User login
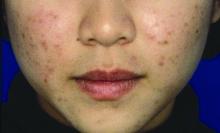
Ovarian reserve markers fall on isotretinoin, but rebound after stopping treatment
MILAN – according to data presented at the World Congress of Dermatology.
Although markers for ovarian reserve, including anti-Müllerian hormone (AMH) serum levels, ovarian volume, and antral follicle count, were significantly lower during a period of isotretinoin use than at baseline, these values were were not significantly different from pretreatment levels by 1 month after stopping isotretinoin.
For patients taking isotretinoin at a dose of 0.5 mg/kg/day, AMH levels fell from a baseline level of 5.29 ng/mL to 4.16 ng/mL during treatment, but rebounded to 4.77 ng/mL 1 month after stopping treatment (P less than .001 for difference between baseline and on-drug values), Tuğba Özkök Akbulut, MD, said during a late-breaking abstracts session.
For women taking isotretinoin 1 mg/kg/day, AMH levels went from 5.14 ng/mL at baseline to 4.24 ng/mL on treatment, to 4.65 ng/mL 1 month after treatment (P less than .001 for difference between baseline and on-drug values), reported Dr. Akbulut a dermatologist at the Haseki Training Research Hospital, Istanbul.
Women on the higher dose of isotretinoin had a similar pattern of decline while on treatment and rebound after ceasing isotretinoin for ovarian volume and antral follicle count (P less than .001 for all values). These differences were not statistically significant for women taking 0.5 mg/kg/day of isotretinoin, except for right ovarian volume (P = 0.013).
Although values were numerically lower for many markers of ovarian reserve after ceasing treatment, compared with baseline figures, these differences were not statistically significantly different. Markers of ovarian reserve did not change significantly for a control group of women without acne.
Dr. Akbulut and her colleagues conducted this prospective case-control study of 42 women of reproductive age who sought dermatologist care for severe acne unresponsive to conservative therapy; 26 women who did not have acne constituted the control group. Smokers, patients with thyroid disease, and those with known polycystic ovary syndrome were excluded from participation.
The women with acne received oral isotretinoin dosed either at 0.5 or 1.0 mg/kg/day, with treatment lasting 5-9 months. For each patient, treatment was stopped when the cumulative dose reached 120 mg/kg.
After an initial visit at which blood was collected from all participants to measure serum AMH levels, those receiving isotretinoin were seen every 4 weeks to check serum lipid and liver enzyme levels.
At the 3-month mark during the study period and 1 month after the end of completing isotretinoin treatment, or at the end of the study period for the control group, blood samples also were drawn for AMH levels.
To measure hormone levels, also blood was drawn between days 2 and 5 of the follicular phase of the menstrual cycle. Participants received ultrasounds to measure antral follicle count and ovarian volume between days 2 and 5 of the menstrual cycle at the initial visit, at the 3-month visit, and at the final visit. Results were interpreted by a trained gynecologist.
Patients, who were mostly in their early 20s, had a mean body mass index of about 22 kg/m2. Hormone levels, ovarian volume, and antral follicle count did not differ among study arms at baseline.
“There are contradictory reports in the literature regarding the effect of retinoic acid on ovarian reserve,” noted Dr. Akbulut. Some preclinical studies found that retinoic acid increased fertility and ovarian reserve in rodents; however, some human studies had shown lower serum AMH concentrations in patients using isotretinoin.
This new demonstration of the reversibility of isotretinoin’s negative effect on ovarian reserve helps clarify a confused picture in the medical literature, said Dr. Akbulut. “The results of our study demonstrated that systemic isotretinoin had a reversible effect on ovarian reserve.”
Dr. Akbulut reported no outside sources of funding and that she had no relevant financial disclosures.
MILAN – according to data presented at the World Congress of Dermatology.
Although markers for ovarian reserve, including anti-Müllerian hormone (AMH) serum levels, ovarian volume, and antral follicle count, were significantly lower during a period of isotretinoin use than at baseline, these values were were not significantly different from pretreatment levels by 1 month after stopping isotretinoin.
For patients taking isotretinoin at a dose of 0.5 mg/kg/day, AMH levels fell from a baseline level of 5.29 ng/mL to 4.16 ng/mL during treatment, but rebounded to 4.77 ng/mL 1 month after stopping treatment (P less than .001 for difference between baseline and on-drug values), Tuğba Özkök Akbulut, MD, said during a late-breaking abstracts session.
For women taking isotretinoin 1 mg/kg/day, AMH levels went from 5.14 ng/mL at baseline to 4.24 ng/mL on treatment, to 4.65 ng/mL 1 month after treatment (P less than .001 for difference between baseline and on-drug values), reported Dr. Akbulut a dermatologist at the Haseki Training Research Hospital, Istanbul.
Women on the higher dose of isotretinoin had a similar pattern of decline while on treatment and rebound after ceasing isotretinoin for ovarian volume and antral follicle count (P less than .001 for all values). These differences were not statistically significant for women taking 0.5 mg/kg/day of isotretinoin, except for right ovarian volume (P = 0.013).
Although values were numerically lower for many markers of ovarian reserve after ceasing treatment, compared with baseline figures, these differences were not statistically significantly different. Markers of ovarian reserve did not change significantly for a control group of women without acne.
Dr. Akbulut and her colleagues conducted this prospective case-control study of 42 women of reproductive age who sought dermatologist care for severe acne unresponsive to conservative therapy; 26 women who did not have acne constituted the control group. Smokers, patients with thyroid disease, and those with known polycystic ovary syndrome were excluded from participation.
The women with acne received oral isotretinoin dosed either at 0.5 or 1.0 mg/kg/day, with treatment lasting 5-9 months. For each patient, treatment was stopped when the cumulative dose reached 120 mg/kg.
After an initial visit at which blood was collected from all participants to measure serum AMH levels, those receiving isotretinoin were seen every 4 weeks to check serum lipid and liver enzyme levels.
At the 3-month mark during the study period and 1 month after the end of completing isotretinoin treatment, or at the end of the study period for the control group, blood samples also were drawn for AMH levels.
To measure hormone levels, also blood was drawn between days 2 and 5 of the follicular phase of the menstrual cycle. Participants received ultrasounds to measure antral follicle count and ovarian volume between days 2 and 5 of the menstrual cycle at the initial visit, at the 3-month visit, and at the final visit. Results were interpreted by a trained gynecologist.
Patients, who were mostly in their early 20s, had a mean body mass index of about 22 kg/m2. Hormone levels, ovarian volume, and antral follicle count did not differ among study arms at baseline.
“There are contradictory reports in the literature regarding the effect of retinoic acid on ovarian reserve,” noted Dr. Akbulut. Some preclinical studies found that retinoic acid increased fertility and ovarian reserve in rodents; however, some human studies had shown lower serum AMH concentrations in patients using isotretinoin.
This new demonstration of the reversibility of isotretinoin’s negative effect on ovarian reserve helps clarify a confused picture in the medical literature, said Dr. Akbulut. “The results of our study demonstrated that systemic isotretinoin had a reversible effect on ovarian reserve.”
Dr. Akbulut reported no outside sources of funding and that she had no relevant financial disclosures.
MILAN – according to data presented at the World Congress of Dermatology.
Although markers for ovarian reserve, including anti-Müllerian hormone (AMH) serum levels, ovarian volume, and antral follicle count, were significantly lower during a period of isotretinoin use than at baseline, these values were were not significantly different from pretreatment levels by 1 month after stopping isotretinoin.
For patients taking isotretinoin at a dose of 0.5 mg/kg/day, AMH levels fell from a baseline level of 5.29 ng/mL to 4.16 ng/mL during treatment, but rebounded to 4.77 ng/mL 1 month after stopping treatment (P less than .001 for difference between baseline and on-drug values), Tuğba Özkök Akbulut, MD, said during a late-breaking abstracts session.
For women taking isotretinoin 1 mg/kg/day, AMH levels went from 5.14 ng/mL at baseline to 4.24 ng/mL on treatment, to 4.65 ng/mL 1 month after treatment (P less than .001 for difference between baseline and on-drug values), reported Dr. Akbulut a dermatologist at the Haseki Training Research Hospital, Istanbul.
Women on the higher dose of isotretinoin had a similar pattern of decline while on treatment and rebound after ceasing isotretinoin for ovarian volume and antral follicle count (P less than .001 for all values). These differences were not statistically significant for women taking 0.5 mg/kg/day of isotretinoin, except for right ovarian volume (P = 0.013).
Although values were numerically lower for many markers of ovarian reserve after ceasing treatment, compared with baseline figures, these differences were not statistically significantly different. Markers of ovarian reserve did not change significantly for a control group of women without acne.
Dr. Akbulut and her colleagues conducted this prospective case-control study of 42 women of reproductive age who sought dermatologist care for severe acne unresponsive to conservative therapy; 26 women who did not have acne constituted the control group. Smokers, patients with thyroid disease, and those with known polycystic ovary syndrome were excluded from participation.
The women with acne received oral isotretinoin dosed either at 0.5 or 1.0 mg/kg/day, with treatment lasting 5-9 months. For each patient, treatment was stopped when the cumulative dose reached 120 mg/kg.
After an initial visit at which blood was collected from all participants to measure serum AMH levels, those receiving isotretinoin were seen every 4 weeks to check serum lipid and liver enzyme levels.
At the 3-month mark during the study period and 1 month after the end of completing isotretinoin treatment, or at the end of the study period for the control group, blood samples also were drawn for AMH levels.
To measure hormone levels, also blood was drawn between days 2 and 5 of the follicular phase of the menstrual cycle. Participants received ultrasounds to measure antral follicle count and ovarian volume between days 2 and 5 of the menstrual cycle at the initial visit, at the 3-month visit, and at the final visit. Results were interpreted by a trained gynecologist.
Patients, who were mostly in their early 20s, had a mean body mass index of about 22 kg/m2. Hormone levels, ovarian volume, and antral follicle count did not differ among study arms at baseline.
“There are contradictory reports in the literature regarding the effect of retinoic acid on ovarian reserve,” noted Dr. Akbulut. Some preclinical studies found that retinoic acid increased fertility and ovarian reserve in rodents; however, some human studies had shown lower serum AMH concentrations in patients using isotretinoin.
This new demonstration of the reversibility of isotretinoin’s negative effect on ovarian reserve helps clarify a confused picture in the medical literature, said Dr. Akbulut. “The results of our study demonstrated that systemic isotretinoin had a reversible effect on ovarian reserve.”
Dr. Akbulut reported no outside sources of funding and that she had no relevant financial disclosures.
REPORTING FROM WCD2019
Ixekizumab surpasses adalimumab in PsA head-to-head study
MADRID – The interleukin-17A inhibitor ixekizumab surpassed the tumor necrosis factor inhibitor adalimumab for treatment of patients with psoriatic arthritis in a multicenter, randomized study with 566 enrolled patients, the first reported results from a head-to-head comparison for this disease of two different classes of biological drugs.
The results showed that a standard, 24-week regimen with each of these agents, both of which already have regulatory approval for treating psoriatic arthritis (PsA), led to achievement of the primary endpoint in 36% of patients treated with ixekizumab (Taltz) and 28% of patients treated with adalimumab (Humira), a statistically significant difference, Philip J. Mease, MD, said at the European Congress of Rheumatology.
“Ixekizumab was superior to adalimumab for improving signs and symptoms of active PsA, as measured by simultaneous achievement of ACR50 [American College of Rheumatology] and PASI 100 [Psoriasis Area and Severity Index],” the study’s primary endpoint that combined a measure of joint disease activity with a measure of skin involvement, said Dr. Mease, a rheumatologist at Swedish Medical Center in Seattle.
This unconventional primary endpoint for testing drugs that treat PsA was called out during discussion of the report for having an inherent bias favoring ixekizumab by its inclusion of a skin outcome that received equal weight with an assessment tool that focused on joint responses. “This was a very unusual primary endpoint that favored ixekizumab,” Roy M. Fleischmann, MD, a Dallas rheumatologist, commented during the discussion.
Dr. Mease readily admitted that the study’s design stacked the deck in favor of ixekizumab, but he added that this decision reflected a desire by the researchers who ran the study to choose a primary endpoint that represented both of the prominent pathologies seen in patients with PsA.
The primary endpoint used in the study “looks at PsA more holistically,” Dr. Mease said in an interview. “It forced clinicians to look beyond just the joints,” in PsA patients. “That has been a limitation of prior PsA treatment assessments,” which until this study have uniformly used single primary outcomes that focus on joint responses, most commonly the ACR20 measure of joint disease activity.
The SPIRIT-H2H (A Study of Ixekizumab [LY2439821] Versus Adalimumab in Participants With Psoriatic Arthritis) study enrolled adults with active PsA who had never before received treatment with a biological drug. Enrolled patients had to have both active disease in their joints and active plaque psoriasis, with an inadequate response to at least one conventional synthetic disease-modifying antirheumatic drug. The 566 patients randomized in the study averaged about 48 years old, a bit more than half were men, and patients averaged about 6 years with diagnosed PsA and about 15 years diagnosed with psoriasis. Just over two-thirds of the patients were on concurrent treatment with a conventional synthetic agent, most often methotrexate.
The two components of the primary endpoint each showed the anticipated result. Among the 269 patients treated with adalimumab for the full 24 weeks, 47% had an ACR50 response, as did 51% of the 262 patients who completed their full course of ixekizumab, a between-group difference that was not statistically significant. In contrast, the PASI 100 measure of complete skin resolution occurred in 47% of the adalimumab-treated patients and 60% of those on ixekizumab, a statistically significant difference.
Dr. Mease reported results for several other efficacy measures, and what was notable was statistically significant superiority for ixekizumab in a measure of entheses disease activity, the SPARCC [Spondyloarthritis Research Consortium of Canada] Enthesitis Index, which fell to zero in 57% of the ixekizumab patients and 45% of those on adalimumab. “It makes you wonder whether there is something special about interleukin-17 in enthesitis,” Dr. Mease said.
The safety results of the study were consistent with the known adverse effect profiles of both drugs.
The impact of these findings on practice remains to be seen, and will likely depend on both cost considerations as well as clinicians trying to tailor drug choices to individual patients. The relatively new drug, ixekizumab, is consequentially more expensive than the older adalimumab, which has had its price depressed by the recent introduction of a biosimilar agent as well as long-standing competition from multiple TNF inhibitors.
“I think in the United States insurers will continue to steer patients toward whichever drug is cheapest among the highly-effective options,” but this result should lead to more use of interleukin-17 inhibitors as second-line agents for PsA, and it might push some clinicians to prescribe it as the first-line treatment, Dr. Mease said. He was confident that the efficacy profile shown by ixekizumab in SPIRIT-H2H was likely a class effect. Economics aside, the impetus to prescribe ixekizumab or another interleukin-17 inhibitor will be greatest when a patient has more extensive skin involvement, while for patients with little or no skin symptoms clinicians will likely stick with the more established TNF inhibitors as the first drug class to prescribe for PsA.
SPIRIT-H2H was sponsored by Eli Lilly, the company that markets ixekizumab (Taltz). Dr. Mease has been a consultant to, speaker for, and received research funding from Eli Lilly and from several other companies. Dr. Fleischmann has been a consultant to and has received research funding from several companies including Eli Lilly.
SOURCE: Mease PJ et al. Ann Rheum Dis. 2019 Jun. doi: 10.1136/annrheumdis-2019-eular.8709.
The results from SPIRIT-H2H confirm with rigorously-collected, prospective data what we had already seen during our routine use of ixekizumab for treating patients with psoriatic arthritis: it does a better job of resolving skin manifestations than any tumor necrosis factor (TNF) inhibitor. For joint symptoms, the two drugs are similar, but for skin ixekizumab has substantial superiority.
I had been hopeful that inhibition of interleukin-17 would surpass TNF inhibition for resolution of joint symptoms, but it looks like they are similar. That’s a little below expectations. But working well for improving skin symptoms is important because it’s something that many patients care about, especially those with more substantial skin symptoms. When matching the best drug to each PsA patient other considerations also exist, such as ease of use. These drugs are delivered by different devices, and ease of administration also matters to patients.
I think it would be premature to presume that the effects shown by ixekizumab extrapolate to all the other interleukin-17 inhibitors. Some of these drugs act via different mechanisms, and so the SPIRIT-H2H results may very well not reflect a class effect.
Thomas Dörner, MD, is professor of rheumatology at Charité University Hospital in Berlin. He has been a consultant to Eli Lilly, as well as to AbbVie, Celgene, Novartis, Pfizer, and Roche, he has been a speaker on behalf of Amgen, Biogen, and Celgene, and he has received research funding from Chugai, Janssen, Roche, and Sanofi. He made these comments in an interview.
The results from SPIRIT-H2H confirm with rigorously-collected, prospective data what we had already seen during our routine use of ixekizumab for treating patients with psoriatic arthritis: it does a better job of resolving skin manifestations than any tumor necrosis factor (TNF) inhibitor. For joint symptoms, the two drugs are similar, but for skin ixekizumab has substantial superiority.
I had been hopeful that inhibition of interleukin-17 would surpass TNF inhibition for resolution of joint symptoms, but it looks like they are similar. That’s a little below expectations. But working well for improving skin symptoms is important because it’s something that many patients care about, especially those with more substantial skin symptoms. When matching the best drug to each PsA patient other considerations also exist, such as ease of use. These drugs are delivered by different devices, and ease of administration also matters to patients.
I think it would be premature to presume that the effects shown by ixekizumab extrapolate to all the other interleukin-17 inhibitors. Some of these drugs act via different mechanisms, and so the SPIRIT-H2H results may very well not reflect a class effect.
Thomas Dörner, MD, is professor of rheumatology at Charité University Hospital in Berlin. He has been a consultant to Eli Lilly, as well as to AbbVie, Celgene, Novartis, Pfizer, and Roche, he has been a speaker on behalf of Amgen, Biogen, and Celgene, and he has received research funding from Chugai, Janssen, Roche, and Sanofi. He made these comments in an interview.
The results from SPIRIT-H2H confirm with rigorously-collected, prospective data what we had already seen during our routine use of ixekizumab for treating patients with psoriatic arthritis: it does a better job of resolving skin manifestations than any tumor necrosis factor (TNF) inhibitor. For joint symptoms, the two drugs are similar, but for skin ixekizumab has substantial superiority.
I had been hopeful that inhibition of interleukin-17 would surpass TNF inhibition for resolution of joint symptoms, but it looks like they are similar. That’s a little below expectations. But working well for improving skin symptoms is important because it’s something that many patients care about, especially those with more substantial skin symptoms. When matching the best drug to each PsA patient other considerations also exist, such as ease of use. These drugs are delivered by different devices, and ease of administration also matters to patients.
I think it would be premature to presume that the effects shown by ixekizumab extrapolate to all the other interleukin-17 inhibitors. Some of these drugs act via different mechanisms, and so the SPIRIT-H2H results may very well not reflect a class effect.
Thomas Dörner, MD, is professor of rheumatology at Charité University Hospital in Berlin. He has been a consultant to Eli Lilly, as well as to AbbVie, Celgene, Novartis, Pfizer, and Roche, he has been a speaker on behalf of Amgen, Biogen, and Celgene, and he has received research funding from Chugai, Janssen, Roche, and Sanofi. He made these comments in an interview.
MADRID – The interleukin-17A inhibitor ixekizumab surpassed the tumor necrosis factor inhibitor adalimumab for treatment of patients with psoriatic arthritis in a multicenter, randomized study with 566 enrolled patients, the first reported results from a head-to-head comparison for this disease of two different classes of biological drugs.
The results showed that a standard, 24-week regimen with each of these agents, both of which already have regulatory approval for treating psoriatic arthritis (PsA), led to achievement of the primary endpoint in 36% of patients treated with ixekizumab (Taltz) and 28% of patients treated with adalimumab (Humira), a statistically significant difference, Philip J. Mease, MD, said at the European Congress of Rheumatology.
“Ixekizumab was superior to adalimumab for improving signs and symptoms of active PsA, as measured by simultaneous achievement of ACR50 [American College of Rheumatology] and PASI 100 [Psoriasis Area and Severity Index],” the study’s primary endpoint that combined a measure of joint disease activity with a measure of skin involvement, said Dr. Mease, a rheumatologist at Swedish Medical Center in Seattle.
This unconventional primary endpoint for testing drugs that treat PsA was called out during discussion of the report for having an inherent bias favoring ixekizumab by its inclusion of a skin outcome that received equal weight with an assessment tool that focused on joint responses. “This was a very unusual primary endpoint that favored ixekizumab,” Roy M. Fleischmann, MD, a Dallas rheumatologist, commented during the discussion.
Dr. Mease readily admitted that the study’s design stacked the deck in favor of ixekizumab, but he added that this decision reflected a desire by the researchers who ran the study to choose a primary endpoint that represented both of the prominent pathologies seen in patients with PsA.
The primary endpoint used in the study “looks at PsA more holistically,” Dr. Mease said in an interview. “It forced clinicians to look beyond just the joints,” in PsA patients. “That has been a limitation of prior PsA treatment assessments,” which until this study have uniformly used single primary outcomes that focus on joint responses, most commonly the ACR20 measure of joint disease activity.
The SPIRIT-H2H (A Study of Ixekizumab [LY2439821] Versus Adalimumab in Participants With Psoriatic Arthritis) study enrolled adults with active PsA who had never before received treatment with a biological drug. Enrolled patients had to have both active disease in their joints and active plaque psoriasis, with an inadequate response to at least one conventional synthetic disease-modifying antirheumatic drug. The 566 patients randomized in the study averaged about 48 years old, a bit more than half were men, and patients averaged about 6 years with diagnosed PsA and about 15 years diagnosed with psoriasis. Just over two-thirds of the patients were on concurrent treatment with a conventional synthetic agent, most often methotrexate.
The two components of the primary endpoint each showed the anticipated result. Among the 269 patients treated with adalimumab for the full 24 weeks, 47% had an ACR50 response, as did 51% of the 262 patients who completed their full course of ixekizumab, a between-group difference that was not statistically significant. In contrast, the PASI 100 measure of complete skin resolution occurred in 47% of the adalimumab-treated patients and 60% of those on ixekizumab, a statistically significant difference.
Dr. Mease reported results for several other efficacy measures, and what was notable was statistically significant superiority for ixekizumab in a measure of entheses disease activity, the SPARCC [Spondyloarthritis Research Consortium of Canada] Enthesitis Index, which fell to zero in 57% of the ixekizumab patients and 45% of those on adalimumab. “It makes you wonder whether there is something special about interleukin-17 in enthesitis,” Dr. Mease said.
The safety results of the study were consistent with the known adverse effect profiles of both drugs.
The impact of these findings on practice remains to be seen, and will likely depend on both cost considerations as well as clinicians trying to tailor drug choices to individual patients. The relatively new drug, ixekizumab, is consequentially more expensive than the older adalimumab, which has had its price depressed by the recent introduction of a biosimilar agent as well as long-standing competition from multiple TNF inhibitors.
“I think in the United States insurers will continue to steer patients toward whichever drug is cheapest among the highly-effective options,” but this result should lead to more use of interleukin-17 inhibitors as second-line agents for PsA, and it might push some clinicians to prescribe it as the first-line treatment, Dr. Mease said. He was confident that the efficacy profile shown by ixekizumab in SPIRIT-H2H was likely a class effect. Economics aside, the impetus to prescribe ixekizumab or another interleukin-17 inhibitor will be greatest when a patient has more extensive skin involvement, while for patients with little or no skin symptoms clinicians will likely stick with the more established TNF inhibitors as the first drug class to prescribe for PsA.
SPIRIT-H2H was sponsored by Eli Lilly, the company that markets ixekizumab (Taltz). Dr. Mease has been a consultant to, speaker for, and received research funding from Eli Lilly and from several other companies. Dr. Fleischmann has been a consultant to and has received research funding from several companies including Eli Lilly.
SOURCE: Mease PJ et al. Ann Rheum Dis. 2019 Jun. doi: 10.1136/annrheumdis-2019-eular.8709.
MADRID – The interleukin-17A inhibitor ixekizumab surpassed the tumor necrosis factor inhibitor adalimumab for treatment of patients with psoriatic arthritis in a multicenter, randomized study with 566 enrolled patients, the first reported results from a head-to-head comparison for this disease of two different classes of biological drugs.
The results showed that a standard, 24-week regimen with each of these agents, both of which already have regulatory approval for treating psoriatic arthritis (PsA), led to achievement of the primary endpoint in 36% of patients treated with ixekizumab (Taltz) and 28% of patients treated with adalimumab (Humira), a statistically significant difference, Philip J. Mease, MD, said at the European Congress of Rheumatology.
“Ixekizumab was superior to adalimumab for improving signs and symptoms of active PsA, as measured by simultaneous achievement of ACR50 [American College of Rheumatology] and PASI 100 [Psoriasis Area and Severity Index],” the study’s primary endpoint that combined a measure of joint disease activity with a measure of skin involvement, said Dr. Mease, a rheumatologist at Swedish Medical Center in Seattle.
This unconventional primary endpoint for testing drugs that treat PsA was called out during discussion of the report for having an inherent bias favoring ixekizumab by its inclusion of a skin outcome that received equal weight with an assessment tool that focused on joint responses. “This was a very unusual primary endpoint that favored ixekizumab,” Roy M. Fleischmann, MD, a Dallas rheumatologist, commented during the discussion.
Dr. Mease readily admitted that the study’s design stacked the deck in favor of ixekizumab, but he added that this decision reflected a desire by the researchers who ran the study to choose a primary endpoint that represented both of the prominent pathologies seen in patients with PsA.
The primary endpoint used in the study “looks at PsA more holistically,” Dr. Mease said in an interview. “It forced clinicians to look beyond just the joints,” in PsA patients. “That has been a limitation of prior PsA treatment assessments,” which until this study have uniformly used single primary outcomes that focus on joint responses, most commonly the ACR20 measure of joint disease activity.
The SPIRIT-H2H (A Study of Ixekizumab [LY2439821] Versus Adalimumab in Participants With Psoriatic Arthritis) study enrolled adults with active PsA who had never before received treatment with a biological drug. Enrolled patients had to have both active disease in their joints and active plaque psoriasis, with an inadequate response to at least one conventional synthetic disease-modifying antirheumatic drug. The 566 patients randomized in the study averaged about 48 years old, a bit more than half were men, and patients averaged about 6 years with diagnosed PsA and about 15 years diagnosed with psoriasis. Just over two-thirds of the patients were on concurrent treatment with a conventional synthetic agent, most often methotrexate.
The two components of the primary endpoint each showed the anticipated result. Among the 269 patients treated with adalimumab for the full 24 weeks, 47% had an ACR50 response, as did 51% of the 262 patients who completed their full course of ixekizumab, a between-group difference that was not statistically significant. In contrast, the PASI 100 measure of complete skin resolution occurred in 47% of the adalimumab-treated patients and 60% of those on ixekizumab, a statistically significant difference.
Dr. Mease reported results for several other efficacy measures, and what was notable was statistically significant superiority for ixekizumab in a measure of entheses disease activity, the SPARCC [Spondyloarthritis Research Consortium of Canada] Enthesitis Index, which fell to zero in 57% of the ixekizumab patients and 45% of those on adalimumab. “It makes you wonder whether there is something special about interleukin-17 in enthesitis,” Dr. Mease said.
The safety results of the study were consistent with the known adverse effect profiles of both drugs.
The impact of these findings on practice remains to be seen, and will likely depend on both cost considerations as well as clinicians trying to tailor drug choices to individual patients. The relatively new drug, ixekizumab, is consequentially more expensive than the older adalimumab, which has had its price depressed by the recent introduction of a biosimilar agent as well as long-standing competition from multiple TNF inhibitors.
“I think in the United States insurers will continue to steer patients toward whichever drug is cheapest among the highly-effective options,” but this result should lead to more use of interleukin-17 inhibitors as second-line agents for PsA, and it might push some clinicians to prescribe it as the first-line treatment, Dr. Mease said. He was confident that the efficacy profile shown by ixekizumab in SPIRIT-H2H was likely a class effect. Economics aside, the impetus to prescribe ixekizumab or another interleukin-17 inhibitor will be greatest when a patient has more extensive skin involvement, while for patients with little or no skin symptoms clinicians will likely stick with the more established TNF inhibitors as the first drug class to prescribe for PsA.
SPIRIT-H2H was sponsored by Eli Lilly, the company that markets ixekizumab (Taltz). Dr. Mease has been a consultant to, speaker for, and received research funding from Eli Lilly and from several other companies. Dr. Fleischmann has been a consultant to and has received research funding from several companies including Eli Lilly.
SOURCE: Mease PJ et al. Ann Rheum Dis. 2019 Jun. doi: 10.1136/annrheumdis-2019-eular.8709.
REPORTING FROM THE EULAR 2019 CONGRESS
Booster vaccines found largely safe in children on immunosuppressive drugs
MADRID – Administration of live attenuated booster of the MMR vaccine with or without varicella (MMR/V) was not associated with serious adverse events in children on immunosuppressive therapy for a rheumatic disease, according to data presented at the European Congress of Rheumatology.
“The study implies that patients can receive booster vaccinations regardless of age, diagnosis, or therapy,” reported Veronica Bergonzo Moshe, MD, a pediatric rheumatologist at Meir Medical Center, Kfar Saba, Israel.
In the absence of safety data, the vaccination of children with rheumatic diseases taking immunosuppressive therapies has been controversial. Although these children face communicable and sometimes life-threatening diseases without vaccination, many clinicians are not offering this protection because they fear adverse consequences.
Current Paediatric Rheumatology European Society (PReS) guidelines have been equivocal, recommending that vaccines be considered on a “case-by-case basis” in children with a rheumatic disease if they are taking high doses of disease-modifying antirheumatic drugs (DMARDs), glucocorticoids, or any dose of biologics.
“The fear is that a state of immune suppression might decrease response to the vaccine or lead to a flare of the rheumatologic disease,” Dr. Moshe said.
In the retrospective study presented by Dr. Moshe, data were collected on 234 children with rheumatic diseases who received a live attenuated MMR/V booster. The children were drawn from 12 pediatric rheumatology centers in 10 countries.
In this relatively large series, 82% of the children had oligoarticular or polyarticular juvenile idiopathic arthritis (JIA). A range of other rheumatic diseases, including juvenile dermatomyositis, localized scleroderma, and isolated idiopathic uveitis were represented among the remaining patients. All were taking medication, and 48% were in remission.
When broken down by therapy, there were three localized reactions in 110 (2.7%) children who received the booster while on methotrexate. No other adverse events, including disease flare, were observed.
Similarly, six of the seven adverse events observed in 76 (8%) patients who were taking methotrexate plus a tumor necrosis factor (TNF) inhibitor biologic at the time of vaccination were local reactions. Fever was reported in one patient. All of these events were transient.
In the 39 patients taking a TNF inhibitor alone, there was a single case of transient fever. There were no adverse events reported in the three patients vaccinated while on tocilizumab, seven patients while on anakinra, or five patients while on canakinumab.
Following vaccination, there were no signs of symptoms of the diseases that the vaccines are designed to prevent. In the minority of patients who did develop localized reactions or fever in this series, there was no apparent relationship with disease activity, age, or sex when compared to those who did not develop an adverse event.
These retrospective data are not definitive, but they are reassuring, according to Dr. Moshe. A larger prospective study by the PReS vaccination study group is now planned. The issue of leaving children unvaccinated is topical due to the recent outbreaks of measles in the United States.
“We must have clear guidelines on how to deal with the administration of live vaccines in this patient population so that we can provide the safest and most effective practice,” Dr. Moshe said.
These data are a first step.
“This large retrospective study demonstrates that live attenuated booster vaccine is probably safe in children with rheumatic diseases,” said Dr. Moshe, but she deferred to the PReS guidelines in suggesting that the decision to vaccinate still might best be performed on a case-by-case basis.
MADRID – Administration of live attenuated booster of the MMR vaccine with or without varicella (MMR/V) was not associated with serious adverse events in children on immunosuppressive therapy for a rheumatic disease, according to data presented at the European Congress of Rheumatology.
“The study implies that patients can receive booster vaccinations regardless of age, diagnosis, or therapy,” reported Veronica Bergonzo Moshe, MD, a pediatric rheumatologist at Meir Medical Center, Kfar Saba, Israel.
In the absence of safety data, the vaccination of children with rheumatic diseases taking immunosuppressive therapies has been controversial. Although these children face communicable and sometimes life-threatening diseases without vaccination, many clinicians are not offering this protection because they fear adverse consequences.
Current Paediatric Rheumatology European Society (PReS) guidelines have been equivocal, recommending that vaccines be considered on a “case-by-case basis” in children with a rheumatic disease if they are taking high doses of disease-modifying antirheumatic drugs (DMARDs), glucocorticoids, or any dose of biologics.
“The fear is that a state of immune suppression might decrease response to the vaccine or lead to a flare of the rheumatologic disease,” Dr. Moshe said.
In the retrospective study presented by Dr. Moshe, data were collected on 234 children with rheumatic diseases who received a live attenuated MMR/V booster. The children were drawn from 12 pediatric rheumatology centers in 10 countries.
In this relatively large series, 82% of the children had oligoarticular or polyarticular juvenile idiopathic arthritis (JIA). A range of other rheumatic diseases, including juvenile dermatomyositis, localized scleroderma, and isolated idiopathic uveitis were represented among the remaining patients. All were taking medication, and 48% were in remission.
When broken down by therapy, there were three localized reactions in 110 (2.7%) children who received the booster while on methotrexate. No other adverse events, including disease flare, were observed.
Similarly, six of the seven adverse events observed in 76 (8%) patients who were taking methotrexate plus a tumor necrosis factor (TNF) inhibitor biologic at the time of vaccination were local reactions. Fever was reported in one patient. All of these events were transient.
In the 39 patients taking a TNF inhibitor alone, there was a single case of transient fever. There were no adverse events reported in the three patients vaccinated while on tocilizumab, seven patients while on anakinra, or five patients while on canakinumab.
Following vaccination, there were no signs of symptoms of the diseases that the vaccines are designed to prevent. In the minority of patients who did develop localized reactions or fever in this series, there was no apparent relationship with disease activity, age, or sex when compared to those who did not develop an adverse event.
These retrospective data are not definitive, but they are reassuring, according to Dr. Moshe. A larger prospective study by the PReS vaccination study group is now planned. The issue of leaving children unvaccinated is topical due to the recent outbreaks of measles in the United States.
“We must have clear guidelines on how to deal with the administration of live vaccines in this patient population so that we can provide the safest and most effective practice,” Dr. Moshe said.
These data are a first step.
“This large retrospective study demonstrates that live attenuated booster vaccine is probably safe in children with rheumatic diseases,” said Dr. Moshe, but she deferred to the PReS guidelines in suggesting that the decision to vaccinate still might best be performed on a case-by-case basis.
MADRID – Administration of live attenuated booster of the MMR vaccine with or without varicella (MMR/V) was not associated with serious adverse events in children on immunosuppressive therapy for a rheumatic disease, according to data presented at the European Congress of Rheumatology.
“The study implies that patients can receive booster vaccinations regardless of age, diagnosis, or therapy,” reported Veronica Bergonzo Moshe, MD, a pediatric rheumatologist at Meir Medical Center, Kfar Saba, Israel.
In the absence of safety data, the vaccination of children with rheumatic diseases taking immunosuppressive therapies has been controversial. Although these children face communicable and sometimes life-threatening diseases without vaccination, many clinicians are not offering this protection because they fear adverse consequences.
Current Paediatric Rheumatology European Society (PReS) guidelines have been equivocal, recommending that vaccines be considered on a “case-by-case basis” in children with a rheumatic disease if they are taking high doses of disease-modifying antirheumatic drugs (DMARDs), glucocorticoids, or any dose of biologics.
“The fear is that a state of immune suppression might decrease response to the vaccine or lead to a flare of the rheumatologic disease,” Dr. Moshe said.
In the retrospective study presented by Dr. Moshe, data were collected on 234 children with rheumatic diseases who received a live attenuated MMR/V booster. The children were drawn from 12 pediatric rheumatology centers in 10 countries.
In this relatively large series, 82% of the children had oligoarticular or polyarticular juvenile idiopathic arthritis (JIA). A range of other rheumatic diseases, including juvenile dermatomyositis, localized scleroderma, and isolated idiopathic uveitis were represented among the remaining patients. All were taking medication, and 48% were in remission.
When broken down by therapy, there were three localized reactions in 110 (2.7%) children who received the booster while on methotrexate. No other adverse events, including disease flare, were observed.
Similarly, six of the seven adverse events observed in 76 (8%) patients who were taking methotrexate plus a tumor necrosis factor (TNF) inhibitor biologic at the time of vaccination were local reactions. Fever was reported in one patient. All of these events were transient.
In the 39 patients taking a TNF inhibitor alone, there was a single case of transient fever. There were no adverse events reported in the three patients vaccinated while on tocilizumab, seven patients while on anakinra, or five patients while on canakinumab.
Following vaccination, there were no signs of symptoms of the diseases that the vaccines are designed to prevent. In the minority of patients who did develop localized reactions or fever in this series, there was no apparent relationship with disease activity, age, or sex when compared to those who did not develop an adverse event.
These retrospective data are not definitive, but they are reassuring, according to Dr. Moshe. A larger prospective study by the PReS vaccination study group is now planned. The issue of leaving children unvaccinated is topical due to the recent outbreaks of measles in the United States.
“We must have clear guidelines on how to deal with the administration of live vaccines in this patient population so that we can provide the safest and most effective practice,” Dr. Moshe said.
These data are a first step.
“This large retrospective study demonstrates that live attenuated booster vaccine is probably safe in children with rheumatic diseases,” said Dr. Moshe, but she deferred to the PReS guidelines in suggesting that the decision to vaccinate still might best be performed on a case-by-case basis.
REPORTING FROM EULAR 2019 CONGRESS
Rosacea: Quality of life improves with treatment success
according to a survey from the National Rosacea Society.
The survey was conducted among 1,044 patients with rosacea, and 76% saw at least some improvement in their skin following treatment. Of all patients who responded to treatment, 40% reported improvement in psychological well-being, 35% reported improvement in social well-being, and 31% reported improvement in occupational well-being.
The percentages of patients reporting improvements were higher or lower depending on the degree of skin clearance. Among patients whose signs and symptoms of rosacea were virtually eliminated, 81% said psychological well-being improved, 71% said social well-being did so, and 62% said occupational well-being improved. Among those whose rosacea improved only slightly or moderately, the percentages of well-being improvement were only 24%, 21%, and 19%, respectively.
The National Rosacea Society serves as a resource to provide information on the disorder, as well as increase awareness and support research.
according to a survey from the National Rosacea Society.
The survey was conducted among 1,044 patients with rosacea, and 76% saw at least some improvement in their skin following treatment. Of all patients who responded to treatment, 40% reported improvement in psychological well-being, 35% reported improvement in social well-being, and 31% reported improvement in occupational well-being.
The percentages of patients reporting improvements were higher or lower depending on the degree of skin clearance. Among patients whose signs and symptoms of rosacea were virtually eliminated, 81% said psychological well-being improved, 71% said social well-being did so, and 62% said occupational well-being improved. Among those whose rosacea improved only slightly or moderately, the percentages of well-being improvement were only 24%, 21%, and 19%, respectively.
The National Rosacea Society serves as a resource to provide information on the disorder, as well as increase awareness and support research.
according to a survey from the National Rosacea Society.
The survey was conducted among 1,044 patients with rosacea, and 76% saw at least some improvement in their skin following treatment. Of all patients who responded to treatment, 40% reported improvement in psychological well-being, 35% reported improvement in social well-being, and 31% reported improvement in occupational well-being.
The percentages of patients reporting improvements were higher or lower depending on the degree of skin clearance. Among patients whose signs and symptoms of rosacea were virtually eliminated, 81% said psychological well-being improved, 71% said social well-being did so, and 62% said occupational well-being improved. Among those whose rosacea improved only slightly or moderately, the percentages of well-being improvement were only 24%, 21%, and 19%, respectively.
The National Rosacea Society serves as a resource to provide information on the disorder, as well as increase awareness and support research.
Ultra low-dose rituximab retains promise in rheumatoid arthritis
MADRID – Retreatment of rheumatoid arthritis (RA) with an ultra low-dose of rituximab failed to meet the predefined noninferiority endpoint relative to a higher dose in a double-blind randomized trial, but the investigators still think this strategy may be viable in selected patients.
Lise M. Verhoef, MSc, a researcher in rheumatology at the Sint Maartenskliniek in Nijmegen, the Netherlands, who presented the data as a late-breaker at the European Congress of Rheumatology, explains in a video interview why the negative trial still might support an ultra low-dose strategy.
This trial, called REDO, was conceived after it was observed that most patients with RA are well controlled on a single injection of 1,000 mg of rituximab even though this is half the standard dose of two 1,000 mg doses given 15 days apart. The study was designed to determine whether even lower doses could be used.
The study enrolled 142 patients with RA who were being retreated with rituximab after responding previously to this therapy. In a 1:2:2 ratio, patients were randomized to single rituximab injections of 1,000 mg, 500 mg, or 200 mg. Outcome then were compared at the end of 6 months.
Noninferiority was defined as 0.5 difference in DAS28-CRP score (disease activity score using C-reactive protein instead of erythrocyte sedimentation rate) score adjusted for baseline disease status and use of conventional disease-modifying antirheumatic drugs (DMARDs).
Although noninferior at 3 months, the 500 mg dose did not meet the noninferiority criteria at 6 months. Due to a hierarchical design, evaluation of the 200 mg dose was precluded by the negative result with the 500 mg dose.
However, the majority of patients did respond to both the 500 mg and 200 mg dose. The failure to meet noninferiority was due to a limited number of patients who required rescue therapy for a flare. As a result, the investigators believe a trial of ultra low-dose therapy still might be reasonable.
In this interview, Ms. Verhoef explains that at her center patients who are well controlled on a 1,000 mg dose of rituximab now are being offered a 500 mg dose for retreatment. If they continue to respond, further retreatment with a 200 mg dose is considered.
Ms. Verhoef had no relevant financial disclosures.
MADRID – Retreatment of rheumatoid arthritis (RA) with an ultra low-dose of rituximab failed to meet the predefined noninferiority endpoint relative to a higher dose in a double-blind randomized trial, but the investigators still think this strategy may be viable in selected patients.
Lise M. Verhoef, MSc, a researcher in rheumatology at the Sint Maartenskliniek in Nijmegen, the Netherlands, who presented the data as a late-breaker at the European Congress of Rheumatology, explains in a video interview why the negative trial still might support an ultra low-dose strategy.
This trial, called REDO, was conceived after it was observed that most patients with RA are well controlled on a single injection of 1,000 mg of rituximab even though this is half the standard dose of two 1,000 mg doses given 15 days apart. The study was designed to determine whether even lower doses could be used.
The study enrolled 142 patients with RA who were being retreated with rituximab after responding previously to this therapy. In a 1:2:2 ratio, patients were randomized to single rituximab injections of 1,000 mg, 500 mg, or 200 mg. Outcome then were compared at the end of 6 months.
Noninferiority was defined as 0.5 difference in DAS28-CRP score (disease activity score using C-reactive protein instead of erythrocyte sedimentation rate) score adjusted for baseline disease status and use of conventional disease-modifying antirheumatic drugs (DMARDs).
Although noninferior at 3 months, the 500 mg dose did not meet the noninferiority criteria at 6 months. Due to a hierarchical design, evaluation of the 200 mg dose was precluded by the negative result with the 500 mg dose.
However, the majority of patients did respond to both the 500 mg and 200 mg dose. The failure to meet noninferiority was due to a limited number of patients who required rescue therapy for a flare. As a result, the investigators believe a trial of ultra low-dose therapy still might be reasonable.
In this interview, Ms. Verhoef explains that at her center patients who are well controlled on a 1,000 mg dose of rituximab now are being offered a 500 mg dose for retreatment. If they continue to respond, further retreatment with a 200 mg dose is considered.
Ms. Verhoef had no relevant financial disclosures.
MADRID – Retreatment of rheumatoid arthritis (RA) with an ultra low-dose of rituximab failed to meet the predefined noninferiority endpoint relative to a higher dose in a double-blind randomized trial, but the investigators still think this strategy may be viable in selected patients.
Lise M. Verhoef, MSc, a researcher in rheumatology at the Sint Maartenskliniek in Nijmegen, the Netherlands, who presented the data as a late-breaker at the European Congress of Rheumatology, explains in a video interview why the negative trial still might support an ultra low-dose strategy.
This trial, called REDO, was conceived after it was observed that most patients with RA are well controlled on a single injection of 1,000 mg of rituximab even though this is half the standard dose of two 1,000 mg doses given 15 days apart. The study was designed to determine whether even lower doses could be used.
The study enrolled 142 patients with RA who were being retreated with rituximab after responding previously to this therapy. In a 1:2:2 ratio, patients were randomized to single rituximab injections of 1,000 mg, 500 mg, or 200 mg. Outcome then were compared at the end of 6 months.
Noninferiority was defined as 0.5 difference in DAS28-CRP score (disease activity score using C-reactive protein instead of erythrocyte sedimentation rate) score adjusted for baseline disease status and use of conventional disease-modifying antirheumatic drugs (DMARDs).
Although noninferior at 3 months, the 500 mg dose did not meet the noninferiority criteria at 6 months. Due to a hierarchical design, evaluation of the 200 mg dose was precluded by the negative result with the 500 mg dose.
However, the majority of patients did respond to both the 500 mg and 200 mg dose. The failure to meet noninferiority was due to a limited number of patients who required rescue therapy for a flare. As a result, the investigators believe a trial of ultra low-dose therapy still might be reasonable.
In this interview, Ms. Verhoef explains that at her center patients who are well controlled on a 1,000 mg dose of rituximab now are being offered a 500 mg dose for retreatment. If they continue to respond, further retreatment with a 200 mg dose is considered.
Ms. Verhoef had no relevant financial disclosures.
REPORTING FROM EULAR 2019 Congress
Systemic sclerosis gastrointestinal symptoms helped by gut microbiota transplant
MADRID – compared with control subjects in a 16-week randomized, double-blind, placebo-controlled pilot study presented at the European Congress of Rheumatology.
The effects were most pronounced on lower GI symptoms, including bloating, diarrhea, and fecal incontinence, with improvement reported by three of five of the patients given the gut microbiota transplant, compared with two of the five patients who received placebo.
“We were surprised by the effect the patients reported, as all had longstanding SSc with GI symptoms,” Anna-Maria Hoffmann-Vold, MD, PhD, of Oslo University Hospital, said in an interview ahead of the congress. “We were especially surprised at the strong effect FMT had on fecal incontinence.”“Patients with systemic sclerosis are very prone to having gastrointestinal involvement – up to 90% of patients have GI symptoms, and it’s associated with very high morbidity and mortality,” she observed during her presentation at the Congress. Despite that, there currently are no disease-modifying treatments that specifically addresses GI involvement in SSc.
It’s been known for a while that patients with SSc have a different intestinal microbiota composition, or dysbiosis, compared with healthy controls, and the possibility of permanent modification of the microbiome through fecal microbiota transplant (FMT) from healthy to ill individuals has become a subject of increased attention in the scientific literature in recent years,. Dr. Hoffmann-Vold said.
In particular, FMT has shown promising results in the treatment of Clostridium difficile infections. While the current study did not focus on mechanistic pathways by which FMT might be exerting its effects, such studies are definitely warranted, she said. “One could speculate that there is a mechanistic link between dysmotility and dysbiosis in SSc, and that the manipulation of gut microbiota with FMT primarily affects motility patterns, which in turn leads to improvement of GI symptoms.”
Together with colleagues at the Oslo University Hospital, Dr. Hoffmann-Vold randomly assigned 10 patients – all women – with limited cutaneous SSc either to treatment with a commercially-available gut microbiota preparation known as anaerobic cultivated human intestinal microbiota (ACHIM) or to placebo. Both ACHIM and placebo were given via gastroduodenoscopy. Their aim was to determine the safety of the approach, as well as to obtain preliminary data on its therapeutic potential.
The UCLA GIT 2.0 score questionnaire was used to assess GI symptoms, with patients defined as responders if they met the questionnaire’s definition of a minimally clinically important difference.
Primary endpoints were safety and clinical efficacy on GI symptoms assessed at weeks 4 and 16, and safety was assessed by observation, interviews, and a standardized safety form.
Results showed improvement in GI symptoms (total UCLA GIT score) in three of the five patients who received the gut microbiota transplant versus two of the five placebo-treated patients at 16 weeks. Two patients in the active treatment versus one in the placebo group had unchanged symptoms, and one patient in the placebo group had worsening symptoms.
Adverse events associated with treatment were “transient and mild”. However, one procedure-related serious adverse event occurred in a placebo-treated patient, which was a duodenal perforation.
Concluding her presentation, Dr. Hoffman-Vold said: “FMT of commercially-available ACHIM in patients with SSc appeared safe, had beneficial effects on lower GI symptoms, altered gut microbiota composition – richness and diversity – and appeared to affect the mucosal immune system.”
The research team has just received national funding for a larger randomized clinical trial that will involve 70 SSc patients and should start towards the end of the year.
The study was sponsored by Helse Sør-øst and NKS. Dr. Hoffmann-Vold has received research funding, consulting fees, or other remuneration from Boehringer Ingelheim, GlaxoSmithKline, and Actelion. A coauthor is the owner of the company that provided the gut microbiota.
SOURCE: Hoffmann-Vold AM et al., Ann Rheum Dis. 2019 Jun. doi: 10.1136/annrheumdis-2019-eular.4684 .
MADRID – compared with control subjects in a 16-week randomized, double-blind, placebo-controlled pilot study presented at the European Congress of Rheumatology.
The effects were most pronounced on lower GI symptoms, including bloating, diarrhea, and fecal incontinence, with improvement reported by three of five of the patients given the gut microbiota transplant, compared with two of the five patients who received placebo.
“We were surprised by the effect the patients reported, as all had longstanding SSc with GI symptoms,” Anna-Maria Hoffmann-Vold, MD, PhD, of Oslo University Hospital, said in an interview ahead of the congress. “We were especially surprised at the strong effect FMT had on fecal incontinence.”“Patients with systemic sclerosis are very prone to having gastrointestinal involvement – up to 90% of patients have GI symptoms, and it’s associated with very high morbidity and mortality,” she observed during her presentation at the Congress. Despite that, there currently are no disease-modifying treatments that specifically addresses GI involvement in SSc.
It’s been known for a while that patients with SSc have a different intestinal microbiota composition, or dysbiosis, compared with healthy controls, and the possibility of permanent modification of the microbiome through fecal microbiota transplant (FMT) from healthy to ill individuals has become a subject of increased attention in the scientific literature in recent years,. Dr. Hoffmann-Vold said.
In particular, FMT has shown promising results in the treatment of Clostridium difficile infections. While the current study did not focus on mechanistic pathways by which FMT might be exerting its effects, such studies are definitely warranted, she said. “One could speculate that there is a mechanistic link between dysmotility and dysbiosis in SSc, and that the manipulation of gut microbiota with FMT primarily affects motility patterns, which in turn leads to improvement of GI symptoms.”
Together with colleagues at the Oslo University Hospital, Dr. Hoffmann-Vold randomly assigned 10 patients – all women – with limited cutaneous SSc either to treatment with a commercially-available gut microbiota preparation known as anaerobic cultivated human intestinal microbiota (ACHIM) or to placebo. Both ACHIM and placebo were given via gastroduodenoscopy. Their aim was to determine the safety of the approach, as well as to obtain preliminary data on its therapeutic potential.
The UCLA GIT 2.0 score questionnaire was used to assess GI symptoms, with patients defined as responders if they met the questionnaire’s definition of a minimally clinically important difference.
Primary endpoints were safety and clinical efficacy on GI symptoms assessed at weeks 4 and 16, and safety was assessed by observation, interviews, and a standardized safety form.
Results showed improvement in GI symptoms (total UCLA GIT score) in three of the five patients who received the gut microbiota transplant versus two of the five placebo-treated patients at 16 weeks. Two patients in the active treatment versus one in the placebo group had unchanged symptoms, and one patient in the placebo group had worsening symptoms.
Adverse events associated with treatment were “transient and mild”. However, one procedure-related serious adverse event occurred in a placebo-treated patient, which was a duodenal perforation.
Concluding her presentation, Dr. Hoffman-Vold said: “FMT of commercially-available ACHIM in patients with SSc appeared safe, had beneficial effects on lower GI symptoms, altered gut microbiota composition – richness and diversity – and appeared to affect the mucosal immune system.”
The research team has just received national funding for a larger randomized clinical trial that will involve 70 SSc patients and should start towards the end of the year.
The study was sponsored by Helse Sør-øst and NKS. Dr. Hoffmann-Vold has received research funding, consulting fees, or other remuneration from Boehringer Ingelheim, GlaxoSmithKline, and Actelion. A coauthor is the owner of the company that provided the gut microbiota.
SOURCE: Hoffmann-Vold AM et al., Ann Rheum Dis. 2019 Jun. doi: 10.1136/annrheumdis-2019-eular.4684 .
MADRID – compared with control subjects in a 16-week randomized, double-blind, placebo-controlled pilot study presented at the European Congress of Rheumatology.
The effects were most pronounced on lower GI symptoms, including bloating, diarrhea, and fecal incontinence, with improvement reported by three of five of the patients given the gut microbiota transplant, compared with two of the five patients who received placebo.
“We were surprised by the effect the patients reported, as all had longstanding SSc with GI symptoms,” Anna-Maria Hoffmann-Vold, MD, PhD, of Oslo University Hospital, said in an interview ahead of the congress. “We were especially surprised at the strong effect FMT had on fecal incontinence.”“Patients with systemic sclerosis are very prone to having gastrointestinal involvement – up to 90% of patients have GI symptoms, and it’s associated with very high morbidity and mortality,” she observed during her presentation at the Congress. Despite that, there currently are no disease-modifying treatments that specifically addresses GI involvement in SSc.
It’s been known for a while that patients with SSc have a different intestinal microbiota composition, or dysbiosis, compared with healthy controls, and the possibility of permanent modification of the microbiome through fecal microbiota transplant (FMT) from healthy to ill individuals has become a subject of increased attention in the scientific literature in recent years,. Dr. Hoffmann-Vold said.
In particular, FMT has shown promising results in the treatment of Clostridium difficile infections. While the current study did not focus on mechanistic pathways by which FMT might be exerting its effects, such studies are definitely warranted, she said. “One could speculate that there is a mechanistic link between dysmotility and dysbiosis in SSc, and that the manipulation of gut microbiota with FMT primarily affects motility patterns, which in turn leads to improvement of GI symptoms.”
Together with colleagues at the Oslo University Hospital, Dr. Hoffmann-Vold randomly assigned 10 patients – all women – with limited cutaneous SSc either to treatment with a commercially-available gut microbiota preparation known as anaerobic cultivated human intestinal microbiota (ACHIM) or to placebo. Both ACHIM and placebo were given via gastroduodenoscopy. Their aim was to determine the safety of the approach, as well as to obtain preliminary data on its therapeutic potential.
The UCLA GIT 2.0 score questionnaire was used to assess GI symptoms, with patients defined as responders if they met the questionnaire’s definition of a minimally clinically important difference.
Primary endpoints were safety and clinical efficacy on GI symptoms assessed at weeks 4 and 16, and safety was assessed by observation, interviews, and a standardized safety form.
Results showed improvement in GI symptoms (total UCLA GIT score) in three of the five patients who received the gut microbiota transplant versus two of the five placebo-treated patients at 16 weeks. Two patients in the active treatment versus one in the placebo group had unchanged symptoms, and one patient in the placebo group had worsening symptoms.
Adverse events associated with treatment were “transient and mild”. However, one procedure-related serious adverse event occurred in a placebo-treated patient, which was a duodenal perforation.
Concluding her presentation, Dr. Hoffman-Vold said: “FMT of commercially-available ACHIM in patients with SSc appeared safe, had beneficial effects on lower GI symptoms, altered gut microbiota composition – richness and diversity – and appeared to affect the mucosal immune system.”
The research team has just received national funding for a larger randomized clinical trial that will involve 70 SSc patients and should start towards the end of the year.
The study was sponsored by Helse Sør-øst and NKS. Dr. Hoffmann-Vold has received research funding, consulting fees, or other remuneration from Boehringer Ingelheim, GlaxoSmithKline, and Actelion. A coauthor is the owner of the company that provided the gut microbiota.
SOURCE: Hoffmann-Vold AM et al., Ann Rheum Dis. 2019 Jun. doi: 10.1136/annrheumdis-2019-eular.4684 .
REPORTING FROM THE EULAR 2019 CONGRESS
Retention rates comparable for biosimilars, original drug in spondyloarthritis
MADRID – judging from data drawn from registries in five Scandinavian countries in a study that evaluated retention rates after 1 year of therapy.
Bente Glintborg, MD, PhD, from the Center for Rheumatology and Spine Diseases, Rigshospitalet, Glostrup, Denmark, explains in a video interview that the indication provided to biosimilars for spondyloarthritis was extended from comparisons conducted in rheumatoid arthritis (RA).
In the absence of a randomized trial in spondyloarthritis, she suggested that this comparison might be the best opportunity to evaluate whether biosimilars perform as well as their biologic originator. This is an important aim based on the theoretical possibility that equivalence in RA does not translate into equivalence in other rheumatic conditions where biologics are indicated.
As she explains, 1,015 biologic-naïve patients initiating etanercept, a tumor necrosis factor (TNF) inhibitor, or a biosimilar were assessed at baseline and at the end of 1 year of therapy. The patients were enrolled in biologic registries maintained in Denmark, Finland, Iceland, Norway, or Sweden.
Retention rates at 1 year were numerically lower on etanercept than the biosimilars, but the difference was not significant (66% vs. 73%; P = 0.18). There also were no significant differences between the biosimilars and etanercept when disease activity was compared at 6 months.
Retention rates are a reasonable surrogate for both efficacy and tolerability based on the expectation that more patients would switch or discontinue agents in the event of lack of efficacy or unacceptable side effects, Dr. Glintborg said at the European Congress of Rheumatology.
In this interview, she notes that a similar study from the Nordic registries led by a coinvestigator also showed equivalent retention rates among spondyloarthritis patients when biosimilars and infliximab were compared at 2 years.
Dr. Glintborg received research support from Biogen, Pfizer, and Abbievie.
MADRID – judging from data drawn from registries in five Scandinavian countries in a study that evaluated retention rates after 1 year of therapy.
Bente Glintborg, MD, PhD, from the Center for Rheumatology and Spine Diseases, Rigshospitalet, Glostrup, Denmark, explains in a video interview that the indication provided to biosimilars for spondyloarthritis was extended from comparisons conducted in rheumatoid arthritis (RA).
In the absence of a randomized trial in spondyloarthritis, she suggested that this comparison might be the best opportunity to evaluate whether biosimilars perform as well as their biologic originator. This is an important aim based on the theoretical possibility that equivalence in RA does not translate into equivalence in other rheumatic conditions where biologics are indicated.
As she explains, 1,015 biologic-naïve patients initiating etanercept, a tumor necrosis factor (TNF) inhibitor, or a biosimilar were assessed at baseline and at the end of 1 year of therapy. The patients were enrolled in biologic registries maintained in Denmark, Finland, Iceland, Norway, or Sweden.
Retention rates at 1 year were numerically lower on etanercept than the biosimilars, but the difference was not significant (66% vs. 73%; P = 0.18). There also were no significant differences between the biosimilars and etanercept when disease activity was compared at 6 months.
Retention rates are a reasonable surrogate for both efficacy and tolerability based on the expectation that more patients would switch or discontinue agents in the event of lack of efficacy or unacceptable side effects, Dr. Glintborg said at the European Congress of Rheumatology.
In this interview, she notes that a similar study from the Nordic registries led by a coinvestigator also showed equivalent retention rates among spondyloarthritis patients when biosimilars and infliximab were compared at 2 years.
Dr. Glintborg received research support from Biogen, Pfizer, and Abbievie.
MADRID – judging from data drawn from registries in five Scandinavian countries in a study that evaluated retention rates after 1 year of therapy.
Bente Glintborg, MD, PhD, from the Center for Rheumatology and Spine Diseases, Rigshospitalet, Glostrup, Denmark, explains in a video interview that the indication provided to biosimilars for spondyloarthritis was extended from comparisons conducted in rheumatoid arthritis (RA).
In the absence of a randomized trial in spondyloarthritis, she suggested that this comparison might be the best opportunity to evaluate whether biosimilars perform as well as their biologic originator. This is an important aim based on the theoretical possibility that equivalence in RA does not translate into equivalence in other rheumatic conditions where biologics are indicated.
As she explains, 1,015 biologic-naïve patients initiating etanercept, a tumor necrosis factor (TNF) inhibitor, or a biosimilar were assessed at baseline and at the end of 1 year of therapy. The patients were enrolled in biologic registries maintained in Denmark, Finland, Iceland, Norway, or Sweden.
Retention rates at 1 year were numerically lower on etanercept than the biosimilars, but the difference was not significant (66% vs. 73%; P = 0.18). There also were no significant differences between the biosimilars and etanercept when disease activity was compared at 6 months.
Retention rates are a reasonable surrogate for both efficacy and tolerability based on the expectation that more patients would switch or discontinue agents in the event of lack of efficacy or unacceptable side effects, Dr. Glintborg said at the European Congress of Rheumatology.
In this interview, she notes that a similar study from the Nordic registries led by a coinvestigator also showed equivalent retention rates among spondyloarthritis patients when biosimilars and infliximab were compared at 2 years.
Dr. Glintborg received research support from Biogen, Pfizer, and Abbievie.
REPORTING FROM EULAR 2019 Congress
Video program engages patients in treat-to-target concept
MADRID – according to data generated from a randomized trial.
One of the major goals of the video program is to inform patients about the treat-to-target concept of RA management, explained Maria I. Danila, MD, a rheumatologist at the University of Alabama at Birmingham.
Although physicians know this to be a guiding tenet for RA management, she explains in this video interview that 50% or more of patients are unaware of this therapeutic goal.
For patients who resist treatment escalation for fear of side effects, this lack of awareness might provide one explanation for failing to adhere to guideline-recommended therapy, Dr. Danila said at the European Congress of Rheumatology. She believes that patients need more information about the potential for treatment escalation to improve function.
To address this issue, a short video was developed to explain the treat-to-target concept. It was then tested in a randomized trial. Those who viewed the video expressed greater willingness to change intervention on the advice of their rheumatologist relative to those who did not (P = 0.01).
Further studies are planned, including studies to test whether willingness to escalate treatment results in better outcomes and whether linking patient behavioral goals such as being able to play golf again will enhance treatment adherence. Dr. Danila envisions wide distribution of this video if further studies demonstrate that it helps patients cooperate with treatment escalation when needed.
Dr. Danila received research support from Pfizer.
MADRID – according to data generated from a randomized trial.
One of the major goals of the video program is to inform patients about the treat-to-target concept of RA management, explained Maria I. Danila, MD, a rheumatologist at the University of Alabama at Birmingham.
Although physicians know this to be a guiding tenet for RA management, she explains in this video interview that 50% or more of patients are unaware of this therapeutic goal.
For patients who resist treatment escalation for fear of side effects, this lack of awareness might provide one explanation for failing to adhere to guideline-recommended therapy, Dr. Danila said at the European Congress of Rheumatology. She believes that patients need more information about the potential for treatment escalation to improve function.
To address this issue, a short video was developed to explain the treat-to-target concept. It was then tested in a randomized trial. Those who viewed the video expressed greater willingness to change intervention on the advice of their rheumatologist relative to those who did not (P = 0.01).
Further studies are planned, including studies to test whether willingness to escalate treatment results in better outcomes and whether linking patient behavioral goals such as being able to play golf again will enhance treatment adherence. Dr. Danila envisions wide distribution of this video if further studies demonstrate that it helps patients cooperate with treatment escalation when needed.
Dr. Danila received research support from Pfizer.
MADRID – according to data generated from a randomized trial.
One of the major goals of the video program is to inform patients about the treat-to-target concept of RA management, explained Maria I. Danila, MD, a rheumatologist at the University of Alabama at Birmingham.
Although physicians know this to be a guiding tenet for RA management, she explains in this video interview that 50% or more of patients are unaware of this therapeutic goal.
For patients who resist treatment escalation for fear of side effects, this lack of awareness might provide one explanation for failing to adhere to guideline-recommended therapy, Dr. Danila said at the European Congress of Rheumatology. She believes that patients need more information about the potential for treatment escalation to improve function.
To address this issue, a short video was developed to explain the treat-to-target concept. It was then tested in a randomized trial. Those who viewed the video expressed greater willingness to change intervention on the advice of their rheumatologist relative to those who did not (P = 0.01).
Further studies are planned, including studies to test whether willingness to escalate treatment results in better outcomes and whether linking patient behavioral goals such as being able to play golf again will enhance treatment adherence. Dr. Danila envisions wide distribution of this video if further studies demonstrate that it helps patients cooperate with treatment escalation when needed.
Dr. Danila received research support from Pfizer.
REPORTING FROM EULAR 2019 Congress
Cardiovascular events in U.S. RA patients fall to non-RA level
MADRID – U.S. patients with rheumatoid arthritis stopped having an excess of cardiovascular disease events during the 2000s.
During both the 1980s and 1990s, patients with rheumatoid arthritis (RA) residing in a 27-county region in southeastern Minnesota and northwestern Wisconsin had cardiovascular disease event rates that were more than twice the rates in similar adults without RA, but that changed during the 2000s, Elena Myasoedova, MD, said in a poster she presented at the European Congress of Rheumatology. During 2000-2009, RA patients enrolled in the Rochester (Minn.) Epidemiology Project had an incidence of cardiovascular disease events at a rate that was 12% lower, compared with matched adults without RA who were also enrolled in the same regional database, reported Dr. Myasoedova, a rheumatologist at the Mayo Clinic in Rochester, and her associates.
“We hypothesize that improved management of RA, including implementation of a treat-to-target strategy and the introduction of biological drugs could have influenced this, as well as increased awareness of and improved prevention of cardiovascular disease,” Dr. Myasoedova said in an interview. The findings “give us a hint that tight control of RA disease activity is also likely to help cardiovascular disease burden.”
She and her associates identified 906 people enrolled in the Rochester Epidemiology Project who had incident RA based on the 1987 criteria of the American College of Rheumatology and matched them by age, sex, and index year with 905 people in the registry without RA. These cohorts included roughly 200 people from each subgroup tracked during the 1980s, 300 from each subgroup tracked during the 1990s, and about 400 in each subgroup tracked during the 2000s. They averaged about 56 years old, and about two-thirds were women.
During the 1980s, the cumulative incidence of nonfatal MI, nonfatal stroke, or cardiovascular disease (CVD) death was 2.11-fold more common among the RA patients than in the matched controls without RA, and during the 1990s this ratio showed a 2.13-fold excess of CVD events among the RA patients. The between-group differences in both decades were statistically significant. During the 2000s, the RA patients actually had a nominally lower rate of CVD events, at 0.88 times the rate of the controls, a difference that was not statistically significant.
Dr. Myasoedova and her associates had previously reported a similar finding in an analysis that used a smaller number of people and focused exclusively on rates of CVD (J Rheumatol. 2017 Jun;44[6]:732-9).
A few factors limit the generalizability of the finding, Dr. Myasoedova cautioned. First, the population studied was about 90% white. Also, people in the Rochester Epidemiology Project receive their medical care from clinicians at the Mayo Clinic or an affiliated hospital in the region covered by the Project.
“These data are from a large, tertiary care center,” and so the findings are most directly applicable to patients who receive medical care in a similar setting that provides guideline-directed management of both RA and CVD risk.
A long-standing hypothesis is that CVD has an inflammatory component. These data support that concept by suggesting that when inflammatory disease is well controlled in RA patients, their CVD risk drops, Dr. Myasoedova said. “CVD has been seen as the number one comorbidity for RA patients, and it remains that way, but it’s very reassuring that the CVD rate has improved. It shows we’re doing something right.”
The study received no commercial funding. Dr. Myasoedova had no relevant disclosures.
SOURCE: Myasoedova E et al. Ann Rheum Dis. Jun 2019;78(Suppl 2):1024-5. Abstract FRI0654. DOI: 10.1136/annrheumdis-2019-eular.4996.
MADRID – U.S. patients with rheumatoid arthritis stopped having an excess of cardiovascular disease events during the 2000s.
During both the 1980s and 1990s, patients with rheumatoid arthritis (RA) residing in a 27-county region in southeastern Minnesota and northwestern Wisconsin had cardiovascular disease event rates that were more than twice the rates in similar adults without RA, but that changed during the 2000s, Elena Myasoedova, MD, said in a poster she presented at the European Congress of Rheumatology. During 2000-2009, RA patients enrolled in the Rochester (Minn.) Epidemiology Project had an incidence of cardiovascular disease events at a rate that was 12% lower, compared with matched adults without RA who were also enrolled in the same regional database, reported Dr. Myasoedova, a rheumatologist at the Mayo Clinic in Rochester, and her associates.
“We hypothesize that improved management of RA, including implementation of a treat-to-target strategy and the introduction of biological drugs could have influenced this, as well as increased awareness of and improved prevention of cardiovascular disease,” Dr. Myasoedova said in an interview. The findings “give us a hint that tight control of RA disease activity is also likely to help cardiovascular disease burden.”
She and her associates identified 906 people enrolled in the Rochester Epidemiology Project who had incident RA based on the 1987 criteria of the American College of Rheumatology and matched them by age, sex, and index year with 905 people in the registry without RA. These cohorts included roughly 200 people from each subgroup tracked during the 1980s, 300 from each subgroup tracked during the 1990s, and about 400 in each subgroup tracked during the 2000s. They averaged about 56 years old, and about two-thirds were women.
During the 1980s, the cumulative incidence of nonfatal MI, nonfatal stroke, or cardiovascular disease (CVD) death was 2.11-fold more common among the RA patients than in the matched controls without RA, and during the 1990s this ratio showed a 2.13-fold excess of CVD events among the RA patients. The between-group differences in both decades were statistically significant. During the 2000s, the RA patients actually had a nominally lower rate of CVD events, at 0.88 times the rate of the controls, a difference that was not statistically significant.
Dr. Myasoedova and her associates had previously reported a similar finding in an analysis that used a smaller number of people and focused exclusively on rates of CVD (J Rheumatol. 2017 Jun;44[6]:732-9).
A few factors limit the generalizability of the finding, Dr. Myasoedova cautioned. First, the population studied was about 90% white. Also, people in the Rochester Epidemiology Project receive their medical care from clinicians at the Mayo Clinic or an affiliated hospital in the region covered by the Project.
“These data are from a large, tertiary care center,” and so the findings are most directly applicable to patients who receive medical care in a similar setting that provides guideline-directed management of both RA and CVD risk.
A long-standing hypothesis is that CVD has an inflammatory component. These data support that concept by suggesting that when inflammatory disease is well controlled in RA patients, their CVD risk drops, Dr. Myasoedova said. “CVD has been seen as the number one comorbidity for RA patients, and it remains that way, but it’s very reassuring that the CVD rate has improved. It shows we’re doing something right.”
The study received no commercial funding. Dr. Myasoedova had no relevant disclosures.
SOURCE: Myasoedova E et al. Ann Rheum Dis. Jun 2019;78(Suppl 2):1024-5. Abstract FRI0654. DOI: 10.1136/annrheumdis-2019-eular.4996.
MADRID – U.S. patients with rheumatoid arthritis stopped having an excess of cardiovascular disease events during the 2000s.
During both the 1980s and 1990s, patients with rheumatoid arthritis (RA) residing in a 27-county region in southeastern Minnesota and northwestern Wisconsin had cardiovascular disease event rates that were more than twice the rates in similar adults without RA, but that changed during the 2000s, Elena Myasoedova, MD, said in a poster she presented at the European Congress of Rheumatology. During 2000-2009, RA patients enrolled in the Rochester (Minn.) Epidemiology Project had an incidence of cardiovascular disease events at a rate that was 12% lower, compared with matched adults without RA who were also enrolled in the same regional database, reported Dr. Myasoedova, a rheumatologist at the Mayo Clinic in Rochester, and her associates.
“We hypothesize that improved management of RA, including implementation of a treat-to-target strategy and the introduction of biological drugs could have influenced this, as well as increased awareness of and improved prevention of cardiovascular disease,” Dr. Myasoedova said in an interview. The findings “give us a hint that tight control of RA disease activity is also likely to help cardiovascular disease burden.”
She and her associates identified 906 people enrolled in the Rochester Epidemiology Project who had incident RA based on the 1987 criteria of the American College of Rheumatology and matched them by age, sex, and index year with 905 people in the registry without RA. These cohorts included roughly 200 people from each subgroup tracked during the 1980s, 300 from each subgroup tracked during the 1990s, and about 400 in each subgroup tracked during the 2000s. They averaged about 56 years old, and about two-thirds were women.
During the 1980s, the cumulative incidence of nonfatal MI, nonfatal stroke, or cardiovascular disease (CVD) death was 2.11-fold more common among the RA patients than in the matched controls without RA, and during the 1990s this ratio showed a 2.13-fold excess of CVD events among the RA patients. The between-group differences in both decades were statistically significant. During the 2000s, the RA patients actually had a nominally lower rate of CVD events, at 0.88 times the rate of the controls, a difference that was not statistically significant.
Dr. Myasoedova and her associates had previously reported a similar finding in an analysis that used a smaller number of people and focused exclusively on rates of CVD (J Rheumatol. 2017 Jun;44[6]:732-9).
A few factors limit the generalizability of the finding, Dr. Myasoedova cautioned. First, the population studied was about 90% white. Also, people in the Rochester Epidemiology Project receive their medical care from clinicians at the Mayo Clinic or an affiliated hospital in the region covered by the Project.
“These data are from a large, tertiary care center,” and so the findings are most directly applicable to patients who receive medical care in a similar setting that provides guideline-directed management of both RA and CVD risk.
A long-standing hypothesis is that CVD has an inflammatory component. These data support that concept by suggesting that when inflammatory disease is well controlled in RA patients, their CVD risk drops, Dr. Myasoedova said. “CVD has been seen as the number one comorbidity for RA patients, and it remains that way, but it’s very reassuring that the CVD rate has improved. It shows we’re doing something right.”
The study received no commercial funding. Dr. Myasoedova had no relevant disclosures.
SOURCE: Myasoedova E et al. Ann Rheum Dis. Jun 2019;78(Suppl 2):1024-5. Abstract FRI0654. DOI: 10.1136/annrheumdis-2019-eular.4996.
REPORTING FROM EULAR 2019 CONGRESS
Sequential dermoscopy imaging helps find melanomas early
WAIKOLOA, HAWAII – Sequential dermoscopy imaging (SDI) is a valuable strategy for diagnosing melanomas early and with better sensitivity and specificity, compared with biopsy decisions based solely on the ugly duckling sign, the ABCDs of melanoma, or other aspects of lesion morphology, Michael A. Marchetti, MD, said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
SDI entails obtaining repeated dermoscopy images over time in order to detect subtle changes. It is typically done short term, over the course of 3-4 months, or longer term, over a period of 6 months to years, with long-term SDI being reserved for monitoring of less suspicious lesions, often in patients with an atypical mole syndrome.
SDI improves diagnostic specificity by dramatically reducing excision of benign pigmented lesions: in one large Belgian study, by up to 75% (Br J Dermatol. 2012 Oct;167[4]:778-86).
Short-term SDI also improves diagnostic sensitivity. That’s because it enables early identification of clinically featureless melanomas that are detected solely based upon change over a 3-month follow-up period. The operative principle here is that 93%-96% of melanomas will show change on dermoscopy within 3 months, while 99% of unchanged melanocytic lesions are benign. Since 16% of benign nevi will change within 3 months, that means 10%-30% of changed lesions are melanomas.
“If there is any change – it doesn’t matter what the change is, but the two images look different – that should lead to a biopsy,” explained Dr. Marchetti, a dermatologist at Memorial Sloan Kettering Cancer Center, New York.
As a result of this improved sensitivity and specificity, SDI has been shown to reduce the cost per melanoma diagnosis by about 40% (PLoS One. 2014 Oct 14;9[10]:e109339. doi: 10.1371/journal.pone.0109339).
Dr. Marchetti considers SDI a second-level diagnostic test for individual equivocal lesions. His first-level diagnostic tool is total-body photography.
SDI needs to be done by scrupulous examination of digital photographic images side-by-side on a computer monitor. A basic rule of SDI is that it should never be used to monitor raised or palpable lesions.
“The only thing you can monitor is something that’s flat,” he stressed.
Nor should SDI be used to monitor lesions with a peripheral globular pattern. And very slow-growing melanomas could potentially be missed by short-term SDI, so suspected lentigo maligna should be monitored for a minimum of 12 months, according to Dr. Marchetti.
Not every patient with an equivocal melanocytic lesion is a good candidate for SDI. It’s a monitoring strategy that should be reserved for reliable patients who will come back in 3 months. “If a patient doesn’t come back I take that very seriously. We call or send a letter,” Dr. Marchetti said.
Moreover, even in a patient who is a good candidate for SDI, he always offers the option of biopsy today rather than short-term monitoring.
SDI employed in conjunction with total-body photography is an extremely effective means of monitoring patients at very high risk for melanoma, Dr. Marchetti said. The power of this combination was illustrated in a prospective Australian study of 311 patients with a history of invasive melanoma plus either a high-risk genetic mutation or a strong family history. During a median follow-up of 3.5 years, 75 melanomas were detected, 14 of them at the baseline visit. The median thickness of melanomas detected post baseline was in situ. Thirty-nine percent of melanomas were detected using SDI and 38% via total body photography. Roughly one in five biopsied melanocytic lesions proved to be melanoma. Of note, five of the melanomas were more than 1 mm in Breslow thickness: Three of them were histologically desmoplastic, and the other two had nodular components (JAMA Dermatol. 2014 Aug;150(8):819-27).
For dermatologists who need to brush up on their dermoscopy skills, Dr. Marchetti recommended dermoscopedia as a useful, free resource.
Legal implications of monitoring via photography
“People often get worked up about this, but I’m not aware of a lawsuit alleging missed melanoma using baseline photography as evidence. And patients, in my experience, are universally appreciative of the use of imaging, although admittedly my experience is biased because people generally come to me for imaging,” Dr. Marchetti said.
He makes a point of telling every patient who opts for short-term SDI that, although the lesion has no features of concern now, it’s important to return promptly for reexamination should any changes occur.
Dr. Marchetti reported having no financial conflicts regarding his presentation.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Sequential dermoscopy imaging (SDI) is a valuable strategy for diagnosing melanomas early and with better sensitivity and specificity, compared with biopsy decisions based solely on the ugly duckling sign, the ABCDs of melanoma, or other aspects of lesion morphology, Michael A. Marchetti, MD, said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
SDI entails obtaining repeated dermoscopy images over time in order to detect subtle changes. It is typically done short term, over the course of 3-4 months, or longer term, over a period of 6 months to years, with long-term SDI being reserved for monitoring of less suspicious lesions, often in patients with an atypical mole syndrome.
SDI improves diagnostic specificity by dramatically reducing excision of benign pigmented lesions: in one large Belgian study, by up to 75% (Br J Dermatol. 2012 Oct;167[4]:778-86).
Short-term SDI also improves diagnostic sensitivity. That’s because it enables early identification of clinically featureless melanomas that are detected solely based upon change over a 3-month follow-up period. The operative principle here is that 93%-96% of melanomas will show change on dermoscopy within 3 months, while 99% of unchanged melanocytic lesions are benign. Since 16% of benign nevi will change within 3 months, that means 10%-30% of changed lesions are melanomas.
“If there is any change – it doesn’t matter what the change is, but the two images look different – that should lead to a biopsy,” explained Dr. Marchetti, a dermatologist at Memorial Sloan Kettering Cancer Center, New York.
As a result of this improved sensitivity and specificity, SDI has been shown to reduce the cost per melanoma diagnosis by about 40% (PLoS One. 2014 Oct 14;9[10]:e109339. doi: 10.1371/journal.pone.0109339).
Dr. Marchetti considers SDI a second-level diagnostic test for individual equivocal lesions. His first-level diagnostic tool is total-body photography.
SDI needs to be done by scrupulous examination of digital photographic images side-by-side on a computer monitor. A basic rule of SDI is that it should never be used to monitor raised or palpable lesions.
“The only thing you can monitor is something that’s flat,” he stressed.
Nor should SDI be used to monitor lesions with a peripheral globular pattern. And very slow-growing melanomas could potentially be missed by short-term SDI, so suspected lentigo maligna should be monitored for a minimum of 12 months, according to Dr. Marchetti.
Not every patient with an equivocal melanocytic lesion is a good candidate for SDI. It’s a monitoring strategy that should be reserved for reliable patients who will come back in 3 months. “If a patient doesn’t come back I take that very seriously. We call or send a letter,” Dr. Marchetti said.
Moreover, even in a patient who is a good candidate for SDI, he always offers the option of biopsy today rather than short-term monitoring.
SDI employed in conjunction with total-body photography is an extremely effective means of monitoring patients at very high risk for melanoma, Dr. Marchetti said. The power of this combination was illustrated in a prospective Australian study of 311 patients with a history of invasive melanoma plus either a high-risk genetic mutation or a strong family history. During a median follow-up of 3.5 years, 75 melanomas were detected, 14 of them at the baseline visit. The median thickness of melanomas detected post baseline was in situ. Thirty-nine percent of melanomas were detected using SDI and 38% via total body photography. Roughly one in five biopsied melanocytic lesions proved to be melanoma. Of note, five of the melanomas were more than 1 mm in Breslow thickness: Three of them were histologically desmoplastic, and the other two had nodular components (JAMA Dermatol. 2014 Aug;150(8):819-27).
For dermatologists who need to brush up on their dermoscopy skills, Dr. Marchetti recommended dermoscopedia as a useful, free resource.
Legal implications of monitoring via photography
“People often get worked up about this, but I’m not aware of a lawsuit alleging missed melanoma using baseline photography as evidence. And patients, in my experience, are universally appreciative of the use of imaging, although admittedly my experience is biased because people generally come to me for imaging,” Dr. Marchetti said.
He makes a point of telling every patient who opts for short-term SDI that, although the lesion has no features of concern now, it’s important to return promptly for reexamination should any changes occur.
Dr. Marchetti reported having no financial conflicts regarding his presentation.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Sequential dermoscopy imaging (SDI) is a valuable strategy for diagnosing melanomas early and with better sensitivity and specificity, compared with biopsy decisions based solely on the ugly duckling sign, the ABCDs of melanoma, or other aspects of lesion morphology, Michael A. Marchetti, MD, said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
SDI entails obtaining repeated dermoscopy images over time in order to detect subtle changes. It is typically done short term, over the course of 3-4 months, or longer term, over a period of 6 months to years, with long-term SDI being reserved for monitoring of less suspicious lesions, often in patients with an atypical mole syndrome.
SDI improves diagnostic specificity by dramatically reducing excision of benign pigmented lesions: in one large Belgian study, by up to 75% (Br J Dermatol. 2012 Oct;167[4]:778-86).
Short-term SDI also improves diagnostic sensitivity. That’s because it enables early identification of clinically featureless melanomas that are detected solely based upon change over a 3-month follow-up period. The operative principle here is that 93%-96% of melanomas will show change on dermoscopy within 3 months, while 99% of unchanged melanocytic lesions are benign. Since 16% of benign nevi will change within 3 months, that means 10%-30% of changed lesions are melanomas.
“If there is any change – it doesn’t matter what the change is, but the two images look different – that should lead to a biopsy,” explained Dr. Marchetti, a dermatologist at Memorial Sloan Kettering Cancer Center, New York.
As a result of this improved sensitivity and specificity, SDI has been shown to reduce the cost per melanoma diagnosis by about 40% (PLoS One. 2014 Oct 14;9[10]:e109339. doi: 10.1371/journal.pone.0109339).
Dr. Marchetti considers SDI a second-level diagnostic test for individual equivocal lesions. His first-level diagnostic tool is total-body photography.
SDI needs to be done by scrupulous examination of digital photographic images side-by-side on a computer monitor. A basic rule of SDI is that it should never be used to monitor raised or palpable lesions.
“The only thing you can monitor is something that’s flat,” he stressed.
Nor should SDI be used to monitor lesions with a peripheral globular pattern. And very slow-growing melanomas could potentially be missed by short-term SDI, so suspected lentigo maligna should be monitored for a minimum of 12 months, according to Dr. Marchetti.
Not every patient with an equivocal melanocytic lesion is a good candidate for SDI. It’s a monitoring strategy that should be reserved for reliable patients who will come back in 3 months. “If a patient doesn’t come back I take that very seriously. We call or send a letter,” Dr. Marchetti said.
Moreover, even in a patient who is a good candidate for SDI, he always offers the option of biopsy today rather than short-term monitoring.
SDI employed in conjunction with total-body photography is an extremely effective means of monitoring patients at very high risk for melanoma, Dr. Marchetti said. The power of this combination was illustrated in a prospective Australian study of 311 patients with a history of invasive melanoma plus either a high-risk genetic mutation or a strong family history. During a median follow-up of 3.5 years, 75 melanomas were detected, 14 of them at the baseline visit. The median thickness of melanomas detected post baseline was in situ. Thirty-nine percent of melanomas were detected using SDI and 38% via total body photography. Roughly one in five biopsied melanocytic lesions proved to be melanoma. Of note, five of the melanomas were more than 1 mm in Breslow thickness: Three of them were histologically desmoplastic, and the other two had nodular components (JAMA Dermatol. 2014 Aug;150(8):819-27).
For dermatologists who need to brush up on their dermoscopy skills, Dr. Marchetti recommended dermoscopedia as a useful, free resource.
Legal implications of monitoring via photography
“People often get worked up about this, but I’m not aware of a lawsuit alleging missed melanoma using baseline photography as evidence. And patients, in my experience, are universally appreciative of the use of imaging, although admittedly my experience is biased because people generally come to me for imaging,” Dr. Marchetti said.
He makes a point of telling every patient who opts for short-term SDI that, although the lesion has no features of concern now, it’s important to return promptly for reexamination should any changes occur.
Dr. Marchetti reported having no financial conflicts regarding his presentation.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
REPORTING FROM SDEF HAWAII DERMATOLOGY SEMINAR