User login
A Rare Case of Leptomeningeal Carcinomatosis From Gastroesophageal Adenocarcinoma Masquerading as Polyneuropathy
INTRODUCTION
Leptomeningeal metastasis (LM) is an extremely rare complication of gastroesophageal (GE) cancer. Diagnosis is challenging due to frequently nonspecific clinical presentations, limited sensitivity of diagnostic testing, and potential overlap with neurologic immune-related adverse events (irAE). We describe a case of metastatic gastroesophageal cancer on immunotherapy presenting with LM masquerading as polyneuropathy.
CASE REPORT
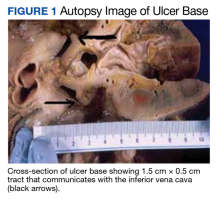
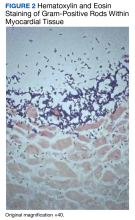
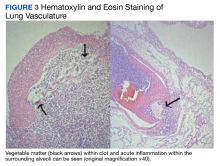
A 74-year-old male with HER2+ GE junction cancer with peritoneal metastases diagnosed 6 months ago, on maintenance trastuzumab/pembrolizumab and with no previous history of cranial or spinal disease, presented with worsening ataxia, headache, and diplopia for one month with multiple negative outpatient MRIs. Examination showed left abducens nerve palsy, dysmetria and absent deep tendon reflexes in upper and lower extremities. CT head was unremarkable, and MRI showed non-specific mild enhancement of the right optic nerve, symmetrical lumbosacral nerve roots and cauda equina concerning for paraneoplastic versus immunotherapy-related polyneuropathy. He was started on empiric high-dose corticosteroids. PET-CT was negative for FDG-avid lesions. Cerebrospinal fluid (CSF) analysis revealed moderate pleocytosis with many large atypical cells, elevated protein (118 mg/dL) and LDH (28 IU/L). Immunohistochemistry was positive for CDX2, CA 19-9, CK7, and pankeratin, consistent with metastatic adenocarcinoma, negative for HER2 in contrast to the original tumor. He subsequently developed hydrocephalus requiring a ventriculoperitoneal shunt. He received ten fractions of whole brain irradiation before electing to pursue hospice care.
DISCUSSION
LM is an extremely rare complication of GE cancer with an incidence of <0.2% and carries a poor prognosis. Differentiation between LM and irAE in patients on immunotherapy can be challenging. Diagnosis relies mostly on CSF cytology, and lumbar puncture should not be delayed in patients with new neurologic symptoms. Treatment options are intrathecal chemotherapy, radiation and steroids. A recent phase II trial has shown promise for intrathecal trastuzumab in patients with HER2+ cancers, but options for HER2 negative disease remain mostly palliative.
CONCLUSIONS
Our case highlights the need for suspecting this rare metastatic site, as early diagnosis and genetic characterization allow for exploring more treatment options including targeted therapies which may improve overall survival and quality of life.
INTRODUCTION
Leptomeningeal metastasis (LM) is an extremely rare complication of gastroesophageal (GE) cancer. Diagnosis is challenging due to frequently nonspecific clinical presentations, limited sensitivity of diagnostic testing, and potential overlap with neurologic immune-related adverse events (irAE). We describe a case of metastatic gastroesophageal cancer on immunotherapy presenting with LM masquerading as polyneuropathy.
CASE REPORT
A 74-year-old male with HER2+ GE junction cancer with peritoneal metastases diagnosed 6 months ago, on maintenance trastuzumab/pembrolizumab and with no previous history of cranial or spinal disease, presented with worsening ataxia, headache, and diplopia for one month with multiple negative outpatient MRIs. Examination showed left abducens nerve palsy, dysmetria and absent deep tendon reflexes in upper and lower extremities. CT head was unremarkable, and MRI showed non-specific mild enhancement of the right optic nerve, symmetrical lumbosacral nerve roots and cauda equina concerning for paraneoplastic versus immunotherapy-related polyneuropathy. He was started on empiric high-dose corticosteroids. PET-CT was negative for FDG-avid lesions. Cerebrospinal fluid (CSF) analysis revealed moderate pleocytosis with many large atypical cells, elevated protein (118 mg/dL) and LDH (28 IU/L). Immunohistochemistry was positive for CDX2, CA 19-9, CK7, and pankeratin, consistent with metastatic adenocarcinoma, negative for HER2 in contrast to the original tumor. He subsequently developed hydrocephalus requiring a ventriculoperitoneal shunt. He received ten fractions of whole brain irradiation before electing to pursue hospice care.
DISCUSSION
LM is an extremely rare complication of GE cancer with an incidence of <0.2% and carries a poor prognosis. Differentiation between LM and irAE in patients on immunotherapy can be challenging. Diagnosis relies mostly on CSF cytology, and lumbar puncture should not be delayed in patients with new neurologic symptoms. Treatment options are intrathecal chemotherapy, radiation and steroids. A recent phase II trial has shown promise for intrathecal trastuzumab in patients with HER2+ cancers, but options for HER2 negative disease remain mostly palliative.
CONCLUSIONS
Our case highlights the need for suspecting this rare metastatic site, as early diagnosis and genetic characterization allow for exploring more treatment options including targeted therapies which may improve overall survival and quality of life.
INTRODUCTION
Leptomeningeal metastasis (LM) is an extremely rare complication of gastroesophageal (GE) cancer. Diagnosis is challenging due to frequently nonspecific clinical presentations, limited sensitivity of diagnostic testing, and potential overlap with neurologic immune-related adverse events (irAE). We describe a case of metastatic gastroesophageal cancer on immunotherapy presenting with LM masquerading as polyneuropathy.
CASE REPORT
A 74-year-old male with HER2+ GE junction cancer with peritoneal metastases diagnosed 6 months ago, on maintenance trastuzumab/pembrolizumab and with no previous history of cranial or spinal disease, presented with worsening ataxia, headache, and diplopia for one month with multiple negative outpatient MRIs. Examination showed left abducens nerve palsy, dysmetria and absent deep tendon reflexes in upper and lower extremities. CT head was unremarkable, and MRI showed non-specific mild enhancement of the right optic nerve, symmetrical lumbosacral nerve roots and cauda equina concerning for paraneoplastic versus immunotherapy-related polyneuropathy. He was started on empiric high-dose corticosteroids. PET-CT was negative for FDG-avid lesions. Cerebrospinal fluid (CSF) analysis revealed moderate pleocytosis with many large atypical cells, elevated protein (118 mg/dL) and LDH (28 IU/L). Immunohistochemistry was positive for CDX2, CA 19-9, CK7, and pankeratin, consistent with metastatic adenocarcinoma, negative for HER2 in contrast to the original tumor. He subsequently developed hydrocephalus requiring a ventriculoperitoneal shunt. He received ten fractions of whole brain irradiation before electing to pursue hospice care.
DISCUSSION
LM is an extremely rare complication of GE cancer with an incidence of <0.2% and carries a poor prognosis. Differentiation between LM and irAE in patients on immunotherapy can be challenging. Diagnosis relies mostly on CSF cytology, and lumbar puncture should not be delayed in patients with new neurologic symptoms. Treatment options are intrathecal chemotherapy, radiation and steroids. A recent phase II trial has shown promise for intrathecal trastuzumab in patients with HER2+ cancers, but options for HER2 negative disease remain mostly palliative.
CONCLUSIONS
Our case highlights the need for suspecting this rare metastatic site, as early diagnosis and genetic characterization allow for exploring more treatment options including targeted therapies which may improve overall survival and quality of life.
Pacify the Prostate, Pop Goes the Pituitary
INTRODUCTION
Excluding skin cancer, prostate cancer is the most common malignancy affecting men in the United States, accounting for ~33% of VA cancer cases. Androgen deprivation therapy (ADT) is considered standard of care in treating advanced prostate cancer. Pituitary apoplexy is a rare and morbid adverse event associated with GnRH agonist treatment. We describe a patient with advanced prostate cancer who developed pituitary apoplexy shortly after leuprolide therapy.
CASE PRESENTATION
A 70-year-old African-American male was diagnosed with a T2aN1M1 stage IVB prostate cancer, Gleason 4+5, PSA 19.5. Four hours after his first leuprolide injection, he developed vomiting, diaphoresis, myalgia, and a severe frontal headache. Brain MRI revealed a 2.4 × 1.3 × 1.3cm pituitary mass, suspicious for an adenoma with hemorrhage. Labs noted low TSH, prolactin, LH, growth hormone, ACTH, cortisol, and testosterone, consistent with pituitary apoplexy. He was treated with steroids. Three weeks later, testosterone levels remained very low. He started abiraterone and prednisone without further leuprolide.
DISCUSSION
Prostate cancer is ubiquitous among VA patients, and ADT with GnRH agonist is vital in their care. These medications stimulate the pituitary to release LH and FSH resulting in a negative feedback loop, ultimately decreasing the levels of testosterone. Common side effects of GnRH agonists include hot flashes, diaphoresis, and sexual dysfunction. We present a patient who started leuprolide for prostate cancer. Symptoms including a severe headache led to an evaluation confirming pituitary apoplexy. Literature review reveals ~ 21 cases of pituitary apoplexy associated with GnRH agonist treatment for prostate cancer, and apoplexy can occur immediately to months later Undiagnosed pituitary adenomas are common among these patients. Treatment includes pituitary surgery or conservative management. Further prostate cancer treatment needs investigation, but we propose that GnRH modifying treatment can be withheld while testosterone levels remain low.
CONCLUSIONS
Prostate cancer is extremely common in the VA population, and treatment with leuprolide is standard. Pituitary apoplexy is a rare, but devastating complication of this treatment, and providers should be aware of the symptoms in order to intervene quickly. Further testosterone lowering treatment may be withheld if testosterone levels remain low.
INTRODUCTION
Excluding skin cancer, prostate cancer is the most common malignancy affecting men in the United States, accounting for ~33% of VA cancer cases. Androgen deprivation therapy (ADT) is considered standard of care in treating advanced prostate cancer. Pituitary apoplexy is a rare and morbid adverse event associated with GnRH agonist treatment. We describe a patient with advanced prostate cancer who developed pituitary apoplexy shortly after leuprolide therapy.
CASE PRESENTATION
A 70-year-old African-American male was diagnosed with a T2aN1M1 stage IVB prostate cancer, Gleason 4+5, PSA 19.5. Four hours after his first leuprolide injection, he developed vomiting, diaphoresis, myalgia, and a severe frontal headache. Brain MRI revealed a 2.4 × 1.3 × 1.3cm pituitary mass, suspicious for an adenoma with hemorrhage. Labs noted low TSH, prolactin, LH, growth hormone, ACTH, cortisol, and testosterone, consistent with pituitary apoplexy. He was treated with steroids. Three weeks later, testosterone levels remained very low. He started abiraterone and prednisone without further leuprolide.
DISCUSSION
Prostate cancer is ubiquitous among VA patients, and ADT with GnRH agonist is vital in their care. These medications stimulate the pituitary to release LH and FSH resulting in a negative feedback loop, ultimately decreasing the levels of testosterone. Common side effects of GnRH agonists include hot flashes, diaphoresis, and sexual dysfunction. We present a patient who started leuprolide for prostate cancer. Symptoms including a severe headache led to an evaluation confirming pituitary apoplexy. Literature review reveals ~ 21 cases of pituitary apoplexy associated with GnRH agonist treatment for prostate cancer, and apoplexy can occur immediately to months later Undiagnosed pituitary adenomas are common among these patients. Treatment includes pituitary surgery or conservative management. Further prostate cancer treatment needs investigation, but we propose that GnRH modifying treatment can be withheld while testosterone levels remain low.
CONCLUSIONS
Prostate cancer is extremely common in the VA population, and treatment with leuprolide is standard. Pituitary apoplexy is a rare, but devastating complication of this treatment, and providers should be aware of the symptoms in order to intervene quickly. Further testosterone lowering treatment may be withheld if testosterone levels remain low.
INTRODUCTION
Excluding skin cancer, prostate cancer is the most common malignancy affecting men in the United States, accounting for ~33% of VA cancer cases. Androgen deprivation therapy (ADT) is considered standard of care in treating advanced prostate cancer. Pituitary apoplexy is a rare and morbid adverse event associated with GnRH agonist treatment. We describe a patient with advanced prostate cancer who developed pituitary apoplexy shortly after leuprolide therapy.
CASE PRESENTATION
A 70-year-old African-American male was diagnosed with a T2aN1M1 stage IVB prostate cancer, Gleason 4+5, PSA 19.5. Four hours after his first leuprolide injection, he developed vomiting, diaphoresis, myalgia, and a severe frontal headache. Brain MRI revealed a 2.4 × 1.3 × 1.3cm pituitary mass, suspicious for an adenoma with hemorrhage. Labs noted low TSH, prolactin, LH, growth hormone, ACTH, cortisol, and testosterone, consistent with pituitary apoplexy. He was treated with steroids. Three weeks later, testosterone levels remained very low. He started abiraterone and prednisone without further leuprolide.
DISCUSSION
Prostate cancer is ubiquitous among VA patients, and ADT with GnRH agonist is vital in their care. These medications stimulate the pituitary to release LH and FSH resulting in a negative feedback loop, ultimately decreasing the levels of testosterone. Common side effects of GnRH agonists include hot flashes, diaphoresis, and sexual dysfunction. We present a patient who started leuprolide for prostate cancer. Symptoms including a severe headache led to an evaluation confirming pituitary apoplexy. Literature review reveals ~ 21 cases of pituitary apoplexy associated with GnRH agonist treatment for prostate cancer, and apoplexy can occur immediately to months later Undiagnosed pituitary adenomas are common among these patients. Treatment includes pituitary surgery or conservative management. Further prostate cancer treatment needs investigation, but we propose that GnRH modifying treatment can be withheld while testosterone levels remain low.
CONCLUSIONS
Prostate cancer is extremely common in the VA population, and treatment with leuprolide is standard. Pituitary apoplexy is a rare, but devastating complication of this treatment, and providers should be aware of the symptoms in order to intervene quickly. Further testosterone lowering treatment may be withheld if testosterone levels remain low.
What We Have Learned About Combining a Ketogenic Diet and Chemoimmunotherapy: A Case Report and Review of Literature
Originally developed for the treatment of refractory epilepsy, the ketogenic diet is distinguished by its high-fat, moderate-protein, and low-carbohydrate food program. Preclinical models provide emerging evidence that a ketogenic diet can have therapeutic potential for a broad range of cancers. The Warburg effect is a condition where cancer cells increase the uptake and fermentation of glucose to produce lactate for their metabolism, which is called aerobic glycolysis. Lactate is the key driver of cancer angiogenesis and proliferation.1,2
The ketogenic diet promotes a metabolic shift from glycolysis to mitochondrial metabolism in normal cells while cancer cells have dysfunction in their mitochondria due to damage in cellular respiration. The ketogenic diet creates a metabolic state whereby blood glucose levels are reduced, and blood ketone bodies (D-β-hydroxybutyrate and acetoacetate) are elevated. In normal cells, the ketogenic diet causes a decrease in glucose intake for glycolysis, which makes them unable to produce enough substrate to enter the tricarboxylic acid (TCA) cycle for adenosine triphosphate (ATP) production. Fatty acid oxidation plays a key role in ketone body synthesis as a “super fuel” that enter the TCA cycle as an alternative pathway to generate ATP. On the other hand, cancer cells are unable to use ketone bodies to produce ATP for energy and metabolism due to mitochondrial defects. Lack of energy subsequently leads to the inhibition of proliferation and survival of cancer cells.3,4
We previously published a safety and feasibility study of the Modified Atkins Diet in metastatic cancer patients after failure of chemotherapy at the US Department of Veterans Affairs (VA) Pittsburgh Healthcare System.1 None of the patients were on chemotherapy at the time of enrollment. The Modified Atkins Diet consists of 60% fat, 30% protein, and 10% carbohydrates and is more tolerable than the ketogenic diet due to higher amounts of protein. Six of 11 patients (54%) had stable disease and partial response on positron emission tomography/computed tomography (PET/CT). Our study showed that patients who lost at least 10% of their body weight had improvement in quality of life (QOL) and cancer response.1 Here we present a case of a veteran with extensive metastatic colon cancer on concurrent ketogenic diet and chemotherapy subsequently followed by concurrent ketogenic diet and immunotherapy at Veterans Affairs Central California Health Care Systems (VACCHCS) in Fresno.
CASE PRESENTATION
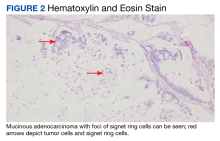
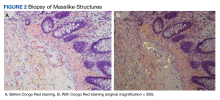
A 69-year-old veteran had iron deficiency anemia (hemoglobin, 6.5 g/dL) about 5 years previously. He underwent a colonoscopy that revealed a near circumferential ulcerated mass measuring 7 cm in the transverse colon. Biopsy results showed mucinous adenocarcinoma of the colon with a foci of signet ring cells (Figure 2).
The patient received adjuvant treatment with FOLFOX (fluorouracil, leucovorin calcium, and oxaliplatin), but within several months he developed pancreatic and worsening omental metastasis seen on PET/CT. He was then started on FOLFIRI (fluorouracil, leucovorin calcium, and irinotecan hydrochloride) plus bevacizumab 16 months after his initial diagnosis. He underwent a pancreatic mastectomy that confirmed adenocarcinoma 9 months later. Afterward, he briefly resumed FOLFIRI and bevacizumab. Next-generation sequencing testing with Foundation One CDx revealed a wild-type (WT) KRAS with a high degree of tumor mutation burden of 37 muts/Mb, BRAF V600E mutation, and high microsatellite instability (MSI-H).
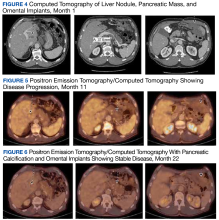
Due to disease progression, the patient’s treatment was changed to encorafenib and cetuximab for 4 months before progressing again with new liver mass and mediastinal lymphadenopathy. He then received pembrolizumab for 4 months until PET/CT showed progression and his carcinoembryonic antigen (CEA) increased from 95 to 1031 ng/mL by January 2021 (Figure 4).
The patient was started on trifluridine/tipiracil, and bevacizumab while concurrently initiating the ketogenic diet in January 2021. Laboratory tests drawn after 1 week of strict dietary ketogenic diet adherence showed low-level ketosis with a glucose ketone index (GKI) of 8.2 (Table 1).
A follow-up PET/CT showed disease progression along with a CEA of 94 ng/mL after 10 months of chemotherapy plus the ketogenic diet (Table 2).
The patient continued to experience excellent QOL based on the QOL Eastern Cooperative Oncology Group (ECOG) core quality of life questionnaire (QLC-C30) forms, which he completed every 3 months. Twenty-two months after starting the ketogenic diet, the patient’s CEA increased to 293 ng/mL although PET/CT continues to show stable disease (Figures 4, 5, and 6).
DISCUSSION
The purpose of this case report is to describe whether a patient receiving active cancer treatment was able to tolerate the ketogenic diet in conjunction with chemotherapy or immunotherapy. Most literature published on the subject evaluated the tolerability and response of the ketogenic diet after the failure of standard therapy. Our patient was diagnosed with stage III mucinous colon adenocarcinoma. He received adjuvant chemotherapy but quickly developed metastatic disease to the pancreas and omentum. We started him on encorafenib and cetuximab based on the BEACON study that showed improvement in response rate and survival when compared with standard chemotherapy for patients with BRAF V600E mutation.5 Unfortunately, his cancer quickly progressed within 4 months and again did not respond to pembrolizumab despite MSI-H, which lasted for another 4 months.
We suggested the ketogenic diet and the patient agreed. He started the diet along with trifluridine/tipiracil, and bevacizumab in January 2021. The patient’s metastatic cancer stabilized for 9 months until his disease progressed again. He was started on doublet immune checkpoint inhibitors ipilimumab and nivolumab based on his MSI-H and high tumor mutation burden with the continuation of the ketogenic diet until now. The CheckMate 142 study revealed that the combination of ipilimumab and nivolumab in patients with MSI-H previously treated for metastatic colon cancer showed some benefit.6
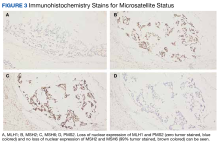
Our patient had the loss of nuclear expression of MLH1 and PMS2 (zero tumor stained) but no evidence of the loss expression of MSH2 and MSH6 genes (99% tumor stained). About 8% to 12% of patients with metastatic colon cancer have BRAF V600E mutations that are usually mucinous type, poorly differentiated, and located in the right side of the colon, which portends to a poor prognosis. Tumor DNA mismatch repair damage results in genetic hypermutability and leads to MSI that is sensitive to treatment with checkpoint inhibitors, as in our patient. Only about 3% of MSI-H tumors are due to germline mutations such as Lynch syndrome (hereditary nonpolyposis colorectal cancer). The presence of both MLH1 hypermethylation and BRAF mutation, as in our patient, is a strong indication of somatic rather than germline mutation.7
GKI, which represents the ratio of glucose to ketone, was developed to evaluate the efficacy of the ketogenic diet. This index measures the degree of metabolic stress on tumor cells through the decrease of glucose levels and increase of ketone bodies. A GKI of ≤ 1.0 has been suggested as the ideal therapeutic goal for cancer management.8 As levels of blood glucose decline, the blood levels of ketone bodies should rise. These 2 lines should eventually intersect at a certain point beyond which one enters the therapeutic zone or therapeutic ketosis zone. This is when tumor growth is expected to slow or cease.9 The patient’s ketone (β-hydroxybutyrate) level was initially high (0.71 mmol/L) with a GKI of 8.2. (low ketotic level), which meant he tolerated a rather strict diet for the first several months. This was also reflected in his 18 lb weight loss (almost 10% of body weight) and cancer stabilization, as in our previous publication.1 Unfortunately, the patient was unable to maintain high ketone and lower GKI levels due to fatigue from depleted carbohydrate intake. He added some carbohydrate snacks in between meals, which improved the fatigue. His ketone level has been < 0.5 mmol/L ever since, albeit his disease continues to be stable. The patient continues his daily work and reports a better QOL, based on the ECOG QLC-C30 form that he completed every 3 months.10 Currently, the patient is still receiving ipilimumab and nivolumab while maintaining the ketogenic diet with stable metastatic disease on PET/CT.
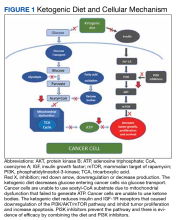
Ketogenic Diet and Cellular Mechanism of Action
PI3K/Akt (phosphatidylinositol-3-kinase) signaling is one of the most important intracellular pathways for tumor cells. It leads to the inhibition of apoptosis and the promotion of cell proliferation, metabolism, and angiogenesis. Deregulation of the PI3K pathway either via amplification of PI3K by tyrosine kinase growth factor receptors or inactivation of the tumor suppressor phosphatase and tensin homolog (PTEN), which is the negative regulator of the PI3K pathway, contributes to the development of cancer cells.11
A study by Goncalves and colleagues revealed an interesting relationship between the PI3K pathway and the benefit of the ketogenic diet to slow tumor growth. PI3K inhibitors inhibit glucose uptake into skeletal muscle and adipose tissue that activate hepatic glycogenolysis. This event results in hyperglycemia due to the pancreas releasing very high levels of insulin into the blood (hyperinsulinemia) that subsequently reactivate PI3K signaling and cause resistance to PI3K inhibitors. The ketogenic diet reportedly minimized the hyperglycemia and hyperinsulinemia induced by the PI3K inhibitor and enhanced the efficacy of PI3K inhibitors in tumor models. Studies combining PI3K inhibitors and ketogenic diet are underway. Hence, combining the ketogenic diet with chemotherapy or other novel treatment should be the focus of ketogenic diet trials.12,13
Ketogenic Diet and Oncology Studies
The impact of the ketogenic diet on the growth of murine pancreatic tumors was evaluated by Yang and colleagues. The ketogenic diet decreased glucose concentration that enters the TCA cycle and increased fatty acid oxidation that produces β-hydroxybutyrate. This event promotes the generation of ATP, although with only modest elevations of NADH with less impact on tumor growth. The combination of ketogenic diet and standard chemotherapy substantially raised tumor NADH and suppressed the growth of murine tumor cells, they noted.14 Furukawa and colleagues compared 10 patients with metastatic colon cancer receiving chemotherapy plus the modified medium-chain triglyceride ketogenic diet for 1 year with 14 patients receiving chemotherapy only. The ketogenic diet group exhibited a response rate of 60% with 5 patients achieving a complete response and a disease control rate of 70%, while the chemotherapy-alone group showed a response rate of only 21% with no complete response and a disease control rate of 64%.15
The ketogenic diet also reportedly stimulates cytokine and CD4+ and CD8+ T-cell production that stimulates T-cell killing activity. The ketogenic diet may overcome several immune escape mechanisms by downregulating the expression of cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein 1 (PD-1) on tumor-infiltrating lymphocytes.16 Our patient tolerated the combination of the ketogenic diet with ipilimumab (CTLA-4 inhibitor) and nivolumab (PD-1 inhibitor) without significant toxicities and stabilization of his disease.
Future Directions
We originally presented the abstract and poster of this case report at the Association of VA Hematology/Oncology annual meeting in San Diego, California, in September 2022.17 Based on our previous experience, we are now using a modified Atkins diet, which is a less strict diet consisting of 60% fat, 30% protein, and 10% carbohydrates combined with chemotherapy and/or immunotherapy. The composition of fat to carbohydrate plus protein in the traditional ketogenic diet is usually 4:1 or 3:1, while in modified Atkins diet the ratio is 1:1 or 2:1. The benefit of the modified Atkins diet is that patients can consume more protein than a strict ketogenic diet and they can be more liberal in carbohydrate allowances. We are about to open a study protocol of combining a modified Atkin diet and chemotherapy and/or immunotherapy as a first-line treatment for veterans with all types of advanced or metastatic solid tumors at VACCHCS. The study protocol was approved by the VA Office of Research and Development and has been submitted to the VACCHCS Institutional Review Board for review. Once approved, we will start patient recruitment.
CONCLUSIONS
Cancer cells have defects in their mitochondria that prevent them from generating energy for metabolism in the absence of glucose. They also depend on the PI3K signaling pathway to survive. The ketogenic diet has the advantage of affecting cancer cell growth by exploiting these mitochondrial defects and blocking hyperglycemia. There is growing evidence that the ketogenic diet is feasible, tolerable, and reportedly inhibits cancer growth. Our case report and previous publications suggest that the ketogenic diet can be added to chemotherapy and/or immunotherapy as an adjunct to standard-of-care cancer treatment while maintaining good QOL. We are planning to open a clinical trial using the modified Atkins diet in conjunction with active cancer treatments as first-line therapy for metastatic solid tumors at the VACCHCS. We are also working closely with researchers from several veteran hospitals to do a diet collaborative study. We believe the ketogenic diet is an important part of cancer treatment and has a promising future. More research should be dedicated to this very interesting field.
Acknowledgments
We previously presented this case report in an abstract and poster at the September 2022 AVAHO meeting in San Diego, California.
1. Tan-Shalaby JL, Carrick J, Edinger K, et al. Modified Atkins diet in advanced malignancies-final results of a safety and feasibility trial within the Veterans Affairs Pittsburgh Healthcare System. Nutr Metab (Lond). 2016;13:52. Published 2016 Aug 12. doi:10.1186/s12986-016-0113-y
2. Talib WH, Mahmod AI, Kamal A, et al. Ketogenic diet in cancer prevention and therapy: molecular targets and therapeutic opportunities. Curr Issues Mol Biol. 2021;43(2):558-589. Published 2021 Jul 3. doi:10.3390/cimb43020042
3. Tan-Shalaby J. Ketogenic diets and cancer: emerging evidence. Fed Pract. 2017;34(suppl 1):37S-42S.
4. Cortez NE, Mackenzie GG. Ketogenic diets in pancreatic cancer and associated cachexia: cellular mechanisms and clinical perspectives. Nutrients. 2021;13(9):3202. Published 2021 Sep 15. doi:10.3390/nu13093202
5. Tabernero J, Grothey A, Van Cutsem E, et al. Encorafenib plus cetuximab as a new standard of care for previously treated BRAF V600E-mutant metastatic colorectal cancer: updated survival results and subgroup analyses from the BEACON study. J Clin Oncol. 2021;39(4):273-284. doi:10.1200/JCO.20.02088
6. André T, Lonardi S, Wong KYM, et al. Nivolumab plus low-dose ipilimumab in previously treated patients with microsatellite instability-high/mismatch repair-deficient metastatic colorectal cancer: 4-year follow-up from CheckMate 142. Ann Oncol. 2022;33(10):1052-1060. doi:10.1016/j.annonc.2022.06.008
7. Grassi E, Corbelli J, Papiani G, Barbera MA, Gazzaneo F, Tamberi S. Current therapeutic strategies in BRAF-mutant metastatic colorectal cancer. Front Oncol. 2021;11:601722. Published 2021 Jun 23. doi:10.3389/fonc.2021.601722
8. Seyfried TN, Mukherjee P, Iyikesici MS, et al. Consideration of ketogenic metabolic therapy as a complementary or alternative approach for managing breast cancer. Front Nutr. 2020;7:21. Published 2020 Mar 11. doi:10.3389/fnut.2020.00021
9. Meidenbauer JJ, Mukherjee P, Seyfried TN. The glucose ketone index calculator: a simple tool to monitor therapeutic efficacy for metabolic management of brain cancer. Nutr Metab (Lond). 2015;12:12. Published 2015 Mar 11. doi:10.1186/s12986-015-0009-2
10. Fayers P, Bottomley A; EORTC Quality of Life Group; Quality of Life Unit. Quality of life research within the EORTC-the EORTC QLQ-C30. European Organisation for Research and Treatment of Cancer. Eur J Cancer. 2002;38(suppl 4):S125-S133. doi:10.1016/s0959-8049(01)00448-8
11. Yang J, Nie J, Ma X, Wei Y, Peng Y, Wei X. Targeting PI3K in cancer: mechanisms and advances in clinical trials. Mol Cancer. 2019;18(1):26. Published 2019 Feb 19. doi:10.1186/s12943-019-0954-x
12. Goncalves MD, Hopkins BD, Cantley LC. Phosphatidylinositol 3-kinase, growth disorders, and cancer. N Engl J Med. 2018;379(21):2052-2062. doi:10.1056/NEJMra1704560
13. Weber DD, Aminzadeh-Gohari S, Tulipan J, Catalano L, Feichtinger RG, Kofler B. Ketogenic diet in the treatment of cancer-where do we stand?. Mol Metab. 2020;33:102-121. doi:10.1016/j.molmet.2019.06.026
14. Yang L, TeSlaa T, Ng S, et al. Ketogenic diet and chemotherapy combine to disrupt pancreatic cancer metabolism and growth. Med. 2022;3(2):119-136. doi:10.1016/j.medj.2021.12.008
15. Furukawa K, Shigematus K, Iwase Y, et al. Clinical effects of one year of chemotherapy with a modified medium-chain triglyceride ketogenic diet on the recurrence of stage IV colon cancer. J Clin Oncol. 2018;36(suppl 15):e15709. doi:10.1200/JCO.2018.36.15_suppl.e15709
16. Zhang X, Li H, Lv X, et al. Impact of diets on response to immune checkpoint inhibitors (ICIs) therapy against tumors. Life (Basel). 2022;12(3):409. Published 2022 Mar 11. doi:10.3390/life12030409
17. Liman, A, Hwang A, Means J, Newson J. Ketogenic diet and cancer: a case report and feasibility study at VA Central California Healthcare System. Fed Pract. 2022;39(suppl 4):S18.
Originally developed for the treatment of refractory epilepsy, the ketogenic diet is distinguished by its high-fat, moderate-protein, and low-carbohydrate food program. Preclinical models provide emerging evidence that a ketogenic diet can have therapeutic potential for a broad range of cancers. The Warburg effect is a condition where cancer cells increase the uptake and fermentation of glucose to produce lactate for their metabolism, which is called aerobic glycolysis. Lactate is the key driver of cancer angiogenesis and proliferation.1,2
The ketogenic diet promotes a metabolic shift from glycolysis to mitochondrial metabolism in normal cells while cancer cells have dysfunction in their mitochondria due to damage in cellular respiration. The ketogenic diet creates a metabolic state whereby blood glucose levels are reduced, and blood ketone bodies (D-β-hydroxybutyrate and acetoacetate) are elevated. In normal cells, the ketogenic diet causes a decrease in glucose intake for glycolysis, which makes them unable to produce enough substrate to enter the tricarboxylic acid (TCA) cycle for adenosine triphosphate (ATP) production. Fatty acid oxidation plays a key role in ketone body synthesis as a “super fuel” that enter the TCA cycle as an alternative pathway to generate ATP. On the other hand, cancer cells are unable to use ketone bodies to produce ATP for energy and metabolism due to mitochondrial defects. Lack of energy subsequently leads to the inhibition of proliferation and survival of cancer cells.3,4
We previously published a safety and feasibility study of the Modified Atkins Diet in metastatic cancer patients after failure of chemotherapy at the US Department of Veterans Affairs (VA) Pittsburgh Healthcare System.1 None of the patients were on chemotherapy at the time of enrollment. The Modified Atkins Diet consists of 60% fat, 30% protein, and 10% carbohydrates and is more tolerable than the ketogenic diet due to higher amounts of protein. Six of 11 patients (54%) had stable disease and partial response on positron emission tomography/computed tomography (PET/CT). Our study showed that patients who lost at least 10% of their body weight had improvement in quality of life (QOL) and cancer response.1 Here we present a case of a veteran with extensive metastatic colon cancer on concurrent ketogenic diet and chemotherapy subsequently followed by concurrent ketogenic diet and immunotherapy at Veterans Affairs Central California Health Care Systems (VACCHCS) in Fresno.
CASE PRESENTATION
A 69-year-old veteran had iron deficiency anemia (hemoglobin, 6.5 g/dL) about 5 years previously. He underwent a colonoscopy that revealed a near circumferential ulcerated mass measuring 7 cm in the transverse colon. Biopsy results showed mucinous adenocarcinoma of the colon with a foci of signet ring cells (Figure 2).
The patient received adjuvant treatment with FOLFOX (fluorouracil, leucovorin calcium, and oxaliplatin), but within several months he developed pancreatic and worsening omental metastasis seen on PET/CT. He was then started on FOLFIRI (fluorouracil, leucovorin calcium, and irinotecan hydrochloride) plus bevacizumab 16 months after his initial diagnosis. He underwent a pancreatic mastectomy that confirmed adenocarcinoma 9 months later. Afterward, he briefly resumed FOLFIRI and bevacizumab. Next-generation sequencing testing with Foundation One CDx revealed a wild-type (WT) KRAS with a high degree of tumor mutation burden of 37 muts/Mb, BRAF V600E mutation, and high microsatellite instability (MSI-H).
Due to disease progression, the patient’s treatment was changed to encorafenib and cetuximab for 4 months before progressing again with new liver mass and mediastinal lymphadenopathy. He then received pembrolizumab for 4 months until PET/CT showed progression and his carcinoembryonic antigen (CEA) increased from 95 to 1031 ng/mL by January 2021 (Figure 4).
The patient was started on trifluridine/tipiracil, and bevacizumab while concurrently initiating the ketogenic diet in January 2021. Laboratory tests drawn after 1 week of strict dietary ketogenic diet adherence showed low-level ketosis with a glucose ketone index (GKI) of 8.2 (Table 1).
A follow-up PET/CT showed disease progression along with a CEA of 94 ng/mL after 10 months of chemotherapy plus the ketogenic diet (Table 2).
The patient continued to experience excellent QOL based on the QOL Eastern Cooperative Oncology Group (ECOG) core quality of life questionnaire (QLC-C30) forms, which he completed every 3 months. Twenty-two months after starting the ketogenic diet, the patient’s CEA increased to 293 ng/mL although PET/CT continues to show stable disease (Figures 4, 5, and 6).
DISCUSSION
The purpose of this case report is to describe whether a patient receiving active cancer treatment was able to tolerate the ketogenic diet in conjunction with chemotherapy or immunotherapy. Most literature published on the subject evaluated the tolerability and response of the ketogenic diet after the failure of standard therapy. Our patient was diagnosed with stage III mucinous colon adenocarcinoma. He received adjuvant chemotherapy but quickly developed metastatic disease to the pancreas and omentum. We started him on encorafenib and cetuximab based on the BEACON study that showed improvement in response rate and survival when compared with standard chemotherapy for patients with BRAF V600E mutation.5 Unfortunately, his cancer quickly progressed within 4 months and again did not respond to pembrolizumab despite MSI-H, which lasted for another 4 months.
We suggested the ketogenic diet and the patient agreed. He started the diet along with trifluridine/tipiracil, and bevacizumab in January 2021. The patient’s metastatic cancer stabilized for 9 months until his disease progressed again. He was started on doublet immune checkpoint inhibitors ipilimumab and nivolumab based on his MSI-H and high tumor mutation burden with the continuation of the ketogenic diet until now. The CheckMate 142 study revealed that the combination of ipilimumab and nivolumab in patients with MSI-H previously treated for metastatic colon cancer showed some benefit.6
Our patient had the loss of nuclear expression of MLH1 and PMS2 (zero tumor stained) but no evidence of the loss expression of MSH2 and MSH6 genes (99% tumor stained). About 8% to 12% of patients with metastatic colon cancer have BRAF V600E mutations that are usually mucinous type, poorly differentiated, and located in the right side of the colon, which portends to a poor prognosis. Tumor DNA mismatch repair damage results in genetic hypermutability and leads to MSI that is sensitive to treatment with checkpoint inhibitors, as in our patient. Only about 3% of MSI-H tumors are due to germline mutations such as Lynch syndrome (hereditary nonpolyposis colorectal cancer). The presence of both MLH1 hypermethylation and BRAF mutation, as in our patient, is a strong indication of somatic rather than germline mutation.7
GKI, which represents the ratio of glucose to ketone, was developed to evaluate the efficacy of the ketogenic diet. This index measures the degree of metabolic stress on tumor cells through the decrease of glucose levels and increase of ketone bodies. A GKI of ≤ 1.0 has been suggested as the ideal therapeutic goal for cancer management.8 As levels of blood glucose decline, the blood levels of ketone bodies should rise. These 2 lines should eventually intersect at a certain point beyond which one enters the therapeutic zone or therapeutic ketosis zone. This is when tumor growth is expected to slow or cease.9 The patient’s ketone (β-hydroxybutyrate) level was initially high (0.71 mmol/L) with a GKI of 8.2. (low ketotic level), which meant he tolerated a rather strict diet for the first several months. This was also reflected in his 18 lb weight loss (almost 10% of body weight) and cancer stabilization, as in our previous publication.1 Unfortunately, the patient was unable to maintain high ketone and lower GKI levels due to fatigue from depleted carbohydrate intake. He added some carbohydrate snacks in between meals, which improved the fatigue. His ketone level has been < 0.5 mmol/L ever since, albeit his disease continues to be stable. The patient continues his daily work and reports a better QOL, based on the ECOG QLC-C30 form that he completed every 3 months.10 Currently, the patient is still receiving ipilimumab and nivolumab while maintaining the ketogenic diet with stable metastatic disease on PET/CT.
Ketogenic Diet and Cellular Mechanism of Action
PI3K/Akt (phosphatidylinositol-3-kinase) signaling is one of the most important intracellular pathways for tumor cells. It leads to the inhibition of apoptosis and the promotion of cell proliferation, metabolism, and angiogenesis. Deregulation of the PI3K pathway either via amplification of PI3K by tyrosine kinase growth factor receptors or inactivation of the tumor suppressor phosphatase and tensin homolog (PTEN), which is the negative regulator of the PI3K pathway, contributes to the development of cancer cells.11
A study by Goncalves and colleagues revealed an interesting relationship between the PI3K pathway and the benefit of the ketogenic diet to slow tumor growth. PI3K inhibitors inhibit glucose uptake into skeletal muscle and adipose tissue that activate hepatic glycogenolysis. This event results in hyperglycemia due to the pancreas releasing very high levels of insulin into the blood (hyperinsulinemia) that subsequently reactivate PI3K signaling and cause resistance to PI3K inhibitors. The ketogenic diet reportedly minimized the hyperglycemia and hyperinsulinemia induced by the PI3K inhibitor and enhanced the efficacy of PI3K inhibitors in tumor models. Studies combining PI3K inhibitors and ketogenic diet are underway. Hence, combining the ketogenic diet with chemotherapy or other novel treatment should be the focus of ketogenic diet trials.12,13
Ketogenic Diet and Oncology Studies
The impact of the ketogenic diet on the growth of murine pancreatic tumors was evaluated by Yang and colleagues. The ketogenic diet decreased glucose concentration that enters the TCA cycle and increased fatty acid oxidation that produces β-hydroxybutyrate. This event promotes the generation of ATP, although with only modest elevations of NADH with less impact on tumor growth. The combination of ketogenic diet and standard chemotherapy substantially raised tumor NADH and suppressed the growth of murine tumor cells, they noted.14 Furukawa and colleagues compared 10 patients with metastatic colon cancer receiving chemotherapy plus the modified medium-chain triglyceride ketogenic diet for 1 year with 14 patients receiving chemotherapy only. The ketogenic diet group exhibited a response rate of 60% with 5 patients achieving a complete response and a disease control rate of 70%, while the chemotherapy-alone group showed a response rate of only 21% with no complete response and a disease control rate of 64%.15
The ketogenic diet also reportedly stimulates cytokine and CD4+ and CD8+ T-cell production that stimulates T-cell killing activity. The ketogenic diet may overcome several immune escape mechanisms by downregulating the expression of cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein 1 (PD-1) on tumor-infiltrating lymphocytes.16 Our patient tolerated the combination of the ketogenic diet with ipilimumab (CTLA-4 inhibitor) and nivolumab (PD-1 inhibitor) without significant toxicities and stabilization of his disease.
Future Directions
We originally presented the abstract and poster of this case report at the Association of VA Hematology/Oncology annual meeting in San Diego, California, in September 2022.17 Based on our previous experience, we are now using a modified Atkins diet, which is a less strict diet consisting of 60% fat, 30% protein, and 10% carbohydrates combined with chemotherapy and/or immunotherapy. The composition of fat to carbohydrate plus protein in the traditional ketogenic diet is usually 4:1 or 3:1, while in modified Atkins diet the ratio is 1:1 or 2:1. The benefit of the modified Atkins diet is that patients can consume more protein than a strict ketogenic diet and they can be more liberal in carbohydrate allowances. We are about to open a study protocol of combining a modified Atkin diet and chemotherapy and/or immunotherapy as a first-line treatment for veterans with all types of advanced or metastatic solid tumors at VACCHCS. The study protocol was approved by the VA Office of Research and Development and has been submitted to the VACCHCS Institutional Review Board for review. Once approved, we will start patient recruitment.
CONCLUSIONS
Cancer cells have defects in their mitochondria that prevent them from generating energy for metabolism in the absence of glucose. They also depend on the PI3K signaling pathway to survive. The ketogenic diet has the advantage of affecting cancer cell growth by exploiting these mitochondrial defects and blocking hyperglycemia. There is growing evidence that the ketogenic diet is feasible, tolerable, and reportedly inhibits cancer growth. Our case report and previous publications suggest that the ketogenic diet can be added to chemotherapy and/or immunotherapy as an adjunct to standard-of-care cancer treatment while maintaining good QOL. We are planning to open a clinical trial using the modified Atkins diet in conjunction with active cancer treatments as first-line therapy for metastatic solid tumors at the VACCHCS. We are also working closely with researchers from several veteran hospitals to do a diet collaborative study. We believe the ketogenic diet is an important part of cancer treatment and has a promising future. More research should be dedicated to this very interesting field.
Acknowledgments
We previously presented this case report in an abstract and poster at the September 2022 AVAHO meeting in San Diego, California.
Originally developed for the treatment of refractory epilepsy, the ketogenic diet is distinguished by its high-fat, moderate-protein, and low-carbohydrate food program. Preclinical models provide emerging evidence that a ketogenic diet can have therapeutic potential for a broad range of cancers. The Warburg effect is a condition where cancer cells increase the uptake and fermentation of glucose to produce lactate for their metabolism, which is called aerobic glycolysis. Lactate is the key driver of cancer angiogenesis and proliferation.1,2
The ketogenic diet promotes a metabolic shift from glycolysis to mitochondrial metabolism in normal cells while cancer cells have dysfunction in their mitochondria due to damage in cellular respiration. The ketogenic diet creates a metabolic state whereby blood glucose levels are reduced, and blood ketone bodies (D-β-hydroxybutyrate and acetoacetate) are elevated. In normal cells, the ketogenic diet causes a decrease in glucose intake for glycolysis, which makes them unable to produce enough substrate to enter the tricarboxylic acid (TCA) cycle for adenosine triphosphate (ATP) production. Fatty acid oxidation plays a key role in ketone body synthesis as a “super fuel” that enter the TCA cycle as an alternative pathway to generate ATP. On the other hand, cancer cells are unable to use ketone bodies to produce ATP for energy and metabolism due to mitochondrial defects. Lack of energy subsequently leads to the inhibition of proliferation and survival of cancer cells.3,4
We previously published a safety and feasibility study of the Modified Atkins Diet in metastatic cancer patients after failure of chemotherapy at the US Department of Veterans Affairs (VA) Pittsburgh Healthcare System.1 None of the patients were on chemotherapy at the time of enrollment. The Modified Atkins Diet consists of 60% fat, 30% protein, and 10% carbohydrates and is more tolerable than the ketogenic diet due to higher amounts of protein. Six of 11 patients (54%) had stable disease and partial response on positron emission tomography/computed tomography (PET/CT). Our study showed that patients who lost at least 10% of their body weight had improvement in quality of life (QOL) and cancer response.1 Here we present a case of a veteran with extensive metastatic colon cancer on concurrent ketogenic diet and chemotherapy subsequently followed by concurrent ketogenic diet and immunotherapy at Veterans Affairs Central California Health Care Systems (VACCHCS) in Fresno.
CASE PRESENTATION
A 69-year-old veteran had iron deficiency anemia (hemoglobin, 6.5 g/dL) about 5 years previously. He underwent a colonoscopy that revealed a near circumferential ulcerated mass measuring 7 cm in the transverse colon. Biopsy results showed mucinous adenocarcinoma of the colon with a foci of signet ring cells (Figure 2).
The patient received adjuvant treatment with FOLFOX (fluorouracil, leucovorin calcium, and oxaliplatin), but within several months he developed pancreatic and worsening omental metastasis seen on PET/CT. He was then started on FOLFIRI (fluorouracil, leucovorin calcium, and irinotecan hydrochloride) plus bevacizumab 16 months after his initial diagnosis. He underwent a pancreatic mastectomy that confirmed adenocarcinoma 9 months later. Afterward, he briefly resumed FOLFIRI and bevacizumab. Next-generation sequencing testing with Foundation One CDx revealed a wild-type (WT) KRAS with a high degree of tumor mutation burden of 37 muts/Mb, BRAF V600E mutation, and high microsatellite instability (MSI-H).
Due to disease progression, the patient’s treatment was changed to encorafenib and cetuximab for 4 months before progressing again with new liver mass and mediastinal lymphadenopathy. He then received pembrolizumab for 4 months until PET/CT showed progression and his carcinoembryonic antigen (CEA) increased from 95 to 1031 ng/mL by January 2021 (Figure 4).
The patient was started on trifluridine/tipiracil, and bevacizumab while concurrently initiating the ketogenic diet in January 2021. Laboratory tests drawn after 1 week of strict dietary ketogenic diet adherence showed low-level ketosis with a glucose ketone index (GKI) of 8.2 (Table 1).
A follow-up PET/CT showed disease progression along with a CEA of 94 ng/mL after 10 months of chemotherapy plus the ketogenic diet (Table 2).
The patient continued to experience excellent QOL based on the QOL Eastern Cooperative Oncology Group (ECOG) core quality of life questionnaire (QLC-C30) forms, which he completed every 3 months. Twenty-two months after starting the ketogenic diet, the patient’s CEA increased to 293 ng/mL although PET/CT continues to show stable disease (Figures 4, 5, and 6).
DISCUSSION
The purpose of this case report is to describe whether a patient receiving active cancer treatment was able to tolerate the ketogenic diet in conjunction with chemotherapy or immunotherapy. Most literature published on the subject evaluated the tolerability and response of the ketogenic diet after the failure of standard therapy. Our patient was diagnosed with stage III mucinous colon adenocarcinoma. He received adjuvant chemotherapy but quickly developed metastatic disease to the pancreas and omentum. We started him on encorafenib and cetuximab based on the BEACON study that showed improvement in response rate and survival when compared with standard chemotherapy for patients with BRAF V600E mutation.5 Unfortunately, his cancer quickly progressed within 4 months and again did not respond to pembrolizumab despite MSI-H, which lasted for another 4 months.
We suggested the ketogenic diet and the patient agreed. He started the diet along with trifluridine/tipiracil, and bevacizumab in January 2021. The patient’s metastatic cancer stabilized for 9 months until his disease progressed again. He was started on doublet immune checkpoint inhibitors ipilimumab and nivolumab based on his MSI-H and high tumor mutation burden with the continuation of the ketogenic diet until now. The CheckMate 142 study revealed that the combination of ipilimumab and nivolumab in patients with MSI-H previously treated for metastatic colon cancer showed some benefit.6
Our patient had the loss of nuclear expression of MLH1 and PMS2 (zero tumor stained) but no evidence of the loss expression of MSH2 and MSH6 genes (99% tumor stained). About 8% to 12% of patients with metastatic colon cancer have BRAF V600E mutations that are usually mucinous type, poorly differentiated, and located in the right side of the colon, which portends to a poor prognosis. Tumor DNA mismatch repair damage results in genetic hypermutability and leads to MSI that is sensitive to treatment with checkpoint inhibitors, as in our patient. Only about 3% of MSI-H tumors are due to germline mutations such as Lynch syndrome (hereditary nonpolyposis colorectal cancer). The presence of both MLH1 hypermethylation and BRAF mutation, as in our patient, is a strong indication of somatic rather than germline mutation.7
GKI, which represents the ratio of glucose to ketone, was developed to evaluate the efficacy of the ketogenic diet. This index measures the degree of metabolic stress on tumor cells through the decrease of glucose levels and increase of ketone bodies. A GKI of ≤ 1.0 has been suggested as the ideal therapeutic goal for cancer management.8 As levels of blood glucose decline, the blood levels of ketone bodies should rise. These 2 lines should eventually intersect at a certain point beyond which one enters the therapeutic zone or therapeutic ketosis zone. This is when tumor growth is expected to slow or cease.9 The patient’s ketone (β-hydroxybutyrate) level was initially high (0.71 mmol/L) with a GKI of 8.2. (low ketotic level), which meant he tolerated a rather strict diet for the first several months. This was also reflected in his 18 lb weight loss (almost 10% of body weight) and cancer stabilization, as in our previous publication.1 Unfortunately, the patient was unable to maintain high ketone and lower GKI levels due to fatigue from depleted carbohydrate intake. He added some carbohydrate snacks in between meals, which improved the fatigue. His ketone level has been < 0.5 mmol/L ever since, albeit his disease continues to be stable. The patient continues his daily work and reports a better QOL, based on the ECOG QLC-C30 form that he completed every 3 months.10 Currently, the patient is still receiving ipilimumab and nivolumab while maintaining the ketogenic diet with stable metastatic disease on PET/CT.
Ketogenic Diet and Cellular Mechanism of Action
PI3K/Akt (phosphatidylinositol-3-kinase) signaling is one of the most important intracellular pathways for tumor cells. It leads to the inhibition of apoptosis and the promotion of cell proliferation, metabolism, and angiogenesis. Deregulation of the PI3K pathway either via amplification of PI3K by tyrosine kinase growth factor receptors or inactivation of the tumor suppressor phosphatase and tensin homolog (PTEN), which is the negative regulator of the PI3K pathway, contributes to the development of cancer cells.11
A study by Goncalves and colleagues revealed an interesting relationship between the PI3K pathway and the benefit of the ketogenic diet to slow tumor growth. PI3K inhibitors inhibit glucose uptake into skeletal muscle and adipose tissue that activate hepatic glycogenolysis. This event results in hyperglycemia due to the pancreas releasing very high levels of insulin into the blood (hyperinsulinemia) that subsequently reactivate PI3K signaling and cause resistance to PI3K inhibitors. The ketogenic diet reportedly minimized the hyperglycemia and hyperinsulinemia induced by the PI3K inhibitor and enhanced the efficacy of PI3K inhibitors in tumor models. Studies combining PI3K inhibitors and ketogenic diet are underway. Hence, combining the ketogenic diet with chemotherapy or other novel treatment should be the focus of ketogenic diet trials.12,13
Ketogenic Diet and Oncology Studies
The impact of the ketogenic diet on the growth of murine pancreatic tumors was evaluated by Yang and colleagues. The ketogenic diet decreased glucose concentration that enters the TCA cycle and increased fatty acid oxidation that produces β-hydroxybutyrate. This event promotes the generation of ATP, although with only modest elevations of NADH with less impact on tumor growth. The combination of ketogenic diet and standard chemotherapy substantially raised tumor NADH and suppressed the growth of murine tumor cells, they noted.14 Furukawa and colleagues compared 10 patients with metastatic colon cancer receiving chemotherapy plus the modified medium-chain triglyceride ketogenic diet for 1 year with 14 patients receiving chemotherapy only. The ketogenic diet group exhibited a response rate of 60% with 5 patients achieving a complete response and a disease control rate of 70%, while the chemotherapy-alone group showed a response rate of only 21% with no complete response and a disease control rate of 64%.15
The ketogenic diet also reportedly stimulates cytokine and CD4+ and CD8+ T-cell production that stimulates T-cell killing activity. The ketogenic diet may overcome several immune escape mechanisms by downregulating the expression of cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein 1 (PD-1) on tumor-infiltrating lymphocytes.16 Our patient tolerated the combination of the ketogenic diet with ipilimumab (CTLA-4 inhibitor) and nivolumab (PD-1 inhibitor) without significant toxicities and stabilization of his disease.
Future Directions
We originally presented the abstract and poster of this case report at the Association of VA Hematology/Oncology annual meeting in San Diego, California, in September 2022.17 Based on our previous experience, we are now using a modified Atkins diet, which is a less strict diet consisting of 60% fat, 30% protein, and 10% carbohydrates combined with chemotherapy and/or immunotherapy. The composition of fat to carbohydrate plus protein in the traditional ketogenic diet is usually 4:1 or 3:1, while in modified Atkins diet the ratio is 1:1 or 2:1. The benefit of the modified Atkins diet is that patients can consume more protein than a strict ketogenic diet and they can be more liberal in carbohydrate allowances. We are about to open a study protocol of combining a modified Atkin diet and chemotherapy and/or immunotherapy as a first-line treatment for veterans with all types of advanced or metastatic solid tumors at VACCHCS. The study protocol was approved by the VA Office of Research and Development and has been submitted to the VACCHCS Institutional Review Board for review. Once approved, we will start patient recruitment.
CONCLUSIONS
Cancer cells have defects in their mitochondria that prevent them from generating energy for metabolism in the absence of glucose. They also depend on the PI3K signaling pathway to survive. The ketogenic diet has the advantage of affecting cancer cell growth by exploiting these mitochondrial defects and blocking hyperglycemia. There is growing evidence that the ketogenic diet is feasible, tolerable, and reportedly inhibits cancer growth. Our case report and previous publications suggest that the ketogenic diet can be added to chemotherapy and/or immunotherapy as an adjunct to standard-of-care cancer treatment while maintaining good QOL. We are planning to open a clinical trial using the modified Atkins diet in conjunction with active cancer treatments as first-line therapy for metastatic solid tumors at the VACCHCS. We are also working closely with researchers from several veteran hospitals to do a diet collaborative study. We believe the ketogenic diet is an important part of cancer treatment and has a promising future. More research should be dedicated to this very interesting field.
Acknowledgments
We previously presented this case report in an abstract and poster at the September 2022 AVAHO meeting in San Diego, California.
1. Tan-Shalaby JL, Carrick J, Edinger K, et al. Modified Atkins diet in advanced malignancies-final results of a safety and feasibility trial within the Veterans Affairs Pittsburgh Healthcare System. Nutr Metab (Lond). 2016;13:52. Published 2016 Aug 12. doi:10.1186/s12986-016-0113-y
2. Talib WH, Mahmod AI, Kamal A, et al. Ketogenic diet in cancer prevention and therapy: molecular targets and therapeutic opportunities. Curr Issues Mol Biol. 2021;43(2):558-589. Published 2021 Jul 3. doi:10.3390/cimb43020042
3. Tan-Shalaby J. Ketogenic diets and cancer: emerging evidence. Fed Pract. 2017;34(suppl 1):37S-42S.
4. Cortez NE, Mackenzie GG. Ketogenic diets in pancreatic cancer and associated cachexia: cellular mechanisms and clinical perspectives. Nutrients. 2021;13(9):3202. Published 2021 Sep 15. doi:10.3390/nu13093202
5. Tabernero J, Grothey A, Van Cutsem E, et al. Encorafenib plus cetuximab as a new standard of care for previously treated BRAF V600E-mutant metastatic colorectal cancer: updated survival results and subgroup analyses from the BEACON study. J Clin Oncol. 2021;39(4):273-284. doi:10.1200/JCO.20.02088
6. André T, Lonardi S, Wong KYM, et al. Nivolumab plus low-dose ipilimumab in previously treated patients with microsatellite instability-high/mismatch repair-deficient metastatic colorectal cancer: 4-year follow-up from CheckMate 142. Ann Oncol. 2022;33(10):1052-1060. doi:10.1016/j.annonc.2022.06.008
7. Grassi E, Corbelli J, Papiani G, Barbera MA, Gazzaneo F, Tamberi S. Current therapeutic strategies in BRAF-mutant metastatic colorectal cancer. Front Oncol. 2021;11:601722. Published 2021 Jun 23. doi:10.3389/fonc.2021.601722
8. Seyfried TN, Mukherjee P, Iyikesici MS, et al. Consideration of ketogenic metabolic therapy as a complementary or alternative approach for managing breast cancer. Front Nutr. 2020;7:21. Published 2020 Mar 11. doi:10.3389/fnut.2020.00021
9. Meidenbauer JJ, Mukherjee P, Seyfried TN. The glucose ketone index calculator: a simple tool to monitor therapeutic efficacy for metabolic management of brain cancer. Nutr Metab (Lond). 2015;12:12. Published 2015 Mar 11. doi:10.1186/s12986-015-0009-2
10. Fayers P, Bottomley A; EORTC Quality of Life Group; Quality of Life Unit. Quality of life research within the EORTC-the EORTC QLQ-C30. European Organisation for Research and Treatment of Cancer. Eur J Cancer. 2002;38(suppl 4):S125-S133. doi:10.1016/s0959-8049(01)00448-8
11. Yang J, Nie J, Ma X, Wei Y, Peng Y, Wei X. Targeting PI3K in cancer: mechanisms and advances in clinical trials. Mol Cancer. 2019;18(1):26. Published 2019 Feb 19. doi:10.1186/s12943-019-0954-x
12. Goncalves MD, Hopkins BD, Cantley LC. Phosphatidylinositol 3-kinase, growth disorders, and cancer. N Engl J Med. 2018;379(21):2052-2062. doi:10.1056/NEJMra1704560
13. Weber DD, Aminzadeh-Gohari S, Tulipan J, Catalano L, Feichtinger RG, Kofler B. Ketogenic diet in the treatment of cancer-where do we stand?. Mol Metab. 2020;33:102-121. doi:10.1016/j.molmet.2019.06.026
14. Yang L, TeSlaa T, Ng S, et al. Ketogenic diet and chemotherapy combine to disrupt pancreatic cancer metabolism and growth. Med. 2022;3(2):119-136. doi:10.1016/j.medj.2021.12.008
15. Furukawa K, Shigematus K, Iwase Y, et al. Clinical effects of one year of chemotherapy with a modified medium-chain triglyceride ketogenic diet on the recurrence of stage IV colon cancer. J Clin Oncol. 2018;36(suppl 15):e15709. doi:10.1200/JCO.2018.36.15_suppl.e15709
16. Zhang X, Li H, Lv X, et al. Impact of diets on response to immune checkpoint inhibitors (ICIs) therapy against tumors. Life (Basel). 2022;12(3):409. Published 2022 Mar 11. doi:10.3390/life12030409
17. Liman, A, Hwang A, Means J, Newson J. Ketogenic diet and cancer: a case report and feasibility study at VA Central California Healthcare System. Fed Pract. 2022;39(suppl 4):S18.
1. Tan-Shalaby JL, Carrick J, Edinger K, et al. Modified Atkins diet in advanced malignancies-final results of a safety and feasibility trial within the Veterans Affairs Pittsburgh Healthcare System. Nutr Metab (Lond). 2016;13:52. Published 2016 Aug 12. doi:10.1186/s12986-016-0113-y
2. Talib WH, Mahmod AI, Kamal A, et al. Ketogenic diet in cancer prevention and therapy: molecular targets and therapeutic opportunities. Curr Issues Mol Biol. 2021;43(2):558-589. Published 2021 Jul 3. doi:10.3390/cimb43020042
3. Tan-Shalaby J. Ketogenic diets and cancer: emerging evidence. Fed Pract. 2017;34(suppl 1):37S-42S.
4. Cortez NE, Mackenzie GG. Ketogenic diets in pancreatic cancer and associated cachexia: cellular mechanisms and clinical perspectives. Nutrients. 2021;13(9):3202. Published 2021 Sep 15. doi:10.3390/nu13093202
5. Tabernero J, Grothey A, Van Cutsem E, et al. Encorafenib plus cetuximab as a new standard of care for previously treated BRAF V600E-mutant metastatic colorectal cancer: updated survival results and subgroup analyses from the BEACON study. J Clin Oncol. 2021;39(4):273-284. doi:10.1200/JCO.20.02088
6. André T, Lonardi S, Wong KYM, et al. Nivolumab plus low-dose ipilimumab in previously treated patients with microsatellite instability-high/mismatch repair-deficient metastatic colorectal cancer: 4-year follow-up from CheckMate 142. Ann Oncol. 2022;33(10):1052-1060. doi:10.1016/j.annonc.2022.06.008
7. Grassi E, Corbelli J, Papiani G, Barbera MA, Gazzaneo F, Tamberi S. Current therapeutic strategies in BRAF-mutant metastatic colorectal cancer. Front Oncol. 2021;11:601722. Published 2021 Jun 23. doi:10.3389/fonc.2021.601722
8. Seyfried TN, Mukherjee P, Iyikesici MS, et al. Consideration of ketogenic metabolic therapy as a complementary or alternative approach for managing breast cancer. Front Nutr. 2020;7:21. Published 2020 Mar 11. doi:10.3389/fnut.2020.00021
9. Meidenbauer JJ, Mukherjee P, Seyfried TN. The glucose ketone index calculator: a simple tool to monitor therapeutic efficacy for metabolic management of brain cancer. Nutr Metab (Lond). 2015;12:12. Published 2015 Mar 11. doi:10.1186/s12986-015-0009-2
10. Fayers P, Bottomley A; EORTC Quality of Life Group; Quality of Life Unit. Quality of life research within the EORTC-the EORTC QLQ-C30. European Organisation for Research and Treatment of Cancer. Eur J Cancer. 2002;38(suppl 4):S125-S133. doi:10.1016/s0959-8049(01)00448-8
11. Yang J, Nie J, Ma X, Wei Y, Peng Y, Wei X. Targeting PI3K in cancer: mechanisms and advances in clinical trials. Mol Cancer. 2019;18(1):26. Published 2019 Feb 19. doi:10.1186/s12943-019-0954-x
12. Goncalves MD, Hopkins BD, Cantley LC. Phosphatidylinositol 3-kinase, growth disorders, and cancer. N Engl J Med. 2018;379(21):2052-2062. doi:10.1056/NEJMra1704560
13. Weber DD, Aminzadeh-Gohari S, Tulipan J, Catalano L, Feichtinger RG, Kofler B. Ketogenic diet in the treatment of cancer-where do we stand?. Mol Metab. 2020;33:102-121. doi:10.1016/j.molmet.2019.06.026
14. Yang L, TeSlaa T, Ng S, et al. Ketogenic diet and chemotherapy combine to disrupt pancreatic cancer metabolism and growth. Med. 2022;3(2):119-136. doi:10.1016/j.medj.2021.12.008
15. Furukawa K, Shigematus K, Iwase Y, et al. Clinical effects of one year of chemotherapy with a modified medium-chain triglyceride ketogenic diet on the recurrence of stage IV colon cancer. J Clin Oncol. 2018;36(suppl 15):e15709. doi:10.1200/JCO.2018.36.15_suppl.e15709
16. Zhang X, Li H, Lv X, et al. Impact of diets on response to immune checkpoint inhibitors (ICIs) therapy against tumors. Life (Basel). 2022;12(3):409. Published 2022 Mar 11. doi:10.3390/life12030409
17. Liman, A, Hwang A, Means J, Newson J. Ketogenic diet and cancer: a case report and feasibility study at VA Central California Healthcare System. Fed Pract. 2022;39(suppl 4):S18.
Naltrexone: a Novel Approach to Pruritus in Polycythemia Vera
P ruritus is a characteristic and often debilitating clinical manifestation reported by about 50% of patients with polycythemia vera (PV). The exact pathophysiology of PV-associated pruritus is poorly understood. The itch sensation may arise from a central phenomenon without skin itch receptor involvement, as is seen in opioid-induced pruritus, or peripherally via unmyelinated C fibers. Various interventions have been used with mixed results for symptom management in this patient population.1
Selective serotonin reuptake inhibitors (SSRIs), such as paroxetine and fluoxetine, have historically demonstrated some efficacy in treating PV-associated pruritus.2 Alongside SSRIs, phlebotomy, antihistamines, phototherapy, interferon a, and myelosuppressive medications also comprise the various current treatment options. In addition to lacking efficacy, antihistamines can cause somnolence, constipation, and xerostomia.3,4 Phlebotomy and cytoreductive therapy are often effective in controlling erythrocytosis but fail to alleviate the disabling pruritus.1,5,6 More recently, suboptimal symptom alleviation has prompted the discovery of agents that target the mammalian target of rapamycin (mTOR) and Janus kinase 2 (Jak2) pathways.1
Naltrexone is an opioid antagonist shown to suppress pruritus in various dermatologic pathologies involving histamine-independent pathways.3,7,8 A systematic search strategy identified 34 studies on PV-associated pruritus, its pathophysiology and interventions, and naltrexone as a therapeutic agent. Only 1 study in the literature has described the use of naltrexone for uremic and cholestatic pruritus.9 We describe the successful use of naltrexone monotherapy for the treatment of pruritus in a patient with PV.
Case Presentation
A 40-year-old man with Jak2-positive PV treated with ruxolitinib presented to the outpatient Michael E. DeBakey Veterans Affairs Medical Center Supportive Care Clinic in Houston, Texas, for severe refractory pruritus. Wheals manifested in pruritic regions of the patient’s skin without gross excoriations or erythema. Pruritus reportedly began diffusely across the posterior torso. Through the rapid progression of an episode lasting 30 to 45 minutes, the lesions and pruritus would spread to the anterior torso, extend to the upper extremities bilaterally, and finally descend to the lower extremities bilaterally. A persistent sensation of heat or warmth on the patient’s skin was present, and periodically, this would culminate in a burning sensation comparable to “lying flat on one’s back directly on a hornet’s nest…[followed by] a million stings” that was inconsistent with erythromelalgia given the absence of erythema. The intensity of the pruritic episodes was subjectively also described as “enough to make [him] want to jump off the roof of a building…[causing] moments of deep, deep frustration…[and] the worst of all the symptoms one may encounter because of [PV].”
Pruritus was exacerbated by sweating, heat, contact with any liquids on the skin, and sunburns, which doubled the intensity. The patient reported minimal, temporary relief with cannabidiol and cold fabric or air on his skin. His current regimen and nonpharmacologic efforts provided no relief and included oatmeal baths, cornstarch after showers, and patting instead of rubbing the skin with topical products. Trials with nonprescription diphenhydramine, loratadine, and calamine and zinc were not successful. He had not pursued phototherapy due to time limitations and travel constraints. He had a history of phlebotomies and hydroxyurea use, which he preferred to avoid and discontinued 1 year before presentation.
Despite improving hematocrit (< 45% goal) and platelet counts with ruxolitinib, the patient reported worsening pruritus that significantly impaired quality of life. His sleep and social and physical activities were hindered, preventing him from working. The patient’s active medications also included low-dose aspirin, sertraline, hydroxyzine, triamcinolone acetonide, and pregabalin for sciatica. Given persistent symptoms despite multimodal therapy and lifestyle modifications, the patient was started on naltrexone 25 mg daily, which provided immediate relief of symptoms. He continues to have adequate symptom control 2 years after naltrexone initiation.
Literature Review
A systematic search strategy was developed with the assistance of a medical librarian in Medline Ovid, using both Medical Subject Heading (MeSH) terms and synonymous keywords. The strategy was then translated to Embase, Web of Science, and Cochrane to extract publications investigating PV, pruritus, and/or naltrexone therapy. All searches were conducted on July 18, 2022, and the results of the literature review were as follows: 2 results from Medline Ovid; 34 results from Embase (2 were duplicates of Medline Ovid results); 3 results from Web of Science (all of which were duplicates of Medline Ovid or Embase results); and 0 results from Cochrane (Figure).
Discussion
Although pruritus is a common and often excruciating manifestation of PV, its pathophysiology remains unclear. Some patients with decreasing or newly normal hematocrit and hemoglobin levels have paradoxically experienced an intensification of their pruritus, which introduces erythropoietin signaling pathways as a potential mechanism of the symptom.8 However, iron replacement therapy for patients with exacerbated pruritus after phlebotomies has not demonstrated consistent relief of pruritus.8 Normalization of platelet levels also has not been historically associated with improvement of pruritus.8,9 It has been hypothesized that cells harboring Jak2 mutations at any stage of the hematopoietic pathway mature and accumulate to cause pruritus in PV.9 This theory has been foundational in the development of drugs with activity against cells expressing Jak2 mutations and interventions targeting histamine-releasing mast cells.9-11
The effective use of naltrexone in our patient suggests that histamine may not be the most effective or sole therapeutic target against pruritus in PV. Naltrexone targets opioid receptors in all layers of the epidermis, affecting cell adhesion and keratinocyte production, and exhibits anti-inflammatory effects through interactions with nonopioid receptors, including Toll-like receptor 4.12 The efficacy of oral naltrexone has been documented in patients with pruritus associated with immune checkpoint inhibitors, psoriasis, eczema, lichen simplex chronicus, prurigo nodularis, cholestasis, uremia, and multiple rheumatologic diseases.3,4,7-9,12-14 Opioid pathways also may be involved in peripheral and/or central processing of pruritus associated with PV.
Importantly, patients who are potential candidates for naltrexone therapy should be notified and advised of the risk of drug interactions with opioids, which could lead to symptoms of opioid withdrawal. Other common adverse effects of naltrexone include hepatotoxicity (especially in patients with a history of significant alcohol consumption), abdominal pain, nausea, arthralgias, myalgias, insomnia, headaches, fatigue, and anxiety.12 Therefore, it is integral to screen patients for opioid dependence and determine their baseline liver function. Patients should be monitored following naltrexone initiation to determine whether the drug is an appropriate and effective intervention against PV-associated pruritus.
CONCLUSIONS
This case study demonstrates that naltrexone may be a safe, effective, nonsedating, and cost-efficient oral alternative for refractory PV-associated pruritus. Future directions involve consideration of case series or randomized clinical trials investigating the efficacy of naltrexone in treating PV-associated pruritus. Further research is also warranted to better understand the pathophysiology of this symptom of PV to enhance and potentially expand medical management for patients.
Acknowledgments
The authors thank Amy Sisson (The Texas Medical Center Library) for her guidance and support in the literature review methodology.
1. Saini KS, Patnaik MM, Tefferi A. Polycythemia vera-associated pruritus and its management. Eur J Clin Invest. 2010;40(9):828-834. doi:10.1111/j.1365-2362.2010.02334.x
2. Tefferi A, Fonseca R. Selective serotonin reuptake inhibitors are effective in the treatment of polycythemia vera-associated pruritus. Blood. 2002;99(7):2627. doi:10.1182/blood.v99.7.2627
3. Lee J, Shin JU, Noh S, Park CO, Lee KH. Clinical efficacy and safety of naltrexone combination therapy in older patients with severe pruritus. Ann Dermatol. 2016;28(2):159-163. doi:10.5021/ad.2016.28.2.159
4. Phan NQ, Bernhard JD, Luger TA, Stander S. Antipruritic treatment with systemic mu-opioid receptor antagonists: a review. J Am Acad Dermatol. 2010;63(4):680-688. doi:10.1016/j.jaad.2009.08.052
5. Metze D, Reimann S, Beissert S, Luger T. Efficacy and safety of naltrexone, an oral opiate receptor antagonist, in the treatment of pruritus in internal and dermatological diseases. J Am Acad Dermatol. 1999;41(4):533-539.
6. Malekzad F, Arbabi M, Mohtasham N, et al. Efficacy of oral naltrexone on pruritus in atopic eczema: a double-blind, placebo-controlled study. J Eur Acad Dermatol Venereol. 2009;23(8):948-950. doi:10.1111/j.1468-3083.2009.03129.x
7. Terg R, Coronel E, Sorda J, Munoz AE, Findor J. Efficacy and safety of oral naltrexone treatment for pruritus of cholestasis, a crossover, double blind, placebo-controlled study. J Hepatol. 2002;37(6):717-722. doi:10.1016/s0168-8278(02)00318-5
8. Lelonek E, Matusiak L, Wrobel T, Szepietowski JC. Aquagenic pruritus in polycythemia vera: clinical characteristics. Acta Derm Venereol. 2018;98(5):496-500. doi:10.2340/00015555-2906
9. Siegel FP, Tauscher J, Petrides PE. Aquagenic pruritus in polycythemia vera: characteristics and influence on quality of life in 441 patients. Am J Hematol. 2013;88(8):665-669. doi:10.1002/ajh.23474
10. Al-Mashdali AF, Kashgary WR, Yassin MA. Ruxolitinib (a JAK2 inhibitor) as an emerging therapy for refractory pruritis in a patient with low-risk polycythemia vera: a case report. Medicine (Baltimore). 2021;100(44):e27722. doi:10.1097/MD.0000000000027722
11. Benevolo G, Vassallo F, Urbino I, Giai V. Polycythemia vera (PV): update on emerging treatment options. Ther Clin Risk Manag. 2021;17:209-221. doi:10.2147/TCRM.S213020
12. Lee B, Elston DM. The uses of naltrexone in dermatologic conditions. J Am Acad Dermatol. 2019;80(6):1746-1752. doi:10.1016/j.jaad.2018.12.031
13. de Carvalho JF, Skare T. Low-dose naltrexone in rheumatological diseases. Mediterr J Rheumatol. 2023;34(1):1-6. doi:10.31138/mjr.34.1.1
14. Singh R, Patel P, Thakker M, Sharma P, Barnes M, Montana S. Naloxone and maintenance naltrexone as novel and effective therapies for immunotherapy-induced pruritus: a case report and brief literature review. J Oncol Pract. 2019;15(6):347-348. doi:10.1200/JOP.18.00797
P ruritus is a characteristic and often debilitating clinical manifestation reported by about 50% of patients with polycythemia vera (PV). The exact pathophysiology of PV-associated pruritus is poorly understood. The itch sensation may arise from a central phenomenon without skin itch receptor involvement, as is seen in opioid-induced pruritus, or peripherally via unmyelinated C fibers. Various interventions have been used with mixed results for symptom management in this patient population.1
Selective serotonin reuptake inhibitors (SSRIs), such as paroxetine and fluoxetine, have historically demonstrated some efficacy in treating PV-associated pruritus.2 Alongside SSRIs, phlebotomy, antihistamines, phototherapy, interferon a, and myelosuppressive medications also comprise the various current treatment options. In addition to lacking efficacy, antihistamines can cause somnolence, constipation, and xerostomia.3,4 Phlebotomy and cytoreductive therapy are often effective in controlling erythrocytosis but fail to alleviate the disabling pruritus.1,5,6 More recently, suboptimal symptom alleviation has prompted the discovery of agents that target the mammalian target of rapamycin (mTOR) and Janus kinase 2 (Jak2) pathways.1
Naltrexone is an opioid antagonist shown to suppress pruritus in various dermatologic pathologies involving histamine-independent pathways.3,7,8 A systematic search strategy identified 34 studies on PV-associated pruritus, its pathophysiology and interventions, and naltrexone as a therapeutic agent. Only 1 study in the literature has described the use of naltrexone for uremic and cholestatic pruritus.9 We describe the successful use of naltrexone monotherapy for the treatment of pruritus in a patient with PV.
Case Presentation
A 40-year-old man with Jak2-positive PV treated with ruxolitinib presented to the outpatient Michael E. DeBakey Veterans Affairs Medical Center Supportive Care Clinic in Houston, Texas, for severe refractory pruritus. Wheals manifested in pruritic regions of the patient’s skin without gross excoriations or erythema. Pruritus reportedly began diffusely across the posterior torso. Through the rapid progression of an episode lasting 30 to 45 minutes, the lesions and pruritus would spread to the anterior torso, extend to the upper extremities bilaterally, and finally descend to the lower extremities bilaterally. A persistent sensation of heat or warmth on the patient’s skin was present, and periodically, this would culminate in a burning sensation comparable to “lying flat on one’s back directly on a hornet’s nest…[followed by] a million stings” that was inconsistent with erythromelalgia given the absence of erythema. The intensity of the pruritic episodes was subjectively also described as “enough to make [him] want to jump off the roof of a building…[causing] moments of deep, deep frustration…[and] the worst of all the symptoms one may encounter because of [PV].”
Pruritus was exacerbated by sweating, heat, contact with any liquids on the skin, and sunburns, which doubled the intensity. The patient reported minimal, temporary relief with cannabidiol and cold fabric or air on his skin. His current regimen and nonpharmacologic efforts provided no relief and included oatmeal baths, cornstarch after showers, and patting instead of rubbing the skin with topical products. Trials with nonprescription diphenhydramine, loratadine, and calamine and zinc were not successful. He had not pursued phototherapy due to time limitations and travel constraints. He had a history of phlebotomies and hydroxyurea use, which he preferred to avoid and discontinued 1 year before presentation.
Despite improving hematocrit (< 45% goal) and platelet counts with ruxolitinib, the patient reported worsening pruritus that significantly impaired quality of life. His sleep and social and physical activities were hindered, preventing him from working. The patient’s active medications also included low-dose aspirin, sertraline, hydroxyzine, triamcinolone acetonide, and pregabalin for sciatica. Given persistent symptoms despite multimodal therapy and lifestyle modifications, the patient was started on naltrexone 25 mg daily, which provided immediate relief of symptoms. He continues to have adequate symptom control 2 years after naltrexone initiation.
Literature Review
A systematic search strategy was developed with the assistance of a medical librarian in Medline Ovid, using both Medical Subject Heading (MeSH) terms and synonymous keywords. The strategy was then translated to Embase, Web of Science, and Cochrane to extract publications investigating PV, pruritus, and/or naltrexone therapy. All searches were conducted on July 18, 2022, and the results of the literature review were as follows: 2 results from Medline Ovid; 34 results from Embase (2 were duplicates of Medline Ovid results); 3 results from Web of Science (all of which were duplicates of Medline Ovid or Embase results); and 0 results from Cochrane (Figure).
Discussion
Although pruritus is a common and often excruciating manifestation of PV, its pathophysiology remains unclear. Some patients with decreasing or newly normal hematocrit and hemoglobin levels have paradoxically experienced an intensification of their pruritus, which introduces erythropoietin signaling pathways as a potential mechanism of the symptom.8 However, iron replacement therapy for patients with exacerbated pruritus after phlebotomies has not demonstrated consistent relief of pruritus.8 Normalization of platelet levels also has not been historically associated with improvement of pruritus.8,9 It has been hypothesized that cells harboring Jak2 mutations at any stage of the hematopoietic pathway mature and accumulate to cause pruritus in PV.9 This theory has been foundational in the development of drugs with activity against cells expressing Jak2 mutations and interventions targeting histamine-releasing mast cells.9-11
The effective use of naltrexone in our patient suggests that histamine may not be the most effective or sole therapeutic target against pruritus in PV. Naltrexone targets opioid receptors in all layers of the epidermis, affecting cell adhesion and keratinocyte production, and exhibits anti-inflammatory effects through interactions with nonopioid receptors, including Toll-like receptor 4.12 The efficacy of oral naltrexone has been documented in patients with pruritus associated with immune checkpoint inhibitors, psoriasis, eczema, lichen simplex chronicus, prurigo nodularis, cholestasis, uremia, and multiple rheumatologic diseases.3,4,7-9,12-14 Opioid pathways also may be involved in peripheral and/or central processing of pruritus associated with PV.
Importantly, patients who are potential candidates for naltrexone therapy should be notified and advised of the risk of drug interactions with opioids, which could lead to symptoms of opioid withdrawal. Other common adverse effects of naltrexone include hepatotoxicity (especially in patients with a history of significant alcohol consumption), abdominal pain, nausea, arthralgias, myalgias, insomnia, headaches, fatigue, and anxiety.12 Therefore, it is integral to screen patients for opioid dependence and determine their baseline liver function. Patients should be monitored following naltrexone initiation to determine whether the drug is an appropriate and effective intervention against PV-associated pruritus.
CONCLUSIONS
This case study demonstrates that naltrexone may be a safe, effective, nonsedating, and cost-efficient oral alternative for refractory PV-associated pruritus. Future directions involve consideration of case series or randomized clinical trials investigating the efficacy of naltrexone in treating PV-associated pruritus. Further research is also warranted to better understand the pathophysiology of this symptom of PV to enhance and potentially expand medical management for patients.
Acknowledgments
The authors thank Amy Sisson (The Texas Medical Center Library) for her guidance and support in the literature review methodology.
P ruritus is a characteristic and often debilitating clinical manifestation reported by about 50% of patients with polycythemia vera (PV). The exact pathophysiology of PV-associated pruritus is poorly understood. The itch sensation may arise from a central phenomenon without skin itch receptor involvement, as is seen in opioid-induced pruritus, or peripherally via unmyelinated C fibers. Various interventions have been used with mixed results for symptom management in this patient population.1
Selective serotonin reuptake inhibitors (SSRIs), such as paroxetine and fluoxetine, have historically demonstrated some efficacy in treating PV-associated pruritus.2 Alongside SSRIs, phlebotomy, antihistamines, phototherapy, interferon a, and myelosuppressive medications also comprise the various current treatment options. In addition to lacking efficacy, antihistamines can cause somnolence, constipation, and xerostomia.3,4 Phlebotomy and cytoreductive therapy are often effective in controlling erythrocytosis but fail to alleviate the disabling pruritus.1,5,6 More recently, suboptimal symptom alleviation has prompted the discovery of agents that target the mammalian target of rapamycin (mTOR) and Janus kinase 2 (Jak2) pathways.1
Naltrexone is an opioid antagonist shown to suppress pruritus in various dermatologic pathologies involving histamine-independent pathways.3,7,8 A systematic search strategy identified 34 studies on PV-associated pruritus, its pathophysiology and interventions, and naltrexone as a therapeutic agent. Only 1 study in the literature has described the use of naltrexone for uremic and cholestatic pruritus.9 We describe the successful use of naltrexone monotherapy for the treatment of pruritus in a patient with PV.
Case Presentation
A 40-year-old man with Jak2-positive PV treated with ruxolitinib presented to the outpatient Michael E. DeBakey Veterans Affairs Medical Center Supportive Care Clinic in Houston, Texas, for severe refractory pruritus. Wheals manifested in pruritic regions of the patient’s skin without gross excoriations or erythema. Pruritus reportedly began diffusely across the posterior torso. Through the rapid progression of an episode lasting 30 to 45 minutes, the lesions and pruritus would spread to the anterior torso, extend to the upper extremities bilaterally, and finally descend to the lower extremities bilaterally. A persistent sensation of heat or warmth on the patient’s skin was present, and periodically, this would culminate in a burning sensation comparable to “lying flat on one’s back directly on a hornet’s nest…[followed by] a million stings” that was inconsistent with erythromelalgia given the absence of erythema. The intensity of the pruritic episodes was subjectively also described as “enough to make [him] want to jump off the roof of a building…[causing] moments of deep, deep frustration…[and] the worst of all the symptoms one may encounter because of [PV].”
Pruritus was exacerbated by sweating, heat, contact with any liquids on the skin, and sunburns, which doubled the intensity. The patient reported minimal, temporary relief with cannabidiol and cold fabric or air on his skin. His current regimen and nonpharmacologic efforts provided no relief and included oatmeal baths, cornstarch after showers, and patting instead of rubbing the skin with topical products. Trials with nonprescription diphenhydramine, loratadine, and calamine and zinc were not successful. He had not pursued phototherapy due to time limitations and travel constraints. He had a history of phlebotomies and hydroxyurea use, which he preferred to avoid and discontinued 1 year before presentation.
Despite improving hematocrit (< 45% goal) and platelet counts with ruxolitinib, the patient reported worsening pruritus that significantly impaired quality of life. His sleep and social and physical activities were hindered, preventing him from working. The patient’s active medications also included low-dose aspirin, sertraline, hydroxyzine, triamcinolone acetonide, and pregabalin for sciatica. Given persistent symptoms despite multimodal therapy and lifestyle modifications, the patient was started on naltrexone 25 mg daily, which provided immediate relief of symptoms. He continues to have adequate symptom control 2 years after naltrexone initiation.
Literature Review
A systematic search strategy was developed with the assistance of a medical librarian in Medline Ovid, using both Medical Subject Heading (MeSH) terms and synonymous keywords. The strategy was then translated to Embase, Web of Science, and Cochrane to extract publications investigating PV, pruritus, and/or naltrexone therapy. All searches were conducted on July 18, 2022, and the results of the literature review were as follows: 2 results from Medline Ovid; 34 results from Embase (2 were duplicates of Medline Ovid results); 3 results from Web of Science (all of which were duplicates of Medline Ovid or Embase results); and 0 results from Cochrane (Figure).
Discussion
Although pruritus is a common and often excruciating manifestation of PV, its pathophysiology remains unclear. Some patients with decreasing or newly normal hematocrit and hemoglobin levels have paradoxically experienced an intensification of their pruritus, which introduces erythropoietin signaling pathways as a potential mechanism of the symptom.8 However, iron replacement therapy for patients with exacerbated pruritus after phlebotomies has not demonstrated consistent relief of pruritus.8 Normalization of platelet levels also has not been historically associated with improvement of pruritus.8,9 It has been hypothesized that cells harboring Jak2 mutations at any stage of the hematopoietic pathway mature and accumulate to cause pruritus in PV.9 This theory has been foundational in the development of drugs with activity against cells expressing Jak2 mutations and interventions targeting histamine-releasing mast cells.9-11
The effective use of naltrexone in our patient suggests that histamine may not be the most effective or sole therapeutic target against pruritus in PV. Naltrexone targets opioid receptors in all layers of the epidermis, affecting cell adhesion and keratinocyte production, and exhibits anti-inflammatory effects through interactions with nonopioid receptors, including Toll-like receptor 4.12 The efficacy of oral naltrexone has been documented in patients with pruritus associated with immune checkpoint inhibitors, psoriasis, eczema, lichen simplex chronicus, prurigo nodularis, cholestasis, uremia, and multiple rheumatologic diseases.3,4,7-9,12-14 Opioid pathways also may be involved in peripheral and/or central processing of pruritus associated with PV.
Importantly, patients who are potential candidates for naltrexone therapy should be notified and advised of the risk of drug interactions with opioids, which could lead to symptoms of opioid withdrawal. Other common adverse effects of naltrexone include hepatotoxicity (especially in patients with a history of significant alcohol consumption), abdominal pain, nausea, arthralgias, myalgias, insomnia, headaches, fatigue, and anxiety.12 Therefore, it is integral to screen patients for opioid dependence and determine their baseline liver function. Patients should be monitored following naltrexone initiation to determine whether the drug is an appropriate and effective intervention against PV-associated pruritus.
CONCLUSIONS
This case study demonstrates that naltrexone may be a safe, effective, nonsedating, and cost-efficient oral alternative for refractory PV-associated pruritus. Future directions involve consideration of case series or randomized clinical trials investigating the efficacy of naltrexone in treating PV-associated pruritus. Further research is also warranted to better understand the pathophysiology of this symptom of PV to enhance and potentially expand medical management for patients.
Acknowledgments
The authors thank Amy Sisson (The Texas Medical Center Library) for her guidance and support in the literature review methodology.
1. Saini KS, Patnaik MM, Tefferi A. Polycythemia vera-associated pruritus and its management. Eur J Clin Invest. 2010;40(9):828-834. doi:10.1111/j.1365-2362.2010.02334.x
2. Tefferi A, Fonseca R. Selective serotonin reuptake inhibitors are effective in the treatment of polycythemia vera-associated pruritus. Blood. 2002;99(7):2627. doi:10.1182/blood.v99.7.2627
3. Lee J, Shin JU, Noh S, Park CO, Lee KH. Clinical efficacy and safety of naltrexone combination therapy in older patients with severe pruritus. Ann Dermatol. 2016;28(2):159-163. doi:10.5021/ad.2016.28.2.159
4. Phan NQ, Bernhard JD, Luger TA, Stander S. Antipruritic treatment with systemic mu-opioid receptor antagonists: a review. J Am Acad Dermatol. 2010;63(4):680-688. doi:10.1016/j.jaad.2009.08.052
5. Metze D, Reimann S, Beissert S, Luger T. Efficacy and safety of naltrexone, an oral opiate receptor antagonist, in the treatment of pruritus in internal and dermatological diseases. J Am Acad Dermatol. 1999;41(4):533-539.
6. Malekzad F, Arbabi M, Mohtasham N, et al. Efficacy of oral naltrexone on pruritus in atopic eczema: a double-blind, placebo-controlled study. J Eur Acad Dermatol Venereol. 2009;23(8):948-950. doi:10.1111/j.1468-3083.2009.03129.x
7. Terg R, Coronel E, Sorda J, Munoz AE, Findor J. Efficacy and safety of oral naltrexone treatment for pruritus of cholestasis, a crossover, double blind, placebo-controlled study. J Hepatol. 2002;37(6):717-722. doi:10.1016/s0168-8278(02)00318-5
8. Lelonek E, Matusiak L, Wrobel T, Szepietowski JC. Aquagenic pruritus in polycythemia vera: clinical characteristics. Acta Derm Venereol. 2018;98(5):496-500. doi:10.2340/00015555-2906
9. Siegel FP, Tauscher J, Petrides PE. Aquagenic pruritus in polycythemia vera: characteristics and influence on quality of life in 441 patients. Am J Hematol. 2013;88(8):665-669. doi:10.1002/ajh.23474
10. Al-Mashdali AF, Kashgary WR, Yassin MA. Ruxolitinib (a JAK2 inhibitor) as an emerging therapy for refractory pruritis in a patient with low-risk polycythemia vera: a case report. Medicine (Baltimore). 2021;100(44):e27722. doi:10.1097/MD.0000000000027722
11. Benevolo G, Vassallo F, Urbino I, Giai V. Polycythemia vera (PV): update on emerging treatment options. Ther Clin Risk Manag. 2021;17:209-221. doi:10.2147/TCRM.S213020
12. Lee B, Elston DM. The uses of naltrexone in dermatologic conditions. J Am Acad Dermatol. 2019;80(6):1746-1752. doi:10.1016/j.jaad.2018.12.031
13. de Carvalho JF, Skare T. Low-dose naltrexone in rheumatological diseases. Mediterr J Rheumatol. 2023;34(1):1-6. doi:10.31138/mjr.34.1.1
14. Singh R, Patel P, Thakker M, Sharma P, Barnes M, Montana S. Naloxone and maintenance naltrexone as novel and effective therapies for immunotherapy-induced pruritus: a case report and brief literature review. J Oncol Pract. 2019;15(6):347-348. doi:10.1200/JOP.18.00797
1. Saini KS, Patnaik MM, Tefferi A. Polycythemia vera-associated pruritus and its management. Eur J Clin Invest. 2010;40(9):828-834. doi:10.1111/j.1365-2362.2010.02334.x
2. Tefferi A, Fonseca R. Selective serotonin reuptake inhibitors are effective in the treatment of polycythemia vera-associated pruritus. Blood. 2002;99(7):2627. doi:10.1182/blood.v99.7.2627
3. Lee J, Shin JU, Noh S, Park CO, Lee KH. Clinical efficacy and safety of naltrexone combination therapy in older patients with severe pruritus. Ann Dermatol. 2016;28(2):159-163. doi:10.5021/ad.2016.28.2.159
4. Phan NQ, Bernhard JD, Luger TA, Stander S. Antipruritic treatment with systemic mu-opioid receptor antagonists: a review. J Am Acad Dermatol. 2010;63(4):680-688. doi:10.1016/j.jaad.2009.08.052
5. Metze D, Reimann S, Beissert S, Luger T. Efficacy and safety of naltrexone, an oral opiate receptor antagonist, in the treatment of pruritus in internal and dermatological diseases. J Am Acad Dermatol. 1999;41(4):533-539.
6. Malekzad F, Arbabi M, Mohtasham N, et al. Efficacy of oral naltrexone on pruritus in atopic eczema: a double-blind, placebo-controlled study. J Eur Acad Dermatol Venereol. 2009;23(8):948-950. doi:10.1111/j.1468-3083.2009.03129.x
7. Terg R, Coronel E, Sorda J, Munoz AE, Findor J. Efficacy and safety of oral naltrexone treatment for pruritus of cholestasis, a crossover, double blind, placebo-controlled study. J Hepatol. 2002;37(6):717-722. doi:10.1016/s0168-8278(02)00318-5
8. Lelonek E, Matusiak L, Wrobel T, Szepietowski JC. Aquagenic pruritus in polycythemia vera: clinical characteristics. Acta Derm Venereol. 2018;98(5):496-500. doi:10.2340/00015555-2906
9. Siegel FP, Tauscher J, Petrides PE. Aquagenic pruritus in polycythemia vera: characteristics and influence on quality of life in 441 patients. Am J Hematol. 2013;88(8):665-669. doi:10.1002/ajh.23474
10. Al-Mashdali AF, Kashgary WR, Yassin MA. Ruxolitinib (a JAK2 inhibitor) as an emerging therapy for refractory pruritis in a patient with low-risk polycythemia vera: a case report. Medicine (Baltimore). 2021;100(44):e27722. doi:10.1097/MD.0000000000027722
11. Benevolo G, Vassallo F, Urbino I, Giai V. Polycythemia vera (PV): update on emerging treatment options. Ther Clin Risk Manag. 2021;17:209-221. doi:10.2147/TCRM.S213020
12. Lee B, Elston DM. The uses of naltrexone in dermatologic conditions. J Am Acad Dermatol. 2019;80(6):1746-1752. doi:10.1016/j.jaad.2018.12.031
13. de Carvalho JF, Skare T. Low-dose naltrexone in rheumatological diseases. Mediterr J Rheumatol. 2023;34(1):1-6. doi:10.31138/mjr.34.1.1
14. Singh R, Patel P, Thakker M, Sharma P, Barnes M, Montana S. Naloxone and maintenance naltrexone as novel and effective therapies for immunotherapy-induced pruritus: a case report and brief literature review. J Oncol Pract. 2019;15(6):347-348. doi:10.1200/JOP.18.00797
A Case Series of Rare Immune-Mediated Adverse Reactions at the New Mexico Veterans Affairs Medical Center
Immune checkpoint inhibitors (ICIs), often broadly referred to as immunotherapy, are being prescribed at increasing rates due to their effectiveness in treating a growing number of advanced solid tumors and hematologic malignancies.1 It has been well established that T-cell signaling mechanisms designed to combat foreign pathogens have been involved in the mitigation of tumor proliferation.2 This protective process can be supported or restricted by infection, medication, or mutations.
ICIs support T-cell–mediated destruction of tumor cells by inhibiting the mechanisms designed to limit autoimmunity, specifically the programmed cell death protein 1/programmed cell death ligand 1 (PD-1/PD-L1) and cytotoxic T-lymphocyte–associated antigen 4 (CTLA-4) pathways. The results have been impressive, leading to an expansive number of US Food and Drug Administration (FDA) approvals across a diverse set of malignancies. Consequently, the Nobel Prize in Physiology or Medicine was awarded for such work in 2018.3
BACKGROUND
While altering these pathways has been shown to hinder tumor growth, the lesser restrictions on the immune system can drive unwanted autoimmune inflammation to host tissue. These toxicities are collectively known as immune-mediated adverse reactions (IMARs). Clinically and histologically, IMARs frequently manifest similarly to other autoimmune conditions and may affect any organ, including skin, liver, lungs, heart, intestine (small and large), kidneys, eyes, endocrine glands, and neurologic tissue.4,5 According to recent studies, as many as 20% to 30% of patients receiving a single ICI will experience at least 1 clinically significant IMAR, and about 13% are classified as severe; however, < 10% of patients will have their ICIs discontinued due to these reactions.6
Though infrequent, a thorough understanding of the severity of IMARs to ICIs is critical for the diagnosis and management of these organ-threatening and potentially life-threatening toxicities. With the growing use of these agents and more FDA approvals for dual checkpoint blockage (concurrent use of CTLA-4 and PD-1/PD-L1 inhibitors), the absolute number of IMARs is expected to rise, thereby leading to more exposure of such events to both oncology and nononcology clinicians. Prior literature has clearly described the treatments and outcomes for many common severe toxicities; however, information regarding presentations and outcomes for rare IMARs is lacking.7
A few fascinating cases of rare toxicities have been observed at the New Mexico Veterans Affairs Medical Center (NMVAMC) in Albuquerque despite its relatively small size compared with other US Department of Veterans Affairs medical centers. As such, herein, the diagnostic evaluation, treatments, and outcomes of rare IMARs are reported for each case, and the related literature is reviewed.
Patient Selection
Patients who were required to discontinue or postpone treatment with any ICI blocking the CTLA-4 (ipilimumab), PD-1 (pembrolizumab, nivolumab, cemiplimab), or PD-L1 (atezolizumab, avelumab, durvalumab) pathways between 2015 to 2021 due to toxicity at the NMVAMC were eligible for inclusion. The electronic health record was reviewed for each eligible case, and the patient demographics, disease characteristics, toxicities, and outcomes were documented for each patient. For the 57 patients who received ICIs within the chosen period, 11 required a treatment break or discontinuation. Of these, 3 cases were selected for reporting due to the rare IMARs observed. This study was approved by the NMVAMC Institutional Review Board.
Case 1: Myocarditis
An 84-year-old man receiving a chemoimmunotherapy regimen consisting of carboplatin, pemetrexed, and pembrolizumab for recurrent, stage IV lung adenocarcinoma developed grade 4 cardiomyopathy, as defined by the Common Terminology Criteria for Adverse Events (CTCAE) v5.0, during his treatment.8 He was treated for 2 cycles before he began experiencing an increase in liver enzymes.
The patient’s presentation was concerning for myocarditis, and he was quickly admitted to NMVAMC. Cardiac catheterization did not reveal any signs of coronary occlusive disease. Prednisone 1 mg/kg was administered immediately; however, given continued chest pain and volume overload, he was quickly transitioned to solumedrol 1000 mg IV daily. After the initiation of his treatment, the patient’s transaminitis began to resolve, and troponin levels began to decrease; however, his symptoms continued to worsen, and his troponin rose again. By the fourth day of hospitalization, the patient was treated with infliximab, a tumor necrosis factor-α inhibitor shown to reverse ICI-induced autoimmune inflammation, with only mild improvement of his symptoms. The patient’s condition continued to deteriorate, his troponin levels remained elevated, and his family decided to withhold additional treatment. The patient died shortly thereafter.
Discussion
Cardiotoxicity resulting from ICI therapy is far less common than the other potential severe toxicities associated with ICIs. Nevertheless, many cases of ICI-induced cardiac inflammation have been reported, and it has been widely established that patients treated with ICIs are generally at higher risk for acute coronary syndrome.9-11 Acute cardiotoxicity secondary to autoimmune destruction of cardiac tissue includes myocarditis, pericarditis, and vasculitis, which may manifest with symptoms of heart failure and/or arrhythmia. Grading of ICI-induced cardiomyopathy has been defined by both CTCAE and the American Society of Clinical Oncology (ASCO), with grade 4 representing moderate to severe clinical decompensation requiring IV medications in the setting of life-threatening conditions.
Review articles have described the treatment options for severe cases.7,12 As detailed in prior reports, once ICI-induced cardiomyopathy is suspected, urgent admission and immediate evaluation to rule out acute coronary syndrome should be undertaken. Given the potential for deterioration despite the occasional insidious onset, aggressive cardiac monitoring, and close follow-up to measure response to interventions should be undertaken.
Case 2: Uveitis
A 70-year-old man who received pembrolizumab as a bladder-sparing approach for his superficial bladder cancer refractory to intravesical treatments developed uveitis. Approximately 3 months following the initiation of treatment, the patient reported bilateral itchy eyes, erythema, and tearing. He had a known history of allergic conjunctivitis that predated the ICI therapy, and consequently, it was unclear whether his symptoms were reflective of a more concerning issue. The patient’s symptoms continued to wax and wane for a few months, prompting a referral to ophthalmology colleagues at NMVAMC.
Ophthalmology evaluation identified uveitic glaucoma in the setting of his underlying chronic glaucoma. Pembrolizumab was discontinued, and the patient was counseled on choosing either cystectomy or locoregional therapies if further tumors arose. However, within a few weeks of administering topical steroid drops, his symptoms markedly improved, and he wished to be restarted on pembrolizumab. His uveitis remained in remission, and he has been treated with pembrolizumab for more than 1 year since this episode. He has had no clear findings of superficial bladder cancer recurrence while receiving ICI therapy.
Discussion
Uveitis is a known complication of pembrolizumab, and it has been shown to occur in 1% of patients with this treatment.13,14 It should be noted that most of the studies of this IMAR occurred in patients with metastatic melanoma; therefore the rate of this condition in other patients is less understood. Overall, ocular IMARs secondary to anti-PD-1 and anti-PD-L1 therapies are rare.
The most common IMAR is surface ocular disease, consisting of dry eye disease (DED), conjunctivitis, uveitis, and keratitis. Of these, the most common ocular surface disease is DED, which occurred in 1% to 4% of patients treated with ICI therapy; most of these reactions are mild and self-limiting.15 Atezolizumab has the highest association with ocular inflammation and ipilimumab has the highest association with uveitis, with reported odds ratios of 18.89 and 10.54, respectively.16 Treatment of ICI-induced uveitis generally includes topical steroids and treatment discontinuation or break.17 Oral or IV steroids, infliximab, and procedural involvement may be considered in refractory cases or those initially presenting with marked vision loss. Close communication with ophthalmology colleagues to monitor visual acuity and ocular pressure multiple times weekly during the acute phase is required for treatment titration.
Case 3: Organizing Pneumonia
A man aged 63 years was diagnosed with malignant mesothelioma after incidentally noting a pleural effusion and thickening on routine low-dose computed tomography surveillance of pulmonary nodules. A biopsy was performed and was consistent with mesothelioma, and the patient was started on nivolumab (PD-1 inhibitor) and ipilimumab (CTLA-4 inhibitor). The patient was initiated on dual ICIs, and after 6 months of therapy, he had a promising complete response. However, after 9 months of therapy, he developed a new left upper lobe (LUL) pleural-based lesion (Figure 2A).
A biopsy was performed, and the histopathologic appearance was consistent with organizing pneumonia (OP) (Figure 3).
Discussion
ICIs can uncommonly drive pneumonitis, with the frequency adjusted based on the number of ICIs prescribed and the primary cancer involved. Across all cancers, up to 5% of patients treated with single-agent ICI therapy may experience pneumonitis, though often the findings may simply be radiographic without symptoms. Moreover, up to 10% of patients undergoing treatment for pulmonary cancer or those with dual ICI treatment regimens experience radiographic and/or clinical pneumonitis.18 The clinical manifestations include a broad spectrum of respiratory symptoms. Given the convoluting concerns of cancer progression and infection, a biopsy is often obtained. Histopathologic findings of pneumonitis may include diffuse alveolar damage and/or interstitial lung disease, with OP being a rare variant of ILD.
Among pulmonologists, OP is felt to have polymorphous imaging findings, and biopsy is required to confirm histology; however, histopathology cannot define etiology, and consequently, OP is somewhat of an umbrella diagnosis. The condition can be cryptogenic (idiopathic) or secondary to a multitude of conditions (infection, drug toxicity, or systemic disease). It is classically described as polypoid aggregations of fibroblasts that obstruct the alveolar spaces.19 This histopathologic pattern was demonstrated in our patient’s lung biopsy. Given a prior case description of ICIs, mesothelioma, OP development, and the unremarkable infectious workup, we felt that the patient’s OP was driven by his dual ICI therapy, thereby leading to the ultimate discontinuation of his ICIs and initiation of steroids.20 Thankfully, the patient had already obtained a complete response to his ICIs, and hopefully, he can attain a durable remission with the addition of maintenance chemotherapy.
CONCLUSIONS
ICIs have revolutionized the treatment of a myriad of solid tumors and hematologic malignancies, and their use internationally is expected to increase. With the alteration in immunology pathways, clinicians in all fields will need to be familiarized with IMARs secondary to these agents, including rare subtypes. In addition, the variability in presentations relative to the patients’ treatment course was significant (between 2-9 months), and this highlights that these IMARs can occur at any time point and clinicians should be ever vigilant to spot symptoms in their patients.
It was unexpected for the 3 aforementioned rare toxicities to arise at NMVAMC among only 57 treated patients, and we speculate that these findings may have been observed for 1 of 3 reasons. First, caring for 3 patients with this collection of rare toxicities may have been due to chance. Second, though there is sparse literature studying the topic, the regional environment, including sunlight exposure and air quality, may play a role in the development of one or all of these rare toxicities. Third, rates of these toxicities may be underreported in the literature or attributed to other conditions rather than due to ICIs at other sites, and the uncommon nature of these IMARs may be overstated. Investigations evaluating rates of toxicities, including those traditionally uncommonly seen, based on regional location should be conducted before any further conclusions are drawn.
1. Bagchi S, Yuan R, Engleman EG. Immune checkpoint inhibitors for the treatment of cancer: clinical impact and mechanisms of response and resistance. Published online 2020. doi:10.1146/annurev-pathol-042020
2. Chen DS, Mellman I. Oncology meets immunology: The cancer-immunity cycle. Immunity. 2013;39(1):1-10. doi:10.1016/j.immuni.2013.07.012
3. Smyth MJ, Teng MWL. 2018 Nobel Prize in physiology or medicine. Clin Transl Immunology. 2018;7(10). doi:10.1002/cti2.1041
4. Baxi S, Yang A, Gennarelli RL, et al. Immune-related adverse events for anti-PD-1 and anti-PD-L1 drugs: Systematic review and meta-analysis. BMJ (Online). 2018;360. doi:10.1136/bmj.k793
5. Ellithi M, Elnair R, Chang GV, Abdallah MA. Toxicities of immune checkpoint inhibitors: itis-ending adverse reactions and more. Cureus. Published online February 10, 2020. doi:10.7759/cureus.6935
6. Berti A, Bortolotti R, Dipasquale M, et al. Meta-analysis of immune-related adverse events in phase 3 clinical trials assessing immune checkpoint inhibitors for lung cancer. Crit Rev Oncol Hematol. 2021;162. doi:10.1016/j.critrevonc.2021.103351
7. Davies M, Duffield EA. Safety of checkpoint inhibitors for cancer treatment: strategies for patient monitoring and management of immune-mediated adverse events. Immunotargets Ther. 2017;Volume 6:51-71. doi:10.2147/itt.s141577
8. US Department of Health and Human Services. Common Terminology Criteria for Adverse Events V5.0. Accessed July 17, 2023. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5584920/
9. Johnson DB, Balko JM, Compton ML, et al. Fulminant myocarditis with combination immune checkpoint blockade. N Engl J Med. 2016;375(18):1749-1755. doi:10.1056/nejmoa1609214
10. Mahmood SS, Fradley MG, Cohen J V., et al. Myocarditis in patients treated with immune checkpoint inhibitors. J Am Coll Cardiol. 2018;71(16):1755-1764. doi:10.1016/j.jacc.2018.02.037
11. Wang DY, Salem JE, Cohen JV, et al. Fatal toxic effects associated with immune checkpoint inhibitors: a systematic review and meta-analysis. JAMA Oncol. 2018;4(12):1721-1728. doi:10.1001/jamaoncol.2018.3923
12. Brahmer JR, Lacchetti C, Schneider BJ, et al; National Comprehensive Cancer Network. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Onc. 2018;36(17):1714-1768. doi:10.1200/JCO
13. Ribas A, Hamid O, Daud A, et al. Association of pembrolizumab with tumor response and survival among patients with advanced melanoma. JAMA. 2016;315:1600-1609. doi:10.1001/jama.2016.4059
14. Dalvin LA, Shields CL, Orloff M, Sato T, Shields JA. Checkpoint inhibitor immune therapy: systemic indications and ophthalmic side effects. Retina. 2018;38(6):1063-1078. doi:10.1097/IAE.0000000000002181
15. Park RB, Jain S, Han H, Park J. Ocular surface disease associated with immune checkpoint inhibitor therapy. Ocular Surface. 2021;20:115-129. doi:10.1016/j.jtos.2021.02.004
16. Fang T, Maberley DA, Etminan M. Ocular adverse events with immune checkpoint inhibitors. J Curr Ophthalmol. 2019;31(3):319-322. doi:10.1016/j.joco.2019.05.002
17. Whist E, Symes RJ, Chang JH, et al. Uveitis caused by treatment for malignant melanoma: a case series. Retin Cases Brief Rep. 2021;15(6):718-723. doi:10.1097/ICB.0000000000000876
18. Naidoo J, Wang X, Woo KM, et al. Pneumonitis in patients treated with anti-programmed death-1/programmed death ligand 1 therapy. J Clin Onc. 2017;35(7):709-717. doi:10.1200/JCO.2016.68.2005
19. Yoshikawa A, Bychkov A, Sathirareuangchai S. Other nonneoplastic conditions, acute lung injury, organizing pneumonia. Accessed July 17, 2023. https://www.pathologyoutlines.com/topic/lungnontumorboop.html
20. Kuint R, Lotem M, Neuman T, et al. Organizing pneumonia following treatment with pembrolizumab for metastatic malignant melanoma–a case report. Respir Med Case Rep. 2017;20:95-97. doi:10.1016/j.rmcr.2017.01.003
Immune checkpoint inhibitors (ICIs), often broadly referred to as immunotherapy, are being prescribed at increasing rates due to their effectiveness in treating a growing number of advanced solid tumors and hematologic malignancies.1 It has been well established that T-cell signaling mechanisms designed to combat foreign pathogens have been involved in the mitigation of tumor proliferation.2 This protective process can be supported or restricted by infection, medication, or mutations.
ICIs support T-cell–mediated destruction of tumor cells by inhibiting the mechanisms designed to limit autoimmunity, specifically the programmed cell death protein 1/programmed cell death ligand 1 (PD-1/PD-L1) and cytotoxic T-lymphocyte–associated antigen 4 (CTLA-4) pathways. The results have been impressive, leading to an expansive number of US Food and Drug Administration (FDA) approvals across a diverse set of malignancies. Consequently, the Nobel Prize in Physiology or Medicine was awarded for such work in 2018.3
BACKGROUND
While altering these pathways has been shown to hinder tumor growth, the lesser restrictions on the immune system can drive unwanted autoimmune inflammation to host tissue. These toxicities are collectively known as immune-mediated adverse reactions (IMARs). Clinically and histologically, IMARs frequently manifest similarly to other autoimmune conditions and may affect any organ, including skin, liver, lungs, heart, intestine (small and large), kidneys, eyes, endocrine glands, and neurologic tissue.4,5 According to recent studies, as many as 20% to 30% of patients receiving a single ICI will experience at least 1 clinically significant IMAR, and about 13% are classified as severe; however, < 10% of patients will have their ICIs discontinued due to these reactions.6
Though infrequent, a thorough understanding of the severity of IMARs to ICIs is critical for the diagnosis and management of these organ-threatening and potentially life-threatening toxicities. With the growing use of these agents and more FDA approvals for dual checkpoint blockage (concurrent use of CTLA-4 and PD-1/PD-L1 inhibitors), the absolute number of IMARs is expected to rise, thereby leading to more exposure of such events to both oncology and nononcology clinicians. Prior literature has clearly described the treatments and outcomes for many common severe toxicities; however, information regarding presentations and outcomes for rare IMARs is lacking.7
A few fascinating cases of rare toxicities have been observed at the New Mexico Veterans Affairs Medical Center (NMVAMC) in Albuquerque despite its relatively small size compared with other US Department of Veterans Affairs medical centers. As such, herein, the diagnostic evaluation, treatments, and outcomes of rare IMARs are reported for each case, and the related literature is reviewed.
Patient Selection
Patients who were required to discontinue or postpone treatment with any ICI blocking the CTLA-4 (ipilimumab), PD-1 (pembrolizumab, nivolumab, cemiplimab), or PD-L1 (atezolizumab, avelumab, durvalumab) pathways between 2015 to 2021 due to toxicity at the NMVAMC were eligible for inclusion. The electronic health record was reviewed for each eligible case, and the patient demographics, disease characteristics, toxicities, and outcomes were documented for each patient. For the 57 patients who received ICIs within the chosen period, 11 required a treatment break or discontinuation. Of these, 3 cases were selected for reporting due to the rare IMARs observed. This study was approved by the NMVAMC Institutional Review Board.
Case 1: Myocarditis
An 84-year-old man receiving a chemoimmunotherapy regimen consisting of carboplatin, pemetrexed, and pembrolizumab for recurrent, stage IV lung adenocarcinoma developed grade 4 cardiomyopathy, as defined by the Common Terminology Criteria for Adverse Events (CTCAE) v5.0, during his treatment.8 He was treated for 2 cycles before he began experiencing an increase in liver enzymes.
The patient’s presentation was concerning for myocarditis, and he was quickly admitted to NMVAMC. Cardiac catheterization did not reveal any signs of coronary occlusive disease. Prednisone 1 mg/kg was administered immediately; however, given continued chest pain and volume overload, he was quickly transitioned to solumedrol 1000 mg IV daily. After the initiation of his treatment, the patient’s transaminitis began to resolve, and troponin levels began to decrease; however, his symptoms continued to worsen, and his troponin rose again. By the fourth day of hospitalization, the patient was treated with infliximab, a tumor necrosis factor-α inhibitor shown to reverse ICI-induced autoimmune inflammation, with only mild improvement of his symptoms. The patient’s condition continued to deteriorate, his troponin levels remained elevated, and his family decided to withhold additional treatment. The patient died shortly thereafter.
Discussion
Cardiotoxicity resulting from ICI therapy is far less common than the other potential severe toxicities associated with ICIs. Nevertheless, many cases of ICI-induced cardiac inflammation have been reported, and it has been widely established that patients treated with ICIs are generally at higher risk for acute coronary syndrome.9-11 Acute cardiotoxicity secondary to autoimmune destruction of cardiac tissue includes myocarditis, pericarditis, and vasculitis, which may manifest with symptoms of heart failure and/or arrhythmia. Grading of ICI-induced cardiomyopathy has been defined by both CTCAE and the American Society of Clinical Oncology (ASCO), with grade 4 representing moderate to severe clinical decompensation requiring IV medications in the setting of life-threatening conditions.
Review articles have described the treatment options for severe cases.7,12 As detailed in prior reports, once ICI-induced cardiomyopathy is suspected, urgent admission and immediate evaluation to rule out acute coronary syndrome should be undertaken. Given the potential for deterioration despite the occasional insidious onset, aggressive cardiac monitoring, and close follow-up to measure response to interventions should be undertaken.
Case 2: Uveitis
A 70-year-old man who received pembrolizumab as a bladder-sparing approach for his superficial bladder cancer refractory to intravesical treatments developed uveitis. Approximately 3 months following the initiation of treatment, the patient reported bilateral itchy eyes, erythema, and tearing. He had a known history of allergic conjunctivitis that predated the ICI therapy, and consequently, it was unclear whether his symptoms were reflective of a more concerning issue. The patient’s symptoms continued to wax and wane for a few months, prompting a referral to ophthalmology colleagues at NMVAMC.
Ophthalmology evaluation identified uveitic glaucoma in the setting of his underlying chronic glaucoma. Pembrolizumab was discontinued, and the patient was counseled on choosing either cystectomy or locoregional therapies if further tumors arose. However, within a few weeks of administering topical steroid drops, his symptoms markedly improved, and he wished to be restarted on pembrolizumab. His uveitis remained in remission, and he has been treated with pembrolizumab for more than 1 year since this episode. He has had no clear findings of superficial bladder cancer recurrence while receiving ICI therapy.
Discussion
Uveitis is a known complication of pembrolizumab, and it has been shown to occur in 1% of patients with this treatment.13,14 It should be noted that most of the studies of this IMAR occurred in patients with metastatic melanoma; therefore the rate of this condition in other patients is less understood. Overall, ocular IMARs secondary to anti-PD-1 and anti-PD-L1 therapies are rare.
The most common IMAR is surface ocular disease, consisting of dry eye disease (DED), conjunctivitis, uveitis, and keratitis. Of these, the most common ocular surface disease is DED, which occurred in 1% to 4% of patients treated with ICI therapy; most of these reactions are mild and self-limiting.15 Atezolizumab has the highest association with ocular inflammation and ipilimumab has the highest association with uveitis, with reported odds ratios of 18.89 and 10.54, respectively.16 Treatment of ICI-induced uveitis generally includes topical steroids and treatment discontinuation or break.17 Oral or IV steroids, infliximab, and procedural involvement may be considered in refractory cases or those initially presenting with marked vision loss. Close communication with ophthalmology colleagues to monitor visual acuity and ocular pressure multiple times weekly during the acute phase is required for treatment titration.
Case 3: Organizing Pneumonia
A man aged 63 years was diagnosed with malignant mesothelioma after incidentally noting a pleural effusion and thickening on routine low-dose computed tomography surveillance of pulmonary nodules. A biopsy was performed and was consistent with mesothelioma, and the patient was started on nivolumab (PD-1 inhibitor) and ipilimumab (CTLA-4 inhibitor). The patient was initiated on dual ICIs, and after 6 months of therapy, he had a promising complete response. However, after 9 months of therapy, he developed a new left upper lobe (LUL) pleural-based lesion (Figure 2A).
A biopsy was performed, and the histopathologic appearance was consistent with organizing pneumonia (OP) (Figure 3).
Discussion
ICIs can uncommonly drive pneumonitis, with the frequency adjusted based on the number of ICIs prescribed and the primary cancer involved. Across all cancers, up to 5% of patients treated with single-agent ICI therapy may experience pneumonitis, though often the findings may simply be radiographic without symptoms. Moreover, up to 10% of patients undergoing treatment for pulmonary cancer or those with dual ICI treatment regimens experience radiographic and/or clinical pneumonitis.18 The clinical manifestations include a broad spectrum of respiratory symptoms. Given the convoluting concerns of cancer progression and infection, a biopsy is often obtained. Histopathologic findings of pneumonitis may include diffuse alveolar damage and/or interstitial lung disease, with OP being a rare variant of ILD.
Among pulmonologists, OP is felt to have polymorphous imaging findings, and biopsy is required to confirm histology; however, histopathology cannot define etiology, and consequently, OP is somewhat of an umbrella diagnosis. The condition can be cryptogenic (idiopathic) or secondary to a multitude of conditions (infection, drug toxicity, or systemic disease). It is classically described as polypoid aggregations of fibroblasts that obstruct the alveolar spaces.19 This histopathologic pattern was demonstrated in our patient’s lung biopsy. Given a prior case description of ICIs, mesothelioma, OP development, and the unremarkable infectious workup, we felt that the patient’s OP was driven by his dual ICI therapy, thereby leading to the ultimate discontinuation of his ICIs and initiation of steroids.20 Thankfully, the patient had already obtained a complete response to his ICIs, and hopefully, he can attain a durable remission with the addition of maintenance chemotherapy.
CONCLUSIONS
ICIs have revolutionized the treatment of a myriad of solid tumors and hematologic malignancies, and their use internationally is expected to increase. With the alteration in immunology pathways, clinicians in all fields will need to be familiarized with IMARs secondary to these agents, including rare subtypes. In addition, the variability in presentations relative to the patients’ treatment course was significant (between 2-9 months), and this highlights that these IMARs can occur at any time point and clinicians should be ever vigilant to spot symptoms in their patients.
It was unexpected for the 3 aforementioned rare toxicities to arise at NMVAMC among only 57 treated patients, and we speculate that these findings may have been observed for 1 of 3 reasons. First, caring for 3 patients with this collection of rare toxicities may have been due to chance. Second, though there is sparse literature studying the topic, the regional environment, including sunlight exposure and air quality, may play a role in the development of one or all of these rare toxicities. Third, rates of these toxicities may be underreported in the literature or attributed to other conditions rather than due to ICIs at other sites, and the uncommon nature of these IMARs may be overstated. Investigations evaluating rates of toxicities, including those traditionally uncommonly seen, based on regional location should be conducted before any further conclusions are drawn.
Immune checkpoint inhibitors (ICIs), often broadly referred to as immunotherapy, are being prescribed at increasing rates due to their effectiveness in treating a growing number of advanced solid tumors and hematologic malignancies.1 It has been well established that T-cell signaling mechanisms designed to combat foreign pathogens have been involved in the mitigation of tumor proliferation.2 This protective process can be supported or restricted by infection, medication, or mutations.
ICIs support T-cell–mediated destruction of tumor cells by inhibiting the mechanisms designed to limit autoimmunity, specifically the programmed cell death protein 1/programmed cell death ligand 1 (PD-1/PD-L1) and cytotoxic T-lymphocyte–associated antigen 4 (CTLA-4) pathways. The results have been impressive, leading to an expansive number of US Food and Drug Administration (FDA) approvals across a diverse set of malignancies. Consequently, the Nobel Prize in Physiology or Medicine was awarded for such work in 2018.3
BACKGROUND
While altering these pathways has been shown to hinder tumor growth, the lesser restrictions on the immune system can drive unwanted autoimmune inflammation to host tissue. These toxicities are collectively known as immune-mediated adverse reactions (IMARs). Clinically and histologically, IMARs frequently manifest similarly to other autoimmune conditions and may affect any organ, including skin, liver, lungs, heart, intestine (small and large), kidneys, eyes, endocrine glands, and neurologic tissue.4,5 According to recent studies, as many as 20% to 30% of patients receiving a single ICI will experience at least 1 clinically significant IMAR, and about 13% are classified as severe; however, < 10% of patients will have their ICIs discontinued due to these reactions.6
Though infrequent, a thorough understanding of the severity of IMARs to ICIs is critical for the diagnosis and management of these organ-threatening and potentially life-threatening toxicities. With the growing use of these agents and more FDA approvals for dual checkpoint blockage (concurrent use of CTLA-4 and PD-1/PD-L1 inhibitors), the absolute number of IMARs is expected to rise, thereby leading to more exposure of such events to both oncology and nononcology clinicians. Prior literature has clearly described the treatments and outcomes for many common severe toxicities; however, information regarding presentations and outcomes for rare IMARs is lacking.7
A few fascinating cases of rare toxicities have been observed at the New Mexico Veterans Affairs Medical Center (NMVAMC) in Albuquerque despite its relatively small size compared with other US Department of Veterans Affairs medical centers. As such, herein, the diagnostic evaluation, treatments, and outcomes of rare IMARs are reported for each case, and the related literature is reviewed.
Patient Selection
Patients who were required to discontinue or postpone treatment with any ICI blocking the CTLA-4 (ipilimumab), PD-1 (pembrolizumab, nivolumab, cemiplimab), or PD-L1 (atezolizumab, avelumab, durvalumab) pathways between 2015 to 2021 due to toxicity at the NMVAMC were eligible for inclusion. The electronic health record was reviewed for each eligible case, and the patient demographics, disease characteristics, toxicities, and outcomes were documented for each patient. For the 57 patients who received ICIs within the chosen period, 11 required a treatment break or discontinuation. Of these, 3 cases were selected for reporting due to the rare IMARs observed. This study was approved by the NMVAMC Institutional Review Board.
Case 1: Myocarditis
An 84-year-old man receiving a chemoimmunotherapy regimen consisting of carboplatin, pemetrexed, and pembrolizumab for recurrent, stage IV lung adenocarcinoma developed grade 4 cardiomyopathy, as defined by the Common Terminology Criteria for Adverse Events (CTCAE) v5.0, during his treatment.8 He was treated for 2 cycles before he began experiencing an increase in liver enzymes.
The patient’s presentation was concerning for myocarditis, and he was quickly admitted to NMVAMC. Cardiac catheterization did not reveal any signs of coronary occlusive disease. Prednisone 1 mg/kg was administered immediately; however, given continued chest pain and volume overload, he was quickly transitioned to solumedrol 1000 mg IV daily. After the initiation of his treatment, the patient’s transaminitis began to resolve, and troponin levels began to decrease; however, his symptoms continued to worsen, and his troponin rose again. By the fourth day of hospitalization, the patient was treated with infliximab, a tumor necrosis factor-α inhibitor shown to reverse ICI-induced autoimmune inflammation, with only mild improvement of his symptoms. The patient’s condition continued to deteriorate, his troponin levels remained elevated, and his family decided to withhold additional treatment. The patient died shortly thereafter.
Discussion
Cardiotoxicity resulting from ICI therapy is far less common than the other potential severe toxicities associated with ICIs. Nevertheless, many cases of ICI-induced cardiac inflammation have been reported, and it has been widely established that patients treated with ICIs are generally at higher risk for acute coronary syndrome.9-11 Acute cardiotoxicity secondary to autoimmune destruction of cardiac tissue includes myocarditis, pericarditis, and vasculitis, which may manifest with symptoms of heart failure and/or arrhythmia. Grading of ICI-induced cardiomyopathy has been defined by both CTCAE and the American Society of Clinical Oncology (ASCO), with grade 4 representing moderate to severe clinical decompensation requiring IV medications in the setting of life-threatening conditions.
Review articles have described the treatment options for severe cases.7,12 As detailed in prior reports, once ICI-induced cardiomyopathy is suspected, urgent admission and immediate evaluation to rule out acute coronary syndrome should be undertaken. Given the potential for deterioration despite the occasional insidious onset, aggressive cardiac monitoring, and close follow-up to measure response to interventions should be undertaken.
Case 2: Uveitis
A 70-year-old man who received pembrolizumab as a bladder-sparing approach for his superficial bladder cancer refractory to intravesical treatments developed uveitis. Approximately 3 months following the initiation of treatment, the patient reported bilateral itchy eyes, erythema, and tearing. He had a known history of allergic conjunctivitis that predated the ICI therapy, and consequently, it was unclear whether his symptoms were reflective of a more concerning issue. The patient’s symptoms continued to wax and wane for a few months, prompting a referral to ophthalmology colleagues at NMVAMC.
Ophthalmology evaluation identified uveitic glaucoma in the setting of his underlying chronic glaucoma. Pembrolizumab was discontinued, and the patient was counseled on choosing either cystectomy or locoregional therapies if further tumors arose. However, within a few weeks of administering topical steroid drops, his symptoms markedly improved, and he wished to be restarted on pembrolizumab. His uveitis remained in remission, and he has been treated with pembrolizumab for more than 1 year since this episode. He has had no clear findings of superficial bladder cancer recurrence while receiving ICI therapy.
Discussion
Uveitis is a known complication of pembrolizumab, and it has been shown to occur in 1% of patients with this treatment.13,14 It should be noted that most of the studies of this IMAR occurred in patients with metastatic melanoma; therefore the rate of this condition in other patients is less understood. Overall, ocular IMARs secondary to anti-PD-1 and anti-PD-L1 therapies are rare.
The most common IMAR is surface ocular disease, consisting of dry eye disease (DED), conjunctivitis, uveitis, and keratitis. Of these, the most common ocular surface disease is DED, which occurred in 1% to 4% of patients treated with ICI therapy; most of these reactions are mild and self-limiting.15 Atezolizumab has the highest association with ocular inflammation and ipilimumab has the highest association with uveitis, with reported odds ratios of 18.89 and 10.54, respectively.16 Treatment of ICI-induced uveitis generally includes topical steroids and treatment discontinuation or break.17 Oral or IV steroids, infliximab, and procedural involvement may be considered in refractory cases or those initially presenting with marked vision loss. Close communication with ophthalmology colleagues to monitor visual acuity and ocular pressure multiple times weekly during the acute phase is required for treatment titration.
Case 3: Organizing Pneumonia
A man aged 63 years was diagnosed with malignant mesothelioma after incidentally noting a pleural effusion and thickening on routine low-dose computed tomography surveillance of pulmonary nodules. A biopsy was performed and was consistent with mesothelioma, and the patient was started on nivolumab (PD-1 inhibitor) and ipilimumab (CTLA-4 inhibitor). The patient was initiated on dual ICIs, and after 6 months of therapy, he had a promising complete response. However, after 9 months of therapy, he developed a new left upper lobe (LUL) pleural-based lesion (Figure 2A).
A biopsy was performed, and the histopathologic appearance was consistent with organizing pneumonia (OP) (Figure 3).
Discussion
ICIs can uncommonly drive pneumonitis, with the frequency adjusted based on the number of ICIs prescribed and the primary cancer involved. Across all cancers, up to 5% of patients treated with single-agent ICI therapy may experience pneumonitis, though often the findings may simply be radiographic without symptoms. Moreover, up to 10% of patients undergoing treatment for pulmonary cancer or those with dual ICI treatment regimens experience radiographic and/or clinical pneumonitis.18 The clinical manifestations include a broad spectrum of respiratory symptoms. Given the convoluting concerns of cancer progression and infection, a biopsy is often obtained. Histopathologic findings of pneumonitis may include diffuse alveolar damage and/or interstitial lung disease, with OP being a rare variant of ILD.
Among pulmonologists, OP is felt to have polymorphous imaging findings, and biopsy is required to confirm histology; however, histopathology cannot define etiology, and consequently, OP is somewhat of an umbrella diagnosis. The condition can be cryptogenic (idiopathic) or secondary to a multitude of conditions (infection, drug toxicity, or systemic disease). It is classically described as polypoid aggregations of fibroblasts that obstruct the alveolar spaces.19 This histopathologic pattern was demonstrated in our patient’s lung biopsy. Given a prior case description of ICIs, mesothelioma, OP development, and the unremarkable infectious workup, we felt that the patient’s OP was driven by his dual ICI therapy, thereby leading to the ultimate discontinuation of his ICIs and initiation of steroids.20 Thankfully, the patient had already obtained a complete response to his ICIs, and hopefully, he can attain a durable remission with the addition of maintenance chemotherapy.
CONCLUSIONS
ICIs have revolutionized the treatment of a myriad of solid tumors and hematologic malignancies, and their use internationally is expected to increase. With the alteration in immunology pathways, clinicians in all fields will need to be familiarized with IMARs secondary to these agents, including rare subtypes. In addition, the variability in presentations relative to the patients’ treatment course was significant (between 2-9 months), and this highlights that these IMARs can occur at any time point and clinicians should be ever vigilant to spot symptoms in their patients.
It was unexpected for the 3 aforementioned rare toxicities to arise at NMVAMC among only 57 treated patients, and we speculate that these findings may have been observed for 1 of 3 reasons. First, caring for 3 patients with this collection of rare toxicities may have been due to chance. Second, though there is sparse literature studying the topic, the regional environment, including sunlight exposure and air quality, may play a role in the development of one or all of these rare toxicities. Third, rates of these toxicities may be underreported in the literature or attributed to other conditions rather than due to ICIs at other sites, and the uncommon nature of these IMARs may be overstated. Investigations evaluating rates of toxicities, including those traditionally uncommonly seen, based on regional location should be conducted before any further conclusions are drawn.
1. Bagchi S, Yuan R, Engleman EG. Immune checkpoint inhibitors for the treatment of cancer: clinical impact and mechanisms of response and resistance. Published online 2020. doi:10.1146/annurev-pathol-042020
2. Chen DS, Mellman I. Oncology meets immunology: The cancer-immunity cycle. Immunity. 2013;39(1):1-10. doi:10.1016/j.immuni.2013.07.012
3. Smyth MJ, Teng MWL. 2018 Nobel Prize in physiology or medicine. Clin Transl Immunology. 2018;7(10). doi:10.1002/cti2.1041
4. Baxi S, Yang A, Gennarelli RL, et al. Immune-related adverse events for anti-PD-1 and anti-PD-L1 drugs: Systematic review and meta-analysis. BMJ (Online). 2018;360. doi:10.1136/bmj.k793
5. Ellithi M, Elnair R, Chang GV, Abdallah MA. Toxicities of immune checkpoint inhibitors: itis-ending adverse reactions and more. Cureus. Published online February 10, 2020. doi:10.7759/cureus.6935
6. Berti A, Bortolotti R, Dipasquale M, et al. Meta-analysis of immune-related adverse events in phase 3 clinical trials assessing immune checkpoint inhibitors for lung cancer. Crit Rev Oncol Hematol. 2021;162. doi:10.1016/j.critrevonc.2021.103351
7. Davies M, Duffield EA. Safety of checkpoint inhibitors for cancer treatment: strategies for patient monitoring and management of immune-mediated adverse events. Immunotargets Ther. 2017;Volume 6:51-71. doi:10.2147/itt.s141577
8. US Department of Health and Human Services. Common Terminology Criteria for Adverse Events V5.0. Accessed July 17, 2023. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5584920/
9. Johnson DB, Balko JM, Compton ML, et al. Fulminant myocarditis with combination immune checkpoint blockade. N Engl J Med. 2016;375(18):1749-1755. doi:10.1056/nejmoa1609214
10. Mahmood SS, Fradley MG, Cohen J V., et al. Myocarditis in patients treated with immune checkpoint inhibitors. J Am Coll Cardiol. 2018;71(16):1755-1764. doi:10.1016/j.jacc.2018.02.037
11. Wang DY, Salem JE, Cohen JV, et al. Fatal toxic effects associated with immune checkpoint inhibitors: a systematic review and meta-analysis. JAMA Oncol. 2018;4(12):1721-1728. doi:10.1001/jamaoncol.2018.3923
12. Brahmer JR, Lacchetti C, Schneider BJ, et al; National Comprehensive Cancer Network. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Onc. 2018;36(17):1714-1768. doi:10.1200/JCO
13. Ribas A, Hamid O, Daud A, et al. Association of pembrolizumab with tumor response and survival among patients with advanced melanoma. JAMA. 2016;315:1600-1609. doi:10.1001/jama.2016.4059
14. Dalvin LA, Shields CL, Orloff M, Sato T, Shields JA. Checkpoint inhibitor immune therapy: systemic indications and ophthalmic side effects. Retina. 2018;38(6):1063-1078. doi:10.1097/IAE.0000000000002181
15. Park RB, Jain S, Han H, Park J. Ocular surface disease associated with immune checkpoint inhibitor therapy. Ocular Surface. 2021;20:115-129. doi:10.1016/j.jtos.2021.02.004
16. Fang T, Maberley DA, Etminan M. Ocular adverse events with immune checkpoint inhibitors. J Curr Ophthalmol. 2019;31(3):319-322. doi:10.1016/j.joco.2019.05.002
17. Whist E, Symes RJ, Chang JH, et al. Uveitis caused by treatment for malignant melanoma: a case series. Retin Cases Brief Rep. 2021;15(6):718-723. doi:10.1097/ICB.0000000000000876
18. Naidoo J, Wang X, Woo KM, et al. Pneumonitis in patients treated with anti-programmed death-1/programmed death ligand 1 therapy. J Clin Onc. 2017;35(7):709-717. doi:10.1200/JCO.2016.68.2005
19. Yoshikawa A, Bychkov A, Sathirareuangchai S. Other nonneoplastic conditions, acute lung injury, organizing pneumonia. Accessed July 17, 2023. https://www.pathologyoutlines.com/topic/lungnontumorboop.html
20. Kuint R, Lotem M, Neuman T, et al. Organizing pneumonia following treatment with pembrolizumab for metastatic malignant melanoma–a case report. Respir Med Case Rep. 2017;20:95-97. doi:10.1016/j.rmcr.2017.01.003
1. Bagchi S, Yuan R, Engleman EG. Immune checkpoint inhibitors for the treatment of cancer: clinical impact and mechanisms of response and resistance. Published online 2020. doi:10.1146/annurev-pathol-042020
2. Chen DS, Mellman I. Oncology meets immunology: The cancer-immunity cycle. Immunity. 2013;39(1):1-10. doi:10.1016/j.immuni.2013.07.012
3. Smyth MJ, Teng MWL. 2018 Nobel Prize in physiology or medicine. Clin Transl Immunology. 2018;7(10). doi:10.1002/cti2.1041
4. Baxi S, Yang A, Gennarelli RL, et al. Immune-related adverse events for anti-PD-1 and anti-PD-L1 drugs: Systematic review and meta-analysis. BMJ (Online). 2018;360. doi:10.1136/bmj.k793
5. Ellithi M, Elnair R, Chang GV, Abdallah MA. Toxicities of immune checkpoint inhibitors: itis-ending adverse reactions and more. Cureus. Published online February 10, 2020. doi:10.7759/cureus.6935
6. Berti A, Bortolotti R, Dipasquale M, et al. Meta-analysis of immune-related adverse events in phase 3 clinical trials assessing immune checkpoint inhibitors for lung cancer. Crit Rev Oncol Hematol. 2021;162. doi:10.1016/j.critrevonc.2021.103351
7. Davies M, Duffield EA. Safety of checkpoint inhibitors for cancer treatment: strategies for patient monitoring and management of immune-mediated adverse events. Immunotargets Ther. 2017;Volume 6:51-71. doi:10.2147/itt.s141577
8. US Department of Health and Human Services. Common Terminology Criteria for Adverse Events V5.0. Accessed July 17, 2023. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5584920/
9. Johnson DB, Balko JM, Compton ML, et al. Fulminant myocarditis with combination immune checkpoint blockade. N Engl J Med. 2016;375(18):1749-1755. doi:10.1056/nejmoa1609214
10. Mahmood SS, Fradley MG, Cohen J V., et al. Myocarditis in patients treated with immune checkpoint inhibitors. J Am Coll Cardiol. 2018;71(16):1755-1764. doi:10.1016/j.jacc.2018.02.037
11. Wang DY, Salem JE, Cohen JV, et al. Fatal toxic effects associated with immune checkpoint inhibitors: a systematic review and meta-analysis. JAMA Oncol. 2018;4(12):1721-1728. doi:10.1001/jamaoncol.2018.3923
12. Brahmer JR, Lacchetti C, Schneider BJ, et al; National Comprehensive Cancer Network. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Onc. 2018;36(17):1714-1768. doi:10.1200/JCO
13. Ribas A, Hamid O, Daud A, et al. Association of pembrolizumab with tumor response and survival among patients with advanced melanoma. JAMA. 2016;315:1600-1609. doi:10.1001/jama.2016.4059
14. Dalvin LA, Shields CL, Orloff M, Sato T, Shields JA. Checkpoint inhibitor immune therapy: systemic indications and ophthalmic side effects. Retina. 2018;38(6):1063-1078. doi:10.1097/IAE.0000000000002181
15. Park RB, Jain S, Han H, Park J. Ocular surface disease associated with immune checkpoint inhibitor therapy. Ocular Surface. 2021;20:115-129. doi:10.1016/j.jtos.2021.02.004
16. Fang T, Maberley DA, Etminan M. Ocular adverse events with immune checkpoint inhibitors. J Curr Ophthalmol. 2019;31(3):319-322. doi:10.1016/j.joco.2019.05.002
17. Whist E, Symes RJ, Chang JH, et al. Uveitis caused by treatment for malignant melanoma: a case series. Retin Cases Brief Rep. 2021;15(6):718-723. doi:10.1097/ICB.0000000000000876
18. Naidoo J, Wang X, Woo KM, et al. Pneumonitis in patients treated with anti-programmed death-1/programmed death ligand 1 therapy. J Clin Onc. 2017;35(7):709-717. doi:10.1200/JCO.2016.68.2005
19. Yoshikawa A, Bychkov A, Sathirareuangchai S. Other nonneoplastic conditions, acute lung injury, organizing pneumonia. Accessed July 17, 2023. https://www.pathologyoutlines.com/topic/lungnontumorboop.html
20. Kuint R, Lotem M, Neuman T, et al. Organizing pneumonia following treatment with pembrolizumab for metastatic malignant melanoma–a case report. Respir Med Case Rep. 2017;20:95-97. doi:10.1016/j.rmcr.2017.01.003
Gastrointestinal Bleeding Caused by Large Intestine Amyloidosis
Gastrointestinal (GI) bleeding is a common cause of hospital admissions. The yearly incidence of upper GI bleeding is 80 to 150/100,000 people and lower GI bleeding is 87/100,000 people.1,2 The differential tends to initially be broad but narrows with good history followed by endoscopic findings. Getting an appropriate history can be difficult at times, which leads health care practitioners to rely more on interventional results.
Amyloidosis is a rare disorder of abnormal protein folding, leading to the deposition of insoluble fibrils that disrupt normal tissues and cause disease.3 There are 2 main types of amyloidosis, systemic and transthyretin, and 4 subtypes. Systemic amyloidosis includes amyloid light-chain (AL) deposition, caused by plasma cell dyscrasia, and amyloid A (AA) protein deposition, caused by systemic autoimmune illness or infections. Transthyretin amyloidosis is caused by changes and deposition of the transthyretin protein consisting of either unstable, mutant protein or wild type protein. Biopsy-proven amyloidosis of the GI tract is rare.4 About 60% of patients with AA amyloidosis and 8% with AL amyloidosis have GI involvement.5
We present a case of nonspecific symptoms that ultimately lined up perfectly with the official histologic confirmation of intestinal amyloidosis.
Case Presentation
A 79-year-old man with a history of type 2 diabetes mellitus, congestive heart failure, hyperlipidemia, obstructive sleep apnea, hypothyroidism, hypertension, coronary artery disease status postcoronary artery bypass grafting, and stent placements presented for 3 episodes of large, bright red bowel movements. He reported past bleeding and straining with stools, but bleeding of this amount had not been noted prior. He also reported dry heaves, lower abdominal pain, constipation with straining, early satiety with dysphagia, weakness, and decreased appetite. Lastly, he mentioned intentionally losing about 35 to 40 pounds in the past 3 to 4 months and over the past several months increased abdominal distention. However, he stated he had no history of alcohol misuse, liver or intestinal disease, cirrhosis, or other autoimmune diseases. His most recent colonoscopy was more than a decade prior and showed no acute process. The patient never had an esophagogastroduodenoscopy (EGD).
On initial presentation, the patient’s vital signs showed no acute findings. His physical examination noted a chronically ill–appearing male with decreased breath sounds to the bases bilaterally and noted abdominal distention with mild generalized tenderness. Laboratory findings were significant for a hemoglobin level, 9.4 g/dL (reference range, 11.6-15.3); iron, 23 ug/dL (reference range, 45-160); transferrin saturation, 8% (reference range, 15-50); ferritin level, 80 ng/mL (reference range, 30-300); and carcinoembryonic antigen level, 1.5 ng/mL (reference range, 0-2.9). Aspartate aminotransferase level was 54 IU/L (reference range, 0-40); alanine transaminase, 24 IU/L (reference range, 7-52); albumin, 2.7 g/dL (reference range, 3.4-5.7); international normalized ratio, 1.3 (reference range, 0-1.1); creatinine, 1.74 mg/dL (reference range, 0.44-1.27); alkaline phosphatase, 369 IU/L (reference range, 39-117). White blood cell count was 15.5 × 109/L (reference range, 3.5-10.3), and lactic acid was 2.5 mmol/L (reference range, 0.5-2.2). He was started on piperacillin/tazobactam in the emergency department and transitioned to ciprofloxacin and metronidazole for presumed intra-abdominal infection. Paracentesis showed a serum ascites albumin gradient of > 1.1 g/dL with no signs of spontaneous bacterial peritonitis. Computed tomography of the abdomen and pelvis with contrast was suspicious for colitis involving the proximal colon, and colonic mass could not be excluded. Also noted was hepatosplenomegaly with abdominopelvic ascites.
Based on these findings, an EGD and colonoscopy were done. The EGD showed mild portal hypertensive gastropathy.
After the biopsy results, the patient was officially diagnosed with intestinal amyloidosis (Figure 2).
He returned to the gastroenterology clinic 2 months later. At that point, he had worsening symptoms, liver function test results, and international normalized ratio. He was admitted for further investigation. A bone biopsy was done to confirm the histology and define the underlying disorder. The biopsy returned showing Waldenstrom macroglobulinemia, and he was started on bortezomib. Unfortunately, his clinical status rapidly worsened, leading to acute renal and hepatic failure and the development of encephalopathy. He eventually died under palliative care services.
Discussion
Amyloidosis is a rare disorder of abnormal protein folding, leading to the deposition of insoluble fibrils that disrupt normal tissues and cause disease.3 There are several variations of amyloid, but the most common type is AL amyloidosis, which affects several organs, including the heart, kidney, liver, nervous system, and GI tract. When AL amyloidosis involves the liver, the median survival time is about 8.5 months.6 There are different ways to diagnose the disease, but a tissue biopsy and Congo Red staining can confirm specific organ involvement as seen in our case.
This case adds another layer to our constantly expanding differential as health care practitioners and proves that atypical patient presentations may not be atypical after all. GI amyloidosis tends to present similarly to our patient with bleeding, malabsorption, dysmotility, and protein-losing gastroenteropathy as ascites, edema, pericardial effusions, and laboratory evidence of hypoalbuminemia.7 Because amyloidosis is a systemic illness, early recognition is important as intestinal complications tend to present as symptoms, but mortality is more often caused by renal failure, cardiomyopathy, or ischemic heart disease, making early multispecialty involvement very important.8
Conclusions
Health care practitioners in all specialties should be aware of and include intestinal amyloidosis in their differential diagnosis when working up GI bleeds with the hope of identifying the disease early. With early recognition, rapid biopsy identification, and early specialist involvement, patients will get the opportunity for expedited multidisciplinary treatment and potentially delay rapid decompensation as shown by the evidence in this case.
1. Antunes C, Copelin II EL. Upper gastrointestinal bleeding. StatPearls [internet]. Updated July 18, 2022. Accessed May 25, 2023. https://www.ncbi.nlm.nih.gov/books/NBK470300
2. Almaghrabi M, Gandhi M, Guizzetti L, et al. Comparison of risk scores for lower gastrointestinal bleeding: a systematic review and meta-analysis. JAMA Netw Open. 2022;5(5):e2214253. doi:10.1001/jamanetworkopen.2022.14253
3. Pepys MB. Pathogenesis, diagnosis and treatment of systemic amyloidosis. Philos Trans R Soc Lond B Biol Sci. 2001;356(1406):203-211. doi:10.1098/rstb.2000.0766
4. Cowan AJ, Skinner M, Seldin DC, et al. Amyloidosis of the gastrointestinal tract: a 13-year, single-center, referral experience. Haematologica. 2013;98(1):141-146. doi:10.3324/haematol.2012.068155
5. Lee BS, Chudasama Y, Chen AI, Lim BS, Taira MT. Colonoscopy leading to the diagnosis of AL amyloidosis in the gastrointestinal tract mimicking an acute ulcerative colitis flare. ACG Case Rep J. 2019;6(11):e00289. doi:10.14309/crj.0000000000000289
6. Zhao L, Ren G, Guo J, Chen W, Xu W, Huang X. The clinical features and outcomes of systemic light chain amyloidosis with hepatic involvement. Ann Med. 2022;54(1):1226-1232. doi:10.1080/07853890.2022.2069281
7. Rowe K, Pankow J, Nehme F, Salyers W. Gastrointestinal amyloidosis: review of the literature. Cureus. 2017;9(5):e1228. doi:10.7759/cureus.1228
8. Kyle RA, Greipp PR, O’Fallon WM. Primary systemic amyloidosis: multivariate analysis for prognostic factors in 168 cases. Blood. 1986;68(1):220-224.
Gastrointestinal (GI) bleeding is a common cause of hospital admissions. The yearly incidence of upper GI bleeding is 80 to 150/100,000 people and lower GI bleeding is 87/100,000 people.1,2 The differential tends to initially be broad but narrows with good history followed by endoscopic findings. Getting an appropriate history can be difficult at times, which leads health care practitioners to rely more on interventional results.
Amyloidosis is a rare disorder of abnormal protein folding, leading to the deposition of insoluble fibrils that disrupt normal tissues and cause disease.3 There are 2 main types of amyloidosis, systemic and transthyretin, and 4 subtypes. Systemic amyloidosis includes amyloid light-chain (AL) deposition, caused by plasma cell dyscrasia, and amyloid A (AA) protein deposition, caused by systemic autoimmune illness or infections. Transthyretin amyloidosis is caused by changes and deposition of the transthyretin protein consisting of either unstable, mutant protein or wild type protein. Biopsy-proven amyloidosis of the GI tract is rare.4 About 60% of patients with AA amyloidosis and 8% with AL amyloidosis have GI involvement.5
We present a case of nonspecific symptoms that ultimately lined up perfectly with the official histologic confirmation of intestinal amyloidosis.
Case Presentation
A 79-year-old man with a history of type 2 diabetes mellitus, congestive heart failure, hyperlipidemia, obstructive sleep apnea, hypothyroidism, hypertension, coronary artery disease status postcoronary artery bypass grafting, and stent placements presented for 3 episodes of large, bright red bowel movements. He reported past bleeding and straining with stools, but bleeding of this amount had not been noted prior. He also reported dry heaves, lower abdominal pain, constipation with straining, early satiety with dysphagia, weakness, and decreased appetite. Lastly, he mentioned intentionally losing about 35 to 40 pounds in the past 3 to 4 months and over the past several months increased abdominal distention. However, he stated he had no history of alcohol misuse, liver or intestinal disease, cirrhosis, or other autoimmune diseases. His most recent colonoscopy was more than a decade prior and showed no acute process. The patient never had an esophagogastroduodenoscopy (EGD).
On initial presentation, the patient’s vital signs showed no acute findings. His physical examination noted a chronically ill–appearing male with decreased breath sounds to the bases bilaterally and noted abdominal distention with mild generalized tenderness. Laboratory findings were significant for a hemoglobin level, 9.4 g/dL (reference range, 11.6-15.3); iron, 23 ug/dL (reference range, 45-160); transferrin saturation, 8% (reference range, 15-50); ferritin level, 80 ng/mL (reference range, 30-300); and carcinoembryonic antigen level, 1.5 ng/mL (reference range, 0-2.9). Aspartate aminotransferase level was 54 IU/L (reference range, 0-40); alanine transaminase, 24 IU/L (reference range, 7-52); albumin, 2.7 g/dL (reference range, 3.4-5.7); international normalized ratio, 1.3 (reference range, 0-1.1); creatinine, 1.74 mg/dL (reference range, 0.44-1.27); alkaline phosphatase, 369 IU/L (reference range, 39-117). White blood cell count was 15.5 × 109/L (reference range, 3.5-10.3), and lactic acid was 2.5 mmol/L (reference range, 0.5-2.2). He was started on piperacillin/tazobactam in the emergency department and transitioned to ciprofloxacin and metronidazole for presumed intra-abdominal infection. Paracentesis showed a serum ascites albumin gradient of > 1.1 g/dL with no signs of spontaneous bacterial peritonitis. Computed tomography of the abdomen and pelvis with contrast was suspicious for colitis involving the proximal colon, and colonic mass could not be excluded. Also noted was hepatosplenomegaly with abdominopelvic ascites.
Based on these findings, an EGD and colonoscopy were done. The EGD showed mild portal hypertensive gastropathy.
After the biopsy results, the patient was officially diagnosed with intestinal amyloidosis (Figure 2).
He returned to the gastroenterology clinic 2 months later. At that point, he had worsening symptoms, liver function test results, and international normalized ratio. He was admitted for further investigation. A bone biopsy was done to confirm the histology and define the underlying disorder. The biopsy returned showing Waldenstrom macroglobulinemia, and he was started on bortezomib. Unfortunately, his clinical status rapidly worsened, leading to acute renal and hepatic failure and the development of encephalopathy. He eventually died under palliative care services.
Discussion
Amyloidosis is a rare disorder of abnormal protein folding, leading to the deposition of insoluble fibrils that disrupt normal tissues and cause disease.3 There are several variations of amyloid, but the most common type is AL amyloidosis, which affects several organs, including the heart, kidney, liver, nervous system, and GI tract. When AL amyloidosis involves the liver, the median survival time is about 8.5 months.6 There are different ways to diagnose the disease, but a tissue biopsy and Congo Red staining can confirm specific organ involvement as seen in our case.
This case adds another layer to our constantly expanding differential as health care practitioners and proves that atypical patient presentations may not be atypical after all. GI amyloidosis tends to present similarly to our patient with bleeding, malabsorption, dysmotility, and protein-losing gastroenteropathy as ascites, edema, pericardial effusions, and laboratory evidence of hypoalbuminemia.7 Because amyloidosis is a systemic illness, early recognition is important as intestinal complications tend to present as symptoms, but mortality is more often caused by renal failure, cardiomyopathy, or ischemic heart disease, making early multispecialty involvement very important.8
Conclusions
Health care practitioners in all specialties should be aware of and include intestinal amyloidosis in their differential diagnosis when working up GI bleeds with the hope of identifying the disease early. With early recognition, rapid biopsy identification, and early specialist involvement, patients will get the opportunity for expedited multidisciplinary treatment and potentially delay rapid decompensation as shown by the evidence in this case.
Gastrointestinal (GI) bleeding is a common cause of hospital admissions. The yearly incidence of upper GI bleeding is 80 to 150/100,000 people and lower GI bleeding is 87/100,000 people.1,2 The differential tends to initially be broad but narrows with good history followed by endoscopic findings. Getting an appropriate history can be difficult at times, which leads health care practitioners to rely more on interventional results.
Amyloidosis is a rare disorder of abnormal protein folding, leading to the deposition of insoluble fibrils that disrupt normal tissues and cause disease.3 There are 2 main types of amyloidosis, systemic and transthyretin, and 4 subtypes. Systemic amyloidosis includes amyloid light-chain (AL) deposition, caused by plasma cell dyscrasia, and amyloid A (AA) protein deposition, caused by systemic autoimmune illness or infections. Transthyretin amyloidosis is caused by changes and deposition of the transthyretin protein consisting of either unstable, mutant protein or wild type protein. Biopsy-proven amyloidosis of the GI tract is rare.4 About 60% of patients with AA amyloidosis and 8% with AL amyloidosis have GI involvement.5
We present a case of nonspecific symptoms that ultimately lined up perfectly with the official histologic confirmation of intestinal amyloidosis.
Case Presentation
A 79-year-old man with a history of type 2 diabetes mellitus, congestive heart failure, hyperlipidemia, obstructive sleep apnea, hypothyroidism, hypertension, coronary artery disease status postcoronary artery bypass grafting, and stent placements presented for 3 episodes of large, bright red bowel movements. He reported past bleeding and straining with stools, but bleeding of this amount had not been noted prior. He also reported dry heaves, lower abdominal pain, constipation with straining, early satiety with dysphagia, weakness, and decreased appetite. Lastly, he mentioned intentionally losing about 35 to 40 pounds in the past 3 to 4 months and over the past several months increased abdominal distention. However, he stated he had no history of alcohol misuse, liver or intestinal disease, cirrhosis, or other autoimmune diseases. His most recent colonoscopy was more than a decade prior and showed no acute process. The patient never had an esophagogastroduodenoscopy (EGD).
On initial presentation, the patient’s vital signs showed no acute findings. His physical examination noted a chronically ill–appearing male with decreased breath sounds to the bases bilaterally and noted abdominal distention with mild generalized tenderness. Laboratory findings were significant for a hemoglobin level, 9.4 g/dL (reference range, 11.6-15.3); iron, 23 ug/dL (reference range, 45-160); transferrin saturation, 8% (reference range, 15-50); ferritin level, 80 ng/mL (reference range, 30-300); and carcinoembryonic antigen level, 1.5 ng/mL (reference range, 0-2.9). Aspartate aminotransferase level was 54 IU/L (reference range, 0-40); alanine transaminase, 24 IU/L (reference range, 7-52); albumin, 2.7 g/dL (reference range, 3.4-5.7); international normalized ratio, 1.3 (reference range, 0-1.1); creatinine, 1.74 mg/dL (reference range, 0.44-1.27); alkaline phosphatase, 369 IU/L (reference range, 39-117). White blood cell count was 15.5 × 109/L (reference range, 3.5-10.3), and lactic acid was 2.5 mmol/L (reference range, 0.5-2.2). He was started on piperacillin/tazobactam in the emergency department and transitioned to ciprofloxacin and metronidazole for presumed intra-abdominal infection. Paracentesis showed a serum ascites albumin gradient of > 1.1 g/dL with no signs of spontaneous bacterial peritonitis. Computed tomography of the abdomen and pelvis with contrast was suspicious for colitis involving the proximal colon, and colonic mass could not be excluded. Also noted was hepatosplenomegaly with abdominopelvic ascites.
Based on these findings, an EGD and colonoscopy were done. The EGD showed mild portal hypertensive gastropathy.
After the biopsy results, the patient was officially diagnosed with intestinal amyloidosis (Figure 2).
He returned to the gastroenterology clinic 2 months later. At that point, he had worsening symptoms, liver function test results, and international normalized ratio. He was admitted for further investigation. A bone biopsy was done to confirm the histology and define the underlying disorder. The biopsy returned showing Waldenstrom macroglobulinemia, and he was started on bortezomib. Unfortunately, his clinical status rapidly worsened, leading to acute renal and hepatic failure and the development of encephalopathy. He eventually died under palliative care services.
Discussion
Amyloidosis is a rare disorder of abnormal protein folding, leading to the deposition of insoluble fibrils that disrupt normal tissues and cause disease.3 There are several variations of amyloid, but the most common type is AL amyloidosis, which affects several organs, including the heart, kidney, liver, nervous system, and GI tract. When AL amyloidosis involves the liver, the median survival time is about 8.5 months.6 There are different ways to diagnose the disease, but a tissue biopsy and Congo Red staining can confirm specific organ involvement as seen in our case.
This case adds another layer to our constantly expanding differential as health care practitioners and proves that atypical patient presentations may not be atypical after all. GI amyloidosis tends to present similarly to our patient with bleeding, malabsorption, dysmotility, and protein-losing gastroenteropathy as ascites, edema, pericardial effusions, and laboratory evidence of hypoalbuminemia.7 Because amyloidosis is a systemic illness, early recognition is important as intestinal complications tend to present as symptoms, but mortality is more often caused by renal failure, cardiomyopathy, or ischemic heart disease, making early multispecialty involvement very important.8
Conclusions
Health care practitioners in all specialties should be aware of and include intestinal amyloidosis in their differential diagnosis when working up GI bleeds with the hope of identifying the disease early. With early recognition, rapid biopsy identification, and early specialist involvement, patients will get the opportunity for expedited multidisciplinary treatment and potentially delay rapid decompensation as shown by the evidence in this case.
1. Antunes C, Copelin II EL. Upper gastrointestinal bleeding. StatPearls [internet]. Updated July 18, 2022. Accessed May 25, 2023. https://www.ncbi.nlm.nih.gov/books/NBK470300
2. Almaghrabi M, Gandhi M, Guizzetti L, et al. Comparison of risk scores for lower gastrointestinal bleeding: a systematic review and meta-analysis. JAMA Netw Open. 2022;5(5):e2214253. doi:10.1001/jamanetworkopen.2022.14253
3. Pepys MB. Pathogenesis, diagnosis and treatment of systemic amyloidosis. Philos Trans R Soc Lond B Biol Sci. 2001;356(1406):203-211. doi:10.1098/rstb.2000.0766
4. Cowan AJ, Skinner M, Seldin DC, et al. Amyloidosis of the gastrointestinal tract: a 13-year, single-center, referral experience. Haematologica. 2013;98(1):141-146. doi:10.3324/haematol.2012.068155
5. Lee BS, Chudasama Y, Chen AI, Lim BS, Taira MT. Colonoscopy leading to the diagnosis of AL amyloidosis in the gastrointestinal tract mimicking an acute ulcerative colitis flare. ACG Case Rep J. 2019;6(11):e00289. doi:10.14309/crj.0000000000000289
6. Zhao L, Ren G, Guo J, Chen W, Xu W, Huang X. The clinical features and outcomes of systemic light chain amyloidosis with hepatic involvement. Ann Med. 2022;54(1):1226-1232. doi:10.1080/07853890.2022.2069281
7. Rowe K, Pankow J, Nehme F, Salyers W. Gastrointestinal amyloidosis: review of the literature. Cureus. 2017;9(5):e1228. doi:10.7759/cureus.1228
8. Kyle RA, Greipp PR, O’Fallon WM. Primary systemic amyloidosis: multivariate analysis for prognostic factors in 168 cases. Blood. 1986;68(1):220-224.
1. Antunes C, Copelin II EL. Upper gastrointestinal bleeding. StatPearls [internet]. Updated July 18, 2022. Accessed May 25, 2023. https://www.ncbi.nlm.nih.gov/books/NBK470300
2. Almaghrabi M, Gandhi M, Guizzetti L, et al. Comparison of risk scores for lower gastrointestinal bleeding: a systematic review and meta-analysis. JAMA Netw Open. 2022;5(5):e2214253. doi:10.1001/jamanetworkopen.2022.14253
3. Pepys MB. Pathogenesis, diagnosis and treatment of systemic amyloidosis. Philos Trans R Soc Lond B Biol Sci. 2001;356(1406):203-211. doi:10.1098/rstb.2000.0766
4. Cowan AJ, Skinner M, Seldin DC, et al. Amyloidosis of the gastrointestinal tract: a 13-year, single-center, referral experience. Haematologica. 2013;98(1):141-146. doi:10.3324/haematol.2012.068155
5. Lee BS, Chudasama Y, Chen AI, Lim BS, Taira MT. Colonoscopy leading to the diagnosis of AL amyloidosis in the gastrointestinal tract mimicking an acute ulcerative colitis flare. ACG Case Rep J. 2019;6(11):e00289. doi:10.14309/crj.0000000000000289
6. Zhao L, Ren G, Guo J, Chen W, Xu W, Huang X. The clinical features and outcomes of systemic light chain amyloidosis with hepatic involvement. Ann Med. 2022;54(1):1226-1232. doi:10.1080/07853890.2022.2069281
7. Rowe K, Pankow J, Nehme F, Salyers W. Gastrointestinal amyloidosis: review of the literature. Cureus. 2017;9(5):e1228. doi:10.7759/cureus.1228
8. Kyle RA, Greipp PR, O’Fallon WM. Primary systemic amyloidosis: multivariate analysis for prognostic factors in 168 cases. Blood. 1986;68(1):220-224.
Spider Bite Wound Care and Review of Traditional and Advanced Treatment Options
The costs for wound care play a significant role in total health care costs and are expected to rise dramatically. A 2018 Medicare analysis estimated chronic wound care cost $28.1 to $96.8 billion in supplies, hospitalization, and nursing care: Most costs were accrued in outpatient wound care.1 The global market for advanced wound care supplies is projected to reach $13.7 billion by 2027, and negative wound pressure therapy alone is projected to grow at a compound annual growth rate of 5% over the analysis period 2020 to 2027.2 Chronic wound care also impacts the patient physiologically, socially, and psychologically. One study compared the 5-year mortality of a patient with a diabetic foot ulcer (30.5%) as similar to those patients with cancer (31%).3 Yet the investment in cancer research far outstrips wound care research.
There is no perfect wound dressing for all chronic wounds, but there is expert consensus on interventions that facilitate wound healing. In 2021, Nuutila and Eriksson stated that wound dressings should fulfill the following criteria: protection against trauma, esthetically acceptable, painless to remove, easy to apply, protection for the wound from contamination and further trauma, a moist environment, and an optimal water vapor transmission rate.4 Balanced moisture control is considered essential for healing chronic wounds. Indeed, moisture control within the wound bed may be the most important factor in chronic wound management and healing. The body communicates through a liquid medium, and if that medium is compromised, communication and marshaling of the immune and healing responses may become inefficient.4 Too much moisture, exudate, or fluid in the wound, and the healing is slowed; too little moisture in the wound results in a compromised responses from the body’s immune system, thus delaying healing. In 1988, Dyson and colleagues demonstrated that moist wound care was superior for the inflammatory and proliferative phases of dermal repair compared with dry wound care. The results showed that 5 days after injury, 66% of the cells in the moist wound were fibroblasts and endothelial cells vs 48% of those in the dry wounds.5
The question of dry vs moist wound care has resulted in various wound dressings that produce favorable moisture balance. Moisture balance in a wound creates the ideal environment for wound healing. Sound wound care practices promote the following physiologic responses: increased probability of autolytic debridement; increased collagen synthesis; keratinocyte migration and reepithelization; decreased pain, inflammation, scarring, and necrosis;enhancement of cell-to-cell signaling; and increase in growth factors.5,6 All these processes are mediated through proper wound moisture control. In addition to proper moisture control, antibiotics added to the wound care milieu (either directly to the wound or systemically) may have a place in chronic wound care. In 2013, Junker and colleagues reported that low-dose antibiotics combined with appropriate moisture balance in wounds demonstrated less scar tissue compared with dry wound care.6
Approaches to chronic wound care are worlds apart: In developing nations the care of chronic wounds often involves traditional management with local products (eg, honey, boiled potato peels, aloe vera gel, banana leaves), whereas in developed nations, more expensive and technologically advanced products are available (eg, wound vacuum, saline wound chamber, hyperbaric oxygen therapy, antibacterial foam). Developing countries often do not have access to technologically advanced wound care products. Local products are often used by local healers, priests, and shamans. The use of these wound interventions in developing countries has produced satisfactory results. In contrast, developed countries have multiple chronic wound care products available (Table).
CASE Presentation
An athletic, healthy 60-year-old Utah National Guard member presented to the George E. Wahlen Department of Veterans Affairs Medical Center in Salt Lake City, Utah, 6 days after experiencing a spider bite. For the first 6 days, the patient applied bacitracin at home. On day 7, the patient noticed that the wound was enlarging and appeared to be fluctuant. The patient was prescribed clindamycin 300 mg 4 times daily on an outpatient basis, which was taken on days 7 to 14.
The wound’s total surface area continued to expand, and the patient returned to the Salt Lake City Veterans Hospital wound care clinic on day 17 stating that the wound was very painful and more fluctuant. The wound care nursing staff were consulted, the wound was debrided, and attempts to drain the wound resulted in minimal exudate expressed from the wound. Clindamycin was increased to 450 mg 4 times daily. However, the wound continued to enlarge and become more painful.
Discussion
Traditional Wound Care
Honey. Honey has been used as a treatment for wounds for almost 3000 years. It has antiseptic and antibacterial properties and contributes to a moist wound care environment. In 2011, Gupta and colleagues reported on the use of honey in 108 patients with burns of < 50% of the total body surface area.7 This report stated that delay in seeking medical care increased wound infection rates, contamination, time to sterilization, and healing. Compared with silver sulfadiazine cream, honey dressings improved the time to wound healing (33 days vs 18 days, respectively), decreased the time to wound sterilization (1 day vs no sterilization), and had better outcomes (37% vs 81%, respectively) with fewer hypertrophic scars and postburn contractures.7
Separate studies in 2011 and 2010 from Fukuda and colleagues and Majtan and colleagues, respectively, reported that honey eliminates pathogens from wounds, augments correct moisture balance, and elevates cytokine activity.8,9 Additional studies in 2006, 2008, and 2014 by Henriques and colleagues, Van den Berg and colleagues, and Majtan suggested that honey reduces reactive oxygen species, is responsible for direct antimicrobial effects in a healing wound, inhibits free radical production, and promotes antitumor activity, respectively.10-12 Van den Berg and colleagues suggested that buckwheat honey is the most effective honey in reducing reactive oxygen species.11
Sterile banana leaves. In medically underserved and rural areas, boiled banana leaves are used to treat burns and nonhealing wounds. In a 2015 study, Waffa and Hayah compared gauze dressings with sterile banana leaves wound dressing in patients with partial thickness burns. Topical antibiotics were added to each type of dressing. The results suggested that the banana leaf dressings were easier to remove, patients reported less pain overall, less pain with dressing changes, and demonstrated a decreased time to healing when contrasted with gauze.13 In 2003, Gore and Akolekar compared autoclaved banana leaves with boiled potato peels in the treatment of patients with partial thickness burns. The time to epithelialization, eschar formation, and skin graft healing were equal in both groups. However, banana leaves were 11 times cheaper and rated easier to prepare than boiled potato peels.14 In a study comparing petroleum gauze with sterile banana leaves, Chendake and colleagues reported that in measures of overall pain and trauma during dressing changes, patients with contused and sutured wounds on the face and neck achieved better outcomes with boiled banana leaves compared with petroleum gauze.15
Boiled potato peels. This treatment is used in rural areas of the world as an adjunct for wound care. In 2015, Manjunath and colleagues theorized that the use of boiled potato peels in patients with necrotizing fasciitis decreased the acidic environment created by the bacteria. Additionally, the study asserted that the toxic wound environment created by the bacteria was neutralized by the potassium content in the peel, and the flavonoids in the peel acted as a free radical scavenger.16 In 2011, Panda and colleagues, using povidone-iodine as a baseline control, reported that peel extract and a peel bandage of sweet potato showed an increased wound closure percentage measured by enhanced epithelialization.17 This increased epithelialization was attributed to the antioxidant effect of the peels enhancing collagen synthesis.17
In contrast, in 1996, a study by Subrahmanyam compared autoclaved potato peel bandages with honey dressings as adjuncts in burn patients with < 40% of the total body surface area affected. The author reported that 90% of the wounds treated with honey were sterile in 7 days, while infection persisted in the potato peel group after 7 days. In the same study, 100% of the wounds treated with honey were healed in 15 days vs 50% in the potato peel group.18 In 1990, Keswani and colleagues compared boiled potato peels with plain gauze as adjuncts in the treatment of burn patients and concluded that although the potato peels had no antibacterial effect, the wounds in both groups had identical bacterial species. But the wounds treated with the potato peels showed reduced desiccation, permitting the survival of skin cells, and enhanced epithelial regeneration.19
Aloe vera. First recorded by the Egyptians and Greeks, aloe vera gel has been used for centuries in many cultures for a variety of ailments, particularly burns and chronic wounds. In a 2016 wound healing study performed on rats, Oryan and colleagues demonstrated that aloe vera gel was superior to saline used as the baseline control. Aloe vera gel used in a dose-dependent fashion demonstrated increased tissue levels of collagen and glycosaminoglycans compared with controls. Aloe vera gel modulated wound inflammation, increased wound contraction, wound epithelialization, decreased scar tissue size, and increased alignment and organization of the scar tissue.20
Gauze. Iodoform gauze is a highly absorbent wound product. Sterile gauze promotes granulation and wound healing. It is well suited for wounds with minimal drainage. However, although gauze is inexpensive, it is easily overwhelmed by the moisture content in the wound, requiring frequent dressing changes (up to 3 times a day), ideally by nursing staff. The resulting increase in nursing care may actually increase the cost of wound care compared with other care modalities.
Petroleum gauze is often used in the care of acute and chronic wounds. However, petroleum-impregnated gauze has a water vapor transmission rate that needs to be remoistened every 4 hours. If the affected area is not remoistened during the exudative phase of wound healing, it may precipitate a delay in healing and increase pain and the prevalence of clinical infections compared with hydrocolloid, film, or foam dressings. Bolton suggested stopping the use of petroleum gauze as the control in studies because it does not provide a balanced and moist wound healing environment.21
Advanced Wound Treatments
Film products. Film products, including plastic food wrap, can be used as wound dressings and meet many of the necessary criteria for enhancing wound healing. These include moisture permeability, carbon dioxide, oxygen transfer, and wound protection. Transmission of moisture varies among products known as the moisture vapor transpiration rate. Film dressings have no absorptive qualities and are unsuited for highly exudative wounds.22,23 Adding polymers, antibacterial, and bioactive agents may increase the wound care properties of film dressings.22 Film dressings excel in protecting shallow nonexudative wounds, are waterproof, and help protect the wound. These products are transparent, allowing clinicians to monitor the progress of the wound without removing the covering, and allowing the dressing to remain in place longer, which decreases the repeated trauma that can occur with dressing changes. Film dressings for wounds differ from those used for IV dressings and should not be used interchangeably.23
Bioactive wound care. These solutions contribute to a moist wound-healing environment. Found naturally in brown seaweed, alginate-containing compounds were used by sailors for centuries to heal wounds. This was known in traditional medicine as the mariner’s cure. Alginate dressings are highly absorbent and can absorb up to 20 times their weight, which makes them desirable for use in highly exudative wounds. First synthesized more than 50 years ago, newer products contain bioactive compounds that prevent tissue damage, stimulate wound healing, improve cell proliferation and migration, and enhance metabolite formation.24-26
In 2018, Aderibigbe and Buyana reported that polymers in the form of hydrogels were able to absorb fluid, making them a suitable choice for minimally exudative wounds. However, in their distended state, the hydrogel subgroup of these products became unstable (perhaps making them a poor choice for extensively exudative wounds), tended to dehydrate, and often needed a secondary dressing, which could lead to wound maceration.22 Most commonly used for wounds with minimal exudate, these dressings shine when used in nominally exudative dry wounds to promote autolytic debridement and hydrate the wound that has formed an eschar.
Hydrocolloid dressings are another type of bioactive wound dressing. These dressings are composed of 2 layers: an inner hydrophilic layer and an outer vapor-permeable layer that promote a moist wound environment. Hydrocolloid dressings assist in hydrating dry eschar wounds and have slight absorbency for exudative wounds. These dressings are not designed to be changed daily and can remain in place for 3 to 6 days. In a 2008 extensive review article, Thomas compared the utility of these dressings in patients with superficial or partial thickness burns, donor sites, surgical wounds, and minor traumatic wounds with basic wound dressings. The results of the review suggested that hydrocolloid dressings conferred statistically significant advantages in measures of decreased pain, healing times (decreased in donor sites by 40%), mobility restriction, and number of dressing changes.27 Although more expensive than basic dressings, the longevity of the hydrocolloid dressing helps defray the original cost. Unfortunately, as these dressings remain in place and continue absorbing exudate, they can take on a very unpleasant odor.
A 2013 Cochrane database review comparing hydrocolloids with foams, alginate, basic wound dressing, and topical treatment found no statistical difference between hydrocolloids and basic wound dressings in patients with diabetes who have noncomplex foot ulcers.28 In 2014, Pott and colleagues suggested a slight superiority in the performance of polyurethane foam dressings over hydrocolloid dressings used in pressure ulcers in older adults.29 In a large pooled analysis in 2010, Davies compared foam to hydrocolloid dressings used in exudative wounds and reported that in 11 of 12 studies, foam dressings were superior to hydrocolloid in terms of exudate management, conformity to the wound, ease of use, decreased trauma and pain at dressing changes, and reduced odor of the wound.30
Foam dressings. These products are typically composed of silicone or polyurethane. Consisting of 2 to 3 layers with a hydrophilic surface, foams are cut to approximate the wound size and serve to wick the macerated wound products to a secondary dressing above the foam. The micropores in the foam matrix absorb exudate from the wound bed while maintaining moisture equilibrium in the wound by donating back moisture to the wound, creating an environment conducive to wound healing. Foam dressings can be combined with various antiseptics (silver, GV/MB, etc) and serve as a delivery vehicle of those products directly to the wound surface.
A 2011 review comparing 8 studies found no difference among foam products available at that time in the use for chronic wounds.31 However, newer products on the market today have produced intriguing results with chronic wounds.
In 2017, Woo and Heil observed that chronic wounds treated with foam products containing GV/MB produced significant improvement when measured at week 4 in the areas of mean wound surface area (42.5%), decrease in baseline Pressure Ulcer Scale for Healing scores (from 13.3 to 10.7), wound coverage by devitalized tissue reduced (from 52.6% to 11.4%), and mean upper and lower wound infection scores were reduced by 75%.32 Further, the researchers reported a moist wound bed was achieved at dressing changes with polyvinyl alcohol (PVA) foam dressing. This led to the presumption that adequate moisture balance and autolytic debridement were facilitated using GV/MB antibacterial PVA foam dressings.
Many foam products on the market today exert an antibacterial effect on the wound bed. Antibiotic properties of various foam dressings create a microenvironment hostile to bacterial growth.32 In addition, the antibacterial properties combined with foam products contribute to the following: autolytic debridement, absorptive qualities (which reduce the bioburden of the wound), and maintenance of moisture in the wound bed. These qualities contribute significantly to the effectiveness of foam products with antibacterial properties.32 The correct balance of moisture in the wound has been identified as a superior environment and perhaps the most important component in chronic wounds.4 Foam dressings are less painful to change, easier to change, and in this case report, contributed to faster wound healing than gauze alone. In 2016, a study by Lee and colleagues suggested that the makeup of the foam product, defined as smaller pore and uniform cell size (foam density), resulted in greater permeability and better moisture absorption and retention capacity, contributing to improved wound healing.33
In 2004, Sibbald and colleagues reported that in a 4-week study of nonhealing chronic wounds, foam wound dressing impregnated with sustained-release silver compared with foam dressing without silver resulted in a reduction in wound size (50% vs 30%, respectively), decreased fluid leakage (27% vs 44% respectively), and reduction in ulcer size measured from baseline (45% vs 25%, respectively).34
In a 2006 study, Varma and colleagues compared sterilized, saline-soaked, nonmedicated polyurethane industrial upholstery foam in nonhealing wounds used in patients with diabetes with conventional techniques using topical antibiotics, hydrocolloid or hydrogel dressings as necessary, and desloughing agents as controls. At the end of a 3-month follow-up period, 100% of the wounds of the foam group had healed compared with 29.2% of the control group. Additionally, the time to wound healing was less than half for the foam group (22.5 days) compared with the control group (52 days), and the time to granulation and epithelialization was faster in the foam group.35
In a 2012 meta-analysis, Aziz and colleagues reported that silver-impregnated dressings and topical silver were no better or worse than controls in preventing wound infection and promoting the healing of burn wounds.36 The authors also noted that the nonsilver dressing groups continuing povidone-iodine, ionic hydrogel, or silicone-coated dressing showed reduced healing time compared with the silver-containing group.36 This is intriguing because silver has long been used as a standard for the treatment of burn wounds.
Conclusions
Although there is no perfect wound dressing, some wound care products seem to perform better due to fewer adverse effects and a much lesser cost. Important aspects of wound care appear to be time from injury to wound care, cleanliness of the wound, moist wound environment, cost, ease of use, and pain of dressing changes.
Primitive wound care products perform admirably in many situations. Modern medicated foam dressings containing antibacterial properties may have beneficial properties compared with other wound care products; however, comparison studies are lacking and need broad-based, randomized, controlled trials to confirm utility. Finally, any choice of wound care product must be tailored to the particular wound and individual patient needs. More large, robust, randomized controlled trials are needed.
Acknowledgments
The authors thank Sarah Maria Paulsen and Rosemary Ellen Brown Smith for their editing, proofreading, and preparation of the manuscript.
1. Nussbaum SR, Carter MJ, Fife CE, et al. An economic evaluation of the impact, cost and Medicare policy implications of chronic non healing wounds. Value Health. 2018;21(1):27-32. doi:10.1016/j.jval.2017.07.007
3. Armstrong DG, Swerdlow MA, Armstrong AA, Conte MS, Padula WV, Bus SA. Five-year mortality and direct costs of care for people with diabetic foot complications are comparable to cancer. J Foot Ankle Res. 2020;13(1)16. doi:10.1186/s13047-020-00383-2
4. Nuutila K, Eriksson E. Moist wound healing with commonly available dressings. Adv Wound Care (New Rochelle). 2021;10(12):685-698. doi:10.1089/wound.2020.1232
5. Dyson M, Young S, Pendle CL, Webster DF, Lang SM. Comparison of the effects of moist and dry conditions on dermal repair. J Investig Dermatol. 1988;91:434-439. doi:10.1111/1523-1747.ep1247646
6. Junker JPE, Kamel RA, Caterson EJ, Eriksson E. Clinical impact upon wound healing and inflammation in moist, wet and dry environments. Adv Wound Care (New Rochelle). 2013;2(7):348-356. doi:10.1089/wound.2012.0412
7. Gupta SS, Singh O, Bhagel PS, Moses S, Shukla S, Mathur RK. Honey dressing versus silver sulfadiazine dressing for wound healing in burn patients: a retrospective study. J Cutan Aesthet Surg. 2011;4(3):183-187. doi:10.4103/0974-2077.91249
8. Fukuda M, Kobayashi K, Hirono Y, et al. Jungle honey enhances immune function and antitumor activity. Evid Based Complement Alternat Med. 2011;2011:1-8. doi:10.1093/ecam/nen086
9. Majtan J, Kumar P, Majtan T, Walls AF, Klaudiny J. Effect of honey and its major royal jelly protein 1 on cytokine and MMP-9 mRNA transcripts in human keratinocytes. Exp Dermatol. 2010;19(8):e73-e79. doi:10.1111/j.1600-0625.2009.00994.x
10. Henriques A, Jackson S, Cooper R, Burton N. Free radical production and quenching in honeys with wound healing potential. J Antimicrob Chemother. 2006;58(4):773-777. doi:10.1093/jac/dkl336
11. Van den Berg AJJ, Van den Worm E, Quarles van Ufford HC, Halkes SBA, M J Hoekstra MJ, Beukelman C J. An in vitro examination of the antioxidant and anti-inflammatory properties of buckwheat honey. J Wound Care. 2008;17(4):172-178. doi:10.12968/jowc.2008.17.4.28839
12. Majtan J. Honey: an immunomodulator in wound healing. Wound Repair Regen. 2014;22(2) 187-192. doi:10.1111/wrr.12117
13. Waffa GA, Hayah AEB. The effectiveness of using banana leaf dressing in management of partial thickness burns’ wound. IJND. 2015;5(4):22-27. doi:10.15520/ijnd.2015.vol5.iss04.70.
14. Gore MA, Akolekar D. Evaluation of banana leaf dressing for partial thickness burn wounds. Burns. 2003;29(5):487-492. doi:10.1016/s0305-4179(03)00050-0
15. Chendake S, Kale T, Manavadaria Y, Motimath AS. Evaluation of banana leaves (Musa paradisiaca) as an alternative wound dressing material compared to conventional petroleum jelly gauze dressing in contused, lacerated and sutured wounds over the head, neck and face region. Cureus. 2021;13(10):1-9. doi:10.7759/cureus.18552
16. Manjunath KS, Bhandage S, Kamat S. ‘Potato peel’ dressing: a novel adjunctive in the management of necrotizing fasciitis. J Maxillofacial Oral Surg. 2015;14(suppl 1):s352-s354. doi:10.1007/s12663-013-0590-8
17. Panda V, Sonkamble M, Patil S. Wound healing activity of Ipomoea batatas tubers (sweet potato). FFHDJ. 2011;1(10):403-415.
18. Subrahmanyam M. Honey dressing versus boiled potato peel in the treatment of burns: a prospective randomized study. Burns. 1996;22(6):491-493. doi:10.1016/0305-4179(96)00007-1.
19. Keswani M H, Vartak AM, Patil A, Davies JW. Histological and bacteriological studies of burn wounds treated with boiled potato peel dressings. Burns. 1990;16(2):137-143. doi:10.1016/0305-4179(90)90175-v
20. Oryan A, Mohammadalipour A, Moshiri A, MR Tabandeh. Topical application of aloe vera accelerated wound healing, modeling, and remodeling, an experimental study. Ann Plast Surg. 2016;77(1)37-46. doi:10.1097/SAP.0000000000000239
21. Bolton L. Evidence corner: April 2007. Wounds. 2007;19(4):A16-A22.
22. Aderibigbe BA, Buyana B. Alginate in wound dressings. Pharmaceutics. 2018;10(2):42. doi.10.3390/pharmaceutics10020042
23. Fletcher J. Using film dressings. Nurs Times. 2003;99(25):57.
24. Ranahewa TH, Premarathna AD, Wijesundara RMKK, Wijewardana V, Jayasooriya AP, Rajapakse RPVJ. Biochemical composition and anticancer effect of different seaweed species (in-vitro and in-vivo studies). Sustainable Marine Structures. 2019;1(2):5-11. doi:10.36956/sms.v1i2.94
25. El Gamal AA. Biological importance of marine algae. Saudi Pharmaceutical J. 2010;18(1):1-25. doi:10.1016/j.jsps.2009.12.001
26. Premarathna AD, Ranahewa TH, Wijesekera RRMKK, et al. Wound healing properties of aqueous extracts of Sargassum Illicifolium: an in vitro assay. Wound Medicine. 2019;24(1):1-7. doi:10.1016/j.wndm.2018.11.001
27. Thomas S. Hydrocolloid dressings in the management of acute wounds: a review of the literature. Int Wound J. 2008;5(5):602-613. doi:10.1111/j.1742-481X.2008.00541.x
28. Dumville JC, Deshpande S, O’Mera K, et al. Hydrocolloid dressing for healing diabetic foot ulcers. Cochrane Database Syst Rev. 2013(8): CD009099. doi:10.1002/14651858.CD009099.pub3
29. Pott FS, Meier MJ, Stocco JGD, Crozeta K, Dayane Ribas J. The effectiveness of hydrocolloid dressings versus other dressings in the healing of pressure ulcers in adults and older adults: a systematic review and meta-analysis. Rev Lat-Am Enfermagem. 2014;22(3):511-520. doi:10.1590/0104-1169.3480.2445
30. Davies P, Rippon M. Comparison of foam and hydrocolloid dressings in the management of wounds: a review of the published literature. Accessed May 17, 2023. http://www.worldwidewounds.com/2010/July/DaviesRippon/DaviesRippon.html
31. Bianchi J, Gray D, Timmons J. Meaume S. Do all foam dressings have the same efficacy in the treatment of chronic wounds? Wounds UK. 2011;7(1):62-67.

33. Lee SM, Park IK, Kim HJ, et al. Physical, morphological, and wound healing properties of a polyurethane foam-film dressing. Biomaterials Res. 2016;20(15):1-11. doi:10.1186/s40824-016-0063-5
34. Sibbald RG, Meaume S, Kirsner RS, et al. Review of the clinical RCT evidence and cost-effectiveness data of a sustained-release silver foam dressing in the healing of critically colonized wounds. December 2005. Updated January 2006. Accessed May 18, 2023. http://www.worldwidewounds.com/2005/december/Sibbald/Silver-Foam-Dressings-Colonised-Wounds.html
35. Varma AK, Kumar H, Kesav Raiesh. Efficacy of polyurethane foam dressing in debrided diabetic lower limb wounds. Wounds. 2006;18(10):300-306.
36. Aziz Z, Abu SF, Chong NJ. A systematic review of silver-containing dressings and topical silver agents (used with dressings) for burn wounds. Burns. 2012;38(3):307-318. doi:10.1016/j.burns.2011.09.020
37. Agarwal P, Kukrele R, Sharma D. Vacuum assisted closure (VAC)/negative pressure wound therapy (NPWT) for difficult wounds: a review. J Clin Orthop Trauma. 2019;10(5):845-848. doi:10.1016/j.jcot.2019.06.015
The costs for wound care play a significant role in total health care costs and are expected to rise dramatically. A 2018 Medicare analysis estimated chronic wound care cost $28.1 to $96.8 billion in supplies, hospitalization, and nursing care: Most costs were accrued in outpatient wound care.1 The global market for advanced wound care supplies is projected to reach $13.7 billion by 2027, and negative wound pressure therapy alone is projected to grow at a compound annual growth rate of 5% over the analysis period 2020 to 2027.2 Chronic wound care also impacts the patient physiologically, socially, and psychologically. One study compared the 5-year mortality of a patient with a diabetic foot ulcer (30.5%) as similar to those patients with cancer (31%).3 Yet the investment in cancer research far outstrips wound care research.
There is no perfect wound dressing for all chronic wounds, but there is expert consensus on interventions that facilitate wound healing. In 2021, Nuutila and Eriksson stated that wound dressings should fulfill the following criteria: protection against trauma, esthetically acceptable, painless to remove, easy to apply, protection for the wound from contamination and further trauma, a moist environment, and an optimal water vapor transmission rate.4 Balanced moisture control is considered essential for healing chronic wounds. Indeed, moisture control within the wound bed may be the most important factor in chronic wound management and healing. The body communicates through a liquid medium, and if that medium is compromised, communication and marshaling of the immune and healing responses may become inefficient.4 Too much moisture, exudate, or fluid in the wound, and the healing is slowed; too little moisture in the wound results in a compromised responses from the body’s immune system, thus delaying healing. In 1988, Dyson and colleagues demonstrated that moist wound care was superior for the inflammatory and proliferative phases of dermal repair compared with dry wound care. The results showed that 5 days after injury, 66% of the cells in the moist wound were fibroblasts and endothelial cells vs 48% of those in the dry wounds.5
The question of dry vs moist wound care has resulted in various wound dressings that produce favorable moisture balance. Moisture balance in a wound creates the ideal environment for wound healing. Sound wound care practices promote the following physiologic responses: increased probability of autolytic debridement; increased collagen synthesis; keratinocyte migration and reepithelization; decreased pain, inflammation, scarring, and necrosis;enhancement of cell-to-cell signaling; and increase in growth factors.5,6 All these processes are mediated through proper wound moisture control. In addition to proper moisture control, antibiotics added to the wound care milieu (either directly to the wound or systemically) may have a place in chronic wound care. In 2013, Junker and colleagues reported that low-dose antibiotics combined with appropriate moisture balance in wounds demonstrated less scar tissue compared with dry wound care.6
Approaches to chronic wound care are worlds apart: In developing nations the care of chronic wounds often involves traditional management with local products (eg, honey, boiled potato peels, aloe vera gel, banana leaves), whereas in developed nations, more expensive and technologically advanced products are available (eg, wound vacuum, saline wound chamber, hyperbaric oxygen therapy, antibacterial foam). Developing countries often do not have access to technologically advanced wound care products. Local products are often used by local healers, priests, and shamans. The use of these wound interventions in developing countries has produced satisfactory results. In contrast, developed countries have multiple chronic wound care products available (Table).
CASE Presentation
An athletic, healthy 60-year-old Utah National Guard member presented to the George E. Wahlen Department of Veterans Affairs Medical Center in Salt Lake City, Utah, 6 days after experiencing a spider bite. For the first 6 days, the patient applied bacitracin at home. On day 7, the patient noticed that the wound was enlarging and appeared to be fluctuant. The patient was prescribed clindamycin 300 mg 4 times daily on an outpatient basis, which was taken on days 7 to 14.
The wound’s total surface area continued to expand, and the patient returned to the Salt Lake City Veterans Hospital wound care clinic on day 17 stating that the wound was very painful and more fluctuant. The wound care nursing staff were consulted, the wound was debrided, and attempts to drain the wound resulted in minimal exudate expressed from the wound. Clindamycin was increased to 450 mg 4 times daily. However, the wound continued to enlarge and become more painful.
Discussion
Traditional Wound Care
Honey. Honey has been used as a treatment for wounds for almost 3000 years. It has antiseptic and antibacterial properties and contributes to a moist wound care environment. In 2011, Gupta and colleagues reported on the use of honey in 108 patients with burns of < 50% of the total body surface area.7 This report stated that delay in seeking medical care increased wound infection rates, contamination, time to sterilization, and healing. Compared with silver sulfadiazine cream, honey dressings improved the time to wound healing (33 days vs 18 days, respectively), decreased the time to wound sterilization (1 day vs no sterilization), and had better outcomes (37% vs 81%, respectively) with fewer hypertrophic scars and postburn contractures.7
Separate studies in 2011 and 2010 from Fukuda and colleagues and Majtan and colleagues, respectively, reported that honey eliminates pathogens from wounds, augments correct moisture balance, and elevates cytokine activity.8,9 Additional studies in 2006, 2008, and 2014 by Henriques and colleagues, Van den Berg and colleagues, and Majtan suggested that honey reduces reactive oxygen species, is responsible for direct antimicrobial effects in a healing wound, inhibits free radical production, and promotes antitumor activity, respectively.10-12 Van den Berg and colleagues suggested that buckwheat honey is the most effective honey in reducing reactive oxygen species.11
Sterile banana leaves. In medically underserved and rural areas, boiled banana leaves are used to treat burns and nonhealing wounds. In a 2015 study, Waffa and Hayah compared gauze dressings with sterile banana leaves wound dressing in patients with partial thickness burns. Topical antibiotics were added to each type of dressing. The results suggested that the banana leaf dressings were easier to remove, patients reported less pain overall, less pain with dressing changes, and demonstrated a decreased time to healing when contrasted with gauze.13 In 2003, Gore and Akolekar compared autoclaved banana leaves with boiled potato peels in the treatment of patients with partial thickness burns. The time to epithelialization, eschar formation, and skin graft healing were equal in both groups. However, banana leaves were 11 times cheaper and rated easier to prepare than boiled potato peels.14 In a study comparing petroleum gauze with sterile banana leaves, Chendake and colleagues reported that in measures of overall pain and trauma during dressing changes, patients with contused and sutured wounds on the face and neck achieved better outcomes with boiled banana leaves compared with petroleum gauze.15
Boiled potato peels. This treatment is used in rural areas of the world as an adjunct for wound care. In 2015, Manjunath and colleagues theorized that the use of boiled potato peels in patients with necrotizing fasciitis decreased the acidic environment created by the bacteria. Additionally, the study asserted that the toxic wound environment created by the bacteria was neutralized by the potassium content in the peel, and the flavonoids in the peel acted as a free radical scavenger.16 In 2011, Panda and colleagues, using povidone-iodine as a baseline control, reported that peel extract and a peel bandage of sweet potato showed an increased wound closure percentage measured by enhanced epithelialization.17 This increased epithelialization was attributed to the antioxidant effect of the peels enhancing collagen synthesis.17
In contrast, in 1996, a study by Subrahmanyam compared autoclaved potato peel bandages with honey dressings as adjuncts in burn patients with < 40% of the total body surface area affected. The author reported that 90% of the wounds treated with honey were sterile in 7 days, while infection persisted in the potato peel group after 7 days. In the same study, 100% of the wounds treated with honey were healed in 15 days vs 50% in the potato peel group.18 In 1990, Keswani and colleagues compared boiled potato peels with plain gauze as adjuncts in the treatment of burn patients and concluded that although the potato peels had no antibacterial effect, the wounds in both groups had identical bacterial species. But the wounds treated with the potato peels showed reduced desiccation, permitting the survival of skin cells, and enhanced epithelial regeneration.19
Aloe vera. First recorded by the Egyptians and Greeks, aloe vera gel has been used for centuries in many cultures for a variety of ailments, particularly burns and chronic wounds. In a 2016 wound healing study performed on rats, Oryan and colleagues demonstrated that aloe vera gel was superior to saline used as the baseline control. Aloe vera gel used in a dose-dependent fashion demonstrated increased tissue levels of collagen and glycosaminoglycans compared with controls. Aloe vera gel modulated wound inflammation, increased wound contraction, wound epithelialization, decreased scar tissue size, and increased alignment and organization of the scar tissue.20
Gauze. Iodoform gauze is a highly absorbent wound product. Sterile gauze promotes granulation and wound healing. It is well suited for wounds with minimal drainage. However, although gauze is inexpensive, it is easily overwhelmed by the moisture content in the wound, requiring frequent dressing changes (up to 3 times a day), ideally by nursing staff. The resulting increase in nursing care may actually increase the cost of wound care compared with other care modalities.
Petroleum gauze is often used in the care of acute and chronic wounds. However, petroleum-impregnated gauze has a water vapor transmission rate that needs to be remoistened every 4 hours. If the affected area is not remoistened during the exudative phase of wound healing, it may precipitate a delay in healing and increase pain and the prevalence of clinical infections compared with hydrocolloid, film, or foam dressings. Bolton suggested stopping the use of petroleum gauze as the control in studies because it does not provide a balanced and moist wound healing environment.21
Advanced Wound Treatments
Film products. Film products, including plastic food wrap, can be used as wound dressings and meet many of the necessary criteria for enhancing wound healing. These include moisture permeability, carbon dioxide, oxygen transfer, and wound protection. Transmission of moisture varies among products known as the moisture vapor transpiration rate. Film dressings have no absorptive qualities and are unsuited for highly exudative wounds.22,23 Adding polymers, antibacterial, and bioactive agents may increase the wound care properties of film dressings.22 Film dressings excel in protecting shallow nonexudative wounds, are waterproof, and help protect the wound. These products are transparent, allowing clinicians to monitor the progress of the wound without removing the covering, and allowing the dressing to remain in place longer, which decreases the repeated trauma that can occur with dressing changes. Film dressings for wounds differ from those used for IV dressings and should not be used interchangeably.23
Bioactive wound care. These solutions contribute to a moist wound-healing environment. Found naturally in brown seaweed, alginate-containing compounds were used by sailors for centuries to heal wounds. This was known in traditional medicine as the mariner’s cure. Alginate dressings are highly absorbent and can absorb up to 20 times their weight, which makes them desirable for use in highly exudative wounds. First synthesized more than 50 years ago, newer products contain bioactive compounds that prevent tissue damage, stimulate wound healing, improve cell proliferation and migration, and enhance metabolite formation.24-26
In 2018, Aderibigbe and Buyana reported that polymers in the form of hydrogels were able to absorb fluid, making them a suitable choice for minimally exudative wounds. However, in their distended state, the hydrogel subgroup of these products became unstable (perhaps making them a poor choice for extensively exudative wounds), tended to dehydrate, and often needed a secondary dressing, which could lead to wound maceration.22 Most commonly used for wounds with minimal exudate, these dressings shine when used in nominally exudative dry wounds to promote autolytic debridement and hydrate the wound that has formed an eschar.
Hydrocolloid dressings are another type of bioactive wound dressing. These dressings are composed of 2 layers: an inner hydrophilic layer and an outer vapor-permeable layer that promote a moist wound environment. Hydrocolloid dressings assist in hydrating dry eschar wounds and have slight absorbency for exudative wounds. These dressings are not designed to be changed daily and can remain in place for 3 to 6 days. In a 2008 extensive review article, Thomas compared the utility of these dressings in patients with superficial or partial thickness burns, donor sites, surgical wounds, and minor traumatic wounds with basic wound dressings. The results of the review suggested that hydrocolloid dressings conferred statistically significant advantages in measures of decreased pain, healing times (decreased in donor sites by 40%), mobility restriction, and number of dressing changes.27 Although more expensive than basic dressings, the longevity of the hydrocolloid dressing helps defray the original cost. Unfortunately, as these dressings remain in place and continue absorbing exudate, they can take on a very unpleasant odor.
A 2013 Cochrane database review comparing hydrocolloids with foams, alginate, basic wound dressing, and topical treatment found no statistical difference between hydrocolloids and basic wound dressings in patients with diabetes who have noncomplex foot ulcers.28 In 2014, Pott and colleagues suggested a slight superiority in the performance of polyurethane foam dressings over hydrocolloid dressings used in pressure ulcers in older adults.29 In a large pooled analysis in 2010, Davies compared foam to hydrocolloid dressings used in exudative wounds and reported that in 11 of 12 studies, foam dressings were superior to hydrocolloid in terms of exudate management, conformity to the wound, ease of use, decreased trauma and pain at dressing changes, and reduced odor of the wound.30
Foam dressings. These products are typically composed of silicone or polyurethane. Consisting of 2 to 3 layers with a hydrophilic surface, foams are cut to approximate the wound size and serve to wick the macerated wound products to a secondary dressing above the foam. The micropores in the foam matrix absorb exudate from the wound bed while maintaining moisture equilibrium in the wound by donating back moisture to the wound, creating an environment conducive to wound healing. Foam dressings can be combined with various antiseptics (silver, GV/MB, etc) and serve as a delivery vehicle of those products directly to the wound surface.
A 2011 review comparing 8 studies found no difference among foam products available at that time in the use for chronic wounds.31 However, newer products on the market today have produced intriguing results with chronic wounds.
In 2017, Woo and Heil observed that chronic wounds treated with foam products containing GV/MB produced significant improvement when measured at week 4 in the areas of mean wound surface area (42.5%), decrease in baseline Pressure Ulcer Scale for Healing scores (from 13.3 to 10.7), wound coverage by devitalized tissue reduced (from 52.6% to 11.4%), and mean upper and lower wound infection scores were reduced by 75%.32 Further, the researchers reported a moist wound bed was achieved at dressing changes with polyvinyl alcohol (PVA) foam dressing. This led to the presumption that adequate moisture balance and autolytic debridement were facilitated using GV/MB antibacterial PVA foam dressings.
Many foam products on the market today exert an antibacterial effect on the wound bed. Antibiotic properties of various foam dressings create a microenvironment hostile to bacterial growth.32 In addition, the antibacterial properties combined with foam products contribute to the following: autolytic debridement, absorptive qualities (which reduce the bioburden of the wound), and maintenance of moisture in the wound bed. These qualities contribute significantly to the effectiveness of foam products with antibacterial properties.32 The correct balance of moisture in the wound has been identified as a superior environment and perhaps the most important component in chronic wounds.4 Foam dressings are less painful to change, easier to change, and in this case report, contributed to faster wound healing than gauze alone. In 2016, a study by Lee and colleagues suggested that the makeup of the foam product, defined as smaller pore and uniform cell size (foam density), resulted in greater permeability and better moisture absorption and retention capacity, contributing to improved wound healing.33
In 2004, Sibbald and colleagues reported that in a 4-week study of nonhealing chronic wounds, foam wound dressing impregnated with sustained-release silver compared with foam dressing without silver resulted in a reduction in wound size (50% vs 30%, respectively), decreased fluid leakage (27% vs 44% respectively), and reduction in ulcer size measured from baseline (45% vs 25%, respectively).34
In a 2006 study, Varma and colleagues compared sterilized, saline-soaked, nonmedicated polyurethane industrial upholstery foam in nonhealing wounds used in patients with diabetes with conventional techniques using topical antibiotics, hydrocolloid or hydrogel dressings as necessary, and desloughing agents as controls. At the end of a 3-month follow-up period, 100% of the wounds of the foam group had healed compared with 29.2% of the control group. Additionally, the time to wound healing was less than half for the foam group (22.5 days) compared with the control group (52 days), and the time to granulation and epithelialization was faster in the foam group.35
In a 2012 meta-analysis, Aziz and colleagues reported that silver-impregnated dressings and topical silver were no better or worse than controls in preventing wound infection and promoting the healing of burn wounds.36 The authors also noted that the nonsilver dressing groups continuing povidone-iodine, ionic hydrogel, or silicone-coated dressing showed reduced healing time compared with the silver-containing group.36 This is intriguing because silver has long been used as a standard for the treatment of burn wounds.
Conclusions
Although there is no perfect wound dressing, some wound care products seem to perform better due to fewer adverse effects and a much lesser cost. Important aspects of wound care appear to be time from injury to wound care, cleanliness of the wound, moist wound environment, cost, ease of use, and pain of dressing changes.
Primitive wound care products perform admirably in many situations. Modern medicated foam dressings containing antibacterial properties may have beneficial properties compared with other wound care products; however, comparison studies are lacking and need broad-based, randomized, controlled trials to confirm utility. Finally, any choice of wound care product must be tailored to the particular wound and individual patient needs. More large, robust, randomized controlled trials are needed.
Acknowledgments
The authors thank Sarah Maria Paulsen and Rosemary Ellen Brown Smith for their editing, proofreading, and preparation of the manuscript.
The costs for wound care play a significant role in total health care costs and are expected to rise dramatically. A 2018 Medicare analysis estimated chronic wound care cost $28.1 to $96.8 billion in supplies, hospitalization, and nursing care: Most costs were accrued in outpatient wound care.1 The global market for advanced wound care supplies is projected to reach $13.7 billion by 2027, and negative wound pressure therapy alone is projected to grow at a compound annual growth rate of 5% over the analysis period 2020 to 2027.2 Chronic wound care also impacts the patient physiologically, socially, and psychologically. One study compared the 5-year mortality of a patient with a diabetic foot ulcer (30.5%) as similar to those patients with cancer (31%).3 Yet the investment in cancer research far outstrips wound care research.
There is no perfect wound dressing for all chronic wounds, but there is expert consensus on interventions that facilitate wound healing. In 2021, Nuutila and Eriksson stated that wound dressings should fulfill the following criteria: protection against trauma, esthetically acceptable, painless to remove, easy to apply, protection for the wound from contamination and further trauma, a moist environment, and an optimal water vapor transmission rate.4 Balanced moisture control is considered essential for healing chronic wounds. Indeed, moisture control within the wound bed may be the most important factor in chronic wound management and healing. The body communicates through a liquid medium, and if that medium is compromised, communication and marshaling of the immune and healing responses may become inefficient.4 Too much moisture, exudate, or fluid in the wound, and the healing is slowed; too little moisture in the wound results in a compromised responses from the body’s immune system, thus delaying healing. In 1988, Dyson and colleagues demonstrated that moist wound care was superior for the inflammatory and proliferative phases of dermal repair compared with dry wound care. The results showed that 5 days after injury, 66% of the cells in the moist wound were fibroblasts and endothelial cells vs 48% of those in the dry wounds.5
The question of dry vs moist wound care has resulted in various wound dressings that produce favorable moisture balance. Moisture balance in a wound creates the ideal environment for wound healing. Sound wound care practices promote the following physiologic responses: increased probability of autolytic debridement; increased collagen synthesis; keratinocyte migration and reepithelization; decreased pain, inflammation, scarring, and necrosis;enhancement of cell-to-cell signaling; and increase in growth factors.5,6 All these processes are mediated through proper wound moisture control. In addition to proper moisture control, antibiotics added to the wound care milieu (either directly to the wound or systemically) may have a place in chronic wound care. In 2013, Junker and colleagues reported that low-dose antibiotics combined with appropriate moisture balance in wounds demonstrated less scar tissue compared with dry wound care.6
Approaches to chronic wound care are worlds apart: In developing nations the care of chronic wounds often involves traditional management with local products (eg, honey, boiled potato peels, aloe vera gel, banana leaves), whereas in developed nations, more expensive and technologically advanced products are available (eg, wound vacuum, saline wound chamber, hyperbaric oxygen therapy, antibacterial foam). Developing countries often do not have access to technologically advanced wound care products. Local products are often used by local healers, priests, and shamans. The use of these wound interventions in developing countries has produced satisfactory results. In contrast, developed countries have multiple chronic wound care products available (Table).
CASE Presentation
An athletic, healthy 60-year-old Utah National Guard member presented to the George E. Wahlen Department of Veterans Affairs Medical Center in Salt Lake City, Utah, 6 days after experiencing a spider bite. For the first 6 days, the patient applied bacitracin at home. On day 7, the patient noticed that the wound was enlarging and appeared to be fluctuant. The patient was prescribed clindamycin 300 mg 4 times daily on an outpatient basis, which was taken on days 7 to 14.
The wound’s total surface area continued to expand, and the patient returned to the Salt Lake City Veterans Hospital wound care clinic on day 17 stating that the wound was very painful and more fluctuant. The wound care nursing staff were consulted, the wound was debrided, and attempts to drain the wound resulted in minimal exudate expressed from the wound. Clindamycin was increased to 450 mg 4 times daily. However, the wound continued to enlarge and become more painful.
Discussion
Traditional Wound Care
Honey. Honey has been used as a treatment for wounds for almost 3000 years. It has antiseptic and antibacterial properties and contributes to a moist wound care environment. In 2011, Gupta and colleagues reported on the use of honey in 108 patients with burns of < 50% of the total body surface area.7 This report stated that delay in seeking medical care increased wound infection rates, contamination, time to sterilization, and healing. Compared with silver sulfadiazine cream, honey dressings improved the time to wound healing (33 days vs 18 days, respectively), decreased the time to wound sterilization (1 day vs no sterilization), and had better outcomes (37% vs 81%, respectively) with fewer hypertrophic scars and postburn contractures.7
Separate studies in 2011 and 2010 from Fukuda and colleagues and Majtan and colleagues, respectively, reported that honey eliminates pathogens from wounds, augments correct moisture balance, and elevates cytokine activity.8,9 Additional studies in 2006, 2008, and 2014 by Henriques and colleagues, Van den Berg and colleagues, and Majtan suggested that honey reduces reactive oxygen species, is responsible for direct antimicrobial effects in a healing wound, inhibits free radical production, and promotes antitumor activity, respectively.10-12 Van den Berg and colleagues suggested that buckwheat honey is the most effective honey in reducing reactive oxygen species.11
Sterile banana leaves. In medically underserved and rural areas, boiled banana leaves are used to treat burns and nonhealing wounds. In a 2015 study, Waffa and Hayah compared gauze dressings with sterile banana leaves wound dressing in patients with partial thickness burns. Topical antibiotics were added to each type of dressing. The results suggested that the banana leaf dressings were easier to remove, patients reported less pain overall, less pain with dressing changes, and demonstrated a decreased time to healing when contrasted with gauze.13 In 2003, Gore and Akolekar compared autoclaved banana leaves with boiled potato peels in the treatment of patients with partial thickness burns. The time to epithelialization, eschar formation, and skin graft healing were equal in both groups. However, banana leaves were 11 times cheaper and rated easier to prepare than boiled potato peels.14 In a study comparing petroleum gauze with sterile banana leaves, Chendake and colleagues reported that in measures of overall pain and trauma during dressing changes, patients with contused and sutured wounds on the face and neck achieved better outcomes with boiled banana leaves compared with petroleum gauze.15
Boiled potato peels. This treatment is used in rural areas of the world as an adjunct for wound care. In 2015, Manjunath and colleagues theorized that the use of boiled potato peels in patients with necrotizing fasciitis decreased the acidic environment created by the bacteria. Additionally, the study asserted that the toxic wound environment created by the bacteria was neutralized by the potassium content in the peel, and the flavonoids in the peel acted as a free radical scavenger.16 In 2011, Panda and colleagues, using povidone-iodine as a baseline control, reported that peel extract and a peel bandage of sweet potato showed an increased wound closure percentage measured by enhanced epithelialization.17 This increased epithelialization was attributed to the antioxidant effect of the peels enhancing collagen synthesis.17
In contrast, in 1996, a study by Subrahmanyam compared autoclaved potato peel bandages with honey dressings as adjuncts in burn patients with < 40% of the total body surface area affected. The author reported that 90% of the wounds treated with honey were sterile in 7 days, while infection persisted in the potato peel group after 7 days. In the same study, 100% of the wounds treated with honey were healed in 15 days vs 50% in the potato peel group.18 In 1990, Keswani and colleagues compared boiled potato peels with plain gauze as adjuncts in the treatment of burn patients and concluded that although the potato peels had no antibacterial effect, the wounds in both groups had identical bacterial species. But the wounds treated with the potato peels showed reduced desiccation, permitting the survival of skin cells, and enhanced epithelial regeneration.19
Aloe vera. First recorded by the Egyptians and Greeks, aloe vera gel has been used for centuries in many cultures for a variety of ailments, particularly burns and chronic wounds. In a 2016 wound healing study performed on rats, Oryan and colleagues demonstrated that aloe vera gel was superior to saline used as the baseline control. Aloe vera gel used in a dose-dependent fashion demonstrated increased tissue levels of collagen and glycosaminoglycans compared with controls. Aloe vera gel modulated wound inflammation, increased wound contraction, wound epithelialization, decreased scar tissue size, and increased alignment and organization of the scar tissue.20
Gauze. Iodoform gauze is a highly absorbent wound product. Sterile gauze promotes granulation and wound healing. It is well suited for wounds with minimal drainage. However, although gauze is inexpensive, it is easily overwhelmed by the moisture content in the wound, requiring frequent dressing changes (up to 3 times a day), ideally by nursing staff. The resulting increase in nursing care may actually increase the cost of wound care compared with other care modalities.
Petroleum gauze is often used in the care of acute and chronic wounds. However, petroleum-impregnated gauze has a water vapor transmission rate that needs to be remoistened every 4 hours. If the affected area is not remoistened during the exudative phase of wound healing, it may precipitate a delay in healing and increase pain and the prevalence of clinical infections compared with hydrocolloid, film, or foam dressings. Bolton suggested stopping the use of petroleum gauze as the control in studies because it does not provide a balanced and moist wound healing environment.21
Advanced Wound Treatments
Film products. Film products, including plastic food wrap, can be used as wound dressings and meet many of the necessary criteria for enhancing wound healing. These include moisture permeability, carbon dioxide, oxygen transfer, and wound protection. Transmission of moisture varies among products known as the moisture vapor transpiration rate. Film dressings have no absorptive qualities and are unsuited for highly exudative wounds.22,23 Adding polymers, antibacterial, and bioactive agents may increase the wound care properties of film dressings.22 Film dressings excel in protecting shallow nonexudative wounds, are waterproof, and help protect the wound. These products are transparent, allowing clinicians to monitor the progress of the wound without removing the covering, and allowing the dressing to remain in place longer, which decreases the repeated trauma that can occur with dressing changes. Film dressings for wounds differ from those used for IV dressings and should not be used interchangeably.23
Bioactive wound care. These solutions contribute to a moist wound-healing environment. Found naturally in brown seaweed, alginate-containing compounds were used by sailors for centuries to heal wounds. This was known in traditional medicine as the mariner’s cure. Alginate dressings are highly absorbent and can absorb up to 20 times their weight, which makes them desirable for use in highly exudative wounds. First synthesized more than 50 years ago, newer products contain bioactive compounds that prevent tissue damage, stimulate wound healing, improve cell proliferation and migration, and enhance metabolite formation.24-26
In 2018, Aderibigbe and Buyana reported that polymers in the form of hydrogels were able to absorb fluid, making them a suitable choice for minimally exudative wounds. However, in their distended state, the hydrogel subgroup of these products became unstable (perhaps making them a poor choice for extensively exudative wounds), tended to dehydrate, and often needed a secondary dressing, which could lead to wound maceration.22 Most commonly used for wounds with minimal exudate, these dressings shine when used in nominally exudative dry wounds to promote autolytic debridement and hydrate the wound that has formed an eschar.
Hydrocolloid dressings are another type of bioactive wound dressing. These dressings are composed of 2 layers: an inner hydrophilic layer and an outer vapor-permeable layer that promote a moist wound environment. Hydrocolloid dressings assist in hydrating dry eschar wounds and have slight absorbency for exudative wounds. These dressings are not designed to be changed daily and can remain in place for 3 to 6 days. In a 2008 extensive review article, Thomas compared the utility of these dressings in patients with superficial or partial thickness burns, donor sites, surgical wounds, and minor traumatic wounds with basic wound dressings. The results of the review suggested that hydrocolloid dressings conferred statistically significant advantages in measures of decreased pain, healing times (decreased in donor sites by 40%), mobility restriction, and number of dressing changes.27 Although more expensive than basic dressings, the longevity of the hydrocolloid dressing helps defray the original cost. Unfortunately, as these dressings remain in place and continue absorbing exudate, they can take on a very unpleasant odor.
A 2013 Cochrane database review comparing hydrocolloids with foams, alginate, basic wound dressing, and topical treatment found no statistical difference between hydrocolloids and basic wound dressings in patients with diabetes who have noncomplex foot ulcers.28 In 2014, Pott and colleagues suggested a slight superiority in the performance of polyurethane foam dressings over hydrocolloid dressings used in pressure ulcers in older adults.29 In a large pooled analysis in 2010, Davies compared foam to hydrocolloid dressings used in exudative wounds and reported that in 11 of 12 studies, foam dressings were superior to hydrocolloid in terms of exudate management, conformity to the wound, ease of use, decreased trauma and pain at dressing changes, and reduced odor of the wound.30
Foam dressings. These products are typically composed of silicone or polyurethane. Consisting of 2 to 3 layers with a hydrophilic surface, foams are cut to approximate the wound size and serve to wick the macerated wound products to a secondary dressing above the foam. The micropores in the foam matrix absorb exudate from the wound bed while maintaining moisture equilibrium in the wound by donating back moisture to the wound, creating an environment conducive to wound healing. Foam dressings can be combined with various antiseptics (silver, GV/MB, etc) and serve as a delivery vehicle of those products directly to the wound surface.
A 2011 review comparing 8 studies found no difference among foam products available at that time in the use for chronic wounds.31 However, newer products on the market today have produced intriguing results with chronic wounds.
In 2017, Woo and Heil observed that chronic wounds treated with foam products containing GV/MB produced significant improvement when measured at week 4 in the areas of mean wound surface area (42.5%), decrease in baseline Pressure Ulcer Scale for Healing scores (from 13.3 to 10.7), wound coverage by devitalized tissue reduced (from 52.6% to 11.4%), and mean upper and lower wound infection scores were reduced by 75%.32 Further, the researchers reported a moist wound bed was achieved at dressing changes with polyvinyl alcohol (PVA) foam dressing. This led to the presumption that adequate moisture balance and autolytic debridement were facilitated using GV/MB antibacterial PVA foam dressings.
Many foam products on the market today exert an antibacterial effect on the wound bed. Antibiotic properties of various foam dressings create a microenvironment hostile to bacterial growth.32 In addition, the antibacterial properties combined with foam products contribute to the following: autolytic debridement, absorptive qualities (which reduce the bioburden of the wound), and maintenance of moisture in the wound bed. These qualities contribute significantly to the effectiveness of foam products with antibacterial properties.32 The correct balance of moisture in the wound has been identified as a superior environment and perhaps the most important component in chronic wounds.4 Foam dressings are less painful to change, easier to change, and in this case report, contributed to faster wound healing than gauze alone. In 2016, a study by Lee and colleagues suggested that the makeup of the foam product, defined as smaller pore and uniform cell size (foam density), resulted in greater permeability and better moisture absorption and retention capacity, contributing to improved wound healing.33
In 2004, Sibbald and colleagues reported that in a 4-week study of nonhealing chronic wounds, foam wound dressing impregnated with sustained-release silver compared with foam dressing without silver resulted in a reduction in wound size (50% vs 30%, respectively), decreased fluid leakage (27% vs 44% respectively), and reduction in ulcer size measured from baseline (45% vs 25%, respectively).34
In a 2006 study, Varma and colleagues compared sterilized, saline-soaked, nonmedicated polyurethane industrial upholstery foam in nonhealing wounds used in patients with diabetes with conventional techniques using topical antibiotics, hydrocolloid or hydrogel dressings as necessary, and desloughing agents as controls. At the end of a 3-month follow-up period, 100% of the wounds of the foam group had healed compared with 29.2% of the control group. Additionally, the time to wound healing was less than half for the foam group (22.5 days) compared with the control group (52 days), and the time to granulation and epithelialization was faster in the foam group.35
In a 2012 meta-analysis, Aziz and colleagues reported that silver-impregnated dressings and topical silver were no better or worse than controls in preventing wound infection and promoting the healing of burn wounds.36 The authors also noted that the nonsilver dressing groups continuing povidone-iodine, ionic hydrogel, or silicone-coated dressing showed reduced healing time compared with the silver-containing group.36 This is intriguing because silver has long been used as a standard for the treatment of burn wounds.
Conclusions
Although there is no perfect wound dressing, some wound care products seem to perform better due to fewer adverse effects and a much lesser cost. Important aspects of wound care appear to be time from injury to wound care, cleanliness of the wound, moist wound environment, cost, ease of use, and pain of dressing changes.
Primitive wound care products perform admirably in many situations. Modern medicated foam dressings containing antibacterial properties may have beneficial properties compared with other wound care products; however, comparison studies are lacking and need broad-based, randomized, controlled trials to confirm utility. Finally, any choice of wound care product must be tailored to the particular wound and individual patient needs. More large, robust, randomized controlled trials are needed.
Acknowledgments
The authors thank Sarah Maria Paulsen and Rosemary Ellen Brown Smith for their editing, proofreading, and preparation of the manuscript.
1. Nussbaum SR, Carter MJ, Fife CE, et al. An economic evaluation of the impact, cost and Medicare policy implications of chronic non healing wounds. Value Health. 2018;21(1):27-32. doi:10.1016/j.jval.2017.07.007
3. Armstrong DG, Swerdlow MA, Armstrong AA, Conte MS, Padula WV, Bus SA. Five-year mortality and direct costs of care for people with diabetic foot complications are comparable to cancer. J Foot Ankle Res. 2020;13(1)16. doi:10.1186/s13047-020-00383-2
4. Nuutila K, Eriksson E. Moist wound healing with commonly available dressings. Adv Wound Care (New Rochelle). 2021;10(12):685-698. doi:10.1089/wound.2020.1232
5. Dyson M, Young S, Pendle CL, Webster DF, Lang SM. Comparison of the effects of moist and dry conditions on dermal repair. J Investig Dermatol. 1988;91:434-439. doi:10.1111/1523-1747.ep1247646
6. Junker JPE, Kamel RA, Caterson EJ, Eriksson E. Clinical impact upon wound healing and inflammation in moist, wet and dry environments. Adv Wound Care (New Rochelle). 2013;2(7):348-356. doi:10.1089/wound.2012.0412
7. Gupta SS, Singh O, Bhagel PS, Moses S, Shukla S, Mathur RK. Honey dressing versus silver sulfadiazine dressing for wound healing in burn patients: a retrospective study. J Cutan Aesthet Surg. 2011;4(3):183-187. doi:10.4103/0974-2077.91249
8. Fukuda M, Kobayashi K, Hirono Y, et al. Jungle honey enhances immune function and antitumor activity. Evid Based Complement Alternat Med. 2011;2011:1-8. doi:10.1093/ecam/nen086
9. Majtan J, Kumar P, Majtan T, Walls AF, Klaudiny J. Effect of honey and its major royal jelly protein 1 on cytokine and MMP-9 mRNA transcripts in human keratinocytes. Exp Dermatol. 2010;19(8):e73-e79. doi:10.1111/j.1600-0625.2009.00994.x
10. Henriques A, Jackson S, Cooper R, Burton N. Free radical production and quenching in honeys with wound healing potential. J Antimicrob Chemother. 2006;58(4):773-777. doi:10.1093/jac/dkl336
11. Van den Berg AJJ, Van den Worm E, Quarles van Ufford HC, Halkes SBA, M J Hoekstra MJ, Beukelman C J. An in vitro examination of the antioxidant and anti-inflammatory properties of buckwheat honey. J Wound Care. 2008;17(4):172-178. doi:10.12968/jowc.2008.17.4.28839
12. Majtan J. Honey: an immunomodulator in wound healing. Wound Repair Regen. 2014;22(2) 187-192. doi:10.1111/wrr.12117
13. Waffa GA, Hayah AEB. The effectiveness of using banana leaf dressing in management of partial thickness burns’ wound. IJND. 2015;5(4):22-27. doi:10.15520/ijnd.2015.vol5.iss04.70.
14. Gore MA, Akolekar D. Evaluation of banana leaf dressing for partial thickness burn wounds. Burns. 2003;29(5):487-492. doi:10.1016/s0305-4179(03)00050-0
15. Chendake S, Kale T, Manavadaria Y, Motimath AS. Evaluation of banana leaves (Musa paradisiaca) as an alternative wound dressing material compared to conventional petroleum jelly gauze dressing in contused, lacerated and sutured wounds over the head, neck and face region. Cureus. 2021;13(10):1-9. doi:10.7759/cureus.18552
16. Manjunath KS, Bhandage S, Kamat S. ‘Potato peel’ dressing: a novel adjunctive in the management of necrotizing fasciitis. J Maxillofacial Oral Surg. 2015;14(suppl 1):s352-s354. doi:10.1007/s12663-013-0590-8
17. Panda V, Sonkamble M, Patil S. Wound healing activity of Ipomoea batatas tubers (sweet potato). FFHDJ. 2011;1(10):403-415.
18. Subrahmanyam M. Honey dressing versus boiled potato peel in the treatment of burns: a prospective randomized study. Burns. 1996;22(6):491-493. doi:10.1016/0305-4179(96)00007-1.
19. Keswani M H, Vartak AM, Patil A, Davies JW. Histological and bacteriological studies of burn wounds treated with boiled potato peel dressings. Burns. 1990;16(2):137-143. doi:10.1016/0305-4179(90)90175-v
20. Oryan A, Mohammadalipour A, Moshiri A, MR Tabandeh. Topical application of aloe vera accelerated wound healing, modeling, and remodeling, an experimental study. Ann Plast Surg. 2016;77(1)37-46. doi:10.1097/SAP.0000000000000239
21. Bolton L. Evidence corner: April 2007. Wounds. 2007;19(4):A16-A22.
22. Aderibigbe BA, Buyana B. Alginate in wound dressings. Pharmaceutics. 2018;10(2):42. doi.10.3390/pharmaceutics10020042
23. Fletcher J. Using film dressings. Nurs Times. 2003;99(25):57.
24. Ranahewa TH, Premarathna AD, Wijesundara RMKK, Wijewardana V, Jayasooriya AP, Rajapakse RPVJ. Biochemical composition and anticancer effect of different seaweed species (in-vitro and in-vivo studies). Sustainable Marine Structures. 2019;1(2):5-11. doi:10.36956/sms.v1i2.94
25. El Gamal AA. Biological importance of marine algae. Saudi Pharmaceutical J. 2010;18(1):1-25. doi:10.1016/j.jsps.2009.12.001
26. Premarathna AD, Ranahewa TH, Wijesekera RRMKK, et al. Wound healing properties of aqueous extracts of Sargassum Illicifolium: an in vitro assay. Wound Medicine. 2019;24(1):1-7. doi:10.1016/j.wndm.2018.11.001
27. Thomas S. Hydrocolloid dressings in the management of acute wounds: a review of the literature. Int Wound J. 2008;5(5):602-613. doi:10.1111/j.1742-481X.2008.00541.x
28. Dumville JC, Deshpande S, O’Mera K, et al. Hydrocolloid dressing for healing diabetic foot ulcers. Cochrane Database Syst Rev. 2013(8): CD009099. doi:10.1002/14651858.CD009099.pub3
29. Pott FS, Meier MJ, Stocco JGD, Crozeta K, Dayane Ribas J. The effectiveness of hydrocolloid dressings versus other dressings in the healing of pressure ulcers in adults and older adults: a systematic review and meta-analysis. Rev Lat-Am Enfermagem. 2014;22(3):511-520. doi:10.1590/0104-1169.3480.2445
30. Davies P, Rippon M. Comparison of foam and hydrocolloid dressings in the management of wounds: a review of the published literature. Accessed May 17, 2023. http://www.worldwidewounds.com/2010/July/DaviesRippon/DaviesRippon.html
31. Bianchi J, Gray D, Timmons J. Meaume S. Do all foam dressings have the same efficacy in the treatment of chronic wounds? Wounds UK. 2011;7(1):62-67.

33. Lee SM, Park IK, Kim HJ, et al. Physical, morphological, and wound healing properties of a polyurethane foam-film dressing. Biomaterials Res. 2016;20(15):1-11. doi:10.1186/s40824-016-0063-5
34. Sibbald RG, Meaume S, Kirsner RS, et al. Review of the clinical RCT evidence and cost-effectiveness data of a sustained-release silver foam dressing in the healing of critically colonized wounds. December 2005. Updated January 2006. Accessed May 18, 2023. http://www.worldwidewounds.com/2005/december/Sibbald/Silver-Foam-Dressings-Colonised-Wounds.html
35. Varma AK, Kumar H, Kesav Raiesh. Efficacy of polyurethane foam dressing in debrided diabetic lower limb wounds. Wounds. 2006;18(10):300-306.
36. Aziz Z, Abu SF, Chong NJ. A systematic review of silver-containing dressings and topical silver agents (used with dressings) for burn wounds. Burns. 2012;38(3):307-318. doi:10.1016/j.burns.2011.09.020
37. Agarwal P, Kukrele R, Sharma D. Vacuum assisted closure (VAC)/negative pressure wound therapy (NPWT) for difficult wounds: a review. J Clin Orthop Trauma. 2019;10(5):845-848. doi:10.1016/j.jcot.2019.06.015
1. Nussbaum SR, Carter MJ, Fife CE, et al. An economic evaluation of the impact, cost and Medicare policy implications of chronic non healing wounds. Value Health. 2018;21(1):27-32. doi:10.1016/j.jval.2017.07.007
3. Armstrong DG, Swerdlow MA, Armstrong AA, Conte MS, Padula WV, Bus SA. Five-year mortality and direct costs of care for people with diabetic foot complications are comparable to cancer. J Foot Ankle Res. 2020;13(1)16. doi:10.1186/s13047-020-00383-2
4. Nuutila K, Eriksson E. Moist wound healing with commonly available dressings. Adv Wound Care (New Rochelle). 2021;10(12):685-698. doi:10.1089/wound.2020.1232
5. Dyson M, Young S, Pendle CL, Webster DF, Lang SM. Comparison of the effects of moist and dry conditions on dermal repair. J Investig Dermatol. 1988;91:434-439. doi:10.1111/1523-1747.ep1247646
6. Junker JPE, Kamel RA, Caterson EJ, Eriksson E. Clinical impact upon wound healing and inflammation in moist, wet and dry environments. Adv Wound Care (New Rochelle). 2013;2(7):348-356. doi:10.1089/wound.2012.0412
7. Gupta SS, Singh O, Bhagel PS, Moses S, Shukla S, Mathur RK. Honey dressing versus silver sulfadiazine dressing for wound healing in burn patients: a retrospective study. J Cutan Aesthet Surg. 2011;4(3):183-187. doi:10.4103/0974-2077.91249
8. Fukuda M, Kobayashi K, Hirono Y, et al. Jungle honey enhances immune function and antitumor activity. Evid Based Complement Alternat Med. 2011;2011:1-8. doi:10.1093/ecam/nen086
9. Majtan J, Kumar P, Majtan T, Walls AF, Klaudiny J. Effect of honey and its major royal jelly protein 1 on cytokine and MMP-9 mRNA transcripts in human keratinocytes. Exp Dermatol. 2010;19(8):e73-e79. doi:10.1111/j.1600-0625.2009.00994.x
10. Henriques A, Jackson S, Cooper R, Burton N. Free radical production and quenching in honeys with wound healing potential. J Antimicrob Chemother. 2006;58(4):773-777. doi:10.1093/jac/dkl336
11. Van den Berg AJJ, Van den Worm E, Quarles van Ufford HC, Halkes SBA, M J Hoekstra MJ, Beukelman C J. An in vitro examination of the antioxidant and anti-inflammatory properties of buckwheat honey. J Wound Care. 2008;17(4):172-178. doi:10.12968/jowc.2008.17.4.28839
12. Majtan J. Honey: an immunomodulator in wound healing. Wound Repair Regen. 2014;22(2) 187-192. doi:10.1111/wrr.12117
13. Waffa GA, Hayah AEB. The effectiveness of using banana leaf dressing in management of partial thickness burns’ wound. IJND. 2015;5(4):22-27. doi:10.15520/ijnd.2015.vol5.iss04.70.
14. Gore MA, Akolekar D. Evaluation of banana leaf dressing for partial thickness burn wounds. Burns. 2003;29(5):487-492. doi:10.1016/s0305-4179(03)00050-0
15. Chendake S, Kale T, Manavadaria Y, Motimath AS. Evaluation of banana leaves (Musa paradisiaca) as an alternative wound dressing material compared to conventional petroleum jelly gauze dressing in contused, lacerated and sutured wounds over the head, neck and face region. Cureus. 2021;13(10):1-9. doi:10.7759/cureus.18552
16. Manjunath KS, Bhandage S, Kamat S. ‘Potato peel’ dressing: a novel adjunctive in the management of necrotizing fasciitis. J Maxillofacial Oral Surg. 2015;14(suppl 1):s352-s354. doi:10.1007/s12663-013-0590-8
17. Panda V, Sonkamble M, Patil S. Wound healing activity of Ipomoea batatas tubers (sweet potato). FFHDJ. 2011;1(10):403-415.
18. Subrahmanyam M. Honey dressing versus boiled potato peel in the treatment of burns: a prospective randomized study. Burns. 1996;22(6):491-493. doi:10.1016/0305-4179(96)00007-1.
19. Keswani M H, Vartak AM, Patil A, Davies JW. Histological and bacteriological studies of burn wounds treated with boiled potato peel dressings. Burns. 1990;16(2):137-143. doi:10.1016/0305-4179(90)90175-v
20. Oryan A, Mohammadalipour A, Moshiri A, MR Tabandeh. Topical application of aloe vera accelerated wound healing, modeling, and remodeling, an experimental study. Ann Plast Surg. 2016;77(1)37-46. doi:10.1097/SAP.0000000000000239
21. Bolton L. Evidence corner: April 2007. Wounds. 2007;19(4):A16-A22.
22. Aderibigbe BA, Buyana B. Alginate in wound dressings. Pharmaceutics. 2018;10(2):42. doi.10.3390/pharmaceutics10020042
23. Fletcher J. Using film dressings. Nurs Times. 2003;99(25):57.
24. Ranahewa TH, Premarathna AD, Wijesundara RMKK, Wijewardana V, Jayasooriya AP, Rajapakse RPVJ. Biochemical composition and anticancer effect of different seaweed species (in-vitro and in-vivo studies). Sustainable Marine Structures. 2019;1(2):5-11. doi:10.36956/sms.v1i2.94
25. El Gamal AA. Biological importance of marine algae. Saudi Pharmaceutical J. 2010;18(1):1-25. doi:10.1016/j.jsps.2009.12.001
26. Premarathna AD, Ranahewa TH, Wijesekera RRMKK, et al. Wound healing properties of aqueous extracts of Sargassum Illicifolium: an in vitro assay. Wound Medicine. 2019;24(1):1-7. doi:10.1016/j.wndm.2018.11.001
27. Thomas S. Hydrocolloid dressings in the management of acute wounds: a review of the literature. Int Wound J. 2008;5(5):602-613. doi:10.1111/j.1742-481X.2008.00541.x
28. Dumville JC, Deshpande S, O’Mera K, et al. Hydrocolloid dressing for healing diabetic foot ulcers. Cochrane Database Syst Rev. 2013(8): CD009099. doi:10.1002/14651858.CD009099.pub3
29. Pott FS, Meier MJ, Stocco JGD, Crozeta K, Dayane Ribas J. The effectiveness of hydrocolloid dressings versus other dressings in the healing of pressure ulcers in adults and older adults: a systematic review and meta-analysis. Rev Lat-Am Enfermagem. 2014;22(3):511-520. doi:10.1590/0104-1169.3480.2445
30. Davies P, Rippon M. Comparison of foam and hydrocolloid dressings in the management of wounds: a review of the published literature. Accessed May 17, 2023. http://www.worldwidewounds.com/2010/July/DaviesRippon/DaviesRippon.html
31. Bianchi J, Gray D, Timmons J. Meaume S. Do all foam dressings have the same efficacy in the treatment of chronic wounds? Wounds UK. 2011;7(1):62-67.

33. Lee SM, Park IK, Kim HJ, et al. Physical, morphological, and wound healing properties of a polyurethane foam-film dressing. Biomaterials Res. 2016;20(15):1-11. doi:10.1186/s40824-016-0063-5
34. Sibbald RG, Meaume S, Kirsner RS, et al. Review of the clinical RCT evidence and cost-effectiveness data of a sustained-release silver foam dressing in the healing of critically colonized wounds. December 2005. Updated January 2006. Accessed May 18, 2023. http://www.worldwidewounds.com/2005/december/Sibbald/Silver-Foam-Dressings-Colonised-Wounds.html
35. Varma AK, Kumar H, Kesav Raiesh. Efficacy of polyurethane foam dressing in debrided diabetic lower limb wounds. Wounds. 2006;18(10):300-306.
36. Aziz Z, Abu SF, Chong NJ. A systematic review of silver-containing dressings and topical silver agents (used with dressings) for burn wounds. Burns. 2012;38(3):307-318. doi:10.1016/j.burns.2011.09.020
37. Agarwal P, Kukrele R, Sharma D. Vacuum assisted closure (VAC)/negative pressure wound therapy (NPWT) for difficult wounds: a review. J Clin Orthop Trauma. 2019;10(5):845-848. doi:10.1016/j.jcot.2019.06.015
30-year-old woman • progressive dyspnea and peripheral edema • 35th week of gestation with a history of mild preeclampsia • Dx?
THE CASE
A 30-year-old woman sought care at her rural family physician’s office for progressive dyspnea and peripheral edema, which she had been experiencing for several weeks. She was G1P0 and in her 35th week of gestation.
Her medical history was remarkable for mild preeclampsia, which was being managed observantly by her obstetrician in consultation with a maternal-fetal medicine specialist. She had been evaluated by her local hospital’s labor and delivery department and her maternal-fetal medicine specialist earlier in the week and seen the previous day by her obstetrician for these signs and symptoms. They all reassured her and told her these symptoms were normal during pregnancy. No diagnostic studies were performed. However, she remained concerned and decided to see her family physician for another opinion.
Upon presentation to her family physician, the patient was afebrile. Her blood pressure was 135/98 mm Hg; heart rate, 96 beats/min; and respiration, 20 breaths/min and slightly labored. Edema of 2 to 3+ was noted in her lower extremities, hands, and face. Bibasilar breath sounds were diminished, and her abdomen was nontender.
The family physician suspected left ventricular systolic dysfunction. He worked in a small office that lacked access to a laboratory or radiographic studies. However, he did have an ultrasound machine available, and although he was not skilled in echocardiography to assess cardiac function, he was able to obtain a bedside lung ultrasound.
THE DIAGNOSIS
While no B-lines were seen on the lung ultrasound, bilateral plural effusions were noted (FIGURE). This finding, paired with the patient’s signs and symptoms, prompted the family physician to suspect a diagnosis of acute decompensated heart failure with presumptive peripartum cardiomyopathy. The patient was immediately driven to the hospital by her family physician for emergency admission with stat obstetric and cardiology consultations.
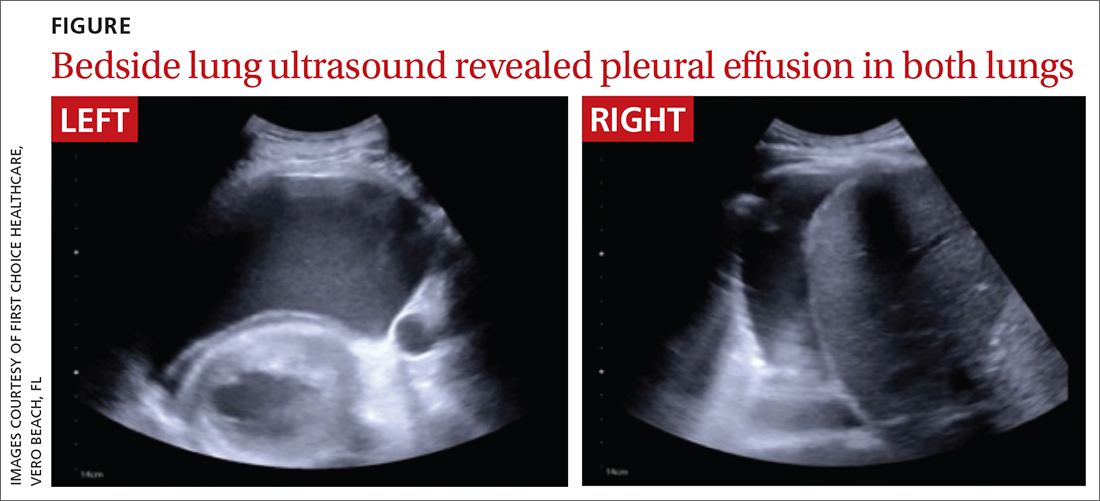
An in-hospital echocardiogram revealed severe global hypokinesia with a left ventricular ejection fraction of 25% to 30%, which confirmed the family physician’s suspicions. Laboratory studies were significant for elevated N-terminal pro-brain natriuretic peptide (43,449 pg/mL; normal, < 125 pg/mL), troponin (1.12 ng/mL; normal range, 0-0.10 ng/mL), and white blood cell count (27.6 x 103/µL). She also had evidence of acute renal injury, with blood urea nitrogen of 46 mg/dL (normal range, 7-18 mg/dL), creatinine of 2.0 mg/dL (normal range, 0.5-1.0 mg/dL), and potassium of 7.6 mmol/L (normal range, 3.5-5.1 mmol/L). Emergency delivery was induced by amniotomy, resulting in the birth of a baby girl weighing 5 lb 4 oz (Apgar scores 6, 8, and 9).
Following delivery, the patient was placed on a milrinone infusion and required dialysis. She was emergently transferred to a tertiary care hospital, where she was admitted to the cardiac intensive care unit by the cardiology/heart transplant service with nephrology and obstetric consultations. Hematology and infectious disease specialists were consulted to rule out HELLP (hemolysis, elevated liver enzymes, and low platelets) syndrome and sepsis, respectively. Her course of care remained complicated with further testing, including cardiac catheterization and biopsy, which was negative for additional pathology.
Continue to: One week after admission...
One week after admission, she was discharged home with a 24-hour wearable external cardiac defibrillator and a confirmed diagnosis of peripartum cardiomyopathy. Her medication regimen included digoxin (125 µg 3 times/wk), spironolactone (25 mg/d), carvedilol (3.125 mg twice daily), sacubitril/valsartan (24 mg/26 mg twice daily), furosemide (20 mg/d as needed for weight gain > 3-4 lb or leg swelling), magnesium oxide (400 mg twice daily), and ferrous sulfate (325 mg/d).
DISCUSSION
Peripartum cardiomyopathy is a rare, life-threatening, idiopathic cardiomyopathy that is responsible for one-half to two-thirds of cardiovascular disease–related maternal deaths in the United States.1,2 It manifests in late pregnancy or early in the postpartum period and is characterized by left ventricular systolic dysfunction with resultant heart failure and an ejection fraction of less than 45%.1,2
Recognized as early as the 1800s by Virchow,2,3 the incidence of peripartum cardiomyopathy in the United States ranges from 1 in 1000 to 4000 live births and is increasing worldwide.1,2 While the cause of peripartum cardiomyopathy remains unknown, risk factors include advanced maternal age, African descent, hypertension, preeclampsia, and multiple gestation pregnancy.1,2
Early diagnosis of peripartum cardiomyopathy is imperative for survival of both mother and baby.4 This may be difficult because the signs and symptoms of heart failure—such as dyspnea, edema, orthopnea, cough, and chest and abdominal pain—overlap with those of a typical pregnancy, resulting in it often being missed on evaluation.1,2
Dx with echocardiography; in a pinch, consider lung ultrasound
Usually a diagnosis of peripartum cardiomyopathy is established with echocardiography.1,2 Thus, this case is of significant importance because it illustrates the successful use of lung ultrasound—a simple and easy test—by a rural family doctor to identify this potentially fatal, elusive condition with no additional studies.
Continue to: Use of lung ultrasound...
Use of lung ultrasound in the detection of acute decompensated heart failure is accepted in the medical literature.5-7 Given clinical correlation, a positive scan is defined by the presence of at least 3 B-lines on a longitudinal plane between 2 ribs or, as seen in our case, by the presence of pleural effusion.5-8 Lung ultrasound is readily available worldwide, is completely safe in pregnancy, and is considered one of the easiest studies to perform.7-10
At the patient’s 9-month follow-up visit, she had made a full clinical recovery. Her ejection fraction was 59.8%, and she had stopped all medications. The patient and her child did not experience any continued complications.
THE TAKEAWAY
Family physicians should be aware of peripartum cardiomyopathy—one of the most elusive and life-threatening diseases of pregnancy. When managing a pregnant patient, it is imperative to follow up on complaints such as dyspnea, peripheral edema, and chest and/or abdominal pain. While these symptoms are not unusual during pregnancy, they should always prompt a more thorough evaluation. If peripartum cardiomyopathy is suspected, lung ultrasound is a valuable diagnostic tool for family physicians. Further research is needed before the findings of this case report can be universally applied in the routine prenatal care of women at risk for peripartum cardiomyopathy.
The authors thank their daughter, Nickel Cielo Abarbanell, for her help in the preparation of this manuscript.
CORRESPONDENCE
Neal Robert Abarbanell, MD, First Choice Healthcare, 1867 20th Avenue, Vero Beach, FL 32960; neal.abarbanell@ gmail.com
1. Honigberg MC, Givertz MM. Peripartum cardiomyopathy. BMJ. 2019;364:k5287. doi: 10.1136/bmj.k5287
2. Arany Z, Elkayam U. Peripartum cardiomyopathy. Circulation. 2016;133:1397-1409. doi: 10.1161/CIRCULATIONAHA.115.020491
3. Porak C. De L’influence reciproque de la grossesse et del maladies du Coceur [thesis]. Medical Faculty of Paris, France: 1880.
4. Lewey J, Levine LD, Elovitz MA, et al. Importance of early diagnosis in peripartum cardiomyopathy. Hypertension. 2020;75:91-97. doi: 10.1161/HYPERTENSIONAHA.119.13291
5. Volpicelli G, Caramello V, Cardinale L, et al. Bedside ultrasound of the lung for the monitoring of acute decompensated heart failure. Am J Emerg Med. 2008;26:585-591. doi: 10.1016/j.ajem.2007.09.014
6. Muniz RT, Mesquita ET, Souza CV Jr, et al. Pulmonary ultrasound in patients with heart failure-systematic review. Arq Bras Cardiol. 2018;110:577-584. doi: 10.5935/abc.20180097
7. Russell FM, Rutz M, Pang PS. Focused ultrasound in the emergency department for patients with acute heart failure. Card Fail Rev. 2015;1:83-86. doi: 10.15420/cfr.2015.1.2.83
8. Gustafsson M, Alehagen U, Johansson P. Imaging congestion with a pocket ultrasound device: prognostic implications in patients with chronic heart failure. J Card Fail. 2015;21:548-554. doi: 10.1016/j.cardfail.2015.02.004
9. Ntusi NA, Samuels P, Moosa S, et al. Diagnosing cardiac disease during pregnancy: imaging modalities. Cardiovasc J Afr. 2016;27:95-103. doi: 10.5830/CVJA-2016-022
10. Kimberly HH, Murray A, Mennicke M, et al. Focused maternal ultrasound by midwives in rural Zambia. Ultrasound Med Biol. 2010;36:1267-1272. doi: 10.1016/j.ultrasmedbio.2010.05.017
THE CASE
A 30-year-old woman sought care at her rural family physician’s office for progressive dyspnea and peripheral edema, which she had been experiencing for several weeks. She was G1P0 and in her 35th week of gestation.
Her medical history was remarkable for mild preeclampsia, which was being managed observantly by her obstetrician in consultation with a maternal-fetal medicine specialist. She had been evaluated by her local hospital’s labor and delivery department and her maternal-fetal medicine specialist earlier in the week and seen the previous day by her obstetrician for these signs and symptoms. They all reassured her and told her these symptoms were normal during pregnancy. No diagnostic studies were performed. However, she remained concerned and decided to see her family physician for another opinion.
Upon presentation to her family physician, the patient was afebrile. Her blood pressure was 135/98 mm Hg; heart rate, 96 beats/min; and respiration, 20 breaths/min and slightly labored. Edema of 2 to 3+ was noted in her lower extremities, hands, and face. Bibasilar breath sounds were diminished, and her abdomen was nontender.
The family physician suspected left ventricular systolic dysfunction. He worked in a small office that lacked access to a laboratory or radiographic studies. However, he did have an ultrasound machine available, and although he was not skilled in echocardiography to assess cardiac function, he was able to obtain a bedside lung ultrasound.
THE DIAGNOSIS
While no B-lines were seen on the lung ultrasound, bilateral plural effusions were noted (FIGURE). This finding, paired with the patient’s signs and symptoms, prompted the family physician to suspect a diagnosis of acute decompensated heart failure with presumptive peripartum cardiomyopathy. The patient was immediately driven to the hospital by her family physician for emergency admission with stat obstetric and cardiology consultations.

An in-hospital echocardiogram revealed severe global hypokinesia with a left ventricular ejection fraction of 25% to 30%, which confirmed the family physician’s suspicions. Laboratory studies were significant for elevated N-terminal pro-brain natriuretic peptide (43,449 pg/mL; normal, < 125 pg/mL), troponin (1.12 ng/mL; normal range, 0-0.10 ng/mL), and white blood cell count (27.6 x 103/µL). She also had evidence of acute renal injury, with blood urea nitrogen of 46 mg/dL (normal range, 7-18 mg/dL), creatinine of 2.0 mg/dL (normal range, 0.5-1.0 mg/dL), and potassium of 7.6 mmol/L (normal range, 3.5-5.1 mmol/L). Emergency delivery was induced by amniotomy, resulting in the birth of a baby girl weighing 5 lb 4 oz (Apgar scores 6, 8, and 9).
Following delivery, the patient was placed on a milrinone infusion and required dialysis. She was emergently transferred to a tertiary care hospital, where she was admitted to the cardiac intensive care unit by the cardiology/heart transplant service with nephrology and obstetric consultations. Hematology and infectious disease specialists were consulted to rule out HELLP (hemolysis, elevated liver enzymes, and low platelets) syndrome and sepsis, respectively. Her course of care remained complicated with further testing, including cardiac catheterization and biopsy, which was negative for additional pathology.
Continue to: One week after admission...
One week after admission, she was discharged home with a 24-hour wearable external cardiac defibrillator and a confirmed diagnosis of peripartum cardiomyopathy. Her medication regimen included digoxin (125 µg 3 times/wk), spironolactone (25 mg/d), carvedilol (3.125 mg twice daily), sacubitril/valsartan (24 mg/26 mg twice daily), furosemide (20 mg/d as needed for weight gain > 3-4 lb or leg swelling), magnesium oxide (400 mg twice daily), and ferrous sulfate (325 mg/d).
DISCUSSION
Peripartum cardiomyopathy is a rare, life-threatening, idiopathic cardiomyopathy that is responsible for one-half to two-thirds of cardiovascular disease–related maternal deaths in the United States.1,2 It manifests in late pregnancy or early in the postpartum period and is characterized by left ventricular systolic dysfunction with resultant heart failure and an ejection fraction of less than 45%.1,2
Recognized as early as the 1800s by Virchow,2,3 the incidence of peripartum cardiomyopathy in the United States ranges from 1 in 1000 to 4000 live births and is increasing worldwide.1,2 While the cause of peripartum cardiomyopathy remains unknown, risk factors include advanced maternal age, African descent, hypertension, preeclampsia, and multiple gestation pregnancy.1,2
Early diagnosis of peripartum cardiomyopathy is imperative for survival of both mother and baby.4 This may be difficult because the signs and symptoms of heart failure—such as dyspnea, edema, orthopnea, cough, and chest and abdominal pain—overlap with those of a typical pregnancy, resulting in it often being missed on evaluation.1,2
Dx with echocardiography; in a pinch, consider lung ultrasound
Usually a diagnosis of peripartum cardiomyopathy is established with echocardiography.1,2 Thus, this case is of significant importance because it illustrates the successful use of lung ultrasound—a simple and easy test—by a rural family doctor to identify this potentially fatal, elusive condition with no additional studies.
Continue to: Use of lung ultrasound...
Use of lung ultrasound in the detection of acute decompensated heart failure is accepted in the medical literature.5-7 Given clinical correlation, a positive scan is defined by the presence of at least 3 B-lines on a longitudinal plane between 2 ribs or, as seen in our case, by the presence of pleural effusion.5-8 Lung ultrasound is readily available worldwide, is completely safe in pregnancy, and is considered one of the easiest studies to perform.7-10
At the patient’s 9-month follow-up visit, she had made a full clinical recovery. Her ejection fraction was 59.8%, and she had stopped all medications. The patient and her child did not experience any continued complications.
THE TAKEAWAY
Family physicians should be aware of peripartum cardiomyopathy—one of the most elusive and life-threatening diseases of pregnancy. When managing a pregnant patient, it is imperative to follow up on complaints such as dyspnea, peripheral edema, and chest and/or abdominal pain. While these symptoms are not unusual during pregnancy, they should always prompt a more thorough evaluation. If peripartum cardiomyopathy is suspected, lung ultrasound is a valuable diagnostic tool for family physicians. Further research is needed before the findings of this case report can be universally applied in the routine prenatal care of women at risk for peripartum cardiomyopathy.
The authors thank their daughter, Nickel Cielo Abarbanell, for her help in the preparation of this manuscript.
CORRESPONDENCE
Neal Robert Abarbanell, MD, First Choice Healthcare, 1867 20th Avenue, Vero Beach, FL 32960; neal.abarbanell@ gmail.com
THE CASE
A 30-year-old woman sought care at her rural family physician’s office for progressive dyspnea and peripheral edema, which she had been experiencing for several weeks. She was G1P0 and in her 35th week of gestation.
Her medical history was remarkable for mild preeclampsia, which was being managed observantly by her obstetrician in consultation with a maternal-fetal medicine specialist. She had been evaluated by her local hospital’s labor and delivery department and her maternal-fetal medicine specialist earlier in the week and seen the previous day by her obstetrician for these signs and symptoms. They all reassured her and told her these symptoms were normal during pregnancy. No diagnostic studies were performed. However, she remained concerned and decided to see her family physician for another opinion.
Upon presentation to her family physician, the patient was afebrile. Her blood pressure was 135/98 mm Hg; heart rate, 96 beats/min; and respiration, 20 breaths/min and slightly labored. Edema of 2 to 3+ was noted in her lower extremities, hands, and face. Bibasilar breath sounds were diminished, and her abdomen was nontender.
The family physician suspected left ventricular systolic dysfunction. He worked in a small office that lacked access to a laboratory or radiographic studies. However, he did have an ultrasound machine available, and although he was not skilled in echocardiography to assess cardiac function, he was able to obtain a bedside lung ultrasound.
THE DIAGNOSIS
While no B-lines were seen on the lung ultrasound, bilateral plural effusions were noted (FIGURE). This finding, paired with the patient’s signs and symptoms, prompted the family physician to suspect a diagnosis of acute decompensated heart failure with presumptive peripartum cardiomyopathy. The patient was immediately driven to the hospital by her family physician for emergency admission with stat obstetric and cardiology consultations.

An in-hospital echocardiogram revealed severe global hypokinesia with a left ventricular ejection fraction of 25% to 30%, which confirmed the family physician’s suspicions. Laboratory studies were significant for elevated N-terminal pro-brain natriuretic peptide (43,449 pg/mL; normal, < 125 pg/mL), troponin (1.12 ng/mL; normal range, 0-0.10 ng/mL), and white blood cell count (27.6 x 103/µL). She also had evidence of acute renal injury, with blood urea nitrogen of 46 mg/dL (normal range, 7-18 mg/dL), creatinine of 2.0 mg/dL (normal range, 0.5-1.0 mg/dL), and potassium of 7.6 mmol/L (normal range, 3.5-5.1 mmol/L). Emergency delivery was induced by amniotomy, resulting in the birth of a baby girl weighing 5 lb 4 oz (Apgar scores 6, 8, and 9).
Following delivery, the patient was placed on a milrinone infusion and required dialysis. She was emergently transferred to a tertiary care hospital, where she was admitted to the cardiac intensive care unit by the cardiology/heart transplant service with nephrology and obstetric consultations. Hematology and infectious disease specialists were consulted to rule out HELLP (hemolysis, elevated liver enzymes, and low platelets) syndrome and sepsis, respectively. Her course of care remained complicated with further testing, including cardiac catheterization and biopsy, which was negative for additional pathology.
Continue to: One week after admission...
One week after admission, she was discharged home with a 24-hour wearable external cardiac defibrillator and a confirmed diagnosis of peripartum cardiomyopathy. Her medication regimen included digoxin (125 µg 3 times/wk), spironolactone (25 mg/d), carvedilol (3.125 mg twice daily), sacubitril/valsartan (24 mg/26 mg twice daily), furosemide (20 mg/d as needed for weight gain > 3-4 lb or leg swelling), magnesium oxide (400 mg twice daily), and ferrous sulfate (325 mg/d).
DISCUSSION
Peripartum cardiomyopathy is a rare, life-threatening, idiopathic cardiomyopathy that is responsible for one-half to two-thirds of cardiovascular disease–related maternal deaths in the United States.1,2 It manifests in late pregnancy or early in the postpartum period and is characterized by left ventricular systolic dysfunction with resultant heart failure and an ejection fraction of less than 45%.1,2
Recognized as early as the 1800s by Virchow,2,3 the incidence of peripartum cardiomyopathy in the United States ranges from 1 in 1000 to 4000 live births and is increasing worldwide.1,2 While the cause of peripartum cardiomyopathy remains unknown, risk factors include advanced maternal age, African descent, hypertension, preeclampsia, and multiple gestation pregnancy.1,2
Early diagnosis of peripartum cardiomyopathy is imperative for survival of both mother and baby.4 This may be difficult because the signs and symptoms of heart failure—such as dyspnea, edema, orthopnea, cough, and chest and abdominal pain—overlap with those of a typical pregnancy, resulting in it often being missed on evaluation.1,2
Dx with echocardiography; in a pinch, consider lung ultrasound
Usually a diagnosis of peripartum cardiomyopathy is established with echocardiography.1,2 Thus, this case is of significant importance because it illustrates the successful use of lung ultrasound—a simple and easy test—by a rural family doctor to identify this potentially fatal, elusive condition with no additional studies.
Continue to: Use of lung ultrasound...
Use of lung ultrasound in the detection of acute decompensated heart failure is accepted in the medical literature.5-7 Given clinical correlation, a positive scan is defined by the presence of at least 3 B-lines on a longitudinal plane between 2 ribs or, as seen in our case, by the presence of pleural effusion.5-8 Lung ultrasound is readily available worldwide, is completely safe in pregnancy, and is considered one of the easiest studies to perform.7-10
At the patient’s 9-month follow-up visit, she had made a full clinical recovery. Her ejection fraction was 59.8%, and she had stopped all medications. The patient and her child did not experience any continued complications.
THE TAKEAWAY
Family physicians should be aware of peripartum cardiomyopathy—one of the most elusive and life-threatening diseases of pregnancy. When managing a pregnant patient, it is imperative to follow up on complaints such as dyspnea, peripheral edema, and chest and/or abdominal pain. While these symptoms are not unusual during pregnancy, they should always prompt a more thorough evaluation. If peripartum cardiomyopathy is suspected, lung ultrasound is a valuable diagnostic tool for family physicians. Further research is needed before the findings of this case report can be universally applied in the routine prenatal care of women at risk for peripartum cardiomyopathy.
The authors thank their daughter, Nickel Cielo Abarbanell, for her help in the preparation of this manuscript.
CORRESPONDENCE
Neal Robert Abarbanell, MD, First Choice Healthcare, 1867 20th Avenue, Vero Beach, FL 32960; neal.abarbanell@ gmail.com
1. Honigberg MC, Givertz MM. Peripartum cardiomyopathy. BMJ. 2019;364:k5287. doi: 10.1136/bmj.k5287
2. Arany Z, Elkayam U. Peripartum cardiomyopathy. Circulation. 2016;133:1397-1409. doi: 10.1161/CIRCULATIONAHA.115.020491
3. Porak C. De L’influence reciproque de la grossesse et del maladies du Coceur [thesis]. Medical Faculty of Paris, France: 1880.
4. Lewey J, Levine LD, Elovitz MA, et al. Importance of early diagnosis in peripartum cardiomyopathy. Hypertension. 2020;75:91-97. doi: 10.1161/HYPERTENSIONAHA.119.13291
5. Volpicelli G, Caramello V, Cardinale L, et al. Bedside ultrasound of the lung for the monitoring of acute decompensated heart failure. Am J Emerg Med. 2008;26:585-591. doi: 10.1016/j.ajem.2007.09.014
6. Muniz RT, Mesquita ET, Souza CV Jr, et al. Pulmonary ultrasound in patients with heart failure-systematic review. Arq Bras Cardiol. 2018;110:577-584. doi: 10.5935/abc.20180097
7. Russell FM, Rutz M, Pang PS. Focused ultrasound in the emergency department for patients with acute heart failure. Card Fail Rev. 2015;1:83-86. doi: 10.15420/cfr.2015.1.2.83
8. Gustafsson M, Alehagen U, Johansson P. Imaging congestion with a pocket ultrasound device: prognostic implications in patients with chronic heart failure. J Card Fail. 2015;21:548-554. doi: 10.1016/j.cardfail.2015.02.004
9. Ntusi NA, Samuels P, Moosa S, et al. Diagnosing cardiac disease during pregnancy: imaging modalities. Cardiovasc J Afr. 2016;27:95-103. doi: 10.5830/CVJA-2016-022
10. Kimberly HH, Murray A, Mennicke M, et al. Focused maternal ultrasound by midwives in rural Zambia. Ultrasound Med Biol. 2010;36:1267-1272. doi: 10.1016/j.ultrasmedbio.2010.05.017
1. Honigberg MC, Givertz MM. Peripartum cardiomyopathy. BMJ. 2019;364:k5287. doi: 10.1136/bmj.k5287
2. Arany Z, Elkayam U. Peripartum cardiomyopathy. Circulation. 2016;133:1397-1409. doi: 10.1161/CIRCULATIONAHA.115.020491
3. Porak C. De L’influence reciproque de la grossesse et del maladies du Coceur [thesis]. Medical Faculty of Paris, France: 1880.
4. Lewey J, Levine LD, Elovitz MA, et al. Importance of early diagnosis in peripartum cardiomyopathy. Hypertension. 2020;75:91-97. doi: 10.1161/HYPERTENSIONAHA.119.13291
5. Volpicelli G, Caramello V, Cardinale L, et al. Bedside ultrasound of the lung for the monitoring of acute decompensated heart failure. Am J Emerg Med. 2008;26:585-591. doi: 10.1016/j.ajem.2007.09.014
6. Muniz RT, Mesquita ET, Souza CV Jr, et al. Pulmonary ultrasound in patients with heart failure-systematic review. Arq Bras Cardiol. 2018;110:577-584. doi: 10.5935/abc.20180097
7. Russell FM, Rutz M, Pang PS. Focused ultrasound in the emergency department for patients with acute heart failure. Card Fail Rev. 2015;1:83-86. doi: 10.15420/cfr.2015.1.2.83
8. Gustafsson M, Alehagen U, Johansson P. Imaging congestion with a pocket ultrasound device: prognostic implications in patients with chronic heart failure. J Card Fail. 2015;21:548-554. doi: 10.1016/j.cardfail.2015.02.004
9. Ntusi NA, Samuels P, Moosa S, et al. Diagnosing cardiac disease during pregnancy: imaging modalities. Cardiovasc J Afr. 2016;27:95-103. doi: 10.5830/CVJA-2016-022
10. Kimberly HH, Murray A, Mennicke M, et al. Focused maternal ultrasound by midwives in rural Zambia. Ultrasound Med Biol. 2010;36:1267-1272. doi: 10.1016/j.ultrasmedbio.2010.05.017
► Progressive dyspnea and peripheral edema
► 35th week of gestation with a history of mild preeclampsia
A Case of Duodenocaval Fistula in the Setting of Respiratory Failure Initially Confused for Transfusion-Related Acute Lung Injury
A duodenocaval fistula (DCF) is seen when a connection exists between the duodenum and the inferior vena cava. It is a rare entity that is commonly missed and presents a diagnostic challenge due to its nonspecific presenting symptoms.1,2 Patients commonly present with gastrointestinal (GI) bleeding or sepsis. Here we present a case of a 37-year-old man who presented to the hospital for a workup related to melena but went into cardiac arrest prior to an esophagogastroduodenoscopy. Unfortunately, on autopsy, the patient was found to have a DCF. We highlight the diagnostic challenge associated with DCF and how in this case the presentation was confused by a diagnosis of possible transfusion-related acute lung injury (TRALI). To the best of our knowledge, this is also the first description of a case of DCF associated with food embolism to the lungs causing respiratory failure.
Case Presentation
A 37-year-old man with a history significant for bulimia presented to the hospital with a 3-day history of melena and reports of dizziness. The patient did not report being on any prescribed medications but noted that he took 4 aspirin daily to “calm his nerves.” The rest of the patient’s history was unremarkable aside from a reported history of induced emesis 3 to 4 times per week for an extended period up until 2 weeks before admission.
On admission, his vital signs demonstrated tachycardia and orthostatic hypotension. Pertinent findings on physical examination were skin pallor, a normal lung examination, mild epigastric tenderness, and guaiac-positive stools. He was alert and oriented to person, place, and time with no focal deficits. His admission laboratory tests were notable for a hemoglobin (Hb) level of 4.6 g/dL (reference range, 14-17.9), a white blood cell count of 13.5 K/cm (reference range, 4.5-11), an international normalized ratio of 1.21, a blood urea nitrogen of 61 mg/dL (reference range, 10-20), and a creatinine of 2.3 mg/dL (reference range, 0.8-1.4). The patient was placed on 2 L of oxygen via nasal cannula for comfort rather than true hypoxia. A chest X-ray on admission was negative with no signs of infiltrate, edema, or widened mediastinum. An abdominal X-ray was significant for a dilated stomach consistent with bulimia with no abdominal free air or signs of obstruction. The case was discussed with the gastroenterology service who felt that the patient needed to be more hemodynamically stable before pursuing endoscopic evaluation.
He was admitted to the intensive care unit and give a transfusion of 4 units of fresh frozen plasma and 2 packed red blood cells (PRBCs) without any issues. During the infusion of a third PRBC, he developed chills, tachycardia, and hypertension with accompanying respiratory distress characterized by wheezing, decreased breath sounds bilaterally, and a decrease in oxygen saturation to 70% on 2 L supplemental oxygen. He responded to treatment with meperidine, methylprednisolone sodium succinate, albuterol nebulizer, and acetaminophen. A new chest X-ray was read as “development of pulmonary edema vs bilateral pneumonitis.” A transfusion reaction was reported to the blood bank and a diagnosis of TRALI was considered. That evening, he completed a dose of platelets and another PRBC without difficulty after he was premedicated with meperidine, methylprednisolone sodium succinate, and acetaminophen. During the night, the patient spiked a temperature of 40.3 °C that was successfully treated with a cooling blanket and acetaminophen.
The following morning the patient was found to be tachypneic and tachycardic with his face mask off. His symptoms were corrected by replacing his face mask. He claimed he felt anxious about getting more transfusions and that he had breathing problems like this at home in the recent past. The patient requested an aspirin to calm his nerves. Over the course of the day, his Hb level dropped from 6.6 g/dL to 5.9 g/dL, and 2 washed leukopoor PRBCs were ordered.
The first unit was infused uneventfully, but after 125 cc of the second unit, the patient developed respiratory distress, rigors, and hypotension to 70/58 mm Hg despite premedication. He again was treated successfully with increased face mask support. A few rales were noted, but his fluid balance was even. A second transfusion reaction was filed with the blood bank and based on the 2 transfusion-associated events with no other clear explanation for his symptoms, the clinical team favored the TRALI diagnosis. However, the blood bank was suspicious this might not be TRALI as the previous night the patient had 2 episodes of respiratory distress with drops in oxygen saturation unassociated with transfusions. The patient was clinically stable for the remainder of the night.
Early the following morning the patient was scheduled for an esophagogastroduodenoscopy to evaluate for a source of his bleeding. At the beginning of the procedure, a unit of washed leukoreduced PRBCs was hung for a Hb level of 6.9 g/dL. No bleeding source was noted in the stomach, but as the endoscope was passed into the duodenum, and after an infusion of only 25 cc of RBCs, the patient became cyanotic and went into cardiac arrest. Despite advanced resuscitation efforts over 90 minutes, the patient could not be successfully resuscitated and died while in the endoscopy suite. A transfusion reaction workup was initiated but was unremarkable. The transfusion medicine staff was suspicious that something other than TRALI was the cause of the patient’s respiratory distress as he had respiratory distress remote to the transfusions and the unit was prepared correctly before administration. The patient’s family agreed to an autopsy.
Pathology
A full autopsy was performed 22 hours after the patient died. The lungs were congested and of increased weight: The right lung was 800 g, and the left was 750 g. The right lower lobe had a wedge-shaped infarction measuring 6 cm × 5 cm fed by a thrombosed vessel. Multiple small hemorrhagic wedge-shaped areas were noted in the left lung. An ulcer measuring 6 cm × 5 cm was noted just distal to the pylorus. At the base of this ulcer was a 1.5 cm × 0.5 cm tract that communicated with the inferior vena cava (Figure 1).
A postmortem blood culture was positive for Clostridium perfringens (C perfringens) and Candida albicans (C Albicans). Interestingly, one of the collected blood culture vials exploded en route to the laboratory, presumably due to the presence of many gas-forming C perfringens bacteria.
On microscopic examination of the autopsy samples, gram-positive rods were observed in the tissue of multiple organs, including the heart, lungs, liver, and kidneys (Figure 2).
Serology
Fourteen days after the patient’s death, both PRBC units infused during transfusion reactions were positive for granulocyte antibodies by immunofluorescence and agglutination techniques. Human leukocyte antigen antibody testing was also sent but was not found in either the donor or patient.
Discussion
Our case illustrates the unique and challenging diagnosis of DCF given the rarity of presentation and how quickly patients may clinically decompensate. After an extensive search of the medical literature, we were only able to identify about 40 previous cases of DCF, of which 37 were described in one review.1 DCF, although rare, should be considered at risk for forming in the following settings: migrating inferior vena cava filter, right nephrectomy and radiotherapy, duodenal peptic ulcer, abdominal trauma, and oncologic settings involving metastatic malignancy requiring radiation and/or surgical grafting of the inferior vena cava.1-4 When the diagnosis is considered, computed tomography (CT) is the best initial imaging modality as it allows for noninvasive evaluation of both the inferior vena cava and nonadjacent structures. A commonality of our case and those described in the literature is the diagnostic mystery and nonspecific symptoms patients present with, thus making CT an appropriate diagnostic modality. Endoscopy is useful for the further workup of GI bleeding and the diagnosis of peptic ulcer disease.5 In our case, given the patient’s autopsy findings and history of extensive nonsteroidal anti-inflammatory drug use, the duodenal peptic ulcer was likely the precipitating factor for his DCF.
The most challenging aspect in diagnosing DCF is that many times patients present with nonspecific symptoms, and given its rarity it is not something that is usually at the forefront of most differentials.2 This diagnostic difficulty may elucidate why there is such a relatively high mortality rate—nearly 40%—associated with DCF and why many times accurate diagnosis is not made until autopsy.1,3 The most common presenting manifestations are sepsis and/or GI bleeding; in less than half the cases described in the literature patients had both sepsis and GI bleeding. In our case, the patient had signs of melena but was not felt to be septic as his presenting signs were felt to be in the setting of blood loss and dehydration (given his history of bulimia), not an acute infectious source.
In retrospect, one of the more confounding aspects of this case is the clinical picture concerning for TRALI. The patient required supplemental oxygen throughout his hospitalization and decompensated while or after receiving a transfusion, thus having TRALI on the differential was not felt inappropriate at that time. However, this case also illustrates the power of an anchoring bias, and perhaps the clinical team anchored on the diagnosis of TRALI too quickly before considering other possible etiologies for the patient’s respiratory distress. TRALI can be one of the most challenging diagnoses to make in the field of transfusion medicine as there are no definitive diagnostic criteria.6 It is felt to be a clinical diagnosis of exclusion as there is no pathognomonic sign or diagnostic test to confirm it as the cause of the patient’s respiratory distress, though anti–human leukocyte antigen antibodies commonly are present.6,7 Considering how quickly the patient decompensated on day 2 of hospitalization and the presence of C perfringens bacteremia, which
Conclusions
Our investigation reports a case of a DCF in the setting of significant duodenal peptic ulcer disease. We highlight the diagnostic challenge that this commonly lethal etiology presents. We believe ours is the first case in which it was confused for TRALI and associated with food embolism to the lungs causing hypoxic respiratory failure. We want to highlight that DCF, though rare, should be considered for patients who present with GI bleeding and hypoxic respiratory failure.
1. Guillem PG, Binot D, Dupuy-Cuny J, et al. Duodenocaval fistula: a life-threatening condition of various origins. J Vasc Surg. 2001;33(3):643-645. doi:10.1067/mva.2001.111741
2. Ippolito D, Querques G, Drago SG, Bonaffini PA, Sironi S. Duodenocaval fistula in a patient with inferior vena cava leiomyosarcoma treated by surgical resection and caval polytetrafluoroethylene prosthesis. Case Rep Radiol. 2015;2015:1-5. doi:10.1155/2015/575961
3. Guo Y, Zhang YQ, Lin W. Radiological diagnosis of duodenocaval fistula: a case report and literature review. World J Gastroenterol. 2010;16(18):2314-2316. doi:10.3748/wjg.v16.i18.2314
4. Perera GB, Wilson SE, Barie PS, Butler JA. Duodenocaval fistula: A late complication of retroperitoneal irradiation and vena cava replacement. Ann Vasc Surg. 2004;18(1):52-58. doi:10.1007/s10016-003-0097-8
5. Addeo P, Rosso E, Oussoultzoglou E, Jaeck D, Pessaux P, Bachellier P. Inferior vena cava graft-enteric fistula after extended hepatectomy with caval replacement. J Vasc Surg. 2012;55(1):226-229. doi:10.1016/j.jvs.2011.05.118
6. Chapman CE, Stainsby D, Jones H, et al. Ten years of hemovigilance reports of transfusion-related acute lung injury in the United Kingdom and the impact of preferential use of male donor plasma. Transfusion. 2009;49(3):440-452. doi:10.1111/j.1537-2995.2008.01948.x
7. Fontaine MJ, Malone J, Mullins FM, Grumet FC. Diagnosis of transfusion-related acute lung injury: TRALI or not TRALI? Ann Clin Lab Sci. 2006;36(1):53-58.
8. Yang C-C, Hsu P-C, Chang H-J, Cheng C-W, Lee M-H. Clinical significance and outcomes of clostridium perfringens bacteremia—a 10-year experience at a tertiary care hospital. Int J Infect Dis. 2013;17(11):e9of55-e960. doi:10.1016/j.ijid.2013.03.001
A duodenocaval fistula (DCF) is seen when a connection exists between the duodenum and the inferior vena cava. It is a rare entity that is commonly missed and presents a diagnostic challenge due to its nonspecific presenting symptoms.1,2 Patients commonly present with gastrointestinal (GI) bleeding or sepsis. Here we present a case of a 37-year-old man who presented to the hospital for a workup related to melena but went into cardiac arrest prior to an esophagogastroduodenoscopy. Unfortunately, on autopsy, the patient was found to have a DCF. We highlight the diagnostic challenge associated with DCF and how in this case the presentation was confused by a diagnosis of possible transfusion-related acute lung injury (TRALI). To the best of our knowledge, this is also the first description of a case of DCF associated with food embolism to the lungs causing respiratory failure.
Case Presentation
A 37-year-old man with a history significant for bulimia presented to the hospital with a 3-day history of melena and reports of dizziness. The patient did not report being on any prescribed medications but noted that he took 4 aspirin daily to “calm his nerves.” The rest of the patient’s history was unremarkable aside from a reported history of induced emesis 3 to 4 times per week for an extended period up until 2 weeks before admission.
On admission, his vital signs demonstrated tachycardia and orthostatic hypotension. Pertinent findings on physical examination were skin pallor, a normal lung examination, mild epigastric tenderness, and guaiac-positive stools. He was alert and oriented to person, place, and time with no focal deficits. His admission laboratory tests were notable for a hemoglobin (Hb) level of 4.6 g/dL (reference range, 14-17.9), a white blood cell count of 13.5 K/cm (reference range, 4.5-11), an international normalized ratio of 1.21, a blood urea nitrogen of 61 mg/dL (reference range, 10-20), and a creatinine of 2.3 mg/dL (reference range, 0.8-1.4). The patient was placed on 2 L of oxygen via nasal cannula for comfort rather than true hypoxia. A chest X-ray on admission was negative with no signs of infiltrate, edema, or widened mediastinum. An abdominal X-ray was significant for a dilated stomach consistent with bulimia with no abdominal free air or signs of obstruction. The case was discussed with the gastroenterology service who felt that the patient needed to be more hemodynamically stable before pursuing endoscopic evaluation.
He was admitted to the intensive care unit and give a transfusion of 4 units of fresh frozen plasma and 2 packed red blood cells (PRBCs) without any issues. During the infusion of a third PRBC, he developed chills, tachycardia, and hypertension with accompanying respiratory distress characterized by wheezing, decreased breath sounds bilaterally, and a decrease in oxygen saturation to 70% on 2 L supplemental oxygen. He responded to treatment with meperidine, methylprednisolone sodium succinate, albuterol nebulizer, and acetaminophen. A new chest X-ray was read as “development of pulmonary edema vs bilateral pneumonitis.” A transfusion reaction was reported to the blood bank and a diagnosis of TRALI was considered. That evening, he completed a dose of platelets and another PRBC without difficulty after he was premedicated with meperidine, methylprednisolone sodium succinate, and acetaminophen. During the night, the patient spiked a temperature of 40.3 °C that was successfully treated with a cooling blanket and acetaminophen.
The following morning the patient was found to be tachypneic and tachycardic with his face mask off. His symptoms were corrected by replacing his face mask. He claimed he felt anxious about getting more transfusions and that he had breathing problems like this at home in the recent past. The patient requested an aspirin to calm his nerves. Over the course of the day, his Hb level dropped from 6.6 g/dL to 5.9 g/dL, and 2 washed leukopoor PRBCs were ordered.
The first unit was infused uneventfully, but after 125 cc of the second unit, the patient developed respiratory distress, rigors, and hypotension to 70/58 mm Hg despite premedication. He again was treated successfully with increased face mask support. A few rales were noted, but his fluid balance was even. A second transfusion reaction was filed with the blood bank and based on the 2 transfusion-associated events with no other clear explanation for his symptoms, the clinical team favored the TRALI diagnosis. However, the blood bank was suspicious this might not be TRALI as the previous night the patient had 2 episodes of respiratory distress with drops in oxygen saturation unassociated with transfusions. The patient was clinically stable for the remainder of the night.
Early the following morning the patient was scheduled for an esophagogastroduodenoscopy to evaluate for a source of his bleeding. At the beginning of the procedure, a unit of washed leukoreduced PRBCs was hung for a Hb level of 6.9 g/dL. No bleeding source was noted in the stomach, but as the endoscope was passed into the duodenum, and after an infusion of only 25 cc of RBCs, the patient became cyanotic and went into cardiac arrest. Despite advanced resuscitation efforts over 90 minutes, the patient could not be successfully resuscitated and died while in the endoscopy suite. A transfusion reaction workup was initiated but was unremarkable. The transfusion medicine staff was suspicious that something other than TRALI was the cause of the patient’s respiratory distress as he had respiratory distress remote to the transfusions and the unit was prepared correctly before administration. The patient’s family agreed to an autopsy.
Pathology
A full autopsy was performed 22 hours after the patient died. The lungs were congested and of increased weight: The right lung was 800 g, and the left was 750 g. The right lower lobe had a wedge-shaped infarction measuring 6 cm × 5 cm fed by a thrombosed vessel. Multiple small hemorrhagic wedge-shaped areas were noted in the left lung. An ulcer measuring 6 cm × 5 cm was noted just distal to the pylorus. At the base of this ulcer was a 1.5 cm × 0.5 cm tract that communicated with the inferior vena cava (Figure 1).
A postmortem blood culture was positive for Clostridium perfringens (C perfringens) and Candida albicans (C Albicans). Interestingly, one of the collected blood culture vials exploded en route to the laboratory, presumably due to the presence of many gas-forming C perfringens bacteria.
On microscopic examination of the autopsy samples, gram-positive rods were observed in the tissue of multiple organs, including the heart, lungs, liver, and kidneys (Figure 2).
Serology
Fourteen days after the patient’s death, both PRBC units infused during transfusion reactions were positive for granulocyte antibodies by immunofluorescence and agglutination techniques. Human leukocyte antigen antibody testing was also sent but was not found in either the donor or patient.
Discussion
Our case illustrates the unique and challenging diagnosis of DCF given the rarity of presentation and how quickly patients may clinically decompensate. After an extensive search of the medical literature, we were only able to identify about 40 previous cases of DCF, of which 37 were described in one review.1 DCF, although rare, should be considered at risk for forming in the following settings: migrating inferior vena cava filter, right nephrectomy and radiotherapy, duodenal peptic ulcer, abdominal trauma, and oncologic settings involving metastatic malignancy requiring radiation and/or surgical grafting of the inferior vena cava.1-4 When the diagnosis is considered, computed tomography (CT) is the best initial imaging modality as it allows for noninvasive evaluation of both the inferior vena cava and nonadjacent structures. A commonality of our case and those described in the literature is the diagnostic mystery and nonspecific symptoms patients present with, thus making CT an appropriate diagnostic modality. Endoscopy is useful for the further workup of GI bleeding and the diagnosis of peptic ulcer disease.5 In our case, given the patient’s autopsy findings and history of extensive nonsteroidal anti-inflammatory drug use, the duodenal peptic ulcer was likely the precipitating factor for his DCF.
The most challenging aspect in diagnosing DCF is that many times patients present with nonspecific symptoms, and given its rarity it is not something that is usually at the forefront of most differentials.2 This diagnostic difficulty may elucidate why there is such a relatively high mortality rate—nearly 40%—associated with DCF and why many times accurate diagnosis is not made until autopsy.1,3 The most common presenting manifestations are sepsis and/or GI bleeding; in less than half the cases described in the literature patients had both sepsis and GI bleeding. In our case, the patient had signs of melena but was not felt to be septic as his presenting signs were felt to be in the setting of blood loss and dehydration (given his history of bulimia), not an acute infectious source.
In retrospect, one of the more confounding aspects of this case is the clinical picture concerning for TRALI. The patient required supplemental oxygen throughout his hospitalization and decompensated while or after receiving a transfusion, thus having TRALI on the differential was not felt inappropriate at that time. However, this case also illustrates the power of an anchoring bias, and perhaps the clinical team anchored on the diagnosis of TRALI too quickly before considering other possible etiologies for the patient’s respiratory distress. TRALI can be one of the most challenging diagnoses to make in the field of transfusion medicine as there are no definitive diagnostic criteria.6 It is felt to be a clinical diagnosis of exclusion as there is no pathognomonic sign or diagnostic test to confirm it as the cause of the patient’s respiratory distress, though anti–human leukocyte antigen antibodies commonly are present.6,7 Considering how quickly the patient decompensated on day 2 of hospitalization and the presence of C perfringens bacteremia, which
Conclusions
Our investigation reports a case of a DCF in the setting of significant duodenal peptic ulcer disease. We highlight the diagnostic challenge that this commonly lethal etiology presents. We believe ours is the first case in which it was confused for TRALI and associated with food embolism to the lungs causing hypoxic respiratory failure. We want to highlight that DCF, though rare, should be considered for patients who present with GI bleeding and hypoxic respiratory failure.
A duodenocaval fistula (DCF) is seen when a connection exists between the duodenum and the inferior vena cava. It is a rare entity that is commonly missed and presents a diagnostic challenge due to its nonspecific presenting symptoms.1,2 Patients commonly present with gastrointestinal (GI) bleeding or sepsis. Here we present a case of a 37-year-old man who presented to the hospital for a workup related to melena but went into cardiac arrest prior to an esophagogastroduodenoscopy. Unfortunately, on autopsy, the patient was found to have a DCF. We highlight the diagnostic challenge associated with DCF and how in this case the presentation was confused by a diagnosis of possible transfusion-related acute lung injury (TRALI). To the best of our knowledge, this is also the first description of a case of DCF associated with food embolism to the lungs causing respiratory failure.
Case Presentation
A 37-year-old man with a history significant for bulimia presented to the hospital with a 3-day history of melena and reports of dizziness. The patient did not report being on any prescribed medications but noted that he took 4 aspirin daily to “calm his nerves.” The rest of the patient’s history was unremarkable aside from a reported history of induced emesis 3 to 4 times per week for an extended period up until 2 weeks before admission.
On admission, his vital signs demonstrated tachycardia and orthostatic hypotension. Pertinent findings on physical examination were skin pallor, a normal lung examination, mild epigastric tenderness, and guaiac-positive stools. He was alert and oriented to person, place, and time with no focal deficits. His admission laboratory tests were notable for a hemoglobin (Hb) level of 4.6 g/dL (reference range, 14-17.9), a white blood cell count of 13.5 K/cm (reference range, 4.5-11), an international normalized ratio of 1.21, a blood urea nitrogen of 61 mg/dL (reference range, 10-20), and a creatinine of 2.3 mg/dL (reference range, 0.8-1.4). The patient was placed on 2 L of oxygen via nasal cannula for comfort rather than true hypoxia. A chest X-ray on admission was negative with no signs of infiltrate, edema, or widened mediastinum. An abdominal X-ray was significant for a dilated stomach consistent with bulimia with no abdominal free air or signs of obstruction. The case was discussed with the gastroenterology service who felt that the patient needed to be more hemodynamically stable before pursuing endoscopic evaluation.
He was admitted to the intensive care unit and give a transfusion of 4 units of fresh frozen plasma and 2 packed red blood cells (PRBCs) without any issues. During the infusion of a third PRBC, he developed chills, tachycardia, and hypertension with accompanying respiratory distress characterized by wheezing, decreased breath sounds bilaterally, and a decrease in oxygen saturation to 70% on 2 L supplemental oxygen. He responded to treatment with meperidine, methylprednisolone sodium succinate, albuterol nebulizer, and acetaminophen. A new chest X-ray was read as “development of pulmonary edema vs bilateral pneumonitis.” A transfusion reaction was reported to the blood bank and a diagnosis of TRALI was considered. That evening, he completed a dose of platelets and another PRBC without difficulty after he was premedicated with meperidine, methylprednisolone sodium succinate, and acetaminophen. During the night, the patient spiked a temperature of 40.3 °C that was successfully treated with a cooling blanket and acetaminophen.
The following morning the patient was found to be tachypneic and tachycardic with his face mask off. His symptoms were corrected by replacing his face mask. He claimed he felt anxious about getting more transfusions and that he had breathing problems like this at home in the recent past. The patient requested an aspirin to calm his nerves. Over the course of the day, his Hb level dropped from 6.6 g/dL to 5.9 g/dL, and 2 washed leukopoor PRBCs were ordered.
The first unit was infused uneventfully, but after 125 cc of the second unit, the patient developed respiratory distress, rigors, and hypotension to 70/58 mm Hg despite premedication. He again was treated successfully with increased face mask support. A few rales were noted, but his fluid balance was even. A second transfusion reaction was filed with the blood bank and based on the 2 transfusion-associated events with no other clear explanation for his symptoms, the clinical team favored the TRALI diagnosis. However, the blood bank was suspicious this might not be TRALI as the previous night the patient had 2 episodes of respiratory distress with drops in oxygen saturation unassociated with transfusions. The patient was clinically stable for the remainder of the night.
Early the following morning the patient was scheduled for an esophagogastroduodenoscopy to evaluate for a source of his bleeding. At the beginning of the procedure, a unit of washed leukoreduced PRBCs was hung for a Hb level of 6.9 g/dL. No bleeding source was noted in the stomach, but as the endoscope was passed into the duodenum, and after an infusion of only 25 cc of RBCs, the patient became cyanotic and went into cardiac arrest. Despite advanced resuscitation efforts over 90 minutes, the patient could not be successfully resuscitated and died while in the endoscopy suite. A transfusion reaction workup was initiated but was unremarkable. The transfusion medicine staff was suspicious that something other than TRALI was the cause of the patient’s respiratory distress as he had respiratory distress remote to the transfusions and the unit was prepared correctly before administration. The patient’s family agreed to an autopsy.
Pathology
A full autopsy was performed 22 hours after the patient died. The lungs were congested and of increased weight: The right lung was 800 g, and the left was 750 g. The right lower lobe had a wedge-shaped infarction measuring 6 cm × 5 cm fed by a thrombosed vessel. Multiple small hemorrhagic wedge-shaped areas were noted in the left lung. An ulcer measuring 6 cm × 5 cm was noted just distal to the pylorus. At the base of this ulcer was a 1.5 cm × 0.5 cm tract that communicated with the inferior vena cava (Figure 1).
A postmortem blood culture was positive for Clostridium perfringens (C perfringens) and Candida albicans (C Albicans). Interestingly, one of the collected blood culture vials exploded en route to the laboratory, presumably due to the presence of many gas-forming C perfringens bacteria.
On microscopic examination of the autopsy samples, gram-positive rods were observed in the tissue of multiple organs, including the heart, lungs, liver, and kidneys (Figure 2).
Serology
Fourteen days after the patient’s death, both PRBC units infused during transfusion reactions were positive for granulocyte antibodies by immunofluorescence and agglutination techniques. Human leukocyte antigen antibody testing was also sent but was not found in either the donor or patient.
Discussion
Our case illustrates the unique and challenging diagnosis of DCF given the rarity of presentation and how quickly patients may clinically decompensate. After an extensive search of the medical literature, we were only able to identify about 40 previous cases of DCF, of which 37 were described in one review.1 DCF, although rare, should be considered at risk for forming in the following settings: migrating inferior vena cava filter, right nephrectomy and radiotherapy, duodenal peptic ulcer, abdominal trauma, and oncologic settings involving metastatic malignancy requiring radiation and/or surgical grafting of the inferior vena cava.1-4 When the diagnosis is considered, computed tomography (CT) is the best initial imaging modality as it allows for noninvasive evaluation of both the inferior vena cava and nonadjacent structures. A commonality of our case and those described in the literature is the diagnostic mystery and nonspecific symptoms patients present with, thus making CT an appropriate diagnostic modality. Endoscopy is useful for the further workup of GI bleeding and the diagnosis of peptic ulcer disease.5 In our case, given the patient’s autopsy findings and history of extensive nonsteroidal anti-inflammatory drug use, the duodenal peptic ulcer was likely the precipitating factor for his DCF.
The most challenging aspect in diagnosing DCF is that many times patients present with nonspecific symptoms, and given its rarity it is not something that is usually at the forefront of most differentials.2 This diagnostic difficulty may elucidate why there is such a relatively high mortality rate—nearly 40%—associated with DCF and why many times accurate diagnosis is not made until autopsy.1,3 The most common presenting manifestations are sepsis and/or GI bleeding; in less than half the cases described in the literature patients had both sepsis and GI bleeding. In our case, the patient had signs of melena but was not felt to be septic as his presenting signs were felt to be in the setting of blood loss and dehydration (given his history of bulimia), not an acute infectious source.
In retrospect, one of the more confounding aspects of this case is the clinical picture concerning for TRALI. The patient required supplemental oxygen throughout his hospitalization and decompensated while or after receiving a transfusion, thus having TRALI on the differential was not felt inappropriate at that time. However, this case also illustrates the power of an anchoring bias, and perhaps the clinical team anchored on the diagnosis of TRALI too quickly before considering other possible etiologies for the patient’s respiratory distress. TRALI can be one of the most challenging diagnoses to make in the field of transfusion medicine as there are no definitive diagnostic criteria.6 It is felt to be a clinical diagnosis of exclusion as there is no pathognomonic sign or diagnostic test to confirm it as the cause of the patient’s respiratory distress, though anti–human leukocyte antigen antibodies commonly are present.6,7 Considering how quickly the patient decompensated on day 2 of hospitalization and the presence of C perfringens bacteremia, which
Conclusions
Our investigation reports a case of a DCF in the setting of significant duodenal peptic ulcer disease. We highlight the diagnostic challenge that this commonly lethal etiology presents. We believe ours is the first case in which it was confused for TRALI and associated with food embolism to the lungs causing hypoxic respiratory failure. We want to highlight that DCF, though rare, should be considered for patients who present with GI bleeding and hypoxic respiratory failure.
1. Guillem PG, Binot D, Dupuy-Cuny J, et al. Duodenocaval fistula: a life-threatening condition of various origins. J Vasc Surg. 2001;33(3):643-645. doi:10.1067/mva.2001.111741
2. Ippolito D, Querques G, Drago SG, Bonaffini PA, Sironi S. Duodenocaval fistula in a patient with inferior vena cava leiomyosarcoma treated by surgical resection and caval polytetrafluoroethylene prosthesis. Case Rep Radiol. 2015;2015:1-5. doi:10.1155/2015/575961
3. Guo Y, Zhang YQ, Lin W. Radiological diagnosis of duodenocaval fistula: a case report and literature review. World J Gastroenterol. 2010;16(18):2314-2316. doi:10.3748/wjg.v16.i18.2314
4. Perera GB, Wilson SE, Barie PS, Butler JA. Duodenocaval fistula: A late complication of retroperitoneal irradiation and vena cava replacement. Ann Vasc Surg. 2004;18(1):52-58. doi:10.1007/s10016-003-0097-8
5. Addeo P, Rosso E, Oussoultzoglou E, Jaeck D, Pessaux P, Bachellier P. Inferior vena cava graft-enteric fistula after extended hepatectomy with caval replacement. J Vasc Surg. 2012;55(1):226-229. doi:10.1016/j.jvs.2011.05.118
6. Chapman CE, Stainsby D, Jones H, et al. Ten years of hemovigilance reports of transfusion-related acute lung injury in the United Kingdom and the impact of preferential use of male donor plasma. Transfusion. 2009;49(3):440-452. doi:10.1111/j.1537-2995.2008.01948.x
7. Fontaine MJ, Malone J, Mullins FM, Grumet FC. Diagnosis of transfusion-related acute lung injury: TRALI or not TRALI? Ann Clin Lab Sci. 2006;36(1):53-58.
8. Yang C-C, Hsu P-C, Chang H-J, Cheng C-W, Lee M-H. Clinical significance and outcomes of clostridium perfringens bacteremia—a 10-year experience at a tertiary care hospital. Int J Infect Dis. 2013;17(11):e9of55-e960. doi:10.1016/j.ijid.2013.03.001
1. Guillem PG, Binot D, Dupuy-Cuny J, et al. Duodenocaval fistula: a life-threatening condition of various origins. J Vasc Surg. 2001;33(3):643-645. doi:10.1067/mva.2001.111741
2. Ippolito D, Querques G, Drago SG, Bonaffini PA, Sironi S. Duodenocaval fistula in a patient with inferior vena cava leiomyosarcoma treated by surgical resection and caval polytetrafluoroethylene prosthesis. Case Rep Radiol. 2015;2015:1-5. doi:10.1155/2015/575961
3. Guo Y, Zhang YQ, Lin W. Radiological diagnosis of duodenocaval fistula: a case report and literature review. World J Gastroenterol. 2010;16(18):2314-2316. doi:10.3748/wjg.v16.i18.2314
4. Perera GB, Wilson SE, Barie PS, Butler JA. Duodenocaval fistula: A late complication of retroperitoneal irradiation and vena cava replacement. Ann Vasc Surg. 2004;18(1):52-58. doi:10.1007/s10016-003-0097-8
5. Addeo P, Rosso E, Oussoultzoglou E, Jaeck D, Pessaux P, Bachellier P. Inferior vena cava graft-enteric fistula after extended hepatectomy with caval replacement. J Vasc Surg. 2012;55(1):226-229. doi:10.1016/j.jvs.2011.05.118
6. Chapman CE, Stainsby D, Jones H, et al. Ten years of hemovigilance reports of transfusion-related acute lung injury in the United Kingdom and the impact of preferential use of male donor plasma. Transfusion. 2009;49(3):440-452. doi:10.1111/j.1537-2995.2008.01948.x
7. Fontaine MJ, Malone J, Mullins FM, Grumet FC. Diagnosis of transfusion-related acute lung injury: TRALI or not TRALI? Ann Clin Lab Sci. 2006;36(1):53-58.
8. Yang C-C, Hsu P-C, Chang H-J, Cheng C-W, Lee M-H. Clinical significance and outcomes of clostridium perfringens bacteremia—a 10-year experience at a tertiary care hospital. Int J Infect Dis. 2013;17(11):e9of55-e960. doi:10.1016/j.ijid.2013.03.001