User login
Contraception for the perimenopausal woman: What’s best?
› Consider long-acting reversible contraception, such as an intrauterine device or an implant, as a first-line option for women who have mild or no symptoms of perimenopause. A
› Unless contraindicated, prescribe combination hormonal contraceptives for women in their 40s who desire them, as they are generally safe and effective in treating perimenopausal symptoms. A
› Use the Centers for Disease Control and Prevention’s evidence-based recommendations to guide your choice of contraceptive for perimenopausal patients based on individual medical history. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
It is no secret that about half of all pregnancies in the United States are unintended, and that teens have the highest rate of unplanned pregnancy. What’s not so well known is that women in their 40s have the second highest rate.1
Optimal use of contraception throughout perimenopause is crucial, but finding the right method of birth control for this patient population can be a bit of a balancing act. Long-acting reversible contraceptives (LARCs), such as an intrauterine device or progestin-only implant, are preferred first-line contraceptive options when preventing pregnancy is the primary goal, given their increased efficacy and limited number of contraindications.2,3 However, women experiencing perimenopausal symptoms often need a combination hormonal contraceptive (CHC)—typically an estrogen-containing pill, a patch, or a vaginal ring—for relief of vasomotor symptoms and cycle control.
Women in their 40s should have access to a full array of options to help improve adherence. However, physicians may be reluctant to prescribe estrogen-containing products for patients who often have a more complex medical history than their younger counterparts, including increased risks for breast cancer, cardiovascular disease, and venous thromboembolism (VTE).
With this in mind, the Centers for Disease Control and Prevention (CDC) has identified medical conditions that may affect the use of the various types of contraceptives by perimenopausal women and issued evidence-based recommendations on the appropriateness of each method using a one-to-4 rating system (TABLE 1).2 To help you address the contraceptive needs of such patients, we review the key risk factors, CDC guidelines, and optimal choices in the 4 case studies that follow.
CASE 1 › Sara G: VTE risk
Sara G, a healthy 45-year-old, recently started dating again following her divorce. She wants to avoid pregnancy. She has no personal or family history of clotting disorders and does not smoke. However, she is obese (body mass index [BMI]=32 kg/m2), and her job as a visiting nurse requires her to spend most of the day in her car. Ms. G also has acne and wants an estrogen-containing contraceptive to help treat it.
If Ms. G were your patient, what would you offer her?
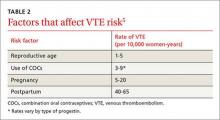
The risk for VTE increases substantially for women older than 40 years. In a recent cohort study, those ages 45 to 49 faced approximately twice the risk of women ages 25 to 29. However, the absolute risk for the older women was still low (4.7-5.3 per 10,000 woman-years).4 What’s more, the risk of VTE from the use of a CHC is substantially less than the risk associated with pregnancy and the postpartum period (TABLE 2).5
Obesity increases the risk. Women like Ms. G who are obese (BMI >30) have an increased risk for VTE associated with CHCs, but the CDC rates them as a Category 2 risk, even for obese women in their 40s—a determination that the advantages outweigh the risks.2
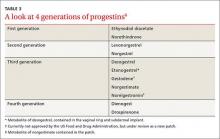
Progestin choice and estrogen dose matter. Combination oral contraceptives (COCs) that contain certain third-generation progestins (gestodene and desogestrel) may be more thrombophilic than those containing first- or second-generation progestins (TABLE 3).6 The relative risk (RR) for VTE with third-generation vs second-generation progestins is 1.3 (95% confidence interval [CI], 1.0-1.8).7 Formulations containing higher doses of estrogen are also more likely to be associated with VTE.7
Drospirenone is a newer progestin. Found in several COCs, drospirenone has antimineralocorticoid properties that help to minimize bloating and fluid retention but may also lead to a hypercoagulable state.5 Numerous studies have investigated the association between drospirenone and VTE risk, with conflicting results.8 Most recently, a large international prospective observational study involving more than 85,000 women showed no increased risk for VTE among women taking COCs with drospirenone compared with pills that do not contain this progestin.9
Non-oral CHCs, including the vaginal ring and the patch, offer the convenience of weekly or monthly use while providing similar benefits to COCs. Some fear that the continuous exposure to hormones associated with these methods may increase the risk for VTE, but evidence is mixed.
A large (N=1.6 million) Danish registry study published in 2012 demonstrated a 2-fold increased risk of VTE among vaginal ring users vs women taking COCs.4 But a multinational prospective cohort study of more than 33,000 women found no increased VTE risk in ring users,10 and a recent US database study involving more than 800,000 women reported nonsignificant VTE risk estimates for both the ring (RR=1.09; 95% CI, 0.55-2.16) and the patch (RR=1.35; 95% CI, 0.90-2.02) compared with COCs.11
THE BOTTOM LINE For Ms. G, the benefits of contraception likely outweigh any small increase in her absolute risk for VTE. To minimize her risk, however, select a pill that contains a low dose (20-35 mcg) of ethinyl estradiol (EE) combined with a progestin that has not been associated with an increased VTE risk. Because of their mechanism of action, most COCs will improve acne, regardless of the progestin in the formulation.12-14
CASE 2 › Stephanie T: CV risk
Stephanie T, 47, is in need of contraception and treatment for severe hot flashes. She has no significant past medical history, but she is obese (BMI =36), her blood pressure (BP) is 130/80 mm Hg, and her most recent labs reveal a fasting glucose of 115 and a hemoglobin A1c of 6.1%. Ms. T is concerned about arterial thromboembolic disease because of her family history: Her father had a myocardial infarction (MI) at age 56 and a maternal aunt had a stroke when she was 65.
What evidence should you consider?
Baseline arterial thromboembolic events are considerably more rare in premenopausal women than VTEs (13.2 MIs vs 24.2 thrombotic strokes per 100,000 woman-years).15 Thus, a small increased RR from a CHC is unlikely to have a significant clinical impact.
A systemic review and meta-analysis of studies between 1995 and 2012 showed that the odds ratio (OR) of ischemic stroke in users of COCs vs nonusers was 1.9 (95% CI, 1.24–2.91).16 This study included very few estrogen formulations with <35 mcg EE, however; even so, no increased risk of MI was found (OR=1.34; 95% CI, 0.87–2.08).16 A 15-year retrospective cohort study of 1.6 million Danish women showed that lowering the dose of EE to 20 mcg (from 30-40 mcg) significantly reduced the risk of arterial events.15 It is unclear whether the vaginal ring is associated with an increased RR of stroke compared with COCs because studies have had mixed results.10,15 There is no compelling evidence to suggest a difference in the risk of arterial events based on the type of progestin used in the COC.15
Hypertension is a key consideration. It is important to remember that perimenopausal women may have comorbid conditions that increase their risk of arterial thromboembolic events. CHCs should be used with caution in women with hypertension, even if BP is adequately controlled—a Category 3 recommendation from the CDC. In such patients, LARC or a progestin-only pill is preferred unless there is a compelling reason to use a CHC, such as acne, vasomotor symptoms, or hirsutism.2
CHCs are contraindicated for women with a BP ≥160/100 mm Hg and/or any manifestation of vascular disease (Category 4).2 Although progestin-only methods are often preferred for women with established vascular disease, depot medroxyprogesterone acetate (DMPA) is an exception (Category 3).2 DMPA is not a first-line choice for such patients because of its potential to cause weight gain and worsening lipids, glucose, and insulin metabolism. Women with hypertriglyceridemia should have follow-up testing of lipid levels after initiation of hormonal contraception, especially if it contains estrogen.
Diabetes is not an absolute contraindication. Many women with diabetes can safely use CHCs (Category 2). The exceptions: those who have vascular disease, nephropathy, retinopathy, or neuropathy (Category 4) or have had diabetes for >20 years and therefore have the potential for undiagnosed vascular disease.2 Generally, the use of insulin should not affect decisions regarding CHCs, and patients can be reassured that the hormones will not worsen their diabetes control.
When caring for women who have multiple risk factors for cardiovascular disease, it is important to exercise clinical judgment regarding the appropriateness of CHCs (Categories 3 and 4). Progestin-only methods have a more favorable risk profile for women at the highest risk and may provide ample relief of perimenopausal symptoms.2
THE BOTTOM LINE Ms. T may benefit from a CHC due to her severe hot flashes. She should be encouraged to adopt healthy lifestyle changes, including diet and exercise, to decrease her risk of arterial thromboembolism and VTE, but she has no contraindications to the use of a CHC at this time.
CASE 3 › Leslie C: Bone health
Leslie C, age 45, is happy with the contraceptive he has used for the past 3 years—DMPA injections every 3 months. She has no perimenopausal symptoms. However, her mother had an osteoporotic hip fracture at age 70 and Ms. C is concerned about the long-term use of DMPA.
Should Ms. C be worried?
Because of DMPA’s association with bone loss, the US Food and Drug Administration issued a black box warning in 2004 recommending that this method be used for more than 2 years only by women for whom other birth control methods are deemed inappropriate.17
The bone loss may be reversed. Evidence suggests that the bone loss is reversible, however, and the American College of Obstetricians and Gynecologists has stated that a potential fracture risk need not limit a woman’s use of DMPA to 2 years.18 A retrospective cohort review of 312,295 women in the United Kingdom did not find evidence of an increased risk of fracture with long-term use of DMPA.19 It is important to note, however, that because of declining estrogen levels, perimenopausal women have fewer years than their younger counterparts to recover bone density upon discontinuation of DMPA.20,21
THE BOTTOM LINE Because Ms. C has no perimenopausal symptoms, she may do well with LARC, which—like DMPA —would free her of the need to remember to take, apply, or insert a contraceptive regularly. It may help to point out that LARCs provide superior contraceptive efficacy compared with DMPA injections (99% vs 94%).3 Nonetheless, she and other women in their 40s who need ongoing contraception should not be discouraged from using DMPA if that is their preference.
CASE 4 › Alissa B: Breast cancer risk
Alissa B, 49, has polycystic ovaries and wonders if it is safe for her to continue her COC. She has been happy with the treatment for years because it gives her relief from hot flashes and regulates her cycles. Her 46-year-old sister was recently diagnosed with invasive breast cancer, however, and Ms. B is afraid that the hormones she takes put her at increased risk.
Should you recommend another method?
Breast cancer is an important concern for many women as they age. Although Ms. B’s family history increases her risk for developing breast cancer, a systematic review indicates that COCs do not add to this risk.22
Weak association between family history and OC use. The review included 10 observational studies and one meta-analysis that investigated the association between COC use and breast cancer in women with a family history of the disease. Only 2 fair-quality studies showed an association, one of which included women who had begun taking the pill before 1975, when formulations typically contained higher doses of estrogen than present-day preparations.22
Data from a recently published meta-analysis also indicate that there is no increased risk for breast cancer from COCs among women with BRCA 1 or BRCA 2 mutations. The summary RR for breast cancer in such patients was 1.13 (95% CI, 0.88-1.45), but OC users had a lower risk for ovarian cancer (summary RR=0.50; 95% CI, 0.33-0.75).23 Additionally, investigators found no association between specific currently used COC formulations and breast cancer.24
THE BOTTOM LINE Based on an independent review of the evidence, the CDC has given a family history of breast cancer a Category 1 rating. Thus, Ms. B can be reassured that she may safely continue taking her COC, which is unlikely to increase her breast cancer risk.
CORRESPONDENCE
Pelin Batur, MD, NCMP, CCD, Cleveland Clinic Independence Family Health Center, 5001 Rockside Road, IN30, Cleveland, OH 44131; baturp@ccf.org.
1. Finer LB, Zolna MR. Unintended pregnancy in the United States: incidence and disparities, 2006. Contraception. 2011;84:478-485.
2. Centers for Disease Control and Prevention (CDC). U.S. medical eligibility criteria for contraceptive use, 2010. MMWR Recomm Rep. 2010;59:1-86.
3. Division of Reproductive Health, National Center for Chronic Disease Prevention and Health Promotion, Centers for Disease Control and Prevention (CDC). U.S. selected practice recommendations for contraceptive use, 2013: adapted from the World Health Organization selected practice recommendations for contraceptive use, 2nd edition. MMWR Recomm Rep. 2013;62:1-60.
4. Lidegaard O, Nielsen LH, Skovlund CW, et al. Venous thrombosis in users of non-oral hormonal contraception: follow-up study, Denmark 2001-10. BMJ. 2012;344:e2990.
5. Committee on gynecologic practice. ACOG committee opinion number 540: Risk of venous thromboembolism among users of drospirenone-containing oral contraceptive pills. Obstet Gynecol. 2012;120:1239-1242.
6. McNamara M, Batur P, DeSapri KT. In the clinic. Perimenopause. Ann Intern Med. 2015;162:ITC1-15.
7. de Bastos M, Stegeman BH, Rosendaal FR, et al. Combined oral contraceptives: venous thrombosis. Cochrane Database Syst Rev. 2014;3:CD010813.
8. Wu CQ, Grandi SM, Filion KB, et al. Drospirenone-containing oral contraceptive pills and the risk of venous and arterial thrombosis: a systematic review. BJOG. 2013;120:801-810.
9. Dinger J, Bardenheuer K, Heinemann K. Cardiovascular and general safety of a 24-day regimen of drospirenone-containing combined oral contraceptives: final results from the international active surveillance study of women taking oral contraceptives. Contraception. 2014;89:253-263.
10. Dinger J, Möhner S, Heinemann K. Cardiovascular risk associated with the use of an etonogestrel-containing vaginal ring. Obstet Gynecol. 2013;122:800-808.
11. Sidney S, Cheetham TC, Connell FA, et al. Recent combined hormonal contraceptives (CHCs) and the risk of thromboembolism and other cardiovascular events in new users. Contraception. 2013;87:93-100.
12. Arowojolu AO, Gallo MF, Lopez LM, et al. Combined oral contraceptive pills for treatment of acne. Cochrane Database Syst Rev. 2012;6:CD004425.
13. Koulianos GT. Treatment of acne with oral contraceptives: criteria for pill selection. Cutis. 2000;66:281-286.
14. Thorneycroft IH. Update on androgenicity. Am J Obstet Gynecol. 1999;180:288-294.
15. Lidegaard Ø, Løkkegaard E, Jensen A, et al. Thrombotic stroke and myocardial infarction with hormonal contraception. N Engl J Med. 2012;366:2257-2266.
16. Peragallo Urrutia R, Coeytaux RR, McBroom AJ, et al. Risk of acute thromboembolic events with oral contraceptive use: a systematic review and meta-analysis. Obstet Gynecol. 2013;122:380-389.
17. U.S. Food and Drug Administration. Safety: Depo-Provera (medroxyprogesterone acetate injectable suspension). U.S. Food and Drug Administration Web site. Available at: http://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm154784.htm. Accessed April 20, 2015.
18. Committee Opinion No. 602: Depot medroxyprogesterone acetate and bone effects. Obstet Gynecol. 2014;123:1398-1402.
19. Lanza LL, McQuay LJ, Rothman KJ, et al. Use of depot medroxyprogesterone acetate contraception and incidence of bone fracture. Obstet Gynecol. 2013;121:593-600.
20. Ettinger B, Pressman A, Sklarin P, et al. Associations between low levels of serum estradiol, bone density, and fractures among elderly women: the study of osteoporotic fractures. J Clin Endocrinol Metab. 1998;83:2239-2243.
21. Reginster JY, Sarlet N, Deroisy R, et al. Minimal levels of serum estradiol prevent postmenopausal bone loss. Calcif Tissue Int. 1992;51:340-343.
22. Gaffield ME, Culwell KR, Ravi A. Oral contraceptives and family history of breast cancer. Contraception. 2009;80:372-380.
23. Iodice S, Barile M, Rotmensz N, et al. Oral contraceptive use and breast or ovarian cancer risk in BRCA1/2 carriers: a meta-analysis. Eur J Cancer. 2010;46:2275-2284.
24. Marchbanks PA, Curtis KM, Mandel MG, et al. Oral contraceptive formulation and risk of breast cancer. Contraception. 2012;85:342-350.
› Consider long-acting reversible contraception, such as an intrauterine device or an implant, as a first-line option for women who have mild or no symptoms of perimenopause. A
› Unless contraindicated, prescribe combination hormonal contraceptives for women in their 40s who desire them, as they are generally safe and effective in treating perimenopausal symptoms. A
› Use the Centers for Disease Control and Prevention’s evidence-based recommendations to guide your choice of contraceptive for perimenopausal patients based on individual medical history. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
It is no secret that about half of all pregnancies in the United States are unintended, and that teens have the highest rate of unplanned pregnancy. What’s not so well known is that women in their 40s have the second highest rate.1
Optimal use of contraception throughout perimenopause is crucial, but finding the right method of birth control for this patient population can be a bit of a balancing act. Long-acting reversible contraceptives (LARCs), such as an intrauterine device or progestin-only implant, are preferred first-line contraceptive options when preventing pregnancy is the primary goal, given their increased efficacy and limited number of contraindications.2,3 However, women experiencing perimenopausal symptoms often need a combination hormonal contraceptive (CHC)—typically an estrogen-containing pill, a patch, or a vaginal ring—for relief of vasomotor symptoms and cycle control.
Women in their 40s should have access to a full array of options to help improve adherence. However, physicians may be reluctant to prescribe estrogen-containing products for patients who often have a more complex medical history than their younger counterparts, including increased risks for breast cancer, cardiovascular disease, and venous thromboembolism (VTE).
With this in mind, the Centers for Disease Control and Prevention (CDC) has identified medical conditions that may affect the use of the various types of contraceptives by perimenopausal women and issued evidence-based recommendations on the appropriateness of each method using a one-to-4 rating system (TABLE 1).2 To help you address the contraceptive needs of such patients, we review the key risk factors, CDC guidelines, and optimal choices in the 4 case studies that follow.
CASE 1 › Sara G: VTE risk
Sara G, a healthy 45-year-old, recently started dating again following her divorce. She wants to avoid pregnancy. She has no personal or family history of clotting disorders and does not smoke. However, she is obese (body mass index [BMI]=32 kg/m2), and her job as a visiting nurse requires her to spend most of the day in her car. Ms. G also has acne and wants an estrogen-containing contraceptive to help treat it.
If Ms. G were your patient, what would you offer her?
The risk for VTE increases substantially for women older than 40 years. In a recent cohort study, those ages 45 to 49 faced approximately twice the risk of women ages 25 to 29. However, the absolute risk for the older women was still low (4.7-5.3 per 10,000 woman-years).4 What’s more, the risk of VTE from the use of a CHC is substantially less than the risk associated with pregnancy and the postpartum period (TABLE 2).5
Obesity increases the risk. Women like Ms. G who are obese (BMI >30) have an increased risk for VTE associated with CHCs, but the CDC rates them as a Category 2 risk, even for obese women in their 40s—a determination that the advantages outweigh the risks.2
Progestin choice and estrogen dose matter. Combination oral contraceptives (COCs) that contain certain third-generation progestins (gestodene and desogestrel) may be more thrombophilic than those containing first- or second-generation progestins (TABLE 3).6 The relative risk (RR) for VTE with third-generation vs second-generation progestins is 1.3 (95% confidence interval [CI], 1.0-1.8).7 Formulations containing higher doses of estrogen are also more likely to be associated with VTE.7
Drospirenone is a newer progestin. Found in several COCs, drospirenone has antimineralocorticoid properties that help to minimize bloating and fluid retention but may also lead to a hypercoagulable state.5 Numerous studies have investigated the association between drospirenone and VTE risk, with conflicting results.8 Most recently, a large international prospective observational study involving more than 85,000 women showed no increased risk for VTE among women taking COCs with drospirenone compared with pills that do not contain this progestin.9
Non-oral CHCs, including the vaginal ring and the patch, offer the convenience of weekly or monthly use while providing similar benefits to COCs. Some fear that the continuous exposure to hormones associated with these methods may increase the risk for VTE, but evidence is mixed.
A large (N=1.6 million) Danish registry study published in 2012 demonstrated a 2-fold increased risk of VTE among vaginal ring users vs women taking COCs.4 But a multinational prospective cohort study of more than 33,000 women found no increased VTE risk in ring users,10 and a recent US database study involving more than 800,000 women reported nonsignificant VTE risk estimates for both the ring (RR=1.09; 95% CI, 0.55-2.16) and the patch (RR=1.35; 95% CI, 0.90-2.02) compared with COCs.11
THE BOTTOM LINE For Ms. G, the benefits of contraception likely outweigh any small increase in her absolute risk for VTE. To minimize her risk, however, select a pill that contains a low dose (20-35 mcg) of ethinyl estradiol (EE) combined with a progestin that has not been associated with an increased VTE risk. Because of their mechanism of action, most COCs will improve acne, regardless of the progestin in the formulation.12-14
CASE 2 › Stephanie T: CV risk
Stephanie T, 47, is in need of contraception and treatment for severe hot flashes. She has no significant past medical history, but she is obese (BMI =36), her blood pressure (BP) is 130/80 mm Hg, and her most recent labs reveal a fasting glucose of 115 and a hemoglobin A1c of 6.1%. Ms. T is concerned about arterial thromboembolic disease because of her family history: Her father had a myocardial infarction (MI) at age 56 and a maternal aunt had a stroke when she was 65.
What evidence should you consider?
Baseline arterial thromboembolic events are considerably more rare in premenopausal women than VTEs (13.2 MIs vs 24.2 thrombotic strokes per 100,000 woman-years).15 Thus, a small increased RR from a CHC is unlikely to have a significant clinical impact.
A systemic review and meta-analysis of studies between 1995 and 2012 showed that the odds ratio (OR) of ischemic stroke in users of COCs vs nonusers was 1.9 (95% CI, 1.24–2.91).16 This study included very few estrogen formulations with <35 mcg EE, however; even so, no increased risk of MI was found (OR=1.34; 95% CI, 0.87–2.08).16 A 15-year retrospective cohort study of 1.6 million Danish women showed that lowering the dose of EE to 20 mcg (from 30-40 mcg) significantly reduced the risk of arterial events.15 It is unclear whether the vaginal ring is associated with an increased RR of stroke compared with COCs because studies have had mixed results.10,15 There is no compelling evidence to suggest a difference in the risk of arterial events based on the type of progestin used in the COC.15
Hypertension is a key consideration. It is important to remember that perimenopausal women may have comorbid conditions that increase their risk of arterial thromboembolic events. CHCs should be used with caution in women with hypertension, even if BP is adequately controlled—a Category 3 recommendation from the CDC. In such patients, LARC or a progestin-only pill is preferred unless there is a compelling reason to use a CHC, such as acne, vasomotor symptoms, or hirsutism.2
CHCs are contraindicated for women with a BP ≥160/100 mm Hg and/or any manifestation of vascular disease (Category 4).2 Although progestin-only methods are often preferred for women with established vascular disease, depot medroxyprogesterone acetate (DMPA) is an exception (Category 3).2 DMPA is not a first-line choice for such patients because of its potential to cause weight gain and worsening lipids, glucose, and insulin metabolism. Women with hypertriglyceridemia should have follow-up testing of lipid levels after initiation of hormonal contraception, especially if it contains estrogen.
Diabetes is not an absolute contraindication. Many women with diabetes can safely use CHCs (Category 2). The exceptions: those who have vascular disease, nephropathy, retinopathy, or neuropathy (Category 4) or have had diabetes for >20 years and therefore have the potential for undiagnosed vascular disease.2 Generally, the use of insulin should not affect decisions regarding CHCs, and patients can be reassured that the hormones will not worsen their diabetes control.
When caring for women who have multiple risk factors for cardiovascular disease, it is important to exercise clinical judgment regarding the appropriateness of CHCs (Categories 3 and 4). Progestin-only methods have a more favorable risk profile for women at the highest risk and may provide ample relief of perimenopausal symptoms.2
THE BOTTOM LINE Ms. T may benefit from a CHC due to her severe hot flashes. She should be encouraged to adopt healthy lifestyle changes, including diet and exercise, to decrease her risk of arterial thromboembolism and VTE, but she has no contraindications to the use of a CHC at this time.
CASE 3 › Leslie C: Bone health
Leslie C, age 45, is happy with the contraceptive he has used for the past 3 years—DMPA injections every 3 months. She has no perimenopausal symptoms. However, her mother had an osteoporotic hip fracture at age 70 and Ms. C is concerned about the long-term use of DMPA.
Should Ms. C be worried?
Because of DMPA’s association with bone loss, the US Food and Drug Administration issued a black box warning in 2004 recommending that this method be used for more than 2 years only by women for whom other birth control methods are deemed inappropriate.17
The bone loss may be reversed. Evidence suggests that the bone loss is reversible, however, and the American College of Obstetricians and Gynecologists has stated that a potential fracture risk need not limit a woman’s use of DMPA to 2 years.18 A retrospective cohort review of 312,295 women in the United Kingdom did not find evidence of an increased risk of fracture with long-term use of DMPA.19 It is important to note, however, that because of declining estrogen levels, perimenopausal women have fewer years than their younger counterparts to recover bone density upon discontinuation of DMPA.20,21
THE BOTTOM LINE Because Ms. C has no perimenopausal symptoms, she may do well with LARC, which—like DMPA —would free her of the need to remember to take, apply, or insert a contraceptive regularly. It may help to point out that LARCs provide superior contraceptive efficacy compared with DMPA injections (99% vs 94%).3 Nonetheless, she and other women in their 40s who need ongoing contraception should not be discouraged from using DMPA if that is their preference.
CASE 4 › Alissa B: Breast cancer risk
Alissa B, 49, has polycystic ovaries and wonders if it is safe for her to continue her COC. She has been happy with the treatment for years because it gives her relief from hot flashes and regulates her cycles. Her 46-year-old sister was recently diagnosed with invasive breast cancer, however, and Ms. B is afraid that the hormones she takes put her at increased risk.
Should you recommend another method?
Breast cancer is an important concern for many women as they age. Although Ms. B’s family history increases her risk for developing breast cancer, a systematic review indicates that COCs do not add to this risk.22
Weak association between family history and OC use. The review included 10 observational studies and one meta-analysis that investigated the association between COC use and breast cancer in women with a family history of the disease. Only 2 fair-quality studies showed an association, one of which included women who had begun taking the pill before 1975, when formulations typically contained higher doses of estrogen than present-day preparations.22
Data from a recently published meta-analysis also indicate that there is no increased risk for breast cancer from COCs among women with BRCA 1 or BRCA 2 mutations. The summary RR for breast cancer in such patients was 1.13 (95% CI, 0.88-1.45), but OC users had a lower risk for ovarian cancer (summary RR=0.50; 95% CI, 0.33-0.75).23 Additionally, investigators found no association between specific currently used COC formulations and breast cancer.24
THE BOTTOM LINE Based on an independent review of the evidence, the CDC has given a family history of breast cancer a Category 1 rating. Thus, Ms. B can be reassured that she may safely continue taking her COC, which is unlikely to increase her breast cancer risk.
CORRESPONDENCE
Pelin Batur, MD, NCMP, CCD, Cleveland Clinic Independence Family Health Center, 5001 Rockside Road, IN30, Cleveland, OH 44131; baturp@ccf.org.
› Consider long-acting reversible contraception, such as an intrauterine device or an implant, as a first-line option for women who have mild or no symptoms of perimenopause. A
› Unless contraindicated, prescribe combination hormonal contraceptives for women in their 40s who desire them, as they are generally safe and effective in treating perimenopausal symptoms. A
› Use the Centers for Disease Control and Prevention’s evidence-based recommendations to guide your choice of contraceptive for perimenopausal patients based on individual medical history. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
It is no secret that about half of all pregnancies in the United States are unintended, and that teens have the highest rate of unplanned pregnancy. What’s not so well known is that women in their 40s have the second highest rate.1
Optimal use of contraception throughout perimenopause is crucial, but finding the right method of birth control for this patient population can be a bit of a balancing act. Long-acting reversible contraceptives (LARCs), such as an intrauterine device or progestin-only implant, are preferred first-line contraceptive options when preventing pregnancy is the primary goal, given their increased efficacy and limited number of contraindications.2,3 However, women experiencing perimenopausal symptoms often need a combination hormonal contraceptive (CHC)—typically an estrogen-containing pill, a patch, or a vaginal ring—for relief of vasomotor symptoms and cycle control.
Women in their 40s should have access to a full array of options to help improve adherence. However, physicians may be reluctant to prescribe estrogen-containing products for patients who often have a more complex medical history than their younger counterparts, including increased risks for breast cancer, cardiovascular disease, and venous thromboembolism (VTE).
With this in mind, the Centers for Disease Control and Prevention (CDC) has identified medical conditions that may affect the use of the various types of contraceptives by perimenopausal women and issued evidence-based recommendations on the appropriateness of each method using a one-to-4 rating system (TABLE 1).2 To help you address the contraceptive needs of such patients, we review the key risk factors, CDC guidelines, and optimal choices in the 4 case studies that follow.
CASE 1 › Sara G: VTE risk
Sara G, a healthy 45-year-old, recently started dating again following her divorce. She wants to avoid pregnancy. She has no personal or family history of clotting disorders and does not smoke. However, she is obese (body mass index [BMI]=32 kg/m2), and her job as a visiting nurse requires her to spend most of the day in her car. Ms. G also has acne and wants an estrogen-containing contraceptive to help treat it.
If Ms. G were your patient, what would you offer her?
The risk for VTE increases substantially for women older than 40 years. In a recent cohort study, those ages 45 to 49 faced approximately twice the risk of women ages 25 to 29. However, the absolute risk for the older women was still low (4.7-5.3 per 10,000 woman-years).4 What’s more, the risk of VTE from the use of a CHC is substantially less than the risk associated with pregnancy and the postpartum period (TABLE 2).5
Obesity increases the risk. Women like Ms. G who are obese (BMI >30) have an increased risk for VTE associated with CHCs, but the CDC rates them as a Category 2 risk, even for obese women in their 40s—a determination that the advantages outweigh the risks.2
Progestin choice and estrogen dose matter. Combination oral contraceptives (COCs) that contain certain third-generation progestins (gestodene and desogestrel) may be more thrombophilic than those containing first- or second-generation progestins (TABLE 3).6 The relative risk (RR) for VTE with third-generation vs second-generation progestins is 1.3 (95% confidence interval [CI], 1.0-1.8).7 Formulations containing higher doses of estrogen are also more likely to be associated with VTE.7
Drospirenone is a newer progestin. Found in several COCs, drospirenone has antimineralocorticoid properties that help to minimize bloating and fluid retention but may also lead to a hypercoagulable state.5 Numerous studies have investigated the association between drospirenone and VTE risk, with conflicting results.8 Most recently, a large international prospective observational study involving more than 85,000 women showed no increased risk for VTE among women taking COCs with drospirenone compared with pills that do not contain this progestin.9
Non-oral CHCs, including the vaginal ring and the patch, offer the convenience of weekly or monthly use while providing similar benefits to COCs. Some fear that the continuous exposure to hormones associated with these methods may increase the risk for VTE, but evidence is mixed.
A large (N=1.6 million) Danish registry study published in 2012 demonstrated a 2-fold increased risk of VTE among vaginal ring users vs women taking COCs.4 But a multinational prospective cohort study of more than 33,000 women found no increased VTE risk in ring users,10 and a recent US database study involving more than 800,000 women reported nonsignificant VTE risk estimates for both the ring (RR=1.09; 95% CI, 0.55-2.16) and the patch (RR=1.35; 95% CI, 0.90-2.02) compared with COCs.11
THE BOTTOM LINE For Ms. G, the benefits of contraception likely outweigh any small increase in her absolute risk for VTE. To minimize her risk, however, select a pill that contains a low dose (20-35 mcg) of ethinyl estradiol (EE) combined with a progestin that has not been associated with an increased VTE risk. Because of their mechanism of action, most COCs will improve acne, regardless of the progestin in the formulation.12-14
CASE 2 › Stephanie T: CV risk
Stephanie T, 47, is in need of contraception and treatment for severe hot flashes. She has no significant past medical history, but she is obese (BMI =36), her blood pressure (BP) is 130/80 mm Hg, and her most recent labs reveal a fasting glucose of 115 and a hemoglobin A1c of 6.1%. Ms. T is concerned about arterial thromboembolic disease because of her family history: Her father had a myocardial infarction (MI) at age 56 and a maternal aunt had a stroke when she was 65.
What evidence should you consider?
Baseline arterial thromboembolic events are considerably more rare in premenopausal women than VTEs (13.2 MIs vs 24.2 thrombotic strokes per 100,000 woman-years).15 Thus, a small increased RR from a CHC is unlikely to have a significant clinical impact.
A systemic review and meta-analysis of studies between 1995 and 2012 showed that the odds ratio (OR) of ischemic stroke in users of COCs vs nonusers was 1.9 (95% CI, 1.24–2.91).16 This study included very few estrogen formulations with <35 mcg EE, however; even so, no increased risk of MI was found (OR=1.34; 95% CI, 0.87–2.08).16 A 15-year retrospective cohort study of 1.6 million Danish women showed that lowering the dose of EE to 20 mcg (from 30-40 mcg) significantly reduced the risk of arterial events.15 It is unclear whether the vaginal ring is associated with an increased RR of stroke compared with COCs because studies have had mixed results.10,15 There is no compelling evidence to suggest a difference in the risk of arterial events based on the type of progestin used in the COC.15
Hypertension is a key consideration. It is important to remember that perimenopausal women may have comorbid conditions that increase their risk of arterial thromboembolic events. CHCs should be used with caution in women with hypertension, even if BP is adequately controlled—a Category 3 recommendation from the CDC. In such patients, LARC or a progestin-only pill is preferred unless there is a compelling reason to use a CHC, such as acne, vasomotor symptoms, or hirsutism.2
CHCs are contraindicated for women with a BP ≥160/100 mm Hg and/or any manifestation of vascular disease (Category 4).2 Although progestin-only methods are often preferred for women with established vascular disease, depot medroxyprogesterone acetate (DMPA) is an exception (Category 3).2 DMPA is not a first-line choice for such patients because of its potential to cause weight gain and worsening lipids, glucose, and insulin metabolism. Women with hypertriglyceridemia should have follow-up testing of lipid levels after initiation of hormonal contraception, especially if it contains estrogen.
Diabetes is not an absolute contraindication. Many women with diabetes can safely use CHCs (Category 2). The exceptions: those who have vascular disease, nephropathy, retinopathy, or neuropathy (Category 4) or have had diabetes for >20 years and therefore have the potential for undiagnosed vascular disease.2 Generally, the use of insulin should not affect decisions regarding CHCs, and patients can be reassured that the hormones will not worsen their diabetes control.
When caring for women who have multiple risk factors for cardiovascular disease, it is important to exercise clinical judgment regarding the appropriateness of CHCs (Categories 3 and 4). Progestin-only methods have a more favorable risk profile for women at the highest risk and may provide ample relief of perimenopausal symptoms.2
THE BOTTOM LINE Ms. T may benefit from a CHC due to her severe hot flashes. She should be encouraged to adopt healthy lifestyle changes, including diet and exercise, to decrease her risk of arterial thromboembolism and VTE, but she has no contraindications to the use of a CHC at this time.
CASE 3 › Leslie C: Bone health
Leslie C, age 45, is happy with the contraceptive he has used for the past 3 years—DMPA injections every 3 months. She has no perimenopausal symptoms. However, her mother had an osteoporotic hip fracture at age 70 and Ms. C is concerned about the long-term use of DMPA.
Should Ms. C be worried?
Because of DMPA’s association with bone loss, the US Food and Drug Administration issued a black box warning in 2004 recommending that this method be used for more than 2 years only by women for whom other birth control methods are deemed inappropriate.17
The bone loss may be reversed. Evidence suggests that the bone loss is reversible, however, and the American College of Obstetricians and Gynecologists has stated that a potential fracture risk need not limit a woman’s use of DMPA to 2 years.18 A retrospective cohort review of 312,295 women in the United Kingdom did not find evidence of an increased risk of fracture with long-term use of DMPA.19 It is important to note, however, that because of declining estrogen levels, perimenopausal women have fewer years than their younger counterparts to recover bone density upon discontinuation of DMPA.20,21
THE BOTTOM LINE Because Ms. C has no perimenopausal symptoms, she may do well with LARC, which—like DMPA —would free her of the need to remember to take, apply, or insert a contraceptive regularly. It may help to point out that LARCs provide superior contraceptive efficacy compared with DMPA injections (99% vs 94%).3 Nonetheless, she and other women in their 40s who need ongoing contraception should not be discouraged from using DMPA if that is their preference.
CASE 4 › Alissa B: Breast cancer risk
Alissa B, 49, has polycystic ovaries and wonders if it is safe for her to continue her COC. She has been happy with the treatment for years because it gives her relief from hot flashes and regulates her cycles. Her 46-year-old sister was recently diagnosed with invasive breast cancer, however, and Ms. B is afraid that the hormones she takes put her at increased risk.
Should you recommend another method?
Breast cancer is an important concern for many women as they age. Although Ms. B’s family history increases her risk for developing breast cancer, a systematic review indicates that COCs do not add to this risk.22
Weak association between family history and OC use. The review included 10 observational studies and one meta-analysis that investigated the association between COC use and breast cancer in women with a family history of the disease. Only 2 fair-quality studies showed an association, one of which included women who had begun taking the pill before 1975, when formulations typically contained higher doses of estrogen than present-day preparations.22
Data from a recently published meta-analysis also indicate that there is no increased risk for breast cancer from COCs among women with BRCA 1 or BRCA 2 mutations. The summary RR for breast cancer in such patients was 1.13 (95% CI, 0.88-1.45), but OC users had a lower risk for ovarian cancer (summary RR=0.50; 95% CI, 0.33-0.75).23 Additionally, investigators found no association between specific currently used COC formulations and breast cancer.24
THE BOTTOM LINE Based on an independent review of the evidence, the CDC has given a family history of breast cancer a Category 1 rating. Thus, Ms. B can be reassured that she may safely continue taking her COC, which is unlikely to increase her breast cancer risk.
CORRESPONDENCE
Pelin Batur, MD, NCMP, CCD, Cleveland Clinic Independence Family Health Center, 5001 Rockside Road, IN30, Cleveland, OH 44131; baturp@ccf.org.
1. Finer LB, Zolna MR. Unintended pregnancy in the United States: incidence and disparities, 2006. Contraception. 2011;84:478-485.
2. Centers for Disease Control and Prevention (CDC). U.S. medical eligibility criteria for contraceptive use, 2010. MMWR Recomm Rep. 2010;59:1-86.
3. Division of Reproductive Health, National Center for Chronic Disease Prevention and Health Promotion, Centers for Disease Control and Prevention (CDC). U.S. selected practice recommendations for contraceptive use, 2013: adapted from the World Health Organization selected practice recommendations for contraceptive use, 2nd edition. MMWR Recomm Rep. 2013;62:1-60.
4. Lidegaard O, Nielsen LH, Skovlund CW, et al. Venous thrombosis in users of non-oral hormonal contraception: follow-up study, Denmark 2001-10. BMJ. 2012;344:e2990.
5. Committee on gynecologic practice. ACOG committee opinion number 540: Risk of venous thromboembolism among users of drospirenone-containing oral contraceptive pills. Obstet Gynecol. 2012;120:1239-1242.
6. McNamara M, Batur P, DeSapri KT. In the clinic. Perimenopause. Ann Intern Med. 2015;162:ITC1-15.
7. de Bastos M, Stegeman BH, Rosendaal FR, et al. Combined oral contraceptives: venous thrombosis. Cochrane Database Syst Rev. 2014;3:CD010813.
8. Wu CQ, Grandi SM, Filion KB, et al. Drospirenone-containing oral contraceptive pills and the risk of venous and arterial thrombosis: a systematic review. BJOG. 2013;120:801-810.
9. Dinger J, Bardenheuer K, Heinemann K. Cardiovascular and general safety of a 24-day regimen of drospirenone-containing combined oral contraceptives: final results from the international active surveillance study of women taking oral contraceptives. Contraception. 2014;89:253-263.
10. Dinger J, Möhner S, Heinemann K. Cardiovascular risk associated with the use of an etonogestrel-containing vaginal ring. Obstet Gynecol. 2013;122:800-808.
11. Sidney S, Cheetham TC, Connell FA, et al. Recent combined hormonal contraceptives (CHCs) and the risk of thromboembolism and other cardiovascular events in new users. Contraception. 2013;87:93-100.
12. Arowojolu AO, Gallo MF, Lopez LM, et al. Combined oral contraceptive pills for treatment of acne. Cochrane Database Syst Rev. 2012;6:CD004425.
13. Koulianos GT. Treatment of acne with oral contraceptives: criteria for pill selection. Cutis. 2000;66:281-286.
14. Thorneycroft IH. Update on androgenicity. Am J Obstet Gynecol. 1999;180:288-294.
15. Lidegaard Ø, Løkkegaard E, Jensen A, et al. Thrombotic stroke and myocardial infarction with hormonal contraception. N Engl J Med. 2012;366:2257-2266.
16. Peragallo Urrutia R, Coeytaux RR, McBroom AJ, et al. Risk of acute thromboembolic events with oral contraceptive use: a systematic review and meta-analysis. Obstet Gynecol. 2013;122:380-389.
17. U.S. Food and Drug Administration. Safety: Depo-Provera (medroxyprogesterone acetate injectable suspension). U.S. Food and Drug Administration Web site. Available at: http://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm154784.htm. Accessed April 20, 2015.
18. Committee Opinion No. 602: Depot medroxyprogesterone acetate and bone effects. Obstet Gynecol. 2014;123:1398-1402.
19. Lanza LL, McQuay LJ, Rothman KJ, et al. Use of depot medroxyprogesterone acetate contraception and incidence of bone fracture. Obstet Gynecol. 2013;121:593-600.
20. Ettinger B, Pressman A, Sklarin P, et al. Associations between low levels of serum estradiol, bone density, and fractures among elderly women: the study of osteoporotic fractures. J Clin Endocrinol Metab. 1998;83:2239-2243.
21. Reginster JY, Sarlet N, Deroisy R, et al. Minimal levels of serum estradiol prevent postmenopausal bone loss. Calcif Tissue Int. 1992;51:340-343.
22. Gaffield ME, Culwell KR, Ravi A. Oral contraceptives and family history of breast cancer. Contraception. 2009;80:372-380.
23. Iodice S, Barile M, Rotmensz N, et al. Oral contraceptive use and breast or ovarian cancer risk in BRCA1/2 carriers: a meta-analysis. Eur J Cancer. 2010;46:2275-2284.
24. Marchbanks PA, Curtis KM, Mandel MG, et al. Oral contraceptive formulation and risk of breast cancer. Contraception. 2012;85:342-350.
1. Finer LB, Zolna MR. Unintended pregnancy in the United States: incidence and disparities, 2006. Contraception. 2011;84:478-485.
2. Centers for Disease Control and Prevention (CDC). U.S. medical eligibility criteria for contraceptive use, 2010. MMWR Recomm Rep. 2010;59:1-86.
3. Division of Reproductive Health, National Center for Chronic Disease Prevention and Health Promotion, Centers for Disease Control and Prevention (CDC). U.S. selected practice recommendations for contraceptive use, 2013: adapted from the World Health Organization selected practice recommendations for contraceptive use, 2nd edition. MMWR Recomm Rep. 2013;62:1-60.
4. Lidegaard O, Nielsen LH, Skovlund CW, et al. Venous thrombosis in users of non-oral hormonal contraception: follow-up study, Denmark 2001-10. BMJ. 2012;344:e2990.
5. Committee on gynecologic practice. ACOG committee opinion number 540: Risk of venous thromboembolism among users of drospirenone-containing oral contraceptive pills. Obstet Gynecol. 2012;120:1239-1242.
6. McNamara M, Batur P, DeSapri KT. In the clinic. Perimenopause. Ann Intern Med. 2015;162:ITC1-15.
7. de Bastos M, Stegeman BH, Rosendaal FR, et al. Combined oral contraceptives: venous thrombosis. Cochrane Database Syst Rev. 2014;3:CD010813.
8. Wu CQ, Grandi SM, Filion KB, et al. Drospirenone-containing oral contraceptive pills and the risk of venous and arterial thrombosis: a systematic review. BJOG. 2013;120:801-810.
9. Dinger J, Bardenheuer K, Heinemann K. Cardiovascular and general safety of a 24-day regimen of drospirenone-containing combined oral contraceptives: final results from the international active surveillance study of women taking oral contraceptives. Contraception. 2014;89:253-263.
10. Dinger J, Möhner S, Heinemann K. Cardiovascular risk associated with the use of an etonogestrel-containing vaginal ring. Obstet Gynecol. 2013;122:800-808.
11. Sidney S, Cheetham TC, Connell FA, et al. Recent combined hormonal contraceptives (CHCs) and the risk of thromboembolism and other cardiovascular events in new users. Contraception. 2013;87:93-100.
12. Arowojolu AO, Gallo MF, Lopez LM, et al. Combined oral contraceptive pills for treatment of acne. Cochrane Database Syst Rev. 2012;6:CD004425.
13. Koulianos GT. Treatment of acne with oral contraceptives: criteria for pill selection. Cutis. 2000;66:281-286.
14. Thorneycroft IH. Update on androgenicity. Am J Obstet Gynecol. 1999;180:288-294.
15. Lidegaard Ø, Løkkegaard E, Jensen A, et al. Thrombotic stroke and myocardial infarction with hormonal contraception. N Engl J Med. 2012;366:2257-2266.
16. Peragallo Urrutia R, Coeytaux RR, McBroom AJ, et al. Risk of acute thromboembolic events with oral contraceptive use: a systematic review and meta-analysis. Obstet Gynecol. 2013;122:380-389.
17. U.S. Food and Drug Administration. Safety: Depo-Provera (medroxyprogesterone acetate injectable suspension). U.S. Food and Drug Administration Web site. Available at: http://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm154784.htm. Accessed April 20, 2015.
18. Committee Opinion No. 602: Depot medroxyprogesterone acetate and bone effects. Obstet Gynecol. 2014;123:1398-1402.
19. Lanza LL, McQuay LJ, Rothman KJ, et al. Use of depot medroxyprogesterone acetate contraception and incidence of bone fracture. Obstet Gynecol. 2013;121:593-600.
20. Ettinger B, Pressman A, Sklarin P, et al. Associations between low levels of serum estradiol, bone density, and fractures among elderly women: the study of osteoporotic fractures. J Clin Endocrinol Metab. 1998;83:2239-2243.
21. Reginster JY, Sarlet N, Deroisy R, et al. Minimal levels of serum estradiol prevent postmenopausal bone loss. Calcif Tissue Int. 1992;51:340-343.
22. Gaffield ME, Culwell KR, Ravi A. Oral contraceptives and family history of breast cancer. Contraception. 2009;80:372-380.
23. Iodice S, Barile M, Rotmensz N, et al. Oral contraceptive use and breast or ovarian cancer risk in BRCA1/2 carriers: a meta-analysis. Eur J Cancer. 2010;46:2275-2284.
24. Marchbanks PA, Curtis KM, Mandel MG, et al. Oral contraceptive formulation and risk of breast cancer. Contraception. 2012;85:342-350.
Renal Denervation
Q) I’ve heard a lot of references to “renal denervation” and its use for resistant hypertension. What is it? Does it work? Is it common in the US?
Renal denervation is a minimally invasive endovascular procedure that ablates (or disrupts) the renal nerves in and around the renal arteries with radiofrequency energy.5 Renal denervation has been approved in the US and other countries and is being used clinically in Europe, Canada, and Australia.6
It is thought that renal denervation interrupts the efferent and afferent signals that stimulate the renin-angiotensin-aldosterone system (RAAS) and regulate whole-body sympathetic nervous system activity.5 Similar to surgical sympathectomy, renal denervation should theoretically lower blood pressure. However, Ezzahti et al found that renin levels did not decrease in patients following renal denervation.7
Drug-resistant hypertension is defined as blood pressure that remains greater than 140/90 mm Hg despite treatment with three or more antihypertensive medications, including a diuretic.8 Patients with resistant hypertension have increased cardiovascular risk.9 Clinical trials of renal denervation have focused on treatment of resistant hypertension, in the hope of reducing the associated morbidity and mortality.
Results of the Symplicity HTN-3 trial, which assessed the safety and efficacy of renal denervation, were anxiously awaited, since prior trials yielded mixed results. Although the Symplicity HTN-1 and Symplicity HTN-2 studies demonstrated a possible benefit of renal denervation to lower office measured blood pressure, other studies did not show a decrease in BP in patients who had undergone renal denervation.6,7 These early trials, however, were small and did not randomize patients to a sham procedure.10
The Symplicity HTN-3 trial included 535 patients at 88 centers in the US. Patients were randomly assigned to receive either renal denervation plus baseline antihypertensive medications or a sham procedure plus baseline antihypertensive medications.
The researchers found that the sham procedure was just as effective as the “true” renal denervation in decreasing systolic blood pressure in patients with resistant hypertension.10 In other words, renal denervation did not demonstrate efficacy for this purpose.
In response to the results of this well-designed trial, the FDA has halted approval to perform renal denervation in patients with resistant hypertension in the US. However, clinical investigation will continue among subgroups of hypertensive patients or separate populations.
Despite a lack of efficacy, renal denervation does appear to be well tolerated, as evidenced by safety data from Symplicity HTN-3. —JK
Jessica Knight, ACNP
University of New Mexico Hospital, Albuquerque
REFERENCES
5. Esler MD, Krum H, Schlaich M, et al. Renal sympathetic denervation for the treatment of drug-resistant hypertension: one-year results from the Symplicity HTN-2 randomized, controlled trial. Circulation. 2012;126(25):2976-2982.
6. Thukkani AK, Bhatt LD. Renal denervation therapy for hypertension. Circulation. 2013;128:2251-2254.
7. Ezzahti M, Moelker A, Friesema E, et al. Blood pressure and neurohormonal responses to renal nerve ablation in treatment-resistant hypertension. J Hypertens. 2014;32(1):135-141.
8. Calhoun DA, Jones D, Textor S, et al. Resistant hypertension: Diagnosis, evaluation, and treatment: A scientific statement from the American Heart Association Professional Education Committee of the Council for High Blood Pressure Research. Hypertension. 2008;51(6):1403-1419.
9. Daugherty SL, Powers JD, Magid DJ, et al. Incidence and prognosis of resistant hypertension in hypertensive patients. Circulation. 2012;125(13):1635-1642.
10. Bhatt DL, Kandzari DE, O’Neill WW, et al; Symplicity HTN-3 Investigators. A controlled trial of renal denervation for resistant hypertension. N Engl J Med. 2014;370(15):1393-1401.
The author would like to thank Eric Judd, MD, of the University of Alabama at Birmingham, for his advice on the preparation of this response.
Q) I’ve heard a lot of references to “renal denervation” and its use for resistant hypertension. What is it? Does it work? Is it common in the US?
Renal denervation is a minimally invasive endovascular procedure that ablates (or disrupts) the renal nerves in and around the renal arteries with radiofrequency energy.5 Renal denervation has been approved in the US and other countries and is being used clinically in Europe, Canada, and Australia.6
It is thought that renal denervation interrupts the efferent and afferent signals that stimulate the renin-angiotensin-aldosterone system (RAAS) and regulate whole-body sympathetic nervous system activity.5 Similar to surgical sympathectomy, renal denervation should theoretically lower blood pressure. However, Ezzahti et al found that renin levels did not decrease in patients following renal denervation.7
Drug-resistant hypertension is defined as blood pressure that remains greater than 140/90 mm Hg despite treatment with three or more antihypertensive medications, including a diuretic.8 Patients with resistant hypertension have increased cardiovascular risk.9 Clinical trials of renal denervation have focused on treatment of resistant hypertension, in the hope of reducing the associated morbidity and mortality.
Results of the Symplicity HTN-3 trial, which assessed the safety and efficacy of renal denervation, were anxiously awaited, since prior trials yielded mixed results. Although the Symplicity HTN-1 and Symplicity HTN-2 studies demonstrated a possible benefit of renal denervation to lower office measured blood pressure, other studies did not show a decrease in BP in patients who had undergone renal denervation.6,7 These early trials, however, were small and did not randomize patients to a sham procedure.10
The Symplicity HTN-3 trial included 535 patients at 88 centers in the US. Patients were randomly assigned to receive either renal denervation plus baseline antihypertensive medications or a sham procedure plus baseline antihypertensive medications.
The researchers found that the sham procedure was just as effective as the “true” renal denervation in decreasing systolic blood pressure in patients with resistant hypertension.10 In other words, renal denervation did not demonstrate efficacy for this purpose.
In response to the results of this well-designed trial, the FDA has halted approval to perform renal denervation in patients with resistant hypertension in the US. However, clinical investigation will continue among subgroups of hypertensive patients or separate populations.
Despite a lack of efficacy, renal denervation does appear to be well tolerated, as evidenced by safety data from Symplicity HTN-3. —JK
Jessica Knight, ACNP
University of New Mexico Hospital, Albuquerque
REFERENCES
5. Esler MD, Krum H, Schlaich M, et al. Renal sympathetic denervation for the treatment of drug-resistant hypertension: one-year results from the Symplicity HTN-2 randomized, controlled trial. Circulation. 2012;126(25):2976-2982.
6. Thukkani AK, Bhatt LD. Renal denervation therapy for hypertension. Circulation. 2013;128:2251-2254.
7. Ezzahti M, Moelker A, Friesema E, et al. Blood pressure and neurohormonal responses to renal nerve ablation in treatment-resistant hypertension. J Hypertens. 2014;32(1):135-141.
8. Calhoun DA, Jones D, Textor S, et al. Resistant hypertension: Diagnosis, evaluation, and treatment: A scientific statement from the American Heart Association Professional Education Committee of the Council for High Blood Pressure Research. Hypertension. 2008;51(6):1403-1419.
9. Daugherty SL, Powers JD, Magid DJ, et al. Incidence and prognosis of resistant hypertension in hypertensive patients. Circulation. 2012;125(13):1635-1642.
10. Bhatt DL, Kandzari DE, O’Neill WW, et al; Symplicity HTN-3 Investigators. A controlled trial of renal denervation for resistant hypertension. N Engl J Med. 2014;370(15):1393-1401.
The author would like to thank Eric Judd, MD, of the University of Alabama at Birmingham, for his advice on the preparation of this response.
Q) I’ve heard a lot of references to “renal denervation” and its use for resistant hypertension. What is it? Does it work? Is it common in the US?
Renal denervation is a minimally invasive endovascular procedure that ablates (or disrupts) the renal nerves in and around the renal arteries with radiofrequency energy.5 Renal denervation has been approved in the US and other countries and is being used clinically in Europe, Canada, and Australia.6
It is thought that renal denervation interrupts the efferent and afferent signals that stimulate the renin-angiotensin-aldosterone system (RAAS) and regulate whole-body sympathetic nervous system activity.5 Similar to surgical sympathectomy, renal denervation should theoretically lower blood pressure. However, Ezzahti et al found that renin levels did not decrease in patients following renal denervation.7
Drug-resistant hypertension is defined as blood pressure that remains greater than 140/90 mm Hg despite treatment with three or more antihypertensive medications, including a diuretic.8 Patients with resistant hypertension have increased cardiovascular risk.9 Clinical trials of renal denervation have focused on treatment of resistant hypertension, in the hope of reducing the associated morbidity and mortality.
Results of the Symplicity HTN-3 trial, which assessed the safety and efficacy of renal denervation, were anxiously awaited, since prior trials yielded mixed results. Although the Symplicity HTN-1 and Symplicity HTN-2 studies demonstrated a possible benefit of renal denervation to lower office measured blood pressure, other studies did not show a decrease in BP in patients who had undergone renal denervation.6,7 These early trials, however, were small and did not randomize patients to a sham procedure.10
The Symplicity HTN-3 trial included 535 patients at 88 centers in the US. Patients were randomly assigned to receive either renal denervation plus baseline antihypertensive medications or a sham procedure plus baseline antihypertensive medications.
The researchers found that the sham procedure was just as effective as the “true” renal denervation in decreasing systolic blood pressure in patients with resistant hypertension.10 In other words, renal denervation did not demonstrate efficacy for this purpose.
In response to the results of this well-designed trial, the FDA has halted approval to perform renal denervation in patients with resistant hypertension in the US. However, clinical investigation will continue among subgroups of hypertensive patients or separate populations.
Despite a lack of efficacy, renal denervation does appear to be well tolerated, as evidenced by safety data from Symplicity HTN-3. —JK
Jessica Knight, ACNP
University of New Mexico Hospital, Albuquerque
REFERENCES
5. Esler MD, Krum H, Schlaich M, et al. Renal sympathetic denervation for the treatment of drug-resistant hypertension: one-year results from the Symplicity HTN-2 randomized, controlled trial. Circulation. 2012;126(25):2976-2982.
6. Thukkani AK, Bhatt LD. Renal denervation therapy for hypertension. Circulation. 2013;128:2251-2254.
7. Ezzahti M, Moelker A, Friesema E, et al. Blood pressure and neurohormonal responses to renal nerve ablation in treatment-resistant hypertension. J Hypertens. 2014;32(1):135-141.
8. Calhoun DA, Jones D, Textor S, et al. Resistant hypertension: Diagnosis, evaluation, and treatment: A scientific statement from the American Heart Association Professional Education Committee of the Council for High Blood Pressure Research. Hypertension. 2008;51(6):1403-1419.
9. Daugherty SL, Powers JD, Magid DJ, et al. Incidence and prognosis of resistant hypertension in hypertensive patients. Circulation. 2012;125(13):1635-1642.
10. Bhatt DL, Kandzari DE, O’Neill WW, et al; Symplicity HTN-3 Investigators. A controlled trial of renal denervation for resistant hypertension. N Engl J Med. 2014;370(15):1393-1401.
The author would like to thank Eric Judd, MD, of the University of Alabama at Birmingham, for his advice on the preparation of this response.
Revenge of the Deer Stand
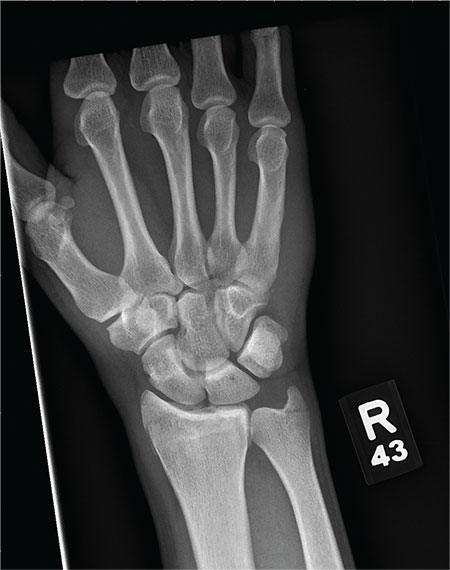
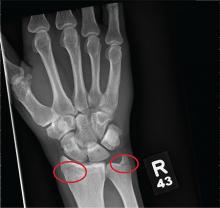
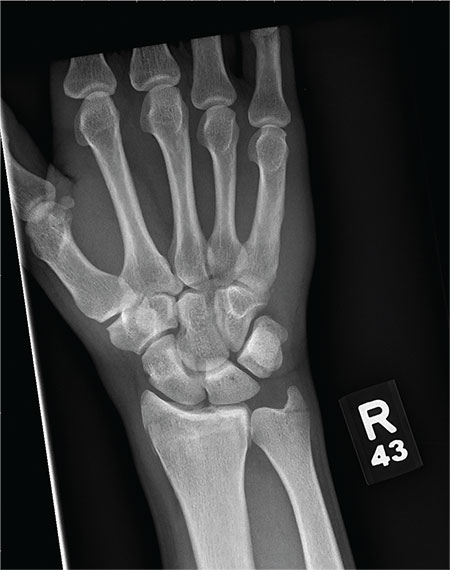
As you begin your shift, knowing deer season has started in your area, you wonder how long it will be before you see your first deer stand casualty of the day. With your first patient, you wonder no more: A 30-year-old man presents for evaluation of right wrist pain after falling from his deer stand. He says one of the straps holding the stand broke, causing him to fall forward and land on his outstretched hands. His medical history is unremarkable. Inspection of the right wrist shows no obvious deformity. No significant swelling is present. There is decreased range of motion and localized tenderness over the radius and ulna. Good pulses and capillary refill are noted. You obtain a radiograph of the wrist. What is your impression?
Time or content?
In May 2015, the American Academy of Pediatrics convened an invitation-only symposium titled Growing Up Digital. Its goal was to reconsider the Academy’s advice on “screen time” and make sure that its policies were “science-driven, not based merely on the precautionary principle.” (“Beyond ‘turn it off’: How to advice families on media use,” Brown et al. AAP News, October 2015). Driven by the concern that the current AAP advice was becoming obsolete and as a result likely to be ignored by parents faced with the realities of our digital culture, the participants investigated the available data on “early learning, game-based learning, social/emotional and developmental concerns, and strategies to foster digital citizenship.”
Their findings have been distilled into a collection of “key messages” for parents published in the October, 2015 AAP News. It’s hard to argue with most of the common sense advice that includes “Role modeling is critical; playtime is important; co-engagement counts; set limits; and create tech-free zones.” A set of formal recommendations is in the works and will be published at a later date.
It is comforting to learn of the academy’s concern to keep its advice current and evidence-based. It is frustrating for those of us expected to deliver the party line when we suspect that parents are muttering to themselves, “Really?” I assume that most pediatricians at the parent/doctor interface will join me in welcoming much of the more nuanced advice in the final recommendations, particularly those for older children and adolescents.
However, if the new document is not carefully worded and promoted, I fear that the potent message of “no screen time under age 2” will be lost or diluted. While the symposium participants may have uncovered some evidence of benefit or at least no serious harm from some digital platforms, does this warrant softening the catchy and clear advice of “no screen time under 2?” I have to ask myself when would a child under the age of 2 being raised in a healthy environment have time for electronic distraction?
As Dr. Ari Brown, Dr. Donald L. Shifrin, and Dr. David L. Hill ask parents in their AAP News piece, “Does your child’s technology use help or hinder participation in other activities?” Just doing a little quick math: Wake up at 7 a.m., breakfast, playground time, maybe a midmorning nap, snack, lunch, afternoon nap, afternoon playground time, maybe another snack, dinner, bedtime story and lights out at 7 p.m. I don’t see a spot to shoehorn in some screen time without eliminating a developmentally and socially important activity. You could replace the hard cover book at bedtime with an electronic one on a tablet, but in my experience that runs the risk of replacing a soporific activity with one that is too visually stimulating.
One could argue that depriving a young child of screen time is going to put him behind his peers who have become masterful web navigators by the time they are 18 months. Rubbish. The learning curve for most electronic devices is so short that the “deprived” child will catch up in a couple of dozen clicks. However, screens require little more than a moving and tapping index finger. What about those other manipulative skills and the strength and coordination of the muscles sitting unused during screen time?
Unfortunately, the crafters of these new guidelines have repeated the same mistake the academy has made before when they observe, “The quality of the content is more important than the platform or time spent with media.” In my opinion, if the time spent on a screen is kept sufficiently short, children won’t squander it on bad stuff for very long nor will what they see be that harmful. Burdening parents with the task of determining quality is unrealistic. However, setting a time limit is far more workable and enforceable.
Finally, when it comes to parents enforcing no screen time under 2, everyone knows that Skyping with Grandma and Grandpa gets a free pass.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “Coping With a Picky Eater.” Email him at pdnews@frontlinemedcom.com.
In May 2015, the American Academy of Pediatrics convened an invitation-only symposium titled Growing Up Digital. Its goal was to reconsider the Academy’s advice on “screen time” and make sure that its policies were “science-driven, not based merely on the precautionary principle.” (“Beyond ‘turn it off’: How to advice families on media use,” Brown et al. AAP News, October 2015). Driven by the concern that the current AAP advice was becoming obsolete and as a result likely to be ignored by parents faced with the realities of our digital culture, the participants investigated the available data on “early learning, game-based learning, social/emotional and developmental concerns, and strategies to foster digital citizenship.”
Their findings have been distilled into a collection of “key messages” for parents published in the October, 2015 AAP News. It’s hard to argue with most of the common sense advice that includes “Role modeling is critical; playtime is important; co-engagement counts; set limits; and create tech-free zones.” A set of formal recommendations is in the works and will be published at a later date.
It is comforting to learn of the academy’s concern to keep its advice current and evidence-based. It is frustrating for those of us expected to deliver the party line when we suspect that parents are muttering to themselves, “Really?” I assume that most pediatricians at the parent/doctor interface will join me in welcoming much of the more nuanced advice in the final recommendations, particularly those for older children and adolescents.
However, if the new document is not carefully worded and promoted, I fear that the potent message of “no screen time under age 2” will be lost or diluted. While the symposium participants may have uncovered some evidence of benefit or at least no serious harm from some digital platforms, does this warrant softening the catchy and clear advice of “no screen time under 2?” I have to ask myself when would a child under the age of 2 being raised in a healthy environment have time for electronic distraction?
As Dr. Ari Brown, Dr. Donald L. Shifrin, and Dr. David L. Hill ask parents in their AAP News piece, “Does your child’s technology use help or hinder participation in other activities?” Just doing a little quick math: Wake up at 7 a.m., breakfast, playground time, maybe a midmorning nap, snack, lunch, afternoon nap, afternoon playground time, maybe another snack, dinner, bedtime story and lights out at 7 p.m. I don’t see a spot to shoehorn in some screen time without eliminating a developmentally and socially important activity. You could replace the hard cover book at bedtime with an electronic one on a tablet, but in my experience that runs the risk of replacing a soporific activity with one that is too visually stimulating.
One could argue that depriving a young child of screen time is going to put him behind his peers who have become masterful web navigators by the time they are 18 months. Rubbish. The learning curve for most electronic devices is so short that the “deprived” child will catch up in a couple of dozen clicks. However, screens require little more than a moving and tapping index finger. What about those other manipulative skills and the strength and coordination of the muscles sitting unused during screen time?
Unfortunately, the crafters of these new guidelines have repeated the same mistake the academy has made before when they observe, “The quality of the content is more important than the platform or time spent with media.” In my opinion, if the time spent on a screen is kept sufficiently short, children won’t squander it on bad stuff for very long nor will what they see be that harmful. Burdening parents with the task of determining quality is unrealistic. However, setting a time limit is far more workable and enforceable.
Finally, when it comes to parents enforcing no screen time under 2, everyone knows that Skyping with Grandma and Grandpa gets a free pass.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “Coping With a Picky Eater.” Email him at pdnews@frontlinemedcom.com.
In May 2015, the American Academy of Pediatrics convened an invitation-only symposium titled Growing Up Digital. Its goal was to reconsider the Academy’s advice on “screen time” and make sure that its policies were “science-driven, not based merely on the precautionary principle.” (“Beyond ‘turn it off’: How to advice families on media use,” Brown et al. AAP News, October 2015). Driven by the concern that the current AAP advice was becoming obsolete and as a result likely to be ignored by parents faced with the realities of our digital culture, the participants investigated the available data on “early learning, game-based learning, social/emotional and developmental concerns, and strategies to foster digital citizenship.”
Their findings have been distilled into a collection of “key messages” for parents published in the October, 2015 AAP News. It’s hard to argue with most of the common sense advice that includes “Role modeling is critical; playtime is important; co-engagement counts; set limits; and create tech-free zones.” A set of formal recommendations is in the works and will be published at a later date.
It is comforting to learn of the academy’s concern to keep its advice current and evidence-based. It is frustrating for those of us expected to deliver the party line when we suspect that parents are muttering to themselves, “Really?” I assume that most pediatricians at the parent/doctor interface will join me in welcoming much of the more nuanced advice in the final recommendations, particularly those for older children and adolescents.
However, if the new document is not carefully worded and promoted, I fear that the potent message of “no screen time under age 2” will be lost or diluted. While the symposium participants may have uncovered some evidence of benefit or at least no serious harm from some digital platforms, does this warrant softening the catchy and clear advice of “no screen time under 2?” I have to ask myself when would a child under the age of 2 being raised in a healthy environment have time for electronic distraction?
As Dr. Ari Brown, Dr. Donald L. Shifrin, and Dr. David L. Hill ask parents in their AAP News piece, “Does your child’s technology use help or hinder participation in other activities?” Just doing a little quick math: Wake up at 7 a.m., breakfast, playground time, maybe a midmorning nap, snack, lunch, afternoon nap, afternoon playground time, maybe another snack, dinner, bedtime story and lights out at 7 p.m. I don’t see a spot to shoehorn in some screen time without eliminating a developmentally and socially important activity. You could replace the hard cover book at bedtime with an electronic one on a tablet, but in my experience that runs the risk of replacing a soporific activity with one that is too visually stimulating.
One could argue that depriving a young child of screen time is going to put him behind his peers who have become masterful web navigators by the time they are 18 months. Rubbish. The learning curve for most electronic devices is so short that the “deprived” child will catch up in a couple of dozen clicks. However, screens require little more than a moving and tapping index finger. What about those other manipulative skills and the strength and coordination of the muscles sitting unused during screen time?
Unfortunately, the crafters of these new guidelines have repeated the same mistake the academy has made before when they observe, “The quality of the content is more important than the platform or time spent with media.” In my opinion, if the time spent on a screen is kept sufficiently short, children won’t squander it on bad stuff for very long nor will what they see be that harmful. Burdening parents with the task of determining quality is unrealistic. However, setting a time limit is far more workable and enforceable.
Finally, when it comes to parents enforcing no screen time under 2, everyone knows that Skyping with Grandma and Grandpa gets a free pass.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “Coping With a Picky Eater.” Email him at pdnews@frontlinemedcom.com.
ITC: Levothyroxine may improve obstetric outcomes in women with low thyroid function
LAKE BUENA VISTA, FLA. – Normalizing maternal thyroid function early in pregnancy may improve obstetric outcomes for women with subclinical hypothyroidism.
A high level of thyroid-stimulating hormone (TSH) was associated with increased stillbirth in women who didn’t receive prenatal levothyroxine, Peter N. Taylor, Ph.D., reported at the International Thyroid Congress.
And women who were untreated for low free thyroxine (T4) were significantly more likely to need an early cesarean section than treated women, suggesting that levothyroxine could be reducing conditions leading to maternal-fetal distress.
“The good thing is that many physicians are already treating with levothyroxine anyway,” said Dr. Taylor of Cardiff (Wales) University. “There are no obvious adverse obstetric events associated with it. It seems relatively benign, and the accepted dose of 150 mcg per day seems adequate.”
He reported data from a subgroup analysis of the large Controlled Antenatal Thyroid Screening Study (N Eng J Med. 2012;366:493-501). That trial enrolled more than 21,000 mother-child pairs, and examined the effect of maternal levothyroxine treatment on the child’s IQ at 3 years. Among the children of women with hypothyroidism, the mean IQ scores were 99.2 and 100 in the treated and control groups – not significantly different. The proportions of children with an IQ of less than 85 were 12% in the treated group and 14% in the control group, also not significantly different.
Dr. Taylor’s cohort involved about 14,000 mother-child pairs from this study, all of whom were from Wales. Using national health care databases as well as study data, he was able to examine obstetric outcomes in women with low thyroid function (664) who received or did not receive levothyroxine early between 11 and 16 weeks’ gestation.
About half of the hypothyroid women (351) had TSH levels above 3.5 mU/L. The remainder had free T4 levels below 10.9 pmol/L. Each of these groups was randomized to levothyroxine treatment or placebo. The rest of the cohort had normal thyroid status.
The study’s primary endpoint was the rate of stillbirth. Secondary endpoints were cesarean section rate (both overall and early); gestational age at birth; and macrosomia.
Among the euthyroid group, the rate of stillbirth was 0.34%, similar to the national background rate. There were no stillbirths among women with high TSH who were treated. Three (1.68%) occurred in the untreated group. The TSH levels in those women before treatment were 3.63 mU/L, 3.66 mU/L, and 4.58 mU/L – not dramatically high, Dr. Taylor noted. “But they could have risen later in pregnancy as stress on the thyroid increased.”
After adjusting for maternal age, weight, parity, birth year, and smoking, stillbirths were five times more likely among the untreated women than the treated women. However, Dr. Taylor cautioned, “This is a very small number of events. But it is quite seductive to think that stillbirths could be prevented by levothyroxine.”
There were no significant associations of high TSH with macrosomia or gestational age.
Untreated low free T4 was not associated with stillbirth. However, Dr. Taylor said, it was very strongly associated with early C-section.
The overall C-section rate was similar between untreated and treated women (28%). But 5.6% of the untreated women had an early C-section, compared with none of the treated women. This hints strongly at a protective effect of levothyroxine, Dr. Taylor said. Early C-sections – between 26 and 32 weeks – are medically driven rather than driven by patient choice. This finding of fewer early interventions among treated women suggests that levothyroxine is exerting some protective effect, especially given the finding that infants of untreated mothers actually tended to be about 133 g lighter at birth.
“We would speculate this is probably due to a decrease in preeclampsia and gestational hypertension, which are more common among women with hypothyroidism.”
Infants of untreated mothers also were born slightly earlier – about half a week, he said at the meeting held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society.
Dr. Taylor had no financial disclosures.
LAKE BUENA VISTA, FLA. – Normalizing maternal thyroid function early in pregnancy may improve obstetric outcomes for women with subclinical hypothyroidism.
A high level of thyroid-stimulating hormone (TSH) was associated with increased stillbirth in women who didn’t receive prenatal levothyroxine, Peter N. Taylor, Ph.D., reported at the International Thyroid Congress.
And women who were untreated for low free thyroxine (T4) were significantly more likely to need an early cesarean section than treated women, suggesting that levothyroxine could be reducing conditions leading to maternal-fetal distress.
“The good thing is that many physicians are already treating with levothyroxine anyway,” said Dr. Taylor of Cardiff (Wales) University. “There are no obvious adverse obstetric events associated with it. It seems relatively benign, and the accepted dose of 150 mcg per day seems adequate.”
He reported data from a subgroup analysis of the large Controlled Antenatal Thyroid Screening Study (N Eng J Med. 2012;366:493-501). That trial enrolled more than 21,000 mother-child pairs, and examined the effect of maternal levothyroxine treatment on the child’s IQ at 3 years. Among the children of women with hypothyroidism, the mean IQ scores were 99.2 and 100 in the treated and control groups – not significantly different. The proportions of children with an IQ of less than 85 were 12% in the treated group and 14% in the control group, also not significantly different.
Dr. Taylor’s cohort involved about 14,000 mother-child pairs from this study, all of whom were from Wales. Using national health care databases as well as study data, he was able to examine obstetric outcomes in women with low thyroid function (664) who received or did not receive levothyroxine early between 11 and 16 weeks’ gestation.
About half of the hypothyroid women (351) had TSH levels above 3.5 mU/L. The remainder had free T4 levels below 10.9 pmol/L. Each of these groups was randomized to levothyroxine treatment or placebo. The rest of the cohort had normal thyroid status.
The study’s primary endpoint was the rate of stillbirth. Secondary endpoints were cesarean section rate (both overall and early); gestational age at birth; and macrosomia.
Among the euthyroid group, the rate of stillbirth was 0.34%, similar to the national background rate. There were no stillbirths among women with high TSH who were treated. Three (1.68%) occurred in the untreated group. The TSH levels in those women before treatment were 3.63 mU/L, 3.66 mU/L, and 4.58 mU/L – not dramatically high, Dr. Taylor noted. “But they could have risen later in pregnancy as stress on the thyroid increased.”
After adjusting for maternal age, weight, parity, birth year, and smoking, stillbirths were five times more likely among the untreated women than the treated women. However, Dr. Taylor cautioned, “This is a very small number of events. But it is quite seductive to think that stillbirths could be prevented by levothyroxine.”
There were no significant associations of high TSH with macrosomia or gestational age.
Untreated low free T4 was not associated with stillbirth. However, Dr. Taylor said, it was very strongly associated with early C-section.
The overall C-section rate was similar between untreated and treated women (28%). But 5.6% of the untreated women had an early C-section, compared with none of the treated women. This hints strongly at a protective effect of levothyroxine, Dr. Taylor said. Early C-sections – between 26 and 32 weeks – are medically driven rather than driven by patient choice. This finding of fewer early interventions among treated women suggests that levothyroxine is exerting some protective effect, especially given the finding that infants of untreated mothers actually tended to be about 133 g lighter at birth.
“We would speculate this is probably due to a decrease in preeclampsia and gestational hypertension, which are more common among women with hypothyroidism.”
Infants of untreated mothers also were born slightly earlier – about half a week, he said at the meeting held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society.
Dr. Taylor had no financial disclosures.
LAKE BUENA VISTA, FLA. – Normalizing maternal thyroid function early in pregnancy may improve obstetric outcomes for women with subclinical hypothyroidism.
A high level of thyroid-stimulating hormone (TSH) was associated with increased stillbirth in women who didn’t receive prenatal levothyroxine, Peter N. Taylor, Ph.D., reported at the International Thyroid Congress.
And women who were untreated for low free thyroxine (T4) were significantly more likely to need an early cesarean section than treated women, suggesting that levothyroxine could be reducing conditions leading to maternal-fetal distress.
“The good thing is that many physicians are already treating with levothyroxine anyway,” said Dr. Taylor of Cardiff (Wales) University. “There are no obvious adverse obstetric events associated with it. It seems relatively benign, and the accepted dose of 150 mcg per day seems adequate.”
He reported data from a subgroup analysis of the large Controlled Antenatal Thyroid Screening Study (N Eng J Med. 2012;366:493-501). That trial enrolled more than 21,000 mother-child pairs, and examined the effect of maternal levothyroxine treatment on the child’s IQ at 3 years. Among the children of women with hypothyroidism, the mean IQ scores were 99.2 and 100 in the treated and control groups – not significantly different. The proportions of children with an IQ of less than 85 were 12% in the treated group and 14% in the control group, also not significantly different.
Dr. Taylor’s cohort involved about 14,000 mother-child pairs from this study, all of whom were from Wales. Using national health care databases as well as study data, he was able to examine obstetric outcomes in women with low thyroid function (664) who received or did not receive levothyroxine early between 11 and 16 weeks’ gestation.
About half of the hypothyroid women (351) had TSH levels above 3.5 mU/L. The remainder had free T4 levels below 10.9 pmol/L. Each of these groups was randomized to levothyroxine treatment or placebo. The rest of the cohort had normal thyroid status.
The study’s primary endpoint was the rate of stillbirth. Secondary endpoints were cesarean section rate (both overall and early); gestational age at birth; and macrosomia.
Among the euthyroid group, the rate of stillbirth was 0.34%, similar to the national background rate. There were no stillbirths among women with high TSH who were treated. Three (1.68%) occurred in the untreated group. The TSH levels in those women before treatment were 3.63 mU/L, 3.66 mU/L, and 4.58 mU/L – not dramatically high, Dr. Taylor noted. “But they could have risen later in pregnancy as stress on the thyroid increased.”
After adjusting for maternal age, weight, parity, birth year, and smoking, stillbirths were five times more likely among the untreated women than the treated women. However, Dr. Taylor cautioned, “This is a very small number of events. But it is quite seductive to think that stillbirths could be prevented by levothyroxine.”
There were no significant associations of high TSH with macrosomia or gestational age.
Untreated low free T4 was not associated with stillbirth. However, Dr. Taylor said, it was very strongly associated with early C-section.
The overall C-section rate was similar between untreated and treated women (28%). But 5.6% of the untreated women had an early C-section, compared with none of the treated women. This hints strongly at a protective effect of levothyroxine, Dr. Taylor said. Early C-sections – between 26 and 32 weeks – are medically driven rather than driven by patient choice. This finding of fewer early interventions among treated women suggests that levothyroxine is exerting some protective effect, especially given the finding that infants of untreated mothers actually tended to be about 133 g lighter at birth.
“We would speculate this is probably due to a decrease in preeclampsia and gestational hypertension, which are more common among women with hypothyroidism.”
Infants of untreated mothers also were born slightly earlier – about half a week, he said at the meeting held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society.
Dr. Taylor had no financial disclosures.
AT ITC 2015
Key clinical point: Levothyroxine may improve obstetric outcomes in women with low thyroid function.
Major finding: Stillbirths were five times more likely among women who were untreated for high levels of thyroid-stimulating hormone during pregnancy.
Data source: The analysis comprised about 14,000 mother-child pairs.
Disclosures: Dr. Taylor had no financial disclosures.
Predictive Factors for CKD
Q) Quite a few of my teenage patients are overweight. I know they are at risk for diabetes, but does their weight also affect their kidneys? Isn’t diabetes the main cause of kidney failure?
The number one cause of chronic kidney disease (CKD) in the United States and worldwide is diabetes, but it is certainly not the only risk factor. Studies have shown a link between obesity and CKD; even in the absence of kidney disease, obesity may cause glomerular dysfunction and an increase in glomerular size.1
Obesity during adolescence has been identified as a strong predictor of CKD in adulthood. Other diseases and conditions that, if present in adolescence, indicate future risk for kidney disease include diabetes, hypertension, inflammation, and proteinuria.
A recent Swedish study followed patients from adolescence to adulthood to identify markers that would predict later kidney disease. In this study, the most predictive factor of kidney failure in adulthood was proteinuria in adolescence (odds ratio, 7.72). These results may be limited by the homogeneity of the predominantly white, male study population, but the extensive follow-up period, which “highlights the long natural history” of kidney disease, is one strength of this study.2
Based on these and other findings, you know that if your teenage patients have proteinuria, they are much more likely to develop kidney failure as an adult. Yet, in the US, the American Academy of Pediatrics and the US Preventive Services Task Force do not recommend urine screening for asymptomatic children.3
Interestingly, however, a survey of pediatric practices revealed that 58% of pediatricians screen adolescents with urinalysis, even if they are asymptomatic.4 In other words, they ignore the guidelines. If they did not, we would likely miss what is possibly the most important predictive factor for kidney failure in adults. —TAH
Tia Austin Hayes, FNP-C
UMMC/JMM Outpatient Dialysis/Renal Clinic, Jackson, Mississippi
REFERENCES
1. Rocchini A. Childhood obesity and a diabetes epidemic. N Engl J Med. 2002;346(11):854-855.
2. Sundin PO, Udumyan R, Sjöström P, Montgomery S. Predictors in adolescence of ESRD in middle-aged men. Am J Kidney Dis. 2014;64(5):723-729.
3. Kaplan RE, Springate JE, Feld LG. Screening dipstick urinalysis: a time to change. Pediatrics. 1997;100(6):919-921.
4. Sox CM, Christakis DA. Pediatricians’ screening urinalysis practices. J Pediatr. 2005; 147(3):362-365.
The author would like to thank Eric Judd, MD, of the University of Alabama at Birmingham, for his advice on the preparation of this response.
Q) Quite a few of my teenage patients are overweight. I know they are at risk for diabetes, but does their weight also affect their kidneys? Isn’t diabetes the main cause of kidney failure?
The number one cause of chronic kidney disease (CKD) in the United States and worldwide is diabetes, but it is certainly not the only risk factor. Studies have shown a link between obesity and CKD; even in the absence of kidney disease, obesity may cause glomerular dysfunction and an increase in glomerular size.1
Obesity during adolescence has been identified as a strong predictor of CKD in adulthood. Other diseases and conditions that, if present in adolescence, indicate future risk for kidney disease include diabetes, hypertension, inflammation, and proteinuria.
A recent Swedish study followed patients from adolescence to adulthood to identify markers that would predict later kidney disease. In this study, the most predictive factor of kidney failure in adulthood was proteinuria in adolescence (odds ratio, 7.72). These results may be limited by the homogeneity of the predominantly white, male study population, but the extensive follow-up period, which “highlights the long natural history” of kidney disease, is one strength of this study.2
Based on these and other findings, you know that if your teenage patients have proteinuria, they are much more likely to develop kidney failure as an adult. Yet, in the US, the American Academy of Pediatrics and the US Preventive Services Task Force do not recommend urine screening for asymptomatic children.3
Interestingly, however, a survey of pediatric practices revealed that 58% of pediatricians screen adolescents with urinalysis, even if they are asymptomatic.4 In other words, they ignore the guidelines. If they did not, we would likely miss what is possibly the most important predictive factor for kidney failure in adults. —TAH
Tia Austin Hayes, FNP-C
UMMC/JMM Outpatient Dialysis/Renal Clinic, Jackson, Mississippi
REFERENCES
1. Rocchini A. Childhood obesity and a diabetes epidemic. N Engl J Med. 2002;346(11):854-855.
2. Sundin PO, Udumyan R, Sjöström P, Montgomery S. Predictors in adolescence of ESRD in middle-aged men. Am J Kidney Dis. 2014;64(5):723-729.
3. Kaplan RE, Springate JE, Feld LG. Screening dipstick urinalysis: a time to change. Pediatrics. 1997;100(6):919-921.
4. Sox CM, Christakis DA. Pediatricians’ screening urinalysis practices. J Pediatr. 2005; 147(3):362-365.
The author would like to thank Eric Judd, MD, of the University of Alabama at Birmingham, for his advice on the preparation of this response.
Q) Quite a few of my teenage patients are overweight. I know they are at risk for diabetes, but does their weight also affect their kidneys? Isn’t diabetes the main cause of kidney failure?
The number one cause of chronic kidney disease (CKD) in the United States and worldwide is diabetes, but it is certainly not the only risk factor. Studies have shown a link between obesity and CKD; even in the absence of kidney disease, obesity may cause glomerular dysfunction and an increase in glomerular size.1
Obesity during adolescence has been identified as a strong predictor of CKD in adulthood. Other diseases and conditions that, if present in adolescence, indicate future risk for kidney disease include diabetes, hypertension, inflammation, and proteinuria.
A recent Swedish study followed patients from adolescence to adulthood to identify markers that would predict later kidney disease. In this study, the most predictive factor of kidney failure in adulthood was proteinuria in adolescence (odds ratio, 7.72). These results may be limited by the homogeneity of the predominantly white, male study population, but the extensive follow-up period, which “highlights the long natural history” of kidney disease, is one strength of this study.2
Based on these and other findings, you know that if your teenage patients have proteinuria, they are much more likely to develop kidney failure as an adult. Yet, in the US, the American Academy of Pediatrics and the US Preventive Services Task Force do not recommend urine screening for asymptomatic children.3
Interestingly, however, a survey of pediatric practices revealed that 58% of pediatricians screen adolescents with urinalysis, even if they are asymptomatic.4 In other words, they ignore the guidelines. If they did not, we would likely miss what is possibly the most important predictive factor for kidney failure in adults. —TAH
Tia Austin Hayes, FNP-C
UMMC/JMM Outpatient Dialysis/Renal Clinic, Jackson, Mississippi
REFERENCES
1. Rocchini A. Childhood obesity and a diabetes epidemic. N Engl J Med. 2002;346(11):854-855.
2. Sundin PO, Udumyan R, Sjöström P, Montgomery S. Predictors in adolescence of ESRD in middle-aged men. Am J Kidney Dis. 2014;64(5):723-729.
3. Kaplan RE, Springate JE, Feld LG. Screening dipstick urinalysis: a time to change. Pediatrics. 1997;100(6):919-921.
4. Sox CM, Christakis DA. Pediatricians’ screening urinalysis practices. J Pediatr. 2005; 147(3):362-365.
The author would like to thank Eric Judd, MD, of the University of Alabama at Birmingham, for his advice on the preparation of this response.
“Spry” Woman Reports Rapid Heart Rate
ANSWER
The correct interpretation includes atrial fibrillation with a rapid ventricular response and aberrantly conducted complexes, left axis deviation, and a left bundle branch block.
Atrial fibrillation is evidenced by the irregularly irregular heart rhythm without a measurable PR interval, and the rapid ventricular response is indicated by a ventricular rate > 100 beats/min.
Aberrant conduction, caused by conduction delay down the His-Purkinje system, is evidenced by the wide QRS complexes with a normally conducted beat (see first beat in leads V1-V3). Criteria for left axis deviation include an R axis between –30° and –90°, and left bundle branch block criteria include a QRS duration > 120 ms, a dominant S wave in V1, and broad monophasic R waves in leads I, aVL, and V5-V6.
ANSWER
The correct interpretation includes atrial fibrillation with a rapid ventricular response and aberrantly conducted complexes, left axis deviation, and a left bundle branch block.
Atrial fibrillation is evidenced by the irregularly irregular heart rhythm without a measurable PR interval, and the rapid ventricular response is indicated by a ventricular rate > 100 beats/min.
Aberrant conduction, caused by conduction delay down the His-Purkinje system, is evidenced by the wide QRS complexes with a normally conducted beat (see first beat in leads V1-V3). Criteria for left axis deviation include an R axis between –30° and –90°, and left bundle branch block criteria include a QRS duration > 120 ms, a dominant S wave in V1, and broad monophasic R waves in leads I, aVL, and V5-V6.
ANSWER
The correct interpretation includes atrial fibrillation with a rapid ventricular response and aberrantly conducted complexes, left axis deviation, and a left bundle branch block.
Atrial fibrillation is evidenced by the irregularly irregular heart rhythm without a measurable PR interval, and the rapid ventricular response is indicated by a ventricular rate > 100 beats/min.
Aberrant conduction, caused by conduction delay down the His-Purkinje system, is evidenced by the wide QRS complexes with a normally conducted beat (see first beat in leads V1-V3). Criteria for left axis deviation include an R axis between –30° and –90°, and left bundle branch block criteria include a QRS duration > 120 ms, a dominant S wave in V1, and broad monophasic R waves in leads I, aVL, and V5-V6.
An 84-year-old woman who recently relocated to be closer to her children presents to your practice as a new patient. She is a resident of an assisted living facility near your office, and although she has no specific complaints, she does report that her home health nurse observed a rapid heart rate and recommended she get it checked. A comprehensive medical history—provided by the patient, her daughter, and the aforementioned nurse—includes hypertension, paroxysmal atrial fibrillation, hypothyroidism, and type 2 diabetes. She has taken medication for these diagnoses for more than 30 years. Surgical history is remarkable for cholecystectomy, appendectomy, and abdominal hysterectomy and oophorectomy, all of which were performed in the 1970s. Her current medication list—confirmed by the assisted living facility—includes furosemide, glyburide, metoprolol, potassium, and levothyroxine. She has not missed any doses. She is allergic to sulfa. The patient, a retired teacher, has never smoked, but she does “enjoy” one martini at dinner on a regular basis. She is widowed; her two daughters and four sons are all alive and well. The review of systems is remarkable for corrective lenses, bilateral hearing aids, and chronic joint pain. The patient does not routinely weigh herself but thinks, based on the fit of her clothes, that she may have gained some weight. She denies constitutional symptoms and shortness of breath. She thinks she may have a urinary tract infection, as she’s had burning with urination for several days, but says this is beginning to improve. Physical exam reveals a blood pressure of 168/90 mm Hg; pulse, 106 beats/min; temperature, 98.4° F; and O2 saturation, 94% on room air. Her weight is 132 lb and her height, 60 in. She is alert and quite spry, with a lot of energy. She wears glasses and bilateral hearing aids. Jugular distention is present to the angle of the jaw. There is no thyromegaly. The pulmonary exam is remarkable for crackles in both lung bases. The heart rhythm is irregularly irregular at a rate of 110 beats/min, and a grade II/VI murmur of mitral regurgitation is heard at the left lower sternal border. The abdomen is soft and nontender, with multiple surgical scars. The lower extremities are remarkable for 2+ pitting edema bilaterally to the level of the mid-calf. Osteoarthritic changes are present in both hands. The neurologic exam is grossly intact. An ECG reveals a ventricular rate of 110 beats/min; PR interval, not measured; QRS duration, 144 ms; QT/QTc interval, 298/403 ms; no P axis; R axis, –36°; and T axis, 169°. What is your interpretation of this ECG?
Sterile or non-sterile gloves for minor skin excisions?
Consider using non-sterile gloves during minor skin excisions (even those that require sutures) because the infection rate is not increased compared to using sterile gloves.1
Strength of recommendation
B: Based on a randomized controlled trial done in a primary care practice.
Heal C, Sriharan S, Buttner PG, et al. Comparing non-sterile to sterile gloves for minor surgery: a prospective randomized controlled noninferiority trial. Med J Aust. 2015;202:27-31.
Illustrative case
A 50-year-old man comes to your office to have a mole removed from his arm. You decide to excise the lesion in your office today. Do you need to use sterile gloves for this procedure, or can you use gloves from the clean non-sterile box in the exam room?
Non-sterile gloves are readily available during a typical office visit and cost up to a dollar less per pair than sterile gloves.1-3 Studies conducted in settings other than primary care offices have shown that non-sterile gloves do not increase the risk of infection during several types of minor skin procedures.
A partially blinded, randomized controlled trial (RCT) in an emergency department found no significant difference in infection rates between the use of sterile (6.1%) vs non-sterile (4.4%) gloves during laceration repairs.2 Similarly, a small RCT in an outpatient dermatology clinic and a larger prospective trial by a Mohs dermatologist showed that infection rates were not increased after Mohs surgery using non-sterile (0.49%) vs sterile (0.50%) gloves.3,4
Guidelines on the use of sterile vs non-sterile gloves for minor skin excisions in outpatient primary care are difficult to come by. Current guidelines from the Centers for Disease Control and Prevention (CDC) and other agencies regarding surgical site infections are broad and focus on the operating room environment.5-7
The American Academy of Dermatology is working on a guideline for treatment of non-melanoma skin cancer that’s due out this winter, and this may provide additional guidance.8 A 2003 review instructed primary care physicians to use sterile gloves for excisional skin biopsies that require sutures.9
The 2015 study by Heal et al1 appears to be the first RCT to address the question of sterile vs non-sterile glove use for minor skin excisions in a primary care outpatient practice.
STUDY SUMMARY: Non-sterile gloves are not inferior to sterile gloves
Heal et al1 conducted a prospective, randomized, controlled, noninferiority trial to compare the incidence of infection after minor skin surgery performed by 6 physicians from a single general practice in Australia using sterile vs non-sterile clean gloves. They evaluated 576 consecutive patients who presented for skin excision between June 2012 and March 2013. Eighty-three patients were excluded because they had a latex allergy, were using oral antibiotics or immunosuppressive drugs, or required a skin flap procedure or excision of a sebaceous cyst. The physicians followed a standard process for performing the procedures and did not use topical antibiotics or antiseptic cleansing after the procedure.
The primary outcome was surgical site infection within 30 days of the excision, defined as purulent discharge, pain or tenderness, localized swelling or redness or heat at the site, or a diagnosis of skin or soft tissue infection by a general practitioner. The clinicians who assessed for infection were blinded to the patient’s assignment to the sterile or non-sterile glove group, and a stitch abscess was not counted as an infection.
The patients’ mean age was 65 years and 59% were men. At baseline, there were no large differences between patients in the sterile and non-sterile glove groups in terms of smoking status, anticoagulant or steroid use, diabetes, excision site, size of excision, and median days until removal of sutures. The lesions were identified histologically as nevus or seborrheic keratosis, skin cancer and precursor, or other.
The incidence of infection in the non-sterile gloves group was 21/241 (8.7%; 95% confidence interval [CI], 4.9%-12.6%) vs 22/237 in the control group (9.3%; 95% CI, 7.4%-11.1%). The CI (95%) for the difference in infection rate (-0.6%) was -4.0% to 2.9%. This was significantly below the predetermined noninferiority margin of 7%. In a sensitivity analysis of patients lost to follow-up (15 patients, 3%) that assumed all of these patients were without infection, or with infection, the CI was still below the noninferiority margin of 7%. The per-protocol analysis showed similar results.
WHAT'S NEW: New evidence questions the need for sterile gloves for in-office excisions
Heal et al1 demonstrated that in a primary care setting, non-sterile gloves are not inferior to sterile gloves for performing excisional procedures that require sutures. While standard practice has many family physicians using sterile gloves for these procedures, this study promotes changing this behavior.
CAVEATS: A high infection rate, other factors might limit generalizability
The overall rate of infection in this study (9%) was higher than that found in the studies from emergency medicine and dermatology literature cited earlier.2-4 A similarly high infection rate has been found in other studies of minor surgery by Heal et al, including a 2006 study that showed a wound infection rate of 8.6%.10 The significance of the higher infection rate is unknown, but there is no clear reason why non-sterile gloves might be less effective in preventing infection in environments with lower infection rates.
This was not a double-blinded study, and physicians might change their behavior during a procedure depending on the type of gloves they are wearing. The sterile gloves used in this study contained powder, while the non-sterile gloves were powderless, but this variable is not known to affect infection rates. A study of Mohs surgery avoided this variable by only using powderless gloves, and had similar outcomes in terms of the difference in infection rate between sterile and non-sterile gloves.4
CHALLENGES TO IMPLEMENTATION: Ingrained habits can be hard to change
Tradition and training die hard. While multiple studies in several settings have found non-sterile gloves are non-inferior to sterile gloves in preventing surgical site infection after minor skin surgeries, this single study in the primary care office setting may not be enough to sway family physicians from ingrained habits.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
1. Heal C, Sriharan S, Buttner PG, et al. Comparing non-sterile to sterile gloves for minor surgery: a prospective randomized controlled non-inferiority trial. Med J Aust. 2015;202:27-31.
2. Perelman VS, Francis GJ, Rutledge T, et al. Sterile versus nonsterile gloves for repair of uncomplicated lacerations in the emergency department: a randomized controlled trial. Ann Emerg Med. 2004;43:362-370.
3. Mehta D, Chambers N, Adams B, et al. Comparison of the prevalence of surgical site infection with use of sterile versus nonsterile gloves for resection and reconstruction during Mohs surgery. Dermatol Surg. 2014;40:234-239.
4. Xia Y, Cho S, Greenway HT, et al. Infection rates of wound repairs during Mohs micrographic surgery using sterile versus nonsterile gloves: a prospective randomized pilot study. Dermatol Surg. 2011;37:651-656.
5. Mangram AJ, Horan TC, Pearson ML, et al. Guideline for prevention of surgical site infection, 1999. Centers for Disease Control and Prevention (CDC) Hospital Infection Control Practices Advisory Committee. Am J Infect Control. 1999;27:97-132.
6. National Institute for Health and Care Excellence. Surgical site infections: prevention and treatment. October 2008. Available at: https://www.nice.org.uk/guidance/cg74. Accessed July 28, 2015.
7. National Health and Medical Research Council. Australian Guidelines for the Prevention and Control of Infection in Healthcare (2010). Updated August 28, 2013. Available at: http://www.nhmrc.gov.au/book/html-australian-guideline-sprevention-and-control-infection-healthcare-2010. Accessed July 31, 2015.
8. American Academy of Dermatology. Clinical Guidelines. American Academy of Dermatology Web site. Available at: https://www.aad.org/education/clinical-guidelines. Accessed July 28, 2015.
9. Zuber TJ. Fusiform excision. Am Fam Physician. 2003;67:1539-1544.
10. Heal C, Buettner P, Browning S. Risk factors for wound infection after minor surgery in general practice. Med J Aust. 2006;18:255-258.
Consider using non-sterile gloves during minor skin excisions (even those that require sutures) because the infection rate is not increased compared to using sterile gloves.1
Strength of recommendation
B: Based on a randomized controlled trial done in a primary care practice.
Heal C, Sriharan S, Buttner PG, et al. Comparing non-sterile to sterile gloves for minor surgery: a prospective randomized controlled noninferiority trial. Med J Aust. 2015;202:27-31.
Illustrative case
A 50-year-old man comes to your office to have a mole removed from his arm. You decide to excise the lesion in your office today. Do you need to use sterile gloves for this procedure, or can you use gloves from the clean non-sterile box in the exam room?
Non-sterile gloves are readily available during a typical office visit and cost up to a dollar less per pair than sterile gloves.1-3 Studies conducted in settings other than primary care offices have shown that non-sterile gloves do not increase the risk of infection during several types of minor skin procedures.
A partially blinded, randomized controlled trial (RCT) in an emergency department found no significant difference in infection rates between the use of sterile (6.1%) vs non-sterile (4.4%) gloves during laceration repairs.2 Similarly, a small RCT in an outpatient dermatology clinic and a larger prospective trial by a Mohs dermatologist showed that infection rates were not increased after Mohs surgery using non-sterile (0.49%) vs sterile (0.50%) gloves.3,4
Guidelines on the use of sterile vs non-sterile gloves for minor skin excisions in outpatient primary care are difficult to come by. Current guidelines from the Centers for Disease Control and Prevention (CDC) and other agencies regarding surgical site infections are broad and focus on the operating room environment.5-7
The American Academy of Dermatology is working on a guideline for treatment of non-melanoma skin cancer that’s due out this winter, and this may provide additional guidance.8 A 2003 review instructed primary care physicians to use sterile gloves for excisional skin biopsies that require sutures.9
The 2015 study by Heal et al1 appears to be the first RCT to address the question of sterile vs non-sterile glove use for minor skin excisions in a primary care outpatient practice.
STUDY SUMMARY: Non-sterile gloves are not inferior to sterile gloves
Heal et al1 conducted a prospective, randomized, controlled, noninferiority trial to compare the incidence of infection after minor skin surgery performed by 6 physicians from a single general practice in Australia using sterile vs non-sterile clean gloves. They evaluated 576 consecutive patients who presented for skin excision between June 2012 and March 2013. Eighty-three patients were excluded because they had a latex allergy, were using oral antibiotics or immunosuppressive drugs, or required a skin flap procedure or excision of a sebaceous cyst. The physicians followed a standard process for performing the procedures and did not use topical antibiotics or antiseptic cleansing after the procedure.
The primary outcome was surgical site infection within 30 days of the excision, defined as purulent discharge, pain or tenderness, localized swelling or redness or heat at the site, or a diagnosis of skin or soft tissue infection by a general practitioner. The clinicians who assessed for infection were blinded to the patient’s assignment to the sterile or non-sterile glove group, and a stitch abscess was not counted as an infection.
The patients’ mean age was 65 years and 59% were men. At baseline, there were no large differences between patients in the sterile and non-sterile glove groups in terms of smoking status, anticoagulant or steroid use, diabetes, excision site, size of excision, and median days until removal of sutures. The lesions were identified histologically as nevus or seborrheic keratosis, skin cancer and precursor, or other.
The incidence of infection in the non-sterile gloves group was 21/241 (8.7%; 95% confidence interval [CI], 4.9%-12.6%) vs 22/237 in the control group (9.3%; 95% CI, 7.4%-11.1%). The CI (95%) for the difference in infection rate (-0.6%) was -4.0% to 2.9%. This was significantly below the predetermined noninferiority margin of 7%. In a sensitivity analysis of patients lost to follow-up (15 patients, 3%) that assumed all of these patients were without infection, or with infection, the CI was still below the noninferiority margin of 7%. The per-protocol analysis showed similar results.
WHAT'S NEW: New evidence questions the need for sterile gloves for in-office excisions
Heal et al1 demonstrated that in a primary care setting, non-sterile gloves are not inferior to sterile gloves for performing excisional procedures that require sutures. While standard practice has many family physicians using sterile gloves for these procedures, this study promotes changing this behavior.
CAVEATS: A high infection rate, other factors might limit generalizability
The overall rate of infection in this study (9%) was higher than that found in the studies from emergency medicine and dermatology literature cited earlier.2-4 A similarly high infection rate has been found in other studies of minor surgery by Heal et al, including a 2006 study that showed a wound infection rate of 8.6%.10 The significance of the higher infection rate is unknown, but there is no clear reason why non-sterile gloves might be less effective in preventing infection in environments with lower infection rates.
This was not a double-blinded study, and physicians might change their behavior during a procedure depending on the type of gloves they are wearing. The sterile gloves used in this study contained powder, while the non-sterile gloves were powderless, but this variable is not known to affect infection rates. A study of Mohs surgery avoided this variable by only using powderless gloves, and had similar outcomes in terms of the difference in infection rate between sterile and non-sterile gloves.4
CHALLENGES TO IMPLEMENTATION: Ingrained habits can be hard to change
Tradition and training die hard. While multiple studies in several settings have found non-sterile gloves are non-inferior to sterile gloves in preventing surgical site infection after minor skin surgeries, this single study in the primary care office setting may not be enough to sway family physicians from ingrained habits.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
Consider using non-sterile gloves during minor skin excisions (even those that require sutures) because the infection rate is not increased compared to using sterile gloves.1
Strength of recommendation
B: Based on a randomized controlled trial done in a primary care practice.
Heal C, Sriharan S, Buttner PG, et al. Comparing non-sterile to sterile gloves for minor surgery: a prospective randomized controlled noninferiority trial. Med J Aust. 2015;202:27-31.
Illustrative case
A 50-year-old man comes to your office to have a mole removed from his arm. You decide to excise the lesion in your office today. Do you need to use sterile gloves for this procedure, or can you use gloves from the clean non-sterile box in the exam room?
Non-sterile gloves are readily available during a typical office visit and cost up to a dollar less per pair than sterile gloves.1-3 Studies conducted in settings other than primary care offices have shown that non-sterile gloves do not increase the risk of infection during several types of minor skin procedures.
A partially blinded, randomized controlled trial (RCT) in an emergency department found no significant difference in infection rates between the use of sterile (6.1%) vs non-sterile (4.4%) gloves during laceration repairs.2 Similarly, a small RCT in an outpatient dermatology clinic and a larger prospective trial by a Mohs dermatologist showed that infection rates were not increased after Mohs surgery using non-sterile (0.49%) vs sterile (0.50%) gloves.3,4
Guidelines on the use of sterile vs non-sterile gloves for minor skin excisions in outpatient primary care are difficult to come by. Current guidelines from the Centers for Disease Control and Prevention (CDC) and other agencies regarding surgical site infections are broad and focus on the operating room environment.5-7
The American Academy of Dermatology is working on a guideline for treatment of non-melanoma skin cancer that’s due out this winter, and this may provide additional guidance.8 A 2003 review instructed primary care physicians to use sterile gloves for excisional skin biopsies that require sutures.9
The 2015 study by Heal et al1 appears to be the first RCT to address the question of sterile vs non-sterile glove use for minor skin excisions in a primary care outpatient practice.
STUDY SUMMARY: Non-sterile gloves are not inferior to sterile gloves
Heal et al1 conducted a prospective, randomized, controlled, noninferiority trial to compare the incidence of infection after minor skin surgery performed by 6 physicians from a single general practice in Australia using sterile vs non-sterile clean gloves. They evaluated 576 consecutive patients who presented for skin excision between June 2012 and March 2013. Eighty-three patients were excluded because they had a latex allergy, were using oral antibiotics or immunosuppressive drugs, or required a skin flap procedure or excision of a sebaceous cyst. The physicians followed a standard process for performing the procedures and did not use topical antibiotics or antiseptic cleansing after the procedure.
The primary outcome was surgical site infection within 30 days of the excision, defined as purulent discharge, pain or tenderness, localized swelling or redness or heat at the site, or a diagnosis of skin or soft tissue infection by a general practitioner. The clinicians who assessed for infection were blinded to the patient’s assignment to the sterile or non-sterile glove group, and a stitch abscess was not counted as an infection.
The patients’ mean age was 65 years and 59% were men. At baseline, there were no large differences between patients in the sterile and non-sterile glove groups in terms of smoking status, anticoagulant or steroid use, diabetes, excision site, size of excision, and median days until removal of sutures. The lesions were identified histologically as nevus or seborrheic keratosis, skin cancer and precursor, or other.
The incidence of infection in the non-sterile gloves group was 21/241 (8.7%; 95% confidence interval [CI], 4.9%-12.6%) vs 22/237 in the control group (9.3%; 95% CI, 7.4%-11.1%). The CI (95%) for the difference in infection rate (-0.6%) was -4.0% to 2.9%. This was significantly below the predetermined noninferiority margin of 7%. In a sensitivity analysis of patients lost to follow-up (15 patients, 3%) that assumed all of these patients were without infection, or with infection, the CI was still below the noninferiority margin of 7%. The per-protocol analysis showed similar results.
WHAT'S NEW: New evidence questions the need for sterile gloves for in-office excisions
Heal et al1 demonstrated that in a primary care setting, non-sterile gloves are not inferior to sterile gloves for performing excisional procedures that require sutures. While standard practice has many family physicians using sterile gloves for these procedures, this study promotes changing this behavior.
CAVEATS: A high infection rate, other factors might limit generalizability
The overall rate of infection in this study (9%) was higher than that found in the studies from emergency medicine and dermatology literature cited earlier.2-4 A similarly high infection rate has been found in other studies of minor surgery by Heal et al, including a 2006 study that showed a wound infection rate of 8.6%.10 The significance of the higher infection rate is unknown, but there is no clear reason why non-sterile gloves might be less effective in preventing infection in environments with lower infection rates.
This was not a double-blinded study, and physicians might change their behavior during a procedure depending on the type of gloves they are wearing. The sterile gloves used in this study contained powder, while the non-sterile gloves were powderless, but this variable is not known to affect infection rates. A study of Mohs surgery avoided this variable by only using powderless gloves, and had similar outcomes in terms of the difference in infection rate between sterile and non-sterile gloves.4
CHALLENGES TO IMPLEMENTATION: Ingrained habits can be hard to change
Tradition and training die hard. While multiple studies in several settings have found non-sterile gloves are non-inferior to sterile gloves in preventing surgical site infection after minor skin surgeries, this single study in the primary care office setting may not be enough to sway family physicians from ingrained habits.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
1. Heal C, Sriharan S, Buttner PG, et al. Comparing non-sterile to sterile gloves for minor surgery: a prospective randomized controlled non-inferiority trial. Med J Aust. 2015;202:27-31.
2. Perelman VS, Francis GJ, Rutledge T, et al. Sterile versus nonsterile gloves for repair of uncomplicated lacerations in the emergency department: a randomized controlled trial. Ann Emerg Med. 2004;43:362-370.
3. Mehta D, Chambers N, Adams B, et al. Comparison of the prevalence of surgical site infection with use of sterile versus nonsterile gloves for resection and reconstruction during Mohs surgery. Dermatol Surg. 2014;40:234-239.
4. Xia Y, Cho S, Greenway HT, et al. Infection rates of wound repairs during Mohs micrographic surgery using sterile versus nonsterile gloves: a prospective randomized pilot study. Dermatol Surg. 2011;37:651-656.
5. Mangram AJ, Horan TC, Pearson ML, et al. Guideline for prevention of surgical site infection, 1999. Centers for Disease Control and Prevention (CDC) Hospital Infection Control Practices Advisory Committee. Am J Infect Control. 1999;27:97-132.
6. National Institute for Health and Care Excellence. Surgical site infections: prevention and treatment. October 2008. Available at: https://www.nice.org.uk/guidance/cg74. Accessed July 28, 2015.
7. National Health and Medical Research Council. Australian Guidelines for the Prevention and Control of Infection in Healthcare (2010). Updated August 28, 2013. Available at: http://www.nhmrc.gov.au/book/html-australian-guideline-sprevention-and-control-infection-healthcare-2010. Accessed July 31, 2015.
8. American Academy of Dermatology. Clinical Guidelines. American Academy of Dermatology Web site. Available at: https://www.aad.org/education/clinical-guidelines. Accessed July 28, 2015.
9. Zuber TJ. Fusiform excision. Am Fam Physician. 2003;67:1539-1544.
10. Heal C, Buettner P, Browning S. Risk factors for wound infection after minor surgery in general practice. Med J Aust. 2006;18:255-258.
1. Heal C, Sriharan S, Buttner PG, et al. Comparing non-sterile to sterile gloves for minor surgery: a prospective randomized controlled non-inferiority trial. Med J Aust. 2015;202:27-31.
2. Perelman VS, Francis GJ, Rutledge T, et al. Sterile versus nonsterile gloves for repair of uncomplicated lacerations in the emergency department: a randomized controlled trial. Ann Emerg Med. 2004;43:362-370.
3. Mehta D, Chambers N, Adams B, et al. Comparison of the prevalence of surgical site infection with use of sterile versus nonsterile gloves for resection and reconstruction during Mohs surgery. Dermatol Surg. 2014;40:234-239.
4. Xia Y, Cho S, Greenway HT, et al. Infection rates of wound repairs during Mohs micrographic surgery using sterile versus nonsterile gloves: a prospective randomized pilot study. Dermatol Surg. 2011;37:651-656.
5. Mangram AJ, Horan TC, Pearson ML, et al. Guideline for prevention of surgical site infection, 1999. Centers for Disease Control and Prevention (CDC) Hospital Infection Control Practices Advisory Committee. Am J Infect Control. 1999;27:97-132.
6. National Institute for Health and Care Excellence. Surgical site infections: prevention and treatment. October 2008. Available at: https://www.nice.org.uk/guidance/cg74. Accessed July 28, 2015.
7. National Health and Medical Research Council. Australian Guidelines for the Prevention and Control of Infection in Healthcare (2010). Updated August 28, 2013. Available at: http://www.nhmrc.gov.au/book/html-australian-guideline-sprevention-and-control-infection-healthcare-2010. Accessed July 31, 2015.
8. American Academy of Dermatology. Clinical Guidelines. American Academy of Dermatology Web site. Available at: https://www.aad.org/education/clinical-guidelines. Accessed July 28, 2015.
9. Zuber TJ. Fusiform excision. Am Fam Physician. 2003;67:1539-1544.
10. Heal C, Buettner P, Browning S. Risk factors for wound infection after minor surgery in general practice. Med J Aust. 2006;18:255-258.
Copyright © 2015. The Family Physicians Inquiries Network. All rights reserved.
Sizing up EMRs and patient care from the other side of the bed rail
Dr. Unger’s guest editorial, “Med students: Look up from your EMRs” (J Fam Pract. 2015;64:517-518), vividly describes what those who have been paying attention see quite clearly: Not only has the widespread implementation of electronic medical records (EMRs) failed to deliver all it has promised, but it has made patient care worse. Many students and members of the health care team spend as little time as possible talking and listening to patients. Instead, the goal is to complete every box in our EMRs to qualify for meaningful use payments and whatever “quality” incentives are available in our local environment.
That said, I believe EMRs are very good at doing the things computers do well, and I hope I never again have to rifle through a paper chart the size of a phone book to find a critical piece of information. The problem lies in the myriad inappropriate ways the EMR is used in place of accurately telling the patient’s story, and the resulting diversion of the entire health care team away from caring for the patients we are supposedly here to serve.
I am tired of complaining to my patients, partners, family, friends, and anyone else who will listen. It is time for family medicine to reclaim its role as “counterculture” and lead the charge for comprehensive, continuous, compassionate care—whose centerpiece is actually talking to, listening to, and examining patients.
David A. Silverstein, MD
Buffalo, NY
While I agree with Dr. Unger about EMRs, I respectfully disagree with his approach when he suspected he had appendicitis. When he initially ordered his own computed tomography scan, rather than seeing his own doctor or going to the emergency department, he (inadvertently) “assigned” himself as his own doctor. He then should have at least offered his history in the hospital, rather than making it a test for the student and the hospital. It sounds like an adversarial situation developed, which did not help matters. Good that he’s doing OK!
Michael Kelly, MD
Minneapolis, Minn
Dr. Unger’s guest editorial, “Med students: Look up from your EMRs” (J Fam Pract. 2015;64:517-518), vividly describes what those who have been paying attention see quite clearly: Not only has the widespread implementation of electronic medical records (EMRs) failed to deliver all it has promised, but it has made patient care worse. Many students and members of the health care team spend as little time as possible talking and listening to patients. Instead, the goal is to complete every box in our EMRs to qualify for meaningful use payments and whatever “quality” incentives are available in our local environment.
That said, I believe EMRs are very good at doing the things computers do well, and I hope I never again have to rifle through a paper chart the size of a phone book to find a critical piece of information. The problem lies in the myriad inappropriate ways the EMR is used in place of accurately telling the patient’s story, and the resulting diversion of the entire health care team away from caring for the patients we are supposedly here to serve.
I am tired of complaining to my patients, partners, family, friends, and anyone else who will listen. It is time for family medicine to reclaim its role as “counterculture” and lead the charge for comprehensive, continuous, compassionate care—whose centerpiece is actually talking to, listening to, and examining patients.
David A. Silverstein, MD
Buffalo, NY
While I agree with Dr. Unger about EMRs, I respectfully disagree with his approach when he suspected he had appendicitis. When he initially ordered his own computed tomography scan, rather than seeing his own doctor or going to the emergency department, he (inadvertently) “assigned” himself as his own doctor. He then should have at least offered his history in the hospital, rather than making it a test for the student and the hospital. It sounds like an adversarial situation developed, which did not help matters. Good that he’s doing OK!
Michael Kelly, MD
Minneapolis, Minn
Dr. Unger’s guest editorial, “Med students: Look up from your EMRs” (J Fam Pract. 2015;64:517-518), vividly describes what those who have been paying attention see quite clearly: Not only has the widespread implementation of electronic medical records (EMRs) failed to deliver all it has promised, but it has made patient care worse. Many students and members of the health care team spend as little time as possible talking and listening to patients. Instead, the goal is to complete every box in our EMRs to qualify for meaningful use payments and whatever “quality” incentives are available in our local environment.
That said, I believe EMRs are very good at doing the things computers do well, and I hope I never again have to rifle through a paper chart the size of a phone book to find a critical piece of information. The problem lies in the myriad inappropriate ways the EMR is used in place of accurately telling the patient’s story, and the resulting diversion of the entire health care team away from caring for the patients we are supposedly here to serve.
I am tired of complaining to my patients, partners, family, friends, and anyone else who will listen. It is time for family medicine to reclaim its role as “counterculture” and lead the charge for comprehensive, continuous, compassionate care—whose centerpiece is actually talking to, listening to, and examining patients.
David A. Silverstein, MD
Buffalo, NY
While I agree with Dr. Unger about EMRs, I respectfully disagree with his approach when he suspected he had appendicitis. When he initially ordered his own computed tomography scan, rather than seeing his own doctor or going to the emergency department, he (inadvertently) “assigned” himself as his own doctor. He then should have at least offered his history in the hospital, rather than making it a test for the student and the hospital. It sounds like an adversarial situation developed, which did not help matters. Good that he’s doing OK!
Michael Kelly, MD
Minneapolis, Minn
Advances in Colorectal Cancer Screening
Colorectal cancer (CRC) screening has been shown to save lives. Screening can prevent CRC by detecting and removing precancerous adenomatous polyps, which are the precursors of most cancers.1 Screening also can detect cancer at an early, asymptomatic stage while it is still localized and amenable to treatment; 5-year survival rates are 80% to 90% for patients with localized, early stage I/II CRC.2
Colorectal cancer (CRC) screening has been shown to save lives. Screening can prevent CRC by detecting and removing precancerous adenomatous polyps, which are the precursors of most cancers.1 Screening also can detect cancer at an early, asymptomatic stage while it is still localized and amenable to treatment; 5-year survival rates are 80% to 90% for patients with localized, early stage I/II CRC.2
Colorectal cancer (CRC) screening has been shown to save lives. Screening can prevent CRC by detecting and removing precancerous adenomatous polyps, which are the precursors of most cancers.1 Screening also can detect cancer at an early, asymptomatic stage while it is still localized and amenable to treatment; 5-year survival rates are 80% to 90% for patients with localized, early stage I/II CRC.2