User login
Identifying and Managing MS Relapse
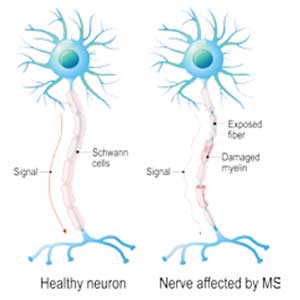
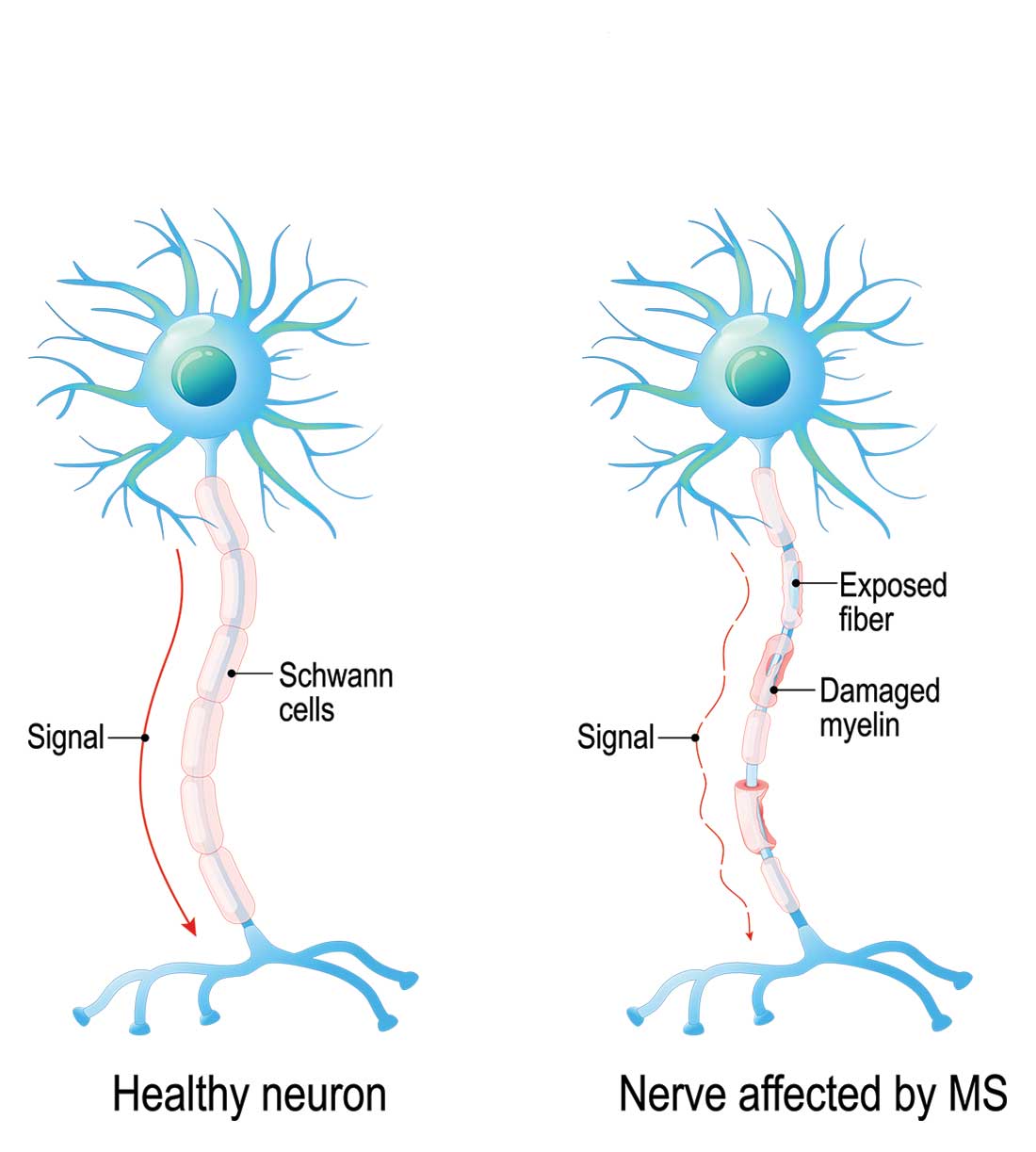
Multiple sclerosis (MS) is a chronic, autoimmune-mediated disorder of the central nervous system that affects more than 40,000 people in the United States. About 85% of cases are categorized as relapsing remitting multiple sclerosis (RRMS), based on the clinical and radiographic pattern of focal demyelination in different regions of the brain and spinal cord over time. Though not fully understood, the pathophysiology of RRMS involves axonal degeneration and inflammatory demyelination; the latter is considered a relapse in patients with an established MS diagnosis.
OVERVIEW
MS relapse can have a significant impact on patients’ short- and long-term function, quality of life, and finances. Relapse may be identified via
- New neurologic symptoms reported by the patient
- New neurologic findings on physical examination
- New radiographic findings on contrast-enhanced MRI of the central nervous system, or
- Abnormal results of cerebrospinal fluid analysis.
Patient-reported symptoms and abnormal signs identified on physical exam should correspond with the area of the central nervous system affected. In some cases, patients may have radiographic evidence of relapse without symptoms or signs.
It is essential for health care providers to identify relapse, as it is an important marker of disease activity that may warrant treatment—particularly if symptoms are impacting function or if there is optic neuritis. MS relapse is also an indicator of suboptimal response to disease-modifying therapies.
Treatment of relapse is one component of RRMS management, which also includes symptom management and use of disease-modifying therapy to reduce risk for disease activity and decline in function.
DIAGNOSING RELAPSE
Because risk for MS relapse cannot be predicted, both patients and providers need to have a high index of suspicion in the setting of new neurologic symptoms or decline in function. Relapse should be considered when these symptoms last longer than 24 hours in the absence of fever or infection. The clinical features of relapse should have corresponding radiographic evidence of active demyelination on contrast-enhanced MRI.
A pseudo-relapse is characterized by new or worsening neurologic symptoms lasting longer than 24 hours with concurrent fever, infection, or other metabolic derangement. Pseudo-relapse does not show radiographic evidence of active demyelination on contrast-enhanced MRI.
Continue to: Aggravation of longstanding neurologic symptoms...
Aggravation of longstanding neurologic symptoms is not considered a relapse, as no new radiographic evidence of disease progression will be seen on MRI. Factors that may contribute to aggravation of established symptoms include an increase in core body temperature, sleep deprivation, and psychosocial stress.
When a patient with suspected or diagnosed RRMS presents with new neurologic symptoms of more than 24 hours’ duration, the first step is to conduct a physical exam to assess for objective evidence of neurologic deficits and signs of infection, including fever. The provider should also order select laboratory testing—including a complete blood cell count and urinalysis with culture—to exclude infection. In certain cases, contrast-enhanced MRI of the brain and/or spine may be ordered; however, this may delay treatment initiation if the study cannot be promptly scheduled.
Evaluation and management should involve communication, if not face-to-face consultation, with the patient’s neurology provider who is responsible for MS management.
IMMEDIATE MANAGEMENT
Acute relapses are managed with anti-inflammatory agents. For some patients, treatment may provide symptomatic relief, shorten the recovery phase, and improve motor function. Long-term benefits have not been demonstrated, except in patients with optic neuritis.
Firstline therapy for MS relapse is high-dose corticosteroids, which can be administered at home, at an ambulatory infusion center, or (in some cases) in a hospital setting. The preferred regimen is methylprednisolone (1 g IV for 3-5 d), with or without prednisone taper. Another option is dexamethasone (80 mg bid for 3-5 d), with or without prednisone taper.1-3
Continue to: Common adverse effects of corticosteroids include...
Common adverse effects of corticosteroids include headache, emotional lability, insomnia, glucose intolerance, hypertension, dyspepsia, and exacerbation of psychiatric conditions; drug interactions should also be considered. Patients with diabetes may need to be admitted to the hospital for glycemic monitoring and control.
High-dose corticosteroids are associated with a rare, non–dose-dependent risk for aseptic femoral necrosis. For patients who are refractory to or not candidates for corticosteroids, adrenocorticotropic hormone (ACTH) gel (80 U/d IM or subQ daily for 10 d) is an option. This medication may be better tolerated, although it is much more expensive than corticosteroids. Plasmapheresis and IV immunoglobulin are also options for patients with refractory symptoms or contraindications to recommended therapies.1-3
ONGOING MANAGEMENT
Once a treatment plan is initiated, providers should carefully follow the patient’s response in terms of adverse effects, symptom improvement, and functional recovery. Those with refractory symptoms may need additional doses of the initial therapy or an alternative therapy.
The relapse recovery period may last several months and be complete or incomplete, so providers may also need to manage neurologic symptoms and functional deficits (with pharmacologic and/or nonpharmacologic options). Patients who have had a relapse should also meet with their neurology provider to discuss their disease-modifying therapy plan, since relapse indicates a suboptimal response to current therapy.
1. Bevan C, Gelfand JM. Therapeutic management of severe relapses in multiple sclerosis. Curr Treat Options Neurol. 2015;17(4):17.
2. Frohman TC, O’Donoghue DL, Northrop D, eds. Multiple Sclerosis for the Physician Assistant. National Multiple Sclerosis Society. 2011.
3. Giesser B, ed. Primer on Multiple Sclerosis . 2nd ed. Oxford, UK: Oxford University Press ; 2015.
Multiple sclerosis (MS) is a chronic, autoimmune-mediated disorder of the central nervous system that affects more than 40,000 people in the United States. About 85% of cases are categorized as relapsing remitting multiple sclerosis (RRMS), based on the clinical and radiographic pattern of focal demyelination in different regions of the brain and spinal cord over time. Though not fully understood, the pathophysiology of RRMS involves axonal degeneration and inflammatory demyelination; the latter is considered a relapse in patients with an established MS diagnosis.
OVERVIEW
MS relapse can have a significant impact on patients’ short- and long-term function, quality of life, and finances. Relapse may be identified via
- New neurologic symptoms reported by the patient
- New neurologic findings on physical examination
- New radiographic findings on contrast-enhanced MRI of the central nervous system, or
- Abnormal results of cerebrospinal fluid analysis.
Patient-reported symptoms and abnormal signs identified on physical exam should correspond with the area of the central nervous system affected. In some cases, patients may have radiographic evidence of relapse without symptoms or signs.
It is essential for health care providers to identify relapse, as it is an important marker of disease activity that may warrant treatment—particularly if symptoms are impacting function or if there is optic neuritis. MS relapse is also an indicator of suboptimal response to disease-modifying therapies.
Treatment of relapse is one component of RRMS management, which also includes symptom management and use of disease-modifying therapy to reduce risk for disease activity and decline in function.
DIAGNOSING RELAPSE
Because risk for MS relapse cannot be predicted, both patients and providers need to have a high index of suspicion in the setting of new neurologic symptoms or decline in function. Relapse should be considered when these symptoms last longer than 24 hours in the absence of fever or infection. The clinical features of relapse should have corresponding radiographic evidence of active demyelination on contrast-enhanced MRI.
A pseudo-relapse is characterized by new or worsening neurologic symptoms lasting longer than 24 hours with concurrent fever, infection, or other metabolic derangement. Pseudo-relapse does not show radiographic evidence of active demyelination on contrast-enhanced MRI.
Continue to: Aggravation of longstanding neurologic symptoms...
Aggravation of longstanding neurologic symptoms is not considered a relapse, as no new radiographic evidence of disease progression will be seen on MRI. Factors that may contribute to aggravation of established symptoms include an increase in core body temperature, sleep deprivation, and psychosocial stress.
When a patient with suspected or diagnosed RRMS presents with new neurologic symptoms of more than 24 hours’ duration, the first step is to conduct a physical exam to assess for objective evidence of neurologic deficits and signs of infection, including fever. The provider should also order select laboratory testing—including a complete blood cell count and urinalysis with culture—to exclude infection. In certain cases, contrast-enhanced MRI of the brain and/or spine may be ordered; however, this may delay treatment initiation if the study cannot be promptly scheduled.
Evaluation and management should involve communication, if not face-to-face consultation, with the patient’s neurology provider who is responsible for MS management.
IMMEDIATE MANAGEMENT
Acute relapses are managed with anti-inflammatory agents. For some patients, treatment may provide symptomatic relief, shorten the recovery phase, and improve motor function. Long-term benefits have not been demonstrated, except in patients with optic neuritis.
Firstline therapy for MS relapse is high-dose corticosteroids, which can be administered at home, at an ambulatory infusion center, or (in some cases) in a hospital setting. The preferred regimen is methylprednisolone (1 g IV for 3-5 d), with or without prednisone taper. Another option is dexamethasone (80 mg bid for 3-5 d), with or without prednisone taper.1-3
Continue to: Common adverse effects of corticosteroids include...
Common adverse effects of corticosteroids include headache, emotional lability, insomnia, glucose intolerance, hypertension, dyspepsia, and exacerbation of psychiatric conditions; drug interactions should also be considered. Patients with diabetes may need to be admitted to the hospital for glycemic monitoring and control.

High-dose corticosteroids are associated with a rare, non–dose-dependent risk for aseptic femoral necrosis. For patients who are refractory to or not candidates for corticosteroids, adrenocorticotropic hormone (ACTH) gel (80 U/d IM or subQ daily for 10 d) is an option. This medication may be better tolerated, although it is much more expensive than corticosteroids. Plasmapheresis and IV immunoglobulin are also options for patients with refractory symptoms or contraindications to recommended therapies.1-3
ONGOING MANAGEMENT
Once a treatment plan is initiated, providers should carefully follow the patient’s response in terms of adverse effects, symptom improvement, and functional recovery. Those with refractory symptoms may need additional doses of the initial therapy or an alternative therapy.
The relapse recovery period may last several months and be complete or incomplete, so providers may also need to manage neurologic symptoms and functional deficits (with pharmacologic and/or nonpharmacologic options). Patients who have had a relapse should also meet with their neurology provider to discuss their disease-modifying therapy plan, since relapse indicates a suboptimal response to current therapy.
Multiple sclerosis (MS) is a chronic, autoimmune-mediated disorder of the central nervous system that affects more than 40,000 people in the United States. About 85% of cases are categorized as relapsing remitting multiple sclerosis (RRMS), based on the clinical and radiographic pattern of focal demyelination in different regions of the brain and spinal cord over time. Though not fully understood, the pathophysiology of RRMS involves axonal degeneration and inflammatory demyelination; the latter is considered a relapse in patients with an established MS diagnosis.
OVERVIEW
MS relapse can have a significant impact on patients’ short- and long-term function, quality of life, and finances. Relapse may be identified via
- New neurologic symptoms reported by the patient
- New neurologic findings on physical examination
- New radiographic findings on contrast-enhanced MRI of the central nervous system, or
- Abnormal results of cerebrospinal fluid analysis.
Patient-reported symptoms and abnormal signs identified on physical exam should correspond with the area of the central nervous system affected. In some cases, patients may have radiographic evidence of relapse without symptoms or signs.
It is essential for health care providers to identify relapse, as it is an important marker of disease activity that may warrant treatment—particularly if symptoms are impacting function or if there is optic neuritis. MS relapse is also an indicator of suboptimal response to disease-modifying therapies.
Treatment of relapse is one component of RRMS management, which also includes symptom management and use of disease-modifying therapy to reduce risk for disease activity and decline in function.
DIAGNOSING RELAPSE
Because risk for MS relapse cannot be predicted, both patients and providers need to have a high index of suspicion in the setting of new neurologic symptoms or decline in function. Relapse should be considered when these symptoms last longer than 24 hours in the absence of fever or infection. The clinical features of relapse should have corresponding radiographic evidence of active demyelination on contrast-enhanced MRI.
A pseudo-relapse is characterized by new or worsening neurologic symptoms lasting longer than 24 hours with concurrent fever, infection, or other metabolic derangement. Pseudo-relapse does not show radiographic evidence of active demyelination on contrast-enhanced MRI.
Continue to: Aggravation of longstanding neurologic symptoms...
Aggravation of longstanding neurologic symptoms is not considered a relapse, as no new radiographic evidence of disease progression will be seen on MRI. Factors that may contribute to aggravation of established symptoms include an increase in core body temperature, sleep deprivation, and psychosocial stress.
When a patient with suspected or diagnosed RRMS presents with new neurologic symptoms of more than 24 hours’ duration, the first step is to conduct a physical exam to assess for objective evidence of neurologic deficits and signs of infection, including fever. The provider should also order select laboratory testing—including a complete blood cell count and urinalysis with culture—to exclude infection. In certain cases, contrast-enhanced MRI of the brain and/or spine may be ordered; however, this may delay treatment initiation if the study cannot be promptly scheduled.
Evaluation and management should involve communication, if not face-to-face consultation, with the patient’s neurology provider who is responsible for MS management.
IMMEDIATE MANAGEMENT
Acute relapses are managed with anti-inflammatory agents. For some patients, treatment may provide symptomatic relief, shorten the recovery phase, and improve motor function. Long-term benefits have not been demonstrated, except in patients with optic neuritis.
Firstline therapy for MS relapse is high-dose corticosteroids, which can be administered at home, at an ambulatory infusion center, or (in some cases) in a hospital setting. The preferred regimen is methylprednisolone (1 g IV for 3-5 d), with or without prednisone taper. Another option is dexamethasone (80 mg bid for 3-5 d), with or without prednisone taper.1-3
Continue to: Common adverse effects of corticosteroids include...
Common adverse effects of corticosteroids include headache, emotional lability, insomnia, glucose intolerance, hypertension, dyspepsia, and exacerbation of psychiatric conditions; drug interactions should also be considered. Patients with diabetes may need to be admitted to the hospital for glycemic monitoring and control.

High-dose corticosteroids are associated with a rare, non–dose-dependent risk for aseptic femoral necrosis. For patients who are refractory to or not candidates for corticosteroids, adrenocorticotropic hormone (ACTH) gel (80 U/d IM or subQ daily for 10 d) is an option. This medication may be better tolerated, although it is much more expensive than corticosteroids. Plasmapheresis and IV immunoglobulin are also options for patients with refractory symptoms or contraindications to recommended therapies.1-3
ONGOING MANAGEMENT
Once a treatment plan is initiated, providers should carefully follow the patient’s response in terms of adverse effects, symptom improvement, and functional recovery. Those with refractory symptoms may need additional doses of the initial therapy or an alternative therapy.
The relapse recovery period may last several months and be complete or incomplete, so providers may also need to manage neurologic symptoms and functional deficits (with pharmacologic and/or nonpharmacologic options). Patients who have had a relapse should also meet with their neurology provider to discuss their disease-modifying therapy plan, since relapse indicates a suboptimal response to current therapy.
1. Bevan C, Gelfand JM. Therapeutic management of severe relapses in multiple sclerosis. Curr Treat Options Neurol. 2015;17(4):17.
2. Frohman TC, O’Donoghue DL, Northrop D, eds. Multiple Sclerosis for the Physician Assistant. National Multiple Sclerosis Society. 2011.
3. Giesser B, ed. Primer on Multiple Sclerosis . 2nd ed. Oxford, UK: Oxford University Press ; 2015.
1. Bevan C, Gelfand JM. Therapeutic management of severe relapses in multiple sclerosis. Curr Treat Options Neurol. 2015;17(4):17.
2. Frohman TC, O’Donoghue DL, Northrop D, eds. Multiple Sclerosis for the Physician Assistant. National Multiple Sclerosis Society. 2011.
3. Giesser B, ed. Primer on Multiple Sclerosis . 2nd ed. Oxford, UK: Oxford University Press ; 2015.
ICYMI: Canakinumab reduced risk of gout attacks
Canakinumab, an interleukin-1–beta blocker, reduced the risk of a gout attack by 52% when administered every 3 months, according to a secondary exploratory analysis of data from the multicenter, randomized, double-blind, placebo-controlled CANTOS trial (NCT01327846). The results of this analysis were published Sept. 17 in the Annals of Internal Medicine (doi: 10.7326/M18-1167).
We covered this story before it was published in the journal. Find our conference coverage at the links below.
Canakinumab, an interleukin-1–beta blocker, reduced the risk of a gout attack by 52% when administered every 3 months, according to a secondary exploratory analysis of data from the multicenter, randomized, double-blind, placebo-controlled CANTOS trial (NCT01327846). The results of this analysis were published Sept. 17 in the Annals of Internal Medicine (doi: 10.7326/M18-1167).
We covered this story before it was published in the journal. Find our conference coverage at the links below.
Canakinumab, an interleukin-1–beta blocker, reduced the risk of a gout attack by 52% when administered every 3 months, according to a secondary exploratory analysis of data from the multicenter, randomized, double-blind, placebo-controlled CANTOS trial (NCT01327846). The results of this analysis were published Sept. 17 in the Annals of Internal Medicine (doi: 10.7326/M18-1167).
We covered this story before it was published in the journal. Find our conference coverage at the links below.
FROM THE ANNALS OF INTERNAL MEDICINE
Elevated type 2 diabetes risk seen in PsA patients
Patients with incident psoriatic arthritis are at a significantly increased risk of developing type 2 diabetes when compared against patients with psoriasis alone and with the general population, according to recent research published in Rheumatology.
Rachel Charlton, PhD, of the department of pharmacy and pharmacology at the University of Bath (England), and her colleagues performed an analysis of 6,783 incident cases of psoriatic arthritis (PsA) from the U.K. Clinical Practice Research Datalink who were diagnosed during 1998-2014. Patients were between 18 years and 89 years old with a median age of 49 years at PsA diagnosis.
In the study, the researchers randomly matched PsA cases at a 1:4 ratio to either a cohort of general population patients with no PsA, psoriasis, or inflammatory arthritis or a cohort of patients with psoriasis but no PsA or inflammatory arthritis. Patients were followed from match to the point where they either no longer met inclusion criteria for the cohort or received a diagnosis of type 2 diabetes, cerebrovascular disease (CVD), ischemic heart disease (IHD), or peripheral vascular disease (PVD) with a mean follow-up duration of approximately 5.5 years across all patient groups.
Patients in the PsA group had a significantly higher incidence of type 2 diabetes, compared with the general population (adjusted relative risk, 1.40; 95% confidence interval, 1.15-1.70; P = .0007) and psoriasis groups (adjusted RR, 1.53; 95% CI, 1.19-1.97; P = .0009). In the PsA group, risk of CVD (adjusted RR, 1.24; 95% CI, 0.99-1.56; P = .06), IHD (adjusted RR, 1.27; 95% CI, 1.05-1.54; P = .02), and PVD (adjusted RR, 1.40; 95% CI, 1.02-1.92; P = .04) were significantly higher than in the general population but not when compared with the psoriasis group. The overall risk of cardiovascular disease (including CVD, IHD, and PVD) for the PsA group was significantly higher (adjusted RR, 1.29; 95% CI, 1.12-1.48; P = .0005), compared with the general population.
“These results support the proposal in existing clinical guidelines that, in order to reduce cardiovascular risk in patients with PsA, it is important to treat inflammatory disease as well as to screen and treat traditional risk factors early in the disease course,” Ms. Charlton and her colleagues wrote in their study.
This study was funded by a grant from the National Institute for Health Research in the United Kingdom. The authors reported no relevant conflicts of interest.
SOURCE: Charlton RA et al. Rheumatology. 2018 Sep 6. doi: 10.1093/rheumatology/key286.
Patients with incident psoriatic arthritis are at a significantly increased risk of developing type 2 diabetes when compared against patients with psoriasis alone and with the general population, according to recent research published in Rheumatology.
Rachel Charlton, PhD, of the department of pharmacy and pharmacology at the University of Bath (England), and her colleagues performed an analysis of 6,783 incident cases of psoriatic arthritis (PsA) from the U.K. Clinical Practice Research Datalink who were diagnosed during 1998-2014. Patients were between 18 years and 89 years old with a median age of 49 years at PsA diagnosis.
In the study, the researchers randomly matched PsA cases at a 1:4 ratio to either a cohort of general population patients with no PsA, psoriasis, or inflammatory arthritis or a cohort of patients with psoriasis but no PsA or inflammatory arthritis. Patients were followed from match to the point where they either no longer met inclusion criteria for the cohort or received a diagnosis of type 2 diabetes, cerebrovascular disease (CVD), ischemic heart disease (IHD), or peripheral vascular disease (PVD) with a mean follow-up duration of approximately 5.5 years across all patient groups.
Patients in the PsA group had a significantly higher incidence of type 2 diabetes, compared with the general population (adjusted relative risk, 1.40; 95% confidence interval, 1.15-1.70; P = .0007) and psoriasis groups (adjusted RR, 1.53; 95% CI, 1.19-1.97; P = .0009). In the PsA group, risk of CVD (adjusted RR, 1.24; 95% CI, 0.99-1.56; P = .06), IHD (adjusted RR, 1.27; 95% CI, 1.05-1.54; P = .02), and PVD (adjusted RR, 1.40; 95% CI, 1.02-1.92; P = .04) were significantly higher than in the general population but not when compared with the psoriasis group. The overall risk of cardiovascular disease (including CVD, IHD, and PVD) for the PsA group was significantly higher (adjusted RR, 1.29; 95% CI, 1.12-1.48; P = .0005), compared with the general population.
“These results support the proposal in existing clinical guidelines that, in order to reduce cardiovascular risk in patients with PsA, it is important to treat inflammatory disease as well as to screen and treat traditional risk factors early in the disease course,” Ms. Charlton and her colleagues wrote in their study.
This study was funded by a grant from the National Institute for Health Research in the United Kingdom. The authors reported no relevant conflicts of interest.
SOURCE: Charlton RA et al. Rheumatology. 2018 Sep 6. doi: 10.1093/rheumatology/key286.
Patients with incident psoriatic arthritis are at a significantly increased risk of developing type 2 diabetes when compared against patients with psoriasis alone and with the general population, according to recent research published in Rheumatology.
Rachel Charlton, PhD, of the department of pharmacy and pharmacology at the University of Bath (England), and her colleagues performed an analysis of 6,783 incident cases of psoriatic arthritis (PsA) from the U.K. Clinical Practice Research Datalink who were diagnosed during 1998-2014. Patients were between 18 years and 89 years old with a median age of 49 years at PsA diagnosis.
In the study, the researchers randomly matched PsA cases at a 1:4 ratio to either a cohort of general population patients with no PsA, psoriasis, or inflammatory arthritis or a cohort of patients with psoriasis but no PsA or inflammatory arthritis. Patients were followed from match to the point where they either no longer met inclusion criteria for the cohort or received a diagnosis of type 2 diabetes, cerebrovascular disease (CVD), ischemic heart disease (IHD), or peripheral vascular disease (PVD) with a mean follow-up duration of approximately 5.5 years across all patient groups.
Patients in the PsA group had a significantly higher incidence of type 2 diabetes, compared with the general population (adjusted relative risk, 1.40; 95% confidence interval, 1.15-1.70; P = .0007) and psoriasis groups (adjusted RR, 1.53; 95% CI, 1.19-1.97; P = .0009). In the PsA group, risk of CVD (adjusted RR, 1.24; 95% CI, 0.99-1.56; P = .06), IHD (adjusted RR, 1.27; 95% CI, 1.05-1.54; P = .02), and PVD (adjusted RR, 1.40; 95% CI, 1.02-1.92; P = .04) were significantly higher than in the general population but not when compared with the psoriasis group. The overall risk of cardiovascular disease (including CVD, IHD, and PVD) for the PsA group was significantly higher (adjusted RR, 1.29; 95% CI, 1.12-1.48; P = .0005), compared with the general population.
“These results support the proposal in existing clinical guidelines that, in order to reduce cardiovascular risk in patients with PsA, it is important to treat inflammatory disease as well as to screen and treat traditional risk factors early in the disease course,” Ms. Charlton and her colleagues wrote in their study.
This study was funded by a grant from the National Institute for Health Research in the United Kingdom. The authors reported no relevant conflicts of interest.
SOURCE: Charlton RA et al. Rheumatology. 2018 Sep 6. doi: 10.1093/rheumatology/key286.
FROM RHEUMATOLOGY
Key clinical point: It is important to treat inflammatory disease as well as to screen and treat traditional cardiovascular risk factors early in the course of PsA.
Major finding: (adjusted RR = 1.53) and a general population control group (adjusted RR = 1.40).
Study details: An analysis of 6,783 patients with psoriatic arthritis in the U.K. Clinical Practice Research Datalink who were diagnosed between 1998 and 2014.
Disclosures: This study was funded by a grant from the National Institute for Health Research in the United Kingdom. The authors reported no relevant conflicts of interest.
Source: Charlton RA et al. Rheumatology. 2018 Sep 6. doi: 10.1093/rheumatology/key286.
Johnson v. Monsanto: Roundup and product liability
QUESTION: A groundskeeper alleges he developed terminal lymphoma from the use of the weed killer Roundup. A homeowner injures his leg while using a lawnmower. One thousand litigants allege Lipitor caused them to develop diabetes. A patient sues his doctor for a serious allergic reaction to an antibiotic.
Which of the following statements is incorrect?
A. These are all examples of product liability.
B. If successful, the groundskeeper may be awarded hundreds of millions of dollars in damages.
C. The homeowner has a cause of action against the manufacturer, even if it’s a borrowed lawnmower from a neighbor.
D. It is now harder to include multiple plaintiffs in a class-action lawsuit.
E. The “learned intermediary doctrine” immunizes the drug manufacturer from liability for a patient’s allergic reaction.
ANSWER: A. Johnson v. Monsanto was a recent case in San Francisco that resulted in a verdict for the plaintiff to the tune of $289 million.1 DeWayne Johnson, a 46-year-old groundskeeper, had developed non-Hodgkin lymphoma after using the weed killer, Roundup, to treat the school grounds, sometimes spraying the herbicide for several hours a day. Mr. Johnson alleged that Roundup’s active ingredient, glyphosate, is a known carcinogen, and that Monsanto, its manufacturer, failed to provide appropriate warning regarding this dangerous product.
The judge in the case allowed into evidence internal emails and experts’ warnings, as well as a critical 2015 position paper of the World Health Organization’s international agency for research on cancer, which classified glyphosate as “probably carcinogenic to humans.”2 Yet the herbicide, used widely in households and in commerce, is registered in 130 countries and approved for use on more than 100 crops. It was the first such case involving Roundup.
At trial, the jury unanimously found that Roundup was a substantial contributing factor in causing Mr. Johnson’s malignancy, that Monsanto failed to warn him of its health hazards (marketing “defect”), and that it knew or should have known that its product was unreasonably dangerous. The main portion, $250 million (of the $289 million award), was awarded as punitive damages.
Tort damages are of two types: compensatory and punitive. The former is to compensate the victim for past and future losses such as wages and medical expenses, pain and suffering, and/or emotional distress. On the other hand, punitive damages, also called exemplary damages, are awarded where there is a reckless, willful, or wanton disregard of the obvious risk of harm.
The case is currently under appeal. Meanwhile, another Roundup trial will soon take place in St. Louis, and the company is facing a class-action suit in U.S. district court in San Francisco, as well as several thousand claims in state courts throughout the country.
Johnson v. Monsanto is a typical product liability action. According to Section 102(2) of the Uniform Product Liability Act, product liability includes “all claims or action brought for personal injury, death, or property damage caused by the manufacture, design, formula, preparation, assembly, installation, testing, warnings, instructions, marketing, packaging, or labeling of any product.”
There are basically three legal theories in a product liability claim: negligence, breach of warranty, and strict product liability. The latter is the most favored by plaintiffs, as there is no need to prove fault or warranty.
In a seminal case in 1963, William Greenman was injured when he used a power tool that was given to him as a gift.3 He sued the manufacturer, although there was no direct contract of warranty between him and the manufacturer, as he did not make the purchase himself.
The California Supreme Court went beyond the law of contracts and negligence by introducing the notion of strict liability, which centers on whether a product is defective and unreasonably dangerous. It holds that a professional supplier who sells a product that is both defective and unreasonably dangerous is strictly liable to foreseeable plaintiffs.
“Defective” is usually defined as product quality that is less than what a reasonable consumer expects. It can be a design, manufacturing, or marketing defect, the latter instance typically showing up as a failure to warn. “Unreasonably dangerous” is a conclusion that the risks that result from its condition outweigh the product’s advantages.
Product liability lawsuits commonly involve pharmaceutical products and medical devices. Recent examples are suits against Pfizer over Lipitor’s alleged role as a cause of diabetes and against Johnson & Johnson over its talcum products purportedly causing ovarian cancer.4
When the same product injures multiple plaintiffs, they may band together to file a common legal action against the manufacturer. This is called a class-action suit, and will proceed if it is certified to satisfy four prerequisites: numerosity, commonality, typicality, and adequacy.5 A class-action lawsuit, governed by Rule 23 of the Federal Rules of Civil Procedure, describes a legal cause of action where a representative plaintiff asserts claims on behalf of a large class of similarly injured members, who then give up their rights to pursue an individual lawsuit. It confers several advantages upon the plaintiffs, including the potential of higher damages.
A recent U.S. Supreme Court decision has, however, put a damper on class-action suits by tightening the jurisdictional requirement.6
The case involved Bristol-Myers Squibb, which was sued in California by several hundred individuals from 33 states for injuries from the platelet inhibitor Plavix (clopidogrel). The issue was whether non–California residents could sue in that state for injuries incurred elsewhere.
In a 8-1 decision, the U.S. Supreme Court held that California courts did not have specific jurisdiction to hear the claims of nonresidents without identifying an adequate link between the state and the nonresidents’ claims, as they weren’t prescribed Plavix in the state, didn’t buy or take the drug there, and weren’t injured by the drug there.
Finally, note that, should a doctor fail to warn an injured patient of a known medication risk, the patient may have a claim against the doctor – but usually not against the drug manufacturer. This is termed the “learned-intermediary doctrine.” The justification is that manufacturers can reasonably rely on the treating doctor to warn of adverse effects, which are disclosed to the profession through its sales reps and in its package insert and the Physician’s Desk Reference. The treating doctor, in turn, is expected to use his or her professional judgment to adequately warn the patient. It is simply not feasible for the manufacturer to directly warn every patient without usurping the doctor-patient relationship.
Such lawsuits fall in the common category of medical negligence and lack of informed consent, and are not considered a product liability action.
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. For additional information, readers may contact the author at siang@hawaii.edu.
References
1. DeWayne Johnson v. Monsanto Co., Superior Court of California, County of San Francisco, Case No. CGC-16-550128, June 18, 2018.
2. The Monsanto Papers: Roundup (Glyphosate) Cancer Case Key Documents & Analysis. Available at usrtk.org.
3. Greenman v. Yuba Power Products Inc., 377 P.2d 897 (Cal. 1963).
4. “Defective and unreasonably dangerous,” Internal Medicine News, Nov. 4, 2014.
5. “Class-action lawsuits,” Internal Medicine News, April 1, 2015.
6. Bristol-Myers Squibb Co. v. Superior Court of California, 582 U.S. ____ (2017).
QUESTION: A groundskeeper alleges he developed terminal lymphoma from the use of the weed killer Roundup. A homeowner injures his leg while using a lawnmower. One thousand litigants allege Lipitor caused them to develop diabetes. A patient sues his doctor for a serious allergic reaction to an antibiotic.
Which of the following statements is incorrect?
A. These are all examples of product liability.
B. If successful, the groundskeeper may be awarded hundreds of millions of dollars in damages.
C. The homeowner has a cause of action against the manufacturer, even if it’s a borrowed lawnmower from a neighbor.
D. It is now harder to include multiple plaintiffs in a class-action lawsuit.
E. The “learned intermediary doctrine” immunizes the drug manufacturer from liability for a patient’s allergic reaction.
ANSWER: A. Johnson v. Monsanto was a recent case in San Francisco that resulted in a verdict for the plaintiff to the tune of $289 million.1 DeWayne Johnson, a 46-year-old groundskeeper, had developed non-Hodgkin lymphoma after using the weed killer, Roundup, to treat the school grounds, sometimes spraying the herbicide for several hours a day. Mr. Johnson alleged that Roundup’s active ingredient, glyphosate, is a known carcinogen, and that Monsanto, its manufacturer, failed to provide appropriate warning regarding this dangerous product.
The judge in the case allowed into evidence internal emails and experts’ warnings, as well as a critical 2015 position paper of the World Health Organization’s international agency for research on cancer, which classified glyphosate as “probably carcinogenic to humans.”2 Yet the herbicide, used widely in households and in commerce, is registered in 130 countries and approved for use on more than 100 crops. It was the first such case involving Roundup.
At trial, the jury unanimously found that Roundup was a substantial contributing factor in causing Mr. Johnson’s malignancy, that Monsanto failed to warn him of its health hazards (marketing “defect”), and that it knew or should have known that its product was unreasonably dangerous. The main portion, $250 million (of the $289 million award), was awarded as punitive damages.
Tort damages are of two types: compensatory and punitive. The former is to compensate the victim for past and future losses such as wages and medical expenses, pain and suffering, and/or emotional distress. On the other hand, punitive damages, also called exemplary damages, are awarded where there is a reckless, willful, or wanton disregard of the obvious risk of harm.
The case is currently under appeal. Meanwhile, another Roundup trial will soon take place in St. Louis, and the company is facing a class-action suit in U.S. district court in San Francisco, as well as several thousand claims in state courts throughout the country.
Johnson v. Monsanto is a typical product liability action. According to Section 102(2) of the Uniform Product Liability Act, product liability includes “all claims or action brought for personal injury, death, or property damage caused by the manufacture, design, formula, preparation, assembly, installation, testing, warnings, instructions, marketing, packaging, or labeling of any product.”
There are basically three legal theories in a product liability claim: negligence, breach of warranty, and strict product liability. The latter is the most favored by plaintiffs, as there is no need to prove fault or warranty.
In a seminal case in 1963, William Greenman was injured when he used a power tool that was given to him as a gift.3 He sued the manufacturer, although there was no direct contract of warranty between him and the manufacturer, as he did not make the purchase himself.
The California Supreme Court went beyond the law of contracts and negligence by introducing the notion of strict liability, which centers on whether a product is defective and unreasonably dangerous. It holds that a professional supplier who sells a product that is both defective and unreasonably dangerous is strictly liable to foreseeable plaintiffs.
“Defective” is usually defined as product quality that is less than what a reasonable consumer expects. It can be a design, manufacturing, or marketing defect, the latter instance typically showing up as a failure to warn. “Unreasonably dangerous” is a conclusion that the risks that result from its condition outweigh the product’s advantages.
Product liability lawsuits commonly involve pharmaceutical products and medical devices. Recent examples are suits against Pfizer over Lipitor’s alleged role as a cause of diabetes and against Johnson & Johnson over its talcum products purportedly causing ovarian cancer.4
When the same product injures multiple plaintiffs, they may band together to file a common legal action against the manufacturer. This is called a class-action suit, and will proceed if it is certified to satisfy four prerequisites: numerosity, commonality, typicality, and adequacy.5 A class-action lawsuit, governed by Rule 23 of the Federal Rules of Civil Procedure, describes a legal cause of action where a representative plaintiff asserts claims on behalf of a large class of similarly injured members, who then give up their rights to pursue an individual lawsuit. It confers several advantages upon the plaintiffs, including the potential of higher damages.
A recent U.S. Supreme Court decision has, however, put a damper on class-action suits by tightening the jurisdictional requirement.6
The case involved Bristol-Myers Squibb, which was sued in California by several hundred individuals from 33 states for injuries from the platelet inhibitor Plavix (clopidogrel). The issue was whether non–California residents could sue in that state for injuries incurred elsewhere.
In a 8-1 decision, the U.S. Supreme Court held that California courts did not have specific jurisdiction to hear the claims of nonresidents without identifying an adequate link between the state and the nonresidents’ claims, as they weren’t prescribed Plavix in the state, didn’t buy or take the drug there, and weren’t injured by the drug there.
Finally, note that, should a doctor fail to warn an injured patient of a known medication risk, the patient may have a claim against the doctor – but usually not against the drug manufacturer. This is termed the “learned-intermediary doctrine.” The justification is that manufacturers can reasonably rely on the treating doctor to warn of adverse effects, which are disclosed to the profession through its sales reps and in its package insert and the Physician’s Desk Reference. The treating doctor, in turn, is expected to use his or her professional judgment to adequately warn the patient. It is simply not feasible for the manufacturer to directly warn every patient without usurping the doctor-patient relationship.
Such lawsuits fall in the common category of medical negligence and lack of informed consent, and are not considered a product liability action.
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. For additional information, readers may contact the author at siang@hawaii.edu.
References
1. DeWayne Johnson v. Monsanto Co., Superior Court of California, County of San Francisco, Case No. CGC-16-550128, June 18, 2018.
2. The Monsanto Papers: Roundup (Glyphosate) Cancer Case Key Documents & Analysis. Available at usrtk.org.
3. Greenman v. Yuba Power Products Inc., 377 P.2d 897 (Cal. 1963).
4. “Defective and unreasonably dangerous,” Internal Medicine News, Nov. 4, 2014.
5. “Class-action lawsuits,” Internal Medicine News, April 1, 2015.
6. Bristol-Myers Squibb Co. v. Superior Court of California, 582 U.S. ____ (2017).
QUESTION: A groundskeeper alleges he developed terminal lymphoma from the use of the weed killer Roundup. A homeowner injures his leg while using a lawnmower. One thousand litigants allege Lipitor caused them to develop diabetes. A patient sues his doctor for a serious allergic reaction to an antibiotic.
Which of the following statements is incorrect?
A. These are all examples of product liability.
B. If successful, the groundskeeper may be awarded hundreds of millions of dollars in damages.
C. The homeowner has a cause of action against the manufacturer, even if it’s a borrowed lawnmower from a neighbor.
D. It is now harder to include multiple plaintiffs in a class-action lawsuit.
E. The “learned intermediary doctrine” immunizes the drug manufacturer from liability for a patient’s allergic reaction.
ANSWER: A. Johnson v. Monsanto was a recent case in San Francisco that resulted in a verdict for the plaintiff to the tune of $289 million.1 DeWayne Johnson, a 46-year-old groundskeeper, had developed non-Hodgkin lymphoma after using the weed killer, Roundup, to treat the school grounds, sometimes spraying the herbicide for several hours a day. Mr. Johnson alleged that Roundup’s active ingredient, glyphosate, is a known carcinogen, and that Monsanto, its manufacturer, failed to provide appropriate warning regarding this dangerous product.
The judge in the case allowed into evidence internal emails and experts’ warnings, as well as a critical 2015 position paper of the World Health Organization’s international agency for research on cancer, which classified glyphosate as “probably carcinogenic to humans.”2 Yet the herbicide, used widely in households and in commerce, is registered in 130 countries and approved for use on more than 100 crops. It was the first such case involving Roundup.
At trial, the jury unanimously found that Roundup was a substantial contributing factor in causing Mr. Johnson’s malignancy, that Monsanto failed to warn him of its health hazards (marketing “defect”), and that it knew or should have known that its product was unreasonably dangerous. The main portion, $250 million (of the $289 million award), was awarded as punitive damages.
Tort damages are of two types: compensatory and punitive. The former is to compensate the victim for past and future losses such as wages and medical expenses, pain and suffering, and/or emotional distress. On the other hand, punitive damages, also called exemplary damages, are awarded where there is a reckless, willful, or wanton disregard of the obvious risk of harm.
The case is currently under appeal. Meanwhile, another Roundup trial will soon take place in St. Louis, and the company is facing a class-action suit in U.S. district court in San Francisco, as well as several thousand claims in state courts throughout the country.
Johnson v. Monsanto is a typical product liability action. According to Section 102(2) of the Uniform Product Liability Act, product liability includes “all claims or action brought for personal injury, death, or property damage caused by the manufacture, design, formula, preparation, assembly, installation, testing, warnings, instructions, marketing, packaging, or labeling of any product.”
There are basically three legal theories in a product liability claim: negligence, breach of warranty, and strict product liability. The latter is the most favored by plaintiffs, as there is no need to prove fault or warranty.
In a seminal case in 1963, William Greenman was injured when he used a power tool that was given to him as a gift.3 He sued the manufacturer, although there was no direct contract of warranty between him and the manufacturer, as he did not make the purchase himself.
The California Supreme Court went beyond the law of contracts and negligence by introducing the notion of strict liability, which centers on whether a product is defective and unreasonably dangerous. It holds that a professional supplier who sells a product that is both defective and unreasonably dangerous is strictly liable to foreseeable plaintiffs.
“Defective” is usually defined as product quality that is less than what a reasonable consumer expects. It can be a design, manufacturing, or marketing defect, the latter instance typically showing up as a failure to warn. “Unreasonably dangerous” is a conclusion that the risks that result from its condition outweigh the product’s advantages.
Product liability lawsuits commonly involve pharmaceutical products and medical devices. Recent examples are suits against Pfizer over Lipitor’s alleged role as a cause of diabetes and against Johnson & Johnson over its talcum products purportedly causing ovarian cancer.4
When the same product injures multiple plaintiffs, they may band together to file a common legal action against the manufacturer. This is called a class-action suit, and will proceed if it is certified to satisfy four prerequisites: numerosity, commonality, typicality, and adequacy.5 A class-action lawsuit, governed by Rule 23 of the Federal Rules of Civil Procedure, describes a legal cause of action where a representative plaintiff asserts claims on behalf of a large class of similarly injured members, who then give up their rights to pursue an individual lawsuit. It confers several advantages upon the plaintiffs, including the potential of higher damages.
A recent U.S. Supreme Court decision has, however, put a damper on class-action suits by tightening the jurisdictional requirement.6
The case involved Bristol-Myers Squibb, which was sued in California by several hundred individuals from 33 states for injuries from the platelet inhibitor Plavix (clopidogrel). The issue was whether non–California residents could sue in that state for injuries incurred elsewhere.
In a 8-1 decision, the U.S. Supreme Court held that California courts did not have specific jurisdiction to hear the claims of nonresidents without identifying an adequate link between the state and the nonresidents’ claims, as they weren’t prescribed Plavix in the state, didn’t buy or take the drug there, and weren’t injured by the drug there.
Finally, note that, should a doctor fail to warn an injured patient of a known medication risk, the patient may have a claim against the doctor – but usually not against the drug manufacturer. This is termed the “learned-intermediary doctrine.” The justification is that manufacturers can reasonably rely on the treating doctor to warn of adverse effects, which are disclosed to the profession through its sales reps and in its package insert and the Physician’s Desk Reference. The treating doctor, in turn, is expected to use his or her professional judgment to adequately warn the patient. It is simply not feasible for the manufacturer to directly warn every patient without usurping the doctor-patient relationship.
Such lawsuits fall in the common category of medical negligence and lack of informed consent, and are not considered a product liability action.
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. For additional information, readers may contact the author at siang@hawaii.edu.
References
1. DeWayne Johnson v. Monsanto Co., Superior Court of California, County of San Francisco, Case No. CGC-16-550128, June 18, 2018.
2. The Monsanto Papers: Roundup (Glyphosate) Cancer Case Key Documents & Analysis. Available at usrtk.org.
3. Greenman v. Yuba Power Products Inc., 377 P.2d 897 (Cal. 1963).
4. “Defective and unreasonably dangerous,” Internal Medicine News, Nov. 4, 2014.
5. “Class-action lawsuits,” Internal Medicine News, April 1, 2015.
6. Bristol-Myers Squibb Co. v. Superior Court of California, 582 U.S. ____ (2017).
CME: Current Trends in Multiple Myeloma: Customizing Treatment Strategies and Optimizing Outcomes
Click here to read the digital edition of the supplement
This supplement is supported by an educational education grant from Celgene and Janssen Biotech, Inc.
Topics Include:
- Advances in diagnosis and prognosis
- Management of the newly diagnosed patient
- Treatment advances and current management strategies for relapsed/refractory patients
- Future treatment directions
About the Author
Thomas R. Chauncey, MD, PhD
Associate Professor of Medicine and Oncology
University of Washington
Associate Member
Fred Hutchinson Cancer Research Center
Director, Marrow Transplant Unit
VA Puget Sound Health Care System
Seattle, WA
Click here to read the digital edition of the supplement
Click here to read the digital edition of the supplement

This supplement is supported by an educational education grant from Celgene and Janssen Biotech, Inc.
Topics Include:
- Advances in diagnosis and prognosis
- Management of the newly diagnosed patient
- Treatment advances and current management strategies for relapsed/refractory patients
- Future treatment directions
About the Author
Thomas R. Chauncey, MD, PhD
Associate Professor of Medicine and Oncology
University of Washington
Associate Member
Fred Hutchinson Cancer Research Center
Director, Marrow Transplant Unit
VA Puget Sound Health Care System
Seattle, WA
Click here to read the digital edition of the supplement
Click here to read the digital edition of the supplement

This supplement is supported by an educational education grant from Celgene and Janssen Biotech, Inc.
Topics Include:
- Advances in diagnosis and prognosis
- Management of the newly diagnosed patient
- Treatment advances and current management strategies for relapsed/refractory patients
- Future treatment directions
About the Author
Thomas R. Chauncey, MD, PhD
Associate Professor of Medicine and Oncology
University of Washington
Associate Member
Fred Hutchinson Cancer Research Center
Director, Marrow Transplant Unit
VA Puget Sound Health Care System
Seattle, WA
Click here to read the digital edition of the supplement
Kymriah cost effectiveness depends on long-term outcomes
The cost-effectiveness of tisagenlecleucel (Kymriah) depends on long-term clinical outcomes, which are presently unknown, according to investigators.
If the long-term outcomes are more modest than clinical trials suggest, then payers may be unwilling to cover the costly therapy, reported John K. Lin, MD, of the Center for Primary Care and Outcomes Research at Stanford (Calif.) University, and his colleagues. Lowering the price or setting up an outcomes-based pricing structure may be necessary to get insurers to cover the therapy.
Tisagenlecleucel is an anti-CD19 chimeric antigen receptor (CAR) T-cell therapy that was approved by the Food and Drug Administration in August 2017 for relapsed or refractory pediatric B-cell acute lymphoblastic leukemia (ALL). In 2018, the FDA expanded the indication for tisagenlecleucel to include adults with relapsed or refractory large B-cell lymphoma, though outcomes from lymphoma trials are not analyzed in the current study.
At a wholesale acquisition cost of $475,000 per infusion, it is the most expensive existing oncology therapy to date, and can be accompanied by expensive, potentially fatal adverse effects. However, clinical trials suggest that tisagenlecleucel can offer years of relapse-free remission, thereby allowing patients to forgo other expensive therapies such as hematopoietic stem cell transplantation (HSCT).
“Although tisagenlecleucel-induced remission rates are promising, compared with those of established therapies (greater than 80% vs. less than 50%), only short-term follow-up data currently exist,” the investigators wrote in the Journal of Clinical Oncology. “Given the high cost and broad applicability in other malignancies of tisagenlecleucel, a pressing question for policy makers, payers, patients, and clinicians is whether the cost of therapy represents reasonable value.”
The study used a Markov model to assess various long-term clinical outcome rates and cost thresholds of tisagenlecleucel. The lifetime cost of therapy was assessed and compared with costs of existing therapies.
The results showed that a 5-year relapse free survival rate of 40% would make the present cost ($475,000) of tisagenlecleucel economically reasonable. In this scenario, the increased life expectancy would be 12.1 years and would result in an additional 5.07 quality-adjusted life years (QALY) gained at a cost of $61,000 per QALY, compared with blinatumomab.
But if long-term outcomes are less favorable, tisagenlecleucel becomes much less cost effective. A 5-year relapse-free survival rate of 20% would drop increased life expectancy to 3.8 years, resulting in 1.80 QALYs gained and raising the cost to $151,000 per QALY.
“Our results suggest that at tisagenlecleucel’s current price and payment structure, its economic value is uncertain,” the investigators wrote.
They suggested a price drop to $200,000 or $350,000, which would allow the drug to remain cost effective even in a worse-case scenario, in which patients relapse and tisagenlecleucel is a bridge to transplant. Another option is to move to outcomes-based pricing. Making payment conditional on 7 months of remission would make the treatment cost effective, according to the analysis.
“Price reductions of tisagenlecleucel or payment only for longer-term remissions would favorably influence cost-effectiveness, even if long-term clinical outcomes are modest,” the investigators wrote.
The study was funded by a Veterans Affairs Office of Academic Affiliations advanced fellowship in health service and research development, and a National Center for Advancing Translational Science Clinical and Translational Science Award. One of the study coauthors reported consulting and research funding from Novartis.
SOURCE: Lin et al. J Clin Oncol. 2018 Sep 13. doi: 10.1200/JCO.2018.79.0642.
The cost-effectiveness of tisagenlecleucel (Kymriah) depends on long-term clinical outcomes, which are presently unknown, according to investigators.
If the long-term outcomes are more modest than clinical trials suggest, then payers may be unwilling to cover the costly therapy, reported John K. Lin, MD, of the Center for Primary Care and Outcomes Research at Stanford (Calif.) University, and his colleagues. Lowering the price or setting up an outcomes-based pricing structure may be necessary to get insurers to cover the therapy.
Tisagenlecleucel is an anti-CD19 chimeric antigen receptor (CAR) T-cell therapy that was approved by the Food and Drug Administration in August 2017 for relapsed or refractory pediatric B-cell acute lymphoblastic leukemia (ALL). In 2018, the FDA expanded the indication for tisagenlecleucel to include adults with relapsed or refractory large B-cell lymphoma, though outcomes from lymphoma trials are not analyzed in the current study.
At a wholesale acquisition cost of $475,000 per infusion, it is the most expensive existing oncology therapy to date, and can be accompanied by expensive, potentially fatal adverse effects. However, clinical trials suggest that tisagenlecleucel can offer years of relapse-free remission, thereby allowing patients to forgo other expensive therapies such as hematopoietic stem cell transplantation (HSCT).
“Although tisagenlecleucel-induced remission rates are promising, compared with those of established therapies (greater than 80% vs. less than 50%), only short-term follow-up data currently exist,” the investigators wrote in the Journal of Clinical Oncology. “Given the high cost and broad applicability in other malignancies of tisagenlecleucel, a pressing question for policy makers, payers, patients, and clinicians is whether the cost of therapy represents reasonable value.”
The study used a Markov model to assess various long-term clinical outcome rates and cost thresholds of tisagenlecleucel. The lifetime cost of therapy was assessed and compared with costs of existing therapies.
The results showed that a 5-year relapse free survival rate of 40% would make the present cost ($475,000) of tisagenlecleucel economically reasonable. In this scenario, the increased life expectancy would be 12.1 years and would result in an additional 5.07 quality-adjusted life years (QALY) gained at a cost of $61,000 per QALY, compared with blinatumomab.
But if long-term outcomes are less favorable, tisagenlecleucel becomes much less cost effective. A 5-year relapse-free survival rate of 20% would drop increased life expectancy to 3.8 years, resulting in 1.80 QALYs gained and raising the cost to $151,000 per QALY.
“Our results suggest that at tisagenlecleucel’s current price and payment structure, its economic value is uncertain,” the investigators wrote.
They suggested a price drop to $200,000 or $350,000, which would allow the drug to remain cost effective even in a worse-case scenario, in which patients relapse and tisagenlecleucel is a bridge to transplant. Another option is to move to outcomes-based pricing. Making payment conditional on 7 months of remission would make the treatment cost effective, according to the analysis.
“Price reductions of tisagenlecleucel or payment only for longer-term remissions would favorably influence cost-effectiveness, even if long-term clinical outcomes are modest,” the investigators wrote.
The study was funded by a Veterans Affairs Office of Academic Affiliations advanced fellowship in health service and research development, and a National Center for Advancing Translational Science Clinical and Translational Science Award. One of the study coauthors reported consulting and research funding from Novartis.
SOURCE: Lin et al. J Clin Oncol. 2018 Sep 13. doi: 10.1200/JCO.2018.79.0642.
The cost-effectiveness of tisagenlecleucel (Kymriah) depends on long-term clinical outcomes, which are presently unknown, according to investigators.
If the long-term outcomes are more modest than clinical trials suggest, then payers may be unwilling to cover the costly therapy, reported John K. Lin, MD, of the Center for Primary Care and Outcomes Research at Stanford (Calif.) University, and his colleagues. Lowering the price or setting up an outcomes-based pricing structure may be necessary to get insurers to cover the therapy.
Tisagenlecleucel is an anti-CD19 chimeric antigen receptor (CAR) T-cell therapy that was approved by the Food and Drug Administration in August 2017 for relapsed or refractory pediatric B-cell acute lymphoblastic leukemia (ALL). In 2018, the FDA expanded the indication for tisagenlecleucel to include adults with relapsed or refractory large B-cell lymphoma, though outcomes from lymphoma trials are not analyzed in the current study.
At a wholesale acquisition cost of $475,000 per infusion, it is the most expensive existing oncology therapy to date, and can be accompanied by expensive, potentially fatal adverse effects. However, clinical trials suggest that tisagenlecleucel can offer years of relapse-free remission, thereby allowing patients to forgo other expensive therapies such as hematopoietic stem cell transplantation (HSCT).
“Although tisagenlecleucel-induced remission rates are promising, compared with those of established therapies (greater than 80% vs. less than 50%), only short-term follow-up data currently exist,” the investigators wrote in the Journal of Clinical Oncology. “Given the high cost and broad applicability in other malignancies of tisagenlecleucel, a pressing question for policy makers, payers, patients, and clinicians is whether the cost of therapy represents reasonable value.”
The study used a Markov model to assess various long-term clinical outcome rates and cost thresholds of tisagenlecleucel. The lifetime cost of therapy was assessed and compared with costs of existing therapies.
The results showed that a 5-year relapse free survival rate of 40% would make the present cost ($475,000) of tisagenlecleucel economically reasonable. In this scenario, the increased life expectancy would be 12.1 years and would result in an additional 5.07 quality-adjusted life years (QALY) gained at a cost of $61,000 per QALY, compared with blinatumomab.
But if long-term outcomes are less favorable, tisagenlecleucel becomes much less cost effective. A 5-year relapse-free survival rate of 20% would drop increased life expectancy to 3.8 years, resulting in 1.80 QALYs gained and raising the cost to $151,000 per QALY.
“Our results suggest that at tisagenlecleucel’s current price and payment structure, its economic value is uncertain,” the investigators wrote.
They suggested a price drop to $200,000 or $350,000, which would allow the drug to remain cost effective even in a worse-case scenario, in which patients relapse and tisagenlecleucel is a bridge to transplant. Another option is to move to outcomes-based pricing. Making payment conditional on 7 months of remission would make the treatment cost effective, according to the analysis.
“Price reductions of tisagenlecleucel or payment only for longer-term remissions would favorably influence cost-effectiveness, even if long-term clinical outcomes are modest,” the investigators wrote.
The study was funded by a Veterans Affairs Office of Academic Affiliations advanced fellowship in health service and research development, and a National Center for Advancing Translational Science Clinical and Translational Science Award. One of the study coauthors reported consulting and research funding from Novartis.
SOURCE: Lin et al. J Clin Oncol. 2018 Sep 13. doi: 10.1200/JCO.2018.79.0642.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point:
Major finding: If 40% of patients achieve 5-year remission without relapse, then tisagenlecleucel would cost $61,000 per quality-adjusted life year.
Study details: An economic analysis involving tisagenlecleucel costs and clinical trial outcomes.
Disclosures: The study was funded by a Veterans Affairs Office of Academic Affiliations advanced fellowship in health service and research development, and a National Center for Advancing Translational Science Clinical and Translational Science Award. One study coauthor reported consulting and research funding from Novartis.
Source: Lin JK et al. J Clin Oncol. 2018 Sep 13. doi: 10.1200/JCO.2018.79.0642.
Predictors for worse outcomes in low–disease activity RA remain tough to come by
Disease flares are common but are hard to predict in people with rheumatoid arthritis who achieve low disease activity (LDA), and these flares are “undeniably” associated with worse disease activity, quality of life, and radiographic progression, researchers report in an analysis of a prospectively observed cohort.
First author Katie Bechman, MBChB, of the academic rheumatology department at King’s College London, and her colleagues reported online in the Journal of Rheumatology that nearly one-third of patients with RA in stable LDA states, including remission, had flares during a year of follow-up in the observational prospective REMIRA study, similar to previous reports in cohort studies.
The cohort of 152 patients had 28-joint Disease Activity Scores (DAS28) less than 3.2 for at least 1 month apart, and 66% (n = 97) of the cohort fulfilled DAS28 remission criteria (DAS28 less than 2.6). The authors defined disease flare as a DAS28 increase of more than 1.2, compared with baseline, or a DAS28 increase of more than 0.6, compared with baseline, and concurrent DAS28 of 3.2 or greater. A total of 69 (35%) were taking disease-modifying antirheumatic drug monotherapy.
Nearly one-third of RA patients (30%, n = 46) with LDA states experienced a flare during 12 months of follow-up. When limiting the cohort to patients who were in remission at baseline, 25% (n = 24) had at least one flare.
Patients who had a flare experienced significantly worse clinical outcomes at 12 months than did patients in sustained remission, reflected by higher disease activity, worse functional outcomes, and higher radiographic progression scores.
For example, patients who flared had more than a threefold greater risk for erosive progression, defined as new or larger erosions over 1 year on radiographs (hazard ratio, 3.6; 95% confidence interval, 2.77-4.67; P less than .01).
Patients’ baseline values on the Health Assessment Questionnaire-Disability Index – a measure of functional activity that is reflected by difficulties in activities of daily living – proved to be a significant independent predictor of flare (HR, 1.76; 95% CI, 1.05-2.93; P = .03) in multivariate analyses.
The researchers also found that serum biomarkers were modestly correlated with DAS28 at the time of flare. DAS28 and its components significantly correlated with multibiomarker disease activity score and calprotectin at time of flare, but correlation coefficients were low at 0.52 and 0.49, respectively. Two-thirds of flares were not associated with a rise in biomarkers.
“This might be because a flare is defined by worsening of the DAS28 composite score, and an increase in [tender joint count] and [patient’s global assessment] alone may increase the DAS28 score to a sufficient level to define a flare,” the investigators suggested.
“It is possible that a flare event is not solely the result of direct synovial inflammation but may be driven by other pathways, for example chronification of pain due to central sensitization and abnormal regulatory mechanisms. This heterogeneity may partly explain why identifying predictors of flare is challenging,” they said.
“In our study, we have shown that the occurrence of a flare is hard to predict, but undeniably associated with worse clinical outcomes at 12 months,” the researchers concluded.
They suggested that it was possible that two “distinct subtypes” of flare might exist: an “inflammatory” flare that was predominantly driven by an increase in swollen joint count and erythrocyte sedimentation rate, and a “noninflammatory” flare with a disproportionately elevated tender joint count and a high patient global assessment score.
“Differentiating these two flare types may identify potential predictors. Further research is needed to determine whether distinct flares exist and to categorize the potential predictors of each,” they added.
The research was partly funded by the U.K. National Institute for Health Research Biomedical Research Centre at Guy’s and St. Thomas’ NHS Foundation Trust and King’s College London.
SOURCE: Bechman K et al. J Rheumatol. 2018 Sep 1. doi: 10.3899/jrheum.171375.
Disease flares are common but are hard to predict in people with rheumatoid arthritis who achieve low disease activity (LDA), and these flares are “undeniably” associated with worse disease activity, quality of life, and radiographic progression, researchers report in an analysis of a prospectively observed cohort.
First author Katie Bechman, MBChB, of the academic rheumatology department at King’s College London, and her colleagues reported online in the Journal of Rheumatology that nearly one-third of patients with RA in stable LDA states, including remission, had flares during a year of follow-up in the observational prospective REMIRA study, similar to previous reports in cohort studies.
The cohort of 152 patients had 28-joint Disease Activity Scores (DAS28) less than 3.2 for at least 1 month apart, and 66% (n = 97) of the cohort fulfilled DAS28 remission criteria (DAS28 less than 2.6). The authors defined disease flare as a DAS28 increase of more than 1.2, compared with baseline, or a DAS28 increase of more than 0.6, compared with baseline, and concurrent DAS28 of 3.2 or greater. A total of 69 (35%) were taking disease-modifying antirheumatic drug monotherapy.
Nearly one-third of RA patients (30%, n = 46) with LDA states experienced a flare during 12 months of follow-up. When limiting the cohort to patients who were in remission at baseline, 25% (n = 24) had at least one flare.
Patients who had a flare experienced significantly worse clinical outcomes at 12 months than did patients in sustained remission, reflected by higher disease activity, worse functional outcomes, and higher radiographic progression scores.
For example, patients who flared had more than a threefold greater risk for erosive progression, defined as new or larger erosions over 1 year on radiographs (hazard ratio, 3.6; 95% confidence interval, 2.77-4.67; P less than .01).
Patients’ baseline values on the Health Assessment Questionnaire-Disability Index – a measure of functional activity that is reflected by difficulties in activities of daily living – proved to be a significant independent predictor of flare (HR, 1.76; 95% CI, 1.05-2.93; P = .03) in multivariate analyses.
The researchers also found that serum biomarkers were modestly correlated with DAS28 at the time of flare. DAS28 and its components significantly correlated with multibiomarker disease activity score and calprotectin at time of flare, but correlation coefficients were low at 0.52 and 0.49, respectively. Two-thirds of flares were not associated with a rise in biomarkers.
“This might be because a flare is defined by worsening of the DAS28 composite score, and an increase in [tender joint count] and [patient’s global assessment] alone may increase the DAS28 score to a sufficient level to define a flare,” the investigators suggested.
“It is possible that a flare event is not solely the result of direct synovial inflammation but may be driven by other pathways, for example chronification of pain due to central sensitization and abnormal regulatory mechanisms. This heterogeneity may partly explain why identifying predictors of flare is challenging,” they said.
“In our study, we have shown that the occurrence of a flare is hard to predict, but undeniably associated with worse clinical outcomes at 12 months,” the researchers concluded.
They suggested that it was possible that two “distinct subtypes” of flare might exist: an “inflammatory” flare that was predominantly driven by an increase in swollen joint count and erythrocyte sedimentation rate, and a “noninflammatory” flare with a disproportionately elevated tender joint count and a high patient global assessment score.
“Differentiating these two flare types may identify potential predictors. Further research is needed to determine whether distinct flares exist and to categorize the potential predictors of each,” they added.
The research was partly funded by the U.K. National Institute for Health Research Biomedical Research Centre at Guy’s and St. Thomas’ NHS Foundation Trust and King’s College London.
SOURCE: Bechman K et al. J Rheumatol. 2018 Sep 1. doi: 10.3899/jrheum.171375.
Disease flares are common but are hard to predict in people with rheumatoid arthritis who achieve low disease activity (LDA), and these flares are “undeniably” associated with worse disease activity, quality of life, and radiographic progression, researchers report in an analysis of a prospectively observed cohort.
First author Katie Bechman, MBChB, of the academic rheumatology department at King’s College London, and her colleagues reported online in the Journal of Rheumatology that nearly one-third of patients with RA in stable LDA states, including remission, had flares during a year of follow-up in the observational prospective REMIRA study, similar to previous reports in cohort studies.
The cohort of 152 patients had 28-joint Disease Activity Scores (DAS28) less than 3.2 for at least 1 month apart, and 66% (n = 97) of the cohort fulfilled DAS28 remission criteria (DAS28 less than 2.6). The authors defined disease flare as a DAS28 increase of more than 1.2, compared with baseline, or a DAS28 increase of more than 0.6, compared with baseline, and concurrent DAS28 of 3.2 or greater. A total of 69 (35%) were taking disease-modifying antirheumatic drug monotherapy.
Nearly one-third of RA patients (30%, n = 46) with LDA states experienced a flare during 12 months of follow-up. When limiting the cohort to patients who were in remission at baseline, 25% (n = 24) had at least one flare.
Patients who had a flare experienced significantly worse clinical outcomes at 12 months than did patients in sustained remission, reflected by higher disease activity, worse functional outcomes, and higher radiographic progression scores.
For example, patients who flared had more than a threefold greater risk for erosive progression, defined as new or larger erosions over 1 year on radiographs (hazard ratio, 3.6; 95% confidence interval, 2.77-4.67; P less than .01).
Patients’ baseline values on the Health Assessment Questionnaire-Disability Index – a measure of functional activity that is reflected by difficulties in activities of daily living – proved to be a significant independent predictor of flare (HR, 1.76; 95% CI, 1.05-2.93; P = .03) in multivariate analyses.
The researchers also found that serum biomarkers were modestly correlated with DAS28 at the time of flare. DAS28 and its components significantly correlated with multibiomarker disease activity score and calprotectin at time of flare, but correlation coefficients were low at 0.52 and 0.49, respectively. Two-thirds of flares were not associated with a rise in biomarkers.
“This might be because a flare is defined by worsening of the DAS28 composite score, and an increase in [tender joint count] and [patient’s global assessment] alone may increase the DAS28 score to a sufficient level to define a flare,” the investigators suggested.
“It is possible that a flare event is not solely the result of direct synovial inflammation but may be driven by other pathways, for example chronification of pain due to central sensitization and abnormal regulatory mechanisms. This heterogeneity may partly explain why identifying predictors of flare is challenging,” they said.
“In our study, we have shown that the occurrence of a flare is hard to predict, but undeniably associated with worse clinical outcomes at 12 months,” the researchers concluded.
They suggested that it was possible that two “distinct subtypes” of flare might exist: an “inflammatory” flare that was predominantly driven by an increase in swollen joint count and erythrocyte sedimentation rate, and a “noninflammatory” flare with a disproportionately elevated tender joint count and a high patient global assessment score.
“Differentiating these two flare types may identify potential predictors. Further research is needed to determine whether distinct flares exist and to categorize the potential predictors of each,” they added.
The research was partly funded by the U.K. National Institute for Health Research Biomedical Research Centre at Guy’s and St. Thomas’ NHS Foundation Trust and King’s College London.
SOURCE: Bechman K et al. J Rheumatol. 2018 Sep 1. doi: 10.3899/jrheum.171375.
FROM THE JOURNAL OF RHEUMATOLOGY
Key clinical point: Flares occur frequently in RA patients with low disease activity and are associated with worse disease activity, quality of life, and radiographic progression.
Major finding:
Study details: A study of 152 RA patients taking part in the observational prospective REMIRA study who had a stable 28-joint Disease Activity Score (DAS28) less than 3.2.
Disclosures: The research was partly funded by the U.K. National Institute for Health Research Biomedical Research Centre at Guy’s and St. Thomas’ NHS Foundation Trust and King’s College London.
Source: Bechman K et al. J Rheumatol. 2018 Sep 1. doi: 10.3899/jrheum.171375.
Pediatric hypertension linked to troubling MRI changes
CHICAGO – There’s another reason to worry about hypertension in children: cognitive decline later in life.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
In a pilot study, Marc Lande, MD, a professor of pediatric nephrology at the University of Rochester (N.Y.), and his colleagues found similar to what’s found in adults with cognitive impairment from hypertension.
The work is ongoing, but it helps explain the subtle deficits on cognitive testing that have been previously demonstrated in children with hypertension.
“The fact that we are finding anything at this very early stage of disease is striking and somewhat bothersome. The hope is that, by improving blood pressure in children, you can improve subsequent cognition and maybe even delay the onset of dementia further down the road,” Dr. Lande said.
For now, the findings underscore the need to diagnose and manage hypertension in children, but there might be additional treatment implications in the future, especially if blood pressure targets are found that ameliorate the problem.
Dr. Lande explained the issues and the emerging evidence in a video interview at the joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
CHICAGO – There’s another reason to worry about hypertension in children: cognitive decline later in life.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
In a pilot study, Marc Lande, MD, a professor of pediatric nephrology at the University of Rochester (N.Y.), and his colleagues found similar to what’s found in adults with cognitive impairment from hypertension.
The work is ongoing, but it helps explain the subtle deficits on cognitive testing that have been previously demonstrated in children with hypertension.
“The fact that we are finding anything at this very early stage of disease is striking and somewhat bothersome. The hope is that, by improving blood pressure in children, you can improve subsequent cognition and maybe even delay the onset of dementia further down the road,” Dr. Lande said.
For now, the findings underscore the need to diagnose and manage hypertension in children, but there might be additional treatment implications in the future, especially if blood pressure targets are found that ameliorate the problem.
Dr. Lande explained the issues and the emerging evidence in a video interview at the joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
CHICAGO – There’s another reason to worry about hypertension in children: cognitive decline later in life.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
In a pilot study, Marc Lande, MD, a professor of pediatric nephrology at the University of Rochester (N.Y.), and his colleagues found similar to what’s found in adults with cognitive impairment from hypertension.
The work is ongoing, but it helps explain the subtle deficits on cognitive testing that have been previously demonstrated in children with hypertension.
“The fact that we are finding anything at this very early stage of disease is striking and somewhat bothersome. The hope is that, by improving blood pressure in children, you can improve subsequent cognition and maybe even delay the onset of dementia further down the road,” Dr. Lande said.
For now, the findings underscore the need to diagnose and manage hypertension in children, but there might be additional treatment implications in the future, especially if blood pressure targets are found that ameliorate the problem.
Dr. Lande explained the issues and the emerging evidence in a video interview at the joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
REPORTING FROM JOINT HYPERTENSION 2018
Dupilumab efficacy extends to chronic rhinosinusitis/nasal polyposis
PARIS – Severe asthma patients with chronic rhinosinusitis (CRS), nasal polyposis (NP), or both derive more protection from severe exacerbations with the monoclonal antibody dupilumab than do those who do not have these comorbidities, according to a post hoc analysis of a phase 3 trial presented at the annual congress of the European Respiratory Society.
“Dupilumab reduced rates of severe exacerbations and improved FEV1 [forced expiratory volume in 1 second] in patients in asthma patients with or without CRS/NP. In those with CRS/NP, dupilumab reduced symptoms associated with these comorbidities,” reported Ian Pavord, MBBS, statutory chair in respiratory medicine at University of Oxford (England).
The data were drawn from the phase 3 Liberty Asthma Quest trial, which was published earlier this year in the New England Journal of Medicine (2018;378:2486-96). In that study, both the 200-mg and 300-mg dose of dupilumab (Dupixent) administered every 2 weeks was associated with about a 50% reduction in the annualized rate of severe exacerbations relative to placebo (P less than .001 for both doses).
In this new post hoc analysis, response in the 382 patients who entered the study with a history of CRS/NP was compared with the 1,520 without CRS/NP. In the CRS/NP patients, the reductions relative to placebo in the rates of severe exacerbations, defined as 3 or more days of systemic glucocorticoids or visit to an emergency department leading to treatment with systemic glucocorticoids, were 63% and 61% for the 200-mg and 300-mg doses of dupilumab, respectively (both P less than .001).
In the non-CRS/NP arms, the reductions relative to placebo were 42% and 40%, respectively (both P less than .001). The greater relative reductions in the CRS/NP patients were achieved even though they were older (mean age approximately 52 vs. 47 years for non-CRS/NP patients), had a significantly greater number of exacerbations in the past year (P = .027), and had higher baseline fractional exhaled nitric oxide and eosinophil levels (both P less than .001), Dr. Pavord reported.
“The greater asthma severity in the CRS/NP patients in this trial is consistent with that reported previously by others,” Dr. Pavord said.
Although the greater asthma severity may have provided a larger margin for benefit, Dr. Pavord also reported that there were improvements in CRS/NP-specific symptoms as measured with the 22-item Sino-Nasal Outcome Test (SNOT-22). By week 12, the total score reduction in SNOT-22 was approximately 15 points (P less than .05) from baseline for both the 200-mg and 300-mg dupilumab doses. This was significantly greater (P less than .05) relative to modest SNOT-22 reductions in the placebo arms (P less than .05). After 52 weeks, the reduction In SNOT-22 scores were sustained, providing an even greater statistical advantage over placebo (P less than .001).
In addition to greater protection against severe exacerbations and CRS/NP-specific symptoms, dupilumab may offer specific improvements on CRS/NP pathology, according to Dr. Pavord. Although imaging was not part of this study, he noted in particular that previous studies with dupilumab as well as other biologics have shown shrinkage of nasal polyps with treatment.
Dupilumab was similarly well tolerated in those with and without CRS/NP. The most common adverse event was injection site reactions in both groups, Dr. Pavord said.
Calling CRS and NP “important comorbidities” in severe asthma patients, Dr. Pavord said that this analysis should be reassuring for those who with CRS/NP who are being considered for dupilumab. Already approved for treatment of atopic dermatitis, dupilumab, which binds to interleukin-4 (IL-4) and IL-13 receptors, is currently under review for the treatment of moderate to severe asthma.
Dr. Pavord has financial relationships with Aerocrine, Almirall, AstraZeneca, Boehringer Ingelheim, Chiesi, GlaxoSmithKline, Knapp, Merck Sharpe, Novartis, Knapp Teva, RespiVert, and Schering-Plough.
PARIS – Severe asthma patients with chronic rhinosinusitis (CRS), nasal polyposis (NP), or both derive more protection from severe exacerbations with the monoclonal antibody dupilumab than do those who do not have these comorbidities, according to a post hoc analysis of a phase 3 trial presented at the annual congress of the European Respiratory Society.
“Dupilumab reduced rates of severe exacerbations and improved FEV1 [forced expiratory volume in 1 second] in patients in asthma patients with or without CRS/NP. In those with CRS/NP, dupilumab reduced symptoms associated with these comorbidities,” reported Ian Pavord, MBBS, statutory chair in respiratory medicine at University of Oxford (England).
The data were drawn from the phase 3 Liberty Asthma Quest trial, which was published earlier this year in the New England Journal of Medicine (2018;378:2486-96). In that study, both the 200-mg and 300-mg dose of dupilumab (Dupixent) administered every 2 weeks was associated with about a 50% reduction in the annualized rate of severe exacerbations relative to placebo (P less than .001 for both doses).
In this new post hoc analysis, response in the 382 patients who entered the study with a history of CRS/NP was compared with the 1,520 without CRS/NP. In the CRS/NP patients, the reductions relative to placebo in the rates of severe exacerbations, defined as 3 or more days of systemic glucocorticoids or visit to an emergency department leading to treatment with systemic glucocorticoids, were 63% and 61% for the 200-mg and 300-mg doses of dupilumab, respectively (both P less than .001).
In the non-CRS/NP arms, the reductions relative to placebo were 42% and 40%, respectively (both P less than .001). The greater relative reductions in the CRS/NP patients were achieved even though they were older (mean age approximately 52 vs. 47 years for non-CRS/NP patients), had a significantly greater number of exacerbations in the past year (P = .027), and had higher baseline fractional exhaled nitric oxide and eosinophil levels (both P less than .001), Dr. Pavord reported.
“The greater asthma severity in the CRS/NP patients in this trial is consistent with that reported previously by others,” Dr. Pavord said.
Although the greater asthma severity may have provided a larger margin for benefit, Dr. Pavord also reported that there were improvements in CRS/NP-specific symptoms as measured with the 22-item Sino-Nasal Outcome Test (SNOT-22). By week 12, the total score reduction in SNOT-22 was approximately 15 points (P less than .05) from baseline for both the 200-mg and 300-mg dupilumab doses. This was significantly greater (P less than .05) relative to modest SNOT-22 reductions in the placebo arms (P less than .05). After 52 weeks, the reduction In SNOT-22 scores were sustained, providing an even greater statistical advantage over placebo (P less than .001).
In addition to greater protection against severe exacerbations and CRS/NP-specific symptoms, dupilumab may offer specific improvements on CRS/NP pathology, according to Dr. Pavord. Although imaging was not part of this study, he noted in particular that previous studies with dupilumab as well as other biologics have shown shrinkage of nasal polyps with treatment.
Dupilumab was similarly well tolerated in those with and without CRS/NP. The most common adverse event was injection site reactions in both groups, Dr. Pavord said.
Calling CRS and NP “important comorbidities” in severe asthma patients, Dr. Pavord said that this analysis should be reassuring for those who with CRS/NP who are being considered for dupilumab. Already approved for treatment of atopic dermatitis, dupilumab, which binds to interleukin-4 (IL-4) and IL-13 receptors, is currently under review for the treatment of moderate to severe asthma.
Dr. Pavord has financial relationships with Aerocrine, Almirall, AstraZeneca, Boehringer Ingelheim, Chiesi, GlaxoSmithKline, Knapp, Merck Sharpe, Novartis, Knapp Teva, RespiVert, and Schering-Plough.
PARIS – Severe asthma patients with chronic rhinosinusitis (CRS), nasal polyposis (NP), or both derive more protection from severe exacerbations with the monoclonal antibody dupilumab than do those who do not have these comorbidities, according to a post hoc analysis of a phase 3 trial presented at the annual congress of the European Respiratory Society.
“Dupilumab reduced rates of severe exacerbations and improved FEV1 [forced expiratory volume in 1 second] in patients in asthma patients with or without CRS/NP. In those with CRS/NP, dupilumab reduced symptoms associated with these comorbidities,” reported Ian Pavord, MBBS, statutory chair in respiratory medicine at University of Oxford (England).
The data were drawn from the phase 3 Liberty Asthma Quest trial, which was published earlier this year in the New England Journal of Medicine (2018;378:2486-96). In that study, both the 200-mg and 300-mg dose of dupilumab (Dupixent) administered every 2 weeks was associated with about a 50% reduction in the annualized rate of severe exacerbations relative to placebo (P less than .001 for both doses).
In this new post hoc analysis, response in the 382 patients who entered the study with a history of CRS/NP was compared with the 1,520 without CRS/NP. In the CRS/NP patients, the reductions relative to placebo in the rates of severe exacerbations, defined as 3 or more days of systemic glucocorticoids or visit to an emergency department leading to treatment with systemic glucocorticoids, were 63% and 61% for the 200-mg and 300-mg doses of dupilumab, respectively (both P less than .001).
In the non-CRS/NP arms, the reductions relative to placebo were 42% and 40%, respectively (both P less than .001). The greater relative reductions in the CRS/NP patients were achieved even though they were older (mean age approximately 52 vs. 47 years for non-CRS/NP patients), had a significantly greater number of exacerbations in the past year (P = .027), and had higher baseline fractional exhaled nitric oxide and eosinophil levels (both P less than .001), Dr. Pavord reported.
“The greater asthma severity in the CRS/NP patients in this trial is consistent with that reported previously by others,” Dr. Pavord said.
Although the greater asthma severity may have provided a larger margin for benefit, Dr. Pavord also reported that there were improvements in CRS/NP-specific symptoms as measured with the 22-item Sino-Nasal Outcome Test (SNOT-22). By week 12, the total score reduction in SNOT-22 was approximately 15 points (P less than .05) from baseline for both the 200-mg and 300-mg dupilumab doses. This was significantly greater (P less than .05) relative to modest SNOT-22 reductions in the placebo arms (P less than .05). After 52 weeks, the reduction In SNOT-22 scores were sustained, providing an even greater statistical advantage over placebo (P less than .001).
In addition to greater protection against severe exacerbations and CRS/NP-specific symptoms, dupilumab may offer specific improvements on CRS/NP pathology, according to Dr. Pavord. Although imaging was not part of this study, he noted in particular that previous studies with dupilumab as well as other biologics have shown shrinkage of nasal polyps with treatment.
Dupilumab was similarly well tolerated in those with and without CRS/NP. The most common adverse event was injection site reactions in both groups, Dr. Pavord said.
Calling CRS and NP “important comorbidities” in severe asthma patients, Dr. Pavord said that this analysis should be reassuring for those who with CRS/NP who are being considered for dupilumab. Already approved for treatment of atopic dermatitis, dupilumab, which binds to interleukin-4 (IL-4) and IL-13 receptors, is currently under review for the treatment of moderate to severe asthma.
Dr. Pavord has financial relationships with Aerocrine, Almirall, AstraZeneca, Boehringer Ingelheim, Chiesi, GlaxoSmithKline, Knapp, Merck Sharpe, Novartis, Knapp Teva, RespiVert, and Schering-Plough.
REPORTING FROM THE ERS CONGRESS 2018
Key clinical point: In asthma patients with chronic rhinosinusitis and/or nasal polyposis (CRS/NP), dupilumab reduces exacerbations.
Major finding: At 52 weeks, severe exacerbations were reduced 61% in CRS/NP patients and 40% in non-CRS/NP patients (both P less than .001).
Study details: Post hoc analysis of phase 3 trial.
Disclosures: Dr. Pavord has financial relationships with Aerocrine, Almirall, AstraZeneca, Boehringer Ingelheim, Chiesi, GlaxoSmithKline, Knapp, Merck Sharpe, Novartis, Knapp Teva, RespiVert, and Schering-Plough.
Updated IPF guideline refines diagnostic criteria with HRCT
A recently updated guideline for idiopathic pulmonary fibrosis (IPF) provides refined diagnostic criteria in an effort to improve clinical application and diagnostic accuracy.
Of note, the guideline recommends that high-resolution CT (HRCT) patterns be used to dictate management course. The guideline also calls for detailed medical history, serological testing to exclude connective tissue disease, and multidisciplinary discussion. Serum biomarkers are recommended against as a means of distinguishing between IPF and other interstitial lung diseases (ILDs).
“Diagnosing IPF is challenging because these symptoms are nonspecific: They occur with all other interstitial lung diseases and with other respiratory problems,” Ganesh Raghu, MD, chair of the guideline committee and professor of medicine and director of the Center for Interstitial Lung Disease at the University of Washington, Seattle, said in a written statement. “Because drugs may slow the progression of IPF, an early and accurate diagnosis is essential for prompt and appropriate treatment for this fatal disease.”
The 2018 guideline represents a second collaborative effort from the American Thoracic Society, European Respiratory Society, Japanese Respiratory Society, and Latin American Thoracic Society. The guideline committee consisted of 29 clinicians, scientists, and a patient with IPF. They evaluated all IPF-related evidence and rated the quality of findings with the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) system. The first IPF guideline was published 7 years ago; the intervening time has revealed some clinical limitations that the 2018 guideline aims to fix.
“The 2011 guideline provided the first evidence-based, formal criteria for diagnosis of IPF and allowed patients with a well-defined diagnosis of IPF to participate in numerous clinical studies and randomized controlled trials that enhanced our understanding of the disease,” Dr. Raghu said. “However, it became clear that there were significant challenges in ascertaining the diagnosis per the 2011 criteria, and abundant evidence accumulated since then allowed the committee to refine the diagnostic criteria now.”
“This [updated] guideline is intended to help clinicians make an accurate diagnosis of IPF,” the authors wrote in the American Journal of Respiratory and Critical Care Medicine, “and to empower them to implement recommended courses of action in the context of individual patient values and preferences, particularly decisions regarding which diagnostic interventions to pursue.”
While the 2011 guideline did not distinguish between patients with different HRCT patterns, the 2018 guideline emphasizes the use of HRCT. It is now recommended that patients undergo HRCT to determine the pattern of usual interstitial pneumonia (UIP). Broadly, patients exhibit the UIP pattern or one of three possible non-UIP patterns (probable UIP, indeterminate UIP, or an alternative diagnosis). Recommendations are specific for UIP and non-UIP.
Patients with probable UIP, indeterminate UIP, or an alternative diagnosis should undergo bronchoalveolar lavage (BAL) and surgical lung biopsy (SLB). Transbronchial lung biopsy (TBBx) and lung cryobiopsy recommendations were not described in these patients because of a lack of evidence. However, patients with UIP should not undergo BAL, SLB, TBBx, or cryobiopsy.
For all patients, the 2018 guideline recommends that medical histories include environmental exposure and medication use, and that serological testing is performed to exclude connective tissue disease. Multidisciplinary discussion is conditionally recommended for diagnostic decision making, particularly when a patient has probable UIP, indeterminate UIP, or an alternative diagnosis.
Finally, the guideline recommends against the use of serum biomarkers as a means of distinguishing between IPF and other ILDs because of weak data on this point. “For the time being, the guideline panel dismissed serum biomarker measurement as an approach to distinguishing IPF from other ILDs because of the high false-positive and false-negative result rates,” the panel wrote.
“Our hope is that this new guideline will bridge the gap between the experienced IPF experts and general pulmonologists in making a prompt and accurate diagnosis of IPF for the individual unfortunately confronted with the disease,” Dr. Raghu said. “This will allow patients to make well-informed decisions about treatment options and participation in clinical trials.”
Looking to the future, the panel described an “urgent need to refine and validate diagnostic approaches to ILD. These needs can be roughly categorized as investigations into the roles of clinical observations, HRCT, bronchoscopy, histopathology, and biomarkers.” The authors also emphasized “the need to refine prognostic approaches, identify risk factors for the development of IPF, and determine the impact and approach to the diagnosis of comorbid illness in the patient with IPF.”
The authors reported funding from Bellerophon, Gilead, Roche, Sanofi, and others.
SOURCE: Raghu G et al. Am J Respir Crit Care Med. 2018 Sep 1. doi: 10.1164/rccm.201807-1255ST.
A recently updated guideline for idiopathic pulmonary fibrosis (IPF) provides refined diagnostic criteria in an effort to improve clinical application and diagnostic accuracy.
Of note, the guideline recommends that high-resolution CT (HRCT) patterns be used to dictate management course. The guideline also calls for detailed medical history, serological testing to exclude connective tissue disease, and multidisciplinary discussion. Serum biomarkers are recommended against as a means of distinguishing between IPF and other interstitial lung diseases (ILDs).
“Diagnosing IPF is challenging because these symptoms are nonspecific: They occur with all other interstitial lung diseases and with other respiratory problems,” Ganesh Raghu, MD, chair of the guideline committee and professor of medicine and director of the Center for Interstitial Lung Disease at the University of Washington, Seattle, said in a written statement. “Because drugs may slow the progression of IPF, an early and accurate diagnosis is essential for prompt and appropriate treatment for this fatal disease.”
The 2018 guideline represents a second collaborative effort from the American Thoracic Society, European Respiratory Society, Japanese Respiratory Society, and Latin American Thoracic Society. The guideline committee consisted of 29 clinicians, scientists, and a patient with IPF. They evaluated all IPF-related evidence and rated the quality of findings with the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) system. The first IPF guideline was published 7 years ago; the intervening time has revealed some clinical limitations that the 2018 guideline aims to fix.
“The 2011 guideline provided the first evidence-based, formal criteria for diagnosis of IPF and allowed patients with a well-defined diagnosis of IPF to participate in numerous clinical studies and randomized controlled trials that enhanced our understanding of the disease,” Dr. Raghu said. “However, it became clear that there were significant challenges in ascertaining the diagnosis per the 2011 criteria, and abundant evidence accumulated since then allowed the committee to refine the diagnostic criteria now.”
“This [updated] guideline is intended to help clinicians make an accurate diagnosis of IPF,” the authors wrote in the American Journal of Respiratory and Critical Care Medicine, “and to empower them to implement recommended courses of action in the context of individual patient values and preferences, particularly decisions regarding which diagnostic interventions to pursue.”
While the 2011 guideline did not distinguish between patients with different HRCT patterns, the 2018 guideline emphasizes the use of HRCT. It is now recommended that patients undergo HRCT to determine the pattern of usual interstitial pneumonia (UIP). Broadly, patients exhibit the UIP pattern or one of three possible non-UIP patterns (probable UIP, indeterminate UIP, or an alternative diagnosis). Recommendations are specific for UIP and non-UIP.
Patients with probable UIP, indeterminate UIP, or an alternative diagnosis should undergo bronchoalveolar lavage (BAL) and surgical lung biopsy (SLB). Transbronchial lung biopsy (TBBx) and lung cryobiopsy recommendations were not described in these patients because of a lack of evidence. However, patients with UIP should not undergo BAL, SLB, TBBx, or cryobiopsy.
For all patients, the 2018 guideline recommends that medical histories include environmental exposure and medication use, and that serological testing is performed to exclude connective tissue disease. Multidisciplinary discussion is conditionally recommended for diagnostic decision making, particularly when a patient has probable UIP, indeterminate UIP, or an alternative diagnosis.
Finally, the guideline recommends against the use of serum biomarkers as a means of distinguishing between IPF and other ILDs because of weak data on this point. “For the time being, the guideline panel dismissed serum biomarker measurement as an approach to distinguishing IPF from other ILDs because of the high false-positive and false-negative result rates,” the panel wrote.
“Our hope is that this new guideline will bridge the gap between the experienced IPF experts and general pulmonologists in making a prompt and accurate diagnosis of IPF for the individual unfortunately confronted with the disease,” Dr. Raghu said. “This will allow patients to make well-informed decisions about treatment options and participation in clinical trials.”
Looking to the future, the panel described an “urgent need to refine and validate diagnostic approaches to ILD. These needs can be roughly categorized as investigations into the roles of clinical observations, HRCT, bronchoscopy, histopathology, and biomarkers.” The authors also emphasized “the need to refine prognostic approaches, identify risk factors for the development of IPF, and determine the impact and approach to the diagnosis of comorbid illness in the patient with IPF.”
The authors reported funding from Bellerophon, Gilead, Roche, Sanofi, and others.
SOURCE: Raghu G et al. Am J Respir Crit Care Med. 2018 Sep 1. doi: 10.1164/rccm.201807-1255ST.
A recently updated guideline for idiopathic pulmonary fibrosis (IPF) provides refined diagnostic criteria in an effort to improve clinical application and diagnostic accuracy.
Of note, the guideline recommends that high-resolution CT (HRCT) patterns be used to dictate management course. The guideline also calls for detailed medical history, serological testing to exclude connective tissue disease, and multidisciplinary discussion. Serum biomarkers are recommended against as a means of distinguishing between IPF and other interstitial lung diseases (ILDs).
“Diagnosing IPF is challenging because these symptoms are nonspecific: They occur with all other interstitial lung diseases and with other respiratory problems,” Ganesh Raghu, MD, chair of the guideline committee and professor of medicine and director of the Center for Interstitial Lung Disease at the University of Washington, Seattle, said in a written statement. “Because drugs may slow the progression of IPF, an early and accurate diagnosis is essential for prompt and appropriate treatment for this fatal disease.”
The 2018 guideline represents a second collaborative effort from the American Thoracic Society, European Respiratory Society, Japanese Respiratory Society, and Latin American Thoracic Society. The guideline committee consisted of 29 clinicians, scientists, and a patient with IPF. They evaluated all IPF-related evidence and rated the quality of findings with the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) system. The first IPF guideline was published 7 years ago; the intervening time has revealed some clinical limitations that the 2018 guideline aims to fix.
“The 2011 guideline provided the first evidence-based, formal criteria for diagnosis of IPF and allowed patients with a well-defined diagnosis of IPF to participate in numerous clinical studies and randomized controlled trials that enhanced our understanding of the disease,” Dr. Raghu said. “However, it became clear that there were significant challenges in ascertaining the diagnosis per the 2011 criteria, and abundant evidence accumulated since then allowed the committee to refine the diagnostic criteria now.”
“This [updated] guideline is intended to help clinicians make an accurate diagnosis of IPF,” the authors wrote in the American Journal of Respiratory and Critical Care Medicine, “and to empower them to implement recommended courses of action in the context of individual patient values and preferences, particularly decisions regarding which diagnostic interventions to pursue.”
While the 2011 guideline did not distinguish between patients with different HRCT patterns, the 2018 guideline emphasizes the use of HRCT. It is now recommended that patients undergo HRCT to determine the pattern of usual interstitial pneumonia (UIP). Broadly, patients exhibit the UIP pattern or one of three possible non-UIP patterns (probable UIP, indeterminate UIP, or an alternative diagnosis). Recommendations are specific for UIP and non-UIP.
Patients with probable UIP, indeterminate UIP, or an alternative diagnosis should undergo bronchoalveolar lavage (BAL) and surgical lung biopsy (SLB). Transbronchial lung biopsy (TBBx) and lung cryobiopsy recommendations were not described in these patients because of a lack of evidence. However, patients with UIP should not undergo BAL, SLB, TBBx, or cryobiopsy.
For all patients, the 2018 guideline recommends that medical histories include environmental exposure and medication use, and that serological testing is performed to exclude connective tissue disease. Multidisciplinary discussion is conditionally recommended for diagnostic decision making, particularly when a patient has probable UIP, indeterminate UIP, or an alternative diagnosis.
Finally, the guideline recommends against the use of serum biomarkers as a means of distinguishing between IPF and other ILDs because of weak data on this point. “For the time being, the guideline panel dismissed serum biomarker measurement as an approach to distinguishing IPF from other ILDs because of the high false-positive and false-negative result rates,” the panel wrote.
“Our hope is that this new guideline will bridge the gap between the experienced IPF experts and general pulmonologists in making a prompt and accurate diagnosis of IPF for the individual unfortunately confronted with the disease,” Dr. Raghu said. “This will allow patients to make well-informed decisions about treatment options and participation in clinical trials.”
Looking to the future, the panel described an “urgent need to refine and validate diagnostic approaches to ILD. These needs can be roughly categorized as investigations into the roles of clinical observations, HRCT, bronchoscopy, histopathology, and biomarkers.” The authors also emphasized “the need to refine prognostic approaches, identify risk factors for the development of IPF, and determine the impact and approach to the diagnosis of comorbid illness in the patient with IPF.”
The authors reported funding from Bellerophon, Gilead, Roche, Sanofi, and others.
SOURCE: Raghu G et al. Am J Respir Crit Care Med. 2018 Sep 1. doi: 10.1164/rccm.201807-1255ST.
FROM THE AMERICAN JOURNAL OF RESPIRATORY AND CRITICAL CARE MEDICINE