User login
Furosemide seen as safe for preventing newborn lung disease
A medication used to reduce fluid retention can also safely be used to prevent a dangerous lung condition that affects newborns, particularly those born premature, according to a new study.
Furosemide (Lasix) – which can reduce excess fluid in the body caused by heart failure, liver disease, and kidney trouble – is commonly used off-label to prevent bronchopulmonary dysplasia (BPD), a disorder that causes irritation and poor development of lungs in premature infants. But until now, researchers have not studied its safety in this setting.
BPD often affects babies born more than 2 months early and can sometimes result in breathing difficulties into adolescence and young adulthood.
“There are so few drugs that have been tested for newborns, and there are very little data to help neonatologists decide if certain medications are safe and effective,” said Rachel Greenberg, MD, MHS, a neonatologist and member of the Duke Clinical Research Institute, Durham, N.C. “We found there was no greater risk of safety events for newborns given furosemide.”
Dr. Greenberg presented the findings at the 2022 Pediatric Academic Societies meeting in Denver.
For the 28-day randomized controlled trial, Dr. Greenberg and colleagues enrolled 80 preterm newborns, born at less than 29 weeks’ gestation, at 17 centers within the Eunice Kennedy Shriver National Institute of Child Health and Human Development Pediatric Trials Network. Of those, 61 received furosemide and 19 received a placebo.
Although babies given furosemide had more problems with electrolytes – an expected outcome from the use of diuretic medications – the researchers observed no greater risk for more serious issues, namely hearing loss or kidney stones, Dr. Greenberg told this news organization.
“The mechanism here is we know that extra fluid can damage the lungs and can cause you to have to use more respiratory support and more oxygen,” she said. “The thought from a physiological standpoint is using a diuretic can decrease fluid in the lungs and lead to improvements in lung outcomes.”
The researchers did not observe a reduction in BDP or death in babies who received furosemide, but Dr. Greenberg said the study was underpowered to detect such an effect.
“We were not powered to detect a difference in that outcome; the overall objective of this study was always to evaluate safety,” she said. “Of course, we wanted to capture variables that would measure effectiveness as well.
“Because this was a pragmatic trial, we did not limit the amount of fluids that the clinicians could give the participating infants. This could have impacted the effectiveness of furosemide. We would need a different design and larger study to truly determine effectiveness.”
Dr. Greenberg said she hoped the new data will provide greater insight to neonatal providers and help bolster future, more large-scale trials using furosemide in premature infants.
The drug has previously been associated with both kidney stones and ototoxicity, which occurs when medication causes a person to develop hearing or balance problems, said Nicolas Bamat, MD, MSCE, assistant professor of pediatrics at the Perelman School of Medicine, University of Pennsylvania, Philadelphia.
Although the number of children in the latest study was too small to generate any firm conclusions, he said, the trial provides the best data to date on furosemide in premature infants.
The medication is used frequently both on babies at risk of developing BPD and babies who have already reached BPD status. Among newborns with highest risk of dying, furosemide is indeed the “most frequently used pharmacotherapy,” Dr. Bamat said.
“What’s worth noting is that furosemide is an old medication that has been used extensively in the neonatal populations for 40 years, and that is occurring in the absence of data,” Dr. Bamat added. “This is a very important step forward.”
Dr. Greenberg and Dr. Bamat have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A medication used to reduce fluid retention can also safely be used to prevent a dangerous lung condition that affects newborns, particularly those born premature, according to a new study.
Furosemide (Lasix) – which can reduce excess fluid in the body caused by heart failure, liver disease, and kidney trouble – is commonly used off-label to prevent bronchopulmonary dysplasia (BPD), a disorder that causes irritation and poor development of lungs in premature infants. But until now, researchers have not studied its safety in this setting.
BPD often affects babies born more than 2 months early and can sometimes result in breathing difficulties into adolescence and young adulthood.
“There are so few drugs that have been tested for newborns, and there are very little data to help neonatologists decide if certain medications are safe and effective,” said Rachel Greenberg, MD, MHS, a neonatologist and member of the Duke Clinical Research Institute, Durham, N.C. “We found there was no greater risk of safety events for newborns given furosemide.”
Dr. Greenberg presented the findings at the 2022 Pediatric Academic Societies meeting in Denver.
For the 28-day randomized controlled trial, Dr. Greenberg and colleagues enrolled 80 preterm newborns, born at less than 29 weeks’ gestation, at 17 centers within the Eunice Kennedy Shriver National Institute of Child Health and Human Development Pediatric Trials Network. Of those, 61 received furosemide and 19 received a placebo.
Although babies given furosemide had more problems with electrolytes – an expected outcome from the use of diuretic medications – the researchers observed no greater risk for more serious issues, namely hearing loss or kidney stones, Dr. Greenberg told this news organization.
“The mechanism here is we know that extra fluid can damage the lungs and can cause you to have to use more respiratory support and more oxygen,” she said. “The thought from a physiological standpoint is using a diuretic can decrease fluid in the lungs and lead to improvements in lung outcomes.”
The researchers did not observe a reduction in BDP or death in babies who received furosemide, but Dr. Greenberg said the study was underpowered to detect such an effect.
“We were not powered to detect a difference in that outcome; the overall objective of this study was always to evaluate safety,” she said. “Of course, we wanted to capture variables that would measure effectiveness as well.
“Because this was a pragmatic trial, we did not limit the amount of fluids that the clinicians could give the participating infants. This could have impacted the effectiveness of furosemide. We would need a different design and larger study to truly determine effectiveness.”
Dr. Greenberg said she hoped the new data will provide greater insight to neonatal providers and help bolster future, more large-scale trials using furosemide in premature infants.
The drug has previously been associated with both kidney stones and ototoxicity, which occurs when medication causes a person to develop hearing or balance problems, said Nicolas Bamat, MD, MSCE, assistant professor of pediatrics at the Perelman School of Medicine, University of Pennsylvania, Philadelphia.
Although the number of children in the latest study was too small to generate any firm conclusions, he said, the trial provides the best data to date on furosemide in premature infants.
The medication is used frequently both on babies at risk of developing BPD and babies who have already reached BPD status. Among newborns with highest risk of dying, furosemide is indeed the “most frequently used pharmacotherapy,” Dr. Bamat said.
“What’s worth noting is that furosemide is an old medication that has been used extensively in the neonatal populations for 40 years, and that is occurring in the absence of data,” Dr. Bamat added. “This is a very important step forward.”
Dr. Greenberg and Dr. Bamat have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A medication used to reduce fluid retention can also safely be used to prevent a dangerous lung condition that affects newborns, particularly those born premature, according to a new study.
Furosemide (Lasix) – which can reduce excess fluid in the body caused by heart failure, liver disease, and kidney trouble – is commonly used off-label to prevent bronchopulmonary dysplasia (BPD), a disorder that causes irritation and poor development of lungs in premature infants. But until now, researchers have not studied its safety in this setting.
BPD often affects babies born more than 2 months early and can sometimes result in breathing difficulties into adolescence and young adulthood.
“There are so few drugs that have been tested for newborns, and there are very little data to help neonatologists decide if certain medications are safe and effective,” said Rachel Greenberg, MD, MHS, a neonatologist and member of the Duke Clinical Research Institute, Durham, N.C. “We found there was no greater risk of safety events for newborns given furosemide.”
Dr. Greenberg presented the findings at the 2022 Pediatric Academic Societies meeting in Denver.
For the 28-day randomized controlled trial, Dr. Greenberg and colleagues enrolled 80 preterm newborns, born at less than 29 weeks’ gestation, at 17 centers within the Eunice Kennedy Shriver National Institute of Child Health and Human Development Pediatric Trials Network. Of those, 61 received furosemide and 19 received a placebo.
Although babies given furosemide had more problems with electrolytes – an expected outcome from the use of diuretic medications – the researchers observed no greater risk for more serious issues, namely hearing loss or kidney stones, Dr. Greenberg told this news organization.
“The mechanism here is we know that extra fluid can damage the lungs and can cause you to have to use more respiratory support and more oxygen,” she said. “The thought from a physiological standpoint is using a diuretic can decrease fluid in the lungs and lead to improvements in lung outcomes.”
The researchers did not observe a reduction in BDP or death in babies who received furosemide, but Dr. Greenberg said the study was underpowered to detect such an effect.
“We were not powered to detect a difference in that outcome; the overall objective of this study was always to evaluate safety,” she said. “Of course, we wanted to capture variables that would measure effectiveness as well.
“Because this was a pragmatic trial, we did not limit the amount of fluids that the clinicians could give the participating infants. This could have impacted the effectiveness of furosemide. We would need a different design and larger study to truly determine effectiveness.”
Dr. Greenberg said she hoped the new data will provide greater insight to neonatal providers and help bolster future, more large-scale trials using furosemide in premature infants.
The drug has previously been associated with both kidney stones and ototoxicity, which occurs when medication causes a person to develop hearing or balance problems, said Nicolas Bamat, MD, MSCE, assistant professor of pediatrics at the Perelman School of Medicine, University of Pennsylvania, Philadelphia.
Although the number of children in the latest study was too small to generate any firm conclusions, he said, the trial provides the best data to date on furosemide in premature infants.
The medication is used frequently both on babies at risk of developing BPD and babies who have already reached BPD status. Among newborns with highest risk of dying, furosemide is indeed the “most frequently used pharmacotherapy,” Dr. Bamat said.
“What’s worth noting is that furosemide is an old medication that has been used extensively in the neonatal populations for 40 years, and that is occurring in the absence of data,” Dr. Bamat added. “This is a very important step forward.”
Dr. Greenberg and Dr. Bamat have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM PAS 2022
Nighttime asthma predicts poor outcomes in teens
Teens with persistent nocturnal asthma symptoms were significantly more likely than were those without nighttime asthma to report poor functional health independent of daytime asthma, based on data from 430 adolescents aged 12-16 years.
Approximately half of children with severe asthma experience at least one night of inadequate sleep per week, and lost sleep among young children with asthma has been associated with impaired physical function, school absence, and worsened mood. However, the effect of asthma-related sleep disruption on daily function in teenagers in particular has not been well studied, according to Anne Zhang of the University of Rochester (N.Y.) and colleagues.
In a poster presented at the virtual meeting of the Pediatric Academic Societies (#542), the researchers reviewed baseline survey data from the School-Based Asthma Care for Teens (SB-ACT) study, a randomized, controlled trial conducted from 2014 to 2018 in Rochester, N.Y.
The average age of the respondents was 13.4 years, 56% were male, 56% were African American, 32% were Hispanic, and 84% had Medicaid insurance.
Persistent nocturnal asthma was defined as 2 or more nights of nighttime awakening in the past 14 days, and intermittent nocturnal asthma was defined as less than 2 nights of nighttime awakening in the past 14 days.
Overall, teens with persistent nocturnal asthma were significantly more likely than were those with intermittent nocturnal asthma to report physical limitations during strenuous activity (58% vs. 41%), moderate activity (32% vs. 19%), and school gym classes (36% vs. 19%; P <.01 for all).
In addition to physical impact, teens with persistent nocturnal asthma were more likely than were those with intermittent nocturnal asthma to report depressive symptoms (41% vs. 23%), asthma-related school absences in the past 14 days (0.81 vs. 0.12), and poorer quality of life (4.6 vs. 5.9, P <.01 for all).
The results remained significant in a multivariate analysis that controlled for daytime asthma symptoms, weight status, race, ethnicity, gender, age, and smoke exposure, the researchers said.
The study findings were limited by several factors including the cross-sectional design, potential of recall bias in survey responses, and lack of data on sleep duration and quality, the researchers noted.
However, the results suggest that improving nighttime asthma control for teens may improve daily function, and providers should ask teens with asthma about the possible effect and burden of nighttime symptoms, they said. Potential strategies to improve persistent nocturnal asthma symptoms include adjusting the timing of medications or physical activity, they added.
“We know that getting adequate, high-quality sleep is important for health - especially for adolescents,” said Kelly A. Curran, MD, of the University of Oklahoma Health Sciences Center, in an interview. “Just like adults, tired teens are not able to function at their best and are at higher risk of developing mood problems,” she said.
However, “There are already so many barriers for teens getting good sleep, such as screen time/social media, homework, busy social calendars, caffeine use, and early morning school start times,” she said. Underlying medical conditions such as depression, anxiety, and obstructive sleep apnea also can contribute to poor sleep for teens, she added.
“In my practice, I frequently counsel about sleep hygiene because it is so essential and not commonly followed,” said Dr. Curran. “Nocturnal asthma is another contributor to poor sleep - not one that I have been regularly screening for - and something we can potentially intervene in to help improve health and quality of life,” she emphasized.
Dr. Curran said that she was not surprised by the study findings, given what is known about the importance of sleep. In clinical practice, “Teens who have asthma should be screened for nocturnal symptoms as these are linked to worsened quality of life, including limitations in activities, depressive symptoms, and asthma-related school absence,” she said.
However, additional research is needed to better understand whether improving nocturnal asthma symptoms can help improve quality of life and daily functioning in adolescents, she noted.
The SB-ACT was supported by the National Institutes of Health. Ms. Zhang was supported in part by the OME-CACHED for medical student research and an NIH grant. The researchers had no financial conflicts to disclose. Dr. Curran had no financial conflicts to disclose.
*This story was updated on May 5. 2021.
Teens with persistent nocturnal asthma symptoms were significantly more likely than were those without nighttime asthma to report poor functional health independent of daytime asthma, based on data from 430 adolescents aged 12-16 years.
Approximately half of children with severe asthma experience at least one night of inadequate sleep per week, and lost sleep among young children with asthma has been associated with impaired physical function, school absence, and worsened mood. However, the effect of asthma-related sleep disruption on daily function in teenagers in particular has not been well studied, according to Anne Zhang of the University of Rochester (N.Y.) and colleagues.
In a poster presented at the virtual meeting of the Pediatric Academic Societies (#542), the researchers reviewed baseline survey data from the School-Based Asthma Care for Teens (SB-ACT) study, a randomized, controlled trial conducted from 2014 to 2018 in Rochester, N.Y.
The average age of the respondents was 13.4 years, 56% were male, 56% were African American, 32% were Hispanic, and 84% had Medicaid insurance.
Persistent nocturnal asthma was defined as 2 or more nights of nighttime awakening in the past 14 days, and intermittent nocturnal asthma was defined as less than 2 nights of nighttime awakening in the past 14 days.
Overall, teens with persistent nocturnal asthma were significantly more likely than were those with intermittent nocturnal asthma to report physical limitations during strenuous activity (58% vs. 41%), moderate activity (32% vs. 19%), and school gym classes (36% vs. 19%; P <.01 for all).
In addition to physical impact, teens with persistent nocturnal asthma were more likely than were those with intermittent nocturnal asthma to report depressive symptoms (41% vs. 23%), asthma-related school absences in the past 14 days (0.81 vs. 0.12), and poorer quality of life (4.6 vs. 5.9, P <.01 for all).
The results remained significant in a multivariate analysis that controlled for daytime asthma symptoms, weight status, race, ethnicity, gender, age, and smoke exposure, the researchers said.
The study findings were limited by several factors including the cross-sectional design, potential of recall bias in survey responses, and lack of data on sleep duration and quality, the researchers noted.
However, the results suggest that improving nighttime asthma control for teens may improve daily function, and providers should ask teens with asthma about the possible effect and burden of nighttime symptoms, they said. Potential strategies to improve persistent nocturnal asthma symptoms include adjusting the timing of medications or physical activity, they added.
“We know that getting adequate, high-quality sleep is important for health - especially for adolescents,” said Kelly A. Curran, MD, of the University of Oklahoma Health Sciences Center, in an interview. “Just like adults, tired teens are not able to function at their best and are at higher risk of developing mood problems,” she said.
However, “There are already so many barriers for teens getting good sleep, such as screen time/social media, homework, busy social calendars, caffeine use, and early morning school start times,” she said. Underlying medical conditions such as depression, anxiety, and obstructive sleep apnea also can contribute to poor sleep for teens, she added.
“In my practice, I frequently counsel about sleep hygiene because it is so essential and not commonly followed,” said Dr. Curran. “Nocturnal asthma is another contributor to poor sleep - not one that I have been regularly screening for - and something we can potentially intervene in to help improve health and quality of life,” she emphasized.
Dr. Curran said that she was not surprised by the study findings, given what is known about the importance of sleep. In clinical practice, “Teens who have asthma should be screened for nocturnal symptoms as these are linked to worsened quality of life, including limitations in activities, depressive symptoms, and asthma-related school absence,” she said.
However, additional research is needed to better understand whether improving nocturnal asthma symptoms can help improve quality of life and daily functioning in adolescents, she noted.
The SB-ACT was supported by the National Institutes of Health. Ms. Zhang was supported in part by the OME-CACHED for medical student research and an NIH grant. The researchers had no financial conflicts to disclose. Dr. Curran had no financial conflicts to disclose.
*This story was updated on May 5. 2021.
Teens with persistent nocturnal asthma symptoms were significantly more likely than were those without nighttime asthma to report poor functional health independent of daytime asthma, based on data from 430 adolescents aged 12-16 years.
Approximately half of children with severe asthma experience at least one night of inadequate sleep per week, and lost sleep among young children with asthma has been associated with impaired physical function, school absence, and worsened mood. However, the effect of asthma-related sleep disruption on daily function in teenagers in particular has not been well studied, according to Anne Zhang of the University of Rochester (N.Y.) and colleagues.
In a poster presented at the virtual meeting of the Pediatric Academic Societies (#542), the researchers reviewed baseline survey data from the School-Based Asthma Care for Teens (SB-ACT) study, a randomized, controlled trial conducted from 2014 to 2018 in Rochester, N.Y.
The average age of the respondents was 13.4 years, 56% were male, 56% were African American, 32% were Hispanic, and 84% had Medicaid insurance.
Persistent nocturnal asthma was defined as 2 or more nights of nighttime awakening in the past 14 days, and intermittent nocturnal asthma was defined as less than 2 nights of nighttime awakening in the past 14 days.
Overall, teens with persistent nocturnal asthma were significantly more likely than were those with intermittent nocturnal asthma to report physical limitations during strenuous activity (58% vs. 41%), moderate activity (32% vs. 19%), and school gym classes (36% vs. 19%; P <.01 for all).
In addition to physical impact, teens with persistent nocturnal asthma were more likely than were those with intermittent nocturnal asthma to report depressive symptoms (41% vs. 23%), asthma-related school absences in the past 14 days (0.81 vs. 0.12), and poorer quality of life (4.6 vs. 5.9, P <.01 for all).
The results remained significant in a multivariate analysis that controlled for daytime asthma symptoms, weight status, race, ethnicity, gender, age, and smoke exposure, the researchers said.
The study findings were limited by several factors including the cross-sectional design, potential of recall bias in survey responses, and lack of data on sleep duration and quality, the researchers noted.
However, the results suggest that improving nighttime asthma control for teens may improve daily function, and providers should ask teens with asthma about the possible effect and burden of nighttime symptoms, they said. Potential strategies to improve persistent nocturnal asthma symptoms include adjusting the timing of medications or physical activity, they added.
“We know that getting adequate, high-quality sleep is important for health - especially for adolescents,” said Kelly A. Curran, MD, of the University of Oklahoma Health Sciences Center, in an interview. “Just like adults, tired teens are not able to function at their best and are at higher risk of developing mood problems,” she said.
However, “There are already so many barriers for teens getting good sleep, such as screen time/social media, homework, busy social calendars, caffeine use, and early morning school start times,” she said. Underlying medical conditions such as depression, anxiety, and obstructive sleep apnea also can contribute to poor sleep for teens, she added.
“In my practice, I frequently counsel about sleep hygiene because it is so essential and not commonly followed,” said Dr. Curran. “Nocturnal asthma is another contributor to poor sleep - not one that I have been regularly screening for - and something we can potentially intervene in to help improve health and quality of life,” she emphasized.
Dr. Curran said that she was not surprised by the study findings, given what is known about the importance of sleep. In clinical practice, “Teens who have asthma should be screened for nocturnal symptoms as these are linked to worsened quality of life, including limitations in activities, depressive symptoms, and asthma-related school absence,” she said.
However, additional research is needed to better understand whether improving nocturnal asthma symptoms can help improve quality of life and daily functioning in adolescents, she noted.
The SB-ACT was supported by the National Institutes of Health. Ms. Zhang was supported in part by the OME-CACHED for medical student research and an NIH grant. The researchers had no financial conflicts to disclose. Dr. Curran had no financial conflicts to disclose.
*This story was updated on May 5. 2021.
FROM PAS 2021
Short-term oxygen prescriptions lead to inappropriate long-term use
In past posts for this news organization, I’ve railed against the cost of inappropriate prescriptions for oxygen. A recent review recommended against prescribing oxygen for patients with isolated exertional or nocturnal desaturations, and recently published randomized trials found no demonstrable benefit to oxygen use in the absence of resting hypoxemia. a common practice in clinics where I’ve worked. However, oxygen prescriptions at hospital discharge are a far more pernicious cause of wasted resources.
Prescriptions at hospital discharge, sometimes referred to as short-term oxygen therapy (STOT), account for a large proportion of total oxygen use. Past data have shown that the term “STOT” is a misnomer, as most patients provided with oxygen at discharge are never reevaluated and become long-term oxygen users. The high cost of durable medical equipment related to oxygen delivery prompted the American Thoracic Society and American College of Chest Physicians to recommend postdischarge reassessment of oxygen needs in their Choosing Wisely campaign for adult pulmonary medicine.
A recent study published in the Annals of the American Thoracic Society (Ann ATS) highlights the benefits available if we decide to “choose wisely.” The authors studied patients covered by Veterans Affairs and discharged on STOT between 2006 and 2011. Only 43.6% (287/659) had complete reassessment (oxygen testing at rest and with ambulation) within 90 days. Of those, 124 (43.2%) were eligible for discontinuation via Centers for Medicare & Medicaid Services guidelines. A total of 70.7% (466/659) were tested at rest, and only 15.7% (73/466) had resting hypoxemia. If one accepts the results of the recently published Long-Term Oxygen Treatment Trial, this means that 84.3% (393/466) would be eligible for oxygen discontinuation.
The Ann ATS study provides a blueprint for how we might improve these dismal numbers. There were five separate sites reviewed in their paper. At one site, reassessment occurred in 78.5% of STOT patients and 100% had oxygen discontinued when appropriate. What was their secret? An automatic alert system and a dedicated clinic, coordinator, and respiratory therapist. Also, among the 124 patients who had a full reassessment and no longer qualified for oxygen, 86.3% had it discontinued.
There are countless reasons why STOT is common, but discontinuation is not. Most COPD exacerbations are managed by nonpulmonologists on general medicine wards prior to discharge. In my experience, these physicians are reluctant to release a patient with exertional hypoxia without STOT. They also assume that the pulmonary clinic will do its job during the obligatory outpatient follow-up appointment they schedule with us. At the follow-up, the patient and physician are reluctant to stop therapy because of psychological dependence and therapeutic overconfidence, respectively.
In summary, STOT following hospitalization comprises the majority of all oxygen prescriptions. Historically, the United States provides far more oxygen than other developed countries, and only CMS reimbursement changes have bent the “overprescription” curve. The Ann ATS study shows that a well-designed program at the hospital level can put oxygen decisions back in the hands of providers.
Let’s “choose wisely” and follow what works, or we’ll have only ourselves to blame when reimbursement decisions are taken out of our hands.
A version of this article first appeared on Medscape.com.
In past posts for this news organization, I’ve railed against the cost of inappropriate prescriptions for oxygen. A recent review recommended against prescribing oxygen for patients with isolated exertional or nocturnal desaturations, and recently published randomized trials found no demonstrable benefit to oxygen use in the absence of resting hypoxemia. a common practice in clinics where I’ve worked. However, oxygen prescriptions at hospital discharge are a far more pernicious cause of wasted resources.
Prescriptions at hospital discharge, sometimes referred to as short-term oxygen therapy (STOT), account for a large proportion of total oxygen use. Past data have shown that the term “STOT” is a misnomer, as most patients provided with oxygen at discharge are never reevaluated and become long-term oxygen users. The high cost of durable medical equipment related to oxygen delivery prompted the American Thoracic Society and American College of Chest Physicians to recommend postdischarge reassessment of oxygen needs in their Choosing Wisely campaign for adult pulmonary medicine.
A recent study published in the Annals of the American Thoracic Society (Ann ATS) highlights the benefits available if we decide to “choose wisely.” The authors studied patients covered by Veterans Affairs and discharged on STOT between 2006 and 2011. Only 43.6% (287/659) had complete reassessment (oxygen testing at rest and with ambulation) within 90 days. Of those, 124 (43.2%) were eligible for discontinuation via Centers for Medicare & Medicaid Services guidelines. A total of 70.7% (466/659) were tested at rest, and only 15.7% (73/466) had resting hypoxemia. If one accepts the results of the recently published Long-Term Oxygen Treatment Trial, this means that 84.3% (393/466) would be eligible for oxygen discontinuation.
The Ann ATS study provides a blueprint for how we might improve these dismal numbers. There were five separate sites reviewed in their paper. At one site, reassessment occurred in 78.5% of STOT patients and 100% had oxygen discontinued when appropriate. What was their secret? An automatic alert system and a dedicated clinic, coordinator, and respiratory therapist. Also, among the 124 patients who had a full reassessment and no longer qualified for oxygen, 86.3% had it discontinued.
There are countless reasons why STOT is common, but discontinuation is not. Most COPD exacerbations are managed by nonpulmonologists on general medicine wards prior to discharge. In my experience, these physicians are reluctant to release a patient with exertional hypoxia without STOT. They also assume that the pulmonary clinic will do its job during the obligatory outpatient follow-up appointment they schedule with us. At the follow-up, the patient and physician are reluctant to stop therapy because of psychological dependence and therapeutic overconfidence, respectively.
In summary, STOT following hospitalization comprises the majority of all oxygen prescriptions. Historically, the United States provides far more oxygen than other developed countries, and only CMS reimbursement changes have bent the “overprescription” curve. The Ann ATS study shows that a well-designed program at the hospital level can put oxygen decisions back in the hands of providers.
Let’s “choose wisely” and follow what works, or we’ll have only ourselves to blame when reimbursement decisions are taken out of our hands.
A version of this article first appeared on Medscape.com.
In past posts for this news organization, I’ve railed against the cost of inappropriate prescriptions for oxygen. A recent review recommended against prescribing oxygen for patients with isolated exertional or nocturnal desaturations, and recently published randomized trials found no demonstrable benefit to oxygen use in the absence of resting hypoxemia. a common practice in clinics where I’ve worked. However, oxygen prescriptions at hospital discharge are a far more pernicious cause of wasted resources.
Prescriptions at hospital discharge, sometimes referred to as short-term oxygen therapy (STOT), account for a large proportion of total oxygen use. Past data have shown that the term “STOT” is a misnomer, as most patients provided with oxygen at discharge are never reevaluated and become long-term oxygen users. The high cost of durable medical equipment related to oxygen delivery prompted the American Thoracic Society and American College of Chest Physicians to recommend postdischarge reassessment of oxygen needs in their Choosing Wisely campaign for adult pulmonary medicine.
A recent study published in the Annals of the American Thoracic Society (Ann ATS) highlights the benefits available if we decide to “choose wisely.” The authors studied patients covered by Veterans Affairs and discharged on STOT between 2006 and 2011. Only 43.6% (287/659) had complete reassessment (oxygen testing at rest and with ambulation) within 90 days. Of those, 124 (43.2%) were eligible for discontinuation via Centers for Medicare & Medicaid Services guidelines. A total of 70.7% (466/659) were tested at rest, and only 15.7% (73/466) had resting hypoxemia. If one accepts the results of the recently published Long-Term Oxygen Treatment Trial, this means that 84.3% (393/466) would be eligible for oxygen discontinuation.
The Ann ATS study provides a blueprint for how we might improve these dismal numbers. There were five separate sites reviewed in their paper. At one site, reassessment occurred in 78.5% of STOT patients and 100% had oxygen discontinued when appropriate. What was their secret? An automatic alert system and a dedicated clinic, coordinator, and respiratory therapist. Also, among the 124 patients who had a full reassessment and no longer qualified for oxygen, 86.3% had it discontinued.
There are countless reasons why STOT is common, but discontinuation is not. Most COPD exacerbations are managed by nonpulmonologists on general medicine wards prior to discharge. In my experience, these physicians are reluctant to release a patient with exertional hypoxia without STOT. They also assume that the pulmonary clinic will do its job during the obligatory outpatient follow-up appointment they schedule with us. At the follow-up, the patient and physician are reluctant to stop therapy because of psychological dependence and therapeutic overconfidence, respectively.
In summary, STOT following hospitalization comprises the majority of all oxygen prescriptions. Historically, the United States provides far more oxygen than other developed countries, and only CMS reimbursement changes have bent the “overprescription” curve. The Ann ATS study shows that a well-designed program at the hospital level can put oxygen decisions back in the hands of providers.
Let’s “choose wisely” and follow what works, or we’ll have only ourselves to blame when reimbursement decisions are taken out of our hands.
A version of this article first appeared on Medscape.com.
Sarcoidosis: An FP’s primer on an enigmatic disease
Sarcoidosis is a multisystem inflammatory disease of unclear etiology that primarily affects the lungs. It can occur at any age but usually develops before the age of 50 years, with an initial peak incidence at 20 to 29 years and a second peak incidence after 50 years of age, especially among women in Scandinavia and Japan.1 Sarcoidosis affects men and women of all racial and ethnic groups throughout the world, but differences based on race, sex, and geography are noted.1
The highest rates are reported in northern European and African-American individuals, particularly in women.1,2 The adjusted annual incidence of sarcoidosis among African Americans is approximately 3 times that among White Americans3 and is more likely to be chronic and fatal in African Americans.3 The disease can be familial with a possible recessive inheritance mode with incomplete penetrance.4 Risk of sarcoidosis in monozygotic twins appears to be 80 times greater than that in the general population, which supports genetic factors accounting for two-thirds of disease susceptibility.5
Likely factors in the development of sarcoidosis
The exact cause of sarcoidosis is unknown, but we have insights into its pathogenesis and potential triggers.1,6-9 Genes involved are being identified: class I and II human leukocyte antigen (HLA) molecules are most consistently associated with risk of sarcoidosis. Environmental exposures can activate the innate immune system and precondition a susceptible individual to react to potential causative antigens in a highly polarized, antigen-specific Th1 immune response. The epithelioid granulomatous response involves local proinflammatory cytokine production and enhanced T-cell immunity at sites of inflammation.10 Granulomas generally form to confine pathogens, restrict inflammation, and protect surrounding tissue.11-13
ACCESS (A Case Control Etiologic Study of Sarcoidosis) identified several environmental exposures such as chemicals used in the agriculture industry, mold or mildew, and musty odors at work.14 Tobacco use was not associated with sarcoidosis.14 Recent studies have shown positive associations with service in the US Navy,15 metal working,16 firefighting,17 the handling of building supplies,18 and onsite exposure while assisting in rescue efforts at the World Trade Center disaster.19 Other data support the likelihood that specific environmental exposures associated with microbe-rich environments modestly increase the risk of sarcoidosis.14 Mycobacterial and propionibacterial DNA and RNA are potentially associated with sarcoidosis.20
Clinical manifestations are nonspecific
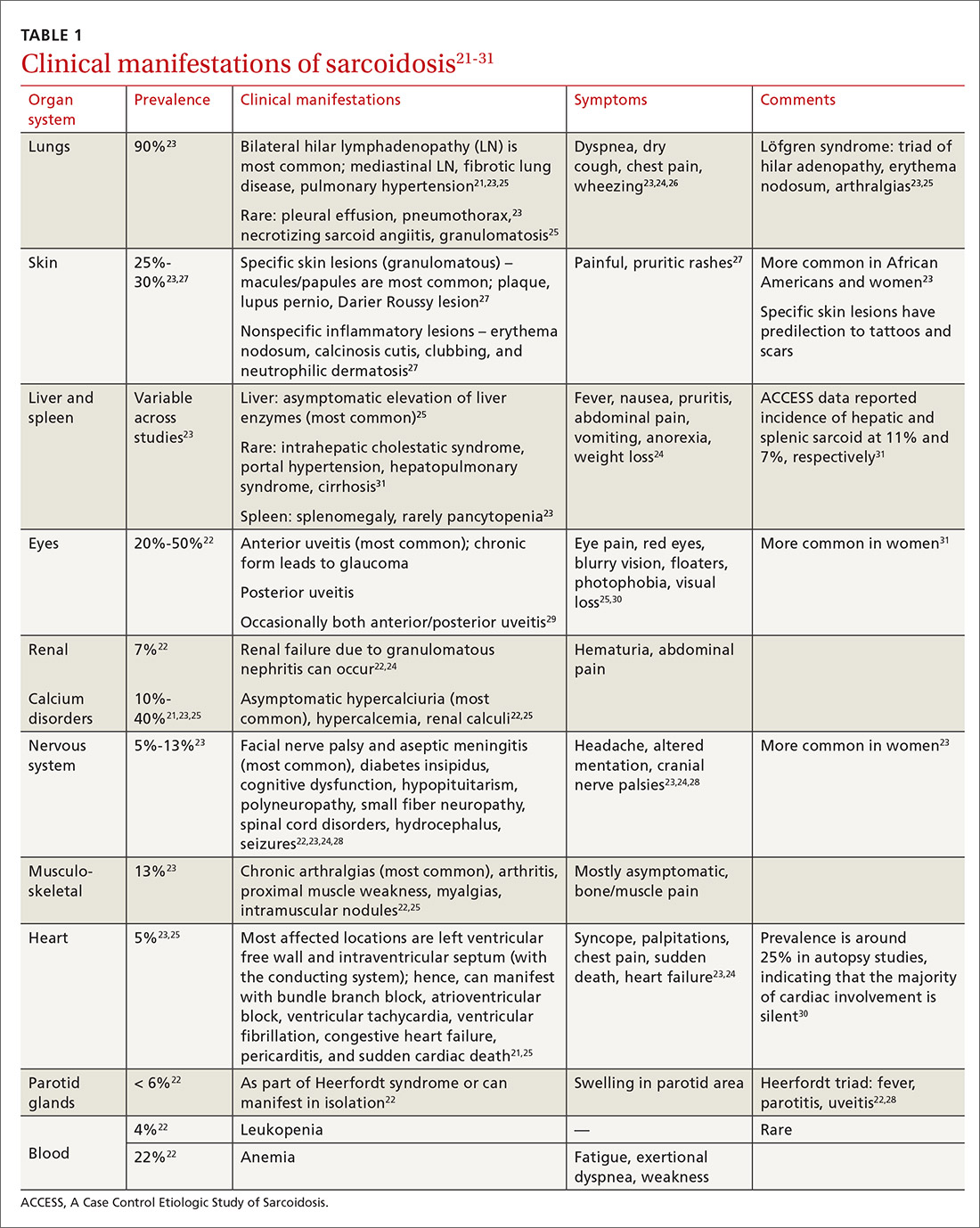
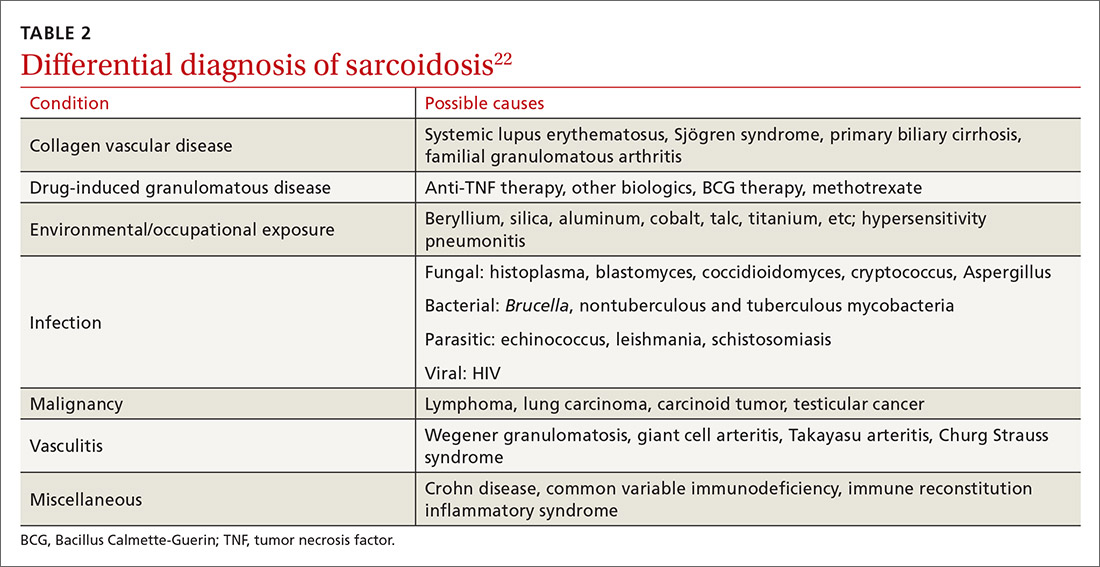
The diagnosis of sarcoidosis can be difficult and delayed due to diverse organ involvement and nonspecific presentations. TABLE 121-31 shows the diverse manifestations in a patient with suspected sarcoidosis. Around 50% of the patients are asymptomatic.23,24 Sarcoidosis is a diagnosis of exclusion, starting with a detailed history to rule out infections, occupational or environmental exposures, malignancies, and other possible disorders (TABLE 2).22
Diagnostic work-up
Radiologic studies
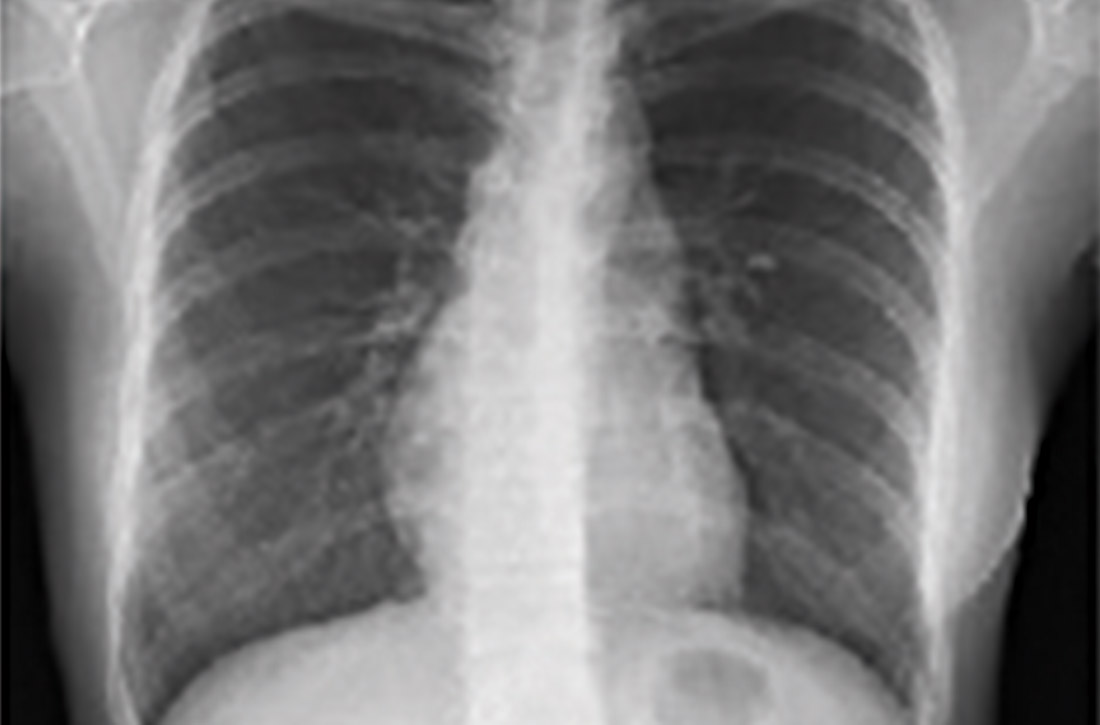
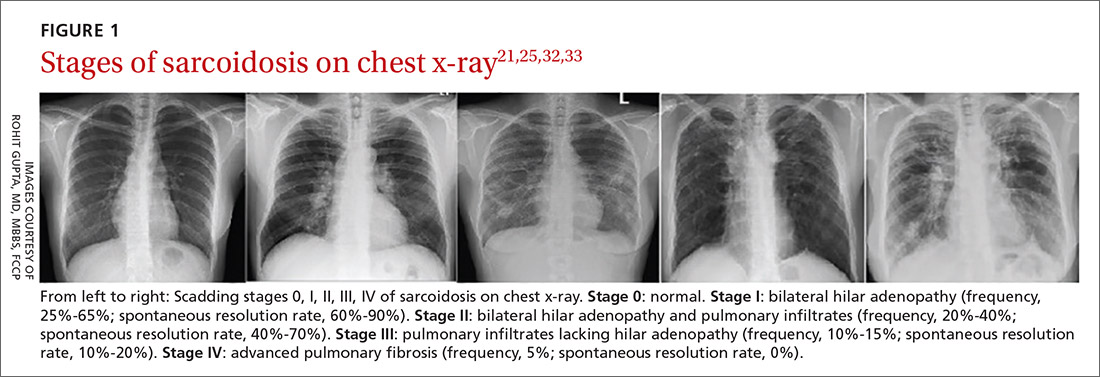
Chest x-ray (CXR) provides diagnostic and prognostic information in the evaluation of sarcoidosis using the Scadding classification system (FIGURE 1).21,25,32,33 Interobserver variability, especially between stages II and III and III and IV is the major limitation of this system.32 At presentation, radiographs are abnormal in approximately 90% of patients.34 Lymphadenopathy is the most common radiographic abnormality, occurring in more than two-thirds of cases, and pulmonary opacities (nodules and reticulation) with a middle to upper lobe predilection are present in 20% to 50% of patients.1,31,35 The nodules vary in size and can coalesce and cause alveolar collapse, thus producing consolidation.36 Linear opacities radiating laterally from the hilum into the middle and upper zones are characteristic in fibrotic disease.
Continue to: High-resoluton computed tomography
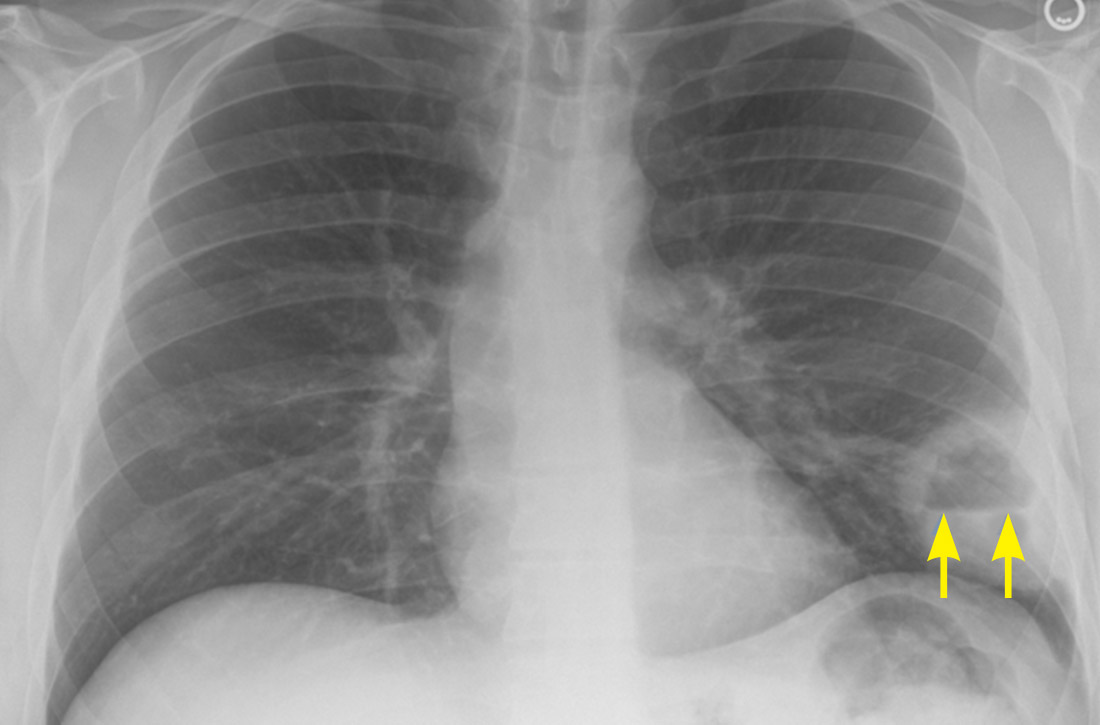
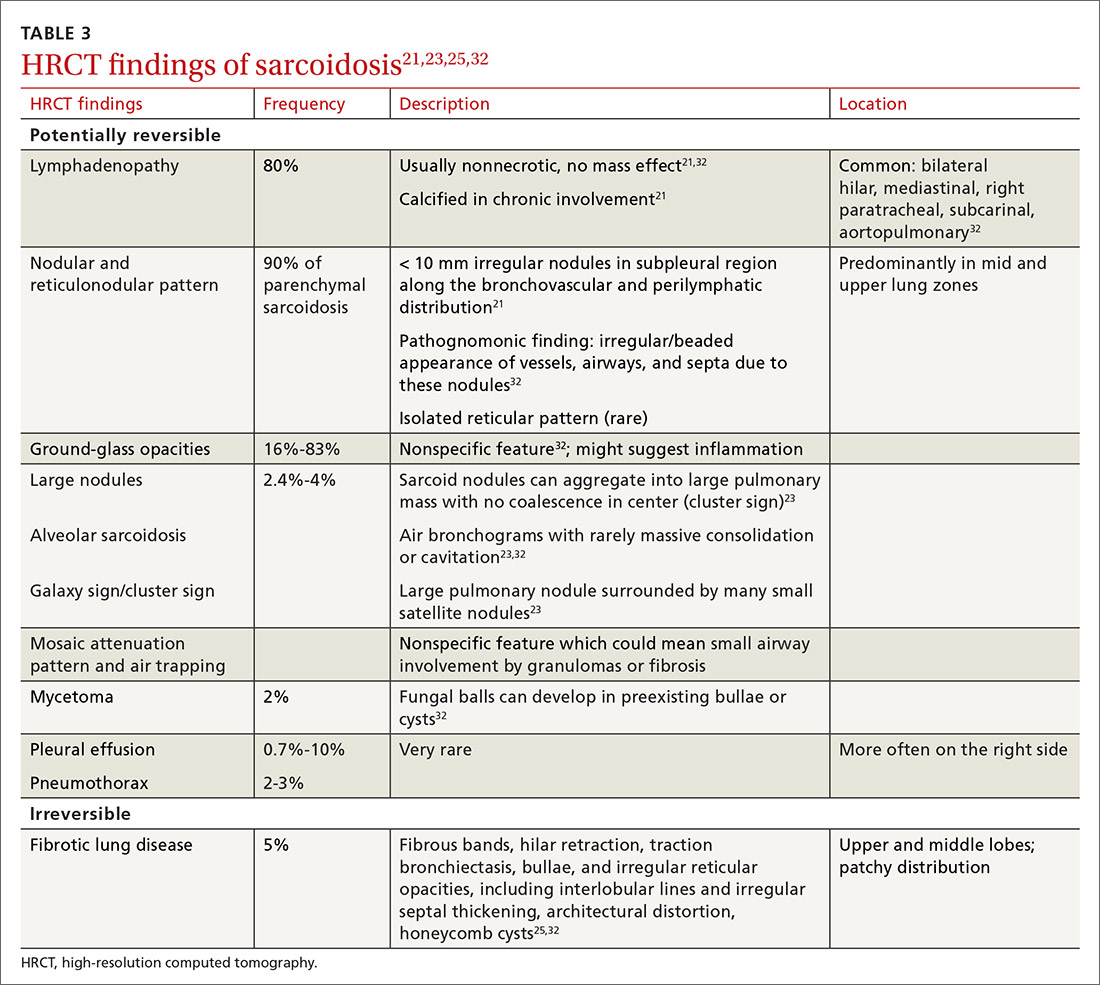
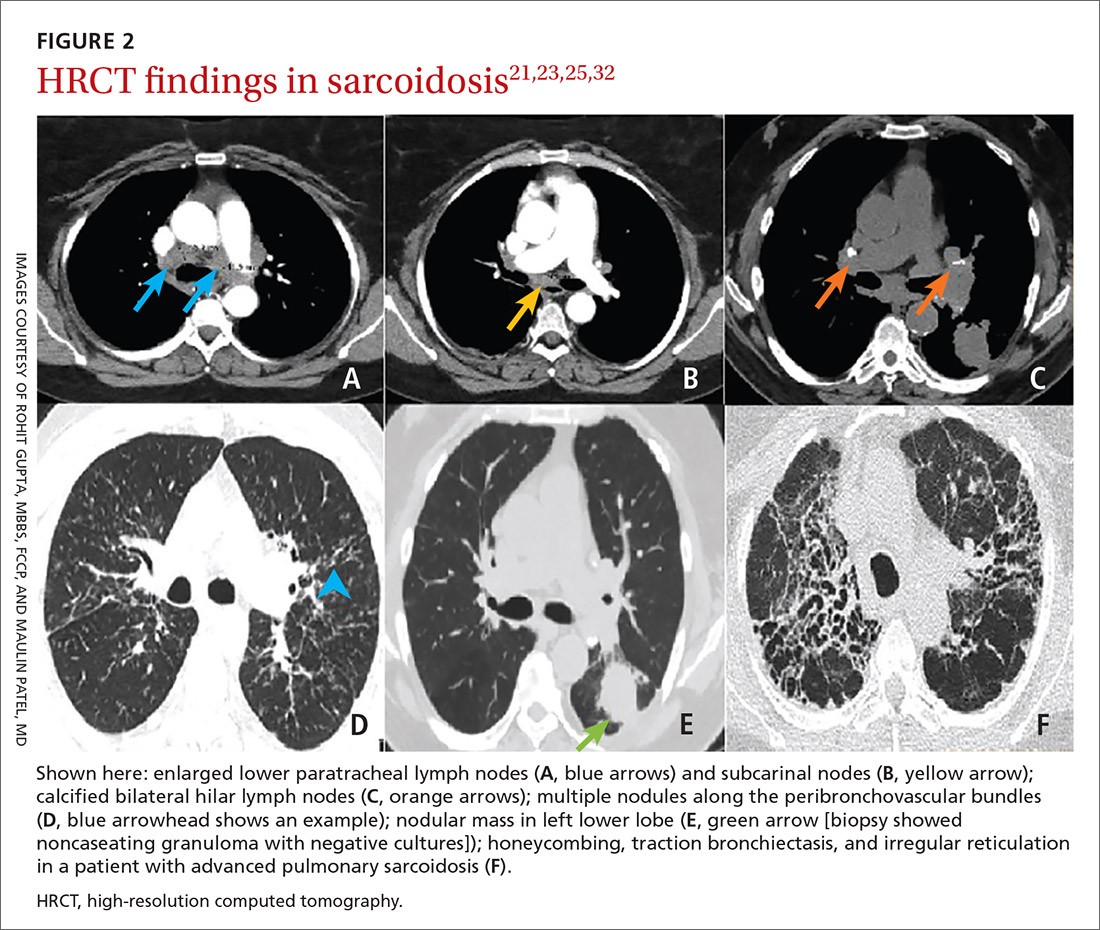
High-resolution computed tomography (HRCT). Micronodules in a perilymphatic distribution with upper lobe predominance combined with subcarinal and symmetrical hilar lymph node enlargement is practically diagnostic of sarcoidosis in the right clinical context. TABLE 321,23,25,32 and FIGURE 221,23,25,32 summarize the common CT chest findings of sarcoidosis.
Advanced imaging such as (18)F-fluorodeoxyglucose positron emission tomography (PET) and magnetic resonance imaging (MRI) are used in specialized settings for advanced pulmonary, cardiac, or neurosarcoidosis.
Tissue biopsy
Skin lesions (other than erythema nodosum), eye lesions, and peripheral lymph nodes are considered the safest extrapulmonary locations for biopsy.21,25 If pulmonary infiltrates or lymphadenopathy are present, or if extrapulmonary biopsy sites are not available, then flexible bronchoscopy with biopsy is the mainstay for tissue sampling.25
Bronchoalveolar lavage (BAL), transbronchial biopsy (TBB), endobronchial biopsy (EBB), and endobronchial ultrasound (EBUS) are invaluable modalities that have reduced the need for open lung biopsy. BAL in sarcoidosis can show lymphocytosis > 15% (nonspecific) and a CD4:CD8 lymphocyte ratio > 3.5 (specificity > 90%).21,22 TBB is more sensitive than EBB; however, sensitivity overall is heightened when both of them are combined. The advent of EBUS has increased the safety and efficiency of needle aspiration of mediastinal lymph nodes. Diagnostic yield of EBUS (~80%) is superior to that with TBB and EBB (~50%), especially in stage I and II sarcoidosis.37 The combination of EBUS with TBB improves the diagnostic yield to ~90%.37
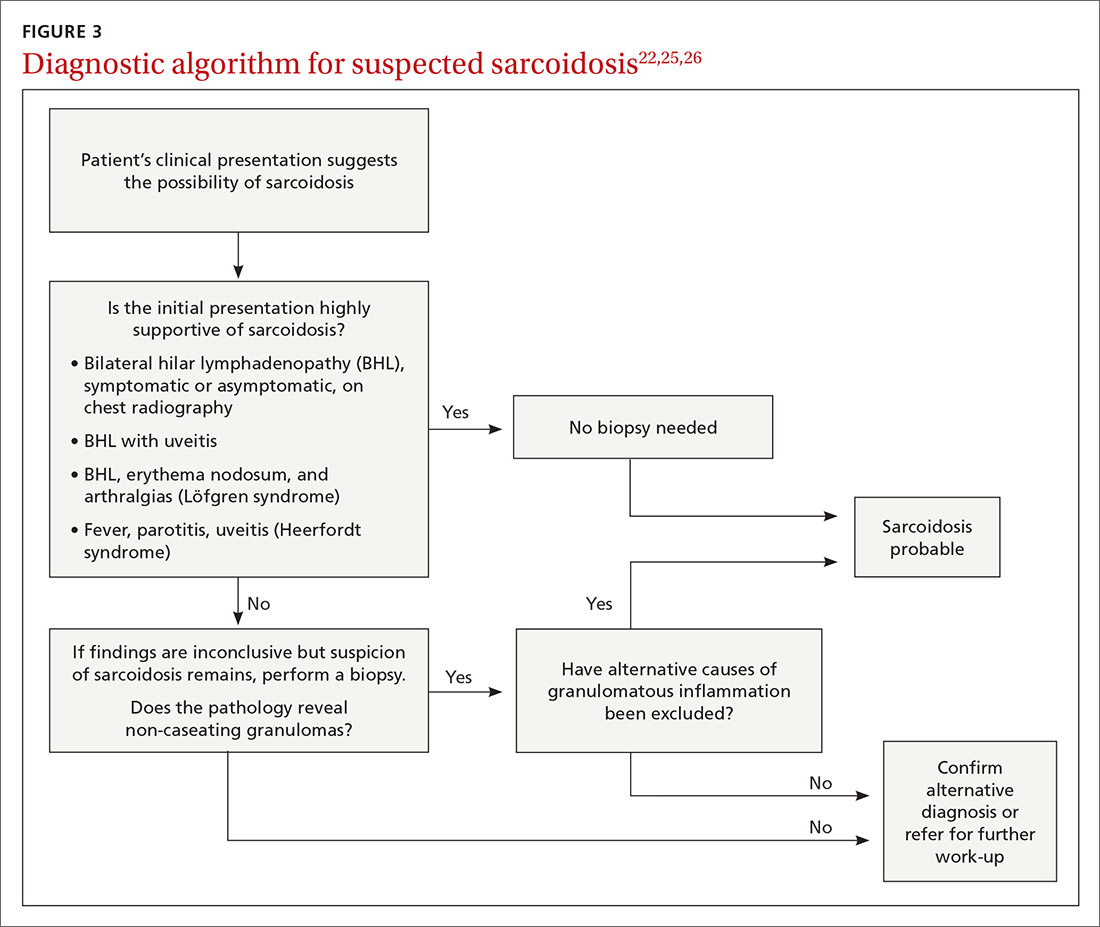
The decision to obtain biopsy samples hinges on the nature of clinical and radiologic findings (FIGURE 3).22,25,26
Continue to: Laboratory studies
Laboratory studies
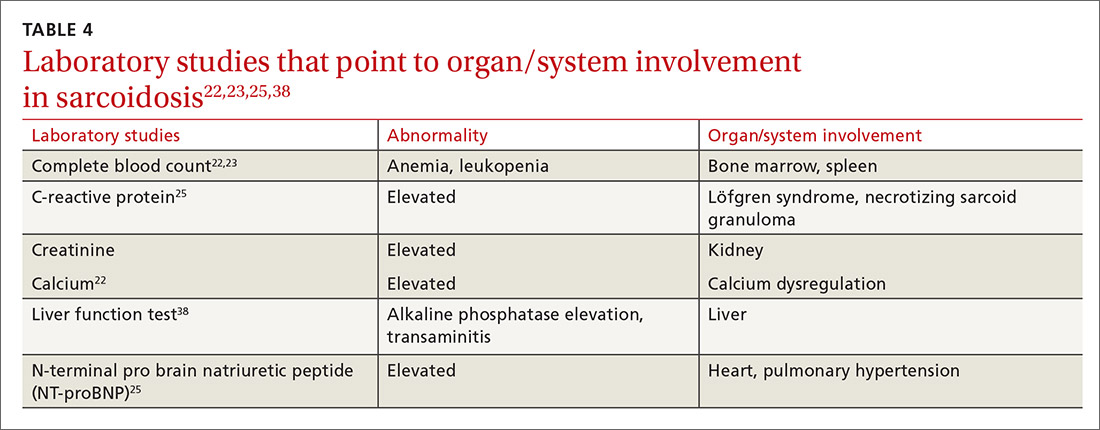
Multiple abnormalities may be seen in sarcoidosis, and specific lab tests may help support a diagnosis of sarcoidosis or detect organ-specific disease activity (TABLE 4).22,23,25,38 However, no consistently accurate biomarkers exist for use in clinical practice. An angiotensin-converting enzyme (ACE) level greater than 2 times the upper limit of normal may be helpful; however, sensitivity remains low, and genetic polymorphisms can influence the ACE level.25 Biomarkers sometimes used to assess disease activity are serum interleukin-2 receptor, neopterin, chitotriosidase, lysozyme, KL-6 glycoprotein, and amyloid A.21
Additional tests to assess specific features or organ involvement
Pulmonary function testing (PFT) is reviewed in detail below under “pulmonary sarcoidosis.”
Electrocardiogram (EKG)/transthoracic echocardiogram (TTE). EKG abnormalities—conduction disturbances, arrhythmias, or nonspecific ST segment and T-wave changes—are the most common nonspecific findings.30 TTE findings are also nonspecific but have value in assessing cardiac chamber size and function and myocardial involvement. TTE is indeed the most common screening modality for sarcoidosis-associated pulmonary hypertension (SAPH), which is definitively diagnosed by right heart catheterization (RHC). Further evaluation for cardiac sarcoidosis can be done with cardiac MRI or fluorodeoxyglucose PET in specialized settings.
Lumbar puncture (LP) may reveal lymphocytic infiltration in suspected neurosarcoidosis, but the finding is nonspecific and can reflect infection or malignancy. Oligoclonal bands may also be seen in about one-third of neurosarcoidosis cases, and it is imperative to rule out multiple sclerosis.28
Pulmonary sarcoidosis
Pulmonary sarcoidosis accounts for most of the morbidity, mortality, and health care use associated with sarcoidosis.39,40
Continue to: Pathology of early and advanced pulmonary sarcoidosis
Pathology of early and advanced pulmonary sarcoidosis
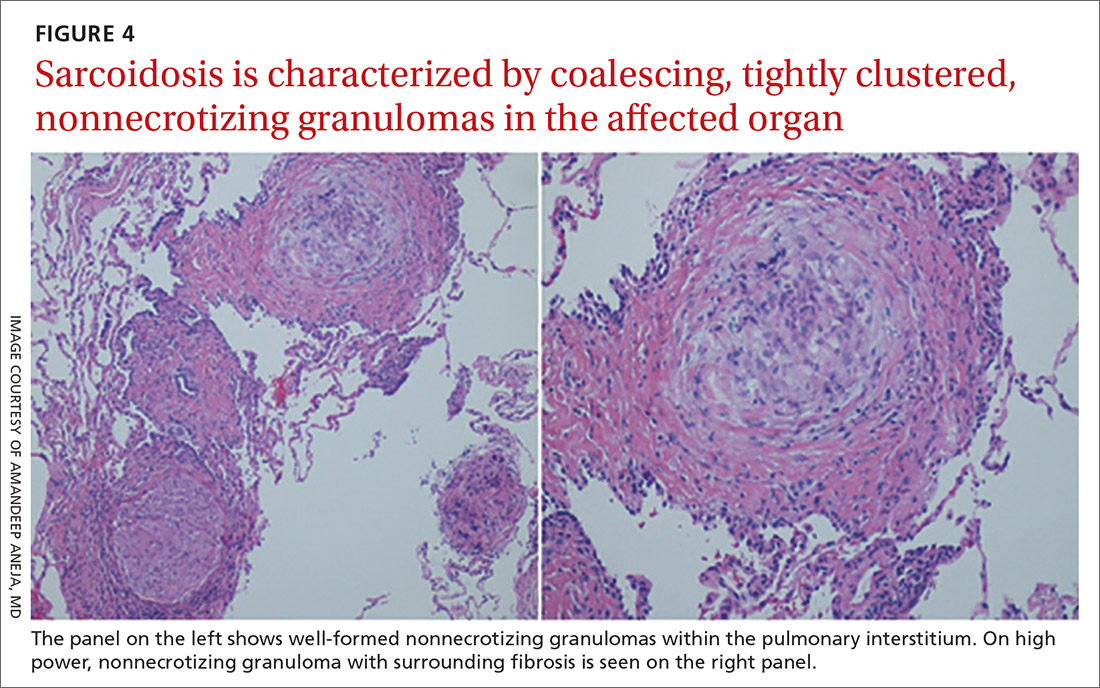
Sarcoidosis is characterized by coalescing, tightly clustered, nonnecrotizing granulomas in the lung (FIGURE 4), most often located along the lymphatic routes of the pleura, interlobular septa, and bronchovascular bundles.41 Granulomas contain epithelioid cells or multinucleated giant cells surrounded by a chronic lymphocytic infiltrate. Typically, intracytoplasmic inclusions, such as Schaumann bodies, asteroid bodies, and blue bodies of calcium oxalates are noted within giant cells.
In chronic disease, lymphocytic infiltrate vanishes and granulomas tend to become increasingly fibrotic and enlarge to form hyalinized nodules rich with densely eosinophilic collagen. In 10% to 30% of cases, the lungs undergo progressive fibrosis.40 Nonresolving inflammation appears to be the major cause of fibrosis and the peribronchovascular localization leading to marked bronchial distortion.
Clinical features, monitoring, and outcomes
Pulmonary involvement occurs in most patients with sarcoidosis, and subclinical pulmonary disease is generally present, even when extrathoracic manifestations predominate.23 Dry cough, dyspnea, and chest discomfort are the most common symptoms. Chest auscultation is usually unremarkable. Wheezing is more common in those with fibrosis and is attributed to airway-centric fibrosis.42 There is often a substantial delay between the onset of symptoms and the diagnosis of pulmonary sarcoidosis, as symptoms are nonspecific and might be mistaken for more common pulmonary diseases, such as asthma or chronic bronchitis.43
Since sarcoidosis can affect pulmonary parenchyma, interstitium, large and small airways, pulmonary vasculature, and respiratory muscles, the pattern of lung function impairment on PFT varies from normal to obstruction, restriction, isolated diffusion defect, or a combination of these. The typical physiologic abnormality is a restrictive ventilatory defect with a decreased diffusing capacity of the lung for carbon monoxide (DLCO). Extent of disease seen on HRCT correlates with level of restriction.44 Airway obstruction can be multifactorial and due to airway distortion (more likely to occur in fibrotic lung disease) and luminal disease.45-48 The 6-minute walk test and DLCO can also aid in the diagnosis of SAPH and advanced parenchymal lung disease.
While monitoring is done clinically and with testing (PFT and imaging) as needed, the optimal approach is unclear. Nevertheless, longitudinal monitoring with testing may provide useful management and prognostic information.40 Pulmonary function can remain stable in fibrotic sarcoidosis over extended periods and actually can improve in some patients.49 Serial spirometry, particularly forced vital capacity, is the most reliable tool for monitoring; when a decline in measurement occurs, chest radiography can elucidate the mechanism.50,51
Continue to: Because sarcoidosis is a multisystem disease...
Because sarcoidosis is a multisystem disease, caution needs to be exercised when evaluating a patient’s new or worsening respiratory symptoms to accurately determine the cause of symptoms and direct therapy accordingly. In addition to refractory inflammatory pulmonary disease, airway disease, infection, fibrosis, and SAPH, one needs to consider extrapulmonary involvement or complications such as cardiac or neurologic disease, musculoskeletal disease, depression, or fatigue. Adverse medication effects, deconditioning, or unrelated (or possibly related) disorders (eg pulmonary embolism) may be to blame.
Determining prognosis
Prognosis of sarcoidosis varies and depends on epidemiologic factors, clinical presentation, and course, as well as specific organ involvement. Patients may develop life-threatening pulmonary, cardiac, or neurologic complications. End-stage disease may require organ transplantation for eligible patients.
Most patients with pulmonary sarcoidosis experience clinical remission with minimal residual organ impairment and a favorable long-term outcome. Advanced pulmonary disease (known as APS) occurs in a small proportion of patients with sarcoidosis but accounts for most of the poor outcomes in sarcoidosis.40 APS is variably defined, but it generally includes pulmonary fibrosis, SAPH, and respiratory infection.
One percent to 5% of patients with sarcoidosis die from complications, and mortality is higher in women and African Americans.52 Mortality and morbidity may be increasing.53 The reasons behind these trends are unclear but could include true increases in disease incidence, better detection rates, greater severity of disease, or an aging population. Increased hospitalizations and health care use might be due to organ damage from granulomatous inflammation (and resultant fibrosis), complications associated with treatment, and psychosocial effects of the disease/treatment.
Management
Management consists primarily of anti-inflammatory or immunosuppressive therapies but can also include measures to address specific complications (such as fatigue) and organ transplant, as well as efforts to counter adverse medication effects. Other supportive and preventive measures may include, on a case-by-case basis, oxygen supplementation, vaccinations, or pulmonary rehabilitation. Details of these are found in other, more in-depth reviews on treatment; we will briefly review anti-inflammatory therapy, which forms the cornerstone of treatment in most patients with sarcoidosis.
Continue to: General approach to treatment decisions
General approach to treatment decisions. Anti-inflammatory therapy is used to reduce granulomatous inflammation, thereby preserving organ function and reducing symptoms. A decision to begin treatment is one shared with the patient and is based on symptoms and potential danger of organ system failure.54 Patients who are symptomatic or have progressive disease or physiologic impairment are generally candidates for treatment. Monitoring usually suffices for those who have minimal symptoms, stable disease, and preserved organ function.
Patients with pulmonary sarcoidosis at CXR stage 0 should not receive treatment, given that large, randomized trials have shown no meaningful benefit and that these patients have a high likelihood of spontaneous remission and excellent long-term prognosis.55-58 However, a subgroup of patients classified as stage 0/I on CXR may show parenchymal disease on HRCT,59 and, if more symptomatic, could be considered for treatment. For patients with stage II to IV pulmonary sarcoidosis with symptoms, there is good evidence that treatment may improve lung function and reduce dyspnea and fatigue.57,60-62
Corticosteroids are first-line treatment for most patients. Based on expert opinion, treatment of pulmonary sarcoidosis is generally started with oral prednisone (or an equivalent corticosteroid). A starting dose of 20 to 40 mg/d generally is sufficient for most patients. If the patient responds to initial treatment, prednisone dose is tapered over a period of months. If symptoms worsen during tapering, the minimum effective dose is maintained without further attempts at tapering. Treatment is continued for at least 3 to 6 months but it might be needed for longer durations; unfortunately, evidence-based guidelines are lacking.63 Once the patient goes into remission, close monitoring is done for possible relapses. Inhaled corticosteroids alone have not reduced symptoms or improved lung function in patients with pulmonary sarcoidosis.64-66
Steroid-sparing agents are added for many patients. For patients receiving chronic prednisone therapy (≥ 10 mg for > 6 months), steroid-sparing agents are considered to minimize the adverse effects of steroids or to better control the inflammatory activity of sarcoidosis. These agents must be carefully selected, and clinical and laboratory monitoring need to be done throughout therapy. TABLE 558,64,67-81
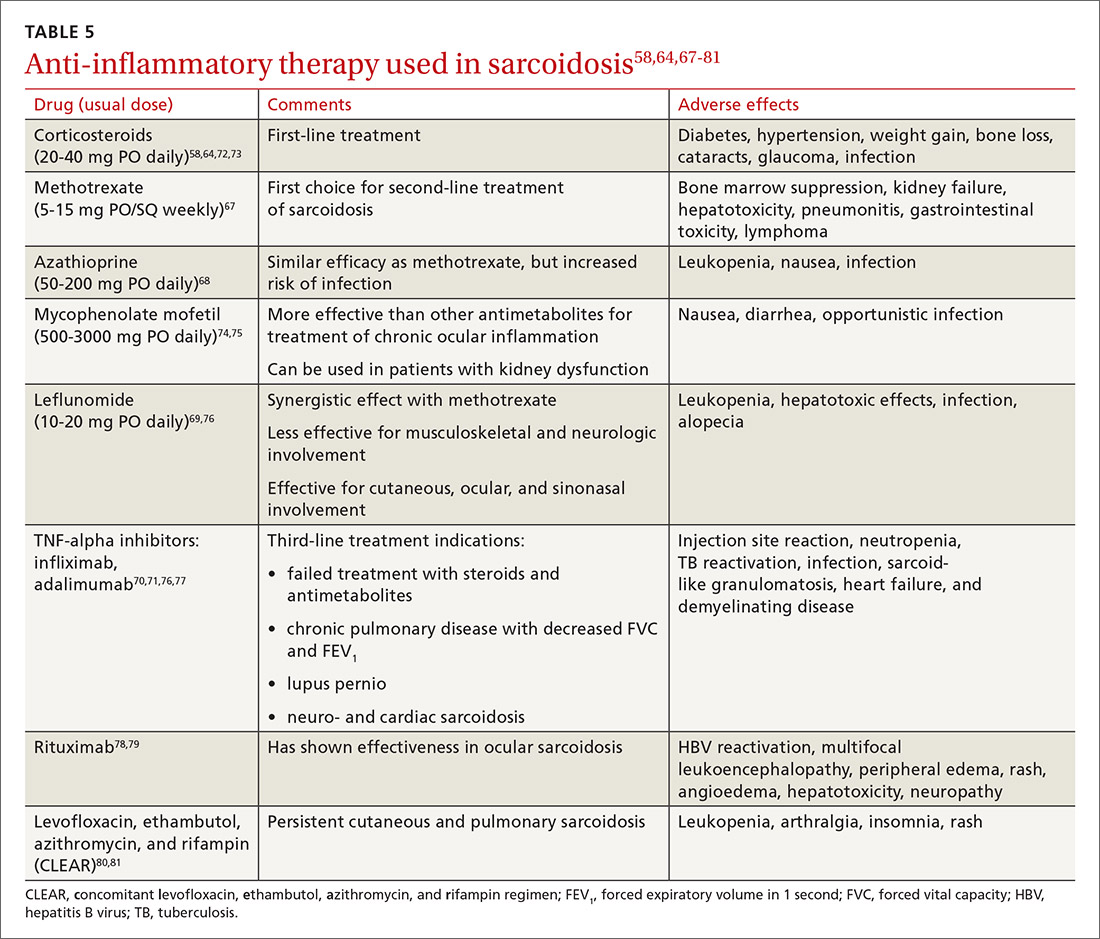
The management might be complicated for extrapulmonary, multi-organ, and advanced sarcoidosis (advanced pulmonary sarcoidosis, cardiac disease, neurosarcoidosis, lupus pernio, etc) when specialized testing, as well as a combination of corticosteroids and steroid-sparing agents (with higher doses or prolonged courses), might be needed. This should be performed at an expert sarcoidosis center, ideally in a multidisciplinary setting involving pulmonologists and/or rheumatologists, chest radiologists, and specialists as indicated, based on specific organ involvement.
Continue to: Research and future directions
Research and future directions
Key goals for research are identifying more accurate biomarkers of disease, improving diagnosis of multi-organ disease, determining validated endpoints of clinical trials in sarcoidosis, and developing treatments for refractory cases.
There is optimism and opportunity in the field of sarcoidosis overall. An example of an advancement is in the area of APS, as the severity and importance of this phenotype has been better understood. Worldwide registries and trials of pulmonary vasodilator therapy (bosentan, sildenafil, epoprostenol, and inhaled iloprost) in patients with SAPH without left ventricular dysfunction are promising.82-85 However, no benefit in survival has been shown.
RioSAPH is a double-blind, placebo-controlled trial of Riociguat (a stimulator of soluble guanylate cyclase) for SAPH (NCT02625558) that is closed to enrollment and undergoing data review. Similarly, results of the phase IV study of pirfenidone, an antifibrotic agent that was shown to decrease disease progression and deaths in idiopathic pulmonary fibrosis,86 are awaited in the near future.
Other potential directions being explored are multicenter patient registries and randomized controlled trials, analyses of existing databases, use of biobanking, and patient-centered outcome measures. Hopefully, the care of patients with sarcoidosis will become more evidence based with ongoing and upcoming research in this field.
CORRESPONDENCE
Rohit Gupta, MBBS, FCCP, 3401 North Broad Street, 7 Parkinson Pavilion, Philadelphia, PA 19140; rohit.gupta@tuhs.temple.edu
1. Costabel U, Hunninghake G. ATS/ERS/WASOG statement on sarcoidosis. Sarcoidosis Statement Committee. American Thoracic Society. European Respiratory Society. World Association for Sarcoidosis and Other Granulomatous Disorders. Eur Respir J. 1999;14:735-737.
2. Hillerdal G, Nöu E, Osterman K, et al. Sarcoidosis: epidemiology and prognosis. A 15-year European study. Am Rev Respir Dis. 1984;130:29-32.
3. Mirsaeidi M, Machado RF, Schraufnagel D, et al. Racial difference in sarcoidosis mortality in the United States. Chest. 2015;147:438-449.
4. Rybicki BA, Iannuzzi MC, Frederick MM, et al. Familial aggregation of sarcoidosis. A case-control etiologic study of sarcoidosis (ACCESS). Am J Resp Crit Care Med. 2001;164:2085-2091.
5. Sverrild A, Backer V, Kyvik KO, et al. Heredity in sarcoidosis:a registry-based twin study. Thorax. 2008;63:894.
6. Vuyst P, Dumortier P, Schandené L, et al. Sarcoidlike lung granulomatosis induced by aluminum dusts. Am Rev Respir Dis. 1987;135:493-497.
7. Werfel U, Schneider J, Rödelsperger K, et al. Sarcoid granulomatosis after zirconium exposure with multiple organ involvement. European Respir J. 1998;12:750.
8. Newman KL, Newman LS. Occupational causes of sarcoidosis. Curr Opin Allergy Clin Immunol. 2012;12:145-150.
9. Zissel G, Müller-Quernheim J. Specific antigen(s) in sarcoidosis:a link to autoimmunity? Eur Respir J. 2016;47:707-709.
10. Chen ES, Moller DR. Etiology of sarcoidosis. Clin Chest Med. 2008;29:365-377.
11. Agostini C, Adami F, Semenzato G. New pathogenetic insights into the sarcoid granuloma. Curr Opin Rheumatol. 2000;12:71-76.
12. Valentonyte R, Hampe J, Huse K, et al. Sarcoidosis is associated with a truncating splice site mutation in BTNL2. Nat Genet. 2005;37:357-364.
13. Rybicki BA, Walewski JL, Maliarik MJ, et al. The BTNL2 gene and sarcoidosis susceptibility in African Americans and Whites. Am J Hum Genet. 2005;77:491-499.
14. Newman LS, Rose CS, Bresnitz EA, et al. A case control etiologic study of sarcoidosis: environmental and occupational risk factors. Am J Respir Crit Care Med. 2004;170:1324-1330.
15. Gorham ED, Garland CF, Garland FC, et al. Trends and occupational associations in incidence of hospitalized pulmonary sarcoidosis and other lung diseases in Navy personnel: a 27-year historical prospective study, 1975-2001. Chest. 2004;126:1431-1438.
16. Kucera GP, Rybicki BA, Kirkey KL, et al. Occupational risk factors for sarcoidosis in African-American siblings. Chest. 2003;123:1527-1535.
17. Prezant DJ, Dhala A, Goldstein A, et al. The incidence, prevalence, and severity of sarcoidosis in New York City firefighters. Chest. 1999;116:1183-1193.
18. Barnard J, Rose C, Newman L, et al. Job and industry classifications associated with sarcoidosis in A Case–Control Etiologic Study of Sarcoidosis (ACCESS). J Occup Environ Med. 2005;47:226-234.
19. Izbicki G, Chavko R, Banauch GI, et al. World Trade Center “sarcoid-like” granulomatous pulmonary disease in New York City Fire Department rescue workers. Chest. 2007;131:1414-1423.
20. Eishi Y, Suga M, Ishige I, et al. Quantitative analysis of mycobacterial and propionibacterial DNA in lymph nodes of Japanese and European patients with sarcoidosis. J Clin Microbiol. 2002;40:198-204.
21. Valeyre D, Prasse A, Nunes H, et al. Sarcoidosis. Lancet. 2014;383:1155-1167.
22. Crouser ED, Maier LA, Wilson KC, et al. Diagnosis and detection of sarcoidosis. An official American Thoracic Society clinical practice guideline. Am J Respir Crit Care Med. 2020;201:e26-51.
23. Judson MA, ed. Pulmonary Sarcoidosis: A Guide for the Practicing Clinician. Springer; 2014.
24. Govender P, Berman JS. The diagnosis of sarcoidosis. Clin Chest Med. 2015;36:585-602.
25. Valeyre D, Bernaudin J-F, Uzunhan Y, et al. Clinical presentation of sarcoidosis and diagnostic work-up. Semin Resp Crit Care Med. 2014;35:336-351.
26. Judson MA. The clinical features of sarcoidosis: a comprehensive review. Clin Rev Allergy Immunol. 2015;49:63-78.
27. Wanat KA, Rosenbach M. Cutaneous sarcoidosis. Clin Chest Med. 2015;36:685-702.
28. Culver DA, Neto ML, Moss BP, et al. Neurosarcoidosis. Semin Resp Crit Care Med. 2017;38:499-513.
29. Pasadhika S, Rosenbaum JT. Ocular sarcoidosis. Clin Chest Med. 2015;36:669-683.
30. Sayah DM, Bradfield JS, Moriarty JM, et al. Cardiac involvement in sarcoidosis: evolving concepts in diagnosis and treatment. Semin Resp Crit Care Med. 2017;38:477-498.
31. Baughman RP, Teirstein AS, Judson MA, et al. Clinical characteristics of patients in a case control study of sarcoidosis. Am J Resp Crit Care. 2012;164:1885-1889.
32. Keijsers RG, Veltkamp M, Grutters JC. Chest imaging. Clin Chest Med. 2015;36:603-619.
33. Scadding J. Prognosis of intrathoracic sarcoidosis in England. A review of 136 cases after five years’ observation. Brit Med J. 1961;2:1165-1172.
34. Miller B, Putman C. The chest radiograph and sarcoidosis. Reevaluation of the chest radiograph in assessing activity of sarcoidosis: a preliminary communication. Sarcoidosis. 1985;2:85-90.
35. Loddenkemper R, Kloppenborg A, Schoenfeld N, et al. Clinical findings in 715 patients with newly detected pulmonary sarcoidosis--results of a cooperative study in former West Germany and Switzerland. WATL Study Group. Wissenschaftliche Arbeitsgemeinschaft für die Therapie von Lungenkrankheitan. Sarcoidosis Vasc Diffuse Lung Dis. 1998;15:178-182.
36. Calandriello L, Walsh SLF. Imaging for sarcoidosis. Semin Resp Crit Care Med. 2017;38:417-436.
37. Gupta D, Dadhwal DS, Agarwal R, et al. Endobronchial ultrasound-guided transbronchial needle aspiration vs conventional transbronchial needle aspiration in the diagnosis of sarcoidosis. Chest. 2014;146:547-556.
38. Baydur A. Recent developments in the physiological assessment of sarcoidosis: clinical implications. Curr Opin Pulm Med. 2012;18:499-505.
39. Jamilloux Y, Maucort-Boulch D, Kerever S, et al. Sarcoidosis-related mortality in France: a multiple-cause-of-death analysis. Eur Respir J. 2016;48:1700-1709.
40. Gupta R, Baughman RP. Advanced pulmonary sarcoidosis. Semin Respir Crit Care Med. 2020;41:700-715.
41. Rossi G, Cavazza A, Colby TV. Pathology of sarcoidosis. Clin Rev Allergy Immunol. 2015;49:36-44.
42. Hansell D, Milne D, Wilsher M, et al. Pulmonary sarcoidosis: morphologic associations of airflow obstruction at thin-section CT. Radiology. 1998;209:697-704.
43. Judson MA, Thompson BW, Rabin DL, et al. The diagnostic pathway to sarcoidosis. Chest. 2003;123:406-412.
44. Müller NL, Mawson JB, Mathieson JR, et al. Sarcoidosis: correlation of extent of disease at CT with clinical, functional, and radiographic findings. Radiology. 1989;171:613-618.
45. Harrison BDW, Shaylor JM, Stokes TC, et al. Airflow limitation in sarcoidosis—a study of pulmonary function in 107 patients with newly diagnosed disease. Resp Med. 1991;85:59-64.
46. Polychronopoulos VS, Prakash UBS. Airway Involvement in sarcoidosis. Chest. 2009;136:1371-1380.
47. Chambellan A, Turbie P, Nunes H, et al. Endoluminal stenosis of proximal bronchi in sarcoidosis: bronchoscopy, function, and evolution. Chest. 2005;127:472-481.
48. Handa T, Nagai S, Fushimi Y, et al. Clinical and radiographic indices associated with airflow limitation in patients with sarcoidosis. Chest. 2006;130:1851-1856.
49. Nardi A, Brillet P-Y, Letoumelin P, et al. Stage IV sarcoidosis: comparison of survival with the general population and causes of death. Eur Respir J. 2011;38:1368-1373.
50. Zappala CJ, Desai SR, Copley SJ, et al. Accuracy of individual variables in the monitoring of long-term change in pulmonary sarcoidosis as judged by serial high-resolution CT scan data. Chest. 2014;145:101-107.
51. Gafà G, Sverzellati N, Bonati E, et al. Follow-up in pulmonary sarcoidosis: comparison between HRCT and pulmonary function tests. Radiol Med. 2012;117:968-978.
52. Gerke AK. Morbidity and mortality in sarcoidosis. Curr Opin Pulm Med. 2014;20:472-478.
53. Kearney GD, Obi ON, Maddipati V, et al. Sarcoidosis deaths in the United States: 1999–2016. Respir Med. 2019;149:30-35.
54. Baughman RP, Judson M, Wells A. The indications for the treatment of sarcoidosis: Wells Law. Sarcoidosis Vasc Diffuse Lung Dis. 2017;34:280-282.
55. Nagai S, Shigematsu M, Hamada K, et al. Clinical courses and prognoses of pulmonary sarcoidosis. Curr Opin Pulm Med. 1999;5:293-298.
56. Neville E, Walker AN, James DG. Prognostic factors predicting the outcome of sarcoidosis: an analysis of 818 patients. Q J Med. 1983;52:525-533.
57. Bradley B, Branley HM, Egan JJ, et al. Interstitial lung disease guideline: the British Thoracic Society in collaboration with the Thoracic Society of Australia and the Irish Thoracic Society. Thorax. 2008;63(suppl 5):v1-v58.
58. Pietinalho A, Tukiainen P, Haahtela T, et al. Oral prednisolone followed by inhaled budesonide in newly diagnosed pulmonary sarcoidosis: a double-blind, placebo-controlled multicenter study. Finnish Pulmonary Sarcoidosis Group. Chest. 1999;116:424-431.
59. Oberstein A, von Zitzewitz H, Schweden F, et al. Non invasive evaluation of the inflammatory activity in sarcoidosis with high-resolution computed tomography. Sarcoidosis Vasc Diffuse Lung Dis. 1997;14:65-72.
60. Gibson G, Prescott RJ, Muers MF, et al. British Thoracic Society Sarcoidosis study: effects of long term corticosteroid treatment. Thorax. 1996;51:238-247.
61. Baughman RP, Nunes H. Therapy for sarcoidosis: evidence-based recommendations. Expert Rev Clin Immunol. 2012;8:95-103.
62. Pietinalho A, Tukiainen P, Haahtela T, et al. Early treatment of stage II sarcoidosis improves 5-year pulmonary function. Chest. 2002;121:24-31.
63. Rahaghi FF, Baughman RP, Saketkoo LA, et al. Delphi consensus recommendations for a treatment algorithm in pulmonary sarcoidosis. Eur Respir Rev. 2020;29:190146.
64. Baughman RP, Iannuzzi MC, Lower EE, et al. Use of fluticasone in acute symptomatic pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis. 2002;19:198-204.
65. du Bois RM, Greenhalgh PM, Southcott AM, et al. Randomized trial of inhaled fluticasone propionate in chronic stable pulmonary sarcoidosis: a pilot study. Eur Respir J. 1999;13:1345-1350.
66. Milman N, Graudal N, Grode G, Munch E. No effect of high‐dose inhaled steroids in pulmonary sarcoidosis: a double‐blind, placebo‐controlled study. J Intern Med. 1994;236:285-290.
67. Baughman RP, Winget DB, Lower EE. Methotrexate is steroid sparing in acute sarcoidosis: results of a double blind, randomized trial. Sarcoidosis Vasc Diffuse Lung Dis. 2000;17:60-66.
68. Vorselaars ADM, Wuyts WA, Vorselaars VMM, et al. Methotrexate vs azathioprine in second-line therapy of sarcoidosis. Chest. 2013;144:805-812.
69. Sahoo D, Bandyopadhyay D, Xu M, et al. Effectiveness and safety of leflunomide for pulmonary and extrapulmonary sarcoidosis. Eur Respir J. 2011;38:1145-1150.
70. Baughman RP, Drent M, Kavuru M, et al. Infliximab therapy in patients with chronic sarcoidosis and pulmonary involvement. Am J Resp Crit Care Med . 2006;174:795-802.
71. Rossman MD, Newman LS, Baughman RP, et al. A double-blinded, randomized, placebo-controlled trial of infliximab in subjects with active pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2006;23:201-208.
72. Selroos O, Sellergren T. Corticosteroid therapy of pulmonary sarcoidosis. A prospective evaluation of alternate day and daily dosage in stage II disease. Scand J Respir Dis . 1979;60:215-221.
73. Israel HL, Fouts DW, Beggs RA. A controlled trial of prednisone treatment of sarcoidosis. Am Rev Respir Dis . 1973;107:609-614.
74. Hamzeh N, Voelker A, Forssén A, et al. Efficacy of mycophenolate mofetil in sarcoidosis. Respir Med . 2014;108:1663-1669.
75. Brill A-K, Ott SR, Geiser T. Effect and safety of mycophenolate mofetil in chronic pulmonary sarcoidosis: a retrospective study. Respiration . 2013;86:376-383.
76. Baughman RP, Lower EE. Leflunomide for chronic sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2004;21:43-48.
77. Sweiss NJ, Noth I, Mirsaeidi M, et al. Efficacy results of a 52-week trial of adalimumab in the treatment of refractory sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2014;31:46-54.
78. Sweiss NJ, Lower EE, Mirsaeidi M, et al. Rituximab in the treatment of refractory pulmonary sarcoidosis. Eur Respir J . 2014;43:1525-1528.
79. Thatayatikom A, Thatayatikom S, White AJ. Infliximab treatment for severe granulomatous disease in common variable immunodeficiency: a case report and review of the literature. Ann Allergy Asthma Immunol . 2005;95:293-300.
80. Drake WP, Oswald-Richter K, Richmond BW, et al. Oral antimycobacterial therapy in chronic cutaneous sarcoidosis: a randomized, single-masked, placebo-controlled study. Jama Dermatol . 2013;149:1040-1049.
81. Drake WP, Richmond BW, Oswald-Richter K, et al. Effects of broad-spectrum antimycobacterial therapy on chronic pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2013;30:201-211.
82. Baughman RP, Culver DA, Cordova FC, et al. Bosentan for sarcoidosis-associated pulmonary hypertension: a double-blind placebo controlled randomized trial. Chest . 2014;145:810-817.
83. Baughman RP, Shlobin OA, Wells AU, et al. Clinical features of sarcoidosis associated pulmonary hypertension: results of a multi-national registry. Respir Med . 2018;139:72-78.
84. Fisher KA, Serlin DM, Wilson KC, et al. Sarcoidosis-associated pulmonary hypertension outcome with long-term epoprostenol treatment. Chest . 2006;130:1481-1488.
85. Baughman RP, Judson MA, Lower EE, et al. Inhaled iloprost for sarcoidosis associated pulmonary hypertension. Sarcoidosis Vasc Diffuse Lung Dis . 2009;26:110-120.
86. King TE, Bradford WZ, Castro-Bernardini S, et al. A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. N Engl J Med . 2014;370:2083-2092.
Sarcoidosis is a multisystem inflammatory disease of unclear etiology that primarily affects the lungs. It can occur at any age but usually develops before the age of 50 years, with an initial peak incidence at 20 to 29 years and a second peak incidence after 50 years of age, especially among women in Scandinavia and Japan.1 Sarcoidosis affects men and women of all racial and ethnic groups throughout the world, but differences based on race, sex, and geography are noted.1
The highest rates are reported in northern European and African-American individuals, particularly in women.1,2 The adjusted annual incidence of sarcoidosis among African Americans is approximately 3 times that among White Americans3 and is more likely to be chronic and fatal in African Americans.3 The disease can be familial with a possible recessive inheritance mode with incomplete penetrance.4 Risk of sarcoidosis in monozygotic twins appears to be 80 times greater than that in the general population, which supports genetic factors accounting for two-thirds of disease susceptibility.5
Likely factors in the development of sarcoidosis
The exact cause of sarcoidosis is unknown, but we have insights into its pathogenesis and potential triggers.1,6-9 Genes involved are being identified: class I and II human leukocyte antigen (HLA) molecules are most consistently associated with risk of sarcoidosis. Environmental exposures can activate the innate immune system and precondition a susceptible individual to react to potential causative antigens in a highly polarized, antigen-specific Th1 immune response. The epithelioid granulomatous response involves local proinflammatory cytokine production and enhanced T-cell immunity at sites of inflammation.10 Granulomas generally form to confine pathogens, restrict inflammation, and protect surrounding tissue.11-13
ACCESS (A Case Control Etiologic Study of Sarcoidosis) identified several environmental exposures such as chemicals used in the agriculture industry, mold or mildew, and musty odors at work.14 Tobacco use was not associated with sarcoidosis.14 Recent studies have shown positive associations with service in the US Navy,15 metal working,16 firefighting,17 the handling of building supplies,18 and onsite exposure while assisting in rescue efforts at the World Trade Center disaster.19 Other data support the likelihood that specific environmental exposures associated with microbe-rich environments modestly increase the risk of sarcoidosis.14 Mycobacterial and propionibacterial DNA and RNA are potentially associated with sarcoidosis.20
Clinical manifestations are nonspecific
The diagnosis of sarcoidosis can be difficult and delayed due to diverse organ involvement and nonspecific presentations. TABLE 121-31 shows the diverse manifestations in a patient with suspected sarcoidosis. Around 50% of the patients are asymptomatic.23,24 Sarcoidosis is a diagnosis of exclusion, starting with a detailed history to rule out infections, occupational or environmental exposures, malignancies, and other possible disorders (TABLE 2).22

Diagnostic work-up

Radiologic studies
Chest x-ray (CXR) provides diagnostic and prognostic information in the evaluation of sarcoidosis using the Scadding classification system (FIGURE 1).21,25,32,33 Interobserver variability, especially between stages II and III and III and IV is the major limitation of this system.32 At presentation, radiographs are abnormal in approximately 90% of patients.34 Lymphadenopathy is the most common radiographic abnormality, occurring in more than two-thirds of cases, and pulmonary opacities (nodules and reticulation) with a middle to upper lobe predilection are present in 20% to 50% of patients.1,31,35 The nodules vary in size and can coalesce and cause alveolar collapse, thus producing consolidation.36 Linear opacities radiating laterally from the hilum into the middle and upper zones are characteristic in fibrotic disease.

Continue to: High-resoluton computed tomography
High-resolution computed tomography (HRCT). Micronodules in a perilymphatic distribution with upper lobe predominance combined with subcarinal and symmetrical hilar lymph node enlargement is practically diagnostic of sarcoidosis in the right clinical context. TABLE 321,23,25,32 and FIGURE 221,23,25,32 summarize the common CT chest findings of sarcoidosis.

Advanced imaging such as (18)F-fluorodeoxyglucose positron emission tomography (PET) and magnetic resonance imaging (MRI) are used in specialized settings for advanced pulmonary, cardiac, or neurosarcoidosis.

Tissue biopsy
Skin lesions (other than erythema nodosum), eye lesions, and peripheral lymph nodes are considered the safest extrapulmonary locations for biopsy.21,25 If pulmonary infiltrates or lymphadenopathy are present, or if extrapulmonary biopsy sites are not available, then flexible bronchoscopy with biopsy is the mainstay for tissue sampling.25
Bronchoalveolar lavage (BAL), transbronchial biopsy (TBB), endobronchial biopsy (EBB), and endobronchial ultrasound (EBUS) are invaluable modalities that have reduced the need for open lung biopsy. BAL in sarcoidosis can show lymphocytosis > 15% (nonspecific) and a CD4:CD8 lymphocyte ratio > 3.5 (specificity > 90%).21,22 TBB is more sensitive than EBB; however, sensitivity overall is heightened when both of them are combined. The advent of EBUS has increased the safety and efficiency of needle aspiration of mediastinal lymph nodes. Diagnostic yield of EBUS (~80%) is superior to that with TBB and EBB (~50%), especially in stage I and II sarcoidosis.37 The combination of EBUS with TBB improves the diagnostic yield to ~90%.37
The decision to obtain biopsy samples hinges on the nature of clinical and radiologic findings (FIGURE 3).22,25,26

Continue to: Laboratory studies
Laboratory studies
Multiple abnormalities may be seen in sarcoidosis, and specific lab tests may help support a diagnosis of sarcoidosis or detect organ-specific disease activity (TABLE 4).22,23,25,38 However, no consistently accurate biomarkers exist for use in clinical practice. An angiotensin-converting enzyme (ACE) level greater than 2 times the upper limit of normal may be helpful; however, sensitivity remains low, and genetic polymorphisms can influence the ACE level.25 Biomarkers sometimes used to assess disease activity are serum interleukin-2 receptor, neopterin, chitotriosidase, lysozyme, KL-6 glycoprotein, and amyloid A.21

Additional tests to assess specific features or organ involvement
Pulmonary function testing (PFT) is reviewed in detail below under “pulmonary sarcoidosis.”
Electrocardiogram (EKG)/transthoracic echocardiogram (TTE). EKG abnormalities—conduction disturbances, arrhythmias, or nonspecific ST segment and T-wave changes—are the most common nonspecific findings.30 TTE findings are also nonspecific but have value in assessing cardiac chamber size and function and myocardial involvement. TTE is indeed the most common screening modality for sarcoidosis-associated pulmonary hypertension (SAPH), which is definitively diagnosed by right heart catheterization (RHC). Further evaluation for cardiac sarcoidosis can be done with cardiac MRI or fluorodeoxyglucose PET in specialized settings.
Lumbar puncture (LP) may reveal lymphocytic infiltration in suspected neurosarcoidosis, but the finding is nonspecific and can reflect infection or malignancy. Oligoclonal bands may also be seen in about one-third of neurosarcoidosis cases, and it is imperative to rule out multiple sclerosis.28
Pulmonary sarcoidosis
Pulmonary sarcoidosis accounts for most of the morbidity, mortality, and health care use associated with sarcoidosis.39,40
Continue to: Pathology of early and advanced pulmonary sarcoidosis
Pathology of early and advanced pulmonary sarcoidosis
Sarcoidosis is characterized by coalescing, tightly clustered, nonnecrotizing granulomas in the lung (FIGURE 4), most often located along the lymphatic routes of the pleura, interlobular septa, and bronchovascular bundles.41 Granulomas contain epithelioid cells or multinucleated giant cells surrounded by a chronic lymphocytic infiltrate. Typically, intracytoplasmic inclusions, such as Schaumann bodies, asteroid bodies, and blue bodies of calcium oxalates are noted within giant cells.

In chronic disease, lymphocytic infiltrate vanishes and granulomas tend to become increasingly fibrotic and enlarge to form hyalinized nodules rich with densely eosinophilic collagen. In 10% to 30% of cases, the lungs undergo progressive fibrosis.40 Nonresolving inflammation appears to be the major cause of fibrosis and the peribronchovascular localization leading to marked bronchial distortion.
Clinical features, monitoring, and outcomes
Pulmonary involvement occurs in most patients with sarcoidosis, and subclinical pulmonary disease is generally present, even when extrathoracic manifestations predominate.23 Dry cough, dyspnea, and chest discomfort are the most common symptoms. Chest auscultation is usually unremarkable. Wheezing is more common in those with fibrosis and is attributed to airway-centric fibrosis.42 There is often a substantial delay between the onset of symptoms and the diagnosis of pulmonary sarcoidosis, as symptoms are nonspecific and might be mistaken for more common pulmonary diseases, such as asthma or chronic bronchitis.43
Since sarcoidosis can affect pulmonary parenchyma, interstitium, large and small airways, pulmonary vasculature, and respiratory muscles, the pattern of lung function impairment on PFT varies from normal to obstruction, restriction, isolated diffusion defect, or a combination of these. The typical physiologic abnormality is a restrictive ventilatory defect with a decreased diffusing capacity of the lung for carbon monoxide (DLCO). Extent of disease seen on HRCT correlates with level of restriction.44 Airway obstruction can be multifactorial and due to airway distortion (more likely to occur in fibrotic lung disease) and luminal disease.45-48 The 6-minute walk test and DLCO can also aid in the diagnosis of SAPH and advanced parenchymal lung disease.
While monitoring is done clinically and with testing (PFT and imaging) as needed, the optimal approach is unclear. Nevertheless, longitudinal monitoring with testing may provide useful management and prognostic information.40 Pulmonary function can remain stable in fibrotic sarcoidosis over extended periods and actually can improve in some patients.49 Serial spirometry, particularly forced vital capacity, is the most reliable tool for monitoring; when a decline in measurement occurs, chest radiography can elucidate the mechanism.50,51
Continue to: Because sarcoidosis is a multisystem disease...
Because sarcoidosis is a multisystem disease, caution needs to be exercised when evaluating a patient’s new or worsening respiratory symptoms to accurately determine the cause of symptoms and direct therapy accordingly. In addition to refractory inflammatory pulmonary disease, airway disease, infection, fibrosis, and SAPH, one needs to consider extrapulmonary involvement or complications such as cardiac or neurologic disease, musculoskeletal disease, depression, or fatigue. Adverse medication effects, deconditioning, or unrelated (or possibly related) disorders (eg pulmonary embolism) may be to blame.
Determining prognosis
Prognosis of sarcoidosis varies and depends on epidemiologic factors, clinical presentation, and course, as well as specific organ involvement. Patients may develop life-threatening pulmonary, cardiac, or neurologic complications. End-stage disease may require organ transplantation for eligible patients.
Most patients with pulmonary sarcoidosis experience clinical remission with minimal residual organ impairment and a favorable long-term outcome. Advanced pulmonary disease (known as APS) occurs in a small proportion of patients with sarcoidosis but accounts for most of the poor outcomes in sarcoidosis.40 APS is variably defined, but it generally includes pulmonary fibrosis, SAPH, and respiratory infection.
One percent to 5% of patients with sarcoidosis die from complications, and mortality is higher in women and African Americans.52 Mortality and morbidity may be increasing.53 The reasons behind these trends are unclear but could include true increases in disease incidence, better detection rates, greater severity of disease, or an aging population. Increased hospitalizations and health care use might be due to organ damage from granulomatous inflammation (and resultant fibrosis), complications associated with treatment, and psychosocial effects of the disease/treatment.
Management
Management consists primarily of anti-inflammatory or immunosuppressive therapies but can also include measures to address specific complications (such as fatigue) and organ transplant, as well as efforts to counter adverse medication effects. Other supportive and preventive measures may include, on a case-by-case basis, oxygen supplementation, vaccinations, or pulmonary rehabilitation. Details of these are found in other, more in-depth reviews on treatment; we will briefly review anti-inflammatory therapy, which forms the cornerstone of treatment in most patients with sarcoidosis.
Continue to: General approach to treatment decisions
General approach to treatment decisions. Anti-inflammatory therapy is used to reduce granulomatous inflammation, thereby preserving organ function and reducing symptoms. A decision to begin treatment is one shared with the patient and is based on symptoms and potential danger of organ system failure.54 Patients who are symptomatic or have progressive disease or physiologic impairment are generally candidates for treatment. Monitoring usually suffices for those who have minimal symptoms, stable disease, and preserved organ function.
Patients with pulmonary sarcoidosis at CXR stage 0 should not receive treatment, given that large, randomized trials have shown no meaningful benefit and that these patients have a high likelihood of spontaneous remission and excellent long-term prognosis.55-58 However, a subgroup of patients classified as stage 0/I on CXR may show parenchymal disease on HRCT,59 and, if more symptomatic, could be considered for treatment. For patients with stage II to IV pulmonary sarcoidosis with symptoms, there is good evidence that treatment may improve lung function and reduce dyspnea and fatigue.57,60-62
Corticosteroids are first-line treatment for most patients. Based on expert opinion, treatment of pulmonary sarcoidosis is generally started with oral prednisone (or an equivalent corticosteroid). A starting dose of 20 to 40 mg/d generally is sufficient for most patients. If the patient responds to initial treatment, prednisone dose is tapered over a period of months. If symptoms worsen during tapering, the minimum effective dose is maintained without further attempts at tapering. Treatment is continued for at least 3 to 6 months but it might be needed for longer durations; unfortunately, evidence-based guidelines are lacking.63 Once the patient goes into remission, close monitoring is done for possible relapses. Inhaled corticosteroids alone have not reduced symptoms or improved lung function in patients with pulmonary sarcoidosis.64-66
Steroid-sparing agents are added for many patients. For patients receiving chronic prednisone therapy (≥ 10 mg for > 6 months), steroid-sparing agents are considered to minimize the adverse effects of steroids or to better control the inflammatory activity of sarcoidosis. These agents must be carefully selected, and clinical and laboratory monitoring need to be done throughout therapy. TABLE 558,64,67-81

The management might be complicated for extrapulmonary, multi-organ, and advanced sarcoidosis (advanced pulmonary sarcoidosis, cardiac disease, neurosarcoidosis, lupus pernio, etc) when specialized testing, as well as a combination of corticosteroids and steroid-sparing agents (with higher doses or prolonged courses), might be needed. This should be performed at an expert sarcoidosis center, ideally in a multidisciplinary setting involving pulmonologists and/or rheumatologists, chest radiologists, and specialists as indicated, based on specific organ involvement.
Continue to: Research and future directions
Research and future directions
Key goals for research are identifying more accurate biomarkers of disease, improving diagnosis of multi-organ disease, determining validated endpoints of clinical trials in sarcoidosis, and developing treatments for refractory cases.
There is optimism and opportunity in the field of sarcoidosis overall. An example of an advancement is in the area of APS, as the severity and importance of this phenotype has been better understood. Worldwide registries and trials of pulmonary vasodilator therapy (bosentan, sildenafil, epoprostenol, and inhaled iloprost) in patients with SAPH without left ventricular dysfunction are promising.82-85 However, no benefit in survival has been shown.
RioSAPH is a double-blind, placebo-controlled trial of Riociguat (a stimulator of soluble guanylate cyclase) for SAPH (NCT02625558) that is closed to enrollment and undergoing data review. Similarly, results of the phase IV study of pirfenidone, an antifibrotic agent that was shown to decrease disease progression and deaths in idiopathic pulmonary fibrosis,86 are awaited in the near future.
Other potential directions being explored are multicenter patient registries and randomized controlled trials, analyses of existing databases, use of biobanking, and patient-centered outcome measures. Hopefully, the care of patients with sarcoidosis will become more evidence based with ongoing and upcoming research in this field.
CORRESPONDENCE
Rohit Gupta, MBBS, FCCP, 3401 North Broad Street, 7 Parkinson Pavilion, Philadelphia, PA 19140; rohit.gupta@tuhs.temple.edu
Sarcoidosis is a multisystem inflammatory disease of unclear etiology that primarily affects the lungs. It can occur at any age but usually develops before the age of 50 years, with an initial peak incidence at 20 to 29 years and a second peak incidence after 50 years of age, especially among women in Scandinavia and Japan.1 Sarcoidosis affects men and women of all racial and ethnic groups throughout the world, but differences based on race, sex, and geography are noted.1
The highest rates are reported in northern European and African-American individuals, particularly in women.1,2 The adjusted annual incidence of sarcoidosis among African Americans is approximately 3 times that among White Americans3 and is more likely to be chronic and fatal in African Americans.3 The disease can be familial with a possible recessive inheritance mode with incomplete penetrance.4 Risk of sarcoidosis in monozygotic twins appears to be 80 times greater than that in the general population, which supports genetic factors accounting for two-thirds of disease susceptibility.5
Likely factors in the development of sarcoidosis
The exact cause of sarcoidosis is unknown, but we have insights into its pathogenesis and potential triggers.1,6-9 Genes involved are being identified: class I and II human leukocyte antigen (HLA) molecules are most consistently associated with risk of sarcoidosis. Environmental exposures can activate the innate immune system and precondition a susceptible individual to react to potential causative antigens in a highly polarized, antigen-specific Th1 immune response. The epithelioid granulomatous response involves local proinflammatory cytokine production and enhanced T-cell immunity at sites of inflammation.10 Granulomas generally form to confine pathogens, restrict inflammation, and protect surrounding tissue.11-13
ACCESS (A Case Control Etiologic Study of Sarcoidosis) identified several environmental exposures such as chemicals used in the agriculture industry, mold or mildew, and musty odors at work.14 Tobacco use was not associated with sarcoidosis.14 Recent studies have shown positive associations with service in the US Navy,15 metal working,16 firefighting,17 the handling of building supplies,18 and onsite exposure while assisting in rescue efforts at the World Trade Center disaster.19 Other data support the likelihood that specific environmental exposures associated with microbe-rich environments modestly increase the risk of sarcoidosis.14 Mycobacterial and propionibacterial DNA and RNA are potentially associated with sarcoidosis.20
Clinical manifestations are nonspecific
The diagnosis of sarcoidosis can be difficult and delayed due to diverse organ involvement and nonspecific presentations. TABLE 121-31 shows the diverse manifestations in a patient with suspected sarcoidosis. Around 50% of the patients are asymptomatic.23,24 Sarcoidosis is a diagnosis of exclusion, starting with a detailed history to rule out infections, occupational or environmental exposures, malignancies, and other possible disorders (TABLE 2).22

Diagnostic work-up

Radiologic studies
Chest x-ray (CXR) provides diagnostic and prognostic information in the evaluation of sarcoidosis using the Scadding classification system (FIGURE 1).21,25,32,33 Interobserver variability, especially between stages II and III and III and IV is the major limitation of this system.32 At presentation, radiographs are abnormal in approximately 90% of patients.34 Lymphadenopathy is the most common radiographic abnormality, occurring in more than two-thirds of cases, and pulmonary opacities (nodules and reticulation) with a middle to upper lobe predilection are present in 20% to 50% of patients.1,31,35 The nodules vary in size and can coalesce and cause alveolar collapse, thus producing consolidation.36 Linear opacities radiating laterally from the hilum into the middle and upper zones are characteristic in fibrotic disease.

Continue to: High-resoluton computed tomography
High-resolution computed tomography (HRCT). Micronodules in a perilymphatic distribution with upper lobe predominance combined with subcarinal and symmetrical hilar lymph node enlargement is practically diagnostic of sarcoidosis in the right clinical context. TABLE 321,23,25,32 and FIGURE 221,23,25,32 summarize the common CT chest findings of sarcoidosis.

Advanced imaging such as (18)F-fluorodeoxyglucose positron emission tomography (PET) and magnetic resonance imaging (MRI) are used in specialized settings for advanced pulmonary, cardiac, or neurosarcoidosis.

Tissue biopsy
Skin lesions (other than erythema nodosum), eye lesions, and peripheral lymph nodes are considered the safest extrapulmonary locations for biopsy.21,25 If pulmonary infiltrates or lymphadenopathy are present, or if extrapulmonary biopsy sites are not available, then flexible bronchoscopy with biopsy is the mainstay for tissue sampling.25
Bronchoalveolar lavage (BAL), transbronchial biopsy (TBB), endobronchial biopsy (EBB), and endobronchial ultrasound (EBUS) are invaluable modalities that have reduced the need for open lung biopsy. BAL in sarcoidosis can show lymphocytosis > 15% (nonspecific) and a CD4:CD8 lymphocyte ratio > 3.5 (specificity > 90%).21,22 TBB is more sensitive than EBB; however, sensitivity overall is heightened when both of them are combined. The advent of EBUS has increased the safety and efficiency of needle aspiration of mediastinal lymph nodes. Diagnostic yield of EBUS (~80%) is superior to that with TBB and EBB (~50%), especially in stage I and II sarcoidosis.37 The combination of EBUS with TBB improves the diagnostic yield to ~90%.37
The decision to obtain biopsy samples hinges on the nature of clinical and radiologic findings (FIGURE 3).22,25,26

Continue to: Laboratory studies
Laboratory studies
Multiple abnormalities may be seen in sarcoidosis, and specific lab tests may help support a diagnosis of sarcoidosis or detect organ-specific disease activity (TABLE 4).22,23,25,38 However, no consistently accurate biomarkers exist for use in clinical practice. An angiotensin-converting enzyme (ACE) level greater than 2 times the upper limit of normal may be helpful; however, sensitivity remains low, and genetic polymorphisms can influence the ACE level.25 Biomarkers sometimes used to assess disease activity are serum interleukin-2 receptor, neopterin, chitotriosidase, lysozyme, KL-6 glycoprotein, and amyloid A.21

Additional tests to assess specific features or organ involvement
Pulmonary function testing (PFT) is reviewed in detail below under “pulmonary sarcoidosis.”
Electrocardiogram (EKG)/transthoracic echocardiogram (TTE). EKG abnormalities—conduction disturbances, arrhythmias, or nonspecific ST segment and T-wave changes—are the most common nonspecific findings.30 TTE findings are also nonspecific but have value in assessing cardiac chamber size and function and myocardial involvement. TTE is indeed the most common screening modality for sarcoidosis-associated pulmonary hypertension (SAPH), which is definitively diagnosed by right heart catheterization (RHC). Further evaluation for cardiac sarcoidosis can be done with cardiac MRI or fluorodeoxyglucose PET in specialized settings.
Lumbar puncture (LP) may reveal lymphocytic infiltration in suspected neurosarcoidosis, but the finding is nonspecific and can reflect infection or malignancy. Oligoclonal bands may also be seen in about one-third of neurosarcoidosis cases, and it is imperative to rule out multiple sclerosis.28
Pulmonary sarcoidosis
Pulmonary sarcoidosis accounts for most of the morbidity, mortality, and health care use associated with sarcoidosis.39,40
Continue to: Pathology of early and advanced pulmonary sarcoidosis
Pathology of early and advanced pulmonary sarcoidosis
Sarcoidosis is characterized by coalescing, tightly clustered, nonnecrotizing granulomas in the lung (FIGURE 4), most often located along the lymphatic routes of the pleura, interlobular septa, and bronchovascular bundles.41 Granulomas contain epithelioid cells or multinucleated giant cells surrounded by a chronic lymphocytic infiltrate. Typically, intracytoplasmic inclusions, such as Schaumann bodies, asteroid bodies, and blue bodies of calcium oxalates are noted within giant cells.

In chronic disease, lymphocytic infiltrate vanishes and granulomas tend to become increasingly fibrotic and enlarge to form hyalinized nodules rich with densely eosinophilic collagen. In 10% to 30% of cases, the lungs undergo progressive fibrosis.40 Nonresolving inflammation appears to be the major cause of fibrosis and the peribronchovascular localization leading to marked bronchial distortion.
Clinical features, monitoring, and outcomes
Pulmonary involvement occurs in most patients with sarcoidosis, and subclinical pulmonary disease is generally present, even when extrathoracic manifestations predominate.23 Dry cough, dyspnea, and chest discomfort are the most common symptoms. Chest auscultation is usually unremarkable. Wheezing is more common in those with fibrosis and is attributed to airway-centric fibrosis.42 There is often a substantial delay between the onset of symptoms and the diagnosis of pulmonary sarcoidosis, as symptoms are nonspecific and might be mistaken for more common pulmonary diseases, such as asthma or chronic bronchitis.43
Since sarcoidosis can affect pulmonary parenchyma, interstitium, large and small airways, pulmonary vasculature, and respiratory muscles, the pattern of lung function impairment on PFT varies from normal to obstruction, restriction, isolated diffusion defect, or a combination of these. The typical physiologic abnormality is a restrictive ventilatory defect with a decreased diffusing capacity of the lung for carbon monoxide (DLCO). Extent of disease seen on HRCT correlates with level of restriction.44 Airway obstruction can be multifactorial and due to airway distortion (more likely to occur in fibrotic lung disease) and luminal disease.45-48 The 6-minute walk test and DLCO can also aid in the diagnosis of SAPH and advanced parenchymal lung disease.
While monitoring is done clinically and with testing (PFT and imaging) as needed, the optimal approach is unclear. Nevertheless, longitudinal monitoring with testing may provide useful management and prognostic information.40 Pulmonary function can remain stable in fibrotic sarcoidosis over extended periods and actually can improve in some patients.49 Serial spirometry, particularly forced vital capacity, is the most reliable tool for monitoring; when a decline in measurement occurs, chest radiography can elucidate the mechanism.50,51
Continue to: Because sarcoidosis is a multisystem disease...
Because sarcoidosis is a multisystem disease, caution needs to be exercised when evaluating a patient’s new or worsening respiratory symptoms to accurately determine the cause of symptoms and direct therapy accordingly. In addition to refractory inflammatory pulmonary disease, airway disease, infection, fibrosis, and SAPH, one needs to consider extrapulmonary involvement or complications such as cardiac or neurologic disease, musculoskeletal disease, depression, or fatigue. Adverse medication effects, deconditioning, or unrelated (or possibly related) disorders (eg pulmonary embolism) may be to blame.
Determining prognosis
Prognosis of sarcoidosis varies and depends on epidemiologic factors, clinical presentation, and course, as well as specific organ involvement. Patients may develop life-threatening pulmonary, cardiac, or neurologic complications. End-stage disease may require organ transplantation for eligible patients.
Most patients with pulmonary sarcoidosis experience clinical remission with minimal residual organ impairment and a favorable long-term outcome. Advanced pulmonary disease (known as APS) occurs in a small proportion of patients with sarcoidosis but accounts for most of the poor outcomes in sarcoidosis.40 APS is variably defined, but it generally includes pulmonary fibrosis, SAPH, and respiratory infection.
One percent to 5% of patients with sarcoidosis die from complications, and mortality is higher in women and African Americans.52 Mortality and morbidity may be increasing.53 The reasons behind these trends are unclear but could include true increases in disease incidence, better detection rates, greater severity of disease, or an aging population. Increased hospitalizations and health care use might be due to organ damage from granulomatous inflammation (and resultant fibrosis), complications associated with treatment, and psychosocial effects of the disease/treatment.
Management
Management consists primarily of anti-inflammatory or immunosuppressive therapies but can also include measures to address specific complications (such as fatigue) and organ transplant, as well as efforts to counter adverse medication effects. Other supportive and preventive measures may include, on a case-by-case basis, oxygen supplementation, vaccinations, or pulmonary rehabilitation. Details of these are found in other, more in-depth reviews on treatment; we will briefly review anti-inflammatory therapy, which forms the cornerstone of treatment in most patients with sarcoidosis.
Continue to: General approach to treatment decisions
General approach to treatment decisions. Anti-inflammatory therapy is used to reduce granulomatous inflammation, thereby preserving organ function and reducing symptoms. A decision to begin treatment is one shared with the patient and is based on symptoms and potential danger of organ system failure.54 Patients who are symptomatic or have progressive disease or physiologic impairment are generally candidates for treatment. Monitoring usually suffices for those who have minimal symptoms, stable disease, and preserved organ function.
Patients with pulmonary sarcoidosis at CXR stage 0 should not receive treatment, given that large, randomized trials have shown no meaningful benefit and that these patients have a high likelihood of spontaneous remission and excellent long-term prognosis.55-58 However, a subgroup of patients classified as stage 0/I on CXR may show parenchymal disease on HRCT,59 and, if more symptomatic, could be considered for treatment. For patients with stage II to IV pulmonary sarcoidosis with symptoms, there is good evidence that treatment may improve lung function and reduce dyspnea and fatigue.57,60-62
Corticosteroids are first-line treatment for most patients. Based on expert opinion, treatment of pulmonary sarcoidosis is generally started with oral prednisone (or an equivalent corticosteroid). A starting dose of 20 to 40 mg/d generally is sufficient for most patients. If the patient responds to initial treatment, prednisone dose is tapered over a period of months. If symptoms worsen during tapering, the minimum effective dose is maintained without further attempts at tapering. Treatment is continued for at least 3 to 6 months but it might be needed for longer durations; unfortunately, evidence-based guidelines are lacking.63 Once the patient goes into remission, close monitoring is done for possible relapses. Inhaled corticosteroids alone have not reduced symptoms or improved lung function in patients with pulmonary sarcoidosis.64-66
Steroid-sparing agents are added for many patients. For patients receiving chronic prednisone therapy (≥ 10 mg for > 6 months), steroid-sparing agents are considered to minimize the adverse effects of steroids or to better control the inflammatory activity of sarcoidosis. These agents must be carefully selected, and clinical and laboratory monitoring need to be done throughout therapy. TABLE 558,64,67-81

The management might be complicated for extrapulmonary, multi-organ, and advanced sarcoidosis (advanced pulmonary sarcoidosis, cardiac disease, neurosarcoidosis, lupus pernio, etc) when specialized testing, as well as a combination of corticosteroids and steroid-sparing agents (with higher doses or prolonged courses), might be needed. This should be performed at an expert sarcoidosis center, ideally in a multidisciplinary setting involving pulmonologists and/or rheumatologists, chest radiologists, and specialists as indicated, based on specific organ involvement.
Continue to: Research and future directions
Research and future directions
Key goals for research are identifying more accurate biomarkers of disease, improving diagnosis of multi-organ disease, determining validated endpoints of clinical trials in sarcoidosis, and developing treatments for refractory cases.
There is optimism and opportunity in the field of sarcoidosis overall. An example of an advancement is in the area of APS, as the severity and importance of this phenotype has been better understood. Worldwide registries and trials of pulmonary vasodilator therapy (bosentan, sildenafil, epoprostenol, and inhaled iloprost) in patients with SAPH without left ventricular dysfunction are promising.82-85 However, no benefit in survival has been shown.
RioSAPH is a double-blind, placebo-controlled trial of Riociguat (a stimulator of soluble guanylate cyclase) for SAPH (NCT02625558) that is closed to enrollment and undergoing data review. Similarly, results of the phase IV study of pirfenidone, an antifibrotic agent that was shown to decrease disease progression and deaths in idiopathic pulmonary fibrosis,86 are awaited in the near future.
Other potential directions being explored are multicenter patient registries and randomized controlled trials, analyses of existing databases, use of biobanking, and patient-centered outcome measures. Hopefully, the care of patients with sarcoidosis will become more evidence based with ongoing and upcoming research in this field.
CORRESPONDENCE
Rohit Gupta, MBBS, FCCP, 3401 North Broad Street, 7 Parkinson Pavilion, Philadelphia, PA 19140; rohit.gupta@tuhs.temple.edu
1. Costabel U, Hunninghake G. ATS/ERS/WASOG statement on sarcoidosis. Sarcoidosis Statement Committee. American Thoracic Society. European Respiratory Society. World Association for Sarcoidosis and Other Granulomatous Disorders. Eur Respir J. 1999;14:735-737.
2. Hillerdal G, Nöu E, Osterman K, et al. Sarcoidosis: epidemiology and prognosis. A 15-year European study. Am Rev Respir Dis. 1984;130:29-32.
3. Mirsaeidi M, Machado RF, Schraufnagel D, et al. Racial difference in sarcoidosis mortality in the United States. Chest. 2015;147:438-449.
4. Rybicki BA, Iannuzzi MC, Frederick MM, et al. Familial aggregation of sarcoidosis. A case-control etiologic study of sarcoidosis (ACCESS). Am J Resp Crit Care Med. 2001;164:2085-2091.
5. Sverrild A, Backer V, Kyvik KO, et al. Heredity in sarcoidosis:a registry-based twin study. Thorax. 2008;63:894.
6. Vuyst P, Dumortier P, Schandené L, et al. Sarcoidlike lung granulomatosis induced by aluminum dusts. Am Rev Respir Dis. 1987;135:493-497.
7. Werfel U, Schneider J, Rödelsperger K, et al. Sarcoid granulomatosis after zirconium exposure with multiple organ involvement. European Respir J. 1998;12:750.
8. Newman KL, Newman LS. Occupational causes of sarcoidosis. Curr Opin Allergy Clin Immunol. 2012;12:145-150.
9. Zissel G, Müller-Quernheim J. Specific antigen(s) in sarcoidosis:a link to autoimmunity? Eur Respir J. 2016;47:707-709.
10. Chen ES, Moller DR. Etiology of sarcoidosis. Clin Chest Med. 2008;29:365-377.
11. Agostini C, Adami F, Semenzato G. New pathogenetic insights into the sarcoid granuloma. Curr Opin Rheumatol. 2000;12:71-76.
12. Valentonyte R, Hampe J, Huse K, et al. Sarcoidosis is associated with a truncating splice site mutation in BTNL2. Nat Genet. 2005;37:357-364.
13. Rybicki BA, Walewski JL, Maliarik MJ, et al. The BTNL2 gene and sarcoidosis susceptibility in African Americans and Whites. Am J Hum Genet. 2005;77:491-499.
14. Newman LS, Rose CS, Bresnitz EA, et al. A case control etiologic study of sarcoidosis: environmental and occupational risk factors. Am J Respir Crit Care Med. 2004;170:1324-1330.
15. Gorham ED, Garland CF, Garland FC, et al. Trends and occupational associations in incidence of hospitalized pulmonary sarcoidosis and other lung diseases in Navy personnel: a 27-year historical prospective study, 1975-2001. Chest. 2004;126:1431-1438.
16. Kucera GP, Rybicki BA, Kirkey KL, et al. Occupational risk factors for sarcoidosis in African-American siblings. Chest. 2003;123:1527-1535.
17. Prezant DJ, Dhala A, Goldstein A, et al. The incidence, prevalence, and severity of sarcoidosis in New York City firefighters. Chest. 1999;116:1183-1193.
18. Barnard J, Rose C, Newman L, et al. Job and industry classifications associated with sarcoidosis in A Case–Control Etiologic Study of Sarcoidosis (ACCESS). J Occup Environ Med. 2005;47:226-234.
19. Izbicki G, Chavko R, Banauch GI, et al. World Trade Center “sarcoid-like” granulomatous pulmonary disease in New York City Fire Department rescue workers. Chest. 2007;131:1414-1423.
20. Eishi Y, Suga M, Ishige I, et al. Quantitative analysis of mycobacterial and propionibacterial DNA in lymph nodes of Japanese and European patients with sarcoidosis. J Clin Microbiol. 2002;40:198-204.
21. Valeyre D, Prasse A, Nunes H, et al. Sarcoidosis. Lancet. 2014;383:1155-1167.
22. Crouser ED, Maier LA, Wilson KC, et al. Diagnosis and detection of sarcoidosis. An official American Thoracic Society clinical practice guideline. Am J Respir Crit Care Med. 2020;201:e26-51.
23. Judson MA, ed. Pulmonary Sarcoidosis: A Guide for the Practicing Clinician. Springer; 2014.
24. Govender P, Berman JS. The diagnosis of sarcoidosis. Clin Chest Med. 2015;36:585-602.
25. Valeyre D, Bernaudin J-F, Uzunhan Y, et al. Clinical presentation of sarcoidosis and diagnostic work-up. Semin Resp Crit Care Med. 2014;35:336-351.
26. Judson MA. The clinical features of sarcoidosis: a comprehensive review. Clin Rev Allergy Immunol. 2015;49:63-78.
27. Wanat KA, Rosenbach M. Cutaneous sarcoidosis. Clin Chest Med. 2015;36:685-702.
28. Culver DA, Neto ML, Moss BP, et al. Neurosarcoidosis. Semin Resp Crit Care Med. 2017;38:499-513.
29. Pasadhika S, Rosenbaum JT. Ocular sarcoidosis. Clin Chest Med. 2015;36:669-683.
30. Sayah DM, Bradfield JS, Moriarty JM, et al. Cardiac involvement in sarcoidosis: evolving concepts in diagnosis and treatment. Semin Resp Crit Care Med. 2017;38:477-498.
31. Baughman RP, Teirstein AS, Judson MA, et al. Clinical characteristics of patients in a case control study of sarcoidosis. Am J Resp Crit Care. 2012;164:1885-1889.
32. Keijsers RG, Veltkamp M, Grutters JC. Chest imaging. Clin Chest Med. 2015;36:603-619.
33. Scadding J. Prognosis of intrathoracic sarcoidosis in England. A review of 136 cases after five years’ observation. Brit Med J. 1961;2:1165-1172.
34. Miller B, Putman C. The chest radiograph and sarcoidosis. Reevaluation of the chest radiograph in assessing activity of sarcoidosis: a preliminary communication. Sarcoidosis. 1985;2:85-90.
35. Loddenkemper R, Kloppenborg A, Schoenfeld N, et al. Clinical findings in 715 patients with newly detected pulmonary sarcoidosis--results of a cooperative study in former West Germany and Switzerland. WATL Study Group. Wissenschaftliche Arbeitsgemeinschaft für die Therapie von Lungenkrankheitan. Sarcoidosis Vasc Diffuse Lung Dis. 1998;15:178-182.
36. Calandriello L, Walsh SLF. Imaging for sarcoidosis. Semin Resp Crit Care Med. 2017;38:417-436.
37. Gupta D, Dadhwal DS, Agarwal R, et al. Endobronchial ultrasound-guided transbronchial needle aspiration vs conventional transbronchial needle aspiration in the diagnosis of sarcoidosis. Chest. 2014;146:547-556.
38. Baydur A. Recent developments in the physiological assessment of sarcoidosis: clinical implications. Curr Opin Pulm Med. 2012;18:499-505.
39. Jamilloux Y, Maucort-Boulch D, Kerever S, et al. Sarcoidosis-related mortality in France: a multiple-cause-of-death analysis. Eur Respir J. 2016;48:1700-1709.
40. Gupta R, Baughman RP. Advanced pulmonary sarcoidosis. Semin Respir Crit Care Med. 2020;41:700-715.
41. Rossi G, Cavazza A, Colby TV. Pathology of sarcoidosis. Clin Rev Allergy Immunol. 2015;49:36-44.
42. Hansell D, Milne D, Wilsher M, et al. Pulmonary sarcoidosis: morphologic associations of airflow obstruction at thin-section CT. Radiology. 1998;209:697-704.
43. Judson MA, Thompson BW, Rabin DL, et al. The diagnostic pathway to sarcoidosis. Chest. 2003;123:406-412.
44. Müller NL, Mawson JB, Mathieson JR, et al. Sarcoidosis: correlation of extent of disease at CT with clinical, functional, and radiographic findings. Radiology. 1989;171:613-618.
45. Harrison BDW, Shaylor JM, Stokes TC, et al. Airflow limitation in sarcoidosis—a study of pulmonary function in 107 patients with newly diagnosed disease. Resp Med. 1991;85:59-64.
46. Polychronopoulos VS, Prakash UBS. Airway Involvement in sarcoidosis. Chest. 2009;136:1371-1380.
47. Chambellan A, Turbie P, Nunes H, et al. Endoluminal stenosis of proximal bronchi in sarcoidosis: bronchoscopy, function, and evolution. Chest. 2005;127:472-481.
48. Handa T, Nagai S, Fushimi Y, et al. Clinical and radiographic indices associated with airflow limitation in patients with sarcoidosis. Chest. 2006;130:1851-1856.
49. Nardi A, Brillet P-Y, Letoumelin P, et al. Stage IV sarcoidosis: comparison of survival with the general population and causes of death. Eur Respir J. 2011;38:1368-1373.
50. Zappala CJ, Desai SR, Copley SJ, et al. Accuracy of individual variables in the monitoring of long-term change in pulmonary sarcoidosis as judged by serial high-resolution CT scan data. Chest. 2014;145:101-107.
51. Gafà G, Sverzellati N, Bonati E, et al. Follow-up in pulmonary sarcoidosis: comparison between HRCT and pulmonary function tests. Radiol Med. 2012;117:968-978.
52. Gerke AK. Morbidity and mortality in sarcoidosis. Curr Opin Pulm Med. 2014;20:472-478.
53. Kearney GD, Obi ON, Maddipati V, et al. Sarcoidosis deaths in the United States: 1999–2016. Respir Med. 2019;149:30-35.
54. Baughman RP, Judson M, Wells A. The indications for the treatment of sarcoidosis: Wells Law. Sarcoidosis Vasc Diffuse Lung Dis. 2017;34:280-282.
55. Nagai S, Shigematsu M, Hamada K, et al. Clinical courses and prognoses of pulmonary sarcoidosis. Curr Opin Pulm Med. 1999;5:293-298.
56. Neville E, Walker AN, James DG. Prognostic factors predicting the outcome of sarcoidosis: an analysis of 818 patients. Q J Med. 1983;52:525-533.
57. Bradley B, Branley HM, Egan JJ, et al. Interstitial lung disease guideline: the British Thoracic Society in collaboration with the Thoracic Society of Australia and the Irish Thoracic Society. Thorax. 2008;63(suppl 5):v1-v58.
58. Pietinalho A, Tukiainen P, Haahtela T, et al. Oral prednisolone followed by inhaled budesonide in newly diagnosed pulmonary sarcoidosis: a double-blind, placebo-controlled multicenter study. Finnish Pulmonary Sarcoidosis Group. Chest. 1999;116:424-431.
59. Oberstein A, von Zitzewitz H, Schweden F, et al. Non invasive evaluation of the inflammatory activity in sarcoidosis with high-resolution computed tomography. Sarcoidosis Vasc Diffuse Lung Dis. 1997;14:65-72.
60. Gibson G, Prescott RJ, Muers MF, et al. British Thoracic Society Sarcoidosis study: effects of long term corticosteroid treatment. Thorax. 1996;51:238-247.
61. Baughman RP, Nunes H. Therapy for sarcoidosis: evidence-based recommendations. Expert Rev Clin Immunol. 2012;8:95-103.
62. Pietinalho A, Tukiainen P, Haahtela T, et al. Early treatment of stage II sarcoidosis improves 5-year pulmonary function. Chest. 2002;121:24-31.
63. Rahaghi FF, Baughman RP, Saketkoo LA, et al. Delphi consensus recommendations for a treatment algorithm in pulmonary sarcoidosis. Eur Respir Rev. 2020;29:190146.
64. Baughman RP, Iannuzzi MC, Lower EE, et al. Use of fluticasone in acute symptomatic pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis. 2002;19:198-204.
65. du Bois RM, Greenhalgh PM, Southcott AM, et al. Randomized trial of inhaled fluticasone propionate in chronic stable pulmonary sarcoidosis: a pilot study. Eur Respir J. 1999;13:1345-1350.
66. Milman N, Graudal N, Grode G, Munch E. No effect of high‐dose inhaled steroids in pulmonary sarcoidosis: a double‐blind, placebo‐controlled study. J Intern Med. 1994;236:285-290.
67. Baughman RP, Winget DB, Lower EE. Methotrexate is steroid sparing in acute sarcoidosis: results of a double blind, randomized trial. Sarcoidosis Vasc Diffuse Lung Dis. 2000;17:60-66.
68. Vorselaars ADM, Wuyts WA, Vorselaars VMM, et al. Methotrexate vs azathioprine in second-line therapy of sarcoidosis. Chest. 2013;144:805-812.
69. Sahoo D, Bandyopadhyay D, Xu M, et al. Effectiveness and safety of leflunomide for pulmonary and extrapulmonary sarcoidosis. Eur Respir J. 2011;38:1145-1150.
70. Baughman RP, Drent M, Kavuru M, et al. Infliximab therapy in patients with chronic sarcoidosis and pulmonary involvement. Am J Resp Crit Care Med . 2006;174:795-802.
71. Rossman MD, Newman LS, Baughman RP, et al. A double-blinded, randomized, placebo-controlled trial of infliximab in subjects with active pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2006;23:201-208.
72. Selroos O, Sellergren T. Corticosteroid therapy of pulmonary sarcoidosis. A prospective evaluation of alternate day and daily dosage in stage II disease. Scand J Respir Dis . 1979;60:215-221.
73. Israel HL, Fouts DW, Beggs RA. A controlled trial of prednisone treatment of sarcoidosis. Am Rev Respir Dis . 1973;107:609-614.
74. Hamzeh N, Voelker A, Forssén A, et al. Efficacy of mycophenolate mofetil in sarcoidosis. Respir Med . 2014;108:1663-1669.
75. Brill A-K, Ott SR, Geiser T. Effect and safety of mycophenolate mofetil in chronic pulmonary sarcoidosis: a retrospective study. Respiration . 2013;86:376-383.
76. Baughman RP, Lower EE. Leflunomide for chronic sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2004;21:43-48.
77. Sweiss NJ, Noth I, Mirsaeidi M, et al. Efficacy results of a 52-week trial of adalimumab in the treatment of refractory sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2014;31:46-54.
78. Sweiss NJ, Lower EE, Mirsaeidi M, et al. Rituximab in the treatment of refractory pulmonary sarcoidosis. Eur Respir J . 2014;43:1525-1528.
79. Thatayatikom A, Thatayatikom S, White AJ. Infliximab treatment for severe granulomatous disease in common variable immunodeficiency: a case report and review of the literature. Ann Allergy Asthma Immunol . 2005;95:293-300.
80. Drake WP, Oswald-Richter K, Richmond BW, et al. Oral antimycobacterial therapy in chronic cutaneous sarcoidosis: a randomized, single-masked, placebo-controlled study. Jama Dermatol . 2013;149:1040-1049.
81. Drake WP, Richmond BW, Oswald-Richter K, et al. Effects of broad-spectrum antimycobacterial therapy on chronic pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2013;30:201-211.
82. Baughman RP, Culver DA, Cordova FC, et al. Bosentan for sarcoidosis-associated pulmonary hypertension: a double-blind placebo controlled randomized trial. Chest . 2014;145:810-817.
83. Baughman RP, Shlobin OA, Wells AU, et al. Clinical features of sarcoidosis associated pulmonary hypertension: results of a multi-national registry. Respir Med . 2018;139:72-78.
84. Fisher KA, Serlin DM, Wilson KC, et al. Sarcoidosis-associated pulmonary hypertension outcome with long-term epoprostenol treatment. Chest . 2006;130:1481-1488.
85. Baughman RP, Judson MA, Lower EE, et al. Inhaled iloprost for sarcoidosis associated pulmonary hypertension. Sarcoidosis Vasc Diffuse Lung Dis . 2009;26:110-120.
86. King TE, Bradford WZ, Castro-Bernardini S, et al. A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. N Engl J Med . 2014;370:2083-2092.
1. Costabel U, Hunninghake G. ATS/ERS/WASOG statement on sarcoidosis. Sarcoidosis Statement Committee. American Thoracic Society. European Respiratory Society. World Association for Sarcoidosis and Other Granulomatous Disorders. Eur Respir J. 1999;14:735-737.
2. Hillerdal G, Nöu E, Osterman K, et al. Sarcoidosis: epidemiology and prognosis. A 15-year European study. Am Rev Respir Dis. 1984;130:29-32.
3. Mirsaeidi M, Machado RF, Schraufnagel D, et al. Racial difference in sarcoidosis mortality in the United States. Chest. 2015;147:438-449.
4. Rybicki BA, Iannuzzi MC, Frederick MM, et al. Familial aggregation of sarcoidosis. A case-control etiologic study of sarcoidosis (ACCESS). Am J Resp Crit Care Med. 2001;164:2085-2091.
5. Sverrild A, Backer V, Kyvik KO, et al. Heredity in sarcoidosis:a registry-based twin study. Thorax. 2008;63:894.
6. Vuyst P, Dumortier P, Schandené L, et al. Sarcoidlike lung granulomatosis induced by aluminum dusts. Am Rev Respir Dis. 1987;135:493-497.
7. Werfel U, Schneider J, Rödelsperger K, et al. Sarcoid granulomatosis after zirconium exposure with multiple organ involvement. European Respir J. 1998;12:750.
8. Newman KL, Newman LS. Occupational causes of sarcoidosis. Curr Opin Allergy Clin Immunol. 2012;12:145-150.
9. Zissel G, Müller-Quernheim J. Specific antigen(s) in sarcoidosis:a link to autoimmunity? Eur Respir J. 2016;47:707-709.
10. Chen ES, Moller DR. Etiology of sarcoidosis. Clin Chest Med. 2008;29:365-377.
11. Agostini C, Adami F, Semenzato G. New pathogenetic insights into the sarcoid granuloma. Curr Opin Rheumatol. 2000;12:71-76.
12. Valentonyte R, Hampe J, Huse K, et al. Sarcoidosis is associated with a truncating splice site mutation in BTNL2. Nat Genet. 2005;37:357-364.
13. Rybicki BA, Walewski JL, Maliarik MJ, et al. The BTNL2 gene and sarcoidosis susceptibility in African Americans and Whites. Am J Hum Genet. 2005;77:491-499.
14. Newman LS, Rose CS, Bresnitz EA, et al. A case control etiologic study of sarcoidosis: environmental and occupational risk factors. Am J Respir Crit Care Med. 2004;170:1324-1330.
15. Gorham ED, Garland CF, Garland FC, et al. Trends and occupational associations in incidence of hospitalized pulmonary sarcoidosis and other lung diseases in Navy personnel: a 27-year historical prospective study, 1975-2001. Chest. 2004;126:1431-1438.
16. Kucera GP, Rybicki BA, Kirkey KL, et al. Occupational risk factors for sarcoidosis in African-American siblings. Chest. 2003;123:1527-1535.
17. Prezant DJ, Dhala A, Goldstein A, et al. The incidence, prevalence, and severity of sarcoidosis in New York City firefighters. Chest. 1999;116:1183-1193.
18. Barnard J, Rose C, Newman L, et al. Job and industry classifications associated with sarcoidosis in A Case–Control Etiologic Study of Sarcoidosis (ACCESS). J Occup Environ Med. 2005;47:226-234.
19. Izbicki G, Chavko R, Banauch GI, et al. World Trade Center “sarcoid-like” granulomatous pulmonary disease in New York City Fire Department rescue workers. Chest. 2007;131:1414-1423.
20. Eishi Y, Suga M, Ishige I, et al. Quantitative analysis of mycobacterial and propionibacterial DNA in lymph nodes of Japanese and European patients with sarcoidosis. J Clin Microbiol. 2002;40:198-204.
21. Valeyre D, Prasse A, Nunes H, et al. Sarcoidosis. Lancet. 2014;383:1155-1167.
22. Crouser ED, Maier LA, Wilson KC, et al. Diagnosis and detection of sarcoidosis. An official American Thoracic Society clinical practice guideline. Am J Respir Crit Care Med. 2020;201:e26-51.
23. Judson MA, ed. Pulmonary Sarcoidosis: A Guide for the Practicing Clinician. Springer; 2014.
24. Govender P, Berman JS. The diagnosis of sarcoidosis. Clin Chest Med. 2015;36:585-602.
25. Valeyre D, Bernaudin J-F, Uzunhan Y, et al. Clinical presentation of sarcoidosis and diagnostic work-up. Semin Resp Crit Care Med. 2014;35:336-351.
26. Judson MA. The clinical features of sarcoidosis: a comprehensive review. Clin Rev Allergy Immunol. 2015;49:63-78.
27. Wanat KA, Rosenbach M. Cutaneous sarcoidosis. Clin Chest Med. 2015;36:685-702.
28. Culver DA, Neto ML, Moss BP, et al. Neurosarcoidosis. Semin Resp Crit Care Med. 2017;38:499-513.
29. Pasadhika S, Rosenbaum JT. Ocular sarcoidosis. Clin Chest Med. 2015;36:669-683.
30. Sayah DM, Bradfield JS, Moriarty JM, et al. Cardiac involvement in sarcoidosis: evolving concepts in diagnosis and treatment. Semin Resp Crit Care Med. 2017;38:477-498.
31. Baughman RP, Teirstein AS, Judson MA, et al. Clinical characteristics of patients in a case control study of sarcoidosis. Am J Resp Crit Care. 2012;164:1885-1889.
32. Keijsers RG, Veltkamp M, Grutters JC. Chest imaging. Clin Chest Med. 2015;36:603-619.
33. Scadding J. Prognosis of intrathoracic sarcoidosis in England. A review of 136 cases after five years’ observation. Brit Med J. 1961;2:1165-1172.
34. Miller B, Putman C. The chest radiograph and sarcoidosis. Reevaluation of the chest radiograph in assessing activity of sarcoidosis: a preliminary communication. Sarcoidosis. 1985;2:85-90.
35. Loddenkemper R, Kloppenborg A, Schoenfeld N, et al. Clinical findings in 715 patients with newly detected pulmonary sarcoidosis--results of a cooperative study in former West Germany and Switzerland. WATL Study Group. Wissenschaftliche Arbeitsgemeinschaft für die Therapie von Lungenkrankheitan. Sarcoidosis Vasc Diffuse Lung Dis. 1998;15:178-182.
36. Calandriello L, Walsh SLF. Imaging for sarcoidosis. Semin Resp Crit Care Med. 2017;38:417-436.
37. Gupta D, Dadhwal DS, Agarwal R, et al. Endobronchial ultrasound-guided transbronchial needle aspiration vs conventional transbronchial needle aspiration in the diagnosis of sarcoidosis. Chest. 2014;146:547-556.
38. Baydur A. Recent developments in the physiological assessment of sarcoidosis: clinical implications. Curr Opin Pulm Med. 2012;18:499-505.
39. Jamilloux Y, Maucort-Boulch D, Kerever S, et al. Sarcoidosis-related mortality in France: a multiple-cause-of-death analysis. Eur Respir J. 2016;48:1700-1709.
40. Gupta R, Baughman RP. Advanced pulmonary sarcoidosis. Semin Respir Crit Care Med. 2020;41:700-715.
41. Rossi G, Cavazza A, Colby TV. Pathology of sarcoidosis. Clin Rev Allergy Immunol. 2015;49:36-44.
42. Hansell D, Milne D, Wilsher M, et al. Pulmonary sarcoidosis: morphologic associations of airflow obstruction at thin-section CT. Radiology. 1998;209:697-704.
43. Judson MA, Thompson BW, Rabin DL, et al. The diagnostic pathway to sarcoidosis. Chest. 2003;123:406-412.
44. Müller NL, Mawson JB, Mathieson JR, et al. Sarcoidosis: correlation of extent of disease at CT with clinical, functional, and radiographic findings. Radiology. 1989;171:613-618.
45. Harrison BDW, Shaylor JM, Stokes TC, et al. Airflow limitation in sarcoidosis—a study of pulmonary function in 107 patients with newly diagnosed disease. Resp Med. 1991;85:59-64.
46. Polychronopoulos VS, Prakash UBS. Airway Involvement in sarcoidosis. Chest. 2009;136:1371-1380.
47. Chambellan A, Turbie P, Nunes H, et al. Endoluminal stenosis of proximal bronchi in sarcoidosis: bronchoscopy, function, and evolution. Chest. 2005;127:472-481.
48. Handa T, Nagai S, Fushimi Y, et al. Clinical and radiographic indices associated with airflow limitation in patients with sarcoidosis. Chest. 2006;130:1851-1856.
49. Nardi A, Brillet P-Y, Letoumelin P, et al. Stage IV sarcoidosis: comparison of survival with the general population and causes of death. Eur Respir J. 2011;38:1368-1373.
50. Zappala CJ, Desai SR, Copley SJ, et al. Accuracy of individual variables in the monitoring of long-term change in pulmonary sarcoidosis as judged by serial high-resolution CT scan data. Chest. 2014;145:101-107.
51. Gafà G, Sverzellati N, Bonati E, et al. Follow-up in pulmonary sarcoidosis: comparison between HRCT and pulmonary function tests. Radiol Med. 2012;117:968-978.
52. Gerke AK. Morbidity and mortality in sarcoidosis. Curr Opin Pulm Med. 2014;20:472-478.
53. Kearney GD, Obi ON, Maddipati V, et al. Sarcoidosis deaths in the United States: 1999–2016. Respir Med. 2019;149:30-35.
54. Baughman RP, Judson M, Wells A. The indications for the treatment of sarcoidosis: Wells Law. Sarcoidosis Vasc Diffuse Lung Dis. 2017;34:280-282.
55. Nagai S, Shigematsu M, Hamada K, et al. Clinical courses and prognoses of pulmonary sarcoidosis. Curr Opin Pulm Med. 1999;5:293-298.
56. Neville E, Walker AN, James DG. Prognostic factors predicting the outcome of sarcoidosis: an analysis of 818 patients. Q J Med. 1983;52:525-533.
57. Bradley B, Branley HM, Egan JJ, et al. Interstitial lung disease guideline: the British Thoracic Society in collaboration with the Thoracic Society of Australia and the Irish Thoracic Society. Thorax. 2008;63(suppl 5):v1-v58.
58. Pietinalho A, Tukiainen P, Haahtela T, et al. Oral prednisolone followed by inhaled budesonide in newly diagnosed pulmonary sarcoidosis: a double-blind, placebo-controlled multicenter study. Finnish Pulmonary Sarcoidosis Group. Chest. 1999;116:424-431.
59. Oberstein A, von Zitzewitz H, Schweden F, et al. Non invasive evaluation of the inflammatory activity in sarcoidosis with high-resolution computed tomography. Sarcoidosis Vasc Diffuse Lung Dis. 1997;14:65-72.
60. Gibson G, Prescott RJ, Muers MF, et al. British Thoracic Society Sarcoidosis study: effects of long term corticosteroid treatment. Thorax. 1996;51:238-247.
61. Baughman RP, Nunes H. Therapy for sarcoidosis: evidence-based recommendations. Expert Rev Clin Immunol. 2012;8:95-103.
62. Pietinalho A, Tukiainen P, Haahtela T, et al. Early treatment of stage II sarcoidosis improves 5-year pulmonary function. Chest. 2002;121:24-31.
63. Rahaghi FF, Baughman RP, Saketkoo LA, et al. Delphi consensus recommendations for a treatment algorithm in pulmonary sarcoidosis. Eur Respir Rev. 2020;29:190146.
64. Baughman RP, Iannuzzi MC, Lower EE, et al. Use of fluticasone in acute symptomatic pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis. 2002;19:198-204.
65. du Bois RM, Greenhalgh PM, Southcott AM, et al. Randomized trial of inhaled fluticasone propionate in chronic stable pulmonary sarcoidosis: a pilot study. Eur Respir J. 1999;13:1345-1350.
66. Milman N, Graudal N, Grode G, Munch E. No effect of high‐dose inhaled steroids in pulmonary sarcoidosis: a double‐blind, placebo‐controlled study. J Intern Med. 1994;236:285-290.
67. Baughman RP, Winget DB, Lower EE. Methotrexate is steroid sparing in acute sarcoidosis: results of a double blind, randomized trial. Sarcoidosis Vasc Diffuse Lung Dis. 2000;17:60-66.
68. Vorselaars ADM, Wuyts WA, Vorselaars VMM, et al. Methotrexate vs azathioprine in second-line therapy of sarcoidosis. Chest. 2013;144:805-812.
69. Sahoo D, Bandyopadhyay D, Xu M, et al. Effectiveness and safety of leflunomide for pulmonary and extrapulmonary sarcoidosis. Eur Respir J. 2011;38:1145-1150.
70. Baughman RP, Drent M, Kavuru M, et al. Infliximab therapy in patients with chronic sarcoidosis and pulmonary involvement. Am J Resp Crit Care Med . 2006;174:795-802.
71. Rossman MD, Newman LS, Baughman RP, et al. A double-blinded, randomized, placebo-controlled trial of infliximab in subjects with active pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2006;23:201-208.
72. Selroos O, Sellergren T. Corticosteroid therapy of pulmonary sarcoidosis. A prospective evaluation of alternate day and daily dosage in stage II disease. Scand J Respir Dis . 1979;60:215-221.
73. Israel HL, Fouts DW, Beggs RA. A controlled trial of prednisone treatment of sarcoidosis. Am Rev Respir Dis . 1973;107:609-614.
74. Hamzeh N, Voelker A, Forssén A, et al. Efficacy of mycophenolate mofetil in sarcoidosis. Respir Med . 2014;108:1663-1669.
75. Brill A-K, Ott SR, Geiser T. Effect and safety of mycophenolate mofetil in chronic pulmonary sarcoidosis: a retrospective study. Respiration . 2013;86:376-383.
76. Baughman RP, Lower EE. Leflunomide for chronic sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2004;21:43-48.
77. Sweiss NJ, Noth I, Mirsaeidi M, et al. Efficacy results of a 52-week trial of adalimumab in the treatment of refractory sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2014;31:46-54.
78. Sweiss NJ, Lower EE, Mirsaeidi M, et al. Rituximab in the treatment of refractory pulmonary sarcoidosis. Eur Respir J . 2014;43:1525-1528.
79. Thatayatikom A, Thatayatikom S, White AJ. Infliximab treatment for severe granulomatous disease in common variable immunodeficiency: a case report and review of the literature. Ann Allergy Asthma Immunol . 2005;95:293-300.
80. Drake WP, Oswald-Richter K, Richmond BW, et al. Oral antimycobacterial therapy in chronic cutaneous sarcoidosis: a randomized, single-masked, placebo-controlled study. Jama Dermatol . 2013;149:1040-1049.
81. Drake WP, Richmond BW, Oswald-Richter K, et al. Effects of broad-spectrum antimycobacterial therapy on chronic pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2013;30:201-211.
82. Baughman RP, Culver DA, Cordova FC, et al. Bosentan for sarcoidosis-associated pulmonary hypertension: a double-blind placebo controlled randomized trial. Chest . 2014;145:810-817.
83. Baughman RP, Shlobin OA, Wells AU, et al. Clinical features of sarcoidosis associated pulmonary hypertension: results of a multi-national registry. Respir Med . 2018;139:72-78.
84. Fisher KA, Serlin DM, Wilson KC, et al. Sarcoidosis-associated pulmonary hypertension outcome with long-term epoprostenol treatment. Chest . 2006;130:1481-1488.
85. Baughman RP, Judson MA, Lower EE, et al. Inhaled iloprost for sarcoidosis associated pulmonary hypertension. Sarcoidosis Vasc Diffuse Lung Dis . 2009;26:110-120.
86. King TE, Bradford WZ, Castro-Bernardini S, et al. A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. N Engl J Med . 2014;370:2083-2092.
PRACTICE RECOMMENDATIONS
› Consider biopsy to aid in diagnosing sarcoidosis; it may be avoided with a high clinical suspicion for sarcoidosis (eg, Löfgren syndrome, lupus pernio, or Heerfordt syndrome). C
› Rule out alternative diagnoses such as infection, malignancy, collagen vascular disease, and vasculitis. C
› Identify extra-pulmonary organ involvement, as clinically indicated, by screening with a baseline eye examination; complete blood count; creatinine, alkaline phosphatase, and calcium levels; electrocardiogram, and other organ-specific studies. C
› Make a patient-centered decision whether to begin antiinflammatory treatment based on symptomatology and risk of organ failure or death. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
37-year-old man • cough • increasing shortness of breath • pleuritic chest pain • Dx?
THE CASE
A 37-year-old man with a history of asthma, schizoaffective disorder, and tobacco use (36 packs per year) presented to the clinic after 5 days of worsening cough, reproducible left-sided chest pain, and increasing shortness of breath. He also experienced chills, fatigue, nausea, and vomiting but was afebrile. The patient had not travelled recently nor had direct contact with anyone sick. He also denied intravenous (IV) drug use, alcohol use, and bloody sputum. Recently, he had intentionally lost weight, as recommended by his psychiatrist.
Medication review revealed that he was taking many central-acting agents for schizoaffective disorder, including alprazolam, aripiprazole, desvenlafaxine, and quetiapine. Due to his intermittent asthma since childhood, he used an albuterol inhaler as needed, which currently offered only minimal relief. He denied any history of hospitalization or intubation for asthma.
During the clinic visit, his blood pressure was 90/60 mm Hg and his heart rate was normal. His pulse oximetry was 92% on room air. On physical examination, he had normal-appearing dentition. Auscultation revealed bilateral expiratory wheezes with decreased breath sounds at the left lower lobe.
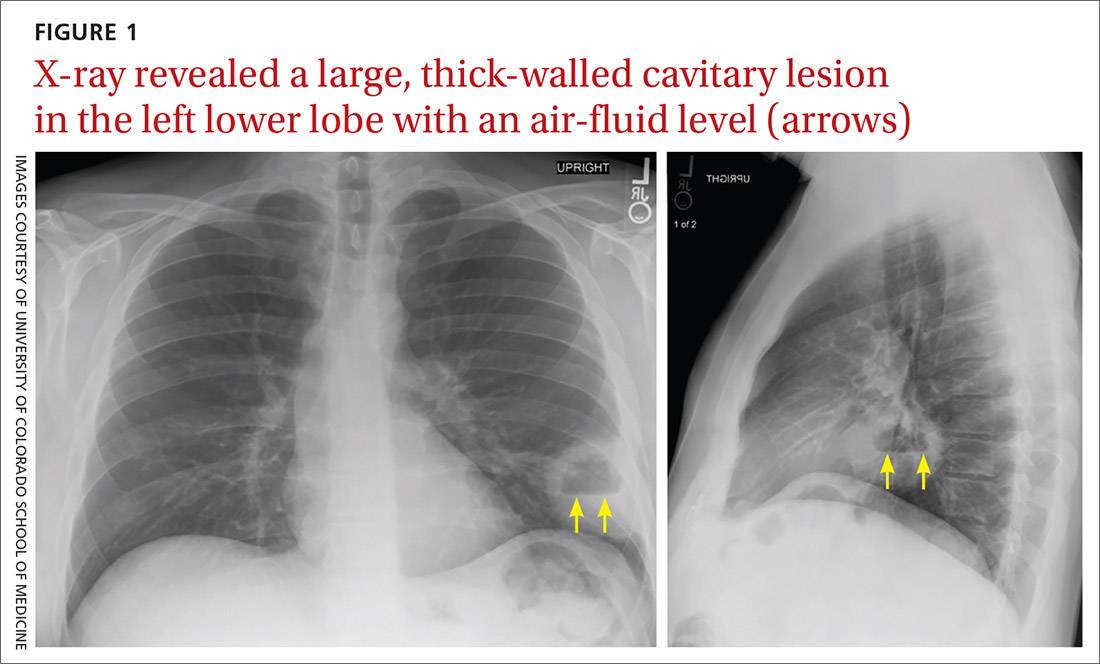
A plain chest radiograph (CXR) performed in the clinic (FIGURE 1) showed a large, thick-walled cavitary lesion with an air-fluid level in the left lower lobe. The patient was directly admitted to the Family Medicine Inpatient Service. Computed tomography (CT) of the chest with contrast was ordered to rule out empyema or malignancy. The chest CT confirmed the previous findings while also revealing a surrounding satellite nodularity in the left lower lobe (FIGURE 2). QuantiFERON-TB Gold and HIV tests were both negative.
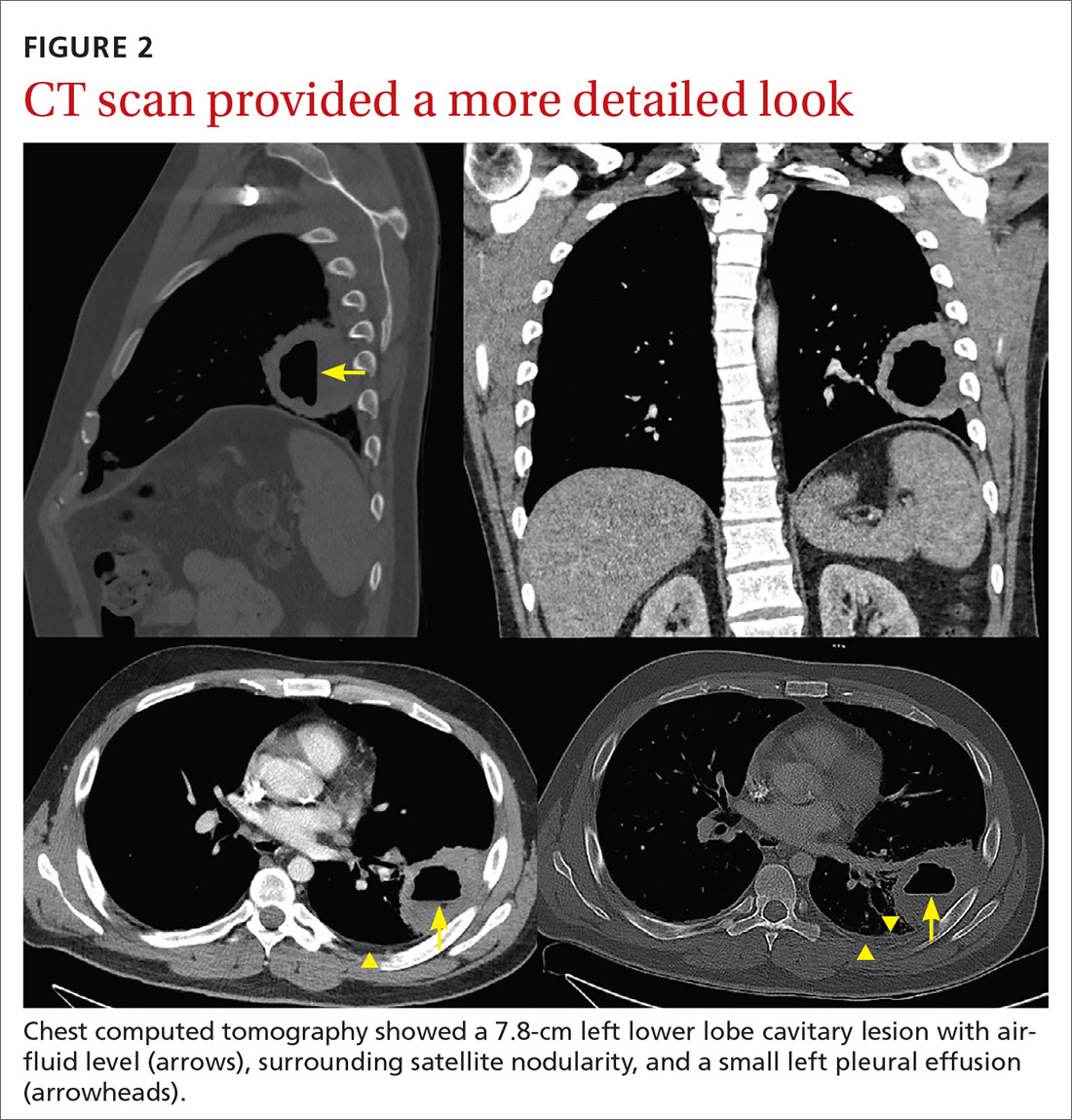
THE DIAGNOSIS
The patient was given a diagnosis of a lung abscess based on symptoms and imaging. An extensive smoking history, as well as multiple sedating medications, increased his likelihood of aspiration.
DISCUSSION
Lung abscess is the probable diagnosis in a patient with indolent infectious symptoms (cough, fever, night sweats) developing over days to weeks and a CXR finding of pulmonary opacity, often with an air-fluid level.1-4 A lung abscess is a circumscribed collection of pus in the lung parenchyma that develops as a result of microbial infection.4
Primary vs secondary abscess. Lung abscesses can be divided into 2 groups: primary and secondary abscesses. Primary abscesses (60%) occur without any other medical condition or in patients prone to aspiration.5 Secondary abscesses occur in the setting of a comorbid medical condition, such as lung disease, heart disease, bronchogenic neoplasm, or immunocompromised status.5
Continue to: With a primary lung abscess...
With a primary lung abscess, oropharyngeal contents are aspirated (generally while the patient is unconscious) and contain mixed flora.2 The aspirate typically migrates to the posterior segments of the upper lobes and to the superior segments of the lower lobes. These abscesses are usually singular and have an air-fluid level.1,2
Secondary lung abscesses occur in bronchial obstruction (by tumor, foreign body, or enlarged lymph nodes), with coexisting lung diseases (bronchiectasis, cystic fibrosis, infected pulmonary infarcts, lung contusion) or by direct spread (broncho-esophageal fistula, subphrenic abscess).6 Secondary abscesses are associated with a poorer prognosis, dependent on the patient’s general condition and underlying disease.7
What to rule out
The differential diagnosis of cavitary lung lesion includes tuberculosis, necrotizing pneumonia, bronchial carcinoma, pulmonary embolism, vasculitis (eg, Churg-Strauss syndrome), and localized pleural empyema.1,4 A CT scan is helpful to differentiate between a parenchymal lesion and pleural collection, which may not be as clear on CXR.1,4
Tuberculosis manifests with fatigue, weight loss, and night sweats; a chest CT will reveal a cavitating lesion (usually upper lobe) with a characteristic “rim sign” that includes caseous necrosis surrounded by a peripheral enhancing rim.8
Necrotizing pneumonia manifests as acute, fulminant infection. The most common causative organisms on sputum culture are Streptococcus pneumoniae, Staphylococcus aureus, Klebsiella pneumoniae, and Pseudomonas species. Plain radiography will reveal multiple cavities and often associated pleural effusion and empyema.9
Continue to: Excavating bronchogenic carcinomas
Excavating bronchogenic carcinomas differ from a lung abscess in that a patient with the latter is typically, but not always, febrile and has purulent sputum. On imaging, a bronchogenic carcinoma has a thicker and more irregular wall than a lung abscess.10
Treatment
When antibiotics first became available, penicillin was used to treat lung abscess.11 Then IV clindamycin became the drug of choice after 2 trials demonstrated its superiority to IV penicillin.12,13 More recently, clindamycin alone has fallen out of favor due to growing anaerobic resistance.14
Current therapy includes beta-lactam with beta-lactamase inhibitors.14 Lung abscesses are typically polymicrobial and thus carry different degrees of antibiotic resistance.15,16 If culture data are available, targeted therapy is preferred, especially for secondary abscesses.7 Antibiotic therapy is usually continued until a CXR reveals a small lesion or is clear, which may require several months of outpatient oral antibiotic therapy.4
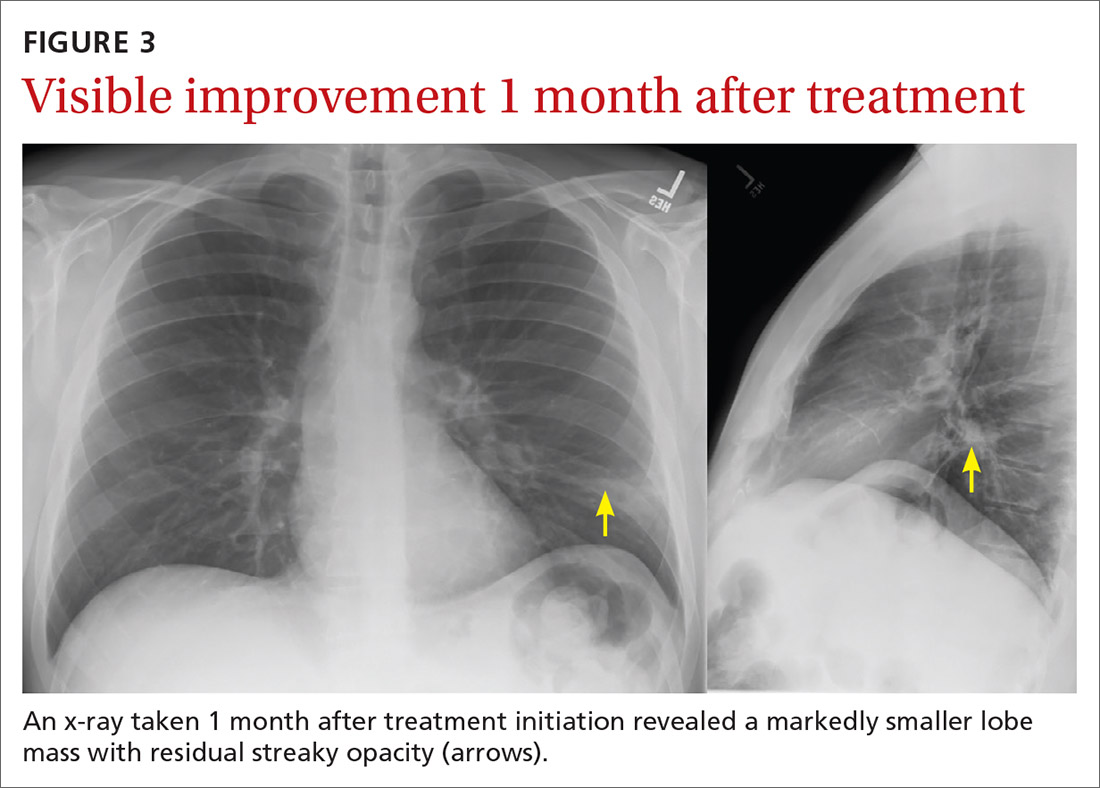
Our patient was treated with IV clindamycin for 3 days in the hospital. Clindamycin was chosen due to his penicillin allergy and started empirically without any culture data. He was transitioned to oral clindamycin and completed a total 3-week course as his CXR continued to show improvement (FIGURE 3). He did not undergo bronchoscopy. A follow-up CXR showed resolution of lung abscess at 9 months. (FIGURE 4).
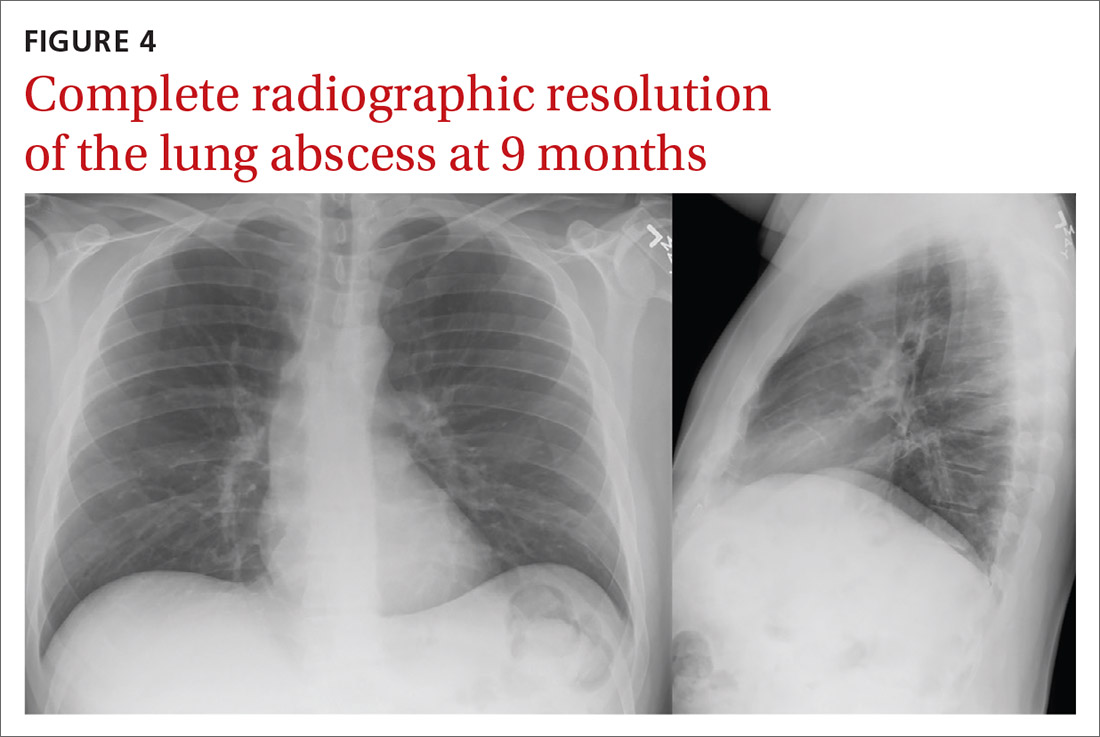
THE TAKEAWAY
All patients with lung abscesses should have sputum culture with gram stain done—ideally prior to starting antibiotics.3,4 Bronchoscopy should be considered for patients with atypical presentations or those who fail standard therapy, but may be used in other cases, as well.3
CORRESPONDENCE
Morteza Khodaee, MD, MPH, AFW Clinic, 3055 Roslyn Street, Denver, CO 80238; morteza.khodaee@cuanschutz.edu
1. Hassan M, Asciak R, Rizk R, et al. Lung abscess or empyema? Taking a closer look. Thorax. 2018;73:887-889. https://doi. org/10.1136/thoraxjnl-2018-211604
2. Moreira J da SM, Camargo J de JP, Felicetti JC, et al. Lung abscess: analysis of 252 consecutive cases diagnosed between 1968 and 2004. J Bras Pneumol. 2006;32:136-43. https://doi.org/10.1590/ s1806-37132006000200009
3. Schiza S, Siafakas NM. Clinical presentation and management of empyema, lung abscess and pleural effusion. Curr Opin Pulm Med. 2006;12:205-211. https://doi.org/10.1097/01. mcp.0000219270.73180.8b
4. Yazbeck MF, Dahdel M, Kalra A, et al. Lung abscess: update on microbiology and management. Am J Ther. 2014;21:217-221. https://doi.org/10.1097/MJT.0b013e3182383c9b
5. Nicolini A, Cilloniz C, Senarega R, et al. Lung abscess due to Streptococcus pneumoniae: a case series and brief review of the literature. Pneumonol Alergol Pol. 2014;82:276-285. https://doi. org/10.5603/PiAP.2014.0033
6. Puligandla PS, Laberge J-M. Respiratory infections: pneumonia, lung abscess, and empyema. Semin Pediatr Surg. 2008;17:42-52. https://doi.org/10.1053/j.sempedsurg.2007.10.007
7. Marra A, Hillejan L, Ukena D. [Management of Lung Abscess]. Zentralbl Chir. 2015;140 (suppl 1):S47-S53. https://doi. org/10.1055/s-0035-1557883
THE CASE
A 37-year-old man with a history of asthma, schizoaffective disorder, and tobacco use (36 packs per year) presented to the clinic after 5 days of worsening cough, reproducible left-sided chest pain, and increasing shortness of breath. He also experienced chills, fatigue, nausea, and vomiting but was afebrile. The patient had not travelled recently nor had direct contact with anyone sick. He also denied intravenous (IV) drug use, alcohol use, and bloody sputum. Recently, he had intentionally lost weight, as recommended by his psychiatrist.
Medication review revealed that he was taking many central-acting agents for schizoaffective disorder, including alprazolam, aripiprazole, desvenlafaxine, and quetiapine. Due to his intermittent asthma since childhood, he used an albuterol inhaler as needed, which currently offered only minimal relief. He denied any history of hospitalization or intubation for asthma.
During the clinic visit, his blood pressure was 90/60 mm Hg and his heart rate was normal. His pulse oximetry was 92% on room air. On physical examination, he had normal-appearing dentition. Auscultation revealed bilateral expiratory wheezes with decreased breath sounds at the left lower lobe.

A plain chest radiograph (CXR) performed in the clinic (FIGURE 1) showed a large, thick-walled cavitary lesion with an air-fluid level in the left lower lobe. The patient was directly admitted to the Family Medicine Inpatient Service. Computed tomography (CT) of the chest with contrast was ordered to rule out empyema or malignancy. The chest CT confirmed the previous findings while also revealing a surrounding satellite nodularity in the left lower lobe (FIGURE 2). QuantiFERON-TB Gold and HIV tests were both negative.

THE DIAGNOSIS
The patient was given a diagnosis of a lung abscess based on symptoms and imaging. An extensive smoking history, as well as multiple sedating medications, increased his likelihood of aspiration.
DISCUSSION
Lung abscess is the probable diagnosis in a patient with indolent infectious symptoms (cough, fever, night sweats) developing over days to weeks and a CXR finding of pulmonary opacity, often with an air-fluid level.1-4 A lung abscess is a circumscribed collection of pus in the lung parenchyma that develops as a result of microbial infection.4
Primary vs secondary abscess. Lung abscesses can be divided into 2 groups: primary and secondary abscesses. Primary abscesses (60%) occur without any other medical condition or in patients prone to aspiration.5 Secondary abscesses occur in the setting of a comorbid medical condition, such as lung disease, heart disease, bronchogenic neoplasm, or immunocompromised status.5
Continue to: With a primary lung abscess...
With a primary lung abscess, oropharyngeal contents are aspirated (generally while the patient is unconscious) and contain mixed flora.2 The aspirate typically migrates to the posterior segments of the upper lobes and to the superior segments of the lower lobes. These abscesses are usually singular and have an air-fluid level.1,2
Secondary lung abscesses occur in bronchial obstruction (by tumor, foreign body, or enlarged lymph nodes), with coexisting lung diseases (bronchiectasis, cystic fibrosis, infected pulmonary infarcts, lung contusion) or by direct spread (broncho-esophageal fistula, subphrenic abscess).6 Secondary abscesses are associated with a poorer prognosis, dependent on the patient’s general condition and underlying disease.7
What to rule out
The differential diagnosis of cavitary lung lesion includes tuberculosis, necrotizing pneumonia, bronchial carcinoma, pulmonary embolism, vasculitis (eg, Churg-Strauss syndrome), and localized pleural empyema.1,4 A CT scan is helpful to differentiate between a parenchymal lesion and pleural collection, which may not be as clear on CXR.1,4
Tuberculosis manifests with fatigue, weight loss, and night sweats; a chest CT will reveal a cavitating lesion (usually upper lobe) with a characteristic “rim sign” that includes caseous necrosis surrounded by a peripheral enhancing rim.8
Necrotizing pneumonia manifests as acute, fulminant infection. The most common causative organisms on sputum culture are Streptococcus pneumoniae, Staphylococcus aureus, Klebsiella pneumoniae, and Pseudomonas species. Plain radiography will reveal multiple cavities and often associated pleural effusion and empyema.9
Continue to: Excavating bronchogenic carcinomas
Excavating bronchogenic carcinomas differ from a lung abscess in that a patient with the latter is typically, but not always, febrile and has purulent sputum. On imaging, a bronchogenic carcinoma has a thicker and more irregular wall than a lung abscess.10
Treatment
When antibiotics first became available, penicillin was used to treat lung abscess.11 Then IV clindamycin became the drug of choice after 2 trials demonstrated its superiority to IV penicillin.12,13 More recently, clindamycin alone has fallen out of favor due to growing anaerobic resistance.14
Current therapy includes beta-lactam with beta-lactamase inhibitors.14 Lung abscesses are typically polymicrobial and thus carry different degrees of antibiotic resistance.15,16 If culture data are available, targeted therapy is preferred, especially for secondary abscesses.7 Antibiotic therapy is usually continued until a CXR reveals a small lesion or is clear, which may require several months of outpatient oral antibiotic therapy.4

Our patient was treated with IV clindamycin for 3 days in the hospital. Clindamycin was chosen due to his penicillin allergy and started empirically without any culture data. He was transitioned to oral clindamycin and completed a total 3-week course as his CXR continued to show improvement (FIGURE 3). He did not undergo bronchoscopy. A follow-up CXR showed resolution of lung abscess at 9 months. (FIGURE 4).

THE TAKEAWAY
All patients with lung abscesses should have sputum culture with gram stain done—ideally prior to starting antibiotics.3,4 Bronchoscopy should be considered for patients with atypical presentations or those who fail standard therapy, but may be used in other cases, as well.3
CORRESPONDENCE
Morteza Khodaee, MD, MPH, AFW Clinic, 3055 Roslyn Street, Denver, CO 80238; morteza.khodaee@cuanschutz.edu
THE CASE
A 37-year-old man with a history of asthma, schizoaffective disorder, and tobacco use (36 packs per year) presented to the clinic after 5 days of worsening cough, reproducible left-sided chest pain, and increasing shortness of breath. He also experienced chills, fatigue, nausea, and vomiting but was afebrile. The patient had not travelled recently nor had direct contact with anyone sick. He also denied intravenous (IV) drug use, alcohol use, and bloody sputum. Recently, he had intentionally lost weight, as recommended by his psychiatrist.
Medication review revealed that he was taking many central-acting agents for schizoaffective disorder, including alprazolam, aripiprazole, desvenlafaxine, and quetiapine. Due to his intermittent asthma since childhood, he used an albuterol inhaler as needed, which currently offered only minimal relief. He denied any history of hospitalization or intubation for asthma.
During the clinic visit, his blood pressure was 90/60 mm Hg and his heart rate was normal. His pulse oximetry was 92% on room air. On physical examination, he had normal-appearing dentition. Auscultation revealed bilateral expiratory wheezes with decreased breath sounds at the left lower lobe.

A plain chest radiograph (CXR) performed in the clinic (FIGURE 1) showed a large, thick-walled cavitary lesion with an air-fluid level in the left lower lobe. The patient was directly admitted to the Family Medicine Inpatient Service. Computed tomography (CT) of the chest with contrast was ordered to rule out empyema or malignancy. The chest CT confirmed the previous findings while also revealing a surrounding satellite nodularity in the left lower lobe (FIGURE 2). QuantiFERON-TB Gold and HIV tests were both negative.

THE DIAGNOSIS
The patient was given a diagnosis of a lung abscess based on symptoms and imaging. An extensive smoking history, as well as multiple sedating medications, increased his likelihood of aspiration.
DISCUSSION
Lung abscess is the probable diagnosis in a patient with indolent infectious symptoms (cough, fever, night sweats) developing over days to weeks and a CXR finding of pulmonary opacity, often with an air-fluid level.1-4 A lung abscess is a circumscribed collection of pus in the lung parenchyma that develops as a result of microbial infection.4
Primary vs secondary abscess. Lung abscesses can be divided into 2 groups: primary and secondary abscesses. Primary abscesses (60%) occur without any other medical condition or in patients prone to aspiration.5 Secondary abscesses occur in the setting of a comorbid medical condition, such as lung disease, heart disease, bronchogenic neoplasm, or immunocompromised status.5
Continue to: With a primary lung abscess...
With a primary lung abscess, oropharyngeal contents are aspirated (generally while the patient is unconscious) and contain mixed flora.2 The aspirate typically migrates to the posterior segments of the upper lobes and to the superior segments of the lower lobes. These abscesses are usually singular and have an air-fluid level.1,2
Secondary lung abscesses occur in bronchial obstruction (by tumor, foreign body, or enlarged lymph nodes), with coexisting lung diseases (bronchiectasis, cystic fibrosis, infected pulmonary infarcts, lung contusion) or by direct spread (broncho-esophageal fistula, subphrenic abscess).6 Secondary abscesses are associated with a poorer prognosis, dependent on the patient’s general condition and underlying disease.7
What to rule out
The differential diagnosis of cavitary lung lesion includes tuberculosis, necrotizing pneumonia, bronchial carcinoma, pulmonary embolism, vasculitis (eg, Churg-Strauss syndrome), and localized pleural empyema.1,4 A CT scan is helpful to differentiate between a parenchymal lesion and pleural collection, which may not be as clear on CXR.1,4
Tuberculosis manifests with fatigue, weight loss, and night sweats; a chest CT will reveal a cavitating lesion (usually upper lobe) with a characteristic “rim sign” that includes caseous necrosis surrounded by a peripheral enhancing rim.8
Necrotizing pneumonia manifests as acute, fulminant infection. The most common causative organisms on sputum culture are Streptococcus pneumoniae, Staphylococcus aureus, Klebsiella pneumoniae, and Pseudomonas species. Plain radiography will reveal multiple cavities and often associated pleural effusion and empyema.9
Continue to: Excavating bronchogenic carcinomas
Excavating bronchogenic carcinomas differ from a lung abscess in that a patient with the latter is typically, but not always, febrile and has purulent sputum. On imaging, a bronchogenic carcinoma has a thicker and more irregular wall than a lung abscess.10
Treatment
When antibiotics first became available, penicillin was used to treat lung abscess.11 Then IV clindamycin became the drug of choice after 2 trials demonstrated its superiority to IV penicillin.12,13 More recently, clindamycin alone has fallen out of favor due to growing anaerobic resistance.14
Current therapy includes beta-lactam with beta-lactamase inhibitors.14 Lung abscesses are typically polymicrobial and thus carry different degrees of antibiotic resistance.15,16 If culture data are available, targeted therapy is preferred, especially for secondary abscesses.7 Antibiotic therapy is usually continued until a CXR reveals a small lesion or is clear, which may require several months of outpatient oral antibiotic therapy.4

Our patient was treated with IV clindamycin for 3 days in the hospital. Clindamycin was chosen due to his penicillin allergy and started empirically without any culture data. He was transitioned to oral clindamycin and completed a total 3-week course as his CXR continued to show improvement (FIGURE 3). He did not undergo bronchoscopy. A follow-up CXR showed resolution of lung abscess at 9 months. (FIGURE 4).

THE TAKEAWAY
All patients with lung abscesses should have sputum culture with gram stain done—ideally prior to starting antibiotics.3,4 Bronchoscopy should be considered for patients with atypical presentations or those who fail standard therapy, but may be used in other cases, as well.3
CORRESPONDENCE
Morteza Khodaee, MD, MPH, AFW Clinic, 3055 Roslyn Street, Denver, CO 80238; morteza.khodaee@cuanschutz.edu
1. Hassan M, Asciak R, Rizk R, et al. Lung abscess or empyema? Taking a closer look. Thorax. 2018;73:887-889. https://doi. org/10.1136/thoraxjnl-2018-211604
2. Moreira J da SM, Camargo J de JP, Felicetti JC, et al. Lung abscess: analysis of 252 consecutive cases diagnosed between 1968 and 2004. J Bras Pneumol. 2006;32:136-43. https://doi.org/10.1590/ s1806-37132006000200009
3. Schiza S, Siafakas NM. Clinical presentation and management of empyema, lung abscess and pleural effusion. Curr Opin Pulm Med. 2006;12:205-211. https://doi.org/10.1097/01. mcp.0000219270.73180.8b
4. Yazbeck MF, Dahdel M, Kalra A, et al. Lung abscess: update on microbiology and management. Am J Ther. 2014;21:217-221. https://doi.org/10.1097/MJT.0b013e3182383c9b
5. Nicolini A, Cilloniz C, Senarega R, et al. Lung abscess due to Streptococcus pneumoniae: a case series and brief review of the literature. Pneumonol Alergol Pol. 2014;82:276-285. https://doi. org/10.5603/PiAP.2014.0033
6. Puligandla PS, Laberge J-M. Respiratory infections: pneumonia, lung abscess, and empyema. Semin Pediatr Surg. 2008;17:42-52. https://doi.org/10.1053/j.sempedsurg.2007.10.007
7. Marra A, Hillejan L, Ukena D. [Management of Lung Abscess]. Zentralbl Chir. 2015;140 (suppl 1):S47-S53. https://doi. org/10.1055/s-0035-1557883
1. Hassan M, Asciak R, Rizk R, et al. Lung abscess or empyema? Taking a closer look. Thorax. 2018;73:887-889. https://doi. org/10.1136/thoraxjnl-2018-211604
2. Moreira J da SM, Camargo J de JP, Felicetti JC, et al. Lung abscess: analysis of 252 consecutive cases diagnosed between 1968 and 2004. J Bras Pneumol. 2006;32:136-43. https://doi.org/10.1590/ s1806-37132006000200009
3. Schiza S, Siafakas NM. Clinical presentation and management of empyema, lung abscess and pleural effusion. Curr Opin Pulm Med. 2006;12:205-211. https://doi.org/10.1097/01. mcp.0000219270.73180.8b
4. Yazbeck MF, Dahdel M, Kalra A, et al. Lung abscess: update on microbiology and management. Am J Ther. 2014;21:217-221. https://doi.org/10.1097/MJT.0b013e3182383c9b
5. Nicolini A, Cilloniz C, Senarega R, et al. Lung abscess due to Streptococcus pneumoniae: a case series and brief review of the literature. Pneumonol Alergol Pol. 2014;82:276-285. https://doi. org/10.5603/PiAP.2014.0033
6. Puligandla PS, Laberge J-M. Respiratory infections: pneumonia, lung abscess, and empyema. Semin Pediatr Surg. 2008;17:42-52. https://doi.org/10.1053/j.sempedsurg.2007.10.007
7. Marra A, Hillejan L, Ukena D. [Management of Lung Abscess]. Zentralbl Chir. 2015;140 (suppl 1):S47-S53. https://doi. org/10.1055/s-0035-1557883
Black nonsmokers still at high risk for secondhand smoke exposure
Despite 30+ years of antismoking public policies and dramatic overall decline in secondhand smoke (SHS) exposure, .
No risk-free SHS exposure
Surendranath S. Shastri, MD, of MD Anderson Cancer Center, Houston, and colleagues underscored the U.S. Surgeon General’s determination that there is no risk-free level of SHS exposure in a recent JAMA Internal Medicine Research Letter.
“With the outbreak of the coronavirus disease 2019, which affects lung function, improving smoke-free policies to enhance air quality should be a growing priority,”they wrote.
Dr. Shastri and colleagues looked at 2011-2018 data from the National Health and Nutrition Examination Survey (NHANES), which detailed prevalence of SHS exposure in the U.S. population aged 3 years and older using interviews and biological specimens to test for cotinine levels. For the survey, nonsmokers having serum cotinine levels of 0.05 to 10 ng/mL were considered to have SHS exposure.
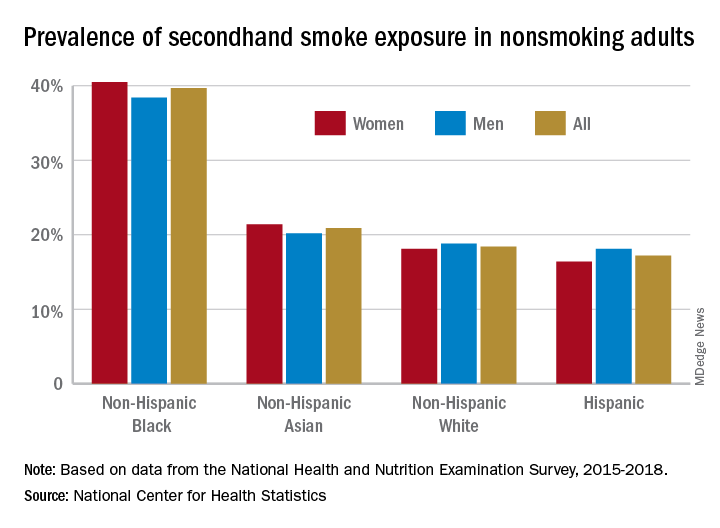
While the prevalence of SHS exposure among nonsmokers declined from 87.5% to 25.3% between 1988 and 2012, levels have stagnated since 2012 and racial and economic disparities are evident. Higher smoking rates, less knowledge about health risks, higher workplace exposure, greater likelihood of living in low-income, multi-unit housing, plus having their communities targeted by tobacco companies, may all help explain higher serum levels of cotinine in populations with lower socioeconomic status.
“Multivariable logistic regression identified younger age (odds ratio [OR], 1.88, for 12-19 years, and OR, 2.29, for 3-11 years), non-Hispanic Black race/ethnicity (OR, 2.75), less than high school education (OR, 1.59), and living below the poverty level (OR, 2.61) as risk factors for SHSe in the 2017-2018 cycle, with little change across all data cycles,” the researchers wrote.
Disparities in SHS exposure
A second report from NHANES data for 2015-2018, published in a National Center for Health Statistics Data Brief (No. 396, February 2021) showed that 20.8% of nonsmoking U.S. adults had SHS exposure, again with greater prevalence among non-Hispanic Black adults (39.7%), than for non-Hispanic White (18.4%), non-Hispanic Asian (20.9%), and Hispanic (17.2%) adults. Exposure was also greater in the younger age groups, with SHS rates for adults aged 18-39 years, 40-59 years, and ≥60 years at 25.6%, 19.1%, and 17.6%, respectively. Lower education (high school or less vs. some college education) and lower income levels were also associated with higher levels of SHS exposure. The investigators noted that among households with smokers, non-Hispanic Black adults are less likely to have complete smoking bans in homes, and among Medicaid or uninsured parents of any race or ethnicity, bans on smoking in family vehicles are less likely.
Overall, the prevalence of SHS exposure declined from 27.7% to 20.7% from 2009 to 2018, but the decreases were mediated by race and income.
SHS exposure in private spaces
A research brief from the Centers for Disease Control and Prevention on SHS exposure in homes and vehicles in the U.S. among middle and high school students also found a general decline in SHS exposure over 2011-2018 in homes (26.8%-20.9%; P < .001) and vehicles (30.2%-19.8%; P < .001). The findings, derived from the National Youth Tobacco Survey for 2011-2019, showed that no reduction occurred in homes among non-Hispanic Black students. Overall, a significant difference in home SHS exposure was observed by race/ethnicity: non-Hispanic Black (28.4%) and non-Hispanic White (27.4%) students both had a higher prevalence compared with Hispanic (20.0%) and non-Hispanic other (20.2%) students (P < .001).
Progress in reducing SHS exposure in public spaces has been made over the last 2 decades, with 27 states and more than 1,000 municipalities implementing comprehensive smoke-free laws that prohibit smoking in indoor public places, including workplaces, restaurants, and bars. While the prevalence of voluntary smoke-free home (83.7%) and vehicle (78.1%) rules has increased over time, private settings remain major sources of SHS exposure for many people, including youths. “Although SHS exposures have declined,” the authors wrote, “more than 6 million young people remain exposed to SHS in these private settings.”
In reviewing the data, Mary Cataletto, MD, FCCP, clinical professor of pediatrics at NYU Long Island School of Medicine, stated that these studies “highlight the need for implementation of smoke-free policies to reduce exposure to secondhand smoke, especially in homes and cars and with focused advocacy efforts in highly affected communities.”
Panagis Galiatsatos, MD, MHS, assistant professor of medicine at Johns Hopkins University, Baltimore, emphasized implementation of smoke-free policies but also treatment for smokers. “I’m not at all surprised by these statistics,” he noted in an interview. “Public health policies have helped us to get to where we are now, but there’s a reason that we have plateaued over the last decade. It’s hard to mitigate secondhand smoke exposure because the ones who are smoking now are the most refractory, challenging cases. ... You need good clinical interventions with counseling supported by pharmacological agents to help them if you want to stop secondhand smoke exposure.” He added, “You have to look at current smokers no differently than you look at patients with stage IV cancer – a group that requires a lot of resources to help them get through. Remember, all of them want to quit, but the promise of well-designed, precision-medicine strategies to help them quit has not been kept. Public health policy isn’t going to do it. We need to manage these patients clinically.”
The investigators had no conflict disclosures.
Despite 30+ years of antismoking public policies and dramatic overall decline in secondhand smoke (SHS) exposure, .
No risk-free SHS exposure
Surendranath S. Shastri, MD, of MD Anderson Cancer Center, Houston, and colleagues underscored the U.S. Surgeon General’s determination that there is no risk-free level of SHS exposure in a recent JAMA Internal Medicine Research Letter.
“With the outbreak of the coronavirus disease 2019, which affects lung function, improving smoke-free policies to enhance air quality should be a growing priority,”they wrote.
Dr. Shastri and colleagues looked at 2011-2018 data from the National Health and Nutrition Examination Survey (NHANES), which detailed prevalence of SHS exposure in the U.S. population aged 3 years and older using interviews and biological specimens to test for cotinine levels. For the survey, nonsmokers having serum cotinine levels of 0.05 to 10 ng/mL were considered to have SHS exposure.

While the prevalence of SHS exposure among nonsmokers declined from 87.5% to 25.3% between 1988 and 2012, levels have stagnated since 2012 and racial and economic disparities are evident. Higher smoking rates, less knowledge about health risks, higher workplace exposure, greater likelihood of living in low-income, multi-unit housing, plus having their communities targeted by tobacco companies, may all help explain higher serum levels of cotinine in populations with lower socioeconomic status.
“Multivariable logistic regression identified younger age (odds ratio [OR], 1.88, for 12-19 years, and OR, 2.29, for 3-11 years), non-Hispanic Black race/ethnicity (OR, 2.75), less than high school education (OR, 1.59), and living below the poverty level (OR, 2.61) as risk factors for SHSe in the 2017-2018 cycle, with little change across all data cycles,” the researchers wrote.
Disparities in SHS exposure
A second report from NHANES data for 2015-2018, published in a National Center for Health Statistics Data Brief (No. 396, February 2021) showed that 20.8% of nonsmoking U.S. adults had SHS exposure, again with greater prevalence among non-Hispanic Black adults (39.7%), than for non-Hispanic White (18.4%), non-Hispanic Asian (20.9%), and Hispanic (17.2%) adults. Exposure was also greater in the younger age groups, with SHS rates for adults aged 18-39 years, 40-59 years, and ≥60 years at 25.6%, 19.1%, and 17.6%, respectively. Lower education (high school or less vs. some college education) and lower income levels were also associated with higher levels of SHS exposure. The investigators noted that among households with smokers, non-Hispanic Black adults are less likely to have complete smoking bans in homes, and among Medicaid or uninsured parents of any race or ethnicity, bans on smoking in family vehicles are less likely.
Overall, the prevalence of SHS exposure declined from 27.7% to 20.7% from 2009 to 2018, but the decreases were mediated by race and income.
SHS exposure in private spaces
A research brief from the Centers for Disease Control and Prevention on SHS exposure in homes and vehicles in the U.S. among middle and high school students also found a general decline in SHS exposure over 2011-2018 in homes (26.8%-20.9%; P < .001) and vehicles (30.2%-19.8%; P < .001). The findings, derived from the National Youth Tobacco Survey for 2011-2019, showed that no reduction occurred in homes among non-Hispanic Black students. Overall, a significant difference in home SHS exposure was observed by race/ethnicity: non-Hispanic Black (28.4%) and non-Hispanic White (27.4%) students both had a higher prevalence compared with Hispanic (20.0%) and non-Hispanic other (20.2%) students (P < .001).
Progress in reducing SHS exposure in public spaces has been made over the last 2 decades, with 27 states and more than 1,000 municipalities implementing comprehensive smoke-free laws that prohibit smoking in indoor public places, including workplaces, restaurants, and bars. While the prevalence of voluntary smoke-free home (83.7%) and vehicle (78.1%) rules has increased over time, private settings remain major sources of SHS exposure for many people, including youths. “Although SHS exposures have declined,” the authors wrote, “more than 6 million young people remain exposed to SHS in these private settings.”
In reviewing the data, Mary Cataletto, MD, FCCP, clinical professor of pediatrics at NYU Long Island School of Medicine, stated that these studies “highlight the need for implementation of smoke-free policies to reduce exposure to secondhand smoke, especially in homes and cars and with focused advocacy efforts in highly affected communities.”
Panagis Galiatsatos, MD, MHS, assistant professor of medicine at Johns Hopkins University, Baltimore, emphasized implementation of smoke-free policies but also treatment for smokers. “I’m not at all surprised by these statistics,” he noted in an interview. “Public health policies have helped us to get to where we are now, but there’s a reason that we have plateaued over the last decade. It’s hard to mitigate secondhand smoke exposure because the ones who are smoking now are the most refractory, challenging cases. ... You need good clinical interventions with counseling supported by pharmacological agents to help them if you want to stop secondhand smoke exposure.” He added, “You have to look at current smokers no differently than you look at patients with stage IV cancer – a group that requires a lot of resources to help them get through. Remember, all of them want to quit, but the promise of well-designed, precision-medicine strategies to help them quit has not been kept. Public health policy isn’t going to do it. We need to manage these patients clinically.”
The investigators had no conflict disclosures.
Despite 30+ years of antismoking public policies and dramatic overall decline in secondhand smoke (SHS) exposure, .
No risk-free SHS exposure
Surendranath S. Shastri, MD, of MD Anderson Cancer Center, Houston, and colleagues underscored the U.S. Surgeon General’s determination that there is no risk-free level of SHS exposure in a recent JAMA Internal Medicine Research Letter.
“With the outbreak of the coronavirus disease 2019, which affects lung function, improving smoke-free policies to enhance air quality should be a growing priority,”they wrote.
Dr. Shastri and colleagues looked at 2011-2018 data from the National Health and Nutrition Examination Survey (NHANES), which detailed prevalence of SHS exposure in the U.S. population aged 3 years and older using interviews and biological specimens to test for cotinine levels. For the survey, nonsmokers having serum cotinine levels of 0.05 to 10 ng/mL were considered to have SHS exposure.

While the prevalence of SHS exposure among nonsmokers declined from 87.5% to 25.3% between 1988 and 2012, levels have stagnated since 2012 and racial and economic disparities are evident. Higher smoking rates, less knowledge about health risks, higher workplace exposure, greater likelihood of living in low-income, multi-unit housing, plus having their communities targeted by tobacco companies, may all help explain higher serum levels of cotinine in populations with lower socioeconomic status.
“Multivariable logistic regression identified younger age (odds ratio [OR], 1.88, for 12-19 years, and OR, 2.29, for 3-11 years), non-Hispanic Black race/ethnicity (OR, 2.75), less than high school education (OR, 1.59), and living below the poverty level (OR, 2.61) as risk factors for SHSe in the 2017-2018 cycle, with little change across all data cycles,” the researchers wrote.
Disparities in SHS exposure
A second report from NHANES data for 2015-2018, published in a National Center for Health Statistics Data Brief (No. 396, February 2021) showed that 20.8% of nonsmoking U.S. adults had SHS exposure, again with greater prevalence among non-Hispanic Black adults (39.7%), than for non-Hispanic White (18.4%), non-Hispanic Asian (20.9%), and Hispanic (17.2%) adults. Exposure was also greater in the younger age groups, with SHS rates for adults aged 18-39 years, 40-59 years, and ≥60 years at 25.6%, 19.1%, and 17.6%, respectively. Lower education (high school or less vs. some college education) and lower income levels were also associated with higher levels of SHS exposure. The investigators noted that among households with smokers, non-Hispanic Black adults are less likely to have complete smoking bans in homes, and among Medicaid or uninsured parents of any race or ethnicity, bans on smoking in family vehicles are less likely.
Overall, the prevalence of SHS exposure declined from 27.7% to 20.7% from 2009 to 2018, but the decreases were mediated by race and income.
SHS exposure in private spaces
A research brief from the Centers for Disease Control and Prevention on SHS exposure in homes and vehicles in the U.S. among middle and high school students also found a general decline in SHS exposure over 2011-2018 in homes (26.8%-20.9%; P < .001) and vehicles (30.2%-19.8%; P < .001). The findings, derived from the National Youth Tobacco Survey for 2011-2019, showed that no reduction occurred in homes among non-Hispanic Black students. Overall, a significant difference in home SHS exposure was observed by race/ethnicity: non-Hispanic Black (28.4%) and non-Hispanic White (27.4%) students both had a higher prevalence compared with Hispanic (20.0%) and non-Hispanic other (20.2%) students (P < .001).
Progress in reducing SHS exposure in public spaces has been made over the last 2 decades, with 27 states and more than 1,000 municipalities implementing comprehensive smoke-free laws that prohibit smoking in indoor public places, including workplaces, restaurants, and bars. While the prevalence of voluntary smoke-free home (83.7%) and vehicle (78.1%) rules has increased over time, private settings remain major sources of SHS exposure for many people, including youths. “Although SHS exposures have declined,” the authors wrote, “more than 6 million young people remain exposed to SHS in these private settings.”
In reviewing the data, Mary Cataletto, MD, FCCP, clinical professor of pediatrics at NYU Long Island School of Medicine, stated that these studies “highlight the need for implementation of smoke-free policies to reduce exposure to secondhand smoke, especially in homes and cars and with focused advocacy efforts in highly affected communities.”
Panagis Galiatsatos, MD, MHS, assistant professor of medicine at Johns Hopkins University, Baltimore, emphasized implementation of smoke-free policies but also treatment for smokers. “I’m not at all surprised by these statistics,” he noted in an interview. “Public health policies have helped us to get to where we are now, but there’s a reason that we have plateaued over the last decade. It’s hard to mitigate secondhand smoke exposure because the ones who are smoking now are the most refractory, challenging cases. ... You need good clinical interventions with counseling supported by pharmacological agents to help them if you want to stop secondhand smoke exposure.” He added, “You have to look at current smokers no differently than you look at patients with stage IV cancer – a group that requires a lot of resources to help them get through. Remember, all of them want to quit, but the promise of well-designed, precision-medicine strategies to help them quit has not been kept. Public health policy isn’t going to do it. We need to manage these patients clinically.”
The investigators had no conflict disclosures.
An alternative regimen to reduce risk of asthma exacerbations
ILLUSTRATIVE CASE
A 37-year-old woman with moderate persistent asthma, controlled on the ICS fluticasone (110 μg twice a day) presents to you for an annual exam. She uses her rescue albuterol inhaler a few times per month. Her last exacerbation was 2 years ago. She has never smoked. She is concerned about continuing to take an ICS every day. What alternative regimen would you recommend for this patient?
According to the Centers for Disease Control and Prevention, asthma affected 24.7 million children and adults in the United States in 2018, accounting for 9.8 million physician visits and 1.6 million emergency department (ED) visits.2 The National Institutes of Health (NIH) asthma care guidelines, updated in 2020, recommend a SABA prn as step 1 for intermittent asthma, along with nonpharmacologic management.3 Once a patient has persistent asthma, treatment escalation to step 2 calls for use of daily maintenance inhalers as the preferred treatment option.3
However, the 2020 Global Initiative for Asthma (GINA) warns that an as-needed SABA does not protect patients from severe exacerbations, and regular use of a SABA alone (> 3 inhalers/year) can increase the risk of exacerbations.4 A meta-analysis and systematic review from 2018 showed that using an ICS/LABA—scheduled and prn for rescue—had lower risk of asthma exacerbations compared with scheduled ICS/LABA with SABA prn for rescue in patients with moderate-to-severe persistent asthma.5 Interestingly, the updated 2020 NIH guidelines have adopted this strategy. SABA use prn is no longer recommended for rescue in mild and moderate persistent asthma, and the guidelines now suggest that ICS/LABA be used as rescue in addition to daily medication.3
Although evidence has been mounting for adding the as-needed ICS/LABA for rescue in patients on daily medication, the mainstay has been to provide a SABA prn for rescue use.5 Confusing matters more, evidence is emerging that as-needed ICS/LABA for rescue alone in certain patients is safe and effective. The randomized controlled Novel START study, an open-label, parallel-group study, compared ICS/LABA prn vs scheduled ICS with SABA prn vs SABA alone prn in adult patients with intermittent or mild persistent asthma.6 ICS/LABA prn prevented more exacerbations and provided better daily control than as-needed SABA alone.6 In addition, ICS/LABA as needed resulted in fewer severe exacerbations but potentially poorer daily control than ICS with SABA as needed.6
The PRACTICAL study investigated treatment of patients with intermittent, mild persistent, and moderate persistent asthma.1
STUDY SUMMARY
ICS/LABA prn reduced risk of severe exacerbations
The randomized controlled PRACTICAL study was a 52-week, open-label, parallel-group, superiority trial in New Zealand that compared as-needed ICS/LABA (n = 437) to scheduled ICS plus as-needed SABA (n = 448). Patients were 18 to 75 years old, with a diagnosis of asthma. Applying NIH guideline definitions, these patients would fall into intermittent, mild persistent, or moderate persistent asthma categories, and were on either as-needed SABA alone or a scheduled low- to moderate-dose ICS plus an as-needed SABA in the previous 12 weeks.
Patients on an as-needed SABA prerandomization had to have at least 1 of the following: (1) asthma symptoms or need for a SABA at least twice in the past 4 weeks; (2) at least 1 nighttime awakening due to asthma in the past 4 weeks; or (3) a severe exacerbation requiring oral corticosteroids in the past year. Patients on scheduled ICS plus SABA prn prerandomization were required to have either: (1) low or moderate ICS dosing with partly or well-controlled asthma; or (2) if uncontrolled, poor inhaler technique or adherence.
Continue to: Patients in the ICS/LABA group...
Patients in the ICS/LABA group were given budesonide 200 µg/formoterol 6 µg, 1 puff prn, and patients in the ICS plus as-needed SABA group were given budesonide 200 µg, 1 puff twice daily, and terbutaline 250 µg, 2 puffs prn. All patients received an asthma action plan that provided guidance on when to seek medical care if asthma worsened, as well as a log to note urgent medical visits and use of systemic corticosteroids. A subset of patients had adherence and dosing monitored by electronic inhaler usage monitors. Patients were seen at 0, 4, 16, 28, 40, and 52 weeks.
Outcomes. The primary outcome was the number of severe exacerbations per patient per year, defined as treatment with oral corticosteroids for ≥ 3 days or ED visit or hospital admission requiring systemic corticosteroids. Among the secondary outcomes were number of moderate and severe exacerbations per patient per year (defined as an unplanned medical visit: primary care, ED, hospital admission, and any duration of steroids); time to first severe exacerbation; assessment with the Asthma Control Questionnaire (ACQ-5); adverse outcomes; and quantity of ICS used (analysis done only for the subset with electronic inhaler monitoring).
ACQ-5 takes the mean of 5 questions assessing asthma control in the previous week, with each question ranging from 0 (no impairment) to 6 (maximum impairment). The statistician was blinded to the primary outcome.
Results. The rate of severe exacerbations per patient per year was 0.119 in the as-needed ICS/LABA group vs 0.172 in the scheduled ICS plus as-needed SABA group (relative rate [RR] = 0.69; 95% confidence interval [CI], 0.48–1.00). Time to first severe asthma exacerbation was longer in the as-needed ICS/LABA group (hazard ratio = 0.60; 95% CI, 0.40–0.91). The rate of moderate and severe exacerbations per patient per year was lower in the as-needed ICS/LABA group: 0.165 vs 0.237 (RR = 0.70; 95% CI, 0.51–0.95).
ACQ-5 scores were similar at all time points (mean difference = 0.07; 95% CI, –0.03 to 0.17). Adverse events were similar between groups (most commonly nasopharyngitis in both groups). Less ICS was used in the ICS/LABA group (difference = –126.5 µg per day; 95% CI, –171.0 to –81.9).
Continue to: WHAT'S NEW
WHAT’S NEW
Study lends support to recent recommendations
This study represents a compelling, real-world look at emerging asthma recommendations. This was the first comprehensive study to show that as-needed ICS/LABA therapy prevents more moderate and severe exacerbations and lengthens the time to first severe exacerbation, compared with scheduled ICS plus SABA prn in intermittent, mild persistent, or moderate persistent asthma. These data have been incorporated into the GINA guidelines, which recommend ICS/LABA prn for step 2.
CAVEATS
Potential bias in study design
The LABA used in this study was formoterol, which has a quicker onset than other LABAs. It is likely that not all LABAs can be used the same way, and both the NIH and GINA guidelines call it out specifically. Additionally, the study’s open-label design can introduce bias but may be the only way to simulate the real-world actions of our patients. Prior studies used placebo inhalers to keep participants and providers blinded but then could not capitalize on the behavior of using only an inhaler prn (as with the ICS/LABA of this study). Finally, there is discordance between the NIH and GINA asthma guidelines on how to use these data.
CHALLENGES TO IMPLEMENTATION
Cost of ICS/LABA may limit its use
Cost is the largest barrier to implementation. Budesonide costs 6 to 10 times more than albuterol per inhaler (retail price of $281-$427 vs $17-$92, respectively).7,8 However, cost differences are likely negated for patients already on a maintenance inhaler.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
1. Hardy J, Baggott C, Fingleton J, et al; PRACTICAL study team. Budesonide-formoterol reliever therapy versus maintenance budesonide plus terbutaline reliever therapy in adults with mild to moderate asthma (PRACTICAL): a 52-week, open-label, multicentre, superiority, randomised controlled trial. Lancet. 2019;394:919-928. Published correction appears in Lancet. 2020;395:1422.
2. Centers for Disease Control and Prevention. Summary Health Statistics: National Health Interview Survey, 2018. Accessed February 17, 2021. https://ftp.cdc.gov/pub/Health_Statistics/NCHS/NHIS/SHS/2018_SHS_Table_A-2.pdf
3. National Institutes of Health. National Heart, Lung, and Blood Institute. 2020 Focused Updates to the Asthma Management Guidelines: A Report from the National Asthma Education and Prevention Program Coordinating Committee Expert Panel Working Group. December 2020. Accessed February 17, 2021. www.nhlbi.nih.gov/health-topics/all-publications-and-resources/2020-focused-updates-asthma-management-guidelines
4. Global Initiative for Asthma (GINA). Global Strategy for Asthma Management and Prevention, 2020. Accessed February 17, 2021. www.ginasthma.org/
5. Sobieraj DM, Weeda ER, Nguyen E, et al. Association of inhaled corticosteroids and long-acting β-agonists as controller and quick relief therapy with exacerbations and symptom control in persistent asthma: a systematic review and meta-analysis. JAMA. 2018;319:1485-1496.
6. Beasley R, Holliday M, Reddel HK, et al; Novel START Study Team. Controlled trial of budesonide-formoterol as needed for mild asthma. N Engl J Med. 2019;380:2020-2030.
7. Albuterol. GoodRx. Accessed February 17, 2021. www.goodrx.com/albuterol
8. Budesonide/formoterol. GoodRx. Accessed February 17, 2021. www.goodrx.com/budesonide-formoterol
ILLUSTRATIVE CASE
A 37-year-old woman with moderate persistent asthma, controlled on the ICS fluticasone (110 μg twice a day) presents to you for an annual exam. She uses her rescue albuterol inhaler a few times per month. Her last exacerbation was 2 years ago. She has never smoked. She is concerned about continuing to take an ICS every day. What alternative regimen would you recommend for this patient?
According to the Centers for Disease Control and Prevention, asthma affected 24.7 million children and adults in the United States in 2018, accounting for 9.8 million physician visits and 1.6 million emergency department (ED) visits.2 The National Institutes of Health (NIH) asthma care guidelines, updated in 2020, recommend a SABA prn as step 1 for intermittent asthma, along with nonpharmacologic management.3 Once a patient has persistent asthma, treatment escalation to step 2 calls for use of daily maintenance inhalers as the preferred treatment option.3
However, the 2020 Global Initiative for Asthma (GINA) warns that an as-needed SABA does not protect patients from severe exacerbations, and regular use of a SABA alone (> 3 inhalers/year) can increase the risk of exacerbations.4 A meta-analysis and systematic review from 2018 showed that using an ICS/LABA—scheduled and prn for rescue—had lower risk of asthma exacerbations compared with scheduled ICS/LABA with SABA prn for rescue in patients with moderate-to-severe persistent asthma.5 Interestingly, the updated 2020 NIH guidelines have adopted this strategy. SABA use prn is no longer recommended for rescue in mild and moderate persistent asthma, and the guidelines now suggest that ICS/LABA be used as rescue in addition to daily medication.3
Although evidence has been mounting for adding the as-needed ICS/LABA for rescue in patients on daily medication, the mainstay has been to provide a SABA prn for rescue use.5 Confusing matters more, evidence is emerging that as-needed ICS/LABA for rescue alone in certain patients is safe and effective. The randomized controlled Novel START study, an open-label, parallel-group study, compared ICS/LABA prn vs scheduled ICS with SABA prn vs SABA alone prn in adult patients with intermittent or mild persistent asthma.6 ICS/LABA prn prevented more exacerbations and provided better daily control than as-needed SABA alone.6 In addition, ICS/LABA as needed resulted in fewer severe exacerbations but potentially poorer daily control than ICS with SABA as needed.6
The PRACTICAL study investigated treatment of patients with intermittent, mild persistent, and moderate persistent asthma.1
STUDY SUMMARY
ICS/LABA prn reduced risk of severe exacerbations
The randomized controlled PRACTICAL study was a 52-week, open-label, parallel-group, superiority trial in New Zealand that compared as-needed ICS/LABA (n = 437) to scheduled ICS plus as-needed SABA (n = 448). Patients were 18 to 75 years old, with a diagnosis of asthma. Applying NIH guideline definitions, these patients would fall into intermittent, mild persistent, or moderate persistent asthma categories, and were on either as-needed SABA alone or a scheduled low- to moderate-dose ICS plus an as-needed SABA in the previous 12 weeks.
Patients on an as-needed SABA prerandomization had to have at least 1 of the following: (1) asthma symptoms or need for a SABA at least twice in the past 4 weeks; (2) at least 1 nighttime awakening due to asthma in the past 4 weeks; or (3) a severe exacerbation requiring oral corticosteroids in the past year. Patients on scheduled ICS plus SABA prn prerandomization were required to have either: (1) low or moderate ICS dosing with partly or well-controlled asthma; or (2) if uncontrolled, poor inhaler technique or adherence.
Continue to: Patients in the ICS/LABA group...
Patients in the ICS/LABA group were given budesonide 200 µg/formoterol 6 µg, 1 puff prn, and patients in the ICS plus as-needed SABA group were given budesonide 200 µg, 1 puff twice daily, and terbutaline 250 µg, 2 puffs prn. All patients received an asthma action plan that provided guidance on when to seek medical care if asthma worsened, as well as a log to note urgent medical visits and use of systemic corticosteroids. A subset of patients had adherence and dosing monitored by electronic inhaler usage monitors. Patients were seen at 0, 4, 16, 28, 40, and 52 weeks.
Outcomes. The primary outcome was the number of severe exacerbations per patient per year, defined as treatment with oral corticosteroids for ≥ 3 days or ED visit or hospital admission requiring systemic corticosteroids. Among the secondary outcomes were number of moderate and severe exacerbations per patient per year (defined as an unplanned medical visit: primary care, ED, hospital admission, and any duration of steroids); time to first severe exacerbation; assessment with the Asthma Control Questionnaire (ACQ-5); adverse outcomes; and quantity of ICS used (analysis done only for the subset with electronic inhaler monitoring).
ACQ-5 takes the mean of 5 questions assessing asthma control in the previous week, with each question ranging from 0 (no impairment) to 6 (maximum impairment). The statistician was blinded to the primary outcome.
Results. The rate of severe exacerbations per patient per year was 0.119 in the as-needed ICS/LABA group vs 0.172 in the scheduled ICS plus as-needed SABA group (relative rate [RR] = 0.69; 95% confidence interval [CI], 0.48–1.00). Time to first severe asthma exacerbation was longer in the as-needed ICS/LABA group (hazard ratio = 0.60; 95% CI, 0.40–0.91). The rate of moderate and severe exacerbations per patient per year was lower in the as-needed ICS/LABA group: 0.165 vs 0.237 (RR = 0.70; 95% CI, 0.51–0.95).
ACQ-5 scores were similar at all time points (mean difference = 0.07; 95% CI, –0.03 to 0.17). Adverse events were similar between groups (most commonly nasopharyngitis in both groups). Less ICS was used in the ICS/LABA group (difference = –126.5 µg per day; 95% CI, –171.0 to –81.9).
Continue to: WHAT'S NEW
WHAT’S NEW
Study lends support to recent recommendations
This study represents a compelling, real-world look at emerging asthma recommendations. This was the first comprehensive study to show that as-needed ICS/LABA therapy prevents more moderate and severe exacerbations and lengthens the time to first severe exacerbation, compared with scheduled ICS plus SABA prn in intermittent, mild persistent, or moderate persistent asthma. These data have been incorporated into the GINA guidelines, which recommend ICS/LABA prn for step 2.
CAVEATS
Potential bias in study design
The LABA used in this study was formoterol, which has a quicker onset than other LABAs. It is likely that not all LABAs can be used the same way, and both the NIH and GINA guidelines call it out specifically. Additionally, the study’s open-label design can introduce bias but may be the only way to simulate the real-world actions of our patients. Prior studies used placebo inhalers to keep participants and providers blinded but then could not capitalize on the behavior of using only an inhaler prn (as with the ICS/LABA of this study). Finally, there is discordance between the NIH and GINA asthma guidelines on how to use these data.
CHALLENGES TO IMPLEMENTATION
Cost of ICS/LABA may limit its use
Cost is the largest barrier to implementation. Budesonide costs 6 to 10 times more than albuterol per inhaler (retail price of $281-$427 vs $17-$92, respectively).7,8 However, cost differences are likely negated for patients already on a maintenance inhaler.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
ILLUSTRATIVE CASE
A 37-year-old woman with moderate persistent asthma, controlled on the ICS fluticasone (110 μg twice a day) presents to you for an annual exam. She uses her rescue albuterol inhaler a few times per month. Her last exacerbation was 2 years ago. She has never smoked. She is concerned about continuing to take an ICS every day. What alternative regimen would you recommend for this patient?
According to the Centers for Disease Control and Prevention, asthma affected 24.7 million children and adults in the United States in 2018, accounting for 9.8 million physician visits and 1.6 million emergency department (ED) visits.2 The National Institutes of Health (NIH) asthma care guidelines, updated in 2020, recommend a SABA prn as step 1 for intermittent asthma, along with nonpharmacologic management.3 Once a patient has persistent asthma, treatment escalation to step 2 calls for use of daily maintenance inhalers as the preferred treatment option.3
However, the 2020 Global Initiative for Asthma (GINA) warns that an as-needed SABA does not protect patients from severe exacerbations, and regular use of a SABA alone (> 3 inhalers/year) can increase the risk of exacerbations.4 A meta-analysis and systematic review from 2018 showed that using an ICS/LABA—scheduled and prn for rescue—had lower risk of asthma exacerbations compared with scheduled ICS/LABA with SABA prn for rescue in patients with moderate-to-severe persistent asthma.5 Interestingly, the updated 2020 NIH guidelines have adopted this strategy. SABA use prn is no longer recommended for rescue in mild and moderate persistent asthma, and the guidelines now suggest that ICS/LABA be used as rescue in addition to daily medication.3
Although evidence has been mounting for adding the as-needed ICS/LABA for rescue in patients on daily medication, the mainstay has been to provide a SABA prn for rescue use.5 Confusing matters more, evidence is emerging that as-needed ICS/LABA for rescue alone in certain patients is safe and effective. The randomized controlled Novel START study, an open-label, parallel-group study, compared ICS/LABA prn vs scheduled ICS with SABA prn vs SABA alone prn in adult patients with intermittent or mild persistent asthma.6 ICS/LABA prn prevented more exacerbations and provided better daily control than as-needed SABA alone.6 In addition, ICS/LABA as needed resulted in fewer severe exacerbations but potentially poorer daily control than ICS with SABA as needed.6
The PRACTICAL study investigated treatment of patients with intermittent, mild persistent, and moderate persistent asthma.1
STUDY SUMMARY
ICS/LABA prn reduced risk of severe exacerbations
The randomized controlled PRACTICAL study was a 52-week, open-label, parallel-group, superiority trial in New Zealand that compared as-needed ICS/LABA (n = 437) to scheduled ICS plus as-needed SABA (n = 448). Patients were 18 to 75 years old, with a diagnosis of asthma. Applying NIH guideline definitions, these patients would fall into intermittent, mild persistent, or moderate persistent asthma categories, and were on either as-needed SABA alone or a scheduled low- to moderate-dose ICS plus an as-needed SABA in the previous 12 weeks.
Patients on an as-needed SABA prerandomization had to have at least 1 of the following: (1) asthma symptoms or need for a SABA at least twice in the past 4 weeks; (2) at least 1 nighttime awakening due to asthma in the past 4 weeks; or (3) a severe exacerbation requiring oral corticosteroids in the past year. Patients on scheduled ICS plus SABA prn prerandomization were required to have either: (1) low or moderate ICS dosing with partly or well-controlled asthma; or (2) if uncontrolled, poor inhaler technique or adherence.
Continue to: Patients in the ICS/LABA group...
Patients in the ICS/LABA group were given budesonide 200 µg/formoterol 6 µg, 1 puff prn, and patients in the ICS plus as-needed SABA group were given budesonide 200 µg, 1 puff twice daily, and terbutaline 250 µg, 2 puffs prn. All patients received an asthma action plan that provided guidance on when to seek medical care if asthma worsened, as well as a log to note urgent medical visits and use of systemic corticosteroids. A subset of patients had adherence and dosing monitored by electronic inhaler usage monitors. Patients were seen at 0, 4, 16, 28, 40, and 52 weeks.
Outcomes. The primary outcome was the number of severe exacerbations per patient per year, defined as treatment with oral corticosteroids for ≥ 3 days or ED visit or hospital admission requiring systemic corticosteroids. Among the secondary outcomes were number of moderate and severe exacerbations per patient per year (defined as an unplanned medical visit: primary care, ED, hospital admission, and any duration of steroids); time to first severe exacerbation; assessment with the Asthma Control Questionnaire (ACQ-5); adverse outcomes; and quantity of ICS used (analysis done only for the subset with electronic inhaler monitoring).
ACQ-5 takes the mean of 5 questions assessing asthma control in the previous week, with each question ranging from 0 (no impairment) to 6 (maximum impairment). The statistician was blinded to the primary outcome.
Results. The rate of severe exacerbations per patient per year was 0.119 in the as-needed ICS/LABA group vs 0.172 in the scheduled ICS plus as-needed SABA group (relative rate [RR] = 0.69; 95% confidence interval [CI], 0.48–1.00). Time to first severe asthma exacerbation was longer in the as-needed ICS/LABA group (hazard ratio = 0.60; 95% CI, 0.40–0.91). The rate of moderate and severe exacerbations per patient per year was lower in the as-needed ICS/LABA group: 0.165 vs 0.237 (RR = 0.70; 95% CI, 0.51–0.95).
ACQ-5 scores were similar at all time points (mean difference = 0.07; 95% CI, –0.03 to 0.17). Adverse events were similar between groups (most commonly nasopharyngitis in both groups). Less ICS was used in the ICS/LABA group (difference = –126.5 µg per day; 95% CI, –171.0 to –81.9).
Continue to: WHAT'S NEW
WHAT’S NEW
Study lends support to recent recommendations
This study represents a compelling, real-world look at emerging asthma recommendations. This was the first comprehensive study to show that as-needed ICS/LABA therapy prevents more moderate and severe exacerbations and lengthens the time to first severe exacerbation, compared with scheduled ICS plus SABA prn in intermittent, mild persistent, or moderate persistent asthma. These data have been incorporated into the GINA guidelines, which recommend ICS/LABA prn for step 2.
CAVEATS
Potential bias in study design
The LABA used in this study was formoterol, which has a quicker onset than other LABAs. It is likely that not all LABAs can be used the same way, and both the NIH and GINA guidelines call it out specifically. Additionally, the study’s open-label design can introduce bias but may be the only way to simulate the real-world actions of our patients. Prior studies used placebo inhalers to keep participants and providers blinded but then could not capitalize on the behavior of using only an inhaler prn (as with the ICS/LABA of this study). Finally, there is discordance between the NIH and GINA asthma guidelines on how to use these data.
CHALLENGES TO IMPLEMENTATION
Cost of ICS/LABA may limit its use
Cost is the largest barrier to implementation. Budesonide costs 6 to 10 times more than albuterol per inhaler (retail price of $281-$427 vs $17-$92, respectively).7,8 However, cost differences are likely negated for patients already on a maintenance inhaler.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
1. Hardy J, Baggott C, Fingleton J, et al; PRACTICAL study team. Budesonide-formoterol reliever therapy versus maintenance budesonide plus terbutaline reliever therapy in adults with mild to moderate asthma (PRACTICAL): a 52-week, open-label, multicentre, superiority, randomised controlled trial. Lancet. 2019;394:919-928. Published correction appears in Lancet. 2020;395:1422.
2. Centers for Disease Control and Prevention. Summary Health Statistics: National Health Interview Survey, 2018. Accessed February 17, 2021. https://ftp.cdc.gov/pub/Health_Statistics/NCHS/NHIS/SHS/2018_SHS_Table_A-2.pdf
3. National Institutes of Health. National Heart, Lung, and Blood Institute. 2020 Focused Updates to the Asthma Management Guidelines: A Report from the National Asthma Education and Prevention Program Coordinating Committee Expert Panel Working Group. December 2020. Accessed February 17, 2021. www.nhlbi.nih.gov/health-topics/all-publications-and-resources/2020-focused-updates-asthma-management-guidelines
4. Global Initiative for Asthma (GINA). Global Strategy for Asthma Management and Prevention, 2020. Accessed February 17, 2021. www.ginasthma.org/
5. Sobieraj DM, Weeda ER, Nguyen E, et al. Association of inhaled corticosteroids and long-acting β-agonists as controller and quick relief therapy with exacerbations and symptom control in persistent asthma: a systematic review and meta-analysis. JAMA. 2018;319:1485-1496.
6. Beasley R, Holliday M, Reddel HK, et al; Novel START Study Team. Controlled trial of budesonide-formoterol as needed for mild asthma. N Engl J Med. 2019;380:2020-2030.
7. Albuterol. GoodRx. Accessed February 17, 2021. www.goodrx.com/albuterol
8. Budesonide/formoterol. GoodRx. Accessed February 17, 2021. www.goodrx.com/budesonide-formoterol
1. Hardy J, Baggott C, Fingleton J, et al; PRACTICAL study team. Budesonide-formoterol reliever therapy versus maintenance budesonide plus terbutaline reliever therapy in adults with mild to moderate asthma (PRACTICAL): a 52-week, open-label, multicentre, superiority, randomised controlled trial. Lancet. 2019;394:919-928. Published correction appears in Lancet. 2020;395:1422.
2. Centers for Disease Control and Prevention. Summary Health Statistics: National Health Interview Survey, 2018. Accessed February 17, 2021. https://ftp.cdc.gov/pub/Health_Statistics/NCHS/NHIS/SHS/2018_SHS_Table_A-2.pdf
3. National Institutes of Health. National Heart, Lung, and Blood Institute. 2020 Focused Updates to the Asthma Management Guidelines: A Report from the National Asthma Education and Prevention Program Coordinating Committee Expert Panel Working Group. December 2020. Accessed February 17, 2021. www.nhlbi.nih.gov/health-topics/all-publications-and-resources/2020-focused-updates-asthma-management-guidelines
4. Global Initiative for Asthma (GINA). Global Strategy for Asthma Management and Prevention, 2020. Accessed February 17, 2021. www.ginasthma.org/
5. Sobieraj DM, Weeda ER, Nguyen E, et al. Association of inhaled corticosteroids and long-acting β-agonists as controller and quick relief therapy with exacerbations and symptom control in persistent asthma: a systematic review and meta-analysis. JAMA. 2018;319:1485-1496.
6. Beasley R, Holliday M, Reddel HK, et al; Novel START Study Team. Controlled trial of budesonide-formoterol as needed for mild asthma. N Engl J Med. 2019;380:2020-2030.
7. Albuterol. GoodRx. Accessed February 17, 2021. www.goodrx.com/albuterol
8. Budesonide/formoterol. GoodRx. Accessed February 17, 2021. www.goodrx.com/budesonide-formoterol
PRACTICE CHANGER
Use an inhaled corticosteroid plus long-acting beta-agonist (ICS/LABA) prn for intermittent, mild persistent, or moderate persistent asthma for fewer moderate and severe exacerbations and the same daily symptom control as scheduled ICS with a short-acting beta-agonist (SABA) prn.1
STRENGTH OF RECOMMENDATION
A: Based on a single, good-quality, multicenter, randomized controlled trial.1
Hardy J, Baggott C, Fingleton J, et al; PRACTICAL study team. Budesonide-formoterol reliever therapy versus maintenance budesonide plus terbutaline reliever therapy in adults with mild to moderate asthma (PRACTICAL): a 52-week, open-label, multicentre, superiority, randomised controlled trial. Lancet. 2019;394:919-928. Published correction appears in Lancet. 2020;395:1422.1
What to do if an employee tests positive for COVID-19
An increasingly common question I’m receiving is:
As always, it depends, but here is some general advice: The specifics will vary depending on state/local laws, or your particular situation.
First, you need to determine the level of exposure, and whether it requires action. According to the Centers for Disease Control and Prevention, actionable exposure occurs 2 days prior to the onset of illness, and lasts 10 days after onset.
If action is required, you’ll need to determine who needs to quarantine and who needs to be tested. Vaccinated employees who have been exposed to suspected or confirmed COVID-19 are not required to quarantine or be tested if they are fully vaccinated and have remained asymptomatic since the exposure. Those employees should, however, follow all the usual precautions (masks, social distancing, handwashing, etc.) with increased diligence. Remind them that no vaccine is 100% effective, and suggest they self-monitor for symptoms (fever, cough, shortness of breath, etc.)
All other exposed employees should be tested. A negative test means an individual was not infected at the time the sample was collected, but that does not mean an individual will not get sick later. Some providers are retesting on days 5 and 7 post exposure.
Some experts advise that you monitor exposed employees (vaccinated or not) yourself, with daily temperature readings and inquiries regarding symptoms, and perhaps a daily pulse oximetry check, for 14 days following exposure. Document these screenings in writing. Anyone testing positive or developing a fever or other symptoms should, of course, be sent home and seek medical treatment as necessary.
Employees who develop symptoms or test positive for COVID-19 should remain out of work until all CDC “return-to-work” criteria are met. At this writing, the basic criteria include:
- At least 10 days pass after symptoms first appeared
- At least 24 hours pass after last fever without the use of fever-reducing medications
- Cough, shortness of breath, and any other symptoms improve
Anyone who is significantly immunocompromised may need more time at home, and probably consultation with an infectious disease specialist.
Your facility should be thoroughly cleaned after the exposure. Close off all areas used by the sick individual, and clean and disinfect all areas such as offices, doorknobs, bathrooms, common areas, and shared electronic equipment. Of course, the cleaners should wear gowns, gloves, masks, and goggles. Some practices are hiring cleaning crews to professionally disinfect their offices. Once the area has been disinfected, it can be reopened for use. Workers without close contact with the person who is sick can return to work immediately after disinfection.
If the potential infected area is widespread and cannot be isolated to a room or rooms where doors can be shut, it may be prudent to temporarily close your office, send staff home, and divert patients to other locations if they cannot be rescheduled. Once your facility is cleaned and disinfected and staff have been cleared, your office may reopen.
Use enhanced precautions for any staff or patients who are immunocompromised, or otherwise fall into the high-risk category, to keep them out of the path of potential exposure areas and allow them to self-quarantine if they desire.
You should continue following existing leave policies (paid time off, vacation, sick, short-term disability, leave of absence, Family and Medical Leave Act, and Americans with Disabilities Act). If the employee was exposed at work, contact your workers’ compensation carrier regarding lost wages. Unless your state laws specify otherwise, you are under no obligation to pay beyond your policies, but you may do so if you choose.
Of course, you can take proactive steps to prevent unnecessary exposure and avoid closures in the first place; for example:
- Call patients prior to their visit, or question them upon arrival, regarding fever, shortness of breath, and other COVID-19 symptoms.
- Check employees’ temperatures every morning.
- Check patients’ temperatures as they enter the office.
- Require everyone, patients and employees alike, to wear face coverings.
- Ask patients to leave friends and family members at home.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a long-time monthly columnist for Dermatology News. Write to him at dermnews@mdedge.com.
An increasingly common question I’m receiving is:
As always, it depends, but here is some general advice: The specifics will vary depending on state/local laws, or your particular situation.
First, you need to determine the level of exposure, and whether it requires action. According to the Centers for Disease Control and Prevention, actionable exposure occurs 2 days prior to the onset of illness, and lasts 10 days after onset.
If action is required, you’ll need to determine who needs to quarantine and who needs to be tested. Vaccinated employees who have been exposed to suspected or confirmed COVID-19 are not required to quarantine or be tested if they are fully vaccinated and have remained asymptomatic since the exposure. Those employees should, however, follow all the usual precautions (masks, social distancing, handwashing, etc.) with increased diligence. Remind them that no vaccine is 100% effective, and suggest they self-monitor for symptoms (fever, cough, shortness of breath, etc.)
All other exposed employees should be tested. A negative test means an individual was not infected at the time the sample was collected, but that does not mean an individual will not get sick later. Some providers are retesting on days 5 and 7 post exposure.
Some experts advise that you monitor exposed employees (vaccinated or not) yourself, with daily temperature readings and inquiries regarding symptoms, and perhaps a daily pulse oximetry check, for 14 days following exposure. Document these screenings in writing. Anyone testing positive or developing a fever or other symptoms should, of course, be sent home and seek medical treatment as necessary.
Employees who develop symptoms or test positive for COVID-19 should remain out of work until all CDC “return-to-work” criteria are met. At this writing, the basic criteria include:
- At least 10 days pass after symptoms first appeared
- At least 24 hours pass after last fever without the use of fever-reducing medications
- Cough, shortness of breath, and any other symptoms improve
Anyone who is significantly immunocompromised may need more time at home, and probably consultation with an infectious disease specialist.
Your facility should be thoroughly cleaned after the exposure. Close off all areas used by the sick individual, and clean and disinfect all areas such as offices, doorknobs, bathrooms, common areas, and shared electronic equipment. Of course, the cleaners should wear gowns, gloves, masks, and goggles. Some practices are hiring cleaning crews to professionally disinfect their offices. Once the area has been disinfected, it can be reopened for use. Workers without close contact with the person who is sick can return to work immediately after disinfection.
If the potential infected area is widespread and cannot be isolated to a room or rooms where doors can be shut, it may be prudent to temporarily close your office, send staff home, and divert patients to other locations if they cannot be rescheduled. Once your facility is cleaned and disinfected and staff have been cleared, your office may reopen.
Use enhanced precautions for any staff or patients who are immunocompromised, or otherwise fall into the high-risk category, to keep them out of the path of potential exposure areas and allow them to self-quarantine if they desire.
You should continue following existing leave policies (paid time off, vacation, sick, short-term disability, leave of absence, Family and Medical Leave Act, and Americans with Disabilities Act). If the employee was exposed at work, contact your workers’ compensation carrier regarding lost wages. Unless your state laws specify otherwise, you are under no obligation to pay beyond your policies, but you may do so if you choose.
Of course, you can take proactive steps to prevent unnecessary exposure and avoid closures in the first place; for example:
- Call patients prior to their visit, or question them upon arrival, regarding fever, shortness of breath, and other COVID-19 symptoms.
- Check employees’ temperatures every morning.
- Check patients’ temperatures as they enter the office.
- Require everyone, patients and employees alike, to wear face coverings.
- Ask patients to leave friends and family members at home.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a long-time monthly columnist for Dermatology News. Write to him at dermnews@mdedge.com.
An increasingly common question I’m receiving is:
As always, it depends, but here is some general advice: The specifics will vary depending on state/local laws, or your particular situation.
First, you need to determine the level of exposure, and whether it requires action. According to the Centers for Disease Control and Prevention, actionable exposure occurs 2 days prior to the onset of illness, and lasts 10 days after onset.
If action is required, you’ll need to determine who needs to quarantine and who needs to be tested. Vaccinated employees who have been exposed to suspected or confirmed COVID-19 are not required to quarantine or be tested if they are fully vaccinated and have remained asymptomatic since the exposure. Those employees should, however, follow all the usual precautions (masks, social distancing, handwashing, etc.) with increased diligence. Remind them that no vaccine is 100% effective, and suggest they self-monitor for symptoms (fever, cough, shortness of breath, etc.)
All other exposed employees should be tested. A negative test means an individual was not infected at the time the sample was collected, but that does not mean an individual will not get sick later. Some providers are retesting on days 5 and 7 post exposure.
Some experts advise that you monitor exposed employees (vaccinated or not) yourself, with daily temperature readings and inquiries regarding symptoms, and perhaps a daily pulse oximetry check, for 14 days following exposure. Document these screenings in writing. Anyone testing positive or developing a fever or other symptoms should, of course, be sent home and seek medical treatment as necessary.
Employees who develop symptoms or test positive for COVID-19 should remain out of work until all CDC “return-to-work” criteria are met. At this writing, the basic criteria include:
- At least 10 days pass after symptoms first appeared
- At least 24 hours pass after last fever without the use of fever-reducing medications
- Cough, shortness of breath, and any other symptoms improve
Anyone who is significantly immunocompromised may need more time at home, and probably consultation with an infectious disease specialist.
Your facility should be thoroughly cleaned after the exposure. Close off all areas used by the sick individual, and clean and disinfect all areas such as offices, doorknobs, bathrooms, common areas, and shared electronic equipment. Of course, the cleaners should wear gowns, gloves, masks, and goggles. Some practices are hiring cleaning crews to professionally disinfect their offices. Once the area has been disinfected, it can be reopened for use. Workers without close contact with the person who is sick can return to work immediately after disinfection.
If the potential infected area is widespread and cannot be isolated to a room or rooms where doors can be shut, it may be prudent to temporarily close your office, send staff home, and divert patients to other locations if they cannot be rescheduled. Once your facility is cleaned and disinfected and staff have been cleared, your office may reopen.
Use enhanced precautions for any staff or patients who are immunocompromised, or otherwise fall into the high-risk category, to keep them out of the path of potential exposure areas and allow them to self-quarantine if they desire.
You should continue following existing leave policies (paid time off, vacation, sick, short-term disability, leave of absence, Family and Medical Leave Act, and Americans with Disabilities Act). If the employee was exposed at work, contact your workers’ compensation carrier regarding lost wages. Unless your state laws specify otherwise, you are under no obligation to pay beyond your policies, but you may do so if you choose.
Of course, you can take proactive steps to prevent unnecessary exposure and avoid closures in the first place; for example:
- Call patients prior to their visit, or question them upon arrival, regarding fever, shortness of breath, and other COVID-19 symptoms.
- Check employees’ temperatures every morning.
- Check patients’ temperatures as they enter the office.
- Require everyone, patients and employees alike, to wear face coverings.
- Ask patients to leave friends and family members at home.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a long-time monthly columnist for Dermatology News. Write to him at dermnews@mdedge.com.
Rollout of COVID-19 monoclonal antibodies lacked unified plan: expert panel
Monoclonal antibodies (mAbs) to treat COVID-19 are in ample supply, but scant evidence on their effectiveness, paltry reimbursement, and a lack of a planned infrastructure to administer them has led to major underutilization of these potentially useful therapies, according to a new report from The National Academies of Sciences, Engineering, and Medicine.
The 35-page report described missed opportunities to work with states and hospitals to establish trust with clinicians and patients and to set up an infusion infrastructure to funnel patients to sites. Though the therapies still need more study, they should be an option for the right patient at the right time, said the National Academies experts in their report, Rapid Expert Consultation on Allocating COVID-19 Monoclonal Antibody Therapies and Other Novel Therapeutics.
“No potentially eligible patient should be left uninformed, and no eligible patient should be denied access, if there are doses available and the patient and doctor agree it is a reasonable course,” they concluded. The report also noted that underuse, and in particular underuse by members of vulnerable and underserved communities “raises concerns about exacerbating already dramatic health disparities.”
The federal government has spent $375 million on Eli Lilly’s bamlanivimab and $450 million on Regeneron’s casirivimab plus imdevimab cocktail, and agreed last month to spend as much as $2.6 billion more on up to 1.25 million additional doses.
Some 785,000 doses of the two therapeutics have been produced and about a half million have been distributed to states. But about three quarters have gone unused. The U.S. Department of Health & Human Services has launched an online treatment locater to try to spur interest in the therapies.
But the federal government hasn’t addressed some of the basic barriers to use of the monoclonals, said the National Academies experts.
“Lack of awareness, interest, and confidence in COVID-19 mAb therapies among patients and providers are major issues,” they said in the report. Patients who have tested positive might not want to travel to an infusion site, while others might not have access to health care or only seek such treatments when it’s too late. Some who are eligible might not have the time, resources, or transportation to go to a site and sit through a 2-hour treatment.
In addition, “the supply and availability of infusion centers and personnel was identified as a greater constraint than the supply of COVID-19 mAbs,” said the report.
Cost a big impediment
While the federal government has covered the cost of the therapies, hospitals and patients inevitably incur related costs.
“The fragmented payment system in the United States has not provided adequate support to cover the spectrum of costs associated with COVID-19 mAb therapies,” said the report. That is compounded by chronic underfunding and restrictions on federally qualified health centers for community health, the report said.
Patients may have to pay for testing, office visits, follow-up appointments, transportation to and from the infusion site, and potentially a copay for the administration of the drug.
While Medicare pays hospitals $309 per infusion, that might not be enough, especially if a hospital or other site had to build out a new infusion center, the report shows. For clinicians, the administrative payment under Medicare Part B does “not cover the total practice cost to furnish infusion services, resulting in a substantial cost-reimbursement disparity,” the report states.
In addition, there are no specific codes for observing patients during the 2-hour procedure.
“The established Medicare payment rate for furnishing COVID-19 mAb therapies does not cover the cost associated with coordinating care for those patients, nor does it justify the risk and opportunity costs associated with investing in infrastructure modifications to safely integrate COVID-19 patients into existing facilities or building temporary infusion capacity,” the report concluded.
More data needed
The U.S. Food and Drug Administration issued emergency-use authorizations (EUAs) for the two monoclonal therapies based on phase 2 trial data, and that leaves a lot of uncertainty, noted the National Academies.
In trials, both therapies reduced COVID-19-related hospitalizations and emergency room visits within 28 days after treatment among patients at high risk of progression, compared with those who received placebo.
But clinicians aren’t certain about who should use the monoclonals, said the report. The underuse has in turn led to trouble collecting data – either through ongoing trials or in starting new trials.
The National Academies recommended allocating the monoclonal antibodies in a way that would give rise to better data collection to inform clinicians. Payers could support the development of a core data platform or registry, or Medicare could develop pilot trials, said the report.
Lilly and UnitedHealth Group are collaborating on a study in high-risk Medicare patients, according to Reuters. Patients who test positive will be given bamlanivimab at home.
“Building infusion capacity and developing the evidence base about the impact of COVID-19 mAbs on clinical outcomes other than hospitalization, including mortality, are the most promising strategies for increasing effective utilization moving forward,” stated the National Academies report.
A version of this article first appeared on Medscape.com.
Monoclonal antibodies (mAbs) to treat COVID-19 are in ample supply, but scant evidence on their effectiveness, paltry reimbursement, and a lack of a planned infrastructure to administer them has led to major underutilization of these potentially useful therapies, according to a new report from The National Academies of Sciences, Engineering, and Medicine.
The 35-page report described missed opportunities to work with states and hospitals to establish trust with clinicians and patients and to set up an infusion infrastructure to funnel patients to sites. Though the therapies still need more study, they should be an option for the right patient at the right time, said the National Academies experts in their report, Rapid Expert Consultation on Allocating COVID-19 Monoclonal Antibody Therapies and Other Novel Therapeutics.
“No potentially eligible patient should be left uninformed, and no eligible patient should be denied access, if there are doses available and the patient and doctor agree it is a reasonable course,” they concluded. The report also noted that underuse, and in particular underuse by members of vulnerable and underserved communities “raises concerns about exacerbating already dramatic health disparities.”
The federal government has spent $375 million on Eli Lilly’s bamlanivimab and $450 million on Regeneron’s casirivimab plus imdevimab cocktail, and agreed last month to spend as much as $2.6 billion more on up to 1.25 million additional doses.
Some 785,000 doses of the two therapeutics have been produced and about a half million have been distributed to states. But about three quarters have gone unused. The U.S. Department of Health & Human Services has launched an online treatment locater to try to spur interest in the therapies.
But the federal government hasn’t addressed some of the basic barriers to use of the monoclonals, said the National Academies experts.
“Lack of awareness, interest, and confidence in COVID-19 mAb therapies among patients and providers are major issues,” they said in the report. Patients who have tested positive might not want to travel to an infusion site, while others might not have access to health care or only seek such treatments when it’s too late. Some who are eligible might not have the time, resources, or transportation to go to a site and sit through a 2-hour treatment.
In addition, “the supply and availability of infusion centers and personnel was identified as a greater constraint than the supply of COVID-19 mAbs,” said the report.
Cost a big impediment
While the federal government has covered the cost of the therapies, hospitals and patients inevitably incur related costs.
“The fragmented payment system in the United States has not provided adequate support to cover the spectrum of costs associated with COVID-19 mAb therapies,” said the report. That is compounded by chronic underfunding and restrictions on federally qualified health centers for community health, the report said.
Patients may have to pay for testing, office visits, follow-up appointments, transportation to and from the infusion site, and potentially a copay for the administration of the drug.
While Medicare pays hospitals $309 per infusion, that might not be enough, especially if a hospital or other site had to build out a new infusion center, the report shows. For clinicians, the administrative payment under Medicare Part B does “not cover the total practice cost to furnish infusion services, resulting in a substantial cost-reimbursement disparity,” the report states.
In addition, there are no specific codes for observing patients during the 2-hour procedure.
“The established Medicare payment rate for furnishing COVID-19 mAb therapies does not cover the cost associated with coordinating care for those patients, nor does it justify the risk and opportunity costs associated with investing in infrastructure modifications to safely integrate COVID-19 patients into existing facilities or building temporary infusion capacity,” the report concluded.
More data needed
The U.S. Food and Drug Administration issued emergency-use authorizations (EUAs) for the two monoclonal therapies based on phase 2 trial data, and that leaves a lot of uncertainty, noted the National Academies.
In trials, both therapies reduced COVID-19-related hospitalizations and emergency room visits within 28 days after treatment among patients at high risk of progression, compared with those who received placebo.
But clinicians aren’t certain about who should use the monoclonals, said the report. The underuse has in turn led to trouble collecting data – either through ongoing trials or in starting new trials.
The National Academies recommended allocating the monoclonal antibodies in a way that would give rise to better data collection to inform clinicians. Payers could support the development of a core data platform or registry, or Medicare could develop pilot trials, said the report.
Lilly and UnitedHealth Group are collaborating on a study in high-risk Medicare patients, according to Reuters. Patients who test positive will be given bamlanivimab at home.
“Building infusion capacity and developing the evidence base about the impact of COVID-19 mAbs on clinical outcomes other than hospitalization, including mortality, are the most promising strategies for increasing effective utilization moving forward,” stated the National Academies report.
A version of this article first appeared on Medscape.com.
Monoclonal antibodies (mAbs) to treat COVID-19 are in ample supply, but scant evidence on their effectiveness, paltry reimbursement, and a lack of a planned infrastructure to administer them has led to major underutilization of these potentially useful therapies, according to a new report from The National Academies of Sciences, Engineering, and Medicine.
The 35-page report described missed opportunities to work with states and hospitals to establish trust with clinicians and patients and to set up an infusion infrastructure to funnel patients to sites. Though the therapies still need more study, they should be an option for the right patient at the right time, said the National Academies experts in their report, Rapid Expert Consultation on Allocating COVID-19 Monoclonal Antibody Therapies and Other Novel Therapeutics.
“No potentially eligible patient should be left uninformed, and no eligible patient should be denied access, if there are doses available and the patient and doctor agree it is a reasonable course,” they concluded. The report also noted that underuse, and in particular underuse by members of vulnerable and underserved communities “raises concerns about exacerbating already dramatic health disparities.”
The federal government has spent $375 million on Eli Lilly’s bamlanivimab and $450 million on Regeneron’s casirivimab plus imdevimab cocktail, and agreed last month to spend as much as $2.6 billion more on up to 1.25 million additional doses.
Some 785,000 doses of the two therapeutics have been produced and about a half million have been distributed to states. But about three quarters have gone unused. The U.S. Department of Health & Human Services has launched an online treatment locater to try to spur interest in the therapies.
But the federal government hasn’t addressed some of the basic barriers to use of the monoclonals, said the National Academies experts.
“Lack of awareness, interest, and confidence in COVID-19 mAb therapies among patients and providers are major issues,” they said in the report. Patients who have tested positive might not want to travel to an infusion site, while others might not have access to health care or only seek such treatments when it’s too late. Some who are eligible might not have the time, resources, or transportation to go to a site and sit through a 2-hour treatment.
In addition, “the supply and availability of infusion centers and personnel was identified as a greater constraint than the supply of COVID-19 mAbs,” said the report.
Cost a big impediment
While the federal government has covered the cost of the therapies, hospitals and patients inevitably incur related costs.
“The fragmented payment system in the United States has not provided adequate support to cover the spectrum of costs associated with COVID-19 mAb therapies,” said the report. That is compounded by chronic underfunding and restrictions on federally qualified health centers for community health, the report said.
Patients may have to pay for testing, office visits, follow-up appointments, transportation to and from the infusion site, and potentially a copay for the administration of the drug.
While Medicare pays hospitals $309 per infusion, that might not be enough, especially if a hospital or other site had to build out a new infusion center, the report shows. For clinicians, the administrative payment under Medicare Part B does “not cover the total practice cost to furnish infusion services, resulting in a substantial cost-reimbursement disparity,” the report states.
In addition, there are no specific codes for observing patients during the 2-hour procedure.
“The established Medicare payment rate for furnishing COVID-19 mAb therapies does not cover the cost associated with coordinating care for those patients, nor does it justify the risk and opportunity costs associated with investing in infrastructure modifications to safely integrate COVID-19 patients into existing facilities or building temporary infusion capacity,” the report concluded.
More data needed
The U.S. Food and Drug Administration issued emergency-use authorizations (EUAs) for the two monoclonal therapies based on phase 2 trial data, and that leaves a lot of uncertainty, noted the National Academies.
In trials, both therapies reduced COVID-19-related hospitalizations and emergency room visits within 28 days after treatment among patients at high risk of progression, compared with those who received placebo.
But clinicians aren’t certain about who should use the monoclonals, said the report. The underuse has in turn led to trouble collecting data – either through ongoing trials or in starting new trials.
The National Academies recommended allocating the monoclonal antibodies in a way that would give rise to better data collection to inform clinicians. Payers could support the development of a core data platform or registry, or Medicare could develop pilot trials, said the report.
Lilly and UnitedHealth Group are collaborating on a study in high-risk Medicare patients, according to Reuters. Patients who test positive will be given bamlanivimab at home.
“Building infusion capacity and developing the evidence base about the impact of COVID-19 mAbs on clinical outcomes other than hospitalization, including mortality, are the most promising strategies for increasing effective utilization moving forward,” stated the National Academies report.
A version of this article first appeared on Medscape.com.
J&J vaccine 85% efficacious against severe COVID globally
The Janssen/Johnson & Johnson single-dose adenovirus vaccine provides 85% efficacy globally against severe COVID-19 illness, according to the highly anticipated interim phase 3 results announced this morning.
The efficacy against severe disease provided by the Janssen/J&J vaccine held true regardless of age, race/ethnicity, absence or presence of comorbidities, and geography. The 44,000-participant ENSEMBLE study was conducted in the United States, South America, and South Africa.
“The team is very diligently monitoring all the variants that come up, and there are literally thousands of these. We are acting in anticipation of a variant being a potential problem. The South African variant we too acted on right away. So we too are preparing that antigen for testing.
“With data today, we do see that not a single South African, after 28 days post vaccination, ended up needing to go to the hospital, no South African died who was vaccinated.
“We do see that 85%-plus protection in South African against severe disease. That is one of the most exciting results in the dataset today,” said Mathai Mammen, MD, PhD, global head of Janssen Research & Development.
The overall efficacy was 66% globally, 72% in the United States, 66% in Latin America, and 57% in South Africa against moderate to severe SARS-CoV-2 28 days post vaccination, officials from the National Institutes of Health and Janssen reported during a media briefing.
But the J&J vaccine has potential advantages over the existing two-dose Pfizer/BioNTech and Moderna mRNA vaccines because it’s single dose and has less stringent storage requirements – only regular refrigeration is needed versus a need to freeze the two-dose Pfizer/BioNTech and Moderna COVID-19 vaccines. The J&J vaccine can be refrigerated for up to 3 months at 36°-46° F (2°-8° C).
But the difference between these just-released efficacy figures and the 94%-95% efficacy provided by the existing Pfizer/BioNTech and Moderna mRNA vaccines generated many questions during the briefing.
Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said the focus should not just be on the overall numbers. “The most important thing from a public health standpoint domestically is to keep people out of the hospital and prevent them from getting severe illness,” he said. “Many in the general public might look at a number and want to know if they get symptomatic disease or not.”
“More important than preventing someone from getting some aches and a sore throat is to prevent people – particularly people who have underlying conditions and the elderly, the ones most susceptible to a severe outcome – [from getting] severe disease,” Dr. Fauci added. Prevention of severe outcomes in a high percentage of individuals “will alleviate so much of the stress, human suffering, and death.”
Dr. Fauci acknowledged that many people will naturally focus on the distinction between 72% efficacy and 94%-95% efficacy. “This could be a messaging challenge [but] you have to make sure people understand the implications.”
It is more complex, he added, than just asking people: “If you go to the door on the left, you get 94% or 95%. If you go to the door to the right, you get 72%. What door do you want to go to?”
Instead, the messaging should be that “this and the other vaccines we have are actually preventing severe disease to a very substantial degree.”
Company defends numbers
Janssen defended their efficacy findings, pointing out that it is not a fair comparison.
“The vaccine programs that went a couple of months ago, they ran their studies during different times, when the pandemic was less complex. There were not these variants, and there was not the same level of incidence, which puts pressure on vaccine efficacy,” said Mathai Mammen, MD, PhD, global head of research and development for Janssen.
“So the numbers cannot really be compared, and that does pose a messaging challenge,” he said. “But the reality is, if one was to run the same studies [for the Pfizer and Moderna vaccines] today you would likely see different results.”
Asked if the efficacy figures could affect vaccine hesitancy, National Institutes of Health Director Francis Collins, MD, PhD, said at the announcement that most reluctance among people to get vaccinated against SARS-CoV-2 stems from concerns about safety. “The safety record is extremely good for this vaccine, as it is for the others that have received emergency use authorization.”
Janssen/J&J plans to submit for emergency use authorization from the U.S. Food and Drug Administration next week, at which point the company plans to release more information on side effects, deaths, and patient subpopulation efficacy, and more from the ENSEMBLE trial.
Janssen is aiming to provide 1 billion doses by the end of this year.
A version of this article first appeared on Medscape.com.
The Janssen/Johnson & Johnson single-dose adenovirus vaccine provides 85% efficacy globally against severe COVID-19 illness, according to the highly anticipated interim phase 3 results announced this morning.
The efficacy against severe disease provided by the Janssen/J&J vaccine held true regardless of age, race/ethnicity, absence or presence of comorbidities, and geography. The 44,000-participant ENSEMBLE study was conducted in the United States, South America, and South Africa.
“The team is very diligently monitoring all the variants that come up, and there are literally thousands of these. We are acting in anticipation of a variant being a potential problem. The South African variant we too acted on right away. So we too are preparing that antigen for testing.
“With data today, we do see that not a single South African, after 28 days post vaccination, ended up needing to go to the hospital, no South African died who was vaccinated.
“We do see that 85%-plus protection in South African against severe disease. That is one of the most exciting results in the dataset today,” said Mathai Mammen, MD, PhD, global head of Janssen Research & Development.
The overall efficacy was 66% globally, 72% in the United States, 66% in Latin America, and 57% in South Africa against moderate to severe SARS-CoV-2 28 days post vaccination, officials from the National Institutes of Health and Janssen reported during a media briefing.
But the J&J vaccine has potential advantages over the existing two-dose Pfizer/BioNTech and Moderna mRNA vaccines because it’s single dose and has less stringent storage requirements – only regular refrigeration is needed versus a need to freeze the two-dose Pfizer/BioNTech and Moderna COVID-19 vaccines. The J&J vaccine can be refrigerated for up to 3 months at 36°-46° F (2°-8° C).
But the difference between these just-released efficacy figures and the 94%-95% efficacy provided by the existing Pfizer/BioNTech and Moderna mRNA vaccines generated many questions during the briefing.
Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said the focus should not just be on the overall numbers. “The most important thing from a public health standpoint domestically is to keep people out of the hospital and prevent them from getting severe illness,” he said. “Many in the general public might look at a number and want to know if they get symptomatic disease or not.”
“More important than preventing someone from getting some aches and a sore throat is to prevent people – particularly people who have underlying conditions and the elderly, the ones most susceptible to a severe outcome – [from getting] severe disease,” Dr. Fauci added. Prevention of severe outcomes in a high percentage of individuals “will alleviate so much of the stress, human suffering, and death.”
Dr. Fauci acknowledged that many people will naturally focus on the distinction between 72% efficacy and 94%-95% efficacy. “This could be a messaging challenge [but] you have to make sure people understand the implications.”
It is more complex, he added, than just asking people: “If you go to the door on the left, you get 94% or 95%. If you go to the door to the right, you get 72%. What door do you want to go to?”
Instead, the messaging should be that “this and the other vaccines we have are actually preventing severe disease to a very substantial degree.”
Company defends numbers
Janssen defended their efficacy findings, pointing out that it is not a fair comparison.
“The vaccine programs that went a couple of months ago, they ran their studies during different times, when the pandemic was less complex. There were not these variants, and there was not the same level of incidence, which puts pressure on vaccine efficacy,” said Mathai Mammen, MD, PhD, global head of research and development for Janssen.
“So the numbers cannot really be compared, and that does pose a messaging challenge,” he said. “But the reality is, if one was to run the same studies [for the Pfizer and Moderna vaccines] today you would likely see different results.”
Asked if the efficacy figures could affect vaccine hesitancy, National Institutes of Health Director Francis Collins, MD, PhD, said at the announcement that most reluctance among people to get vaccinated against SARS-CoV-2 stems from concerns about safety. “The safety record is extremely good for this vaccine, as it is for the others that have received emergency use authorization.”
Janssen/J&J plans to submit for emergency use authorization from the U.S. Food and Drug Administration next week, at which point the company plans to release more information on side effects, deaths, and patient subpopulation efficacy, and more from the ENSEMBLE trial.
Janssen is aiming to provide 1 billion doses by the end of this year.
A version of this article first appeared on Medscape.com.
The Janssen/Johnson & Johnson single-dose adenovirus vaccine provides 85% efficacy globally against severe COVID-19 illness, according to the highly anticipated interim phase 3 results announced this morning.
The efficacy against severe disease provided by the Janssen/J&J vaccine held true regardless of age, race/ethnicity, absence or presence of comorbidities, and geography. The 44,000-participant ENSEMBLE study was conducted in the United States, South America, and South Africa.
“The team is very diligently monitoring all the variants that come up, and there are literally thousands of these. We are acting in anticipation of a variant being a potential problem. The South African variant we too acted on right away. So we too are preparing that antigen for testing.
“With data today, we do see that not a single South African, after 28 days post vaccination, ended up needing to go to the hospital, no South African died who was vaccinated.
“We do see that 85%-plus protection in South African against severe disease. That is one of the most exciting results in the dataset today,” said Mathai Mammen, MD, PhD, global head of Janssen Research & Development.
The overall efficacy was 66% globally, 72% in the United States, 66% in Latin America, and 57% in South Africa against moderate to severe SARS-CoV-2 28 days post vaccination, officials from the National Institutes of Health and Janssen reported during a media briefing.
But the J&J vaccine has potential advantages over the existing two-dose Pfizer/BioNTech and Moderna mRNA vaccines because it’s single dose and has less stringent storage requirements – only regular refrigeration is needed versus a need to freeze the two-dose Pfizer/BioNTech and Moderna COVID-19 vaccines. The J&J vaccine can be refrigerated for up to 3 months at 36°-46° F (2°-8° C).
But the difference between these just-released efficacy figures and the 94%-95% efficacy provided by the existing Pfizer/BioNTech and Moderna mRNA vaccines generated many questions during the briefing.
Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said the focus should not just be on the overall numbers. “The most important thing from a public health standpoint domestically is to keep people out of the hospital and prevent them from getting severe illness,” he said. “Many in the general public might look at a number and want to know if they get symptomatic disease or not.”
“More important than preventing someone from getting some aches and a sore throat is to prevent people – particularly people who have underlying conditions and the elderly, the ones most susceptible to a severe outcome – [from getting] severe disease,” Dr. Fauci added. Prevention of severe outcomes in a high percentage of individuals “will alleviate so much of the stress, human suffering, and death.”
Dr. Fauci acknowledged that many people will naturally focus on the distinction between 72% efficacy and 94%-95% efficacy. “This could be a messaging challenge [but] you have to make sure people understand the implications.”
It is more complex, he added, than just asking people: “If you go to the door on the left, you get 94% or 95%. If you go to the door to the right, you get 72%. What door do you want to go to?”
Instead, the messaging should be that “this and the other vaccines we have are actually preventing severe disease to a very substantial degree.”
Company defends numbers
Janssen defended their efficacy findings, pointing out that it is not a fair comparison.
“The vaccine programs that went a couple of months ago, they ran their studies during different times, when the pandemic was less complex. There were not these variants, and there was not the same level of incidence, which puts pressure on vaccine efficacy,” said Mathai Mammen, MD, PhD, global head of research and development for Janssen.
“So the numbers cannot really be compared, and that does pose a messaging challenge,” he said. “But the reality is, if one was to run the same studies [for the Pfizer and Moderna vaccines] today you would likely see different results.”
Asked if the efficacy figures could affect vaccine hesitancy, National Institutes of Health Director Francis Collins, MD, PhD, said at the announcement that most reluctance among people to get vaccinated against SARS-CoV-2 stems from concerns about safety. “The safety record is extremely good for this vaccine, as it is for the others that have received emergency use authorization.”
Janssen/J&J plans to submit for emergency use authorization from the U.S. Food and Drug Administration next week, at which point the company plans to release more information on side effects, deaths, and patient subpopulation efficacy, and more from the ENSEMBLE trial.
Janssen is aiming to provide 1 billion doses by the end of this year.
A version of this article first appeared on Medscape.com.