vastly disproportionate rate. The result is far from true justice, but what we as a country do have is a moment of accountability – and perhaps an opportunity for true system-level reform.
The final report of the President’s Task Force on 21st Century Policing, released in May 2015, recommended major policy changes at the federal level and developed key pillars aimed at promoting effective crime reduction while building public trust. Based on this report, four key takeaways are relevant to any discussion of police reform. All are vitally important, but two stand out as particularly relevant in the aftermath of the verdict. One of the key recommendations was “embracing a guardian – rather than a warrior mindset” in an effort to build trust and legitimacy. Another was ensuring that “peace officer and standards training (POST) boards include mandatory Crisis Intervention Training.”
As health professionals, we know that the ultimate effectiveness of any intervention is based upon the amount of shared trust and collaboration in the patient-physician relationship. As a consultation-liaison psychiatrist, I’ve been trained to recognize that, when requested to consult on a case, I’m frequently not making a medical diagnosis or delivering an intervention; I’m helping the team and patient reestablish trust in each other. Communication skills and techniques help start a dialogue, but you will ultimately fall short of shared understanding without trust. The underpinning of trust could begin with a commitment to procedural justice. Procedural justice, as described in The Justice Collaboratory of Yale Law School, “speaks to the idea of fair processes and how the quality of their experiences strongly impacts people’s perception of fairness.” There are four central tenets of procedural justice:
- Whether they were treated with dignity and respect.
- Whether they were given voice.
- Whether the decision-maker was neutral and transparent.
- Whether the decision-maker conveyed trustworthy motives.
These four tenets have been researched and shown to improve the trust and confidence a community has in police, and lay the foundation for creating a standard set of shared interests and values.
As health professionals, there are many aspects of procedural justice that we can and should embrace, particularly as we come to our reckoning with the use of restraints in medical settings.
Building on the work of the Task Force on 21st Century Policing, the National Initiative for Building Community Trust and Justice, from January 2015 through December 2018, implemented a six-city intervention aimed at generating measurable improvements in officer behavior, public safety, and community trust in police. The National Initiative was organized around three principal ideas: procedural justice, implicit bias training, and reconciliation and candid conversations about law enforcement’s historic role in racial tensions.
In addition to the recommendations of the federal government and independent institutions, national-level health policy organizations have made clear statements regarding police brutality and the need for systemic reform to address police brutality and systemic racism. In 2018, the American Psychiatric Association released a position statement on Police Brutality and Black Males. This was then followed in 2020 with a joint statement from the National Medical Association and the APA condemning systemic racism and police violence against Black Americans. Other health policy associations, including the American Medical Association and the American Association of Medical Colleges, have made clear statements condemning systemic racism and police brutality.
In the aftermath of the verdict, we also saw something very different. In our partisan country, there appeared to be uniform common ground. Statements were made acknowledging the importance of this historic moment, from police unions, and both political parties, and various invested grassroots organizations. In short, we may have true agreement and motivation to take the next hard steps in police reform for this country. There will be policy discussions and new mandates for training, and certainly a push to ban the use of lethal techniques, such as choke holds. While helpful, these will ultimately fall short unless we hold ourselves accountable for a true culture change.
The challenge of implementing procedural justice shouldn’t be just a law enforcement challenge, and it shouldn’t fall on the shoulders of communities with high crime areas. In other words, no single racial group should own it. Ultimately, procedural justice will need to be embraced by all of us.
On April 20, as I watched the verdict, my oldest daughter watched with me, and she asked, “What do you think, Dad?” I responded: “It’s accountability and an opportunity.” She nodded her head with resolve. She then grabbed her smartphone and jumped into social media and proclaimed in her very knowledgeable teenage voice, “See Dad, one voice is cool, but many voices in unison is better; time to get to work!” To Darnella Frazier, who captured the crime on video at age 17, and all in your generation who dare to hold us accountable, I salute you. I thank you for forcing us to look even when it was painful and not ignore the humanity of our fellow man. It is indeed time to get to work.
Dr. Norris is associate dean of student affairs and administration at George Washington University, Washington. He has no disclosures.
VEXAS syndrome: Implications for dermatologists
When I was a medical student, I always found it gratifying when there was a unifying mechanism that explained the symptoms of a disease. Part of the reason I chose dermatology as a specialty was how frequently we are able to “see” these mechanisms in the skin, both clinically and histologically. What’s even more interesting is that this condition is caused by a postzygotic somatic mutation, an apparently underrecognized cause of disease that we are just now beginning to understand. An example of a postzygotic somatic mutation causing a disease that we all learned about in medical school is paroxysmal nocturnal hemoglobinuria.
Using a “bottom-up” approach, researchers at the National Institutes of Health and in the United Kingdom identified 25 patients with somatic UBA1 mutations and noticed that they had strikingly similar autoinflammatory syndromes. UBA1 encodes ubiquitin E1, which is part of the pathway the breaks down proteins as part of the normal cellular machine. It is localized to the X chromosome, so all 25 affected patients were males, and most were aged between 40 and 70 years. These patients had an autoinflammatory syndrome characterized by fever, chondritis (similar to relapsing polychondritis), vasculitis, and neutrophilic dermatoses. Many patients also had features of myelodysplastic syndrome and plasma cell dyscrasia. The inflammatory pattern in this condition seems to show elevations in tumor necrosis factor, interleukin-6, and interferon-gamma.
So why is this syndrome relevant to dermatology? We are often asked to evaluate patients for neutrophilic dermatosis and vasculitis, and many affected patients had clinical and histologic findings compatible with polyarteritis nodosa and Sweet syndrome. When confronted with a neutrophilic dermatosis, we’ve all been taught to evaluate for myelodysplastic syndrome, which many of these patients appeared to have, at least on the surface. When bone marrow biopsies were done, the myeloid cell precursors that give rise to neutrophils were noted to have prominent cytoplasmic vacuoles, hence the “V” in VEXAS.
In reading the article describing 25 patients with this syndrome, which was published in the New England Journal of Medicine, I was struck by how refractory they were to treatment. Most patients had been treated with systemic steroids, multiple biologics, and several nonbiologic medications that are mainstays of treatment for neutrophilic dermatosis like dapsone and colchicine. I was fortunate enough to speak to Amanda Ombrello, MD, of the National Human Genome Research Institute, one of the lead authors of the paper, who drew my attention to the supplementary appendix, which showed the marked injection-site reactions some patients had to anakinra – yet another reason why a patient might end up in a dermatology clinic. In my mind, all of these features could be a clue to a diagnosis of VEXAS syndrome.
Many patients seemed to fare poorly, with 40% of patients dying before the completion of the study. When it comes to extremely rare diseases, it seems that the more physicians who are aware of the existence of a particular syndrome, the more likely it is a patient will come under our care and be correctly diagnosed.
Dr. Saardi is a dermatologist and internist, and is director of the inpatient dermatology service at the George Washington University Hospital, Washington. He has no disclosures.
When I was a medical student, I always found it gratifying when there was a unifying mechanism that explained the symptoms of a disease. Part of the reason I chose dermatology as a specialty was how frequently we are able to “see” these mechanisms in the skin, both clinically and histologically. What’s even more interesting is that this condition is caused by a postzygotic somatic mutation, an apparently underrecognized cause of disease that we are just now beginning to understand. An example of a postzygotic somatic mutation causing a disease that we all learned about in medical school is paroxysmal nocturnal hemoglobinuria.
Using a “bottom-up” approach, researchers at the National Institutes of Health and in the United Kingdom identified 25 patients with somatic UBA1 mutations and noticed that they had strikingly similar autoinflammatory syndromes. UBA1 encodes ubiquitin E1, which is part of the pathway the breaks down proteins as part of the normal cellular machine. It is localized to the X chromosome, so all 25 affected patients were males, and most were aged between 40 and 70 years. These patients had an autoinflammatory syndrome characterized by fever, chondritis (similar to relapsing polychondritis), vasculitis, and neutrophilic dermatoses. Many patients also had features of myelodysplastic syndrome and plasma cell dyscrasia. The inflammatory pattern in this condition seems to show elevations in tumor necrosis factor, interleukin-6, and interferon-gamma.
So why is this syndrome relevant to dermatology? We are often asked to evaluate patients for neutrophilic dermatosis and vasculitis, and many affected patients had clinical and histologic findings compatible with polyarteritis nodosa and Sweet syndrome. When confronted with a neutrophilic dermatosis, we’ve all been taught to evaluate for myelodysplastic syndrome, which many of these patients appeared to have, at least on the surface. When bone marrow biopsies were done, the myeloid cell precursors that give rise to neutrophils were noted to have prominent cytoplasmic vacuoles, hence the “V” in VEXAS.
In reading the article describing 25 patients with this syndrome, which was published in the New England Journal of Medicine, I was struck by how refractory they were to treatment. Most patients had been treated with systemic steroids, multiple biologics, and several nonbiologic medications that are mainstays of treatment for neutrophilic dermatosis like dapsone and colchicine. I was fortunate enough to speak to Amanda Ombrello, MD, of the National Human Genome Research Institute, one of the lead authors of the paper, who drew my attention to the supplementary appendix, which showed the marked injection-site reactions some patients had to anakinra – yet another reason why a patient might end up in a dermatology clinic. In my mind, all of these features could be a clue to a diagnosis of VEXAS syndrome.
Many patients seemed to fare poorly, with 40% of patients dying before the completion of the study. When it comes to extremely rare diseases, it seems that the more physicians who are aware of the existence of a particular syndrome, the more likely it is a patient will come under our care and be correctly diagnosed.
Dr. Saardi is a dermatologist and internist, and is director of the inpatient dermatology service at the George Washington University Hospital, Washington. He has no disclosures.
When I was a medical student, I always found it gratifying when there was a unifying mechanism that explained the symptoms of a disease. Part of the reason I chose dermatology as a specialty was how frequently we are able to “see” these mechanisms in the skin, both clinically and histologically. What’s even more interesting is that this condition is caused by a postzygotic somatic mutation, an apparently underrecognized cause of disease that we are just now beginning to understand. An example of a postzygotic somatic mutation causing a disease that we all learned about in medical school is paroxysmal nocturnal hemoglobinuria.
Using a “bottom-up” approach, researchers at the National Institutes of Health and in the United Kingdom identified 25 patients with somatic UBA1 mutations and noticed that they had strikingly similar autoinflammatory syndromes. UBA1 encodes ubiquitin E1, which is part of the pathway the breaks down proteins as part of the normal cellular machine. It is localized to the X chromosome, so all 25 affected patients were males, and most were aged between 40 and 70 years. These patients had an autoinflammatory syndrome characterized by fever, chondritis (similar to relapsing polychondritis), vasculitis, and neutrophilic dermatoses. Many patients also had features of myelodysplastic syndrome and plasma cell dyscrasia. The inflammatory pattern in this condition seems to show elevations in tumor necrosis factor, interleukin-6, and interferon-gamma.
So why is this syndrome relevant to dermatology? We are often asked to evaluate patients for neutrophilic dermatosis and vasculitis, and many affected patients had clinical and histologic findings compatible with polyarteritis nodosa and Sweet syndrome. When confronted with a neutrophilic dermatosis, we’ve all been taught to evaluate for myelodysplastic syndrome, which many of these patients appeared to have, at least on the surface. When bone marrow biopsies were done, the myeloid cell precursors that give rise to neutrophils were noted to have prominent cytoplasmic vacuoles, hence the “V” in VEXAS.
In reading the article describing 25 patients with this syndrome, which was published in the New England Journal of Medicine, I was struck by how refractory they were to treatment. Most patients had been treated with systemic steroids, multiple biologics, and several nonbiologic medications that are mainstays of treatment for neutrophilic dermatosis like dapsone and colchicine. I was fortunate enough to speak to Amanda Ombrello, MD, of the National Human Genome Research Institute, one of the lead authors of the paper, who drew my attention to the supplementary appendix, which showed the marked injection-site reactions some patients had to anakinra – yet another reason why a patient might end up in a dermatology clinic. In my mind, all of these features could be a clue to a diagnosis of VEXAS syndrome.
Many patients seemed to fare poorly, with 40% of patients dying before the completion of the study. When it comes to extremely rare diseases, it seems that the more physicians who are aware of the existence of a particular syndrome, the more likely it is a patient will come under our care and be correctly diagnosed.
Dr. Saardi is a dermatologist and internist, and is director of the inpatient dermatology service at the George Washington University Hospital, Washington. He has no disclosures.
Open Notes
. While some clinicians consider it an unwelcome intrusion, advocates say it will improve communication and compliance.
Patient access to notes is not new. In many states, patients already have the ability to request copies of their charts, or to access truncated information via clinic websites. The difference is that most patients will now be able to click on a patient portal – such as MyChart, or other similar apps – and gain instantaneous, unfettered access to everything in their records.
Clinicians have traditionally thought of medical notes as private journal entries; but in the last few decades they have become an important component of the documentation necessary for billing, as well as evidence in the event of litigation. Now, with the implementation of the Cures Act, medical notes have evolved into a tool to communicate with the patient, rather than just among health care providers, lawyers, and billing departments.
Supporters contend that this change will make a big difference, because patients will be able to see exactly what their doctors have written, rather than just a list of confusing test results and diagnosis lists in “medicalese.”
OpenNotes, a think tank that has promoted the sharing of clinical notes with patients for years, calls the Cures Act legislation a “new world” where shared notes are valuable tools to improve communication between patients and physicians while strengthening their relationship. They cite evidence indicating that “when health professionals offer patients and families ready access to clinical notes, the quality and safety of care improves.”
Not all doctors are as enthusiastic. Many are concerned that patients might misinterpret what they see in their doctors’ notes, including complex descriptions of clinical assessments and decisions.
Others worry about patients having immediate access to their records, perhaps even before their physicians. The American Academy of Dermatology is working with the American Medical Association and other groups to gather real-world instances where the release of lab results, reports, or notes directly to patients before their physician could review the information with them caused emotional harm or other adverse consequences.
Undoubtedly, there are scenarios where unrestricted display of clinical notes could be problematic. One example is the issue of adolescents and reproductive health. Since parents now have access to their children’s records, some teenagers might hesitate to confide in their physicians and deny themselves important medical care.
The new rules permit blocking access to records if there is clear evidence that doing so “will substantially reduce the risk of harm” to patients or third parties. Psychotherapy counseling notes, for example, are completely exempt from the new requirements.
There are also state-level laws that can supersede the new federal law and block access to notes. For example, California law forbids providers from posting cancer test results without discussing them with the patient first.
Research indicates that shared notes have benefits that should outweigh the concerns of most physicians. One study showed that about 70% of patients said reviewing their notes helped them understand why medications were prescribed, which improved their compliance. This was particularly true for patients whose primary language is not English. A British study found that patients felt empowered by shared notes, and thought they improved their relationship with their physicians.
Other advantages of sharing notes include the ability of family members to review what happened at visits, which can be particularly important when dementia or other disabilities are involved. Patients will also be able to share their medical records with physicians outside of their health network, thus avoiding unnecessary or repetitious workups.
OpenNotes contends that when patients review their doctors’ notes, they gain “a newfound, deeper respect for what physicians have to understand to do their jobs.” Other predicted advantages include improved medical record accuracy and less miscommunication. In a study published in 2019 that evaluated experiences of patients who read ambulatory visit notes, only 5% were more worried after reading the notes and 3% were confused.
Alleviating worry among clinicians may be a bigger problem; but as a general principle, you should avoid judgmental language, and never write anything in a chart that you wouldn’t want your patients or their family members – or lawyers – to see.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at dermnews@mdedge.com.
. While some clinicians consider it an unwelcome intrusion, advocates say it will improve communication and compliance.
Patient access to notes is not new. In many states, patients already have the ability to request copies of their charts, or to access truncated information via clinic websites. The difference is that most patients will now be able to click on a patient portal – such as MyChart, or other similar apps – and gain instantaneous, unfettered access to everything in their records.
Clinicians have traditionally thought of medical notes as private journal entries; but in the last few decades they have become an important component of the documentation necessary for billing, as well as evidence in the event of litigation. Now, with the implementation of the Cures Act, medical notes have evolved into a tool to communicate with the patient, rather than just among health care providers, lawyers, and billing departments.
Supporters contend that this change will make a big difference, because patients will be able to see exactly what their doctors have written, rather than just a list of confusing test results and diagnosis lists in “medicalese.”
OpenNotes, a think tank that has promoted the sharing of clinical notes with patients for years, calls the Cures Act legislation a “new world” where shared notes are valuable tools to improve communication between patients and physicians while strengthening their relationship. They cite evidence indicating that “when health professionals offer patients and families ready access to clinical notes, the quality and safety of care improves.”
Not all doctors are as enthusiastic. Many are concerned that patients might misinterpret what they see in their doctors’ notes, including complex descriptions of clinical assessments and decisions.
Others worry about patients having immediate access to their records, perhaps even before their physicians. The American Academy of Dermatology is working with the American Medical Association and other groups to gather real-world instances where the release of lab results, reports, or notes directly to patients before their physician could review the information with them caused emotional harm or other adverse consequences.
Undoubtedly, there are scenarios where unrestricted display of clinical notes could be problematic. One example is the issue of adolescents and reproductive health. Since parents now have access to their children’s records, some teenagers might hesitate to confide in their physicians and deny themselves important medical care.
The new rules permit blocking access to records if there is clear evidence that doing so “will substantially reduce the risk of harm” to patients or third parties. Psychotherapy counseling notes, for example, are completely exempt from the new requirements.
There are also state-level laws that can supersede the new federal law and block access to notes. For example, California law forbids providers from posting cancer test results without discussing them with the patient first.
Research indicates that shared notes have benefits that should outweigh the concerns of most physicians. One study showed that about 70% of patients said reviewing their notes helped them understand why medications were prescribed, which improved their compliance. This was particularly true for patients whose primary language is not English. A British study found that patients felt empowered by shared notes, and thought they improved their relationship with their physicians.
Other advantages of sharing notes include the ability of family members to review what happened at visits, which can be particularly important when dementia or other disabilities are involved. Patients will also be able to share their medical records with physicians outside of their health network, thus avoiding unnecessary or repetitious workups.
OpenNotes contends that when patients review their doctors’ notes, they gain “a newfound, deeper respect for what physicians have to understand to do their jobs.” Other predicted advantages include improved medical record accuracy and less miscommunication. In a study published in 2019 that evaluated experiences of patients who read ambulatory visit notes, only 5% were more worried after reading the notes and 3% were confused.
Alleviating worry among clinicians may be a bigger problem; but as a general principle, you should avoid judgmental language, and never write anything in a chart that you wouldn’t want your patients or their family members – or lawyers – to see.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at dermnews@mdedge.com.
. While some clinicians consider it an unwelcome intrusion, advocates say it will improve communication and compliance.
Patient access to notes is not new. In many states, patients already have the ability to request copies of their charts, or to access truncated information via clinic websites. The difference is that most patients will now be able to click on a patient portal – such as MyChart, or other similar apps – and gain instantaneous, unfettered access to everything in their records.
Clinicians have traditionally thought of medical notes as private journal entries; but in the last few decades they have become an important component of the documentation necessary for billing, as well as evidence in the event of litigation. Now, with the implementation of the Cures Act, medical notes have evolved into a tool to communicate with the patient, rather than just among health care providers, lawyers, and billing departments.
Supporters contend that this change will make a big difference, because patients will be able to see exactly what their doctors have written, rather than just a list of confusing test results and diagnosis lists in “medicalese.”
OpenNotes, a think tank that has promoted the sharing of clinical notes with patients for years, calls the Cures Act legislation a “new world” where shared notes are valuable tools to improve communication between patients and physicians while strengthening their relationship. They cite evidence indicating that “when health professionals offer patients and families ready access to clinical notes, the quality and safety of care improves.”
Not all doctors are as enthusiastic. Many are concerned that patients might misinterpret what they see in their doctors’ notes, including complex descriptions of clinical assessments and decisions.
Others worry about patients having immediate access to their records, perhaps even before their physicians. The American Academy of Dermatology is working with the American Medical Association and other groups to gather real-world instances where the release of lab results, reports, or notes directly to patients before their physician could review the information with them caused emotional harm or other adverse consequences.
Undoubtedly, there are scenarios where unrestricted display of clinical notes could be problematic. One example is the issue of adolescents and reproductive health. Since parents now have access to their children’s records, some teenagers might hesitate to confide in their physicians and deny themselves important medical care.
The new rules permit blocking access to records if there is clear evidence that doing so “will substantially reduce the risk of harm” to patients or third parties. Psychotherapy counseling notes, for example, are completely exempt from the new requirements.
There are also state-level laws that can supersede the new federal law and block access to notes. For example, California law forbids providers from posting cancer test results without discussing them with the patient first.
Research indicates that shared notes have benefits that should outweigh the concerns of most physicians. One study showed that about 70% of patients said reviewing their notes helped them understand why medications were prescribed, which improved their compliance. This was particularly true for patients whose primary language is not English. A British study found that patients felt empowered by shared notes, and thought they improved their relationship with their physicians.
Other advantages of sharing notes include the ability of family members to review what happened at visits, which can be particularly important when dementia or other disabilities are involved. Patients will also be able to share their medical records with physicians outside of their health network, thus avoiding unnecessary or repetitious workups.
OpenNotes contends that when patients review their doctors’ notes, they gain “a newfound, deeper respect for what physicians have to understand to do their jobs.” Other predicted advantages include improved medical record accuracy and less miscommunication. In a study published in 2019 that evaluated experiences of patients who read ambulatory visit notes, only 5% were more worried after reading the notes and 3% were confused.
Alleviating worry among clinicians may be a bigger problem; but as a general principle, you should avoid judgmental language, and never write anything in a chart that you wouldn’t want your patients or their family members – or lawyers – to see.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at dermnews@mdedge.com.
Addressing women’s concerns about the J&J vaccine
A rare form of venous thromboembolism (VTE) has developed in premenopausal women who have received the Johnson & Johnson (J&J) SARS-CoV-2 vaccine.
This week we learned that of the more than 6.8 million individuals in the United States who received the single-dose J&J vaccine, six women aged 18-48 years have been diagnosed with cerebral venous sinus thrombosis, and all had thrombocytopenia. In each case, symptoms were first noted 1-2 weeks after vaccination. The Food and Drug Administration and Centers for Disease Control and Prevention have recommended a pause in the administration of this vaccine.
Women’s health clinicians are already hearing from concerned patients, who understandably have questions about what this news means for them.
If they have already received the J&J vaccine within the past 3 weeks, I advise them that, although risks for any vaccine-related problems are extremely low, they should be mindful of new-onset leg or abdominal pain, or an unusual or severe headache. Such patients should contact their physician as soon as possible, and if they cannot be seen quickly, it would be appropriate to visit a hospital ED. When seeking medical care, patients should specify details of their vaccination history. Depending on the individual issues present, women with suggestive symptoms should receive blood work, Doppler venous studies (if there is a suspicion of lower-extremity deep vein thrombosis), and appropriate imaging (if there is concern for cerebral venous sinus thrombosis or pulmonary embolism).
As physicians and scientists at the CDC and FDA dig into this issue, I assume they are asking questions to determine whether the affected women have any factors that might increase their baseline risk for VTE, such as:
- A body mass index of at least 30 kg/m2
- Use of combination estrogen-progestin contraceptives (pill, ring, or patch)
- Known or suspected chronic inflammatory conditions such as rheumatoid arthritis, systemic lupus erythematosus, or
- Known familial or other thrombophilic conditions or chronic
- Recent prolonged immobility, such as a long airplane or automobile trip, which might increase risk for VTE
Experts say that the risk for a serious adverse event following receipt of the J&J vaccine is outweighed by the benefits of vaccination against COVID disease. However, that may not be enough to allay concerns among some premenopausal women.
Even if the “pause” in the administration of the vaccine is lifted, some women may be asking whether they should receive J&J’s viral vector vaccine or request one of the messenger RNA vaccines. I will be looking to the expert opinions of Anthony S. Fauci, MD, and advice from the CDC and FDA for guidance here. However, it may be reasonable to steer high-risk reproductive-age women away from the J&J vaccine in favor of the Moderna and Pfizer vaccines, if these options are available.
A version of this article first appeared on Medscape.com.
A rare form of venous thromboembolism (VTE) has developed in premenopausal women who have received the Johnson & Johnson (J&J) SARS-CoV-2 vaccine.
This week we learned that of the more than 6.8 million individuals in the United States who received the single-dose J&J vaccine, six women aged 18-48 years have been diagnosed with cerebral venous sinus thrombosis, and all had thrombocytopenia. In each case, symptoms were first noted 1-2 weeks after vaccination. The Food and Drug Administration and Centers for Disease Control and Prevention have recommended a pause in the administration of this vaccine.
Women’s health clinicians are already hearing from concerned patients, who understandably have questions about what this news means for them.
If they have already received the J&J vaccine within the past 3 weeks, I advise them that, although risks for any vaccine-related problems are extremely low, they should be mindful of new-onset leg or abdominal pain, or an unusual or severe headache. Such patients should contact their physician as soon as possible, and if they cannot be seen quickly, it would be appropriate to visit a hospital ED. When seeking medical care, patients should specify details of their vaccination history. Depending on the individual issues present, women with suggestive symptoms should receive blood work, Doppler venous studies (if there is a suspicion of lower-extremity deep vein thrombosis), and appropriate imaging (if there is concern for cerebral venous sinus thrombosis or pulmonary embolism).
As physicians and scientists at the CDC and FDA dig into this issue, I assume they are asking questions to determine whether the affected women have any factors that might increase their baseline risk for VTE, such as:
- A body mass index of at least 30 kg/m2
- Use of combination estrogen-progestin contraceptives (pill, ring, or patch)
- Known or suspected chronic inflammatory conditions such as rheumatoid arthritis, systemic lupus erythematosus, or
- Known familial or other thrombophilic conditions or chronic
- Recent prolonged immobility, such as a long airplane or automobile trip, which might increase risk for VTE
Experts say that the risk for a serious adverse event following receipt of the J&J vaccine is outweighed by the benefits of vaccination against COVID disease. However, that may not be enough to allay concerns among some premenopausal women.
Even if the “pause” in the administration of the vaccine is lifted, some women may be asking whether they should receive J&J’s viral vector vaccine or request one of the messenger RNA vaccines. I will be looking to the expert opinions of Anthony S. Fauci, MD, and advice from the CDC and FDA for guidance here. However, it may be reasonable to steer high-risk reproductive-age women away from the J&J vaccine in favor of the Moderna and Pfizer vaccines, if these options are available.
A version of this article first appeared on Medscape.com.
A rare form of venous thromboembolism (VTE) has developed in premenopausal women who have received the Johnson & Johnson (J&J) SARS-CoV-2 vaccine.
This week we learned that of the more than 6.8 million individuals in the United States who received the single-dose J&J vaccine, six women aged 18-48 years have been diagnosed with cerebral venous sinus thrombosis, and all had thrombocytopenia. In each case, symptoms were first noted 1-2 weeks after vaccination. The Food and Drug Administration and Centers for Disease Control and Prevention have recommended a pause in the administration of this vaccine.
Women’s health clinicians are already hearing from concerned patients, who understandably have questions about what this news means for them.
If they have already received the J&J vaccine within the past 3 weeks, I advise them that, although risks for any vaccine-related problems are extremely low, they should be mindful of new-onset leg or abdominal pain, or an unusual or severe headache. Such patients should contact their physician as soon as possible, and if they cannot be seen quickly, it would be appropriate to visit a hospital ED. When seeking medical care, patients should specify details of their vaccination history. Depending on the individual issues present, women with suggestive symptoms should receive blood work, Doppler venous studies (if there is a suspicion of lower-extremity deep vein thrombosis), and appropriate imaging (if there is concern for cerebral venous sinus thrombosis or pulmonary embolism).
As physicians and scientists at the CDC and FDA dig into this issue, I assume they are asking questions to determine whether the affected women have any factors that might increase their baseline risk for VTE, such as:
- A body mass index of at least 30 kg/m2
- Use of combination estrogen-progestin contraceptives (pill, ring, or patch)
- Known or suspected chronic inflammatory conditions such as rheumatoid arthritis, systemic lupus erythematosus, or
- Known familial or other thrombophilic conditions or chronic
- Recent prolonged immobility, such as a long airplane or automobile trip, which might increase risk for VTE
Experts say that the risk for a serious adverse event following receipt of the J&J vaccine is outweighed by the benefits of vaccination against COVID disease. However, that may not be enough to allay concerns among some premenopausal women.
Even if the “pause” in the administration of the vaccine is lifted, some women may be asking whether they should receive J&J’s viral vector vaccine or request one of the messenger RNA vaccines. I will be looking to the expert opinions of Anthony S. Fauci, MD, and advice from the CDC and FDA for guidance here. However, it may be reasonable to steer high-risk reproductive-age women away from the J&J vaccine in favor of the Moderna and Pfizer vaccines, if these options are available.
A version of this article first appeared on Medscape.com.