User login
Sharon Worcester is an award-winning medical journalist for MDedge News. She has been with the company since 1996, first as the Southeast Bureau Chief (1996-2009) when the company was known as International Medical News Group, then as a freelance writer (2010-2015) before returning as a reporter in 2015. She previously worked as a daily newspaper reporter covering health and local government. Sharon currently reports primarily on oncology and hematology. She has a BA from Eckerd College and an MA in Mass Communication/Print Journalism from the University of Florida. Connect with her via LinkedIn and follow her on twitter @SW_MedReporter.
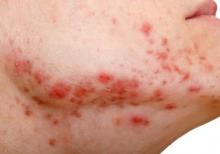
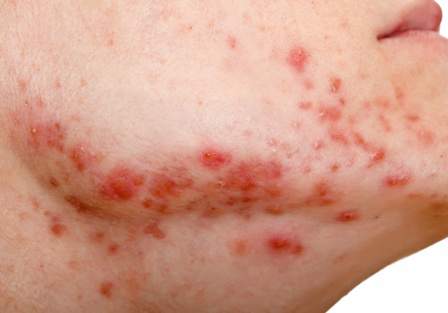
Wrinkle filler also works for acne scars
ORLANDO – A non-resorbable wrinkle filler proved highly effective and durable for the treatment of atrophic acne scars in a randomized, controlled, multicenter study.
At 1 month after treatment with polymethylmethacrylate-collagen, or PMMA-collagen (Artefill, Suneva Medical, Inc.), nearly 70% of 97 subjects showed at least a 2-point improvement on the validated 4-point Acne Scar Rating Scale, compared with about 40% of 50 control subjects injected with saline. At 6 months, the response rate remained above 60% in the PMMA-collagen group, but dropped closer to 30% among those in the control group, Dr. James M. Spencer of Mount Sinai School of Medicine, New York reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
The control group subjects were then crossed over to the treatment group, and, at 12 months, the response rates were about 70% and nearly 60% in the treatment and control groups, respectively, he said.
Similarly, both Physician and Subject Global Aesthetic Improvement Scale (PGAIS/SGAIS) scores diverged during a 6-month evaluator-blinded phase of the study, then converged after crossover by the control group subjects. For example, the percentage of treatment and control group subjects with improvement at 1 month and 6 months according to the 5-point PGAIS was about 90% vs. less than 65%, and about 80% vs. about 30%, respectively. More than 90% in both groups showed improvement at 12 months, after control group crossover.
Additionally, subject satisfaction at 1 and 6 months in the treatment and control groups based on assessment of scar correction using Patient Satisfaction Scale scores was above 80% vs. about 60%, and about 80% vs. about 50%, respectively. Satisfaction in both groups was between 80% and 90% at 12 months, after control group crossover.
Study subjects, who had a mean age of 44 years, were enrolled from 10 U.S. centers and were treated during one injection session. An additional touch-up injection was allowed as needed. A total of 1,292 scars were treated in the 97 treatment group subjects, and 424 were treated in the 50 control group subjects. Participants were evaluated by blinded assessors at 2 weeks and 1, 3, and 6 months, after which control group subjects were treated with PMMA-collagen. Assessments were made in open-label fashion at 9 and 12 months. Most subjects (61%) were women, and 20% had Fitzpatrick skin types 5 or 6.
During the blinded portion of the study, six treatment-related adverse events were reported among treatment group subjects, and two were reported among control group subjects. None of the subjects experienced granulomas, changes in pigmentation, or hypertrophic scarring.
This study is the first randomized, blinded study of PMMA-collagen for treating acne scars, Dr. Spencer noted, adding that the findings demonstrate the efficacy and safety of PMMA-collagen for this purpose.
“The improvement is durable, lasting for 12 months,” he wrote, noting that the filler is easily administered and requires minimal training in those who are familiar with dermal fillers.
“PMMA-collagen works very well on deep, severe acne scars, and should also work very well on shallow scars,” he said.
The product may enable practitioners to effectively treat acne scarring without a large capital equipment expenditure and without the risks associated with resurfacing procedures,” he said.
This study was sponsored by Suneva Medical, Inc.
ORLANDO – A non-resorbable wrinkle filler proved highly effective and durable for the treatment of atrophic acne scars in a randomized, controlled, multicenter study.
At 1 month after treatment with polymethylmethacrylate-collagen, or PMMA-collagen (Artefill, Suneva Medical, Inc.), nearly 70% of 97 subjects showed at least a 2-point improvement on the validated 4-point Acne Scar Rating Scale, compared with about 40% of 50 control subjects injected with saline. At 6 months, the response rate remained above 60% in the PMMA-collagen group, but dropped closer to 30% among those in the control group, Dr. James M. Spencer of Mount Sinai School of Medicine, New York reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
The control group subjects were then crossed over to the treatment group, and, at 12 months, the response rates were about 70% and nearly 60% in the treatment and control groups, respectively, he said.
Similarly, both Physician and Subject Global Aesthetic Improvement Scale (PGAIS/SGAIS) scores diverged during a 6-month evaluator-blinded phase of the study, then converged after crossover by the control group subjects. For example, the percentage of treatment and control group subjects with improvement at 1 month and 6 months according to the 5-point PGAIS was about 90% vs. less than 65%, and about 80% vs. about 30%, respectively. More than 90% in both groups showed improvement at 12 months, after control group crossover.
Additionally, subject satisfaction at 1 and 6 months in the treatment and control groups based on assessment of scar correction using Patient Satisfaction Scale scores was above 80% vs. about 60%, and about 80% vs. about 50%, respectively. Satisfaction in both groups was between 80% and 90% at 12 months, after control group crossover.
Study subjects, who had a mean age of 44 years, were enrolled from 10 U.S. centers and were treated during one injection session. An additional touch-up injection was allowed as needed. A total of 1,292 scars were treated in the 97 treatment group subjects, and 424 were treated in the 50 control group subjects. Participants were evaluated by blinded assessors at 2 weeks and 1, 3, and 6 months, after which control group subjects were treated with PMMA-collagen. Assessments were made in open-label fashion at 9 and 12 months. Most subjects (61%) were women, and 20% had Fitzpatrick skin types 5 or 6.
During the blinded portion of the study, six treatment-related adverse events were reported among treatment group subjects, and two were reported among control group subjects. None of the subjects experienced granulomas, changes in pigmentation, or hypertrophic scarring.
This study is the first randomized, blinded study of PMMA-collagen for treating acne scars, Dr. Spencer noted, adding that the findings demonstrate the efficacy and safety of PMMA-collagen for this purpose.
“The improvement is durable, lasting for 12 months,” he wrote, noting that the filler is easily administered and requires minimal training in those who are familiar with dermal fillers.
“PMMA-collagen works very well on deep, severe acne scars, and should also work very well on shallow scars,” he said.
The product may enable practitioners to effectively treat acne scarring without a large capital equipment expenditure and without the risks associated with resurfacing procedures,” he said.
This study was sponsored by Suneva Medical, Inc.
ORLANDO – A non-resorbable wrinkle filler proved highly effective and durable for the treatment of atrophic acne scars in a randomized, controlled, multicenter study.
At 1 month after treatment with polymethylmethacrylate-collagen, or PMMA-collagen (Artefill, Suneva Medical, Inc.), nearly 70% of 97 subjects showed at least a 2-point improvement on the validated 4-point Acne Scar Rating Scale, compared with about 40% of 50 control subjects injected with saline. At 6 months, the response rate remained above 60% in the PMMA-collagen group, but dropped closer to 30% among those in the control group, Dr. James M. Spencer of Mount Sinai School of Medicine, New York reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
The control group subjects were then crossed over to the treatment group, and, at 12 months, the response rates were about 70% and nearly 60% in the treatment and control groups, respectively, he said.
Similarly, both Physician and Subject Global Aesthetic Improvement Scale (PGAIS/SGAIS) scores diverged during a 6-month evaluator-blinded phase of the study, then converged after crossover by the control group subjects. For example, the percentage of treatment and control group subjects with improvement at 1 month and 6 months according to the 5-point PGAIS was about 90% vs. less than 65%, and about 80% vs. about 30%, respectively. More than 90% in both groups showed improvement at 12 months, after control group crossover.
Additionally, subject satisfaction at 1 and 6 months in the treatment and control groups based on assessment of scar correction using Patient Satisfaction Scale scores was above 80% vs. about 60%, and about 80% vs. about 50%, respectively. Satisfaction in both groups was between 80% and 90% at 12 months, after control group crossover.
Study subjects, who had a mean age of 44 years, were enrolled from 10 U.S. centers and were treated during one injection session. An additional touch-up injection was allowed as needed. A total of 1,292 scars were treated in the 97 treatment group subjects, and 424 were treated in the 50 control group subjects. Participants were evaluated by blinded assessors at 2 weeks and 1, 3, and 6 months, after which control group subjects were treated with PMMA-collagen. Assessments were made in open-label fashion at 9 and 12 months. Most subjects (61%) were women, and 20% had Fitzpatrick skin types 5 or 6.
During the blinded portion of the study, six treatment-related adverse events were reported among treatment group subjects, and two were reported among control group subjects. None of the subjects experienced granulomas, changes in pigmentation, or hypertrophic scarring.
This study is the first randomized, blinded study of PMMA-collagen for treating acne scars, Dr. Spencer noted, adding that the findings demonstrate the efficacy and safety of PMMA-collagen for this purpose.
“The improvement is durable, lasting for 12 months,” he wrote, noting that the filler is easily administered and requires minimal training in those who are familiar with dermal fillers.
“PMMA-collagen works very well on deep, severe acne scars, and should also work very well on shallow scars,” he said.
The product may enable practitioners to effectively treat acne scarring without a large capital equipment expenditure and without the risks associated with resurfacing procedures,” he said.
This study was sponsored by Suneva Medical, Inc.
AT THE ODAC CONFERENCE
Key clinical point: PMMA-collagen is safe, effective, and practical for treating atrophic acne scars.
Major finding: Nearly 70% of treated subjects vs. 40% of controls showed at least a 2-point improvement on the Acne Scar Rating Scale
Data source: A randomized, controlled, multicenter study of 147 subjects.
Disclosures: This study was sponsored by Suneva Medical, Inc.
Post-lumpectomy radiotherapy benefits good-risk DCIS patients
The ipsilateral local failure rate in women with good-risk ductal carcinoma in situ was low with observation after breast-conserving surgery, but was significantly lower with the addition of radiotherapy, according to findings from a prospective, randomized, Radiation Therapy Oncology Group trial.
Failure occurred in 19 of 298 patients in the observation group, compared with 2 of 287 patients in the radiotherapy group; the cumulative rates of ipsilateral local failure at 7 years were 6.7% and 0.9% in the groups, respectively (hazard ratio, 0.11), Dr. Beryl McCormick of Memorial Sloan Kettering Cancer Center, New York, and her colleagues reported online Jan. 19 in the Journal of Clinical Oncology.
Local failures were invasive in 42% of cases in the observation arm and in one of the two patients in the radiotherapy arm. The 7-year cumulative mastectomy incidence was 2.8% in the observation arm and 1.5% in the radiotherapy arm. Survival was excellent and did not differ between the arms, the investigators said (J. Clin. Oncol. 2015 Jan. 19 [doi:10.1200/JCO.2014.57.9029]).
Study subjects were women with a mean age of 58 years and mammographically detected low- or intermediate-grade ductal carcinoma in situ (DCIS) that measured less than 2.5 cm with margins of at least 3 mm. Those assigned to the radiotherapy group underwent whole-breast radiotherapy at a dose of 50 Gy in 25 fractions, 50.4 Gy in 28 fractions, or 42.5 Gy in 16 fractions, and most received tamoxifen, which was required in both arms when the study opened before being made optional in 2001.
The women were followed for a median of 7.2 years.
Grade 1 and 2 toxicities occurred more often in the radiotherapy group (30% vs. 76%), but grade 3 and 4 toxicities occurred in 4% of women in both groups. Late toxicity in the radiotherapy patients was grade 1 in 30%, grade 2 in 4.6%, and grade 3 in 0.7% of subjects, the investigators said.
Breast-conserving surgery and radiation have been shown to produce results that are equivalent to mastectomy in patients with DCIS, and in four large prospective trials, radiation therapy reduced the risk of local failure by at least 50%. About half of the recurrences in those trials were DCIS and half were invasive breast cancer, but survival was excellent regardless of local treatment, the investigators explained.
“More recently, an understanding of DCIS as not just one disease but a group of related subtypes of cancers has emerged, with a spectrum of local failure risk. Is there a low-risk DCIS for which the benefit of radiotherapy would not be seen?” they wrote.
The current study was designed to address the question of radiotherapy benefit in a good-risk DCIS subset. Although it didn’t meet the targeted accrual of 1,790 patients and was thus closed early, the findings – along with those from an Eastern Cooperative Oncology Group trial, “imply that clinical pathologic criteria can be used to define a cohort of patients with DCIS who can be expected to have a much lower rate of in-breast recurrence without radiotherapy in the first 7 years after lumpectomy than previously reported in past randomized trials,” they said.
These data may support decisions to forego adjuvant radiotherapy after breast-conserving surgery, particularly given the low mastectomy rate, but they also confirm that radiotherapy provides significant benefit with respect to further reducing local failure risk, they added.
However, given historic patterns of increasing local failure rates over 10 to 15 years, longer follow-up of these patients is needed to fully realize the rates of local failure, they said.
This study was supported by grants from the National Institutes of Health. Dr. McCormick reported having no disclosures.
The ipsilateral local failure rate in women with good-risk ductal carcinoma in situ was low with observation after breast-conserving surgery, but was significantly lower with the addition of radiotherapy, according to findings from a prospective, randomized, Radiation Therapy Oncology Group trial.
Failure occurred in 19 of 298 patients in the observation group, compared with 2 of 287 patients in the radiotherapy group; the cumulative rates of ipsilateral local failure at 7 years were 6.7% and 0.9% in the groups, respectively (hazard ratio, 0.11), Dr. Beryl McCormick of Memorial Sloan Kettering Cancer Center, New York, and her colleagues reported online Jan. 19 in the Journal of Clinical Oncology.
Local failures were invasive in 42% of cases in the observation arm and in one of the two patients in the radiotherapy arm. The 7-year cumulative mastectomy incidence was 2.8% in the observation arm and 1.5% in the radiotherapy arm. Survival was excellent and did not differ between the arms, the investigators said (J. Clin. Oncol. 2015 Jan. 19 [doi:10.1200/JCO.2014.57.9029]).
Study subjects were women with a mean age of 58 years and mammographically detected low- or intermediate-grade ductal carcinoma in situ (DCIS) that measured less than 2.5 cm with margins of at least 3 mm. Those assigned to the radiotherapy group underwent whole-breast radiotherapy at a dose of 50 Gy in 25 fractions, 50.4 Gy in 28 fractions, or 42.5 Gy in 16 fractions, and most received tamoxifen, which was required in both arms when the study opened before being made optional in 2001.
The women were followed for a median of 7.2 years.
Grade 1 and 2 toxicities occurred more often in the radiotherapy group (30% vs. 76%), but grade 3 and 4 toxicities occurred in 4% of women in both groups. Late toxicity in the radiotherapy patients was grade 1 in 30%, grade 2 in 4.6%, and grade 3 in 0.7% of subjects, the investigators said.
Breast-conserving surgery and radiation have been shown to produce results that are equivalent to mastectomy in patients with DCIS, and in four large prospective trials, radiation therapy reduced the risk of local failure by at least 50%. About half of the recurrences in those trials were DCIS and half were invasive breast cancer, but survival was excellent regardless of local treatment, the investigators explained.
“More recently, an understanding of DCIS as not just one disease but a group of related subtypes of cancers has emerged, with a spectrum of local failure risk. Is there a low-risk DCIS for which the benefit of radiotherapy would not be seen?” they wrote.
The current study was designed to address the question of radiotherapy benefit in a good-risk DCIS subset. Although it didn’t meet the targeted accrual of 1,790 patients and was thus closed early, the findings – along with those from an Eastern Cooperative Oncology Group trial, “imply that clinical pathologic criteria can be used to define a cohort of patients with DCIS who can be expected to have a much lower rate of in-breast recurrence without radiotherapy in the first 7 years after lumpectomy than previously reported in past randomized trials,” they said.
These data may support decisions to forego adjuvant radiotherapy after breast-conserving surgery, particularly given the low mastectomy rate, but they also confirm that radiotherapy provides significant benefit with respect to further reducing local failure risk, they added.
However, given historic patterns of increasing local failure rates over 10 to 15 years, longer follow-up of these patients is needed to fully realize the rates of local failure, they said.
This study was supported by grants from the National Institutes of Health. Dr. McCormick reported having no disclosures.
The ipsilateral local failure rate in women with good-risk ductal carcinoma in situ was low with observation after breast-conserving surgery, but was significantly lower with the addition of radiotherapy, according to findings from a prospective, randomized, Radiation Therapy Oncology Group trial.
Failure occurred in 19 of 298 patients in the observation group, compared with 2 of 287 patients in the radiotherapy group; the cumulative rates of ipsilateral local failure at 7 years were 6.7% and 0.9% in the groups, respectively (hazard ratio, 0.11), Dr. Beryl McCormick of Memorial Sloan Kettering Cancer Center, New York, and her colleagues reported online Jan. 19 in the Journal of Clinical Oncology.
Local failures were invasive in 42% of cases in the observation arm and in one of the two patients in the radiotherapy arm. The 7-year cumulative mastectomy incidence was 2.8% in the observation arm and 1.5% in the radiotherapy arm. Survival was excellent and did not differ between the arms, the investigators said (J. Clin. Oncol. 2015 Jan. 19 [doi:10.1200/JCO.2014.57.9029]).
Study subjects were women with a mean age of 58 years and mammographically detected low- or intermediate-grade ductal carcinoma in situ (DCIS) that measured less than 2.5 cm with margins of at least 3 mm. Those assigned to the radiotherapy group underwent whole-breast radiotherapy at a dose of 50 Gy in 25 fractions, 50.4 Gy in 28 fractions, or 42.5 Gy in 16 fractions, and most received tamoxifen, which was required in both arms when the study opened before being made optional in 2001.
The women were followed for a median of 7.2 years.
Grade 1 and 2 toxicities occurred more often in the radiotherapy group (30% vs. 76%), but grade 3 and 4 toxicities occurred in 4% of women in both groups. Late toxicity in the radiotherapy patients was grade 1 in 30%, grade 2 in 4.6%, and grade 3 in 0.7% of subjects, the investigators said.
Breast-conserving surgery and radiation have been shown to produce results that are equivalent to mastectomy in patients with DCIS, and in four large prospective trials, radiation therapy reduced the risk of local failure by at least 50%. About half of the recurrences in those trials were DCIS and half were invasive breast cancer, but survival was excellent regardless of local treatment, the investigators explained.
“More recently, an understanding of DCIS as not just one disease but a group of related subtypes of cancers has emerged, with a spectrum of local failure risk. Is there a low-risk DCIS for which the benefit of radiotherapy would not be seen?” they wrote.
The current study was designed to address the question of radiotherapy benefit in a good-risk DCIS subset. Although it didn’t meet the targeted accrual of 1,790 patients and was thus closed early, the findings – along with those from an Eastern Cooperative Oncology Group trial, “imply that clinical pathologic criteria can be used to define a cohort of patients with DCIS who can be expected to have a much lower rate of in-breast recurrence without radiotherapy in the first 7 years after lumpectomy than previously reported in past randomized trials,” they said.
These data may support decisions to forego adjuvant radiotherapy after breast-conserving surgery, particularly given the low mastectomy rate, but they also confirm that radiotherapy provides significant benefit with respect to further reducing local failure risk, they added.
However, given historic patterns of increasing local failure rates over 10 to 15 years, longer follow-up of these patients is needed to fully realize the rates of local failure, they said.
This study was supported by grants from the National Institutes of Health. Dr. McCormick reported having no disclosures.
Key clinical point: Foregoing adjuvant radiotherapy after breast-conserving surgery is a reasonable choice, but radiotherapy provides significant benefit with respect to reducing the local failure risk.
Major finding: The cumulative rates of ipsilateral local failure at 7 years were 6.7% and 0.9% in the observation and radiotherapy groups, respectively (HR, 0.11)
Data source: A prospective randomized trial involving 585 women.
Disclosures: This study was supported by grants from the National Institutes of Health. Dr. McCormick reported having no disclosures.
Vitiligo indicates effective melanoma treatment, predicts survival benefit
The development of vitiligo in melanoma patients on immunotherapy may predict improved survival, according to findings from a systematic review and meta-analysis.
In 137 studies reported between 1995 and 2013 and including 5,737 patients with stage III to IV melanoma who were treated with immunotherapy, the pooled cumulative incidence of vitiligo was 3.4%. In those with vitiligo for whom individual patient data were available, both progression-free and overall survival were significantly improved, compared with those without vitiligo after researchers adjusted for age and sex (hazard ratio, 0.51 and 0.25, respectively), Dr. Hansje-Eva Teulings of the University of Amsterdam and her colleagues reported online Jan. 19 in the Journal of Clinical Oncology.
Immune-related effects after melanoma immunotherapy have been linked to increased clinical efficacy. Vitiligo, which results from “strong antimelanoma immunity that also targets healthy melanocytes as a result of shared expression melanocyte differentiation antigens,” is no exception, but it was unclear whether data from individual studies showing tumor regression and improved survival in those with vitiligo could be extrapolated to all immunotherapy studies, the investigators explained (J. Clin. Oncol. 2015 Jan. 19 [doi:10.1200/JCO.2014.57.4756]).
The current findings highlight the significance of vitiligo as a clinical marker for effective antimelanoma immunity and for improved clinical outcome, they said, concluding that “more awareness of vitiligo induction in patients with melanoma by oncologists may contribute to better recognition of patients with effective antimelanoma immunity and may influence their treatment options and prognosis.”
The development of vitiligo in melanoma patients on immunotherapy may predict improved survival, according to findings from a systematic review and meta-analysis.
In 137 studies reported between 1995 and 2013 and including 5,737 patients with stage III to IV melanoma who were treated with immunotherapy, the pooled cumulative incidence of vitiligo was 3.4%. In those with vitiligo for whom individual patient data were available, both progression-free and overall survival were significantly improved, compared with those without vitiligo after researchers adjusted for age and sex (hazard ratio, 0.51 and 0.25, respectively), Dr. Hansje-Eva Teulings of the University of Amsterdam and her colleagues reported online Jan. 19 in the Journal of Clinical Oncology.
Immune-related effects after melanoma immunotherapy have been linked to increased clinical efficacy. Vitiligo, which results from “strong antimelanoma immunity that also targets healthy melanocytes as a result of shared expression melanocyte differentiation antigens,” is no exception, but it was unclear whether data from individual studies showing tumor regression and improved survival in those with vitiligo could be extrapolated to all immunotherapy studies, the investigators explained (J. Clin. Oncol. 2015 Jan. 19 [doi:10.1200/JCO.2014.57.4756]).
The current findings highlight the significance of vitiligo as a clinical marker for effective antimelanoma immunity and for improved clinical outcome, they said, concluding that “more awareness of vitiligo induction in patients with melanoma by oncologists may contribute to better recognition of patients with effective antimelanoma immunity and may influence their treatment options and prognosis.”
The development of vitiligo in melanoma patients on immunotherapy may predict improved survival, according to findings from a systematic review and meta-analysis.
In 137 studies reported between 1995 and 2013 and including 5,737 patients with stage III to IV melanoma who were treated with immunotherapy, the pooled cumulative incidence of vitiligo was 3.4%. In those with vitiligo for whom individual patient data were available, both progression-free and overall survival were significantly improved, compared with those without vitiligo after researchers adjusted for age and sex (hazard ratio, 0.51 and 0.25, respectively), Dr. Hansje-Eva Teulings of the University of Amsterdam and her colleagues reported online Jan. 19 in the Journal of Clinical Oncology.
Immune-related effects after melanoma immunotherapy have been linked to increased clinical efficacy. Vitiligo, which results from “strong antimelanoma immunity that also targets healthy melanocytes as a result of shared expression melanocyte differentiation antigens,” is no exception, but it was unclear whether data from individual studies showing tumor regression and improved survival in those with vitiligo could be extrapolated to all immunotherapy studies, the investigators explained (J. Clin. Oncol. 2015 Jan. 19 [doi:10.1200/JCO.2014.57.4756]).
The current findings highlight the significance of vitiligo as a clinical marker for effective antimelanoma immunity and for improved clinical outcome, they said, concluding that “more awareness of vitiligo induction in patients with melanoma by oncologists may contribute to better recognition of patients with effective antimelanoma immunity and may influence their treatment options and prognosis.”
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Vitiligo appears to serve as a clinical marker for effective antimelanoma immunity and improved clinical outcome.
Major finding: Progression-free and overall survival were improved in patients who developed vitiligo (HR, 0.51 and 0.25, respectively).
Data source: A systematic review and meta-analysis of 139 studies including 5,737 patients.
Disclosures: Dr. Teulings reported having no disclosures.
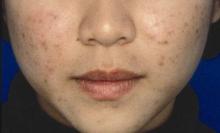
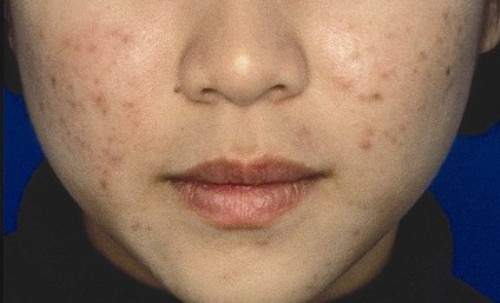
Combined OTC, prescription acne regimen satisfied young patients
ORLANDO – A three-component combined over-the-counter and prescription skin care regimen was safe, effective, well tolerated, and well liked by acne vulgaris patients aged 12 years and older in an open-label multicenter study.
Of 81 participants with a mean age of 19 years, mild or moderate facial acne vulgaris, and mean acne vulgaris duration of 4.4 years, 89.2% agreed or strongly agreed that they liked the regimen, 87.9% reported overall satisfaction, and 87.9% said they would recommend the regimen to others.
Nearly all participants (95.9%) said the regimen was easy to use, 73% reported improved skin texture, and 70.3% said the regimen met their needs, Dr. Michael H. Gold of Nashville, Tenn., reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
Treatment involved once-daily application of a prescription topical gel containing adapalene 1% and benzoyl peroxide 2.5%, as well as the use of two over-the-counter products designed for acne-prone skin: a foaming cleanser used twice daily and a moisturizer with broad spectrum SPF 30 sunscreen used once daily. Treatment continued for 8 weeks.
In addition to the subjective patient satisfaction questionnaire, subjects were assessed based on total inflammatory and noninflammatory lesion counts; photographic evaluation of skin shininess, texture, and presence of Propionibacterium acnes; cutaneous tolerability scores for stinging, burning, erythema, scaling, and dryness; and adverse events.
The therapeutic effect was evident in most patients at 2 weeks, with a reduction in the number of lesions. After 8 weeks, total inflammatory and noninflammatory lesion counts were significantly reduced, compared with baseline. Skin shininess and P. acnes also were significantly reduced by 8 weeks, Dr. Gold noted.
Most patients had no cutaneous irritation; the proportion of patients experiencing irritation was smaller than in prior phase II and III studies that evaluated benzoyl peroxide once-daily gel with or without moisturizer. Of 553 patients from those prior studies, 4% and 1% reported moderate and severe erythema, respectively, compared with 1% and 0% of the patients in the current study. In addition 3% and 1% of patients in earlier phase II and III studies, respectively, reported moderate or severe stinging/burning, compared with none of the patients in the current study.
A total of 18 adverse events were reported by 13 patients in the current study, and 17 events were considered to be related to the skin care regimen. None of the affected patients discontinued the regimen, and none of the patients reported serious adverse events.
Of note, 70% of patients agreed or strongly agreed that a sunscreen made for acne-prone skin was important to them, Dr. Gold said.
He and his colleagues concluded that a complete three-component, acne-specific regimen designed to clean, moisturize, medicate, and photoprotect is important for optimizing patient outcomes, and that the regimen used in this study was associated with good outcomes and high levels of patient satisfaction.
This study was funded by Galderma Laboratories.
ORLANDO – A three-component combined over-the-counter and prescription skin care regimen was safe, effective, well tolerated, and well liked by acne vulgaris patients aged 12 years and older in an open-label multicenter study.
Of 81 participants with a mean age of 19 years, mild or moderate facial acne vulgaris, and mean acne vulgaris duration of 4.4 years, 89.2% agreed or strongly agreed that they liked the regimen, 87.9% reported overall satisfaction, and 87.9% said they would recommend the regimen to others.
Nearly all participants (95.9%) said the regimen was easy to use, 73% reported improved skin texture, and 70.3% said the regimen met their needs, Dr. Michael H. Gold of Nashville, Tenn., reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
Treatment involved once-daily application of a prescription topical gel containing adapalene 1% and benzoyl peroxide 2.5%, as well as the use of two over-the-counter products designed for acne-prone skin: a foaming cleanser used twice daily and a moisturizer with broad spectrum SPF 30 sunscreen used once daily. Treatment continued for 8 weeks.
In addition to the subjective patient satisfaction questionnaire, subjects were assessed based on total inflammatory and noninflammatory lesion counts; photographic evaluation of skin shininess, texture, and presence of Propionibacterium acnes; cutaneous tolerability scores for stinging, burning, erythema, scaling, and dryness; and adverse events.
The therapeutic effect was evident in most patients at 2 weeks, with a reduction in the number of lesions. After 8 weeks, total inflammatory and noninflammatory lesion counts were significantly reduced, compared with baseline. Skin shininess and P. acnes also were significantly reduced by 8 weeks, Dr. Gold noted.
Most patients had no cutaneous irritation; the proportion of patients experiencing irritation was smaller than in prior phase II and III studies that evaluated benzoyl peroxide once-daily gel with or without moisturizer. Of 553 patients from those prior studies, 4% and 1% reported moderate and severe erythema, respectively, compared with 1% and 0% of the patients in the current study. In addition 3% and 1% of patients in earlier phase II and III studies, respectively, reported moderate or severe stinging/burning, compared with none of the patients in the current study.
A total of 18 adverse events were reported by 13 patients in the current study, and 17 events were considered to be related to the skin care regimen. None of the affected patients discontinued the regimen, and none of the patients reported serious adverse events.
Of note, 70% of patients agreed or strongly agreed that a sunscreen made for acne-prone skin was important to them, Dr. Gold said.
He and his colleagues concluded that a complete three-component, acne-specific regimen designed to clean, moisturize, medicate, and photoprotect is important for optimizing patient outcomes, and that the regimen used in this study was associated with good outcomes and high levels of patient satisfaction.
This study was funded by Galderma Laboratories.
ORLANDO – A three-component combined over-the-counter and prescription skin care regimen was safe, effective, well tolerated, and well liked by acne vulgaris patients aged 12 years and older in an open-label multicenter study.
Of 81 participants with a mean age of 19 years, mild or moderate facial acne vulgaris, and mean acne vulgaris duration of 4.4 years, 89.2% agreed or strongly agreed that they liked the regimen, 87.9% reported overall satisfaction, and 87.9% said they would recommend the regimen to others.
Nearly all participants (95.9%) said the regimen was easy to use, 73% reported improved skin texture, and 70.3% said the regimen met their needs, Dr. Michael H. Gold of Nashville, Tenn., reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
Treatment involved once-daily application of a prescription topical gel containing adapalene 1% and benzoyl peroxide 2.5%, as well as the use of two over-the-counter products designed for acne-prone skin: a foaming cleanser used twice daily and a moisturizer with broad spectrum SPF 30 sunscreen used once daily. Treatment continued for 8 weeks.
In addition to the subjective patient satisfaction questionnaire, subjects were assessed based on total inflammatory and noninflammatory lesion counts; photographic evaluation of skin shininess, texture, and presence of Propionibacterium acnes; cutaneous tolerability scores for stinging, burning, erythema, scaling, and dryness; and adverse events.
The therapeutic effect was evident in most patients at 2 weeks, with a reduction in the number of lesions. After 8 weeks, total inflammatory and noninflammatory lesion counts were significantly reduced, compared with baseline. Skin shininess and P. acnes also were significantly reduced by 8 weeks, Dr. Gold noted.
Most patients had no cutaneous irritation; the proportion of patients experiencing irritation was smaller than in prior phase II and III studies that evaluated benzoyl peroxide once-daily gel with or without moisturizer. Of 553 patients from those prior studies, 4% and 1% reported moderate and severe erythema, respectively, compared with 1% and 0% of the patients in the current study. In addition 3% and 1% of patients in earlier phase II and III studies, respectively, reported moderate or severe stinging/burning, compared with none of the patients in the current study.
A total of 18 adverse events were reported by 13 patients in the current study, and 17 events were considered to be related to the skin care regimen. None of the affected patients discontinued the regimen, and none of the patients reported serious adverse events.
Of note, 70% of patients agreed or strongly agreed that a sunscreen made for acne-prone skin was important to them, Dr. Gold said.
He and his colleagues concluded that a complete three-component, acne-specific regimen designed to clean, moisturize, medicate, and photoprotect is important for optimizing patient outcomes, and that the regimen used in this study was associated with good outcomes and high levels of patient satisfaction.
This study was funded by Galderma Laboratories.
AT THE ODAC CONFERENCE
Key clinical point: Children and young adults with acne vulgaris adhered to a three-part skin care regimen involving prescription and OTC products.
Major finding: 87.9% of patients reported overall satisfaction.
Data source: An open-label, multicenter study.
Disclosures: The study was funded by Galderma Laboratories.
OTC acne product equals benzoyl peroxide with clindamycin
ORLANDO – An over-the-counter 5.5% benzoyl peroxide preparation with lipohydroxy acid was a safe and effective alternative to prescription 5% benzoyl peroxide with 1% clindamycin in a 12-week randomized, double-blind, multicenter study of patients with mild to moderate acne vulgaris who also were treated with generic 0.025% tretinoin.
Statistically significant improvement on a variety of efficacy endpoints was seen at weeks 8 and 12 in 60 men and women aged 18-50 years who were randomized to either the OTC-based or prescription-based regimen, Susana Raab of L’Oreal Research & Innovation, Clark, N.J., reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
The mean reduction in total acne lesion count at week 8 was approximately 60% with the OTC regimen and 64% in the prescription regimen. At week 12, the mean reduction was about 72% in both groups. The mean improvement in global acne assessment was approximately 31% and 33% in the groups, respectively, at 8 weeks, and about 33% in both groups at 12 weeks.
The mean improvement in overall appearance was 37%-38% in both groups at 8 weeks, and was close to 50% in both groups at 12 weeks, Ms. Raab said.
All skin types and a range of ethnicities were represented in the study population. Subjects applied the OTC benzoyl peroxide with lipohydroxy acid or the prescription benzoyl peroxide and clindamycin twice daily for 12 weeks. The tretinoin was applied daily at night after the other products were dried and absorbed. Three blinded board-certified dermatologists assessed the subjects for tolerability, and lesion counts were performed on the entire face to assess for open and closed comedones, papules, and pustules.
The dermatologists also assessed for tone, smoothness, brightness, appearance of pores, global acne, overall skin appearance, and tolerability.
The degree of acne relapse was assessed during a 4-week regression phase at which time the treatment was discontinued.
Both treatments improved inflammatory and noninflammatory lesions to a similar degree within 2 weeks of use. Dryness and peeling were seen with both treatments, but resolved by week 12. Stinging, tingling, itching, and burning also were common with both treatments, and largely resolved in both groups by week 8, except for significant stinging in the OTC treatment group. No significant overall differences were seen between the groups with respect to tolerance parameters, Ms. Raab noted.
“When compared, both treatments were at parity in improvement of all efficacy attributes at all time points,” she and her colleagues concluded.
ORLANDO – An over-the-counter 5.5% benzoyl peroxide preparation with lipohydroxy acid was a safe and effective alternative to prescription 5% benzoyl peroxide with 1% clindamycin in a 12-week randomized, double-blind, multicenter study of patients with mild to moderate acne vulgaris who also were treated with generic 0.025% tretinoin.
Statistically significant improvement on a variety of efficacy endpoints was seen at weeks 8 and 12 in 60 men and women aged 18-50 years who were randomized to either the OTC-based or prescription-based regimen, Susana Raab of L’Oreal Research & Innovation, Clark, N.J., reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
The mean reduction in total acne lesion count at week 8 was approximately 60% with the OTC regimen and 64% in the prescription regimen. At week 12, the mean reduction was about 72% in both groups. The mean improvement in global acne assessment was approximately 31% and 33% in the groups, respectively, at 8 weeks, and about 33% in both groups at 12 weeks.
The mean improvement in overall appearance was 37%-38% in both groups at 8 weeks, and was close to 50% in both groups at 12 weeks, Ms. Raab said.
All skin types and a range of ethnicities were represented in the study population. Subjects applied the OTC benzoyl peroxide with lipohydroxy acid or the prescription benzoyl peroxide and clindamycin twice daily for 12 weeks. The tretinoin was applied daily at night after the other products were dried and absorbed. Three blinded board-certified dermatologists assessed the subjects for tolerability, and lesion counts were performed on the entire face to assess for open and closed comedones, papules, and pustules.
The dermatologists also assessed for tone, smoothness, brightness, appearance of pores, global acne, overall skin appearance, and tolerability.
The degree of acne relapse was assessed during a 4-week regression phase at which time the treatment was discontinued.
Both treatments improved inflammatory and noninflammatory lesions to a similar degree within 2 weeks of use. Dryness and peeling were seen with both treatments, but resolved by week 12. Stinging, tingling, itching, and burning also were common with both treatments, and largely resolved in both groups by week 8, except for significant stinging in the OTC treatment group. No significant overall differences were seen between the groups with respect to tolerance parameters, Ms. Raab noted.
“When compared, both treatments were at parity in improvement of all efficacy attributes at all time points,” she and her colleagues concluded.
ORLANDO – An over-the-counter 5.5% benzoyl peroxide preparation with lipohydroxy acid was a safe and effective alternative to prescription 5% benzoyl peroxide with 1% clindamycin in a 12-week randomized, double-blind, multicenter study of patients with mild to moderate acne vulgaris who also were treated with generic 0.025% tretinoin.
Statistically significant improvement on a variety of efficacy endpoints was seen at weeks 8 and 12 in 60 men and women aged 18-50 years who were randomized to either the OTC-based or prescription-based regimen, Susana Raab of L’Oreal Research & Innovation, Clark, N.J., reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
The mean reduction in total acne lesion count at week 8 was approximately 60% with the OTC regimen and 64% in the prescription regimen. At week 12, the mean reduction was about 72% in both groups. The mean improvement in global acne assessment was approximately 31% and 33% in the groups, respectively, at 8 weeks, and about 33% in both groups at 12 weeks.
The mean improvement in overall appearance was 37%-38% in both groups at 8 weeks, and was close to 50% in both groups at 12 weeks, Ms. Raab said.
All skin types and a range of ethnicities were represented in the study population. Subjects applied the OTC benzoyl peroxide with lipohydroxy acid or the prescription benzoyl peroxide and clindamycin twice daily for 12 weeks. The tretinoin was applied daily at night after the other products were dried and absorbed. Three blinded board-certified dermatologists assessed the subjects for tolerability, and lesion counts were performed on the entire face to assess for open and closed comedones, papules, and pustules.
The dermatologists also assessed for tone, smoothness, brightness, appearance of pores, global acne, overall skin appearance, and tolerability.
The degree of acne relapse was assessed during a 4-week regression phase at which time the treatment was discontinued.
Both treatments improved inflammatory and noninflammatory lesions to a similar degree within 2 weeks of use. Dryness and peeling were seen with both treatments, but resolved by week 12. Stinging, tingling, itching, and burning also were common with both treatments, and largely resolved in both groups by week 8, except for significant stinging in the OTC treatment group. No significant overall differences were seen between the groups with respect to tolerance parameters, Ms. Raab noted.
“When compared, both treatments were at parity in improvement of all efficacy attributes at all time points,” she and her colleagues concluded.
Key clinical point: An OTC benzoyl peroxide–based product compared well with a prescription benzoyl peroxide/clindamycin product for acne vulgaris.
Major finding: The mean improvement in overall appearance was 37%-38% in both treatment groups at 8 weeks and was close to 50% in both groups at 12 weeks.
Data source: A randomized, double-blind, multicenter study of 60 adults.
Disclosures: The lead author is employed by L’Oreal Research & Innovation, manufacturer of the OTC product.
MRI shows ongoing inflammation despite clinical remission in early RA
Two years of either triple therapy or treatment with tumor necrosis factor plus methotrexate failed to eliminate joint inflammation on MRI in a subcohort of patients with early rheumatoid arthritis from the randomized, double-blind Treatment of Early Aggressive Rheumatoid Arthritis (TEAR) trial.
In 118 patients with a mean age of 51 years, short disease duration, and severe disease at TEAR trial entry – 92% of whom were seropositive – only 29 had wrist pain, tenderness, or swelling at 2-year follow-up. However, all 118 patients had MRI evidence of residual joint inflammation after 2 years, and 78% had evidence of osteitis, Dr. Veena K. Ranganath of the University of California, Los Angeles, and her colleagues reported (Arthritis Care Res. 2015 Jan. 7 [doi:10.1002/acr.22541]).
Inflammation remained despite significant improvement of disease activity measures at the time of the MRI, compared with baseline (for example, 28-joint disease activity score using erythrocyte sedimentation rate [DAS28-ESR] decreased from 5.8 to 2.9). Total MRI inflammation scores were significantly lower in patients who met 2011 American College of Rheumatology (ACR)/European League Against Rheumatism (EULAR) Boolean remission criteria and remission by Chronic Disease Activity Index (CDAI), but not in those with DAS28-ESR remission, they noted.
The findings demonstrate that total MRI inflammatory scores are “best differentiated by the most stringent clinical remission criteria” – CDAI and 2011 ACR/EULAR Boolean Criteria, as opposed to DAS28-ESR (with a 2.6 cutpoint). Further, no differences were seen in damage or MRI inflammatory scores based on treatment regimen, which supports methotrexate-first recommendations for the TEAR trial, they said, noting that the long-term prognostic implications of the study findings are unclear because of short-follow-up, and that it remains unclear whether attainment of clinical remission warrants a drug holiday or cessation of RA treatment.
Thus, it is “ill-advised to discontinue therapy until future studies suggest otherwise,” they concluded, adding that this is particularly true given that prior published data suggest a link between osteitis – which occurred at a high rate in this study despite clinical remission – and future radiographic progression.
The TEAR trial was supported by Amgen. The current research was supported by a National Institutes of Health/National Center for Advancing Translational Science UCLA CTSI grant, and individual authors were supported by ACR/REF grants, a National Institutes of Health award, the Margaret J. Miller Endowed Professor of Research Chair, and the Agency for Healthcare Research and Quality.
Two years of either triple therapy or treatment with tumor necrosis factor plus methotrexate failed to eliminate joint inflammation on MRI in a subcohort of patients with early rheumatoid arthritis from the randomized, double-blind Treatment of Early Aggressive Rheumatoid Arthritis (TEAR) trial.
In 118 patients with a mean age of 51 years, short disease duration, and severe disease at TEAR trial entry – 92% of whom were seropositive – only 29 had wrist pain, tenderness, or swelling at 2-year follow-up. However, all 118 patients had MRI evidence of residual joint inflammation after 2 years, and 78% had evidence of osteitis, Dr. Veena K. Ranganath of the University of California, Los Angeles, and her colleagues reported (Arthritis Care Res. 2015 Jan. 7 [doi:10.1002/acr.22541]).
Inflammation remained despite significant improvement of disease activity measures at the time of the MRI, compared with baseline (for example, 28-joint disease activity score using erythrocyte sedimentation rate [DAS28-ESR] decreased from 5.8 to 2.9). Total MRI inflammation scores were significantly lower in patients who met 2011 American College of Rheumatology (ACR)/European League Against Rheumatism (EULAR) Boolean remission criteria and remission by Chronic Disease Activity Index (CDAI), but not in those with DAS28-ESR remission, they noted.
The findings demonstrate that total MRI inflammatory scores are “best differentiated by the most stringent clinical remission criteria” – CDAI and 2011 ACR/EULAR Boolean Criteria, as opposed to DAS28-ESR (with a 2.6 cutpoint). Further, no differences were seen in damage or MRI inflammatory scores based on treatment regimen, which supports methotrexate-first recommendations for the TEAR trial, they said, noting that the long-term prognostic implications of the study findings are unclear because of short-follow-up, and that it remains unclear whether attainment of clinical remission warrants a drug holiday or cessation of RA treatment.
Thus, it is “ill-advised to discontinue therapy until future studies suggest otherwise,” they concluded, adding that this is particularly true given that prior published data suggest a link between osteitis – which occurred at a high rate in this study despite clinical remission – and future radiographic progression.
The TEAR trial was supported by Amgen. The current research was supported by a National Institutes of Health/National Center for Advancing Translational Science UCLA CTSI grant, and individual authors were supported by ACR/REF grants, a National Institutes of Health award, the Margaret J. Miller Endowed Professor of Research Chair, and the Agency for Healthcare Research and Quality.
Two years of either triple therapy or treatment with tumor necrosis factor plus methotrexate failed to eliminate joint inflammation on MRI in a subcohort of patients with early rheumatoid arthritis from the randomized, double-blind Treatment of Early Aggressive Rheumatoid Arthritis (TEAR) trial.
In 118 patients with a mean age of 51 years, short disease duration, and severe disease at TEAR trial entry – 92% of whom were seropositive – only 29 had wrist pain, tenderness, or swelling at 2-year follow-up. However, all 118 patients had MRI evidence of residual joint inflammation after 2 years, and 78% had evidence of osteitis, Dr. Veena K. Ranganath of the University of California, Los Angeles, and her colleagues reported (Arthritis Care Res. 2015 Jan. 7 [doi:10.1002/acr.22541]).
Inflammation remained despite significant improvement of disease activity measures at the time of the MRI, compared with baseline (for example, 28-joint disease activity score using erythrocyte sedimentation rate [DAS28-ESR] decreased from 5.8 to 2.9). Total MRI inflammation scores were significantly lower in patients who met 2011 American College of Rheumatology (ACR)/European League Against Rheumatism (EULAR) Boolean remission criteria and remission by Chronic Disease Activity Index (CDAI), but not in those with DAS28-ESR remission, they noted.
The findings demonstrate that total MRI inflammatory scores are “best differentiated by the most stringent clinical remission criteria” – CDAI and 2011 ACR/EULAR Boolean Criteria, as opposed to DAS28-ESR (with a 2.6 cutpoint). Further, no differences were seen in damage or MRI inflammatory scores based on treatment regimen, which supports methotrexate-first recommendations for the TEAR trial, they said, noting that the long-term prognostic implications of the study findings are unclear because of short-follow-up, and that it remains unclear whether attainment of clinical remission warrants a drug holiday or cessation of RA treatment.
Thus, it is “ill-advised to discontinue therapy until future studies suggest otherwise,” they concluded, adding that this is particularly true given that prior published data suggest a link between osteitis – which occurred at a high rate in this study despite clinical remission – and future radiographic progression.
The TEAR trial was supported by Amgen. The current research was supported by a National Institutes of Health/National Center for Advancing Translational Science UCLA CTSI grant, and individual authors were supported by ACR/REF grants, a National Institutes of Health award, the Margaret J. Miller Endowed Professor of Research Chair, and the Agency for Healthcare Research and Quality.
FROM ARTHRITIS CARE & RESEARCH
Key clinical point: Until data suggest otherwise, treatment should continue despite clinical remission in early RA patients.
Major finding: Only 29 of 118 patients had symptoms, but all 118 had MRI evidence of inflammation.
Data source: A subcohort of 118 patients from the randomized, double-blind TEAR trial .
Disclosures: The TEAR trial was supported by Amgen. The current research was supported by a National Institutes of Health/National Center for Advancing Translational Science UCLA CTSI grant, and individual authors were supported by ACR/REF grants, a National Institutes of Health award, the Margaret J. Miller Endowed Professor of Research Chair, and the Agency for Healthcare Research and Quality.
Case series: Ustekinumab for psoriasis helps skin, hurts joints
Ustekinumab treatment was associated with new-onset or worsening psoriatic arthritis in a series of seven patients with psoriasis.
The findings, which support previous observations that patients treated with ustekinumab (Stelara) “sometimes have discordant responses of their skin and joint disease,” underscore the need for regularly asking patients about joint symptoms, and for referral to a rheumatologist for suspected psoriatic arthritis, Ben B. Jones of the University of Utah, Salt Lake City, and his colleagues reported (Br. J. Dermatol. 2014 Dec. 30 [doi:10.1111/bjd.13645]).
All seven patients in the case series had well-controlled psoriasis on ustekinumab. Five had new-onset psoriatic arthritis, and two had worsening psoriatic arthritis on treatment. The patients had phenotypic similarities; most were women over age 49 years, and all five of those with new-onset disease were women. Also, five of the seven patients had exposure to tumor necrosis factor inhibitors prior to switching to ustekinumab.
Three other case series have reported similar findings of marked improvement in cutaneous symptoms with worsening of joint symptoms among patients treated with ustekinumab, the investigators noted, concluding that the findings – which may reflect a lack of efficacy at the administered doses or a need for more frequent dosing – may support arguments that psoriatic arthritis and psoriasis involve distinct inflammatory pathways.
“It is also possible that ustekinumab may trigger or unmask inflammation in the joints of patients with psoriatic arthritis,” they wrote, concluding that “larger epidemiologic studies comparing patients with discordant and concordant cutaneous and articular responses to ustekinumab may better define patients at risk for psoriatic arthritis worsening with ustekinumab.”
Two of the five authors have reported serving as a consultant or advisory board member, receiving payment for lectures, and/or serving as an investigator for Janssen, which markets ustekinumab, as well as other manufacturers of biologics. The other authors reported having no conflicts of interest.
Ustekinumab treatment was associated with new-onset or worsening psoriatic arthritis in a series of seven patients with psoriasis.
The findings, which support previous observations that patients treated with ustekinumab (Stelara) “sometimes have discordant responses of their skin and joint disease,” underscore the need for regularly asking patients about joint symptoms, and for referral to a rheumatologist for suspected psoriatic arthritis, Ben B. Jones of the University of Utah, Salt Lake City, and his colleagues reported (Br. J. Dermatol. 2014 Dec. 30 [doi:10.1111/bjd.13645]).
All seven patients in the case series had well-controlled psoriasis on ustekinumab. Five had new-onset psoriatic arthritis, and two had worsening psoriatic arthritis on treatment. The patients had phenotypic similarities; most were women over age 49 years, and all five of those with new-onset disease were women. Also, five of the seven patients had exposure to tumor necrosis factor inhibitors prior to switching to ustekinumab.
Three other case series have reported similar findings of marked improvement in cutaneous symptoms with worsening of joint symptoms among patients treated with ustekinumab, the investigators noted, concluding that the findings – which may reflect a lack of efficacy at the administered doses or a need for more frequent dosing – may support arguments that psoriatic arthritis and psoriasis involve distinct inflammatory pathways.
“It is also possible that ustekinumab may trigger or unmask inflammation in the joints of patients with psoriatic arthritis,” they wrote, concluding that “larger epidemiologic studies comparing patients with discordant and concordant cutaneous and articular responses to ustekinumab may better define patients at risk for psoriatic arthritis worsening with ustekinumab.”
Two of the five authors have reported serving as a consultant or advisory board member, receiving payment for lectures, and/or serving as an investigator for Janssen, which markets ustekinumab, as well as other manufacturers of biologics. The other authors reported having no conflicts of interest.
Ustekinumab treatment was associated with new-onset or worsening psoriatic arthritis in a series of seven patients with psoriasis.
The findings, which support previous observations that patients treated with ustekinumab (Stelara) “sometimes have discordant responses of their skin and joint disease,” underscore the need for regularly asking patients about joint symptoms, and for referral to a rheumatologist for suspected psoriatic arthritis, Ben B. Jones of the University of Utah, Salt Lake City, and his colleagues reported (Br. J. Dermatol. 2014 Dec. 30 [doi:10.1111/bjd.13645]).
All seven patients in the case series had well-controlled psoriasis on ustekinumab. Five had new-onset psoriatic arthritis, and two had worsening psoriatic arthritis on treatment. The patients had phenotypic similarities; most were women over age 49 years, and all five of those with new-onset disease were women. Also, five of the seven patients had exposure to tumor necrosis factor inhibitors prior to switching to ustekinumab.
Three other case series have reported similar findings of marked improvement in cutaneous symptoms with worsening of joint symptoms among patients treated with ustekinumab, the investigators noted, concluding that the findings – which may reflect a lack of efficacy at the administered doses or a need for more frequent dosing – may support arguments that psoriatic arthritis and psoriasis involve distinct inflammatory pathways.
“It is also possible that ustekinumab may trigger or unmask inflammation in the joints of patients with psoriatic arthritis,” they wrote, concluding that “larger epidemiologic studies comparing patients with discordant and concordant cutaneous and articular responses to ustekinumab may better define patients at risk for psoriatic arthritis worsening with ustekinumab.”
Two of the five authors have reported serving as a consultant or advisory board member, receiving payment for lectures, and/or serving as an investigator for Janssen, which markets ustekinumab, as well as other manufacturers of biologics. The other authors reported having no conflicts of interest.
Key clinical point: Increasing evidence suggests that ustekinumab is associated with discordant joint and skin responses.
Major finding: Five of seven psoriasis patients treated with ustekinumab experienced new-onset psoriatic arthritis, and two had worsening psoriatic arthritis.
Data source: A series of seven cases.
Disclosures: Two of the five authors have reported serving as a consultant or advisory board member, receiving payment for lectures, and/or serving as an investigator for Janssen, which markets ustekinumab, as well as other manufacturers of biologics. The other authors reported having no conflicts of interest.
Framingham score underestimates CVD risk in psoriatic arthritis patients
Most newly diagnosed psoriatic arthritis patients have an increased risk for cardiovascular disease that is markedly underestimated by the Framingham Risk Score, according to findings from a retrospective, population-based, cohort study.
The mean Framingham Risk Score in 126 patients with psoriatic arthritis who were aged 30 years or older and who had no prior cardiovascular disease (CVD) history, was 9.7% during the first 10 years of follow-up. However, the 10-year cumulative incidence of CVD events was nearly double that at 17% (standardized incidence ratio, 1.80), Dr. Floranne C. Ernste of the Mayo Clinic, Rochester, Minn., and her colleagues reported (Arthritis Care Res. 2015 Jan. 7 [doi:10.1002/acr.22536]).
Age-based analysis showed that the CVD risk in these patients was consistently twice as high as predicted by the FRS beginning after age 40 years, the authors noted.
The findings underscore the importance of CVD risk assessment in patients with psoriatic arthritis but suggest that the Framingham Risk Score may not be applicable in such patients, the investigators said, adding that “This study serves to illustrate the important need for further research to focus on the development of CVD risk assessment tools specific to psoriatic arthritis patients.”
The findings also suggest that aggressive therapy may be warranted early in the course of psoriatic arthritis to “attenuate the long-term burden of CVD,” they said.
This study was supported by the Rochester Epidemiology Project and by Amgen. Dr. Ernste reported having no disclosures.
Most newly diagnosed psoriatic arthritis patients have an increased risk for cardiovascular disease that is markedly underestimated by the Framingham Risk Score, according to findings from a retrospective, population-based, cohort study.
The mean Framingham Risk Score in 126 patients with psoriatic arthritis who were aged 30 years or older and who had no prior cardiovascular disease (CVD) history, was 9.7% during the first 10 years of follow-up. However, the 10-year cumulative incidence of CVD events was nearly double that at 17% (standardized incidence ratio, 1.80), Dr. Floranne C. Ernste of the Mayo Clinic, Rochester, Minn., and her colleagues reported (Arthritis Care Res. 2015 Jan. 7 [doi:10.1002/acr.22536]).
Age-based analysis showed that the CVD risk in these patients was consistently twice as high as predicted by the FRS beginning after age 40 years, the authors noted.
The findings underscore the importance of CVD risk assessment in patients with psoriatic arthritis but suggest that the Framingham Risk Score may not be applicable in such patients, the investigators said, adding that “This study serves to illustrate the important need for further research to focus on the development of CVD risk assessment tools specific to psoriatic arthritis patients.”
The findings also suggest that aggressive therapy may be warranted early in the course of psoriatic arthritis to “attenuate the long-term burden of CVD,” they said.
This study was supported by the Rochester Epidemiology Project and by Amgen. Dr. Ernste reported having no disclosures.
Most newly diagnosed psoriatic arthritis patients have an increased risk for cardiovascular disease that is markedly underestimated by the Framingham Risk Score, according to findings from a retrospective, population-based, cohort study.
The mean Framingham Risk Score in 126 patients with psoriatic arthritis who were aged 30 years or older and who had no prior cardiovascular disease (CVD) history, was 9.7% during the first 10 years of follow-up. However, the 10-year cumulative incidence of CVD events was nearly double that at 17% (standardized incidence ratio, 1.80), Dr. Floranne C. Ernste of the Mayo Clinic, Rochester, Minn., and her colleagues reported (Arthritis Care Res. 2015 Jan. 7 [doi:10.1002/acr.22536]).
Age-based analysis showed that the CVD risk in these patients was consistently twice as high as predicted by the FRS beginning after age 40 years, the authors noted.
The findings underscore the importance of CVD risk assessment in patients with psoriatic arthritis but suggest that the Framingham Risk Score may not be applicable in such patients, the investigators said, adding that “This study serves to illustrate the important need for further research to focus on the development of CVD risk assessment tools specific to psoriatic arthritis patients.”
The findings also suggest that aggressive therapy may be warranted early in the course of psoriatic arthritis to “attenuate the long-term burden of CVD,” they said.
This study was supported by the Rochester Epidemiology Project and by Amgen. Dr. Ernste reported having no disclosures.
FROM ARTHRITIS CARE & RESEARCH
Key clinical point: The Framingham Risk Score may not be applicable for estimating CVD risk in psoriatic arthritis patients.
Major finding: The 10-year cumulative CVD incidence rate was 17%, compared with 9.7% predicted by Framingham Risk Score.
Data source: A retrospective, population-based, cohort study of 126 patients.
Disclosures: This study was supported by the Rochester Epidemiology Project and by Amgen. Dr. Ernste reported having no disclosures.
CDC: Flu Remains Widespread; Antivirals Underutilized
The approximate midpoint of the 2014-2015 flu season has been reached, disease remains widespread, and antiviral flu medications remain underutilized, according to the Centers for Disease Control and Prevention.
As predicted, this season is proving particularly severe due to the predominance of antigenically drifted H3N2 virus strains, and the CDC is urging clinicians to maintain a high index of suspicion for flu and to prescribe antivirals earlier and more aggressively for patients presenting with flulike illness – especially young children, those over age 65 years, and those with underlying conditions, even if their symptoms are mild, CDC Director Thomas R. Frieden said during a press briefing.
A health advisory on the topic was issued Jan. 9, he noted.
“The CDC has recommended the use of antiviral drugs as an adjunct to vaccination. They are the only medicines that can specifically treat influenza illness, and in the context of an H3N2 predominant season with a less effective vaccine, treatment with antiflu drugs is even more important than usual,” he said, referring to the two neuraminidase inhibitors currently approved for treating influenza: oseltamivir and zanamivir.
About two-thirds of the H3N2 viruses analyzed this season are different from the H3N2 virus included in this year’s vaccine, which is why the vaccine is expected to have reduced effectiveness, he explained.
This makes early antiviral treatment all the more important, he added, noting that treatment within 2 days of symptom onset is optimal, but later treatment can also offer benefit.
“CDC scientists have looked very carefully at the use of influenza drugs in the clinical setting, and the conclusion is clear: They work, but they aren’t being used nearly enough. They can reduce symptoms, shorten duration of illness, and prevent serious complications,” he said.
Continue for early treatments >>
Early treatment may help patients avoid hospitalization and could be life saving, he added.
Many patients are unaware that effective prescription treatments exist, and many doctors are not using the treatments as recommended. In one study, fewer than one in five eligible high-risk patients received treatment.
“So we’re expanding our efforts to reach clinicians about reminders about the importance of these drugs,” he said, noting that if high-risk patients with underlying conditions such as asthma, sickle cell disease, renal disease, or diabetes were treated, “tens of thousands of hospitalizations, and thousands of deaths could potentially be prevented.”
Dr. Frieden suggested that clinicians consider phone triage lines for high-risk patients to discuss symptoms over the phone and to “facilitate early initiation of treatment.”
An antiviral prescription can be provided without testing and before an office visit, he added.
The typical flu season lasts about 13 weeks on average, and the season began about 7 weeks ago. The continued high rate of H3N2 disease has had the greatest impact on older adults; that is typical of such seasons, which often involve a higher rate of hospitalizations and deaths.
In fact, hospitalization rates in the over 65 age group are rising sharply, Dr. Frieden said.
Last week the rate was 52 per 100,000 population, and this week the rate was 92 per 100,000 population. The cumulative rate 2 years ago when H3N2 viruses last predominated was 183 per 100,000, he noted.
“We wouldn’t be surprised to see something very similar this year,” he said.
Young children are also severely affected; thus far in the season there have been 26 reported pediatric flu-related deaths.
However, there are early signs that infection rates are declining in areas where the season started earlier, such as the Southeast, and the number of patients presenting to a doctor with flulike symptoms has also declined slightly, but it is too soon to tell whether disease activity has peaked, he said, stressing that “we still have several weeks of flu activity ahead.”
It’s not too late to get vaccinated, and despite the reduced efficacy of this year’s vaccine against the circulating H3N2 strains, vaccination may still offer some benefit.
“There are other strains out there as well,” he said, noting that a proportion of cases from other strains, such as influenza B, tend to occur late in the season, and this year’s vaccine offers a good match for influenza B viruses.
Pneumococcal vaccination is also important, particularly for older adults and those at high risk , he said.
Last September the CDC announced a new recommendation that all adults over age 65 years should get two different pneumococcal vaccines. Such vaccination may help prevent flu-related pneumonia.
Patients with the flu or flulike illness should be advised to cover their cough and to stay home from work or school to protect those who are most vulnerable to the flu, he said.
The approximate midpoint of the 2014-2015 flu season has been reached, disease remains widespread, and antiviral flu medications remain underutilized, according to the Centers for Disease Control and Prevention.
As predicted, this season is proving particularly severe due to the predominance of antigenically drifted H3N2 virus strains, and the CDC is urging clinicians to maintain a high index of suspicion for flu and to prescribe antivirals earlier and more aggressively for patients presenting with flulike illness – especially young children, those over age 65 years, and those with underlying conditions, even if their symptoms are mild, CDC Director Thomas R. Frieden said during a press briefing.
A health advisory on the topic was issued Jan. 9, he noted.
“The CDC has recommended the use of antiviral drugs as an adjunct to vaccination. They are the only medicines that can specifically treat influenza illness, and in the context of an H3N2 predominant season with a less effective vaccine, treatment with antiflu drugs is even more important than usual,” he said, referring to the two neuraminidase inhibitors currently approved for treating influenza: oseltamivir and zanamivir.
About two-thirds of the H3N2 viruses analyzed this season are different from the H3N2 virus included in this year’s vaccine, which is why the vaccine is expected to have reduced effectiveness, he explained.
This makes early antiviral treatment all the more important, he added, noting that treatment within 2 days of symptom onset is optimal, but later treatment can also offer benefit.
“CDC scientists have looked very carefully at the use of influenza drugs in the clinical setting, and the conclusion is clear: They work, but they aren’t being used nearly enough. They can reduce symptoms, shorten duration of illness, and prevent serious complications,” he said.
Continue for early treatments >>
Early treatment may help patients avoid hospitalization and could be life saving, he added.
Many patients are unaware that effective prescription treatments exist, and many doctors are not using the treatments as recommended. In one study, fewer than one in five eligible high-risk patients received treatment.
“So we’re expanding our efforts to reach clinicians about reminders about the importance of these drugs,” he said, noting that if high-risk patients with underlying conditions such as asthma, sickle cell disease, renal disease, or diabetes were treated, “tens of thousands of hospitalizations, and thousands of deaths could potentially be prevented.”
Dr. Frieden suggested that clinicians consider phone triage lines for high-risk patients to discuss symptoms over the phone and to “facilitate early initiation of treatment.”
An antiviral prescription can be provided without testing and before an office visit, he added.
The typical flu season lasts about 13 weeks on average, and the season began about 7 weeks ago. The continued high rate of H3N2 disease has had the greatest impact on older adults; that is typical of such seasons, which often involve a higher rate of hospitalizations and deaths.
In fact, hospitalization rates in the over 65 age group are rising sharply, Dr. Frieden said.
Last week the rate was 52 per 100,000 population, and this week the rate was 92 per 100,000 population. The cumulative rate 2 years ago when H3N2 viruses last predominated was 183 per 100,000, he noted.
“We wouldn’t be surprised to see something very similar this year,” he said.
Young children are also severely affected; thus far in the season there have been 26 reported pediatric flu-related deaths.
However, there are early signs that infection rates are declining in areas where the season started earlier, such as the Southeast, and the number of patients presenting to a doctor with flulike symptoms has also declined slightly, but it is too soon to tell whether disease activity has peaked, he said, stressing that “we still have several weeks of flu activity ahead.”
It’s not too late to get vaccinated, and despite the reduced efficacy of this year’s vaccine against the circulating H3N2 strains, vaccination may still offer some benefit.
“There are other strains out there as well,” he said, noting that a proportion of cases from other strains, such as influenza B, tend to occur late in the season, and this year’s vaccine offers a good match for influenza B viruses.
Pneumococcal vaccination is also important, particularly for older adults and those at high risk , he said.
Last September the CDC announced a new recommendation that all adults over age 65 years should get two different pneumococcal vaccines. Such vaccination may help prevent flu-related pneumonia.
Patients with the flu or flulike illness should be advised to cover their cough and to stay home from work or school to protect those who are most vulnerable to the flu, he said.
The approximate midpoint of the 2014-2015 flu season has been reached, disease remains widespread, and antiviral flu medications remain underutilized, according to the Centers for Disease Control and Prevention.
As predicted, this season is proving particularly severe due to the predominance of antigenically drifted H3N2 virus strains, and the CDC is urging clinicians to maintain a high index of suspicion for flu and to prescribe antivirals earlier and more aggressively for patients presenting with flulike illness – especially young children, those over age 65 years, and those with underlying conditions, even if their symptoms are mild, CDC Director Thomas R. Frieden said during a press briefing.
A health advisory on the topic was issued Jan. 9, he noted.
“The CDC has recommended the use of antiviral drugs as an adjunct to vaccination. They are the only medicines that can specifically treat influenza illness, and in the context of an H3N2 predominant season with a less effective vaccine, treatment with antiflu drugs is even more important than usual,” he said, referring to the two neuraminidase inhibitors currently approved for treating influenza: oseltamivir and zanamivir.
About two-thirds of the H3N2 viruses analyzed this season are different from the H3N2 virus included in this year’s vaccine, which is why the vaccine is expected to have reduced effectiveness, he explained.
This makes early antiviral treatment all the more important, he added, noting that treatment within 2 days of symptom onset is optimal, but later treatment can also offer benefit.
“CDC scientists have looked very carefully at the use of influenza drugs in the clinical setting, and the conclusion is clear: They work, but they aren’t being used nearly enough. They can reduce symptoms, shorten duration of illness, and prevent serious complications,” he said.
Continue for early treatments >>
Early treatment may help patients avoid hospitalization and could be life saving, he added.
Many patients are unaware that effective prescription treatments exist, and many doctors are not using the treatments as recommended. In one study, fewer than one in five eligible high-risk patients received treatment.
“So we’re expanding our efforts to reach clinicians about reminders about the importance of these drugs,” he said, noting that if high-risk patients with underlying conditions such as asthma, sickle cell disease, renal disease, or diabetes were treated, “tens of thousands of hospitalizations, and thousands of deaths could potentially be prevented.”
Dr. Frieden suggested that clinicians consider phone triage lines for high-risk patients to discuss symptoms over the phone and to “facilitate early initiation of treatment.”
An antiviral prescription can be provided without testing and before an office visit, he added.
The typical flu season lasts about 13 weeks on average, and the season began about 7 weeks ago. The continued high rate of H3N2 disease has had the greatest impact on older adults; that is typical of such seasons, which often involve a higher rate of hospitalizations and deaths.
In fact, hospitalization rates in the over 65 age group are rising sharply, Dr. Frieden said.
Last week the rate was 52 per 100,000 population, and this week the rate was 92 per 100,000 population. The cumulative rate 2 years ago when H3N2 viruses last predominated was 183 per 100,000, he noted.
“We wouldn’t be surprised to see something very similar this year,” he said.
Young children are also severely affected; thus far in the season there have been 26 reported pediatric flu-related deaths.
However, there are early signs that infection rates are declining in areas where the season started earlier, such as the Southeast, and the number of patients presenting to a doctor with flulike symptoms has also declined slightly, but it is too soon to tell whether disease activity has peaked, he said, stressing that “we still have several weeks of flu activity ahead.”
It’s not too late to get vaccinated, and despite the reduced efficacy of this year’s vaccine against the circulating H3N2 strains, vaccination may still offer some benefit.
“There are other strains out there as well,” he said, noting that a proportion of cases from other strains, such as influenza B, tend to occur late in the season, and this year’s vaccine offers a good match for influenza B viruses.
Pneumococcal vaccination is also important, particularly for older adults and those at high risk , he said.
Last September the CDC announced a new recommendation that all adults over age 65 years should get two different pneumococcal vaccines. Such vaccination may help prevent flu-related pneumonia.
Patients with the flu or flulike illness should be advised to cover their cough and to stay home from work or school to protect those who are most vulnerable to the flu, he said.
Analysis: Push for Expanded Hepatitis C Screening Appears Premature
The recent advent of new treatments for hepatitis C prompted organizations including the Centers for Disease Control and Prevention, the U.S. Preventive Services Task Force, and the World Health Organization to recommend expanded hepatitis C screening, but such screening may be premature, according to a subject analysis.
Too much uncertainty exists regarding the validity of surrogate markers for treatment efficacy that were used in trials, and evidence regarding clinical outcomes and screening strategies is lacking, according to Dr. Ronald L. Koretz of the University of California, Los Angeles, and his colleagues, who evaluated the current understanding of the incidence and natural course of hepatitis C infection, treatment efficacy, and potential harms of treatment for their analysis.
The best available data suggest that 80%-85% of patients with chronic hepatitis C will die of nonhepatic causes; thus screening could lead to unnecessary treatment. This is important, as safety data for newer drugs are limited, and the existing data suggest a small but concerning rate of serious adverse events associated with the use of some treatments and treatment regimens; the risk-benefit profile of treatment cannot be adequately evaluated because of the lack of data regarding treatment benefits, the investigators reported online Jan. 13 in the British Medical Journal ([doi:10.10036/bmj.g7809]).
Clinical trials to determine the outcomes of treatment in screen-detected patients, as well as the long-term hazards of treatment, are needed, they said, noting that currently available trials included small numbers of patients and/or only short-term follow-up. Until data from such trials are available, physicians should not be pressured to enforce recommended screening strategies “out of enthusiasm for new treatments that have not yet been shown to cause long-term clinical improvement,” they concluded.
Dr. Koretz is a member of the editorial board of the Cochrane Hepato-Biliary Group. The authors reported having no other financial conflicts.
The authors question the merits of hepatitis C (HCV) screening despite the endorsement of the Centers for Disease Control and Prevention, U.S. Preventive Services Task Force, and World Health Organization. They suggest HCV does not lead to sufficient mortality, treatment may cause harm, sustained virologic response (SVR) does not represent a ‘cure’ and question if treating HCV truly reduces long term morbidity/mortality. While it is true the majority of patients with HCV will not develop cirrhosis or death attributable to their liver disease, the authors downplayed the morbidity and mortality related to HCV and the significant cost to the health care system as well (Ann. Intern. Med. 2012;156:271; Hepatology 2013;57:2164]. The authors also downplayed the importance of sustained viral response (SVR), suggesting that it does not represent a cure, yet neglect to mention several studies demonstrating late recurrence in only 1%-3% of patients who have achieved SVR. Furthermore, the authors describe the “harms of treatment,” yet they largely reference therapies that are no longer used and make minimal comment on the clear safety and efficacy of the current interferon-free regimens. Additionally, they report that while clearing HCV may reduce the risk of decompensated cirrhosis and hepatocellular carcinoma, it does not completely eliminate these risks and therefore treatment may not be of value. This brings us back to the motivation of the CDC, USPSTF, and WHO to recommend screening for HCV – identifying asymptomatic infection to allow for administration of safe, effective antiviral therapies before the development of cirrhosis and all of its complications.
Dr. Sean Koppe is director of hepatology, University of Illinois Hospital & Health Sciences System. He has no conflicts of interest.
The authors question the merits of hepatitis C (HCV) screening despite the endorsement of the Centers for Disease Control and Prevention, U.S. Preventive Services Task Force, and World Health Organization. They suggest HCV does not lead to sufficient mortality, treatment may cause harm, sustained virologic response (SVR) does not represent a ‘cure’ and question if treating HCV truly reduces long term morbidity/mortality. While it is true the majority of patients with HCV will not develop cirrhosis or death attributable to their liver disease, the authors downplayed the morbidity and mortality related to HCV and the significant cost to the health care system as well (Ann. Intern. Med. 2012;156:271; Hepatology 2013;57:2164]. The authors also downplayed the importance of sustained viral response (SVR), suggesting that it does not represent a cure, yet neglect to mention several studies demonstrating late recurrence in only 1%-3% of patients who have achieved SVR. Furthermore, the authors describe the “harms of treatment,” yet they largely reference therapies that are no longer used and make minimal comment on the clear safety and efficacy of the current interferon-free regimens. Additionally, they report that while clearing HCV may reduce the risk of decompensated cirrhosis and hepatocellular carcinoma, it does not completely eliminate these risks and therefore treatment may not be of value. This brings us back to the motivation of the CDC, USPSTF, and WHO to recommend screening for HCV – identifying asymptomatic infection to allow for administration of safe, effective antiviral therapies before the development of cirrhosis and all of its complications.
Dr. Sean Koppe is director of hepatology, University of Illinois Hospital & Health Sciences System. He has no conflicts of interest.
The authors question the merits of hepatitis C (HCV) screening despite the endorsement of the Centers for Disease Control and Prevention, U.S. Preventive Services Task Force, and World Health Organization. They suggest HCV does not lead to sufficient mortality, treatment may cause harm, sustained virologic response (SVR) does not represent a ‘cure’ and question if treating HCV truly reduces long term morbidity/mortality. While it is true the majority of patients with HCV will not develop cirrhosis or death attributable to their liver disease, the authors downplayed the morbidity and mortality related to HCV and the significant cost to the health care system as well (Ann. Intern. Med. 2012;156:271; Hepatology 2013;57:2164]. The authors also downplayed the importance of sustained viral response (SVR), suggesting that it does not represent a cure, yet neglect to mention several studies demonstrating late recurrence in only 1%-3% of patients who have achieved SVR. Furthermore, the authors describe the “harms of treatment,” yet they largely reference therapies that are no longer used and make minimal comment on the clear safety and efficacy of the current interferon-free regimens. Additionally, they report that while clearing HCV may reduce the risk of decompensated cirrhosis and hepatocellular carcinoma, it does not completely eliminate these risks and therefore treatment may not be of value. This brings us back to the motivation of the CDC, USPSTF, and WHO to recommend screening for HCV – identifying asymptomatic infection to allow for administration of safe, effective antiviral therapies before the development of cirrhosis and all of its complications.
Dr. Sean Koppe is director of hepatology, University of Illinois Hospital & Health Sciences System. He has no conflicts of interest.
The recent advent of new treatments for hepatitis C prompted organizations including the Centers for Disease Control and Prevention, the U.S. Preventive Services Task Force, and the World Health Organization to recommend expanded hepatitis C screening, but such screening may be premature, according to a subject analysis.
Too much uncertainty exists regarding the validity of surrogate markers for treatment efficacy that were used in trials, and evidence regarding clinical outcomes and screening strategies is lacking, according to Dr. Ronald L. Koretz of the University of California, Los Angeles, and his colleagues, who evaluated the current understanding of the incidence and natural course of hepatitis C infection, treatment efficacy, and potential harms of treatment for their analysis.
The best available data suggest that 80%-85% of patients with chronic hepatitis C will die of nonhepatic causes; thus screening could lead to unnecessary treatment. This is important, as safety data for newer drugs are limited, and the existing data suggest a small but concerning rate of serious adverse events associated with the use of some treatments and treatment regimens; the risk-benefit profile of treatment cannot be adequately evaluated because of the lack of data regarding treatment benefits, the investigators reported online Jan. 13 in the British Medical Journal ([doi:10.10036/bmj.g7809]).
Clinical trials to determine the outcomes of treatment in screen-detected patients, as well as the long-term hazards of treatment, are needed, they said, noting that currently available trials included small numbers of patients and/or only short-term follow-up. Until data from such trials are available, physicians should not be pressured to enforce recommended screening strategies “out of enthusiasm for new treatments that have not yet been shown to cause long-term clinical improvement,” they concluded.
Dr. Koretz is a member of the editorial board of the Cochrane Hepato-Biliary Group. The authors reported having no other financial conflicts.
The recent advent of new treatments for hepatitis C prompted organizations including the Centers for Disease Control and Prevention, the U.S. Preventive Services Task Force, and the World Health Organization to recommend expanded hepatitis C screening, but such screening may be premature, according to a subject analysis.
Too much uncertainty exists regarding the validity of surrogate markers for treatment efficacy that were used in trials, and evidence regarding clinical outcomes and screening strategies is lacking, according to Dr. Ronald L. Koretz of the University of California, Los Angeles, and his colleagues, who evaluated the current understanding of the incidence and natural course of hepatitis C infection, treatment efficacy, and potential harms of treatment for their analysis.
The best available data suggest that 80%-85% of patients with chronic hepatitis C will die of nonhepatic causes; thus screening could lead to unnecessary treatment. This is important, as safety data for newer drugs are limited, and the existing data suggest a small but concerning rate of serious adverse events associated with the use of some treatments and treatment regimens; the risk-benefit profile of treatment cannot be adequately evaluated because of the lack of data regarding treatment benefits, the investigators reported online Jan. 13 in the British Medical Journal ([doi:10.10036/bmj.g7809]).
Clinical trials to determine the outcomes of treatment in screen-detected patients, as well as the long-term hazards of treatment, are needed, they said, noting that currently available trials included small numbers of patients and/or only short-term follow-up. Until data from such trials are available, physicians should not be pressured to enforce recommended screening strategies “out of enthusiasm for new treatments that have not yet been shown to cause long-term clinical improvement,” they concluded.
Dr. Koretz is a member of the editorial board of the Cochrane Hepato-Biliary Group. The authors reported having no other financial conflicts.