User login
Therapies for Actinic Keratosis With a Focus on Cosmetic Outcomes
Actinic keratosis (AK), also referred to as solar keratosis or senile keratosis, is an intraepidermal proliferation of dysplastic keratinocytes that develops in response to chronic exposure to UV radiation. Actinic keratoses are among the most commonly encountered lesions seen by dermatologists, and it has been estimated that 60% of predisposed individuals older than 40 years have at least one AK.1,2 Prevalence is notably higher in light-skinned individuals and increases with age, presumably from higher cumulative sun exposure and decreased effectiveness of the immune system.1,3 It remains a point of contention as to whether or not AKs actually represent squamous cell carcinoma (SCC) in situ, but the potential for progression to invasive disease has been well demonstrated, as the majority of SCCs develop from preexisting AKs.4-6 The risk for progression to invasive disease for an individual AK has been estimated to range from 0.025% to 16% per year, with an average of approximately 8% in immunocompetent patients.7
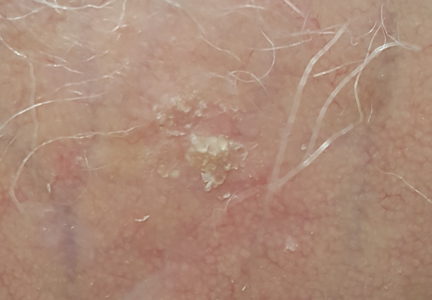
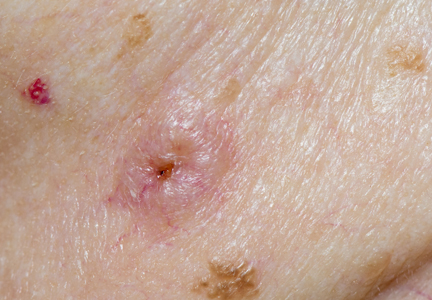
The clinical morphology of AK can vary widely, but the most common presentation is an erythematous scaly macule, papule, or plaque on sun-exposed skin. The skin surrounding AKs typically shows evidence of solar damage with deep wrinkling, mottled pigmentation, scattered telangiectases, purpura, or xerosis (Figure). A variety of clinical variants with unique presentations exist, including atrophic, hypertrophic, acantholytic, lichenoid, bowenoid, and pigmented subtypes. Because more than 80% of AKs occur on highly visible areas such as the head, neck, back of the hands, and forearms, AKs can have an obvious detrimental effect on cosmetic appearance. Studies also have shown a strong association between AKs and decreased overall quality of life (QOL).3,8,9

Because of the risk for AK progression to invasive cancer along with its negative impact on cosmesis and QOL, clinicians generally opt to treat AKs. Numerous different treatment options exist, including topical medications, procedural modalities, and light-based therapies. Here, we review the efficacy of the most commonly utilized treatments and discuss the relevant cosmetic considerations and outcomes.
Topical Treatments
5-Fluorouracil
5-Fluorouracil (5-FU) is a US Food and Drug Administration (FDA)–approved, topically applied pyrimidine analogue that inhibits thymidylate synthase. The resulting suppression of DNA and RNA synthesis induces cell death with a preference for mitotically active cells.10 5-Fluorouracil has been used for more than 50 years as a treatment of AK and its efficacy is well established. A systematic review of 5 randomized controlled studies of topical 5-FU reported an average of 49% of 423 patients achieving complete lesion clearance with 5-FU cream 5% applied once or twice daily for up to 7 weeks.11 Some notable drawbacks of 5-FU, however, are application-site erythema, blistering, pruritus, necrosis, erosion, and pain. These effects often lead to premature cessation of therapy, but newer formulations of 5-FU cream 0.5% have shown good efficacy with better tolerability.12 A randomized, double-blind, multicenter, parallel-group study of 177 patients using 5-FU cream 0.5% once daily for either 1, 2, or 4 weeks demonstrated significant (P<.001) efficacy over vehicle gel in all treatment arms.13 The most effective therapy was 4 weeks of treatment, which achieved a mean 91.7% reduction in lesion count as assessed 1 month after cessation of therapy. The primary adverse effect (AE) reported in this trial was mild to moderate facial irritation, which generally resolved within 18 to 21 days after treatment cessation.13 Overall, 5-FU is a highly effective therapy for treating AKs that also can improve signs of photoaging, but patients should be aware of cosmetically unappealing effects that generally occur throughout therapy and during the immediate posttreatment period.14
Chemical Peels
Chemical peels traditionally employ acidic compounds to strip away outer layers of skin to variable depths depending on the concentration of the agent being applied. For treatment of AK, trichloroacetic acid (TCA) is a commonly employed cauterant that has shown efficacy comparable to topical 5-FU as well as ablative CO2 laser resurfacing.15 Trichloroacetic acid peels also are a convenient therapy, as good results can be achieved after a single treatment session. A split-face study of 15 patients treated with either a single application of 35% TCA and Jessner solution or twice-daily application of 5-FU cream 5% for 3 weeks demonstrated a reduction in 75% of visible AKs in both treatment arms over a 1-year follow-up period.16 Although 80% of patients self-reported considerable cosmetic improvement with both therapies, patient preference was reported to be in favor of the TCA peel, given its quick results and relatively mild side effects as compared to 5-FU. Treatment with chemical peels will result in temporary erythema and mild desquamation that usually resolves within 2 weeks; however, there are cases in which erythema has been reported to persist for several months.16 Adverse effects such as permanent scarring or pigmentation changes rarely are seen with TCA concentrations less than 45%.17 Caution should be used in patients with a history of herpes simplex virus, keloids, postinflammatory hyperpigmentation, radiation exposure, immunosuppression, and those unable or unwilling to use sunscreen and avoid sun exposure in the immediate posttreatment period.
Diclofenac Sodium
Diclofenac sodium (DFS) is an FDA-approved topical, nonsteroidal,
anti-inflammatory drug whose mechanism of action in the treatment of AK is thought to involve inhibition of the cyclooxygenase 2 enzyme.18 The resulting reduction of prostaglandins is believed to inhibit tumor angiogenesis, induce apoptosis, and inhibit cell differentiation.19-22 In a multicenter, double-blind, placebo-controlled study of 195 patients, application of DFS 3% in hyaluronan gel 2.5% twice daily for 60 days showed significant (P<.05) efficacy over placebo in achieving complete resolution of target lesions during a 30-day follow-up period (31% vs 10%). Furthermore, qualitative patient assessment of complete global improvement also was significantly (P<.05) higher in the active treatment group as compared to placebo (31% vs 10%).23 Additional studies of DFS 3% in hyaluronan gel 2.5% applied twice daily for 90 days have shown even higher rates of success, with complete resolution of target lesions in 40% to 58% of cases.24,25 This therapy also has been reported to substantially improve QOL following treatment completion.26 The most frequently cited AEs include pruritus, rash, dry skin, erythema, and application-site reactions. Overall, DFS is a
well-tolerated therapy with efficacy comparable to that of 5-FU but with a lower incidence of AEs
and higher patient satisfaction as determined in
2 head-to-head studies.27,28
ImiquimodImiquimod (IMQ) is an FDA-approved topical agent that functions as an immune response modifier via agonism of toll-like receptor 7.18 The resulting cytokine production and release enhances the innate and acquired immune responses leading to anticancer activity.29 The efficacy of IMQ for treatment of AK has been demonstrated in numerous well-designed clinical trials. A
meta-analysis of 5 randomized, double-blind trials including 1293 patients treated with IMQ cream 5%
2 to 3 times per week for 12 to 16 weeks reported complete clearance of AKs in 50% of patients treated with IMQ as compared to 5% of patients treated with vehicle.30 The most frequently reported AEs with this therapy include erythema, scabbing, flaking, and erosion. These effects generally resolve following cessation of treatment, and therapy is considered to be well tolerated; however, there are case reports of IMQ triggering or exacerbating existing inflammatory conditions.31 Imiquimod cream also is approved at 2.5% and 3.75% concentrations, which have demonstrated significant (P<.001) efficacy over placebo and a reduced incidence of AEs; complete clearance rates have been reported as 30.6% and 35.6%, respectively.32 Notably, a study comparing 75 patients randomized to either IMQ cream 5%
3 times per week for 4 weeks, 1 or 2 courses of cryosurgery, or 5-FU ointment 5% twice daily for 4 weeks reported that IMQ achieved significantly (P<.01) superior sustained clearance rates during a 12-month follow-up period over cryosurgery and 5-FU
(73% vs 4% vs 33%).33 Additionally, cosmetic outcomes as determined by both participants and investigators were reported as excellent at 12 months posttreatment in more than 80% of participants treated with IMQ. These excellent, long-lasting cosmetic outcomes also were determined to be significantly (P<.0001) superior to the cosmetic outcomes of 5-FU and cryotherapy, which both reported excellent outcomes in less than 10% of cases.33
Ingenol MebutateIngenol mebutate (IM) is a macrocyclic diterpene ester derived from the Euphorbia peplus plant that is FDA approved for the treatment of AK.1 Ingenol mebutate’s mechanism of action is thought to involve induction of cell death via disruption of the plasma membrane and mitochondria in addition to production of an inflammatory response, which produces tumor-specific antibodies and a large influx of neutrophils.34,35 The overall evidence for the efficacy of IM is strong. A combined analysis of 4 multicenter, randomized, double-blind studies of 1005 participants reported that IM gel 0.015% applied once daily for 3 days to the face or scalp was significantly superior (P<.001) to placebo in achieving complete clearance as assessed 54 days after completion of therapy (42.2% vs 3.7%) and that IM gel 0.05% applied once daily for 2 days to the trunk or extremities also was significantly superior (P<.001) to placebo in achieving complete clearance as determined 55 days after completion of therapy (34.1% vs 4.7%).36 A follow-up report to this study indicated that IM also appears to achieve long-lasting effects with an overall 87% decrease in total AKs at 12 months follow-up in both trial groups.37 Additionally, it has been recently reported that treatment with IM in these trials was associated with significantly higher overall treatment satisfaction (P<.001) and improved QOL (P<.001) as compared to vehicle.38 Cosmetic outcomes of IM therapy have been assessed in a trial analyzing the efficacy of IM gel 0.025% for 3 days or IM gel 0.05% for 2 or 3 days on nonfacial AKs. This study reported significantly (P<.0001) higher patient satisfaction with the cosmetic outcome at 8 weeks after therapy as compared to vehicle.34 Studies performed in mice have demonstrated that IM is able to promote collagen matrix turnover and impose dermal elasticity, which may contribute to these good cosmetic outcomes.39 The most common AEs of IM therapy are erythema, crusting, and flaking; these effects generally occur 3 to 8 days after starting treatment. These effects, however, generally are short lived and resolve within 2 weeks of treatment cessation when IM is applied to the face or scalp or 4 weeks when applied to the trunk or extremities.40 Overall, IM is a useful therapeutic option given its relatively short treatment course as compared to other topically applied agents, as well as its lasting efficacy, mild AEs, and good cosmetic outcomes.
Procedural Modalities
Surgical Procedures
Surgical approaches for the treatment of AK include excision, curettage with or without electrodesiccation, and dermabrasion. In the past, these modalities were used with greater frequency, but the advent of effective topical medications with lower risks of AEs has largely reduced their use.41 Excision may still be indicated in cases where SCC is suspected, and curettage can be used for treatment of thicker hypertrophic AKs.42 Although these approaches have not been evaluated in clinical trials, they are generally effective but require the use of local anesthetics and come with substantial risk for infection, permanent scarring, and hypopigmentation. Dermabrasion employs the use of a motorized device equipped with an abrasive material to physically remove superficial layers of the skin. Studies are limited, but this method has been reported as an effective treatment in a retrospective review of 23 participants in which 96% remained free of AKs at 1 year, 83% at 2 years, 64% at 4 years, and 54% at 5 years posttherapy.43 Notably, one split-face study of 40 participants treated with dermabrasion followed by 25% TCA on one side and either Jessner solution and 35% TCA or dermabrasion alone on the other side reported that the combination of dermabrasion with 25% TCA consistently produced excellent cosmetic results with nearly complete eradication of AKs.44 In general, however, cosmetic outcomes with dermabrasion are variable, as the technique is highly operator dependent and treatment is associated with notable discomfort as well as risk for scarring and permanent pigmentation alteration.
Cryotherapy
Cryotherapy remains one of the most commonly utilized treatments of AK and involves the delivery of liquid nitrogen via a spray device or a cotton tip applicator to rapidly freeze cells, thus causing cellular destruction via ice crystal formation and protein denaturation.45 Efficacy with this technique has been reported to be as high as 98.8% at 12 months follow-up, but more recent studies cite lower rates of success.46 A prospective multicenter study of 90 participants with 421 AKs on the face or scalp treated with a single freeze-thaw cycle of liquid nitrogen reported an overall complete response rate of 67.2% at 3 months posttherapy. Additionally, higher complete response rates were associated with longer freeze times, and cosmetic outcomes were reported as good to excellent in 94% of complete response lesions.47 Similar results were reported in an open-label, prospective, randomized, controlled clinical trial of 200 participants with 543 AKs, which compared a single freeze-thaw cycle with liquid nitrogen to a single session of CO2 laser ablation in the treatment of isolated AKs of the face and scalp.48 At 3 months posttherapy, complete clearance was observed in 71.6% of participants treated with cryotherapy and in 65.3% of participants treated with laser ablation (P=.532). At 12 months posttherapy, participants who originally showed complete response at 3 months were assessed for relapse. Complete clearance was preserved in 72.6% of participants treated with cryotherapy versus 21.9% of participants treated with laser ablation (P<.0001), and cosmetic outcomes were reported by participants as good or excellent at 3 months follow-up in more than 93% of participants for both treatment arms.48 Possible AEs of cryotherapy include pain during treatment, blister formation with possible hemorrhage, infection, scarring, and permanent pigmentary changes.47,48 Notably, the risk for hypopigmentation increases with longer freezing times, thus requiring clinicians to consider the balance between improved efficacy and reduced cosmetic outcomes.47
Light-Based Therapies
Laser Therapy
Ablative laser resurfacing with either the CO2 or erbium-doped:YAG (Er:YAG) laser utilizes light of specific wavelengths to selectively induce thermolysis and destruction of the epidermal layer. Both lasers have been studied as treatments of AK, but there is a lack of large, well-designed studies. In one small study of 14 participants treated with 1 to 2 passes of the CO2 laser, complete clearance was reported in all cases without any recurrences during a follow-up period of 6 to 24 months. Additionally, all participants in this study reported satisfaction with the cosmetic outcome.49 The CO2 laser also has demonstrated efficacy comparable to that of the TCA peel and 5-FU therapy in a prospective randomized trial of 34 patients with facial or scalp AKs who received either CO2 laser with 2 passes, 30% TCA peel, or 5-FU cream 5% twice daily for 3 weeks.15 Reduction in mean AK counts at 3 months posttherapy was significantly (P<.03) higher in all treatment arms as compared to the control group (92% for CO2 laser, 89% for TCA peel, and 83% for 5-FU cream). No significant (P=.31) difference in outcomes was noted among the different treatment arms.15 Similar results were reported for the Er:YAG laser in a small prospective study of 5 participants treated with 2 to 3 passes with the Er:YAG laser in which reduction in mean AK counts was reported as ranging from 86% to 96% at 3 months posttherapy.50 The Er:YAG laser in combination with the CO2 laser has shown notable long-term efficacy in achieving higher lesion clearance rates and sustained complete clearance rates over treatment with topical 5-FU.51 In a prospective randomized study of 55 par-ticipants with multiple AKs on the face or scalp, participants were assigned to receive either combination laser ablation with the Er:YAG and CO2 lasers down to the level of the papillary dermis or 5-FU cream 5% applied twice daily for 2 to 7 weeks until an appropriate clinical inflammatory response was achieved. At 12 months follow-up, the laser treatment group achieved significantly (P=.048) higher mean lesion clearance rates (91.1%) as compared to the 5-FU arm (76.6%) and significantly (P=.003) higher sustained complete clearance rates (59.3%) as compared to 5-FU (29.2%). The proportion of participants with an improvement in photoaging score at 12 months follow-up approached statistical significance (P=.07), with 74% of the laser-treated group showing improvement as compared to 43% of the 5-FU–treated group. Long-term, cosmetically unappealing side effects such as erythema and hypopigmentation occurred notably more often in the laser-treated group as compared to the 5-FU group.51 In summary, ablative lasers appear to be a highly effective therapy for AK but at the cost of increased risk for AEs such as permanent pigmentary changes, prolonged erythema lasting up to several months, and scarring.50,52-55
Fractional photothermolysis is a relatively new advancement in the field of laser therapy that has received FDA approval for the treatment of AK.56 This treatment works by creating multiple noncontiguous microscopic columns of thermal injury while sparing adjacent zones of viable tissue.57 Although there are limited studies involving the use of such lasers in the treatment of AK, initial findings suggest that 1927-nm thulium lasers may be more effective than 1550-nm erbium lasers in achieving lesion clearance. A trial of 14 participants who received 5 laser treatments with a 1550-nm fractionated erbium-doped fiber laser reported an average reduction in AK counts of 66.2% at 3 months follow-up and a 55.6% reduction at 6 months follow-up. A participant-determined marked or very significant improvement of lesions was reported in 83% of participants at 1 month posttreatment but only in 44% of participants at 6 months posttreatment.58 A similar trial of 24 participants treated with up to 4 treatment sessions of the fractionated 1927-nm thulium laser reported an 87.3% reduction in number of AKs at 3 months follow-up and an 86.6% reduction at 6 months follow-up.56 The primary advantage of fractional laser therapy is a faster recovery period generally lasting only 2 or 3 days as compared to 2 weeks or more with traditional ablative lasers, thus limiting the amount of time a patient must tolerate cosmetically unappealing erythema.59,60 The quick recovery time has been attributed to the fractional laser’s ability to preserve the stratum corneum and skin barrier, which also helps reduce the risk for other AEs such as scarring and infection.56,59-61 Additional studies are needed to better assess the true efficacy of fractional laser therapy, but treatment with the fractional 1927-nm thulium laser appears to be a promising and well-tolerated therapeutic option for treatment of AK with similar efficacy to traditional ablative lasers but with a lower risk of AEs.
Photodynamic TherapyPhotodynamic therapy (PDT) is an FDA-approved treatment that involves the use of a topical photosensitizing agent such as 5-aminolevulinic acid (ALA) or methyl aminovulinate (MAL) before exposure to an activating light source to generate reactive oxygen species that lead to cell death.62-65 Multiple PDT regimens with varying combinations of photosensitizers, incubation time, and light sources have been studied, but a 2012 Cochrane review determined that treatment with conventional formulations of MAL and ALA with either blue- or red-light PDT were similarly efficacious for treatment of individual AKs as compared to vehicle with blue- or red-light PDT. One exception was that longer incubation time (ie, 4 hours) with ALA resulted in better results than shorter incubation times (ie, 0.5, 1, 2 hours) with ALA.66
Standard PDT treatment with MAL also has consistently demonstrated superior efficacy in achieving complete clearance rates in addition to superior cosmetic outcomes over treatment with either cryotherapy, DFS, or 5-FU.67-73 Three studies in particular noted an excellent or good investigator-determined cosmetic outcome in 96% to 98% of participants treated with MAL-PDT.69,71,74 Photodynamic therapy with ALA also has been reported as superior over CO2 laser ablation for AK reduction as well as both patient and investigator overall satisfaction.75
More recently, several methods of improving photosensitizer delivery have been studied, which have demonstrated remarkable efficacy at achieving lesion clearance over standard cream formulations or application routines. One such method involves the use of gentle heating to increase photosensitizer uptake. In a split-extremity study of 20 participants who were treated with 20% ALA under occlusion for 1 hour with one side heated to 38.8°C, the heated side demonstrated significant (P<.0001) efficacy at achieving higher median clearance rates over control when evaluated at 2 and 6 months posttherapy.76 Notably, occlusion of ALA in itself during the incubation period also has been demonstrated to significantly (P<.0001) improve clearance rates.77 Another method involves the use of a new nanoemulsion-based formulation of ALA gel, known as BF-200 ALA, which has demonstrated remarkable efficacy over standard MAL cream and placebo in a long-term follow-up analysis of 2 prospective, randomized, controlled trials.78 In a similar vein, 3 prospective randomized trials with a minimum follow-up time of 3 months demonstrated that MAL-PDT in combination with fractional ablative laser pretreatment has significant (P<.02 in all trials) efficacy over MAL-PDT without pretreatment in achieving complete AK clearance. Although the cosmetic outcomes were good or excellent in 87% to 100% of patients, they were not significantly different from stand-alone MAL-PDT treatment in any of the trials.79-81 However, pretreatment with microneedling in MAL-PDT has been shown to achieve superior cosmetic outcomes over MAL-PDT without microneedling, according to one small split-face study of 10 participants.82
Overall, PDT is an excellent therapeutic option that is able to provide efficacious clearance of AKs as well as superior cosmetic outcomes. Common AEs of PDT include burning, itching, and stinging during therapy, but pain intensity decreases dramatically upon termination of illumination, with cessation of most symptoms by 12 hours posttherapy.73 Permanent pigmentation changes have been reported to occasionally occur following PDT therapy.81
Conclusion
When determining which therapy to use in a patient, clinicians must take into account a variety of factors such as patient preference, cost of treatment, availability, tolerance for AEs, and the need for field therapy. Although all therapies discussed within this article are effective and reasonable treatment choices, patients who are particularly concerned about cosmetic outcomes would most likely benefit from either IMQ or PDT, as the data for cosmetic outcomes with these therapies are the strongest. Combination or sequential treatments may be required in some cases and all patients should be monitored for lesion recurrence regardless of treatment choice. A summary of the therapies and key studies discussed here is available in the PDF.
- Lebwohl M. Actinic keratosis: epidemiology and progression to squamous cell carcinoma. Br J Dermatol. 2003;149(suppl 66):31-33.
- Drake LA, Ceilley RI, Cornelison RL, et al. Guidelines of care for actinic keratoses. Committee on Guidelines of Care. J Am Acad Dermatol. 1995;32:95-98.
- Salasche SJ. Epidemiology of actinic keratoses and squamous cell carcinoma. J Am Acad Dermatol. 2000;42(1, pt 2):4-7.
- Ackerman AB, Mones JM. Solar (actinic) keratosis is squamous cell carcinoma. Br J Dermatol. 2006;155:9-22.
- Anwar J, Wrone DA, Kimyai-Asadi A, et al. The development of actinic keratosis into invasive squamous cell carcinoma: evidence and evolving classification schemes. Clin Dermatol. 2004;22:189-196.
- Criscione VD, Weinstock MA, Naylor MF, et al. Actinic keratoses: natural history and risk of malignant transformation in the Veterans Affairs Topical Tretinoin Chemoprevention Trial. Cancer. 2009;115:2523-2530.
- Glogau RG. The risk of progression to invasive disease. J Am Acad Dermatol. 2000;42(1, pt 2):23-24.
- Esmann S, Jemec GB. Management of actinic keratosis patients: a qualitative study. J Dermatolog Treat. 2007;18:53-58.
- Weinstock MA, Lee KC, Chren MM, et al. Quality of life in the actinic neoplasia syndrome: the VA Topical Tretinoin Chemoprevention (VATTC) trial. J Am Acad Dermatol. 2009;61:207-215.
- Berman B, Villa AM, Ramirez CC. Mechanisms of action of new treatment modalities for actinic keratosis. J Drugs Dermatol. 2006;5:167-173.
- Askew DA, Mickan SM, Soyer HP. Effectiveness of 5-fluorouracil treatment for actinic keratosis: a systematic review of randomized controlled trials. Int J Dermatol. 2009;46:452-463.
- Levy S, Furst K, Chern W. A pharmacokinetic evaluation of 0.5% and 5% fluorouracil topical cream in patients with actinic keratosis. Clin Ther. 2001;23:908-920.
- Jorizzo J, Stewart D, Bucko A, et al. Randomized trial evaluating a new 0.5% fluorouracil formulation demonstrates efficacy after 1-, 2-, or 4-week treatment in patients with actinic keratosis. Cutis. 2002;70:335-359.
- Sachs DL, Kang S, Hammerberg C, et al. Topical fluorouracil for actinic keratoses and photoaging: a clinical and molecular analysis. Arch Dermatol. 2009;145:659-666.
- Hantash BM, Stewart DB, Cooper ZA, et al. Facial resurfacing for nonmelanoma skin cancer prophylaxis. Arch Dermatol. 2006;142:976-982.
- Lawrence N, Cox SE, Cockerell CJ, et al. A comparison of the efficacy and safety of Jessner’s solution and 35% trichloroacetic acid vs 5% fluorouracil in the treatment of widespread facial actinic keratoses. Arch Dermatol. 1995;131:176-181.
- Monheit GD. The Jessner’s + TCA peel: a medium-depth chemical peel. J Dermatol Surg Oncol. 1989;15:945-950.
- Hemmi H, Kaisho T, Takeuchi O, et al. Small anti-viral compounds activate immune cells via the TLR7 MyD88-dependent signaling pathway. Nat Immunol. 2002;3:196-200.
- Adamson DJ, Frew D, Tatoud R, et al. Diclofenac antagonizes peroxisome proliferator-activated receptor-gamma signaling. Mol Pharmacol. 2002;61:7-12.
- Alam CA, Seed MP, Willoughby DA. Angiostasis and vascular regression in chronic granulomatous inflammation induced by diclofenac in combination with hyaluronan in mice. J Pharm Pharmacol. 1995;47:407-411.
- Lu X, Xie W, Reed D, et al. Nonsteroidal antiinflammatory drugs cause apoptosis and induce cyclooxygenases in chicken embryo fibroblasts. Proc Natl Acad Sci USA. 1995;92:7961-7965.
- Seed MP, Brown JR, Freemantle CN, et al. The inhibition of colon-26 adenocarcinoma development and angiogenesis by topical diclofenac in 2.5% hyaluronan. Cancer Res. 1997;57:1625-1629.
- Rivers JK, Arlette J, Shear N, et al. Topical treatment of actinic keratoses with 3.0% diclofenac in 2.5% hyaluronan gel. Br J Dermatol. 2002;146:94-100.
- Wolf JE, Taylor JR, Tschen E, et al. Topical 3.0% diclo-fenac in 2.5% hyaluronan gel in the treatment of actinic keratoses. Int J Dermatol. 2001;40:709-713.
- Nelson C, Rigel D, Smith S, et al. Phase IV, open-label assessment of the treatment of actinic keratosis with 3.0% diclofenac sodium topical gel (Solaraze). J Drugs Dermatol. 2004;3:401-407.
- Pflugfelder A, Welter AK, Leiter U, et al. Open label randomized study comparing 3 months vs. 6 months treatment of actinic keratoses with 3% diclofenac in 2.5% hyaluronic acid gel: a trial of the German Dermatologic Cooperative Oncology Group. J Eur Acad Dermatol Venereol. 2012;26:48-53.
- Smith SR, Morhenn VB, Piacquadio DJ. Bilateral comparison of the efficacy and tolerability of 3% diclofenac sodium gel and 5% 5-fluorouracil cream in the treatment of actinic keratoses of the face and scalp. J Drugs Dermatol. 2006;5:156-159.
- Segatto MM, Dornelles SI, Silveira VB, et al. Comparative study of actinic keratosis treatment with 3% diclo- fenac sodium and 5% 5-fluorouracil. An Bras Dermatol. 2013;88:732-738.
- Vidal D. Topical imiquimod: mechanism of action and clinical applications. Mini Rev Med Chem. 2006;6:499-503.
- Hadley G, Derry S, Moore RA. Imiquimod for actinic keratosis: systematic review and meta-analysis. J Invest Dermatol. 2006;126:1251-1255.
- Caperton C, Berman B. Safety, efficacy, and patient acceptability of imiquimod for topical treatment of actinic keratoses. Clin Cosmet Investig Dermatol. 2011;4:35-40.
- Swanson N, Smith CC, Kaur M, et al. Imiquimod 2.5% and 3.75% for the treatment of actinic keratoses: two phase 3, multicenter, randomized, double-blind, placebo-controlled studies. J Drugs Dermatol. 2014;13:166-169.
- Krawtchenko N, Roewert-Huber J, Ulrich M, et al. A randomised study of topical 5% imiquimod vs. topical 5-fluorouracil vs. cryosurgery in immunocompetent patients with actinic keratoses: a comparison of clinical and histological outcomes including 1-year follow-up. Br J Dermatol. 2007;157(suppl 2):34-40.
- Anderson L, Schmieder GJ, Werschler WP, et al. Randomized, double-blind, double-dummy, vehicle-controlled study of ingenol mebutate gel 0.025% and 0.05% for actinic keratosis. J Am Acad Dermatol. 2009;60:934-943.
- Ogbourne SM, Suhrbier A, Jones B, et al. Antitumor activity of 3-ingenyl angelate: plasma membrane and mitochondrial disruption and necrotic cell death. Cancer Res. 2004;64:2833-2839.
- Lebwohl M, Swanson N, Anderson LL, et al. Ingenol mebutate gel for actinic keratosis. N Engl J Med. 2012;366:1010-1019.
- Lebwohl M, Shumack S, Stein-Gold L, et al. Long-term follow-up study of ingenol mebutate gel for the treatment of actinic keratoses. JAMA Dermatol. 2013;149:666-670.
- Augustin M, Tu JH, Knudsen KM, et al. Ingenol mebutate gel for actinic keratosis: the link between quality of life, treatment satisfaction, and clinical outcomes. J Am Acad Dermatol. 2015;72:816-821.
- Kane-Maguire N, Moseley R, Cozzi S, et al. Modulation of fibroblast phenotype and extracellular matrix composition by ingenol mebutate may be associated with scar resolution and improved dermal cosmesis. J Am Acad Dermatol. 2012;66:AB218.
- Martin G, Swanson N. Clinical findings using ingenol mebutate gel to treat actinic keratoses. J Am Acad Dermatol. 2013;68(1, suppl 1):S39-S48.
- Feldman SR, Fleischer AB, Williford PM, et al. Destructive procedures are the standard of care for treatment of actinic keratoses. J Am Acad Dermatol. 1999;40:43-47.
- Berlin JM. Current and emerging treatment strategies for the treatment of actinic keratosis. Clin Cosmet Investig Dermatol. 2010;3:119-126.
- Coleman WP, Yarborough JM, Mandy SH. Dermabrasion for prophylaxis and treatment of actinic keratoses. Dermatol Surg. 1996;22:17-21.
- Cooley JE, Casey DL, Kauffman CL. Manual resurfacing and trichloroacetic acid for the treatment of patients with widespread actinic damage. clinical and histologic observations. Dermatol Surg. 1997;23:373-379.
- Goldberg LH, Kaplan B, Vergilis-Kalner I, et al. Liquid nitrogen: temperature control in the treatment of actinic keratosis. Dermatol Surg. 2010;36:1956-1961.
- Lubritz RR, Smolewski SA. Cryosurgery cure rate of actinic keratoses. J Am Acad Dermatol. 1982;7:631-632.
- Thai KE, Fergin P, Freeman M, et al. A prospective study of the use of cryosurgery for the treatment of actinic keratoses. Int J Dermatol. 2004;43:687-692.
- Zane C, Facchinetti E, Rossi MT, et al. Cryotherapy is preferable to ablative CO2 laser for the treatment of isolated actinic keratoses of the face and scalp: a randomized clinical trial. Br J Dermatol. 2014;170:1114-1121.
- Trimas SJ, Ellis DA, Metz RD. The carbon dioxide laser. an alternative for the treatment of actinically damaged skin. Dermatol Surg. 1997;23:885-889.
- Jiang SB, Levine VJ, Nehal KS, et al. Er:YAG laser for the treatment of actinic keratoses. Dermatol Surg. 2000;26:437-440.
- Ostertag JU, Quaedvlieg PJ, Van der geer S, et al. A clinical comparison and long-term follow-up of topical 5-fluorouracil versus laser resurfacing in the treatment of widespread actinic keratoses. Lasers Surg Med. 2006;38:731-739.
- Iyer S, Friedli A, Bowes L, et al. Full face laser resurfacing: therapy and prophylaxis for actinic keratoses and non-melanoma skin cancer. Lasers Surg Med. 2004;34:114-119.
- Rubin MG. A peeler’s thoughts on skin improvement with chemical peels and laser resurfacing. Clin Plast Surg. 1997;24:407-409.
- Riggs K, Keller M, Humphreys TR. Ablative laser resurfacing: high-energy pulsed carbon dioxide and erbium:yttrium-aluminum-garnet. Clin Dermatol. 2007;25:462-473.
- Adrian RM. Pulsed carbon dioxide and long pulse 10-ms erbium-YAG laser resurfacing: a comparative clinical and histological study. J Cutan Laser Ther. 1999;1:197-202.
- Weiss ET, Brauer JA, Anolik R, et al. 1927-nm fractional resurfacing of facial actinic keratoses: a promising new therapeutic option. J Am Acad Dermatol. 2013; 68:98-102.
- Manstein D, Herron GS, Sink RK, et al. Fractional photothermolysis: a new concept for cutaneous remodeling using microscopic patterns of thermal injury. Lasers Surg Med. 2004;34:426-438.
- Katz TM, Goldberg LH, Marquez D, et al. Nonablative fractional photothermolysis for facial actinic keratoses: 6-month follow-up with histologic evaluation. J Am Acad Dermatol. 2011;65:349-356.
- Prens SP, De Vries K, Neumann HA, et al. Non-ablative fractional resurfacing in combination with topical tretinoin cream as a field treatment modality for multiple actinic keratosis: a pilot study and a review of other field treatment modalities. J Dermatolog Treat. 2013;24:227-231.
- Alexiades-Armenakas MR, Dover JS, Arndt KA. The spectrum of laser skin resurfacing: nonablative, fractional, and ablative laser resurfacing. J Am Acad Dermatol. 2008;58:719-737.
- Tannous Z. Fractional resurfacing. Clin Dermatol. 2007;25:480-486.
- Gold MH. Continuing medical education article-skin treatment: photodynamic therapy: indications and treatment. Aesthet Surg J. 2008;28:545-552.
- Juarranz A, Jaén P, Sanz-Rodríguez F, et al. Photodynamic therapy of cancer. basic principles and applications. Clin Transl Oncol. 2008;10:148-154.
- Juzeniene A, Peng Q, Moan J. Milestones in the development of photodynamic therapy and fluorescence diagnosis. Photochem Photobiol Sci. 2007;6:1234-1245.
- Moan J, Berg K. The photodegradation of porphyrins in cells can be used to estimate the lifetime of singlet oxygen. Photochem Photobiol. 1991;53:549-553.
- Gupta AK, Paquet M, Villanueva E, et al. Interventions for actinic keratoses. Cochrane Database Syst Rev. 2012;12:CD004415.
- Patel G, Armstrong AW, Eisen DB. Efficacy of photodynamic therapy vs other interventions in randomized clinical trials for the treatment of actinic keratoses: a systematic review and meta-analysis. JAMA Dermatol. 2014;150:1281-1288.
- Kaufmann R, Spelman L, Weightman W, et al. Multicentre intraindividual randomized trial of topical methyl aminolaevulinate-photodynamic therapy vs. cryotherapy for multiple actinic keratoses on the extremities. Br J Dermatol. 2008;158:994-999.
- Freeman M, Vinciullo C, Francis D, et al. A comparison of photodynamic therapy using topical methyl aminolevulinate (Metvix) with single cycle cryotherapy in patients with actinic keratosis: a prospective, randomized study. J Dermatolog Treat. 2003;14:99-106.
- Morton C, Campbell S, Gupta G, et al. Intraindividual, right-left comparison of topical methyl aminolaevulinate-photodynamic therapy and cryotherapy in subjects with actinic keratoses: a multicentre, randomized controlled study. Br J Dermatol. 2006;155:1029-1036.
- Pariser DM, Lowe NJ, Stewart DM, et al. Photodynamic therapy with topical methyl aminolevulinate for actinic keratosis: results of a prospective randomized multicenter trial. J Am Acad Dermatol. 2003;48:227-232.
- Zane C, Facchinetti E, Rossi MT, et al. A randomized clinical trial of photodynamic therapy with methyl aminolaevulinate vs. diclofenac 3% plus hyaluronic acid gel for the treatment of multiple actinic keratoses of the face and scalp. Br J Dermatol. 2014;170:1143-1150.
- Perrett CM, McGregor JM, Warwick J, et al. Treatment of post-transplant premalignant skin disease: a randomized intrapatient comparative study of 5-fluorouracil cream and topical photodynamic therapy. Br J Dermatol. 2007;156:320-328.
- Szeimies RM, Karrer S, Radakovic-Fijan S, et al. Photodynamic therapy using topical methyl 5-aminolevulinate compared with cryotherapy for actinic keratosis: a prospective, randomized study. J Am Acad Dermatol. 2002; 47:258-262.
- Scola N, Terras S, Georgas D, et al. A randomized, half-side comparative study of aminolaevulinate photodynamic therapy vs. CO(2) laser ablation in immunocompetent patients with multiple actinic keratoses. Br J Dermatol. 2012;167:1366-1373.
- Willey A, Anderson RR, Sakamoto FH. Temperature-modulated photodynamic therapy for the treatment of actinic keratosis on the extremities: a pilot study. Dermatol Surg. 2014;40:1094-1102.
- Pariser DM. Management of Actinic Keratoses: Treatment Selection and Optimizing Outcomes. Presented at: Winter Clinical Dermatology Conference Hawaii; January 18, 2015; Kaanapali, HI.
- Dirschka T, Radny P, Dominicus R, et al. Long-term (6 and 12 months) follow-up of two prospective, randomized, controlled phase III trials of photodynamic therapy with BF-200 ALA and methyl aminolaevulinate for the treatment of actinic keratosis. Br J Dermatol. 2013;168:825-836.
- Choi SH, Kim KH, Song KH. Efficacy of ablative fractional laser-assisted photodynamic therapy with short-incubation time for the treatment of facial and scalp actinic keratosis: 12-month follow-up results of a randomized, prospective, comparative trial. J Eur Acad Dermatol Venereol. 2015;29:1598-1605.
- Ko DY, Jeon SY, Kim KH, et al. Fractional erbium:YAG laser-assisted photodynamic therapy for facial actinic keratoses: a randomized, comparative, prospective study. J Eur Acad Dermatol Venereol. 2014;28:1529-1539.
- Togsverd-Ho K, Haak CS, Thaysen-Petersen D, et al. Intensified photodynamic therapy of actinic keratoses with fractional CO2 laser: a randomized clinical trial. Br J Dermatol. 2012;166:1262-1269.
- Torezan L, Chaves Y, Niwa A, et al. A pilot split-face study comparing conventional methyl aminolevulinate-photodynamic therapy (PDT) with microneedling-assisted PDT on actinically damaged skin. Dermatol Surg. 2013;39:1197-1201.
Actinic keratosis (AK), also referred to as solar keratosis or senile keratosis, is an intraepidermal proliferation of dysplastic keratinocytes that develops in response to chronic exposure to UV radiation. Actinic keratoses are among the most commonly encountered lesions seen by dermatologists, and it has been estimated that 60% of predisposed individuals older than 40 years have at least one AK.1,2 Prevalence is notably higher in light-skinned individuals and increases with age, presumably from higher cumulative sun exposure and decreased effectiveness of the immune system.1,3 It remains a point of contention as to whether or not AKs actually represent squamous cell carcinoma (SCC) in situ, but the potential for progression to invasive disease has been well demonstrated, as the majority of SCCs develop from preexisting AKs.4-6 The risk for progression to invasive disease for an individual AK has been estimated to range from 0.025% to 16% per year, with an average of approximately 8% in immunocompetent patients.7
The clinical morphology of AK can vary widely, but the most common presentation is an erythematous scaly macule, papule, or plaque on sun-exposed skin. The skin surrounding AKs typically shows evidence of solar damage with deep wrinkling, mottled pigmentation, scattered telangiectases, purpura, or xerosis (Figure). A variety of clinical variants with unique presentations exist, including atrophic, hypertrophic, acantholytic, lichenoid, bowenoid, and pigmented subtypes. Because more than 80% of AKs occur on highly visible areas such as the head, neck, back of the hands, and forearms, AKs can have an obvious detrimental effect on cosmetic appearance. Studies also have shown a strong association between AKs and decreased overall quality of life (QOL).3,8,9

Because of the risk for AK progression to invasive cancer along with its negative impact on cosmesis and QOL, clinicians generally opt to treat AKs. Numerous different treatment options exist, including topical medications, procedural modalities, and light-based therapies. Here, we review the efficacy of the most commonly utilized treatments and discuss the relevant cosmetic considerations and outcomes.
Topical Treatments
5-Fluorouracil
5-Fluorouracil (5-FU) is a US Food and Drug Administration (FDA)–approved, topically applied pyrimidine analogue that inhibits thymidylate synthase. The resulting suppression of DNA and RNA synthesis induces cell death with a preference for mitotically active cells.10 5-Fluorouracil has been used for more than 50 years as a treatment of AK and its efficacy is well established. A systematic review of 5 randomized controlled studies of topical 5-FU reported an average of 49% of 423 patients achieving complete lesion clearance with 5-FU cream 5% applied once or twice daily for up to 7 weeks.11 Some notable drawbacks of 5-FU, however, are application-site erythema, blistering, pruritus, necrosis, erosion, and pain. These effects often lead to premature cessation of therapy, but newer formulations of 5-FU cream 0.5% have shown good efficacy with better tolerability.12 A randomized, double-blind, multicenter, parallel-group study of 177 patients using 5-FU cream 0.5% once daily for either 1, 2, or 4 weeks demonstrated significant (P<.001) efficacy over vehicle gel in all treatment arms.13 The most effective therapy was 4 weeks of treatment, which achieved a mean 91.7% reduction in lesion count as assessed 1 month after cessation of therapy. The primary adverse effect (AE) reported in this trial was mild to moderate facial irritation, which generally resolved within 18 to 21 days after treatment cessation.13 Overall, 5-FU is a highly effective therapy for treating AKs that also can improve signs of photoaging, but patients should be aware of cosmetically unappealing effects that generally occur throughout therapy and during the immediate posttreatment period.14
Chemical Peels
Chemical peels traditionally employ acidic compounds to strip away outer layers of skin to variable depths depending on the concentration of the agent being applied. For treatment of AK, trichloroacetic acid (TCA) is a commonly employed cauterant that has shown efficacy comparable to topical 5-FU as well as ablative CO2 laser resurfacing.15 Trichloroacetic acid peels also are a convenient therapy, as good results can be achieved after a single treatment session. A split-face study of 15 patients treated with either a single application of 35% TCA and Jessner solution or twice-daily application of 5-FU cream 5% for 3 weeks demonstrated a reduction in 75% of visible AKs in both treatment arms over a 1-year follow-up period.16 Although 80% of patients self-reported considerable cosmetic improvement with both therapies, patient preference was reported to be in favor of the TCA peel, given its quick results and relatively mild side effects as compared to 5-FU. Treatment with chemical peels will result in temporary erythema and mild desquamation that usually resolves within 2 weeks; however, there are cases in which erythema has been reported to persist for several months.16 Adverse effects such as permanent scarring or pigmentation changes rarely are seen with TCA concentrations less than 45%.17 Caution should be used in patients with a history of herpes simplex virus, keloids, postinflammatory hyperpigmentation, radiation exposure, immunosuppression, and those unable or unwilling to use sunscreen and avoid sun exposure in the immediate posttreatment period.
Diclofenac Sodium
Diclofenac sodium (DFS) is an FDA-approved topical, nonsteroidal,
anti-inflammatory drug whose mechanism of action in the treatment of AK is thought to involve inhibition of the cyclooxygenase 2 enzyme.18 The resulting reduction of prostaglandins is believed to inhibit tumor angiogenesis, induce apoptosis, and inhibit cell differentiation.19-22 In a multicenter, double-blind, placebo-controlled study of 195 patients, application of DFS 3% in hyaluronan gel 2.5% twice daily for 60 days showed significant (P<.05) efficacy over placebo in achieving complete resolution of target lesions during a 30-day follow-up period (31% vs 10%). Furthermore, qualitative patient assessment of complete global improvement also was significantly (P<.05) higher in the active treatment group as compared to placebo (31% vs 10%).23 Additional studies of DFS 3% in hyaluronan gel 2.5% applied twice daily for 90 days have shown even higher rates of success, with complete resolution of target lesions in 40% to 58% of cases.24,25 This therapy also has been reported to substantially improve QOL following treatment completion.26 The most frequently cited AEs include pruritus, rash, dry skin, erythema, and application-site reactions. Overall, DFS is a
well-tolerated therapy with efficacy comparable to that of 5-FU but with a lower incidence of AEs
and higher patient satisfaction as determined in
2 head-to-head studies.27,28
ImiquimodImiquimod (IMQ) is an FDA-approved topical agent that functions as an immune response modifier via agonism of toll-like receptor 7.18 The resulting cytokine production and release enhances the innate and acquired immune responses leading to anticancer activity.29 The efficacy of IMQ for treatment of AK has been demonstrated in numerous well-designed clinical trials. A
meta-analysis of 5 randomized, double-blind trials including 1293 patients treated with IMQ cream 5%
2 to 3 times per week for 12 to 16 weeks reported complete clearance of AKs in 50% of patients treated with IMQ as compared to 5% of patients treated with vehicle.30 The most frequently reported AEs with this therapy include erythema, scabbing, flaking, and erosion. These effects generally resolve following cessation of treatment, and therapy is considered to be well tolerated; however, there are case reports of IMQ triggering or exacerbating existing inflammatory conditions.31 Imiquimod cream also is approved at 2.5% and 3.75% concentrations, which have demonstrated significant (P<.001) efficacy over placebo and a reduced incidence of AEs; complete clearance rates have been reported as 30.6% and 35.6%, respectively.32 Notably, a study comparing 75 patients randomized to either IMQ cream 5%
3 times per week for 4 weeks, 1 or 2 courses of cryosurgery, or 5-FU ointment 5% twice daily for 4 weeks reported that IMQ achieved significantly (P<.01) superior sustained clearance rates during a 12-month follow-up period over cryosurgery and 5-FU
(73% vs 4% vs 33%).33 Additionally, cosmetic outcomes as determined by both participants and investigators were reported as excellent at 12 months posttreatment in more than 80% of participants treated with IMQ. These excellent, long-lasting cosmetic outcomes also were determined to be significantly (P<.0001) superior to the cosmetic outcomes of 5-FU and cryotherapy, which both reported excellent outcomes in less than 10% of cases.33
Ingenol MebutateIngenol mebutate (IM) is a macrocyclic diterpene ester derived from the Euphorbia peplus plant that is FDA approved for the treatment of AK.1 Ingenol mebutate’s mechanism of action is thought to involve induction of cell death via disruption of the plasma membrane and mitochondria in addition to production of an inflammatory response, which produces tumor-specific antibodies and a large influx of neutrophils.34,35 The overall evidence for the efficacy of IM is strong. A combined analysis of 4 multicenter, randomized, double-blind studies of 1005 participants reported that IM gel 0.015% applied once daily for 3 days to the face or scalp was significantly superior (P<.001) to placebo in achieving complete clearance as assessed 54 days after completion of therapy (42.2% vs 3.7%) and that IM gel 0.05% applied once daily for 2 days to the trunk or extremities also was significantly superior (P<.001) to placebo in achieving complete clearance as determined 55 days after completion of therapy (34.1% vs 4.7%).36 A follow-up report to this study indicated that IM also appears to achieve long-lasting effects with an overall 87% decrease in total AKs at 12 months follow-up in both trial groups.37 Additionally, it has been recently reported that treatment with IM in these trials was associated with significantly higher overall treatment satisfaction (P<.001) and improved QOL (P<.001) as compared to vehicle.38 Cosmetic outcomes of IM therapy have been assessed in a trial analyzing the efficacy of IM gel 0.025% for 3 days or IM gel 0.05% for 2 or 3 days on nonfacial AKs. This study reported significantly (P<.0001) higher patient satisfaction with the cosmetic outcome at 8 weeks after therapy as compared to vehicle.34 Studies performed in mice have demonstrated that IM is able to promote collagen matrix turnover and impose dermal elasticity, which may contribute to these good cosmetic outcomes.39 The most common AEs of IM therapy are erythema, crusting, and flaking; these effects generally occur 3 to 8 days after starting treatment. These effects, however, generally are short lived and resolve within 2 weeks of treatment cessation when IM is applied to the face or scalp or 4 weeks when applied to the trunk or extremities.40 Overall, IM is a useful therapeutic option given its relatively short treatment course as compared to other topically applied agents, as well as its lasting efficacy, mild AEs, and good cosmetic outcomes.
Procedural Modalities
Surgical Procedures
Surgical approaches for the treatment of AK include excision, curettage with or without electrodesiccation, and dermabrasion. In the past, these modalities were used with greater frequency, but the advent of effective topical medications with lower risks of AEs has largely reduced their use.41 Excision may still be indicated in cases where SCC is suspected, and curettage can be used for treatment of thicker hypertrophic AKs.42 Although these approaches have not been evaluated in clinical trials, they are generally effective but require the use of local anesthetics and come with substantial risk for infection, permanent scarring, and hypopigmentation. Dermabrasion employs the use of a motorized device equipped with an abrasive material to physically remove superficial layers of the skin. Studies are limited, but this method has been reported as an effective treatment in a retrospective review of 23 participants in which 96% remained free of AKs at 1 year, 83% at 2 years, 64% at 4 years, and 54% at 5 years posttherapy.43 Notably, one split-face study of 40 participants treated with dermabrasion followed by 25% TCA on one side and either Jessner solution and 35% TCA or dermabrasion alone on the other side reported that the combination of dermabrasion with 25% TCA consistently produced excellent cosmetic results with nearly complete eradication of AKs.44 In general, however, cosmetic outcomes with dermabrasion are variable, as the technique is highly operator dependent and treatment is associated with notable discomfort as well as risk for scarring and permanent pigmentation alteration.
Cryotherapy
Cryotherapy remains one of the most commonly utilized treatments of AK and involves the delivery of liquid nitrogen via a spray device or a cotton tip applicator to rapidly freeze cells, thus causing cellular destruction via ice crystal formation and protein denaturation.45 Efficacy with this technique has been reported to be as high as 98.8% at 12 months follow-up, but more recent studies cite lower rates of success.46 A prospective multicenter study of 90 participants with 421 AKs on the face or scalp treated with a single freeze-thaw cycle of liquid nitrogen reported an overall complete response rate of 67.2% at 3 months posttherapy. Additionally, higher complete response rates were associated with longer freeze times, and cosmetic outcomes were reported as good to excellent in 94% of complete response lesions.47 Similar results were reported in an open-label, prospective, randomized, controlled clinical trial of 200 participants with 543 AKs, which compared a single freeze-thaw cycle with liquid nitrogen to a single session of CO2 laser ablation in the treatment of isolated AKs of the face and scalp.48 At 3 months posttherapy, complete clearance was observed in 71.6% of participants treated with cryotherapy and in 65.3% of participants treated with laser ablation (P=.532). At 12 months posttherapy, participants who originally showed complete response at 3 months were assessed for relapse. Complete clearance was preserved in 72.6% of participants treated with cryotherapy versus 21.9% of participants treated with laser ablation (P<.0001), and cosmetic outcomes were reported by participants as good or excellent at 3 months follow-up in more than 93% of participants for both treatment arms.48 Possible AEs of cryotherapy include pain during treatment, blister formation with possible hemorrhage, infection, scarring, and permanent pigmentary changes.47,48 Notably, the risk for hypopigmentation increases with longer freezing times, thus requiring clinicians to consider the balance between improved efficacy and reduced cosmetic outcomes.47
Light-Based Therapies
Laser Therapy
Ablative laser resurfacing with either the CO2 or erbium-doped:YAG (Er:YAG) laser utilizes light of specific wavelengths to selectively induce thermolysis and destruction of the epidermal layer. Both lasers have been studied as treatments of AK, but there is a lack of large, well-designed studies. In one small study of 14 participants treated with 1 to 2 passes of the CO2 laser, complete clearance was reported in all cases without any recurrences during a follow-up period of 6 to 24 months. Additionally, all participants in this study reported satisfaction with the cosmetic outcome.49 The CO2 laser also has demonstrated efficacy comparable to that of the TCA peel and 5-FU therapy in a prospective randomized trial of 34 patients with facial or scalp AKs who received either CO2 laser with 2 passes, 30% TCA peel, or 5-FU cream 5% twice daily for 3 weeks.15 Reduction in mean AK counts at 3 months posttherapy was significantly (P<.03) higher in all treatment arms as compared to the control group (92% for CO2 laser, 89% for TCA peel, and 83% for 5-FU cream). No significant (P=.31) difference in outcomes was noted among the different treatment arms.15 Similar results were reported for the Er:YAG laser in a small prospective study of 5 participants treated with 2 to 3 passes with the Er:YAG laser in which reduction in mean AK counts was reported as ranging from 86% to 96% at 3 months posttherapy.50 The Er:YAG laser in combination with the CO2 laser has shown notable long-term efficacy in achieving higher lesion clearance rates and sustained complete clearance rates over treatment with topical 5-FU.51 In a prospective randomized study of 55 par-ticipants with multiple AKs on the face or scalp, participants were assigned to receive either combination laser ablation with the Er:YAG and CO2 lasers down to the level of the papillary dermis or 5-FU cream 5% applied twice daily for 2 to 7 weeks until an appropriate clinical inflammatory response was achieved. At 12 months follow-up, the laser treatment group achieved significantly (P=.048) higher mean lesion clearance rates (91.1%) as compared to the 5-FU arm (76.6%) and significantly (P=.003) higher sustained complete clearance rates (59.3%) as compared to 5-FU (29.2%). The proportion of participants with an improvement in photoaging score at 12 months follow-up approached statistical significance (P=.07), with 74% of the laser-treated group showing improvement as compared to 43% of the 5-FU–treated group. Long-term, cosmetically unappealing side effects such as erythema and hypopigmentation occurred notably more often in the laser-treated group as compared to the 5-FU group.51 In summary, ablative lasers appear to be a highly effective therapy for AK but at the cost of increased risk for AEs such as permanent pigmentary changes, prolonged erythema lasting up to several months, and scarring.50,52-55
Fractional photothermolysis is a relatively new advancement in the field of laser therapy that has received FDA approval for the treatment of AK.56 This treatment works by creating multiple noncontiguous microscopic columns of thermal injury while sparing adjacent zones of viable tissue.57 Although there are limited studies involving the use of such lasers in the treatment of AK, initial findings suggest that 1927-nm thulium lasers may be more effective than 1550-nm erbium lasers in achieving lesion clearance. A trial of 14 participants who received 5 laser treatments with a 1550-nm fractionated erbium-doped fiber laser reported an average reduction in AK counts of 66.2% at 3 months follow-up and a 55.6% reduction at 6 months follow-up. A participant-determined marked or very significant improvement of lesions was reported in 83% of participants at 1 month posttreatment but only in 44% of participants at 6 months posttreatment.58 A similar trial of 24 participants treated with up to 4 treatment sessions of the fractionated 1927-nm thulium laser reported an 87.3% reduction in number of AKs at 3 months follow-up and an 86.6% reduction at 6 months follow-up.56 The primary advantage of fractional laser therapy is a faster recovery period generally lasting only 2 or 3 days as compared to 2 weeks or more with traditional ablative lasers, thus limiting the amount of time a patient must tolerate cosmetically unappealing erythema.59,60 The quick recovery time has been attributed to the fractional laser’s ability to preserve the stratum corneum and skin barrier, which also helps reduce the risk for other AEs such as scarring and infection.56,59-61 Additional studies are needed to better assess the true efficacy of fractional laser therapy, but treatment with the fractional 1927-nm thulium laser appears to be a promising and well-tolerated therapeutic option for treatment of AK with similar efficacy to traditional ablative lasers but with a lower risk of AEs.
Photodynamic TherapyPhotodynamic therapy (PDT) is an FDA-approved treatment that involves the use of a topical photosensitizing agent such as 5-aminolevulinic acid (ALA) or methyl aminovulinate (MAL) before exposure to an activating light source to generate reactive oxygen species that lead to cell death.62-65 Multiple PDT regimens with varying combinations of photosensitizers, incubation time, and light sources have been studied, but a 2012 Cochrane review determined that treatment with conventional formulations of MAL and ALA with either blue- or red-light PDT were similarly efficacious for treatment of individual AKs as compared to vehicle with blue- or red-light PDT. One exception was that longer incubation time (ie, 4 hours) with ALA resulted in better results than shorter incubation times (ie, 0.5, 1, 2 hours) with ALA.66
Standard PDT treatment with MAL also has consistently demonstrated superior efficacy in achieving complete clearance rates in addition to superior cosmetic outcomes over treatment with either cryotherapy, DFS, or 5-FU.67-73 Three studies in particular noted an excellent or good investigator-determined cosmetic outcome in 96% to 98% of participants treated with MAL-PDT.69,71,74 Photodynamic therapy with ALA also has been reported as superior over CO2 laser ablation for AK reduction as well as both patient and investigator overall satisfaction.75
More recently, several methods of improving photosensitizer delivery have been studied, which have demonstrated remarkable efficacy at achieving lesion clearance over standard cream formulations or application routines. One such method involves the use of gentle heating to increase photosensitizer uptake. In a split-extremity study of 20 participants who were treated with 20% ALA under occlusion for 1 hour with one side heated to 38.8°C, the heated side demonstrated significant (P<.0001) efficacy at achieving higher median clearance rates over control when evaluated at 2 and 6 months posttherapy.76 Notably, occlusion of ALA in itself during the incubation period also has been demonstrated to significantly (P<.0001) improve clearance rates.77 Another method involves the use of a new nanoemulsion-based formulation of ALA gel, known as BF-200 ALA, which has demonstrated remarkable efficacy over standard MAL cream and placebo in a long-term follow-up analysis of 2 prospective, randomized, controlled trials.78 In a similar vein, 3 prospective randomized trials with a minimum follow-up time of 3 months demonstrated that MAL-PDT in combination with fractional ablative laser pretreatment has significant (P<.02 in all trials) efficacy over MAL-PDT without pretreatment in achieving complete AK clearance. Although the cosmetic outcomes were good or excellent in 87% to 100% of patients, they were not significantly different from stand-alone MAL-PDT treatment in any of the trials.79-81 However, pretreatment with microneedling in MAL-PDT has been shown to achieve superior cosmetic outcomes over MAL-PDT without microneedling, according to one small split-face study of 10 participants.82
Overall, PDT is an excellent therapeutic option that is able to provide efficacious clearance of AKs as well as superior cosmetic outcomes. Common AEs of PDT include burning, itching, and stinging during therapy, but pain intensity decreases dramatically upon termination of illumination, with cessation of most symptoms by 12 hours posttherapy.73 Permanent pigmentation changes have been reported to occasionally occur following PDT therapy.81
Conclusion
When determining which therapy to use in a patient, clinicians must take into account a variety of factors such as patient preference, cost of treatment, availability, tolerance for AEs, and the need for field therapy. Although all therapies discussed within this article are effective and reasonable treatment choices, patients who are particularly concerned about cosmetic outcomes would most likely benefit from either IMQ or PDT, as the data for cosmetic outcomes with these therapies are the strongest. Combination or sequential treatments may be required in some cases and all patients should be monitored for lesion recurrence regardless of treatment choice. A summary of the therapies and key studies discussed here is available in the PDF.
Actinic keratosis (AK), also referred to as solar keratosis or senile keratosis, is an intraepidermal proliferation of dysplastic keratinocytes that develops in response to chronic exposure to UV radiation. Actinic keratoses are among the most commonly encountered lesions seen by dermatologists, and it has been estimated that 60% of predisposed individuals older than 40 years have at least one AK.1,2 Prevalence is notably higher in light-skinned individuals and increases with age, presumably from higher cumulative sun exposure and decreased effectiveness of the immune system.1,3 It remains a point of contention as to whether or not AKs actually represent squamous cell carcinoma (SCC) in situ, but the potential for progression to invasive disease has been well demonstrated, as the majority of SCCs develop from preexisting AKs.4-6 The risk for progression to invasive disease for an individual AK has been estimated to range from 0.025% to 16% per year, with an average of approximately 8% in immunocompetent patients.7
The clinical morphology of AK can vary widely, but the most common presentation is an erythematous scaly macule, papule, or plaque on sun-exposed skin. The skin surrounding AKs typically shows evidence of solar damage with deep wrinkling, mottled pigmentation, scattered telangiectases, purpura, or xerosis (Figure). A variety of clinical variants with unique presentations exist, including atrophic, hypertrophic, acantholytic, lichenoid, bowenoid, and pigmented subtypes. Because more than 80% of AKs occur on highly visible areas such as the head, neck, back of the hands, and forearms, AKs can have an obvious detrimental effect on cosmetic appearance. Studies also have shown a strong association between AKs and decreased overall quality of life (QOL).3,8,9

Because of the risk for AK progression to invasive cancer along with its negative impact on cosmesis and QOL, clinicians generally opt to treat AKs. Numerous different treatment options exist, including topical medications, procedural modalities, and light-based therapies. Here, we review the efficacy of the most commonly utilized treatments and discuss the relevant cosmetic considerations and outcomes.
Topical Treatments
5-Fluorouracil
5-Fluorouracil (5-FU) is a US Food and Drug Administration (FDA)–approved, topically applied pyrimidine analogue that inhibits thymidylate synthase. The resulting suppression of DNA and RNA synthesis induces cell death with a preference for mitotically active cells.10 5-Fluorouracil has been used for more than 50 years as a treatment of AK and its efficacy is well established. A systematic review of 5 randomized controlled studies of topical 5-FU reported an average of 49% of 423 patients achieving complete lesion clearance with 5-FU cream 5% applied once or twice daily for up to 7 weeks.11 Some notable drawbacks of 5-FU, however, are application-site erythema, blistering, pruritus, necrosis, erosion, and pain. These effects often lead to premature cessation of therapy, but newer formulations of 5-FU cream 0.5% have shown good efficacy with better tolerability.12 A randomized, double-blind, multicenter, parallel-group study of 177 patients using 5-FU cream 0.5% once daily for either 1, 2, or 4 weeks demonstrated significant (P<.001) efficacy over vehicle gel in all treatment arms.13 The most effective therapy was 4 weeks of treatment, which achieved a mean 91.7% reduction in lesion count as assessed 1 month after cessation of therapy. The primary adverse effect (AE) reported in this trial was mild to moderate facial irritation, which generally resolved within 18 to 21 days after treatment cessation.13 Overall, 5-FU is a highly effective therapy for treating AKs that also can improve signs of photoaging, but patients should be aware of cosmetically unappealing effects that generally occur throughout therapy and during the immediate posttreatment period.14
Chemical Peels
Chemical peels traditionally employ acidic compounds to strip away outer layers of skin to variable depths depending on the concentration of the agent being applied. For treatment of AK, trichloroacetic acid (TCA) is a commonly employed cauterant that has shown efficacy comparable to topical 5-FU as well as ablative CO2 laser resurfacing.15 Trichloroacetic acid peels also are a convenient therapy, as good results can be achieved after a single treatment session. A split-face study of 15 patients treated with either a single application of 35% TCA and Jessner solution or twice-daily application of 5-FU cream 5% for 3 weeks demonstrated a reduction in 75% of visible AKs in both treatment arms over a 1-year follow-up period.16 Although 80% of patients self-reported considerable cosmetic improvement with both therapies, patient preference was reported to be in favor of the TCA peel, given its quick results and relatively mild side effects as compared to 5-FU. Treatment with chemical peels will result in temporary erythema and mild desquamation that usually resolves within 2 weeks; however, there are cases in which erythema has been reported to persist for several months.16 Adverse effects such as permanent scarring or pigmentation changes rarely are seen with TCA concentrations less than 45%.17 Caution should be used in patients with a history of herpes simplex virus, keloids, postinflammatory hyperpigmentation, radiation exposure, immunosuppression, and those unable or unwilling to use sunscreen and avoid sun exposure in the immediate posttreatment period.
Diclofenac Sodium
Diclofenac sodium (DFS) is an FDA-approved topical, nonsteroidal,
anti-inflammatory drug whose mechanism of action in the treatment of AK is thought to involve inhibition of the cyclooxygenase 2 enzyme.18 The resulting reduction of prostaglandins is believed to inhibit tumor angiogenesis, induce apoptosis, and inhibit cell differentiation.19-22 In a multicenter, double-blind, placebo-controlled study of 195 patients, application of DFS 3% in hyaluronan gel 2.5% twice daily for 60 days showed significant (P<.05) efficacy over placebo in achieving complete resolution of target lesions during a 30-day follow-up period (31% vs 10%). Furthermore, qualitative patient assessment of complete global improvement also was significantly (P<.05) higher in the active treatment group as compared to placebo (31% vs 10%).23 Additional studies of DFS 3% in hyaluronan gel 2.5% applied twice daily for 90 days have shown even higher rates of success, with complete resolution of target lesions in 40% to 58% of cases.24,25 This therapy also has been reported to substantially improve QOL following treatment completion.26 The most frequently cited AEs include pruritus, rash, dry skin, erythema, and application-site reactions. Overall, DFS is a
well-tolerated therapy with efficacy comparable to that of 5-FU but with a lower incidence of AEs
and higher patient satisfaction as determined in
2 head-to-head studies.27,28
ImiquimodImiquimod (IMQ) is an FDA-approved topical agent that functions as an immune response modifier via agonism of toll-like receptor 7.18 The resulting cytokine production and release enhances the innate and acquired immune responses leading to anticancer activity.29 The efficacy of IMQ for treatment of AK has been demonstrated in numerous well-designed clinical trials. A
meta-analysis of 5 randomized, double-blind trials including 1293 patients treated with IMQ cream 5%
2 to 3 times per week for 12 to 16 weeks reported complete clearance of AKs in 50% of patients treated with IMQ as compared to 5% of patients treated with vehicle.30 The most frequently reported AEs with this therapy include erythema, scabbing, flaking, and erosion. These effects generally resolve following cessation of treatment, and therapy is considered to be well tolerated; however, there are case reports of IMQ triggering or exacerbating existing inflammatory conditions.31 Imiquimod cream also is approved at 2.5% and 3.75% concentrations, which have demonstrated significant (P<.001) efficacy over placebo and a reduced incidence of AEs; complete clearance rates have been reported as 30.6% and 35.6%, respectively.32 Notably, a study comparing 75 patients randomized to either IMQ cream 5%
3 times per week for 4 weeks, 1 or 2 courses of cryosurgery, or 5-FU ointment 5% twice daily for 4 weeks reported that IMQ achieved significantly (P<.01) superior sustained clearance rates during a 12-month follow-up period over cryosurgery and 5-FU
(73% vs 4% vs 33%).33 Additionally, cosmetic outcomes as determined by both participants and investigators were reported as excellent at 12 months posttreatment in more than 80% of participants treated with IMQ. These excellent, long-lasting cosmetic outcomes also were determined to be significantly (P<.0001) superior to the cosmetic outcomes of 5-FU and cryotherapy, which both reported excellent outcomes in less than 10% of cases.33
Ingenol MebutateIngenol mebutate (IM) is a macrocyclic diterpene ester derived from the Euphorbia peplus plant that is FDA approved for the treatment of AK.1 Ingenol mebutate’s mechanism of action is thought to involve induction of cell death via disruption of the plasma membrane and mitochondria in addition to production of an inflammatory response, which produces tumor-specific antibodies and a large influx of neutrophils.34,35 The overall evidence for the efficacy of IM is strong. A combined analysis of 4 multicenter, randomized, double-blind studies of 1005 participants reported that IM gel 0.015% applied once daily for 3 days to the face or scalp was significantly superior (P<.001) to placebo in achieving complete clearance as assessed 54 days after completion of therapy (42.2% vs 3.7%) and that IM gel 0.05% applied once daily for 2 days to the trunk or extremities also was significantly superior (P<.001) to placebo in achieving complete clearance as determined 55 days after completion of therapy (34.1% vs 4.7%).36 A follow-up report to this study indicated that IM also appears to achieve long-lasting effects with an overall 87% decrease in total AKs at 12 months follow-up in both trial groups.37 Additionally, it has been recently reported that treatment with IM in these trials was associated with significantly higher overall treatment satisfaction (P<.001) and improved QOL (P<.001) as compared to vehicle.38 Cosmetic outcomes of IM therapy have been assessed in a trial analyzing the efficacy of IM gel 0.025% for 3 days or IM gel 0.05% for 2 or 3 days on nonfacial AKs. This study reported significantly (P<.0001) higher patient satisfaction with the cosmetic outcome at 8 weeks after therapy as compared to vehicle.34 Studies performed in mice have demonstrated that IM is able to promote collagen matrix turnover and impose dermal elasticity, which may contribute to these good cosmetic outcomes.39 The most common AEs of IM therapy are erythema, crusting, and flaking; these effects generally occur 3 to 8 days after starting treatment. These effects, however, generally are short lived and resolve within 2 weeks of treatment cessation when IM is applied to the face or scalp or 4 weeks when applied to the trunk or extremities.40 Overall, IM is a useful therapeutic option given its relatively short treatment course as compared to other topically applied agents, as well as its lasting efficacy, mild AEs, and good cosmetic outcomes.
Procedural Modalities
Surgical Procedures
Surgical approaches for the treatment of AK include excision, curettage with or without electrodesiccation, and dermabrasion. In the past, these modalities were used with greater frequency, but the advent of effective topical medications with lower risks of AEs has largely reduced their use.41 Excision may still be indicated in cases where SCC is suspected, and curettage can be used for treatment of thicker hypertrophic AKs.42 Although these approaches have not been evaluated in clinical trials, they are generally effective but require the use of local anesthetics and come with substantial risk for infection, permanent scarring, and hypopigmentation. Dermabrasion employs the use of a motorized device equipped with an abrasive material to physically remove superficial layers of the skin. Studies are limited, but this method has been reported as an effective treatment in a retrospective review of 23 participants in which 96% remained free of AKs at 1 year, 83% at 2 years, 64% at 4 years, and 54% at 5 years posttherapy.43 Notably, one split-face study of 40 participants treated with dermabrasion followed by 25% TCA on one side and either Jessner solution and 35% TCA or dermabrasion alone on the other side reported that the combination of dermabrasion with 25% TCA consistently produced excellent cosmetic results with nearly complete eradication of AKs.44 In general, however, cosmetic outcomes with dermabrasion are variable, as the technique is highly operator dependent and treatment is associated with notable discomfort as well as risk for scarring and permanent pigmentation alteration.
Cryotherapy
Cryotherapy remains one of the most commonly utilized treatments of AK and involves the delivery of liquid nitrogen via a spray device or a cotton tip applicator to rapidly freeze cells, thus causing cellular destruction via ice crystal formation and protein denaturation.45 Efficacy with this technique has been reported to be as high as 98.8% at 12 months follow-up, but more recent studies cite lower rates of success.46 A prospective multicenter study of 90 participants with 421 AKs on the face or scalp treated with a single freeze-thaw cycle of liquid nitrogen reported an overall complete response rate of 67.2% at 3 months posttherapy. Additionally, higher complete response rates were associated with longer freeze times, and cosmetic outcomes were reported as good to excellent in 94% of complete response lesions.47 Similar results were reported in an open-label, prospective, randomized, controlled clinical trial of 200 participants with 543 AKs, which compared a single freeze-thaw cycle with liquid nitrogen to a single session of CO2 laser ablation in the treatment of isolated AKs of the face and scalp.48 At 3 months posttherapy, complete clearance was observed in 71.6% of participants treated with cryotherapy and in 65.3% of participants treated with laser ablation (P=.532). At 12 months posttherapy, participants who originally showed complete response at 3 months were assessed for relapse. Complete clearance was preserved in 72.6% of participants treated with cryotherapy versus 21.9% of participants treated with laser ablation (P<.0001), and cosmetic outcomes were reported by participants as good or excellent at 3 months follow-up in more than 93% of participants for both treatment arms.48 Possible AEs of cryotherapy include pain during treatment, blister formation with possible hemorrhage, infection, scarring, and permanent pigmentary changes.47,48 Notably, the risk for hypopigmentation increases with longer freezing times, thus requiring clinicians to consider the balance between improved efficacy and reduced cosmetic outcomes.47
Light-Based Therapies
Laser Therapy
Ablative laser resurfacing with either the CO2 or erbium-doped:YAG (Er:YAG) laser utilizes light of specific wavelengths to selectively induce thermolysis and destruction of the epidermal layer. Both lasers have been studied as treatments of AK, but there is a lack of large, well-designed studies. In one small study of 14 participants treated with 1 to 2 passes of the CO2 laser, complete clearance was reported in all cases without any recurrences during a follow-up period of 6 to 24 months. Additionally, all participants in this study reported satisfaction with the cosmetic outcome.49 The CO2 laser also has demonstrated efficacy comparable to that of the TCA peel and 5-FU therapy in a prospective randomized trial of 34 patients with facial or scalp AKs who received either CO2 laser with 2 passes, 30% TCA peel, or 5-FU cream 5% twice daily for 3 weeks.15 Reduction in mean AK counts at 3 months posttherapy was significantly (P<.03) higher in all treatment arms as compared to the control group (92% for CO2 laser, 89% for TCA peel, and 83% for 5-FU cream). No significant (P=.31) difference in outcomes was noted among the different treatment arms.15 Similar results were reported for the Er:YAG laser in a small prospective study of 5 participants treated with 2 to 3 passes with the Er:YAG laser in which reduction in mean AK counts was reported as ranging from 86% to 96% at 3 months posttherapy.50 The Er:YAG laser in combination with the CO2 laser has shown notable long-term efficacy in achieving higher lesion clearance rates and sustained complete clearance rates over treatment with topical 5-FU.51 In a prospective randomized study of 55 par-ticipants with multiple AKs on the face or scalp, participants were assigned to receive either combination laser ablation with the Er:YAG and CO2 lasers down to the level of the papillary dermis or 5-FU cream 5% applied twice daily for 2 to 7 weeks until an appropriate clinical inflammatory response was achieved. At 12 months follow-up, the laser treatment group achieved significantly (P=.048) higher mean lesion clearance rates (91.1%) as compared to the 5-FU arm (76.6%) and significantly (P=.003) higher sustained complete clearance rates (59.3%) as compared to 5-FU (29.2%). The proportion of participants with an improvement in photoaging score at 12 months follow-up approached statistical significance (P=.07), with 74% of the laser-treated group showing improvement as compared to 43% of the 5-FU–treated group. Long-term, cosmetically unappealing side effects such as erythema and hypopigmentation occurred notably more often in the laser-treated group as compared to the 5-FU group.51 In summary, ablative lasers appear to be a highly effective therapy for AK but at the cost of increased risk for AEs such as permanent pigmentary changes, prolonged erythema lasting up to several months, and scarring.50,52-55
Fractional photothermolysis is a relatively new advancement in the field of laser therapy that has received FDA approval for the treatment of AK.56 This treatment works by creating multiple noncontiguous microscopic columns of thermal injury while sparing adjacent zones of viable tissue.57 Although there are limited studies involving the use of such lasers in the treatment of AK, initial findings suggest that 1927-nm thulium lasers may be more effective than 1550-nm erbium lasers in achieving lesion clearance. A trial of 14 participants who received 5 laser treatments with a 1550-nm fractionated erbium-doped fiber laser reported an average reduction in AK counts of 66.2% at 3 months follow-up and a 55.6% reduction at 6 months follow-up. A participant-determined marked or very significant improvement of lesions was reported in 83% of participants at 1 month posttreatment but only in 44% of participants at 6 months posttreatment.58 A similar trial of 24 participants treated with up to 4 treatment sessions of the fractionated 1927-nm thulium laser reported an 87.3% reduction in number of AKs at 3 months follow-up and an 86.6% reduction at 6 months follow-up.56 The primary advantage of fractional laser therapy is a faster recovery period generally lasting only 2 or 3 days as compared to 2 weeks or more with traditional ablative lasers, thus limiting the amount of time a patient must tolerate cosmetically unappealing erythema.59,60 The quick recovery time has been attributed to the fractional laser’s ability to preserve the stratum corneum and skin barrier, which also helps reduce the risk for other AEs such as scarring and infection.56,59-61 Additional studies are needed to better assess the true efficacy of fractional laser therapy, but treatment with the fractional 1927-nm thulium laser appears to be a promising and well-tolerated therapeutic option for treatment of AK with similar efficacy to traditional ablative lasers but with a lower risk of AEs.
Photodynamic TherapyPhotodynamic therapy (PDT) is an FDA-approved treatment that involves the use of a topical photosensitizing agent such as 5-aminolevulinic acid (ALA) or methyl aminovulinate (MAL) before exposure to an activating light source to generate reactive oxygen species that lead to cell death.62-65 Multiple PDT regimens with varying combinations of photosensitizers, incubation time, and light sources have been studied, but a 2012 Cochrane review determined that treatment with conventional formulations of MAL and ALA with either blue- or red-light PDT were similarly efficacious for treatment of individual AKs as compared to vehicle with blue- or red-light PDT. One exception was that longer incubation time (ie, 4 hours) with ALA resulted in better results than shorter incubation times (ie, 0.5, 1, 2 hours) with ALA.66
Standard PDT treatment with MAL also has consistently demonstrated superior efficacy in achieving complete clearance rates in addition to superior cosmetic outcomes over treatment with either cryotherapy, DFS, or 5-FU.67-73 Three studies in particular noted an excellent or good investigator-determined cosmetic outcome in 96% to 98% of participants treated with MAL-PDT.69,71,74 Photodynamic therapy with ALA also has been reported as superior over CO2 laser ablation for AK reduction as well as both patient and investigator overall satisfaction.75
More recently, several methods of improving photosensitizer delivery have been studied, which have demonstrated remarkable efficacy at achieving lesion clearance over standard cream formulations or application routines. One such method involves the use of gentle heating to increase photosensitizer uptake. In a split-extremity study of 20 participants who were treated with 20% ALA under occlusion for 1 hour with one side heated to 38.8°C, the heated side demonstrated significant (P<.0001) efficacy at achieving higher median clearance rates over control when evaluated at 2 and 6 months posttherapy.76 Notably, occlusion of ALA in itself during the incubation period also has been demonstrated to significantly (P<.0001) improve clearance rates.77 Another method involves the use of a new nanoemulsion-based formulation of ALA gel, known as BF-200 ALA, which has demonstrated remarkable efficacy over standard MAL cream and placebo in a long-term follow-up analysis of 2 prospective, randomized, controlled trials.78 In a similar vein, 3 prospective randomized trials with a minimum follow-up time of 3 months demonstrated that MAL-PDT in combination with fractional ablative laser pretreatment has significant (P<.02 in all trials) efficacy over MAL-PDT without pretreatment in achieving complete AK clearance. Although the cosmetic outcomes were good or excellent in 87% to 100% of patients, they were not significantly different from stand-alone MAL-PDT treatment in any of the trials.79-81 However, pretreatment with microneedling in MAL-PDT has been shown to achieve superior cosmetic outcomes over MAL-PDT without microneedling, according to one small split-face study of 10 participants.82
Overall, PDT is an excellent therapeutic option that is able to provide efficacious clearance of AKs as well as superior cosmetic outcomes. Common AEs of PDT include burning, itching, and stinging during therapy, but pain intensity decreases dramatically upon termination of illumination, with cessation of most symptoms by 12 hours posttherapy.73 Permanent pigmentation changes have been reported to occasionally occur following PDT therapy.81
Conclusion
When determining which therapy to use in a patient, clinicians must take into account a variety of factors such as patient preference, cost of treatment, availability, tolerance for AEs, and the need for field therapy. Although all therapies discussed within this article are effective and reasonable treatment choices, patients who are particularly concerned about cosmetic outcomes would most likely benefit from either IMQ or PDT, as the data for cosmetic outcomes with these therapies are the strongest. Combination or sequential treatments may be required in some cases and all patients should be monitored for lesion recurrence regardless of treatment choice. A summary of the therapies and key studies discussed here is available in the PDF.
- Lebwohl M. Actinic keratosis: epidemiology and progression to squamous cell carcinoma. Br J Dermatol. 2003;149(suppl 66):31-33.
- Drake LA, Ceilley RI, Cornelison RL, et al. Guidelines of care for actinic keratoses. Committee on Guidelines of Care. J Am Acad Dermatol. 1995;32:95-98.
- Salasche SJ. Epidemiology of actinic keratoses and squamous cell carcinoma. J Am Acad Dermatol. 2000;42(1, pt 2):4-7.
- Ackerman AB, Mones JM. Solar (actinic) keratosis is squamous cell carcinoma. Br J Dermatol. 2006;155:9-22.
- Anwar J, Wrone DA, Kimyai-Asadi A, et al. The development of actinic keratosis into invasive squamous cell carcinoma: evidence and evolving classification schemes. Clin Dermatol. 2004;22:189-196.
- Criscione VD, Weinstock MA, Naylor MF, et al. Actinic keratoses: natural history and risk of malignant transformation in the Veterans Affairs Topical Tretinoin Chemoprevention Trial. Cancer. 2009;115:2523-2530.
- Glogau RG. The risk of progression to invasive disease. J Am Acad Dermatol. 2000;42(1, pt 2):23-24.
- Esmann S, Jemec GB. Management of actinic keratosis patients: a qualitative study. J Dermatolog Treat. 2007;18:53-58.
- Weinstock MA, Lee KC, Chren MM, et al. Quality of life in the actinic neoplasia syndrome: the VA Topical Tretinoin Chemoprevention (VATTC) trial. J Am Acad Dermatol. 2009;61:207-215.
- Berman B, Villa AM, Ramirez CC. Mechanisms of action of new treatment modalities for actinic keratosis. J Drugs Dermatol. 2006;5:167-173.
- Askew DA, Mickan SM, Soyer HP. Effectiveness of 5-fluorouracil treatment for actinic keratosis: a systematic review of randomized controlled trials. Int J Dermatol. 2009;46:452-463.
- Levy S, Furst K, Chern W. A pharmacokinetic evaluation of 0.5% and 5% fluorouracil topical cream in patients with actinic keratosis. Clin Ther. 2001;23:908-920.
- Jorizzo J, Stewart D, Bucko A, et al. Randomized trial evaluating a new 0.5% fluorouracil formulation demonstrates efficacy after 1-, 2-, or 4-week treatment in patients with actinic keratosis. Cutis. 2002;70:335-359.
- Sachs DL, Kang S, Hammerberg C, et al. Topical fluorouracil for actinic keratoses and photoaging: a clinical and molecular analysis. Arch Dermatol. 2009;145:659-666.
- Hantash BM, Stewart DB, Cooper ZA, et al. Facial resurfacing for nonmelanoma skin cancer prophylaxis. Arch Dermatol. 2006;142:976-982.
- Lawrence N, Cox SE, Cockerell CJ, et al. A comparison of the efficacy and safety of Jessner’s solution and 35% trichloroacetic acid vs 5% fluorouracil in the treatment of widespread facial actinic keratoses. Arch Dermatol. 1995;131:176-181.
- Monheit GD. The Jessner’s + TCA peel: a medium-depth chemical peel. J Dermatol Surg Oncol. 1989;15:945-950.
- Hemmi H, Kaisho T, Takeuchi O, et al. Small anti-viral compounds activate immune cells via the TLR7 MyD88-dependent signaling pathway. Nat Immunol. 2002;3:196-200.
- Adamson DJ, Frew D, Tatoud R, et al. Diclofenac antagonizes peroxisome proliferator-activated receptor-gamma signaling. Mol Pharmacol. 2002;61:7-12.
- Alam CA, Seed MP, Willoughby DA. Angiostasis and vascular regression in chronic granulomatous inflammation induced by diclofenac in combination with hyaluronan in mice. J Pharm Pharmacol. 1995;47:407-411.
- Lu X, Xie W, Reed D, et al. Nonsteroidal antiinflammatory drugs cause apoptosis and induce cyclooxygenases in chicken embryo fibroblasts. Proc Natl Acad Sci USA. 1995;92:7961-7965.
- Seed MP, Brown JR, Freemantle CN, et al. The inhibition of colon-26 adenocarcinoma development and angiogenesis by topical diclofenac in 2.5% hyaluronan. Cancer Res. 1997;57:1625-1629.
- Rivers JK, Arlette J, Shear N, et al. Topical treatment of actinic keratoses with 3.0% diclofenac in 2.5% hyaluronan gel. Br J Dermatol. 2002;146:94-100.
- Wolf JE, Taylor JR, Tschen E, et al. Topical 3.0% diclo-fenac in 2.5% hyaluronan gel in the treatment of actinic keratoses. Int J Dermatol. 2001;40:709-713.
- Nelson C, Rigel D, Smith S, et al. Phase IV, open-label assessment of the treatment of actinic keratosis with 3.0% diclofenac sodium topical gel (Solaraze). J Drugs Dermatol. 2004;3:401-407.
- Pflugfelder A, Welter AK, Leiter U, et al. Open label randomized study comparing 3 months vs. 6 months treatment of actinic keratoses with 3% diclofenac in 2.5% hyaluronic acid gel: a trial of the German Dermatologic Cooperative Oncology Group. J Eur Acad Dermatol Venereol. 2012;26:48-53.
- Smith SR, Morhenn VB, Piacquadio DJ. Bilateral comparison of the efficacy and tolerability of 3% diclofenac sodium gel and 5% 5-fluorouracil cream in the treatment of actinic keratoses of the face and scalp. J Drugs Dermatol. 2006;5:156-159.
- Segatto MM, Dornelles SI, Silveira VB, et al. Comparative study of actinic keratosis treatment with 3% diclo- fenac sodium and 5% 5-fluorouracil. An Bras Dermatol. 2013;88:732-738.
- Vidal D. Topical imiquimod: mechanism of action and clinical applications. Mini Rev Med Chem. 2006;6:499-503.
- Hadley G, Derry S, Moore RA. Imiquimod for actinic keratosis: systematic review and meta-analysis. J Invest Dermatol. 2006;126:1251-1255.
- Caperton C, Berman B. Safety, efficacy, and patient acceptability of imiquimod for topical treatment of actinic keratoses. Clin Cosmet Investig Dermatol. 2011;4:35-40.
- Swanson N, Smith CC, Kaur M, et al. Imiquimod 2.5% and 3.75% for the treatment of actinic keratoses: two phase 3, multicenter, randomized, double-blind, placebo-controlled studies. J Drugs Dermatol. 2014;13:166-169.
- Krawtchenko N, Roewert-Huber J, Ulrich M, et al. A randomised study of topical 5% imiquimod vs. topical 5-fluorouracil vs. cryosurgery in immunocompetent patients with actinic keratoses: a comparison of clinical and histological outcomes including 1-year follow-up. Br J Dermatol. 2007;157(suppl 2):34-40.
- Anderson L, Schmieder GJ, Werschler WP, et al. Randomized, double-blind, double-dummy, vehicle-controlled study of ingenol mebutate gel 0.025% and 0.05% for actinic keratosis. J Am Acad Dermatol. 2009;60:934-943.
- Ogbourne SM, Suhrbier A, Jones B, et al. Antitumor activity of 3-ingenyl angelate: plasma membrane and mitochondrial disruption and necrotic cell death. Cancer Res. 2004;64:2833-2839.
- Lebwohl M, Swanson N, Anderson LL, et al. Ingenol mebutate gel for actinic keratosis. N Engl J Med. 2012;366:1010-1019.
- Lebwohl M, Shumack S, Stein-Gold L, et al. Long-term follow-up study of ingenol mebutate gel for the treatment of actinic keratoses. JAMA Dermatol. 2013;149:666-670.
- Augustin M, Tu JH, Knudsen KM, et al. Ingenol mebutate gel for actinic keratosis: the link between quality of life, treatment satisfaction, and clinical outcomes. J Am Acad Dermatol. 2015;72:816-821.
- Kane-Maguire N, Moseley R, Cozzi S, et al. Modulation of fibroblast phenotype and extracellular matrix composition by ingenol mebutate may be associated with scar resolution and improved dermal cosmesis. J Am Acad Dermatol. 2012;66:AB218.
- Martin G, Swanson N. Clinical findings using ingenol mebutate gel to treat actinic keratoses. J Am Acad Dermatol. 2013;68(1, suppl 1):S39-S48.
- Feldman SR, Fleischer AB, Williford PM, et al. Destructive procedures are the standard of care for treatment of actinic keratoses. J Am Acad Dermatol. 1999;40:43-47.
- Berlin JM. Current and emerging treatment strategies for the treatment of actinic keratosis. Clin Cosmet Investig Dermatol. 2010;3:119-126.
- Coleman WP, Yarborough JM, Mandy SH. Dermabrasion for prophylaxis and treatment of actinic keratoses. Dermatol Surg. 1996;22:17-21.
- Cooley JE, Casey DL, Kauffman CL. Manual resurfacing and trichloroacetic acid for the treatment of patients with widespread actinic damage. clinical and histologic observations. Dermatol Surg. 1997;23:373-379.
- Goldberg LH, Kaplan B, Vergilis-Kalner I, et al. Liquid nitrogen: temperature control in the treatment of actinic keratosis. Dermatol Surg. 2010;36:1956-1961.
- Lubritz RR, Smolewski SA. Cryosurgery cure rate of actinic keratoses. J Am Acad Dermatol. 1982;7:631-632.
- Thai KE, Fergin P, Freeman M, et al. A prospective study of the use of cryosurgery for the treatment of actinic keratoses. Int J Dermatol. 2004;43:687-692.
- Zane C, Facchinetti E, Rossi MT, et al. Cryotherapy is preferable to ablative CO2 laser for the treatment of isolated actinic keratoses of the face and scalp: a randomized clinical trial. Br J Dermatol. 2014;170:1114-1121.
- Trimas SJ, Ellis DA, Metz RD. The carbon dioxide laser. an alternative for the treatment of actinically damaged skin. Dermatol Surg. 1997;23:885-889.
- Jiang SB, Levine VJ, Nehal KS, et al. Er:YAG laser for the treatment of actinic keratoses. Dermatol Surg. 2000;26:437-440.
- Ostertag JU, Quaedvlieg PJ, Van der geer S, et al. A clinical comparison and long-term follow-up of topical 5-fluorouracil versus laser resurfacing in the treatment of widespread actinic keratoses. Lasers Surg Med. 2006;38:731-739.
- Iyer S, Friedli A, Bowes L, et al. Full face laser resurfacing: therapy and prophylaxis for actinic keratoses and non-melanoma skin cancer. Lasers Surg Med. 2004;34:114-119.
- Rubin MG. A peeler’s thoughts on skin improvement with chemical peels and laser resurfacing. Clin Plast Surg. 1997;24:407-409.
- Riggs K, Keller M, Humphreys TR. Ablative laser resurfacing: high-energy pulsed carbon dioxide and erbium:yttrium-aluminum-garnet. Clin Dermatol. 2007;25:462-473.
- Adrian RM. Pulsed carbon dioxide and long pulse 10-ms erbium-YAG laser resurfacing: a comparative clinical and histological study. J Cutan Laser Ther. 1999;1:197-202.
- Weiss ET, Brauer JA, Anolik R, et al. 1927-nm fractional resurfacing of facial actinic keratoses: a promising new therapeutic option. J Am Acad Dermatol. 2013; 68:98-102.
- Manstein D, Herron GS, Sink RK, et al. Fractional photothermolysis: a new concept for cutaneous remodeling using microscopic patterns of thermal injury. Lasers Surg Med. 2004;34:426-438.
- Katz TM, Goldberg LH, Marquez D, et al. Nonablative fractional photothermolysis for facial actinic keratoses: 6-month follow-up with histologic evaluation. J Am Acad Dermatol. 2011;65:349-356.
- Prens SP, De Vries K, Neumann HA, et al. Non-ablative fractional resurfacing in combination with topical tretinoin cream as a field treatment modality for multiple actinic keratosis: a pilot study and a review of other field treatment modalities. J Dermatolog Treat. 2013;24:227-231.
- Alexiades-Armenakas MR, Dover JS, Arndt KA. The spectrum of laser skin resurfacing: nonablative, fractional, and ablative laser resurfacing. J Am Acad Dermatol. 2008;58:719-737.
- Tannous Z. Fractional resurfacing. Clin Dermatol. 2007;25:480-486.
- Gold MH. Continuing medical education article-skin treatment: photodynamic therapy: indications and treatment. Aesthet Surg J. 2008;28:545-552.
- Juarranz A, Jaén P, Sanz-Rodríguez F, et al. Photodynamic therapy of cancer. basic principles and applications. Clin Transl Oncol. 2008;10:148-154.
- Juzeniene A, Peng Q, Moan J. Milestones in the development of photodynamic therapy and fluorescence diagnosis. Photochem Photobiol Sci. 2007;6:1234-1245.
- Moan J, Berg K. The photodegradation of porphyrins in cells can be used to estimate the lifetime of singlet oxygen. Photochem Photobiol. 1991;53:549-553.
- Gupta AK, Paquet M, Villanueva E, et al. Interventions for actinic keratoses. Cochrane Database Syst Rev. 2012;12:CD004415.
- Patel G, Armstrong AW, Eisen DB. Efficacy of photodynamic therapy vs other interventions in randomized clinical trials for the treatment of actinic keratoses: a systematic review and meta-analysis. JAMA Dermatol. 2014;150:1281-1288.
- Kaufmann R, Spelman L, Weightman W, et al. Multicentre intraindividual randomized trial of topical methyl aminolaevulinate-photodynamic therapy vs. cryotherapy for multiple actinic keratoses on the extremities. Br J Dermatol. 2008;158:994-999.
- Freeman M, Vinciullo C, Francis D, et al. A comparison of photodynamic therapy using topical methyl aminolevulinate (Metvix) with single cycle cryotherapy in patients with actinic keratosis: a prospective, randomized study. J Dermatolog Treat. 2003;14:99-106.
- Morton C, Campbell S, Gupta G, et al. Intraindividual, right-left comparison of topical methyl aminolaevulinate-photodynamic therapy and cryotherapy in subjects with actinic keratoses: a multicentre, randomized controlled study. Br J Dermatol. 2006;155:1029-1036.
- Pariser DM, Lowe NJ, Stewart DM, et al. Photodynamic therapy with topical methyl aminolevulinate for actinic keratosis: results of a prospective randomized multicenter trial. J Am Acad Dermatol. 2003;48:227-232.
- Zane C, Facchinetti E, Rossi MT, et al. A randomized clinical trial of photodynamic therapy with methyl aminolaevulinate vs. diclofenac 3% plus hyaluronic acid gel for the treatment of multiple actinic keratoses of the face and scalp. Br J Dermatol. 2014;170:1143-1150.
- Perrett CM, McGregor JM, Warwick J, et al. Treatment of post-transplant premalignant skin disease: a randomized intrapatient comparative study of 5-fluorouracil cream and topical photodynamic therapy. Br J Dermatol. 2007;156:320-328.
- Szeimies RM, Karrer S, Radakovic-Fijan S, et al. Photodynamic therapy using topical methyl 5-aminolevulinate compared with cryotherapy for actinic keratosis: a prospective, randomized study. J Am Acad Dermatol. 2002; 47:258-262.
- Scola N, Terras S, Georgas D, et al. A randomized, half-side comparative study of aminolaevulinate photodynamic therapy vs. CO(2) laser ablation in immunocompetent patients with multiple actinic keratoses. Br J Dermatol. 2012;167:1366-1373.
- Willey A, Anderson RR, Sakamoto FH. Temperature-modulated photodynamic therapy for the treatment of actinic keratosis on the extremities: a pilot study. Dermatol Surg. 2014;40:1094-1102.
- Pariser DM. Management of Actinic Keratoses: Treatment Selection and Optimizing Outcomes. Presented at: Winter Clinical Dermatology Conference Hawaii; January 18, 2015; Kaanapali, HI.
- Dirschka T, Radny P, Dominicus R, et al. Long-term (6 and 12 months) follow-up of two prospective, randomized, controlled phase III trials of photodynamic therapy with BF-200 ALA and methyl aminolaevulinate for the treatment of actinic keratosis. Br J Dermatol. 2013;168:825-836.
- Choi SH, Kim KH, Song KH. Efficacy of ablative fractional laser-assisted photodynamic therapy with short-incubation time for the treatment of facial and scalp actinic keratosis: 12-month follow-up results of a randomized, prospective, comparative trial. J Eur Acad Dermatol Venereol. 2015;29:1598-1605.
- Ko DY, Jeon SY, Kim KH, et al. Fractional erbium:YAG laser-assisted photodynamic therapy for facial actinic keratoses: a randomized, comparative, prospective study. J Eur Acad Dermatol Venereol. 2014;28:1529-1539.
- Togsverd-Ho K, Haak CS, Thaysen-Petersen D, et al. Intensified photodynamic therapy of actinic keratoses with fractional CO2 laser: a randomized clinical trial. Br J Dermatol. 2012;166:1262-1269.
- Torezan L, Chaves Y, Niwa A, et al. A pilot split-face study comparing conventional methyl aminolevulinate-photodynamic therapy (PDT) with microneedling-assisted PDT on actinically damaged skin. Dermatol Surg. 2013;39:1197-1201.
- Lebwohl M. Actinic keratosis: epidemiology and progression to squamous cell carcinoma. Br J Dermatol. 2003;149(suppl 66):31-33.
- Drake LA, Ceilley RI, Cornelison RL, et al. Guidelines of care for actinic keratoses. Committee on Guidelines of Care. J Am Acad Dermatol. 1995;32:95-98.
- Salasche SJ. Epidemiology of actinic keratoses and squamous cell carcinoma. J Am Acad Dermatol. 2000;42(1, pt 2):4-7.
- Ackerman AB, Mones JM. Solar (actinic) keratosis is squamous cell carcinoma. Br J Dermatol. 2006;155:9-22.
- Anwar J, Wrone DA, Kimyai-Asadi A, et al. The development of actinic keratosis into invasive squamous cell carcinoma: evidence and evolving classification schemes. Clin Dermatol. 2004;22:189-196.
- Criscione VD, Weinstock MA, Naylor MF, et al. Actinic keratoses: natural history and risk of malignant transformation in the Veterans Affairs Topical Tretinoin Chemoprevention Trial. Cancer. 2009;115:2523-2530.
- Glogau RG. The risk of progression to invasive disease. J Am Acad Dermatol. 2000;42(1, pt 2):23-24.
- Esmann S, Jemec GB. Management of actinic keratosis patients: a qualitative study. J Dermatolog Treat. 2007;18:53-58.
- Weinstock MA, Lee KC, Chren MM, et al. Quality of life in the actinic neoplasia syndrome: the VA Topical Tretinoin Chemoprevention (VATTC) trial. J Am Acad Dermatol. 2009;61:207-215.
- Berman B, Villa AM, Ramirez CC. Mechanisms of action of new treatment modalities for actinic keratosis. J Drugs Dermatol. 2006;5:167-173.
- Askew DA, Mickan SM, Soyer HP. Effectiveness of 5-fluorouracil treatment for actinic keratosis: a systematic review of randomized controlled trials. Int J Dermatol. 2009;46:452-463.
- Levy S, Furst K, Chern W. A pharmacokinetic evaluation of 0.5% and 5% fluorouracil topical cream in patients with actinic keratosis. Clin Ther. 2001;23:908-920.
- Jorizzo J, Stewart D, Bucko A, et al. Randomized trial evaluating a new 0.5% fluorouracil formulation demonstrates efficacy after 1-, 2-, or 4-week treatment in patients with actinic keratosis. Cutis. 2002;70:335-359.
- Sachs DL, Kang S, Hammerberg C, et al. Topical fluorouracil for actinic keratoses and photoaging: a clinical and molecular analysis. Arch Dermatol. 2009;145:659-666.
- Hantash BM, Stewart DB, Cooper ZA, et al. Facial resurfacing for nonmelanoma skin cancer prophylaxis. Arch Dermatol. 2006;142:976-982.
- Lawrence N, Cox SE, Cockerell CJ, et al. A comparison of the efficacy and safety of Jessner’s solution and 35% trichloroacetic acid vs 5% fluorouracil in the treatment of widespread facial actinic keratoses. Arch Dermatol. 1995;131:176-181.
- Monheit GD. The Jessner’s + TCA peel: a medium-depth chemical peel. J Dermatol Surg Oncol. 1989;15:945-950.
- Hemmi H, Kaisho T, Takeuchi O, et al. Small anti-viral compounds activate immune cells via the TLR7 MyD88-dependent signaling pathway. Nat Immunol. 2002;3:196-200.
- Adamson DJ, Frew D, Tatoud R, et al. Diclofenac antagonizes peroxisome proliferator-activated receptor-gamma signaling. Mol Pharmacol. 2002;61:7-12.
- Alam CA, Seed MP, Willoughby DA. Angiostasis and vascular regression in chronic granulomatous inflammation induced by diclofenac in combination with hyaluronan in mice. J Pharm Pharmacol. 1995;47:407-411.
- Lu X, Xie W, Reed D, et al. Nonsteroidal antiinflammatory drugs cause apoptosis and induce cyclooxygenases in chicken embryo fibroblasts. Proc Natl Acad Sci USA. 1995;92:7961-7965.
- Seed MP, Brown JR, Freemantle CN, et al. The inhibition of colon-26 adenocarcinoma development and angiogenesis by topical diclofenac in 2.5% hyaluronan. Cancer Res. 1997;57:1625-1629.
- Rivers JK, Arlette J, Shear N, et al. Topical treatment of actinic keratoses with 3.0% diclofenac in 2.5% hyaluronan gel. Br J Dermatol. 2002;146:94-100.
- Wolf JE, Taylor JR, Tschen E, et al. Topical 3.0% diclo-fenac in 2.5% hyaluronan gel in the treatment of actinic keratoses. Int J Dermatol. 2001;40:709-713.
- Nelson C, Rigel D, Smith S, et al. Phase IV, open-label assessment of the treatment of actinic keratosis with 3.0% diclofenac sodium topical gel (Solaraze). J Drugs Dermatol. 2004;3:401-407.
- Pflugfelder A, Welter AK, Leiter U, et al. Open label randomized study comparing 3 months vs. 6 months treatment of actinic keratoses with 3% diclofenac in 2.5% hyaluronic acid gel: a trial of the German Dermatologic Cooperative Oncology Group. J Eur Acad Dermatol Venereol. 2012;26:48-53.
- Smith SR, Morhenn VB, Piacquadio DJ. Bilateral comparison of the efficacy and tolerability of 3% diclofenac sodium gel and 5% 5-fluorouracil cream in the treatment of actinic keratoses of the face and scalp. J Drugs Dermatol. 2006;5:156-159.
- Segatto MM, Dornelles SI, Silveira VB, et al. Comparative study of actinic keratosis treatment with 3% diclo- fenac sodium and 5% 5-fluorouracil. An Bras Dermatol. 2013;88:732-738.
- Vidal D. Topical imiquimod: mechanism of action and clinical applications. Mini Rev Med Chem. 2006;6:499-503.
- Hadley G, Derry S, Moore RA. Imiquimod for actinic keratosis: systematic review and meta-analysis. J Invest Dermatol. 2006;126:1251-1255.
- Caperton C, Berman B. Safety, efficacy, and patient acceptability of imiquimod for topical treatment of actinic keratoses. Clin Cosmet Investig Dermatol. 2011;4:35-40.
- Swanson N, Smith CC, Kaur M, et al. Imiquimod 2.5% and 3.75% for the treatment of actinic keratoses: two phase 3, multicenter, randomized, double-blind, placebo-controlled studies. J Drugs Dermatol. 2014;13:166-169.
- Krawtchenko N, Roewert-Huber J, Ulrich M, et al. A randomised study of topical 5% imiquimod vs. topical 5-fluorouracil vs. cryosurgery in immunocompetent patients with actinic keratoses: a comparison of clinical and histological outcomes including 1-year follow-up. Br J Dermatol. 2007;157(suppl 2):34-40.
- Anderson L, Schmieder GJ, Werschler WP, et al. Randomized, double-blind, double-dummy, vehicle-controlled study of ingenol mebutate gel 0.025% and 0.05% for actinic keratosis. J Am Acad Dermatol. 2009;60:934-943.
- Ogbourne SM, Suhrbier A, Jones B, et al. Antitumor activity of 3-ingenyl angelate: plasma membrane and mitochondrial disruption and necrotic cell death. Cancer Res. 2004;64:2833-2839.
- Lebwohl M, Swanson N, Anderson LL, et al. Ingenol mebutate gel for actinic keratosis. N Engl J Med. 2012;366:1010-1019.
- Lebwohl M, Shumack S, Stein-Gold L, et al. Long-term follow-up study of ingenol mebutate gel for the treatment of actinic keratoses. JAMA Dermatol. 2013;149:666-670.
- Augustin M, Tu JH, Knudsen KM, et al. Ingenol mebutate gel for actinic keratosis: the link between quality of life, treatment satisfaction, and clinical outcomes. J Am Acad Dermatol. 2015;72:816-821.
- Kane-Maguire N, Moseley R, Cozzi S, et al. Modulation of fibroblast phenotype and extracellular matrix composition by ingenol mebutate may be associated with scar resolution and improved dermal cosmesis. J Am Acad Dermatol. 2012;66:AB218.
- Martin G, Swanson N. Clinical findings using ingenol mebutate gel to treat actinic keratoses. J Am Acad Dermatol. 2013;68(1, suppl 1):S39-S48.
- Feldman SR, Fleischer AB, Williford PM, et al. Destructive procedures are the standard of care for treatment of actinic keratoses. J Am Acad Dermatol. 1999;40:43-47.
- Berlin JM. Current and emerging treatment strategies for the treatment of actinic keratosis. Clin Cosmet Investig Dermatol. 2010;3:119-126.
- Coleman WP, Yarborough JM, Mandy SH. Dermabrasion for prophylaxis and treatment of actinic keratoses. Dermatol Surg. 1996;22:17-21.
- Cooley JE, Casey DL, Kauffman CL. Manual resurfacing and trichloroacetic acid for the treatment of patients with widespread actinic damage. clinical and histologic observations. Dermatol Surg. 1997;23:373-379.
- Goldberg LH, Kaplan B, Vergilis-Kalner I, et al. Liquid nitrogen: temperature control in the treatment of actinic keratosis. Dermatol Surg. 2010;36:1956-1961.
- Lubritz RR, Smolewski SA. Cryosurgery cure rate of actinic keratoses. J Am Acad Dermatol. 1982;7:631-632.
- Thai KE, Fergin P, Freeman M, et al. A prospective study of the use of cryosurgery for the treatment of actinic keratoses. Int J Dermatol. 2004;43:687-692.
- Zane C, Facchinetti E, Rossi MT, et al. Cryotherapy is preferable to ablative CO2 laser for the treatment of isolated actinic keratoses of the face and scalp: a randomized clinical trial. Br J Dermatol. 2014;170:1114-1121.
- Trimas SJ, Ellis DA, Metz RD. The carbon dioxide laser. an alternative for the treatment of actinically damaged skin. Dermatol Surg. 1997;23:885-889.
- Jiang SB, Levine VJ, Nehal KS, et al. Er:YAG laser for the treatment of actinic keratoses. Dermatol Surg. 2000;26:437-440.
- Ostertag JU, Quaedvlieg PJ, Van der geer S, et al. A clinical comparison and long-term follow-up of topical 5-fluorouracil versus laser resurfacing in the treatment of widespread actinic keratoses. Lasers Surg Med. 2006;38:731-739.
- Iyer S, Friedli A, Bowes L, et al. Full face laser resurfacing: therapy and prophylaxis for actinic keratoses and non-melanoma skin cancer. Lasers Surg Med. 2004;34:114-119.
- Rubin MG. A peeler’s thoughts on skin improvement with chemical peels and laser resurfacing. Clin Plast Surg. 1997;24:407-409.
- Riggs K, Keller M, Humphreys TR. Ablative laser resurfacing: high-energy pulsed carbon dioxide and erbium:yttrium-aluminum-garnet. Clin Dermatol. 2007;25:462-473.
- Adrian RM. Pulsed carbon dioxide and long pulse 10-ms erbium-YAG laser resurfacing: a comparative clinical and histological study. J Cutan Laser Ther. 1999;1:197-202.
- Weiss ET, Brauer JA, Anolik R, et al. 1927-nm fractional resurfacing of facial actinic keratoses: a promising new therapeutic option. J Am Acad Dermatol. 2013; 68:98-102.
- Manstein D, Herron GS, Sink RK, et al. Fractional photothermolysis: a new concept for cutaneous remodeling using microscopic patterns of thermal injury. Lasers Surg Med. 2004;34:426-438.
- Katz TM, Goldberg LH, Marquez D, et al. Nonablative fractional photothermolysis for facial actinic keratoses: 6-month follow-up with histologic evaluation. J Am Acad Dermatol. 2011;65:349-356.
- Prens SP, De Vries K, Neumann HA, et al. Non-ablative fractional resurfacing in combination with topical tretinoin cream as a field treatment modality for multiple actinic keratosis: a pilot study and a review of other field treatment modalities. J Dermatolog Treat. 2013;24:227-231.
- Alexiades-Armenakas MR, Dover JS, Arndt KA. The spectrum of laser skin resurfacing: nonablative, fractional, and ablative laser resurfacing. J Am Acad Dermatol. 2008;58:719-737.
- Tannous Z. Fractional resurfacing. Clin Dermatol. 2007;25:480-486.
- Gold MH. Continuing medical education article-skin treatment: photodynamic therapy: indications and treatment. Aesthet Surg J. 2008;28:545-552.
- Juarranz A, Jaén P, Sanz-Rodríguez F, et al. Photodynamic therapy of cancer. basic principles and applications. Clin Transl Oncol. 2008;10:148-154.
- Juzeniene A, Peng Q, Moan J. Milestones in the development of photodynamic therapy and fluorescence diagnosis. Photochem Photobiol Sci. 2007;6:1234-1245.
- Moan J, Berg K. The photodegradation of porphyrins in cells can be used to estimate the lifetime of singlet oxygen. Photochem Photobiol. 1991;53:549-553.
- Gupta AK, Paquet M, Villanueva E, et al. Interventions for actinic keratoses. Cochrane Database Syst Rev. 2012;12:CD004415.
- Patel G, Armstrong AW, Eisen DB. Efficacy of photodynamic therapy vs other interventions in randomized clinical trials for the treatment of actinic keratoses: a systematic review and meta-analysis. JAMA Dermatol. 2014;150:1281-1288.
- Kaufmann R, Spelman L, Weightman W, et al. Multicentre intraindividual randomized trial of topical methyl aminolaevulinate-photodynamic therapy vs. cryotherapy for multiple actinic keratoses on the extremities. Br J Dermatol. 2008;158:994-999.
- Freeman M, Vinciullo C, Francis D, et al. A comparison of photodynamic therapy using topical methyl aminolevulinate (Metvix) with single cycle cryotherapy in patients with actinic keratosis: a prospective, randomized study. J Dermatolog Treat. 2003;14:99-106.
- Morton C, Campbell S, Gupta G, et al. Intraindividual, right-left comparison of topical methyl aminolaevulinate-photodynamic therapy and cryotherapy in subjects with actinic keratoses: a multicentre, randomized controlled study. Br J Dermatol. 2006;155:1029-1036.
- Pariser DM, Lowe NJ, Stewart DM, et al. Photodynamic therapy with topical methyl aminolevulinate for actinic keratosis: results of a prospective randomized multicenter trial. J Am Acad Dermatol. 2003;48:227-232.
- Zane C, Facchinetti E, Rossi MT, et al. A randomized clinical trial of photodynamic therapy with methyl aminolaevulinate vs. diclofenac 3% plus hyaluronic acid gel for the treatment of multiple actinic keratoses of the face and scalp. Br J Dermatol. 2014;170:1143-1150.
- Perrett CM, McGregor JM, Warwick J, et al. Treatment of post-transplant premalignant skin disease: a randomized intrapatient comparative study of 5-fluorouracil cream and topical photodynamic therapy. Br J Dermatol. 2007;156:320-328.
- Szeimies RM, Karrer S, Radakovic-Fijan S, et al. Photodynamic therapy using topical methyl 5-aminolevulinate compared with cryotherapy for actinic keratosis: a prospective, randomized study. J Am Acad Dermatol. 2002; 47:258-262.
- Scola N, Terras S, Georgas D, et al. A randomized, half-side comparative study of aminolaevulinate photodynamic therapy vs. CO(2) laser ablation in immunocompetent patients with multiple actinic keratoses. Br J Dermatol. 2012;167:1366-1373.
- Willey A, Anderson RR, Sakamoto FH. Temperature-modulated photodynamic therapy for the treatment of actinic keratosis on the extremities: a pilot study. Dermatol Surg. 2014;40:1094-1102.
- Pariser DM. Management of Actinic Keratoses: Treatment Selection and Optimizing Outcomes. Presented at: Winter Clinical Dermatology Conference Hawaii; January 18, 2015; Kaanapali, HI.
- Dirschka T, Radny P, Dominicus R, et al. Long-term (6 and 12 months) follow-up of two prospective, randomized, controlled phase III trials of photodynamic therapy with BF-200 ALA and methyl aminolaevulinate for the treatment of actinic keratosis. Br J Dermatol. 2013;168:825-836.
- Choi SH, Kim KH, Song KH. Efficacy of ablative fractional laser-assisted photodynamic therapy with short-incubation time for the treatment of facial and scalp actinic keratosis: 12-month follow-up results of a randomized, prospective, comparative trial. J Eur Acad Dermatol Venereol. 2015;29:1598-1605.
- Ko DY, Jeon SY, Kim KH, et al. Fractional erbium:YAG laser-assisted photodynamic therapy for facial actinic keratoses: a randomized, comparative, prospective study. J Eur Acad Dermatol Venereol. 2014;28:1529-1539.
- Togsverd-Ho K, Haak CS, Thaysen-Petersen D, et al. Intensified photodynamic therapy of actinic keratoses with fractional CO2 laser: a randomized clinical trial. Br J Dermatol. 2012;166:1262-1269.
- Torezan L, Chaves Y, Niwa A, et al. A pilot split-face study comparing conventional methyl aminolevulinate-photodynamic therapy (PDT) with microneedling-assisted PDT on actinically damaged skin. Dermatol Surg. 2013;39:1197-1201.
Practice Points
- In addition to their risk for progression to malignancy, actinic keratoses (AKs) can have negative impacts on cosmetic appearance and quality of life.
- A variety of topical medications, procedural modalities, and light-based therapies are available for treatment of AKs, which offer varying degrees of efficacy for clearance of lesions and cosmetic outcomes. Based on the current data, imiquimod and photodynamic therapy are the treatments most likely to provide an excellent cosmetic outcome.
Medically Unlikely Edits
Medically Unlikely Edits (MUEs) are benchmarks recognized by the Centers for Medicare & Medicaid Services (CMS) that are designed to prevent incorrect or excessive coding. Specifically, an MUE is an edit that tests medical claims for services billed in excess of the maximum number of units of service permitted for a single beneficiary on the same date of service from the same provider (eg, multiples of the same Healthcare Common Procedure Coding System [HCPCS] code listed on different claim lines).1
The MUE System
If the number of units of service billed by the same physician for the same patient on the same day exceeds the maximum number permitted by the CMS, the Medicare Administrative Contractor (MAC) will deny the code or return the claim to the provider for correction (return to provider [RTP]). Units of service billed in excess of the MUE will not be paid, but other services billed on the same claim form may still be paid. In the case of an MUE-associated RTP, the provider should resubmit a corrected claim, not an appeal; however, an appeal is possible in the case of an MUE-associated denial. An MUE-associated denial is a coding denial, not a medical necessity denial; therefore, the provider cannot use an Advance Beneficiary Notice to transfer liability for claim payment to the patient.
MUE Adjudication Indicators
In 2013, the CMS modified the MUE process to include 3 different MUE adjudication indicators (MAIs) with a value of 1, 2, or 3 so that some MUE values would be date of service edits rather than claim line edits.2 Medically Unlikely Edits for HCPCS codes with an MAI of 1 are identical to the prior claim line edits. If a provider needs to report excess units of service with an MAI of 1, appropriate modifiers should be used to report them on separate lines of a claim. Current Procedural Terminology (CPT) modifiers such as -76 (repeat procedure or service by the same physician) and -91 (repeat clinical diagnostic laboratory test) as well as anatomic modifiers (eg, RT, LT, F1, F2) may be used, with modifier -59 (distinct procedural service) used only if no other modifier suffices. An example of an MUE with an MAI of 1 is CPT code 17264 (destruction, malignant lesion [eg, laser surgery, electrosurgery, cryosurgery, chemosurgery, surgical curettement], trunk, arms or legs; lesion diameter 3.1–4.0 cm), for which the MUE threshold is 3, meaning no more than 3 destructions can be submitted per claim line without triggering an edit-based rejection or RTP.
An MAI of 2 denotes absolute date of service edits, or so-called “per day edits based on policy.” Such edits are in place because units of service billed in excess of the MUE value on the same date of service are considered to be impossible by the CMS based on regulatory guidance or anatomic considerations.2 For instance, although the same physician may destroy multiple actinic keratoses in a single patient on the same date of service, it would not be possible to code more than one unit of service as
CPT code 17000, which specifically and exclusively
refers to the first lesion destroyed. Similarly,
CPT code 13101 (repair, complex, trunk; lesion diameter 2.6–7.5 cm) could only be reported once that day, as all complex repairs at that anatomic site must be summed and smaller or larger totals would be reported with another code.
Anatomic limitations are sometimes obvious and do not require specific coding rules. For example, only 1 gallbladder can be removed per patient. Although Qualified Independent Contractors and Administrative Law Judges are not bound by MAIs, they do give particular deference to an MAI of
2 given its definitive nature.2 Because ambulatory surgical center providers (Medicare specialty code 49) cannot report modifier -50 for bilateral
procedures, the MUE value used for editing is doubled for HCPCS codes with an MAI of 2 or 3 if the bilateral surgery indicator for the HCPCS code is 1.3
An MAI of 3 describes less strict date of service edits, so-called “per day edits based on clinical benchmarks.”2 Similar to MAIs of 1, MUEs for MAIs of 3 are based on medically likely daily frequencies of services provided in most settings. To determine if an MUE with an MAI of 3 has been reached, the MAC sums the units of service billed on all claim lines of the current claim as well as all prior paid claims for the same patient billed by the same provider on the same date of service. If the total units of service obtained in this manner exceeds the MUE value, then all claim lines with the relevant code for the current claim will be denied, but prior paid claims will not be adjusted. Denials based on MUEs for codes with an MAI of 3 can be appealed to the local MAC. Successful appeals require documentation that the units of service in excess of the MUE value were actually delivered and demonstration of medical necessity.2 An example of a CPT code with an MAI of 3 is 40490 (biopsy of lip) for which the MUE value is 3.
Complications With MUE and MAI
Because MUEs are based on current coding guidelines as well as current clinical practice, they are only applicable for the time period in which they are in effect. A change made to an MUE value for a particular code is not retroactive; however, in exceptional circumstances when a retroactive effective date is applied, MACs are not directed to examine prior claims but only “claims that are brought to their attention.”2
It also is important to realize that not all MUEs are publicly available and many are confidential. When claim denials occur, particularly in the context of multiple units of a particular code, automated MUE edits should be among the issues that are suspected. Physicians may resubmit RTP claims on separate lines if a claim line edit (MAI of 1) is operative. An MAI of 2 suggests a coding error that needs to be corrected, as these coding approaches are generally impossible based on definitional issues or anatomy. If an MUE with an MAI of 3 is the reason for denial, an appeal is possible, provided there is documentation to show that the service was actually provided and that it was medically necessary.
Final Thoughts
Dermatologists should be vigilant for unexpected payment denials, which may coincide with the implementation of new MUE values. When such denials occur and MUE values are publicly available, dermatologists should consider filing an appeal if the relevant MUEs were associated with an MAI of 1 or 3. Overall, dermatologists should be aware that many MUEs that were formerly claim line edits (MAI of 1) have been recently transitioned to date of service edits (MAI of 3), which are more restrictive.
1. American Academy of Dermatology. Medicare’s expanded medically unlikely edits. https://www.aad.org/members
/practice-and-advocacy-resource-center/coding-resources
/derm-coding-consult-library/winter-2014/medicare-
s-expanded-medically-unlikely-edits. Published Winter 2014. Accessed August 6, 2015.
2. Centers for Medicare & Medicaid Services. Revised modification to the Medically Unlikely Edit (MUE) program. MLN Matters. Number MM8853. https://www.cms.gov
/Outreach-and-Education/Medicare-Learning-Network-MLN/MLNMattersArticles/downloads/MM8853.pdf. Published January 1, 2015. Accessed August 6, 2015.
3. Centers for Medicare & Medicaid Services.
Medically Unlikely Edits (MUE) and bilateral procedures. MLN Matters. Number SE1422.
https://www.cms.gov/Regulations-and-Guidance
/Guidance/Transmittals/2014-Transmittals-Items/SE1422.html?DLPage=2&DLEntries=10&DLSort=1&DLSort
Dir=ascending. Accessed July 28, 2015.
Medically Unlikely Edits (MUEs) are benchmarks recognized by the Centers for Medicare & Medicaid Services (CMS) that are designed to prevent incorrect or excessive coding. Specifically, an MUE is an edit that tests medical claims for services billed in excess of the maximum number of units of service permitted for a single beneficiary on the same date of service from the same provider (eg, multiples of the same Healthcare Common Procedure Coding System [HCPCS] code listed on different claim lines).1
The MUE System
If the number of units of service billed by the same physician for the same patient on the same day exceeds the maximum number permitted by the CMS, the Medicare Administrative Contractor (MAC) will deny the code or return the claim to the provider for correction (return to provider [RTP]). Units of service billed in excess of the MUE will not be paid, but other services billed on the same claim form may still be paid. In the case of an MUE-associated RTP, the provider should resubmit a corrected claim, not an appeal; however, an appeal is possible in the case of an MUE-associated denial. An MUE-associated denial is a coding denial, not a medical necessity denial; therefore, the provider cannot use an Advance Beneficiary Notice to transfer liability for claim payment to the patient.
MUE Adjudication Indicators
In 2013, the CMS modified the MUE process to include 3 different MUE adjudication indicators (MAIs) with a value of 1, 2, or 3 so that some MUE values would be date of service edits rather than claim line edits.2 Medically Unlikely Edits for HCPCS codes with an MAI of 1 are identical to the prior claim line edits. If a provider needs to report excess units of service with an MAI of 1, appropriate modifiers should be used to report them on separate lines of a claim. Current Procedural Terminology (CPT) modifiers such as -76 (repeat procedure or service by the same physician) and -91 (repeat clinical diagnostic laboratory test) as well as anatomic modifiers (eg, RT, LT, F1, F2) may be used, with modifier -59 (distinct procedural service) used only if no other modifier suffices. An example of an MUE with an MAI of 1 is CPT code 17264 (destruction, malignant lesion [eg, laser surgery, electrosurgery, cryosurgery, chemosurgery, surgical curettement], trunk, arms or legs; lesion diameter 3.1–4.0 cm), for which the MUE threshold is 3, meaning no more than 3 destructions can be submitted per claim line without triggering an edit-based rejection or RTP.
An MAI of 2 denotes absolute date of service edits, or so-called “per day edits based on policy.” Such edits are in place because units of service billed in excess of the MUE value on the same date of service are considered to be impossible by the CMS based on regulatory guidance or anatomic considerations.2 For instance, although the same physician may destroy multiple actinic keratoses in a single patient on the same date of service, it would not be possible to code more than one unit of service as
CPT code 17000, which specifically and exclusively
refers to the first lesion destroyed. Similarly,
CPT code 13101 (repair, complex, trunk; lesion diameter 2.6–7.5 cm) could only be reported once that day, as all complex repairs at that anatomic site must be summed and smaller or larger totals would be reported with another code.
Anatomic limitations are sometimes obvious and do not require specific coding rules. For example, only 1 gallbladder can be removed per patient. Although Qualified Independent Contractors and Administrative Law Judges are not bound by MAIs, they do give particular deference to an MAI of
2 given its definitive nature.2 Because ambulatory surgical center providers (Medicare specialty code 49) cannot report modifier -50 for bilateral
procedures, the MUE value used for editing is doubled for HCPCS codes with an MAI of 2 or 3 if the bilateral surgery indicator for the HCPCS code is 1.3
An MAI of 3 describes less strict date of service edits, so-called “per day edits based on clinical benchmarks.”2 Similar to MAIs of 1, MUEs for MAIs of 3 are based on medically likely daily frequencies of services provided in most settings. To determine if an MUE with an MAI of 3 has been reached, the MAC sums the units of service billed on all claim lines of the current claim as well as all prior paid claims for the same patient billed by the same provider on the same date of service. If the total units of service obtained in this manner exceeds the MUE value, then all claim lines with the relevant code for the current claim will be denied, but prior paid claims will not be adjusted. Denials based on MUEs for codes with an MAI of 3 can be appealed to the local MAC. Successful appeals require documentation that the units of service in excess of the MUE value were actually delivered and demonstration of medical necessity.2 An example of a CPT code with an MAI of 3 is 40490 (biopsy of lip) for which the MUE value is 3.
Complications With MUE and MAI
Because MUEs are based on current coding guidelines as well as current clinical practice, they are only applicable for the time period in which they are in effect. A change made to an MUE value for a particular code is not retroactive; however, in exceptional circumstances when a retroactive effective date is applied, MACs are not directed to examine prior claims but only “claims that are brought to their attention.”2
It also is important to realize that not all MUEs are publicly available and many are confidential. When claim denials occur, particularly in the context of multiple units of a particular code, automated MUE edits should be among the issues that are suspected. Physicians may resubmit RTP claims on separate lines if a claim line edit (MAI of 1) is operative. An MAI of 2 suggests a coding error that needs to be corrected, as these coding approaches are generally impossible based on definitional issues or anatomy. If an MUE with an MAI of 3 is the reason for denial, an appeal is possible, provided there is documentation to show that the service was actually provided and that it was medically necessary.
Final Thoughts
Dermatologists should be vigilant for unexpected payment denials, which may coincide with the implementation of new MUE values. When such denials occur and MUE values are publicly available, dermatologists should consider filing an appeal if the relevant MUEs were associated with an MAI of 1 or 3. Overall, dermatologists should be aware that many MUEs that were formerly claim line edits (MAI of 1) have been recently transitioned to date of service edits (MAI of 3), which are more restrictive.
Medically Unlikely Edits (MUEs) are benchmarks recognized by the Centers for Medicare & Medicaid Services (CMS) that are designed to prevent incorrect or excessive coding. Specifically, an MUE is an edit that tests medical claims for services billed in excess of the maximum number of units of service permitted for a single beneficiary on the same date of service from the same provider (eg, multiples of the same Healthcare Common Procedure Coding System [HCPCS] code listed on different claim lines).1
The MUE System
If the number of units of service billed by the same physician for the same patient on the same day exceeds the maximum number permitted by the CMS, the Medicare Administrative Contractor (MAC) will deny the code or return the claim to the provider for correction (return to provider [RTP]). Units of service billed in excess of the MUE will not be paid, but other services billed on the same claim form may still be paid. In the case of an MUE-associated RTP, the provider should resubmit a corrected claim, not an appeal; however, an appeal is possible in the case of an MUE-associated denial. An MUE-associated denial is a coding denial, not a medical necessity denial; therefore, the provider cannot use an Advance Beneficiary Notice to transfer liability for claim payment to the patient.
MUE Adjudication Indicators
In 2013, the CMS modified the MUE process to include 3 different MUE adjudication indicators (MAIs) with a value of 1, 2, or 3 so that some MUE values would be date of service edits rather than claim line edits.2 Medically Unlikely Edits for HCPCS codes with an MAI of 1 are identical to the prior claim line edits. If a provider needs to report excess units of service with an MAI of 1, appropriate modifiers should be used to report them on separate lines of a claim. Current Procedural Terminology (CPT) modifiers such as -76 (repeat procedure or service by the same physician) and -91 (repeat clinical diagnostic laboratory test) as well as anatomic modifiers (eg, RT, LT, F1, F2) may be used, with modifier -59 (distinct procedural service) used only if no other modifier suffices. An example of an MUE with an MAI of 1 is CPT code 17264 (destruction, malignant lesion [eg, laser surgery, electrosurgery, cryosurgery, chemosurgery, surgical curettement], trunk, arms or legs; lesion diameter 3.1–4.0 cm), for which the MUE threshold is 3, meaning no more than 3 destructions can be submitted per claim line without triggering an edit-based rejection or RTP.
An MAI of 2 denotes absolute date of service edits, or so-called “per day edits based on policy.” Such edits are in place because units of service billed in excess of the MUE value on the same date of service are considered to be impossible by the CMS based on regulatory guidance or anatomic considerations.2 For instance, although the same physician may destroy multiple actinic keratoses in a single patient on the same date of service, it would not be possible to code more than one unit of service as
CPT code 17000, which specifically and exclusively
refers to the first lesion destroyed. Similarly,
CPT code 13101 (repair, complex, trunk; lesion diameter 2.6–7.5 cm) could only be reported once that day, as all complex repairs at that anatomic site must be summed and smaller or larger totals would be reported with another code.
Anatomic limitations are sometimes obvious and do not require specific coding rules. For example, only 1 gallbladder can be removed per patient. Although Qualified Independent Contractors and Administrative Law Judges are not bound by MAIs, they do give particular deference to an MAI of
2 given its definitive nature.2 Because ambulatory surgical center providers (Medicare specialty code 49) cannot report modifier -50 for bilateral
procedures, the MUE value used for editing is doubled for HCPCS codes with an MAI of 2 or 3 if the bilateral surgery indicator for the HCPCS code is 1.3
An MAI of 3 describes less strict date of service edits, so-called “per day edits based on clinical benchmarks.”2 Similar to MAIs of 1, MUEs for MAIs of 3 are based on medically likely daily frequencies of services provided in most settings. To determine if an MUE with an MAI of 3 has been reached, the MAC sums the units of service billed on all claim lines of the current claim as well as all prior paid claims for the same patient billed by the same provider on the same date of service. If the total units of service obtained in this manner exceeds the MUE value, then all claim lines with the relevant code for the current claim will be denied, but prior paid claims will not be adjusted. Denials based on MUEs for codes with an MAI of 3 can be appealed to the local MAC. Successful appeals require documentation that the units of service in excess of the MUE value were actually delivered and demonstration of medical necessity.2 An example of a CPT code with an MAI of 3 is 40490 (biopsy of lip) for which the MUE value is 3.
Complications With MUE and MAI
Because MUEs are based on current coding guidelines as well as current clinical practice, they are only applicable for the time period in which they are in effect. A change made to an MUE value for a particular code is not retroactive; however, in exceptional circumstances when a retroactive effective date is applied, MACs are not directed to examine prior claims but only “claims that are brought to their attention.”2
It also is important to realize that not all MUEs are publicly available and many are confidential. When claim denials occur, particularly in the context of multiple units of a particular code, automated MUE edits should be among the issues that are suspected. Physicians may resubmit RTP claims on separate lines if a claim line edit (MAI of 1) is operative. An MAI of 2 suggests a coding error that needs to be corrected, as these coding approaches are generally impossible based on definitional issues or anatomy. If an MUE with an MAI of 3 is the reason for denial, an appeal is possible, provided there is documentation to show that the service was actually provided and that it was medically necessary.
Final Thoughts
Dermatologists should be vigilant for unexpected payment denials, which may coincide with the implementation of new MUE values. When such denials occur and MUE values are publicly available, dermatologists should consider filing an appeal if the relevant MUEs were associated with an MAI of 1 or 3. Overall, dermatologists should be aware that many MUEs that were formerly claim line edits (MAI of 1) have been recently transitioned to date of service edits (MAI of 3), which are more restrictive.
1. American Academy of Dermatology. Medicare’s expanded medically unlikely edits. https://www.aad.org/members
/practice-and-advocacy-resource-center/coding-resources
/derm-coding-consult-library/winter-2014/medicare-
s-expanded-medically-unlikely-edits. Published Winter 2014. Accessed August 6, 2015.
2. Centers for Medicare & Medicaid Services. Revised modification to the Medically Unlikely Edit (MUE) program. MLN Matters. Number MM8853. https://www.cms.gov
/Outreach-and-Education/Medicare-Learning-Network-MLN/MLNMattersArticles/downloads/MM8853.pdf. Published January 1, 2015. Accessed August 6, 2015.
3. Centers for Medicare & Medicaid Services.
Medically Unlikely Edits (MUE) and bilateral procedures. MLN Matters. Number SE1422.
https://www.cms.gov/Regulations-and-Guidance
/Guidance/Transmittals/2014-Transmittals-Items/SE1422.html?DLPage=2&DLEntries=10&DLSort=1&DLSort
Dir=ascending. Accessed July 28, 2015.
1. American Academy of Dermatology. Medicare’s expanded medically unlikely edits. https://www.aad.org/members
/practice-and-advocacy-resource-center/coding-resources
/derm-coding-consult-library/winter-2014/medicare-
s-expanded-medically-unlikely-edits. Published Winter 2014. Accessed August 6, 2015.
2. Centers for Medicare & Medicaid Services. Revised modification to the Medically Unlikely Edit (MUE) program. MLN Matters. Number MM8853. https://www.cms.gov
/Outreach-and-Education/Medicare-Learning-Network-MLN/MLNMattersArticles/downloads/MM8853.pdf. Published January 1, 2015. Accessed August 6, 2015.
3. Centers for Medicare & Medicaid Services.
Medically Unlikely Edits (MUE) and bilateral procedures. MLN Matters. Number SE1422.
https://www.cms.gov/Regulations-and-Guidance
/Guidance/Transmittals/2014-Transmittals-Items/SE1422.html?DLPage=2&DLEntries=10&DLSort=1&DLSort
Dir=ascending. Accessed July 28, 2015.
Practice Points
- Medically Unlikely Edits (MUEs) are designed to prevent incorrect or excessive coding. Units of service billed in excess of the MUE will not be paid.
- Three different MUE adjudication indicators (MAIs) were added so that some MUE values would be date of service edits.
- Dermatologists should be vigilant for unexpected payment denials.
Electronic Brachytherapy: Overused and Overpriced?
The introduction of high-density radiation electronic brachytherapy (eBX) for the treatment of nonmelanoma skin cancers has induced great angst within the dermatology community.1 The Current Procedural Terminology (CPT) code 0182T (high dose rate eBX) reimburses at an extraordinarily high rate, which has drawn a substantial amount of attention. Some critics see it as another case of overutilization, of sucking more money out of a bleeding Medicare system. The financial opportunity afforded by eBX has even led some entrepreneurs to purchase dermatology clinics so that skin cancer patients can be treated via this modality instead of more traditional and less costly techniques (personal communication, 2014).
Among radiation oncologists, high-density radiation eBX is considered to be an important treatment option for select patients who have skin cancers staged as T1 or T2 tumors that are 4 cm or smaller in diameter and 5 mm or less in depth.2 Additionally, ideal candidates for nonsurgical treatment options such as eBX include patients with lesions in cosmetically challenging areas (eg, ears, nose), those who may experience problematic wound healing due to tumor location (eg, lower extremities) or medical conditions (eg, diabetes mellitus, peripheral vascular disease), those with medical comorbidities that may preclude them from surgery, those currently taking anticoagulants, and those who are not interested in undergoing surgery.
A common criticism of eBX is that there is little data on long-term treatment outcomes, which will soon be addressed by a 5-year multicenter, prospective, randomized study of 720 patients with basal cell carcinoma and squamous cell carcinoma led by the University of California, Irvine, and the University of California, San Diego (study protocol currently with institutional review board). Another criticism is that some manufacturers of eBX devices gained the less rigorous US Food and Drug Administration Premarket Notification 510(k) certification; however, this certification is quite commonplace in the United States, and an examination of the data actually shows a lower recall rate with this method when compared to the longer premarket approval application process.3 A more important criticism of eBX might be that radiation therapy is associated with a substantial increase in skin cancers that may occur decades later in irradiated areas; however, there remains a paucity of studies examining the safety data on eBX during the posttreatment period when such effects would be expected.
In practice, the forces for good and evil are not only limited to those who utilize eBX. It is widely known that CPT codes for Mohs micrographic surgery also have been abused—that is, the procedure has been used in circumstances where it was not absolutely necessary4—which led to an effort by dermatologic surgery organizations to agree on appropriate use criteria for Mohs surgery.5 These criteria are not perfect but should help curb the misuse of a valuable technique, which is one that is recognized as being optimal for the treatment of complex skin cancers. One might suggest forming similar appropriate use criteria for eBX and limiting this treatment to patients who either are older than 65 years, have serious medical issues, are currently taking anticoagulants, are immobile, or simply cannot handle further dermatologic surgeries.
The American Medical Association has developed new Category III CPT codes for treatment of the skin with eBX that will become effective January 2016.6 These codes take into consideration the need for a radiation oncologist and a physicist to be present for planning, dosimetry, simulation, and selection of parameters for the appropriate depth. Although I do not know the reimbursement rates for these new codes yet, they will likely be substantially less than the current payment for treatment with eBX. That said, the gravy train has left the station, and those who have invested in the devices for eBX will either see the benefit of continued treatment for their patients or divest themselves of eBX now that the reimbursement will be more modest.
Some of my dermatology colleagues, who also are some of my very good friends, have a visceral and absolute objection to the use of any form of radiation therapy, and I respect their opinions. However, eBX does play a role in treating cutaneous malignancies, and our radiation oncology colleagues—many who treat patients with extensive, aggressive, and recurrent skin cancers—also have a place at the table.
Speaking as a fellowship-trained dermatologic surgeon and a department chair, I am very aware that the teaching we provide today for our dermatology residents and fellows is likely to be their modus operandi for the future, a future in which the Patient Protection and Affordable Care Act will force physicians to carefully choose quality of care over personal gain and where financial rewards will be based on appropriate utilization and measurable outcomes. Electronic brachytherapy is one tool amongst many. I have a plethora of patients in their 70s and 80s who have given up on surgery for skin cancer and who would prefer painless treatment with eBX, which allows for the appropriate use of such a controversial therapy.
Acknowledgments—I would like to thank Janellen Smith, MD (Irvine, California), Joshua Spanogle, MD (Saint Augustine, Florida), and Jordan V. Wang, MBE (Philadelphia, Pennsylvania), for their constructive comments.
1. Linos E, VanBeek M, Resneck JS Jr. A sudden and concerning increase in the use of electronic brachytherapy for skin cancer. JAMA Dermatol. 2015;151:699-700.
2. Bhatnagar A. Nonmelanoma skin cancer treated with electronic brachytherapy: results at 1 year [published online ahead of print January 9, 2013]. Brachytherapy. 2013;12:134-140.
3. Connor JT, Lewis RJ, Berry DA, et al. FDA recalls not as alarming as they seem. Arch Intern Med. 2011;171:1044-1046.
4. Goldman G. Mohs surgery comes under the microscope. Member to Member American Academy of Dermatology E-newsletter. https://www.aad.org/members/publications /member-to-member/2013-archive/november-8-2013 /mohs-surgery-comes-under-the-microscope. Published November 8, 2013. Accessed August 10, 2015.
5. Ad Hoc Task Force, Connolly SM, Baker DR, et al. AAD/ACMS/ASDSA/ASMS 2012 appropriate use criteria for Mohs micrographic surgery: a report of the American Academy of Dermatology, American College of Mohs Surgery, American Society for Dermatologic Surgery Association, and the American Society for Mohs Surgery [published online ahead of print September 5, 2012]. J Am Acad Dermatol. 2012;67:531-550.
6. ACR Radiology Coding Source: CPT 2016 anticipated code changes. American College of Radiology Web site. http://www.acr.org/Advocacy/Economics-Health-Policy /Billing-Coding/Coding-Source-List/2015/Mar-Apr-2015 /CPT-2016-Anticipated-Code-Changes. Published March 2015. Accessed August 21, 2015.
The introduction of high-density radiation electronic brachytherapy (eBX) for the treatment of nonmelanoma skin cancers has induced great angst within the dermatology community.1 The Current Procedural Terminology (CPT) code 0182T (high dose rate eBX) reimburses at an extraordinarily high rate, which has drawn a substantial amount of attention. Some critics see it as another case of overutilization, of sucking more money out of a bleeding Medicare system. The financial opportunity afforded by eBX has even led some entrepreneurs to purchase dermatology clinics so that skin cancer patients can be treated via this modality instead of more traditional and less costly techniques (personal communication, 2014).
Among radiation oncologists, high-density radiation eBX is considered to be an important treatment option for select patients who have skin cancers staged as T1 or T2 tumors that are 4 cm or smaller in diameter and 5 mm or less in depth.2 Additionally, ideal candidates for nonsurgical treatment options such as eBX include patients with lesions in cosmetically challenging areas (eg, ears, nose), those who may experience problematic wound healing due to tumor location (eg, lower extremities) or medical conditions (eg, diabetes mellitus, peripheral vascular disease), those with medical comorbidities that may preclude them from surgery, those currently taking anticoagulants, and those who are not interested in undergoing surgery.
A common criticism of eBX is that there is little data on long-term treatment outcomes, which will soon be addressed by a 5-year multicenter, prospective, randomized study of 720 patients with basal cell carcinoma and squamous cell carcinoma led by the University of California, Irvine, and the University of California, San Diego (study protocol currently with institutional review board). Another criticism is that some manufacturers of eBX devices gained the less rigorous US Food and Drug Administration Premarket Notification 510(k) certification; however, this certification is quite commonplace in the United States, and an examination of the data actually shows a lower recall rate with this method when compared to the longer premarket approval application process.3 A more important criticism of eBX might be that radiation therapy is associated with a substantial increase in skin cancers that may occur decades later in irradiated areas; however, there remains a paucity of studies examining the safety data on eBX during the posttreatment period when such effects would be expected.
In practice, the forces for good and evil are not only limited to those who utilize eBX. It is widely known that CPT codes for Mohs micrographic surgery also have been abused—that is, the procedure has been used in circumstances where it was not absolutely necessary4—which led to an effort by dermatologic surgery organizations to agree on appropriate use criteria for Mohs surgery.5 These criteria are not perfect but should help curb the misuse of a valuable technique, which is one that is recognized as being optimal for the treatment of complex skin cancers. One might suggest forming similar appropriate use criteria for eBX and limiting this treatment to patients who either are older than 65 years, have serious medical issues, are currently taking anticoagulants, are immobile, or simply cannot handle further dermatologic surgeries.
The American Medical Association has developed new Category III CPT codes for treatment of the skin with eBX that will become effective January 2016.6 These codes take into consideration the need for a radiation oncologist and a physicist to be present for planning, dosimetry, simulation, and selection of parameters for the appropriate depth. Although I do not know the reimbursement rates for these new codes yet, they will likely be substantially less than the current payment for treatment with eBX. That said, the gravy train has left the station, and those who have invested in the devices for eBX will either see the benefit of continued treatment for their patients or divest themselves of eBX now that the reimbursement will be more modest.
Some of my dermatology colleagues, who also are some of my very good friends, have a visceral and absolute objection to the use of any form of radiation therapy, and I respect their opinions. However, eBX does play a role in treating cutaneous malignancies, and our radiation oncology colleagues—many who treat patients with extensive, aggressive, and recurrent skin cancers—also have a place at the table.
Speaking as a fellowship-trained dermatologic surgeon and a department chair, I am very aware that the teaching we provide today for our dermatology residents and fellows is likely to be their modus operandi for the future, a future in which the Patient Protection and Affordable Care Act will force physicians to carefully choose quality of care over personal gain and where financial rewards will be based on appropriate utilization and measurable outcomes. Electronic brachytherapy is one tool amongst many. I have a plethora of patients in their 70s and 80s who have given up on surgery for skin cancer and who would prefer painless treatment with eBX, which allows for the appropriate use of such a controversial therapy.
Acknowledgments—I would like to thank Janellen Smith, MD (Irvine, California), Joshua Spanogle, MD (Saint Augustine, Florida), and Jordan V. Wang, MBE (Philadelphia, Pennsylvania), for their constructive comments.
The introduction of high-density radiation electronic brachytherapy (eBX) for the treatment of nonmelanoma skin cancers has induced great angst within the dermatology community.1 The Current Procedural Terminology (CPT) code 0182T (high dose rate eBX) reimburses at an extraordinarily high rate, which has drawn a substantial amount of attention. Some critics see it as another case of overutilization, of sucking more money out of a bleeding Medicare system. The financial opportunity afforded by eBX has even led some entrepreneurs to purchase dermatology clinics so that skin cancer patients can be treated via this modality instead of more traditional and less costly techniques (personal communication, 2014).
Among radiation oncologists, high-density radiation eBX is considered to be an important treatment option for select patients who have skin cancers staged as T1 or T2 tumors that are 4 cm or smaller in diameter and 5 mm or less in depth.2 Additionally, ideal candidates for nonsurgical treatment options such as eBX include patients with lesions in cosmetically challenging areas (eg, ears, nose), those who may experience problematic wound healing due to tumor location (eg, lower extremities) or medical conditions (eg, diabetes mellitus, peripheral vascular disease), those with medical comorbidities that may preclude them from surgery, those currently taking anticoagulants, and those who are not interested in undergoing surgery.
A common criticism of eBX is that there is little data on long-term treatment outcomes, which will soon be addressed by a 5-year multicenter, prospective, randomized study of 720 patients with basal cell carcinoma and squamous cell carcinoma led by the University of California, Irvine, and the University of California, San Diego (study protocol currently with institutional review board). Another criticism is that some manufacturers of eBX devices gained the less rigorous US Food and Drug Administration Premarket Notification 510(k) certification; however, this certification is quite commonplace in the United States, and an examination of the data actually shows a lower recall rate with this method when compared to the longer premarket approval application process.3 A more important criticism of eBX might be that radiation therapy is associated with a substantial increase in skin cancers that may occur decades later in irradiated areas; however, there remains a paucity of studies examining the safety data on eBX during the posttreatment period when such effects would be expected.
In practice, the forces for good and evil are not only limited to those who utilize eBX. It is widely known that CPT codes for Mohs micrographic surgery also have been abused—that is, the procedure has been used in circumstances where it was not absolutely necessary4—which led to an effort by dermatologic surgery organizations to agree on appropriate use criteria for Mohs surgery.5 These criteria are not perfect but should help curb the misuse of a valuable technique, which is one that is recognized as being optimal for the treatment of complex skin cancers. One might suggest forming similar appropriate use criteria for eBX and limiting this treatment to patients who either are older than 65 years, have serious medical issues, are currently taking anticoagulants, are immobile, or simply cannot handle further dermatologic surgeries.
The American Medical Association has developed new Category III CPT codes for treatment of the skin with eBX that will become effective January 2016.6 These codes take into consideration the need for a radiation oncologist and a physicist to be present for planning, dosimetry, simulation, and selection of parameters for the appropriate depth. Although I do not know the reimbursement rates for these new codes yet, they will likely be substantially less than the current payment for treatment with eBX. That said, the gravy train has left the station, and those who have invested in the devices for eBX will either see the benefit of continued treatment for their patients or divest themselves of eBX now that the reimbursement will be more modest.
Some of my dermatology colleagues, who also are some of my very good friends, have a visceral and absolute objection to the use of any form of radiation therapy, and I respect their opinions. However, eBX does play a role in treating cutaneous malignancies, and our radiation oncology colleagues—many who treat patients with extensive, aggressive, and recurrent skin cancers—also have a place at the table.
Speaking as a fellowship-trained dermatologic surgeon and a department chair, I am very aware that the teaching we provide today for our dermatology residents and fellows is likely to be their modus operandi for the future, a future in which the Patient Protection and Affordable Care Act will force physicians to carefully choose quality of care over personal gain and where financial rewards will be based on appropriate utilization and measurable outcomes. Electronic brachytherapy is one tool amongst many. I have a plethora of patients in their 70s and 80s who have given up on surgery for skin cancer and who would prefer painless treatment with eBX, which allows for the appropriate use of such a controversial therapy.
Acknowledgments—I would like to thank Janellen Smith, MD (Irvine, California), Joshua Spanogle, MD (Saint Augustine, Florida), and Jordan V. Wang, MBE (Philadelphia, Pennsylvania), for their constructive comments.
1. Linos E, VanBeek M, Resneck JS Jr. A sudden and concerning increase in the use of electronic brachytherapy for skin cancer. JAMA Dermatol. 2015;151:699-700.
2. Bhatnagar A. Nonmelanoma skin cancer treated with electronic brachytherapy: results at 1 year [published online ahead of print January 9, 2013]. Brachytherapy. 2013;12:134-140.
3. Connor JT, Lewis RJ, Berry DA, et al. FDA recalls not as alarming as they seem. Arch Intern Med. 2011;171:1044-1046.
4. Goldman G. Mohs surgery comes under the microscope. Member to Member American Academy of Dermatology E-newsletter. https://www.aad.org/members/publications /member-to-member/2013-archive/november-8-2013 /mohs-surgery-comes-under-the-microscope. Published November 8, 2013. Accessed August 10, 2015.
5. Ad Hoc Task Force, Connolly SM, Baker DR, et al. AAD/ACMS/ASDSA/ASMS 2012 appropriate use criteria for Mohs micrographic surgery: a report of the American Academy of Dermatology, American College of Mohs Surgery, American Society for Dermatologic Surgery Association, and the American Society for Mohs Surgery [published online ahead of print September 5, 2012]. J Am Acad Dermatol. 2012;67:531-550.
6. ACR Radiology Coding Source: CPT 2016 anticipated code changes. American College of Radiology Web site. http://www.acr.org/Advocacy/Economics-Health-Policy /Billing-Coding/Coding-Source-List/2015/Mar-Apr-2015 /CPT-2016-Anticipated-Code-Changes. Published March 2015. Accessed August 21, 2015.
1. Linos E, VanBeek M, Resneck JS Jr. A sudden and concerning increase in the use of electronic brachytherapy for skin cancer. JAMA Dermatol. 2015;151:699-700.
2. Bhatnagar A. Nonmelanoma skin cancer treated with electronic brachytherapy: results at 1 year [published online ahead of print January 9, 2013]. Brachytherapy. 2013;12:134-140.
3. Connor JT, Lewis RJ, Berry DA, et al. FDA recalls not as alarming as they seem. Arch Intern Med. 2011;171:1044-1046.
4. Goldman G. Mohs surgery comes under the microscope. Member to Member American Academy of Dermatology E-newsletter. https://www.aad.org/members/publications /member-to-member/2013-archive/november-8-2013 /mohs-surgery-comes-under-the-microscope. Published November 8, 2013. Accessed August 10, 2015.
5. Ad Hoc Task Force, Connolly SM, Baker DR, et al. AAD/ACMS/ASDSA/ASMS 2012 appropriate use criteria for Mohs micrographic surgery: a report of the American Academy of Dermatology, American College of Mohs Surgery, American Society for Dermatologic Surgery Association, and the American Society for Mohs Surgery [published online ahead of print September 5, 2012]. J Am Acad Dermatol. 2012;67:531-550.
6. ACR Radiology Coding Source: CPT 2016 anticipated code changes. American College of Radiology Web site. http://www.acr.org/Advocacy/Economics-Health-Policy /Billing-Coding/Coding-Source-List/2015/Mar-Apr-2015 /CPT-2016-Anticipated-Code-Changes. Published March 2015. Accessed August 21, 2015.
Sept. JVS: Vascular surgeons do higher percentage of AAA repairs
The impact of endovascular repair on specialties performing abdominal aortic aneurysm repair.
Klaas H. J. Ultee, BSc,Rob Hurks, MD, PhD, Dominique B. Buck, MD, George S. DaSilva, BS, Peter A. Soden, MD,Joost A. van Herwaarden, MD, PhD, Hence J. M. Verhagen, MD, PhD, and Marc L. Schermerhorn, MD
Due to the increased use of EVAR for both intact and ruptured AAA repair, vascular surgeons are performing an increasing majority of AAA repairs, according to a new study reported in the September edition of Journal of Vascular Surgery.
The study examined the years 2001 through 2009 using the Nationwide Inpatient Sample, the largest national administrative database, which is maintained by the Agency for Healthcare Research and Quality as part of the Healthcare Cost and Utilization Project.
After 2009 the surgeon identification variables in the database were discontinued so more recent data were unavailable for the study.
“We do plan to analyze (this same subject) using Medicare data,” according to Dr. Marc L. Schermerhorn, “but our access to it lags several years behind. It will allow better risk adjustment as well.”
The study was interested in AAA repairs by the following types of physicians: vascular surgeons, general surgeons, cardiac surgeons, as well as nonsurgical specialists such as interventional cardiologists and interventional radiologists.
Overall, 108,587 EVARS and 85,080 open AAA repairs were identified. Of all repairs, 61 percent were performed by vascular surgeons, 20 percent by general surgeons, and 16 percent by cardiac surgeons. ICs and IRs performed the remaining 3 percent.
Significantly, the absolute number of vascular surgeons performing AAA repair increased 30 percent during the study period, whereas the number of GS and CS repairs decreased 46 and 30 percent, respectively.
AAA repairs are still done by general surgeons and cardiovascular surgeons; however, in those cases, patients are less likely to receive EVAR.
Researchers also found that whether patients received open or endovascular repair varied with the type of surgeon, but also by the patient’s gender, emergent admission, and race.
Other influencing factors were age of patient, treatment in a teaching hospital, year, and whether or not the hospital was in an urban area.
“The big question,” Schermerhorn noted, “is whether specialty has an influence on outcomes. We chose not to try to analyze this using this database because we did not think we could adequately do risk adjustment. It is difficult to distinguish a pre-existing condition from a post-op complication, for example, renal failure.”
The impact of endovascular repair on specialties performing abdominal aortic aneurysm repair.
Klaas H. J. Ultee, BSc,Rob Hurks, MD, PhD, Dominique B. Buck, MD, George S. DaSilva, BS, Peter A. Soden, MD,Joost A. van Herwaarden, MD, PhD, Hence J. M. Verhagen, MD, PhD, and Marc L. Schermerhorn, MD
Due to the increased use of EVAR for both intact and ruptured AAA repair, vascular surgeons are performing an increasing majority of AAA repairs, according to a new study reported in the September edition of Journal of Vascular Surgery.
The study examined the years 2001 through 2009 using the Nationwide Inpatient Sample, the largest national administrative database, which is maintained by the Agency for Healthcare Research and Quality as part of the Healthcare Cost and Utilization Project.
After 2009 the surgeon identification variables in the database were discontinued so more recent data were unavailable for the study.
“We do plan to analyze (this same subject) using Medicare data,” according to Dr. Marc L. Schermerhorn, “but our access to it lags several years behind. It will allow better risk adjustment as well.”
The study was interested in AAA repairs by the following types of physicians: vascular surgeons, general surgeons, cardiac surgeons, as well as nonsurgical specialists such as interventional cardiologists and interventional radiologists.
Overall, 108,587 EVARS and 85,080 open AAA repairs were identified. Of all repairs, 61 percent were performed by vascular surgeons, 20 percent by general surgeons, and 16 percent by cardiac surgeons. ICs and IRs performed the remaining 3 percent.
Significantly, the absolute number of vascular surgeons performing AAA repair increased 30 percent during the study period, whereas the number of GS and CS repairs decreased 46 and 30 percent, respectively.
AAA repairs are still done by general surgeons and cardiovascular surgeons; however, in those cases, patients are less likely to receive EVAR.
Researchers also found that whether patients received open or endovascular repair varied with the type of surgeon, but also by the patient’s gender, emergent admission, and race.
Other influencing factors were age of patient, treatment in a teaching hospital, year, and whether or not the hospital was in an urban area.
“The big question,” Schermerhorn noted, “is whether specialty has an influence on outcomes. We chose not to try to analyze this using this database because we did not think we could adequately do risk adjustment. It is difficult to distinguish a pre-existing condition from a post-op complication, for example, renal failure.”
The impact of endovascular repair on specialties performing abdominal aortic aneurysm repair.
Klaas H. J. Ultee, BSc,Rob Hurks, MD, PhD, Dominique B. Buck, MD, George S. DaSilva, BS, Peter A. Soden, MD,Joost A. van Herwaarden, MD, PhD, Hence J. M. Verhagen, MD, PhD, and Marc L. Schermerhorn, MD
Due to the increased use of EVAR for both intact and ruptured AAA repair, vascular surgeons are performing an increasing majority of AAA repairs, according to a new study reported in the September edition of Journal of Vascular Surgery.
The study examined the years 2001 through 2009 using the Nationwide Inpatient Sample, the largest national administrative database, which is maintained by the Agency for Healthcare Research and Quality as part of the Healthcare Cost and Utilization Project.
After 2009 the surgeon identification variables in the database were discontinued so more recent data were unavailable for the study.
“We do plan to analyze (this same subject) using Medicare data,” according to Dr. Marc L. Schermerhorn, “but our access to it lags several years behind. It will allow better risk adjustment as well.”
The study was interested in AAA repairs by the following types of physicians: vascular surgeons, general surgeons, cardiac surgeons, as well as nonsurgical specialists such as interventional cardiologists and interventional radiologists.
Overall, 108,587 EVARS and 85,080 open AAA repairs were identified. Of all repairs, 61 percent were performed by vascular surgeons, 20 percent by general surgeons, and 16 percent by cardiac surgeons. ICs and IRs performed the remaining 3 percent.
Significantly, the absolute number of vascular surgeons performing AAA repair increased 30 percent during the study period, whereas the number of GS and CS repairs decreased 46 and 30 percent, respectively.
AAA repairs are still done by general surgeons and cardiovascular surgeons; however, in those cases, patients are less likely to receive EVAR.
Researchers also found that whether patients received open or endovascular repair varied with the type of surgeon, but also by the patient’s gender, emergent admission, and race.
Other influencing factors were age of patient, treatment in a teaching hospital, year, and whether or not the hospital was in an urban area.
“The big question,” Schermerhorn noted, “is whether specialty has an influence on outcomes. We chose not to try to analyze this using this database because we did not think we could adequately do risk adjustment. It is difficult to distinguish a pre-existing condition from a post-op complication, for example, renal failure.”
EMA recommends orphan designation for CAR T-cell therapy

The European Medicines Agency’s (EMA’s) Committee for Orphan Medicinal Products has adopted a positive opinion recommending that KTE-C19 receive orphan designation to treat primary mediastinal B-cell lymphoma (PMBCL) and mantle cell lymphoma.
KTE-C19 is an investigational chimeric antigen receptor (CAR) T-cell therapy designed to target CD19, a protein expressed on the cell surface of B-cell lymphomas and leukemias.
No other product candidate currently has orphan drug designation for the treatment of PMBCL in the European Union (EU).
KTE-C19 already has orphan drug designation to treat diffuse large B-cell lymphoma (DLBCL) in the US and the EU.
“We are conducting a phase 1/2 clinical trial of KTE-C19 in patients with refractory, aggressive non-Hodgkin lymphoma, including DLBCL and PMBCL, and plan to report initial topline results from the phase 1 portion of the trial later this year [at the ASH Annual Meeting],” said Arie Belldegrun, MD, Chairman, President, and Chief Executive Officer of Kite Pharmaceuticals, the company developing KTE-C19.
Trial results
Last year, researchers reported results with KTE-C19 in the Journal of Clinical Oncology. The study included 15 patients with advanced B-cell malignancies.
The patients received a conditioning regimen of cyclophosphamide and fludarabine, followed 1 day later by a single infusion of KTE-C19. The researchers noted that the conditioning regimen is known to be active against B-cell malignancies and could have made a direct contribution to patient responses.
Thirteen patients were evaluable for response. Eight patients achieved a complete response (CR), and 4 had a partial response (PR).
Of the 7 patients with chemotherapy-refractory DLBCL, 4 achieved a CR, 2 achieved a PR, and 1 had stable disease. Of the 4 patients with chronic lymphocytic leukemia, 3 had a CR, and 1 had a PR. Among the 2 patients with indolent lymphomas, 1 achieved a CR, and 1 had a PR.
KTE-C19 was associated with fever, low blood pressure, focal neurological deficits, and delirium. Toxicities largely occurred in the first 2 weeks after infusion.
All but 2 patients experienced grade 3/4 adverse events. Four patients had grade 3/4 hypotension.
All patients had elevations in serum interferon gamma and/or interleukin 6 around the time of peak toxicity, but most did not develop elevations in serum tumor necrosis factor.
Neurologic toxicities included confusion and obtundation, which have been reported in previous studies. However, 3 patients developed unexpected neurologic abnormalities.
About orphan designation
The EMA’s Committee for Orphan Medicinal Products adopts an opinion on the granting of orphan designation, and that opinion is submitted to the European Commission for endorsement.
In the EU, orphan designation is granted to therapies intended to treat a life-threatening or chronically debilitating condition that affects no more than 5 in 10,000 persons and where no satisfactory treatment is available.
Companies that obtain orphan designation for a drug benefit from a number of incentives, including protocol assistance, a type of scientific advice specific for designated orphan medicines, and 10 years of market exclusivity once the medicine is approved. Fee reductions are also available, depending on the status of the sponsor and the type of service required. ![]()

The European Medicines Agency’s (EMA’s) Committee for Orphan Medicinal Products has adopted a positive opinion recommending that KTE-C19 receive orphan designation to treat primary mediastinal B-cell lymphoma (PMBCL) and mantle cell lymphoma.
KTE-C19 is an investigational chimeric antigen receptor (CAR) T-cell therapy designed to target CD19, a protein expressed on the cell surface of B-cell lymphomas and leukemias.
No other product candidate currently has orphan drug designation for the treatment of PMBCL in the European Union (EU).
KTE-C19 already has orphan drug designation to treat diffuse large B-cell lymphoma (DLBCL) in the US and the EU.
“We are conducting a phase 1/2 clinical trial of KTE-C19 in patients with refractory, aggressive non-Hodgkin lymphoma, including DLBCL and PMBCL, and plan to report initial topline results from the phase 1 portion of the trial later this year [at the ASH Annual Meeting],” said Arie Belldegrun, MD, Chairman, President, and Chief Executive Officer of Kite Pharmaceuticals, the company developing KTE-C19.
Trial results
Last year, researchers reported results with KTE-C19 in the Journal of Clinical Oncology. The study included 15 patients with advanced B-cell malignancies.
The patients received a conditioning regimen of cyclophosphamide and fludarabine, followed 1 day later by a single infusion of KTE-C19. The researchers noted that the conditioning regimen is known to be active against B-cell malignancies and could have made a direct contribution to patient responses.
Thirteen patients were evaluable for response. Eight patients achieved a complete response (CR), and 4 had a partial response (PR).
Of the 7 patients with chemotherapy-refractory DLBCL, 4 achieved a CR, 2 achieved a PR, and 1 had stable disease. Of the 4 patients with chronic lymphocytic leukemia, 3 had a CR, and 1 had a PR. Among the 2 patients with indolent lymphomas, 1 achieved a CR, and 1 had a PR.
KTE-C19 was associated with fever, low blood pressure, focal neurological deficits, and delirium. Toxicities largely occurred in the first 2 weeks after infusion.
All but 2 patients experienced grade 3/4 adverse events. Four patients had grade 3/4 hypotension.
All patients had elevations in serum interferon gamma and/or interleukin 6 around the time of peak toxicity, but most did not develop elevations in serum tumor necrosis factor.
Neurologic toxicities included confusion and obtundation, which have been reported in previous studies. However, 3 patients developed unexpected neurologic abnormalities.
About orphan designation
The EMA’s Committee for Orphan Medicinal Products adopts an opinion on the granting of orphan designation, and that opinion is submitted to the European Commission for endorsement.
In the EU, orphan designation is granted to therapies intended to treat a life-threatening or chronically debilitating condition that affects no more than 5 in 10,000 persons and where no satisfactory treatment is available.
Companies that obtain orphan designation for a drug benefit from a number of incentives, including protocol assistance, a type of scientific advice specific for designated orphan medicines, and 10 years of market exclusivity once the medicine is approved. Fee reductions are also available, depending on the status of the sponsor and the type of service required. ![]()

The European Medicines Agency’s (EMA’s) Committee for Orphan Medicinal Products has adopted a positive opinion recommending that KTE-C19 receive orphan designation to treat primary mediastinal B-cell lymphoma (PMBCL) and mantle cell lymphoma.
KTE-C19 is an investigational chimeric antigen receptor (CAR) T-cell therapy designed to target CD19, a protein expressed on the cell surface of B-cell lymphomas and leukemias.
No other product candidate currently has orphan drug designation for the treatment of PMBCL in the European Union (EU).
KTE-C19 already has orphan drug designation to treat diffuse large B-cell lymphoma (DLBCL) in the US and the EU.
“We are conducting a phase 1/2 clinical trial of KTE-C19 in patients with refractory, aggressive non-Hodgkin lymphoma, including DLBCL and PMBCL, and plan to report initial topline results from the phase 1 portion of the trial later this year [at the ASH Annual Meeting],” said Arie Belldegrun, MD, Chairman, President, and Chief Executive Officer of Kite Pharmaceuticals, the company developing KTE-C19.
Trial results
Last year, researchers reported results with KTE-C19 in the Journal of Clinical Oncology. The study included 15 patients with advanced B-cell malignancies.
The patients received a conditioning regimen of cyclophosphamide and fludarabine, followed 1 day later by a single infusion of KTE-C19. The researchers noted that the conditioning regimen is known to be active against B-cell malignancies and could have made a direct contribution to patient responses.
Thirteen patients were evaluable for response. Eight patients achieved a complete response (CR), and 4 had a partial response (PR).
Of the 7 patients with chemotherapy-refractory DLBCL, 4 achieved a CR, 2 achieved a PR, and 1 had stable disease. Of the 4 patients with chronic lymphocytic leukemia, 3 had a CR, and 1 had a PR. Among the 2 patients with indolent lymphomas, 1 achieved a CR, and 1 had a PR.
KTE-C19 was associated with fever, low blood pressure, focal neurological deficits, and delirium. Toxicities largely occurred in the first 2 weeks after infusion.
All but 2 patients experienced grade 3/4 adverse events. Four patients had grade 3/4 hypotension.
All patients had elevations in serum interferon gamma and/or interleukin 6 around the time of peak toxicity, but most did not develop elevations in serum tumor necrosis factor.
Neurologic toxicities included confusion and obtundation, which have been reported in previous studies. However, 3 patients developed unexpected neurologic abnormalities.
About orphan designation
The EMA’s Committee for Orphan Medicinal Products adopts an opinion on the granting of orphan designation, and that opinion is submitted to the European Commission for endorsement.
In the EU, orphan designation is granted to therapies intended to treat a life-threatening or chronically debilitating condition that affects no more than 5 in 10,000 persons and where no satisfactory treatment is available.
Companies that obtain orphan designation for a drug benefit from a number of incentives, including protocol assistance, a type of scientific advice specific for designated orphan medicines, and 10 years of market exclusivity once the medicine is approved. Fee reductions are also available, depending on the status of the sponsor and the type of service required. ![]()
Tiering dermatologists without the benefit of true quality measures
Tiering is the “ranking” of physicians by insurance companies. These rankings are used to decide who gets to participate in the networks, how you get paid, the patient’s copay, and on and on. The rankings are also published. Insurance companies want to save money, and are attempting to do this under the guise of enhancing quality.
Fine, you say, I am an efficient dermatologist and ready to be ranked against anyone.
No, it is not fine, because there are no validated quality measures for dermatologists.
Well great, you say, then dermatologists can’t be ranked.
No, unfortunately, dermatologists are getting ranked anyway, and the process is little more than just making up a ranking.
Let me give you an example. Cigna has a two star system and ranks specialists according to “practice of evidence-based medicine” and “quality of care.” (See “How are specialists chosen for Cigna Care Designation” on Frequently Asked Questions on the Cigna web site). If there aren’t any quality measures for dermatologists, how can they do it? Well, they give the first star to a dermatologist if primary care doctors in their medical group check glycosylated hemoglobins and blood pressures.
Yes, some dermatologists get credit and a star for something that has nothing to do with them.
The second measure of quality is even more preposterous. Cigna uses cost-per-patient software, and the least expensive dermatologist gets the second star – no matter who or what they are treating, or what procedures they are performing.
This approach introduces multiple perversions into the system. First, the primary care doctors are under huge pressure to get their patients to comply with testing measures. Consequently, the systems they work for are insisting that they “fire” patients who do not come in for their checkups and get their blood checks.
Closer to home, dermatologists who do Mohs surgery full time, or who are in solo or small practices, or who prescribe expensive medications are penalized.
Cigna is one of six health insurers tiering dermatologists, but soon all insurers will be doing the same. Representatives from the American Academy of Dermatology, including myself, have met with Cigna and pointed out how meaningless it is to rank dermatologists without having specialty-specific quality parameters. The less-than-adequate response has been that “the lack of quality measures is a problem with several specialties.”
Given the lack of validated quality measures for dermatology, I find it bizarre that Health and Human Services Secretary Sylvia M. Burwell has set the goal of tying 85% of all traditional Medicare payments to quality or value by 2016 and 90% by 2018. I’m afraid this is going to be a very blunt axe resulting in splintered health care.
The AAD is doing its best to delay this deadline, at least until there are some relevant quality measures for dermatology, and has launched a major data collection initiative – DataDerm. Amassing that information should give us some decent benchmarks in a few years. DataDerm will ultimately provide benchmark reports, access to clinically relevant data, quality measurement, and information to improve patient care.
Until then we will argue, reason, and cajole as best we can. Meanwhile, the AAD will need your help with DataDerm, and it won’t do any good for you to stomp your feet and just say ‘no.’ In future columns, I will discuss the impacts of UnitedHealth Group’s misguided “lab benefit program” and the unfortunate Optum360 physician profiling software.
Dr. Coldiron is a past president of the American Academy of Dermatology. He is currently in private practice, but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. Reach him at dermnews@frontlinemedcom.com
Tiering is the “ranking” of physicians by insurance companies. These rankings are used to decide who gets to participate in the networks, how you get paid, the patient’s copay, and on and on. The rankings are also published. Insurance companies want to save money, and are attempting to do this under the guise of enhancing quality.
Fine, you say, I am an efficient dermatologist and ready to be ranked against anyone.
No, it is not fine, because there are no validated quality measures for dermatologists.
Well great, you say, then dermatologists can’t be ranked.
No, unfortunately, dermatologists are getting ranked anyway, and the process is little more than just making up a ranking.
Let me give you an example. Cigna has a two star system and ranks specialists according to “practice of evidence-based medicine” and “quality of care.” (See “How are specialists chosen for Cigna Care Designation” on Frequently Asked Questions on the Cigna web site). If there aren’t any quality measures for dermatologists, how can they do it? Well, they give the first star to a dermatologist if primary care doctors in their medical group check glycosylated hemoglobins and blood pressures.
Yes, some dermatologists get credit and a star for something that has nothing to do with them.
The second measure of quality is even more preposterous. Cigna uses cost-per-patient software, and the least expensive dermatologist gets the second star – no matter who or what they are treating, or what procedures they are performing.
This approach introduces multiple perversions into the system. First, the primary care doctors are under huge pressure to get their patients to comply with testing measures. Consequently, the systems they work for are insisting that they “fire” patients who do not come in for their checkups and get their blood checks.
Closer to home, dermatologists who do Mohs surgery full time, or who are in solo or small practices, or who prescribe expensive medications are penalized.
Cigna is one of six health insurers tiering dermatologists, but soon all insurers will be doing the same. Representatives from the American Academy of Dermatology, including myself, have met with Cigna and pointed out how meaningless it is to rank dermatologists without having specialty-specific quality parameters. The less-than-adequate response has been that “the lack of quality measures is a problem with several specialties.”
Given the lack of validated quality measures for dermatology, I find it bizarre that Health and Human Services Secretary Sylvia M. Burwell has set the goal of tying 85% of all traditional Medicare payments to quality or value by 2016 and 90% by 2018. I’m afraid this is going to be a very blunt axe resulting in splintered health care.
The AAD is doing its best to delay this deadline, at least until there are some relevant quality measures for dermatology, and has launched a major data collection initiative – DataDerm. Amassing that information should give us some decent benchmarks in a few years. DataDerm will ultimately provide benchmark reports, access to clinically relevant data, quality measurement, and information to improve patient care.
Until then we will argue, reason, and cajole as best we can. Meanwhile, the AAD will need your help with DataDerm, and it won’t do any good for you to stomp your feet and just say ‘no.’ In future columns, I will discuss the impacts of UnitedHealth Group’s misguided “lab benefit program” and the unfortunate Optum360 physician profiling software.
Dr. Coldiron is a past president of the American Academy of Dermatology. He is currently in private practice, but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. Reach him at dermnews@frontlinemedcom.com
Tiering is the “ranking” of physicians by insurance companies. These rankings are used to decide who gets to participate in the networks, how you get paid, the patient’s copay, and on and on. The rankings are also published. Insurance companies want to save money, and are attempting to do this under the guise of enhancing quality.
Fine, you say, I am an efficient dermatologist and ready to be ranked against anyone.
No, it is not fine, because there are no validated quality measures for dermatologists.
Well great, you say, then dermatologists can’t be ranked.
No, unfortunately, dermatologists are getting ranked anyway, and the process is little more than just making up a ranking.
Let me give you an example. Cigna has a two star system and ranks specialists according to “practice of evidence-based medicine” and “quality of care.” (See “How are specialists chosen for Cigna Care Designation” on Frequently Asked Questions on the Cigna web site). If there aren’t any quality measures for dermatologists, how can they do it? Well, they give the first star to a dermatologist if primary care doctors in their medical group check glycosylated hemoglobins and blood pressures.
Yes, some dermatologists get credit and a star for something that has nothing to do with them.
The second measure of quality is even more preposterous. Cigna uses cost-per-patient software, and the least expensive dermatologist gets the second star – no matter who or what they are treating, or what procedures they are performing.
This approach introduces multiple perversions into the system. First, the primary care doctors are under huge pressure to get their patients to comply with testing measures. Consequently, the systems they work for are insisting that they “fire” patients who do not come in for their checkups and get their blood checks.
Closer to home, dermatologists who do Mohs surgery full time, or who are in solo or small practices, or who prescribe expensive medications are penalized.
Cigna is one of six health insurers tiering dermatologists, but soon all insurers will be doing the same. Representatives from the American Academy of Dermatology, including myself, have met with Cigna and pointed out how meaningless it is to rank dermatologists without having specialty-specific quality parameters. The less-than-adequate response has been that “the lack of quality measures is a problem with several specialties.”
Given the lack of validated quality measures for dermatology, I find it bizarre that Health and Human Services Secretary Sylvia M. Burwell has set the goal of tying 85% of all traditional Medicare payments to quality or value by 2016 and 90% by 2018. I’m afraid this is going to be a very blunt axe resulting in splintered health care.
The AAD is doing its best to delay this deadline, at least until there are some relevant quality measures for dermatology, and has launched a major data collection initiative – DataDerm. Amassing that information should give us some decent benchmarks in a few years. DataDerm will ultimately provide benchmark reports, access to clinically relevant data, quality measurement, and information to improve patient care.
Until then we will argue, reason, and cajole as best we can. Meanwhile, the AAD will need your help with DataDerm, and it won’t do any good for you to stomp your feet and just say ‘no.’ In future columns, I will discuss the impacts of UnitedHealth Group’s misguided “lab benefit program” and the unfortunate Optum360 physician profiling software.
Dr. Coldiron is a past president of the American Academy of Dermatology. He is currently in private practice, but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. Reach him at dermnews@frontlinemedcom.com
HIT risk rises with obesity
LAS VEGAS – High body mass index is strongly associated with increased rates of heparin-induced thrombocytopenia, based on findings from a review of prospectively collected data from surgical and cardiac intensive care unit patients presumed to have the condition.
Of 304 patients included in the review, 36 (12%) were positive for heparin-induced thrombocytopenia (HIT). The rates increased in tandem with BMI. For example, the rate was 0% among 9 underweight individuals (BMI less than 18.5 kg/m2), 8% among 119 normal-weight individuals (BMI of 18.5-24.9 kg/m2), 11% among 98 overweight individuals (BMI of 25-29.9 kg/m2), 18% among 67 obese individuals (BMI of 30-39.9 kg/m2), and 36% among 11 morbidly obese individuals (BMI of 40 kg/m2 or greater), Dr. Matthew B. Bloom reported at the annual meeting of the American Association for the Surgery of Trauma.
The odds of HIT were 170% greater among obese patients, compared with normal-weight patients (odds ratio, 2.67), and 600% greater among morbidly obese patients, compared with normal-weight patient (odds ratio, 6.98), said Dr. Bloom of Cedars-Sinai Medical Center, Los Angeles.
Logistic regression showed that each 1 unit increase in BMI was associated with a 7.7% increase in the odds of developing HIT, he noted.
Additionally, an anti-heparin/PF4 (platelet factor 4) antibody OD (optical density) value of 2.0 or greater, but not of 0.4 or greater or 0.8 or greater, was also significantly increased with BMI, and in-hospital mortality increased significantly with BMI above normal, he said.
Warkentin 4T scores used to differentiate HIT from other types of thrombocytopenia were not found to correlate with changes in BMI in this study, nor were deep vein thrombosis, pulmonary embolism, or stroke.
The increase in PF4 with increasing BMI may be a marker for overall increasing levels of circulating antibodies in the obese ICU population, but more biochemical studies are needed to tease this out, he said.
Patients included in the review were all those admitted to the surgical and cardiac ICUs at Cedars-Sinai over a more than 7 year period. They had a mean age of 62.1 years, 59% were men, and their mean BMI was 27 kg/m2.
The findings are among the first to show a strong association between BMI and HIT in ICU patients, Dr. Bloom said, noting that several other studies have shown that obesity is linked with increased incidence and increased severity of immune-mediated diseases, including rheumatoid arthritis, systemic lupus erythematosus, and inflammatory bowel disease.
“And HIT is an immune-mediated disease,” he added.
“BMI may be an important new clinical variable for estimating the pre-test probability of HIT, and perhaps, in the future, patient ‘thickness’ could be considered a new ‘T’ in the 4T score, he concluded.
Dr. Bloom reported having no disclosures.
LAS VEGAS – High body mass index is strongly associated with increased rates of heparin-induced thrombocytopenia, based on findings from a review of prospectively collected data from surgical and cardiac intensive care unit patients presumed to have the condition.
Of 304 patients included in the review, 36 (12%) were positive for heparin-induced thrombocytopenia (HIT). The rates increased in tandem with BMI. For example, the rate was 0% among 9 underweight individuals (BMI less than 18.5 kg/m2), 8% among 119 normal-weight individuals (BMI of 18.5-24.9 kg/m2), 11% among 98 overweight individuals (BMI of 25-29.9 kg/m2), 18% among 67 obese individuals (BMI of 30-39.9 kg/m2), and 36% among 11 morbidly obese individuals (BMI of 40 kg/m2 or greater), Dr. Matthew B. Bloom reported at the annual meeting of the American Association for the Surgery of Trauma.
The odds of HIT were 170% greater among obese patients, compared with normal-weight patients (odds ratio, 2.67), and 600% greater among morbidly obese patients, compared with normal-weight patient (odds ratio, 6.98), said Dr. Bloom of Cedars-Sinai Medical Center, Los Angeles.
Logistic regression showed that each 1 unit increase in BMI was associated with a 7.7% increase in the odds of developing HIT, he noted.
Additionally, an anti-heparin/PF4 (platelet factor 4) antibody OD (optical density) value of 2.0 or greater, but not of 0.4 or greater or 0.8 or greater, was also significantly increased with BMI, and in-hospital mortality increased significantly with BMI above normal, he said.
Warkentin 4T scores used to differentiate HIT from other types of thrombocytopenia were not found to correlate with changes in BMI in this study, nor were deep vein thrombosis, pulmonary embolism, or stroke.
The increase in PF4 with increasing BMI may be a marker for overall increasing levels of circulating antibodies in the obese ICU population, but more biochemical studies are needed to tease this out, he said.
Patients included in the review were all those admitted to the surgical and cardiac ICUs at Cedars-Sinai over a more than 7 year period. They had a mean age of 62.1 years, 59% were men, and their mean BMI was 27 kg/m2.
The findings are among the first to show a strong association between BMI and HIT in ICU patients, Dr. Bloom said, noting that several other studies have shown that obesity is linked with increased incidence and increased severity of immune-mediated diseases, including rheumatoid arthritis, systemic lupus erythematosus, and inflammatory bowel disease.
“And HIT is an immune-mediated disease,” he added.
“BMI may be an important new clinical variable for estimating the pre-test probability of HIT, and perhaps, in the future, patient ‘thickness’ could be considered a new ‘T’ in the 4T score, he concluded.
Dr. Bloom reported having no disclosures.
LAS VEGAS – High body mass index is strongly associated with increased rates of heparin-induced thrombocytopenia, based on findings from a review of prospectively collected data from surgical and cardiac intensive care unit patients presumed to have the condition.
Of 304 patients included in the review, 36 (12%) were positive for heparin-induced thrombocytopenia (HIT). The rates increased in tandem with BMI. For example, the rate was 0% among 9 underweight individuals (BMI less than 18.5 kg/m2), 8% among 119 normal-weight individuals (BMI of 18.5-24.9 kg/m2), 11% among 98 overweight individuals (BMI of 25-29.9 kg/m2), 18% among 67 obese individuals (BMI of 30-39.9 kg/m2), and 36% among 11 morbidly obese individuals (BMI of 40 kg/m2 or greater), Dr. Matthew B. Bloom reported at the annual meeting of the American Association for the Surgery of Trauma.
The odds of HIT were 170% greater among obese patients, compared with normal-weight patients (odds ratio, 2.67), and 600% greater among morbidly obese patients, compared with normal-weight patient (odds ratio, 6.98), said Dr. Bloom of Cedars-Sinai Medical Center, Los Angeles.
Logistic regression showed that each 1 unit increase in BMI was associated with a 7.7% increase in the odds of developing HIT, he noted.
Additionally, an anti-heparin/PF4 (platelet factor 4) antibody OD (optical density) value of 2.0 or greater, but not of 0.4 or greater or 0.8 or greater, was also significantly increased with BMI, and in-hospital mortality increased significantly with BMI above normal, he said.
Warkentin 4T scores used to differentiate HIT from other types of thrombocytopenia were not found to correlate with changes in BMI in this study, nor were deep vein thrombosis, pulmonary embolism, or stroke.
The increase in PF4 with increasing BMI may be a marker for overall increasing levels of circulating antibodies in the obese ICU population, but more biochemical studies are needed to tease this out, he said.
Patients included in the review were all those admitted to the surgical and cardiac ICUs at Cedars-Sinai over a more than 7 year period. They had a mean age of 62.1 years, 59% were men, and their mean BMI was 27 kg/m2.
The findings are among the first to show a strong association between BMI and HIT in ICU patients, Dr. Bloom said, noting that several other studies have shown that obesity is linked with increased incidence and increased severity of immune-mediated diseases, including rheumatoid arthritis, systemic lupus erythematosus, and inflammatory bowel disease.
“And HIT is an immune-mediated disease,” he added.
“BMI may be an important new clinical variable for estimating the pre-test probability of HIT, and perhaps, in the future, patient ‘thickness’ could be considered a new ‘T’ in the 4T score, he concluded.
Dr. Bloom reported having no disclosures.
AT THE AAST ANNUAL MEETING
Key clinical point: Higher body mass index is strongly associated with increased rates of heparin-induced thrombocytopenia.
Major finding: The HIT rate was 0%, 8%, 11%, 18,%, and 36% among underweight, normal-weight, overweight, obese, and morbidly obese individuals, respectively.
Data source: A review of prospectively collected data for 304 patients.
Disclosures: Dr. Bloom reported having no disclosures.
FDA grants drug orphan designation for hemophilia B

The US Food and Drug Administration (FDA) has granted orphan designation for DTX101 as a treatment for hemophilia B.
DTX101 is designed to deliver factor IX gene expression in a durable fashion to prevent the long-term complications of hemophilia B.
Preclinical studies have indicated that DTX101 has the potential to be a well-tolerated, effective therapy for hemophilia B, according to Dimension Therapeutics, Inc., the company developing DTX101.
The company said it expects to initiate a multicenter, phase 1/2 study to evaluate DTX101 in adults with moderate/severe to severe hemophilia B by the end of this year.
About orphan designation
The FDA grants orphan designation to drugs and biologics intended to treat rare diseases or conditions that affect fewer than 200,000 people in the US.
Orphan designation provides the company developing a drug with certain benefits, such as tax credits for qualified clinical trials, exemption from FDA user fees, and 7 years of market exclusivity if the drug is approved. ![]()

The US Food and Drug Administration (FDA) has granted orphan designation for DTX101 as a treatment for hemophilia B.
DTX101 is designed to deliver factor IX gene expression in a durable fashion to prevent the long-term complications of hemophilia B.
Preclinical studies have indicated that DTX101 has the potential to be a well-tolerated, effective therapy for hemophilia B, according to Dimension Therapeutics, Inc., the company developing DTX101.
The company said it expects to initiate a multicenter, phase 1/2 study to evaluate DTX101 in adults with moderate/severe to severe hemophilia B by the end of this year.
About orphan designation
The FDA grants orphan designation to drugs and biologics intended to treat rare diseases or conditions that affect fewer than 200,000 people in the US.
Orphan designation provides the company developing a drug with certain benefits, such as tax credits for qualified clinical trials, exemption from FDA user fees, and 7 years of market exclusivity if the drug is approved. ![]()

The US Food and Drug Administration (FDA) has granted orphan designation for DTX101 as a treatment for hemophilia B.
DTX101 is designed to deliver factor IX gene expression in a durable fashion to prevent the long-term complications of hemophilia B.
Preclinical studies have indicated that DTX101 has the potential to be a well-tolerated, effective therapy for hemophilia B, according to Dimension Therapeutics, Inc., the company developing DTX101.
The company said it expects to initiate a multicenter, phase 1/2 study to evaluate DTX101 in adults with moderate/severe to severe hemophilia B by the end of this year.
About orphan designation
The FDA grants orphan designation to drugs and biologics intended to treat rare diseases or conditions that affect fewer than 200,000 people in the US.
Orphan designation provides the company developing a drug with certain benefits, such as tax credits for qualified clinical trials, exemption from FDA user fees, and 7 years of market exclusivity if the drug is approved. ![]()
Troponin Test Can Predict Mortality Risk in Cardiovascular Patients
NEW YORK — An experimental, highly sensitive troponin test cannot predict when type 2 diabetics with stable ischemic heart disease will benefit from prompt coronary revascularization, but it can show which patients are more likely to die from myocardial infarction, stroke, or other cardiovascular cases, according to a new study.
Because the troponin test measures damage to heart muscle, the outcome "suggests there's ongoing injury to patients with stable heart disease and diabetes. It has a strong association with death, heart attack, stroke, heart failure," the chief author, Dr. Brendan Everett, director of inpatient general cardiology at Brigham and Women's Hospital in Boston, told Reuters Health in a telephone interview.
But when it comes to heading off a higher risk, "we need to do more research to understand where it's coming from and what therapies might be appropriate. So it does not appear in this population that opening the coronary arteries offers any long-term benefit with respect to death or heart attack," he said.
"It tells you this is not the type of disease status that can be managed by bypass or stenting," said Dr. David Zhao, chairman of cardiology at Wake Forest Baptist Medical Center in Winston-Salem, North Carolina. He was not involved in the research.
"From the patient's standpoint, it is disappointing," Dr. Zhao said. "But from a science standpoint, it all comes together nicely. These patients tend to have more small-vessel microvascular disease and the constant elevation of troponin indicates that there is more severe microvascular disease and more endomyocardial injury. I think it gives us the opportunity to look at more intensified medical management in this group of patients."
The study was an offshoot of the BARI 2D trial, conducted at 49 sites in six countries. It found that aggressive revascularization didn't reduce the risk of death or cardiovascular outcomes compared to intensive medical therapy alone in type 2 diabetics with stable heart disease.
In the new research, the team went back and used a highly sensitive troponin test sold in Europe but not available in the United States to see "if it could be used in stable patients to identify those who would benefit from having their coronary arteries opened with either angioplasty and stenting or bypass surgery," Dr. Everett said.
"There aren't many studies addressing that question and luckily BARI 2D had samples that were stored and available to us after the completion of the trial," he said. "What we did find was that there's clearly a group of patients that are at very high risk for death, heart attack, heart failure, and stroke."
The researchers found 27.1% of patients with a troponin T level of 14.0 ng/L or higher died from cardiovascular cause, or had a heart attack or stroke, compared to 12.9% of patients whose levels were lower, they reported online August 12 in the New England Journal of Medicine.
Adjusting for various factors, the risk of reaching one of those primary endpoints after five years was 85% greater with the higher troponin levels (P<0.001).
The risks for reaching individual components of the composite endpoint were also higher. Unadjusted five-year rates for patients with higher versus lower troponin levels were, respectively, 10.9% versus 3.5% for cardiovascular death, 18.7% versus 9.2% for myocardial infarction, 4.4% versus 2.3% for stroke, and 25.7% versus 11.1% for heart failure.
The Everett team also found that among the 897 volunteers whose levels had been at 14 or above, receiving a prompt intervention did not reduce their likelihood of reaching a primary endpoint compared to the 2,277 with normal concentrations.
The five-year rates of heart attack, stroke, or death from any cardiovascular cause were 26.5% in the revascularization group and 27.6% with medical therapy alone.
"We need to figure out how to better treat them, because fixing their coronary arteries doesn't seem to be giving them the benefit we thought it would, or hoped it would," said Dr. Everett. "For the time being, this is not a test that should be routinely used on stable patients."
The researchers also looked at how troponin levels changed over time.
"Despite aggressive medical therapy for type 2 diabetes and stable ischemic heart disease, the median troponin T concentration increased over one year of follow-up, and no significant reductions in troponin T concentrations were observed in patients who underwent coronary revascularization," the researchers said.
"If this type of patient has elevated troponin, I would be more aggressive in medical management, including statins, smoking cessation, and lifestyle changes," said Dr. Zhao of Wake Forest. "I think the next step would be to ideally run a clinical trial and see if, in patients with elevated troponin levels, intensified medical management reduces the number of events as compared to standard therapy.
NEW YORK — An experimental, highly sensitive troponin test cannot predict when type 2 diabetics with stable ischemic heart disease will benefit from prompt coronary revascularization, but it can show which patients are more likely to die from myocardial infarction, stroke, or other cardiovascular cases, according to a new study.
Because the troponin test measures damage to heart muscle, the outcome "suggests there's ongoing injury to patients with stable heart disease and diabetes. It has a strong association with death, heart attack, stroke, heart failure," the chief author, Dr. Brendan Everett, director of inpatient general cardiology at Brigham and Women's Hospital in Boston, told Reuters Health in a telephone interview.
But when it comes to heading off a higher risk, "we need to do more research to understand where it's coming from and what therapies might be appropriate. So it does not appear in this population that opening the coronary arteries offers any long-term benefit with respect to death or heart attack," he said.
"It tells you this is not the type of disease status that can be managed by bypass or stenting," said Dr. David Zhao, chairman of cardiology at Wake Forest Baptist Medical Center in Winston-Salem, North Carolina. He was not involved in the research.
"From the patient's standpoint, it is disappointing," Dr. Zhao said. "But from a science standpoint, it all comes together nicely. These patients tend to have more small-vessel microvascular disease and the constant elevation of troponin indicates that there is more severe microvascular disease and more endomyocardial injury. I think it gives us the opportunity to look at more intensified medical management in this group of patients."
The study was an offshoot of the BARI 2D trial, conducted at 49 sites in six countries. It found that aggressive revascularization didn't reduce the risk of death or cardiovascular outcomes compared to intensive medical therapy alone in type 2 diabetics with stable heart disease.
In the new research, the team went back and used a highly sensitive troponin test sold in Europe but not available in the United States to see "if it could be used in stable patients to identify those who would benefit from having their coronary arteries opened with either angioplasty and stenting or bypass surgery," Dr. Everett said.
"There aren't many studies addressing that question and luckily BARI 2D had samples that were stored and available to us after the completion of the trial," he said. "What we did find was that there's clearly a group of patients that are at very high risk for death, heart attack, heart failure, and stroke."
The researchers found 27.1% of patients with a troponin T level of 14.0 ng/L or higher died from cardiovascular cause, or had a heart attack or stroke, compared to 12.9% of patients whose levels were lower, they reported online August 12 in the New England Journal of Medicine.
Adjusting for various factors, the risk of reaching one of those primary endpoints after five years was 85% greater with the higher troponin levels (P<0.001).
The risks for reaching individual components of the composite endpoint were also higher. Unadjusted five-year rates for patients with higher versus lower troponin levels were, respectively, 10.9% versus 3.5% for cardiovascular death, 18.7% versus 9.2% for myocardial infarction, 4.4% versus 2.3% for stroke, and 25.7% versus 11.1% for heart failure.
The Everett team also found that among the 897 volunteers whose levels had been at 14 or above, receiving a prompt intervention did not reduce their likelihood of reaching a primary endpoint compared to the 2,277 with normal concentrations.
The five-year rates of heart attack, stroke, or death from any cardiovascular cause were 26.5% in the revascularization group and 27.6% with medical therapy alone.
"We need to figure out how to better treat them, because fixing their coronary arteries doesn't seem to be giving them the benefit we thought it would, or hoped it would," said Dr. Everett. "For the time being, this is not a test that should be routinely used on stable patients."
The researchers also looked at how troponin levels changed over time.
"Despite aggressive medical therapy for type 2 diabetes and stable ischemic heart disease, the median troponin T concentration increased over one year of follow-up, and no significant reductions in troponin T concentrations were observed in patients who underwent coronary revascularization," the researchers said.
"If this type of patient has elevated troponin, I would be more aggressive in medical management, including statins, smoking cessation, and lifestyle changes," said Dr. Zhao of Wake Forest. "I think the next step would be to ideally run a clinical trial and see if, in patients with elevated troponin levels, intensified medical management reduces the number of events as compared to standard therapy.
NEW YORK — An experimental, highly sensitive troponin test cannot predict when type 2 diabetics with stable ischemic heart disease will benefit from prompt coronary revascularization, but it can show which patients are more likely to die from myocardial infarction, stroke, or other cardiovascular cases, according to a new study.
Because the troponin test measures damage to heart muscle, the outcome "suggests there's ongoing injury to patients with stable heart disease and diabetes. It has a strong association with death, heart attack, stroke, heart failure," the chief author, Dr. Brendan Everett, director of inpatient general cardiology at Brigham and Women's Hospital in Boston, told Reuters Health in a telephone interview.
But when it comes to heading off a higher risk, "we need to do more research to understand where it's coming from and what therapies might be appropriate. So it does not appear in this population that opening the coronary arteries offers any long-term benefit with respect to death or heart attack," he said.
"It tells you this is not the type of disease status that can be managed by bypass or stenting," said Dr. David Zhao, chairman of cardiology at Wake Forest Baptist Medical Center in Winston-Salem, North Carolina. He was not involved in the research.
"From the patient's standpoint, it is disappointing," Dr. Zhao said. "But from a science standpoint, it all comes together nicely. These patients tend to have more small-vessel microvascular disease and the constant elevation of troponin indicates that there is more severe microvascular disease and more endomyocardial injury. I think it gives us the opportunity to look at more intensified medical management in this group of patients."
The study was an offshoot of the BARI 2D trial, conducted at 49 sites in six countries. It found that aggressive revascularization didn't reduce the risk of death or cardiovascular outcomes compared to intensive medical therapy alone in type 2 diabetics with stable heart disease.
In the new research, the team went back and used a highly sensitive troponin test sold in Europe but not available in the United States to see "if it could be used in stable patients to identify those who would benefit from having their coronary arteries opened with either angioplasty and stenting or bypass surgery," Dr. Everett said.
"There aren't many studies addressing that question and luckily BARI 2D had samples that were stored and available to us after the completion of the trial," he said. "What we did find was that there's clearly a group of patients that are at very high risk for death, heart attack, heart failure, and stroke."
The researchers found 27.1% of patients with a troponin T level of 14.0 ng/L or higher died from cardiovascular cause, or had a heart attack or stroke, compared to 12.9% of patients whose levels were lower, they reported online August 12 in the New England Journal of Medicine.
Adjusting for various factors, the risk of reaching one of those primary endpoints after five years was 85% greater with the higher troponin levels (P<0.001).
The risks for reaching individual components of the composite endpoint were also higher. Unadjusted five-year rates for patients with higher versus lower troponin levels were, respectively, 10.9% versus 3.5% for cardiovascular death, 18.7% versus 9.2% for myocardial infarction, 4.4% versus 2.3% for stroke, and 25.7% versus 11.1% for heart failure.
The Everett team also found that among the 897 volunteers whose levels had been at 14 or above, receiving a prompt intervention did not reduce their likelihood of reaching a primary endpoint compared to the 2,277 with normal concentrations.
The five-year rates of heart attack, stroke, or death from any cardiovascular cause were 26.5% in the revascularization group and 27.6% with medical therapy alone.
"We need to figure out how to better treat them, because fixing their coronary arteries doesn't seem to be giving them the benefit we thought it would, or hoped it would," said Dr. Everett. "For the time being, this is not a test that should be routinely used on stable patients."
The researchers also looked at how troponin levels changed over time.
"Despite aggressive medical therapy for type 2 diabetes and stable ischemic heart disease, the median troponin T concentration increased over one year of follow-up, and no significant reductions in troponin T concentrations were observed in patients who underwent coronary revascularization," the researchers said.
"If this type of patient has elevated troponin, I would be more aggressive in medical management, including statins, smoking cessation, and lifestyle changes," said Dr. Zhao of Wake Forest. "I think the next step would be to ideally run a clinical trial and see if, in patients with elevated troponin levels, intensified medical management reduces the number of events as compared to standard therapy.
Solitary confinement
A recent study released by the Association of State Correctional Administrators and researchers from Yale Law School has found that federal and state prisons are holding as many as 100,000 inmates in solitary confinement or isolated housing (“Large Number of Inmates in Solitary Poses Problem for Justice System, Study Says,” by Jess Bravin, Wall Street Journal, Sept. 2, 2015). This new data has turned up the volume of voices calling for abolishment of solitary confinement on the grounds that not only is it inhumane but also counterproductive.
Do you agree with abolitionists or are you sympathetic to some prison workers and administrators who say that there are situations in which social isolation is the best and maybe the only solution when a prisoner is a serious threat to the safety of his fellow inmates and staff?
While you are mulling over your answer, here is a related question more relevant to your own situation. How do you feel about solitary confinement (a.k.a., time-out) as a consequence for a misbehaving preschooler?
Do you think it is cruel and inhumane? Do you recommend it to parents as part of a comprehensive behavior-management strategy? Will many parents try it? Or, do they recoil and wonder why you would suggest that they become prison wardens in their own homes? If parents try it, is it effective?
In my experience, if done correctly in the right circumstances, time-out for a young child in his room – even if it requires latching the door – can be a safe, humane, and effective consequence for misbehavior. Sometimes, it is the only thing that works. But the devil is in the “ifs.”
First, time-out should be the last step in a comprehensive behavior-management strategy that begins with prevention – by assuring that the child is getting enough sleep and the right kind of attention from his parents who have expectations for their child that are appropriate for his age and temperament. The child’s environment and schedule should be structured to minimize the temptation to misbehave. Other less-drastic-sounding consequences must have been tried unsuccessfully. And ... both parent and child must be psychologically and developmentally normal.
Will brief episodes of solitary confinement make a young child feel insecure or unloved? Not if his parents make it clear by their behavior that she is loved and living in a stable environment, regardless of whether she is in time-out or not. Will time-out make a child hate her room? I’ve never seen it happen. If the child plays happily in her room during her sentence, does this render time-out ineffective? No, that’s a win-win situation. The misbehavior has stopped and the child is happy. Does this mean that time-out may not be a good deterrent? It might. But I have found that the only effective deterrent is consistent follow-up of every threat with the promised consequence – regardless of the consequence.
What if the child “destroys” his room during time-out? And is it safe to leave a child alone in his room? The solutions to these challenges can be found in Lowes or Home Depot.
I’m not going to take up any more of your recreational reading time describing the details of how time-out can be made more effective and palatable for parents. But it can be done and may require purchasing a latch or some kind of child-resistant door closure device. It will most likely be used briefly – if at all – but it can remain as a tangible reminder to the child that his parent follows up on his threats.
I won’t be surprised if some of you are shocked that I would advocate solitary confinement for young children. I am interested to hear what you recommend to parents who are struggling to keep their child’s behavior in bounds.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “Coping With a Picky Eater.”
A recent study released by the Association of State Correctional Administrators and researchers from Yale Law School has found that federal and state prisons are holding as many as 100,000 inmates in solitary confinement or isolated housing (“Large Number of Inmates in Solitary Poses Problem for Justice System, Study Says,” by Jess Bravin, Wall Street Journal, Sept. 2, 2015). This new data has turned up the volume of voices calling for abolishment of solitary confinement on the grounds that not only is it inhumane but also counterproductive.
Do you agree with abolitionists or are you sympathetic to some prison workers and administrators who say that there are situations in which social isolation is the best and maybe the only solution when a prisoner is a serious threat to the safety of his fellow inmates and staff?
While you are mulling over your answer, here is a related question more relevant to your own situation. How do you feel about solitary confinement (a.k.a., time-out) as a consequence for a misbehaving preschooler?
Do you think it is cruel and inhumane? Do you recommend it to parents as part of a comprehensive behavior-management strategy? Will many parents try it? Or, do they recoil and wonder why you would suggest that they become prison wardens in their own homes? If parents try it, is it effective?
In my experience, if done correctly in the right circumstances, time-out for a young child in his room – even if it requires latching the door – can be a safe, humane, and effective consequence for misbehavior. Sometimes, it is the only thing that works. But the devil is in the “ifs.”
First, time-out should be the last step in a comprehensive behavior-management strategy that begins with prevention – by assuring that the child is getting enough sleep and the right kind of attention from his parents who have expectations for their child that are appropriate for his age and temperament. The child’s environment and schedule should be structured to minimize the temptation to misbehave. Other less-drastic-sounding consequences must have been tried unsuccessfully. And ... both parent and child must be psychologically and developmentally normal.
Will brief episodes of solitary confinement make a young child feel insecure or unloved? Not if his parents make it clear by their behavior that she is loved and living in a stable environment, regardless of whether she is in time-out or not. Will time-out make a child hate her room? I’ve never seen it happen. If the child plays happily in her room during her sentence, does this render time-out ineffective? No, that’s a win-win situation. The misbehavior has stopped and the child is happy. Does this mean that time-out may not be a good deterrent? It might. But I have found that the only effective deterrent is consistent follow-up of every threat with the promised consequence – regardless of the consequence.
What if the child “destroys” his room during time-out? And is it safe to leave a child alone in his room? The solutions to these challenges can be found in Lowes or Home Depot.
I’m not going to take up any more of your recreational reading time describing the details of how time-out can be made more effective and palatable for parents. But it can be done and may require purchasing a latch or some kind of child-resistant door closure device. It will most likely be used briefly – if at all – but it can remain as a tangible reminder to the child that his parent follows up on his threats.
I won’t be surprised if some of you are shocked that I would advocate solitary confinement for young children. I am interested to hear what you recommend to parents who are struggling to keep their child’s behavior in bounds.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “Coping With a Picky Eater.”
A recent study released by the Association of State Correctional Administrators and researchers from Yale Law School has found that federal and state prisons are holding as many as 100,000 inmates in solitary confinement or isolated housing (“Large Number of Inmates in Solitary Poses Problem for Justice System, Study Says,” by Jess Bravin, Wall Street Journal, Sept. 2, 2015). This new data has turned up the volume of voices calling for abolishment of solitary confinement on the grounds that not only is it inhumane but also counterproductive.
Do you agree with abolitionists or are you sympathetic to some prison workers and administrators who say that there are situations in which social isolation is the best and maybe the only solution when a prisoner is a serious threat to the safety of his fellow inmates and staff?
While you are mulling over your answer, here is a related question more relevant to your own situation. How do you feel about solitary confinement (a.k.a., time-out) as a consequence for a misbehaving preschooler?
Do you think it is cruel and inhumane? Do you recommend it to parents as part of a comprehensive behavior-management strategy? Will many parents try it? Or, do they recoil and wonder why you would suggest that they become prison wardens in their own homes? If parents try it, is it effective?
In my experience, if done correctly in the right circumstances, time-out for a young child in his room – even if it requires latching the door – can be a safe, humane, and effective consequence for misbehavior. Sometimes, it is the only thing that works. But the devil is in the “ifs.”
First, time-out should be the last step in a comprehensive behavior-management strategy that begins with prevention – by assuring that the child is getting enough sleep and the right kind of attention from his parents who have expectations for their child that are appropriate for his age and temperament. The child’s environment and schedule should be structured to minimize the temptation to misbehave. Other less-drastic-sounding consequences must have been tried unsuccessfully. And ... both parent and child must be psychologically and developmentally normal.
Will brief episodes of solitary confinement make a young child feel insecure or unloved? Not if his parents make it clear by their behavior that she is loved and living in a stable environment, regardless of whether she is in time-out or not. Will time-out make a child hate her room? I’ve never seen it happen. If the child plays happily in her room during her sentence, does this render time-out ineffective? No, that’s a win-win situation. The misbehavior has stopped and the child is happy. Does this mean that time-out may not be a good deterrent? It might. But I have found that the only effective deterrent is consistent follow-up of every threat with the promised consequence – regardless of the consequence.
What if the child “destroys” his room during time-out? And is it safe to leave a child alone in his room? The solutions to these challenges can be found in Lowes or Home Depot.
I’m not going to take up any more of your recreational reading time describing the details of how time-out can be made more effective and palatable for parents. But it can be done and may require purchasing a latch or some kind of child-resistant door closure device. It will most likely be used briefly – if at all – but it can remain as a tangible reminder to the child that his parent follows up on his threats.
I won’t be surprised if some of you are shocked that I would advocate solitary confinement for young children. I am interested to hear what you recommend to parents who are struggling to keep their child’s behavior in bounds.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “Coping With a Picky Eater.”